Abstract
Background
Brain metastases represent a significant healthcare problem. It is estimated that 20% to 40% of patients with cancer will develop metastatic cancer to the brain during the course of their illness. The burden of brain metastases impacts on quality and length of survival. Presenting symptoms include headache (49%), focal weakness (30%), mental disturbances (32%), gait ataxia (21%), seizures (18%), speech difficulty (12%), visual disturbance (6%), sensory disturbance (6%) and limb ataxia (6%).
Brain metastases may spread from any primary site. The most common primary site is the lung, followed by the breast then gastrointestinal sites. Eighty‐five per cent of brain metastases are found in the cerebral hemispheres, 10% to 15% in the cerebellum and 1% to 3% in the brainstem. Brain radiotherapy is used to treat cancer participants who have brain metastases from various primary malignancies.
This is an update to the original review published in Issue 3, 2006.
Objectives
To assess the effectiveness and adverse effects of whole brain radiotherapy (WBRT) in adult participants with multiple metastases to the brain.
Search methods
We searched the Cochrane Central Register of Controlled Trials (CENTRAL) (Issue 3, 2011), MEDLINE and EMBASE to July 2011.
Selection criteria
Randomized controlled trials (RCTs) comparing WBRT either alone or with other treatments in adults with newly diagnosed multiple metastases to the brain from any primary cancer. Trials of prophylactic WBRT were excluded as well as trials that dealt with surgery or WBRT, or both, for the treatment of single brain metastasis.
Data collection and analysis
Two review authors independently assessed trial quality and abstracted information. Adverse effects information was also collected from the trials.
Main results
Nine RCTs involving 1420 participants were added in this updated review. This updated review now includes a total of 39 trials involving 10,835 participants.
Eight published reports (nine RCTs) showed no benefit of altered dose‐fractionation schedules as compared to the control fractionation (3000 cGy in 10 fractions daily) of WBRT for overall survival. These studies also showed no improvement in symptom control nor neurologic improvement among the different dose‐fractionation schemes as compared to 3000 cGy in 10 daily fractions of WBRT. This updated review includes two trials comparing 4000 cGy in 20 fractions given twice daily versus 2000 cGy in 4 or 5 daily fractions. Overall, there was no survival advantage (hazard ratio (HR) 1.18, 95% confidence interval (CI) 0.89 to 1.56, P = 0.25) with the use of 4000 cGy in 20 fractions given twice daily compared to 2000 cGy in 4 or 5 daily fractions.
The addition of radiosensitizers in six RCTs did not confer additional benefit to WBRT in either the overall survival times (HR 1.08, 95% CI 0.98 to 1.18, P = 0.11) or brain tumour response rates (HR 0.87, 95% CI 0.60 to 1.26, P = 0.46).
Two RCTs found no benefit in overall survival (HR 0.61, 95% CI 0.27 to 1.39, P = 0.24) with the use of WBRT and radiosurgery boost as compared to WBRT alone for selected participants with multiple brain metastases (up to four brain metastases). Overall, there was a statistically significant improvement in local brain control (HR 0.35, 95% CI 0.20 to 0.61, P = 0.0003) favouring the WBRT and radiosurgery boost arm. Only one trial of radiosurgery boost with WBRT reported an improved Karnofsky performance score outcome and improved ability to reduce the dexamethasone dose.
In this updated review, a total of three RCTs reported on selected patients (with up to three or four brain metastases) treated with radiosurgery alone versus WBRT and radiosurgery. Based on two trials, there was no difference in overall survival (HR 0.98, 95% CI 0.71 to 1.35, P = 0.88). The addition of WBRT when added to radiosurgery significantly improved locally treated brain metasatases control (HR 2.61, 95% CI 1.68 to 4.06, P < 0.0001) and distant brain control (HR 2.15, 95% CI 1.55 to 2.99, P < 0.00001). On the other hand, one trial concluded that patients treated with WBRT and radiosurgery boost were significantly more likely to show a decline in learning and memory function as compared to those treated with radiosurgery alone.
One RCT examined the use of WBRT and prednisone versus prednisone alone and produced inconclusive results.
Authors' conclusions
None of the RCTs with altered WBRT dose‐fractionation schemes as compared to standard (3000 cGy in 10 daily fractions or 2000 cGy in 4 or 5 daily fractions) found a benefit in terms of overall survival, neurologic function, or symptom control.
The use of radiosensitizers or chemotherapy in conjunction with WBRT remains experimental.
Radiosurgery boost with WBRT may improve local disease control in selected participants as compared to WBRT alone, although survival remains unchanged for participants with multiple brain metastases.
This updated review now includes a total of three RCTs examining the use of radiosurgery alone versus WBRT and radiosurgery. The addition of WBRT to radiosurgery improves local and distant brain control but there is no difference in overall survival. Patients treated with radiosurgery alone were found to have better neurocognitive outcomes in one trial as compared to patients treated with WBRT and radiosurgery.
The benefit of WBRT as compared to supportive care alone has not been studied in RCTs. It may be that supportive care alone, without WBRT, is appropriate for some participants, particularly those with advanced disease and poor performance status.
Keywords: Adult, Humans, Brain Neoplasms, Brain Neoplasms/radiotherapy, Brain Neoplasms/secondary, Brain Neoplasms/surgery, Combined Modality Therapy, Combined Modality Therapy/methods, Cranial Irradiation, Cranial Irradiation/methods, Dose Fractionation, Radiation‐Sensitizing Agents, Radiation‐Sensitizing Agents/therapeutic use, Radiosurgery, Radiosurgery/methods, Randomized Controlled Trials as Topic, Survival Analysis, Treatment Outcome
Whole brain radiotherapy for the treatment of multiple brain metastases
Radiotherapy is commonly used to treat patients with cancer that has spread to the brain. The aim of this review was to determine the effectiveness and adverse effects of whole brain radiotherapy (WBRT) alone or in combination with other treatments in adult patients with multiple brain metastases. Thirty‐nine trials involving 10,835 participants were included following the update in 2012. There does not appear to be any additional benefit of altered WBRT dose schedules compared to standard doses. The use of chemotherapy or radiosensitizers in conjunction with WBRT has not yet been shown to confer any additional benefit. Radiosurgery boost with WBRT does not improve survival in selected patients with multiple brain metastases but local control may be improved with the addition of radiosurgery boost to WBRT. WBRT when added to radiosurgery improves local and distant brain control but neurocognitive outcomes may be better in patients treated with radiosurgery alone as compared to WBRT and radiosurgery.
Background
This review is an update of a previously published review in the Cochrane Database of Systematic Reviews (The Cochrane Library 2006, Issue 3) on 'Whole brain radiotherapy for the treatment of multiple brain metastases'.
Brain metastases represent a significant healthcare problem. It is estimated that 20% to 40% of participants with cancer will develop metastatic cancer to the brain during the course of their illness (Loeffler 1997). The burden of brain metastases impacts on quality and length of survival. Presenting symptoms include headache (49%), focal weakness (30%), mental disturbances (32%), gait ataxia (21%), seizures (18%), speech difficulty (12%), visual disturbance (6%), sensory disturbance (6%) and limb ataxia (6%) (Posner 1995).
Brain metastases may spread from any primary site. The most common primary site is the lung, followed by the breast then gastrointestinal sites (Walker 1985). Eighty‐five per cent of brain metastases are found in the cerebral hemispheres, 10% to 15% in the cerebellum and 1% to 3% in the brainstem (Arbit 1995).
The mainstay of treatment for brain metastases has been corticosteroids (for the treatment of peritumoral edema) and whole brain radiotherapy (WBRT).
WBRT has been shown to improve neurological symptoms and function with minimal morbidity (Chao 1954; Posner 1977). Non‐randomized studies suggest that WBRT increases median survival up to three to six months (Chao 1954; Katz 1981; Posner 1977; Zimm 1981). Median survival of participants with symptomatic brain metastases is approximately one month without treatment and two months with corticosteroid use. The overall response rate to whole brain radiotherapy is dependent on the symptoms reported, and ranges from 64% to 85% (Borgelt 1980; Katz 1981; Sneed 1996). However, the definition of symptom response has on the whole been vaguely defined in studies. In one study, 74% of participants showed improvement in neurologic symptoms and 65% maintained this improvement for at least nine months (Cairncross 1980).
Even with treatment, brain metastases still cause significant morbidity and mortality. Despite WBRT, up to a half of participants will die from intracranial progression (Borgelt 1980; Chao 1954; Gelber 1981; Katz 1981; Noordijk 1994). As the majority of participants with metastatic cancer to the brain have, or soon develop, widely disseminated disease, in many cases their survival is dependent upon their overall response to cancer treatment and not on treatment for their brain metastases alone.
In an attempt to improve outcomes, studies have examined the use of WBRT combined with systemic treatment (chemotherapy) or radiosensitizers (agents which enhance the radiation effect). Altered dose‐fractionation schedules have also been studied. Surgery has been combined with WBRT for single metastasis (Grant 2001) and radiosurgery (a specialized, focused radiation technique) has been used for selected participants. The benefits and harms of these therapies were assessed in that systematic review. Studies that examined the use of surgery or WBRT, or both, for single brain metastases were excluded, as this is the subject of another Cochrane systematic review (Hart 2004).
Objectives
The aim of this review was to assess the effectiveness of WBRT either alone or in combination with other therapies in adult participants with newly diagnosed multiple brain metastases and to assess any adverse effects from these treatments.
Methods
Criteria for considering studies for this review
Types of studies
Trials of participants with newly diagnosed multiple metastases to the brain who were randomized to treatment with WBRT either alone or with other therapies were included in this review.
Types of participants
Adult participants receiving WBRT for newly diagnosed multiple metastases to the brain from any primary cancer were eligible for inclusion.
Types of interventions
Trials that compared the following interventions were eligible for inclusion:
altered WBRT dose‐fractionation schedules versus conventional WBRT fractionation schedules (*3000 cGy in 10 fractions or 2000 cGy in 4 or 5 fractions);
WBRT* and systemic therapy;
WBRT plus radiosensitizers versus WBRT*;
WBRT plus radiosurgery versus WBRT*;
radiosurgery alone versus radiosurgery and WBRT*;
steroids alone versus WBRT and steroids*.
*control arm
Trials of prophylactic WBRT in which WBRT was used without evidence of existing brain metastases were excluded. Studies that examined the use of surgery or WBRT, or both, for single brain metastases were also excluded as this is the subject of another Cochrane systematic review (Hart 2004). This review excludes management for patients with recurrent brain metastases.
Types of outcome measures
Data for the following outcome measures were sought:
overall survival;
intracranial progression‐free duration (defined as the time from randomization or entry to the trial until progressive brain disease was diagnosed (i.e., enlarging brain metastases or the development of new brain metastases based on a contrast‐enhanced computed tomography (CT) or magnetic resonance imaging (MRI) scan);
brain response (reported as the percentage of participants achieving a complete response (CR) or partial response (PR) of existing brain metastases to treatment. Complete response was defined as complete radiographic disappearance of the brain metastases. Partial response was defined as more than 50% decrease in size of the brain metastases on CT or MRI);
local brain control (reported as the percentage of participants with unchanged or improved serial post‐treatment CT or MRI scans judged as either a complete response (CR), partial response (PR) or stable disease (SD) with improving or stable neurologic symptoms or neurologic examination. SD was defined as a 0% to 50% decrease in size of all lesions with stabilization of neurologic symptoms or neurologic examination and stable dexamethasone dose. Progressive disease was defined as an increase in the size of any lesion);
distant brain control (defined as CR, PR or SD of brain metastases not treated with radiosurgery);
quality of life assessed by any scale;
symptom control;
neurologic function;
proportion of participants who were able to reduce their daily dexamethasone dose and duration of reduced dexamethasone requirements;
adverse effects.
Search methods for identification of studies
Electronic searches
Review update
In July 2011, we revised and updated the search strategies. The following databases were searched: CENTRAL (The Cochrane Library Issue 3, 2011), MEDLINE (to week 3 July 2011), EMBASE (to week 3 July 2011). Refer to Appendix 1, Appendix 2 and Appendix 3 for the updated search strategies. Only published reports in English were searched.
Searching other resources
We handsearched the references of included studies to identify additional studies.
Data collection and analysis
Study selection
Two radiation oncologists (MNT and EC) assessed the titles and abstracts retrieved by the search strategy. We obtained the full published reports for all references deemed to meet the inclusion criteria. We also retrieved articles if it was felt that the article's reference list should be reviewed for additional relevant studies.
Assessment of the full reports enabled the identification of studies for inclusion in the review. Those that were excluded at this stage are listed in the 'Characteristics of excluded studies' table and the reasons for exclusion stated.
Assessors were not blinded to author, institution, journal of publication or results as the review authors were familiar with most studies and the typographical layout of journals.
Data extraction and management
Two independent review authors (MNT and NL) extracted data using standard data extraction forms. Any disagreements or discrepancies were resolved by a third assessor (RW).
We extracted the following data items.
Study characteristics.
Patient characteristics.
Interventions.
Outcome data including:
overall survival;
intracranial progression‐free duration (defined as the time from randomization or entry to the trial until progressive brain disease was diagnosed (i.e., enlarging brain metastases or the development of new brain metastases based on a contrast‐enhanced computed tomography (CT) or magnetic resonance imaging (MRI) scan);
brain response (reported as the percentage of participants achieving a complete response (CR) or partial response (PR) of existing brain metastases to treatment. Complete response was defined as complete radiographic disappearance of the brain metastases. Partial response was defined as more than 50% decrease in size of the brain metastases on CT or MRI);
local brain control (reported as the percentage of participants with unchanged or improved serial post‐treatment CT or MRI scans judged as either a complete response (CR), partial response (PR) or stable disease (SD) with improving or stable neurologic symptoms or neurologic examination. SD was defined as a 0% to 50% decrease in size of all lesions with stabilization of neurologic symptoms or neurologic examination and stable dexamethasone dose. Progressive disease was defined as an increase in the size of any lesion;
distant brain control (defined as CR, PR or SD of brain metastases not treated with radiosurgery);
quality of life assessed by any scale;
symptom control; neurologic function;
proportion of participants who were able to reduce their daily dexamethasone dose and duration of reduced dexamethasone requirements;
adverse effects;
study withdrawals, patient deaths, losses to follow up.
Assessment of methodological quality of included studies
Two review authors (MNT and EC) independently assessed the quality of the studies using the criteria described in the Oxford Quality Scale (Jadad 1996). The Oxford Quality Scale produces a maximum score of five, based on three domains (randomization process, whether the study was double blind, and whether all study participants were accounted for).
However, because trials involving WBRT were likely to lack double blinding and thus limit the usefulness of the Oxford Quality Scale, the quality assessment tool described by Detsky 1989 was used in parallel. This scale uses five domains: randomization process, outcome assessment, inclusion and exclusion criteria, details of intervention, and appropriateness of statistics. Seven items are scored using 'Yes or No' responses, seven items are scored using 'Yes, Partial, No' responses and one item addresses the study's power and is applicable for negative trials. For the purposes of this review, 'Yes' is given a score of 1, 'No' is given a score of 0 and 'Partial' a score of 0.5.
Sensitivity analysis
We planned to undertake a sensitivity analysis using scores of methodological quality. The quality scores that were derived were used to identify the strengths or weaknesses of trial designs, or both, and enable the effect of study quality on outcomes to be assessed.
Dichotomous data and continuous data
This updated review includes the following updated statistical analyses.
For the pooled analysis of overall survival, local control and distant brain control, the generic inverse variance method and fixed‐effect model in Review Manager (RevMan5) was used. Data pooling for overall survival, local control and distant control were estimated using the Hazard Ratio Meta‐analysis Box (Parmar 1998).
For the pooled analysis of brain tumour response, we abstracted the proportion of participants with a complete or partial response from the tables, figures or text of published reports. We determined tumour response using the proportion of participants achieving a complete response (CR) or partial response (PR). These data were treated as dichotomous outcome measures.
Neurologic function was described as the proportion of participants with improved neurologic function and was treated as a dichotomous outcome.
Due to the heterogeneity of instruments used and differences in reporting, we described and did not pool quality of life, symptom control and adverse effects outcomes.
Missing data
We abstracted study withdrawals, patient deaths and losses to follow up. All analyses were conducted on an intention‐to‐treat basis.
Assessment of heterogeneity
The Radiation Therapy Oncology Group (RTOG) analyzed the database of 1200 participants from three consecutive RTOG trials which examined several dose fractionation schemes of WBRT and the use of radiosensitizers with WBRT (Gaspar 1997). Using recursive partitioning analysis (RPA), three prognostic classes based on survival were generated, Class 1: participants with Karnofsky performance status (KPS) ≥ 70, < 65 years of age with controlled primary and no extracranial metastases; Class 3: KPS < 70; Class 2: all others. These classes enable the classification of patient populations with brain metastases to be separated into homogeneous patient populations based on the survival outcome.
Other patient characteristics predicting other outcomes of interest (intracranial progression‐free survival, quality of life, symptom control, neurologic function, ability to taper down on dexamethasone dose) have not previously been defined.
We extracted patient characteristics based on age, KPS and status of extracranial disease from the trials. Treatment characteristics were also extracted. If patient populations or treatment characteristics were deemed to be heterogeneous, we did not pool the data for meta‐analysis.
Data synthesis
If pooling of the data was deemed to be appropriate, we used the statistical package RevMan 5.1. In this updated review, we reported hazard ratios (HR) with 95% confidence intervals (CI) for the outcomes of overall survival, local control, and distant brain control. Risk ratios (RR) with 95% CI using the random‐effects model were reported for neurocognitive improvement and brain tumour response.
We extracted data from full published trials. Data from abstracts could not be pooled as these reports lacked the detail to permit pooling of outcomes. Analyses were based on the reported abstract. Authors of these abstracts were not contacted for further information.
Subgroup analysis
No subgroup analyses were planned.
Results
Description of studies
Nine new trials (Chang 2009; Davey 2008; Graham 2010; Knisely 2008; Kocher 2011; Lee 2008; Neuhaus 2009; Scott 2007; Suh 2008) involving 1420 participants were added in this updated review. This updated review now includes a total of 39 trials involving 10,835 participants.
Altered WBRT schedules
