ABSTRACT
Detailed annotation of an IncHI2 plasmid, pSTM6-275, from Salmonella enterica serotype 1,4,5,12:i:- strain TW-Stm6 revealed a composite structure, including antimicrobial resistance genes on mobile genetic elements. The plasmid was thermosensitive for transfer to Escherichia coli and conferred reduced susceptibility to antibiotics, copper sulfate, and silver nitrate. Metal ion susceptibility was dependent on physiological conditions, giving an insight into the environments where this trait might confer a fitness advantage.
KEYWORDS: IncHI2, Salmonella, copper, heavy metals, integrons, plasmid-mediated resistance, silver
TEXT
Salmonella enterica is a common enteric pathogen of humans and animals and is found in many environmental and animal reservoirs with zoonotic potential. Distinct clones of multidrug-resistant S. enterica serovar Typhimurium have emerged and dominated in succession (1, 2). A recent clone of S. Typhimurium (strain SO4698-09) carries a Salmonella genomic island (SGI) which contributes to enhanced resistance to copper sulfate, a common animal feed additive (3). We recently reported the genome sequence of S. enterica 1,4,[5],12:i:- strain TW-Stm6, an isolate recovered from pig feces which has the same antigenic formula, phage type, and sequence type as strain SO4698-09, and also carries the genomic island, SGI-4 (3–5). The assembled genome of TW-Stm6 comprised a 4,999,862-bp chromosome, a 4-kb MOBQ plasmid (pSTM6-4), and a 275.8-kb IncHI2 plasmid (pSTM6-275). Here, we report the detailed annotation of pSTM6-275 and its genetic structure, function, and transmission of antibiotic and heavy-metal resistance genes to other bacteria.
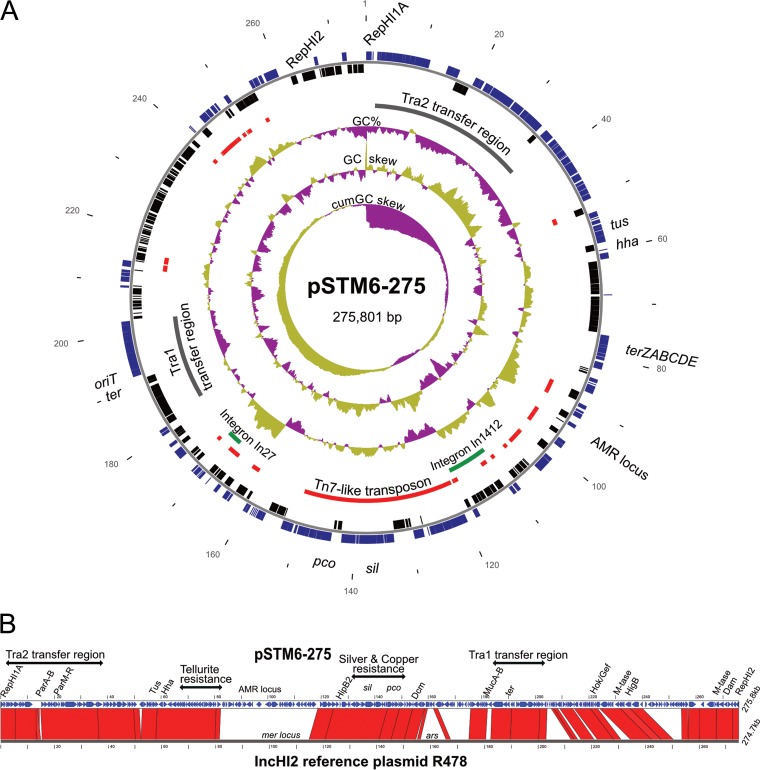
Annotations were revised using EcoGene version 3 (www.ecogene.org), UniProt (www.uniprot.org), Rfam (rfam.xfam.org), BLAST (https://blast.ncbi.nlm.nih.gov/Blast.cgi), and literature searches. Plasmid typing was performed using PlasmidFinder (cge.cbs.dtu.dk/services/PlasmidFinder-1.3), the Plasmid MLST database (pubmlst.org/plasmid), and local searches of custom database sequences and IncHI typing (6). Insertion sequence (IS) elements were typed using ISFinder (https://www-is.biotoul.fr). A diagram of the 275,801-bp plasmid pSTM6-275 (accession no. CP019647.1) is shown in Fig. 1A. Many genes were clustered in functional units, such as the ter operon (tellurium resistance), the sil locus (silver efflux), the pco locus (copper efflux) and the transfer regions Tra1/Tra2. Tra2 has an origin of plasmid transfer, oriT, a potential replication terminus (ter) site, and a tus gene encoding the replication-termination protein Tus. The plasmid copy number, determined by normalizing the sequence depth relative to the chromosome, was 1.4. In silico typing of plasmid pSTM6-275 indicated it belonged to the IncHI2 (subtype 3) and was similar to plasmid IncHI2 reference plasmid R478 (accession no. BX664015.1) (Fig. 1B). BLASTN searches of the