Abstract
Supplementation with B vitamins (vitamin B9(folic acid), vitamin B12 and vitamin B6) lowers blood total homocysteine (tHcy) concentrations by about 25% and reduces the relative risk of stroke overall by about 10% (risk ratio (RR) 0.90, 95% CI 0.82 to 0.99) compared with placebo. Homocysteine-lowering interventions have no significant effect on myocardial infarction, death from any cause or adverse outcomes. Factors that appear to modify the effect of B vitamins on stroke risk include low folic acid status, high tHcy, high cyanocobalamin dose in patients with impaired renal function and concurrent antiplatelet therapy. In regions with increasing levels or established policies of population folate supplementation, evidence from observational genetic epidemiological studies and randomised controlled clinical trials is concordant in suggesting an absence of benefit from lowering of homocysteine with folic acid for prevention of stroke. Clinical trials indicate that in countries which mandate folic acid fortification of food, folic acid supplementation has no significant effect on reducing stroke risk (RR 1.05, 95% CI 0.90 to 1.23). However, in countries without mandatory folic acid food fortification, folic acid supplementation reduces the risk of stroke by about 15% (RR 0.85, 95% CI 0.77 to 0.94). Folic acid alone or in combination with minimal cyanocobalamin (≤0.05 mg/day) is associated with an even greater reduction in risk of future stroke by 25% (RR 0.75, 95% CI 0.66 to 0.86), whereas the combination of folic acid and a higher dose of cyanocobalamin (≥0.4 mg/day) is not associated with a reduced risk of future stroke (RR 0.95, 95% CI 0.86 to 1.05). The lack of benefit of folic acid plus higher doses of cyanocobalamin (≥0.4 mg/day) was observed in trials which all included participants with chronic kidney disease. Because metabolic B12 deficiency is very common and usually not diagnosed, future randomised trials of homocysteine-lowering interventions for stroke prevention should probably test a combination of folic acid and methylcobalamin or hydroxocobalamin instead of cyanocobalamin, and perhaps vitamin B6.
Keywords: B vitamins, folic acid, homocysteine, stroke prevention, meta-analysis
The homocysteine hypothesis
The hypothesis that B vitamin therapy (folic acid, vitamin B6 and vitamin B12) may prevent stroke emerged more than 15 years ago after early case and epidemiological studies reported an association between high plasma concentrations of total homocysteine (tHcy) and incident cardiovascular diseases,1–7 laboratory studies reported atherogenic and thrombogenic properties of high tHcy,8–10 and randomised controlled trials (RCTs) showed that supplementation with B vitamins lowers tHcy.11
What is homocysteine?
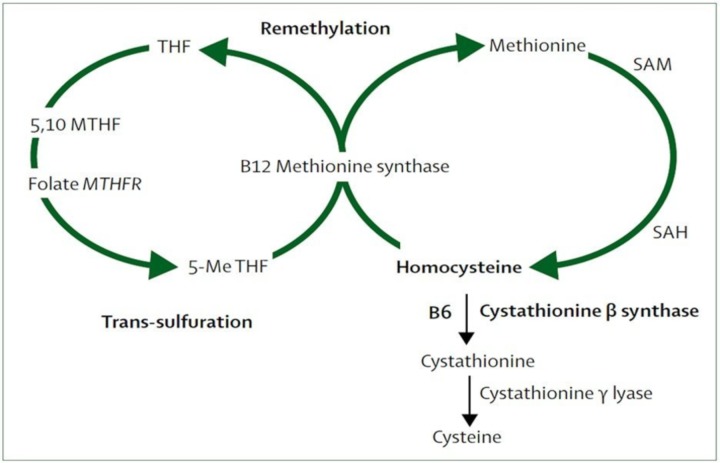
Homocysteine is derived from the essential amino acid methionine, which is abundant in animal sources of protein. Methionine is converted to homocysteine via the intermediate S-adenosylmethionine, which acts as a methyl donor, as in several other biochemical processes such as DNA methylation and creatine and phosphatidylcholine synthesis. Homocysteine can be remethylated back to methionine or converted to cysteine by transsulfuration (figure 1).12
Figure 1.
Homocysteine metabolism and the methionine and folate cycles. 5,10-MTHF, 5,10-methylenetetrahydrofolate; 5-Me THF, 5-methyltetrahydrofolate; B6, pyridoxine; B12, cobalamin; MTHFR, methylenetetrahydrofolate reductase; SAH, S-adenosylhomocysteine; SAM, S-adenosylmethionine; THF, tetrahydrofolate. Reproduced with permission from Elsevier.12
Factors influencing homocysteine concentration
Plasma tHcy reflects free-bound and protein-bound homocysteine, its dimer homocysteine and cysteine-homocysteine mixed disulfide.13 The causes of high tHcy are listed in box 1.14–19
Box 1. Causes of hyperhomocysteinemia.
Genetic factors
5,10-Methylenetetrahydrofolate reductase C677T homozygosity (common).16–18
Cystathionine gamma-lyase (common).14
Heterozygosity for cystathionine β synthase (CBS) defects (uncommon).
Homocystinuria (homozygous CBS) (very rare).19
Physiological factors
Increased age.
Male sex.
Menopause.
Reduced glomerular filtration rate.
Increased muscle mass.
Lifestyle factors
Reduced B vitamin intake (folate, vitamin B12, vitamin B6).
Smoking.
Coffee.
Alcohol consumption.
Physical inactivity.
Disease states
B vitamin deficiency (due to malabsorption, including inflammatory bowel disease).
Renal impairment.
Hypothyroidism.
Diabetes mellitus.
Psoriasis.
Malignancies.
Drugs
Lipid-lowering*—cholestyramine, nicotinic acid, fibric acid derivatives (eg, fenofibrate).
