Abstract
A non-resolving inflammation of the endothelium is recognised to be an important process leading to atherosclerosis. In diabetes, this process is thought to account for a significant number of cardiovascular disease-associated death and disability. However, the molecular mechanisms by which diabetes contributes to endothelial inflammation remain to be established. Whilst there is some evidence linking hyperglycaemia-induced reactive oxygen species (ROS) formation by the mitochondrial electron-transport chain to oxidative stress, cellular injury and apoptosis in the endothelium, a clear link to endothelium inflammation has not yet been established. The mitochondrial molecular stress protein Hsp60 is known to be secreted from mammalian cells and is capable of activating pro-inflammatory mediators on target cells expressing Toll-like receptors (TLRs). Hsp60 is also known to be elevated in serum of diabetes patients and has been shown to be upregulated by hyperglycaemic growth conditions in cultured human HeLa cells. This study shows that Hsp60 induced in human acute monocyte leukaemia cell line (THP-1) cells grown under hyperglycaemic conditions (25 mM glucose) was able to be secreted into growth media. Furthermore, the secretion of Hsp60 from THP-1 cells was able to be inhibited by 5,5-(N-N-dimethyl)-amiloride hydrochloride (DMA), an exosomal inhibitor. Interestingly, the conditioned media obtained from THP-1 cells grown in the presence of 25 mM glucose was able to induce the secretion of TNF-α in human vascular endothelium cell line (HUVEC). When conditioned media was immuno-depleted of Hsp60, there was a significant reduction in the release of TNF-α from the HUVEC cells. This suggests that a potential link may exist between hyperglycaemia-induced expression of Hsp60 in monocyte cells and vascular inflammation. Circulating levels of Hsp60 due to mitochondrial stress in diabetes patients could therefore be an important modulator of inflammation in endothelial cells and thus contribute to the increased incidences of atherosclerosis in diabetes mellitus.
Keywords: Hsp60, Hyperglycaemia, Inflammation, THP-1 monocytes, TNF-α, Endothelial cells
Introduction
Atherosclerosis is the major cause of cardiovascular disease. For reasons unknown, the underlying molecular changes associated with atherosclerosis are exacerbated in diabetes mellitus (DM) (King and Grant 2016). Atherosclerosis is triggered by complex interactions between a number of cell types and oxidised low-density lipoprotein molecules leading to the formation of plaques (Tabas et al. 2015). This process is also associated with non-resolving inflammation that can lead to endothelial cell destruction. Whilst it is clear that hyperglycaemia-induced reactive oxygen species (ROS) formation by the mitochondrial electron-transport chain can lead to oxidative stress, cellular injury and apoptosis in the endothelium (Shah and Brownlee 2016), there is no clear evidence to suggest that ROS are indeed key contributors to endothelium inflammation.
Heat-shock protein 60 (Hsp60) is a molecular stress protein predominantly localised to the mitochondrion matrix functioning as a protein-folding machine and is considered to be a good indicator of mitochondrial stress (Cabiscol et al. 2002; Haak et al. 2009; Jovaisaite et al. 2014; Martinus et al. 1996). Interestingly, Hsp60 has also been shown to be secreted from a variety of mammalian cell types (Bassan et al. 1998; Merendino et al. 2010) and is known to be found at elevated levels in saliva and serum of both type 1 and 2 diabetes mellitus patients (Verrijn Stuart et al. 2012; Yuan et al. 2011). However, the pathological consequence of having elevated levels of Hsp60 in systemic circulation and the cell types responsible for secretion of Hsp60 into circulation under hyperglycaemia is not yet known.
Interestingly, Hsp60 is capable of modulating the expression of pro-inflammatory cytokines such as interleukins 6 and 8 (IL-6, IL-8) and tumour necrosis factor alpha (TNF-α) in cells expressing Toll-like receptors 2 and 4 (TLR2/4) (Swaroop et al. 2016). A number of studies suggest that extracellular Hsp60 plays a role as a cellular “danger” signal for cellular and humoral immune reactions (Asea and De Maio 2007; Chen et al. 1999; Grundtman et al. 2011; Pockley 2003). Hsp60 has also been suggested to play a role in the pathogenesis of atherosclerosis in DM (Frostegard 2013; Rabczynski et al. 2012). There is also some evidence to suggest that anti-Hsp60 antibodies are involved with the destruction of vascular endothelial cells (Almanzar et al. 2012; Mandal et al. 2005). However, the precise mechanisms by which Hsp60 promotes atherosclerosis remain unclear (Zhao et al. 2015a).
We have recently proposed a hypothesis that Hsp60 secreted from monocyte cells under hyperglycaemic stress could play a key role as a trigger of endothelial inflammation in DM (Juwono and Martinus 2016). The objective of this investigation was to establish if there is indeed an association between monocyte-derived mitochondrial Hsp60 and vascular inflammation. Using human acute monocyte leukaemia cell line (THP-1) and vascular endothelium cell line (HUVEC) as model in vitro cell systems, we investigated to see if (a) Hsp60 was induced and secreted into the media by the THP-1 monocyte cells that were subjected to hyperglycaemic growth conditions and (b) if the Hsp60 containing conditioned media was subsequently able to induce the expression of TNF-α in cultured human endothelial cells.
