Abstract
Some of trauma-exposed individuals develop posttraumatic stress disorder (PTSD), an incapacitating psychiatric disorder that is characterized by intrusion, avoidance, negative changes in mood and cognition, and hyperarousal. A number of other trauma-related conditions are very frequently found in individuals with PTSD. Traumatic brain injury (TBI) is one of the most frequently observed trauma-related conditions that trauma-exposed individuals with PTSD may experience. TBI refers to transient or permanent brain dysfunction that results in a wide range of neurological, cognitive, and psychiatric symptoms. These trauma-related conditions significantly affect one’s quality of life, leading to substantial disability and socioeconomic burden. As the prevalence of PTSD with comorbid TBI is increasing in the general population along with the rates of crimes and accidents, effective prevention and intervention strategies are necessitated. However, a definitive treatment for PTSD with comorbid TBI is still lacking, resulting in high rates of treatment resistance and chronicity. It is essential to investigate the neurobiological mechanisms and potential therapeutics of PTSD with comorbid TBI. Yet, a few repetitive transcranial magnetic stimulation (rTMS) studies have recently investigated therapeutic efficacy in treatment-resistant patients with PTSD and/or TBI. Thus, this article reviews rTMS studies in trauma-related conditions, mainly focusing on PTSD and PTSD with TBI as one of the comorbidities. The review focuses on the applications of rTMS in reducing PTSD symptoms with and without comorbidities based on differential parameters and effects of rTMS as well as concomitant clinical conditions. The section on PTSD with comorbidities focuses on TBI with neurological, cognitive, and psychiatric symptoms. Although there were some inconsistencies in the clinical outcomes and optimized parameters of rTMS applied in PTSD and TBI, low frequency stimulation over the hyperactive frontal regions and/or high frequency stimulation over the hypoactive frontal regions generally improved the clinical symptoms of PTSD and TBI. Lastly, the limitations of the rTMS studies in PTSD and TBI as well as potential directions for future research are discussed.
Keywords: trauma, posttraumatic stress disorder, traumatic brain injury, repetitive transcranial magnetic stimulation, brain, neuromodulation
Introduction
Some of trauma-exposed individuals experience posttraumatic stress disorder (PTSD), an incapacitating psychiatric disorder that is characterized by intrusion, avoidance, negative changes in mood and cognition, and hyperarousal after a traumatic experience.1 A number of other trauma-related conditions are increasingly found in individuals with PTSD that include neuropsychiatric disorders such as traumatic brain injury (TBI), major depression, and alcohol use disorders (AUDs).1–3 One of the most frequently observed comorbidities in PTSD is TBI. TBI refers to transient or permanent brain dysfunction that results in a wide range of neurological, cognitive, and psychiatric symptoms after a head trauma and categorized as “mild” or “moderate to severe” depending on its severity.2 The prevalence of PTSD with TBI in the general population has been increasing along with the growing rates of crimes and accidents, necessitating effective interventions.3 Both PTSD and TBI, as posttraumatic conditions, highly overlap and co-occur, suggesting shared etiology and pathophysiology between these two.3 The co-occurrence of PTSD and TBI has fewer treatment options available and increases prognostic factors for chronicity, leading to substantial disability and socioeconomic burden.3
Among the PTSD patients who received standard pharmacotherapy (anticonvulsants, antipsychotics, and antidepressants) and psychotherapy (cognitive–behavioral treatment and eye movement desensitization, and reprocessing), one-third of them remain diagnosed.4,5 Moreover, TBI and related clinical symptoms often show low response and compliance to standard pharmacological and non-pharmacological interventions (exercise and rehabilitation) potentially due to its complicated pathophysiology.6 As a definitive treatment for PTSD with multiple comorbidities is still lacking, treatment of chronic PTSD with comorbid TBI as shown in veterans is particularly challenging.
Repetitive transcranial magnetic stimulation (rTMS) is a noninvasive neuromodulation treatment by rapidly and repetitively changing electromagnetic fields through the coil applied over the scalp.7 It has been increasingly applied in various clinical fields including psychiatry and neurology. Depending on clinical applications, differential rTMS parameters such as target brain region and frequency have been selected for optimized usage. It has been believed that low frequency (LF) rTMS (≤1 Hz) reduces and high frequency (HF) rTMS (>1 Hz) enhances cortical excitability.7,8 The dorsolateral prefrontal cortex (DLPFC) has been a promising target brain region as a hub mainly involved in emotion and cognitive control.9 It has been suggested that differential clinical effects may occur depending on the hemisphere that is targeted by rTMS.10–12 Considering the previously suggested antidepressant effects of rTMS, a growing number of rTMS studies have recently investigated therapeutic efficacy in treatment-resistant PTSD patients with multiple comorbidities.13 As trauma-exposed individuals with PTSD commonly experience TBI, the neurobiological mechanisms of rTMS applications in PTSD with comorbid TBI needs to be investigated. An integrated information on optimized rTMS protocol for PTSD with comorbid TBI may provide a therapeutic benefit in actual clinical settings.
Multimodal neuroimaging has been used to investigate the pathophysiological correlates of PTSD with comorbid TBI, potentially providing a rationale for optimized rTMS parameters (target site and frequency) for effective treatment. Previous functional neuroimaging studies of PTSD have suggested dysfunctional top-down control (characterized by hypoactive prefrontal and hyperactive limbic regions) as the pathophysiological underpinnings.14–16 Functional and structural neuroimaging studies of TBI revealed altered resting functional connectivity in the frontal region as well as microstructural and macrostructural white matter changes that correlate with clinical dysfunction.17–20 The prefrontal cortex including the DLPFC is suggested as the pathophysiological brain correlate of PTSD with comorbid TBI. PTSD and TBI may be explained by dysfunctional cognitive and emotional control of psychological distress and neurological symptoms following trauma exposure.
The aim of the present study was, thus, to critically review the clinical studies of rTMS in trauma-related conditions. The current review is divided into two main sections of PTSD and PTSD with comorbidities mainly focusing on TBI. The section on PTSD is further divided based on the rTMS parameters, concomitant psychotherapy, main clinical effects, and multiple comorbidities. The section on PTSD with comorbidities focuses on TBI with neurological, cognitive, and psychiatric symptoms, considering that these highly co-occurring trauma-related conditions share pathophysiology and clinical manifestations.
Methods
A literature search was conducted in Google Scholar, PubMed/MEDLINE, and Web of Science using the terms that include “rTMS,” “repetitive transcranial magnetic stimulation,” “trauma,” “posttraumatic stress disorder,” “PTSD,” “TBI,” “traumatic brain injury,” “brain injuries,” and “concussion.” The bibliographies of review articles on rTMS in PTSD and TBI published to date were ensured to be included. Two authors (EN and MK) independently performed the search. The third author (SY) discussed the disagreements and led to consensus.
