Abstract
Hydrogels (HGs) are attractive matrices for cell-based cartilage tissue regeneration given their injectability and ability to fill defects with irregular shapes. However, most HGs developed to date often lack cell scale macroporosity, which restrains the encapsulated cells, leading to delayed new extracellular matrix deposition restricted to pericellular regions. Furthermore, tissue-engineered cartilage using conventional HGs generally suffers from poor mechanical property and fails to restore the load-bearing property of articular cartilage. The goal of this study was to evaluate the potential of macroporous gelatin-based microribbon (μRB) HGs as novel 3D matrices for accelerating chondrogenesis and new cartilage formation by human mesenchymal stem cells (MSCs) in 3D with improved mechanical properties. Unlike conventional HGs, these μRB HGs are inherently macroporous and exhibit cartilage-mimicking shock-absorbing mechanical property. After 21 days of culture, MSC-seeded μRB scaffolds exhibit a 20-fold increase in compressive modulus to 225 kPa, a range that is approaching the level of native cartilage. In contrast, HGs only resulted in a modest increase in compressive modulus of 65 kPa. Compared with conventional HGs, macroporous μRB scaffolds significantly increased the total amount of neocartilage produced by MSCs in 3D, with improved interconnectivity and mechanical strength. Altogether, these results validate gelatin-based μRBs as promising scaffolds for enhancing and accelerating MSC-based cartilage regeneration and may be used to enhance cartilage regeneration using other cell types as well.
Keywords: : macroporous, hydrogels, cartilage, mesenchymal stem cells, gelatin, three-dimensional
Introduction
Despite decades of research and clinical efforts, cartilage loss and damage remain the leading cause of disability among adults and pose a significant socioeconomic burden.1,2 Articular cartilage loss can be caused by trauma, sports-related injury, chronic wear and tear, and pathological conditions such as arthritis.3 Being aneural, shallow cartilage defects can often go unnoticed, whereas the avascular nature of the tissue prevents repair and causes the damage to be irreversible.4 Untreated focal cartilage defects can progress to osteoarthritis, a disease where the prevalence dramatically correlates with age and substantially contributes to healthcare costs.1 To prevent this progression, an effective treatment for focal cartilage damage must be developed. Articular cartilage serves as a shock absorber for joints that are subject to loading and high impact. Restoring such shock-absorbing properties of articular cartilage in a timely manner is an important criterion for successful cartilage regeneration.
Stem cells offer a promising potential solution for regenerating lost cartilage given their potent ability to expand and undergo chondrogenesis.5,6 Past research has investigated multiple stem cell sources such as mesenchymal stem cells (MSCs),7 umbilical cord-derived stem cells,8 adipose-derived stem cells,9 and chondrocyte-derived cartilage stem/progenitor cells.10 MSCs present a particularly attractive cell source given that they naturally serve as progenitor cells in the bone marrow for forming bone and cartilage during musculoskeletal development.11,12 MSCs express very little or no histocompatibility complex class II molecules and induce no T cell proliferation, making them nonimmunogenic.13 These properties make MSCs particularly attractive as a source for cartilage tissue engineering.
Cartilage is a load-bearing tissue, and cartilage defects are often irregular in shape with a slippery interface. Given these properties, injecting cells alone would result in rapid cell loss, poor survival, and no structural support. To overcome these issues, tissue engineering scaffolds have been widely employed as 3D matrices to enhance cartilage repair by providing transplanted cells with structural support and desirable niche cues to promote cell survival,14 engraftment,15 differentiation,16 and integration.17 Hydrogels (HGs) are attractive for cartilage repair as their sol–gel transition enables homogeneous cell mixture and makes it easier for in situ polymerization to fill cartilage defects in a minimally invasive manner.18,19 Various HG compositions have been explored to induce chondrogenesis of stem cells, including hyaluronan,20 chondroitin sulfate,21 gelatin,22 and polyethylene glycol.23
Despite the promise of HGs to enhance cartilage repair, success has been limited by several factors. First, upon polymerization, most HG networks are nanoporous, imposing physical constraints on the encapsulated cells with sizes ranging in micron range.24–27 Such physical restriction often leads to inhibited stem cell proliferation and delay in new matrix deposition.28–31 While introducing degradable matrix cues, such as matrix metalloproteinase, degradable peptides can facilitate cell-mediated degradation,30,32 MSCs are less able to degrade HGs than chondrocytes.32,33 To facilitate MSC-based cartilage formation in 3D, HGs generally need to be very soft to reduce the physical constraint that MSCs must overcome to deposit matrix and to proliferate.28,29 To overcome the physical constraint in 3D HGs, degradable porogens can be encapsulated in bulk HGs to create space, enabling cells to be deployed in a macroporous space within HGs. Our research group and others have demonstrated that such macroporosity substantially accelerates new cartilage matrix deposition by removing physical constraints.25,34,35 However, HGs generally lose integrity when subject to cyclic mechanical loading, and porogen incorporation further decreases the already weak mechanical strength of the HGs. As such, it remains a challenge to use HGs in a load-bearing environment such as articular cartilage defects.25
To overcome the limitations of HGs, our group reported a gelatin-based microribbon (μRB) scaffold that combined injectability with macroporosity while still supporting homogeneous cell encapsulation. Unlike other macroporous HGs, the intercrosslinked μRB scaffolds exhibit unique shock-absorbing capacity and maintain structural integrity when subject to cyclic mechanical loading.36 This was achieved by intercrosslinking microscale μRB HG building blocks into a highly interconnected macroporous structure, which exhibits a spring-like mechanical property upon compression. These unique mechanical properties combined with macroporosity makes μRB scaffolds an attractive scaffold for articular cartilage repair. Unlike HGs, these μRB-based scaffolds form through a two-step crosslinking process. First, the precursor solution is wet-spun into μRB-shaped building blocks and intracrosslinked to fix the morphology. These μRBs can subsequently mix with cells homogeneously, then intercrosslink into a cell-laden macroporous scaffold.36 When cultured in stem cell growth medium, the macroporosity within μRB scaffolds encourages adipose-derived stem cells to proliferate up to 30-fold by day 21.36 These results validated the advantage of introducing macroporosity in scaffolds on accelerating stem cell proliferation in vitro. However, the potential of such macroporous μRB scaffolds for supporting stem cell-based chondrogenesis and matrix deposition remains to be explored.
