Abstract
Background
The benefits of adding upfront whole‐brain radiotherapy (WBRT) to surgery or stereotactic radiosurgery (SRS) when compared to surgery or SRS alone for treatment of brain metastases are unclear.
Objectives
To compare the efficacy and safety of surgery or SRS plus WBRT with that of surgery or SRS alone for treatment of brain metastases in patients with systemic cancer.
Search methods
We searched MEDLINE, EMBASE and The Cochrane Central Register of Controlled Trials (CENTRAL) up to May 2013 and annual meeting proceedings of ASCO and ASTRO up to September 2012 for relevant studies.
Selection criteria
Randomised controlled trials (RCTs) comparing surgery or SRS plus WBRT with surgery or SRS alone for treatment of brain metastases.
Data collection and analysis
Two review authors undertook the quality assessment and data extraction. The primary outcome was overall survival (OS). Secondary outcomes include progression free survival (PFS), local and distant intracranial disease progression, neurocognitive function (NF), health related quality of life (HRQL) and neurological adverse events. Hazard ratios (HR), risk ratio (RR), confidence intervals (CI), P‐values (P) were estimated with random effects models using Revman 5.1
Main results
We identified five RCTs including 663 patients with one to four brain metastases. The risk of bias associated with lack of blinding was high and impacted to a greater or lesser extent on the quality of evidence for all of the outcomes. Adding upfront WBRT decreased the relative risk of any intracranial disease progression at one year by 53% (RR 0.47, 95% CI 0.34 to 0.66, P value < 0.0001, I2 =34%, Chi2 P value = 0.21, low quality evidence) but there was no clear evidence of a difference in OS (HR 1.11, 95% CI 0.83 to 1.48, P value = 0.47, I2 = 52%, Chi2 P value = 0.08, low quality evidence) and PFS (HR 0.76, 95% CI 0.53 to 1.10, P value = 0.14, I2 = 16%, Chi2 P value = 0.28, low quality evidence). Subgroup analyses showed that the effects on overall survival were similar regardless of types of focal therapy used, number of brain metastases, dose and sequence of WBRT. The evaluation of the impact of upfront WBRT on NF, HRQL and neurological adverse events was limited by the unclear and high risk of reporting, performance and detection bias, and inconsistency in the instruments and methods used to measure and report results across studies.
Authors' conclusions
There is low quality evidence that adding upfront WBRT to surgery or SRS decreases any intracranial disease progression at one year. There was no clear evidence of an effect on overall and progression free survival. The impact of upfront WBRT on neurocognitive function, health related quality of life and neurological adverse events was undetermined due to the high risk of performance and detection bias, and inconsistency in the instruments and methods used to measure and report results across studies
Plain language summary
The benefits and side effects of adding upfront whole brain radiotherapy to surgery or radiosurgery for treatment of brain metastases
Background For certain patients with a single brain metastasis, surgery to remove the single brain metastasis may be undertaken. For certain patients with small brain metastatic disease, a highly focused single radiation treatment called radiosurgery, may be used. It is unclear the benefits and side effects of adding upfront (i.e. a therapy given to previously untreated patients) whole brain radiation to surgery or radiosurgery. We wanted to establish whether adding upfront whole brain radiation to surgery or radiosurgery was a useful treatment for brain metastases.
Study characteristics We included five studies. These studies reported the effect of adding whole brain radiation to surgery or radiosurgery in terms of survival, brain disease progression, quality of life and treatment side effects. The sample size of these studies ranged from 19 to 359 patients.
Key findings Adding whole brain radiotherapy to surgery or radiosurgery reduces brain metastases progression rates substantially but there was no clear evidence of an effect on survival and it is unclear whether it may cause side effects such as memory loss.
Quality of the evidence We considered the evidence on survival, intracranial disease progression, neurocognitive function, quality of life and treatment side effects to be of low quality.
Summary of findings
for the main comparison.
| Surgery or SRS plus WBRT compared with Surgery or SRS for brain metastases | |||||||||||
|
Patient or population: Patients with brain metastases Settings: Outpatient Intervention: Surgery or SRS plus WBRT Comparison: Surgery or SRS | |||||||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect (95% CI) | No of Participants (studies) | Quality assessment | Quality of the evidence (GRADE) | Comments | |||||
| Assumed risk | Corresponding risk | Risk of bias | Inconsistency | Indirectness | Imprecision | Other considerations | |||||
|
Overall Survival (median follow up: 7.8 months to 11.2 months) |
Median overall survival (months): 6.2 to 15.2 | Median overall survival (months): 5.7 to 11.2 | HR 1.11 (0.83 to 1.48) | 663 (5) | Serious | Not serious | Not serious | Serious | Publication bias strongly suspected; All plausible residual confounding would suggest spurious effect, while no effect was observed | Low | Two of the included studies were judged to be at high risk of bias i.e. Chang 2009 was judged to be at high risk of reporting bias while Roos 2006 was judged to be at high risk of attrition bias. |
| Progression free survival | Median progression free survival (months): 4.5 to 4.6 | Median progression free survival (months): 4.3 to 6.1 | HR 0.76 (0.53 to 1.10) | 378 (2) | Very serious | Not serious | Not serious | Serious | Publication bias strongly suspected; All plausible residual confounding would suggest spurious effect, while no effect was observed | Low | All studies were unblinded and this could impact on subjective outcomes such as disease progression. The criteria for disease progression was not well defined in the included trials. |
| Any intracranial disease progression at 1 year | 73‐76% | 22 ‐ 47% | RR 0.46 (0.35 to 0.60) | 304 (4) | Very serious | Not serious | Not serious | Not serious | Publication bias strongly suspected; All plausible residual confounding would suggest spurious effect, while no effect was observed | Low | All studies were unblinded and this could impact on subjective outcomes such as intracranial disease progression. The criteria for intracranial disease progression was not well defined in the included trials. |
| Neurocognitive function | Chang 2009 reported worse neurocognitive function with upfront WBRT but this study was at high risk for selection, performance, detection, attrition and reporting bias. Aoyama 2006 and Roos 2006 did not find any differences in neurocognitive function between the two treatment groups. | Indeterminate | 209 (3) | Very serious | Serious | Not serious | Serious | Publication bias strongly suspected; All plausible residual confounding would suggest spurious effect, while no effect was observed | Low | All studies were unblinded and this could impact on subjective outcomes such as neurocognitive function. We were unable to pool the results statistically due to differences among studies in the instruments and methods used to measure and report results. | |
| Health related quality of life | Kocher 2011 reported worse health related quality of life with upfront WBRT. Chang 2009 and Roos 2006 did not find any differences in health related quality of life between the two treatment groups. | Favours observation | 436 (3) | Very serious | Serious | Not serious | Serious | Publication bias strongly suspected; All plausible residual confounding would suggest spurious effect, while no effect was observed | Low | All studies were unblinded and this could impact on subjective outcomes such as health related quality of life. We were unable to pool the results statistically due to differences among studies in the instruments and methods used to measure and report results. | |
| Adverse events | Kocher 2011 and Roos 2006 reported more severe neurological adverse events with upfront WBRT. However Chang 2009 reported fewer neurological adverse events with upfront WBRT. Aoyama 2006 did not find any difference in the incidence of severe neurotoxicity events between the two treatment groups. | Favours observation | 568 (4) | Very serious | Serious | Not serious | Serious | Publication bias strongly suspected; All plausible residual confounding would suggest spurious effect, while no effect was observed | Low | All studies were unblinded and this could impact on subjective outcomes such as adverse events. We were unable to pool the results statistically due to differences among studies in the instruments and methods used to measure and report results. | |
| *The basis for the assumed risk (e.g. the median control group risk across studies) is provided in footnotes. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: Confidence interval; RR: Risk Ratio; HR: Hazard Ratio | |||||||||||
| GRADE Working Group grades of evidence High quality: Further research is very unlikely to change our confidence in the estimate of effect. Moderate quality: Further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate. Low quality: Further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate. Very low quality: We are very uncertain about the estimate. | |||||||||||
Background
Description of the condition
It is estimated that approximately 12.7 million cancer cases and 7.6 million cancer deaths occurred world‐wide in 2008 (Jemal 2011). Cancer can spread or metastasize to the brain from its main site, e.g. the lung or breast. Brain metastases are 10 times more common than tumours that originate in the brain (NCCN 2012) and approximately 170,000 new cases of brain metastases are diagnosed annually in the United States each year (Platta 2010). The incidence of brain metastases has increased two to five times over the last 40 years (Paterson 1982; Thomas 2000; Sundermeyer 2005; Smedby 2009). Possible reasons include better control of extra‐cranial disease with more effective systemic therapy and improved detection of small brain metastases with newer imaging modalities such as magnetic resonance imaging (MRI) (Paterson 1982; Mamon 2005; Sundermeyer 2005). It is estimated that 15% of patients who die from cancer have brain metastases detected at autopsy (Gavrilovic 2005).
Description of the intervention
When brain metastases occur, there is often also active disease at the primary site or elsewhere in the body. In few cases the brain is the only site with active disease. Local treatments such as surgery, radiotherapy (either stereotactic, directed only at the metastases; or directed to the whole brain) will only treat the brain disease. Surgery may be technically challenging in people where the metastases are close to important areas for speech and language or strength or sensation as surgery can also be associated with risks of bleeding or local damage. Treatment of brain metastases with surgery alone was also observed in randomized clinical trials (Patchell 1998; Roos 2006; Kocher 2011). Stereotactic radiosurgery (SRS) involves a single shot of radiotherapy and can treat metastases both close to the surface or deep in the brain. Stereotactic radiosurgery will only affect the targeted metastases and not the rest of the brain. Whole brain radiotherapy covers the whole brain and will treat the metastases and may also prevent the growth of new metastases.
The prognosis of patients with untreated brain metastases is poor, with a median survival of one month (Khuntia 2006). With the introduction of whole brain radiation therapy (WBRT) and corticosteroids in the mid 1900s the median survival time improved to four to six months (Al‐Shamy 2009).
Advances in imaging techniques over the last few decades, such as computer assisted image guided surgery and intra‐operative MRI, may have strengthened the role of surgery in the management of brain metastases (Vogelbaum 2006). Despite these advances, the survival benefits of surgery seem uncertain. A Cochrane review found that the combination of surgery and WBRT might improve functionally independent survival and reduce the risk of death due to neurological causes but not overall survival in patients with single brain metastasis when compared to WBRT alone (Hart 2011).
Stereotactic radiosurgery is a specialized radiation technique that delivers a single dose of highly collimated (i.e.to make rays parallel) radiation to a precisely defined target while minimizing the dose to surrounding areas (Mehta 2005). The advantages of SRS are its potential to treat lesions not amenable to surgical resection, minimal invasiveness and that it can be performed in an outpatient setting (Stafinski 2006). Examples of available photon‐based SRS delivery systems include the Gamma Knife, Linac and Cyberknife. The Gamma Knife is a dedicated cranial SRS unit through its geometric design, with 192 or 201 cobalt‐60 sources that are channeled through a tungsten collimator helmet, generating narrow radiation pencil beams and requires an invasive stereotactic head frame for localization and immobilisation. The accuracy of the treatment is within less than a millimetre and is ideal for small tumours (less than 3.5cm). The linac based SRS system use x‐rays generated by the linear accelerator system and micro‐multileaf collimators or circular collimators of various diameters (four to 50 millimetres) to shape the beam to the size of the tumour. It can be used to treat both small and large (> 3.5cm) tumours. The Cyberknife is a frame less, non‐invasive robotic radiosurgery technology which combines a compact six MV linear accelerator with a robotic arm of six degrees of freedom. It contains highly adjustable collimator cones ranging from five to 60 millimetres to deliver highly conformal treatment through several hundred narrow radiation beams from many different angles. It uses two orthogonal x‐ray cameras mounted to the ceiling to allow for real‐time tracking based upon bony landmarks or implanted fiducial (i.e. an object placed in the field of view of an imaging system for use as a point of reference). It can be used to treat lesions throughout the body, even in the difficult to reach or sensitive areas (McHaffie 2012).
A Cochrane review found that a combination of SRS with WBRT improved local brain control but not overall survival as compared with WBRT alone when all patients with brain metastases were included in the analysis (Patil 2010). However, subgroup analysis from one trial (Andrews 2004) indicated that survival was significantly longer in patients with single brain metastasis and in patients with a good prognosis (that is, under the age of 65 years, Karnofsky performance score 70 or higher, a controlled primary tumour, and no extra‐cranial metastases).
Tsao 2012 is an updated Cochrane review on whole brain radiotherapy for the treatment of newly diagnosed multiple brain metastases. In this updated review, Tsao and colleagues included three randomised controlled trials comparing SRS alone with SRS plus upfront WBRT. They concluded that the addition of upfront WBRT to radiosurgery improves local and distant brain control but there is no difference in overall survival. Patients treated with radiosurgery alone were found to have better neurocognitive outcomes in one trial as compared to patients treated with SRS and WBRT.
One of the key elements in deciding the appropriate treatment modalities for brain metastases is the patient's overall prognosis. The prognosis for survival depends on age; neurological fitness (performance score); extent of disease in other areas of the body; whether the disease is active at the primary site and the number of brain metastases. Two different prognostic scores, the Radiation Therapy Oncology Group (RTOG) recursive partitioning analysis (RPA) classification (Gaspar 1997) (Table 2) and Graded Prognostic Assessment (GPA) scores (Sperduto 2008) (Table 3; Table 4), are available to assist the clinician and patient decide on the relative risks and possible benefits of different treatments. According to the RPA classification, patients with the best prognosis (Class I) are less than 65 years old, have a Karnofsy performance score of 70 or higher and a controlled primary tumour without extra‐cranial metastases. The GPA system has demonstrated that more favourable prognosis is associated with fewer numbers of brain metastases (one versus two to three versus more than three). Wen 2011 suggested that more aggressive treatment should be considered in patients who have a more favourable prognosis with the goal of eradication or control of the brain metastases.
