Abstract
Background
Since lifestyle changes are main therapies for non-alcoholic fatty liver disease (NAFLD), changing dietary components (nutritional or bioactive) may play a parallel important role. Few studies have assessed the effects of curcumin on NAFLD (mainly antioxidant and anti-inflammatory effects). We aimed to determine the effects of nano-curcumin (NC) on overweight/obese NAFLD patients by assessing glucose, lipids, inflammation, insulin resistance, and liver function indices, especially through nesfatin.
Methods
This double-blind, randomized, placebo-controlled clinical trial was conducted in the Oil Company Central Hospital, Tehran. 84 overweight/obese patients with NAFLD diagnosed using ultrasonography were recruited according to the eligibility criteria (age 25–50 yrs., body mass index [BMI] 25–35 kg/m2). The patients were randomly divided into two equal NC (n = 42) and placebo (n = 42) groups. Interventions were two 40 mg capsules/day after meals for 3 months. Lifestyle changes were advised. A general questionnaire, a 24-h food recall (at the beginning, middle and end), and the short-form international physical activity questionnaire (at the beginning and end) were completed. Also, blood pressure, fatty liver degree, anthropometrics, fasting blood sugar (FBS) and insulin (FBI), glycated hemoglobin (HbA1c), homeostasis model assessment-insulin resistance (HOMA-IR), quantitative insulin sensitivity check index (QUICKI), total cholesterol (TC), triglyceride (TG), low-density lipoprotein cholesterol (LDL-c), high-density lipoprotein cholesterol (HDL-c), tumor necrosis factor-alpha (TNF-α), high sensitive c-reactive protein (hs-CRP), interleukin-6 (IL-6), liver transaminases, and nesfatin were determined at the beginning and end.
Results
NC compared with placebo significantly increased HDL, QUICKI, and nesfatin and decreased fatty liver degree, liver transaminases, waist circumference (WC), FBS, FBI, HbA1c, TG, TC, LDL, HOMA-IR, TNF-α, hs-CRP, and IL-6 (P < 0.05). The mean changes in weight, BMI, body composition (BC), and blood pressure were not significant (P > 0.05). After adjustment for confounders, the changes were similar to the unadjusted model.
Conclusion
NC supplementation in overweight/obese NAFLD patients improved glucose indices, lipids, inflammation, WC, nesfatin, liver transaminases, and fatty liver degree. Accordingly, the proposed mechanism for ameliorating NAFLD with NC was approved by the increased serum nesfatin and likely consequent improvements in inflammation, lipids, and glucose profile. Further trials of nano-curcumin’s effects are suggested.
Trial registration
Iranian Registry of Clinical Trials, IRCT2016071915536N3. Registered 2016-08-02.
Keywords: Non-alcoholic fatty liver disease, Nano-curcumin, Obesity, Overweight, Iran
Introduction
Non-alcoholic fatty liver disease (NAFLD) is the deposition of triglycerides (TG) in hepatocytes over than 5% of liver weight/volume and has three grades according to the liver biopsy (mild: < 33%, moderate: 33–66%, severe: > 66%). Usually, non-invasive diagnostic methods such as ultrasound, CT scan, and MRI are widely utilized that exact differentiation between the stages is difficult. Also, the liver enzymes (alanine transaminase [ALT] and aspartate transaminase [AST]) may be elevated 1.5–2 times above normal levels. However, many individuals with advanced non-alcoholic steatohepatitis (NASH) and even cirrhosis have normal liver enzyme levels. NAFLD rates are therefore likely higher than reported. Symptoms frequently include fatigue and upper-right quarter abdominal discomfort. The average adult prevalence is roughly 30% (65–85% and 15–20% in obese [BMI ≥ 25] and non-obese [BMI < 25] patients, respectively). It is more prevalent in males [1–4].
Two phases of pathology of NAFLD are fat deposition with hepatic steatosis and NASH. Insulin resistance plays a key role in both phases and common irritants include oxidative stress and inflammation. Liver fat content directly correlates with insulin resistance. Activating nuclear factor kappa B (NF-κB) upregulates pro-inflammatory cytokines, which can affect insulin activity. Inflammation, adipokines, oxidative stress or lipid metabolites can all, therefore, adjust insulin sensitivity, even though the intrahepatic fat content may not be necessarily directly related to these factors. The other risk factors include increased blood insulin levels, central obesity, type 2 diabetes, certain medications, nutrition status (starvation, protein or choline deficiency), some diseases, jejunum bypass, age, family history, malnutrition, severe weight loss, and gastrointestinal tract infections. Also, NAFLD incidence may correlate to the high saturated fat and/or carbohydrate intake. Other patients can display normal weight despite having abdominal obesity and insulin resistance [1–4].
Nesfatin as a neuropeptide of the hypothalamus is involved in appetite regulation and body fat storage by important functions in metabolizing glucose, phosphorylating specific signaling proteins, and increasing liver insulin sensitivity, specifically by AMP-activated protein kinase. Its gene is expressed in some locations including the brain, pancreas, stomach endocrine cells, and adipocytes. Nesfatin gene expression is activated by peroxisome proliferator-activated receptors (PPARs), especially PPARγ [5, 6]. Recently, serum nesfatin levels in overweight/obese NAFLD patients aged 30–60 years was significantly lower than in healthy individuals [1, 2, 6].
The common prescription for NAFLD involves lifestyle changes (weight loss and physical activity). Since weight loss and long-term maintenance is not always an easy task, the dietary modifications can be seen as a therapeutic approach for such patients. Accordingly, it is needed to investigate the effects of certain nutrients and/or dietary ingredients on NAFLD [1–4].
Curcumin as the effective ingredient in turmeric (ginger family) expresses multiple effects including antioxidant, anti-inflammatory, antimicrobial, and anti-carcinogenic ones. It raises both PPARγ activity and expression, which are important for inhibiting inflammation and oxidative stress as major factors involved in insulin resistance and NAFLD [1, 2].
The prevalence and implications of NAFLD are rising. Since there are no medications to combat it and the role of nutrition is a key treatment factor; by examining dietary components such as curcumin for NAFLD improvement, researchers can begin to uncover new treatments. Curcumin is involved in a number of metabolic functions in improving insulin resistance. Despite multiple health benefits, its low stability and bioavailability impact its therapeutic efficiency. Various techniques have been explored recently to improve this problem by using of polymeric nanoparticles called Nano-curcumin (NC) (for example, polylactic-co-glycolic acid (PLGA) nanoparticles can increase curcumin bioavailability 22-fold) [1, 2]. In this trial, NC was used, therefore. The onset and progression of NAFLD may be improved by curcumin through inhibiting inflammation and oxidative stress. The severity of NAFLD is increased by overweight or obesity; however, no human studies have been carried out to determine how these are affected by curcumin. This study, therefore, was designed to examine effects of curcumin on blood glucose indices, lipids, inflammatory profiles, liver function (fatty liver degree, ALT, AST), and insulin resistance (HOMA-IR, QUICKI), especially by looking at serum nesfatin in obese NAFLD patients.
Materials and methods
study design and participants
The ethics committee of Tehran University of Medical Sciences approved this double-blind randomized placebo-controlled clinical trial as IR.TUMS.REC.1394.791. The study was registered to the Iranian Registry of Clinical Trials as IRCT2016071915536N3 on 02/08/2016. The participants were overweight or obese NAFLD patients referred to the sonography department of the NIOC Central Hospital in Tehran.
Inclusion criteria included NAFLD according to ultrasonography, aged 25–50, and 25 ≤ BMI < 35 kg/m2. Exclusion criteria included alcohol intake during the previous year, inability/unwillingness to cooperate, other liver conditions, secondary NAFLD, uncontrolled hypertension (> 140/90 mmHg), pregnancy or lactation, professional athlete status, use of statins, ursodeoxycholic acid, probiotics, antihypertensive, curcumin-interactive drugs, consumption of multivitamin/mineral and antioxidant supplements over the previous 3 months, weight loss over the previous 3 months, and not taking more than 10% of the intervention supplement [1–4, 7–9].
Randomization and intervention
Subjects were divided into two equal groups by the block randomization method, carried out by an assistant (NC [n = 42] or placebo [n = 42]). Stratified randomization was used to control for sex. The ratio of the two groups was 1:1. 3 patients from the NC group and 2 patients from the placebo group declined to participate (Fig. 1).
Fig. 1.
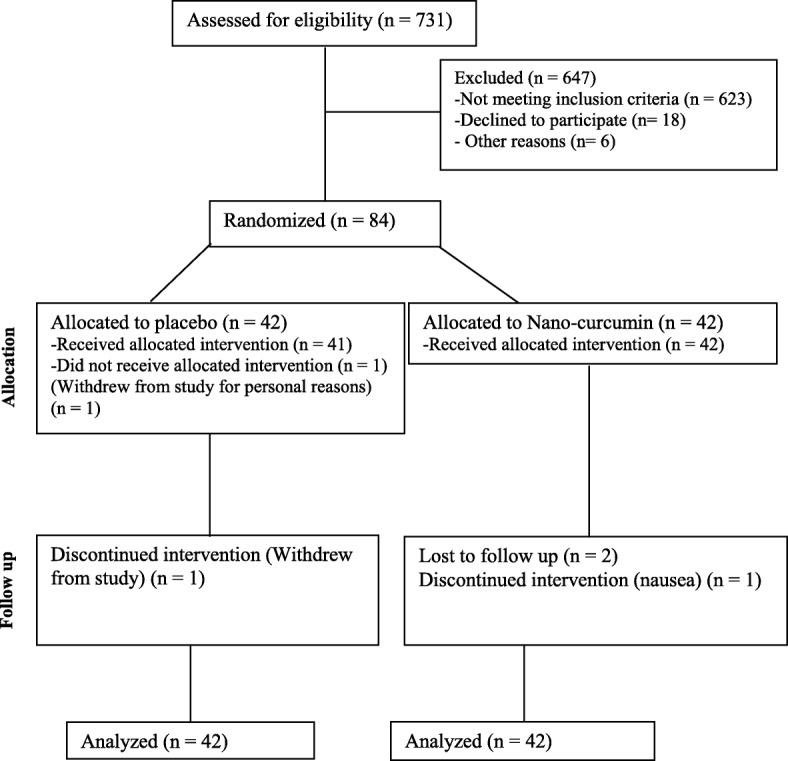
Flowchart of participants with overweight/obesity and NAFLD
Intervention allocation blinding was performed for both participants and investigators before the beginning, kept during the intervention, and opened after the data analysis by a field worker. The NC and placebo capsules were supplied by the Exir-Nano-Sina Company. The capsules were similar in shape, size, and color. The sinacurcumin® dose was 80 mg/day (two 40-mg capsules per day, according to the manufacturer’s instructions: one capsule at breakfast and another at dinner). The supplements were distributed monthly, and compliance status was assessed monthly by face-to-face consultation, and weekly by phone. The compliance percentage of supplements was calculated according to the mean percentage of the number of capsules consumed by the individuals of any groups. The lifestyle advice was equally presented by a trained dietician (SAJT) at the hospital.
