Abstract
Regulatory T cells (Tregs) are essential for maintaining immune tolerance and preventing autoimmune and inflammatory diseases. The activity and function of Tregs are in large part determined by various intracellular metabolic processes. Recent findings have focused on how intracellular metabolism can shape the development, trafficking, and function of Tregs. In this review, we summarize and discuss current research that reveals how distinct metabolic pathways modulate Tregs differentiation, phenotype stabilization, and function. These advances highlight numerous opportunities to alter Tregs frequency and function in physiopathologic conditions via metabolic manipulation and have important translational implications.
Keywords: cell metabolism, T cells, Foxp3, mTOR
1. Overview of the Effects of Cellular Metabolism on Tregs
Regulatory T cells (Tregs) are crucial for immune homeostasis and the control of inflammatory disorders [1,2,3,4]. We mainly focus the discussion on CD4+CD25+Foxp3+ Tregs in this review. Tregs subsets include: thymus derived Tregs (tTregs), peripheral derived Tregs (pTregs) which are generated extrathymically at peripheral sites, and iTregs that are induced ex vivo following TCR stimulation in the presence of transforming growth factor β [5]. Tregs-mediated protection has been applied in numerous preclinical models of autoimmune diseases and transplantation, which informs on their therapeutic potential for human diseases [6,7,8,9].
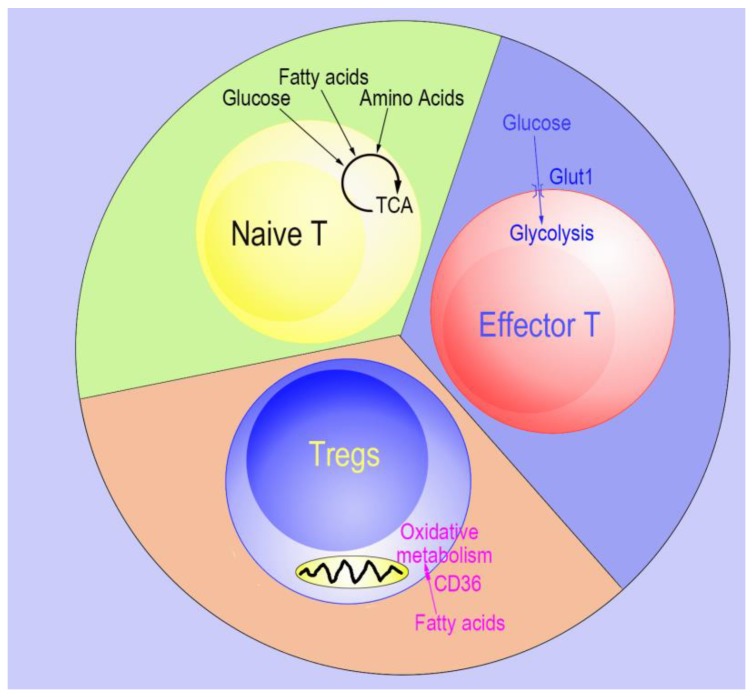
Like conventional CD4+ T cells, Tregs also have a high degree of plasticity related to different transcriptional programs [10,11,12,13], which are in turn impacted by cellular metabolism [14]. Recent findings show that Tregs use glycolysis and fatty acid oxidation differently than effector conventional T cells and naïve T cells [15]. Compared to effector conventional T cells, mouse Tregs oxidize lipids at higher rates and exhibit low glycolytic flux in vitro. On the other hand, the modest energy and biosynthesis demands of naïve T cells are typically met by the tricarboxylic acid cycle, lipid oxidation, and glycolysis (Figure 1) [16]. Moreover, glycolysis inhibits Tregs differentiation and promotes Tregs expansion, whereas fatty-acid oxidation (FAO) promotes Tregs differentiation [15,17]. In addition, the expression of Foxp3 in Tregs inhibits Myc expression and reduces glycolysis, which can be suitable for Tregs in low glucose condition [17]. Foxp3, in turn, induces oxidate phosphorylation and increases the ratio of oxidized nicotinamide adenine dinucleotide (NAD) over the reduced form (NADH), allowing Tregs to survive in elevated lactate environments [17]. Furthermore, autophagy is one of the first responses when cells experience nutrient limitation and is critical for Tregs fitness. Deficient autophagy leads to the upregulation of the mechanistic target of rapamycin (mTOR) and c-Myc (Myc proto-oncogene) expression as well as an increase in glycolysis, resulting in impaired Tregs function [18,19].
Figure 1.
Model of energy usage by naïve T, effector T, and Regulatory T cells (Tregs). Naïve T cells use glucose, fatty acids, and amino acids as their energy source. Effector T cells have higher energy efficiency and use glucose as their primary energy source. In contrast, the glucose transporter 1 is absent in Tregs and Tregs use fatty-acid oxidation (FAO) as their main energy source.
Different chain lengths of fatty acids have dissimilar effects on Tregs differentiation. Adding a short chain fatty acid to mouse or human naïve CD4+ T cells enhances Tregs differentiation, while a long chain fatty acid (LC-FA) decreases Tregs differentiation [20]. Our previous work demonstrated that sodium butyrate, which belongs to the short-chain fatty acid family, promotes Tregs induction and displays therapeutic potential in several inflammatory disorders [21]. However, Raud et al. recently reported that Carnitine palmitoyltransferase 1a (Cpt1a), a critical regulator of LC-FA oxidation, is largely dispensable for Tregs generation [22].
mTOR is a 289 kDa serine/threonine protein kinase that is highly evolutionarily conserved and has two complexes mTORC1 and mTORC2 [23]. It can directly affect T cell proliferation and differentiation through the integration of environmental cues such as energy stores, nutrients, and growth factors; and can be selectively inhibited by rapamycin [24]. Generally, mTORC1 is more sensitive to rapamycin than mTORC2 [25], however, in naïve CD4+ T cells, mTORC1 and mTORC2 have essentially the same sensitivity to rapamycin [26]. This review describes the effects of mTOR signaling dependent cellular metabolic regulation on Tregs phenotype and differentiation/suppressive function. Moreover, we discuss the role of mTOR in its modulation of T cell metabolism, which could provide targets for metabolic manipulation.
