Abstract
Regenerative endodontic procedures for immature permanent teeth with apical periodontitis confer biological advantages such as tooth homeostasis, enhanced immune defense system, and a functional pulp-dentin complex, in addition to clinical advantages such as the facilitation of root development. Currently, this procedure is recognized as a paradigm shift from restoration using materials to regenerate pulp-dentin tissues. Many studies have been conducted with regard to stem/progenitor cells, scaffolds, and biomolecules, associated with pulp tissue engineering. However, preclinical and clinical studies have evidently revealed several drawbacks in the current clinical approach to revascularization that may lead to unfavorable outcomes. Therefore, our review examines the challenges encountered under clinical conditions and summarizes current research findings in an attempt to provide direction for transition from basic research to clinical practice.
Keywords: Regenerative endodontics, pulp regeneration, revascularization, tissue engineering
Introduction
Endodontic treatment is a treatment modality that involves removal of contaminated tissue from teeth with necrotic pulp and replacement of the removed tissue with an inert material. This treatment has been known for many years as predictable and reliable. However, immature teeth with a necrotic pulp and apical periodontitis present multiple challenges to a successful outcome.1 They present large open apices along with divergent and thin root walls that are susceptible to fractures. Filling the root canal is a challenging task because the open apex does not provide a barrier which is able to stop the filling material. Apexification is a conventional treatment, specially indicated for the treatment of necrotic, immature permanent teeth, with demonstrated efficacy in achieving closure of the apex.2 However, it does not promote root development which is vital for preventing root fractures.
In 2004, Banchs and Trope3 published a case report, suggestive of a new treatment procedure for immature permanent teeth with apical periodontitis called “revascularization,” also referred to as regenerative endodontic therapy. Regenerative endodontic procedures (REPs) are defined as “biologically based procedures designed to replace damaged structures, including dentin and root structures, as well as cells of the pulp-dentin complex.”4 It is a new paradigm advocating the complete replacement of compromised tissue, based on tissue engineering rather than traditional restoration.
Since the first case report, a large volume of clinical studies and case reports have been published describing favorable clinical outcomes.5–9 Recently, however, it has been suggested that outcomes of REPs should be evaluated from a science-based aspect as well as from a patient-based or clinical aspect, and that these have indicated unfavorable and/or unexpected outcomes.10–13 Obviously, there is a gap between the clinical outcomes of the current protocol and the expected outcomes predicted by the research concept. This review presents the clinical protocol that is currently in use, its challenges and limitations, and details current research attempting to translate biological concepts into clinical practice.
Clinical procedure for pulp-dentin regeneration
The approach of regenerating dental pulp tissue by inducing blood into the root canal was first proposed by Ostby in the 1960s14 and then abandoned for over 20 years with no obvious outcome. In the 1970s, the understanding that revascularization, or reestablishment of a vascular network within the root canal, is essential for the completion of root development, came to the fore, having originated from traumatology.15,16 The term, revascularization, was then used in initial case reports on regenerative endodontic therapies.3,17 During the period of more than a decade that followed the first case report, various protocols for pulp-dentin regeneration were introduced. This new treatment modality was adopted by the American Dental Association in 2011. However, evidence-based guidelines that provide the most favorable results have not yet been fully established, but are being gradually developed, based on many clinical and basic research studies.
Differences from conventional endodontic treatment
Similar to conventional root canal therapy, the primary goal of REPs as an endodontic treatment is the resolution of apical periodontitis. However, there are certain differences in the basic concept and related procedures. First, REPs are originally applied to immature permanent teeth, with thin walls and wide-opened apices. Aggressive filing is performed for infection control in endodontic treatment. However, in REPs, mechanical debridement using endodontic files is contraindicated to avoid further weakening of the thin root canal wall and to protect the vitality of apical tissue stem cells.17,18 Instead, sufficient chemical disinfection using an irrigant and intracanal medicaments is proposed.
Second, disinfection in REPs should be performed with thorough consideration to cell cytotoxicity. Even though sterile environment may be achieved by sufficient disinfection, pulp tissue regeneration requires a balance between disinfection and the microenvironment necessary for cell viability, in order to induce stem cell survival and differentiation. Various concentrations of sodium hypochlorite (NaOCl), ranging from 0.5% to 6 %, have been used for disinfection.19 Recently, several studies have reported that concentrations of NaOCl higher than 3% may exhibit cytotoxicity toward stem cells of apical papilla (SCAP) and interfere with cell adhesion on the dentin surface.20–22 For these reasons, recent studies including clinical considerations of the American Association of Endodontists (AAE) recommend the use of lower concentration of NaOCl in REPs. In the same vein, the use of lower concentrations of triple antibiotic paste (TAP) or calcium hydroxide were recommended as intracanal medicaments.23
Finally, REPs utilize tissue engineering to form a pulp-dentin structure in the canal. Intracanal bleeding has a positive impact on the three requirements for tissue engineering: stem/progenitor cells, scaffolds, and growth factors.10,24,25 It is proposed that inducing bleeding into the canal results in the delivery of mesenchymal stem cells (MSCs) to the site.25 The blood clot that forms acts as the scaffold, as well as a rich source of growth factors that may play an important role in the regeneration process.10,26 Since a recent study reported that ethylenediaminetetraacetic acid (EDTA) solution may release various growth factors entrapped in dentin, thereby promoting differentiation of dental pulp stem cells (DPSCs) into odontoblast-like cells, EDTA has been recommended as the final irrigation.27
Clinical protocol: revascularization
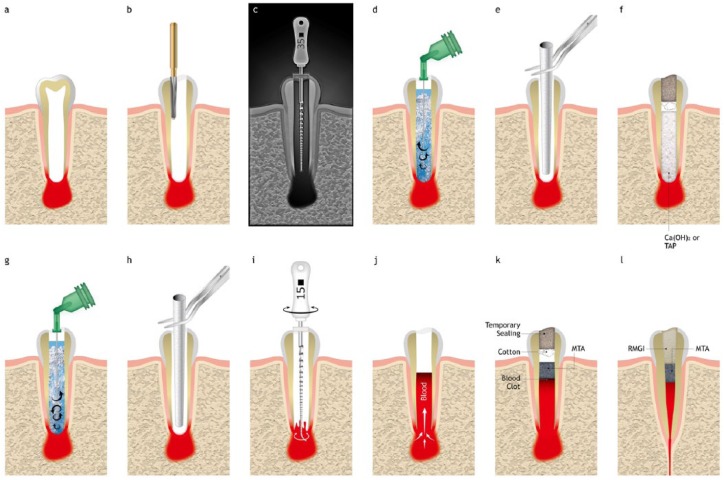
REP is performed within the principles of conventional endodontic treatments, but with some major changes, such as a disinfection process which totally relies on chemical irrigation (while taking into consideration cytotoxicity toward cells to be recruited for the canal), as well as the stimulation of pulp-dentin regeneration via bleeding induction. The currently recommended procedures are as follows (Figure 1).24,28
Figure 1.
Schematic illustration of revascularization procedure. Revascularization is considered for immature teeth with open apices, pulp necrosis, and apical periodontitis (a). After accessing the opening (b), gentle irrigation limited to coronal part of the chamber is performed. A radiograph with K-file insertion (c) provides the approximate tooth length, which helps to determine a working length. Low concentration of NaOCl (1.5 or less than 3%, 20 mL/canal, 5 min) is used for disinfection (d), following which saline or 17% EDTA is used. After copious irrigation and canal drying with paper point (e), intracanal medicaments, such as Ca(OH)2 or TAP were placed, and covered with temporary filling material (f). After confirming the absence of any signs of infection, the final step is initiated. Final irrigation is performed with sterile saline and 17% EDTA (g). After the canal has dried (h), a pre-curved K-file is introduced 2 mm past the apical foramen and rotated to induce bleeding (i). Blood fills the canal from the bottom and the blood clot can be identified after 15 min (j). After the blood clot is confirmed, capping materials such as MTA are placed over the blood clot (k). Regeneration of pulp-dentin leads to root development with thickening, lengthening, and apical closure, as well as maintenance of tooth vitality (l).
