Abstract
Carbon monoxide (CO) is a gaseous molecule produced from heme by heme oxygenase (HO). Endogenous CO production occurring at low concentrations is thought to have several useful biological roles. In mammals, especially humans, a proper neurovascular unit comprising endothelial cells, pericytes, astrocytes, microglia, and neurons is essential for the homeostasis and survival of the central nervous system (CNS). In addition, the regeneration of neurovascular systems from neural stem cells and endothelial precursor cells after CNS diseases is responsible for functional repair. This review focused on the possible role of CO/HO in the neurovascular unit in terms of neurogenesis, angiogenesis, and synaptic plasticity, ultimately leading to behavioral changes in CNS diseases. CO/HO may also enhance cellular networks among endothelial cells, pericytes, astrocytes, and neural stem cells. This review highlights the therapeutic effects of CO/HO on CNS diseases involved in neurogenesis, synaptic plasticity, and angiogenesis. Moreover, the cellular mechanisms and interactions by which CO/HO are exploited for disease prevention and their therapeutic applications in traumatic brain injury, Alzheimer’s disease, and stroke are also discussed.
Keywords: Carbon monoxide, Heme oxygenase, Pericytes, Neurovascular unit, Astrocytes, Neural stem cells
OVERVIEW
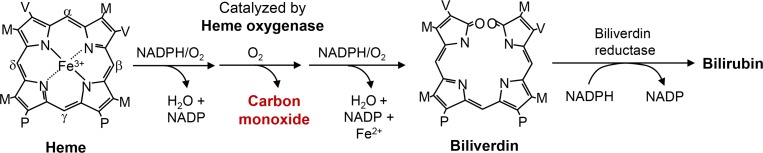
Similar to nitric oxide (NO), produced by a family of nitric oxide synthases (NOSs), carbon monoxide (CO) is also endogenously generated from heme by the catalytic reaction of heme oxygenase (HO) (Fig. 1). CO is endogenously produced, and three isoforms of HO (e.g., HO-1, HO-2, and HO-3) have been identified. These isoforms of HO differ in their tissue distributions and molecular properties. HO-1, induced in response to a broad spectrum of stimuli, was first identified as a stress protein and acted as cytoprotective agents (Wu et al., 2011). The second isoform, HO-2, is constitutively expressed (Trakshel and Maines, 1989). The third isoform, HO-3, is highly homologous to HO-2 but possesses significantly lower catalytic activity. These enzymes, both HO-1 and HO-2, catalyze the oxidative conversion of heme to ferrous iron, CO, and biliverdin. Biliverdin is reduced to bilirubin by biliverdin reductase (reviewed in Kim et al., 2011) (Fig. 1).
Fig. 1.
Production of CO by heme degradation. Heme oxygenase degrades heme to generate carbon monoxide, free iron, and biliverdin. Biliverdin can be rapidly reduced into bilirubin by biliverdin reductase.
HO is expressed in the brain; however, the relative distributions of the three isoforms varies greatly depending on injury and age (Maines, 1997). Inducible HO-1 is not normally detectable in the healthy brains, and its expression is detected in glia and macrophages 24 hours after trauma rat brains (Fukuda et al., 1995). HO-1 is also induced in astrocytes of the peri-infarct regions 48 hours after macular cerebral artery occlusion (MCAO) in the mouse brain (Choi et al., 2017). HO-2 is highly expressed in brain cells, astrocytes, cerebral endothelial cells (ECs) and neurons (Scapagnini et al., 2002; Parfenova et al., 2006). HO-2 appears to protect against lipid peroxidation-mediated cell loss and impaired motor recovery after traumatic brain injury (Chang et al., 2003), whereas its deletion exacerbated cerebral hemorrhage-induced brain edema (Wang and Dore, 2008). HO-2 is abundantly expressed in the cerebellum and hippocampus (Ewing and Maines, 1992; Maines, 1997; McCoubrey, 1997). In the hippocampus, HO-2 is highly expressed in hippocampal granule cells and pyramidal neurons of the CA1 and CA3 regions (Verma et al., 1993), suggesting its role in memory function.
The beneficial or detrimental effects of CO may depend on its concentration. Higher than 500 ppm may lead to toxic effects. Rats exposed to CO at 500 ppm showed cardiac function abnormalities via the induction of the late Na+ current (Dallas et al., 2012). CO inhalation of 3 mg/kg for one hour can lead to critical concentrations at the upper limit of 14.3% carboxyhemoglobin in human blood; therefore, clinical trials have used up to 250 ppm (Motterlini and Otterbein, 2010; Peers, 2012). The protective effects of 250 ppm CO in the CNS have been demonstrated in ischemic brain injury such as stroke and traumatic brain injury in animal models (Wang et al., 2011; Choi et al., 2016a).
The development of safe and efficient CO-releasing molecules (CORMs) for therapy, as well as therapeutic use of CO gas in treatment of neurovascular diseases, is of great current interest. These CORMs possess transition metal carriers that requires stringent characterization from a metabolic and toxicological states. Several CORMs have been synthesized as therapeutic agents aimed at delivering controlled amounts of CO to tissues and organs (Motterlini et al., 2002). CORM-A1 (Na2(H3BCO2)) is soluble and stable in water and decomposes to release CO under physiological conditions with slow kinetics (t1/2=21 min) (Motterlini et al., 2003). CORM-2 ([Ru(CO)3Cl2]2) is soluble in dimethyl sulfoxide and releases CO with fast kinetics (t1/2=1 min) (Motterlini et al., 2002). CORM-3 ([Ru(CO)3Cl(glycinate)]) is a water-soluble CO-releasing agent with fast kinetics (t1/2=1 min) (Clark et al., 2003). A newly synthesized CO-releasing agent, CORM-401 ([Mn(CO)4(S2CNMeCH2CO2H)]), contains a manganese metal center and releases up to 3 CO/mole of compound (Fayad-Kobeissi et al., 2016). Most of the beneficial cellular effects of CORMs were demonstrated at concentrations of 10-200 μM (Hettiarachchi et al., 2014; Choi et al., 2016a).
A protective role for low-dose CO in vascular and neuronal systems in the central nervous system (CNS) has been reported, implying its beneficial effects on neurovascular diseases such as traumatic brain injury, Alzheimer’s disease, and stroke (Yabluchanskiy et al., 2012; Hettiarachchi et al., 2014; Choi et al., 2016b). It may be involved in improving angiogenesis, neurogenesis, and memory functions. This review highlights the therapeutic effects of CO/HO on CNS diseases involved in neurogenesis, synaptic plasticity and angiogenesis. Moreover, the cellular mechanisms and interactions by which CO/HO are exploited for therapeutic applications in traumatic brain injury, Alzheimer’s disease, and stroke are also discussed.
