Abstract
Background
Incomplete adherence to tuberculosis (TB) treatment increases the risk of delayed culture conversion with continued transmission in the community, as well as treatment failure, relapse, and development or amplification of drug resistance. We conducted a systematic review and meta-analysis of adherence interventions, including directly observed therapy (DOT), to determine which approaches lead to improved TB treatment outcomes.
Methods and findings
We systematically reviewed Medline as well as the references of published review articles for relevant studies of adherence to multidrug treatment of both drug-susceptible and drug-resistant TB through February 3, 2018. We included randomized controlled trials (RCTs) as well as prospective and retrospective cohort studies (CSs) with an internal or external control group that evaluated any adherence intervention and conducted a meta-analysis of their impact on TB treatment outcomes. Our search identified 7,729 articles, of which 129 met the inclusion criteria for quantitative analysis. Seven adherence categories were identified, including DOT offered by different providers and at various locations, reminders and tracers, incentives and enablers, patient education, digital technologies (short message services [SMSs] via mobile phones and video-observed therapy [VOT]), staff education, and combinations of these interventions. When compared with DOT alone, self-administered therapy (SAT) was associated with lower rates of treatment success (CS: risk ratio [RR] 0.81, 95% CI 0.73–0.89; RCT: RR 0.94, 95% CI 0.89–0.98), adherence (CS: RR 0.83, 95% CI 0.75–0.93), and sputum smear conversion (RCT: RR 0.92, 95% CI 0.87–0.98) as well as higher rates of development of drug resistance (CS: RR 4.19, 95% CI 2.34–7.49). When compared to DOT provided by healthcare providers, DOT provided by family members was associated with a lower rate of adherence (CS: RR 0.86, 95% CI 0.79–0.94). DOT delivery in the community versus at the clinic was associated with a higher rate of treatment success (CS: RR 1.08, 95% CI 1.01–1.15) and sputum conversion at the end of two months (CS: RR 1.05, 95% CI 1.02–1.08) as well as lower rates of treatment failure (CS: RR 0.56, 95% CI 0.33–0.95) and loss to follow-up (CS: RR 0.63, 95% CI 0.40–0.98). Medication monitors improved adherence and treatment success and VOT was comparable with DOT. SMS reminders led to a higher treatment completion rate in one RCT and were associated with higher rates of cure and sputum conversion when used in combination with medication monitors. TB treatment outcomes improved when patient education, healthcare provider education, incentives and enablers, psychological interventions, reminders and tracers, or mobile digital technologies were employed. Our findings are limited by the heterogeneity of the included studies and lack of standardized research methodology on adherence interventions.
Conclusion
TB treatment outcomes are improved with the use of adherence interventions, such as patient education and counseling, incentives and enablers, psychological interventions, reminders and tracers, and digital health technologies. Trained healthcare providers as well as community delivery provides patient-centered DOT options that both enhance adherence and improve treatment outcomes as compared to unsupervised, SAT alone.
In this systematic review and meta-analysis, Payam Nahid and colleagues investigate results from trials and observational studies to identify methods for encouraging patients to complete tuberculosis treatment that are associated with improved TB outcomes.
Author summary
Why was this study done?
Tuberculosis remains a global problem and is the leading cause of death from a single infectious pathogen.
Tuberculosis treatment involves taking multiple medications daily for months to years, depending on the level of drug resistance, and failure to complete therapy as prescribed can lead to poor outcomes, including increased risk of failure, disease relapse, continued transmission, development of drug resistance, and death.
Public health programs and researchers have studied different ways of encouraging patients to take tuberculosis medications as prescribed in order to improve the likelihood of curing patients, but it is unclear which interventions are most effective.
What did the researchers do and find?
We systematically reviewed all published articles on this topic and found 129 research studies on different methods to encourage patients to complete tuberculosis treatment.
We found that providing patients with more support during their treatment increases the number of patients who successfully complete their treatment and reduces the number of patients who develop drug-resistant tuberculosis.
Some of these support measures include different methods of reminding patients to take their medication doses, such as having a healthcare worker observe them, sending them reminder text messages, or helping them with financial resources to make it to their clinic appointments.
What do these findings mean?
In addition to prescribing tuberculosis medications to patients with active tuberculosis, public health programs should include resources to help patients overcome individual challenges to completing treatment.
Using financial incentives, tools such as text messaging, and the use of healthcare workers to observe patients taking their medications at home or in the community can help patients overcome some of the barriers to treatment completion.
Combining multiple different types of patient-centered support interventions tailored to an individual’s needs and values can improve tuberculosis control efforts.
Introduction
Adherence to treatment is challenging, given the complexity, modest tolerability, and long duration of treatment regimens currently available for both drug-susceptible and -resistant tuberculosis (TB). In turn, low adherence increases the risk of poor outcomes, including treatment failure, relapse, and development or amplification of drug resistance [1–6]. Public health programs have used a variety of strategies to improve adherence at the health system level via financial incentives or enablers to offset the cost of accessing treatment, improving coordination and logistics around TB treatment delivery, and training healthcare providers. Other strategies tackle barriers to completing TB treatment by addressing knowledge gaps, attitudes, and behaviors surrounding adherence to TB treatment [7–11]. One of the most commonly used adherence interventions is directly observed therapy (DOT), in which a health worker, family member, or community member observes the patient taking TB medications [12]. In recent years, video-observed therapy (VOT) has gained attention as an alternative way of delivering DOT [13,14]. Other interventions aimed at supporting adherence through DOT include incentives, which are material or financial rewards provided to those adhering treatment [15], and enablers, which are interventions that allow patients to overcome economic constraints associated with DOT, such as absence from work or the direct and indirect patient costs of accessing TB treatment. Other interventions focus on providing education on TB, its treatment, and prevention to help patients make informed decisions and the healthcare team to deliver patient-centered care [16]. Reminder systems and patient tracers are targeted at assisting patients to keep appointments and to take action when patients miss appointments [17]. These interventions include reminder letters, phone calls, home visits, and, more recently, short message service (SMS) technology as well as electronic pill boxes. Psychological interventions aim to support via psychological or emotional counseling or a social network of peers undergoing TB treatment as a means of improving adherence to TB treatment [18,19]. Given the significant losses patients and the health system incur as a result of poor TB treatment outcomes, identifying those interventions that are most likely to improve adherence and outcomes, especially in resource-limited settings, is crucial.
The first pillar of the End TB Strategy of the World Health Organization (WHO)—Integrated, Patient Centered Care and Prevention—calls for “treatment of all people with tuberculosis including drug-resistant tuberculosis; and patient support” [20]. In 2015, WHO commissioned a series of systematic reviews and meta-analyses ahead of a Guideline Development Group meeting tasked with the revision of its TB treatment guidelines in accordance with the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) approach [21–23]. Until then, no evidence-based recommendations on TB treatment adherence and delivery existed. In order to inform this process, we conducted a systematic review and meta-analysis of studies of adherence interventions on drug-susceptible and -resistant TB treatment outcomes. Our goal was to identify any adherence interventions associated with improvement in TB treatment outcomes.
Methods
Search strategy and selection criteria
This study has been designed and reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA, S1 Text). The full protocol for this study is available in the supplementary material (S2 Text).
We included all randomized controlled trials (RCTs) as well as prospective and retrospective cohort studies (CSs). The population of interest included all adults or children in any setting undergoing active TB treatment. This included patients with pulmonary, extrapulmonary, smear-positive or -negative, and drug-susceptible and -resistant TB as well as patients with HIV coinfection. To be included, studies must have had an intervention targeted to increase adherence to TB treatment and an internal or historical control group (Table 1). We excluded articles on patients with only latent TB infection. We also excluded studies that compared DOT delivered in a hospital versus clinic setting because of a separate focused systematic review being conducted at the time of this review.
Table 1. PICO question breakdown for adherence interventions in TB treatment.
| Population | Intervention | Comparator | Outcome |
|---|---|---|---|
| Patients on treatment for drug-sensitive TB Patients on MDR-TB treatment Children (0–14 y) and adults TB patients infected with HIV and not infected with HIV |
Any intervention to promote treatment adherence
|
Routine practice* |
|
*Routine practice refers to regular TB drugs pickup and consultations with a physician or other healthcare workers being available when necessary, TB treatment being free of charge; and essential information/health education in relation to TB treatment being provided [24].
Abbreviations: DOT, directly observed therapy; MDR-TB, multidrug-resistant tuberculosis; PICO, population, intervention, comparison, outcome; SMS, short message service; TB, tuberculosis; VOT, video-observed therapy.
The following were the primary outcomes of interest as defined by WHO [25]: Cure, a pulmonary TB patient with bacteriologically confirmed TB at the beginning of treatment who was smear or culture negative in the last month of treatment and on at least one previous occasion; Treatment completion, a TB patient who completed treatment without evidence of failure but with no record to show that sputum smear or culture results in the last month of treatment and on at least one previous occasion were negative, either because tests were not done or because results are unavailable; Treatment success, the sum of “cure” and “treatment completion”; Treatment failure, a TB patient whose sputum smear or culture is positive at month 5 or later during treatment; Death, a TB patient who dies for any reason before starting or during the course of treatment; Loss to follow-up, a TB patient who did not start treatment or whose treatment was interrupted for two consecutive months or more; Relapse, a patient with a bacteriologically positive sputum smear or culture after the completion of any of the study TB regimens; Adherence, defined using parent study definitions such as being lost to follow-up (or default), isoniazid (INH) urine test, appointment keeping, etc.; and Development of resistance, identification of new drug resistance in a subsequent isolate otherwise matched with the baseline isolate. In post hoc analyses, given the number of articles that defined adherence as having taken more than 80%–90% of treatment doses, and for ease of comparison between studies, we chose to use this definition for the outcome of adherence when available.
We searched the literature using Medline with two search strategies, one to include all types of adherence interventions and the second targeted at SMS/VOT through February 3, 2018. The complete search strategy is available in S1 and S2 Tables. We also reviewed references of relevant articles and systematic reviews and contacted experts in the field for unpublished studies. We included all studies in the English language regardless of publication status or date. However, two foreign language articles were included, as data from them were previously abstracted by a different systematic review. Titles and abstracts were reviewed by one investigator (NA) and full manuscript content reviewed by multiple investigators (NA, PN, LJ, CM). Ethics approval was not required for this study, as all information was abstracted from published literature without access to any individual and/or identifiable data.
Data extraction and variable definitions
Using a standard data abstraction sheet, the following data were recorded from articles that met our inclusion criteria: patient selection, type of TB (pulmonary or extrapulmonary), HIV coinfection, treatment outcomes, method of blinding and randomization, results of drug-susceptibility testing, acid-fast bacilli (AFB) smear and culture results, mode of supervision of therapy, type of adherence intervention, and study results for the outcomes of interest (adjusted or unadjusted risk ratios [RRs], risk differences [RDs], raw data). We chose to use unadjusted RRs preferentially if these data were available. When available, data were gathered from both per protocol and intention-to-treat analyses. The quality of the studies was assessed using the Cochrane Risk of Bias tool for RCTs, and the Newcastle-Ottawa Scale was used for observational studies [26,27].
We grouped the adherence interventions identified across studies into the following categories: (1) DOT referred to the act of observing a patient swallow medications, and self-administered therapy (SAT) was defined as the patient taking each medication dose without supervision; (2) education and counseling interventions were those aimed at providing adequate knowledge and ensuring patient understanding of the disease process and risks and benefits associated with treatment adherence; (3) incentives were interventions to promote treatment adherence through a financial or material reward and enablers were interventions that allowed patients to overcome barriers to treatment adherence (cost, distance, availability); (4) reminders included any intervention made prior to the patient taking medications or attending appointments to serve as a reminder and tracers involved contacts made after a patient had failed adherence in order to improve subsequent adherence to treatment; (5) psychological interventions aimed to provide emotional or psychological support aimed at reducing stigma and increasing treatment adherence; (6) digital health interventions included any of the above categories implemented via mobile electronic devices (SMS, VOT, medication monitors); and lastly, (7) mixed interventions included a combination of the aforementioned interventions to address barriers to adherence based on patient-specific needs and values. Thus, the term “patient-centered DOT” or “enhanced DOT” was used to designate any study using adherence interventions spanning the multiple categories mentioned above in conjunction with DOT. With respect to DOT provider type, a lay provider was defined as an untrained volunteer, including family members or other patient-designated person. A trained health worker included any health worker, community member, or volunteer who had received any form of formal training for DOT. For the categories of patient education and counseling, staff education, incentives and enablers, reminders and tracers, and psychological interventions, studies included in the meta-analysis had to have employed the same supervision modality (either DOT or SAT) for both the intervention and control arms.
All estimates of effect for dichotomous outcomes were reported as RRs with 95% confidence intervals. When two or more studies were available on a particular outcome, random effects meta-analysis was performed to obtain a pooled estimate of treatment effect and pooled RR between the intervention and control arms. Heterogeneity was assessed visually using forest plots and statistically using the χ2 and I2 tests. If more than 10 studies were available for a particular comparison, we used funnel plots to determine publication bias. All analyses were conducted in RevMan5 [28]. All figures were generated using RevMan and GraphPad Prism 7.0 [29]. If a study reported no events in the intervention or control group for an outcome of interest, the RD was calculated.
Results
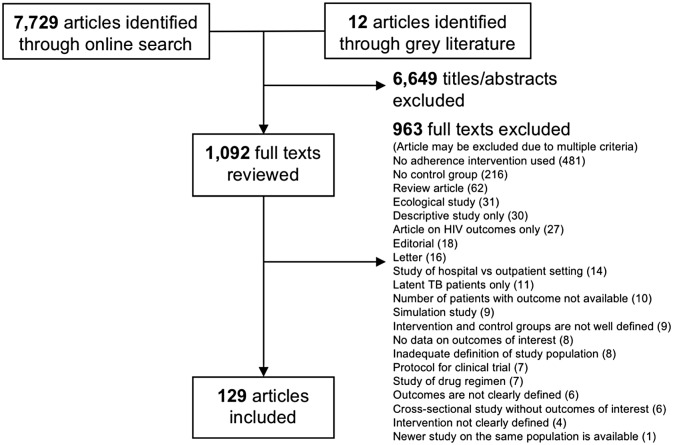
Title and abstract literature review yielded 7,729 articles, of which 1,092 met the inclusion criteria for full text review (Fig 1). References of 32 systematic reviews found through our online search were also reviewed for relevant articles. A final 129 articles met the inclusion criteria for quantitative analysis. Characteristics of included studies as well as quality assessments are summarized in S3 Table, S1 and S2 Figs. The overall quality of included studies varied significantly amongst CSs. The quality of RCTs was limited predominantly by lack of blinding, given the nature of adherence interventions. Below is a brief summary of findings by each type of adherence intervention (Table 2). When available, data from RCTs and CSs are listed in parentheses. Some of the analyses contained significant heterogeneity, as measured by I2, which has been denoted on their corresponding figures.
Fig 1. PRISMA summary.
TB, tuberculosis.
Table 2. Summary of the effects of different types of adherence interventions on TB treatment outcomes.
