Abstract
Background:
Pancreatic cancer is a deadly disease for which available biomarkers, such as CA19–9, lack the desired sensitivity and specificity for early detection. Additional biomarkers are needed to improve both its sensitivity and specificity.
Methods:
Multiplex immunoassays were developed for selected biomarkers using a Bio-Plex 200 system and analytical performance optimized. All proteins were analyzed in sera of patients diagnosed with pancreatic ductal adenocarcinoma (PDAC, n=188) or benign pancreatic conditions (131), and healthy controls (89). The clinical performance of these markers were evaluated individually or in combination for their ability to complement CA19–9 for the early detection of pancreatic cancer.
Results:
A 6-plex immunoassay was developed with negligible cross-reactivity, wide dynamic ranges, recovery of 89–104%, and intra-assay and inter-assay precision of 10.2–19.6% and 13.7–29.3%, respectively. Individually, the best biomarkers to separate PDAC early stage from chronic pancreatitis or intraductal papillary mucinous neoplasm (IPMN) were CA19–9 and MIA or CA19–9 and MIC-1. Logistic regression modelling selected the two-marker panels that significantly improved the individual biomarker performance in discriminating PDAC early stage from chronic pancreatitis (AUCCA19–9+MIA=0.86 versus AUCCA19–9=0.81 or AUCMIA=0.75 only, p<0.05) or IPMN (AUCCA19–9+MIC-1=0.81 versus AUCCA19–9=0.75 or AUCMIC-1=0.73 only, p<0.05). It was observed that OPN outperformed CA19–9 in separating IPMN from chronic pancreatitis (AUCOPN=0.80 versus AUCCA19–9=0.70, p<0.01).
Conclusions:
The biomarker panels evaluated by assays with high analytical performance demonstrated potential complementary values to CA19–9, warranting additional clinical validation to determine their role in early detection of pancreatic cancer.
Impact:
The validated biomarker panels could lead to earlier intervention and better outcomes.
Keywords: Multiplex, immunoassay, Serum, Biomarker, Pancreatic Cancer
Introduction
Pancreatic cancer is the fourth leading cause of cancer death in the United States (1). The majority of patients are diagnosed in advanced and unresectable stages with a median survival of 6 months and an overall 5-year survival of < 5% (2). The early detection of pancreatic ductal adenocarcinoma (PDAC) is critical because surgery at an early stage is the most promising therapy that greatly improves prognosis (3). However, there are currently no available screening tests for the early detection of PDAC. Conventional imaging tools such as abdominal computerized tomography (CT) scanning, magnetic resonance imaging (MRI), endoscopic retrograde cholangiopancreatography (ERCP) and endoscopic ultrasound (EUS) are inadequate for detecting small premalignant lesions and are relatively costly, time-consuming and invasive (4,5). The current gold-standard serum marker cancer antigen 19–9 (CA19–9) is an FDA-cleared marker with the intended clinical use for monitoring disease status (6–8). It lacks the necessary sensitivity and specificity and has been shown to be ineffective in mass screening of asymptomatic subjects (6–8). CA19–9 is frequently elevated in non-malignant conditions such as pancreatitis, obstructive jaundice, and other benign conditions (6–8). It is also unsuitable for use in 5–10% patients who have a Lewis-negative genotype and do not express the antigen (8). All of these factors limit its clinical utility in a screening and early detection setting. There is an urgent clinical need to identify additional biomarkers to complement CA19–9 for use in the early detection of pancreatic cancer.
Recently, several alternative serum biomarkers have been identified for use in the detection of PDAC. These include macrophage inhibitory cytokine-1 (MIC-1), carcinoembryonic antigen cell adhesion molecule-1 (CEACAM-1), regenerating islet-derived protein 4 (REG-4), osteopontin (OPN), tissue inhibitor of metalloproteinase 1 (TIMP-1), tissue polypeptide-specific antigen (TPS), and others (9–16). However, none of these have been clinically proved to be superior to CA19–9 (11,17) even though as reported by several research groups that the combinations of individual serum biomarkers could improve their performance for the detection of pancreactic cancer (18,19). An increasing number of novel candidate biomarkers are being identified using high-throughput proteomic technologies (9–19). Such candidates, along with potential biomarkers reported in the literature, need to be rigorously validated using assays with sufficiently high analytical performances on clinical samples from relevant patient populations. Traditional enzyme-linked immunosorbent assays (ELISAs) only measure a single antigen at a time, which can be a major challenge for simultaneously quantification of multiple potential biomarkers across large cohorts of patient samples of which available sample volumes are often a limiting factor. Magnetic bead-based multiplex immunoassays use differentially detectable bead sets as substrates capturing analytes in solution and detection antibodies measuring quantities of analytes in a single sample, and represent a promising solution to simultaneously measure multiple analytes in a single sample using minimum sample volume. Compared with traditional ELISA and planar microarray, magnetic bead-based immunoassays may demonstrate faster solution-phase kinetics instead of solid-phase kinetics and lower limits of quantification (20,21). In this study, we hypothesized that combinations of individual serum biomarkers could offer a superior diagnostic ability in early detection of pancreatic cancer. To this end, we have performed bioinformatical analysis of publicly available gene, protein, and PUBMED databases to identify candidate biomarkers based on reported fold changes and/or sensitivity/specificity weighted by number of publications and cumulative study sample sizes. Magnetic bead-based multiplex immunoassays were developed for the selected candidate serum biomarkers using a Bio-Plex 200 suspension array system (Bio-Rad), and applied to a case-control set of serum samples from subjects with PDAC or benign conditions, and healthy controls. The performance of these candidate biomarkers were evaluated individually and in combination for their ability to complement CA19–9 for the early detection of pancreatic cancer.
Materials and Methods
Specimens
A total of 408 archived serum samples obtained from 188 patients with histologically diagnosed PDAC, 131 patients with benign pancreatic conditions, and 89 healthy controls without a history of pancreatic diseases were studied with institutional approval. All patient serum samples were obtained before surgery or other treatment, and stored at −80°C until analysis.
Reagents and antibodies
All of the recombinant proteins and antibodies were purchased from R&D Systems (Minneapolis, MN)), except the detection antibody for SPON1 which was biotinylated in-house. The majority of the antibodies except those for OPN and SPON1 were from the DuoSet ELISA kits (R&D). Detailed information on the recombinant proteins and antibodies are provided in Supplement Table 1. Magnetic COOH beads, amine coupling kits, and Bio-Plex Pro Reagent kits were purchased from Bio-Rad Laboratories (Hercules, CA). NHS and Sulfo-NHS, EDC, EZ-Link™ Sulfo-NHS-Biotin, and Zeba™ Spin Desalting Columns were purchased from Thermo Scientific (Rockford, IL). Serum CA19–9 concentrations were measured using an FDA cleared assay on the Tosoh AIA-600II immunoassay analyser (Tosoh Bioscience, S. San Francisco, CA). The human osteopontin ELISA kit (ABIN414433) and the human heat shock protein 27 ELISA kit (ab113334) were purchased from Antibodies-Online (Atlanta, GA) and Abcam (Cambridge, MA), respectively.