Anticonvulsants—phenytoin, carbamazepine.
Sex hormones—androgens.
Antirheumatic drugs—methotrexate.
Other—ciclosporin, diuretics, levodopa, methionine loading (oral, intravenous, peritoneal), theophylline, trimethoprim.
*HMG-CoA reductase inhibitors have no effect on total plasma homocysteine levels.
Genetic mutations
The cause of inherited hyperhomocysteinaemia can be broadly separated into remethylation disturbances (methylenetetrahydrofolate reductase (MTHFR) deficiency and vitamin B12 and folate-related defects) and transsulfuration defects (cystathionine β-synthase deficiency and cystathionine gamma-lyase deficiency).15–19
Remethylation disturbances
In order for homocysteine to be remethylated to methionine, it requires a methyl donor in the form of 5-methyl tetrahydrofolate, which is produced from 5,10-methylenetetrahydrofolate in a reaction catalysed by 5,10-MTHFR. About 10% of the population is homozygous for a point mutation (C-to-T substitution at nucleotide 677 (677 C→T polymorphism)) in the coding region of the gene for MTHFR. The TT polymorphism is associated with a thermolabile variant of MTHFR that has reduced activity (by about 50%) and thus reduced production of the methyl donor 5-methyl tetrahydrofolate (akin to low dietary intake of folate), which increases tHcy moderately by about 20% (ie, about 2 µmol/L).16–18
Transsulfuration defects
The most common, although rare, genetic cause of severe hyperhomocysteinaemia (>100 µmol/L) and homocystinuria is homozygous deficiency of cystathionine β-synthase, which is inherited as an autosomal recessive trait in about 1 in 100 000 live births.19
Nutritional deficiencies in B vitamin cofactors required for homocysteine metabolism
The metabolic (methylation) pathway that converts homocysteine to methionine is catalysed by methionine synthase and requires folic acid (5-methyl tetrahydrofolate) and vitamin B12 (cobalamin) as an essential cofactor (figure 1). The metabolic (transsulfuration) pathway that converts homocysteine to cystathionine is catalysed by cystathionine β-synthase and requires vitamin B6 as a cofactor (figure 1). Hence, the total plasma concentration of homocysteine (tHcy) is influenced by blood concentrations of the B vitamins folic acid (B9), cyanocobalamin (B12) and pyridoxine (B6).
Other causes of hyperhomocysteinaemia
Other factors affecting tHcy include increasing age, smoking, hypothyroidism, renal impairment, malabsorption of vitamin B12 and numerous other diseases and drugs (box 1). Fasting tHcy rises as serum creatinine rises because of impaired metabolism and clearance of homocysteine by the kidney.
Evidence for homocysteine as a causal risk factor for stroke
A causal association between a risk factor, such as tHcy, and a disease, such as stroke, is inferred by several factors, including the following:
Temporal sequence of exposure to the risk factor first, followed then by the occurrence of the disease.
Independence from confounding factors (known and unknown) that are associated with both the exposure and the disease but are not on the causal pathway between the exposure and the disease (eg, are not mediators of the causal effect).
Strength of the association (high relative risk or relative odds).
A dose–response relationship (the greater degree of exposure to the risk factor, the greater the frequency or severity of the disease).
Consistency of the association among different studies and populations.
Biological and epidemiological plausibility.
A decline in the incidence of the disease after experimental removal or reduction of the risk factor (ideally in RCTs).20
Observational epidemiological studies
Systematic reviews of observational (cohort and case–control) studies show a strong, positive and dose-related association between the serum concentration of tHcy and the risk of stroke, independent of other vascular risk factors.6 7 An increase of 3 μmol/L in tHcy was associated with an increase in risk of stroke by about 24% and increased risk of myocardial infarction by about 15% after a mean follow-up of 7.3 years and after adjusting for confounding by other cardiovascular risk factors.6 7 However, absent other evidence, reverse causation and residual confounding, by failure to measure and adjust for confounding factors that increase both tHcy and stroke risk but are not on the causal pathway between tHcy and stroke (eg, smoking, lower socioeconomic class and renal impairment), might be possible explanations for the observed association.
Genetic epidemiological studies
A meta-analysis of 111 literature-based genetic studies examining the association between MTHFR and stroke found that people who are homozygous for the T allele (TT) of the MTHFR 677 C→T polymorphism have a significantly greater mean tHcy (weighted mean difference 1.93 μmol/L; 95% CI 1.38 to 2.47) and also a significantly greater risk of stroke (OR1.26, 95% CI 1.14 to 1.40) than people who are unaffected and are homozygous for the CC genotype.21 The greater risk of stroke conferred by MTHFR-TT was in proportion to the difference in tHcy that can be attributed to the polymorphism. The effects were greatest in regions with low dietary folate consumption.
As the 677 C→T polymorphism is distributed by random assortment of alleles during gamete production and fertilisation, this is an example of natural (Mendelian) randomisation whereby people are randomly allocated to groups with higher (TT) or lower tHcy (CC). The observed triangular association between MTHFR genotype, tHcy and stroke supports the hypothesis that increased concentrations of tHcy (by about 2 µmol/L) may be causal in increasing the odds of stroke (by about a quarter). The similar estimate of risk of stroke obtained from the meta-analysis of genetic association studies21 and from meta-analyses of non-genetic observational studies,6 7 which have different sources of error, also supports a causal association.
However, there are caveats to these literature-based genetic meta-analyses.21 The results could have been affected by small-study bias, as only two studies had more than 400 stroke cases, and the effect of the MTHFR 677C→T variant on tHcy could have been modified by the prevailing concentrations of folic acid, with high folate being associated with a reduced effect of MTHFR 677C→T on tHcy.