| Outcomes |
SAT versus any DOT (No. of studies) |
SAT versus any DOT-HIV/TB (No. of studies) |
Family/community DOT versus HCW DOT (No. of studies) |
Lay provider DOT versus HCW DOT (No. of studies) |
Home DOT versus clinic DOT (No. of studies) |
Community DOT versus clinic DOT (No. of studies) |
Home DOT versus community DOT (No. of studies) |
Patient education2 (No. of studies) |
Incentives/enablers2 (No. of studies) |
Reminders/tracers2 (No. of studies) |
Patient-centered DOT versus SAT (No. of studies) |
Patient-centered DOT versus DOT (No. of studies) |
Patient-centered SAT versus SAT (No. of studies) |
Psychological interventions2 (No. of studies) |
Staff education2 (No. of studies) |
Phone reminders2 (No. of studies) |
VOT versus DOT (No. of studies) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mortality—CSs | Ø (23) |
↑ (4) |
Ø (3) |
Ø (3) |
↑ (8) |
Ø (6) |
Ø (2) |
-- | ↓ (3) |
Ø (3) |
Ø (4) |
Ø (4) |
-- | Ø (1) |
-- | Ø (2) |
Ø (1) |
| Mortality—RCTs | Ø (4) |
-- | -- | -- | -- | Ø (2) |
Ø (1) |
Ø (2) |
Ø (2) |
Ø (3) |
Ø (1) |
↓ (2) |
-- | -- | Ø (2) |
Ø (1) |
-- |
| Success—CSs | ↓ (4) |
↓ (3) |
Ø (3) |
Ø (2) |
↑ (3) |
↑ (9) |
↓ (1) |
-- | ↑ (4) |
Ø (2) |
↑ (2) |
↑ (4) |
-- | -- | ↑ (1) |
-- | -- |
| Success—RCTs | ↓ (5) |
-- | -- | -- | -- | Ø (1) |
Ø (2) |
Ø (2) |
↑ (3) |
↑ (4) |
↑ (1) |
↑ (2) |
-- | -- | Ø (3) |
Ø (3) |
-- |
| Completion—CSs | Ø (14) |
↓ (1) |
Ø (2) |
Ø (1) |
Ø (5) |
Ø (3) |
Ø (2) |
-- | Ø (4) |
Ø (1) |
↑ (2) |
Ø (2) |
-- | ↑ (1) |
-- | Ø (2) |
Ø (2) |
| Completion—RCTs | Ø (5) |
-- | -- | -- | -- | Ø (1) |
-- | ↑ (1) |
↑ (2) |
Ø (3) |
↑ (1) |
Ø (2) |
-- | ↑ (1) |
Ø (2) |
Ø (1) |
-- |
| Cure—CSs | ↓ (18) |
↓ (2) |
Ø (3) |
Ø (1) |
Ø (6) |
Ø (6) |
Ø (2) |
-- | ↑ (4) |
↑ (2) |
↑ (2) |
Ø (2) |
-- | -- | -- | ↑ (2) |
-- |
| Cure—RCTs | Ø (4) |
-- | -- | -- | -- | Ø (2) |
Ø (1) |
↑ (1) |
Ø (1) |
Ø (2) |
↑ (2) |
↑ (2) |
-- | Ø (1) |
Ø (3) |
Ø (1) |
-- |
| Failure—CSs | Ø (15) |
Ø (5) |
↑ (3) |
Ø (2) |
Ø (4) |
↓ (6) |
Ø (2) |
-- | Ø (2) |
Ø (3) |
Ø (2) |
Ø (2) |
-- | -- | -- | Ø (3) |
-- |
| Failure—RCTs | Ø (2) |
-- | -- | -- | -- | Ø (1) |
Ø (1) |
Ø (1) |
↓ (1) |
Ø (3) |
-- | Ø (2) |
-- | -- | Ø (2) |
Ø (1) |
-- |
| Loss to follow-up—CSs | Ø (21) |
Ø (3) |
Ø (3) |
Ø (2) |
Ø (7) |
↓ (6) |
Ø (2) |
-- | ↓ (5) |
↓ (4) |
Ø (4) |
Ø (4) |
-- | ↓ (1) |
-- | Ø (2) |
-- |
| Loss to follow-up—RCTs | Ø (4) |
-- | -- | -- | -- | Ø (2) |
Ø (1) |
Ø (3) |
↓ (1) |
Ø (4) |
↓ (1) |
↓ (3) |
-- | Ø (1) |
Ø (2) |
Ø (1) |
-- |
| Relapse—CSs | Ø (6) |
Ø (1) |
-- | -- | -- | -- | -- | -- | -- | -- | Ø (1) |
-- | -- | -- | -- | -- | -- |
| Relapse—RCTs | Ø (1) |
-- | -- | -- | -- | -- | -- | -- | -- | -- | -- | -- | -- | -- | -- | -- | -- |
| Adherence—CSs | ↓ (1) |
-- | ↓ (1) |
-- | ↓ | -- | -- | ↑ (1) |
-- | -- | -- | -- | -- | -- | -- | -- | -- |
| Adherence—RCTs | Ø (1) |
-- | -- | -- | -- | -- | -- | ↑ (1) |
-- | ↑ (1) |
-- | ↑ (1) |
Ø (1) |
-- | -- | -- | -- |
| Smear conversion—CSs | Ø (2) |
-- | -- | -- | ↑ | ↑ (2) |
-- | -- | -- | -- | -- | -- | -- | -- | -- | ↑ (1) |
-- |
| Smear conversion—RCTs | ↓ (1) |
-- | -- | -- | -- | Ø (1) |
-- | -- | ↑ (1) |
↑ (3) |
↑ (1) |
-- | -- | -- | -- | Ø (1) |
-- |
| Development of drug resistance—CSs | ↓ (3) |
-- | -- | -- | -- | -- | -- | -- | -- | ↓ (1) |
Ø (1) |
-- | -- | -- | -- | -- | -- |
| Development of drug resistance-RCTs | -- | -- | -- | -- | -- | -- | -- | -- | Ø (1) |
-- | -- | -- | -- | -- | -- | -- | -- |
| Unfavorable outcome*—CSs | -- | -- | -- | -- | -- | ↓ (1) |
-- | -- | -- | -- | -- | -- | -- | -- | -- | -- | -- |
| Poor adherence1—RCTs | -- | -- | -- | -- | -- | -- | -- | -- | -- | -- | -- | -- | -- | -- | -- | Ø (1) |
-- |
Ø No evidence of a difference with the use of intervention versus control.
↑ Statistically significant increased risk of outcome associated with the use of intervention versus control.
↓ Statistically significant decreased risk of outcome associated with the use of intervention versus control.
-- No available data for comparison.
*Unfavorable outcome is defined as combined failure, default, death, or transfer out by the study.
1Percentage of patient-months during which >20% of doses were missed.
2Comparison of adherence intervention in addition to standard of care versus standard of care alone. Standard of care was DOT or SAT, depending on study setting.
Abbreviations: CS, cohort study; DOT, directly observed therapy; HCW, healthcare worker; RCT, randomized controlled trial; SAT, self-administered therapy.
SAT versus DOT
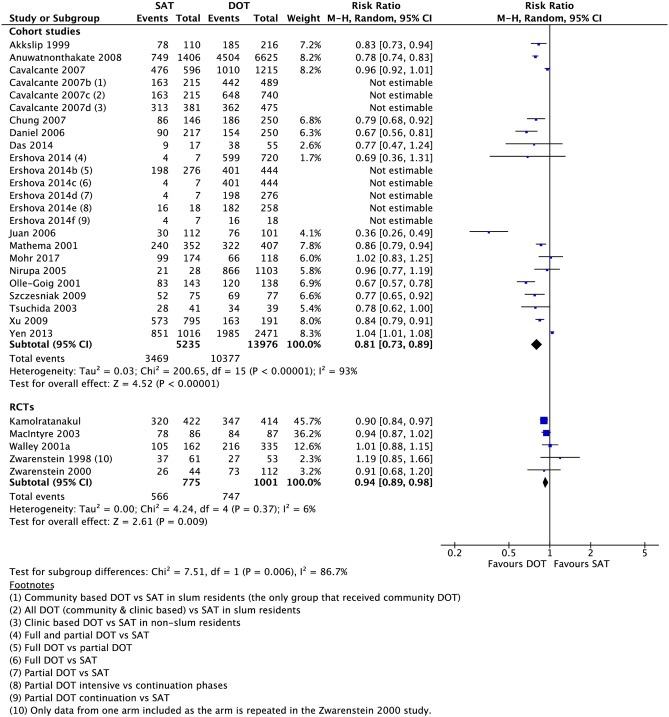
Forty-six studies were included in the meta-analysis, seven of which were RCTs [4,30–74]. Six CSs included primarily HIV/TB patients [51,53–55,57,72] and two CSs involved primarily MDR-TB patients [56,72]. DOT was offered daily or intermittently at home, clinic, or in the community. DOT providers ranged from family members to trained lay providers and healthcare providers.
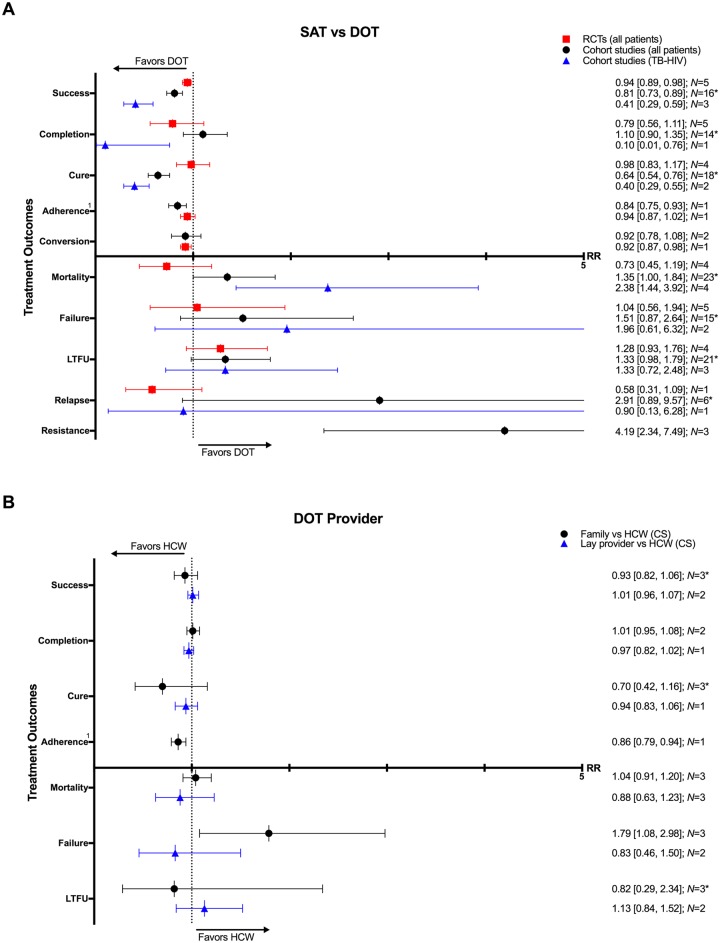
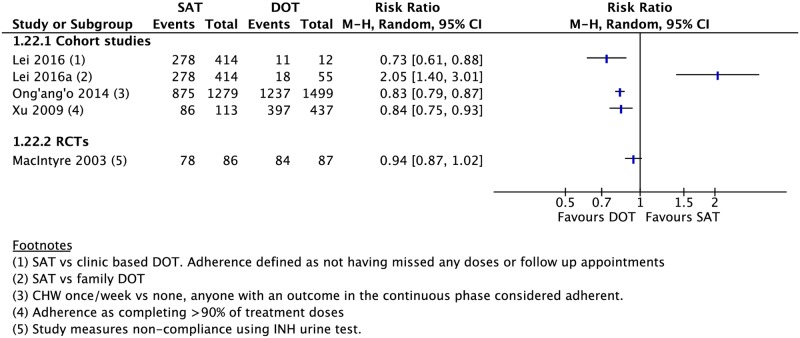
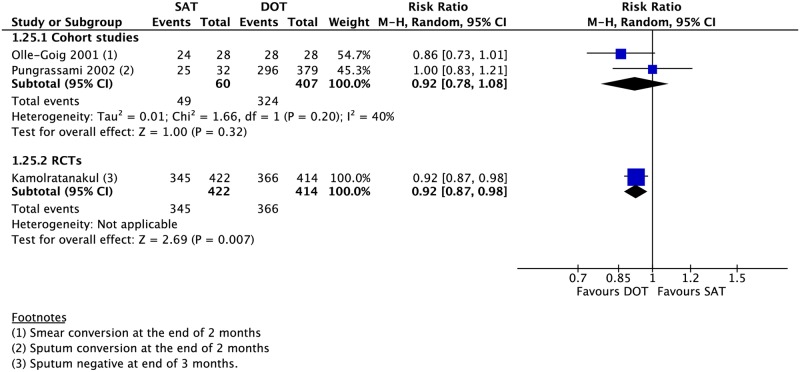
Compared to those undergoing any DOT, participants who received SAT had a lower rate of treatment success (16 CSs: RR 0.81, 95% CI 0.73–0.89; 5 RCTs: RR 0.94, 95% CI 0.89–0.98), cure (18 CSs: RR 0.64, 95% CI 0.54–0.76; 4 RCTs: 0.98, 95% CI 0.83–1.17), and adherence (1 CS: RR 0.84, 95% CI 0.75–0.93; 1 RCT: RR 0.94, 95% CI 0.87–1.02) (Figs 2–5). One RCT found a lower rate of smear conversion at the end of two months amongst SAT patients (RR 0.92, 95% CI 0.87–0.98) (Fig 6). There was no significant difference between DOT and SAT amongst all CSs and RCTs for the outcomes of mortality (23 CSs, 4 RCTs), treatment completion (14 CSs, 5 RCTs), treatment failure (15 CSs, 5 RCTs), loss to follow-up (21 CSs, 4 RCTs), relapse (6 CSs, 1 RCT), or development of drug resistance (3 CSs) (S3–S14 Figs).
Fig 2.
(A) SAT compared with DOT on TB treatment outcomes. (B) Impact of any DOT provided by lay providers, family members, or healthcare workers on TB treatment outcomes. * = significant heterogeneity in the meta-analysis as determined by I2 statistic. 1 = depicted is the rate of adherence in one study defined as completing >90% of treatment doses by pill counting in one CS and based on six positive INH urine tests done at random in one RCT. Conversion = sputum conversion to negative at the end of two months (CS) and three months (RCT). N = number of studies included within the meta-analysis. Resistance = development of drug resistance. CS, cohort study; DOT, directly observed therapy; HCW, healthcare worker; INH, isoniazid; LTFU, loss to follow-up; RCT, randomized controlled trial; RR, risk ratio; SAT, self-administered therapy; TB, tuberculosis.
Fig 5. Forest plot of adherence rates in studies comparing patients undergoing SAT versus DOT.
CHW, community health worker; DOT, directly observed therapy; INH, isoniazid; M-H, Mantel-Haenszel; SAT, self-administered therapy.
Fig 6. Meta-analysis of rates of sputum conversion in patients undergoing SAT versus DOT.
DOT, directly observed therapy; M-H, Mantel-Haenszel; RCT, randomized controlled trial; SAT, self-administered therapy.
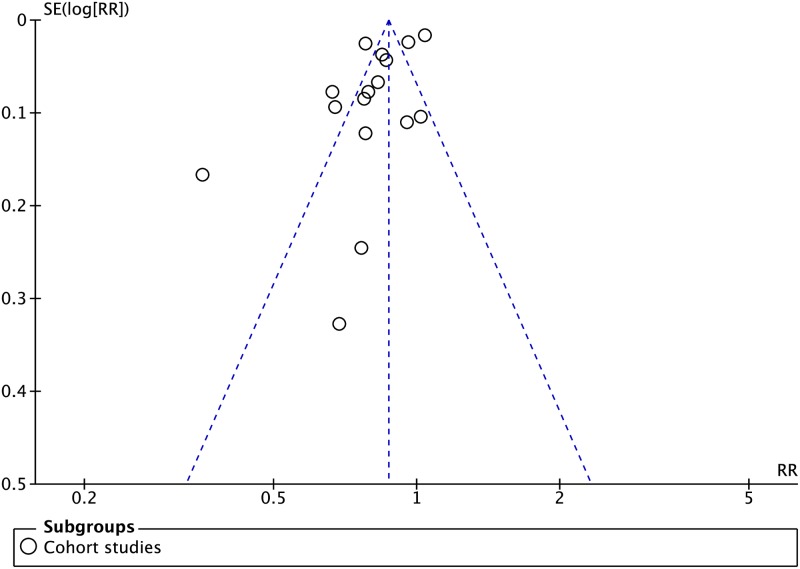
Fig 3. Funnel plot of cohort studies comparing treatment success rates in patients undergoing SAT versus DOT. No funnel plot of RCTs has been included as there were fewer than 10 RCTs.
DOT, directly observed therapy; RCT, randomized controlled trial; RR, risk ratio; SAT, self-administered therapy; SE, standard error.
Fig 4. Meta-analysis of treatment success rates in patients undergoing SAT versus DOT.
DOT, directly observed therapy; M-H, Mantel-Haenszel; SAT, self-administered therapy.
An assessment of the publication bias (S3, S5, S7, S9 and S11 Figs) showed asymmetry for the outcomes of mortality, treatment success, and loss to follow-up. Notably, a few smaller studies found worse outcomes with SAT, as compared with DOT. The plots reflect the significant heterogeneity between the studies and may also be suggestive of publication bias (i.e., reporting bias in CSs).
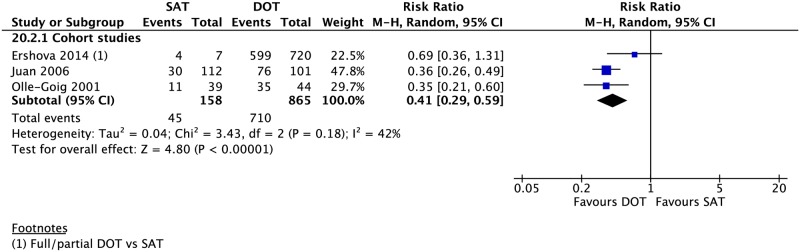
In patients with HIV/TB, SAT was associated with lower rates of treatment success (3 CSs: RR 0.41, 95% CI 0.29–0.59), completion (1 CS: RR 0.10, 95% CI 0.01–0.76), and cure (2 CSs: RR 0.40, 95% CI 0.29–0.55) as well as a higher rate of mortality (4 CSs: RR 2.38, 95% CI 1.44–3.92). The difference in the rates of loss to follow-up (3 CSs: RR 1.33, 95% CI 0.72–2.48), treatment failure (2 CSs: RR 1.96, 95% CI 0.61–6.32), and relapse (1 CS: RR 0.90, 95% CI 0.13–6.28) between the two groups (Figs 2, 7 and S16–S20 Figs) was not significant. Although several studies in our meta-analysis included patients with MDR-TB in their cohort, all but two reported aggregate data on treatment outcomes without stratifying by drug-resistant status. Of the 28 MDR-TB patients in one retrospective CS in New York City from 1987–1997 [56], 11 underwent SAT and 17 received DOT. The mortality, treatment completion, and nonadherence rates were not significantly different between the two groups (S21–S24 Figs). A prospective CS in South Africa of rifampicin-resistant patients found no evidence of a difference in rates of mortality, treatment failure, and loss to follow-up between those undergoing DOT for the entire duration of TB treatment and those who only received DOT during the first six months [72].
Fig 7. Meta-analysis of treatment success rates in HIV/TB patients undergoing SAT versus DOT.
DOT, directly observed therapy; M-H, Mantel-Haenszel; SAT, self-administered therapy; TB, tuberculosis.
