Abstract
Purpose
Despite the established role of O6-methyl-guanine-DNA methyltransferase (MGMT) as a marker for temozolomide response, consensus of the most reliable method to assess MGMT expression in pituitary adenomas is still missing. Currently, immunohistochemistry (IHC) assessment of formaldehyde fixed tissue samples is most widely used in a semiquantitative description. As formaldehyde fails to completely preserve nucleic acids, RCL2, an alcohol-based formaldehyde-free fixative, has been proposed as a more reliable alternative in terms of cell stability. Furthermore, as the current method of IHC is semiquantitative and observer-dependent, pyrosequencing, an objective tool to evaluate the methylation status of the MGMT promoter, has emerged as a reliable and accurate alternative. The aim of this study was to validate the current IHC method for assessment of MGMT protein expression in pituitary adenomas.
Methods
The tissue samples of 8 macroadenomas with positive IHC MGMT expression (> 50%) were investigated: first, we compared the time dependent stability of MGMT protein expression after pituitary adenoma removal between formaldehyde vs. RCL2. Then, we compared positive IHC MGMT expression with methylated promoter status using pyrosequencing.
Results
In the first 12 h after adenoma removal, tissue samples remained MGMT positive in significantly more samples when fixated with formaldehyde than with RCL2, respectively (96 vs. 81%, p = 0.025).
Conclusion
Our data confirm that the current method using formaldehyde tissue fixation and IHC reveals stable and reliable results of MGMT assessment in pituitary adenomas.
Keywords: Pituitary adenoma, MGMT, Time dependent, Promoter methylation
Introduction
Aggressive pituitary adenomas and carcinomas pose a treatment challenge because they often remain incurable despite multiple surgeries, endocrine therapy and radiation treatment.
Temozolomide (TMZ), an alkylating agent, which is the mainstay of treatment for high-grade gliomas and advanced melanoma [1–4], has also shown effectiveness against aggressive pituitary adenomas and carcinomas [5–12].
A positive response to TMZ has been found in association with downregulation of the DNA repair protein O6-methyl-guanine-DNA methyltransferase (MGMT) which removes alkylating adducts induced by TMZ and counteracts its antineoplastic action [6, 9, 11–22]. MGMT is a ubiquitously present protein in human cells but the amount of expression varies greatly within normal tissues. It is located on chromosome 10q26 and consists of 207 amino acids with a molecular mass of 21,645 Da [23–26] in human cells. The half-life of the mRNA as estimated in different cell lines is approximately 10 h [27, 28].
Despite the established role of MGMT as a possible marker for TMZ response, consensus of the most reliable method to assess MGMT expression in pituitary adenomas is still missing. Currently, immunohistochemistry (IHC) assessment of formaldehyde fixed tissue samples is most widely used in a semiquantiative description [6, 9, 11, 12, 15, 21]. Differences in methodology of MGMT immunostaining and assessment however, complicate the comparison of existing studies thus the clinical value of MGMT expression for pituitary tumours [12, 29, 30].
However, formaldehyde fails to completely preserve nucleic acids, and RCL2, an alcohol-based formaldehyde-free fixative, has been proposed as a more reliable alternative in terms of cell stability [31–33]. Furthermore, the current method is semiquantitative and observer-dependent. Pyrosequencing, an objective tool to evaluate the methylation status of the MGMT promoter which detects pyrophosphate release on nucleotide of the next complementary nucleotide, has emerged as a reliable and accurate alternative in other tumor types [34].
The aim of this study was to validate the current IHC method for assessment of MGMT protein expression in pituitary adenomas.
Materials and methods
Patient series
We evaluated 8 MGMT positive pituitary adenomas of a consecutive series of 16 patients with macroadenomas (diameter > 1 cm) at the immediate time point of tumor tissue removal. The tissue of these operations was divided into two equal parts, one for neuropathological examination the other for research purposes. The tumor samples were evaluated by the consent of the patients for further histopathological examination (EC Nr:1008/2014).
Histopathologic examinations
Immediately after surgical removal, each tumor sample was divided into 18 pieces of at least 2 mm3 size. In a time dependent mode (at 0 min, 30 min, 1 h, 2 h, 6 h, 12 h after operation) one piece was fixed in 4.5% neutral buffered formaldehyde and one in RCL2, respectively at each time. Furthermore, one piece was frozen in liquid nitrogen in case of possible unclear results or necessary evaluation at a later date.
All samples were embedded in paraffin, cut at 5 μm and stained with both hematoxylin and eosin and the periodic acid-Schiff method. Paraffin-embedded tissue sections were examined immunohistochemically using the mouse monoclonal antibodies, MGMT (Ab-1, Clone MT 3.1; Waltham, Massachusetts, USA; 1:50 dilution). Staining was performed with a Ventana BenchMark ULTRA (Ventana Medical Systems Inc., Tucson, Arizona, USA) automated immunostainer.
After deparaffinization, 5 μm thick sections have undergone heat-induced epitope retrieval in citrate buffer, pH 6.0. Tissue sections were incubated overnight at 4 °C with the primary antibody. The next day, sections were labelled with the appropriate secondary antibody, incubated with avidin-biotin-peroxidase, and visualized with a standard diaminobenzidine (DAB) detection kit (Ventana iVIEW DAB Universal Kit). Sections were then counterstained with Mayer’s hematoxylin.