First visit
All visits, except for the final visit, are designed with a focus on disinfection of the root canal. After local anesthesia, rubber dam isolation, and access, gentle irrigation which is limited to the coronal part of the pulp chamber is performed. It is recommended that the canal be inspected using a dental microscope to confirm the presence of residual vital tissue and the level to which it is present.24 A K-file, or alternatively a gutta-percha cone, should be introduced into the canal to establish a working length.11,29 In case when inserting a file into the canal, a little resistance caused by viable tissue or pain sensation is reported, a file should not progress deeper. A file could be fixed with wax or cotton pellet during radiographs taken (Figure 1(c)).
Removal of necrotic tissue and the disinfection of the canal were accomplished by gently irrigating the canal with a minimum 20 mL NaOCl (Figure 1(d)). Lower concentrations of NaOCl are recommended (1.5 or under 3%, 20 mL/canal, 5 min).20,30 The canal is then irrigated with saline or 17% EDTA (20 mL/canal, 5 min). The needle should be positioned at a point 2 mm short of the apical foramen in order to minimize cytotoxicity to apical tissues. Negative pressure irrigation procedures such as EndoVac (Discus Dental, Culver city, CA) may be considered.31 Mechanical debridement is contraindicated so as not to weaken the root wall.
After the canal is disinfected with copious irrigation and dried with paper points (Figure 1(e)), it is recommended to place intracanal medicaments, either calcium hydroxide (Ca(OH)2) or TAP using a lentulo spiral or syringe. Treatment with TAP (a 1:1:1 volume combination of ciprofloxacin, metronidazole, and minocycline) at a low concentration (0.1–1.0 mg/mL) is recommended to lower cytotoxicity toward stem/progenitor cells. Double antibiotic paste (DAP) without minocycline may be considered if avoidance of tooth discoloration is desired. In order to prevent coronal leakage of bacteria, a sterile cotton pellet may be placed over the medicaments and the pellet covered with 3–4 mm of temporary filling material, such as Cavit (3M ESPE, St Paul, MN), IRM, and glass ionomer.
Interim visit
The patients could be recalled within a time interval of 1–4 weeks. If clinical signs and symptoms persist, the disinfection procedures implemented during the first visit should be repeated.
Final visit
After confirming that signs of persistent infection are absent, the tooth is anesthetized with 3% mepivacaine without vasoconstrictor, and the temporary restoration removed following rubber dam isolation. Copious and gentle irrigation with sterile saline or 17% EDTA should be repeated until no medicament is evident in the canal (Figure 1(g)). After the canal is dried with paper points, bleeding is induced by over-instrumenting with K-file. A pre-curved K-file is introduced 2 mm past the apical foramen and rotated to induce bleeding below a point approximately 3 mm apical to the cemento-enamel junction (CEJ; Figure 1(i) and (j)). The time estimated for blood clot formation is 15 min.3,29,32 The stability of the blood clot could be confirmed using the reverse side of a paper point. An alternative method of inducing a blood clot is the use of platelet-rich plasma (PRP) or platelet-rich fibrin (PRF). After stability of the blood clot is confirmed, mineral trioxide aggregate (MTA) cement is placed over the clot as capping material. A 3–4 mm layer of MTA is recommended. In order to minimize apical displacement of MTA, resorbable matrix, such as collagen matrix, may be placed over the blood clot. In case MTA with a long setting time is used, final restoration is performed during the next visit.
Clinical issues and limitation
Most studies analyzing revascularization have reported favorable outcomes with apical healing, mineralized tissue apposition, and continued root development of varying degrees (Figure 1(l)).5–7,26,33 However, some recent studies have reported drawbacks and unfavorable outcomes resulting from recurrence of periapical lesions, absence of continued root formation, and partial or complete intracanal obliteration.10,12,34,35 There are three issues relating to the current clinical protocol with regard to unfavorable outcome and those reasons are as follows: (1) insufficient bleeding,7,12,36 (2) incomplete disinfection,34,37,38 and (3) ectopic tissue formation instead of pulp-dentin regeneration.39–41
Because mesenchymal stem/progenitor cells are delivered into the root canal space only after bleeding is induced, bleeding induction may play a critical role in successful pulp-dentin regeneration.25 Therefore, utilization of anesthesia without vasoconstrictors is recommended to facilitate bleeding. However, some reports point to difficulties encountered in bleeding induction, such as insufficient bleeding despite following protocol. Such difficulties could be related to the absence of root development or poor root development due to a lack of recruited cells.7,8,36 Therefore, it may not be prudent to rely on bleeding induction to produce the predicted outcome.
Unlike conventional root canal therapy, current REPs suggest low concentrations of irrigation and intracanal medication for disinfection (e.g. 1.5% of NaOCl and 0.1–1 mg/mL of TAP). In addition, mechanical debridement is contraindicated to avoid weakening the root wall and protect viable tissue in the canal. However, once a microbial infection is well established in the root canal system, it is well known that complete elimination is difficult, even with mechanical instrumentation and stronger concentrations of irrigation.42–44 Complete disinfection is a major factor required for successful endodontic treatment. Therefore, a protocol which relies on thorough chemical disinfection using low concentrations of irrigants and medicaments may be questionable. Several studies have indicated that failed revascularization, with the presence of apical periodontitis and absence of root development, may be due to insufficiency of the disinfection levels, recommended by the current protocol.34,38,40 Lin et al.45 and Zizka et al.46 reported that deposition of hard tissue or maturation of roots continued to occur despite persistent apical periodontitis in failed REPs. It appears that root development is dependent on the condition of the Hertwig epithelial sheath rather than pulp regeneration or the bacterial load in the root canal system.47 Current REPs mainly focus on achieving root maturation by protecting the viability and adherence of stem cells, which may lead to the failure of achieving even the primary objective of REPs, which is the resolution of apical periodontitis. Therefore, the disinfection protocol of REPs should be carefully re-evaluated, in order to achieve a proper balance between disinfection of the root canal system and survival of the stem/progenitor cells, which may lead to a successful outcome in the long term.
Despite the clinical success as evidenced by the healing of apical periodontitis, continued root development, and in certain cases, vitality responses, it has been questioned whether REPs result in true regeneration of pulp-dentin-like tissue. Several preclinical studies reported apposition of osteoid-/cementoid-like tissue lacking an organized pulp-dentin complex.32,48,49 Histology reports of human teeth have confirmed that canals are filled with ectopic bone, fibrous tissue, and cement apposition up to the inner wall of the root.39,40,44 If recruited cells originate from the periodontal ligament (PDL), cementum, or alveolar bone, they would generate the same tissue that they originated from.50,51 However, there are a few instances where newly formed dentin-like and pulp-like tissue is evident in the canal.52,53 All these cases are diagnosed with reversible or irreversible pulpitis, where dental pulp progenitor cells or stem cells derived from the remaining vital pulp and apical papilla remained. Similarly, several studies have demonstrated promising outcomes related to regeneration of pulp-dentin-like tissue using transplantation with DPSCs or SCAP in vivo.54,55 However, currently, cell transplantation is not feasible in a clinical environment. Currently, revascularization appears to be the best practice based on tissue engineering for pulp-dentin complex. However, there may yet be room for improvement and much basic research is in progress with the objective of translating biological concepts into clinical practice.
Current approaches for pulp-dentin regeneration
Similar to general tissue engineering, pulp tissue engineering aimed at regenerating the pulp-dentin complex also requires three essential factors: SCAPs, scaffolds, and signaling molecules. It is speculated that the ability of the tissues or organs to regenerate is dependent on the availability of resident SCAPs and microenvironmental cues.56–59
Stem/progenitor cells
Many studies have been conducted on stem/progenitor cell sources with the ability to regenerate the pulp-dentin complex. The issue of whether odontoblast-like cells that differentiate from heterogeneous stem cell populations have the same biological activities remains unresolved. Currently, MSCs of pulp origin are considered to have the highest potential for differentiating into odontoblast-like cells.60–62 Seo et al.50 reported that MSCs from pulp, PDL, and bone marrow generate the same tissues as their origins, which supports the view that pulp-dentin regeneration would require pulpal MSCs present in the root canal. In addition to DPSCs, SCAPs and stem cells from human exfoliated deciduous teeth (SHED) are known as potential cell sources for regeneration of the pulp-dentin complex.63–66
Considering the fact that the availability and quality of dental pulp tissue decline sharply with age, nonodontogenic stem cells have been investigated as alternative sources. The generation of induced pluripotent stem cells (iPSCs) is considered a groundbreaking development that revolutionized regenerative medicine. This is achieved by reprograming adult somatic cells or terminally differentiated cells, back to a pluripotent state via overexpression of four defined transcription factors.67–69 Analogous, but superior, to embryonic stem cells, patient-specific iPSCs can give rise to all cell lineages in the body, circumventing the clinical barrier of immunological rejection or ethical controversy.68 An efficient induction protocol has been developed recently to facilitate the differentiation of murine iPSCs (miPSCs) into neural crest-like cells (NCLCs) in vitro.70 These NCLCs further demonstrated their potential to differentiate into dental mesenchymal cells including odontoblasts upon co-culture with mouse dental epithelium. Despite that, not all of the reconstituted tooth germs produced perfect tissue-engineered tooth-like structures. These interesting findings demonstrate much potential for iPSCs in future regenerative dentistry research.