DOT provider type
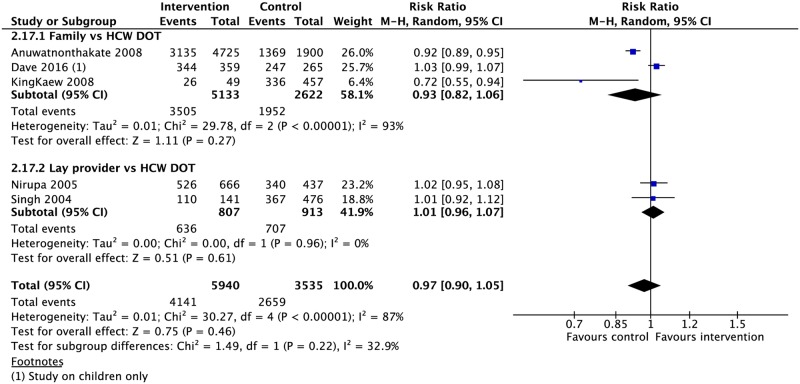
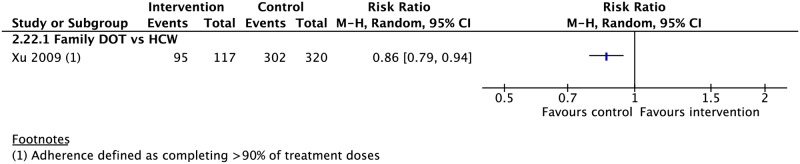
Seven CSs compared treatment outcomes in patients who received DOT by family/lay providers or healthcare workers [42,49,63,75–78]. Family member DOT was associated with a lower rate of adherence (1 CS: RR 0.86, 95% CI 0.79–0.94) and higher rate of treatment failure (3 CSs: RR 1.79, 95% CI 1.08–2.98) compared with trained health worker DOT (Fig 2). There was no evidence of a difference in rates of treatment success, completion, cure, mortality, and loss to follow-up between the two groups (Figs 2, 8, 9 and S25–S29 Figs). DOT by lay providers showed no significant difference in outcomes when compared with healthcare worker (HCW) DOT.
Fig 8. Meta-analysis of treatment success rates in patients receiving DOT by different types of providers.
DOT, directly observed therapy; HCW, healthcare worker; M-H, Mantel-Haenszel.
Fig 9. Comparison of adherence rates in patients receiving DOT by a family member versus HCW.
DOT, directly observed therapy; HCW, healthcare worker; M-H, Mantel-Haenszel.
DOT locations
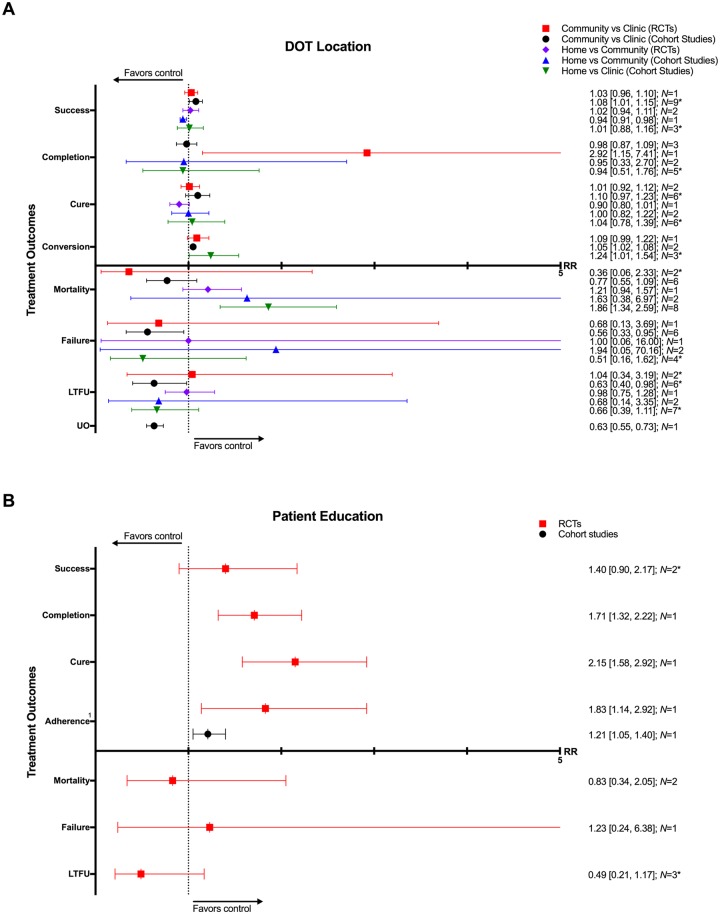
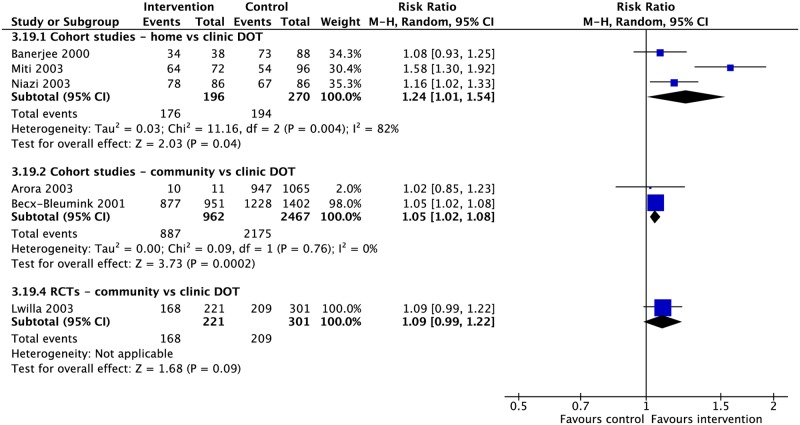
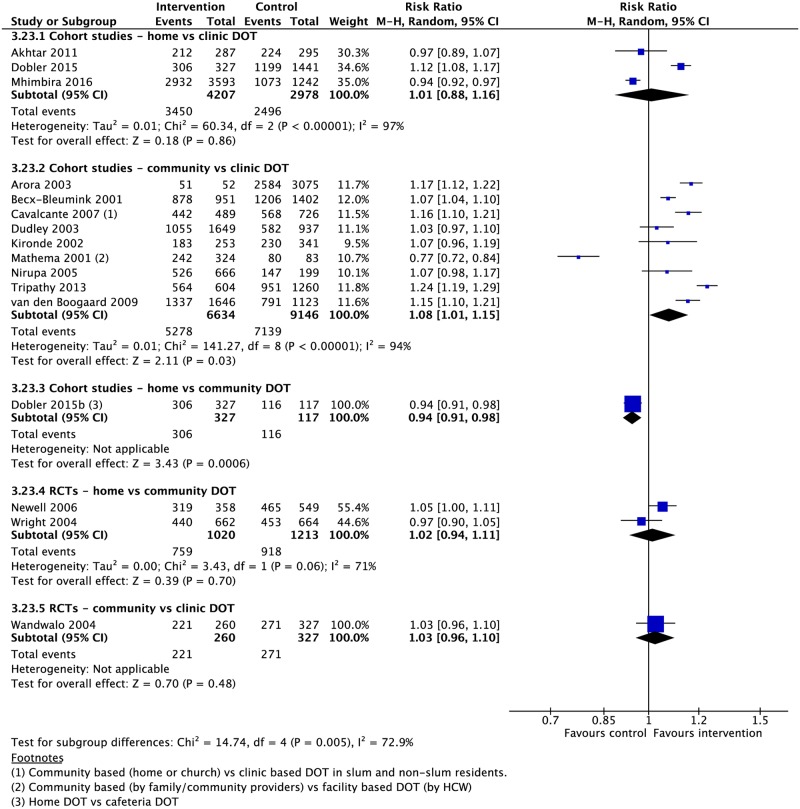
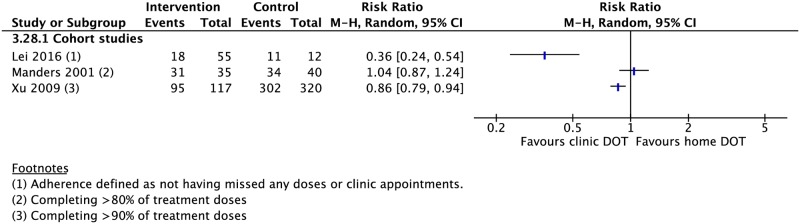
Four RCTs and 21 CSs compared DOT offered at different locations, including the home, community, and clinic [37,39,42,49,61,73,79–97]. In comparison with clinic-based DOT, treatment in the community was associated with higher rates of treatment completion (1 RCT: RR 2.92, 95% CI 1.15–7.41; 3 CSs: RR 0.87, 95% CI 0.87–1.09) and sputum conversion at the end of two months (2 CSs: RR 1.05, 95% CI 1.02–1.08; 1 RCT: RR 1.09, 95% CI 0.99–1.22) as well as lower rates of loss to follow-up (6 CSs: RR 0.63, 95% CI 0.40–0.98; 2 RCTs: RR 1.04, 95% CI 0.34–3.19), treatment failure (6 CSs: RR 0.56, 95% CI 0.33–0.95; 1 RCT: RR 0.68, 95% CI 0.13–3.69), and unfavorable outcome (1 CS: RR 0.63, 95% CI 0.55–0.73) (Figs 10–13 and S30–S35 Figs). No significant differences were noted for the outcomes of mortality or cure. Home-based DOT was associated with a lower rate of treatment adherence when compared with clinic-based DOT in one CS, in which patients were observed by family members or lay providers (1 CS: RR 0.86, 95% CI 0.79–0.94), while another found no significant difference in adherence rates when comparing home and clinic based DOT. Home-based DOT was associated with a higher mortality rate (8 CSs: RR 1.86, 95% CI 1.34–2.59). This effect is nullified with the removal of the study by Mhimbira and colleagues, who defined home-based DOT as DOT delivered at home by a treatment supporter of patients’ choosing. When compared with a particular type of community-based DOT that included offering a free lunch to patients, home-based DOT had a lower success rate in one CS (1 CS: RR 0.94, 95% CI 0.92–0.98; 2 RCTs: RR 1.02, 95% CI 0.94–1.11) but no significant difference in rates of treatment completion, cure, mortality, failure, and loss to follow-up in other studies. Home-based DOT was associated with marginally higher rates of sputum conversion at two months, when compared with clinic-based DOT (3 CSs: RR 1.24, 95% CI 1.01–1.54).
Fig 10.
(A) Impact of DOT provided at home, in the community, or in clinic on TB treatment outcomes. (B) Impact of patient education and counseling interventions on TB treatment outcomes. * = significant heterogeneity in the meta-analysis, as determined by I2 statistic. 1 = adherence defined as the proportion of patients that took >75% of prescribed doses in one CS and the proportion of patients attending all appointments in one RCT. Conversion = sputum conversion to negative at the end of two months. N = number of studies included within the meta-analysis. Composite outcome reported by one study defined as combined failure, default, death, or transfer out. CS, cohort study; DOT, directly observed therapy; LTFU, loss to follow-up; RCT, randomized controlled trial; RR, risk ratio; TB, tuberculosis; UO, unfavorable outcome.
Fig 13. Meta-analysis of sputum conversion rates at two months in patients receiving DOT at various locations.
DOT, directly observed therapy; M-H, Mantel-Haenszel.
Fig 11. Meta-analysis of treatment success rates in patients receiving DOT in various locations.
DOT, directly observed therapy; HCW, healthcare worker; M-H, Mantel-Haenszel; RCT, randomized controlled trial.
Fig 12. Adherence rates in patients receiving DOT at various locations.
DOT, directly observed therapy; M-H, Mantel-Haenszel.
Patient education and counseling
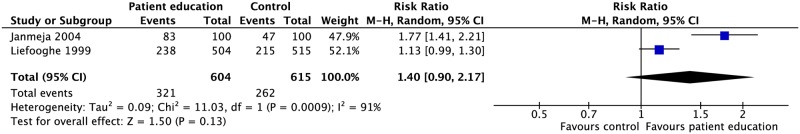
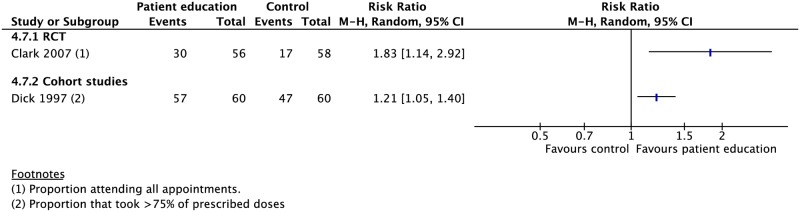
Four RCTs and one CS evaluated the effect of oral and written educational material as well as counseling on TB treatment outcomes [7–11]. Education and counseling was associated with a higher rate of treatment completion (1 RCT: RR 1.71, 95% CI 1.32–2.22), cure (1 RCT: RR 2.15, 95% CI 1.58–2.92), and adherence (1 RCT: RR 1.83, 95% CI 1.14–2.92; 1 CS: RR 1.21, 95% CI 1.05–1.40) (Figs 10, 14, 15 and S36–S40 Figs). These interventions had no meaningful impact on rates of mortality, treatment success, failure, or loss to follow-up.
Fig 14. Meta-analysis of treatment success rates in patients receiving patient education and counseling interventions in addition to standard care versus standard care alone.
M-H, Mantel-Haenszel.
Fig 15. Adherence rates in patients receiving patient education and counseling interventions in addition to standard care versus standard care alone.
M-H, Mantel-Haenszel; RCT, randomized controlled trial.
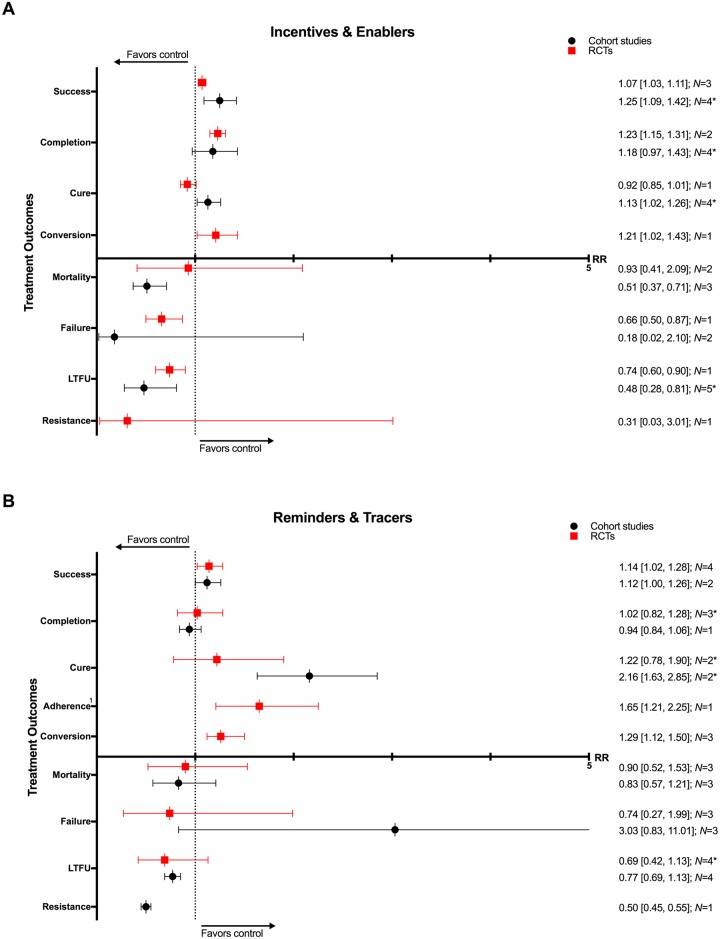
Incentives and enablers
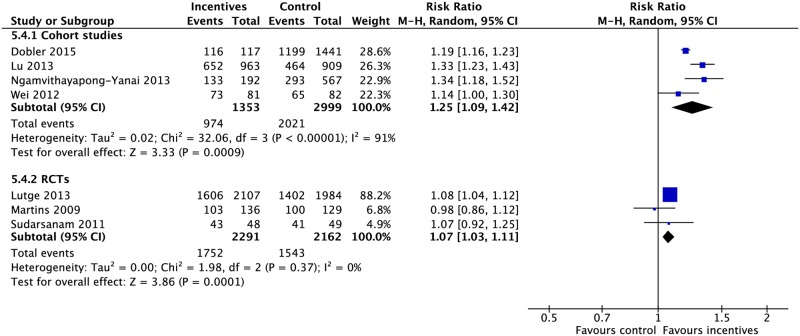
Incentives and enablers in four RCTs and eleven CSs were associated with lower rates of mortality (3 CSs: RR 0.51, 95% CI 0.37–0.71; 2 RCTs: RR 0.93, 95% CI 0.41–2.09), treatment failure (1 RCT: RR 0.66, 95% CI 0.50–0.87; 2 CSs: RR 0.18, 95% CI 0.02–2.10), and loss to follow-up (1 RCT: RR 0.74, 95% CI 0.60–0.90; 5 CSs: RR 0.48, 95% CI 0.28–0.81) as well as higher rates of treatment success (3 RCTs: RR 1.07, 95% CI 1.03–1.11; 4 CSs: RR 1.25, 95% CI 1.09–1.42), completion (2 RCTs: RR 1.23, 95% CI 1.15–1.31; 4 CSs: RR 1.18, 95% CI 0.97–1.43), cure (4 CSs: 1.13, 95% CI 1.02–1.26; 1 RCT: RR 0.92, 95% CI 0.85–1.01), and sputum conversion at two months (1 RCT: RR 1.21, 95% CI 1.02–1.43). (Figs 16, 17 and S41–S47 Figs) [83,98–111].
Fig 16.
(A) Impact of incentives and enablers on TB treatment outcomes. (B) Impact of reminders and tracers on TB treatment outcomes. * = significant heterogeneity in the meta-analysis as determined by I2 statistic. 1 = adherence defined as the proportion of patients who presented for all drug collections in the first six months of treatment. Conversion = sputum conversion at the end of two months. Resistance = development of drug resistance. N = number of studies included within the meta-analysis. LTFU, loss to follow-up; RCT, randomized controlled trial; RR, risk ratio; TB, tuberculosis.
