Abstract
Background
Drugs can prevent postoperative nausea and vomiting, but their relative efficacies and side effects have not been compared within one systematic review.
Objectives
The objective of this review was to assess the prevention of postoperative nausea and vomiting by drugs and the development of any side effects.
Search methods
We searched The Cochrane Central Register of Controlled Trials (CENTRAL) (The Cochrane Library, Issue 2, 2004), MEDLINE (January 1966 to May 2004), EMBASE (January 1985 to May 2004), CINAHL (1982 to May 2004), AMED (1985 to May 2004), SIGLE (to May 2004), ISI WOS (to May 2004), LILAC (to May 2004) and INGENTA bibliographies.
Selection criteria
We included randomized controlled trials that compared a drug with placebo or another drug, or compared doses or timing of administration, that reported postoperative nausea or vomiting as an outcome.
Data collection and analysis
Two authors independently assessed trial quality and extracted outcome data.
Main results
We included 737 studies involving 103,237 people. Compared to placebo, eight drugs prevented postoperative nausea and vomiting: droperidol, metoclopramide, ondansetron, tropisetron, dolasetron, dexamethasone, cyclizine and granisetron. Publication bias makes evidence for differences among these drugs unreliable. The relative risks (RR) versus placebo varied between 0.60 and 0.80, depending upon the drug and outcome. Evidence for side effects was sparse: droperidol was sedative (RR 1.32) and headache was more common after ondansetron (RR 1.16).
Authors' conclusions
Either nausea or vomiting is reported to affect, at most, 80 out of 100 people after surgery. If all 100 of these people are given one of the listed drugs, about 28 would benefit and 72 would not. Nausea and vomiting are usually less common and, therefore, drugs are less useful. For 100 people, of whom 30 would vomit or feel sick after surgery if given placebo, 10 people would benefit from a drug and 90 would not. Between one to five patients out of every 100 people may experience a mild side effect, such as sedation or headache, when given an antiemetic drug. Collaborative research should focus on determining whether antiemetic drugs cause more severe, probably rare, side effects. Further comparison of the antiemetic effect of one drug versus another is not a research priority.
Keywords: Humans, Antiemetics, Antiemetics/therapeutic use, Postoperative Nausea and Vomiting, Postoperative Nausea and Vomiting/prevention & control, Randomized Controlled Trials as Topic
Drugs for preventing nausea and vomiting after surgery
We found eight drugs that reliably prevented nausea or vomiting after surgery. The drugs prevented nausea or vomiting in three or four people out of every 10 who would have vomited or felt nauseated with a placebo. We did not find reliable evidence that one drug was better than another. A person's age or sex, the type of surgery, or the time the drug was given did not change the effect of a drug. When drugs were given together, their effects simply added. Side effects were mild and affected four out of 100 people for the two drugs most studied.
Either nausea or vomiting are reported to affect, at most, 80 out of 100 people after surgery. If all 100 of these people are given a drug, about 28 would benefit and 72 would not. Nausea or vomiting are usually less common and therefore drugs are usually less useful.
Doctors should research how often drugs cause severe side effects.
Background
Postoperative nausea and vomiting (PONV) are unwanted outcomes after anaesthesia or sedation (Watcha 1992). Patients rate PONV as one of the least desirable events after surgery (Eberhart 2002; Engoren 2000; Gan 2001; Rashiq 2003). Postoperative nausea and vomiting can delay hospital discharge or result in unplanned admission. Vomiting can stress wounds, imbalance body electrolytes and cause bleeding (Watcha 1995c). Only a few factors, in just a few studies, have been shown to independently predict PONV: sex, history of smoking, motion sickness or PONV, duration of operation, and opioid administration (Apfel 2002b; Rüsch 2005; Van den Bosch 2005). Nausea or vomiting may be more frequent after some types of surgery, for example laparoscopy, strabismus and middle ear surgery (Cohen 1994; Kapur 1991; Kenny 1994; Kortilla 1992; Watcha 1992; Watcha 1995c). The risks of nausea or vomiting may vary with: preanaesthetic medication; anaesthetic drugs and techniques; postoperative pain management (Watcha 1992).
There are a number of published systematic reviews that report on one or more antiemetic drugs (Figueredo 1998; Gupta 2003; Henzi 1999; Henzi 2000; Hirayama 2001; Steward 2002; Tramèr 1995; Tramèr 1997; Tramèr 1999). These systematic reviews can tell the reader how well those drugs prevent PONV. The effects of some drugs have not been summarized in systematic reviews. We have tried to provide the reader a single place to find the effect on PONV of any drug that has been studied. We will update this review on a regular basis.
Objectives
Our objectives for this review were to determine the efficacy and safety of drugs for preventing postoperative nausea and vomiting. 'Prevention' means that the drug was given before a participant experienced either nausea or vomiting.
We assessed whether drugs changed the risks of two types of postoperative outcomes:
the risk of postoperative nausea or vomiting;
the risk of other adverse event/side effects.
We assessed each drug separately for these two primary analyses.
We also assessed whether:
the risks of postoperative nausea or vomiting are altered by the route of drug administration;
the risks of postoperative nausea or vomiting are altered by the timing of drug administration;
the risks of postoperative nausea or vomiting are altered by the dose of drug administered.
We only analysed the relative risks from within studies in these secondary analyses (intrastudy comparisons) ‐ we did not compare the risks between one study and another (interstudy comparisons).
We performed four subgroup analyses (interstudy comparisons) based upon:
the age of the participant;
the sex of the participant;
the type of surgery;
the time the drug was administered.
These four exploratory interstudy subgroup analyses are not as reliable as the intrastudy analyses (primary and secondary analyses) because participants were not randomly allocated to one study or another. For the first subgroup analysis, we categorized studies as assessing adults, children, or both. If the study authors did not define their participants as child or adult, we categorized participants of more than 17 years old as adult. We examined the effect of timing of drug administration with the fourth subgroup analysis. This interstudy analysis compared event rates between different trials; this is not the same as the third of the secondary analyses, that only included trials within which participants were allocated to receive a drug at different times.
We performed two post‐hoc analyses that we did not anticipate in the protocol. One assessed our decision to treat all control groups the same, whether or not the placebo group received a recognised antiemetic. The other analysis assessed studies of granisetron.
Methods
Criteria for considering studies for this review
Types of studies
We included randomized controlled trials (RCTs) that evaluated the effect of a drug or drugs given before the onset of postoperative nausea and vomiting. We excluded studies of treatment for established postoperative nausea or vomiting and studies of anaesthetic drugs or analgesics.
Types of participants
We included participants undergoing general anaesthesia, regional anaesthesia or sedation.
Types of interventions
We included any drug allocated before the onset of postoperative nausea or vomiting compared with placebo, compared with no treatment or compared with another drug. The drug could be given preoperatively, at induction of anaesthesia, intraoperatively or postoperatively (before nausea or vomiting had occurred).
Types of outcome measures
We analysed:
the proportion of participants nauseated postoperatively;
the proportion of participants vomiting postoperatively;
the proportion of participants who were either nauseated or who vomited;
the proportion of participants treated for nausea or vomiting postoperatively;
the proportion of participants who experienced side effects (any adverse outcome).
Search methods for identification of studies
We searched The Cochrane Central Register of Controlled Trials (CENTRAL) (The Cochrane Library, Issue 2, 2004) and DARE databases (to May 2004), MEDLINE (PubMed1966 to May 2004), EMBASE (1980 to May 2004), CINAHL (1982 to May 2004), AMED (1985 to May 2004), SIGLE (to May 2004), ISI WOS (to May 2004), LILAC (to May 2004) and INGENTA bibliographies. We used free text and their associated exploded MeSH terms. We assessed the studies we retrieved for any free text terms, MeSH terms for drugs that we had not already included. We updated the search strategy with new terms to increase the number of studies that we retrieved. We did not restrict the language. Please see Appendix 1.
Data collection and analysis
Trial identification
We first assessed study title and abstract. We retrieved copies of all eligible studies. We stated why we excluded studies (please see the table, 'Characteristics of excluded studies').
Quality assessment
We independently assessed: the method of allocation concealment (adequate, inadequate, unclear, not used); the method of randomization (adequate, inadequate, unclear); the blinding (yes, no) of allocation separately to the anaesthetist and the outcome assessor; follow up (complete, incomplete); and intention to treat analysis (yes, no). Please see the table 'Characteristics of included studies' for more information.
Data extraction
We recorded the type of participant, interventions and outcomes on a data extraction form. We did not contact study authors to supply missing data. We hope to retrieve some of these data when we update this systematic review.
Analysis
We made the following comparisons:
drug(s) versus placebo;
drug(s) versus no treatment;
drug(s) versus drug(s).
Authors used one or more of four outcomes to measure the effect of a drug: nausea; vomiting; nausea or vomiting; antiemetic treatment. We analysed these outcomes as dichotomous variables that participants either did or did not experience. Some authors graded nausea or vomiting, using distinctions such as 'mild', 'moderate', or 'severe'. We did not analyse grades of nausea or vomiting, as different studies used different scales. Some authors categorized PONV by the 'severest' symptom, for instance "vomiting (worse than) retching (worse than) nausea". We did not assume that someone categorized as vomiting was also nauseated. We categorized studies that compared a combination of two drugs versus one of those drugs (for instance dexamethasone and ondansetron versus dexamethasone) as 'drug versus placebo', in this example ondansetron versus placebo. We have analysed this decision in a post‐hoc analysis that we did not list in the protocol ('giving one antiemetic with another' in Results and 'Does it matter what you give the drug with?' in Discussion).
Studies recorded outcomes during different postoperative periods, for instance six hours, or 24 hours or 72 hours. Some authors divided the postoperative observation period, for instance dividing a 24‐hour observation period into a "0 to 4 hour" period and a "4 to 24 hours" period, but then did not report the risk for the complete observation period (0 to 24 hours in this example). We reported the risk of an outcome once for each study. We used the risk for the period in which the outcome was most common (all groups combined). A study with three groups, for instance placebo, dexamethasone and metoclopramide, allows three comparisons: placebo versus dexamethasone; placebo versus metoclopramide; dexamethasone versus metoclopramide. This means that the data from each group are used twice. Although each datum is used only once in each of the three analyses, we thought that such studies, with more than two groups, would have an exaggerated effect on the total review. We therefore reduced the contribution of such a study by adjusting for the number of times each datum was used: therefore if a datum was used twice, we divided the proportion by two. For instance, if 12 of 40 participants vomited in a group that was analysed twice, we used the proportion 6/20 for each analysis. If division resulted in numbers that were not integers, we used the next integer (Review Manager (RevMan 4.2) analyses only handle integers).
We constructed Funnel plots and Forest plots for each outcome and drug. We then constructed plots for subgroup analyses. We used a random‐effects model for all analyses. We expressed the treatment effects as relative risks. We discuss how the Number Needed to Treat changes with the control risk of PONV in the Discussion (Additional Table 14).
Table 1.
Turning relative risk into numbers needed to treat
| Relative Risk | Control incidence .8 | Control incidence .7 | Control incidence .6 | Control incidence .5 | Control incidence .4 | Control incidence .3 | Control incidence .2 | Control incidence .1 |
| Relative risk 0.96 Absolute risk AR AR reduction NNT | 0.96 0.77 0.03 31 | 0.96 0.67 0.03 36 | 0.96 0.58 0.02 42 | 0.96 0.48 0.02 50 | 0.96 0.384 0.016 63 | 0.96 0.288 0.012 83 | 0.96 0.192 0.008 125 | 0.96 0.096 0.004 250 |
| Relative risk 0.90 Absolute risk AR reduction NNT | 0.90 0.72 0.08 13 | 0.90 0.63 0.07 14 | 0.90 0.54 0.06 17 | 0.90 0.45 0.05 20 | 0.90 0.364 0.040 25 | 0.90 0.270 0.030 33 | 0.90 0.180 0.020 50 | 0.90 0.090 0.010 100 |
| Relative risk 0.86 Absolute risk AR reduction NNT | 0.86 0.69 0.11 9 | 0.86 0.60 0.10 10 | 0.86 0.52 0.08 12 | 0.86 0.43 0.07 14 | 0.86 0.344 0.056 18 | 0.86 0.258 0.042 24 | 0.86 0.172 0.028 36 | 0.86 0.086 0.014 71 |
| Relative risk 0.80 Absolute risk AR reduction NNT | 0.80 0.64 0.16 6 | 0.80 0.56 0.14 7 | 0.80 0.48 0.12 8 | 0.80 0.40 0.10 10 | 0.80 0.320 0.080 13 | 0.80 0.240 0.060 17 | 0.80 0.160 0.040 25 | 0.80 0.080 0.020 50 |
| Relative risk 0.74 Absolute risk AR reduction NNT | 0.74 0.59 0.21 5 | 0.74 0.52 0.18 5 | 0.74 0.44 0.16 6 | 0.74 0.37 0.13 8 | 0.74 0.296 0.104 10 | 0.74 0.222 0.078 13 | 0.74 0.148 0.052 19 | 0.74 0.074 0.026 38 |
| Relative risk 0.70 Absolute risk AR reduction NNT | 0.70 0.56 0.24 4 | 0.70 0.49 0.21 5 | 0.70 0.42 0.18 6 | 0.70 0.35 0.15 7 | 0.70 0.280 0.120 8 | 0.70 0.210 0.090 11 | 0.70 0.140 0.060 17 | 0.70 0.070 0.030 33 |
| Relative risk 0.64 Absolute risk AR reduction NNT | 0.64 0.51 0.29 3 | 0.64 0.45 0.25 4 | 0.64 0.38 0.22 5 | 0.64 0.32 0.18 6 | 0.64 0.256 0.144 7 | 0.64 0.192 0.108 9 | 0.64 0.128 0.072 14 | 0.64 0.064 0.036 28 |
| Relative risk 0.56 Absolute risk AR reduction NNT | 0.56 0.45 0.35 3 | 0.56 0.39 0.31 3 | 0.56 0.34 0.26 4 | 0.56 0.28 0.22 5 | 0.56 0.224 0.176 6 | 0.56 0.168 0.132 8 | 0.56 0.112 0.088 11 | 0.56 0.056 0.044 23 |
| Relative risk 0.50 Absolute risk AR reduction NNT | 0.50 0.40 0.40 3 | 0.50 0.35 0.35 3 | 0.50 0.30 0.30 3 | 0.50 0.25 0.25 4 | 0.50 0.200 0.200 5 | 0.50 0.150 0.150 7 | 0.50 0.100 0.100 10 | 0.50 0.050 0.050 20 |
Results
Description of studies
We retrieved 863 studies, of which we included 737 and excluded 126. Some of these excluded studies were abstracts of work subsequently published in full, or were incorporated into larger studies that referenced them, or had been previously identified as duplicates (Tramèr 1997b).
Age and sex of participants
The included studies contained 103,237 participants. The age of 98,474 participants was reported, of whom 21,632 were children and 76,842 were adults. The sex of 87,225 participants was reported, of whom 20,916 were male and 66,309 were female. Age and sex were reported for 85,737 participants: 8180 were boys; 5967 were girls; 11,916 were men; and 59,674 were women.
Drugs and number of studies
The included studies examined the effects of 60 different drugs (number of studies in brackets): alizapride (3); alprazolam (1); atropine (13); betamethasone (1); bromazepam (1); bromopride (1); butorphanol (1); chloral hydrate (1); chlorpromazine (2); cimetidine (2); cisapride (1); clebopride (2); clonidine (30); cp 122721 (2); cyclizine (10); dexamethasone (88); dexmedetomidine (1); diazepam (35); difenidol (1); dimenhydrinate (15); dixyrazine (4); dolasetron (26); domperidone (11); droperidol (222); edrophonium (2); ephedrine (4); flunitrazepam (4); flurbiprofen (1); ginger (6); glycopyrrolate (9); granisetron (81); hydroxyzine (1); hyoscine (16); intralipid (1); lidocaine (4); lorazepam (8); lormetazepam (1); magnesium (2); medazepam (1); methylnaltrexone (2); methylprednisolone (2); metoclopramide (158); midazolam (20); nabilone (1); naloxone (1); neostigmine (26); ondansetron (263); oxygen (7); palonosetron (2); perphenazine (11); physostigmine (1); prochlorperazine (13); promethazine (9); ramosetron (10); ranitidine (3); sulpiride (1); tandospirone (1); tiapride (1); trimethobenzamide (2); tropisetron (42). Some studies (318) assessed more than one drug.
Control
The control group in 510 studies received a placebo. The control group in 68 studies received no treatment. There was no control group in 159 studies. All of the studies that did not contain a control group compared two or more drugs (or two or more doses of a drug, or both). Many of the studies that contained a control group also compared drugs. In seven studies, the authors controlled for one intervention with a placebo and for another intervention with no treatment.
Number of interventions
There were 1442 intervention groups: 1316 groups received one drug; 125 groups received two drugs; and one group received four drugs. Three hundred and seventy‐nine studies assessed one drug, 297 studies assessed two drugs, 52 studies assessed three drugs and nine studies assessed four drugs. There was one intervention group in 276 studies, two in 273 studies, three in 146 studies, four in 31 studies, five groups in seven studies, six groups in three studies and seven intervention groups in one study.
Timing of interventions
A drug was given before anaesthesia was induced in 185 studies, at induction in 369 studies, during surgery in 183 studies, and after surgery in 107 studies. Ninety‐nine studies gave a drug during two of these four periods: before induction and after anaesthesia (16); before induction and on induction (14); before induction and during the maintenance of anaesthesia (12); on induction and during the maintenance of anaesthesia (16); on induction and after anaesthesia (22); during, and after, anaesthesia (19). Four studies gave an antiemetic drug during three of these four periods; before induction, on induction, and after anaesthesia (1); before induction, during the maintenance of, and after anaesthesia (1); on induction, during the maintenance of, and after anaesthesia (2).
Outcomes
The risk of nausea or vomiting was measured once in 406 studies, twice in 204 studies, thrice in 70 studies, four times in 39 studies, five times in 15 studies, six times in nine studies and seven times in four studies. The majority of studies ‐ including the 396 studies that measured the outcome once and 194 others ‐ reported risks for the total postoperative observation period. The remaining 147 studies only reported the risks during different parts of the observation period. For instance the risk of an outcome was reported during the first three hours and the subsequent 21 hours of a study but not for the total 24 hours. Side effects were looked for and reported in 380 studies.
Route and timing and dosage
Fourteen studies assessed how the route of administration changed drug effect; 15 studies assessed how timing of the intervention changed the effect of a drug; and 133 studies assessed the effect of a drug given at different doses.
Risk of bias in included studies
A sample size calculation was reported by 276 of the 737 included studies. We assessed the concealment of group allocation as adequate in 178 studies and inadequate in nine studies. The authors of the remaining 550 studies did not state how they concealed group allocation ‐ we categorized these studies as unclear. We assessed the allocation sequence as random in 195 studies and not random in seven studies. The authors of the remaining 535 studies did not state how they generated the allocation sequence ‐ we categorized these studies as unclear. Six hundred and thirty‐five studies blinded the outcome assessor to the intervention received by the participant, and 102 studies did not report blinding the outcome assessor. Two hundred and ninety studies blinded the anaesthesiologist (or other practitioner) who gave the anaesthetic (or sedation) to the intervention received by the participant, and 447 studies did not report blinding the anaesthesiologist. Five hundred and twelve studies analysed their results by intention to treat, and 225 studies did not. Five hundred and six studies included the results from all the participants (complete follow up), whilst 231 studies lost some participants to follow up.
Effects of interventions
Our results are summarized graphically as Forest plots. The numbers preceding each heading, for instance '1.1 Nausea', correspond to the numbered Forest plot. To save space, we have not presented the Forest plots for all of the subgroup analyses.
Primary analysis: the risk of postoperative nausea or vomiting
We separated the results into six divisions on the basis of what the control was, and whether an intervention group received a drug or a combination of drugs:
placebo versus drug;
no treatment versus drug;
drug versus drug;
placebo versus drugs;
no treatment versus drugs;
drugs versus drugs.
We used a random‐effects model to calculate the relative risk of the event and the 95% confidence intervals.
Placebo versus drug
These results are summarized in Additional Table 15 as well as the Forest plots. 1.1 Nausea (Analysis 1.1) We calculated that the risk (95% confidence interval) for postoperative nausea is decreased compared to placebo by: alizapride 0.65 (0.46 to 0.92); cyclizine 0.67 (0.51 to 0.89); dexamethasone 0.58 (0.48 to 0.69); diazepam 0.50 (0.25 to 0.99); dolasetron 0.82 (0.76 to 0.90); droperidol 0.65 (0.60 to 0.71); granisetron 0.53 (0.45 to 0.63); hyoscine 0.63 (0.47 to 0.83); lorazepam 0.55 (0.33 to 0.93); metoclopramide 0.82 (0.76 to 0.89); ondansetron 0.68 (0.63 to 0.74); prochlorperazine 0.73 (0.56 to 0.96); ramosetron 0.62 (0.40 to 0.96); tropisetron 0.77 (0.71 to 0.84).
Table 2.
Placebo versus Drug
| Drug | Nausea | Vomiting | Nausea or Vomiting | Rescue antiemetic |
| RR (95% CI) | RR (95% CI) | RR (95% CI) | RR (95% CI) | |
| Alizapride | 0.65 (0.46 ‐ 0.92) | 0.49 (0.29 ‐ 0.84) | 0.68 (0.39 ‐ 1.19) | no result |
| Atropine | no result | 1.11 (0.78 ‐ 1.58) | 0.91 (0.36 ‐ 2.31) | no result |
| Cimetidine | 0.66 (0.16 ‐ 2.68) | 0.47 (0.17 ‐ 1.32) | no result | no result |
| Clonidine | 0.69 (0.46 ‐ 1.05) | 0.75 (0.53 ‐ 1.06) | 0.73 (0.52 ‐ 1.02) | 1.09 (0.94 ‐ 1.27) |
| Cyclizine | 0.65 (0.47 ‐ 0.90) | 0.57 (0.43 ‐ 0.75) | 0.68 (0.58 ‐ 0.80) | 0.27 (0.14 ‐ 0.62) |
| Dexamethasone | 0.57 (0.48 ‐ 0.69) | 0.51 (0.46 ‐ 0.57) | 0.49 (0.44 ‐ 0.54) | 0.50 (0.42 ‐ 0.59) |
| Diazepam | 0.50 (0.25 ‐ 0.99) | 0.85 (0.58 ‐ 1.24) | 1.04 (0.51 ‐ 2.10) | no result |
| Dimenhydrinate | 0.72 (0.47 ‐ 1.13) | 0.61 (0.46 ‐ 0.81) | 0.71 (0.59 ‐ 0.86) | 0.62(0.33 ‐ 1.15) |
| Dixyrazine | no result | no result | 0.83 (0.67 ‐ 1.02) | 0.49 (0.30 ‐ 0.80) |
| Dolasetron | 0.82 (0.76 ‐ 0.90) | 0.63 (0.51 ‐ 0.76) | 0.72 (0.62 ‐ 0.83) | 0.67 (0.57 to 0.79) |
| Domperidone | 0.62 (0.20 ‐ 1.94) | 0.80 (0.52 ‐ 1.23) | 0.71 (0.44 ‐ 1.13) | no result |
| Droperidol | 0.65 (0.60 ‐ 0.71) | 0.65 (0.61 ‐ 0.70) | 0.62 (0.58 ‐ 0.67) | 0.53 (0.47 ‐ 0.60) |
| Ephedrine | 0.50 (0.20 ‐ 1.23) | 0.91 (0.64 ‐ 1.27) | 0.79 (0.55 ‐ 1.15) | 0.82 (0.41 ‐ 1.66) |
| Ginger | 0.87 (0.62 ‐ 1.23) | 1.04 (0.66 ‐ 1.64) | 1.02 (0.73 ‐ 1.42) | 0.40 (0.18 ‐ 0.88) |
| Glycopyrrolate | no result | no result | 0.67 (0.35 ‐ 1.29) | 0.52 (0.18 ‐ 1.48) |
| Granisetron | 0.53 (0.45 to 0.63) | 0.40 (0.35 ‐ 0.46) | 0.39 (0.31 ‐ 0.48) | 0.29 (0.22 ‐ 0.39) |
| Hyoscine | 0.63 (0.47 ‐ 0.83) | 0.66 (0.56 ‐ 0.77) | 0.71 (0.56 ‐ 0.90) | 0.92 (0.69 ‐ 1.21) |
| Lorazepam | 0.55 (0.33 ‐ 0.93) | 0.61(0.33 ‐ 1.13) | no result | no result |
| Magnesium | no result | no result | 0.79 (0.36 ‐ 1.72) | no result |
| Methylnaltrexone | no result | 0.64 (0.30 ‐ 1.33) | no result | 0.63 (0.33 ‐ 1.21) |
| Metoclopramide | 0.82 (0.76 ‐ 0.88) | 0.75 (0.70 ‐ 0.81) | 0.76 (0.70 ‐ 0.82) | 0.78 (0.69 ‐ 0.88) |
| Midazolam | 0.90 (0.64 ‐ 1.28) | 0.73 (0.56 ‐ 0.95) | 1.44 (0.52 ‐ 3.94) | 0.61 (0.38 ‐ 0.98) |
| Neostigmine | 2.73 (1.15 ‐ 6.48) | 3.87 (0.79 ‐ 19.0) | 3.19 (1.71 ‐ 5.93) | 1.39 (0.55 ‐ 3.50) |
| Ondansetron | 0.68 (0.63 ‐ 0.74) | 0.55 (0.50 ‐ 0.59) | 0.56 (0.50 ‐ 0.63) | 0.55 (0.49 ‐ 0.61) |
| Perphenazine | 1.15 (0.42 ‐ 3.12) | 0.70 (0.51 ‐ 0.96) | 0.71 (0.43 ‐ 1.15) | no result |
| Prochlorperazine | 0.73 (0.56 ‐ 0.96) | 0.68 (0.52 ‐ 0.89) | 0.68 (0.55 ‐ 0.86) | 0.49 (0.22 ‐ 1.08) |
| Promethazine | no result | 0.76 (0.40 ‐ 1.45) | 0.46 (0.25 ‐ 0.82) | no result |
| Ramosetron | 0.62 (0.40 ‐ 0.96) | 0.42 (0.28 ‐ 0.63) | 0.51 (0.39 ‐ 0.68) | 0.38 (0.15 ‐ 0.99) |
| Tropisetron | 0.77 (0.71 ‐ 0.84) | 0.59 (0.50 ‐ 0.69) | 0.70 (0.61 ‐ 0.81) | 0.62 (0.53 ‐ 0.72) |
Analysis 1.1.
Comparison 1 PRIMARY ANALYSIS: Placebo versus Drug, Outcome 1 Nausea.
We calculated that there is no evidence that the risk of postoperative nausea is changed by: cimetidine 0.66 (0.16 to 2.68); clonidine 0.69 (0.46 to 1.05); dimenhydrinate 0.72 (0.47 to 1.13); domperidone 0.62 (0.20 to 1.94); ginger 0.87 (0.62 to 1.23); midazolam 0.90 (0.64 to 1.28); perphenazine 1.15 (0.42 to 3.12). We calculated that neostigmine increases the risk of postoperative nausea, relative risk 2.73 (1.15 to 6.48).
1.2 Vomiting (Analysis 1.2) We calculated that the risk (95% confidence interval) for postoperative vomiting is decreased compared to placebo by: alizapride 0.49 (0.29 to 0.84); cyclizine 0.55 (0.43 to 0.71); dexamethasone 0.51 (0.46 to 0.56); dimenhydrinate 0.61 (0.46 to 0.81); dolasetron 0.62 (0.51 to 0.76); droperidol 0.65 (0.60 to 0.70); granisetron 0.40 (0.35 to 0.46); hyoscine 0.65 (0.55 to 0.77); metoclopramide 0.76 (0.70 to 0.81); midazolam 0.73 (0.56 to 0.95); ondansetron 0.54 (0.50 to 0.59); perphenazine 0.70 (0.51 to 0.96); prochlorperazine 0.68 (0.52 to 0.89); ramosetron 0.42 (0.28 to 0.63); tropisetron 0.60 (0.51 to 0.70).
Analysis 1.2.
Comparison 1 PRIMARY ANALYSIS: Placebo versus Drug, Outcome 2 Vomiting.
We calculated that there is no evidence that the risk of postoperative vomiting is changed by: atropine 1.11 (0.78 to 1.58); cimetidine 0.47 (0.17 to 1.32); clonidine 0.75 (0.53 to 1.06); diazepam 0.85 (0.58 to 1.24); domperidone 0.80 (0.52 to 1.23); ephedrine 1.00 (0.69 to 1.45); ginger 1.00 (0.65 to 1.54); lorazepam 0.61 (0.33 to 1.13); methylnaltrexone 0.64 (0.30 to 1.33); neostigmine 3.87 (0.79 to 18.99); promethazine 0.76 (0.40 to 1.45).
1.3 Nausea or vomiting (Analysis 1.3) We calculated that the risk (95% confidence interval) for postoperative 'nausea or vomiting' is decreased compared to placebo by: cyclizine 0.67 (0.56 to 0.79); dexamethasone 0.48 (0.43 to 0.54); dimenhydrinate 0.71 (0.59 to 0.86); dolasetron 0.72 (0.62 to 0.83); droperidol 0.62 (0.58 to 0.67); granisetron 0.39 (0.31 to 0.48); hyoscine 0.71 (0.56 to 0.90); metoclopramide 0.76 (0.70 to 0.82); ondansetron 0.56 (0.50 to 0.62); prochlorperazine 0.68 (0.55 to 0.86); promethazine 0.46 (0.25 to 0.82); ramosetron 0.51(0.39 to 0.68); tropisetron 0.72 (0.63 to 0.82).
Analysis 1.3.
Comparison 1 PRIMARY ANALYSIS: Placebo versus Drug, Outcome 3 Nausea or Vomiting.
We calculated that there is no evidence that the risk of postoperative 'nausea or vomiting' is changed by: alizapride 0.68 (0.39 to 1.19); atropine 0.91 (0.36 to 2.91); clonidine 0.73 (0.52 to 1.02); diazepam 1.04 (0.51 to 2.10); dixyrazine 0.83 (0.67 to 1.02); domperidone 0.71 (0.44 to 1.13); ephedrine 0.84 (0.52 to 1.34); ginger 0.79 (0.55 to 1.14); glycopyrrolate 0.67 (0.35 to 1.29); magnesium 0.79 (0.36 to 1.72); midazolam 1.44 (0.52 to 3.94); perphenazine 0.71 (0.43 to 1.15). We calculated that neostigmine increased the risk of postoperative nausea or vomiting ‐ relative risk 3.19 (95% confidence interval 1.71 to 5.93).
1.4 Rescue antiemetic (Analysis 1.4) We calculated that the risk (95% confidence interval) of treatment for postoperative nausea or vomiting is decreased compared to placebo by: cyclizine 0.27 (0.15 to 0.48); dexamethasone 0.49 (0.41 to 0.58); dixyrazine 0.49 (0.30 to 0.80); dolasetron 0.67 (0.57 to 0.79); droperidol 0.53 (0.47 to 0.59); ginger 0.40 (0.18 to 0.88); granisetron 0.29 (0.22 to 0.39); lorazepam 0.55 (0.33 to 0.93); metoclopramide 0.78 (0.69 to 0.88); midazolam 0.61 (0.38 to 0.98); ondansetron 0.54 (0.48 to 0.60); ramosetron 0.38 (0.15 to 0.99); tropisetron 0.63 (0.55 to 0.73).
Analysis 1.4.
Comparison 1 PRIMARY ANALYSIS: Placebo versus Drug, Outcome 4 Rescue antiemetic.
We calculated that there is no evidence that the risk of treatment for postoperative nausea or vomiting is changed by: clonidine 1.09 (0.94 to 1.27); dimenhydrinate 0.62 (0.33 to 1.15); ephedrine 0.82 (0.41 to 1.66); glycopyrrolate 0.52 (0.18 to 1.48); hyoscine 0.92 (0.69 to 1.21); methylnaltrexone 0.63 (0.33 to 1.21); neostigmine 1.39 (0.55 to 3.50); prochlorperazine 0.49 (0.22 to 1.08).
No treatment versus drug
2.1 Nausea (Analysis 2.1) We calculated that the risk (95% confidence interval) for postoperative nausea is decreased compared to no treatment by: droperidol 0.58 (0.41 to 0.81); metoclopramide 0.34 (0.17 to 0.66); ondansetron 0.66 (0.49 to 0.88).
Analysis 2.1.

Comparison 2 PRIMARY ANALYSIS: No Treatment versus Drug, Outcome 1 Nausea.
We calculated that there is no evidence that promethazine changes the risk of postoperative nausea ‐ relative risk 0.81 (0.55 to 1.20).
2.2 Vomiting (Analysis 2.2) We calculated that the risk (95% confidence interval) for postoperative vomiting is decreased compared to no treatment by: dexamethasone 0.40 (0.24 to 0.65); dixyrazine 0.31 (0.18 to 0.53); droperidol 0.65 (0.53 to 0.79); metoclopramide 0.49 (0.30 to 0.79); ondansetron 0.43 (0.34 to 0.54).
Analysis 2.2.

Comparison 2 PRIMARY ANALYSIS: No Treatment versus Drug, Outcome 2 Vomiting.
We calculated that there is no evidence that promethazine changes the risk of postoperative vomiting ‐ relative risk 0.53 (0.15 to 1.84).
2.3 Nausea or Vomiting (Analysis 2.3) We calculated that the risk (95% confidence interval) for postoperative 'nausea or vomiting' is decreased compared to no treatment by: droperidol 0.56 (0.41 to 0.78); metoclopramide 0.35 (0.17 to 0.74); ondansetron 0.61 (0.46 to 0.81).
Analysis 2.3.

Comparison 2 PRIMARY ANALYSIS: No Treatment versus Drug, Outcome 3 Nausea or Vomiting.
2.4 Rescue antiemetic (Analysis 2.4)
Analysis 2.4.

Comparison 2 PRIMARY ANALYSIS: No Treatment versus Drug, Outcome 4 Rescue antiemetic.
We calculated that the risk (95% confidence interval) of treatment for postoperative nausea or vomiting is decreased compared to no treatment by: dixyrazine 0.08 (0.01 to 0.61); droperidol 0.57 (0.40 to 0.82); ondansetron 0.62 (0.43 to 0.90).
Drug versus drug
Most of these results are summarized in Additional Table 16, as well as the Forest plots.
Table 3.
Effective drug versus effective drug
| Comparison | Nausea | Vomiting | Nausea or Vomiting | Rescue antiemetic | Differences |
| Drug versus Drug | RR (95% CI) | RR (95% CI) | RR (95% CI) | RR (95% CI) | Number of outcomes different |
| Cyclizine ‐ Dexamethasone | One study | One study | One study | One study | No result |
| Cyclizine ‐ Dolasetron | No study | No study | No study | No study | No result |
| Cyclizine ‐ Droperidol | One study | One study | One study | No study | No result |
| Cyclizine ‐ Granisetron | No study | No study | No study | No study | No result |
| Cyclizine ‐ Metoclopramide | No study | No study | One study | No study | No result |
| Cyclizine ‐ Ondansetron | 1.00 (0.69 to 1.44) | 1.36 (0.58 to 3.18) | 1.19 (0.73 to 1.95) | 0.66 (0.31 to 1.40) | 0/4 |
| Cyclizine ‐ Ramosetron | No study | No study | No study | No study | No result |
| Cyclizine ‐ Tropisetron | No study | No study | No study | No study | No result |
| Dexamethasone ‐ Dolasetron | No study | No study | No study | No study | No result |
| Dexamethasone ‐ Droperidol | 1.08 (0.64 to 1.84) | 0.96 (0.48 to 1.93) | 1.04 (0.72 to 1.52) | 1.17 (0.68 to 2.04) | 0/4 |
| Dexamethasone ‐ Granisetron | 1.65 (0.54 to 5.04) | 1.75 (0.85 to 3.62) | No study | 8.00 (1.04 to 61.5) | 1/3 |
| Dexamethasone Metoclopramide | 0.61 (0.28 to 1.34) | 0.45 (0.17 to 1.20) | 0.59 (0.35 to 0.99) | 0.50 (0.19 to 1.33) | No result |
| Dexamethasone ‐ Ondansetron | 1.27 (0.94 to 1.71) | 1.38 (0.84 to 2.26) | 1.23 (0.96 to 1.59) | 1.19 (0.78 to 1.80) | 0/4 |
| Dexamethasone ‐ Ramosetron | No study | No study | No study | No study | No result |
| Dexamethasone ‐ Tropisetron | 0.41 (0.22 to 0.78) | 0.38 (0.13 to 1.11) | 0.41 (0.22 to 0.78) | 0.44 (0.19 to 1.04) | 2/4 |
| Dolasetron ‐ Droperidol | 1.06 (0.62 to 1.82) | 0.80 (0.50 to 1.30) | 0.95 (0.77 to 1.17) | No study | 0/3 |
| Dolasetron ‐ Granisetron | No study | No study | No study | No study | No result |
| Dolasetron ‐ Metoclopramide | 0.85 (0.57 to 1.26) | 0.36 (0.19 to 0.65) | 0.70 (0.47 to 1.04) | 0.55 (0.32 to 0.94) | 2/4 |
| Dolasetron ‐ Ondansetron | 1.02 (0.81 to 1.28) | 1.17 (0.94 to 1.45) | 1.03 (0.83 to 1.27) | 0.98 (0.75 to 1.29) | 0/4 |
| Dolasetron ‐ Ramosetron | No study | No study | No study | No study | No result |
| Dolasetron ‐ Tropisetron | One study | One study | No study | No study | No result |
| Droperidol ‐ Granisetron | 1.36 (1.05 to 1.77) | 2.16 (1.71 to 2.72) | 2.08 (1.55 to 2.80) | 3.62 (2.41 to 5.46) | 4/4 |
| Droperidol ‐ Metoclopramide | 0.90 (0.74 to 1.10) | 0.83 (0.71 to 0.96) | 0.77 (0.65 to 0.91) | 0.78 (0.58 to 1.03) | 2/4 |
| Droperidol ‐ Ondansetron | 0.95 (0.88 to 1.03) | 1.22 (1.09 to 1.37) | 0.99 (0.88 to 1.12) | 1.01 (0.89 to 1.14) | 1/4 |
| Droperidol ‐ Ramosetron | No study | No study | No study | No study | No result |
| Droperidol ‐ Tropisetron | 1.07 (0.86 to 1.33) | 1.10 (0.54 to 2.22) | 1.03 (0.81 to 1.30) | 1.07 (0.78 to 1.46) | 0/4 |
| Granisetron ‐ Metoclopramide | 0.50 (0.31 to 0.81) | 0.39 (0.26 to 0.59) | 0.38 (0.27 to 0.55) | 0.21 (0.11 to 0.42) | 4/4 |
| Granisetron ‐ Ondansetron | No study | No study | No study | 1.12 (0.38 to 3.34) | 0/1 |
| Granisetron ‐ Ramosetron | 2.34 (1.11 to 4.94) | 2.82 (1.69 to 4.71) | 2.50 (1.18 to 5.29) | One study | 3/3 |
| Granisetron ‐ Tropisetron | No study | No study | No study | 1.00 (0.34 to 2.91) | 0/1 |
| Metoclopramide ‐ Ondansetron | 1.22 (1.01 to 1.47) | 1.48 (1.23 to 1.77) | 1.28 (1.03 to 1.58) | 1.12 (0.99 to 1.27) | 3/4 |
| Metoclopramide ‐ Ramosetron | No study | No study | No study | No study | No result |
| Metoclopramide ‐ Tropisetron | 0.86 (0.50 to 1.48) | 1.33 (0.70 to 2.53) | 1.20 (0.88 to 1.62) | 1.29 (0.90 to 1.85) | 0/4 |
| Ondansetron ‐ Ramosetron | No study | No study | No study | No study | No result |
| Ondansetron ‐ Tropisetron | 1.15 (0.82 to 1.60) | 1.53 (1.15 to 2.04) | 1.09 (0.88 to 1.36) | 1.08 (0.85 to 1.39) | 1/4 |
| Ramosetron ‐ Tropisetron | No study | No study | No study | No study | No result |
3.1 Nausea (Analysis 3.1) We calculated that the risk (95% confidence interval) of postoperative nausea was different when the following drugs were compared: dexamethasone was superior to tropisetron 0.41 (0.22 to 0.78); droperidol was inferior to granisetron 1.36 (1.05 to 1.77); granisetron was superior to metoclopramide 0.50 (0.31 to 0.81); granisetron was inferior to ramosetron 2.34 (1.11 to 4.94).
Analysis 3.1.

Comparison 3 PRIMARY ANALYSIS: Drug versus Drug, Outcome 1 Nausea.
We calculated that there is no evidence of different risks for postoperative nausea when the following drugs were compared: atropine versus hyoscine 2.33 (0.98 to 5.58); cyclizine versus ondansetron 1.00 (0.69 to 1.44); dexamethasone versus droperidol 1.08 (0.65 to 1.78); dexamethasone versus granisetron 1.07 (0.15 to 7.57); dexamethasone versus metoclopramide 0.61 (0.28 to 1.34); dexamethasone versus ondansetron 1.27 (0.94 to 1.71); diazepam versus promethazine 0.83 (0.39 to 1.76); dimenhydrinate versus droperidol 1.70 (0.73 to 3.99); dimenhydrinate versus metoclopramide 1.51 (0.43 to 5.33); dimenhydrinate versus ondansetron 0.80 (0.51 to 1.26); dolasetron versus droperidol 1.06 (0.62 to 1.82); dolasetron versus metoclopramide 0.85 (0.57 to 1.26); dolasetron versus ondansetron 1.02 (0.81 to 1.28); domperidone versus droperidol 0.96 (0.23 to 4.05); domperidone versus metoclopramide 0.94 (0.62 to 1.43); droperidol versus metoclopramide 0.91 (0.73 to 1.31); droperidol versus ondansetron 0.95 (0.88 to 1.03); droperidol versus propofol 3.48 (0.78 to 15.46); droperidol versus tropisetron 1.07 (0.86 to 1.33); ginger versus metoclopramide 0.92 (0.54 to 1.59); metoclopramide versus ondansetron 1.19 (0.99 to 1.44); metoclopramide versus tropisetron 0.86 (0.50 to 1.48); ondansetron versus prochlorperazine 0.96 (0.49 to 1.86); ondansetron versus promethazine 0.81 (0.46 to 1.40); ondansetron versus tropisetron 1.15 (0.82 to 1.60).
3.2 Vomiting (Analysis 3.2) We calculated that the risk (95% confidence interval) of postoperative vomiting was different when the following drugs were compared: atropine was superior to glycopyrrolate 0.67 (0.50 to 0.90); atropine was inferior to hyoscine 3.12 (1.56 to 6.25); diazepam was inferior to droperidol 2.16 (1.39 to 3.34); diazepam was inferior to flunitrazepam 1.74 (1.04 to 2.91); dimenhydrinate was inferior to ondansetron 1.76 (1.09 to 2.85); dolasetron was superior to metoclopramide 0.36 (0.19 to 0.65); droperidol was inferior to granisetron 2.16 (1.71 to 2.72); droperidol was superior to metoclopramide 0.83 (0.71 to 0.97); droperidol was superior to midazolam 0.77 (0.63 to 0.94); droperidol was inferior to ondansetron 1.20 (1.07 to 1.34); granisetron was superior to metoclopramide 0.39 (0.26 to 0.59); granisetron was superior to perphenazine 0.36 (0.21 to 0.62); granisetron was inferior to ramosetron 2.82 (1.69 to 4.71); metoclopramide was inferior to ondansetron 1.44 (1.20 to 1.73); ondansetron was inferior to tropisetron 1.54 (1.15 to 2.06).
Analysis 3.2.
Comparison 3 PRIMARY ANALYSIS: Drug versus Drug, Outcome 2 Vomiting.
We calculated that there is no evidence of different risks for postoperative vomiting when the following drugs were compared: clonidine versus diazepam 0.58 (0.29 to 1.15); clonidine versus midazolam 0.81 (0.31 to 2.10); cyclizine versus ondansetron 1.36 (0.58 to 3.18); dexamethasone versus droperidol 0.97 (0.51 to 1.84); dexamethasone versus granisetron 1.72 (0.80 to 3.70); dexamethasone versus metoclopramide 0.45 (0.17 to 1.20); dexamethasone versus ondansetron 1.38 (0.84 to 2.26); dexamethasone versus tropisetron 0.38 (0.13 to 1.11); diazepam versus midazolam 2.08 (0.28 to 15.60); diazepam versus phenobarbitone 0.95 (0.66 to 1.38); diazepam versus promethazine 1.78 (0.32 to 10.03); diazepam versus trimeprazine 1.96 (0.98 to 3.90); dimenhydrinate versus droperidol 0.93 (0.53 to 1.64); dimenhydrinate versus metoclopramide 0.79 (0.46 to 1.36); dolasetron versus droperidol 0.80 (0.50 to 1.30); dolasetron versus ondansetron 1.17 (0.94 to 1.45); domperidone versus droperidol 2.13 (0.82 to 5.53); domperidone versus metoclopramide 1.01 (0.64 to 1.59); droperidol versus ephedrine 1.00 (0.15 to 6.45); droperidol versus propofol 3.00 (0.66 to 13.69); droperidol versus tropisetron 1.10 (0.54 to 2.22); metoclopramide versus perphenazine 0.75 (0.37 to 1.54); metoclopramide versus tropisetron 1.33 (0.70 to 2.53); ondansetron versus prochlorperazine 0.87 (0.50 to 1.50); ondansetron versus promethazine 0.84 (0.48 to 1.45); pentobarbitone versus trimeprazine 1.19 (0.33 to 4.32).
3.3 Nausea or Vomiting (Analysis 3.3) We calculated that the risk (95% confidence interval) of the combined outcome postoperative 'nausea or vomiting' was different when the following drugs were compared: atropine was inferior to hyoscine 2.79 (1.74 to 4.45); clonidine was superior to neostigmine 0.31 (0.11 to 0.86); dexamethasone was superior to metoclopramide 0.59 (0.35 to 0.99); dexamethasone was superior to tropisetron 0.41 (0.22 to 0.78); domperidone was inferior to droperidol 1.80 (1.05 to 3.08); droperidol was inferior to granisetron 2.08 (1.55 to 2.80); droperidol was superior to metoclopramide 0.77 (0.65 to 0.92); droperidol was inferior to propofol 2.98 (1.08 to 8.24); granisetron was superior to metoclopramide 0.35 (0.24 to 0.51); granisetron was inferior to ramosetron 2.50 (1.18 to 5.29); metoclopramide was inferior to ondansetron 1.28 (1.03 to 1.58); ondansetron was superior to prochlorperazine 0.61 (0.43 to 0.87).
Analysis 3.3.

Comparison 3 PRIMARY ANALYSIS: Drug versus Drug, Outcome 3 Nausea or Vomiting.
We calculated that there is no evidence of different risks for postoperative 'nausea or vomiting' when the following drugs were compared: atropine versus glycopyrrolate 0.65 (0.20 to 2.17); clonidine versus midazolam 0.75 (0.41 to 1.37); cyclizine versus ondansetron 1.19 (0.73 to 1.95); dexamethasone versus droperidol 1.04 (0.72 to 1.52); dexamethasone versus granisetron 0.96 (0.10 to 9.32); dexamethasone versus ondansetron 1.29 (0.99 to 1.68); diazepam versus flunitrazepam 1.41 (0.44 to 4.56); dimenhydrinate versus droperidol 1.31 (0.58 to 2.96); dimenhydrinate versus metoclopramide 1.09 (0.44 to 2.70); dolasetron versus droperidol 0.95 (0.77 to 1.17); dolasetron versus metoclopramide 0.70 (0.47 to 1.04); dolasetron versus ondansetron 1.03 (0.83 to 1.27); domperidone versus metoclopramide 0.90 (0.72 to 1.13); droperidol versus granisetron 2.08 (1.55 to 2.80); droperidol versus ondansetron 0.99 (0.86 to 1.14); droperidol versus tropisetron 1.03 (0.81 to 1.30); ginger versus metoclopramide 0.94 (0.57 to 1.53); metoclopramide versus tropisetron 1.20 (0.88 to 1.62); ondansetron versus promethazine 0.75 (0.46 to 1.22); ondansetron versus tropisetron 1.09 (0.88 to 1.36).
3.4 Rescue antiemetic (Analysis 3.4) We calculated that the risk (95% confidence interval) of treatment for postoperative nausea or vomiting was different when the following drugs were compared: atropine was inferior to hyoscine 3.00 (1.49 to 6.03); dexamethasone was inferior to granisetron 7.95 (1.03 to 61.15); dolasetron was superior to metoclopramide 0.55 (0.33 to 0.94); droperidol was inferior to granisetron 2.77 (1.82 to 4.21); granisetron was superior to metoclopramide 0.32 (0.17 to 0.62).
Analysis 3.4.

Comparison 3 PRIMARY ANALYSIS: Drug versus Drug, Outcome 4 Rescue antiemetic.
We calculated that there is no evidence of different risks of treatment for postoperative nausea or vomiting when the following drugs were compared: atropine versus glycopyrrolate 0.69 (0.21 to 2.27); cyclizine versus ondansetron 0.65 (0.30 to 1.39); dexamethasone versus droperidol 1.18 (0.68 to 2.06); dexamethasone versus metoclopramide 0.50 (0.19 to 1.33); dexamethasone versus ondansetron 1.32 (0.83 to 2.10); dexamethasone versus tropisetron 0.44 (0.19 to 1.04); dimenhydrinate versus ondansetron 0.95 (0.64 to 1.43); dolasetron versus ondansetron 0.97 (0.77 to 1.22); domperidone versus metoclopramide 0.93 (0.58 to 1.48); droperidol versus ephedrine 0.80 (0.24 to 2.59); droperidol versus metoclopramide 0.85 (0.64 to 1.14); droperidol versus ondansetron 1.01 (0.89 to 1.14); droperidol versus propofol 2.93 (0.63 to 13.61); droperidol versus tropisetron 1.11 (0.81 to 1.52); granisetron versus ondansetron 1.14 (0.39 to 3.31); granisetron versus tropisetron 1.00 (0.35 to 2.82); metoclopramide versus ondansetron 1.11 (0.97 to 1.27); metoclopramide versus tropisetron 1.31 (0.93 to 1.85); ondansetron versus prochlorperazine 1.45 (0.65 to 3.28); ondansetron versus tropisetron 1.08 (0.86 to 1.34).
Placebo versus drugs
4.1 Nausea (Analysis 4.1) We calculated that dexamethasone combined with ondansetron decreases the risk for postoperative nausea compared to placebo ‐ relative risk 0.32 (95% confidence interval 0.17 to 0.60). We calculated that there is no evidence that the following drug combinations change the risk of postoperative nausea compared to placebo: dexamethasone and granisetron 0.26 (0.06 to 1.12); dimenhydrinate and droperidol 0.45 (0.18 to 1.13); dimenhydrinate and metoclopramide 0.74 (0.24 to 2.25); dolasetron and droperidol 0.43 (0.09 to 2.11); droperidol and ondansetron 0.43 (0.11 to 1.67); glycopyrrolate and neostigmine 1.38 (0.95 to 1.99).
Analysis 4.1.

Comparison 4 PRIMARY ANALYSIS: Placebo versus Drugs, Outcome 1 Nausea.
4.2 Vomiting (Analysis 4.2) We calculated that the following drug combinations decrease the risk (95% confidence interval) for postoperative vomiting compared to placebo: dexamethasone and ondansetron 0.31 (0.14 to 0.70); droperidol and ondansetron 0.36 (0.19 to 0.67). We calculated that there is no evidence that the following drug combinations change the risk for postoperative vomiting compared to placebo: dexamethasone and granisetron 0.28 (0.06 to 1.23); dimenhydrinate and droperidol 0.31 (0.08 to 1.17); dimenhydrinate and metoclopramide 0.40 (0.09 to 1.85); dolasetron and droperidol 0.33 (0.08 to 1.45); droperidol and metoclopramide 0.68 (0.27 to 1.71); glycopyrrolate and neostigmine 0.97 (0.68 to 1.38).
Analysis 4.2.

Comparison 4 PRIMARY ANALYSIS: Placebo versus Drugs, Outcome 2 Vomiting.
4.3 Nausea or vomiting (Analysis 4.3) We calculated that the following drug combinations decrease the risk (95% confidence interval) for postoperative 'nausea or vomiting' compared to placebo: dexamethasone and ondansetron 0.33 (0.22 to 0.49); droperidol and ondansetron 0.38 (0.18 to 0.81).
Analysis 4.3.

Comparison 4 PRIMARY ANALYSIS: Placebo versus Drugs, Outcome 3 Nausea or Vomiting.
We calculated that there is no evidence that following drug combinations change the risk for 'nausea or vomiting' compared to placebo: clonidine and neostigmine 1.59 (0.12 to 21.80); dimenhydrinate and droperidol 0.45 (0.18 to 1.13); dimenhydrinate and metoclopramide 0.58 (0.23 to 1.46); dolasetron and droperidol 0.35 (0.12 to 1.03); glycopyrrolate and neostigmine 1.03 (0.86 to 1.23).
4.4 Rescue antiemetic (Analysis 4.4) We calculated that the following combinations of drugs decrease the risk (95% confidence interval) of treatment for nausea or vomiting compared to placebo: dexamethasone and ondansetron 0.19 (0.07 to 0.52); droperidol and ondansetron 0.32 (0.14 to 0.76).
Analysis 4.4.

Comparison 4 PRIMARY ANALYSIS: Placebo versus Drugs, Outcome 4 Rescue antiemetic.
We calculated that there is no evidence that glycopyrrolate combined with neostigmine changes the risk of treatment for nausea or vomiting compared to placebo ‐ relative risk 1.42 (95% confidence interval 0.71 to 2.86).
No treatment versus drugs
5.1 Nausea (Analysis 5.1) We calculated that there is no evidence that atropine combined with neostigmine changes the risk of postoperative nausea compared to no treatment ‐ relative risk 1.57 (95% confidence interval 0.96 to 2.59).
Analysis 5.1.

Comparison 5 PRIMARY ANALYSIS: No Treatment versus Drugs, Outcome 1 Nausea.
5.2 Vomiting (Analysis 5.2) We calculated that there is no evidence that atropine combined with neostigmine changes the risk of postoperative vomiting compared to no treatment ‐ relative risk 2.19 (95% confidence interval 0.77 to 6.21).
Analysis 5.2.

Comparison 5 PRIMARY ANALYSIS: No Treatment versus Drugs, Outcome 2 Vomiting.
5.3 Nausea or vomiting (Analysis 5.3)
Analysis 5.3.

Comparison 5 PRIMARY ANALYSIS: No Treatment versus Drugs, Outcome 3 Nausea or Vomiting.
No results.
5.4 Rescue antiemetic (Analysis 5.4)
Analysis 5.4.

Comparison 5 PRIMARY ANALYSIS: No Treatment versus Drugs, Outcome 4 Rescue antiemetic.
No results.
Drugs versus drugs
6.1 Nausea (Analysis 6.1) We calculated that there is no evidence of different risks of postoperative nausea following droperidol versus dexamethasone combined with granisetron ‐ relative risk 1.21 (95% confidence interval 0.83 to 1.76).
Analysis 6.1.

Comparison 6 PRIMARY ANALYSIS: Drugs versus Drugs, Outcome 1 Nausea.
6.2 Vomiting (Analysis 6.2) We calculated that the risk of postoperative vomiting is greater following droperidol than following dexamethasone combined with granisetron ‐ relative risk 2.11 (95% confidence interval 1.35 to 3.32).
Analysis 6.2.

Comparison 6 PRIMARY ANALYSIS: Drugs versus Drugs, Outcome 2 Vomiting.
We calculated that there is no evidence that the risk of postoperative vomiting is different following droperidol combined with metoclopramide compared to ondansetron 0.67 (0.13 to 3.53).
6.3 Nausea or vomiting (Analysis 6.3) We calculated that there is no evidence that the risk of postoperative 'nausea or vomiting' is different following dexamethasone combined with granisetron compared to droperidol ‐ relative risk 1.37 (95% confidence interval 0.76 to 2.48).
Analysis 6.3.
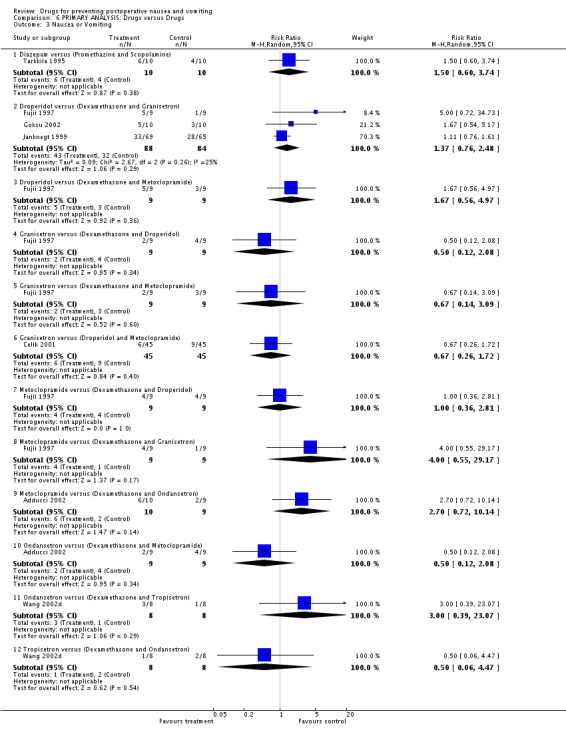
Comparison 6 PRIMARY ANALYSIS: Drugs versus Drugs, Outcome 3 Nausea or Vomiting.
6.4 Rescue antiemetic (Analysis 6.4) We calculated that there is no evidence that the risk of treatment for postoperative nausea or vomiting is different for the following drug comparisons ‐ relative risk (95% confidence interval): droperidol versus dexamethasone and granisetron 1.73 (0.79 to 3.81); ondansetron versus droperidol and metoclopramide 1.00 (0.38 to 2.63).
Analysis 6.4.