Positive control tissues for IHC consisted of paraffin-embedded sections of colon cancer for immunostaining of MGMT. A positive internal control was done by staining of vascular endothelial cells. We used a non-relevant antibody of the same species (mouse) and of the same immunoglobulin isotype (IgG1) as negative control.
Assessment
For MGMT protein expression evaluation, each tumor sample was classified as: < 10, 10–25, 25–50, 50–75 and > 75% immunopositive cells [9, 35]. According to their clinical relevance these groups were further stratified to the two distinguished groups, < 50% (negative and intermediate immunoexpression), > 50% (positive immunoexpression) as proposed [35, 36].
The immunoreactivity of MGMT was evaluated under light-microscopy at ×20–40 magnification by three observers (A.M., A.W. and R.H) in a randomized and blinded mode. Only areas with highest immunoreactivity and minimal necrosis, fibrosis or other artifacts were selected for evaluation.
Pyrosequencing
For pyrosequencing analysis, 5 μm slices were cut from the formaldehyde stained and paraffin embedded blocks and were investigated at the Institute of Cancer Research, Medical University of Vienna. DNA isolation was performed using the EpiTect FFPE Lysis Kit (Qiagen, Hilden, Germany) according to manufacturer’s recommendations.
For definition of methylated/unmethylated MGMT promoter, the percentage mean value of the four investigated CpG dinucleotides (genomic sequence on chromosome 10 from 131,265,519 to 131,265,537:CGACGCCCGCAGGTCCTCG) was calculated.
A cut-off percentage of mean methylation, due to clinical relevance, was determined at 8% (< 8% MGMT promoter unmethylated; ≥ 8% MGMT promoter methylated) as previous publications described for glioblastomas [34, 37, 38].
Statistical analysis
To assess differences within formaldehyde and RCL2 samples χ2 test was used. The same method was chosen to evaluate differences between IHC and pyrosequencing formaldehyde fixated samples.
A p-value < 0.05 was considered significant. For statistical analyses SPSS® version 23.0 software (SPSS Inc., Chicago, IL, USA) has been used.
Results
The tissue samples of 8 macroadenomas with positive IHC MGMT expression (> 50%) were investigated. The histological examination showed that there were 4/8 null-cell adenomas and 4/8 gonadotropinomas.
First, we compared the stability of MGMT protein expression after pituitary adenoma removal in formaldehyde vs. RCL2 in a time dependent mode. Then, we compared positive IHC MGMT expression with methylated promoter status using pyrosequencing.
Formaldehyde vs. RCL2
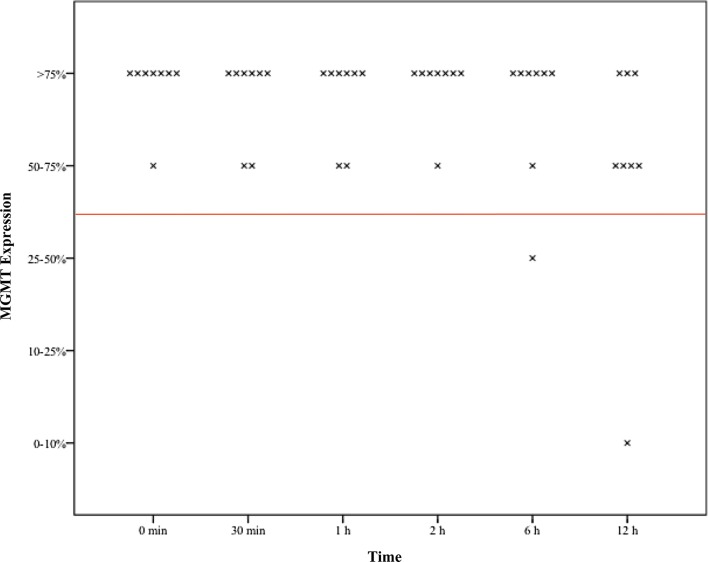
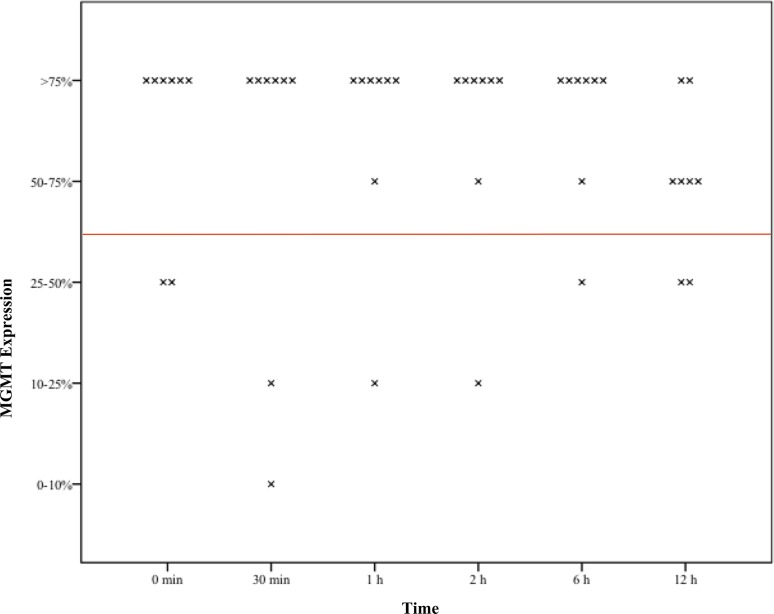
Overall eight pituitary adenoma samples fixated at six different time points, 46/48 (96%) formaldehyde cases remained positive (MGMT expression > 50%), while 39/48 (81%) cases showed stable results in the RCL2 group (p = 0.025).