Oh et al. developed a method to generate MSCs from primary normal human epidermal keratinocytes (NHEKs) by inducing epithelial–mesenchymal transition (EMT). They coined the term induced MSCs (iMSCs) to describe these cells, which are distinct from iPSCs.51 Clinically, these unique cell populations may represent an alternative source of autologous MSCs for patients who lack adequate availability of tissue for MSCs, since iMSCs are easily obtained from skin tissue. Further investigation may elucidate the potency of these iMSCs and assess their capacity for trans-differentiation into functional odontoblasts when transplanted in the root canal microenvironment.
Microenvironment
Some studies have suggested that the fate of transplanted stem/progenitor cells might be site-associated rather than origin-associated.59 In this respect, revascularization may have to consider providing bioengineered microenvironments, using three-dimensional scaffold, and chemotactic growth factors.
Scaffolds
Functionally, scaffolds provide a solid environment for stem cells and signaling molecules. In regenerative endodontics, an ideal scaffold should be biodegradable and deliver cells containing growth factors. Various types of scaffolds such as natural, or synthetic, polymers are being studied and reported with varying results (Table 1).87
Table 1.
Summary of studies on the biological effects of scaffolds. Biologic effect of each scaffold was organized according to experimental design, for example, in vitro, in vivo using cell-based or cell-homing approaches, and/or clinical study.
| Type | Scaffold | Authors | Methods | Conclusion |
|---|---|---|---|---|
| Natural | Collagen matrix | Nakashima71 | In vivo (animal) Cell homing |
BMP-2 and 4 induce osteodentin formation if combined with collagen matrix. |
| Kwon et al.72 | In vitro | Collagen scaffold has beneficial effects on proliferation and differentiation of hDPCs. | ||
| Chitosan | Farea et al.73 | In vitro | Combined with chitosan scaffold and TGF-β1 in conjunction with SHED might be beneficial for bone regeneration. | |
| PRP | Torabinejad et al.74 | In vivo (animal) Cell homing |
PRP or blood clots as scaffolds result in the in-growth of bone-like, cementum-like, and connective tissue in the apical third of the roots. | |
| Rodríguez-Benítez et al.75 | In vivo (animal) Cell homing |
PRP scaffold improves the success rate of the revascularization procedure. | ||
| Bezgin et al.76 | Clinical Cell homing |
Treatment outcomes did not differ significantly between PRP and conventional scaffold. | ||
| Zhu et al.77 | In vivo (animal) cell based | Tissues formed using PRP scaffolds were not pulp tissues but mainly periodontal tissues. | ||
| Martin et al.40 | Clinical Cell homing |
Regardless of PRP treatment, cementoid/osteoid tissues and uninflamed fibrous connective tissues were formed. | ||
| Torabinejad and Turman33 | Clinical Cell homing |
PRP is potentially an ideal scaffold for regenerative endodontics. | ||
| Torabinejad et al.78 | In vivo (animal) Cell homing |
Either PRP or blood clots form bonelike tissue without continual root maturation. | ||
| PRF | Chen et al.79 | In vitro | PRF scaffolds serve as a potential therapy in regenerative endodontics. | |
| He et al.80 | In vitro | PRF released the maximum quantity of growth factors. | ||
| Woo et al.81 | In vitro | Combination of MTA and PRF has a synergistic effect on odontoblastic differentiation of hDPCs. | ||
| PRP, PRF | Shivashankar et al.82 | Clinical Cell homing |
PRP was better than PRF in periapical wound healing when used in regenerative procedures. | |
| PRGF | Dianat et al.83 | In vivo (animal) Cell homing |
PRGF scaffolds showed no advantage over blood clots regarding regenerative healing. | |
| Synthetic | PLGA | Chen et al.84 | In vitro | PLGA/gelatin electrospun sheet made up a microenvironment for tooth root regeneration. |
| PLLA | Cordeiro et al.65 | In vitro and clinical Cell homing |
SHED with PLLA scaffold suggested viable source of stem cells for dental pulp tissue engineering. | |
| β-tricalcium phosphate/chitosan | Liao et al.85 | In vitro | Porous beta-tricalcium phosphate/chitosan composite scaffolds upregulated expressions of ALP and OPN. | |
| Biphasic calcium phosphate | AbdulQader et al.86 | In vitro | Biphasic calcium phosphate scaffolds support hDPC differentiation for dentin tissue regeneration. |
ALP: alkaline phosphatase; BMP: bone morphogenetic protein; hDPCs: human dental pulp cells; MTA: mineral trioxide aggregate; OPN: osteopontin; PLGA: poly(d,l-lactide-co-glycolide); PLLA: poly(l-lactic acid); PRGF: plasma rich in growth factors; PRF: platelet-rich fibrin; PRP: platelet-rich plasma; SHED: stem cells from human exfoliated deciduous teeth; TGF: transforming growth factor.
Collagen scaffold
Animal-derived and recombinant collagens, especially type I, are known to be the most useful biomaterials available for tissue engineering, cosmetic surgery, and drug delivery systems.88 Collagen scaffolds are used either in native fibrillar forms or in denaturized forms such as sponges, sheets, plugs, and pellets.88 Kwon et al.72 showed that cross-linking of collagen scaffolds has beneficial effects in the attachment, proliferation, and differentiation of human dental pulp cells (hDPCs). In an animal-based study, Nakashima71 showed that teeth-implanted growth factors and collagen matrix scaffolds induced osteodentin.
PRP
PRP, an autologous source of blood, contains elevated platelet concentrations, growth factors, and cytokines that enhance wound healing, attract pulp stem cells, and promote SCAP proliferation.89 Clinical studies have indicated that PRP enables a higher percentage of hard tissue deposition compared to blood clot scaffold, while other studies showed no advantage over blood clots regarding periapical healing, apical closure, or new tissue formation.74–76,83 Despite these contrasting results, it is evident that tissue regenerated using either PRP or blood clots as scaffolds is a bone-like tissue without continual root maturation.78 Martin et al.40 concluded that PRP enhanced wound healing only if parenchymal tissue has not been completely destroyed, but otherwise did not induce tissue regeneration. The advantages of using PRP scaffold are ease of application and the shorter time taken to induce vital tissues within the root canal. However, as special equipment and medications are required to prepare PRP, the cost of treatment may increase.33
PRF
PRF, a second-generation platelet concentrate, contains a multitude of growth factors and exhibits cell differentiation properties, in addition to degrading quickly. PRF is not only a scaffold for cell adhesion and migration but also a growth factor for dental pulp cells.79 The intracanal transplantation of DPSCs with PRF might serve as potential therapy for regenerative endodontics, pulp revitalization, or revascularization.79 He et al.80 introduced a new method, which involves adding hDPC suspension before centrifugation and demonstrated that PRF may play a synergistic role with dentin matrix in the formation of odontoblast cells when used as scaffold. MTA used in revascularization also had a synergic effect with PRF when compared to that with dental pulp cells cultured with MTA or PRF individually.81 However, a study has also indicated that PRP performed better than PRF and blood clot in periapical wound healing when used in REPs.82
Synthetic polymers
Synthetic polymers are easily manufactured from a wide range of biodegradable polymers and are susceptible to modification, whereas their degradation is easily controlled.88 Scaffold from biodegradable polymers serves as a temporary supporting structure for growing cells and tissues prior to degradation.90 Poly(d,l-lactide-co-glycolide) (PLGA) with gelatin scaffold promoted endodontic regeneration via simulation of extracellular matrix environments of stem cells.84 Incorporation of gelatin into PLGA-based scaffolds alleviated inflammatory reactions initiated by acidic degradation products.84 PLGA scaffold with simulated microgravity groups upregulated a broad range of integrins and may have contributed to regulating the proliferation, migration, and adhesion of hDPCs.91
Inorganic scaffolds
Inorganic regenerative scaffold materials include calcium alginate, demineralized bone matrix (DBM), and mostly calcium phosphate.88 Calcium phosphate scaffolds have been widely and successfully used with osteoblast cells for bone tissue regeneration. The biological properties of this porous structure are determined by pore shape and size, porosity percentage, and the pore interconnection pathway.92 Microporosity under 10 μm diameter permits body-fluid circulation, while macroporosity over 100 μm is prerequisite for migration and proliferation of cells and tissue formation.93 It is reported that a pore size of 300 μm was adequate for dental pulp–derived cells to align on the surface and regenerate dentin-like tissue.94 Biphasic calcium phosphate(BCP), an intimate mixture of more stable phase hydroxyapatite (HA) and more soluble phase beta-tricalcium phosphate (β-TCP), is known as an appropriate material for endodontic regeneration as it resembles their inorganic part.86 AbdulQader et al.86 showed that BCP scaffold of 65% porosity can support hDPC differentiation for dentin tissue regeneration and concluded that a high rate of degradation of BCP with 65% porosity produced a localized alkaline, calcium-, and phosphate-rich environment that is optimal for odontoblast differentiation.