Assessments and measurements
General characteristics, dietary intakes, and physical activity
The main investigator identified NAFLD patients, checked their eligibility criteria, clarified the study details, and obtained informed consent. Interviews were conducted to fill the questionnaires, including a general questionnaire, a 24-h food recall (at beginning, middle, and end), and the short-form IPAQ (SF-IPAQ) (at beginning and end). At the commencement of the trial, lifestyle advice [10] was presented by a qualified dietician, including adhering to a low-calorie diet aimed at achieving weight loss of about 0.5–1 kg/week, and increasing physical activity by undertaking moderate-intensity aerobic exercise at least 3 times/week for 30–45 min [11].
Subjects’ dietary status was determined by Nutritionist-4 software, using gram/day values from the 24-h food recall (validated in Iran [12]) [12, 13].
The IPAQ short-form questionnaire has 7 stratified questions that determine one of three overall activity levels. This questionnaire has previously been validated in Iran [14, 15].
Anthropometric measurements
Weight (at beginning and end) and height (at the beginning) were determined using a digital scale and stadiometer (Seca® Germany, Model: 7551021994). BMI was calculated by dividing weight in kilograms by height in meters squared. Body composition percentages, including body fat and lean body mass, were measured using the Bioimpedance Analyzer device (Tanita®). At the beginning and end of the study, systolic and diastolic blood pressures were obtained with the use of a mercury manometer (Riester®) and were reported in mmHg. Waist circumference was measured at the middle of the last rib and at the iliac crest with minimal clothing, using a non-elastic tape, at the beginning and end of the trial. Blood sampling, storage, and laboratory tests were carried out at the NIOC Central Hospital, Tehran.
Sonography and blood biomarkers measurements
The ultrasound test was performed by a radiologist, using a sonography device (General Electric (GE)®, Model: Voluson E8) after a 12-h fast, to reduce potential human error.
10 ml of blood (at the beginning and end) was taken from the peripheral vein following a 12-h overnight fast and was centrifuged for 20 min (3000 g). Serum glucose indices, lipids, and liver transaminases were assessed on the day of the blood sample. The remaining serum was frozen and stored at − 80 °C before analysis.
Serum nesfatin, IL-6, and TNF-α were measured using sandwich ELISA and the following kits: Shanghai Crystal Day Biotech Co. Ltd®; Intra-assay CV < 8%, Inter-assay CV < 10% with an automatic device (Elisys Uno Human®). The ELISA kit for FBI and hs-CRP were DiaMetra® Co. of Italy, DCM076–8; Intra-assay CV ≤ 5%, Inter-assay CV ≤ 10% and Diagnostics Biochem Canada (DBC) Inc®, REF: CAN-CRP-4360, Version 5.0; Intra-assay CV ≤ 15.2%, Inter-assay CV ≤ 9.9%, respectively. Serum glucose was determined with the glucose oxidase method, using a Hitachi analyzer device (q17®) and the specified kit Bionik®, Liquid Stable, Glucose oxidase GOD-POD, Mono-reagent; Intra-assay CV ≤ 2.10%, Inter-assay CV ≤ 3.09%. HOMA-IR and QUICKI indexes were calculated by the following formulas:
HOMA ‐ IR = FBI [μIU/ml] × FBS [mg/dl]/405.
Serum lipids (TC, TG, HDL-c, LDL-c), ALT, AST, and HbA1c were assessed with the Hitachi analyzer device (q17®) and specific kits (Table 1).
Table 1.
The used kits for determination of the serum lipids, liver enzymes, and glycosylated hemoglobin
| Factors | Kits |
|---|---|
| TC | Bionik®, Liquid Stable, Enzymatic Colorimetric CHOD-POD, Intra-assay CV ≤ 1.216%, Inter-assay CV ≤ 6.906% |
| HDL | Bionik®, Liquid Stable, Direct. Enzymatic Colorimetric, Intra-assay CV ≤ 0.7%, Inter-assay CV ≤ 1.5% |
| TG | Bionik®, Liquid Stable, GPO-POD, Mono-reagent, Intra-assay CV ≤ 1.573%, Inter-assay CV ≤ 7.704% |
| LDL | Bionik®, Liquid Stable, Direct. Enzymatic Colorimetric, Intra-assay CV ≤ 1.76%, Inter-assay CV ≤ 0.65% |
| AST | Bionik®, Liquid Stable, NADH. Kinetic UV.Liquid, Intra-assay CV ≤ 3.02%, Inter-assay CV ≤ 3.00% |
| ALT | Bionik®, Liquid Stable, NADH. Kinetic UV.IFCC, Intra-assay CV ≤ 4.27%, Inter-assay CV ≤ 4.68% |
| HbA1c | Bionik®, Liquid Reagents, Particle Enhanced Immuno-Assay [PEIA], Intra-assay CV ≤ 1.72%, Inter-assay CV ≤ 2.77% |
Sample size
According to Chuengsamarn et al. [16], the mean ± SD of HOMA-IR indices in the curcumin and placebo groups were 3.22 ± 1.30 and 4.08 ± 1.35, respectively. There were 42 participants in each sample group, with a CI of 95%, power of 80%, and a drop of 15%. 84 individuals in total were chosen and split into two equal groups (NC and placebo) using a block randomization method.
Data analysis and accessibility
Data management was performed, including entry, security, coding, and storage. The follow-up data missing from patients was estimated by the modified-intention to treat (m-ITT) analysis, and the regression imputation method. The Kolmogorov-Smirnov, Chi-square, Fisher Exact, and t or Mann-Whitney tests were implemented to assess the normality of continuous variables and categorical and continuous baseline characteristics, respectively. Two-way repeated measures analysis of variance (TWRM-ANOVA) was used to compute time effects and time by treatment interaction effects with all the dependent variables. Moreover, TWRM-ANOVA was adjusted for participants’ dietary intake of energy, total fat, saturated fat, monounsaturated fatty acid, vitamins D, B1, B6, and folate. A 95% confidence interval (CI) and a P-value of < 0.05 were used to report data. Data analysis was performed using SPSS16 (statistical package for the social sciences) and STATA11SE (general-purpose statistical software package by Stata Corp) software. The main researcher had full access to the finalized dataset and presented the results.
Results
Participant traits
As shown in Figs. 1, 731 people were screened based on their medical history. 108 subjects met the eligibility criteria, of whom 18 declined to take part, and 6 could not participate. 84 subjects were randomized into 2 groups and completed the first visit. At the follow-up stage, 5 subjects could not continue (for personal reasons and/or travel; NC n = 3; placebo n = 2). Eventually, data analysis was carried out on the 84 subjects, according to the modified-ITT analysis.
Participant general traits and physical activity levels are displayed in Table 2. The majority of the subjects had similar education levels and presented the high economic status and low physical activity scores. Both groups well consumed the prescribed supplements. The compliance percentage of supplements were 95.98% in NC group and 97.95% in placebo group.
Table 2.
General traits and physical activity levels of overweight/obese patients with non-alcoholic fatty liver disease (NAFLD)
| Variables | Nanocurcumin (n = 42) n(%) or Mean(SD) | Placebo (n = 42) n(%) or Mean(SD) | P-value | ||
|---|---|---|---|---|---|
| Age (yrs) | 41.8(5.6) | 42.5(6.2) | 0.2* | ||
| Gender | male | 23(54.8) | 23(54.8) | 1.0** | |
| female | 19(45.2) | 19(45.2) | |||
| Height (cm) | 167.8(9.8) | 167.7(9.0) | 0.8* | ||
| Marriage status | single | 5(11.9) | 7(16.7) | 0.5** | |
| married | 37(88.1) | 35(83.3) | |||
| Job status | employee, free job/retired | 31(73.8) | 25(59.5) | 0.1** | |
| housewife, unemployed | 11(26.2) | 17(40.5) | |||
| Education level | up to associate degree | 21(50) | 20(47.6) | 0.8** | |
| Bachelor and higher | 21(50) | 22(52.4) | |||
| Economic level | Low (≤3 living items) | 0(0) | 0(0) | 0.8** | |
| moderate (4–6 living items) | 19(45.2) | 18(42.9) | |||
| High (≥7 living items) | 23(54.8) | 24(57.1) | |||
| Physical activity level (Baseline) | low (< 600 MET-minutes/week) | 31(73.8) | 28(66.7) | 0.4** | |
| Moderate (600 to < 1500 MET-minutes/week) | 11(26.2) | 14(33.3) | |||
| High (≥ 1500 MET-minutes/week) | 0(0) | 0(0) | |||
| Physical activity level (After 3 months) | low (< 600 MET-minutes/week) | 27(64.3) | 26(61.9) | 0.8$ | |
| Moderate (600 to < 1500 MET-minutes/week) | 15(35.7) | 16(38.1) | |||
| High (≥ 1500 MET-minutes/week) | 0(0) | 0(0) | |||
| Fatty liver (Baseline) | No | 0(0) | 0(0) | 0.5** | |
| Yes | Mild | 35(83.3) | 37(88.1) | ||
| Moderate | 7(16.7) | 5(11.9) | |||
| Severe | 0(0) | 0(0) | |||
| Fatty liver (After 3 months) | No | 20(47.6) | 5(11.9) | < 0.001** | |
| Yes | Mild | 21(50.0) | 33(78.6) | ||
| Moderate | 1(2.4) | 4(9.5) | |||
| Severe | 0(0) | 0(0) | |||
*Mann-Whitney; **Chi-square; $Fisher exact test
Dietary vitamin D intake at baseline was greater in the placebo group, while other baseline features between the two groups were similar (Tables 2, 3, 4).
