Abstract
We present an unusual case of Eagle’s syndrome with bilateral internal carotid artery dissection in a 45-year-old man. Initial symptomatology included ipsilateral headaches and facial sensory symptoms. A right horner’s syndrome was present on clinical examination. Radiological imaging revealed an old infarct, with bilateral carotid dissections and bilateral elongated styloid processes consistent with Eagle’s syndrome. Despite initiation of secondary prevention with antiplatelet therapy, he had two further ischaemic events. The case highlights the symptomatology and complications of Eagle’s syndrome, with its management discussed through a review of similar case reports.
Keywords: neurology, stroke, neuroimaging
Background
The incidence of internal carotid artery dissection (ICAD) is underestimated due to underdiagnosis and unawareness, with an annual reported incidence of 2.6 per 1 000 000. However, ICAD contributes to 20% of ischaemic strokes in the younger generation.1
ICAD can be a complication of ‘stylocarotid’ variant of Eagle’s syndrome (ES). This occurs due to impingement of the carotid artery from an elongated styloid process (ESP) resulting in dissection and cerebrovascular complications.2
Due to its symptomatic variation and unspecific features, ES can be easily missed. This case highlights the importance of angiography in such circumstances, especially when tackling a differential diagnosis of cerebral ischaemia and cervicofacial pain.3 4
Surgical treatment remains the most definitive treatment for prevention of further ischaemic episodes in ICAD secondary to ES as highlighted in the literature review.
In view of the clinical data available, an aggressive surgical approach to avert further neurological sequelae in such patients is advisable.
Case presentation
A 45-year-old man presented to accident and emergency with right-sided headache and facial numbness that developed over the previous 2 days. He also had a ‘heavy tongue’, with decreased movement to the right side. There was no recent history of abrupt head movements but he had been involved in a traumatic motor vehicle accident 15 years prior to presentation.
His medical history was unremarkable and he was on no regular medications. He professed a healthy active life style, was a non-smoker and teetotal and lived with his partner.
Clinical evaluation revealed a right Horner’s syndrome with the rest of the neurological examination being unremarkable. He was significantly hypertensive with a blood pressure of 201/121 mm Hg. Other parameters were stable.
Blood investigations yielded no significant abnormalities. An ECG showed diffuse T wave inversions and CT of the brain displayed an old left frontal lobe infarct (figure 1). No previous imaging was available for comparison.
Figure 1.
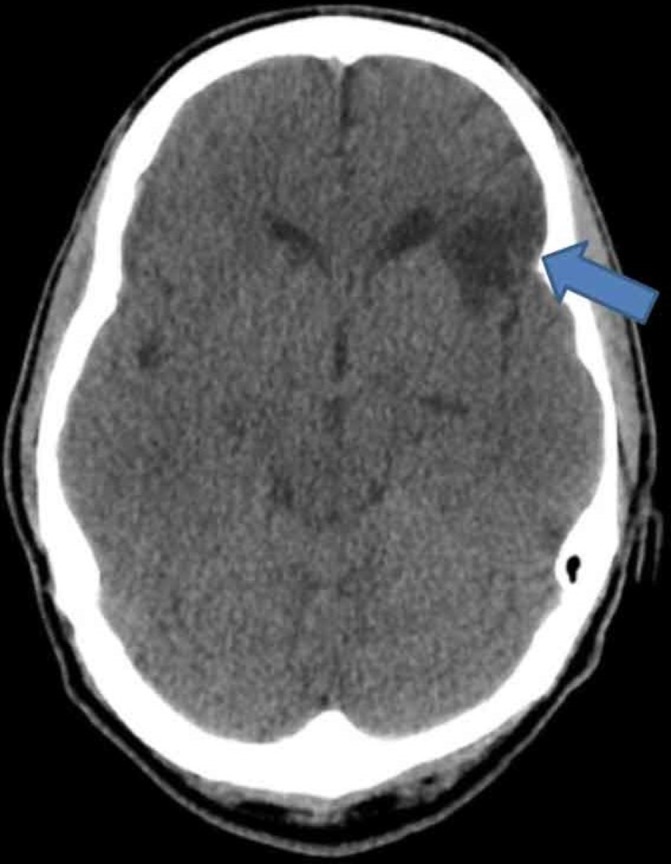
CT of the brain displaying an old left frontal lobe infarct, as indicated by the blue arrow.