Comparison 6 PRIMARY ANALYSIS: Drugs versus Drugs, Outcome 4 Rescue antiemetic.
Primary analysis: the risk of side effects
Studies reported the postoperative risks of the following: abdominal pain (or bloating or constipation); agitation (or confusion or restlessness); bradycardia; dizziness (or vertigo); drowsiness (or sedation); dry mouth; extrapyramidal reaction; headache; infection; itch (or pruritus); shivering. Some studies reported the combined risks of: 'dizziness or headache'; 'dizziness or shivering'; 'drowsiness or headache'; 'dizziness or drowsiness or headache'. Three hundred and eighty studies reported how many participants experienced side effects, 148 studies said that they recorded side effects in the methodology but did not report the number who experienced a side effect, and 209 studies did not report side effects. We have performed a post‐hoc analysis that we did not list in the protocol (see 'Post‐hoc interstudy analysis: studies authored by Fujii et al' in Results and Discussion). Exclusion of results by Fujii et al did not alter the number of side effects caused by drugs but it did widen the confidence intervals.
Placebo versus drug
We calculated that the risk of side effects was changed by the following drugs compared to placebo ‐ relative risk (95% confidence interval): dizziness is increased by neostigmine 6.82 (1.31 to 35.41) and decreased by tropisetron 0.37 (0.14 to 0.96); drowsiness is increased by dimenhydrinate 9.01 (2.18 to 37.23) and by droperidol 1.32 (1.16 to 1.51); dry mouth is increased by hyoscine 1.25 (1.05 to 1.49); headache is decreased by droperidol 0.79 (0.65 to 0.95) and increased by ondansetron 1.16 (1.03 to 1.30).
We calculated that there is no evidence for a difference in the risk of any other side effect for a drug compared to placebo.
No treatment versus drug
We calculated that droperidol increased the risk of drowsiness compared to no treatment ‐ relative risk 2.57 (95% confidence interval 1.02 to 6.43).
Drug versus drug
We calculated that dimenhydrinate increased the risk of drowsiness compared to ondansetron ‐ relative risk 7.22 (95% confidence interval 1.52 to 34.36). We calculated that there is no evidence that the risk of any other side effect studied differs when drugs were compared.
Placebo versus drugs We calculated that there is no evidence that the risk of any side effect studied is increased by a combination of drugs compared to placebo.
No treatment versus drugs We calculated that there is no evidence that the risk of any side effect studied is increased by a combination of drugs compared to no treatment.
Drugs versus drugs We calculated that there is no evidence that the risk of any side effect studied is increased by a combination of drugs compared to another drug or combination of drugs.
Secondary analysis: the route of administration
Only one author (in four studies) assessed route of administration for a drug (van den Berg 1995; van den Berg 1996; van den Berg 1996b; van den Berg 1996c). We calculated that two outcomes are less common following intramuscular than intravenous prochlorperazine ‐ relative risk (95% confidence interval): nausea 0.53 (0.33 to 0.83); nausea or vomiting 0.78 (0.62 to 0.97).
Secondary analysis: the timing of drug administration
Only droperidol (Klockgether 1993; Korttila 1985; Kraus 1991; Nakata 2002) and ondansetron (Madan 2000; Polati 1995; Sun 1997c; Tang 1998; Trakya 1996) were studied. There was no evidence that the risk of postoperative nausea and vomiting differed for groups given ondansetron before induction, at induction, intraoperatively or postoperatively. Nausea and vomiting were treated more often after ondansetron had been given at induction than when it had been given intraoperatively ‐ relative risk 1.76 (95% confidence interval 1.12 to 2.76). There were no differences in outcomes when droperidol was given at different times.
Secondary analysis: the dose of drug
We found no evidence for the following drugs that the risk of any emetic outcome was affected by dose: alizapride; dolasetron; domperidone; ginger; tropisetron. The risk of at least one outcome was decreased by larger doses of the drugs listed below.
Clonidine
The risks for two outcomes were greater after smaller doses of clonidine ‐ relative risk (95% confidence interval): vomiting 2.68 (1.17 to 6.16); 'nausea or vomiting' 3.41 (1.34 to 8.71). When we only analysed the effect of doubling the dose of clonidine only the risk for 'nausea or vomiting' 1.41 (1.05 to 1.88) was greater with half the dose (Bock 2002; Carabine 1992; Grottke 2003; Mikawa 1995; Paech 1997; Sites 2003).
Dexamethasone
The risks for most outcomes were greater after smaller doses of dexamethasone ‐ relative risk (95% confidence interval): vomiting 1.57 (1.07 to 2.30); nausea or vomiting 1.44 (1.10 to 1.90); nausea 1.41 (0.98 to 2.03); treatment 1.48 (1.00 to 2.20). When we only analysed the effect of doubling the dose of dexamethasone the risks for both nausea 1.51 (1.02 to 2.24) and 'nausea or vomiting' 1.41 (1.05 to 1.88) were greater with half the dose (Elhakim 2002; Fujii 2002; Ho 2001; Lee 2001; Liu 1999; Wang 2000c; Wang 2001).
Droperidol
The risks for all outcomes were greater after smaller doses of droperidol ‐ relative risk (95% confidence interval): nausea 1.23 (1.12 to 1.36); vomiting 1.26 (1.01 to 1.57); nausea or vomiting 1.20 (1.08 to 1.33); treatment 1.21 (1.02 to 1.44). When we only analysed the effect of doubling the dose of droperidol we found that the relative risks stayed about the same: nausea 1.28 (1.05 to 1.56); vomiting 1.33 (1.01 to 1.77); nausea or vomiting 1.20 (1.07 to 1.34); treatment 1.22 (1.02 to 1.46) (Beattie 1993; Brown 1991; Culebras 2003; Eustis 1987; Fortney 1998; Foster 1996; Fujii 1995b; Jorgensen 1990; Klahsen 1996; Koivuranta 1997; Korttila 1985; Lamond 1998; Lim 1991; Lim 1999; McKenzie 1995; Millar 1987; Morin 1999; Mortensen 1982; Nicolson 1988; O'Donovan 1984; Spadafora 1994; Stead 1994; Tang 1996; TerRiet 1997; Tripple 1989).
Granisetron
The risks for all outcomes were greater after smaller doses of granisetron ‐ relative risk (95% confidence interval): nausea 1.21 (1.05 to 1.40); vomiting 1.50 (1.26 to 1.79); nausea or vomiting 1.50 (1.19 to 1.89); treatment 1.66 (1.15 to 2.40). When we only analysed the effect of doubling the dose of granisetron, the risks for vomiting 1.64 (1.23 to 2.20), 'nausea or vomiting' 2.12 (1.48 to 3.05) and treatment 2.10 (1.21 to 3.66) were greater with half the dose. Removal of studies by Fujii removes any effect of dose on outcome (please see post‐hoc analysis and Discussion) (Cieslak 1996; Fujii 1994b; Fujii 1996e; Fujii 1997f; Fujii 1998o; Fujii 1998q; Fujii 1998r; Fujii 1998s; Fujii 1998t; Fujii 1999L; Fujii 1999n; Fujii 2001f; Fujii 2001g; Fujii 2002b; Fujii 2002b; McAllister 1996; Mikawa 1995b; Mikawa 1997b; Munro 1999; Wilson 1996).
Metoclopramide
The risk for vomiting was greater after smaller doses of metoclopramide ‐ relative risk 1.82 (95% confidence interval 1.16 to 2.87) but was not when we only analysed the effect of doubling the dose of metoclopramide (Diamond 1988; Lin 1992; Vollmer 1988).
Ondansetron
The risks for most outcomes were greater after smaller doses of ondansetron ‐ relative risk (95% confidence interval): vomiting 1.13 (1.02 to 1.26); nausea or vomiting 1.39 (1.08 to 1.79); nausea 1.07 (1.00 to 1.15). When we only analysed the effect of doubling the dose of ondansetron, these differences disappeared except for the outcome 'nausea or vomiting': nausea 1.07 (0.97 to 1.18); vomiting 1.08 (0.97 to 1.20); nausea or vomiting 1.43 (1.08 to 1.90); treatment 1.22 (0.85 to 1.74) (Alon 1993b; Bowhay 2001; Charuluxananan 2003; Davis 1995b; Dershwitz 1998; Goodarzi 1998; Helmers 1993; Honkavaara 1996b; Lawhorn 1997; Le Roy 1995; Paventi 2001; Pearman 1994; Principi 1996; Rose 1996b; Rust 1994; Sadhasivam 2000; Saur 1996; Splinter 1997c; TerRiet 1997; Trakya 1996; Tur 1995; Watcha 1995b; Zarate 2000).
Ramosetron
Two outcomes were more frequent after smaller doses of ramosetron (half the dose) ‐ relative risk (95% confidence interval): vomiting 2.12 (1.05 to 4.27); nausea or vomiting 2.20 (1.23 to 3.92) (Fujii 2000c; Fujii 2002e; Fujii 2003). Please see the post‐hoc analysis and Discussion.
The risk of 'nausea or vomiting' was less with smaller doses of neostigmine ‐ relative risk 0.66 (95%CI 0.47 to 0.91).
Interstudy analyses: subgroup analyses, sensitivity analyses and post‐hoc analyses
We examined the effect of subgrouping studies using the following four variables:
the age of the participant;
the type of surgery;
the sex of the participant;
the timing of antiemetic used (before, during, or after the operation, or at induction).
We examined the effect of each variable using the same outcomes that we used for the main analyses: nausea; vomiting; nausea or vomiting; rescue antiemetic. We compared subgroups that contained at least two studies. We compared the 95% confidence intervals of the treatment effect and we interpreted the absence of overlap as an indication that the treatment effect differed significantly between subgroups.
Subgroup analysis: the age of the participant
There were no consistent differences in the effects of any drug on any outcome when studies were subgrouped on the basis of participant age ‐ children or adults. Subgrouping studies by age did not decrease statistical heterogeneity. The confidence intervals for all outcomes in children and adults overlapped, except for two outcomes with ondansetron that were prevented more in children than adults ‐ relative risk (95% confidence interval): vomiting 0.49 (0.44 to 0.53) compared with 0.62 (0.59 to 0.65) in adults; treatment 0.35 (0.29 to 0.42) compared with 0.54 (0.51 to 0.58) in adults. And one outcome with tropisetron that was prevented more in children than adults: treatment 0.44 (0.35 to 0.56) compared with 0.67 (0.63 to 0.71) in adults.
Subgroup analysis: the type of operation
There were no differences in the effects of any drug on any outcome when studies were subgrouped on the basis of type of surgery: dental; otorhinolaryngological (ENT); general; gynaecological; maxillofacial; neurosurgical; obstetrical; ophthalmological; orthopaedic; plastic; urological. Subgrouping studies by type of operation did not decrease statistical heterogeneity. The only exception was the risk of nausea after granisetron versus placebo that differed in three comparisons. The relative risk in studies of neurosurgical participants was 0.94 (0.71 to 1.25) compared to 0.38 (0.21 to 0.67) in studies of ENT participants, 0.47 (0.35 to 0.65) in studies of gynaecological participants and 0.48 (0.38 to 0.61) in studies of general surgical participants. These three isolated differences contrast with the remaining 354 comparisons that showed no effect of type of operation (summarized in Additional Table 17).
Table 4.
Subgroup analysis: type of operation; placebo versus drug
| Outcome: specialty | Clonidine | Dexamethasone | Dolasetron | Droperidol | Granisetron | Metoclopramide | Ondansetron | Tropisetron |
| Clonidine | Dexamethasone | Dolasetron | Droperidol | Granisetron | Metoclopramide | Ondansetron | Tropisetron | |
| Nausea: dental | 0.52 (0.30 to 0.90) | 0.73 (0.24 to 2.20) | ||||||
| Nausea: ENT | 0.51 (0.36 to 0.71) | 0.63 (0.49 to 0.81) | 0.38 (0.21 to 0.67) | 0.89 (0.64 to 1.25) | 0.73 (0.62 to 0.85) | |||
| Nausea: general | 0.59 (0.48 to 0.72) | 0.75 (0.33 to 1.70) | 0.64 (0.48 to 0.84) | 0.47 (0.35 to 0.65) | 0.86 (0.62 to 1.19) | 0.72 (0.59 to 0.88) | 0.70 (0.54 to 0.90) | |
| Nausea: gynaecological | 0.56 (0.36 to 0.88) | 0.81 (0.71 to 0.93) | 0.70 (0.58 to 0.84) | 0.48 (0.38 to 0.61) | 0.86 (0.77 to 0.96) | 0.66 (0.57 to 0.76) | 0.76 (0.66 to 0.87) | |
| Nausea: neurosurgical | 0.94 (0.71 to 1.25) | 0.88 (0.56 to 1.38) | ||||||
| Nausea: obstetrical | 0.61 (0.43 to 0.87) | 0.50 (0.34 to 0.73) | 0.69 (0.48 to 0.99) | 0.41 (0.25 to 0.65) | ||||
| Nausea: ophthalmological | 0.59 (0.20 to 1.75) | 0.56 (0.31 to 1.01) | 0.29 (0.06 to 1.39) | |||||
| Nausea: orthopaedic | 0.64 (0.31 to 1.31) | 0.39 (0.24 to 0.64) | 0.54 (0.41 to 0.71) | 0.70 (0.51 to 0.94) | 0.82 (0.50 to 1.33) | |||
| Nausea: plastic | 0.82 (0.50 to 1.36) | |||||||
| Vomiting: dental | 0.38 (0.02 to 6.98) | 0.73 (0.38 to 1.40) | 0.39 (0.08 to 1.85) | |||||
| Vomiting: ENT | 0.49 (0.41 to 0.60) | 0.62 (0.45 to 0.86) | 0.32 (0.23 to 0.44) | 0.80 (0.62 to 1.04) | 0.49 (0.39 to 0.61) | 0.53 (0.41 to 0.69) | ||
| Vomiting: general | 0.51 (0.40 to 0.66) | 0.30 (0.14 to 0.66) | 0.73 (0.55 to 0.96) | 0.43 (0.32 to 0.59) | 0.79 (0.60 to 1.04) | 0.55 (0.44 to 0.70) | 0.27 (0.11 to 0.67) | |
| Vomiting: gynaecological | 0.44 (0.35 to 0.55) | 0.57 (0.37 to 0.89) | 0.57 (0.46 to 0.71) | 0.40 (0.33 to 0.49) | 0.75 (0.65 to 0.86) | 0.61 (0.51 to 0.72) | 0.56 (0.45 to 0.71) | |
| Vomiting: maxillofacial | 1.19 (0.35 to 4.03) | |||||||
| Vomiting: neurosurgical | 0.48 (0.30 to 0.78) | 0.48 (0.29 to 0.81) | ||||||
| Vomiting: obstetrical | 0.66 (0.46 to 0.94) | 0.54 (0.35 to 0.85) | 0.65 (0.38 to 1.09) | 0.55 (0.25 to 1.22) | ||||
| Vomiting: ophthalmological | 0.77 (0.50 to 1.18) | 0.54 (0.35 to 0.83) | 0.62 (0.51 to 0.75) | 0.41 (0.31 to 0.54) | 0.73 (0.59 to 0.89) | 0.47 (0.32 to 0.70) | ||
| Vomiting: orthopaedic | 0.91 (0.45 to 1.84) | 0.32 (0.15 to 0.69) | 0.50 (0.41 to 0.63) | 0.69 (0.49 to 0.97) | 0.48 (0.30 to 0.76) | |||
| Vomiting: plastic | 0.44 (0.26 to 0.73) | |||||||
| Vomiting: urological | 0.63 (0.28 to 1.39) | |||||||
| Nausea or Vomiting: ENT | 0.58 (0.41 to 0.81) | 0.51 (0.32 to 0.82) | 0.29 (0.15 to 0.58) | 0.82 (0.50 to 1.39) | 0.54 (0.42 to 0.70) | |||
| Nausea or Vomiting: general | 0.47 (0.29 to 0.76) | 0.57 (0.44 to 0.74) | 0.46 (0.37 to 0.56) | 0.83 (0.68 to 1.02) | 0.58 (0.45 to 0.74) | 0.55 (0.44 to 0.63) | ||
| Nausea or Vomiting: gynaecological | 0.46 (0.38 to 0.55) | 0.81 (0.74 to 0.89) | 0.61 (0.52 to 0.71) | 0.37 (0.28 to 0.47) | 0.77 (0.65 to 0.90) | 0.63 (0.53 to 0.76) | 0.60 (0.43 to 0.84) | |
| Nausea or Vomiting: neurosurgical | 0.48 (0.29 to 0.80) | |||||||
| Nausea or Vomiting: obstetrical | 0.46 (0.35 to 0.62) | 0.53 (0.40 to 0.71) | 0.75 (0.54 to 1.03) | 0.44 (0.31 to 0.62) | ||||
| Nausea or Vomiting: ophthalmological | 0.56 (0.39 to 0.79) | 0.89 (0.67 to 1.18) | 0.51 (0.44 to 0.61) | |||||
| Nausea or Vomitiing: orthopaedic | 0.34 (0.22 to 0.52) | 0.56 (0.45 to 0.70) | 0.72 (0.52 to 1.00) | 0.48 (0.35 to 0.65) | ||||
| Rescue antiemetic: dental | 0.17 (0.03 to 0.91) | |||||||
| Rescue antiemetic: ENT | 0.47 (0.28 to 0.80) | 0.70 (0.48 to 1.02) | 0.19 (0.11 to 0.34) | 0.86 (0.63 to 1.15) | 0.53 (0.42 to 0.67) | 0.30 (0.03 to 3.46) | ||
| Rescue antiemetic: general | 0.40 (0.30 to 0.53) | 0.49 (0.31 to 0.76) | 0.30 (0.20 to 0.45) | 0.84 (0.61 to 1.17) | 0.67 (0.50 to 0.89) | 0.56 (0.36 to 0.88) | ||
| Rescue antiemetic: gynaecological | 0.44 (0.31 to 0.62) | 0.69 (0.55 to 0.85) | 0.46 (0.32 to 0.67) | 0.32 (0.23 to 0.46) | 0.71 (0.57 to 0.89) | 0.62 (0.53 to 0.73) | 0.63 (0.47 to 0.83) | |
| Rescue antiemetic: neurosurgical | 0.63 (0.36 to 1.10) | |||||||
| Rescue antiemetic: obstetrical | 0.61 (0.39 to 0.96) | 0.45 (0.25 to 0.81) | 0.49 (0.18 to 1.31) | 0.23 (0.09 to 0.58) | ||||
| Rescue antiemetic: ophthalmological | 0.37 (0.19 to 0.71) | 0.36 (0.20 to 0.64) | 0.56 (0.22 to 1.40) | 0.44 (0.35 to 0.55) | 0.52 (0.21 to 1.26) | |||
| Rescue antiemetic: orthopaedic | 1.13 (0.76 to 1.67) | 0.35 (0.21 to 0.59) | 0.55 (0.44 to 0.69) | 0.78 (0.59 to 1.02) | 0.45 (0.30 to 0.66) | 1.05 (0.55 to 1.98) |
Subgroup analysis: the sex of the participant
There were no differences in the effects of any drug on any outcome when studies were subgrouped on the basis of participant sex: male (men or boys) or female (women or girls). Subgrouping studies by sex did not decrease statistical heterogeneity.
Subgroup analysis: the time of drug administration
There were no consistent differences in the effects of any drug on any outcome when studies were subgrouped on the basis of timing of administration (preoperatively, at induction, intraoperatively, postoperatively). Subgrouping studies did not reduce statistical heterogeneity. Only three of 245 subgroup comparisons suggested a possible effect of timing (95% confidence intervals overlapped for the other 242 comparisons). It is possible that when ondansetron is given late (after the participant awoke from anaesthesia) it fails to prevent nausea ‐ relative risk (95% confidence interval): preoperative 0.67 (0.54 to 0.84); induction 0.68 (0.61 to 0.76); intraoperative 0.61 (0.48 to 0.78); postoperative 1.17 (0.93 to 1.48).
Sensitivity analysis: measures of methodological quality
There were no differences in the effects of any drug on any outcome when studies were subgrouped on the basis of: allocation concealment; sequence generation; blinding of outcome assessor; blinding of anaesthetist. Subgrouping studies by methodological quality did not decrease statistical heterogeneity.
Post‐hoc interstudy analysis: studies authored by Fujii et al
We performed this subgroup analysis because studies of granisetron authored by Fujii et al have been criticized (please see Discussion for details). 13.1 to 13.4 Placebo versus Granisetron (Analysis 13.1 to Analysis 13.4) There was no consistent difference in the results of studies authored by Fujii and other studies. The effect of granisetron in both groups was similar for two outcomes ‐ relative risk (95% confidence interval): vomiting 0.38 (0.33 to 0.44) for 39 Fujii studies (2719 participants) compared with 0.42 (0.33 to 0.54) for the other 12 studies (1369 participants); nausea or vomiting 0.41 (0.36 to 0.47) for 27 Fujii studies (1908 participants) compared with 0.53 (0.35 to 0.80) for the other seven studies (744 participants). The corresponding P values from interaction analyses are 0.25 and 0.50 respectively. There were differences for the other two outcomes; nausea 0.42 (0.34 to 0.53) for 28 Fujii studies (1839 participants) compared with 0.67 (0.55 to 0.81) for the other nine studies (1091 participants); treatment 0.23 (0.17 to 0.30) for 30 Fujii studies (2413 participants) compared with 0.48 (0.34 to 0.69) for the other nine studies (997 participants). The corresponding P values from interaction analyses are 0.002 and 0.001.
Analysis 13.1.

Comparison 13 POSTHOC ANALYSIS: Fujii et al versus other authors, Outcome 1 Nausea: granisetron.
Analysis 13.4.

Comparison 13 POSTHOC ANALYSIS: Fujii et al versus other authors, Outcome 4 Rescue antiemetic: granisetron.
The Funnel plot for granisetron (versus placebo) appeared to be the most asymmetric of any drug. Therefore the effect of granisetron may be overestimated more than any other drug (see additional Figure 1 and Figure 2).
Figure 1.

Severe Funnel plot asymmetry: granisetron's effectiveness versus placebo is less than implied by the relative risk. [Each dot is an outcome from one study. Nausea is green. Vomiting is light blue. 'Nausea or Vomiting' is dark blue. Rescue antiemetic is pink. Dots overlap. Coloured vertical lines mark the summative relative risk for each outcome. The outcomes of dots closer to the top (SE 0.0) are more precise]
Figure 2.

Funnel plot of studies of granisetron versus placebo (compare with additional figure 02). In this plot results from studies authored by Fujii et al are red; results from other studies are blue. The vertical lines are the corresponding estimates of effect: green is nausea; light blue is vomiting; dark blue is nausea or vomiting; pink is treatment.
13.5 to 13.8 Droperidol versus Granisetron (Analysis 13.5 to Analysis 13.8) There was no consistent difference in the results of studies authored by Fujii and other studies. The effect of droperidol versus granisetron was similar in the two groups for two outcomes ‐ relative risk (95% confidence interval): vomiting 2.42 (1.82 to 3.22) for 21 Fujii studies (838 participants) compared with 1.70 (1.14 to 2.55) for the other three studies (170 participants); nausea or vomiting 2.43 (1.84 to 3.22) for 15 Fujii studies (574 participants) compared with 1.22 (0.61 to 2.48) for the other three studies (170 participants). The corresponding P values from interaction analyses are 0.16 and 0.08 respectively (Altman 2003). There were differences for the other two outcomes; nausea 2.33 (1.54 to 3.52) for 16 Fujii studies (612 participants) compared with 0.94 (0.67 to 1.33) for the other three studies (170 participants); treatment 5.10 (2.75 to 9.44) for 17 Fujii studies (700 participants) compared with 1.63 (0.91 to 2.89) for the other two studies (150 participants). The corresponding P values from interaction analyses are 0.001 and 0.008 respectively. The Funnel plot for droperidol versus granisetron appeared to be asymmetric. Therefore the effect of granisetron may be overestimated (see additional Figure 3 and Figure 4).
Analysis 13.5.

Comparison 13 POSTHOC ANALYSIS: Fujii et al versus other authors, Outcome 5 Nausea: droperidol versus granisetron.
Analysis 13.8.

Comparison 13 POSTHOC ANALYSIS: Fujii et al versus other authors, Outcome 8 Rescue antiemetic: droperidol versus granisetron.
Figure 3.

Severe Funnel plot asymmetry: droperidol and granisetron's effectiveness are more similar than implied by the relative risk. [Each dot is an outcome from one study. Nausea is green. Vomiting is light blue. 'Nausea or Vomiting' is dark blue. Rescue antiemetic is pink. Dots overlap. Coloured vertical lines mark the summative relative risk for each outcome. The outcomes of dots closer to the top (SE 0.0) are more precise]
Figure 4.

Funnel plot of studies of droperidol versus granisetron (compare with additional figure 09). In this plot results from studies authored by Fujii et al are red; results from other studies are blue. The vertical lines are the corresponding estimates of effect: green is nausea; light blue is vomiting; dark blue is nausea or vomiting; pink is treatment.
13.9 Risk of side effects (Analysis 13.9)
Analysis 13.9.

Comparison 13 POSTHOC ANALYSIS: Fujii et al versus other authors, Outcome 9 Side effects.
There were no differences in the effect estimates for side effects reported by Fujii et al compared to other authors. The pattern of relative risks reported by Fujii et al appeared different to other studies. Fujii reported exactly the same risks of side effects for most groups in each study. Table of comparison 13.09 lists the results of 261 studies in which side effects were assessed by Fujii and other authors. The risk for 140 of these 261 side effects was exactly the same in each group (the relative risk is exactly 1.0). The relative risk was exactly one in 128 of the 159 studies published by Fujii and exactly one in 12 of the 102 studies published by other authors (see additional Figure 5).
Figure 5.

One relative risk is plotted for each side‐effect reported by each study: red are from studies by Fujii et al; blue are from studies by other authors. An equal risk for a side effect (relative risk one) was reported by Fujii et al for 128 out of 159 risks (they overlap on the dashed line RR=1 and so appear fewer than 128), and reported by other authors for 12 out of 102 risks.
Post‐hoc interstudy analysis: giving one antiemetic with another
(Additional Table 18) The IMPACT study (Apfel 2004) found that the effects of three drugs (dexamethasone, droperidol, ondansetron) were independent of whether the drug was given alone or with one or both of the other two drugs. To illustrate this result, consider a drug that confers a relative risk for PONV of 0.65 when compared with placebo. One can infer from the results of the IMPACT study that the drug would still confer a relative risk of 0.65 if it is given with another drug. If the second drug confers a relative risk of 0.7 for PONV (compared to placebo) the combined relative risk for PONV is 0.65 multiplied by 0.7, equalling 0.46. In summary, relative risks of the three drugs assessed in the IMPACT study were unaffected by coadministration with another drug.
Table 5.
Posthoc analysis: control with or without antiemetic
| Comparison | Nausea | Vomiting | Nausea or Vomiting | Rescue antiemetic | 95% CI overlap? |
| RR (95% CI) heterogeneity | RR (95% CI) heterogeneity | RR (95% CI) heterogeneity | RR (95% CI) heterogeneity | Outcomes overlapping | |
| CYCLIZINE | |||||
| Control did not receive antiemetic | 0.65 (0.46 to 0.92) 0% | 0.56 (0.43 to 0.72) 0% | 0.62 (0.51 to 0.75) 22% | 0.29 (0.14 to 0.60) 0% | |
| Control did receive antiemetic | 0.83 (0.28 to 2.44) na | 0.17 (0.02 to 1.30) na | 0.77 (0.53 to 1.13) na | 0.10 (0.01 to 0.73) na | Yes (4/4) |
| Total | 0.67 (0.48 to 0.92) 0% | 0.54 (0.42 to 0.70) 5% | 0.64 (0.54 to 0.76) 5% | 0.24 (0.12 to 0.47) 0% | |
| DEXAMETHASONE | |||||
| Control did not receive antiemetic | 0.62 (0.55 to 0.69) 80% | 0.47 (0.42 to 0.53) 11% | 0.43 (0.38 to 0.48) 0% | 0.49 (0.43 to 0.55) 45% | |
| Control did receive antiemetic | 0.59 (0.47 to 0.74) 0% | 0.43 (0.33 to 0.57) 0% | 0.55 (0.47 to 0.65) 15% | 0.62 (0.45 to 0.84) 13% | Yes (4/4) |
| Total | 0.61 (0.55 to 0.68) 71% | 0.46 (0.42 to 0.52) 0% | 0.47 (0.43 to 0.51) 16% | 0.50 (0.45 to 0.57) 39% | |
| DOLASETRON | |||||
| Control did not receive antiemetic | 0.81 (0.74 to 0.88) 0% | 0.70 (0.64 to 0.78) 52% | 0.77 (0.71 to 0.83) 37% | 0.68 (0.60 to 0.76) 24% | |
| Control did receive antiemetic | 0.42 (0.13 to 1.40) 5% | 0.27 (0.08 to 0.90) 0% | 0.30 (0.12 to 0.73) 0% | na | Yes (3/3) |
| Total | 0.81 (0.74 to 0.88) 0% | 0.69 (0.63 to 0.77) 50% | 0.76 (0.70 to 0.82) 44% | 0.68 (0.60 to 0.76) 24% | |
| DROPERIDOL | |||||
| Control did not receive antiemetic | 0.66 (0.62 to 0.70) 58% | 0.59 (0.55 to 0.64) 8% | 0.63 (0.59 to 0.68) 12% | 0.48 (0.44 to 0.53) 22% | |
| Control did receive antiemetic | 0.58 (0.46 to 0.75): 0% | 0.55 (0.43 to 0.71) 0% | 0.46 (0.36 to 0.60) 0% | 0.49 (0.35 to 0.69) 37% | Yes (4/4) |
| Total | 0.65 (0.62 to 0.69) 38% | 0.59 (0.55 to 0.64) 0% | 0.62 (0.58 to 0.66) 10% | 0.48 (0.44 to 0.52) 24% | |
| GRANISETRON | |||||
| Control did not receive antiemetic | 0.60 (0.55 to 0.66) 29% | 0.43 (0.39 to 0.48) 12% | 0.50 (0.46 to 0.56) 58% | 0.35 (0.30 to 0.42) 52% | |
| Control did receive antiemetic | 0.22 (0.12 to 0.41) 0% | 0.16 (0.09 to 0.29) 0% | 0.16 (0.09 to 0.28) 0% | 0.10 (0.04 to 0.27) 0% | No (0/4) |
| Total | 0.56 (0.51 to 0.62) 40% | 0.41 (0.37 to 0.45) 0% | 0.46 (0.42 to 0.51) 65% | 0.33 (0.28 to 0.38) 50% | |
| METOCLOPRAMIDE | |||||
| Control did not receive antiemetic | 0.78 (0.71 to 0.85) 0% | 0.72 (0.66 to 0.78) 0% | 0.73 (0.67 to 0.79) 0% | 0.74 (0.64 to 0.84) 0% | |
| Control did receive antiemetic | 0.76 (0.47 to 1.22) 0% | 0.96 (0.65 to 1.42) 0% | 0.83 (0.52 to 1.31) 0% | 0.87 (0.54 to 1.41) 0% | Yes (4/4) |
| Total | 0.78 (0.71 to 0.85) 0% | 0.73 (0.67 to 0.79) 0% | 0.73 (0.67 to 0.79) 0% | 0.74 (0.65 to 0.85) 0% | |
| ONDANSETRON | |||||
| Control did not receive antiemetic | 0.73 (0.71 to 0.75) 81% | 0.60 (0.57 to 0.62) 68% | 0.55 (0.52 to 0.58) 77% | 0.51 (0.48 to 0.54) 57% | |
| Control did receive antiemetic | 0.61 (0.48 to 0.78) 0% | 0.56 (0.42 to 0.74) 0% | 0.66 (0.54 to 0.82) 2% | 0.62 (0.46 to 0.82) 0% | Yes (4/4) |
| Total | 0.73 (0.70 to 0.75) 79% | 0.59 (0.57 to 0.62) 65% | 0.55 (0.53 to 0.58) 75% | 0.52 (0.49 to 0.55) 52% | |
| TROPISETRON | |||||
| Control did not receive antiemetic | 0.76 (0.69 to 0.83) 0% | 0.62 (0.56 to 0.69) 44% | 0.72 (0.64 to 0.80) 15% | 0.62 (0.54 to 0.71) 4% | |
| Control did receive antiemetic | na | na | 0.39 (0.19 to 0.78) na | 0.20 (0.05 to 0.89) na | Yes (2/2) |
| Total | 0.76 (0.69 to 0.83) 0% | 0.62 (0.56 to 0.69) 44% | 0.69 (0.62 to 0.78) 24% | 0.61 (0.53 to 0.70) 9% |
We performed a post‐hoc subgroup analysis to determine whether our results were consistent with the results of the IMPACT study. This subgroup analysis compared the relative risks in studies that gave an antiemetic drug by itself with studies that coadministered an antiemetic drug with another. This subgroup analysis is an indirect comparison and so the results are less reliable than the direct comparison of the IMPACT study.
Our results were consistent with the IMPACT study. We did not find any evidence that the relative risk conferred by one drug was affected by coadministration with another drug. The only exception was granisetron, which was more effective when given with dexamethasone (please see the Discussion).
Discussion
How effective is a drug?
A drug may appear to have different effects on the risks of nausea, vomiting, nausea or vomiting, and treatment. This systematic review cannot determine whether such differences were chance, due to systematic differences in measurement or systematic differences in effect. It may be more difficult to 'measure' nausea because it is an internal symptom and its measurement depends upon communication between the participant and the assessor. Rescue treatment also depends upon someone choosing whether or not to give a rescue antiemetic. For most comparisons in this review, the 95% confidence intervals overlap for all four outcomes. The decision to administer a prophylactic antiemetic should probably use the most precise measure of effect from the four outcomes (narrowest 95% confidence interval).
Please see the section "Which drug is best?" below for a discussion of whether one drug prevented nausea and vomiting more than another drug. This question is properly answered through direct comparisons of drugs within randomized controlled trials. The differences between the results of different drugs in different studies cannot be taken as reliable evidence that one drug is more effective versus placebo than another.
Are the calculated effects overestimates?
Funnel plot asymmetry may be caused by: selection bias (publication or location bias); poor methodological quality of smaller studies (design, analysis or fraud); true heterogeneity (varying effect with study size); artefact or chance. We interpret most Funnel plots of drug versus placebo as asymmetric. All of the asymmetric plots had an excess of imprecise studies, with relative risks that favoured the drug. The asymmetry was present for each outcome for most drugs. For example look at some of the Funnel plots for studies of a drug versus placebo. The Funnel plot for metoclopramide showed asymmetry that we think is mild (Figure 6); we think that the plots for droperidol (Figure 7) and ondansetron (Figure 8) showed moderate asymmetry; and we think that the plot for granisetron versus placebo showed the most asymmetry (Figure 1 and Figure 2).
Figure 6.

Mild Funnel plot asymmetry: metoclopramide's effectiveness versus placebo is similar to the relative risk (closer than other antiemetics). Each dot is an outcome from one study. Nausea is green. Vomiting is light blue. 'Nausea or Vomiting' is dark blue. Rescue antiemetic is pink. Dots overlap. Coloured vertical lines mark the summative relative risk for each outcome. The outcomes of dots closer to the top (SE 0.0) are more precise].
Figure 7.

Moderate Funnel plot asymmetry: droperidol's effectiveness versus placebo is less than implied by the relative risk. [Each dot is an outcome from one study. Nausea is green. Vomiting is light blue. 'Nausea or Vomiting' is dark blue. Rescue antiemetic is pink. Dots overlap. Coloured vertical lines mark the summative relative risk for each outcome. The outcomes of dots closer to the top (SE 0.0) are more precise]
Figure 8.

Moderate Funnel plot asymmetry: ondansetron's effectiveness versus placebo is less than implied by the relative risk. [Each dot is an outcome from one study. Nausea is green. Vomiting is light blue. 'Nausea or Vomiting' is dark blue. Rescue antiemetic is pink. Dots overlap. Coloured vertical lines mark the summative relative risk for each outcome. The outcomes of dots closer to the top (SE 0.0) are more precise]
Because this asymmetric effect was constant across all four outcomes and was present for many drugs, we discount chance and true heterogeneity as likely causes. We think that the cause is either publication bias or poor methodological quality of smaller studies. Either cause justifies an adjustment of the calculated results that we reported in the review. The results for the comparisons with the most asymmetric Funnel plots overestimate the effect the most.
Which drug is best?
We included 60 drugs. Twenty drugs had an effect compared to placebo (relative risk 95th percentile less than one) for at least one outcome (nausea, vomiting, 'nausea or vomiting', rescue antiemetic). We concentrate here on the nine drugs that had an effect on all four outcomes: cyclizine, dexamethasone, dolasetron, droperidol, granisetron, metoclopramide, ondansetron, ramosetron and tropisetron.
There are 36 ways of comparing two of nine drugs (see Additional Table 16). Twelve of the 36 comparisons have not been studied. Eleven of the 24 comparisons that have been studied produced evidence that one drug was more effective than another. Three comparisons have been studied extensively ‐ droperidol versus metoclopramide, droperidol versus ondansetron, metoclopramide versus ondansetron ‐ with between 22 and 45 studies contributing to each comparison. Because of the Funnel plot asymmetry for these comparisons, we cannot draw any conclusion as to whether these drugs differ in their ability to prevent PONV (additional Figure 9, Figure 10 and Figure 11). We believe that the likely causes for these asymmetries are the same as for comparisons of drugs versus placebo ‐ either publication bias or poor methodological quality of smaller studies.
Figure 9.

Severe Funnel plot asymmetry: metoclopramide and ondansetron's effectiveness are more similar than implied by the relative risk. [Each dot is an outcome from one study. Nausea is green. Vomiting is light blue. 'Nausea or Vomiting' is dark blue. Rescue antiemetic is pink. Dots overlap. Coloured vertical lines mark the summative relative risk for each outcome. The outcomes of dots closer to the top (SE 0.0) are more precise]
Figure 10.
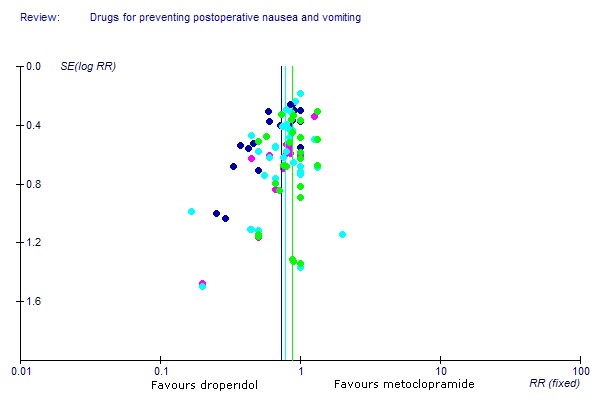
Moderate Funnel plot asymmetry: droperidol and metoclopramide's effectiveness are more similar than implied by the relative risks. [Each dot is an outcome from one study. Nausea green, Vomiting light blue, 'Nausea or vomiting' dark blue, Rescue antiemetic pink. Dots overlap. Coloured vertical lines mark the summative relative risk for each outcome. The outcomes of dots closer to the top (SE 0.0) are more precise]
Figure 11.