A subsequent meta-analysis of 67 data sets from genetic studies involving 53 643 individuals without cardiovascular disease reported that the effect of the MTHFR 677C→T variant on tHcy was larger in regions with low folate intake (Asia; difference between individuals with TT vs CC genotype, 3.12 μmol/L, 95% CI 2.23 to 4.01) than in areas with high folate intake due to folate fortification (America, Australia and New Zealand, 0.13 μmol/L, 95% CI −0.85 to 1.11).22 A concurrent and complementary meta-analysis of 140 data sets from 101 genetic studies including 20 885 stroke events reported that the effect of the MTHFR 677C→T variant on the OR for stroke was also higher in regions with low folate intake (Asia, OR 1.68, 95% CI 1.44 to 1.97) than in in areas with high folate intake due to folate fortification (America, Australia and New Zealand, OR 1.03, 95% CI 0.84 to 1.25).22
Clinical trials
Effect of B vitamin supplementation on plasma homocysteine concentration
A meta-analysis of 12 clinical trials involving 1114 individuals without renal failure, and most with normal folate status, showed that between 0.5 mg and 5 mg of folic acid daily lowered tHcy concentrations by 25% (95% CI 23% to 28%).11
The minimum effective daily dose of folic acid for achieving maximal homocysteine-lowering efficacy was about 0.5 mg. Higher dose of folic acid, up to 5 mg daily, had similar effects on tHcy to lower doses, in the range of 0.5–5 mg folic acid daily. Vitamin B12 (0.02–1 mg daily further reduced tHcy levels by about 7% (95% CI 3% to 10%). Vitamin B6 (2–50 mg daily) and betaine (a methyl group donor involved in the metabolism of methionine) were less effective.11 The effect of B vitamin supplementation was more pronounced in people with higher tHcy and lower folate concentrations before treatment.11
Effect of B vitamin supplementation on stroke and other cardiovascular events
A recently updated systematic review for the Cochrane Library identified 15 RCTs that assessed the effects of homocysteine-lowering interventions, in the form of supplements of vitamins B6, B9 or B12 given alone or in combination, for preventing cardiovascular events over 1–7.3 years of follow-up, in 71 422 participants (excluding patients with end-stage renal disease).23 Nine trials (60%) had low risk of bias.
Stroke
A meta-analysis, using a random-effects model (which is more conservative than a fixed-effects model), of the 10 RCTs that compared homocysteine-lowering interventions with placebo in a total of 44 224 participants showed homocysteine-lowering treatment was associated with a reduced risk of stroke (4.3% homocysteine-lowering vs 5.1% placebo, risk ratio (RR) 0.90, 95% CI 0.82 to 0.99, I2=8%).23
In two RCTs that compared homocysteine-lowering treatment at high dose versus low dose in 3929 participants, there were uncertain effects of high doses of homocysteine-lowering interventions versus low dose on stroke (10.8% high dose vs 11.2% low dose, RR 0.90, 95% CI 0.66 to 1.22, I2=72%).23
In one RCT that compared homocysteine-lowering treatment (folic acid) plus antihypertensive therapy (enalapril) versus antihypertensive therapy (enalapril) in 20 702 participants, homocysteine-lowering treatment was associated with a reduced risk of stroke (2.7% homocysteine-lowering vs 3.4% placebo, RR 0.79, 95% CI 0.68 to 0.93, I2=8%).23
Other events
There were no differences in the effects of homocysteine-lowering interventions compared with placebo on myocardial infarction (7.1% vs 6.0%; RR 1.02, 95% CI 0.95 to 1.10, I2=0%; 12 trials, n=46 699), and compared with other controls on death from any cause (11.7% vs 12.3%; RR 1.01, 95% CI 0.96 to 1.06, I2=0%; 11 trials, n=44 817) or serious adverse events such as cancer (8.3% vs 8.5%; RR 1.07, 95% CI 1.00 to 1.14, I2=0%; 8 trials, n=35 788).23
Other recent meta-analyses have produced similar results.24–26
Modifiers of the effect of B vitamin supplementation on stroke
Folic acid fortification of food
A recent systematic review identified 13 RCTs of folic acid therapy in which stroke was reported as an outcome and most participants (>50%) in a trial resided in countries without mandatory fortification of food with folic acid.27 At total of 65 812 participants were enrolled in 13 trials; 12 trials included individuals with pre-existing conditions: stroke (one trial), coronary heart disease (five trials), cardiovascular disease (one trial), end-stage renal disease or advanced chronic kidney disease (three trials), hypertension (one trial) and oesophageal dysplasia (one trial), while one trial included individuals living in high altitude. Folic acid alone or the combination of folic acid and minimal cyanocobalamin (≤0.05 mg/day) was used in an active treatment group in eight trials, whereas the combination of folic acid and cyanocobalamin (≥0.4 mg/day) was used in an active treatment group in five trials. Three trials excluded participants with chronic kidney disease while other trials included some participants with chronic kidney disease.
