Abstract
Aims:
We sought to examine whether disturbances in central and peripheral circadian rhythms were related to the experience of fatigue in patients with chronic liver disease (CLD).
Methods:
Fatigued and non-fatigued patients with compensated CLD were enrolled in a prospective pilot study. Patients underwent a one week evaluation of free-living sleep and physical activity patterns, followed by a 24-hour admission, during which they underwent serial blood sampling, polysomnography, a 6-minute walk test and continuous core temperature measurements under standardized conditions. Blood samples were analyzed for liver tests, melatonin levels, lipids, and cortisol. Circadian rhythms were analyzed using single cosinor analyses.
Results:
Six fatigued and six non-fatigued patients were studied; five participants had cirrhosis. Fatigue severity was positively associated higher peak melatonin levels (rho=0.59, p=0.04) and a delay in night-time melatonin peak and inversely associated with sleep efficiency (rho=−0.63, p=0.04). Polysomnography, 6-minute walk test, and core temperature measurements did not differ significantly between the fatigued and non-fatigued patients. Although liver enzymes, bilirubin and albumin demonstrated a circadian pattern, it was not associated with fatigue. Fatigued patients showed a blunted and delayed cortisol rhythm and fatigue was strongly correlated with cortisol amplitude (rho=−0.77, p=0.004) and phase (r=−0.66, p=0.02).
Conclusion:
Subtle aberrations in melatonin and adrenal circadian rhythms, as well as reduced sleep efficiency, likely contribute to fatigue in patients with CLD. These abnormalities may ultimately be a therapeutic target to improve quality of life for fatigued patients with CLD.
Keywords: fatigue, liver disease, sleep, circadian rhythms, melatonin, cortisol
Introduction
Fatigue is a significant symptom in chronic liver disease (CLD), can severely impact quality of life, and is not correlated with the severity of liver disease[1, 2, 3, 4, 5]. The pathogenesis of fatigue in chronic liver disease is largely unknown, and little progress has been made in the treatment of this frequently debilitating symptom[6]. Cirrhosis and hepatic encephalopathy have a known association with sleep disturbance and altered day-night pattern, which could contribute to fatigue[7]. However, cirrhotic patients tend to have complex multi-organ dysfunction, and findings in them may not be generalizable to all patients with liver disease. Yet, fatigue occurs in patients with non-cirrhotic liver disease and remains poorly understood.
The circadian system is important for nearly every physiologic and behavioral system in the body[8]. Apart from the central master oscillator in the suprachiasmatic nucleus (SCN) there is a growing body of literature on the presence of peripheral circadian clocks, particularly in the liver and heart[9]. Studies have identified a set of rhythmically-expressed, clock-regulated genes in the livers of mice, and animal models of liver fibrosis have demonstrated alterations in the circadian rhythm of hepatic clock genes[10, 11, 12]. Although the master oscillator in the SCN is known to be entrained by light through retinal input, it is less clear how other external signals such as nutrition and temperature affect the central and peripheral clocks. Importantly, the peripheral oscillators may be coordinated with the master SCN clock through neural signaling and by variation in the level of melatonin[13].
Melatonin (N-acetyl-5-methoxytryptamine) is a neurohormone produced predominantly in the pineal gland but data also suggest it is produced in lymphoid organs, the gut, and liver[14, 15]. Melatonin secretion peaks at nighttime and is virtually undetectable during the day. Melatonin has been found to play a role in multiple functions at the cellular, organ, and organism level, including synchronization of peripheral clocks, an antioxidant and anti-inflammatory effect, and control of sleep-wake cycles[16].
In patients with cirrhosis, alterations in plasma melatonin levels and rhythm have been demonstrated and are explained, at least in part, by impaired hepatic catabolism[17, 18, 19]. Following liver transplantation, these abnormalities may be corrected[20]. In advanced cirrhosis, fatigue has been linked to hepatic encephalopathy, sleep disturbance, and altered circadian rhythms[21]. In non-cirrhotics, there is a dearth of information on circadian abnormalities and no data on their possible association with fatigue.
We hypothesized that circadian abnormalities in either the central clock or the peripheral hepatic clock occur in patients across all stages of liver disease and that desynchronization of peripheral and central rhythms may be a possible cause of fatigue. We performed a prospective pilot study to define the role of circadian rhythms and sleep-wake abnormalities in the physiology of liver disease-related fatigue.
Methods
Patient population
Adult patients (age > 18) with CLD of any etiology were eligible for inclusion. Exclusion criteria included decompensated cirrhosis, untreated or uncontrolled comorbidities, active substance abuse, fatigue-causing medications, travel across time zones during the study period, pregnancy/breastfeeding, and inability to provide informed consent. Patients were stratified by their self-reported fatigue status as fatigued or non-fatigued, and a similar number of cirrhotics were recruited in both groups. This study was approved by the NIDDK/NIAMS Institutional Review Board on March 26, 2013 (clinicaltrials.gov NCT01867385) and all subjects provided written informed consent.
Assessment and Quantitation of Fatigue
Fatigue was assessed using the Patient Reported Outcomes Measurement Information System (PROMIS) fatigue SF7a questionnaire, administered at the time of enrollment[22, 23]. The questionnaire consists of 7 questions rating symptoms over the prior seven days on a scale of 1-5, with the total score ranging from 7-35; higher numbers indicate more severe fatigue. For dichotomous analyses, we defined a priori as fatigued patients with verbal indication of fatigue for at least 6 months and with an SF7 score > 13. The SF7 cut-off was based on a preliminary analysis in a large sample of outpatients with chronic liver disease. In brief, 116 outpatients with varying etiologies and severity of liver disease (age 55±14 years, 59% male, 81% viral hepatitis, 15% cirrhosis) were asked if they have fatigue (as a yes/no questions) and were administered the SF7 at the same time. 52 patients (45%) reported having fatigue and their SF7 score was significantly higher than non-fatigued subjects (20±5 vs. 11±4, p<0.001). An SF7 cutoff > 13 (identified using ROC analysis, AUROC=0.9, CI=0.85-0.96) had a sensitivity of 90.4% to detect self-reported fatigue, with a specificity of 75.8%.
Study Procedures
To assess free-living sleep quality and activity patterns, subjects were provided a small, watch-like accelerometer (GT3X+ v.3.2.1, Actigraph, Pensacola, FL), and wore it on the non-dominant wrist at home for seven days[24, 25]. Following that week, subjects were admitted to the NIH Clinical Center for a 24-hour evaluation under standardized conditions, including uniform sleep and wake times, meal timing and composition, and light exposure. Light intensity was not measured but was similar for all patients during the admission. Table 1 details the protocolized admission schedule. Subjects underwent standard EEG and polysomnography in the sleep lab, with measurement of respiratory patterns, EKG, and leg movement. Detailed questionnaires on fatigue, caffeine intake, sleep disturbance, depression, and global health were administered during the admission[26]. Fasting blood was drawn for baseline testing with additional blood samples drawn every 2-3 hours throughout the 24 hour period, for a total of 10 time points, to allow for measurement of melatonin and circadian biomarkers. Night time blood draws were done through an extension tubing without the need to wake the subjects. Samples were separated and stored in −80C for subsequent analysis. The central circadian rhythm was assessed by continuous measurement of core body temperature via a swallowed temperature-monitoring capsule (Vital Sense, Philips Respironics, Bend, OR). Prior to discharge, a six-minute walk test was performed to assess physical fitness.