A total of 30 articles published from 2002 to 2018 were initially searched and 15 of them were excluded due to the following reasons: full-text unavailability, duplication, language written, animal studies, poster presentations, and study design (open-label studies in PTSD patients using rTMS, case studies in PTSD patients using rTMS, and case studies in TBI patients using rTMS). The current review includes a total of 15 studies on rTMS applications in PTSD and TBI: 12 randomized-controlled trials (RCTs) that are sham controlled; one case study; and two open-label studies.
rTMS in PTSD
The potential therapeutic efficacy of rTMS in PTSD has been investigated through a series of pilot studies, case studies, and randomized-controlled trials. All rTMS studies of PTSD reviewed in this review are presented in Table 1. The target brain regions and frequency of rTMS applied in the studies are schematically presented in Figure 1. Considering the heterogeneous nature of treatment-resistant PTSD, the first section on rTMS in PTSD is divided into three sections based on concomitant psychotherapy, main clinical effects, and multiple comorbidities. The rTMS on PTSD with multiple comorbidities main focused on TBI with neurological, cognitive, and psychiatric symptoms given the high co-occurrence of PTSD and TBI.
Table 1.
rTMS studies in patients with PTSD
| rTMS for the treatment of PTSD | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Study | PTSD (n) | Group (n) | Methodology | rTMS parameters | Outcomes | ||||
| Site | Frequency (Hz) | Intensity (% MT) | Total sessions | Total pulses | Post rTMS – pre rTMS | ||||
| Nam et al28 | 16 | (A) active (7) (B) sham (9) |
RCT 90° off sham | R DLPFC | 1 | 100 | 15 | 18,000 | [CAPS] ↓ total PTSD severity (A)>(B) – ↓ re-experiencing (A)>(B) |
| Kozel et al29 | 103 | (A) active + CPT (54) (B) sham + CPT (49) |
RCT sham coil | R DLPFC | 1 | 110 | 12–15 | 21,600–27,000 | [CAPS] ↓ PTSD symptoms (A)>(B) |
| Osuch et al30 | 9 | (A) active + imaginal ET (9) (B) sham + imaginal ET (9) |
RCT sham- controlled 45° off sham cross-over | R DLPFC | 1 | 100 | 20 | 36,000 | [CAPS/IES] – ↓ hyperarousal (A)>(B) [↑ 24-hour urinary NE, ↑ T4, ↓ serum prolactin] (A)>(B) |
| Isserles et al31 | 26 | (A) active DTMS + traumatic SI + ET (9) (B) active DTMS + non-traumatic SI + ET (8) (C) sham DTMS + traumatic SI + ET (9) |
RCT sham coil | R & L mPFC | 20 | 120 | 12 | 20,160 | [CAPS] ↓ PTSD severity (A)>(B)>(C) – ↓ intrusion (A)>(B)>(C) [HR] ↓ response to traumatic script (A)>(B)>(C) |
| Boggio et al32 | 26 | (A) active, R DLPFC (9) (B) active, L DLPFC (9) (C) sham (8) |
RCT double-blind sham coil | (A) R DLPFC (B) L DLPFC | 20 | 80 | 10 | 16,000 | [PCL/TOP] ↓ core PTSD symptoms (A)>(B)>(C) – ↓ avoidance (A)>(B) – re-experiencing (A)=(B) – ↓ hyperarousal (A)<(B) [HDRS] ↓ mood (B)>(A) [HARS] ↓ anxiety (A)>(B) |
| Cohen et al33 | 24 | (A) active, 1 Hz, R DLPFC (8) (B) active, 10 Hz, R DLPFC (10) (C) sham, R DLPFC (6) |
RCT double-blind 90° off sham | R DLPFC | (A) 1 (B) 10 |
80 | 10 | (A) 1,000 (B) 4,000 |
[PCL/TOP/CAPS] ↓ core PTSD symptoms (A)=(B)>(C) – ↓ re-experiencing (A)=(B)>(C) – ↓ avoidance (A)=(B)>(C) [HARS] ↓ anxiety (B)>(A)>(C) |
| George et al34 | + mTBI (41) | (A) active (20) (B) sham (21) |
RCT double-blind sham coil | L DLPFC | 10 | 120 | 9 | 54,000 | [SIS] ↓ suicidal ideation Day 1 (A)>(B) Day 2, 3 (A)=(B) |
| Watts et al35 | + PD/OCD (20) | (A) active (10) (B) sham (10) |
RCT double-blind sham coil | R DLPFC | 1 | 90 | 10 | 4,000 | [PCL] ↓ core PTSD symptoms, (A)>(B) – re-experiencing=avoidance=hyperarousal [BDI] ↓ depression symptoms, (A)>(B) [STAI] ↓ anxiety symptoms, (A)>(B) [BNCE] cognitive improvement, (A)=(B) |
Note: The ↑ and ↓ symbols indicate an increase and a decrease, respectively.
Abbreviations: BDI, Beck Depression Inventory; BNCE, Brief Neuropsychological Cognitive Examination; CAPS, Clinician-Administered Posttraumatic Stress Disorder Scale; CPT, cognitive processing therapy; DLPFC, dorsolateral prefrontal cortex; DTMS, deep transcranial magnetic stimulation; ET, exposure therapy; HARS, Hamilton Anxiety Rating Scale; HDRS, Hamilton Depression Rating Scale; HR, heart rate; IES, Impact of Event Scale; L, left; MT, motor threshold; NE, norepinephrine; OCD, obsessive compulsive disorder; PCL, Posttraumatic Stress Disorder Checklist; PD, panic disorder; mPFC, medial prefrontal cortex; PTSD, posttraumatic stress disorder; R, right; RCT, randomized-controlled trial; SI, script-driven imagery; STAI, State-Trait Anxiety Inventory; SIS, Suicide Intention Scale; T4, thyroxine; TOP, Treatment Outcome Posttraumatic Stress Disorder Scale; mTBI, mild traumatic brain injury; rTMS, repetitive transcranial magnetic stimulation.
Figure 1.
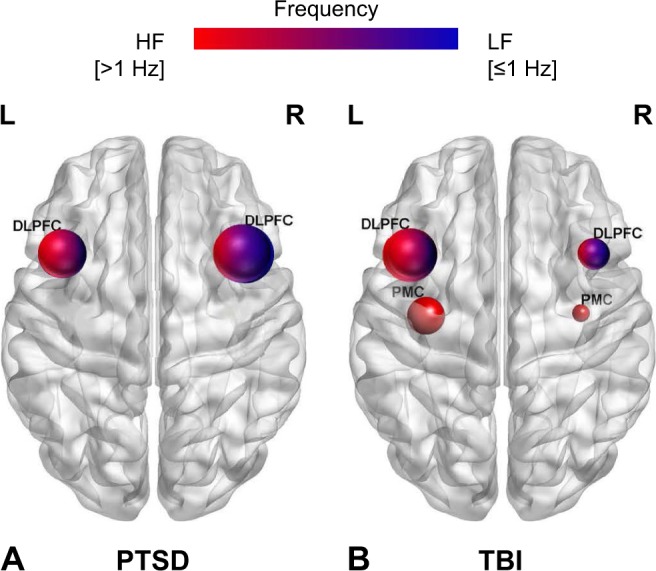
Schematic representation of the target brain regions according to frequency of rTMS among trauma-exposed individuals in the review: (A) PTSD patients and (B) TBI patients.
Notes: The size of the nodes is proportional to the number of rTMS studies allocated to each target brain region. The color of the nodes reflects the frequency of rTMS applied to each target region: high frequency stimulation (>1 Hz) (red color) and low frequency stimulation (≤1 Hz) (blue color).
