Complex interactions of gut microbiota activate the production of bioactive polyamine, which exerts strong effects on the host.
Abstract
Metabolites of the intestinal microbiota are thought to be generated through metabolic pathways spanning multiple taxa of intestinal bacteria. We have previously shown that the level of putrescine, a polyamine found abundantly in the human intestinal lumen, is increased in the colonic lumen following administration of arginine and the probiotic Bifidobacterium sp.; however, the underlying mechanism remained poorly understood. We report a novel pathway for putrescine production from arginine through agmatine involving the collaboration of two bacterial groups, and triggered by environmental acidification (drop in pH to below 6.5 from neutral). This pathway comprises the acid tolerance system of Escherichia coli, representing bacteria that have an arginine-dependent acid resistance system; the energy production system of Enterococcus faecalis, representing bacteria that have an agmatine deiminase system; and the acid production system of the acid-producing bacteria, represented by Bifidobacterium spp. This pathway is unique in that it represents a relationship between the independent survival strategies of multiple bacteria.
INTRODUCTION
Metabolites derived from gut microbes are associated with host health and disease (1), and probiotic bacteria are expected to optimize the metabolite composition of the intestinal microbiota by affecting their biochemical reactions. Polyamines (for example, putrescine and spermidine) are important metabolites of intestinal bacteria, and putrescine is present in the intestinal lumen in 0.5 to 1 mM concentrations in healthy humans (2, 3). These organic cations are found in almost all organisms (4, 5) and are required for cell growth and proliferation. Polyamines promote animal health through induction of autophagy (6), suppression of inflammation (7), improvement of cognitive function (8), and suppression of the progression of heart failure (9). High levels of polyamines are also observed in tumors (10), and the selective inhibition of ornithine decarboxylase, a key enzyme in polyamine synthesis, is effective against malignancies, including colon cancer (10, 11). Hence, whether or not polyamines in the intestinal lumen are harmful or beneficial for health, strategies to regulate and optimize their concentrations in the intestinal tract are likely to have practical applications. Our group explored the relationship between polyamines and prevention of senescence with the ultimate aim to create novel foods and/or technologies that facilitate bacterial putrescine production to increase the polyamine levels in elderly individuals who have weakened polyamine biosynthesis systems. In contrast, if the mechanism of polyamine production in the intestine is elucidated, then there is a possibility that polyamine concentration can be decreased in patients with colon cancer, for whom decreased polyamine levels in the colon have been found to be beneficial. As food-derived polyamines are absorbed before they reach the lower intestine (12), most polyamines in this region of the gut are produced by intestinal microbiota (13); therefore, regulation of microbial polyamine metabolism and transport is required to maintain polyamine concentrations at 0.5 to 1 mM, the normal level in healthy individuals, in the intestinal lumen of individuals with low level of intestinal luminal polyamines, represented by the elderly (2). However, the mechanisms underlying the up-regulation of colonic luminal polyamines following administration of arginine and/or probiotics (14, 15) are poorly understood at the genetic level, precluding the regulation of gene expression to optimize the polyamine concentrations in the intestinal lumen.
Putrescine is biosynthesized in bacterial cells from agmatine, which is produced by decarboxylation of arginine by the arginine decarboxylases SpeA and AdiA. Two metabolic pathways for the conversion of agmatine to putrescine have been reported to date: The first is the agmatine ureohydrolase (SpeB) pathway, where agmatine is directly converted to putrescine; the second is the N-carbamoylputrescine pathway, which involves N-carbamoylputrescine production from agmatine by agmatine iminohydrolase and its conversion to putrescine by N-carbamoylputrescine amidohydrolase. In silico genome analysis of the 56 most abundant bacterial species in the human microbiome (16) has predicted that many species are unable to produce putrescine from arginine using their own enzymes, owing to incomplete synthetic pathways (17, 18), in turn suggesting the existence of a metabolic pathway spanning multiple bacterial species in the gut. However, to our knowledge, there currently is no genetic proof for such a hybrid metabolic pathway. Moreover, even if a microorganism has the synthetic pathway of a specific metabolite, it does not necessarily secrete this metabolite. To confirm the release of a target metabolite from specific bacteria, it is necessary to analyze gene expression related to both the synthetic pathway and the transporters of the target metabolite. However, it is very difficult to identify links between expression in synthetic pathways and that of specific metabolite transporters using metagenomics and transcriptomics alone. Here, by using traditional cultivation and molecular biological techniques, we present evidence of novel hybrid pathways for the synthesis and release of target metabolites, and demonstrate that this system consists of independent pathways involving various bacterial species, rather than of pathways for metabolite synthesis and release within a single bacterial species.
Probiotics are defined as live microbial food supplements that confer a health benefit to the host by improving intestinal balance (19), such as by influencing the composition of metabolites derived from the intestinal microbiota. Bifidobacterium spp. is frequently used in probiotic products (20). Supplementation of Bifidobacterium animalis subsp. lactis has been shown to increase the colonic polyamine concentration (15). However, Bifidobacterium spp. do not have homologs of enzymes involved in polyamine biosynthesis; therefore, the increased concentration of polyamine in the intestinal lumen likely results from the activation of polyamine biosynthesis by indigenous gut microbiota.
Here, we used traditional cultivation bacterial culture methods, molecular biological techniques, and literature searches for combinations of commensal bacterial metabolic pathways to identify a novel putrescine production pathway, from arginine through agmatine, shared between two bacterial species: Enterococcus faecalis and Escherichia coli. Activity of this pathway is stimulated by environmental acidification (drop in pH to below 6.5 from neutral) induced by acid-producing bacteria, represented by Bifidobacterium spp. Putrescine, which is biosynthesized by the two bacteria, is a byproduct of two independent survival strategies in the acidic environment caused by acid-producing bacteria, represented by Bifidobacterium spp., namely, the acid resistance of E. coli and the energy production of En. faecalis.
RESULTS
Induction of putrescine production by mixed microbial cultures
First, we examined the difference between the effects of single and mixed bacterial cultures on putrescine production. The putrescine concentration in the culture supernatant from a mixed microbial culture including 14 species from the four main phyla in the human intestine in medium containing arginine was 8.5 mM, which was more than 10 times higher than the average concentration (0.21 mM; range from 0 to 1.02 mM) for each species cultured individually (Fig. 1A). To determine which bacteria were responsible for the induced putrescine production, we analyzed the putrescine concentration in medium from cocultured pairs of bacterial species. The highest concentration of 8.0 mM was found in medium from mixed E. coli and En. faecalis culture (Fig. 1B), and this concentration was over eight times higher than those observed when either species was cultured separately (fig. S1A). The putrescine production in E. coli and En. faecalis mixed culture was highest when the pH of the medium was 5.5 (fig. S1B), suggesting that the induction of putrescine production might be attributed to a bacterial acid resistance system. E. coli has an acid resistance system involving arginine and agmatine, while En. faecalis has an agmatine utilization pathway that contributes indirectly to mitigating growth medium acidification as a consequence of adenosine 5′-triphosphate (ATP) production (see below) (21); notably, agmatine is an intermediate between arginine and putrescine in these pathways.
Fig. 1. Induction of putrescine production by mixed microbial cultures.
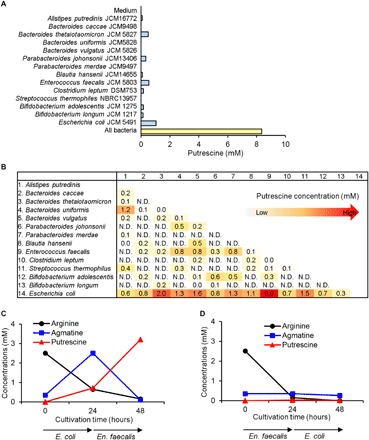
(A) Extracellular putrescine concentrations in cocultures and individual cultures of 14 bacterial strains in GAM medium supplemented with 2 mM arginine. (B) Extracellular putrescine concentrations in cocultures of each pair of 14 bacterial strains (total 91 combinations). N.D. means not detected; that is, the levels fell below the minimum detectable amount. (C) Extracellular concentration of l-arginine, putrescine, and agmatine in the medium after culture of E. coli for 24 hours (0 to 24 hours), followed by culture of En. faecalis for another 24 hours (24 to 48 hours) in the same medium; E. coli was removed by filtration after 24 hours. (D) Extracellular concentrations of l-arginine, putrescine, and agmatine in the medium after culture of En. faecalis for 24 hours (0 to 24 hours), followed by culture of E. coli for another 24 hours (24 to 48 hours) in the same medium. En. faecalis was removed by filtration after 24 hours.
To assess the possibility that either E. coli or En. faecalis synthesizes putrescine from arginine via extracellular agmatine released by the other bacterium, we used the spent medium from culture of each bacterium to culture the other and quantified arginine, putrescine, and agmatine. During cultivation of E. coli, arginine decreased from 2.51 to 0.64 mM (26% of the initial concentration), agmatine increased from 0.35 to 2.51 mM, and putrescine increased from 0 to 0.71 mM (0 to 24 hours; Fig. 1C). When spent medium from E. coli cultivation was used to culture En. faecalis, agmatine decreased from 2.51 to 0.10 mM (3.9% of the initial concentration), while putrescine increased from 0.71 to 3.21 mM (24 to 48 hours; Fig. 1C). In contrast, when spent medium from En. faecalis culture was used to cultivate E. coli, neither agmatine nor putrescine production was observed, and arginine was almost completely absent (Fig. 1D). Together, these results suggest that induction of putrescine production from arginine by a mixed culture of E. coli and En. faecalis is due to the transmission of agmatine, a reactive intermediate between arginine and putrescine, from E. coli to En. faecalis.
