Abstract
Laparoscopic transgastric stapler assisted mucosectomy (SAM) has been described for minimally invasive circumferential en-bloc resection of Barrett’s esophagus (BE). Conceivably long-term disease control might be achieved by adding anti-reflux surgery after resection of BE by SAM. The aim of this study was to assess the feasibility of combined SAM and fundoplication in one laparoscopic procedure in 6 pigs. Furthermore, the competence of the gastroesophageal junction (GEJ) was assessed at baseline, after SAM, and after subsequent laparoscopic fundoplication. At each measuring point reflux measurements were repeated 6 times in each pig. Blue-colored water was infused into the stomach for provocation of reflux. Intragastric yield pressure and volume until drainage of blue solution (DBS) were recorded. Time to reflux by DBS and by multichannel intraluminal impedance (MII) was measured. Laparoscopic fundoplication after SAM was feasible in all animals in a single-session. A weakening of the GEJ was found after SAM, indicated by decreased yield pressure, yield volume, time to DBS and MII (p=0.007, p<0.001, p<0.001 and p=0.008). After adding a Nissen-fundoplication the GEJ was restored. The measurements of the competence of the GEJ returned to baseline values. (Yield volume, p=0.11; time to DBS, p=0.15; MII p=0.84). The yield pressure increased from 11.5 mmHg at baseline to 19.7 mmHg after SAM and fundoplication (p<0.001). Laparoscopic fundoplication and SAM may be combined in a single laparoscopic session. Although weakened after SAM Nissen fundoplication restored the GEJ as an effective reflux barrier in this experiment. Clinical validation is needed to confirm the results in a prospective human trial.
Keywords: Fundoplication, Barrett´s esophagus, Impedance manometry, Gastroesophageal reflux, minimally invasive surgery
Introduction
Barrett’s esophagus (BE) is a transformation of esophageal squamous epithelium into metaplastic columnar epithelium. In this complex process of tissue transformation severe and ongoing gastro-esophageal reflux disease (GERD) plays an important role and has been identified as a risk factor for the development of BE.1, 2 The incidence of BE in the Western population is estimated to be 1.5%.3 A minority of patients with BE will develop dysplasia.4 Eventually, the risk of development of high-grade dysplasia (HGD) is as high as 9% per year.5 Being diagnosed with HGD the annual progress rate to esophageal adenocarcinoma (EAC) is 10% to 25%.6, 7
Endoscopic therapy has become an alternative to esophagectomy for dysplastic BE and early adenocarcinoma of the esophagus in the last decade. Nevertheless, when histopathologic risk factors for lymph node involvement are present and based on the depth of submucosal infiltration esophagectomy may still be required.8, 9 Endoscopic therapy consists in endoscopic mucosal resection (EMR) or endoscopic submucosa dissection (ESD). In large, close to or circumferential lesions EMR may be performed stepweise in two or more sessions.10 Alternatively, the EMR is limited to the dysplastic region while the remaining BE is treated by ablation such as radiofrequency ablation (RFA), cryoablation or Argon beam coagulation.11, 12 Drawbacks of endoscopic therapy are the piecemeal technique prone to incomplete resection13, 14, the risk for post-intervention stenosis10, 12 and the limitation of the maximum area that is resectable. Furthermore, GERD as the underlying disease of BE remains untreated.
GERD in the context of BE is usually treated either medically, typically involving a proton pump inhibitor, or by laparoscopic fundoplication. It remains controversial if antireflux surgery does reduce the risk of disease progression in BE more effectively compared to medical treatment.15 However, after fundoplication studies have shown a regression in length of BE, a loss of BE, and a loss of dysplasia in some patients.16–18 In a 5-years observation study none of 85 patients with BE undergoing antireflux surgery developed disease progression to HGD or EAC.19 Conflicting conclusions were drawn by meta-analyses analyzing BE progression towards EAC after antireflux surgery compared to medical treatment. While two previous meta-analysis failed to show a difference between medical and surgical treatment16, 19 the most recent meta-analysis demonstrated a strong tendency to a decreased risk of EAC in patients that underwent fundoplication.20 The analysis of seven studies showed a decreased pooled incidence rate ratio of EAC of 0.46 (95% confidence interval 0.20-1.08) in patients treated with antireflux surgery compared to the medically treated patients.