Conjugation of antibodies to microspheres
Capture antibodies for OPN, MIA, CEACAM-1, MIC-1, SPON1 and HSP27 were coupled to magnetic beads of different regions (64, 46, 26, 34, 43 & 63) using the Bio-Rad amine coupling kit according to the manufacturer’s instructions. The use of differentially detectable beads of the different regions enables the simultaneous identification and quantification of multiple analytes in the same sample and the individual immunoassays therefore can be multiplexed. The optimal amount of capture antibodies for one coupling reaction after titration was either 6 μg for OPN, MIA, CEACAM-1, MIC-1 and HSP27 or 9 μg for SPON1. The coupled beads were counted using a Coulter Z2 counter, validated using biotinylated rabbit anti-mouse (B8520) or rabbit anti-goat (B7014) IgG antibodies (Sigma-Aldrich, St. Louis, MO), and stored in storage buffer provided in the kit at 4°C in the dark.
Multiplex immunoassay
A magnetic bead-based multiplex immunoassay was developed for the selected candidate serum biomarkers using a Bio-Plex 200 suspension array system (Bio-Rad, Hercules, CA). The monoplex immunoassays for individual candidates were first developed using the Bio-Plex Pro Reagent kit. Briefly, 2500 coupled beads were incubated with 50 μl of a sample diluted in sample diluent for 1 hour. The beads were washed and incubated with 25 μl of the detection antibody diluted in the detection antibody diluent for 30 minutes. The beads were then washed again and incubated with 50 μl of 2 μg/mL streptavidin-phycoerytherin (SA-PE) diluted in the assay buffer for 10 minutes. The beads were finally washed and suspended in 125 μl of the assay buffer for the analysis by the Bio-Plex 200 system. All assays were carried out at room temperature and protected from light. All washing steps were performed with the wash buffer with an automated plate washer (Bio-Plex Pro™ II wash station, Bio-Rad). Calibration curves were established using 9 calibrators in a 2-fold dilution series. Two pooled human normal sera (one internal pooled sera and the other S7023 from Sigma-Aldrich) were used for the optimization of the assay conditions.
Before multiplexing the individual assays, assay specificity was examined by performing single-detection and multiplexed-detection antibody cross-reactivity studies to detect the fluorescence signals in response to high concentrations of the recombinant proteins at the first dilution point of the standard curve (except SPON1 at the third dilution). The single detection antibody study was conducted by testing an individual detection antibody in the presence of multiplexed capture beads and a single antigen, which evaluates the specificity of a capture antibody. The multiplexed-detection antibody study was conducted by testing multiplexed detection antibodys in the presence of multiplexed capture beads and a single antigen, which evaluates the specificity of a detection antibody and to some degree the specificity of the capture antibody. Cross-reactivity was defined as the percentage of nonspecific cross-reacting signal detected relative to the specific signal for that analyte.
For the multiplex immunoassay, the capture beads and the detection antibodies were prepared by mixing the 2500 coupled beads and the detection antibodies used in the monoplex assays. The final concentrations of the detection antibodies in the multiplex assay were used at 0.4 μg/mL for OPN and CEACAM-1, 2 μg/mL for SPON1, 0.2 μg/mL for MIA and HSP27, and 0.0125 μg/mL for MIC-1, respectively, after the titration. The calibration curve was established using 9 calibrators in 2-fold dilution series in the standard diluent derived from a mixture of the highest standard points of the 6 recombinant proteins. The highest standards for the 6 recombinant proteins in the multiplex assay were 40, 1.5, 20, 3, 15 and 3 ng/mL for OPN, MIA, CEACAM-1, MIC-1, SPON1 and HSP27, respectively. The multiplex immunoassays were compared to the monoplex immunoassays by measuring 4 independent doses of individual recombinant proteins based on their respective calibration curves. The correlations of the developed multiplex immunoassays and commercial ELISA kits in serum OPN or HSP27 protein quantifications were also determined in 7 or 13 patient sera, respectively. The multiplex immunoassay was carried out using the Bio-Plex Pro Reagent kit in the same procedures as those in the monoplex assays described above. The serum samples were 4-fold diluted in the sample diluent in the multiplex immunoassay. Two quality controls (QC) were prepared by diluting the mixture of the highest standards of 6 recombinant proteins at either 3-fold (QC1) or 30-fold (QC2) in the standard diluent. Two pooled human sera with the known CA19–9 measurements at either high or low levels were used as the additional controls. The multiplex immunoassay was performed in duplicate on 13× 96-well Bio-Plex flat bottom plates. All samples were randomized with regard to their plate locations.
Data acquisition and primary data analysis were performed on the Bio-Plex 200 system in combination with Bio-Plex Manager Software version 6.1.1 by use of a 5-parametric (5-PL) nonlinear logistic regression curve fitting model (Bio-Rad). Assay sensitivity (limit of blank, LOB) was defined as the concentration of analyte corresponding to the median fluorescent intensity (MFI) of the background plus two standard deviations (SD) of the mean background MFI. Intra-assay precision was calculated as the coefficient of variance (%CV) on 4 replicates of the pooled normal sera or two QCs on a single assay plate. Inter-assay precision was calculated as the %CV from 6 independent assays. The assay recovery was calculated as the percentage of the observed concentration relative to the expected concentration of each standard point or QC. The assay working dynamic range was defined as the range between the lower limit of quantification (LLOQ) and the upper limit of quantification (ULOQ) in which an assay is both precise (intra-assay %CV ≤10% and inter-assay %CV ≤15%) and accurate (80–120% recovery).
Data analysis
Analysis of variance (ANOVA) and the nonparametric Mann-Whitney U test were used to compare serum biomarker levels between subjects with PDAC or benign pancreatic conditions and healthy controls, with a p-value less than 0.05 considered significant. Receiver-operating-characteristic (ROC) curve analysis was performed and the area under the curve (AUC) was calculated separately for each of 7 biomarkers and the combinations of biomarkers. The Delong test was used to compare the AUCs. Pearson correlation coefficients were determined to assess correlation of the measurements between the multiplex and monoplex immunoassays or commercial ELISA kits, and were also used to evaluate the association of markers with age separately in the healthy controls, benign conditions and PDAC patient groups. Logistic regression modeling was constructed including age as a covariate and backward stepwise selected z-score transformed variables with the highest performance. To ensure that the observed performance was not due to overfitting, selected models were further compared against the performance of models constructed with label permuted samples. The Statistica 13 (StatSoft), GraphPad Prism 6 (GraphPad Software), and R (https://www.R-project.org) were used for statistical analysis.