1. Recursive partitioning analysis classification.
| Class 1 | Class 2 | Class 3 | |
| Karnofsky Performance Status | ≥ 70 | ≥ 70 | < 70 |
| Age | < 65 | ≥ 65 | ≥ 65 |
| Systemic disease status | Controlled | Uncontrolled | Uncontrolled |
| Presence of extracranial metastasis | No | Yes | Yes |
| Median survival (months) | 7.1 | 4.2 | 2.3 |
2. Graded Prognostic Assessment scores.
| Scores | |||
| 0 | 0.5 | 1 | |
| Karnofsky Performance Status | < 70 | 70‐80 | 90‐100 |
| Age | > 60 | 50‐59 | < 50 |
| Presence of extracranial metastases | Yes | Not applicable | No |
| Number of central nervous system metastases | > 3 | 2‐3 | 1 |
3. Median survival for total Graded Prognostic Assessment scores.
| Total Score | Median Survival (months) |
| 0‐1 | 2.6 |
| 1.5‐2.5 | 3.8 |
| 3 | 6.9 |
| 3.5‐4 | 11 |
Why it is important to do this review
The role of WBRT additional to surgery or SRS versus surgery or SRS alone for one to three brain metastases in patients with stable systemic cancer remains unclear. The National Comprehensive Cancer Network (NCCN) guidelines suggest three different treatment options for one to three resectable brain metastases in patients with limited systemic disease (NCCN 2012). The options include 1) surgery followed by WBRT or SRS, 2) SRS plus WBRT or 3) SRS alone. For unresectable disease, WBRT or SRS or both can be used. There is also no clear consensus on the treatment recommendation for single brain metastasis from the American College of Radiology's Appropriateness Panel as there were potential concerns regarding the adverse effects of WBRT on neuro cognition (Suh 2010). A Cochrane review found that there was no good evidence from randomised controlled trials that surgery in addition to WBRT improves overall survival for patients with single brain metastasis compared with WBRT alone (Hart 2011).
The authors of Suh 2010a reported that the uncertainty in recommending the addition of WBRT to SRS in patients with a limited number of brain metastases is due to controversy over the relative effects of tumour progression and whole brain radiation on neurocognitive function. DeAngelis and colleagues reported that approximately 11% of patients who were treated with WBRT for brain metastases and had survived more than 12 months, developed severe dementia, especially with the use of larger dose per fraction schedules (DeAngelis 1989). Li and colleagues had observed in a randomised phase III trial that for patients who demonstrated good radiologic response to WBRT (30Gy in 10 fractions), they also had an improvement in executive function and fine motor coordination but not memory function, specifically recall and delayed recall as assessed with the Hopkins Verbal Learning Test (Li 2007). They had observed that memory function was most susceptible to early decline, even in patients with non‐progressing brain metastases, implying the selective effect of WBRT in preserving certain neurocognitive domains over others and the differential sensitivity of certain domains to radiation effects. Weizel and colleagues had also observed a decline in verbal memory function as assessed by the auditory verbal learning test at six to eight weeks after completion of WBRT for brain metastases (Welzel 2008). Hence, advocates for treatment with SRS alone believed that there was potential worsening of neuro cognition and no overall survival benefits associated with a combination of SRS and WBRT when compared to SRS alone. They preferred the option of salvage therapy with further SRS or subsequent WBRT when the disease progressed. However, advocates for combining SRS with WBRT argue that intra‐cranial disease progression is more frequent in the absence of WBRT and the recurrence of brain metastases can lead to more severe neurocognitive function deficits than those caused by WBRT.
Objectives
To compare the efficacy and safety of surgery or SRS plus WBRT with that of surgery or SRS alone for treatment of brain metastases in patients with systemic cancer.
Methods
Criteria for considering studies for this review
Types of studies
Randomized controlled trials (RCTs) meeting the eligibility criteria were included. Blinding and placebo control are difficult in trials of surgical interventions and were desirable but not mandatory requirements.
Types of participants
Patients (aged 18 years or older) with brain metastases (on imaging) and systemic cancer (primary site confirmed by histology). The brain metastases do not have to be histologically proven.
Types of interventions
Surgery: intended complete macroscopic resection of the brain metastases by a neurosurgical technique. Post‐operative MRI within 48 hours to define extent of resection would be desirable.
Stereotactic radiosurgery: delivery of highly collimated radiation to a precisely defined target i.e. brain metastases.
Whole brain radiation therapy (WBRT): delivery of radiation to the entire brain with various dose‐fractionation schedules such as 30Gy in 10 daily fractions or 20Gy in 4 or 5 daily fractions.
Intervention
Surgery or SRS plus WBRT
Comparison
Surgery or SRS alone
Types of outcome measures
Primary outcomes
(1) Overall survival (OS): survival until death from all causes. Survival was assessed from the time when patients were randomised.
Secondary outcomes
(2) Progression free survival (PFS): from time of randomizations to time of disease progression (an increase in the size of any lesion, development of new lesions or decline in performance status or worsening in symptoms) or death.
(3) Local intra‐cranial disease progression: from the time of randomizations to time of progression (i.e. enlarging brain metastases or development of new brain metastases on contrast enhanced computed tomography or magnetic resonance imaging) at the initial surgery or SRS treated site in the brain.
(4) Distant intra‐cranial disease progression: from time of randomizations to time of tumour development on contrast enhanced computed tomography or magnetic resonance imaging at new sites (not treated by surgery or SRS before) in the brain.
(5) Neurocognitive function (NF), measured using a scale that has been validated through reporting of norms in a peer‐reviewed publication. Examples of such scales are Hopkins Verbal Learning Test, Cogstate Test, International Shopping List Test and Mini Mental State Examination.
(6) Health related quality of life (HRQL), measured using a scale that has been validated through reporting of norms in a peer reviewed publication. An example of such a scale is the Functional Assessment of Cancer Therapy to Brain and European Organization for Research and Treatment of Cancer (EORTC) Quality of Life Questionnaire Brain Module.
(7) Neurological adverse events, nature, timing and management: defined as per trial protocol. We looked at the common neurological toxicities, namely nausea, vomiting, headache, fatigue and seizures and extracted only grades 3 or 4 toxicity data.
Search methods for identification of studies
Papers in all languages were sought and translations were carried out if necessary.
Electronic searches
Cochrane Central Register of Controlled Trials (CENTRAL) (The Cochrane Library), MEDLINE and EMBASE were searched up to May 2013.
The CENTRAL, MEDLINE and EMBASE search strategies are presented in Appendix 1, Appendix 2 and Appendix 3.
Searching other resources
Unpublished literature
We identified prospective and ongoing trials by searching the prospective trials registers: http://www.controlled‐trials.com (International Standard Randomized Controlled Trial Number Registry); http://www.clinicaltrials.gov (U.S. National Institutes of Health); http://www.cancer.gov/clinicaltrials/search (U.S. National Cancer Institute); http://www.who.int/trialsearch (World Health Organization International Clinical Trials Registry Platform) and http://www.anzctr.org.au (Australia and New Zealand Clinical Trials Registry).
Handsearching
The citation lists of included studies, key textbooks and previous systematic reviews were checked through hand searching. Reports of conferences were hand searched in the following sources:
Annual meeting of the American Society of Clinical Oncology;
Annual meeting of the American Society for Radiation Oncology.
Data collection and analysis
Selection of studies
All titles and abstracts retrieved by electronic searching were downloaded to a reference management database (that is Endnote), duplicates were removed and the remaining references were examined independently by four review authors (SYY, LKHC, ITWK, KWY). Those studies which did not meet the inclusion criteria were excluded and copies of the full texts of potentially relevant references were obtained. The eligibility of retrieved papers was assessed independently by four review authors (SYY, LKHC, ITWK, KWY). Disagreements were resolved by discussion between the four review authors and if necessary by a fifth review author (LJJ). Reasons for exclusion were documented.
Data extraction and management
For included studies, data on characteristics of patients (inclusion criteria, age, primary cancer histology, co‐morbidity, previous treatment, number enrolled in each arm) and interventions (types of focal therapy, that is surgery or SRS, dose of WBRT, SRS, sequence of focal therapy and WBRT), risk of bias, duration of follow‐up, outcomes and deviations from the protocol were extracted independently by four review authors (SYY, LKHC, ITWK, KWY) onto a data extraction form specially designed for the review.
For time to event (survival and disease progression) data, we extracted the log of the hazard ratio (log(HR)) and its standard error from trial reports; if these were not reported, we attempted to estimate the log (HR) and its standard error using published methods (Parmar 1998; Tierney 2007).
For dichotomous outcomes (for example deaths or cases of disease progression if it was not possible to use a hazard ratio) we extracted the number of patients in each treatment arm who experienced the outcome of interest and the number of patients assessed at the endpoint in order to estimate a risk ratio.
For continuous outcomes (for example quality of life measures) we extracted the final value and standard deviation of the outcome of interest and the number of patients assessed in each treatment arm at the endpoint at the end of follow‐up in order to estimate the mean difference between treatment arms and its standard error.
Both unadjusted and adjusted statistics were extracted, if reported. Where possible, all the data extracted were those relevant to an intention‐to‐treat analysis, in which participants were analysed in the groups to which they were assigned. The time points at which outcomes were collected and reported were noted.
Data were extracted independently by four review authors (SYY, LKHC, ITWK, KWY) onto a data extraction form specially designed for the review. Differences between review authors were resolved by discussion or by appeal to a fifth review author (LJJ) if necessary.
Assessment of risk of bias in included studies
Risk of bias in the included RCTs was assessed using the Cochrane Collaboration's risk of bias tool (Higgins 2011) and the following criteria.
Was the allocation sequence adequately generated?
Yes, e.g. participants assigned to treatments on the basis of a computer generated random sequence or a table of random numbers
No, e.g. participants assigned to treatments on the basis of date of birth, clinic identification number or surname, or no attempt to randomise participants
Unclear, e.g. not reported, information not available
Was allocation adequately concealed?
Yes, e.g. where the allocation sequence could not be foretold
No, e.g. allocation sequence could be foretold by patients, investigators or treatment providers
Unclear, e.g. not reported
Were outcome assessors adequately prevented from knowing the allocated interventions during the study?
Yes
No
Unclear
Was loss to follow‐up less than 20% and were the reasons for loss to follow‐up similar in both arms?
Yes
No
Unclear
Are reports of the study free of suggestion of selective outcome reporting?
Yes, e.g. if study reports all outcomes specified in the protocol
No
Unclear
Was the study apparently free of other problems that could put it at a high risk of bias?
Yes
No
Unclear
The risk of bias tool was applied independently by four review authors (SYY, LKHC, ITWK, KWY) and differences resolved by discussion or by appeal to a fifth review author (LJJ). Results were summarized in both a risk of bias graph and a risk of bias summary. Results of meta‐analyses were interpreted in the light of the findings with respect to risk of bias.
Measures of treatment effect
We used the following measures of the effect of treatment.
For time to event data (such as survival, progression free survival, intracranial disease progression), we used the hazard ratio if possible
For dichotomous outcomes (such as neurological adverse events), we used the risk ratio if possible.
For continuous outcomes (neurocognitive function, health related quality of life), we used the mean difference between treatment arms if possible.
Dealing with missing data
We did not impute missing outcome data for the primary outcome. When data were missing or only imputed data were reported, trial authors were contacted for data on the outcomes only among the participants who were assessed.
Assessment of heterogeneity
Heterogeneity between studies was assessed by visual inspection of forest plots, by estimation of the percentage heterogeneity between trials which cannot be ascribed to sampling variation (Higgins 2003), by a formal statistical test of the significance of the heterogeneity (Deeks 2001) and, by subgroup analyses (see below). When there was evidence of substantial heterogeneity, the possible reasons for this were investigated and reported.
Assessment of reporting biases
The funnel plot corresponding to the meta‐analysis of the primary outcome was examined to assess the potential for small study effects such as publication bias. When the plot suggested that treatment effects were not sampled from a symmetric distribution, as assumed by the random‐effects model, further meta‐analyses were performed using fixed‐effect models.
Data synthesis
When sufficient clinically similar studies were available, their results were pooled in meta‐analyses. Adjusted summary statistics were used if available; otherwise unadjusted results were used.
For time‐to‐event data, hazard ratios were pooled using the generic inverse variance facility of RevMan 5.
We pooled the hazard ratios only if the time to event data were analysed using the Kaplan Meier methods. The Kaplan‐Meier method and competing risk method are two different methods used in the estimation of cumulative incidence of an event of interest. The Kaplan‐Meier method does not account for any competing risk factors such as death but competing risk method does. This means that Kaplan‐Meier approach and the competing risk method will result in identical estimate of cumulative incidence of an event only when there are no competing risk. However when competing risks are present, the estimate obtained using the Kaplan‐Meier method is numerically larger than the competing risk method (Satagopan 2004).
For any dichotomous outcomes, the risk ratio was calculated for each study and was pooled when the outcomes were measured at the same time point with the same instruments.
For continuous outcomes, the mean differences between the treatment arms were pooled when all trials measured the outcome on the same scale and at the same time point.
Random‐effects models with inverse variance weighting was used for all meta‐analyses (DerSimonian 1986).
Subgroup analysis and investigation of heterogeneity
The following subgroup analyses, determined a priori were performed,
Types of focal interventions i.e. surgery versus SRS.
Number of brain metastases i.e. one brain metastasis only versus one or more brain metastases.
Sequence of WBRT i.e. before or after focal interventions.
Dose of WBRT in terms of Equivalent Dose in 2Gy Fractions with alpha/beta ratio of 2 (EQD22) i.e. EQD22>40Gy versus <40Gy
Sensitivity analysis
Sensitivity analyses was performed by excluding studies with moderate to high risk of bias.
Results
Description of studies
Results of the search
We identified five trials (Aoyama 2006; Chang 2009; Kocher 2011; Patchell 1998; Roos 2006) including 663 patients using the search strategy summarized in Figure 1
1.