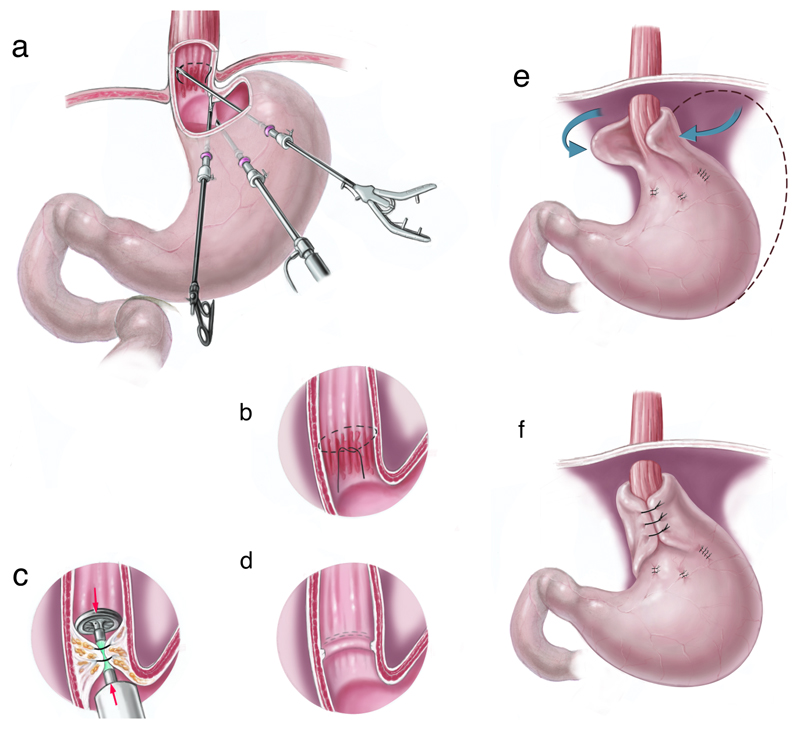
As an alternative to EMR or ESD, a novel surgical minimally invasive technique for laparoscopic transgastric stapler-assisted esophageal mucosectomy (SAM) has been proposed previously.21 By SAM, the BE is resected circumferentially using a circular endostapler. In brief, by repositioning of laparoscopic trocars through the anterior gastric wall, the mucosal aspect of the gastroesphageal junction is visualized. By help of rigid laparoscopic instruments and an circumferential endostapler, the BE is circumferentially resected while the mucosal borders are sutured by staples (Figure 1). This obtains a circumferential, single, oriented mucosectomy specimen thus facilitating pathologic work-up. Unlike as in EMR or ESD no mucosal defect is left to open wound healing. Previously, wound healing and stricture formation has been compared after open esophageal mucosectomy with leaving the defect uncovered or advancing the mucosa and closing the defect by hand sutures. While no stenosis was observed in covered defects, dense fibrotic strictures formed when the defect was left uncovered after six weeks in a porcine experiment.22 These findings were confirmed when SAM was compared to EMR. In a pig modell after 6-weeks of survival no strictures occurred in SAM while all animals of the EMR group showed a significant narrowing of the esophageal lumen.21
Figure 1.
a: Principle of transgastric stapler-assisted mucosectomy: By help of three transabdominal-transgastric trocars two mucosal pursestring sutures are placed in the distal esophagus orally and aborally of the Barrett Epithelium. The anvil of the circular stapler is inserted and the purse-string sutures are tightened around the spine of the anvil. By firing the stapler a circumferential mucosectomy specimen is obtained. Staples approximate the mucosal borders and no mucosal defect is left open. b: After mobilization, the fundus is positioned in a loose manner around the esophagus. The 360°-Nissen-fundoplication is secured with three sutures.
When combining SAM and a Nissen fundoplication, BE and GERD, as an important underlying cause of BE, might be controlled in a single-session laparoscopic surgery. This experimental study was aimed to assess the feasibility of combined SAM and 360° Nissen fundoplication in a single laparoscopic session in a porcine in-vivo reflux model. Furthermore, the effectiveness of reflux control using a Nissen fundoplication after SAM was evaulated.
Material and Methods
This in-vivo study was performed in a porcine model using six German Landrace pigs of both sexes weighting 34-kg to 42-kg. Ethical approval was obtained from the Animal Care Committee of Karlsruhe, Germany (Reference-Number: 95-3185.81/G-62/15). Treatment of the animals was in accordance with the Animal Research: Reporting of In Vivo Experiments (ARRIVE) guidelines for animal research.23 All procedures were performed by a single, attending surgeon (D.C.S.). He has extensive experience in laparoscopic surgery and holds a certificate in advanced laparoscopic surgery from the Swiss Association for Laparoscopic and Thoracoscopic Surgery.
Study design
The hypothesis was that the combination of SAM and fundoplication is feasible and that fundoplication represents an effective reflux barrier after SAM. For this pilot study on a novel surgical technique there is no data on which a sample size calculation might be based in the current literature. The reproducibility of the procedure is, however, usually determined after the procedure is performed in 6 pigs.24, 25
The feasibility of fundoplication after SAM was assessed. SAM was estimated successful when a circumferential, complete mucosal resection was achieved. Competence of the gastroesophageal junction (GEJ) was assessed intraoperatively at three time points: (1) at baseline, (2) after SAM, and (3) after SAM and fundoplication. At each time point the measurements were repeated six times in each animal. The competence of the GEJ was assessed 4-fold: (1) Blue-colored water was infused in the gastric lumen. The time until drainage of blue solution (DBS) over an esophageal tube was measured. (2) The intragastric volume infused until DBS was recorded (yield volume). (3) The intragatric pressure when DBS occurred was recorded (yield pressure). (4) While infusing the blue-colored water the time until first appearance of GER detected by intraesophageal multichannel intraluminal impedance (MII) was detected. The infusion was stopped when reaching an intragastric pressure of 45-mmHg and no reflux occurred. Then the GEJ was declared insurmountable for provoked reflux.