In one patient MGMT continued to be positive during the whole investigation period whereas in RCL2 samples every sample showed negative (< 50%) results (p = 0.001). In one patient MGMT remained positive in 5/6 cases whereas all samples of RCL2 showed positive results (p = 0.296). In another patient, 5/6 formaldehyde samples were positive, however 2/5 RCL2 samples were positive (p = 0.376) (Table 1) (Figs. 1, 2).
Table 1.
Comparison of IHC MGMT expression > 50%, formaldehyde vs. RCL2
| Patient | Formaldehyde > 50% | RCL2 > 50% | p |
|---|---|---|---|
| Patient 1 | 6/6 | 6/6 | NS |
| Patient 2 | 6/6 | 0/6 | 0.001 |
| Patient 3 | 6/6 | 6/6 | NS |
| Patient 4 | 5/6 | 4/6 | NS |
| Patient 5 | 6/6 | 6/6 | NS |
| Patient 6 | 5/6 | 5/6 | NS |
| Patient 7 | 6/6 | 6/6 | NS |
| Patient 8 | 6/6 | 6/6 | NS |
| Total | 46/48 | 39/48 | 0.025 |
NS non significant (p > 0.05)
Fig. 1.
Boxplots of assessed time dependent MGMT expression in formaldehyde fixed tissue samples x—demonstrating the respective MGMT expression to the defined time point red line—demonstrating values above and below 50% MGMT Expression
Fig. 2.
Boxplots of assessed time dependent MGMT expression in RCL2 fixed tissue sample x—demonstrating the respective MGMT expression to the defined time point red line—demonstrating values above and below 50% MGMT expression
Pyrosequencing
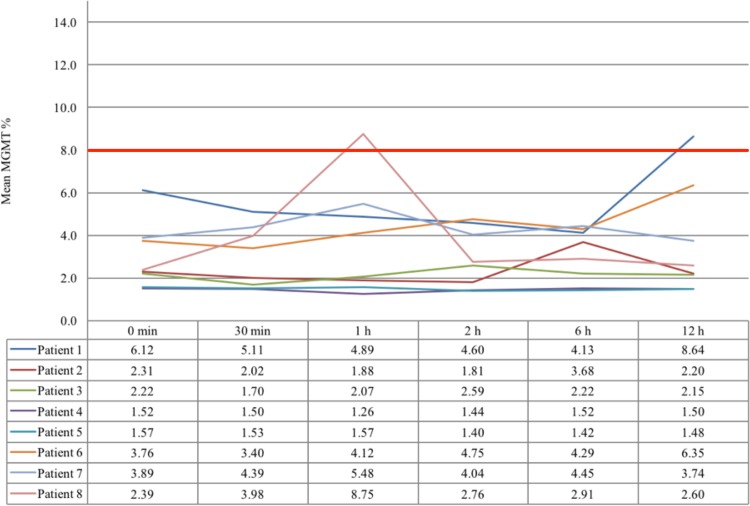
We observed an unmethylated MGMT promoter in all adenoma tissue samples immediately after the operation (1.52–6.12%). In 2/48 (4%) cases the MGMT promoter changed to methylated (defining a cut-off for methylated MGMT promoter ≥ 8%) in one patient after 1 h and in another patient after 12 h after tumor removal. MGMT pyrosequencing results are shown in detail in Fig. 3.
Fig. 3.
Time dependent MGMT pyrosequencing results bold red line—demonstrating values above and below 8% MGMT promotor methylation
Discussion
Aggressive pituitary adenomas and pituitary carcinomas unresponsive to multiple surgeries, drug treatment and radiotherapy pose a considerable therapeutic challenge. To date, chemotherapy with the alkylating drug TMZ has been the most effective treatment alternative for approximately half of these patients [5–13, 30, 36, 39–46].
In clinical routine, negative MGMT expression was found the most reliable predictive marker for tumor response to TMZ. However, MGMT expression was found to not correlate with biological tumor behavior and TMZ treatment success in every case (positive response to TMZ in only 73% of MGMT negative cases) [12]. In this respect, we validated the time-dependent stability of MGMT expression with the currently performed tissue fixation and assessment against alternative methods.
Pyrosequencing
Pyrosequencing, a relatively new technique compared to IHC, has proven stable interobserver results in promoter methylation analysis of glioblastoma samples and thus has found its way into clinical routine setting [34]. Furthermore, methylation of the CpG islands of the MGMT promoter has shown to correlate with loss of MGMT protein expression in tumor tissue [47]. This finding was also shown in pituitary adenomas using methylation analysis of the MGMT promoter. However, the frequency of methylation is considerably lower in pituitary tumors than in glioma cell lines [11]. Furthermore, pyrosequencing of promoter methylation in tumor samples can be contaminated with MGMT positive normal cells like leukocytes and endothelial cells and therefore maybe show false positive results [48–51].