Table 3.
Comparison of baseline mean for weight, BMI, glucose indices, nesfatin, inflammatory factors, and liver enzymes in overweight/obese patients with non-alcoholic fatty liver disease (NAFLD)
| Variables | Nanocurcumin (n = 42) Mean(SD) | Placebo (n = 42) Mean(SD) | P-value |
|---|---|---|---|
| Weight (kg) | 86.54(10.98) | 86.70(11.15) | 0.9* |
| BMI (kg/m2) | 30.67(2.14) | 30.75(2.35) | 0.9** |
| Waist circumference (cm) | 105.4(6.2) | 103.8(6.7) | 0.8* |
| Fat Mass (%) | 31.6(6.4) | 31.9(4.7) | 0.8* |
| SBP (mmhg) | 120.3(4.7) | 120.7(4.3) | 0.7** |
| DBP (mmhg) | 78.8(4.5) | 79.8(4.3) | 0.1* |
| ALT (u/l) | 42.8(11.6) | 42.1(8.2) | 0.7** |
| AST (u/l) | 28.4(6.7) | 27.6(7.8) | 0.4** |
| TC (mg/dl) | 212.9(18.9) | 211.8(21.4) | 0.8* |
| LDL-c (mg/dl) | 135.6(17.6) | 133.0(20.7) | 0.6* |
| TG (mg/dl) | 175.9(70.3) | 181.2(65.6) | 0.7** |
| HDL-c (mg/dl) | 41.8(5.6) | 42.7(5.7) | 0.4* |
| FBS (mg/dl) | 89.1(5.4) | 89.3(5.8) | 0.9* |
| HbA1c (%) | 5.2(0.2) | 5.3(0.2) | 0.4** |
| Nesfatin (ng/ml) | 1.8(0.5) | 1.8(0.4) | 0.5* |
| FBI (μIU/ml) | 8.0(0.5) | 7.9(0.6) | 0.7* |
| TNF-α (ng/l) | 14.7(3.3) | 15.1(2.7) | 0.2* |
| IL-6 (ng/l) | 7.6(1.5) | 7.9(1.5) | 0.2* |
| hs-CRP (mg/l) | 5.9(2.5) | 5.3(2.5) | 0.2** |
| HOMA-IR (score) | 1.77(0.16) | 1.75(0.17) | 0.7** |
| QUICKI (score) | 0.3505(0.0049) | 0.3509(0.0057) | 0.7* |
*t-test; **Mann-Whitney; ALT alanine transaminase, AST aspartate transaminase, BMI body mass index, FBI fasting blood insulin,FBS fasting blood sugar, HOMA-IR homeostasis model assessment-insulin resistance, hs-CRP high-sensitive C-reactive protein, HDL-C high density lipoprotein-cholesterol, IL-6 interleukin-6, LDL-C low density lipoprotein-cholesterol, QUICKI quantitative insulin sensitivity check index, TC total cholesterol, TNF-α tumor necrosis factor-alpha
Table 4.
Dietary intakes of overweight/obese patients with non-alcoholic fatty liver disease (NAFLD)
| Dietary intakes | Nanocurcumin (n = 42) Mean(SD) | Placebo (n = 42) Mean(SD) | P-value | ||
|---|---|---|---|---|---|
| Energy (kcal) | Baseline | 2473.2(470.5) | 2338.6(496.3) | 0.2* | 0.001** |
| 1.5 Months | 2089.9(412.9) | 2267.0(468.4) | 0.07 | ||
| 3 Months | 2019.4(380.5) | 2205.9(476.0) | 0.05 | ||
| Protein (g) | Baseline | 100.7(30.7) | 97.1(32.7) | 0.6 | 0.3 |
| 1.5 Months | 93.7(24.9) | 93.8(31.8) | 0.8 | ||
| 3 Months | 84.1(28.7) | 92.9(31.4) | 0.2 | ||
| Protein (%) | Baseline | 16.3(3.7) | 16.5(4.1) | 0.7 | 0.2 |
| 1.5 Months | 18.0(3.8) | 16.4(3.9) | 0.06 | ||
| 3 Months | 16.6(4.8) | 16.7(4.1) | 0.8 | ||
| Carbohydrate (g) | Baseline | 296.1(55.6) | 280.5(81.9) | 0.3 | 0.3 |
| 1.5 Months | 253.8(68.9) | 252.3(58.9) | 0.9 | ||
| 3 Months | 256.2(47.8) | 266.0(73.3) | 0.3 | ||
| Carbohydrate (%) | Baseline | 48.5(7.7) % | 47.8(9.0) % | 0.7 | 0.5 |
| 1.5 Months | 48.4(8.7) % | 45.1(8.7) % | 0.08 | ||
| 3 Months | 51.2(7.4) % | 48.1(8.0) % | 0.07 | ||
| Fat total (g) | Baseline | 103.1(32.8) | 97.0(27.4) | 0.3 | 0.001 |
| 1.5 Months | 82.3(22.5) | 102.4(31.1) | 0.001 | ||
| 3 Months | 78.7(20.6) | 90.4(24.6) | 0.02 | ||
| Fat total (%) | Baseline | 37.00(6.9) % | 37.5(7.4) % | 0.7 | 0.1 |
| 1.5 Months | 35.5(6.7) % | 40.2(7.1) % | 0.003 | ||
| 3 Months | 34.8(4.9) % | 37.0(6.6) % | 0.09 | ||
| Cholesterol (mg) | Baseline | 268.2(168.8) | 282.3(178.7) | 0.7 | 0.3 |
| 1.5 Months | 273.9(150.8) | 271.3(153.1) | 0.9 | ||
| 3 Months | 198.3(126.8) | 263.9(125.6) | 0.01 | ||
| Saturated fat (g) | Baseline | 27.3(11.4) | 25.5(9.2) | 0.7 | 0.03 |
| 1.5 Months | 22.5(7.2) | 27.9(9.7) | 0.005 | ||
| 3 Months | 21.2(8.5) | 25.6(9.8) | 0.03 | ||
| Monounsaturated fatty acid (g) | Baseline | 37.8(13.7) | 36.1(9.8) | 0.5 | 0.008 |
| 1.5 Months | 30.4(8.9) | 36.7(11.6) | 0.006 | ||
| 3 Months | 28.5(8.2) | 33.7(9.0) | 0.008 | ||
| Polyunsaturated fatty acid (g) | Baseline | 26.4(13.6) | 25.0(11.6) | 0.5 | 0.06 |
| 1.5 Months | 20.3(7.6) | 26.6(15.3) | 0.1 | ||
| 3 Months | 20.2(6.8) | 22.1(7.1) | 0.4 | ||
| Vitamin A (RAE) (μg) | Baseline | 316.8(210.7) | 349.0(264.2) | 0.6 | 0.6 |
| 1.5 Months | 366.9(287.1) | 319.7(270.6) | 0.2 | ||
| 3 Months | 416.2(378.3) | 407.0(344.8) | 0.4 | ||
| Carotenoids (μg) | Baseline | 8009.3(6994.8) | 7580.6(6601.1) | 0.7 | 0.8 |
| 1.5 Months | 8527.0(6850.7) | 8068.6(6627.9) | 0.7 | ||
| 3 Months | 8184.9(6718.9) | 8705.7(7724.2) | 0.9 | ||
| Vitamin C (mg) | Baseline | 97.0(84.4) | 79.8(61.3) | 0.4 | 0.9 |
| 1.5 Months | 83.8(61.0) | 74.2(69.4) | 0.1 | ||
| 3 Months | 95.1(73.5) | 85.7(71.8) | 0.2 | ||
| Calcium (mg) | Baseline | 1086.0(502.2) | 1115.7(521.2) | 0.7 | 0.1 |
| 1.5 Months | 1025.8(360.1) | 910.2(473.1) | 0.03 | ||
| 3 Months | 897.8(408.9) | 1076.8(475.1) | 0.06 | ||
| Iron (mg) | Baseline | 15.0(3.5) | 14.8(5.0) | 0.7 | 0.3 |
| 1.5 Months | 14.1(4.0) | 13.7(3.1) | 0.6 | ||
| 3 Months | 13.1(3.1) | 14.1(4.0) | 0.1 | ||
| Vitamin D (μg) | Baseline | 1.0(1.8) | 1.9(2.1) | 0.01 | 0.02 |
| 1.5 Months | 2.4(3.6) | 1.1(1.6) | 0.03 | ||
| 3 Months | 2.1(3.5) | 1.4(1.6) | 0.9 | ||
| Vitamin E (mg) | Baseline | 29.7(19.0) | 28.7(13.1) | 0.7 | 0.5 |
| 1.5 Months | 24.9(9.2) | 27.0(14.6) | 0.4 | ||
| 3 Months | 23.7(8.6) | 26.9(8.2) | 0.1 | ||
| Vitamin B1 (mg) | Baseline | 1.9(0.3) | 1.7(0.5) | 0.08 | 0.01 |
| 1.5 Months | 1.6(0.4) | 1.5(0.4) | 0.3 | ||
| 3 Months | 1.5(0.3) | 1.7(0.5) | 0.06 | ||
| Vitamin B2 (mg) | Baseline | 1.9(0.7) | 2.0(0.8) | 0.5 | 0.1 |
| 1.5 Months | 1.9(0.6) | 1.8(0.6) | 0.1 | ||
| 3 Months | 1.7(0.7) | 1.9(0.7) | 0.1 | ||
| Vitamin B3 (mg) | Baseline | 30.3(13.2) | 27.8(12.9) | 0.4 | 0.4 |
| 1.5 Months | 25.7(10.2) | 27.2(14.7) | 0.8 | ||
| 3 Months | 24.2(10.6) | 26.3(11.9) | 0.4 | ||
| Vitamin B6 (mg) | Baseline | 2.2(0.7) | 1.9(0.6) | 0.05 | 0.02 |
| 1.5 Months | 1.8(0.5) | 1.8(0.7) | 0.5 | ||
| 3 Months | 1.7(0.6) | 1.9(0.6) | 0.1 | ||
| Folate (DFE) (μg) | Baseline | 504.0(222.7) | 443.6(181.5) | 0.3 | 0.04 |
| 1.5 Months | 425.7(155.8) | 426.5(149.7) | 0.7 | ||
| 3 Months | 413.0(126.7) | 471.7(152.9) | 0.05 | ||
| Vitamin B12 (μg) | Baseline | 4.1(2.5) | 4.4(2.5) | 0.6 | 0.6 |
| 1.5 Months | 5.1(2.8) | 4.6(2.5) | 0.5 | ||
| 3 Months | 4.3(3.0) | 4.4(2.4) | 0.9 | ||
| Vitamin K (μg) | Baseline | 148.6(224.4) | 134.9(287.3) | 0.1 | 0.8 |
| 1.5 Months | 142.6(279.7) | 86.5(135.3) | 0.5 | ||
| 3 Months | 192.2(366.3) | 168.8(419.1) | 0.6 | ||
| Zinc (mg) | Baseline | 12.4(3.3) | 12.7(3.7) | 0.7 | 0.7 |
| 1.5 Months | 12.6(4.0) | 12.4(3.3) | 0.7 | ||
| 3 Months | 11.5(3.7) | 12.1(4.5) | 0.6 | ||
| Selenium (μg) | Baseline | 113.7(34.2) | 119.9(54.1) | 0.7 | 0.2 |
| 1.5 Months | 120.4(58.1) | 99.2(37.9) | 0.09 | ||
| 3 Months | 112.0(57.7) | 106.6(44.5) | 0.6 | ||
