Abstract
Nitric oxide (NO) is a gaseous free radical endogenously generated in plant cells. Peroxisomes are cell organelles characterized by an active metabolism of reactive oxygen species (ROS) and are also one of the main cellular sites of NO production in higher plants. In this mini-review, an updated and comprehensive overview is presented of the evidence available demonstrating that plant peroxisomes have the capacity to generate NO, and how this molecule and its derived products, peroxynitrite (ONOO−) and S-nitrosoglutathione (GSNO), can modulate the ROS metabolism of peroxisomes, mainly throughout protein posttranslational modifications (PTMs), including S-nitrosation and tyrosine nitration. Several peroxisomal antioxidant enzymes, such as catalase (CAT), copper-zinc superoxide dismutase (CuZnSOD), and monodehydroascorbate reductase (MDAR), have been demonstrated to be targets of NO-mediated PTMs. Accordingly, plant peroxisomes can be considered as a good example of the interconnection existing between ROS and reactive nitrogen species (RNS), where NO exerts a regulatory function of ROS metabolism acting upstream of H2O2.
Keywords: catalase, monodehydroascorbate reductase, tyrosine nitration, nitric oxide, peroxisome, reactive oxygen species, S-nitrosation, superoxide dismutase
1. Introduction
Peroxisomes are organelles with an essential oxidative metabolism present in almost all categories of eukaryotic cells. In higher plants, these organelles are recognized to have a versatile metabolism because their enzymatic composition can adapt to different cell and organ types, stages of development, and environmental conditions [1,2,3,4,5,6]. However, there is a common battery of enzymes that are present in all types of plant peroxisomes. This includes a set of antioxidant systems whose functions are to keep under control the internal active metabolism of reactive oxygen species (ROS), mainly superoxide radicals (O2·−) and hydrogen peroxide (H2O2). These ROS are generated under physiological conditions by different pathways, such as purine catabolism, fatty acid β-oxidation, and photorespiration [7,8,9,10]. These antioxidant systems acquire a special relevance in those situations where the ROS generation is intensified, like under plant stress conditions [11].
In recent years, different experimental data have demonstrated that plant peroxisomes also have the capacity to generate another free radical—nitric oxide (NO)—and a family of derived molecules designated as reactive nitrogen species (RNS), including peroxynitrite (ONOO−) [12] and S-nitrosoglutathione (GSNO) [13]. The production of these two families of reactive species—ROS and RNS—raises new questions about their potential functions in peroxisomes, either as simple byproducts of the peroxisomal metabolism or perhaps having a regulatory function in the peroxisome and also outside these organelles, due to the characteristic signaling properties of ROS and RNS.
In this work, the interconnections existing between the metabolism of ROS and RNS in peroxisomes are presented. In this relationship, NO exerts a regulatory function by controlling the activity of some target enzymes through posttranslational modifications (PTMs), mainly S-nitrosation (or S-nitrosylation) and tyrosine nitration. It should be pointed out that the NO-generating capacity of peroxisomes may have significant implications in the cellular metabolism of plants under physiological conditions, including leaf senescence [14], pollen tube growth [15], and auxin-induced root organogenesis [16]. However, peroxisomal NO metabolism is particularly exacerbated under oxidative stress situations induced by abiotic conditions like salinity [17], and the heavy-metals cadmium [12,18], and lead [19].
2. Nitric Oxide Generation in Plant Peroxisomes
In higher plants, NO is a key signaling molecule [20,21] involved in numerous processes, including seed germination [22,23], primary and lateral root growth [24,25], plant development [26,27], stomatal closure [28], flowering [29], reproductive tissues [15,30,31], fruit ripening [32,33], senescence [14,34], abiotic stresses [35,36,37,38,39] and biotic stresses [40]. However, the enzymatic source(s) of NO in plant cells is still a controversial matter subject to intense discussions [41,42,43]. Different pieces of biochemical evidence have demonstrated the presence of L-arginine-dependent nitric oxide synthase (NOS)-like activity in plant peroxisomes. Data accumulated during the last twenty years indicate that the hypothetical protein responsible for the NO generation in peroxisomes has biochemical requirements similar to that of animal NOS, including substrate, cofactors and sensitivity to inhibitors [14,44], dependence on calcium and calmodulin [45], as well as dependence on the mechanism of the import system to peroxisomes through a peroxisomal targeting signal type 2 (PTS-2) [46]. The known biochemical properties of the protein responsible for NO generation in plant peroxisomes, in comparison with those described for animal NOS, are summarized in Table 1. Additionally, there are experimental data that have corroborated the presence of NO in plant peroxisomes and that were obtained by complementary approaches, including electron paramagnetic resonance (EPR) spectroscopy, ozone chemiluminescence, and NO-specific fluorescence probes [14,19]. It should be mentioned that in other cellular compartments a reductive NO generation involving nitrite/nitrate or nitrate reductase (NR) has been described, as well as a non-enzymatic production of NO at acidic pH in the presence of reductants like ascorbate [43,47]. However, peroxisomes have at oxidative metabolism and, to our knowledge, there is not any experimental evidence of the presence of nitrite/nitrate or NR in these plant organelles. Moreover, it has been reported that peroxisomes have an alkaline pH [48], what suggests that the mentioned non-enzymatic generation of NO in peroxisomes is not likely under normal physiological conditions.
Table 1.
Biochemical requirements of the peroxisomal protein responsible for the L-arginine-dependent nitric oxide synthase (NOS)-like activity in higher plants.
| Requirements | Peroxisomal NOS-Like Protein |
|---|---|
| Substrate | L-Arginine |
| Cofactor requirement | NADPH, Ca2+, FAD, FMN, BH4 |
| Sensitivity to inhibitor | Aminiguanidine, L-NNA, L-NAME, L-NMMA |
| Peroxisomal targeting signal (PTS) | Type 2 (PTS2) |
| Dependence of peroxisomal protein import system | PEX5, PEX7, PEX12, PEX13, Ca2+, CaM |
| Localization | Matrix |
BH4, tetrabiopterin; PEX, peroxin; L-NNA, L-NG-Nitroarginine; L-NAME, Nω-Nitro-L-arginine methyl ester hydrochloride; L-NMMA, NG-Monomethyl-L-arginine, monoacetate salt; CaM, calmodulin.
