Abstract
The gut microbiota, as an important factor affecting host health, plays a significant role in the occurrence and development of diabetes mellitus (DM), and the mechanism may be related to excessive endotoxins, altered short-chain fatty acids (SCFAs), and disordered bile acid metabolism. Traditional Chinese medicine (TCM) has a long history of treating DM, but its mechanism is not very clear. Recent research has suggested that Chinese herbal medicine can improve glucose metabolism by remodeling the gut microbiota, which opens new avenues for further research on hypoglycemic mechanisms. This review presents the recent progress of Chinese herbs, herbal extracts, and herbal compound preparations in treating DM through regulating the gut microbiota and summarizes the main mechanisms involved, namely, anti-inflammatory and antioxidative effects, protecting the intestinal barrier and inhibiting lipotoxicity. In addition, some suggestions for improvement are also proposed.
1. Introduction
Diabetes mellitus (DM) is a significant health concern that threatens people around the world. In 2017, the International Diabetes Federation (IDF) released the eighth edition of the “IDF diabetes map”, which showed that there were approximately 425 million adults (20-79 years old) worldwide suffering from DM, and this number might reach 629 million by 2045. In China, approximately 114.4 million people (20-79 years old) are living with DM; thus, China ranks the highest in the world (http://www.diabetesatlas.org). In the last few years, people have gradually recognized that the function of the human metabolism is not only determined by ourselves but is also closely related to our “second genome”— the gut microbiota, which has opened another door for a better understanding of DM [1–3]. To find better treatment effects for DM, a multitude of probiotic, prebiotic, and synbiotic products have been developed and gradually introduced in clinical practice [4].
Traditional Chinese medicine (TCM) has a longer than 2000-year history of treating DM. In recent years, accumulating evidence has confirmed that TCM can improve DM through different molecular mechanisms [5], and remodeling the gut microbiota is an important research focus [6]. In addition to the specific changes in some intestinal bacteria after herbal medicine is taken, researchers also pay attention to how the improved gut microbiota affects the metabolism of its host. In this review, we summarize the related literature, attempt to extract several main pathways linking the gut microbiota and glycometabolism, and then provide potential future directions that may improve the limitations of current research.
A literature search was performed using the PubMed, CNKI, Chinese Biomedical Literature, Wanfang, and VIP databases. The keywords for the search included “diabetes”, “gut microbiota”, “gut microflora”,“gut flora”, “intestinal microflora”, “Traditional Chinese medicine”, “herbal medicine”, and “herbs”, and we also searched related references.
2. The Relationship between DM and the Gut Microbiota
The gut microbiota not only affects the intestinal tract but also has a profound impact on the host's metabolic function through various mechanisms [1]. In recent decades, accumulating evidence has suggested a close connection between DM and the gut microbiota. Two cohort studies on individuals in China and Europe revealed that there are compositional and functional alterations in the gut microbiota of patients with type 2 diabetes mellitus (T2DM). For example, the amount of butyrate-producing bacteria Roseburia and Faecalibacterium prausnitzii was lower [7, 8]. In addition, a study based on the Han Chinese population showed that, compared to healthy subjects, people with type 1 diabetes mellitus (T1DM) had distinctly different gut microbiota, and Bacteroides abundance was positively correlated with autoantibodies [9].
How does a disturbed gut microbiota affect the carbohydrate metabolism of its host? According to current research, the reasons may be related to the following factors: (1) lipopolysaccharides (LPS): LPS are the vital component of the outer membrane of gram-negative bacteria, while they may cause low-grade inflammation associated with DM when excessive LPS enter into the blood circulation [10]; (2) short-chain fatty acids (SCFAs): SCFAs, mainly including acetic acid, propionic acid, and butyric acid, are the major fermentation products produced from indigestible fiber and polysaccharides, and reduced SCFA levels may destroy the host's metabolic homeostasis because they are related to energy metabolism, GLP-1 secretion, and the integrity of the intestinal mucosa [11]; (3) bile acids (BAs): BAs are produced by cholesterol degradation and are metabolized by the gut microbiota, and disordered metabolism of BAs may affect the expression of bile acid receptors in the intestine and then impair related glucose metabolism pathways [12]; (4) gut permeability: a disturbed gut microbiota may disrupt intestinal tight junction proteins, enhance gut permeability, and lead to the consistent leakage of LPS, which in turn triggers the systemic low-grade inflammation [13]; (5) energy harvest: an obesity-associated gut microbiota is more effective in utilizing energy from the diet, resulting in energy overload in the host, which forms a crucial foundation for the onset of insulin resistance and T2DM [14]; (6) intestinal immune: the gut microbiota is crucial to the development and modulation of the intestinal mucosal immune, and the dysfunctional interaction between the gut microbiota and the immune system is related to the occurrence of T1DM [15]. Conversely, improved gut microbiota may regulate one or more of the above pathways, exerting a comprehensive therapeutic effects on DM.
3. Evidence of Chinese Herbal Medicines Treating DM by Regulating the Gut Microbiota
3.1. Herbs and Herbal Extracts
Folium Mori for the treatment of Xiaoke (the name of DM in TCM) is recorded in the Compendium of Materia Medica [16], and its hypoglycemic effect has been confirmed in animal experiments [17] and clinical trials [18]. Feeding a diet containing 20% Folium Mori power to diabetic rats for 8 weeks can inhibit nonesterified fatty acid (NEFA) signaling pathway and ameliorate hyperglycemia. At the same time, the proportion of Bacteroidetes in intestinal bacteria is restored after Folium Mori treatment [19]. Another study confirmed that Folium Mori could promote the production of SCFAs and regulate the metabolism of steroids and BAs [20]. In addition to Folium Mori, Dendrobium candidum can alleviate oxidative stress in liver [21], and Rhizoma Dioscoreae is conducive to reducing the blood glucose of patients with T2DM [22]. Their details are summarized in Table 1.
Table 1.
Chinese herbs and herbal extracts treating DM by regulating the gut microbiota.