| Fiber total (g) | Baseline | 29.8(12.8) | 31.0(16.1) | 0.9 | 0.09 |
| 1.5 Months | 30.1(16.5) | 27.1(15.1) | 0.5 | ||
| 3 Months | 24.5(9.5) | 31.5(15.6) | 0.03 | ||
*Total of the column: t-test or Mann-Whitney; **Total of the column: Two way repeated measures-ANOVA (TWRM-ANOVA)
Dietary status and measured biomarkers changes
The dietary intakes of energy, total fat, saturated fat, monounsaturated fatty acid, vitamins D, B1, B6, and folate during the study were higher in the placebo group (P < 0.05, Table 4), while other dietary patterns were almost identical between groups. These significant differences in terms of consumption were later considered as confounders in the final analysis model.
In the NC group, the mean difference of DBP was not significant (P > 0.05), although weight, BMI, WC, SBP, ALT, AST, TC, LDL-c, TG, FBS, FBI, HbA1c, FM, TNF-α, IL-6, hs-CRP, and HOMA-IR decreased, and HDL-c, QUICKI, and Nesfatin increased significantly (P < 0.05). In the placebo group, the mean difference of SBP was not significant (P > 0.05), but the weight, BMI, WC, DBP, ALT, AST, TC, LDL-c, TG, FBS, FBI, HbA1c, FM, TNF-α, IL-6, hs-CRP, and HOMA-IR decreased, and HDL-c, QUICKI, and Nesfatin increased significantly (P < 0.05).
The mean ± standard deviation of BMI at baseline and at the end of intervention were 30.6 ± 2.14 kg/m2 and 29.7 ± 2.10 kg/m2 in NC group, and 30.7 ± 2.35 kg/m2 and 29.9 ± 2.53 kg/m2 in the placebo group. These mean changes remained non-significant in both the unadjusted (P = 0.2) and adjusted models (P = 0.3).
As can be seen in the time by treatment interaction effect in both the unadjusted and adjusted analysis models, WC, ALT, AST, TC, LDL-c, TG, FBS, FBI, HbA1c, TNF-α, IL-6, hs-CRP, and HOMA-IR decreased, and HDL-c, QUICKI, and Nesfatin increased significantly among the NC group in comparison with the placebo group (P < 0.05) (Table 5). In other words, NC, in comparison with placebo, significantly increased HDL-c, QUICKI, and Nesfatin and decreased WC, ALT, AST, TC, LDL-c, TG, FBS, FBI, HbA1c, TNF-α, IL-6, hs-CRP, and HOMA-IR (P < 0.05). After adjustment for confounders, the significant changes remained similar (P < 0.05).
Table 5.
The changes in weight, BMI, glucose indices, nesfatin, inflammatory factors, and liver enzymes in overweight/obese NAFLD patients
| Variables | Intervention | Baseline Mean(SD) | 3 Months Mean(SD) | P-value$ | Mean Difference (95% CI) | P-value# | ||
|---|---|---|---|---|---|---|---|---|
| Time | Treatment | Interaction | ||||||
| Weight (kg) | Nanocurcumin (n = 42) | 86.5 (10.9) | 83.7 (10.7) | < 0.001 | −2.8 (−3.5, −2.0) | < 0.001 | 0.8 | 0.3 |
| Placebo (n = 42) | 86.7(11.0) | 84.3(11.0) | < 0.001 | −2.4 (−3.1, −1.6) | < 0. 1 | 0.3 | 0.3 | |
| BMI (kg/m2)* | Nanocurcumin (n = 42) | 30.6(2.14) | 29.7(2.10) | < 0.001 | −0.9 (− 1.0, −0.7) | < 0.001 | 0.6 | 0.2 |
| Placebo (n = 42) | 30.7(2.35) | 29.9(2.53) | < 0.001 | − 0.8 (− 0.9, − 0.6) | 0.03 | 0.9 | 0.3 | |
| Fat Mass (%) | Nanocurcumin (n = 42) | 31.6(6.4) | 30.3(6.5) | < 0.001 | −1.3 (− 1.7, − 0.8) | < 0.001 | 0.7 | 0.3 |
| Placebo (n = 42) | 31.9(4.7) | 30.7(4.9) | < 0.001 | −1.2 (− 1.5, − 0.8) | 0.003 | 0.1 | 0.9 | |
| Waist circumference (cm) | Nanocurcumin (n = 42) | 105.4(6.2) | 99.6(5.7) | < 0.001 | −5.8 (−6.2, −5.3) | < 0.001 | 0.6 | < 0.001 |
| Placebo (n = 42) | 103.8(6.7) | 102.5(6.9) | < 0.001 | −1.3 (− 1.7, − 0.8) | 0.07 | 0.9 | < 0.001 | |
| SBP (mmhg) | Nanocurcumin (n = 42) | 120.3(4.7) | 118.2(4.4) | 0.008 | −2.1 (−2.4, −1.7) | 0.001 | 0.3 | 0.3 |
| Placebo (n = 42) | 120.7(4.3) | 119.6(4.9) | 0.07 | −1.1 (−1.4, −0.7) | 0.7 | 0.4 | 0.8 | |
| DBP (mmhg) | Nanocurcumin (n = 42) | 78.8(4.5) | 77.9(2.9) | 0.102 | −0.9 (−1.1, − 0.6) | 0.008 | 0.2 | 0.7 |
| Placebo (n = 42) | 79.8(4.3) | 78.7(3.4) | 0.03 | −1.1 (−1.3, 0.8) | 0.4 | 0.4 | 0.7 | |
| ALT (u/l)** | Nanocurcumin (n = 42) | 42.8(11.6) | 32.6(9.9) | < 0.001 | −10.2 (− 10.9, −9.4) | < 0.001 | 0.1 | < 0.001 |
| Placebo (n = 42) | 42.1(8.2) | 39.6(7.5) | 0.001 | −2.5 (−3.0, 1.9) | 0.4 | 0.3 | < 0.001 | |
| AST (u/l)^ | Nanocurcumin (n = 42) | 28.43(6.7) | 22.03(5.9) | < 0.001 | −6.4 (−6.8, −5.9) | < 0.001 | 0.3 | < 0.001 |
| Placebo (n = 42) | 27.60(7.8) | 25.63(7.2) | 0.002 | −1.97 (−2.47, − 1.46) | 0.052 | 0.7 | < 0.001 | |
| TC (mg/dl) | Nanocurcumin (n = 42) | 212.9(18.9) | 195.2(19.8) | < 0.001 | −17.7 (− 19.0, − 16.3) | < 0.001 | 0.2 | 0.002 |
| Placebo (n = 42) | 211.8(21.8) | 205.0(20.5) | 0.005 | −6.8 (−8.2, 5.3) | 0.3 | 0.9 | 0.01 | |
| LDL-c (mg/dl) | Nanocurcumin (n = 42) | 135.6(17.6) | 114.6(20.5) | < 0.001 | −21.0 (−22.2, −19.7) | < 0.001 | 0.3 | < 0.001 |
| Placebo (n = 42) | 133.0(20.7) | 125.7(22.2) | < 0.001 | −7.3 (−8.7, −5.8) | 0.01 | 0.9 | 0.003 | |
| TG (mg/dl) | Nanocurcumin (n = 42) | 175.9(70.3) | 142.5(49.9) | < 0.001 | −33.4 (−37.5, −29.2) | < 0.001 | 0.1 | < 0.001 |
| Placebo (n = 42) | 181.2(65.6) | 175.3(62.5) | 0.007 | −5.9 (−10.2, −1.5) | 0.5 | 0.057 | 0.001 | |
| HDL-c (mg/dl) | Nanocurcumin (n = 42) | 41.8(5.6) | 51.4(6.6) | < 0.001 | 9.6 (9.1, 10.0) | < 0.001 | 0.009 | < 0.001 |
| Placebo (n = 42) | 42.7(5.7) | 43.8(5.4) | 0.001 | 1.1 (0.7, 1.4) | < 0.001 | 0.04 | < 0.001 | |
| FBS (mg/dl) | Nanocurcumin (n = 42) | 89.1(5.4) | 86.3(5.2) | < 0.001 | −2.8 (−3.1, −2.4) | < 0.001 | 0.3 | < 0.001 |
| Placebo (n = 42) | 89.3(5.8) | 88.2(5.5) | < 0.001 | −1.1 (−1.4, 0.7) | 0.6 | 0.2 | 0.03 | |
| HbA1c (%) | Nanocurcumin (n = 42) | 5.2(0.218) | 5.1(0.229) | < 0.001 | −0.1 (−0.115, −0.085) | < 0.001 | 0.1 | 0.001 |
| Placebo (n = 42) | 5.3(0.218) | 5.2(0.188) | < 0.001 | −0.1 (− 0.114, − 0.086) | 0.01 | 0.8 | 0.002 | |
| Nesfatin (ng/ml) | Nanocurcumin (n = 42) | 1.81(0.54) | 3.37(8.8) | < 0.001 | 1.56 (1.52, 1.59) | < 0.001 | < 0.001 | < 0.001 |
| Placebo (n = 42) | 1.88(0.49) | 2.06(7.6) | < 0.001 | .18 (0.14, 0.21) | < 0.001 | 0.003 | < 0.001 | |
| FBI (μIU/ml) | Nanocurcumin (n = 42) | 8.0(0.5) | 6.5(0.9) | < 0.001 | −1.5 (−1.54, −1.45) | < 0.001 | 0.001 | < 0.001 |
| Placebo (n = 42) | 7.9(0.6) | 7.6(0.7) | < 0.001 | −0.3 (−0.34, −0.25) | 0.3 | 0.08 | < 0.001 | |
| TNF-α (ng/l) | Nanocurcumin (n = 42) | 14.7(3.3) | 7.3(2.9) | < 0.001 | −7.4 (−7.5, −7.2) | < 0.001 | < 0.001 | < 0.001 |
| Placebo (n = 42) | 15.1(2.7) | 13.7(4.0) | < 0.001 | 1.4 (−1.63, −1.16) | 0.002 | < 0.001 | < 0.001 | |
| IL-6 (ng/l) | Nanocurcumin (n = 42) | 7.60(1.56) | 3.81(1.63) | < 0.001 | −3.79 (−3.88, −3.69) | < 0.001 | < 0.001 | < 0.001 |
| Placebo (n = 42) | 7.99(1.52) | 7.02(7.6) | < 0.001 | −0.97 (−1.07, −0.86) | < 0.001 | < 0.001 | < 0.001 | |
| hs-CRP (mg/l) | Nanocurcumin (n = 42) | 5.9(2.57) | 3.6(1.58) | < 0.001 | −2.3 (−2.44, −2.15) | < 0.001 | 0.3 | < 0.001 |