Abbreviations: DLPFC, dorsolateral prefrontal cortex; HF, high frequency; L, left; LF, low frequency; PMC, primary motor cortex; PTSD, posttraumatic stress disorder; R, right; rTMS, repetitive transcranial magnetic stimulation; TBI, traumatic brain injury.
Previous functional neuroimaging studies of PTSD have suggested dysfunctional top-down control as the pathophysiological underpinning, providing a rationale for optimized rTMS parameters.14–16 Heightened resting connectivity in the salience network (SN) and default mode network (DMN) has been suggested to underlie compromised threat detection system and emotional dysregulation in PTSD.21 This was further supported by lower gray matter density and white matter integrity in the frontal region as indicated by structural neuroimaging in PTSD.14,22 Diffusion tensor imaging reported microstructural and macrostructural abnormalities, suggesting pathological sensory gating in PTSD.23 Interestingly, preferential right hemispheric involvement in adverse emotion and left hemispheric predominance in episodic memory retrieval have been implicated based on the task-dependent hemispheric activation of previous functional neuroimaging studies in PTSD.24–27
For the treatment of PTSD,9,11,28–35 differential combinations of rTMS parameters have been used to augment psychotherapy29–31 and examine therapeutic effects on anxiety vs depression32,33 as well as on PTSD with multiple comorbidities.34,35 Most of the rTMS studies of PTSD have suggested LF stimulation over the right DLPFC as an effective treatment for PTSD,28–30,33,35 particularly in treating anxiety-related hyperarousal.30 HF rTMS, mostly applied over the left DLPFC, showed comparable improvement in PTSD and major depressive disorder (MDD) symptoms.32,34 HF rTMS applied to the right frontal regions32,33 or bilateral frontal regions31 also improved PTSD core symptoms. Generally, the rTMS treatment for PTSD was well-tolerated without serious adverse events.28,29,32 Mild and transient adverse effects included headache, vertigo, uneasiness, and sleep problem.28,29,32
The right DLPFC, primarily involved in the regulation of negative emotion, has been a promising target region for HF and LF rTMS in PTSD.28,33 LF (≤1 Hz) rTMS treatment over the right DLPFC revealed alleviation in reexperiencing and overall severity of PTSD symptoms.28
Chronic PTSD patients tend to be highly unresponsive to standard pharmacotherapy and psychotherapy, possibly due to high comorbidity and heterogeneity. Therefore, PTSD patients who are chronic and treatment-refractory with high comorbidity have been extensively studied in relation to rTMS. This is particularly true for veterans who are repetitively exposed to traumatic experiences.
It has been reported that more than 50% of the PTSD patients develop MDD, suggesting a high comorbidity rate and shared neurobiological mechanisms between these two common psychiatric disorders.36,37 Since PTSD patients with comorbid MDD show relatively poorer outcomes to standard treatment,38–40 rTMS studies have investigated clinical improvement in treatment-resistant PTSD patients with comorbid MDD.
rTMS for the treatment of PTSD with concomitant psychotherapy
rTMS has been applied to augment standard psychotherapy in treatment-refractory and chronic PTSD patients. Specifically, evidence-based psychotherapies such as cognitive processing therapy (CPT) and prolonged exposure therapy have been recommended in longstanding patients with combat-related PTSD. However, two-thirds of them did not fully remit, necessitating additional rTMS to augment standard psychotherapy.41 It has been believed that standard psychotherapy followed by rTMS treatment elicits traumatic experiences and emotions that are targeted by rTMS.
Two LF (≤1 Hz) rTMS studies have been performed on the right DLPFC to augment psychotherapy in chronic PTSD patients.29,30 These results further provided evidence in support of LF rTMS as an effective treatment for PTSD-related adverse emotions.11
Veterans with chronic combat-related PTSD received 12–15 sessions of LF (≤1 Hz) rTMS to the right DLPFC prior to weekly CPT. CPT aims to alleviate negative emotions during elicitation of traumatic experiences. The active group (rTMS + CPT) revealed greater PTSD symptom reductions across CPT sessions and at 6-month posttreatment assessment as compared to the sham group (sham + CPT).29
In line with this, nine treatment-resistant PTSD patients had 20 sessions of LF (≤1 Hz) rTMS to the right DLPFC combined with exposure therapy in a placebo-controlled crossover study. Each session consisted of a 30-minute active or sham rTMS treatment, an imaginal exposure therapy of discussing personally distressing topics, and a 20-minute active or sham rTMS treatment. The active group with concomitant exposure therapy showed greater improvement in the hyperarousal symptom, higher 24-hour urinary norepinephrine, higher serum thyroxin, and lower serum prolactin relative to the sham group with concomitant exposure therapy. This finding indicates physiological and symptomatic treatment effects of active rTMS combined with exposure therapy.30
Isserles et al31 suggested that effectiveness of deep bilateral HF (20 Hz) rTMS preceded by a script-driven exposure therapy depends on the inclusion of traumatic script. Interestingly, lower intrusion symptom and heart rate response were observed only when traumatic script-driven imagery preceded active rTMS treatment. This finding suggests a need for elicitation booktraumatic memory followed by rTMS treatment in alleviating intrusion symptoms. Treatment-resistant PTSD patients were divided into three treatment groups, and they received 12 sessions of deep HF (20 Hz) repeated TMS (DTMS): group A receiving DTMS after traumatic script-driven imagery, group B receiving DTMS after positive experience-driven imagery, and group C receiving sham DTMS after traumatic script-driven imagery. Group A demonstrated post-rTMS improvement in intrusion symptoms relative to groups B and C. In accordance with this finding, increasingly lower heart rate during cued traumatic re-experiencing was observed only in group A. This was further supported by a strong post-rTMS association between heart rate response and PTSD severity scores.31 One patient in the group A had a self-limited tonic-clonic generalized seizure that was transient and did not require any treatment. Taken together, LF rTMS to the right DLPFC with concomitant psychotherapy has improved hyperarousal and overall PTSD symptoms;29,30 HF rTMS to the bilateral medial prefrontal cortex augmented the preceded exposure therapy that used traumatic script-driven imagery, leading to improvement in intrusion symptoms.31
rTMS for the treatment of PTSD with main clinical effects
Two double-blind placebo-controlled trials compared clinical efficacy of rTMS depending on differential rTMS parameters (target sites – the right DLPFC vs left DLPFC and frequency – HF vs LF). Notably, PTSD-related anxiety symptoms improved with HF rTMS over the right DLPFC. PTSD-related depressive symptoms improved with HF rTMS over the left DLPFC and LF rTMS over the right DLPFC.32,33