Methods
Cell culture of THP-1 monocytes and HUVEC endothelial cells
Human acute monocytic leukaemia cell line (THP-1) and vascular endothelium cell line (HUVEC) were obtained from the American Tissue Culture Collection. THP-1 cells were cultured in RPMI Media 1640 (Gibco) supplemented with 10% foetal bovine serum (FBS) and 1 mM pyruvate. HUVEC cells were grown in F-12K medium containing 0.1 mg/ml heparin, 0.05 mg/ml endothelial cell growth supplement (ECGS) and 10% FBS. Cells were grown at 37 °C, 5% CO2 in a humidified incubator. All experiments were carried out using near confluent cells which had been passaged 2 days prior the experiment.
THP-1 cells were grown in the presence of 5 mM glucose (control condition) and 25 mM glucose as described previously (Hall and Martinus 2013).
MTT assay
The conversion of the tetrazolium salt redox dye, 3-(4,5-dimethylthiazol-2yl)-2,5-diphenlytetrazolium bromide (MTT), into a formazan product was used as an assay for mitochondrial dehydrogenase activity. After exposure of the THP-1 cells to 5 and 25 mM glucose, the MTT reductions were determined at 570 and 655 nm using the Sigma MTT-based In vitro Toxicology Assay Kit according to the manufacturer’s protocol.
LDH assay
The lactate dehydrogenase (LDH) assays were carried out using a CytoTox 96®Non-radioactive Cytotoxicity Assay commercial kit (Promega) as per the manufacturer’s protocol as described previously (Wang et al. 2016). The assay basically involved adding supernatant from cells to reconstituted substrate mixture. After incubation at room temperature for 30 min (protected from light), stop solution was added and the absorbance was measured at 490 nm in a plate reader (Bio-Rad 680). A number of controls were included in these experiments: spontaneous control corrected for the spontaneous release of LDH from cells; maximum control represented 100% release of LDH upon cell lysis and culture medium background control corrected for any LDH activity contributed by FBS in the growth media and the varying amounts of phenol red in the media. The percentage of LDH content (relative to maximum control samples) was interpreted as the percentage of lysed cells on the assumption that every dead cell released equal amounts of LDH.
Heat shock of THP-1 cells
As a means of obtaining a positive control for expression of Hsp60, heat-shock treatment was utilised (Martinus et al. 1996). Essentially, THP-1 cells were grown in culture flasks for 3 days, so as to reach the exponential growth stage. The media was then replaced with RPMI pre-warmed at 42 °C, and the flask was placed into a 42 °C incubator for 20 min. Following heat treatment, the media was again replaced, with RPMI pre-warmed to 37 °C. The culture was returned to the standard incubating conditions for 6 h. After this time, the cells were harvested for protein extraction and Western blot analysis.
Protein extraction and Western blot analysis
The extraction of cellular protein, SDS-PAGE and Western blot analysis was carried out as described in Tsuei and Martinus (2012). Briefly, total cellular protein was extracted using the TENT buffer (50 mM Tris, 250 mM NaCl, 5 mM EDTA, 1% Triton X-100 and freshly supplemented with 0.4 mM phenlymethylsulfonyl fluoride). Protein concentrations were estimated using the BCA Protein Assay Kit (Pierce), and 15 μg of total protein was separated by the SDS-PAGE. Proteins were then transferred to a nitrocellulose membrane (Bio-Rad) and probed with Ponceau S staining to ensure that there was equal loading of proteins across all the lanes prior to the Western blot analysis. After blocking the blots with 10% skim milk powder, the membranes were incubated in the primary polyclonal rabbit Hsp60 (Stressgen SPA805) followed by peroxidase conjugated goat anti-rabbit IgG (Sigma). The membrane was then incubated with SuperSignal West Pico Chemiluminescent Substrate (Pierce) and the Hsp60 protein band visualized through LAS-100 Plus Gel Documentation System (Fujifilm). The intensity of the bands was analysed using the Gel Quant software. After stripping, the same blots were then probed with the anti-actin antibody. The Gel Quant values generated for the Hsp60 protein were normalised to the housekeeping protein actin after probing the same membrane with the anti-actin antibody.
Preparation of conditioned media
Conditioned media from THP-1 cells grown for 72 h in the presence of 5 mM glucose (GCM 5 mM) and 25 mM glucose (GCM 25 mM) was collected and centrifuged at 800g for 10 min to remove cells and debris. This media was lyophilised (as this procedure is known to retain the biological activity of cellular proteins) and reconstituted in sterile phosphate-buffered saline (PBS). Levels of Hsp60 in the conditioned media were determined by anti-Hsp60 ELISA kit according to the manufacturer’s instructions (sensitivity 1.37 ng/ml) (Stressgen). Results obtained were normalised for cell number and expressed as ng/106 cells. The conditioned media obtained from THP-1 cells grown in the presence of 5 and 25 mM glucose was then added to cultures of newly seeded HUVEC cells and grown under standard growth conditions for 48 h. The tumour necrosis factor-alpha (TNF-α) level in the HUVEC cell media was determined by human TNF-α ELISA kit according to the manufacturer’s instructions (sensitivity 1.7 pg/ml) (ThermoFisher Scientific) and expressed as pg/ml. As a positive control, HUVEC cells (106) were also treated with 1 ng/ml interleukin-1α (IL-1α) (Sigma) for 24 h, and the media collected and assayed for TNF-α by ELISA.
Immuno-depletion of Hsp60 from conditioned media
The Hsp60 secreted into conditioned media obtained fromTHP-1 cells grown in the presence of 25 mM glucose was immune-precipitated by a protocol adapted from Merendino et al. (2010). Essentially, conditioned media (500 μg of protein) was incubated with polyclonal anti-Hsp60 antibodies (5 μg) at room temperature for 2 h followed by incubation with 20 μl of protein A-sepharose at 4 °C for 12 h. The incubation mixture was then centrifuged at 14,000g for 20 s at 4 °C, the pellet was discarded and the supernatant retained and added to HUVEC cells grown under standard growth conditions for 48 h. TNF-α secreted was determined by ELISA as described previously.