GenBank database (4 October 2017) failed to find any other plasmid that matched (≥99% nt identity) more than about 70% of pSTM6-275. Figure 2A depicts regions that are rich in mobile genetic elements, including IS elements, transposons, and integrons, and containing multiple resistance genes (blaTEM, strA, strB, sul3, aadA1, aadA2, cmlA, aphA2, and two copies of tetA) encoding resistance to ampicillin, streptomycin, spectinomycin, sulfonamide, trimethoprim, chloramphenicol, kanamycin, and tetracycline, respectively. Copies of blaTEM, strA, strB, tetB, and sul2 occur on the chromosome in SGI-4 (5). The class I integron In27 (dfrA12_gcuF_aadA2_ΔqacE), encoding resistance to trimethoprim and streptomycin/spectinomycin, also contains a truncated sul1 gene, disrupted by IS26. A second integron, In1412, classified as a novel class I integron (7), was 9,240 bp and contained the array estX3_psp_aadA2_cmlA1_aadA1_qacHD4::IS1203 conferring resistance to streptomycin/spectinomycin and chloramphenicol (Fig. 2). A fragment of the macrolide efflux major facilitator superfamily (MFS) gene mefB (8) and a sul3 element/domain (9) lie proximal to qacH and are flanked by divergent IS26 elements.
FIG 1.
(A) Diagram of plasmid pSTM6-275 (275.8 kb). Tracks show (from outermost to center): scale in kilobase pairs; predicted coding sequences (CDS) of the top (blue) and bottom (black) strands; transposons and IS elements (red); integrons (green) and Tra regions (gray); GC%; GC-skew; and cumulative GC-skew. (B) Alignment of pSTM6-275 and the IncHI2 reference plasmid R478 (accession no. BX664015.1). Regions of nucleotide sequence similarity of ≥85% are indicated in red. Backbone regions and some of the more important proteins and loci of pSTM6-275 are indicated at the top. AMR, antimicrobial resistance.
FIG 2.
(A) Gene map of the resistance regions of pSTM6-275. Antibiotic, metal, and disinfectant resistance genes are clustered into two nearby regions, 87.1 to 151.4 kb (main sequence) and 162.1 to 173.7 kb (lower sequence connected by dotted line to the right). In each sequence, genes and operons are shown above the horizontal line, and IS elements, transposons, and integrons are displayed under the line. Integron attC sites are shown as triangles within integron borders. The scale at the top indicates kilobase pairs. (B) Comparison of sul3-integron type IIIc region of the E. coli plasmid pRYC306 (accession no. HQ875016) with the corresponding region of pSTM6-275. Nearly identical sequences are shaded red, or gray for IS26 elements. As indicated in the diagram, the flanking 8-bp direct repeats (CTTAGGTC) of pRYC306 IS element IS26 (nucleotides [nt] 7321 to 6502) are found split between the two leftmost copies of IS26 in pSTM6-275. A 49-bp sequence, depicted as solid blue arrows close to the horizontal lines, occurs twice in pRYC306 (near IS26 and near sul3) and once in pSTM6-275. Size scales, in kilobase pairs, are shown at the top and bottom.