2. mTOR
As a member of phosphatidylinositol-3 kinases (PI3K) family, mTOR contains two N-terminal HEAT domains (binding domain), which are important for protein–protein interactions. It also includes an FRB region (rapamycin binding domain of mTOR), a FAT domain (a domain in PI3K-related kinases), a structurally supportive C-terminal FATC domain (a domain in PI3K-related kinases), and a kinase domain [27]. During T cell activation, T cell receptor (TCR) stimulates the mTORC1 and mTORC2 via triggering the recruitment of PI3K to the TCR receptor [28]. The activation of PI3K leads to activation of the serine–threonine kinase AKT (also known as protein kinase B) by pyruvate dehydrogenase kinase 1 (PDK1), following the activation of mTOR signaling [29]. Additionally, PI3K can directly induce the activation of mTORC2 [30]. Diverse environmental inputs can be integrated into the mTOR pathway. For example, through mTOR, metabolic cues and immune signals have an ability to direct T cell fate decisions [31]. Moreover, co-stimulatory signals, TCR and cytokine can activate mTOR via PI3K-AKT signaling to meet energy demands and activate T cells.
2.1. mTOR and Tregs Differentiation
The most profound function of mTOR in Tregs generation was first revealed using the selective inhibitor of mTOR, rapamycin, which decreased the production of effector T cells and increased the generation of Tregs [32]. Furthermore, a lack of mTORC1 signaling may lead to a failure of differentiation from naïve CD4+ T cells to Th17 lineage. When mTORC2 and mTORC1 were both mutually absent, however, naïve CD4+ T cells were differentiated into Foxp3+ Tregs [33]. This research underscores the significant role of mTOR as a fundamental regulatory factor in the differentiation of Tregs and Th17 cells (Figure 2).
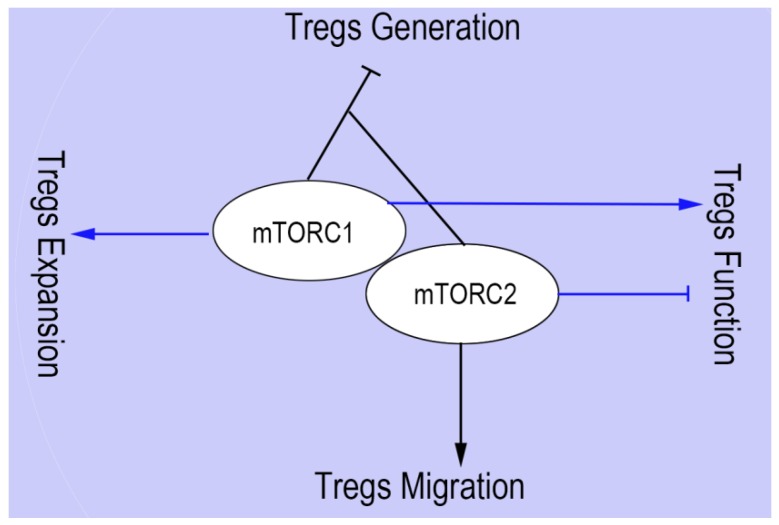
Figure 2.
The roles of mTORC1 and mTORC2 on Tregs generation, expansion, function, and migration. The absence of mTOR signaling dramatically increase Tregs generation, while deleting either mTORC1 or mTORC2 signaling does not lead to the upregulation of Foxp3+ Tregs. mTORC1 and mTORC2 play opposite roles in Tregs function, the absence of main component Raptor of mTORC1 limits Tregs function, and lack of mTORC2 increases Tregs function via promoting the activity of mTORC1. mTORC2 promotes the migration of Tregs to inflammatory sites. However, the effects of mTORC1 on the Tregs migration remain unclear. mTOR signaling is essential for Tregs expansion. Consequently, Slc3a2-deficient Tregs have an impaired mTORC1 pathway and show lower proliferation ability. However, the role of mTORC2 on Tregs expansion remains unclear.
2.2. mTOR and Tregs Function
Tregs generation is enhanced during an immune response by inhibition of mTOR. Such activity is considered a required step in maintaining Tregs suppressive capabilities. Recent evidence has revealed a critical role of mTORC1 complex in the development of Tregs suppressive activity [34] (Figure 2). If Raptor is specifically deleted from Tregs, mTORC1 is disrupted. This leads not only to a profound loss of Tregs suppressive activity, but also causes the development of a fatal early-onset inflammatory disorder. Mechanistically, cholesterol/lipid metabolism is enhanced through Raptor/mTORC1 signaling in Tregs [34]. In order to establish Tregs functional competency, the mevalonate pathway can up-regulate the Tregs suppressive molecules CTLA-4 and ICOS, as well as coordinate Tregs proliferation. Inhibition of the mTORC2 pathway is partly involved in maintaining Tregs function by mTORC1 [34]. Nevertheless, mTOR signaling is critical for properly programming activated Tregs function in order to protect tissue homeostasis and preserve immune tolerance. Tregs-specific deletion of mTOR impairs Tregs function and homeostasis, resulting in the spontaneous effector T cell activation and in the development of inflammation in barrier tissues, which is correlated with the reduction in the local tissues of both peripheral Tregs (pTregs) and thymic-derived Tregs (tTregs) [35]. In contrast, Toll-like receptor (TLR) signals enhance Tregs proliferation through mTORC1 signaling pathway, glucose transporter 1 (Glut1) upregulation, and glycolysis. However, these signals decrease the suppressive ability of Tregs [36]. It is likely that the TLR signal results in high levels of pro-inflammatory cytokines such as IL-6, IL-1, TNFα, and these pro-inflammatory cytokines decrease Tregs functionality even as Tregs maintain mTORC1 expression.
2.3. mTOR and Tregs Expansion
Although rapamycin is commonly used to block tumor cell growth, it is interesting that proliferation of CD4+ T cell and activation-induced cell death cannot be blocked by rapamycin in vitro and in kidney transplant rejection [37]. On the contrary, expanded Tregs may suppress the proliferation of effector T cells in vitro as well as prevent allograft rejection in vivo [37,38]. A new study has revealed that branched-chain amino acids (BCAAs) could be essential for the maintenance of Tregs profiling state, through metabolic reprogramming of the amino acid transporter solute carrier family 3 member 1 (Slc3a2) dependent pathway. Slc3a2-deficient Tregs impair the mTORC1 pathway and show lower proliferation ability [39] (Figure 2). The expansion of Tregs by rapamycin usually requires the addition of IL-2. Thus, even when mTORC1 is inhibited by rapamycin [40], IL-2 maintains the ability to expand Treg cells.
2.4. mTOR and Tregs Migration
As one of the complexes of mTOR, mTORC2 has been proven to have control of spatial aspects of cell growth via actin reorganization [41,42]. The immune-modulatory function is critically interrelated with the migration of activated Tregs to inflammatory tissue [43]. A recent study demonstrated that glycolysis was beneficial for their relocation. Migration of Tregs to inflamed tissue was initiated by pro-migratory stimuli through a PI3K-mTORC2-mediated signaling pathway, which culminated in stimulation of the enzyme glucokinase (GCK). Subsequently, GCK increases cytoskeletal rearrangements by interacting with actin. If Tregs lack this pathway, they will still be functionally suppressive but will fail to migrate to skin allografts as well as inhibit rejection [44] (Figure 2).