| Herbs/Herbal extracts | Models | Changes of the gut microbiota and their metabolites | Core mechanisms | Ref. |
|---|---|---|---|---|
| Folium Mori | SD rats | Increased: the phyla Bacteroidetes and Proteobacteria and class Clostridia | Improve NEFA metabolism | [19] |
|
| ||||
| Dendrobium candidum | KM mice | Increased: the abundance and diversity of gut microbiota | Attenuate oxidative stress | [21] |
|
| ||||
| Rhizoma Dioscoreae | human | Increased: Bifidobacterium | N/A | [22] |
|
| ||||
| Berberine | Wistar/SD rats |
Increased: Bifidobacterium, Lactobacillus Decreased: Escherichia coli, Enterococcus |
Protect intestinal barrier; suppress inflammatory response; promote GLP-2 secretion | [24–26] |
|
| ||||
| Rhein | db/db mice |
Increased: Bacteroidetes, Akkermansia Decreased: Firmicutes, F/B ratio |
Promote GLP-1 secretion | [27] |
|
| ||||
| Seabuckthorn Protein | ICR mice |
Increased: Bifidobacterium, Lactobacillus, Bacteroides Decreased: Clostridium coccoides, PH value |
Improve fatty acids metabolism | [28] |
|
| ||||
| Zanthoxylum alkylamides | SD rats |
Increased: Lactobacillus, Bifidobacterium, Clostridium, SCFAs Decreased: Enterococcus, Enterobacterium, Bacteroides, PH value, free ammonia |
Inhibit hepatic gluconeogenesis; promote insulin secretion |
[29] |
|
| ||||
| Polyphenols from Fructus Mori L. | db/db mice |
Increased: Bacteroidetes, Anaeroplasmatales, butyrate, propionate Decreased: Firmicutes, F/B ratio, Bacillus, Lactobacillus |
Attenuate oxidative stress | [30] |
|
| ||||
| Polysaccharide from the seed of Plantago asiatica L. | Wistar rats |
Increased: Bacteroides, Lactobacillus, Prevotella, SCFAs Decreased: Alistipes obesi |
Improve NEFA metabolism; attenuate oxidative stress | [31] |
|
| ||||
| Ganoderma atrum Polysaccharide | Wistar rats | Increased: SCFAs | Attenuate oxidative stress | [32] |
|
| ||||
| Maydis stigma Polysaccharide | KM mice | Increased: Lactobacillus, Bacteroides | N/A | [33] |
|
| ||||
| Radix Pseudostellariae Polysaccharide | C57 mice | Increased: Lactobacillus, Bifidobacterium | Attenuate oxidative stress; suppress inflammatory response | [34] |
|
| ||||
| Morus nigra Polysaccharide | db/db mice |
Increased: Bacteroidales, Bacteroides Lactobacillus, Allobaculum, Akkermansia Decreased: F/B ratio, opportunistic pathogens such as Staphylococcus and Enterococcus |
Regulate lipid metabolism; attenuate oxidative stress | [35] |
|
| ||||
| Polysaccharide from the fermented Momordica charantia L | Wistar rats |
Increased: Lactococcus laudensis, Prevotella loescheii, SCFAs Decreased: pH value |
Attenuate oxidative stress | [36] |
Abbreviations. NEFA: nonesterified fatty acid; F/B: Firmicutes/Bacteroidetes; GLP-2: glucagon-like peptide-2; GLP-1: glucagon-like peptide-1; SCFAs: short chain fatty acids; N/A: not applicable.
Berberine is the principle bioactive alkaloid of some heat-clearing herbs, such as Coptis chinensis [23]. Recent studies have shown that berberine is an excellent gut microbiota modulator in animals and humans with DM. For example, it can promote the proliferation of Bifidobacterium and Lactobacillus, inhibit the growth of Escherichia coli, and reduce the levels of LPS in the intestine, thereby relieving chronic systemic inflammation [24]. In addition, the damaged intestinal mucosa and immune barrier can also be repaired by berberine, which contributes to preventing intestinal endotoxins from entering the blood and maintaining the host's metabolic homeostasis [25, 26]. In addition, many other phytochemicals, such as rhein [27], Seabuckthorn protein [28], Zanthoxylum alkylamides [29], and polyphenols from Fructus MoriL. [30], also have a hypoglycemic effect by regulating the gut microbiota (Table 1).
Besides, polysaccharides extracted from herbal medicines are also potential prebiotics. Qixing Nie and his colleagues [31] confirmed that the polysaccharides from the seeds of Plantago asiatica L. could reduce body weight, decrease blood glucose, and repair damaged kidney function in diabetic rats. In addition, it could also increase the number of bacteria such as Bacteroides vulgatus, Lactobacillus fermentum, Prevotella loescheii, and Bacteroides ovatus and promote the production of SCFAs. Moreover, the polysaccharides extracted from Ganoderma atrum [32], Maydis stigma[33], Radix Pseudostellariae [34], Morus nigra [35], and Momordica charantia L. fermented with Lactobacillus plantarum NCU116 [36] also can treat DM by regulating intestinal microecology (Table 1).
3.2. Herbal Compound Preparations
TCM formula is the combination of several herbs, and the compatibility of the herbs is the key to playing a synergistic therapeutic role [37]. Xiexin Tang, including three herbs: Rhei rhizome, Scutellaria radix, and Coptidis rhizome, shows ideal anti-inflammation, hypoglycemic and hypolipidemic effects in rats with T2DM. In addition, Xiexin Tang can increase the number of some anti-inflammatory bacteria such as Adlercreutzia, Alloprevotella, and the concentration of SCFAs in feces. The correlation analysis suggests that the increased intestinal bacteria may be a direct regulator of metabolic function [38]. In addition to animal experiments, the relevant evidences are also confirmed by clinical randomized controlled trials. The classic formula Gegen Qinlian Decoction and the modern herbal formula AMC both have significant hypoglycemic effects, and the gut microbiota structure is optimized after 12 weeks of administration. For example, levels of Faecalibacterium, the bacteria with anti-inflammatory effects, are profoundly increased by the two formulae; the SCFA-producing bacteria, Roseburia and Blautia, are also promoted after AMC treatment [39, 40]. Besides, the hypoglycemic effect of Gegen Qinlian Decoction (animal experiment)[41], Huanglian Jiedu Decoction [42, 43], Qijian mixture [44], Banxia Xiexin Decoction [45], and three innovative herbal compound preparations [46–48] also benefits the regulation of the gut microbiota, and their details are listed in Table 2.
Table 2.
Herbal compound preparations treating DM by regulating the gut microbiota.
| Preparations | Composition of preparations | Models | Changes of the gut microbiota and metabolites | Core mechanisms | Ref. |
|---|---|---|---|---|---|
| Xiexin Tang | Rhizome Rhei, Radix Scutellaria, Rhizome Coptidis | SD rats | Increased: some SCFAs producing and anti-inflammatory bacteria such as Adlercreutzia and Alloprevotella ; SCFAs | Suppress inflammatory response | [38] |
|
| |||||
| Gegen Qinlian Decoction | Radix Puerariae, Radix Scutellariae, Rhizoma Coptidis, Honey-fried Licorice Root | human |
Increased: Faecalibacterium, Bifidobacterium, Gemmiger Decreased: Alistipes, Odoribacter |
N/A | [39] |
| KK-Ay mice | Increased: Lactobacillus johnsonii, Bacteroides vulgatus | Suppress inflammatory response | [41] | ||
|
| |||||
| AMC | Rhizoma Anemarrhenae, Momordica charantia, Rhizoma Coptidis, Salvia miltiorrhiza, Red yeast rice, Aloe vera, Schisandra chinensis, Rhizoma zingiberis | human | Increased: Faecalibacterium, Roseburia, Gemmiger, Coprococcus, Megamonas, Blautia | N/A | [40] |
|
| |||||
| Huanglian Jiedu Decoction | Coptidis Rhizoma, Scutellariae Radix, Phellodendri Cortex, Gardeniae Fructus | ZDF rats |
Increased: Bacteroides, Clostridium, Roseburia, Akkermansia, Oscillospira, Aggregatibacter, Eubacterium Decreased: Prevotella |
Protect intestinal barrier; suppress inflammatory response; promote GLP-1and GLP-2 secretion | [42] |
| SD rats |
Increased: Parabacteroides, Blautia, Akkermansia Decreased: Aerococcus, Staphylococcus, Corynebacterium |
Regulate lipid metabolism; suppress inflammatory response; attenuate oxidative stress | [43] | ||
|
| |||||
| Qijian mixture | Astragalus membranaceus, Ramulus euonymi, Coptis chinensis, Pueraria lobata | KKay mice | Increased: Bacteroidetes | Improve carbohydrate and amino acid metabolism disorder | [44] |
|
| |||||
| Banxia Xiexin Decoction | Rhizoma Pinelliae Praeparata, Radix Scutellaria, Rhizome Coptidis, Panax ginseng, Rhizoma zingiberis, Radix liquiritiae, Fructus Ziziphi Jujubae | SD rats | Increased: Bacteroides, Bifidobacterium, Lactobacilli, Enterococcus | Suppress inflammatory response | [45] |
|
| |||||
| Herbal extract + Chemical drugs | Houttuynia cordata Thunb., Metformin | OLETF rats |
Increased: Roseburia, Akkermansia, SCFAs Decreased: Gram-negative bacteria, prevotella, Escherichia coli, LPS |
Suppress inflammatory response | [46] |
|
| |||||
| Herbal extract +Probiotics | Burdock Fructooligosaccharide GF13 Lactobacillus plantarum Sc 52 | C57 mice |
Increased: Lactobacillus, Bifidobacterium, SCFAs Decreased: Enterobacterium, Enterococcus |
Protect intestinal barrier; suppress inflammatory response; | [47] |
|
| |||||
| Herbal extract +Prebiotics | Berberine, stachyose | KKay mice | Increased: Lactobacillus, Bifidobacterium | N/A | [48] |
Abbreviations. SCFAs: short chain fatty acids; GLP-1: glucagon-like peptide-1; GLP-2: glucagon-like peptide-2; LPS: lipopolysaccharides; N/A: not applicable.