A Tn7 family transposon (32.4 kb) carrying silver and copper resistance loci, silESRCFBAGP and pcoGE1ABCDRSE2 (Fig. 2A), occurs between the integrons and is delimited by inverted terminal repeats and flanking 5-bp direct repeats (Fig. 1.) Similar elements have been detected in other IncHI2 plasmids from animal-associated bacteria (10). Sil and Pco systems are composed of metal ion-binding proteins and transporters (11–13). A sil-pco locus with the same gene arrangement also occurs on the TW-Stm6 chromosome in SGI-4, but unlike the plasmid version, it lacks Tn7-like tnsABCD genes and inverted terminal repeats, suggesting a different evolutionary history.
Many IncHI2 plasmids are thermosensitive for transfer, with 27 to 33°C being the permissive temperature and >37°C being the nonpermissive temperature (14). Since pSTM6-275 has all the genes for proteins required for self-transmission, this function and its thermosensitive character were examined. Plasmid stability and conjugal transfer of pSTM6-275 from Salmonella to Escherichia coli DH5α were tested according to published methods (14), and the results are from two experiments performed in duplicate. Transfer occurred at 1.3 × 10−5 transconjugants per donor at 27°C, but no transfer was detected at 37°C. Transconjugants coinherited resistance to ampicillin, sulfonamide, streptomycin, spectinomycin, kanamycin, tetracycline, trimethoprim, and chloramphenicol, consistent with the plasmid structure. Colonies (n = 112) derived from a culture of the transconjugants, grown at 44°C for 24 h without antibiotic selection, retained all resistance markers, indicating that the plasmid was not thermosensitive for maintenance in E. coli. In this respect, pSTM6-275 differs from the reported phenotype of plasmid R478 (14).
The MICs of CuSO4 and AgNO3 were determined by agar dilution assays using LB agar (pH 7.2; 25 mM HEPES) (10, 12) using Oxoid AnaeroGen sachets if required. The sensitivities to of Salmonella TW-Stm6, E. coli DH5α, and two E. coli pSTM-275 transconjugants are shown in Table 1. The two transconjugants had the same MICs. The Salmonella donor had a higher AgNO3 MIC at 27°C (800 μM) than at 37°C (50 μM), and the MIC was not influenced by oxygen availability. The plasmid increased the MIC of AgNO3 for E. coli from 50 to 800 μM at 27°C; however, this effect was not seen at the higher temperature, where the MIC for all strains was 50 μM. The MIC to CuSO4 for E. coli was affected by oxygen. E. coli was most sensitive to CuSO4 under anaerobic conditions, but plasmid-bearing transconjugants were less sensitive possibly due to more efficient efflux.
TABLE 1.
MICs of CuSO4 and AgNO3a
| Substance | Strain type (strain) | MICs by temp and oxygen availability |
|||
|---|---|---|---|---|---|
| 37°C |
27°C |
||||
| With oxygen | Without oxygen | With oxygen | Without oxygen | ||
| AgNO3 | Donor (Salmonella Typhimurium TW-Stm6) | 50 | 50 | 800 | 800 |
| Recipient (E. coli DH5α) | 50 | 50 | 50 | 50 | |
| 2 transconjugants | 50 | 50 | 800 | 800 | |
| CuSO4 | Donor (Salmonella Typhimurium TW-Stm6) | 12.5 | 12.5 | 12.5 | 12.5 |
| Recipient (E. coli DH5α) | 6.25 | 1.56 | 6.26 | 1.56 | |
| 2 transconjugants | 6.25 | 6.25 | 6.25 | 6.25 | |
MICs for AgNO3 are given in micromolar, and those for CuSO4 are given in millimolar.
