Abstract
Acute myeloid leukemia (AML) is an aggressive hematological malignancy with high incidence in the aging population. In addition, AML is one of the more common pediatric malignancies. Unfortunately, both of these patient groups are quite sensitive to chemotherapy toxicities. Investigation of blueberries specifically as an anti-AML agent has been limited, despite being a prominent natural product with no reported toxicity. In this study, blueberry extracts are reported for the first time to exert a dietary therapeutic effect in animal models of AML. Furthermore, in vitro studies revealed that blueberry extracts exerted anti-AML efficacy against myeloid leukemia cell lines as well as against primary AML, and specifically provoked Erk and Akt regulation within the leukemia stem cell subpopulation. This study provides evidence that blueberries may be unique sources for anti-AML biopharmaceutical compound discovery, further warranting fractionation of this natural product. More so, blueberries themselves may provide an intriguing dietary option to enhance the anti-AML efficacy of traditional therapy for subsets of patients that otherwise may not tolerate rigorous combinations of therapeutics.
Keywords: acute myeloid leukemia, blueberry, mouse models
Introduction
Natural product research has described numerous plant-derived compounds and extracts with beneficial health effects. Blueberries are rich sources for polyphenols, and are generally recognized for their health benefits arising from studies in models of aging, inflammation, diabetes, and cancer [1–5]. In contrast, the efficacy of blueberries or their components in cellular models of acute myeloid leukemia (AML) and other leukemias has minimally been explored [6]. AML is a heterogeneous group of hematological cancers defined by clonal expansion of immature myeloid progenitors in the blood and bone marrow [7–9]. It is an aggressive disease that is defined by various potential cytogenetic and molecular alterations and immunophenotypes [10]. While the incidence of AML is greatest in the aging population, it can occur at any age and is one of the more common pediatric malignancies [7–9]. The prognosis for most AML is poor, with minimal clinical advances being made over the past few decades [10]. Remission is sometimes achieved, but only after aggressive chemotherapy that can be detrimental to pediatric and elderly patients. Therefore, studies aimed at identifying bioactive compounds with anti-AML efficacy from blueberries and other natural products are of interest for biopharmaceutical development given the need for effective and minimally toxic therapies. In the current study, we evaluated an anti-AML effect for blueberry extracts in both cellular and animal models of AML. By defining a role for blueberries as an anti-AML natural product this study may also lead to dietary interventions to augment the treatment of AML in pediatric or elderly patients that otherwise may not respond well to aggressive chemotherapy.
Materials and Methods
Cell Culture
Murine C1498, 32D-p210-GFP, and 32D-FLT3-ITD cells, and human HL-60/vcr cells were maintained at 37°C, and 5% CO2, in RPMI-1640 supplemented with 10% fetal bovine serum (FBS) and 1% penicillin/streptomycin. Human KG-1 cells were likewise maintained, in IMDM supplemented with 20% FBS and 1% penicillin/streptomycin. Patient AML samples were obtained using informed consent approved by the Penn State College of Medicine Institutional Review Board. Samples were prepared from peripheral blood or bone marrow using Ficoll-Paque separation of white blood cells, and for short term assays were maintained in RPMI-1640 supplemented with 10% FBS.
In Vitro Assays
Cellular viability assays were performed as previously described using a Cell Titer 96 AQueous Non-Radioactive Cell Proliferation Assay according to the manufacturer’s instructions (Promega, Madison, WI) [11,12]. Colony forming assays were performed as previously described using MethoCult H4434 according to the manufacturer’s instructions (Stem Cell Technologies, Vancouver, BC, Canada) [11]. Apoptosis assays were performed as previously described using Annexin V and 7-aminoactinomycin D from BD Biosciences (San Jose, CA) [11,12], and a fluorophore-conjugated antibody targeting CD45 from Biolegend (San Diego, CA) to gate the blast population. Phosflow analysis was performed by using the BD Cytofix/Cytoperm kit according to the manufacturer’s instructions (BD Biosciences, San Jose, CA). Briefly, cells were fixed following treatment and then their membranes were permeabilized to permit intracellular staining with fluorophore-conjugated antibodies targeting phosphorylated Akt and Erk (BD Biosciences, San Jose, CA). Cells were subsequently stained to evaluate leukemia stem cells with fluorophore-conjugated antibodies targeting CD34 and CD38 (BD Biosciences, San Jose, CA), as well as CD96 (eBioscience, San Diego, CA). Flow cytometry was performed at the Penn State College of Medicine Flow Cytometry Core using a BD Biosciences LSR II flow cytometer and BD FACS Diva software.