The goal of this study was to evaluate the ability of gelatin-based μRB scaffolds for enhancing MSC-based cartilage regeneration in vitro; gelatin HGs were included as a control. We hypothesized that the macroporosity within the 3D μRB scaffolds would accelerate and enhance deposition of new cartilage matrix by MSCs in a highly interconnected manner, with higher mechanical strength than HG scaffolds. To test our hypothesis, human MSCs were encapsulated in μRB scaffolds or HGs, and cultured in chondrogenic medium supplemented with TGF-β3 for 21 days. Outcomes were analyzed using mechanical testing, biochemical assays, gene expression, and histology.
Materials and Methods
Materials
Dimethyl sulfoxide, gelatin type-A, glutaraldehyde, methacrylic anhydride, and L-lysine hydrochloride were purchased from Sigma-Aldrich (St. Louis, MO). All materials were used as received.
HG and gelatin μRB synthesis
HG (methacrylated gelatin) precursor solution was fabricated by dissolving gelatin (10 g) in 100 mL of phosphate-buffered saline (PBS) at 50°C. Under continuous stirring at 1000 rpm, methacrylic anhydride (20 mL) was added dropwise to the solution and allowed to fully react for 2 h at 50°C. Dripping the solution into acetone (3 L) removed unreacted methacrylic anhydride and byproducts while precipitating the methacrylated gelatin. The crude product was purified by dialysis in deionized water, lyophilized, and stored at −20°C.
μRBs were synthesized by dissolving gelatin (12 g) in dimethyl sulfoxide (48 g) at 60°C under constant stirring at 60 rpm for 18 h. The viscous solution was transferred to a 60-mL syringe and aged for 18 h. The gelatin solution was ejected using a syringe pump set to 5 mL/h from a height of 1.8 m into ethanol (3.5 L) being stirred at 500 rpm. The precipitated gelatin microfiber was transferred to acetone for 3 h, where it fully dried and formed μRBs. After being transferred back to ethanol, the μRBs were chopped to length (∼3 mm) using a homogenizer. To functionalize the μRBs, they were transferred to methanol (100 mL) containing methacrylic acid N-hydroxysuccinimide ester (15 wt%) and stirred at room temperature for 18 h. Next, the μRBs were transferred to fresh methanol (200 mL) containing glutaraldehyde (0.1 wt%) and stirred vigorously for 18 h at room temperature. Unreacted glutaraldehyde was neutralized by adding L-lysine hydrochloride (1% in 200 mL PBS) and stirring for 2 h. The product was washed eight times with PBS and three times with deionized water to remove any unreacted reagents before being lyophilized and stored at −20°C.
Scanning electron microscopy
The same amount of gelatin (7.5% w/v) was used to fabricate μRB or HG scaffolds. μRBs were fabricated using wet spinning as reported previously and photocrosslinked into a bulk scaffold at 7.5 wt% in PBS containing a photoinitiator under ultraviolet light (365 nm, 4 m, 2.5 mW/c).36 A Hitachi S-3400 N variable pressure scanning electron microscope was used to image the morphology of hydrated μRB and HG scaffolds. After scaffold formation, samples were incubated in PBS at 37°C for 18 h and washed with deionized water before being loaded into the scanning electron microscope chamber. To ensure samples remained hydrated and did not dry or freeze, the sample stage was cooled gradually from room temperature to operating temperature (−25°C); simultaneously, the chamber pressure was reduced from 1 atm to 50 Pa, following a pressure–temperature curve, at which water stays in the liquid phase. Images were taken using an electron beam of 15 kV and a working distance of ∼7 mm.
Human bone marrow-derived MSC culture and expansion
Cryopreserved second-passage human MSCs (Lonza, Basel, Switzerland) were seeded in tissue-culture flasks coated with gelatin (0.1 wt%). The cells were expanded at 37°C and 5% CO2 in Dulbecco's modified Eagle's medium with 10% fetal bovine serum, 1% Penicillin–Streptomycin, and 10 ng/mL basic fibroblast growth factor (PeproTech, Rocky Hill, NJ). Cells were passaged upon 85–90% confluency and passage 6 cells were used for all experiments.
Cell encapsulation in 3D
Lyophilized HG precursor was rehydrated with PBS containing 0.05% lithium phenyl-2,4,6-trimethylbenzoylphosphinate (LAP) photoinitiator.36 Trypsinized passage-six MSCs suspended in PBS were gently mixed with the HG solution to achieve a final density of 7.5 wt% of HG to PBS and a final cell concentration of 10 million cells/mL. The HG solution was sandwiched between two glass slides (with a 2 mm spacer) and exposed to ultraviolet light (365 nm, 2 mW/cm2) for 4 min, yielding HG scaffolds. After 24 h of incubation (37°C, 5% CO2), cylindrical scaffolds (4.5 mm in diameter) were punched out using a biopsy punch and transferred to a 24-well plate.
Lyophilized μRBs were hydrated with PBS containing 0.05% LAP photoinitiator. The mixture was incubated at 37°C for 1 h to ensure that the μRBs were fully hydrated. Trypsinized passage-six MSCs suspended in PBS were gently mixed with the μRBs to achieve a final density of 7.5 wt% of μRBs to PBS and a final cell concentration of 10 million cells/mL. The cell-containing material was sandwiched between two glass slides (with a 2 mm spacer) and exposed to ultraviolet light (365 nm, 2 mW/cm2) for 4 min, producing macroporous scaffolds. After 24 h of incubation (37°C, 5% CO2) in MSC growth medium, cylindrical scaffolds (4.5 mm in diameter) were punched out using a biopsy punch and transferred to a 24-well plate.