Fig 17. Meta-analysis of treatment success rates in patients receiving incentives and enablers in addition to standard care versus standard care alone.
M-H, Mantel-Haenszel; RCT, randomized controlled trial.
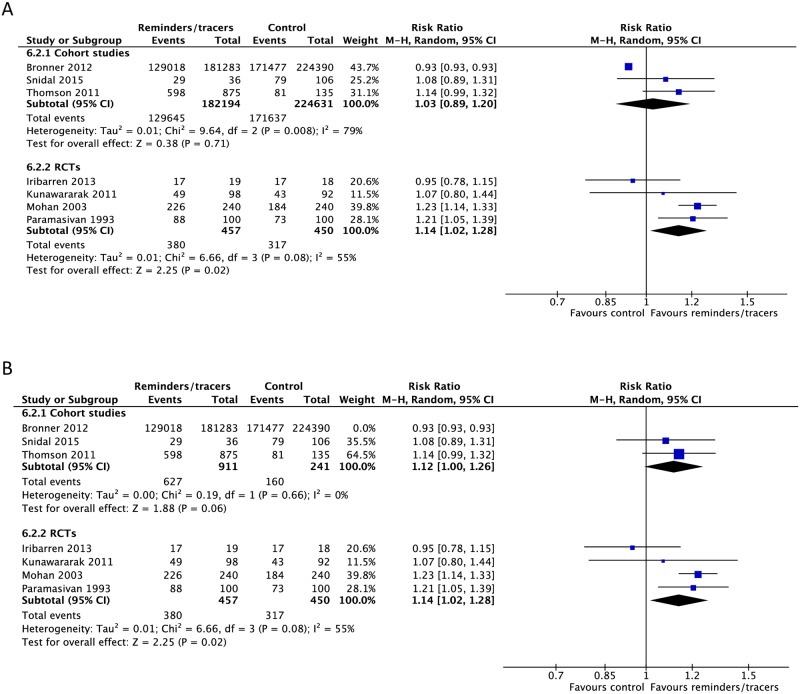
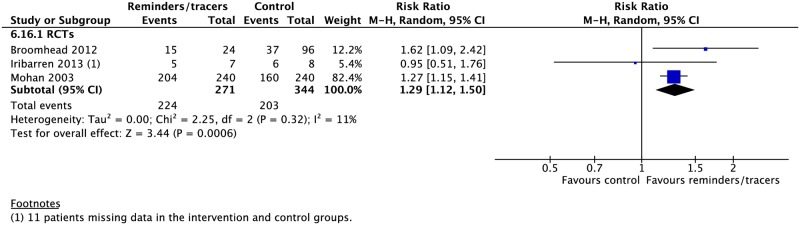
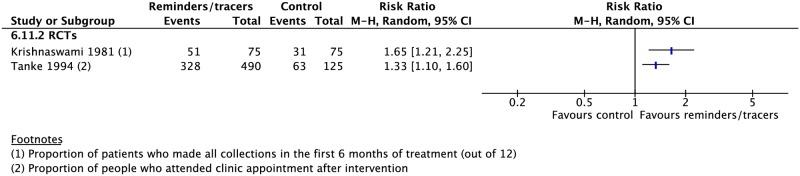
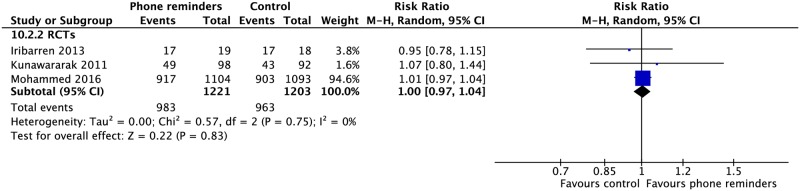
Reminders and tracers
Nine RCTs and six CSs found higher rates of treatment success (4 RCTs: RR 1.14, 95% CI 1.02–1.28; 2 CSs: RR 1.12, 95% CI 1.00–1.26), cure (2 CSs: RR 2.16, 95% CI 1.63–2.85; 2 RCTs: RR 1.22, 95% CI 0.78–1.90), adherence (1 RCT: RR 1.65, 95% CI 1.21–2.25), and sputum conversion at two months (3 RCTs: RR 1.29, 95% CI 1.12–1.50) and lower rates of development of drug resistance (1 CS: RR 0.50, 95% CI 0.45–0.55) and loss to follow-up (4 CSs: RR 0.77, 95% CI 0.69–1.13; 4 RCTs: RR 0.69, 95% CI 0.42–1.13) with the use of reminders and tracers (Figs 16, 18–20 and S48–S55 Figs) [112–126]. One study by Bronner and colleagues was removed in sensitivity analyses due to lack of controlling for baseline differences in treatment outcomes prior to intervention in the intervention and control districts (Fig 18).
Fig 18.
(A) Meta-analysis of treatment success rates in patients receiving reminders/tracers in addition to standard care versus standard care alone. (B) Sensitivity analysis: removing the heaviest weighted study (Bronner 2012) in which control and intervention cohorts had different pre-intervention success rates. M-H, Mantel-Haenszel; RCT, randomized controlled trial.
Fig 20. Meta-analysis of rates of sputum conversion at two months in patients receiving reminders/tracers in addition to standard care versus standard care alone.
M-H, Mantel-Haenszel; RCT, randomized controlled trial.
Fig 19. Adherence rates in patients receiving reminders/tracers in addition to standard care versus standard care alone.
M-H, Mantel-Haenszel; RCT, randomized controlled trial.
Staff education
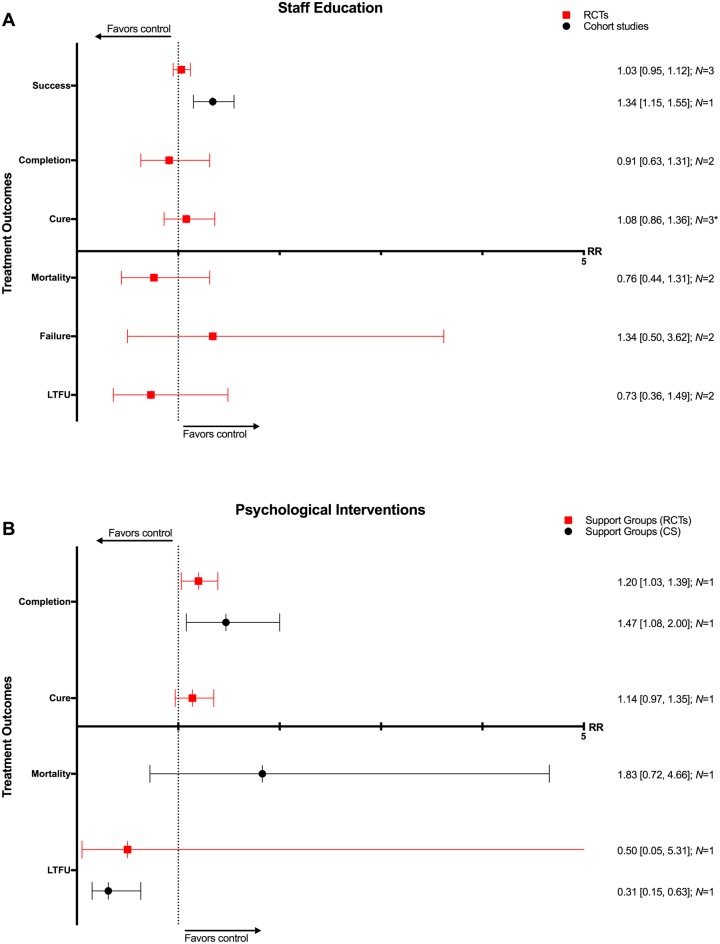
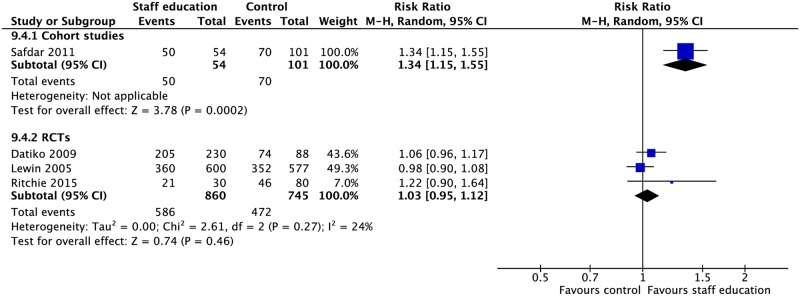
Three RCTs and one CS involved interventions such as adherence education for staff, peer training for lay health workers, reminders to initiate adherence discussions, and educational tools and aids for decision-making [127–130]. These interventions were associated with higher rates of treatment success (1 CS: RR 1.34, 95% CI 1.15–1.55) and lower rates of loss to follow-up (1 CS: RD −0.18, 95% CI −0.26–−0.10) in the one CS. The three RCTs found no significant difference in rates of TB treatment outcomes with the use of such interventions (Figs 21, 22 and S56–S61 Figs).
Fig 21.
(A) Impact of staff education on TB treatment outcomes. (B) Impact of using psychological interventions, such as mental health counseling and support groups, on TB treatment outcomes. * = significant heterogeneity in the meta-analysis, as determined by I2 statistic. N = number of studies included within the meta-analysis. CS, cohort study; LTFU, loss to follow-up; RCT, randomized controlled trial; RR, risk ratio; TB, tuberculosis.
Fig 22. Meta-analysis of the impact of staff education on treatment success rates.
M-H, Mantel-Haenszel; RCT, randomized controlled trial.
Psychological interventions
One RCT focused on brief counseling interventions for alcohol cessation and another offered self-help groups [131,132]. One prospective CS evaluated the impact of TB clubs as a support network [133]. Support groups were associated with higher rates of treatment completion (1 CS: RR 1.47, 95% CI 1.08–2.00; 1 RCT: RR 1.20, 95% CI 1.03–1.39) and lower rates of treatment failure (1 RCT: RD −0.12, 95%CI −0.22–−0.01; 1 CS: RD −0.02, 95% CI −0.06–0.03) and loss to follow-up (1 CS: RR 0.31, 95% CI 0.15–0.63; 1 RCT: RR 0.50, 95% CI 0.05–5.31) (Fig 21 and S62–S67 Figs).
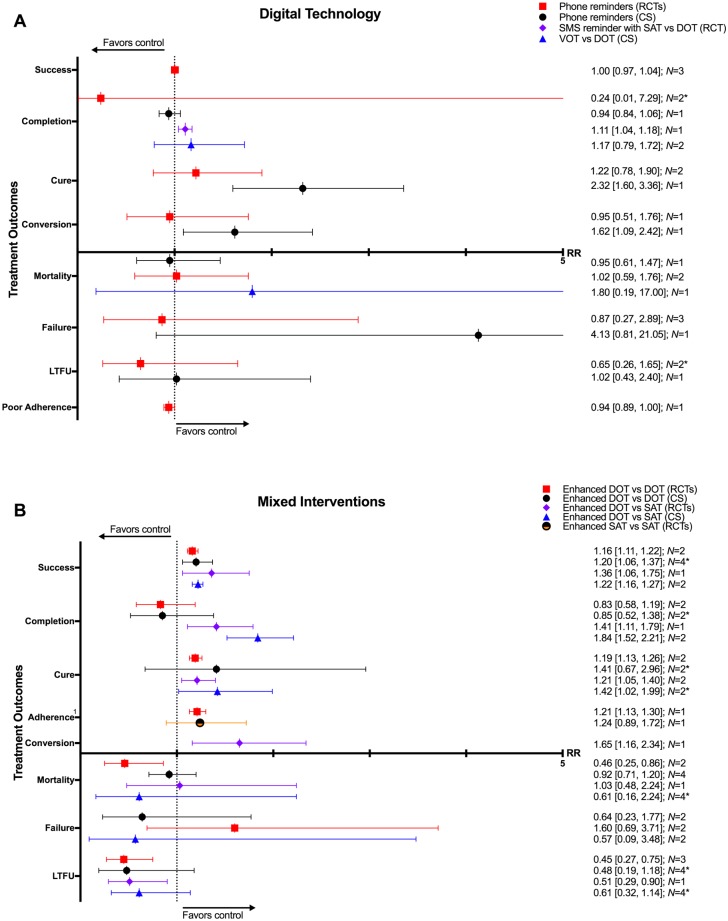
Digital health
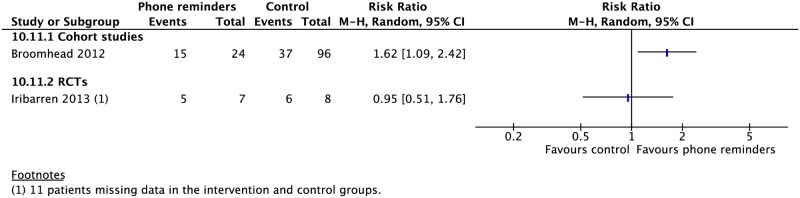
Five RCTs and two CSs looked at daily reminder texts or phone call reminders to take medications for patients undergoing SAT or family DOT [114,117,119,120,125,126,134]. Two CSs evaluated the impact of VOT [135,136]. In one RCT comparing SMS reminders in patients undergoing SAT versus those undergoing DOT, SAT patients had a higher treatment completion rate (1 RCT: RR 1.11, 95% CI 1.04–1.18). One CS on patients using a combination wireless pill box/SMS reminder system found higher rates of cure (RR 2.32, 95% CI 1.60–3.36) and sputum conversion at two months (RR 1.62, 95% CI 1.09–2.42) compared to those without this system. Other studies found no difference in rates of mortality, treatment success, failure, or loss to follow-up (Figs 23–25 and S68–S74 Figs).
Fig 23.
(A) Impact of digital technologies on TB treatment outcomes. (B) Impact of combining different types of adherence interventions on TB treatment outcomes. * = significant heterogeneity in the meta-analysis, as determined by I2 statistic. 1 = defined as proportion of patients taking >90% of pills. Conversion = sputum conversion at the end of two months. N = number of studies included within the meta-analysis. Poor adherence = percentage of patient-months in which >20% of doses were missed. CS, cohort study; DOT, directly observed therapy; LTFU, loss to follow-up; RCT, randomized controlled trial; RR, risk ratio; TB, tuberculosis; VOT, video-observed therapy.
Fig 25. Rates of sputum conversion at two months in patients receiving phone reminders in addition to standard care versus standard care alone.
M-H, Mantel-Haenszel; RCT, randomized controlled trial.
Fig 24. Meta-analysis of treatment success rates in patients receiving phone reminders in addition to standard care versus standard care alone.
M-H, Mantel-Haenszel; RCT, randomized controlled trial.
Electronic medication monitor boxes were associated with lower rates of loss to follow-up (1 CS: RR 0.59, 95% CI 0.43–0.80), poor outcome (1 CS: RR 0.63, 95% CI 0.47–0.83), and poor adherence (1 CS: RR 0.57, 95% CI 0.53–0.61). Phone reminders and medication monitor boxes combined were associated with lower rates of poor adherence (1 CS: RR 0.56, 95% CI 0.52–0.60). Compared with DOT, VOT rates of treatment completion (2 CSs: RR 1.17, 95% CI 0.79–1.72) and mortality (1 CS: RR 1.80, 95% CI 0.19–17) were not significantly different [135,136].
Mixed interventions
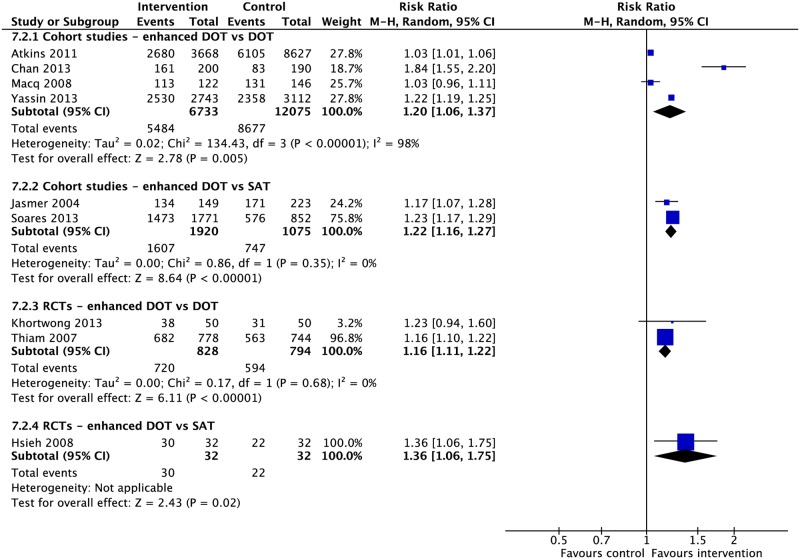
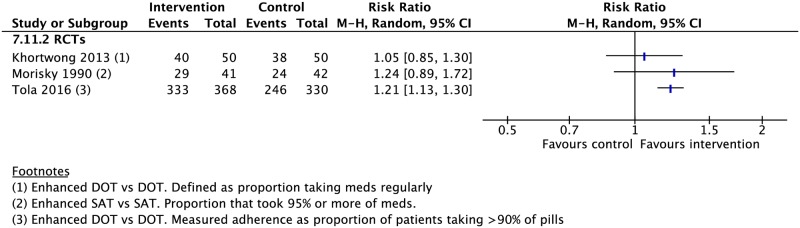
Seven RCTs and eleven CSs combined multiple adherence interventions from the aforementioned categories to DOT or SAT [10,59,137–152]. Patient-centered DOT (enhanced DOT) was associated with lower rates of loss to follow-up (4 RCTs: RR 0.45, 95% CI 0.32–0.65) and higher rates of treatment success (3 CSs: RR 1.27, 95% CI 1.09–1.49; 2 RCTs: RR 1.16, 95% CI 1.11–1.22) and cure (3 RCTs: RR 1.19, 95% CI 1.13–1.25) when compared with DOT alone (Figs 23, 26, 27 and S75–S82 Figs). When compared with SAT, patient-centered DOT was associated with higher rates of treatment success (2 CSs: RR 1.22, 95% CI 1.16–1.27; 1 RCT: RR 1.36, 95% CI 1.06–1.75), treatment completion (2 CSs: RR 1.84, 95% CI 1.52–2.21; 1 RCT: RR 1.41, 95% CI 1.11–1.79), cure (2 CSs: RR 1.42, 95% CI 1.02–1.99; 1 RCT: RR 1.36, 95% CI 1.06–1.75), and sputum conversion at two months (1 RCT: RR 1.65, 95% CI 1.16–2.34). We did not find any studies that compared patient-centered DOT to patient-centered SAT.