Moderate Funnel plot asymmetry: droperidol and ondansetron's effectiveness are more similar than implied by the relative risk for vomiting (light blue). [Each dot is an outcome from one study. Nausea green, Vomiting light blue, 'Nausea or vomiting' dark blue, Rescue antiemetic pink. Dots overlap. Coloured vertical lines mark the summative relative risk for each outcome. The outcomes of dots closer to the top (SE 0.0) are more precise]
Is more drug better than less drug?
We expected that the dose of a drug would have some effect on the risk of nausea or vomiting.
We found a consistent dose‐response for only one drug ‐ droperidol. When a larger dose of droperidol was given, the risk for PONV was decreased compared to smaller doses. Put another way, the risk for PONV was greater when less drug was given. We found only one or two studies for each dose comparison for droperidol. In isolation, few of these comparisons showed an effect of dose (the 95% confidence intervals included one). We found that when we combined studies that compared a dose of droperidol with a half‐dose, the risk for each outcome was greater by about 1.2 for the smaller dose: nausea 1.28 (1.05 to 1.56); vomiting 1.33 (1.01 to 1.77); nausea or vomiting 1.20 (1.07 to 1.34); treatment 1.22 (1.02 to 1.46). The results of this systematic review do not show a maximum dose above which droperidol has no further effect and they do not show whether the dose‐response is linear.
We calculated a dose‐response for at least one outcome for four drugs: clonidine, dexamethasone, granisetron and ondansetron. We do not know how to interpret the dose‐response for clonidine, because clonidine was not effective when we combined all placebo‐controlled studies. It is possible that higher doses of clonidine may have an effect when compared to placebo. We found that halving the dose of granisetron increased the risk for three outcomes: vomiting 1.64 (1.23 to 2.20), 'nausea or vomiting' 2.12 (1.48 to 3.05) and treatment 2.10 (1.21 to 3.66). We found that halving the dose of dexamethasone increased the risk of nausea by 1.5 (1.02 to 2.24) and 'nausea or vomiting' by 1.41 (1.05 to 1.88). We found that halving the dose of ondansetron increased the risk of 'nausea or vomiting' by 1.43 (1.08 to 1.90).
Overall we did not find convincing evidence that more drug is more effective for most of the drugs we assessed.
Is the timing of drug administration important?
There were few direct comparisons of timing of drug administration. There were few statistically different direct or indirect comparisons and these were sporadic. Timing may be important but there is no evidence at the moment to define what aspect of timing is important.
Does it matter what you give the drug with?
We did a post‐hoc analysis following the publication of the IMPACT study (Apfel 2004). This factorial study allowed the authors to assess the interaction of three antiemetic drugs (dexamethasone, droperidol, ondansetron) in 4083 participants. They found that the effect of each drug was independent of whether the drug was given alone or with one or both of the other two drugs.
We had decided to assume independence of effect in the design (protocol) of this systematic review. Imagine a study in which all participants received one antiemetic drug, and some participants are allocated to receive a second antiemetic drug. The participants who received one antiemetic (and a placebo for the second drug) were the control for participants that received the additional antiemetic. In Additional Table 18 we have tabulated a post‐hoc subgroup analysis of this decision. We have categorized studies into those in which the control group participants received a recognised antiemetic ("Control received antiemetic") and those that did not ("Control did not receive antiemetic"). The final column "95% CI overlap?" details whether the confidence intervals overlapped and for how many outcomes.
We calculated that the 95% confidence intervals for these two categories of studies overlapped for every outcome for every drug; that is there was no evidence that the effects of these drugs were altered by coadministration with other drugs. This is an indirect comparison and is not as reliable as the result from the IMPACT study.
The only exception was for the drug granisetron. The 95% confidence intervals did not overlap for any of the outcomes, and the relative risk for each outcome was less for the subgroup of studies that coadministered granisetron with another antiemetic. By itself this result would support research into the hypothesis that the effect of granisetron is synergistic with other antiemetics.
Do side effects outweigh benefits?
We don't know. Approximately one‐third of the studies we included did not report side effects. We think that the range of side effects reported was partly dependent on the authors' expectations. It is possible that there are rare side effects that are either too infrequent to be reported or did occur but were not reported because the authors did not consider them to be potential side effects of a drug.
Clinicians should use other sources of information about side effects. For instance, in 2001 the United States of America's Food and Drug Administration revised the labelling for droperidol with a black box warning for cases of electrocardiographic QT wave prolongation, torsades de pointes arrhythmia and death. The evidence and lack of evidence for this side effect have since been discussed and some clinicians do not think that the association is valid and causal (Kao 2003). In addition, a systematic review could include the results from all studies of these drugs versus placebo irrespective of the outcome being analysed, for instance the treatment of established nausea or vomiting and the prevention or treatment of nausea or vomiting after chemotherapy and radiotherapy.
Studies authored by Fujii et al
We read a number of criticisms of studies authored by Fujii et al in the course of preparing this systematic review. These criticisms focus on studies of granisetron and the reported risks of side effects (Apfel 1999; Kranke 1999c; Kranke 2000; Kranke 2001). We therefore performed subgroup analyses that compared studies authored by Fujii et al with other studies (comparisons 13.01 to 13.09). Analyses for interactions do not show consistent differences in effect for the two groups of studies, either for granisetron versus placebo or droperidol versus granisetron. The most important aspect of results for granisetron that readers should take into account is the marked asymmetry of the Funnel plots (see "Are the calculated effects overestimates?" and "Which drug is best?", additional Figure 1; Figure 2, Figure 3, and Figure 4). These asymmetries suggest that the effect of granisetron is overestimated (versus placebo) and that there is not a reliable difference between granisetron and other antiemetics. We believe that the results for side effects support our conclusion that the research priority is the incidence of important adverse events.
Is prophylactic antiemesis worthwhile?
Whether a prophylactic antiemetic drug causes net benefit or harm depends upon the risks and severities of the bad outcomes prevented (nausea, vomiting, sequelae, side effects of rescue antiemetic) and the unwanted outcomes caused (side effects, failure of expected benefit). The majority of people do not benefit when given a prophylactic antiemetic, but all are exposed to the risk of side effects. Attempts have been made to reduce the exposure of patients to unnecessary antiemetic prophylaxis by using scoring systems to predict the likelihood of postoperative nausea or vomiting, but in some circumstances these may not have useful predictive power (Van den Bosch 2005). We do not know of any attempt to predict the likelihood of a patient experiencing a side effect. We have presented the drug costs to prevent one episode of postoperative nausea or vomiting in Additional Table 19.
Table 6.
Drug costs to prevent one person experiencing PONV (£ or $ per event)
| Drug(s) | US$ per dose | UK£ per dose | Control incidence .2 | Control incidence .3 | Control incidence .4 | Control incidence .5 | Control incidence .6 | Control incidence .7 | Control incidence .8 |
| Metoclopramide 10mg [US pharmacy price] | $0.20 | £0.28 | £4.62 $3.30 | £3.08 $2.20 | £2.31 $1.64 | £1.85 $1.32 | £1.54 $1.10 | £1.32 $0.94 | £1.15 $0.82 |
| Cyclizine 50mg [meclizine 50mg: US pharmacy price] | $0.74 | £0.74 | £12.20 $12.20 | £8.13 $8.13 | £6.10 $6.10 | £4.88 $4.88 | £4.07 $4.07 | £3.49 $3.49 | £3.05 $3.05 |
| Dexamethasone 8mg [US pharmacy price] | $2.25 | £1.27 | £18.14 $32.14 | £12.10 $21.44 | £9.07 $16.07 | £7.26 $12.86 | £6.05 $10.72 | £5.18 $9.18 | £4.54 $8.05 |
| Ondansetron 4mg [US pharmacy price] | $21.23 | £6.45 | £92.14 $303.28 | £61.43 $202.20 | £46.07 $151.64 | £36.86 $121.32 | £30.71 $101.08 | £26.33 $86.66 | £23.04 $75.84 |
| Granisetron 1mg [US pharmacy price] | $46.87 | £11.46 | £188.90 $772.58 | £125.93 $515.04 | £94.45 $386.29 | £75.56 $309.03 | £62.97 $257.54 | £53.97 $220.73 | £47.23 $193.16 |
| Cyclizine 50mg and Metoclopramide 10mg [US pharmacy prices] | $0.94 | £1.02 | £11.66 $10.75 | £7.77 $7.16 | £5.83 $5.37 | £4.66 $4.29 | £3.89 $3.58 | £3.33 $3.07 | £2.91 $2.68 |
| Dexamethasone 8mg and Metoclopramide 10mg [US pharmacy prices | $2.45 | £1.55 | £15.12 $23.90 | £10.08 $15.93 | £7.56 $11.95 | £6.05 $9.56 | £5.04 $7.97 | £4.32 $6.83 | £3.78 $5.97 |
| Cyclizine 50mg and Dexamethasone 8mg and Metoclopramide 10mg | $3.19 | £2.29 | £18.05 $25.14 | £12.03 $16.76 | £9.02 $12.56 | £7.22 $10.06 | £6.02 $8.39 | £5.16 $7.19 | £4.51 $6.28 |
Does it matter what your age or sex is, what operation you are having or when you're given the antiemetic?
None of these variables alter the effect of antiemetic drugs in a consistent way. Subgrouping studies did not reduce statistical heterogeneity. It is possible that metaregression may reveal subtle effects. However, even in the absence of an effect on the prophylactic power of drugs, these variables may indeed 'matter', because the decision to administer an antiemetic depends upon the absolute effect of a drug (control rate of nausea or vomiting multiplied by the relative risk), not just on the relative risk alone. The control rate itself may be altered by these variables even if the relative risk is not.
Turning relative risks into decisions
The decision to use a prophylactic antiemetic drug depends upon the expected wanted and unwanted effects caused by the drug, on their risks and severities.
The efficacy and safety of a drug depend upon the risks of nausea and vomiting and of 'side effects' in an untreated population. We have prepared a selection of control group risks and calculated absolute risk reductions and numbers needed to treat (Additional Table 14). A commonly reported risk for postoperative nausea or vomiting is 30 out of 100 people. If an antiemetic is given to all 100 people before surgery 20 will still vomit or be nauseated, so 10 people will have benefited and 90 people will not. When the risk of nausea or vomiting is higher more people will benefit, so if the risk is 80 out of 100 people, 28 would benefit and 72 would not.
Authors' conclusions
Most patients given a drug to prevent nausea or vomiting after surgery will not benefit from it. Nausea or vomiting is reported to affect at most 80 out of 100 people after surgery. If all 100 of these people are given a drug, 28 would benefit, and 72 would not. Nausea and vomiting are usually less common and therefore drugs are less useful. For 100 people, of whom 30 would vomit or feel sick after surgery if given placebo, 10 people would benefit from a drug and 90 would not. Between one to five patients out of every 100 given a prophylactic antiemetic may expect to experience a mild side effect such as headache, sedation or dry mouth. There is convincing evidence that eight drugs reduce PONV by a similar amount: cyclizine, droperidol, granisetron, metoclopramide, ondansetron, tropisetron, dolasetron and dexamethasone. There is only limited evidence that more drug is more effective: there is convincing evidence that more drug is more effective for droperidol and limited evidence for dexamethasone and ondansetron. Evidence for differences in the efficacy of these eight drugs is not convincing.
There are some questions that are inadequately answered by the evidence that we have reviewed.
What are the types and risks of side effects experienced by patients exposed to prophylactic antiemetics?
Are cyclizine, droperidol, granisetron, metoclopramide, ondansetron, tropisetron, dolasetron and dexamethasone equipotent?
Are there reproducible dose‐effect relationships for these drugs?
Are clonidine, dimenhydrinate, hyoscine, prochlorperazine and ramosetron effective prophylactic antiemetics?
Do other drugs included in this review prevent PONV, such as alizapride, benzodiazepines, perphenazine and promethazine?
Because the majority of patients do not benefit from exposure to prophylactic antiemetics, the most important question to answer is "What are the types and risks of side effects experienced by patients exposed to prophylactic antiemetics?". Although we have produced evidence for the risks of 'mild' adverse outcomes in this review, such as constipation, dizziness, itchiness and sedation, there were little data on more severe outcomes such as infection, cardiac arrhythmia, extrapyramidal reactions and death. This information will probably require multicentre international collaboration to acquire because these side effects, if they occur, will be rare. Researchers may be tempted to answer easier but less important questions.
Researchers should use Additional Table 16 to decide what drugs to compare. Researchers should aim to conduct single studies that compare all eight drugs, as results from such studies will be most reliable. Potential researchers should conduct realistic sample size calculations before committing to research. Researchers should remember to distinguish between masking of allocation and generation of a (random) allocation sequence. When reporting results, authors should list all quality criteria clearly. Readers of studies should be able to determine how many people were nauseated, how many vomited, how many were both nauseated and vomited, and how many vomited without experiencing nausea. Authors should specify how they categorized retching and, again, readers should be able to determine how many people retched and whether they experienced any other symptoms. Authors should report how many people feel nauseated or vomit before operation (because drug administration would be treatment, not prophylaxis). Authors should report the risk of each outcome for the total observation period: if authors report risks during two or more periods, readers should be able to determine how many people experienced an outcome in each period and how many periods each person experienced an outcome for. Otherwise the units of analysis are unclear.
Feedback
Feedback submitted: 04‐Feb‐2014
Summary
The review is out of date, and needs to be updated with included summary of findings table, and risk of bias figures.
Due to the very large nature of the topic, it is very difficult to find specific information from the review. Perhaps dividing it into smaller more specific reviews would allow scientific questions to be more easily answered, and would also allow a shorter time to update each review.
In terms of outcomes, we do not see the clinical utility of having separate analyses for nausea, vomiting, and nausea or vomiting.
In presentation of data, the paragaphs of listed drugs with results is not an effective way to display results, perhaps tables would be easier to read.
The overall recommendation was against the use of routine PONV prophylaxis, unless the patient has a high risk. However in the recommendations there was no suggestions as to what would put a person at higher risk (are we to assume the usual risk factors? did the outcomes in the RCTs reflect this?). In order to assess the usefulness of the review, we used the example of answering the question: does ondansetron prevent post‐operative nausea or vomiting in a certain population (ex. gyne procedures).
The trials in the results forest plot showed a very high level of heterogeneity, thus results are questionable as to how this can be applicable to real life. Sub‐group analysis within the forest plot could allow for more specific structure of information and answer more specific questions in terms of either patient population of the RCT, dose of drug used, or procedure that was performed.No recommended dosage was given (or easily found) although the drug was shown to be efficacious.
Reply
The author agrees with this comment.
The author disagrees with this comment. The summary (particularly when tabulated as suggested in point 1.) can concisely communicate the results of the review. The review is answering a simple question: the outcomes assessed (PONV) are the same for all the drugs. An update might identify around 13 effective drugs, the effects of which can still be communicated simply. There were few side‐effects that achieved statistical significance, so these do not complicate the summary findings.The question being addressed by the review – that might be asked by patients or clinicians – is ‘what drugs can reduce the rate of PONV and by how much?’ and ‘what are their relative effects and side effects?’ This question is very difficult to answer if a patient or clinician has to hunt down multiple systematic reviews that have only looked at one or two drugs. Division of this review into multiple smaller reviews doesn’t allow for meta‐regression or indirect comparisons, it does not so easily permit the identification of fraudulent research or bias.
The systematic review reflects the outcomes that authors of RCTs have chosen to report. Some patients dislike vomiting but don’t mind being nauseated (so much), whilst other patients don’t mind vomiting as long as they don’t feel nauseated (much): not all patients who vomit are recorded as complaining of nausea. For these reasons separate analyses are helpful.
This will be considered in the update.
No, there was no recommendation. The summary factually stated how many patients might be expected to benefit and how many would not.
Due to lack of capacity for the Cochrane Library (in 2006) to accommodate subgroup analyses we did not display the various subgroup results, although we did report that type of surgery was not associated with interactions with antiemetic effects. Subgroup analyses did not reduce heterogeneity. There was also no interaction with age (adult vs child), sex (male vs female) and other factors as reported. We also reported on the effects of dosage.
Contributors
Summary: Andrew Wilson, Senior Student, University of British Columbia Medical School. I certify that I have no affiliations with or involvement in any organization or entity with a financial interest in the subject matter of my feedback.
Reply: John Carlisle, lead author.
Acknowledgements
The authors thank the editorial team, particularly Dr Mathew Zacharias, Professor Nathan Pace and Jane Cracknell. We also thank Antoinette Bradley for retrieving most of the papers. We thank those who helped translate studies. And John Carlisle thanks Joanna Carlisle for her patience and begs her forgiveness for the update.
Appendices
Appendix 1. Search strategy
MeSH‐NAUSEA OR NAUSEA* OR INAPPETENCE
MeSH‐VOMITING OR VOMIT* OR EMESIS OR EMET*
MeSH‐POSTOPERATIVE NAUSEA AND VOMITING OR POSTOPERATIVE NAUSEA AND VOMITING
#1 OR #2 OR #3
MeSH‐POSTOPERATIVE OR POST‐OPERATIVE
MeSH‐ANESTHESIA OR ANAESTHESIA OR ANESTHET* OR ANAESTHET*
#5 OR #6
MeSH‐ANTIEMETICS OR ANTIEMESIS OR ANTIEMETIC* OR ANTIEMETOGENIC
ALIZAPRIDE OR ALPRAZOLAM OR ATROPINE OR BETAMETHASONE OR BETHAMETHAZONE OR BROMAZEPAM OR CHLORAL HYDRATE OR CHLORPROMAZINE OR CIMETIDINE OR CLEBOPRIDE OR CLONIDINE OR CYCLIZINE OR DEXAMETHASONE OR DEXMEDETOMIDINE OR DIAZEPAM OR DIFENIDOL OR DIMENHYDRINATE OR DIXYRAZINE OR DOLASETRON OR DOMPERIDONE OR DROPERIDOL OR EPHEDRINE OR ERYTHROMYCIN OR FAMOTIDINE OR FLUNITRAZEPAM OR FLURBIPROFEN OR GINGER OR GLYCOPYRROLATE OR GRANISETRON OR HYOSCINE OR INTRALIPID OR ITASETRON OR LIDOCAINE OR LORAZEPAM OR LORMETAZEPAM OR MAGNESIUM OR MEDAZEPAM OR METHYLNALTREXONE OR METHYLPREDNISOLONE OR METOCLOPRAMIDE OR MIDAZOLAM OR NALOXONE OR NEOSTIGMINE OR ONDANSETRON OR OXYGEN OR PALONOSETRON OR PENTOBARBITONE OR PERPHENAZINE OR PREDNISOLONE OR PROCHORPERAZINE OR PENTOBARBITONE OR PROMETHAZINE OR PROPOFOL OR RAMOSETRON OR RANITIDINE OR SULPIRIDE OR TIAPRIDE OR TRIMETHOBENZAMINE OR TROPISETRON
#8 OR #9
#4 AND #7 AND #10
Data and analyses
Comparison 1.
PRIMARY ANALYSIS: Placebo versus Drug
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Nausea | 337 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 1.1 Alizapride | 3 | 185 | Risk Ratio (M‐H, Random, 95% CI) | 0.65 [0.46, 0.92] |
| 1.2 Atropine | 1 | 21 | Risk Ratio (M‐H, Random, 95% CI) | 0.91 [0.07, 12.69] |
| 1.3 Betamethasone | 1 | 78 | Risk Ratio (M‐H, Random, 95% CI) | 0.28 [0.10, 0.77] |
| 1.4 Bromopride | 1 | 200 | Risk Ratio (M‐H, Random, 95% CI) | 0.36 [0.18, 0.70] |
| 1.5 Chlorpromazine | 1 | 26 | Risk Ratio (M‐H, Random, 95% CI) | 1.36 [0.10, 19.50] |
| 1.6 Cimetidine | 2 | 146 | Risk Ratio (M‐H, Random, 95% CI) | 0.66 [0.16, 2.68] |
| 1.7 Clebopride | 1 | 80 | Risk Ratio (M‐H, Random, 95% CI) | 0.67 [0.20, 2.18] |
| 1.8 Clonidine | 10 | 576 | Risk Ratio (M‐H, Random, 95% CI) | 0.69 [0.46, 1.05] |
| 1.9 CP‐122,721 | 1 | 53 | Risk Ratio (M‐H, Random, 95% CI) | 1.00 [0.90, 1.11] |
| 1.10 Cyclizine | 5 | 638 | Risk Ratio (M‐H, Random, 95% CI) | 0.65 [0.47, 0.90] |
| 1.11 Dexamethasone | 51 | 4163 | Risk Ratio (M‐H, Random, 95% CI) | 0.59 [0.50, 0.69] |
| 1.12 Diazepam | 7 | 609 | Risk Ratio (M‐H, Random, 95% CI) | 0.50 [0.25, 0.99] |
| 1.13 Dimenhydrinate | 6 | 278 | Risk Ratio (M‐H, Random, 95% CI) | 0.72 [0.47, 1.13] |
| 1.14 Dixyrazine | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 1.23 [0.73, 2.09] |
| 1.15 Dolasetron | 13 | 2812 | Risk Ratio (M‐H, Random, 95% CI) | 0.82 [0.76, 0.90] |
| 1.16 Domperidone | 3 | 129 | Risk Ratio (M‐H, Random, 95% CI) | 0.62 [0.20, 1.94] |
| 1.17 Droperidol | 80 | 7174 | Risk Ratio (M‐H, Random, 95% CI) | 0.65 [0.60, 0.71] |
| 1.18 Ephedrine | 2 | 46 | Risk Ratio (M‐H, Random, 95% CI) | 0.5 [0.20, 1.23] |
| 1.19 Ginger | 6 | 460 | Risk Ratio (M‐H, Random, 95% CI) | 0.87 [0.62, 1.23] |
| 1.20 Granisetron | 37 | 2950 | Risk Ratio (M‐H, Random, 95% CI) | 0.53 [0.45, 0.63] |
| 1.21 Hydroxyzine | 1 | 50 | Risk Ratio (M‐H, Random, 95% CI) | 0.18 [0.04, 0.74] |
| 1.22 Hyoscine | 14 | 1008 | Risk Ratio (M‐H, Random, 95% CI) | 0.63 [0.47, 0.83] |
| 1.23 Intralipid | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 1.2 [0.41, 3.51] |
| 1.24 Lorazepam | 2 | 50 | Risk Ratio (M‐H, Random, 95% CI) | 0.55 [0.33, 0.93] |
| 1.25 Lormetazepam | 1 | 39 | Risk Ratio (M‐H, Random, 95% CI) | 0.79 [0.20, 3.07] |
| 1.26 Magnesium | 1 | 44 | Risk Ratio (M‐H, Random, 95% CI) | 0.91 [0.35, 2.40] |
| 1.27 Methylnaltrexone | 1 | 120 | Risk Ratio (M‐H, Random, 95% CI) | 0.75 [0.39, 1.45] |
| 1.28 Methylprednisolone | 1 | 72 | Risk Ratio (M‐H, Random, 95% CI) | 0.33 [0.01, 7.92] |
| 1.29 Metoclopramide | 58 | 3001 | Risk Ratio (M‐H, Random, 95% CI) | 0.82 [0.76, 0.88] |
| 1.30 Midazolam | 3 | 145 | Risk Ratio (M‐H, Random, 95% CI) | 0.90 [0.64, 1.28] |
| 1.31 Naloxone | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.47 [0.30, 0.74] |
| 1.32 Neostigmine | 5 | 198 | Risk Ratio (M‐H, Random, 95% CI) | 2.73 [1.15, 6.48] |
| 1.33 Ondansetron | 114 | 15861 | Risk Ratio (M‐H, Random, 95% CI) | 0.68 [0.63, 0.74] |
| 1.34 Perphenazine | 3 | 595 | Risk Ratio (M‐H, Random, 95% CI) | 1.15 [0.42, 3.12] |
| 1.35 Physostigmine | 1 | 100 | Risk Ratio (M‐H, Random, 95% CI) | 4.80 [1.11, 20.82] |
| 1.36 Prochlorperazine | 7 | 453 | Risk Ratio (M‐H, Random, 95% CI) | 0.73 [0.56, 0.96] |
| 1.37 Promethazine | 1 | 29 | Risk Ratio (M‐H, Random, 95% CI) | 0.41 [0.09, 1.88] |
| 1.38 Propofol | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 1.5 [0.29, 7.73] |
| 1.39 Ramosetron | 4 | 339 | Risk Ratio (M‐H, Random, 95% CI) | 0.62 [0.40, 0.96] |
| 1.40 Ranitidine | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.54 [0.39, 0.75] |
| 1.41 Tandospirone | 1 | 90 | Risk Ratio (M‐H, Random, 95% CI) | 0.66 [0.41, 1.06] |
| 1.42 Tiapride | 1 | 26 | Risk Ratio (M‐H, Random, 95% CI) | 0.17 [0.02, 1.20] |
| 1.43 Trimethobenzamide | 1 | 94 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.44, 2.30] |
| 1.44 Tropisetron | 19 | 1989 | Risk Ratio (M‐H, Random, 95% CI) | 0.77 [0.71, 0.84] |
| 2 Vomiting | 445 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 2.1 Alizapride | 3 | 185 | Risk Ratio (M‐H, Random, 95% CI) | 0.49 [0.29, 0.84] |
| 2.2 Alprazolam | 1 | 43 | Risk Ratio (M‐H, Random, 95% CI) | 2.10 [0.20, 21.42] |
| 2.3 Atropine | 2 | 121 | Risk Ratio (M‐H, Random, 95% CI) | 1.11 [0.78, 1.58] |
| 2.4 Betamethasone | 1 | 78 | Risk Ratio (M‐H, Random, 95% CI) | 0.18 [0.02, 1.39] |
| 2.5 Bromopride | 1 | 200 | Risk Ratio (M‐H, Random, 95% CI) | 0.22 [0.08, 0.63] |
| 2.6 Chloral Hydrate | 1 | 65 | Risk Ratio (M‐H, Random, 95% CI) | 0.51 [0.03, 7.80] |
| 2.7 Chlorpromazine | 1 | 26 | Risk Ratio (M‐H, Random, 95% CI) | 0.34 [0.04, 2.64] |
| 2.8 Cimetidine | 2 | 146 | Risk Ratio (M‐H, Random, 95% CI) | 0.47 [0.17, 1.32] |
| 2.9 Cisapride | 1 | 96 | Risk Ratio (M‐H, Random, 95% CI) | 1.06 [0.69, 1.62] |
| 2.10 Clebopride | 1 | 80 | Risk Ratio (M‐H, Random, 95% CI) | 0.05 [0.00, 0.87] |
| 2.11 Clonidine | 12 | 739 | Risk Ratio (M‐H, Random, 95% CI) | 0.75 [0.53, 1.06] |
| 2.12 CP‐122,721 | 1 | 53 | Risk Ratio (M‐H, Random, 95% CI) | 0.16 [0.02, 1.24] |
| 2.13 Cyclizine | 6 | 688 | Risk Ratio (M‐H, Random, 95% CI) | 0.57 [0.43, 0.75] |
| 2.14 Dexamethasone | 66 | 5594 | Risk Ratio (M‐H, Random, 95% CI) | 0.51 [0.46, 0.57] |
| 2.15 Diazepam | 12 | 854 | Risk Ratio (M‐H, Random, 95% CI) | 0.85 [0.58, 1.24] |
| 2.16 Dimenhydrinate | 12 | 940 | Risk Ratio (M‐H, Random, 95% CI) | 0.61 [0.46, 0.81] |
| 2.17 Dixyrazine | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.68 [0.48, 0.97] |
| 2.18 Dolasetron | 15 | 2458 | Risk Ratio (M‐H, Random, 95% CI) | 0.62 [0.51, 0.76] |
| 2.19 Domperidone | 4 | 159 | Risk Ratio (M‐H, Random, 95% CI) | 0.80 [0.52, 1.23] |
| 2.20 Droperidol | 110 | 8084 | Risk Ratio (M‐H, Random, 95% CI) | 0.65 [0.61, 0.70] |
| 2.21 Ephedrine | 4 | 179 | Risk Ratio (M‐H, Random, 95% CI) | 0.91 [0.64, 1.27] |
| 2.22 Flurbiprofen | 1 | 90 | Risk Ratio (M‐H, Random, 95% CI) | 0.89 [0.45, 1.77] |
| 2.23 Ginger | 5 | 440 | Risk Ratio (M‐H, Random, 95% CI) | 1.04 [0.66, 1.64] |
| 2.24 Granisetron | 52 | 4088 | Risk Ratio (M‐H, Random, 95% CI) | 0.40 [0.35, 0.46] |
| 2.25 Haloperidol | 1 | 70 | Risk Ratio (M‐H, Random, 95% CI) | 0.38 [0.19, 0.79] |
| 2.26 Hydroxyzine | 1 | 50 | Risk Ratio (M‐H, Random, 95% CI) | 0.25 [0.06, 1.06] |
| 2.27 Hyoscine | 16 | 1385 | Risk Ratio (M‐H, Random, 95% CI) | 0.66 [0.56, 0.77] |
| 2.28 Intralipid | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.62, 1.61] |
| 2.29 Lidocaine | 1 | 50 | Risk Ratio (M‐H, Random, 95% CI) | 1.33 [0.54, 3.29] |
| 2.30 Lorazepam | 3 | 91 | Risk Ratio (M‐H, Random, 95% CI) | 0.61 [0.33, 1.13] |
| 2.31 Lormetazepam | 1 | 39 | Risk Ratio (M‐H, Random, 95% CI) | 3.15 [0.14, 72.88] |
| 2.32 Magnesium | 1 | 44 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 2.33 Methylnaltrexone | 2 | 233 | Risk Ratio (M‐H, Random, 95% CI) | 0.64 [0.30, 1.33] |
| 2.34 Metoclopramide | 80 | 4050 | Risk Ratio (M‐H, Random, 95% CI) | 0.75 [0.70, 0.81] |
| 2.35 Midazolam | 5 | 458 | Risk Ratio (M‐H, Random, 95% CI) | 0.73 [0.56, 0.95] |
| 2.36 Naloxone | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.36 [0.17, 0.76] |
| 2.37 Neostigmine | 7 | 307 | Risk Ratio (M‐H, Random, 95% CI) | 3.87 [0.79, 18.99] |
| 2.38 Ondansetron | 142 | 17958 | Risk Ratio (M‐H, Random, 95% CI) | 0.55 [0.50, 0.59] |
| 2.39 Palonosetron | 1 | 205 | Risk Ratio (M‐H, Random, 95% CI) | 0.74 [0.49, 1.11] |
| 2.40 Pentobarbitone | 1 | 24 | Risk Ratio (M‐H, Random, 95% CI) | 1.11 [0.74, 1.68] |
| 2.41 Perphenazine | 4 | 864 | Risk Ratio (M‐H, Random, 95% CI) | 0.70 [0.51, 0.96] |
| 2.42 Physostigmine | 1 | 100 | Risk Ratio (M‐H, Random, 95% CI) | 1.44 [0.25, 8.26] |
| 2.43 Prochlorperazine | 8 | 502 | Risk Ratio (M‐H, Random, 95% CI) | 0.68 [0.52, 0.89] |
| 2.44 Promethazine | 3 | 56 | Risk Ratio (M‐H, Random, 95% CI) | 0.76 [0.40, 1.45] |
| 2.45 Propofol | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 0.56 [0.24, 1.27] |
| 2.46 Ramosetron | 4 | 339 | Risk Ratio (M‐H, Random, 95% CI) | 0.42 [0.28, 0.63] |
| 2.47 Ranitidine | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.40 [0.24, 0.68] |
| 2.48 Sulpiride | 1 | 50 | Risk Ratio (M‐H, Random, 95% CI) | 0.25 [0.06, 1.06] |
| 2.49 Tandospirone | 1 | 90 | Risk Ratio (M‐H, Random, 95% CI) | 0.56 [0.34, 0.94] |
| 2.50 Tiapride | 1 | 26 | Risk Ratio (M‐H, Random, 95% CI) | 0.20 [0.03, 1.48] |
| 2.51 Trimeprazine | 1 | 24 | Risk Ratio (M‐H, Random, 95% CI) | 0.56 [0.26, 1.17] |
| 2.52 Trimethobenzamide | 1 | 94 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.41, 2.44] |
| 2.53 Tropisetron | 25 | 2428 | Risk Ratio (M‐H, Random, 95% CI) | 0.59 [0.50, 0.69] |
| 3 Nausea or Vomiting | 271 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 3.1 Alizapride | 2 | 95 | Risk Ratio (M‐H, Random, 95% CI) | 0.68 [0.39, 1.19] |
| 3.2 Atropine | 2 | 62 | Risk Ratio (M‐H, Random, 95% CI) | 0.91 [0.36, 2.31] |
| 3.3 Chlorpromazine | 1 | 26 | Risk Ratio (M‐H, Random, 95% CI) | 0.91 [0.18, 4.55] |
| 3.4 Cimetidine | 1 | 26 | Risk Ratio (M‐H, Random, 95% CI) | 1.33 [0.37, 4.82] |
| 3.5 Clebopride | 1 | 80 | Risk Ratio (M‐H, Random, 95% CI) | 0.27 [0.10, 0.73] |
| 3.6 Clonidine | 8 | 457 | Risk Ratio (M‐H, Random, 95% CI) | 0.73 [0.52, 1.02] |
| 3.7 Cyclizine | 4 | 608 | Risk Ratio (M‐H, Random, 95% CI) | 0.68 [0.58, 0.80] |
| 3.8 Dexamethasone | 46 | 3530 | Risk Ratio (M‐H, Random, 95% CI) | 0.49 [0.44, 0.54] |
| 3.9 Diazepam | 2 | 195 | Risk Ratio (M‐H, Random, 95% CI) | 1.04 [0.51, 2.10] |
| 3.10 Dimenhydrinate | 6 | 377 | Risk Ratio (M‐H, Random, 95% CI) | 0.71 [0.59, 0.86] |
| 3.11 Dixyrazine | 2 | 111 | Risk Ratio (M‐H, Random, 95% CI) | 0.82 [0.67, 1.02] |
| 3.12 Dolasetron | 12 | 1794 | Risk Ratio (M‐H, Random, 95% CI) | 0.72 [0.62, 0.83] |
| 3.13 Domperidone | 6 | 289 | Risk Ratio (M‐H, Random, 95% CI) | 0.71 [0.44, 1.13] |
| 3.14 Droperidol | 69 | 6101 | Risk Ratio (M‐H, Random, 95% CI) | 0.62 [0.58, 0.67] |
| 3.15 Ephedrine | 3 | 163 | Risk Ratio (M‐H, Random, 95% CI) | 0.79 [0.55, 1.15] |
| 3.16 Ginger | 3 | 235 | Risk Ratio (M‐H, Random, 95% CI) | 0.97 [0.75, 1.25] |
| 3.17 Glycopyrrolate | 2 | 93 | Risk Ratio (M‐H, Random, 95% CI) | 0.67 [0.35, 1.29] |
| 3.18 Granisetron | 34 | 2652 | Risk Ratio (M‐H, Random, 95% CI) | 0.39 [0.31, 0.48] |
| 3.19 Hydroxyzine | 1 | 50 | Risk Ratio (M‐H, Random, 95% CI) | 0.21 [0.07, 0.65] |
| 3.20 Hyoscine | 8 | 629 | Risk Ratio (M‐H, Random, 95% CI) | 0.71 [0.56, 0.90] |
| 3.21 Intralipid | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 1.05 [0.76, 1.44] |
| 3.22 Lidocaine | 1 | 16 | Risk Ratio (M‐H, Random, 95% CI) | 0.50 [0.06, 4.47] |
| 3.23 Magnesium | 2 | 94 | Risk Ratio (M‐H, Random, 95% CI) | 0.79 [0.36, 1.72] |
| 3.24 Methylprednisolone | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.36, 2.75] |
| 3.25 Metoclopramide | 54 | 2707 | Risk Ratio (M‐H, Random, 95% CI) | 0.76 [0.70, 0.82] |
| 3.26 Midazolam | 2 | 91 | Risk Ratio (M‐H, Random, 95% CI) | 1.44 [0.52, 3.94] |
| 3.27 Neostigmine | 9 | 488 | Risk Ratio (M‐H, Random, 95% CI) | 3.19 [1.71, 5.93] |
| 3.28 Ondansetron | 79 | 6721 | Risk Ratio (M‐H, Random, 95% CI) | 0.56 [0.50, 0.63] |
| 3.29 Palonosetron | 1 | 348 | Risk Ratio (M‐H, Random, 95% CI) | 0.72 [0.58, 0.89] |
| 3.30 Perphenazine | 3 | 610 | Risk Ratio (M‐H, Random, 95% CI) | 0.71 [0.43, 1.15] |
| 3.31 Prochlorperazine | 5 | 438 | Risk Ratio (M‐H, Random, 95% CI) | 0.68 [0.55, 0.86] |
| 3.32 Promethazine | 2 | 41 | Risk Ratio (M‐H, Random, 95% CI) | 0.46 [0.25, 0.82] |
| 3.33 Propofol | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 0.8 [0.44, 1.45] |
| 3.34 Ramosetron | 5 | 341 | Risk Ratio (M‐H, Random, 95% CI) | 0.51 [0.39, 0.68] |
| 3.35 Ranitidine | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.6 [0.36, 1.00] |
| 3.36 Sulpiride | 1 | 50 | Risk Ratio (M‐H, Random, 95% CI) | 0.27 [0.10, 0.69] |
| 3.37 Tandospirone | 1 | 90 | Risk Ratio (M‐H, Random, 95% CI) | 0.68 [0.46, 0.98] |
| 3.38 Tiapride | 1 | 26 | Risk Ratio (M‐H, Random, 95% CI) | 0.17 [0.02, 1.20] |
| 3.39 Trimethobenzamide | 1 | 94 | Risk Ratio (M‐H, Random, 95% CI) | 0.92 [0.47, 1.81] |
| 3.40 Tropisetron | 16 | 1184 | Risk Ratio (M‐H, Random, 95% CI) | 0.70 [0.61, 0.81] |
| 4 Rescue antiemetic | 294 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 4.1 Alizapride | 1 | 35 | Risk Ratio (M‐H, Random, 95% CI) | 0.59 [0.31, 1.12] |
| 4.2 Atropine | 1 | 41 | Risk Ratio (M‐H, Random, 95% CI) | 0.95 [0.06, 14.22] |
| 4.3 Betamethasone | 1 | 78 | Risk Ratio (M‐H, Random, 95% CI) | 0.67 [0.29, 1.55] |
| 4.4 Cimetidine | 1 | 26 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.07, 14.34] |
| 4.5 Clonidine | 7 | 382 | Risk Ratio (M‐H, Random, 95% CI) | 1.09 [0.94, 1.27] |
| 4.6 CP‐122,721 | 1 | 53 | Risk Ratio (M‐H, Random, 95% CI) | 0.66 [0.38, 1.14] |
| 4.7 Cyclizine | 4 | 198 | Risk Ratio (M‐H, Random, 95% CI) | 0.27 [0.14, 0.52] |
| 4.8 Dexamethasone | 47 | 4111 | Risk Ratio (M‐H, Random, 95% CI) | 0.50 [0.42, 0.59] |
| 4.9 Diazepam | 1 | 25 | Risk Ratio (M‐H, Random, 95% CI) | 1.85 [0.41, 8.32] |
| 4.10 Dimenhydrinate | 8 | 721 | Risk Ratio (M‐H, Random, 95% CI) | 0.62 [0.33, 1.15] |
| 4.11 Dixyrazine | 2 | 111 | Risk Ratio (M‐H, Random, 95% CI) | 0.49 [0.30, 0.80] |
| 4.12 Dolasetron | 10 | 2805 | Risk Ratio (M‐H, Random, 95% CI) | 0.67 [0.57, 0.79] |
| 4.13 Domperidone | 1 | 37 | Risk Ratio (M‐H, Random, 95% CI) | 0.90 [0.42, 1.92] |
| 4.14 Droperidol | 74 | 6241 | Risk Ratio (M‐H, Random, 95% CI) | 0.53 [0.47, 0.60] |
| 4.15 Ephedrine | 3 | 149 | Risk Ratio (M‐H, Random, 95% CI) | 0.82 [0.41, 1.66] |
| 4.16 Ginger | 4 | 177 | Risk Ratio (M‐H, Random, 95% CI) | 0.40 [0.18, 0.88] |
| 4.17 Glycopyrrolate | 2 | 93 | Risk Ratio (M‐H, Random, 95% CI) | 0.52 [0.18, 1.48] |
| 4.18 Granisetron | 42 | 3410 | Risk Ratio (M‐H, Random, 95% CI) | 0.29 [0.22, 0.39] |
| 4.19 Hyoscine | 10 | 949 | Risk Ratio (M‐H, Random, 95% CI) | 0.92 [0.69, 1.21] |
| 4.20 Intralipid | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 1.25 [0.37, 4.21] |
| 4.21 Lidocaine | 1 | 50 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 4.22 Lorazepam | 1 | 26 | Risk Ratio (M‐H, Random, 95% CI) | 0.5 [0.05, 4.86] |
| 4.23 Methylnaltrexone | 2 | 233 | Risk Ratio (M‐H, Random, 95% CI) | 0.63 [0.33, 1.21] |
| 4.24 Metoclopramide | 44 | 2075 | Risk Ratio (M‐H, Random, 95% CI) | 0.78 [0.69, 0.88] |
| 4.25 Midazolam | 3 | 115 | Risk Ratio (M‐H, Random, 95% CI) | 0.61 [0.38, 0.98] |
| 4.26 Naloxone | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.54 [0.32, 0.92] |
| 4.27 Neostigmine | 2 | 150 | Risk Ratio (M‐H, Random, 95% CI) | 1.39 [0.55, 3.50] |
| 4.28 Ondansetron | 106 | 11383 | Risk Ratio (M‐H, Random, 95% CI) | 0.55 [0.49, 0.61] |
| 4.29 Palonosetron | 1 | 218 | Risk Ratio (M‐H, Random, 95% CI) | 0.78 [0.61, 0.99] |
| 4.30 Pentobarbitone | 1 | 24 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.17, 5.98] |
| 4.31 Perphenazine | 1 | 258 | Risk Ratio (M‐H, Random, 95% CI) | 0.52 [0.25, 1.07] |
| 4.32 Prochlorperazine | 5 | 281 | Risk Ratio (M‐H, Random, 95% CI) | 0.49 [0.22, 1.08] |
| 4.33 Promethazine | 1 | 29 | Risk Ratio (M‐H, Random, 95% CI) | 0.27 [0.03, 2.31] |
| 4.34 Ramosetron | 3 | 259 | Risk Ratio (M‐H, Random, 95% CI) | 0.38 [0.15, 0.99] |
| 4.35 Ranitidine | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.86 [0.33, 2.25] |
| 4.36 Tandospirone | 1 | 90 | Risk Ratio (M‐H, Random, 95% CI) | 0.5 [0.25, 1.02] |
| 4.37 Tiapride | 1 | 26 | Risk Ratio (M‐H, Random, 95% CI) | 0.2 [0.01, 3.80] |
| 4.38 Trimeprazine | 1 | 24 | Risk Ratio (M‐H, Random, 95% CI) | 0.50 [0.05, 4.81] |
| 4.39 Tropisetron | 24 | 1997 | Risk Ratio (M‐H, Random, 95% CI) | 0.62 [0.53, 0.72] |
Comparison 2.
PRIMARY ANALYSIS: No Treatment versus Drug
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Nausea | 27 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 1.1 Atropine | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.07, 14.55] |
| 1.2 Butorphanol | 1 | 20 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.17, 5.77] |
| 1.3 Chlorpromazine | 1 | 25 | Risk Ratio (M‐H, Random, 95% CI) | 1.5 [0.11, 21.31] |
| 1.4 Cimetidine | 1 | 231 | Risk Ratio (M‐H, Random, 95% CI) | 0.39 [0.20, 0.76] |
| 1.5 Dexamethasone | 1 | 16 | Risk Ratio (M‐H, Random, 95% CI) | 0.57 [0.27, 1.20] |
| 1.6 Diazepam | 1 | 40 | Risk Ratio (M‐H, Random, 95% CI) | 0.67 [0.22, 2.01] |
| 1.7 Dolasetron | 1 | 92 | Risk Ratio (M‐H, Random, 95% CI) | 1.08 [0.81, 1.44] |
| 1.8 Droperidol | 11 | 1672 | Risk Ratio (M‐H, Random, 95% CI) | 0.62 [0.44, 0.86] |
| 1.9 Ephedrine | 1 | 45 | Risk Ratio (M‐H, Random, 95% CI) | 0.25 [0.02, 2.54] |
| 1.10 Hyoscine | 1 | 34 | Risk Ratio (M‐H, Random, 95% CI) | 1.27 [0.73, 2.21] |
| 1.11 Metoclopramide | 3 | 199 | Risk Ratio (M‐H, Random, 95% CI) | 0.34 [0.17, 0.66] |
| 1.12 Ondansetron | 7 | 439 | Risk Ratio (M‐H, Random, 95% CI) | 0.65 [0.50, 0.86] |
| 1.13 Promethazine | 2 | 76 | Risk Ratio (M‐H, Random, 95% CI) | 0.81 [0.55, 1.20] |
| 1.14 Ranitidine | 1 | 420 | Risk Ratio (M‐H, Random, 95% CI) | 0.28 [0.15, 0.52] |
| 1.15 Tropisetron | 1 | 68 | Risk Ratio (M‐H, Random, 95% CI) | 0.92 [0.49, 1.72] |
| 2 Vomiting | 42 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 2.1 Atropine | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 0.67 [0.23, 1.89] |
| 2.2 Butorphanol | 1 | 20 | Risk Ratio (M‐H, Random, 95% CI) | 0.6 [0.19, 1.86] |
| 2.3 Chlorpromazine | 1 | 25 | Risk Ratio (M‐H, Random, 95% CI) | 0.25 [0.04, 1.77] |
| 2.4 Cimetidine | 1 | 231 | Risk Ratio (M‐H, Random, 95% CI) | 0.30 [0.15, 0.59] |
| 2.5 Dexamethasone | 2 | 384 | Risk Ratio (M‐H, Random, 95% CI) | 0.40 [0.24, 0.65] |
| 2.6 Diazepam | 1 | 40 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.34, 2.93] |
| 2.7 Dixyrazine | 2 | 136 | Risk Ratio (M‐H, Random, 95% CI) | 0.31 [0.18, 0.53] |
| 2.8 Dolasetron | 1 | 92 | Risk Ratio (M‐H, Random, 95% CI) | 0.52 [0.30, 0.91] |
| 2.9 Domperidone | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 0.29 [0.07, 1.16] |
| 2.10 Droperidol | 15 | 2060 | Risk Ratio (M‐H, Random, 95% CI) | 0.63 [0.52, 0.76] |
| 2.11 Ephedrine | 1 | 45 | Risk Ratio (M‐H, Random, 95% CI) | 0.56 [0.29, 1.07] |
| 2.12 Hyoscine | 2 | 84 | Risk Ratio (M‐H, Random, 95% CI) | 0.30 [0.13, 0.74] |
| 2.13 Lidocaine | 1 | 79 | Risk Ratio (M‐H, Random, 95% CI) | 0.92 [0.58, 1.48] |
| 2.14 Metoclopramide | 5 | 470 | Risk Ratio (M‐H, Random, 95% CI) | 0.49 [0.30, 0.79] |
| 2.15 Ondansetron | 12 | 868 | Risk Ratio (M‐H, Random, 95% CI) | 0.42 [0.34, 0.52] |
| 2.16 Prochlorperazine | 1 | 158 | Risk Ratio (M‐H, Random, 95% CI) | 0.78 [0.49, 1.24] |
| 2.17 Promethazine | 2 | 76 | Risk Ratio (M‐H, Random, 95% CI) | 0.53 [0.15, 1.84] |
| 2.18 Ranitidine | 1 | 420 | Risk Ratio (M‐H, Random, 95% CI) | 0.17 [0.09, 0.32] |
| 2.19 Tropisetron | 1 | 68 | Risk Ratio (M‐H, Random, 95% CI) | 1.33 [0.32, 5.51] |
| 3 Nausea or Vomiting | 20 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 3.1 Atropine | 1 | 68 | Risk Ratio (M‐H, Random, 95% CI) | 0.70 [0.38, 1.26] |
| 3.2 Butorphanol | 1 | 20 | Risk Ratio (M‐H, Random, 95% CI) | 0.83 [0.37, 1.85] |
| 3.3 Chlorpromazine | 1 | 53 | Risk Ratio (M‐H, Random, 95% CI) | 0.28 [0.10, 0.76] |
| 3.4 Droperidol | 9 | 1619 | Risk Ratio (M‐H, Random, 95% CI) | 0.56 [0.41, 0.78] |
| 3.5 Ephedrine | 1 | 45 | Risk Ratio (M‐H, Random, 95% CI) | 0.55 [0.30, 0.99] |
| 3.6 Metoclopramide | 2 | 181 | Risk Ratio (M‐H, Random, 95% CI) | 0.35 [0.17, 0.74] |
| 3.7 Neostigmine | 1 | 120 | Risk Ratio (M‐H, Random, 95% CI) | 2.53 [0.87, 7.41] |
| 3.8 Ondansetron | 5 | 388 | Risk Ratio (M‐H, Random, 95% CI) | 0.56 [0.44, 0.72] |
| 3.9 Promethazine | 1 | 36 | Risk Ratio (M‐H, Random, 95% CI) | 0.86 [0.57, 1.29] |
| 3.10 Tropisetron | 1 | 68 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.60, 1.66] |
| 4 Rescue antiemetic | 18 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 4.1 Butorphanol | 1 | 20 | Risk Ratio (M‐H, Random, 95% CI) | 1.5 [0.32, 7.14] |
| 4.2 Dexamethasone | 1 | 16 | Risk Ratio (M‐H, Random, 95% CI) | 0.5 [0.13, 2.00] |
| 4.3 Dixyrazine | 2 | 136 | Risk Ratio (M‐H, Random, 95% CI) | 0.08 [0.01, 0.61] |
| 4.4 Dolasetron | 1 | 92 | Risk Ratio (M‐H, Random, 95% CI) | 0.97 [0.71, 1.33] |
| 4.5 Droperidol | 6 | 291 | Risk Ratio (M‐H, Random, 95% CI) | 0.57 [0.40, 0.82] |
| 4.6 Hyoscine | 1 | 34 | Risk Ratio (M‐H, Random, 95% CI) | 1.01 [0.33, 3.13] |
| 4.7 Lidocaine | 1 | 78 | Risk Ratio (M‐H, Random, 95% CI) | 0.21 [0.05, 0.91] |
| 4.8 Metoclopramide | 1 | 80 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 4.9 Neostigmine | 1 | 120 | Risk Ratio (M‐H, Random, 95% CI) | 3.95 [0.24, 65.27] |
| 4.10 Ondansetron | 6 | 544 | Risk Ratio (M‐H, Random, 95% CI) | 0.50 [0.32, 0.79] |
| 4.11 Tropisetron | 1 | 68 | Risk Ratio (M‐H, Random, 95% CI) | 0.91 [0.45, 1.85] |
Comparison 3.
PRIMARY ANALYSIS: Drug versus Drug
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Nausea | 188 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 1.1 Alizapride versus Droperidol | 1 | 35 | Risk Ratio (M‐H, Random, 95% CI) | 1.43 [0.58, 3.55] |
| 1.2 Alizapride versus Metoclopramide | 1 | 35 | Risk Ratio (M‐H, Random, 95% CI) | 0.82 [0.43, 1.56] |
| 1.3 Alizapride versus Ondansetron | 1 | 52 | Risk Ratio (M‐H, Random, 95% CI) | 1.08 [0.07, 16.36] |
| 1.4 Atropine versus Chlorpromazine | 1 | 25 | Risk Ratio (M‐H, Random, 95% CI) | 0.67 [0.05, 9.47] |
| 1.5 Atropine versus Glycopyrrolate | 1 | 100 | Risk Ratio (M‐H, Random, 95% CI) | 0.64 [0.31, 1.35] |
| 1.6 Atropine versus Hyoscine | 2 | 100 | Risk Ratio (M‐H, Random, 95% CI) | 2.33 [0.98, 5.58] |
| 1.7 Bromazepam versus Lorazepam | 1 | 153 | Risk Ratio (M‐H, Random, 95% CI) | 0.74 [0.38, 1.41] |
| 1.8 Butorphanol versus Droperidol | 1 | 20 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.17, 5.77] |
| 1.9 Cimetidine versus Metoclopramide | 1 | 14 | Risk Ratio (M‐H, Random, 95% CI) | 5.0 [0.28, 88.53] |
| 1.10 Clonidine versus Diazepam | 1 | 100 | Risk Ratio (M‐H, Random, 95% CI) | 1.38 [0.76, 2.51] |
| 1.11 Clonidine versus Droperidol | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 0.11 [0.01, 1.90] |
| 1.12 Clonidine versus Midazolam | 1 | 100 | Risk Ratio (M‐H, Random, 95% CI) | 0.57 [0.18, 1.83] |
| 1.13 Clonidine versus Neostigmine | 1 | 10 | Risk Ratio (M‐H, Random, 95% CI) | 0.25 [0.04, 1.52] |
| 1.14 CP‐122,721 versus Ondansetron | 1 | 53 | Risk Ratio (M‐H, Random, 95% CI) | 1.00 [0.90, 1.11] |
| 1.15 Cyclizine versus Dexamethasone | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 0.56 [0.24, 1.27] |
| 1.16 Cyclizine versus Droperidol | 1 | 50 | Risk Ratio (M‐H, Random, 95% CI) | 0.7 [0.32, 1.54] |
| 1.17 Cyclizine versus Ondansetron | 2 | 133 | Risk Ratio (M‐H, Random, 95% CI) | 1.00 [0.69, 1.44] |
| 1.18 Cyclizine versus Perphenazine | 2 | 425 | Risk Ratio (M‐H, Random, 95% CI) | 0.49 [0.08, 2.92] |
| 1.19 Dexamethasone versus Dolasetron | 1 | 14 | Risk Ratio (M‐H, Random, 95% CI) | 0.5 [0.06, 4.33] |
| 1.20 Dexamethasone versus Droperidol | 6 | 313 | Risk Ratio (M‐H, Random, 95% CI) | 1.08 [0.64, 1.84] |
| 1.21 Dexamethasone versus Granisetron | 2 | 54 | Risk Ratio (M‐H, Random, 95% CI) | 1.65 [0.54, 5.04] |
| 1.22 Dexamethasone versus Metoclopramide | 3 | 92 | Risk Ratio (M‐H, Random, 95% CI) | 0.61 [0.28, 1.34] |
| 1.23 Dexamethasone versus Ondansetron | 7 | 318 | Risk Ratio (M‐H, Random, 95% CI) | 1.27 [0.94, 1.71] |
| 1.24 Dexamethasone versus Tropisetron | 2 | 78 | Risk Ratio (M‐H, Random, 95% CI) | 0.41 [0.22, 0.78] |
| 1.25 Diazepam versus Droperidol | 1 | 50 | Risk Ratio (M‐H, Random, 95% CI) | 1.95 [0.76, 5.00] |
| 1.26 Diazepam versus Flunitrazepam | 2 | 130 | Risk Ratio (M‐H, Random, 95% CI) | 0.8 [0.42, 1.51] |
| 1.27 Diazepam versus Midazolam | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.54, 1.86] |
| 1.28 Diazepam versus Pentobarbitone | 1 | 50 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 1.29 Diazepam versus Promethazine | 2 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.83 [0.39, 1.76] |
| 1.30 Difenidol versus Metoclopramide | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.07, 13.64] |
| 1.31 Difenidol versus Ondansetron | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 3.0 [0.14, 65.16] |
| 1.32 Dimenhydrinate versus Droperidol | 2 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 1.70 [0.73, 3.99] |
| 1.33 Dimenhydrinate versus Metoclopramide | 2 | 43 | Risk Ratio (M‐H, Random, 95% CI) | 1.51 [0.43, 5.33] |
| 1.34 Dimenhydrinate versus Ondansetron | 3 | 156 | Risk Ratio (M‐H, Random, 95% CI) | 0.80 [0.51, 1.26] |
| 1.35 Dolasetron versus Droperidol | 3 | 109 | Risk Ratio (M‐H, Random, 95% CI) | 1.06 [0.62, 1.82] |
| 1.36 Dolasetron versus Metoclopramide | 4 | 227 | Risk Ratio (M‐H, Random, 95% CI) | 0.85 [0.57, 1.26] |
| 1.37 Dolasetron versus Ondansetron | 5 | 527 | Risk Ratio (M‐H, Random, 95% CI) | 1.02 [0.81, 1.28] |
| 1.38 Dolasetron versus Tropisetron | 1 | 41 | Risk Ratio (M‐H, Random, 95% CI) | 1.05 [0.72, 1.52] |
| 1.39 Domperidone versus Droperidol | 2 | 69 | Risk Ratio (M‐H, Random, 95% CI) | 0.96 [0.23, 4.05] |
| 1.40 Domperidone versus Granisetron | 1 | 100 | Risk Ratio (M‐H, Random, 95% CI) | 4.0 [1.20, 13.32] |
| 1.41 Domperidone versus Metoclopramide | 4 | 232 | Risk Ratio (M‐H, Random, 95% CI) | 0.94 [0.62, 1.43] |
| 1.42 Droperidol versus Ephedrine | 1 | 16 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.28, 3.54] |
| 1.43 Droperidol versus Ginger | 1 | 19 | Risk Ratio (M‐H, Random, 95% CI) | 0.9 [0.16, 5.13] |
| 1.44 Droperidol versus Granisetron | 19 | 782 | Risk Ratio (M‐H, Random, 95% CI) | 1.36 [1.05, 1.77] |
| 1.45 Droperidol versus Hydroxyzine | 1 | 50 | Risk Ratio (M‐H, Random, 95% CI) | 3.5 [0.80, 15.23] |
| 1.46 Droperidol versus Hyoscine | 1 | 40 | Risk Ratio (M‐H, Random, 95% CI) | 0.65 [0.42, 1.00] |
| 1.47 Droperidol versus Metoclopramide | 27 | 726 | Risk Ratio (M‐H, Random, 95% CI) | 0.90 [0.74, 1.10] |
| 1.48 Droperidol versus Ondansetron | 33 | 2386 | Risk Ratio (M‐H, Random, 95% CI) | 0.95 [0.88, 1.03] |
| 1.49 Droperidol versus Propofol | 2 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 3.48 [0.78, 15.46] |
| 1.50 Droperidol versus Tropisetron | 2 | 169 | Risk Ratio (M‐H, Random, 95% CI) | 1.07 [0.86, 1.33] |
| 1.51 Ephedrine versus Propofol | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 0.33 [0.04, 2.85] |
| 1.52 Erythromycin versus Metoclopramide | 1 | 74 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.32, 3.17] |
| 1.53 Ginger versus Metoclopramide | 2 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.92 [0.54, 1.59] |
| 1.54 Glycopyrrolate versus Hyoscine | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 2.67 [0.78, 9.09] |
| 1.55 Granisetron versus Metoclopramide | 11 | 318 | Risk Ratio (M‐H, Random, 95% CI) | 0.50 [0.31, 0.81] |
| 1.56 Granisetron versus Ramosetron | 5 | 416 | Risk Ratio (M‐H, Random, 95% CI) | 2.34 [1.11, 4.94] |
| 1.57 Lidocaine versus Metoclopramide | 1 | 100 | Risk Ratio (M‐H, Random, 95% CI) | 4.06 [1.45, 11.39] |
| 1.58 Metoclopramide versus Nabilone | 1 | 53 | Risk Ratio (M‐H, Random, 95% CI) | 0.96 [0.69, 1.35] |
| 1.59 Metoclopramide versus Ondansetron | 31 | 1970 | Risk Ratio (M‐H, Random, 95% CI) | 1.22 [1.01, 1.47] |
| 1.60 Metoclopramide versus Perphenazine | 1 | 25 | Risk Ratio (M‐H, Random, 95% CI) | 0.31 [0.01, 6.94] |
| 1.61 Metoclopramide versus Propofol | 2 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 3.48 [0.78, 15.46] |
| 1.62 Metoclopramide versus Tiapride | 1 | 26 | Risk Ratio (M‐H, Random, 95% CI) | 2.0 [0.21, 19.44] |
| 1.63 Metoclopramide versus Trimethobenzamide | 1 | 95 | Risk Ratio (M‐H, Random, 95% CI) | 0.87 [0.37, 2.06] |
| 1.64 Metoclopramide versus Tropisetron | 3 | 120 | Risk Ratio (M‐H, Random, 95% CI) | 0.86 [0.50, 1.48] |
| 1.65 Ondansetron versus Prochlorperazine | 4 | 301 | Risk Ratio (M‐H, Random, 95% CI) | 0.96 [0.49, 1.86] |
| 1.66 Ondansetron versus Prometazine | 3 | 83 | Risk Ratio (M‐H, Random, 95% CI) | 0.81 [0.46, 1.40] |
| 1.67 Ondansetron versus Tropisetron | 8 | 377 | Risk Ratio (M‐H, Random, 95% CI) | 1.15 [0.82, 1.60] |
| 2 Vomiting | 258 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 2.1 Alizapride versus Droperidol | 1 | 35 | Risk Ratio (M‐H, Random, 95% CI) | 1.17 [0.45, 3.03] |
| 2.2 Alizapride versus Metoclopramide | 1 | 35 | Risk Ratio (M‐H, Random, 95% CI) | 0.78 [0.37, 1.68] |
| 2.3 Alizapride versus Ondansetron | 1 | 52 | Risk Ratio (M‐H, Random, 95% CI) | 7.54 [0.41, 139.04] |
| 2.4 Alprazolam versus Chloral hydrate | 1 | 32 | Risk Ratio (M‐H, Random, 95% CI) | 1.05 [0.11, 10.31] |
| 2.5 Alprazolam versus Diazepam | 1 | 32 | Risk Ratio (M‐H, Random, 95% CI) | 1.05 [0.11, 10.31] |
| 2.6 Alprazolam versus Midazolam | 1 | 41 | Risk Ratio (M‐H, Random, 95% CI) | 0.95 [0.15, 6.13] |
| 2.7 Atropine versus Chlorpromazine | 1 | 25 | Risk Ratio (M‐H, Random, 95% CI) | 2.0 [0.24, 16.61] |
| 2.8 Atropine versus Glycopyrrolate | 2 | 190 | Risk Ratio (M‐H, Random, 95% CI) | 0.67 [0.50, 0.90] |
| 2.9 Atropine versus Hyoscine | 2 | 100 | Risk Ratio (M‐H, Random, 95% CI) | 3.12 [1.56, 6.25] |
| 2.10 Bromazepam versus Lorazepam | 1 | 153 | Risk Ratio (M‐H, Random, 95% CI) | 0.48 [0.12, 1.85] |
| 2.11 Butorphanol versus Droperidol | 1 | 20 | Risk Ratio (M‐H, Random, 95% CI) | 3.0 [0.37, 24.17] |
| 2.12 Chloral Hydrate versus Diazepam | 1 | 22 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.07, 14.05] |
| 2.13 Chloral Hydrate versus Midazolam | 1 | 31 | Risk Ratio (M‐H, Random, 95% CI) | 0.91 [0.09, 8.93] |
| 2.14 Cimetidine versus Metoclopramide | 1 | 14 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.08, 13.02] |
| 2.15 Clonidine versus Diazepam | 3 | 213 | Risk Ratio (M‐H, Random, 95% CI) | 0.58 [0.29, 1.15] |
| 2.16 Clonidine versus Midazolam | 3 | 274 | Risk Ratio (M‐H, Random, 95% CI) | 0.81 [0.31, 2.10] |
| 2.17 Clonidine versus Neostigmine | 1 | 10 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 2.18 CP‐122,721 versus Ondansetron | 1 | 53 | Risk Ratio (M‐H, Random, 95% CI) | 0.35 [0.08, 1.56] |
| 2.19 Cyclizine versus Dexamethasone | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 0.56 [0.24, 1.27] |
| 2.20 Cyclizine versus Droperidol | 1 | 50 | Risk Ratio (M‐H, Random, 95% CI) | 1.5 [0.27, 8.22] |
| 2.21 Cyclizine versus Ondansetron | 3 | 183 | Risk Ratio (M‐H, Random, 95% CI) | 1.36 [0.58, 3.18] |
| 2.22 Cyclizine versus Perphenazine | 2 | 440 | Risk Ratio (M‐H, Random, 95% CI) | 0.80 [0.55, 1.16] |
| 2.23 Dexamethasone versus Dolasetron | 1 | 14 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.08, 13.02] |
| 2.24 Dexamethasone versus Droperidol | 6 | 313 | Risk Ratio (M‐H, Random, 95% CI) | 0.96 [0.48, 1.93] |
| 2.25 Dexamethasone versus Granisetron | 3 | 104 | Risk Ratio (M‐H, Random, 95% CI) | 1.75 [0.85, 3.62] |
| 2.26 Dexamethasone versus Metoclopramide | 3 | 92 | Risk Ratio (M‐H, Random, 95% CI) | 0.45 [0.17, 1.20] |
| 2.27 Dexamethasone versus Ondansetron | 7 | 318 | Risk Ratio (M‐H, Random, 95% CI) | 1.38 [0.84, 2.26] |
| 2.28 Dexamethasone versus Perphenazine | 1 | 226 | Risk Ratio (M‐H, Random, 95% CI) | 1.38 [0.99, 1.92] |
| 2.29 Dexamethasone versus Tropisetron | 2 | 78 | Risk Ratio (M‐H, Random, 95% CI) | 0.38 [0.13, 1.11] |
| 2.30 Diazepam versus Droperidol | 2 | 116 | Risk Ratio (M‐H, Random, 95% CI) | 2.16 [1.39, 3.34] |
| 2.31 Diazepam versus Flunitrazepam | 3 | 271 | Risk Ratio (M‐H, Random, 95% CI) | 1.74 [1.04, 2.91] |
| 2.32 Diazepam versus Midazolam | 2 | 71 | Risk Ratio (M‐H, Random, 95% CI) | 2.08 [0.28, 15.60] |
| 2.33 Diazepam versus Phenobarbitone | 3 | 130 | Risk Ratio (M‐H, Random, 95% CI) | 0.95 [0.66, 1.38] |
| 2.34 Diazepam versus Promethazine | 2 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 1.78 [0.32, 10.03] |
| 2.35 Diazepam versus Triclofos | 1 | 200 | Risk Ratio (M‐H, Random, 95% CI) | 1.06 [0.82, 1.37] |
| 2.36 Diazepam versus Trimeprazine | 3 | 126 | Risk Ratio (M‐H, Random, 95% CI) | 1.96 [0.98, 3.90] |
| 2.37 Difenidol versus Metoclopramide | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 0.25 [0.03, 1.82] |
| 2.38 Difenidol versus Ondansetron | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.07, 13.64] |
| 2.39 Dimenhydrinate versus Droperidol | 3 | 179 | Risk Ratio (M‐H, Random, 95% CI) | 0.93 [0.53, 1.64] |
| 2.40 Dimenhydrinate versus Metoclopramide | 3 | 162 | Risk Ratio (M‐H, Random, 95% CI) | 0.79 [0.46, 1.36] |
| 2.41 Dimenhydrinate versus Ondansetron | 4 | 181 | Risk Ratio (M‐H, Random, 95% CI) | 1.76 [1.09, 2.85] |
| 2.42 Dimenhydrinate versus Tropisetron | 1 | 118 | Risk Ratio (M‐H, Random, 95% CI) | 0.81 [0.45, 1.44] |
| 2.43 Dixyrazine versus Droperidol | 1 | 21 | Risk Ratio (M‐H, Random, 95% CI) | 0.66 [0.21, 2.08] |
| 2.44 Dolasetron versus Droperidol | 3 | 109 | Risk Ratio (M‐H, Random, 95% CI) | 0.80 [0.50, 1.30] |
| 2.45 Dolasetron versus Metoclopramide | 4 | 227 | Risk Ratio (M‐H, Random, 95% CI) | 0.36 [0.19, 0.65] |
| 2.46 Dolasetron versus Ondansetron | 7 | 781 | Risk Ratio (M‐H, Random, 95% CI) | 1.17 [0.94, 1.45] |
| 2.47 Dolasetron versus Tropisetron | 1 | 41 | Risk Ratio (M‐H, Random, 95% CI) | 1.31 [0.84, 2.06] |
| 2.48 Domperidone versus Droperidol | 2 | 69 | Risk Ratio (M‐H, Random, 95% CI) | 2.13 [0.82, 5.53] |
| 2.49 Domperidone versus Granisetron | 1 | 100 | Risk Ratio (M‐H, Random, 95% CI) | 3.2 [1.27, 8.07] |
| 2.50 Domperidone versus Metoclopramide | 4 | 231 | Risk Ratio (M‐H, Random, 95% CI) | 1.01 [0.64, 1.59] |
| 2.51 Droperidol versus Ephedrine | 2 | 48 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.15, 6.45] |
| 2.52 Droperidol versus Ginger | 1 | 19 | Risk Ratio (M‐H, Random, 95% CI) | 0.45 [0.05, 4.16] |
| 2.53 Droperidol versus Granisetron | 24 | 1008 | Risk Ratio (M‐H, Random, 95% CI) | 2.16 [1.71, 2.72] |
| 2.54 Droperidol versus Haloperidol | 1 | 69 | Risk Ratio (M‐H, Random, 95% CI) | 0.61 [0.15, 2.52] |
| 2.55 Droperidol versus Hydroxyzine | 1 | 50 | Risk Ratio (M‐H, Random, 95% CI) | 3.0 [0.67, 13.46] |
| 2.56 Droperidol versus Hyoscine | 1 | 40 | Risk Ratio (M‐H, Random, 95% CI) | 0.5 [0.21, 1.20] |
| 2.57 Droperidol versus Lidocaine | 1 | 50 | Risk Ratio (M‐H, Random, 95% CI) | 0.46 [0.21, 1.02] |
| 2.58 Droperidol versus Lorazepam | 1 | 41 | Risk Ratio (M‐H, Random, 95% CI) | 1.90 [0.55, 6.60] |
| 2.59 Droperidol versus Metoclopramide | 39 | 1206 | Risk Ratio (M‐H, Random, 95% CI) | 0.83 [0.71, 0.96] |
| 2.60 Droperidol versus Midazolam | 2 | 543 | Risk Ratio (M‐H, Random, 95% CI) | 0.77 [0.63, 0.94] |
| 2.61 Droperidol versus Ondansetron | 45 | 2529 | Risk Ratio (M‐H, Random, 95% CI) | 1.22 [1.09, 1.37] |
| 2.62 Droperidol versus Prochlorperazine | 1 | 34 | Risk Ratio (M‐H, Random, 95% CI) | 2.0 [0.42, 9.50] |
| 2.63 Droperidol versus Promethazine | 1 | 100 | Risk Ratio (M‐H, Random, 95% CI) | 5.6 [2.35, 13.33] |
| 2.64 Droperidol versus Propofol | 2 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 3.0 [0.66, 13.69] |
| 2.65 Droperidol versus Sulpiride | 1 | 38 | Risk Ratio (M‐H, Random, 95% CI) | 1.92 [0.30, 12.13] |
| 2.66 Droperidol versus Tropisetron | 4 | 326 | Risk Ratio (M‐H, Random, 95% CI) | 1.10 [0.54, 2.22] |
| 2.67 Ephedrine versus Propofol | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.36, 2.75] |
| 2.68 Erythromycin versus Metoclopramide | 1 | 74 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.32, 3.17] |
| 2.69 Ginger versus Metoclopramide | 1 | 40 | Risk Ratio (M‐H, Random, 95% CI) | 0.5 [0.10, 2.43] |
| 2.70 Glycopyrrolate versus Hyoscine | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 2.67 [1.21, 5.88] |
| 2.71 Granisetron versus Metoclopramide | 13 | 376 | Risk Ratio (M‐H, Random, 95% CI) | 0.39 [0.26, 0.59] |
| 2.72 Granisetron versus Perphenazine | 2 | 190 | Risk Ratio (M‐H, Random, 95% CI) | 0.36 [0.21, 0.62] |
| 2.73 Granisetron versus Ramosetron | 7 | 546 | Risk Ratio (M‐H, Random, 95% CI) | 2.82 [1.69, 4.71] |
| 2.74 Haloperidol versus Metoclopramide | 1 | 85 | Risk Ratio (M‐H, Random, 95% CI) | 0.91 [0.38, 2.15] |
| 2.75 Lidocaine versus Metoclopramide | 1 | 100 | Risk Ratio (M‐H, Random, 95% CI) | 3.25 [0.35, 30.19] |
| 2.76 Lorazepam versus Promethazine | 1 | 138 | Risk Ratio (M‐H, Random, 95% CI) | 6.36 [0.79, 51.43] |
| 2.77 Lorazepam versus Trimeprazine | 1 | 199 | Risk Ratio (M‐H, Random, 95% CI) | 3.42 [1.83, 6.40] |
| 2.78 Metoclopramide versus Nabilone | 1 | 53 | Risk Ratio (M‐H, Random, 95% CI) | 1.24 [0.79, 1.93] |
| 2.79 Metoclopramide versus Ondansetron | 43 | 2624 | Risk Ratio (M‐H, Random, 95% CI) | 1.48 [1.23, 1.77] |
| 2.80 Metoclopramide versus Perphenazine | 3 | 327 | Risk Ratio (M‐H, Random, 95% CI) | 0.75 [0.37, 1.54] |
| 2.81 Metoclopramide versus Prochlorperazine | 1 | 34 | Risk Ratio (M‐H, Random, 95% CI) | 2.5 [0.56, 11.16] |
| 2.82 Metoclopramide versus Promethazine | 1 | 11 | Risk Ratio (M‐H, Random, 95% CI) | 2.4 [0.30, 19.34] |
| 2.83 Metoclopramide versus Propofol | 2 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 3.48 [0.78, 15.46] |
| 2.84 Metoclopramide versus Tiapride | 1 | 26 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.07, 14.34] |
| 2.85 Metoclopramide versus Trimethobenzamide | 1 | 95 | Risk Ratio (M‐H, Random, 95% CI) | 0.98 [0.40, 2.39] |
| 2.86 Metoclopramide versus Tropisetron | 5 | 257 | Risk Ratio (M‐H, Random, 95% CI) | 1.33 [0.70, 2.53] |
| 2.87 Ondansetron versus Perphenazine | 1 | 216 | Risk Ratio (M‐H, Random, 95% CI) | 1.07 [0.79, 1.46] |
| 2.88 Ondansetron versus Prochlorperazine | 4 | 324 | Risk Ratio (M‐H, Random, 95% CI) | 0.87 [0.50, 1.50] |
| 2.89 Ondansetron versus Prometazine | 4 | 98 | Risk Ratio (M‐H, Random, 95% CI) | 0.84 [0.48, 1.45] |
| 2.90 Ondansetron versus Tropisetron | 8 | 406 | Risk Ratio (M‐H, Random, 95% CI) | 1.53 [1.15, 2.04] |
| 2.91 Pentobarbitone versus Trimeprazine | 2 | 78 | Risk Ratio (M‐H, Random, 95% CI) | 1.19 [0.33, 4.32] |
| 3 Nausea or Vomiting | 167 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 3.1 Alizapride versus Droperidol | 1 | 35 | Risk Ratio (M‐H, Random, 95% CI) | 1.43 [0.58, 3.55] |
| 3.2 Alizapride versus Metoclopramide | 1 | 35 | Risk Ratio (M‐H, Random, 95% CI) | 0.82 [0.43, 1.56] |
| 3.3 Alizapride versus Ondansetron | 1 | 52 | Risk Ratio (M‐H, Random, 95% CI) | 4.32 [0.52, 36.09] |
| 3.4 Atropine versus Chlorpromazine | 1 | 25 | Risk Ratio (M‐H, Random, 95% CI) | 2.0 [0.24, 16.61] |
| 3.5 Atropine versus Glycopyrrolate | 2 | 71 | Risk Ratio (M‐H, Random, 95% CI) | 0.65 [0.20, 2.17] |
| 3.6 Atropine versus Hyoscine | 2 | 100 | Risk Ratio (M‐H, Random, 95% CI) | 2.79 [1.74, 4.45] |
| 3.7 Butorphanol versus Droperidol | 1 | 20 | Risk Ratio (M‐H, Random, 95% CI) | 2.5 [0.63, 10.00] |
| 3.8 Chlorpromazine versus Droperidol | 1 | 16 | Risk Ratio (M‐H, Random, 95% CI) | 3.0 [0.85, 10.63] |
| 3.9 Chlorpromazine versus Lidocaine | 1 | 16 | Risk Ratio (M‐H, Random, 95% CI) | 2.0 [0.75, 5.33] |
| 3.10 Cimetidine versus Metoclopramide | 1 | 14 | Risk Ratio (M‐H, Random, 95% CI) | 2.0 [0.23, 17.34] |
| 3.11 Clonidine versus Diazepam | 1 | 100 | Risk Ratio (M‐H, Random, 95% CI) | 1.07 [0.78, 1.45] |
| 3.12 Clonidine versus Midazolam | 3 | 175 | Risk Ratio (M‐H, Random, 95% CI) | 0.75 [0.41, 1.37] |
| 3.13 Clonidine versus Neostigmine | 2 | 23 | Risk Ratio (M‐H, Random, 95% CI) | 0.31 [0.11, 0.86] |
| 3.14 Cyclizine versus Droperidol | 1 | 50 | Risk Ratio (M‐H, Random, 95% CI) | 0.7 [0.32, 1.54] |
| 3.15 Cyclizine versus Metoclopramide | 1 | 55 | Risk Ratio (M‐H, Random, 95% CI) | 2.07 [0.99, 4.34] |
| 3.16 Cyclizine versus Ondansetron | 3 | 190 | Risk Ratio (M‐H, Random, 95% CI) | 1.19 [0.73, 1.95] |
| 3.17 Cyclizine versus Perphenazine | 2 | 440 | Risk Ratio (M‐H, Random, 95% CI) | 0.85 [0.58, 1.26] |
| 3.18 Dexamethasone versus Dolasetron | 1 | 14 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.30, 3.35] |
| 3.19 Dexamethasone versus Droperidol | 6 | 313 | Risk Ratio (M‐H, Random, 95% CI) | 1.04 [0.72, 1.52] |
| 3.20 Dexamethasone versus Granisetron | 1 | 40 | Risk Ratio (M‐H, Random, 95% CI) | 2.5 [0.94, 6.66] |
| 3.21 Dexamethasone versus Metoclopramide | 3 | 92 | Risk Ratio (M‐H, Random, 95% CI) | 0.59 [0.35, 0.99] |
| 3.22 Dexamethasone versus Ondansetron | 7 | 304 | Risk Ratio (M‐H, Random, 95% CI) | 1.23 [0.96, 1.59] |
| 3.23 Dexamethasone versus Tropisetron | 2 | 78 | Risk Ratio (M‐H, Random, 95% CI) | 0.41 [0.22, 0.78] |
| 3.24 Diazepam versus Flunitrazepam | 3 | 287 | Risk Ratio (M‐H, Random, 95% CI) | 1.41 [0.44, 4.56] |
| 3.25 Diazepam versus Heptabarbitone | 1 | 48 | Risk Ratio (M‐H, Random, 95% CI) | 1.28 [0.73, 2.27] |
| 3.26 Diazepam versus Lorazepam | 1 | 46 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.60, 1.66] |
| 3.27 Diazepam versus Pentobarbitone | 1 | 50 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 3.28 Diazepam versus Promethazine | 1 | 20 | Risk Ratio (M‐H, Random, 95% CI) | 0.86 [0.45, 1.64] |
| 3.29 Difenidol versus Metoclopramide | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 0.4 [0.10, 1.55] |
| 3.30 Difenidol versus Ondansetron | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 2.0 [0.22, 18.33] |
| 3.31 Dimenhydrinate versus Droperidol | 2 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 1.31 [0.58, 2.96] |
| 3.32 Dimenhydrinate versus Metoclopramide | 2 | 43 | Risk Ratio (M‐H, Random, 95% CI) | 1.09 [0.44, 2.70] |
| 3.33 Dimenhydrinate versus Ondansetron | 1 | 32 | Risk Ratio (M‐H, Random, 95% CI) | 0.85 [0.38, 1.89] |
| 3.34 Dolasetron versus Droperidol | 4 | 161 | Risk Ratio (M‐H, Random, 95% CI) | 0.95 [0.77, 1.17] |
| 3.35 Dolasetron versus Metoclopramide | 4 | 227 | Risk Ratio (M‐H, Random, 95% CI) | 0.70 [0.47, 1.04] |
| 3.36 Dolasetron versus Ondansetron | 4 | 327 | Risk Ratio (M‐H, Random, 95% CI) | 1.03 [0.83, 1.27] |
| 3.37 Dolasetron versus Tropisetron | 1 | 41 | Risk Ratio (M‐H, Random, 95% CI) | 1.05 [0.76, 1.45] |
| 3.38 Domperidone versus Droperidol | 3 | 102 | Risk Ratio (M‐H, Random, 95% CI) | 1.80 [1.05, 3.08] |
| 3.39 Domperidone versus Granisetron | 1 | 100 | Risk Ratio (M‐H, Random, 95% CI) | 3.71 [1.78, 7.76] |
| 3.40 Domperidone versus Metoclopramide | 7 | 335 | Risk Ratio (M‐H, Random, 95% CI) | 0.90 [0.72, 1.13] |
| 3.41 Droperidol versus Ephedrine | 1 | 32 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.41, 2.45] |
| 3.42 Droperidol versus Granisetron | 18 | 744 | Risk Ratio (M‐H, Random, 95% CI) | 2.08 [1.55, 2.80] |
| 3.43 Droperidol versus Hydroxyzine | 1 | 50 | Risk Ratio (M‐H, Random, 95% CI) | 3.67 [1.16, 11.58] |
| 3.44 Droperidol versus Hyoscine | 1 | 40 | Risk Ratio (M‐H, Random, 95% CI) | 0.65 [0.42, 1.00] |
| 3.45 Droperidol versus Lidocaine | 1 | 10 | Risk Ratio (M‐H, Random, 95% CI) | 0.5 [0.06, 3.91] |
| 3.46 Droperidol versus Metoclopramide | 23 | 742 | Risk Ratio (M‐H, Random, 95% CI) | 0.77 [0.65, 0.91] |
| 3.47 Droperidol versus Midazolam | 1 | 150 | Risk Ratio (M‐H, Random, 95% CI) | 0.70 [0.55, 0.88] |
| 3.48 Droperidol versus Ondansetron | 23 | 1572 | Risk Ratio (M‐H, Random, 95% CI) | 0.99 [0.88, 1.12] |
| 3.49 Droperidol versus Phenobarbitone | 1 | 56 | Risk Ratio (M‐H, Random, 95% CI) | 0.69 [0.39, 1.24] |
| 3.50 Droperidol versus Propofol | 2 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 2.98 [1.08, 8.24] |
| 3.51 Droperidol versus Ranitidine | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.92 [0.48, 1.74] |
| 3.52 Droperidol versus Sulpiride | 1 | 38 | Risk Ratio (M‐H, Random, 95% CI) | 2.40 [0.78, 7.45] |
| 3.53 Droperidol versus Tropisetron | 3 | 218 | Risk Ratio (M‐H, Random, 95% CI) | 1.03 [0.81, 1.30] |
| 3.54 Ephedrine versus Propofol | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 0.75 [0.34, 1.64] |
| 3.55 Ginger versus Metoclopramide | 2 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.94 [0.57, 1.53] |
| 3.56 Glycopyrrolate versus Hyoscine | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 2.67 [1.50, 4.74] |
| 3.57 Granisetron versus Metoclopramide | 11 | 318 | Risk Ratio (M‐H, Random, 95% CI) | 0.38 [0.27, 0.55] |
| 3.58 Granisetron versus Ondansetron | 1 | 13 | Risk Ratio (M‐H, Random, 95% CI) | 1.17 [0.36, 3.76] |
| 3.59 Granisetron versus Ramosetron | 5 | 416 | Risk Ratio (M‐H, Random, 95% CI) | 2.50 [1.18, 5.29] |
| 3.60 Granisetron versus Tropisetron | 1 | 12 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.32, 3.10] |
| 3.61 Metoclopramide versus Ondansetron | 22 | 1016 | Risk Ratio (M‐H, Random, 95% CI) | 1.28 [1.03, 1.58] |
| 3.62 Metoclopramide versus Perphenazine | 1 | 25 | Risk Ratio (M‐H, Random, 95% CI) | 0.92 [0.06, 13.18] |
| 3.63 Metoclopramide versus Promethazine | 1 | 11 | Risk Ratio (M‐H, Random, 95% CI) | 2.4 [0.30, 19.34] |
| 3.64 Metoclopramide versus Propofol | 2 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 3.48 [1.29, 9.39] |
| 3.65 Metoclopramide versus Tiapride | 1 | 26 | Risk Ratio (M‐H, Random, 95% CI) | 2.0 [0.21, 19.44] |
| 3.66 Metoclopramide versus Trimethobenzamide | 1 | 95 | Risk Ratio (M‐H, Random, 95% CI) | 0.90 [0.44, 1.83] |
| 3.67 Metoclopramide versus Tropisetron | 4 | 142 | Risk Ratio (M‐H, Random, 95% CI) | 1.20 [0.88, 1.62] |
| 3.68 Ondansetron versus Prochlorperazine | 4 | 387 | Risk Ratio (M‐H, Random, 95% CI) | 0.61 [0.43, 0.87] |
| 3.69 Ondansetron versus Prometazine | 3 | 83 | Risk Ratio (M‐H, Random, 95% CI) | 0.75 [0.46, 1.22] |
| 3.70 Ondansetron versus Tropisetron | 7 | 300 | Risk Ratio (M‐H, Random, 95% CI) | 1.09 [0.88, 1.36] |
| 4 Rescue antiemetic | 167 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 4.1 Alizapride versus Droperidol | 1 | 35 | Risk Ratio (M‐H, Random, 95% CI) | 1.17 [0.45, 3.03] |
| 4.2 Alizapride versus Metoclopramide | 1 | 35 | Risk Ratio (M‐H, Random, 95% CI) | 0.94 [0.40, 2.18] |
| 4.3 Atropine versus Glycopyrrolate | 3 | 231 | Risk Ratio (M‐H, Random, 95% CI) | 0.69 [0.21, 2.27] |
| 4.4 Atropine versus Hyoscine | 2 | 100 | Risk Ratio (M‐H, Random, 95% CI) | 3.0 [1.49, 6.03] |
| 4.5 Butorphanol versus Droperidol | 1 | 20 | Risk Ratio (M‐H, Random, 95% CI) | 7.0 [0.41, 120.16] |
| 4.6 Cimetidine versus Metoclopramide | 1 | 14 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.08, 13.02] |
| 4.7 Clonidine versus Diazepam | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 5.0 [0.62, 40.28] |
| 4.8 Clonidine versus Droperidol | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 0.11 [0.01, 1.90] |
| 4.9 CP‐122,721 versus Ondansetron | 1 | 52 | Risk Ratio (M‐H, Random, 95% CI) | 0.75 [0.45, 1.25] |
| 4.10 Cyclizine versus Dexamethasone | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 0.22 [0.06, 0.86] |
| 4.11 Cyclizine versus Ondansetron | 3 | 183 | Risk Ratio (M‐H, Random, 95% CI) | 0.65 [0.30, 1.39] |
| 4.12 Dexamethasone versus Droperidol | 5 | 274 | Risk Ratio (M‐H, Random, 95% CI) | 1.18 [0.68, 2.06] |
| 4.13 Dexamethasone versus Granisetron | 3 | 104 | Risk Ratio (M‐H, Random, 95% CI) | 7.95 [1.03, 61.15] |
| 4.14 Dexamethasone versus Metoclopramide | 2 | 78 | Risk Ratio (M‐H, Random, 95% CI) | 0.51 [0.19, 1.36] |
| 4.15 Dexamethasone versus Ondansetron | 7 | 304 | Risk Ratio (M‐H, Random, 95% CI) | 1.16 [0.76, 1.78] |
| 4.16 Dexamethasone versus Perphenazine | 1 | 226 | Risk Ratio (M‐H, Random, 95% CI) | 1.84 [0.81, 4.17] |
| 4.17 Dexamethasone versus Tropisetron | 2 | 78 | Risk Ratio (M‐H, Random, 95% CI) | 0.44 [0.19, 1.04] |
| 4.18 Diazepam versus Droperidol | 1 | 65 | Risk Ratio (M‐H, Random, 95% CI) | 2.44 [1.13, 5.30] |
| 4.19 Diazepam versus Pentobarbitone | 2 | 75 | Risk Ratio (M‐H, Random, 95% CI) | 1.38 [0.28, 6.91] |
| 4.20 Diazepam versus Promethazine | 1 | 20 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.42, 2.40] |
| 4.21 Diazepam versus Trimeprazine | 1 | 25 | Risk Ratio (M‐H, Random, 95% CI) | 2.77 [0.33, 23.14] |
| 4.22 Dimenhydrinate versus Droperidol | 1 | 24 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.07, 14.21] |
| 4.23 Dimenhydrinate versus Metoclopramide | 1 | 26 | Risk Ratio (M‐H, Random, 95% CI) | 2.0 [0.21, 19.44] |
| 4.24 Dimenhydrinate versus Ondansetron | 3 | 156 | Risk Ratio (M‐H, Random, 95% CI) | 0.95 [0.64, 1.43] |
| 4.25 Dixyrazine versus Droperidol | 1 | 21 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 4.26 Dolasetron versus Droperidol | 1 | 53 | Risk Ratio (M‐H, Random, 95% CI) | 1.06 [0.79, 1.42] |
| 4.27 Dolasetron versus Metoclopramide | 2 | 180 | Risk Ratio (M‐H, Random, 95% CI) | 0.55 [0.33, 0.94] |
| 4.28 Dolasetron versus Ondansetron | 6 | 732 | Risk Ratio (M‐H, Random, 95% CI) | 0.97 [0.77, 1.22] |
| 4.29 Dolasetron versus Tropisetron | 1 | 41 | Risk Ratio (M‐H, Random, 95% CI) | 1.05 [0.76, 1.45] |
| 4.30 Domperidone versus Droperidol | 1 | 36 | Risk Ratio (M‐H, Random, 95% CI) | 1.67 [0.56, 4.95] |
| 4.31 Domperidone versus Granisetron | 1 | 100 | Risk Ratio (M‐H, Random, 95% CI) | 23.0 [1.39, 380.01] |
| 4.32 Domperidone versus Metoclopramide | 3 | 186 | Risk Ratio (M‐H, Random, 95% CI) | 0.93 [0.58, 1.48] |
| 4.33 Droperidol versus Ephedrine | 2 | 48 | Risk Ratio (M‐H, Random, 95% CI) | 0.80 [0.24, 2.59] |
| 4.34 Droperidol versus Ginger | 1 | 19 | Risk Ratio (M‐H, Random, 95% CI) | 0.30 [0.01, 6.62] |
| 4.35 Droperidol versus Granisetron | 19 | 850 | Risk Ratio (M‐H, Random, 95% CI) | 2.77 [1.82, 4.21] |
| 4.36 Droperidol versus Hyoscine | 1 | 40 | Risk Ratio (M‐H, Random, 95% CI) | 0.25 [0.08, 0.75] |
| 4.37 Droperidol versus Lidocaine | 1 | 50 | Risk Ratio (M‐H, Random, 95% CI) | 0.07 [0.00, 1.11] |
| 4.38 Droperidol versus Metoclopramide | 23 | 639 | Risk Ratio (M‐H, Random, 95% CI) | 0.84 [0.64, 1.11] |
| 4.39 Droperidol versus Midazolam | 1 | 150 | Risk Ratio (M‐H, Random, 95% CI) | 0.76 [0.54, 1.06] |
| 4.40 Droperidol versus Ondansetron | 38 | 2773 | Risk Ratio (M‐H, Random, 95% CI) | 1.01 [0.89, 1.14] |
| 4.41 Droperidol versus Phenobarbitone | 1 | 56 | Risk Ratio (M‐H, Random, 95% CI) | 0.75 [0.11, 4.95] |
| 4.42 Droperidol versus Propofol | 2 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 2.93 [0.63, 13.61] |
| 4.43 Droperidol versus Ranitidine | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.33 [0.07, 1.52] |
| 4.44 Droperidol versus Tropisetron | 4 | 234 | Risk Ratio (M‐H, Random, 95% CI) | 1.11 [0.81, 1.52] |
| 4.45 Ginger versus Metoclopramide | 2 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.43 [0.13, 1.43] |
| 4.46 Glycopyrrolate versus Hyoscine | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 2.43 [1.18, 4.99] |
| 4.47 Granisetron versus Metoclopramide | 11 | 318 | Risk Ratio (M‐H, Random, 95% CI) | 0.32 [0.17, 0.62] |
| 4.48 Granisetron versus Ondansetron | 2 | 29 | Risk Ratio (M‐H, Random, 95% CI) | 1.14 [0.39, 3.31] |
| 4.49 Granisetron versus Perphenazine | 2 | 190 | Risk Ratio (M‐H, Random, 95% CI) | 0.06 [0.01, 0.43] |
| 4.50 Granisetron versus Ramosetron | 3 | 236 | Risk Ratio (M‐H, Random, 95% CI) | 0.5 [0.10, 2.40] |
| 4.51 Granisetron versus Tropisetron | 2 | 28 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.35, 2.82] |
| 4.52 Metoclopramide versus Nabilone | 1 | 53 | Risk Ratio (M‐H, Random, 95% CI) | 1.22 [0.81, 1.84] |
| 4.53 Metoclopramide versus Ondansetron | 27 | 1776 | Risk Ratio (M‐H, Random, 95% CI) | 1.11 [0.98, 1.26] |
| 4.54 Metoclopramide versus Propofol | 2 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 3.48 [0.78, 15.46] |
| 4.55 Metoclopramide versus Tiapride | 1 | 26 | Risk Ratio (M‐H, Random, 95% CI) | 3.0 [0.13, 67.51] |
| 4.56 Metoclopramide versus Tropisetron | 5 | 182 | Risk Ratio (M‐H, Random, 95% CI) | 1.31 [0.93, 1.85] |
| 4.57 Ondansetron versus Perphenazine | 1 | 216 | Risk Ratio (M‐H, Random, 95% CI) | 0.56 [0.17, 1.86] |
| 4.58 Ondansetron versus Prochlorperazine | 3 | 241 | Risk Ratio (M‐H, Random, 95% CI) | 1.46 [0.65, 3.28] |
| 4.59 Ondansetron versus Promethazine | 1 | 15 | Risk Ratio (M‐H, Random, 95% CI) | 1.14 [0.09, 15.08] |
| 4.60 Ondansetron versus Tropisetron | 8 | 365 | Risk Ratio (M‐H, Random, 95% CI) | 1.09 [0.88, 1.36] |
| 4.61 Pentobarbitone versus Trimeprazine | 1 | 24 | Risk Ratio (M‐H, Random, 95% CI) | 2.0 [0.21, 19.23] |
Comparison 4.
PRIMARY ANALYSIS: Placebo versus Drugs
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Nausea | 25 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 1.1 Cimetidine and Metoclopramide | 1 | 14 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.08, 13.02] |
| 1.2 Clonidine and Neostigmine | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 0.51 [0.17, 1.52] |
| 1.3 Cyclizine and Ondansetron | 1 | 40 | Risk Ratio (M‐H, Random, 95% CI) | 0.56 [0.16, 1.92] |
| 1.4 Dexamethasone and Droperidol | 1 | 25 | Risk Ratio (M‐H, Random, 95% CI) | 0.27 [0.04, 2.10] |
| 1.5 Dexamethasone and Granisetron | 3 | 62 | Risk Ratio (M‐H, Random, 95% CI) | 0.26 [0.06, 1.12] |
| 1.6 Dexamethasone and Metoclopramide | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 0.75 [0.23, 2.44] |
| 1.7 Dexamethasone and Ondansetron | 3 | 109 | Risk Ratio (M‐H, Random, 95% CI) | 0.32 [0.17, 0.60] |
| 1.8 Dimenhydrinate and Droperidol | 2 | 63 | Risk Ratio (M‐H, Random, 95% CI) | 0.45 [0.18, 1.13] |
| 1.9 Dimenhydrinate and Metoclopramide | 2 | 44 | Risk Ratio (M‐H, Random, 95% CI) | 0.74 [0.24, 2.25] |
| 1.10 Dimenhydrinate and Ondansetron | 1 | 16 | Risk Ratio (M‐H, Random, 95% CI) | 0.25 [0.04, 1.77] |
| 1.11 Dolasetron and Droperidol | 2 | 38 | Risk Ratio (M‐H, Random, 95% CI) | 0.43 [0.09, 2.11] |
| 1.12 Droperidol and Ginger | 1 | 36 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.34, 2.91] |
| 1.13 Droperidol and Metoclopramide | 2 | 29 | Risk Ratio (M‐H, Random, 95% CI) | 1.00 [0.61, 1.65] |
| 1.14 Droperidol and Ondansetron | 2 | 58 | Risk Ratio (M‐H, Random, 95% CI) | 0.43 [0.11, 1.67] |
| 1.15 Glycopyrrolate and Neostigmine | 2 | 248 | Risk Ratio (M‐H, Random, 95% CI) | 1.38 [0.95, 1.99] |
| 1.16 Ondansetron and Promethazine | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 0.33 [0.10, 1.11] |
| 2 Vomiting | 30 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 2.1 Atropine and Edrophonium | 1 | 38 | Risk Ratio (M‐H, Random, 95% CI) | 1.38 [0.72, 2.64] |
| 2.2 Cimetidine and Dimetindene | 1 | 144 | Risk Ratio (M‐H, Random, 95% CI) | 0.20 [0.07, 0.54] |
| 2.3 Cimetidine and Metoclopramide | 1 | 14 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.08, 13.02] |
| 2.4 Clonidine and Neostigmine | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 2.5 Cyclizine and Ondansetron | 1 | 40 | Risk Ratio (M‐H, Random, 95% CI) | 0.07 [0.01, 0.50] |
| 2.6 Dexamethasone and Droperidol | 1 | 25 | Risk Ratio (M‐H, Random, 95% CI) | 0.36 [0.04, 3.02] |
| 2.7 Dexamethasone and Granisetron. | 2 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.28 [0.06, 1.23] |
| 2.8 Dexamethasone and Metoclopramide | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 0.5 [0.12, 2.08] |
| 2.9 Dexamethasone and Ondansetron | 3 | 109 | Risk Ratio (M‐H, Random, 95% CI) | 0.31 [0.14, 0.70] |
| 2.10 Dimenhydrinate and Droperidol | 2 | 63 | Risk Ratio (M‐H, Random, 95% CI) | 0.31 [0.08, 1.17] |
| 2.11 Dimenhydrinate and Metoclopramide | 2 | 44 | Risk Ratio (M‐H, Random, 95% CI) | 0.40 [0.09, 1.85] |
| 2.12 Dimenhydrinate and Ondansetron | 1 | 16 | Risk Ratio (M‐H, Random, 95% CI) | 0.50 [0.06, 4.47] |
| 2.13 Dolasetron and Droperidol | 2 | 38 | Risk Ratio (M‐H, Random, 95% CI) | 0.33 [0.08, 1.45] |
| 2.14 Droperidol and Ginger | 1 | 36 | Risk Ratio (M‐H, Random, 95% CI) | 0.78 [0.25, 2.39] |
| 2.15 Droperidol and Metoclopramide | 3 | 52 | Risk Ratio (M‐H, Random, 95% CI) | 0.68 [0.27, 1.71] |
| 2.16 Droperidol and Ondansetron | 3 | 126 | Risk Ratio (M‐H, Random, 95% CI) | 0.36 [0.19, 0.67] |
| 2.17 Droperidol and Phenoperidine | 1 | 163 | Risk Ratio (M‐H, Random, 95% CI) | 0.59 [0.27, 1.29] |
| 2.18 Glycopyrrolate and Neostigmine | 3 | 286 | Risk Ratio (M‐H, Random, 95% CI) | 0.97 [0.68, 1.38] |
| 2.19 Metoclopramide and Ondansetron | 1 | 133 | Risk Ratio (M‐H, Random, 95% CI) | 0.33 [0.16, 0.67] |
| 2.20 Ondansetron and Promethazine | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 0.25 [0.09, 0.72] |
| 3 Nausea or Vomiting | 26 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 3.1 Atropine and Edrophonium | 1 | 40 | Risk Ratio (M‐H, Random, 95% CI) | 1.25 [0.39, 3.99] |
| 3.2 Cimetidine and Metoclopramide | 1 | 14 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.08, 13.02] |
| 3.3 Clonidine and Neostigmine | 2 | 32 | Risk Ratio (M‐H, Random, 95% CI) | 1.59 [0.12, 21.80] |
| 3.4 Cyclizine and Ondansetron | 1 | 40 | Risk Ratio (M‐H, Random, 95% CI) | 0.63 [0.43, 0.92] |
| 3.5 Dexamethasone and Droperidol | 1 | 25 | Risk Ratio (M‐H, Random, 95% CI) | 0.31 [0.08, 1.21] |
| 3.6 Dexamethasone and Granisetron | 1 | 46 | Risk Ratio (M‐H, Random, 95% CI) | 0.10 [0.01, 0.72] |
| 3.7 Dexamethasone and Metoclopramide | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 0.67 [0.28, 1.58] |
| 3.8 Dexamethasone and Ondansetron | 3 | 109 | Risk Ratio (M‐H, Random, 95% CI) | 0.33 [0.22, 0.49] |
| 3.9 Dimenhydrinate and Droperidol | 2 | 63 | Risk Ratio (M‐H, Random, 95% CI) | 0.45 [0.18, 1.13] |
| 3.10 Dimenhydrinate and Metoclopramide | 2 | 44 | Risk Ratio (M‐H, Random, 95% CI) | 0.58 [0.23, 1.46] |
| 3.11 Dolasetron and Droperidol | 2 | 38 | Risk Ratio (M‐H, Random, 95% CI) | 0.35 [0.12, 1.03] |
| 3.12 Droperidol and Ondansetron | 4 | 117 | Risk Ratio (M‐H, Random, 95% CI) | 0.38 [0.18, 0.81] |
| 3.13 Glycopyrrolate and Neostigmine | 4 | 434 | Risk Ratio (M‐H, Random, 95% CI) | 1.03 [0.86, 1.23] |
| 3.14 Metoclopramide and Ondansetron | 1 | 16 | Risk Ratio (M‐H, Random, 95% CI) | 0.67 [0.30, 1.48] |
| 3.15 Ondansetron and Promethazine | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 0.35 [0.17, 0.72] |
| 4 Rescue antiemetic | 21 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 4.1 Atropine and Edrophonium | 1 | 38 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.07, 14.85] |
| 4.2 Cimetidine and Metoclopramide | 1 | 14 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 4.3 Cyclizine and Ondansetron | 1 | 40 | Risk Ratio (M‐H, Random, 95% CI) | 0.08 [0.01, 0.66] |
| 4.4 Dexamethasone and Droperidol | 1 | 25 | Risk Ratio (M‐H, Random, 95% CI) | 0.43 [0.10, 1.83] |
| 4.5 Dexamethasone and Granisetron | 2 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.11 [0.01, 1.95] |
| 4.6 Dexamethasone and Ondansetron | 2 | 91 | Risk Ratio (M‐H, Random, 95% CI) | 0.19 [0.07, 0.52] |
| 4.7 Dimenhydrinate and Droperidol | 1 | 24 | Risk Ratio (M‐H, Random, 95% CI) | 0.33 [0.04, 2.77] |
| 4.8 Dimenhydrinate and Metoclopramide | 1 | 26 | Risk Ratio (M‐H, Random, 95% CI) | 0.33 [0.04, 2.80] |
| 4.9 Dimenhydrinate and Ondansetron | 1 | 16 | Risk Ratio (M‐H, Random, 95% CI) | 0.2 [0.03, 1.35] |
| 4.10 Droperidol and Ginger | 1 | 36 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 4.11 Droperidol and Metoclopramide | 1 | 12 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.60, 1.66] |
| 4.12 Droperidol and Ondansetron | 5 | 143 | Risk Ratio (M‐H, Random, 95% CI) | 0.32 [0.14, 0.76] |
| 4.13 Glycopyrrolate and Neostigmine | 3 | 224 | Risk Ratio (M‐H, Random, 95% CI) | 1.44 [0.70, 2.95] |
| 4.14 Ondansetron and Promethazine | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 0.13 [0.01, 1.16] |
Comparison 5.
PRIMARY ANALYSIS: No Treatment versus Drugs
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Nausea | 5 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 1.1 Atropine and Chlorpromazine | 1 | 26 | Risk Ratio (M‐H, Random, 95% CI) | 1.36 [0.10, 19.50] |
| 1.2 Atropine and neostigmine | 2 | 118 | Risk Ratio (M‐H, Random, 95% CI) | 1.57 [0.96, 2.59] |
| 1.3 Dexamethasone and Droperidol and Metoclopramide and Ondansetron | 1 | 115 | Risk Ratio (M‐H, Random, 95% CI) | 0.34 [0.21, 0.56] |
| 1.4 Glycopyrrolate and Neostigmine | 1 | 40 | Risk Ratio (M‐H, Random, 95% CI) | 2.6 [1.14, 5.93] |
| 2 Vomiting | 5 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 2.1 Atropine and Chlorpromazine | 1 | 26 | Risk Ratio (M‐H, Random, 95% CI) | 0.23 [0.03, 1.63] |
| 2.2 Atropine and Neostigmine | 2 | 118 | Risk Ratio (M‐H, Random, 95% CI) | 2.19 [0.77, 6.21] |
| 2.3 Dexamethasone and Droperidol and Metoclopramide and Ondansetron | 1 | 115 | Risk Ratio (M‐H, Random, 95% CI) | 0.11 [0.03, 0.45] |
| 2.4 Glycopyrrolate and Neostigmine | 1 | 40 | Risk Ratio (M‐H, Random, 95% CI) | 2.25 [0.83, 6.13] |
| 3 Nausea or Vomiting | 5 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 3.1 Atropine and Chlorpromazine | 1 | 26 | Risk Ratio (M‐H, Random, 95% CI) | 0.39 [0.10, 1.53] |
| 3.2 Atropine and Diazepam | 1 | 50 | Risk Ratio (M‐H, Random, 95% CI) | 0.09 [0.01, 1.56] |
| 3.3 Atropine and Neostigmine | 1 | 38 | Risk Ratio (M‐H, Random, 95% CI) | 2.17 [1.05, 4.49] |
| 3.4 Dexamethasone and Droperidol and Metoclopramide and Ondansetron | 1 | 115 | Risk Ratio (M‐H, Random, 95% CI) | 0.34 [0.21, 0.56] |
| 3.5 Droperidol and Metoprolol | 1 | 56 | Risk Ratio (M‐H, Random, 95% CI) | 0.33 [0.04, 3.01] |
| 4 Rescue antiemetic | 3 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 4.1 Atropine and Diazepam | 1 | 50 | Risk Ratio (M‐H, Random, 95% CI) | 0.33 [0.01, 7.81] |
| 4.2 Atropine and Neostigmine | 1 | 80 | Risk Ratio (M‐H, Random, 95% CI) | 3.0 [1.06, 8.52] |
| 4.3 Glycopyrrolate and Neostigmine | 1 | 40 | Risk Ratio (M‐H, Random, 95% CI) | 1.4 [0.53, 3.68] |
Comparison 6.
PRIMARY ANALYSIS: Drugs versus Drugs
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Nausea | 8 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 1.1 Diazepam versus (Promethazine and Scopolamine) | 1 | 20 | Risk Ratio (M‐H, Random, 95% CI) | 1.33 [0.40, 4.49] |
| 1.2 Droperidol versus (Dexamethasone and Granisetron) | 3 | 172 | Risk Ratio (M‐H, Random, 95% CI) | 1.21 [0.83, 1.76] |
| 1.3 Droperidol versus (Dexamethasone and Metoclopramide) | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 1.5 [0.32, 6.94] |
| 1.4 Granisetron versus (Dexamethasone and Droperidol) | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 0.5 [0.05, 4.58] |
| 1.5 Granisetron versus (Dexamethasone and Metoclopramide) | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 0.5 [0.05, 4.58] |
| 1.6 Granisetron versus (Droperidol and Metoclopramide) | 1 | 90 | Risk Ratio (M‐H, Random, 95% CI) | 0.75 [0.18, 3.16] |
| 1.7 Metoclopramide versus (Dexamethasone and Droperidol) | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 1.5 [0.32, 6.94] |
| 1.8 Metoclopramide versus (Dexamethasone and Granisetron) | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 3.0 [0.38, 23.68] |
| 1.9 Metoclopramide versus (Dexamethasone and Ondansetron) | 1 | 19 | Risk Ratio (M‐H, Random, 95% CI) | 4.5 [0.64, 31.60] |
| 1.10 Ondansetron versus (Dimenhydrinate and Droperidol) | 1 | 161 | Risk Ratio (M‐H, Random, 95% CI) | 0.90 [0.67, 1.20] |
| 1.11 Ondansetron versus (Dexamethasone and Metoclopramide) | 1 | 17 | Risk Ratio (M‐H, Random, 95% CI) | 0.59 [0.13, 2.70] |
| 1.12 Ondansetron versus (Droperidol and Metoclopramide) | 1 | 12 | Risk Ratio (M‐H, Random, 95% CI) | 0.8 [0.41, 1.56] |
| 2 Vomiting | 12 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 2.1 (Atropine and Edrophonium) versus (Glycopyrrolate and Neostigmine) | 1 | 38 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.58, 1.72] |
| 2.2 Diazepam versus (Droperidol and Trimeprazine) | 1 | 22 | Risk Ratio (M‐H, Random, 95% CI) | 9.00 [1.36, 59.54] |
| 2.3 Diazepam versus (Promethazine and Scopolamine) | 1 | 40 | Risk Ratio (M‐H, Random, 95% CI) | 2.0 [0.58, 6.91] |
| 2.4 Droperidol versus (Dexamethasone and Granisetron) | 3 | 172 | Risk Ratio (M‐H, Random, 95% CI) | 2.11 [1.35, 3.32] |
| 2.5 Droperidol versus (Dexamethasone and Metoclopramide) | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 2.0 [0.22, 18.33] |
| 2.6 Droperidol versus (Metoclopramide and Ondansetron) | 1 | 16 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.07, 13.37] |
| 2.7 Granisetron versus (Dexamethasone and Droperidol) | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 0.5 [0.05, 4.58] |
| 2.8 Granisetron versus (Dexamethasone and Metoclopramide) | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.07, 13.64] |
| 2.9 Granisetron versus (Droperidol and Metoclopramide) | 1 | 90 | Risk Ratio (M‐H, Random, 95% CI) | 0.6 [0.15, 2.36] |
| 2.10 Metoclopramide versus (Dexamethasone and Droperidol) | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.18, 5.63] |
| 2.11 Metoclopramide versus (Dexamethasone and Granisetron) | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 5.0 [0.27, 91.52] |
| 2.12 Metoclopramide versus (Dexamethasone and Ondansetron) | 1 | 19 | Risk Ratio (M‐H, Random, 95% CI) | 5.4 [0.79, 36.68] |
| 2.13 Ondansetron versus (Dexamethasone and Metoclopramide) | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 0.5 [0.05, 4.58] |
| 2.14 Ondansetron versus (Dimenhydrinate and Droperidol) | 1 | 162 | Risk Ratio (M‐H, Random, 95% CI) | 0.80 [0.49, 1.31] |
| 2.15 Ondansetron versus (Droperidol and Metoclopramide) | 2 | 28 | Risk Ratio (M‐H, Random, 95% CI) | 0.67 [0.13, 3.53] |
| 2.16 Perphenazine versus (Droperidol and Metoclopramide) | 1 | 67 | Risk Ratio (M‐H, Random, 95% CI) | 0.97 [0.15, 6.49] |
| 2.17 Perphenazine versus (Droperidol and Ondansetron) | 1 | 67 | Risk Ratio (M‐H, Random, 95% CI) | 1.94 [0.18, 20.40] |
| 2.18 Trimeprazine versus (Diazepam and Droperidol) | 1 | 26 | Risk Ratio (M‐H, Random, 95% CI) | 1.29 [0.69, 2.39] |
| 3 Nausea or Vomiting | 7 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 3.1 Diazepam versus (Promethazine and Scopolamine) | 1 | 20 | Risk Ratio (M‐H, Random, 95% CI) | 1.5 [0.60, 3.74] |
| 3.2 Droperidol versus (Dexamethasone and Granisetron) | 3 | 172 | Risk Ratio (M‐H, Random, 95% CI) | 1.37 [0.76, 2.48] |
| 3.3 Droperidol versus (Dexamethasone and Metoclopramide) | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 1.67 [0.56, 4.97] |
| 3.4 Granisetron versus (Dexamethasone and Droperidol) | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 0.5 [0.12, 2.08] |
| 3.5 Granisetron versus (Dexamethasone and Metoclopramide) | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 0.67 [0.14, 3.09] |
| 3.6 Granisetron versus (Droperidol and Metoclopramide) | 1 | 90 | Risk Ratio (M‐H, Random, 95% CI) | 0.67 [0.26, 1.72] |
| 3.7 Metoclopramide versus (Dexamethasone and Droperidol) | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.36, 2.81] |
| 3.8 Metoclopramide versus (Dexamethasone and Granisetron) | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 4.0 [0.55, 29.17] |
| 3.9 Metoclopramide versus (Dexamethasone and Ondansetron) | 1 | 19 | Risk Ratio (M‐H, Random, 95% CI) | 2.70 [0.72, 10.14] |
| 3.10 Ondansetron versus (Dexamethasone and Metoclopramide) | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 0.5 [0.12, 2.08] |
| 3.11 Ondansetron versus (Dexamethasone and Tropisetron) | 1 | 16 | Risk Ratio (M‐H, Random, 95% CI) | 3.0 [0.39, 23.07] |
| 3.12 Tropisetron versus (Dexamethasone and Ondansetron) | 1 | 16 | Risk Ratio (M‐H, Random, 95% CI) | 0.50 [0.06, 4.47] |
| 4 Rescue antiemetic | 9 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 4.1 (Atropine and Edrophonium) versus (Glycopyrrolate and Neostigmine) | 1 | 38 | Risk Ratio (M‐H, Random, 95% CI) | 0.2 [0.03, 1.55] |
| 4.2 Diazepam versus (Promethazine and Scopolamine) | 1 | 20 | Risk Ratio (M‐H, Random, 95% CI) | 1.25 [0.47, 3.33] |
| 4.3 Droperidol versus (Dexamethasone and Granisetron) | 2 | 152 | Risk Ratio (M‐H, Random, 95% CI) | 1.73 [0.79, 3.81] |
| 4.4 Droperidol versus (Dexamethasone and Metoclopramide) | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 3.0 [0.38, 23.68] |
| 4.5 Droperidol versus (Metoclopramide and Ondansetron) | 1 | 16 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 4.6 Granisetron versus (Dexamethasone and Droperidol) | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 0.5 [0.05, 4.58] |
| 4.7 Granisetron versus (Dexamethasone and Metoclopramide) | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.07, 13.64] |
| 4.8 Granisetron versus (Droperidol and Metoclopramide) | 1 | 90 | Risk Ratio (M‐H, Random, 95% CI) | 2.0 [0.19, 21.28] |
| 4.9 Metoclopramide versus (Dexamethasone and Droperidol) | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 1.5 [0.32, 6.94] |
| 4.10 Metoclopramide versus (Dexamethasone and Granisetron) | 1 | 18 | Risk Ratio (M‐H, Random, 95% CI) | 7.0 [0.41, 118.69] |
| 4.11 Ondansetron versus (Droperidol and Metoclopramide) | 3 | 228 | Risk Ratio (M‐H, Random, 95% CI) | 1.00 [0.38, 2.63] |
| 4.12 Perphenazine versus (Droperidol and Metoclopramide) | 1 | 67 | Risk Ratio (M‐H, Random, 95% CI) | 0.85 [0.35, 2.08] |
| 4.13 Perphenazine versus (Droperidol and Ondansetron) | 1 | 67 | Risk Ratio (M‐H, Random, 95% CI) | 0.97 [0.38, 2.46] |
Comparison 7.
PRIMARY ANALYSIS: Side effects; Placebo versus Drug
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Dizziness or vertigo | 7 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 1.1 Neostigmine | 2 | 86 | Risk Ratio (M‐H, Random, 95% CI) | 6.82 [1.31, 35.41] |
| 1.2 Tropisetron | 5 | 833 | Risk Ratio (M‐H, Random, 95% CI) | 0.37 [0.14, 0.96] |
| 2 Drowsiness or sedation | 26 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 2.1 Dimenhydrinate | 2 | 335 | Risk Ratio (M‐H, Random, 95% CI) | 9.01 [2.18, 37.23] |
| 2.2 Droperidol | 24 | 2220 | Risk Ratio (M‐H, Random, 95% CI) | 1.32 [1.16, 1.51] |
| 3 Dry mouth | 10 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 3.1 Hyoscine | 10 | 806 | Risk Ratio (M‐H, Random, 95% CI) | 1.25 [1.05, 1.49] |
| 4 Extrapyramidal reaction | 8 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 4.1 Dimenhydrinate | 1 | 47 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 4.2 Droperidol | 5 | 1694 | Risk Ratio (M‐H, Random, 95% CI) | 4.37 [0.74, 25.76] |
| 4.3 Granisetron | 1 | 40 | Risk Ratio (M‐H, Random, 95% CI) | 0.33 [0.01, 7.72] |
| 4.4 Metoclopramide | 2 | 102 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 4.5 Ondansetron | 2 | 202 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 5 Headache | 65 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 5.1 Droperidol | 25 | 2343 | Risk Ratio (M‐H, Random, 95% CI) | 0.79 [0.65, 0.95] |
| 5.2 Ondansetron | 45 | 10808 | Risk Ratio (M‐H, Random, 95% CI) | 1.16 [1.03, 1.30] |
| 6 Infection | 2 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 6.1 Dexamethasone | 2 | 340 | Risk Ratio (M‐H, Random, 95% CI) | 1.5 [0.25, 8.85] |
| 6.2 Ondansetron | 1 | 46 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
Analysis 7.1.