A total of nine published reports involved participants randomized to altered WBRT dose‐fractionation schedules compared to standard 3000 cGy in 10 daily fractions (Borgelt 1980; Borgelt 1981; Chatani 1985; Chatani 1994; Haie‐Meder 1993; Harwood 1977; Kurtz 1981; Murray 1997; Priestman 1996). One study (Haie‐Meder 1993) was excluded because the trial design did not include a standard WBRT dose‐fractionation arm (3000 cGy in 10 fractions or 2000 cGy in five fractions).
This left eight reports (on altered WBRT dose‐fractionation schedules) which met inclusion criteria for this review (3645 participants).
The published report by Borgelt 1980 included the results of two sequential trials (Study 1 and Study 2). Participants were randomized to one of five WBRT schedules ranging from 4000 cGy/4 weeks to 2000 cGy/1 week. Study 1 (Borgelt 1980a) randomized participants to one of four regimens (3000 cGy/2 weeks, 3000 cGy/3 weeks, 4000 cGy/3 weeks, 4000 cGy/4 weeks). Study 2 (Borgelt 1980b) randomized participants to one of three regimens (2000 cGy/1 week, 3000 cGy/2 weeks, 4000 cGy/3 weeks).
Borgelt 1981 reported on participants randomized to 1000 cGy/1 fraction in Study 1 (Borgelt 1981a) or 1200 cGy/2 fractions in Study 2 (Borgelt 1981b) as compared to participants treated on one of five schedules ranging from 4000 cGy/4 weeks to 2000 cGy/1 week. Thus, there is some duplication of patient reporting in Borgelt 1980 and Borgelt 1981 for participants treated with one of five WBRT schedules ranging from 4000 cGy/4 weeks to 2000 cGy/1 week. However, Borgelt 1981 reported on participants treated with 1000 cGy/1 fraction or 1200 cGy/2 fractions, not included in the Borgelt 1980 report. It was not possible to separate the subgroup of participants duplicated in the Borgelt 1980 and Borgelt 1981 reports. As such, the data for these reports were entered as completely separate groups of participants.
Chatani 1985 and Chatani 1994 reported on separate groups of participants randomized to trials that were enrolled sequentially, 1980 to 1983 (Chatani 1985) and 1985 to 1992 (Chatani 1994). There was also no duplication of participants reported in the other trials (Harwood 1977; Kurtz 1981; Murray 1997; Priestman 1996).
Neurocognitive outcomes in the trial participants from Murray 1997 were subsequently reported by Regine 2001.
This updated review now includes two RCTs examining the use of 4000 cGy in 20 twice daily fractions of WBRT versus 2000 cGy in 4 or 5 daily fractions of WBRT (Davey 2008; Graham 2010).
WBRT with or without radiosensitizers
Six fully published trials (DeAngelis 1989; Eyre 1984; Komarnicky 1991; Mehta 2003; Phillips 1995; Suh 2006) examined the use of radiosensitizers in addition to WBRT (2016 participants). The radiosensitizers used were lonidamide (DeAngelis 1989), metronidazole (Eyre 1984), misonidazole (Komarnicky 1991), bromodeoxyuridine (BrdU) (Phillips 1995), motexafin gadolinium (Mehta 2003) and efaproxiral (Suh 2006). Another published trial on the same participants as in the Suh 2006 trial was reported by Stea 2006, in which the prognostic significance of the radiographic response was explored. Suh 2006 reported on survival as the primary endpoint with response rate as the secondary endpoint.
Since the last published version of this review, a post hoc subgroup analysis of breast cancer patients treated wtih the radiosensitizer efaproxiral and WBRT versus WBRT was published (Scott 2007). Subsequently Suh 2008 reported, in abstract form, a trial which a priori included only breast cancer patients randomized to WBRT plus efaproxiral versus WBRT alone.
Mehta 2003 reported on survival and neurologic outcomes in patients randomized to WBRT and motexafin gadolinium as compared to WBRT alone. A follow‐up report by Meyers 2004 reported specifically on neurocognitive outcomes on the same group of participants that were randomized in the motexafin gadolium trial by Mehta 2003.
WBRT and chemotherapy
Since the last update, three trials (Knisely 2008; Lee 2008; Neuhaus 2009) have been added.
Nine studies (975 participants) examined the use of chemotherapy and WBRT (Antonadou 2002; Guerrieri 2004; Knisely 2008; Lee 2008; Mornex 2003; Neuhaus 2009; Postmus 2000; Robinet 2001; Ushio 1991). Eight studies were fully published reports (Guerrieri 2004; Knisely 2008; Lee 2008; Mornex 2003; Neuhaus 2009; Postmus 2000; Robinet 2001; Ushio 1991). The Antonadou 2002 trial has been published in abstract form.
Antonadou 2002 randomized participants to WBRT with or without temozolomide chemotherapy. Guerrieri 2004 randomized participants with non‐small cell lung cancer metastatic to brain to either WBRT alone or WBRT and concomitant carboplatin chemotherapy. Mornex 2003 randomized participants with metastatic malignant melanoma to brain to fotemustine and WBRT versus fotemustine alone. Postmus 2000 examined participants with metastatic small cell lung cancer to brain and randomized these participants to teniposide versus teniposide and WBRT. Robinet 2001 reported on participants with non‐small cell lung cancer metastatic to brain and randomized these participants to early versus delayed WBRT with concurrent cisplatin and vinorelbine chemotherapy. Ushio 1991 randomized participants with metastatic lung cancer to brain to either WBRT alone versus WBRT and chloroethylnitrosoureas versus WBRT and chloroethylnitrosoureas and tegafur.
WBRT with or without radiosurgery boost
Three trials (Andrews 2004; Chougule 2000; Kondziolka 1999) have been reported examining the use of WBRT with or without radiosurgery boost for up to four brain metastases (469 participants in total). Two trials are fully published (Andrews 2004; Kondziolka 1999). The Kondziolka 1999 trial included participants with two to four brain metastases (all 25 mm in diameter or less). Andrews 2004 reported on participants with one to three brain metastases, with a maximum diameter of 4 cm for the largest lesion and additional lesions not exceeding 3 cm in diameter. The Chougule 2000 trial has been published in abstract form. This trial included participants with one to three brain metastases, tumor volume of 30 cc or less and minimum life expectancy of three months.
Radiosurgery alone versus radiosurgery and WBRT
Since the last update, two additional RCTs have been fully published (Chang 2009; Kocher 2011).
Aoyama 2006 randomized 132 participants with one to four brain metastases (3 cm or less in size) to either radiosurgery alone or radiosurgery and WBRT. Chang 2009 randomized 58 patients with one to three brain metastases to radiosurgery alone versus WBRT and radiosurgery. Kocher 2011 reported on 359 patients randomized to radiosurgery alone versus WBRT and radiosurgery or surgery versus surgery and WBRT.
Steroids with or without WBRT
One trial (48 participants) reported on the use of oral prednisone with or without WBRT (Horton 1971).
Risk of bias in included studies
We could only assess the quality of fully published trials. Due to lack of detail, quality scores could not be assigned for trials published in abstract form.
Studies comparing different dose‐fractionation schedules of WBRT
The 10 reports examining altered WBRT dose‐fractionation schedules achieved the following scores on the Oxford Quality Scale (Jadad 1996) for methodological quality.
Quality score 4: none. Quality score 3: Davey 2008; Graham 2010; Kurtz 1981; Priestman 1996. Quality score 2: Borgelt 1980; Borgelt 1981; Harwood 1977; Murray 1997. Quality score 1: Chatani 1985; Chatani 1994.
All studies were described as randomized, but three studies (Chatani 1994; Chatani 1985; Harwood 1977) did not report the method of randomization. None of the studies were double‐blind. Five studies (Davey 2008; Graham 2010; Harwood 1977; Kurtz 1981; Priestman 1996) described withdrawals and drop‐outs in terms of numbers and reasons per group.
The Detsky score (Detsky 1989) ranged from eight to 10.5 (out of a possible maximum score of 15) for these 10 publications. None of the negative trials reported confidence intervals or post hoc power calculations. None of the trials were double‐blind. While the outcome of survival is objective, outcomes such as symptom control and neurologic function class are prone to subjective interpretation.
Studies comparing WBRT with or without radiosensitizers
The six fully published trials examining WBRT with or without radiosensitizers achieved the following scores on the Oxford Quality Scale (Jadad 1996) for methodological quality.
Quality score 4: none. Quality score 3: Komarnicky 1991; Phillips 1995; Suh 2006. Quality score 2: Eyre 1984; Mehta 2003. Quality score 1: DeAngelis 1989.
All studies were described as randomized except two studies (DeAngelis 1989; Eyre 1984) which did not report the method of randomization. None of the studies were double‐blind. Two studies (DeAngelis 1989; Mehta 2003) did not describe withdrawals or drop‐outs.
The Detsky score for these five trials ranged from 10 to 13 out of a possible maximum score of 15. None of the negative trials reported confidence intervals or post hoc power calculations. The outcomes of survival and brain tumor response were objective. None of the trials reported on sample size justification except for the Mehta 2003 trial.
Studies comparing WBRT and systemic therapy
The eight fully published reports comparing WBRT with and without systemic therapy (chemotherapy) achieved the following scores using the Oxford scale (Jadad 1996) for methodological quality.
Quality score 4: none. Quality score 3: Knisely 2008; Lee 2008; Neuhaus 2009; Robinet 2001. Quality score 2: Ushio 1991. Quality score 1: Guerrieri 2004; Mornex 2003; Postmus 2000.
The Detsky scores for these four trials ranged from 10 to 11 out of a possible maximum score of 15.
All studies were described as randomized but two studies (Mornex 2003; Postmus 2000) did not report the method of randomization. None of the studies were double‐blind. Four trials (Knisely 2008; Lee 2008; Neuhaus 2009; Robinet 2001) reported on withdrawals and drop‐outs in terms of numbers and reasons per group. The outcome measures of survival and brain tumor response were objective. Ushio 1991 did not report sample size justification. The Guerrieri 2004 trial was closed early because of poor accrual (42 participants randomized and reported on out of a planned accrual of 300 participants). The Knisely 2008 trial was also closed early because of poor accrual (183 patients randomized). A protocol‐planned interim analysis showed that the Knisely 2008 trial had extremely low probability of ever showing a significant difference favoring the radiosensitizer arm.
Studies comparing WBRT with or without radiosurgery
For the two fully published trials comparing WBRT with or without radiosurgery, the scores using the Oxford Quality Scale (Jadad 1996) for methodological quality were as follows.
Quality score 4: none. Quality score 3: Andrews 2004. Quality score 2: none. Quality score 1: Kondziolka 1999.
The Detsky score was 11 out of 15 for both trials. Neither trial was double‐blinded. Withdrawals and drop‐outs were described. The outcome measures of survival and brain tumor response were objective and sample size justification was reported.
Study comparing radiosurgery alone versus radiosurgery and WBRT
For the three fully published trials comparing radiosurgery alone versus WBRT and radiosurgery, the scores using the Oxford Quality Scale (Jadad 1996) for methodological quality were as follows.
Quality score 4: none.
Quality score 3: Aoyama 2006; Chang 2009; Kocher 2011.
Quality score 2: none.
Quality score 1: none.
The Detsky score was 13 for all of these trials. None of the trials were double‐blinded. Withdrawals and drop‐outs were described. The outcome measures of survival and neurocognitive function were objective. However, duration of functional independence (the primary endpoint for the Kocher 2011 trial) was subjective. Sample size justifications were reported.
Studies comparing steroids with or without WBRT
One study was found for this comparison and the score using the Oxford Quality Scale (Jadad 1996) for methodological quality was two. The Detsky score was three. This was an older trial carried out in the pre‐CT era. Diagnoses of brain metastases were made with outdated techniques. The sample size was small (48 participants) with no sample size justification and statistical analysis of outcomes was not performed.
Sensitivity analysis was performed using the Detsky score. Studies and their corresponding hazard ratios (HR) or risk ratios (RR) were presented in ascending order in the sensitivity analysis. This allowed the effect of study quality on outcomes to be explored visually in the forest plots (see comparisons 05 (01 to 06)). Overall, outcomes were insensitive to study quality.
Effects of interventions
The outcomes for each of the interventions are reported below.
Altered WBRT dose‐fractionation schedules versus conventional WBRT fractionation schedule (control: 3000 cGy in 10 daily fractions or 2000 cGy in 4 or 5 daily fractions)
The eight reports (Borgelt 1980; Borgelt 1981; Chatani 1985; Chatani 1994; Harwood 1977; Kurtz 1981; Murray 1997; Priestman 1996) examining the effectiveness of different dose‐fractionation schedules of WBRT provided data for statistical analysis. Regine 2001 reported on the neurocognitive outcome in brain metastases participants treated in the Murray 1997 trial of accelerated fractionation versus accelerated hyperfractionated WBRT.
Dose response was the primary outcome for this comparison. In order to evaluate dose response many different dose‐fractionation schedules were compared. The most commonly employed 'control' regimen was 3000 cGy in 10 daily fractions. The concept of 'biological equivalent dose' (BED) was used to facilitate comparison between different dose‐fractionation regimens.
BED can be calculated using the equation BED = nd [1+d/(alpha/beta)], where n = number of fractions, d = dose per fraction, and alpha/beta = 10 for tumor (Hall 2000). For the purpose of assessing dose response, studies were divided into those comparing lower biological doses compared to 3000 cGy in 10 daily fractions and higher biological doses compared with 3000 cGy in 10 daily fractions. In order to explore if a dose response relationship was present, the relative biological effectiveness (RBE) = 39 Gy for the fractionation scheme of 3000 cGy in 10 daily fractions was used as control. Outcome comparisons were made between RBE < 39 Gy versus 39 Gy and 39 Gy versus > 39 Gy.
With regard to the outcomes of interest, none of the trials reported on:
intracranial progression‐free duration;
local brain control (CR + PR + SD);
local brain tumour response (CR + PR);
quality of life;
the proportion of participants able to reduce their dexamethasone dose.
Overall survival
(See comparisons 1.1, 1.2, 1.3)
Data for this outcome were available from six trials (Chatani 1985; Chatani 1994; Harwood 1977; Kurtz 1981; Murray 1997; Priestman 1996).
Three trials (Chatani 1994; Harwood 1977; Priestman 1996) compared lower dose radiation with RBE < 39 Gy (2000 cGy in 5 fractions, 1000 cGy in a single fraction or 1200 cGy in 2 fractions) to standard dose WBRT with RBE = 39 Gy (3000 cGy in 10 fractions). Combining these data in a meta‐analysis using RevMan analyses in RevMan 5.1 showed a significant difference (P = 0.01) in overall survival favoring 3000 cGy in 10 fractions (HR 1.21, 95% CI 1.04 to 1.40). Of note, Chatani 1994 reported no statistically significant difference in median nor one‐year survival for those patients treated with 3000 cGy in 10 daily fractions (3.4 months and 6%, respectively) versus 2000 cGy in 5 daily fractions of WBRT (2.4 months and 4%, respectively). Harwood 1977 also reported no statistically significant difference in survival for patients treated with a single 1000 cGy of WBRT versus 3000 cGy in 10 daily fractions. Priestman 1996, however, reported a statistically significant difference (P = 0.04) in overall survival favoring patients treated with 3000 cGy in 10 daily fractions of WBRT (84 days median survival) as compared to patients treated with 1200 cGy in 2 daily fractions (77 days median survival).
Four trials (Chatani 1985; Chatani 1994; Kurtz 1981; Murray 1997) compared higher dose WBRT with RBE > 39 Gy (5000 cGy in 20 fractions or 5440 cGy in 34 fractions twice daily) to standard dose WBRT with RBE = 39 Gy (3000 cGy in 10 fractions). Pooling of the data from these four trials showed no statistically significant difference (P = 0.65) between higher dose WBRT and control in overall survival (HR 0.97, 95% CI 0.83 to 1.12).
Overall survival was not obtainable from the published reports of Borgelt 1980 or Borgelt 1981. However, the reports indicated that overall median survival was not statistically different among the different dose‐fractionation schemes of WBRT in these trials.
Although the Haie‐Meder 1993 trial was excluded based on the lack of a standard arm, this study also reported no difference in overall survival with the different dose‐fractionation schemes studied.
Two trials (Borgelt 1980b; Chatani 1994) provided data comparing two fractionation schedules commonly employed in Canada (control: 2000 cGy in five fractions or 3000 cGy in 10 fractions). Neither trial detected a significant difference in overall survival between the two control fractionation schemes.
Since the last update, two randomized controlled trials were published which examined patients treated with WBRT 4000 cGy in 20 fractions BID versus standard WBRT 2000 cGy in 4 or 5 daily fractions. Davey 2008 randomized patients to 4000 cGy in 20 fractions twice daily (BID) WBRT versus 2000 cGy in 5 daily fractions WBRT. Graham 2010 randomized patients to 4000 cGy in 20 fractions BID WBRT versus 2000 cGy in 4 daily fractions WBRT. Pooling the data from these two trials showed no statistically significant difference in overall survival (HR 1.18, 95% CI 0.89 to 1.56, P = 0.25).
Symptom control
Symptom control was assessed in seven published reports (Borgelt 1980; Borgelt 1981; Chatani 1985; Chatani 1994; Harwood 1977; Kurtz 1981; Priestman 1996). However, a variety of different scales were used (for example neurological symptom relief, palliative index, performance status) and statistical pooling of the data was therefore not meaningful. Considered individually, none of these trials showed a difference in symptom control with altered dose‐fractionation schedules as compared to control (3000 cGy in 10 fractions).
Overall brain control
A post hoc subset analysis in the Davey 2008 trial showed time to retreatment for intracranial relapse was 14 weeks in the control WBRT arm (2000 cGy in 5 daily fractions) as compared to 32 weeks in the 4000 cGy in 20 BID fractions arm (P = 0.03). The primary endpoint for the Graham 2010 trial was proportion of patients with overall brain progression. There was a statistically significant difference in brain progression favouring patients treated with 4000 cGy in 20 BID fractions as compared to 2000 cGy in 4 daily fractions (44% versus 64%, P = 0.03, respectively).
Neurological function
(See comparisons 02 01 and 02 02)
Neurological function outcomes were pooled from seven reports (Borgelt 1980; Borgelt 1981; Chatani 1985; Chatani 1994; Harwood 1977; Kurtz 1981; Priestman 1996). The grading scales utilized were similar across trials and were typically based on a four‐point scale ranging from one (minimal interference) to four (comatose or requiring constant nursing care).
For three reports (Borgelt 1980; Borgelt 1981; Kurtz 1981) neurological function improvement was reported only for participants with a baseline neurological function of grade two or three. Within this limitation, there was no statistically significant difference (P = 0.23) in the pooled rates of neurologic function improvement for those treated with a biologically higher dose versus the control dose (odds ratio (OR) 1.14, 95% CI 0.92 to 1.42). Overall, there was a statistically significant difference (P = 0.03) in neurological function improvement favoring the control WBRT dose of 3000 cGy in 10 daily fractions as compared to a lower dose (OR 1.74, 95% CI 1.06 to 2.84). The duration of any improvement was not consistently reported.
Regine 2001 reported on the neurocognitive outcome in brain metastases participants treated in the Murray 1997 trial of accelerated fractionation versus accelerated hyperfractionated WBRT. Based on Mini‐Mental Status Examination scores, there was no significant difference in neurocognitive function between the two treatment arms of the trial. However, at three months, those participants with uncontrolled brain metastases had a significant drop in Mini‐Mental Status Examination scores as compared to those participants with radiographically controlled brain metastases.
Adverse effects
Adverse effects were inconsistently reported among the trials in terms of incidence and grade. In Borgelt 1981, there was no difference in treatment morbidity (defined as worsening of neurological symptoms or the appearance of new symptoms) among the treatment arms. For the Chatani 1994 study, no patient experienced grade three or more acute toxicity.
In the Harwood 1977 study, 40% (out of 51 participants) treated with a single dose of WBRT developed acute complications (increased headache, nausea and vomiting, neurologic deficit; or a fall in the level of consciousness) compared with 27% (out of 50 participants) treated with the fractionated course. This difference did not reach statistical significance (P = 0.254).
Murray's study (Murray 1997) of accelerated hyperfractionation versus 3000 cGy in 10 fractions over 10 days resulted in one grade four ototoxicity and one grade five toxicity (patient death) due to cerebral edema in the accelerated hyperfractionation arm (N = 216 participants). There was no difference in the incidence of acute grade three toxicity nor in the incidence of grade three or grade four late toxicity.