Biomolecules
Combined with scaffolds, biomolecules (BM) that are released from demineralized dentin matrix or delivered exogenously have been found to play an important role in pulp revascularization by forming favorable microenvironments.95 They are thought to recruit endogenous cells by chemotactic effects or induce differentiation of recruited cells in order to initiate dental pulp regeneration. Numerous studies using BM, either singularly or in combination, have reported various aspects of its function (Table 2).
Table 2.
Summary of the studies on the function of bioactive molecules. It was organized according to the experimental design, for example, in vitro or in vivo using cell-based or cell-homing approaches.
| Category | Bioactive molecule | Authors | Methods | Conclusion |
|---|---|---|---|---|
| TGF | TGF-β1 and BMP-2 | Begue-Kirn et al.96 | In vivo (animal) Cell homing |
TGF-β1 and BMP-2 stimulated odontoblast differentiation. |
| TGF-β3 | Huojia et al.97 | In vitro | TGF-β3 promoted mRNA expression, and increased protein levels of osteocalcin and type I collagen in dental pulp cells. | |
| TGF-β superfamily | Liu et al.98 | In vitro | TGF-β-related gene profiles are reported to be associated with odontoblast-like differentiation and mineralization. | |
| BMP | BMP-2 | Iohara et al.99 | In vivo (animal) Cell homing |
BMP-2 can direct pulp progenitor/stem cell differentiation into odontoblasts and result in dentin formation. |
| BMP-4 | About et al.100 | In vitro | BMP-4 is involved in nestin up-regulation which promotes odontoblast differentiation during normal and pathological conditions. | |
| Blood derived | PDGF alpha | Morrison-Graham et al.101 | In vitro | Functional PDGFR alpha can affect crest development both directly and indirectly. |
| PDGF, IGF-1, aFGF, and IGF-2 | Nakashima102 | In vitro | Proliferation of pulp cells may be stimulated mainly by PDGF and IGF-1. Production of extracellular matrix proteoglycan may be enhanced by aFGF, IGF-1, IGF-2, TGF-β, and PDGF. |
|
| PDGF | Yokose et al.103 | In vitro | PDGF exerts diverse effects on odontoblastic differentiation and acts in dentinogenesis during the repair process of damaged dental pulp. | |
| FGF, VEGF, PDGF, and NGF | Kim et al.104 | In vivo (animal) Cell homing |
Delivery of bFGF, VEGF, or PDGF with NGF and BMP-7 has potent cell-homing effects for recellularization and revascularization. | |
| SDF-1, bFGF, and PDGF | Yang et al.105 | In vitro | SDF-1alpha, bFGF, PDGF, SCF, and G-CSF could achieve pulp-like tissue formation via a cell-homing strategy. | |
| VEGF and BMP-2 | Aksel and Huang106 | In vitro | Both VEGF and BMP-2 enhances odonto/osteogenic differentiation of DPSCs. | |
| VEGF and FGF-2 | Mullane et al.107 | In vitro | VEGF and FGF-2 enhanced neovascularization of severed human dental pulps. | |
| General | IGF-1 | Feng et al.108 | In vitro | IGF-1 could promote proliferation and osteogenic differentiation of DPSCs. |
| IGF-1 | Wang et al.109 | In vitro | IGF-1 plays an important role in the regulation of tooth root development. | |
| Neurotrophic | BDNF | De Almeida et al.110 | In vitro | SCAP release BDNF in a concentration-dependent manner and trigger directed axonal targeting. |
aFGF: acidic fibroblast growth factor; BDNF: brain-derived neurotrophic factor; bFGF: basic fibroblast growth factor; BMP: bone morphogenetic protein; DPSC: dental pulp stem cell; FGF: fibroblast growth factor; G-CSF: granulocyte colony-stimulating factor; IGF: insulin growth factor; NGF: nerve growth factor; PDGF: platelet-derived growth factor; SCAP: stem cells from apical papilla; SCF: stem cell factor; SDF: stromal cell-derived factor; TGF: transforming growth factor; VEGF: vascular endothelial growth factor.
Transforming growth factor-β superfamily and bone morphogenetic protein family
A key family of growth factors, which have been identified in the dentin matrix, are members of the transforming growth factor-β (TGF-β) family of growth factors. The TGF-β family comprises a group of diverse growth factors including TGF-β, bone morphogenetic protein (BMP), growth and differentiation factors (GDFs), and anti-Mullerian hormone (AMH). TGF-β1, one of the three isoforms of TGF-β, is involved in primary odontoblastic differentiation and promoting tertiary dentin generation together with the regulation of dentin extracellular matrix synthesis, cell growth, cell proliferation, cell differentiation, and cell apoptosis.111 In addition, TGF-β3 induced ectopic mineralization in dental pulp during tooth germ development of fetal mouse and increased osteocalcin and type 1 collagen levels.97 Begue-kirn et al.96 reported that TGF-β-like molecules may interact with a component that acts as a regulator of its activity in initiating functional differentiation of odontoblasts. BMPs, a subgroup of the TGF-β superfamily, are involved in many biological activities including cell proliferation, differentiation, and apoptosis. BMPs are known to have strong osteoinductive and chondrogenic effects. BMP-2 stimulates the differentiation of dental pulp stem/progenitor cells into odontoblasts in vivo and in vitro, and it induces dentin sialophosphoprotein (DSPP) expressions to enhance the angiogenic potential of DPSCs.106 It also increases alkaline phosphatase activity and stimulates reparative dentin formation.98
Blood-derived growth factors
Blood-derived growth factors such as platelet-derived growth factor (PDGF), TGF-β, fibroblast growth factor (FGF), vascular endothelial growth factor (VEGF), and insulin growth factor (IGF) are found in blood clots produced by bleeding induction. VEGF is regarded as a dominant signaling protein involved in lymphangiogenesis, vasculogenesis (de novo formation of the embryonic circulatory system), and angiogenesis (growth of blood vessels from pre-existing vasculature).112 It has been shown to promote blood vessel formation enhancing neovascularization in vivo.107 PDGF, a potent mitogen for cells of mesenchymal origin, consists of a family of polypeptides (A, B, C, and D) that are placenta growth factors.113 It promotes angiogenesis and regulates the process of odontoblastic differentiation, acting synergistically with other growth factors.114 Kim et al.104 delivered combinatory cytokines of VEGF, bFGF, or PDGF with basal nerve growth factor (NGF) and BMP-7 into the root canal and suggested that chemotaxis-induced cell homing is sufficient for the regeneration of pulp-dentin regeneration leading to a complete fill of dental-pulp-like tissue.