| Placebo (n = 42) | 5.3(2.50) | 5.2(2.47) | < 0.001 | 0.1 (−0.26, 0.06) | < 0.001 | 0.3 | < 0.001 | |
| HOMA-IR (score) | Nanocurcumin (n = 42) | 1.77(0.16) | 1.39(0.21) | < 0.001 | −0.38 (− 0.39, − 0.36) | < 0.001 | 0.001 | < 0.001 |
| Placebo (n = 42) | 1.75(0.17) | 1.65(0.18) | < 0.001 | 0.1 (−0.11, − 0.08) | 0.3 | 0.04 | < 0.001 | |
| QUICKI (score) | Nanocurcumin (n = 42) | 0.3505(0.0049) | 0.3643(0.0092) | < 0.001 | 0.0138 0.0143, 0.0133) | < 0.001 | 0.001 | < 0.001 |
| Placebo (n = 42) | 0.3509(0.0057) | 0.3543(0.0068) | < 0.001 | 0.0034 (0.0038, 0.0029) | 0.3 | 0.3 | < 0.001 | |
*Inversely transformed; **Transformed by square root; ^Logarithmically transformed; $Paired t-test; #Two way repeated measures-ANOVA (TWRM-ANOVA), top row: unadjusted; bottom row: adjusted for energy, total fat, saturated fat, monounsaturated fatty acid, vitamins D, B1, B6, and folate. ALT alanine transaminase, AST aspartate transaminase, BMI body mass index, FBI fasting blood insulin,FBS fasting blood sugar, HOMA-IR homeostasis model assessment-insulin resistance, hs-CRP: high-sensitive C-reactive protein, HDL-C: high density lipoprotein-cholesterol, IL-6: interleukin-6, LDL-C: low density lipoprotein-cholesterol, QUICKI: quantitative insulin sensitivity check index, TC: total cholesterol, TNF-α: tumor necrosis factor-alpha
Safety
The patients reported no side-effects and side-events associated with treatment during the study, with the exception of one patient in the NC group, who reported nausea.
Discussion
This trial was the first to assess the effects of NC on serum levels of some important factors related to overweight, obesity, and NAFLD.
The baseline variables were similar between the two groups with an exception for the dietary vitamin D intake was higher in the placebo group. Possible reasons for high entry similarity include single center selection and similar participant socio-economic levels.
According to both the unadjusted and adjusted analysis, NC (in comparison with placebo) significantly increased HDL-c, QUICKI, and nesfatin, and decreased WC, ALT, AST, TC, LDL-c, TG, FBS, FBI, HbA1c, TNF-α, IL-6, hs-CRP, and HOMA-IR. The changes in weight, BMI, and FM among the NC group in comparison with placebo were not significant.
Many studies have shown the significant effects of curcumin (especially more accessible forms such as NC) on anthropometric measurements (weight, BMI, WC, FM) [6, 17–25]. The different mechanisms have been proposed for effects of curcumin on anthropometric measurements including inhibition of the adipocytes, lipogenesis, fat mass, and inflammation, increasing lipolysis, energy consumption [26, 27], and brown fat tissue, and probiotics-like effects [24]. Also, the effects of nesfatin on anthropometrics include decreased appetite [28, 29], BMI [26, 30, 31], and WC [26]. Therefore, the WC improvement in this study may be attributed to the increased levels of serum nesfatin by NC supplementation.
In a study on NAFLD, curcumin not changed SBP and DBP significantly [17], although, SBP was significantly decreased in the NC group in this study. However, the beneficial effects of curcumin on blood pressure have been reported in two reviews [20, 27]. Some mechanisms have been proposed including improvement of vascular reactivity [27], inflammation, oxidative stress, and anthropometrics [20]. The studies of nesfatin’s effects on hypertension are divisive [32]. The reason for no changes of BP may be the normal BP of the participants at baseline.
The turmeric in a study on NAFLD patients significantly reduced, HOMA-R, FBS, and FBI [22]. Several reviews have reported the beneficial effects of curcumin on the glucose indices (FBS, HbA1c) [23, 24, 33, 34]. Some mechanisms were decreasing inflammatory factors, glucose production, glycogenolysis, insulin resistance, adipocytes, FM, weight and increasing antioxidant activity, glucose uptake, catabolism, and insulin sensitivity [24, 33, 35, 36]. In a study among diabetics, curcumin significantly reduced TC, non-HDL-c, and Lp(a) and increased HDL-c. TG and LDL-c significantly decreased only in the curcumin group [37]. The reasons for some mismatches may be the differences among patients and the supplement type and dose. In two separate studies in diabetic [18] and dyslipidemic patients [38], curcumin significantly reduced TG but no other lipids. The reasons of difference may be the different patients, intervention duration, and supplementation type and dose. The beneficial effects of curcumin on lipids have been reported in several reviews [18, 24, 33–53]. The proposed mechanisms were reducing inflammation, oxidative stress [20, 21, 41, 42], obesity, adipogenesis [20], HMG-CoA reductase, cholesterol absorption and intestinal transmission, apo-B100 expression [51], and lipogenic genes expression, increasing LDL receptors [52], regulating of some genes involved in lipoprotein [20] and lipid metabolism, anti-atherogenic effects, and statins-like functions [51]. Also, nesfatin can improve glucose profiles by many mechanisms [5, 54–58]. Thus, the present improvements may be attributed to increased nesfatin levels. In addition, the effect of NC on the WC can improve glucose indices [28, 55] and lipids [59, 60].
The NC in infertile men significantly increased total antioxidant capacity and reduced CRP and TNF-α levels [61]. The anti-inflammatory targets of curcumin have been mentioned TNF-α, IL-1β, NF-κB, IL-6, COX2, 5-LOX, iNOS, IL-17A, IL-17F, and IL-22 [23]. Also, several reviews have shown the anti-inflammatory effects of curcumin [27, 36, 41–49, 62–64]. Its proposed anti-inflammatory mechanisms were reducing expression and release of inflammatory factors [27, 36, 39, 44, 63, 65] and PPARγ activation [66]. Also, decreases in WC can improve inflammatory factors including TNF-α, IL-6, and hs-CRP [49, 50]. The beneficial effects of nesfatin on inflammation and oxidative stress have been reported in the previous studies including reducing the NF-κB expression, IL-6, IL-1β, TNF-α, and apoptosis [67, 68].
In only two separate studies of Jujube and Nigella sativa effects on nesfatin, Jujube increased liver and plasma levels of it. The reported important effects for nesfatin are appetite loss, anti-hyperglycemic, anti-inflammation, neuroendocrine regulators, reducing body fat, and metabolic regulation [69]. Thus, the improvements in glucose indices [5, 54, 56–58], lipids [57, 58], inflammation [67], and anthropometrics [5, 54, 56], and subsequently NAFLD, may be attributed to increased nesfatin levels by NC supplementation.