Table 1:
Protocolized Admission Schedule
| Day One | |
|---|---|
| 7-8am | Breakfast at home (guidelines per dietician) |
| 8:30am | Arrive at hospital ward, swallow capsule |
| 9am-noon | Admission H&P |
| Noon | Standardized Lunch |
| 1pm | 6 minute walk test |
| 2pm | Start serial blood draws |
| 2:30pm | place head leads, 20 min baseline EEG |
| 6pm | Standardized Dinner |
| 10pm | Polysomnography begins |
| Day Two | |
| 6am | Fasting Bloodwork |
| 7-8am | Standardized Breakfast |
| 10am | Questionnaires |
| Noon | Standardized Lunch |
Biomarker Measurements
Serum levels of liver enzymes, bilirubin, albumin, cortisol, insulin, free fatty acids, and lipids were measured by the NIH Clinical Center Department of Laboratory Medicine using standard techniques. Immunoassays were used to measure serum levels of melatonin (GenWay Biotech, San Diego, CA), IL-6 (V-Plex human IL-6 kit, Meso Scale Discovery, Rockville, MD), corticotropin releasing factor (CRF) (Phoenix Pharmaceuticals, Burlingame, CA), cortisol binding globulin (CBG) (BioVendor, Asheville, NC), factor VII and alpha-1 antitrypsin (A1AT) (Abcam, San Francisco, CA). All assays were performed in duplicate and according to the manufacturer’s instructions.
Statistical Analyses
Melatonin levels were log-transformed for parametric tests. Continuous variables were compared between the fatigued and non-fatigued groups using Student’s t-test or Mann-Whitney U test, as appropriate. For serum biomarkers with circadian rhythmicity we performed a single cosinor analysis with a predefined period of 24 hours and from it determined the MESOR (midline statistic of rhythm, a mean adjusted for the rhythmic changes), amplitude (the variation around the midline) and acrophase (location of the peak)[27]. Given the small number of participants and the anticipated inter-individual variability, we did not group subjects together, but extracted MESOR, amplitude, and acrophase for each individual and each biomarker separately. These were then compared between the two groups using Student’s t-test (dichotomous analysis), and were correlated with individual fatigue scores across all subjects (continuous analysis). Effect size was calculated using Cohen’s d for Student’s t-test, rank-biserial correlation coefficient for Mann-Whitney U (using the Wendt formula), and Cramer’s v for categorical tests.
In the absence of preliminary data on which power calculations could be based, the sample size was determined based on feasibility, cost, and the pilot design with a plan for exploratory analysis of the data[28].
Analyses were performed in R [R Core Team (2014). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL http://www.R-project.org/], SAS (Version 9.3, Cary, NC, USA), SPSS Statistics (Version 19, IBM) and GraphPad Prism (Version 6).
Results
Twelve patients were enrolled, 6 with fatigue and 6 without (Table 2). In the fatigued group, four patients had chronic hepatitis C, one patient had nonalcoholic fatty liver disease, and one patient had chronic hepatitis B & D. In the non-fatigued group, five patients had chronic hepatitis C and one had chronic hepatitis B. There were no significant differences between fatigued and non-fatigued patients beyond their fatigue scores. The PROMIS fatigue scores did not correlate with features of liver disease such as alanine aminotransferase (ALT), platelet count or presence of cirrhosis.
Table 2.
Patient characteristics.
| Fatigued (n=6) | Non-fatigued (n=6) | Test statistic | P-value | Effect size | |
|---|---|---|---|---|---|
| Age (27-68) | 45 years | 56 years | −1.474a | 0.17 | 0.85c |
| Male | 3 (25%) | 4 (33%) | 1 | 0.17d | |
| HCV Etiology | 4 (67%) | 5 (83%) | 0.37 | 0.51d | |
| ALT (u/L) (±SD) | 83.5 ±43 | 97±50 | −1.209 a | 0.63 | 0.29 c |
| Platelets (×109/L) (±SD) | 144 ±48 | 153 ±89 | −0.209 a | 0.84 | 0.12 c |
| Cirrhosis (Child A) | 3 (50%) | 2 (33%) | 1 | 0.18c | |
| Caffeine intake (mg/d) (±SD) | 238±279 | 122±195 | 9 b | 0.55 | 0.28e |
| Median PROMIS Score (range) | 21 (17-26) | 10.5 (9-13) | 36 b | <0.005 | 1e |
Student’s t
Mann-Whitney U
Cohen’s d
Cramer’s v
Rank-biserial correlation coefficient r
Sleep Studies
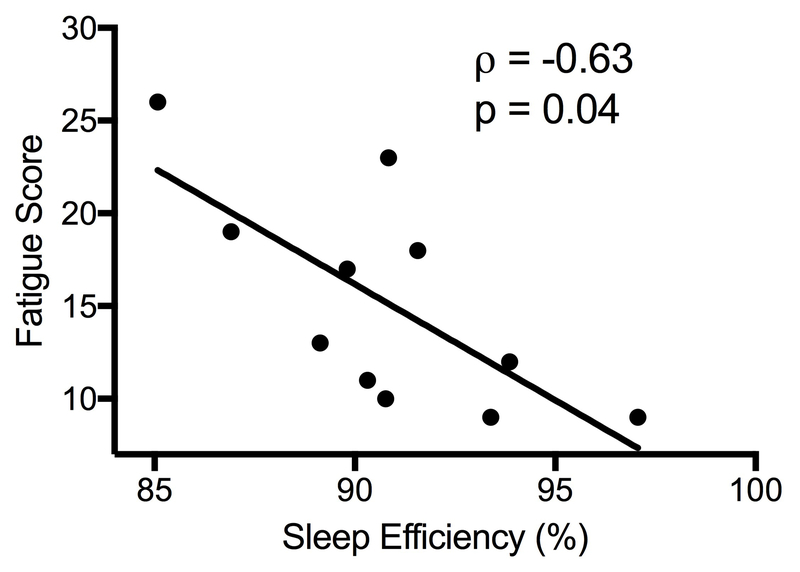
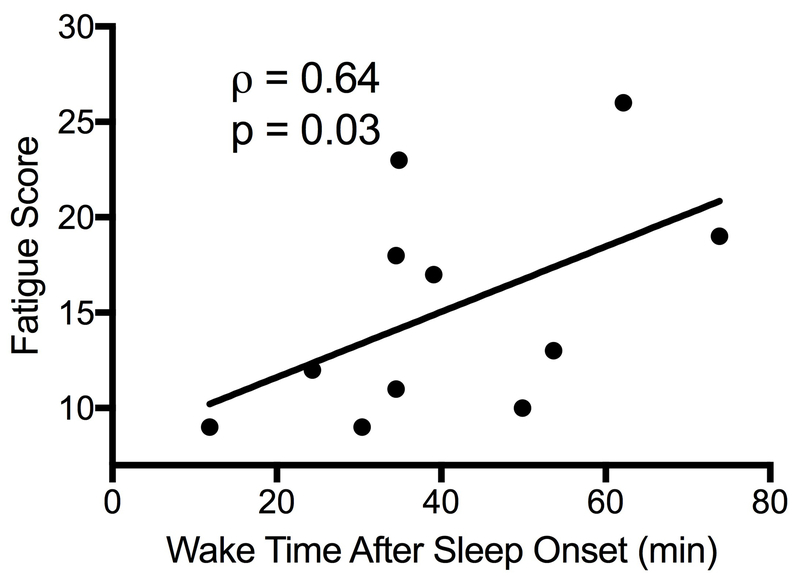
Sleep parameters averaged over 7 days in the home setting are shown in Table 3. While statistical significance was not met, actigraphy revealed that the fatigued patients were on average less physically active during the day, slept less, and awoke more at night. There was no significant difference between the bed times and rise times of the fatigued and non-fatigued patients. However, fatigued patients tended to go to bed later and rose later than non-fatigued patients. Despite spending the same amount of time in bed (nearly 7.5 hours), patients with fatigue demonstrated a trend for decreased sleep efficiency (proportion of time in bed actually spent sleeping) compared to those without fatigue (p=0.07). The PROMIS Fatigue score was strongly correlated with sleep efficiency (Spearman’s rho=−0.63, p=0.04) and wake time after sleep onset (rho=0.64, p=0.03) (Figure 1a-b).