A meta-analysis of these trials among people who were living in countries without mandatory folic acid fortification in food found that folic acid therapy was associated with a lower risk of any future stroke compared with control (RR 0.85, 95% CI 0.77 to 0.95; p=0.004; 3–8 fewer events per 1000 patients).27
Other meta-analyses have reported similar results.26 The latter meta-analysis of 22 RCTs comparing folic acid with placebo or very low dose control group for at least 6 months of intervention in a total of 82 723 participants reported that folic acid supplementation significantly reduced the stroke risk by about 10% (RR 0.89, 95% CI 0.84 to 0.96) and that the effect was greater in low folate regions (2 trials, n=24 020; Asia, RR 0.78, 95% CI 0.67 to 0.90) compared with high folate regions (7 trials, n=14 655; America, RR 1.05, 95% CI 0.90 to 1.23), and among patients without folic acid fortification (11 trials, n=49 957; RR 0.85, 95% CI 0.77 to 0.94) compared with those with folic acid fortification (7 trials, n=14 655; RR 1.05, 95% CI 0.90 to 1.23).26
Hyperhomocysteinaemia realising greater degrees of homocysteine reduction with B vitamins
Larger decreases in tHcy with B vitamin supplementation, as seen in populations with elevated tHcy due to insufficient folate intake, have been associated with greater reductions in stroke risk in Chinese adults with hypertension enrolled in the China Stroke Primary Prevention Trial (CSPPT),28 and greater reductions in risk of cardiovascular diseases in a meta-analysis of RCTs of folic acid supplementation.24
This effect probably underpins the observation in a stratified meta-analysis of RCTs comparing folic acid with placebo (or a very low dose control group) in patients without folic acid fortification that the beneficial effect of folic acid in preventing stroke was larger in participants with low baseline serum vitamin B12 levels (<384 pg/mL: relative risk: 0.78, 95% CI 0.68 to 0.89), as low vitamin B12 increases tHcy.26 However, it is important to acknowledge that methodological problems may compromise the sensitivity and specificity of assays measuring the serum vitamin B12 concentration; both false negative and false positive values are common with the use of the laboratory-reported lower limit of the normal range as a cut-off point for B12 deficiency.29 Further, as only about 25% of serum total B12 is in the active form of holotranscobalamin, total serum B12 determination is not sensitive for B12 deficiency.29 30 To establish the diagnosis of vitamin B12 deficiency in patients who have not received B12 treatment, it is recommended to measure holotranscobalamin or to measure functional or metabolic markers of B12 adequacy such as plasma methylmalonic acid or tHcy.29 30 The levels of both methylmalonic acid and tHcy are markedly elevated in the vast majority (>98%) of patients with clinical B12 deficiency. Metabolic B12 deficiency is an elevated level of methylmalonic acid or elevated tHcy in folate-replete individuals. An elevated level of methylmalonic acid is reasonably specific for vitamin B12 deficiency (but also moderately elevated in renal failure) and decreases with vitamin B12 therapy, whereas the level of serum tHcy is less specific (see other causes in box 1).
Like vitamin B12 deficiency, the MTHFR 677TT genotype is also associated with elevated tHcy concentrations. A substudy of the CSPPT recently reported an interactive effect of the MTHFR C677T gene variant and serum folate levels on the serum tHcy-lowering response to long-term folic acid supplementation.31 As in previous studies, the MTHFR 677TT genotype was associated with elevated tHcy levels, but it was mainly in participants with low folate intake and low serum folate concentrations. The study reported that the adverse effects of the TT genotype on tHcy levels were ameliorated by raising serum folate levels above the threshold (15 ng/mg or 34 nmol/L) with folic acid supplementation. The data also indicated that individuals with the TT genotype and low serum folate, by having the highest levels of tHcy, are also the subgroup likely to benefit most from folic acid therapy, particularly when serum folate levels can increase to the reported threshold of >34 nmol/L.31 The authors suggested that adequate nutrition (ie, folate intake) may be able to counteract genetic susceptibility for hyperhomocysteinaemia.
Cyanocobalamin toxicity in patients with impaired renal function
A meta-analysis of individual patient data from two large trials of B vitamin supplementation in patients with prior stroke and TIA (Vitamins Intervention for Stroke Prevention (VISP)32 and VITAmins TO Prevent Stroke (VITATOPS)33) and tabulated data from other trials found that patients with impaired renal function (estimated glomerular filtration rate (eGFR) <50 mL/min/1.73 m2) who were allocated to high-dose cyanocobalamin (at least 0.4 mg daily) did not benefit from therapy with B vitamins for the prevention of stroke (RR 1.04, 95% CI 0.84 to 1.27), whereas patients with normal renal function who were not exposed to high-dose cyanocobalamin (ie, no cyanocobalamin or 0.02 mg cyanocobalamin daily) did benefit significantly from B vitamin supplementation (RR 0.78, 95% CI 0.67 to 0.90; interaction p=0.03) (table 1).12
Table 1.
Effect of renal function and dose of cyanocobalamin on stroke reduction by B vitamins
| Trial | Dose of cyanocobalamin (μg) | Serum creatinine (μmol/L*) B vitamins | Control | Stroke HR (95% CI) |
P values |
| VISP† 32 | 400 | 99.9 (55.7) | 97.2 (47.7) | 1.0 (0.8 to 1.3) | 0.80 |
| VITATOPS‡ 33 | 500 | 92.4 (40.3) | 91.4 (34.6) | 0.92 (0.81 to 1.06) | 0.25 |
| DIVINe34 | 1000 | 141.4 (97.2) | 123.8 (79.6) | 6.6 (0.8 to 54.4) | 0.08 |
| SEARCH39 | 1000 | NA | NA | 1.02 (0.86 to 1.21) | NA |
| CSPPT35 | 0 | 65.95 (19.0) | 65.95 (19.0) | 0.79 (0.68 to 0.93) | 0.003 |
| CSPPT35 | 0 | 126.6 (72.7) | 130.6 (68.6) | 0.88 (0.33 to 2.36) | 0.81 |
| eGFR<60§ | |||||
| CSPPT35 | 0 | 64.7 (13.8) | 64.6 (13.6) | 0.79 (0.67 to 0.92) | 0.003 |
| Excluding eGFR<60§ | |||||
| NORVIT36 | 400 | 91 (27) | 91 (24) | 0.83 (0.47 to 1.47) | 0.52 |
| HOPE37 | 1000 | 88.4 (26.5) | 88.4 (26.5) | 0.75 (0.59 to 0.97) | 0.03 |
| SU.Fol.OM338 | 20 | 78.0 (70.0-88.0) | 78.0 (69.0–88.0) | 0.57 (0.33 to 0.97) | 0.04 |
Individual data were obtained from both VISP32 and VITATOPS33 trials. Reproduced with permission from Elsevier.12
*Data are presented as mean (SD), unless otherwise specified.