THE ROLE OF CO/HO IN THE NEUROVASCULAR UNIT
Brain ECs closely interact with other types of cells, including pericytes, astrocytes, microglia, and neurons, to form a functional ‘neurovascular unit (NVU)’, which maintains proper brain homeostasis. The NVU is a highly dynamic multicellular structure capable of integrating and responding to both neuronal and vascular systems. Miscommunication and malfunction in members of the NVU are important in many neurologic diseases.
CO may provide neuroprotective mechanisms via direct binding with heme proteins or by indirectly inducing HO-1 (Chun and Kim, 2016; Han et al., 2016). HO-1 is a neuroprotective factor that is upregulated by an array of conditions including elevated CO. Bursts of physiological intracellular reactive oxygen species (ROS) levels activates the nuclear factor erythroid 2-related factor 2 (Nrf2) pathway and induces HO-1, leading to CO production (Kim et al., 2012; Jeong et al., 2016; Jin et al., 2016). These results imply that there is positive circuit between the HO-1/CO and ROS/Nrf2 pathways, resulting in the continuous production of CO. CO/CORMs protect cells from cytotoxicity induced by pathological levels of ROS, which may be a crucial factor in the pathogenesis of human ischemic diseases by providing anti-inflammation, anti-apoptosis, and anti-oxidative injury mechanisms. This review focuses on the possible role of CO/HO in neurovascular repair processing in terms of neurogenesis, synaptic plasticity, and angiogenesis in neurologic diseases (e.g., traumatic brain injury, Alzheimer’s disease, and stroke).
The effects of CO/HO on neurogenesis
Cerebral ischemia and neurodegenerative diseases lead to impairment or death of neurons in the CNS. Regeneration of functional neurons from stem cell-based therapies is a promising strategy currently under investigation. Neurogenesis is the process by which neural stem cells (NSCs) generate newly synthesized neurons to replace damaged neurons or maintain brain functions. NO promotes adult neurogenesis after brain damage (Zhang et al., 2001). Similar to NO, low doses of CO may induce endogenous neurogenesis, providing a potential therapeutic strategy for neurodegenerative diseases. In the traumatic brain injury mouse model, 4 mg/kg CORM-3 rescues pericyte-NSC crosstalk, possibly promoting neurorecovery during the chronic phase (Choi et al., 2016a). Inhibition of HO by tin-protoporphyrin significantly blocked CORM-3-mediated neurogenesis and neuroscore improvements after traumatic brain injury, suggesting reciprocal signaling between CO and HO (Choi et al., 2016a). Moreover, inhibition of NOS with L-NAME blocked CORM-3-mediated neurogenesis and migration of NSCs (Choi et al., 2016a), suggesting that CO may activate NOS to boost neurogenesis. In this study, CO could not induce conversion of pericytes into NSCs in in vitro or in vivo models (Choi et al., 2016a); instead, conditioned medium from cultured oxygen-glucose deprived pericytes treated with CORM-3 enhanced the differentiation of NSCs into mature neurons. These findings suggest that no direct pericyte-to-NSC conversion was taking place; instead, the effects on neurogenesis may be indirectly mediated by the ability of CORM-3 to modulate pericyte-NSC crosstalk (Choi et al., 2016a).
In the MCAO mouse models, HO-1−/− mice had fewer positive cells for both the axon migrating marker netrin and 5-Bromo-2′-deoxyuridine (BrdU; a marker for proliferating cells), as compared to vehicle (Nada et al., 2014). In addition, the MCAO mice brains showed upregulation of nicotinamide phosphoribosyltransferase (Nampt) levels compared with levels in the sham mice, and this effect was significantly blocked in the brains of the HO-1+/− mice (Choi et al., 2017). Nampt-transgenic mice showed an increased number of NSCs and improved neural functional recovery compared with those in wild-type mice after MCAO (Zhao et al., 2015), suggesting that the HO-1/Nampt axis may be involved in neuronal recovery after ischemic stroke. For neuronal differentiation, combinatory treatment of CORM-A1 with retinoic acid induced the neuronal marker Tuj1 in the human neuroblastoma SH-SY5Y cell line, human teratocarcinoma NT2 cell line and organotypic hippocampal slice cultures (Almeida et al., 2016). Recently, the beneficial effects of the CORM-2 and HO-1 pathways on the reduction of neuronal cell death (SH-SY5Y cells and hippocampal neurons) against amyloid β peptide toxicity has been reported (Hettiarachchi et al., 2014). More investigation, however, will be necessary to ascertain whether CO/HO can induce neurogenesis to rescue the cognitive functions in Alzheimer’s diseases. Taken together, these findings suggest that the CO/HO pathway may induce neurogenesis in several neurovascular diseases.
The effects of CO/HO on synaptic plasticity
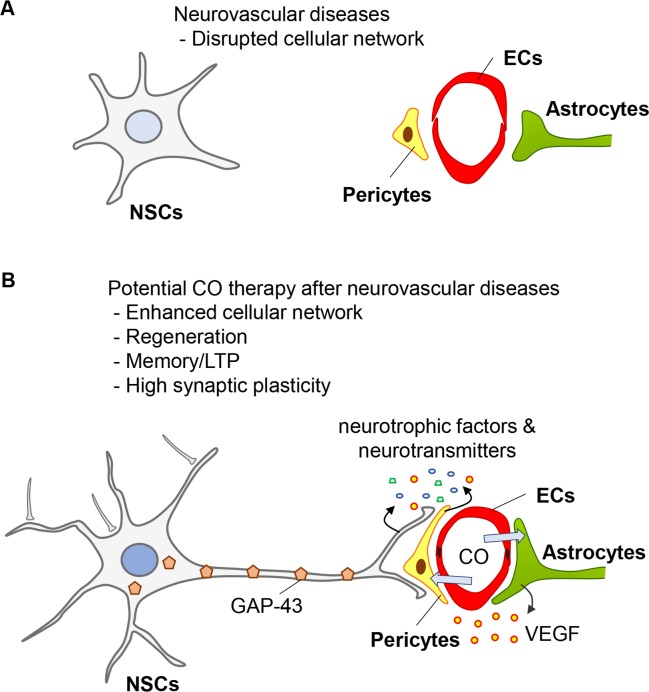
Synapses are the basic structural and functional component for neural communication in the brain. A neuron may receive information via neurotransmitters from hundreds of other neurons connecting thousands of synaptic terminals, as well as from cellular crosstalk. The inputs are highly varied because each transmitting neuron may secrete a different quantity or kind of neurotransmitter in various pathophysiological conditions (Fig. 2A). Endogenous CO production occurring at comparatively low concentrations is thought to have a role at the synapse leading to long-term potentiation (LTP) (Verma et al., 1993; Zhuo et al., 1993). CO can release glutamate from synaptosomes, and has a role in LTP, possibly resulting in memory processing (Shinomura et al., 1994) (Fig. 2B). Poss et al. (1995) noted that although brain HO activity was markedly reduced in HO-2-deficient mice compared to those in wild-type (Poss et al., 1995), both groups demonstrated an equal ability for synaptic potentiation, suggesting that direct CO or HO-1 may be important for synaptic potentiation. In particular, high expression levels of HO-1 in the hippocampus may provide further evidence linking endogenous CO with cognitive processing; however, more investigation is required to determine the role of HO-1 on cognitive functions.