A definitive cut-off for MGMT promotor methylation at the CpG position of 8% in pituitary adenomas, like in glioma cell lines, has not been defined to date. We applied the same criteria to the adenoma samples in our present series and revealed no statistical significant difference compared to MGMT positive IHC tissue samples.
However, comparing the costs between IHC and pyrosequencing at our institute, examinations revealed that taken into account only the pure costs for material (without acquisition or staff costs), the charges for one sample pyrosequencing were 63.5€ vs. 2.3€ for one IHC sample.
Immunostaining of MGMT
Differences in the technique of fixation, preservation and duration of paraffin-embedding have been reported to alter results in immunostaining of MGMT [11]. IHC has the advantage that technical expertise and equipment is widely available, in contrast to pyrosequencing. Furthermore, IHC allows differentiating between adenoma cells and non-neoplastic cells as well as identification of heterogeneity within tumor samples.
A relatively small variability of MGMT expression within a given pituitary adenoma has been attributed to the homogeneous population of adenoma cells in contrast to more heterogeneous glioma cells [52].
Time of adenoma tissue fixation
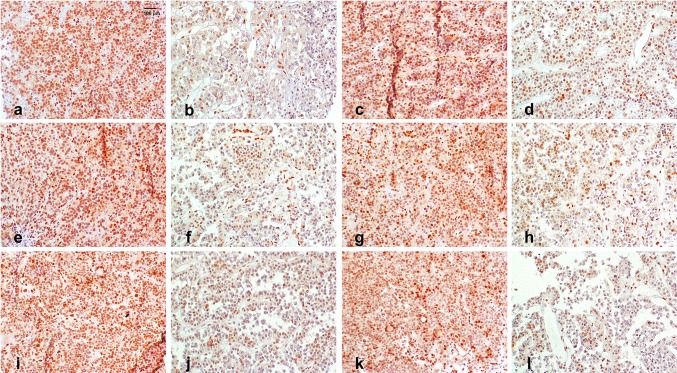
Furthermore, false negative results of MGMT expression may be due to different time points of fixation after tissue removal. The positive MGMT expression of neuron and glia cells has been found to decrease and vanish after exitus [53, 54]. We therefore investigated tissue samples over a time period of 12 h of 8 patients in detail to investigate a possible decrease of MGMT expression. We found that in formaldehyde fixation samples the results remained positive (IHC MGMT expression > 50%) in 96% in formaldehyde fixed samples (Fig. 4).
Fig. 4.
Case 2: time dependent MGMT expression, ×40 magnification (a, b) time point = 0 min after tumour removal (a formalin > 75%; b RCL2 0–10%), c, d time point = 30 min after tumour removal (c formalin 50–75%; d RCL2 10–25%), e, f time point = 1 h after tumour removal (e formalin 50–75%; f RCL2 10–25%), g, h time point = 2 h after tumour removal (g formalin > 75%; h RCL2 10–25%), i, j time point = 6 h after tumour removal (i formalin > 75%; j RCL2 25–50%), k, l time point = 12 h after tumour removal (k formalin 50–75%; l RCL2 25–50%)
Conclusion
In conclusion, our data confirm that the current method using formaldehyde tissue fixation and IHC reveals stable and reliable results of MGMT assessment in pituitary adenomas.
Acknowledgements
Open access funding provided by Medical University of Vienna. We thank Anita Brandstetter for evaluation of pyrosequencing results.
Funding
This research did not receive any specific grant from any funding agency in the public, commercial or not-for-profit sector.
Compliance with ethical standards
Conflict of interest
All authors have no financial interest/arrangement or affiliation with one or more organizations that could be perceived as a real or apparent conflict of interest in the context of the article.