Further CT imaging of the neck revealed bilateral dissection of the internal carotid arteries (figures 2 and 3). CT angiography of the aortic arch displayed pathologically ESPs, with the right and left processes being 4.0 and 4.2 cm in length, respectively (figure 4).
Figure 2.
CT angiography of the aortic arch and neck revealing bilateral internal carotid dissection - shown by the two white arrows.
Figure 3.

CT angiography of the aortic arch and neck revealing right internal carotid artery dissection at the level of the blue arrow.
Figure 4.
CT angiography of the aortic arch displaying elongated styloid processes, as annotated by the two white arrows.
Aspirin 75 mg, clopidogrel 75 mg, amlodipine 5 mg and simvastatin 20 mg were prescribed as part of the initial management. Two days after hospitalisation, he experienced photophobia, tenderness over the right neck muscles and trapezius as well as right ocular pain and headaches.
MRI of the head, neck and MR angiography showed a subacute embolic infarction in the distribution of the right anterior circulation (figure 5). Furthermore, occlusion of the right dissected internal carotid artery was confirmed.
Figure 5.
Diffusion-weighted MRI indicating acute infarction of the right caudate head in the territory of the anterior cerebral artery (white arrow).
During his 5th day of hospitalisation, he suffered a mechanical fall with acute confusion. Examination revealed left homonymous hemianopia with visual neglect, dense left hemiplegia with hemianaesthesia, dysarthria and an up-going left plantar response.
A repeat MRI showed an acute right middle cerebral artery territory infarct involving the inferior division of the M2 segment (figure 6).
Figure 6.
Diffusion-weighted MRI revealing right middle cerebral artery territory infarct.
Hypertension was ultimately controlled with three different antihypertensive medications (amlodipine 10 mg, perindopril 8 mg and atenolol 25 mg daily). Blood and urine investigations for secondary causes of hypertension were negative and an ultrasound of the renal arteries revealed no abnormalities.
An echocardiogram was also carried out showing apical hypertrophic cardiomyopathy (AHCM) with no embolic source. The patient was referred for follow-up at the cardiomyopathy clinic.
The patient was referred for otolaryngology review, with the case discussed at a multidisciplinary team review for possible bilateral styloidectomy. He was deemed medically unfit for the procedure at that stage.
Outcome and follow-up
This man is able to mobilise independently with a circumducting gait secondary to left hemiparesis after weeks in a rehabilitation facility. He is receiving botulinum toxin injections in his left elbow flexors and left long finger flexors for the residual upper extremity spasticity.
Currently, he remains unemployed but is seeking alternative employment.
Discussion
First described in 1937 by Watt Eagle, ES represents a collection of symptoms secondary to impingement caused by the styloid process.4
The incidence of an ESP varies from 4.0% to 7.3% due to discrepancies in the quoted upper limit of the normal length of the styloid process (2.5–3.0 cm). While only 4% of the described population is symptomatic, it is suggested that ES has been underestimated due to its erratic manifestations.3 4
Two symptomatic variants are described, depending on the ESP’s angulation; the classic stylohyoid syndrome and stylocarotid artery syndrome.
Symptoms concerning the ‘stylocarotid’ variant are primarily related to compression of the sympathetic chain and carotid artery. Presentations may include syncope, visual symptoms, facial pain and cerebrovascular symptoms.5 6
CAD with related Horner’s syndrome1 3 7–9 and sinus thrombosis10 are uncommon complications of this variant.