3. Promising Metabolic Targets to Manipulate Tregs Frequency and Function
mTOR is a very important regulator of cell survival and plays bidirectional roles in Tregs induction and function. The absence of mTOR signaling dramatically increases Tregs generation and inhibits the function of Tregs. To advance therapeutics and promote homeostasis of the immune system, it is necessary to identify more specific targets that modify Tregs function or induction. Next, we discuss the effects of some mTOR interrelated metabolic regulators on Tregs phenotype.
3.1. Hypoxia-Inducible Factor 1α (HIF1α)
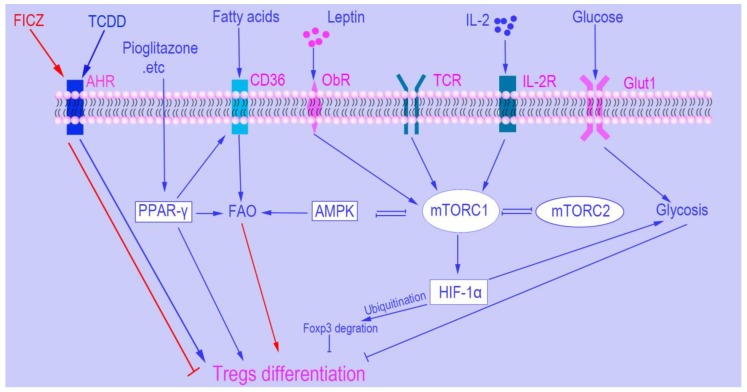
The transcription factor hypoxia-inducible factor (HIF1α) is a necessary protein for sensing oxygen saturation and subsequently initiating the cellular response to hypoxia [45,46]. It also is closely associated with Tregs and the mTOR pathway. Furthermore, impeding glycolysis up-regulates production of Tregs that occurs through inhibition of mTOR-mediated induction of HIF1α [45]. Interestingly, HIF1α inhibits Tregs differentiation in a transcription-independent manner [47], while not affecting Foxp3 mRNA levels. HIF1α exerts this inhibition through promoting the degradation of Foxp3 protein by ubiquitination (Figure 3). Moreover, lack of HIF1α promotes Tregs induction and protects mice from autoimmune neuro-inflammation [45].
Figure 3.
Metabolic and mTOR signaling control of Regulatory T cells’ differentiation. Different ligand binding with aryl hydrocarbon receptor (AHR) leads to dissimilar outcome in Tregs induction. For example, 6-formylindolo [3,2-b] carbazole (FICZ) binding with AHR inhibits Tregs generation while 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) promotes Tregs generation. Peroxisome proliferator-activated receptor-γ (PPAR-γ) increase Tregs differentiation via regulating the balance between fatty-acid oxidation (FAO) and glycometabolism. AMP-activated protein kinase (AMPK) is an important metabolic checkpoint in Tregs differentiation, and there is controversy regarding the role of the AMPK in Tregs differentiation. mTOR signaling is the most important metabolic regulator of Tregs. If mTOR is absent in naïve CD4+ T cells, it can dramatically increase Tregs differentiation even under normal activating conditions. HIF1α does not directly regulate the expression of Foxp3. However, it promotes the degradation of Foxp3 protein by ubiquitination.
Inflammation and hypoxia are two independent factors regulating the balance between Th17 and Tregs. Our group reported that tTregs are unstable in the inflammatory environment and fail to suppress collagen-induced arthritis [48]. Mechanistically, the presence of the inflammatory cytokine IL-6 converts tTregs to Th17-like cells in vitro [2,11,12,49], and IL-6 also increases HIF1α expression in a stat3-dependent manner [47]. Up to this date, it is unclear whether IL-6 regulates Tregs stability via metabolic alteration through HIF1α. A number of published articles reported that HIF1α is a key regulator in inflammation and autoimmune diseases, such as systemic lupus erythematosus (SLE) [50], rheumatoid arthritis (RA) [51,52], type 1 diabetes (T1DM) [53], multiple sclerosis (MS) [54], psoriasis [55], and inflammatory bowel disease (IBD) [56]. These findings indicate that HIF1α is a potential target to manipulate Tregs phenotype in autoimmune diseases.
3.2. AMP-Activated Protein Kinase (AMPK)
AMPK senses the cellular AMP/ATP ratio and is activated by low energy balance (high AMP/ATP ratio) [57,58,59]. Activated AMPK promotes FAO via upregulating a series of lipid oxidation related genes, such as Acetyl-CoA carboxylase 1 (ACC1), Acetyl-CoA carboxylase 2 (ACC2), Cpt1a, and sterol regulatory element binding transcription factor 1 (SREVP-1c) [60]. In addition, AMPK also regulates glycolysis via adjusting the expression of Glut1 [61]. AMPK is responsible for Tregs differentiation via regulating the balance of FAO and glycolysis. Activation of AMPK by metformin increases Tregs induction; and in murine models inhibits the progression of experimental autoimmune encephalomyelitis (EAE), and inflammatory bowel disease [15,62,63]. TCR activates both AMPK and mTOR, the latter active kinase is a negative regulator for the former one under limited nutrient condition [64]. Interestingly, the absence of AMPK has no effect on Tregs differentiation even though it enhances mTORC1 activity. Liver kinase B1 (LKB1) is a best-studied upstream kinase of AMPK and is an important metabolic sensor of Tregs. The absence of LKB1 in Foxp3+ Tregs limits the number and function of Tregs, and the effects of LKB1 on Tregs generation and function are independent of AMPK and mTOR [65,66]. Moreover, a recent paper demonstrated that Tregs differentiation was independent of the AMPK-Driven LC-FAO [22].
3.3. Leptin
Leptin is a cytokine-like hormone and is structurally similar to IL-6. Leptin mediates metabolism and T cell function [67,68]. Chronic leptin- and leptin-receptor deficiency is correlated with resistance to autoimmunity and high susceptibility to infection [69,70]. For example, leptin levels in SLE patients are correlated with regulatory T cell frequency [71]. De Rosa et al. reported that both leptin and its receptor are constitutively expressed in freshly isolated human Tregs. Increased leptin signaling acts as an antagonist during Tregs proliferation (Figure 3) [72].