Comparison 7 PRIMARY ANALYSIS: Side effects; Placebo versus Drug, Outcome 1 Dizziness or vertigo.
Analysis 7.2.

Comparison 7 PRIMARY ANALYSIS: Side effects; Placebo versus Drug, Outcome 2 Drowsiness or sedation.
Analysis 7.3.

Comparison 7 PRIMARY ANALYSIS: Side effects; Placebo versus Drug, Outcome 3 Dry mouth.
Analysis 7.4.

Comparison 7 PRIMARY ANALYSIS: Side effects; Placebo versus Drug, Outcome 4 Extrapyramidal reaction.
Analysis 7.5.
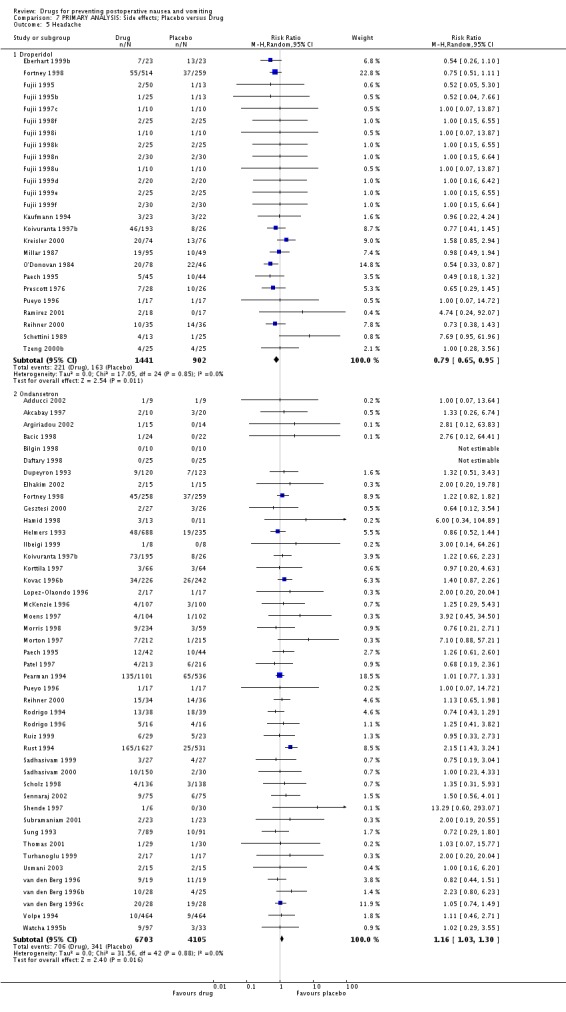
Comparison 7 PRIMARY ANALYSIS: Side effects; Placebo versus Drug, Outcome 5 Headache.
Analysis 7.6.

Comparison 7 PRIMARY ANALYSIS: Side effects; Placebo versus Drug, Outcome 6 Infection.
Comparison 8.
PRIMARY ANALYSIS: Side effects; No Treatment versus Drug
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Drowsiness or sedation | 6 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 1.1 Droperidol | 6 | 784 | Risk Ratio (M‐H, Random, 95% CI) | 2.57 [1.02, 6.43] |
| 2 Extrapyramidal reaction | 1 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 2.1 Droperidol | 1 | 12 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 2.2 Metoclopramide | 1 | 12 | Risk Ratio (M‐H, Random, 95% CI) | 3.0 [0.15, 61.74] |
Analysis 8.1.

Comparison 8 PRIMARY ANALYSIS: Side effects; No Treatment versus Drug, Outcome 1 Drowsiness or sedation.
Analysis 8.2.

Comparison 8 PRIMARY ANALYSIS: Side effects; No Treatment versus Drug, Outcome 2 Extrapyramidal reaction.
Comparison 9.
PRIMARY ANALYSIS: Side effects; Drug versus Drug
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Drowsiness or sedation | 2 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 1.1 Dimenhydrinate versus Ondansetron | 2 | 124 | Risk Ratio (M‐H, Random, 95% CI) | 7.22 [1.52, 34.36] |
Analysis 9.1.

Comparison 9 PRIMARY ANALYSIS: Side effects; Drug versus Drug, Outcome 1 Drowsiness or sedation.
Comparison 10.
SECONDARY ANALYSIS: Route versus Route
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Nausea | 3 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 1.1 Prochlorperazine intramuscular versus intravenous | 3 | 298 | Risk Ratio (M‐H, Random, 95% CI) | 0.53 [0.33, 0.83] |
| 2 Vomiting | 3 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 2.1 Prochlorperazine intramuscular versus intravenous | 3 | 327 | Risk Ratio (M‐H, Random, 95% CI) | 1.04 [0.60, 1.80] |
| 3 Nausea or Vomiting | 6 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 3.1 Droperidol epidural versus intravenous | 2 | 118 | Risk Ratio (M‐H, Random, 95% CI) | 1.32 [0.36, 4.80] |
| 3.2 Prochlorperazine intramuscular versus intravenous | 4 | 514 | Risk Ratio (M‐H, Random, 95% CI) | 0.78 [0.62, 0.97] |
| 4 Rescue antiemetic | 4 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 4.1 Droperidol epidural versus intravenous | 2 | 118 | Risk Ratio (M‐H, Random, 95% CI) | 2.84 [0.45, 18.01] |
| 4.2 Prochlorperazine intramuscular versus intravenous | 2 | 217 | Risk Ratio (M‐H, Random, 95% CI) | 0.74 [0.17, 3.19] |
Analysis 10.1.

Comparison 10 SECONDARY ANALYSIS: Route versus Route, Outcome 1 Nausea.
Analysis 10.2.

Comparison 10 SECONDARY ANALYSIS: Route versus Route, Outcome 2 Vomiting.
Analysis 10.3.

Comparison 10 SECONDARY ANALYSIS: Route versus Route, Outcome 3 Nausea or Vomiting.
Analysis 10.4.

Comparison 10 SECONDARY ANALYSIS: Route versus Route, Outcome 4 Rescue antiemetic.
Comparison 11.
SECONDARY ANALYSIS: Timing versus Timing
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Nausea | 4 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 1.1 Ondansetron | 4 | 219 | Risk Ratio (M‐H, Random, 95% CI) | 1.17 [0.79, 1.73] |
| 2 Vomiting | 7 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 2.1 Droperidol | 2 | 173 | Risk Ratio (M‐H, Random, 95% CI) | 1.15 [0.83, 1.59] |
| 2.2 Ondansetron | 5 | 339 | Risk Ratio (M‐H, Random, 95% CI) | 1.20 [0.79, 1.80] |
| 3 Nausea or Vomiting | 3 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 3.1 Droperidol | 3 | 168 | Risk Ratio (M‐H, Random, 95% CI) | 0.89 [0.56, 1.41] |
| 4 Rescue antiemetic | 5 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 4.1 Droperidol | 2 | 127 | Risk Ratio (M‐H, Random, 95% CI) | 0.69 [0.35, 1.36] |
| 4.2 Ondansetron | 3 | 248 | Risk Ratio (M‐H, Random, 95% CI) | 1.76 [1.12, 2.76] |
Analysis 11.1.

Comparison 11 SECONDARY ANALYSIS: Timing versus Timing, Outcome 1 Nausea.
Analysis 11.2.

Comparison 11 SECONDARY ANALYSIS: Timing versus Timing, Outcome 2 Vomiting.
Analysis 11.3.

Comparison 11 SECONDARY ANALYSIS: Timing versus Timing, Outcome 3 Nausea or Vomiting.
Analysis 11.4.

Comparison 11 SECONDARY ANALYSIS: Timing versus Timing, Outcome 4 Rescue antiemetic.
Comparison 12.
SECONDARY ANALYSIS: Dose versus Dose
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Nausea alizapride | 2 | 134 | Risk Ratio (M‐H, Random, 95% CI) | 1.11 [0.65, 1.88] |
| 1.1 Alizapride intravenous 50 mg versus 100 mg | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 1.13 [0.46, 2.77] |
| 1.2 Alizapride intravenous 100 mg versus 200 mg | 2 | 104 | Risk Ratio (M‐H, Random, 95% CI) | 1.28 [0.47, 3.51] |
| 2 Nausea clonidine | 2 | 90 | Risk Ratio (M‐H, Random, 95% CI) | 2.59 [0.62, 10.86] |
| 2.1 Clonidine intrathecal 25 µcg/kg versus 75 µcg/kg | 1 | 40 | Risk Ratio (M‐H, Random, 95% CI) | 1.6 [0.63, 4.05] |
| 2.2 Clonidine epidural 25 µcg/hr versus 50 µcg/kg | 1 | 50 | Risk Ratio (M‐H, Random, 95% CI) | 7.00 [0.93, 52.80] |
| 3 Nausea dexamethasone | 6 | 733 | Risk Ratio (M‐H, Random, 95% CI) | 1.41 [0.98, 2.03] |
| 3.1 Dexamethasone intravenous 1.25 mg versus 2.5 mg | 1 | 66 | Risk Ratio (M‐H, Random, 95% CI) | 1.5 [0.55, 4.12] |
| 3.2 Dexamethasone intravenous 2.5 mg versus 5 mg | 3 | 173 | Risk Ratio (M‐H, Random, 95% CI) | 1.49 [0.73, 3.07] |
| 3.3 Dexamethasone intravenous 2 mg versus 4 mg | 1 | 45 | Risk Ratio (M‐H, Random, 95% CI) | 1.5 [0.47, 4.74] |
| 3.4 Dexamethasone intravenous 4 mg versus 8 mg | 2 | 75 | Risk Ratio (M‐H, Random, 95% CI) | 1.5 [0.50, 4.49] |
| 3.5 Dexamethasone intravenous 5 mg versus 8 mg | 1 | 88 | Risk Ratio (M‐H, Random, 95% CI) | 1.34 [0.46, 3.90] |
| 3.6 Dexamethasone intravenous 5 mg versus 10 mg | 3 | 196 | Risk Ratio (M‐H, Random, 95% CI) | 1.13 [0.53, 2.43] |
| 3.7 Dexamethasone intravenous 8 mg versus 16 mg | 2 | 90 | Risk Ratio (M‐H, Random, 95% CI) | 2.0 [0.43, 9.25] |
| 4 Nausea dolasetron | 5 | 2113 | Risk Ratio (M‐H, Random, 95% CI) | 1.05 [0.95, 1.16] |
| 4.1 Dolasetron oral 25 mg versus 50 mg | 2 | 355 | Risk Ratio (M‐H, Random, 95% CI) | 1.01 [0.78, 1.31] |
| 4.2 Dolasetron oral 50 mg versus 100 mg | 2 | 234 | Risk Ratio (M‐H, Random, 95% CI) | 0.95 [0.71, 1.28] |
| 4.3 Dolasetron oral 100 mg versus 200 mg | 2 | 343 | Risk Ratio (M‐H, Random, 95% CI) | 1.01 [0.78, 1.31] |
| 4.4 Dolasetron intravenous 12.5 mg versus 25 mg | 2 | 409 | Risk Ratio (M‐H, Random, 95% CI) | 1.10 [0.59, 2.06] |
| 4.5 Dolasetron intravenous 25 mg versus 50 mg | 2 | 461 | Risk Ratio (M‐H, Random, 95% CI) | 1.23 [0.96, 1.57] |
| 4.6 Dolasetron intravenous 50 mg versus 100 mg | 1 | 311 | Risk Ratio (M‐H, Random, 95% CI) | 1.13 [0.91, 1.40] |
| 5 Nausea domperidone | 2 | 132 | Risk Ratio (M‐H, Random, 95% CI) | 0.68 [0.26, 1.79] |
| 5.1 Domperidone intravenous 5 mg versus 10 mg | 1 | 72 | Risk Ratio (M‐H, Random, 95% CI) | 0.5 [0.10, 2.56] |
| 5.2 Domperidone intramuscular 10 mg versus 15 mg | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.8 [0.24, 2.69] |
| 6 Nausea droperidol | 14 | 2324 | Risk Ratio (M‐H, Random, 95% CI) | 1.23 [1.12, 1.36] |
| 6.1 Droperidol intravenous 5 µcg/kg versus 10 µcg/kg | 1 | 32 | Risk Ratio (M‐H, Random, 95% CI) | 2.25 [0.87, 5.83] |
| 6.2 Droperidol intravenous 10 µcg/kg versus 20 µcg/kg | 2 | 173 | Risk Ratio (M‐H, Random, 95% CI) | 0.89 [0.15, 5.22] |
| 6.3 Droperidol intravenous 20 µcg/kg versus 30 µcg/kg | 1 | 39 | Risk Ratio (M‐H, Random, 95% CI) | 0.67 [0.08, 5.80] |
| 6.4 Droperidol intravenous 0.25 mg versus 0.5 mg | 1 | 95 | Risk Ratio (M‐H, Random, 95% CI) | 1.09 [0.49, 2.44] |
| 6.5 Droperidol intravenous 0.25 mg versus 1.25 mg | 1 | 78 | Risk Ratio (M‐H, Random, 95% CI) | 0.52 [0.16, 1.62] |
| 6.6 Droperidol intravenous 0.5 mg versus 1 mg | 1 | 75 | Risk Ratio (M‐H, Random, 95% CI) | 2.57 [0.87, 7.62] |
| 6.7 Droperidol intravenous 0.625 mg versus 1.25 mg | 3 | 1107 | Risk Ratio (M‐H, Random, 95% CI) | 1.02 [0.71, 1.47] |
| 6.8 Droperidol intravenous 0.75 mg versus 1.25 mg | 1 | 91 | Risk Ratio (M‐H, Random, 95% CI) | 1.06 [0.62, 1.83] |
| 6.9 Droperidol intravenous 1.25 mg versus 2.5 mg | 2 | 120 | Risk Ratio (M‐H, Random, 95% CI) | 2.25 [1.19, 4.23] |
| 6.10 Droperidol intravenous 2.5 mg versus 5 mg | 1 | 196 | Risk Ratio (M‐H, Random, 95% CI) | 1.47 [0.69, 3.11] |
| 6.11 Droperidol intravenous PCA bolus 5 µcg versus 15 µcg | 1 | 123 | Risk Ratio (M‐H, Random, 95% CI) | 1.25 [0.76, 2.05] |
| 6.12 Droperidol intravenous PCA bolus 15 µcg versus 50 µcg | 1 | 124 | Risk Ratio (M‐H, Random, 95% CI) | 1.57 [0.87, 2.84] |
| 6.13 Droperidol intravenous PCA bolus 50 µcg versus 100 µcg | 1 | 26 | Risk Ratio (M‐H, Random, 95% CI) | 1.24 [0.42, 3.65] |
| 6.14 Droperidol intravenous PCA bolus 100 µcg versus 150 µcg | 1 | 19 | Risk Ratio (M‐H, Random, 95% CI) | 1.11 [0.30, 4.17] |
| 6.15 Droperidol intravenous PCA bolus 150 µcg versus 200 µcg | 1 | 26 | Risk Ratio (M‐H, Random, 95% CI) | 0.8 [0.26, 2.50] |
| 7 Nausea ginger | 2 | 188 | Risk Ratio (M‐H, Random, 95% CI) | 1.03 [0.77, 1.39] |
| 7.1 Ginger oral 100 mg versus 200 mg | 1 | 116 | Risk Ratio (M‐H, Random, 95% CI) | 1.06 [0.76, 1.49] |
| 7.2 Ginger oral 0.5 g versus 1 g | 1 | 72 | Risk Ratio (M‐H, Random, 95% CI) | 0.92 [0.49, 1.74] |
| 8 Nausea granisetron | 11 | 1312 | Risk Ratio (M‐H, Random, 95% CI) | 1.21 [1.05, 1.40] |
| 8.1 Granisetron oral 1 mg versus 2 mg | 3 | 128 | Risk Ratio (M‐H, Random, 95% CI) | 2.17 [0.78, 6.05] |
| 8.2 Granisetron oral 2 mg versus 4 mg | 3 | 128 | Risk Ratio (M‐H, Random, 95% CI) | 1.14 [0.35, 3.67] |
| 8.3 Granisetron intravenous 2 µcg/kg versus 5 µcg/kg | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 1.64 [1.09, 2.46] |
| 8.4 Granisetron intravenous 5 µcg/kg versus 10 µcg/kg | 1 | 40 | Risk Ratio (M‐H, Random, 95% CI) | 1.1 [0.61, 1.99] |
| 8.5 Granisetron intravenous 10 µcg/kg versus 20 µcg/kg | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 1.11 [0.64, 1.94] |
| 8.6 Granisetron intravenous 20 µcg/kg versus 30 µcg/kg | 1 | 45 | Risk Ratio (M‐H, Random, 95% CI) | 2.0 [0.24, 16.36] |
| 8.7 Granisetron intravenous 20 µcg/kg versus 40 µcg/kg | 5 | 246 | Risk Ratio (M‐H, Random, 95% CI) | 1.55 [0.82, 2.91] |
| 8.8 Granisetron intravenous 30 µcg/kg versus 40 µcg/kg | 1 | 45 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.10, 10.17] |
| 8.9 Granisetron intravenous 40 µcg/kg versus 60 µcg/kg | 1 | 38 | Risk Ratio (M‐H, Random, 95% CI) | 1.92 [0.13, 28.32] |
| 8.10 Granisetron intravenous 40 µcg/kg versus 80 µcg/kg | 1 | 45 | Risk Ratio (M‐H, Random, 95% CI) | 0.67 [0.08, 5.88] |
| 8.11 Granisetron intravenous 40 µcg/kg versus 100 µcg/kg | 2 | 83 | Risk Ratio (M‐H, Random, 95% CI) | 1.19 [0.31, 4.62] |
| 8.12 Granisetron intravenous 100 µcg versus 1 mg | 1 | 199 | Risk Ratio (M‐H, Random, 95% CI) | 1.42 [1.09, 1.84] |
| 8.13 Granisetron intravenous 1 mg versus 3 mg | 1 | 195 | Risk Ratio (M‐H, Random, 95% CI) | 0.88 [0.66, 1.16] |
| 9 Nausea neostigmine | 2 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.69 [0.43, 1.09] |
| 9.1 Neostigmine intrathecal 25 µcg versus 50 µcg | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 0.88 [0.43, 1.80] |
| 9.2 Neostigmine intrathecal 100 µcg versus 200 µcg | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 0.58 [0.32, 1.06] |
| 10 Nausea ondansetron | 11 | 3545 | Risk Ratio (M‐H, Random, 95% CI) | 1.07 [1.00, 1.15] |
| 10.1 Ondansetron oral 4 mg versus 8 mg | 1 | 808 | Risk Ratio (M‐H, Random, 95% CI) | 1.01 [0.92, 1.11] |
| 10.2 Ondansetron oral 8 mg versus 16 mg | 2 | 855 | Risk Ratio (M‐H, Random, 95% CI) | 1.12 [1.02, 1.24] |
| 10.3 Ondansetron intravenous 1 mg versus 4 mg | 1 | 387 | Risk Ratio (M‐H, Random, 95% CI) | 1.13 [0.96, 1.34] |
| 10.4 Ondansetron intravenous 1 mg versus 8 mg | 1 | 345 | Risk Ratio (M‐H, Random, 95% CI) | 1.07 [0.92, 1.23] |
| 10.5 Ondansetron intravenous 4 mg versus 8 mg | 8 | 807 | Risk Ratio (M‐H, Random, 95% CI) | 1.32 [0.97, 1.79] |
| 10.6 Ondansetron intravenous 8 mg versus 16 mg | 1 | 343 | Risk Ratio (M‐H, Random, 95% CI) | 0.94 [0.81, 1.09] |
| 11 Nausea ramosetron | 3 | 226 | Risk Ratio (M‐H, Random, 95% CI) | 2.09 [0.86, 5.09] |
| 11.1 Ramosetron intravenous 0.15 mg versus 0.3 mg | 3 | 113 | Risk Ratio (M‐H, Random, 95% CI) | 3.08 [0.97, 9.83] |
| 11.2 Ramosetron intravenous 0.3 mg versus 0.6 mg | 3 | 113 | Risk Ratio (M‐H, Random, 95% CI) | 1.19 [0.30, 4.79] |
| 12 Nausea tropisetron | 2 | 291 | Risk Ratio (M‐H, Random, 95% CI) | 1.24 [0.44, 3.52] |
| 12.1 Tropisetron intravenous 2 mg versus 5 mg | 2 | 291 | Risk Ratio (M‐H, Random, 95% CI) | 1.24 [0.44, 3.52] |
| 13 Vomiting alizapride | 2 | 114 | Risk Ratio (M‐H, Random, 95% CI) | 1.09 [0.48, 2.46] |
| 13.1 Alizapride intravenous 100 mg versus 200 mg | 2 | 114 | Risk Ratio (M‐H, Random, 95% CI) | 1.09 [0.48, 2.46] |
| 14 Vomiting clonidine | 3 | 160 | Risk Ratio (M‐H, Random, 95% CI) | 2.68 [1.17, 6.16] |
| 14.1 Clonidine oral 2 µcg/kg versus 4 µcg/kg | 1 | 70 | Risk Ratio (M‐H, Random, 95% CI) | 2.5 [0.87, 7.22] |
| 14.2 Clonidine intrathecal 25 µcg/kg versus 75 µcg/kg | 1 | 40 | Risk Ratio (M‐H, Random, 95% CI) | 3.0 [0.69, 13.12] |
| 14.3 Clonidine epidural 25 µcg/hr versus 50 µcg/hr | 1 | 50 | Risk Ratio (M‐H, Random, 95% CI) | 3.0 [0.13, 70.30] |
| 15 Vomiting dexamethasone | 7 | 869 | Risk Ratio (M‐H, Random, 95% CI) | 1.57 [1.07, 2.30] |
| 15.1 Dexamethasone intravenous 1.25 mg versus 2.5 mg | 2 | 111 | Risk Ratio (M‐H, Random, 95% CI) | 1.77 [0.81, 3.86] |
| 15.2 Dexamethasone intravenous 2 mg versus 4 mg | 1 | 45 | Risk Ratio (M‐H, Random, 95% CI) | 1.55 [0.07, 35.89] |
| 15.3 Dexamethasone intravenous 2.5 mg versus 5 mg | 4 | 204 | Risk Ratio (M‐H, Random, 95% CI) | 1.45 [0.69, 3.08] |
| 15.4 Dexamethasone intravenous 4 mg versus 8 mg | 2 | 90 | Risk Ratio (M‐H, Random, 95% CI) | 2.33 [0.73, 7.41] |
| 15.5 Dexamethasone intravenous 5 mg versus 8 mg | 1 | 88 | Risk Ratio (M‐H, Random, 95% CI) | 2.87 [0.61, 13.44] |
| 15.6 Dexamethasone intravenous 5 mg versus 10 mg | 4 | 241 | Risk Ratio (M‐H, Random, 95% CI) | 1.20 [0.55, 2.60] |
| 15.7 Dexamethasone intravenous 8 mg versus 16 mg | 2 | 90 | Risk Ratio (M‐H, Random, 95% CI) | 1.19 [0.31, 4.67] |
| 16 Vomiting dolasetron | 5 | 1678 | Risk Ratio (M‐H, Random, 95% CI) | 1.11 [0.96, 1.28] |
| 16.1 Dolasetron oral 25 mg versus 50 mg | 1 | 113 | Risk Ratio (M‐H, Random, 95% CI) | 1.08 [0.79, 1.48] |
| 16.2 Dolasetron oral 50 mg versus 100 mg | 1 | 74 | Risk Ratio (M‐H, Random, 95% CI) | 1.29 [0.83, 2.01] |
| 16.3 Dolasetron oral 100 mg versus 200 mg | 1 | 112 | Risk Ratio (M‐H, Random, 95% CI) | 0.91 [0.60, 1.37] |
| 16.4 Dolasetron intravenous 12.5 mg versus 25 mg | 3 | 493 | Risk Ratio (M‐H, Random, 95% CI) | 1.21 [0.68, 2.15] |
| 16.5 Dolasetron intravenous 25 mg versus 50 mg | 3 | 518 | Risk Ratio (M‐H, Random, 95% CI) | 1.29 [0.90, 1.84] |
| 16.6 Dolasetron intravenous 50 mg versus 100 mg | 2 | 368 | Risk Ratio (M‐H, Random, 95% CI) | 0.98 [0.75, 1.29] |
| 17 Vomiting domperidone | 2 | 132 | Risk Ratio (M‐H, Random, 95% CI) | 1.03 [0.71, 1.49] |
| 17.1 Domperidone intravenous 5 mg versus 10 mg | 1 | 72 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.61, 1.63] |
| 17.2 Domperidone intramuscular 10 mg versus 15 mg | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 1.08 [0.62, 1.89] |
| 18 Vomiting droperidol | 17 | 1518 | Risk Ratio (M‐H, Random, 95% CI) | 1.26 [1.01, 1.57] |
| 18.1 Droperidol oral 50 µcg/kg versus 75 µcg/kg | 1 | 43 | Risk Ratio (M‐H, Random, 95% CI) | 0.92 [0.40, 2.08] |
| 18.2 Droperidol intravenous 5 µcg/kg versus 10 µcg/kg | 1 | 32 | Risk Ratio (M‐H, Random, 95% CI) | 1.5 [0.52, 4.32] |
| 18.3 Droperidol intravenous 10 µcg/kg versus 20 µcg/kg | 2 | 173 | Risk Ratio (M‐H, Random, 95% CI) | 2.57 [1.32, 5.03] |
| 18.4 Droperidol intravenous 20 µcg/kg versus 30 µcg/kg | 1 | 39 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.37, 2.71] |
| 18.5 Droperidol intravenous 25 µcg/kg versus 50 µcg/kg | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 1.03 [0.55, 1.93] |
| 18.6 Droperidol intravenous 0.25 mg versus 0.5 mg | 1 | 95 | Risk Ratio (M‐H, Random, 95% CI) | 0.61 [0.22, 1.74] |
| 18.7 Droperidol intravenous 0.25 mg versus 1.25 mg | 1 | 78 | Risk Ratio (M‐H, Random, 95% CI) | 0.60 [0.11, 3.40] |
| 18.8 Droperidol intravenous 0.5 mg versus 1 mg | 1 | 75 | Risk Ratio (M‐H, Random, 95% CI) | 3.42 [0.14, 81.27] |
| 18.9 Droperidol intravenous 0.625 mg versus 1.25 mg | 3 | 195 | Risk Ratio (M‐H, Random, 95% CI) | 1.09 [0.47, 2.56] |
| 18.10 Droperidol intravenous 0.75 mg versus 1.25 mg | 1 | 91 | Risk Ratio (M‐H, Random, 95% CI) | 1.28 [0.49, 3.34] |
| 18.11 Droperidol intravenous 1.25 mg versus 2.5 mg | 2 | 120 | Risk Ratio (M‐H, Random, 95% CI) | 2.84 [0.89, 9.11] |
| 18.12 Droperidol intravenous 2.5 mg versus 5 mg | 1 | 196 | Risk Ratio (M‐H, Random, 95% CI) | 1.22 [0.50, 2.97] |
| 18.13 Droperidol intravenous PCA bolus 5 µcg versus 15 µcg | 1 | 123 | Risk Ratio (M‐H, Random, 95% CI) | 1.06 [0.52, 2.12] |
| 18.14 Droperidol intravenous PCA bolus 15 µcg versus 50 µcg | 1 | 124 | Risk Ratio (M‐H, Random, 95% CI) | 1.82 [0.80, 4.13] |
| 18.15 Droperidol intravenous PCA bolus 17 µcg versus 33 µcg | 1 | 40 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.29, 3.45] |
| 18.16 Droperidol intravenous PCA bolus 50 µcg versus 100 µcg | 1 | 34 | Risk Ratio (M‐H, Random, 95% CI) | 0.75 [0.20, 2.86] |
| 19 Vomiting ginger | 2 | 188 | Risk Ratio (M‐H, Random, 95% CI) | 0.77 [0.34, 1.72] |
| 19.1 Ginger oral 100 mg versus 200 mg | 1 | 116 | Risk Ratio (M‐H, Random, 95% CI) | 1.05 [0.68, 1.62] |
| 19.2 Ginger oral 0.5 g versus 1 g | 1 | 72 | Risk Ratio (M‐H, Random, 95% CI) | 0.45 [0.18, 1.18] |
| 20 Vomiting granisetron | 18 | 1837 | Risk Ratio (M‐H, Random, 95% CI) | 1.50 [1.26, 1.79] |
| 20.1 Granisetron oral 20 µcg/kg versus 40 µcg/kg | 3 | 131 | Risk Ratio (M‐H, Random, 95% CI) | 1.76 [0.85, 3.67] |
| 20.2 Granisetron oral 40 µcg/kg versus 80 µcg/kg | 2 | 83 | Risk Ratio (M‐H, Random, 95% CI) | 1.64 [0.55, 4.90] |
| 20.3 Granisetron oral 1 mg versus 2 mg | 3 | 128 | Risk Ratio (M‐H, Random, 95% CI) | 2.22 [0.80, 6.14] |
| 20.4 Granisetron oral 2 mg versus 4 mg | 3 | 128 | Risk Ratio (M‐H, Random, 95% CI) | 1.32 [0.39, 4.44] |
| 20.5 Granisetron intravenous 2 µcg/kg versus 5 µcg/kg | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 2.21 [1.19, 4.12] |
| 20.6 Granisetron intravenous 5 µcg/kg versus 10 µcg/kg | 1 | 40 | Risk Ratio (M‐H, Random, 95% CI) | 1.4 [0.53, 3.68] |
| 20.7 Granisetron intravenous 10 µcg/kg versus 20 µcg/kg | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.39, 2.53] |
| 20.8 Granisetron intravenous 10 µcg/kg versus 40 µcg/kg | 2 | 101 | Risk Ratio (M‐H, Random, 95% CI) | 7.59 [1.86, 30.96] |
| 20.9 Granisetron intravenous 20 µcg/kg versus 30 µcg/kg | 1 | 45 | Risk Ratio (M‐H, Random, 95% CI) | 2.0 [0.48, 8.28] |
| 20.10 Granisetron intravenous 20 µcg/kg versus 40 µcg/kg | 7 | 351 | Risk Ratio (M‐H, Random, 95% CI) | 1.99 [1.13, 3.50] |
| 20.11 Granisetron intravenous 30 µcg/kg versus 40 µcg/kg | 1 | 45 | Risk Ratio (M‐H, Random, 95% CI) | 2.0 [0.31, 12.84] |
| 20.12 Granisetron intravenous 40 µcg/kg versus 60 µcg/kg | 1 | 38 | Risk Ratio (M‐H, Random, 95% CI) | 1.92 [0.13, 28.32] |
| 20.13 Granisetron intravenous 40 µcg/kg versus 80 µcg/kg | 2 | 105 | Risk Ratio (M‐H, Random, 95% CI) | 1.59 [0.46, 5.51] |
| 20.14 Granisetron intravenous 40 µcg/kg versus 100 µcg/kg | 3 | 128 | Risk Ratio (M‐H, Random, 95% CI) | 1.32 [0.39, 4.44] |
| 20.15 Granisetron intravenous 100 µcg versus 1 mg | 1 | 199 | Risk Ratio (M‐H, Random, 95% CI) | 1.48 [1.05, 2.10] |
| 20.16 Granisetron intravenous 1 mg versus 3 mg | 1 | 195 | Risk Ratio (M‐H, Random, 95% CI) | 0.97 [0.67, 1.43] |
| 21 Vomiting metoclopramide | 3 | 179 | Risk Ratio (M‐H, Random, 95% CI) | 1.82 [1.16, 2.87] |
| 21.1 Metoclopramide intravenous 0.15 mg/kg versus 0.25 mg/kg | 1 | 53 | Risk Ratio (M‐H, Random, 95% CI) | 2.38 [1.25, 4.53] |
| 21.2 Metoclopramide intravenous 10 mg versus 20 mg | 1 | 66 | Risk Ratio (M‐H, Random, 95% CI) | 1.36 [0.49, 3.77] |
| 21.3 Metoclopramide intravenous 20 mg versus 50 mg | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 1.43 [0.63, 3.25] |
| 22 Vomiting neostigmine | 2 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.83 [0.43, 1.63] |
| 22.1 Neostigmine intrathecal 25 µcg versus 50 µcg | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 0.83 [0.32, 2.15] |
| 22.2 Neostigmine intrathecal 100 µcg versus 200 µcg | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 0.83 [0.32, 2.15] |
| 23 Vomiting ondansetron | 19 | 4474 | Risk Ratio (M‐H, Random, 95% CI) | 1.15 [1.04, 1.27] |
| 23.1 Ondansetron oral 0.075 mg/kg versus 0.15 mg/kg | 1 | 91 | Risk Ratio (M‐H, Random, 95% CI) | 2.39 [0.66, 8.65] |
| 23.2 Ondansetron oral 4 mg versus 8 mg | 2 | 838 | Risk Ratio (M‐H, Random, 95% CI) | 0.57 [0.11, 3.12] |
| 23.3 Ondansetron oral 8 mg versus 16 mg | 3 | 875 | Risk Ratio (M‐H, Random, 95% CI) | 1.20 [1.05, 1.37] |
| 23.4 Ondansetron intravenous 10 µcg/kg versus 50 µcg/kg | 1 | 48 | Risk Ratio (M‐H, Random, 95% CI) | 2.83 [0.97, 8.27] |
| 23.5 Ondansetron intravenous 40 µcg/kg versus 100 µcg/kg | 1 | 42 | Risk Ratio (M‐H, Random, 95% CI) | 0.91 [0.63, 1.30] |
| 23.6 Ondansetron intravenous 50 µcg/kg versus 100 µcg/kg | 2 | 189 | Risk Ratio (M‐H, Random, 95% CI) | 0.98 [0.50, 1.90] |
| 23.7 Ondansetron intravenous 50 µcg/kg versus 150 µcg/kg | 1 | 239 | Risk Ratio (M‐H, Random, 95% CI) | 1.46 [1.08, 1.97] |
| 23.8 Ondansetron intravenous 75 µcg/kg versus 150 µcg/kg | 1 | 142 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.51, 1.94] |
| 23.9 Ondansetron intravenous 100 µcg/kg versus 150 µcg/kg | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.28, 3.59] |
| 23.10 Ondansetron intravenous 100 µcg/kg versus 200 µcg/kg | 1 | 36 | Risk Ratio (M‐H, Random, 95% CI) | 1.44 [0.90, 2.30] |
| 23.11 Ondansetron intravenous 1 mg versus 4 mg | 1 | 400 | Risk Ratio (M‐H, Random, 95% CI) | 1.36 [1.02, 1.82] |
| 23.12 Ondansetron intravenous 1 mg versus 8 mg | 1 | 345 | Risk Ratio (M‐H, Random, 95% CI) | 1.28 [1.07, 1.53] |
| 23.13 Ondansetron intravenous 2 mg versus 4 mg | 1 | 36 | Risk Ratio (M‐H, Random, 95% CI) | 2.68 [0.31, 23.43] |
| 23.14 Ondansetron intravenous 4 mg versus 8 mg | 7 | 790 | Risk Ratio (M‐H, Random, 95% CI) | 1.17 [0.71, 1.92] |
| 23.15 Ondansetron intravenous 8 mg versus 16 mg | 1 | 343 | Risk Ratio (M‐H, Random, 95% CI) | 0.92 [0.76, 1.11] |
| 24 Vomiting ramosetron | 3 | 226 | Risk Ratio (M‐H, Random, 95% CI) | 2.12 [1.05, 4.27] |
| 24.1 Ramosetron intravenous 0.15 mg versus 0.3 mg | 3 | 113 | Risk Ratio (M‐H, Random, 95% CI) | 2.20 [0.91, 5.29] |
| 24.2 Ramosetron intravenous 0.3 mg versus 0.6 mg | 3 | 113 | Risk Ratio (M‐H, Random, 95% CI) | 1.99 [0.62, 6.38] |
| 25 Vomiting tropisetron | 3 | 332 | Risk Ratio (M‐H, Random, 95% CI) | 1.38 [0.57, 3.34] |
| 25.1 Tropisetron intravenous 2 mg versus 5 mg | 3 | 332 | Risk Ratio (M‐H, Random, 95% CI) | 1.38 [0.57, 3.34] |
| 26 Nausea or Vomiting clonidine | 3 | 116 | Risk Ratio (M‐H, Random, 95% CI) | 3.41 [1.34, 8.71] |
| 26.1 Clonidine oral 2 µcg/kg versus 5 µcg/kg | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 4.0 [1.01, 15.81] |
| 26.2 Clonidine epidural 25 µcg/hr versus 50 µcg/hr | 1 | 50 | Risk Ratio (M‐H, Random, 95% CI) | 8.00 [1.08, 59.32] |
| 26.3 Clonidine epidural 1 µcg/kg versus 3 µcg/kg | 1 | 36 | Risk Ratio (M‐H, Random, 95% CI) | 1.5 [0.28, 7.93] |
| 27 Nausea or Vomiting dexamethasone | 6 | 733 | Risk Ratio (M‐H, Random, 95% CI) | 1.44 [1.10, 1.90] |
| 27.1 Dexamethasone intravenous 1.25 mg versus 2.5 mg | 1 | 66 | Risk Ratio (M‐H, Random, 95% CI) | 1.5 [0.70, 3.24] |
| 27.2 Dexamethasone intravenous 2 mg versus 4 mg | 1 | 45 | Risk Ratio (M‐H, Random, 95% CI) | 1.25 [0.61, 2.56] |
| 27.3 Dexamethasone intravenous 2.5 mg versus 5 mg | 3 | 173 | Risk Ratio (M‐H, Random, 95% CI) | 1.62 [0.92, 2.83] |
| 27.4 Dexamethasone intravenous 4 mg versus 8 mg | 2 | 75 | Risk Ratio (M‐H, Random, 95% CI) | 2.47 [1.01, 6.03] |
| 27.5 Dexamethasone intravenous 5 mg versus 8 mg | 1 | 88 | Risk Ratio (M‐H, Random, 95% CI) | 1.77 [0.78, 4.02] |
| 27.6 Dexamethasone intravenous 5 mg versus 10 mg | 3 | 196 | Risk Ratio (M‐H, Random, 95% CI) | 1.06 [0.58, 1.93] |
| 27.7 Dexamethasone intravenous 8 mg versus 16 mg | 2 | 90 | Risk Ratio (M‐H, Random, 95% CI) | 1.12 [0.41, 3.03] |
| 28 Nausea or Vomiting dolasetron | 3 | 1188 | Risk Ratio (M‐H, Random, 95% CI) | 1.06 [0.96, 1.19] |
| 28.1 Dolasetron oral 25 mg versus 50 mg | 2 | 355 | Risk Ratio (M‐H, Random, 95% CI) | 1.06 [0.91, 1.24] |
| 28.2 Dolasetron oral 50 mg versus 100 mg | 2 | 234 | Risk Ratio (M‐H, Random, 95% CI) | 1.05 [0.82, 1.34] |
| 28.3 Dolasetron oral 100 mg versus 200 mg | 2 | 343 | Risk Ratio (M‐H, Random, 95% CI) | 0.97 [0.82, 1.14] |
| 28.4 Dolasetron intravenous 25 mg versus 50 mg | 1 | 256 | Risk Ratio (M‐H, Random, 95% CI) | 1.41 [1.09, 1.82] |
| 29 Nausea or Vomiting domperidone | 2 | 132 | Risk Ratio (M‐H, Random, 95% CI) | 0.98 [0.74, 1.31] |
| 29.1 Domperidone intravenous 5 mg versus 10 mg | 1 | 72 | Risk Ratio (M‐H, Random, 95% CI) | 0.90 [0.60, 1.37] |
| 29.2 Domperidone intramuscular 10 mg versus 15 mg | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 1.06 [0.71, 1.57] |
| 30 Nausea or Vomiting droperidol | 13 | 2272 | Risk Ratio (M‐H, Random, 95% CI) | 1.20 [1.08, 1.33] |
| 30.1 Droperidol intravenous 5 µcg/kg versus 10 µcg/kg | 1 | 32 | Risk Ratio (M‐H, Random, 95% CI) | 2.00 [0.42, 9.42] |
| 30.2 Droperidol intravenous 10 µcg/kg versus 20 µcg/kg | 2 | 79 | Risk Ratio (M‐H, Random, 95% CI) | 2.02 [1.01, 4.03] |
| 30.3 Droperidol intravenous 20 µcg/kg versus 30 µcg/kg | 1 | 39 | Risk Ratio (M‐H, Random, 95% CI) | 0.91 [0.40, 2.07] |
| 30.4 Droperidol intravenous 0.25 mg versus 1.25 mg | 1 | 116 | Risk Ratio (M‐H, Random, 95% CI) | 1.10 [0.52, 2.30] |
| 30.5 Droperidol intravenous 0.5 mg versus 1 mg | 1 | 75 | Risk Ratio (M‐H, Random, 95% CI) | 2.86 [0.98, 8.31] |
| 30.6 Droperidol intravenous 0.625 mg versus 1.25 mg | 3 | 1126 | Risk Ratio (M‐H, Random, 95% CI) | 0.98 [0.69, 1.41] |
| 30.7 Droperidol intravenous 0.75 mg versus 1.25 mg | 1 | 94 | Risk Ratio (M‐H, Random, 95% CI) | 0.40 [0.08, 1.86] |
| 30.8 Droperidol intravenous 1.25 mg versus 2.5 mg | 3 | 201 | Risk Ratio (M‐H, Random, 95% CI) | 1.59 [0.96, 2.65] |
| 30.9 Droperidol intravenous 2.5 mg versus 5 mg | 1 | 192 | Risk Ratio (M‐H, Random, 95% CI) | 1.5 [0.71, 3.17] |
| 30.10 Droperidol intravenous PCA bolus 5 µcg versus 15 µcg | 1 | 123 | Risk Ratio (M‐H, Random, 95% CI) | 1.36 [0.84, 2.20] |
| 30.11 Droperidol intravenous PCA bolus 15 µcg versus 50 µcg | 1 | 124 | Risk Ratio (M‐H, Random, 95% CI) | 1.35 [0.77, 2.37] |
| 30.12 Droperidol intravenous PCA bolus 50 µcg versus 100 µcg | 1 | 26 | Risk Ratio (M‐H, Random, 95% CI) | 1.06 [0.52, 2.15] |
| 30.13 Droperidol intravenous PCA bolus 100 µcg versus 150 µcg | 1 | 19 | Risk Ratio (M‐H, Random, 95% CI) | 1.85 [0.61, 5.63] |
| 30.14 Droperidol intravenous PCA bolus 150 µcg versus 200 µcg | 1 | 26 | Risk Ratio (M‐H, Random, 95% CI) | 0.8 [0.26, 2.50] |
| 31 Nausea or Vomiting granisetron | 10 | 1132 | Risk Ratio (M‐H, Random, 95% CI) | 1.50 [1.19, 1.89] |
| 31.1 Granisetron oral 1 mg versus 2 mg | 3 | 128 | Risk Ratio (M‐H, Random, 95% CI) | 2.42 [1.17, 5.01] |
| 31.2 Granisetron oral 2 mg versus 4 mg | 3 | 128 | Risk Ratio (M‐H, Random, 95% CI) | 1.26 [0.53, 3.00] |
| 31.3 Granisetron intravenous 20 µcg/kg versus 30 µcg/kg | 1 | 45 | Risk Ratio (M‐H, Random, 95% CI) | 3.0 [0.77, 11.72] |
| 31.4 Granisetron intravenous 20 µcg/kg versus 40 µcg/kg | 5 | 196 | Risk Ratio (M‐H, Random, 95% CI) | 2.80 [1.63, 4.79] |
| 31.5 Granisetron intravenous 30 µcg/kg versus 40 µcg/kg | 1 | 45 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.21, 4.86] |
| 31.6 Granisetron intravenous 40 µcg/kg versus 60 µcg/kg | 1 | 38 | Risk Ratio (M‐H, Random, 95% CI) | 1.92 [0.30, 12.13] |
| 31.7 Granisetron intravenous 40 µcg/kg versus 80 µcg/kg | 2 | 75 | Risk Ratio (M‐H, Random, 95% CI) | 1.25 [0.46, 3.43] |
| 31.8 Granisetron intravenous 40 µcg/kg versus 100 µcg/kg | 2 | 83 | Risk Ratio (M‐H, Random, 95% CI) | 1.41 [0.49, 4.04] |
| 31.9 Granisetron intravenous 100 µcg versus 1 mg | 1 | 199 | Risk Ratio (M‐H, Random, 95% CI) | 1.45 [1.12, 1.87] |
| 31.10 Granisetron intravenous 1 mg versus 3 mg | 1 | 195 | Risk Ratio (M‐H, Random, 95% CI) | 0.88 [0.66, 1.16] |
| 32 Nausea or Vomiting neostigmine | 4 | 190 | Risk Ratio (M‐H, Random, 95% CI) | 0.66 [0.47, 0.91] |
| 32.1 Neostigmine intrathecal 50 µcg versus 100 µcg | 1 | 40 | Risk Ratio (M‐H, Random, 95% CI) | 0.8 [0.40, 1.60] |
| 32.2 Neostigmine intrathecal 100 µcg versus 200 µcg | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 0.58 [0.32, 1.06] |
| 32.3 Neostigmine intrathecal 200 µcg versus 500 µcg | 1 | 20 | Risk Ratio (M‐H, Random, 95% CI) | 0.25 [0.07, 0.90] |
| 32.4 Neostigmine epidural 10 µcg/kg versus 20 µcg/kg | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 0.67 [0.18, 2.42] |
| 32.5 Neostigmine epidural 20 µcg/kg versus 30 µcg/kg | 1 | 20 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.26, 3.81] |
| 32.6 Neostigmine epidural 30 µcg/kg versus 40 µcg/kg | 1 | 20 | Risk Ratio (M‐H, Random, 95% CI) | 0.6 [0.19, 1.86] |
| 32.7 Neostigmine epidural 40 µcg/kg versus 50 µcg/kg | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 0.77 [0.38, 1.55] |
| 33 Nausea or Vomiting ondansetron | 8 | 556 | Risk Ratio (M‐H, Random, 95% CI) | 1.39 [1.08, 1.79] |
| 33.1 Ondansetron intavenous 25 µcg/kg versus 50 µcg/kg | 1 | 45 | Risk Ratio (M‐H, Random, 95% CI) | 1.64 [0.92, 2.92] |
| 33.2 Ondansetron intavenous 50 µcg/kg versus 75 µcg/kg | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 1.4 [0.57, 3.43] |
| 33.3 Ondansetron intavenous 50 µcg/kg versus 100 µcg/kg | 1 | 40 | Risk Ratio (M‐H, Random, 95% CI) | 1.6 [0.63, 4.05] |
| 33.4 Ondansetron intavenous 75 µcg/kg versus 100 µcg/kg | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.36, 2.75] |
| 33.5 Ondansetron intavenous 100 µcg/kg versus 150 µcg/kg | 1 | 45 | Risk Ratio (M‐H, Random, 95% CI) | 1.43 [0.54, 3.75] |
| 33.6 Ondansetron intravenous 2 mg versus 4 mg | 1 | 36 | Risk Ratio (M‐H, Random, 95% CI) | 1.57 [0.55, 4.43] |
| 33.7 Ondansetron intravenous 4 mg versus 8 mg | 5 | 330 | Risk Ratio (M‐H, Random, 95% CI) | 1.31 [0.85, 2.02] |
| 34 Nausea or Vomiting ramosetron | 3 | 226 | Risk Ratio (M‐H, Random, 95% CI) | 2.20 [1.23, 3.92] |
| 34.1 Ramosetron intravenous 0.15 mg versus 0.3 mg | 3 | 113 | Risk Ratio (M‐H, Random, 95% CI) | 2.98 [1.43, 6.23] |
| 34.2 Ramosetron intravenous 0.3 mg versus 0.6 mg | 3 | 113 | Risk Ratio (M‐H, Random, 95% CI) | 1.34 [0.53, 3.42] |
| 35 Rescue antiemetic clonidine | 2 | 110 | Risk Ratio (M‐H, Random, 95% CI) | 1.18 [0.73, 1.92] |
| 35.1 Clonidine intrathecal 25 µcg/kg versus 75 µcg/kg | 1 | 40 | Risk Ratio (M‐H, Random, 95% CI) | 1.08 [0.67, 1.75] |
| 35.2 Clonidine epidural 2 µcg/ml versus 3 µcg/ml | 1 | 36 | Risk Ratio (M‐H, Random, 95% CI) | 0.89 [0.74, 1.08] |
| 35.3 Clonidine epidural 3 µcg/ml versus 4 µcg/ml | 1 | 34 | Risk Ratio (M‐H, Random, 95% CI) | 1.77 [1.20, 2.61] |
| 36 Rescue antiemetic dexamethasone | 6 | 763 | Risk Ratio (M‐H, Random, 95% CI) | 1.48 [1.00, 2.20] |
| 36.1 Dexamethasone intravenous 1.25 mg versus 2.5 mg | 2 | 111 | Risk Ratio (M‐H, Random, 95% CI) | 1.25 [0.44, 3.54] |
| 36.2 Dexamethasone intravenous 2 mg versus 4 mg | 1 | 45 | Risk Ratio (M‐H, Random, 95% CI) | 2.5 [0.32, 19.53] |
| 36.3 Dexamethasone intravenous 2.5 mg versus 5 mg | 4 | 204 | Risk Ratio (M‐H, Random, 95% CI) | 1.49 [0.73, 3.06] |
| 36.4 Dexamethasone intravenous 4 mg versus 8 mg | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 3.0 [0.13, 68.26] |
| 36.5 Dexamethasone intravenous 5 mg versus 8 mg | 1 | 88 | Risk Ratio (M‐H, Random, 95% CI) | 1.77 [0.78, 4.02] |
| 36.6 Dexamethasone intravenous 5 mg versus 10 mg | 4 | 240 | Risk Ratio (M‐H, Random, 95% CI) | 1.29 [0.59, 2.82] |
| 36.7 Dexamethasone intravenous 8 mg versus 16 mg | 1 | 45 | Risk Ratio (M‐H, Random, 95% CI) | 0.65 [0.03, 14.97] |
| 37 Rescue antiemetic dolasetron | 5 | 2114 | Risk Ratio (M‐H, Random, 95% CI) | 1.05 [0.92, 1.19] |
| 37.1 Dolasetron oral 25 mg versus 50 mg | 2 | 354 | Risk Ratio (M‐H, Random, 95% CI) | 0.97 [0.63, 1.48] |
| 37.2 Dolasetron oral 50 mg versus 100 mg | 2 | 234 | Risk Ratio (M‐H, Random, 95% CI) | 1.10 [0.77, 1.57] |
| 37.3 Dolasetron oral 100 mg versus 200 mg | 2 | 343 | Risk Ratio (M‐H, Random, 95% CI) | 0.95 [0.71, 1.28] |
| 37.4 Dolasetron intravenous 12.5 mg versus 25 mg | 2 | 410 | Risk Ratio (M‐H, Random, 95% CI) | 0.92 [0.65, 1.30] |
| 37.5 Dolasetron intravenous 25 mg versus 50 mg | 2 | 462 | Risk Ratio (M‐H, Random, 95% CI) | 1.28 [0.93, 1.76] |
| 37.6 Dolasetron intravenous 50 mg versus 100 mg | 1 | 311 | Risk Ratio (M‐H, Random, 95% CI) | 1.19 [0.78, 1.82] |
| 38 Rescue antiemetic droperidol | 12 | 1862 | Risk Ratio (M‐H, Random, 95% CI) | 1.21 [1.02, 1.44] |
| 38.1 Droperidol oral 50 µcg/kg versus 75 µcg/kg | 1 | 43 | Risk Ratio (M‐H, Random, 95% CI) | 1.05 [0.30, 3.66] |
| 38.2 Droperidol intravenous 5 µcg/kg versus 10 µcg/kg | 1 | 32 | Risk Ratio (M‐H, Random, 95% CI) | 2.0 [0.20, 19.91] |
| 38.3 Droperidol intravenous 10 µcg/kg versus 20 µcg/kg | 1 | 134 | Risk Ratio (M‐H, Random, 95% CI) | 1.33 [0.56, 3.15] |
| 38.4 Droperidol intravenous 25 µcg/kg versus 50 µcg/kg | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.29 [0.01, 6.91] |
| 38.5 Droperidol intravenous 0.25 mg versus 0.5 mg | 1 | 95 | Risk Ratio (M‐H, Random, 95% CI) | 0.98 [0.26, 3.69] |
| 38.6 Droperidol intravenous 0.625 mg versus 1.25 mg | 4 | 1223 | Risk Ratio (M‐H, Random, 95% CI) | 1.20 [0.99, 1.45] |
| 38.7 Droperidol intravenous 0.75 mg versus 1.25 mg | 1 | 94 | Risk Ratio (M‐H, Random, 95% CI) | 0.59 [0.06, 6.32] |
| 38.8 Droperidol intravenous 1.25 mg versus 2.5 mg | 1 | 70 | Risk Ratio (M‐H, Random, 95% CI) | 3.83 [0.37, 40.16] |
| 38.9 Droperidol intravenous PCA bolus 17 µcg versus 33 µcg | 1 | 40 | Risk Ratio (M‐H, Random, 95% CI) | 2.0 [0.41, 9.71] |
| 38.10 Droperidol intravenous PCA bolus 50 µcg versus 100 µcg | 1 | 26 | Risk Ratio (M‐H, Random, 95% CI) | 1.41 [0.49, 4.04] |
| 38.11 Droperidol intravenous PCA bolus 100 µcg versus 150 µcg | 1 | 19 | Risk Ratio (M‐H, Random, 95% CI) | 1.67 [0.36, 7.82] |
| 38.12 Droperidol intravenous PCA bolus 150 µcg versus 200 µcg | 1 | 26 | Risk Ratio (M‐H, Random, 95% CI) | 0.8 [0.18, 3.59] |
| 39 Rescue antiemetic granisetron | 16 | 1569 | Risk Ratio (M‐H, Random, 95% CI) | 1.66 [1.15, 2.40] |
| 39.1 Granisetron oral 20 µcg/kg versus 40 µcg/kg | 2 | 93 | Risk Ratio (M‐H, Random, 95% CI) | 1.09 [0.06, 18.65] |
| 39.2 Granisetron oral 40 µcg/kg versus 80 µcg/kg | 1 | 45 | Risk Ratio (M‐H, Random, 95% CI) | 2.0 [0.13, 29.81] |
| 39.3 Granisetron oral 1 mg versus 2 mg | 2 | 83 | Risk Ratio (M‐H, Random, 95% CI) | 6.79 [0.93, 49.51] |
| 39.4 Granisetron oral 2 mg versus 4 mg | 2 | 83 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 39.5 Granisetron intravenous 2 µcg/kg versus 5 µcg/kg | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 2.0 [0.77, 5.20] |
| 39.6 Granisetron intravenous 5 µcg/kg versus 10 µcg/kg | 1 | 40 | Risk Ratio (M‐H, Random, 95% CI) | 1.33 [0.34, 5.21] |
| 39.7 Granisetron intravenous 10 µcg/kg versus 20 µcg/kg | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 1.5 [0.37, 6.07] |
| 39.8 Granisetron intravenous 10 µcg/kg versus 40 µcg/kg | 1 | 66 | Risk Ratio (M‐H, Random, 95% CI) | 4.0 [0.47, 33.91] |
| 39.9 Granisetron intravenous 20 µcg/kg versus 30 µcg/kg | 1 | 45 | Risk Ratio (M‐H, Random, 95% CI) | 4.65 [0.27, 81.01] |
| 39.10 Granisetron intravenous 20 µcg/kg versus 40 µcg/kg | 8 | 386 | Risk Ratio (M‐H, Random, 95% CI) | 2.86 [1.20, 6.81] |
| 39.11 Granisetron intravenous 30 µcg/kg versus 40 µcg/kg | 1 | 45 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 39.12 Granisetron intravenous 40 µcg/kg versus 60 µcg/kg | 1 | 38 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 39.13 Granisetron intravenous 40 µcg/kg versus 80 µcg/kg | 3 | 135 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 39.14 Granisetron intravenous 40 µcg/kg versus 100 µcg/kg | 3 | 128 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 39.15 Granisetron intravenous 1 mg versus 3 mg | 1 | 262 | Risk Ratio (M‐H, Random, 95% CI) | 1.09 [0.70, 1.68] |
| 40 Rescue antiemetic neostigmine | 2 | 166 | Risk Ratio (M‐H, Random, 95% CI) | 0.87 [0.59, 1.29] |
| 40.1 Neostigmine intrathecal 25 µcg/kg versus 50 µcg/kg | 1 | 33 | Risk Ratio (M‐H, Random, 95% CI) | 0.86 [0.48, 1.54] |
| 40.2 Neostigmine intrathecal 50 µcg/kg versus 75 µcg/kg | 1 | 33 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.58, 1.73] |
| 40.3 Neostigmine epidural 10 µcg/kg versus 20 µcg/kg | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 40.4 Neostigmine epidural 20 µcg/kg versus 30 µcg/kg | 1 | 20 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 40.5 Neostigmine epidural 30 µcg/kg versus 40 µcg/kg | 1 | 20 | Risk Ratio (M‐H, Random, 95% CI) | 0.33 [0.02, 7.32] |
| 40.6 Neostigmine epidural 40 µcg/kg versus 50 µcg/kg | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 0.29 [0.04, 2.01] |
| 41 Rescue antiemetic ondansetron | 10 | 754 | Risk Ratio (M‐H, Random, 95% CI) | 1.22 [0.90, 1.65] |
| 41.1 Ondansetron intravenous 10 µcg/kg versus 50 µcg/kg | 1 | 48 | Risk Ratio (M‐H, Random, 95% CI) | 3.61 [0.20, 65.86] |
| 41.2 Ondansetron intavenous 25 µcg/kg versus 50 µcg/kg | 1 | 45 | Risk Ratio (M‐H, Random, 95% CI) | 3.5 [0.91, 13.44] |
| 41.3 Ondansetron intravenous 40 µcg/kg versus 100 µcg/kg | 1 | 49 | Risk Ratio (M‐H, Random, 95% CI) | 2.13 [0.26, 17.54] |
| 41.4 Ondansetron intavenous 50 µcg/kg versus 75 µcg/kg | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.16, 6.20] |
| 41.5 Ondansetron intravenous 50 µcg/kg versus 100 µcg/kg | 2 | 89 | Risk Ratio (M‐H, Random, 95% CI) | 7.0 [0.38, 127.32] |
| 41.6 Ondansetron intavenous 75 µcg/kg versus 100 µcg/kg | 1 | 30 | Risk Ratio (M‐H, Random, 95% CI) | 2.0 [0.20, 19.78] |
| 41.7 Ondansetron intravenous 100 µcg/kg versus 200 µcg/kg | 1 | 49 | Risk Ratio (M‐H, Random, 95% CI) | 5.5 [0.24, 128.18] |
| 41.8 Ondansetron intavenous 100 µcg/kg versus 150 µcg/kg | 1 | 45 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.10, 10.17] |
| 41.9 Ondansetron intravenous 0.5 mg versus 1 mg | 1 | 38 | Risk Ratio (M‐H, Random, 95% CI) | 1.46 [0.67, 3.15] |
| 41.10 Ondansetron intravenous 1 mg versus 2 mg | 1 | 26 | Risk Ratio (M‐H, Random, 95% CI) | 1.25 [0.43, 3.63] |
| 41.11 Ondansetron intravenous 2 mg versus 4 mg | 2 | 62 | Risk Ratio (M‐H, Random, 95% CI) | 1.17 [0.41, 3.36] |
| 41.12 Ondansetron intravenous 4 mg versus 8 mg | 4 | 205 | Risk Ratio (M‐H, Random, 95% CI) | 1.08 [0.55, 2.13] |
| 41.13 Ondansetron intravenous 8 mg versus 16 mg | 1 | 38 | Risk Ratio (M‐H, Random, 95% CI) | 0.70 [0.28, 1.77] |
| 42 Rescue antiemetic ramosetron | 2 | 166 | Risk Ratio (M‐H, Random, 95% CI) | 8.77 [0.54, 142.51] |
| 42.1 Ramosetron intravenous 0.15 mg versus 0.3 mg | 2 | 83 | Risk Ratio (M‐H, Random, 95% CI) | 8.77 [0.54, 142.51] |
| 42.2 Ramosetron intravenous 0.3 mg versus 0.6 mg | 2 | 83 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 43 Rescue antiemetic tropisetron | 3 | 331 | Risk Ratio (M‐H, Random, 95% CI) | 1.15 [0.44, 2.99] |
| 43.1 Tropisetron intravenous 2 mg versus 5 mg | 3 | 331 | Risk Ratio (M‐H, Random, 95% CI) | 1.15 [0.44, 2.99] |
Analysis 12.1.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 1 Nausea alizapride.
Analysis 12.2.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 2 Nausea clonidine.
Analysis 12.3.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 3 Nausea dexamethasone.
Analysis 12.4.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 4 Nausea dolasetron.
Analysis 12.5.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 5 Nausea domperidone.
Analysis 12.6.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 6 Nausea droperidol.
Analysis 12.7.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 7 Nausea ginger.
Analysis 12.8.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 8 Nausea granisetron.
Analysis 12.9.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 9 Nausea neostigmine.
Analysis 12.10.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 10 Nausea ondansetron.
Analysis 12.11.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 11 Nausea ramosetron.
Analysis 12.12.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 12 Nausea tropisetron.
Analysis 12.13.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 13 Vomiting alizapride.
Analysis 12.14.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 14 Vomiting clonidine.
Analysis 12.15.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 15 Vomiting dexamethasone.
Analysis 12.16.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 16 Vomiting dolasetron.
Analysis 12.17.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 17 Vomiting domperidone.
Analysis 12.18.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 18 Vomiting droperidol.
Analysis 12.19.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 19 Vomiting ginger.
Analysis 12.20.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 20 Vomiting granisetron.
Analysis 12.21.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 21 Vomiting metoclopramide.
Analysis 12.22.
Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 22 Vomiting neostigmine.
Analysis 12.23.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 23 Vomiting ondansetron.
Analysis 12.24.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 24 Vomiting ramosetron.
Analysis 12.25.
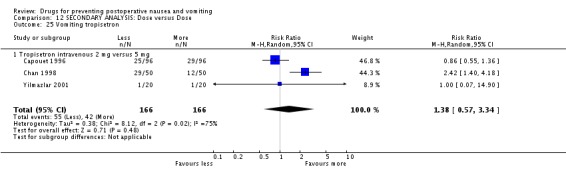
Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 25 Vomiting tropisetron.
Analysis 12.26.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 26 Nausea or Vomiting clonidine.
Analysis 12.27.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 27 Nausea or Vomiting dexamethasone.
Analysis 12.28.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 28 Nausea or Vomiting dolasetron.
Analysis 12.29.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 29 Nausea or Vomiting domperidone.
Analysis 12.30.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 30 Nausea or Vomiting droperidol.
Analysis 12.31.
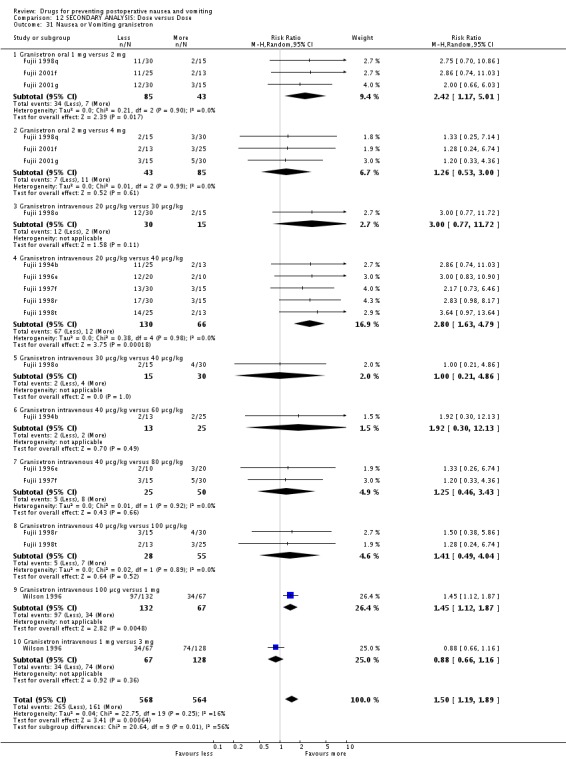
Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 31 Nausea or Vomiting granisetron.
Analysis 12.32.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 32 Nausea or Vomiting neostigmine.
Analysis 12.33.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 33 Nausea or Vomiting ondansetron.
Analysis 12.34.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 34 Nausea or Vomiting ramosetron.
Analysis 12.35.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 35 Rescue antiemetic clonidine.
Analysis 12.36.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 36 Rescue antiemetic dexamethasone.
Analysis 12.37.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 37 Rescue antiemetic dolasetron.
Analysis 12.38.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 38 Rescue antiemetic droperidol.
Analysis 12.39.
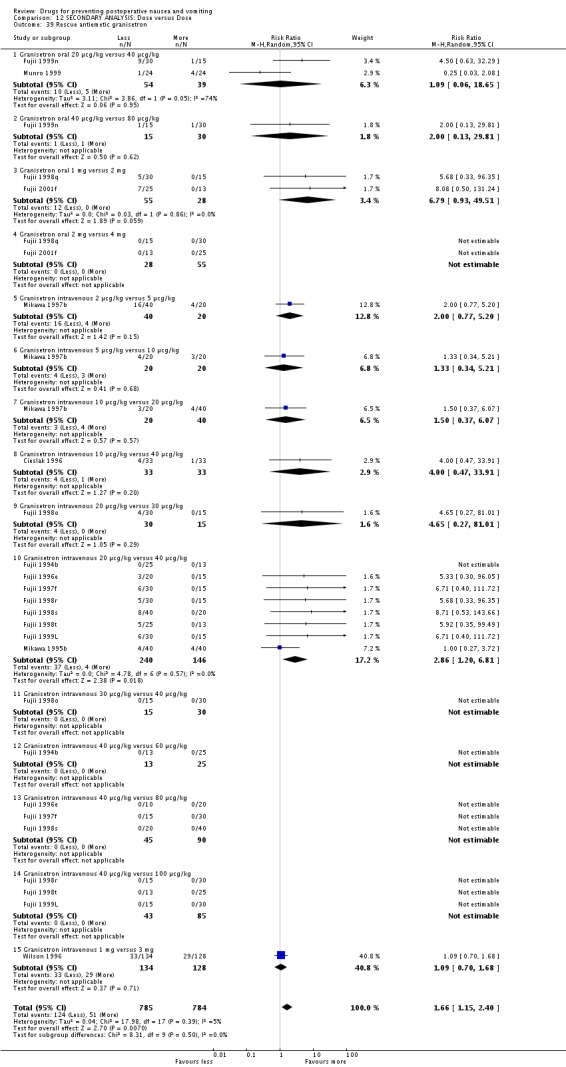
Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 39 Rescue antiemetic granisetron.
Analysis 12.40.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 40 Rescue antiemetic neostigmine.
Analysis 12.41.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 41 Rescue antiemetic ondansetron.
Analysis 12.42.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 42 Rescue antiemetic ramosetron.
Analysis 12.43.