Fig 26. Meta-analysis of treatment success rates in patients receiving combination adherence interventions (enhanced DOT) in addition to standard care versus standard care alone.
DOT, directly observed therapy; M-H, Mantel-Haenszel; RCT, randomized controlled trial; SAT, self-administered therapy.
Fig 27. Adherence rates in patients receiving combination adherence interventions (enhanced DOT or enhanced SAT) in addition to standard care versus standard care alone.
DOT, directly observed therapy; M-H, Mantel-Haenszel; RCT, randomized controlled trial; SAT, self-administered therapy.
Discussion
To our knowledge, this is the first comprehensive systematic review of prospective and retrospective CSs as well as RCTs studying any interventions used to increase adherence to treatment of either drug-susceptible or -resistant TB. The studies included represent high- and low-resource settings and therefore are broadly representative across a variety of TB program settings. Our meta-analysis identified several adherence interventions that consistently showed associations with improved adherence and TB treatment outcomes. Of note, DOT was associated with better treatment outcomes compared to SAT, especially for TB patients living with HIV. The addition of other adherence interventions to DOT, such as education (for staff or patients), material or psychological support, or reminder systems (including SMS technology and phone reminders), correlated with reduced rates of mortality and loss to follow-up and higher rates of treatment success and cure. Combining other types of adherence interventions with DOT or SAT was associated with improved outcomes compared to either treatment modality alone. Lastly, VOT had no evidence of a difference in rates of treatment completion and mortality when compared to DOT.
Our conclusions differ from prior reviews. Karumbi and colleagues focused on data from RCTs and concluded that DOT did not improve TB treatment completion and cure when compared to self-administered therapy [153]. When our meta-analysis was limited to datasets from RCTs, we found similar results on these outcomes. Whereas RCTs are a superior study design because of their strong internal validity, they can lack external validity [154–156]. For example, RCTs provide rigorous oversight of participants that does not correspond to routine practice in program settings. Furthermore, without allocation concealment, alterations of behavior because of the awareness of being observed can further hamper the ability of RCTs to assess the value of adherence interventions. Another review by Pasipanodya and colleagues included RCTs as well as prospective CSs and concluded that DOT was not significantly better than SAT in preventing microbiologic failure, relapse, or development of drug resistance [157]. Our review found similar results with respect to these particular outcomes of interest. However, because our inclusion criteria were broader than these three outcomes, we included a larger number of CSs and RCTs in our review, resulting in a larger dataset for analysis.
Important findings in our analysis are that a variety of adherence interventions are effective in improving TB outcomes, certain subpopulations are more likely to benefit from particular types of interventions (i.e., DOT for TB patients with HIV), and certain modalities of DOT are more effective than others. Furthermore, treatment supervision alone in the form of DOT is not likely to guarantee improved TB treatment outcomes in all TB patients across all settings. As such, a patient-centered care approach to TB adherence using a package of interventions tailored to a patient’s needs and values is more likely to improve TB outcomes. It must be noted that most of the literature on adherence interventions included in our meta-analysis did not evaluate the impact of adherence interventions on relapse and development of drug resistance, which are critical outcomes of interest for population-level TB management [2]. Only three CSs compared the impact of DOT on the development of drug resistance compared with SAT, one of which found no significant difference [4,59,70]. One CS found reminders correlated with reduced rates of drug resistance [113], and another CS on incentives found no benefit [109]. With regard to relapse rates, one RCT [158] and four CSs [40,54,59,67] found no significant differences between DOT and SAT, while two CSs saw a decline in relapse rates with DOT [4,38]. Lastly, relapse rates were similar between patient-centered DOT and SAT in one CS [59]. Given the paucity of data on such critical outcomes, the effectiveness of adherence interventions based on our analyses should be assessed cautiously.
DOT has several limitations, including the cost imposed on patients and the health system [33–35,49,159,160]. Furthermore, WHO’s guidance on TB ethics argues that DOT is only ethically justifiable in the context of a patient-centered approach and provides an ethical framework for its implementation [161,162]. It must be noted that many of the adherence interventions in these studies involve significant resource utilization, which makes it necessary to mobilize the resources needed to facilitate their implementation. Comparing the cost-effectiveness of different adherence interventions would be crucial to strengthen and expand patient-centered approaches for adherence to TB treatment. Data from studies on digital technology and VOT are promising and have the potential to reduce costs to the patient and the health system [163]. The key challenges to address in implementing SMS and VOT adherence interventions are ensuring access to smartphones, coverage of data transmission costs, data encryption, and patient privacy.
Our meta-analysis has limitations. First, most of the studies on DOT in TB patients living with HIV are from the pre-ART era or were conducted in patients at highest risk of loss to follow-up. Contemporary, more integrated approaches to HIV/TB care were not assessed in this review. Second, the studies in our review were heterogeneous in their methodology. It is difficult to standardize methodology in these types of studies, given the numerous disparate interventions possible, evolving technologies, and the complexities of behavior change. We grouped similar interventions within categories of adherence interventions to draw on their similarities for practice recommendations. As the way in which such interventions are implemented largely determines their success, inter-study variability impacts our meta-analytic findings. Third, our literature review was restricted to English language articles in Medline. The absence of potentially relevant studies published in non-English language journals or via other electronic databases is a limitation. However, we reviewed the references of 32 systematic reviews on this topic with search strategies that spanned European (Embase), Latin American (LILACS), Cochrane Central Register of Controlled Trials (CENTRAL), and metaRegister of Controlled Trials (mRCT) databases. Additionally, we consulted experts in studies of adherence interventions to ascertain that all relevant studies were included. Lastly, the results from meta-analyses of CSs should be interpreted with caution given uncontrolled confounding inherent to such studies, as noted by our quality assessment.
In this review, we sought to evaluate the impact of adherence interventions in the treatment of drug-resistant TB, and whereas some of the studies included in our review had cohorts of patients with drug resistance, no studies focused only on MDR-TB met our inclusion criteria. A systematic review in 2009 that was limited to case series without an internal or historic control group found that rates of treatment success were higher in patients with MDR-TB who received DOT throughout treatment than those who did not [164]. Another meta-analysis also limited to case series found lower rates of loss to follow-up with DOT delivery at home, use of community health workers for DOT (as opposed to nurses or healthcare providers), a standardized treatment regimen, provision of DOT throughout treatment, and patient education [165]. Given the increased length and duration of treatment necessary for patients with MDR-TB, more studies on adherence interventions tailored to this population are needed.
Conclusion
We have found that TB treatment outcomes improve with the use of adherence interventions such as patient education and counseling, material support, psychological support interventions, reminders and tracers, and digital health technologies. DOT provided by trained health workers in the community is associated with better treatment outcomes than DOT provided by family members or untrained lay workers. DOT provided in the community is associated with better treatment outcomes than clinic-based DOT. TB patients living with HIV have significantly better outcomes when treated with DOT as opposed to SAT. VOT may be an appropriate alternative to in-person DOT if the resources for its use are available. More importantly, a patient-centered approach to TB treatment using a package of adherence interventions tailored to patients’ needs and values leads to improved TB treatment outcomes. The optimal package of adherence interventions to implement may vary by setting, resources, and the local epidemiology of TB (e.g., prevalence of comorbidities, including HIV coinfection), among other factors. The WHO TB treatment guidelines update, for which this review was conducted, provides additional information on selecting patient-centered approaches for enhancing adherence [166]. Based on our review, studies on adherence interventions in patients with MDR-TB as well as cost-effectiveness analyses will be helpful in identifying the optimal interventions to implement in various settings.
Supporting information
(DOC)
(DOC)
TB, tuberculosis.
(DOCX)
SMS, short message service; TB, tuberculosis.
(DOCX)
1 = Newcastle-Ottawa Score provided for cohort studies. Quality of RCTs are presented separately. 2 = Study includes >50% HIV/TB patients. 3 = Study includes >50% MDR-TB patients. RCT, randomized controlled trial; TB, tuberculosis.
(DOCX)
(TIF)
(TIF)
DOT, directly observed therapy; SAT, self-administered therapy.
(TIF)
“Not estimable” denotes a subgroup within a study not included in the meta-analysis. DOT, directly observed therapy; SAT, self-administered therapy.
(TIF)
DOT, directly observed therapy; SAT, self-administered therapy.
(TIF)
“Not estimable” denotes a subgroup within a study not included in the meta-analysis. DOT, directly observed therapy; SAT, self-administered therapy.
(TIF)
DOT, directly observed therapy; SAT, self-administered therapy.
(TIF)
“Not estimable” denotes a subgroup within a study not included in the meta-analysis. DOT, directly observed therapy; SAT, self-administered therapy.
(TIF)
DOT, directly observed therapy; SAT, self-administered therapy.
(TIF)
“Not estimable” denotes a subgroup within a study not included in the meta-analysis. DOT, directly observed therapy; SAT, self-administered therapy.
(TIF)
DOT, directly observed therapy; SAT, self-administered therapy.
(TIF)
“Not estimable” denotes a subgroup within a study not included in the meta-analysis. DOT, directly observed therapy; SAT, self-administered therapy.
(TIF)
DOT, directly observed therapy; SAT, self-administered therapy.
(TIF)
DOT, directly observed therapy; SAT, self-administered therapy.
(TIF)
DOT, directly observed therapy; SAT, self-administered therapy; TB, tuberculosis.
(TIF)
DOT, directly observed therapy; SAT, self-administered therapy; TB, tuberculosis.
(TIF)
DOT, directly observed therapy; SAT, self-administered therapy; TB, tuberculosis.
(TIF)
DOT, directly observed therapy; SAT, self-administered therapy; TB, tuberculosis.
(TIF)
DOT, directly observed therapy; SAT, self-administered therapy; TB, tuberculosis.
(TIF)
DOT, directly observed therapy; SAT, self-administered therapy; TB, tuberculosis.
(TIF)
DOT, directly observed therapy; MDR-TB, multidrug-resistant tuberculosis; SAT, self-administered therapy.
(TIF)
DOT, directly observed therapy; MDR-TB, multidrug-resistant tuberculosis; SAT, self-administered therapy.
(TIF)
DOT, directly observed therapy; MDR-TB, multidrug-resistant tuberculosis; SAT, self-administered therapy.
(TIF)
DOT, directly observed therapy; MDR-TB, multidrug-resistant tuberculosis; SAT, self-administered therapy.
(TIF)
DOT, directly observed therapy.
(TIF)
DOT, directly observed therapy.
(TIF)
DOT, directly observed therapy.
(TIF)
DOT, directly observed therapy.
(TIF)
DOT, directly observed therapy.
(TIF)
DOT, directly observed therapy.
(TIF)
DOT, directly observed therapy.
(TIF)
DOT, directly observed therapy.
(TIF)
DOT, directly observed therapy.
(TIF)
DOT, directly observed therapy.
(TIF)
DOT, directly observed therapy.
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(A) Meta-analysis of mortality rates in patients receiving reminders/tracers in addition to standard care versus standard care alone. (B) Sensitivity analysis: removing the heaviest weighted study (Bronner 2012) in which control and intervention cohorts had significantly different pre-intervention mortality rates. “Not estimable” denotes a subgroup within a study not included in the meta-analysis.
(TIF)
(TIF)
(A) Meta-analysis of cure rates in patients receiving reminders/tracers in addition to standard care versus standard care alone. (B) Sensitivity analysis: removing the heaviest weighted study (Bronner 2012) in which control and intervention cohorts had significantly different pre-intervention cure rates.
(TIF)
(A) Meta-analysis of treatment failure rates in patients receiving reminders/tracers in addition to standard care versus standard care alone—cohort studies. (B) Sensitivity analysis: removing the heaviest weighted study (Bronner 2012) in which control and intervention cohorts had significantly different pre-intervention treatment failure rates.
(TIF)
RCT, randomized controlled trial.
(TIF)
(A) Meta-analysis of rates of loss to follow-up in patients receiving reminders/tracers in addition to standard care versus standard care alone. (B) Sensitivity analysis: removing the heaviest weighted study (Bronner 2012) in which control and intervention cohorts had significantly different pre-intervention loss to follow-up rates.
(TIF)
(TIF)
Poor adherence is defined as the percentage of patient-months in which at least 20% of doses were missed.
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
RCT, randomized controlled trial.
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
SMS, short message service; VOT, video-observed therapy.
(TIF)
SMS, short message service; VOT, video-observed therapy.
(TIF)
RCT, randomized controlled trial; SMS, short message service.
(TIF)
SMS, short message service.
(TIF)
SMS, short message service.
(TIF)
SMS, short message service.
(TIF)
Poor adherence is defined as the percentage of patient-months in which at least 20% of doses were missed. SMS, short message service.
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
Acknowledgments
The authors alone are responsible for the views expressed in this publication, and these views do not necessarily represent the decisions or policies of WHO. The designations used and the presentation of the material in this publication do not imply the expression of any opinion whatsoever on the part of WHO concerning the legal status of any country, territory, city or area, or of its authorities, nor concerning the delimitation of its frontiers or boundaries.
Abbreviations
- AFB
acid-fast bacilli
- CS
cohort study
- DOT
directly observed therapy
- GRADE
Grading of Recommendations Assessment, Development, and Evaluation
- HCW
healthcare worker
- INH
isoniazid
- M-H
Mantel-Haenszel
- PICO
population, intervention, comparison, outcome
- PRISMA
Preferred Reporting Items for Systematic Reviews and Meta-Analysis
- RCT
randomized controlled trial
- RD
risk difference
- RR
risk ratio
- SAT
self-administered therapy
- SMS
short message service
- TB
tuberculosis
- VOT
video-observed therapy
- WHO
World Health Organization
Data Availability
All relevant data are within the paper and its Supporting information files.