†Patients with an eGFR of less than 50 mL/min/1.73 m² had a mean eGFR rate (SD) of 38.7 mL/min/1.73 m² (9.2) (control 39.3 mL/min/1.73 m² (8.6); composite outcomes HR (95% CI) 1.01 (0.74 to 1.37; p=0.977)). Patients with an eGFR of 50 mL/min/1.73 m² or more had mean eGFR rate of 89.2 mL/min/1.73 m² (44.6) (control 87.3 (31.1); composite outcomes HR 0.92 (0.77 to 1.11; p=0.382)). Composite outcomes refer to myocardial infarction, stroke or death.
‡Patients with an eGFR of 50 mL/min/1.73 m² or less had a mean eGFR rate (SD) of 38.6 mL/min/1.73 m² (10.0) (control 38.3 (9.5); composite outcomes HR (95% CI) 0.88 (0.59 to 1.32; p=0.54)). Patients with an eGFR of more than 50 mL/min/1.73 m² had a mean eGFR rate of 80.2 (18.1) (control 80.9 (18.3); composite outcomes HR 0.82 (0.68 to 0.98; p=0.03)). Composite outcomes refer to myocardial infarction, stroke or death.
§mL/min/1.73 m².
CSPPT, China Stroke Primary Prevention Trial; eGFR, estimated glomerular filtration rate (mL/min/1.73 m²); NA, not applicable; VISP, Vitamins Intervention for Stroke Prevention; VITATOPS, VITAmins TO Prevent Stroke.
SEARCH: Study of the Effectiveness of Additional Reductions of Cholesterol and Homocysteine
Table 1 shows the relationship of renal function and dose of cyanocobalamin to reduction of stroke with B vitamins.12 32–39
A subsequent subgroup analysis of the meta-analysis of RCTs27 of folic acid therapy among 65 812 participants who were mainly living in countries without mandatory folic acid fortification (referred to in the above section on folic acid fortification of food) found that folic acid alone or the combination of folic acid and minimal cyanocobalamin (≤0.05 mg/day) was associated with a lower risk of future stroke (RR 0.75, 95% CI 0.66 to 0.86), whereas the combination of folic acid and a higher dose of cyanocobalamin (≥0.4 mg/day) was not associated with a lower risk of future stroke (RR 0.95, 95% CI 0.86 to 1.05).27 The lack of benefit of folic acid plus higher doses of cyanocobalamin (≥0.4 mg/day) was observed in trials which all included some participants with chronic kidney disease.27 In patients with diabetic nephropathy who were enrolled in the Diabetic Intervention with Vitamins to Improve Nephropathy (DIVINe) trial, B vitamins, which included cyanocobalamin in a dose of 1000 μg daily, accelerated the decline in renal function and doubled the risk of stroke and other cardiovascular events compared with placebo (HR 2.0, 95% CI 1.0 to 4.0).34 All the events occurred among participants with eGFR <50 mL/min/1.73 m2.34 In a subgroup analysis of the VISP trial, there was a significant benefit of B vitamins including cyanocobalamin among participants with an eGFR >46 mL/min/1.73 m2 (the lowest 10%) compared with individuals with renal impairment.32
These observations have generated the hypothesis that renal impairment in patients who have high levels of thiocyanate, cyanocobalamin or impaired decyanation of cyanocobalamin leads to adverse effects of cyanocobalamin, which offset the benefits of folic acid alone12 (box 2, adapted from ref 12). This hypothesis does not mean that individuals with renal impairment do not benefit from B vitamins in terms of stroke prevention; they are harmed when cyanocobalamin is added in high doses. For example, in the large Chinese trial (CSPPT), the benefit of folic acid monotherapy in preventing stroke was also found in participants with mild to moderate chronic kidney disease.35 Hence, if B vitamin preparations are to contain vitamin B12, methylcobalamin or hydroxocobalamin should be used, not cyanocobalamin, particularly in patients with renal impairment.
Box 2. Summary of the evidence regarding B vitamins for stroke prevention.
The overall results of previous trials of vitamin B therapy for the prevention of stroke might be the result of the increased risk of stroke and myocardial infarction in participants with impaired renal function, cancelling out the benefits observed in participants with good renal function.
Koyama and colleagues have reported that patients with renal failure have increased concentrations of thiocyanate and that coadministration of intravenous methylcobalamin and oral folate normalises hyperhomocysteinemia in haemodialysis patients.40 These findings, in addition to two plausible mechanisms for increased risk of cardiovascular events with exposure to thiocyanate—thiocyanate increases the oxidation of low-density lipoprotein and thiocyanate formation consumes hydrogen sulfide—suggest that cyanide from cyanocobalamin (or impaired decyanation of cyanocobalamin) in patients with impaired renal function might be harmful.
If the harm from B vitamins in the Diabetic Intervention with Vitamins to Improve Nephropathy (DIVINe) study34 was caused by folic acid, then it would be expected that in the CSPPT study35 patients with impaired renal function who were treated with folic acid alone would also have a higher risk of stroke and an increased decline in renal function. However, folic acid improved renal function and reduced the number of composite events, including overall mortality.35 These results suggest that possible harm from B vitamins in other studies was due to cyanocobalamin or pyridoxine (B6).
In the VITATOPS trial,33 B vitamin therapy with 400 µg cyanocobalamin was not beneficial in patients with diabetes who had an eGFR <50 mL/min/1.73 m² (HR 0.88, 95% CI 0.59 to 1.32, p=0.54), but was beneficial in patients with an eGFR >50 mL/min/1.73 m² (HR 0.82, 95% CI 0.68 to 0.98, p=0.03).