3.3. Summary
According to the existing literature, the effects of antidiabetic herbal medicines on the gut microbiota have the following rules: (1) regulation of the microbiota structure by increasing microbial diversity and reducing the Firmicutes/Bacteroidetes(F/B)ratio; (2) increasing the anti-inflammatory bacteria such as Bifidobacterium, Lactobacillus, Akkermansia, and Faecalibacterium; (3) increasing the SCFAs producing bacteria, such as Roseburia and Eubacterium, and promoting the concentration of SCFAs in the intestine; (4) decreasing the abundance of pathogenic bacteria such as Escherichia coli and Enterococcus. These changes in the gut microbiota may trigger a series of chain reactions that may then improve the glucose metabolism of the hosts. The specific mechanisms are discussed below.
4. Mechanisms of Chinese Herbal Medicines Treating DM by Regulating the Gut Microbiota
4.1. Anti-Inflammatory Effect
Chronic low-grade inflammation has been recognized as a characteristic of some metabolic diseases such as DM and obesity, and the state of the gut microbiota is closely related to this type of inflammation [49]. Lactobacillus, the probiotics widely used in various fields, has the function of reducing metabolism-related inflammation in both STZ-treated rats [50] and humans with DM [51]. Bifidobacterium can also reduce the release of proinflammatory cytokines by restoring the balance between regulatory T cells (Tregs) and B lymphocytes and reversing the bacterial translocation process from the intestine to tissues [52, 53]. As we have introduced above, the Chinese herbal medicines berberine [24], Seabuckthorn protein [28], Zanthoxylum alkylamides [29], Radix Pseudostellariae polysaccharides [34], Gegen Qinlian Decoction [39, 41], and so on are all promoters of Lactobacillus and Bifidobacterium proliferation, which may be a significant reason that they exert better therapeutic benefits. In addition to promoting the growth of probiotics, alleviating the adverse effects of LPS can also attenuate inflammation response. Microbiota disorder may increase the LPS level in intestine and then aggravate the inflammatory injury of intestinal epithelium. When excessive LPS enter the blood through damaged intestinal mucosa, the low-grade systemic inflammation may be triggered [54]. In the above herbal medicines, Gegen Qinlian Decoction [41] and Banxia Xiexin Decoction [45] can reduce the leakage of LPS and decrease the inflammatory factors in serum; Huanglian Jiedu Decoction [42]can downregulate the expression of LPS-related inflammatory proteins and improve the intestinal mucosal barrier function.
Taken together, increasing anti-inflammatory bacteria, reducing the production and leakage of LPS, and downregulating the LPS-related inflammatory factors are the key links for the anti-inflammatory effects of TCM.
4.2. Antioxidative Effect
Oxidative stress plays an important role in the pathological process of DM, and it not only destroys islet β cells and insulin signaling pathways, but also may contribute to serious complications such as diabetic cardiovascular disease and diabetic nephropathy [55]. Recent studies show that some intestinal bacteria have antioxidant activity. For one thing, the probiotics reduce the level of intestinal oxidative stress through their own antioxidases and antioxidant metabolites; for another, they can also activate the antioxidant system of the host and improve oxidative stress via different pathways [56]. A healthy intestinal microbiota is the basis for maintaining redox homeostasis, while a disordered microbiota composition may induce gut epithelial reactive oxygen species (ROS) generation, causing intestinal injuries or even systemic diseases [57, 58]. In recent years, researchers have confirmed that some pro- and prebiotics can improve the oxidative stress state [59]. Similarly, the aforementioned hypoglycemic TCM such as Dendrobium candidum [21], Polyphenols from Fructus Mori L. [30], and several herbal polysaccharides [31, 32, 34–36] also have antioxidant activities. In the diabetic state, enterogenous endotoxin not only is a source of inflammation but may lead to oxidative stress in some organs, such as pancreas [60] and liver [61]. Through regulating the gut microbiota and repairing the intestinal mucosal barrier, herbal medicines can reduce endotoxin damage and avoid a series of problems caused by it. In addition, accumulating evidence indicates that some bacteria such as Lactobacillus, Bifidobacterium, and Akkermansia and the bacterial metabolite butyrate also have potential antioxidant features [56, 62–64]; as listed above, plenty of herbal medicines can promote their increase.
Thus, some herbal medicines, especially the herbal polysaccharides, may play an antioxidant role by promoting probiotic growth and inhibiting enterogenous endotoxin damage.
4.3. Protecting the Intestinal Barrier
In 1986, researchers already observed increased intestinal permeability in diabetic patients [65], and in recent years people have been concerned about the “second attack” to the organism caused by the damaged intestinal barrier. In the physiological state, the intestinal mucosa absorbs nutrients and prevents pathogenic bacteria or endotoxins from invading the blood. However, under the influence of a high fat diet, inflammatory stimulation, oxidative stress, and other factors, the structure and function of the intestinal mucosa could be disrupted, which may result in intestinal injury and systemic diseases such as DM [66]. Although the intestinal barrier plays an important role in health, there are only few drugs focusing on it [67]. By regulating the microbial composition and increasing SCFAs, some herbal medicines can improve intestinal barrier function and inhibit the damage of LPS. Specifically, adherent-invasive Escherichia coli may trigger inflammatory response and break the mucosa homeostasis [68]. On the contrary, Akkermansia and Lactobacillus can restore the integrity of intestinal epithelium by reducing proinflammatory cytokine [69, 70], and Bifidobacterium can maintain intestinal health by promoting the secretion of glucagon-like peptide-2 (GLP-2) [71]; SCFAs, as the fuel for the intestinal mucosa, not only provide energy for epithelial cells [72] but also facilitate tight junction assembly [73]. As mentioned above, berberine [24–26] can effectively regulate the gut microbiota, improve the intestinal permeability by inhibiting TLR4/MyD88/NF-kB signaling pathway, and increase the secretion of GLP-2. The compound preparation containing Burdock Fructooligosaccharide GF13 and Lactobacillus plantarum Sc 52 [47] can significantly promote SCFA production and restore the impaired intestinal barrier, which may be a potential prebiotic product for the treatment of DM.