A double-blind placebo-controlled trial compared therapeutic efficacy of rTMS depending on two different target sites – the right DLPFC vs left DLPFC. Thirty PTSD patients were divided into three groups – the active right DLPFC group, active left DLPFC group, and sham group – and received 10 sessions of HF (20 Hz) rTMS. The right DLPFC group (48.6%) showed clinical efficacy in overall PTSD symptoms more so than the left DLPFC group (22.8%) at the posttreatment assessment. Verbal fluency also improved after the TMS treatment only in the right DLPFC group. Notably, anxiety symptoms improved particularly in the right DLPFC group, while depressive symptoms demonstrated meaningful improvement only in the left DLPFC group, suggesting differential hemispheric involvement in anxiety vs depression related to PTSD.32
Another double-blind placebo-controlled trial compared high (10 Hz) vs low (≤1 Hz) frequency rTMS treatment over the right DLPFC in 24 PTSD patients. It suggested therapeutic efficacy of HF rTMS in alleviating PTSD core symptoms and anxiety symptoms and LF rTMS in improving depressive symptoms. The HF group demonstrated post-rTMS improvement in hyperarousal, reexperiencing, and avoidance symptoms relative to the LF and sham groups. Scores in the PTSD checklist decreased in the high (29.3%) vs low (10.4%) frequency group at posttreatment. Interestingly, the HF group revealed marked improvement in anxiety, but not in depression, as compared to the LF and sham groups. One patient in LF group and one in HF group reported a transient manic episode after receiving rTMS. Other adverse events included muscular discomfort (n=2), transient ear discomfort, dizziness (n=1), and a mild rage attack (n=1) that did not require additional treatments.33 Taken together, HF rTMS to the right DLPFC improved anxiety and overall PTSD symptoms of PTSD, along with verbal fluency, while LF rTMS to the right DLPFC and HF rTMS to the left DLPFC improved depressive symptoms of PTSD.32,33
rTMS for the treatment of PTSD with multiple comorbidities
Due to the heterogeneous nature of psychiatric disorders, PTSD patients with comorbid MDD also had mild TBI (mTBI) and other psychiatric comorbidities including panic disorder (PD), AUD, obsessive-compulsive disorder (OCD), borderline personality disorder (BPD), social anxiety disorder (SAD), and eating disorder. Considering a suggested relationship between comorbidity and treatment resistance in PTSD, rTMS studies have also examined PTSD patients with multiple comorbidities that include depression.34,35
Patients with PTSD, mTBI, or both who had a depressive episode received nine sessions of HF (10 Hz) rTMS over the left DLPFC. The active group transiently showed lower suicidal ideation relative to the sham group only at post-rTMS day 1 vs baseline. One patient discontinued the treatment due to erythema and second-degree burn at the coil stimulation site, but fully recovered with the appropriate treatment.34 PTSD patients with comorbid eating disorder also had other psychiatric disorders including MDD, BPD, PD, and SAD. PTSD patients with comorbid psychiatric disorders including MDD, PD, and OCD demonstrated improvement in PTSD and MDD symptoms after active LF (≤1 Hz) rTMS to the right DLPFC.35 Overall, PTSD patients with multiple comorbidities including depression were treated using various combinations of rTMS parameters.
rTMS in TBI
As the prevalence of PTSD with comorbid TBI is increasing in the general population along with the rates of crimes and accidents, effective prevention and intervention strategies are necessitated. However, the neurobiological underpinnings for PTSD with comorbid TBI have not yet been clearly investigated, resulting in high rates of treatment resistance and chronicity. As the need to deepen the understanding of neurobiological mechanisms about the therapeutic efficacy of rTMS is of utmost importance in treatment-resistant patients with PTSD and/or TBI, we focused on rTMS applications of TBI among PTSD-related comorbidities.
The potential therapeutic efficacy of rTMS in TBI has been investigated through a series of pilot studies, case studies, and randomized-controlled trials. All rTMS studies of TBI reviewed in this review are presented in Table 2. The target brain regions and frequency of the stimulation applied in the studies are schematically presented in Figure 1. The rTMS studies of TBI presented in this review mainly focused on the three most common clinical symptoms following TBI, which are neurological, cognitive, and psychiatric symptoms, in consideration of the complicated pathophysiology of TBI. The section on the TBI-related neurological symptoms are further divided into subsections of headache, central pain, altered consciousness, and tinnitus subsequent to mild or severe TBI, given the high prevalence and multiple subtypes of TBI-related neurological symptoms.
Table 2.
rTMS studies in patients with TBI
| Study | TBI (n) | Methodology | rTMS parameters | Outcomes | ||||
|---|---|---|---|---|---|---|---|---|
| Site | Frequency (Hz) | Intensity (% MT) | Total sessions | Total pulses | Post rTMS – pre rTMS | |||
| rTMS for the treatment of TBI-related neurological symptoms | ||||||||
| Leung et al42 | Post mTBI (6) | Case study | L PMC L DLPFC |
10 | 80 | 4 | 8,000 | [NPRS] ↓ headache |
| Leung et al43 | Post mTBI (active: 15 sham: 16) | RCT 180° off sham neuronavigation |
L PMC | 10 | 80 | 3 | 6,000 | [NPRS] ↓ headache |
| Leung et al44 | Post mTBI (active: 14 sham: 15) | RCT 180° off sham neuronavigation |
L DLPFC | 10 | 80 | 4 | 8,000 | [NPRS] ↓ headache |
| Koski et al45 | 6 mo post mTBI (15) | Open label | L DLPFC | 10 | 110 | 20 | – | [PCSS] ↓ Post-concussion symptoms (headache, sleep deprivation)(BR/)[task fMRI]: ↑ NP task-related activation peaks in the DLPFC and ↑ deactivation in the ACC |
| Choi et al46 | Headache post mTBI (active: 6 sham: 6) | RCT 90° off sham |
PMC | 10 | 90 | 10 | 10,000 | [NPRS] ↓ central pain |
| Manganotti et al47 | Post severe TBI (A) VS: 3 (B) MCS: 3 | Open label | L PMC R PMC |
20 | 120 | 1 | – | [DRS/JFK CRS] coma/consciousness 1 patient (MCS) ↑, 5 patients: pre = post [MEP]: ↑ motor excitability [EEG]: no altered consciousness or slightly ↑ |
| rTMS for the treatment of TBI-related cognitive symptoms | ||||||||
| Leung et al43 | Post mTBI (active: 15 sham: 16) | RCT 180° off sham neuronavigation |
L PMC | 10 | 80 | 3 | 6,000 | [CPT] (=) attention |
| Leung et al44 | Post mTBI (active: 14 sham: 15) | RCT 180° off sham neuronavigation |
L DLPFC | 10 | 80 | 4 | 8,000 | [CPT] (=) attention, concentration, working memory |
| Koski et al45 | 6 mo post mTBI (15) | Open label | L DLPFC | 10 | 110 | 20 | – | [NP battery] ↑ attention, processing speed, executive function, memory [task-fMRI]: ↑ NP task-related activation peaks in the DLPFC & ↑ deactivation in the ACC |
| Neville et al48 | Post severe TBI (active: 18 sham: 18) | RCT double-blind sham coil | L DLPFC | 10 | 110 | 10 | 20,000 | [NP battery] ↑ executive function, attention, memory |
| rTMS for the treatment of TBI-related psychiatric symptoms | ||||||||
| George et al34 | (A) active (20) (B) sham (21) | RCT double-blind sham coil | L DLPFC | 10 | 120 | 9 | 54,000 | [SIS] suicidal ideation (A) = (B) |
| Leung et al43 | Post mTBI (active: 15 sham: 16) | RCT 180° off sham neuronavigation |
L PMC | 10 | 80 | 3 | 6,000 | [HDRS] (=) depression |
| Leung et al44 | Post mTBI (active: 14 sham: 15) | RCT 180° off sham neuronavigation |
L DLPFC | 10 | 80 | 4 | 8,000 | [HDRS] ↓ depression |
Note: The ↑ and ↓ symbols indicate an increase and a decrease, respectively.