Results of search strategy
Our initial search of MEDLINE, EMBASE and CENTRAL until June 2013 yielded 2454 references. We screened through the titles and/or abstracts of these references and excluded 2419 references as they did not meet our eligibility criteria. Of these, 655 references were not randomised trials, 1715 references did not include patients with brain metastases and 49 references did not have the appropriate trial design / comparisons. After removing the duplicates, we retrieved the remaining 10 references for detailed evaluation.
We excluded Chougule 2000 as it was published as an abstract form and did not provide sufficient information for qualitative and quantitative analysis. We excluded Fogarty 2011 as it is still an ongoing trial, ACTRN12607000512426 which we identified from the Australian New Zealand Clinical Trials Registry. We also identified another prospective trial, NCT00377156 from the US National Institute of Health Clinical Trials Registry.
The remaining eight references were reports of five randomised controlled trials (Aoyama 2006; Chang 2009; Kocher 2011; Patchell 1998; Roos 2006).
Included studies
All included five trials (Aoyama 2006; Chang 2009; Kocher 2011; Patchell 1998; Roos 2006) were published as full journal articles. The median sample size was 95 patients, ranging from 19 to 359 across all five trials. The mean age of patients ranged from 58 to 63 years old. The number of brain metastases allowed in these trials were one to four. The percentage of patients with primary breast or lung cancers ranged from 47 to 69. The percentage of patients with RPA Class I were 14 to 21 across three trials (Aoyama 2006; Chang 2009; Roos 2006). The remaining two trials (Patchell 1998; Kocher 2011) did not report the percentage of patients with RPA Class I.
Across all five trials, two trials employed SRS only (Aoyama 2006; Chang 2009), two trials employed either SRS or Surgery (Kocher 2011; Roos 2006) and one trial used Surgery alone as focal intervention (Patchell 1998). The median WBRT EQD22 was 37.5Gy, ranging from 33.75Gy to 47.88Gy across all five trials.
We extracted time to event data for OS from all five trials and PFS from two trials (Kocher 2011; Roos 2006). Intracranial disease progression was reported inconsistently across all five trials. Neurocognitive function was reported in three trials (Aoyama 2006; Chang 2009; Roos 2006). Health related quality of life was reported in three trials (Chang 2009; Kocher 2011; Roos 2006). Neurological adverse events were reported in four trials (Aoyama 2006; Chang 2009; Kocher 2011; Roos 2006).
Excluded studies
Chougule 2000 was excluded as it was published as an abstract and did not provide sufficient information for analysis. Fogarty 2011 was excluded as it was published as a study protocol and is an ongoing trial.
Risk of bias in included studies
The risk of bias in included studies was summarized in Figure 2 and Figure 3
2.