Table 3.
Sleep parameters at home, measured by actigraphy and averaged over 7 days. Values presented as median (range).
| Fatigued (n=6) | Non-fatigued (n=6) | Test Statistic | P-value | Effect Size | |
|---|---|---|---|---|---|
| Sleep Efficiency (%) | 90% (85-92%) | 92% (89-97%) | −2.08a | 0.07 | 1.26 b |
| Daily Activity Level (counts) | 2.7×106 (1.9-3.9) | 2.8×106 (1.3-3.4) | 0.505 a | 0.63 | 0.30 b |
| Total Time in Bed (min) | 417 (410-553) | 425 (386-555) | 0.003 a | 0.99 | 0.00 b |
| Total Sleep Time (min) | 375 (355-479) | 403 (359-505) | −0.484 a | 0.64 | 0.29 b |
| Wake Time After Sleep Onset (min) | 39 (35-74) | 33 (12-54) | 1.455 a | 0.19 | 0.87 b |
| Number of Awakenings | 13.8 (11.3-23.7) | 9.7 (6.3-16.4) | 1.756 a | 0.13 | 1.05 b |
| Average Awakening Length (min) | 3.5 (3.0-3.6) | 3.6 (3.1-3.9) | −0.847 a | 0.41 | 0.52 b |
| Bed Time (HH:MM±SD) | 23:54±68 | 23:20±61 | 0.41 | ||
| Rise Time (HH:MM±SD) | 07:16±108 | 06:45±49 | 0.58 |
Student’s t
Cohen’s d
Figure 1.
Participants’ level of fatigue is associated with sleep parameters measured by actigraphy.
In contrast to the actigraphy data, sleep efficiency measured by polysomnography during the inpatient stay did not differ between fatigued and non-fatigued patients and did not correlate with the PROMIS fatigue score (Spearman’s rho=0.33, p=0.30). Total sleep time and apnea-hypopnea index were also not correlated with fatigue scores. Abnormalities in the polysomnogram were found in 9 (75%) of the 12 subjects, 6/6 (100%) of fatigued patients and 3/6 (50%) of non-fatigued (p=0.18). Of the fatigued patients, two had primary snoring, one had mild periodic leg movements (PLM), one had mild obstructive sleep apnea (OSA), one had moderate OSA, and one had mild OSA and moderate-severe PLM. In the non-fatigued group, one patient had severe complex sleep apnea and severe PLM, one had mild OSA and frequent PLM, and one had moderate OSA.
Six-Minute Walk Test
Physical stamina and cardiovascular fitness were assessed using a six-minute walk test, which was performed successfully in 11 of the 12 patients. There was no difference in mean distance walked between fatigued and non-fatigued patients (493 vs. 432 meters, p=0.48) and the distance did not correlate with PROMIS fatigue score (Spearman’s rho=0.44, p=0.17).
Circadian Rhythms
We assessed the central clock by measuring the core temperature. Central clock parameters including nadir night-time temperature, time to reach nadir, maximal 24-hour temperature and peak-to-nadir temperature difference were not different between patients with and without fatigue (Table 4).
Table 4.
Circadian rhythm parameters in fatigued and non-fatigued patients.
| Fatigued (n=6) | Non-Fatigued (n=6) | Test Statistic | p-value | Effect Size | |
|---|---|---|---|---|---|
| Core temperature night-time nadir (°C) | 36.43±0.14 | 36.36±0.37 | 0.389a | 0.69 | 0.25b |
| Core temperature nadir time | 04:05 (00:57-07:46) | 05:44 (02:34-07:31) | 0.25 | ||
| Peak Melatonin (pg/ml) | 90.3±70.1 | 71.2±45.6 | 0.558a | 0.59 | 0.32a |
| Melatonin peak time | 3am | 1am | 0.048 |
Student’s t
Cohen’s d
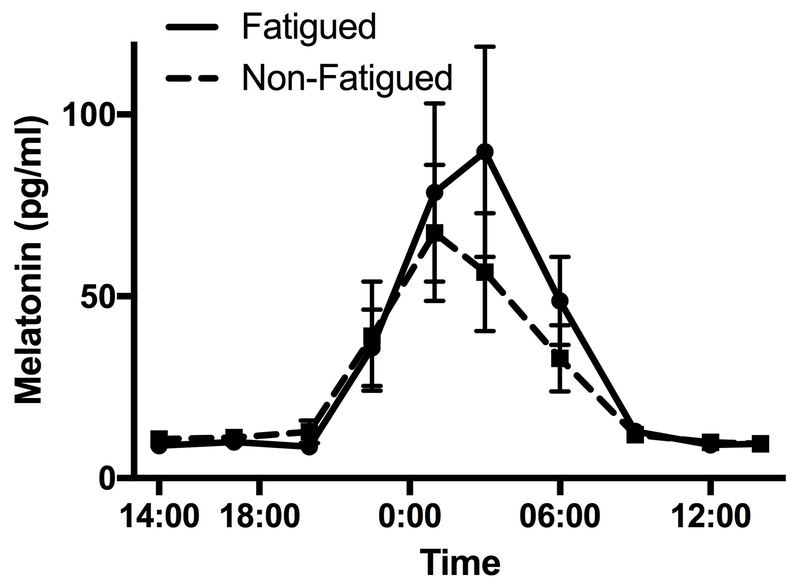
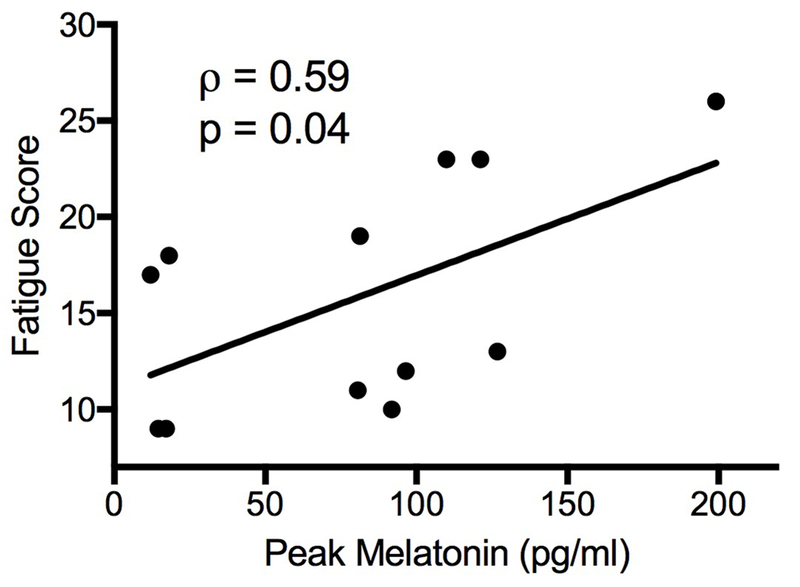
Melatonin levels measured over the 24-hour period, demonstrated the expected oscillating pattern (Figure 2a). Fatigue score was strongly correlated with peak melatonin level, with more fatigued patients having higher peak melatonin levels (rho=0.59, p=0.04, Figure 2b).