3.4. Peroxisome Proliferator-Activated Receptors (PPARs)
Peroxisome proliferator-activated receptors (PPARs) are nuclear hormone receptors that function to regulate cell growth, homeostasis, and differentiation. PPARα, β/δ, and γ are three primary isoforms, each with distinct functions and tissue distribution [73]. PPARs play an important role in peroxisomal mediated β-oxidation of FAO. When PPARs are activated with specific ligands, conformational changes occur, resulting in heterodimerization with retinoid X receptor (RXRα), which then binds to promoter regions of target genes involved in FAO [74,75]. PPARs also regulate glucose metabolism. Recent studies have demonstrated that agonists of PPARs inhibit inflammatory and immune responses in non-alcoholic fatty liver disease, at least in part, through increased expression of Foxp3 and induction of Tregs (Figure 3) [76,77]. Although studies have revealed a clear picture of anti-inflammatory function by PPAR-γ, the action of other PPARs on the Tregs population remains uncertain. Recent studies have focused on the relationship between PPARs and visceral adipose tissue (VAT) Tregs. VAT Tregs are a unique subset of Tregs that uniquely express PPARγ and are specifically recruited to adipose tissue to suppress the inflammatory process [78]. In mice that specifically lack PPARγ in Tregs, VAT Treg cell population is reduced. These mice display enhanced insulin resistance and increased susceptibility to diabetic pathology [79,80]. These findings suggest that PPARγ may be a promising target for obesity-associated insulin resistance (IR). However, the absence of PPARγ in VAT Tregs does not perturb their frequency in aged mice, and VAT Tregs show a gene expression profile more similar to fat effector conventional T cells than splenic Tregs [81]. One possible reason is that long term inflammation in aged mice may change the characteristics of Tregs and promote the transdifferentiation. These results highlight the importance of PPARγ in Treg differentiation, migration, and function although aging and chronic inflammation may affect the role of PPARγ in Treg biology.
3.5. The Aryl Hydrocarbon Receptor (AHR)
AHR exists as a receptor and transcription factor, which is essential for xenobiotic metabolism and shows a cital function in immunity [82]. AHR has a high-affinity ligand, TCDD (2,3,7,8-tetrachlorodibenzo-p-dioxin) and when activated in vivo, AHR–TCDD complex leads to the induction of CD4+CD25+Foxp3+ Tregs. Alternatively, 6-formylindolo [3,2-b] carbazole (FICZ) may activate AHR to interfere with Tregs differentiation, boosting Th17 cell differentiation and worsening experimental autoimmune encephalomyelitis (EAE). Therefore, AHR regulates the balance of Tregs and Th17 cell differentiation in a ligand-specific manner (Figure 3) [83] and can be a unique target for immunosuppression therapy.
3.6. Interleukin 2 (IL-2)
IL-2 was first found as a T cell growth factor and plays an important role in T cell proliferation and differentiation [84]. We confirmed, along with several groups, that IL-2 is still necessary to induce and expand Tregs [10,85]. Zeng et al. demonstrated that IL-2 enhancement of Tregs function was dependent on the activation of mTORC1 [34]. Interestingly, IL-2 may partner with rapamycin (mTOR inhibitor) in Tregs expansion in vitro (Box 1, Figure 3). Low dose IL-2 is a promising method for the treatment of autoimmune diseases like lupus [86,87], T1DM [88], and graft-versus-host (GVHD) [89,90].
Box 1. Some unsolved questions in the metabolic regulation of Tregs.
The different roles of mTORC1 and mTORC2 in Treg induction, migration, expansion, and function;
The different metabolic profiles of Tregs during steady states and inflammatory conditions;
Identification of metabolic factors that correlate Tregs development, function, and expansion with environment cues;
The basis of the requirement of high doses of IL-2 (mTOR activator) along with rapamycin (inhibit mTOR) to expand Tregs in vitro.
4. Concluding Remarks and Perspectives
Recently, research has highlighted the complex roles of intrinsic metabolic pathways in the development and function of Tregs, which may have significant implications on immune diseases and responses. In this review, we briefly summarize the roles of metabolic sensors in the biological features of Tregs as well as their potential to be targets of clinical immune-modifying therapies (Figure 3). Immunometabolism is a promising new field, and profound questions remain to be answered (Box 1).
Funding
This work was supported by NIH R01 AR059103, NIH Star award and NIH R61 AR073409.
Conflicts of Interest
The authors declare no financial or commercial conflicts of interest.
References
- 1.Qian X., Wang K., Wang X., Zheng S.G., Lu L. Generation of human regulatory t cells de novo with suppressive function prevent xenogeneic graft versus host disease. Int. Immunopharmacol. 2011;11:630–637. doi: 10.1016/j.intimp.2010.11.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kong N., Lan Q., Chen M., Wang J., Shi W., Horwitz D.A., Quesniaux V., Ryffel B., Liu Z., Brand D., et al. Antigen-specific transforming growth factor beta-induced treg cells, but not natural treg cells, ameliorate autoimmune arthritis in mice by shifting the th17/treg cell balance from th17 predominance to treg cell predominance. Arthritis Rheum. 2012;64:2548–2558. doi: 10.1002/art.34513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lan Q., Zhou X., Fan H., Chen M., Wang J., Ryffel B., Brand D., Ramalingam R., Kiela P.R., Horwitz D.A., et al. Polyclonal cd4+foxp3+ treg cells induce tgfbeta-dependent tolerogenic dendritic cells that suppress the murine lupus-like syndrome. J. Mol. Cell Biol. 2012;4:409–419. doi: 10.1093/jmcb/mjs040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Li B., Zheng S.G. How regulatory t cells sense and adapt to inflammation. Cell. Mol. Immunol. 2015;12:519–520. doi: 10.1038/cmi.2015.65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Shevach E.M., Thornton A.M. Ttregs, ptregs, and itregs: Similarities and differences. Immunol. Rev. 2014;259:88–102. doi: 10.1111/imr.12160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bluestone J.A., Bour-Jordan H. Current and future immunomodulation strategies to restore tolerance in autoimmune diseases. Cold Spring Harb. Perspect. Biol. 2012;4 doi: 10.1101/cshperspect.a007542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Juvet S.C., Whatcott A.G., Bushell A.R., Wood K.J. Harnessing regulatory t cells for clinical use in transplantation: The end of the beginning. Am. J. Transplant. 2014;14:750–763. doi: 10.1111/ajt.12647. [DOI] [PubMed] [Google Scholar]