In Priestman's study (Priestman 1996), 22 participants (out of 274 participants) treated with 1200 cGy in two fractions of WBRT developed drowsiness or lethargy, headache, nausea or vomiting, dizziness or ataxia, cerebral hemorrhage, blurred vision or fits as compared with 13 participants (out of 270 participants) treated with 3000 cGy in 10 fractions.
WBRT plus radiosensitizers versus WBRT
Since the last review, two additional trials (Scott 2007; Suh 2008) have been added. Results from the Scott 2007 trial were not pooled as this report was a post hoc subset analysis of breast cancer patients randomized to WBRT and efaproxiral versus WBRT alone. Furthermore, the larger confirmatory trial which included breast cancer patients only (a priori) was subsequently published in abstract form (Suh 2008). No difference in overall survival was reported in breast cancer patients treated with WBRT and efaproxiral versus WBRT alone. The results from Suh 2008 could not be pooled as they were published in abstract form only.
Six fully published RCTs (DeAngelis 1989; Eyre 1984; Komarnicky 1991; Mehta 2003; Phillips 1995; Suh 2006) examined the use of radiosensitizers in addition to WBRT. The radiosensitizers studied were:
lonidamide,
metronidazole,
misonidazole,
motexafin gadolinium,
bromodeoxyuridine (BrdU),
RSR13 (efaproxiral).
With regard to the outcomes of interest, none of the trials reported on:
neurologic function, or
proportion of participants who were able to reduce their daily dexamethasone dose and the duration of reduced dexamethasone requirements.
Overall survival
(See comparison 3.1)
Data on survival was provided by five of the six trials (Eyre 1984; Komarnicky 1991; Mehta 2003; Phillips 1995; Suh 2006). When these data were combined, no difference (P = 0.11) was found in overall survival (HR 1.08, 95% CI 0.98 to 1.18).
The DeAngelis 1989 trial examined the use of WBRT with or without lonidamine and found no improvement (P = 0.42) in median survival (165 days for the WBRT group versus 120 days for the lonidamine group). Survival at six months could not be obtained from this study. As such, survival data from this study (DeAngelis 1989) could not be pooled.
Local brain tumor response
(See comparison 3.2)
Four trials (DeAngelis 1989; Eyre 1984; Phillips 1995; Suh 2006) reported on local brain tumour response rates (either complete response (CR) or partial response (PR)). There was no significant difference (P = 0.46) in response rate between those participants receiving only WBRT and those receiving treatment with WBRT and radiosensitizers (OR 0.87, 95% CI 0.60 to 1.26).
Quality of life
Only one trial (Mehta 2003) reported quality of life outcomes. In this trial, 401 participants with lung cancer, breast cancer, melanoma, and other cancers were randomized to motexafin gadolinium and WBRT or WBRT alone. The trial reported no significant difference in time to progression of the brain‐specific quality of life (FACT‐BR) assessment in either of the treatment groups.
There was also no statistically significant difference between treatment arms in time to neurocognitive progression as reported by Meyers 2004 on the participants in the Mehta 2003 trial of WBRT with or without motexafin gadolinium. Participants with lung cancer (but not other types of cancer) who were treated with motexafin gadolinium in addition to WBRT tended to have improved memory and executive function (P = 0.062) and improved neurological function as assessed by a blinded events review committee (P = 0.048).
It was concluded that motexafin gadolinium did not confer an overall advantage in survival or time to neurologic progression for the entire cohort. Based on the subgroup analysis, there was a suggestion that participants with lung cancer may benefit (based on improved neurologic function, memory, and executive function). A randomized trial examining this specific subgroup has been launched.
Symptom control
For participants receiving WBRT with or without the radiosensitizer misonidazole (Komarnicky 1991), there was no difference between the treatment arms in the percentage of total survival time spent in an improved or stable Karnofsky Performance Status (KPS), or in the median time to deterioration of KPS. The percentage of participants who spent 90% to 100% of their survival time in an improved or stable neurological state was also similar among the treatment and control arms.
Suh 2006 reported on Spitzer quality of life and KPS outcomes for participants randomized to efaproxiral and WBRT versus WBRT alone. A higher proportion of participants in the efaproxiral arm had stable or improving quality of life scores (OR 1.21) and KPS (OR 1.38) (P = 0.008) as compared to participants treated with WBRT alone.
Adverse effects
All six studies that assessed the addition of radiosensitizers to WBRT reported serious adverse effects.
The study conducted by DeAngelis 1989 found that the most common side effects of lonidamide and WBRT were myalgia (68% or 21/31), testicular pain (42% or 8/19 of men), anorexia (26% or 8/31), ototoxicity (26% or 8/31), malaise or fatigue (26% or 8/31), and nausea and vomiting (19% or 6/31). No acute or subacute radiation‐related neurotoxicity was observed in either treatment group.
Participants who received WBRT and metronidazole in the Eyre 1984 study reported a 51% (out of 57 participants) incidence of nausea and vomiting compared with 3.2 % (out of 54 participants) in the WBRT alone arm.
In the study conducted by Komarnicky 1991, misonidazole administration with WBRT was well tolerated and produced no grade three neurotoxicity or ototoxicity. However, several (number not reported) grade three symptoms of nausea and vomiting (defined as occurring one to three times daily) were noted.
Phillips 1995 reported three fatal toxicities in 34 participants randomized to WBRT with administration of the radiosensitizer BrdU. One death resulted from a severe Stevens‐Johnson skin reaction (a rare and severe adverse reaction to sulphonamides involving skin and mucous membranes of the eyes, mouth, nose and genitals with ulceration and loss of epithelium) and two other deaths were due to neutropenia and infection. No increased rate of radiation skin reactions or central nervous system injuries were noted in the BrdU arm of this study.
The trial by Mehta 2003 reported grade three and four adverse events: hypotension (5.8%), asthenia (2.6%), hyponatremia (2.1%), leukopenia (2.1%), hyperglycemia (1.6%) and vomiting (1.6%) out of 193 participants randomized to the WBRT and motexafin gadolinium arm.
In the Suh 2006 trial, 28% of participants in the efaproxiral arm experienced grade 3 or 4 treatment‐emergent adverse events. The most common efaproxiral severe adverse effect was hypoxemia (11%, 29 out of 266 participants).
WBRT plus radiosurgery versus WBRT
Three trials provided information on the effectiveness of WBRT alone compared with WBRT plus radiosurgery (Andrews 2004; Chougule 2000; Kondziolka 1999). Two trials were published in full. The Chougule 2000 trial has been published in abstract form.
The following outcomes of interest were not reported in any of the trials:
brain metastases response (CR + PR),
quality of life,
symptom control.
Overall survival
(See comparison 4.1)
The results of the two fully published reports (Andrews 2004; Kondziolka 1999) were pooled. Overall, there was no difference (P = 0.24) in overall survival (HR 0.61, 95% CI 0.27 to 1.39) between those participants with multiple brain metastases treated with WBRT and radiosurgery boost as compared to WBRT alone. The Chougule 2000 trial (reported in abstract form) also did not find median survival differences (P value not stated) among the randomized arms (radiosurgery alone: seven months, radiosurgery and WBRT: five months, WBRT alone: nine months). The results of the Chougule 2000 trial could not be pooled as insufficient detail was available from the published abstract. The Chougule 2000 trial has never been fully published. The Andrews 2004 trial reported on improved survival (P = 0.0393) for the subset of participants with surgically unresectable single brain metastasis treated with WBRT and radiosurgery boost as compared to WBRT alone (median survival 6.5 months versus 4.9 months).
Local brain tumor control
(See comparison 4.2)
Local control was defined as unchanged or improved serial post‐treatment MRI scans judged as either a complete response, partial response or stable disease. Progressive disease was defined as an increase in size in any brain lesion, the development of new brain lesions, or stable disease with neurologic deterioration. Data for local brain control in the Andrews 2004 and Kondziolka 1999 trials were pooled. Overall, there was a statistically significant (P = 0.0003) improvement in local brain control favouring the WBRT and radiosurgery boost arm (HR 0.35, 95% CI 0.20 to 0.61). Data on one year local brain control were not obtainable from the Chougule 2000 abstract.
Intracranial progression‐free duration
In the Kondziolka 1999 trial, the median time to local brain failure was six months after WBRT alone (95% CI 3.5 to 8.5) in comparison to 36 months after WBRT and radiosurgery boost (95% CI 15.6 to 57). In the Andrews 2004 trial, there was no statistically significant difference (P = 0.1278) with respect to overall time to intracranial tumour progression or neurologic deaths rates between those treated with WBRT and radiosurgery boost as compared to WBRT alone. The Chougule 2000 abstract did not report on intracranial progression‐free duration. These results could not be pooled due to the differences in reporting this outcome.
Neurologic function
Neither Kondziolka 1999 nor Chougule 2000 reported on neurologic function outcomes. Andrews 2004 reported that the KPS was improved at six months in 10/79 or 13% of participants treated with WBRT and radiosurgery boost as compared to 3/75 or 4% of participants treated with WBRT alone (P = 0.0331). However, mental status as measured using the Mini‐Mental Status examination was not statistically different between the two arms.
Proportion of participants who are able to reduce their daily dexamethasone dose and duration
Neither Kondziolka 1999 nor Chougule 2000 reported on dexamethasone outcomes. Andrews 2004 reported that a higher proportion of participants (41/79) in the WBRT and radiosurgery arm had decreased steroid requirements at six months as compared to 25/75 participants in the WBRT alone arm (P = 0.0158).
Adverse effects
Kondziolka 1999 reported no neurologic or systemic morbidity related to stereotactic radiosurgery. After WBRT, participants expectedly developed mild scalp erythema and hair loss. In the Andrews 2004 trial, early and late toxicities did not differ greatly between the two treatment arms. However, there were more participants with acute grade three and four toxicity in the WBRT and radiosurgery boost arm (4/160 participants) as compared to the WBRT alone arm (0/166 participants). Also, there were more late grade three and four toxicity in the combined arm (6/160 participants) compared to the WBRT alone arm (3/166 participants).
Radiosurgery alone versus radiosurgery and WBRT
In this updated review, two fully published trials (Chang 2009; Kocher 2011) are now included on the area of radiosurgery alone versus radiosurgery and WBRT.
None of the trials (Aoyama 2006; Chang 2009; Kocher 2011) examining the use of radiosurgery alone versus radiosurgery and WBRT reported on brain metastases response rates (CR + PR) nor on the percentage of patients able to reduce the dexamethasone dose or duration of reduced dexamethasone dose.
Overall survival
(See comparison 5.1)
The pooled overall survival analysis for the two randomized controlled trials (Aoyama 2006; Chang 2009) showed no significant difference (HR 0.98, 95% CI 0.71 to 1.35, P = 0.88).
In the Kocher 2011 trial, overall survival for the radiosurgery alone arm versus WBRT and radiosurgery boost coud not be isolated. Nevertheless, Kocher 2011 reported no significant difference in overall survival among patients treated with combined therapy (either surgery and WBRT or radiosurgery and WBRT) as compared to patients treated with surgery alone or radiosurgery alone. Median survival was 10.9 months (in patients treated with combined therapy) versus 10.7 months (in those treated with either surgery or radiosurgery alone).
Local control
(See comparison 5.2)
From the three trials (Aoyama 2006; Chang 2009; Kocher 2011), there was a statistically significant difference (P < 0.0001) in pooled local brain control (HR 2.61, 95% CI 1.68 to 4.06) favouring WBRT and radiosurgery versus radiosurgery alone.
Distant brain control
(See comparison 5.3)
From the three trials (Aoyama 2006; Chang 2009; Kocher 2011), there was a statistically significant difference (P < 0.00001) in pooled distant brain control (HR 2.15, 95% CI 1.55 to 2.99) favoring WBRT and radiosurgery versus radiosurgery alone.
Neurologic function
In the Kocher et al trial (Kocher 2011), the duration of functional independence was the primary endpoint (defined as the date of randomization to the first report of WHO performance status decline of more than 2). The median time to WHO performance status > 2 (survival with functional independence) was 10 months in the surgery or radiosurgery alone arm versus 9.5 months in the surgery and WBRT or radiosurgery and WBRT arms (P = 0.71).
In the Aoyama et al trial (Aoyama 2006), systemic functional preservation rates (KPS ≥ 70) at 12 months was 33.9% in the WBRT and radiosurgery arm as compared to 26.9% in the radiosurgery alone arm (P = 0.53). Neurologic preservation rate at 12 months was 72.1% in the WBRT and radiosurgery arm, similar to 70.3% in the radiosurgery alone arm (P = 0.99).
The Chang et al trial (Chang 2009) did not describe neurologic function outcomes.
Neurocognition
Neurocognition was the primary outcome in the Chang et al trial (Chang 2009). A formal battery of neurocognitive tests was used. After 58 patients were entered into the Chang et al trial (Chang 2009) the trial was stopped early based on the early stopping rules that there was a high probability (96%) that patients randomized to radiosurgery and WBRT were significantly more likely to show a decline in learning and memory function (mean posterior probability of decline 52%) at four months compared to patients treated with radiosurgery alone (mean posterior probability of decline 24%).
The Aoyama et al trial (Aoyama 2006) used a crude measure of neurocognition, the Mini‐Mental Status examination. Aoyama et al (Aoyama 2006) reported that the average duration until Mini‐Mental Status examination deterioration was 16.5 months in the WBRT and radiosurgery arm versus 7.6 months in the radiosurgery alone arm (P = 0.05).
The Kocher et al trial (Kocher 2011) did not report neurocognitive outcomes.
Quality of life
The Aoyama et al trial (Aoyama 2006) did not report on quality of life outcomes.
The Kocher et al trial (Kocher 2011) indicated that quality of life was a secondary endpoint, but the results relating to quality of life have not yet been reported.
In the Chang et al trial (Chang 2009), the validated quality of life instrument (FACT‐BR) was used. The FACT‐BR mean difference between the randomized arms at four months compared to baseline was 2.8 (95% CI ‐26 to 21, P = 0.76). The authors indicated, however, that the wide confidence interval implied that the quality of life results were inconclusive.
Side effects
In the Aoyama et al trial (Aoyama 2006) symptomatic acute neurologic toxicity was observed in 4 patients (out of 65) receiving WBRT and radiosurgery and in 8 patients (out of 67) receiving radiosurgery alone (P = 0.36). Symptomatic late neurologic toxic effects were seen in 7 patients in the WBRT and radiosurgery group versus 3 patients in the radiosurgery alone arm (P = 0.20).
In the Kocher et al trial (Kocher 2011), there were 16 serious acute toxicities (13 patients in the surgery and WBRT or radiosurgery and WBRT arms and 3 patients in the surgery or radiosurgery alone arm). Symptomatic radionecrosis was reported in 8% (7/90 patients) after radiosurgery alone and in 13% (12/95 patients) treated with radiosurgery and WBRT.
In the Chang et al trial (Chang 2009) there was one case of grade 3 toxicity in the radiosurgery and WBRT arm and one case of grade 3 toxicity in the radiosurgery alone arm that was attributable to radiation. Two cases of grade 4 toxicity (pathologically proven radiation necrosis) were reported in the radiosurgery alone arm.
WBRT and systemic therapy
Three trials (Knisely 2008; Lee 2008; Neuhaus 2009) which examined the use of WBRT and systemic therapy have been added to the update of this review.
Seven studies provided information for a comparison of the effectiveness of WBRT plus systemic therapy and WBRT alone (Antonadou 2002; Guerrieri 2004; Knisely 2008; Mornex 2003; Neuhaus 2009; Postmus 2000; Ushio 1991). However, the results from the trials of systemic therapy and WBRT could not be pooled due to the heterogeneity of the study interventions. The results for each trial were therefore reported separately.
In the randomized controlled trial by Ushio 1991, 100 participants were randomized to one of three treatment arms and brain tumor regression (more than 50%) was reported as follows.
WBRT alone (36% or 5/14).
WBRT plus chloroethylnitrosoureas (methyl‐CCNU or ACNU) (69% or 11/16).
WBRT plus chloroethylnitrosoureas plus tegafur (74% or 14/19).
Response rates were significantly different (P < 0.05) between the WBRT alone 36% (5/14) and the WBRT + chloroethylnitrosoureas + tegafur 74% (14/19) arms. However, with regard to median survival times, no significant differences were found among the three groups:
WBRT alone, 27 weeks;
WBRT plus chloroethylnitrosoureas (methyl‐CCNU or ACNU), 29 weeks;
WBRT plus chloroethylnitrosoureas plus tegafur, 30.5 weeks.
Information regarding intracranial progression‐free duration, quality of life, symptom control, neurological function and ability to taper down on dexamethasone dose was not reported. Two participants died of probable adverse effects of chemotherapy.
Postmus 2000 conducted a RCT on the use of teniposide chemotherapy with or without WBRT in participants with small cell lung cancer metastatic to brain. A 57% response rate was seen in the combined‐modality arm and a 22% response rate was seen in the teniposide alone arm (P < 0.001). Time to brain progression was longer in the combined arm (P = 0.005). Response outside the brain was no different. Overall survival was not significantly different: median survival 3.5 months for the combined arm versus 3.2 months for the teniposide only arm (P = 0.87). Forty‐five teniposide only participants and 43 combined‐modality participants had both a neurologic function baseline score and a score after cycle two. Of the participants in the teniposide only arm, eight improved and 34 remained stable compared with 12 and 23 respectively in the combined‐modality arm. Quality of life outcomes were not reported. The proportion of participants able to reduce the dexamethasone dose was also not reported. Toxicities were noted to be mild. The most common toxicity was hematological, resulting in dose delays in 45 participants and dose reductions in eight participants. Non‐hematologic toxicities occurred infrequently, for example, WHO grade three or four nausea and vomiting occurred in 11% out of 60 participants treated with combined modality and 5% out of 60 participants treated in the teniposide only group. WHO grade three or four infection occurred in 4% out of 60 participants treated with combined modality and 6% out of 60 participants treated in the teniposide only group. Less than 3% out of 60 participants in each arm experienced mucositis, headache or cutaneous reactions not exceeding WHO grade three toxicity.
Robinet 2001 examined early versus delayed WBRT with concurrent cisplatin and vinorelbine chemotherapy in participants with metastatic non‐small cell lung cancer. WBRT was either given delayed (after two to six cycles of chemotherapy for intracranial non‐responders) or early (on day one to 12 during the first cycle of chemotherapy). The study found no difference in survival times between the two arms of the study (median survival duration 24 weeks in the delayed radiotherapy arm and 21 weeks in the early radiotherapy arm). Intracranial progression‐free duration, quality of life, symptom control, neurologic function and ability to reduce dexamethasone dose were not reported. A high number of patient deaths were reported:
as a result of toxicity, 13 participants (six in the delayed radiotherapy arm (6.9%) and seven in the early radiotherapy arm (8.2%);
as a result of sepsis during severe neutropenia, 10 participants;
from pneumonia (without neutropenia), one participant in each study arm following the second cycle of chemotherapy;
from renal failure, one participant in the delayed chemotherapy arm after the first cycle.
Antonadou 2002 conducted a randomized trial where 134 participants received either WBRT or WBRT with temozolomide chemotherapy. The results were published in abstract form only. The median survival time was reported as not significantly different between the two treatment arms (8.3 months WBRT plus temozolomide versus 6.3 months for WBRT alone, P = 0.179). Improved brain response was seen in the combined arm 53.4% (95% CI 40% to 66.7%) compared to the WBRT alone arm 33.3% (95% CI 21.4% to 47.1%, P = 0.039). Intracranial progression‐free duration, toxicities, quality of life, symptom control, neurological function, and ability to reduce dexamethasone dose were not reported in the abstract.