Figure 2.
Melatonin profiles differ by degree of fatigue. Panel (a) shows 24 h profile by group. Panel (b) shows association between individual participants’ PROMIS Fatigue SF7a score and peak melatonin level.
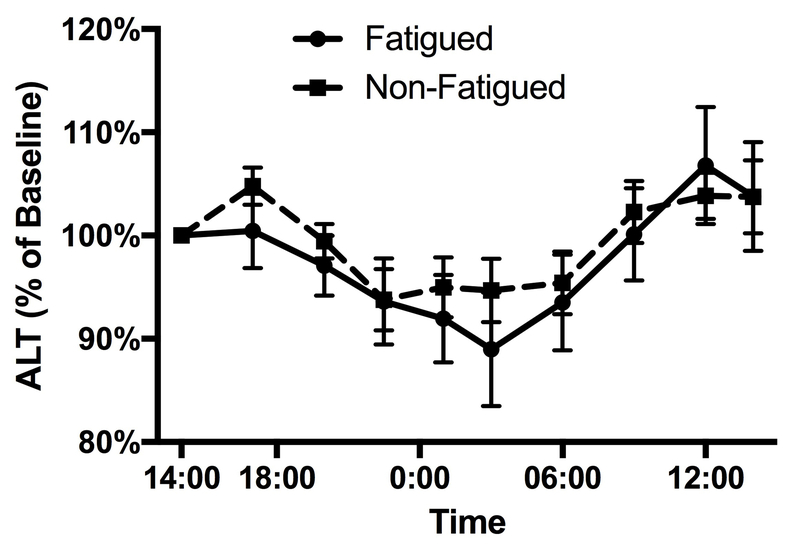
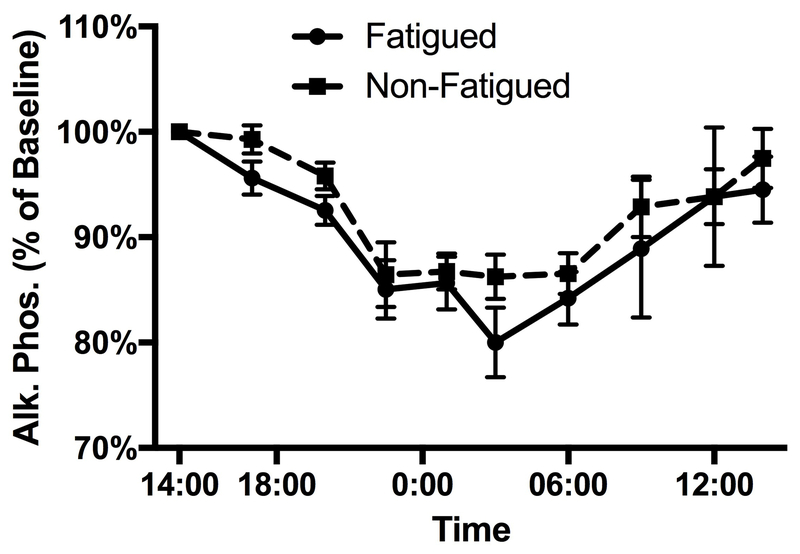
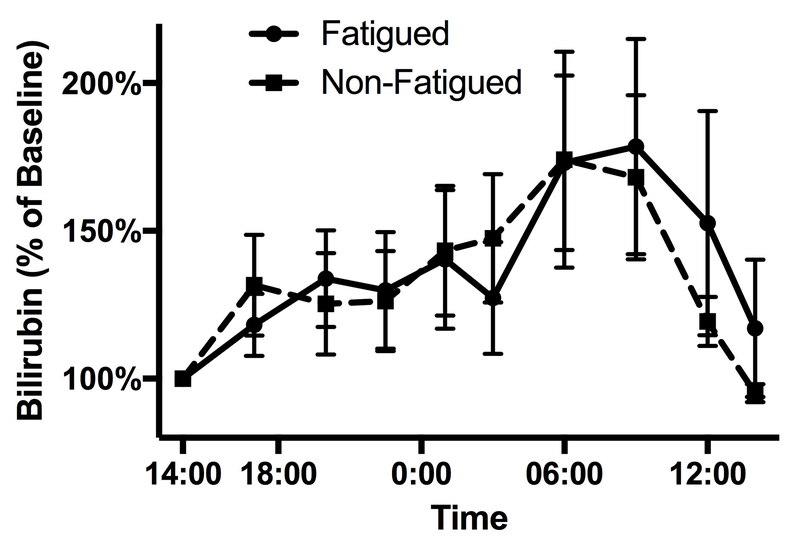
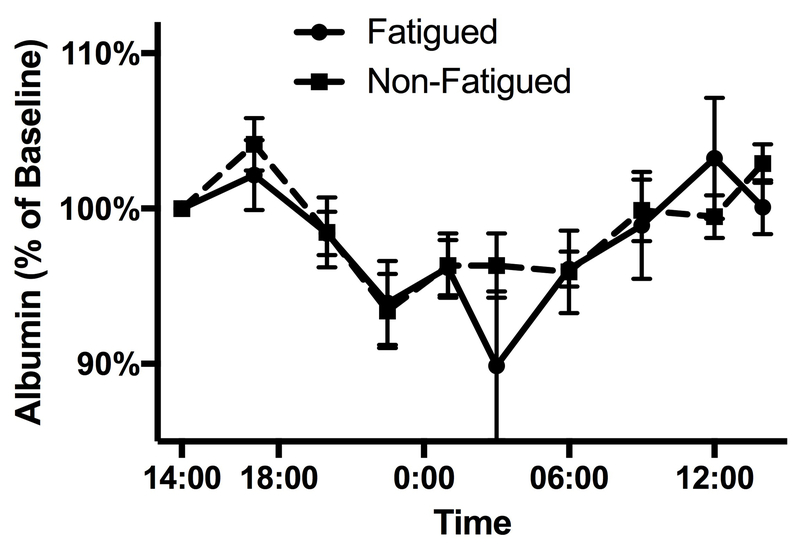
In order to assess liver-specific circadian rhythms we measured the serum levels of several liver-derived factors over the 24-hour period (Figure 3). Liver enzymes demonstrated a diurnal fluctuation, peaking in the afternoon (alkaline phosphatase) or early evening (ALT) and reaching a nadir at night, while bilirubin peaked in the early morning. Although ALT (Figure 3a), aspartate aminotransferase (AST), alkaline phosphatase (Figure 3b), total bilirubin (Figure 3c) and albumin (Figure 3d) displayed significant (p<0.05) circadian rhythmicity, their amplitude and acrophase did not differ between groups and were not correlated with fatigue score.
Figure 3.
Diurnal variation of liver-derived factors does not differ significantly between fatigued and nonfatigued participants. Panel (a) displays ALT, (b) alkaline phosphatase, (c) total bilirubin, and (d) albumin.
Beyond the typical liver function tests, we also assessed circadian patterns in hepatic protein synthesis and secretion using serum levels of the short half-life liver-derived factor VII and a1AT; these proteins did not vary with time in a statistically significant manner.