In the DIVINe study,34 B vitamins (including 1000 µg cyanocobalamin) administered to patients with diabetic nephropathy (eGFR >50 mL min/1.73 m²) accelerated decline in renal function and doubled the number of cardiovascular events compared with individuals who did not receive B vitamins.
In the VISP subgroup analysis,32 which excluded patients with an eGFR <46.18 mL/min/1.73 m² and patients who received cyanocobalamin injections, a 34% reduction in the composite outcome of stroke, myocardial infarction or vascular death was observed in patients with normal cyanocobalamin absorption (baseline B12above the median) who received high-dose B vitamin therapy compared with patients with poor cyanocobalamin absorption (baseline B12 below the median) who received low-dose vitamin B therapy.
The Su.Fol.OM3 trial38 reported that B vitamins reduced stroke incidence by 43%, and it is hypothesised that this was because the trial included relatively young participants with good renal function and minimal exposure to cyanocobalamin (only 20 µg daily).
The Heart Outcomes Protection Evaluation-2 (HOPE-2) trial37 reported that B vitamins reduced the risk of stroke by 23%, and it is hypothesised that this was because the trial included a younger study population with better renal function than other trials such as Norwegian Vitamin (NORVIT)36 and VISP.32
The VISP31 32 and NORVIT36 trials showed no benefit of B vitamin therapy for stroke prevention, and it is hypothesised that this was because the participants in these trials were older and had poorer renal function than the study populations of subsequent trials. In the NORVIT trial there was an increased risk of stroke among participants who received cyanocobalamin. Reproduced with permission from Elsevier.12
*eGFR, estimated glomerular filtration rate; CSPPT, China Stroke Primary Prevention Trial; VISP, Vitamins Intervention for Stroke Prevention; VITATOPS, VITAmins TO Prevent Stroke
Smoking
Cigarette smoke, which contains thiocyanate, and cigarette smoking, which has been associated with lower folate and higher tHcy concentrations than never smokers, may also attenuate the effect of folic acid in preventing stroke.41 A subgroup analysis from the CSPPT reported that compared with never smokers, ever smokers may require a higher dosage of folic acid to achieve a greater beneficial effect of folic acid therapy on preventing stroke.41
Antiplatelet therapy
In the three RCTs of B vitamins which assessed stroke as an outcome and reported event rates according to whether or not individuals were taking antiplatelet drugs, a total of 4643 high vascular risk subjects who were not taking antiplatelet drugs were enrolled. Compared with the control group, those allocated B vitamin supplementation had a 29% lower risk of recurrent stroke overall (HR 0.71, 95% CI 0.58 to 0.88).42 These data raise the hypothesis that B vitamins may be an effective strategy to reduce stroke risk in patients who cannot tolerate antiplatelet therapy. A clinical trial of B vitamins may be warranted in this group.42
Conclusion
Epidemiological studies report an independent, triangular association between MTHFR genotype, high tHcy and increased risk of stroke. RCTs show that B vitamin supplementation (vitamin B9 (folic acid), vitamin B12 and vitamin B6) lowers tHcy by about 25% and reduces the relative risk of stroke overall by about 10% compared with placebo.
Factors that appear to modify the effect of B vitamins on stroke risk include low folic acid status, high tHcy (and hence low B12), high cyanocobalamin dose in patients with impaired renal function and concurrent antiplatelet therapy.
For patients with ischaemic stroke in whom the underlying cause is uncertain (ie, cryptogenic) after standard evaluation,43 it is recommended that tHcy be measured as part of the advanced evaluation.
If tHcy is raised, potential cause(s) of the hyperhomocysteinaemia (box 1) should be sought from the clinical assessment and measurement of renal function, thyroid function, glycated haemoglobin and holotranscobalamin or methylmalonic acid in the blood, and treated if possible.
If the cause of the hyperhomocysteinaemia remains unexplained and persistent, measurement of possible genetic determinants (eg, MTHFR) is indicated and daily supplementation with 0.5 mg folic acid and about 0.5 mg vitamin B12 is recommended. This combination would be expected to reduce tHcy by about a quarter to a third (eg, from about 12 umol/L to 8–9 umol/L).11 While vitamin B12 can be administered as cyanocobalamin in patients with normal renal function, it is recommended that vitamin B12 be administered as methylcobalamin or hydroxocobalamin, particularly in patients with impaired renal function.
Any future trials of B vitamins for stroke prevention in regions without folic acid fortification should endeavour to determine whether the relative effects of B vitamin supplementation in reducing stroke risk are consistent throughout the range of baseline tHcy concentrations, or whether there is a threshold tHcy level below which B vitamins become ineffective (eg, as also for blood pressure-lowering and cholesterol-lowering44).
Footnotes
Contributors: GJH wrote the paper.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: The author was the principal investigator of the VITAmins TO Prevent Stroke (VITATOPS) trial. The author received honoraria from AC Immune for chairing the data safety monitoring committee of two clinical trials of vaccines for Alzheimer’s disease, and from Bayer for lecturing about stroke prevention in atrial fibrillation at sponsored scientific symposia.
Provenance and peer review: Commissioned; externally peer reviewed.
Data sharing statement: No additional data are available.