References
- 1.Middleton MR, Grob JJ, Aaronson N, Fierlbeck G, Tilgen W, Seiter S, Gore M, Aamdal S, Cebon J, Coates A, Dreno B, Henz M, Schadendorf D, Kapp A, Weiss J, Fraass U, Statkevich P, Muller M, Thatcher N. Randomized phase III study of temozolomide versus dacarbazine in the treatment of patients with advanced metastatic malignant melanoma. J Clin Oncol. 2000;18(1):158–166. doi: 10.1200/JCO.2000.18.1.158. [DOI] [PubMed] [Google Scholar]
- 2.Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn U, Curschmann J, Janzer RC, Ludwin SK, Gorlia T, Allgeier A, Lacombe D, Cairncross JG, Eisenhauer E, Mirimanoff RO, European Organisation for R, Treatment of Cancer Brain., Radiotherapy T, G., National Cancer Institute of Canada Clinical Trials, G Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. New Engl J Med. 2005;352(10):987–996. doi: 10.1056/NEJMoa043330. [DOI] [PubMed] [Google Scholar]
- 3.Augustine CK, Yoo JS, Potti A, Yoshimoto Y, Zipfel PA, Friedman HS, Nevins JR, Ali-Osman F, Tyler DS. Genomic and molecular profiling predicts response to temozolomide in melanoma. Clin Cancer Res. 2009;15(2):502–510. doi: 10.1158/1078-0432.CCR-08-1916. [DOI] [PubMed] [Google Scholar]
- 4.Neyns B, Tosoni A, Hwu WJ, Reardon DA. Dose-dense temozolomide regimens: antitumor activity, toxicity, and immunomodulatory effects. Cancer. 2010;116(12):2868–2877. doi: 10.1002/cncr.25035. [DOI] [PubMed] [Google Scholar]
- 5.Bengtsson D, Schroder HD, Andersen M, Maiter D, Berinder K, Feldt Rasmussen U, Rasmussen AK, Johannsson G, Hoybye C, van der Lely AJ, Petersson M, Ragnarsson O, Burman P. Long-term outcome and MGMT as a predictive marker in 24 patients with atypical pituitary adenomas and pituitary carcinomas given treatment with temozolomide. J Clin Endocrinol Metab. 2015;100(4):1689–1698. doi: 10.1210/jc.2014-4350. [DOI] [PubMed] [Google Scholar]
- 6.Bush ZM, Longtine JA, Cunningham T, Schiff D, Jane JA, Jr, Vance ML, Thorner MO, Laws ER, Jr, Lopes MB. Temozolomide treatment for aggressive pituitary tumors: correlation of clinical outcome with O(6)-methylguanine methyltransferase (MGMT) promoter methylation and expression. J Clin Endocrinol Metab. 2010;95(11):E280–E290. doi: 10.1210/jc.2010-0441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hagen C, Schroeder HD, Hansen S, Hagen C, Andersen M. Temozolomide treatment of a pituitary carcinoma and two pituitary macroadenomas resistant to conventional therapy. Eur J Endocrinol. 2009;161(4):631–637. doi: 10.1530/EJE-09-0389. [DOI] [PubMed] [Google Scholar]
- 8.Hirohata T, Asano K, Ogawa Y, Takano S, Amano K, Isozaki O, Iwai Y, Sakata K, Fukuhara N, Nishioka H, Yamada S, Fujio S, Arita K, Takano K, Tominaga A, Hizuka N, Ikeda H, Osamura RY, Tahara S, Ishii Y, Kawamata T, Shimatsu A, Teramoto A, Matsuno A. DNA mismatch repair protein (MSH6) correlated with the responses of atypical pituitary adenomas and pituitary carcinomas to temozolomide: the national cooperative study by the Japan Society for Hypothalamic and Pituitary Tumors. J Clin Endocrinol Metab. 2013;98(3):1130–1136. doi: 10.1210/jc.2012-2924. [DOI] [PubMed] [Google Scholar]
- 9.Kovacs K, Scheithauer BW, Lombardero M, McLendon RE, Syro LV, Uribe H, Ortiz LD, Penagos LC. MGMT immunoexpression predicts responsiveness of pituitary tumors to temozolomide therapy. Acta Neuropathol. 2008;115(2):261–262. doi: 10.1007/s00401-007-0279-5. [DOI] [PubMed] [Google Scholar]
- 10.Losa M, Mazza E, Terreni MR, McCormack A, Gill AJ, Motta M, Cangi MG, Talarico A, Mortini P, Reni M. Salvage therapy with temozolomide in patients with aggressive or metastatic pituitary adenomas: experience in six cases. Eur J Endocrinol. 2010;163(6):843–851. doi: 10.1530/EJE-10-0629. [DOI] [PubMed] [Google Scholar]
- 11.McCormack AI, McDonald KL, Gill AJ, Clark SJ, Burt MG, Campbell KA, Braund WJ, Little NS, Cook RJ, Grossman AB, Robinson BG, Clifton-Bligh RJ. Low O6-methylguanine-DNA methyltransferase (MGMT) expression and response to temozolomide in aggressive pituitary tumours. Clinical endocrinology. 2009;71(2):226–233. doi: 10.1111/j.1365-2265.2008.03487.x. [DOI] [PubMed] [Google Scholar]
- 12.Raverot G, Sturm N, de Fraipont F, Muller M, Salenave S, Caron P, Chabre O, Chanson P, Cortet-Rudelli C, Assaker R, Dufour H, Gaillard S, Francois P, Jouanneau E, Passagia JG, Bernier M, Cornelius A, Figarella-Branger D, Trouillas J, Borson-Chazot F, Brue T. Temozolomide treatment in aggressive pituitary tumors and pituitary carcinomas: a French multicenter experience. J Clin Endocrinol Metab. 2010;95(10):4592–4599. doi: 10.1210/jc.2010-0644. [DOI] [PubMed] [Google Scholar]