To visualize cell viability and morphology, cell-encapsulated scaffolds were stained using a LIVE/DEAD Viability/Cytotoxicity Kit for mammalian cells (Invitrogen, Carlsbad, CA) for 30 min (37°C, 5% CO2). Images were acquired using a Zeiss fluorescence microscope (Axio Observer 3.1; Zeiss, Oberkochen, Germany) and the ZEN 2 (blue edition) software (Zeiss).
Mechanical testing
Unconfined compression tests were conducted using an Instron 5944 materials testing system (Instron Corporation, Norwood, MA) fitted with a 10 N load cell (Interface, Inc., Scottsdale, AZ). Sample constructs were tested on day 1 and 21 of culture (n = 5) The test set-up consisted of custom-made aluminum compression platens lined with polytetrafluoroethylene to minimize friction. All tests were conducted in PBS at room temperature.
Specimen diameter (∼4.5 mm) and thickness (∼2 mm) were measured using digital calipers and the material testing system's position readout, respectively. Before each test, a preload of approximately 10 mN was applied. The upper plate was then lowered at a rate of 1% strain/s to a maximum strain of 30%. Load and displacement data were recorded at 100 Hz. The compressive modulus was determined for a strain range of 20–30% from a linear curve fit of the stress versus strain curve.
Biochemical assays
The masses of the μRB and HG scaffolds (n = 5) were measured wet (prelyophilized) and dry (postlyophilized) before samples were digested in papainase solution (Worthington Biochemical, Lakewood, NJ) at 60°C for 16 h. DNA content was quantified using the PicoGreen assay (Molecular Probes, Eugene, OR) with lambda phage DNA as a standard. Sulfated glycosaminoglycan (sGAG) content was measured using the 1,9-dimethylmethylene blue dye-binding assay and shark chondroitin sulfate (Sigma-Aldrich) as a standard.
Histology and immunostaining
μRB and HG scaffolds were washed twice in PBS at 37°C, fixed in 4% paraformaldehyde for 2 h, washed twice in PBS, and transferred to 30 wt% sucrose in PBS for 18 h. The samples were then soaked in optimal cutting temperature (OCT) compound (VWR International, Radnor, PA) for 1 h and frozen in imbedding molds using liquid nitrogen vapor. Using a Microtome Cryostat (Triangle Biomedical Sciences, Durham, NC), 8 μm-thick sections were cut from the OCT-imbedded samples and stored at −20°C before use. sGAG deposition was visualized using Safranin-O staining (Sigma-Aldrich). Tissue morphology was examined with Masson Trichrome staining (Thermo Scientific, Waltham, MA). For immunostaining, sections were treated with blocking buffer (2% goat serum and 3% bovine serum albumin in 1X PBS) for 1 h and incubated overnight at 4°C in rabbit polyclonal antibody against collagen type I (ab34710, 1:100), II (ab34712, 1:80), or X (ab58632, 1:100) (Abcam, Cambridge, MA). The sections were then incubated in a secondary antibody (1:100, AlexaFluor 488 goat anti-rabbit; Invitrogen) for 1 h at room temperature. Nuclei were counterstained using Hoechst 33342 stain (Thermo Scientific) and images were taken with a Zeiss fluorescence microscope (Axio Observer 3.1) and the ZEN 2 (blue edition) software (Zeiss). Sections without primary antibody incubation served as negative controls.
Gene expression
Total RNA was extracted from cells in μRB and HG scaffolds (n = 5) using TRIzol (Invitrogen) and the RNeasy Mini Kit (Qiagen, Valencia, CA) after the samples had been homogenized mechanically using a Pellet Pestle Cordless Motor (Sigma-Aldrich). One mg of RNA from each sample was reverse transcribed into cDNA using the Superscript First-Strand Synthesis System (Invitrogen). Real-time polymerase chain reaction (PCR) was performed on an Applied Biosystems 7900 Real-Time PCR system using SYBR Green master mix (Applied Biosystems, Carlsbad, CA). The expression of genes encoding chondrogenic markers, including, Sox 9, aggrecan, and type II collagen, as well as fibroblastic markers type I collagen and type X collagen, were quantified using the ΔΔCt method.37 Gene expression levels were normalized internally to expression of the gene encoding glyceraldehyde 3-phosphate dehydrogenase. Relative fold changes represent changes in gene expression compared with the MSC control group on day 1.
Statistical analysis
All data were expressed as mean ± standard error. Statistical significance was determined through analysis of variance using Student's t-test with equal variance. p-values (two-tailed) less than 0.05 were considered statistically significant; p-values less than 0.005 were considered highly significant.
Results
μRB scaffolds exhibit macroporosity and support homogeneous cell encapsulation with high cell viability
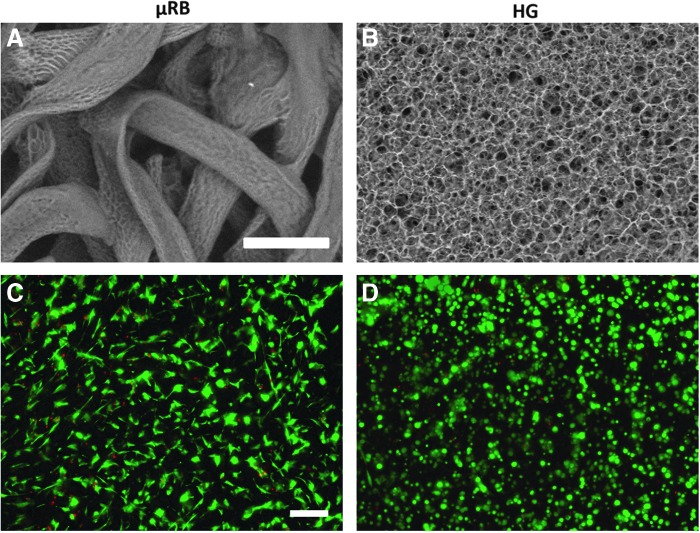
A variable pressure scanning electron microscope was used to characterize the structure and morphology of scaffolds made of μRBs or HGs. The crosslinked μRB scaffolds showed highly interconnected macroporosity (Fig. 1A), yet restricted and smaller porosity was observed in conventional HGs (Fig. 1B). Next, we examined the ability of μRB scaffolds to support homogeneous cell encapsulation in 3D and assessed cell viability by performing LIVE/DEAD staining. Twenty-four hours after encapsulation, cells in μRB scaffolds were highly viable, attached extensively on the μRBs, and were distributed homogenously in 3D (Fig. 1C). Cells encapsulated in HG scaffolds also demonstrated high cell viability, but exhibited round morphology due to physical restriction of the HG network and lack of macroporosity at the cellular scale (Fig. 1D).