Guest chief editor: J David Spence
References
- 1. McCully KS. Vascular pathology of homocysteinemia: implications for the pathogenesis of arteriosclerosis. Am J Pathol 1969;56:111–28. [PMC free article] [PubMed] [Google Scholar]
- 2. Boers GH, Smals AG, Trijbels FJ, et al. . Heterozygosity for homocystinuria in premature peripheral and cerebral occlusive arterial disease. N Engl J Med 1985;313:709–15. 10.1056/NEJM198509193131201 [DOI] [PubMed] [Google Scholar]
- 3. Wilcken DE, Wilcken B. The natural history of vascular disease in homocystinuria and the effects of treatment. J Inherit Metab Dis 1997;20:295–300. 10.1023/A:1005373209964 [DOI] [PubMed] [Google Scholar]
- 4. Clarke R, Daly L, Robinson K, et al. . Hyperhomocysteinemia: an independent risk factor for vascular disease. N Engl J Med 1991;324:1149–55. 10.1056/NEJM199104253241701 [DOI] [PubMed] [Google Scholar]
- 5. Graham IM. Plasma homocysteine as a risk factor for vascular disease. JAMA 1997;277:1775–81. 10.1001/jama.1997.03540460039030 [DOI] [PubMed] [Google Scholar]
- 6. Homocysteine Studies Collaboration. Homocysteine and risk of ischemic heart disease and stroke: a meta-analysis. JAMA 2002;288:2015–22. [DOI] [PubMed] [Google Scholar]
- 7. Wald DS, Law M, Morris JK. Homocysteine and cardiovascular disease: evidence on causality from a meta-analysis. BMJ 2002;325:1202–6. 10.1136/bmj.325.7374.1202 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Chambers JC, Obeid OA, Kooner JS. Physiological increments in plasma homocysteine induce vascular endothelial dysfunction in normal human subjects. Arterioscler Thromb Vasc Biol 1999;19:2922–7. 10.1161/01.ATV.19.12.2922 [DOI] [PubMed] [Google Scholar]
- 9. Faraci FM, Lentz SR. Hyperhomocysteinemia, oxidative stress, and cerebral vascular dysfunction. Stroke 2004;35:345–7. 10.1161/01.STR.0000115161.10646.67 [DOI] [PubMed] [Google Scholar]
- 10. Zhang D, Jiang X, Fang P, et al. . Hyperhomocysteinemia promotes inflammatory monocyte generation and accelerates atherosclerosis in transgenic cystathionine beta-synthase-deficient mice. Circulation 2009;120:1893–902. 10.1161/CIRCULATIONAHA.109.866889 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Homocysteine lowering trialist’s collaboration. Lowering blood homocysteine with folic acid based supplements: meta-analysis of randomised trials. BMJ 1998;316:894–8. [PMC free article] [PubMed] [Google Scholar]
- 12. Spence JD, Yi Q, Hankey GJ. B vitamins in stroke prevention: time to reconsider. Lancet Neurol 2017;16:750–60. 10.1016/S1474-4422(17)30180-1 [DOI] [PubMed] [Google Scholar]
- 13. Mudd SH, Finkelstein JD, Refsum H, et al. . Homocysteine and its disulfide derivatives: a suggested consensus terminology. Arterioscler Thromb Vasc Biol 2000;20:1704–6. 10.1161/01.ATV.20.7.1704 [DOI] [PubMed] [Google Scholar]
- 14. Wang J, Huff AM, Spence JD, et al. . Single nucleotide polymorphism in CTH associated with variation in plasma homocysteine concentration. Clin Genet 2004;65:483–6. 10.1111/j.1399-0004.2004.00250.x [DOI] [PubMed] [Google Scholar]
- 15. Fowler B. Homocysteine: overview of biochemistry, molecular biology, and role in disease processes. Semin Vasc Med 2005;5:77–86. 10.1055/s-2005-872394 [DOI] [PubMed] [Google Scholar]
- 16. Kang SS, Zhou J, Wong PW, et al. . Intermediate homocysteinemia: a thermolabile variant of methylenetetrahydrofolate reductase. Am J Hum Genet 1988;43:414–21. [PMC free article] [PubMed] [Google Scholar]
- 17. Frosst P, Blom HJ, Milos R, et al. . A candidate genetic risk factor for vascular disease: a common mutation in methylenetetrahydrofolate reductase. Nat Genet 1995;10:111–3. 10.1038/ng0595-111 [DOI] [PubMed] [Google Scholar]
- 18. Brattstrom L, Wilcken DEL, Ohrvik J, et al. . Common methylenetetrahydrofolate reductase gene mutation leads to hyperhomocysteinemia but not to vascular disease : the result of a meta-analysis. Circulation 1998;98:2520–6. 10.1161/01.CIR.98.23.2520 [DOI] [PubMed] [Google Scholar]
- 19. Mudd SH, Skovby F, Levy HL, et al. . The natural history of homocystinuria due to cystathionine beta-synthase deficiency. Am J Hum Genet 1985;37:1–31. [PMC free article] [PubMed] [Google Scholar]
- 20. Hill AB. The environment and disease: association or causation? Proc R Soc Med 1965;58:295–300. [PMC free article] [PubMed] [Google Scholar]
- 21. Casas JP, Bautista LE, Smeeth L, et al. . Homocysteine and stroke: evidence on a causal link from mendelian randomisation. Lancet 2005;365:224–32. 10.1016/S0140-6736(05)70152-5 [DOI] [PubMed] [Google Scholar]