Comparison 12 SECONDARY ANALYSIS: Dose versus Dose, Outcome 43 Rescue antiemetic tropisetron.
Comparison 13.
POSTHOC ANALYSIS: Fujii et al versus other authors
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Nausea: granisetron | 37 | 2950 | Risk Ratio (M‐H, Random, 95% CI) | 0.53 [0.45, 0.63] |
| 1.1 Fujii | 28 | 1859 | Risk Ratio (M‐H, Random, 95% CI) | 0.42 [0.34, 0.53] |
| 1.2 Others | 9 | 1091 | Risk Ratio (M‐H, Random, 95% CI) | 0.67 [0.55, 0.81] |
| 2 Vomiting: granisetron | 52 | 4088 | Risk Ratio (M‐H, Random, 95% CI) | 0.40 [0.35, 0.46] |
| 2.1 Fujii | 39 | 2719 | Risk Ratio (M‐H, Random, 95% CI) | 0.38 [0.33, 0.44] |
| 2.2 Others | 13 | 1369 | Risk Ratio (M‐H, Random, 95% CI) | 0.42 [0.33, 0.54] |
| 3 Nausea or Vomiting: granisetron | 34 | 2652 | Risk Ratio (M‐H, Random, 95% CI) | 0.39 [0.31, 0.48] |
| 3.1 Fujii | 27 | 1908 | Risk Ratio (M‐H, Random, 95% CI) | 0.41 [0.36, 0.47] |
| 3.2 Others | 7 | 744 | Risk Ratio (M‐H, Random, 95% CI) | 0.53 [0.35, 0.80] |
| 4 Rescue antiemetic: granisetron | 42 | 3410 | Risk Ratio (M‐H, Random, 95% CI) | 0.29 [0.22, 0.39] |
| 4.1 Fujii | 33 | 2413 | Risk Ratio (M‐H, Random, 95% CI) | 0.23 [0.17, 0.30] |
| 4.2 Others | 9 | 997 | Risk Ratio (M‐H, Random, 95% CI) | 0.48 [0.34, 0.69] |
| 5 Nausea: droperidol versus granisetron | 19 | 782 | Risk Ratio (M‐H, Random, 95% CI) | 1.36 [1.05, 1.77] |
| 5.1 Fujii | 16 | 612 | Risk Ratio (M‐H, Random, 95% CI) | 2.33 [1.54, 3.52] |
| 5.2 Others | 3 | 170 | Risk Ratio (M‐H, Random, 95% CI) | 0.94 [0.67, 1.33] |
| 6 Vomiting: droperidol versus granisetron | 24 | 1008 | Risk Ratio (M‐H, Random, 95% CI) | 2.16 [1.71, 2.72] |
| 6.1 Fujii | 21 | 838 | Risk Ratio (M‐H, Random, 95% CI) | 2.42 [1.82, 3.22] |
| 6.2 Others | 3 | 170 | Risk Ratio (M‐H, Random, 95% CI) | 1.70 [1.14, 2.55] |
| 7 Nausea or Vomiting: droperidol versus granisetron | 18 | 744 | Risk Ratio (M‐H, Random, 95% CI) | 2.08 [1.55, 2.80] |
| 7.1 Fujii | 15 | 574 | Risk Ratio (M‐H, Random, 95% CI) | 2.43 [1.84, 3.22] |
| 7.2 Others | 3 | 170 | Risk Ratio (M‐H, Random, 95% CI) | 1.23 [0.61, 2.48] |
| 8 Rescue antiemetic: droperidol versus granisetron | 19 | 850 | Risk Ratio (M‐H, Random, 95% CI) | 2.77 [1.82, 4.21] |
| 8.1 Fujii | 17 | 700 | Risk Ratio (M‐H, Random, 95% CI) | 5.10 [2.75, 9.44] |
| 8.2 Others | 2 | 150 | Risk Ratio (M‐H, Random, 95% CI) | 1.63 [0.91, 2.89] |
| 9 Side effects | 97 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 9.1 Constipation: Placebo versus Dexamethasone; Fujii | 1 | 50 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.07, 15.12] |
| 9.2 Constipation: Placebo versus Dexamethasone; Not Fujii | 1 | 125 | Risk Ratio (M‐H, Random, 95% CI) | 0.41 [0.17, 1.02] |
| 9.3 Constipation: Placebo versus Granisetron; Fujii | 4 | 330 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.37, 2.68] |
| 9.4 Constipation: Placebo versus Granisetron; Not Fujii | 2 | 113 | Risk Ratio (M‐H, Random, 95% CI) | 1.18 [0.21, 6.74] |
| 9.5 Dizziness: Placebo versus Dexamethasone; Fujii | 7 | 576 | Risk Ratio (M‐H, Random, 95% CI) | 1.00 [0.50, 2.03] |
| 9.6 Dizziness: Placebo versus Dexamethasone; Not Fujii | 9 | 685 | Risk Ratio (M‐H, Random, 95% CI) | 1.06 [0.77, 1.47] |
| 9.7 Dizziness: Placebo versus Droperidol; Fujii | 8 | 308 | Risk Ratio (M‐H, Random, 95% CI) | 1.01 [0.40, 2.54] |
| 9.8 Dizziness: Placebo versus Droperidol; Not Fujii | 11 | 1749 | Risk Ratio (M‐H, Random, 95% CI) | 0.98 [0.73, 1.31] |
| 9.9 Dizziness: Placebo versus Granisetron; Fujii | 19 | 1232 | Risk Ratio (M‐H, Random, 95% CI) | 0.76 [0.47, 1.25] |
| 9.10 Dizziness: Placebo versus Granisetron; Not Fujii | 1 | 39 | Risk Ratio (M‐H, Random, 95% CI) | 1.17 [0.08, 17.35] |
| 9.11 Dizziness: Placebo versus Metoclopramide; Fujii | 3 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.22, 4.56] |
| 9.12 Dizziness: Placebo versus Metoclopramide; Not Fujii | 7 | 614 | Risk Ratio (M‐H, Random, 95% CI) | 0.81 [0.41, 1.60] |
| 9.13 Headache: Placebo versus Dexamethasone; Fujii | 9 | 686 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.61, 1.65] |
| 9.14 Headache: Placebo versus Dexamethasone; Not Fujii | 13 | 901 | Risk Ratio (M‐H, Random, 95% CI) | 0.77 [0.55, 1.08] |
| 9.15 Headache: Placebo versus Droperidol; Fujii | 10 | 408 | Risk Ratio (M‐H, Random, 95% CI) | 0.96 [0.49, 1.87] |
| 9.16 Headache: Placebo versus Droperidol; Not Fujii | 14 | 1872 | Risk Ratio (M‐H, Random, 95% CI) | 0.79 [0.62, 1.00] |
| 9.17 Headache: Placebo versus Granisetron; Fujii | 27 | 1788 | Risk Ratio (M‐H, Random, 95% CI) | 0.99 [0.72, 1.36] |
| 9.18 Headache: Placebo versus Granisetron; Not Fujii | 2 | 112 | Risk Ratio (M‐H, Random, 95% CI) | 0.66 [0.22, 1.91] |
| 9.19 Headache: Placebo versus Metoclopramide; Fujii | 4 | 84 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.27, 3.74] |
| 9.20 Headache: Placebo versus Metoclopramide; Not Fujii | 8 | 566 | Risk Ratio (M‐H, Random, 95% CI) | 1.01 [0.51, 2.00] |
| 9.21 Itch: Placebo versus Dexamethasone; Fujii | 1 | 120 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.15, 6.87] |
| 9.22 Itch: Placebo versus Dexamethasone; Not Fujii | 3 | 144 | Risk Ratio (M‐H, Random, 95% CI) | 0.99 [0.76, 1.28] |
| 9.23 Sedation: Placebo versus Dexamethasone; Fujii | 5 | 275 | Risk Ratio (M‐H, Random, 95% CI) | 0.81 [0.31, 2.13] |
| 9.24 Sedation: Placebo versus Dexamethasone; Not Fujii | 1 | 36 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 9.25 Sedation: Placebo versus Droperidol; Fujii | 8 | 338 | Risk Ratio (M‐H, Random, 95% CI) | 1.17 [0.51, 2.64] |
| 9.26 Sedation: Placebo versus Droperidol; Not Fujii | 16 | 1882 | Risk Ratio (M‐H, Random, 95% CI) | 1.32 [1.14, 1.53] |
| 9.27 Sedation: Placebo versus Metoclopramide; Fujii | 3 | 64 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.22, 4.59] |
| 9.28 Sedation: Placebo versus Metoclopramide; Not Fujii | 9 | 397 | Risk Ratio (M‐H, Random, 95% CI) | 1.12 [0.74, 1.71] |
| 9.29 Dizziness: Droperidol versus Granisetron; Fujii | 10 | 398 | Risk Ratio (M‐H, Random, 95% CI) | 1.00 [0.48, 2.09] |
| 9.30 Dizziness: Droperidol versus Granisetron; Not Fujii | 1 | 134 | Risk Ratio (M‐H, Random, 95% CI) | 0.72 [0.34, 1.54] |
| 9.31 Dizziness: Droperidol versus Metoclopramide; Fujii | 9 | 310 | Risk Ratio (M‐H, Random, 95% CI) | 1.19 [0.52, 2.69] |
| 9.32 Dizziness: Droperidol versus Metoclopramide; Not Fujii | 1 | 36 | Risk Ratio (M‐H, Random, 95% CI) | 3.0 [0.13, 69.09] |
| 9.33 Dizziness: Granisetron versus Ramosetron; Fujii | 3 | 300 | Risk Ratio (M‐H, Random, 95% CI) | 1.0 [0.30, 3.38] |
| 9.34 Dizziness: Granisetron versus Ramosetron; Not Fujii | 1 | 36 | Risk Ratio (M‐H, Random, 95% CI) | 0.33 [0.04, 2.91] |
| 9.35 Headache: Droperidol versus Granisetron; Fujii | 11 | 428 | Risk Ratio (M‐H, Random, 95% CI) | 0.91 [0.48, 1.73] |
| 9.36 Headache: Droperidol versus Granisetron; Not Fujii | 1 | 134 | Risk Ratio (M‐H, Random, 95% CI) | 0.58 [0.32, 1.07] |
| 9.37 Headache: Droperidol versus Metoclopramide; Fujii | 10 | 340 | Risk Ratio (M‐H, Random, 95% CI) | 1.44 [0.68, 3.04] |
| 9.38 Headache: Droperidol versus Metoclopramide; Not Fujii | 2 | 83 | Risk Ratio (M‐H, Random, 95% CI) | 1.00 [0.31, 3.29] |
| 9.39 Headache: Granisetron versus Ramosetron; Fujii | 3 | 300 | Risk Ratio (M‐H, Random, 95% CI) | 1.25 [0.51, 3.09] |
| 9.40 Headache: Granisetron versus Ramosetron; Not Fujii | 1 | 36 | Risk Ratio (M‐H, Random, 95% CI) | 0.71 [0.28, 1.84] |
| 9.41 Sedation: Droperidol versus Metoclopramide; Fujii | 4 | 130 | Risk Ratio (M‐H, Random, 95% CI) | 1.48 [0.43, 5.05] |
| 9.42 Sedation: Droperidol versus Metoclopramide; Not Fujii | 4 | 204 | Risk Ratio (M‐H, Random, 95% CI) | 1.26 [0.88, 1.80] |
Analysis 13.2.

Comparison 13 POSTHOC ANALYSIS: Fujii et al versus other authors, Outcome 2 Vomiting: granisetron.
Analysis 13.3.

Comparison 13 POSTHOC ANALYSIS: Fujii et al versus other authors, Outcome 3 Nausea or Vomiting: granisetron.
Analysis 13.6.