Inhibition of Hsp60 secretion
THP-1 cells grown in the presence of 5 and 25 mM glucose (as described previously) were incubated with 5 and 10 nM 5,5-(N-N-dimethyl)-amiloride hydrochloride (DMA), an exosomal inhibitor for 1 h (Lyu et al. 2015). The amount of Hsp60 secreted into the growth medium in the presence of DMA was determined by anti-Hsp60 ELISA and compared to untreated controls. LDH assays were carried out on THP-1 cells exposed to DMA as described previously. Conditioned media obtained from these cells were added to HUVEC cells grown under standard growth conditions for 48 h, and the TNF-α secreted was determined by anti-TNF-α ELISA as described previously.
Statistical analysis
All statistical analysis in this study was carried out using Microsoft Excel. Data was averaged where appropriate, and the standard error of the mean (S.E.M.) was calculated using Excel. A two-tailed Student’s t test was carried out to determine the significance of the data, and the accepted level of significance at p < 0.05 and p < 0.005, which was denoted as (*) and (**), respectively.
Results/discussion
Hyperglycaemia induced Hsp60 expression in THP-1 monocyte cells
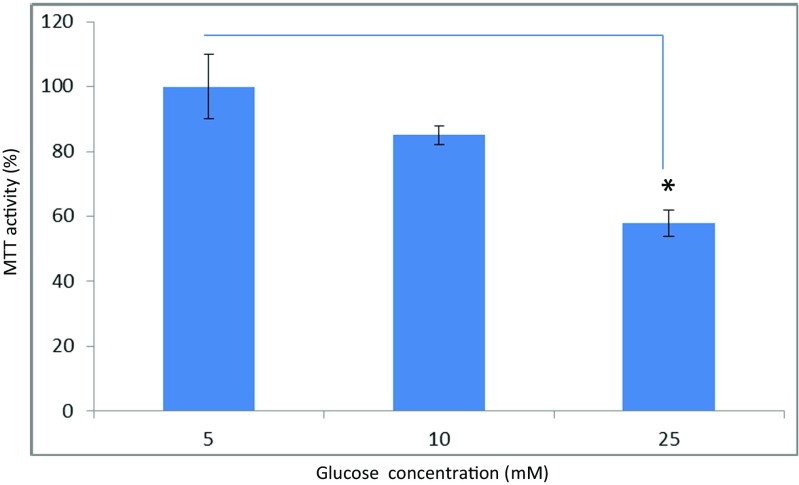
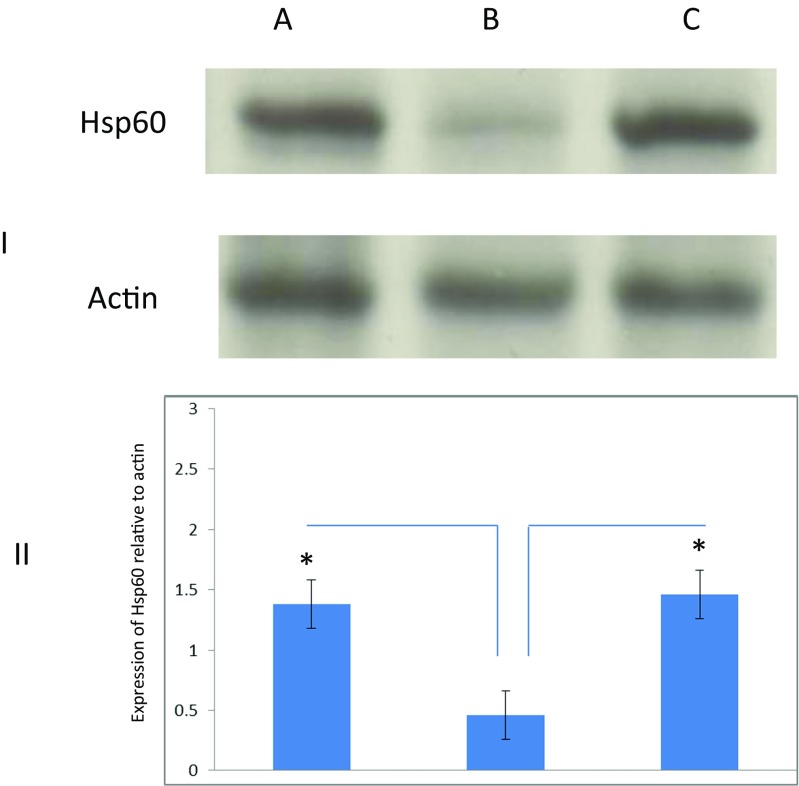
The conversion of MTT to a formazan product by mitochondrial dehydrogenases was used as an index of mitochondrial viability. When human THP-1 monocyte cells were cultured in the presence of 5, 10 and 25 mM glucose, there was a significant (p < 0.05) 30% decrease in mitochondrial dehydrogenase activity under the hyperglycaemic growth condition (25 mM glucose) compared to the normal glucose levels of 5 mM (Fig. 1). To determine if growing THP-1 cells under hyperglycaemic conditions also led to an increase in expression of Hsp60 (as an indicator of mitochondrial stress), total cell protein extracted from THP-1 cells grown in the presence of 5 and 25 mM glucose was separated by SDS-PAGE and probed with an ant-Hsp60 antibody by Western blot analysis. When THP-1 cells were grown in the presence of 25 mM glucose, there was a 3-fold increase in Hsp60 protein levels compared to cells grown in the presence of 5 mM glucose (Fig. 2 A). This was comparable to the 3.17-fold levels of Hsp60 induction observed in the heat-shocked THP-1 cells (Fig. 2 C) which acted as a positive control. These results also align well with our previous findings that human HeLa cells grown in the presence of hyperglycaemic conditions (25 mM glucose) led to a 3.5-fold increase in Hsp60 protein levels when compared to cells grown in the presence of 5 mM glucose (Hall and Martinus 2013). Hsp60 is a constitutively expressed protein (D’Souza and Brown 1998); therefore, the levels expressed under normal glucose (5 mM) growth conditions (Fig. 2 B) represents basal levels of expression in THP-1 cells used in this study. These observations confirm that exposure of THP-1 monocyte cells to hyperglycaemic growth conditions does indeed lead to an increase in the protein levels of mitochondrial Hsp60.