FIG. 1.
Gelatin μRB scaffolds displayed improved macroporosity compared with conventional HGs while supporting MSC viability after encapsulation. (A, B) Scanning electron microscopy showed highly interconnected macroporosity in μRB scaffolds, whereas pores were more restricted and smaller in HG scaffolds. (C, D) LIVE/DEAD staining showed comparable high cell viability in both μRB and gelatin HG scaffolds. Only μRB scaffolds supported rapid cell spreading. Green: live cells; red: dead cells; Scale bars: 100 μm (A, B), 200 μm (C, D). μRB, microribbon; HG, hydrogel; MSC, mesenchymal stem cell.
μRB scaffolds accelerate increases in mechanical strength of engineered neocartilage compared with HGs
To examine and compare the ability of μRB scaffolds to induce chondrogenesis and to produce cartilage-like mechanical properties in HG scaffolds, cell-laden μRB and HG scaffolds (μRB + MSCs and HG + MSCs, respectively) were transferred to chondrogenic medium, including TGF-β, 24 h after encapsulation. Acellular scaffolds were included as a negative control for both groups (μRB-Only and HG-Only). All scaffolds were 4.5 mm in diameter and 2 mm in height. μRB scaffolds (both with and without MSCs) were opaque on day 1; HG scaffolds (both with and without MSCs) were transparent (Fig. 2A). After 21 days of culture in chondrogenic medium, the opacity of the μRB scaffolds with MSCs increased with the appearance of articular cartilage, suggesting significant matrix deposition (Fig. 2A). In contrast, the acellular μRB scaffolds had started to degrade and were losing integrity (Fig. 2A). The HGs with MSCs had lost transparency but were not as opaque as the μRB + MSC samples. The acellular HG samples looked unchanged and remained transparent (Fig. 2A). The change in gross morphology predicted a change in the compressive moduli of the scaffolds over the 21-day culture. On day 1, μRB + MSC and μRB-Only samples exhibited similar compressive moduli (9.9 ± 2.4 kPa and 5.8 ± 0.9 kPa, respectively). The HG samples were slightly stiffer: the HG + MSC samples had a modulus of 23.1 ± 0.4 kPa and the HG-Only samples had a modulus of 40.0 ± 4.9 kPa. On day 21, the μRB + MSC samples exhibited an over 20-fold increase in stiffness to 224.8 ± 19.0 kPa. The acellular μRB samples degraded during the same time, with moduli of 0.7 ± 0.4 kPa. HG + MSC samples increased to 64.6 ± 15.9 kPa, whereas HG-Only samples decreased to 39.6 ± 3.3 kPa. Because both scaffold types offered similar biochemical cues, these compressive moduli results indicated the importance of interconnected macropores in remodeling the scaffold and translating matrix deposition into cartilage-mimicking mechanical properties.
FIG. 2.

μRB scaffolds, but not HGs, led to accelerated increases in compressive moduli of MSC-based neocartilage formation after 21 days of culture in chondrogenic medium in vitro. (A) MSC-seeded μRBs or HG samples showed increased opacity over time, suggesting matrix deposition by MSCs in 3D. Acellular μRB scaffolds degraded and lost integrity over time while no significant change was observed in acellular HG controls. (B) Unconfined compressive testing revealed a 20-fold increase in compressive modulus in MSC-seeded μRBs, while only a three-fold increase was observed in MSC-seeded HGs. The moduli of acellular μRB scaffolds decreased significantly while the compressive modulus of acellular HGs remained the same. Scale bar: 2 mm. ns, nonsignificant (p > 0.05), **p < 0.01, ****p < 0.0001.
Macroporous μRB scaffolds increase cell proliferation and matrix production
To assess how the macroporosity of μRB scaffolds influence MSC proliferation, DNA content of the scaffolds was quantified using a PicoGreen assay 24 h after encapsulation and after 21 days of incubation (Fig. 3A). The initial DNA content was similar between the μRB and HG groups, indicating a comparable cell seeding number and efficient cell encapsulation (Fig. 3A). After 21 days of culture in chondrogenic medium, the μRB scaffolds allowed almost four-fold proliferation (247.69 ± 34.16 μg/g). In contrast, the HG samples did not support significant cell proliferation (102.82 ± 14.16 μg/g). The 1,9-dimethylmethylene blue assay was used to quantify the deposition of sGAG, a major component of cartilage (Fig. 3B). On day 1, negligible amounts of sGAGs were detected in both μRB and conventional HG scaffolds (Fig. 3B). By day 21, both μRB and HG scaffolds induced a substantial amount of sGAG deposition, with μRB induced over 60% more sGAG deposition than the HG group (8581.30 ± 1356.67 μg/g and 5319.93 ± 514.40 μg/g, respectively).
FIG. 3.
The μRB scaffolds led to significantly enhanced MSC proliferation and cartilage matrix production compared with HGs in 3D. (A) DNA content per wet weight increased ∼3-fold in μRB scaffolds, and no significant change in DNA was observed in HGs. (B) Quantitative GAG assay showed both μRB and HG scaffolds led to significantly enhanced GAG deposition by MSCs over 21 days, with μRB scaffolds leading to ∼50% more total sGAG deposition than HG scaffolds. ns = nonsignificant (p > 0.05), **p < 0.01, ****p < 0.0001.