- 13.Takeshita A, Inoshita N, Taguchi M, Okuda C, Fukuhara N, Oyama K, Ohashi K, Sano T, Takeuchi Y, Yamada S. High incidence of low O(6)-methylguanine DNA methyltransferase expression in invasive macroadenomas of Cushing’s disease. Euro J Endocrinol/Euro Fed Endocr Soc. 2009;161(4):553–559. doi: 10.1530/EJE-09-0414. [DOI] [PubMed] [Google Scholar]
- 14.Fealey ME, Scheithauer BW, Horvath E, Erickson D, Kovacs K, McLendon R, Lloyd RV. MGMT immunoexpression in silent subtype 3 pituitary adenomas: possible therapeutic implications. Endocr Pathol. 2010;21(3):161–165. doi: 10.1007/s12022-010-9120-0. [DOI] [PubMed] [Google Scholar]
- 15.Lau Q, Scheithauer B, Kovacs K, Horvath E, Syro LV, Lloyd R. MGMT immunoexpression in aggressive pituitary adenoma and carcinoma. Pituitary. 2010;13(4):367–379. doi: 10.1007/s11102-010-0249-0. [DOI] [PubMed] [Google Scholar]
- 16.Syro LV, Ortiz LD, Scheithauer BW, Lloyd R, Lau Q, Gonzalez R, Uribe H, Cusimano M, Kovacs K, Horvath E. Treatment of pituitary neoplasms with temozolomide: a review. Cancer. 2011;117(3):454–462. doi: 10.1002/cncr.25413. [DOI] [PubMed] [Google Scholar]
- 17.Salehi F, Scheithauer BW, Kros JM, Lau Q, Fealey M, Erickson D, Kovacs K, Horvath E, Lloyd RV. MGMT promoter methylation and immunoexpression in aggressive pituitary adenomas and carcinomas. J Neurooncol. 2011;104(3):647–657. doi: 10.1007/s11060-011-0532-6. [DOI] [PubMed] [Google Scholar]
- 18.McCormack AI, Wass JA, Grossman AB. Aggressive pituitary tumours: the role of temozolomide and the assessment of MGMT status. Eur J Clin Invest. 2011;41(10):1133–1148. doi: 10.1111/j.1365-2362.2011.02520.x. [DOI] [PubMed] [Google Scholar]
- 19.Moshkin O, Syro LV, Scheithauer BW, Ortiz LD, Fadul CE, Uribe H, Gonzalez R, Cusimano M, Horvath E, Rotondo F, Kovacs K. Aggressive silent corticotroph adenoma progressing to pituitary carcinoma: the role of temozolomide therapy. Hormones. 2011;10(2):162–167. doi: 10.14310/horm.2002.1307. [DOI] [PubMed] [Google Scholar]
- 20.Zuhur SS, Tanik C, Karaman O, Velet S, Cil E, Ozturk FY, Ozkayalar H, Musluman AM, Altuntas Y. MGMT immunoexpression in growth hormone-secreting pituitary adenomas and its correlation with Ki-67 labeling index and cytokeratin distribution pattern. Endocrine. 2011;40(2):222–227. doi: 10.1007/s12020-011-9485-y. [DOI] [PubMed] [Google Scholar]
- 21.Salehi F, Scheithauer BW, Kovacs K, Horvath E, Syro LV, Sharma S, Manoranjan B, Cusimano M. O-6-methylguanine-DNA methyltransferase (MGMT) immunohistochemical expression in pituitary corticotroph adenomas. Neurosurgery. 2012;70(2):491–496. doi: 10.1227/NEU.0b013e318230ac63. [DOI] [PubMed] [Google Scholar]
- 22.McCormack A, Kaplan W, Gill AJ, Little N, Cook R, Robinson B, Clifton-Bligh R. MGMT expression and pituitary tumours: relationship to tumour biology. Pituitary. 2013;16(2):208–219. doi: 10.1007/s11102-012-0406-8. [DOI] [PubMed] [Google Scholar]
- 23.Hongeng S, Brent TP, Sanford RA, Li H, Kun LE, Heideman RL. O6-Methylguanine-DNA methyltransferase protein levels in pediatric brain tumors. Clin Cancer Res. 1997;3(12 Pt 1):2459–2463. [PubMed] [Google Scholar]
- 24.Nagane M, Kobayashi K, Ohnishi A, Shimizu S, Shiokawa Y. Prognostic significance of O6-methylguanine-DNA methyltransferase protein expression in patients with recurrent glioblastoma treated with temozolomide. Japn J Clin Oncol. 2007;37(12):897–906. doi: 10.1093/jjco/hym132. [DOI] [PubMed] [Google Scholar]
- 25.Kishida Y, Natsume A, Toda H, Toi Y, Motomura K, Koyama H, Matsuda K, Nakayama O, Sato M, Suzuki M, Kondo Y, Wakabayashi T. Correlation between quantified promoter methylation and enzymatic activity of O6-methylguanine-DNA methyltransferase in glioblastomas. Tumour Biol. 2012;33(2):373–381. doi: 10.1007/s13277-012-0319-1. [DOI] [PubMed] [Google Scholar]
- 26.Pieper RO. Understanding and manipulating O6-methylguanine-DNA methyltransferase expression. Pharmacol Ther. 1997;74(3):285–297. doi: 10.1016/S0163-7258(97)00003-X. [DOI] [PubMed] [Google Scholar]
- 27.Kroes RA, Erickson LC. The role of mRNA stability and transcription in O6-methylguanine DNA methyltransferase (MGMT) expression in Mer + human tumor cells. Carcinogenesis. 1995;16(9):2255–2257. doi: 10.1093/carcin/16.9.2255. [DOI] [PubMed] [Google Scholar]