Comparison 13 POSTHOC ANALYSIS: Fujii et al versus other authors, Outcome 6 Vomiting: droperidol versus granisetron.
Analysis 13.7.
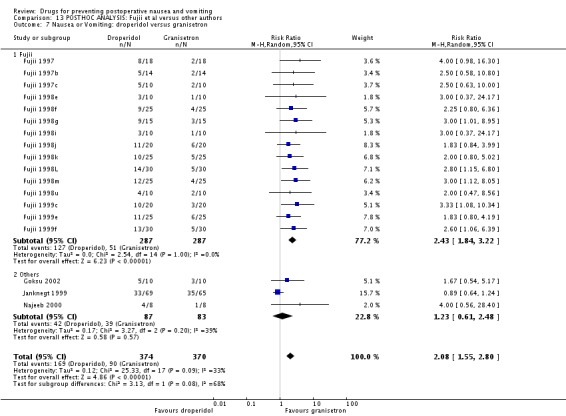
Comparison 13 POSTHOC ANALYSIS: Fujii et al versus other authors, Outcome 7 Nausea or Vomiting: droperidol versus granisetron.
What's new
Last assessed as up‐to‐date: 14 May 2004.
| Date | Event | Description |
|---|---|---|
| 7 December 2016 | Amended | The future of this review is being considered. This is because it includes a large number of retracted studies. |
History
Protocol first published: Issue 2, 2003 Review first published: Issue 3, 2006
| Date | Event | Description |
|---|---|---|
| 4 March 2014 | Amended | Feedback submitted |
| 20 June 2008 | Amended | Converted to new review format. |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
| Methods | Y; B; Y; Y; N; N | |
| Participants | 80 adults; 63 women; orthopaedic/general; >18; ASA1‐3; exc' study drug contraindication, pregnant | |
| Interventions | Induction Either: PLACEBO; or BETAMETHASONE 12mg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐4; 5‐24 hours. | |
| Notes | Male/female incidences not reported. Nausea and vomiting commonest 5‐24 hours, rescue antiemetic 0‐4 hours. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; N; Y; Y | |
| Participants | 60 boys; hypospadias; ASA1; 2‐10; exc' study drug allergy, bleeding tendency, CNS/spinal disease | |
| Interventions | Induction Either: PLACEBO +/‐ caudal NEOSTIGMINE 2 mcg/kg | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐2 hours. | |
| Notes | No side effects. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 80 women; breast; ASA1‐3; mean 50; exc lung/cardiac/liver/renal disease, fever, antiemetic, N&V, hypertension, obese, alcoholic | |
| Interventions | Intraoperative Either: PLACEBO; or DEXAMETHASONE 8mg iv; or DOLASETRON 12.5mg iv; or METOCLOPRAMIDE 20mg iv | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | No side effects. Unclear if retchers/vomiters nauseated. Retching categorized separately. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 52 children; no sex data; 2‐13; strabismus; ASA1; exc' drug/infection | |
| Interventions | Intraoperative Either: PLACEBO; or DROPERIDOL 75 to 100 mcg/kg i.v. | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐48 hours. | |
| Notes | Male/female incidences not recorded. No side effects. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; N; Y; Y | |
| Participants | 180 adults; 127 women; lap' chole'; 18‐75 (mean 49); ASA1,2; exc' GI/ NM disease, drug abuse, <18 >75 years, antiemetic | |
| Interventions | Induction Either: PLACEBO; or METOCLOPRAMIDE 10mg iv / ONDANSETRON 8mg iv +/‐ DEXAMETHASONE 8mg iv | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐6; 6‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Unclear if retchers/vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; A; N; N; Y; cY | |
| Participants | 150 adults; 101 women; lap' chole'; ASA1,2; 18‐60 (mean 40); exc' PONV/motion sickness, renal disorder, DM, obese, antiemetic | |
| Interventions | Induction Either: PLACEBO; ONDANSETRON 4mg iv; PLACEBO and P6 acupressure | |
| Outcomes | All outcomes. Postop 0‐6; 6‐24 hours. | |
| Notes | Outcomes commonest 0‐6 hours. Male/female incidences not reported. Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; N; N | |
| Participants | 139 women; gynaecological; exc' pregnant/breastfeeding, antiemetic | |
| Interventions | Intraoperative Either: PLACEBO; or ONDANSETRON 4mg i.v.+/‐ CYCLIZINE 50 mg i.v. | |
| Outcomes | All outcomes. Postop 0‐3; 3‐24; 0‐24 hours. | |
| Notes | Vomiting commonest 0‐3 hours. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 60 adults; no sex data; ASA1,2; thyroidectomy; exc' abnormal blood | |
| Interventions | Induction Either: NO TREATMENT; or PLACEBO; or ONDANSETRON 4mg i.v. | |
| Outcomes | Nausea; vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 45 adults; 24 women; mastoidectomy; ASA1,2; exc' antiemetic, PONV, GI disease, pregnant/menstrual | |
| Interventions | Induction Either: PLACEBO; or DEXAMETHASONE 8mg i.v. AND GRANISETRON 3mg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetic Postop 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 145 adults; 77 women; ASA1,2; orthopaedic; exc' asthma, antiemetic, alcoholic, dopamine drug, renal failure, study drug allergy | |
| Interventions | Intraoperative AND postoperative PCA Either: PLACEBO twice; or DROPERIDOL 1.25mg i.v. then 5mg/60ml; or ONDANSETRON 4mg i.v. then 8mg/60ml | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Unclear how retching categorized or if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; N; N | |
| Participants | 124 adults; 76 women; ASA 1,2; orthopaedic; 16‐80 (mean 56); exc antiemetic, alcoholic, renal failure | |
| Interventions | Preoperative Either: PLACEBO; or METOCLOPRAMIDE 10mg i.v.; or ONDANSETRON 8mg i.v. | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 120 adults; 50 women; 18‐75 (mean 45); ASA1‐3; eyes; exc' fertile, lactating, confounding condition, heart/liver/renal disease, study drug allergy, drug abuse | |
| Interventions | Intraoperative end Either: PLACEBO; or METOCLOPRAMIDE 0.25mg/kg i.v.; or TROPISETRON 0.1mg/kg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Unclear if vomiters nauseated or categorized once or twice. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 58 children; 33 girls; 8‐15 (mean 12); various surgeries; exc' antiemetic | |
| Interventions | Induction Either: PLACEBO i.v.; or TROPISETRON 0.1mg/kg i.v. | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐2; 2‐6; 6‐12; 12‐18; 18‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; N; N; Y; Y | |
| Participants | 30 adults; no sex data; 19‐79 (mean 46); exc' DM, liver/CNS diseases. | |
| Interventions | Intraoperative Either: NO TREATMENT; or DOMPERIDONE 0.2mg/kg i.v. | |
| Outcomes | Vomiting. Postop 0‐4 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 362 women; gynaecological | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 1.25mg i.v.; or HALOPERIDOL 1 or 2 or 5mg i.v.; or METOCLOPRAMIDE 10 or 20mg i.v. | |
| Outcomes | Vomiting. Postop time unclear. | |
| Notes | Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; N; N | |
| Participants | 67 women; STOP; ASA1; 15‐37 (mean 25); exc' renal, blood, liver disease, drug abuse, study drug allergy, antiemetic | |
| Interventions | Preoperative Either: DROPERIDOL 1.25mg i.v.; or METOCLOPRAMIDE 10mg i.v.; or ONDANSETRON 8mg i.v. | |
| Outcomes | All outcomes Postop 0‐3 hours. | |
| Notes | Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 40 women; gynaecological; ASA1,2 | |
| Interventions | Induction Either: ONDANSETRON 4 or 8mg i.v. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 40 women; gynaecological; ASA1,2; exc' antiemetic, allergy, renal/liver/blood disease | |
| Interventions | Preoperative Either: DROPERIDOL 1.25mg i.v; or ONDANSETRON 4mg i.v. | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | D ‐ Not used |
| Methods | N; B; Y; N; N; N | |
| Participants | 84 women; gynaecological; 17‐72 (mean 34); ASA1,2; exc' renal/blood/liver disease, drug abuse/allergy, antiemetic | |
| Interventions | Preoperative Either: PLACEBO; or TROPISETRON 5mg i.v. | |
| Outcomes | All outcomes. Postop 0‐4 ; 4‐24; 0‐24 hours. | |
| Notes | Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 84 adults; 52 women; general; 16‐73 (mean 46); exc' cardiac/lung disease, DM, disorder affecting outcome | |
| Interventions | Preoperative Either: DROPERIDOL 0.2mg/kg (max 10mg) i.m.; or PENTOBARBITONE 2mg/kg i.m. | |
| Outcomes | Nausea or vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. No side effects. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 30 adults; no sex data; urological/plastic; ASA1,2; mean 26 | |
| Interventions | Induction Either: PLACEBO; or intrathecal NEOSTIGMINE 200 or 500 mcg | |
| Outcomes | Nausea or vomiting. Postop 0‐4 hours. | |
| Notes | Male/female incidences not reported. No side effects. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 40 adults; 19 women; thyroidectomy; ASA1,2; 18‐60 (mean 43); exc' GI disease, antiemetic | |
| Interventions | Induction Either: PLACEBO; or DEXAMETHASONE 8mg iv | |
| Outcomes | Nausea; vomiting. Postop immediate; 0‐15 minutes. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear if vomiters nauseated or categorized once or twice. Retching categorized as nausea. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N, B, Y; Y, Y; Y | |
| Participants | 339 children; 92 girls; various surgeries; exc' preference for IM drugs | |
| Interventions | Preoperative Either: Placebo; or Alprazolam 5mcg/kg; or Chloral hydrate 40mg/kg; or Diazepam 0.25mg/kg; or Midazolam 0.3mg/kg | |
| Outcomes | Vomiting. Postop 0‐1; 1‐3 hours. | |
| Notes | Vomiting commonest 1‐3 hours. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; Y; Y; N; N; N | |
| Participants | 48 children; 2‐12 years (mean 6); ASA1,2; tonsil +/‐ adenoids; excluded cardiac/renal/liver disease | |
| Interventions | Intraoperative Either: PLACEBO; or TROPISETRON 0.1mg/kg i.v. | |
| Outcomes | Vomiting. Postop 0‐2;2‐6; 6‐12 ;12‐18; 18‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; N; N | |
| Participants | 110 children; 74 girls; tonsil +/‐ adenoids; ASA1,2; exc' drug affecting outcomes, study contraindication. | |
| Interventions | Induction Either: PLACEBO; or DEXAMETHASONE 0.5 mg/kg (max 8mg) i.v. | |
| Outcomes | Vomiting; rescue antiemetics. Postop 0‐2; 2‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 473 patients; no sex or age data; ENT or eye | |
| Interventions | Intraoperative Either: NO TREATMENT; or DROPERIDOL 50 mcg/kg (route unclear) | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐1; 1‐2; 2‐6; 6‐24; 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; N; N | |
| Participants | 1180 children & adults; excluded <20% incidence of PV, antiemetic | |
| Interventions | Intraoperative Either: PLACEBO; or DIMENHYDRINATE 62.5 mg i.v. (1.25mg/kg); or DROPERIDOL 2.5mg i.v. (50 mcg/kg); or METOCLOPRAMIDE 50 mg/kg i.v.; or TROPISETRON 2.5mg i.v. (50 mcg/kg) | |
| Outcomes | Vomiting. Postop 0‐24 hours. | |
| Notes | Side effects not recorded. Nausea and 'nausea or vomiting' reported for adults. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; N; Y; N | |
| Participants | 5199 adults, PONV risk > 0.40; six interventions; 4086 analysed; 3279 women; mean 47; various surgeries | |
| Interventions | Intraoperative 20 minutes after start. Either: DEXAMETHASONE 4mg i.v.; or DROPERIDOL 1.25mg i.v.; or NO TREATMENT. Intraoperative 20 minutes before end. Either; ONDANSETRON 4mg i.v.; or NO TREATMENT. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐2; 2‐24; 0‐24 hours. | |
| Notes | Male/female incidences reported. Side effects not reported. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 80 children; 35 girls; tonsil & adenoids; 3‐15 (mean 7); exc' ulcer, DM, chronic disease, steroid | |
| Interventions | Intraoperative Either: PLACEBO; DEXAMETHASONE 1mg/kg (max 16mg) i.v. | |
| Outcomes | Vomiting. Postop 0‐6 hours. | |
| Notes | Male/female incidences not reported. No side effects. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; N; N | |
| Participants | 60 adults; 7 women; various surgeries; mean 62; exc' pregnant, ASA>3, unable to use PCA, drug allergy/abuse, epilepsy, antiemetic, antidepressant | |
| Interventions | Postoperative infusion Either: PLACEBO; or ONDANSETRON 1mg/ml iv | |
| Outcomes | See notes. | |
| Notes | Incidence not reported. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; N; N | |
| Participants | 108 women; gynaecological; ASA1,2; 18‐75 (mean 32); exc' study drug allergy, pregnant/lactating, dislike spicy food, antiemetic | |
| Interventions | Preoperative Either: PLACEBO; or oral GINGER 500mg or 1g | |
| Outcomes | Postop 0‐24 hours. | |
| Notes | Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; Y; Y; Y | |
| Participants | 87 adults; 66 women; lap' chole'; mean 44; ASA1,2; exc' PONV/motion sickness, GI disease, antiemetic | |
| Interventions | Induction Either PLACEBO; or ONDANSETRON 4mg iv; or TROPISETRON 5mg iv | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Immediate; 3; 6; 12; 0‐12 hours. | |
| Notes | Nausea commonest 3 hours. Unclear if vomiters nauseated or vomiters categorized once or twice. Male/female incidences not reported. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; N; N; Y; Y | |
| Participants | 89 adults; various surgeries | |
| Interventions | Intraoperative Either: PLACEBO i.v.; or DROPERIDOL 0.625mg i.v. | |
| Outcomes | Nausea or vomiting. PACU; 24 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 251 children & adults; 122 female; 5‐89 (mean 69); ASA1,2; cataracts; exc' confounding conditions | |
| Interventions | Induction Either: PLACEBO; or METOCLOPRAMIDE 10mg i.v.; or ONDANSETRON 4mg i.v. | |
| Outcomes | Nausea; vomiting, Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects: "no differences". Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; N; N | |
| Participants | 64 adults; 50 women; lap' chole' 18‐80 (mean 50); ASA1,2; exc' motion sickness/PONV, antiemetic, GI disease, steroid | |
| Interventions | Induction Either: DROPERIDOL 1.25mg iv +/‐ ONDANSETRON 4mg iv | |
| Outcomes | All outcomes. Postop 0‐1; 1‐3; 3‐24; 0‐24 hours. | |
| Notes | Nausea or vomiting only 0‐24 hours. Other outcomes commonest 3‐24 hours. Male/female incidences not reported. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; N; N. | |
| Participants | ?52 or 101 children; no sex data; eye; no exclusion | |
| Interventions | Induction Either: PLACEBO; or METOCLOPRAMIDE 0.25mg/kg i.v.; or ONDANSETRON 0.15mg/kg i.v. | |
| Outcomes | See notes. Postop 0‐4; 0‐24 hours. | |
| Notes | No group numbers. Percentage (not incidence) of retching/vomiting. Male/female incidences not recorded. Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; N; N | |
| Participants | 50 women; thyroid; 18‐45 (mean 33); exc' irregular cycle, oestrogen/progestagen, PONV/motion sickness | |
| Interventions | Preoperative Either: PLACEBO; or ONDANSETRON 8mg i.v. | |
| Outcomes | Nausea or vomiting. Postop (time unclear). | |
| Notes | Unclear how retching categorized. Unclear if nauseated vomiters categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 68 adults; 45 women; lap' chole'; >18 (mean 54); ASA1‐3 | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 1mg i.v.; or ONDANSETRON 4mg i.v. | |
| Outcomes | Nausea; vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 300; ASA 1 or 2; general | |
| Interventions | Preoperative Either: PLACEBO; or DROPERIDOL 1mg i.v.; or ONDANSETRON 4mg i.v. | |
| Outcomes | Nausea; vomiting. Postop 4; 8; 12; 24 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 42 women; gynaecogical; ASA1,2; mean 44; exc' opioid, antiemetic, antihistamine, benzodiazepine | |
| Interventions | Induction Either: PLACEBO;or epidural DROPERIDOL 2.5mg | |
| Outcomes | Nausea; vomiting. Postop 0‐24 hours. | |
| Notes | Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 60 women; hysterectomy; ASA1,2 | |
| Interventions | Induction AND postoperative PCA Either: PLACEBO twice; or DROPERIDOL 1.25mg i.v. then PLACEBO; or DROPERIDOL 1.25mg i.v. then 0.1mg/ml | |
| Outcomes | Postop 0‐24 hours. | |
| Notes | Unclear how retching categorized or if nauseated vomiters categorized once or twice. No side effects. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; Y; Y; N; N; N | |
| Participants | 92 children; 43 girls; ASA1,2; 1‐18 (mean 7); tonsil +/‐ adenoids; exc' study drug allergy, motion sickness | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 0.1 mg/kg (max 4mg) i.v. | |
| Outcomes | Vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; N; N; Y; Y | |
| Participants | 120 boys; hypospadia; ASA1; 2‐8 (mean 6); exc' bleeding tendency, spinal/CNS disease | |
| Interventions | Intraoperative Either: NO TREATMENT; or caudal NEOSTIGMINE 10 or 20 or 30 or 40 or 50 mcg/kg. | |
| Outcomes | Nausea or vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | No side effects. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; N | |
| Participants | 50 adults; 30 women; ASA1,2; dental extraction; 18‐45 (mean 23) | |
| Interventions | Preoperative Either: PLACEBO; or oral DEXAMETHASONE 8mg | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Only incidence of vomiting clear. Male/female incidences not reported. No side effects. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; Y; Y | |
| Participants | 100 women; lap' steri'; ASA1,2; exc' oral contraceptive, antiemetic, unclear or no menses | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 10 or 20 or 30 mcg/kg i.v. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐6; 6‐24; 0‐24 hours. | |
| Notes | Side effects not recorded. Categorized by 'worst' symptom. Retching categorized with vomiting. Unclear if vomiters retched or nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y. | |
| Participants | 40 adults; abdominal; exc' >75, opioid abuse, heart/renal/hepatic/lung failure, high BP, postop' ventilation | |
| Interventions | Preoperative AND postoperative Either: PLACEBO twice; or oral CLONIDINE 300 mcg/kg twice | |
| Outcomes | Postop 0‐24 hours. | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y. | |
| Participants | 120 children; 63 girls; strabismus; 1‐15 (mean 6); ASA1,2; exc' motion sickness/PONV | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 25 mcg/kg iv; or ONDANSETRON 150 mcg/kg iv | |
| Outcomes | Nausea or vomiting; rescue antiemetic. Postop 0‐2; 2‐6; 6‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. No side effects. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 390 women; gynaecological; mean 51; exc' <18, >100kg, ASA>3, recent antiemetic/N&V, study drug allergy, pregnant | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 8mg i.v. | |
| Outcomes | Nausea; vomiting;rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Side effects not reported. Retching categorized as vomiting. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 60 women; gynaecological; ASA1,2; 21‐42 (mean 33); exc' pregnant, breast feeding, antiemetic, abnormal blood | |
| Interventions | Intraoperative Either: PLACEBO; or ONDANSETRON 8mg i.v.; or TROPISETRON 5mg i.v. | |
| Outcomes | Nausea; vomiting. Postop 0‐10; 10‐20; 20‐30; 30‐60 min; 1‐6; 0‐6 hours. | |
| Notes | Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; N; N | |
| Participants | 88 adults; 50 women; lap' chole'; ASA 1,2; mean 43; excluded >75, ASA>2, pregnant, ERCP, chronic pain, disease, opioid/tranquilizer, drug abuse | |
| Interventions | Preoperative Either: PLACEBO; or DEXAMETHASONE 8mg iv | |
| Outcomes | All outcomes. Postop 0‐6; 6‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. No side effects. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; Y; Y; Y. | |
| Participants | 120 adults; 96 women; lap' chole'; ASA1,2; 21‐55 (mean 42); exc' GI/NM disease, motion sickness/PONV, pregnant, menstrual, smoked, DM, antiemetic | |
| Interventions | Induction Either: GRANISETRON 50 mcg/kg +/‐ DEXAMETHASONE 8mg iv | |
| Outcomes | All outcomes. Postop 0‐4; 4‐24; 0‐24 hours. | |
| Notes | Nausea and rescue antiemetic commonest 0‐4 hours, vomiting 4‐24 hours. Male/female incidences not reported. Unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 100 children; 53 girls; strabismus; ASA1 | |
| Interventions | Intraoperative Either: DROPERIDOL 75mcg/kg i.v. and PLACEBO; or PROMETHAZINE 0.5mg/kg i.v. and 0.5mg/kg i.m. (max 25mg) | |
| Outcomes | Vomiting. Postop 0‐6; 6‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; N; N; N | |
| Participants | 80 children; 9 girls; minor surgery; ASA1,2; 3‐8 (mean 5); exc' endocrine disease, MH risk, aortic stenosis, infection, preop' agitation | |
| Interventions | Induction Either: PLACEBO twice; or caudal CLONIDINE 1 or 3 mcg/kg AND PLACEBO; or PLACEBO AND CLONIDINE 3 mcg/kg iv | |
| Outcomes | Nausea or vomiting. Postop 0‐2 hours. | |
| Notes | Male/female incidences not reported. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 80 adults; 38 women; general; ASA1,2 | |
| Interventions | Intraoperative Either: No treatment; or Neostigmine 1.5mg i.v. AND Atropine 0.5mg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postoperative 0‐5; 5‐24; 24‐48; 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 60 women; gynaecological; ASA1,2; 16‐65 (mean 41); exc' antiemetic/opioid | |
| Interventions | Preoperative then induction Either; PLACEBO twice; or oral GINGER 1g then PLACEBO; or PLACEBO then METOCLOPRAMIDE 10mg i.v. | |
| Outcomes | Nausea; nausea or vomiting; rescue antiemetic. PACU; PACU‐4 hours; 4‐12; 12‐24; 0‐24 hours. | |
| Notes | Nausea commonest PACU and 4‐12 hours. No side effects. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 40 women; caesarean | |
| Interventions | Postoperative PCA Either: PLACEBO; or DROPERIDOL PCA 83mcg/ml | |
| Outcomes | See notes. | |
| Notes | Percentage not incidences. Side effects: "droperidol sedated breast‐fed infant". | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 90 women; gynaecological; ASA1,2 | |
| Interventions | Intraoperative Either: PLACEBO; or ALIZAPRIDE 50 or 100 or 200mg i.v | |
| Outcomes | Nausea, vomiting, rescue antiemetics Postop 0‐4 hours | |
| Notes | Side effects not reported. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | Unclear number or sex of children; strabismus; 3‐14 | |
| Interventions | Intraoperative Either: PLACEBO; or ONDANSETRON 5mg/msq | |
| Outcomes | See notes. | |
| Notes | Percentage not incidence of nausea or vomiting. Male/female incidences not recorded. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; Y; N | |
| Participants | 90 adults; 31 women; plastic/orthopaedic; ASA1,2; mean 33 | |
| Interventions | Induction Either: PLACEBO; or plexal NEOSTIGMINE 500 mcg; or s/c | |
| Outcomes | Nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. No side effects. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 106 patient; no other data | |
| Interventions | Postoperative four times Either: PLACEBO; or DOMPERIDONE 30mg i.v. then 10mg i.v. thrice | |
| Outcomes | Nausea or vomiting. Postop 0‐3; 0‐6; 0‐9; 0‐12; 0‐15; 0‐18; 0‐21; 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 60 adults; 53 women; thyroid; mean 50; exc' drug interaction | |
| Interventions | Preoperative Either: PLACEBO; or oral ONDANSETRON 8mg | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐12 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Retching categorized as vomiting. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 60 adults; no sex data; thyroid | |
| Interventions | Preoperative Either: PLACEBO; or oral ONDANSETRON 8mg | |
| Outcomes | See notes. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. No number per group. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; Y; N | |
| Participants | 131 children; 53 girls; strabismus; ASA1,2; 2‐13 (mean 6); exc' antiemetic, renal/liver/metabolic/endocrine disease | |
| Interventions | Intraoperative Either: PLACEBO; or ONDANSETRON 0.04 or 0.1 or 0.2mg/kg i.v. | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐8; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects incidence not reported. Unclear how retching categorized | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; Y; Y; Y. | |
| Participants | 200 children; no sex data; 2‐9; ENT surgery | |
| Interventions | Preoperative Either: oral DIAZEPAM 0.2mg/kg; or oral TRICLOFOS 71mg/kg | |
| Outcomes | Vomiting. Postop period unclear. | |
| Notes | No side effects. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; N; N | |
| Participants | 64 adults; 38 women; orthopaedic | |
| Interventions | Induction Either: PLACEBO; or METOCLOPRAMIDE 10mg i.v. | |
| Outcomes | Nausea. PACU | |
| Notes | Nausea not reported for 0‐24 hours. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 60 women; cholecystectomy; 20‐69 (mean 44); exc' post hoc NG & 'pethidine intolerance' | |
| Interventions | Intraoperative AND postoperative thrice Either: PLACEBO; or METOCLOPRAMIDE 10mg i.m. each time | |
| Outcomes | Vomiting; nausea or vomiting. Postop 0‐6; 6‐18 hours. | |
| Notes | Outcomes commonest 6‐18 hours. Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 120 women; gynaecological; 18‐65; ASA1,2; exc' liver, renal, blood/metabolic disease, obese, pregnant | |
| Interventions | Preoperative Either: NO TREATMENT; or oral DIAZEPAM 10 mg | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop time unclear. | |
| Notes | No incidences (physiology & propofol). | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 126 children; 57 girls; ASA1,2; 2‐18 (mean 7.3); strabismus; exc' N&V risk, disorder affecting outcome | |
| Interventions | Postoperative Either: PLACEBO; or METOCLOPRAMIDE 0.15 mg/kg i.v. | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐8 hours. | |
| Notes | Male/female incidences not reported. No side effects. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 100 children; no sex data; strabismus; ASA1,2; 2‐18 years | |
| Interventions | INDUCTION Either: PLACEBO; or DROPERIDOL 20 or 75 mcg/kg i.v. | |
| Outcomes | Vomiting. Postop 0‐2 hours. | |
| Notes | Male/female incidences not recorded. Side effects "droperidol sedative". Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 242 adults; 193 women; 18‐60; ASA1,2; gynaecological/biliary | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 2.5mg i.v. +/‐ ONDANSETRON 4mg i.v. | |
| Outcomes | See notes. | |
| Notes | Male/female incidences not reported. Side effects 'droperidol sedative'. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; N; Y; Y | |
| Participants | 40 adults; 23 women; lithotripsy; 20‐77 (mean 48); ASA1,2; exc' ASA>3, COPD | |
| Interventions | Preoperative Either: PLACEBO; or DOLASETRON 12.5mg iv | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐1; 1‐2; 0‐2 hours. | |
| Notes | Outcomes commonest 0‐1 hour. Male/female incidences not reported. Side effects not recorded. Unclear how retching was categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; N; N; Y; Y | |
| Participants | 569 boys; general; 2‐12 (mean 5); ASA1,2; exc' sleep apnoea, PONV, antiemetic, benzodiazepine | |
| Interventions | Induction Either: NO TREATMENT; or DEXAMETHASONE 150 mcg/kg i.v. | |
| Outcomes | Vomiting. Postop 0‐24 hours. | |
| Notes | Side effects not recorded. Retching not categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; N; N; Y; Y | |
| Participants | 120 adults; 90 women; rhinoplasty (ENT); ASA1,2; 15‐45 | |
| Interventions | Induction Either: METOCLOPRAMIDE 10mg i.v. +/‐ CIMETIDINE 300mg i.v. | |
| Outcomes | Nausea; vomiting. Postop (time unclear). | |
| Notes | Male/female incidences not reported. No side effects. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 60 women; gynaecological; ASA1 | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 1.5mg i.v. | |
| Outcomes | ||
| Notes | Side effects "no differences". Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 60 children; no sex data; 4‐12 (mean 6); ASA1,2; general; exc' previous GA, motion sickness, antiemetic, obese | |
| Interventions | Induction Either: PLACEBO; or METOCLOPRAMIDE 0.12mg/kg i.v.; or ONDANSETRON 5mg/msq | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐8 hours. | |
| Notes | Male/female incidences not recorded. Side effects: 'no difference'. Retching categorized as nausea. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; N; N. | |
| Participants | 74 adults; ASA1,2; 18‐59 (mean 31); no sex data; various surgeries | |
| Interventions | Induction Either: NO TREATMENT; or DROPERIDOL 0.015 mg/kg i.v. | |
| Outcomes | See notes. | |
| Notes | Percentages not incidences reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; Y | |
| Participants | 160 adults; 84 women; dental | |
| Interventions | Induction Either: PLACEBO; or DEXAMETHASONE 0.15mg/kg (max 12mg); or PENTAZOCINE 0.4mg/kg (max 30mg) i.v. | |
| Outcomes | Vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 32 adults; 15 women; various surgeries; ASA1,2; 18‐70 (mean 36); exc' pregnant, renal/blood/liver disease, obese, drugs affecting outcome, N&V | |
| Interventions | Preoperative Either: PLACEBO; or ONDANSETRON 4mg i.v. | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not reported. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; N; N | |
| Participants | 385 women; gynaecological; ASA1,2; 18‐75 (mean 40); exc' N&V, antiemetic, NG, drug abuse, study drug allergy, pregnant | |
| Interventions | Induction Either: PLACEBO; or TROPISETRON 0.5 or 2 or 5mg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 100 adults; 54 women; ASA1,2; 30‐75 (mean 64); orthopaedic; exc' drugs, contraindication to extradural | |
| Interventions | Intraoperative AND postoperative Either: CLONIDINE 150 mcg then 25 or 50 mcg/hr; or MORPHINE 1mg then 0.1mg/hr; or both (CLONIDINE 150 mcg then MORPHINE) | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Unclear how retching categorized. Unclear if all vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 54 children; 26 girls; ASA1,2; tonsil + adenoid; exc'CNS/liver/renal/cardiac disease, study drug allergy | |
| Interventions | Induction Either: PLACEBO; or GRANISETRON 10 mcg/kg i.v. | |
| Outcomes | Vomiting. Postop 0‐8; 8‐24 hours. | |
| Notes | Vomiting commonest 0‐8 hours. Male/female incidences not reported. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 172 children; 73 girls; strabismus; 3‐10; exc' motion sickness/PONV, antiemetic/N&V, liver/renal/gastric disease | |
| Interventions | Intraoperative AND postoperative every 8 hours Either: DROPERIDOL 0.05mg/kg iv then +/‐ oral DIMENHYDRINATE 1.25mg/kg; or ONDANSETRON 0.1mg/kg iv then +/‐ oral 0.15mg/kg | |
| Outcomes | Nausea; vomiting. In hospital; discharge‐24 hours; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 234 children; 121 girls; strabismus; 2‐12 (mean 6); ASA1,2 | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 0.1mg/kg (max 4mg) i.v. | |
| Outcomes | Vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 90 children; 46 girls; 4‐10 (mean 7); tonsils +/‐ adenoids; ASA1 | |
| Interventions | Induction Either: GRANISETRON 40 mcg/kg i.v.; or DROPERIDOL 50 mcg/kg i.v. AND METOCLOPRAMIDE 0.25mg/kg i.v. | |
| Outcomes | All outcomes Postop 0‐3; 3‐24 hours. | |
| Notes | Outcomes commonest 3‐24 hours. Male/female incidences not reported. Unclear how retching categorized or if vomiters nauseated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 431 women; STOP | |
| Interventions | Preoperative Either: No treatment; or METOCLOPRAMIDE 10mg i.v.; or PROCHLORPERAZINE 6.25 or 12.5mg i.m. | |
| Outcomes | Vomiting. PACU | |
| Notes | Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; Y; N; N | |
| Participants | 150 women; breast; ASA1,2; exc' GI disease, drug abuse, pregnant/menstrual, N&V/antiemetic | |
| Interventions | Induction Either: PLACEBO; or TROPISETRON 2 or 5mg i.v. | |
| Outcomes | All outcomes Postop 0‐2; 2‐6; 6‐12; 12‐18; 18‐24; 0‐24 hours. | |
| Notes | Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; N; N; Y; Y | |
| Participants | 60 adults; 53 women; liver surgery; 20‐94 (mean 43); exc' NG | |
| Interventions | Preoperative AND postoperative Either: PLACEBO twice; or RANITIDINE 50mg i.v. twice. | |
| Outcomes | Nausea; vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 240 women; caesarean; exc' study drug allergy, skin disorder | |
| Interventions | Intraoperative Either: PLACEBO; or NALBUPHINE 4mg iv; or ONDANSETRON 4 or 8mg iv | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐4 hours. [see notes]. | |
| Notes | Unclear how retching categorized. No side effects. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 351 adults; 308 women; laparoscopies; ASA1,2; 19‐75; exc' fertile women, study drug allergy | |
| Interventions | Preoperative Either: oral PLACEBO; or oral POLONASETRON 0.3 or 1 or 3 or 10 or 30 mcg/kg | |
| Outcomes | Nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; N; N | |
| Participants | 50 women; gynaecological; ASA1,2; 18‐60 (mean 40); exc' N/V, opioids/antiemetic, pregnant, reflux, drug abuse | |
| Interventions | Intraoperative Either: METOCLOPRAMIDE 10mg i.v.; or ONDANSETRON 4mg i.v. | |
| Outcomes | All outcomes Postop 0‐1; 1‐4; 4‐12; 12‐24; 0‐4; 0‐24 hours. | |
| Notes | Side effects "sedation same". Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; N; Y; N | |
| Participants | 80 adults; 49 women; orthopaedic; >17 (mean 63); ASA1‐3; exc' antiemetic, study drug allergy, pregnant/breast feeding | |
| Interventions | Introperative Either: PLACEBO AND ONDANSETRON 4mg iv; or PROCHLORPERAZINE 10mg im AND PLACEBO | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐48 hours. | |
| Notes | Side effects not recorded. Retching categorized as vomiting. Unclear if nauseated retchers/vomiters categorized once, twice, thrice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; N; N | |
| Participants | 105 women; laparoscopy; ASA1,2; 19‐56 (mean 38); exc' antiemetic, psychoactive drug, obese, pregnant, ASA>2, N&V | |
| Interventions | Preoperative AND intraoperative twice Either: DOLASETRON 12.5mg i.v. then PLACEBO twice; or PLACEBO then DOLASETRON 12.5mg i.v. then PLACEBO; or PLACEBO twice then DOLASETRON 12.5mg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐4; 0‐24 hours. | |
| Notes | Retching categorized as vomiting. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; N; Y; Y | |
| Participants | 81 women; Caesarean; exc' liver/renal/ psychiatric/CNS disease, preeclampsia | |
| Interventions | Intraoperative AND postoperative Either: NO TREATMENT then PCA; or ONDANSETRON 4mg i.v. then 0.13 mg/ml PCA | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized or if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 120 women; gynaecological | |
| Interventions | Preoperative Either: PLACEBO; or CYCLIZINE 50 mg i.m.; or PERPHENAZINE 2.5mg i.m. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐1; 1‐6 hours. | |
| Notes | Nausea commonest 1‐6 hours, vomiting and 'nausea or vomiting' 0‐1 hour. Unclear how retching categorized or if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; N; N; N | |
| Participants | 93 children; 48 girls; tonsil +/‐ adenoids; 3‐16 (mean 7); ASA1,2; exc' drugs, post hoc bradycardia | |
| Interventions | Postoperative Either: ATROPINE 15 mcg/kg i.v.; or GLYCOPYRROLATE 10 mcg/kg i.v. | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐6; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Retching not categorized as vomiting | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N, B, Y, N, Y, Y | |
| Participants | 121 children; 58 girls; strabismus; 1‐12 (mean 6); exc' anaesthetic reaction | |
| Interventions | Induction Either: Placebo ; or Atropine 15 mcg/kg i.v.; or Glycopyrrolate 7.5 mcg/kg i.v. | |
| Outcomes | Nausea or vomiting; rescue antiemetic. Postoperative 0‐24 hours. | |
| Notes | Male/female incidences not recorded. No side effects reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; N; N | |
| Participants | 180 women; ASA1,2; gynaecological; exc' pregnant, breast feeding, obese, antiemetic | |
| Interventions | Induction Either: PLACEBO; or CYCLIZINE 50 mg i.v.; or ONDANSETRON 4 mg i.v. | |
| Outcomes | All outcomes. Postop 0‐6; 6‐24; 0‐24 hours. | |
| Notes | Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 150 children; no sex data; strabismus; ASA1,2; 2‐15 (mean 5 ) | |
| Interventions | Induction Either: DROPERIDOL 0.075mg/kg i.v.; or LIDOCAINE 1.5mg/kg i.v.; or DROPERIDOL 0.025mg/kg i.v. AND LIDOCAINE 1.5mg/kg i.v. | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐6; 0‐72 hours. | |
| Notes | Male/female incidences not reported. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; N; N | |
| Participants | 80 pregnant women; caesarean; ASA 1,2; exc' obstetric/fetal complications | |
| Interventions | Induction Either: PLACEBO; or intrathecal MORPHINE 100 mcg; or NEOSTIGMINE 25 mcg; or both | |
| Outcomes | Nausea or vomitiing; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 97 children; 47 girls; ASA1,2; 2‐16 (mean 5); various surgeries; exc' antiemetic, study drug allergy' | |
| Interventions | INDUCTION Either: PLACEBO; or GRANISETRON 10 or 40 mcg/kg i.v. | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐2; 2‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; N; N; Y; Y | |
| Participants | 60 women; gynaecological; ASA1,2; 15‐65 (mean 28); exc' liver/renal/blood/metabolic diseases, drug abuse, neuroleptic | |
| Interventions | Preoperative Either: DIAZEPAM 150 mcg/kg i.v.; or MIDAZOLAM 70 mcg/kg i.v. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐3 hours. | |
| Notes | Incidences of vomiting and 'nausea or vomiting' not reported. Side effects not reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 80 adults; 43 women; general; ASA1‐3; exc' NSAID allergy, heart/kidney/liver disease, steroid | |
| Interventions | Induction Either: PLACEBO; or DEXAMETHASONE 4mg i.v. | |
| Outcomes | Nausea Postop 0‐2; 2‐24 hours. | |
| Notes | Nausea commonest 2‐24 hours. Male/female incidences not reported. Side effects not reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 140 adults; 110 women; lap' chole'; ASA1,2; exc' antiemetic, disease, drug abuse, pregnant, obese, study drug allergy | |
| Interventions | Intraoperative Either: DOLASETRON 12.5mg i.v. AND PLACEBO or DEXAMETHASONE 4mg i.v. | |
| Outcomes | Nausea; vomiting. Postop 0‐5; 5‐24 hours. | |
| Notes | Nausea commonest 0‐5 hours, vomiting 5‐24 hours. Male/female incidences and side effects not reported. Unclear if nauseated retching vomiters categorized once, twice or thrice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 44 children; no sex data; strabismus | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 75 mcg/kg i.v.; or ONDANSETRON 0.15mg/kg i.v. | |
| Outcomes | Nausea or vomiting. Postop 0‐3; 3‐24; 0‐24 hours. | |
| Notes | Male/female not recorded. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; N; N | |
| Participants | 124 children; 24 girls; general; ASA1,2; 2‐12 (mean 5); exc' reflux, gastroparesis, motion sickness, prior POV, renal/heart disease, antibiotic | |
| Interventions | Preoperative AND postoperative Either: PLACEBO; or oral CISAPRIDE 0.3mg/kg then PLACEBO; or PLACEBO then oral CISAPRIDE 0.3mg/kg | |
| Outcomes | Vomiting. Postop 0‐4; 4‐24; 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 195 women; gynae/caesarean; exc' renal, liver, CNS, cardiac disease | |
| Interventions | Induction Either: PLACEBO; or DOMPERIDONE 4mg i.v.; or METOCLOPRAMIDE 10mg i.v. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop time unclear. | |
| Notes | Unclear how retching categorized or if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; N; N; Y; Y. | |
| Participants | 30 women; gynaecological; ASA1,2 | |
| Interventions | Intraoperative Either: PLACEBO; or TROPISETRON 5mg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐2; 2‐24; 0‐24 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; N; N | |
| Participants | 405 children; no sex data; various surgeries; ASA1‐3; 0.5‐16 (mean 5); exc' seizures, confounding condition | |
| Interventions | Preoperative Either: oral MIDAZOLAM 0.25 or 0.5 or 1mg/kg | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop (time unclear). | |
| Notes | Male/female incidences not recorded. No side effects. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 60 women; hysterectomy | |
| Interventions | Preoperative twice AND intraoperative Either: PLACEBO; or PLACEBO twice then DROPERIDOL 0.75mg i.v.; or oral RANITIDINE 300mg twice then PLACEBO | |
| Outcomes | Nausea or vomiting; rescue antiemetic. PACU; discharge‐24 hours. | |
| Notes | Nausea or vomiting commonest second period, rescue antiemetic commonest first period. No side effects. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; N; N; N | |
| Participants | 100 adults; 84 women; ASA1,2; abdominal/head & neck; mean 39; exc' drug allergy, phenothiazine, pregnant/breast feeding | |
| Interventions | Intraoperative Either: PLACEBO; or PROCHLORPERAZINE 10mg i.v. | |
| Outcomes | Nausea; vomiting. Postop (pre‐narcotics); (post‐narcotics). | |
| Notes | Outcomes commonest first period. Male/female incidences not reported. Side effects not recorded. Unclear if vomiters nauseated. Retching a subset of vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; Y; N; N | |
| Participants | 340 adults; 174 women; various surgery; ASA1‐3; 18‐80; exc' neurolepsis, spinal opioids, extrapyramidal, butyrophenones allergy, renal disease | |
| Interventions | Postoperative PCA Either: PLACEBO; or DROPERIDOL 5 or 15 or 50 mcg/ml | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; N; N; Y; Y | |
| Participants | 192 women; laparoscopies | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 0.5mg i.v.; or METOCLOPRAMIDE 15mg i.v.; or ONDANSETRON 4mg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetic. 0‐PACU discharge; 0‐24 hours. | |
| Notes | Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 85 adults; 53 women; lap' chole'; ASA1,2 | |
| Interventions | Induction Either: METOCLOPRAMIDE 10mg i.v.; or ONDANSETRON 4mg i.v. | |
| Outcomes | Vomiting. Postop 0‐24 hours. | |
| Notes | Side effects not recorded. Retching not categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 150 adults & children; 71 female; tonsil +/‐ adenoids; ASA1; mean 12; exc' <4, antiemetic | |
| Interventions | Induction Either: PLACEBO; or METOCLOPRAMIDE 0.2 mg/kg i.v.; or ONDANSETRON 0.1 mg/kg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐2; 2‐6 hours. | |
| Notes | Vomiting commonest 0‐2 hours. Male/female incidences not reported. Unclear if retching vomiters categorized once or twice or if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 136 adults; 93 women; thyroid; ASA1‐3; excluded 5HT/droperidol antagonist, antiemetic, heart/thyroid/renal/liver disease | |
| Interventions | Preoperative AND induction Either: oral DOLASETRON 50mg then NO TREATMENT; or NO TREATMENT then PLACEBO/DOLASETRON 12.5mg i.v./DROPERIDOL 1.25mg i.v. | |
| Outcomes | Nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 480 children; no sex data; strabismus | |
| Interventions | Preoperative Either: PLACEBO; or DROPERIDOL 0.22mg/kg i.m. +/‐ PHENOPERIDINE 0.044 mg/kg i.m. | |
| Outcomes | Vomiting. Postop 0‐4; 4‐24 hours. | |
| Notes | Outcomes commonest 4‐24 hours. Male/female incidences not reported. Unclear how retching was categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 40 adults; 20 women; septoplasty; ASA1,2; mean 31 | |
| Interventions | Preoperative Either: PLACEBO; or oral DIAZEPAM 10mg | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; N; N; N | |
| Participants | 70 women; hysterectomy; ASA1,2; excluded study drug allergy | |
| Interventions | Induction AND postoperative Either: PLACEBO; or ONDANSETRON 4mg i.v. twice | |
| Outcomes | Nausea; vomiting. Postop 0‐24; 0‐120 hours. | |
| Notes | Side effects not recorded. Retching categorized as vomiting. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; N; N | |
| Participants | 102 children; no sex data; dental; ASA1,2; 2‐8 (mean 4) | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 75 mcg/kg i.v.; or ONDANSETRON 100 mcg/kg i.v. | |
| Outcomes | Vomiting. Postop 0‐2; 2‐24; 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 213 children & adults; 84 female; strabismus; ASA1,2; mean 12 | |
| Interventions | Induction Either: DROPERIDOL 75 mcg/kg i.v.; or ONDANSETRON 75 or 150 mcg/kg i.v. | |
| Outcomes | Vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. No side effects. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; Y; Y; Y | |
| Participants | 175 women; 18‐80 (mean 41); excluded pregnant, N&V, antiemetic, postop' ETT/NG/ICU | |
| Interventions | Preoperative Either: PLACEBO; or ONDANSETRON 0.5. or 1 or 2 or 4 or 8 or 16 mg i.v. | |
| Outcomes | Rescue antiemetic. Postop 0‐6; 0‐24 hours. | |
| Notes | Side effects not reported. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 206 adults; 115 women; orthopaedic/gynaecological | |
| Interventions | Preoperative Either: PLACEBO; or oral METOCLOPRAMIDE 20mg | |
| Outcomes | Nausea or vomiting. Postop 0‐1; 1‐3; 3‐6 hours. | |
| Notes | Nausea or vomiting commonest 1‐3 hours. Unclear how retching categorized | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 90 women; laparoscopies; ASA1,2; excluded pregnant | |
| Interventions | Induction Either: PLACEBO; or METOCLOPRAMIDE 20 or 50mg i.v. | |
| Outcomes | Vomiting. Postop time unclear. | |
| Notes | Side effects not recorded. Unclear how retching categorized or if vomiters nauseated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 281 women; gynae; ASA1,2; 18‐55 (mean 37); excluded pregnant/breast feeding/menstrual, liver/renal/cardiac/metabolic disease, cardiac drug, bowel obstruction, antiemetic, NG, drug abuse | |
| Interventions | Intraoperative Either: PLACEBO; or DOLASETRON 12.5 or 25 or 50 or 100mg i.v. | |
| Outcomes | Vomiting. Postop 0‐2; 0‐6; 0‐12; 0‐24 hours. | |
| Notes | Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 793 women; gynae; ASA1‐3; 18‐60 (mean 43); excluded pregnant, liver/renal/endocrine/cardiac disease, antiemetic, post hoc NG | |
| Interventions | Preoperative 1‐2 hours Either: PLACEBO; or oral DOLASETRON 25 or 50 or 100 or 200mg | |
| Outcomes | Nausea; nausea or vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 98 children; 41 girls; tonsil +/‐ adenoids; ASA1,2; exc' N&V, study drug allergy | |
| Interventions | Induction Either: PLACEBO; or TROPISETRON 0.1mg/kg (max 2mg) i.v. | |
| Outcomes | Vomiting. Postop 0‐24 hours. | |
| Notes | Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 60 women; lap' steri'; ASA1,2; exc' analgesic/antiemetic, study drug allergy, lung disease | |
| Interventions | Intraoperative Either: NO TREATMENT; or NEOSTIGMINE 2.5mg i.v. AND GLYCOPYRROLATE 0.5mg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐5; 5‐24 hours. | |
| Notes | Outcomes commonest 0‐5 hours. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 284 adults; 159 women; GI surgery | |
| Interventions | Intraoperative Either: PLACEBO; or METOCLOPRAMIDE 20mg i.v.; or TRIMETHOBENZAMIDE 300mg i.v. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; N; N | |
| Participants | 138 women; 16 to 70; gynaecological; exc' <16 >70, premed' preference | |
| Interventions | Preoperative Either: oral LORAZEPAM 2.5mg; or PROMETHAZINE 50mg | |
| Outcomes | Vomiting. Postop 1 hour. | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 45 adults; 19 women; strabismus; ASA1‐3; 15‐65; excluded obese, liver disease, seizure, difficult airway, antiemetic/opioid, study drug allergy | |
| Interventions | Induction Either: DROPERIDOL 1.25mg i.v.; or ONDANSETRON 4mg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐1; 1‐5; 5‐24 hours. | |
| Notes | Nausea commonest 1‐5 hours, vomiting 5‐24 hours. Male/female incidences not reported. Retching categorized as vomiting. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 1094 adults; regional/GA; ASA1‐4; no sex data; no exclusion data | |
| Interventions | Preoperative twice Either: NO TREATMENT; or NO TREATMENT then CIMETIDINE 5mg/kg/RANITIDINE 1.25mg/kg i.v.; or oral RANITIDINE 300mg then NO TREATMENT | |
| Outcomes | Nausea; vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 40 children; 27 girls; general; 6‐14 (mean 11); exc' antemetics | |
| Interventions | Induction Either: PLACEBO; or HYOSCINE 140 mcg, 5 microgram/hr (patch) | |
| Outcomes | All outcomes Postop 0‐72 hours. | |
| Notes | Male/female incidences not reported. Unclear how retching categorized or if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 60 women; hysterectomy; ASA1,2; exc' study drug allergy, PONV | |
| Interventions | Intraoperative AND postoperative Either: DROPERIDOL 1.25mg i.v. then PCA 0.1mg/ml ; or ONDANSETRON 4 mg i.v. then PCA 1mg/ml | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 4; 8; 12; 24; 0‐24 hours. | |
| Notes | Nausea commonest at 8 hours. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 300 women; ASA1,2; excluded pregnant/breast feeding, heart/kidney/liver disease, postop NG | |
| Interventions | Postoperative Either:PLACEBO; or CLEBOPRIDE 2mg i.m. | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐12 hours. | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 1300 women; gynaecological | |
| Interventions | Preoperative Either: PLACEBO; or CYCLIZINE 50mg i.m.; or PERPHENAZINE 2.5 or 5mg i.m. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐6 hours. | |
| Notes | Summary of publications. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; N; N | |
| Participants | 243 women; gynaecological; exc' pregnant, ASA>3, N&V/antiemetic, cardiac/lung/renal/liver/CNS/disease | |
| Interventions | Preoperative AND postoperative (twice) Either: PLACEBO; or oral ONDANSETRON 8mg thrice | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Nausea or vomiting incidences may be adjusted. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 304 adults; 183 women; general/gynaecological; 18‐76 (mean 48); exc' N&V, antiemetic, ASA>3, NYHA>2, prostatism, glaucoma | |
| Interventions | Preoperative Either: PLACEBO; or SCOPOLAMINE patch | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐2; 2‐6; 6‐10; 10‐24; 24‐48; 0‐48 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; Y; N; N | |
| Participants | 150 women; various surgeries; ASA1,2; 18‐70 (mean 45); exc' antiemetic/N&V | |
| Interventions | Induction AND postoperative suppository thrice Either: PLACEBO; or DIMENHYDRINATE 62mg i.v. then 150mg thrice | |
| Outcomes | Nausea or vomiting; rescue antiemetic. Postop 0‐48 hours. | |
| Notes | Side effects: "dry mouth with dimenhydramine". Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; Y; Y; Y. | |
| Participants | 140 men; ENT; ASA1,2; exc' antiemetic/N&V | |
| Interventions | Induction AND Postoperative Either: PLACEBO; or DIMENHYDRINATE 1mg/kg i.v. twice; or DROPERIDOL 15 mcg/kg i.v. then PLACEBO; or DIMENHYDRINATE 1mg/kg i.v. AND DROPERIDOL 15 mcg/kg i.v. twice. | |
| Outcomes | All outcomes Postop 0‐24 hours. | |
| Notes | Unclear if vomiters nauseated or if nauseated vomiters categorized once or twice. Retching was categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; A; Y; N; N; N | |
| Participants | 120 women; ENT; ASA1,2; 18‐69 | |
| Interventions | Intraoperative AND postoperative Either: PLACEBO; or DIMENHYDRINATE 1mg/kg i.v. twice; or METOCLOPRAMIDE 0.3mg/kg i.v. twice; or DIMENHYDRINATE 1mg/kg i.v. AND METOCLOPRAMIDE 0.3mg/kg i.v. twice | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Side effects "not different ". Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; A; Y; N; Y; Y | |
| Participants | 148 adults; cataracts; ASA1‐3; 52‐93 | |
| Interventions | Intraoperative Either: PLACEBO; or DOLASETRON 12.5mg i.v.; or DROPERIDOL 10 mcg/kg i.v.; or both | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Unclear how retching categorized | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; A; Y; N; N; N | |
| Participants | 160 adults; 102 women; thyroid/lap' chole'; exc' mood‐altering drugs, antiemetic, psychiatric disease/cancer, N&V | |
| Interventions | Induction Either: 5 to 7.5 mg i.v. of DROPERIDOL or MIDAZOLAM | |
| Outcomes | Vomiting; nausea or vomiting; rescue antiemetic. Postop 0‐2; 2‐6; 6‐10; 10‐24; 24‐48; 0‐48 hours. | |
| Notes | Male/female incidences not reported. Side effects "impaired mood". Retching categorized as vomiting. Unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; N; Y; Y | |
| Participants | 160 men; ENT; ASA1,2; exc' study drug allergy, antiemetic/N&V | |
| Interventions | Induction AND postoperative Either: PLACEBO; or DIMENHYDRINATE 1mg/kg i.v. twice; or METOCLOPRAMIDE 0.3mg/kg i.v. twice; or both twice | |
| Outcomes | All outcomes. Postop 0‐6 hours. | |
| Notes | Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; N; Y; Y | |
| Participants | 100 adults; oral surgery | |
| Interventions | PREOPERATIVE Either: CLONIDINE 1.5 mcg/kg i.v.; or MIDAZOLAM 0.05mg/kg i.v. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | High risk | C ‐ Inadequate |
| Methods | N; A; Y; Y; N; N | |
| Participants | 240 adults; ENT; ASA1,2 | |
| Interventions | Induction AND postoperative Either: PLACEBO; or DIMENHYDRINATE 1mg/kg i.v. twice.; or DROPERIDOL 15 mcg/kg i.v. twice; or both twice | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects "no difference". Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; Y; N; N | |
| Participants | 184 women; gynaecological; mean 37; ASA1‐3; exc' BMI>35, incomprehension, antiemetics | |
| Interventions | Preoperative AND postoperative twice Either: PLACEBO; or oral GINGER 100 or 200mg thrice | |
| Outcomes | All outcomes. Postop 0‐3; 0‐24 hours. | |
| Notes | Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 40 adults; 18 women; craniotomy; ASA 2,3; 20‐70 (mean 55); exc' ASA>3, antiemetic, study drug allergy, pregnant/breast feeding, psychiatric | |
| Interventions | Intraoperative Either: PLACEBO; or GRANISETRON 3mg iv | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐30 mins; 0‐1; 0‐4; 0‐8; 0‐12; 0‐24; 0‐48 hours. | |
| Notes | Male/female incidences not reported. Unclear if vomiters nauseated or if nauseated vomiters categorized once or twice. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 75 adults; 53 women; lap' chole'; exc' reflux | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 4mg i.v. +/‐ TENOXICAM 20mg i.v. | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Retching not categorized as vomiting. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 180 adults; 129 women; lap' chole'; mean 42; exc' opioid/NSAID | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 4mg iv +/‐ DEXAMETHASONE (2 or 4 or 8 or 16mg i.v.) | |
| Outcomes | All outcomes. Postop 0‐6; 6‐12; 12‐24 hours. | |
| Notes | Outcomes commonest 12‐24 hours. Male/female incidences not reported. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; N; N | |
| Participants | 120 children; 49 girls; tonsil +/‐ adenoids; 4‐11 (mean 5); exc' antiemetic/steroid/antihistamine/psychoactive | |
| Interventions | Induction Either: PLACEBO; or DEXAMETHASONE 0.5mg/kg iv (max 8mg iv) | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐3; 3‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; N; N; N | |
| Participants | 50 women; laparoscopies; ASA1,2; mean 32; exc' antiemetic, DM, GI disease, fertility | |
| Interventions | Preoperative Either: PLACEBO; or oral METOCLOPRAMIDE 30mg | |
| Outcomes | Nausea; vomiting; rescue antiemetic. PACU; discharge‐6; 6‐24; 0‐24 hours. | |
| Notes | Side effects not recorded. Unclear how retching treated or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; Y; Y; Y | |
| Participants | 44 women; gynaecological; 20‐57 years; ASA1 | |
| Interventions | Induction Either: PLACEBO; or METOCLOPRAMIDE 20mg i.v. | |
| Outcomes | Vomiting. Postop (time unclear). | |
| Notes | Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 100 children; 48 girls; strabismus; ASA1; mean 7; exc' antiemetic/PONV | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 0.075mg/kg i.v.; or METOCLOPRAMIDE 0.1mg/kg i.v.; or ONDANSETRON 0.1mg/kg i.v. | |
| Outcomes | Nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects "droperidol sedative". Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 90 women; laparoscopy; ASA1,2; exc' pregnant, tubal ligations, obese, antiemetic | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 4mg i.v. | |
| Outcomes | All outcomes. Postop 0‐3; 3‐8; 8‐24 hours. | |
| Notes | Rescue antiemetic only 8‐24 hours. Nausea and 'nausea or vomiting' commonest 0‐3 hours, vomiting 8‐24 hours. Side effects not reported. Retching categorized vomiting, unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 60 children; no sex data; strabismus; >2 (mean 6); ASA1,2; exc' motion sickness, POV | |
| Interventions | Induction Either: DROPERIDOL 25 or 50 mcg/kg i.v | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐2; 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 60 adults; 32 women; 18‐75 (mean 48); craniotomy ; exc' ASA>3, antiemetic, pregnant/breast feeding, obese, mental retardation, psychiatric | |
| Interventions | Intraoperative Either: PLACEBO; or DROPERIDOL 0.625mg i.v.; or ONDANSETRON 4mg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetics. Postop 0‐1; 0‐4; 0‐8; 0‐12; 0‐24; 0‐48 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear if vomiters nauseated or categorized once or twice. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; N; N | |
| Participants | 50 adults; 23 women; craniotomy; 18‐75 (mean 54); excluded ASA>3, antiemetic, study drug allergy, pregnant/breast feeding, obese, mental retardation, psychiatric | |
| Interventions | Intraoperative Either: PLACEBO; or ONDANSETRON 8mg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐1; 0‐4; 0‐8; 0‐12; 0‐24; 0‐48 hours. | |
| Notes | Male/female incidences and side effects not reported. Retching categorised as vomiting. Unclear if vomiters nauseated or categorized once of twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; N; N; N; N | |
| Participants | 585 adults; general; no exclusion | |
| Interventions | Preoperative Either: NO TREATMENT; or DROPERIDOL 5mg i.m. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐36 hours. | |
| Notes | Male/female incidences not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 134 children; 74 girls; tonsil +/‐ adenoid; exc' hypertension, CNS disease, obese, malabsorption, study drug allergy | |
| Interventions | Preoperative AND preoperative Either: oral CLONIDINE 4 mcg/kg then PLACEBO; or PLACEBO then oral MIDAZOLAM 0.5mg/kg | |
| Outcomes | Vomiting. Postop 0‐10; 10‐24; 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Side effects "clonidine group excited". Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 30 adults; no sex data; cardiac; exc' prior cardiac surgery, bacterial infection, immunodysfunction | |
| Interventions | Preoperative AND postoperative four times Either: PLACEBO; or METHYLPREDNISOLONE 15mg i.v. each time | |
| Outcomes | Nausea or vomiting. Postop 0‐24; 24‐48; 48‐72 hours. | |
| Notes | Male/female incidences not recorded. Nausea or vomiting commonest 24‐48 hours. No side effects. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; N; N; Y; Y | |
| Participants | 75 children; 23 girls; day case; ASA1,2; 1‐10 (mean 5) | |
| Interventions | Induction Either: PLACEBO; or METOCLOPRAMIDE 150 mcg/kg i.v.; or ONDANSETRON 100 mcg/kg i.v. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. No side effects. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 90 adults; 35 women; orthopaedic; ASA1,2 | |
| Interventions | Induction Either: PLACEBO; or intrathecal CLONIDINE 75‐100 mcg | |
| Outcomes | Nausea; vomiting. Postop 0‐24 hours. | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; N; Y; Y | |
| Participants | 40 women; D&C; 16‐39; exc' ASA>1, hypertension, DM, asthma | |
| Interventions | Induction Either: PLACEBO; or METOCLOPRAMIDE 10mg i.v. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐1; 1‐3; 3‐6; 6‐12; 12‐24 hours. | |
| Notes | Nausea and vomiting commonest 0‐1 hour. Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; N; N | |
| Participants | 2061 adults; 1817 women; day case; exc' ASA>1, obese, pregnant/breastfeed, drug abuse, antiemetic | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 0.625 or 1.25 mg i.v.; or ONDANSETRON 4mg i.v. | |
| Outcomes | Nausea; nausea or vomiting; rescue antiemetic. Postop 0‐2; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Unclear how retching categorized or if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; N; N | |
| Participants | 180 adults; ASA 1,2; post hoc exclusion participants (56/180) | |
| Interventions | Postoperative on analgesic request Either: PLACEBO; or METHYLNALTREXONE 0.01 or 0.1 or 0.3mg/kg i.v. | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐3 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 120 women; gynaecological; exc' intubation, psychosis/psychotropic drug | |
| Interventions | Induction Either: NO TREATMENT; or DROPERIDOL 0.5 or 1mg i.v. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Unclear how retching categorized or if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 45 adults; 21 women; orthopaedic; ASA2‐4; >70 (mean 78); exc' psychiatric, study drug allergy, COPD, bleeding disorder | |
| Interventions | Postoperative if pain score >3/10 Either: PLACEBO; or intrathecal CLONIDINE 30 mcg; or EPINEPHRINE 200 mcg | |
| Outcomes | Rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 87 children; 2‐9; ENT, orthopaedic/general; <29kg | |
| Interventions | Preoperative Either: oral DROPERIDOL 0.4mg/kg; or DROPERIDOL 0.2mg/kg AND DIAZEPAM 0.1mg/kg | |
| Outcomes | Vomiting. Postop time unclear. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 30 adults; 16 women; mean 31; ASA1,2; maxillofacial | |
| Interventions | Preoperative Either: oral CLONIDINE 5 mcg/kg; or MIDAZOLAM 100 mcg/kg | |
| Outcomes | Nausea or vomiting. Postop 0‐2 hours. | |
| Notes | Male/female incidences not reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 40 adults; 20 women; orthopaedic; ASA1,2; exc' CNS/GI disease, anxiety, chronic pain, opioid abuse, study drug allergy | |
| Interventions | Postoperative Either: PLACEBO; or PCA DROPERIDOL 0.0625 mcg/hr | |
| Outcomes | Vomiting Postop 0‐12; 12‐24; 24‐36; 36‐48; 48‐60; 0‐60 hours. | |
| Notes | Male/female incidences not reported. No side effects. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; Y; Y; Y | |
| Participants | 100 children; 34 girls; >2 (mean 4); various surgeries; exc' ENT | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 50mcg/kg | |
| Outcomes | Nausea or vomiting. Postop 0‐4 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 60 women; gynaecological; ASA1,2; 28‐67 (46); exc' cardiac/lung/renal/liver/CNS disease, antiemetic | |
| Interventions | Postoperative Either: PLACEBO; or GRANISETRON 3mg i.v.; or METOCLOPRAMIDE 10mg i.v. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐3; 3‐24 hours. | |
| Notes | Nausea and 'nausea or vomiting' commonest 3‐24 hours, vomiting 0‐3 hours. No side effects. Unclear how retching categorized or if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 100 women; gynaecological; ASA1,2; 25‐65 (mean 45); exc' cardiac/lung/renal/liver/CNS disease, antiemetic | |
| Interventions | Postoperative Either: PLACEBO; or GRANISETRON 20 or 40 or 60 mcg/kg i.v. | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | No side effects. Retching categorized as nausea. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 88 women; gynaecological; ASA1,2; 25‐68 (mean 43); exc' cardiac/lung/renal/liver/CNS disease, antiemetic | |
| Interventions | Postoperative Either; PLACEBO; or DEXAMETHASONE 8mg i.v.; or GRANISETRON 20 mcg/kg i.v.; or both | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Retching categorized as nausea. Unclear if vomiters nauseated/retching or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 100 women; gynaecological; ASA1,2; 23‐67 (mean 42); exc' cardiac/renal/lung/liver/CNS disease, antiemetic | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 1.25 or 2.5mg i.v.; or GRANISETRON 40 mcg/kg i.v. | |
| Outcomes | Nausea; vomiting. Postop 0‐3; 3‐24 hours. | |
| Notes | Nausea commonest 3‐24 hours, vomiting 0‐3 hours. Retching categorized as nausea. Unclear if vomiters nauseated/retching or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 60 children; 25 girls; strabismus/tonsil +/‐ adenoid; 4‐10 (mean 7); ASA1,2; exc' antiemetic, motion sickness | |
| Interventions | Induction Either: GRANISETRON 40 mcg/kg i.v. +/‐ DEXAMETHASONE 4mg | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Unclear if vomiters retched. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 50 children; 21 girls; strabismus/tonsil +/‐ adenoid; ASA1,2; 4‐10 (mean 7); exc' antiemetic/N&V | |
| Interventions | Induction Either: PLACEBO; or GRANISETRON 40 mcg/kg i.v. | |
| Outcomes | Vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Unclear if vomiters retched. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 70 children; 32 girls; strabismus/tonsil +/‐ adenoid; excluded motion sickness/PONV | |
| Interventions | Induction Either: PLACEBO; or GRANISETRON 40 mcg/kg i.v.; or METOCLOPRAMIDE 0.25mg/kg i.v. | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐3; 3‐24 hours. | |
| Notes | Rescue antiemetic only 3‐24 hours. Vomiting commonest 0‐3 hours. Male/female incidences not reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 110 women; gynaecological; 21‐63 (mean 43); ASA1,2; exc' cardiac/lung/GI/renal/liver/CNS disease, antiemetic, pregnant | |
| Interventions | Induction Either: PLACEBO; or GRANISETRON 40 mcg/kg i.v. | |
| Outcomes | All outcomes Postop 0‐24 hours. | |
| Notes | Retching categorized as nausea. Unclear if vomiters retched or were nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 80 children; 34 girls; strabismus/tonsil +/‐ adenoid; ASA1,2; >4 (mean 7); exc' antiemetic/vomiting | |
| Interventions | Induction Either: PLACEBO; or GRANISETRON 20 or 40 or 80 mcg/kg i.v. | |
| Outcomes | Nausea or vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 270 women; gynaecological; ASA1,2; 23‐63 (mean 43); exc' GI disease, motion sickness, PONV, pregnant/menstrual, antiemetic | |
| Interventions | Induction Either: DROPERIDOL 1.25mg i.v.; or GRANISETRON 40 mcg/kg; or METOCLOPRAMIDE 10mg; or each drug with DEXAMETHASONE 8mg i.v. | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Unclear if nauseated retching vomiters categorized once, twice or thrice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 80 adults; 53 women; lap' chole'; 25‐65, ASA1,2; exc' GI disease, pregnant/menstrual, antiemetic | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 1.25mg i.v.; or GRANISETRON 3mg i.v. | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Unclear if retchers/vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 120 women; gynaecological; 23‐63; ASA1,2; exc' GI disease, pregnant/menstrual antiemetic | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 1.25mg i.v; or GRANISETRON 40 mcg/kg; or METOCLOPRAMIDE 10mg | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 120 women; gynaecological; 21‐45 (mean 38); ASA1,2; exc' GI disease, contraceptive, antiemetic | |
| Interventions | INDUCTION Either: PLACEBO; or GRANISETRON 40 mcg/kg i.v. | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 90 women; gynaecological; 23‐63; ASA1,2; exc' GI disease, pregnant/menstrual, antiemetic | |
| Interventions | Intraoperative Either: PLACEBO; or GRANISETRON 40 mcg/kg i.v. | |
| Outcomes | All outcomes Postop 0‐24 hours. | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 120 women; breast; 42‐66 (mean 53); exc' GI disease, antiemetic, menstrual/hormone drug | |
| Interventions | Induction Either: PLACEBO; or GRANISETRON 20 or 40 or 80 mcg/kg i.v. | |
| Outcomes | All outcomes Postop 0‐24 hours. | |
| Notes | Retching categorized as nausea. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 50 women; breast; 45‐68 (mean 55); ASA1,2; exc' GI disease, menstrual, hormone drug, antiemetic | |
| Interventions | Induction Either: PLACEBO; or GRANISETRON 2.5mg i.v. | |
| Outcomes | All outcomes Postop 0‐24 hours. | |
| Notes | Retching categorized as nausea. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 60 adults; 43 women; ASA1; ENT; exc' pregnancy/menstrual, GI disease, antiemetic | |
| Interventions | Induction Either: PLACEBO; or GRANISETRON 40 mcg/kg i.v. | |
| Outcomes | All outcomes Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. No side effects. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 120 adults; 85 women; ASA 1; middle ear; exc' pregnant/menstrual, GI disease, antiemetic | |
| Interventions | Induction Either: DEXAMETHASONE 8mg i.v.; or GRANISETRON 3mg i.v.; or both | |
| Outcomes | All outcomes Postop 0‐3; 3‐24 hours | |
| Notes | Outcomes commonest 0‐3 hours. Male/female incidences not reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; A; Y; Y; Y; Y | |
| Participants | 150 women; breast; ASA1,2; exc' GI disease, motion sickness, PONV, pregnant/menstrual, antiemetic | |
| Interventions | Induction Either: PLACEBO; or GRANISETRON 40 mcg/kg i.v. +/‐ or DEXAMETHASONE 8mg i.v. | |
| Outcomes | All outcomes. Postop 0‐3; 3‐24 hours | |
| Notes | Outcomes commonest 0‐3 hours. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 100 women; gynaecological; ASA1,2; 23‐63; exc' GI disease, motion sickness, PONV, pregnant/menstrual, antiemetic | |
| Interventions | Preoperative Either: DOMPERIDONE 20mg oral; or GRANISETRON 2mg | |
| Outcomes | All outcomes. Postop 0‐3; 3‐24 hours. | |
| Notes | Outcomes commonest 3‐24 hours. Unclear if vomiters retched or nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 80 children; 37 girls; strabismus/tonsil +/‐ adenoid; ASA1; 4‐10 (mean 7); exc' motion sickness, PONV, antiemetic | |
| Interventions | Induction Either: DROPERIDOL mcg/kg i.v.; or GRANISETRON 40 mcg/kg i.v. | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 120 women; lap' chole'; ASA1,2; 25‐65 years; exc' GI disease, pregnant/menstrual, antiemetic | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 1.25 mg/kg i.v.; or GRANISETRON 3mg i.v.; or METOCLOPRAMIDE 10mg i.v. | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; Y; Y; Y | |
| Participants | 150 women; 23‐65; elective lap' chole'; ASA1; exc' obese, GI disease, pregnant/menstrual, antiemetic | |
| Interventions | Induction Either: DROPERIDOL 1.25 mg i.v.; or GRANISETRON 3mg i.v.; or both | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 90 women; gynaecological; ASA1,2; 25‐63 (mean 44); exc' GI disease, pregnancy/menstrual, antiemetic | |
| Interventions | Induction Either: DROPERIDOL 1.25mg i.v.; or GRANISETRON 2.5mg i.v.; or METOCLOPRAMIDE 10mg i.v. | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Categorized by severest (vomiting>retching>nausea). I combined retching and vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 100 children; 44 girls; extremity surgery; ASA1; 4‐10 (mean 7); exc' motion sickness, PONV, antiemetic | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 50 mcg/kg i.v.; or GRANISETRON 40 mcg/kg; or METOCLOPRAMIDE 0.25 mg/kg | |
| Outcomes | Vomiting; rescue antiemetics. Postop 0‐24 hours. | |
| Notes | Unclear how retching categorized or if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; Y; Y; Y | |
| Participants | 120 women; caesarean; ASA1,2; 22‐35; exc' motion sickness, PONV, antiemetic, GI disease | |
| Interventions | Intraoperative Either: PLACEBO; or DROPERIDOL 1.25 mg i.v.; or GRANISETRON 3mg; or METOCLOPRAMIDE 10mg | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐3; 3‐24 hours. | |
| Notes | Outcomes commonest 0‐3 hours. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 120 women; gynaecological; ASA 1,2; menstruating; exc' antiemetic, GI disease | |
| Interventions | Induction Either: DROPERIDOL 25 mcg/kg i.v.; or GRANISETRON 40 mcg/kg; or METOCLOPRAMIDE 0.2 mg/kg | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | No side effects. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; Y; Y; Y | |
| Participants | 150 women; ASA1,2; breast; exc' motion sickness, PONV, menstrual, hormone drug, antiemetic | |
| Interventions | Induction Either: DROPERIDOL 1.25 mg i.v.; or GRANISETRON 3mg; or both | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐3; 3‐24; 0‐24 hours. | |
| Notes | I combined retching and vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 180 women; ASA1,2; middle ear; exc' pregnant, menstrual motion sickness, previous PONV, antiemetic | |
| Interventions | Induction Either: DROPERIDOL 20 mcg/kg i.v.; or GRANISETRON 40 mcg/kg; or METOCLOPRAMIDE 0.2 mg/kg | |
| Outcomes | All outcomes. Postop 0‐3; 3‐24 hours. | |
| Notes | Outcomes commonest 3‐24 hours. I combined retching and vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 150 women; ASA1,2; gynaecological; 21‐63; exc' motion sickness, PONV, GI disease, pregnancy/menstrual, antiemetic | |
| Interventions | Induction Either: DROPERIDOL 1.25 mg i.v.; or GRANISETRON 2.5 mg; or both | |
| Outcomes | All outcomes. Postop 0‐3; 3‐24; 0‐24 hours. | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 180 children; 88 girls; tonsil +/‐ adenoids; 4‐10 (mean 7); exc' antiemetic, motion sickness | |
| Interventions | Induction Either: DROPERIDOL 50 mcg/kg i.v.; or GRANISETRON 40 mcg/kg; or both | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐3; 3‐24 hours. | |
| Notes | Outcomes commonest 3‐24 hours. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 120 women; lap' chole'; ASA1,2; 25‐63; exc' GI disease, pregnant/menstrual, antiemetic | |
| Interventions | Induction Either: PLACEBO; or GRANISETRON 20 or 30 or 40 mcg/kg i.v. | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | I combined retching and vomiting. Unclear if vomiters retched or nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 120 women; ASA1,2; gynaecological; exc' GI disease, pregnant/menstrual, antiemetic | |
| Interventions | Preoperative Either: PLACEBO; or oral GRANISETRON 1 or 2 or 4mg | |
| Outcomes | All outcomes. Postop 0‐3; 3‐24; 0‐24 hours. | |
| Notes | I combined retching and vomiting. Unclear if vomiters retched or nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 120 women; ENT; ASA1; 25‐60 (mean 44) ; exc' GI disease, motion sickness/PONV, pregnancy/menstrual, antiemetic | |
| Interventions | Induction Either: PLACEBO; or GRANISETRON 20 or 40 or 100 mcg/kg i.v. | |
| Outcomes | All outcomes. Postop 0‐3; 3‐24 hours. | |
| Notes | Outcomes commonest 0‐3 hours. Unclear if all retchers vomited or nauseated. I combined retching and vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 160 children; 89 girls; tonsil +/‐ adenoid; 4‐10 (mean 7); ASA 1; exc' motion sickness, antiemetic | |
| Interventions | Preoperative Either: PLACEBO; or GRANISETRON 20 or 40 or 80 mcg/kg i.v. | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects "no association". | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 100 women; thyroid; ASA 1; exc' motion sickness, PONV, menstrual, antiemetic | |
| Interventions | Induction Either: PLACEBO; or GRANISETRON 20 or 40 or 100 mcg/kg | |
| Outcomes | All outcomes. Postop 0‐3; 3‐24 hours. | |
| Notes | Nausea commonest 0‐3 hours, vomiting and 'nausea or vomiting' 0‐24 hours. I combined retching and vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 120 women; breast; 43‐64 (mean 53); exc' GI disease, antiemetic, menstrual/hormone drug | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 2.5mg i.v.; or GRANISETRON 40 mcg/kg; or METOCLOPRAMIDE 0.2 mg/kg | |
| Outcomes | All outcomes. Postop 0‐3; 3‐24; 0‐24 hours. | |
| Notes | Categorized by severest symptom (vomiting>retching>nausea). | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 150 children; 55 girls; ASA1; 4‐10 (mean 7); general; exc' motion sickness, PONV, antiemetic | |
| Interventions | INDUCTION Either: DEXAMETHASONE 150 mcg/kg i.v.; or GRANISETRON 40 mcg/kg; or both | |
| Outcomes | Vomiting; rescue antiemetics Postop 0‐3; 3‐24; 0‐24 hours | |
| Notes | Male/female incidences not reported. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 120 women; caesarean; ASA1,2; 24‐38 (mean 29); exc' GI disease, motion sickness/PONV, antiemetic | |
| Interventions | Intraoperative Either: GRANISETRON 3mg i.v. +/‐ DEXAMETHASONE 8mg | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | I combined retching and vomiting. Unclear if retchers or vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 120 women; GA; thyroid; ASA 1; excluded PONV/motion sickness, pregnant/menstrual, GI disease, antiemetic | |
| Interventions | Induction Either: DROPERIDOL 20 mcg/kg i.v.; or GRANISETRON 40 mcg/kg; or METOCLOPRAMIDE 0.2 mg/kg | |
| Outcomes | All outcomes. Postop 0‐3; 3‐24 hours. | |
| Notes | Outcomes commonest 3‐24 hours. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 120 children; 63 girls; strabismus; 4‐10 (mean 7); ASA1; exc' motion sickness/POV, antiemetic | |
| Interventions | Induction Either: DROPERIDOL 50 mcg/kg i.v.; or GRANISETRON 40 mcg/kg; or both | |
| Outcomes | Vomiting: rescue antiemetic. Postop 0‐3; 3‐24 hours. | |
| Notes | Vomiting commonest 3‐24 hours. Male/female incidences not reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; N; Y; Y | |
| Participants | 150 adults; 108 women; middle ear; ASA1; 20‐68 (mean 45); exc' GI disease, motion sickness/PONV, antiemetic | |
| Interventions | Induction Either: DROPERIDOL 20 mcg/kg i.v.; or GRANISETRON 40 mcg/kg; or both | |
| Outcomes | All outcomes. Postop 0‐3; 3‐24 hours. | |
| Notes | Outcomes commonest 0‐3 hours. Male/female incidences not reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 180 women; thyroid; ASA1; 32‐58 (mean 46); exc' GI disease, motion sickness, PONV, pregnant/menstrual, antiemetic | |
| Interventions | Induction Either: DROPERIDOL 20 mcg/kg i.v.; or GRANISETRON 40 mcg/kg; or both | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | I combined retching and vomiting. Unclear if retchers/vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 100 children; 55 girls; tonsil +/‐ adenoid; ASA1; 4‐10 (mean 7); exc' POV, antiemetic, motion sickness | |
| Interventions | Preoperative Either: GRANISETRON 40 mcg/kg i.v.; or PERPHENAZINE 70 mcg/kg | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐3; 3‐24 hours. | |
| Notes | Vomiting commonest 0‐3 hours. Male/female incidences not reported. I combined retching and vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 100 adults; 69 women; ASA 1; middle ear; exc' previous PONV/motion sickness, pregnant/menstrual, antiemetic | |
| Interventions | Induction Either: GRANISETRON 3mg; or RAMOSETRON 0.3mg | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24; 24‐48 hours. | |
| Notes | Outcomes commonest 24‐48 hours. Male/female incidences not reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 80 women: lap' chole'; ASA1,2; 25‐65 (mean 46); exc' GI disease, pregnant/menstrual, antiemetic | |
| Interventions | Intraoperative end Either: GRANISETRON 3mg i.v.; or RAMOSETRON 0.3mg | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24; 24‐48 hours. | |
| Notes | Outcomes commonest 24‐48 hours. Side effects "no differences". Unclear if retchers vomited or if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 120 women; gynaecological; ASA1,2; 23‐65 (mean 44); exc' GI disease, motion sickness, pregnant/menstrual, antiemetic, PONV | |
| Interventions | Intraoperative end Either: GRANISETRON 2.5mg i.v.; or RAMOSETRON 0.3mg | |
| Outcomes | All outcomes. Postop 0‐3; 3‐24; 24‐48 hours. | |
| Notes | Nausea and nausea or vomiting commonest 24‐48 hours, vomiting 3‐24. I combined retching and vomiting. Unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 90 children; 47 girls; tonsil +/‐ adenoid; ASA 1; 4‐10 (mean 7); exc' motion sickness, PONV, antiemetic | |
| Interventions | Induction Either: GRANISETRON 40 mcg/kg i.v.; or PERPHENAZINE 70 mcg/kg | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐3; 3‐24 hours. | |
| Notes | Outcomes commonest 3‐24 hours. Male/female incidences not reported. Granisetron and perphenazine dose units incorrect. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; N; Y; Y | |
| Participants | 120 children; 54 girls; 4‐10 (mean 6); general; exc' motion sickness/PONV, antiemetic | |
| Interventions | Induction Either: PLACEBO; or GRANISETRON 20 or 40 or 100 mcg/kg i.v. | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 120 children; 57 girls; 6 (4‐10) years; ASA1,2; tonsil +/‐ adenoids; exc' POV, GI disease, antiemetic | |