Some animal studies of curcumin/turmemeric supplementation have shown the beneficial effects on fatty liver and serum aminotransferases [61, 70–73]. In a study on NAFLD patients, curcumin significantly reduced fatty liver degree, ALT, and AST [17]. According to three separate reviews, curcumin can reduce ALT, AST [39, 40], ALP, GGT [40], steatosis, inflammation, and ROS [21]. However, a study of the turmeric effects on liver enzymes and fatty liver degree in NAFLD patients showed no significant changes [22]. The reason for the differences may be the type and form of supplementation. The other proposed mechanisms in improving liver enzymes and fatty liver by curcumin were reducing inflammatory markers, lipid synthesis/accumulation, anthropometric measurements [17], and oxidative stress [40, 70], PPARγ activation, and effects on glycolysis [73]. Also, the effect of nesfatin on steatosis improvement has been reported in a previous animal study [57].
The strengths were first examining the effect of NC in obese NAFLD patients, especially by assessing nefatin levels, stratified blocked randomization, the newly diagnosed NAFLD patients no receiving treatment, and assessing dietary intakes and physical activity levels. However, some limitations were no liver biopsy and measuring GGT and certain factors mentioned (PPAR-γ, etc.), self-reporting of dietary intakes and physical activity, selecting a specific center, and no checking the bioavailability and blood levels of NC.
Conclusion
NC supplementation in overweight and obese NAFLD patients improved some markers related to obesity and NAFLD including nesfatin, QUICKI, fatty liver degree, WC, glucose indices, lipids, inflammation, and liver transaminases. The effects of NC on weight, BMI, FM, SBP, and DBP were not significant. Accordingly, the proposed mechanism for ameliorating NAFLD with NC supplementation was approved by increasing serum nesfatin levels and likely subsequently improving inflammatory, lipid and glucose profiles. Further trials on effects of curcumin are suggested, involving larger sample sizes, longer durations, non-obese patients, and considering the mentioned limitations.
Acknowledgments
The support of Tehran University of Medical Sciences and cooperation of the central hospital of the NIOC, Tehran, Iran, are acknowledged (grant no. 31581).
Patient consent
Details have been removed from this case description/these case descriptions to ensure anonymity. The editors and reviewers have seen the detailed information available and are satisfied that the information backs up the case the authors are making.
Provenance and peer review
Not commissioned; externally peer reviewed.
Funding
The trial funding was supported by Tehran University of Medical Sciences (grant no. 31581).
Availability of data and materials
Not applicable.
Abbreviations
- ALT
Alanine transaminase
- AMPK
Adenosine monophosphate-activated protein kinase
- AST
Aspartate transaminase
- BMI
Body mass index
- CNS
Central nervous system
- COX2
Cyclooxygenase-2
- DBP
Diastolic blood presure
- DFE
Dietary Folate Equivalent
- ECL
Electrochemiluminescence
- EGCG
Epigallocatechin gallate
- EIA
Enzyme immunoassay
- ELISA
Enzyme-linked immunosorbent assay
- FBI
Fasting blood insulin
- FBS
Fasting blood sugar
- GGT
Gamma-glutamyltransferase
- GGT
Gamma-glutamyltransferase
- HbA1c
hemoglobin A1c
- HDL-C
High density lipoprotein-cholesterol
- HOMA-IR
Homeostasis model assessment-insulin resistance
- hs-CRP
High-sensitivity C-reactive protein
- IL-1β
Interleukin-1 beta
- IL-6
Interleukin-6
- iNOS
inducible-nitric oxide synthase
- LDH
Lactate dehydrogenase
- LDL-C
Low density lipoprotein-cholesterol
- LXR
Liver X receptor
- MAPKs
Mitogen-activated protein kinases
- MCP
Monocyte chemoattractant protein
- NAFLD
Non-alcoholic fatty liver disease
- NF-κB
Nnuclear factor kappa B
- NIOC
National Iranian Oil Company
- NO
Nitric oxide
- PGC-1α
PPAR-γ co-activator-1 alpha
- PGC-1α
PPAR-γ co-activator-1 alpha
- PPAR
Peroxisome proliferation activated receptor
- QUICKI
Quantitative insulin sensitivity check index
- ROS
Reactive oxygen species
- SBP
Systolic blood presure
- SF-IPAQ
Short Form-International physical activity questionnaire
- SREBPs
Sterol regulatory element–binding proteins
- SREBPs
Sterol regulatory element–binding proteins
- TC
Total cholesterol
- TNF-α
Ttumor necrosis factor-alpha
- TPN
Total parenteral nutrition
- TWRM-ANOVA
Two way repeated measures-Analysis of variance
- UCPs
Uncoupling proteins
- USDA
United States Department of Agriculture
Authors contributions
SAJT, MJHA, and SMR conceived and developed the idea for the paper and revised the manuscript. SAJT, MDM, SM, and SMA contributed to data collection. SAJT and MDM wrote numerous drafts. MQ contributed to statistical interpretations. All authors read and approved the final manuscript.
Ethics approval and consent to participate
The ethical approval of this trial was conducted by the ethics committee of Tehran University of Medical Sciences (Ethical Code: IR.TUMS.REC.1395.2612). All the participants completed an informed consent form (in Persian). Participation in and continuation of the supplementation were free and voluntary for the patients. In the trial, advice on the lifestyle modification was presented to the patients free of charge. The health care services of the hospital were provided without inconsistency. No side effects of the supplements were reported. The patients’ personal information was kept confidential.
Consent for publication
Not applicable.
Competing interests
None declared.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Seyed Ali Jazayeri-Tehrani, Email: jazayeri.sa@gmail.com.
Seyed Mahdi Rezayat, Phone: +989124708873, Email: rezayat@tums.ac.ir.
Siavash Mansouri, Email: siavash.mansouri@gmail.com.
Mostafa Qorbani, Email: mqorbani1379@yahoo.com.
Seyed Moayed Alavian, Email: alavian@thc.ir.
Milad Daneshi-Maskooni, Email: miladdaneshi@gmail.com.
Mohammad-Javad Hosseinzadeh-Attar, Phone: +989123717305, Email: mhosseinzadeh@tums.ac.ir, Email: mjha3@yahoo.com.
References
- 1.Jazayeri-Tehrani SA, Rezayat SM, Mansouri S, Qorbani M, Alavian SM, Daneshi-Maskooni M, Hosseinzadeh-Attar MJ. Efficacy of nanocurcumin supplementation on insulin resistance, lipids, inflammatory factors and nesfatin among obese patients with non-alcoholic fatty liver disease (NAFLD): a trial protocol. BMJ Open. 2017;7(7):e016914. doi: 10.1136/bmjopen-2017-016914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Jazayeri-Tehrani SA, Rezayat SM, Mansouri S, Qorbani M, Alavian SM, Daneshi-Maskooni M, Hosseinzadeh-Attar MJ. The nanocurcumin reduces appetite in obese patients with non-alcoholic fatty liver disease (nafld): a double-blind randomized placebo-controlled clinical trial. Nanomedicine Journal. 2018;5(2):67–76. [Google Scholar]
- 3.Daneshi-Maskooni M, Keshavarz SA, Qorbani M, Mansouri S, Alavian SM, Badri-Fariman M, Jazayeri-Tehrani SA, Sotoudeh G. Green cardamom increases Sirtuin-1 and reduces inflammation in overweight or obese patients with non-alcoholic fatty liver disease: a double-blind randomized placebo-controlled clinical trial. Nutrition & metabolism. 2018;15(1):63. doi: 10.1186/s12986-018-0297-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Daneshi-Maskooni M, Keshavarz SA, Mansouri S, Qorbani M, Alavian SM, Badri-Fariman M, Jazayeri-Tehrani SA, Sotoudeh G. The effects of green cardamom on blood glucose indices, lipids, inflammatory factors, paraxonase-1, sirtuin-1, and irisin in patients with nonalcoholic fatty liver disease and obesity: study protocol for a randomized controlled trial. Trials. 2017;18(1):260. doi: 10.1186/s13063-017-1979-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ayada C, Toru Ü, Korkut Y. Nesfatin-1 and its effects on different systems. Hippokratia. 2015;19:4. [PMC free article] [PubMed] [Google Scholar]
- 6.Başar O, Akbal E, Köklü S, et al. A novel appetite peptide, nesfatin-1 in patients with the non-alcoholic fatty liver disease. Scand J Clin Lab Invest. 2012;72:479–483. doi: 10.3109/00365513.2012.699097. [DOI] [PubMed] [Google Scholar]
- 7.Dixon JB, Bhathal PS, Hughes NR, O’Brien PE. Nonalcoholic fatty liver disease: improvement in liver histological analysis with weight loss. Hepatology. 2004;39:1647–1654. doi: 10.1002/hep.20251. [DOI] [PubMed] [Google Scholar]
- 8.Luyckx FH, Desaive C, Thiry A, Dewe W, Scheen AJ, Gielen JE, Lefebvre PJ. Liver abnormalities in severely obese subjects: effect of drastic weight loss after gastroplasty. Int J Obes Relat Metab Disord. 1998;22:222–226. doi: 10.1038/sj.ijo.0800571. [DOI] [PubMed] [Google Scholar]
- 9.Verma SK, Jain V, Singh DP. Effect of greater cardamom (Amomum subulatum Roxb.) on blood lipids, fibrinolysis and total antioxidant status in patients with ischemic heart disease. Asian Pacific J Trop Dis. 2012;2:S739–S743. [Google Scholar]
- 10.Palmer M, Schaffner F. Effect of weight reduction on hepatic abnormalities in overweight patients. Gastroenterology. 1990;99:1408–1413. doi: 10.1016/0016-5085(90)91169-7. [DOI] [PubMed] [Google Scholar]
- 11.The lab tests online. Laboratory methods used for medical lab testing (enzyme-linked immunosorbent assay (ELISA)) [Internet]. 2012. [cited 26 Feb 2017]. Available from: https://labtestsonline.org/understanding/features/methods/start/2/. Accessed 26 Feb 2017.