Bilateral carotid dissection secondary to ES is currently documented in only five other cases in the medical literature.3 7–10
However, in our case, dissections of the carotid arteries were unlikely to have occurred simultaneously. Due to the old frontal infarct, it is highly probable that the left ICAD had already manifested prior to hospitalisation, with the right ICAD transpiring with his onset of symptoms.
Due to its ambiguous presentation, ES is considered a diagnosis of exclusion.11 12 From our case, AHCM was considered as a possible alternative diagnosis for the ischaemic events, as one-third of AHCM patients experience cardiovascular complications and arrhythmias.13 There were no arrhythmias on cardiac monitoring and no thrombi or other embolic sources on the echocardiogram. This, together with the radiological findings, made AHCM a less likely aetiological factor.
CT angiography of the neck is considered the gold standard of the vascular variant of ES.14 15 In our case, this was essential in providing three-dimensional reconstruction of the distance between the ESP and carotid arteries, clinching the diagnosis of ES.
Over the past 10 years, 17 cases of carotid dissection secondary to ES were reported in literature, with triggering events and management reviewed in table 1.
Table 1.
Literature review of bilateral CAD secondary to ESPs
| Author (year) | Age, sex | Laterality of ESP | Triggering event | Diagnosis | Initial treatment | Additional treatment |
| Farhat et al 6 2009 | 70 M | Left | Mechanical fall | TIA and right ICAD | Styloidectomy | None |
| Faivre et al 7 2009 | 60 M | Bilateral | Shake dancing | Right MCA infarct and bilateral ICAD | Antithrombotic therapy and styloidectomy | None |
| Razak et al 28 2014 | 41 M | Right | Forced sustained head turning | Right MCA territory infarct and Right ICAD | Intravenous thrombolysis and styloidectomy | None |
| Todo et al 8 2012 | 57 M | Bilateral | Vigorous cervical massage | TIA and bilateral ICAD | Intravenous anticoagulation therapy | Following further TIA, Thrombectomy and CAS |
| Ohara et al 21 2012 | 43 M | Bilateral | Spontaneous | Right ICAD | Intravenous anticoagulation and oral antiplatelet | Declined surgical treatment |
| Sveinsson et al 16 2013 | 38 M | Left | Spontaneous | Left parietal and occipital lobe infarcts and left CAD | Intravenous thrombolysis, thrombectomy, dual anti platelets and CAS | After recurrent lacunar infarct, styloidectomy |
| Sveinsson et al 16 2013 | 41 F | Right | Intensive physical exercise | Right small pseudo aneurysm and ICAD | Intravenous anticoagulation then oral anticoagulation | None after discussion with otolaryngologist |
| Yamamoto et al 23 2013 | 51 M | Right | Repeated neck rotation | Right ICAD | Single antiplatelet | None |
| Ogura et al 9 2015 | 55 M | Bilateral | Spontaneous | Left temporal lobe infarct and bilateral ICAD | Intravenous anticoagulation with heparin | After ICA occlusion, styloidectomy and CAS |
| Ogura et al 9 2015 | 55 M | Right | Cervical massage | Right cerebral infarct and Right ICAD | Endovascular thrombectomy and CAS, intravenous anticoagulation with heparin | None |
| Ogura et al 9 2015 | 80 M | Left | Spontaneous | Left ICA dissecting aneurysm | Styloidectomy | None |
| Aydin et al 25 2015 | 57 F | Right | Spontaneous | Right MCA infarct and right ICAD | Oral anticoagulation | None |
| Dewan et al 3 2016 | 49 M | Bilateral | Spontaneous | Left parietal and occipital lobe infarcts and bilateral ICAD | Dual antiplatelets and CAS | None |
| Hebant et al 22 2017 | 57 M | Bilateral | Spontaneous | Left MCA territory infarct and left ICAD | Single antiplatelet | None |
| Subedi et al 26 2017 | 47 F | Right | Prolonged head positioning using cellphone | Right MCA territory infarct and Right ICAD | Anticoagulation and CAS | None |
| Smoot et al 24 2017 | 60 M | Bilateral | Boxing career | infarcts in the right parietal cortex and Right ICAD | Intravenous anticoagulation with heparin | CAS after new parietal lobe infarct episode |
| Jelodar et al 10 2018 | 40 M | Bilateral | Spontaneous | Multiple ischaemic strokes | Antiplatelet medication and bilateral styloidectomy | None |
CAD, carotid artery dissection; CAS, carotid artery stenting; ESP, elongated styloid process; ICAD, internal carotid artery dissection; MCA, middle cerebral artery; TIA, transient ischaemic attack.