Putrescine production by sequential reactions in E. coli and En. faecalis
E. coli has an acid resistance system composed of arginine decarboxylase (AdiA) and an arginine-agmatine antiporter (AdiC) (22); the equivalent function in En. faecalis is achieved by agmatine deiminase (AguA) and an agmatine-putrescine antiporter (AguD), although it was previously reported that acidic stress does not induce this system (21). We hypothesized that the two acid resistance systems of these individual bacteria could combine, resulting in induced putrescine production. In this hypothetical pathway (Fig. 2), arginine is taken up from the medium into E. coli cells by AdiC and converted to agmatine by AdiA/SpeA. The generated agmatine is then exported from the E. coli cells to the medium via AdiC. Next, En. faecalis takes up the agmatine from the medium using AguD. Agmatine is then converted to putrescine by the sequential actions of AguA and a putrescine carbamoyl transferase (AguB). The generated putrescine is exported from En. faecalis cells via AguD. Hence, the combination of these metabolic and transport pathways of the two bacteria could promote the production of putrescine.
Fig. 2. Hypothetical putrescine production pathway consisting of the acid resistance system of E. coli and an ATP synthesis system of En. faecalis.
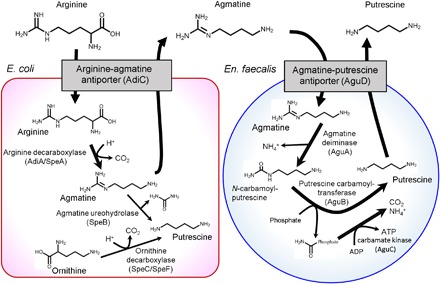
A putrescine biosynthesis pathway consisting of sequential reactions accomplished by E. coli and En. faecalis. Details are shown in the text.
To test this hypothesis, we deleted the adiA gene and complemented it in E. coli, which we then cocultured with wild-type En. faecalis (Fig. 3A). In LB medium containing 2 mM l-arginine, 1.5 g/liter d-glucose, and 0.5 g/liter l-cysteine-hydrochloride (LB-RGC medium), cocultures of wild-type E. coli (LKM10096) and wild-type En. faecalis (SK947) produced 1.82 mM putrescine in the supernatant. When adiA-deleted E. coli (SK914) was cocultured with wild-type En. faecalis (SK947), putrescine production was abolished. Coculture of adiA-complemented E. coli (SK912) and wild-type En. faecalis (SK947) completely restored putrescine production. Similarly, when adiC-deleted E. coli (LKM10097) was cocultured with wild-type En. faecalis (SK947), putrescine production was abolished, and in complementation experiments using adiC-complemented E. coli (LKM10100) and wild-type En. faecalis (SK947), putrescine production was completely restored (Fig. 3B).
Fig. 3. Validation of the hypothesis of sequential reactions in E. coli and En. faecalis using bacteria whose genes encoding transporters and enzymes were deleted or complemented.
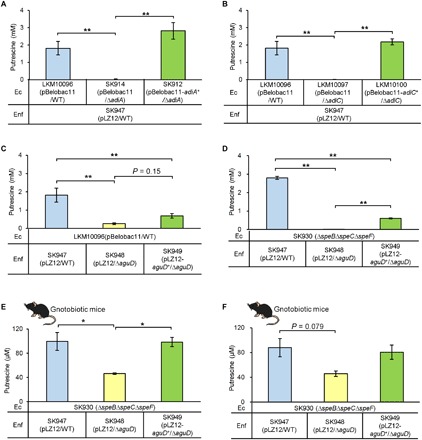
(A and B) Extracellular putrescine concentration in cocultures of wild-type (WT) En. faecalis (Enf; SK947) and E. coli (Ec) including gene knockout mutants or complementary transformants of arginine decarboxylase (adiA) of E. coli (A) or arginine-agmatine antiporter (adiC ) of E. coli (B). (C) Extracellular putrescine concentrations in cocultures of wild-type E. coli (LKM10096) and En. faecalis including the wild type, gene knockout mutants, or complementary transformants of agmatine-putrescine antiporter (aguD). (D) Extracellular putrescine concentration in cocultures of putrescine-deficient E. coli (SK930) and wild-type En. faecalis (SK947). Coculture of these bacteria was conducted under anaerobic conditions at 37°C for 24 hours in LB-RGC medium. (E and F) Putrescine concentrations in the content of cecal lumen (E) and colonic lumen (F) of gnotobiotic mice colonized with putrescine-deficient E. coli (SK930) and wild-type En. faecalis (SK947). Error bars represent SEs. *P < 0.05 and **P < 0.01, one-way analysis of variance (ANOVA) with Tukey’s test.
Similarly, we investigated the role of the aguD gene in En. faecalis (Fig. 3C). When wild-type E. coli (LKM10096) was cocultured with aguD-deleted En. faecalis (SK948), the putrescine concentration decreased to 14% (0.26 mM) of the value when wild-type E. coli was cocultured with wild-type En. faecalis (SK947) (1.82 mM). In the complementation experiment using wild-type E. coli (LKM10096) and aguD-complemented En. faecalis (SK949), the putrescine concentration was restored to 0.68 mM (37% of the value from coculture of wild-type E. coli and wild-type En. faecalis). These results demonstrated that a hybrid system composed of E. coli and En. faecalis enzymes and transporters transported and metabolized arginine to putrescine in the medium (Fig. 2).
The hybrid system for putrescine production by two bacteria in the mouse intestinal tract
To determine whether the putrescine production pathway (Fig. 2) is functional in the mouse intestine, we performed experiments using gnotobiotic mice colonized with E. coli and En. faecalis. As putrescine production was not completely abolished when wild-type E. coli and aguD-deleted En. faecalis were cocultured (Fig. 3C), putrescine-deficient E. coli (pBelobac11/ΔspeBΔspeCΔspeF) (SK930), which produces agmatine but not putrescine, owing to the deletion of all putrescine biosynthetic genes, was used for this experiment. When putrescine-deficient E. coli (SK930) and wild-type En. faecalis (SK947) were cocultured in LB-RGC medium, the putrescine concentration in the culture supernatant reached 2.8 mM (Fig. 3D). In contrast, when putrescine-deficient E. coli (SK930) and aguD-deleted En. faecalis (SK948) were cocultured, putrescine production was completely abolished. Coculture of putrescine-deficient E. coli (SK930) and aguD-complemented En. faecalis (SK949) led to putrescine production at a level of 21% of that in coculture of putrescine-deficient E. coli (SK930) and wild-type En. faecalis (SK947) (Fig. 3D). Next, we administered each pair of bacterial strains to germ-free mice and quantified putrescine concentrations in the contents of their ceca and colons. In gnotobiotic mice colonized with putrescine-deficient E. coli (SK930) and wild-type En. faecalis (SK930), the cecal putrescine concentration was 100 μM (Fig. 3E). In contrast, that in mice colonized with putrescine-deficient E. coli (SK930) and aguD-deleted En. faecalis (SK948) dropped to 46 μM but was restored to 99 μM in mice colonized with putrescine-deficient E. coli (SK930) and aguD-complemented En. faecalis (SK949) (Fig. 3E). Colonic luminal concentrations of putrescine showed a similar trend, although the differences were not statistically significant (P = 0.079) (Fig. 3F). These results indicated that a system composed of enzymes and transporters of E. coli and En. faecalis was functional in the mouse intestine.
Physiological importance of the hybrid system involving two bacterial species for putrescine production by E. coli and En. faecalis
The increased production of putrescine observed in this study appeared to result from a combination of the acid resistance systems of E. coli and En. faecalis. It is known that E. coli has arginine-dependent “acid resistance systems” (AR3) that exchange intracellular agmatine for extracellular arginine (23). When wild-type E. coli (MG1655) was monocultured in arginine-containing medium at different pH values, the agmatine concentration decreased gradually with a pH increase from 5 to 9 (bacterial growth was inhibited at pH 4 and 9) (fig. S2A), demonstrating that the production of agmatine from arginine in E. coli depends on this system. However, wild-type En. faecalis (V583) monocultured in medium at different pH values (5 to 8) containing agmatine exhibited no significant difference in putrescine production (bacterial growth was inhibited at pH 4 and 9) (fig. S2B), demonstrating that environmental pH did not influence putrescine production from agmatine in En. faecalis. In contrast, putrescine production by wild-type En. faecalis (SK947) was inhibited in the presence of glucose (fig. S2C), indicating that this pathway is involved in energy production. These results are consistent with those of a previous report that showed that this pathway is activated to obtain energy but is not strongly correlated with protection of bacterial cells against acidic stress (21). The viable count of wild-type En. faecalis (SK948) monocultured in medium containing 5 mM agmatine at pH 6 was significantly higher than that of aguD-deleted En. faecalis monocultured in the same medium (fig. S2D). Furthermore, the viable count of En. faecalis in coculture of wild-type En. faecalis (SK947) with putrescine-deficient E. coli (SK930) was significantly higher than that in coculture of aguD-deleted En. faecalis (SK948) with putrescine-deficient E. coli (SK930) (P < 0.05). The viable count of En. faecalis was restored when aguD-complemented En. faecalis (SK949) was cocultured with putrescine-deficient E. coli (SK930) (fig. S2E). These results indicate that En. faecalis grows using agmatine produced by E. coli from arginine.