Abbreviations: ACC, anterior cingulate cortex; JFK CRS, JFK Coma Recovery Scale; CPT, Conner’s Continuous Performance Test; DLPFC, dorsolateral prefrontal cortex; DRS, Disability Rating Scale; EEG, electroencephalography; fMRI, functional magnetic resonance imaging; HDRS, Hamilton Depression Rating Scale; L, left; MCS, minimally conscious state; MEP, motor evoked potentials; mo, months; MT, motor threshold; NP, neuropsychological test; NPRS, Numeric Pain Rating Scale; PCSS, Post-Concussion Symptom Scale; PMC, primary motor cortex; R, right; RCT, randomized-controlled trial; SIS, Suicide Intention Scale; TBI, traumatic brain injury; mTBI, mild traumatic brain injury; rTMS, repetitive transcranial magnetic stimulation; VS, vegetative state.
Altered resting functional connectivity in the frontal region as well as microstructural and macrostructural white matter changes that correlate with clinical dysfunction underlying TBI may provide insights regarding the optimized rTMS parameters in TBI-related conditions.17–20 Altered functional connectivity in the DMN and lower integrity in the corpus callosum were reported in previous neuroimaging studies of mTBI.17,18 Moderate to severe TBI patients have shown lower resting functional connectivity in the SN and frontoparietal network, indicating dysfunctions in self-awareness and attention, as well as widespread white matter loss.19,20
Most rTMS studies of TBI have focused on the DLPFC as the target brain region as well as HF to facilitate clinical dysfunction. With regard to neurological symptoms, HF rTMS applied over the left DLPFC or left primary motor cortex (PMC) alleviated mTBI-related headache.42–45 HF rTMS also improved central pain related to mTBI46 as well as altered consciousness related to severe TBI.47 HF rTMS over the left hemisphere improved cognitive dysfunction in mTBI45 and severe TBI patients.48 HF rTMS to the left DLPFC improved depression in mTBI.43,44 The rTMS treatment for TBI was generally well-tolerated without serious adverse events; mild and transient adverse events included headache, dizziness, eye twitch, and muscular discomfort.46
rTMS for the treatment of TBI-related neurological symptoms
rTMS has been increasingly applied in TBI-related headache that often becomes treatment-refractory to standard treatment despite its high prevalence. In four recent studies, an active HF (10 Hz) rTMS treatment over the left PMC and/or left DLPFC showed therapeutic efficacy in relieving mTBI-related headache.42–45
A case study reported lower intensity, duration, and frequency of mild TBI headache (mTBI-HA) after HF (10 Hz) rTMS over the left PMC and left DLPFC. Six patients who completed rTMS treatment reported 53.1% decrease in headache intensity and 79% decrease in headache exacerbation frequency. Among the completers, four patients with persistent headache exacerbations reported 50% and 31% reduction in duration and intensity of the exacerbation, respectively. Complete cessation of severe headache episodes was reported by two patients.42 Although HF rTMS to the left PMC and left DLPFC both showed therapeutic efficacy on mTBI-related headache, a different brain pathway was suggested to be involved for each target region.44
To confirm the potential therapeutic effects in the case study, Leung et al43,44 performed two other randomized controlled-trials in veterans with chronic MTBI-HA. The patients received active vs sham HF (10 Hz) rTMS over the left PMC. The active group (58.3%) reported at least 50% lower intensity of headache compared to the sham group (16.6%) at 1-week follow-up assessment. The active group also demonstrated lower scores of functionally debilitating headache relative to the sham group at 4-week follow-up assessment, suggesting long-lasting effects of the rTMS treatment in relieving mTBI-related headache.43
In a most recent study, veterans with MTBI-HA were randomized into the active vs sham group and received HF (10 Hz) rTMS over the left DLPFC. The active group reported 50% and 57% reduction in the prevalence of persistent headache at 1 week and at 2 weeks following the rTMS completion, respectively. In contrast, the sham group reported 7% and 20% reduction at 1-week and 2-week follow-up assessments, respectively.44
mTBI patients with post-concussive symptoms received 20 sessions of HF (10 Hz) rTMS to the left DLPFC in an open-label study. The post-concussive symptoms including headache, depression, sleep deprivation, and cognitive impairment were examined at pre-rTMS, post-rTMS, and post 3-month follow-up assessment. Those who completed the treatment showed improvement in sleep functioning, mental focus, and particularly headache. The adverse events reported included increased sleep problems (n=3), anxiety about the treatment (n=1), an episode of vertigo (n=1), and headache (n=3).45 HF (10 Hz) rTMS to the left PMC and/or left DLPFC have improved mTBI-related headache.42–45
A pilot study suggested that HF (10 Hz) rTMS over the PMC of the affected hemisphere may alleviate mTBI-related central pain, thus improving quality of life of those affected. Twelve mTBI patients with central pain revealed partial injury in the spinothalamocortical tracts. They received either 10 sessions of rTMS treatment or sham rTMS treatment over the PMC of the affected hemisphere. The active group reported lower intensity of central pain and greater improvement in physical and mental health status during and after the rTMS treatment sessions when compared to the sham group.46
Three patients with minimally conscious state (MCS) and other three with vegetative state received a single HF (20 Hz) rTMS session to the bilateral PMC. After the rTMS treatment, only one patient with MCS showed long-lasting improvement in consciousness and motor excitability as manifested by EEG. All other patients showed greater motor excitability, lower motor threshold, as well as a trend toward improvement in consciousness. This finding suggests that a single HF rTMS session may not be enough to restore consciousness in severe TBI.47 Whether severe TBI patients in minimally conscious or vegetative state may restore unconsciousness after HF rTMS remains debated.47
rTMS for the treatment of TBI-related cognitive symptoms
rTMS, mostly HF stimulation over the left hemisphere improved cognitive dysfunction in severe TBI patients.48 Yet, some of the mTBI patients did not show cognitive enhancement after HF rTMS over the left hemisphere.43,44
mTBI patients with somatic, psychiatric, and cognitive impairments received HF (10 Hz) rTMS over the left DLPFC and reported cognitive enhancement in working memory, executive function, processing speed, and attention. Moreover, a task-related functional magnetic resonance imaging showed that this cognitive improvement was related to stronger activation in the DLPFC and deactivation in the anterior cingulate cortex (ACC) during a verbal and nonverbal working memory task at post-rTMS vs pre-rTMS.45