Fig. 2.
The role of CO in neurovascular diseases by enhancing cellular networks. (A) Cellular networks among ECs, pericytes, astrocytes and NSCs are diminished after neurovascular diseases. (B) CO may induce repairing systems by promoting cellular networks in the neurovascular unit, regeneration, LTP/memory functions, and synaptic plasticity.
The growth-associated protein (GAP-43) is associated with presynaptic neuronal outgrowth and neuronal plasticity in general (Snipes et al., 1987). Transgenic overexpression of GAP-43 can result in the formation of new synapses, neurite outgrowth, and synaptogenesis after injury (Routtenberg et al., 2000). The expression of GAP-43 is restricted in neurons, and its expression is stabilized by HuD, a neuronal-specific RNA-binding protein (Mobarak et al., 2000). Recently, we reported that conditioned media from pericytes exposed to CORM-3 during oxygen-glucose deprived conditions induce the production of GAP-43 protein in embryonic and adult rat neural stem cells (Choi et al., 2016a), suggesting that pericyte-NSC crosstalk may induce neuronal regeneration, presynaptic outgrowth and plasticity (Fig. 2B).
The effects of CO/HO on angiogenesis
Blood vessel development is a regulated process involving the proliferation, migration, and remodeling of ECs from adjacent pre-existing blood vessels (angiogenesis) or following differentiation of endothelial progenitor cells (EPCs) from mesodermal precursors (vasculogenesis). Because expression of HO-1 can protect EPCs from oxidative injury and stimulate EPC homing to injured regions to promote angiogenesis, this positive feedback may be beneficial for EPC function (Li et al., 2012). Vascular endothelial growth factor (VEGF), a potent angiogenic and permeability factor, induces pathologic vessel formation in neurovascular diseases; therefore, anti-VEGF (bevacizumab) therapy has been used to prevent vision loss in patients with eye diseases such as diabetic retinopathy and retinopathy of prematurity (Spaide and Fisher, 2006; Mintz-Hittner et al., 2011). Secretion of VEGF by astrocytes is increased in response to low oxygen tension (Stone et al., 1995). In a mouse model of multiple sclerosis, astrocytic VEGF expression was a key driver of blood-brain barrier (BBB) permeability, lymphocyte infiltration and neuropathology in inflammation and demyelinating lesions (Argaw et al., 2012).
Ischemia insults or ROS results in the upregulation of hypoxia-inducible factor-1 alpha (HIF-1α), which regulates several target genes including VEGF, with functions ranging from EC fate decisions to vasculogenesis and angiogenesis (Semenza, 2003). Overexpression of HO-1 or 100 μM CORM-2 stabilizes the HIF-1α protein in astrocytes, leading to up-regulation of VEGF expression (Choi et al., 2010). HIF-1α can also activate the transcription of stromal cell-derived factor 1 (SDF-1) in ECs, resulting in increased adhesion, migration and homing of circulating C-X-C chemokine receptor type 4 (CXCR4, a cognate receptor for SDF-1)-positive progenitor cells to ischemic tissue (Ceradini et al., 2004). Mice exposed to 250 ppm CO gas for 2 h per day had enhanced vasculogenesis after vascular injury through increased circulating EPCs and elevated serum SDF-1 levels (Lin et al., 2009). SDF-1 also promoted angiogenesis via an HO-1-dependent mechanism in EPCs and aortic ECs isolated from wild-type, but not from HO-1−/− mice (Deshane et al., 2007). Similar to CO, NO is a gaseous signaling molecule that plays an important role in homeostatic vascular health. Interestingly, NO induces angiogenesis by upregulating pro-angiogenic mediators, such as HO-1, VEGF and interleukin (IL)-8 (Pae et al., 2005), suggesting NO crosstalk with HO-1/CO for angiogenic pathway regulation. Endothelial nitric oxide synthase (eNOS) is involved in SDF-1/CXCR4-mediated bone marrow stem cell recruitment following injury (Li et al., 2009).
Overexpression of HO-1 or pretreatment of 100 μM CORM-2 followed by recovery stabilizes peroxisome proliferator-activated receptor-γ (PPAR-γ) coactivator-1α (PGC-1α) protein in astrocytes, leading to upregulation of VEGF expression in an HIF-1α-independent manner (Choi et al., 2017). CO stimulates angiogenesis and energy metabolism via the induction of metabolic modulators such as PPAR-γ and PGC-1α (Bilban et al., 2006; Suliman et al., 2007). In addition, CO elicits a mild oxidative stress response that stimulates mitochondrial energy metabolism, PGC-1α protein expression and mitochondrial DNA copy number (Lancel et al., 2009). Adenosine monophosphate protein kinase (AMPK) can enhance sirtuin 1 (SIRT1, an NAD+-dependent class III histone deacetylase) activity by increasing cellular NAD+ levels, resulting in the deacetylation and modulation of downstream SIRT1 targets, including PGC-1α (Canto et al., 2009). Therefore, CO/HO pathway may promote angiogenesis and vasculogenesis.
THE EFFECTS OF CO/HO ON CELLULAR NETWORK IN PATHOLOGIC CONDITIONS
Coordinated regulation of vascular and neuronal systems in the CNS is associated with normal brain functions. Knowledge of the neurovascular crosstalk among endothelial cells, pericytes, astrocytes, and neurons is important to understanding the molecular basis of neurological disease (Lo et al., 2004). Recently, a link between pericyte loss and neurologic disease has gained interest (Winkler et al., 2010). Pericytes in the NVU are a key effector for the regulation of BBB permeability (Armulik et al., 2010; Daneman et al., 2010), cerebral blood flow (Bell et al., 2010), and neuronal morphology and behavior (Bell et al., 2010). Astrocytes are also involved in neurovascular coupling. Astrocytes play a critical role in the maintenance of the BBB through vasoactive endothelial growth factor signaling mechanisms and interactions with extracellular matrix proteins (Lee et al., 2003; Argaw et al., 2012).
There is an abundance of preclinical evidence in both large and small animals demonstrating the beneficial effects of CO, administered either as CO gas or CORMs, in neurodegenerative diseases such as traumatic brain injury, Alzheimer’s disease and stroke (Bauer and Pannen, 2009; Motterlini and Otterbein, 2010; Choi et al., 2016a). The possible therapeutic roles of CO in neurovascular diseases are discussed as follows.
