Abstract
Fufang Danshen (FFDS), a Chinese medicine formula widely used in the clinic, has proven therapeutic effects on pain relief. However, the mechanisms of these effects have not been elucidated. Here, we performed a systematic analysis to discover the mechanisms of FFDS in attenuating pain to gain a better understanding of FFDS in the treatment of other diseases accompanied by pain. Relevance analysis showed that Salvia miltiorrhizae was the best studied herb in FFDS. Most compounds in FFDS have good bioavailability, and we collected 223 targets for 35 compounds in FFDS. These targets were significantly enriched in many pathways related to pain and can be classified as signal transduction, endocrine system, nervous system and lipid metabolism. We compared Salvia miltiorrhizae and Panax notoginseng and found that they can significantly affect different pathways. Moreover, ten pain disease proteins and 45 therapeutic targets can be directly targeted by FFDS. All 45 therapeutic targets have direct or indirect connections with pain disease proteins. Forty-six pain disease proteins can be indirectly affected by FFDS, especially through heat shock cognate 71 kDa protein (HSPA8) and transcription factor AP-1 (JUN). A total of 109 targets of FFDS were identified as significant targets.
Introduction
Pain, a major symptom related to cancer, inflammation and other diseases, has been defined as an unpleasant sensory and emotional experience associated with actual or potential tissue damage or described in terms of such damage by the International Association for the Study of Pain (IASP)1. Pain is not always good for us. In a normal state, pain may help us avoid injury, but in a pathological state, it evolves from a symptom indicating tissue damage to a disease itself 2. The mechanisms accounting for pain have not yet been fully elucidated. The discovery of neurons and their roles in pain3 invalidated many theories related to pain. Currently, the specificity or labeled line theory and gate control theory are the most controversial topics. According to different characteristics, pain can be classified into various types, such as acute pain, chronic pain, inflammatory pain, and neuropathic pain. Clinical pain is a serious public health issue. As the primary drugs, opioids and nonsteroidal anti-inflammatory drugs (NSAIDS) are the most widely used in the treatment of pain. However, these drugs have many severe adverse effects that have often been observed in a large number of patients. Common side effects of opioids include constipation, nausea, vomiting, respiratory depression, and urinary retention. Common side effects of NASIDS include injury to the gastrointestinal tract, liver and kidney dysfunction, and hematological system damage. All of these factors have necessitated the development of alternative analgesics4. Although many new analgesics have been introduced to the clinic for the treatment of pain in the past decades, we cannot deny the lack of real breakthrough drugs in clinical pain control5. Improvements have been made in our understanding of pain mechanisms, and many therapeutic targets and disease proteins related to pain have been found6. However, there may be more undiscovered successful drugs for treating various types of pain.
Traditional Chinese medicine (TCM) is an important part of world medicine. Despite its unknown molecular mechanisms, the therapeutic effects of TCM on curing diseases are recognized by hundreds and thousands of people. TCM is an important complementary and alternative medicine accepted by 183 countries and regions worldwide. The most famous theory of TCM is the balance-regulation theory, which emphasizes the integrity of the human body as well as the interaction between human individuals and their environment7. Scientists have studied herbal medicines and found that more than 800 types of TCM are effective in relieving pain8. Fufang Danshen (FFDS), recorded in Chinese Pharmacopoeia (2015), has been used clinically to treat coronary arteriosclerosis, angina pectoris, hyperlipemia and Alzheimer’s disease in China and is also available as a dietary supplement or drug in other countries9. FFDS comprises three herbs, including Salvia miltiorrhizae (Danshen) as a Jun drug (monarch drug), Panax notoginseng (Sanqi) as a Chen drug (ministerial drug), and Borneolum (Bing Pian) as Zuo and Shi drugs (adjuvant drug and messenger drug). In TCM, herbal formulas are organized based on the rule of “Jun-Chen-Zuo-Shi” to synergize therapeutic effects and integrally minimize adverse effects10,11. Many studies have proven that FFDS has many biological functions, including relieving pain, promoting blood circulation, improving reduced blood flow, reducing blood lipids, protecting blood vessels and myocardium, and improving heart function12–14. Herbs in this formula were recently found to affect other diseases, such as cancer, and osteoporosis15,16. Although many studies have demonstrated the significant therapeutic effects of FFDS on attenuating neuropathic pain, cancer pain, osteoarthritis pain, migraine and angina pectoris, few studies have been conducted to uncover the mechanisms. As pain is a common, severe symptom related to diseases that FFDS can cure, uncovering the mechanisms of this formula in treating pain will provide a better understanding of FFDS in the treatment of those diseases.
Network pharmacology is an approach to drug design that encompasses systems biology, network analysis, connectivity, redundancy and pleiotropy17. Network pharmacology is recognized as a new strategy and powerful tool for the exploration of drug targets and the identification of potentially active ingredients in TCM research18,19. In the present study, we conducted a bioinformatics investigation to elucidate the multilevel mechanisms of FFDS in attenuating pain. This investigation includes the following main steps: (1) assess the relevance of FFDS and its herbs with pain; (2) drug-likeness and bioavailability analysis of compounds in FFDS; (3) collection of targets for FFDS; and (4) analysis of the potential pharmacological mechanisms of FFDS in the treatment of pain. Compared with other TCM network pharmacology approaches, such as Li’s20, our approach focused on uncovering potential pathways and processes that a herbal formula affects to cure a disease. Similar steps include preparing data (compounds in a formula, target collection or prediction for a formula, disease proteins and therapeutic targets or proteins involved in disease-related pathways, and protein-protein interaction (PPI) data), analyzing networks and discovering the potential pathways or processes that the formula affects to cure a specific disease. Most TCM network pharmacology studies or informatics investigations focus on uncovering the mechanisms of action of an entire formula and ignore the discovery of active compounds in the formula (with the exception of Li21, who has introduced a novel method to predict active compounds in a formula), which will hinder the modernization of TCM. In our investigation, we used the “drug-target” principle and “essential protein” theory, which suggests that “a highly connected protein is more important” to uncover potentially active compounds for the treatment of pain. Our results successfully uncovered some active compounds that have experimentally proven antinociceptive effects. In addition, we analyzed disease proteins and therapeutic targets separately since they are different concepts, while most studies analyze them as a whole. Disease proteins are products of disease genes, which are the main factors that can cause diseases, but therapeutic targets are important factors that can cure diseases. We also evaluated therapeutic targets based on their degrees in the drug-target network, and we suggested that therapeutic targets with higher degrees were more likely or more efficient therapeutic targets for a specific disease. In addition, we discussed the relevance of FFDS and its herbs with pain based on text mining. The discussion covers the extent to which FFDS and its herbs have been associated with pain and which herb is more efficient for the treatment of pain. Based on these TCM network pharmacology approaches, we will have a clearer, further understanding of the pharmacological mechanisms of FFDS in attenuating pain.
Results
FFDS, its herbs and pain
As shown in Table 1, Salvia miltiorrhizae (43 papers) is the best studied herb in FFDS due to its importance in TCM, followed by Panax notoginseng (13 papers). Pain is a major, common symptom related to various diseases; thus, the volume of research on pain is extensive (599207). The number of papers relating FFDS or its herbs to pain is very small, and the P-value for FFDS and Panax notoginseng was nonsignificant, which may be because western medicine is more efficient and convenient than TCM in the treatment of pain. Additionally, TCM is preferred for use in complex diseases, such as cancer and Alzheimer’s disease. The study of TCM in the treatment of pain can provide a better understanding of its efficacy in the treatment of other diseases accompanied by pain and aid in the development of new analgesics from TCM.
Table 1.
Relevance between FFDS, its herbs and pain.
| Herb | Total | Relevant to diseases | Relevant to pain | Ratio | P-value |
|---|---|---|---|---|---|
| Fufang Danshen | 38 | 15 | 1 | 6.67% | 0.2858 |
| Salvia miltiorrhizae | 2296 | 1105 | 43 | 3.89% | 0.0049 |
| Panax notoginseng | 1031 | 410 | 13 | 3.17% | 0.1291 |
| Borneolum | 25 | 16 | 3 | 18.75% | 0.0004 |
The ratio is the volume of papers relevant to pain/the volume of papers relevant to diseases.
Drug-likeness and bioavailability analysis for compounds in FFDS
Ultimately, we obtained 123 compounds for Salvia miltiorrhizae (Danshen), 78 compounds for Panax notoginseng (Sanqi), and 20 compounds for Borneolum (Bing Pian). Dauricine and gamma-Sitosterol are shared by Salvia miltiorrhizae and Panax notoginseng; Bata-Caryophyllene and Elemicin are shared by Panax notoginseng and Borneolum. By contrast, the other compounds belong to only one of the three herbs. In total, 217 compounds were obtained for FFDS. The detailed information is listed in Table S1. The statistics for the six main properties of all compounds in FFDS are shown in Table 2. As shown, the mean and median values of properties of compounds in FFDS conform to “the rule of five”, and most (173/217) of the ingredients in FFDS conform to drug-likeness rules. A bioavailability score was also obtained to evaluate the bioavailability of ingredients in FFDS. The result showed that 75.6% of ingredients have good bioavailability. Conforming to the predictions, the poor bioavailability of ginsenosides and salvianolic acids has been proven.
