Abstract
The sunshine vitamin has been associated with reduced risk for many chronic illnesses including cancer and cancer mortality. Epidemiologic and ecological studies have suggested that living at higher latitudes and having lower blood levels of 25-hydroxyvitamin D are associated with increased risk for up to 15 cancers including breast, colon, lung, lymphoma, pancreatic, ovarian and prostate cancer. Most randomized controlled trials using appropriate doses of vitamin D have suggested that improvement in vitamin D status reduces risk for several cancers. Although the exact mechanism by which enhanced vitamin D status reduces risk for cancer is not completely understood, there is evidence that by raising blood levels of 25-hydroxyvitamin D this metabolite can enter a wide variety of cells in the body and then be converted to 1,25-dihydroxyvitamin D3. The vitamin D metabolite, 1,25-dihydroxyvitamin D3, has been demonstrated to markedly reduce cellular proliferation especially of malignant cells that have a vitamin D receptor. It also induces terminal differentiation. 1,25-dihydroxyvitamin D3 is also anti-angiogenic and pro-apoptotic which also plays a role in reducing the growth and spread of malignant cells. Thus improvement in vitamin D status with sensible sun exposure, vitamin D supplementation and ingesting foods containing vitamin D is a reasonable strategy to reduce risk of malignancy.
Keywords: Vitamin D; Cancer; 25-Hydroxyvitamin D; 1,25-Dihydroxyvitamin D; Apoptosis; Anti-angiogenesis; Malignancy
Introduction
For more than a decade research has suggested that there may be an association with sun exposure, vitamin D status and risk for developing a variety of some of the most common cancers. A variety of basic and clinical research activities have provided insights as to how the sunshine vitamin may play a role in reducing risk for deadly cancers including breast and colon cancers. The goal of this review is to give a broad overview of studies and mechanisms related to vitamin D and cancer.
Physiology and metabolism of vitamin D
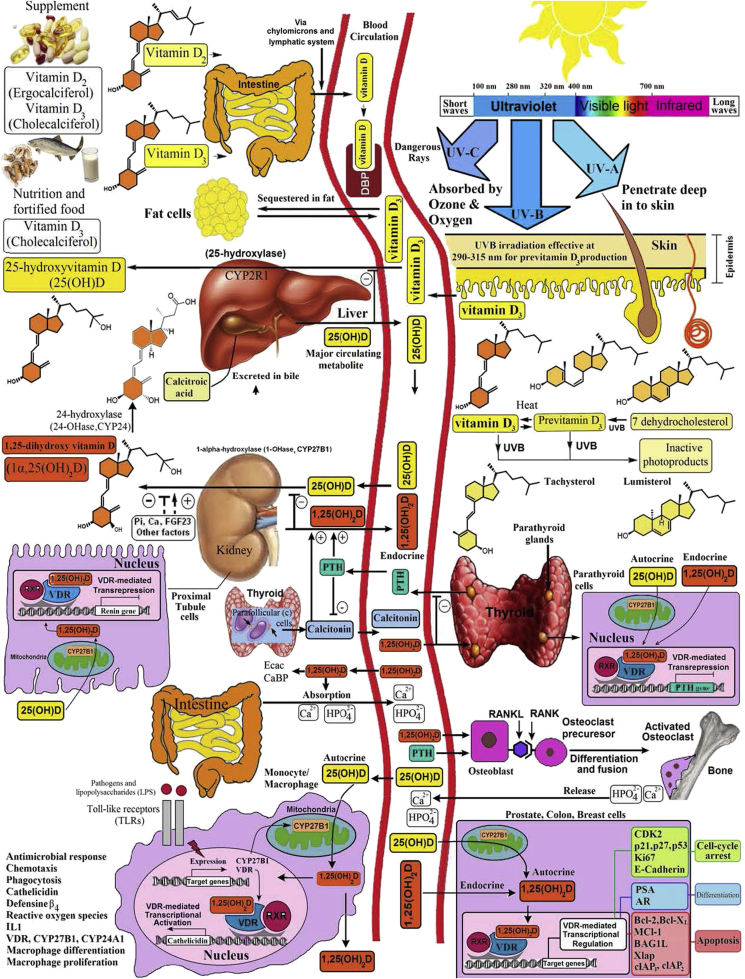
There are two sources of vitamin D and two major forms of vitamin D. During sun exposure the precursor of cholesterol, 7-dehydrocholesterol, in the epidermis and dermis absorbs solar ultraviolet B radiation (290–315 nm) and is converted to previtamin D3 [1]. Once formed previtamin D3 undergoes a rearrangement of its double bonds to form vitamin D3 which then exits the plasma membrane of the skin cells into the extravascular space. It diffuses into the dermal capillary bed for transport to the liver 2, 3 (Fig. 1). Vitamin D3 is also present in oily fish, cod liver oil and fortified foods such as dairy products, margarine, cereals and some juice products as well as in supplements. Vitamin D2 which is produced from its precursor ergosterol and is found in yeast and mushrooms exposed to ultraviolet radiation. It is also used in the fortification of some foods and in supplements and pharmaceutical vitamin D preparations [2]. Both vitamin D2 and vitamin D3 (D represents either) when ingested is incorporated into the chylomicrons and is transported from the lymphatic system into the venous blood stream for transport to the liver. In the liver, vitamin D is converted to its major circulating form 25-hydroxyvitamin D [25(OH)D]. 25(OH)D bound to its vitamin D binding protein (DBP) is transported to the kidneys where it is converted to its active form, 1,25-dihydroxyvitamin D [1,25(OH)2D]. 1,25(OH)2D exits the kidneys and is transported to the small intestine where it interacts with its nuclear vitamin D receptor (VDR) and the retinoic acid X receptor (RxR) to form a heterodimer complex. This complex interacts with specific vitamin D responsive elements to initiate or inhibit transcription of genes that control intestinal calcium absorption (Fig. 1). 1,25(OH)2D also travels to the skeleton where it binds to the VDR in osteoblasts to induce RANK ligand (RANKL). RANKL interacts with its receptor on monocytes resulting in the transformation into bone resorbing osteoclasts 2, 3 (Fig. 1). Thus the major function of vitamin D is to maintain calcium homeostasis by increasing intestinal calcium transport. When there is inadequate calcium absorption 1,25(OH)2D then mobilizes precious calcium from the skeleton (Fig. 1).
Figure 1.