| Interventions | Induction Either: PLACEBO; or GRANISETRON 40 mcg/kg i.v. | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. I combined retching and vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 120 children; 62 girls; 6‐10 (mean 7); ASA1; strabismus; exc' motion sickness, PONV, antiemetic | |
| Interventions | Preoperative Either: PLACEBO; or oral GRANISETRON 20 or 40 or 80 mcg/kg | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 120 adults; 83 women; lap' chole'; ASA1; 25‐65 (47 mean); exc' GI disease, pregnant/menstrual, antiemetic | |
| Interventions | Induction Either: GRANISETRON 40 mcg/kg i.v. +/‐ DEXAMETHASONE 8mg | |
| Outcomes | All outcomes. Postop 0‐3; 3‐24 hours. | |
| Notes | Nausea commonest 0‐3 hours, vomiting 3‐24 hours. Male/female incidences not reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 130 women; thyroid; ASA1; 33‐58 (mean 47); exc' GI disease, pregnant/menstrual, antiemetic | |
| Interventions | Induction Either: GRANISETRON 40 mcg/kg i.v. +/‐ DEXAMETHASONE 8mg | |
| Outcomes | All outcomes. Postop 0‐3; 3‐24 hours | |
| Notes | I combined retching and vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 120 women; gynaecological; 21‐63; ASA1,2; exc' GI disease, antiemetic, PONV | |
| Interventions | Intraoperative end Either: PLACEBO; or RAMOSETRON 0.15 or 0.3 or 0.6mg i.v. | |
| Outcomes | All outcomes. Postop 0‐3; 3‐24; 24‐48 hours. | |
| Notes | Outcomes commonest 0‐3 hours. Side effects "no difference". Unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 90 adults; 48 women; middle ear; ASA1; exc' pregnant/menstrual, antiemetic, disease | |
| Interventions | Intraoperative Either: DROPERIDOL 20mcg/kg i.v.; or METOCLOPRAMIDE 0.2mg/kg; or PROPOFOL 0.5mg/kg | |
| Outcomes | All outcomes. Postop 0‐3; 3‐24 hours. | |
| Notes | Outcomes commonest 3‐24 hours. Male/female incidences not reported. Unclear how retching categorized or if nauseated vomiters categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 90 adults; 71 women; thyroid; ASA1; 33‐58 (mean 46); exc' GI disease, pregnant/menstrual, antiemetic | |
| Interventions | Intraoperative Either: DROPERIDOL 20mcg/kg i.v.; or METOCLOPRAMIDE 0.2mg/kg; or PROPOFOL 0.5mg/kg | |
| Outcomes | All outcomes. Postop 0‐3; 3‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Categorized by severest symptom (vomiting>retching>nausea). I combined retching and vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 90 children; 43 girls; tonsil +/‐ adenoid; ASA1; 4‐10 (mean 6); exc' GI disease, POV, antiemetic | |
| Interventions | Induction Either: DROPERIDOL 50 mcg/kg i.v.; or GRANISETRON 40 mcg/kg; or METOCLOPRAMIDE 0.25 mg/kg | |
| Outcomes | Vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 90 children; 47 girls; tonsil +/‐ adenoid; ASA 1; 4‐10 (mean 6); exc' motion sickness, PONV, antiemetic | |
| Interventions | Intraoperative Either: GRANISETRON 40 mcg/kg i.v.; or RAMOSETRON 6 mcg/kg | |
| Outcomes | Vomiting. Postop 0‐24; 24‐48 hours. | |
| Notes | Vomiting commonest 24‐48 hours. Male/female incidences not reported. Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 80 children; 42 girls; 4‐10 (mean 7); strabismus; ASA1; exc' motion sickness, PONV, antiemetic | |
| Interventions | Intraoperative Either: GRANISETRON 40 mcg/kg i.v.; or RAMOSETRON 6 mcg/kg | |
| Outcomes | Vomiting. Postop 0‐24; 24‐48 hours. | |
| Notes | Vomiting commonest 24‐48 hours. Male/female incidences not reported. No side effects. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 100 women; mastectomy; ASA1; 29‐66; exc' pregnant/menstrual; GI disease, antiemetic | |
| Interventions | Preoperative Either: PLACEBO; or oral GRANISETRON 1 or 2 or 4mg | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | No side effects. Categorized by severest symptom (vomiting>retching>nausea). I combined retching and vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 130 adults; 98 women; lap' chole'; ASA1; 25‐63 (mean 47); exc' antiemetic, steroid, post‐hoc conversion | |
| Interventions | Preoperative Either: PLACEBO; or oral GRANISETRON 1 or 2 or 4mg | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. No side effects. Unclear if vomiters retched or nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; Y; Y; Y | |
| Participants | 120 women; STOP; ASA1; 19‐47 (mean 33); exc' antiemetic, GI disease, motion sickness, PONV | |
| Interventions | Induction Either: PLACEBO; or DEXAMETHASONE 4 or 8 or 16mg i.v. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | No side effects. Unclear if vomiters nauseated. I combined retching and vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 100 children; 41 girls; general; 4‐11 (mean 7); ASA1; exc' POV/motion sickness, antiemetic | |
| Interventions | Preoperative Either: PLACEBO; or oral GRANISETRON 20 or 40 or 80 mcg/kg | |
| Outcomes | Vomiting. Postop 0‐6; 6‐24 hours. | |
| Notes | Vomiting commonest 0‐6 hours. Male/female incidences not reported. No side effects. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 80 adults; 65 women; thyroid; ASA1; median 46; exc' antiemetic, GI disease, motion sickness, PONV, pregnant/menstrual | |
| Interventions | Induction Either: GRANISETRON 3mg i.v.; or RAMOSETRON 0.3mg | |
| Outcomes | All outcomes. Postop 0‐24; 24‐48 hours. | |
| Notes | Nausea, vomiting, 'nausea or vomiting, commonest 24‐48 hours. Male/female incidences not reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; Y; Y. | |
| Participants | 110 adults; 71 women; lap' chole'; ASA1; 23‐68 (mean 48); exc' antiemetic, cholecystitis, steroid, post‐hoc conversion | |
| Interventions | Intraoperative Either: PLACEBO; or RAMOSETRON 0.15 or 0.3 or 0.6mg iv | |
| Outcomes | All outcomes. Postop 0‐24; 24‐48 hours. | |
| Notes | Nausea commonest 24‐48 hours, others 0‐24 hours. Side effects "headache 8% to 12%". I combined retching and vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 90 women; dental; 15‐57 (mean 36); ASA1; exc' GI disease, pregnant/menstrual, antiemetic | |
| Interventions | Postoperative Either placebo; or PROPOFOL 0.25 or 0.5mg/kg iv. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐3; 3‐24 hours. | |
| Notes | Nausea and 'nausea or vomiting' commonest 3‐24 hours, vomiting 0‐3 hours. Unclear if retchers or vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 80 adults; 58 women; orthopaedic; mean 61; exc' GI disease, motion sickness/PONV, antiemetic | |
| Interventions | Intraoperative Either: PLACEBO; or RAMOSETRON 0.15 or 0.3 or 0.6mg iv | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Categorized by severest symptom (vomiting>retching>nausea). I combined retching and vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 256 children; 117 girls; ASA1,2; 2‐12 (mean 6); tonsil +/‐ adenoid | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 75 mcg/kg i.v.; or METOCLOPRAMIDE 0.5 mg/kg i.v.; or ONDANSETRON 0.15 mg/kg i.v. | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐7 hours. | |
| Notes | Vomiting reported at 0‐8 hours and 8‐24 hours. Male/female incidences not reported. No side effects. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; N; Y; Y | |
| Participants | 67 children; 27 girls; craniotomy; 2‐18 (mean 9); exc' antiemetic | |
| Interventions | Induction AND postoperative Either: PLACEBO; or ONDANSETRON 0.15mg/kg i.v. | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐8; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 84 women; D&C | |
| Interventions | Preoperative Either: DIAZEPAM 10mg/70kg i.m.; or LORAZEPAM 5mg/70kg | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Incidences not reported. No side effects. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 120 adults; 67 women; orthopaedic; ASA1,2; no exclusion | |
| Interventions | Postoperative Either: PLACEBO; or DROPERIDOL 1.25 mg i.v.; or ONDANSETRON 4mg | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. No side effects. Unclear how retching categorized or if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; N; N | |
| Participants | 82 adults; 40 women; orthopaedic; ASA1,2; exc' phenothiazine, study drug allergy | |
| Interventions | Postoperative immediate AND PCA Either: PLACEBO/DROPERIDOL 1.25mg i.v. then PLACEBO/DROPERIDOL PCA (0.08mg/ml) | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects "no extrapyramidal reaction". Retching categorized as vomiting. Unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 60 women; hysterectomy; ASA < 4 | |
| Interventions | Postoperative Either: PLACEBO; or NALOXONE 0.25 or 1 mcg/kg/h | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized or if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 60 women; gynaecological; ASA1,2 | |
| Interventions | Postoperative Either: PLACEBO; or oral ONDANSETRON 8mg | |
| Outcomes | Nausea; vomiting. Postop 0‐22 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. Unclear if drug treatment not prophylaxis. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 52 women; laparoscopies; ASA1,2; 21‐50 (mean 34); exc' BMI>29, PONV, motion sickness, psychiatric, menstrual, drug abuse | |
| Interventions | Induction Either: ALIZAPRIDE 50mg i.v.; or ONDANSETRON 4mg | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐12 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 45 women; laparoscopies; ASA1,2; 18‐46 (mean 31); exc' GI/psychiatric disease, PONV motion sickness, menstrual, drug abuse | |
| Interventions | Induction Either: PLACEBO; or DEXAMETHASONE 8mg iv | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐12 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; N; Y; Y | |
| Participants | 232 adults; no sex data; various surgeries; exc' <15 years | |
| Interventions | Preoperative Either: DIAZEPAM 2mg/10kg i.v.; or FLUNITRAZEPAM; or METHOHEXITONE 10mg/10kg | |
| Outcomes | Nausea or vomiting. Postop time unclear. | |
| Notes | Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 60 women; gynaecological; ASA1 | |
| Interventions | Induction Either: DROPERIDOL 1.25mg i.v.; or METOCLOPRAMIDE 10mg; or ONDANSETRON 8mg | |
| Outcomes | Nausea; vomiting. Postop 0‐24 hours. | |
| Notes | No side effect. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; N; N | |
| Participants | 88 adults; 38 women; arthroscopy; ASA1,2; mean 42; exc' analgesics, alpha 2 blockers | |
| Interventions | Intraoperative Either: PLACEBO; or articular CLONIDINE 150 mcg or NEOSTIGMINE 500 mcg or both and PLACEBO s/c; or articular CLONIDINE 150 mcg and NEOSTIGMINE 500 mcg s/c; or articular NEOSTIGMINE 500 mcg and CLONIDINE 150 mcg s/c | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not given. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; N; N | |
| Participants | 157 women; hysterectomy; ASA1,2; exc' antiemetic, N&V, obese | |
| Interventions | Preoperative AND intraoperative Either: oral CP‐122,721 200mg then PLACEBO / ONDANSETRON 4mg i.v.; or oral PLACEBO then ONDANSETRON 4mg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐8; 0‐24 hours. | |
| Notes | Retching categorized as vomiting. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; A; Y; N; N; N | |
| Participants | 50 children; 25 girls; tonsil +/‐ adenoids; 3‐15 | |
| Interventions | Intraoperative Either: PLACEBO; or DEXAMETHASONE 1mg/kg (max 16mg) i.v. | |
| Outcomes | Nausea or vomiting; rescue antiemetic Postop 0‐4 hours; 0‐5 days. | |
| Notes | Nausea or vomiting 0‐5 days, rescue antiemetic 0‐4 hours. Male/female incidences not reported. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; N; N; N; N | |
| Participants | 50 women; hysterectomy; ASA1,2; exc' >75years, >90kg | |
| Interventions | Intraoperative AND postoperative Either: PLACEBO twice; or MIDAZOLAM 0.07mg/kg i.v. then 0.014mg/kg/hr | |
| Outcomes | Nausea; rescue antiemetic. Postop 0‐48 hours. | |
| Notes | Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 40 adults; 17 women; various surgeries; ASA1‐4; exc' antiepileptic drug, obese, renal/liver disease | |
| Interventions | Induction Either: Propofol +/‐ Midazolam 2.5 to 5 mg | |
| Outcomes | All outcomes. Postop in PACU; at 24 hours | |
| Notes | Nausea or vomiting commonest 24 hours, others PACU. Male/female incidences not reported. No side effects. Unclear how retching categorized | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 60 adults; 27 women; middle ear; ASA1,2; 20‐60 (mean 37); exc' motion sickness, antiemetic, GI disease, menstrual | |
| Interventions | Induction Either: DROPERIDOL 1.25mg iv; or GRANISETRON 3mg +/‐ DEXAMETHASONE 8mg | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐3; 3‐24 hours. | |
| Notes | Outcomes commonest 0‐3 hours. Male/female incidences not reported. Inconsistent results. Unclear if vomiters nauseated or if retchers/vomiters categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; N; Y; Y | |
| Participants | 240 women; laparoscopy; ASA1,2; 19‐70 (mean 37); exc' pregnant/breastfeeding, postmenopausal, obese, renal/liver disease, CNS injury, cytostatic therapy, antiemetic | |
| Interventions | Induction Either: OXYGEN 30% or 80%; or ONDANSETRON 8 mg i.v. | |
| Outcomes | All outcomes. Postop 0‐6; 6‐24; 0‐24 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 80 children; 35 girls; ASA1,2; 2‐14 (mean 10); orthopaedic | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 60 mcg/kg i.v.; or ONDANSETRON 50 or 100 mcg/kg | |
| Outcomes | Nausea or vomiting; rescue antiemetics. Postop 0‐3; 3‐48 hours. | |
| Notes | Outcomes commonest 3‐48 hours. Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; N; N; Y; Y | |
| Participants | 70 children; no sex data; 2‐10 years; tonsil +/‐ adenoids; ASA1,2; exc' ASA>2, obese, PONV, study drug allergy | |
| Interventions | Induction Either: PLACEBO; or GRANISETRON 1 or 10 or 100 mcg/kg i.v. | |
| Outcomes | Vomiting. Postop PACU; 0‐hospital discharge; 0‐24 hours. | |
| Notes | Unclear which groups had which drugs. Unclear how retching categorized. Male/female incidences not recorded. Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; N | |
| Participants | 161 children; 83 girls; adenotonsillectomy; 3‐12 | |
| Interventions | Preoperative Either: oral DIAZEPAM 0.22mg/kg; or PHENOBARBITONE 4.4mg/kg; or TRIMEPRAZINE 3.3mg/kg | |
| Outcomes | Vomiting. Postop 0‐18 hours. | |
| Notes | Male/female incidences not reported. No side effects. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 200 women; caesarean; mean 27 | |
| Interventions | Postoperative Either: PLACEBO; or BROMOPRIDE 10mg i.m. | |
| Outcomes | Nausea; vomiting. Postop 2‐24 hours. | |
| Notes | Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 26 women; ASA1,2; hysterectomy; 37‐60; exc' drug abuse | |
| Interventions | Preoperative Either: NO TREATMENT; or oral CLONIDINE 5 mcg/kg | |
| Outcomes | Nausea; rescue antiemetic. Postop 0‐48 hours. | |
| Notes | Incidences not reported. No side effects. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; N, N. | |
| Participants | 60 women; hysterectomy; ASA1,2; 17‐56 (mean 44); exc' drug abuse | |
| Interventions | Preoperative Either: PLACEBO; or oral CLONIDINE 5 mcg/kg | |
| Outcomes | Nausea. 0‐48 hours. | |
| Notes | Incidence not reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 90 adults; 48 women; orthopaedic; ASA1,2; 18‐80 (mean 67); exc' obese, adrenergic/psychotropic/analgesic drugs, fertile | |
| Interventions | Induction Either: PLACEBO; or intrathecal CLONIDINE 75 mcg | |
| Outcomes | Vomiting; rescue antiemetics. Postop 0‐6 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear how 'emesis', retching or vomiting defined. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; N; Y | |
| Participants | 442 adults; 98 women; cardiac; mean age 62 | |
| Interventions | Postoperative Either: PLACEBO; or DROPERIDOL 0.3 mg/hr | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Retching categorized as vomiting. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 231 adults; 98 women; general; 18‐80; exc' obese, antiemetic, N&V, fever/infection | |
| Interventions | Intraoperative AND postoperative Either: OXYGEN 30% or 80% | |
| Outcomes | All outcomes. Postop 0‐6; 6‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; N; N | |
| Participants | 74 women; laparoscopies; ASA1,2 | |
| Interventions | Induction Either: CYCLIZINE 50 mg i.v.; or ONDANSETRON 4mg | |
| Outcomes | All outcomes. Postop 0‐6; 6‐24 hours | |
| Notes | Outcomes commonest 0‐6 hours. Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 80 women; gynaecological; ASA1,2; 18‐65 (mean 35); exc' pregnant, drug abuse, psychiatric, study drug allergy, antiemetic | |
| Interventions | Induction Either: DROPERIDOL 2.5mg i.v.; or ONDANSETRON 8mg | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 45 adults; 23 women; orthopaedic; ASA1‐3; 18‐78 (mean 52) | |
| Interventions | Preoperative Either: oral CLONIDINE 2 or 5 mcg/kg; or oral MIDAZOLAM 0.1mg/kg | |
| Outcomes | Nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; N; N | |
| Participants | 60 women; lap' steri'; ASA1,2; 25‐44; exc' NSAID intolerance, intubation | |
| Interventions | Induction Either: PLACEBO; or GLYCOPYRROLATE 0.3mg i.v. | |
| Outcomes | Nausea or vomiting; rescue antiemetic. Postop 0‐3 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 50 women; STOP; mean 24 | |
| Interventions | Induction Either: DOMPERIDONE 10mg i.v.; or METOCLOPRAMIDE 10mg | |
| Outcomes | Nausea or vomiting; rescue antiemetic. Postop 0‐0.5; 0.5; 1‐2; 0‐2 hours. | |
| Notes | Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 58 adults; 32‐65 (mean 43); ASA1,2 | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 5mg i.v.; or ONDANSETRON 8mg; or DROPERIDOL 5mg AND ONDANSETRON 4mg | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐1; 1‐6; 6‐24; 0‐24 hours. | |
| Notes | Incidences not reported. Unclear how retching categorized or if nauseated vomiters categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 60 children; 32 girls; inguinal/urogenital; ASA1,2; 1‐12 (mean 5) | |
| Interventions | Postoperative Either: PLACEBO; or intrathecal MIDAZOLAM 50 mcg/kg | |
| Outcomes | Vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. No side effects. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 80 children; 24 girls; strabismus; ASA1; 3‐12 (mean 7); exc' obese, GI disease | |
| Interventions | Preoperative Either: PLACEBO; or oral CLONIDINE 4 mcg/kg | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐6; 6‐24; 24‐48; 0‐48 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 40 children; no sex data; craniofacial; mean 1 | |
| Interventions | Induction AND postoperative Either: PLACEBO twice; or ONDANSETRON 0.15mg/kg i.v. twice | |
| Outcomes | Vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 90 women; hysterectomy; 20‐68 (mean 52); ASA 1,2; exc' heart/lung/CNS disease, bleeding disorder, pregnancy | |
| Interventions | Postoperative immediate Either: PLACEBO; or epidural DROPERIDOL 2.5mg or CLONIDINE 150 mcg | |
| Outcomes | Nausea; rescue antiemetics. Postop 10 minutes; 0‐24 hours. | |
| Notes | Unclear how retching categorized. Side effects "clonidine sedative". | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 60 participants; 38 women; laminectomy; ASA1,2 | |
| Interventions | Intraoperative AND postoperative Either: DEXAMETHASONE 4mg iv then 0.005mg/kg/hour; or ONDANSETRON 4mg then 0.005mg/kg/hr | |
| Outcomes | Nausea; vomiting. Postop 0‐18 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; N; N | |
| Participants | 109 women; hysterectomy; ASA1,2; exc' heart disease, antiemetic | |
| Interventions | Intraoperative Either: PLACEBO; or EPHEDRINE 0.5 mg/kg i.m. | |
| Outcomes | Vomiting; nausea or vomiting; rescue antiemetics. Postop 0‐1; 1‐2; 2‐3; 3‐24 hours. | |
| Notes | Outcomes commonest 3‐24 hours. Retching categorized as vomiting. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; Y; N; N | |
| Participants | 300 adults; 60 women; CABG; mean 64; exc' steroid, arrhythmia | |
| Interventions | Induction AND postoperative Either: PLACEBO twice; or DEXAMETHASONE 4mg iv twice | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐24; 24‐48 hours. | |
| Notes | Rescue antiemetic only 0‐24 hours. Nausea and vomiting commonest 0‐24 hours. Male/female incidences not reported. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; Y; N; N | |
| Participants | 74 children; 39 girls; tonsil + adenoids; ASA1,2; 2‐10 (mean 6); exc' sleep apnoea, PONV, obese, antiemetic, study drug allergy | |
| Interventions | Induction Either: PLACEBO; or DIMENHYDRINATE 0.5mg/kg i.v.; or ONDANSETRON 0.1 mg/kg | |
| Outcomes | Vomiting. Postop 0‐10; 10‐24; 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; A; N; N; N; N | |
| Participants | 180 adults; 106 women; breast or abdominal | |
| Interventions | Induction or continuous AND postoperative Either: NO TREATMENT twice; or DEXAMETHASONE 4mg i.v.+ DROPERIDOL 1.25mg + METOCLOPRAMIDE 10mg + ONDANSETRON 4mg then NO TREATMENT; or PROPOFOL 1mg/kg/hr | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐4; 5‐8; 0‐24 hours. | |
| Notes | Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; N; N; Y; Y | |
| Participants | 100 adults; no sex data; corneal graft | |
| Interventions | Induction Either: DOMPERIDONE 10mg i.v.; or METOCLOPRAMIDE 10mg i.v. | |
| Outcomes | All outcomes. Postop 0‐1; 1‐6; 6‐15; 0‐15 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 60 children; 29 girls; 2‐12 (mean 8); ASA1; strabismus; exc' motion sickness, POV, antiemetic | |
| Interventions | Preoperative Either: oral CLONIDINE 4 mcg/kg; or oral DIAZEPAM 0.4 mg/kg | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Male/female incidences not given. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 65 women; gynaecological; <60 years | |
| Interventions | Intraoperative Either: PLACEBO; or METOCLOPRAMIDE 10mg i.m.; or PERPHENAZINE 5mg | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐4 hours. | |
| Notes | Unclear how retching categorized or if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; N; N | |
| Participants | 80 children; 28 girls; strabismus; ASA1,2; 1‐6 (mean 3) | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 50 mcg/kg i.v. | |
| Outcomes | Nausea or vomiting; rescue antiemetic. 0‐PACU discharge; 0‐5; 5‐24; 24‐48; 5‐48 hours. | |
| Notes | Rescue antiemetic only 0‐5 hours. Nausea or vomiting commonest 5‐48 hours. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; N; N | |
| Participants | 40 women; gynaecological; ASA1,2; exc' study drug allergy, glaucoma, bladder atony, H2 antagonist, antiemetic, NG | |
| Interventions | Postoperative Either: NO TREATMENT; or SCOPOLAMINE patch | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐2; 2‐4; 4‐6; 6‐24 hours. | |
| Notes | Outcomes commonest 0‐2 hours. Unclear how retching categorized. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 36 men; colorectal; mean 51 | |
| Interventions | Induction Either: NO TREATMENT; or DROPERIDOL 0.5mg/kg i.v.; or METOCLOPRAMIDE 0.5mg/kg | |
| Outcomes | Nausea; vomiting. Postop 0‐2; 2‐12; 12‐24 hours. | |
| Notes | Outcomes commonest 0‐2 hours. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; N; N; N; N | |
| Participants | 1334 adults; 992 women; various surgeries; exc' <18 >75 | |
| Interventions | Intraoperative Either: PLACEBO; or DROPERIDOL mcg/kg i.v. | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; N; N | |
| Participants | 923 women; gynaecological; ASA1‐3; 18‐65 (mean 43); exc' CNS, renal, liver, heart, metabolic/endocrine disease, N&V/antiemetic, NG, pregnant/breastfeeding | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 1 or 8 or 16mg i.v. | |
| Outcomes | Nausea; vomiting. Postop 0‐24 hours. | |
| Notes | Retching categorized as vomiting. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 160 adults; 125 women; ASA1,2; lap' chole'; exc' study drug allergy, N&V/antiemetic, drug abuse, renal/liver/heart/lung/CNS/blood or endocrine disease | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 1.25mg i.v.; or METOCLOPRAMIDE 10mg; or ONDANSETRON 4mg | |
| Outcomes | Nausea; vomiting. Postop 0‐1; 1‐4; 4‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. No side effects. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; N; N | |
| Participants | 224 women; pregnant; caesarean; ASA1,2; post‐hoc exclusion | |
| Interventions | INTRAOPERATIVE Either: PLACEBO; or CYCLIZINE 50mg i.m.; or PROCHLORPERAZINE 12.5mg | |
| Outcomes | All outcomes. Postop 0‐8; 8‐24 hours. | |
| Notes | Side effects not recorded. Incidences not reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; N; N | |
| Participants | 225 women; hysterectomy; ASA1,2; 35‐55 (mean 45); exc' PONV, motion sickness/GI disease, antiemetic | |
| Interventions | Intraoperative end Either: PLACEBO; or DEXAMETHASONE 2.5 or 5 or 10mg i.v.; or DROPERIDOL 1.25mg | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Side effects: "restless with droperidol". Retching categorized as vomiting. Unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; N; N; N | |
| Participants | 132 children; 60 girls; tonsil +/‐ adenoids; ASA1,2; 2‐14 (mean 6); exc' previous PONV, GI ulcer/bleeding, HZV, hypertension, study drug allergy | |
| Interventions | Induction Either: TROPISETRON 0.1mg/kg i.v. (maximum 2mg) +/‐ DEXAMETHASONE 0.5mg/kg (maximum 8mg) | |
| Outcomes | All outcomes. Postop 0‐1; 0‐2; 0‐4; 0‐8; 0‐12 hours. | |
| Notes | Male/female incidences not reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; Y; Y; Y | |
| Participants | 60 adults; 29 women; ASA1,2; 15‐60 (mean 41); middle ear, exc' antiemetic | |
| Interventions | Preoperative AND induction Either: PLACEBO then GLYCOPYRRONIUM 0.2mg i.v.; or HYOSCINE patch then PLACEBO | |
| Outcomes | All outcomes. Postop 0‐2; 2‐6; 6‐12; 12‐18; 18‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Unclear if vomiters nauseated or categorized once or twice. I combined retching and vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 50 adults; 27 women; plastic; exc' antiemetic, pregnancy/breastfeeding, obese | |
| Interventions | Preoperative AND induction Either: PLACEBO then ATROPINE 100 mcg/kg i.v.; or SCOPOLAMINE patch 0.5mg then PLACEBO | |
| Outcomes | All outcomes. Postop 0‐2; 2‐6; 6‐12; 12‐18; 18‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. No side effects. Unclear if vomiters retched or if either nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 56 adults; 33 women; ENT; ASA1,2; 21‐55 (mean 40); exc' antiemetic/N&V | |
| Interventions | Preoperative Either: PLACEBO; or HYOSCINE 0.5mg | |
| Outcomes | All outcomes Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Categorized by severest symptom (vomiting>retching>nausea). Unclear if vomiters retched or if either nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 75 adults; ASA1,2; 15‐62; middle ear; exc' antiemetic or N&V | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 4 or 8mg i.v. | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 50 children; mean 10; ASA1,2; otoplasty; exc' obese, antiemetic | |
| Interventions | Preoperative AND induction Either: SCOPOLAMINE patch (0.25 or 0.5mg) then placebo; or PLACEBO then ATROPINE 10mcg/kg i.v. | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; N; N | |
| Participants | 54 children; 25 girls; ASA1,2; strabismus; 1‐11 (mean 6) | |
| Interventions | Preoperative Either: NO TREATMENT; or SCOPOLAMINE 0.38 mg patch | |
| Outcomes | Vomiting. Postop 0‐48 hours. | |
| Notes | Male/female incidences not reported. No side effects. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; N; N. | |
| Participants | 112 women; caesarean; ASA1,2 | |
| Interventions | Intraoperative Either: NO TREATMENT; or DROPERIDOL 2.5mg i.v. | |
| Outcomes | Nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 50 adults; dental ; no sex data | |
| Interventions | Preoperative Either: oral DIAZEPAM 10mg; or oral PENTOBARBITONE 100mg | |
| Outcomes | All outcomes. Postop 0‐1 hour. | |
| Notes | Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; N; N | |
| Participants | 162 women; hysterectomy; ASA1,2; 35‐65 (mean 49) | |
| Interventions | Intraoperative end Either: PLACEBO; or NEOSTIGMINE 2mg i.v. AND GYLCOPYRROLATE 0.4mg | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐1; 1‐2; 2‐3; 3‐9; 9‐15; 15‐21; 21‐27; 0‐27 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized. Unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; N; N; N | |
| Participants | 120 women; tubal ligation; ASA1,2; mean 34; exc' breast feeding, PONV, motion sickness, antiemetic | |
| Interventions | Intraoperative Either: PLACEBO; or DEXAMETHASONE 5mg i.v.; or METOCLOPRAMIDE 10mg | |
| Outcomes | All outcomes. Postop 0‐4; 4‐24; 0‐24 hours. | |
| Notes | No side effects. Retching categorized as vomiting. Unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 80 women; breast; 27‐69 (mean 50); ASA1,2 | |
| Interventions | Induction Either: METOCLOPRAMIDE 10mg i.v.; or ONDANSETRON 4mg | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 60 women; laparoscopies; ASA1,2; 18‐60 | |
| Interventions | Preoperative AND induction Either: PLACEBO then DROPERIDOL 1.25mg i.v. or GRANISETRON 10 microgram/kg; or oral GRANISETRON 1mg then PLACEBO | |
| Outcomes | See notes. | |
| Notes | Unclear how retching categorized or if vomiters nauseated or categorized once or twice. Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 72 adults; 39 women; dental; exc' allergy | |
| Interventions | Preoperative Either: PLACEBO; or METHYLPREDNISOLONE 40mg i.v. | |
| Outcomes | Nausea. Postop 0‐8 hours. | |
| Notes | Male/female incidences not reported. No side effects. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 30 adults; 20 women; hepato‐biliary; ASA1,2; mean 48; exc' liver/renal/cardiac/blood/endocrine disease, antiemetic | |
| Interventions | Preoperative Either: NO TREATMENT; or ONDANSETRON 4mg i.v. | |
| Outcomes | Nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. No side effects. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 45 women; gynaecological; 18‐63 (mean 38) | |
| Interventions | Induction AND intraoperative Either: NO TREATMENT then METOCLOPRAMIDE 10mg i.v.; or ONDANSETRON 4mg i.v. then NO TREATMENT; or both | |
| Outcomes | Nausea; vomiting. Postop 0‐24 hours. | |
| Notes | Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 40 adults; 13 women; abdominal and leg; ASA1,2; 16‐82 (mean 50); exc' cardiac/lung disease | |
| Interventions | Preoperative Either: oral DIAZEPAM 10mg; or sublingual FLUNITRAZEPAM 2mg | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop time unclear. | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 60 children; 32 girls; strabismus; ASA1,2; 3‐14 (mean 6); exc' N&V/antiemetic, trial drug, NG | |
| Interventions | Induction Either: PLACEBO; or DEXAMETHASONE 0.15mg/kg iv | |
| Outcomes | Vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 165 adults; 87 women; eye; 15‐87 (mean 60) | |
| Interventions | Postoperative Either: NO TREATMENT; or DROPERIDOL 5mg i.v. | |
| Outcomes | Nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 68 women; laparoscopies; ASA1,2; 18‐50 (mean 35) | |
| Interventions | Preoperative Either: NO TREATMENT; or oral TROPISETRON 5mg | |
| Outcomes | All outcomes. Postop 0‐3; 3‐24; 0‐24 hours. | |
| Notes | Side effects not recorded. Unclear if vomiters nauseated. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; N; Y; N | |
| Participants | 397 adults; 339 women; various surgeries; exc' ASA>3, antiemetic, pregnant/breastfeeding, glaucoma, study drug allergy, opiates/cytotoxic | |
| Interventions | Induction Either: DROPERIDOL 1.25mg i.v.; or GRANISETRON 1mg i.v. +/‐ DEXAMETHASONE 5mg | |
| Outcomes | All outcomes. Postop 0‐24; 72‐120 hours. | |
| Notes | Outcomes commonest 72‐120 hours. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 60 adults; 50 women; general/gynaecological; ASA1,2; 18‐75 (mean 45); exc' hypertension, IHD, depression, alcohol abuse, antidepressant, beta blocker, liver/renal disease | |
| Interventions | Intraoperative AND postoperative PCA. Either: PLACEBO twice; or CLONIDINE 4 mcg/kg i.v. then PCA bolus 20 mcg i.v. | |
| Outcomes | Nausea or vomiting. Postop 0‐12; 12‐24; 24‐36 hours. | |
| Notes | Outcomes commonest 0‐12 hours. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 102 adults; 61 women; middle ear; 18‐65 (mean 45); exc' gastropathy, obese | |
| Interventions | Induction Either: NO TREATMENT; or DROPERIDOL 25 mcg/kg i.v. | |
| Outcomes | Nausea; vomiting. Postop 0‐PACU discharge; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; Y; Y; Y | |
| Participants | 120 adults; 62 women; middle ear; ASA1,2; exc' DM, cholecystitis, neuropathy, NM disorder, pregnant, obese | |
| Interventions | Preoperative Either: PLACEBO; or DROPERIDOL 25 mcg/kg i.v.; or ONDANSETRON 4mg i.v. | |
| Outcomes | All outcomes. Postop 0‐2; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; N; N; N; N | |
| Participants | 120 adults; 36 women; laminectomy; mean 51; ASA1‐3; exc' pregnant, heart/liver/renal disease/bleeding abnormality | |
| Interventions | Induction Either: PLACEBO; or epidural CLONIDINE 150 mcg | |
| Outcomes | Nausea; vomiting. Postop 0‐1; 1‐25 hours. | |
| Notes | Outcomes commonest 1‐25 hours. Male/female incidences not reported. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; N; N; N | |
| Participants | 76 children; 29 female; tonsil +/‐ adenoids; ASA1,2; 2‐14 (mean 6) | |
| Interventions | Induction Either: PLACEBO; or TROPISETRON 0.2mg/kg (maximum 5mg) | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐2; 2‐4; 4‐8; 8‐12; 12‐24; 0‐24 hours. | |
| Notes | Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; Y; N; Y | |
| Participants | 120 women; lap' chole'; ASA1‐3; exc' pregnant/breastfeeding; Parkinson's, metabolic disease, antiemetic | |
| Interventions | Induction Either: DROPERIDOL 1.25mg i.v.; or TROPISETRON 5mg i.v. | |
| Outcomes | All outcomes. Postop 0‐2; 2‐24 hours. | |
| Notes | Unclear how retching categorized or if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 180 women; breast; ASA1‐3 | |
| Interventions | Intraoperative Either: NO TREATMENT; or ONDANSETRON 8mg i.v. | |
| Outcomes | All outcomes. Postop 0‐2; 2‐24; 0‐24 hours. | |
| Notes | Side effects "did not differ". Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; N; N | |
| Participants | 200 women; thyroid/parathyroid; ASA1‐3; mean 50 | |
| Interventions | Preoperative Either: oral METOCLOPRAMIDE 10mg; or ONDANSETRON 16mg; or TROPISETRON 5mg | |
| Outcomes | All outcomes. Postop 0‐2; 2‐24; 0‐24 hours. | |
| Notes | Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 60 adults; 44 female; various surgeries; ASA1,2 | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 10 or 20 mcg/kg i.v. | |
| Outcomes | Nausea or vomiting. Postop 0‐2 hours. | |
| Notes | Male/female incidences not reported. No side effects. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 150 adults; 126 women; thyroid; 19‐70 (mean 50); ASA1‐3; exc' obese, reflux, PONV/motion sickness, antiemetic, lung disease | |
| Interventions | Induction AND intraoperative AND postoperative Either: No treatment then 30% O2; or Droperidol 0.625mg iv then O2 30%; or air then 80% O2 | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐2; 2‐6; 6‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Retching categorized as vomiting. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 75 women; ERPC; ASA1; 17‐40 (mean 29); exc' hyperemesis, antiemetic, sedative/analgesic | |
| Interventions | INDUCTION Either: PLACEBO; or METOCLOPRAMIDE 10mg i.v.; or TIAPRIDE 100mg | |
| Outcomes | All outcomes. Postop 0‐1; 1‐6; 0‐6 hours. | |
| Notes | Nausea and vomiting commonest 0‐1 hour, rescue antiemetic 1‐6 hours. Unclear how retching treated or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 40 women; gynaecological; ASA1,2; 40‐65 years | |
| Interventions | Induction AND postoperative Either: METOCLOPRAMIDE 10mg i.v. thrice; ONDANSETRON 8mg thrice | |
| Outcomes | Nausea or vomiting. Postop 0‐24 hours. | |
| Notes | No side effects. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 47 adults; 21 female; 55‐72 (mean 60); exc' heart/liver/GI/renal disease | |
| Interventions | Induction AND postoperative Either: PLACEBO twice; or METOCLOPRAMIDE 1mg/kg i.v. then 1.5mg/kg | |
| Outcomes | Nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 62 adults; 36 female; middle ear; ASA1,2; 18‐60 (mean 34); exc' CNS/ear disease, motion sickness/PONV | |
| Interventions | Induction AND postoperative Either: NO TREATMENT or DEXAMETHASONE 5mg i.v. or ONDANSETRON 8mg then 50% O2 + 50% N2O; or NO TREATMENT then 75% O2 + 25% N2O | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐6; 6‐24 hours. | |
| Notes | Rescue antiemetic only 6‐24 hours. Nausea commonest 6‐24 hours. Male/female incidences not reported. No side effects. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 150 children; 74 female; various surgery; exc' antiemetics, DM, reflux | |
| Interventions | Preoperative Either: PLACEBO; or oral DOLASETRON 1.8mg/kg; or ONDANSETRON 0.15mg/kg iv | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐1; 1‐2; 0‐24 hours. | |
| Notes | Male/female incidences not reported. No side effects. I combined retching and vomiting. Unclear if retchers or vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; N; N; Y; Y | |
| Participants | 100 women; cataract; mean 72 | |
| Interventions | Preoperative Either: NO TREATMENT; or DROPERIDOL 0.04 mg/kg | |
| Outcomes | Nausea; vomiting. Postop 0‐8; 8‐14; 14‐20; 20‐26 hours. | |
| Notes | Nausea commonest 8‐14 hours, vomiting 0‐8 hours. No side effects. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 116 children; no sex data; strabismus; 2‐16 (mean 5) | |
| Interventions | Intraoperative Either: NO TREATMENT; or DIXYRAZINE 0.25mg/kg i.v. | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 84 adults; 37 female; ENT; exc' ASA>1 | |
| Interventions | Induction AND intraoperative Either NO TREATMENT twice; or DROPERIDOL 0.15mg/kg i.v. then METOPROLOL 0.1mg/kg | |
| Outcomes | Nausea or vomiting. Postop 0‐3 hours. | |
| Notes | Male/female incidences not reported. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y. | |
| Participants | 90 adults; no sex data; ASA1,2; craniotomy; 18‐72; exc' PONV/motion sickness | |
| Interventions | Intraoperative Either: PLACEBO; or ONDANSETRON 8mg i.v. | |
| Outcomes | Nausea; vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 100 children; 49 female; strabismus; ASA1,2; 1‐15 (mean 7); exc' motion sickness/PONV | |
| Interventions | Induction Either: PLACEBO; or METOCLOPRAMIDE 250 mcg/kg i.v.; or ONDANSETRON 150 mcg/kg; or METOCLOPRAMIDE 150 mcg/kg AND ONDANSETRON 100 mcg/kg | |
| Outcomes | Nausea or vomiting. Postop 0‐2; 2‐6; 6‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; N; N; N | |
| Participants | 170 adults; 58 female; craniotomy; ASA1,2; exc' motion sickness/PONV, VP shunt, antiemetic/N&V | |
| Interventions | Intraoperative Either: PLACEBO; or ONDANSETRON 4mg i.v. | |
| Outcomes | All outcomes. Postop 0‐6; 6‐24; 0‐24 hours. | |
| Notes | Unclear how retching categorized or if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 49 children; no sex data; ASA1‐3; 1‐17; ophthalmic | |
| Interventions | Intraoperative Either: PLACEBO; or METOCLOPRAMIDE 0.25mg/kg i.v.; or ONDANSETRON 0.15mg/kg | |
| Outcomes | Postop before discharge. | |
| Notes | Incidences not reported. Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; Y; Y; Y | |
| Participants | 286 adults; 156 female; orthopaedic; ASA1,2; exc' PONV, ASA>2, N&V | |
| Interventions | Postoperative AND PCA Either: PLACEBO twice; or DROPERIDOL 2.5mg i.v. then 0.125mg bolus; or METOCLOPRAMIDE 20mg, 1mg bolus; or TROPISETRON 5mg then PLACEBO | |
| Outcomes | Nausea or vomiting; rescue antiemetic. Postop 0‐10; 10‐18; 0‐18; 18‐36; 0‐36 hours. | |
| Notes | Male/female incidences not reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 241 adults; 133 female; elective surgery; ASA1,2; <65 years; mean 39; exc' liver/renal/CNS/endocrine disease, pregnant/lactating, study drug allergy, emesis, NG | |
| Interventions | Preoperative Either: PLACEBO; or oral METOCLOPRAMIDE 10mg; or ONDANSETRON 8mg | |
| Outcomes | Nausea; vomitiing; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Unclear how retching categorized or if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N, B, Y, N, Y, Y | |
| Participants | 182 women; orthopaedic; ASA1,2; 16‐60 (mean 36); exc' antiemetic, alcoholism, GI disease, DM | |
| Interventions | Intraoperative Either: PLACEBO; or ALIZAPRIDE 100 or 200 mg i.v.; or DROPERIDOL 1.25mg; or METOCLOPRAMIDE 20 mg | |
| Outcomes | All outcomes. Postop 0‐24 hours | |
| Notes | I combined retching and vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 90 adults; no sex data; inguinal/anal; ASA1,2; 18‐74 (mean 34) | |
| Interventions | Induction Either: PLACEBO; or intrathecal MORPHINE 100 or 200 mcg; or NEOSTIGMINE 100 or 200 mcg; or MORPHINE 50 mcg AND NEOSTIGMINE 50 mcg | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 129 children; strabismus; 63 female; 1‐13 (mean 6) | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 75 mcg/kg i.v.; or LORAZEPAM 10 mcg/kg | |
| Outcomes | Vomiting. Postop 0‐6; 6‐24 hours. | |
| Notes | Vomiting commonest 6‐24 hours. Male/female incidences not reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 41 children; no sex data; middle ear | |
| Interventions | Intraoperative Either: PLACEBO; or ONDANSETRON 0.15mg/kg (maximum 4mg) i.v.; or PROMETHAZINE 0.25mg/kg | |
| Outcomes | Vomiting. Postop 0‐2; 2‐24; 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. I combined retching and vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 45 adults; no sex data; middle ear | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 4mg i.v.; or PROMETHAZINE 25mg; or ONDANSETRON 2mg AND PROMETHAZINE 12.5mg | |
| Outcomes | See notes. | |
| Notes | Side effects not recorded. Unclear how retching categorized. Percentages of an unknown number. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 87 adults; 41 female; middle ear; ASA1,2; exc' obese, retardation | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 4mg i.v.; or PROMETHAZINE 12.5mg; or ONDANSETRON 2mg AND PROMETHAZINE 12.5mg | |
| Outcomes | All outcomes. Postop 0‐3; 0‐24 hours. | |
| Notes | Rescue antiemetic only 0‐3 hours. Male/female incidences not reported. No side effect. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 140 women; gynaecological; exc' pregnant, liver/CNS disease | |
| Interventions | Induction Either: NO TREATMENT; or ONDANSETRON 8mg i.v. | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | No side effect. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 38 adults; no sex data; ASA1,2; orthopaedic; exc' significant disease, antiemetic | |
| Interventions | Intraoperative Either: No treatment; or Atropine 1.2mg i.v. AND neostigmine 2.5mg | |
| Outcomes | All outcomes. Period: 0‐24 hours postoperatively. | |
| Notes | Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; N; N | |
| Participants | 80 women; gynaecological; ASA1,2; 18‐70 (mean 41); exc' pregnancy/lactation, Parkinsonism, GI disease, study drug allergy, antipsychotic, antihistamine | |
| Interventions | Induction AND postoperative Either: PLACEBO twice; or DROPERIDOL 1mg i.v. then PLACEBO/DROPERIDOL 0.02mg/ml or 0.04mg/ml PCA | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 100 children; 50 female; strabismus; 3‐10 (mean 6); ASA1,2 | |
| Interventions | Postoperative Either: PLACEBO; or DROPERIDOL 0.075mg/kg i.v.; or METOCLOPRAMIDE 0.15mg/kg | |
| Outcomes | Vomiting. Postop 0‐3; 0‐24 hours. | |
| Notes | Male/female incidences not reported. No side effects. Retching categorized as vomiting. Unclear if retching vomiters categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 100 children; 46 female; strabismus; 3‐12 (mean 6) | |
| Interventions | Induction AND postoperative Either: DROPERIDOL 75 mcg/kg +/‐ ATROPINE 10 mcg/kg i.v. then no treatment; or no treatment then DROPERIDOL 75 mcg/kg +/‐ ATROPINE 10 mcg/kg | |
| Outcomes | Vomiting. Postoperative 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 160 children; 78 female; ASA1,2; 4‐14 years; strabismus | |
| Interventions | Induction AND intraoperative Either: PLACEBO twice; or DROPERIDOL 75 mcg/kg i.v. then PLACEBO or ONDANSETRON 0.1 mg/kg; or ONDANSETRON 0.1 mg/kg then PLACEBO | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. No side effects. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 60 adults; 34 women; hip arthoplasty; exc' heart/GI/renal/extrapyramidal disease | |
| Interventions | Induction AND postoperative Either: PLACEBO twice; or METOCLOPRAMIDE 20mg i.m. twice | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐5 hours. | |
| Notes | Male/female incidences not reported. No side effects. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; A; Y; Y; Y; Y | |
| Participants | 63 adults; female 49; ASA1,2; exc' pregnant/breastfeeding, antiemetic, liver disease | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 4mg/kg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐1; 1‐2; 2‐24; 0‐24 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 94 women; laparoscopies; previous PONV; ASA 1‐3; exc' breastfeeding/pregnant, antiemetic, liver or Parkinson's disease | |
| Interventions | Intraoperative Either: DROPERIDOL or 1.25mg i.v. | |
| Outcomes | All outcomes. Postop 0‐2; 0‐24 hours. | |
| Notes | Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; N; Y; Y | |
| Participants | 439 women; laparoscopies; > 15 years; ASA1‐3; exc' pregnant/breastfeeding, liver/Parkinson's disease, antiemetic | |
| Interventions | INDUCTION Either: PLACEBO; or DROPERIDOL 1.25mg i.v.; or ONDANSETRON 8mg | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐2; 0‐24 hours. | |
| Notes | Retching categorized as vomiting. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; N; N; N | |
| Participants | 88 women; gynaecological; >18 years (mean 40); ASA1‐3; exc' pregnant/breastfeeding, liver/metabolic/Parkinson's disease, antiemetic | |
| Interventions | Intraoperative Either: ONDANSETRON 8mg i.v.; or TROPISETRON 5mg | |
| Outcomes | All outcomes. Postop 0‐2; 0‐24 hours. | |
| Notes | Nausea or vomiting only 0‐2 hours. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 60 children; 30 female; various surgeries; 5‐16 (mean 11); exc' epilepsy | |
| Interventions | Induction Either: PLACEBO; or DIXYRAZINE 0.25 mg/kg i.v. | |
| Outcomes | All outcomes. Postop 0‐2; 2‐24 hours. | |
| Notes | Outcomes commonest 2‐24 hours. Male/female incidences not reported. Side effects "dixyrazine sedative". Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 185 women; orthopaedic; ASA1,2; mean 42; exc' antiemetics | |
| Interventions | Intraoperative Either: PLACEBO; or DOMPERIDONE 5 or 10mg i.v.; or DROPERIDOL 1.25mg; or METOCLOPRAMIDE 10mg | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | No side effects. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 112 women; orthopaedic; ASA1,2; exc' antiemetic | |
| Interventions | Preoperative OR intraoperative Either: PLACEBO twice; or PLACEBO then DROPERIDOL 1.25mg i.v.; or DROPERIDOL 2.5mg i.m. then PLACEBO | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Side effects not recorded. I combined retching and vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 517 adults; 486 female; various surgeries; ASA1‐3; 18‐65 (mean 43); exc' pregnant, NG, antiemetic, cardiac/liver disease, obese, alcohol abuse | |
| Interventions | Induction Either: PLACEBO; or DOLASETRON 25 or 50mg i.v.; or ONDANSETRON 4mg | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 283 women; various surgeries; ASA1‐3; mean 22; exc' study drug allergy | |
| Interventions | Preoperative 12 hours for 48 hours Either: PLACEBO; or HYOSCINE patch (140 mcg then 5 mcg/hr) | |
| Outcomes | All outcomes Postop 0‐3; 3‐48 hours. | |
| Notes | Rescue antiemetic only 0‐3 hours. Others commonest 3‐48 hours. Unclear if vomiters nauseated or categorized once or twice. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; N; Y; Y | |
| Participants | 60 adults; 24 female; anal; ASA1,2 | |
| Interventions | Intraoperative AND postoperative Either; NO TREATMENT; or epidural BUTORPHANOL 0.85 mg/hr; or DROPERIDOL 0.11 mg/hr | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Unclear if vomiters and retchers nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; N; N; N | |
| Participants | 138 adults; 79 female; lap' chole'; ASA1‐3; mean 49 | |
| Interventions | Induction Either: DIMENHYDRINATE 50mg i.v.; or ONDANSETRON 4mg | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Retching categorized as vomiting. Unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 468 men; various surgeries; ASA1,2; exc' obese, NG, antiemetic, study drug | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 4mg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐2; 0‐24 hours. | |
| Notes | Retching categorized as vomiting. Unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 380 adults and children; no sex or age data; ENT/strabismus. | |
| Interventions | Induction AND intraoperative Either: PLACEBO twice; or METOCLOPRAMIDE 1mg/kg (max 50mg) i.v. then PLACEBO; or vice versa | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not reported. Unclear if vomiters nauseated or categorized once or twice. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 372 adults (increased risk) and children; no sex or age data; ENT/strabismus | |
| Interventions | Induction AND intraoperative Either: PLACEBO twice; or TROPISETRON 50 mcg/kg (max 2.5mg) i.v. then PLACEBO; or vice versa | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 61 children; 34 female; strabismus; 3‐14 (mean 6); ASA1,2 | |
| Interventions | Induction AND intraoperative Either: NO TREATMENT twice; or DROPERIDOL 0.075mg/kg i.v. then NO TREATMENT; or vice versa | |
| Outcomes | Nausea or vomiting. Postop 0‐4 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 150 adults; 91 women; various surgeries; exc' pregnant, study drug allergy, antiemetic | |
| Interventions | Intraoperative Either: PLACEBO; or DROPERIDOL 0.625mg i.v. | |
| Outcomes | All outcomes. Postop 0‐4; 4‐24 hours. | |
| Notes | Nausea or vomiting commonest 4‐24 hours. Male/female incidences not reported. Unclear how retching categorized or if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 58 adults; 33 women; tympanoplasty; excluded <18 >75, drug abuse, antiemetic, renal/liver disease, pregnant | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 4mg i.v. | |
| Outcomes | All outcomes. Postop 0‐1; 1‐2; 2‐4; 4‐8; 8‐24; 0‐24 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 100 adults; 49 women; eye; ASA1,2; exc' IOP>25mmHg, clonidine, calcium/betablocker | |
| Interventions | Preoperative 2 hours. Either: oral CLONIDINE 300 mcg/kg; or DIAZEPAM 0.15 mg/kg | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐6 hours, | |
| Notes | Male/female incidences not given. Unclear how retching categorized. Unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 30 women; gynaecological | |
| Interventions | Preoperative Either: METOCLOPRAMIDE 10mg i.v.; or ONDANSETRON 4mg | |
| Outcomes | Nausea; vomiting. Postop 0‐24 hours. | |
| Notes | Retching categorized as vomiting. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 154 children; no sex data; strabismus; ASA1,2; 1‐15 (mean 4) | |
| Interventions | Preoperative Either: PLACEBO; or oral DROPERIDOL 300 mcg/kg; or METOCLOPRAMIDE 0.15mg/kg; or both | |
| Outcomes | Vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; N; N | |
| Participants | 408 adults; 242 women; GI; ASA1,2; 18‐75 (mean 50); exc' N&V/antiemetic, pregnant/breastfeeding, >100kg, drug abuse, study drug allergy | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 4mg i.v. | |
| Outcomes | Nausea; vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not reported. Retching categorized as vomiting. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; Y; N; N | |
| Participants | 80 women; gynaecological; 18‐85 years; ASA1,2; exc' opioid, antiemetic, Parkinsonism, study drug allergy | |
| Interventions | Postoperative PCA Either: DROPERIDOL bolus 0.05 or 0.1 or 0.15 or 0.2 mg/ml | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Unclear how retching categorized. Patients categorized by severest symptom (vomiting>nausea). | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 26 adults; 24 female; thyroidectomy | |
| Interventions | Preoperative 16 AND 1 hour Either: PLACEBO twice; or oral LORAZEPAM 2.5mg twice | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐2; 2‐12; 0‐12 hours. | |
| Notes | Nausea and vomiting commonest 0‐2 hours. Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 90 adults; 49 female; various surgeries | |
| Interventions | Preoperative Either: PLACEBO; or DIXYRAZINE 0.5mg/kg i.m. | |
| Outcomes | Nausea or vomiting; rescue antiemetic. Postop 0‐6 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 61 children; no sex data; strabismus ; ASA1; 2‐14 (mean 7) | |
| Interventions | Intraoperative Either: NO TREATMENT; or DIXYRAZINE 0.25mg/kg i.v.; or DROPERIDOL 0.075mg/kg | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐2; 0‐4; 0‐6; 0‐8; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 100 adults; 85 women; cholecystectomy; mean 45 | |
| Interventions | Intraoperative Either: PLACEBO; or PHYSOSTIGMINE 2mg iv | |
| Outcomes | Nausea; vomiting. Postop 0‐10; 10‐20; 20‐40; 40‐60 minutes. | |
| Notes | Outcomes commonest 40‐60 minutes. Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; N; N | |
| Participants | 100 adults; 48 female; orthopaedic; ASA1,2; exc' analgesic/antiemetic, epilepsy | |
| Interventions | Preoperative Either: PLACEBO; or DROPERIDOL 0.5mg i.v.; or METOCLOPRAMIDE 10mg | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B;Y; N; N; N | |
| Participants | 100 women; hysterectomy; ASA1,2; mean 47; exc' analgesic/antiemetic, epilepsy, N&V | |
| Interventions | Preoperative Either: PLACEBO; or DROPERIDOL 0.5mg i.v.; or METOCLOPRAMIDE 10mg | |
| Outcomes | Nausea; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 60 adults; 23 female; orthopaedic; ASA1,2; mean 38 | |
| Interventions | Induction Either: PLACEBO; or intrathecal NEOSTIGMINE 25 or 50 or 100 mcg | |
| Outcomes | See notes. | |
| Notes | Incidences not reported. Male/female incidences not reported. Side effects not reported. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 92 women; hysterectomy; ASA1,2; mean 53; exc' >70 years, study drug allergy, bradycardia | |
| Interventions | Intraoperative Either; PLACEBO; or intrathecal NEOSTIGMINE 25 or 50 or 75 mcgs | |
| Outcomes | Rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 60 children; no sex data; ASA1,2; 1‐12; tonsils +/‐ adenoids | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 20 mcg/kg i.v.; or ONDANSETRON 0.15mg/kg | |
| Outcomes | Vomiting. PACU. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 60 children; no sex data; strabismus; ASA1,2 | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 20 mcg/kg i.v.; or ONDANSETRON 0.15mg/kg | |
| Outcomes | Vomiting. PACU. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; N; N | |
| Participants | 165 children; no sex data; adenotonsillectomy; ASA1,2; 1‐12 (mean 5) | |
| Interventions | Preoperative AND intraoperative Either: PLACEBO then DROPERIDOL mcg/kg i.v. or ONDANSETRON 0.15mg/kg; or oral METOCLOPRAMIDE 0.15mg/kg then PLACEBO or DROPERIDOL or ONDANSETRON | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐3; 3‐24 hours. | |
| Notes | Vomiting commonest 3‐24 hours. Male/female incidences not recorded. Side effects not recorded. Unclear how many in each group. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; N; Y; Y | |
| Participants | 320 children; tonsil + adenoids, strabismus; exc' PONV, motion sickness, antiemetic | |
| Interventions | Preoperative Either: NO TREATMENT or oral METOCLOPRAMIDE 0.15mg/kg AND PLACEBO or ONDANSETRON 0.05 or 0.1 or 0.15mg/kg i.v. | |
| Outcomes | Vomiting. Postop 0‐3; 3‐24 hours. | |
| Notes | Vomiting commonest 3‐24 hours. Male/female incidences not recorded. Side effects not recorded. Unclear how many in each group. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; N; Y; Y | |
| Participants | 50 adults; various surgeries; 1 female; ASA1,2; exc' fertile women, >100kg | |
| Interventions | Preoperative Either: PLACEBO; or DEXMEDETOMIDINE 2 mcg/kg i.v. | |
| Outcomes | Rescue antiemetic. Postop 0‐3 hours. | |
| Notes | Incidence not reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 60 children; no sex data; <33kg; 1‐9; ENT/general. | |
| Interventions | Preoperative Either: NO TREATMENT; or DROPERIDOL 0.2mg/kg oral | |
| Outcomes | Vomiting. Postop time unclear. | |
| Notes | Male/female incidences not recorded. Side effects "no extrapyramidal". Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 35 adults; 19 female; 21‐69 years; middle ear | |
| Interventions | Preoperative Either: PLACEBO; or SCOPOLAMINE patch | |
| Outcomes | Nausea; vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 45 women; gynaecological | |
| Interventions | Induction AND intraoperative Either: PLACEBO twice; or ONDANSETRON 4 or 8mg i.v. twice | |
| Outcomes | Nausea; rescue antiemetic. Postop 0‐30 hours. | |
| Notes | Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 135 women; thyroidectomy; ASA1,2; exc' motion sickness, GI disease, antiemetic, obese, smoker | |
| Interventions | Induction Either: PLACEBO; or DEXAMETHASONE 5 or 8mg i.v. | |
| Outcomes | All outcomes. Postop 0‐2; 2‐24; 0‐24 hours. | |
| Notes | No side effects. Unclear if vomiters nauseated. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 113 adults; 104 women; thyroid; ASA1,2; 18‐65 (mean 40); exc' obese, GI disease, antiemetic | |
| Interventions | Induction Either: PLACEBO; or GRANISETRON 20 mcg/kg iv; or RAMOSETRON 4 mcg/kg | |
| Outcomes | All outcomes. Postop 0‐1; 1‐2; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Retching categorized as vomiting. Categorized by severest symptom (vomiting > nausea). Unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; N; N; N | |
| Participants | 90 women; orthopaedic; ASA1,2; 28‐69 (mean 43); exc' GI disease, liver/renal disease, antiemetic, N&V, obese, smoker | |
| Interventions | Induction Either: PLACEBO; or DEXAMETHASONE 8mg iv | |
| Outcomes | All outcomes Postop 0‐6; 6‐12; 12‐24; 0‐24 hours. | |
| Notes | No side effects. Unclear how retching categorized or if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 168 women; laparoscopies; 28‐58 (mean 39); ASA1,2; exc' pregnant, obese, smoker, steroid, GI/liver/renal/ear disease, N&V, antiemetic | |
| Interventions | Induction Either: PLACEBO; or DEXAMETHASONE 8mg iv | |
| Outcomes | All outcomes. Postop 0‐2; 0‐6; 6‐12; 12‐18; 0‐24 hours. | |
| Notes | No side effects. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 84 women; gynaecological; 18‐65 (mean 44); exc' pregnant/breastfeeding, renal/hepatic/heart/metabolic/endocrine disease, antiemetic | |
| Interventions | Preoperative AND postoperative Either: PLACEBO twice; or oral ONDANSETRON 8mg twice | |
| Outcomes | All outcomes. Postop 0‐1; 0‐24 hours. | |
| Notes | No side effects. Retching categorized as nausea. Vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 82 adults; 61 female; ASA1,2; 12‐75 (mean 50); GI; exc' obese, ASA>2, N&V/antiemetic, pregnant/breastfeeding, drug abuse, study drug allergy | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 4mg i.v. | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; N; N | |
| Participants | 60 adults; no sex data; thyroid/breast | |
| Interventions | Postoperative Either: PLACEBO; or ONDANSETRON 4mg i.v. +/‐ DEXAMETHASONE 8mg | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Side effects not recorded. Incidences not reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; N; N | |
| Participants | 100 adults; 35 female; various surgeries; ASA1,2; exc' CNS, NM, renal, liver disease, BMI <20 >30, pregnant | |
| Interventions | Intraoperative Either: PLACEBO; or NEOSTIGMINE AND GLYCOPYRROLATE (10 mcg/kg AND 2.5 mcg/kg i.v.; or double or quadruple doses). | |
| Outcomes | Nausea or vomiting. Postop 0‐1 hour. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; N; Y; N; N | |
| Participants | 60 women; <70 years; ASA1,2; hysterectomy; exc' psychiatric, antiemetic | |
| Interventions | Preoperative Either: oral METOCLOPRAMIDE 10mg; or oral NABILONE 2mg | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop about 4 hours; 4‐24 hours. | |
| Notes | Outcomes commonest 4‐24 hours. No side effects. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; Y; N; N | |
| Participants | 100 adults; 66 women; lap' chole'; mean 34; exc' < 15 >65, pregnant | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 4mg i.v. | |
| Outcomes | Nausea; vomiting. Postop 0‐12 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; N; N; Y; Y | |
| Participants | 325 women; STOP; ASA1,2; exc' antiemetic/N&V, ergometrine | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 0.25 or 1.25 or 2.5mg i.v.; or METOCLOPRAMIDE 10 mg | |
| Outcomes | Nausea or vomiting. Postop 0‐6 hours. | |
| Notes | Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; N; N | |
| Participants | 228 women; laparoscopies; ASA1,2 | |
| Interventions | Intraoperative Either: DROPERIDOL 10 or 20 mcg/kg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐7; 7‐24 hours. | |
| Notes | Rescue antiemetic only 0‐7 hours, nausea 7‐24 hours. Vomiting commonest 0‐7hours. Retching categorized with vomiting. Unclear if nauseated vomiters categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 110 children; 57 female; strabismus; mean 5; ASA1,2 | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 0.075mg/kg i.v.; or METOCLOPRAMIDE 0.15 or 0.25 mg/kg | |
| Outcomes | Vomiting. Postop 0‐3 hours. | |
| Notes | Male/female incidences not reported. No side effects. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; Y; Y; Y | |
| Participants | 188 women; gynaecological. | |
| Interventions | Intraoperative Either: METOCLOPRAMIDE 10mg i.m.; or PERPHENAZINE 5mg i.m. | |
| Outcomes | Vomiting. Postop 0‐6 hours. | |
| Notes | Side effects "metoclopramide sedating". Retching categorized as vomiting. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; N; N | |
| Participants | 67 children; 34 girls; ASA1,2; mean 7; tonsil +/‐ adenoids; exc' motion sickness/PONV | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 0.15mg/kg (to 8mg) i.v. | |
| Outcomes | Vomiting. Postop 0‐24; 24‐48 hours. | |
| Notes | Vomiting commonest 0‐24 hours. Male/female incidences not reported. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; A; Y; Y; Y; Y | |
| Participants | 57 children; 23 female; strabismus; ASA1,2; 3‐14 (mean 6); exc' GI dysmotility | |
| Interventions | Induction Either: DROPERIDOL 0.075mg/kg i.v.; or ONDANSETRON 0.15mg/kg | |
| Outcomes | Vomiting. Postop 0‐24; 24‐48 hours. | |
| Notes | Vomiting commonest 0‐48 hours. Male/female incidences not reported. Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 150 women; gynaecological; ASA1,2; exc' motion sickness/PONV, GI disease, menstrual, hormone | |
| Interventions | Induction Either: PLACEBO; or DEXAMETHASONE 1.25 or 2.5 or 5 or 10mg i.v. | |
| Outcomes | Vomiting; rescue antiemetic Postop 0‐24 hours. | |
| Notes | No side effects. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 80 adults; 47 female; middle ear; ASA1,2; exc' antiemetic, GI disease, PONV | |
| Interventions | Induction Either: PLACEBO; or DEXAMETHASONE 10mg i.v. | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. No side effects. Retching categorized as vomiting. Categorized by severest symptom (vomiting>nausea). | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; N; N; N | |
| Participants | 22 women; gynaecological; ASA1; 21‐64 | |
| Interventions | Preoperative Either: PLACEBO; or LORAZEPAM 2mg oral | |
| Outcomes | Nausea; vomiting. Postop 0‐6 hours. | |
| Notes | Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 73 women; breast; mean 50; exc' study drug allergy, liver/renal/cardiac/CNS/GI disease, pregnant, drug abuse, antiemetic | |
| Interventions | Intraoperative Either: DOLASETRON 50mg iv; or DROPERIDOL 1mg | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 80 women; STOP; ASA1; mean 34; exc' pessary, N&V | |
| Interventions | Induction Either: NO TREATMENT; or METOCLOPRAMIDE 10mg i.v. | |
| Outcomes | All outcomes. Postop 0‐1; 1‐2 hours. | |
| Notes | Outcomes commonest 0‐1 hour. No side effects. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 32 women; gynaecological | |
| Interventions | Preoperative Either: PLACEBO; or HYOSCINE patch (1.5mg / 3 days) | |
| Outcomes | Nausea. Postop 0‐24 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 100 women; gynaecological; ASA1,2; 18‐65 (mean 47); exc' opioid, NSAID, steroid/antiemetic, study drug allergy | |
| Interventions | Induction Either: PLACEBO; or DEXAMETHASONE 8mg i.v.; or ONDANSETRON 4mg; or both | |
| Outcomes | All outcomes. Postop 0‐2; 0‐12; 0‐24; 0‐48 hours | |
| Notes | Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; N; N; N | |
| Participants | 90 women; laparoscopies; ASA1,2; 18‐60 (mean 37); exc' pregnant, antiemetic/analgesic/bambuterol, >100kg | |
| Interventions | Intraoperative Either: PLACEBO; or NEOSTIGMINE 50 mcg/kg AND GLYCOPYRROLATE 10 mcg/kg | |
| Outcomes | All outcomes. Postop 0‐6; 6‐24; 0‐24 hours. | |
| Notes | Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; N; N; N | |
| Participants | 270 children; various surgeries; 31 female; 1‐15 (mean 5) | |
| Interventions | Induction Either: NO TREATMENT; or DROPERIDOL 20 mcg/kg i.v. | |
| Outcomes | Vomiting. Postop 0‐6; 6‐24 hours. | |
| Notes | Vomiting commonest 6‐24 hours. Male/female incidences not recorded. Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 40 adults; laparoscopies; 33 female; ASA1,2; mean 42 | |
| Interventions | Preoperative AND postoperative twice Either: ONDANSETRON 4mg i.v. thrice; or TROPISETRON 5mg i.v. then NO TREATMENT twice | |
| Outcomes | Nausea; vomiting. Postop 0‐8 hours. | |
| Notes | Male/female incidences not reported. No side effects. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; Y; Y; Y | |
| Participants | 120 children; 58 female; strabismus; ASA1,2; 1‐15 (mean 6); exc' motion sickness/PONV | |
| Interventions | Induction AND intraoperative Either: ONDANSETRON 100 mcg/kg i.v. then PLACEBO; or vice versa. | |
| Outcomes | Vomiting; rescue antiemetics. Postop 0‐2; 2‐6; 6‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not reported. Retching not categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; N; N | |
| Participants | 200 women; gynaecological; ASA1,2; 16‐70 (mean 39); exc' antiemetic | |
| Interventions | Intraoperative Either: PLACEBO; or DOMPERIDONE 20mg i.v.; or DROPERIDOL 2.5mg; or METOCLOPRAMIDE 10mg | |
| Outcomes | Nausea or vomiting. Postop 0‐6; 0‐24 hours. | |
| Notes | Unclear if 'nausea and vomiting' is 'nausea or vomiting'. Unclear if nauseated vomiters categorized once or twice. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 201 women; gynaecological; ASA1,2; 16‐70 (mean 36); exc' antiemetic | |
| Interventions | Induction Either: PLACEBO; or DOMPERIDONE 20mg i.v.; or DROPERIDOL 2.5mg; or METOCLOPRAMIDE 10mg | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐1; 0‐2; 0‐3; 0‐4 hours. | |
| Notes | Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; N; N | |
| Participants | 65 adults; 31 women; craniectomy; 18‐76 (mean 44); ASA1‐3; exc' N&V, antiemetic, study drug allergy, pregnant/breastfeeding, obese, psychiatric | |
| Interventions | Intraoperative Either: PLACEBO; or TROPISETRON 2mg iv | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐2; 2‐12; 12‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Retching categorized as vomiting. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 153 women; gynaecological; ASA1,2; exc' postmenopausal, ectopic, reflux | |
| Interventions | Preoperative Either: PLACEBO; or oral METOCLOPRAMIDE 10mg; or ONDANSETRON 4mg | |
| Outcomes | All outcomes. Postop 0‐48 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 60 children; 31 female; strabismus | |
| Interventions | Intraoperative Either: DROPERIDOL 0.05mg/kg i.v.; or METOCLOPRAMIDE 0.15mg/kg; or ONDANSETRON 0.15mg/kg | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not reported. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 200 children; 109 girls; strabismus; exc' premature, obese, GI/liver disease, DM, inner ear disease | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 0.075mg/kg i.v; or ONDANSETRON 0.4mg/kg | |
| Outcomes | Vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 40 women; various surgeries; ASA1,2; exc' drugs, drug abuse, pregnant, renal disease | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 8mg i.v. | |
| Outcomes | Nausea; vomiting. Postop 0‐24 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 120 adults; 104 female; plastic | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 4mg i.v. | |
| Outcomes | Nausea; vomiting. | |
| Notes | Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 60 adults; no sex data; ASA1,2; 17‐59; various surgeries; exc' antiemetic, cold, ear infection | |
| Interventions | Intraoperative Either: NO TREATMENT; or DROPERIDOL 0.015 mg/kg i.v. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 to 72 hours. | |
| Notes | Male/female incidences not reported. I combined retching and vomiting. Unclear if vomiters retched or nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; N; N; Y; Y | |
| Participants | 105 adults; 92 female; urological/gynaecological; ASA1,2; 18‐65; exc' ASA>3 | |
| Interventions | Preoperative AND induction Either: NO TREATMENT twice; or NO TREATMENT then ONDANSETRON 4mg i.v.; or PROMETHAZINE 25mg i.m. then NO TREATMENT | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 40 children; 20 female; dental; 3‐5 (mean 4) | |
| Interventions | Preoperative Either: oral DIAZEPAM 0.3mg/kg; or nasal MIDAZOLAM 0.3mg/kg | |
| Outcomes | Vomiting. Postop time unclear. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 52 women; gynaecological; ASA1,2; 19‐45 (mean 31) | |
| Interventions | Preoperative twice Either: oral PLACEBO twice; or oral METOCLOPRAMIDE 20mg twice; or NO TREATMENT twice | |
| Outcomes | All outcomes. Postop 0‐1; 1‐2; 2‐3; 3‐4; 0‐24 hours. | |
| Notes | Nausea and rescue antiemetic commonest 2‐3 hours, vomiting 0‐1 hour. Side effects not reported. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 102 women; STOP | |
| Interventions | Induction Either: PLACEBO; or DIAZEPAM 10mg i.v. | |
| Outcomes | Nausea; vomiting. Postop time unclear. | |
| Notes | Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 109 women; STOP/D&C; 14‐49; exc' psychiatric | |
| Interventions | Induction Either: PLACEBO; or DIAZEPAM 10 mg i.v. | |
| Outcomes | Nausea; vomiting. Postop 0‐3 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; N; N | |
| Participants | 90 women; gyneacological/general; exc' poor risk | |
| Interventions | Intraoperative Either: DIAZEPAM 10mg i.v.; or FLUNITRAZEPAM 1mg | |
| Outcomes | All outcomes. Postop time unclear. | |
| Notes | Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 41 adults; no sex data; 65‐85; laparotomy | |
| Interventions | Intraoperative twice Either: PLACEBO twice; or DIAZEPAM 5mg twice | |
| Outcomes | Nausea; vomiting. Postop time unclear. | |
| Notes | Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 113 women; gynaecological | |
| Interventions | Induction Either: PLACEBO; or DIAZEPAM 10mg i.v. | |
| Outcomes | Nausea; vomiting. Postop 0‐24 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; N; N | |
| Participants | 52 men; prostatectomy; ASA1‐3; mean 62; exc' clonidine, study drug allergy, cardiac disease, alcohol abuse, psychotropic/analgesic | |
| Interventions | Preoperative Either: oral PLACEBO; or CLONIDINE 3 mcg/kg | |
| Outcomes | Nausea. Postop 0‐48 hours. | |
| Notes | The incidence not reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 32 children; no sex data; strabismus; ASA1; median 10 | |
| Interventions | Preoperative AND induction AND postoperative twice Either: NO TREATMENT then PROMETAZINE 0.5mg/kg i.m. then oral PROMETAZINE 3.3 or 6.6mg/kg twice; or ONDANSETRON 0.1mg/kg i.v. then NO TREATMENT then ONDANSETRON 0.1mg/kg i.v. twice | |
| Outcomes | Nausea; vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Unclear how retching categorized. Second postoperative ondansetron not given if asymptomatic. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 50 children; no sex data; strabismus; ASA1,2 | |
| Interventions | Intraoperative Either: PLACEBO; or GRANISETRON 10 or 40 mcg/kg i.v. | |
| Outcomes | Vomiting. Postop hospital; home; 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 104 adults/children; 50 female; burns; 2‐25 (mean 12) | |
| Interventions | Intraoperative AND postoperative Either: PLACEBO twice; or DIMENHYDRINATE 0.5mg/kg i.v. twice; or ONDANSETRON 0.1mg/kg twice | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 54 adults; 24 women; hand; mean 44; ASA1,2; exc' Raynaud's, sickle cell, study drug allergy, conduction abnormality, COPD | |
| Interventions | Induction Either: PLACEBO; or NEOSTIGMINE 1mg iv | |
| Outcomes | Nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; N; Y; Y | |
| Participants | 150 women; STOP; exc' ASA>2, epilepsy, renal/liver disease | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 2.5mg i.m.; or HYDROXYZINE 100mg i.m. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐3 hours. | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 207 women; various surgeries; ASA1‐3; 18‐65 (mean 39); exc' antiemetic, obese, liver disease, NG | |
| Interventions | Induction AND postoperative Either: PLACEBO twice; or ONDANSETRON 8mg i.v. twice | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Retching categorized as vomiting. Unclear if vomiters/retchers nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 180 women; gynaecological; ASA1,2; 18‐70; antiemetics, study drug allergy, NG | |
| Interventions | Induction Either: PLACEBO; or DEXAMETHASONE 8mg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐2; 0‐4; 0‐24 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; Y; Y; Y | |
| Participants | 60 women; hysterectomy; ASA1,2; mean 43; exc' NG, study drug contraindicated | |
| Interventions | Postoperative PCA Either: PLACEBO; or DROPERIDOL 0.5 or 1mg/30ml | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | No side effects. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; N; Y | |
| Participants | 120 women; laparoscopies; ASA1,2 | |
| Interventions | Intraoperative Either: PLACEBO; or ONDANSETRON 4mg i.v. | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Side effects "droperidol sedative". Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 80 women; gynaecological; ASA1‐3; exc' DM, antiemetic, NG, study drug allergy | |
| Interventions | Induction Either: (PLACEBO or DEXAMETHASONE 20mg i.v.) AND ONDANSETRON 4mg | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 100 adults; 72 female; various surgeries; exc' sedative/analgesic, study drug allergy | |
| Interventions | Induction Either: LIDOCAINE 10mg i.v.; or METOCLOPRAMIDE 20mg | |
| Outcomes | Nausea; vomiting. Postop 0‐1 hour. | |
| Notes | Male/female incidences not reported. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; N; N; N; N | |
| Participants | 45 children; no sex data; hernia; 1‐5 (mean 3); ASA1; exc' caudal contraindicated, CNS disease | |
| Interventions | Induction Either: PLACEBO; or caudal NEOSTIGMINE 1 mcg/kg | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not recorded. No side effects. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 80 women; gynaecological | |
| Interventions | Postoperative Either PLACEBO; or CLEBOPRIDE 1mg i.v. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐6 hours. | |
| Notes | Unclear how retching categorized. Unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 140 children; 84 girls; strabismus; 3‐12 (mean 7); ASA1; exc' GI disease, obese, previous operation | |
| Interventions | Preoperative Either: PLACEBO; or oral CLONIDINE 2 or 4 mcg/kg; or oral DIAZEPAM 0.4mg/kg | |
| Outcomes | Vomiting. Postop (time unclear) | |
| Notes | Male/female incidences not recorded. Retching not categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 120 women; gynaecologic; ASA1; 22‐65 (mean 45); exc' antiemetic, GI disease, pregnant, obese, NG | |
| Interventions | Intraoperative Either: PLACEBO; or GRANISETRON 20 or 40 microgram/kg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Retching categorized as vomiting. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 90 children; no sex data; ASA1; 2‐11 (mean 6); exc' ASA>1, obese, previous operation, GI disease | |
| Interventions | Induction Either: PLACEBO; or FLURBIPROFEN 0.5 or 1 mg/kg i.v. | |
| Outcomes | Vomiting. Postop 0‐8 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Retching not categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 200 women; gynaecological; ASA1; 20‐67 (mean 46); exc' antiemetic, GI disease, pregnant, obese, liver/renal disease, NG | |
| Interventions | Induction Either: PLACEBO; or GRANISETRON 2 or 5 or 10 or 20 mcg/kg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Side effects not reported. Retching categorized as vomiting. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; N; N | |
| Participants | 105 adults; no sex data; ASA1‐3; various surgeries; exc' obese, NG, motion sickness/PONV, antiemetic, pregnancy | |
| Interventions | Preoperative Either: DROPERIDOL 1.25mg i.v.; or ONDANSETRON 4mg | |
| Outcomes | Unclear. Postop unclear. | |
| Notes | Male/female incidences not recorded. Unclear number per group or how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; N; N | |
| Participants | 150 women; STOP; ASA1,2; exc' antiemetics, drugs interacting with droperidol | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 0.25 or 0.5mg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetics. Postop 0‐24 hours. | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; N; N; Y; Y | |
| Participants | 101 women; gynaecological; exc' GI complaints | |
| Interventions | Preoperative Either: NO TREATMENT; or oral METOCLOPRAMIDE 10mg | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop time unclear. | |
| Notes | Side effects not recorded. Unclear how retching categorized. All vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; N; N | |
| Participants | 142 women; hysterectomy; exc' PONV, study drug allergy, opioid, liver disease | |
| Interventions | Induction AND postoperative Either: DROPERIDOL 0.5mg i.v. then 3mg/60ml PCA; or ONDANSETRON 4mg then 8mg/60ml PCA | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; A; Y; N; Y; Y | |
| Participants | 30 children; 15 female; strabismus; ASA1; 4‐12 (mean 10) | |
| Interventions | Preoperative AND postoperative twice Either; oral METOCLOPRAMIDE 5mg thrice; or oral ONDANSETRON 4mg thrice | |
| Outcomes | Nausea; vomiting. Postop 0‐2; 2‐12; 12‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Retching not categorized as vomiting. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; N; Y | |
| Participants | 208 adults; 144 women; laparoscopies; 18‐75 (mean 47); exc' antiemetic/N&V, NG, <45kg >100kg, pregnancy/breastfeeding | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 4mg i.v. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 120 women; gynaecological; ASA1‐3; mean age 38; exc' analgesic, pregnant/breastfeeding, obese or thin, post hoc NG | |
| Interventions | Intraoperative end Either: PLACEBO; or METHYLNALTREXONE 20mg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetic Postop 0‐6 hours. | |
| Notes | Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 30 adults; 23 women; orthopaedic; 50‐80 (mean 70); exc' hypertensive, heart/lung/kidney/CNS disease, alcoholism, psychoactive drug | |