Table 2.
Statistics of the six main properties of compounds in FFDS.
| Descriptors | Max value | Min value | Mean value | Median value |
|---|---|---|---|---|
| TPSA | 447.21 | 0 | 105.52 | 67.51 |
| Num. H-bond acceptors | 27 | 0 | 6.28 | 4 |
| Num. H-bond donors | 18 | 0 | 3.29 | 1 |
| Num. rotatable bonds | 21 | 0 | 4.82 | 2 |
| MLOGP | 6.92 | −6.15 | 1.46 | 1.48 |
| Molecular weight (MW) | 1269.46 | 44.1 | 402.74 | 311.33 |
FFDS targets and their functions
A total of 223 targets were retained for 35 compounds, with 99 targets belonging to Salvia miltiorrhizae, 143 targets to Panax notoginseng and 17 targets to Borneolum. Among the 35 compounds, 18 compounds belong to Salvia miltiorrhizae, 17 belong to Panax notoginseng, and 2 belong to Borneolum. Based on these two perspectives, we concluded that the rule of “Jun-Chen-Zuo-Shi” is reasonable to organize herbs in a formula. Detailed information is available in Table S2. Salvia miltiorrhizae and Panax notoginseng shared 24 targets; Salvia miltiorrhizae and Borneolum shared 7 targets; Panax notoginseng and Borneolum shared 8 targets; and cytochrome P450 3A4 (CYP3A4), high mobility group protein B1 (HMGB1), and caspase-3 (CASP3) belong to the three herbs. Seventy-one targets belong to only Salvia miltiorrhizae, and these targets were significantly enriched in steroid hormone biosynthesis, HIF-1 signaling pathway, Jak-STAT signaling pathway and PI3K-Akt signaling pathway. These four pathways have relationships with pain and may constitute the mechanisms of Salvia miltiorrhizae in the treatment of pain. For example, steroids can affect the nervous system and are of particular interest in the modulation of pain22,23; molecules targeting the Jak-STAT signaling cascade are successful even though the specific contribution of this pathway in the modulation of pain is unknown24. Additionally, Jak-STAT and PI3K-Akt are important microglial intracellular signaling cascades that are essential for neuropathic pain development and maintenance25. A total of 114 targets belong to only Panax notoginseng, and they were significantly enriched in 13 pathways related to pain, including arachidonic acid metabolism, adipocytokine signaling pathway, tumor necrosis factor (TNF) signaling pathway, serotonergic synapse, inflammatory mediator regulation of TRP channels, Toll-like receptor signaling pathway, linoleic acid metabolism, oxytocin signaling pathway, NF-kappa B signaling pathway, MAPK signaling pathway, GnRH signaling pathway, mTOR signaling pathway and vascular endothelial growth factor (VEGF) signaling pathway, which were ranked by P-values. Leptin, an adipocytokine, plays an important role in nociceptive behavior induced by nerve injury26. Modulators of the arachidonic acid cascade have been the focus of research on treatments for inflammation and pain for several decades, and the design and development of multitarget inhibitors of this pathway that exhibit improved efficacy and less undesired side effects is a new paradigm27. The targets belonging to only Salvia miltiorrhizae or Panax notoginseng were also involved in pathways significantly enriched by targets belonging to the opposite herb only. For example, the targets of Panax notoginseng were involved in the HIF-1 signaling pathway and PI3K-Akt signaling pathway, which were significantly enriched by targets of Salvia miltiorrhizae. Additionally, targets of Salvia miltiorrhizae were involved in the TNF signaling pathway and NF-kappa B signaling pathway, which were significantly enriched by targets of Panax notoginseng. Five targets belong to only Borneolum, while 12 targets are shared with Salvia miltiorrhizae or Panax notoginseng. Five unique targets, including UDP-glucuronosyltransferase 2B10 (UGT2B10), UDP-glucuronosyltransferase 2B11 (UGT2B11), Nuclear factor erythroid 2-related factor 2 (NFE2L2), NAD(P)H dehydrogenase [quinone] 1 (NQO1) and Prostacyclin receptor (PTGIR), may explain why Borneolum is necessary for the formula. UGT2B10, UGT2B11 and NQO1 play important roles in the elimination of toxic materials. NFE2L2 is a transcription activator that binds to antioxidant response elements (AREs) in the promoter regions of target genes and is important for the coordinated upregulation of genes in response to oxidative stress. FFDS has further potential to exert therapeutic effects through targets with at least two herbs or compounds; thus, we retained 51 targets with at least two herbs or compounds in Table S3. Thirteen targets can interact with at least three compounds, and CASP3 is targeted by the most compounds (7), followed by CYP3A4 (4). Of note, Salvia miltiorrhizae and Panax notoginseng possess the most targets and most of the same targets, indicating that they are major herbs in FFDS. However, Borneolum is also essential for its unique targets.
Subsequently, we performed enrichment analysis for 223 FFDS targets, and pathways with P-value <= 0.01 and Fold Enrichment >= 1.5 were retained to find their relationships with pain. A total of 26 pathways may have relationships with pain. Among the 26 pathways, fourteen pathways with Bonferroni <= 0.01 were retained in Table 3. The other 12 pathways include the ErbB signaling pathway, prolactin signaling pathway, oxytocin signaling pathway, insulin signaling pathway, sphingolipid signaling pathway, neurotrophin signaling pathway, GnRH signaling pathway, cholinergic synapse, Jak-STAT signaling pathway, inflammatory mediator regulation of TRP channels, estrogen signaling pathway and AMPK signaling pathway. These pathways were also significantly enriched by targets with at least two compounds except for the adipocytokine signaling pathway, NF-kappa B signaling pathway, ErbB signaling pathway, GnRH signaling pathway, Jak-STAT signaling pathway, inflammatory mediator regulation of TRP channels and AMPK signaling pathway. Among the 26 pathways, 16 pathways were also enriched by 24 targets shared by Salvia miltiorrhizae and Panax notoginseng, including the TNF signaling pathway, VEGF signaling pathway, apoptosis, HIF-1 signaling pathway, toll-like receptor signaling pathway, neurotrophin signaling pathway, sphingolipid signaling pathway, insulin signaling pathway, PI3K-Akt signaling pathway, prolactin signaling pathway, MAPK signaling pathway, estrogen signaling pathway, cholinergic synapse, serotonergic synapse, mTOR signaling pathway and ErbB signaling pathway. These 16 pathways may constitute the same major functions of Salvia miltiorrhizae and Panax notoginseng in the treatment of pain. Inhibition of tumor necrosis factor-alpha (TNF-α) can reduce inflammation and pain28. Toll-like receptors are now recognized to contribute to the chronic pain process29. Moreover, myocyte apoptosis was detected as a possible promoter of pain and motor dysfunction in neuropathic rats30. These 26 pathways can be classified into the following categories: environmental information processing, organismal systems, metabolism and cellular processes. Pathways in signal transduction (11) account for the most pathways. Pain is transmitted by neurons and nerve conduction. The nerve conduction of pain can be classified into four cascades, including pain sensing of the nociceptive receptor, pain transmission of the primary afferent fiber, dorsal horn of the spinal cord, spinal cord-fasciculus thalamicus and other ascending tracts, including pain integration in the cortical and limbic systems, descending control and pain modulation of neurotransmitters. Therefore, there is no doubt that many pathways in signal transduction are involved in pain signal transduction, and these pathways represent therapeutic targets in the treatment of pain. Pathways in the endocrine system (6) account for the second highest number of pathways. Many painful conditions appear to be induced, reduced, and in some cases, modulated by hormones; additionally, knowledge of the role of the endocrine system in chronic pain mechanisms is slowly increasing in experimental and clinical studies31. Pathways in the nervous system (3) and lipid metabolism (3) account for the third highest number of pathways. Pain is a nervous system disease. Specialized pro-resolving lipid mediators have a function in pain32. Analgesic lipid mediators include enzyme pathways, such as endogenous agonists of cannabinoid receptors (endocannabinoids), lipid-amide agonists of peroxisome proliferator-activated receptor-α, and products of oxidative metabolism of polyunsaturated fatty acids via cytochrome P450. These lipid messengers are produced and act at different stages of the response to tissue injury and may be part of a peripheral gating mechanism that regulates the access of nociceptive information to the spinal cord and brain33.
Table 3.