Risk of bias graph: review authors' judgements about each risk of bias item presented as percentages across all included studies.
3.

Risk of bias summary: review authors' judgements about each risk of bias item for each included study.
Allocation
The risk of bias in random sequence generation is low in all trials (Aoyama 2006; Chang 2009; Kocher 2011; Patchell 1998; Roos 2006). Aoyama 2006 and Chang 2009 used permutated block randomizations algorithm while Kocher 2011 used the minimization algorithm to generate the sequence. Patchell 1998 and Roos 2006 used computer generated random numbers. There risk of bias for allocation concealment is low in four trials (Aoyama 2006; Chang 2009; Roos 2006; Patchell 1998), unclear in one trials (Kocher 2011). Aoyama 2006, Roos 2006 and Patchell 1998 performed its allocation at a central site. Kocher 2011 did not describe its method of allocation concealment.
Blinding
Blinding of participants and personnel and outcome assessment was not reported in any of the five trials. The risk of bias from lack of blinding was high for subjective outcomes such as progression free survival, intracranial disease progression, neurocognitive function, health related quality of life and neurological adverse events and low for objective outcomes such as overall survival.
Incomplete outcome data
The risk of bias in incomplete outcome data is low for three trials (Aoyama 2006; Kocher 2011; Patchell 1998) and high in two trials (Chang 2009; Roos 2006). Chang 2009 included 53% of the patients (31 out of 58) for analysis of neurocognitive function outcome. Roos 2006 closed prematurely due to poor accrual. All trials followed intention to treat principle in the analysis of survival results.
Selective reporting
The risk of bias in selective reporting is low in all trials except Chang 2009. As mentioned above, it is likely that Chang 2009 is at high risk of reporting bias as patients in the observation arm survived significantly longer compared with WBRT arm.
Other potential sources of bias
We did not find any other potential sources of bias in the included study. Although a funnel plot was initially planned to examine possible publication bias, but due to the small number of included studies, funnel plot analysis was deemed not useful and hence not conducted.
Effects of interventions
See: Table 1
Overall survival
There was no clear evidence of a difference in overall survival with additional upfront WBRT when compared with observation (HR 1.11; 95%CI 0.83 to 1.48, P value = 0.47(Analysis 1.1)). There was significant heterogeneity in the hazard ratios for OS from the individual trials (P value = 0.08, I2 = 52%). However there is no significant differences in effects on OS between subgroups defined by types of focal interventions (Analysis 2.1) , number of brain metastases (Analysis 3.1), sequence of WBRT (Analysis 4.1) and EQD22 dose of WBRT (Analysis 5.1).
1.1. Analysis.
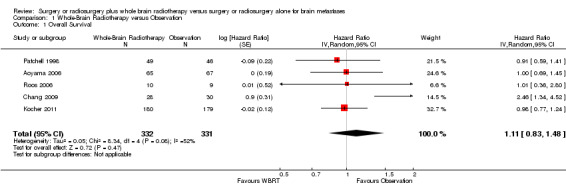
Comparison 1 Whole‐Brain Radiotherapy versus Observation, Outcome 1 Overall Survival.
2.1. Analysis.

Comparison 2 Whole‐Brain Radiotherapy versus Observation (subgroup analysis: Focal intervention), Outcome 1 Overall Survival.
3.1. Analysis.

Comparison 3 Whole‐Brain Radiotherapy versus Observation (subgroup analysis: Number of brain metastases), Outcome 1 Overall Survival.
4.1. Analysis.

Comparison 4 Whole‐Brain Radiotherapy versus Observation (subgroup analysis: Sequence of WBRT), Outcome 1 Overall Survival.
5.1. Analysis.

Comparison 5 Whole‐Brain Radiotherapy versus Observation (subgroup analysis: Equivalent Dose in 2Gy Fractions with α/β ratio of 2 (EQD22)), Outcome 1 Overall Survival.
Progression free survival
There was no clear evidence of a difference in progression free survival with additional upfront WBRT when compared with observation (HR 0.76; 95%CI 0.53 to 1.10, P value = 0.14 (Analysis 1.2)). There was no significant heterogeneity in the hazard ratios for PFS from the individual trials (P value = 0.28, I2=16%). There were no significant differences in effects on PFS between subgroups defined by number of brain metastases (Analysis 3.2).
1.2. Analysis.
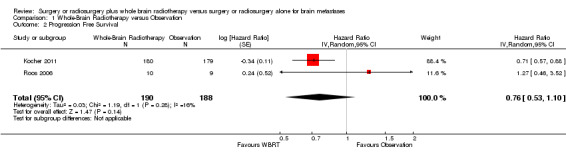
Comparison 1 Whole‐Brain Radiotherapy versus Observation, Outcome 2 Progression Free Survival.
3.2. Analysis.

Comparison 3 Whole‐Brain Radiotherapy versus Observation (subgroup analysis: Number of brain metastases), Outcome 2 Progression Free Survival.
Intracranial disease progression
We were unable to pool the intracranial disease progression results statistically using time to events analysis due to the differences among studies in the methods of analysis. Patchell 1998 and Roos 2006 analysed the results using the Kaplan‐Meier methods, while Kocher 2011 used the competing risk method to analyse the cumulative incidence. Both Aoyama 2006 and Chang 2009 analysed the results of local intracranial disease progression with Kaplan‐Meier method but used the competing risk method to analyse the distant intracranial disease progression results.
We instead pooled the risk of intracranial disease progression at one year as risk ratio. Adding upfront WBRT significantly reduced the risk of any intracranial disease progression at one year by 53% (RR 0.47; 95%CI 0.34 to 0.66, P value < 0.0001 Analysis 1.3), risk of local intracranial disease progression at one year by 62% (RR 0.38; 95%CI 0.24 to 0.60, P value < 0.0001 Analysis 1.4) and risk of distant intracranial disease progression at one year by 44% (RR 0.56; 95%CI 0.44 to 0.71, P value < 0.00001 Analysis 1.5). There was no significant heterogeneity in the risk ratios for intracranial disease progression at one year from the individual trials (P value = 0.21, I2 = 34% for any intracranial disease progression, P value = 0.14, I2 = 45% for local intracranial disease progression and P value = 0.31, I2 = 17% for distant intracranial disease). Although one study that used EQD22 of more than 40Gy for WBRT showed an apparently greater reduction in the risk of any or distant intracranial disease progression at one year, the strength of this subgroup effect was weak (P = 0.08) (Analysis 5.2; Analysis 5.4). There were no significant differences in effects on intracranial disease progression at one year between subgroups defined by types of focal intervention (Analysis 2.2; Analysis 2.3; Analysis 2.4) , number of brain metastases (Analysis 3.3; Analysis 3.4; Analysis 3.5) and sequence of WBRT (Analysis 4.2; Analysis 4.3; Analysis 4.4).
1.3. Analysis.

Comparison 1 Whole‐Brain Radiotherapy versus Observation, Outcome 3 Any intracranial disease progression at one year.
1.4. Analysis.

Comparison 1 Whole‐Brain Radiotherapy versus Observation, Outcome 4 Local intracranial disease progression at one year.
1.5. Analysis.

Comparison 1 Whole‐Brain Radiotherapy versus Observation, Outcome 5 Distant intracranial disease progression at one year.
5.2. Analysis.
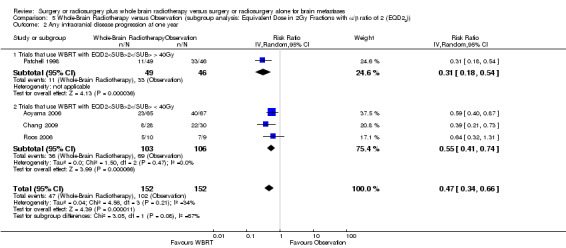
Comparison 5 Whole‐Brain Radiotherapy versus Observation (subgroup analysis: Equivalent Dose in 2Gy Fractions with α/β ratio of 2 (EQD22)), Outcome 2 Any intracranial disease progression at one year.
5.4. Analysis.
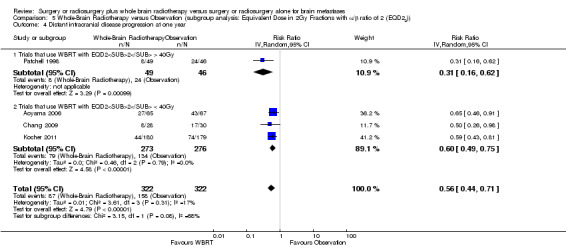
Comparison 5 Whole‐Brain Radiotherapy versus Observation (subgroup analysis: Equivalent Dose in 2Gy Fractions with α/β ratio of 2 (EQD22)), Outcome 4 Distant intracranial disease progression at one year.
2.2. Analysis.

Comparison 2 Whole‐Brain Radiotherapy versus Observation (subgroup analysis: Focal intervention), Outcome 2 Any intracranial disease progression at one year.
2.3. Analysis.

Comparison 2 Whole‐Brain Radiotherapy versus Observation (subgroup analysis: Focal intervention), Outcome 3 Local intracranial disease progression at one year.
2.4. Analysis.

Comparison 2 Whole‐Brain Radiotherapy versus Observation (subgroup analysis: Focal intervention), Outcome 4 Distant intracranial disease progression at one year.
3.3. Analysis.

Comparison 3 Whole‐Brain Radiotherapy versus Observation (subgroup analysis: Number of brain metastases), Outcome 3 Any intracranial disease progression at one year.
3.4. Analysis.
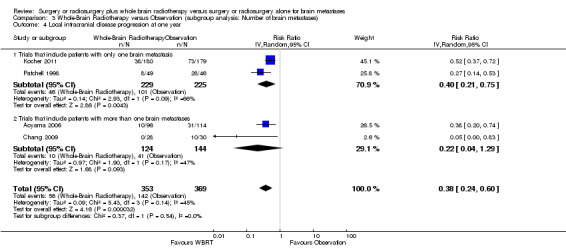
Comparison 3 Whole‐Brain Radiotherapy versus Observation (subgroup analysis: Number of brain metastases), Outcome 4 Local intracranial disease progression at one year.
3.5. Analysis.

Comparison 3 Whole‐Brain Radiotherapy versus Observation (subgroup analysis: Number of brain metastases), Outcome 5 Distant intracranial disease progression at one year.
4.2. Analysis.

Comparison 4 Whole‐Brain Radiotherapy versus Observation (subgroup analysis: Sequence of WBRT), Outcome 2 Any intracranial disease progression at one year.
4.3. Analysis.