- 8.Horwitz D.A., Zheng S.G., Gray J.D., Wang J.H., Ohtsuka K., Yamagiwa S. Regulatory t cells generated ex vivo as an approach for the therapy of autoimmune disease. Semin. Immunol. 2004;16:135–143. doi: 10.1016/j.smim.2003.12.009. [DOI] [PubMed] [Google Scholar]
- 9.Horwitz D.A., Gray J.D., Zheng S.G. The potential of human regulatory t cells generated ex vivo as a treatment for lupus and other chronic inflammatory diseases. Arthritis Res. 2002;4:241–246. doi: 10.1186/ar414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zheng S.G., Wang J., Wang P., Gray J.D., Horwitz D.A. Il-2 is essential for tgf-beta to convert naive cd4+cd25- cells to cd25+foxp3+ regulatory t cells and for expansion of these cells. J. Immunol. 2007;178:2018–2027. doi: 10.4049/jimmunol.178.4.2018. [DOI] [PubMed] [Google Scholar]
- 11.Zheng S.G., Wang J., Horwitz D.A. Cutting edge: Foxp3+cd4+cd25+ regulatory t cells induced by il-2 and tgf-beta are resistant to th17 conversion by il-6. J. Immunol. 2008;180:7112–7116. doi: 10.4049/jimmunol.180.11.7112. [DOI] [PubMed] [Google Scholar]
- 12.Zhou X., Kong N., Wang J., Fan H., Zou H., Horwitz D., Brand D., Liu Z., Zheng S.G. Cutting edge: All-trans retinoic acid sustains the stability and function of natural regulatory t cells in an inflammatory milieu. J. Immunol. 2010;185:2675–2679. doi: 10.4049/jimmunol.1000598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gu J., Lu L., Chen M., Xu L., Lan Q., Li Q., Liu Z., Chen G., Wang P., Wang X., et al. Tgf-beta-induced cd4+foxp3+ t cells attenuate acute graft-versus-host disease by suppressing expansion and killing of effector cd8+ cells. J. Immunol. 2014;193:3388–3397. doi: 10.4049/jimmunol.1400207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Procaccini C., Carbone F., Di Silvestre D., Brambilla F., De Rosa V., Galgani M., Faicchia D., Marone G., Tramontano D., Corona M., et al. The proteomic landscape of human ex vivo regulatory and conventional t cells reveals specific metabolic requirements. Immunity. 2016;44:406–421. doi: 10.1016/j.immuni.2016.01.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Michalek R.D., Gerriets V.A., Jacobs S.R., Macintyre A.N., MacIver N.J., Mason E.F., Sullivan S.A., Nichols A.G., Rathmell J.C. Cutting edge: Distinct glycolytic and lipid oxidative metabolic programs are essential for effector and regulatory cd4+ t cell subsets. J. Immunol. 2011;186:3299–3303. doi: 10.4049/jimmunol.1003613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Pearce E.L. Metabolism in t cell activation and differentiation. Curr. Opin. Immunol. 2010;22:314–320. doi: 10.1016/j.coi.2010.01.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Angelin A., Gil-de-Gomez L., Dahiya S., Jiao J., Guo L., Levine M.H., Wang Z., Quinn W.J., 3rd, Kopinski P.K., Wang L., et al. Foxp3 reprograms t cell metabolism to function in low-glucose, high-lactate environments. Cell Metab. 2017;25:1282–1293.e7. doi: 10.1016/j.cmet.2016.12.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kabat A.M., Harrison O.J., Riffelmacher T., Moghaddam A.E., Pearson C.F., Laing A., Abeler-Dorner L., Forman S.P., Grencis R.K., Sattentau Q., et al. The autophagy gene atg16l1 differentially regulates treg and th2 cells to control intestinal inflammation. Elife. 2016;5:e12444. doi: 10.7554/eLife.12444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wei J., Long L., Yang K., Guy C., Shrestha S., Chen Z., Wu C., Vogel P., Neale G., Green D.R., et al. Autophagy enforces functional integrity of regulatory t cells by coupling environmental cues and metabolic homeostasis. Nat. Immunol. 2016;17:277–285. doi: 10.1038/ni.3365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Haghikia A., Jorg S., Duscha A., Berg J., Manzel A., Waschbisch A., Hammer A., Lee D.H., May C., Wilck N., et al. Dietary fatty acids directly impact central nervous system autoimmunity via the small intestine. Immunity. 2015;43:817–829. doi: 10.1016/j.immuni.2015.09.007. [DOI] [PubMed] [Google Scholar]
- 21.Chen X., Su W., Wan T., Yu J., Zhu W., Tang F., Liu G., Olsen N., Liang D., Zheng S.G. Sodium butyrate regulates th17/treg cell balance to ameliorate uveitis via the nrf2/ho-1 pathway. Biochem. Pharmacol. 2017;142:111–119. doi: 10.1016/j.bcp.2017.06.136. [DOI] [PubMed] [Google Scholar]
- 22.Raud B., Roy D.G., Divakaruni A.S., Tarasenko T.N., Franke R., Ma E.H., Samborska B., Hsieh W.Y., Wong A.H., Stuve P., et al. Etomoxir actions on regulatory and memory t cells are independent of cpt1a-mediated fatty acid oxidation. Cell Metab. 2018;28:504–515.e7. doi: 10.1016/j.cmet.2018.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Yang Q., Guan K.L. Expanding mtor signaling. Cell Res. 2007;17:666–681. doi: 10.1038/cr.2007.64. [DOI] [PubMed] [Google Scholar]