General growth factors
IGF-I is a multifunctional peptide, which promotes osteogenic proliferation and differentiation of DPSCs108 and SCAPs into a mineralizing phenotype,109 and plays a role in the promotion of cell proliferation and the inhibition of cell death (apoptosis).115 It is required for achieving maximal growth during development. IGF-I is a key regulator of bone formation and mineralization,116 where it stimulates osteoblastic cells in culture to proliferate and synthesize bone matrix proteins and also stimulates mRNA expression for alkaline phosphatase.117
Neurotrophic factors
Although its exact role is yet to be clarified, brain-derived neurotrophic factor (BDNF) is expressed in the vascular wall and changes vascular function and biology.118 It is related to angiogenesis, nitric oxide production, and capillary density. De Almeida et al. performed human SCAPs co-culturing of rat-derived trigeminal neurons and confirmed that NGF, glial-derived neurotrophic factor (GDNF), and BDNF show potency in SCAP-mediated neurite outgrowth. BDNF, especially, was the critical factor among the neutrophins.110
Direction and strategy of research
The AAE recognized the tremendous future potential in regenerative endodontics and invested US$2.5 million in a multi-year grant to support clinical research in regeneration. In addition to this trend, a large volume of basic research has also been carried out at a global level. Various research studies have been conducted on scaffolds and BM as well as on stem/progenitor cells to elucidate the right microenvironment. These findings may suggest directions to be followed in order to obtain ideal pulp-dentin regeneration. Despite such accomplishments, many hurdles have to be overcome in the translation of research results into clinical practice. Many basic studies have produced outstanding data using cell-based approaches. However, cell-based approaches are still faced with practical challenges in a clinical environment such as time constraints, cost, and safety issues, and therefore using such findings in clinical practice has not been feasible as of yet. Like in other fields of tissue engineering, there is also a consideration that advanced technologies such as three-dimensional cell cultures, three-dimensional cell printing, and gene therapy could be applied for pulp-dentin regeneration.4 These advanced technologies could lead to more ideal pulp-dentin regeneration. However, more active research is required to generate predictable outcomes with these technologies.
In the short term, it would be most helpful to promote research studies that may be applied practically in the current clinical environment, such as the study supporting cell-homing approach with delivery of BM and scaffolds and the study using minced pulp as a source of pulpal MSCs.119 In addition, even with positive research findings, ideal methods that are difficult to apply in a clinic situation should only be considered as a research for research’s sake. Over the past 20 years, there has been a lot of research conducted; however, recently, most of the revisions to the revascularization protocol have been concerned with the concentration of intracanal medication. This might be because there is a limited connection between the basic research and the actual clinic. We hope that this review will serve as an opportunity to introduce researchers to the clinical environment and limitations and hope that more research will be conducted that can be applied within the clinic.
Conclusion
Pulp-dentin regeneration based on tissue engineering was performed under clinical conditions, using the revascularization protocol. This protocol is recognized as a paradigm shift from the formerly used method of restoration with materials. Lately, it has been considered as a treatment option for mature teeth as well as immature teeth.120 However, there is a broad consent that the final tissue acquired by REPs is more likely to bone-like tissue mixed with connective tissue rather than the pulp-dentin complex. A controversy has surfaced over the terms “pulp-dentin regeneration,” “pulpal repair,” and “wound healing.” The current protocol, revascularization, based on the cell-homing approach is considered as one possibility for generating ectopic mineralized tissue formation. It also revealed the limitations such as unpredictability in bleeding induction and imbalance between complete disinfection and stem/progenitor cell viability. Nevertheless, it is evident that pulp-dentin regeneration has biological advantages such as tooth homeostasis, an enhanced immune defense system, and a functional pulp-dentin complex, in addition to the clinical advantages of promoting root development. Understanding current clinical approach and challenges and encouraging applicable research studies may help develop regenerative procedures with more predictable and ideal outcomes.
Footnotes
Declaration of conflicting interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by a National Research Foundation of Korea (NRF) grant funded by the Korea government [NRF-2017R1D1A3B03031966] and Global Research Development Center Program through the National Research Foundation of Korea(NRF) funded by the Ministry of Science and ICT(MSIT)[NRF-2018K1A4A3A01064257].
ORCID iDs: Chanyong Jung  https://orcid.org/0000-0002-6533-4787
https://orcid.org/0000-0002-6533-4787
Minju Song  https://orcid.org/0000-0001-7586-2359
https://orcid.org/0000-0001-7586-2359
References
- 1. Trope M. Treatment of the immature tooth with a non-vital pulp and apical periodontitis. Dent Clin North Am 2010; 54: 313–324. [DOI] [PubMed] [Google Scholar]
- 2. Torabinejad M, Nosrat A, Verma P, et al. Regenerative endodontic treatment or mineral trioxide aggregate apical plug in teeth with necrotic pulps and open apices: a systematic review and meta-analysis. J Endod 2017; 43: 1806–1820. [DOI] [PubMed] [Google Scholar]
- 3. Banchs F, Trope M. Revascularization of immature permanent teeth with apical periodontitis: new treatment protocol? J Endod 2004; 30: 196–200. [DOI] [PubMed] [Google Scholar]
- 4. Murray PE, Garcia-Godoy F, Hargreaves KM. Regenerative endodontics: a review of current status and a call for action. J Endod 2007; 33: 377–390. [DOI] [PubMed] [Google Scholar]
- 5. Bose R, Nummikoski P, Hargreaves K. A retrospective evaluation of radiographic outcomes in immature teeth with necrotic root canal systems treated with regenerative endodontic procedures. J Endod 2009; 35: 1343–1349. [DOI] [PubMed] [Google Scholar]
- 6. Jeeruphan T, Jantarat J, Yanpiset K, et al. Mahidol study 1: comparison of radiographic and survival outcomes of immature teeth treated with either regenerative endodontic or apexification methods: a retrospective study. J Endod 2012; 38: 1330–1336. [DOI] [PubMed] [Google Scholar]
- 7. Ding RY, Cheung GS, Chen J, et al. Pulp revascularization of immature teeth with apical periodontitis: a clinical study. J Endod 2009; 35: 745–749. [DOI] [PubMed] [Google Scholar]
- 8. Cehreli ZC, Isbitiren B, Sara S, et al. Regenerative endodontic treatment (revascularization) of immature necrotic molars medicated with calcium hydroxide: a case series. J Endod 2011; 37: 1327–1330. [DOI] [PubMed] [Google Scholar]
- 9. Gelman R, Park H. Pulp revascularization in an immature necrotic tooth: a case report. Pediatr Dent 2012; 34: 496–499. [PubMed] [Google Scholar]
- 10. Nosrat A, Homayounfar N, Oloomi K. Drawbacks and unfavorable outcomes of regenerative endodontic treatments of necrotic immature teeth: a literature review and report of a case. J Endod 2012; 38: 1428–1434. [DOI] [PubMed] [Google Scholar]
- 11. Chen MYH, Chen KL, Chen CA, et al. Responses of immature permanent teeth with infected necrotic pulp tissue and apical periodontitis/abscess to revascularization procedures. Int Endod J 2012; 45: 294–305. [DOI] [PubMed] [Google Scholar]