Fig. 1.
Mitochondrial dehydrogenase activity of THP-1 cells grown for 5 days in the presence of 10 and 25 mM glucose expressed as a percentage of the 5 mM glucose levels. N = 3 independent experiments. The error bars represent ±S.E.M. (*) represents a statistically significant value (p < 0.05)
Fig. 2.
I. Western blot of Hsp60 and actin expression from THP-1 cells grown for 5 days in the presence of 5 and 25 mM glucose. Lanes A and B cells grown in 25 and 5 mM glucose, respectively. Lane C is a heat-shocked positive control. The Western blot is a representative of N = 3 independent experiments. II. Expression of Hp60 relative to actin. Cells exposed to 25 mM glucose (lane A) had a 3.0-fold induction compared with cells grown in 5 mM glucose (lane B). The heat-shocked cells (lane C) which acted as a positive control had a 3.17-fold induction compared to cells grown in 5 mM glucose (lane B). The error bars represent ±S.E.M. (*) represents a statistically significant value (p < 0.05)
Hyperglycaemia is known to induce reactive oxygen species (ROS) formation by impairment of the mitochondrial electron-transport chain activity (Giacco and Brownlee 2010; Hall and Martinus 2013), which can lead to cellular injury and apoptosis in the endothelium (Fiorentino et al. 2013). High glucose has also been shown to induce the secretion of TNF-α by primary human monocytes via down-regulation of transmembrane receptor CD33 expression (Gonzalez et al. 2012). However, a clear link between mitochondrial stress protein Hsp60 released from monocyte cells and endothelium inflammation has not yet been established.
Secretion of Hsp60 into culture medium
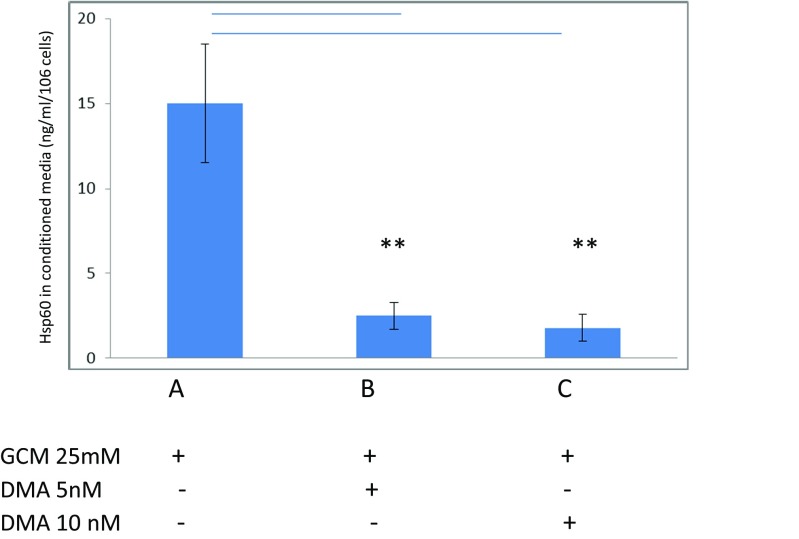
To determine if the increase in Hsp60 protein observed in THP-1 cells grown under hyperglycaemic conditions was actively secreted into the culture media, Hsp60 levels in conditioned media obtained fromTHP-1 monocyte cells grown in the presence of 5 and 25 mM glucose were measured using an anti-Hsp60 ELISA. There was no detectable Hsp60 protein in GCM 5 mM. This was consistent with the MTT and Western blot data which indicated that under normal growth conditions, the THP-1 monocyte cells were not subjected to mitochondrial stress and that at the basal levels of Hsp60 expression, there was no detectable release into the culture medium. However, the GCM 25 mM obtained from THP-1 cells that were exposed to 25 mM glucose were shown to contain 15 ± 3.5 ng/ml/106 cells of Hsp60 protein (Fig. 3 A). Interestingly, the GCM 25 mM also contained TNF-α (3.2 ± 1.2 pg/ml). The presence of TNF-α in the GCM 25 mM was not unexpected as high glucose-induced TNF-α secretion from monocyte cells has been previously documented (Gonzalez et al. 2012; Shanmugam et al. 2003).
Fig. 3.