Similarly, in animal peroxisomes, the presence of an inducible NOS isozyme [49,50], which is imported to the peroxisomal matrix using a PTS2 [51], has also been demonstrated. In conclusion, the above data indicated for the protein responsible for NO generation in peroxisomes from plant origin are in good agreement with the data reported for the animal peroxisomal NOS activity.
3. Peroxisomal Proteins: Targets of NO-mediated PTMs
At present, the number of potential targets that undergo NO-mediated PTMs is increasing. This is due to the identifications obtained by specific proteomic methodologies combined with biochemical analyses, such as the biotin switch method and labeling with isotope-coded affinity tags (ICAT). These approaches have also allowed confirming whether a specific protein is S-nitrosated and/or nitrated. In some cases, even the affected amino acid residues of the protein have been identified [52]. Furthermore, the existence of any NO-derived PTM is additional evidence of, at least, the presence of NO and its derived molecules in a specific subcellular compartment [53]. So far, the number of identified plant peroxisomal proteins susceptible to undergo a specific NO-derived PTM has also increased with the development of the mentioned methodologies. The characteristic peroxisomal proteins that have been identified as targets of NO in higher plants are summarized in Table 2. Among the different peroxisomal proteins undergoing NO-derived PTMs, in this article, we have focused on some of the key antioxidant enzymes of peroxisomes, including catalase (CAT), monodehydroascorbate reductase (MDAR), and copper-zinc superoxide dismutase (CuZnSOD).
Table 2.
Some proteins from higher plant peroxisomes that undergo nitric oxide (NO)-derived posttranslational modifications (PTMs), either by S-nitrosation or tyrosine nitration.
| Peroxisomal Enzyme | NO-Derived PTM | References |
|---|---|---|
| 3-ketoacyl-CoA thiolase 1 | S-nitrosation | [52] |
| Hydroxypyruvate reductase | S-nitrosation/nitration | [54,55,56] |
| Glycolate oxidase | S-nitrosation/nitration | [55,57,58] |
| Malate dehydrogenase | S-nitrosation/nitration | [55,59] |
| Catalase | S-nitrosation/nitration | [55,56,60,61] |
| CuZn superoxide dismutase (CSD3) | Nitration | [62] |
| Monodehydroascorbate reductase | S-nitrosation/nitration | [63] |
3.1. Catalase (CAT, EC 1.11.1.6)
CAT is a heme-containing protein and one of the key H2O2-scavenging enzymes present in prokaryotic and eukaryotic cells [64,65,66,67]. Additionally, CAT is recognized as a constitutive enzyme of all kinds of peroxisomes from eukaryotic cells, being used as a biochemical marker of these organelles. The information available, at present, indicates that this enzyme is the main target of NO in animals and plants. In fact, initial in vitro assays showed that the bovine liver CAT was rapidly and reversibly inhibited by NO [68,69]. In plants, using purified tobacco CAT, similar studies demonstrated that both NO donors and ONOO− (a nitrating molecule) had the capacity to inhibit the enzyme activity [70]. More recently, studies carried out in different plant species have shown that CAT is a target of S-nitrosation in sunflower hypocotyls [60], pea leaves [55], and Arabidopsis [56], and of tyrosine nitration in pepper fruits [61]. Moreover, it was demonstrated that both S-nitrosation and tyrosine nitration inhibited CAT activity in pea leaves and pepper fruits [55,61]. It has been proposed that the potential target of S-nitrosation in Arabidopsis CAT is Cys86 [56], although this should be corroborated by specific mass spectrometry analyses. However, it must be taken into account that NO could also interact with the Fe atoms present in the heme groups of CAT, forming a metal nitrosyl complex, that perhaps could affect its activity, although, to our knowledge, there is no information on this mechanism in plant CAT. In any case, all the data available suggest that NO acts upstream of H2O2, thereby regulating CAT activity. This inhibition of CAT by NO could imply a lower capacity to remove H2O2, and consequently it could be well correlated with those physiological or adverse processes that have associated an increase of their oxidative metabolism [18,61].
3.2. Monodehydroascorbate Reductase (MDAR, EC 1.6.5.4)
This enzyme is part of the ascorbate-glutathione (ASC-GSH) cycle, whose function is also to control the cellular content of H2O2 [71]. The ASC-GSH cycle is present in different subcellular compartments, including peroxisomes [72,73,74]. However, very little information is available on how RNS can regulate the specific isozymes of this cycle present in peroxisomes. MDAR catalyzes the NADH-dependent conversion of monodehydroascorbate to ascorbate, and peroxisomal MDAR has been characterized in pea leaves [75] and Arabidopsis [76]. Further in vitro analysis of recombinant MDAR from pea leaf peroxisomes in the presence of nitrating or S-nitrosylating agents (ONOO− or GSNO, respectively) demonstrated that both processes caused inhibition of the MDAR activity [63]. Mass spectrometric analysis and site-directed mutagenesis confirmed that Tyr345 was the primary site of nitration by ONOO− responsible for the inhibition of MDAR activity. On the other hand, in silico analysis of the MDAR indicated that Cys68 was the best candidate for S-nitrosylation [63]. This implies a possible modulation in peroxisomes of the ascorbate regeneration and the H2O2 scavenging by RNS.