The mean age of onset was 49.7 years (ranging from 38 to 80). The female to male ratio was 3:14, showing a strong male predominance.
In nine of the above cases, it was suggested that strenuous neck movements acted as triggering events. The aetiology of ES is still uncertain, with suggestions including genetic hypothesis, persistence of cartilaginous styloid process precursors and age-related inflammatory changes.15
A popular proposition involves ossification of the stylohyoid complex secondary to trauma or persistent irritation.12 16 With the patient’s involvement in a motor vehicle accident a few years ago, it could be hypothesised that bilateral ESP may have occurred due to trauma inflicted on the styloid processes.
No randomised control trials for guidelines concerning medical, interventional or surgical treatment of ICAD for ES are present.5 11
Medical therapy is often considered as first line. The American College of Cardiology Foundation & American Heart Association guidelines for CAD17 advise anticoagulation or antiplatelet use for least 3–6 months (class IIa evidence). No randomised studies have yet compared antiplatelet against anticoagulation use.18
In the Cervical Artery Dissection in Stroke Study trial, aspirin, dipyridamole or clopidogrel was administered as monotherapy or in combination.19 Treatment was at the discretion of the physician with no current guidelines in place.
Positive effects of dual antiplatelet use were hinted at by Perry and Al-Ali when reporting lower stroke recurrence or haemorrhage in CAD compared with the literature. However, the authors further proposed larger prospective studies to confirm optimal medical treatment combinations.20
In our index patient, due to the multiple ischaemic infarcts with no evidence of haemorrhagic conversion, the treating neurologist opted for dual antiplatelet therapy.
From the literature review, eight patients underwent solely medical therapy initially,8 9 16 21–25 with three patients requiring surgical or endovascular interventions.8 9 26
In contrast, use of endovascular therapy is not well established in literature with intrastent thrombosis and thromboembolism after carotid artery stenting (CAS) recently reported.9
From our review four patients underwent initial CAS,3 9 16 26 with one patient requiring further management due to stroke recurrence.16 However, two cases reported use of CAS as additional treatment following recurrent ischaemic events, with no other episodes reported.8 24
Hoving et al have advised further analysis of treatment trials to assess its therapeutic effectiveness.27
Surgical management with styloidectomy is considered to be the definitive treatment of ES.3 This is clearly seen from table 1, with seven patients who underwent surgical interventions (as initial or secondary treatment) not experiencing further symptoms.6 7 9 10 16 28
Therefore, we strongly recommend considering surgical management in the early stage of treatment to prevent further cerebrovascular events.
The intraoral or transcervical approach can be used, both having similar success rates.6 The latter is generally preferred in view of improved sterility and better visualisation for easier removal of the ESP.3 6 The advantages of the intraoral approach are decreased operative timing and risk of facial nerve damage.11
Finally, long-term treatment of such patients involves continuation of antithrombotic therapy and risk factor modification. The latter includes treatment of hypertension, hyperlipidaemia, diabetes and smoking cessation.17
Learning points.
Diagnosis is confirmed based on both symptomatology and radiological evidence.
Underestimation of Eagle’s syndrome may occur due to its diffuse array of presentation.
CT angiography with three-dimensional reconstruction is the gold standard for confirming an elongated styloid process and its spatial relationships.
No clinical practice guidelines are present for carotid artery dissection secondary to Eagle’s syndrome. While medical and endovascular therapy play a therapeutic role, early surgical consideration is strongly advised for prevention of further ischaemic events.