Funding Statement
The project was funded by the World Health Organization (http://www.who.int/en/) to conduct the evidence reviews required for the revision of the World Health Organization TB Treatment Guidelines (PN). The funders specified the PICO for this study, which was addressed in accordance with GRADE. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Hirpa S, Medhin G, Girma B, Melese M, Mekonen A, Suarez P, et al. Determinants of multidrug-resistant tuberculosis in patients who underwent first-line treatment in Addis Ababa: a case control study. BMC Public Health. 2013;13:782 Epub 2013/08/29. doi: 10.1186/1471-2458-13-782 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Moonan PK, Quitugua TN, Pogoda JM, Woo G, Drewyer G, Sahbazian B, et al. Does directly observed therapy (DOT) reduce drug resistant tuberculosis? BMC Public Health. 2011;11:19 Epub 2011/01/11. doi: 10.1186/1471-2458-11-19 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Munro SA, Lewin SA, Smith HJ, Engel ME, Fretheim A, Volmink J. Patient adherence to tuberculosis treatment: a systematic review of qualitative research. PLoS Med. 2007;4(7):e238 doi: 10.1371/journal.pmed.0040238 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Weis SE, Slocum PC, Blais FX, King B, Nunn M, Matney GB, et al. The effect of directly observed therapy on the rates of drug resistance and relapse in tuberculosis. N Engl J Med. 1994;330(17):1179–84. Epub 1994/04/28. doi: 10.1056/NEJM199404283301702 . [DOI] [PubMed] [Google Scholar]
- 5.Ormerod LP, Prescott RJ. Inter-relations between relapses, drug regimens and compliance with treatment in tuberculosis. Respir Med. 1991;85(3):239–42. Epub 1991/05/01. . [DOI] [PubMed] [Google Scholar]
- 6.Mitchison DA. How drug resistance emerges as a result of poor compliance during short course chemotherapy for tuberculosis. Int J Tuberc Lung Dis. 1998;2(1):10–5. Epub 1998/04/30. . [PubMed] [Google Scholar]
- 7.Clark PM, Karagoz T, Apikoglu-Rabus S, Izzettin FV. Effect of pharmacist-led patient education on adherence to tuberculosis treatment. Am J Health Syst Pharm. 2007;64(5):497–505. Epub 2007/02/27. doi: 10.2146/ajhp050543 . [DOI] [PubMed] [Google Scholar]
- 8.Janmeja AK D S.K.; Bhargava R.; Chavan B.S. Psychotherapy improves compliance with tuberculosis treatment. Respiration. 2005;72:375–80. doi: 10.1159/000086251 [DOI] [PubMed] [Google Scholar]
- 9.Liefooghe R, Suetens C, Meulemans H, Moran MB, De Muynck A. A randomised trial of the impact of counselling on treatment adherence of tuberculosis patients in Sialkot, Pakistan. Int J Tuberc Lung Dis. 1999;3(12):1073–80. Epub 1999/12/22. . [PubMed] [Google Scholar]
- 10.Baral SC, Aryal Y, Bhattrai R, King R, Newell JN. The importance of providing counselling and financial support to patients receiving treatment for multi-drug resistant TB: mixed method qualitative and pilot intervention studies. BMC Public Health. 2014;14:46 Epub 2014/01/21. doi: 10.1186/1471-2458-14-46 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Dick J, Lombard C. Shared vision—a health education project designed to enhance adherence to anti-tuberculosis treatment. Int J Tuberc Lung Dis. 1997;1(2):181–6. Epub 1997/04/01. . [PubMed] [Google Scholar]
- 12.Bayer R, Wilkinson D. Directly observed therapy for tuberculosis: history of an idea. Lancet. 1995;345(8964):1545–8. Epub 1995/06/17. . [DOI] [PubMed] [Google Scholar]
- 13.Mirsaeidi M, Farshidpour M, Banks-Tripp D, Hashmi S, Kujoth C, Schraufnagel D. Video directly observed therapy for treatment of tuberculosis is patient-oriented and cost-effective. Eur Respir J. 2015;46(3):871–4. Epub 2015/03/21. doi: 10.1183/09031936.00011015 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sinkou H, Hurevich H, Rusovich V, Zhylevich L, Falzon D, de Colombani P, et al. Video-observed treatment for tuberculosis patients in Belarus: findings from the first programmatic experience. Eur Respir J. 2017;49(3). Epub 2017/03/24. doi: 10.1183/13993003.02049-2016 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lutge EE, Wiysonge CS, Knight SE, Sinclair D, Volmink J. Incentives and enablers to improve adherence in tuberculosis. Cochrane Database Syst Rev. 2015;(9):CD007952 Epub 2015/09/04. doi: 10.1002/14651858.CD007952.pub3 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.M'Imunya J M, Kredo T, Volmink J. Patient education and counselling for promoting adherence to treatment for tuberculosis. Cochrane Database Syst Rev. 2012;(5):CD006591 Epub 2012/05/18. doi: 10.1002/14651858.CD006591.pub2 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Liu Q, Abba K, Alejandria MM, Sinclair D, Balanag VM, Lansang MA. Reminder systems to improve patient adherence to tuberculosis clinic appointments for diagnosis and treatment. Cochrane Database Syst Rev. 2014;(11):CD006594 Epub 2014/11/19. doi: 10.1002/14651858.CD006594.pub3 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Denkinger CM, Grenier J, Stratis AK, Akkihal A, Pant-Pai N, Pai M. Mobile health to improve tuberculosis care and control: a call worth making. Int J Tuberc Lung Dis. 2013;17(6):719–27. Epub 2013/04/02. doi: 10.5588/ijtld.12.0638 . [DOI] [PubMed] [Google Scholar]
- 19.Free C, Phillips G, Galli L, Watson L, Felix L, Edwards P, et al. The effectiveness of mobile-health technology-based health behaviour change or disease management interventions for health care consumers: a systematic review. PLoS Med. 2013;10(1):e1001362 doi: 10.1371/journal.pmed.1001362 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Global strategy and targets for tuberculosis prevention, care and control after 2015 Geneva: World Health Organization; 2014. [cited 2017 Sept 23]. http://apps.who.int/gb/ebwha/pdf_files/EB134/B134_R4-en.pdf?ua=1. [Google Scholar]
- 21.Guyatt GH, Oxman AD, Vist GE, Kunz R, Falck-Ytter Y, Alonso-Coello P, et al. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ. 2008;336(7650):924–6. Epub 2008/04/26. doi: 10.1136/bmj.39489.470347.AD . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.WHO handbook for guideline development, 2nd edition. 2014 [cited 2017 Sept 23]. http://www.who.int/publications/guidelines/handbook_2nd_ed.pdf.
- 23.Guidelines for treatment of drug-susceptible tuberculosis and patient care 2017 [cited 2017 Sept 23]. http://www.who.int/tb/publications/2017/dstb_guidance_2017/en/.
- 24.Alipanah N, Jarlsberg L, Miller C, Lechner A, Wai K, Nahid P. Report for Systematic Review for Adherence Interventions in TB Treatment. World Health Organization guidelines for treatment of drug-susceptible tuberculosis and patient care. 2017;Annex 5.
- 25.Definitions and reporting framework for tuberculosis: World Health Organization; 2014. [cited 2017 Sept 23]. http://apps.who.int/iris/bitstream/10665/79199/1/9789241505345_eng.pdf. [Google Scholar]
- 26.Cochrane Risk of Bias Tool [cited 2017 Sept 23]. http://handbook.cochrane.org/.
- 27.Wells GA S B.; O’Connell D.O.; Peterson J.; Welch V.; Losos M.; Tugwell P. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. 2014. [Google Scholar]
- 28.Review Manager (RevMan). 5.3 ed Copenhagen: The Nordic Cochrane Centre: The Cochrane Collaboration; 2014. [Google Scholar]
- 29.GraphPad Prism. GraphPad Software Inc.
- 30.Kamolratanakul P, Sawert H, Lertmaharit S, Kasetjaroen Y, Akksilp S, Tulaporn C, et al. Randomized controlled trial of directly observed treatment (DOT) for patients with pulmonary tuberculosis in Thailand. Trans R Soc Trop Med Hyg. 1999;93(5):552–7. Epub 2000/03/04. . [DOI] [PubMed] [Google Scholar]
- 31.MacIntyre CR, Goebel K, Brown GV, Skull S, Starr M, Fullinfaw RO. A randomised controlled clinical trial of the efficacy of family-based direct observation of anti-tuberculosis treatment in an urban, developed-country setting. Int J Tuberc Lung Dis. 2003;7(9):848–54. Epub 2003/09/16. . [PubMed] [Google Scholar]
- 32.Chennai T. A controlled clinical trial of oral short-course regimens in the treatment of sputum-positive pulmonary tuberculosis. Tuberculosis Research Centre. Int J Tuberc Lung Dis. 1997;1(6):509–17. Epub 1998/03/06. . [PubMed] [Google Scholar]
- 33.Walley JD, Khan MA, Newell JN, Khan MH. Effectiveness of the direct observation component of DOTS for tuberculosis: a randomised controlled trial in Pakistan. Lancet. 2001;357(9257):664–9. Epub 2001/03/15. doi: 10.1016/S0140-6736(00)04129-5 . [DOI] [PubMed] [Google Scholar]
- 34.Zwarenstein M, Schoeman JH, Vundule C, Lombard CJ, Tatley M. Randomised controlled trial of self-supervised and directly observed treatment of tuberculosis. Lancet. 1998;352(9137):1340–3. Epub 1998/11/05. doi: 10.1016/S0140-6736(98)04022-7 . [DOI] [PubMed] [Google Scholar]
- 35.Zwarenstein M, Schoeman JH, Vundule C, Lombard CJ, Tatley M. A randomised controlled trial of lay health workers as direct observers for treatment of tuberculosis. Int J Tuberc Lung Dis. 2000;4(6):550–4. Epub 2000/06/23. . [PubMed] [Google Scholar]
- 36.Tandon M, Gupta M, Tandon S, Gupta KB. DOTS versus self administered therapy (SAT) for patients of pulmonary tuberculosis: a randomised trial at a tertiary care hospital. Indian J Med Sci. 2002;56(1):19–21. Epub 2003/01/02. . [PubMed] [Google Scholar]
- 37.Akkslip S, Rasmithat S, Maher D, Sawert H. Direct observation of tuberculosis treatment by supervised family members in Yasothorn Province, Thailand. Int J Tuberc Lung Dis. 1999;3(12):1061–5. Epub 1999/12/22. . [PubMed] [Google Scholar]
- 38.Balasubramanian VN, Oommen K, Samuel R. DOT or not? Direct observation of anti-tuberculosis treatment and patient outcomes, Kerala State, India. Int J Tuberc Lung Dis. 2000;4(5):409–13. Epub 2000/05/18. . [PubMed] [Google Scholar]
- 39.Mathema B, Pande SB, Jochem K, Houston RA, Smith I, Bam DS, et al. Tuberculosis treatment in nepal: a rapid assessment of government centers using different types of patient supervision. Int J Tuberc Lung Dis. 2001;5(10):912–9. Epub 2001/10/19. . [PubMed] [Google Scholar]
- 40.Ormerod LP, Horsfield N, Green RM. Tuberculosis treatment outcome monitoring: Blackburn 1988–2000. Int J Tuberc Lung Dis. 2002;6(8):662–5. Epub 2002/08/02. . [PubMed] [Google Scholar]
- 41.Tsuchida K, Koyanagi H. Outcome of directly observed therapy for tuberculosis in Yokohama City, Japan. Int J Tuberc Lung Dis. 2003;7(8):730–4. Epub 2003/08/19. . [PubMed] [Google Scholar]
- 42.Nirupa C S G.; Santha T.; Ponnuraja C.; Fathima R.; Chandrasekharan V.; Jaggarajamma K.; Thomas A.; Gopi P.G.; Narayanan P.R. Evaluation of directly observed therapy treatment providers in the revised national tuberculosis control programme. Indian J Tuberc. 2005;52:73–7. [Google Scholar]
- 43.Daniel OJ. Pre- and post-directly observed treatment era in the management of TB: a teachiing hospital experience. Trop Doct. 2006;36(3):163–5. Epub 2006/08/04. doi: 10.1258/004947506777978280 . [DOI] [PubMed] [Google Scholar]
- 44.Okanurak K, Kitayaporn D, Wanarangsikul W, Koompong C. Effectiveness of DOT for tuberculosis treatment outcomes: a prospective cohort study in Bangkok, Thailand. Int J Tuberc Lung Dis. 2007;11(7):762–8. Epub 2007/07/05. . [PubMed] [Google Scholar]
- 45.Abassi A, Mansourian AR. Efficacy of DOTS strategy in treatment of respiratory tuberculosis in Gorgan, Islamic Republic of Iran. East Mediterr Health J. 2007;13(3):664–9. Epub 2007/08/11. . [PubMed] [Google Scholar]
- 46.Siemion-Szczesniak I K J. Treatment outcomes in culture-positive pulmonary tuberculosis. Pneumonol Alergol Pol. 2009;77:11–22. [PubMed] [Google Scholar]
- 47.Cayla JA, Rodrigo T, Ruiz-Manzano J, Caminero JA, Vidal R, Garcia JM, et al. Tuberculosis treatment adherence and fatality in Spain. Respir Res. 2009;10:121 Epub 2009/12/03. doi: 10.1186/1465-9921-10-121 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Zvavamwe Z, Ehlers VJ. Experiences of a community-based tuberculosis treatment programme in Namibia: a comparative cohort study. Int J Nurs Stud. 2009;46(3):302–9. Epub 2008/11/11. doi: 10.1016/j.ijnurstu.2008.09.013 . [DOI] [PubMed] [Google Scholar]
- 49.Xu W, Lu W, Zhou Y, Zhu L, Shen H, Wang J. Adherence to anti-tuberculosis treatment among pulmonary tuberculosis patients: a qualitative and quantitative study. BMC Health Serv Res. 2009;9:169 Epub 2009/09/22. doi: 10.1186/1472-6963-9-169 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Abuaku B T H.; Li X.; Chen M.; Huang X. Treatment default and death among tuberculosis patients in Hunan, China. Scandinavian Journal of Infectious Diseases. 2010;42:281–7. doi: 10.3109/00365540903493723 [DOI] [PubMed] [Google Scholar]
- 51.Alwood K, Keruly J, Moore-Rice K, Stanton DL, Chaulk CP, Chaisson RE. Effectiveness of supervised, intermittent therapy for tuberculosis in HIV-infected patients. AIDS. 1994;8(8):1103–8. Epub 1994/08/01. . [DOI] [PubMed] [Google Scholar]
- 52.Das M, Isaakidis P, Armstrong E, Gundipudi NR, Babu RB, Qureshi IA, et al. Directly-observed and self-administered tuberculosis treatment in a chronic, low-intensity conflict setting in India. PLoS ONE. 2014;9(3):e92131 doi: 10.1371/journal.pone.0092131 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Alvarez-Uria G, Pakam R, Midde M, Naik PK. Incidence and mortality of tuberculosis before and after initiation of antiretroviral therapy: an HIV cohort study in India. J Int AIDS Soc. 2014;17:19251 Epub 2014/12/17. doi: 10.7448/IAS.17.1.19251 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Juan G, Lloret T, Perez C, Lopez P, Navarro R, Ramon M, et al. Directly observed treatment for tuberculosis in pharmacies compared with self-administered therapy in Spain. Int J Tuberc Lung Dis. 2006;10(2):215–21. Epub 2006/02/28. . [PubMed] [Google Scholar]
- 55.Ershova JV, Podewils LJ, Bronner LE, Stockwell HG, Dlamini SS, Mametja LD. Evaluation of adherence to national treatment guidelines among tuberculosis patients in three provinces of South Africa. S Afr Med J. 2014;104(5):362–8. Epub 2014/09/13. doi: 10.7196/samj.7655 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Bashar M, Alcabes P, Rom WN, Condos R. Increased incidence of multidrug-resistant tuberculosis in diabetic patients on the Bellevue Chest Service, 1987 to 1997. Chest. 2001;120(5):1514–9. Epub 2001/11/20. . [DOI] [PubMed] [Google Scholar]
- 57.Olle-Goig JE, Alvarez J. Treatment of tuberculosis in a rural area of Haiti: directly observed and non-observed regimens. The experience of H pital Albert Schweitzer. Int J Tuberc Lung Dis. 2001;5(2):137–41. Epub 2001/03/22. . [PubMed] [Google Scholar]
- 58.Pungrassami P, Johnsen SP, Chongsuvivatwong V, Olsen J. Has directly observed treatment improved outcomes for patients with tuberculosis in southern Thailand? Trop Med Int Health. 2002;7(3):271–9. Epub 2002/03/21. . [DOI] [PubMed] [Google Scholar]
- 59.Jasmer RM, Seaman CB, Gonzalez LC, Kawamura LM, Osmond DH, Daley CL. Tuberculosis treatment outcomes: directly observed therapy compared with self-administered therapy. Am J Respir Crit Care Med. 2004;170(5):561–6. Epub 2004/06/09. doi: 10.1164/rccm.200401-095OC . [DOI] [PubMed] [Google Scholar]
- 60.Cayla JA, Caminero JA, Rey R, Lara N, Valles X, Galdos-Tanguis H, et al. Current status of treatment completion and fatality among tuberculosis patients in Spain. Int J Tuberc Lung Dis. 2004;8(4):458–64. Epub 2004/05/15. . [PubMed] [Google Scholar]
- 61.Cavalcante SC, Soares EC, Pacheco AG, Chaisson RE, Durovni B, Team DE. Community DOT for tuberculosis in a Brazilian favela: comparison with a clinic model. Int J Tuberc Lung Dis. 2007;11(5):544–9. Epub 2007/04/19. . [PubMed] [Google Scholar]
- 62.Radilla-Chavez P, Laniado-Laborin R. Results of directly observed treatment for tuberculosis in Ensenada, Mexico: not all DOTS programs are created equally. Int J Tuberc Lung Dis. 2007;11(3):289–92. Epub 2007/03/14. . [PubMed] [Google Scholar]
- 63.Anuwatnonthakate A, Limsomboon P, Nateniyom S, Wattanaamornkiat W, Komsakorn S, Moolphate S, et al. Directly observed therapy and improved tuberculosis treatment outcomes in Thailand. PLoS ONE. 2008;3(8):e3089 doi: 10.1371/journal.pone.0003089 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Kapella BK, Anuwatnonthakate A, Komsakorn S, Moolphate S, Charusuntonsri P, Limsomboon P, et al. Directly observed treatment is associated with reduced default among foreign tuberculosis patients in Thailand. Int J Tuberc Lung Dis. 2009;13(2):232–7. Epub 2009/01/17. . [PubMed] [Google Scholar]
- 65.Vieira AA, Ribeiro SA. Compliance with tuberculosis treatment after the implementation of the directly observed treatment, short-course strategy in the city of Carapicuiba, Brazil. J Bras Pneumol. 2011;37(2):223–31. Epub 2011/05/04. . [DOI] [PubMed] [Google Scholar]