Fourteen pathways related to pain are significantly enriched by 223 targets of FFDS.
| KEGG pathway | Count | P-Value | Fold Enrichment | Bonferroni | Class |
|---|---|---|---|---|---|
| TNF signaling pathway | 23 | 2.56E-14 | 8.15 | 6.13E-12 | Environmental information processing; signal transduction |
| Arachidonic acid metabolism | 16 | 3.91E-11 | 9.69 | 9.34E-09 | Metabolism; lipid metabolism |
| HIF-1 signaling pathway | 18 | 5.19E-10 | 6.90 | 1.24E-07 | Environmental information processing; signal transduction |
| Toll-like receptor signaling pathway | 18 | 1.84E-09 | 6.38 | 4.40E-07 | Organismal systems; immune system |
| Adipocytokine signaling pathway | 15 | 2.68E-09 | 8.05 | 6.42E-07 | Organismal systems; endocrine system |
| Linoleic acid metabolism | 10 | 3.34E-08 | 12.95 | 7.98E-06 | Metabolism; lipid metabolism |
| Serotonergic synapse | 16 | 1.83E-07 | 5.41 | 4.38E-05 | Organismal systems; nervous system |
| PI3K-Akt signaling pathway | 28 | 3.12E-07 | 3.05 | 7.42E-05 | Environmental information processing; signal transduction |
| Apoptosis | 12 | 5.22E-07 | 7.27 | 1.25E-04 | Cellular processes; cell growth and death |
| Steroid hormone biosynthesis | 11 | 2.30E-06 | 7.12 | 5.49E-04 | Metabolism; lipid metabolism |
| NF-kappa B signaling pathway | 13 | 2.60E-06 | 5.61 | 6.21E-04 | Environmental information processing; signal transduction |
| VEGF signaling pathway | 11 | 3.71E-06 | 6.77 | 8.86E-04 | Environmental information processing; signal transduction |
| mTOR signaling pathway | 10 | 1.83E-05 | 6.47 | 4.36E-03 | Environmental information processing; signal transduction |
| MAPK signaling pathway | 20 | 4.03E-05 | 2.95 | 9.58E-03 | Environmental information processing; signal transduction |
The relationships of herbs, compounds and FFDS targets with pain disease proteins
We built the FFDS targets-other human proteins PPI network and pain disease proteins-other human proteins PPI network. We used 141 pain disease proteins (Table S4) and 223 FFDS targets to identify FFDS-related pain disease proteins. Pain disease proteins that can be directly targeted by FFDS were treated as FFDS primary pain disease proteins. Pain disease proteins that can directly interact with FFDS targets but cannot be directly targeted by FFDS were treated as FFDS secondary pain disease proteins. FFDS primary and secondary pain disease proteins can be defined as FFDS-related pain disease proteins. Fifty-six FFDS-related pain disease proteins (Table S5) were identified, including 10 primary pain disease proteins and 46 secondary pain disease proteins. The primary pain disease proteins include catechol O-methyltransferase (COMT), endothelin-1 (EDN1), interleukin-2 (IL2), RAC-alpha serine/threonine-protein kinase (AKT1), neuronal acetylcholine receptor subunit alpha-7 (CHRNA7), prostaglandin G/H synthase 2 (PTGS2), serum albumin (ALB), protachykinin-1 (TAC1), oxytocin-neurophysin 1 (OXT) and TNF. Five primary pain disease proteins have direct interactions with the pain disease protein set, including AKT1, ALB, PTGS2, TNF and COMT, and the minimum path lengths are 2 between any of the other primary pain disease proteins and the pain disease protein set except for IL2 (3). Two proteins may have no direct interaction, but they may have indirect connections through other human proteins called intermediators, which form several paths to connect these proteins. The minimum path length can show whether a protein has close interaction or distant interaction with a protein set (without that protein). AKT1 and ALB can interact with most pain disease proteins. Among the 46 secondary pain disease proteins, 27 proteins have direct interactions with the pain disease protein set, and the minimum path lengths between any of the other 17 secondary pain disease proteins and the pain disease protein set (except for the proteins themselves) are 2, and the minimum path lengths are 3 and 4 for the remaining two secondary pain disease proteins. Among the 27 secondary pain disease proteins that have direct interactions with the pain disease protein set, high affinity nerve growth factor receptor (NTRK1) can interact with the most pain disease proteins (14) directly, followed by transitional endoplasmic reticulum ATPase (VCP) (11), heterogeneous nuclear ribonucleoprotein A1 (HNRNPA1) (9) and RNA-binding protein FUS (FUS) (8). These results suggest that all 56 FFDS-related pain disease proteins have direct or indirect connections with the pain disease protein set except for themselves.
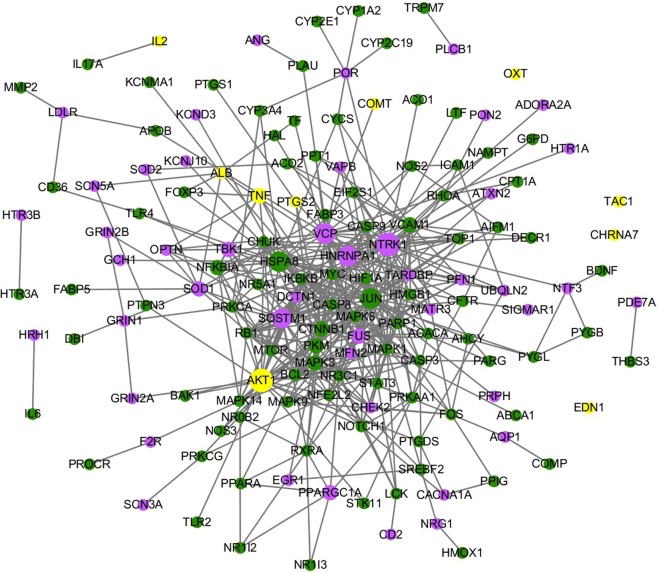
Six primary pain disease proteins belong to Panax notoginseng, 3 belong to Salvia miltiorrhizae, and AKT1 is shared by Panax notoginseng and Salvia miltiorrhizae. AKT1 can be targeted by three compounds, while COMT, EDN1 and IL2 can be targeted by two compounds in Salvia miltiorrhizae. PTGS2 can be targeted by two compounds in Panax notoginseng, and CHRNA7, ALB, TAC1, OXT and TNF can be targeted by one compound in Panax notoginseng. AKT1 is involved in the PI3K-Akt signaling pathway. IL2 is a cytokine; cytokines play an important role in pain through different mechanisms in several sites of pain transmission pathways34. PTGS2 inhibitors have been widely used to treat inflammation and pain35,36. As shown in Fig. 1, we built a network for the 56 FFDS-related pain disease proteins and their interactive FFDS targets. AKT1 is a FFDS primary pain disease protein, and it interacts with the most FFDS targets (19), followed by TNF (5). Among the 46 secondary pain disease proteins, NTRK1 is associated with most FFDS targets (23), followed by VCP (19), HNRNPA1 (16) and sequestosome-1 (SQSTM1) (14). NTRK1 and VCP can be indirectly targeted by 13 compounds in the three herbs, HNRNPA1 can be indirectly targeted by 12 compounds in three herbs, and SQSTM1 can be targeted by 11 compounds in three herbs; all of these proteins are associated with the highest number of compounds. Nerve growth factor (NGF) is a neurotrophic factor that acts as a mediator of pain, and NTRK1 encodes a receptor tyrosine kinase for NGF37. In addition, heat shock cognate 71 kDa protein (HSPA8) and transcription factor AP-1 (JUN) are FFDS targets instead of pain disease proteins, but they are associated with most pain disease proteins. Thus, HSPA8 and AP-1 may be factors that contribute to the therapeutic effects of FFDS in the treatment of pain. HSPA8 is implicated in a wide variety of cellular processes, including the protection of the proteome from stress, folding and transport of newly synthesized polypeptides, activation of proteolysis of misfolded proteins and the formation and dissociation of protein complexes. JUN directly interacts with specific target DNA sequences to regulate gene expression.
Figure 1.
The network for 56 FFDS-related pain disease proteins and their interactive FFDS targets. Green nodes represent FFDS targets, blue nodes represent pain disease proteins, and yellow nodes represent FFDS targets/pain disease proteins.