Macroporous μRB scaffolds improve the distribution and interconnectivity of newly deposited cartilage compared with HGs
Scaffold cross-sections were stained with Safranin-O to reveal the morphology and distribution of sGAGs in cellular and acellular samples after 21 days in culture (Fig. 4A). At low magnification, μRB + MSC samples contained sGAGs distributed throughout the scaffold in a dense manner (Fig. 4A). Higher magnification revealed the highly interconnected morphology of the matrix. The μRB-Only group showed only the background of the μRB scaffold (Fig. 4A). At low magnification, the HG + MSC group contained dense pericellular sGAG deposition throughout the scaffold (Fig. 4A). A higher magnification revealed that matrix deposition was limited to the pores in which the cells were trapped, signifying that the matrix is not interconnected. The HG-Only group had morphology similar to that of acellular samples (Fig. 4A).
FIG. 4.
Macroporous μRB scaffolds, and not HGs, enabled increased amounts of new cartilage matrix deposition in a highly interconnected manner throughout the 3D scaffold at day 21. (A) Safranin-O staining depicted μRB + MSC group with enhanced interconnected GAG deposition, whereas HG + MSCs had limited GAG deposition restricted to pericellular regions. Acellular samples were stained as background controls. (B) Trichrome staining of total collagen showed a similar trend to the GAG staining. Red: GAG; blue: collagen. For both stainings, low-magnification (upper row) and high-magnification (lower row) images are provided. Scale bar in upper row: 1 mm; scale bar in lower row: 200 μm.
Masson's Trichrome was used to stain the collagen deposition in the scaffolds after 21 days of culture (Fig. 4B). Collagen deposition was similar to the sGAG distribution in cellular samples after 21 days (Fig. 4B). The μRB + MSC group was marked by robust and interconnected collagen distribution throughout the scaffold (Fig. 4B). The HG + MSC group permitted dense pericellular collagen formation throughout the scaffold that was limited to the isolated pores in which the cells were trapped (Fig. 4B).
μRB scaffolds direct a hyaline cartilage phenotype, as indicated by gene expression and interconnected collagen type II matrix deposition
Gene expression (Supplementary Fig. S1; Supplementary Data are available online at www.liebertpub.com/tea) and immunostaining (Fig. 5 and Supplementary Fig. S2) were used to characterize the cartilage phenotype generated by MSC-seeded μRB and HG scaffolds. Gene expression was monitored through real-time PCR on day 14 and normalized to day 1 samples (Supplementary Fig. S1). The hyaline cartilage markers, aggrecan and collagen type II, increased by >1000-fold and by ∼2,000,000-fold in the μRB and HG samples, respectively (Supplementary Fig. S1). While collagen type X and type I expression was upregulated (60,000-fold and 10-fold, respectively) in both μRB and HG samples, their increases were orders of magnitude lower than that of collagen type II.
FIG. 5.

The μRB scaffolds promoted deposition of collagen that mimics phenotype of articular cartilage. Immunofluorescence staining indicated that collagen produced by MSCs was predominantly type II collagen, a major constituent of native cartilage. Collagen X, a hypertrophy cartilage marker, was observed in HG scaffolds, but not in the μRB group. Minimal collagen I deposition was observed in both μRB and HG samples, suggesting the absence of an undesirable fibrocartilage phenotype. Scale bar: 400 μm.
These findings were consistent with immunostaining results: collagen type II immunostaining (Fig. 5 and Supplementary Fig. S2) revealed matrix morphology similar to that indicated by Safranin-O and Masson's Trichrome staining (Fig. 4). The μRB + MSC scaffolds induced dense, interconnected collagen type II matrix deposition (Fig. 5). In the HG + MSC group, collagen type II deposition occurred only in the pericellular region and was not interconnected (Fig. 5). Staining for collagens type I and X was minimal or negative for all cellular samples (Fig. 5), despite upregulation of the RNA encoding these proteins (Supplementary Fig. S1), indicating a hyaline cartilage phenotype. Acellular control samples did not stain for any collagen subtype (Supplementary Fig. S3).
Gene expression captures cellular fates at one specific time point, whereas histology and immunostaining show the accumulative total amount of matrix over time. As such, the gene expression pattern does not necessarily agree with the matrix deposition observed through biochemical assays or histology. In our study, MSCs in both μRB and HG groups showed comparable levels of cartilage markers (i.e., Sox 9 and AGG), indicating that MSCs are upregulating chondrogenic markers in a similar way. However, the actual matrix deposition not only depends on the cells, but also depends on how permissive the scaffold is in allowing new matrix deposition. In HGs, the polymeric network is nanoporous, and cells must degrade the matrix first before any new matrix can be deposited. In contrast, the μRB-based scaffolds already contain macroporosity and cells can deposit new matrix right away without physical constraint. Our data indeed show that, despite comparable chondrogenic marker expressions (Supplementary Fig. S1), μRB-based scaffolds resulted in increased amount of new cartilage formation with improved interconnectivity (Figs. 3–5).
Discussion
In this study, we evaluated the potential of macroporous μRB scaffolds for accelerating and enhancing MSC-based cartilage formation in vitro. Compared with HG controls, MSC-seeded μRB scaffolds exhibited a >20-fold increase in compressive modulus after only 21 days of culture to a range comparable to that of native cartilage (Fig. 2B). In contrast, HG scaffolds led to a less than 3-fold increase in compressive moduli, with inferior mechanical properties (Fig. 2B). Furthermore, macroporosity within the μRB scaffolds increased the quantity and homogeneous distribution of newly deposited cartilage matrix (Figs. 3 and 4). Altogether, these results highlight the advantages of macroporous μRB scaffolds over nanoporous HGs. This study demonstrates that macroporosity helps accelerate new cartilage deposition and improves the interconnectivity of newly formed cartilage by MSCs in 3D (Figs. 4 and 5). This improvement in cartilage structure also translates to improved mechanical functions, with compressive moduli of engineered cartilage approaching the range of compressive moduli of native articular cartilage (Fig. 2B).