- 12. Song M, Cao Y, Shin S-J, et al. Revascularization-associated intracanal calcification: assessment of prevalence and contributing factors. J Endod 2017; 43: 2025–2033. [DOI] [PubMed] [Google Scholar]
- 13. Diogenes A, Ruparel NB, Shiloah Y, et al. Regenerative endodontics: a way forward. J Am Dent Assoc 2016; 147: 372–380. [DOI] [PubMed] [Google Scholar]
- 14. Ostby BN. The role of the blood clot in endodontic therapy. An experimental histologic study. Acta Odontol Scand 1961; 19: 324–353. [PubMed] [Google Scholar]
- 15. Galler KM. Clinical procedures for revitalization: current knowledge and considerations. Int Endod J 2016; 49: 929–936. [DOI] [PubMed] [Google Scholar]
- 16. Skoglund A, Tronstad L, Wallenius K. A microangiographic study of vascular changes in replanted and autotransplanted teeth of young dogs. Oral Surg Oral Med Oral Pathol 1978; 45: 17–28. [DOI] [PubMed] [Google Scholar]
- 17. Iwaya SI, Ikawa M, Kubota M. Revascularization of an immature permanent tooth with apical periodontitis and sinus tract. Dent Traumatol 2001; 17: 185–187. [DOI] [PubMed] [Google Scholar]
- 18. Cvek M. Prognosis of luxated non-vital maxillary incisors treated with calcium hydroxide and filled with gutta-percha. A retrospective clinical study. Endod Dent Traumatol 1992; 8: 45–55. [DOI] [PubMed] [Google Scholar]
- 19. Kontakiotis EG, Filippatos CG, Tzanetakis GN, et al. Regenerative endodontic therapy: a data analysis of clinical protocols. J Endod 2015; 41: 146–154. [DOI] [PubMed] [Google Scholar]
- 20. Martin DE, De Almeida JF, Henry MA, et al. Concentration-dependent effect of sodium hypochlorite on stem cells of apical papilla survival and differentiation. J Endod 2014; 40: 51–55. [DOI] [PubMed] [Google Scholar]
- 21. Chang YC, Huang FM, Tai KW, et al. The effect of sodium hypochlorite and chlorhexidine on cultured human periodontal ligament cells. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2001; 92: 446–450. [DOI] [PubMed] [Google Scholar]
- 22. Ring KC, Murray PE, Namerow KN, et al. The comparison of the effect of endodontic irrigation on cell adherence to root canal dentin. J Endod 2008; 34: 1474–1479. [DOI] [PubMed] [Google Scholar]
- 23. Ruparel NB, Teixeira FB, Ferraz CC, et al. Direct effect of intracanal medicaments on survival of stem cells of the apical papilla. J Endod 2012; 38: 1372–1375. [DOI] [PubMed] [Google Scholar]
- 24. Wigler R, Kaufman AY, Lin S, et al. Revascularization: a treatment for permanent teeth with necrotic pulp and incomplete root development. J Endod 2013; 39: 319–326. [DOI] [PubMed] [Google Scholar]
- 25. Lovelace TW, Henry MA, Hargreaves KM, et al. Evaluation of the delivery of mesenchymal stem cells into the root canal space of necrotic immature teeth after clinical regenerative endodontic procedure. J Endod 2011; 37: 133–138. [DOI] [PubMed] [Google Scholar]
- 26. Shah N, Logani A, Bhaskar U, et al. Efficacy of revascularization to induce apexification/apexogensis in infected, nonvital, immature teeth: a pilot clinical study. J Endod 2008; 34: 919–925; discussion 1157. [DOI] [PubMed] [Google Scholar]
- 27. Galler KM, D’Souza RN, Federlin M, et al. Dentin conditioning codetermines cell fate in regenerative endodontics. J Endod 2011; 37: 1536–1541. [DOI] [PubMed] [Google Scholar]
- 28. American Association of Endodontics. American Association of Endodontics clinical considerations for a regenerative procedure, https://www.aae.org/specialty/wp-content/uploads/sites/2/2017/06/currentregenerativeendodonticconsiderations.pdf (2016, accessed 27 May 2018).
- 29. Reynolds K, Johnson JD, Cohenca N. Pulp revascularization of necrotic bilateral bicuspids using a modified novel technique to eliminate potential coronal discolouration: a case report. Int Endod J 2009; 42: 84–92. [DOI] [PubMed] [Google Scholar]
- 30. Siqueira JF, Jr, Rocas IN, Favieri A, et al. Chemomechanical reduction of the bacterial population in the root canal after instrumentation and irrigation with 1%, 2.5%, and 5.25% sodium hypochlorite. J Endod 2000; 26: 331–334. [DOI] [PubMed] [Google Scholar]
- 31. da Silva LA, Nelson-Filho P, da Silva RA, et al. Revascularization and periapical repair after endodontic treatment using apical negative pressure irrigation versus conventional irrigation plus triantibiotic intracanal dressing in dogs’ teeth with apical periodontitis. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2010; 109: 779–787. [DOI] [PubMed] [Google Scholar]
- 32. Thibodeau B, Teixeira F, Yamauchi M, et al. Pulp revascularization of immature dog teeth with apical periodontitis. J Endod 2007; 33: 680–689. [DOI] [PubMed] [Google Scholar]
- 33. Torabinejad M, Turman M. Revitalization of tooth with necrotic pulp and open apex by using platelet-rich plasma: a case report. J Endod 2011; 37: 265–268. [DOI] [PubMed] [Google Scholar]
- 34. Lin LM, Shimizu E, Gibbs JL, et al. Histologic and histobacteriologic observations of failed revascularization/revitalization therapy: a case report. J Endod 2014; 40: 291–295. [DOI] [PubMed] [Google Scholar]
- 35. Kahler B, Mistry S, Moule A, et al. Revascularization outcomes: a prospective analysis of 16 consecutive cases. J Endod 2014; 40: 333–338. [DOI] [PubMed] [Google Scholar]
- 36. Nosrat A, Seifi A, Asgary S. Regenerative endodontic treatment (revascularization) for necrotic immature permanent molars: a review and report of two cases with a new biomaterial. J Endod 2011; 37: 562–567. [DOI] [PubMed] [Google Scholar]
- 37. Chaniotis A. Treatment options for failing regenerative endodontic procedures: report of 3 cases. J Endod 2017; 43: 1472–1478. [DOI] [PubMed] [Google Scholar]
- 38. Verma P, Nosrat A, Kim JR, et al. Effect of residual bacteria on the outcome of pulp regeneration in vivo. J Dent Res 2017; 96: 100–106. [DOI] [PubMed] [Google Scholar]
- 39. Shimizu E, Ricucci D, Albert J, et al. Clinical, radiographic, and histological observation of a human immature permanent tooth with chronic apical abscess after revitalization treatment. J Endod 2013; 39: 1078–1083. [DOI] [PubMed] [Google Scholar]
- 40. Martin G, Ricucci D, Gibbs JL, et al. Histological findings of revascularized/revitalized immature permanent molar with apical periodontitis using platelet-rich plasma. J Endod 2013; 39: 138–144. [DOI] [PubMed] [Google Scholar]
- 41. Lei L, Chen Y, Zhou R, et al. Histologic and immunohistochemical findings of a human immature permanent tooth with apical periodontitis after regenerative endodontic treatment. J Endod 2015; 41: 1172–1179. [DOI] [PubMed] [Google Scholar]
- 42. Siqueira JF, Jr, Rocas IN. Clinical implications and microbiology of bacterial persistence after treatment procedures. J Endod 2008; 34: 1291–1301.e1293. [DOI] [PubMed] [Google Scholar]
- 43. Wu MK, Dummer PM, Wesselink PR. Consequences of and strategies to deal with residual post-treatment root canal infection. Int Endod J 2006; 39: 343–356. [DOI] [PubMed] [Google Scholar]
- 44. Ricucci D, Siqueira JF., Jr. Biofilms and apical periodontitis: study of prevalence and association with clinical and histopathologic findings. J Endod 2010; 36: 1277–1288. [DOI] [PubMed] [Google Scholar]
- 45. Lin LM, Kim SG, Martin G, et al. Continued root maturation despite persistent apical periodontitis of immature permanent teeth after failed regenerative endodontic therapy. Aust Endod J. Epub ahead of print 16 January 2018. DOI: 10.1111/aej.12252. [DOI] [PubMed] [Google Scholar]
- 46. Zizka R, Buchta T, Voborna I, et al. Root maturation in teeth treated by unsuccessful revitalization: 2 case reports. J Endod 2016; 42: 724–729. [DOI] [PubMed] [Google Scholar]
- 47. Nosrat A, Li KL, Vir K, et al. Is pulp regeneration necessary for root maturation? J Endod 2013; 39: 1291–1295. [DOI] [PubMed] [Google Scholar]