- 28.Fritz G, Tano K, Mitra S, Kaina B. Inducibility of the DNA repair gene encoding O6-methylguanine-DNA methyltransferase in mammalian cells by DNA-damaging treatments. Mol Cell Biol. 1991;11(9):4660–4668. doi: 10.1128/MCB.11.9.4660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Raverot G, Castinetti F, Jouanneau E, Morange I, Figarella-Branger D, Dufour H, Trouillas J, Brue T. Pituitary carcinomas and aggressive pituitary tumours: merits and pitfalls of temozolomide treatment. Clin Endocrinol (Oxf) 2012;76(6):769–775. doi: 10.1111/j.1365-2265.2012.04381.x. [DOI] [PubMed] [Google Scholar]
- 30.Annamalai AK, Dean AF, Kandasamy N, Kovacs K, Burton H, Halsall DJ, Shaw AS, Antoun NM, Cheow HK, Kirollos RW, Pickard JD, Simpson HL, Jefferies SJ, Burnet NG, Gurnell M. Temozolomide responsiveness in aggressive corticotroph tumours: a case report and review of the literature. Pituitary. 2012;15(3):276–287. doi: 10.1007/s11102-011-0363-7. [DOI] [PubMed] [Google Scholar]
- 31.Bellet V, Boissiere F, Bibeau F, Desmetz C, Berthe ML, Rochaix P, Maudelonde T, Mange A, Solassol J. Proteomic analysis of RCL2 paraffin-embedded tissues. J Cell Mol Med. 2008;12(5B):2027–2036. doi: 10.1111/j.1582-4934.2008.00186.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Delfour C, Roger P, Bret C, Berthe ML, Rochaix P, Kalfa N, Raynaud P, Bibeau F, Maudelonde T, Boulle N. RCL2, a new fixative, preserves morphology and nucleic acid integrity in paraffin-embedded breast carcinoma and microdissected breast tumor cells. J Mol Diagn. 2006;8(2):157–169. doi: 10.2353/jmoldx.2006.050105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Denouel A, Boissiere-Michot F, Rochaix P, Bibeau F, Boulle N. An alternative fixative to formalin fixation for molecular applications: the RCL2((R))-CS100 approach. Methods Mol Biol. 2011;724:297–307. doi: 10.1007/978-1-61779-055-3_19. [DOI] [PubMed] [Google Scholar]
- 34.Preusser M, Berghoff AS, Manzl C, Filipits M, Weinhausel A, Pulverer W, Dieckmann K, Widhalm G, Wohrer A, Knosp E, Marosi C, Hainfellner JA. Clinical neuropathology practice news 1–2014: pyrosequencing meets clinical and analytical performance criteria for routine testing of MGMT promoter methylation status in glioblastoma. Clin Neuropathol. 2014;33(1):6–14. doi: 10.5414/NP300730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Widhalm G, Wolfsberger S, Preusser M, Woehrer A, Kotter MR, Czech T, Marosi C, Knosp E. O(6)-methylguanine DNA methyltransferase immunoexpression in nonfunctioning pituitary adenomas: are progressive tumors potential candidates for temozolomide treatment? Cancer. 2009;115(5):1070–1080. doi: 10.1002/cncr.24053. [DOI] [PubMed] [Google Scholar]
- 36.Micko ASG, Wohrer A, Hoftberger R, Vila G, Marosi C, Knosp E, Wolfsberger S. MGMT and MSH6 immunoexpression for functioning pituitary macroadenomas. Pituitary. 2017 doi: 10.1007/s11102-017-0829-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Felsberg J, Thon N, Eigenbrod S, Hentschel B, Sabel MC, Westphal M, Schackert G, Kreth FW, Pietsch T, Loffler M, Weller M, Reifenberger G, Tonn JC, German Glioma N. Promoter methylation and expression of MGMT and the DNA mismatch repair genes MLH1, MSH2, MSH6 and PMS2 in paired primary and recurrent glioblastomas. Int J Cancer. 2011;129(3):659–670. doi: 10.1002/ijc.26083. [DOI] [PubMed] [Google Scholar]
- 38.Reifenberger G, Hentschel B, Felsberg J, Schackert G, Simon M, Schnell O, Westphal M, Wick W, Pietsch T, Loeffler M, Weller M, German Glioma N. Predictive impact of MGMT promoter methylation in glioblastoma of the elderly. Int J Cancer. 2012;131(6):1342–1350. doi: 10.1002/ijc.27385. [DOI] [PubMed] [Google Scholar]
- 39.Mohammed S, Kovacs K, Mason W, Smyth H, Cusimano MD. Use of temozolomide in aggressive pituitary tumors: case report. Neurosurgery. 2009;64(4):E773–E774. doi: 10.1227/01.NEU.0000339115.12803.4E. [DOI] [PubMed] [Google Scholar]
- 40.Moyes VJ, Alusi G, Sabin HI, Evanson J, Berney DM, Kovacs K, Monson JP, Plowman PN, Drake WM. Treatment of Nelson’s syndrome with temozolomide. Eur J Endocrinol. 2009;160(1):115–119. doi: 10.1530/EJE-08-0557. [DOI] [PubMed] [Google Scholar]