Animal Trials
NOD-Scid and C57BL/6J mice were bred from founders obtained from the Jackson Laboratory (Bar Harbor, ME). Female NOD-Scid mice were xenografted with HL-60/vcr (2×106 cells/mouse) or KG-1 (2.5×106 cells/mouse) cells by tail vein injection. After one week, mice were randomized and given ad libitum access to either control drinking water or crude blueberry extract dissolved in drinking water (1% w/v). Male and female C57BL/6J mice were engrafted with C1498 cells (1×106 cells/mouse) by retro-orbital injection. After one week, mice were randomized and given a crude blueberry preparation to eat (25 mg/mouse/week), ad libitum access to chloroquine in the drinking water (0.288 mg/ml sweetened with 15 mg/ml glucose to alleviate bitterness) [13], a combination of both, or control water (sweetened with 15 mg/ml glucose). Mice were euthanized once they reached moribund status. All procedures were approved by the Institutional Animal Care and Use Committee of the Penn State College of Medicine.
Blueberry Extraction
Solvents and reagents were obtained from VWR (Radnor, PA) and Sigma (St. Louis, MO). Briefly, whole blueberries were lyophilized, crushed to powder, and a crude extract was prepared by extracting for 30 minutes with agitation using aqueous acetone (70/30 acetone/water), and dried by rotory-evaporation and lyophilization [3,4]. As these were whole extracts, the mass yield was slightly over 50% of the original starting material. Standardized blueberry powder was alternatively obtained from FutureCeuticals (Mornence, IL). No differences were noted in cell culture studies between the blueberry crude extracts, and the standardized blueberry powder.
Statistical Analysis
Colony-forming and apoptosis assay comparisons of untreated controls and treatments with blueberry extracts were made using an unpaired t-test. For in vivo studies, survival analysis was performed using the Mantel-Cox Logrank test. All experiments were performed with an n≥3 bioloigcal replicate sample size, and graphical averages are depicted +/− standard deviation from the mean.
Results and Discussion
Initially, blueberry extracts were evaluated for anti-AML therapeutic efficacy using cell lines and patient samples. Crude preparations were observed to diminish the viability of the AML cell lines HL-60/vcr, C1498, and 32D-FLT3-ITD, as well as the chronic myeloid leukemia (CML) cell line 32D-p210-GFP (Fig. 1A). Anti-AML efficacy for crude blueberry extracts was further demonstrated using a colony forming assay with a poor prognosis patient sample (#329: inv3, −7) (Fig. 1B), as well as several patient samples using short-term apoptosis assays (Fig. 1C). Apoptosis assays used several different patient cases including three poor prognosis AML (#651: −7, −5, #652: −7, #658: complex cytogenetics), one intermediate prognosis AML (#657: +8), one favorable prognosis AML (#661: inv16), and one CML case (#653). Overall, these results demonstrated in vitro anti-leukemia efficacy for blueberry extracts using both cell lines and primary patient samples. Further studies are warranted to identify the specific components of blueberries that exert anti-leukemia efficacy to facilitate their potential biopharmaceutical development. These constituent components of the blueberries may target signaling pathways critical to survival and proliferation of leukemia or related hematological disorders.
Figure 1. In vitro anti-AML therapeutic effect of blueberry extracts.