CO/HO in traumatic brain injury
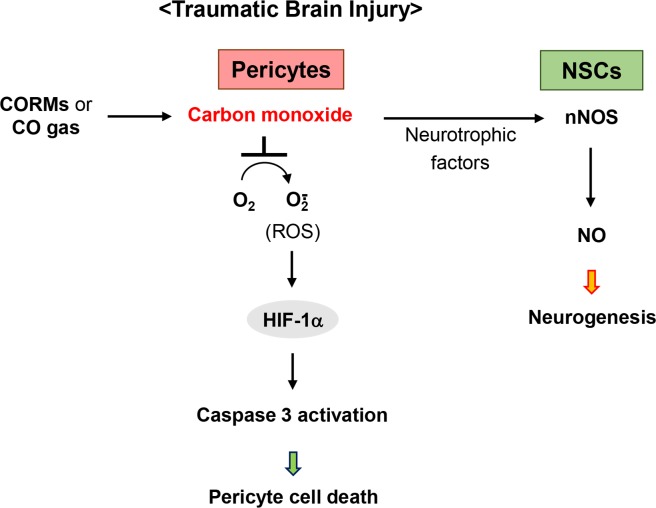
Primary injury is the immediate mechanical and ischemic damage that occurs at the time of the traumatic event, while secondary injury evolves over a period of days to months following the initial injury. Oxidative-nitrative stress is one of the important mechanisms underlying secondary injury following traumatic brain injury (Lynch and Dawson, 1994). Another result of traumatic brain injury is long-term cognitive dysfunction, memory loss and altered motor skills (Xiong et al., 2013). The restoration of high-level cognitive and motor functions requires mature neural circuitry through the induction of angiogenesis, the process by which EPCs may induce new blood vessels to support glucose and oxygen supply. Therefore, therapeutic agents for the treatment of neurodegenerative diseases may require multifunctional mechanisms to reduce oxidative-nitrative stress, and enhance neurorestorative processes, including neurogenesis, synaptogenesis, and angiogenesis, consequently contributing to the recovery of cognitive and motor functions. The beneficial role of CO in traumatic brain injury has been reported, leading to neurogenesis which may be influenced by rescued pericyte-derived factors, consequently activating the neuronal NOS/NO axis (Choi et al., 2016a) (Fig. 3). It is increasingly recognized that pericytes are a critical neurovascular target cell in stroke and neurodegeneration (Yemisci et al., 2009; Winkler et al., 2011). Consistent with these data, widespread pericyte cell death occurs after traumatic brain injury, and the ability to prevent pericyte dropout with CO suggests that its potential role as a neurovascular target. Interestingly, cleaved caspase 3 (apoptotic marker) expression coincides with that of pericyte marker, and ROS/HIF-1α axis may act as the pro-apoptotic initiator for pericyte cell death (Choi et al., 2016a) (Fig. 3). Rescuing pericytes also amplified the endogenous recovery response, suggesting that pericyte-NSC crosstalk may be a mechanism for the regulation of neurogenesis in the recovering brain. These cell-cell interactions reinforce the importance of help-me signaling between multiple cell types in the CNS (Xing and Lo, 2016), and may provide novel opportunities for amplifying endogenous pathways for remodeling injured brain tissue.
Fig. 3.
Mechanisms of CO for repair process in traumatic brain injury. CO, provided from CORM-3 or CO gas, may offer a novel therapeutic approach for traumatic brain injury by preventing pericyte cell death from ROS-dependent signaling, resulting in the activation of redox-sensitive transcription factor HIF-1α, consequently leading to cleaved caspase 3-mediated apoptosis. Moreover, CO therapy may promote pericyte-NSC crosstalk to boost nNOS/NO-mediated neurogenesis.
CO/HO in Alzheimer’s diseases
A form of mental deterioration, or dementia, Alzheimer’s disease is characterized by confusion and memory loss. Its incidence is usually age-related; however, traumatic brain injury and cerebral vascular diseases are factors conferring increased risk for late-onset Alzheimer’s disease (reviewed in Ikonomovic et al., 2017). Alzheimer’s disease results in a characteristic brain pathology: neurons die in large areas of the brain, and brain tissue often shrinks. It is diagnosed based on postmortem findings of two features–neurofibrillary tangles (bundles of tau protein), and senile plaques–in the remaining brain tissue is diagnostic. Senile plaques are aggregates of beta-amyloid, an insoluble peptide of 40 to 42 amino acids that is cleaved from a membrane protein normally present in neurons. Apoptosis contributes to the neuronal loss associated with amyloid-β-peptide toxicity in Alzheimer’s disease. 10 μM CORM-2 protects against amyloid-β-induced toxicity in the human neuroblastoma SH-SY5Y, and in rat hippocampal neurons (Hettiarachchi et al., 2014). In Alzheimer’s disease, molecular signaling is compromised between pericytes and other cell components of the NVU (Winkler et al., 2014) and pericyte loss is accelerated. An association between pericyte loss and the accumulation of soluble Aβ was demonstrated in transgenic APPswe mice (Sagare et al., 2013). Therefore, the link between CO and pericytes requires investigation in the Alzheimer’s disease model.
This perspective is changing with the growing realization that glia actively crosstalk with neurons and influence synaptic development, transmission, and plasticity through an array of secreted and contact-dependent signals. Elevated astrocyte Ca2+ can trigger the release of gliotransmitters, which modulate neuronal activity as well as synaptic transmission and plasticity (Araque, 2008). 100 μM CORM-2 pre-treatment or HO-1 overexpression in astrocytes secretes VEGF, an angiogenic and neurogenic factor, through an L-type Ca2+ channel activation-mediated signaling cascade (Choi et al., 2017). Disruptions in neuron-glia signaling may contribute to synaptic and cognitive impairment in Alzheimer’s disease (Chung et al., 2015). Age results in increased astrocyte expression of glial fibrillary acidic mRNA and a shift to a pro-inflammatory phenotype, as well as signs of oxidative damage and expression of the senescence-associated secretory phenotype (reviewed in Garwood et al., 2017). The possible role of CO/HO in the promotion of cellular network requires further investigation.
CO/HO in stroke
Stroke is a clinical condition in which the blood supply to the brain is limited or severed by embolism and blood clotting, or by the rupture of blood vessels and subsequent cerebral hemorrhage. Ischemic stroke resulting from vascular disorders induces several biochemical and cellular reactions such as increased calcium overload, ROS production, inflammatory response, and impairment of BBB. During reperfusion, ROS concentration peaks, which causes cytotoxicity through lipid peroxidation, protein oxidation, and DNA fragmentation (Nelson et al., 1992; Gursoy-Ozdemir et al., 2004). The potential efficacy of CO therapy in neurovascular diseases may be consistent with past attempts to utilize CO for ischemia-reperfusion injury in other organs (Fujita et al., 2001). Interestingly, 4 mg/kg CORM-3, administered either before or three days after intracerebral hemorrhage, altered the inflammatory response and reduces brain damage in a rat model of hemorrhagic stroke (Yabluchanskiy et al., 2012). Astrocytes isolated from HO-1-deficient animals were more vulnerable to hemoglobin and to hemin toxicity than were wild-type cells (Chen-Roetling et al., 2005; Chen-Roetling and Regan, 2006). HO-1 may be essential for astrocyte resistance to hemoglobin, but it has no protective effect in neurons. As a consequence of the differential effects of HO-1 in neurons and in astrocytes, it is important to selectively induce HO-1 overexpression in astrocytes. An interesting approach has been utilized to selectively express HO-1 in astrocytes using the glial fibrillary acidic protein promoter. Exclusive overexpression of HO-1 in astrocytes, but not in neurons, resulted in reduced hemin-induced cell death (Benvenisti-Zarom and Regan, 2007). HO-1 induction is generally considered an adaptive cytoprotective response against the toxicity of oxidative stress. Overexpression of HO-1 in the mouse brain revealed a reduction in infarct volumes induced by MCAO (Panahian et al., 1999). Challenged with glutamate, an acute excitotoxic amino acid, resulted in reduced viability of primary neuronal cells from HO-1-knockout mice, compared with those of the wild-type mice (Ahmad et al., 2006). Therefore, the CO/HO pathway may be useful for the therapeutic treatment of stroke.