Results
Biomarker selection and development of a 6-plex immunoassay
Comprehensive literature search and in silico analysis of publicly available gene and protein databases were performed to identify biomarker candidates reported to be involved in the development and progression of PDAC and also measureable in human serum. The final candidates were selected for the multiplex immunoassay development based on the commercial availability of appropriate pairs of capture and detection antibodies and their relative abundances in human serum samples. Customized magnetic bead-based multiplex immunoassays were developed for the selected candidate serum biomarkers using a Bio-Plex 200 suspension array system. Magnetic bead-based monoplex immunoassays were first developed for OPN, MIA, CEACAM-1, MIC-1, SPON1 and HSP27. The cross-reactivity studies through single-detection and mutiplexed-detection antibody experiments indicated that the degree of cross-reactivity across the 6 immunoassays was generally <1%, based on the measurements in response to high concentrations of the recombinant proteins at first dilution point (except SPON1 at the third dilution because only 1.4% of sera with SPON1 exceeded the third dilution) of the standard curve. Between 1.3–3.3% of nonspecific cross-reactions were observed with the SPON1 antibody against other proteins (data not shown). It should be noted that majority of these nonspecific cross-reactions were observed at recombinant protein concentrations that exceeded physiological levels, thereby reducing the chance of cross-reactivity in physiological human serum samples.
By combining the capture antibody-coupled beads and detection antibodies used in the monoplex immunoassays, a 6-plex immunoassay of OPN, MIA, CEACAM-1, MIC-1, SPON1 and HSP27 was developed and evaluated. The calibration curves of the 6-plex immunoassay were generated using the 5PL logistic regression models (Supplement Figure 1A–F). The 6-plex immunoassay results correlated significantly with their respective monoplex immunoassay results (p<0.05), suggesting that the 6-plex immunoassay was comparable to the monoplex immunoassays for protein quantification. Furthermore, there were significant correlations of OPN and HSP27 protein measurements using the 6-plex immunoassay compared to commercial ELISA kits (Table 1).
Table 1.
Analytical performance of the 6-plex immunoassay.
| Mean (pg/mL) | Intra-assay Precision (%CV) | Inter-assay Precision (%CV) | Intra-assay Precision (%CV) | Inter-assay Precision (%CV) | LOB (pg/mL) | LLOQ (pg/mL) | ULOQ (pg/mL) | 6-plex vs Monoplex, Pearson R/p value | 6-plex vs ELISA, Pearson R/p value | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| QC1 | QC2 | QC1 | QC2 | QC1 | QC2 | Pooled Normal Serum | Pooled Normal Se rum | ||||||
| OPN | 13448.3 | 1228.3 | 2.1 | 5.1 | 3.7 | 4.2 | 12.9 | 13.7 | 19.9 | 157.3 | 37174.3 | 0.9987/0.0013 | 0.8945/0.0066 |
| MIA | 481.7 | 48.3 | 8.6 | 10.0 | 4.4 | 8.4 | 10.2 | 13.7 | 0.1 | 6.7 | 1550.0 | 0.9888/0.0112 | ND* |
| CEACAM-1 | 6141.7 | 658.3 | 9.3 | 6.6 | 8.6 | 16.3 | 16.5 | 19.3 | 19.8 | 96.0 | 20042.7 | 0.9715/0.0285 | ND |
| MIC-1 | 963.3 | 93.3 | 8.1 | 10.3 | 21.5 | 18.8 | 14.1 | 17.4 | 0.1 | 12.0 | 2383.7 | 0.9996/0.0004 | ND |
| SPON1 | 4746.7 | 473.3 | 4.3 | 2.7 | 5.0 | 6.8 | 14.8 | 16.8 | 12.5 | 60.3 | 15124.7 | 0.9675/0.0325 | ND |
| HSP27 | 933.3 | 88.3 | 15.4 | 12.6 | 14.5 | 13.2 | 19.6 | 29.3 | 0.5 | 12.0 | 3078.0 | 0.9997/0.0003 | 0.9254/<0.00001 |
NOTE: QC1, high control. QC2, low control. LOB, limit of blank. LLOQ, lower limit of quantitation. ULOQ, upper limit of quantification.
The correlation of 6-plex vs monoplex was examed on 4 doses of individual recombinant proteins. The correlation of 6-plex vs the
commercial ELISA kit was examed on 7 (OPN) or 13 (HSP27) patient sera.
, ND, not determined.
The analytical performance of the 6-plex immunoassay is shown in Table 1, with recovery of 89–104% (standard curve points and QCs), intra-assay precision of 2.1–15.4% (QCs) or 10.2–19.6% (pooled normal serum) and inter-assay precision of 3.7–21.5% (QCs) or 13.7–29.3% (pooled normal serum). The 6-plex immunoassay exhibited wide dynamic concentration ranges; the calibration curves covered (>2 logs) defined by LLOQ and ULOQ, and low LOBs for target protein quantification.
Application of the 6-plex immunoassay in the detection of PDAC
The developed 6-plex immunoassay was used to analyze the target protein levels in sera of 188 patients diagnosed with PDAC [mean (SD) age, 65 (10) years; M/F, 81/107], 131 patients with benign pancreatic conditions [57 (15) years; 71/60], and 89 healthy controls [35 (14) years; 45/44]. Among the 188 patients with PDAC, there were 96 with early stage [IA/IB/IIA/IIB, 13/18/17/48; 65 (10) years; 34/62] and 92 with late stage [III/IV, 19/73; 64 (10) years; 47/45] disease. Among 131 patients with benign pancreatic conditions, there were 63 with intraductal papillary mucinous neoplasms (IPMN) [64 (12) years; 24/39] and 68 with chronic pancreatitis [51 (15) years; 47/21]. Detailed clinicopathologic characteristics of the study cohort, including diagnosis, age, sex and anatomic stage, are shown in Table 2. The performance of the individual markers was compared to CA19–9 to discriminate between PDAC patients and benign conditions or healthy controls (Supplement Figure 2A–G). Serum levels of OPN, CEACAM-1, MIC-1, SPON1 & CA19–9 or MIA were significantly increased or decreased in benign conditions compared to healthy controls (OPN, CEACAM-1 & MIC-1 at p<0.0001, SPON1 & CA19–9 at p<0.01, and MIA at p<0.0001). Serum levels of OPN, CEACAM-1, MIC-1, SPON1 & CA19–9 were also significantly increased in PDAC patients compared to healthy controls (all at p<0.0001). Furthermore, serum levels of OPN, MIA, CEACAM-1, MIC-1 & CA19–9 were significantly increased in PDAC patients compared to benign conditions (all at p<0.0001, except MIA at p<0.001). Based on ROC curve analysis (Supplement Figure 3A, C&E), individually, the four best biomarkers to separate benign conditions from healthy controls were MIC-1 (AUC=0.91, [0.86–0.94]), CEACAM-1 (0.81, [0.75–0.86]), MIA (0.81, [0.75–0.86]), and OPN (0.80, [0.74–0.85]). The four best biomarkers to separate PDAC patients from healthy controls were MIC-1 (0.97, [0.94–0.98]), CA19–9 (0.93, [0.89–0.96]), CEACAM-1 (0.91, [0.87–0.94]), and OPN (0.90, [0.86–0.94]). The four best biomarkers to separate PDAC from benign condisions were CA19–9 (0.82, [0.77–0.86]), MIC-1 (0.69, [0.64–0.74]), CEACAM-1 (0.67, [0.62–0.73]), and MIA (0.66, [0.60–0.71]). Logistic regression modeling was constructed by backward stepwise selection using z-score transformed variables including age as a covariate (Supplement Table 2 & Supplement Figure 3B,D&F). A three-marker panel of MIC-1 (p<0.0001), MIA (p=0.0001) & OPN (p=0.0037) remained in the model which had an AUC of 0.94 (0.90–0.96) that was greater than the individual biomarkers for benign conditions versus healthy controls (p value: MIC-1 at 0.0073, MIA at <0.0001, OPN at <0.0001, or CA19–9 at <0.0001). A three-marker panel of CA19–9 (p=0.0014), MIC-1 (p<0.0001) & OPN (p=0.0060) remained in the model which had an AUC of 0.99 (0.97–1.00) that was greater than the individual biomarkers for PDAC versus healthy controls (p value: CA19–9 at <0.0001, MIC-1 at 0.0005, or OPN at <0.0001). A three-marker panel of CA19–9 (p<0.0001), CEACAM-1 (p=0.0520) & MIA (p=0.0001) remained in the model which had an AUC of 0.86 (0.81–0.89) that was greater than the individual biomarkers for PDAC versus benign conditions (p value: CA19–9 at 0.0055, CEACAM-1 at <0.0001, or MIA at <0.0001). In comparison, the AUC of models of the same three-marker panel constructed using label-permuted samples was 0.56 (0.50–0.60).