- 48. Yamauchi N, Nagaoka H, Yamauchi S, et al. Immunohistological characterization of newly formed tissues after regenerative procedure in immature dog teeth. J Endod 2011; 37: 1636–1641. [DOI] [PubMed] [Google Scholar]
- 49. Wang X, Thibodeau B, Trope M, et al. Histologic characterization of regenerated tissues in canal space after the revitalization/revascularization procedure of immature dog teeth with apical periodontitis. J Endod 2010; 36: 56–63. [DOI] [PubMed] [Google Scholar]
- 50. Seo BM, Miura M, Gronthos S, et al. Investigation of multipotent postnatal stem cells from human periodontal ligament. Lancet 2004; 364: 149–155. [DOI] [PubMed] [Google Scholar]
- 51. Cao Y, Song M, Kim E, et al. Pulp-dentin regeneration: current state and future prospects. J Dent Res 2015; 94: 1544–1551. [DOI] [PubMed] [Google Scholar]
- 52. Torabinejad M, Faras H. A clinical and histological report of a tooth with an open apex treated with regenerative endodontics using platelet-rich plasma. J Endod 2012; 38: 864–868. [DOI] [PubMed] [Google Scholar]
- 53. Peng C, Zhao Y, Wang W, et al. Histologic findings of a human immature revascularized/regenerated tooth with symptomatic irreversible pulpitis. J Endod 2017; 43: 905–909. [DOI] [PubMed] [Google Scholar]
- 54. Huang GTJ, Yamaza T, Shea LD, et al. Stem/progenitor cell-mediated de novo regeneration of dental pulp with newly deposited continuous layer of dentin in an in vivo model. Tissue Eng Part A 2010; 16: 605–615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Iohara K, Imabayashi K, Ishizaka R, et al. Complete pulp regeneration after pulpectomy by transplantation of CD105+ stem cells with stromal cell-derived factor-1. Tissue Eng Part A 2011; 17: 1911–1920. [DOI] [PubMed] [Google Scholar]
- 56. Fukuda K, Yuasa S. Stem cells as a source of regenerative cardiomyocytes. Circ Res 2006; 98: 1002–1013. [DOI] [PubMed] [Google Scholar]
- 57. Alsberg E, von Recum HA, Mahoney MJ. Environmental cues to guide stem cell fate decision for tissue engineering applications. Expert Opin Biol Ther 2006; 6: 847–866. [DOI] [PubMed] [Google Scholar]
- 58. Lin LM, Ricucci D, Huang GT. Regeneration of the dentine-pulp complex with revitalization/revascularization therapy: challenges and hopes. Int Endod J 2014; 47: 713–724. [DOI] [PubMed] [Google Scholar]
- 59. Chmilewsky F, Jeanneau C, Dejou J, et al. Sources of dentin-pulp regeneration signals and their modulation by the local microenvironment. J Endod 2014; 40: S19–25. [DOI] [PubMed] [Google Scholar]
- 60. Gronthos S, Brahim J, Li W, et al. Stem cell properties of human dental pulp stem cells. J Dent Res 2002; 81: 531–535. [DOI] [PubMed] [Google Scholar]
- 61. Shi S, Bartold PM, Miura M, et al. The efficacy of mesenchymal stem cells to regenerate and repair dental structures. Orthod Craniofac Res 2005; 8: 191–199. [DOI] [PubMed] [Google Scholar]
- 62. Huang GT, Gronthos S, Shi S. Mesenchymal stem cells derived from dental tissues vs. those from other sources: their biology and role in regenerative medicine. J Dent Res 2009; 88: 792–806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Huang GT, Sonoyama W, Liu Y, et al. The hidden treasure in apical papilla: the potential role in pulp/dentin regeneration and bioroot engineering. J Endod 2008; 34: 645–651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Sonoyama W, Liu Y, Yamaza T, et al. Characterization of the apical papilla and its residing stem cells from human immature permanent teeth: a pilot study. J Endod 2008; 34: 166–171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Cordeiro MM, Dong Z, Kaneko T, et al. Dental pulp tissue engineering with stem cells from exfoliated deciduous teeth. J Endod 2008; 34: 962–969. [DOI] [PubMed] [Google Scholar]
- 66. Kerkis I, Caplan AI. Stem cells in dental pulp of deciduous teeth. Tissue Eng Part B Rev 2012; 18: 129–138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 2006; 126: 663–676. [DOI] [PubMed] [Google Scholar]
- 68. Yamanaka S. Induced pluripotent stem cells: past, present, and future. Cell Stem Cell 2012; 10: 678–684. [DOI] [PubMed] [Google Scholar]
- 69. Park IH, Arora N, Huo H, et al. Disease-specific induced pluripotent stem cells. Cell 2008; 134: 877–886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Otsu K, Kishigami R, Oikawa-Sasaki A, et al. Differentiation of induced pluripotent stem cells into dental mesenchymal cells. Stem Cells Dev 2012; 21: 1156–1164. [DOI] [PubMed] [Google Scholar]
- 71. Nakashima M. Induction of dentine in amputated pulp of dogs by recombinant human bone morphogenetic proteins-2 and -4 with collagen matrix. Arch Oral Biol 1994; 39: 1085–1089. [DOI] [PubMed] [Google Scholar]
- 72. Kwon YS, Lee SH, Hwang YC, et al. Behaviour of human dental pulp cells cultured in a collagen hydrogel scaffold cross-linked with cinnamaldehyde. Int Endod J 2017; 50: 58–66. [DOI] [PubMed] [Google Scholar]
- 73. Farea M, Husein A, Halim AS, et al. Synergistic effects of chitosan scaffold and TGFbeta1 on the proliferation and osteogenic differentiation of dental pulp stem cells derived from human exfoliated deciduous teeth. Arch Oral Biol 2014; 59: 1400–1411. [DOI] [PubMed] [Google Scholar]
- 74. Torabinejad M, Milan M, Shabahang S, et al. Histologic examination of teeth with necrotic pulps and periapical lesions treated with 2 scaffolds: an animal investigation. J Endod 2015; 41: 846–852. [DOI] [PubMed] [Google Scholar]
- 75. Rodríguez-Benítez S, Stambolsky C, Gutierrez-Perez JL, et al. Pulp revascularization of immature dog teeth with apical periodontitis using triantibiotic paste and platelet-rich plasma: a radiographic study. J Endod 2015; 41: 1299–1304. [DOI] [PubMed] [Google Scholar]
- 76. Bezgin T, Yilmaz AD, Celik BN, et al. Efficacy of platelet-rich plasma as a scaffold in regenerative endodontic treatment. J Endod 2015; 41: 36–44. [DOI] [PubMed] [Google Scholar]
- 77. Zhu X, Wang Y, Liu Y, et al. Immunohistochemical and histochemical analysis of newly formed tissues in root canal space transplanted with dental pulp stem cells plus platelet-rich plasma. J Endod 2014; 40: 1573–1578. [DOI] [PubMed] [Google Scholar]
- 78. Torabinejad M, Faras H, Corr R, et al. Histologic examinations of teeth treated with 2 scaffolds: a pilot animal investigation. J Endod 2014; 40: 515–520. [DOI] [PubMed] [Google Scholar]
- 79. Chen YJ, Zhao YH, Zhao YJ, et al. Potential dental pulp revascularization and odonto-/osteogenic capacity of a novel transplant combined with dental pulp stem cells and platelet-rich fibrin. Cell Tissue Res 2015; 361: 439–455. [DOI] [PubMed] [Google Scholar]
- 80. He X, Chen WX, Ban G, et al. A new method to develop human dental pulp cells and platelet-rich fibrin complex. J Endod 2016; 42: 1633–1640. [DOI] [PubMed] [Google Scholar]
- 81. Woo SM, Kim WJ, Lim HS, et al. Combination of mineral trioxide aggregate and platelet-rich fibrin promotes the odontoblastic differentiation and mineralization of human dental pulp cells via BMP/Smad signaling pathway. J Endod 2016; 42: 82–88. [DOI] [PubMed] [Google Scholar]
- 82. Shivashankar VY, Johns DA, Maroli RK, et al. Comparison of the effect of PRP, PRF and induced bleeding in the revascularization of teeth with necrotic pulp and open apex: a triple blind randomized clinical trial. J Clin Diagn Res 2017; 11: ZC34–ZC39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83. Dianat O, Mashhadi Abas F, Paymanpour P, et al. Endodontic repair in immature dogs’ teeth with apical periodontitis: blood clot vs plasma rich in growth factors scaffold. Dent Traumatol 2017; 33: 84–90. [DOI] [PubMed] [Google Scholar]
- 84. Chen G, Chen J, Yang B, et al. Combination of aligned PLGA/Gelatin electrospun sheets, native dental pulp extracellular matrix and treated dentin matrix as substrates for tooth root regeneration. Biomaterials 2015; 52: 56–70. [DOI] [PubMed] [Google Scholar]