Mornex 2003 reported on the use of fotemustine combined with WBRT (37 participants) versus fotemustine alone (39 participants) for participants with metastatic melanoma to brain. There was no significant difference in overall survival (median survival was 86 days in the fotemustine alone arm versus 105 days in the combined arm) or in cerebral response (7.4%, 95% CI 1 to 24 in the fotemustine alone arm versus 10%, 95% CI 2 to 27% in the combined treatment arm). However, there was a significant difference in favour of the combined arm for time to cerebral progression (P = 0.028). Quality of life, symptom control, neurological function and ability to reduce dexamethasone dose were not reported. Myelosuppression was the most severe adverse event. Delayed grade three to four neutropenia occurred in 46% of 39 participants in the fotemustine alone arm and 35% of 37 participants in the combined arm. Delayed grade three to four thrombocytopenia occurred in 44% of 39 participants in the fotemustine alone arm and 38% of 37 participants in the combined arm. Severe anemia occurred in 5% of 39 participants in the fotemustine alone arm and 11% of 37 participants in the combined arm. One participant suffered a cerebral hemorrhage and three participants died of pneumonia.
Guerrieri 2004 randomized brain metastases participants from non‐small cell lung cancer to either whole brain radiotherapy alone versus whole brain radiotherapy and carboplatin chemotherapy. The planned accrual target was 300 participants. However, the study was closed early due to poor accrual and the results of the 42 participants entered into the study were reported. As such, no firm conclusions could be made. From this small sample, the median survival was 4.4 months in the WBRT arm versus 3.7 months in the combined arm (P = 0.64). The objective brain response rates were 10% in the whole brain radiotherapy arm and 29% in the combined arm (P = 0.24).
Knisely 2008 compared WBRT versus WBRT and thalidomide. Median survival was 3.9 months for both arms. No novel toxicities were seen but thalidomide was not tolerated. Forty‐eight per cent of patients discontinued thalidomide because of side effects. The time to brain progression curves did not differ significantly (P = 0.097) between the two treatment arms.
Neuhaus 2009 randomized brain metastases patients from lung cancer (small cell lung cancer or non‐small cell lung cancer) to either WBRT and topotecan chemotherapy versus WBRT alone. The trial was stopped early because of poor accrual (total of 96 patients compared to a planned 320 patients). No significant differences in overall survival (HR 1.32, 95% CI 0.83 to 2.10, P = 0.43) or progression‐free survival (HR 1.28, 95% CI 0.73 to 2.43, P = 0.89) were seen. Hematological events occurred mainly in the combined treatment arm.
Lee 2008 reported a trial in patients with metastatic non‐small cell lung cancer to the brain treated with primary chemotherapy (gemcitabine and vinorelbine) followed by WBRT versus WBRT first then followed by the same chemotherapy. The response rates of chemotherapy and survival outcomes were not statistically different between the two arms. The overall response rate was 28% versus 39.1%, progression‐free survival was 3.6 months versus 4.4 months, and overall survival was 9.1 months versus 9.9 months respectively. In the WBRT first arm, grade three or four neutorpenia was more frequent (79% versus 40%) during chemotherapy.
Steroids alone versus steroids and WBRT
One randomized trial (Horton 1971) examined the use of prednisone with or without WBRT. This was a small trial (48 participants were recruited) conducted at a time when computed tomography (CT) scanning was not available. Participants were enrolled in the study if they had a histologically proven cancer and clinical symptoms and signs of brain metastases, such as abnormalities identified by radioisotope brain scans, electro‐encephalograms (EEGs), angiograms, and spinal fluid chemistry and cytology. The proportion of participants with improved performance status was similar in both the prednisone alone and the WBRT plus prednisone arms (63% out of 19 participants and 61% out of 28 participants respectively). The median survival time in the prednisone alone arm was ten weeks as compared to 14 weeks in the combined treatment arm (P value not stated). Participants were not stratified for other known prognostic factors such as age, performance status, and extent of extracranial disease. Other outcomes such as intracranial progression‐free duration, quality of life, symptom control, neurologic function, toxicities and ability to taper steroid dose were not reported.
Discussion
In this updated review, the addition of nine fully published articles (Chang 2009; Davey 2008; Graham 2010; Knisely 2008; Kocher 2011; Lee 2008; Neuhaus 2009; Scott 2007; Suh 2008) did not change the conclusions of the original Cochrane review. This updated review now includes the pooled results for radiosurgery alone versus radiosurgery and WBRT trials based on a total of three randomized controlled trials (Aoyama 2006; Chang 2009; Kocher 2011).
Altered WBRT dose‐fractionation schedules versus conventional WBRT fractionation schedule
All these trials had well balanced arms with respect to variables known to affect survival (such as age, performance status and status of extracranial disease activity).
In summary, none of the randomized controlled trials have found a benefit (in terms of overall survival or neurologic function) with altered dose‐fractionation schedules as compared to standard (3000 cGy in 10 fractions or 2000 cGy in 4 or 5 daily fractions). Two studies (Borgelt 1980b; Chatani 1994) provided data comparing two fractionation schedules commonly employed in Canada (2000 cGy in 5 fractions or 3000 cGy in 10 fractions). Neither trial detected a significant difference in overall survival nor neurologic function between these two fractionation schemes. Symptom control was assessed in seven reports or nine trials (Borgelt 1980a; Borgelt 1980b; Borgelt 1981a; Borgelt 1981b; Chatani 1985; Chatani 1994; Harwood 1977; Kurtz 1981; Priestman 1996). All trials showed no difference in symptom control with altered WBRT dose‐fractionation schedules as compared to control (3000 cGy in 10 fractions).
Two trials (Davey 2008; Graham 2010) reported on 4000 cGy in 20 BID fractions WBRT versus 2000 cGy in 4 or 5 daily fractions WBRT. No statistically significant difference in overall survival was seen between these two arms. Graham 2010 reported a statistically significant difference (P = 0.03) in brain progression favoring patients treated with 4000 cGy in 20 BID fractions WBRT (44% versus 64% respectively). A post hoc subset analysis in the Davey 2008 trial showed that the time to retreatment for intracranial relapse was 14 weeks in the control WBRT arm (2000 cGy in 5 daily fractions) as compared to 32 weeks in the 4000 cGy in 20 BID fractions arm (P = 0.03).
The major deficiencies in these trials are lack of comprehensive quality of life outcomes and lack of information regarding the proportion of participants able to reduce the steroid dose in each arm. With no overall improvement in overall survival and neurologic function classification, palliative endpoints such as quality of life and steroid use are important. As such, a lack of sufficient high quality evidence precludes recommendations as to which dose‐fractionation regimen provides the greatest improvement in quality of life and steroid dose. Furthermore, the trials did not address whether a particular dose‐fractionation regimen is superior in terms of intracranial progression‐free duration.
The results of these studies examining the use of altered dose‐fractionation schemes of WBRT may not be generalizable to all tumor types. The majority of participants in these studies had lung, breast or colorectal cancer primaries. Participants with diagnosed small cell lung cancer, germ cell tumors and hematological malignancies were often excluded from the trials.
WBRT plus radiosensitizers versus WBRT
In an attempt to improve local brain control, radiosensitizers have been added to WBRT. However, none of the randomized controlled trials showed a benefit in terms of overall survival or brain response (CR + PR). The use of radiosensitizers in these trials (lonidamide, metronidazole, misonidazole, BrdU, motexafin gadolinium, efaproxiral) was associated with toxicity.
WBRT plus radiosurgery versus WBRT
The two fully published RCTs (Andrews 2004; Kondziolka 1999) examining the use of radiosurgery boost and WBRT versus WBRT alone revealed no significant difference in overall survival between the two arms. Although these trials were well balanced for baseline prognostic factors, the Kondziolka 1999 trial was methodologically weaker (small trial size and early trial closure) as compared to the Andrews 2004 trial. The Andrews 2004 trial included participants with one to three brain metastases. Although overall there was no improvement in survival with the addition of radiosurgery boost in the Andrews 2004 trial, survival was improved in participants with surgically unresectable single brain metastasis. The trial was designed with an a priori sample size calculation to test the hypothesis of improved survival in participants with single brain metastasis. The median survival time was 6.5 months for those with single brain metastasis treated with WBRT and radiosurgery boost versus 4.9 months for those treated with WBRT alone (P = 0.0393). The Kondziolka 1999 trial excluded participants with single brain metastasis (two to four brain metastases were included).
There was a statistically significant improvement in local brain control for both the Andrews 2004 and Kondziolka 1999 trials favouring the radiosurgery boost arm. The Chougule 2000 trial is difficult to analyse since the data remains in abstract form. Despite the lapse of many years, the full report from Chougule 2000 remains to be published. The Chougule 2000 abstract reported local brain control of 87%, 91% and 62% for gamma knife radiosurgery, gamma knife radiosurgery + WBRT and WBRT alone respectively (P values not given). These three reports support the conclusion that WBRT and radiosurgery boost improves local brain control (CR + PR + SD).
Thus, the addition of radiosurgery boost to WBRT does not improve survival for participants with two to four brain metastases but, in one trial, survival was increased in selected participants with surgically unresectable single brain metastasis treated with WBRT and radiosurgery boost as compared to WBRT alone. However, these trials were not designed to answer the question as to whether radiosurgery boost with WBRT is superior or equivalent to surgical resection and WBRT for participants with single resectable brain metastases. WBRT and radiosurgery boost also improves local brain control in selected participants with one to four brain metastases.
Radiosurgery alone versus WBRT and radiosurgery
Pooled results from the three trials (Aoyama 2006; Chang 2009; Kocher 2011) show no statistically significant difference in overall survival between the two arms. However, the addition of WBRT to radiosurgery significantly improves both local brain metastases control and distant brain control. The Chang 2009 trial evaluated neurocognition as the primary endpoint. Patients assigned to WBRT and radiosurgery boost were significantly more likely to show a decline in learning and memory function compared to those assigned to radiosurgery alone.
For selected patients with multiple brain metastases and eligible for radiosurgery, it remains controversial whether these patients should be treated with WBRT and radiosurgery boost to maximize local brain metastases and distant brain metastases control or whether they should be treated wtih radiosurgery alone (which may be associated with better neurocognitive outcomes). Overall survival between these two treatment options was not statistically different provided patients were followed closely and treated with salvage brain therapies at the time of relapse. However, it is possible that with a strategy of radiosurgery alone the increased risk of brain relapse may be associated with symptoms which are not reversible (Regine 2002).
The optimal timing of radiosurgery in selected patients has not been elucidated. The question as to whether radiosurgery should be used as boost treatment with WBRT at the time of relapse after WBRT failure or used alone, reserving WBRT for future extensive brain relapse, remains unanswered.
Improved reporting of quality of life, neurologic and neurocognitive outcomes is needed.
WBRT and systemic therapy
All trials (Antonadou 2002; Guerrieri 2004; Knisely 2008; Mornex 2003; Neuhaus 2009; Postmus 2000; Ushio 1991) examining the use of WBRTand chemotherapy versus WBRT alone showed no survival benefit with the addition of chemotherapy to WBRT. These trials also showed an increase in toxicity when chemotherapy was added to WBRT.
The two trials (Lee 2008; Robinet 2001) which examined the timing to WBRT, either concurrent or sequential to chemotherapy, also showed no statistically significant difference in overall survial wtih respect to WBRT timing with the use of chemotherapy.
Steroids versus steroids and WBRT
One small randomized trial (Horton 1971) examined the use of prednisone with or without WBRT. The proportion of participants with improvement in performance status was similar in the steroid alone and combined WBRT and steroid arms. In addition, the median survival of the steroid alone arm was 10 weeks as compared to 14 weeks in the combined arm (P value not stated). It is unknown whether both groups were balanced with respect to other prognostic factors such as age, performance status, extent of extracranial disease. Thus, the magnitude of benefit with the use of WBRT over steroids alone remains unknown.
Supportive care has been defined as "multi‐professional attention to the individual's overall physical, psychosocial, spiritual and cultural needs and should be available at all stages of the illness for participants of all ages and regardless of the current intention of any anti‐cancer treatment" (Ahmed 2003). The use of supportive care alone without WBRT is an option, for example, in participants with poor performance status and extensive active extracranial disease based on short life expectancy. These participants would fit into the recursive partitioning analysis (RPA) class III, where median survival is only 2.3 months with WBRT (Gaspar 1997). In comparison, participants in RPA classes I and II have median survivals of 7.1 months and 4.2 months respectively. There are, however, no RCTs which have been reported to help guide practitioners as to which subsets of participants with brain metastases benefit (in terms of survival, symptom control or quality of life) from WBRT and which subsets do not.
Authors' conclusions
This updated review did not change the original conclusions or implications for practice. The option of radiosurgery alone (without WBRT) in selected patients with newly diagnosed brain metastases has been added.
Altered WBRT dose‐fractionation schedules
There is no benefit (in terms of overall survival or neurologic function classification) with altered WBRT dose‐fractionation schedules as compared to standard fractionation (3000 cGy in 10 daily fractions or 2000 cGy in 4 or 5 daily fractions).
WBRT plus radiosensitizers versus WBRT
Of the radiosensitizers reported in randomized trials, none have shown a benefit as compared to WBRT alone. The use of radiosensitizers is not recommended outside research studies.
WBRT plus radiosurgery versus WBRT
Radiosurgery (boost with WBRT) may be considered in selected participants to improve local brain control. The radiosurgery trials included participants with up to four small brain metastases (less than 4 cm) and with a good Karnofsky performance status of 70 or more. However, there was no improvement in overall survival for participants with multiple brain metastases treated with radiosurgery boost and WBRT as compared to WBRT alone.
Radiosurgery alone versus radiosurgery and WBRT
The omission of WBRT in selected participants with multiple brain metastases who are treated with radiosurgery alone is associated with increased risk of local and distant brain relapse, but there is no detriment to overall survival. Based on one trial, the use of radiosurgery alone was associated with a better neurocognitive outcome as compared to WBRT and radiosurgery.
WBRT and systemic therapy
The use of chemotherapy as primary therapy for brain metastases or the use of chemotherapy with WBRT to treat brain metastases remains experimental.
Supportive care versus WBRT
There is a lack of high quality randomized evidence to clarify the value of WBRT versus supportive care alone in participants with multiple brain metastases. Supportive care alone without WBRT is an option (for example, for participants with poor performance status or widely disseminated cancer based on short life expectancy). However, there is a lack of contemporary high quality trials (only one older randomized trial has been published) to guide practitioners as to which subsets of participants with brain metastases should be managed with supportive care alone, without WBRT.
Future trials should be directed at examining the use of WBRT versus supportive care alone in participants with multiple brain metastases. Outcomes of interest include quality of life, symptom control, neurological function, steroid toxicity and overall survival. Novel radiosensitizers or chemotherapy agents which improve outcomes as compared to WBRT alone need exploration. The optimal timing of radiosurgery in relation to WBRT in selected participants with brain metastases remains to be defined.
Acknowledgements
The authors thank Dr A Ohlsson for his guidance in developing and reviewing this document. The Cancer Care Ontario Program in Evidence‐based Care's Supportive Care Guidelines Group and Neuro‐oncology Disease Site Group also contributed to the development of this document. Please see Cancer Care Ontario Practice Guidelines Initiative (CCOPGI) website (http://www.cancercare.on.ca/access_PEBC.htm) for a complete list of current Supportive Care Guidelines Group and Neuro‐oncology Disease Site Group members.
Appendices
Appendix 1. MEDLINE search strategy
1 exp Brain Neoplasms/ 2 ((brain or brainstem or intracranial or posterior fossa) adj3 (cancer* or carcinom* or tumor* or tumour* or neoplasm*)).mp. 3 1 or 2 4 exp Neoplasm Metastasis/ or metastas*.mp. 5 Radiotherapy/ 6 Radiotherapy, Adjuvant/ 7 (radiotherapy or radiat* or radiosurg*).mp. 8 Combined Modality Therapy/ 9 Radiosurgery/ 10 gamma knife.mp. 11 or/5‐10 12 3 and 4 and 11 13 randomized controlled trial.pt. 14 controlled clinical trial.pt. 15 randomized.ab. 16 placebo.ab. 17 radiotherapy.fs. 18 randomly.ab. 19 trial.ab. 20 groups.ab. 21 or/13‐20 22 12 and 21
key: mp=title, original title, abstract, name of substance word, subject heading word, unique identifier, pt=publication type, ab=abstract, fs=floating subheading, sh=subject heading
Appendix 2. EMBASE search strategy
1 exp Central Nervous System Tumor/ 2 exp brain cortex/ 3 ((brain or brainstem or intracranial or posterior fossa) adj3 (neoplasm* or cancer* or carcinoma* or tumor* or tumour*)).mp. 4 or/1‐3 5 Brain Metastasis/ or metastas*.mp. 6 4 and 5 7 exp radiosurgery/ 8 multimodality cancer therapy/ 9 Radiotherapy/ 10 Cancer radiotherapy/ 11 (radiotherap* or radiosurg*).mp. 12 gamma knife.mp. 13 radiat*.mp. 14 or/7‐13 15 6 and 14 16 random*.ti,ab. 17 factorial*.ti,ab. 18 (crossover* or cross over* or cross‐over*).ti,ab. 19 placebo*.ti,ab. 20 (doubl* adj blind*).ti,ab. 21 (singl* adj blind*).ti,ab. 22 assign*.ti,ab. 23 allocat*.ti,ab. 24 volunteer*.ti,ab. 25 crossover procedure/ 26 double blind procedure/ 27 randomized controlled trial/ 28 single blind procedure/ 29 or/16‐28 30 15 and 29
key:, mp=title, abstract, subject headings, heading word, drug trade name, original title, device manufacturer, drug manufacturer name, ti=title, ab=abstract
Appendix 3. CENTRAL search strategy
#1 MeSH descriptor Brain Neoplasms explode all trees #2 brain* near/3 (cancer* or carcinoma* or tumor* or tumour* or neoplasm*) #3 brainstem near/3 (cancer* or carcinoma* or tumor* or tumour* or neoplasm*) #4 intracranial near/3 (cancer* or carcinoma* or tumor* or tumour* or neoplasm*) #5 posterior fossa near/3 (cancer* or carcinoma* tumor* or tumour* or neoplasm*) #6 (#1 OR #2 OR #3 OR #4 OR #5) #7 MeSH descriptor Neoplasm Metastasis explode all trees #8 metastas* #9 (#7 OR #8) #10 MeSH descriptor Radiotherapy explode all trees #11 MeSH descriptor Radiotherapy, Adjuvant explode all trees #12 radiotherapy or radiat* or radiosurg* #13 MeSH descriptor Combined Modality Therapy explode all trees #14 MeSH descriptor Radiosurgery explode all trees #15 gamma knife #16 (#7 OR #8 OR #9 OR #10 OR #11 OR #12 OR #13 OR #14 OR #15) #17 (#6 AND #9 AND #16)
Data and analyses
Comparison 1.
Altered WBRT fractionation schedules versus WBRT control
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Overall survival: lower dose WBRT versus control WBRT (3000 cGy/10 daily fractions) | 3 | Hazard Ratio (Fixed, 95% CI) | 1.21 [1.04, 1.40] | |
| 2 Overall survival: higher dose WBRT versus control (3000 cGy/10 daily fractions) | 4 | Hazard Ratio (Fixed, 95% CI) | 0.97 [0.83, 1.12] | |
| 3 Overall survival: WBRT 4000 cGy/20 fractions BID versus control WBRT (2000 cGy/ 4‐5 daily fractions) | 2 | Hazard Ratio (Fixed, 95% CI) | 1.18 [0.89, 1.56] |
Analysis 1.1.
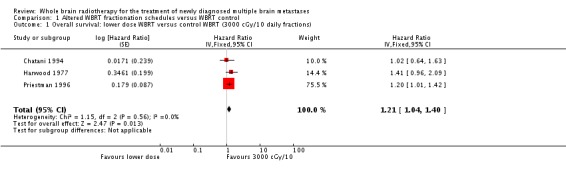
Comparison 1 Altered WBRT fractionation schedules versus WBRT control, Outcome 1 Overall survival: lower dose WBRT versus control WBRT (3000 cGy/10 daily fractions).
Analysis 1.2.