Comparison 4 Whole‐Brain Radiotherapy versus Observation (subgroup analysis: Sequence of WBRT), Outcome 3 Local intracranial disease progression at one year.
4.4. Analysis.
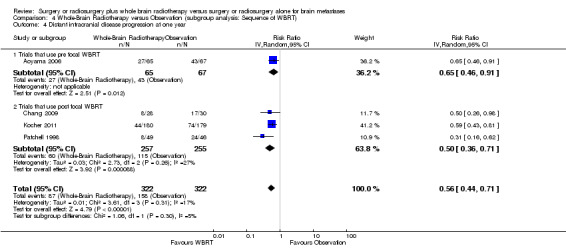
Comparison 4 Whole‐Brain Radiotherapy versus Observation (subgroup analysis: Sequence of WBRT), Outcome 4 Distant intracranial disease progression at one year.
Neurocognitive function
The impact of upfront WBRT on neurocognitive function was unclear. We were unable to pool the NF results statistically due to the differences among studies in the instruments and methods used to measure and report NF results (Table 5). Aoyama 2007 (Aoyama 2006 trial ) and Roos 2006 used mini‐mental state examination (MMSE) while Chang 2009 used Hopkins Verbal Learning Test‐Revised (HVLT‐R), Wechsler Adult Intelligence Scale‐III digit span and digit symbol, Trail Making Test parts A and B, Multilingual Aphasia Examination Controlled Oral Word Association and Lafayette Grooved Pegboard.
4. Neurocognitive function.
| Study | Instrument | Rating |
| Patchell 1998 | Not reported | Not applicable |
| Aoyama 2006 | MMSE | Favor neither |
| Chang 2009 | HVLT‐R | Favor Observation |
| Kocher 2011 | Not reported | Not applicable |
| Roos 2006 | MMSE | Favor neither |
Aoyama 2007 measured NF at baseline, one month, 3 months after treatment and if possible every 3 months thereafter. Aoyama 2007 reported that improvements of >=3 points in the MMSE were similar between two groups (P = 0.85). For patients who started with at MMSE of >= 27, or achieved this after initial brain treatment, 76% of cases who had upfront WBRT still had not dropped 3 points in MMSE by year 1, 69% by year 2 and 15% by 3 years. This compares with 59%, 52% and 52% respectively in patients without upfront WBRT (P = 0.73). The average duration until deterioration was 16.5 months in the upfront WBRT group and 7.6 months in the observation group (P = 0.05). Roos 2006 measured the NF with MMSE annually after treatment and found that there was no difference in MMSE between the groups (P = 0.50). Chang 2009 planned to measure the NF at 1,2,4,6,9,12,15,18 months and then every six months thereafter following completion of the last radiation treatment. A decline for total recall score (based on HVLT‐R) was defined as a drop in five points or greater compared with baseline. However the trial was halted early as there was 96% confidence that total recall at 4 months for upfront WBRT group was inferior to total recall for observation based on a mean posterior probability of decline of 52% for the upfront WBRT group and 24% for the observation group. The total recall difference persisted at six months, with a mean posterior probability of decline of 28% for the upfront WBRT group and 8% for the observation group with 90% confidence. The Bayesian posterior probability that proportion with a significant neurocognitive worsening is higher in upfront WBRT group at 4 months ranged from 38% to 86% for the remaining neurocognitive tests. However the authors did caution that the analysis for other neurocognitive function tests besides the HLVT‐R may be underpowered.
Health related quality of life
The impact of upfront WBRT on HRQL was unclear. We were unable to pool the HRQL results statistically due to the differences among studies in the instruments and methods used to measure and report HRQL results (Table 6). Chang 2009 used the Functional Assessment of Cancer Therapy ‐ Brain (FACT‐BR) while Soffietti 2013 (Kocher 2011 trial) and Roos 2006 used the EORTC Quality of life‐C30 and BN 20 to measure HRQL.
5. Health related quality of life.
| Study | Instrument | Rating |
| Patchell 1998 | Not reported | Not applicable |
| Aoyama 2006 | Not reported | Not applicable |
| Chang 2009 | FACT‐BR | Favor neither |
| Kocher 2011 | EORTC Quality of life‐ C30 and BN 20 | Favor Observation |
| Roos 2006 | EORTC Quality of life‐ C30 and BN 20 | Favor neither |
Chang 2009 planned to measure HRQL at 1,2,4,6,9,12,15,18 months and then every six months thereafter following completion of the last radiation treatment but the trial was halted early according to the early stopping rules stated above. The FACT BR mean difference between the groups at 4 months compared with baseline was 2.8 (95% CI ‐26 to 21, P value = 0.76). Chang 2009 wrote that these results were inconclusive and should not be interpreted as indicating no difference between the two groups. Soffietti 2013 measured HRQL at baseline, at 8 weeks (between 2nd and 6 weeks after start of focal treatment) and every 3 months for 3 years after 8 weeks assessment. Soffietti 2013 reported that patients in the observation arm have statistically significant higher mean global HRQL scores compared with patients with WBRT at 9 months (63.2 versus 52.2, P value = 0.01). Roos 2006 measured HRQL at two, five and six months following completion of the last radiation treatment and found no significant differences in the global HRQL between the two groups at two and five months. Roos 2006 also reported that they were unable to conduct detailed HRQL analysis due to small sample size and there were too few surviving patients for meaningful statistical comparisons after five months.
Neurological adverse events
The impact of upfront WBRT on neurological adverse events was unclear. We were unable to pool the neurotoxicity results statistically due to the differences among studies in the instruments and methods used to measure and report neurotoxicity results (Table 7). Aoyama 2006 used the National Cancer Institute's Common Toxicity Criteria version 2.0 (NCI CTC v2.0) and found no significant differences in severe neurotoxicities between the two groups. There were 5 cases of grade 3 or 4 neurotoxicity (i.e. one case of grade 3 seizure, two cases of grade 3 leukoencephalopathy and two cases of grade 4 radiation necrosis) in the upfront WBRT group compared with 4 cases (i.e. one case of grade 3 seizure, one case of grade 3 neurotoxicity (details not given), one case of grade 4 seizure and one case of grade 4 radiation necrosis) in the observation group. Chang 2009 did not report which instrument was used to grade neurotoxicity and found that there were slightly more neurotoxic events in the observation group. There was only one case of grade 3 seizure in the upfront WBRT group and three cases grade 3 or 4 neurotoxicity namely one case of grade 3 aphasia, two cases of grade 4 radiation necrosis in the observation group. Kocher 2011 reported the use of the Late Effects of Normal Tissues ‐ Subjective, Objective, Management, Analytic brain toxicity scales for grading of late neurotoxicity effects but not for acute neurotoxicity. Kocher 2011 wrote that the acute toxicity of upfront WBRT was generally mild with one percent of the patients having severe vomiting and four percent of patients having severe headache. There were no significant differences in the severe late neurotoxicity between the two groups. There were 14 patients with grade 3 seizures, 22 patients with grade 4 seizures, 7 patients with grade 3 headache and 2 patients with grade 4 headache in the upfront WBRT group and10 patients with grade 3 seizures, 23 patients with grade 4 seizures, 10 patients with grade 3 headache and 1 patient with grade 4 headache in the observation group. Roos 2006 used the National Cancer Institute (NCI) acute radiation toxicity score and reported two cases of neurotoxicity in the upfront WBRT group and none in the observation group.
6. Neurological Adverse Events.
| Study | Instrument | Rating |
| Patchell 1998 | Not reported | Not applicable |
| Aoyama 2006 | NCI CTC v2.0 | Favor neither |
| Chang 2009 | Not reported | Favor WBRT |
| Kocher 2011 | LENT‐SOMA | Favor Observation |
| Roos 2006 | NCI Acute Radiation Toxicity Scores | Favor Observation |
Discussion
Summary of main results
This meta‐analysis showed that adding upfront WBRT to surgery or SRS delayed any intracranial disease progression at one year but did not improve overall or progression free survival. The impact of upfront WBRT on neurocognitive function, health related quality of life and neurological adverse events remained undetermined.
Overall completeness and applicability of evidence
We felt that the included studies provided insufficient evidence for us to draw reliable conclusions on the effects of upfront WBRT on overall survival, progression free survival, intracranial disease progression, neurocognitive function, health related quality of life and neurological adverse events outcomes.
Although two trials (Aoyama 2006; Roos 2006) showed that use of upfront WBRT does not worsen neurocognitive function, it is important to note that these two trials used MMSE to measure neurocognitive outcomes and brief mental status evaluations such as MMSE has been found to have extremely poor sensitivity in detecting neurocognitive problems in brain tumour patients (Meyers 2003). Among the included trials, only one trial (Chang 2009) used a battery of validated neurocognitive tests to measure various domains of neuro cognition. This trial was also designed with a primary endpoint of neurocognitive function, as objectively measured by significant deterioration in HVLT‐R total recall at 4 months unlike Aoyama 2006 and Li 2007 who measured neurocognitive outcomes at multiple time points. It is unfortunate that the trial was halted early and failed to meet its accrual target. This trial was also at high risk of unintentional selection, performance, detection and reporting bias as the patients in the observation arm was significantly longer survival than the WBRT arm and this was not observed in other studies.
It remains to be determined what is the optimal method of analysing neurocognitive tests results i.e. whether we should use bayesian statistical methods, use a fixed drop from baseline scores or presenting results at a fixed time. We agree with Meyers and colleagues (Meyers 2006) that it is crucial for future clinical trials to continue to use validated instruments such as HVLT, controlled oral word association and trail making tests to detect meaningful cognitive effects of various treatment modalities i.e. surgery, radiotherapy or drugs. Future clinical trials should also design to measure neurocognitive function at multiple time points and include sufficient sample size for reliable statistical analysis of the results.
The use of various salvage treatments for brain metastases may confound the assessment of intracranial disease progression and overall survival when the rates of use differ between treatment groups. The criteria for various salvage treatments were not reported in all trials. Only three trials reported the utilization rate of salvage treatment (Aoyama 2006; Chang 2009; Kocher 2011). All three trials consistently showed that more patients in the observation group received salvage treatment compared to upfront WBRT group. Aoyama 2006 reported that 16% of patients received salvage WBRT, 28% of patients received salvage SRS in the observation group while 14% of patients received salvage SRS in the upfront WBRT group. Chang 2009 reported that 33% of the patients received salvage WBRT, 20% of patients received salvage SRS, 33% of patients received salvage surgery in the observation group while 7% of the patients received salvage SRS in the upfront WBRT group. Kocher 2011 reported that 31% of patients received salvage WBRT, 12% of patients received salvage SRS, 6% of patients received salvage surgery while 3% of patients received salvage WBRT, 11% of patients received salvage SRS and 2% of patients received salvage surgery in the upfront WBRT group. Hence patients who did not receive upfront WBRT require close surveillance and have a higher rate of salvage therapy.
These results are applicable to routine practice of delivering WBRT based on standard whole brain fields and opposed lateral techniques for treatment of one to four brain metastases but not necessarily applicable to treatment of more than four brain metastases, or brain metastases secondary to small cell lung carcinoma, lymphoma, leukaemia, multiple myeloma and germ cell tumours or with intensity‐modulation radiotherapy techniques to spare the critical brain structures such as hippocampus or use of concurrent radiation sensitizer or neuro protectors.
Quality of the evidence
All five trials provide low quality evidence for overall survival, progression free survival, intracranial disease progression, neurocognitive function, health related quality of life and neurological adverse events outcomes. We determined the quality of evidence using the guideline development tool developed by the GRADE Working Group (GRADE Working Group 2012) with the following scoring criteria: Risk of bias ‐ Serious for overall survival and Very serious for all other outcomes; Inconsistency ‐ Not serious for overall survival, progression free survival and intracranial disease progression, and Serious for all other outcomes; Indirectness ‐ Not serious for all outcomes; Imprecison ‐ Serious for all outcomes except for intracranial disease progression at one year; Publication bias ‐ Strongly suspected for all outcomes; Plausible confounding ‐ Would suggest spurious effect for all outcomes; Dose‐response gradient ‐ No for all outcomes.
Potential biases in the review process
The strengths of this review are that it addresses an important pragmatic question, includes trials such as Roos 2006 and Kocher 2011 which were not discussed in recent reviews (Brown 2008; Kalkanis 2010; Linskey 2010; Platta 2010; Tsao 2012a; Tsao 2012) and shows consistent results in sensitivity analyses.
A limitation of this review is the use of published results rather than individually updated patient data. Although these results may overestimate the benefits of additional upfront WBRT (Stewart 1993), it is unlikely that an individual patient data‐based meta‐analysis will changes the conclusions of this review. This is because the effects of additional upfront WBRT on intracranial disease progression are still likely to be significant while the effects on OS are likely to remain non‐significant.
Another limitation is the inability to summarize the data on intracranial disease progression using time to event analysis methods due to the differences among studies in the methods of analysis. As mentioned earlier, the two methods of analysis used by the studies in the estimation of cumulative incidence of events were Kaplan‐Meier and the competing risk method.The Kaplan‐Meier method does not account for any competing risk factors such as death but competing risk method does. This means that the Kaplan‐Meier approach and the competing risk method will result in an identical estimate of cumulative incidence of an event only when there are no competing risks. However when competing risks are present, the estimate obtained using the Kaplan‐Meier method is numerically larger than the competing risk method (Satagopan 2004). Hence, we could only summarize the intracranial disease progression data as a dichotomous outcome i.e. risk of intracranial disease progression at one year.
The other limitation is heterogeneity in the reporting of neurocognitive function, health related quality of life and neurological adverse events. Due to the heterogeneity, we were unable to perform quantitative analysis on these outcomes. The qualitative evaluation of these subjective outcomes was also limited by the potential performance and detection bias as blinding of participants, personnel and outcome assessment was not performed.
Agreements and disagreements with other studies or reviews
These results are fairly consistent with other reviews (Brown 2008; Platta 2010; Tsao 2012; Tsao 2012a) and practice guidelines from NCCN (NCCN 2012), American Association of Neurological Surgeons and Congress of Neurological Surgeons (Kalkanis 2010; Linskey 2010) and American Society for Radiation Oncology (Tsao 2012b).