Experimental setting
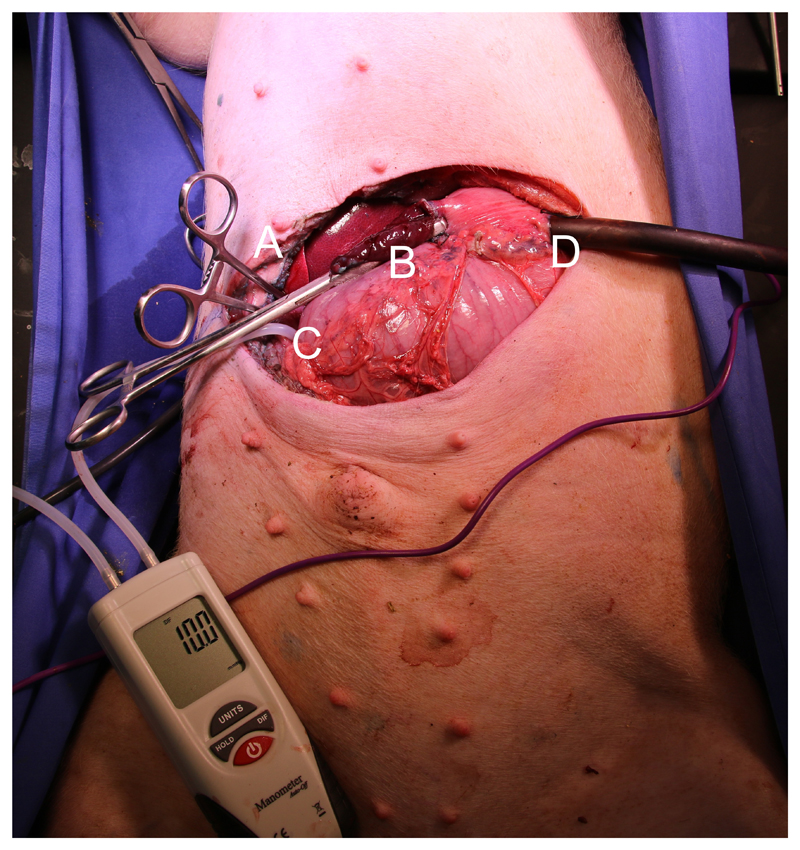
Animals fasted for 24-hours and were premedicated intramuscularly with ketamine (10 mg/kg), azaperone (4 mg/kg) and midazolam (0.5 mg/kg). Subsequently the animals were intubated and general anesthesia was maintained using sevoflurane. No muscle relaxation was used. A transverse laparotomy in the left upper quadrant was performed. The greater omentum was dissected along the gastroepiploic vessels and the short gastric vessels were divided. A 24-french catheter was inserted into the gastric lumen through a small ventral gastrostomy and secured using a purse-string suture. This tube was connected to a roll-pump (Heidolph™ PD Series, Heidolph Instruments GmbH & Co. KG, Schwabach, Germany) for infusion of blue-colored water (Toluidin, F. Köhler Chemie GmbH, Bensheim, Germany) at a constant flow rate of 14 ml/second. An esophageal tube was placed 5-cm above the GEJ and constantly sucked using a low-pressure suction device. A further small gastrostomy was performed for the insertion of a tube for the intragastric pressure monitoring using a portable pressure gauge (Digital LCD manometer, Rise Ltd, Hong Kong). A third gastrostomy was created to empty the stomach of the blue solution using suction after each measurement. During the measurements this gastrostomy was temporarily closed with a clamp. The duodenum was mobilized and clamped to avoid run-off of blue solution into the intestine (Figure 2).
Figure 2.
Setting for intraoperative measurement of the competence of the gastroesophageal junction. A) Clamp to close the duodenum, B) Gastrostomy for suction of blue-colored water between the measurement, C) Tube and instrument for intragastric pressure measurement, D) Tube for infusion of blue-colored water by means of a roll-pump.
Stapler-assisted mucosectomy
Before performing the SAM procedure the transverse laparotomy was closed by peritoneal and facial running sutures to allow the establishment of a pneumoperitoneum. A 12-mmHg pneumoperitoneum was established and three 12-mm trocars (Kii® Fios, Applied Medical, Rancho Santa Margarita, California) were placed in left subcostal, epigastric and in the left upper quadrant. The technique of laparoscopic transgastric stapler-assisted mucosectomy has been reported before and is shown in Figure 1.21 After redeploying the trocars through the ventral gastric wall, a 12-mmHg pneumogastrium was established. Two submucosal purse-string sutures in the distal oesophagus were placed. The left subcostal facial incision was subsequently enlarged to 2.5-cm and a wound protector was inserted into the stomach (Alexis™ XXS, Applied Medical, Rancho Santa Margarita, California). The anvil of a 25-mm circular stapler (CDH25A, 25mm/5.5mm, Ethicon, Somerville, New Jersey) was inserted in the esophagus. The purse-string sutures were knotted and the anvil connected to the endostapler. An en-bloc mucosectomy was performed by means of the circular endostapler. Gastrotomies were closed by sutures. The procedure was terminated after an air leak-test and intra-cutaneous wound closure. Contrary to the original description of the technique the hiatus was not mobilized in this experiment. After SAM the laparotomy was re-opened for the measurements.
360° Nissen-fundoplication
Again, before laparoscopic fundoplication the laparotomy was closed. Four 12-mm trocars were placed in the right subcostal, epigastric, and in the middle and left upper abdomen. A 12-mmHg pneumoperitoneum established. A liver retractor was introduced in the right subcostal trocar and the left liver lobe was lifted to expose the hiatus. The hiatus was completely mobilized using the monopolar hook. Then gastric fundus was mobilized and remaining short gastric vessels were cut using LigaSure (Medtronic plc, Dublin, Ireland). A 36-french gastric tube was inserted transorally for calibration of the fundoplication. The fundus was positioned in a loose manner behind the esophagus. The fundoplication was secured with three braided sutures (Figure 1). The large gastric tube was removed. Subsequently the pneumoperitoneum was released, the trocars removed, and the laparotomy re-opened for measurements.