mTBI patients with headache did not report cognitive enhancement after the HF (10 Hz) rTMS treatment to the left DLPFC.44 In line with this, attention preservation transiently improved at 1 week after completing HF (10 Hz) rTMS over the left PMC, but this cognitive enhancement did not last long.43
HF rTMS to the left DLPFC may improve cognitive dysfunction related to severe TBI. Severe TBI patients with diffuse axonal injury underwent 10 sessions of HF (10 Hz) rTMS over the left DLPFC. The completers demonstrated improvement in mood, cortical excitability, and cognition such as executive function and non-verbal memory.48 These results are consistent with the previous HF rTMS studies that have shown cognitive enhancement through stimulation of the hypoactive brain regions.49,50 Overall, HF rTMS to the left frontal regions has improved cognitive dysfunction related to mTBI45 and severe TBI.48
rTMS for the treatment of TBI-related psychiatric symptoms
Three rTMS studies suggested that LF rTMS to the right DLPFC, HF rTMS to the left DLPFC, or both may improve depression in mTBI.34,43,44 These results are in line with the pathophysiological correlates that previous rTMS studies of MDD have suggested.51,52
Leung et al44 suggested transient antidepressant effects of HF (10 Hz) rTMS treatment to the left DLPFC in mTBI patients with headache. The active group showed lower depressive symptoms than the sham group at 1-week follow-up. A trend toward lower depressive symptoms was observed in the active group than in the sham group at 2-week follow-up, suggesting decreasing antidepressant effects over time. However, the two groups did not show differences in PTSD severity at pre-rTMS vs post-rTMS.44 These findings consistent with the HF (10 Hz) rTMS treatment may stimulate and normalize the hypoactive left DLPFC responsible for emotional dysregulation in MDD.53–55 This finding contrasts with the finding that HF (10 Hz) rTMS therapy over the left PMC did not improve depression in mTBI patients with headache.43 These contrasting results of the two studies further add evidence in support of the left DLPFC as the neural correlate underlying depression.43
Yet, suicidal ideation was not improved in mTBI patients with PTSD and a depressive episode, who received HF rTMS to the left DLPFC. Lower suicidal ideation was reported only at day 1 after completing nine sessions of HF (10 Hz) rTMS treatment to the left DLPFC. At 6-month follow-up assessment, no one committed a suicide. The reported adverse events included erythema (n=1) that was appropriately treated and recovered as well as temporary eye pain (n=1) and jaw pain (n=1).34
Conclusion
The present study reviews the applications of rTMS in trauma-related conditions divided into PTSD and PTSD with comorbidities. Among PTSD-related comorbidities, TBI mainly co-occurs with PTSD and may increase prognostic factors for chronicity, leading to substantial disability and socioeconomic burden. This review provides a clue regarding the optimized rTMS parameters for the trauma-exposed individuals with comorbid PTSD and TBI that share common etiology and pathophysiology. It is generally agreed that LF rTMS to the right DLPFC effectively improves PTSD-related anxiety symptoms and HF rTMS to the left DLPFC improves headache, cognitive dysfunction, and depression related to TBI. Thus, the present review suggests the DLPFC to be a promising target brain region for rTMS interventions on trauma-related conditions. The current review supports that the DLPFC plays a pivotal role in emotion and cognitive control of psychological distress and neurological symptoms following trauma exposures.
The present critical review on rTMS applied in trauma-related conditions provides insights into protective and risk factors for the development of PTSD and/or TBI in trauma-exposed individuals. Taken together, rTMS studies reviewed in this article generally demonstrated therapeutic efficacy on PTSD symptoms with multiple comorbidities as well as neurological, cognitive, and psychiatric symptoms related to TBI without serious adverse events. Although there is some inconsistency depending on the characteristics and main symptoms of the patients, rTMS has been mostly applied to normalize the frontal dysfunction related to PTSD and TBI through HF over the hypo-active or LF over the hyper-active brain regions. This may provide a clue regarding the optimized rTMS parameters for the trauma-exposed individuals with comorbid PTSD and TBI.
Clinical outcomes and applied parameters of rTMS have rather been inconsistent, in part, because the pathophysiology underlying PTSD and TBI is multifaceted and complex. Furthermore, differences in sample size, experimental paradigms, comorbidity, concomitant therapy, as well as duration, type, and/or severity of illnesses may potentially explain these discrepancies. Alternatively, future larger longitudinal studies with optimized experimental designs are needed to control these factors. Moreover, an integration of multimodal neuroimaging with rTMS may offer an expansive view of neurobiological mechanisms underlying PTSD and TBI, potentially providing an important insight into the early prevention and intervention. Future systematic review and meta-analysis are warranted to investigate clinical efficacy of rTMS in trauma-related conditions with confidence interval and odds ratio of randomized-controlled trials that used neuroimaging.
Acknowledgments
This research was supported by the Brain Research Program through the National Research Foundation of Korea funded by the Ministry of Science and ICT (2015M3C7A1028373), from the ICT R&D program of Institute for Information & Communications Technology Promotion (B0132-15-1001), by the Field-oriented Support of Fire Fighting Technology Research and Development Program funded by the National Fire Agency (MPSS-Fire Fighting Safety-2016-86), and by the National Institute on Drug Abuse (R01DA024070).
Footnotes
Disclosure
The authors report no conflicts of interest in this work.
References
- 1.American Psychiatric Association . Diagnostic and Statistical Manual of Mental Disorders (DSM-5 TM) 5th ed. Arlington: American Psychiatric Publishing; 2013. [Google Scholar]
- 2.American Congress of Rehabilitation Medicines Definition of mild traumatic brain injury. J Head Trauma Rehabil. 1993;8:86–87. [Google Scholar]
- 3.Vanderploeg RD, Belanger HG, Curtiss G. Mild traumatic brain injury and posttraumatic stress disorder and their associations with health symptoms. Arch Phys Med Rehabil. 2009;90(7):1084–1093. doi: 10.1016/j.apmr.2009.01.023. [DOI] [PubMed] [Google Scholar]
- 4.Kessler RC, Sonnega A, Bromet E, Hughes M, Nelson CB. Posttraumatic stress disorder in the National comorbidity survey. Arch Gen Psychiatry. 1995;52(12):1048–1060. doi: 10.1001/archpsyc.1995.03950240066012. [DOI] [PubMed] [Google Scholar]