- 12.Jazayeri S, Nouri M, Pour-Ebrahim R, Fakhrzadeh H, Larijani B. Food and nutrient intake status in urban population (20–60 years) of population research center of Tehran University of Medical Sciences. Iran J Diab Lipid. 2005;1(3):81–89. [Google Scholar]
- 13.Qaffarpour M, Houshyar-Rad A, Kianfar H. Guides of home scales, conversion coefficients and percentage of Iranian foods. Tehran: Agricultural Sciences Publication; 2000. [Google Scholar]
- 14.The IPAQ group. International Physical Activity Questionnaire [Internet]. 2017. [cited 26 Feb 2017]. Available from: https://sites.google.com/site/theipaq/.
- 15.Booth ML, Ainsworth BE, Pratt MI, Ekelund U, Yngve AG, Sallis JF, Oja PE. International physical activity questionnaire: 12-country reliability and validity. Med Sci Sports Exerc. 2003;195(9131/03):3508–1381. doi: 10.1249/01.MSS.0000078924.61453.FB. [DOI] [PubMed] [Google Scholar]
- 16.Chuengsamarn S, Rattanamongkolgul S, Luechapudiporn R, et al. Curcumin extract for prevention of type 2 diabetes. Diabetes Care. 2012;35:2121–2127. doi: 10.2337/dc12-0116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Panahi Y, Kianpour P, Mohtashami R, Jafari R, Simental-Mendía LE, Sahebkar A. Efficacy and safety of phytosomal curcumin in non-alcoholic fatty liver disease: a randomized controlled trial. Drug research. 2017;67(04):244–251. doi: 10.1055/s-0043-100019. [DOI] [PubMed] [Google Scholar]
- 18.Chuengsamarn S, Rattanamongkolgul S, Phonrat B, Tungtrongchitr R, Jirawatnotai S. Reduction of atherogenic risk in patients with type 2 diabetes by curcuminoid extract: a randomized controlled trial. J Nutr Biochem. 2014;25(2):144–150. doi: 10.1016/j.jnutbio.2013.09.013. [DOI] [PubMed] [Google Scholar]
- 19.Panahi Y, Khalili N, Sahebi E, Namazi S, Karimian MS, Majeed M, Sahebkar A. Antioxidant effects of curcuminoids in patients with type 2 diabetes mellitus: a randomized controlled trial. Inflammopharmacology. 2017;25(1):25–31. doi: 10.1007/s10787-016-0301-4. [DOI] [PubMed] [Google Scholar]
- 20.Hewlings SJ, Kalman DS. Curcumin: a review of its’ effects on human health. Foods. 2017;6(10):92. doi: 10.3390/foods6100092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lizana P, Galdames M, Rodrigo R. Oxidative stress and endoplasmic reticulum stress as potential therapeutic targets in non-alcoholic fatty liver disease. Reactive Oxygen Species. 2017;4(10):266–274. [Google Scholar]
- 22.Navekar R, Rafraf M, Ghaffari A, Asghari-Jafarabadi M, Khoshbaten M. Turmeric supplementation improves serum glucose indices and leptin levels in patients with nonalcoholic fatty liver diseases. J Am Coll Nutr. 2017;36(4):261–267. doi: 10.1080/07315724.2016.1267597. [DOI] [PubMed] [Google Scholar]
- 23.Kocaadam B, Şanlier N. Curcumin, an active component of turmeric (Curcuma longa), and its effects on health. Crit Rev Food Sci Nutr. 2017;57(13):2889–2895. doi: 10.1080/10408398.2015.1077195. [DOI] [PubMed] [Google Scholar]
- 24.Jin T, Song Z, Weng J, Fantus IG. Curcumin and other dietary polyphenols: potential mechanisms of metabolic actions and therapy for diabetes and obesity. American Journal of Physiology-Endocrinology and Metabolism. 2018;314(3):E201–E205. doi: 10.1152/ajpendo.00285.2017. [DOI] [PubMed] [Google Scholar]
- 25.Hariri M, Haghighatdoost F. Effect of curcumin on anthropometric measures: a systematic review on randomized clinical trials. J Am Coll Nutr. 2018;37(3):215–222. doi: 10.1080/07315724.2017.1392263. [DOI] [PubMed] [Google Scholar]
- 26.Shen P, Han Y, Cai B, Wang Y. Decreased levels of serum nesfatin-1 in patients with obstructive sleep apnea syndrome. Sleep and Breathing. 2015;19(2):515–522. doi: 10.1007/s11325-014-1039-0. [DOI] [PubMed] [Google Scholar]
- 27.Zhang DW, Fu M, Gao SH, Liu JL. Curcumin and diabetes: a systematic review. Evid Based Complement Alternat Med. 2013;2013. [DOI] [PMC free article] [PubMed]
- 28.Syabaniarti AN. The Relationship between Body Mass Index (BMI) and Waist Circumference (WC) to fasting blood glucose level based on the medical check-up result of PT. X Regional Jakarta Employees Year 2016. 2nd International Seminar on Global Health (ISGH); 2018: 306–312. http://repository.stikesayani.ac.id/index.php/isgh2/article/view/66.
- 29.Dong J, Xu H, Wang PF, Cai GJ, Song HF, Wang CC, Dong ZT, Ju YJ, Jiang ZY. Nesfatin-1 stimulates fatty-acid oxidation by activating AMP-activated protein kinase in STZ-induced type 2 diabetic mice. PLoS One. 2013;8(12):e83397. doi: 10.1371/journal.pone.0083397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kim SH, Ahn M, Jung IA, Jeon YJ, Cho WK, Cho KS, Park SH, Jung MH, Suh BK. The relation of serum Nesfatin-1 level with anthropometric and metabolic parameters in Korean children and young adolescents. ESPE Abstracts 2015; 84: P2–375. http://abstracts.eurospe.org/hrp/0084/hrp0084p2-375.htm.
- 31.Abaci A, Catli G, Anik A, Kume T, Bober E. The relation of serum nesfatin-1 level with metabolic and clinical parameters in obese and healthy children. Pediatr Diabetes. 2013;14(3):189–195. doi: 10.1111/pedi.12009. [DOI] [PubMed] [Google Scholar]
- 32.Hamedani NK, Hosseinzadeh-Attar MJ, Hosseini M. The correlation between nesfatin-1 and blood pressure in healthy normal weight and obese adults. International Research Journal of Applied and Basic Sciences. 2014;8(4):398–400. [Google Scholar]
- 33.de Meloa IS, dos Santosb AF, Buenoc NB. Curcumin or combined curcuminoids are effective in lowering the fasting blood glucose concentrations of individuals with dysglycemia: systematic review and meta-analysis of randomized controlled trials. Pharmacol Res. 2017;128:137–144. doi: 10.1016/j.phrs.2017.09.010. [DOI] [PubMed] [Google Scholar]
- 34.Rahimi HR, Mohammadpour AH, Dastani M, Jaafari MR, Abnous K, Ghayour MM, Kazemi RO. The effect of nano-curcumin on HbA1c, fasting blood glucose, and lipid profile in diabetic subjects: a randomized clinical trial. Avicenna journal of phytomedicine. 2016;6(5):567–577. [PMC free article] [PubMed] [Google Scholar]
- 35.Kim HG, Lee SB, Lee JS, Kim WY, Choi SH, Son CG. Artemisia iwayomogi plus Curcuma longa Synergistically Ameliorates Nonalcoholic Steatohepatitis in HepG2 Cells. Evidence-Based Complementary and Alternative Medicine. 2017; Article ID 4390636. 9 pages. Doi: 10.1155/2017/4390636. [DOI] [PMC free article] [PubMed]
- 36.Ghorbani Z, Hekmatdoost A, Mirmiran P. Anti-hyperglycemic and insulin sensitizer effects of turmeric and its principal constituent curcumin. International journal of endocrinology and metabolism. 2014;12(4):e18081. doi: 10.5812/ijem.18081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Panahi Y, Khalili N, Sahebi E, Namazi S, Reiner Ž, Majeed M, Sahbekar A. Curcuminoids modify lipid profile in type 2 diabetes mellitus: a randomized controlled trial. Complementary therapies in medicine. 2017;33:1–5. doi: 10.1016/j.ctim.2017.05.006. [DOI] [PubMed] [Google Scholar]
- 38.Mohammadi A, Sahebkar A, Iranshahi M, Amini M, Khojasteh R, Ghayour-Mobarhan M, Ferns GA. Effects of supplementation with curcuminoids on dyslipidemia in obese patients: a randomized crossover trial. Phytother Res. 2013;27(3):374–379. doi: 10.1002/ptr.4715. [DOI] [PubMed] [Google Scholar]
- 39.Gupta SC, Patchva S, Aggarwal BB. Therapeutic roles of curcumin: lessons learned from clinical trials. AAPS J. 2013;15(1):195–218. doi: 10.1208/s12248-012-9432-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.S Darvesh A, B Aggarwal B, Bishayee A. Curcumin and liver cancer: a review. Curr Pharm Biotechnol. 2012;13(1):218–228. doi: 10.2174/138920112798868791. [DOI] [PubMed] [Google Scholar]
- 41.Ballantyne M. The Pharmacognosy and Therapeutic Efficacy of Turmeric (Curcuma longa): A Systematic Review. Senior Honors Projects. 2017; Paper 573. http://digitalcommons.uri.edu/srhonorsprog/573
- 42.Kapakos G, Youreva V, Srivastava AK. Cardiovascular protection by curcumin: molecular aspects. Indian J Biochem Biophys. 2012;49(5):306–315. [PubMed] [Google Scholar]
- 43.Lopresti AL, Hood SD, Drummond PD. Multiple antidepressant potential modes of action of curcumin: a review of its anti-inflammatory, monoaminergic, antioxidant, immune-modulating and neuroprotective effects. J Psychopharmacol. 2012;26(12):1512–1524. doi: 10.1177/0269881112458732. [DOI] [PubMed] [Google Scholar]