Salvia miltiorrhizae possesses 36 pain disease proteins, and four of these proteins can be directly targeted by it. Panax notoginseng possesses 49 pain disease proteins, and seven of these proteins can be directly targeted by it. Borneolum possesses 12 pain disease proteins, and none of them can be directly targeted by it. Twenty-eight compounds in FFDS have direct or indirect interactions with pain disease proteins. Eicosatetraenoic acid in Salvia miltiorrhizae can directly target the most pain disease proteins (5) and is directly or indirectly related to the most pain disease proteins (29). Polysaccharide, ursolic acid, hexadecanoic acid, baicalin, rosmarinic acid and oleanolic acid are directly or indirectly related to at least ten pain disease proteins. We retrieved the literature in PubMed and found that 13 compounds in FFDS have antinociceptive effects. These compounds include ursolic acid, baicalin, rosmarinic acid, ferulic acid, caffeic acid, tanshinone IIA, protocatechuic acid, tetramethylpyrazine and gamma-sitosterol in Salvia miltiorrhizae; polysaccharide, ginsenoside Rg1 and ginsenoside Rg3 in Panax notoginseng; and oleanolic acid in Borneolum. For example, ursolic acid has anti-inflammatory, antioxidant, and antinociceptive effects in different animal models38. Polysaccharide pretreatment significantly reduced the number of writhes and licking time but did not increase the latency time of responses, demonstrating its antinociceptive effects39,40. Orally administered oleanolic acid showed an antinociceptive effect in a dose-dependent manner as measured in the acetic acid-induced writhing test41. We used a scoring system to assess herbs and compounds in FFDS that affect pain disease proteins and found that 13 compounds (ursolic acid:3.65, polysaccharide:3.13, baicalin:2.17, oleanolic acid:1.78, rosmarinic acid:1.33, ferulic acid:1.08, caffeic acid:1.04, tanshinone IIA:0.92, protocatechuic acid:0.76, tetramethylpyrazine:0.73, gamma-Sitosterol:0.52, ginsenoside Rg3:0.28 and ginsenoside Rg1:0.22. P <= 0.01) with known antinociceptive effects had scores. Additionally, 15 other compounds (eicosatetraenoic acid:4.90, hexadecanoic acid:3.21, ginsenoside Rd:1.96, ginsenoside Rg2:1.60, tanshinone I:1.07, ginsenoside Re:0.79, pentadecanoic acid: 0.71, cryptotanshinone:0.63, magnesium lithospermate B:0.44, ginsenoside Rh1:0.29, dauricine:0.17, miltirone:0.17, isoferulic acid:0.16, Danshensu:0.12 and asiatic acid:0.03; P <= 0.01 except for Dauricine and miltirone (P = 0.029)) in FFDS had scores. The maximum score, mean score and minimum score of the 13 known compounds are 3.65, 1.36 and 0.22, respectively, and the maximum score, mean score and minimum score of the 15 unknown compounds are 4.90, 1.08 and 0.03, respectively. We compared unknown compounds with known compounds by using t-tests and found no significant difference in the score values between them (P = 0.57), indicating that these unknown compounds are of equal importance in the treatment of pain. Six known compounds could directly target pain disease proteins. At least two targets (including pain disease proteins) of each of the 13 known compounds have direct associations with pain disease proteins. The percentages (number of targets directly associated with pain disease proteins (including pain disease proteins)/all targets of a compound) of 10 known compounds exceed 0.5. Additionally, the scores of 11 known compounds exceed 0.5. To improve prediction accuracy, we required satisfying at least two of the following conditions: directly targeting a pain disease protein, at least 2 targets with direct associations with pain disease proteins (including pain disease proteins), percentage >= 0.5 and score >= 0.5 to determine that a compound has effects in the treatment of pain. Indeed, all known compounds can be determined to have effects in treating pain by using this method. This method has been proven reliable in screening active compounds from herbal formulas. Using this method, eicosatetraenoic acid, hexadecanoic acid, ginsenoside Rd, ginsenoside Rg2, tanshinone I, ginsenoside Re, pentadecanoic acid, cryptotanshinone, magnesium lithospermate B, isoferulic acid, danshensu and asiatic acid can be determined to have effects in treating pain. These compounds and the 13 known compounds may be the main active ingredients of FFDS in treating pain. Relevant information about these compounds and the 13 known compounds is listed in Table 4. However, scores always show the comprehensive relevance of compounds to pain. The scores of eicosatetraenoic acid, hexadecanoic acid, ginsenoside Rd, ginsenoside Rg2, tanshinone I, ginsenoside Re and pentadecanoic acid in the 15 unknown compounds exceed 0.7, and these compounds have more potential as drugs for pain. In fact, the scores of 7/15 unknown compounds exceed 0.7, while the scores of 10/13 known compounds exceed 0.7.
Table 4.
Relevance information about herbs and compounds (determined to have effects in treating pain) with regard to pain.
| Herb/compound | n | k | Number of directly targeted pain disease proteins | Percentage (k/n) | Sum score | Max score | P-Value | Type |
|---|---|---|---|---|---|---|---|---|
| Salvia miltiorrhizae (herb) | 99 | 58 | 4 | 0.586 | 14.97 | 0.54 | 0.00 | known |
| Ursolic acid | 16 | 13 | 1 | 0.812 | 3.65 | 0.54 | 0.00 | known |
| Baicalin | 23 | 13 | 0 | 0.565 | 2.17 | 0.42 | 9.55E-15 | known |
| Rosmarinic acid | 7 | 6 | 1 | 0.857 | 1.33 | 0.37 | 4.23E-09 | known |
| Ferulic acid | 16 | 7 | 0 | 0.438 | 1.08 | 0.36 | 1.64E-07 | known |
| Tanshinone I | 7 | 5 | 0 | 0.714 | 1.07 | 0.45 | 4.25E-07 | unknown |
| Caffeic acid | 20 | 5 | 1 | 0.250 | 1.04 | 0.40 | 2.28E-04 | known |
| Tanshinone IIA | 11 | 8 | 1 | 0.727 | 0.93 | 0.22 | 8.05E-11 | known |
| Protocatechuic acid | 4 | 3 | 1 | 0.750 | 0.76 | 0.43 | 9.75E-05 | known |
| Tetramethylpyrazine | 3 | 2 | 0 | 0.667 | 0.73 | 0.49 | 2.51E-3 | known |
| Cryptotanshinone | 4 | 3 | 1 | 0.750 | 0.63 | 0.35 | 9.75E-05 | unknown |
| Gamma-Sitosterol | 4 | 4 | 0 | 1.000 | 0.52 | 0.15 | 7.28E-07 | known |
| Magnesium lithospermate B | 5 | 3 | 1 | 0.600 | 0.44 | 0.19 | 2.39E-04 | unknown |
| Isoferulic acid | 3 | 2 | 0 | 0.667 | 0.16 | 0.12 | 2.51E-03 | unknown |
| Danshensu | 2 | 2 | 0 | 1.000 | 0.12 | 0.06 | 8.54E-04 | unknown |
| Panax notoginseng (herb) | 143 | 43 | 7 | 0.301 | 17.78 | 0.67 | 0.00 | known |
| Eicosanetetraenoic acid | 39 | 16 | 5 | 0.410 | 4.90 | 0.67 | 5.00E-15 | unknown |
| Hexadecanoic acid | 48 | 15 | 2 | 0.313 | 3.21 | 0.63 | 4.15E-12 | unknown |
| Polysaccharide | 14 | 14 | 0 | 1.000 | 3.13 | 0.53 | 0.00 | known |
| Ginsenoside Rd | 3 | 3 | 1 | 1.000 | 1.96 | 0.62 | 2.49E-05 | unknown |
| Ginsenoside Rg2 | 3 | 3 | 0 | 1.000 | 1.60 | 0.36 | 2.49E-05 | unknown |
| Ginsenoside Re | 4 | 4 | 0 | 1.000 | 0.79 | 0.39 | 7.28E-07 | unknown |
| Pentadecanoic acid | 2 | 2 | 0 | 1.000 | 0.71 | 0.33 | 8.54E-04 | unknown |
| Gamma-Sitosterol | 4 | 4 | 0 | 1.000 | 0.52 | 0.15 | 7.28E-07 | known |
| Ginsenoside Rg3 | 4 | 3 | 0 | 0.750 | 0.28 | 0.13 | 9.75E-05 | known |
| Ginsenoside Rg1 | 5 | 2 | 1 | 0.400 | 0.22 | 0.14 | 8.05E-03 | known |
| Dauricine | 1 | 1 | 0 | 1.000 | 0.17 | 0.17 | 2.92E-02 | unknown |
| Borneolum (herb) | 17 | 11 | 0 | 0.647 | 1.80 | 0.37 | 1.38E-13 | known |
| Oleanolic acid | 14 | 9 | 0 | 0.643 | 1.78 | 0.37 | 2.70E-11 | known |
| Asiatic acid | 3 | 2 | 0 | 0.667 | 0.03 | 0.03 | 2.51E-03 | unknown |
Lines 2, 17 and 29 are herbal items. Compounds below an herb belong to this herb. The “Materials and methods” section contains definitions for n and k.
Overlap analysis between FFDS targets and drug targets with indications for pain
To explore similarities between FFDS and known pain drugs, we collected known drugs in treating pain as well as their affected targets in humans from the DrugBank and TTD databases. These drug targets can be used as therapeutic targets. Subsequently, we built a drug-target network to explore frequently affected therapeutic targets. If a therapeutic target is affected by more drugs in treating the same disease, this target has more potential as an efficient approach for treating the disease. After we analyzed the networks (Table S6), the Mu-type opioid receptor (OPRM1) was targeted by the most drugs (55), and PTGS2 was targeted by the most approved drugs (14), indicating that they are efficient therapeutic approaches for pain treatment. PTGS2 can be directly targeted by FFDS. Prostaglandin G/H synthase 1 (PTGS1) is associated with 22 drugs, including 8 approved drugs, and can be directly targeted by FFDS.