In short, a damaged intestinal barrier is the hidden killers of metabolic homeostasis, and herbal medicines can improve this pathological state by decreasing pathogenic bacteria, increasing mucosal protective bacteria, and promoting the production of SCFAs.
4.4. Inhibiting Lipotoxicity
Lipotoxicity refers to cell dysfunction or death caused by excess lipid accumulation in nonadipose tissues [74], which not only further aggravates the progression of DM [75, 76] but also leads to diabetic complications such as cardiomyopathy [77] and angiopathy [78]. Recent studies suggest that the gut microbiota plays multiple fundamentally roles in maintaining lipid metabolic homeostasis and may be the potential target for improving dyslipidemia [79]. Above all, regulating the gut microbial ratio is an effective approach. In general, the obese usually have a higher Firmicutes/Bacteroidetes (F/B) ratio than the normal-weight [80, 81]. After treatment with rhein [27], polyphenols from Fructus Mori L. [30], and Morus nigra polysaccharides [35], a lower F/B ratio is observed. Besides, herbal medicines such as Zanthoxylum alkylamides [29], polysaccharides from the seeds of Plantago asiatica L. [31], and the fermented Momordica charantia L.[36] can inhibit lipotoxicity by promoting the production of the gut microbiota-derived metabolites acetate and butyrate. Specifically, acetate can regulate the expression of beige adipogenesis-related genes in white adipose tissue, thereby promoting the release of heat [82]. Butyrate is helpful for enhancing the mitochondrial function of skeletal muscle and brown fat and increasing energy expenditure [83]. In addition, in the abdominally obese individuals, serum lipopolysaccharide-binding protein (LBP) and NEFAs are highly correlated, which suggests LPS also lead to the disorder of lipid metabolism [84]. As discussed above, some herbal medicines can inhibit LPS by regulating the microbiota. Finally, the bacterial metabolites BAs can modulate systemic lipid metabolism via the nuclear farnesoid X receptor (FXR) and the G protein-coupled receptor 5 (GPR5) [85], and it may be a potential significant target for herbal medicines, while the relevant evidence is lacking and further study is needed.
Altogether, TCM attenuates lipotoxicity mainly through reducing F/B ratio, regulating microbial metabolites acetate, butyrate, and decreasing the damage of LPS.
4.5. Summary
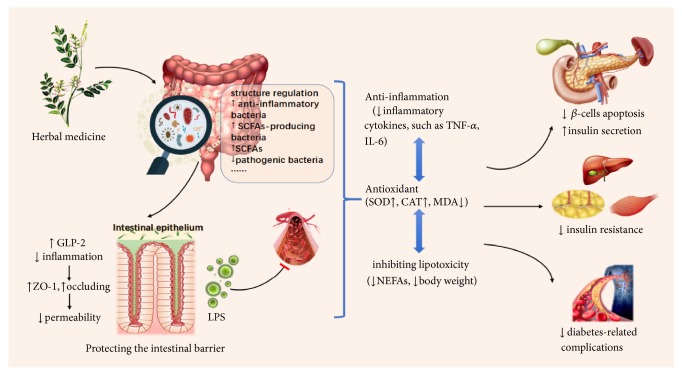
In summary, herbal medicines can exert multiple beneficial therapeutic effects on DM by regulating the gut microbiota, and anti-inflammatory and antioxidant effects, protecting the intestinal barrier and inhibiting lipotoxicity are the main mechanisms. Moreover, promoting GLP-1 secretion and inhibiting hepatic gluconeogenesis may also be related to the microbiota regulation. In addition to restoring islet function and improving insulin resistance, these mechanisms are also conducive to prevent the development of diabetes-related complications. In a word, the effects of herbal medicines on the gut microbiota are comprehensive and far-reaching (Figure 1).
Figure 1.
The mechanisms of Chinese herbal medicines treating DM by regulating the gut microbiota. SCFAs: short-chain fatty acids, LPS: lipopolysaccharide, GLP-2: glucagon-like peptide-2, TNF-α: tumor necrosis factor-α; IL-6: interleukins-6, SOD: superoxide dismutase; CAT: catalase, MDA: malondialdehyde; NEFAs: nonesterified fatty acids.
5. Discussion and Future Perspectives
Data from numerous studies have supported an essential role for the gut microbiota in the process of TCM treating DM, but in the large number of bacterial species, which may exert the most important effects? It is necessary to pay attention to the core microbiota. Although Lactobacillus species account for only 0.01 to 0.06 of all bacterial species, it plays a significant role in protecting the epithelial barrier, producing antipathogenic compounds and regulating immunity [86]. Recently, accumulating data suggests probiotic Lactobacillus strains can play an auxiliary role in treating DM [87], while other studies show that the abundance of Lactobacillus is positively correlated with the level of blood glucose [8, 88], so the crosstalk between Lactobacillus and glucose metabolism requires further investigation. Bifidobacteria are among the dominant members of the breastfed infant's gastrointestinal tract [89], and selective increases of bifidobacteria could improve high-fat-diet-induced DM and metabolic endotoxaemia in mice [90]. Akkermansia muciniphila is regarded as a next-generation benefical microbe that can activate Toll-like receptor 2 signaling pathways and promote the production of IL-10, thus playing a protective role for the intestinal mucosal barrier [91]. Roseburia has a strong butyrate production capacity that can provide energy for intestinal epithelial cells and inhibit the release of proinflammatory cytokines [92]. Normally, the abundance of Bifidobacterium, Akkermansia, and Roseburia is negatively correlated with the level of blood glucose [91–93]. As shown above, most TCM can promote the growth of these beneficial bacteria, which then elicit multitarget therapeutic effects for the treatment of DM.
However, we must recognize that plenty of factors can affect the gut microbiota [94]. In order to make use of TCM better, it is necessary to discuss it in the real world, not just in the laboratory. Among the influencing factors of adult microbiota, dietary factors can account for up to 57% of gut microbiota changes [95], and recently the gut microbiota-targeted diet for treating DM has become a topic of widespread concern [96]. Liping Zhao and his colleagues [97] found that a diet rich in fiber could optimize the gut microbiota, produce more SCFAs, and help to control blood glucose more effectively, which presented a novel ecological approach for managing T2DM. In addition, the influence of diet on microbiota may be related to the efficacy of herbal medicines. Intestinal bacteria can convert herbal chemicals into various bioactive substances, thus promoting better absorption and utilization [98], while disordered microbiota may affect the normal metabolism of drugs to some extent [99]. So, we speculate that the gut microbiota shaped by a healthy diet will enhance the efficacy of TCM, but the hypothesis warrants further investigation.
The further perspectives may include but are not limited to the following directions: above all, more advanced technical means such as metagenomic sequencing and germ-free animal should be applied to better reveal the relationship between TCM and the gut microbiota. Besides, the current studies mainly focus on intestinal bacteria, LPS and SCFAs, and the further exploration can aim at the intestinal fungi and other bacterial metabolites such as BAs. In addition, the research results based on mice/rat are difficult to apply in clinical practice directly, and the studies on human gut microbiota are urgently needed. Last and the most important, more attention should be shifted from mechanism to application. On the one hand, innovate and improve TCM in the direction of individualized targeted therapy; on the other hand, attempt to combine herbal medicines with the probiotics, probiotics or healthy food, and develop new and more effective compound preparations.