Hsp60 detected in glucose-conditioned media (GCM) by anti-Hsp60 ELISA. The GCM (25 mM) contained 15 ± 3.5 ng/ml/106 cells of Hsp60 protein (lane A). In the presence of DMA (at 5 or 10 nM), there was an 83–88% decrease in Hsp60 detected compared to levels seen in GCM (25 mM) (lanes B and C, respectively). The data is representative of N = 3 independent experiments. The error bars represent ±S.E.M. (**) represents a statistically significant value (p < 0.005)
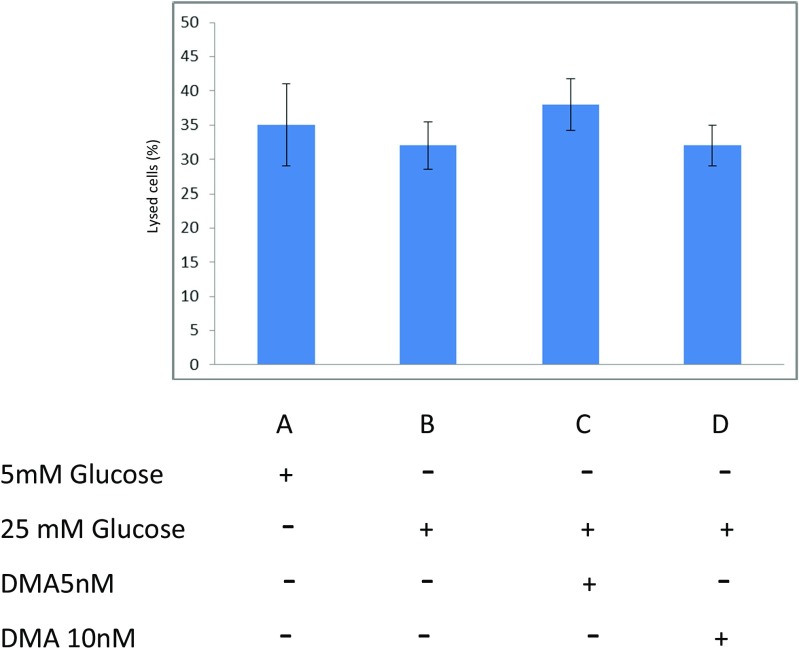
Interestingly, when THP1 monocyte cells that were grown in the presence of 25 mM glucose were treated with the exosomal inhibitor DMA at either 5 or 10 nM, there was a significant decrease (p < 0.005) in Hsp60 protein detected in the GCM 25 mM (Fig. 3 B and C). The LDH assay data indicated that cells grown in the presence of 25 mM glucose had similar levels of cell lysis to cells grown in 5 mM glucose (Fig. 4 A and B) and that treatment of THP cells with DMA did not lead to any adverse effects (Fig. 4 C and D). This indicated that the secretion of Hsp60 from THP-1 cells grown under hyperglycaemic conditions was indeed an active process involving exosomes, rather than non-specific leakage from the cells due to cell breakage and that the THP-1 cells maintained their plasma membrane integrity when treated with DMA. Extracellular release of Hsp60 from a variety of mammalian cell types via exosomes has been well-documented (Campanella et al. 2015; Gupta and Knowlton 2007; Malik et al. 2013). In our experiments, active secretion of Hsp60 was not observed in THP-1 cell grown in the presence of 5 mM glucose. This, however, contrasts to a study using cultured human tumour cell lines H292, A549 and K562 which demonstrated that Hsp60 could be actively secreted into the growth medium in unstressed tumour cells (Merendino et al. 2010). The reason for this discrepancy is not yet known.
Fig. 4.
LDH assays of THP-1 cells grown in the presence of 5 and 25 mM glucose which were exposed to DMA (5 or 10 nM). The percentage of LDH content (relative to maximum control samples) was interpreted as the percentage of lysed cells on the assumption that every dead cell released equal amounts of LDH. The data is representative of N = 3 independent experiments. The error bars represent ±S.E.M.
Effect of THP-1-derived conditioned media on the expression of TNF-α from endothelial cells
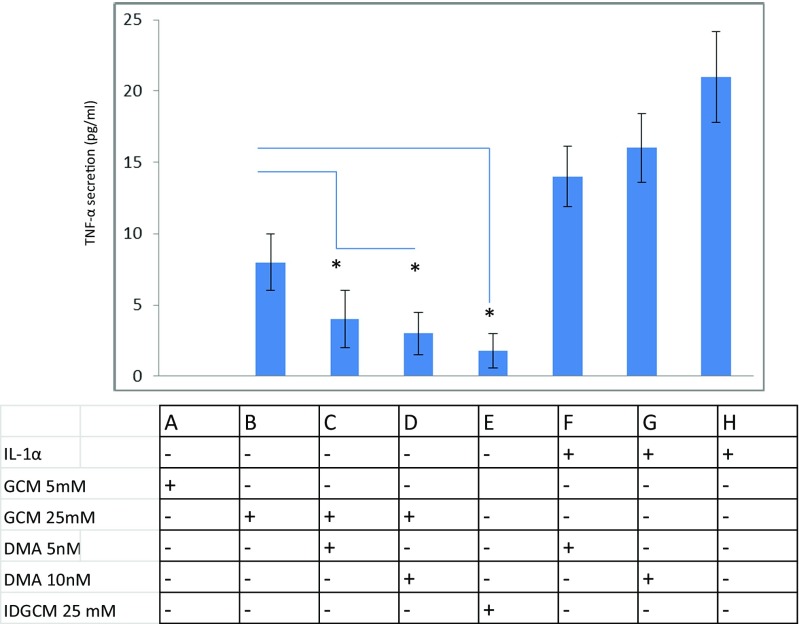
To determine if Hsp60 secreted from THP-1 monocyte cells was able to illicit an inflammatory response from endothelial cells, the human endothelial cell line HUVEC was exposed to conditioned medium obtained from the THP-1 monocyte cells grown in the presence of glucose at 5 and 25 mM. After a 3-day growth period, the level of TNF-α secreted into media was measured using an anti-TNF-α ELISA. There was no detectable TNF-α from HUVEC cells when cells were exposed to GCM 5 mM (Fig. 5 A). Exposing HUVEC cells to THP-1 cell-derived GCM 25 mM resulted in the secretion of 8 ± 2 pg/ml of TNF-α (Fig. 5 B). Interestingly, the levels of TNF-α secreted were significantly reduced (p < 0.05) when the GCM obtained from THP-1 cells grown in 25 mM glucose was treated with either 5 or 10 nM DMA (Fig. 5 C and D). Since DMA is an inhibitor of the exosomal protein secretion pathway (Lyu et al. 2015), this suggested that the compound responsible for triggering the inflammatory response from HUVEC cells was actively secreted via the exosomal pathway from the THP-1 cells. We were unable to detect any Hsp60 present in the glucose (25 mM)-conditioned media after the hsp60 immuno-depletion procedure. When this Hsp60 immuno-deleted media was exposed to HUVEC cells, the TNF-α secretion response of the HUVEC cells was significantly reduced (p < 0.05) (Fig. 5 E). This is a critical result in support of our central hypothesis that monocyte-derived mitochondrial Hsp60 (actively secreted from cells under hyperglycaemic conditions) is capable of inducing an inflammatory response in endothelial cells. Exposure of HUVEC cells to interleukin-1α (IL-1α) led to a robust TNF-α response (Fig. 5 H). This indicated that the HUVEC cells used during this investigation were immune responsive. IL-1α was used as a positive control for this study as it had been shown to be an effective inducer of the expression and secretion of TNF-α from HUVEC endothelial cells (Merendino et al. 2010). The exosomal inhibitor DMA made no difference to the ability of HUVEC cells to respond to IL-1α (Fig. 5 F and G).