PERSPECTIVES
The various involvements of the CO/HO system have received considerable attention as targets for therapeutic interventions against neurovascular diseases in the CNS. CO/CORMs may offer a novel therapeutic approach for neurovascular diseases by preventing cell death and promoting cellular networks to boost regeneration for functional recovery. The results of this and other studies suggest that CO/CORMs may be a translationally viable and novel approach for CNS disorders. CO/CORMs may modify both aspects of CNS pathophysiology, thus providing proof-of-concept that it is feasible to pursue dual-effect therapies to prevent apoptosis and brain damage during the acute phase of brain injury as well as enhance neurogenic responses to restore cognitive and motor function during the recovery phase.
Acknowledgments
YKC is thankful to Dr. Eng H Lo (Massachusetts General Hospital, Harvard Medical School, USA) for his useful discussion. This research was supported from a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: HI17C2160).
Footnotes
CONFLICT OF INTEREST
There is no conflict of interest.
REFERENCES
- Ahmad AS, Zhuang H, Dore S. Heme oxygenase-1 protects brain from acute excitotoxicity. Neuroscience. 2006;141:1703–1708. doi: 10.1016/j.neuroscience.2006.05.035. [DOI] [PubMed] [Google Scholar]
- Almeida AS, Soares L, Vieira M, Gramsbergen JB, Vieira HL. Carbon Monoxide Releasing Molecule-A1 (CORM-A1) improves neurogenesis: increase of neuronal differentiation yield by preventing cell death. PLoS ONE. 2016;11:e0154781. doi: 10.1371/journal.pone.0154781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Araque A. Astrocytes process synaptic information. Neuron Glia Biol. 2008;4:3–10. doi: 10.1017/S1740925X09000064. [DOI] [PubMed] [Google Scholar]
- Argaw AT, Asp L, Zhang J, Navrazhina K, Pham T, Mariani JN, Mahase S, Dutta DJ, Seto J, Kramer EG, Ferrara N, Sofroniew MV, John GR. Astrocyte-derived VEGF-A drives blood-brain barrier disruption in CNS inflammatory disease. J Clin Invest. 2012;122:2454–2468. doi: 10.1172/JCI60842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Armulik A, Genove G, Mae M, Nisancioglu MH, Wallgard E, Niaudet C, He L, Norlin J, Lindblom P, Strittmatter K, Johansson BR, Betsholtz C. Pericytes regulate the blood-brain barrier. Nature. 2010;468:557–561. doi: 10.1038/nature09522. [DOI] [PubMed] [Google Scholar]
- Bauer I, Pannen BH. Bench-to-bedside review: Carbon monoxide--from mitochondrial poisoning to therapeutic use. Crit Care. 2009;13:220. doi: 10.1186/cc7887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bell RD, Winkler EA, Sagare AP, Singh I, LaRue B, Deane R, Zlokovic BV. Pericytes control key neurovascular functions and neuronal phenotype in the adult brain and during brain aging. Neuron. 2010;68:409–427. doi: 10.1016/j.neuron.2010.09.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benvenisti-Zarom L, Regan RF. Astrocyte-specific heme oxygenase-1 hyperexpression attenuates heme-mediated oxidative injury. Neurobiol Dis. 2007;26:688–695. doi: 10.1016/j.nbd.2007.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bilban M, Bach FH, Otterbein SL, Ifedigbo E, d’Avila JC, Esterbauer H, Chin BY, Usheva A, Robson SC, Wagner O, Otterbein LE. Carbon monoxide orchestrates a protective response through PPARγ. Immunity. 2006;24:601–610. doi: 10.1016/j.immuni.2006.03.012. [DOI] [PubMed] [Google Scholar]
- Canto C, Gerhart-Hines Z, Feige JN, Lagouge M, Noriega L, Milne JC, Elliott PJ, Puigserver P, Auwerx J. AMPK regulates energy expenditure by modulating NAD+ metabolism and SIRT1 activity. Nature. 2009;458:1056–1060. doi: 10.1038/nature07813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ceradini DJ, Kulkarni AR, Callaghan MJ, Tepper OM, Bastidas N, Kleinman ME, Capla JM, Galiano RD, Levine JP, Gurtner GC. Progenitor cell trafficking is regulated by hypoxic gradients through HIF-1 induction of SDF-1. Nat Med. 2004;10:858–864. doi: 10.1038/nm1075. [DOI] [PubMed] [Google Scholar]
- Chang EF, Wong RJ, Vreman HJ, Igarashi T, Galo E, Sharp FR, Stevenson DK, Noble-Haeusslein LJ. Heme oxygenase-2 protects against lipid peroxidation-mediated cell loss and impaired motor recovery after traumatic brain injury. J Neurosci. 2003;23:3689–3696. doi: 10.1523/JNEUROSCI.23-09-03689.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen-Roetling J, Benvenisti-Zarom L, Regan RF. Cultured astrocytes from heme oxygenase-1 knockout mice are more vulnerable to heme-mediated oxidative injury. J Neurosci Res. 2005;82:802–810. doi: 10.1002/jnr.20681. [DOI] [PubMed] [Google Scholar]
- Chen-Roetling J, Regan RF. Effect of heme oxygenase-1 on the vulnerability of astrocytes and neurons to hemoglobin. Biochem Biophys Res Commun. 2006;350:233–237. doi: 10.1016/j.bbrc.2006.09.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choi YK, Kim CK, Lee H, Jeoung D, Ha KS, Kwon YG, Kim KW, Kim YM. Carbon monoxide promotes VEGF expression by increasing HIF-1α protein level via two distinct mechanisms, translational activation and stabilization of HIF-1α protein. J Biol Chem. 2010;285:32116–32125. doi: 10.1074/jbc.M110.131284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choi YK, Kim JH, Lee DK, Lee KS, Won MH, Jeoung D, Lee H, Ha KS, Kwon YG, Kim YM. Carbon monoxide potentiation of L-type Ca2+ channel activity increases HIF-1α-independent VEGF expression via an AMPKα/SIRT1-mediated PGC-1α/ERRα axis. Antioxid Redox Signal. 2017;27:21–36. doi: 10.1089/ars.2016.6684. [DOI] [PubMed] [Google Scholar]