Acknowledgments
Consultant neurologist Josanne Aquilina, Nicola Dingli (Resident Specialist Neurology) and Ruth Attard (BST General Medicine) who where responsible for the care of the patient at Mater Dei Hospital.
Footnotes
Contributors: MZ wrote up the case report and discussion. MD and CC reviewed the report, with corrections and necessary alterations to the case report carried out. VA provided the imaging figures.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: None declared.
Patient consent: Obtained.
Provenance and peer review: Not commissioned; externally peer reviewed.
References
- 1. Zetterling M, Carlström C, Conrad P. Review article - Internal carotid artery dissection. Acta Neurol Scand 2000;101:1–7. 10.1034/j.1600-0404.2000.00001.x [DOI] [PubMed] [Google Scholar]
- 2. Mathews T, Achar R, Rattan G. Eagle Syndrome-vascular type-A case presentation and a short review of literature. Case Report 2012. 10.1594/ranzcraocr2012/R-0032 [DOI] [Google Scholar]
- 3. Dewan MC, Morone PJ, Zuckerman SL, et al. Paradoxical ischemia in bilateral Eagle syndrome: a case of false-localization from carotid compression. Clin Neurol Neurosurg 2016;141:30–2. 10.1016/j.clineuro.2015.12.004 [DOI] [PubMed] [Google Scholar]
- 4. Piagkou M, Anagnostopoulou S, Kouladouros K, et al. Eagle’s syndrome: a review of the literature. Clin Anat 2009;22:545–58. 10.1002/ca.20804 [DOI] [PubMed] [Google Scholar]
- 5. Badhey A, Jategaonkar A, Anglin Kovacs AJ, et al. Eagle syndrome: a comprehensive review. Clin Neurol Neurosurg 2017;159:34–8. 10.1016/j.clineuro.2017.04.021 [DOI] [PubMed] [Google Scholar]
- 6. Farhat HI, Elhammady MS, Ziayee H, et al. Eagle syndrome as a cause of transient ischemic attacks. J Neurosurg 2009;110:90–3. 10.3171/2008.3.17435 [DOI] [PubMed] [Google Scholar]
- 7. Faivre A, Abdelfettah Z, Rodriguez S, et al. Neurological picture. Bilateral internal carotid artery dissection due to elongated styloid processes and shaking dancing. J Neurol Neurosurg Psychiatry 2009;80:1154–5. 10.1136/jnnp.2008.159954 [DOI] [PubMed] [Google Scholar]
- 8. Todo T, Alexander M, Stokol C, et al. Eagle syndrome revisited: cerebrovascular complications. Ann Vasc Surg 2012;26:729.e1–5. 10.1016/j.avsg.2011.12.005 [DOI] [PubMed] [Google Scholar]
- 9. Ogura T, Mineharu Y, Todo K, et al. Carotid artery dissection caused by an elongated styloid process: three case reports and review of the literature. NMC Case Rep J 2015;2:21–5. 10.2176/nmccrj.2014-0179 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Jelodar S, Ghadirian H, Ketabchi M, et al. Bilateral ischemic stroke due to carotid artery compression by abnormally elongated styloid process at both sides: a case report. J Stroke Cerebrovasc Dis 2018;27:e89–91. 10.1016/j.jstrokecerebrovasdis.2017.12.018 [DOI] [PubMed] [Google Scholar]
- 11. Bokhari M, Bhimji S. Eagle Syndrome. 2017.