Measuring unit
A 6-chanel impedance probe containing 8 measuring segments between 2-cm and 13-cm in various distances (SI-ZNIS-L, Standard Instruments, Karlsruhe, Germany) was used for impedance measurement. For registration Vizion® portable system and for analysis ViMeDat™ software (Standard Instruments GmbH, Karlsruhe, Germany) was employed. For detection of DBS a 14-french gastric tube was employed. Both catheters were fixed together using hydrophobic scotch tape (Figure 3). The tip of the gastric probe was positioned 5-cm above the GEJ. After insertion of the combined catheter the position of the tip of the MII was positioned at the GEJ guided by digital palpation. The distance of the tip from the incisior teeth was marked. The correct position of the tube was controlled after each measurement. After each measurement the ventral gastrostomy was opened by removal of the clamp and the intragastric fluid was sucked. The volume of sucked fluid was recorded. Then a further 14-french gastric tube was inserted through the gastrostomy retrograde in the esophagus and remaining fluid in the esophagus was sucked using a low-pressure suction device. Subsequently the correct positioning of the measurement probe was checked.
Figure 3.

Combined impedance probe and gastric tube that was positioned in the esophagus.
Definition of gastroesophageal reflux
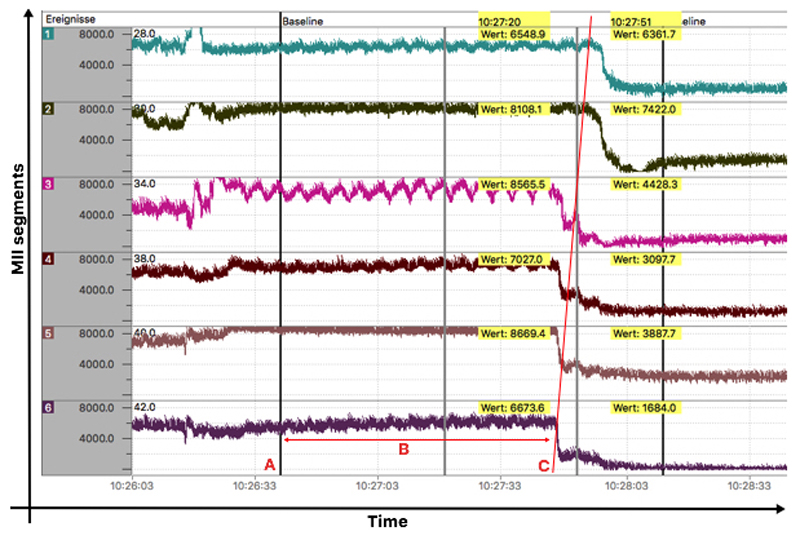
GER detected by DBS was defined as time to first visible appearance of blue-colored solution.20 The intragastric yield pressure was set at visible DBS. GER detected by MII was defined as a decrease in impedance of at least 50% for the baseline registered in the first segment from the tip. The decrease of impedance had to be sequential, beginning in the most distal measuring segment from the tip of the probe (Figure 4).
Figure 4.
Sample for detection of provoked reflux by multichannel intraluminal impedance (MII) measurement at baseline. A) Start of infusion of blue-colored water into stomach, B) time to detection of reflux by MII, C) line connecting the time points when decrease in impedance at different impedance segments occurs.
Statistical analysis
Statistical analysis was performed using SPSS software (IBM® SPSS®Statistics, Version 22, Chicago, Illinois). The results of parametric data were expressed as mean and 95% confidence interval. Normal distributions of the values for each measuring point were tested using the Kolmogorov-Smirnov-test. Non-parametric data were expressed as median and interquartile range. Comparisons between the different measuring points of the results were performed using a paired student´s t test. Multiple group comparisons were performed using a one-way ANOVA. The level of significance for a difference between the measuring points was set at a p value of ≤0.05.
Results
The median weight of the animals was 39.5-kg [36.3-41.3]. The median operation time for SAM and for fundoplication was 90 minutes [80-107] and 35 minutes [24-86], respectively.
Feasibility
Fundoplication was feasible after SAM procedure without any technical difficulties. Furthermore, the SAM procedure revealed an intact circumferential mucosectomy specimen. Macroscopically the specimens did not contain muscle portions. No transmural resection occurred.