Brown 2008 and Platta 2010 are qualitative reviews which discussed the benefits and sides effects of additional upfront WBRT to surgery or SRS for treatment of brain metastases. Both reviews found that additional upfront WBRT provide excellent intracranial disease control with no improvement in overall survival and associated with uncertain effects on neurocognitive function and quality of life. Brown 2008 recommended that adjuvant WBRT should be considered as standard following surgical resection of a single brain metastasis based on Patchell 1998 and Roos 2006 trials. However Brown 2008 was unable to recommend adjuvant WBRT to patients who had stereotactic radiosurgery because of concern about neurocognitive impairment. Platta 2010 did not provide any treatment recommendations.
Tsao 2012a is a systematic review and meta‐analysis attempting to address two clinical questions in patients with limited brain metastases: 1. What are the effects of additional upfront WBRT to SRS versus SRS alone and 2. What are the effects of additional upfront SRS to WBRT versus WBRT alone. This systematic review and meta‐analysis was published while we were preparing the manuscript for this review. The results of this meta‐analysis is consistent and complementary with our results. Tsao 2012a found that additional upfront WBRT improves both local and distant intracranial disease control with no overall survival benefit and reports worse neuro cognition with Hopkins Verbal Learning Test (test of memory). Tsao 2012a concluded that SRS alone should be considered as a routine treatment option due to favourable neuro cognition outcomes, less risk of late side effects and does not adversely affect patients performance status.
NCCN (NCCN 2012) recommended three treatment options for treatment of one to three resectable brain metastases for patients who had stable systemic disease. The options include 1) surgery followed by WBRT or SRS, 2) SRS plus WBRT or 3) SRS alone. NCCN also gave a category one recommendation to two options (I) surgery followed WBRT for one to three brain metastases (ii) SRS plus WBRT for single brain metastasis. The American Association of Neurological Surgeons and Congress of Neurological Surgeons gave a level one recommendation for use of surgical resection followed by WBRT (Kalkanis 2010) and level two recommendation for use of stereotactic radiosurgery alone (Linskey 2010) for treatment of patients with single brain metastasis. The American Society for Radiation Oncology (ASTRO) (Tsao 2012b) made its treatment recommendations based on the number of brain metastasis and estimated prognosis. For patients with single brain metastasis and good prognosis (expected survival 3 months or more), there are level one evidence supporting the use of WBRT and surgery, WBRT and SRS and SRS alone. For patients with more than one brain metastases and good prognosis, there are level one evidence supporting the use of WBRT and SRS, SRS alone and WBRT alone.
Although this review overlaps with Tsao 2012 and Tsao 2012a, there are a few differences. Firstly, the focus of this review is whether to add a diffuse therapy early after a focal therapy and we have considered both surgery and SRS to be valid focal therapy as there is no evidence to suggest either focal therapy is better than the other, unlike Tsao and colleagues who looked at patients receiving only SRS as focal therapy. Secondly, this review offers a more detailed discussion of neurocognitive functioning, health related quality of life and neurological adverse events.
Authors' conclusions
Implications for practice.
There is low quality evidence that adding upfront WBRT to surgery or SRS decreases any intracranial disease progression at one year. There was no clear evidence of an effect on overall and progression free survival. The impact of upfront WBRT on neurocognitive function, health related quality of life and neurological adverse events was undetermined due to the high risk of performance and detection bias, and inconsistency in the instruments and methods used to measure and report results across studies. Patients who do not have WBRT after Surgery or SRS require careful surveillance and have a high rate of salvage therapy which may include further SRS or WBRT.
Implications for research.
Properly blinded randomised trials assessing of the impact of additional upfront WBRT to surgery or SRS on neurocognitive function, health related quality of life and neurological adverse events are warranted. Future research should also focus on improving the precision of radiation delivery to avoid critical structures such as hippocampus or developing effective chemical modifiers of radiation i.e. radioprotectors to ameliorate the neurotoxicity of WBRT and improve quality of life. To assess neurocognitive function accurately, a number of tools need to be used in conjunction with one another. A single tool for neuro cognition assessment that is sensitive and easy to perform is needed.
What's new
| Date | Event | Description |
|---|---|---|
| 21 September 2016 | Amended | Contact details updated. |
History
Protocol first published: Issue 11, 2011 Review first published: Issue 3, 2014
| Date | Event | Description |
|---|---|---|
| 27 March 2014 | Amended | Contact details updated. |
Acknowledgements
We thank Robin Grant and Mike Hart for their clinical expertise. We thank Jane Hayes for designing the search strategy and Gail Quinn for her contribution to the editorial process.
The National Institute for Health Research (NIHR) is the largest single funder of the Cochrane Gynaecological Cancer Group.
The views and opinions expressed therein are those of the authors and do not necessarily reflect those of the NIHR, NHS or the Department of Health.
Appendices
Appendix 1. MEDLINE search strategy
1 exp Brain Neoplasms/ 2 ((brain or cerebral or intracranial or intracerebral) adj5 (metasta* or cancer* or tumor* or tumour* or neoplas* or carcinoma* or malignan*)).mp. 3 1 or 2 4 exp Radiotherapy/ 5 radiotherapy.fs. 6 radiotherap*.mp. 7 radiation.mp. 8 irradiation.mp. 9 WBRT.mp. 10 4 or 5 or 6 or 7 or 8 or 9 11 exp Neurosurgical Procedures/ 12 surgery.fs. 13 neurosurg*.mp. 14 surg*.mp. 15 Radiosurgery/ 16 radiosurg*.mp. 17 resect*.mp. 18 11 or 12 or 13 or 14 or 15 or 16 or 17 19 3 and 10 and 18 20 randomized controlled trial.pt. 21 controlled clinical trial.pt. 22 randomized.ab. 23 placebo.ab. 24 clinical trials as topic.sh. 25 randomly.ab. 26 trial.ti. 27 20 or 21 or 22 or 23 or 24 or 25 or 26 28 19 and 27
key:
mp=protocol supplementary concept, rare disease supplementary concept, title, original title, abstract, name of substance word, subject heading word, unique identifier fs=floating subheading pt=publication type ab=abstract ti=title
Appendix 2. EMBASE search strategy
1 exp brain tumor/ 2 ((brain* or cerebral or intracranial or intra‐cranial or intracerebral or intra‐cerebral) adj5 (metasta* or cancer* or tumor* or tumour* or neoplas* or carcinoma* or malignan*)).mp. 3 1 or 2 4 exp radiotherapy/ 5 rt.fs. 6 radiotherap*.mp. 7 radiation.mp. 8 irradiation.mp. 9 WBRT.mp. 10 4 or 5 or 6 or 7 or 8 or 9 11 exp neurosurgery/ 12 su.fs. 13 neurosurg*.mp. 14 exp radiosurgery/ 15 radiosurg*.mp. 16 resect*.mp. 17 11 or 12 or 13 or 14 or 15 or 16 18 3 and 10 and 17 19 crossover procedure/ 20 double‐blind procedure/ 21 randomized controlled trial/ 22 single‐blind procedure/ 23 random*.mp. 24 factorial*.mp. 25 (crossover* or cross over* or cross‐over*).mp. [mp=title, abstract, subject headings, heading word, drug trade name, original title, device manufacturer, drug manufacturer, device trade name, keyword] 26 placebo*.mp. 27 (double* adj blind*).mp. 28 (singl* adj blind*).mp. 29 assign*.mp. 30 allocat*.mp. 31 volunteer*.mp. 32 19 or 20 or 21 or 22 or 23 or 24 or 25 or 26 or 27 or 28 or 29 or 30 or 31 33 18 and 32
Appendix 3. CENTRAL search strategy
#1 MeSH descriptor Brain Neoplasms explode all trees #2 (brain or cerebral or intracranial or intra‐cranial or intracerebral or intra‐cerebral) near/5 (metasta* or cancer* or tumor* or tumour* or neoplas* or carcinoma* or malignan*) #3 (#1 OR #2) #4 MeSH descriptor Radiotherapy explode all trees #5 Any MeSH descriptor with qualifier: RT #6 radiotherap* #7 radiation #8 irradiation #9 WBRT #10 (#4 OR #5 OR #6 OR #7 OR #8 OR #9) #11 MeSH descriptor Neurosurgical Procedures explode all trees #12 Any MeSH descriptor with qualifier: SU #13 neurosurg* #14 surg* #15 MeSH descriptor Radiosurgery, this term only #16 radiosurg* #17 resect* #18 (#11 OR #12 OR #13 OR #14 OR #15 OR #16 OR #17) #19 (#3 AND #10 AND #18)
Data and analyses
Comparison 1. Whole‐Brain Radiotherapy versus Observation.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Overall Survival | 5 | 663 | Hazard Ratio (Random, 95% CI) | 1.11 [0.83, 1.48] |
| 2 Progression Free Survival | 2 | 378 | Hazard Ratio (Random, 95% CI) | 0.76 [0.53, 1.10] |
| 3 Any intracranial disease progression at one year | 4 | 304 | Risk Ratio (IV, Random, 95% CI) | 0.47 [0.34, 0.66] |
| 4 Local intracranial disease progression at one year | 4 | 722 | Risk Ratio (IV, Random, 95% CI) | 0.38 [0.24, 0.60] |
| 5 Distant intracranial disease progression at one year | 4 | 644 | Risk Ratio (IV, Random, 95% CI) | 0.56 [0.44, 0.71] |
Comparison 2. Whole‐Brain Radiotherapy versus Observation (subgroup analysis: Focal intervention).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Overall Survival | 5 | 663 | Hazard Ratio (Random, 95% CI) | 1.11 [0.83, 1.48] |
| 1.1 Trials that use SRS alone as focal therapy | 2 | 190 | Hazard Ratio (Random, 95% CI) | 1.52 [0.63, 3.66] |
| 1.2 Trials that use Surgery alone as focal therapy | 1 | 95 | Hazard Ratio (Random, 95% CI) | 0.91 [0.59, 1.41] |
| 1.3 Trials that use either Surgery or SRS as focal therapy | 2 | 378 | Hazard Ratio (Random, 95% CI) | 0.98 [0.78, 1.23] |
| 2 Any intracranial disease progression at one year | 4 | 304 | Risk Ratio (IV, Random, 95% CI) | 0.47 [0.34, 0.66] |
| 2.1 Trials that use SRS alone as focal therapy | 2 | 190 | Risk Ratio (IV, Random, 95% CI) | 0.52 [0.35, 0.76] |
| 2.2 Trials that use Surgery alone as focal therapy | 1 | 95 | Risk Ratio (IV, Random, 95% CI) | 0.31 [0.18, 0.54] |
| 2.3 Trials that use either Surgery or SRS as focal therapy | 1 | 19 | Risk Ratio (IV, Random, 95% CI) | 0.64 [0.32, 1.31] |
| 3 Local intracranial disease progression at one year | 4 | 722 | Risk Ratio (IV, Random, 95% CI) | 0.38 [0.24, 0.60] |
| 3.1 Trials that use SRS alone as focal therapy | 2 | 268 | Risk Ratio (IV, Random, 95% CI) | 0.22 [0.04, 1.29] |
| 3.2 Trials that use Surgery alone as focal therapy | 1 | 95 | Risk Ratio (IV, Random, 95% CI) | 0.27 [0.14, 0.53] |
| 3.3 Trials that use either Surgery or SRS as focal therapy | 1 | 359 | Risk Ratio (IV, Random, 95% CI) | 0.52 [0.37, 0.72] |
| 4 Distant intracranial disease progression at one year | 4 | 644 | Risk Ratio (IV, Random, 95% CI) | 0.56 [0.44, 0.71] |
| 4.1 Trials that use SRS alone as focal therapy | 2 | 190 | Risk Ratio (IV, Random, 95% CI) | 0.61 [0.45, 0.83] |
| 4.2 Trials that use Surgery alone as focal therapy | 1 | 95 | Risk Ratio (IV, Random, 95% CI) | 0.31 [0.16, 0.62] |
| 4.3 Trials that use either Surgery or SRS as focal therapy | 1 | 359 | Risk Ratio (IV, Random, 95% CI) | 0.59 [0.43, 0.81] |
Comparison 3. Whole‐Brain Radiotherapy versus Observation (subgroup analysis: Number of brain metastases).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Overall Survival | 5 | 663 | Hazard Ratio (Random, 95% CI) | 1.11 [0.83, 1.48] |
| 1.1 Trials that include patients with only one brain metastasis | 2 | 114 | Hazard Ratio (Random, 95% CI) | 0.93 [0.62, 1.38] |
| 1.2 Trials that include patients with more than one brain metastases | 3 | 549 | Hazard Ratio (Random, 95% CI) | 1.24 [0.80, 1.91] |
| 2 Progression Free Survival | 2 | 378 | Hazard Ratio (Random, 95% CI) | 0.76 [0.53, 1.10] |
| 2.1 Trials that include patients with only one brain metastasis | 1 | 19 | Hazard Ratio (Random, 95% CI) | 1.27 [0.46, 3.52] |
| 2.2 Trials that include patients with more than one brain metastases | 1 | 359 | Hazard Ratio (Random, 95% CI) | 0.71 [0.57, 0.88] |
| 3 Any intracranial disease progression at one year | 4 | 304 | Risk Ratio (IV, Random, 95% CI) | 0.47 [0.34, 0.66] |
| 3.1 Trials that include patients with only one brain metastasis | 2 | 114 | Risk Ratio (IV, Random, 95% CI) | 0.43 [0.21, 0.87] |
| 3.2 Trials that include patients with more than one brain metastases | 2 | 190 | Risk Ratio (IV, Random, 95% CI) | 0.52 [0.35, 0.76] |
| 4 Local intracranial disease progression at one year | 4 | 722 | Risk Ratio (IV, Random, 95% CI) | 0.38 [0.24, 0.60] |
| 4.1 Trials that include patients with only one brain metastasis | 2 | 454 | Risk Ratio (IV, Random, 95% CI) | 0.40 [0.21, 0.75] |
| 4.2 Trials that include patients with more than one brain metastases | 2 | 268 | Risk Ratio (IV, Random, 95% CI) | 0.22 [0.04, 1.29] |
| 5 Distant intracranial disease progression at one year | 4 | 644 | Risk Ratio (IV, Random, 95% CI) | 0.56 [0.44, 0.71] |
| 5.1 Trials that include patients with only one brain metastasis | 2 | 454 | Risk Ratio (IV, Random, 95% CI) | 0.47 [0.25, 0.85] |
| 5.2 Trials that include patients with more than one brain metastases | 2 | 190 | Risk Ratio (IV, Random, 95% CI) | 0.61 [0.45, 0.83] |
Comparison 4. Whole‐Brain Radiotherapy versus Observation (subgroup analysis: Sequence of WBRT).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Overall Survival | 5 | 663 | Hazard Ratio (Random, 95% CI) | 1.11 [0.83, 1.48] |
| 1.1 Trials that use pre focal WBRT | 1 | 132 | Hazard Ratio (Random, 95% CI) | 1.0 [0.69, 1.45] |
| 1.2 Trials that use post focal WBRT | 4 | 531 | Hazard Ratio (Random, 95% CI) | 1.18 [0.79, 1.77] |
| 2 Any intracranial disease progression at one year | 4 | 304 | Risk Ratio (IV, Random, 95% CI) | 0.47 [0.34, 0.66] |
| 2.1 Trials that use pre focal WBRT | 1 | 132 | Risk Ratio (IV, Random, 95% CI) | 0.59 [0.40, 0.87] |
| 2.2 Trials that use post focal WBRT | 3 | 172 | Risk Ratio (IV, Random, 95% CI) | 0.41 [0.27, 0.61] |
| 3 Local intracranial disease progression at one year | 4 | 722 | Risk Ratio (IV, Random, 95% CI) | 0.38 [0.24, 0.60] |
| 3.1 Trials that use pre focal WBRT | 1 | 210 | Risk Ratio (IV, Random, 95% CI) | 0.38 [0.20, 0.74] |
| 3.2 Trials that use post focal WBRT | 3 | 512 | Risk Ratio (IV, Random, 95% CI) | 0.35 [0.17, 0.72] |
| 4 Distant intracranial disease progression at one year | 4 | 644 | Risk Ratio (IV, Random, 95% CI) | 0.56 [0.44, 0.71] |
| 4.1 Trials that use pre focal WBRT | 1 | 132 | Risk Ratio (IV, Random, 95% CI) | 0.65 [0.46, 0.91] |
| 4.2 Trials that use post focal WBRT | 3 | 512 | Risk Ratio (IV, Random, 95% CI) | 0.50 [0.36, 0.71] |
Comparison 5. Whole‐Brain Radiotherapy versus Observation (subgroup analysis: Equivalent Dose in 2Gy Fractions with α/β ratio of 2 (EQD22)).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Overall Survival | 5 | 663 | Hazard Ratio (Random, 95% CI) | 1.11 [0.83, 1.48] |
| 1.1 Trials that use WBRT with EQD22 > 40Gy | 1 | 95 | Hazard Ratio (Random, 95% CI) | 0.91 [0.59, 1.41] |
| 1.2 Trials that use WBRT with EQD22 < 40Gy | 4 | 568 | Hazard Ratio (Random, 95% CI) | 1.20 [0.82, 1.74] |
| 2 Any intracranial disease progression at one year | 4 | 304 | Risk Ratio (IV, Random, 95% CI) | 0.47 [0.34, 0.66] |
| 2.1 Trials that use WBRT with EQD22 > 40Gy | 1 | 95 | Risk Ratio (IV, Random, 95% CI) | 0.31 [0.18, 0.54] |
| 2.2 Trials that use WBRT with EQD22 < 40Gy | 3 | 209 | Risk Ratio (IV, Random, 95% CI) | 0.55 [0.41, 0.74] |
| 3 Local intracranial disease progression at one year | 4 | 722 | Risk Ratio (IV, Random, 95% CI) | 0.38 [0.24, 0.60] |
| 3.1 Trials that use WBRT with EQD22 > 40Gy | 1 | 95 | Risk Ratio (IV, Random, 95% CI) | 0.27 [0.14, 0.53] |
| 3.2 Trials that use WBRT with EQD22 < 40Gy | 3 | 627 | Risk Ratio (IV, Random, 95% CI) | 0.43 [0.26, 0.72] |
| 4 Distant intracranial disease progression at one year | 4 | 644 | Risk Ratio (IV, Random, 95% CI) | 0.56 [0.44, 0.71] |
| 4.1 Trials that use WBRT with EQD22 > 40Gy | 1 | 95 | Risk Ratio (IV, Random, 95% CI) | 0.31 [0.16, 0.62] |
| 4.2 Trials that use WBRT with EQD22 < 40Gy | 3 | 549 | Risk Ratio (IV, Random, 95% CI) | 0.60 [0.49, 0.75] |
5.3. Analysis.