Comparison 1 Altered WBRT fractionation schedules versus WBRT control, Outcome 2 Overall survival: higher dose WBRT versus control (3000 cGy/10 daily fractions).
Analysis 1.3.

Comparison 1 Altered WBRT fractionation schedules versus WBRT control, Outcome 3 Overall survival: WBRT 4000 cGy/20 fractions BID versus control WBRT (2000 cGy/ 4‐5 daily fractions).
Comparison 2.
Altered WBRT fractionation schedules versus WBRT control: neurological function improvement
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Neurologic function improvement: lower dose WBRT versus control dose WBRT (3000 cGy/10 fractions) | 5 | 1612 | Odds Ratio (M‐H, Random, 95% CI) | 1.74 [1.06, 2.84] |
| 2 Neurologic function improvement: higher dose WBRT versus control dose WBRT (3000 cGy/10 fractions) | 4 | 1480 | Odds Ratio (M‐H, Random, 95% CI) | 1.14 [0.92, 1.42] |
Analysis 2.1.

Comparison 2 Altered WBRT fractionation schedules versus WBRT control: neurological function improvement, Outcome 1 Neurologic function improvement: lower dose WBRT versus control dose WBRT (3000 cGy/10 fractions).
Analysis 2.2.

Comparison 2 Altered WBRT fractionation schedules versus WBRT control: neurological function improvement, Outcome 2 Neurologic function improvement: higher dose WBRT versus control dose WBRT (3000 cGy/10 fractions).
Comparison 3.
WBRT with radiosensitizers (radiosen) versus WBRT alone
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Overall survival | 5 | Hazard Ratio (Fixed, 95% CI) | 1.08 [0.98, 1.18] | |
| 2 Brain tumor response rates: complete response (CR) and partial response (PR) combined | 4 | 754 | Odds Ratio (M‐H, Random, 95% CI) | 0.87 [0.60, 1.26] |
Analysis 3.1.

Comparison 3 WBRT with radiosensitizers (radiosen) versus WBRT alone, Outcome 1 Overall survival.
Analysis 3.2.

Comparison 3 WBRT with radiosensitizers (radiosen) versus WBRT alone, Outcome 2 Brain tumor response rates: complete response (CR) and partial response (PR) combined.
Comparison 4.
WBRT and radiosurgery versus WBRT
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Overall survival | 2 | Hazard Ratio (Fixed, 95% CI) | 0.61 [0.27, 1.39] | |
| 2 Local control | 2 | Hazard Ratio (Fixed, 95% CI) | 0.35 [0.20, 0.61] |
Analysis 4.1.

Comparison 4 WBRT and radiosurgery versus WBRT, Outcome 1 Overall survival.
Analysis 4.2.

Comparison 4 WBRT and radiosurgery versus WBRT, Outcome 2 Local control.
Comparison 5.
Radiosurgery alone versus WBRT and radiosurgery
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Overall survival | 2 | Hazard Ratio (Fixed, 95% CI) | 0.98 [0.71, 1.35] | |
| 2 Local Control | 3 | Hazard Ratio (Fixed, 95% CI) | 2.61 [1.68, 4.06] | |
| 3 Distant brain control | 3 | Hazard Ratio (Fixed, 95% CI) | 2.15 [1.55, 2.99] |
Analysis 5.1.
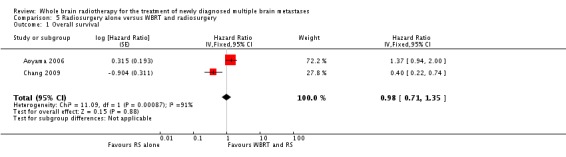
Comparison 5 Radiosurgery alone versus WBRT and radiosurgery, Outcome 1 Overall survival.
Analysis 5.2.

Comparison 5 Radiosurgery alone versus WBRT and radiosurgery, Outcome 2 Local Control.
Analysis 5.3.

Comparison 5 Radiosurgery alone versus WBRT and radiosurgery, Outcome 3 Distant brain control.
Comparison 6.
Sensitivity analysis
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Quality of studies: Overall survival (lower dose versus control 3000 cGy/10 daily fractions)) | 3 | Hazard Ratio (Fixed, 95% CI) | 1.21 [1.04, 1.40] | |
| 1.1 QS =1 | 1 | Hazard Ratio (Fixed, 95% CI) | 1.02 [0.64, 1.63] | |
| 1.2 QS = 2 | 1 | Hazard Ratio (Fixed, 95% CI) | 1.41 [0.96, 2.09] | |
| 1.3 QS = 3 | 1 | Hazard Ratio (Fixed, 95% CI) | 1.20 [1.01, 1.42] | |
| 2 Quality of studies: Overall survival (higher dose versus control 3000 cGy/10 daily fractions) | 4 | Hazard Ratio (Fixed, 95% CI) | 0.97 [0.83, 1.12] | |
| 2.1 QS =1 | 2 | Hazard Ratio (Fixed, 95% CI) | 0.79 [0.56, 1.11] | |
| 2.2 QS =2 | 1 | Hazard Ratio (Fixed, 95% CI) | 1.07 [0.87, 1.33] | |
| 2.3 QS =3 | 1 | Hazard Ratio (Fixed, 95% CI) | 0.93 [0.71, 1.21] | |
| 3 Quality of studies: Overall survival (4000 cGy/20 fractions BID versus 2000 cGy in 4‐5 daily fractions) | 2 | Hazard Ratio (Fixed, 95% CI) | 1.18 [0.89, 1.56] | |
| 3.1 QS = 3 | 2 | Hazard Ratio (Fixed, 95% CI) | 1.18 [0.89, 1.56] | |
| 4 Quality of studies: Neurologic function improvement (lower dose versus control) | 5 | 1612 | Odds Ratio (M‐H, Random, 95% CI) | 1.74 [1.06, 2.84] |
| 4.1 QS =1 | 1 | 116 | Odds Ratio (M‐H, Random, 95% CI) | 2.04 [0.75, 5.53] |
| 4.2 QS =2 | 3 | 963 | Odds Ratio (M‐H, Random, 95% CI) | 1.87 [0.94, 3.72] |
| 4.3 QS =3 | 1 | 533 | Odds Ratio (M‐H, Random, 95% CI) | 1.30 [0.92, 1.83] |
| 5 Quality of studies: Neurologic function improvement (higher dose versus control) | 4 | 1480 | Odds Ratio (M‐H, Random, 95% CI) | 1.14 [0.92, 1.42] |
| 5.1 QS =1 | 2 | 196 | Odds Ratio (M‐H, Random, 95% CI) | 0.96 [0.51, 1.82] |
| 5.2 QS =2 | 1 | 1100 | Odds Ratio (M‐H, Random, 95% CI) | 1.14 [0.88, 1.47] |
| 5.3 QS =3 | 1 | 184 | Odds Ratio (M‐H, Random, 95% CI) | 1.35 [0.75, 2.41] |
| 6 Quality of studies: Overall survival (WBRT with or without radiosensitizers) | 5 | Hazard Ratio (Fixed, 95% CI) | 1.08 [0.98, 1.18] | |
| 6.1 QS =2 | 2 | Hazard Ratio (Fixed, 95% CI) | 1.06 [0.91, 1.24] | |
| 6.2 QS =3 | 3 | Hazard Ratio (Fixed, 95% CI) | 1.08 [0.97, 1.21] | |
| 7 Quality of studies: Brain tumor response (WBRT with or without radiosensitizers) | 4 | 754 | Odds Ratio (M‐H, Random, 95% CI) | 0.87 [0.60, 1.26] |
| 7.1 QS = 1 | 1 | 58 | Odds Ratio (M‐H, Random, 95% CI) | 2.36 [0.75, 7.37] |
| 7.2 QS = 2 | 1 | 111 | Odds Ratio (M‐H, Random, 95% CI) | 0.89 [0.42, 1.88] |
| 7.3 QS = 3 | 2 | 585 | Odds Ratio (M‐H, Random, 95% CI) | 0.76 [0.55, 1.06] |
| 8 Quality of studies: Overall survival (WBRT and radiosurgery versus WBRT) | 2 | Hazard Ratio (Fixed, 95% CI) | 0.61 [0.27, 1.39] | |
| 8.1 QS=3 | 2 | Hazard Ratio (Fixed, 95% CI) | 0.61 [0.27, 1.39] | |
| 9 Quality of studies: Local control (WBRT and radiosurgery versus WBRT) | 2 | Hazard Ratio (Fixed, 95% CI) | 0.35 [0.20, 0.61] | |
| 9.1 QS=3 | 2 | Hazard Ratio (Fixed, 95% CI) | 0.35 [0.20, 0.61] | |
| 10 Quality of studies: Overall survival (radiosurgery alone versus WBRT and radiosurgery) | 2 | Hazard Ratio (Fixed, 95% CI) | 0.98 [0.71, 1.35] | |
| 10.1 Quality score =3 | 2 | Hazard Ratio (Fixed, 95% CI) | 0.98 [0.71, 1.35] | |
| 11 Quality of studies: Local control (radiosurgery alone versus WBRT and radiosurgery) | 3 | Hazard Ratio (Fixed, 95% CI) | 2.61 [1.68, 4.06] | |
| 11.1 QS =3 | 3 | Hazard Ratio (Fixed, 95% CI) | 2.61 [1.68, 4.06] | |
| 12 Quality of studies: Distant brain control (radiosurgery alone versus WBRT and radiosurgery) | 3 | Hazard Ratio (Fixed, 95% CI) | 2.15 [1.55, 2.99] | |
| 12.1 QS=3 | 3 | Hazard Ratio (Fixed, 95% CI) | 2.15 [1.55, 2.99] |
Analysis 6.1.
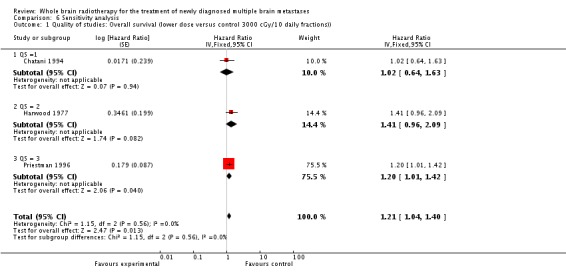
Comparison 6 Sensitivity analysis, Outcome 1 Quality of studies: Overall survival (lower dose versus control 3000 cGy/10 daily fractions)).
Analysis 6.2.

Comparison 6 Sensitivity analysis, Outcome 2 Quality of studies: Overall survival (higher dose versus control 3000 cGy/10 daily fractions).
Analysis 6.3.
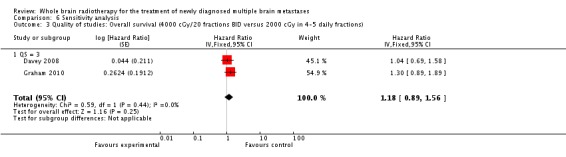
Comparison 6 Sensitivity analysis, Outcome 3 Quality of studies: Overall survival (4000 cGy/20 fractions BID versus 2000 cGy in 4‐5 daily fractions).
Analysis 6.4.

Comparison 6 Sensitivity analysis, Outcome 4 Quality of studies: Neurologic function improvement (lower dose versus control).
Analysis 6.5.