- 22. Holmes MV, Newcombe P, Hubacek JA, et al. . Effect modification by population dietary folate on the association between MTHFR genotype, homocysteine, and stroke risk: a meta-analysis of genetic studies and randomised trials. Lancet 2011;378:584–94. 10.1016/S0140-6736(11)60872-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Martí-Carvajal AJ, Solà I, Lathyris D, et al. . Homocysteine-lowering interventions for preventing cardiovascular events. Cochrane Database Syst Rev 2017;8:CD006612 10.1002/14651858.CD006612.pub5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Li Y, Huang T, Zheng Y, et al. . Folic acid supplementation and the risk of cardiovascular diseases: a meta-analysis of randomized controlled trials. J Am Heart Assoc 2016;5:e003768 10.1161/JAHA.116.003768 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Tian T, Yang KQ, Cui JG, et al. . Folic acid supplementation for stroke prevention in patients with cardiovascular disease. Am J Med Sci 2017;354:379–87. 10.1016/j.amjms.2017.05.020 [DOI] [PubMed] [Google Scholar]
- 26. Zhao M, Wu G, Li Y, et al. . Meta-analysis of folic acid efficacy trials in stroke prevention: Insight into effect modifiers. Neurology 2017;88:1830–8. 10.1212/WNL.0000000000003909 [DOI] [PubMed] [Google Scholar]
- 27. Hsu CY, Chiu SW, Hong KS, et al. . Folic acid in stroke prevention in countries without mandatory folic acid food fortification: a meta-analysis of randomized controlled trials. J Stroke 2018;20:99–109. 10.5853/jos.2017.01522 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Huang X, Li Y, Li P, et al. . Association between percent decline in serum total homocysteine and risk of first stroke. Neurology 2017;89:2101–7. 10.1212/WNL.0000000000004648 [DOI] [PubMed] [Google Scholar]
- 29. Stabler SP. Vitamin B12 deficiency. N Engl J Med 2013;368:149–60. [DOI] [PubMed] [Google Scholar]
- 30. Spence JD. Metabolic vitamin B12 deficiency: a missed opportunity to prevent dementia and stroke. Nutr Res 2016;36:109–16. 10.1016/j.nutres.2015.10.003 [DOI] [PubMed] [Google Scholar]
- 31. Huang X, Qin X, Yang W, et al. . MTHFR gene and serum folate interaction on serum homocysteine lowering: prospect for precision folic acid treatment. Arterioscler Thromb Vasc Biol 2018;38:679–85. 10.1161/ATVBAHA.117.310211 [DOI] [PubMed] [Google Scholar]
- 32. Spence JD, Bang H, Chambless LE, et al. . Vitamin intervention for stroke prevention trial: an efficacy analysis. Stroke 2005;36:2404–9. 10.1161/01.STR.0000185929.38534.f3 [DOI] [PubMed] [Google Scholar]
- 33. VITATOPS Trial Study Group. B vitamins in patients with recent transient ischaemic attack or stroke in the VITAmins TO Prevent Stroke (VITATOPS) trial: a randomised, double-blind, parallel, placebo-controlled trial. Lancet Neurol 2010;9:855–65. 10.1016/S1474-4422(10)70187-3 [DOI] [PubMed] [Google Scholar]
- 34. House AA, Eliasziw M, Cattran DC, et al. . Effect of B-vitamin therapy on progression of diabetic nephropathy: a randomized controlled trial. JAMA 2010;303:1603–9. 10.1001/jama.2010.490 [DOI] [PubMed] [Google Scholar]
- 35. Xu X, Qin X, Li Y, et al. . Efficacy of folic acid therapy on the progression of chronic kidney disease: the renal substudy of the china stroke primary prevention trial. JAMA Intern Med 2016;176:1443–50. 10.1001/jamainternmed.2016.4687 [DOI] [PubMed] [Google Scholar]
- 36. Bønaa KH, Njølstad I, Ueland PM, et al. . Homocysteine lowering and cardiovascular events after acute myocardial infarction. N Engl J Med 2006;354:1578–88. 10.1056/NEJMoa055227 [DOI] [PubMed] [Google Scholar]
- 37. Lonn E, Yusuf S, Arnold MJ, et al. . Homocysteine lowering with folic acid and B vitamins in vascular disease. N Engl J Med 2006;354:1567–77. 10.1056/NEJMoa060900 [DOI] [PubMed] [Google Scholar]
- 38. Galan P, Kesse-Guyot E, Czernichow S, et al. . Effects of B vitamins and omega 3 fatty acids on cardiovascular diseases: a randomised placebo controlled trial. BMJ 2010;341:c6273 10.1136/bmj.c6273 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Armitage JM, Bowman L, Clarke RJ, et al. . Effects of homocysteine-lowering with folic acid plus vitamin B12 vs placebo on mortality and major morbidity in myocardial infarction survivors: a randomized trial. JAMA 2010;303:2486–94. 10.1001/jama.2010.840 [DOI] [PubMed] [Google Scholar]
- 40. Koyama K, Ito A, Yamamoto J, et al. . Randomized controlled trial of the effect of short-term coadministration of methylcobalamin and folate on serum ADMA concentration in patients receiving long-term hemodialysis. Am J Kidney Dis 2010;55:1069–78. 10.1053/j.ajkd.2009.12.035 [DOI] [PubMed] [Google Scholar]
- 41. Zhou Z, Li J, Yu Y, et al. . Effect of smoking and folate levels on the efficacy of folic acid therapy in prevention of stroke in hypertensive men. Stroke 2018;49:114–20. 10.1161/STROKEAHA.117.018273 [DOI] [PubMed] [Google Scholar]
- 42. Park JH, Saposnik G, Ovbiagele B, et al. . Effect of B-vitamins on stroke risk among individuals with vascular disease who are not on antiplatelets: A meta-analysis. Int J Stroke 2016;11:206–11. 10.1177/1747493015616512 [DOI] [PubMed] [Google Scholar]
- 43. Saver JL. Cryptogenic Stroke. N Engl J Med Overseas Ed 2016;374:2065–74. 10.1056/NEJMcp1503946 [DOI] [PubMed] [Google Scholar]
- 44. Navarese EP, Robinson JG, Kowalewski M, et al. . Association between baseline LDL-C level and total and cardiovascular mortality after LDL-C lowering: a systematic review and meta-analysis. JAMA 2018;319:1566–79. 10.1001/jama.2018.2525 [DOI] [PMC free article] [PubMed] [Google Scholar]