- 66.Ong'ang'o JR, Mwachari C, Kipruto H, Karanja S. The effects on tuberculosis treatment adherence from utilising community health workers: a comparison of selected rural and urban settings in Kenya. PLoS ONE. 2014;9(2):e88937 doi: 10.1371/journal.pone.0088937 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Mac JT, Doordan A, Carr CA. Evaluation of the effectiveness of a directly observed therapy program with Vietnamese tuberculosis patients. Public Health Nurs. 1999;16(6):426–31. Epub 2000/01/05. . [DOI] [PubMed] [Google Scholar]
- 68.Chung WS, Chang YC, Yang MC. Factors influencing the successful treatment of infectious pulmonary tuberculosis. Int J Tuberc Lung Dis. 2007;11(1):59–64. Epub 2007/01/16. . [PubMed] [Google Scholar]
- 69.Yen YF, Yen MY, Lin YP, Shih HC, Li LH, Chou P, et al. Directly observed therapy reduces tuberculosis-specific mortality: a population-based follow-up study in Taipei, Taiwan. PLoS ONE. 2013;8(11):e79644 doi: 10.1371/journal.pone.0079644 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Chien JY, Lai CC, Tan CK, Chien ST, Yu CJ, Hsueh PR. Decline in rates of acquired multidrug-resistant tuberculosis after implementation of the directly observed therapy, short course (DOTS) and DOTS-Plus programmes in Taiwan. J Antimicrob Chemother. 2013;68(8):1910–6. Epub 2013/04/13. doi: 10.1093/jac/dkt103 . [DOI] [PubMed] [Google Scholar]
- 71.Yen YF, Feng JY, Pan SW, Chuang PH, Su VY, Su WJ. Determinants of mortality in elderly patients with tuberculosis: a population-based follow-up study. Epidemiol Infect. 2017;145(7):1374–81. Epub 2017/02/14. doi: 10.1017/S0950268817000152 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Mohr E, Daniels J, Beko B, Isaakidis P, Cox V, Steele SJ, et al. DOT or SAT for Rifampicin-resistant tuberculosis? A non-randomized comparison in a high HIV-prevalence setting. PLoS ONE. 2017;12(5):e0178054 doi: 10.1371/journal.pone.0178054 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Lei X, Huang K, Liu Q, Jie YF, Tang SL. Are tuberculosis patients adherent to prescribed treatments in China? Results of a prospective cohort study. Infect Dis Poverty. 2016;5:38 Epub 2016/05/07. doi: 10.1186/s40249-016-0134-9 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Snyder RE, Marlow MA, Phuphanich ME, Riley LW, Maciel EL. Risk factors for differential outcome following directly observed treatment (DOT) of slum and non-slum tuberculosis patients: a retrospective cohort study. BMC Infect Dis. 2016;16:494 Epub 2016/09/21. doi: 10.1186/s12879-016-1835-1 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Dave PV, Shah AN, Nimavat PB, Modi BB, Pujara KR, Patel P, et al. Direct Observation of Treatment Provided by a Family Member as Compared to Non-Family Member among Children with New Tuberculosis: A Pragmatic, Non-Inferiority, Cluster-Randomized Trial in Gujarat, India. PLoS ONE. 2016;11(2):e0148488 doi: 10.1371/journal.pone.0148488 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Kingkaew N S B.; Amnusiphon W.; Jongpalbulpatana J.; Anuwatnonthakate A. Effectiveness of and results from directly observed treatment of tuberculosis patients by health-care workers vs. family members, Vachira Phuket Hospital, 2005–2006. Journal of Health Systems Research. 2008;2(2). [Google Scholar]
- 77.Singh AA, Parasher D, Shekhavat GS, Sahu S, Wares DF, Granich R. Effectiveness of urban community volunteers in directly observed treatment of tuberculosis patients: a field report from Haryana, North India. Int J Tuberc Lung Dis. 2004;8(6):800–2. Epub 2004/06/09. . [PubMed] [Google Scholar]
- 78.Wilkinson D, Davies GR. Coping with Africa's increasing tuberculosis burden: are community supervisors an essential component of the DOT strategy? Directly observed therapy. Trop Med Int Health. 1997;2(7):700–4. Epub 1997/07/01. . [DOI] [PubMed] [Google Scholar]
- 79.Akhtar S, Rozi S, White F, Hasan R. Cohort analysis of directly observed treatment outcomes for tuberculosis patients in urban Pakistan. Int J Tuberc Lung Dis. 2011;15(1):90–6. Epub 2011/02/01. . [PubMed] [Google Scholar]
- 80.Arora VK S N.; Gupta R. Community mediated domiciliary DOTS execution—A study from New Delhi. Ind J Tub. 2003;50:143–50. [Google Scholar]
- 81.Banerjee A, Harries AD, Mphasa N, Nyirenda TE, Veen J, Ringdal T, et al. Evaluation of a unified treatment regimen for all new cases of tuberculosis using guardian-based supervision. Int J Tuberc Lung Dis. 2000;4(4):333–9. Epub 2000/04/25. . [PubMed] [Google Scholar]
- 82.Becx-Bleumink M W H.; Apriani W.; Vrakking H. High tuberculosis notification and treatment success rates through community participation in central Sulawesi, Republic of Indonesia. Int J Tuberc Lung Dis. 2001;5(10):920–5. [PubMed] [Google Scholar]
- 83.Dobler CC, Korver S, Batbayar O, Oyuntsetseg S, Tsolmon B, Wright C, et al. Success of community-based directly observed anti-tuberculosis treatment in Mongolia. Int J Tuberc Lung Dis. 2015;19(6):657–62. Epub 2015/05/07. doi: 10.5588/ijtld.14.0927 . [DOI] [PubMed] [Google Scholar]
- 84.Dudley L, Azevedo V, Grant R, Schoeman JH, Dikweni L, Maher D. Evaluation of community contribution to tuberculosis control in Cape Town, South Africa. Int J Tuberc Lung Dis. 2003;7(9 Suppl 1):S48–55. Epub 2003/09/16. . [PubMed] [Google Scholar]
- 85.Kironde S, Meintjies M. Tuberculosis treatment delivery in high burden settings: does patient choice of supervision matter? Int J Tuberc Lung Dis. 2002;6(7):599–608. Epub 2002/07/10. . [PubMed] [Google Scholar]
- 86.Maciel EL, Guidoni LM, Brioshi AP, do Prado TN, Fregona G, Hadad DJ, et al. Household members and health care workers as supervisors of tuberculosis treatment. Rev Saude Publica. 2010;44(2):339–43. Epub 2010/03/27. . [DOI] [PubMed] [Google Scholar]
- 87.Manders AJ, Banerjee A, van den Borne HW, Harries AD, Kok GJ, Salaniponi FM. Can guardians supervise TB treatment as well as health workers? A study on adherence during the intensive phase. Int J Tuberc Lung Dis. 2001;5(9):838–42. Epub 2001/09/28. . [PubMed] [Google Scholar]
- 88.Mhimbira F, Hella J, Maroa T, Kisandu S, Chiryamkubi M, Said K, et al. Home-Based and Facility-Based Directly Observed Therapy of Tuberculosis Treatment under Programmatic Conditions in Urban Tanzania. PLoS ONE. 2016;11(8):e0161171 doi: 10.1371/journal.pone.0161171 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Miti S, Mfungwe V, Reijer P, Maher D. Integration of tuberculosis treatment in a community-based home care programme for persons living with HIV/AIDS in Ndola, Zambia. Int J Tuberc Lung Dis. 2003;7(9 Suppl 1):S92–8. Epub 2003/09/16. . [PubMed] [Google Scholar]
- 90.Moalosi G, Floyd K, Phatshwane J, Moeti T, Binkin N, Kenyon T. Cost-effectiveness of home-based care versus hospital care for chronically ill tuberculosis patients, Francistown, Botswana. Int J Tuberc Lung Dis. 2003;7(9 Suppl 1):S80–5. Epub 2003/09/16. . [PubMed] [Google Scholar]
- 91.Niazi AD, Al-Delaimi AM. Impact of community participation on treatment outcomes and compliance of DOTS patients in Iraq. East Mediterr Health J. 2003;9(4):709–17. Epub 2005/03/08. . [PubMed] [Google Scholar]
- 92.Tripathy SK K P.; Sagili K.D.; Enarson A. Effectiveness of a community-based observation of anti-tuberculosis treatment in Bangalore City, India, 2010–2011. Public Health Action. 2013;3(3):230–4. doi: 10.5588/pha.13.0043 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.van den Boogaard J, Lyimo R, Irongo CF, Boeree MJ, Schaalma H, Aarnoutse RE, et al. Community vs. facility-based directly observed treatment for tuberculosis in Tanzania's Kilimanjaro Region. Int J Tuberc Lung Dis. 2009;13(12):1524–9. Epub 2009/11/19. . [PubMed] [Google Scholar]
- 94.Lwilla F, Schellenberg D, Masanja H, Acosta C, Galindo C, Aponte J, et al. Evaluation of efficacy of community-based vs. institutional-based direct observed short-course treatment for the control of tuberculosis in Kilombero district, Tanzania. Trop Med Int Health. 2003;8(3):204–10. Epub 2003/03/13. . [DOI] [PubMed] [Google Scholar]
- 95.Newell JN, Baral SC, Pande SB, Bam DS, Malla P. Family-member DOTS and community DOTS for tuberculosis control in Nepal: cluster-randomised controlled trial. Lancet. 2006;367(9514):903–9. Epub 2006/03/21. doi: 10.1016/S0140-6736(06)68380-3 . [DOI] [PubMed] [Google Scholar]
- 96.Wandwalo E, Kapalata N, Egwaga S, Morkve O. Effectiveness of community-based directly observed treatment for tuberculosis in an urban setting in Tanzania: a randomised controlled trial. Int J Tuberc Lung Dis. 2004;8(10):1248–54. Epub 2004/11/06. . [PubMed] [Google Scholar]
- 97.Wright J, Walley J, Philip A, Pushpananthan S, Dlamini E, Newell J, et al. Direct observation of treatment for tuberculosis: a randomized controlled trial of community health workers versus family members. Trop Med Int Health. 2004;9(5):559–65. Epub 2004/05/01. doi: 10.1111/j.1365-3156.2004.01230.x . [DOI] [PubMed] [Google Scholar]
- 98.Bock NN, Sales RM, Rogers T, DeVoe B. A spoonful of sugar …: improving adherence to tuberculosis treatment using financial incentives. Int J Tuberc Lung Dis. 2001;5(1):96–8. Epub 2001/03/27. . [PubMed] [Google Scholar]
- 99.Cantalice Filho JP. Food baskets given to tuberculosis patients at a primary health care clinic in the city of Duque de Caxias, Brazil: effect on treatment outcomes. J Bras Pneumol. 2009;35(10):992–7. Epub 2009/11/18. . [DOI] [PubMed] [Google Scholar]
- 100.Chua AP, Lim LK, Ng H, Chee CB, Wang YT. Outcome of a grocery voucher incentive scheme for low-income tuberculosis patients on directly observed therapy in Singapore. Singapore Med J. 2015;56(5):274–9. Epub 2015/03/20. doi: 10.11622/smedj.2015054 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Lu H, Yan F, Wang W, Wu L, Ma W, Chen J, et al. Do transportation subsidies and living allowances improve tuberculosis control outcomes among internal migrants in urban Shanghai, China? Western Pac Surveill Response J. 2013;4(1):19–24. Epub 2013/08/03. doi: 10.5365/WPSAR.2013.4.1.003 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Ngamvithayapong-Yanai J, Luangjina S, Nedsuwan S, Kantipong P, Wongyai J, Ishikawa N. Engaging women volunteers of high socioeconomic status in supporting socioeconomically disadvantaged tuberculosis patients in Chiang Rai, Thailand. Western Pac Surveill Response J. 2013;4(1):34–8. Epub 2013/08/03. doi: 10.5365/WPSAR.2012.3.4.013 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Sripad A, Castedo J, Danford N, Zaha R, Freile C. Effects of Ecuador's national monetary incentive program on adherence to treatment for drug-resistant tuberculosis. Int J Tuberc Lung Dis. 2014;18(1):44–8. Epub 2013/12/25. doi: 10.5588/ijtld.13.0253 . [DOI] [PubMed] [Google Scholar]
- 104.Torrens AW, Rasella D, Boccia D, Maciel EL, Nery JS, Olson ZD, et al. Effectiveness of a conditional cash transfer programme on TB cure rate: a retrospective cohort study in Brazil. Trans R Soc Trop Med Hyg. 2016;110(3):199–206. Epub 2016/02/18. doi: 10.1093/trstmh/trw011 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Tsai WC, Kung PT, Khan M, Campbell C, Yang WT, Lee TF, et al. Effects of pay-for-performance system on tuberculosis default cases control and treatment in Taiwan. J Infect. 2010;61(3):235–43. Epub 2010/07/14. doi: 10.1016/j.jinf.2010.06.016 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Wei X, Zou G, Yin J, Walley J, Yang H, Kliner M, et al. Providing financial incentives to rural-to-urban tuberculosis migrants in Shanghai: an intervention study. Infect Dis Poverty. 2012;1(1):9 Epub 2012/01/01. doi: 10.1186/2049-9957-1-9 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Zou G, Wei X, Witter S, Yin J, Walley J, Liu S, et al. Incremental cost-effectiveness of improving treatment results among migrant tuberculosis patients in Shanghai. Int J Tuberc Lung Dis. 2013;17(8):1056–64. Epub 2013/07/06. doi: 10.5588/ijtld.12.0799 . [DOI] [PubMed] [Google Scholar]
- 108.Jahnavi G, Sudha CH. Randomised controlled trial of food supplements in patients with newly diagnosed tuberculosis and wasting. Singapore Med J. 2010;51(12):957–62. Epub 2011/01/12. . [PubMed] [Google Scholar]
- 109.Lutge E, Lewin S, Volmink J, Friedman I, Lombard C. Economic support to improve tuberculosis treatment outcomes in South Africa: a pragmatic cluster-randomized controlled trial. Trials. 2013;14:154 Epub 2013/05/30. doi: 10.1186/1745-6215-14-154 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Martins N, Morris P, Kelly PM. Food incentives to improve completion of tuberculosis treatment: randomised controlled trial in Dili, Timor-Leste. BMJ. 2009;339:b4248 Epub 2009/10/28. doi: 10.1136/bmj.b4248 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Sudarsanam TD, John J, Kang G, Mahendri V, Gerrior J, Franciosa M, et al. Pilot randomized trial of nutritional supplementation in patients with tuberculosis and HIV-tuberculosis coinfection receiving directly observed short-course chemotherapy for tuberculosis. Trop Med Int Health. 2011;16(6):699–706. Epub 2011/03/23. doi: 10.1111/j.1365-3156.2011.02761.x . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Al-Hajjaj MS, Al-Khatim IM. High rate of non-compliance with anti-tuberculosis treatment despite a retrieval system: a call for implementation of directly observed therapy in Saudi Arabia. Int J Tuberc Lung Dis. 2000;4(4):345–9. Epub 2000/04/25. . [PubMed] [Google Scholar]
- 113.Bronner LE, Podewils LJ, Peters A, Somnath P, Nshuti L, van der Walt M, et al. Impact of community tracer teams on treatment outcomes among tuberculosis patients in South Africa. BMC Public Health. 2012;12:621 Epub 2012/08/09. doi: 10.1186/1471-2458-12-621 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Hermans SM, Elbireer S, Tibakabikoba H, Hoefman BJ, Manabe YC. Text messaging to decrease tuberculosis treatment attrition in TB-HIV coinfection in Uganda. Patient Prefer Adherence. 2017;11:1479–87. Epub 2017/09/19. doi: 10.2147/PPA.S135540 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Snidal SJ, Barnard G, Atuhairwe E, Ben Amor Y. Use of eCompliance, an innovative biometric system for monitoring of tuberculosis treatment in rural Uganda. Am J Trop Med Hyg. 2015;92(6):1271–9. Epub 2015/04/08. doi: 10.4269/ajtmh.14-0413 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Thomson KA, Cheti EO, Reid T. Implementation and outcomes of an active defaulter tracing system for HIV, prevention of mother to child transmission of HIV (PMTCT), and TB patients in Kibera, Nairobi, Kenya. Trans R Soc Trop Med Hyg. 2011;105(6):320–6. Epub 2011/04/23. doi: 10.1016/j.trstmh.2011.02.011 . [DOI] [PubMed] [Google Scholar]
- 117.Iribarren S, Beck S, Pearce PF, Chirico C, Etchevarria M, Cardinale D, et al. TextTB: A Mixed Method Pilot Study Evaluating Acceptance, Feasibility, and Exploring Initial Efficacy of a Text Messaging Intervention to Support TB Treatment Adherence. Tuberc Res Treat. 2013;2013:349394 Epub 2014/01/24. doi: 10.1155/2013/349394 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Krishnaswami KV S P.R.; Tripathy S.P.; Vaidyanathan B.; Radhakrishna S.; Fox W. A randomised study of two policies for managing default in out-patients collecting supplies of drugs for pulmonary tuberculosis in a large city in South India. Tubercle. 1981;61:103–12. [DOI] [PubMed] [Google Scholar]
- 119.Kunawarak P P S.; Chantawong S.; Pokaew P.; Traisathit P.; Srithanaviboonchai K.; Plipat T. Tuberculosis treatment with mobile-phone medication reminders in Nothern Thailand. Southeast Asian J Trop Med Public Health. 2011;42(6):1444–51. [PubMed] [Google Scholar]
- 120.Mohammed S, Glennerster R, Khan AJ. Impact of a Daily SMS Medication Reminder System on Tuberculosis Treatment Outcomes: A Randomized Controlled Trial. PLoS ONE. 2016;11(11):e0162944 doi: 10.1371/journal.pone.0162944 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Mohan A, Nassir H, Niazi A. Does routine home visiting improve the return rate and outcome of DOTS patients who delay treatment? East Mediterr Health J. 2003;9(4):702–8. Epub 2005/03/08. . [PubMed] [Google Scholar]
- 122.Moulding TS, Caymittes M. Managing medication compliance of tuberculosis patients in Haiti with medication monitors. Int J Tuberc Lung Dis. 2002;6(4):313–9. Epub 2002/04/09. . [PubMed] [Google Scholar]
- 123.Paramasivan RP, R. T.; Rajasekaran S. Short course chemotherapy: A controlled study of indirect default retrieval method. Ind J Tub. 1993;40:185–90. [Google Scholar]
- 124.Tanke ED, Leirer VO. Automated telephone reminders in tuberculosis care. Med Care. 1994;32(4):380–9. Epub 1994/04/01. . [DOI] [PubMed] [Google Scholar]