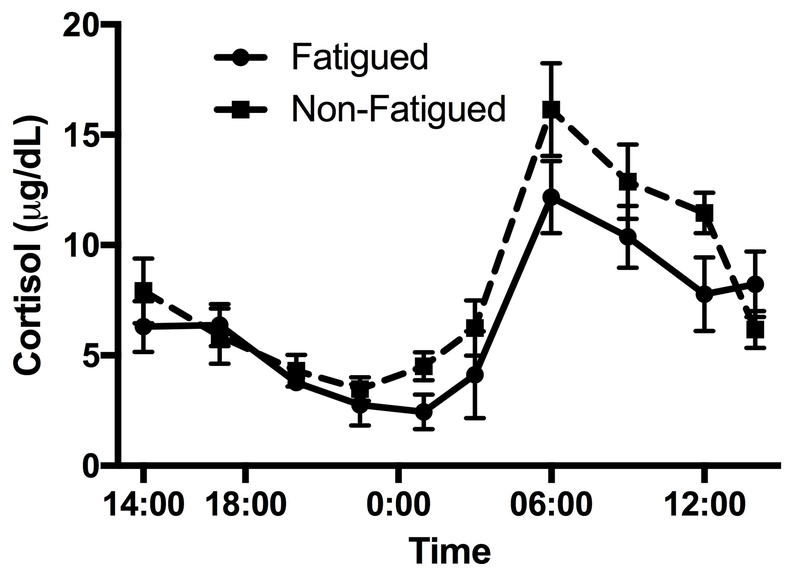
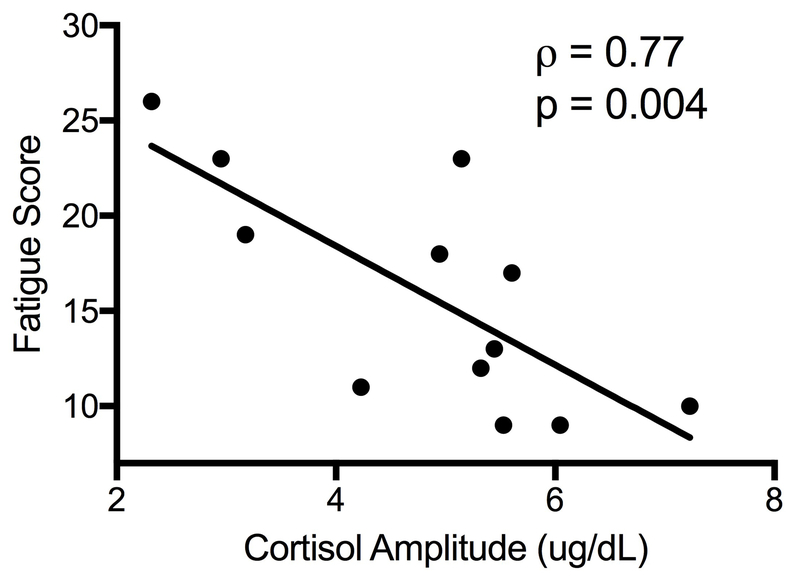
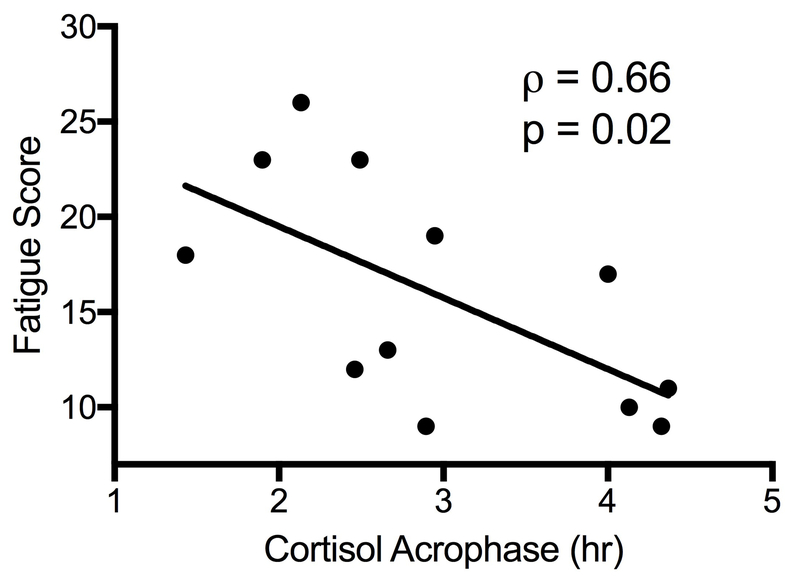
For comparison with the liver clock, we measured serum cortisol and cortisol releasing factor (CRF) as markers of the adrenal clock and the hypothalamic-pituitary-adrenal (HPA) axis, respectively, and the liver-secreted cortisol binding globulin (CBG). As expected, serum cortisol levels displayed significant periodicity (p=0.0002, Figure 4a). Circadian parameters of cortisol fluctuation were associated with fatigue; fatigued patients had a lower amplitude of fluctuation of cortisol levels (4.0±1.4 vs. 5.6±1 μg/dL, p=0.04) and a trend towards a phase shift causing a 1 hour delay (p=0.086). Fatigue scores showed a strong negative correlation with cortisol amplitude (rho=−0.77, p=0.004, Fig. 4B) and acrophase (r=−0.66, p=0.02, Fig. 4C). Fatigued subjects showed a trend for lower 6AM cortisol (12.2 vs. 16.1 μg/dL), average 24-hour cortisol (6.6 vs. 8.0 μg/dL) and total 24-hour cortisol output (156.0 vs. 191.3 μg/dL) but the differences did not reach statistical significance (p=0.13 for all). Cortisol amplitude and acrophase were not correlated with peak melatonin levels and their association with fatigue severity was independent of melatonin on multivariate regression. Circadian features of CRF and CBG did not show a significant relationship with fatigue score. IL-6 was suggested as a mediator of somnolence and fatigue in adrenal dysfunction; its levels and circadian features were not associated with fatigue or with cortisol fluctuations in our subjects.
Figure 4.
Circadian rhythm of cortisol varies by fatigue level. Panel (a) shows the expected periodicity. Panel (b) shows the relationship between PROMIS Fatigue SF7a score and cortisol amplitude. Panel (c) shows the relationship between the PROMIS score and cortisol acrophase.
Insulin and free fatty acids showed a consistent temporal pattern across all subjects, driven by meal timing and validating the standardization of experimental conditions during the 24-hour assessment, but were not associated with fatigue score. Similarly, components of the lipid profile (triglycerides, total cholesterol and HDL-cholesterol) displayed circadian rhythmicity but were not significantly associated with fatigue score.
Relationships between Peak Melatonin Level, Sleep Efficiency, and Cortisol Amplitude
We found associations of fatigue with sleep efficiency, melatonin level, and cortisol amplitude. To determine if these variables were related to each other, we performed bivariate correlation analyses and found no significant associations between the 3 variables (Peak Melatonin vs. Sleep Efficiency, ρ=−0.42, 0.20; Peak Melatonin vs. Cortisol Amplitude, ρ=−0.52, 0.085; Cortisol Amplitude vs. Sleep Efficiency, ρ=0.51, 0.11).