Selection of Bifidobacterium strains that induce putrescine production through the hybrid system comprisingE. coli and En. faecalis
We screened for Bifidobacterium spp. that strongly produced putrescine and added nine identified strains to the mixed cultures of the putrescine-deficient E. coli (SK930) strain and wild-type En. faecalis (SK947). We determined the concentrations of putrescine in the culture supernatants of these mixtures (Fig. 4A). Among cocultures tested, putrescine-deficient E. coli (SK930), wild-type En. faecalis (V583), and B. animalis subsp. lactis LKM512 produced the highest putrescine concentration. Moreover, putrescine concentrations in the presence of the two strains of B. animalis subsp. lactis (LKM512 and DSM10140T) differed, indicating that induction of putrescine production by Bifidobacterium spp. is strain-dependent. Bifidobacterium spp. produce lactate and acetate (24), which decrease the pH in the intestinal lumen (25). Next, we investigated the mechanism by which B. animalis subsp. lactis LKM512 induced putrescine production in cocultures of En. faecalis and E. coli. The pH of culture supernatants from individually cultured putrescine-deficient E. coli (SK930), wild-type En. faecalis (V583), and B. animalis subsp. lactis LKM512 decreased from an initial value of 6.5 to approximately 5.2, 5.3, and 4.4, respectively, after 24 hours (Fig. 4B), and their putrescine concentrations were very low (Fig. 4C). However, the median pH of supernatants from cocultures of these three strains was 5.0, which was significantly lower than that of cocultures without LKM512 (pH 5.9) (Fig. 4B). Furthermore, the putrescine concentration in culture supernatants from cocultures of the three strains was more than twice that from cocultures without LKM512 (P < 0.01; Fig. 4C), suggesting that the acidic environment created by B. animalis subsp. lactis LKM512 probably induced putrescine production in cocultures of E. coli and En. faecalis. Nevertheless, it could be possible that factors other than environmental acidity are involved in this process because the extracellular putrescine concentration positively correlated with the number of both E. coli and En. faecalis in the coculture system (fig. S3A). Notably, in the culture with B. animalis subsp. lactis LKM512 or B. adolescentis JCM1275T, which induced higher production of putrescine, large numbers of E. coli [108.5 colony-forming units (CFU)/ml or more] and En. faecalis (107.9 CFU/ml or more) were detected. However, the culture with B. longum JCM1217T or B. pseudocatenulatum JCM1200T, which induced only slight putrescine production, contained fewer than 1/10 of the E. coli and En. faecalis (less than 106.8 CFU/ml) than those with B. animalis subsp. lactis LKM512 or B. adolescentis JCM1275T. All cultures reached pH 5.5 at which maximal putrescine production occurred within 12 hours (fig. S3B).
Fig. 4. Effects of acidification by bifidobacteria on putrescine production by E. coli and En. faecalis.
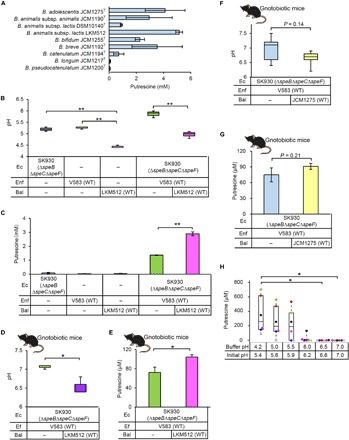
(A) Extracellular putrescine concentration in cocultures of putrescine-deficient E. coli (SK930), wild-type En. faecalis (V583), and each Bifidobacterium sp. (B and C) Extracellular pH (B) and putrescine concentration (C) in cocultures of putrescine-deficient E. coli (SK930), wild-type En. faecalis (V583), and B. animalis subsp. lactis LKM512 (Bal). Coculture of these bacteria was conducted under anaerobic conditions at 37°C for 24 hours in LB medium containing 2 mM l-arginine, d-glucose (1.5 g/liter), galacto-oligosaccharide (5 g/liter), 2 mM MgSO4, 60 mM NH4Cl, and LB-RGC (0.5 g/liter) (pH 6.5). (D and E) Fecal pH (D) and putrescine concentration (E) in gnotobiotic mice colonized with putrescine-deficient E. coli (SK930), wild-type En. faecalis (V583), and B. animalis subsp. lactis LKM512. (F and G) Fecal pH (F) and putrescine (G) concentration in gnotobiotic mice inoculated with putrescine-deficient E. coli (SK930), wild-type En. faecalis (V583), and B. adolescentis JCM1275T. (H) Extracellular putrescine concentration in human feces incubated at different pHs. Error bars represent SEs. *P < 0.05 and **P < 0.01, one-way ANOVA with Tukey’s test, Student’s t tests, and Steel-Dwass test.
Induction of polyamine production in the feces of gnotobiotic mice by oral administration of Bifidobacterium spp.
To test the effect of the environment created by Bifidobacterium spp. on putrescine production in vivo, gnotobiotic mice were colonized with the putrescine-deficient E. coli (SK930) and wild-type En. faecalis (V583), with or without one of two Bifidobacterium spp. (B. animalis subsp. lactis LKM512 or B. adolescentis JCM1275T). Compared with mice colonized with putrescine-deficient E. coli (SK930) and wild-type En. faecalis (V583) alone, the fecal pH (Fig. 4D) and putrescine concentrations (Fig. 4E) in mice additionally colonized with LKM512 were significantly lower (P < 0.01) and higher (P < 0.05), respectively. However, there was no significant difference in fecal pH (Fig. 4F) and putrescine concentration (Fig. 4G) between gnotobiotic mice colonized with and without B. adolescentis JCM1275T. Although we confirmed the colonization of both bacteria, the number of B. animalis subsp. lactis LKM512 was higher than that of B. adolescentis JCM1275T (fig. S4, A and B). These results suggested that the ability of Bifidobacterium spp. to increase putrescine depended on the ability of acidification of the intestinal luminal environment and that this ability differs among species.
To investigate the potential effect of acidification of the intestinal luminal environment on putrescine production, we suspended human feces in buffers with different pH and incubated for 24 hours (n = 6). We increased the putrescine concentrations at pH 5.4 to 5.9 and observed the increase in all samples at pH 5.4, suggesting that acidification of the intestinal luminal environment might trigger putrescine production by intestinal bacteria (Fig. 4H). In an additional experiment (n = 5), wherein we suspended fecal samples in 0.1 N HCl to kill bacterial cells, putrescine concentration was slightly increased (fig. S5A), suggesting that the putrescine production in the fecal suspensions incubated at pH 5.4 to 5.9 (Fig. 4H) was not derived from the rupture of bacterial cells caused by the acidic conditions.
Putrescine production by collaboration of En. faecalis and other intestinal bacterial species
En. faecalis, which contributes to the second half of the putrescine production process identified in this study (Fig. 2), is a dominant intestinal bacterial species (16), and consequently, it is expected to play a significant role in metabolite flow in the intestinal lumen. However, E. coli is a relatively minor species in the intestine; therefore, to determine whether other intestinal bacteria can substitute for E. coli in the first half of putrescine production, we identified species that have AdiA and AdiC from a list of intestinal bacteria by in silico analyses (table S1). Two species identified by this screen, Citrobacter youngae ATCC 29220 and Fusobacterium varium ATCC 27725, were cocultured with wild-type En. faecalis (V583). When Ci. youngae ATCC 29220 and En. faecalis were cocultured, putrescine production tended to increase as compared to that in separate Ci. youngae culture (Fig. 5A). In addition, when F. varium ATCC 27725 and En. faecalis were cocultured, the putrescine concentration in the culture supernatant was more than 28 times higher than when F. varium was cultured alone. Furthermore, the concentration of putrescine in feces of gnotobiotic mice cocolonized with F. varium ATCC 27725 and wild-type En. faecalis (V583) was significantly higher (P < 0.05) than that of those monocolonized with wild-type En. faecalis (V583) or F. varium ATCC 27725 alone (Fig. 5B). These results indicated that several indigenous intestinal bacterial species that have AdiA and AdiC homologs contributed to the compound putrescine metabolic pathway. Although the agmatine production in Ci. youngae ATCC 29220 monoculture was highest when the pH of the medium was 5.0, that in F. varium ATCC 27725 monoculture was highest when the pH of the medium was 7.0 (fig. S6). This result showed that the AdiC/AdiA pathway of Ci. youngae, as well as that of E. coli, is activated by environmental acidification, whereas the pathway of F. varium is not.
Fig. 5. General putrescine production pathway from arginine via agmatine in the human intestinal microbiome.
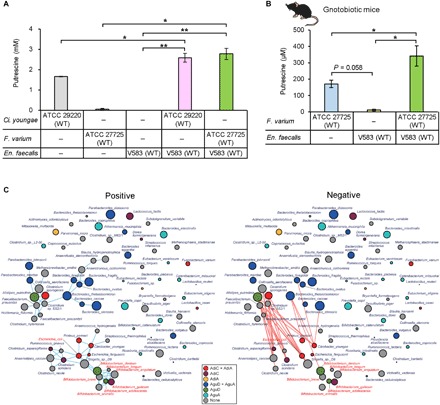
(A) Extracellular putrescine concentration in monoculture of Ci. youngae ATCC 29220 or F. varium ATCC 27725 and in coculture of Ci. youngae ATCC 29220 or F. varium ATCC 27725 with wild-type En. faecalis (V583). (B) Fecal putrescine concentration in gnotobiotic mice inoculated with F. varium ATCC 27725 and wild-type En. faecalis (V583). Mono- and cocultures of these bacteria were conducted under anaerobic conditions at 37°C for 24 hours in LB-RGC medium. (C) Symbiont-symbiont co-occurrence networks of key genes in the putrescine production pathway using previously described human microbiome data from U.S. metropolitan areas (26). Blue and red lines between bacterial species indicate statistically significant aggregation and segregation, respectively. Names of Bifidobacterium spp. and E. coli are shown in red. The size of the circles represents the number of reads identified. Circle colors: red, bacterial species that have AdiC and AdiA; deep pink, bacterial species that have AdiC; gold, bacterial species that have AdiA; blue, bacterial species that have AguD and AguA; chartreuse, bacterial species that have AguD; cyan, bacterial species that have AguA; gray, other bacterial species. Error bars represent SE. *P < 0.05 and **P < 0.01, one-way ANOVA with Tukey’s test and Student’s t tests.