- 125.Broomhead S, Mars M. Retrospective return on investment analysis of an electronic treatment adherence device piloted in the Northern Cape Province. Telemed J E Health. 2012;18(1):24–31. Epub 2011/12/14. doi: 10.1089/tmj.2011.0143 . [DOI] [PubMed] [Google Scholar]
- 126.Liu X, Lewis JJ, Zhang H, Lu W, Zhang S, Zheng G, et al. Effectiveness of Electronic Reminders to Improve Medication Adherence in Tuberculosis Patients: A Cluster-Randomised Trial. PLoS Med. 2015;12(9):e1001876 doi: 10.1371/journal.pmed.1001876 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Lewin S, Dick J, Zwarenstein M, Lombard CJ. Staff training and ambulatory tuberculosis treatment outcomes: a cluster randomized controlled trial in South Africa. Bull World Health Organ. 2005;83(4):250–9. Epub 2005/05/04. . [PMC free article] [PubMed] [Google Scholar]
- 128.Puchalski Ritchie LM, Schull MJ, Martiniuk AL, Barnsley J, Arenovich T, van Lettow M, et al. A knowledge translation intervention to improve tuberculosis care and outcomes in Malawi: a pragmatic cluster randomized controlled trial. Implement Sci. 2015;10:38 Epub 2015/04/19. doi: 10.1186/s13012-015-0228-y . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Datiko DG, Lindtjorn B. Health extension workers improve tuberculosis case detection and treatment success in southern Ethiopia: a community randomized trial. PLoS ONE. 2009;4(5):e5443 Epub 2009/05/09. doi: 10.1371/journal.pone.0005443 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Safdar N, Hinderaker SG, Baloch NA, Enarson DA, Khan MA, Morkve O. Childhood tuberculosis deskguide and monitoring: an intervention to improve case management in Pakistan. BMC Health Serv Res. 2011;11:187 Epub 2011/08/13. doi: 10.1186/1472-6963-11-187 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Shin S, Livchits V, Connery HS, Shields A, Yanov S, Yanova G, et al. Effectiveness of alcohol treatment interventions integrated into routine tuberculosis care in Tomsk, Russia. Addiction. 2013;108(8):1387–96. Epub 2013/03/16. doi: 10.1111/add.12148 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Alvarez Gordillo G AG J.; Dorantes Jimenez J.E. Estrategia educativa para incrementar el cumplimiento del regimen antituberculoso en Chiapas, Mexico. Pan Am J Public Health. 2003;14(6):402–8. [DOI] [PubMed] [Google Scholar]
- 133.Demissie M, Getahun H, Lindtjorn B. Community tuberculosis care through "TB clubs" in rural North Ethiopia. Soc Sci Med. 2003;56(10):2009–18. Epub 2003/04/17. . [DOI] [PubMed] [Google Scholar]
- 134.Fang XH, Guan SY, Tang L, Tao FB, Zou Z, Wang JX, et al. Effect of Short Message Service on Management of Pulmonary Tuberculosis Patients in Anhui Province, China: A Prospective, Randomized, Controlled Study. Med Sci Monit. 2017;23:2465–9. Epub 2017/05/24. doi: 10.12659/MSM.904957 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Chuck C, Robinson E, Macaraig M, Alexander M, Burzynski J. Enhancing management of tuberculosis treatment with video directly observed therapy in New York City. Int J Tuberc Lung Dis. 2016;20(5):588–93. Epub 2016/04/17. doi: 10.5588/ijtld.15.0738 . [DOI] [PubMed] [Google Scholar]
- 136.Wade VA, Karnon J, Eliott JA, Hiller JE. Home videophones improve direct observation in tuberculosis treatment: a mixed methods evaluation. PLoS ONE. 2012;7(11):e50155 doi: 10.1371/journal.pone.0050155 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Atkins S, Lewin S, Jordaan E, Thorson A. Lay health worker-supported tuberculosis treatment adherence in South Africa: an interrupted time-series study. Int J Tuberc Lung Dis. 2011;15(1):84–9, i Epub 2011/02/01. . [PubMed] [Google Scholar]
- 138.Chan PC, Huang SH, Yu MC, Lee SW, Huang YW, Chien ST, et al. Effectiveness of a government-organized and hospital-initiated treatment for multidrug-resistant tuberculosis patients—a retrospective cohort study. PLoS ONE. 2013;8(2):e57719 doi: 10.1371/journal.pone.0057719 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Davidson BL. A controlled comparison of directly observed therapy vs self-administered therapy for active tuberculosis in the urban United States. Chest. 1998;114(5):1239–43. Epub 1998/11/21. . [DOI] [PubMed] [Google Scholar]
- 140.Farmer P, Robin S, Ramilus SL, Kim JY. Tuberculosis, poverty, and "compliance": lessons from rural Haiti. Semin Respir Infect. 1991;6(4):254–60. Epub 1991/12/01. . [PubMed] [Google Scholar]
- 141.Garden B, Samarina A, Stavchanskaya I, Alsterlund R, Ovregaard A, Taganova O, et al. Food incentives improve adherence to tuberculosis drug treatment among homeless patients in Russia. Scand J Caring Sci. 2013;27(1):117–22. Epub 2012/06/08. doi: 10.1111/j.1471-6712.2012.01009.x . [DOI] [PubMed] [Google Scholar]
- 142.Soares EC, Vollmer WM, Cavalcante SC, Pacheco AG, Saraceni V, Silva JS, et al. Tuberculosis control in a socially vulnerable area: a community intervention beyond DOT in a Brazilian favela. Int J Tuberc Lung Dis. 2013;17(12):1581–6. Epub 2013/11/10. doi: 10.5588/ijtld.13.0152 . [DOI] [PubMed] [Google Scholar]
- 143.Yassin MA, Datiko DG, Tulloch O, Markos P, Aschalew M, Shargie EB, et al. Innovative community-based approaches doubled tuberculosis case notification and improve treatment outcome in Southern Ethiopia. PLoS ONE. 2013;8(5):e63174 doi: 10.1371/journal.pone.0063174 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Drabo M, Zerbo R, Berthe A, Ouedrago L, Konfe S, Mugishe E, et al. [Community involvement in tuberculosis care in three rural health districts of Burkina Faso]. Sante Publique. 2009;21(5):485–97. Epub 2010/03/17. . [PubMed] [Google Scholar]
- 145.Hsieh CJ, Lin LC, Kuo BI, Chiang CH, Su WJ, Shih JF. Exploring the efficacy of a case management model using DOTS in the adherence of patients with pulmonary tuberculosis. J Clin Nurs. 2008;17(7):869–75. Epub 2007/09/14. doi: 10.1111/j.1365-2702.2006.01924.x . [DOI] [PubMed] [Google Scholar]
- 146.Khortwong P, Kaewkungwal J. Thai health education program for improving TB migrant's compliance. J Med Assoc Thai. 2013;96(3):365–73. Epub 2013/04/02. . [PubMed] [Google Scholar]
- 147.Morisky DE, Malotte CK, Choi P, Davidson P, Rigler S, Sugland B, et al. A patient education program to improve adherence rates with antituberculosis drug regimens. Health Educ Q. 1990;17(3):253–67. Epub 1990/01/01. . [DOI] [PubMed] [Google Scholar]
- 148.Thiam S, LeFevre AM, Hane F, Ndiaye A, Ba F, Fielding KL, et al. Effectiveness of a strategy to improve adherence to tuberculosis treatment in a resource-poor setting: a cluster randomized controlled trial. JAMA. 2007;297(4):380–6. Epub 2007/01/25. doi: 10.1001/jama.297.4.380 . [DOI] [PubMed] [Google Scholar]
- 149.Duarte R, Santos A, Mota M, Carvalho A, Marques A, Barros H. Involving community partners in the management of tuberculosis among drug users. Public Health. 2011;125(1):60–2. Epub 2011/01/25. doi: 10.1016/j.puhe.2010.09.002 . [DOI] [PubMed] [Google Scholar]
- 150.Macq J, Solis A, Martinez G, Martiny P. Tackling tuberculosis patients' internalized social stigma through patient centred care: an intervention study in rural Nicaragua. BMC Public Health. 2008;8:154 Epub 2008/05/10. doi: 10.1186/1471-2458-8-154 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Wai PP, Shewade HD, Kyaw NTT, Kyaw KWY, Thein S, Si Thu A, et al. Patients with MDR-TB on domiciliary care in programmatic settings in Myanmar: Effect of a support package on preventing early deaths. PLoS ONE. 2017;12(12):e0187223 doi: 10.1371/journal.pone.0187223 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Tola HH, Shojaeizadeh D, Tol A, Garmaroudi G, Yekaninejad MS, Kebede A, et al. Psychological and Educational Intervention to Improve Tuberculosis Treatment Adherence in Ethiopia Based on Health Belief Model: A Cluster Randomized Control Trial. PLoS ONE. 2016;11(5):e0155147 doi: 10.1371/journal.pone.0155147 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Karumbi J, Garner P. Directly observed therapy for treating tuberculosis. Cochrane Database Syst Rev. 2015;(5):CD003343 Epub 2015/05/30. doi: 10.1002/14651858.CD003343.pub4 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Bothwell LE, Greene JA, Podolsky SH, Jones DS. Assessing the Gold Standard—Lessons from the History of RCTs. N Engl J Med. 2016;374(22):2175–81. Epub 2016/06/02. doi: 10.1056/NEJMms1604593 . [DOI] [PubMed] [Google Scholar]
- 155.Rothwell PM. External validity of randomised controlled trials: "to whom do the results of this trial apply?". Lancet. 2005;365(9453):82–93. Epub 2005/01/11. doi: 10.1016/S0140-6736(04)17670-8 . [DOI] [PubMed] [Google Scholar]
- 156.Chavez-MacGregor M, Giordano SH. Randomized Clinical Trials and Observational Studies: Is There a Battle? J Clin Oncol. 2016;34(8):772–3. Epub 2016/01/21. doi: 10.1200/JCO.2015.64.7487 . [DOI] [PubMed] [Google Scholar]
- 157.Pasipanodya JG, Gumbo T. A meta-analysis of self-administered vs directly observed therapy effect on microbiologic failure, relapse, and acquired drug resistance in tuberculosis patients. Clin Infect Dis. 2013;57(1):21–31. Epub 2013/03/15. doi: 10.1093/cid/cit167 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.A controlled clinical trial of oral short-course regimens in the treatment of sputum-positive pulmonary tuberculosis. Tuberculosis Research Centre. Int J Tuberc Lung Dis. 1997;1(6):509–17. Epub 1998/03/06. . [PubMed] [Google Scholar]
- 159.Lienhardt C, Ogden JA. Tuberculosis control in resource-poor countries: have we reached the limits of the universal paradigm? Trop Med Int Health. 2004;9(7):833–41. Epub 2004/07/02. doi: 10.1111/j.1365-3156.2004.01273.x . [DOI] [PubMed] [Google Scholar]
- 160.Khan MA, Walley JD, Witter SN, Imran A, Safdar N. Costs and cost-effectiveness of different DOT strategies for the treatment of tuberculosis in Pakistan. Directly Observed Treatment. Health Policy Plan. 2002;17(2):178–86. Epub 2002/05/10. . [DOI] [PubMed] [Google Scholar]
- 161.Ethics guidance for the implementation of the End TB strategy 2017.
- 162.Guidance on ethics of tuberculosis prevention, care and control 2010 [cited 2017 Nov 5]. http://whqlibdoc.who.int/publications/2010/9789241500531_eng.pdf?ua=1
- 163.Krueger K, Ruby D, Cooley P, Montoya B, Exarchos A, Djojonegoro BM, et al. Videophone utilization as an alternative to directly observed therapy for tuberculosis. Int J Tuberc Lung Dis. 2010;14(6):779–81. Epub 2010/05/22. . [PubMed] [Google Scholar]
- 164.Orenstein EW, Basu S, Shah NS, Andrews JR, Friedland GH, Moll AP, et al. Treatment outcomes among patients with multidrug-resistant tuberculosis: systematic review and meta-analysis. Lancet Infect Dis. 2009;9(3):153–61. Epub 2009/02/28. doi: 10.1016/S1473-3099(09)70041-6 . [DOI] [PubMed] [Google Scholar]
- 165.Toczek A, Cox H, du Cros P, Cooke G, Ford N. Strategies for reducing treatment default in drug-resistant tuberculosis: systematic review and meta-analysis. Int J Tuberc Lung Dis. 2013;17(3):299–307. Epub 2012/12/06. doi: 10.5588/ijtld.12.0537 . [DOI] [PubMed] [Google Scholar]
- 166.Nahid P, Dorman SE, Alipanah N, Barry PM, Brozek JL, Cattamanchi A, et al. Official American Thoracic Society/Centers for Disease Control and Prevention/Infectious Diseases Society of America Clinical Practice Guidelines: Treatment of Drug-Susceptible Tuberculosis. Clin Infect Dis. 2016;63(7):e147–e95. Epub 2016/08/16. doi: 10.1093/cid/ciw376 . [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(DOC)
(DOC)
TB, tuberculosis.
(DOCX)
SMS, short message service; TB, tuberculosis.
(DOCX)
1 = Newcastle-Ottawa Score provided for cohort studies. Quality of RCTs are presented separately. 2 = Study includes >50% HIV/TB patients. 3 = Study includes >50% MDR-TB patients. RCT, randomized controlled trial; TB, tuberculosis.
(DOCX)
(TIF)
(TIF)
DOT, directly observed therapy; SAT, self-administered therapy.
(TIF)
“Not estimable” denotes a subgroup within a study not included in the meta-analysis. DOT, directly observed therapy; SAT, self-administered therapy.
(TIF)
DOT, directly observed therapy; SAT, self-administered therapy.
(TIF)
“Not estimable” denotes a subgroup within a study not included in the meta-analysis. DOT, directly observed therapy; SAT, self-administered therapy.
(TIF)
DOT, directly observed therapy; SAT, self-administered therapy.
(TIF)
“Not estimable” denotes a subgroup within a study not included in the meta-analysis. DOT, directly observed therapy; SAT, self-administered therapy.
(TIF)
DOT, directly observed therapy; SAT, self-administered therapy.
(TIF)
“Not estimable” denotes a subgroup within a study not included in the meta-analysis. DOT, directly observed therapy; SAT, self-administered therapy.
(TIF)
DOT, directly observed therapy; SAT, self-administered therapy.
(TIF)
“Not estimable” denotes a subgroup within a study not included in the meta-analysis. DOT, directly observed therapy; SAT, self-administered therapy.
(TIF)
DOT, directly observed therapy; SAT, self-administered therapy.
(TIF)
DOT, directly observed therapy; SAT, self-administered therapy.
(TIF)
DOT, directly observed therapy; SAT, self-administered therapy; TB, tuberculosis.
(TIF)
DOT, directly observed therapy; SAT, self-administered therapy; TB, tuberculosis.
(TIF)
DOT, directly observed therapy; SAT, self-administered therapy; TB, tuberculosis.
(TIF)
DOT, directly observed therapy; SAT, self-administered therapy; TB, tuberculosis.
(TIF)
DOT, directly observed therapy; SAT, self-administered therapy; TB, tuberculosis.
(TIF)
DOT, directly observed therapy; SAT, self-administered therapy; TB, tuberculosis.
(TIF)
DOT, directly observed therapy; MDR-TB, multidrug-resistant tuberculosis; SAT, self-administered therapy.
(TIF)
DOT, directly observed therapy; MDR-TB, multidrug-resistant tuberculosis; SAT, self-administered therapy.
(TIF)
DOT, directly observed therapy; MDR-TB, multidrug-resistant tuberculosis; SAT, self-administered therapy.
(TIF)
DOT, directly observed therapy; MDR-TB, multidrug-resistant tuberculosis; SAT, self-administered therapy.
(TIF)
DOT, directly observed therapy.
(TIF)
DOT, directly observed therapy.
(TIF)
DOT, directly observed therapy.
(TIF)
DOT, directly observed therapy.
(TIF)
DOT, directly observed therapy.
(TIF)
DOT, directly observed therapy.
(TIF)
DOT, directly observed therapy.
(TIF)
DOT, directly observed therapy.
(TIF)
DOT, directly observed therapy.
(TIF)
DOT, directly observed therapy.
(TIF)
DOT, directly observed therapy.
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(A) Meta-analysis of mortality rates in patients receiving reminders/tracers in addition to standard care versus standard care alone. (B) Sensitivity analysis: removing the heaviest weighted study (Bronner 2012) in which control and intervention cohorts had significantly different pre-intervention mortality rates. “Not estimable” denotes a subgroup within a study not included in the meta-analysis.
(TIF)
(TIF)
(A) Meta-analysis of cure rates in patients receiving reminders/tracers in addition to standard care versus standard care alone. (B) Sensitivity analysis: removing the heaviest weighted study (Bronner 2012) in which control and intervention cohorts had significantly different pre-intervention cure rates.
(TIF)
(A) Meta-analysis of treatment failure rates in patients receiving reminders/tracers in addition to standard care versus standard care alone—cohort studies. (B) Sensitivity analysis: removing the heaviest weighted study (Bronner 2012) in which control and intervention cohorts had significantly different pre-intervention treatment failure rates.
(TIF)
RCT, randomized controlled trial.
(TIF)
(A) Meta-analysis of rates of loss to follow-up in patients receiving reminders/tracers in addition to standard care versus standard care alone. (B) Sensitivity analysis: removing the heaviest weighted study (Bronner 2012) in which control and intervention cohorts had significantly different pre-intervention loss to follow-up rates.
(TIF)
(TIF)
Poor adherence is defined as the percentage of patient-months in which at least 20% of doses were missed.
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
RCT, randomized controlled trial.
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
SMS, short message service; VOT, video-observed therapy.
(TIF)
SMS, short message service; VOT, video-observed therapy.
(TIF)
RCT, randomized controlled trial; SMS, short message service.
(TIF)
SMS, short message service.
(TIF)
SMS, short message service.
(TIF)
SMS, short message service.
(TIF)
Poor adherence is defined as the percentage of patient-months in which at least 20% of doses were missed. SMS, short message service.
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
(TIF)
Data Availability Statement
All relevant data are within the paper and its Supporting information files.