3.3. Superoxide Dismutase (SOD; EC 1.15.1.1)
Superoxide dismutases (SODs) are a family of metalloenzymes that catalyze the disproportionation of O2·− radicals into H2O2 and O2. In higher plants, there are three main types of SODs, containing prosthetic metals Mn (Mn-SODs), Fe (Fe-SODs), or Cu plus Zn (Cu,Zn-SODs) [77,78]. The presence of SOD activity in peroxisomes was reported for the first time in plant tissues—in pea (Pisum sativum L.) leaves—in the early 1980s [79]. However, this report, in general, passed unnoticed and was even questioned until it was described in human cells years later [80]. Since then, the occurrence of different types of SODs in plant peroxisomes has been described in at least ten distinct plant species [11,78]. At present, SOD is considered a constitutive enzyme in all types of peroxisomes, although the family of isozyme present depends on the organ and plant species.
In relation to the susceptibility of SOD to different RNS-induced modifications, previous reports indicated that the recombinant human Mn-SOD and Cu,Zn-SOD were prone to be inactivated by ONOO− [81,82]. In the case of plant peroxisomes, recently the recombinant peroxisomal Cu,Zn-SOD (designated as CSD3) was obtained in Arabidopsis, and in vitro assays in the presence of nitrating or S-nitrosylating agents showed that 500 μM ONOO− provoked a 65% inhibition of the Cu,Zn-SOD activity, whereas GSNO did not cause any effect [62]. Regarding mass spectrometric analyses, Tyr115 was identified as the potential target of nitration [62]. Accordingly, SOD seems to be a relevant protein to be further investigated as a target of NO-mediated PTMs, since it appears to be sensitive to exert some discrimination between nitration and nitrosation processes.
4. Conclusions and Future Perspectives
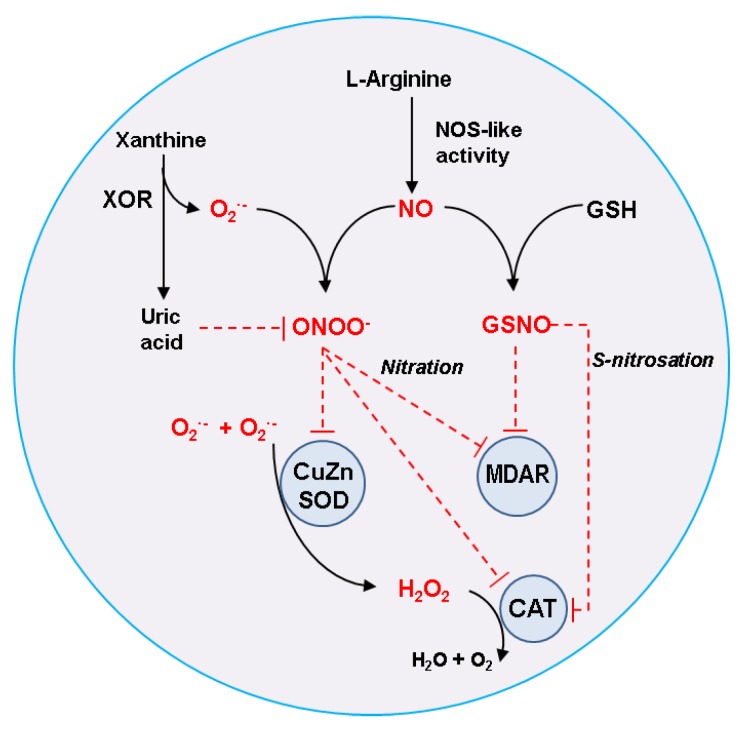
Plant peroxisomes have relevant antioxidant systems comprised mainly of CAT, SOD, and the ASC-GSH cycle, which are present in all types of plant peroxisomes [7]. Likewise, results obtained in previous research works have demonstrated that besides an active ROS metabolism in peroxisomes, these organelles also have an active RNS metabolism. Although there are few specific studies on how distinct RNS can regulate the different peroxisomal antioxidant systems, the data available suggest the NO may act upstream of the H2O2 metabolism. A scheme based on previous reports [7,11,83,84], showing how NO can modulate the activity of peroxisomal antioxidant enzymes throughout either nitration or S-nitrosation, is presented in Figure 1. The peroxisomal xanthine oxidoreductase (XOR) activity catalyzes the oxidation of xanthine with the production of uric acid and O2·− [85]. On the other hand, L-arginine-dependent NOS-like activity generates NO, which can react with O2·− to produce ONOO−, a powerful oxidant and strong nitrating molecule that can mediate PTMs through tyrosine nitration [86]. NO can also interact with reduced glutathione (GSH) to form GSNO, a NO donor that can mediate S-nitrosation of proteins [87]. Uric acid is a recognized inhibitor of ONOO−-mediated toxicity [88,89], and this brings out a new potential mechanism of peroxisomal auto-regulation through this powerful nitrating molecule. In this scenario, the identified targets of NO-derived PTMs in peroxisomes, CAT, CuZnSOD, and MDAR, which are either directly or indirectly linked to the H2O2 pool, are key points to be modulated by nitration or S-nitrosation.
Figure 1.
The interrelationship between nitric oxide (NO) metabolism and antioxidant enzymes in plant peroxisomes. Peroxisomal xanthine oxidoreductase (XOR) activity produces uric acid and superoxide radicals (O2·−). On the other hand, an L-arginine-dependent nitric oxide synthase (NOS)-like activity generates NO, which can react with O2·− to give rise to peroxynitrite (ONOO−), which is a powerful oxidant and strong nitrating molecule that can mediate posttranslational modifications (PTMs), such as tyrosine nitration. NO can also interact with reduced glutathione (GSH) to form S-nitrosoglutathione (GSNO), a NO donor that can mediate S-nitrosation reactions. Uric acid is a recognized ONOO− scavenger that could be part of a mechanism of peroxisomal auto-regulation. With all these components, the identified targets of NO-derived PTMs in peroxisomes, catalase (CAT), copper, zinc superoxide dismutase (CuZnSOD), and monodehydroascorbate reductase (MDAR) can undergo inhibition of their activity either by nitration or S-nitrosation.