Symbiont-symbiont co-occurrence networks and analysis of metatranscriptome of human feces
To analyze the coexistence of the three groups of bacteria identified in the current study as involved in putrescine production in the human intestinal lumen, we constructed a symbiont-symbiont co-occurrence network using previously reported human microbiome data from U.S. metropolitan areas (26). We identified Bifidobacterium spp. coexistent with bacterial species that have AdiC and/or AdiA (including E. coli) (Fig. 5C, left, and fig. S7, top). In addition, Clostridium sporogenes, which has both AdiC and AdiA, was found to coexist with several species of the genera Bacteroides and Parabacteroides, which have both AguA and AguD. However, these two coexistent clusters occurred separately. In addition, there was segregation between several bacterial species that have both AdiC and AdiA and those that have AguA and/or AguD (Fig. 5C, right, and fig. S7, bottom). The results of analyses using data from Venezuela and Malawi (figs. S8 and S9) (26) were similar to those generated from U.S. fecal microbiomes. To confirm expression of the adiA, adiC, aguA, and aguD genes in colonic lumen of human, we analyzed the number of RNA sequences of these genes using previously reported metatranscriptome data obtained eight human feces (27). We observed expression of the adiA, adiC, and aguA genes derived from various bacterial species in all fecal samples (table S2), demonstrating that three of the four key genes of this metabolic system were activated in the human intestine. However, we did not find the expression of the aguD gene in this analysis. We also did not detect this gene (DNA sequence) in the metagenome data (table S2).
DISCUSSION
Metabolites of intestinal bacteria are considered to be synthesized by reactions within single bacterial cells or by sequential reactions in different bacterial species in the intestinal tract. Studies to experimentally identify enzymes involved in the synthesis of metabolites by intestinal bacteria are in progress and focus on pathways within individual bacterial cells (28–30). Only a few studies have explored the metabolic mechanisms involving the collaboration of more than two intestinal bacteria using genetically engineered bacterial strains. For example, in an excellent study describing the cross-feeding system of polysaccharides by intestinal microbes (31), gene deletions of one of two bacterial species involved in the cross-feeding demonstrated a symbiotic association between bacteria. Here, we have identified a novel hybrid system for putrescine production by two bacteria using deletion and complementation strains of both bacterial species involved.
This study has focused on the metabolism of the polyamine, putrescine, in the ecosystem of the intestinal lumen and has demonstrated genetically that independent metabolic pathways and transport systems of two intestinal bacterial species (En. faecalis and E. coli) can combine to induce the production of putrescine both in vitro and in vivo. In the in vivo experiment, the putrescine level in the colonic lumen was found to be insufficient, suggesting that the produced putrescine may be absorbed by the host. Further study is required to clarify this possibility. Putrescine production via this pathway does not depend on the specific combination of En. faecalis and E. coli, since pairings of En. faecalis and Ci. youngae or F. varium, which have AdiA and AdiC homologs, also produced putrescine. As there are many types of bacteria with homologs of the enzymes AdiA, AdiC, AguA, and/or AguD in the human colon, there are likely countless combinations that can produce putrescine via this pathway. The optimal pH to produce agmatine was different for Ci. youngae and F. varium in monoculture, indicating that the AdiC/AdiA system of some bacteria in the human gut is activated while that of others is not activated by environmental acidification (drop in pH to below 6.5 from neutral).
The induction of putrescine production observed in the present study was triggered by a resistance system of E. coli, which is induced to ensure survival in acidic environments, and the putrescine production depended on acidic pH. E. coli has two arginine decarboxylases, SpeA (32) and AdiA (33), and is able to biosynthesize agmatine. An operon consisting of adiA and adiC encodes an arginine-agmatine antiporter that exchanges agmatine inside the cell for arginine outside (33). In sequential reactions in E. coli, arginine imported from outside the cell by AdiC is decarboxylated by AdiA to generate agmatine with the consumption of protons, increasing the environmental pH. This alkalization system is induced in response to low pH and has been extensively studied as AR3 (22).
In En. faecalis, putrescine is synthesized from agmatine by sequential reactions catalyzed by AguA and AguB (34, 35). As an agmatine biosynthetic pathway has not previously been reported in En. faecalis, and because homologs of agmatine biosynthetic enzymes are not present in En. faecalis, it is thought that this bacterium imports agmatine from outside the cell and converts it to putrescine. The uptake of agmatine is catalyzed by the agmatine-putrescine antiporter AguD, which exchanges putrescine inside the cell for agmatine outside (36). The pathway of agmatine catabolism to yield putrescine via agmatine deiminase consists of AguA, AguB, and AguD and can enhance the growth of En. faecalis by neutralizing the external medium (21). However, this pathway is not influenced by environmental pH but is induced by exogenous agmatine and completely suppressed by environmental glucose (fig. S1), corroborating the report of Suárez et al. (21) that suggested that the agmatine deiminase pathway of En. faecalis has a role not only in acid resistance but also in energy production.
In in silico analysis using human fecal microbiome data, among the three types of bacteria involved in putrescine production in the intestinal lumen, acid-producing Bifidobacterium spp. were coexistent with E. coli as representative bacteria that have systems that release agmatine, suggesting that agmatine production is most likely facilitated by AdiC/AdiA in response to acid stress caused by Bifidobacterium spp. Cl. sporogenes, which has AdiC and AdiA homologs, might play an important role in this putrescine biosynthetic pathway because only Cl. sporogenes coexists with several Bacteroides species, which have AguD and AguA homologs. Both coexistence and segregation were observed among bacterial species that have AdiC/AdiA and those that harbor AguD/AguA, which import agmatine and release putrescine, suggesting that the relationship between these two types of bacteria is not specific and they do not depend on each other. In other words, this relationship is not a specific cosurvival strategy. In this analysis, no existing cluster of the three types of bacteria–those with AdiC/AdiA, those with AguD/AguA, and Bifidobacterium spp.–was found. This may be because only sequenced bacterial genomes were included. In the future, if the number of bacteria with sequenced genomes derived from microbiomes of other ethnic groups increases, then clusters harboring these three bacterial types might be found. Furthermore, the possible involvement of other acid-producing bacteria in environmental acidification should also be considered. Analysis of metatranscriptomic data of human feces obtained from published articles revealed that three of the four key genes of this metabolic system (AdiA, AdiC, and AguA) are activated in the human intestine. AguD gene expression was not found in these data. Because this gene was also not detected in the metagenome data, the absence of AguD expression might be caused by the absence of AguD-harboring bacteria from the fecal samples used in this study, rather than by down-regulation of gene expression. The AguD gene can be detected in DNA sequences of some human feces samples in the database of the National Institutes of Health Human Metagenome Project (www.hmpdacc.org/hmp/), by searching the Joint Genome Institute Integrated Microbial Genomes and Mircobiomes system (JGI-IMG/M: https://img.jgi.doe.gov/). Therefore, there is a possibility that AguD gene expression may be detected in feces with high putrescine concentrations. Further studies involving fecal metatranscriptome analysis after putrescine concentration determination are needed to confirm this notion.
The well-known benefits of Bifidobacterium spp. depend on acidification of the luminal environment by acetate and lactate production by these bacteria. For example, constipation can be alleviated by the activation of peristaltic movement in the digestive tract stimulated by these short-chain fatty acids (37). In addition, beneficial effects occur via the improvement of gut microbiota and the beneficial metabolites produced by Bifidobacterium spp. For example, Bifidobacterium spp. were shown to protect their hosts from enteropathogenic infections through acetate production in the intestinal tract (38). However, there are no previous reports showing that short-chain fatty acids derived from Bifidobacterium spp. induce the production of other metabolites by indigenous bacteria. Here, we describe a novel mechanism whereby acidification of the intestinal environment by Bifidobacterium spp. facilitates the synthesis and export of a polyamine by coculture of E. coli and En. faecalis in vitro and in vivo. The findings of the present study indicate the need for a major paradigm shift away from the concept that Bifidobacterium spp. only generate direct health benefits toward the idea that they facilitate indirect benefits through modification of metabolite generation by the gut microbiota in the large intestine. Putrescine production was inhibited by mixed cultures containing several Bifidobacterium spp. (B. longum and B. pseudocatenulatum) that suppressed growth of E. coli and En. faecalis. This finding suggests that only those Bifidobacterium spp. that do not suppress commensal bacteria, especially bacteria that have AdiC/AdiA or AguD/AguA, might be involved in inducing putrescine production via this hybrid system.
In conclusion, we identified a novel pathway for putrescine production from arginine through agmatine involving collaboration of two different bacterial species, triggered by environmental acidification (drop in pH to below 6.5 from neutral) in hosts harboring acid-producing bacteria in the gut. This pathway consists of independent strategies in the individual contributing bacteria: the acid tolerance system of E. coli (arginine-dependent acid resistance system) and the energy production system of En. faecalis (agmatine deiminase system), in the context of acid production by acid-producing bacteria, represented here by Bifidobacterium spp. (fig. S10).