Schematic representation of the synthesis and metabolism of vitamin D for skeletal and non-skeletal function. During exposure to sunlight, 7-dehydrocholesterol in the skin is converted to previtamin D3. Previtamin D3 immediately converts by a heat-dependent process to vitamin D3. Excessive exposure to sunlight degrades previtamin D3 and vitamin D3 into inactive photoproducts. Vitamin D2 and vitamin D3 from dietary sources are incorporated into chylomicrons, transported by the lymphatic system into the venous circulation. Vitamin D (D represents D2 or D3) made in the skin or ingested in the diet can be stored in and then released from fat cells. Vitamin D in the circulation is bound to the vitamin D-binding protein(DBP), which transports it to the liver, where vitamin D is converted by the vitamin D-25-hydroxylase to 25-hydroxyvitamin D [25(OH)D]. This is the major circulating form of vitamin D that is used by clinicians to measure vitamin D status (although most reference laboratories report the normal range to be 20–100 ng/mL, the preferred healthful range is 30–60 ng/mL). It is biologically inactive and must be converted in the kidneys by the 25-hydroxyvitamin D-1a-hydroxylase (1-OHase) to its biologically active form 1,25-dihydroxyvitamin D [1,25(OH)2D]. 1,25(OH)2D3 is then taken up by target cells and targeted to intracellular D-binding proteins (IDBP) to mitochondrial 24-hydroxylase or to the vitamin D receptor (VDR). The 1,25(OH)2D3-VDR complex heterodimerizes with the retinoic acid receptor (RXR) and binds to specific sequences in the promoter regions of the target gene. The DNA bound heterodimer attracts components of the RNA polymerase II complex and nuclear transcription regulators. Serum phosphorus, calcium fibroblast growth factors (FGF-23), and other factors can either increase or decrease the renal production of 1,25(OH)2D. 1,25(OH)2D feedback regulates its own synthesis and decreases the synthesis and secretion of parathyroid hormone (PTH) in the parathyroid glands. 1,25(OH)2D increases the expression of the 25-hydroxyvitamin D-24-hydroxylase (24-OHase) to catabolize 1,25(OH)2D to the water-soluble, biologically inactive calcitroic acid, which is excreted in the bile. 1,25(OH)2D enhances intestinal calcium absorption in the small intestine by stimulating the expression of the epithelial calcium channel (ECaC) and the calbindin 9K (calcium-binding protein, CaBP). 1,25(OH)2D is recognized by its receptor in osteoblasts, causing an increase in the expression of the receptor activator of the NF-kB ligand (RANKL). Its receptor RANK on the preosteoclast binds RANKL, which induces the preosteoclast to become a mature osteoclast. The mature osteoclast removes calcium and phosphorus from the bone to maintain blood calcium and phosphorus levels. Adequate calcium and phosphorus levels promote the mineralization of the skeleton. Autocrine metabolism of 25(OH)D; when a macrophage or monocyte is stimulated through its toll-like receptor 2/1 (TLR2/1) by an infectious agent such as Mycobacterium tuberculosis or its lipopolysaccharide, the signal up-regulates the expression of VDR and 1-OHase. A 25(OH)D level of 30 ng/mL or higher provides adequate substrate for 1-OHase to convert 25(OH)D to 1,25(OH)2D in mitochondria. 1,25(OH)2D travels to the nucleus, where it increases the expression of cathelicidin, a peptide capable of promoting innate immunity and inducing the destruction of infectious agents such as M. tuberculosis. It is also likely that the 1,25(OH)2D produced in monocytes or macrophages is released to act locally on activated T lymphocytes, which regulate cytokine synthesis, and activated B lymphocytes, which regulate immunoglobulin synthesis. When the 25(OH)D level is approximately 30 ng/mL, the risk of many common cancers is reduced. It is believed that the local production of 1,25(OH)2D in the breast, colon, prostate, and other tissues regulates a variety of genes that control proliferation, including p21 and p27, as well as genes that inhibit angiogenesis and induce differentiation and apoptosis. Once 1,25(OH)2D completes the task of maintaining normal cellular proliferation and differentiation, it induces expression of the enzyme 24-OHase, which enhances the catabolism of 1,25(OH)2D to the biologically inert calcitroic acid. Thus, locally produced (autocrine) 1,25(OH)2D does not enter the circulation and has no influence on calcium metabolism. The parathyroid glands have 1-OHase activity, and the local production of 1,25(OH)2D inhibits the expression and synthesis of parathyroid hormone. The 1,25(OH)2D produced in the kidney enters the circulation and can down-regulate renin production in the kidney and stimulate insulin secretion in the beta islet cells of the pancreas.
Holick copyright 2013 Reproduced with permission.
Association studies linking sun exposure and vitamin D status with cancer risk
In 1916 Hoffman [4] reported that living at higher latitudes was associated with increased risk for mortality from cancer. He compared cancer mortality between 1908 and 1912 and concluded that cancer mortality increased with increasing distance from the equator. 20 years later Peller and Stephenson [5] reported that United States Navy personnel who principally worked outside had a 60% less likelihood of dying from cancer compared to the civilian population who principally worked indoors. In 1941 Apperly [6] made a similar observation when he compared cancer mortality in Americans and Canadians who were in the agricultural business and exposed to a lot of sunlight and observed that cancer mortality significantly declined for farmers who lived in the south compared to the Northeast. Both Apperly and Peller and Stephenson [5] also recognized that those who were most exposed to sunlight had up to an 8 times higher risk of developing nonmelanoma skin cancer which they noted were easy to detect and easy to treat. Apperly [6] had suggested that by developing nonmelanoma skin cancer that this imparted at an immunity to all cancers including those with the highest mortality breast, colon and prostate cancers.
Little attention was paid to these insightful observations until the 1980s when Garland et al. [7] reported a strong positive correlation with reduced risk for colorectal cancer mortality with increased mean daily solar radiation i.e. living at lower latitudes in United States. This initiated an 8 year prospective case-controlled study of adults living in Washington County, MD relating their vitamin D status i.e. serum 25(OH)D with their risk of developing colon cancer. They observed that for those who had at the initiation of the trial a 25(OH)D > 20 ng/mL had a 3 fold decreased risk for developing colon cancer [8]. Thus Garland et al. [8] made the connection with living at higher latitudes and having less sun exposure and less vitamin D production and was associated with a lower blood level of 25(OH)D with increased risk for colon cancer.