In summary, the data presently available indicate that plant peroxisomes contain multiple elements of ROS and RNS metabolism, where NO seems to act upstream of H2O2 routes throughout the regulation of the peroxisomal antioxidant enzymes. Nevertheless, it should be taken into account that both NO and H2O2 could be released to the cytosol, acting as signal molecules among the different subcellular compartments. However, in plants under certain abiotic stress conditions an overproduction of H2O2 and NO could take place in peroxisomes, and a high accumulation of these signal molecules can mediate a nitro-oxidative stress in plant cells [11,90].
Author Contributions
F.J.C. conceived and wrote the manuscript. L.A.d.R. and J.M.P. critically revised the manuscript. All authors approved the final submitted version of the manuscript.
Funding
Research in our laboratory is supported by an ERDF-co-financed grant from the Ministry of Economy and Competitiveness (AGL2015-65104-P) and Junta de Andalucía (group BIO-192), Spain.
Conflicts of Interest
The authors declare no conflict of interest.
References
- 1.Nishimura M., Hayashi M., Kato A., Yamaguchi K., Mano S. Functional transformation of microbodies in higher plant cells. Cell Struct. Funct. 1996;21:387–393. doi: 10.1247/csf.21.387. [DOI] [PubMed] [Google Scholar]
- 2.Hayashi M., Nishimura M. Entering a new era of research on plant peroxisomes. Curr. Opin. Plant Biol. 2003;6:577–582. doi: 10.1016/j.pbi.2003.09.012. [DOI] [PubMed] [Google Scholar]
- 3.Hu J., Baker A., Bartel B., Linka N., Mullen R.T., Reumann S., Zolman B.K. Plant peroxisomes: Biogenesis and function. Plant Cell. 2012;24:2279–2303. doi: 10.1105/tpc.112.096586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Sørhagen K., Laxa M., Peterhänsel C., Reumann S. The emerging role of photorespiration and non-photorespiratory peroxisomal metabolism in pathogen defence. Plant Biol. (Stuttg) 2013;15:723–736. doi: 10.1111/j.1438-8677.2012.00723.x. [DOI] [PubMed] [Google Scholar]
- 5.Goto-Yamada S., Mano S., Yamada K., Oikawa K., Hosokawa Y., Hara-Nishimura I., Nishimura M. Dynamics of the light-dependent transition of plant peroxisomes. Plant Cell Physiol. 2015;56:1264–1271. doi: 10.1093/pcp/pcv081. [DOI] [PubMed] [Google Scholar]
- 6.Kao Y.T., González K.L., Bartel B. Peroxisome function, biogenesis, and dynamics in plants. Plant Physiol. 2018;176:162–177. doi: 10.1104/pp.17.01050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.del Río L.A., Corpas F.J., Sandalio L.M., Palma J.M., Gómez M., Barroso J.B. Reactive oxygen species, antioxidant systems and nitric oxide in peroxisomes. J. Exp. Bot. 2002;53:1255–1272. doi: 10.1093/jxb/53.372.1255. [DOI] [PubMed] [Google Scholar]
- 8.Weber H. Fatty acid-derived signals in plants. Trends Plant Sci. 2002;7:217–224. doi: 10.1016/S1360-1385(02)02250-1. [DOI] [PubMed] [Google Scholar]
- 9.del Río L.A., López-Huertas E. ROS generation in peroxisomes and its role in cell signaling. Plant Cell Physiol. 2016;57:1364–1376. doi: 10.1093/pcp/pcw076. [DOI] [PubMed] [Google Scholar]
- 10.Corpas F.J., del Río L.A., Palma J.M. Plant peroxisomes at the crossroad of NO and H2O2 metabolism. J. Integr. Plant Biol. 2019 doi: 10.1111/jipb.12772. [DOI] [PubMed] [Google Scholar]
- 11.Corpas F.J., Barroso J.B., Palma J.M., Rodríguez-Ruiz M. Plant peroxisomes: A nitro-oxidative cocktail. Redox Biol. 2017;11:535–542. doi: 10.1016/j.redox.2016.12.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Corpas F.J., Barroso J.B. Peroxynitrite (ONOO−) is endogenously produced in Arabidopsis peroxisomes and is overproduced under cadmium stress. Ann. Bot. 2014;113:87–96. doi: 10.1093/aob/mct260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Barroso J.B., Valderrama R., Corpas F.J. Immunolocalization of S-nitrosoglutathione, S-nitrosoglutathione reductase and tyrosine nitration in pea leaf organelles. Acta Physiol Plant. 2013;35:2635–2640. doi: 10.1007/s11738-013-1291-0. [DOI] [Google Scholar]
- 14.Corpas F.J., Barroso J.B., Carreras A., Quirós M., León A.M., Romero-Puertas M.C., Esteban F.J., Valderrama R., Palma J.M., Sandalio L.M., et al. Cellular and subcellular localization of endogenous nitric oxide in young and senescent pea plants. Plant Physiol. 2004;136:2722–2733. doi: 10.1104/pp.104.042812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Prado A.M., Porterfield D.M., Feijó J.A. Nitric oxide is involved in growth regulation and reorientation of pollen tubes. Development. 2004;131:2707–2714. doi: 10.1242/dev.01153. [DOI] [PubMed] [Google Scholar]
- 16.Schlicht M., Ludwig-Müller J., Burbach C., Volkmann D., Baluska F. Indole-3-butyric acid induces lateral root formation via peroxisome-derived indole-3-acetic acid and nitric oxide. New Phytol. 2013;200:473–482. doi: 10.1111/nph.12377. [DOI] [PubMed] [Google Scholar]