Comparison 5 Whole‐Brain Radiotherapy versus Observation (subgroup analysis: Equivalent Dose in 2Gy Fractions with α/β ratio of 2 (EQD22)), Outcome 3 Local intracranial disease progression at one year.
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Aoyama 2006.
| Methods | Randomized Controlled Study Multi centre Sample size calculation ‐ Power:80% to detect an absolute difference of 30% in median survival time with a 2 sided alpha level of 0.05 ‐ Calculated sample size: 178 patients ‐ Randomized: 132 patients |
|
| Participants | Mean age: 62 % patients with primary breast or lung cancers: 73 % patients with RPA Class 1: 14 % Withdrawals: 0 No. of patients in the Focal therapy plus WBRT arm: 65 No. of patients in the Focal therapy only arm: 67 Baseline Characteristics Comparability: Comparable totally Inclusion criteria ‐ 18 years or older ‐ One to four brain metastases, each with maximum diameter of no more than 3cm on contrast enhanced magnetic resonance imaging scans, derived from a histologically confirmed systemic cancer. ‐ KPS score >=70 Exclusion criteria ‐ Metastases from small cell carcinoma, lymphoma, germinoma and multiple myeloma |
|
| Interventions | Type of Focal intervention: Stereotactic Radiosurgery Dose of WBRT: 30Gy in 10 fractions, EQD22: 37.5Gy Sequence of WBRT: Pre focal intervention Time interval between focal intervention and WBRT: not reported |
|
| Outcomes | Overall survival Local intracranial disease progression Distant intracranial disease progression Neurocognitive function Toxicity |
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | A permutated blocks randomizations algorithm was used with a block size of 4 |
| Allocation concealment (selection bias) | Low risk | Randomization was performed at the Hokkaido University Hospital Data Center |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No missing outcome data |
| Selective reporting (reporting bias) | Low risk | All pre‐specified primary and secondary outcomes were reported |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | The risk of bias from lack of blinding was high for subjective outcomes such as progression free survival, intracranial disease progression, neurocognitive function, health related quality of life and neurological adverse events and low for objective outcomes such as overall survival |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | The risk of bias from lack of blinding was high for subjective outcomes such as progression free survival, intracranial disease progression, neurocognitive function, health related quality of life and neurological adverse events and low for objective outcomes such as overall survival |
Chang 2009.
| Methods | Randomized Controlled Study Single Center Sample size calculation ‐ Power 80% to detect a 30% difference based on the neurocognitive endpoint of total recall ‐ Calculated sample size: 90 patients ‐ Randomized: 58 patients |
|
| Participants | Mean age: 63 % patients with primary breast or lung cancers: 63 % patients with RPA Class 1: 17 % Withdrawals: 0 No. of patiens in the Focal therapy plus WBRT arm: 28 No. of patients in the Focal therapy only arm: 30 Baseline Characteristics Comparability: Comparable mostly Inclusion criteria ‐ 18 years or older ‐ RPA class one to two ‐ KPS score >= 70 ‐ one to three newly diagnosed brain metastases eligible for stereotactic radiosurgery ‐ brain magnetic resonance imaging within one month of enrolment Exclusion criteria ‐ prior brain surgery, radiosurgery or whole brain radiotherapy ‐ leukaemia, lymphoma, germ cell tumour, small cell lung cancer, leptomeningeal disease or unknown primary tumour ‐ pregnant |
|
| Interventions | Type of Focal intervention: Stereotactic Radiosurgery Dose of WBRT: 30Gy in 12 fractions, EQD22: 33.75Gy Sequence of WBRT: Post focal intervention Time interval between focal intervention and WBRT: 3 weeks |
|
| Outcomes | Overall survival Local intracranial disease progression Distant intracranial disease progression Neurocognitive function Quality of life Toxicity |
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Randomization was done by computer using a standard permutated block algorithm |
| Allocation concealment (selection bias) | Low risk | The sequence was concealed until interventions were assigned by the Clinical Oncology Research database computer |
| Incomplete outcome data (attrition bias) All outcomes | High risk | While intention to treat analysis was utilized for survival outcomes, only 53% of the patients were included for analysis of neurocognitive function outcomes |
| Selective reporting (reporting bias) | High risk | Although all pre‐specified primary and secondary outcomes were reported, it is likely that there is high risk of reporting bias as the patients in the observation arm had significantly better survival than WBRT arm while no other study had such a survival difference |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | The risk of bias from lack of blinding was high for subjective outcomes such as progression free survival, intracranial disease progression, neurocognitive function, health related quality of life and neurological adverse events and low for objective outcomes such as overall survival |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | The risk of bias from lack of blinding was high for subjective outcomes such as progression free survival, intracranial disease progression, neurocognitive function, health related quality of life and neurological adverse events and low for objective outcomes such as overall survival |
Kocher 2011.
| Methods | Randomized Controlled Study Multi centre Sample size calculation ‐ Power:80% to detect a 11% difference in the proportion of patients alive with performance status <= 2 at 6 months, with a 2 sided alpha level of 0.05 ‐ Calculated sample size: 340 patients ‐ Randomized: 359 patients |
|
| Participants | Mean age: 60 % patients with primary breast or lung cancers: 65 % patients with RPA Class 1: Not reported % Withdrawals: 0 No. of patiens in the Focal therapy plus WBRT arm: 180 No. of patients in the Focal therapy only arm: 179 Baseline Characteristics Comparability: Comparable mostly Inclusion criteria ‐ 18 years or older ‐ WHO performance status <=2 ‐ One to three brain metastases ‐ Radiosurgery: single metastasis <= 3.5cm, multiple metastases <= 2.5cm in diameter ‐ Surgery: complete surgical resection ‐ Radiosurgery: histologic confirmation of primary tumour or other metastases <= 4 years ago, stereotactic biopsy of the brain metastases otherwise ‐ Stable systemic cancer for => 3 months and/or asymptomatic synchronous primary tumour without metastases outside the CNS or unknown primary tumour Exclusion criteria ‐ Brain metastasis of small cell lung cancer, lymphoma, leukaemia, myeloma, germ cell tumours ‐ Brain stem metastases ‐ Leptomeningeal metastases ‐ Recurrent brain metastases after surgery and/or radiosurgery and/or brain irradiation ‐ Inability to interrupt chemotherapy during whole brain radiotherapy |
|
| Interventions | Type of Focal intervention: Stereotactic radiosurgery or Surgery Dose of WBRT: 30Gy in 10 fractions, EQD22: 37.5Gy Sequence of WBRT: Post focal intervention Time interval between focal intervention and WBRT: 4 weeks (surgery), 2 weeks (radiosurgery) |
|
| Outcomes | Overall survival Progression free survival Local intracranial disease progression Distant intracranial disease progression Quality of life Toxicity |
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Randomized using a minimization algorithm |
| Allocation concealment (selection bias) | Unclear risk | The method of concealment is not described |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No missing outcome data |
| Selective reporting (reporting bias) | Low risk | All pre‐specified primary and secondary outcomes were reported |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | The risk of bias from lack of blinding was high for subjective outcomes such as progression free survival, intracranial disease progression, neurocognitive function, health related quality of life and neurological adverse events and low for objective outcomes such as overall survival |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | The risk of bias from lack of blinding was high for subjective outcomes such as progression free survival, intracranial disease progression, neurocognitive function, health related quality of life and neurological adverse events and low for objective outcomes such as overall survival |
Patchell 1998.
| Methods | Randomized Controlled Study Multi centre Sample size calculation ‐ Power:80% to detect a 34% difference in brain recurrence rate with a 2 sided alpha test of 0.05 ‐ Calculated sample size: 80 patients ‐ Randomized: 95 patients |
|
| Participants | Mean age: 59 % patients with primary breast or lung cancers: 69 % patients with RPA Class 1: Not reported % Withdrawals: 0 No. of patiens in the Focal therapy plus WBRT arm: 49 No. of patients in the Focal therapy only arm: 46 Baseline Characteristics Comparability: Comparable totally Inclusion criteria ‐ 18 years or older ‐ tissue proven diagnosis of metastatic brain tumour obtained from a complete resection of a single brain metastasis Exclusion criteria ‐ brain metastases that had not been completely removed by surgery ‐ leptomeningeal metastases ‐ previous cranial radiotherapy ‐ need for immediate treatment to prevent acute neurologic deterioration ‐ concomitant second malignancies ‐ KPS score < 70 ‐ small cell lung cancer, germ cell tumour, lymphoma, leukaemia, multiple myeloma |
|
| Interventions | Type of Focal intervention: Surgery Dose of WBRT: 50.4Gy in 28 fractions, EQD22: 47.88Gy Sequence of WBRT: Post focal intervention Time interval between focal intervention and WBRT: 28 days |
|
| Outcomes | Overall survival Local intracranial disease progression Distant intracranial disease progression |
|
| Notes | Trial protocol obtained from the authors | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Sequence was generated using computer generated random numbers |
| Allocation concealment (selection bias) | Low risk | Allocation was done at a central site |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No missing outcome data |
| Selective reporting (reporting bias) | Low risk | All pre‐specified primary and secondary outcomes were reported |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | The risk of bias from lack of blinding was high for subjective outcomes such as progression free survival, intracranial disease progression, neurocognitive function, health related quality of life and neurological adverse events and low for objective outcomes such as overall survival |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | The risk of bias from lack of blinding was high for subjective outcomes such as progression free survival, intracranial disease progression, neurocognitive function, health related quality of life and neurological adverse events and low for objective outcomes such as overall survival |
Roos 2006.
| Methods | Randomized Controlled Study Multi centre Sample size calculation ‐ Power:80% to detect a 3 months difference in median duration of central nervous system failure free survival ‐ Calculated sample size: 130 patients ‐ Randomized: 19 patients |
|
| Participants | Mean age: 58 % patients with primary breast or lung cancers: 47 % patients with RPA Class 1: 21 % Withdrawals: 0 No. of patiens in the Focal therapy plus WBRT arm: 10 No. of patients in the Focal therapy only arm: 9 Baseline Characteristics Comparability: Comparable mostly Inclusion criteria ‐ 18 years or older ‐ Single brain metastasis from an extra‐cranial primary malignancy with complete surgical resection or radiosurgery within 6 weeks of registration ‐ Post surgery or radiosurgery WHO performance status <= 2 Exclusion criteria ‐ primary brain tumour ‐ small cell lung cancer ‐ seminoma ‐ lymphoma ‐ myeloma ‐ leukaemia ‐ macroscopic residual disease following surgery ‐ meningeal disease ‐ life expectancy due to extra‐cranial disease predicted to be less than 6 months ‐ prior brain radiation |
|
| Interventions | Type of Focal intervention: Stereotactic Radiosurgery or Surgery Dose of WBRT: 30Gy in 10 fractions, EQD22: 37.5Gy Sequence of WBRT: Post focal intervention Time interval between focal intervention and WBRT: not reported |
|
| Outcomes | Overall survival Progression free survival Local or Distant intracranial disease progression Neurocognitive function Quality of life Toxicity |
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Sequence was generated using a computer generated randomizations chart |
| Allocation concealment (selection bias) | Low risk | Allocation was done at a central site |
| Incomplete outcome data (attrition bias) All outcomes | High risk | The trial closed prematurely due to poor accrual |
| Selective reporting (reporting bias) | Low risk | All pre‐specified primary and secondary outcomes were reported |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | The risk of bias from lack of blinding was high for subjective outcomes such as progression free survival, intracranial disease progression, neurocognitive function, health related quality of life and neurological adverse events and low for objective outcomes such as overall survival |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | The risk of bias from lack of blinding was high for subjective outcomes such as progression free survival, intracranial disease progression, neurocognitive function, health related quality of life and neurological adverse events and low for objective outcomes such as overall survival |
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Chougule 2000 | Abstract only. Did not provide sufficient data for qualitative or quantitative analysis |
| Fogarty 2011 | Ongoing trial. Only the trial protocol was published. |
Characteristics of ongoing studies [ordered by study ID]
ACTRN12607000512426.
| Trial name or title | Whole Brain Radiotherapy following local treatment of intracranial metastases of melanoma – A randomised phase III trial |
| Methods | Randomized Controlled Study Multi centre The randomisation process will be coordinated centrally and sites will phone to request randomisation for a patient and confirm eligibility information by fax. Once randomised, the investigator and patient will be made aware of the assigned treatment Minimisation will be used as the randomisation method for patients in this trial Open (masking not used) Parallel Assignment |
| Participants | Inclusion criteria 1.1‐3 intracranial metastases on MRI from melanoma, locally treated with either surgical excision and/or stereotactic irradiation. 2.Life expectancy of at least 6 months 3.Aged 18 years or older 4.WBRT must be within 8 weeks of completion of localised treatment and within 4 weeks of randomisation 5.Able to have an MRI brain scan with contrast. ‘estimated Glomerular Filtrate Rate’ (eGFR) is adequate at the discretion of the radiologist and capable of having gadolinium‐containing contrast medium for MRI as per practice guidelines 6.Complete localised treatment of all these metastases no more than 6 weeks prior to randomisation 7.An Eastern Cooperative Oncology Group (ECOG) performance status of 2 or less at randomisation 8.CT scan of chest, abdomen and pelvis as a minimum prior to randomisation. Scans must be within 8 weeks of randomisation 9.Serum Lactate Dehydrogenase (LDH) must be = 2 x upper limit of normal 10.Able to provide written informed consent Age 18 years and above Exclusion criteria 1.Any untreated intracranial disease 2.Any previous intracranial treatment (surgical excision and/or stereotactic irradiation treatment and/or WBRT) prior to this diagnosis of intracranial melanoma 3.Evidence of leptomeningeal disease on pre‐local treatment MRI scan 4.Patients with prior cancers, except: ‐ Those diagnosed more than five years ago with no evidence of disease recurrence within this time; ‐ Successfully treated basal cell and squamous cell skin carcinoma; ‐ Carcinoma in‐situ of the cervix 5.A medical or psychiatric condition that compromises ability to give informed consent or complete the protocol 6.Positive urine pregnancy test for women of childbearing potential within a week of registration onto the trial |
| Interventions | (i) Whole brain radiotherapy ‐ a minimum of 30 Gy in 10 fractions given as one fraction per day within 4 weeks of randomisation (ii) Observation |
| Outcomes | Primary Proportion of patients with distant intracranial failure as determined by magnetic resonance imaging (MRI) assessment Secondary Time to intracranial failure (local, distant and overall (local+ distant)) as determined by MRI Quality of life as measured by QLQ‐C30 and BN‐20 Neurocognitive function as measured by Hopkins Verbal Learning Test, Controlled Oral Word Association Test, Trail Making Test Part A and B Progression‐free and overall survival |
| Starting date | 1/12/2008 |
| Contact information | Name: Elizabeth Paton Address:The Australia and New Zealand Melanoma Trials Group (ANZMTG), University of Sydney Level 2 1A Eden St North Sydney NSW 2060 Country:Australia Tel:+61 2 9911 7354 Fax:+61 2 9954 9435 Email:anzmtg@usyd.edu.au |
| Notes |
NCT00377156.
| Trial name or title | Stereotactic Radiation Therapy With or Without Whole‐Brain Radiation Therapy in Treating Patients With Brain Metastases |
| Methods | Randomized Controlled Study Multi centre |
| Participants | Ages Eligible for Study: 18 Years and older
Genders Eligible for Study: Both
Accepts Healthy Volunteers: No Criteria DISEASE CHARACTERISTICS: Diagnosis of cerebral metastases meeting the following criteria: One to three presumed brain metastases Metastases must be from a histologically confirmed extracerebral site (e.g., lung, breast, prostate) Histological confirmation may have been from the primary tumour site, from another metastatic site (e.g., osseous metastasis, adrenal metastasis), or from the metastatic brain lesion(s) Each lesion must measure less than 3.0 cm by contrast MRI of the brain performed within the past 21 days Lesions must not be within 5 mm of the optic chiasm or within the brainstem Eligibility for treatment with gamma knife or linear accelerator‐based radiosurgery confirmed by a radiation oncologist No primary germ cell tumour, small cell carcinoma, or lymphoma No leptomeningeal metastases Hormone receptor status not specified PATIENT CHARACTERISTICS: Male or female Menopausal status not specified ECOG performance status 0‐2 Not pregnant Negative pregnancy test Fertile patients must use effective contraception Male patients must continue to use contraception for 3 months after the completion of radiotherapy No pacemaker or other MRI‐incompatible metal in the body No known allergy to gadolinium PRIOR CONCURRENT THERAPY: More than 7 days since prior and no concurrent chemotherapy No prior cranial radiotherapy No prior resection of cerebral metastases Concurrent hormonal agents, steroids, and/or anticonvulsants allowed |