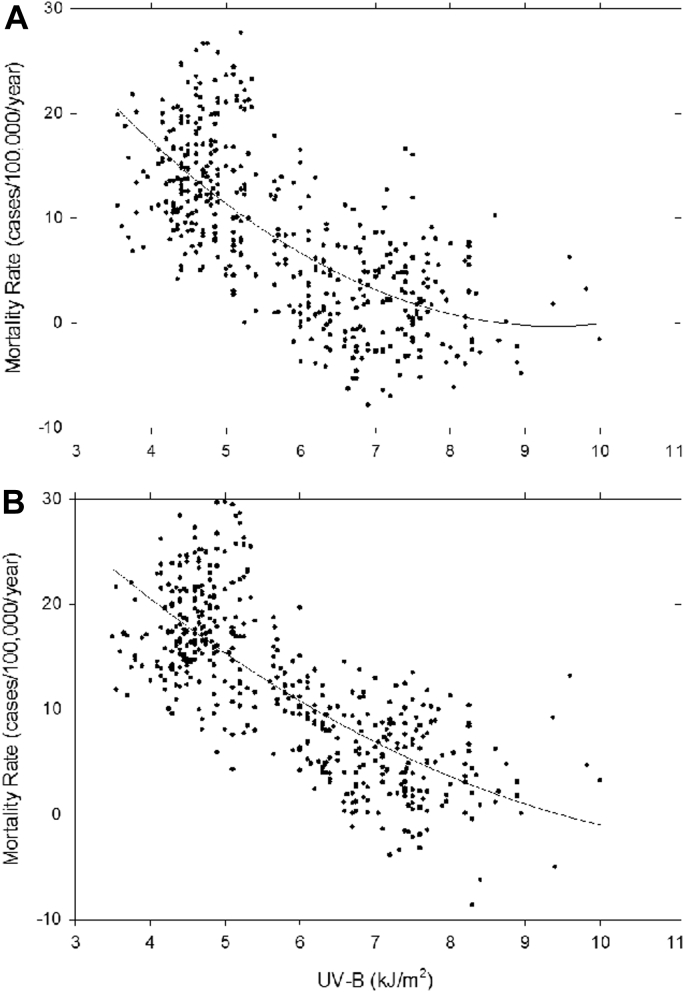
Since these initial insightful observations a multitude of epidemiologic studies have reported an association with decreased sun exposure or lower vitamin D status with increased risk for a variety of cancers including breast, colon, lymphoma, lung and prostate cancers 9, 10, 11. Grant [12] evaluated mortality due to cancer and related it to UV exposure in both men and women. He found a dramatic inverse relationship (Fig. 2) confirming the earlier studies of Apperly [6] and Peller and Stephenson [5]. Grant 12, 13, 14 also reported that more than 13 cancers were reduced by adequate exposure to solar UVB radiation. He calculated that in the span of 24 years (1970–1994) that 566,400 Americans died of cancer because of inadequate exposure to solar UVB radiation. He also concluded that approximately 50,000–63,000 Americans and 1900–25,000 British citizens died prematurely of cancer each year due to vitamin D deficiency 13, 14. His conclusions are consistent with the recent meta-analysis by Keum and Giovannucci [15] who concluded that several lines of evidence suggests that the effects of vitamin D may be stronger for cancer mortality than for incidence. Indeed Maalmi and et al. [16]. reported a meta-analysis of prospective cohort studies and concluded among colorectal cancer patients pooled hazard ratios (95% confidence intervals) comparing highest and lowest 25(OH)D levels were 0.71 (0.55–0.91) and 0.65 (0.49–0.86) for overall and disease-specific mortality respectively. For breast cancer patients the corresponding pooled estimates were 0.62 (0.49–0.78) and 0.58 (0.38–0.84) respectively. It was concluded that a 25(OH)D > 30 ng/mL was associated with this significantly reduced mortality in patients with colorectal and breast cancer.
Figure 2.
Showing the relation of total cancer mortality rates to Smith's Solar Radiation Index in the American states, (white population only).
Reproduced with permission.
Epidemiologic studies have suggested that adequate levels of 25(OH)D are critical for the prevention of various solid tumors including ovarian, breast, colon and prostate cancers. A meta-analysis for the US Preventative Services Task Force regarding vitamin D supplementation concluded that each 4-ng/mL increase in blood 25(OH)D levels was associated with a 6% reduced risk of colorectal cancer [17]. One of the outcome measures for the large Women's Health Initiative Study (WHI) was evaluation for the effect of calcium and vitamin D on reducing risk for colorectal cancer. It was concluded that there was no benefit for ingesting 1000 mg of calcium and 400 IUs of vitamin D daily regarding colon cancer risk [18]. However women who had a baseline level of 25(OH)D < 12 ng/mL and who took the calcium and vitamin D supplement had a 253% increased risk of developing colorectal cancer compared to women who took the same amount of vitamin D3 and calcium for 7 years and had a baseline 25(OH)D level of at least 24 ng/mL [19]. Furthermore a reanalysis of the WHI revealed that 15,646 women (43%) who were not taking personal calcium or vitamin D supplements at the initiation of the trial and who are now randomized to be taking the calcium and vitamin D supplement had a significantly decreased risk of colorectal cancer by 17% and total breast and invasive breast cancers by 14% and 20% [20]. In a randomized controlled trial of osteoporotic women who received 1500 mg of elemental calcium and 1100 IUs of vitamin D3 daily for 4 years had a reduced risk of developing all cancers by 60%. The study was strengthened by a meta-analysis whereby if it was assumed that during the first year some of the women may have already had a developing cancer when the first year was removed from the analysis and even stronger 77% reduction in all cancers was observed [21].
The associations with reduced exposure to solar ultraviolet radiation with increased risk for cancer has been observed in more than 100 countries including Australia, Japan, China where it is been reported that there is an inverse relationship with solar UVB exposure for 15 types of cancer including breast, cervical, colon, endometrial, esophageal, gastric, lung, ovarian, bladder and non-Hodgkin's lymphoma 3, 10, 22, 23. Luscombe et al. [24] reported that men in Finland who worked outdoors had a 3 year hiatus before developing prostate cancer compared to indoor workers. Adults who developed lymphoma had decreased risk for mortality if they had more sun exposure as a teenager [25]. The concept that sensible sun exposure during teenage and young adult years is supported by the study of Knight et al. [26] who reported that women who had the most sun exposure from ages 10–19 years reduced risk of developing breast cancer by more than 60% compared to those with the least sun exposure. This benefit was lost in women who had increased their sun exposure after the age of 40 years.