Table 2.
Clinicopathologic characteristics of the study cohort.
| Variables | Number (%) |
|---|---|
| Total | 408 |
| Healthy controls | 89 (21.8) |
| Age (years) | |
| Mean±SD | 35±14 |
| Range (Median) | 21–67 (29) |
| Gender | |
| Male | 45 (50.6) |
| Female | 44 (49.4) |
| Benign conditions | 131 (32.1) |
| Age (years) | |
| Mean±SD | 57±15 |
| Range (Median) | 13–89 (59) |
| Gender | |
| Male | 71 (54.2) |
| Female | 60 (45.8) |
| Chronic pancreatitis | 68 (51.9) |
| IPMN | 63 (48.1) |
| PDAC | 188 (46.1) |
| Age (years) | |
| Mean±SD | 65±10 |
| Range (Median) | 30–92 (65) |
| Gender | |
| Male | 81 (43.1) |
| Female | 107 (56.9) |
| Early stage | 96 (51.1) |
| IA/IB/IIA/IIB | 13/18/17/48 |
| Late stage | 92 (48.9) |
| III/IV | 19/73 |
NOTE: IPMN, intraductal papillary mucinous neoplasm.
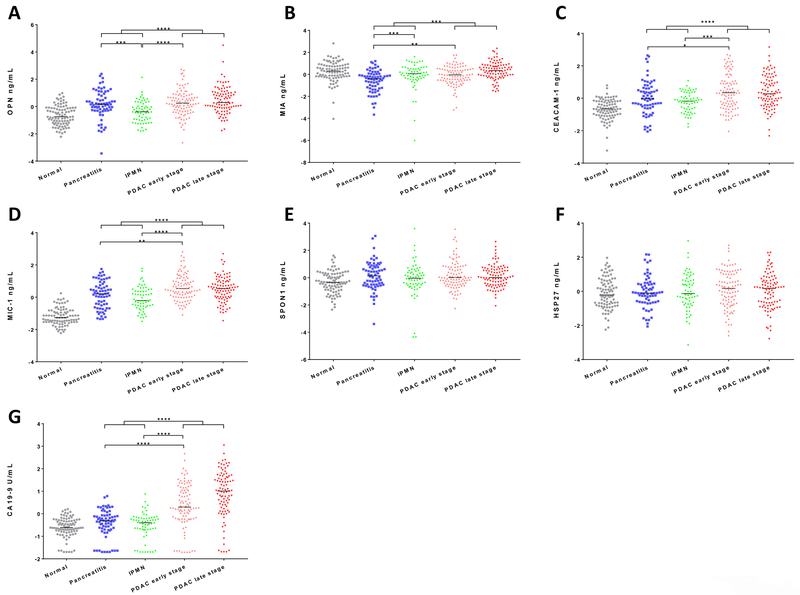
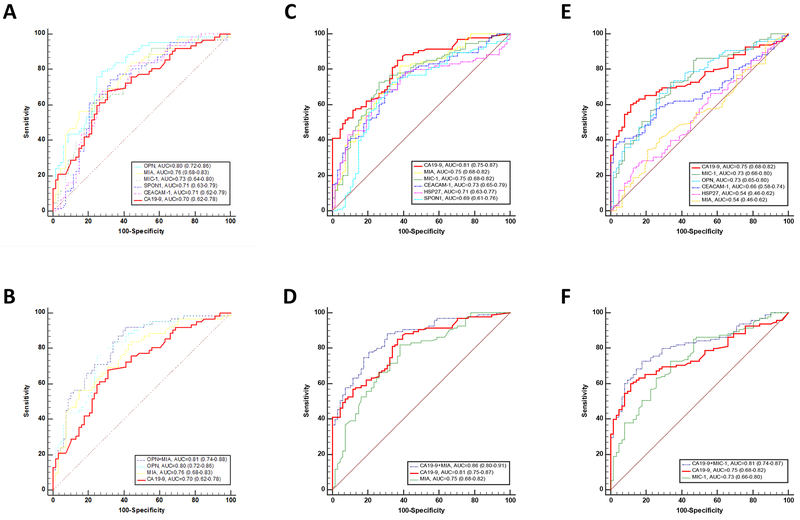
Serum levels of individual biomarkers were further analyzed in different subgroups consisting of 89 healthy controls, 68 chronic pancreatitis, 63 IPMN, 96 PDAC early stage, and 92 PDAC late stage patients (Table 3 & Figure 1A–G). Serum levels of OPN or MIA were significantly decreased or increased in IPMN compared to chronic pancreatitis patients (both at p<0.001); however, there was no significant difference in serum CA19–9 levels between IPMN and chronic pancreatitis patients. Serum levels of MIA, CEACAM-1, MIC-1 & CA19–9 were significantly increased in PDAC early stage compared to chronic pancreatitis patients (MIA & MIC-1 at p<0.01, CEACAM-1 at p<0.05, and CA19–9 at p<0.0001). Serum levels of OPN, CEACAM-1, MIC-1 & CA19–9 were also significantly increased in PDAC early stage compared to IPMN patients (OPN, MIC-1 & CA19–9 at p<0.0001, CEACAM-1 at p<0.001). Based on ROC curve analysis (Figure 2A,C&E), individually, the three best biomarkers to separate IPMN from chronic pancreatitis based on the ROC curve analysis were OPN (0.80, [0.72–0.86]), MIA (0.76, [0.68–0.83]), and MIC-1 (0.73, [0.64–0.80]). The three best biomarkers to separate PDAC early stage from chronic pancreatitis were CA19–9 (0.81, [0.75–0.87]), MIA (0.75, [0.68–0.82]), and MIC-1 (0.75, [0.68–0.82]). The three best biomarkers to separate PDAC early stage from IPMN were CA19–9 (0.75, [0.68–0.82]), MIC-1 (0.73, [0.66–0.80]), and OPN (0.73, [0.65–0.80]). Logistic regression modeling was constructed by backward stepwise selection using z-score transformed variables including age as a covariate (Figure 2B,D&F). A two-marker panel of OPN (p=0.0110) & MIA (p=0.0143) remained in the model which had an AUC of 0.81 (0.74–0.88) that was greater than the individual biomarkers for IPMN versus chronic pancreatitis (p value: OPN at 0.5506, MIA at 0.0241, or CA19–9 at 0.0018). A two-marker panel of CA19–9 (p=0.0004) & MIA (p=0.0004) remained in the model which had an AUC of 0.86 (0.80–0.91) that was greater than the individual biomarkers for PDAC early stage versus chronic pancreatitis (p value: CA19–9 at 0.0314 or MIA at 0.0001). A two-marker panel of CA19–9 (p=0.0021) & MIC-1 (p=0.0122) remained in the model which had an AUC of 0.81 (0.74–0.87) that was greater than the individual biomarkers for PDAC early stage versus IPMN (p value: CA19–9 at 0.0208 or MIC-1 at 0.0291). In comparison, logistic regresion models of the above panels constructed using label-permuted samples all failed to be diagnostic with AUCs at 0.57 (0.47–0.65), 0.56 (0.48–0.63), and 0.56 (0.47–0.64) for IPMN versus chronic pancreatitis, PDAC early stage versus chronic pancreatitis, and PDAC early stage versus IPMN, respectively.