- Choi YK, Maki T, Mandeville ET, Koh SH, Hayakawa K, Arai K, Kim YM, Whalen MJ, Xing C, Wang X, Kim KW, Lo EH. Dual effects of carbon monoxide on pericytes and neurogenesis in traumatic brain injury. Nat Med. 2016a;22:1335–1341. doi: 10.1038/nm.4188. [DOI] [PubMed] [Google Scholar]
- Choi YK, Park JH, Baek YY, Won MH, Jeoung D, Lee H, Ha KS, Kwon YG, Kim YM. Carbon monoxide stimulates astrocytic mitochondrial biogenesis via L-type Ca2+ channel-mediated PGC-1α/ERRα activation. Biochem Biophys Res Commun. 2016b;479:297–304. doi: 10.1016/j.bbrc.2016.09.063. [DOI] [PubMed] [Google Scholar]
- Chun YJ, Kim D. Cancer activation and polymorphisms of human cytochrome 450 1B1. Toxicol Res. 2016;32:89–93. doi: 10.5487/TR.2016.32.2.089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chung WS, Welsh CA, Barres BA, Stevens B. Do glia drive synaptic and cognitive impairment in disease? Nat Neurosci. 2015;18:1539–1545. doi: 10.1038/nn.4142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark JE, Naughton P, Shurey S, Green CJ, Johnson TR, Mann BE, Foresti R, Motterlini R. Cardioprotective actions by a water-soluble carbon monoxide-releasing molecule. Circ Res. 2003;93:e2–e8. doi: 10.1161/01.RES.0000084381.86567.08. [DOI] [PubMed] [Google Scholar]
- Dallas ML, Yang Z, Boyle JP, Boycott HE, Scragg JL, Milligan CJ, Elies J, Duke A, Thireau J, Reboul C, Richard S, Bernus O, Steele DS, Peers C. Carbon monoxide induces cardiac arrhythmia via induction of the late Na+ current. Am J Respir Crit Care Med. 2012;186:648–656. doi: 10.1164/rccm.201204-0688OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Daneman R, Zhou L, Kebede AA, Barres BA. Pericytes are required for blood-brain barrier integrity during embryogenesis. Nature. 2010;468:562–566. doi: 10.1038/nature09513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deshane J, Chen S, Caballero S, Grochot-Przeczek A, Was H, Li Calzi S, Lach R, Hock TD, Chen B, Hill-Kapturczak N, Siegal GP, Dulak J, Jozkowicz A, Grant MB, Agarwal A. Stromal cell-derived factor 1 promotes angiogenesis via a heme oxygenase 1-dependent mechanism. J Exp Med. 2007;204:605–618. doi: 10.1084/jem.20061609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ewing JF, Maines MD. In situ hybridization and immunohistochemical localization of heme oxygenase-2 mRNA and protein in normal rat brain: differential distribution of isozyme 1–2. Mol Cell Neurosci. 1992;3:559–570. doi: 10.1016/1044-7431(92)90068-D. [DOI] [PubMed] [Google Scholar]
- Fayad-Kobeissi S, Ratovonantenaina J, Dabire H, Wilson JL, Rodriguez AM, Berdeaux A, Dubois-Rande JL, Mann BE, Motterlini R, Foresti R. Vascular and angiogenic activities of CORM-401, an oxidant-sensitive CO-releasing molecule. Biochem Pharmacol. 2016;102:64–77. doi: 10.1016/j.bcp.2015.12.014. [DOI] [PubMed] [Google Scholar]
- Fujita T, Toda K, Karimova A, Yan SF, Naka Y, Yet SF, Pinsky DJ. Paradoxical rescue from ischemic lung injury by inhaled carbon monoxide driven by derepression of fibrinolysis. Nat Med. 2001;7:598–604. doi: 10.1038/87929. [DOI] [PubMed] [Google Scholar]
- Fukuda K, Panter SS, Sharp FR, Noble LJ. Induction of heme oxygenase-1 (HO-1) after traumatic brain injury in the rat. Neurosci Lett. 1995;199:127–130. doi: 10.1016/0304-3940(95)12042-3. [DOI] [PubMed] [Google Scholar]
- Garwood CJ, Ratcliffe LE, Simpson JE, Heath PR, Ince PG, Wharton SB. Review: astrocytes in Alzheimer’s disease and other age-associated dementias: a supporting player with a central role. Neuropathol Appl Neurobiol. 2017;43:281–298. doi: 10.1111/nan.12338. [DOI] [PubMed] [Google Scholar]
- Gursoy-Ozdemir Y, Can A, Dalkara T. Reperfusion-induced oxidative/nitrative injury to neurovascular unit after focal cerebral ischemia. Stroke. 2004;35:1449–1453. doi: 10.1161/01.STR.0000126044.83777.f4. [DOI] [PubMed] [Google Scholar]
- Han S, Pham TV, Kim JH, Lim YR, Park HG, Cha GS, Yun CH, Chun YJ, Kang LW, Kim D. Structural analysis of the Streptomyces avermitilis CYP107W1-oligomycin A complex and role of the tryptophan 178 residue. Mol Cells. 2016;39:211–216. doi: 10.14348/molcells.2016.2226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hettiarachchi N, Dallas M, Al-Owais M, Griffiths H, Hooper N, Scragg J, Boyle J, Peers C. Heme oxygenase-1 protects against Alzheimer’s amyloid-β(1–42)-induced toxicity via carbon monoxide production. Cell Death Dis. 2014;5:e1569. doi: 10.1038/cddis.2014.529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ikonomovic MD, Mi Z, Abrahamson EE. Disordered APP metabolism and neurovasculature in trauma and aging: Combined risks for chronic neurodegenerative disorders. Ageing Res Rev. 2017;34:51–63. doi: 10.1016/j.arr.2016.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jeong YH, Park JS, Kim DH, Kim HS. Lonchocarpine increases Nrf2/ARE-mediated antioxidant enzyme expression by modulating AMPK and MAPK signaling in brain astrocytes. Biomol Ther (Seoul) 2016;24:581–588. doi: 10.4062/biomolther.2016.141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin CH, So YK, Han SN, Kim JB. Isoegomaketone upregulates heme oxygenase-1 in RAW264.7 cells via ROS/p38 MAPK/Nrf2 pathway. Biomol Ther (Seoul) 2016;24:510–516. doi: 10.4062/biomolther.2015.194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim JH, Choi YK, Lee KS, Cho DH, Baek YY, Lee DK, Ha KS, Choe J, Won MH, Jeoung D, Lee H, Kwon YG, Kim YM. Functional dissection of Nrf2-dependent phase II genes in vascular inflammation and endotoxic injury using Keap1 siRNA. Free Radic Biol Med. 2012;53:629–640. doi: 10.1016/j.freeradbiomed.2012.04.019. [DOI] [PubMed] [Google Scholar]