In total, 45 therapeutic targets are affected by both FFDS and known drugs. All therapeutic targets have direct or indirect connections with the pain disease protein set. Six therapeutic targets are also pain disease proteins, including PTGS2, CHRNA7, COMT, TNF, ALB and OXT, while 24 therapeutic targets are not pain disease proteins but can directly interact with the pain disease proteins. For the remaining 15 therapeutic targets, the minimum path lengths between each target and pain disease proteins are 2. As shown in Fig. 2, we built a network to show the direct connections between the 45 therapeutic targets and pain disease proteins. For the six therapeutic targets/pain disease proteins, ALB can interact with 4 other pain disease proteins, TNF can interact with 2 other pain disease proteins, and PTGS2 and COMT can interact with 1 other pain disease protein. By contrast, CHRNA7 and OXT have no direct interactions with other pain disease proteins. Vascular cell adhesion protein 1 (VCAM1), which is not a pain disease protein, can directly interact with the most pain disease proteins (6), followed by NF-kappa B inhibitor alpha (NFKBIA) (5), mitogen-activated protein kinase 3 (MAPK3) (5) and inhibitor of nuclear factor kappa B kinase subunit beta (IKBKB) (4). Seven therapeutic targets associated with at least four known drugs are listed in Table 5. According to the enrichment results, the 45 therapeutic targets were significantly associated with 13 pain-related pathways, including the TNF signaling pathway, NF-kappa B signaling pathway, apoptosis, HIF-1 signaling pathway, Toll-like receptor signaling pathway, serotonergic synapse, VEGF signaling pathway, arachidonic acid metabolism, adipocytokine signaling pathway, cholinergic synapse, mTOR signaling pathway, sphingolipid signaling pathway and neurotrophin signaling pathway. All of these pathways could be found in the 26 pathways enriched by all FFDS targets. When we compared those pathways enriched by the 45 therapeutic targets and pathways enriched by all FFDS targets, we found that some of the 45 therapeutic targets and other FFDS targets were involved in the same pathways.
Figure 2.

The direct interactions between 45 common therapeutic targets and pain disease proteins. Green nodes represent therapeutic targets, blue nodes represent pain disease proteins, and yellow nodes represent therapeutic targets/pain disease proteins.
Table 5.
The 7 common targets associated with the most drugs known for the treatment of pain.
| Gene | Protein | Compound | Type |
|---|---|---|---|
| PTGS2 | Prostaglandin G/H synthase 2 | Hexadecanoic acid, eicosatetraenoic acid | successful |
| PTGS1 | Prostaglandin G/H synthase 1 | Eicosatetraenoic acid | successful |
| HTR3A | 5-hydroxytryptamine receptor 3A | Ginsenoside Rg3 | successful |
| NR3C1 | Glucocorticoid receptor | Ginsenoside Re | investigational |
| CHRNA7 | Neuronal acetylcholine receptor subunit alpha-7 | Ginsenoside Rg1 | investigational |
| TOP2A | DNA topoisomerase 2-alpha | Ursolic acid, oleanolic acid | investigational |
| ALOX5 | Arachidonate 5-lipoxygenase | Baicalin, caffeic acid, eicosatetraenoic acid | investigational |
Evaluation of FFDS targets and analysis of significant targets
Highly connected proteins in PPIs can be defined as significant proteins. To identify the significant targets of FFDS, we constructed a FFDS targets-pain disease proteins-other human proteins PPI network to evaluate FFDS targets. We calculated the degrees of FFDS targets, and 109 FFDS targets (Table S7) with higher degrees than the median value of the degrees of all FFDS targets in the network can be defined as significant targets of FFDS. The degrees of JUN, Myc proto-oncogene protein (MYC), VCAM1, HSPA8, Catenin beta-1 (CTNNB1) and AKT1 are the highest at 1474, 892, 656, 428, 368 and 314, respectively. VCAM1 and AKT1 are therapeutic targets of drugs with indications for pain and pain disease protein, respectively. Five significant targets (AKT1, ALB, TNF, COMT, and PTGS2) are pain disease proteins, and ALB, TNF, COMT and PTGS2 are also therapeutic targets. There are 25 therapeutic targets except for those four therapeutic targets/pain disease proteins. As shown in Fig. 3, a highly connected subnetwork was extracted from the FFDS targets-pain disease proteins-other human proteins PPI network. The highly connected subnetwork can be defined as a core of FFDS. The highly connected subnetwork includes 29 pain disease proteins or therapeutic targets of drugs with indications for pain. Thus, FFDS may significantly affect pain through this network.
Figure 3.
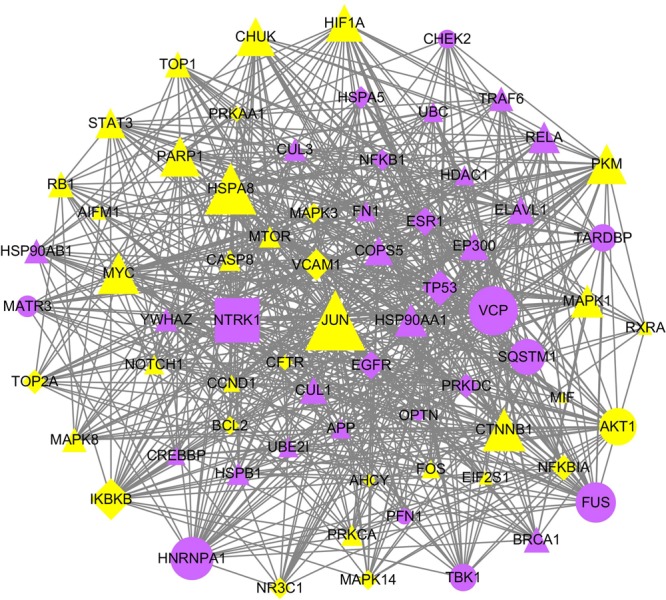
A highly connected subnetwork in the FFDS targets-pain disease proteins-other human proteins network. Triangle nodes correspond to FFDS targets or other human proteins, circle nodes are pain disease proteins, diamond nodes are therapeutic targets, rectangle nodes represent therapeutic targets/pain disease proteins, yellow nodes can be directly targeted by FFDS, and blue nodes can be indirectly affected by FFDS.
Then, we performed pathway enrichment analysis, and 109 significant targets were significantly associated with 28 pathways related to pain. The TNF signaling pathway, NF-kappa B signaling pathway, apoptosis, HIF-1 signaling pathway, Toll-like receptor signaling pathway, serotonergic synapse, VEGF signaling pathway, adipocytokine signaling pathway, cholinergic synapse, mTOR signaling pathway, sphingolipid signaling pathway and neurotrophin signaling pathway were also enriched by all FFDS targets and 45 therapeutic targets. This finding indicates that the significant therapeutic targets are involved in those pathways. The PI3K-Akt signaling pathway, MAPK signaling pathway, ErbB signaling pathway, prolactin signaling pathway, oxytocin signaling pathway, insulin signaling pathway, GnRH signaling pathway, estrogen signaling pathway, AMPK signaling pathway and inflammatory mediator regulation of TRP channels were also enriched by all FFDS targets. Other pathways, including the cAMP signaling pathway, retrograde endocannabinoid signaling, Wnt signaling pathway, dopaminergic synapse and Rap1 signaling pathway, can be classified into environmental information processing (signal transduction) and organismal systems (nervous system). VEGF is a selective endothelial cell mitogen that promotes angiogenesis and increases blood vessel permeability. Inhibiting VEGF provides effective pain relief42. Endocannabinoids modulate neuronal, glial and endothelial cell function and have neuromodulatory, anti-excitotoxic, anti-inflammatory and vasodilatory effects. Endocannabinoids behave as analgesics in acute nociception and clinical pain models, such as inflammation and painful neuropathy43,44.
Discussion
Pain is a severe symptom related to many diseases as well as a disease itself that affects thousands of people. As quality of life has improved and the need for painless treatment is increasing, studies on pain are attracting more attention than ever. Though many new analgesics have been introduced to the clinic, pain remains an issue. Additionally, alternative analgesics are required because of the side effects of currently available analgesics. Chinese herbal formulas are well known for their advantages in curing complex diseases with few side effects. FFDS is a Chinese herbal formula that has significant therapeutic effects on attenuating pain. FFDS has been widely used to treat various diseases. One common symptom among these diseases, such Alzheimer’s disease, is pain; thus, determining the mechanisms of FFDS in attenuating pain will aid in the development of many new therapeutic approaches for patients in pain and provide better understanding of pain in the treatment of other diseases.
In the present study, we conducted a bioinformatics investigation to uncover the potential pharmacological mechanisms of FFDS in the treatment of pain. A large-scale text mining revealed that three herbs in FFDS have effects on pain, and some active compounds in each herb have antinociceptive effects. New drugs that may have analgesic effects were discovered based on drug-target principles, including tanshinone I in Salvia miltiorrhizae, eicosatetraenoic acid, hexadecanoic acid, ginsenoside Rg2 and ginsenoside Rd in Panax notoginseng. The pathways constituting the foundational mechanisms of the effects of FFDS on pain have been enriched by 223 targets of FFDS, and they can be classified into signal transduction, endocrine system, lipid metabolism and nervous system. Salvia miltiorrhizae and Panax notoginseng can significantly affect different pathways related to pain, but they also contribute to the effects of the opposite herb in a corresponding pathway, such as the HIF-1 signaling pathway, PI3K-Akt signaling pathway, TNF signaling pathway and NF-kappa B signaling pathway.