Discussion
This prospective trial determined that fatigue in CLD patients was associated with impaired sleep efficiency and altered melatonin and cortisol profiles. We found that fatigued patients had reduced free-living sleep quality measured using actigraphy (sleep efficiency and wake time after sleep onset) compared with the non-fatigued group and these sleep quality indices were strongly correlated with fatigue severity. These differences were not confirmed using one-night polysomnography, perhaps due to patient unfamiliarity and discomfort in the lab-based sleep conditions in both participant groups. Interestingly, however, polysomnography detected abnormalities in 9 of 12 subjects, though no significant difference was noted between fatigued and non-fatigued patients. While prior work has documented circadian abnormalities and sleep disturbance in patients with cirrhosis, to our knowledge, our study represents the first evidence of these abnormalities in patients with CLD across the spectrum of fibrosis[29].
We found several aberrations of circadian rhythm that were associated with fatigue in our participants. The peak melatonin levels correlated with severity of fatigue, and the peak occurred later during the night in fatigued subjects. This is similar to the derangements seen in delayed sleep phase syndrome (DSPS) and is consistent with previously published data[30, 31]. However, in contrast to DSPS, we have not found a delay in the night-time core temperature nadir[32]. Melatonin secretion is regulated by the central pacemaker in the SCN and is modulated by ambient light[33]. In our study, ambient light was turned on and off at a uniform time for all subjects to ensure standardization of experimental conditions. Thus, our findings suggest that central clock disturbances are present even in patients with non-cirrhotic CLD and may be impacting their fatigue levels. As the liver is the main site of melatonin catabolism, it is also possible that melatonin breakdown at the liver, rather than melatonin secretion from the pineal gland, is the cause for the difference in diurnal patterns; however, as there was no difference in severity of liver disease between the two groups, this is unlikely to be the cause[34]. Although melatonin peak was not significantly correlated with sleep efficiency, it is possible that the lack of correlation is due to the small number of subjects.
Fatigue and Circadian Rhythms
Our initial hypothesis was that fatigue in CLD is related to aberrations in hepatic circadian rhythms or lack of synchronization between hepatic and central circadian rhythms. The liver clock is entrained to the central clock by neuronal, hormonal and body temperature signals, and is highly dependent on feeding cycles[35]. In animal models the intra-hepatic rhythm is typically assessed at the transcriptional level but this is not feasible in human studies. We attempted to assess the hepatic clock in our subjects by measuring serum markers that are predominantly secreted from the liver, including typical liver function tests, and proteins with a short half-life like factor VII and alpha-1 antitrypsin. Although we were able to demonstrate a diurnal pattern of transaminases, alkaline phosphatase and bilirubin, as previously described, we did not find an association of any of these rhythms with the presence or severity of fatigue[36]. It is possible that the serum markers, which reflect hepatic secretion but also non-hepatic clearance, are not a reliable measure of the intra-hepatic clock or alternatively, that hepatic rhythms are not underlying the fatigue of CLD.
In contrast to the liver clock, we found a close association of fatigue with the adrenal clock in our patients. Fatigued subjects had a lower amplitude of cortisol fluctuation and a delay in its peak, both of which correlated with fatigue severity. These features required assessment of circadian rhythmicity and were not easily discernibly by typically measured values, such as morning cortisol levels or total cortisol output. We did not measure levels of ACTH due to concern about its stability in stored serum. We did, however, assay CRF and did not detect a similar change, suggesting the changes in cortisol are not centrally mediated. This is further supported by the absence of association between the changes in cortisol and melatonin patterns in fatigued subjects.
The absence of diurnal variations in serum CBG levels suggest that it was not involved in the shift of cortisol diurnal rhythm[37]. The adrenal gland exhibits an autonomous clock that is at least partially independent of the SCN central clock, and in fact, glucocorticoids themselves may serve to synchronize the liver clock[38, 39]. Our findings suggest that in CLD, an aberrant adrenal clock may underlie the associated fatigue.
Cirrhosis has been associated with impaired function of the hypothalamic-pituitary adrenal (HPA) axis[40, 41, 42]. IL-6 has been suggested as an important mediator linking adrenal corticosteroid secretion to sleepiness and fatigue and elevated IL-6 was linked to fatigue in cirrhotics[42, 43]. We measured IL-6 levels in our subjects and, although the typical circadian pattern was seen, no association with fatigue was noted. Fatigue is difficult to define and quantify, no “objective” test exists, and it has diverse manifestations. Patients with fatigue can be limited physically and mentally. While our study included a six-minute walk test, we did not conduct neuropsychiatric testing.
Fatigue Assessment
To quantify fatigue, our main instrument has been the PROMIS SF7a questionnaire. PROMIS is a set of reliable and precise tools to measure patient-reported health status, and has been shown to be an adequate tool for assessment of fatigue in patients with chronic liver disease as well as other medical conditions[44]. We previously observed excellent concordance between self-reported fatigue (present or absent), fatigue scores using the Modified Fatigue Impact Scale (MFIS) and the PROMIS SF7 (Sarkar & Rotman, unpublished). Given the excellent validation of PROMIS across several patient categories, its standardization and the availability of population norms, we chose it as the tool for our study. While we used multiple questionnaires and tests to assess various aspects of fatigue, ultimately we dichotomized the patients into fatigued and non-fatigued groups based on the PROMIS score. This categorization of participants limits our ability to tease apart various manifestations of fatigue but was appropriate for analysis of the pilot data.
Limitations and Strengths
The main limitation of our study is its small sample size. This is evident for example in our inability to determine whether the three main findings that are associated with fatigue - sleep efficiency, melatonin and cortisol - are truly independent of each other. Our study was, by definition, a pilot study aiming to collect a large amount of data on each individual subject, and limited in size by practical considerations of effort and budget. Fatigue is a symptom that crosses the spectrum of liver disease etiology and severity; this suggests a common pathway that is not necessarily disease-specific[45]. In light of that fact and because this was an exploratory study, we enrolled patients with diverse forms of chronic liver disease. While we matched the fatigued and non-fatigued patients by fibrosis stage, we did not perform a subgroup analysis by etiology because of the small numbers. Our findings merit confirmation in studies with more subjects but a narrower scope, which can focus on specific endpoints.
The main strength of our study is its careful and standardized design. Circadian rhythms and biomarkers vary rapidly in response to external cues. By carefully maintaining uniform meal timing and content (as proven by the insulin and free fatty acid curves) and by standardizing ambient light times and sleep conditions, we were able to minimize environmental factors that could introduce error. Furthermore, by performing all of the analyses (i.e. blood tests, sleep study questionnaires and 6-minute walk test) within the same short admission, we could assess for correlations across those different modalities.
We intentionally chose to recruit subjects with a variety of liver disease, but the majority were infected with HCV, reflecting the typical population of subjects at our center at the time of recruitment. Our aim was to identify mechanisms for liver-associated fatigue that are independent of the etiology of CLD, as suggested by its presence across different disorders. It is possible that these mechanisms are modulated differently in different liver diseases, or supplemented by additional disease-specific mechanisms; larger studies would be needed to assess that question.
In conclusion, we identified an association of liver-related fatigue with impaired sleep efficiency and with subtle aberrations in melatonin and cortisol circadian rhythms. Mechanistic studies to understand how CLD affects adrenal and pineal function may help in further elucidating the pathogenesis of this common and debilitating syndrome. Our finding of delayed and blunted melatonin and cortisol rhythms suggest that therapeutic approaches that aim to reset or modulate the circadian rhythm may provide relief for fatigued patients with liver disease[46].
Acknowledgements
Andrea Beri, Wally Duncan, Jackie Greenfield, Jay Hoofnagle, Jake Liang, Vicky Liberty, Lynette Niemann, Dante Picchioni, Ann Smith, Sarah Smyth, Windy Wallin, Allison Weiss, Edythe Wiggs, NIDDK Liver Diseases Branch staff and patients.