Measuring parameters
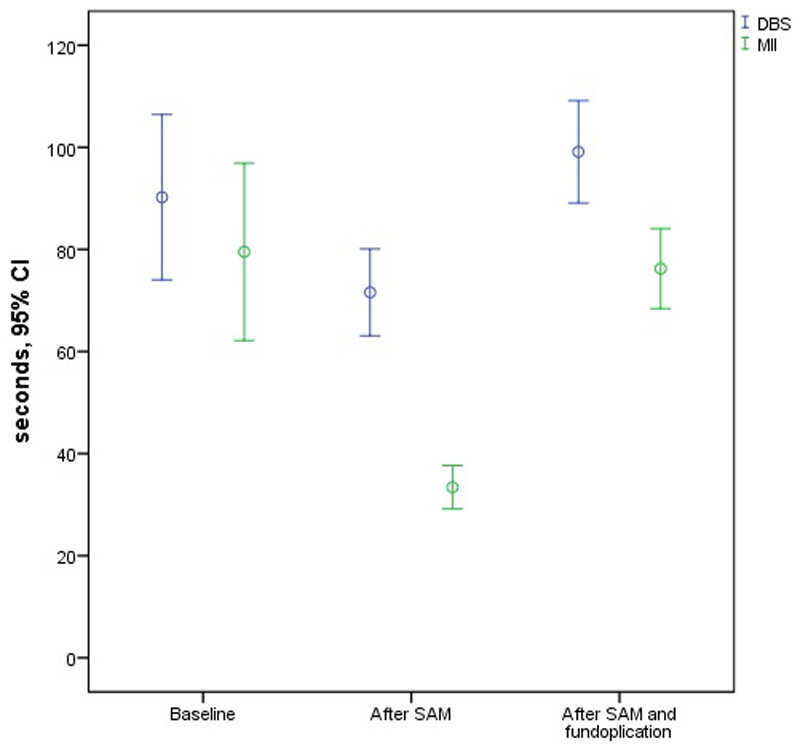
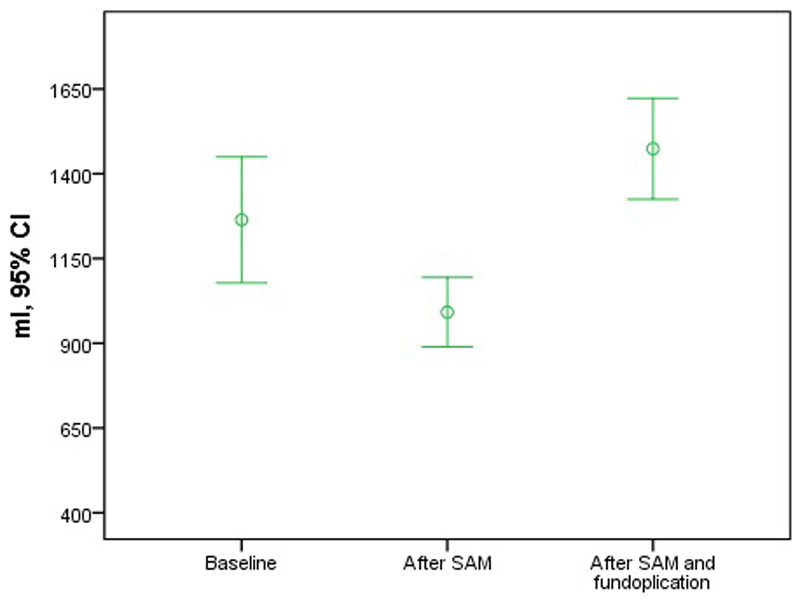
The mean time until reflux detected by DBS, by MII, yield volume, and yield pressure for the measurements at baseline and after SAM are depicted in table 1 and Figures 5–7. After SAM and fundoplication in 19 out of 36 measurements the reflux barrier was insurmountable and no reflux could be provoked. The respective measuring values after SAM and fundoplication are those of the remaining 17 measurements when reflux could still be provoked (Table 1, Figures 5-7).
Table 1.
Time to reflux detected by drainage of blue solution (DBS), by decrease of multichannel intraluminal impedance (MII), and intragastric yield pressure at DBS as well as intragastric volume at DBS.
| Baseline (N=36) |
After SAM (N=36) |
p§ | After SAM and fundoplication* (N=17) |
p§ | |
|---|---|---|---|---|---|
| DBS, seconds (95% CI) |
90 (74-106) | 72 (63-80) | 0.008 | 99 (89-109) | 0.147 |
| MII, seconds (95% CI) |
80 (62-97) | 33 (29-38) | <0.001 | 76 (68-84) | 0.84 |
| Yield pressure, mmHg (95% CI) |
11.5 (10.4-12.7) | 8.5 (7.7-9.4) | <0.001 | 19.7 (18.1-21.3) | <0.001 |
| Yield volume, ml (95% CI) |
1’248 (1’024-1’472) | 987 (870-1’104) | 0.007 | 1’474 (1’324-1’623) | 0.106 |
SAM: stapler-assisted mucosectomy; CI: confidence interval;
compared to baseline,
values for fundoplications that yielded to intragastric infusion (17/36 measurements)
Figure 5.
Mean time and 95% confidence interval for first detection of reflux by drainage of blue solution (DBS) and multichannel intraluminal impedance (MII) at baseline, after stapler assisted mucosectomy (SAM), and after SAM and fundoplication. After fundoplication in 19 measurements the reflux barrier was insurmountable and no reflux could be provoked. The respective values for the measurements after SAM and fundoplication are those of the remaining 17 measurements when reflux could still be provoked.
Figure 7.
Mean time and 95% confidence interval for yield pressure at baseline, after stapler assisted mucosectomy (SAM), and after SAM and fundoplication. After fundoplication in 19 measurements the reflux barrier was insurmountable and no reflux could be provoked. The respective values for the measurements after SAM and fundoplication are those of the remaining 17 measurements when reflux could still be provoked.
Correlation of measuring parameters
Time to reflux detected by DBS was positively correlated to the time until decrease of MII (r=0.973, p<0.0001), to the intragastric yield pressure at DBS (r=0.765, p<0.0001), and to the intragastric volume at DBS (r=1.0, p<0.0001).