We believe that similar interspecies connections are likely to exist in the production of other metabolites by intestinal microbiota. It will be important to consider this possibility in future analyses of metabolite production by gut microbial communities. It should be noted that novel pathways for the production of specific metabolites via microbial networks cannot be readily identified from metagenome data alone because, even if a microorganism has the synthetic pathway of a specific metabolite, it does not necessarily release this metabolite.
MATERIALS AND METHODS
Bacterial strains and plasmids
Most gut microbial species belong to four major phyla: Firmicutes, Actinobacteria, Proteobacteria, and Bacteroidetes. We selected 14 intestinal bacterial species that are the predominant species in human microbiota (16) from these four phyla (table S3). The strains, plasmids, and primers used in this study are listed in table S4.
In E. coli, P1 transduction (39) was used to transfer chromosomes with gene deletions ΔadiC (JW4076) and ΔadiA (JW5731) from the Keio collection (40) into a wild-type MG1655 background to generate SK900 (ΔadiA:: FRT-kan+-FRT) and SK902 (ΔadiC:: FRT-kan+-FRT). The plasmid pCP20 (41) was then introduced to eliminate the kanamycin resistance cassettes, generating SK901 (ΔadiA:: FRT) and SK903 (ΔadiC:: FRT).
pBelobac11-adiC+ was constructed as follows: A 1725–base pair (bp) DNA fragment including adiC and a 300-bp region upstream of this gene was amplified by polymerase chain reaction (PCR) using KOD-Plus (Toyobo Co. Ltd.) polymerase, “adiC_300bp-up_for” and “adiC_term_rev” as primers, and genomic DNA of wild-type E. coli (MG1655) as a template. The amplified fragment was cloned into pBelobac11 digested with Hpa I. The cloned region was sequenced, demonstrating that it contained no mutations.
pBelobac11-adiA+ was constructed as follows: A 2598-bp DNA fragment including adiA and a 300-bp upstream region was amplified by PCR using KOD-Plus polymerase, “adiA_300bp-up_for” and “adiA_term_rev” as primers, and genomic DNA of wild-type E. coli (MG1655) as template. The amplified fragment was cloned into pBelobac11 digested with Hpa I. The cloned region was sequenced, and there were no mutations.
A derivative of the temperature-sensitive plasmid pLT06 (42), pLT06-ΔaguD, was used for allelic exchange of aguD in En. faecalis, following methods described previously (43, 44). Briefly, a sequence extending from −1 to −1042, with reference to the aguD start codon, and a second fragment extending from +1 to +1096 relative to the aguD stop codon were amplified by PCR. The two amplicons were concatenated using PCR, and the resulting 2140-bp DNA fragment was cloned into the Eco RI site of pLT06 to generate pLT06-ΔaguD. pLT06-ΔaguD was introduced into V583, resulting in SK932. A strain with a single-crossover event in the region flanking aguD on the chromosome of SK932 was screened on THB plates (Becton, Dickinson and Company) supplemented with chloramphenicol (10 μg/ml) and 5-bromo-4-chloro-3-indolyl-β-d-galactopyranoside (X-gal; 120 μg/ml) after incubation at 42°C to eliminate the temperature-sensitive plasmid, pLT06-ΔaguD, generating SK934. The single crossover was confirmed in a temperature- and chloramphenicol-resistant blue colony by PCR. Then, a strain with a double-crossover event was generated by two passages of culture in medium without chloramphenicol and screened on MM9YEG plates (45) containing X-gal (120 μg/ml) and p-chloro-phenylalanine (p-Cl-Phe; 10 μg/ml) to generate SK937. The double crossover generating ΔaguD was confirmed in a p-Cl-Phe–resistant white colony by PCR. The manipulated chromosomal region in SK937 was sequenced to confirm that no unintended mutations had been introduced.
pLZ12-aguB+D+ was constructed as follows: A 2985-bp DNA fragment, including the aguBD operon and a 500-bp region upstream of aguB on the chromosome of wild-type En. faecalis (V583), was amplified by PCR using PrimeSTAR polymerase (Takara Bio Inc.), the primers Clone_agcB+0. 5K_FWD and Clone_agcD+COMPL, and genomic DNA of wild-type En. faecalis (V583) as a template. The amplified fragment was cloned into pLZ12 (46) digested with Bam HI. The cloned region was sequenced, and no mutations were identified.
Screening of polyamine-producing bacteria
We searched for combinations of intestinal bacterial species that produced polyamines from l-arginine among 14 different intestinal bacteria; in total, 91 combinations were tested. Strains were each precultured in suitable medium at 37°C for 24 hours under anaerobic conditions. Bacteroides vulgatus, Ba. thetaiotaomicron, Ba. caccae, Ba. uniformis, Parabacteroides merdae, Pa. johnsonii, En. faecalis, and E. coli were precultured in GAM Broth (Nissui Pharmaceutical Co. Ltd.). Alistipes putredinis was precultured in Eggerth-Gagnon (EG) broth (JCM medium 14). Blautia hansenii was precultured in peptone yeast glucose broth (JCM medium 676). Cl. leptum was precultured in PY+X broth (DSMZ medium 104c). Streptococcus thermophiles was precultured in M17 broth (Merck KGaA). B. adolescentis and B. longum were precultured in blood liver (BL) broth (Nissui Pharmaceutical Co. Ltd.). Each bacterial preculture was inoculated at a final optical density at 600 nm (OD600) of 1.0 × 10−2 in gifu anaerobe medium (GAM) broth containing 2 mM l-arginine. Strains were grown under anaerobic conditions at 37°C for 24 hours. Culture supernatants were collected by centrifugation at 9100g for 10 min, and the putrescine concentrations were measured.
Mixed cultures of E. coli and En. faecalis
Strains were grown under anaerobic conditions at 37°C for 24 hours in LB medium (Becton, Dickinson and Company) containing 2 mM l-arginine, 1.5 g/liter d-glucose, and 0.5 g/liter l-cysteine-hydrochloride (LB-RGC medium) in screw-top test tubes with butyl rubber inner plugs (Sanshin Industrial Co. Ltd.). The gas-phase portions in screw-top test tubes were replaced with N2/CO2 (80:20, v/v) before autoclaving (115°C, 15 min). Chloramphenicol was added to the medium at a final concentration of 10 μg/ml, where required. E. coli precultures were harvested after 24 hours and inoculated at a final OD600 of 1.0 × 10−2. En. faecalis was harvested after 24 hours and inoculated at a final OD600 of 1.0 × 10−5. After 24 hours of mono- or coculture, culture supernatants were collected by centrifugation (9100g, 10 min), and the putrescine concentration was determined. To measure viable bacterial counts of En. faecalis, aliquots (10 μl) were removed and serially diluted, 20 μl of each dilution was plated on KF Streptococcus Agar (Oxoid), and the CFU were counted.
Incubation using culture supernatants from E. coli or En. faecalis
Strains were grown under anaerobic conditions at 37°C for 24 hours in LB-RGC medium in screw-top test tubes with butyl rubber inner plugs. The gas-phase portions in the screw-top test tubes were replaced with N2/CO2 (80:20, v/v) before autoclaving (115°C, 15 min). E. coli and En. faecalis precultures were harvested after 24 hours and inoculated at a final OD600 of 1.0 × 10−2. E. coli or En. faecalis was cultured for 24 hours, and culture supernatants were collected by centrifugation (1500g, 10 min) and sterilized through commercial filter membrane units (Millex-GV, 0.22 μm; polyvinylidene difluoride, 33 mm; Merck Millipore). The other strain was then cultured for 24 hours in the sterilized culture supernatant, and l-arginine, agmatine, and putrescine concentrations were measured in the final culture supernatant.
Monoculture of E. coli, En. faecalis, Ci. youngae, and F. varium
Precultures were harvested and inoculated at a final OD600 of 1.0 × 10−2 in the media described below. Wild-type E. coli (MG1655) was cultured under anaerobic conditions at 37°C for 24 hours in LB-RGC medium (pH 4.0 to 9.0). Wild-type En. faecalis (V583) was cultured in LB medium containing l-cysteine-hydrochloride (0.5 g/liter) and 5 mM agmatine (pH 4.0 to 9.0) or in LB medium containing glucose (0 to 3 g/liter), l-cysteine-hydrochloride (0.5 g/liter), and 5 mM agmatine (pH 6.0). Culture supernatants were collected by centrifugation (9100g, 10 min), and the putrescine and agmatine concentrations were measured. Ci. youngae ATCC 29220 and F. varium ATCC 27725 were cultured under anaerobic conditions at 37°C for 24 hours in LB-RGC medium (pH 4.0 to 9.0). Culture supernatants were collected by centrifugation (9100g, 10 min), and the agmatine and putrescine concentrations and pH were measured. For viable bacterial counts of Bifidobacterium spp., aliquots (10 μl) were removed and serially diluted, 20 μl of each dilution was plated on TOS propionate agar (Yakult Pharmaceutical Ind. Co. Ltd.), and the CFU were counted.
Mixed cultures of E. coli, En. faecalis, and Bifidobacterium spp.