- 5.Bisson J, Andrew M. Psychological treatment of post-traumatic stress disorder (PTSD) Cochrane Database Syst Rev. 2007;3:CD003388. doi: 10.1002/14651858.CD003388.pub3. [DOI] [PubMed] [Google Scholar]
- 6.Reti IM, Schwarz N, Bower A, Tibbs M, Rao V. Transcranial magnetic stimulation: a potential new treatment for depression associated with traumatic brain injury. Brain Inj. 2015;29(7–8):789–797. doi: 10.3109/02699052.2015.1009168. [DOI] [PubMed] [Google Scholar]
- 7.George MS, Raman R, Benedek DM, et al. A two-site pilot randomized 3 day trial of high dose left prefrontal repetitive transcranial magnetic stimulation (rTMS) for suicidal inpatients. Brain Stimul. 2014;7(3):421–431. doi: 10.1016/j.brs.2014.03.006. [DOI] [PubMed] [Google Scholar]
- 8.Rosa MA, Lisanby SH. Somatic treatments for mood disorders. Neuropsychopharmacology. 2012;37(1):102–116. doi: 10.1038/npp.2011.225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Philip NS, Barredo J, van’t Wout-Frank M, Tyrka AR, Price LH, Carpenter LL. Network mechanisms of clinical response to transcranial magnetic stimulation in posttraumatic stress disorder and major depressive disorder. Biol Psychiatry. 2018;83(3):263–272. doi: 10.1016/j.biopsych.2017.07.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Iannone A, Cruz Apdem, Brasil-Neto JP, Boechat-Barros R. Transcranial magnetic stimulation and transcranial direct current stimulation appear to be safe neuromodulatory techniques useful in the treatment of anxiety disorders and other neuropsychiatric disorders. Arq Neuropsiquiatr. 2016;74(10):829–835. doi: 10.1590/0004-282X20160115. [DOI] [PubMed] [Google Scholar]
- 11.Tillman GD, Kimbrell TA, Calley CS, Kraut MA, Freeman TW, Hart J., Jr Repetitive transcranial magnetic stimulation and threat memory: selective reduction of combat threat memory p300 response after right frontal-lobe stimulation. J Neuropsychiatry Clin Neurosci. 2011;23(1):40–47. doi: 10.1176/jnp.23.1.jnp40. [DOI] [PubMed] [Google Scholar]
- 12.Wassermann EM, Lisanby SH. Therapeutic application of repetitive transcranial magnetic stimulation: a review. Clin Neurophysiol. 2001;112(8):1367–1377. doi: 10.1016/s1388-2457(01)00585-5. [DOI] [PubMed] [Google Scholar]
- 13.Herrold AA, Kletzel SL, Harton BC, Chambers RA, Jordan N, Pape TL. Transcranial magnetic stimulation: potential treatment for co-occurring alcohol, traumatic brain injury and posttraumatic stress disorders. Neural Regen Res. 2014;9(19):1712–1730. doi: 10.4103/1673-5374.143408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Rauch SL, Shin LM, Phelps EA. Neurocircuitry models of posttraumatic stress disorder and extinction: human neuroimaging research-past, present, and future. Biol Psychiatry. 2006;60(4):376–382. doi: 10.1016/j.biopsych.2006.06.004. [DOI] [PubMed] [Google Scholar]
- 15.Brewin CR. What is it that a neurobiological model of PTSD must explain? Prog Brain Res. 2008;167:217–228. doi: 10.1016/S0079-6123(07)67015-0. [DOI] [PubMed] [Google Scholar]
- 16.Koenigs M, Huey ED, Raymont V, et al. Focal brain damage protects against post-traumatic stress disorder in combat veterans. Nat Neurosci. 2008;11(2):232–237. doi: 10.1038/nn2032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Johnson B, Zhang K, Gay M, et al. Alteration of brain default network in subacute phase of injury in concussed individuals: resting-state fMRI study. Neuroimage. 2012;59(1):511–518. doi: 10.1016/j.neuroimage.2011.07.081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zhou Y, Milham MP, Lui YW, et al. Default-mode network disruption in mild traumatic brain injury. Radiology. 2012;265(3):882–892. doi: 10.1148/radiol.12120748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ham TE, Bonnelle V, Hellyer P, et al. The neural basis of impaired self-awareness after traumatic brain injury. Brain. 2014;137(2):586–597. doi: 10.1093/brain/awt350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sharp DJ, Beckmann CF, Greenwood R, et al. Default mode network functional and structural connectivity after traumatic brain injury. Brain. 2011;134(8):2233–2247. doi: 10.1093/brain/awr175. [DOI] [PubMed] [Google Scholar]
- 21.Koch SB, van Zuiden M, Nawijn L, Frijling JL, Veltman DJ, Olff M. Aberrant resting-state brain activity in posttraumatic stress disorder: a meta-analysis and systematic review. Depress Anxiety. 2016;33(7):592–605. doi: 10.1002/da.22478. [DOI] [PubMed] [Google Scholar]
- 22.Kim MJ, Lyoo IK, Kim SJ, et al. Disrupted white matter tract integrity of anterior cingulate in trauma survivors. Neuroreport. 2005;16(10):1049–1053. doi: 10.1097/00001756-200507130-00004. [DOI] [PubMed] [Google Scholar]
- 23.Olson EA, Cui J, Fukunaga R, Nickerson LD, Rauch SL, Rosso IM. Disruption of white matter structural integrity and connectivity in posttraumatic stress disorder: a TBSS and tractography study. Depress Anxiety. 2017;34(5):437–445. doi: 10.1002/da.22615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rauch SL, van der Kolk BA, Fisler RE, et al. A symptom provocation study of posttraumatic stress disorder using positron emission tomography and script-driven imagery. Arch Gen Psychiatry. 1996;53(5):380–387. doi: 10.1001/archpsyc.1996.01830050014003. [DOI] [PubMed] [Google Scholar]
- 25.Simmons A, Matthews SC, Stein MB, Paulus MP. Anticipation of emotionally aversive visual stimuli activates right insula. Neuroreport. 2004;15(14):2261–2265. doi: 10.1097/00001756-200410050-00024. [DOI] [PubMed] [Google Scholar]
- 26.Whalley MG, Kroes MC, Huntley Z, Rugg MD, Davis SW, Brewin CR. An fMRI investigation of posttraumatic flashbacks. Brain Cogn. 2013;81(1):151–159. doi: 10.1016/j.bandc.2012.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Nardo D, Högberg G, Flumeri F, et al. Self-rating scales assessing subjective well-being and distress correlate with rCBF in PTSD-sensitive regions. Psychol Med. 2011;41(12):2549–2561. doi: 10.1017/S0033291711000912. [DOI] [PubMed] [Google Scholar]
- 28.Nam DH, Pae CU, Chae JH. Low-frequency, repetitive transcranial magnetic stimulation for the treatment of patients with posttraumatic stress disorder: a double-blind, sham-controlled study. Clin Psychopharmacol Neurosci. 2013;11(2):96–102. doi: 10.9758/cpn.2013.11.2.96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kozel FA, Motes MA, Didehbani N, et al. Repetitive TMS to augment cognitive processing therapy in combat veterans of recent conflicts with PTSD: a randomized clinical trial. J Affect Disord. 2018;229:506–514. doi: 10.1016/j.jad.2017.12.046. [DOI] [PubMed] [Google Scholar]
- 30.Osuch EA, Benson BE, Luckenbaugh DA, Geraci M, Post RM, Mccann U. Repetitive TMS combined with exposure therapy for PTSD: a preliminary study. J Anxiety Disord. 2009;23(1):54–59. doi: 10.1016/j.janxdis.2008.03.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Isserles M, Shalev AY, Roth Y, et al. Effectiveness of deep transcranial magnetic stimulation combined with a brief exposure procedure in post-traumatic stress disorder-a pilot study. Brain Stimul. 2013;6(3):377–383. doi: 10.1016/j.brs.2012.07.008. [DOI] [PubMed] [Google Scholar]