Discussion
While EMR does not treat GERD as the underlying disease of BE, SAM may be combined with a laparoscopic fundoplication in a single-session as proposed in the current experiment. In the experiments the intragastric yield pressure and the yield volume when reflux occurs were decreased after SAM indicating a weakening of the GEJ competence as reflux barrier. At least when measured immediately after surgery, it seemed that SAM impaired the GEJ competence. Macro- and microscopic studies, however, did not reveal resection of muscle tissue by SAM.21 Therefore a resection of parts of the esophageal sphincter apparatus may be excluded. An explanation for the finding may albeit be that the stiff circular stapler line holds the esophageal lumen open. Moreover, introducing the anvil of the circular stapler may have widened the GEJ although a lag time of at least 30 minutes after SAM until measuring has been respected. However, after fundoplication the reflux barrier was restored in all animals.
SAM was developped and evaluated in a previous experiment.21 It was considered safe in a 6-weeks porcine survival experiment comparing SAM and circumferential EMR. All animals thrived well after surgery. Circumferential intact specimens were obtained in all pigs. The median maximum length resected was 22 mm using a circular endostapler with a 25 mm circumference. The muscularis layer including muscularis mucosae was preserved in all specimens but the submucosa was present only in half of the mucosectomies. The resection, however, did not involve the muscularis propria. Interestingly, 6 weeks after intervention stricture formation was observed in all animals undergoing EMR but in none after SAM despite a significant larger resection area in the SAM group. Two out of 6 animals in the EMR were sacrificed prematurely due to symptomatic stenosis. At necropsy the lumen at level of resection was narrowed in EMR but not in SAM (7.45 mm vs 16.8 mm, p<0.001). The ratio of the esophageal width at level of resection divided by the width proximal to the resection zone was 0.71 for EMR and 0.98 for SAM (p<0.001). In EMR, in 3 out of 6 pigs damage and collagen deposits in the tunica muscularis were found. In contrast mucosal healing in SAM without stenosis or fibrosis was demonstrated.
Frantzides et al have described laparoscopic mucosal resection by scissors for HGD via an anterior gastrotomy in combination with Nissen fundoplication before in humans.26–28 A resection extending 5-cm from the Z-line has been described to be feasible. The access by transgastric endoscopy and the use of a circumferential endostapler for mucosectomy in SAM, as proposed, however, is novel. In the technique described by Frantzides26–28, the mucosal wound was left to open wound healing. Consequently in two out of 11 patients stenosis necessitating endoscopic dilatation occurred. Farrell has reported in a previous porcine study circular mucosal resection of the distal esophagus of segments up to 5-cm in length in an open surgical technique.22 The defects were then covered and hand sutures reconstructed the mucosal integrity. As in SAM no or only mild fibrosis and no stenosis was observed.
EMR of HGD followed by RFA of the remaining BE is currently considered the first-line treatment.7 Nevertheless, despite EMR and RFA in a cohort of 335 patients from the UK National Halo RFA Registry 86% of patients were cleared from HGD and only 62% from BE 12-months after a mean of 2.5 RFA procedures. Invasive cancer developed in 3% of patients despite endoscopic treatment.29 The US Multicenter Consortium Recurrence reports complete remission of BE in 56% 24-months after combined EMR and RFA. BE recurrence after 2 years after complete eradication was 33%.30 These findings warrant a close lifetime follow-up with repeated endoscopy studies in patients treated for HGD with EMR and RFA. In a decision analysis-model the effectiveness of combined EMR-RFA against esophagectomy was investigated. EMR-RFA seemed to be equivalent to esophagectomy irrespective of patients age.31 Nevertheless, alternative treatment options may be indicated in patients preferring a single, definitive treatment and patients necessitating a surgical anti-reflux procedure. Moreover, in the subgroup of patients suffering from an extensive or even circumferential HGD beyond the limits of combined EMR and RFA, SAM combined with fundoplication may represent a safe alternative treatment to esophagectomy.
Currently the extent of resection is limited by the capacity of the circular stapler to contain tissue. The used circular staplers are not designed for resection but to fashion anastomosis. The relatively short resection may also explain the missing submucosal layer in part of the specimens. In the further development of SAM a modified circular stapler must be designed with the capacity to resection longer segments.
The specific advantages of SAM versus endoscopic treatment are a) the circular en-bloc mucosectomy enabling a higher probability of complete BE eradication, b) the possibility for a more accurate microscopic evaluation of the lateral and deep resection margins, c) a low probability of post-intervention stenosis and d) the option for concomitant anti-reflux surgery providing not only the eradication of BE and HGD but also to cure the underlying GERD. These advantages of SAM compared to endoscopic therapy must be counterbalanced with the increased invasiveness and potential serious complications such as intraabdominal infection, wound infection, leak of the gastrostomy closure, or esophageal performation.
Conclusions
This porcine in-vivo experiment demonstrated that laparoscopic SAM and fundoplication may be combined in a single laparoscopic session. Although weakened after SAM Nissen fundoplication restored the GEJ as an effective reflux barrier in this experiment. Clinical validation is needed to confirm the results in a prospective human trial.
Figure 6.
Mean time and 95% confidence interval for yield volume at baseline, after stapler assisted mucosectomy (SAM), and after SAM and fundoplication. After fundoplication in 19 measurements the reflux barrier was insurmountable and no reflux could be provoked. The respective values for the measurements after SAM and fundoplication are those of the remaining 17 measurements when reflux could still be provoked.
Acknowledgement
-
Funding
The German Research Foundation (DFG) funded the study (STE 2590/1-1). Impedance monitoring instruments were loaned free of charge from Standard Instruments GmbH, Werftstrasse 12, 76189 Karlsruhe, Germany.