Comparison 6 Sensitivity analysis, Outcome 5 Quality of studies: Neurologic function improvement (higher dose versus control).
Analysis 6.6.
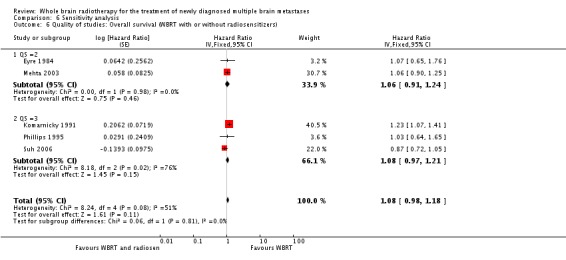
Comparison 6 Sensitivity analysis, Outcome 6 Quality of studies: Overall survival (WBRT with or without radiosensitizers).
Analysis 6.7.
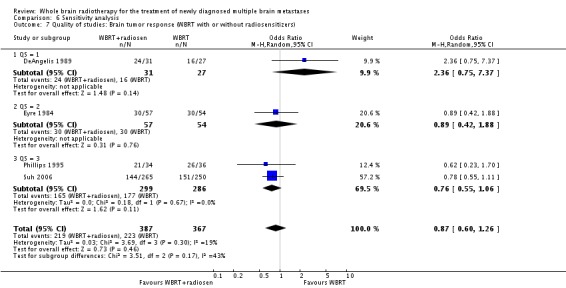
Comparison 6 Sensitivity analysis, Outcome 7 Quality of studies: Brain tumor response (WBRT with or without radiosensitizers).
Analysis 6.8.

Comparison 6 Sensitivity analysis, Outcome 8 Quality of studies: Overall survival (WBRT and radiosurgery versus WBRT).
Analysis 6.9.

Comparison 6 Sensitivity analysis, Outcome 9 Quality of studies: Local control (WBRT and radiosurgery versus WBRT).
Analysis 6.10.
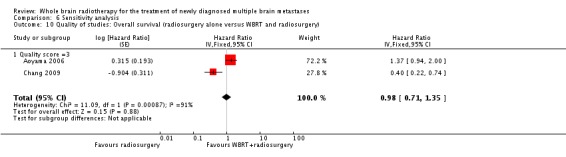
Comparison 6 Sensitivity analysis, Outcome 10 Quality of studies: Overall survival (radiosurgery alone versus WBRT and radiosurgery).
Analysis 6.11.

Comparison 6 Sensitivity analysis, Outcome 11 Quality of studies: Local control (radiosurgery alone versus WBRT and radiosurgery).
Analysis 6.12.