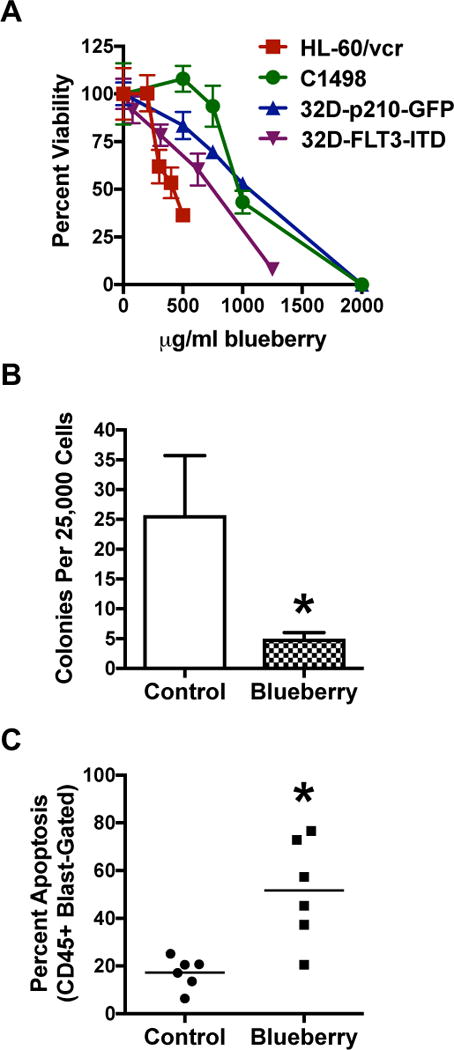
(A) Cellular viability was determined by MTS assay following 48-hour exposure of AML cell lines (dashed line with circles: C1498, solid line with squares: HL-60/vcr, dashed line with upside down triangles: 32D-FLT3-ITD) and a CML cell line (solid line with right side up triangles: 32D-p210-GFP) to crude blueberry extract. (B) Colony forming capacity of a poor prognosis patient AML case #329 was determined in the presence of 500 μg/ml crude blueberry extract (t-test, p=0.0245, n≥3). (C) Ficol-prepared WBC fractions of patient AML (case #651, #652, #657, #658, and #661) and patient CML (case #653) were exposed to 500 μg/ml crude blueberry extracts for 48 hours followed by flow cytometry to determine percent apoptosis (Annexin V+) in the CD45-gated blast population (t-test, p=0.0098, n=6).
Phosflow analysis was used to evaluate the effects of blueberry extracts on leukemia stem cells. This technique allows single cell analysis of proteins such as Akt and Erk, which are kinases critical to regulating signaling networks classically responsible for growth and survival. In this study, HL-60/vcr cells displayed dose-dependent increases in the phosphorylation of Erk, indicative of activation, in response to blueberry extract exposure (Fig. 2A). This effect was evident in both the leukemia stem cells, defined as CD34+CD38− or CD34+CD38−CD96+, as well as within the bulk leukemia cell population. Previously, Hosen et al. used CD96 to further define the AML stem cell fraction [14]. Our group later evaluated CD96 as an AML stem cell marker for targeted therapeutics [15]. In the present research study, Phosflow analysis of a poor prognosis AML patient sample revealed that blueberry extracts augmented Erk phosphorylation, as well as Akt phosphorylation, within both the leukemia stem cell (CD34+CD38− or CD34+CD38−CD96+) and bulk leukemia cell populations (Fig. 2B). This observation of an increase in Erk and Akt signaling may seem counterintuitive at first, but is specifically meaningful within the context of leukemia stem cell biology. Leukemia stem cells are quiescent and not cycling, which is important in protecting them from DNA-damaging chemotherapeutics [16,17]. Viale et al. demonstrated that disruption of the cell cycle inhibitor p21 caused leukemia stem cells to hyper-proliferate which ultimately depleted their population [16]. This study provided support for the concept of a two-step approach to leukemia treatment whereby initial treatment provokes the leukemia stem cell to enter the cell cycle and hyper-proliferate, followed by a second more cytotoxic therapy. In the context of the current study, where blueberry extracts represent a complex mixture of potentially therapeutic compounds, it is possible that particular compound(s) provoke the leukemia stem cell to enter the cell cycle by promoting Erk and/or Akt phosphorylation. At the same time, other blueberry compound(s) may exert a cytotoxic effect on these newly cycling leukemia stem cells as well as the overall leukemia cell population. This suggests that even blueberry extracts themselves may have utility as a therapeutic alternative or adjuvant to traditional anti-leukemia therapeutics which may be effective given the ability to provoke leukemia stem cells into an ultimately detrimental proliferative cycle. Studies to define and isolate constituent components of blueberries that influence leukemia stem cells specifically are warranted.