Fig. 5.
TNF-α secretion from HUVEC cells as measured by anti-TNF-α ELISA after incubation with GCM from THP-1 cells. When HUVEC cells were exposed to GCM (5 mM), no TNF-α was detected (A); when HUVEC were exposed to GCM (25 mM), 8 ± 2 pg/ml of TNF-α was detected (B); when cells were exposed to GCM (25 mM) in the presence of 5 and 10 nM DMA, there was a significant reduction in the amount of TNF-a secreted (p < 0.05) (C and D, respectively); when HUVEC cells were exposed to Hsp60 immuno-depleted GCM (25 mM), there was a significant reduction in the amount of TNF-α secreted (p < 0.05) (E); responsiveness of the HUVEC cells to IL-1α is shown in (H); DMA had no effect on the TNF-α responsiveness of HUVEC cells to stimulation by IL-1α (F and G). The data is representative of N = 3 independent experiments. Error bars represent ±S.E.M. (*) represents a statistically significant value (p < 0.05)
The ability of the conditioned media obtained from THP-1 cells grown in 25 mM glucose (which had been immune-depleted with anti-Hsp60 antibodies prior to adding to HUVEC cells) to significantly reduce the TNF-α secretion clearly demonstrated that it was indeed the Hsp60 present in conditioned media from THP-1 cells subjected to hyperglycaemia that is the trigger factor for the TNF-α pro-inflammatory response observed in HUVEC cells in this study. Whilst it is possible that the TNF-α detected in the conditioned media of THP-1 cells gown in the presence of high glucose may have contributed to the TNF-α secreted from HUVEC endothelial cells exposed to high glucose-conditioned media from THP-1 cells, it was considered unlikely given the documented inability of TNF-α to activate p38MAPK signalling pathway (Said et al. 2002). Although this study did not investigate the receptor being activated by the response documented, extracellular Hsp60 has been shown to induce inflammation through activating and up-regulating TLRs in cardiomyocytes (Tian et al. 2013). It would be interesting to see if Hsp60 produced by THP-1 monocytes could also modulate the expression of TLRs on HUVEC cells. The ability of Hsp60 to activate the expression of TNF-α via TLR4-p38 MAPK receptor mediated signal transduction has been documented (Zhao et al. 2015b). Interestingly, Hsp60 has been also shown to play a regulatory role in IL-1β-induced microglial inflammation via TLR4-p38 MAPK axis (Swaroop et al. 2016). It is tempting to speculate that the TLR-p38 MAPK signalling pathway may also be in operation in the system described in this study but will require further investigation to determine if it is indeed responsible for the monocyte-derived Hsp60 activation of the endothelial TNF-α inflammatory response.
Concluding remarks
Monocyte endothelial interactions are recognised as an important early event trigger for a number of vascular pathologies, including atherosclerosis. Atherosclerosis is a complex process involving the binding and infiltration of monocytes through the endothelium (Daiber et al. 2016). The binding of monocytes to the endothelium is thought to be initiated by the expression of VCAM-1, ICAM-1 and E-selectins as a result of inflammatory activation of the endothelium (Grenon et al. 2012). The molecular triggers responsible for this non-resolved inflammation of the endothelium are still not fully understood. The data presented in this study show that Hsp60 released from monocytes exposed to hyperglycaemic conditions is capable of modulating the TNF-α (inflammatory) response of endothelial cells, suggesting that mitochondrial Hsp60 in monocyte-endothelial interactions in vascular disease warrant further investigation.