- Kim YM, Pae HO, Park JE, Lee YC, Woo JM, Kim NH, Choi YK, Lee BS, Kim SR, Chung HT. Heme oxygenase in the regulation of vascular biology: from molecular mechanisms to therapeutic opportunities. Antioxid Redox Signal. 2011;14:137–167. doi: 10.1089/ars.2010.3153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lancel S, Hassoun SM, Favory R, Decoster B, Motterlini R, Neviere R. Carbon monoxide rescues mice from lethal sepsis by supporting mitochondrial energetic metabolism and activating mitochondrial biogenesis. J Pharmacol Exp Ther. 2009;329:641–648. doi: 10.1124/jpet.108.148049. [DOI] [PubMed] [Google Scholar]
- Lee SW, Kim WJ, Choi YK, Song HS, Son MJ, Gelman IH, Kim YJ, Kim KW. SSeCKS regulates angiogenesis and tight junction formation in blood-brain barrier. Nat Med. 2003;9:900–906. doi: 10.1038/nm889. [DOI] [PubMed] [Google Scholar]
- Li FY, Lam KS, Tse HF, Chen C, Wang Y, Vanhoutte PM, Xu A. Endothelium-selective activation of AMP-activated protein kinase prevents diabetes mellitus-induced impairment in vascular function and reendothelialization via induction of heme oxygenase-1 in mice. Circulation. 2012;126:1267–1277. doi: 10.1161/CIRCULATIONAHA.112.108159. [DOI] [PubMed] [Google Scholar]
- Li N, Lu X, Zhao X, Xiang FL, Xenocostas A, Karmazyn M, Feng Q. Endothelial nitric oxide synthase promotes bone marrow stromal cell migration to the ischemic myocardium via upregulation of stromal cell-derived factor-1α. Stem Cells. 2009;27:961–970. doi: 10.1002/stem.6. [DOI] [PubMed] [Google Scholar]
- Lin HH, Chen YH, Yet SF, Chau LY. After vascular injury, heme oxygenase-1/carbon monoxide enhances re-endothelialization via promoting mobilization of circulating endothelial progenitor cells. J Thromb Haemost. 2009;7:1401–1408. doi: 10.1111/j.1538-7836.2009.03478.x. [DOI] [PubMed] [Google Scholar]
- Lo EH, Broderick JP, Moskowitz MA. tPA and proteolysis in the neurovascular unit. Stroke. 2004;35:354–356. doi: 10.1161/01.STR.0000115164.80010.8A. [DOI] [PubMed] [Google Scholar]
- Lynch DR, Dawson TM. Secondary mechanisms in neuronal trauma. Curr Opin Neurol. 1994;7:510–516. doi: 10.1097/00019052-199412000-00007. [DOI] [PubMed] [Google Scholar]
- Maines MD. The heme oxygenase system: a regulator of second messenger gases. Annu Rev Pharmacol Toxicol. 1997;37:517–554. doi: 10.1146/annurev.pharmtox.37.1.517. [DOI] [PubMed] [Google Scholar]
- McCoubrey WK, Jr, Huang TJ, Maines MD. Isolation and characterization of a cDNA from the rat brain that encodes hemoprotein heme oxygenase-3. Eur J Biochem. 1997;247:725–732. doi: 10.1111/j.1432-1033.1997.00725.x. [DOI] [PubMed] [Google Scholar]
- Mintz-Hittner HA, Kennedy KA, Chuang AZ. Efficacy of intravitreal bevacizumab for stage 3+ retinopathy of prematurity. N Engl J Med. 2011;364:603–615. doi: 10.1056/NEJMoa1007374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mobarak CD, Anderson KD, Morin M, Beckel-Mitchener A, Rogers SL, Furneaux H, King P, Perrone-Bizzozero I. The RNA-binding protein HuD is required for GAP-43 mRNA stability, GAP-43 gene expression, and PKC-dependent neurite outgrowth in PC12 cells. Mol Biol Cell. 2000;11:3191–3203. doi: 10.1091/mbc.11.9.3191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Motterlini R, Clark JE, Foresti R, Sarathchandra P, Mann BE, Green CJ. Carbon monoxide-releasing molecules: characterization of biochemical and vascular activities. Circ Res. 2002;90:E17–E24. doi: 10.1161/hh0202.104530. [DOI] [PubMed] [Google Scholar]
- Motterlini R, Mann BE, Johnson TR, Clark JE, Foresti R, Green CJ. Bioactivity and pharmacological actions of carbon monoxide-releasing molecules. Curr Pharm Des. 2003;9:2525–2539. doi: 10.2174/1381612033453785. [DOI] [PubMed] [Google Scholar]
- Motterlini R, Otterbein LE. The therapeutic potential of carbon monoxide. Nat Rev Drug Discov. 2010;9:728–743. doi: 10.1038/nrd3228. [DOI] [PubMed] [Google Scholar]
- Nada SE, Tulsulkar J, Shah ZA. Heme oxygenase 1-mediated neurogenesis is enhanced by Ginkgo biloba (EGb 761®) after permanent ischemic stroke in mice. Mol Neurobiol. 2014;49:945–956. doi: 10.1007/s12035-013-8572-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelson CW, Wei EP, Povlishock JT, Kontos HA, Moskowitz MA. Oxygen radicals in cerebral ischemia. Am J Physiol. 1992;263:H1356–H1362. doi: 10.1152/ajpheart.1992.263.5.H1356. [DOI] [PubMed] [Google Scholar]
- Pae HO, Oh GS, Choi BM, Kim YM, Chung HT. A molecular cascade showing nitric oxide-heme oxygenase-1-vascular endothelial growth factor-interleukin-8 sequence in human endothelial cells. Endocrinology. 2005;146:2229–2238. doi: 10.1210/en.2004-1431. [DOI] [PubMed] [Google Scholar]
- Panahian N, Yoshiura M, Maines MD. Overexpression of heme oxygenase-1 is neuroprotective in a model of permanent middle cerebral artery occlusion in transgenic mice. J Neurochem. 1999;72:1187–1203. doi: 10.1111/j.1471-4159.1999.721187.x. [DOI] [PubMed] [Google Scholar]
- Parfenova H, Basuroy S, Bhattacharya S, Tcheranova D, Qu Y, Regan RF, Leffler CW. Glutamate induces oxidative stress and apoptosis in cerebral vascular endothelial cells: contributions of HO-1 and HO-2 to cytoprotection. Am J Physiol Cell Physiol. 2006;290:C1399–C1410. doi: 10.1152/ajpcell.00386.2005. [DOI] [PubMed] [Google Scholar]
- Peers C. Modulation of ion channels and transporters by carbon monoxide: causes for concern? Front Physiol. 2012;3:477. doi: 10.3389/fphys.2012.00477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poss KD, Thomas MJ, Ebralidze AK, O’Dell TJ, Tonegawa S. Hippocampal long-term potentiation is normal in heme oxygenase-2 mutant mice. Neuron. 1995;15:867–873. doi: 10.1016/0896-6273(95)90177-9. [DOI] [PubMed] [Google Scholar]