- 24.Blouet C., Ono H., Schwartz G.J. Mediobasal hypothalamic p70 s6 kinase 1 modulates the control of energy homeostasis. Cell Metab. 2008;8:459–467. doi: 10.1016/j.cmet.2008.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Verbist K.C., Guy C.S., Milasta S., Liedmann S., Kaminski M.M., Wang R., Green D.R. Metabolic maintenance of cell asymmetry following division in activated t lymphocytes. Nature. 2016;532:389–393. doi: 10.1038/nature17442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Delgoffe G.M., Pollizzi K.N., Waickman A.T., Heikamp E., Meyers D.J., Horton M.R., Xiao B., Worley P.F., Powell J.D. The kinase mtor regulates the differentiation of helper t cells through the selective activation of signaling by mtorc1 and mtorc2. Nat. Immunol. 2011;12:295–303. doi: 10.1038/ni.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Weichhart T., Hengstschlager M., Linke M. Regulation of innate immune cell function by mtor. Nat. Rev. Immunol. 2015;15:599–614. doi: 10.1038/nri3901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Laplante M., Sabatini D.M. Mtor signaling in growth control and disease. Cell. 2012;149:274–293. doi: 10.1016/j.cell.2012.03.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Saxton R.A., Sabatini D.M. Mtor signaling in growth, metabolism, and disease. Cell. 2017;168:960–976. doi: 10.1016/j.cell.2017.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Shimobayashi M., Hall M.N. Making new contacts: The mtor network in metabolism and signalling crosstalk. Nat. Rev. Mol. Cell Biol. 2014;15:155–162. doi: 10.1038/nrm3757. [DOI] [PubMed] [Google Scholar]
- 31.Chi H. Regulation and function of mtor signalling in t cell fate decisions. Nat. Rev. Immunol. 2012;12:325–338. doi: 10.1038/nri3198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zheng Y., Collins S.L., Lutz M.A., Allen A.N., Kole T.P., Zarek P.E., Powell J.D. A role for mammalian target of rapamycin in regulating t cell activation versus anergy. J. Immunol. 2007;178:2163–2170. doi: 10.4049/jimmunol.178.4.2163. [DOI] [PubMed] [Google Scholar]
- 33.Powell J.D., Pollizzi K.N., Heikamp E.B., Horton M.R. Regulation of immune responses by mtor. Annu. Rev. Immunol. 2012;30:39–68. doi: 10.1146/annurev-immunol-020711-075024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Zeng H., Yang K., Cloer C., Neale G., Vogel P., Chi H. Mtorc1 couples immune signals and metabolic programming to establish t(reg)-cell function. Nature. 2013;499:485–490. doi: 10.1038/nature12297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Chapman N.M., Zeng H., Nguyen T.M., Wang Y., Vogel P., Dhungana Y., Liu X., Neale G., Locasale J.W., Chi H. Mtor coordinates transcriptional programs and mitochondrial metabolism of activated treg subsets to protect tissue homeostasis. Nat. Commun. 2018;9:2095. doi: 10.1038/s41467-018-04392-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Gerriets V.A., Kishton R.J., Johnson M.O., Cohen S., Siska P.J., Nichols A.G., Warmoes M.O., de Cubas A.A., MacIver N.J., Locasale J.W., et al. Foxp3 and toll-like receptor signaling balance treg cell anabolic metabolism for suppression. Nat. Immunol. 2016;17:1459–1466. doi: 10.1038/ni.3577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Battaglia M., Stabilini A., Roncarolo M.G. Rapamycin selectively expands cd4+cd25+foxp3+ regulatory t cells. Blood. 2005;105:4743–4748. doi: 10.1182/blood-2004-10-3932. [DOI] [PubMed] [Google Scholar]
- 38.Wang G.Y., Zhang Q., Yang Y., Chen W.J., Liu W., Jiang N., Chen G.H. Rapamycin combined with allogenic immature dendritic cells selectively expands cd4+cd25+foxp3+ regulatory t cells in rats. Hepatobiliary Pancreat. Dis. Int. 2012;11:203–208. doi: 10.1016/S1499-3872(12)60149-0. [DOI] [PubMed] [Google Scholar]
- 39.Ikeda K., Kinoshita M., Kayama H., Nagamori S., Kongpracha P., Umemoto E., Okumura R., Kurakawa T., Murakami M., Mikami N., et al. Slc3a2 mediates branched-chain amino-acid-dependent maintenance of regulatory t cells. Cell Rep. 2017;21:1824–1838. doi: 10.1016/j.celrep.2017.10.082. [DOI] [PubMed] [Google Scholar]
- 40.Asanuma S., Tanaka J., Sugita J., Kosugi M., Shiratori S., Wakasa K., Shono Y., Shigematsu A., Kondo T., Kobayashi T., et al. Expansion of cd4(+)cd25 (+) regulatory t cells from cord blood cd4(+) cells using the common gamma-chain cytokines (il-2 and il-15) and rapamycin. Ann. Hematol. 2011;90:617–624. doi: 10.1007/s00277-010-1121-z. [DOI] [PubMed] [Google Scholar]
- 41.Cybulski N., Hall M.N. Tor complex 2: A signaling pathway of its own. Trends Biochem. Sci. 2009;34:620–627. doi: 10.1016/j.tibs.2009.09.004. [DOI] [PubMed] [Google Scholar]
- 42.Oh W.J., Jacinto E. Mtor complex 2 signaling and functions. Cell Cycle. 2011;10:2305–2316. doi: 10.4161/cc.10.14.16586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Gao Y., Tang J., Chen W., Li Q., Nie J., Lin F., Wu Q., Chen Z., Gao Z., Fan H., et al. Inflammation negatively regulates foxp3 and regulatory t-cell function via dbc1. Proc. Natl. Acad. Sci. USA. 2015;112:E3246–E3254. doi: 10.1073/pnas.1421463112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Kishore M., Cheung K.C.P., Fu H., Bonacina F., Wang G., Coe D., Ward E.J., Colamatteo A., Jangani M., Baragetti A., et al. Regulatory t cell migration is dependent on glucokinase-mediated glycolysis. Immunity. 2017;47:875–889.e10. doi: 10.1016/j.immuni.2017.10.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Shi L.Z., Wang R., Huang G., Vogel P., Neale G., Green D.R., Chi H. Hif1alpha-dependent glycolytic pathway orchestrates a metabolic checkpoint for the differentiation of th17 and treg cells. J. Exp. Med. 2011;208:1367–1376. doi: 10.1084/jem.20110278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Guan S.Y., Leng R.X., Tao J.H., Li X.P., Ye D.Q., Olsen N., Zheng S.G., Pan H.F. Hypoxia-inducible factor-1alpha: A promising therapeutic target for autoimmune diseases. Expert Opin. Ther. Targets. 2017;21:715–723. doi: 10.1080/14728222.2017.1336539. [DOI] [PubMed] [Google Scholar]
- 47.Dang E.V., Barbi J., Yang H.Y., Jinasena D., Yu H., Zheng Y., Bordman Z., Fu J., Kim Y., Yen H.R., et al. Control of t(h)17/t(reg) balance by hypoxia-inducible factor 1. Cell. 2011;146:772–784. doi: 10.1016/j.cell.2011.07.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Kong N., Lan Q., Chen M., Zheng T., Su W., Wang J., Yang Z., Park R., Dagliyan G., Conti P.S., et al. Induced t regulatory cells suppress osteoclastogenesis and bone erosion in collagen-induced arthritis better than natural t regulatory cells. Ann. Rheum. Dis. 2012;71:1567–1572. doi: 10.1136/annrheumdis-2011-201052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Xu L., Kitani A., Fuss I., Strober W. Cutting edge: Regulatory t cells induce cd4+cd25-foxp3- t cells or are self-induced to become th17 cells in the absence of exogenous tgf-beta. J. Immunol. 2007;178:6725–6729. doi: 10.4049/jimmunol.178.11.6725. [DOI] [PubMed] [Google Scholar]