Abbreviations
- BE
Barrett´s esophagus
- DBS
Drainage of blue solution
- EAC
Esophageal adenocarcinoma
- EMR
Endoscopic mucosa resection
- GEJ
Gastroesophageal junction
- GER
Gastroesophageal reflux
- MII
Multichannel intraluminal impedance measurement
- RFA
Radiofrequency Ablation
- SAM
Stapler-assisted mucosectomy
Footnotes
Meeting presentation: The results of this study were presented at the 25th International Congress of the European Association for Endoscopic Surgery in Frankfurt, Germany, June 14-17, 2017.
Conflicts of interest
Daniel C. Steinemann is supported by the Swiss National Science Foundation (Grant P300PB-161099/1) and the Margarete and Walter Lichtenstein Foundation, Basel, Switzerland (DMS2321). All other authors have nothing to disclose.
Author contributions
DCS and PCM designed the study, performed the experiments, analyzed the data and drafted the manuscript. ACS performed the experiments and performed a critical revision. GRL, AZ and BPM designed the study and performed a critical revision. All authors gave their final approval.
References
- 1.Lagergren J, Bergstrom R, Lindgren A, et al. Symptomatic gastroesophageal reflux as a risk factor for esophageal adenocarcinoma. N Engl J Med. 1999;340(11):825–31. doi: 10.1056/NEJM199903183401101. [DOI] [PubMed] [Google Scholar]
- 2.Ireland CJ, Fielder AL, Thompson SK, et al. Development of a risk prediction model for Barrett's esophagus in an Australian population. Dis Esophagus. 2017;30(11):1–8. doi: 10.1093/dote/dox033. [DOI] [PubMed] [Google Scholar]
- 3.Findlay JM, Middleton MR, Tomlinson I. Genetic Biomarkers of Barrett's Esophagus Susceptibility and Progression to Dysplasia and Cancer: A Systematic Review and Meta-Analysis. Dig Dis Sci. 2016;61(1):25–38. doi: 10.1007/s10620-015-3884-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bhat S, Coleman HG, Yousef F, et al. Risk of malignant progression in Barrett's esophagus patients: results from a large population-based study. J Natl Cancer Inst. 2011;103(13):1049–57. doi: 10.1093/jnci/djr203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Duits LC, Phoa KN, Curvers WL, et al. Barrett's oesophagus patients with low-grade dysplasia can be accurately risk-stratified after histological review by an expert pathology panel. Gut. 2015;64(5):700–6. doi: 10.1136/gutjnl-2014-307278. [DOI] [PubMed] [Google Scholar]
- 6.Bennett C, Vakil N, Bergman J, et al. Consensus statements for management of Barrett's dysplasia and early-stage esophageal adenocarcinoma, based on a Delphi process. Gastroenterology. 2012;143(2):336–46. doi: 10.1053/j.gastro.2012.04.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kastelein F, van Olphen S, Steyerberg EW, et al. Surveillance in patients with long-segment Barrett's oesophagus: a cost-effectiveness analysis. Gut. 2015;64(6):864–71. doi: 10.1136/gutjnl-2014-307197. [DOI] [PubMed] [Google Scholar]
- 8.Fitzgerald RC, di Pietro M, Ragunath K, et al. British Society of Gastroenterology guidelines on the diagnosis and management of Barrett's oesophagus. Gut. 2014;63(1):7–42. doi: 10.1136/gutjnl-2013-305372. [DOI] [PubMed] [Google Scholar]
- 9.Schoon E. Eradication of Barrett's neoplasia: endoscopy vs. laparoscopy. Endoscopy. 2017;49(7):629–630. doi: 10.1055/s-0043-109020. [DOI] [PubMed] [Google Scholar]
- 10.Pouw RE, Seewald S, Gondrie JJ, et al. Stepwise radical endoscopic resection for eradication of Barrett's oesophagus with early neoplasia in a cohort of 169 patients. Gut. 2010;59(9):1169–77. doi: 10.1136/gut.2010.210229. [DOI] [PubMed] [Google Scholar]
- 11.Hamade N, Sharma P. Ablation Therapy for Barrett's Esophagus: New Rules for Changing Times. Curr Gastroenterol Rep. 2017;19(10):48. doi: 10.1007/s11894-017-0589-2. [DOI] [PubMed] [Google Scholar]
- 12.Katada C, Muto M, Manabe T, et al. Esophageal stenosis after endoscopic mucosal resection of superficial esophageal lesions. Gastrointest Endosc. 2003;57(2):165–9. doi: 10.1067/mge.2003.73. [DOI] [PubMed] [Google Scholar]
- 13.Giovannini M, Bories E, Pesenti C, et al. Circumferential endoscopic mucosal resection in Barrett's esophagus with high-grade intraepithelial neoplasia or mucosal cancer. Preliminary results in 21 patients. Endoscopy. 2004;36(9):782–7. doi: 10.1055/s-2004-825813. [DOI] [PubMed] [Google Scholar]
- 14.Pech O, Behrens A, May A, et al. Long-term results and risk factor analysis for recurrence after curative endoscopic therapy in 349 patients with high-grade intraepithelial neoplasia and mucosal adenocarcinoma in Barrett's oesophagus. Gut. 2008;57(9):1200–6. doi: 10.1136/gut.2007.142539. [DOI] [PubMed] [Google Scholar]