The vitamin D, sunlight and cancer conundrum
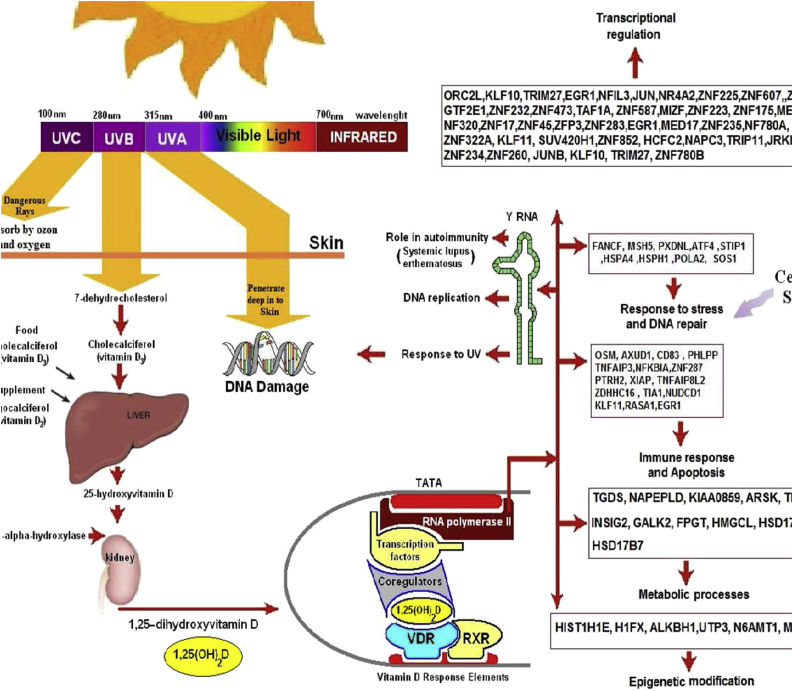
It is well documented that 1,25(OH)2D3 is one of the most potent hormones in decreasing cellular proliferation and inducing terminal differentiation in a variety of normal and malignant cells that contain a VDR 1, 2, 3, 27. Although the exact mechanism by which 1,25(OH)2D3 is able to regulate cellular proliferation and differentiation a large number of genes that control proliferation, differentiation, apoptosis and angiogenosis are either directly or indirectly influenced by 1,25(OH)2D3. 1,25(OH)2D3 increased inhibitors and decreased activators of cyclin-cyclin-dependent kinase complexes in addition to increasing levels of cyclin-dependent kinase inhibitors Cip/Kip proteins P 21 and P 27. These proteins are responsible for keeping the cell cycle in G1/S phase, preventing DNA synthesis and therefore cellular growth 1, 2, 3, 28, 29, 30, 31, 32. Other strategies that 1,25(OH)2D3 uses to help reduce the development of malignancy is to induce apoptosis and to reduce the blood supply to the developing tumor 28, 29, 30, 31, 32, 33, 34 (Fig. 3). A recent study in healthy adults who receives vitamin D supplementation for 4 months reported that 291 genes were significantly influenced many of which control cellular proliferation, maturation, immune function as well as antioxidant activity all that are related to the potential for regulating cellular growth and reducing risk for malignancy [35] (Fig. 3).
Figure 3.
Biological functions for genes whose expression levels were altered after 2 months of vitamin D3 supplementation. After receiving vitamin D3 supplementation we identified 291 genes whose expression was significantly decreased or increased. Some of these genes influence several pathways that are involved in response to stress and DNA repair, DNA replication, immune regulation, epigenetic modification, transcriptional regulation and other biological functions. In addition vitamin D3 supplementation influenced the expression of Y RNA and CETN3 that are involved in DNA repair in response to UVR exposure.
Holick copyright 2013 reproduced with permission.
In the 1980s and 90s numerous publications reported that 1,25(OH)2D3 was a potent inhibitor of a wide variety of cultured cancer cells including breast, colon and prostate cell lines 1, 3, 31. However clinical studies with 1,25(OH)2D3 for treating preleukemia and prostate cancer were unsuccessful in part due to the observed toxicity i.e. hypercalcemia associated with the hormone 36, 37. However the observation that topically applied or orally administered 1,25(OH)2D3 was effective for the treatment of the non-malignant hyperproliferative skin disorder psoriasis demonstrated that this hormone did in fact have potent antiproliferative in pro-differentiating properties that could be used clinically 3, 38.
There is significant scientific evidence to suggest that maintenance of adequate vitamin D status is important for the prevention of a wide variety of deadly cancers 9, 10, 11, 12, 13, 14, 15, 16, 17, 39, 40, 41, 42. Woo et al. [43] reported that men with metastatic prostate cancer who received 2000 IUs of vitamin D3 daily had as much is a 50% reduction in prostate-specific antigen levels after 21 months. Tangpricha et al. [44] observed that mice who were vitamin D deficient and injected with mouse colon cancer cells (MC-26) had 60% greater tumor growth compared to the mice who received the same colon cancer cells but who were vitamin D sufficient throughout the study. Similarly a study in immunodeficient mice revealed that maintaining a normal vitamin D status reduced the growth of a human androgen insensitive prostate cancer (DU-145) [45].
Based on all the evidence it was concluded that the most likely explanation for why improvement in vitamin D status reduced risk of deadly cancers was because the increased circulating concentrations of 25(OH)D could be converted in the kidneys to increased amounts of 1,25(OH)2D which in turn would be released back into the circulation to provide its anti-tumor potential to any cell that was prone to developing a malignancy. However because 1,25(OH)2D is such a potent hormone for regulating calcium homeostasis if there was a significant increase in the circulating level of this hormone it would likely lead to toxicity including hypercalcemia, hyperphosphatemia, soft tissue calcification, nephrocalcinosis and ultimately death [3]. In fact the production of 1,25(OH)2D is tightly regulated in the kidneys by a variety of factors including parathyroid hormone, calcium, phosphate and fibroblast growth factor 23 2, 3 (Fig. 1).
Herein was the conundrum. The epidemiologic, association studies and randomized controlled trials demonstrated that improvement in vitamin D status or UVB exposure reduced risk of deadly cancers. However, the kidneys were not producing more 1,25(OH)2D in response to higher circulating levels of 25(OH)D; how was vitamin D having its effect? It had been previously reported in the 1980s by Bikle et al. [46] that cultured human keratinocytes were capable of converting 25(OH)D to 1,25(OH)2D3. It was also reported that activated macrophages were also able to locally produce 1,25(OH)2D3 which was responsible for the induction of cathelicidin a defensen protein that helps to lyse infective agents engulfed by the macrophage [47]. However these observations although interesting did not seem to have any physiologic significance for cancer reduction until Schwartz et al. [48] reported that normal human prostate cells also had capacity to convert 25(OH)D to 1,25(OH)2D. This observation was confirmed in a variety of normal and malignant cell lines including those of breast, colon, lung and melanocytes 47, 48. Based on these observations it was concluded that the likely explanation for why improvement in vitamin D status was associated with reduced risk for malignancies was due to increased circulating 25(OH)D which could enter cells throughout the body and be converted to 1,25(OH)2D. 1,25(OH)2D in turn controlled wide variety of genes many of which maintains cellular health and reduced risk for malignant transformation 1, 2, 3, 28, 48, 49. Thus vitamin D has 2 functions the first and foremost an endocrine function to maintain calcium homeostasis which is critically important for not only skeletal health but for a variety of metabolic processes (Fig. 1). The second and equally important function is for vitamin D to function in an autocrine/paracrine function whereby 25(OH)D is converted to 1,25(OH)2D in non-calcium regulating cells. Once formed this hormone not only helps to regulate cellular growth and a variety of other physiologic functions but also induces the 25-hydroxyvitamin D-24 hydroxylase (CYP24A1) [27]. This enzyme rapidly destroys 1,25(OH)2D into a water-soluble calcitroic acid thus preventing this calcium active hormone from entering into the circulation to have any influence on calcium metabolism potentially causing toxicity 1, 2, 3 (Fig. 1).