- 32.Boggio PS, Rocha M, Oliveira MO, et al. Noninvasive brain stimulation with high-frequency and low-intensity repetitive transcranial magnetic stimulation treatment for posttraumatic stress disorder. J Clin Psychiatry. 2010;71(8):992–999. doi: 10.4088/JCP.08m04638blu. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Cohen H, Kaplan Z, Kotler M, Kouperman I, Moisa R, Grisaru N. Repetitive transcranial magnetic stimulation of the right dorsolateral prefrontal cortex in posttraumatic stress disorder: a double-blind, placebo-controlled study. Am J Psychiatry. 2004;161(3):515–524. doi: 10.1176/appi.ajp.161.3.515. [DOI] [PubMed] [Google Scholar]
- 34.George MS, Raman R, Benedek DM, et al. A two-site pilot randomized 3 day trial of high dose left prefrontal repetitive transcranial magnetic stimulation (rTMS) for suicidal inpatients. Brain Stimul. 2014;7(3):421–431. doi: 10.1016/j.brs.2014.03.006. [DOI] [PubMed] [Google Scholar]
- 35.Watts BV, Landon B, Groft A, Young-Xu Y. A sham controlled study of repetitive transcranial magnetic stimulation for posttraumatic stress disorder. Brain Stimul. 2012;5(1):38–43. doi: 10.1016/j.brs.2011.02.002. [DOI] [PubMed] [Google Scholar]
- 36.Rytwinski NK, Scur MD, Feeny NC, Youngstrom EA. The co-occurrence of major depressive disorder among individuals with posttraumatic stress disorder: a meta-analysis. J Trauma Stress. 2013;26(3):299–309. doi: 10.1002/jts.21814. [DOI] [PubMed] [Google Scholar]
- 37.Flory JD, Yehuda R. Comorbidity between post-traumatic stress disorder and major depressive disorder: alternative explanations and treatment considerations. Dialogues Clin Neurosci. 2015;17(2):141–150. doi: 10.31887/DCNS.2015.17.2/jflory. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Holtzheimer PE, Russo J, Zatzick D, Bundy C, Roy-Byrne PP. The impact of comorbid posttraumatic stress disorder on short-term clinical outcome in hospitalized patients with depression. Am J Psychiatry. 2005;162(5):970–976. doi: 10.1176/appi.ajp.162.5.970. [DOI] [PubMed] [Google Scholar]
- 39.Campbell DG, Felker BL, Liu CF, et al. Prevalence of depression-PTSD comorbidity: implications for clinical practice guidelines and primary care-based interventions. J Gen Intern Med. 2007;22(6):711–718. doi: 10.1007/s11606-006-0101-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Dunner DL, Aaronson ST, Sackeim HA, et al. A multisite, naturalistic, observational study of transcranial magnetic stimulation for patients with pharmacoresistant major depressive disorder: durability of benefit over a 1-year follow-up period. J Clin Psychiatry. 2014;75(12):1394–1401. doi: 10.4088/JCP.13m08977. [DOI] [PubMed] [Google Scholar]
- 41.Steenkamp MM, Litz BT, Hoge CW, Marmar CR. Psychotherapy for military-related PTSD: a review of randomized clinical trials. JAMA. 2015;314(5):489–500. doi: 10.1001/jama.2015.8370. [DOI] [PubMed] [Google Scholar]
- 42.Leung A, Fallah A, Shukla S, et al. rTMS in alleviating mild TBI related headaches–a case series. Pain Physician. 2016;19(2):E347–E354. [PubMed] [Google Scholar]
- 43.Leung A, Shukla S, Fallah A, et al. Repetitive transcranial magnetic stimulation in managing mild traumatic brain injury-related headaches. Neuromodulation: Technology at the Neural Interface. 2016;19(2):133–141. doi: 10.1111/ner.12364. [DOI] [PubMed] [Google Scholar]
- 44.Leung A, Metzger-Smith V, He Y, et al. Left dorsolateral prefrontal cortex rTMS in alleviating MTBI related headaches and depressive symptoms. Neuromodulation. 2018;21(4):390–401. doi: 10.1111/ner.12615. [DOI] [PubMed] [Google Scholar]
- 45.Koski L, Kolivakis T, Yu C, Chen JK, Delaney S, Ptito A. Noninvasive brain stimulation for persistent postconcussion symptoms in mild traumatic brain injury. J Neurotrauma. 2015;32(1):38–44. doi: 10.1089/neu.2014.3449. [DOI] [PubMed] [Google Scholar]
- 46.Choi GS, Kwak SG, Lee HD, Chang MC. Effect of high-frequency repetitive transcranial magnetic stimulation on chronic central pain after mild traumatic brain injury: a pilot study. J Rehabil Med. 2018;50(3):246–252. doi: 10.2340/16501977-2321. [DOI] [PubMed] [Google Scholar]
- 47.Manganotti P, Formaggio E, Storti SF, et al. Effect of high-frequency repetitive transcranial magnetic stimulation on brain excitability in severely brain-injured patients in minimally conscious or vegetative state. Brain Stimul. 2013;6(6):913–921. doi: 10.1016/j.brs.2013.06.006. [DOI] [PubMed] [Google Scholar]
- 48.Neville IS, Hayashi CY, El Hajj SA, et al. Repetitive transcranial magnetic stimulation (rTMS) for the cognitive rehabilitation of traumatic brain injury (TBI) victims: study protocol for a randomized controlled trial. Trials. 2015;16(1):440. doi: 10.1186/s13063-015-0944-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Levkovitz Y, Rabany L, Harel EV, Zangen A. Deep transcranial magnetic stimulation add-on for treatment of negative symptoms and cognitive deficits of schizophrenia: a feasibility study. Int J Neuropsychopharmacol. 2011;14(7):991–996. doi: 10.1017/S1461145711000642. [DOI] [PubMed] [Google Scholar]
- 50.Drumond Marra HL, Myczkowski ML, Maia Memória C, et al. Transcranial magnetic stimulation to address mild cognitive impairment in the elderly: a randomized controlled study. Behavioural Neurology. 2015;2015(1):1–13. doi: 10.1155/2015/287843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Brunoni AR, Chaimani A, Moffa AH, et al. Repetitive transcranial magnetic stimulation for the acute treatment of major depressive episodes: a systematic review with network meta-analysis. JAMA Psychiatry. 2017;74(2):143–152. doi: 10.1001/jamapsychiatry.2016.3644. [DOI] [PubMed] [Google Scholar]
- 52.Philip NS, Barredo J, Aiken E, Carpenter LL. Neuroimaging mechanisms of therapeutic transcranial magnetic stimulation for major depressive disorder. Biol Psychiatry Cogn Neurosci Neuroimaging. 2018;3(3):211–222. doi: 10.1016/j.bpsc.2017.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Baeken C, De Raedt R, van Hove C, Clerinx P, De Mey J, Bossuyt A. HF-rTMS treatment in medication-resistant melancholic depression: results from 18FDG-PET brain imaging. CNS Spectr. 2009;14(8):439–448. doi: 10.1017/s1092852900020411. [DOI] [PubMed] [Google Scholar]
- 54.Liston C, Chen AC, Zebley BD, et al. Default mode network mechanisms of transcranial magnetic stimulation in depression. Biol Psychiatry. 2014;76(7):517–526. doi: 10.1016/j.biopsych.2014.01.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Kang JI, Lee H, Jhung K, et al. Frontostriatal connectivity changes in major depressive disorder after repetitive transcranial magnetic stimulation: a randomized sham-controlled study. J Clin Psychiatry. 2016;77(9):e1137–e1143. doi: 10.4088/JCP.15m10110. [DOI] [PubMed] [Google Scholar]