ST3 IncHI2 plasmids are widespread in food-producing animals (10), and despite their potential to disseminate antimicrobial resistance genes, few complete sequences have been characterized in detail. Our results suggest that the transmission of pSTM6-275 is probably restricted to outside a mammalian host given the thermosensitive nature of transfer, suggesting that it is well adapted for persistence in the environment. Furthermore, the expression of at least some of the metal resistance traits was influenced by physiological conditions. Copper metabolism in enterobacteria is complex, as several genes can be involved, including those involved in transport, oxidation, and regulation. In the present study, E. coli DH5α was sensitive to 1.56 mM CuSO4 without oxygen, and the acquisition of pSTM6-275 decreased the sensitivity to 6.25 mM under anaerobic conditions. E. coli can regulate copper levels by expressing chromosomal genes encoding a periplasmic copper oxidase, CueO, a cytoplasmic copper transporter, CopA, and the Cus efflux system (15). In the presence of oxygen and amino acids, copper homeostasis is achieved by CueO oxidation and CopA-mediated efflux. Cus is induced under anaerobic conditions or nutrient limitation, and E. coli CueO and Cus are not sufficient to confer Cu(I) resistance under anaerobic conditions, where nutrients are plentiful (16). Salmonella does not have a cus-encoded copper efflux pump and relies on CueO and CopA for copper homeostasis (15). CueO is sufficient for low-level Cu(I) tolerance and is required for virulence in mice (17).
The high level (800 μM) and temperature dependence of silver sensitivity shown by TW-Stm6 and E. coli pSTM6-275 transconjugants were unexpected. To our knowledge, this has not been previously reported, and the mechanism underlying this phenotype is unclear. It may be due to differences in thermoregulation of sil gene expression, the effect of temperature on the secondary structure of SilE/PcoE that alters the amount of ion binding, or changes in the outer membrane composition (18, 19).
pSTM6-275 carries a novel class I integron, In1412, that is most similar to the sul3-integron type IIIc region of E. coli plasmid pRYC306 (accession no. HQ875016.1). To evolve from pRYC306 to pSTM-275, one could hypothesize that (i) an IS440 element inserted into qacH; (ii), an IS26 element inserted in mefB, in the opposite orientation to the IS26 element near sul3; and (iii) an inversion occurred via the outward-facing IS26 elements, splitting the 8-bp direct repeats originally on the IS26 near the sul3 gene so they end up on two separate IS26 copies. IS26 is a frequently occurring and highly active insertion element in the genomes and plasmids of Salmonella spp., commonly mediating recombination events that generate new types or combinations of virulence determinants (20). Recently documented examples include novel plasmids and chromosomal loci (21–23).
The evolution of pSTM6-275 appears to be complex, and the function and regulation of many of its genes remain to be fully characterized, particularly for accessory genes, such as those involved in resistance, regulatory cross talk, and those specifying uncharacterized proteins with unknown function. Identification of other sequence type 3 (ST3) IncHI2 plasmids from human, veterinary, and environmental sources may provide further insights into the evolution of these plasmids and their role in the dissemination of resistance. This work adds to our understanding of the organization and function of an ST3 IncHI2 plasmid which may confer a fitness advantage for persistence in agricultural effluent.
ACKNOWLEDGMENTS
This research was funded by the NH&MRC through the Centres of Research Excellence program, grant no. 1079625. Yuhong Liu received a Melbourne Research Scholarship.
The funders had no role in the study design, data collection and interpretation, or the decision to submit the work for publication.
H.B.-J. and M.D.-S. designed the research; TW, LR, RH and YL performed the research; HBJ, MDS, YL and RH analyzed the results; HBJ and MDS wrote the manuscript which was read, edited and approved by all authors.