The authors would like to acknowledge the UCSF Liver Center P30 DK026743 and Division of Gastroenterology, as well as the NIH intramural grant Z01 999999.
Conflicts of Interest and Source of Funding: No relevant disclosures declared. NIH intramural grant Z01 999999, UCSF Liver Center P30 DK026743.
References:
- 1.Foster GR, Goldin RD, Thomas HC. Chronic hepatitis C virus infection causes a significant reduction in quality of life in the absence of cirrhosis. Hepatology. 1998. January;27(1):209–12. 10.1002/hep.510270132. PubMed PMID: 9425939; eng. [DOI] [PubMed] [Google Scholar]
- 2.Gumber SC, Chopra S. Hepatitis C: a multifaceted disease. Review of extrahepatic manifestations. Ann Intern Med. 1995. October;123(8):615–20. PubMed PMID: 7677303; eng. [DOI] [PubMed] [Google Scholar]
- 3.Kallman J, O’Neil MM, Larive B, et al. Fatigue and health-related quality of life (HRQL) in chronic hepatitis C virus infection. Dig Dis Sci. 2007. October;52(10):2531–9. doi: 10.1007/s10620-006-9708-x. PubMed PMID: 17406828; eng. [DOI] [PubMed] [Google Scholar]
- 4.Abbas G, Jorgensen RA, Lindor KD. Fatigue in primary biliary cirrhosis. Nat Rev Gastroenterol Hepatol. 2010. June;7(6):313–9. 10.1038/nrgastro.2010.62. PubMed PMID: 20458334; eng. [DOI] [PubMed] [Google Scholar]
- 5.Gutteling JJ, de Man RA, Busschbach JJ, et al. Overview of research on health-related quality of life in patients with chronic liver disease. Neth J Med. 2007. 2007 Jul-Aug;65(7):227–34. PubMed PMID: 17656809; eng. [PubMed] [Google Scholar]
- 6.D’Mello C, Swain MG. Liver-brain inflammation axis. Am J Physiol Gastrointest Liver Physiol. 2011. November;301(5):G749–61. 10.1152/ajpgi.00184.2011. PubMed PMID: 21868631; eng. [DOI] [PubMed] [Google Scholar]
- 7.Córdoba J, Cabrera J, Lataif L, et al. High prevalence of sleep disturbance in cirrhosis. Hepatology. 1998. February;27(2):339–45. 10.1002/hep.510270204. PubMed PMID: 9462628; eng. [DOI] [PubMed] [Google Scholar]
- 8.Kushida CA. Sleep deprivation : basic science, physiology, and behavior. New York: Marcel Dekker; 2005. (Lung biology in health and disease; 192). [Google Scholar]
- 9.Hoogerwerf WA. Role of biological rhythms in gastrointestinal health and disease. Rev Endocr Metab Disord. 2009. Dec;10(4):293–300. doi: 10.1007/s11154-009-9119-3. PubMed PMID: 19798581; eng. [DOI] [PubMed] [Google Scholar]
- 10.Panda S, Antoch MP, Miller BH, et al. Coordinated transcription of key pathways in the mouse by the circadian clock. Cell. 2002. May;109(3):307–20. doi: S0092867402007225 [pii]. PubMed PMID: 12015981; eng. [DOI] [PubMed] [Google Scholar]
- 11.Storch KF, Lipan O, Leykin I, et al. Extensive and divergent circadian gene expression in liver and heart. Nature. 2002. May;417(6884):78–83. 10.1038/nature744. PubMed PMID: 11967526; eng. [DOI] [PubMed] [Google Scholar]
- 12.Chen P, Kakan X, Zhang J. Altered circadian rhythm of the clock genes in fibrotic livers induced by carbon tetrachloride. FEBS Lett. 2010. April;584(8):1597–601. 10.1016/j.febslet.2010.03.019. PubMed PMID: 20233594; eng. [DOI] [PubMed] [Google Scholar]
- 13.Mohawk JA, Green CB, Takahashi JS. Central and peripheral circadian clocks in mammals. Annu Rev Neurosci. 2012. July;35:445–62. doi: 10.1146/annurev-neuro-060909-153128. PubMed PMID: 22483041; eng. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chen CQ, Fichna J, Bashashati M, et al. Distribution, function and physiological role of melatonin in the lower gut. World J Gastroenterol. 2011. September;17(34):3888–98. doi: 10.3748/wjg.v17.i34.3888. PubMed PMID: 22025877; PubMed Central PMCID: PMCPMC3198018. eng. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Srinivasan V, Spence DW, Trakht I, et al. Immunomodulation by melatonin: its significance for seasonally occurring diseases. Neuroimmunomodulation. 2008;15(2):93–101. 10.1159/000148191. PubMed PMID: 18679047; eng. [DOI] [PubMed] [Google Scholar]
- 16.Pevet P, Challet E. Melatonin: both master clock output and internal time-giver in the circadian clocks network [; Research Support, Non-U.S. Gov’t; Review]. Journal of physiology, Paris. 2011. 2011 Dec (Epub 2011 Jul;105(4–6):170–82. PubMed PMID: MEDLINE:21914478; English. [DOI] [PubMed] [Google Scholar]
- 17.Steindl PE, Finn B, Bendok B, et al. Disruption of the diurnal rhythm of plasma melatonin in cirrhosis. Ann Intern Med. 1995. August;123(4):274–7. PubMed PMID: 7611593; eng. [DOI] [PubMed] [Google Scholar]
- 18.Celinski K, Konturek PC, Slomka M, et al. Altered basal and postprandial plasma melatonin, gastrin, ghrelin, leptin and insulin in patients with liver cirrhosis and portal hypertension without and with oral administration of melatonin or tryptophan. J Pineal Res. 2009. May;46(4):408–14. 10.1111/j.1600-079X.2009.00677.x. PubMed PMID: 19552764; eng. [DOI] [PubMed] [Google Scholar]
- 19.Steindl PE, Ferenci P, Marktl W. Impaired hepatic catabolism of melatonin in cirrhosis. Ann Intern Med. 1997. September;127(6):494 PubMed PMID: 9313015; eng. [DOI] [PubMed] [Google Scholar]
- 20.Cordoba J, Steindl P, Blei AT. Melatonin arrhythmia is corrected after liver transplantation. Am J Gastroenterol. 2009. July;104(7):1862–3. 10.1038/ajg.2009.171. PubMed PMID: 19455119; eng. [DOI] [PubMed] [Google Scholar]
- 21.SHERLOCK S, SUMMERSKILL WH, WHITE LP, et al. Portal-systemic encephalopathy; neurological complications of liver disease. Lancet. 1954. September;267(6836):454–7. PubMed PMID: 13193045; eng. [PubMed] [Google Scholar]
- 22.Junghaenel DU, Christodoulou C, Lai JS, et al. Demographic correlates of fatigue in the US general population: results from the patient-reported outcomes measurement information system (PROMIS) initiative. J Psychosom Res. 2011. September;71(3):117–23. 10.1016/j.jpsychores.2011.04.007. PubMed PMID: 21843744; eng. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bajaj JS, Thacker LR, Wade JB, et al. PROMIS computerised adaptive tests are dynamic instruments to measure health-related quality of life in patients with cirrhosis. Aliment Pharmacol Ther. 2011. November;34(9):1123–32. doi: 10.1111/j.1365-2036.2011.04842.x. PubMed PMID: 21929591; PubMed Central PMCID: PMCPMC3989141. eng. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.de Souza L, Benedito-Silva AA, Pires ML, et al. Further validation of actigraphy for sleep studies. Sleep. 2003. Feb;26(1):81–5. PubMed PMID: 12627737; eng. [DOI] [PubMed] [Google Scholar]