- 41.Curto L, Torre ML, Ferrau F, Pitini V, Altavilla G, Granata F, Longo M, Hofland LJ, Trimarchi F, Cannavo S. Temozolomide-induced shrinkage of a pituitary carcinoma causing Cushing’s disease–report of a case and literature review. Sci World J. 2010;10:2132–2138. doi: 10.1100/tsw.2010.210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Ersen A, Syro LV, Penagos L, Uribe H, Scheithauer BW, Ortiz LD, Rotondo F, Horvath E, Kovacs K. Non-uniform response to temozolomide therapy in a pituitary gonadotroph adenoma. Can J Neurol Sci. 2012;39(5):683–685. doi: 10.1017/S0317167100018242. [DOI] [PubMed] [Google Scholar]
- 43.Ortiz LD, Syro LV, Scheithauer BW, Ersen A, Uribe H, Fadul CE, Rotondo F, Horvath E, Kovacs K. Anti-VEGF therapy in pituitary carcinoma. Pituitary. 2012;15(3):445–449. doi: 10.1007/s11102-011-0346-8. [DOI] [PubMed] [Google Scholar]
- 44.Whitelaw BC, Dworakowska D, Thomas NW, Barazi S, Riordan-Eva P, King AP, Hampton T, Landau DB, Lipscomb D, Buchanan CR, Gilbert JA, Aylwin SJ. Temozolomide in the management of dopamine agonist-resistant prolactinomas. Clin Endocrinol. 2012;76(6):877–886. doi: 10.1111/j.1365-2265.2012.04373.x. [DOI] [PubMed] [Google Scholar]
- 45.Batisse M, Raverot G, Maqdasy S, Durando X, Sturm N, Montoriol PF, Kemeny JL, Chazal J, Trouillas J, Tauveron I. Aggressive silent GH pituitary tumor resistant to multiple treatments, including temozolomide. Cancer Investig. 2013;31(3):190–196. doi: 10.3109/07357907.2013.775293. [DOI] [PubMed] [Google Scholar]
- 46.Murakami M, Mizutani A, Asano S, Katakami H, Ozawa Y, Yamazaki K, Ishida Y, Takano K, Okinaga H, Matsuno A. A mechanism of acquiring temozolomide resistance during transformation of atypical prolactinoma into prolactin-producing pituitary carcinoma: case report. Neurosurgery. 2011;68(6):E1761–E1767. doi: 10.1227/NEU.0b013e318217161a. [DOI] [PubMed] [Google Scholar]
- 47.Esteller M, Hamilton SR, Burger PC, Baylin SB, Herman JG. Inactivation of the DNA repair gene O6-methylguanine-DNA methyltransferase by promoter hypermethylation is a common event in primary human neoplasia. Cancer Res. 1999;59(4):793–797. [PubMed] [Google Scholar]
- 48.Sasai K, Nodagashira M, Nishihara H, Aoyanagi E, Wang L, Katoh M, Murata J, Ozaki Y, Ito T, Fujimoto S, Kaneko S, Nagashima K, Tanaka S. Careful exclusion of non-neoplastic brain components is required for an appropriate evaluation of O6-methylguanine-DNA methyltransferase status in glioma: relationship between immunohistochemistry and methylation analysis. Am J Surg Pathol. 2008;32(8):1220–1227. doi: 10.1097/PAS.0b013e318164c3f0. [DOI] [PubMed] [Google Scholar]
- 49.Silber JR, Blank A, Bobola MS, Mueller BA, Kolstoe DD, Ojemann GA, Berger MS. Lack of the DNA repair protein O6-methylguanine-DNA methyltransferase in histologically normal brain adjacent to primary human brain tumors. Proc Natl Acad Sci USA. 1996;93(14):6941–6946. doi: 10.1073/pnas.93.14.6941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Cao VT, Jung TY, Jung S, Jin SG, Moon KS, Kim IY, Kang SS, Park CS, Lee KH, Chae HJ. The correlation and prognostic significance of MGMT promoter methylation and MGMT protein in glioblastomas. Neurosurgery. 2009;65(5):866–875. doi: 10.1227/01.NEU.0000357325.90347.A1. [DOI] [PubMed] [Google Scholar]
- 51.Preusser M, Charles Janzer R, Felsberg J, Reifenberger G, Hamou MF, Diserens AC, Stupp R, Gorlia T, Marosi C, Heinzl H, Hainfellner JA, Hegi M. Anti-O6-methylguanine-methyltransferase (MGMT) immunohistochemistry in glioblastoma multiforme: observer variability and lack of association with patient survival impede its use as clinical biomarker. Brain Pathol. 2008;18(4):520–532. doi: 10.1111/j.1750-3639.2008.00153.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Rodriguez FJ, Thibodeau SN, Jenkins RB, Schowalter KV, Caron BL, O’Neill BP, James CD, Passe S, Slezak J, Giannini C. MGMT immunohistochemical expression and promoter methylation in human glioblastoma. Appl Immunohistochem Mol Morphol. 2008;16(1):59–65. doi: 10.1097/PAI.0b013e31802fac2f. [DOI] [PubMed] [Google Scholar]
- 53.Nakasu S, Fukami T, Baba K, Matsuda M. Immunohistochemical study for O6-methylguanine-DNA methyltransferase in the non-neoplastic and neoplastic components of gliomas. J Neurooncol. 2004;70(3):333–340. doi: 10.1007/s11060-004-9170-6. [DOI] [PubMed] [Google Scholar]
- 54.Hsu CY, Ho HL, Chang-Chien YC, Chang YW, Ho DM. MGMT promoter methylation in non-neoplastic brain. J Neurooncol. 2015;121(3):459–467. doi: 10.1007/s11060-014-1663-3. [DOI] [PubMed] [Google Scholar]