We prepared mixed cultures of putrescine-deficient E. coli (SK930) and wild-type En. faecalis (V583) and of putrescine-deficient E. coli (SK930), wild-type En. faecalis (V583), and Bifidobacterium spp. Strains were grown under anaerobic conditions at 37°C for 24 hours in LB medium containing 2 mM l-arginine, d-glucose (1.5 g/liter), galacto-oligosaccharide (5 g/liter), 2 mM MgSO4, 60 mM NH4Cl, and l-cysteine-hydrochloride (0.5 g/liter; pH 6.5) in screw-top test tubes with butyl rubber inner plugs. The gas-phase portions in the screw-top test tubes were replaced with N2/CO2 (80:20, v/v) before autoclaving (115°C, 15 min). E. coli precultures were harvested and inoculated at a final OD600 of 1.0 × 10–3 in the above-mentioned medium, En. faecalis precultures were inoculated at a final OD600 of 1.0 × 10–5, and each Bifidobacterium sp. was inoculated at a final OD600 of 1.0 × 10–2. Culture supernatants were collected by centrifugation (9100g, 10 min), and the putrescine concentration and pH were measured. For viable bacterial counts of E. coli, En. faecalis, and Bifidobacterium spp., aliquots (10 μl) were removed and serially diluted, 20 μl of each dilution was plated on deoxycholate agar (Merck KGaA), KF Streptococcus Agar (Oxoid), and TOS propionate agar (Yakult Pharmaceutical Ind. Co. Ltd.), respectively, and the CFU were counted.
Colonization of gnotobiotic mice
Germ-free mice (C57BL/6) were purchased from Sankyo Labo Service Co. Inc. and were bred at Kyodo Milk Industry Co. Ltd. Mice were housed in flexible film plastic isolators with sterilized bedding and provided with sterilized water [containing chloramphenicol (10 μg/ml), where required] and sterilized commercial CMF pellets (Oriental Yeast Co. Ltd.) ad libitum. Six-week-old germ-free mice were used in all experiments and were divided into three groups (n = 3 per group): a control group colonized with putrescine-deficient E. coli (SK930) and wild-type En. faecalis (SK947), a knockout group colonized with putrescine-deficient E. coli (SK930) and aguD-deleted En. faecalis (SK948), and a complement group colonized with putrescine-deficient E. coli (SK930) and aguD-complemented En. faecalis (SK949). The experimental schedule is presented in fig. S11A. First, germ-free mouse feces were collected. Then, the three groups of mice were gavaged with phosphate-buffered saline (PBS) containing different En. faecalis strains (1.0 × 109 CFU). After 20 hours, mouse feces were collected, and all groups of mice were gavaged with PBS containing putrescine-deficient E. coli (SK930) (1.0 × 1010 CFU). After 20 hours, feces were collected. The mice were sacrificed, the colonic and cecal contents were obtained, and the putrescine concentration of these samples was measured.
In experiments to test the effects of three types of bacteria, 6-week-old germ-free C57BL/6 mice were divided into two groups (n = 3 per group): one colonized with two types of bacteria [putrescine-deficient E. coli (SK930) and wild-type En. faecalis (V583)] and one with three types of bacteria [putrescine-deficient E. coli (SK930), wild-type En. faecalis (V583), and B. animalis subsp. lactis (LKM512)]. The experimental schedule is presented in fig. S11B. First, germ-free mice feces were collected, and then, the first group of mice was gavaged with PBS containing putrescine-deficient E. coli (SK930) (1.0 × 1010 CFU) and wild-type En. faecalis (V583) (3.0 × 109 CFU), while the latter group was gavaged with PBS containing putrescine-deficient E. coli (SK930) (1.0 × 1010 CFU), wild-type En. faecalis (V583) (3.0 × 109 CFU), and B. animalis subsp. lactis LKM512 (1.0 × 108 CFU). After 20 hours, feces were collected, and the polyamine concentrations and pH were measured.
To test the effects of colonization with F. varium and En. faecalis, experimental mice were divided into two groups (n = 3 per group). The experimental schedule is presented in fig. S11C. One group was gavaged with PBS containing F. varium ATCC 27725 (5.0 × 108 CFU) on the first day and wild-type En. faecalis (V583) (3.0 × 109 CFU) on the second day. A second group of mice was gavaged with PBS containing wild-type En. faecalis (V583) (3.0 × 109 CFU) on the first day and F. varium ATCC 27725 (5.0 × 108 CFU) on the second day. After the administration of bacteria (20 hours), mouse feces were collected, and the putrescine concentrations were measured. All animal experiments were approved by the Kyodo Milk Animal Use Committee (permit numbers 2015-01 and 2016-10) and were in accordance with the Guide for the Care and Use of Laboratory Animals, published by the National Academies Press.
Culture of human feces at various pH values
Feces were obtained from six volunteers and suspended in nine volumes of 100 mM MES buffer (Dojindo Laboratories) (pH 4.2, 5.0, 5.5, 6.0, 6.5, and 7.0). The suspensions were incubated for 24 hours under anaerobic conditions. Polyamine concentrations were measured in the culture supernatants.
In an additional experiment, feces were obtained from five volunteers and suspended in nine volumes of 100 mM HCl or 100 mM MES buffer (Dojindo Laboratories) (pH 4.2, 5.0, 6.0, and 7.0). The suspensions were incubated for 24 hours under anaerobic conditions. Polyamine concentrations and pH were measured in the culture supernatants. For viable bacterial counts, aliquots (10 μl) were removed and serially diluted; 20 μl of each dilution was plated on EG agar (JCM medium 14), BL agar (Nissui Pharmaceutical Co. Ltd.), and GAM agar (Nissui Pharmaceutical Co. Ltd.); and the CFUs were counted.
Determination of polyamine, l-arginine, and agmatine concentrations
The polyamine, l-arginine, and agmatine concentrations in culture supernatants were measured, as described by Reguera et al. (47), with some modifications, using an Acquity ultra-performance liquid chromatography system with an FLR Detector (Waters), as previously described (14).
In silico pathway analysis using BLAST
Protein BLAST (48) analysis was performed using protein sequence data from 126 bacterial strains present in the human gut (26). The accession IDs and sources of the protein sequences used for the BLAST search, as well as the identity scores, are listed in table S1.
Mixed cultures of F. varium and Ci. youngae
Similar to the mixed culture of E. coli and En. faecalis, F. varium and Ci. youngae were grown in LB-RGC medium under anaerobic conditions at 37°C for 24 hours. F. varium and Ci. youngae were harvested after 24 hours of preculture and inoculated at a final OD600 of 1.0 × 10−2. After 24 hours of culture, culture supernatants were collected by centrifugation (9100g, 10 min), and the putrescine concentration was determined.
Symbiont-symbiont co-occurrence network analysis
Corresponding bacterial operational taxonomic units (OTUs) for each of the 126 bacterial species genome sequences were identified among the 16S ribosomal RNA data of Yatsunenko et al. (26) using the program Minimo (49). Corresponding 16S OTUs were found for 84 of the 126 bacteria. Patterns of co-occurrence among the 84 bacteria were examined using the sparse correlations for compositional data (SparCC) method (50). A positive correlation coefficient in the SparCC analysis represents a pair of bacteria potentially sharing environmental preferences and/or mutually interacting with each other, while a negative correlation coefficient represents diverged environmental preferences and/or competitive or antagonistic interactions. Only links with absolute correlation coefficients >0.3 were included in the networks.
Analysis of the expression of AdiA, AdiC, AguA, and AguD genes using metatranscriptomic data
The expression of AdiA, AdiC, AguA, and AguD genes in the metatranscriptome of human feces was analyzed using the metatranscriptomic data of Franzosa et al. (27).
Statistical analyses
Extracellular and fecal putrescine concentrations, pH, and viable bacterial counts were compared between the groups with the Student’s t test, one-way ANOVA (followed by Tukey’s test), Steel-Dwass test, or Wilcoxon signed-rank test. Comparisons at weeks 0 and 12 were tested using paired t test or Wilcoxon signed-rank test. Student’s t test and one-way ANOVA (followed by Tukey’s test) were performed using SPSS version 22 (IBM), and other analyses were performed using R statistical software version 3.4.2 or SPSS version 22.
Supplementary Material
Acknowledgments
We are grateful to B. A. Duerkop, and L. Hooper from the University of Texas Southwestern Medical Center, Dallas for technical advice and for the gift of pLZ12. We acknowledge L. E. Hancock, University of Kansas, for providing pLT06. We are grateful to Y. Doi, Okayama University of Science, for technical advice regarding electroporation of En. faecalis. We thank A. Yamashita and A. Nakamura of Kyodo Milk Industry Co. Ltd. for supporting this study. Funding: This work was funded by Kyodo Milk Industry Co. Ltd. The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript. This study was partially supported by Grants-in-Aid from the Institute for Fermentation, Osaka (to S.K.). Author contributions: Y.K. performed the experiments in Figs. 3 to 5 (A and B), wrote the Materials and Methods, and drew the figures. K.M. hypothesized and performed the experiments in Fig. 1. H.T. performed the symbiont-symbiont co-occurrence network analysis and edited the manuscript. R.K. performed and analyzed the experiment in Fig. 1 and table S2. Y.B. supervised this study. S.K. designed and prepared the genetically modified bacterial experiments, analyzed the data, and wrote the manuscript. M.M. conceived the study, analyzed the data, and wrote the manuscript. Competing interests: Kyodo Milk Industry Co. Ltd., which funded this research, has commercial products (yogurt and bacterial powder) containing B. animalis subsp. lactis LKM512 that is related to the content of this article to declare. The authors declare that they have no competing interests. Data and materials availability: All data needed to evaluate the conclusions in the paper are present in the paper and/or the Supplementary Materials. Additional data related to this paper may be requested from the authors.