- 50.Deng W., Ren Y., Feng X., Yao G., Chen W., Sun Y., Wang H., Gao X., Sun L. Hypoxia inducible factor-1 alpha promotes mesangial cell proliferation in lupus nephritis. Am. J. Nephrol. 2014;40:507–515. doi: 10.1159/000369564. [DOI] [PubMed] [Google Scholar]
- 51.Hu F., Shi L., Mu R., Zhu J., Li Y., Ma X., Li C., Jia R., Yang D., Li Y., et al. Hypoxia-inducible factor-1alpha and interleukin 33 form a regulatory circuit to perpetuate the inflammation in rheumatoid arthritis. PLoS ONE. 2013;8:e72650. doi: 10.1371/journal.pone.0072650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Giatromanolaki A., Sivridis E., Maltezos E., Athanassou N., Papazoglou D., Gatter K.C., Harris A.L., Koukourakis M.I. Upregulated hypoxia inducible factor-1alpha and -2alpha pathway in rheumatoid arthritis and osteoarthritis. Arthritis Res. Ther. 2003;5:193–201. doi: 10.1186/ar756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Loukovaara S., Koivunen P., Ingles M., Escobar J., Vento M., Andersson S. Elevated protein carbonyl and hif-1alpha levels in eyes with proliferative diabetic retinopathy. Acta Ophthalmol. 2014;92:323–327. doi: 10.1111/aos.12186. [DOI] [PubMed] [Google Scholar]
- 54.Graumann U., Reynolds R., Steck A.J., Schaeren-Wiemers N. Molecular changes in normal appearing white matter in multiple sclerosis are characteristic of neuroprotective mechanisms against hypoxic insult. Brain Pathol. 2003;13:554–573. doi: 10.1111/j.1750-3639.2003.tb00485.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Vasilopoulos Y., Sourli F., Zafiriou E., Klimi E., Ioannou M., Mamuris Z., Simos G., Koukoulis G., Roussaki-Schulze A. High serum levels of hif-1alpha in psoriatic patients correlate with an over-expression of il-6. Cytokine. 2013;62:38–39. doi: 10.1016/j.cyto.2013.02.029. [DOI] [PubMed] [Google Scholar]
- 56.Mimouna S., Goncalves D., Barnich N., Darfeuille-Michaud A., Hofman P., Vouret-Craviari V. Crohn disease-associated escherichia coli promote gastrointestinal inflammatory disorders by activation of hif-dependent responses. Gut Microbes. 2011;2:335–346. doi: 10.4161/gmic.18771. [DOI] [PubMed] [Google Scholar]
- 57.Bai A., Ma A.G., Yong M., Weiss C.R., Ma Y., Guan Q., Bernstein C.N., Peng Z. Ampk agonist downregulates innate and adaptive immune responses in tnbs-induced murine acute and relapsing colitis. Biochem. Pharmacol. 2010;80:1708–1717. doi: 10.1016/j.bcp.2010.08.009. [DOI] [PubMed] [Google Scholar]
- 58.Gualdoni G.A., Mayer K.A., Goschl L., Boucheron N., Ellmeier W., Zlabinger G.J. The amp analog aicar modulates the treg/th17 axis through enhancement of fatty acid oxidation. FASEB J. 2016;30:3800–3809. doi: 10.1096/fj.201600522R. [DOI] [PubMed] [Google Scholar]
- 59.Park M.J., Lee S.Y., Moon S.J., Son H.J., Lee S.H., Kim E.K., Byun J.K., Shin D.Y., Park S.H., Yang C.W., et al. Metformin attenuates graft-versus-host disease via restricting mammalian target of rapamycin/signal transducer and activator of transcription 3 and promoting adenosine monophosphate-activated protein kinase-autophagy for the balance between t helper 17 and tregs. Transl. Res. 2016;173:115–130. doi: 10.1016/j.trsl.2016.03.006. [DOI] [PubMed] [Google Scholar]
- 60.Tamas P., Hawley S.A., Clarke R.G., Mustard K.J., Green K., Hardie D.G., Cantrell D.A. Regulation of the energy sensor amp-activated protein kinase by antigen receptor and ca2+ in t lymphocytes. J. Exp. Med. 2006;203:1665–1670. doi: 10.1084/jem.20052469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Barnes K., Ingram J.C., Porras O.H., Barros L.F., Hudson E.R., Fryer L.G., Foufelle F., Carling D., Hardie D.G., Baldwin S.A. Activation of glut1 by metabolic and osmotic stress: Potential involvement of amp-activated protein kinase (ampk) J. Cell Sci. 2002;115:2433–2442. doi: 10.1242/jcs.115.11.2433. [DOI] [PubMed] [Google Scholar]
- 62.Sun Y., Tian T., Gao J., Liu X., Hou H., Cao R., Li B., Quan M., Guo L. Metformin ameliorates the development of experimental autoimmune encephalomyelitis by regulating t helper 17 and regulatory t cells in mice. J. Neuroimmunol. 2016;292:58–67. doi: 10.1016/j.jneuroim.2016.01.014. [DOI] [PubMed] [Google Scholar]
- 63.Lee S.Y., Lee S.H., Yang E.J., Kim E.K., Kim J.K., Shin D.Y., Cho M.L. Metformin ameliorates inflammatory bowel disease by suppression of the stat3 signaling pathway and regulation of the between th17/treg balance. PLoS ONE. 2015;10:e0135858. doi: 10.1371/journal.pone.0135858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Waickman A.T., Powell J.D. Mtor, metabolism, and the regulation of t-cell differentiation and function. Immunol. Rev. 2012;249:43–58. doi: 10.1111/j.1600-065X.2012.01152.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Yang K., Blanco D.B., Neale G., Vogel P., Avila J., Clish C.B., Wu C., Shrestha S., Rankin S., Long L., et al. Homeostatic control of metabolic and functional fitness of treg cells by lkb1 signalling. Nature. 2017;548:602–606. doi: 10.1038/nature23665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.He N., Fan W., Henriquez B., Yu R.T., Atkins A.R., Liddle C., Zheng Y., Downes M., Evans R.M. Metabolic control of regulatory t cell (treg) survival and function by lkb1. Proc. Natl. Acad. Sci. USA. 2017;114:12542–12547. doi: 10.1073/pnas.1715363114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Farooqi I.S., Matarese G., Lord G.M., Keogh J.M., Lawrence E., Agwu C., Sanna V., Jebb S.A., Perna F., Fontana S., et al. Beneficial effects of leptin on obesity, t cell hyporesponsiveness, and neuroendocrine/metabolic dysfunction of human congenital leptin deficiency. J. Clin. Investig. 2002;110:1093–1103. doi: 10.1172/JCI0215693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Matarese G., Procaccini C., De Rosa V., Horvath T.L., La Cava A. Regulatory t cells in obesity: The leptin connection. Trends Mol. Med. 2010;16:247–256. doi: 10.1016/j.molmed.2010.04.002. [DOI] [PubMed] [Google Scholar]
