Histiocytoses are rare diseases involving any tissue or organ of adults or children, and with variable clinical presentation and prognosis.1 They can be classified into five main groups.2 Most histiocytoses in the L group [HL; this group includes Langerhans cell histiocytosis (LCH), Erdheim-Chester Disease (ECD), mixed histiocytosis, indeterminate cell histiocytosis, and extracutaneous juvenile xanthogranuloma with mutation of the MAPkinase pathway] have oncogenic mutations of BRAF,3–4 and the remaining cases often have mutations of other genes, also activating the MAPkinase cell signaling pathway.5–7
Treatment of histiocytoses with MAPkinase pathway inhibitors induces major tumor response.8–10 Vemurafenib (a selective BRAF inhibitor) has obtained orphan disease designation for treatment of LCH and ECD in Europe and has recently been approved by the FDA for the treatment of ECD with BRAF mutation. The detection of BRAF mutations is therefore a major issue for patients with HL such as ECD and LCH.
We previously reported that in some cases, one of the methods currently used for the detection of BRAF mutations in melanomas was responsible for false negative results, but that this problem can be corrected by using highly sensitive pico-droplet digital polymerase chain reaction (pddPCR).5 Indeed, pddPCR allows the detection of mutations with a variant allele frequency (VAF) as low as 0.01%.11 However a low VAF in solid tumors or leukemia usually corresponds to a subclone, and may not be relevant clinically.12 We thus decided to investigate VAF and response to BRAF inhibitors in a large series of patients with histiocytosis.
All patients were included in the French Histiocytosis Registry approved by the Comité de Protection des Personnes Ile de France III (#2011-A00447-34). Selection criteria for the present study were: age at the time of diagnosis of at least 18 years, and histiocyte-infiltrated sample available for histology review and molecular analysis. Patients with histiocytoses in the C, R, M and H groups2 were excluded (Online Supplementary Figures S2 and S3). Detection of somatic mutations was performed on DNA extracted from areas infiltrated with the highest percentage of histiocytes, using pyrosequencing or pddPCR as previously described.5,13 Both pyrosequencing and pddPCR are quantitative methods, however a pyrosequencing result is based on an average of two PCR, while each pddPCR is obtained from 4 to 5,000,000 PCR. Thus pddPCR is highly sensitive, and it provides absolute quantification when the sample is diluted. For this latter method, BRAF was considered as mutated when at least 3 droplets in the cluster were positive.11 Cases without mutation were screened for other mutations of genes in the MAPkinase pathway by targeted NGS. Cases without any mutations were classified as either wild type (WT) for BRAFV600E when at least 1,000 droplets were amplified, or as non-conclusive when the number of amplified droplets was lower. The sensitivity to targeted treatments was evaluated using the best metabolic response (MR) at 3 or 6 months. MR was determined by [18F]fluorodeoxyglucose (FDG) positron emission tomography (PET) scan as previously described.8
We analyzed 577 tissue samples with histiocytosis infiltration, from 474 adult patients (flowchart, Online Supplementary Figure S2). Most (n=432) of the patients had HL, and the others were unclassified (Online Supplementary Figure S3). Among the 432 adult patients with HL, the median age at the time of diagnosis was 54.1 years (range 18.0 to 90.7 years), and the male/female ratio was 1.81 (270/150). The mutational status of 287 (66.4%) of the patients was identified (i.e., either BRAFV600E mutation, another mutation in the MAPkinase pathway, or WT). In the 145 (33.6%) other cases, DNA obtained from infiltrated FFPE tissues could not be amplified (n=101), or no mutations were detected, but the amount or quality of DNA did not allow 1,000 amplicons to be obtained by pddPCR.
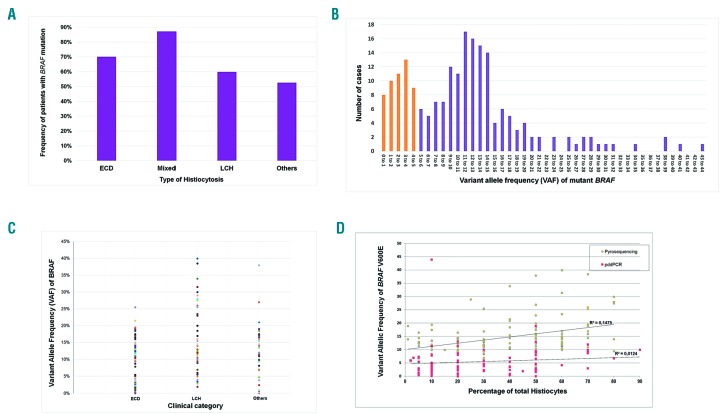
The frequency of mutations in the MAPkinase pathway in adults with HL was 70.4% (202/287). A BRAFV600E mutation was detected in 177/287 (61.9%) patients. Another type of BRAF mutation or a mutation of another gene of the MAPkinase cell-signaling pathway were detected in 5/25 and 20/25 patients respectively. Among patients with HL, BRAFV600E mutations were more frequent in mixed histiocytoses than in ECD and LCH (80.7%, 64.7%, and 57.0% respectively, P<0.05, Chi2 test) (Figure 1A).