Table 3.
Statistics of individual biomarkers in healthy controls, benign conditions and PDAC patients.
| Biomarker | Subgroup | Number | Min | Max | Median | Mean | IQR |
|---|---|---|---|---|---|---|---|
| OPN | Healthy Control | 89 | 2.1 | 21.7 | 6.9 | 7.9 | 4.2 |
| Chronic Pancreatitis | 68 | 0.8 | 103.6 | 13.6 | 20.7 | 15.3 | |
| IPMN | 63 | 2.3 | 84.8 | 8.5 | 12.1 | 9.1 | |
| PDAC early stage | 96 | 2.0 | 135.3 | 14.3 | 19.1 | 15.5 | |
| PDAC late stage | 92 | 2.3 | 154.1 | 14.5 | 21.5 | 17.3 | |
| MIA | Healthy Control | 89 | 0.1 | 1.5 | 0.6 | 0.7 | 0.5 |
| Chronic Pancreatitis | 68 | 0.1 | 1.2 | 0.5 | 0.5 | 0.3 | |
| IPMN | 62* | 0.1 | 1.5 | 0.6 | 0.6 | 0.4 | |
| PDAC early stage | 96 | 0.1 | 1.4 | 0.5 | 0.6 | 0.4 | |
| PDAC late stage | 92 | 0.3 | 2.1 | 0.6 | 0.7 | 0.4 | |
| CEACAM-1 | Healthy Control | 89 | 2.3 | 26.9 | 12.7 | 13.0 | 7.1 |
| Chronic Pancreatitis | 68 | 5.2 | 93.2 | 17.1 | 23.4 | 16.7 | |
| IPMN | 63 | 5.7 | 35.4 | 16.1 | 17.0 | 9.8 | |
| PDAC early stage | 96 | 4.8 | 120.9 | 22.2 | 31.4 | 25.7 | |
| PDAC late stage | 92 | 4.6 | 117.8 | 21.9 | 29.3 | 18.9 | |
| MIC-1 | Healthy Control | 89 | 0.1 | 0.8 | 0.3 | 0.3 | 0.2 |
| Chronic Pancreatitis | 68 | 0.2 | 3.4 | 0.9 | 1.0 | 0.9 | |
| IPMN | 63 | 0.3 | 3.0 | 0.7 | 0.8 | 0.6 | |
| PDAC early stage | 95* | 0.4 | 7.9 | 1.1 | 1.5 | 1.0 | |
| PDAC late stage | 92 | 0.2 | 7.1 | 1.0 | 1.3 | 0.9 | |
| SPON1 | Healthy Control | 89 | 1.9 | 14.9 | 4.7 | 5.0 | 2.0 |
| Chronic Pancreatitis | 68 | 1.0 | 17.5 | 5.9 | 6.3 | 3.2 | |
| IPMN | 63 | 0.6 | 21.8 | 5.2 | 5.9 | 2.7 | |
| PDAC early stage | 96 | 2.0 | 42.8 | 5.8 | 7.3 | 3.4 | |
| PDAC late stage | 92 | 2.1 | 21.1 | 5.9 | 6.5 | 3.2 | |
| HSP27 | Healthy Control | 89 | 0.2 | 4.2 | 0.9 | 1.2 | 0.9 |
| Chronic Pancreatitis | 68 | 0.2 | 4.8 | 1.0 | 1.2 | 0.7 | |
| IPMN | 63 | 0.1 | 8.6 | 0.9 | 1.3 | 1.0 | |
| PDAC early stage | 96 | 0.2 | 7.1 | 1.2 | 1.5 | 1.5 | |
| PDAC late stage | 92 | 0.2 | 5.2 | 1.2 | 1.3 | 1.1 | |
| CA19–9 | Healthy Control | 89 | <0.1 | 71.6 | 11.0 | 15.6 | 13.5 |
| Chronic Pancreatitis | 68 | <0.1 | 203.2 | 20.1 | 32.2 | 32.0 | |
| IPMN | 63 | <0.1 | 386. | 16.8 | 26.9 | 18.8 | |
| PDAC early stage | 96 | <0.1 | 27027.8 | 85.3 | 827.5 | 583.2 | |
| PDAC late stage | 92 | <0.1 | 25110.7 | 354.8 | 1638.7 | 1465.9 |
NOTE: all biomarkers are at ng/mL, except CA19–9 at U/mL. IQR, interquartile range.
, 2 missed data due to Out of Range.
Figure 1. Analysis of biomarkers in sera from PDAC patients, benign conditions, and healthy controls.
A-G, Concentrations of OPN, MIA, CEACAM-1, MIC-1, SPON1, HSP27 and CA19–9 in PDAC patients, benign conditions, and healthy controls. Only serum levels of biomarkers demonstrating significant differences between pancreatitis, IPMN, and PDAC early stage, (or benign and PDAC) are asterisked (Mann-Whitney U test). Bars indicate median value. *, p<0.05; **, p<0.01; ***, p<0.001; ****, p<0.0001.
Figure 2. Diagnostic performance of individual or combination of serum biomarkers in detection of early stage PDAC.
Diagnostic performance of CA19–9, OPN, MIA, CEACAM-1, MIC-1, SPON1 & HSP27 as individual markers (A, C&E) and their complementary (B, D&F) in differentiating patients with IPMN versus pancreatitis (A&B) or PDAC early stage versus pancreatitis (C&D) or PDAC early stage versus IPMN (E&F). ROC curves with AUCs are presented along with their 95% CI in brackets. Logistic regression modeling was constructed including age as a covariate and backward stepwise selected z-score transformed variables with the highest performance. The two-marker panel of OPN & MIA with an AUC=0.81 (0.74–0.88) for pancreatitis versus IPMN, CA19–9 & MIA with an AUC=0.86 (0.80–0.91) for pancreatitis versus PDAC early stage, and CA19–9 & MIC-1 with an AUC=0.81 (0.74–0.87) for IPMN versus PDAC early stage significantly improved the individual biomarker performance (p value: 0.0018, 0.0314, or 0.0208 for CA19–9; Delong test).