Figure 2. Blueberry extracts regulate Erk and Akt phosphorylation in leukemia stem cells.

(A) HL-60/vcr cells were exposed overnight (12 hours) to blueberry extracts and phosphorylation of Akt and Erk were monitored by Phosflow analysis (n=3 per data point). Antibodies were used to define leukemia stem cells as CD34+CD38− or CD34+CD38−CD96+. (B) Phosflow analysis was similarly used to evaluate cells exposed overnight to blueberry extracts from AML patient case #329 (poor prognosis) (n=3 per data point).
Anti-AML efficacy for crude blueberry extracts was next evaluated using in vivo models. The lifespan of NOD-Scid mice xenografted with human HL-60/vcr (Fig. 3A), or KG-1 cells (Fig. 3B), was significantly extended by blueberry supplemented in drinking water. Additionally, blueberry extracts formulated as an edible snack modestly extended the lifespan of C57BL/6J mice engrafted with the aggressive murine AML cell line C1498 (Fig. 4). Intriguingly, when combined with the autophagy inhibitor chloroquine, blueberry treatment substantially augmented the lifespan of these mice. The combination with chloroquine was selected in this instance after we previously observed and reported that C1498 cells upregulate autophagy as a mechanism of resistance to therapy [11]. Overall, these in vivo studies showed that dietary supplementation of blueberry extracts is sufficient to exert an anti-AML effect in animal models. This is important because pediatric and elderly AML patients may benefit from dietary therapeutic strategies that otherwise exert no toxicity.
Figure 3. Survival extended in mice with AML following dietary blueberry extract supplementation.
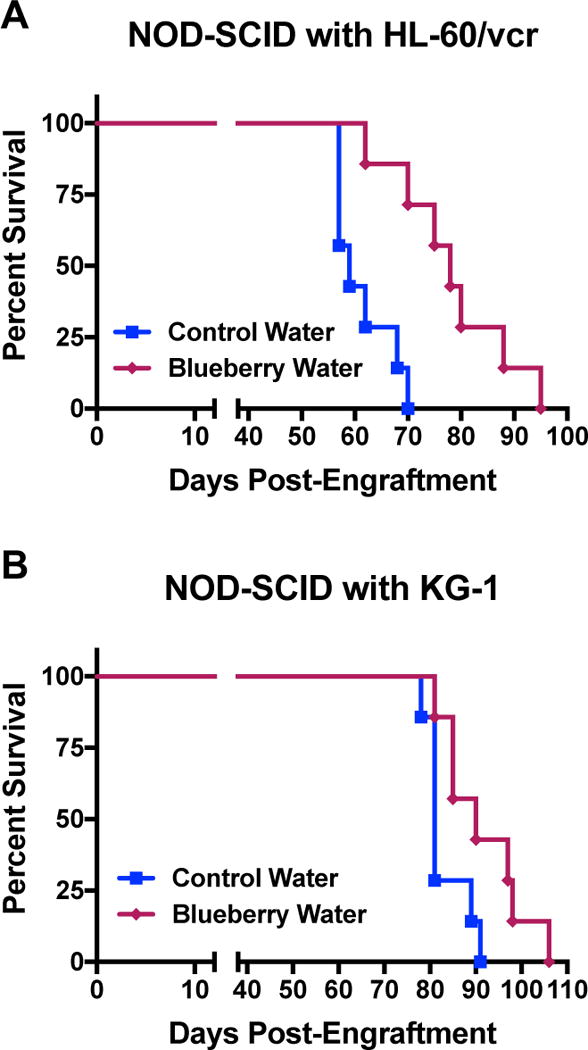
(A) NOD-Scid mice were xenografted with the human AML cell line HL-60/vcr (2×106 cells/mouse by tail vein injection) and survival was monitored after mice were given crude blueberry extract (1% w/v) (dark line with diamonds) ad libitum in the drinking water, or control water (light gray line with squares), starting one week following engraftment (Mantel-Cox Logrank test, p=0.0017, n=7/group). (B) NOD-Scid mice were xenografted with the human AML cell line KG-1 (2.5×106 cells/mouse by tail vein injection) and survival was monitored after mice were given crude blueberry extract (1% w/v) (dark line with diamonds) ad libitum in the drinking water, or control water (light gray line with squares), starting one week following engraftment (Mantel-Cox Logrank test, p=0.0382, n=7/group).