- 17.Corpas F.J., Hayashi M., Mano S., Nishimura M., Barroso J.B. Peroxisomes are required for in vivo nitric oxide accumulation in the cytosol following salinity stress of Arabidopsis plants. Plant Physiol. 2009;151:2083–2094. doi: 10.1104/pp.109.146100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Smiri M., Chaoui A., Rouhier N., Gelhaye E., Jacquot J.P., El Ferjani E. Oxidative damage and redox change in pea seeds treated with cadmium. C R Biol. 2010;333:801–807. doi: 10.1016/j.crvi.2010.09.002. [DOI] [PubMed] [Google Scholar]
- 19.Corpas F.J., Barroso J.B. Lead-induced stress, which triggers the production of nitric oxide (NO) and superoxide anion (O2·−) in Arabidopsis peroxisomes, affects catalase activity. Nitric Oxide. 2017;68:103–110. doi: 10.1016/j.niox.2016.12.010. [DOI] [PubMed] [Google Scholar]
- 20.Wilson I.D., Neill S.J., Hancock J.T. Nitric oxide synthesis and signalling in plants. Plant Cell Environ. 2008;31:622–631. doi: 10.1111/j.1365-3040.2007.01761.x. [DOI] [PubMed] [Google Scholar]
- 21.Neill S., Bright J., Desikan R., Hancock J., Harrison J., Wilson I. Nitric oxide evolution and perception. J. Exp Bot. 2008;59:25–35. doi: 10.1093/jxb/erm218. [DOI] [PubMed] [Google Scholar]
- 22.Beligni M.V., Lamattina L. Nitric oxide stimulates seed germination and de-etiolation, and inhibits hypocotyl elongation, three light-inducible responses in plants. Planta. 2000;210:215–221. doi: 10.1007/PL00008128. [DOI] [PubMed] [Google Scholar]
- 23.Bethke P.C., Gubler F., Jacobsen J.V., Jones R.L. Dormancy of Arabidopsis seeds and barley grains can be broken by nitric oxide. Planta. 2004;219:847–855. doi: 10.1007/s00425-004-1282-x. [DOI] [PubMed] [Google Scholar]
- 24.Pagnussat G.C., Lanteri M.L., Lombardo M.C., Lamattina L. Nitric oxide mediates the indole acetic acid induction activation of a mitogen-activated protein kinase cascade involved in adventitious root development. Plant Physiol. 2004;135:279–286. doi: 10.1104/pp.103.038554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Corpas F.J., Barroso J.B. Functions of Nitric Oxide (NO) in roots during development and under adverse stress conditions. Plants (Basel) 2015;4:240–252. doi: 10.3390/plants4020240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sanz L., Albertos P., Mateos I., Sánchez-Vicente I., Lechón T., Fernández-Marcos M., Lorenzo O. Nitric oxide (NO) and phytohormones crosstalk during early plant development. J. Exp Bot. 2015;66:2857–2868. doi: 10.1093/jxb/erv213. [DOI] [PubMed] [Google Scholar]
- 27.Corpas F.J., Barroso J.B., Carreras A., Valderrama R., Palma J.M., León A.M., Sandalio L.M., del Río L.A. Constitutive arginine-dependent nitric oxide synthase activity in different organs of pea seedlings during plant development. Planta. 2006;224:246–254. doi: 10.1007/s00425-005-0205-9. [DOI] [PubMed] [Google Scholar]
- 28.Bright J., Desikan R., Hancock J.T., Weir I.S., Neill S.J. ABA-induced NO generation and stomatal closure in Arabidopsis are dependent on H2O2 synthesis. Plant J. 2006;45:113–122. doi: 10.1111/j.1365-313X.2005.02615.x. [DOI] [PubMed] [Google Scholar]
- 29.Senthil Kumar R., Shen C.H., Wu P.Y., Suresh Kumar S., Hua M.S., Yeh K.W. Nitric oxide participates in plant flowering repression by ascorbate. Sci Rep. 2016;6:35246. doi: 10.1038/srep35246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Prado A.M., Colaço R., Moreno N., Silva A.C., Feijó J.A. Targeting of pollen tubes to ovules is dependent on nitric oxide (NO) signaling. Mol Plant. 2008;1:703–714. doi: 10.1093/mp/ssn034. [DOI] [PubMed] [Google Scholar]
- 31.Zafra A., Rodríguez-García M.I., Alché Jde D. Cellular localization of ROS and NO in olive reproductive tissues during flower development. BMC Plant Biol. 2010;10:36. doi: 10.1186/1471-2229-10-36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Corpas F.J., Freschi L., Rodríguez-Ruiz M., Mioto P.T., González-Gordo S., Palma J.M. Nitro-oxidative metabolism during fruit ripening. J. Exp Bot. 2018;69:3449–3463. doi: 10.1093/jxb/erx453. [DOI] [PubMed] [Google Scholar]
- 33.Corpas F.J., Palma J.M. Nitric oxide on/off in fruit ripening. Plant Biol (Stuttg). 2018;20:805–807. doi: 10.1111/plb.12852. [DOI] [PubMed] [Google Scholar]
- 34.Du J., Li M., Kong D., Wang L., Lv Q., Wang J., Bao F., Gong Q., Xia J., He Y. Nitric oxide induces cotyledon senescence involving co-operation of the NES1/MAD1 and EIN2-associated ORE1 signalling pathways in Arabidopsis. J. Exp. Bot. 2014;65:4051–4063. doi: 10.1093/jxb/ert429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Signorelli S., Corpas F.J., Borsani O., Barroso J.B., Monza J. Water stress induces a differential and spatially distributed nitro-oxidative stress response in roots and leaves of Lotus japonicus. Plant Sci. 2013;201–202:137–146. doi: 10.1016/j.plantsci.2012.12.004. [DOI] [PubMed] [Google Scholar]
- 36.Manai J., Gouia H., Corpas F.J. Redox and nitric oxide homeostasis are affected in tomato (Solanum lycopersicum) roots under salinity-induced oxidative stress. J. Plant Physiol. 2014;171:1028–1035. doi: 10.1016/j.jplph.2014.03.012. [DOI] [PubMed] [Google Scholar]