Discussion
In this study, a 6-plex immunoassay of OPN, MIA, CEACAM-1, MIC-1, SPON1 and HSP27 was in-house developed with high analytical performance appropriate for biomarker validation studies. It was applied to a set of serum samples of PDAC patients, those with benign pancreatic conditions, and healthy controls to evaluate the performance of 6 biomarkers individually and in combination for their ability to complement CA19–9 for the early detection of pancreatic cancer. The assay was characterized by LOB/LLOQ, cross-reactivity, recovery, intra- and inter-assay precision; and demonstrated wide dynamic ranges for the target protein measurements that significantly correlated with their respective monoplex assays and/or commercial ELISAs. The multiplex assay measures 6 candidate proteins using 12.5 μL of neat serum, and could include more candidate proteins with available pairs of capture and detection antibodies.
In this study, we identifed two-marker panels of CA19–9 and MIA or CA19–9 and MIC-1, both showed strong diagnostic performance and significant improvement over the use of CA19–9 alone in detecting early stage PDAC from chronic pancreatitis or IPMN. We also demonstrated that three-marker panels of CA19–9, MIC-1 and OPN or CA19–9, CEACAM-1 and MIA significantly improved upon the individual biomarker performances in the detection of PDAC from healthy controls or benign conditions. MIA is a small secreted protein coded by a single copy gene on chromosome 19q13.31-q13.33 and acts as an autocrine growth factor. MIA is strongly expressed by malignant melanoma cells and interacts with extracellular matrix proteins. Its overexpreesion promotes the metastatic behaviour of malignant melanoma, thus making it a potential prognostic marker in malignant melanoma (22). MIA was found to be overexpressed in pancreatic cancer and has the potential to promote the invasiveness of pancreatic cancer cells, but its serum levels were not significantly different between healthy donors and pancreatic cancer patients (23). MIC-1 belongs to the transforming growth factor-ß superfamily, originally identified in activated macrophages, and is found to be overexpressed in several cancer types (24). MIC-1 may have anticancer functions as its promoter region is a target for p53. Koopmann et al reported that serum MIC-1 outperformed CA19–9 in the differention of patients with resectable pancreatic cancer from healthy controls with an AUC=0.99 (MIC-1) versue 0.78 (CA19–9) but not from chronic pancreatitis (0.81 versue 0.74) (11). OPN is a glycophosphoprotein normally produced and secreted into most body fluids by osteoblasts, arterial smooth muscle cells, various epithelia, activated T cells and macrophages, and often found to be overexpressed in different cancer types (24). OPN is most likely related to tumorigenesis, cancer cell proliferation and progression, migration and invasion, protection from apoptosis, and enhancement of metastatic ability. Koopmann et al reported that serum OPN outperformed CA19–9 in the differention of patients with resectable pancreatic cancer from healthy controls with a sensitivity of 80% and a specificity of 97% (OPN) versus a sensitivity of 62% (CA19–9) (10). CEACAM-1 is a member of of the human carcinoembryonic antigen (CEA) family. The CEACAM subgroup members belong to the immunoglobulin superfamily of adhesion molecules. CEACAM1 is expressed in a number of epithelia, granulocytes, and lymphocytes, and the expression of CEACAM-1 was also reported in different cancer types (24). CEACAM-1 plays an important role in the regulation of tumor growth, angiogenesis, and immune modulation. Simeone et al reported that the combination of CEACAM-1 and CA19–9 had a significantly higher diagnostic accuracy than CA19–9 alone (12,25). Our study provides additional validation of the utilities of these serum biomarkers in early detection of PDAC (9–12).
Brand et al (18) reported that the panel of CA19–9, intercellular adhesion molecule 1 (ICAM-1) and osteoprotegerin (OPG) offered 78% of sensitivity (SN) at 94.1% specificity (SP) and an AUC of 0.91 in ROC curve analysis for the discrimination of PDAC versue healthy individuals providing the correct classification of 70.7% of stage I/II cancers. This compared to a 51.4% SN at 90% SP (AUC=0.82) with the correct classification of 40% of stage I/II cancers for CA19–9 alone. Comparing PDAC versus benign disease, a panel of CA19–9, CEA and TIMP-1 demonstrated an improvement over CA19–9 alone in terms of AUC (0.83 vs 0.78) and SN/SP (71.2%/88.6% vs 52.1%/90.2%) (18). Chan et al (19) showed that a biomarker panel consisting of CA19–9, cancer antigen 125 (CA-125) and laminin gamma 2 (LAMC2) significantly improved the performance of CA19–9 alone in discriminating early stage PDAC from benign conditions or chronic pancreatitis (AUC: 0.76 vs 0.69 or 0.74 vs 0.59). Our study provides additional evidence that combinations of individual serum biomarkers can offer superior diagnostic ability in early detection of pancreatic cancer.
Interestingly, our study showed that OPN alone or in combination with MIA outperforms CA19–9 in separating patients with IPMN from chronic pancreatitis with an AUC=0.80 (OPN; p=0.0042), or 0.81 (OPN and MIA; p=0.0018) versus 0.70 (CA19–9). Aprroximately 25% of the pancreatic neoplasms resected surgically and 50% of pancreatic cysts detected incidentally are IPMNs, which are major precursors of pancreatic cancer (26,27). It was observed in a prospective study that patients with pancreatic cysts had a remarkably increased (22.5-fold greater than expected) rate of developing pancreatic cancer (28,29). CEA and CA19–9 are the two most commonly used biomarkers for the diagnosis and follow-up of IPMN, however their sensitivities are too low to be used as a screening method, especially in high-risk patients (30). Novel serum biomarkers of precursor lesions are needed to devise timely therapies. Our study therefore provides new insight into the differentiation of IPMN from chronic pancreatitis.
In this study, we identified and validated the biomarker panels consisting of CA19–9 and MIC-1 or MIA that were better at detecting PDAC patients than CA19–9 alone, most notably at early disease stages. This study serves as a cross-validation of our previous studies (9,11) in a large, independent sample cohort in a multiplex immunoassay format. Our study is also in agreement with the reports from other research groups (31–34), including two recently published meta-analyses consisting of a total of 1235 (35) or 1698 (36) PDAC patients, demonstrating that serum MIC-1 may be a useful diagnostic biomarker with high sensitivity and specificity for identifying PDAC patients. The diagnostic performance of the biomarkers may be enhanced using a combination of CA19–9 and MIC-1 (9,11,31–34), considering MIC-1 had a sensitivity of 63.1% in detecting patients with CA19–9-negative PDAC (32). Notably, serum MIC-1 levels were significantly decreased in patients with PDAC after curative resection and returned to elevated levels when tumor relapse occurred (32), indicating the potential ability to complement CA19–9 in prognosis or therapeutic PDAC monitoring. Recently, there has been increased attention in the detection of tumor-specific molecular alterations by high-throughput screening – ‘omic’ technologies (37). There are a large number of promising biomarkers, including various tumor and serum proteins, microRNAs, as well as genetic markers that may be combined as diagnostic or prognostic indices (34). Combining circulating tumor DNA and protein biomarker-based liquid biopsies recently demonstrated an increased sensitivity of 64% in a blood test for early stage PDAC (38).