Figure 4. Dietary blueberry extract supplementation in combination with chloroquine extended the survival of mice with AML.
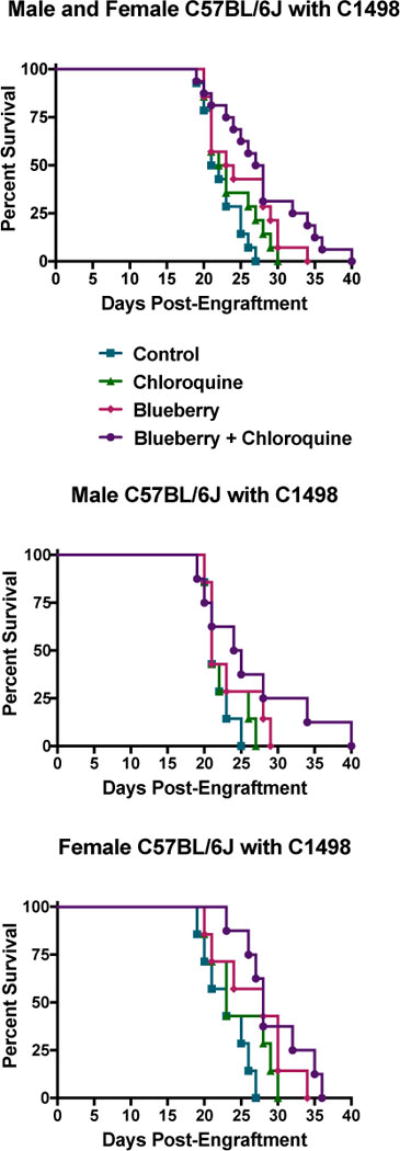
C57BL/6J mice were engrafted with the murine AML cell line C1498 (1×106 cells/mouse by retro-orbital injection) and survival was monitored after mice were given a crude blueberry preparation to eat (25 mg/mouse/week) (line with upside down triangles), chloroquine in the drinking water (25 μM sweetened with 1% w/v glucose to alleviate bitterness) ad libitum (line with right side up triangles), or a combination of both (line with circles), or control water alone (line with squares), starting one week following engraftment (Mantel-Cox Logrank tests: Males & Females together, p=0.0263, n≥14/group; Males only, not significant at p=0.1225, n≥7/group; Females only, p=0.0479, n≥7/group).
Conclusions
There is an urgent need for more effective and less toxic therapies for the treatment of AML. Many patients are not effectively treated with current standard of care therapies due to severe toxicity. Many times treatment will not commence, or will be halted before completion. Dietary interventions may offer alternative or adjuvants that are minimally toxic if at all. In the present study, blueberry extracts were evaluated for the first time for anti-AML efficacy in both cellular and animal models of AML. In addition to a demonstration of therapeutic efficacy in these models, the present study further demonstrated that blueberry extracts could regulate the AML stem cell. Therefore, constituent components of the blueberry hold promise not only as anti-AML therapeutics, but as anti-AML stem cell therapeutics. As part of an extract, or isolated and in combination with cytotoxic chemotherapeutics, blueberry components hold the potential to eradicate the specific cells that are primarily responsible for AML development, progression, relapse, and therapy resistance. More so, the success of the in vivo studies further showed good bioavailability for the components of the blueberry that exert anti-AML efficacy. Altogether suggesting that blueberries could be a profound source for anti-leukemia biopharmaceutical discovery and development.