- 37.Feigl G., Lehotai N., Molnár Á., Ördög A., Rodríguez-Ruiz M., Palma J.M., Corpas F.J., Erdei L., Kolbert Z. Zinc induces distinct changes in the metabolism of reactive oxygen and nitrogen species (ROS and RNS) in the roots of two Brassica species with different sensitivity to zinc stress. Ann Bot. 2015;116:613–625. doi: 10.1093/aob/mcu246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Houmani H., Rodríguez-Ruiz M., Palma J.M., Corpas F.J. Mechanical wounding promotes local and long distance response in the halophyte Cakile maritima through the involvement of the ROS and RNS metabolism. Nitric Oxide. 2018;74:93–101. doi: 10.1016/j.niox.2017.06.008. [DOI] [PubMed] [Google Scholar]
- 39.Kharbech O., Houmani H., Chaoui A., Corpas F.J. Alleviation of Cr(VI)-induced oxidative stress in maize (Zea mays L.) seedlings by NO and H2S donors through differential organ-dependent regulation of ROS and NADPH-recycling metabolisms. J. Plant Physiol. 2017;219:71–80. doi: 10.1016/j.jplph.2017.09.010. [DOI] [PubMed] [Google Scholar]
- 40.Trapet P., Kulik A., Lamotte O., Jeandroz S., Bourque S., Nicolas-Francès V., Rosnoblet C., Besson-Bard A., Wendehenne D. NO signaling in plant immunity: A tale of messengers. Phytochemistry. 2015;112:72–79. doi: 10.1016/j.phytochem.2014.03.015. [DOI] [PubMed] [Google Scholar]
- 41.Santolini J., André F., Jeandroz S., Wendehenne D. Nitric oxide synthase in plants: where do we stand? Nitric Oxide. 2017;63:30–38. doi: 10.1016/j.niox.2016.09.005. [DOI] [PubMed] [Google Scholar]
- 42.Corpas F.J., Barroso J.B. Nitric oxide synthase-like activity in higher plants. Nitric Oxide. 2017;68:5–6. doi: 10.1016/j.niox.2016.10.009. [DOI] [PubMed] [Google Scholar]
- 43.Astier J., Gross I., Durner J. Nitric oxide production in plants: An update. J. Exp. Bot. 2018;69:3401–3411. doi: 10.1093/jxb/erx420. [DOI] [PubMed] [Google Scholar]
- 44.Barroso J.B., Corpas F.J., Carreras A., Sandalio L.M., Valderrama R., Palma J.M., Lupiáñez J.A., del Río L.A. Localization of nitric-oxide synthase in plant peroxisomes. J. Biol. Chem. 1999;274:36729–36733. doi: 10.1074/jbc.274.51.36729. [DOI] [PubMed] [Google Scholar]
- 45.Corpas F.J., Barroso J.B. Calmodulin antagonist affects peroxisomal functionality by disrupting both peroxisomal Ca2+ and protein import. J. Cell Sci. 2018;131 doi: 10.1242/jcs.201467. [DOI] [PubMed] [Google Scholar]
- 46.Corpas F.J., Barroso J.B. Peroxisomal plant nitric oxide synthase (NOS) protein is imported by peroxisomal targeting signal type 2 (PTS2) in a process that depends on the cytosolic receptor PEX7 and calmodulin. FEBS Lett. 2014;588:2049–2054. doi: 10.1016/j.febslet.2014.04.034. [DOI] [PubMed] [Google Scholar]
- 47.Gupta K.J., Igamberdiev A.U. The anoxic plant mitochondrion as a nitrite: NO reductase. Mitochondrion. 2011;11:537–543. doi: 10.1016/j.mito.2011.03.005. [DOI] [PubMed] [Google Scholar]
- 48.Shen J., Zeng Y., Zhuang X., Sun L., Yao X., Pimpl P., Jiang L. Organelle pH in the Arabidopsis endomembrane system. Mol. Plant. 2013;6:1419–1437. doi: 10.1093/mp/sst079. [DOI] [PubMed] [Google Scholar]
- 49.Stolz D.B., Zamora R., Vodovotz Y., Loughran P.A., Billiar T.R., Kim Y.M., Simmons R.L., Watkins S.C. Peroxisomal localization of inducible nitric oxide synthase in hepatocytes. Hepatology. 2002;36:81–93. doi: 10.1053/jhep.2002.33716. [DOI] [PubMed] [Google Scholar]
- 50.Loughran P.A., Stolz D.B., Vodovotz Y., Watkins S.C., Simmons R.L., Billiar T.R. Monomeric inducible nitric oxide synthase localizes to peroxisomes in hepatocytes. Proc. Natl. Acad. Sci. USA. 2005;102:13837–13842. doi: 10.1073/pnas.0503926102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Loughran P.A., Stolz D.B., Barrick S.R., Wheeler D.S., Friedman P.A., Rachubinski R.A., Watkins S.C., Billiar T.R. PEX7 and EBP50 target iNOS to the peroxisome in hepatocytes. Nitric Oxide. 2013;31:9–19. doi: 10.1016/j.niox.2013.02.084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Fares A., Rossignol M., Peltier J.B. Proteomics investigation of endogenous S-nitrosylation in Arabidopsis. Biochem. Biophys. Res. Commun. 2011;416:331–336. doi: 10.1016/j.bbrc.2011.11.036. [DOI] [PubMed] [Google Scholar]
- 53.Heijnen H.F., van Donselaar E., Slot J.W., Fries D.M., Blachard-Fillion B., Hodara R., Lightfoot R., Polydoro M., Spielberg D., Thomson L., et al. Subcellular localization of tyrosine-nitrated proteins is dictated by reactive oxygen species generating enzymes and by proximity to nitric oxide synthase. Free Radic. Biol. Med. 2006;40:1903–1913. doi: 10.1016/j.freeradbiomed.2005.09.006. [DOI] [PubMed] [Google Scholar]
- 54.Corpas F.J., Leterrier M., Begara-Morales J.C., Valderrama R., Chaki M., López-Jaramillo J., Luque F., Palma J.M., Padilla M.N., Sánchez-Calvo B., et al. Inhibition of peroxisomal hydroxypyruvate reductase (HPR1) by tyrosine nitration. Biochim. Biophys. Acta. 2013;1830:4981–4989. doi: 10.1016/j.bbagen.2013.07.002. [DOI] [PubMed] [Google Scholar]