The selection of optimal panels through step-wise multivariate logistic regression allowed us to identify markers that are complementary in detecting specific conditions. Evidence for such complementary values were strengthened by comparison to null hypothesis performances generated from label permutation analysis. However, for such panels to be used as an in vitro diagnostic multivariate index assay (IVDMIA), additional development work and large-scale multi-site independent validation studies will be required (39–41).
In summary, a magnetic bead-based multiplex immunoassay was developed demonstrating sufficient analytical performance to evaluate serum biomarkers that may complement CA19–9 for early detection of PDAC. The biomarker panels identified in this study warrant additional clinical validation to determine their role in early detection of pancreatic cancer, which could lead to earlier intervention and better outcomes.
Supplementary Material
Acknowledgments:
This work was supported by funding from the NIH/NCI Early Detection Research Network, U24 CA115102 and U01 CA200469.
Abbreviations:
- CA19–9
cancer antigen 19–9
- OPN
osteopontin
- CEACAM-1
carcinoembryonic antigen-related cell adhesion molecule 1
- MIC-1
macrophage inhibitory cytokine-1
- MIA
melanoma inhibitory activity
- SPON1
spondin-1
- HSP27
heat shock protein 27
Footnotes
Disclosure: The authors declare no potential conflicts of interest.
References
- 1.Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin 2013;63(1):11–30 doi 10.3322/caac.21166. [DOI] [PubMed] [Google Scholar]
- 2.Michaud DS. Epidemiology of pancreatic cancer. Minerva Chir 2004;59(2):99–111. [PubMed] [Google Scholar]
- 3.Vincent A, Herman J, Schulick R, Hruban RH, Goggins M. Pancreatic cancer. Lancet 2011;378(9791):607–20 doi 10.1016/S0140-6736(10)62307-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Brand RE, Matamoros A. Imaging techniques in the evaluation of adenocarcinoma of the pancreas. Dig Dis 1998;16(4):242–52. [DOI] [PubMed] [Google Scholar]
- 5.Hidalgo M Pancreatic cancer. N Engl J Med 2010;362(17):1605–17 doi 10.1056/NEJMra0901557. [DOI] [PubMed] [Google Scholar]
- 6.Goonetilleke KS, Siriwardena AK. Systematic review of carbohydrate antigen (CA 19–9) as a biochemical marker in the diagnosis of pancreatic cancer. Eur J Surg Oncol 2007;33(3):266–70 doi 10.1016/j.ejso.2006.10.004. [DOI] [PubMed] [Google Scholar]
- 7.Duffy MJ, Sturgeon C, Lamerz R, Haglund C, Holubec VL, Klapdor R, et al. Tumor markers in pancreatic cancer: a European Group on Tumor Markers (EGTM) status report. Ann Oncol 2010;21(3):441–7 doi 10.1093/annonc/mdp332. [DOI] [PubMed] [Google Scholar]
- 8.Locker GY, Hamilton S, Harris J, Jessup JM, Kemeny N, Macdonald JS, et al. ASCO 2006 update of recommendations for the use of tumor markers in gastrointestinal cancer. J Clin Oncol 2006;24(33):5313–27 doi 10.1200/JCO.2006.08.2644. [DOI] [PubMed] [Google Scholar]
- 9.Koopmann J, Buckhaults P, Brown DA, Zahurak ML, Sato N, Fukushima N, et al. Serum macrophage inhibitory cytokine 1 as a marker of pancreatic and other periampullary cancers. Clin Cancer Res 2004;10(7):2386–92. [DOI] [PubMed] [Google Scholar]
- 10.Koopmann J, Fedarko NS, Jain A, Maitra A, Iacobuzio-Donahue C, Rahman A, et al. Evaluation of osteopontin as biomarker for pancreatic adenocarcinoma. Cancer Epidemiol Biomarkers Prev 2004;13(3):487–91. [PubMed] [Google Scholar]
- 11.Koopmann J, Rosenzweig CN, Zhang Z, Canto MI, Brown DA, Hunter M, et al. Serum markers in patients with resectable pancreatic adenocarcinoma: macrophage inhibitory cytokine 1 versus CA19–9. Clin Cancer Res 2006;12(2):442–6 doi 10.1158/1078-0432.CCR-05-0564. [DOI] [PubMed] [Google Scholar]
- 12.Simeone DM, Ji B, Banerjee M, Arumugam T, Li D, Anderson MA, et al. CEACAM1, a novel serum biomarker for pancreatic cancer. Pancreas 2007;34(4):436–43 doi 10.1097/MPA.0b013e3180333ae3. [DOI] [PubMed] [Google Scholar]
- 13.Takayama R, Nakagawa H, Sawaki A, Mizuno N, Kawai H, Tajika M, et al. Serum tumor antigen REG4 as a diagnostic biomarker in pancreatic ductal adenocarcinoma. J Gastroenterol 2010;45(1):52–9 doi 10.1007/s00535-009-0114-y. [DOI] [PubMed] [Google Scholar]
- 14.Poruk KE, Firpo MA, Scaife CL, Adler DG, Emerson LL, Boucher KM, et al. Serum osteopontin and tissue inhibitor of metalloproteinase 1 as diagnostic and prognostic biomarkers for pancreatic adenocarcinoma. Pancreas 2013;42(2):193–7 doi 10.1097/MPA.0b013e31825e354d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Pasanen PA, Eskelinen M, Partanen K, Pikkarainen P, Penttila I, Alhava E. Diagnostic value of tissue polypeptide specific antigen in patients with pancreatic carcinoma. Tumour Biol 1994;15(1):52–60. [DOI] [PubMed] [Google Scholar]
- 16.Zhou W, Sokoll LJ, Bruzek DJ, Zhang L, Velculescu VE, Goldin SB, et al. Identifying markers for pancreatic cancer by gene expression analysis. Cancer Epidemiol Biomarkers Prev 1998;7(2):109–12. [PubMed] [Google Scholar]
- 17.Slesak B, Harlozinska-Szmyrka A, Knast W, Sedlaczek P, van Dalen A, Einarsson R. Tissue polypeptide specific antigen (TPS), a marker for differentiation between pancreatic carcinoma and chronic pancreatitis. A comparative study with CA 19–9. Cancer 2000;89(1):83–8. [DOI] [PubMed] [Google Scholar]