6. Conclusion
Chinese herbal medicines used to treat DM are closely related to regulating the gut microbiota, and the optimized gut microbiota improves the glucose metabolism of hosts, mainly by exerting anti-inflammatory and antioxidant effects, protecting the intestinal barrier, and inhibiting lipotoxicity. In the next step, more studies need to be based on the human microbiota and take individualization, precision, and multidisciplinary integration as the development goals. In short, the gut microbiota provides a new opportunity to elucidate the mechanism of TCM in treating DM; at the same time, herbal medicine is also a treasury of potential prebiotics and needs more in-depth study.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (no. 81774279) and the Key Research and Development Project of Sichuan Province (no. 2018SZ0068).
Disclosure
Boxun Zhang and Rensong Yue are co-first authors.
Conflicts of Interest
The authors declare that that there are no conficts of interest regarding the publication of this paper.
Authors' Contributions
All authors were responsible for the study design. Boxun Zhang and Rensong Yue have contributed equally in this work. Boxun Zhang and Rensong Yue drafted the paper. Yuan Chen, Maoyi Yang, and Xiaoying Huang participated in the literature search; Jiacheng Shui, Yuliang Peng, and Jiawei Chin supervised the study; all authors approved the final paper.
References
- 1.Sonnenburg J. L., Bäckhed F. Diet-microbiota interactions as moderators of human metabolism. Nature. 2016;535(7610):56–64. doi: 10.1038/nature18846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hartstra A. V., Bouter K. E. C., Bäckhed F., Nieuwdorp M. Insights into the role of the microbiome in obesity and type 2 diabetes. Diabetes Care. 2015;38(1):159–165. doi: 10.2337/dc14-0769. [DOI] [PubMed] [Google Scholar]
- 3.Knip M., Siljander H. The role of the intestinal microbiota in type 1 diabetes mellitus. Nature Reviews Endocrinology. 2016;12(3):154–167. doi: 10.1038/nrendo.2015.218. [DOI] [PubMed] [Google Scholar]
- 4.Kim Y. A., Keogh J. B., Clifton P. M. Probiotics, prebiotics, synbiotics and insulin sensitivity. Nutrition Research Reviews. 2017;31(1):1–17. doi: 10.1017/S095442241700018X. [DOI] [PubMed] [Google Scholar]
- 5.Tong X.-L., Dong L., Chen L., Zhen Z. Treatment of diabetes using traditional Chinese medicine: past, present and future. American Journal of Chinese Medicine. 2012;40(5):877–886. doi: 10.1142/S0192415X12500656. [DOI] [PubMed] [Google Scholar]
- 6.Gao Z., Li Q., Wu X., Zhao X., Zhao L., Tong X. New insights into the mechanisms of chinese herbal products on diabetes: a focus on the “Bacteria-mucosal immunity-inflammation-diabetes” axis. Journal of Immunology Research. 2017;2017:13. doi: 10.1155/2017/1813086.1813086 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Qin J., Li Y., Cai Z. A metagenome-wide association study of gut microbiota in type 2 diabetes. Nature. 2012;490(7418):55–60. doi: 10.1038/nature11450. [DOI] [PubMed] [Google Scholar]
- 8.Karlsson F. H., Tremaroli V., Nookaew I., et al. Gut metagenome in European women with normal, impaired and diabetic glucose control. Nature. 2013;498(7452):99–103. doi: 10.1038/nature12198. [DOI] [PubMed] [Google Scholar]
- 9.Huang Y., Li S. C., Hu J., et al. Gut microbiota profiling in han chinese with type 1 diabetes. Diabetes Research & Clinical Practice. 2018;141:256–263. doi: 10.1016/j.diabres.2018.04.032. [DOI] [PubMed] [Google Scholar]
- 10.Cani P. D., Osto M., Geurts L., Everard A. Involvement of gut microbiota in the development of low-grade inflammation and type 2 diabetes associated with obesity. Gut Microbes. 2012;3(4):279–288. doi: 10.4161/gmic.19625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Morrison D. J., Preston T. Formation of short chain fatty acids by the gut microbiota and their impact on human metabolism. Gut Microbes. 2016;7(3):189–200. doi: 10.1080/19490976.2015.1134082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Fiorucci S., Distrutti E. Bile acid-activated receptors, intestinal microbiota, and the treatment of metabolic disorders. Trends in Molecular Medicine. 2015;21(11):702–714. doi: 10.1016/j.molmed.2015.09.001. [DOI] [PubMed] [Google Scholar]
- 13.Moreira A. P. B., Texeira T. F. S., Ferreira A. B., Do Carmo Gouveia Peluzio M., De Cássia Gonçalves Alfenas R. Influence of a high-fat diet on gut microbiota, intestinal permeability and metabolic endotoxaemia. British Journal of Nutrition. 2012;108(5):801–809. doi: 10.1017/s0007114512001213. [DOI] [PubMed] [Google Scholar]
- 14.Greiner T., Bäckhed F. Effects of the gut microbiota on obesity and glucose homeostasis. Trends in Endocrinology & Metabolism. 2011;22(4):117–123. doi: 10.1016/j.tem.2011.01.002. [DOI] [PubMed] [Google Scholar]
- 15.Wen L., Ley R. E., Volchkov P. Y., et al. Innate immunity and intestinal microbiota in the development of type 1 diabetes. Nature. 2008;455(7216):1109–1113. doi: 10.1038/nature07336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ji T., Su S.-L., Guo S., et al. Research progress on bioactive component groups and their action mechanisms of Mori Folium for prevention and treatment of diabetes. Chinese Traditional and Herbal Drugs. 2015;46(5):778–784. [Google Scholar]
- 17.Cai S., Sun W., Fan Y., et al. Effect of mulberry leaf (Folium Mori) on insulin resistance via IRS-1/PI3K/Glut-4 signalling pathway in type 2 diabetes mellitus rats. Pharmaceutical Biology. 2016;54(11):2685–2691. doi: 10.1080/13880209.2016.1178779. [DOI] [PubMed] [Google Scholar]
- 18.Murata K., Yatsunami K., Fukuda E., et al. Antihyperglycemic effects of propolis mixed with mulberry leaf extract on patients with type 2 diabetes. Alternative Therapies in Health and Medicine. 2004;10(3):78–79. [PubMed] [Google Scholar]
- 19.Sheng Y., Zheng S., Ma T., et al. Mulberry leaf alleviates streptozotocin-induced diabetic rats by attenuating NEFA signaling and modulating intestinal microflora. Scientific Reports. 2017;7(1) doi: 10.1038/s41598-017-12245-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zou L. F. Research on Hypocholesterolemic Effects and Mechanism of Mulberry Leaves Water Extract on Cholesterol Metabolism in High Fat Diet Female Mice. Southwestern University; 2017. [Google Scholar]
- 21.Zhou Q. The Influence of Dendrobium Candidum Extract on Blood Glucose and the Intestinal Microflora in Diabetic Mice. Dalian, China: Dalian Medical University; 2017. [Google Scholar]
- 22.Xin H. M. Effect Study of Yam Gruel on Bifidobacterium in the Gut with Diabetic Patients of Type 2. Fuzhou, China: Fujian University of Traditional Chinese Medicine; 2016. [Google Scholar]