- 44.Meng B, Li J, Cao H. Antioxidant and anti-inflammatory activities of curcumin on diabetes mellitus and its complications. Curr Pharm Des. 2013;19(11):2101–2113. [PubMed] [Google Scholar]
- 45.Mirzaei H, Shakeri A, Rashidi B, Jalili A, Banikazemi Z, Sahebkar A. Phytosomal curcumin: a review of pharmacokinetic, experimental and clinical studies. Biomed Pharmacother. 2017;85:102–112. doi: 10.1016/j.biopha.2016.11.098. [DOI] [PubMed] [Google Scholar]
- 46.Noorafshan A, Ashkani-Esfahani S. A review of the therapeutic effects of curcumin. Curr Pharm Des. 2013;19(11):2032–2046. [PubMed] [Google Scholar]
- 47.Sahebkar A, Cicero AF, Simental-Mendía LE, Aggarwal BB, Gupta SC. Curcumin downregulates human tumor necrosis factor-α levels: a systematic review and meta-analysis of randomized controlled trials. Pharmacol Res. 2016;107:234–242. doi: 10.1016/j.phrs.2016.03.026. [DOI] [PubMed] [Google Scholar]
- 48.Shehzad A, Lee YS. Molecular mechanisms of curcumin action: signal transduction. Biofactors. 2013;39(1):27–36. doi: 10.1002/biof.1065. [DOI] [PubMed] [Google Scholar]
- 49.Schlecht I, Fischer B, Behrens G, Leitzmann MF. Relations of visceral and abdominal subcutaneous adipose tissue, body mass index, and waist circumference to serum concentrations of parameters of chronic inflammation. Obesity facts. 2016;9(3):144–157. doi: 10.1159/000443691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Stepanikova I, Oates GR, Bateman LB. Does one size fit all? The role of body mass index and waist circumference in systemic inflammation in midlife by race and gender. Ethnicity & health. 2017;22(2):169–183. doi: 10.1080/13557858.2016.1235681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Panahi Y, Ahmadi Y, Teymouri M, Johnston TP, Sahebkar A. Curcumin as a potential candidate for treating hyperlipidemia: a review of cellular and metabolic mechanisms. J Cell Physiol. 2018;233(1):141–152. doi: 10.1002/jcp.25756. [DOI] [PubMed] [Google Scholar]
- 52.Qin S, Huang L, Gong J, Shen S, Huang J, Ren H, Hu H. Efficacy and safety of turmeric and curcumin in lowering blood lipid levels in patients with cardiovascular risk factors: a meta-analysis of randomized controlled trials. Nutr J. 2017;16(1):68. doi: 10.1186/s12937-017-0293-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Sahebkar A. A systematic review and meta-analysis of randomized controlled trials investigating the effects of curcumin on blood lipid levels. Clin Nutr. 2014;33(3):406–414. doi: 10.1016/j.clnu.2013.09.012. [DOI] [PubMed] [Google Scholar]
- 54.Li ZL, Xu L, Sun XR, Guo FF, Gong YL, Gao SL. Central nesfatin-1 influences the excitability of ghrelin-responsive gastric distension neurons in the arcuate nucleus and reduces gastric motility in rats. Eur J Neurosci. 2013;38(11):3636–3643. doi: 10.1111/ejn.12366. [DOI] [PubMed] [Google Scholar]
- 55.Veghari G, Sedaghat M, Joshaghani H, Banihashem S, Moharloei P, Angizeh A, Tazik E, Moghaddami A, Hajian-Tilaki K, ZahedPasha Y. The association of fasting blood glucose (FBG) and waist circumference in northern adults in Iran: a population-based study. Journal of Diabetes & Metabolic Disorders 2014 Dec;13(1):2. http://www.jdmdonline.com/content/13/1/2 [DOI] [PMC free article] [PubMed]
- 56.Ramesh N, Gawli K, Kiran Pasupulleti V, Unniappan S. Metabolic and cardiovascular actions of nesfatin-1: implications in health and disease. Curr Pharm Des. 2017;23(10):1453–1464. doi: 10.2174/1381612823666170130154407. [DOI] [PubMed] [Google Scholar]
- 57.Yin Y, Li Z, Gao L, Li Y, Zhao J, Zhang W. AMPK-dependent modulation of hepatic lipid metabolism by nesfatin-1. Mol Cell Endocrinol. 2015;417:20–26. doi: 10.1016/j.mce.2015.09.006. [DOI] [PubMed] [Google Scholar]
- 58.Catak Z, Aydin S, Sahin İ, Kuloglu T, Aksoy A, Dagli AF. Regulatory neuropeptides (ghrelin, obestatin, and nesfatin-1) levels in serum and reproductive tissues of female and male rats with the fructose-induced metabolic syndrome. Neuropeptides. 2014;48(3):167–177. doi: 10.1016/j.npep.2014.04.002. [DOI] [PubMed] [Google Scholar]
- 59.Mohanna S, Baracco R, Seclén S. Lipid profile, waist circumference, and body mass index in a high altitude population. High Alt Med Biol. 2006;7(3):245–255. doi: 10.1089/ham.2006.7.245. [DOI] [PubMed] [Google Scholar]
- 60.Kang HT, Yoon JH, Kim JY, Ahn SK, Linton JA, Koh SB, Kim JK. The association between the ratio of triglyceride to HDL-C and insulin resistance according to waist circumference in a rural Korean population. Nutr Metab Cardiovasc Dis. 2012;22(12):1054–1060. doi: 10.1016/j.numecd.2011.01.013. [DOI] [PubMed] [Google Scholar]
- 61.Sengupta M, Sharma GD, Chakraborty B. Hepatoprotective and immunomodulatory properties of aqueous extract of Curcuma longa in carbon tetrachloride intoxicated Swiss albino mice. Asian Pacific journal of tropical biomedicine. 2011;1(3):193–199. doi: 10.1016/S2221-1691(11)60026-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Jurenka JS. Anti-inflammatory properties of curcumin, a major constituent of Curcuma longa: a review of preclinical and clinical research. Altern Med Rev. 2009;14(2):141–153. [PubMed] [Google Scholar]
- 63.Ghandadi M, Sahebkar A. Curcumin: an effective inhibitor of interleukin-6. Curr Pharm Des. 2017;23(6):921–931. doi: 10.2174/1381612822666161006151605. [DOI] [PubMed] [Google Scholar]
- 64.Hatcher H, Planalp R, Cho J, Torti FM, Torti SV. Curcumin: from ancient medicine to current clinical trials. Cell Mol Life Sci. 2008;65(11):1631–1652. doi: 10.1007/s00018-008-7452-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Derosa G, Maffioli P, Simental-Mendía LE, Bo S, Sahebkar A. Effect of curcumin on circulating interleukin-6 concentrations: a systematic review and meta-analysis of randomized controlled trials. Pharmacol Res. 2016;111:394–404. doi: 10.1016/j.phrs.2016.07.004. [DOI] [PubMed] [Google Scholar]
- 66.Jacob A, Wu R, Zhou M, Wang P. Mechanism of the Anti-inflammatory Effect of Curcumin: PPAR-γ Activation. PPAR Research 2007; Article ID: 89369. 5 pages. doi:10.1155/2007/89369. [DOI] [PMC free article] [PubMed]
- 67.Tang CH, Fu XJ, Xu XL, Wei XJ, Pan HS. The anti-inflammatory and anti-apoptotic effects of nesfatin-1 in the traumatic rat brain. Peptides. 2012;36(1):39–45. doi: 10.1016/j.peptides.2012.04.014. [DOI] [PubMed] [Google Scholar]
- 68.Kolgazi M, Cantali-Ozturk C, Deniz R, Ozdemir-Kumral ZN, Yuksel M, Sirvanci S, Yeğen BC. Nesfatin-1 alleviates gastric damage via direct antioxidant mechanisms. journal of surgical research. 2015;193(1):111–118. doi: 10.1016/j.jss.2014.06.057. [DOI] [PubMed] [Google Scholar]
- 69.Shirvani H, Ghanbari-Niaki A, Rahmati-Ahmadabad S. Effects of endurance training and herb supplementation on tissue nesfatin-1/nucleobindin-2 and ghrelin mRNA expression. International Journal of Applied Exercise Physiology. 2017;6(1):71–84. [Google Scholar]
- 70.Afrin R, Arumugam S, Rahman A, Wahed MI, Karuppagounder V, Harima M, Suzuki H, Miyashita S, Suzuki K, Yoneyama H, Ueno K. Curcumin ameliorates liver damage and progression of NASH in NASH-HCC mouse model possibly by modulating HMGB1-NF-κB translocation. Int Immunopharmacol. 2017;44:174–182. doi: 10.1016/j.intimp.2017.01.016. [DOI] [PubMed] [Google Scholar]
- 71.Bao W, Li K, Rong S, Yao P, Hao L, Ying C, Zhang X, Nussler A, Liu L. Curcumin alleviates ethanol-induced hepatocytes oxidative damage involving heme oxygenase-1 induction. J Ethnopharmacol. 2010;128(2):549–553. doi: 10.1016/j.jep.2010.01.029. [DOI] [PubMed] [Google Scholar]
- 72.Chi X, Yu D, Li P, Lu Q, Jiang W, Hao K. The protective effects of (1E, 6E)-1, 7-diphenylhepta-1, 6-diene-3, 5-dione, a curcumin analogue, against operative liver injury in rats. Eur J Pharm Sci. 2017;100:94–101. doi: 10.1016/j.ejps.2016.12.042. [DOI] [PubMed] [Google Scholar]
- 73.Guo C, Ma J, Zhong Q, Zhao M, Hu T, Chen T, Qiu L, Wen L. Curcumin improves alcoholic fatty liver by inhibiting fatty acid biosynthesis. Toxicol Appl Pharmacol. 2017;328:1–9. doi: 10.1016/j.taap.2017.05.001. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.