Pain disease proteins and therapeutic targets of drugs with indications for pain are directly or indirectly affected by FFDS, and these proteins may be the main factors that FFDS targets to treat pain. Most pain disease proteins and therapeutic targets are highly connected in the PPI network and can be defined as significant proteins. By comparing their locations in KEGG pathways, we found that most of these proteins were located at the beginning of these pathways or at important points within the pathways. Targeting these proteins can affect an entire pathway and has significant therapeutic effects. Other targets of FFDS can affect many pain disease proteins, which cannot be ignored, such as HSPA8 and JUN. Of course, a single drug affects only a few therapeutic targets. However, a formula can affect more targets due to its complex composition. Except for those disease proteins and therapeutic targets, other targets of FFDS were also involved in the same pathways as those disease proteins and therapeutic targets. These “unnecessary targets” were located in unimportant locations or could be indirectly affected by the disease proteins and therapeutic targets. “Unnecessary targets” may bridge several points to enhance the functions of FFDS or they may be therapeutic targets that have not yet been discovered. Moreover, we speculate that for a pathway related to a disease, affecting only a small part of the pathway is necessary to cure this disease; thus, these “unnecessary targets” are affected by FFDS only to reverse its side effects on the human body. Some “unnecessary targets” may be harmful to the human body; therefore, the formula should be optimized.
Based on the published literature, we can conclude that FFDS has therapeutic effects in the treatment of neuropathic pain, cancer pain and arthralgia. In terms of neuropathic pain, three disease proteins of neuropathic pain can be directly targeted by FFDS, including TNF45, PTGS146 and PTGS247; twenty therapeutic targets of drugs with indications including neuropathic pain can be directly affected by FFDS, such as PTGS2, PTGS1 and 5-hydroxytryptamine receptor 3A (HTR3A), which have all been associated with at least four drugs known for the treatment of neuropathic pain. Additionally, fourteen pathways that have relationships with neuropathic pain therapy can be significantly enriched by targets of FFDS, for example, the TNF signaling pathway45, Toll-like receptor signaling pathway48, PI3K-Akt signaling pathway49 and apoptosis50. No disease proteins for cancer pain can be directly targeted by FFDS, while one therapeutic target (HTR3A) can be directly targeted by FFDS, and two pathways (HIF-1 signaling pathway51 and T cell receptor signaling pathway)52 that have relationships with cancer pain therapy are significantly enriched by FFDS targets. FFDS has proven anticancer properties; in our investigation, many cancer-related proteins and pathways could be affected by FFDS. Thus, many indirect factors should be affected by FFDS to attenuate cancer pain, including proteins that can interact with cancer pain-related proteins. Seven disease proteins for arthralgia can be directly targeted by FFDS, including macrophage migration inhibitory factor (MIF), interleukin-6 (IL6), 72 kDa type IV collagenase (MMP2), Toll-like receptor 4 (TLR4), connective tissue growth factor (CTGF), cartilage oligomeric matrix protein (COMP) and interleukin-10 (IL10). Arthrogenic alphaviruses can cause debilitating illnesses characterized by arthritis and arthralgia, and evidence suggests that both MIF and CD74 play a critical role in mediating alphaviral disease53. In the chronic phase, the level of IL-6 has been associated with persistent arthralgia, providing a possible explanation for the etiology of arthralgia that plagues CHIKV-infected patients54. No therapeutic targets of drugs with indications including arthralgia can be targeted by FFDS. Two pathways (the Toll-like receptor signaling pathway55 and osteoclast differentiation)56 that have relationships with arthralgia can be significantly enriched by FFDS targets. Based on our results, we can conclude that FFDS can treat the aforementioned diseases through a target network, not simply one or two targets. Due to space limitations, we cannot discuss many mechanisms of action of FFDS in the treatment of these diseases. Therefore, we suggest that more investigators should use TCM network pharmacology approaches to unveil these mechanisms. In conclusion, this paper has fully elucidated the potential mechanisms of the effects of FFDS on pain from a systemic perspective based on targets of FFDS and network analysis. Moreover, this study will provide basic theories for the usage of this formula and its herbs.
Materials and Methods
FFDS, its herbs and pain
We referred to an article and used a computational method to assess the relevance between FFDS, its herbs and pain. The more papers relating an herb to pain, the more efficient this herb will be in treating pain. A parameter was used to balance bias and further assess the relevance between FFDS, its herbs and pain. The parameter is the ratio of pain herb-related papers and disease herb-related papers. The P-value was calculated by Eq. (1) and used to examine the error.
| 1 |
where N is the total number of papers in PubMed (27 million), K is the number of papers related to pain (599207, using “Pain” as a keyword), n is the number of papers about FFDS or its herbs and diseases (using synonyms for FFDS or its herbs and disease as keywords), and k is the number of papers about FFDS or its herbs and pain (using synonyms for FFDS or its herbs and pain as keywords). We used synonyms of an item (such as Salvia miltiorrhizae, its synonyms are Salvia miltiorrhizae and Danshen, which are wildly used in papers) to search PubMed and download Pubmed IDs, then we removed duplicate IDs and calculated the total number of the searched papers.
Data preparation
Herbal compounds in FFDS were collected and extracted from TCM database@TW57 (http://tcm.cmu.edu.tw/zh-tw/index.php, updated 2013 July) and PubMed literature. Therapeutic targets and their associated drugs were collected from DrugBank58 (https://www.drugbank.ca/, version 5.0) and Therapeutic Target Database59 (http://bidd.nus.edu.sg/BIDD-Databases/TTD/TTD.asp, last updated on Sep 10, 2015). Pain disease proteins were collected from the Therapeutic Target Database and DisGeNET60 (with score >= 0.2, http://www.disgenet.org, version 5.0). We retained only the drugs (their therapeutic targets) and disease proteins that clearly belonged to “Pain”. PPI data were obtained from Mentha61 (http://mentha.uniroma2.it/). The known targets of compounds in FFDS were collected from STITCH62 (http://stitch.embl.de/, version 5.0) with a higher interaction score (confidence >= 0.7).
Drug-likeness and bioavailability analysis
Compounds with drug-likeness properties and high bioavailability are more likely to be drugs. To explore the drug-likeness and bioavailability of the compounds in FFDS, we used SwissADME (http://www.swissadme.ch/)63,64 as a tool to calculate the drug-likeness and bioavailability of each compound. The drug-likeness rules include Lipinski’s rule, Ghose’s rule, Veber’s rule, Egan’s rule and Muegge’s rule.
Statistical analysis to assess the probabilities of compounds and herbs in FFDS in the treatment of pain
Uncovering active compounds in Chinese herbal formulas for specific diseases can facilitate the modernization of TCM. Here, we built a new computational method to simply assess compounds and herbs in FFDS in treating pain. First, we constructed the pain disease proteins-other human proteins PPI network to evaluate the significance of pain disease proteins for pain. Increased significance of a protein in the network indicates that this protein is more essential for pain65. According to the centrality of a protein in the PPI network, which has a strong correlation with the essentiality of a protein, the protein will be given a weight value Si to show its relative significance. First, we calculated the degrees of pain disease proteins in the pain disease proteins-other human proteins PPI network. Then, we calculated log10 values of degrees. Finally, we divided log10 values by the maximum log10 value for normalization. We used those normalized values as the weight values Si of each pain disease protein. As degrees 1 and 2 are similarly important and log10 of 1 is 0, we changed the weight values of pain disease proteins with degrees 1 or 2 to 0.4 (log10 of 3 is 0.4771) divided by the maximum log10 value. In truth, the degrees of only two pain disease proteins used here were 1 or 2. The probability scores of PPIs were obtained from the Mentha database. The product of the weight value of a pain disease protein and the maximum probability of a specified compound directly or indirectly interacting with that protein will be considered the computational relevance score of the compound with pain through this pain disease protein. The sum of the relevance scores of a compound with regard to all its directly or indirectly interactive pain disease proteins will be treated as the computational relevance score of the compound with pain through all its related pain disease proteins. Ultimately, the sum of relevance scores of all compounds in one herb was treated as the computational relevance score of the herb with pain through all its related pain disease proteins. The score could not represent the real therapeutic effects, but it may show the probabilities and relative significance of herbs and their compounds in the treatment of diseases. Similar to Eq. (1), we also used P-values to examine the possibility of finding that a certain number of proteins were targets of a compound or an herb and had direct associations with pain disease proteins (including pain disease proteins themselves) in at least k proteins by chance. Here, N is the total number of human proteins in UniProt (162191), and K is the number of proteins that have direct associations with pain disease proteins (including pain disease proteins) (4739). n is the number of targets of a compound or an herb, and k is the number of proteins that were targets of a compound or an herb and had direct associations with pain disease proteins (including pain disease proteins).
Network construction and analysis
Many types of networks were built and analyzed to explore the pharmacological mechanisms of the effects of FFDS on pain. The degree was calculated to assess the significance of a protein in a network by NetworkAnalyzer in Cytoscape 3.2. The centrality-lethality rule shows that a highly connected protein is more important to an organism than a poorly connected protein65,66. All networks were visualized and analyzed by Cytoscape 3.2.
Pathway enrichment and analysis
Database for Annotation, Visualization, and Integrated Discovery67 (DAVID, https://david.ncifcrf.gov/, version 6.8) was used as a tool for KEGG pathway enrichment by using default settings. Related pathways are available in the KEGG pathway database68 (KEGG: Kyoto Encyclopedia of Genes and Genomes, http://www.kegg.jp/, release 81.0).