- 18.Brand RE, Nolen BM, Zeh HJ, Allen PJ, Eloubeidi MA, Goldberg M, et al. Serum biomarker panels for the detection of pancreatic cancer. Clin Cancer Res 2011;17(4):805–16 doi 10.1158/1078-0432.CCR-10-0248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Chan A, Prassas I, Dimitromanolakis A, Brand RE, Serra S, Diamandis EP, et al. Validation of biomarkers that complement CA19.9 in detecting early pancreatic cancer. Clin Cancer Res 2014;20(22):5787–95 doi 10.1158/1078-0432.CCR-14-0289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Fu Q, Zhu J, Van Eyk JE. Comparison of multiplex immunoassay platforms. Clin Chem 2010;56(2):314–8 doi 10.1373/clinchem.2009.135087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Houser B Bio-Rad’s Bio-Plex(R) suspension array system, xMAP technology overview. Arch Physiol Biochem 2012;118(4):192–6 doi 10.3109/13813455.2012.705301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bosserhoff AK, Kaufmann M, Kaluza B, Bartke I, Zirngibl H, Hein R, et al. Melanoma-inhibiting activity, a novel serum marker for progression of malignant melanoma. Cancer Res 1997;57(15):3149–53. [PubMed] [Google Scholar]
- 23.El Fitori J, Kleeff J, Giese NA, Guweidhi A, Bosserhoff AK, Buchler MW, et al. Melanoma Inhibitory Activity (MIA) increases the invasiveness of pancreatic cancer cells. Cancer Cell Int 2005;5(1):3 doi 10.1186/1475-2867-5-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ballehaninna UK, Chamberlain RS. Biomarkers for pancreatic cancer: promising new markers and options beyond CA 19–9. Tumour Biol 2013;34(6):3279–92 doi 10.1007/s13277-013-1033-3. [DOI] [PubMed] [Google Scholar]
- 25.Bunger S, Laubert T, Roblick UJ, Habermann JK. Serum biomarkers for improved diagnostic of pancreatic cancer: a current overview. J Cancer Res Clin Oncol 2011;137(3):375–89 doi 10.1007/s00432-010-0965-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Andrejevic-Blant S, Kosmahl M, Sipos B, Kloppel G. Pancreatic intraductal papillary-mucinous neoplasms: a new and evolving entity. Virchows Arch 2007;451(5):863–9 doi 10.1007/s00428-007-0512-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kosmahl M, Pauser U, Peters K, Sipos B, Luttges J, Kremer B, et al. Cystic neoplasms of the pancreas and tumor-like lesions with cystic features: a review of 418 cases and a classification proposal. Virchows Arch 2004;445(2):168–78 doi 10.1007/s00428-004-1043-z. [DOI] [PubMed] [Google Scholar]
- 28.Hruban RH, Maitra A, Kern SE, Goggins M. Precursors to pancreatic cancer. Gastroenterol Clin North Am 2007;36(4):831–49, vi doi 10.1016/j.gtc.2007.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Tada M, Kawabe T, Arizumi M, Togawa O, Matsubara S, Yamamoto N, et al. Pancreatic cancer in patients with pancreatic cystic lesions: a prospective study in 197 patients. Clin Gastroenterol Hepatol 2006;4(10):1265–70 doi 10.1016/j.cgh.2006.07.013. [DOI] [PubMed] [Google Scholar]
- 30.Moris D, Damaskos C, Spartalis E, Papalampros A, Vernadakis S, Dimitroulis D, et al. Updates and Critical Evaluation on Novel Biomarkers for the Malignant Progression of Intraductal Papillary Mucinous Neoplasms of the Pancreas. Anticancer Res 2017;37(5):2185–94 doi 10.21873/anticanres.11553. [DOI] [PubMed] [Google Scholar]
- 31.Mohamed AA, Soliman H, Ismail M, Ziada D, Farid TM, Aref AM, et al. Evaluation of circulating ADH and MIC-1 as diagnostic markers in Egyptian patients with pancreatic cancer. Pancreatology 2015;15(1):34–9 doi 10.1016/j.pan.2014.10.008. [DOI] [PubMed] [Google Scholar]
- 32.Wang X, Li Y, Tian H, Qi J, Li M, Fu C, et al. Macrophage inhibitory cytokine 1 (MIC-1/GDF15) as a novel diagnostic serum biomarker in pancreatic ductal adenocarcinoma. BMC Cancer 2014;14:578 doi 10.1186/1471-2407-14-578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kaur S, Chakraborty S, Baine MJ, Mallya K, Smith LM, Sasson A, et al. Potentials of plasma NGAL and MIC-1 as biomarker(s) in the diagnosis of lethal pancreatic cancer. PLoS One 2013;8(2):e55171 doi 10.1371/journal.pone.0055171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Loosen SH, Neumann UP, Trautwein C, Roderburg C, Luedde T. Current and future biomarkers for pancreatic adenocarcinoma. Tumour Biol 2017;39(6):1010428317692231 doi 10.1177/1010428317692231. [DOI] [PubMed] [Google Scholar]
- 35.Chen YZ, Liu D, Zhao YX, Wang HT, Gao Y, Chen Y. Diagnostic performance of serum macrophage inhibitory cytokine-1 in pancreatic cancer: a meta-analysis and meta-regression analysis. DNA Cell Biol 2014;33(6):370–7 doi 10.1089/dna.2013.2237. [DOI] [PubMed] [Google Scholar]
- 36.Yang Y, Yan S, Tian H, Bao Y. Macrophage inhibitory cytokine-1 versus carbohydrate antigen 19–9 as a biomarker for diagnosis of pancreatic cancer: A PRISMA-compliant meta-analysis of diagnostic accuracy studies. Medicine (Baltimore) 2018;97(9):e9994 doi 10.1097/MD.0000000000009994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Boulaiz H, Ramos MC, Grinan-Lison C, Garcia-Rubino ME, Vicente F, Marchal JA. What’s new in the diagnosis of pancreatic cancer: a patent review (2011-present). Expert Opin Ther Pat 2017;27(12):1319–28 doi 10.1080/13543776.2017.1379991. [DOI] [PubMed] [Google Scholar]
- 38.Cohen JD, Javed AA, Thoburn C, Wong F, Tie J, Gibbs P, et al. Combined circulating tumor DNA and protein biomarker-based liquid biopsy for the earlier detection of pancreatic cancers. Proc Natl Acad Sci U S A 2017;114(38):10202–7 doi 10.1073/pnas.1704961114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Bristow RE, Smith A, Zhang Z, Chan DW, Crutcher G, Fung ET, et al. Ovarian malignancy risk stratification of the adnexal mass using a multivariate index assay. Gynecol Oncol 2013;128(2):252–9 doi 10.1016/j.ygyno.2012.11.022. [DOI] [PubMed] [Google Scholar]
- 40.Zhang Z An In Vitro Diagnostic Multivariate Index Assay (IVDMIA) for Ovarian Cancer: Harvesting the Power of Multiple Biomarkers. Rev Obstet Gynecol 2012;5(1):35–41. [PMC free article] [PubMed] [Google Scholar]
- 41.Zhang Z, Chan DW. The road from discovery to clinical diagnostics: lessons learned from the first FDA-cleared in vitro diagnostic multivariate index assay of proteomic biomarkers. Cancer Epidemiol Biomarkers Prev 2010;19(12):2995–9 doi 10.1158/1055-9965.EPI-10-0580. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.