References
- Almanzar G, Ollinger R, Leuenberger J, Onestingel E, Rantner B, Zehm S, Cardini B, van der Zee R, Grundtman C, Wick G. Autoreactive HSP60 epitope-specific T-cells in early human atherosclerotic lesions. J Autoimmun. 2012;39:441–450. doi: 10.1016/j.jaut.2012.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Asea AAA, De Maio A (2007) Heat shock proteins: potent mediators of inflammation and immunity: potent mediators of inflammation and immunity. Springer
- Bassan M, Zamostiano R, Giladi E, Davidson A, Wollman Y, Pitman J, Hauser J, Brenneman DE, Gozes I. The identification of secreted heat shock 60-like protein from rat glial cells and a human neuroblastoma cell line. Neurosci Lett. 1998;250:37–40. doi: 10.1016/S0304-3940(98)00428-5. [DOI] [PubMed] [Google Scholar]
- Cabiscol E, Bellí G, Tamarit J, Echave P, Herrero E, Ros J. Mitochondrial Hsp60, resistance to oxidative stress, and the labile iron pool are closely connected in Saccharomyces cerevisiae. J Biol Chem. 2002;277:44531–44538. doi: 10.1074/jbc.M206525200. [DOI] [PubMed] [Google Scholar]
- Campanella C, D'Anneo A, Gammazza AM, Bavisotto CC, Barone R, Emanuele S, Lo Cascio F, Mocciaro E, Fais S, De Macario EC, et al. The histone deacetylase inhibitor SAHA induces HSP60 nitration and its extracellular release by exosomal vesicles in human lung-derived carcinoma cells. Oncotarget. 2015;7:28849–28867. doi: 10.18632/oncotarget.6680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen W, Syldath U, Bellmann K, Burkart V, Kolb H. Human 60-kDa heat-shock protein: a danger signal to the innate immune system. J Immunol. 1999;162:3212–3219. [PubMed] [Google Scholar]
- Daiber A, Steven S, Weber A, Shuvaev VV, Muzykantov VR, Laher I, Li H, Lamas S, Munzel T. Targeting vascular (endothelial) dysfunction. Br J Pharmacol. 2016;174:1591–1619. doi: 10.1111/bph.13517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- D'Souza SM, Brown IR. Constitutive expression of heat shock proteins Hsp90, Hsc70, Hsp70 and Hsp60 in neural and non-neural tissues of the rat during postnatal development. Cell Stress Chaperones. 1998;3:188–199. doi: 10.1379/1466-1268(1998)003<0188:CEOHSP>2.3.CO;2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fiorentino TV, Prioletta A, Zuo P, Folli F. Hyperglycemia-induced oxidative stress and its role in diabetes mellitus related cardiovascular diseases. Curr Pharm Des. 2013;19:5695–5703. doi: 10.2174/1381612811319320005. [DOI] [PubMed] [Google Scholar]
- Frostegard J. Immunity, atherosclerosis and cardiovascular disease. BMC Med. 2013;11:117. doi: 10.1186/1741-7015-11-117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giacco F, Brownlee M. Oxidative stress and diabetic complications. Circ Res. 2010;107:1058–1070. doi: 10.1161/CIRCRESAHA.110.223545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gonzalez Y, Herrera MT, Soldevila G, Garcia-Garcia L, Fabián G, Pérez-Armendariz EM, Bobadilla K, Guzmán-Beltrán S, Sada E, Torres M. High glucose concentrations induce TNF-α production through the down-regulation of CD33 in primary human monocytes. BMC Immunol. 2012;13:1–14. doi: 10.1186/1471-2172-13-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grenon SM, Aguado-Zuniga J, Hatton JP, Owens CD, Conte MS, Hughes-Fulford M. Effects of fatty acids on endothelial cells: inflammation and monocyte adhesion. J Surg Res. 2012;177:e35–e43. doi: 10.1016/j.jss.2012.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grundtman C, Kreutmayer SB, Almanzar G, Wick MC, Wick G. Heat shock protein 60 and immune inflammatory responses in atherosclerosis. Arterioscler Thromb Vasc Biol. 2011;31:960–968. doi: 10.1161/ATVBAHA.110.217877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gupta S, Knowlton AA. HSP60 trafficking in adult cardiac myocytes: role of the exosomal pathway. Am J Physiol Heart Circ Physiol. 2007;292:H3052–H3056. doi: 10.1152/ajpheart.01355.2006. [DOI] [PubMed] [Google Scholar]
- Haak JL, Buettner GR, Spitz DR, Kregel KC. Aging augments mitochondrial susceptibility to heat stress. Am J Physiol Regul Integr Comp Physiol. 2009;296:R812–R820. doi: 10.1152/ajpregu.90708.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hall L, Martinus RD. Hyperglycaemia and oxidative stress upregulate HSP60 & HSP70 expression in HeLa cells. SpringerPlus. 2013;2:1–10. doi: 10.1186/2193-1801-2-431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jovaisaite V, Mouchiroud L, Auwerx J. The mitochondrial unfolded protein response, a conserved stress response pathway with implications in health and disease. J Exp Biol. 2014;217:137–143. doi: 10.1242/jeb.090738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Juwono J, Martinus RD. Does Hsp60 provide a link between mitochondrial stress and inflammation in diabetes mellitus? J Diab Res. 2016;2016:8017571. doi: 10.1155/2016/8017571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- King RJ, Grant PJ. Diabetes and cardiovascular disease: pathophysiology of a life-threatening epidemic. Herz. 2016;41:184–192. doi: 10.1007/s00059-016-4414-8. [DOI] [PubMed] [Google Scholar]