The importance of maintaining a healthy vitamin D status to reduce risk of cancer
To date that has been very disappointing that neither 1,25(OH)2D3 or its active analogs have been proven to be effective in the treatment of any cancer. There are likely several reasons for this separate from the potential calcemic toxicity associated with 1,25(OH)2D3 and its active analogs. The simplest strategy is for a cell that is malignant to markedly increase the expression of CYP24A1 which would rapidly destroy any 1,25(OH)2D3 that enters its domain preventing it from docking in the nucleus to regulate genes to control and destroy the malignant cell. Human prostate cancer cells have been reported to have robust expression of this 1,25(OH)2D3 destructive enzyme [50].
Another clever strategy that malignant cells have developed to mitigate 1,25(OH)2D3's antitumor activity is to express the transcription factor SNAIL [51]. This transcription factor is a zinc-finger transcription factor that is involved in cell movement and exists in both invertebrates and vertebrates. The over expression of this gene in the human colon cancer cell line (SW 480-ADH) resulted in decreasing the expression of VDR thus preventing 1,25(OH)2D3 from docking with its nuclear receptor. The over expression of SNAIL 1 induced epithelial-to-mesenchymal transition and also inhibited E-cadherin which is important for cellular adhesion and differentiation [51]. It was concluded that the ratio of VDR and SNAIL expression in cells may be critical for E-cadherin expression which ultimately influences cellular growth and thus colon cancer progression [51].
Thus based on the autocrine/paracrine hypothesis, the ability of many cells and organs not related to calcium metabolism being able to locally produce 1,25(OH)2D3 is the impetus for increasing circulating concentrations of 25(OH)D in order to reduce risk of developing deadly cancers and the mortality associated with them.
Approaches for raising blood levels of 25-hydroxyvitamin D
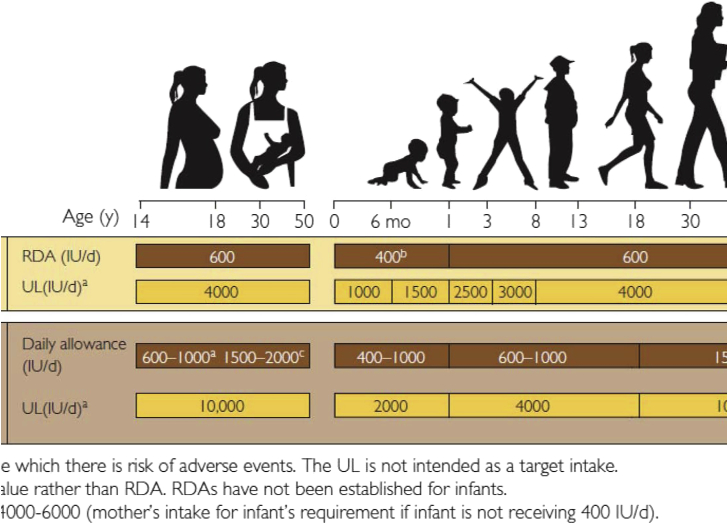
The Institute of Medicine (IOM) and the Endocrine Society have both concluded that vitamin D deficiency should be defined as a serum 25(OH)D > 20 ng/mL [52] (Fig. 4). The IOM suggests that a 25(OH)D > 20 ng/mL is all that is required to maximize bone health and they ignored the association studies concluding that there is no benefit of improving vitamin D status for other health outcomes measures including reducing risk of cancer and cancer mortality [52]. The Endocrine Society concluded based on all of the available literature that a minimum serum level of 25(OH)D to maximize bone health was 30 ng/mL [53]. Furthermore, the Endocrine Society guidelines committee reviewed many of the epidemiologic studies and concluded that there may be additional health benefits that require further investigation including reducing risk of cancers. To achieve a blood level of at least 20 ng/mL the IOM recommended 400 IUs daily for children under one year of age, 600 IUs daily for children one year and older and all adults up to the age of 70 years and 800 IUs daily for adults over the age of 70. The Endocrine Society recommends children under one year of age require 400–1000 IUs daily, children one year and older 600–1000 IUs daily and all adults 1500–2000 IUs daily to sustain a circulating serum level of 25(OH)D > 30 ng/mL (Fig. 4). They also recognized that obese children and adults required 2–3 times more vitamin D to treat and prevent vitamin D deficiency. To treat vitamin D deficiency various strategies can be employed the most common is to give 50,000 IUs of vitamin D2 (the pharmaceutical form of vitamin D available in United States) once a week for 8 weeks which is equivalent to ingesting approximately 6000 IUs daily which can be equally effective. To prevent recurrence it was recommended that 50,000 IUs of vitamin D2 once every 2 weeks which is equivalent to approximately 3000 IUs daily is effective [54]. This strategy has been successfully employed for up to 6 years without any untoward toxicity [55]. Additional sources of vitamin D which can be variable can be obtained from dietary sources and sensible sun exposure. The use of the mobile phone app Dminder.info provides guidelines anywhere on the globe at any time of the year for how much sun exposure is required to make sufficient vitamin D while at the same time alerting the user after the sensible sun exposure to either wear sun protection or get out of the sun to prevent the damaging effects from excessive exposure to sunlight.
Figure 4.
Vitamin D intakes recommended by the Institute of Medicine and the Endocrine Practice Guidelines Committee. 25(OH) D = 25-hydroxyvitamin D; AI = adequate intake; RDA = recommended dietary allowance; SE = standard error; UL = tolerableupper intake level.
Copyright Holick 2013, reproduced with permission.