- 23.Meng F., Wu Z., Yin Z., Lin L., Wang R., Zhang Q. Coptidis rhizoma and its main bioactive components: recent advances in chemical investigation, quality evaluation and pharmacological activity. Chinese Medicine. 2018;13(1) doi: 10.1186/s13020-018-0171-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Liu D., Zhang Y., Liu Y., et al. Berberine modulates gut microbiota and reduces insulin resistance via the TLR4 signaling pathway. Experimental and Clinical Endocrinology & Diabetes. 2018;126(8):513–520. doi: 10.1055/s-0043-125066. [DOI] [PubMed] [Google Scholar]
- 25.Gong J., Hu M., Huang Z., et al. Berberine attenuates intestinal mucosal barrier dysfunction in type 2 diabetic rats. Frontiers in Pharmacology. 2017;8(42) doi: 10.3389/fphar.2017.00042.e1001212 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Shan C. Y., Yang J. H., Kong Y., et al. Alteration of the intestinal barrier and GLP2 secretion in berberine-treated type 2 diabetic rats. Journal of Endocrinology. 2013;218(3):255–262. doi: 10.1530/JOE-13-0184. [DOI] [PubMed] [Google Scholar]
- 27.Wang R., Zang P., Chen J., et al. Gut microbiota play an essential role in the antidiabetic effects of rhein. Evidence-Based Complementary and Alternative Medicine. 2018;2018:8. doi: 10.1155/2018/6093282.6093282 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wang W. J. Effect of Seabuckthorn Protein on Lipid Metabolism and Intestinal Microbial Community in Streptozotocin-Induced Diabetic Mice. Hefei, China: Hefei University of Technology; 2017. [Google Scholar]
- 29.You Y. M. The Effects and Mechanisms of Zanthoxylum Alkylamides on Sugar and Lipid Metabolism in Rats. Chongqing, China: Southwest University; 2016. [Google Scholar]
- 30.Li F. H. The Hypoglycemic Activity of Polyphenols Extracted from Mulberry (Fructus Mori L.) Fruit. Guangzhou, China: South China University of Technology; [Google Scholar]
- 31.Nie Q., Hu J., Gao H., Fan L., Chen H., Nie S. Polysaccharide from Plantago asiatica L. attenuates hyperglycemia, hyperlipidemia and affects colon microbiota in type 2 diabetic rats. Food Hydrocolloids. 2019;86:34–42. doi: 10.1016/j.foodhyd.2017.12.026. [DOI] [Google Scholar]
- 32.Zhu K.-X., Nie S.-P., Tan L.-H., Li C., Gong D.-M., Xie M.-Y. A polysaccharide from ganoderma atrum improves liver function in type 2 diabetic rats via antioxidant action and short-chain fatty acids excretion. Journal of Agricultural and Food Chemistry. 2016;64(9):1938–1944. doi: 10.1021/acs.jafc.5b06103. [DOI] [PubMed] [Google Scholar]
- 33.Wang C., Yin Y., Cao X., Li X. Effects of Maydis stigma polysaccharide on the intestinal microflora in type-2 diabetes. Pharmaceutical Biology. 2016;54(12):3086–3092. doi: 10.1080/13880209.2016.1211153. [DOI] [PubMed] [Google Scholar]
- 34.Wang Q. Mechanism of Radix Pseudostellariae Polysaccharide through the Nrf2 Pathway to Improve Hepatic IR. Xining,China: Qinghai University; 2015. [Google Scholar]
- 35.Chen C., You L.-J., Huang Q., et al. Modulation of gut microbiota by mulberry fruit polysaccharide treatment of obese diabetic db/db mice. Food & Function. 2018;9(7):3732–3742. doi: 10.1039/C7FO01346A. [DOI] [PubMed] [Google Scholar]
- 36.Gao H., Wen J.-J., Hu J.-L., et al. Polysaccharide from fermented Momordica charantia L. with Lactobacillus plantarum NCU116 ameliorates type 2 diabetes in rats. Carbohydrate Polymers. 2018;201:624–633. doi: 10.1016/j.carbpol.2018.08.075. [DOI] [PubMed] [Google Scholar]
- 37.Zhou M., Hong Y., Lin X., Shen L., Feng Y. Recent pharmaceutical evidence on the compatibility rationality of traditional Chinese medicine. Journal of Ethnopharmacology. 2017;206:363–375. doi: 10.1016/j.jep.2017.06.007. [DOI] [PubMed] [Google Scholar]
- 38.Wei X., Tao J., Xiao S., et al. Xiexin Tang improves the symptom of type 2 diabetic rats by modulation of the gut microbiota. Scientific Reports. 2018;8(1) doi: 10.1038/s41598-018-22094-2.3685 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Xu J., Lian F., Zhao L., et al. Structural modulation of gut microbiota during alleviation of type 2 diabetes with a Chinese herbal formula. The ISME Journal. 2015;9(3):552–562. doi: 10.1038/ismej.2014.177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Tong X., Xu J., Lian F., et al. Structural alteration of gut microbiota during the amelioration of human type 2 diabetes with hyperlipidemia by metformin and a traditional chinese herbal formula: a multicenter, randomized, open label clinical trial. Mbio. 2018;9(3) doi: 10.1128/mBio.02392-17.e02392-17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Zhang C.-H., Ma G.-Q., Deng Y.-B., et al. Effect of gegen qinlian decoction on LPS, TNF-α, IL-6, and intestinal flora in diabetic KK-Ay mice. Chinese Traditional and Herbal Drugs. 2017;48(8):1611–1616. [Google Scholar]
- 42.Liu H. Y. Huang-Lian-Jie-Du Decoction Prevents Type 2 Diabetes in IGT Rats through Its Improvement of Intestinal Hormones and Gut Microbiota. Tianjin, China: Tianjin Medical University; 2017. [Google Scholar]
- 43.Chen M., Liao Z., Lu B., et al. Huang-Lian-Jie-Du-decoction ameliorates hyperglycemia and insulin resistant in association with gut microbiota modulation. Frontiers in Microbiology. 2018;9(2380) doi: 10.3389/fmicb.2018.02380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Gao K., Yang R., Zhang J., et al. Effects of Qijian mixture on type 2 diabetes assessed by metabonomics, gut microbiota and network pharmacology. Pharmacological Research. 2018;130:93–109. doi: 10.1016/j.phrs.2018.01.011. [DOI] [PubMed] [Google Scholar]
- 45.Xu M., Yue R., Yang M., et al. Effects of Banxia Xiexin Decoction on intestinal flora and inflammatory factors of diabetic gastroparesis rats. Chinese Traditional and Herbal Drugs. 2018;49(13):3056–3061. [Google Scholar]
- 46.Wang J.-H., Bose S., Lim S.-K., et al. Houttuynia cordata facilitates metformin on ameliorating insulin resistance associated with gut microbiota alteration in OLETF rats. Genes. 2017;8(10):p. 239. doi: 10.3390/genes8100239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Luan C. Therapeutic Effects of Burdock Fructooligosaccharide Combined with Lactobacillus Plantarum Sc52 on Type 2 Diabetes. Changchun, China: Jilin Agricultural University; 2015. [Google Scholar]
- 48.Han Y., Li C., Huan Y., et al. Effects of berberine compatible with stachyose on glucolipid metabolism and gut microbiota in diabetic mice. Chinese Journal of Clinical Pharmacology. 2016;32(12):1121–1124. [Google Scholar]