Finally, we have demonstrated that blueberry extracts can impede pro-inflammatory signaling pathways mediated by a neutral sphingomyelinase [3], as well as by an NADPH oxidase [4]. These particular pathways may augment leukemogenic signaling by redox-mediated inactivation of tyrosine phosphatases, which can lead to increased tyrosine kinase-mediated signaling [18,19]. In addition to the prevalence of dysregulated growth factor signaling pathways in AML and other myeloid hematological disorders [10,20–22], inflammatory cytokines have been associated with relapsing AML and AML secondary to Fanconi anemia [23,24]. Altogether, the persistence of these conditions in AML suggests that novel anti-inflammatory agents such as those derived from blueberry extracts may be of particular benefit for the treatment of AML. In conclusion, this study demonstrated anti-AML efficacy for blueberry extracts. This is of significance because natural products have long served as sources and blueprints for the discovery and development of novel biopharmaceuticals [25–27]. Therefore, the present study may hold promise to identify minimally toxic and potentially combinatorial anti-AML compounds that can be utilized in anti-AML biopharmaceutical development.
Acknowledgments
We would like to thank Dr. Hubert Serve of the University of Münster for generously providing the 32D-FLT3-ITD cells. This study was funded in part by the National Institute for General Medical Sciences of the National Institutes for Health under award number P20-GM103395 (K.L.D.), the National Cancer Institute of the National Institutes for Health under award number K22-CA190674 (B.M.B.), as well as the Penn State University Kiesendahl Family Endowed Leukemia Research Fund (D.F.C.), the Kenneth Noel Memorial Fund (D.F.C.), and PA Tobacco Settlement funds (M.K. and D.F.C.).
References
- 1.Dunlap KL, Reynolds AJ, Duffy LK. Total antioxidant power in sled dogs supplemented with blueberries and the comparison of blood parameters associated with exercise. Comparative Biochemistry and Physiology Part A: Molecular & Integrative Physiology. 2006;143:429–434. doi: 10.1016/j.cbpa.2005.09.007. [DOI] [PubMed] [Google Scholar]
- 2.Grace MH, Esposito D, Dunlap KL, Lila MA. Comparative analysis of phenolic content and profile, antioxidant capacity, and anti-inflammatory bioactivity in wild Alaskan and commercial Vaccinium berries. Journal of Agricultural and Food Chemistry. 2014;62:4007–4017. doi: 10.1021/jf403810y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Gustafson SJ, Barth BM, McGill CM, Clauson TP, Kuhn TB. Wild Alaskan Blueberry Extracts Inhibit a Magnesium-Dependent Neutral Sphingomyelinase Activity in Neurons Exposed to TNFα. Current Topics in Nutraceutical Research. 2007;5:183–188. [Google Scholar]
- 4.Gustafson SJ, Dunlap KL, McGill CM, Kuhn TB. A nonpolar blueberry fraction blunts NADPH oxidase activation in neuronal cells exposed to tumor necrosis factor-α. Oxidative Medicine and Cellular Longevity. 2012;2012:768101. doi: 10.1155/2012/768101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kellogg J, Wang J, Flint C, Ribnicky D, Kuhn P, De Mejia EG, Raskin I, Lila MA. Alaskan wild berry resources and human health under the cloud of climate change. Journal of Agricultural and Food Chemistry. 2010;58:3884–3900. doi: 10.1021/jf902693r. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kai H, Akamatsu E, Torii E, Kodama H, Yukizaki C, Sakakibara Y, Suiko M, Morishita K, Kataoka H, Matsuno K. Inhibition of proliferation by agricultural plant extracts in seven human adult T-cell leukaemia (ATL)-related cell lines. Journal of Natural Medicines. 2011;65:651–655. doi: 10.1007/s11418-011-0510-5. [DOI] [PubMed] [Google Scholar]