Vitamin D toxicity
Vitamin D toxicity is extremely uncommon. Studies have shown in adults that 10,000 IUs daily for at least 5 months did not cause any toxicity [56]. Even in pregnant women who talk 4000 IUs daily throughout her pregnancy raise her blood levels of 25(OH)D to approximately 60 ng/mL without any evidence of toxicity [57]. Vitamin D intoxication which is associated with hypercalcemia hypercalciuria hypophosphatemia soft tissue calcification and nephrocalcinosis is usually observed when blood levels of 25(OH)D > 300 ng/mL for a prolonged period of time [2].
There are however are a few exceptions. Patients with granulomatous disorders such as sarcoidosis the to be more careful about her vitamin D intake due to the extra renal production of 1,25(OH)2D.Toxicity is usually observed when blood levels of 25(OH)D greater than 30 ng/mL. Some patients with Hodgkin's and non-Hodgkin's lymphoma also have invasion of macrophages into their lymphoma. These activated macrophages like those in granulomatous disorders in the non-regulated fashion produce 1,25(OH)2D which can exit the macrophage into the circulation causing hypercalciuria and hypercalcemia [53].
Conclusion
There are now tens of thousands of publications that suggest that improvement in vitamin D status not only is important for musculoskeletal health but for overall health and well-being by reducing risk of both acute and chronic illnesses 1, 2, 3. This is especially true for the association of improved vitamin D status with decreased risk for developing many cancers and cancer mortality. Patients with cancer who are undergoing chemotherapy are at especially high risk for vitamin D deficiency. They often are advised to avoid all sun exposure because of photosensitivity to their medications and they often have a lot of GI distress leading to a decrease in nutrient intake including vitamin D. Patients on chemotherapy often complain of muscle weakness and aches and pains in her bones and muscles which are classic signs for vitamin D deficiency and osteomalacia. These patients should be monitored carefully and receive appropriate treatment for the prevention and treatment of vitamin D deficiency. It has been speculated that improvement in vitamin D status may help improve cancer therapeutic outcomes although there are no control trials to support the speculation. There is however no downside to increasing everyone's vitamin D status. There is enough ecological, association and meta-analyses studies supporting the role of vitamin D in reducing risk of deadly malignancies to warrant the recommendation for everyone to improve their vitamin D status with increasing ingestion of foods fortified with vitamin D, obtaining sensible sun exposure when appropriate and taking a pharmaceutical or supplemental form of vitamin D to maintain a circulating level of 25(OH)D of at least 30 ng/mL with a preferred range of 40–60 ng/mL with up to 100 ng/mL being perfectly safe.
Footnotes
This is an open access article under the CC BY-NC-ND license (http://creativecommons.org/licenses/by-nc-nd/3.0/).
References
- 1.Wacker M., Holick M.F. Sunlight and vitamin D: A global perspective for health. Dermato-endocrinol. 2013;5(1):51–108. doi: 10.4161/derm.24494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Holick M.F. Vitamin D deficiency. N Eng J Med. 2007;357:266–281. doi: 10.1056/NEJMra070553. [DOI] [PubMed] [Google Scholar]
- 3.Hossein-nezhad A., Holick M.F. Vitamin D for Health: a global perspective. Mayo Clin Proc. 2013;88(7):720–755. doi: 10.1016/j.mayocp.2013.05.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hoffman F.L. Prudential Press; 1915. The mortality of cancer throughout the world. Appendix E. [Google Scholar]
- 5.Peller S., Stephenson C.S. Skin irritation and cancer in the United States Navy. Am J Med Sci. 1937;194:326–333. [Google Scholar]
- 6.Apperly F.L. The relation of solar radiation to cancer mortality in North America. Cancer Res. 1941;1:191–195. doi: 10.1158/0008-5472.CAN-15-3169. [DOI] [PubMed] [Google Scholar]
- 7.Garland C.F., Garland F.F. Do sunlight and vitamin D reduce the likelihood of colon cancer? Int J Epidemiol. 1980;9:227–231. doi: 10.1093/ije/9.3.227. [DOI] [PubMed] [Google Scholar]
- 8.Garland C.F., Garland F.C., Shaw E.K., Comstock G.W., Helsing K.J., Gorham E.D. Serum 25-hydroxyvitamin D and colon cancer: eight-year prospective study. Lancet. 1989;18:1176–1178. doi: 10.1016/s0140-6736(89)91789-3. [DOI] [PubMed] [Google Scholar]
- 9.Holick M.F. Vitamin D and sunlight: strategies for cancer prevention and other health benefits. Clin J Am Soc Nephrol. 2008;3(5):1548–1554. doi: 10.2215/CJN.01350308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Freedman D.M., Looker A.C., Abnet C.C., Linet M.S., Graubard B.I. Serum 25-hydroxyvitamin D and cancer mortality in the NHANES III study (1988-2006) Cancer Res. 2010;70(21):8587–8597. doi: 10.1158/0008-5472.CAN-10-1420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Garland C.F., Gorham E.D., Mohr S.B., Grant W.B., Giovannucci E.L., Lipkin M. Vitamin D and prevention of breast cancer: pooled analysis. J Steroid Biochem Mol Biol. 2007;103(3-5):708–711. doi: 10.1016/j.jsbmb.2006.12.007. [DOI] [PubMed] [Google Scholar]
- 12.Grant W.B. An estimate of premature cancer mortality in the U.S. due to inadequate doses of solar ultraviolet-B radiation. Cancer. 2002;94:1867. doi: 10.1002/cncr.10427. [DOI] [PubMed] [Google Scholar]
- 13.Grant W.B. An estimate of the global reduction in mortality rates through doubling vitamin D levels. Eur J Clin Nutr. 2011;65(9):1016–1026. doi: 10.1038/ejcn.2011.68. [DOI] [PubMed] [Google Scholar]