- 49.Patterson E. E., Ryan P. M., Cryan J. F., et al. Gut microbiota, obesity and diabetes. Postgraduate Medical Journal. 2016;92(1087):286–300. doi: 10.1136/postgradmedj-2015-133285. [DOI] [PubMed] [Google Scholar]
- 50.Singh S., Sharma R. K., Malhotra S., Pothuraju R., Shandilya U. K. Lactobacillus rhamnosus NCDC17 ameliorates type-2 diabetes by improving gut function, oxidative stress and inflammation in high-fat-diet fed and streptozotocintreated rats. Beneficial Microbes. 2017;8(2):243–255. doi: 10.3920/BM2016.0090. [DOI] [PubMed] [Google Scholar]
- 51.Andreasen A. S., Larsen N., Pedersen-Skovsgaard T., et al. Effects of Lactobacillus acidophilus NCFM on insulin sensitivity and the systemic inflammatory response in human subjects. British Journal of Nutrition. 2010;104(12):1831–1838. doi: 10.1017/S0007114510002874. [DOI] [PubMed] [Google Scholar]
- 52.Le T. K. C., Hosaka T., Le T. T. T., et al. Oral administration of Bifidobacterium spp. improves insulin resistance, induces adiponectin, and prevents inflammatory adipokine expressions. Biomedical Research. 2014;35(5):303–310. doi: 10.2220/biomedres.35.303. [DOI] [PubMed] [Google Scholar]
- 53.Amar J., Chabo C., Waget A., et al. Intestinal mucosal adherence and translocation of commensal bacteria at the early onset of type 2 diabetes: molecular mechanisms and probiotic treatment. EMBO Molecular Medicine. 2011;3(9):559–572. doi: 10.1002/emmm.201100159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Gil-Cardoso K., Ginés I., Pinent M., Ardévol A., Blay M., Terra X. Effects of flavonoids on intestinal inflammation, barrier integrity and changes in gut microbiota during diet-induced obesity. Nutrition Research Reviews. 2016;29(2):234–248. doi: 10.1017/S0954422416000159. [DOI] [PubMed] [Google Scholar]
- 55.Domingueti C. P., Dusse L. M. S., Carvalho M. D. G., De Sousa L. P., Gomes K. B., Fernandes A. P. Diabetes mellitus: The linkage between oxidative stress, inflammation, hypercoagulability and vascular complications. Journal of Diabetes and its Complications. 2016;30(4):738–745. doi: 10.1016/j.jdiacomp.2015.12.018. [DOI] [PubMed] [Google Scholar]
- 56.Wang Y., Wu Y., Wang Y., et al. Antioxidant properties of probiotic bacteria. Nutrients. 2017;9(5):p. 521. doi: 10.3390/nu9050521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Neish A. S. Redox signaling mediated by the gut microbiota. Free Radical Research. 2013;47(11):950–957. doi: 10.3109/10715762.2013.833331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Qiao Y., Sun J., Ding Y., Le G., Shi Y. Alterations of the gut microbiota in high-fat diet mice is strongly linked to oxidative stress. Applied Microbiology and Biotechnology. 2013;97(4):1689–1697. doi: 10.1007/s00253-012-4323-6. [DOI] [PubMed] [Google Scholar]
- 59.Salehi-Abargouei A., Ghiasvand R., Hariri M. Prebiotics, prosynbiotics and synbiotics: can they reduce plasma oxidative stress parameters? a systematic review. Probiotics and Antimicrobial Proteins. 2017;9(1):1–11. doi: 10.1007/s12602-016-9248-4. [DOI] [PubMed] [Google Scholar]
- 60.Choudhury S., Ghosh S., Gupta P., Mukherjee S., Chattopadhyay S. Inflammation-induced ROS generation causes pancreatic cell death through modulation of Nrf2/NF-κB and SAPK/JNK pathway. Free Radical Research. 2015;49(11):1371–1383. doi: 10.3109/10715762.2015.1075016. [DOI] [PubMed] [Google Scholar]
- 61.Yang Y., Yan X., Wang K., et al. Triptriolide alleviates lipopolysaccharide-induced liver injury by Nrf2 and NF-κB signaling pathways. Frontiers in Pharmacology. 2018;9:p. 999. doi: 10.3389/fphar.2018.00999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Amaretti A., di Nunzio M., Pompei A., Raimondi S., Rossi M., Bordoni A. Antioxidant properties of potentially probiotic bacteria: in vitro and in vivo activities. Applied Microbiology and Biotechnology. 2013;97(2):809–817. doi: 10.1007/s00253-012-4241-7. [DOI] [PubMed] [Google Scholar]
- 63.Zhang L., Qin Q., Liu M., Zhang X., He F., Wang G. Akkermansia muciniphila can reduce the damage of gluco/lipotoxicity, oxidative stress and inflammation, and normalize intestine microbiota in streptozotocin-induced diabetic rats. Pathogens and Disease. 2018;76(4) doi: 10.1093/femspd/fty028.fty028 [DOI] [PubMed] [Google Scholar]
- 64.Jahns F., Wilhelm A., Jablonowski N., Mothes H., Greulich K. O., Glei M. Butyrate modulates antioxidant enzyme expression in malignant and non-malignant human colon tissues. Molecular Carcinogenesis. 2015;54(4):249–260. doi: 10.1002/mc.22102. [DOI] [PubMed] [Google Scholar]
- 65.Mooradian A. D., Morley J. E., Levine A. S., Prigge W. F., Gebhard R. L. Abnormal intestinal permeability to sugars in diabetes mellitus. Diabetologia. 1986;29(4):221–224. doi: 10.1007/BF00454879. [DOI] [PubMed] [Google Scholar]
- 66.Turner J. R. Intestinal mucosal barrier function in health and disease. Nature Reviews Immunology. 2009;9(11):799–809. doi: 10.1038/nri2653. [DOI] [PubMed] [Google Scholar]
- 67.Odenwald M. A., Turner J. R. The intestinal epithelial barrier: a therapeutic target? Nature Reviews Gastroenterology & Hepatology. 2017;14(1):9–21. doi: 10.1038/nrgastro.2016.169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Martinez-Medina M., Denizot J., Dreux N., et al. Western diet induces dysbiosis with increased E. coli in CEABAC10 mice, alters host barrier function favouring AIEC colonisation. Gut. 2014;63(1):116–124. doi: 10.1136/gutjnl-2012-304119. [DOI] [PubMed] [Google Scholar]
- 69.Bron P. A., Kleerebezem M., Brummer R.-J., et al. Can probiotics modulate human disease by impacting intestinal barrier function? British Journal of Nutrition. 2017;117(1):93–107. doi: 10.1017/S0007114516004037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Ottman N., Reunanen J., Meijerink M., et al. Pili-like proteins of Akkermansia muciniphila modulate host immune responses and gut barrier function. PLoS ONE. 2017;12(3) doi: 10.1371/journal.pone.0173004.e0173004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Cani P. D., Possemiers S., van de Wiele T., et al. Changes in gut microbiota control inflammation in obese mice through a mechanism involving GLP-2-driven improvement of gut permeability. Gut. 2009;58(8):1091–1103. doi: 10.1136/gut.2008.165886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Puertollano E., Kolida S., Yaqoob P. Biological significance of short-chain fatty acid metabolism by the intestinal microbiome. Current Opinion in Clinical Nutrition & Metabolic Care. 2014;17(2):139–144. doi: 10.1097/MCO.0000000000000025. [DOI] [PubMed] [Google Scholar]