- 15.de Jonge PJ, Spaander MC, Bruno MJ, et al. Acid suppression and surgical therapy for Barrett's oesophagus. Best Pract Res Clin Gastroenterol. 2015;29(1):139–50. doi: 10.1016/j.bpg.2014.11.005. [DOI] [PubMed] [Google Scholar]
- 16.Chang EY, Morris CD, Seltman AK, et al. The effect of antireflux surgery on esophageal carcinogenesis in patients with barrett esophagus: a systematic review. Ann Surg. 2007;246(1):11–21. doi: 10.1097/01.sla.0000261459.10565.e9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.DeMeester SR, DeMeester TR. Columnar mucosa and intestinal metaplasia of the esophagus: fifty years of controversy. Ann Surg. 2000;231(3):303–21. doi: 10.1097/00000658-200003000-00003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Knight BC, Devitt PG, Watson DI, et al. Long-term Efficacy of Laparoscopic Antireflux Surgery on Regression of Barrett's Esophagus Using BRAVO Wireless pH Monitoring: A Prospective Clinical Cohort Study. Ann Surg. 2017;266(6):1000–1005. doi: 10.1097/SLA.0000000000002019. [DOI] [PubMed] [Google Scholar]
- 19.Hofstetter WL, Peters JH, DeMeester TR, et al. Long-term outcome of antireflux surgery in patients with Barrett's esophagus. Ann Surg. 2001;234(4):532–8. doi: 10.1097/00000658-200110000-00012. discussion 538-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Maret-Ouda J, Konings P, Lagergren J, et al. Antireflux Surgery and Risk of Esophageal Adenocarcinoma: A Systematic Review and Meta-analysis. Ann Surg. 2016;263(2):251–7. doi: 10.1097/SLA.0000000000001438. [DOI] [PubMed] [Google Scholar]
- 21.Steinemann DC, Zerz A, Muller PC, et al. Laparoscopic transgastric circumferential stapler-assisted vs. endoscopic esophageal mucosectomy in a porcine model. Endoscopy. 2017;49(7):668–674. doi: 10.1055/s-0043-103407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Farrell TM, Archer SB, Metreveli RE, et al. Resection and advancement of esophageal mucosa. A potential therapy for Barrett's esophagus. Surg Endosc. 2001;15(9):937–41. doi: 10.1007/s004640080057. [DOI] [PubMed] [Google Scholar]
- 23.Kilkenny C, Browne WJ, Cuthill IC, et al. Improving bioscience research reporting: the ARRIVE guidelines for reporting animal research. PLoS Biol. 2010;8(6):e1000412. doi: 10.1371/journal.pbio.1000412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Bordeianou L, Sylla P, Kinnier CV, et al. Perineal sigmoidopexy utilizing transanal endoscopic microsurgery (TEM) to treat full thickness rectal prolapse: a feasibility trial in porcine and human cadaver models. Surg Endosc. 2015;29(3):686–91. doi: 10.1007/s00464-014-3722-4. [DOI] [PubMed] [Google Scholar]
- 25.Zdichavsky M, Krautwald M, Meile T, et al. Single-port live donor nephrectomy using a novel Curved Radius r2 Surgical System in an in vivo model. Minim Invasive Ther Allied Technol. 2015;24(2):63–7. doi: 10.3109/13645706.2014.975134. [DOI] [PubMed] [Google Scholar]
- 26.Frantzides CT, Carlson MA, Keshavarzian A, et al. Laparoscopic transgastric esophageal mucosal resection: 4-year minimum follow-up. Am J Surg. 2010;200(2):305–7. doi: 10.1016/j.amjsurg.2009.10.011. [DOI] [PubMed] [Google Scholar]
- 27.Frantzides CT, Daly SC, Frantzides AT, et al. Laparoscopic transgastric esophageal mucosal resection: a treatment option for patients with high-grade dysplasia in Barrett's esophagus. Am J Surg. 2016;211(3):534–6. doi: 10.1016/j.amjsurg.2015.12.008. [DOI] [PubMed] [Google Scholar]
- 28.Frantzides CT, Madan AK, Moore RE, et al. Laparoscopic transgastric esophageal mucosal resection for high-grade dysplasia. J Laparoendosc Adv Surg Tech A. 2004;14(5):261–5. doi: 10.1089/lap.2004.14.261. [DOI] [PubMed] [Google Scholar]
- 29.Haidry RJ, Dunn JM, Butt MA, et al. Radiofrequency ablation and endoscopic mucosal resection for dysplastic barrett's esophagus and early esophageal adenocarcinoma: outcomes of the UK National Halo RFA Registry. Gastroenterology. 2013;145(1):87–95. doi: 10.1053/j.gastro.2013.03.045. [DOI] [PubMed] [Google Scholar]
- 30.Gupta M, Iyer PG, Lutzke L, et al. Recurrence of esophageal intestinal metaplasia after endoscopic mucosal resection and radiofrequency ablation of Barrett's esophagus: results from a US Multicenter Consortium. Gastroenterology. 2013;145(1):79–86 e1. doi: 10.1053/j.gastro.2013.03.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hu Y, Puri V, Shami VM, et al. Comparative Effectiveness of Esophagectomy Versus Endoscopic Treatment for Esophageal High-grade Dysplasia. Ann Surg. 2016;263(4):719–26. doi: 10.1097/SLA.0000000000001387. [DOI] [PubMed] [Google Scholar]