- 14.Grant W.B. Lower vitamin-D production from solar ultraviolet-B irradiance may explain some differences in cancer survival rates. J Natl Med Assoc. 2006;98(3):357–364. [PMC free article] [PubMed] [Google Scholar]
- 15.Keum N., Giovannucci E. Vitamin D supplements and cancer incidence and mortality: A meta-analysis. Br J Cancer. 2014;294:1–5. doi: 10.1038/bjc.2014.294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Maalmi H., Ordonez-Mena J.M., Schottker B., Brenner H. Serum 25-hydroxyvitamin D levels and survival in colorectal and breast cancer patients: Systematic review and meta-analysis of prospective cohort studies. Eur J Cancer. 2014;50:1510–1521. doi: 10.1016/j.ejca.2014.02.006. [DOI] [PubMed] [Google Scholar]
- 17.Chung M., Lee J., Terasawa T., Lau J., Trikalinos T.A. Vitamin D with or without calcium supplementation for prevention of cancer and fractures: an updated meta-analysis for the U.S. Preventive Services Task Force. Ann Intern Med. 2011;155(12):827–838. doi: 10.7326/0003-4819-155-12-201112200-00005. [DOI] [PubMed] [Google Scholar]
- 18.Wactawski-Wende J., Kotchen J.M., Anderson G.L., Assaf A.R., Brunner R.L., O'Sullivan M.J. Calcium plus vitamin D supplementation and the risk of colorectal cancer. N Engl J Med. 2006;354(7):684–696. doi: 10.1056/NEJMoa055222. [DOI] [PubMed] [Google Scholar]
- 19.Holick M.F. Calcium plus vitamin D and the risk of colorectal cancer. N Engl J Med. 2006;354(21):2287–2288. doi: 10.1056/NEJMc060753. [DOI] [PubMed] [Google Scholar]
- 20.Bolland M.J., Grey A., Gamble G.D., Reid I.R. Calcium and vitamin D supplements and health outcomes: a reanalysis of the Women's Health Initiative (WHI) limited access data set. Am J Clin Nutr. 2011;94(4):1144–1149. doi: 10.3945/ajcn.111.015032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lappe J.M., Travers-Gustafson D., Davies K.M., Recker R.R., Heaney R.P. Vitamin D and calcium supplementation reduces cancer risk: results of a randomized trial. Am J Clin Nutr. 2007;85(6):1586–1591. doi: 10.1093/ajcn/85.6.1586. [DOI] [PubMed] [Google Scholar]
- 22.Boscoe F.P., Schymura M.J. Solar ultraviolet-B exposure and cancer incidence and mortality in the United States, 1993-2002. BMC Cancer. 2006;6:264. doi: 10.1186/1471-2407-6-264. http://dx.doi.org/10.1186/1471-2407-6-264 PMID:17096841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Grant W.B. Ecological studies of the UVB-vitamin D-cancer hypothesis. Anticancer Res. 2012;32:223–236. PMID:22213311. [PubMed] [Google Scholar]
- 24.Luscombe C.J., Fryer A.A., French M.E., Liu S., Saxby M.F., Jones P.W. Exposure to ultraviolet radiation: association with susceptibility and age at presentation with prostate cancer. Lancet. 2001;358:641–642. doi: 10.1016/S0140-6736(01)05788-9. http://dx.doi.org/10.1016/S0140-6736(01)05788-9 PMID:11530156. [DOI] [PubMed] [Google Scholar]
- 25.Chang E.T., Smedby K.E., Hjalgrim H., Porwit-MacDonald A., Roos G., Glimelius B. Family history of hematopoietic malignancy and risk of lymphoma. J Natl Cancer Inst. 2005;97:1466–1474. doi: 10.1093/jnci/dji293. http://dx.doi.org/10.1093/jnci/dji293 PMID:16204696. [DOI] [PubMed] [Google Scholar]
- 26.Knight J.A., Lesosky M., Barnett H., Raboud J.M., Vieth R. Vitamin D and reduced risk of breast cancer: a population-based case-control study. Cancer Epidemiol Biomarkers Prev. 2007;16:422–429. doi: 10.1158/1055-9965.EPI-06-0865. http://dx.doi.org/10.1158/1055-9965.EPI-06-0865 PMID:17372236. [DOI] [PubMed] [Google Scholar]
- 27.Jones G. Extrarenal vitamin D activation and interactions between vitamin D2, vitamin D3, and vitamin D analogs. Annu Rev Nutr. 2013;33:23–44. doi: 10.1146/annurev-nutr-071812-161203. [DOI] [PubMed] [Google Scholar]
- 28.Spina C.S., Tangpricha V., Uskokovic M., Adorinic L., Maehr H., Holick M.F. Vitamin D and cancer. Anticancer Res. 2006;26:2515–2524. [PubMed] [Google Scholar]
- 29.Krishnam A.V., Peehl D.M., Feldman D. The role of vitamin D in prostate cancer: recent results. Cancer Res. 2003;164:205–221. doi: 10.1007/978-3-642-55580-0_15. [DOI] [PubMed] [Google Scholar]
- 30.Bernardi R.J., Johnson C.S., Modzelewski R.A., Trump D.L. Antiproliferative effects of 1_, 25-dihydroxyvitamin D3 and vitamin D analogs on tumor-derived endothelial cells. Endocrinology. 2002;143:2508–2514. doi: 10.1210/endo.143.7.8887. [DOI] [PubMed] [Google Scholar]
- 31.Swami S., Raghavachari M., Muller U.R., Bao Y.P., Feldman D. Vitamin D growth inhibition of breast cancer cells: Gene expression patterns expressed by cDNA microarray. Breast Cancer Res Treat. 2003;80:49–62. doi: 10.1023/A:1024487118457. [DOI] [PubMed] [Google Scholar]
- 32.Cross H.S., Kallay E., Lechner D., Gerdenitsch W., Aldercreutz H., Armbrecht H.J. Phytoestrogens and vitamin D metabolism: A new concept for the prevention and therapy of colorectal, prostate and mammary carcinoma. J Nutr. 2004;134:1207S–1212S. doi: 10.1093/jn/134.5.1207S. [DOI] [PubMed] [Google Scholar]
- 33.Zhao X., Feldman D. Regulation of vitamin D receptor abundance and responsiveness during differentiation of HT-29 human colon cancer cells. Endocrinology. 1993;132:1808–1813. doi: 10.1210/endo.132.4.8384998. [DOI] [PubMed] [Google Scholar]
- 34.Mantell D.J., Owens P.E., Bundred N.J., Mawer E.B., Canfield A.E. 1,25-Dihydroxyvitamin D3 inhibits angiogenesis in vitro and in vivo. Circ Res. 2000;87:214–220. doi: 10.1161/01.res.87.3.214. [DOI] [PubMed] [Google Scholar]
- 35.Hossein-nezhad A., Spira A., Holick M.F. Influence of vitamin D status and vitamin D3 supplementation on genome wide expression of White blood cells: a randomized double-blind clinical trial. PLoS One. 2013;8(3):e58725. doi: 10.1371/journal.pone.0058725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Koeffler H.P., Hirjik J., Iti L., the Southern California Leukemia Group 1,25-Dihydroxyvitamin D3: In vivo and in vitro effects on human preleukemic and leukemic cells. Cancer Treat Rep. 1985;69:1399–1407. [PubMed] [Google Scholar]