Articular cartilage protects underlying bone tissue in load-bearing joints from chronic wear3 and hence endures significant stress. The compressive modulus of normal articular cartilage typically ranges from 500 to 900 kPa.38 Given the importance of cartilage for shock absorbance and load bearing, it is critical for engineered cartilage to restore mechanical strength to the tissue as quickly as possible. However, previous attempts using HGs for cartilage tissue engineering generally led to compressive moduli that were one order of magnitude lower than the physiological range of native articular cartilage.32,39 Even prolonging the in vitro culture period up to 8 weeks only led to average moduli ranging from 50 to 60 kPa.32 Similar to previous reports, in this study, MSC-seeded HG scaffolds also had a compressive modulus that was one order of magnitude lower than that of native cartilage (Fig. 2B). While increasing HG concentration can lead to higher initial stiffness, this increased concentration leads to even more physical restrictions to cells encapsulated in 3D, which is undesirable for new cartilage deposition.24 Alternatively, soft HGs provide a slightly more permissive network for cells, but further decrease the already weak mechanical strength of HGs.24 This dilemma greatly limits the application of HGs to engineering load-bearing tissues such as cartilage. Unlike HGs, while the initial compressive modulus of the macroporous μRB scaffold was low, intercrosslinking among the μRB building blocks confers upon great shock-absorbing capacity when the macroporous scaffold is subject to cyclic loading.36 In this study, the use of μRB scaffolds led to a rapid enhancement of mechanical strength approaching the range of healthy cartilage (224.8 ± 19.0 kPa) after only 3 weeks of culture in vitro. This efficacy in restoration is most likely due to the inherent macroporosity of the μRB scaffold (Fig. 1B), which removes the physical restrictions to which cells are subject in nanoporous HGs and allows fast deposition of new cartilage matrix in 3D in a highly interconnected manner without the need for HG degradation.
An ideal scaffold for cartilage regeneration should be degradable but also support rapid new cartilage formation to allow fast restoration of cartilage structures and load-bearing function. It is important that degradation of the initial scaffold will not compromise the long-term mechanical strength and stability of the newly regenerated cartilage. Our data show that the mechanical strength of cell-seeded μRBs increased ∼20-fold within just 3 weeks due to the rapid cartilage deposition in μRB scaffolds (Fig. 2B). As such, mechanical strength of the resulting cartilage no longer depends on the initial scaffold, and degradation-induced decreases in compressive moduli become negligible. We showed that acellular μRB scaffolds degraded over 3 weeks, with loss of integrity and decrease in compressive moduli. In contrast, cell-seeded μRBs maintained structural integrity and resulted in an over 20-fold increase in compressive moduli (from ∼10 kPa to over 200 kPa). These results confirm that long-term mechanical strength of engineered cartilage is dominated by the newly deposited matrix rather than the original μRB scaffold. Altogether, our results demonstrate the μRB scaffold is degradable, yet degradation does not interfere with long-term mechanical strength due to an accelerated enhancement of the compressive modulus by cell-secreted matrix.
The dramatic increase in the mechanical strength of the μRB group within a short time compared with the HG group (Fig. 2B) is not only a result of the amount of matrix, but also the distribution of the newly deposited matrix. Quantitative sGAG assays revealed that MSCs in the μRB group produced 60% more sGAG than MSCs in the HG group (Fig. 3B), yet the compressive moduli of μRB group were 250% higher than those of the HG group (Fig. 2B). While biochemical assays show the total amount of newly deposited matrix, histological staining indicates the distribution of newly deposited tissue. While μRB scaffolds led to dense and highly interconnected matrix deposition, newly deposited cartilage matrix in the HG group was limited to pericellular regions (Fig. 4). Other studies using various HGs typically reported pericellular deposition of new cartilage, due to the physical restriction of HG network.39,40 These results indicate that μRB scaffolds increased both the amount (Fig. 3B) and the interconnected distribution (Fig. 4) of neocartilage, which together contribute to a rapid improvement in the mechanical properties (Fig. 2B) of engineered cartilage that cannot be achieved by nanoporous HGs.
Another strategy to introduce macroporosity in HGs is using porogens, which are microspheres that can be encapsulated within HGs and degraded to yield pores.25,34,35 Our group and others have shown that porogen-induced macroporosity can accelerate and enhance new cartilage tissue formation by cells in 3D.25,34,35 However, porogen-induced macroporosity further decreases the already weak mechanical strength of conventional HGs, which leads to undesirable collapses due to the scaffold's inability to resist cell traction forces.35 To achieve interconnected pores, porogens need to be packed closely, leading to further decreases in HG mechanical strength and stability. In contrast, macroporosity within μRB scaffolds is inherently interconnected regardless of μRB density, and the intercrosslinking of μRBs renders the resulting scaffolds mechanically stable and able to absorb shock.36
Driven by the need to introduce macroporosity into scaffolds, researchers have developed various technologies to fabricate solid macroporous scaffolds using collagen,41 silk,42 poly (2-hydroxyethyl methacrylate),43 poly (lactic-co-glycolic acid),44 and hydroxyapatite.45 Unlike HGs, these sponge-like solid scaffolds have high mechanical strength. However, the fabrication process for these scaffolds is not cell friendly and cells can only be seeded after fabrication, leading to heterogeneous cell distribution in 3D.46 Another advantage of μRB scaffold is the ease of achieving homogeneous cell distribution in 3D macroporous matrices (Fig. 1), while maintaining high mechanical stability and rapid enhancement of mechanical strength approaching the range of healthy cartilage (Fig. 2).
In summary, our results validate μRB scaffolds as a promising scaffold for supporting stem cell-based cartilage regeneration by overcoming key limitations associated with conventional HGs. While both μRB and HG scaffolds allowed upregulations of chondrogenic markers, the macroporosity within μRB scaffolds provide a more permissive 3D niche that allow for rapid new cartilage deposition with improved interconnectivity throughout the scaffolds. Such improvements in cartilage structures also translates to improved mechanical properties of resulting cartilage, with MSC-seeded μRB scaffolds leading to over 20-fold increase in compressive moduli to a range that approaches that of native articular cartilage after only 21 days of culture in vitro. The ability to support direct and homogeneous cell encapsulation also makes μRBs a more desirable scaffold choice than solid macroporous scaffolds. Future work will evaluate the potential of such μRB scaffolds in supporting cartilage regeneration in vivo using animal models. While this study focused on MSCs as a model cell type, this strategy may be easily applied to enhance cartilage formation with other stem cell types.