SUPPLEMENTARY MATERIALS
Supplementary material for this article is available at http://advances.sciencemag.org/cgi/content/full/4/6/eaat0062/DC1
fig. S1. Induction of putrescine production by coculture of E. coli and En. faecalis.
fig. S2. Effects of extracellular pH and glucose concentration on bacterial metabolism in monocultures of En. faecalis and E. coli.
fig. S3. Viable bacterial counts and change of extracellular pH in cocultures of putrescine-deficient E. coli (SK930), wild-type En. faecalis (V583), and each Bifidobacterium sp.
fig. S4. Viable bacterial counts in feces of gnotobiotic mice.
fig. S5. Extracellular putrescine concentration and total viable bacterial counts in human feces incubated at different pH values (n = 5).
fig. S6. Effect of pH on extracellular agmatine concentration in monocultures of E. coli, Ci. youngae, and F. varium.
fig. S7. Symbiont-symbiont co-occurrence networks of key genes in the putrescine production pathway using previously described human microbiome data from U.S. metropolitan areas.
fig. S8. Symbiont-symbiont co-occurrence network patterns of key genes in the putrescine production pathway using previously described human microbiome data from Venezuela.
fig. S9. Symbiont-symbiont co-occurrence network patterns of key genes in the putrescine production pathway using previously described human microbiome data from Malawi.
fig. S10. Mechanistic model of a novel pathway for putrescine production from arginine through agmatine via the collaboration of three different bacterial species.
fig. S11. Outline of gnotobiotic mouse experiments.
table S1. List of species expressing homologs of the enzymes AdiA and AdiC, as determined by in silico analyses of 126 bacterial strains present in the human gut.
table S2. Detection of RNA sequences of AdiA, AdiC, AguA, and AguD by metatranscriptomic analysis of human feces.
table S3. List of bacteria used for screening of polyamine producing bacteria.
table S4. Strains, plasmids, and oligonucleotides used in this study.
REFERENCES AND NOTES
- 1.Sharon G., Garg N., Debelius J., Knight R., Dorrestein P. C., Mazmanian S. K., Specialized metabolites from the microbiome in health and disease. Cell Metab. 20, 719–730 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Matsumoto M., Benno Y., The relationship between microbiota and polyamine concentration in the human intestine: A pilot study. Microbiol. Immunol. 51, 25–35 (2007). [DOI] [PubMed] [Google Scholar]
- 3.Matsumoto M., Kakizoe K., Benno Y., Comparison of fecal microbiota and polyamine concentration in adult patients with intractable atopic dermatitis and healthy adults. Microbiol. Immunol. 51, 37–46 (2007). [DOI] [PubMed] [Google Scholar]
- 4.Tabor C. W., Tabor H., Polyamines in microorganisms. Microbiol. Rev. 49, 81–99 (1985). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Pegg A. E., Recent advances in the biochemistry of polyamines in eukaryotes. Biochem. J. 234, 249–262 (1986). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Eisenberg T., Knauer H., Schauer A., Büttner S., Ruckenstuhl C., Carmona-Gutierrez D., Ring J., Schroeder S., Magnes C., Antonacci L., Fussi H., Deszcz L., Hartl R., Schraml E., Criollo A., Megalou E., Weiskopf D., Laun P., Heeren G., Breitenbach M., Grubeck-Loebenstein B., Herker E., Fahrenkrog B., Fröhlich K.-U., Sinner F., Tavernarakis N., Minois N., Kroemer G., Madeo F., Induction of autophagy by spermidine promotes longevity. Nat. Cell Biol. 11, 1305–1314 (2009). [DOI] [PubMed] [Google Scholar]
- 7.Soda K., Dobashi Y., Kano Y., Tsujinaka S., Konishi F., Polyamine-rich food decreases age-associated pathology and mortality in aged mice. Exp. Gerontol. 44, 727–732 (2009). [DOI] [PubMed] [Google Scholar]
- 8.Gupta V. K., Scheunemann L., Eisenberg T., Mertel S., Bhukel A., Koemans T. S., Kramer J. M., Liu K. S. Y., Schroeder S., Stunnenberg H. G., Sinner F., Magnes C., Pieber T. R., Dipt S., Fiala A., Schenck A., Schwaerzel M., Madeo F., Sigrist S. J., Restoring polyamines protects from age-induced memory impairment in an autophagy-dependent manner. Nat. Neurosci. 16, 1453–1460 (2013). [DOI] [PubMed] [Google Scholar]
- 9.Eisenberg T., Abdellatif M., Schroeder S., Primessnig U., Stekovic S., Pendl T., Harger A., Schipke J., Zimmermann A., Schmidt A., Tong M., Ruckenstuhl C., Dammbrueck C., Gross A. S., Herbst V., Magnes C., Trausinger G., Narath S., Meinitzer A., Hu Z., Kirsch A., Eller K., Carmona-Gutierrez D., Büttner S., Pietrocola F., Knittelfelder O., Schrepfer E., Rockenfeller P., Simonini C., Rahn A., Horsch M., Moreth K., Beckers J., Fuchs H., Gailus-Durner V., Neff F., Janik D., Rathkolb B., Rozman J., de Angelis M. H., Moustafa T., Haemmerle G., Mayr M., Willeit P., von Frieling-Salewsky M., Pieske B., Scorrano L., Pieber T., Pechlaner R., Willeit J., Sigrist S. J., Linke W. A., Mühlfeld C., Sadoshima J., Dengjel J., Kiechl S., Kroemer G., Sedej S., Madeo F., Cardioprotection and lifespan extension by the natural polyamine spermidine. Nat. Med. 22, 1428–1438 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gerner E. W., Meyskens F. L. Jr, Polyamines and cancer: Old molecules, new understanding. Nat. Rev. Cancer 4, 781–792 (2004). [DOI] [PubMed] [Google Scholar]
- 11.Laukaitis C. M., Gerner E. W., DFMO: Targeted risk reduction therapy for colorectal neoplasia. Best Pract. Res. Clin. Gastroenterol. 25, 495–506 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Uda K., Tsujikawa T., Fujiyama Y., Bamba T., Rapid absorption of luminal polyamines in a rat small intestine ex vivo model. J. Gastroenterol. Hepatol. 18, 554–559 (2003). [DOI] [PubMed] [Google Scholar]
- 13.Matsumoto M., Kibe R., Ooga T., Aiba Y., Kurihara S., Sawaki E., Koga Y., Benno Y., Impact of intestinal microbiota on intestinal luminal metabolome. Sci. Rep. 2, 233 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kibe R., Kurihara S., Sakai Y., Suzuki H., Ooga T., Sawaki E., Muramatsu K., Nakamura A., Yamashita A., Kitada Y., Kakeyama M., Benno Y., Matsumoto M., Upregulation of colonic luminal polyamines produced by intestinal microbiota delays senescence in mice. Sci. Rep. 4, 4548 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Matsumoto M., Kurihara S., Kibe R., Ashida H., Benno Y., Longevity in mice is promoted by probiotic-induced suppression of colonic senescence dependent on upregulation of gut bacterial polyamine production. PLOS ONE 6, e23652 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Qin J., Li R., Raes J., Arumugam M., Burgdorf K. S., Manichanh C., Nielsen T., Pons N., Levenez F., Yamada T., Mende D. R., Li J., Xu J., Li S., Li D., Cao J., Wang B., Liang H., Zheng H., Xie Y., Tap J., Lepage P., Bertalan M., Batto J.-M., Hansen T., Le Paslier D., Linneberg A., Nielsen H. B., Pelletier E., Renault P., Sicheritz-Ponten T., Turner K., Zhu H., Yu C., Li S., Jian M., Zhou Y., Li Y., Zhang X., Li S., Qin N., Yang H., Wang J., Brunak S., Doré J., Guarner F., Kristiansen K., Pedersen O., Parkhill J., Weissenbach J. ; MetaHIT Consortium, Bork P., Ehrlich S. D., Wang J., A human gut microbial gene catalogue established by metagenomic sequencing. Nature 464, 59–65 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Burrell M., Hanfrey C. C., Murray E. J., Stanley-Wall N. R., Michael A. J., Evolution and multiplicity of arginine decarboxylases in polyamine biosynthesis and essential role in Bacillus subtilis biofilm formation. J. Biol. Chem. 285, 39224–39238 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Sugiyama Y., Nara M., Sakanaka M., Gotoh A., Kitakata A., Okuda S., Kurihara S., Comprehensive analysis of polyamine transport and biosynthesis in the dominant human gut bacteria: Potential presence of novel polyamine metabolism and transport genes. Int. J. Biochem. Cell Biol. 93, 52–61 (2017). [DOI] [PubMed] [Google Scholar]
- 19.Rijkers G. T., Bengmark S., Enck P., Haller D., Herz U., Kalliomaki M., Kudo S., Lenoir-Wijnkoop I., Mercenier A., Myllyluoma E., Rabot S., Rafter J., Szajewska H., Watzl B., Wells J., Wolvers D., Antoine J.-M., Guidance for substantiating the evidence for beneficial effects of probiotics: Current status and recommendations for future research. J. Nutr. 140, 671S–676S (2010). [DOI] [PubMed] [Google Scholar]
- 20.Fuller R., Probiotics in man and animals. J. Appl. Bacteriol. 66, 365–378 (1989). [PubMed] [Google Scholar]
- 21.Suárez C., Espariz M., Blancato V. S., Magni C., Expression of the agmatine deiminase pathway in Enterococcus faecalis is activated by the AguR regulator and repressed by CcpA and PTSMan systems. PLOS ONE 8, e76170 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Foster J. W., Escherichia coli acid resistance: Tales of an amateur acidophile. Nat. Rev. Microbiol. 2, 898–907 (2004). [DOI] [PubMed] [Google Scholar]