We acknowledge the contribution of the South Australia Research and Development Institute, and the Microbiological Diagnostic Laboratory, University of Melbourne.
REFERENCES
- 1.Soyer Y, Switt AM, Davis MA, Maurer J, McDonough PL, Schoonmaker-Bopp DJ, Dumas NB, Root T, Warnick LD, Grohn YT, Wiedmann M. 2009. Salmonella enterica serotype 4,5,12:i:-, an emerging Salmonella serotype that represents multiple distinct clones. J Clin Microbiol 47:3546–3556. doi: 10.1128/JCM.00546-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.García P, Malorny B, Rodicio MR, Stephan R, Hachler H, Guerra B, Lucarelli C. 2016. Horizontal acquisition of a multidrug-resistance module (R-type ASSuT) is responsible for the monophasic phenotype in a widespread clone of Salmonella serovar 4,5,12:i. Front Microbiol 7:680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Petrovska L, Mather AE, AbuOun M, Branchu P, Harris SR, Connor T, Hopkins KL, Underwood A, Lettini AA, Page A, Bagnall M, Wain J, Parkhill J, Dougan G, Davies R, Kingsley RA. 2016. Microevolution of monophasic Salmonella Typhimurium during epidemic, United Kingdom, 2005–2010. Emerg Infect Dis 22:617–624. doi: 10.3201/eid2204.150531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Weaver T, Valcanis M, Mercoulia K, Sait M, Tuke J, Kiermeier A, Hogg G, Pointon A, Hamilton D, Billman-Jacobe H. 2017. Longitudinal study of Salmonella 1,4,5,12:i:- shedding in five Australian pig herds. Prev Vet Med 136:19–28. doi: 10.1016/j.prevetmed.2016.11.010. [DOI] [PubMed] [Google Scholar]
- 5.Dyall-Smith M, Liu Y, Billman-Jacobe H. 2017. Genome sequence of an Australian monophasic Salmonella enterica subsp. enterica Typhimurium Isolate (TW-Stm6) carrying a large plasmid with multiple antimicrobial resistance genes. Genome Announc 5:e00793–17. doi: 10.1128/genomeA.00793-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Liang QH, Yin Z, Zhao YC, Liang LQ, Feng J, Zhan Z, Wang H, Song YJ, Tong YG, Wu WL, Chen WJ, Wang JL, Lingxiao JAX, Zhou DS. 2017. Sequencing and comparative genomics analysis of the IncHI2 plasmids pT5282-mphA and p112298-catA and the IncHI5 plasmid pYNKP001-dfrA. Int J Antimicrob Agents 49:709–718. doi: 10.1016/j.ijantimicag.2017.01.021. [DOI] [PubMed] [Google Scholar]
- 7.Moura A, Soares M, Pereira C, Leitao N, Henriques I, Correia A. 2009. INTEGRALL: a database and search engine for integrons, integrases and gene cassettes. Bioinformatics 25:1096–1098. doi: 10.1093/bioinformatics/btp105. [DOI] [PubMed] [Google Scholar]
- 8.Curiao T, Canton R, Garcillan-Barcia MP, de la Cruz F, Baquero F, Coque TM. 2011. Association of composite IS26-sul3 elements with highly transmissible IncI1 plasmids in extended-spectrum-beta-lactamase-producing Escherichia coli clones from humans. Antimicrob Agents Chemother 55:2451–2457. doi: 10.1128/AAC.01448-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Perreten V, Boerlin P. 2003. A new sulfonamide resistance gene (sul3) in Escherichia coli is widespread in the pig population of Switzerland. Antimicrob Agents Chemother 47:1169–1172. doi: 10.1128/AAC.47.3.1169-1172.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Fang LX, Li XP, Li L, Li SM, Liao XP, Sun J, Liu YH. 2016. Co-spread of metal and antibiotic resistance within ST3-IncHI2 plasmids from E. coli isolates of food-producing animals. Sci Rep 6:25312. doi: 10.1038/srep25312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Randall CP, Gupta A, Jackson N, Busse D, O'Neill AJ. 2015. Silver resistance in Gram-negative bacteria: a dissection of endogenous and exogenous mechanisms. J Antimicrob Chemother 70:1037–1046. doi: 10.1093/jac/dku523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gupta A, Matsui K, Lo JF, Silver S. 1999. Molecular basis for resistance to silver cations in Salmonella. Nat Med 5:183–188. doi: 10.1038/5545. [DOI] [PubMed] [Google Scholar]