- Lyu L, Wang H, Li B, Qin Q, Qi L, Nagarkatti M, Nagarkatti P, Janicki JS, Wang XL, Cui T. A critical role of cardiac fibroblast-derived exosomes in activating renin angiotensin system in cardiomyocytes. J Mol Cell Cardiol. 2015;89:268–279. doi: 10.1016/j.yjmcc.2015.10.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malik ZA, Kott KS, Poe AJ, Kuo T, Chen L, Ferrara KW, Knowlton AA. Cardiac myocyte exosomes: stability, HSP60, and proteomics. Am J Physiol Heart Circ Physiol. 2013;304:H954–H965. doi: 10.1152/ajpheart.00835.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mandal K, Foteinos G, Jahangiri M, Xu Q. Role of antiheat shock protein 60 autoantibodies in atherosclerosis. Lupus. 2005;14:742–746. doi: 10.1191/0961203305lu2212oa. [DOI] [PubMed] [Google Scholar]
- Martinus RD, Garth GP, Webster TL, Cartwright P, Naylor DJ, Hoj PB, Hoogenraad NJ. Selective induction of mitochondrial chaperones in response to loss of the mitochondrial genome. Eur J Biochem/FEBS. 1996;240:98–103. doi: 10.1111/j.1432-1033.1996.0098h.x. [DOI] [PubMed] [Google Scholar]
- Merendino AM, Bucchieri F, Campanella C, Marcianò V, Ribbene A, David S, Zummo G, Burgio G, Corona DFV, de Macario EC, et al. Hsp60 is actively secreted by human tumor cells. PLoS One. 2010;5:e9247. doi: 10.1371/journal.pone.0009247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pockley AG. Heat shock proteins as regulators of the immune response. Lancet (London, England) 2003;362:469–476. doi: 10.1016/S0140-6736(03)14075-5. [DOI] [PubMed] [Google Scholar]
- Rabczynski M, Fiodorenko-Dumas Z, Adamiec R, Paprocka-Borowicz M, Dumas I. Role of anti-HSP 60/65 antibodies in atherogenesis in patients with type 2 diabetes and lower limb ischemia. J Physiol Pharmacol. 2012;63:691–696. [PubMed] [Google Scholar]
- Said FA, Werts C, Elalamy I, Couetil JP, Jacquemin C, Hatmi M. TNF-alpha, inefficient by itself, potentiates IL-1beta-induced PGHS-2 expression in human pulmonary microvascular endothelial cells: requirement of NF-kappaB and p38 MAPK pathways. Br J Pharmacol. 2002;136:1005–1014. doi: 10.1038/sj.bjp.0704811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shah MS, Brownlee M. Molecular and cellular mechanisms of cardiovascular disorders in diabetes. Circ Res. 2016;118:1808–1829. doi: 10.1161/CIRCRESAHA.116.306923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shanmugam N, Reddy MA, Guha M, Natarajan R. High glucose-induced expression of proinflammatory cytokine and chemokine genes in monocytic cells. Diabetes. 2003;52:1256–1264. doi: 10.2337/diabetes.52.5.1256. [DOI] [PubMed] [Google Scholar]
- Swaroop S, Sengupta N, Suryawanshi AR, Adlakha YK, Basu A. HSP60 plays a regulatory role in IL-1beta-induced microglial inflammation via TLR4-p38 MAPK axis. J Neuroinflammation. 2016;13:27. doi: 10.1186/s12974-016-0486-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tabas I, García-Cardeña G, Owens GK. Recent insights into the cellular biology of atherosclerosis. J Cell Biol. 2015;209:13–22. doi: 10.1083/jcb.201412052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tian J, Guo X, Liu X-M, Liu L, Weng Q-F, Dong S-J, Knowlton AA, Yuan W-J, Lin L. Extracellular HSP60 induces inflammation through activating and up-regulating TLRs in cardiomyocytes. Cardiovasc Res. 2013;98:391–401. doi: 10.1093/cvr/cvt047. [DOI] [PubMed] [Google Scholar]
- Tsuei A-C, Martinus RD. Metformin induced expression of Hsp60 in human THP-1 monocyte cells. Cell Stress Chaperones. 2012;17:23–28. doi: 10.1007/s12192-011-0282-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verrijn Stuart AA, de Jager W, Klein MR, Teklenburg G, Nuboer R, Hoorweg JJ, de Vroede MA, de Kruijff I, Fick M, Schroor EJ, et al. Recognition of heat shock protein 60 epitopes in children with type 1 diabetes. Diabetes Metab Res Rev. 2012;28:527–534. doi: 10.1002/dmrr.2306. [DOI] [PubMed] [Google Scholar]
- Wang AT, Prinsep MR, Martinus RD. Pterocellin A isolated from marine bryozoan Pterocella vesiculosa is cytotoxic to human HeLa cells via mitochondrial apoptotic processes. Spring. 2016;5:742. doi: 10.1186/s40064-016-2397-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yuan J, Dunn P, Martinus RD. Detection of Hsp60 in saliva and serum from type 2 diabetic and non-diabetic control subjects. Cell Stress Chaperones. 2011;16:689–693. doi: 10.1007/s12192-011-0281-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao Y, Zhang C, Wei X, Li P, Cui Y, Qin Y, Wei X, Jin M, Kohama K, Gao Y. Heat shock protein 60 stimulates the migration of vascular smooth muscle cells via Toll-like receptor 4 and ERK MAPK activation. Sci Rep. 2015;5:15352. doi: 10.1038/srep15352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao Y, Zhang C, Wei X, Li P, Cui Y, Qin Y, Wei X, Jin M, Kohama K, Gao Y. Heat shock protein 60 stimulates the migration of vascular smooth muscle cells via Toll-like receptor 4 and ERK MAPK activation. Sci Rep. 2015;5:15352. doi: 10.1038/srep15352. [DOI] [PMC free article] [PubMed] [Google Scholar]