Comparison 6 Sensitivity analysis, Outcome 12 Quality of studies: Distant brain control (radiosurgery alone versus WBRT and radiosurgery).
What's new
Last assessed as up‐to‐date: 6 March 2012.
| Date | Event | Description |
|---|---|---|
| 21 September 2016 | Amended | Contact details updated. |
History
Protocol first published: Issue 4, 2002 Review first published: Issue 3, 2006
| Date | Event | Description |
|---|---|---|
| 8 March 2012 | New search has been performed | New search conducted on 19 July 2011. |
| 6 March 2012 | New citation required but conclusions have not changed | This update does not provide substantive changes to the original review. The title has been amended to: Whole brain radiotherapy for the treatment of newly diagnosed multiple brain metastases. The descriptor 'newly diagnosed' was added to clarify that the review only deals with upfront management rather than management at relapse. The literature search was updated in July 2011. Nine new trials (Chang 2009, Davey 2008, Graham 2010, Knisely 2008, Kocher 2011, Lee 2008, Neuhaus 2009, Scott 2007, Suh 2008) involving 1420 participants were added to this updated review. This updated review now includes a total of 39 trials involving 10,835 participants. The statistical analyses have now been revised. Hazard ratios (rather than odds ratios) are now provided for the outcomes of overall survival, local control and distant brain control. Risk ratios with 95% CI were recalculated with formal statistical input for the outcomes of neurologic function improvement and brain tumour response. In terms of implications for practice, the option of radiosurgery alone (without WBRT) in selected patients with newly diagnosed brain metastases has been added. |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Andrews 2004
| Methods | RCT enrolled: 333 evaluated: 331 | |
| Participants | Inclusion: 1. 1‐3 brain metastases 2. maximum size 4 cm (largest lesion) and others not more than 3 cm Exclusion: 1. KPS < 70 2. brainstem metastasis 3. metastasis less than 1 cm from optic apparatus 4. active extracranial disease judged by systemic treatment given within 1 month of enrollment | |
| Interventions | WBRT and radiosurgery boost vs WBRT | |
| Outcomes | 1. Overall survival 2. Local control, overall intracranial recurrence rates 3. cause of death 4. performance status 5. steroid usage | |
| Notes | QS = 3 | |
Antonadou 2002
| Methods | RCT enrolled: 134 evaluated: not reported | |
| Participants | Inclusion: 1. previously untreated brain metastases | |
| Interventions | WBRT with or without temozolomide chemotherapy | |
| Outcomes | 1. Response rates 2. Neurologic response 3. Overall survival | |
| Notes | Abstract | |
Aoyama 2006
| Methods | RCT enrolled: 160 evaluated: 132 | |
| Participants | Inclusion: 1. 1‐ 4 brain metastases 2. size ≤ 3 cm 3. pathologically confirmed systemic cancer Exclusion: 1. History of brain irradiation or surgery 2. small cell cancer, lymphoma, germinoma, myeloma, non‐melanomatous skin cancer | |
| Interventions | Radiosurgery alone versus WBRT and radiosurgery | |
| Outcomes | 1. Overall survival 2. Cause of death 3. Proportion with KPS ≥ 70 4. Neurologic functional preservation rate 5. Freedom from metachronous brain metastases 6. Freedom from new brain metastases 7. Salvage treatment for new/recurrent brain metastases 8. Local tumor control rate | |
| Notes | ||
Borgelt 1980
| Methods | RCT (2 studies) study 1: enrolled: 993 evaluated: 910 study 2: enrolled: 1001 evaluated: 902 | |
| Participants | Inclusion: 1. Brain metastases by clinical symptoms, EEG, radioisotope brain scans, arteriogram, pneumoencephalogram, biopsy Exclusion: 1. General medical or neoplastic status prohibiting adequate assessment 2. Other cancer treatment within two weeks of entry 3. Lesions too numerous or symptoms too vague for follow up | |
| Interventions | study 1: 4000 cGy/20 fr (227 pts) versus 4000 cGy/15 fr (233 pts) versus 3000 cGy/15 fr (217 pts) versus 3000 cGy/10 fr (233 pts) study 2: 4000 cGy/15 fr (227 pts) versus 3000 cGy/10 fr (228 pts) versus 2000 cGy/5 fr (447 pts) |
|
| Outcomes | 1. Survival 2. Palliative index | |
| Notes | QS = 2 | |
Borgelt 1980a
| Methods | study 1: enrolled: 993 evaluated: 910 | |
| Participants | See above | |
| Interventions | study 1: 4000 cGy/20 fr (227 pts) versus 4000 cGy/15 fr (233 pts) versus 3000 cGy/15 fr (217 pts) versus 3000 cGy/10 fr (233 pts) | |
| Outcomes | See above | |
| Notes | See above | |
Borgelt 1980b
| Methods | study 2: enrolled: 1001 evaluated: 902 | |
| Participants | See above | |
| Interventions | study 2: 4000 cGy/15 fr (227 pts) versus 3000 cGy/10 fr (228 pts) versus 2000 cGy/5 fr (447 pts) | |
| Outcomes | See above | |
| Notes | See above | |
Borgelt 1981
| Methods | RCT (2 studies) study 1: enrolled: 155 evaluated: 138 study 2: enrolled: 78 evaluated: 64 |
|
| Participants | Inclusion: 1. Brain metastases diagnosed by clinical symptoms and EEG, radioisotope brain scans, arteriograms, pneumoencephalograms, or biopsy Exclusion: ‐medical condition precluding adequate follow up ‐new anti‐cancer treatment within two weeks ‐lesions too numerous ‐symptoms too vague for adequate assessment |
|
| Interventions | Study 1: 1000 cGy/1 fr 3000 cGy ‐ 4000 cGy/10‐20 fr Study 2: 1200 cGy/ 2 fr 2000 cGy/5 fr |
|
| Outcomes | 1. Survival 2. Improvement in neurologic function (3. Treatment morbidity) | |
| Notes | QS = 2 | |
Borgelt 1981a
| Methods | study 1: enrolled: 155 evaluated: 138 | |
| Participants | See above | |
| Interventions | Study 1: 1000 cGy/1 fr versus 3000 cGy ‐ 4000 cGy/10‐20 fr | |
| Outcomes | See above | |
| Notes | See above | |
Borgelt 1981b
| Methods | study 2: enrolled: 78 evaluated: 64 | |
| Participants | See above | |
| Interventions | Study 2: 1200 cGy/ 2 fr versus 2000 cGy/5 fr | |
| Outcomes | See above | |
| Notes | See above | |
Chang 2009
| Methods | RCT enrolled: 58 evaluated: 58 |
|
| Participants | older than 18 years of age recursive partitioning analysis (RPA) class 1 or 2 Karnofsky performance status 70 or more one to three newly diagnosed brain metastases eligible for radiosurgery excluded if leukemia, lymphoma, germ cell, small cell cancer, leptomeningeal disease or unknown primary tumour |
|
| Interventions | radiosurgery and whole brain radiotherapy versus radiosurgery alone |
|
| Outcomes | Primary: neurocognitive endpoint of total recall | |
| Notes | the trial was stopped early (based on early stopping rules) on the basis that there was a high probability (96%) that patients randomized to radiosurgery and whole brain radiotherapy were more likely to show a decline in learning and memory | |
Chatani 1985
| Methods | RCT: enrolled: not stated evaluated: 69 | |
| Participants | Inclusion: 1. Brain metastases from lung cancer 2. Diagnosed with clinical symptoms and CT Exclusion: Not specified | |
| Interventions | 5000 cGy/20 fr versus 3000 cGy/10 fr | |
| Outcomes | 1. Survival 2. Neurologic improvement | |
| Notes | QS =1 | |
Chatani 1994
| Methods | RCT: enrolled: not stated evaluated: 162 | |
| Participants | Inclusion: 1. Brain metastases from lung cancer 2. Diagnosed with clinical symptoms and CT | |
| Interventions | Normal LDH: 5000 cGy/20 fr versus 3000 cGy/10 fr Elevated LDH: 3000 cGy/10 fr versus 2000 cGy/5 fr |
|
| Outcomes | 1. Survival 2. Neurologic improvement (3. Acute side‐effects) | |
| Notes | QS = 1 | |
Chougule 2000
| Methods | RCT: evaluated: 104 | |
| Participants | Inclusion: 1. Three lesions or less 2. tumor volume 30 cc or less 3. minimum life expectancy three months | |
| Interventions | Gamma knife radiosurgery versus Gamma knife radiosurgery and WBRT (3000 cGy/10 fr) versus WBRT (3000 cGy/10 fr) | |
| Outcomes | 1. Survival 2. Brain local control | |
| Notes | Abstract | |
Davey 2008
| Methods | RCT evaluated: 90 |
|
| Participants | confirmed malignant disease brain metastases ECOG performance status 0‐2 excluded patients with complete excision of a single brain metastasis and life expectancy less than 6 weeks |
|
| Interventions | whole brain radiotherapy 40 Gy in 20 fractions twice daily versus whole brain radiotherapy 20 Gy in 5 fractions once daily |
|
| Outcomes | Primary: overall survival |
|
| Notes | ||
DeAngelis 1989
| Methods | RCT: enrolled: not stated evaluated: 58 | |
| Participants | Inclusion: 1. Histologically confirmed cancer and brain mets (CT) Exclusion: 1. Prior WBRT | |
| Interventions | 3000 cGy/10 fr and lonidamide versus 3000 cGy/10 fr | |
| Outcomes | 1. Survival 2. Response rates (CR + PR) | |
| Notes | QS = 1 | |
Eyre 1984
| Methods | RCT: enrolled: 116 evaluated: 111 | |
| Participants | Inclusion: 1. Histological proof of malignancy 2. Clinical documentation of brain mets (isotope or CT) 3. Neurologic symptoms 4. No prior cranial radiation 5. Expected survival more than 4 wks 6. Adults Exclusion: 1. Systemic chemotherapy with agents that cross blood brain barrier |
|
| Interventions | 3000 cGy/10 fr and metronidazole versus 3000 cGy/10 fr | |
| Outcomes | 1. Survival 2. Response rates (CR + PR) | |
| Notes | QS = 2 | |
Graham 2010
| Methods | RCT: evaluated: 113 |
|
| Participants | ECOG performance status <3 life expectancy > 2 months extracranial disease (stable for 2 months, or absent or concurrent with brain metastases at time of initial cancer diagnosis) |
|
| Interventions | whole brain radiotherapy 40 Gy in 20 twice daily fractions versus whole brain radiotherapy 20 Gy in 4 daily fractions |
|
| Outcomes | primary: intracranial control and quality of life secondary: interval to CNS progression overall survival |
|
| Notes | ||
Guerrieri 2004
| Methods | RCT: planned sample: 300 closed due to poor accrual: 42 | |
| Participants | Inclusion: 1. metastatic NSCLC to brain 2. inoperable CNS disease or refused surgery 3. WHO performance status zero to two 4. adequate blood counts 5. urinary creatinine ≥ 50 mL/min Exclusion: 1. Prior chemotherapy 2. Prior brain radiotherapy |
|
| Interventions | 2000 cGy/5 fr versus 2000 cGy/5 fr and carboplatin | |
| Outcomes | 1. Overall survival 2. Radiographic brain response 3. Symptom control 4. Toxicity | |
| Notes | QS = 1 | |
Harwood 1977
| Methods | RCT: enrolled: 108 evaluated: 101 | |
| Participants | Inclusion: 1. Brain metastases Exclusion: 1. Previous WBRT 2. Chemotherapy given three weeks prior | |
| Interventions | 1000 cGy/1 fr versus 3000 cGy/10 fr | |
| Outcomes | 1. Survival 2. Local control (3. Response frequency) (4. Complication rate) | |
| Notes | QS = 2 | |
Horton 1971
| Methods | RCT: evaluated: 47 | |
| Participants | Inclusion: 1. Histologically proven cancer 2. Parenchymal brain metastases (radio‐isotope brain scans, EEG, echo‐encephalograms, angiograms, spinal fluid cytology and chemistry) Exclusion: 1. All gross brain tumour surgically excised |
|
| Interventions | Supportive care (oral prednisone) versus Supportive care (oral prednisone) and WBRT | |
| Outcomes | 1. Survival 2. Improvement in performance status | |
| Notes | QS =2 | |
Knisely 2008
| Methods | RCT enrolled: 332 evaluated: 183 |
|
| Participants | 18 years of age or older anticipated survival more than 8 weeks Zubrod performance status of 0‐1 MRI documented brain metastases histologically proven extracranial solid tumour not eligible for radiosurgery (due to size more than 4 cm, number more than 3 or location eg. midbrain) |
|
| Interventions | whole brain radiotherapy versus whole brain radiotherapy and thalidomide |
|
| Outcomes | primary: survival secondary: central nervous system progression rates toxicity |
|
| Notes | ||
Kocher 2011
| Methods | RCT enrolled: 359 evaluated: 359 |
|
| Participants | 1. age 18 years or older 2. WHO performance status 2 or less 3. 1‐3 brain metastases 4. Radiosurgery: single metastasis 3.5 cm or less, multiple metastases 2.5 cm or less 5. Surgery: complete surgical resection 6. stable systemic cancer for 3 months or more and/or asymptomatic synchronous primary tumor wtihout metastases outside the CNS or unknown primary tumor 7. excluded small cell lung cancer, lymphoma, leukemia, myeloma, germ cell tumors, brainstem metastases, leptomeningeal metastases, recurrent brain metastases |
|
| Interventions | 1. radiosurgery alone 2. radiosurgery and whole brain radiotherapy 1. surgery alone 2. surgery and whole brain radiotherapy |
|
| Outcomes | Primary outcome: ‐duration of functional independence (measured from the day of random assignment to the first report of deterioration of a WHO PS of more than 2 Secondary outcomes: ‐frequency of intracranial relapse at initially treated sites and at new sites ‐progression‐free survival ‐overall survival ‐late toxicities ‐quality of life |
|
| Notes | ||
Komarnicky 1991
| Methods | RCT: enrolled: 859 evaluated: 779 | |
| Participants | Inclusion: 1. Brain metastases with measurable disease 2. 18 to 75 years of age 3. KPS ≥ 40 4. Neurologic function class (NFC) 1, 2, 3 5. If on chemo must not have had change in drugs within two weeks prior to randomization, during RT/misonidazole or two weeks after completion of RT | |
| Interventions | 3000 cGy/10 fr and misonidazole versus 3000 cGy/10 fr | |
| Outcomes | 1. Survival | |
| Notes | QS = 3 | |
Kondziolka 1999
| Methods | RCT: evaluated: 27 | |
| Participants | Inclusion: 1. Histologic confirmation of cancer 2. 25 mm or less in size 3. More than 5 mm from the optic chiasm 4. two to four brain metastases Exclusion: Participants unable to undergo and MRI |
|
| Interventions | WBRT (3000 cGy/10 fr) versus WBRT (3000 cGy/10 fr) and radiosurgery | |
| Outcomes | 1. Survival 2. Local brain control | |
| Notes | QS = 1 | |
Kurtz 1981
| Methods | RCT: enrolled: 309 evaluated: 255 | |
| Participants | Inclusion: 1. Cancer patients with positive isotope brain scans 2. No evidence of disseminated disease Exclusion: 1. NFC IV 2. Anti‐cancer treatment within previous 2 wks 3. Progressive untreated primary | |
| Interventions | 5000 cGy/20 fr versus 3000 cGy/10 fr | |
| Outcomes | 1. Survival 2. Median time to progression 3. Palliation of symptoms (4. Improvement rate) (5. Cause of death) | |
| Notes | QS = 3 | |
Lee 2008
| Methods | RCT enrolled: 48 evaluated: 48 |
|
| Participants | pathologically confirmed nonsmall cell lung cancer clinically silent brain metastases (not surgically resectable) age 18‐75 years ECOG performance status 0‐3 adequate bone marrow, hepatic, renal functions no prior chemotherapy nor molecular‐targeted therapy |
|
| Interventions | chemotherapy first followed by whole brain radiation versus whole brain radiation followed by chemotherapy |
|
| Outcomes | progression‐free survival tumour response quality of life toxicity |
|
| Notes | ||
Mehta 2003
| Methods | RCT: enrolled: not reported evaluated: 401 | |
| Participants | Inclusion: 1. MRI demonstrated brain metastases 2. histologically proven solid tumours 3. required WBRT 4. KPS of at least 70 Exclusion: 1. small cell lung cancer, lymphoma or germ‐cell tumors 2. brain metastases partially or completely resected 3. received prior cranial irradiation 4. leptomeningeal metastases 5. two or more sites of extracranial metastases except when breast was the primary cancer 6. chemotherapy planned during WBRT | |
| Interventions | WBRT (3000 cGy/10 fr and motexafin gadolinium vs WBRT (3000 cGy/10 fr) | |
| Outcomes | 1. Survival 2. Neurologic progression | |
| Notes | QS = 2 | |
Meyers 2004
| Methods | RCT: enrolled: not reported evaluated: 401 | |
| Participants | see Mehta 2003 | |
| Interventions | WBRT (3000 cGy/10 fr and motexafin gadolinium vs. WBRT (3000 cGy/ 10 fr) | |
| Outcomes | 1. neurocognitive function and progression | |
| Notes | Same trial as Mehta | |
Mornex 2003
| Methods | RCT: enrolled: 76 evaluated: 76 | |
| Participants | Inclusion: 1. histologically confirmed malignant melanoma 2. at least one measurable, non‐resectable cerebral lesion 3. age 18 to 70 years 4. life expectancy greater than three months 5. adequate blood counts 6. satisfactory liver and renal function 7. no previous chemotherapy within last four weeks 8. No previous cranial irradiation 9. Informed consent | |
| Interventions | fotemustine versus fotemustine and WBRT | |
| Outcomes | 1. cerebral response rates (complete and partial) and control rate (complete, partial and stable disease) and time to cerebral progression on day 50 2. overall response rate, time to overall progression, overall survival | |
| Notes | QS = 1 | |
Murray 1997
| Methods | RCT: enrolled:445 evaluated: 429 | |
| Participants | Inclusion: 1. Proof of underlying primary 2. Measureable brain lesion(s) on CT/MRI 3. KPS ≥ 70 4. NFC 1 or 2 Exclusion: 1. Primary site hematopoeitc origin 2. Metastatic lymphoma involving meninges | |
| Interventions | 5440 cGy/34 fr (BID over 17 days) versus 3000 cGy/10 fr (over 10 days) | |
| Outcomes | 1. Survival (2. Acute toxicity) | |
| Notes | QS = 2 | |
Neuhaus 2009
| Methods | RCT enrolled: 96 evaluated: 96 |
|
| Participants | histologically proven lung cancer (small cell or nonsmall cell) 18‐75 years of age at least one measurable lesion in the brain sufficient bone marrow reserve, renal function tests ECOG performance status 0‐2 excluded leptomeningeal metastases |
|
| Interventions | whole brain radiation alone versus whole brain radiation and topotecan |
|
| Outcomes | response rate progression‐free survival overall survival quality of life |
|
| Notes | ||
Phillips 1995
| Methods | RCT: evaluated: 72 | |
| Participants | Inclusion: 1. Metastatic cancer to brain (based on neurodiagnostic tests, biopsy of primary, biopsy of brain lesion) 2. KPS ≥ 70 3. Age > 18 y.o. 4. Measurable lesion(s) 5. WBC > 4000/mm3 6. Platelets > 125 000/mm3 7. NFC 1, 2 Exclusion: 1. Prior brain RT 2. Complete surgical excision 3. CNS primary, leukemia, lymphoma 4. Concurrent chemotherapy 5. Primary absent or controlled or under RT |
|
| Interventions | 3750 cGy/15 fr and BrdU versus 3750 cGy/15 fr | |
| Outcomes | 1. Survival 2. Response (CR + PR) | |
| Notes | QS = 3 | |
Postmus 2000
| Methods | RCT enrolled: 134 evaluated: 120 | |
| Participants | Inclusion: 1. metastatic small cell lung cancer 2. brain metastases (CT) 3. extracranial tumour 4. no previous treatment with chemotherapy or radiation for brain metastases 5. no prior teniposide 6. age less than 76 years Exclusion: 1. uncontrolled infection 2. serious non‐malignant disease | |
| Interventions | teniposide chemotherapy with or without WBRT | |
| Outcomes | 1. Survival 2. Time to progression 3. Duration of response | |
| Notes | QS =1 | |
Priestman 1996
| Methods | RCT: enrolled: 533 evaluated: 352 | |
| Participants | Inclusion: 1. Symptomatic cerebral metastases confirmed on CT/radioisotope scans/biopsy 2. Histologic confirmation of primary 3. If no obvious primary, biopsy brain metastases 4. Presence of symptoms related to brain metastases 5. Stable dose of decadron 1 wk prior to randomization 6. 16 y.o. and over 7. WHO performance status zero to three; NFC < 4 (MRC) Exclusion: 1. Cytotoxic chemotherapy in prior four weeks | |
| Interventions | 1200 cGy/2 fr versus 3000 cGy/10 fr | |
| Outcomes | 1. Survival 2. Neurologic symptom response (3. Cause of death) (4. Morbidity) | |
| Notes | QS = 3 | |
Robinet 2001
| Methods | RCT: enrolled: 176 evaluated: 171 | |
| Participants | Inclusion: 1. metastatic non‐small cell lung cancer 2. at least one measurable brain metastasis 3. ECOG performance two or less 4. good renal and hematologic function Exclusion: 1. recent myocardial infarction, congestive heart failure or arrhythmia 3. prior chemotherapy or brain radiotherapy 4. previous cancer (non‐melanoma skin cancer or in situ cervix cancer allowed) |
|
| Interventions | early versus delayed WBRT and concurrent cisplatin and vinorelbine chemotherapy | |
| Outcomes | 1. Survival 2. Progression‐free survival 3. Response | |
| Notes | QS = 3 | |
Scott 2007
| Methods | RCT enrolled: 538 (non‐small cell lung cancer, breast cancer and other primaries) evaluated: 106 (subgroup of breast cancer) |
|
| Participants | brain metastases RTOG recursive partitioning analysis (class I or II) no prior treatment of brain metastases (except partial resection allowed) chemotherapy discontinued at least 7 days prior to the start of whole brain radiotherapy and not to be restarted until at least 1 month after whole brain radiotherapy excluded small cell lung cancer, germ cell tumours and lymphomas |
|
| Interventions | whole brain radiotherapy versus whole brain radiotherapy and efaproxiral |
|
| Outcomes | Primary: overall survival Secondary: quality of life |
|
| Notes | subgroup analysis (not a priori) | |
Stea 2006
| Methods | ||
| Participants | ||
| Interventions | ||
| Outcomes | ||
| Notes | ||
Suh 2006
| Methods | RCT: enrolled: 538 evaluated: 515 | |
| Participants | Inclusion: 1. RPA class I or II 2. no prior brain metastases treatment 3. age ≥ 18 years 4. adequate hematologic, hepatic, renal function 5. no other concurrent active malignancy 6. no planned therapy for brain metastases one month post follow‐up visit 7. pulse oximetry ≥ 90% Exclusion: 1. small cell lung cancer, germ cell or lymphoma 2. pregnant or breast‐feeding 3. prior exposure to efaproxiral 4. chemotherapy within seven days or investigational agents within 28 days of whole brain radiotherapy |
|
| Interventions | 3000 cGy/10 fr versus 3000 cGy/10 fr plus efaproxiral | |
| Outcomes | 1. Survival 2. Radiographic brain response 3. Radiologic and clinical brain progression 4. Cause of death 5. Quality of life | |
| Notes | QS = 3 | |
Suh 2008
| Methods | RCT enrolled: 368 evaluated: 365 |
|
| Participants | breast cancer brain metastases Karnofsky performance status 70 or more no other active concurrent malignancy |
|
| Interventions | whole brain radiotherapy versus whole brain radiotherapy and efaproxiral |
|
| Outcomes | overall survival | |
| Notes | ||
Ushio 1991
| Methods | RCT enrolled: 100 evaluated: 88 | |
| Participants | Inclusion: 1. brain metastases from lung cancer 2. extracranial cancer relatively well controlled 3. good general condition | |
| Interventions | WBRT versus WBRT + chloroethylnitrosoureas versus WBRT + chloroethylnitrosoureas + tegafur | |
| Outcomes | 1. Response rates 2. Survival | |
| Notes | QS = 2 | |
CNS = Central Nervous System CR = complete response CT = computed tomography ECOG = Eastern Co‐operative Oncology Group EEG = electroencephalography KPS = Karnofsky performance status MRC = Medical Research Council NFC = neurologic functional classification NSCLC = non‐small cell lung cancer PR = partial response QS = quality score RCT = randomized controlled trial RPA = recursive partitioning analysis RT = radiotherapy y.o. = years old WBC = white blood count WBRT = whole brain radiotherapy
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Haie‐Meder 1993 | This RCT examined altered dose‐fractionation schedules and was excluded based on no standard arm (3000 cGy/10 fr or 2000 cGy/5 fr). The randomizations were: One course of WBRT: 1800 cGy/3 fr/3 d versus Two courses of WBRT: 1800 cGy/3 fr/3 d followed 1 month later by another 1800 cGy/3 fr/3 d or 1800 cGy/3 fr/3 d followed one month later by another 2500 cGy/10 fr/2 weeks |
Characteristics of ongoing studies [ordered by study ID]
ASCOSOG‐Z0300
| Trial name or title | Phase III randomized study of radiosurgery with or without WBRT in patients with one to three cerebral metastases |
| Methods | |
| Participants | 1. participants with one to three brain metastases (less than 3 cm) 2. not within 5 mm of optic chiasm or within brainstem 3. no germ cell tumour, small cell carcioma or lymphoma 4. no leptomeningeal disease |
| Interventions | Radiosurgery versus radiosurgery and WBRT |
| Outcomes | 1. survival 2. time brain failure 3. quality of life, functional independence, neurocognitive status 4. toxicity |
| Starting date | |
| Contact information | |
| Notes |
CCCWFU‐97600
| Trial name or title | NCI‐P02‐0211 Phase III randomized study of d‐methylphenidate to improve quality of life in patients receiving radiotherapy for primary or metastatic brain tumours |
| Methods | |