- 55.Ortega-Galisteo A.P., Rodríguez-Serrano M., Pazmiño D.M., Gupta D.K., Sandalio L.M., Romero-Puertas M.C. S-Nitrosylated proteins in pea (Pisum sativum L.) leaf peroxisomes: Changes under abiotic stress. J. Exp. Bot. 2012;63:2089–2103. doi: 10.1093/jxb/err414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Puyaubert J., Fares A., Rézé N., Peltier J.B., Baudouin E. Identification of endogenously S-nitrosylated proteins in Arabidopsis plantlets: effect of cold stress on cysteine nitrosylation level. Plant Sci. 2014;215–216:150–156. doi: 10.1016/j.plantsci.2013.10.014. [DOI] [PubMed] [Google Scholar]
- 57.Abat J.K., Mattoo A.K., Deswal R. S-nitrosylated proteins of a medicinal CAM plant Kalanchoe pinnata- ribulose-1,5-bisphosphate carboxylase/oxygenase activity targeted for inhibition. FEBS J. 2008;275:2862–2872. doi: 10.1111/j.1742-4658.2008.06425.x. [DOI] [PubMed] [Google Scholar]
- 58.Tanou G., Job C., Rajjou L., Arc E., Belghazi M., Diamantidis G., Molassiotis A., Job D. Proteomics reveals the overlapping roles of hydrogen peroxide and nitric oxide in the acclimation of citrus plants to salinity. Plant J. 2009;60:795–804. doi: 10.1111/j.1365-313X.2009.04000.x. [DOI] [PubMed] [Google Scholar]
- 59.Lozano-Juste J., Colom-Moreno R., León J. In vivo protein tyrosine nitration in Arabidopsis thaliana. J. Exp. Bot. 2011;62:3501–3517. doi: 10.1093/jxb/err042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Begara-Morales J.C., López-Jaramillo F.J., Sánchez-Calvo B., Carreras A., Ortega-Muñoz M., Santoyo-González F., Corpas F.J., Barroso J.B. Vinyl sulfone silica: Application of an open preactivated support to the study of transnitrosylation of plant proteins by S-nitrosoglutathione. BMC Plant Biol. 2013;13:61. doi: 10.1186/1471-2229-13-61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Chaki M., Álvarez de Morales P., Ruiz C., Begara-Morales J.C., Barroso J.B., Corpas F.J., Palma J.M. Ripening of pepper (Capsicum annuum) fruit is characterized by an enhancement of protein tyrosine nitration. Ann Bot. 2015;116:637–647. doi: 10.1093/aob/mcv016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Holzmeister C., Gaupels F., Geerlof A., Sarioglu H., Sattler M., Durner J., Lindermayr C. Differential inhibition of Arabidopsis superoxide dismutases by peroxynitrite-mediated tyrosine nitration. J. Exp. Bot. 2015;66:989–999. doi: 10.1093/jxb/eru458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Begara-Morales J.C., Sánchez-Calvo B., Chaki M., Mata-Pérez C., Valderrama R., Padilla M.N., López-Jaramillo J., Luque F., Corpas F.J., Barroso J.B. Differential molecular response of monodehydroascorbate reductase and glutathione reductase by nitration and S-nitrosylation. J. Exp. Bot. 2015;66:5983–5996. doi: 10.1093/jxb/erv306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Mhamdi A., Queval G., Chaouch S., Vanderauwera S., Van Breusegem F., Noctor G. Catalase function in plants: A focus on Arabidopsis mutants as stress-mimic models. J. Exp. Bot. 2010;61:4197–4220. doi: 10.1093/jxb/erq282. [DOI] [PubMed] [Google Scholar]
- 65.Mhamdi A., Noctor G., Baker A. Plant catalases: Peroxisomal redox guardians. Arch. Biochem. Biophys. 2012;525:181–194. doi: 10.1016/j.abb.2012.04.015. [DOI] [PubMed] [Google Scholar]
- 66.Nicholls P. Classical catalase: ancient and modern. Arch. Biochem. Biophys. 2012;525:95–101. doi: 10.1016/j.abb.2012.01.015. [DOI] [PubMed] [Google Scholar]
- 67.Glorieux C., Calderon P.B. Catalase, a remarkable enzyme: Targeting the oldest antioxidant enzyme to find a new cancer treatment approach. Biol. Chem. 2017;398:1095–1108. doi: 10.1515/hsz-2017-0131. [DOI] [PubMed] [Google Scholar]
- 68.Brown G.C. Reversible binding and inhibition of catalase by nitric oxide. Eur. J. Biochem. 1995;232:188–191. doi: 10.1111/j.1432-1033.1995.tb20798.x. [DOI] [PubMed] [Google Scholar]
- 69.Purwar N., McGarry J.M., Kostera J., Pacheco A.A., Schmidt M. Interaction of nitric oxide with catalase: Structural and kinetic analysis. Biochemistry. 2011;50:4491–4503. doi: 10.1021/bi200130r. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Clark D., Durner J., Navarre D.A., Klessig D.F. Nitric oxide inhibition of tobacco catalase and ascorbate peroxidase. Mol. Plant Microbe Interact. 2000;13:1380–1384. doi: 10.1094/MPMI.2000.13.12.1380. [DOI] [PubMed] [Google Scholar]
- 71.Noctor G., Reichheld J.P., Foyer C.H. ROS-related redox regulation and signaling in plants. Semin. Cell Dev. Biol. 2018;80:3–12. doi: 10.1016/j.semcdb.2017.07.013. [DOI] [PubMed] [Google Scholar]
- 72.Jiménez A., Hernández J.A., del Río L.A., Sevilla F. Evidence for the presence of the ascorbate-glutathione cycle in mitochondria and peroxisomes of pea leaves. Plant Physiol. 1997;114:275–284. doi: 10.1104/pp.114.1.275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Kuźniak E., Skłodowska M. Compartment-specific role of the ascorbate-glutathione cycle in the response of tomato leaf cells to Botrytis cinerea infection. J. Exp. Bot. 2005;56:921–933. doi: 10.1093/jxb/eri086. [DOI] [PubMed] [Google Scholar]