- Routtenberg A, Cantallops I, Zaffuto S, Serrano P, Namgung U. Enhanced learning after genetic overexpression of a brain growth protein. Proc Natl Acad Sci USA. 2000;97:7657–7662. doi: 10.1073/pnas.97.13.7657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sagare AP, Bell RD, Zhao Z, Ma Q, Winkler EA, Ramanathan A, Zlokovic BV. Pericyte loss influences Alzheimer-like neurodegeneration in mice. Nat Commun. 2013;4:2932. doi: 10.1038/ncomms3932. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- Scapagnini G, D’Agata V, Calabrese V, Pascale A, Colombrita C, Alkon D, Cavallaro S. Gene expression profiles of heme oxygenase isoforms in the rat brain. Brain Res. 2002;954:51–59. doi: 10.1016/S0006-8993(02)03338-3. [DOI] [PubMed] [Google Scholar]
- Semenza GL. Targeting HIF-1 for cancer therapy. Nat Rev Cancer. 2003;3:721–732. doi: 10.1038/nrc1187. [DOI] [PubMed] [Google Scholar]
- Shinomura T, Nakao S, Mori K. Reduction of depolarization-induced glutamate release by heme oxygenase inhibitor: possible role of carbon monoxide in synaptic transmission. Neurosci Lett. 1994;166:131–134. doi: 10.1016/0304-3940(94)90468-5. [DOI] [PubMed] [Google Scholar]
- Snipes GJ, Chan SY, McGuire CB, Costello BR, Norden JJ, Freeman JA, Routtenberg A. Evidence for the coidentification of GAP-43, a growth-associated protein, and F1, a plasticity-associated protein. J Neurosci. 1987;7:4066–4075. doi: 10.1523/JNEUROSCI.07-12-04066.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spaide RF, Fisher YL. Intravitreal bevacizumab (Avastin) treatment of proliferative diabetic retinopathy complicated by vitreous hemorrhage. Retina. 2006;26:275–278. doi: 10.1097/00006982-200603000-00004. [DOI] [PubMed] [Google Scholar]
- Stone J, Itin A, Alon T, Pe’er J, Gnessin H, Chan-Ling T, Keshet E. Development of retinal vasculature is mediated by hypoxia-induced vascular endothelial growth factor (VEGF) expression by neuroglia. J Neurosci. 1995;15:4738–4747. doi: 10.1523/JNEUROSCI.15-07-04738.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suliman HB, Carraway MS, Tatro LG, Piantadosi CA. A new activating role for CO in cardiac mitochondrial biogenesis. J Cell Sci. 2007;120:299–308. doi: 10.1242/jcs.03318. [DOI] [PubMed] [Google Scholar]
- Trakshel GM, Maines MD. Multiplicity of heme oxygenase isozymes. HO-1 and HO-2 are different molecular species in rat and rabbit. J Biol Chem. 1989;264:1323–1328. [PubMed] [Google Scholar]
- Verma A, Hirsch DJ, Glatt CE, Ronnett GV, Snyder SH. Carbon monoxide: a putative neural messenger. Science. 1993;259:381–384. doi: 10.1126/science.7678352. [DOI] [PubMed] [Google Scholar]
- Wang B, Cao W, Biswal S, Dore S. Carbon monoxide-activated Nrf2 pathway leads to protection against permanent focal cerebral ischemia. Stroke. 2011;42:2605–2610. doi: 10.1161/STROKEAHA.110.607101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang J, Dore S. Heme oxygenase 2 deficiency increases brain swelling and inflammation after intracerebral hemorrhage. Neuroscience. 2008;155:1133–1141. doi: 10.1016/j.neuroscience.2008.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winkler EA, Bell RD, Zlokovic BV. Pericyte-specific expression of PDGF β receptor in mouse models with normal and deficient PDGF β receptor signaling. Mol Neurodegener. 2010;5:32. doi: 10.1186/1750-1326-5-32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winkler EA, Bell RD, Zlokovic BV. Central nervous system pericytes in health and disease. Nat Neurosci. 2011;14:1398–1405. doi: 10.1038/nn.2946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winkler EA, Sagare AP, Zlokovic BV. The pericyte: a forgotten cell type with important implications for Alzheimer’s disease? Brain Pathol. 2014;24:371–386. doi: 10.1111/bpa.12152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu ML, Ho YC, Yet SF. A central role of heme oxygenase-1 in cardiovascular protection. Antioxid Redox Signal. 2011;15:1835–1846. doi: 10.1089/ars.2010.3726. [DOI] [PubMed] [Google Scholar]
- Xing C, Lo EH. Help-me signaling: Non-cell autonomous mechanisms of neuroprotection and neurorecovery. Prog Neurobiol. 2017;152:181–199. doi: 10.1016/j.pneurobio.2016.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiong Y, Mahmood A, Chopp M. Animal models of traumatic brain injury. Nat Rev Neurosci. 2013;14:128–142. doi: 10.1038/nrn3407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yabluchanskiy A, Sawle P, Homer-Vanniasinkam S, Green CJ, Foresti R, Motterlini R. CORM-3, a carbon monoxide-releasing molecule, alters the inflammatory response and reduces brain damage in a rat model of hemorrhagic stroke. Crit Care Med. 2012;40:544–552. doi: 10.1097/CCM.0b013e31822f0d64. [DOI] [PubMed] [Google Scholar]
- Yemisci M, Gursoy-Ozdemir Y, Vural A, Can A, Topalkara K, Dalkara T. Pericyte contraction induced by oxidative-nitrative stress impairs capillary reflow despite successful opening of an occluded cerebral artery. Nat Med. 2009;15:1031–1037. doi: 10.1038/nm.2022. [DOI] [PubMed] [Google Scholar]
- Zhang R, Zhang L, Zhang Z, Wang Y, Lu M, Lapointe M, Chopp M. A nitric oxide donor induces neurogenesis and reduces functional deficits after stroke in rats. Ann Neurol. 2001;50:602–611. doi: 10.1002/ana.1249. [DOI] [PubMed] [Google Scholar]
- Zhao Y, Guan YF, Zhou XM, Li GQ, Li ZY, Zhou CC, Wang P, Miao CY. Regenerative neurogenesis after ischemic stroke promoted by nicotinamide phosphoribosyltransferase-nicotinamide adenine dinucleotide cascade. Stroke. 2015;46:1966–1974. doi: 10.1161/STROKEAHA.115.009216. [DOI] [PubMed] [Google Scholar]
- Zhuo M, Small SA, Kandel ER, Hawkins RD. Nitric oxide and carbon monoxide produce activity-dependent long-term synaptic enhancement in hippocampus. Science. 1993;260:1946–1950. doi: 10.1126/science.8100368. [DOI] [PubMed] [Google Scholar]