- 73.Peng L., Li Z., Green R. S., Holzman I. R., Lin J. Butyrate enhances the intestinal barrier by facilitating tight junction assembly via activation of AMP-activated protein kinase in Caco-2 cell monolayers. Journal of Nutrition. 2009;139(9):1619–1625. doi: 10.3945/jn.109.104638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Schaffer J. E. Lipotoxicity: when tissues overeat. Current Opinion in Lipidology. 2003;14(3):281–287. doi: 10.1097/00041433-200306000-00008. [DOI] [PubMed] [Google Scholar]
- 75.Las G., Shirihai O. S. The role of autophagy in β-cell lipotoxicity and type 2 diabetes. Diabetes, Obesity and Metabolism. 2010;12(2):15–19. doi: 10.1111/j.1463-1326.2010.01268.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Cusi K. The role of adipose tissue and lipotoxicity in the pathogenesis of type 2 diabetes. Current Diabetes Reports. 2010;10(4):306–315. doi: 10.1007/s11892-010-0122-6. [DOI] [PubMed] [Google Scholar]
- 77.Ussher J. R. The role of cardiac lipotoxicity in the pathogenesis of diabetic cardiomyopathy. Expert Review of Cardiovascular Therapy. 2014;12(3):345–358. doi: 10.1586/14779072.2014.891939. [DOI] [PubMed] [Google Scholar]
- 78.Bardini G., Rotella C. M., Giannini S. Dyslipidemia and diabetes: reciprocal impact of impaired lipid metabolism and Beta-cell dysfunction on micro- and macrovascular complications. The Review of Diabetic Studies. 2012;9(2-3):82–93. doi: 10.1900/rds.2012.9.82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Ghazalpour A., Cespedes I., Bennett B. J., Allayee H. Expanding role of gut microbiota in lipid metabolism. Current Opinion in Lipidology. 2016;27(2):141–147. doi: 10.1097/MOL.0000000000000278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Koliada A., Syzenko G., Moseiko V., et al. Association between body mass index and Firmicutes/Bacteroidetes ratio in an adult Ukrainian population. BMC Microbiology. 2017;17(1):p. 120. doi: 10.1186/s12866-017-1027-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Ismail N. A., Ragab S. H., ElBaky A. A., Shoeib A. R. S., Alhosary Y., Fekry D. Frequency of firmicutes and bacteroidetes in gut microbiota in obese and normal weight egyptian children and adults. Archives of Medical Science. 2011;7(3):501–507. doi: 10.5114/aoms.2011.23418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Hanatani S., Motoshima H., Takaki Y., et al. Acetate alters expression of genes involved in beige adipogenesis in 3T3-L1 cells and obese KK-Ay mice. Journal of Clinical Biochemistry and Nutrition. 2016;59(3):207–214. doi: 10.3164/jcbn.16-23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Gao Z., Yin J., Zhang J., et al. Butyrate improves insulin sensitivity and increases energy expenditure in mice. Diabetes. 2009;58(7):1509–1517. doi: 10.2337/db08-1637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Saghafi-Asl M., Amiri P., Naghizadeh M., Ghavami S. M., Karamzad N. Association of endotoxaemia with serum free fatty acids in metabolically healthy and unhealthy abdominally obese individuals: a case-control study in northwest of Iran. BMJ Open. 2017;7(5) doi: 10.1136/bmjopen-2017-015910.e015910 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Wahlström A., Sayin S. I., Marschall H.-U., Bäckhed F. Intestinal crosstalk between bile acids and microbiota and its impact on host metabolism. Cell Metabolism. 2016;24(1):41–50. doi: 10.1016/j.cmet.2016.05.005. [DOI] [PubMed] [Google Scholar]
- 86.Zhang Z., Lv J., Pan L., Zhang Y. Roles and applications of probiotic Lactobacillus strains. Applied Microbiology and Biotechnology. 2018;102(19):8135–8143. doi: 10.1007/s00253-018-9217-9. [DOI] [PubMed] [Google Scholar]
- 87.Miraghajani M., Dehsoukhteh S. S., Rafie N., Hamedani S. G., Sabihi S., Ghiasvand R. Potential mechanisms linking probiotics to diabetes: a narrative review of the literature. São Paulo Medical Journal. 2017;135(2):169–178. doi: 10.1590/1516-3180.2016.0311271216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Sato J., Kanazawa A., Ikeda F., et al. Gut Dysbiosis and Detection of “Live Gut Bacteria” in Blood of Japanese Patients With Type 2 Diabetes. Diabetes Care. 2014;37(8):2343–2350. doi: 10.2337/dc13-2817. [DOI] [PubMed] [Google Scholar]
- 89.Bottacini F., Ventura M., Sinderen D. V., Motherway M. O. Diversity, ecology and intestinal function of bifidobacteria. Microbial Cell Factories. 2014;13(1) doi: 10.1186/1475-2859-13-S1-S4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Cani P. D., Neyrinck A. M., Fava F., et al. Selective increases of bifidobacteria in gut microflora improve high-fat-diet-induced diabetes in mice through a mechanism associated with endotoxaemia. Diabetologia. 2007;50(11):2374–2383. doi: 10.1007/s00125-007-0791-0. [DOI] [PubMed] [Google Scholar]
- 91.Yuji N., Kazuhiko U., Tomohisa T. A next-generation beneficial microbe: Akkermansia muciniphila. Journal of Clinical Biochemistry and Nutrition. 2018 doi: 10.3164/jcbn.18-57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Tamanai-Shacoori Z., Smida I., Bousarghin L., et al. Roseburia spp.: a marker of health? Future Microbiology. 2017;12(2):157–170. doi: 10.2217/fmb-2016-0130. [DOI] [PubMed] [Google Scholar]
- 93.Larsen N., Vogensen F. K., van den Berg F. W. J., et al. Gut microbiota in human adults with type 2 diabetes differs from non-diabetic adults. PLoS ONE. 2010;5(2) doi: 10.1371/journal.pone.0009085.e9085 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Gilbert J. A., Blaser M. J., Caporaso J. G., Jansson J. K., Lynch S. V., Knight R. Current understanding of the human microbiome. Nature Medicine. 2018;24(4):392–400. doi: 10.1038/nm.4517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Zhang N., Ju Z., Zuo T. Time for food: the impact of diet on gut microbiota and human health. Nutrition Journal. 2018;51-52:80–85. doi: 10.1016/j.nut.2017.12.005. [DOI] [PubMed] [Google Scholar]
- 96.Gentile C. L., Weir T. L. The gut microbiota at the intersection of diet and human health. Science. 2018;362(6416):776–780. doi: 10.1126/science.aau5812. [DOI] [PubMed] [Google Scholar]
- 97.Zhao L., Zhang F., Ding X., et al. Gut bacteria selectively promoted by dietary fibers alleviate type 2 diabetes. Science. 2018;359(6380):1151–1156. doi: 10.1126/science.aao5774. [DOI] [PubMed] [Google Scholar]
- 98.Xu J., Chen H.-B., Li S.-L. Understanding the molecular mechanisms of the interplay between herbal medicines and gut microbiota. Medicinal Research Reviews. 2017;37(5):1140–1185. doi: 10.1002/med.21431. [DOI] [PubMed] [Google Scholar]
- 99.Wilson I. D., Nicholson J. K. Gut microbiome interactions with drug metabolism, efficacy, and toxicity. Translational Research. 2017;179:204–222. doi: 10.1016/j.trsl.2016.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]