Supplementary Material
Acknowledgments
The authors would like to thank NIH R01DE024772-01 (F. Y.), National Science Foundation CAREER award program (CBET-1351289) (F. Y.), California Institute for Regenerative Medicine Tools and Technologies award (Grant #TR3-05569) (F. Y.), Stanford Chem-H Institute New Materials for Applications in Biology and Medicine Seed Grant (F. Y.), Stanford Child Health Research Institute (F. Y.), California Institute for Regenerative Medicine predoctoral fellowship (B. C.), and NIH NRSA predoctoral fellowship (5F31DE025788-03) (B. C.) for support. They also thank Dr. Lydia-Marie Joubert from the Cell Sciences Imaging Facility at Stanford for use of, and assistance with, the Hitachi electron microscope. Finally, they thank Anthony Behn from the Department of Orthopedic Surgery at Stanford for his assistance with mechanical testing.
Disclosure Statement
No competing financial interests exist.
References
- 1.Griffin T.M., and Guilak F. The role of mechanical loading in the onset and progression of osteoarthritis. Exer Sport Sci Rev 33, 195, 2005 [DOI] [PubMed] [Google Scholar]
- 2.Lai J.H., Kajiyama G., Smith R.L., Maloney W., and Yang F. Stem cells catalyze cartilage formation by neonatal articular chondrocytes in 3D biomimetic hydrogels. Sci Rep 3, 3553, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Sophia Fox A.J., Bedi A., and Rodeo S.A. The Basic Science of Articular Cartilage: structure, Composition, and Function. Sports Health 1, 461, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wang T., Lai J.H., Han L.H., Tong X., and Yang F. Chondrogenic differentiation of adipose-derived stromal cells in combinatorial hydrogels containing cartilage matrix proteins with decoupled mechanical stiffness. Tissue Eng A 20, 2131, 2014 [DOI] [PubMed] [Google Scholar]
- 5.Guilak F., Awad H.A., Fermor B., Leddy H.A., and Gimble J.M. Adipose-derived adult stem cells for cartilage tissue engineering. Biorheology 41, 389, 2004 [PubMed] [Google Scholar]
- 6.Zuk P.A., Zhu M., Mizuno H., et al. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng 7, 211, 2001 [DOI] [PubMed] [Google Scholar]
- 7.Pittenger M.F., Mackay A.M., Beck S.C., et al. Multilineage potential of adult human mesenchymal stem cells. Science 284, 143, 1999 [DOI] [PubMed] [Google Scholar]
- 8.Klontzas M.E., Kenanidis E.I., Heliotis M., Tsiridis E., and Mantalaris A. Bone and cartilage regeneration with the use of umbilical cord mesenchymal stem cells. Expert Opin Biol Ther 15, 1541, 2015 [DOI] [PubMed] [Google Scholar]
- 9.Ogawa R., and Mizuno S. Cartilage regeneration using adipose-derived stem cells. Curr Stem Cell Res Ther 5, 129, 2010 [DOI] [PubMed] [Google Scholar]
- 10.Jiang Y., Cai Y., Zhang W., et al. Human Cartilage-Derived Progenitor Cells From Committed Chondrocytes for Efficient Cartilage Repair and Regeneration. Stem Cells Transl Med 5, 733, 2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gupta P.K., Das A.K., Chullikana A., and Majumdar A.S. Mesenchymal stem cells for cartilage repair in osteoarthritis. Stem Cell Res Ther 3, 25, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hollander A.P., Dickinson S.C., and Kafienah W. Stem Cells and Cartilage Development: complexities of a Simple Tissue. Stem Cells (Dayton, Ohio) 28, 1992, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Machado C.d.V., Telles P.D.d.S., and Nascimento I.L.O. Immunological characteristics of mesenchymal stem cells. Rev Bras Hematol Hemoter 35, 62, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Spiller K.L., Maher S.A., and Lowman A.M. Hydrogels for the Repair of Articular Cartilage Defects. Tissue Eng B Rev 17, 281, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Cushing M.C., and Anseth K.S. Materials science. Hydrogel cell cultures. Science 316, 1133, 2007 [DOI] [PubMed] [Google Scholar]
- 16.Toh W.S., Lee E.H., Guo X.-M., et al. Cartilage repair using hyaluronan hydrogel-encapsulated human embryonic stem cell-derived chondrogenic cells. Biomaterials 31, 6968, 2010 [DOI] [PubMed] [Google Scholar]
- 17.Wang D.-A., Varghese S., Sharma B., et al. Multifunctional chondroitin sulphate for cartilage tissue-biomaterial integration. Nat Mater 6, 385, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.DeForest C.A., Sims E.A., Anseth K.S., and Click P.-F. Hydrogels with Independently Tunable Mechanics and Chemical Functionality for 3D Cell Culture. Chem Mat 22, 4783, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Nii M., Lai J.H., Keeney M., et al. The effects of interactive mechanical and biochemical niche signaling on osteogenic differentiation of adipose-derived stem cells using combinatorial hydrogels. Acta Biomater 9, 5475, 2013 [DOI] [PubMed] [Google Scholar]
- 20.Nettles D.L., Vail T.P., Morgan M.T., Grinstaff M.W., and Setton L.A. Photocrosslinkable hyaluronan as a scaffold for articular cartilage repair. Ann Biomed Eng 32, 391, 2004 [DOI] [PubMed] [Google Scholar]
- 21.Varghese S., Hwang N.S., Canver A.C., Theprungsirikul P., Lin D.W., and Elisseeff J. Chondroitin sulfate based niches for chondrogenic differentiation of mesenchymal stem cells. Matrix Biol J Int Soc Matrix Biol 27, 12, 2008 [DOI] [PubMed] [Google Scholar]
- 22.Awad H.A., Wickham M.Q., Leddy H.A., Gimble J.M., and Guilak F. Chondrogenic differentiation of adipose-derived adult stem cells in agarose, alginate, and gelatin scaffolds. Biomaterials 25, 3211, 2004 [DOI] [PubMed] [Google Scholar]