- 25.Kushida CA, Chang A, Gadkary C, et al. Comparison of actigraphic, polysomnographic, and subjective assessment of sleep parameters in sleep-disordered patients. Sleep Med. 2001. September;2(5):389–96. doi: S1389-9457(00)00098-8 [pii] PubMed PMID: 14592388; eng. [DOI] [PubMed] [Google Scholar]
- 26.Modi AA, Feld JJ, Park Y, et al. Increased caffeine consumption is associated with reduced hepatic fibrosis. Hepatology. 2010. January;51(1):201–9. doi: 10.1002/hep.23279. PubMed PMID: 20034049; PubMed Central PMCID: PMCPMC2801884. eng. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Cornelissen G Cosinor-based rhythmometry. Theor Biol Med Model. 2014. April;11:16. doi: 10.1186/1742-4682-11-16. PubMed PMID: 24725531; PubMed Central PMCID: PMCPMC3991883. eng. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Bacchetti P, Deeks SG, McCune JM. Breaking free of sample size dogma to perform innovative translational research. Sci Transl Med. 2011. June 15;3(87):87ps24. doi: 10.1126/scitranslmed.3001628. PubMed PMID: 21677197; PubMed Central PMCID: PMCPMC3134305. eng. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Swain MG. Fatigue in liver disease: pathophysiology and clinical management. Can J Gastroenterol. 2006. March;20(3):181–8. PubMed PMID: 16550262; PubMed Central PMCID: PMCPMC2582971. eng. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Agnew HW, Webb WB, Williams RL. The first night effect: an EEG study of sleep. Psychophysiology. 1966. January;2(3):263–6. PubMed PMID: 5903579; eng. [DOI] [PubMed] [Google Scholar]
- 31.Montagnese S, Middleton B, Mani AR, et al. Sleep and circadian abnormalities in patients with cirrhosis: features of delayed sleep phase syndrome? Metab Brain Dis. 2009. September;24(3):427–39. doi: 10.1007/s11011-009-9146-5. PubMed PMID: 19756996; eng. [DOI] [PubMed] [Google Scholar]
- 32.Montagnese S, Middleton B, Mani AR, et al. On the origin and the consequences of circadian abnormalities in patients with cirrhosis. Am J Gastroenterol. 2010. August;105(8):1773–81. 10.1038/ajg.2010.86. PubMed PMID: 20332771; eng. [DOI] [PubMed] [Google Scholar]
- 33.Ozaki S, Uchiyama M, Shirakawa S, et al. Prolonged interval from body temperature nadir to sleep offset in patients with delayed sleep phase syndrome. Sleep. 1996. January;19(1):36–40. PubMed PMID: 8650460; eng. [PubMed] [Google Scholar]
- 34.Johnston JD, Skene DJ. 60 YEARS OF NEUROENDOCRINOLOGY: Regulation of mammalian neuroendocrine physiology and rhythms by melatonin. J Endocrinol. 2015. August;226(2):T187–98. doi: 10.1530/JOE-15-0119. PubMed PMID: 26101375; eng. [DOI] [PubMed] [Google Scholar]
- 35.Skene DJ, Papagiannidou E, Hashemi E, et al. Contribution of CYP1A2 in the hepatic metabolism of melatonin: studies with isolated microsomal preparations and liver slices. J Pineal Res. 2001. November;31(4):333–42. PubMed PMID: 11703563; eng. [DOI] [PubMed] [Google Scholar]
- 36.Reinke H, Asher G. Circadian Clock Control of Liver Metabolic Functions. Gastroenterology. 2015. December 1. doi: 10.1053/j.gastro.2015.11.043. PubMed PMID: 26657326; Eng. [DOI] [PubMed] [Google Scholar]
- 37.Sennels HP, Jorgensen HL, Goetze JP, et al. Rhythmic 24-hour variations of frequently used clinical biochemical parameters in healthy young males--the Bispebjerg study of diurnal variations. Scand J Clin Lab Invest. 2012. July;72(4):287–95. doi: 10.3109/00365513.2012.662281. PubMed PMID: 22397709; eng. [DOI] [PubMed] [Google Scholar]
- 38.Chung TT, Gunganah K, Monson JP, et al. Circadian variation in serum cortisol during hydrocortisone replacement is not attributable to changes in cortisol-binding globulin concentrations. Clin Endocrinol (Oxf). 2016. April;84(4):496–500. doi: 10.1111/cen.12982. PubMed PMID: 26603673; eng. [DOI] [PubMed] [Google Scholar]
- 39.Gamble KL, Berry R, Frank SJ, et al. Circadian clock control of endocrine factors. Nat Rev Endocrinol. 2014. August;10(8):466–75. doi: 10.1038/nrendo.2014.78. PubMed PMID: 24863387; PubMed Central PMCID: PMCPMC4304769. eng. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Dickmeis T Glucocorticoids and the circadian clock. J Endocrinol. 2009. January;200(1):3–22. doi: 10.1677/JOE-08-0415. PubMed PMID: 18971218; eng. [DOI] [PubMed] [Google Scholar]
- 41.Galbois A, Rudler M, Massard J, et al. Assessment of adrenal function in cirrhotic patients: salivary cortisol should be preferred. J Hepatol. 2010. June;52(6):839–45. doi: 10.1016/j.jhep.2010.01.026. PubMed PMID: 20385427; eng. [DOI] [PubMed] [Google Scholar]
- 42.Kalaitzakis E, Josefsson A, Castedal M, et al. Factors related to fatigue in patients with cirrhosis before and after liver transplantation. Clin Gastroenterol Hepatol. 2012. February;10(2):174–81, 181.e1 10.1016/j.cgh.2011.07.029. PubMed PMID: 21839709; eng. [DOI] [PubMed] [Google Scholar]
- 43.Tan T, Chang L, Woodward A, et al. Characterising adrenal function using directly measured plasma free cortisol in stable severe liver disease. J Hepatol. 2010. November;53(5):841–8. doi: 10.1016/j.jhep.2010.05.020. PubMed PMID: 20739086; eng. [DOI] [PubMed] [Google Scholar]
- 44.Cella D, Lai JS, Jensen SE, et al. PROMIS Fatigue Item Bank had Clinical Validity across Diverse Chronic Conditions. J Clin Epidemiol. 2016. May;73:128–34. doi: 10.1016/j.jclinepi.2015.08.037. PubMed PMID: 26939927; PubMed Central PMCID: PMCPMC4902759. eng. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.An K, Jallo N, Menzies V, et al. Integrative Review of Co-Occurring Symptoms Across Etiologies of Chronic Liver Disease and Implications for Symptom Management Research and Practice. J Nurs Scholarsh. 2015. July;47(4):310–7. doi: 10.1111/jnu.12146. PubMed PMID: 26077958; eng. [DOI] [PubMed] [Google Scholar]
- 46.Ballesta A, Innominato PF, Dallmann R, et al. Systems Chronotherapeutics. Pharmacol Rev 2017. April;69(2):161–199. doi: 10.1124/pr.116.013441. PubMed PMID: 28351863; eng. [DOI] [PMC free article] [PubMed] [Google Scholar]