Figure 1.
Variant allele frequencies of BRAF mutations in adult histiocytoses. (A) Frequencies of patients with BRAF mutations in the different types of histiocytoses. (B) Distribution of the histiocytoses according to their VAF. For instance, the numbers of cases with VAF 0 to 1%, 1 to 2% and 2 to 3% were 8, 10 and 11 respectively. (C) VAF values in the main types of histiocytoses. (D) Correlation of VAF with the percentage of histiocytes (i.e., all the histiocytes identified by histology among all cells present within the tissue). Whatever the method used, the correlation of VAF with the percentage of histiocytes was low: R2 was 0.15 and 0.01 in pyrosequencing and pddPCR respectively.
Of the 577 samples, 135 were found by pyro -sequencing to have a BRAFV600E mutation. PddPCR analysis - which we had previously shown to have high sensitivity11,14 – was then run on 173 samples thought not to have the BRAFV600E mutation. This detected mutations in 41 of them (23.6%) (Online Supplementary Figure S4). Thus, the use of a highly sensitive method is mandatory to reliably detect BRAFV600E mutation in histiocytoses.
Median VAF was 11.0% (range 0.04 to 44.0%) in the 197 samples for which it was available. VAF in histiocytoses was thus obviously lower than the median VAF of 43.6% that we observed in melanomas over the same decade.14 Distribution of VAF in histiocytosis samples was bimodal (Figure 1B). Interestingly, VAF were lower than 5% and 2% in 49 (24.8%) and 16 (8.1%) of cases respectively. Most of the methods used to detect somatic mutations in FFPE samples of solid tumors, including NGS, do not routinely detect VAF as low as 5% or 2%, and are thus not appropriate to detect BRAFV600E mutation in histiocytoses. This is an important message for non-specialized platforms, because of the risk of false negatives. We suspect that these methods can also fail to detect other mutations in such samples. Supporting this, in 54 samples of histiocytoses (children and adults) in which we detected mutations of MAP2K1, the median VAF was 7.5%, and 21/54 (38.9%) samples had a VAF <5% (J.F. Emile, MD, PhD, unpublished data, July 2018).
We then investigated the causes of the low VAF in histiocytoses. It was similar in ECD, LCH and unclassified histiocytoses (median 10.0%, 13.0% and 11.3% respectively) (Figure 1C). As VAF obviously depends on the respective amount of tumor and reactive cells within the analyzed tissues,14 we compared VAF with the evaluated percentage of histiocytes among all cell types (Figure 1D). Surprisingly, VAF was not correlated, which suggests that, at least in ECD, a variable and sometimes high proportion of histiocytes do not harbor the oncogenic mutation and are reactive stromal cells. This was confirmed by immunohistochemistry showing that the proportion of histiocytes stained with BRAFV600E-specific antibody was very different in each biopsy (Online Supplementary Figure S1). Alternatively, the histiocytes with BRAF mutation might be a subclone of the neoplasia.
Patients having ECD with BRAFV600E mutation can benefit from treatment with BRAF inhibitors,8–10 and this treatment is now considered a standard of care in patients with life-threatening disease.1,15 The low VAF might be indicative of a subclone, and some oncologists suspect that patients with very low VAF will not respond to BRAF-targeted therapies. Among the 126 patients in our series with ECD or mixed histiocytosis and BRAFV600E mutation, 48 were treated for at least 3 months with either vemurafenib or dabrafenib. We used the best metabolic response at 3 or 6 months of treatment to compare patients with low and high VAF (Figure 2). None of the patients had disease progression during treatment, and response rates were similar between patients with a VAF <5% and those with a higher VAF [90.0 % (9/10) and 89.5 % (34/38)] respectively. Thus, even in patients with low VAF the response rate to BRAF inhibitors was good. Furthermore there was no difference in the delay of response to targeted therapy. However, the first PET scan was performed at 3 months, and all but four patients were already responding to treatment at this time. The four patients whose best response occurred at 6 months had a VAF>5%.
Figure 2.
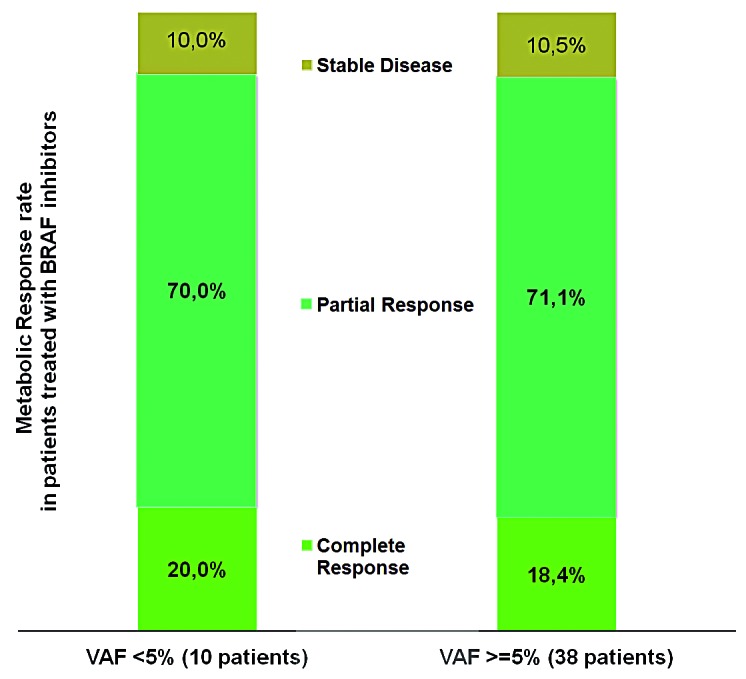
Best metabolic response to treatment with BRAF inhibitors at 3 or 6 months in patients, with either low (<5%) or high VAF.