- 37.Beer T.M., Ryan C.W., Venner P.M., Petrylak D.P., Chatta G.S., Ruether J.D. Double-blinded randomized study of high-dose calcitriol plus docetaxel compared with placebo plus docetaxel in androgen-independent prostate cancer: A report from the ASCENT investigators. J Clin Oncol. 2007;25:669–674. doi: 10.1200/JCO.2006.06.8197. [DOI] [PubMed] [Google Scholar]
- 38.Perez A., Chen T.C., Turner A., Raab R., Bhawan J., Poche P. Efficacy and safety of topical calcitriol (1,25- dihydroxyvitamin D3) for the treatment of psoriasis. Br J Dermatol. 1996;134:238–246. [PubMed] [Google Scholar]
- 39.Gorham E.D., Garland C.F., Garland F.C., Grant W.B., Mohr S.B., Lipkin M. Optimal vitamin D status for colorectal cancer prevention: A quantitative meta-analysis. Am J Prev Med. 2007;32:210–216. doi: 10.1016/j.amepre.2006.11.004. [DOI] [PubMed] [Google Scholar]
- 40.Shui I.M., Mucci L.A., Kraft P., Tamimi R.M., Lindstrom S., Penney K.L. Vitamin D-related genetic variation, plasma vitamin D, and risk of lethal prostate cancer: a prospective nested case-control study. J Natl Cancer Inst. 2012;104(9):690–699. doi: 10.1093/jnci/djs189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Buttigliero C., Monagheddu C., Petroni P., Saini A., Dogliotti L., Ciccone G. Prognostic role of vitamin D status and efficacy of vitamin D supplementation in cancer patients: a systematic review. Oncologist. 2011;16(9):1215–1227. doi: 10.1634/theoncologist.2011-0098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Stolzenberg-Solomon R.Z., Hayes R.B., Horst R.L., Anderson K.E., Hollis B.W., Silverman D.T. Serum vitamin D and risk of pancreatic cancer in the prostate, lung, colorectal, and ovarian screening trial. Cancer Res. 2009;69(4):1439–1447. doi: 10.1158/0008-5472.CAN-08-2694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Woo T.C., Choo R., Jamieson M., Chander S., Vieth R. Pilot study: potential role of vitamin D (cholecalciferol) in patients with PSA relapse after definitive therapy. Nutr Cancer. 2005;51:32–36. doi: 10.1207/s15327914nc5101_5. [DOI] [PubMed] [Google Scholar]
- 44.Tangpricha V., Spina C., Yao M., Chen T.C., Wolfe M.M., Holick M.F. Vitamin D deficiency enhances the growth of MC-26 colon cancer xenografts in Balb/c mice. J Nutr. 2005;135:2350–2354. doi: 10.1093/jn/135.10.2350. [DOI] [PubMed] [Google Scholar]
- 45.Ray R., Banks M., Abuzahra H., Eddy V., Persons K., Lucia M.S. Effect of dietary vitamin D and calcium on the growth of androgen-insensitive human prostate tumor in a murine model. Anticancer Res. 2012;32:727–732. [PMC free article] [PubMed] [Google Scholar]
- 46.Bikle D.D. Vitamin D: role in skin and hair. In: Feldman D., Pike J.W., Glorieux F.H., editors. Vitamin D. Elsevier Academic Press; Boston, MA: 2005. pp. 609–630. [Google Scholar]
- 47.Adams J.S., Hewison M. Update in Vitamin D. J Clin Endocrinol Metab. 2010;95(2):471–478. doi: 10.1210/jc.2009-1773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Schwartz G.G., Whitlatch L.W., Chen T.C., Lokeshwar B.L., Holick M.F. Human prostate cells synthesize 1,25-dihydroxyvitamin D3 from 25-hydroxyvitamin D3. Cancer Epidemiol Biomarkers Prev. 1998;7:391–395. [PubMed] [Google Scholar]
- 49.Hummel D., Fetahu I.S., Groschel C., Manhardt T., Kally E. Role of proinflammatory cytokines on expression of vitamin D metabolism and target genes in colon cancer cells. J Steroid Biochem Mol Biol. 2014;144:91–95. doi: 10.1016/j.jsbmb.2013.09.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Wang T.T., Tavera-Mendoza L.E., Laperriere D., Libby E., MacLeod N.B., Nagai Y. Large-scale in silico and microarray-based identification of direct 1,25-dihydroxyvitamin D3 target genes. Mol Endocrinol. 2005;19(11):2685–2695. doi: 10.1210/me.2005-0106. [DOI] [PubMed] [Google Scholar]
- 51.Palmer H.G., Larriba M.J., Garcia J.M., Ordonez-Moran P., Pena C., Peiro S. The transcription factor SNAIL represses vitamin D receptor expression and responsiveness in human colon cancer. Nat Med. 2004;10:917–919. doi: 10.1038/nm1095. [DOI] [PubMed] [Google Scholar]
- 52.Ross A.C., Manson J.E., Abrams S.A., Aloia J.F., Brannon P.M., Clinton S.K. The 2011 report on dietary reference intakes for calcium and vitamin D from the Institute of Medicine: what clinicians need to know. J Clin Endocrinol Metab. 2011;96(1):53–58. doi: 10.1210/jc.2010-2704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Holick M.F., Binkley N.C., Bischoff-Ferrari H.A., Gordon C.M., Hanley D.A., Heaney R.P. Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(7):1911–1930. doi: 10.1210/jc.2011-0385. [DOI] [PubMed] [Google Scholar]
- 54.Malabanan A., Veronikis I.E., Holick M.F. Redefining vitamin D insufficiency. Lancet. 1998;351:805–806. doi: 10.1016/s0140-6736(05)78933-9. [DOI] [PubMed] [Google Scholar]
- 55.Pietras S.M., Obayan B.K., Cai M.H., Holick M.F. Vitamin D2 treatment for vitamin D deficiency and insufficiency for up to 6 years. Arch Intern Med. 2009;169:1806–1808. doi: 10.1001/archinternmed.2009.361. [DOI] [PubMed] [Google Scholar]
- 56.Heaney R.P., Davies K.M., Chen T.C., Holick M.F., Barger-Lux M.J. Human serum 25-hydroxycholecalciferol response to extended oral dosing with cholecalciferol. Am J Clin Nutr. 2003;77:204–210. doi: 10.1093/ajcn/77.1.204. [DOI] [PubMed] [Google Scholar]
- 57.Hollis B.W. Vitamin D requirement during pregnancy and lactation. J Bone Miner Res. 2007;22:V39–V44. doi: 10.1359/jbmr.07s215. [DOI] [PubMed] [Google Scholar]