- 7.Baldus CD, Mrózek K, Marcucci G, Bloomfield CD. Clinical outcome of de novo acute myeloid leukaemia patients with normal cytogenetics is affected by molecular genetic alterations: a concise review. British Journal of Haematology. 2007;137:387–400. doi: 10.1111/j.1365-2141.2007.06566.x. [DOI] [PubMed] [Google Scholar]
- 8.Becker H, Marcucci G, Maharry K, Radmacher MD, Mrózek K, Margeson D, Whitman SP, Wu YZ, Schwind S, Paschka P, Powell BL, Carter TH, Kolitz JE, Wetzler M, Carroll AJ, Baer MR, Caligiuri MA, Larson RA, Bloomfield CD. Favorable prognostic impact of NPM1 mutations in older patients with cytogenetically normal de novo acute myeloid leukemia and associated gene-and microRNA-expression signatures: a Cancer and Leukemia Group B study. Journal of Clinical Oncology. 2010;28:596–604. doi: 10.1200/JCO.2009.25.1496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kottaridis PD, Gale RE, Frew ME, Harrison G, Langabeer SE, Belton AA, Walker H, Wheatley K, Bowen DT, Burnett AK, Goldstone AH, Linch DC. The presence of a FLT3 internal tandem duplication in patients with acute myeloid leukemia (AML) adds important prognostic information to cytogenetic risk group and response to the first cycle of chemotherapy: analysis of 854 patients from the United Kingdom Medical Research Council AML 10 and 12 trials. Blood. 2011;98:1752–1759. doi: 10.1182/blood.v98.6.1752. [DOI] [PubMed] [Google Scholar]
- 10.Patel JP, Gönen M, Figueroa ME, Fernandez H, Sun Z, Racevskis J, Van Vlierberghe P, Dolgalev I, Thomas S, Aminova O, Huberman K, Cheng J, Viale A, Socci ND, Heguy A, Cherry A, Vance G, Higgins RR, Ketterling RP, Gallagher RE, Litzow M, van den Brink MR, Lazarus HM, Rowe JM, Luger S, Ferrando A, Paietta E, Tallman MS, Melnick A, Abdel-Wahab O, Levine RL. Prognostic relevance of integrated genetic profiling in acute myeloid leukemia. New England Journal of Medincine. 2012;366:1079–1089. doi: 10.1056/NEJMoa1112304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Brown TJ, Garcia A, Kissinger LN, Shanmugavelandy SS, Wang X, Cabot MC, Kester M, Claxton DF, Barth BM. Therapeutic Combination of Nanoliposomal Safingol and Nanoliposomal Ceramide for Acute Myeloid Leukemia. Journal of Leukemia (Los Angeles) 2013;1:110. [Google Scholar]
- 12.McGill CM, Alba-Rodriguez EJ, Li S, Benson CJ, Ondrasik RM, Fisher LN, Claxton DF, Barth BM. Extracts of Devil’s club (Oplopanax horridus) exert therapeutic efficacy in experimental models of acute myeloid leukemia. Phytotherapy Research. 2014;28:1308–1314. doi: 10.1002/ptr.5129. [DOI] [PubMed] [Google Scholar]
- 13.Lewis MD, Pfeil J, Mueller AK. Continuous oral chloroquine as a novel route for Plasmodium prophylaxis and cure in experimental murine models. BMC Research Notes. 2011;4:262. doi: 10.1186/1756-0500-4-262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hosen N, Park CY, Tatsumi N, Oji Y, Sugiyama H, Gramatzki M, Krensky AM, Weissman IL. CD96 is a leukemic stem cell-specific marker in human acute myeloid leukemia. Proceedings of the National Academy of Sciences USA. 2007;104:11008–11013. doi: 10.1073/pnas.0704271104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Barth BM, Altinoğlu EI, Shanmugavelandy SS, Kaiser JM, Crespo-Gonzalez D, DiVittore NA, McGovern C, Goff TM, Keasey NR, Adair JH, Loughran TP, Claxton DF, Kester M. Targeted indocyanine-green-loaded calcium phosphosilicate nanoparticles for in vivo photodynamic therapy of leukemia. ACS Nano. 2011;5:5325–5337. doi: 10.1021/nn2005766. [DOI] [PubMed] [Google Scholar]