Supplementary information
Acknowledgements
This work was supported by the Natural Science Foundation of China (J1210026), the Doctoral Program of Higher Education of China (20120091110038), and the Open Project of State Key Laboratory of Proteomics (SKLP-O201415). We are grateful for our colleagues’ helpful assistance.
Author Contributions
J.Y. conceived and guided the investigation, Y.S. performed the investigation, and J.Y. and Y.S. contributed to the writing of the manuscript. All authors reviewed the manuscript.
Competing Interests
The authors declare no financial competing interests. Nonfinancial competing interests include PubMed, TCM database@TW, SwissADME, DrugBank, Therapeutic Target Database, Cytoscape, UniProt and KEGG.
Footnotes
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary information accompanies this paper at 10.1038/s41598-019-40694-4.
References
- 1.Bonica JJ, et al. The need of a taxonomy. Pain. 1979;6(3):247–248. doi: 10.1016/0304-3959(79)90046-0. [DOI] [PubMed] [Google Scholar]
- 2.Vardeh D, Mannion RJ, Woolf CJ. Toward a Mechanism-Based Approach to Pain Diagnosis. The Journal of Pain. 2016;17(9):50–69. doi: 10.1016/j.jpain.2016.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Woolf CJ, Ma Q. Nociceptors—Noxious Stimulus Detectors. Neuron. 2007;55(03):353–364. doi: 10.1016/j.neuron.2007.07.016. [DOI] [PubMed] [Google Scholar]
- 4.Li JX, Zhang Y. Emerging drug targets for pain treatment. Eur J Pharmacol. 2012;681(1–3):1–5. doi: 10.1016/j.ejphar.2012.01.017. [DOI] [PubMed] [Google Scholar]
- 5.Kissin I. The development of new analgesics over the past 50 years: a lack of real breakthrough drugs. Anesthesia & Analgesia. 2010;110(3):780–789. doi: 10.1213/ANE.0b013e3181cde882. [DOI] [PubMed] [Google Scholar]
- 6.Kissin I. Scientometrics of drug discovery efforts: pain-related molecular targets. Drug Des Devel Ther. 2015;9:3393–3404. doi: 10.2147/DDDT.S85633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zhang Y, et al. A systems biology-based investigation into the therapeutic effects of Gansui Banxia Tang on reversing the imbalanced network of hepatocellular carcinoma. Sci Rep. 2014;4:4154. doi: 10.1038/srep04154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Du GH, Yuan TY, Du LD, Zhang YX. The Potential of Traditional Chinese Medicine in the Treatment and Modulation of Pain. Advances in Pharmacology. 2016;75:325–361. doi: 10.1016/bs.apha.2016.01.001. [DOI] [PubMed] [Google Scholar]
- 9.Chu Y, et al. The effect of Compound Danshen Dripping Pills, a Chinese herb medicine, on the pharmacokinetics and pharmacodynamics of warfarin in rats. Journal of Ethnopharmacology. 2011;137(3):1457–1461. doi: 10.1016/j.jep.2011.08.035. [DOI] [PubMed] [Google Scholar]
- 10.Yao Y, et al. Deciphering the combination principles of Traditional Chinese Medicine from a systems pharmacology perspective based on Ma-huang Decoction. Journal of Ethnopharmacology. 2013;150(2):619–638. doi: 10.1016/j.jep.2013.09.018. [DOI] [PubMed] [Google Scholar]
- 11.Guo Q, et al. Guizhi-Shaoyao-Zhimu decoction attenuates rheumatoid arthritis partially by reversing inflammation-immune system imbalance. J Transl Med. 2016;14:165. doi: 10.1186/s12967-016-0921-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wang H, Gao X, Zhang B. Tanshinone: An inhibitor of proliferation of vascular smooth muscle cells. Journal of Ethnopharmacology. 2005;99(1):93–98. doi: 10.1016/j.jep.2005.01.057. [DOI] [PubMed] [Google Scholar]
- 13.Fan GW, et al. The anti-inflammatory activities of Tanshinone IIA, an active component of TCM, are mediated by estrogen receptor activation and inhibition of iNOS. J Steroid Biochem Mol Biol. 2009;113(4):275–280. doi: 10.1016/j.jsbmb.2009.01.011. [DOI] [PubMed] [Google Scholar]
- 14.Mao H, et al. Dual effects of lipophilic extract of Salvia miltiorrhiza (Danshen) on catecholamine secretion in cultured bovine adrenal medullary cells. J Ethnopharmacol. 2009;125(1):59–67. doi: 10.1016/j.jep.2009.06.010. [DOI] [PubMed] [Google Scholar]
- 15.Chen X, et al. The Anticancer Properties of Salvia Miltiorrhiza Bunge (Danshen): A Systematic Review. Med Res Rev. 2014;34(4):768–794. doi: 10.1002/med.21304. [DOI] [PubMed] [Google Scholar]
- 16.Guo Y, et al. Salvia miltiorrhiza: An ancient Chinese herbal medicine as a source for anti-osteoporotic drugs. J Ethnopharmacol. 2014;155(3):1401–1416. doi: 10.1016/j.jep.2014.07.058. [DOI] [PubMed] [Google Scholar]
- 17.Hopkins AL. Network pharmacology: the next paradigm in drug discovery. Nat Chem Biol. 2008;4:682–690. doi: 10.1038/nchembio.118. [DOI] [PubMed] [Google Scholar]
- 18.Gao L, et al. Molecular targets of Chinese herbs: a clinical study of hepatoma based on network pharmacology. Sci Rep. 2016;6:24944. doi: 10.1038/srep24944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Xiang H, et al. Yin-Chen-Hao Tang attenuates severe acute pancreatitis in rat: an experimental verification of In silico network target prediction. Front Pharmacol. 2016;7:378. doi: 10.3389/fphar.2016.00378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Liang X, Li H, Li S. A novel network pharmacology approach to analyse traditional herbal formulae: the Liu-Wei-Di-Huang pill as a case study. Mol Biosyst. 2014;10(5):1014–1022. doi: 10.1039/C3MB70507B. [DOI] [PubMed] [Google Scholar]
- 21.Zhang B, Wang X, Li S. An Integrative Platform of TCM Network Pharmacology and Its Application on a Herbal Formula, Qing-Luo-Yin. Evid Based Complement Alternat Med. 2013;2013:456747. doi: 10.1155/2013/456747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lewis SS, et al. Select steroid hormone glucuronide metabolites can cause toll-like receptor 4 activation and enhanced pain. Brain Behav Immun. 2015;44:128–136. doi: 10.1016/j.bbi.2014.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mensah-Nyagan AG, et al. Evidence for a key role of steroids in the modulation of pain. Psychoneuroendocrinology. 2009;34(s1):S169–S177. doi: 10.1016/j.psyneuen.2009.06.004. [DOI] [PubMed] [Google Scholar]
- 24.Busch-Dienstfertig M, González-Rodríguez S. IL-4, JAK-STAT signaling, and pain. Jakstat. 2013;2(4):e27638. doi: 10.4161/jkst.27638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Jin, D., Yang, J. P., Hu, J. H., Wang, L. N. & Zuo, J. L. MCP-1 stimulates spinal microglia via PI3K/Akt pathway in bone cancer pain. 1599, 158–67 (2015). [DOI] [PubMed]
- 26.Tian Y, et al. Leptin enhances NMDA-induced spinal excitation in rats: A functional link between adipocytokine and neuropathic pain. Pain. 2011;152(6):1263–1271. doi: 10.1016/j.pain.2011.01.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Meirer K, Steinhilber D, Proschak E. Inhibitors of the arachidonic acid cascade: interfering with multiple pathways. Basic Clin Pharmacol Toxicol. 2014;114(1):83–91. doi: 10.1111/bcpt.12134. [DOI] [PubMed] [Google Scholar]
- 28.Yamacita-Borin FY. Superoxide anion-induced pain and inflammation depends on TNFα/TNFR1 signaling in mice. Neurosci Lett. 2015;605(25):53–58. doi: 10.1016/j.neulet.2015.08.015. [DOI] [PubMed] [Google Scholar]
- 29.Nicotra L, Loram LC, Watkins LR, Hutchinson MR. Toll-like receptors in chronic pain. Exp Neurol. 2012;234(2):316–329. doi: 10.1016/j.expneurol.2011.09.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Gradl G, et al. A pan-caspase inhibitor reduces myocyte apoptosis and neuropathic pain in rats with chronic constriction injury of the sciatic nerve. Anesthesia & Analgesia. 2013;116(1):216–223. doi: 10.1213/ANE.0b013e31826e0fe0. [DOI] [PubMed] [Google Scholar]
- 31.Aloisi AM, Vodo S, Buonocore M. Pain and thyroid hormones. Neurol Sci. 2013;34(9):1501–1508. doi: 10.1007/s10072-013-1440-7. [DOI] [PubMed] [Google Scholar]
- 32.Serhan CN. Pro-resolving lipid mediators are leads for resolution physiology. Nature. 2014;510(7503):92–101. doi: 10.1038/nature13479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Piomelli D, Hohmann AG, Seybold V, Hammock BD. A lipid gate for the peripheral control of pain. J Neurosci. 2014;34(46):15184–191. doi: 10.1523/JNEUROSCI.3475-14.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.de Oliveira CM, Sakata RK, Issy AM, Gerola LR, Salomão R. Cytokines and pain. Rev Bras Anestesiol. 2011;61(2):255–265. doi: 10.1016/S0034-7094(11)70029-0. [DOI] [PubMed] [Google Scholar]