- 69.De Rosa V., Procaccini C., La Cava A., Chieffi P., Nicoletti G.F., Fontana S., Zappacosta S., Matarese G. Leptin neutralization interferes with pathogenic t cell autoreactivity in autoimmune encephalomyelitis. J. Clin. Investig. 2006;116:447–455. doi: 10.1172/JCI26523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Matarese G., Di Giacomo A., Sanna V., Lord G.M., Howard J.K., Di Tuoro A., Bloom S.R., Lechler R.I., Zappacosta S., Fontana S. Requirement for leptin in the induction and progression of autoimmune encephalomyelitis. J. Immunol. 2001;166:5909–5916. doi: 10.4049/jimmunol.166.10.5909. [DOI] [PubMed] [Google Scholar]
- 71.Wang X., Qiao Y., Yang L., Song S., Han Y., Tian Y., Ding M., Jin H., Shao F., Liu A. Leptin levels in patients with systemic lupus erythematosus inversely correlate with regulatory t cell frequency. Lupus. 2017;26:1401–1406. doi: 10.1177/0961203317703497. [DOI] [PubMed] [Google Scholar]
- 72.De Rosa V., Procaccini C., Cali G., Pirozzi G., Fontana S., Zappacosta S., La Cava A., Matarese G. A key role of leptin in the control of regulatory t cell proliferation. Immunity. 2007;26:241–255. doi: 10.1016/j.immuni.2007.01.011. [DOI] [PubMed] [Google Scholar]
- 73.Dreyer C., Krey G., Keller H., Givel F., Helftenbein G., Wahli W. Control of the peroxisomal beta-oxidation pathway by a novel family of nuclear hormone receptors. Cell. 1992;68:879–887. doi: 10.1016/0092-8674(92)90031-7. [DOI] [PubMed] [Google Scholar]
- 74.Kliewer S.A., Umesono K., Noonan D.J., Heyman R.A., Evans R.M. Convergence of 9-cis retinoic acid and peroxisome proliferator signalling pathways through heterodimer formation of their receptors. Nature. 1992;358:771–774. doi: 10.1038/358771a0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Choi J.M., Bothwell A.L. The nuclear receptor ppars as important regulators of t-cell functions and autoimmune diseases. Mol. Cells. 2012;33:217–222. doi: 10.1007/s10059-012-2297-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Pawlak M., Lefebvre P., Staels B. Molecular mechanism of pparalpha action and its impact on lipid metabolism, inflammation and fibrosis in non-alcoholic fatty liver disease. J. Hepatol. 2015;62:720–733. doi: 10.1016/j.jhep.2014.10.039. [DOI] [PubMed] [Google Scholar]
- 77.Zhu Y., Ni Y., Liu R., Hou M., Yang B., Song J., Sun H., Xu Z., Ji M. Ppar-gamma agonist alleviates liver and spleen pathology via inducing treg cells during schistosoma japonicum infection. J. Immunol. Res. 2018;2018:6398078. doi: 10.1155/2018/6398078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Cipolletta D., Feuerer M., Li A., Kamei N., Lee J., Shoelson S.E., Benoist C., Mathis D. Ppar-gamma is a major driver of the accumulation and phenotype of adipose tissue treg cells. Nature. 2012;486:549–553. doi: 10.1038/nature11132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Hontecillas R., Bassaganya-Riera J. Peroxisome proliferator-activated receptor gamma is required for regulatory cd4+ t cell-mediated protection against colitis. J. Immunol. 2007;178:2940–2949. doi: 10.4049/jimmunol.178.5.2940. [DOI] [PubMed] [Google Scholar]
- 80.Cipolletta D. Adipose tissue-resident regulatory t cells: Phenotypic specialization, functions and therapeutic potential. Immunology. 2014;142:517–525. doi: 10.1111/imm.12262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Bapat S.P., Myoung Suh J., Fang S., Liu S., Zhang Y., Cheng A., Zhou C., Liang Y., LeBlanc M., Liddle C., et al. Depletion of fat-resident treg cells prevents age-associated insulin resistance. Nature. 2015;528:137–141. doi: 10.1038/nature16151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Stockinger B., Di Meglio P., Gialitakis M., Duarte J.H. The aryl hydrocarbon receptor: Multitasking in the immune system. Annu. Rev. Immunol. 2014;32:403–432. doi: 10.1146/annurev-immunol-032713-120245. [DOI] [PubMed] [Google Scholar]
- 83.Quintana F.J., Basso A.S., Iglesias A.H., Korn T., Farez M.F., Bettelli E., Caccamo M., Oukka M., Weiner H.L. Control of t(reg) and t(h)17 cell differentiation by the aryl hydrocarbon receptor. Nature. 2008;453:65–71. doi: 10.1038/nature06880. [DOI] [PubMed] [Google Scholar]
- 84.Smith K.A. Interleukin-2: Inception, impact, and implications. Science. 1988;240:1169–1176. doi: 10.1126/science.3131876. [DOI] [PubMed] [Google Scholar]
- 85.Sakaguchi S. Naturally arising foxp3-expressing cd25+cd4+ regulatory t cells in immunological tolerance to self and non-self. Nat. Immunol. 2005;6:345–352. doi: 10.1038/ni1178. [DOI] [PubMed] [Google Scholar]
- 86.Mizui M., Tsokos G.C. Low-dose il-2 in the treatment of lupus. Curr. Rheumatol. Rep. 2016;18:68. doi: 10.1007/s11926-016-0617-5. [DOI] [PubMed] [Google Scholar]
- 87.Von Spee-Mayer C., Siegert E., Abdirama D., Rose A., Klaus A., Alexander T., Enghard P., Sawitzki B., Hiepe F., Radbruch A., et al. Low-dose interleukin-2 selectively corrects regulatory t cell defects in patients with systemic lupus erythematosus. Ann. Rheum. Dis. 2016;75:1407–1415. doi: 10.1136/annrheumdis-2015-207776. [DOI] [PubMed] [Google Scholar]
- 88.Rosenzwajg M., Churlaud G., Mallone R., Six A., Derian N., Chaara W., Lorenzon R., Long S.A., Buckner J.H., Afonso G., et al. Low-dose interleukin-2 fosters a dose-dependent regulatory t cell tuned milieu in t1d patients. J. Autoimmun. 2015;58:48–58. doi: 10.1016/j.jaut.2015.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Koreth J., Kim H.T., Jones K.T., Lange P.B., Reynolds C.G., Chammas M.J., Dusenbury K., Whangbo J., Nikiforow S., Alyea E.P., 3rd, et al. Efficacy, durability, and response predictors of low-dose interleukin-2 therapy for chronic graft-versus-host disease. Blood. 2016;128:130–137. doi: 10.1182/blood-2016-02-702852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Matsuoka K., Koreth J., Kim H.T., Bascug G., McDonough S., Kawano Y., Murase K., Cutler C., Ho V.T., Alyea E.P., et al. Low-dose interleukin-2 therapy restores regulatory t cell homeostasis in patients with chronic graft-versus-host disease. Sci. Transl. Med. 2013;5 doi: 10.1126/scitranslmed.3005265. [DOI] [PMC free article] [PubMed] [Google Scholar]