- 23.Elisseeff J., Anseth K., Sims D., et al. Transdermal photopolymerization of poly(ethylene oxide)-based injectable hydrogels for tissue-engineered cartilage. Plast Reconstr Surg 104, 1014, 1999 [DOI] [PubMed] [Google Scholar]
- 24.Erickson I.E., Kestle S.R., Zellars K.H., et al. High mesenchymal stem cell seeding densities in hyaluronic acid hydrogels produce engineered cartilage with native tissue properties. Acta Biomater 8, 3027, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Dadsetan M., Hefferan T.E., Szatkowski J.P., et al. Effect of Hydrogel Porosity on Marrow Stromal Cell Phenotypic Expression. Biomaterials 29, 2193, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Han L.H., Conrad B., Chung M.T., et al. Winner of the Young Investigator Award of the Society for Biomaterials at the 10th World Biomaterials Congress, May 17–22, 2016, Montreal QC, Canada: microribbon-based hydrogels accelerate stem cell-based bone regeneration in a mouse critical-size cranial defect model. J Biomed Mater Res A 104, 2016, 1321 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hollister S.J. Porous scaffold design for tissue engineering. Nat Mater 4, 518, 2005 [DOI] [PubMed] [Google Scholar]
- 28.Chung C., Mesa J., Randolph M.A., Yaremchuk M., and Burdick J.A. Influence of gel properties on neocartilage formation by auricular chondrocytes photoencapsulated in hyaluronic acid networks. J Biomed Mater Res A 77A, 518, 2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kim I.L., Mauck R.L., and Burdick J.A. Hydrogel design for cartilage tissue engineering: a case study with hyaluronic acid. Biomaterials 32, 8771, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Feng Q., Zhu M., Wei K., and Bian L. Cell-mediated degradation regulates human mesenchymal stem cell chondrogenesis and hypertrophy in MMP-sensitive hyaluronic acid hydrogels. PLoS One 9, e99587, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Carter D.R., and Beaupré G.S. Skeletal Function and Form: Mechanobiology of Skeletal Development, Aging, and Regeneration. Cambridge: Cambridge University Press, 2000 [Google Scholar]
- 32.Chung C., Beecham M., Mauck R.L., and Burdick J.A. The influence of degradation characteristics of hyaluronic acid hydrogels on in vitro neocartilage formation by mesenchymal stem cells. Biomaterials 30, 4287, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Nicodemus G.D., and Bryant S.J. Cell encapsulation in biodegradable hydrogels for tissue engineering applications. Tissue Eng B Rev 14, 149, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bencherif S.A., Braschler T.M., and Renaud P. Advances in the design of macroporous polymer scaffolds for potential applications in dentistry. J Periodont Implant Sci 43, 251, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Han L.H., Lai J.H., Yu S., and Yang F. Dynamic tissue engineering scaffolds with stimuli-responsive macroporosity formation. Biomaterials 34, 4251, 2013 [DOI] [PubMed] [Google Scholar]
- 36.Han L.-H., Yu S., Wang T., Behn A.W., and Yang F. Tissue engineering: microribbon-like elastomers for fabricating macroporous and highly flexible scaffolds that support cell proliferation in 3D. Adv Funct Mater 23, 266, 2013 [Google Scholar]
- 37.Schmittgen T.D., and Livak K.J. Analyzing real-time PCR data by the comparative CT method. Nat Protoc 3, 1101, 2008 [DOI] [PubMed] [Google Scholar]
- 38.Athanasiou K.A., Rosenwasser M.P., Buckwalter J.A., Malinin T.I., and Mow V.C. Interspecies comparisons of in situ intrinsic mechanical properties of distal femoral cartilage. J Orthop Res 9, 330, 1991 [DOI] [PubMed] [Google Scholar]
- 39.Sridhar B.V., Brock J.L., Silver J.S., Leight J.L., Randolph M.A., and Anseth K.S. Development of a cellularly degradable PEG hydrogel to promote articular cartilage extracellular matrix deposition. Adv Healthc Mater 4, 702, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Sridhar B.V., Doyle N.R., Randolph M.A., and Anseth K.S. Covalently tethered TGF-beta1 with encapsulated chondrocytes in a PEG hydrogel system enhances extracellular matrix production. J Biomed Mater Res A 102, 4464, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Nehrer S., Breinan H.A., Ramappa A., et al. Chondrocyte-seeded collagen matrices implanted in a chondral defect in a canine model. Biomaterials 19, 2313, 1998 [DOI] [PubMed] [Google Scholar]
- 42.Mandal B.B., Grinberg A., Gil E.S., Panilaitis B., and Kaplan D.L. High-strength silk protein scaffolds for bone repair. Proc Natl Acad Sci U S A 109, 7699, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Chirila T.V., Constable I.J., Crawford G.J., et al. Poly(2-hydroxyethyl methacrylate) sponges as implant materials: in vivo and in vitro evaluation of cellular invasion. Biomaterials 14, 26, 1993 [DOI] [PubMed] [Google Scholar]
- 44.Thevenot P.T., Nair A.M., Shen J.H., Lotfi P., Ko C.Y., and Tang L.P. The effect of incorporation of SDF-1 alpha into PLGA scaffolds on stem cell recruitment and the inflammatory response. Biomaterials 31, 3997, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Fierz F.C., Beckmann F., Huser M., et al. The morphology of anisotropic 3D-printed hydroxyapatite scaffolds. Biomaterials 29, 3799, 2008 [DOI] [PubMed] [Google Scholar]
- 46.Melchels F.P.W., Tonnarelli B., Olivares A.L., et al. The influence of the scaffold design on the distribution of adhering cells after perfusion cell seeding. Biomaterials 32, 2878, 2011 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.