- 35.Chen L, Yang G, Grosser T. Prostanoids and inflammatory pain. Prostaglandins & Other Lipid Mediators. 2013;104–105:58–66. doi: 10.1016/j.prostaglandins.2012.08.006. [DOI] [PubMed] [Google Scholar]
- 36.Applebaum E, Nackley AG, Bair E, Maixner W, Khan AA. Genetic Variants in Cyclooxygenase-2 Contribute to Post-treatment Pain among Endodontic Patients. J Endod. 2015;41(8):1214–1218. doi: 10.1016/j.joen.2015.04.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Indo Y. Neurobiology of pain, interoception and emotional response: lessons from nerve growth factor-dependent neurons. Eur J Neurosci. 2014;39(3):375–391. doi: 10.1111/ejn.12448. [DOI] [PubMed] [Google Scholar]
- 38.Bhat RA, et al. Effect of ursolic acid in attenuating chronic constriction injury-induced neuropathic pain in rats. Fundam Clin Pharmacol. 2016;30(6):517–528. doi: 10.1111/fcp.12223. [DOI] [PubMed] [Google Scholar]
- 39.Pereira JG, et al. Polysaccharides isolated from Digenea simplex inhibit inflammatory and nociceptive responses. Carbohydr Polym. 2014;108:17–25. doi: 10.1016/j.carbpol.2014.01.105. [DOI] [PubMed] [Google Scholar]
- 40.Batista JA, et al. Polysaccharide isolated from Agardhiella ramosissima: chemical structure and anti-inflammation activity. Carbohydr Polym. 2014;99:59–67. doi: 10.1016/j.carbpol.2013.08.071. [DOI] [PubMed] [Google Scholar]
- 41.Park SH, et al. Mechanisms involved in the antinociceptive effects of orally administered oleanolic acid in the mouse. Arch Pharm Res. 2013;36(7):905–911. doi: 10.1007/s12272-013-0093-7. [DOI] [PubMed] [Google Scholar]
- 42.Lin J, et al. VEGF and its receptor-2 involved in neuropathic pain transmission mediated by P2X2(/)3 receptor of primary sensory neurons. Brain Res Bull. 2010;83(5):284–291. doi: 10.1016/j.brainresbull.2010.08.002. [DOI] [PubMed] [Google Scholar]
- 43.Zogopoulos P, Vasileiou I, Patsouris E, Theocharis SE. The role of endocannabinoids in pain modulation. Fundam Clin Pharmacol. 2013;27(1):64–80. doi: 10.1111/fcp.12008. [DOI] [PubMed] [Google Scholar]
- 44.Piomelli D. More surprises lying ahead. The endocannabinoids keep us guessing. Neuropharmacology. 2014;76(Pt B):228–234. doi: 10.1016/j.neuropharm.2013.07.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Lu Y, et al. TRAF6 upregulation in spinal astrocytes maintains neuropathic pain by integrating TNF-α and IL-1β signaling. Pain. 2014;155(12):2618–2629. doi: 10.1016/j.pain.2014.09.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Lau YM, et al. Cellular sources of cyclooxygenase-1 and -2 up-regulation in the spinal dorsal horn after spinal nerve ligation. Neuropathol Appl Neurobiol. 2014;40(4):452–463. doi: 10.1111/nan.12078. [DOI] [PubMed] [Google Scholar]
- 47.Ma W, St-Jacques B, Duarte PC. Targeting pain mediators induced by injured nerve-derived COX2 and PGE2 to treat neuropathic pain. Expert Opin Ther Targets. 2012;16(6):527–540. doi: 10.1517/14728222.2012.680955. [DOI] [PubMed] [Google Scholar]
- 48.Kuang X, et al. Effects of intrathecal epigallocatechin gallate, an inhibitor of Toll-like receptor 4, on chronic neuropathic pain in rats. Eur J Pharmacol. 2012;676(1–3):51–56. doi: 10.1016/j.ejphar.2011.11.037. [DOI] [PubMed] [Google Scholar]
- 49.Popiolek-Barczyk K, Mika J. Targeting the Microglial Signaling Pathways: New Insights in the Modulation of Neuropathic Pain. Curr Med Chem. 2016;23(26):2908–2928. doi: 10.2174/0929867323666160607120124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Hu, Q., Fang, L., Li, F., Thomas, S. & Yang, Z. Hyperbaric oxygenation treatment alleviates CCI-induced neuropathic pain and decreases spinal apoptosis. 19(7), 920–928 (2015). [DOI] [PubMed]
- 51.Wang, J. Z., Li, L., Pan, L. L. & Chen, J. H. Hypnosis and music interventions (HMIs) inactivate HIF-1: A potential curative efficacy for cancers and hypertension. 85(5), 551–557 (2015). [DOI] [PubMed]
- 52.Börner C, et al. Mechanisms of opioid-mediated inhibition of human T cell receptor signaling. J Immunol. 2009;183(2):882–889. doi: 10.4049/jimmunol.0802763. [DOI] [PubMed] [Google Scholar]
- 53.Herrero, L. J. et al. Macrophage migration inhibitory factor receptor CD74 mediates alphavirus-induced arthritis and myositis in murine models of alphavirus infection. 65(10), 2724–2736 (2013). [DOI] [PMC free article] [PubMed]
- 54.Chow, A. et al. Persistent arthralgia induced by Chikungunya virus infection is associated with interleukin-6 and granulocyte macrophage colony-stimulating factor. 203(2), 149–157 (2011). [DOI] [PMC free article] [PubMed]
- 55.Xu S, Li L, Zhang W, Wang Y. Comparative study of regulation of TLR/TRAF signaling pathway of classic prescriptions that deals with arthralgia syndrome based on relevent theory of prescription and syndrome. Zhongguo Zhong Yao Za Zhi. 2010;35(8):1025–1029. [PubMed] [Google Scholar]
- 56.Phuklia W, et al. Osteoclastogenesis induced by CHIKV-infected fibroblast-like synoviocytes: a possible interplay between synoviocytes and monocytes/macrophages in CHIKV-induced arthralgia/arthritis. Virus Research. 2013;177(2):179–188. doi: 10.1016/j.virusres.2013.08.011. [DOI] [PubMed] [Google Scholar]
- 57.Chen CY. TCM Database@Taiwan: the world’s largest traditional Chinese medicine database for drug screening in silico. Plos One. 2011;6(1):e15939. doi: 10.1371/journal.pone.0015939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Wishart DS, et al. DrugBank: a comprehensive resource for in silico drug discovery and exploration. Nucleic Acids Res. 2006;34(Database issue):D668–72. doi: 10.1093/nar/gkj067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Yang H, et al. Therapeutic target database update 2016: enriched resource for bench to clinical drug target and targeted pathway information. Nucleic Acids Res. 2016;44(D1):D1069–D1074. doi: 10.1093/nar/gkv1230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Piñero J, et al. DisGeNET: a comprehensive platform integrating information on human disease-associated genes and variants. Nucleic Acids Res. 2017;45(D1):D833–D839. doi: 10.1093/nar/gkw943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Calderone A, Castagnoli L, Cesareni G. mentha: a resource for browsing integrated protein-interaction networks. Nat Methods. 2013;10(8):690–691. doi: 10.1038/nmeth.2561. [DOI] [PubMed] [Google Scholar]
- 62.Szklarczyk D, et al. STITCH 5: augmenting protein-chemical interaction networks with tissue and affinity data. Nucleic Acids Res. 2016;44(D1):D380–D384. doi: 10.1093/nar/gkv1277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Daina, A., Michielin, O. & Zoete, V. iLOGP: a simple, robust, and efficient description of n-octanol/water partition coefficient for drug design using the GB/SA approach. 54(12), 3284–3301 (2014). [DOI] [PubMed]
- 64.Daina A, Zoete V. A BOILED-Egg To Predict Gastrointestinal Absorption and Brain Penetration of Small Molecules. Chem Med Chem. 2016;11(11):1117–1121. doi: 10.1002/cmdc.201600182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Jeong H, Mason SP, Barabási AL, Oltvai ZN. Lethality and centrality in protein networks. Nature. 2001;411(6833):41–42. doi: 10.1038/35075138. [DOI] [PubMed] [Google Scholar]
- 66.He X, Zhang J. Why do hubs tend to be essential in protein networks? Plos Genet. 2006;2(6):e88. doi: 10.1371/journal.pgen.0020088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Huang DW, Sherman BT, Lempicki RA. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc. 2009;4(1):44–57. doi: 10.1038/nprot.2008.211. [DOI] [PubMed] [Google Scholar]
- 68.Kanehisa M, Furumichi M, Tanabe M, Sato Y, Morishima K. KEGG: new perspectives on genomes, pathways, diseases and drugs. Nucleic Acids Res. 2017;45(D1):D353–D361. doi: 10.1093/nar/gkw1092. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.