- 74.Palma J.M., Jiménez A., Sandalio L.M., Corpas F.J., Lundqvist M., Gómez M., Sevilla F., del Río L.A. Antioxidative enzymes from chloroplasts, mitochondria, and peroxisomes during leaf senescence of nodulated pea plants. J. Exp. Bot. 2006;57:1747–1758. doi: 10.1093/jxb/erj191. [DOI] [PubMed] [Google Scholar]
- 75.Leterrier M., Corpas F.J., Barroso J.B., Sandalio L.M., del Río L.A. Peroxisomal monodehydroascorbate reductase. Genomic clone characterization and functional analysis under environmental stress conditions. Plant Physiol. 2005;138:2111–2123. doi: 10.1104/pp.105.066225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Lisenbee C.S., Lingard M.J., Trelease R.N. Arabidopsis peroxisomes possess functionally redundant membrane and matrix isoforms of monodehydroascorbate reductase. Plant J. 2005;43:900–914. doi: 10.1111/j.1365-313X.2005.02503.x. [DOI] [PubMed] [Google Scholar]
- 77.Fridovich I. Superoxide radical and superoxide dismutases. Annu Rev Biochem. 1995;64:97–112. doi: 10.1146/annurev.bi.64.070195.000525. [DOI] [PubMed] [Google Scholar]
- 78.del Río L.A., Corpas F.J., López-Huertas E., Palma J.M. Plant superoxide dismutases: Function under abiotic stress conditions. In: Gupta D., Palma J., Corpas F., editors. Antioxidants and Antioxidant Enzymes in Higher Plants. Springer; Cham, Switzerland: 2018. pp. 1–26. [Google Scholar]
- 79.del Río L.A., Lyon D.S., Olah I., Glick B., Salin M.L. Immunocytochemical evidence for a peroxisomal localization of manganese superoxide dismutase in leaf protoplasts from a higher plant. Planta. 1983;158:216–224. doi: 10.1007/BF01075257. [DOI] [PubMed] [Google Scholar]
- 80.Keller G.A., Warner T.G., Steimer K.S., Hallewell R.A. Cu,Zn superoxide dismutase is a peroxisomal enzyme in human fibroblasts and hepatoma cells. Proc. Natl. Acad. Sci. USA. 1991;88:7381–8735. doi: 10.1073/pnas.88.16.7381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Alvarez B., Demicheli V., Durán R., Trujillo M., Cerveñansky C., Freeman B.A., Radi R. Inactivation of human Cu,Zn superoxide dismutase by peroxynitrite and formation of histidinyl radical. Free Radic. Biol. Med. 2004;37:813–822. doi: 10.1016/j.freeradbiomed.2004.06.006. [DOI] [PubMed] [Google Scholar]
- 82.Demicheli V., Quijano C., Alvarez B., Radi R. Inactivation and nitration of human superoxide dismutase (SOD) by fluxes of nitric oxide and superoxide. Free Radic. Biol. Med. 2007;42:1359–1368. doi: 10.1016/j.freeradbiomed.2007.01.034. [DOI] [PubMed] [Google Scholar]
- 83.Corpas F.J., Barroso J.B., del Río L.A. Peroxisomes as a source of reactive oxygen species and nitric oxide signal molecules in plant cells. Trends Plant Sci. 2001;6:145–150. doi: 10.1016/S1360-1385(01)01898-2. [DOI] [PubMed] [Google Scholar]
- 84.del Río L.A., Sandalio L.M., Corpas F.J., Palma J.M., Barroso J.B. Reactive oxygen species and reactive nitrogen species in peroxisomes. Production, scanvenging, and role in cell signaling. Plant Physiol. 2006;141:330–335. doi: 10.1104/pp.106.078204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Corpas F.J., Palma J.M., Sandalio L.M., Valderrama R., Barroso J.B., del Río L.A. Peroxisomal xanthine oxidoreductase: Characterization of the enzyme from pea (Pisum sativum L.) leaves. J. Plant Physiol. 2008;165:1319–1330. doi: 10.1016/j.jplph.2008.04.004. [DOI] [PubMed] [Google Scholar]
- 86.Ferrer-Sueta G., Campolo N., Trujillo M., Bartesaghi S., Carballal S., Romero N., Alvarez B., Radi R. Biochemistry of peroxynitrite and protein tyrosine nitration. Chem. Rev. 2018;118:1338–1408. doi: 10.1021/acs.chemrev.7b00568. [DOI] [PubMed] [Google Scholar]
- 87.Broniowska K.A., Diers A.R., Hogg N. S-nitrosoglutathione. Biochim. Biophys. Acta. 2013;1830:3173–3181. doi: 10.1016/j.bbagen.2013.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Alamillo J.M., García-Olmedo F. Effects of urate, a natural inhibitor of peroxynitrite-mediated toxicity, in the response of Arabidopsis thaliana to the bacterial pathogen Pseudomonas syringae. Plant J. 2001;25:529–540. doi: 10.1046/j.1365-313x.2001.00984.x. [DOI] [PubMed] [Google Scholar]
- 89.Signorelli S., Imparatta C., Rodríguez-Ruiz M., Borsani O., Corpas F.J., Monza J. In vivo and in vitro approaches demonstrate proline is not directly involved in the protection against superoxide, nitric oxide, nitrogen dioxide and peroxynitrite. Funct. Plant Biol. 2016;43:870–879. doi: 10.1071/FP16060. [DOI] [PubMed] [Google Scholar]
- 90.del Río L.A., Palma J.M., Sandalio L.M., Corpas F.J., Pastori G.M., Bueno P., López-Huertas E. Peroxisomes as a source of superoxide and hydrogen peroxide in stressed plants. Biochem. Soc. Trans. 1996;24:434–438. doi: 10.1042/bst0240434. [DOI] [PubMed] [Google Scholar]