- 85. Liao F, Chen Y, Li Z, et al. A novel bioactive three-dimensional beta-tricalcium phosphate/chitosan scaffold for periodontal tissue engineering. J Mater Sci Mater Med 2010; 21: 489–496. [DOI] [PubMed] [Google Scholar]
- 86. AbdulQader ST, Rahman IA, Thirumulu KP, et al. Effect of biphasic calcium phosphate scaffold porosities on odontogenic differentiation of human dental pulp cells. J Biomater Appl 2016; 30: 1300–1311. [DOI] [PubMed] [Google Scholar]
- 87. Galler KM, D’Souza RN, Hartgerink JD, et al. Scaffolds for dental pulp tissue engineering. Adv Dent Res 2011; 23: 333–339. [DOI] [PubMed] [Google Scholar]
- 88. Cen L, Liu W, Cui L, et al. Collagen tissue engineering: development of novel biomaterials and applications. Pediatr Res 2008; 63: 492–496. [DOI] [PubMed] [Google Scholar]
- 89. Alsousou J, Ali A, Willett K, et al. The role of platelet-rich plasma in tissue regeneration. Platelets 2013; 24: 173–182. [DOI] [PubMed] [Google Scholar]
- 90. Ji W, Yang F, Seyednejad H, et al. Biocompatibility and degradation characteristics of PLGA-based electrospun nanofibrous scaffolds with nanoapatite incorporation. Biomaterials 2012; 33: 6604–6614. [DOI] [PubMed] [Google Scholar]
- 91. He L, Pan S, Li Y, et al. Increased proliferation and adhesion properties of human dental pulp stem cells in PLGA scaffolds via simulated microgravity. Int Endod J 2016; 49: 161–173. [DOI] [PubMed] [Google Scholar]
- 92. Feng B, Jinkang Z, Zhen W, et al. The effect of pore size on tissue ingrowth and neovascularization in porous bioceramics of controlled architecture in vivo. Biomed Mater 2011; 6: 015007. [DOI] [PubMed] [Google Scholar]
- 93. Sopyan I. Recent development on porous calcium phosphate ceramics for biomedical application. Med J Malaysia 2008; 63(suppl. A): 14–15. [PubMed] [Google Scholar]
- 94. Ryu HS, Hong KS, Lee JK, et al. Magnesia-doped HA/beta-TCP ceramics and evaluation of their biocompatibility. Biomaterials 2004; 25: 393–401. [DOI] [PubMed] [Google Scholar]
- 95. Kim SG. Biological molecules for the regeneration of the pulp-dentin complex. Dent Clin North Am 2017; 61: 127–141. [DOI] [PubMed] [Google Scholar]
- 96. Begue-Kirn C, Smith AJ, Ruch JV, et al. Effects of dentin proteins, transforming growth factor beta 1 (TGF beta 1) and bone morphogenetic protein 2 (BMP2) on the differentiation of odontoblast in vitro. Int J Dev Biol 1992; 36: 491–503. [PubMed] [Google Scholar]
- 97. Huojia M, Muraoka N, Yoshizaki K, et al. TGF-beta3 induces ectopic mineralization in fetal mouse dental pulp during tooth germ development. Dev Growth Differ 2005; 47: 141–152. [DOI] [PubMed] [Google Scholar]
- 98. Liu J, Jin T, Chang S, et al. Matrix and TGF-beta-related gene expression during human dental pulp stem cell (DPSC) mineralization. In Vitro Cell Dev Biol Anim 2007; 43: 120–128. [DOI] [PubMed] [Google Scholar]
- 99. Iohara K, Nakashima M, Ito M, et al. Dentin regeneration by dental pulp stem cell therapy with recombinant human bone morphogenetic protein 2. J Dent Res 2004; 83: 590–595. [DOI] [PubMed] [Google Scholar]
- 100. About I, Laurent-Maquin D, Lendahl U, et al. Nestin expression in embryonic and adult human teeth under normal and pathological conditions. Am J Pathol 2000; 157: 287–295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101. Morrison-Graham K, Schatteman GC, Bork T, et al. A PDGF receptor mutation in the mouse (Patch) perturbs the development of a non-neuronal subset of neural crest-derived cells. Development 1992; 115: 133–142. [DOI] [PubMed] [Google Scholar]
- 102. Nakashima M. The effects of growth factors on DNA synthesis, proteoglycan synthesis and alkaline phosphatase activity in bovine dental pulp cells. Arch Oral Biol 1992; 37: 231–236. [DOI] [PubMed] [Google Scholar]
- 103. Yokose S, Kadokura H, Tajima N, et al. Platelet-derived growth factor exerts disparate effects on odontoblast differentiation depending on the dimers in rat dental pulp cells. Cell Tissue Res 2004; 315: 375–384. [DOI] [PubMed] [Google Scholar]
- 104. Kim JY, Xin X, Moioli EK, et al. Regeneration of dental-pulp-like tissue by chemotaxis-induced cell homing. Tissue Eng Part A 2010; 16: 3023–3031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105. Yang J, Yuan G, Chen Z. Pulp regeneration: current approaches and future challenges. Front Physiol 2016; 7: 58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106. Aksel H, Huang GT. Combined effects of vascular endothelial growth factor and bone morphogenetic protein 2 on odonto/osteogenic differentiation of human dental pulp stem cells in vitro. J Endod 2017; 43: 930–935. [DOI] [PubMed] [Google Scholar]
- 107. Mullane EM, Dong Z, Sedgley CM, et al. Effects of VEGF and FGF2 on the revascularization of severed human dental pulps. J Dent Res 2008; 87: 1144–1148. [DOI] [PubMed] [Google Scholar]
- 108. Feng X, Huang D, Lu X, et al. Insulin-like growth factor 1 can promote proliferation and osteogenic differentiation of human dental pulp stem cells via mTOR pathway. Dev Growth Differ 2014; 56: 615–624. [DOI] [PubMed] [Google Scholar]
- 109. Wang S, Mu J, Fan Z, et al. Insulin-like growth factor 1 can promote the osteogenic differentiation and osteogenesis of stem cells from apical papilla. Stem Cell Res 2012; 8: 346–356. [DOI] [PubMed] [Google Scholar]
- 110. De Almeida JF, Chen P, Henry MA, et al. Stem cells of the apical papilla regulate trigeminal neurite outgrowth and targeting through a BDNF-dependent mechanism. Tissue Eng Part A 2014; 20: 3089–3100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111. Cassidy N, Fahey M, Prime SS, et al. Comparative analysis of transforming growth factor-beta isoforms 1–3 in human and rabbit dentine matrices. Arch Oral Biol 1997; 42: 219–223. [DOI] [PubMed] [Google Scholar]
- 112. Holmes K, Roberts OL, Thomas AM, et al. Vascular endothelial growth factor receptor-2: structure, function, intracellular signalling and therapeutic inhibition. Cell Signal 2007; 19: 2003–2012. [DOI] [PubMed] [Google Scholar]
- 113. Kim SG, Zhou J, Solomon C, et al. Effects of growth factors on dental stem/progenitor cells. Dent Clin North Am 2012; 56: 563–575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114. Wu E, Palmer N, Tian Z, et al. Comprehensive dissection of PDGF-PDGFR signaling pathways in PDGFR genetically defined cells. PLoS ONE 2008; 3: e3794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115. D’Mello SR, Galli C, Ciotti T, et al. Induction of apoptosis in cerebellar granule neurons by low potassium: inhibition of death by insulin-like growth factor I and cAMP. Proc Natl Acad Sci U S A 1993; 90: 10989–10993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116. Fang Y, Wang LP, Du FL, et al. Effects of insulin-like growth factor I on alveolar bone remodeling in diabetic rats. J Periodontal Res 2013; 48: 144–150. [DOI] [PubMed] [Google Scholar]
- 117. Alkharobi H, Alhodhodi A, Hawsawi Y, et al. IGFBP-2 and -3 co-ordinately regulate IGF1 induced matrix mineralisation of differentiating human dental pulp cells. Stem Cell Res 2016; 17: 517–522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118. Pedard M, Quirie A, Totoson P, et al. Vascular brain-derived neurotrophic factor pathway in rats with adjuvant-induced arthritis: effect of anti-rheumatic drugs. Atherosclerosis 2018; 274: 77–85. [DOI] [PubMed] [Google Scholar]
- 119. Liang Z, Kawano S, Chen W, et al. Minced pulp as source of pulpal mesenchymal stem cells with odontogenic differentiation capacity. J Endod 2018; 44: 80–86. [DOI] [PubMed] [Google Scholar]
- 120. He L, Kim SG, Gong Q, et al. Regenerative endodontics for adult patients. J Endod 2017; 43: S57–S64. [DOI] [PubMed] [Google Scholar]