In conclusion, we report that VAF is low in patients with histiocytosis, and is not related to percentage of histiocytes. We also show that low VAF does not impact on response to BRAF inhibitors. Therefore, highly sensitive methods are both necessary and appropriate to detect mutations in histiocytoses.
Supplementary Material
Footnotes
Funding: SM received a fellowship from the Fondation pour la Recherche Médicale (FRM DEA20170637843). The study was supported by grants from the Association pour la Recherche et l’Enseignement en Pathologie (AREP). The authors want to acknowledge all the pathologists who send histiocytosis samples, and R. Ben Jannet, C.G. Kotokpo Youkou. D. Pechaud, and Y. Pothin for their expert technical work
Information on authorship, contributions, and financial & other disclosures was provided by the authors and is available with the online version of this article at www.haematologica.org.
References
- 1.Haroche J, Cohen-Aubart F, Rollins BJ, et al. Histiocytoses: emerging neoplasia behind inflammation. Lancet Oncol. 2017;18(2):e113–e125. [DOI] [PubMed] [Google Scholar]
- 2.Emile JF, Abla O, Fraitag S, et al. Revised classification of histiocytoses and neoplasms of the macrophage-dendritic cell lineages. Blood. 2016;127(22):2672–2681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Badalian-Very G, Vergilio JA, Degar BA, et al. Recurrent BRAF mutations in Langerhans cell histiocytosis. Blood. 2010;116(11):1919–1923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Haroche J, Charlotte F, Arnaud L, et al. High prevalence of BRAF V600E mutations in Erdheim-Chester disease but not in other non-Langerhans cell histiocytoses. Blood. 2012;120(13):2700–2703. [DOI] [PubMed] [Google Scholar]
- 5.Emile JF, Diamond EL, Hélias-Rodzewicz Z, et al. Recurrent RAS and PIK3CA mutations in Erdheim-Chester disease. Blood. 2014;124(19): 3016–3019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chakraborty R, Hampton OA, Shen X, et al. Mutually exclusive recurrent somatic mutations in MAP2K1 and BRAF support a central role for ERK activation in LCH pathogenesis. Blood. 2014;124(19):3007–3015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Diamond EL, Durham BH, Haroche J, et al. Diverse and targetable kinase alterations drive histiocytic neoplasms. Cancer Discov. 2016;6(2):154–165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Haroche J, Cohen-Aubart F, Emile JF, et al. Reproducible and sustained efficacy of targeted therapy with vemurafenib in patients with BRAF(V600E)-mutated Erdheim-Chester disease. J Clin Oncol. 2015;33(5):411–418. [DOI] [PubMed] [Google Scholar]
- 9.Cohen Aubart F, Emile JF, Carrat F, et al. Targeted therapies in 54 patients with Erdheim-Chester disease, including follow-up after interruption (the LOVE study). Blood. 2017;130(11):1377–1380. [DOI] [PubMed] [Google Scholar]
- 10.Bhatia A, Ulaner G, Rampal R, et al. Single-agent dabrafenib for BRAFV600E-mutated histiocytosis. Haematologica. 2018;103(4): e177–e180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Héritier S, Hélias-Rodzewicz Z, Lapillonne H, et al. Circulating cell-free BRAFV600E as a biomarker in children with Langerhans cell histiocytosis. Br J Haematol. 2017;178(3):457–467. [DOI] [PubMed] [Google Scholar]
- 12.Laurent-Puig P, Pekin D, Normand C, et al. Clinical relevance of KRAS-mutated subclones detected with picodroplet digital PCR in advanced colorectal cancer treated with anti-EGFR therapy. Clin Cancer Res. 2015;21(5):1087–1097. [DOI] [PubMed] [Google Scholar]
- 13.Colomba E, Hélias-Rodzewicz Z, Von Deimling A, et al. Detection of BRAF p.V600E mutations in melanomas: comparison of four methods argues for sequential use of immunohistochemistry and pyrosequencing. J Mol Diagn. 2013;15(1):94–100. [DOI] [PubMed] [Google Scholar]
- 14.Hélias-Rodzewicz Z, Funck-Brentano E, Baudoux L, et al. Variations of BRAF mutant allele percentage in melanomas. BMC Cancer. 2015;15:497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Diamond EL, Dagna L, Hyman DM, et al. Consensus guidelines for the diagnosis and clinical management of Erdheim-Chester disease. Blood. 2014;124(4):483–492. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.