| Participants | 1. histologically confirmed metastatic cancer to brain or primary brain tumor 2. planned cranial irradiation (partial or whole brain) with a total dose of at least 25 Gy |
| Interventions | d‐methylphenidate (oral) versus placebo |
| Outcomes | 1. fatigue score 2. quality of life 3. depression 4. neurocognitive function |
| Starting date | |
| Contact information | |
| Notes |
EORTC‐18981
| Trial name or title | Phase III randomized study of temozolomide with or without WBRT in patients with stage IV melanoma with asymptomatic brain metastases |
| Methods | |
| Participants | 1. histologically confirmed unresectable metastatic melanoma 2. concurrent visceral/soft tissue metastases 3. at least one site of measurable disease 4. documented evidence of disease progression 5. no neurologic symptoms |
| Interventions | Temozolomide chemotherapy versus temozolomide chemotherapy and WBRT |
| Outcomes | 1. survival 2. time to appearance of neurologic symptoms 3. progression‐free survival 4. quality of life and quality‐adjusted survival 5. toxicity |
| Starting date | Closed |
| Contact information | |
| Notes |
| Trial name or title | Protocol ID: 040066 Surgery versus radiosurgery to treat metastatic brain tumors |
| Methods | |
| Participants | Eligibility: 1. histologically confirmed cancer 2. one to three brain metastases 3. less than or equal to 4 cm Exclusion: 1. brainstem lesions 2. tumours within 10 mm from optic apparatus 3. poor operative candidates 4. leptomeningeal disease 5. psychiatric impairments 6. infection 7. germ cell tumour, lymphoma, small cell lung cancer |
| Interventions | Surgery versus radiosurgery |
| Outcomes | 1. survival 2. tumour recurrence or progression 3. neurologic sign or symptom developmen 4. functional independencet 5. steroid and anticonvulsant use 6. quality of life |
| Starting date | |
| Contact information | |
| Notes |
| Trial name or title | Protocol ID: P03247 Temozolomide for treatment of brain metastases from non‐small cell lung cancer |
| Methods | |
| Participants | 1. histologically confirmed non‐small cell lung cancer 2. at least one brain metastases 3. no previous WBRT or radiosurgery 4. no more than two sites of extracranial disease 5. KPS greater than 70 |
| Interventions | WBRT plus temozolomide versus WBRT plus placebo |
| Outcomes | 1. survival 2. improvement in time to radiologic CNS progression |
| Starting date | |
| Contact information | |
| Notes |
QUARTZ
| Trial name or title | Medical Research Council: Quality of Life After Radiotherapy and Steroids |
| Methods | |
| Participants | 1. non‐small cell lung cancer 2. inoperable brain metastases |
| Interventions | WBRT plus optimal supportive care including dexamethasone vs optimal supportive care alone (including dexamethasone) |
| Outcomes | 1. quality adjusted life years 2. overall survival 3. KPS 4. neurologic symptoms 5. effect on the career's quality of life |
| Starting date | |
| Contact information | |
| Notes |
RTOG‐BR0118/ DEV1006
| Trial name or title | Phase III randomized study of radiotherapy with or without thalidomide in patients with multiple brain metastases |
| Methods | |
| Participants | 1. histologically confirmed cancer 2. multiple brain metastases 3. at least one measurable brain metastasis by MRI more than 4 cm or located in brainstem |
| Interventions | WBRT versus WBRT and thalidomide |
| Outcomes | 1. survival 2. time to tumour progression 3. time to neurocognitive progression 4. cause of death 5. toxic effects 6. quality of life |
| Starting date | |
| Contact information | |
| Notes |
UCLA‐0302038
| Trial name or title | PCI‐P‐PCYC‐0211 Phase III randomized study of motexafin gadolinium and WBRT versus WBRT alone in patients with brain metastases secondary to non‐small cell lung cancer |
| Methods | |
| Participants | 1. histologically confirmed non‐small cell lung cancer 2. no liver metastases 3. no extracranial metastases to more than one organ 4. no leptomeningeal metastases |
| Interventions | WBRT versus WBRT and motexafin gadolinium |
| Outcomes | 1. time to neurologic progression or death (with neurologic progression) 2. time to neurocognitive progression 3. time to loss of functional independence 4. time to all cause mortality |
| Starting date | |
| Contact information | |
| Notes |
CNS = central nervous system KPS = Karnofsky performance status MRI = magnetic resonance imaging RSR 13 = efaproxiral WBRT = whole brain radiotherapy
Contributions of authors
MNT: literature search and identification of trials for inclusion; evaluation of methodological quality of included trials; abstraction of data; writing review. Main responsibility for updating review (August 2007). NL: literature search and identification of trials for inclusion; abstraction of data; assistance in writing review. RW: assistance in writing review; abstraction of data. EC: literature search and identification of trials for inclusion; evaluation of methodological; quality of included trials; abstraction of data. ER: assistance in writing review.
WX: statistical analysis.
AS: assistance in writing review.
Sources of support
Internal sources
University of Toronto, Odette Cancer Centre, Toronto, ON, Canada.
External sources
Cancer Care Ontario Program in Evidence‐based Care's Supportive Care Guidelines Group and Neuro‐oncology Disease Site Group, Canada.
Declarations of interest
None known
Edited (no change to conclusions)
References
References to studies included in this review
- Andrews DW, Scott CB, Sperduto PW, et al. Whole brain radiation therapy with or without stereotactic radiosurgery boost for patients with one to three brain metastases: phase III results of the RTOG 9508 randomised trial. Lancet 2004;363:1665‐72. [DOI] [PubMed] [Google Scholar]
- Antonadou D, Coliarakis N, Paraskevaidis M, Athansiou H, Sarris G, Synodinou M, et al. Whole brain radiotherapy alone or in combination with temozolamide for brain metastases. A phase III study (abstract). International Journal of Radiation Oncology, Biology, Physics 2002;54:93. [Google Scholar]
- Aoyama H, Shirato H, Tago M, et al. Stereotactic radiosurgery plus whole‐brain radiation therapy vs stereotactic radiosurgery alone for treatment of brain metastases. A randomized controlled trial. JAMA 2006;295(21):2483‐91. [DOI] [PubMed] [Google Scholar]
- Borgelt B, Gelber R, Kramer S, Brady LW, Chang CH, Davis LW, et al. The palliation of brain metastases: final results of the first two studies by the Radiation Therapy Oncology Group. International Journal of Radiation Oncology, Biology, Physics 1980;6:1‐9. [DOI] [PubMed] [Google Scholar]
- Borgelt B, Gelber R, Kramer S, et al. The palliation of brain metastases: final results of the first two studies by the Radiation Therapy Oncology Group. International Journal of Radiation Oncology, Biology, Physics 1980;6:1‐9. [DOI] [PubMed] [Google Scholar]
- Borgelt B, Gelber R, Kramer S, et al. The palliation of brain metastases: Final results of the first two studies by the Radiation Therapy Oncology Group. International Journal of Radiation Oncology, Biology, Physics 1980;6:1‐9. [DOI] [PubMed] [Google Scholar]
- Borgelt B, Gelber R, Larson M, Hendrickson F, Griffin T, Roth R. Ultra‐rapid high dose irradiation schedules for the palliation of brain metastases: final results of the first two studies by the Radiation Therapy Oncology Group. International Journal of Radiation Oncology, Biology, Physics 1981;7:1633‐8. [DOI] [PubMed] [Google Scholar]
- Borgelt B, Gelber R, Larson M, et al. Ultra‐rapid high dose irradiation schedules for the palliation of brain metastases: final results of the first two studies by the Radiation Therapy Oncology Group. International Journal of Radiation Oncology, Biology, Physics 1981;7:1633‐8. [DOI] [PubMed] [Google Scholar]
- Borgelt B, Gelber R, Larson M, et al. Ultra‐rapid high dose irradiation schedules for the palliation of brain metastases: final results of the first two studies by the Radiation Therapy Oncology Group. International Journal of Radiation Oncology, Biology, Physics 1981;7:1633‐8. [DOI] [PubMed] [Google Scholar]
- Chang E, Wefel JS, Hess KR, et al. Neurocognition in patients with brain metastases treated with radiosurgery or radiosurgery plus whole‐brain irradiation: a randomised controlled trial. Lancet Oncology 2009;10:1037‐44. [DOI] [PubMed] [Google Scholar]
- Chatani M, Teshima T, Hata K, Inoue T, Suzuki T. Whole brain irradiation for metastases from lung carcinoma. A clinical investigation. Acta Radiological Oncology 1985;24:311‐4. [DOI] [PubMed] [Google Scholar]
- Chatani M, Matayoshi Y, Masaki N, Inoue T. Radiation therapy for brain metastases from lung carcinoma. Prospective randomized trial according to level of lactate dehydrogenase. Strahlentherapie Onkologie 1994;170:155‐61. [PubMed] [Google Scholar]
- Chougule PB, Burton‐Williams M, Saris S, Zheng Z, Ponte B, Noren G, et al. Randomized treatment of brain metastases with gamma knife radiosurgery, whole brain radiotherapy or both (abstract). International Journal of Radiation Oncology, Biology, Physics 2000;48:114. [Google Scholar]
- Davey P, Hoegler D, Ennis M, Smith J. A phase III study of accelerated versus conventional hypofractionated whole brain irradiation in patients of good performance status with brain metastases not suitable for surgical excision. Radiotherapy and Oncology 2008;88:173‐6. [DOI] [PubMed] [Google Scholar]
- DeAngelis LM, Currie VE, Kim J‐H, Krol G, O'Hehir MA, Farag FM, et al. The combined use of radiation therapy and lonidamide in the treatment of brain metastases. Journal of Neuro‐oncology 1989;7:241‐7. [DOI] [PubMed] [Google Scholar]
- Eyre HJ, Ohlsen JD, Frank J, LoBuglio AF, McCracken JD, Weatherall TJ, Mansfield CM. Randomized trial of radiotherapy versus radiotherapy plus metronidazole for the treatment of metastatic cancer to brain. Journal of Neuro‐oncology 1984;2:325‐30. [DOI] [PubMed] [Google Scholar]
- Graham PH, Bucci J, Browne L. Randomized comparison of whole brain radiotherapy, 20 gy in four daily fractions versus 40 Gy in 20 twice‐daily fractions, for brain metastases. International Journal of Radiation Oncology, Biology, Physics 2010;77(3):648‐54. [DOI] [PubMed] [Google Scholar]
- Guerrieri M, Wong K, Ryan G, Millward M, Quong G, Ball DL. A randomised phase III study of palliative radiation with concomitant carboplatin for brain metastases from non‐small cell carcinoma of lung. Lung Cancer 2004;46:107‐11. [DOI] [PubMed] [Google Scholar]
- Harwood AR, Simpson WJ. Radiation therapy of cerebral metastases: A randomized prospective clinical trial. International Journal of Radiation Oncology, Biology, Physics 1977;2:1091‐4. [DOI] [PubMed] [Google Scholar]
- Horton J, Baxter DH, Olson KB. The management of metastases to the brain by irradiation and corticosteroids. The American Journal of Roentgenology, Radium Therapy, and Nuclear Medicine 1971;3:334‐6. [DOI] [PubMed] [Google Scholar]
- Knisely J, Berkey B, Chakravarti A, et al. A phase III study of conventional radiation therapy plus thalidomide versus conventional radiation therapy for multiple brain metastases (RTOG 0118). International Journal of Radiation Oncology, Biology, Physics 2008;71(1):79‐86. [DOI] [PubMed] [Google Scholar]
- Kocher M, Soffietti R, Abacioglu U, et al. Adjuvant whole‐brain radiotherapy versus observation after radiosurgery or surgical resection of one to three cerebral metastases: results of the EORTC 22952‐26001 study. Journal of Clinical Oncology 2011;29(2):134‐41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Komarnicky LT, Phillips TL, Martz K, Asbell S, Isaacson S, Urtasun R. A randomized phase III protocol for the evaluation of misonidazole combined with radiation in the treatment of patients with brain metastases (RTOG‐7916). International Journal of Radiation Oncology, Biology, Physics 1991;20:53‐8. [DOI] [PubMed] [Google Scholar]
- Kondziolka D, Patel A, Lunsford LD, Kassam A, Flickinger JC. Stereotactic radiosurgery plus whole brain radiotherapy versus radiotherapy alone for patients with multiple brain metastases. International Journal of Radiation Oncology, Biology, Physics 1999;45(2):427‐34. [DOI] [PubMed] [Google Scholar]
- Kurtz JM, Gelber R, Brady LW, Carella RJ, Cooper JS. The palliation of brain metastases in a favourable patient population: a randomized clinical trial by the Radiation Therapy Oncology Group. International Journal of Radiation Oncology, Biology, Physics 1981;7:891‐5. [DOI] [PubMed] [Google Scholar]
- Lee DH, Han J‐Y, Kim HT, et al. Primary chemotherapy for newly diagnosed nonsmall cell lung cancer patients with synchronous brain metastases compared with whole‐brain radiotherapy administered first. Results of a randomized pilot study. Cancer 2008;113:143‐9. [DOI] [PubMed] [Google Scholar]
- Mehta MP, Rodrigus P, Terhaard CHJ, Rao A, Suh J, Roa W, et al. Survival and neurologic outcomes in a randomized trial of motexafin gadolinium and whole‐brain radiation therapy in brain metastases. Journal of Clinical Oncology 2003;21:2529‐36. [DOI] [PubMed] [Google Scholar]
- Meyers CA, Smith JA, Bezjak A, Mehta MP, Liebmann J, Illidge T, et al. Neurocognitive function and progression in patients with brain metastases treated with whole‐brain radiation and motexafin gadolinium: results of a randomized phase III trial. Journal of Clinical Oncology 2004;22(1):157‐65. [DOI] [PubMed] [Google Scholar]
- Mornex F, Thomas L, Mohr P, Hauschild A, Delaunay MM, Lesimple T. A prospective randomized multicentre phase III trial of fotemustine plus whole brain irradiation versus fotemustine alone in cerebral metastases of malignant melanoma. Melanoma Research 2003;13(1):97‐103. [DOI] [PubMed] [Google Scholar]
- Murray KJ, Scott C, Greenberg HM, Emami B, Seider M, Vora NL, et al. A randomized phase III study of accelerated hyperfractionation versus standard in patients with unresected brain metastases: a report of the Radiation Therapy Oncology Group (RTOG) 9104. International Journal of Radiation Oncology, Biology, Physics 1997;39:571‐4. [DOI] [PubMed] [Google Scholar]
- Neuhaus T, Ko Y, Muller RP, et al. A phase III trial of topotecan and whole brain radiation therapy for patients with CNS‐metastases due to lung cancer. British Journal of Cancer 2009;100:291‐7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- PhillipsTL, Scott CB, Leibel SA, Rotman M, Weigensberg IJ. Results of a randomized comparison of radiotherapy and bromodeoxyuridine with radiotherapy alone for brain metastases: report of RTOG trial 89‐05. International Journal of Radiation Oncology, Biology, Physics 1995;33:339‐48. [DOI] [PubMed] [Google Scholar]
- Postmus PE, Haaxma‐Reiche H, Smit EF, Groen HJM, Karnicka H, Lewinski T, et al. Treatment of brain metastases of small‐cell lung cancer: comparing teniposide and teniposide with whole‐brain radiotherapy‐ a phase III study of the European Organization for the Research and Treatment of Lung Cancer Cooperative Group. Journal of Clinical Oncology 2000;18:3400‐8. [DOI] [PubMed] [Google Scholar]
- Priestman TJ, Dunn J, Brada M, Rampling R, Baker PG. Final results of the Royal College of Radiologists' trial comparing two different radiotherapy schedules in the treatment of cerebral metastases. Clinical Oncology 1996;8:308‐15. [DOI] [PubMed] [Google Scholar]
- Robinet G, Thomas P, Breton JL, Lena H, Gouva S, Dabouis G, et al. Results of a phase III study of early versus delayed whole brain radiotherapy with concurrent cisplatin and vinorelbine combination in inoperable brain metastasis of non‐small cell lung cancer: Groupe Francaise de Pneumo‐Cancerologie (GFPC) protocol 95‐1. Annals of Oncology 2001;12:59‐67. [DOI] [PubMed] [Google Scholar]
- Scott C, Suh J, Stea B, Nabid A, Hackman J. Improved survival, quality of life, and quality‐adjusted survival in breast cancer patients treated with efaproxiral (Efaproxyn) plus whole‐brain radiation therapy for brain metastases. American Journal of Clinical Oncology 2007;30:580‐7. [DOI] [PubMed] [Google Scholar]
- Stea B, Suh JH, Boyd AP, et al. Whole‐brain radiotherapy with or without efaproxiral for the treatment of brain metastases: determinants of response and its prognostic value for subsequent survival. International Journal of Radiation Oncology, Biology, Physics 2006;64(4):1023‐30. [DOI] [PubMed] [Google Scholar]
- Stea B, Suh JH, Boyd AP, Cagnoni PJ, Shaw E. Whole‐brain radiotherapy with or without efaproxiral for the treatment of brain metastases: determinants of response and its prognostic value for subsequent survival. International Journal of Radiation Oncology, Biology, Physics 2006;64(4):1023‐30. [DOI] [PubMed] [Google Scholar]; Suh JH, Stea B, Nabid A, Kresl JJ, Fortin A, Mercier J‐P, et al. Phase III study of efaproxiral as an adjunct to whole‐brain radiation therapy for brain metastases. Journal of Clinical Oncology 2006;24:106‐14. [DOI] [PubMed] [Google Scholar]
- Suh JH, Stea B, Tankel K, et al. Results of the phase III ENRICH (RT‐016) study of efaproxiral administered concurrent with whole brain radiation therapy (WBRT) in women with brain metastases from breast cancer. International Journal of Radiation Oncology, Biology, Physics 2008;72 Suppl(1):50‐1. [Google Scholar]
- Ushio Y, Arita N, Hayakawa T, Mogami H, Hasegawa H, Bitoh S, et al. Chemotherapy of brain metastases from lung carcinoma: a controlled randomized study. Neurosurgery 1991;28:201‐5. [DOI] [PubMed] [Google Scholar]
References to studies excluded from this review
- Haie‐Meder C, Pellae‐Cosset B, Laplanche A, Lagrange JL, Tuchais C, Nogues C, et al. Results of a randomized clinical trial comparing two radiation schedules in the palliative treatment of brain metastases. Radiotherapy and Oncology 1993;26:111‐6. [DOI] [PubMed] [Google Scholar]
References to ongoing studies
- Phase III randomized study of radiosurgery with or without WBRT in patients with one to three cerebral metastases. Ongoing study Starting date of trial not provided. Contact author for more information.
- NCI‐P02‐0211 Phase III randomized study of d‐methylphenidate to improve quality of life in patients receiving radiotherapy for primary or metastatic brain tumours. Ongoing study Starting date of trial not provided. Contact author for more information.
- Phase III randomized study of temozolomide with or without WBRT in patients with stage IV melanoma with asymptomatic brain metastases. Ongoing study Closed.
- Protocol ID: 040066 Surgery versus radiosurgery to treat metastatic brain tumors. Ongoing study Starting date of trial not provided. Contact author for more information.
- Protocol ID: P03247 Temozolomide for treatment of brain metastases from non‐small cell lung cancer. Ongoing study Starting date of trial not provided. Contact author for more information.
- Medical Research Council: Quality of Life After Radiotherapy and Steroids. Ongoing study Starting date of trial not provided. Contact author for more information.
- Phase III randomized study of radiotherapy with or without thalidomide in patients with multiple brain metastases. Ongoing study Starting date of trial not provided. Contact author for more information.
- PCI‐P‐PCYC‐0211 Phase III randomized study of motexafin gadolinium and WBRT versus WBRT alone in patients with brain metastases secondary to non‐small cell lung cancer. Ongoing study Starting date of trial not provided. Contact author for more information.
Additional references
- Ahmed N, Ahmedzai S, Vora V, Hilam S, Paz S. Supportive care for patients with gastrointestinal cancer. Cochrane Database of Systematic Reviews 2004, Issue 3. [DOI: 10.1002/14651858.CD004282.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arbit E, Wronski M. The treatment of brain metastases. Neurosurgery Quarterly 1995;5:1. [Google Scholar]
- Cairncross JG, Kim JH, Posner JB. Radiation therapy for brain metastases. Annals of Neurology 1980;7:529‐41. [DOI] [PubMed] [Google Scholar]
- Chao JH, Philips R, Nickson JJ. Roentgen ray therapy for cerebral metastases. Cancer 1954;7:682‐9. [DOI] [PubMed] [Google Scholar]
- Detsky AS, Naylor CD, O'Rourke K, McGeer AJ, L'Abbe KA. Incorporating variations in the quality of individual randomized trials into meta‐analysis. Journal of Clinical Epidemiology 1992;45:255‐65. [DOI] [PubMed] [Google Scholar]
- Gaspar L, Scott C, Rotman M, Asbell S, Phillips T, Wasserman T, et al. Recursive partitioning analysis (RPA) of prognostic factors in three Radiation Therapy Oncology Group (RTOG) brain metastases trials. International Journal of Radiation Oncology, Biology, Physics 1997;37:745‐51. [DOI] [PubMed] [Google Scholar]
- Gelber RD, Larson M, Borgelt BB, Kramer S. Equivalence of radiation schedules for the palliative treatment of brain metastases in patients with favorable prognosis. Cancer 1981;48:1749‐53. [DOI] [PubMed] [Google Scholar]
- Grant R, Walker M. Surgical resection and whole brain radiation therapy versus whole brain radiation therapy for solitary brain metastases. Cochrane Database of Systematic Reviews 2001, Issue 4. [DOI: 10.1002/14651858.CD003292] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hall EJ. Time, dose and fractionation in radiotherapy. In: John JR, Pamela Sutton P, Deirdre Marino D editor(s). Radiobiology for the radiologist. 5th Edition. Philadelphia: Lippincott Williams and Wilkins, 2000:412. [Google Scholar]
- MG Hart, R Grant, M Walker, H Dickinson. Surgical resection and whole brain radiation therapy versus whole brain radiation therapy alone for single brain metastases. Cochrane Database of Systematic Reviews 2004, Issue 4. [DOI: 10.1002/14651858.CD003292] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jadad AR, Moore RA, Carroll D. .Assessing the quality of reports of randomized clinical trials: is blinding necessary?. Controlled Clinical Trials 1996;17:1‐12. [DOI] [PubMed] [Google Scholar]
- Katz HR. The relative effectiveness of radiation therapy, corticosteroids and surgery in the management of melanoma metastatic to the central nervous system. International Journal of Radiation Oncology, Biology, Physics 1981;7:897‐906. [DOI] [PubMed] [Google Scholar]
- Loeffler JS, Patchell RA, Sawaya R. Treatment of metastatic cancer. In: Devita VT, Hellman S, Rosenberg SA editor(s). Cancer: Principles and Practice of Oncology. Fifth. Philadelphia: Lippincott‐Raven, 1997:2523. [Google Scholar]
- Noordijk EM, Vecht CJ, Haaxma‐Reiche H, Padberg GW, Voormolen JHC, Hoekstra FH, et al. The choice of treatment of single brain metastasis should be based on extracranial tumor activity and age. International Journal of Radiation Oncology, Biology, Physics 1994;29:711‐7. [DOI] [PubMed] [Google Scholar]
- Parmar MK, Torri V, Stewart L. Extracting summary statistics to perform meta‐analyses of the published literature for survival endpoints. Statistics in Medicine 1998;17:2815‐34. [DOI] [PubMed] [Google Scholar]
- Posner JB. Management of central nervous system metastases. Seminars in Oncology 1977;4:81‐91. [PubMed] [Google Scholar]
- Posner JB. Neurologic complications of cancer. Vol. 37, Philadelphia: Davis FA, 1995:311. [Google Scholar]
- Regine WF, Scott C, Murray K, Curran W. Neurocognitive outcome in brain metastases patients treated with accelerated‐fractionation vs accelerated‐hyperfractionated radiotherapy: an analysis from Radiation Therapy Oncology Group study 91‐04. International Journal of Radiation Oncology, Biology, Physics 2001;51(3):711‐7. [DOI] [PubMed] [Google Scholar]
- Regine WF, Huhn JL, Patchell RA, et al. Risk of symptomatic brain tumor recurrence and neurologic deficit after radiosurgery alone in patients with newly diagnosed brain metastases: results and implications. International Journal of Radiation Oncology, Biology, Physics 2002;52(2):333‐8. [DOI] [PubMed] [Google Scholar]
- Sneed PK, Larson DA, Wara WM. Radiotherapy for cerebral metastases. Neurosurgery Clinics of North America 1996;7:505‐16. [PubMed] [Google Scholar]
- Walker AE, Robins M, Weinfield FD. Epidemiology of brain tumors: The national survey of intracranial neoplasms. Neurology 1985;35:219‐26. [DOI] [PubMed] [Google Scholar]
- Zimm S, Wampler GL, Stablein D. Intracerebral metastases in solid tumor patients: Natural history and results of treatment. Cancer 1981;48:384‐94. [DOI] [PubMed] [Google Scholar]