- 12. Bahgat M, Bahgat Y, Bahgat A. Eagle’s syndrome, a rare cause of neck pain. BMJ Case Rep 2012;2012:bcr2012006278 10.1136/bcr-2012-006278 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Haruki S, Minami Y, Hagiwara N. Stroke and embolic events in hypertrophic cardiomyopathy: risk stratification in patients without atrial fibrillation. Stroke 2016;47:936–42. 10.1161/STROKEAHA.115.012130 [DOI] [PubMed] [Google Scholar]
- 14. Chuang WC, Short JH, McKinney AM, et al. Reversible left hemispheric ischemia secondary to carotid compression in Eagle syndrome: surgical and CT angiographic correlation. AJNR Am J Neuroradiol 2007;28:143–5. [PMC free article] [PubMed] [Google Scholar]
- 15. Steinmann EP. A new light on the pathogenesis of the styloid syndrome. Arch Otolaryngol 1970;91:171–4. 10.1001/archotol.1970.00770040241013 [DOI] [PubMed] [Google Scholar]
- 16. Sveinsson O, Kostulas N, Herrman L. Internal carotid dissection caused by an elongated styloid process (Eagle syndrome). BMJ Case Rep 2013;2013:bcr2013009878 10.1136/bcr-2013-009878 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Brott TG, Halperin JL, Abbara S, et al. Guideline on the management of patients with extracranial carotid and vertebral artery disease. J Am Coll Cardiol 2011;57:1002–44. [DOI] [PubMed] [Google Scholar]
- 18. Chowdhury MM, Sabbagh CN, Jackson D, et al. Antithrombotic treatment for acute extracranial carotid artery dissections: a meta-analysis. Eur J Vasc Endovasc Surg 2015;50:148–56. 10.1016/j.ejvs.2015.04.034 [DOI] [PubMed] [Google Scholar]
- 19. Markus HS, Hayter E, Levi C, et al. Antiplatelet treatment compared with anticoagulation treatment for cervical artery dissection (CADISS): a randomised trial. Lancet Neurol 2015;14:361–7. 10.1016/S1474-4422(15)70018-9 [DOI] [PubMed] [Google Scholar]
- 20. Perry BC, Al-Ali F. Spontaneous cervical artery dissection: the borgess classification. Front Neurol 2013;4:133 10.3389/fneur.2013.00133 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Ohara N, Sakaguchi M, Okazaki S, et al. Internal carotid artery dissection caused by an elongated styloid process: usefulness of transoral ultrasonography. J Stroke Cerebrovasc Dis 2012;21:918.e7–8. 10.1016/j.jstrokecerebrovasdis.2012.05.014 [DOI] [PubMed] [Google Scholar]
- 22. Hebant B, Guegan-Massardier E, Macaigne V, et al. Ischemic stroke due to internal carotid artery dissection associated with an elongated styloid process (Eagle syndrome). J Neurol Sci 2017;372:466–7. 10.1016/j.jns.2016.10.055 [DOI] [PubMed] [Google Scholar]
- 23. Yamamoto S, Todo K, Kawamoto M, et al. Carotid artery dissection associated with an elongated styloid process. Intern Med 2013;52:1005–6. 10.2169/internalmedicine.52.0024 [DOI] [PubMed] [Google Scholar]
- 24. Smoot TW, Taha A, Tarlov N, et al. Eagle syndrome: A case report of stylocarotid syndrome with internal carotid artery dissection. Interv Neuroradiol 2017;23:433–6. 10.1177/1591019917706050 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. AYDIN E, QULIYEV H, CINAR C. Eagle Syndrome Presenting with Neurological Symptoms. Turk Neurosurg 2016:1. [DOI] [PubMed] [Google Scholar]
- 26. Subedi R, Dean R, Baronos S, et al. Carotid artery dissection: a rare complication of Eagle syndrome. BMJ Case Rep 2017;2017:bcr2016218184 10.1136/bcr-2016-218184 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Hoving JW, Marquering HA, Majoie C. Endovascular treatment in patients with carotid artery dissection and intracranial occlusion: a systematic review. Neuroradiology 2017;59:641–7. 10.1007/s00234-017-1850-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Razak A, Short JL, Hussain SI. Carotid artery dissection due to elongated styloid process: a self-stabbing phenomenon. J Neuroimaging 2014;24:298–301. 10.1111/j.1552-6569.2012.00759.x [DOI] [PubMed] [Google Scholar]