- 23.Richard H., Foster J. W., Escherichia coli glutamate- and arginine-dependent acid resistance systems increase internal pH and reverse transmembrane potential. J. Bacteriol. 186, 6032–6041 (2004). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.de Vries W., Stouthamer A. H., Fermentation of glucose, lactose, galactose, mannitol, and xylose by bifidobacteria. J. Bacteriol. 96, 472–478 (1968). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Bullen C. L., Tearle P. V., Willis A. T., Bifidobacteria in the intestinal tract of infants: An in-vivo study. J. Med. Microbiol. 9, 325–333 (1976). [DOI] [PubMed] [Google Scholar]
- 26.Yatsunenko T., Rey F. E., Manary M. J., Trehan I., Dominguez-Bello M. G., Contreras M., Magris M., Hidalgo G., Baldassano R. N., Anokhin A. P., Heath A. C., Warner B., Reeder J., Kuczynski J., Caporaso J. G., Lozupone C. A., Lauber C., Clemente J. C., Knights D., Knight R., Gordon J. I., Human gut microbiome viewed across age and geography. Nature 486, 222–227 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Franzosa E. A., Morgan X. C., Segata N., Waldron L., Reyes J., Earl A. M., Giannoukos G., Boylan M. R., Ciulla D., Gevers D., Izard J., Garrett W. S., Chan A. T., Huttenhower C., Relating the metatranscriptome and metagenome of the human gut. Proc. Natl. Acad. Sci. U.S.A. 111, E2329–E2338 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wu M., McNulty N. P., Rodionov D. A., Khoroshkin M. S., Griffin N. W., Cheng J., Latreille P., Kerstetter R. A., Terrapon N., Henrissat B., Osterman A. L., Gordon J. I., Genetic determinants of in vivo fitness and diet responsiveness in multiple human gut Bacteroides. Science 350, aac5992 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Rogowski A., Briggs J. A., Mortimer J. C., Tryfona T., Terrapon N., Lowe E. C., Baslé A., Morland C., Day A. M., Zheng H., Rogers T. E., Thompson P., Hawkins A. R., Yadav M. P., Henrissat B., Martens E. C., Dupree P., Gilbert H. J., Bolam D. N., Glycan complexity dictates microbial resource allocation in the large intestine. Nat. Commun. 6, 7481 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Degnan P. H., Barry N. A., Mok K. C., Taga M. E., Goodman A. L., Human gut microbes use multiple transporters to distinguish vitamin B12 analogs and compete in the gut. Cell Host Microbe 15, 47–57 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Rakoff-Nahoum S., Foster K. R., Comstock L. E., The evolution of cooperation within the gut microbiota. Nature 533, 255–259 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Boyle S. M., Markham G. D., Hafner E. W., Wright J. M., Tabor H., Tabor C. W., Expression of the cloned genes encoding the putrescine biosynthetic enzymes and methionine adenosyltransferase of Escherichia coli (speA, speB, speC and metK). Gene 30, 129–136 (1984). [DOI] [PubMed] [Google Scholar]
- 33.Gong S., Richard H., Foster J. W., YjdE (AdiC) is the arginine:agmatine antiporter essential for arginine-dependent acid resistance in Escherichia coli. J. Bacteriol. 185, 4402–4409 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Landete J. M., Arena M. E., Pardo I., Manca de Nadra M. C., Ferrer S., The role of two families of bacterial enzymes in putrescine synthesis from agmatine via agmatine deiminase. Int. Microbiol. 13, 169–177 (2010). [DOI] [PubMed] [Google Scholar]
- 35.Llácer J. L., Polo L. M., Tavarez S., Alarcon B., Hilario R., Rubio V., The gene cluster for agmatine catabolism of Enterococcus faecalis: Study of recombinant putrescine transcarbamylase and agmatine deiminase and a snapshot of agmatine deiminase catalyzing its reaction. J. Bacteriol. 189, 1254–1265 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Driessen A. J., Smid E. J., Konings W. N., Transport of diamines by Enterococcus faecalis is mediated by an agmatine-putrescine antiporter. J. Bacteriol. 170, 4522–4527 (1988). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Shi Y., Chen Q., Huang Y., Ni L., Liu J., Jiang J., Li N., Function and clinical implications of short-chain fatty acids in patients with mixed refractory constipation. Colorectal Dis. 18, 803–810 (2016). [DOI] [PubMed] [Google Scholar]
- 38.Fukuda S., Toh H., Hase K., Oshima K., Nakanishi Y., Yoshimura K., Tobe T., Clarke J. M., Topping D. L., Suzuki T., Taylor T. D., Itoh K., Kikuchi J., Morita H., Hattori M., Ohno H., Bifidobacteria can protect from enteropathogenic infection through production of acetate. Nature 469, 543–547 (2011). [DOI] [PubMed] [Google Scholar]
- 39.Miller J. H., A short course in bacterial genetics: A laboratory manual and handbook for Escherichia coli and related bacteria. Trends Biochem. Sci. 18, 193 (1993). [Google Scholar]
- 40.Baba T., Ara T., Hasegawa M., Takai Y., Okumura Y., Baba M., Datsenko K. A., Tomita M., Wanner B. L., Mori H., Construction of Escherichia coli K-12 in-frame, single-gene knockout mutants: The Keio collection. Mol. Syst. Biol. 2, 2006.0008 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Datsenko K. A., Wanner B. L., One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proc. Natl. Acad. Sci. U.S.A. 97, 6640–6645 (2000). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Thurlow L. R., Thomas V. C., Hancock L. E., Capsular polysaccharide production in Enterococcus faecalis and contribution of CpsF to capsule serospecificity. J. Bacteriol. 191, 6203–6210 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Shepard B. D., Gilmore M. S., Electroporation and efficient transformation of Enterococcus faecalis grown in high concentrations of glycine. Methods Mol. Biol. 47, 217–226 (1995). [DOI] [PubMed] [Google Scholar]
- 44.Duerkop B. A., Clements C. V., Rollins D., Rodrigues J. L. M., Hooper L. V., A composite bacteriophage alters colonization by an intestinal commensal bacterium. Proc. Natl. Acad. Sci. U.S.A. 109, 17621–17626 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Dunny G. M., Clewell D. B., Transmissible toxin (hemolysin) plasmid in Streptococcus faecalis and its mobilization of a noninfectious drug resistance plasmid. J. Bacteriol. 124, 784–790 (1975). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Perez-Casal J., Caparon M. G., Scott J. R., Mry, a trans-acting positive regulator of the M protein gene of Streptococcus pyogenes with similarity to the receptor proteins of two-component regulatory systems. J. Bacteriol. 173, 2617–2624 (1991). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Reguera R. M., Balaña-Fouce R., Showalter M., Hickerson S., Beverley S. M., Leishmania major lacking arginase (ARG) are auxotrophic for polyamines but retain infectivity to susceptible BALB/c mice. Mol. Biochem. Parasitol. 165, 48–56 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Camacho C., Coulouris G., Avagyan V., Ma N., Papadopoulos J., Bealer K., Madden T. L., BLAST+: Architecture and applications. BMC Bioinformatics 10, 421 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Treangen T. J., Sommer D. D., Angly F. E., Koren S., Pop M., Next generation sequence assembly with AMOS. Curr. Protoc. Bioinformatics 33, 11.8.1–11.8.18 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Friedman J., Alm E. J., Inferring correlation networks from genomic survey data. PLOS Comput. Biol. 8, e1002687 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary material for this article is available at http://advances.sciencemag.org/cgi/content/full/4/6/eaat0062/DC1
fig. S1. Induction of putrescine production by coculture of E. coli and En. faecalis.
fig. S2. Effects of extracellular pH and glucose concentration on bacterial metabolism in monocultures of En. faecalis and E. coli.
fig. S3. Viable bacterial counts and change of extracellular pH in cocultures of putrescine-deficient E. coli (SK930), wild-type En. faecalis (V583), and each Bifidobacterium sp.
fig. S4. Viable bacterial counts in feces of gnotobiotic mice.
fig. S5. Extracellular putrescine concentration and total viable bacterial counts in human feces incubated at different pH values (n = 5).
fig. S6. Effect of pH on extracellular agmatine concentration in monocultures of E. coli, Ci. youngae, and F. varium.
fig. S7. Symbiont-symbiont co-occurrence networks of key genes in the putrescine production pathway using previously described human microbiome data from U.S. metropolitan areas.
fig. S8. Symbiont-symbiont co-occurrence network patterns of key genes in the putrescine production pathway using previously described human microbiome data from Venezuela.
fig. S9. Symbiont-symbiont co-occurrence network patterns of key genes in the putrescine production pathway using previously described human microbiome data from Malawi.
fig. S10. Mechanistic model of a novel pathway for putrescine production from arginine through agmatine via the collaboration of three different bacterial species.
fig. S11. Outline of gnotobiotic mouse experiments.
table S1. List of species expressing homologs of the enzymes AdiA and AdiC, as determined by in silico analyses of 126 bacterial strains present in the human gut.
table S2. Detection of RNA sequences of AdiA, AdiC, AguA, and AguD by metatranscriptomic analysis of human feces.
table S3. List of bacteria used for screening of polyamine producing bacteria.
table S4. Strains, plasmids, and oligonucleotides used in this study.