- 16.Viale A, De Franco F, Orleth A, Cambiaghi V, Giuliani V, Bossi D, Ronchini C, Ronzoni S, Muradore I, Monestiroli S, Gobbi A, Alcalay M, Minucci S, Pelicci PG. Cell-cycle restriction limits DNA damage and maintains self-renewal of leukaemia stem cells. Nature. 2009;457:51–56. doi: 10.1038/nature07618. [DOI] [PubMed] [Google Scholar]
- 17.Viale A, Pelicci PG. Awaking stem cells from dormancy: growing old and fighting cancer. EMBO Molecular Medicine. 2009;1:88–91. doi: 10.1002/emmm.200900019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Truong TH, Carroll KS. Redox regulation of epidermal growth factor receptor signaling through cysteine oxidation. Biochemistry. 2012;51:9954–9965. doi: 10.1021/bi301441e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wall SB, Oh JY, Diers AR, Landar A. Oxidative modification of proteins: an emerging mechanism of cell signaling. Frontiers in Physiology. 2012;3:369. doi: 10.3389/fphys.2012.00369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bar-Natan M, Nelson EA, Xiang M, Frank DA. STAT signaling in the pathogenesis and treatment of myeloid malignancies. JAKSTAT. 2012;1:55–64. doi: 10.4161/jkst.20006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Levine RL. JAK-mutant myeloproliferative neoplasms. Current Topics in Microbiology and Immunology. 2012;355:119–133. doi: 10.1007/82_2011_170. [DOI] [PubMed] [Google Scholar]
- 22.Pikman Y, Lee BH, Mercher T, McDowell E, Ebert BL, Gozo M, Cuker A, Wernig G, Moore S, Galinsky I, DeAngelo DJ, Clark JJ, Lee SJ, Golub TR, Wadleigh M, Gilliland DG, Levine RL. MPLW515L is a novel somatic activating mutation in myelofibrosis with myeloid metaplasia. PLoS Medicine. 2006;3:e270. doi: 10.1371/journal.pmed.0030270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Li J, Sejas DP, Zhang X, Qiu Y, Nattamai KJ, Rani R, Rathbun KR, Geiger H, Williams DA, Bagby GC, Pang Q. TNF-alpha induces leukemic clonal evolution ex vivo in Fanconi anemia group C murine stem cells. Journal of Clinical Investigation. 2007;117:3283–3295. doi: 10.1172/JCI31772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rickmann M, Macke L, Sundarasetty BS, Stamer K, Figueiredo C, Blasczyk R, Heuser M, Krauter J, Ganser A, Stripecke R. Monitoring dendritic cell and cytokine biomarkers during remission prior to relapse in patients with FLT3-ITD acute myeloid leukemia. Annals of Hematology. 2013;92:1079–1090. doi: 10.1007/s00277-013-1744-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Gerber HP, Koehn FE, Abraham RT. The antibody-drug conjugate: an enabling modality for natural products-based cancer drug discovery. Natural Products Reports. 2013;30:625–639. doi: 10.1039/c3np20113a. [DOI] [PubMed] [Google Scholar]
- 26.Giddings LA, Newman DJ. Microbial natural products: molecular blueprints for antitumor drugs. Journal of Industrial Microbiology and Biotechnology. 2013;40:1181–1210. doi: 10.1007/s10295-013-1331-1. [DOI] [PubMed] [Google Scholar]
- 27.Newman DJ, Giddings LA. Natural products as leads to antitumor drugs. Phytochemistry Reviews. 2014;13:123–137. [Google Scholar]