| Interventions | Active Comparator: Arm I
Patients undergo stereotactic radiosurgery (SRS) Experimental: Arm II Patients undergo SRS as in arm I. Within 14 days, patients then undergo whole‐brain radiotherapy 5 days a week for 2.5 weeks. |
| Outcomes | Primary Outcome Measures: Survival as measured by stratified logrank test and Cox proportional hazards models at 6 months [ Designated as safety issue: No ] Secondary Outcome Measures: Time to CNS failure [ Designated as safety issue: No ] Quality of life, in terms of brain sub scale and physical and emotional functioning, as measured by FACT‐BR questionnaire, physician‐assessed neurological signs and symptoms, and treatment‐related adverse events at 3 months post treatment [ Designated as safety issue: Yes ] Functional independence as measured by Barthel Activities of Daily Living Index and ECOG performance status [ Designated as safety issue: No ] Duration of functional independence [ Designated as safety issue: No ] Neurocognitive status as measured by objective assessment of changes in cognitive signs and symptoms and physician‐assessed toxicities at 3 months post treatment [ Designated as safety issue: Yes ] |
| Starting date | Jul 2006 |
| Contact information | Study Chair:Paul D. Brown, MD Mayo Clinic Investigator:Kurt A. Jaeckle, MD Mayo Clinic Investigator:Richard L. Deming, MD Mercy Therapeutic Radiology Associates, PC at Mercy Medical Center ‐ Des Moines Investigator:Elana Farace, PhD Milton S. Hershey Medical Center Investigator:Bruce Pollock, MD Mayo Clinic Study Chair:Anthony Asher, MD, FACS Carolina Neurosurgery and Spine Associates Investigator:Fred G. Barker, MD Massachusetts General Hospital Study Chair:Larry Kleinberg, MD Sidney Kimmel Comprehensive Cancer Center |
| Notes |
Differences between protocol and review
With regards to time to event data synthesis, we have noted in the post‐protocol stage that the included studies use either the Kaplan‐Meier or the competing risk methods to analyse intracranial disease progression. We have found that the Kaplan‐Meier and the competing risk methods are two different methods used in the estimation of cumulative incidence of an event of interest. The Kaplan‐Meier method does not account for any competing risk factors such as death but competing risk method does. This means that the Kaplan‐Meier approach and the competing risk method will result in an identical estimate of cumulative incidence of an event only when there are no competing risks. However when competing risks are present, the estimate obtained using the Kaplan‐Meier method is numerically larger than the competing risk method (Satagopan 2004). Since death is a competing risk factor in the analysis of intracranial disease progression data, we believed that it is inappropriate to pool the intracranial disease progression results across studies. We have also amended the data synthesis section accordingly to reflect this difference between protocol and review.
Although a funnel plot was initially planned during the protocol to examine publication bias, but due to the small number of included studies, funnel plot analysis was deemed not useful and hence not conducted.
Contributions of authors
Protocol development: YYS, IWKT, KHL, KWY, JJL. Selection of trials for inclusion: YYS, IWKT, KHL, KWY, JJL. Methodological assessment and data extraction: YYS, IWKT, KHL, KWY, JJL. Data entry and management: YYS. Data analysis: YYS, IWKT, KHL, KWY, JJL. Preparation of manuscript: YYS, IWKT, KHL, KWY, JJL.
Sources of support
Internal sources
None, Not specified.
External sources
No sources of support supplied
Declarations of interest
None known
Edited (no change to conclusions)
References
References to studies included in this review
Aoyama 2006 {published data only}
- Aoyama H, Shirato H, Tago M, Nakagawa K, Toyoda T, Hatano K, et al. Stereotactic Radiosurgery Plus Whole‐Brain Radiation Therapy vs Stereotactiv Radiosurgery alone for Treatment of Brain Metastases. The Journal of the American Medical Association 2006;295(21):2483‐90. [DOI] [PubMed] [Google Scholar]
Chang 2009 {published data only}
- Chang E, Wefel J, Hess K, Allen P, Lang F, Kornguth D, et al. Neurocognition in patients with brain metastases treated with radiosurgery or radiosurgery plus whole‐brain irradiation: a randomised controlled trial. Lancet Oncology 2009;10(11):1037‐44. [DOI] [PubMed] [Google Scholar]
Kocher 2011 {published data only}
- Kocher M, Soffietti R, Abacioglu U, Villa S, Fauchon F, Baumert B, et al. Adjuvant whole‐brain radiotherapy versus observation after radiosurgery or surgical resection of one to three cerebral metastases: Results of the EORTC 22952‐26001 Study. Journal of Clinical Oncology 2011;29(2):134‐41. [DOI] [PMC free article] [PubMed] [Google Scholar]
Patchell 1998 {published data only}
- Patchell R, Tibbs P, Regine W, Dempsey R, Mohiuddin M, Kryscio R, et al. Postoperative Radiotherapy in the Treatment of Single Metastases to the Brain. The Journal of the American Medical Association 1998;280(17):1485‐9. [DOI] [PubMed] [Google Scholar]
Roos 2006 {published data only}
- Roos D, Writh A, Burmeister B, Spry N, Drummond K, Beresford J, et al. Whole brain irradiation following surgery or radiosurgery for solitary brain metastases: Mature results of a prematurely closed randomized Trans‐Tasman Radiation Oncology Group trial (TROG 98.05). Radiotherapy and Oncology 2006;80(3):318‐22. [DOI] [PubMed] [Google Scholar]
References to studies excluded from this review
Chougule 2000 {published data only}
- Chougule P, Burton‐Williams M, Saris S, Zheng Z, Ponte B, Noren G, et al. Randomized treatment off brain metastasis with gamma knife radiosurgery, whole brain radiotherapy or both. International Journal of Radiation Oncology Biology and Physics. 2000; Vol. 48:114.
Fogarty 2011 {published data only}
- Fogarty G, Morton R, Vardy J, Nowak A, Mandel C, Forder P, et al. Whole brain radiotherapy after local treatment of brain metastases in melanoma patients ‐ a randomised phase III trial. BMC Cancer 2011;11(142):1‐8. [DOI] [PMC free article] [PubMed] [Google Scholar]
References to ongoing studies
ACTRN12607000512426 {unpublished data only}
- ACTRN12607000512426. Whole Brain Radiotherapy following local treatment of intracranial metastases of melanoma – A randomised phase III trial. www.anzctr.org.au/ACTRN12607000512426.aspx (accessed 11 October 2011).
NCT00377156 {unpublished data only}
- NCT00377156. Stereotactic Radiation Therapy With or Without Whole‐Brain Radiation Therapy in Treating Patients With Brain Metastases. clinicaltrials.gov/ct/show/NCT00377156 (accessed 11 October 2011).
Additional references
Al‐Shamy 2009
- Al‐Shamy G, Sawaya R. Management of brain metastases: the indispensable role of surgery. Journal of Neuro‐Oncology 2009;92(3):275‐82. [DOI] [PubMed] [Google Scholar]
Andrews 2004
- Andrews DW, Scott CB, Sperduto PW, Flanders AE, Gaspar LE, Schell MC, et al. Whole brain radiation therapy with or without stereotactic radiosurgery boost for patients with one to three brain metastases: phase III results of RTOG 9508 randomised trial. Lancet 2004;363:1665‐72. [DOI] [PubMed] [Google Scholar]
Aoyama 2007
- Aoyama H, Tago M, Kato N, Toyoda T, Kenjyo M, Hirota S, et al. Neurocognitive function of patients with brain metastasis who received either whole brain radiotherapy plus stereotactic radiosurgery or radiosurgery alone. International Journal of Radiation Oncology Biology and Physics 2007;68(5):1388‐95. [DOI] [PubMed] [Google Scholar]
Brown 2008
- Brown P, Asher A, Farace E. Adjuvant whole brain radiotherapy: strong emotions decide but rational studies are needed. International Journal of Radiation Oncology, Biology, Physics 2008;70(5):1305‐9. [DOI] [PubMed] [Google Scholar]
DeAngelis 1989
- DeAngelis L, Delattrre JY, Posner J. Radiation‐induced dementia in patients cured of brain metastases. Neurology 1989;39(6):789‐96. [DOI] [PubMed] [Google Scholar]
Deeks 2001
- Deeks JJ, Altman DG, Bradburn MJ. Statistical methods for examining heterogeneity and combining results from several studies in meta‐analysis. In: Egger M, Davey Smith G, Altman DG editor(s). Systematic Reviews in Health Care: Meta‐analysis in Context. 2nd Edition. London: BMJ Publication Group, 2001. [Google Scholar]
DerSimonian 1986
- DerSimonian R, Laird N. Meta‐analyses in clinical trials. Controlled Clinical Trials 1986;7:177‐88. [DOI] [PubMed] [Google Scholar]
Gaspar 1997
- Gaspar L, Scott C, Rotman M, Asbell S, Phillips T, Wasserman T, et al. Recursive partitioning analysis (RPA) of prognostic factors in three radiation therapy oncology group (RTOG) brain metastases trials. International Journal of Radiation Oncology, Biology, Physics 1997;37(4):745‐51. [DOI] [PubMed] [Google Scholar]
Gavrilovic 2005
- Gavrilovic IT, Poser JB. Brain metastases: epidemiology and pathophysiology. Journal of Neuro‐Oncology 2005;75(1):5‐14. [DOI] [PubMed] [Google Scholar]
GRADE Working Group 2012 [Computer program]
- GRADE Working Group. Guideline Development Tool. McMaster University and Synaway.com, 2012.
Hart 2011
- Hart MG, Grant R, Walker M, Dickinson HO. Surgical resection and whole brain radiation therapy versus whole brain radiation therapy alone for single brain metastases. Cochrane Database of Systematic Reviews 2011, Issue 3. [DOI: 10.1002/14651858.CD003292.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Higgins 2003
- Higgins JPT, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta‐analyses. BMJ 2003;327(7416):557‐60. [DOI] [PMC free article] [PubMed] [Google Scholar]
Higgins 2011
- Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0. The Cochrane Collaboration, March 2011. [Google Scholar]
Jemal 2011
- Jemal A, Bray F, Center M, Ferlay J, Ward E, Forman D. Global Cancer Statistics. CA: A Cancer Journal for Clinicians 2011;61:69‐90. [DOI] [PubMed] [Google Scholar]
Kalkanis 2010
- Kalkanis S, Kondziolka D, Gaspar L, Burri S, Asher A, Cobbs C, et al. The role of surgical resection in the management of newly diagnosed brain metastases: a systematic review and evidence‐based clinical guidelines. Journal of Neuro‐oncology 2010;96:33‐43. [DOI] [PMC free article] [PubMed] [Google Scholar]
Khuntia 2006
- Khuntia D, Brown P, Li J, Mehta MP. Whole‐brain radiotherapy in the management of brain metastasis. Journal of Clinical Oncology 2006;24(8):1295‐304. [DOI] [PubMed] [Google Scholar]
Li 2007
- Li J, Bentzen SM, Renschler M, Mehta M. Regression after whole‐brain radiation therapy for brain metastases correlates with survival and improved neurocognitive function. Journal of Clinical Oncology 2007;25(10):1260‐6. [DOI] [PubMed] [Google Scholar]
Linskey 2010
- Linskey M, Andrews D, Asher A, Burri S, Kondziolka D, Robinson P, et al. The role of stereotactic radiosurgery in the management of patients with newly diagnosed brain metastases: a systematic review and evidence‐based clinical practice guidelines. Journal of Neuro‐Oncology 2010;96:45‐68. [DOI] [PMC free article] [PubMed] [Google Scholar]
Mamon 2005
- Mamon HJ, Yeap BY, Janne PA, Reblando J, Shrager S, Jaklitsch MT, et al. High risk of brain metastases in surgically staged IIIA non‐small cell lung cancer patients treated with surgery, chemotherapy and radiation. Journal of Clinical Oncology 2005;23(7):1530‐7. [DOI] [PubMed] [Google Scholar]
McHaffie 2012
- McHaffie DR, Kuntia D, Suh JH, Tome W, Mehta MP. Stereotactic Irradiation: Linear accelerator and Gamma knife. In: Gunderson LL, Tepper JE editor(s). Clinical Radiation Oncology. 3rd Edition. Saunders, 2012:331‐43. [Google Scholar]
Mehta 2005
- Mehta MP, Tsao MN, Whelan TJ, Morris DE, Hayman JA, Flickinger JC, et al. The American Society of Therapeutic Radiology and Oncology (ASTRO) evidence‐based review of the role of radiosurgery for brain metastases. International Journal of Radiation Oncology, Biology, Physics 2005;63(1):37‐46. [DOI] [PubMed] [Google Scholar]
Meyers 2003
- Meyers CA, Wefel JS. The use of the Mini‐Mental State Examination to assess cognitive function in cancer trials: No, Ifs, Ands, Buts, or Sensitivity. Journal of Clinical Oncology 2003;21(19):3557‐8. [DOI] [PubMed] [Google Scholar]
Meyers 2006
- Meyers CA, Brown PD. Role and relevance of neurocognitive assessment in clinical trials of patients with CNS tumors. Journal of Clinical Oncology 2006;24(8):1305‐09. [DOI] [PubMed] [Google Scholar]
NCCN 2012
- National Comprehensive Cancer Network. Central Nervous System Cancers. http://www.nccn.org/professionals/physician_gls/pdf/cns.pdf 2012; Vol. Version I.2012.
Parmar 1998
- Parmar MK, Torri V, Stewart L. Extracting summary statistics to perform meta‐analyses of the published literature for survival endpoints. Statistics in Medicine 1998;17(24):2815‐34. [DOI] [PubMed] [Google Scholar]
Paterson 1982
- Paterson AH, Agarwal M, Lees A, Hanson J, Szafran O. Brain metastases in breast cancer patients receiving adjuvant chemotherapy. Cancer 1982;49(4):651‐4. [DOI] [PubMed] [Google Scholar]
Patil 2010
- Patil CG, Pricola K, Garg SK, Bryant A, Black KL. Whole brain radiation therapy (WBRT) alone versus WBRT and radiosurgery for the treatment of brain metastases. Cochrane Database of Systematic Reviews 2010, Issue 6. [DOI: 10.1002/14651858.CD006121.pub2] [DOI] [PubMed] [Google Scholar]
Platta 2010
- Platta CS, Khuntia D, Mehta MP, Suh JH. Current treatment strategies for brain metastasis and complications from therapeutic techniques: a review of current literature. American Journal of Clinical Oncology 2010;33(4):398‐407. [DOI] [PubMed] [Google Scholar]
Satagopan 2004
- Satagopan JM, Ben‐Porat L, Berwick M, Robson M, Kutler D, Auerback AD. A note on competing risks in survival data analysis. British Journal of Cancer 2004;91:1229‐35. [DOI] [PMC free article] [PubMed] [Google Scholar]
Smedby 2009
- Smedby KE, Brandt L, Backlund ML, Blomqvist P. Brain metastases admissions in Sweden between 1987 and 2006. British Journal of Cancer 2009;101(11):1919‐24. [DOI] [PMC free article] [PubMed] [Google Scholar]
Soffietti 2013
- Soffietti R, Kocher M, Abacioglu U, Villa S, Fauchon F, Baumert B, et al. A European Organization for Research and Treatment of Cancer phase III trial of adjuvant whole‐brain radiotherapy versus observation in patients with one to three brain metastases from solid tumors after surgical resection or radiosurgery: Quality of life results. Journal of Clinical Oncology 2013;31:65‐72. [DOI] [PubMed] [Google Scholar]
Sperduto 2008
- Sperduto PW, Berkey B, Gaspar L, Mehta M, Curran W. A new prognostic index and comparison to three other indices for patients with brain metastases: an analysis of 1960 patients in the RTOG database. International Journal of Radiation Oncology, Biology, Physics 2008;70(2):510‐4. [DOI] [PubMed] [Google Scholar]
Stafinski 2006
- Stafinski T, Jhangri GS, Yan E, Menon D. Effectiveness of stereotactic radiosurgery alone or in combination with whole brain radiotherapy compared to conventional surgery and/or whole brain radiotherapy for the treatment of one or more brain metastases: a systematic review and meta‐analysis. Cancer Treatment Reviews 2006;32(3):203‐13. [DOI] [PubMed] [Google Scholar]
Stewart 1993
- Stewart LA, Parmar MKB. Meta‐analysis of the literature or of individual patient data: is there a difference?. Lancet 1993;341:418‐22. [DOI] [PubMed] [Google Scholar]
Suh 2010
- Suh JH, Videtic GM, Aref AM, Germano I, Goldsmith BJ, Imperato JP, et al. ACR Appropriateness Criteria: single brain metastasis. Current Problems in Cancer 2010;34(3):162‐74. [DOI] [PubMed] [Google Scholar]
Suh 2010a
- Suh JH. Stereotactic radiosurgery for the management of brain metastases. The New England Journal of Medicine 2010;362(12):1119‐27. [DOI] [PubMed] [Google Scholar]
Sundermeyer 2005
- Sundermeyer ML, Meropol NJ, Rogatko A, Wang H, Cohen SJ. Changing patterns of bone and brain metastases in patients with colorectal cancer. Clinical Colorectal Cancer 2005;5(2):108‐13. [DOI] [PubMed] [Google Scholar]
Thomas 2000
- Thomas AJ, Rock JP, Johnson CC, Weiss L, Jacobsen G, Rosenblum M. Survival of patients with synchronous brain metastases: an epidemiological study in southeastern Michigan. Neurosurgical Focus 2000;9(2):1‐5. [DOI] [PubMed] [Google Scholar]
Tierney 2007
- Tierney JF, Stewart LA, Ghersi D, Burdett S, Sydes MR. Practical methods for incorporating summary time‐to‐event data into meta‐analysis. Trials 2007;8:16. [DOI] [PMC free article] [PubMed] [Google Scholar]
Tsao 2012
- Tsao MN, Lloyd N, Wong RKS, Chow E, Rakovitch E, Laperriere N, Xu W, Sahgal A. Whole brain radiotherapy for the treatment of newly diagnosed multiple brain metastases. Cochrane Database of Systematic Reviews 2012, Issue 4. [DOI: 10.1002/14651858.CD003869.pub3] [DOI] [PMC free article] [PubMed] [Google Scholar]
Tsao 2012a
- Tsao M, Xu W, Sahgal A. A Meta‐Analysis Evaluating Stereotactic Radiosurgery, Whole‐Brain Radiotherapy, or Both for Patients Presenting with a Limited Number of Brain Metastases. Cancer 2012;118:2486‐93. [DOI] [PubMed] [Google Scholar]
Tsao 2012b
- Tsao M, Rades D, Wirth A, Lo S, Danielson B, Gaspar L, et al. Radiotherapeutic and surgical management for newly diagnosed brain metastasis (es): An American Society for Radiation Oncology evidence‐based guideline. Practical Radiation Oncology 2012;2:210‐25. [DOI] [PMC free article] [PubMed] [Google Scholar]
Vogelbaum 2006
- Vogelbaum MA, Suh JH. Resectable brain metastases. Journal of Clinical Oncology 2006;24(8):1289‐94. [DOI] [PubMed] [Google Scholar]
Wen 2011
- Wen PY, Loeffler JS. Overview of the clinical manifestations, diagnosis and management of patients with brain metastases. In: Basow DS editor(s). Uptodate. Waltham: Uptodate, 2011. [Google Scholar]