- 13.Hernández-Montes G, Arguello JM, Valderrama B. 2012. Evolution and diversity of periplasmic proteins involved in copper homeostasis in gamma proteobacteria. BMC Microbiol 12:249–263. doi: 10.1186/1471-2180-12-249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Taylor DE, Levine JG. 1980. Studies of temperature-sensitive transfer and maintenance of H incompatibility group plasmids. J Gen Microbiol 116:475–484. [DOI] [PubMed] [Google Scholar]
- 15.Munson GP, Lam DL, Outten FW, O'Halloran TV. 2000. Identification of a copper-responsive two-component system on the chromosome of Escherichia coli K-12. J Bacteriol 182:5864–5871. doi: 10.1128/JB.182.20.5864-5871.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Fung DKC, Lau WY, Chan WT, Yan A. 2013. Copper efflux is induced during anaerobic amino acid limitation in Escherichia coli to protect iron-sulfur cluster enzymes and biogenesis. J Bacteriol 195:4556–4568. doi: 10.1128/JB.00543-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Achard MES, Tree JJ, Holden JA, Simpfendorfer KR, Wijburg OLC, Strugnell RA, Schembri MA, Sweet MJ, Jennings MP, McEwan AG. 2010. The multi-copper-ion oxidase CueO of Salmonella enterica serovar Typhimurium is required for systemic virulence. Infect Immun 78:2312–2319. doi: 10.1128/IAI.01208-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Asiani KR, Williams H, Bird L, Jenner M, Searle MS, Hobman JL, Scott DJ, Soultanas P. 2016. SilE is an intrinsically disordered periplasmic “molecular sponge.” Mol Microbiol 101:731–742. doi: 10.1111/mmi.13399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lundrigan MD, Earhart CF. 1984. Gene envY of Escherichia coli K-12 affects thermoregulation of major porin expression. J Bacteriol 157:262–268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Doublet B, Praud K, Weill F-X, Cloeckaert A. 2009. Association of IS26-composite transposons and complex In4-type integrons generates novel multidrug resistance loci in Salmonella genomic island 1. J Antimicrob Chemother 63:282–289. doi: 10.1093/jac/dkn500. [DOI] [PubMed] [Google Scholar]
- 21.Boland C, Bertrand S, Mattheus W, Dierick K, Jasson V, Rosseel T, Van Borm S, Mahillon J, Wattiau P. 2015. Extensive genetic variability linked to IS26 insertions in the fljB promoter region of atypical monophasic variants of Salmonella enterica serovar Typhimurium. Appl Environ Microbiol 81:3169–3175. doi: 10.1128/AEM.00270-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.García V, García P, Rodríguez I, Rodicio R, Rodicio MR. 2016. The role of IS26 in evolution of a derivative of the virulence plasmid of Salmonella enterica serovar Enteritidis which confers multiple drug resistance. Infect Genet Evol 45:246–249. doi: 10.1016/j.meegid.2016.09.008. [DOI] [PubMed] [Google Scholar]
- 23.Wong MH, Chan EW, Chen S. 2017. IS26-mediated formation of a virulence and resistance plasmid in Salmonella Enteritidis. J Antimicrob Chemother 72:2750–2754. doi: 10.1093/jac/dkx238. [DOI] [PubMed] [Google Scholar]