| Interventions | Preoperative AND intraoperative Either: ATROPINE 0.01 mg/kg i.m. then 1mg i.v.; or GLYCOPYRROLATE 5 mcg/kg i.m. then 0.5 mg i.v. | |
| Outcomes | Nausea or vomiting. Postop 0‐3 hours. | |
| Notes | Male/female incidences not reported. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; N; N | |
| Participants | 96 women; gynaecological; exc' multiple pregnancy, hydatiform, antiemetic, allergy, epilepsy, liver/renal disease | |
| Interventions | Induction Either: METOCLOPRAMIDE 0.4mg/kg i.v.; or ONDANSETRON 4mg | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐1; 1‐discharge; discharge‐24 hours. | |
| Notes | Nausea commonest 1 hour‐discharge, vomiting discharge‐24 hours, rescue antiemetic 0‐1 hour. Side effects not reported. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 200 women; gynaecological; ASA1,2; mean 35; exc' study drug allergy, psychiatric, pregnancy, antiemetic | |
| Interventions | Intraoperative Either: PLACEBO; or DROPERIDOL 0.625 or 1.25 or 2.5mg i.v. | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | No side effects. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; N; N | |
| Participants | 1074 women; gynaecological; exc' propofol, pregnant/breastfeeding, >100kg, ASA>3, antiemetic, NG | |
| Interventions | Induction Either: PLACEBO; or METOCLOPRAMIDE 10mg i.v.; or ONDANSETRON 4mg | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Retching categorized as vomiting. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 300 women; gynaecological; exc' >70 years, asthma/COPD, caesarean | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 2.5 or 5mg i.v. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 427 children; female 217; tonsil +/‐ adenoids; ASA1,2; 2‐12 (mean 6) | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 0.1mg/kg (to 4mg) | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Unclear if vomiters nauseated or categorized once or twice. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 50 adults; various surgeries; no sex data; ASA1‐3; 18‐78 (mean 42) | |
| Interventions | Intraoperative Either: PLACEBO; or epidural SCOPOLAMINE 0.25mg | |
| Outcomes | Nausea; vomiting. Postop 0‐2; 2‐4; 4‐6; 6‐24 hours. | |
| Notes | Nausea and vomiting commonest 0‐2 hours. Male/female incidences not reported. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 56 women; laparoscopies | |
| Interventions | Induction Either: PLACEBO; or METOCLOPRAMIDE 10mg i.v. | |
| Outcomes | Postop recovery (time unclear). | |
| Notes | Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; N; N | |
| Participants | 76 children; 39 female; strabismus; ASA1,2; 1‐12 (mean 5); exc' allergy, antiemetic | |
| Interventions | Preoperative Either: PLACEBO; or oral GRANISETRON 20 or 40 mcg/kg | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐3; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; N; N; N | |
| Participants | 60 children; 21 girls; appendicectomy; 5‐13 (mean 10) | |
| Interventions | Intraoperative AND postoperative PCA Either: NO TREATMENT then PLACEBO; or DROPERIDOL 10 mcg/kg then 0.2 mcg/kg/ml; or ONDANSETRON 100 mcg/kg then ONDANSETRON 2 mcg/kg/ml | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. No side effects. Unclear how retching categorized. Unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 40 adults; 19 female; eye; ASA1,2; mean 30; exc' <18 years | |
| Interventions | Preoperative Either: oral CLONIDINE 5 mcg/kg; or oral MIDAZOLAM 100 mcg/kg | |
| Outcomes | Vomiting. Postop (time unclear) | |
| Notes | Male/female incidences not reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 132 adults; 108 women; lap' chole'; 21‐68 (mean 37); ASA1,2; 40‐101 (mean 73) kg; exc', antiemetic/N&V | |
| Interventions | Induction Either: PLACEBO; or GRANISETRON 3mg i.v.; or METOCLOPRAMIDE 10mg; or ONDANSETRON 4mg; or TROPISETRON 5mg | |
| Outcomes | Nausea or vomiting; rescue antiemetic. Postop 0‐24 horus. | |
| Notes | Male/female incidences not reported. Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 90 adults; 65 women; mean age 30; laparoscopies; ASA1,2; exc' hypertension, heart/GI/CNS disease, drugs affecting LOS tone | |
| Interventions | Intraoperative end before reversal Either: PLACEBO i.v. and NO TREATMENT i.m.; or EPHEDRINE 0.5 mg/kg i.m. and NO TREATMENT i.v.; or PROPOFOL 0.25 mg/kg i.v. and NO TREATMENT i.m. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear if retchers and vomiters nauseated or if vomiters retched. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 45 adults; 28 women; various surgeries; 25‐60 (mean 40) | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 2.5mg i.v.; or GRANISETRON 3mg | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not recorded. No side effects. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; N; N; Y; Y | |
| Participants | 90 adults; 81 female; lap' chole' | |
| Interventions | Preoperative Either: NO TREATMENT; or PLACEBO ; or oral ONDANSETRON 8mg | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐6; 6‐12; 12‐24; >24 hours. | |
| Notes | Male/female incidences not reported. No side effects. Unclear how retching categorized. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; N; N; N | |
| Participants | 120 adults; 55 women; general surgery; 16‐70 (mean 57); ASA1,2; exc' opioids, convulsions, Parkinson's, drug abuse, psychiatric problems | |
| Interventions | Intraoperative AND postoperative Either: PLACEBO twice; or DROPERIDOL 2.5mg i.v. then PLACEBO or epidural DROPERIDOL 2.5mg; or PLACEBO then epidural DROPERIDOL 2.5mg/day | |
| Outcomes | Nausea or vomiting; rescue antiemetic. Postop 0‐72 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 120 children; no sex data; tonsil + adenoids; 4‐14; exc' ASA>1, steroid, infection | |
| Interventions | Preoperative Either: PLACEBO; or DEXAMETHASONE 1mg/kg (max 16mg) i.v. | |
| Outcomes | Vomiting. Postop 0‐6 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; N; N | |
| Participants | 100 women; laparoscopy; ASA 1; exc' BMI>27, study drug allergy | |
| Interventions | Intraoperative Either: PLACEBO; or GLYCOPYRROLATE 0.4mg AND NEOSTIGMINE 2mg | |
| Outcomes | Nausea or vomiting; rescue antiemetic. Postop 3; 0‐24 hours. | |
| Notes | Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 34 adults; 17 women; colorectal/head & neck; exc' <18, ASA>3, drug abuse, pregnant/breastfeeding | |
| Interventions | Postoperative PCA Either: NO TREATMENT; or DROPERIDOL 0.1mg i.v./bolus | |
| Outcomes | Rescue antiemetic. Postop 0‐48 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 65 children; 23 girls; strabismus; 1‐16 (mean 5); ASA1,2 | |
| Interventions | Preoperative Either: oral DIAZEPAM 0.15 mg/kg; or DROPERIDOL 50 or 75 mcg/kg | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐1; 0‐7; 7‐24; 0‐24 hours. | |
| Notes | Outcomes commonest 0‐7 hours. Male/female incidences not reported. Side effect "droperidol sedative, no extrapyramidal". Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; Y; N; N | |
| Participants | 99 women; caesarean; mean 31; ASA1,2; exc' study drug allergy, hypertension/DM, GI disease, antiemetic | |
| Interventions | Postoperative Either: PLACEBO; or CYCLIZINE 50mg iv; or DEXAMETHASONE 8mg | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Retching categorized as vomiting. Unclear if vomiters nauseated. Unclear if nauseated vomiters categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 150 boys; hypospadia; 3‐5 (mean 4) | |
| Interventions | Induction Either: PLACEBO; or CYCLIZINE 20mg iv; or ONDANSETRON 0.1mg/kg iv | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; A; Y; Y; N; N | |
| Participants | 140 adults & children; 87 female; dental; ASA1,2; 15‐65 (mean 28); exc' ASA>2, antiemetic | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 0.25 or 1.25mg i.v. | |
| Outcomes | Nausea; vomiting. Postop 0‐8; 24 hours. | |
| Notes | Outcomes commonest 0‐8 hours. Male/female incidences not reported. Retching categorized as nausea. Unclear if vomiters nauseated (or retched) or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; Y; N; N | |
| Participants | 68 women; ASA1,2; mastectomy | |
| Interventions | Induction Either: PLACEBO; or CLONIDINE 2 mcg/kg i.v. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐5; 5‐24; 0‐24 hours. | |
| Notes | Side effects "no difference". Categorized by severest symptom (vomiting>retching>nausea). I combined retching and vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; Y; N; N. | |
| Participants | 216 children; 55 girls; various surgeries; ASA1,2; 2‐12 (mean 6); exc' ASA>2, reflux, vomiting, obese, emergency, antiemetic | |
| Interventions | Intraoperative Either: DOLASETRON 45 or 175 or 350 or 700 mcg/kg iv; or ONDANSETRON 100 mcg/kg iv | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐6; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; N; Y; Y | |
| Participants | 60 adults; 28 women; orthopaedic; ASA1,2; mean 41 | |
| Interventions | Induction Either: PLACEBO; or epidural MORPHINE 0.6mg; or NEOSTIGMINE 60 mcg; or both | |
| Outcomes | Vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 90 adults; 51 women; tympanoplasty; ASA1,2; 18‐64 (mean 14); exc' menstrual, asthma, antiemetic | |
| Interventions | Preoperative Either: PLACEBO; or TANDOSPIRONE 10 or 30mg | |
| Outcomes | All outcomes. Postop 0‐3; 3‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Retching categorized separately. I combined retching and vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 60 women; gynaecological; ASA1,2 | |
| Interventions | Intraoperative Either: 5% dextrose; or 10% intralipid | |
| Outcomes | All outcomes. Postop 0‐4 hours. | |
| Notes | No side effects. I combined retching and vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 150 adults; 78 women; abdominal; ASA1,2 | |
| Interventions | Postoperative Either: PLACEBO; or ONDANSETRON 4mg i.v.; or TROPISETRON 5mg | |
| Outcomes | Nausea or vomiting; rescue antiemetic. Postop 0‐18; 0‐36 hours. | |
| Notes | Male/female incidences not reported. No side effects. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 50 children; 26 female; strabismus; ASA1,2; 4‐15 (mean 8); exc' motion sickness/PONV | |
| Interventions | Preoperative Either: NO TREATMENT; or ATROPINE 0.015mg/kg iv + DIAZEPAM 0.15mg/kg iv | |
| Outcomes | Nausea or vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 60 adults; 42 women; lap' chole'; ASA1,2; mean 45 | |
| Interventions | Induction Either: DROPERIDOL 1.25mg iv +/‐ GRANISETRON 3mg | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 15; 30 minutes; 1; 2; 4; 12; 24 hours. | |
| Notes | Outcomes commonest at 1 hour. Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 44 women; hysterectomy; ASA 1,2; 18‐65 (mean 45); exc' PONV/motion sickness, study drug allergy | |
| Interventions | Intraoperative Either: DROPERIDOL 15 mcg/kg iv; or TROPISETRON 0.05mg/kg | |
| Outcomes | Vomiting. Postop immediate; 2; 6; 12; 24; 48 hours. | |
| Notes | Vomiting commonest at 6 hours. Side effect "no difference". Selective reporting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; N; N; N | |
| Participants | 270 women; gynaecological; exc' bowel, N&V, antiemetic | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 2.5mg i.v.; or ONDANSETRON 8mg | |
| Outcomes | Nausea; vomiting; rescue antiemetic. PACU;PACU discharge‐6; 6‐24; 0‐24 hours. | |
| Notes | Nausea commonest 6‐24 hours. Unclear if vomiters/retchers nauseated or categorized twice. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N | |
| Participants | 100 women; gynaecological; exc' clonidine/opioid | |
| Interventions | Induction AND intraoperative Either: PLACEBO; or epidural CLONIDINE 2 or 3 or 4 mcg/ml | |
| Outcomes | All outcomes Postop 0‐24hours. | |
| Notes | Side effects "no difference". | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; N; N; N | |
| Participants | 144 women; gynaecological; exc' antiemetic, N&V, study drug allergy | |
| Interventions | Induction Either: NO TREATMENT; or DOLASETRON 12.5 mg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐2; 0‐4; 0‐6; 6‐24 hours. | |
| Notes | Outcomes commonest 0‐6 hours. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. Graph/text discrepancies. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; N; Y; N | |
| Participants | 120 women; gynaecological; mean 49; exc' N&V, antiemetic, study drug contraindication | |
| Interventions | Induction AND intraoperative Either: NO TREATMENT then DOLASETRON 12.5mg iv; or ONDANSETRON 4mg or TROPISETRON 2mg then NO TREATMENT | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | No side effects. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 50 children; 26 female; tonsil +/‐ adenoids; exc' steroid contraindication | |
| Interventions | Postoperative day 1‐7 Either: PLACEBO; or oral DEXAMETHASONE 0.5mg/kg | |
| Outcomes | Nausea or vomiting. Postop day one; two; three; four; five; two to eight. | |
| Notes | Male/female incidences not recorded. Outcomes commonest days 2‐8. Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 80 women; caesarean; ASA1 | |
| Interventions | Intraoperative Either: PLACEBO; or intrathecal CLONIDINE 150 mcg; or NEOSTIGMINE 50 mcg; or both | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; N; N; N | |
| Participants | 164 women; Caesarean; exc' breastfeeding, psychiatric, antiemetic | |
| Interventions | Intraoperative Either: PLACEBO; or METOCLOPRAMIDE 10mg i.v.; or ONDANSETRON 4mg | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 80 women; laparoscopies; ASA1,2 | |
| Interventions | Preoperative AND induction Either: PLACEBO twice; or PLACEBO then METOCLOPRAMIDE 10mg i.v.; or oral CIMETIDINE 300mg then PLACEBO; or both | |
| Outcomes | All outcomes. Postop (time unclear) | |
| Notes | No side effects. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 40 adults; 22 women; arthroplasties; ASA1,2; mean 70; exc' study drug allergy, liver/heart/renal disease, drug abuse, obese | |
| Interventions | Postoperative PCA Either: PLACEBO; or METOCLOPRAMIDE 1mg/ml | |
| Outcomes | Nausea; vomiting. PACU (time unclear); 0‐24; 24‐48 hours. | |
| Notes | Outcomes commonest 0‐24 hours. Male/female incidences not reported. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 120 women; gynaecological; 27‐43 (mean 36); ASA1,2; exc' pregnant, GI/liver disease, motion sickness, Parkinson's, antiemetic, menstrual | |
| Interventions | Induction Either: METOCLOPRAMIDE 10mg i.v.; or METOCLOPRAMIDE 5mg i.v. AND TROPISETRON 5mg | |
| Outcomes | Nausea or vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Unclear how retching categorized. Unclear if nauseated vomiters categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 130 children; no sex datum; tonsil +/‐ adenoid; ASA1,2; 2‐12 (mean 6); exc' antiemetic, antihistamine, steroid, psychoactive drug, DM | |
| Interventions | Induction Either: PLACEBO; or DEXAMETHASONE 1mg/kg (max 25mg) i.v. | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐3; 3‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Retching categorized separately. I combined retching and vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; Y; N; N | |
| Participants | 44 adults; 23 female; hemiarthroplasty; >50 (mean 69); ASA1‐3; exc' allergy, various drugs, heart/liver disease, regional anaesthesia | |
| Interventions | Preoperative AND postoperative twice Either; PLACEBO thrice; or oral CLONIDINE 5 mcg/kg thrice | |
| Outcomes | Nausea; vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effect "clonidine sedative". Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N. | |
| Participants | 90 adults, no sex data, arthoplasty, ASA1‐3, >60 | |
| Interventions | Induction AND postoperative thrice Either: PLACEBO; or ONDANSETRON 16mg four times | |
| Outcomes | See notes. | |
| Notes | Side effects not recorded. No numbers reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 200 children; 122 girls; mean 4; ASA1,2; various surgeries; exc' anticonvulsant/sedative, GI disease | |
| Interventions | Preoperative Either: PLACEBO; or oral DIAZEPAM 0.5mg/kg ; or MIDAZOLAM 0.25 or 0.5mg/kg | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐1; 1‐4; 0‐24 hours. | |
| Notes | Incidences not reported. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 60 adults; no sex data; lap' chole'; 22‐84; exc' NG, liver/GI disease, DM, infection | |
| Interventions | Preoperative AND intraoperative Either: ONDANSETRON 4mg i.v. then PLACEBO; or PLACEBO then TROPISETRON 5mg | |
| Outcomes | Nausea; vomiting. Postop 0‐1; 0‐4; 0‐8; 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; N; N | |
| Participants | 433 children; 158 girls; various surgeries; 2‐12 (mean 5); exc' antiemetic, liver/renal disease, PONV | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 0.1 mg/kg (max 4mg) i.v. | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐2; 0‐24 hours. | |
| Notes | Rescue antiemetic only 0‐2 hours. Male/female incidences not reported. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 35 women; gynaecological/breast; ASA1,2; mean 41 | |
| Interventions | Preoperative Either: NO TREATMENT; or buccal PROCHLORPERAZINE 3mg | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐1; 1‐4; 4‐24; 0‐24 hours. | |
| Notes | Side effects not recorded. Unclear if vomiters nauseated or categorized once or twice. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 52 women; gynaecological/breast; ASA1,2; exc' study drug allergy, pregnant/breastfeeding | |
| Interventions | Preoperative Either: NO TREATMENT; or buccal PROCHLORPERAZINE 6mg | |
| Outcomes | All outcomes. Postop 0‐1; 1‐4; 4‐24; 0‐24 hours. | |
| Notes | No side effects. Unclear how retching categorized or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 160 adults; 115 women; various surgery | |
| Interventions | Intraoperative Either: NO TREATMENT; or METOCLOPRAMIDE 10mg i.v. | |
| Outcomes | Vomiting. Postop 0‐4 hours. | |
| Notes | Male/female incidences not reported. No side effects. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | High risk | C ‐ Inadequate |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 60 adults; 28 women; lap' chole'; ASA1,2; exc' opioid/antiemetic/N&V, reflux, drug abuse | |
| Interventions | Induction Either: ONDANSETRON 4 or 8mg i.v. | |
| Outcomes | Nausea or vomiting; rescue antiemetic. Postop 0‐2; 2‐6; 6‐12; 12‐18; 18‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Inconsistent data. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; N; N | |
| Participants | 140 women; gynaecological | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 1mg i.v.; or METOCLOPRAMIDE 10mg; or ONDANSETRON 4mg | |
| Outcomes | Nausea; vomiting; rescue antiemetics. Postop 0‐2; 2‐6; 6‐24; 0‐24 hours. | |
| Notes | Nausea only 0‐24 hours. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 60 children; girls 28; 3‐14 (mean 10); otoplasty | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 75 mcg/kg i.v.; or ONDANSETRON 0.1mg/kg | |
| Outcomes | Vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects nodal rhythm with ondansetron. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | a) 1169 women; various surgeries; ASA1,2; 18‐70; exc' pregnant/breastfeeding, NG, obese: b) 468 men & boys; >12; ASA1,2; exc' antiemetic, liver biopsy, NG, obese | |
| Interventions | Induction a) Either: PLACEBO; or ONDANSETRON 1 or 4 or 8mg i.v. b) Either: PLACEBO; or ONDANSETRON 4mg i.v. | |
| Outcomes | Nausea; Vomiting. Postop 0‐24 hours. | |
| Notes | Retching categorized as vomiting. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 90 women; gynaecological; 18‐70 (mean 46); ASA1,2; exc' <45 >90 kg, opioid | |
| Interventions | Induction Either: DROPERIDOL 1.25mg i.v.; or ONDANSETRON 4mg; or both | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | No side effects. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 104 children; no sex data; strabismus; ASA1,2; mean 5. | |
| Interventions | Induction AND intraoperative Either: PLACEBO or DROPERIDOL 10 mcg/kg i.v. then NO TREATMENT; or NO TREATMENT or DROPERIDOL 10 mcg/kg then METOCLOPRAMIDE 0.1mg/kg | |
| Outcomes | Nausea; vomiting. PACU; 0‐1; 6‐12; 24 hours. | |
| Notes | Nausea and vomiting commonest 0‐1 hour. Male/female incidences not recorded. No side effects. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 100 adults; 63 women; lap' chole'; 20‐70 (mean 53); ASA1‐3; exc' jaundice/cholecystitis, study drug allergy, steroid, renal/liver/cardiac disease, ASA>3, post‐hoc conversion | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 1.25mg i.v.; or METOCLOPRAMIDE 10mg; or ONDANSETRON 4mg | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 199 children >1 (mean 7.5); ENT | |
| Interventions | Preoperative Either: LORAZEPAM 0.05mg/kg oral; or TRIMEPRAZINE 3mg/kg oral | |
| Outcomes | Vomiting. Postop at 4 hours. | |
| Notes | Male/female incidences not recorded. No side effects. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 1030 adults; 722 women; various surgeries; >17 (mean 37); exc' antiemetic, disease, obese, liver/GI surgery | |
| Interventions | Intraoperative Either: PLACEBO; or DOLASETRON 12.5 or 25 or 50 or 100mg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Unclear if vomiters nauseated or categorized once or twice. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 120 women; laparoscopies; ASA1‐3; exc' pregnancy, alcohol, opioid/antiemetic | |
| Interventions | Preoperative Either: PLACEBO; or GINGER 1g oral; or METOCLOPRAMIDE 10mg oral | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Side effect "no difference". Unclear how retching categorized or if nauseated vomiters categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 150 women; hysterectomy; ASA1,2; mean 50; exc' lung/heart/liver/kidney disease, fever, antiemetic, obese, N&V, alcoholic | |
| Interventions | Preoperative AND intraoperative Either: PLACEBO then NO TREATMENT/METOCLOPRAMIDE 20mg i.v.; or oral DOLASETRON 50mg then NO TREATMENT | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized or if vomiters nauseated. Patients categorized by severest symptom. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; N | |
| Participants | 387 adults; 288 women; lap' chole'; mean 53; ASA1‐3; exc' antiemetic, obese, cardiac, Parkinson's disease, study drug allergy, alcohol abuse | |
| Interventions | Induction Either: PLACEBO; or DOLASETRON 12.5mg iv; or METOCLOPRAMIDE 20mg | |
| Outcomes | All outcomes. Postop 0‐4; 4‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. I combined retching and vomiting. Unclear if retchers/vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 150 women; hysterectomy/breast; ASA1‐3; mean 53; exc' study drug allergy, GI/heart/renal/liver/CNS disease, pregnant/menstrual | |
| Interventions | Preoperative Either: PLACEBO/DEXAMETHASONE 8mg iv AND oral DOLASETRON 50mg | |
| Outcomes | All outcomes. Postop 0‐2; 0‐24 hours. | |
| Notes | No side effects. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 54 adults; 39 women; orthopaedic; ASA1‐3 | |
| Interventions | Intraoperative Either: PLACEBO; or TROPISETRON 5mg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐6; 7‐12; 13‐18; 19‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 73 adults; no sex data; ASA1‐3; 50‐85 (mean 65); orthopaedic. | |
| Interventions | Induction AND postoperative twice Either: PLACEBO thrice; or METOCLOPRAMIDE 20mg i.v. thrice; or ONDANSETRON 8mg thrice | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; N; Y; Y | |
| Participants | 120 adults; 75 women; various operations; exc' <18 >75, ASA>2, BMI>28, heart disease, antiemetic/N&V | |
| Interventions | Preoperative AND intraoperative Either: PLACEBO twice; or PLACEBO then ONDANSETRON 4mg i.v.; or ONDANSETRON 4mg i.v. then PLACEBO | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 90 adults; 71 women; general/gynaecological; mean 45; exc' ASA>3, <21 >75 years, obese, PONV, liver/renal/heart disease, pregnant/breastfeeding, anxiety, antiemetic | |
| Interventions | Preoperative AND intraoperative Either: PLACEBO twice; or PLACEBO then ONDANSETRON 4mg i.v.; or ONDANSETRON 4mg i.v. then PLACEBO | |
| Outcomes | Nausea; vomiting. Postop 0‐6 days. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 80 women; gynaecological; 20‐50 years; exc' pregnanct liver/GI disease, opioids/alcohol/antiemetic, post‐hoc conversion | |
| Interventions | Preoperative Either: PLACEBO; or oral GINGER 1g | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 2; 4; 0‐24 hours. | |
| Notes | Side effects not reported. Unclear if vomiters nauseated or categorized once or twice. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y. | |
| Participants | 153 women; gynaecological; exc' disease, psychoactive/sedative drugs | |
| Interventions | Preoperative Either: oral BROMAZEPAM 6mg; or oral LORAZEPAM 2mg | |
| Outcomes | Nausea; vomiting. Postop 0‐24 hours. | |
| Notes | Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 158 adults; 90 female; mean 43; hernia/varicose veins; exc' short operations | |
| Interventions | Preoperative Either: ATROPINE 0.6 mg +/‐ DIAZEPAM 5 or 10mg | |
| Outcomes | Nausea; vomiting. Postop 0‐3; 3‐24 hours. | |
| Notes | Outcomes commonest 3‐24 hours. Male/female incidences not reported. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 90 women; gynaecological; ASA1,2; 21‐73 (mean 49); exc' renal/liver/cardiac/blood/endocrine disease, pregnant/breastfeeding, antiemetic, NG | |
| Interventions | Preoperative ten minutes Either: PLACEBO; or ONDANSETRON 4 or 8mg i.v. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | No side effects. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 100 women; various surgeries; ASA1,2; 18‐65 (mean 48); exc' antiemetic, obese, study drug allergy | |
| Interventions | Induction then postoperative Either: PLACEBO thrice; or DROPERIDOL 2.5mg i.v. then PLACEBO/ONDANSETRON 4mg then DROPERIDOL 1.25mg; or ONDANSETRON 4mg AND PLACEBO twice | |
| Outcomes | All outcomes. Postop 0‐2; 0‐12; 0‐24; 0‐48 hours. | |
| Notes | Retching categorized as vomiting. Unclear if vomiters/retchers nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; N; N; Y; Y | |
| Participants | 60 adults; female 37; ASA1‐3; neurosurgery; exc' antiemetic | |
| Interventions | Intraoperative Either: METOCLOPRAMIDE 10mg i.v.; or ONDANSETRON 8mg | |
| Outcomes | All outcomes. Postop 0‐48 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Retching categorized as vomiting, unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; N; N | |
| Participants | 150 women; gynaecological; ASA1‐3; mean 50; exc' antiemetic | |
| Interventions | Intraoperative Either: PLACEBO; or DROPERIDOL 1.25mg i.v.; or TROPISETRON 5mg | |
| Outcomes | All outcomes. Postop 0‐2; 2‐6; 6‐24; 24‐48; 0‐48 hours. | |
| Notes | Side effects period unclear. Participants categorized by severest symptom (vomit>retch>nausea). Unclear if retchers/vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; N; N; N | |
| Participants | 100 women; gynaecological; ASA1,2; mean 37; exc' N&V/antiemetic, pregnant/breastfeeding, obese, lung disease | |
| Interventions | Intraoperative AND postoperative Either: OXYGEN 30% or 80% | |
| Outcomes | All outcomes PACU (time unclear); discharge‐24; 0‐24 hours. | |
| Notes | No side effects. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; N; N | |
| Participants | 100 women; breast; ASA1‐3; | |
| Interventions | Intraoperative AND postoperative Either: OXYGEN 30% or 50% | |
| Outcomes | All outcomes. Postop 0‐2; 2‐6; 6‐24; 0‐24 hours. | |
| Notes | Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 100 adults; 58 women; elective surgery; ASA1‐3; 17‐80 (mean 42); exc' antiemetic, N&V, renal/liver disease, reflux | |
| Interventions | Induction Either: METOCLOPRAMIDE 10mg i.v.; or ONDANSETRON 4mg | |
| Outcomes | Nausea; vomiting. Postop 0‐1; 1‐12; 12‐24; 0‐24 hours. | |
| Notes | Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; Y; N; N | |
| Participants | 122 adults; 93 women; lap' chole'; ASA1,2; exc' antiemetic, <18 | |
| Interventions | Intraoperative Either: METOCLOPRAMIDE 20mg i.v.; or ONDANSETRON 8mg | |
| Outcomes | Vomiting; nausea or vomiting; rescue antiemetic. Postop 0‐6; 6‐12; 12‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Retching categorized as vomiting. Unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; A; Y; Y; Y; Y | |
| Participants | 45 women; caesarean; ASA1,2; mean 26; exc' ASA>2, CNS/spinal disease, opioid/naloxone, epidural contraindication/vein/dural puncture, post‐hoc surgery | |
| Interventions | Postoperative Either: PLACEBO; or epidural CLONIDINE 2 mcg/kg/day | |
| Outcomes | Nausea; vomiting; rescue antiemetics. Postop 0‐48 hours. | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; A; Y; N; Y; Y | |
| Participants | 41 women; laparoscopy; 20‐40 (mean 29); ASA1; exc' pregnant, obese, motion sickness/PONV, antiemetic, steroid, DM, HH | |
| Interventions | Induction Either: DEXAMETHASONE 8mg i.v. + ONDANSETRON 4mg; or ONDANSETRON 8mg | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐2; 2‐24; 0‐24 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 104 women; gynaecological; ASA1,2; 18‐65 (mean 43); exc' obese, heart/lung/renal/liver/CNS/GI disease, PONV, N&V, antiemetic | |
| Interventions | Intraoperative end Either: PLACEBO; or DROPERIDOL 1.25mg i.v.; or METOCLOPRAMIDE 10mg | |
| Outcomes | Nausea; vomiting. Postop 0‐6; 7‐24; 13‐18; 19‐24; 0‐24 hours. | |
| Notes | Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; N; N; N | |
| Participants | 123 women; laparoscopies; exc' pregnant/breastfeeding, drug | |
| Interventions | Induction Either: METOCLOPRAMIDE 10mg i.v.; or ONDANSETRON 4mg | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐6; 6‐24; 0‐24 hours. | |
| Notes | Nausea and vomiting only 0‐6 hours. Retching categorized as vomiting. Unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; N, N; Y; Y | |
| Participants | 252 women; gynaecological; 16‐50; 28‐68kg; exc' surgery > 10 min, disease affecting outcome, study drug allergy | |
| Interventions | Preoperative Either: No treatment; or ATROPINE 0.65mg i.m.; or CHLORPROMAZINE 25mg; or both | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postoperatively at 0‐1; 1‐6; 0‐6 hours. | |
| Notes | Retching categorized as vomiting. Unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; N; N | |
| Participants | 216 women; breast; ASA1,2; 18‐80 (mean 54); exc' pregnant, obese, steroid | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 1.25mg i.v.; or ONDANSETRON 8mg | |
| Outcomes | All outcomes. Postop 0‐2; 0‐24 hours. | |
| Notes | Unclear how retching categorized. Unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; N; N | |
| Participants | 39 adults; 18 women; ENT; 18‐65 (mean 44); ASA1,2; exc' anticholinergic/antihistamine, glaucoma, GI/renal tract obstruction | |
| Interventions | Preoperative Either: PLACEBO; or SCOPOLAMINE 1.5mg patch | |
| Outcomes | Nausea or vomiting. Postop 0‐12; 12‐36 hours. | |
| Notes | Male/female incidences not reported. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; Y; Y; Y | |
| Participants | 142 children; general surgery; >30kg; exc' unsuitable for study | |
| Interventions | Preoperative Either: DIAZEPAM 0.3mg/kg oral; or FLUNITRAZEPAM 0.03 mg/kg | |
| Outcomes | Vomiting. Postop 24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; N; N; N | |
| Participants | 30 women; lap' steri'; ASA1,2; exc' alcoholism, drug abuse, sedative, antidepressant, antiepileptic, other surgery, study drug allergy | |
| Interventions | Preoperative Either: PLACEBO; or MIDAZOLAM 0.04 mg/kg i.v. | |
| Outcomes | Rescue antiemetic. Postop 0‐5 hours. | |
| Notes | Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 160 women; hysterectomy; ASA1,2; exc' <30 >45 years, ASA>2, antiemetic, drug abuse | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 4mg i.v. | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Side effects not reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; N; N; N; N | |
| Participants | 200 children, 1‐15 years, ASA 1&2, orthopaedic | |
| Interventions | Intraoperative Either: PLACEBO; or DROPERIDOL 5 mcg/kg i.v. | |
| Outcomes | Vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; N; N | |
| Participants | 45 adults; 19 women; laminectomy; exc pregnant, HH | |
| Interventions | Postoperative PCA Either: PLACEBO; or DROPERIDOL bolus 0.15mg | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐48 hours. | |
| Notes | Male/female incidences not reported. Side effect "droperidol sedative". Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 120 adults; 62 women; orthopaedic; 18‐40 (mean 26) | |
| Interventions | Preoperative Either: NO TREATMENT; or DIAZEPAM 0.2mg/kg i.m.; or PROMETHAZINE 1mg/kg | |
| Outcomes | Nausea; vomiting. Postop 0‐1; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effect "sedation". Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; N; N | |
| Participants | 80 adults; 55 women; dental; ASA1,2; 18‐48 (mean 25); exc' men>120 kg, women > 100kg, pregnant/breastfeeding, study drug allergy | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 4mg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐1; 0‐4; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; N; N; N | |
| Participants | 34 adults; 11 women; maxillofacial; 19‐68 (mean 40); exc' men>120kg, women>100kg, pregnant/breastfeeding | |
| Interventions | Preoperative Either: PLACEBO; or ONDANSETRON 4mg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐1; 0‐4; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 60 adults; no sex data/age data; ASA1,2; maxillofacial. | |
| Interventions | Intraoperative Either: PLACEBO; or METOCLOPRAMIDE 10mg i.v.; or TROPISETRON 5mg | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; Y; Y; Y | |
| Participants | 90 children; 44 female; strabismus; ASA1,2 | |
| Interventions | Induction Either: PLACEBO; or METOCLOPRAMIDE 0.25mg/kg i.v.; or ONDANSETRON 0.15mg/kg | |
| Outcomes | Vomiting; rescue antiemetic Postop 0‐4; 0‐24 hours. | |
| Notes | Rescue antiemetic only 0‐4 hours. Male/female incidences not reported. Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; Y; N; N | |
| Participants | 212 children; 96 girls; tonsillectomy; ASA1,2; 2‐12 (mean 7); exc' POV, study drug allergy | |
| Interventions | Induction AND postoperative Either: PLACEBO twice; or METOCLOPRAMIDE 0.25mg/kg i.v. or ONDANSETRON 0.15mg/kg i.v. then PLACEBO; or either drug twice | |
| Outcomes | Vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; Y; N; N | |
| Participants | 140 children; 60 female; tonsil +/‐ adenoid; ASA1,2; 1‐12 (mean 6); exc' PONV, motion sickness, reflux, study drug allergy | |
| Interventions | Preoperative Either: PLACEBO; or oral ONDANSETRON 0.075 or 0.15mg/kg | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐18; 18‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; N; N | |
| Participants | 100 women; gynaecological; ASA1,2; exc' hypertension/heart disease, antiemetic | |
| Interventions | Intraoperative Either: PLACEBO; or DROPERIDOL 0.04mg/kg i.m.; EPHEDRINE 0.5mg/kg | |
| Outcomes | Vomiting; nausea or vomiting; rescue antiemetic. Postop 0‐2; 2‐24 hours. | |
| Notes | Vomiting only 2‐24 hours. Nausea or vomiting and rescue antiemetic only 0‐2 hours. No side effects. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; N; N | |
| Participants | 100 women; gynaecological; ASA1,2; exc' antiemetic, steroid, ulcer, DM, study drug contraindication | |
| Interventions | Intraoperative Either: DEXAMETHASONE 0.17 mg/kg i.v.; or DROPERIDOL 0.02 mg/kg | |
| Outcomes | All outcomes. Postop 0‐2; 2‐8; 8‐24; 0‐24 hours. | |
| Notes | Nausea commonest 0‐2 hours, vomiting 2‐8 hours. Side effects not reported. Retching categorized as vomiting. Unclear if vomiters nauseated. Categorized by severest symptom. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; Y; N; N | |
| Participants | 56 adults; women 13; lap' chole'; ASA1,2; exc' post hoc conversion | |
| Interventions | Preoperative Either: PLACEBO; or ONDANSETRON 4mg i.v. | |
| Outcomes | Nausea or vomiting. Postop 0‐72 hours. | |
| Notes | Male/female incidences not reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; N; N; N | |
| Participants | 120 women; laparoscopy; 18‐55 (mean 34); ASA1,2; exc' study drug allergy, pregnancy, GI disease, antiemetic | |
| Interventions | Intraoperative Either: PLACEBO; or DOLASETRON 12.5mg i.v.; or METOCLOPRAMIDE 10mg | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; N; N; N | |
| Participants | 170 women; gynaecological; 18‐50 (mean 35); exc' ASA>2, <18 >50, pregnant/breastfeeding, study drug allergy, antiemetic, BM><30%, post‐hoc protocol violation | |
| Interventions | Intraoperative Either: PLACEBO; or DROPERIDOL 2.5mg i.v.; or METOCLOPRAMIDE 10mg | |
| Outcomes | Nausea; vomiting. Postop 0‐24 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; N; N | |
| Participants | 40 women; caesarean; exc' liver/renal/psychiatric disease, pre‐eclampsia, epilepsy, drug abuse, antiemetic, breastfeeding | |
| Interventions | Postoperative PCA Either: PLACEBO; or DROPERIDOL 10mg | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐20 hours. | |
| Notes | No side effects. Unclear if vomiters/retchers nauseated. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 2158 women; 12‐75 (mean 45); ASA1‐3; various surgeries; exc' pregnant/breastfeeding, obese, N&V/antiemetic, NG, liver/renal disease | |
| Interventions | Preoperative Either: PLACEBO; or oral ONDANSETRON 4 or 8 or 16mg | |
| Outcomes | Nausea; vomiting. Postop 0‐24 hours. | |
| Notes | Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 54 women; mastectomy; ASA1,2; exc' obese, gastric obstruction, antiemetic | |
| Interventions | Intraoperative end Either: PLACEBO; or ONDANSETRON 4mg i.v. | |
| Outcomes | All outcomes. Postop 0‐2; 2‐6; 6‐24; 0‐24 hours. | |
| Notes | Unclear how retching categorized or if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 180 children; 95 female; ASA1,2; 2‐12 (mean 7); strabismus; exc' antiemetic | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 25/50/75/100/150 mcg/kg i.v. | |
| Outcomes | Nausea or vomiting; rescue antiemetic. Postop 0‐2; 2‐6; 0‐6; 6‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Unclear how retching categorized. Unclear if nauseated vomiters categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; Y; N | |
| Participants | 30 adults; 16 women; subdural electrodes; mean 37; exc' headache/pain syndrome, study drug allergy | |
| Interventions | Preoperative AND postoperative 6 hourly Either: PLACEBO each time; or DEXAMETHASONE 10mg iv then 4mg 6 hourly for 1 day, 2mg 6 hourly for 1 day, 2mg 12 hourly for 1 day | |
| Outcomes | See notes. | |
| Notes | Incidences not reported. Male/female incidences not recorded. No side effects. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; A; Y; N; Y; Y | |
| Participants | 100 adults; 49 women; various surgeries; ASA1,2; exc' drugs. | |
| Interventions | Induction AND intraoperative Either: ATROPINE 6 then 15 mcg/kg i.v.; or GLYCOPYRROLATE 3 then 7.5 mcg/kg | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐2; 2‐24; 0‐24 hours | |
| Notes | Retching categorized as nausea. Unclear if vomiters nauseated or categorized once or twice. Vomiting and rescue antiemetic commonest 0‐2 hours. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; N | |
| Participants | 97 women; caesarean; ASA1,2; mean 29; exc' convulsions/Parkinsonism, drug abuse, psychiatric | |
| Interventions | Intraoperative twice Either: PLACEBO or epidural DROPERIDOL 2.5mg then NO TREATMENT; or epidural PLACEBO then DROPERIDOL 2.5mg i.v. | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | No side effects. Unclear how retching categorized. Vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y. | |
| Participants | 94 women; hysterectomy; ASA1,2; mean 43; exc' asthma, study drug allergy, drug abuse, psychiatric/convulsions/parkinsonism, GI disease | |
| Interventions | Postoperative PCA. Either: PLACEBO; or DROPERIDOL 0.0625mg/bolus | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐2; 2‐6; 6‐18; 18‐24; 0‐24 hours. | |
| Notes | Retching categorized as vomiting. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 90 women; gynaecological; ASA1,2; 18‐65 (mean 48); exc' opioids/NSAID/steroids/antiemetic, study drug allergy | |
| Interventions | Induction AND postoperative Either: DROPERIDOL 1.25mg iv AND DEXAMETHASONE 8mg/ONDANSETRON 4mg then DROPERIDOL 1.25mg; or DEXAMETHASONE 8mg AND ONDANSETRON 4mg then PLACEBO | |
| Outcomes | All outcomes. Postop 0‐2; 2‐12; 12‐24; 24‐48; 0‐48 hours. | |
| Notes | Nausea commonest 2‐12 hours, vomiting 12‐24 hours. Side effect "no difference". Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; N; N; Y; Y | |
| Participants | 32 women; STOP; ASA1 | |
| Interventions | Induction Either: PLACEBO; or METOCLOPRAMIDE 0.2mg/kg i.v.; or PROMETHAZINE 0.5mg/kg | |
| Outcomes | Vomiting; nausea or vomiting. Postop 0‐4 hours. | |
| Notes | Side effect "promethazine sedative". Data inconsistent. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 51 women; gynaecological; 18‐75 (mean 47); exc' <18 >75 years, >100kg, antiemetic/N&V, ASA>3, renal disease, drug abuse, study drug allergy | |
| Interventions | (a) Preoperative Either: PLACEBO; or oral ONDANSETRON 8 or 16mg (b) Preoperative AND postoperative twice Either: PLACEBO; or oral ONDANSETRON 8 or 16mg thrice | |
| Outcomes | Nausea; vomiting. Postop 0‐24 hours. | |
| Notes | Side effects not reported. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; N; N | |
| Participants | 100 adults | |
| Interventions | Preoperative Either: PLACEBO; or DROPERIDOL 5mg i.v.; or SULPIRIDE 50 or 100mg | |
| Outcomes | Vomiting; nausea or vomiting. Postop 0‐48 hours. | |
| Notes | Male/female incidences not recorded. Methodology unclear, data inconsistent. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 40 children; 19 female; 3‐12 (mean 6); ASA1; strabismus; exc' N&V or drug | |
| Interventions | Preoperative Either: PLACEBO; or DIMENHYDRINATE 50 mg suppository | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Male/female incidences not recorded. No side effects. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N, B, Y, N, Y; Y | |
| Participants | 64 adults; 40 women; middle ear; exc' <18 >60, neuroleptic, pregnant, study drug allergy, renal/heart disease | |
| Interventions | Preoperative thrice then intraoperative Either: PLACEBO; or ALIZAPRIDE oral 50mg thrice then 50mg i.v. | |
| Outcomes | Nausea, vomiting, nausea or vomiting. Postop 0‐8; 8‐24; 24‐30; 30‐48 hours | |
| Notes | Male/female incidences not given. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. Nausea or vomiting commonest 0‐8 hours. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 842 adults; 622 women; ASA1‐3; 18‐75; various surgeries; exc' ASA>3, pregnant, allergy, NG, drug affecting outcome, drug abuse | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 4mg i.v.; or TROPISETRON 2mg | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Retching categorized as vomiting. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; N; N; Y; Y | |
| Participants | 40 adults; 31 women; cholecystectomy; ASA1,2; mean 49; exc' >70, glaucoma, anticholinesterase, psychiatric | |
| Interventions | Preoperative AND intraoperative Either: PLACEBO then DROPERIDOL 7.5mg i.v.; or SCOPOLAMINE patch then PLACEBO | |
| Outcomes | All outcomes Postop 0‐1; 1‐2; 2‐6; 6‐18; 0‐18 hours. | |
| Notes | No side effects. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; N; N; Y; N | |
| Participants | 143 women; gynaecological; mean 47; exc' pregnant, cancer last 5 years, antiemetic, acupressure, neuropathy | |
| Interventions | Preoperative twice Either: PLACEBO tice; or PLACEBO then DROPERIDOL 1.25mg iv; or ACUPRESSURE then PLACEBO/DROPERIDOL 1.25mg | |
| Outcomes | Nausea; vomiting. Postop day one; day two; 0‐36 hours. | |
| Notes | Vomiting commonest day one. Side effects not recorded. Retching categorized as vomiting. Unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 50 adults; 23 women; vitrectomy; exc' ASA>3, renal/CNS/cardiac disease, pregnant, drug abuse | |
| Interventions | Induction Either: PLACEBO; or MAGNESIUM 50mg/kg i.v. | |
| Outcomes | Nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 160 children; 83 girls; strabismus; ASA1,2; 1‐12 (mean 4); exc' antiemetic, obese | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 75 mcg/kg i.v.; or METOCLOPRAMIDE 250 mcg/kg; or ONDANSETRON 100 mcg/kg | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐2; 2‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. No side effects. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; N; Y; Y | |
| Participants | 575 adults; 364 women; various surgery; ASA1‐3 | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 4mg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetic. PACU; PACU ‐ hospital discharge (time unclear). | |
| Notes | Nausea and rescue antiemetic only first period, vomiting second period. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 79 women; gynaecological; exc' pregnant/breastfeeding, antiemetic | |
| Interventions | Intraoperative Either: PLACEBO; or ONDANSETRON 4mg i.v. | |
| Outcomes | All outcomes. Postop 0‐4; after 4 hours. | |
| Notes | Nausea, 'nausea or vomiting' and rescue antiemetic only 0‐4 hours. Vomiting commonest >4 hours. Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 72 women; hysterectomy; 18‐65 (mean 46); ASA1,2 | |
| Interventions | Preoperative Either: PLACEBO; or HYOSCINE patch | |
| Outcomes | All outcomes. PACU; 3‐24; 24‐48;48‐72 hours. | |
| Notes | Outcomes commonest 3‐24 hours. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 150 children; 88 girls; strabismus; ASA1,2; 2‐15 (mean 7); exc' antiemetic | |
| Interventions | Intraoperative Either: PLACEBO; or ONDANSETRON 100 mcg/kg i.v. | |
| Outcomes | Nausea; nausea or vomiting; rescue antiemetics. Postop 0‐2; 2‐6; 6‐24; 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 51 adults; 28 women; ophthalmological; 21‐90 (mean 57); ASA1,2 | |
| Interventions | Intraoperative Either: DIFENIDOL 40mg i.m.; or METOCLOPRAMIDE 10mg; or ONDANSETRON 4mg | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐4 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 82 women; gynaecological; mean 35; exc' lung/cardiac disease | |
| Interventions | Preoperative Either: PLACEBO; or METOCLOPRAMIDE 10mg i.m. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐6 hours. | |
| Notes | No side effects. Retching categorized as nausea. Unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; N; N | |
| Participants | 60 adults; no sex data; various surgeries; mean 29; ASA1,2; exc' benzodiazepine, drug abuse/allergy, pregnant/breastfeeding, bleeding disorder, CNS disease | |
| Interventions | Induction Either: PLACEBO; or intrathecal MIDAZOLAM 2.5mg | |
| Outcomes | Nausea; vomiting. Postop 0‐48 hours. | |
| Notes | Male/female incidences not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; N; N | |
| Participants | 50 women; hysterectomy; ASA1,2; exc' premedicant except temazepam | |
| Interventions | Postoperative PCA Either: PLACEBO; or DROPERIDOL 0.05mg/ml | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | No side effects. Retching categorized as vomiting. Unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; N; N | |
| Participants | 100 women; gynaecological surgery; ASA1,2; 16‐70 (mean 50); exc' antiemetic | |
| Interventions | Intraoperative Either: PLACEBO; or DROPERIDOL 2.5mg i.v.; or METOCLOPRAMIDE 10mg; or ONDANSETRON 4mg | |
| Outcomes | Nausea or vomiting. Postop 0‐6 hours. | |
| Notes | Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 176 children; strabismus; 9 months to 12; ASA1;exc' motion sickness/PONV | |
| Interventions | Intraoperative Either: PLACEBO; or METOCLOPRAMIDE 0.25mg/kg i.v.; or ONDANSETRON 0.15mg/kg | |
| Outcomes | Vomiting. Postop 0‐2; 2‐6; 6‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; N; Y; Y | |
| Participants | 76 children; 36 girls; strabismus; ASA1,2; 1‐12 (mean 7); exc' reflux, motion sickness, CNS/inner ear disease | |
| Interventions | Induction Either: PLACEBO; or METOCLOPRAMIDE 0.25mg/kg i.v. | |
| Outcomes | Nausea or vomiting. Postop 0‐2; 2‐6; 6‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. No side effects. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; Y; Y; Y | |
| Participants | 240 children; 121 girls; strabismus; ASA1,2; exc' motion sickness/PONV | |
| Interventions | Induction AND intraoperative Either: PLACEBO twice; or DROPERIDOL 25 mcg/kg/ONDANSETRON 150 mcg/kg i.v. then PLACEBO; or DROPERIDOL 15 mcg/kg then ONDANSETRON 100 mcg/kg | |
| Outcomes | Nausea or vomiting; rescue antiemetic. Postop 0‐2; 2‐6; 6‐24; 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 61 women; gynaecological; ASA1,2; 25‐60 (mean 43); exc' analgesic/psychotropic drug | |
| Interventions | Preoperative Either: PLACEBO; or oral DIAZEPAM 10mg; or HYOSCINE 0.6mg | |
| Outcomes | Vomiting. Postop 0‐3 hours. | |
| Notes | Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; N; N | |
| Participants | 45 adults; 21 women; neurosurgery; ASA1‐3; 13‐60 (mean 35); exc' N&V/antiemetic, VP shunt, study drug allergy, drug abuse | |
| Interventions | Preoperative Either: PLACEBO; or ONDANSETRON 4mg i.v. | |
| Outcomes | All outcomes. Postop 0‐24; 24‐48; 0‐48 hours. | |
| Notes | Male/female incidences not reported. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | High risk | C ‐ Inadequate |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 81 adults; 45 women; knee replacement; mean 66; exc' pregnant, COPD, study drug allergy, chronic pain/opioid, RA | |
| Interventions | Induction Either: PLACEBO; or intrathecal MORPHINE 250 mcg +/‐ CLONIDINE 25 or 75 mcg | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; Y; N; N | |
| Participants | 160 women; gynaecological; ASA1,2; 18‐50 (mean 33); exc' study drug allergy | |
| Interventions | Induction Either: DROPERIDOL 20 mcg/kg i.v.; or ONDANSETRON 4mg | |
| Outcomes | Nausea; vomiting; rescue antiemetic. PACU; 0‐1; 1‐24 hours. | |
| Notes | Rescue antiemetic only 0‐1 hour, vomiting 1‐24 hours. Nausea commonest 1‐24 hours. No side effects. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 376 adults; 176 women; eyes; 13‐82 years | |
| Interventions | Postoperative twice Either: PLACEBO twice; or TRIMETHOBENZAMIDE 200mg i.m. twice | |
| Outcomes | See notes. | |
| Notes | Incidences not reported. Unclear how retching categorized. Male/female incidences not reported. Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 68 adults; 36 women; lap' chole'; ASA1,2 | |
| Interventions | Intraoperative Either: NO TREATMENT; or ONDANSETRON 4mg i.v. | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐2; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; Y; N; N | |
| Participants | 125 adults; 90 analyzed; 72 women; 20‐60 (mean 41); lap' chole'; exc' pregnant/breastfeeding, glaucoma, GI/renal obstruction, antiemetic/‐cholinergic/‐histamine, post‐hoc protocol breach | |
| Interventions | Preoperative Either: PLACEBO; or SCOPOLAMINE 1.5mg patch | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Unclear how retching categorized. Unclear if a nauseated vomiter categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; N; N; N | |
| Participants | 90 children; 41 girls; dental; 4‐12 (mean 7); ASA1; exc' antiemetic, ASA>1, infection, study drug allergy | |
| Interventions | Intraoperative Either: PLACEBO; or ONDANSETRON 0.15mg/kg (to 8mg) | |
| Outcomes | Vomiting; rescue antiemetics. Postop 0‐2; 2‐5; 5‐24; 0‐24 hours. | |
| Notes | Rescue antiemetic commonest 2‐5 hours. Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; N; N; N | |
| Participants | 120 women; D&C; ASA1; 18‐50; exc' drug abuse, musculoskeletal/psychological/vestibular/neurological disease | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 0.625mg i.v. | |
| Outcomes | All outcomes. Postop 0‐2 hours. | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 150 women; laparoscopy; ASA1,2 | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 0.625 or 1.25mg i.v.; or METOCLOPRAMIDE 10mg; or ONDANSETRON 4mg | |
| Outcomes | PACU; PACU discharge‐24 hours | |
| Notes | Unclear numbers per group. Unclear how retching categorized. Side effect "droperidol sedative". | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 393 children; no sex data; 2‐14 (mean 6); strabismus | |
| Interventions | Induction Either: DROPERIDOL 50 mcg/kg i.v.; or MIDAZOLAM 50 mcg/kg | |
| Outcomes | Vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 276 children; no sex data; tonsil +/‐ adenoids; 2‐12 (mean 7); exc' study drug allergy | |
| Interventions | Induction Either: DROPERIDOL 50 mcg/kg i.v.; or ONDANSETRON 150 mcg/kg | |
| Outcomes | Vomiting; rescue antiemetic. Hospital; discharge‐8; 8‐24; 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 215 children; 115 girls; tonsil +/‐ adenoid; mean 7; exc' study drug allergy | |
| Interventions | Induction Either: PLACEBO; or MIDAZOLAM 75 mcg/kg i.v. | |
| Outcomes | Vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. No side effects. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 233 children; no sex data; tonsil +/‐ adenoid; 2‐14 (mean 7); ASA1; exc' study drug allergy | |
| Interventions | Preoperative Either: PLACEBO; or oral ONDANSETRON 0.1mg/kg | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐4; 4‐8; 8‐24; 0‐24 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; N; N; N | |
| Participants | 140 children; no sex data; ASA1; 2‐12 (mean 7); tonsil +/‐ adenoid; exc' study drug allergy, sleep apnoea | |
| Interventions | Induction Either: PLACEBO; or DEXAMETHASONE 150 mcg/kg i.v. | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐6; 6‐14; 14‐24; 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Retching not categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; N; N | |
| Participants | 230 children; 114 girls; tonsil +/‐ adenoid; 2‐12 (mean 7); exc' study drug allergy, sleep apnoea, ASA>2 | |
| Interventions | Induction Either: DEXAMETHASONE 150 mcg/kg (max 8mg) i.v.; or PERPHENAZINE 70 mcg/kg (max 5mg) | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐6; 6‐24; 0‐24 hours, | |
| Notes | Side effects not recorded. Retching categorized as nausea. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; N; N; N | |
| Participants | 240 children; no sex data; ASA1,2; 2‐12 (mean 7); exc' sleep apnoea, study drug allergy | |
| Interventions | Induction Either: ONDANSETRON 50 or 150 mcg/kg i.v. | |
| Outcomes | Vomiting. Hospital; discharge; next day (time unclear); 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 260 children; no sex data; 2‐12 (mean 7); tonsil +/‐ adenoid; exc' study drug allergy | |
| Interventions | Induction Either: PLACEBO; or PERPHENAZINE 70 mcg/kg i.v. | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐1; 1‐5; 0‐24; 24‐48; 0‐48 hours. | |
| Notes | No side effects. Retching not categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; N; N; N | |
| Participants | 197 children; strabismus; 2‐14 (mean 6); ASA1; exc' study drug allergy, illness | |
| Interventions | Intraoperative Either: DEXAMETHASONE 150 mcg/kg and ONDANSETRON 50 mcg/kg; or ONDANSETRON 150 mcg/kg AND PLACEBO | |
| Outcomes | Vomiting. 0 to about 3; 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Retching not categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 216 children; 118 girls; 2‐12 (mean 7); ASA1,2; tonsil +/‐ adenoid; exc' study drug allergy, sleep apnoea, ASA>2 | |
| Interventions | Induction Either: ONDANSETRON 150 mcg/kg i.v.; or PERPHENAZINE 70mcg/kg | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | No side effect. Retching not categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; N; N | |
| Participants | 200 children; no sex data; strabismus; 2‐14 (mean 6); exc' study drug allergy, ASA>1 | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 50 mcg/kg i.v. | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐1; 1‐3; 0‐3; 3‐24; 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 82 children; no sex data; strabismus; 1‐13 years; ASA1,2 | |
| Interventions | Induction Either: DROPERIDOL 10 or 20 or 40 or 80 mcg/kg i.v. | |
| Outcomes | See notes. Postop 0‐6 hours. | |
| Notes | Group sizes unclear. Unclear if nauseated retching vomiters categorized once, twice or thrice. Male/female incidences not recorded. Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; Y; N; N | |
| Participants | 215 adults; 172 women; lap' chole' | |
| Interventions | Induction Either: DROPERIDOL 0.625mg i.v. AND METOCLOPRAMIDE 10mg; or ONDANSETRON 4mg AND SALINE | |
| Outcomes | Rescue antiemetic. PACU | |
| Notes | Male/female incidences not reported. Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; N; N | |
| Participants | 212 adults; 161 women; lap' chole'; ASA1,2; exc' post‐hoc conversion | |
| Interventions | Induction Either: PERPHENAZINE 5mg i.v.; or DROPERIDOL 0.625mg AND METOCLOPRAMIDE 10mg/ONDANSETRON 4mg | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐4; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; N; N | |
| Participants | 132 children; no sex data; tonsil +/‐ adenoid; ASA1,2; 2‐12 (mean 6) | |
| Interventions | Induction Either: PLACEBO; or METOCLOPRAMIDE 0.25mg/kg i.v.; or ONDANSETRON 0.15mg/kg | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐5 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 201 adults; 105 women; various surgeries; ASA1‐3; mean 40; exc' ENT/eye surgery, NG, study drug contraindication, antiemetic/anticholinergic, drug abuse | |
| Interventions | Preoperative 8 hours Either: PLACEBO; or HYOSCINE patch (1.5mg) | |
| Outcomes | All outcomes. Postop 0‐8; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Participants | 135 children; 73 girls; strabismus; 2‐15 (mean 7); ASA1,2; exc' antiemetic | |
| Interventions | Induction Either: PLACEBO; or DEXAMETHASONE 1mg/kg iv (max 25mg); or ONDANSETRON 100 mcg/kg (max 4mg) | |
| Outcomes | Nausea or vomiting; rescue antiemetic. Postop 0‐6; 6‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | D ‐ Not used |
| Methods | N; B; Y; Y; N; N | |
| Participants | 210 women; gynaecological; ASA1,2; exc' N&V/opioid/antiemetic, opioid, pregnant, reflux | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 4mg i.v. | |
| Outcomes | All outcomes Postop 0‐1; 1‐5; 5‐24; 0‐24 hours. | |
| Notes | Rescue antiemetic commonest 0‐1 hour. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 150 children; 72 girls; tonsil +/‐ adenoid; 2‐12 (mean 6); ASA 1,2; exc' antiemetic, antihistamine, psychoactive drug, DM | |
| Interventions | Induction Either: PLACEBO; or DOLASETRON 0.5mg/kg iv (max 25mg); or ONDANSETRON 0.15mg/kg (max 4mg) | |
| Outcomes | Vomiting; rescue antiemetic. PACU; discharge‐24; 24‐48; 0‐48 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 125 adults; 58 women; mean 45; ENT | |
| Interventions | Intraoperative Either: PLACEBO; or METOCLOPRAMIDE 20mg i.v.; or ONDANSETRON 4mg; or DROPERIDOL 0.625mg +/‐ METOCLOPRAMIDE 20mg | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop recovery‐5; 0‐24; 0‐7 days. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 105 adults; ENT surgery | |
| Interventions | Intraoperative Either: PLACEBO; or DROPERIDOL 0.625mg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop time unclear. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 315 adults; 158 women; ENT surgery | |
| Interventions | Intraoperative Either: PLACEBO; or DROPERIDOL 0.625mg i.v.; or ONDANSETRON 4mg | |
| Outcomes | Postop | |
| Notes | Incidences not reported. Male/female incidences not reported. Side effects not reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; N; N | |
| Participants | 75 adults; 35 women; ENT; ASA1,2; 20‐70 (mean 43); exc' antiemetic, disease, drug abuse, pregnant, obese | |
| Interventions | Induction AND intraoperative Either: PLACEBO twice; or PLACEBO then ONDANSETRON 4mg i.v.; or ONDANSETRON 4mg i.v. then PLACEBO | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐2; 24 hours. | |
| Notes | Outcomes commonest 0‐2 hours. Retching categorized as vomiting. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; N; N | |
| Participants | 180 women; laparoscopy; ASA1,2; 18‐65 (mean 32); exc' antiemetic, pregnant, obese, NG, liver disease | |
| Interventions | Preoperative Either: PLACEBO; or ONDANSETRON 8mg i.v. | |
| Outcomes | Nausea; vomiting. Postop 0‐2; 0‐24 hours. | |
| Notes | Retching categorized as vomiting. Unclear if vomiters/retchers nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 134 women; lap' chole' or gynaecological; ASA1,2; exc' antiemetic/N&V, benzodiazepine, phenothiazine, allergy, epilepsy, reflux, liver/renal disease | |
| Interventions | Induction Either: DROPERIDOL 75 mcg/kg (max 5mg) i.v.; or ONDANSETRON 4mg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐1; 1‐6; 6‐12; 12‐24 hours. | |
| Notes | Rescue antiemetic only 0‐1 hour. Nausea and vomiting commonest 0‐1 hour. Side effect "no difference". Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | High risk | C ‐ Inadequate |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 130 adults; 61 women; orthopaedic; mean 69; ASA1‐3; exc' opioid/antiemetic, motion sickness, pruritus, study drug allergy, steroid | |
| Interventions | Induction Either: DEXAMETHASONE 8mg iv; or ONDANSETRON 8mg iv; or both | |
| Outcomes | All outcomes. Postop 0‐4; 4‐8; 8‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. No side effects. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 50 women; gynaecological; 20‐70 | |
| Interventions | Intraoperative Either: DROPERIDOL 1.25mg i.v.; or METOCLOPRAMIDE 10mg i.v. | |
| Outcomes | See notes. | |
| Notes | "The incidence of PONV was significantly higher in the metoclopramide group than the droperidol group". No incidences given. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 60 men; herniae; ASA1; mean 23; exc' study drug allergy | |
| Interventions | Induction AND intraoperative Either: PLACEBO; or intrathecal NEOSTIGMINE 50 or 100 mcg | |
| Outcomes | Nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 60 men; herniae; ASA1; exc' study drug allergy, contraindication to spinal | |
| Interventions | Induction Either: PLACEBO; or DEXAMETHASONE 10mg i.v. | |
| Outcomes | Nausea; vomiting. Postop 0‐24 hours. | |
| Notes | Retching categorized as vomiting. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 161 women; gynaecological; ASA1,2; exc' pregnant, antiemetic/psychotropic, obese | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 0.625 or 1.25mg i.v.; or ONDANSETRON 4mg | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐1;1‐6; 6‐24; 0‐24 hours. | |
| Notes | Rescue antiemetic only 0‐24 hours. Side effects not recorded. Retching categorized as vomiting. Unclear if vomiters/retchers nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; Y; N; N | |
| Participants | 164 women; ASA1,2; laparoscopy; exc' antiemetic, antipsychotic, obese, pregnant, vomiting | |
| Interventions | Induction AND intraopeative Either: PLACEBO twice; or PLACEBO then ONDANSETRON 4mg i.v.; or ONDANSETRON 4mg then PLACEBO; or ONDANSETRON 2mg twice | |
| Outcomes | All outcomes. Postop 0‐5; 0‐24 hours. | |
| Notes | Side effects not recorded. Retching categorized as vomiting. Unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 218 women; hysterectomy; ASA1,2; exc' pregnant, obese, N&V/antiemetic/antipsychotic, drug abuse, renal/liver/cardiac/metabolic/endocrine | |
| Interventions | Intraoperative Either: PLACEBO; or PALONOSETRON 0.1 or 0.3 or 1.0 or 3.0 or 30 mcg/kg i.v. | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐2; 0‐12; 0‐24 hours. | |
| Notes | Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 135 adults; 85 women; mean 55; various surgeries; ASA1‐3; exc' pregnant, menstrual, obese, N&V/antiemetic/psychoactive, cardiac/lung/CNS/metabolic/endocrine disease | |
| Interventions | Intraoperative Either: PLACEBO; or DOLASETRON 12.5mg iv; or ONDANSETRON 4mg | |
| Outcomes | All outcomes. Postop 0‐1; 1‐24; 0‐24 hours. | |
| Notes | Nausea and vomiting commonest 1‐24 hours, rescue antiemetic 0‐1 hour. Male/female incidences not reported. Side effects not reported. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 60 adults; no sex data; 50‐83 (mean 69); ASA1‐3; exc' urinary retention, cardiac disease, glaucoma | |
| Interventions | Preoperative twice Either: PLACEBO then oral DIAZEPAM 5‐15mg/PROMETHAZINE 10mg; or SCOPOLAMINE patch then oral PROMETHAZINE 10mg | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 142 adults; no sex or age data; ASA1,2 | |
| Interventions | Intraoperative Either: PLACEBO; or DROPERIDOL 0.625 or 1.25mg i.v.; or ONDANSETRON 2 or 4mg i.v.; or THIETHYLPERAZINE 5 or 10mg i.v. | |
| Outcomes | Vomiting; nausea or vomiting; rescue antiemetic. Postop 0‐2; 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 45 women; gynaecological; ASA1,2; mean 29; exc' antiemetic, hypertension, disease | |
| Interventions | Intraoperative Either: PLACEBO; or DROPERIDOL 0.04mg/kg i.m.; or EPHEDRINE 0.5mg/kg | |
| Outcomes | Nausea; vomiting. PACU; PACU‐2; 2‐12 hours. | |
| Notes | Nausea commonest PACU, vomiting PACU‐2 hours. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; N; N | |
| Participants | 101 adults; 66 women; laparoscopy; mean 43; ASA1,2; exc' study drug contraindicated, antiemetic, motion sickness/PONV/N&V | |
| Interventions | Postoperative six times Either: PLACEBO; or oral ONDANSETRON 8mg each time | |
| Outcomes | Nausea; vomiting. Postop 4‐24; 24‐72 hours. | |
| Notes | Nausea commonest 4‐24 hours, vomiting commonest 24‐72 hours. Male/female incidences not reported. No side effects. Retching categorized as vomiting. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 177 women; gynaecological; pre‐menopausal 19‐53 (mean 36); ASA1,2; exc' GI/liver/renal disease, antiemetic/N&V, pregnant/breastfeeding, BMI>35 | |
| Interventions | Induction Either: DEXAMETHASONE 8mg i.v.; or ONDANSETRON 4mg i.v.; or both | |
| Outcomes | All outcomes. Postop 0‐3; 3‐12; 12‐24; 0‐24 hours. | |
| Notes | Outcomes commonest 3‐12 hours. Unclear if vomiters retched or nauseated or if nauseated vomiter categorized once or twice. Retching categorized separately. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 96 women; superficial surgery; ASA1,2; 18‐65 (mean 41); exc' study drug contraindication, pregnant | |
| Interventions | Preoperative AND intraoperative Either: PLACEBO; or PLACEBO then DROPERIDOL 1.25mg i.v.; or SCOPOLAMINE patch 1.5mg then PLACEBO | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐2; 2‐24 hours. | |
| Notes | Rescue antiemetic only 0‐2 hours. Nausea and vomiting commonest 2‐24 hours. Unclear how retching categorized or if nauseated vomiter categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 40 adults; 17 women; ENT; 24‐54 (mean 37); ASA1,2; exc' study drug contraindication | |
| Interventions | Intraoperative Either: PLACEBO; or ATROPINE 0.01mg/kg iv AND EDROPHONIUM 1mg/kg | |
| Outcomes | Nausea or vomiting. Postop 0‐3 hours. | |
| Notes | Male/female incidences not reported. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 40 women; gynaecological; mean 44; ASA1,2 | |
| Interventions | Preoperative AND postoperative Either: PLACEBO twice; or ONDANSETRON 4mg i.v. twice | |
| Outcomes | Nausea; vomiting. PACU; PACU ‐ 8 hours. | |
| Notes | Outcomes commonest PACU. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 40 women; gynaecological; 21‐77 (mean 42); exc' glaucoma, study drug allergy | |
| Interventions | Preoperative Either: PLACEBO; or SCOPOLAMINE 1.5mg patch | |
| Outcomes | All outcomes Postop 0‐15; 15 min to 1 hour; 1‐2; 2‐3; 3‐4; 4‐5; 5‐6; 0‐6 hours. | |
| Notes | Nausea commonest 15 min to 1 hour, vomiting 0‐15 min. Unclear how retching categorized or if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; N; N | |
| Participants | 61 adults; 37 women; 50‐80 (mean 64); gynaecological/general | |
| Interventions | Preoperative Either: PLACEBO; or SCOPOLAMINE patch | |
| Outcomes | Vomiting. Postop 0‐18 hours. | |
| Notes | Male/female incidences not reported. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 60 women; D&C; exc' contraindications to study drug | |
| Interventions | Preoperative Either: PLACEBO; or oral LORMETAZEPAM 1mg; or MORPHINE 30mg | |
| Outcomes | Nausea; vomiting. Postop 0‐24 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; N; N | |
| Participants | 70 children; 31 girls; tonsil + adenoids; exc' cardiac/NM disease, bleeding disorder | |
| Interventions | Induction Either: PLACEBO; or DEXAMETHASONE 1 mg/kg (max 10mg) i.v. | |
| Outcomes | Vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. No side effects. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; N | |
| Participants | 40 adults; laparotomies; ASA1,2; exc' antiemetic | |
| Interventions | Induction Either: ONDANSETRON 4mg i.v. +/‐ DEXAMETHASONE 8mg i.v. | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Side effects not reported. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 40 children; 10 girls; strabismus; ASA1,2; 3‐12 (mean 6); exc' antiemetic, retardation | |
| Interventions | Induction Either: PLACEBO saline iv; or TROPISETRON 1mg/msq | |
| Outcomes | Nausea or vomiting; rescue antiemetic. Postop 0‐2; 2‐6; 6‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 30 women; hysterectomy; ASA1,2; 18‐65 (mean 46); | |
| Interventions | Preoperative thrice AND induction Either: NO TREATMENT thrice then ONDANSETRON 4 or 8mg i.v.; or oral ONDANSETRON 8mg thrice then NO TREATMENT | |
| Outcomes | Nausea; vomiting. Postop 0‐1; 1‐4; 4‐24; 0‐24 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 180 children; 77 girls; 3‐16 (mean 7); strabismus | |
| Interventions | Induction Either: NO TREATMENT; or LIGNOCAINE 2 mg/kg i.v.; or ONDANSETRON 5mg/m2 | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐6; 0‐24; 24‐48 hours. | |
| Notes | Rescue antiemetic only 0‐24 hours, vomiting commonest 0‐24 hours. Male/female incidences not reported. Side effects not reported. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; N; N; Y; Y | |
| Participants | 80 women; hysterectomy; ASA1‐3; mean 53; exc' liver/renal disease, chemotherapy, study drug allergy, obese, antiemetic/N&V, psychiatric | |
| Interventions | Preoperative AND induction Either: PLACEBO/oral DOLASETRON 50mg then NO TREATMENT/DROPERIDOL 2.5mg i.v. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐4 hours. | |
| Notes | Side effects not recorded. Categorized by severest symptom (vomiting>retching>nausea). I combined retching and vomiting. Unclear if retchers/vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 32 women; gynaecological; exc' PONV, inner ear problems, obese, reflux risk, antiemetic | |
| Interventions | Preoperative Either: DROPERIDOL 5mcg/kg or 0.01mg/kg i.v. | |
| Outcomes | All outcomes. PACU; PACU‐6 hours. | |
| Notes | Nausea or vomiting only PACU. Other outcomes commonest PACU‐6 hours. No side effects. Unclear how retching categorized or if nauseated vomiter categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 121 women; gynaecologial; mean 50; exc' >ASA2, >65, study drug allergy, antiemetic. | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 4mg i.v.; or TROPISETRON 5mg | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐16; 0‐24 hours. | |
| Notes | Side effect "no severe reaction". Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 50 adults; 16 women; ASA1,2; orthopaedic; exc' ASA>2, PONV, GI disease, study drug contraindication, N&V | |
| Interventions | Postoperative Either: PLACEBO; or DEXAMETHASONE 150 mcg/kg | |
| Outcomes | All outcomes. Postop 4; 8; 12; 24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Categorized by severest symptom (vomiting>retching>nausea) | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 62 adults; 42 women; ENT/eyes/gynaecological; 16‐75 (mean 39); | |
| Interventions | Preoperative Either: NO TREATMENT; or oral ONDANSETRON 4mg | |
| Outcomes | Vomiting. Postop 0‐10; 10‐60 minutes; 1‐6; 6‐12; 12‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 80 adults; 59 women; 17‐65 (mean 43) | |
| Interventions | Preoperative Either: NO TREATMENT; or oral ONDANSETRON 4 or 8 or 16mg | |
| Outcomes | Vomiting. Postop 0‐10; 10‐60 minutes; 1‐6; 6‐12; 12‐24 hours. | |
| Notes | Vomiting commonest 1‐6 hours. Male/female incidences not recorded. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 44 children; no sex data; general; ASA1; 1‐6 (mean 4); exc' contraindication to caudal | |
| Interventions | Induction Either: PLACEBO; or caudal NEOSTIGMINE 2 mcg/kg | |
| Outcomes | Vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 100 women; gynaecological; ASA1,2; 30‐56 (mean 43); exc' motion sickness/PONV, obese, study drug allergy, liver disease | |
| Interventions | Preoperative AND induction Either: PLACEBO twice; or PLACEBO then ONDANSETRON 4mg i.v.; or DIMENHYDRINATE 50mg i.m. then PLACEBO; or both | |
| Outcomes | Nausea; vomiting. Postop 0‐2; 2‐6; 6‐24 hours. | |
| Notes | Outcomes commonest 0‐2 hours. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 60 children; no sex data; strabismus; 3‐14 (mean 11); ASA1,2 | |
| Interventions | Preoperative Either CHLORPROMAZINE 0.2mg/kg i.v.; or LIDOCAINE 1.5mg/kg; or DROPERIDOL 0.075mg/kg AND LIDOCAINE 1.5mg/kg | |
| Outcomes | Nausea or vomiting. 0‐24 hours. | |
| Notes | Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; N; N | |
| Participants | 120 pregnant women; Caesarean; ASA1,2 | |
| Interventions | Intraoperative Either: PLACEBO; or DEXAMETHASONE 8mg i.v.; or DROPERIDOL 1.25 mg | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Unclear how retching categorized or if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; N; N | |
| Participants | 160 women; STOP; ASA1,2; 20‐45 (mean 32); exc' renal, blood/liver disease, drug abuse, antiemetic | |
| Interventions | Induction Either: PLACEBO; or DEXAMETHASONE 8mg i.v.; or DROPERIDOL 1.25mg; or both | |
| Outcomes | All outcomes. Postop 0‐2; 2‐24; 0‐24 hours. | |
| Notes | Retching categorized as vomiting. Unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; N; N | |
| Participants | 120 women; hysterectomy; ASA1,2; 35‐55 (mean 45); exc' motion sickness/PONV, GI disease, antiemetic | |
| Interventions | Intraoperative Either: PLACEBO; or DEXAMETHASONE 5mg i.v.; or METOCLOPRAMIDE 10mg | |
| Outcomes | All outcomes. Postop 0‐6; 0‐12; 0‐24 hours. | |
| Notes | No side effects. Retching categorized as vomiting, unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; N; N | |
| Participants | 70 women; hysterectomy; mean age 43; ASA1,2; exc' motion sickness/PONV, GI disorder, disease, opioid, antiemetic | |
| Interventions | Intraoperative Either: PLACEBO; or ONDANSETRON 4mg iv | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 150 women; gynaecological; 18‐50 (mean 33); ASA1,2; 40‐70 kg; exc' antiemetic, cardiac or lung disease | |
| Interventions | Intraoperative Either: PLACEBO; or DROPERIDOL 0.5mg i.v.; or METOCLOPRAMIDE 5mg | |
| Outcomes | Vomiting. Postop 0‐6; 6‐24 hours. | |
| Notes | Vomiting commonest 6‐24 hours. No side effects. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; Y; Y; Y | |
| Participants | 200 children; 68 girls; various surgeries; ASA1, 2; 2‐11 (mean 5); exc' liver disease | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 0.1mg/kg i.v. | |
| Outcomes | All outcomes. Postop 0‐1; 1‐2; 2‐3; 3‐4; 0‐4 hours. | |
| Notes | Male/female incidences not reported. No side effects. Retching categorized as nausea. Unclear if vomiter/retcher nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; Y; N; Y | |
| Participants | 66 adults; 38 women; laparotomy; ASA 1, 2; exc' opioid, chronic pain | |
| Interventions | Postoperative PCA Either: PLACEBO; or MAGNESIUM 30mg/ml; or KETAMINE 1mg/ml | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; N; Y | |
| Participants | 144 women; gynaecological; ASA1,2 | |
| Interventions | Preoperative Either: PLACEBO; or DIMETINDENE 12mg i.v. AND CIMETIDINE 600mg | |
| Outcomes | Vomiting. Postop 0‐24 hours. | |
| Notes | Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; N; N | |
| Participants | 42 women; gynaecological; ASA1,2; 18‐65 (mean 40) | |
| Interventions | Preoperative AND postoperative Either: PLACEBO; or HYOSCINE (0.5mg /3 days) patch | |
| Outcomes | Nausea; vomiting; rescue antiemetic Postop 0‐24; 24‐48; 48‐72 hours. | |
| Notes | Nausea commonest 24‐48 hours, vomiting 0‐24 hours. Retching categorized as vomiting. Unclear if nauseated retcher/vomiter categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 92 women; gynaecological; exc' <18 >65, CNS/renal/liver/cardiac/metabolic/endocrine disease, antiemetic, NG | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 8mg i.v. | |
| Outcomes | Nausea; vomiting; rescue antimetic. Postop 0‐24 hours. | |
| Notes | Incidence of specific side effects not reported. Retching categorized as vomiting. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 90 participants; no sex or age data; middle ear; ASA1,2 | |
| Interventions | Preoperative Either: DEXAMETHASONE 0.15mg/kg iv; or ONDANSETRON 0.1mg/kg; or both | |
| Outcomes | All outcomes. Postop 0‐4; 4‐24 hours. | |
| Notes | Nausea commonest 0‐4 hours, vomiting and 'nausea or vomiting' 4‐24 hours. Male/female incidences not reported. Unclear if retchers/vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 100 adults; 32 women; oral | |
| Interventions | Intraoperative Either: PLACEBO; or DROPERIDOL 0.014mg/kg i.v. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐1; 1‐6; 6‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not reported. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 52 women; Caesarean; ASA1,2; exc' chronic pain, foetus <36/40, study drug allergy | |
| Interventions | Induction Either: PLACEBO; or intrathecal MIDAZOLAM 1mg | |
| Outcomes | Nausea or vomiting. Postop 1; 6; 24 hours | |
| Notes | Nausea or vomiting commonest at one hour. No side effects. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 200 children & adults; female 76; eyes; 1‐83 (mean 43); | |
| Interventions | Intraoperative Either: PLACEBO; or DROPERIDOL 0.035mg/kg i.v.; or METOCLOPRAMIDE 0.15mg/kg; or PROCHLORPERAZINE 0.18mg/kg i.m. | |
| Outcomes | Vomiting. Postop 0‐24 hours. | |
| Notes | No side effects. Unclear if retching vomiters categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 374 patients; no sex or age data; ENT | |
| Interventions | Induction Either: PLACEBO/ONDANSETRON 0.06mg/kg/PROCHLORPERAZINE 0.1mg/kg i.v. and NO TREATMENT; or NO TREATMENT i.v. and PROCHLORPERAZINE 0.2mg i.m. | |
| Outcomes | Nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; Y; Y; Y | |
| Participants | 148 adults and children; 69 female; tympanoplasty; 7‐61 (mean 30); ASA1,2; exc' pregnant, antiemetic | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 0.06mg/kg i.v.; or PROCHLORPERAZINE 0.1 or 0.2mg/kg i.m. | |
| Outcomes | All outcomes. PACU; PACU ‐ 24 hours. | |
| Notes | Rescue antiemetic only PACU‐24 hours. Other outcomes commonest PACU‐24 hours. Male/female incidences not reported. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 282 children & adults; female 109; 2‐65 (mean 18); tonsil +/‐ adenoids; ASA1,2 | |
| Interventions | Induction Either: PLACEBO; or PROCHLORPERAZINE 0.1 or 0.2mg/kg i.v. AND NO TREATMENT i.m.; or ONDANSETRON 0.06mg/kg i.v. AND NO TREATMENT | |
| Outcomes | All outcomes. Postop 0‐1; 1‐24 hours. | |
| Notes | Outcomes commonest 1‐24 hours. Male/female incidences not reported. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 220 adults; 46 female; ENT; ASA1,2; exc' antiemetic, pregnant | |
| Interventions | Induction Either: PLACEBO/ONDANSETRON 0.06mg/kg i.v./PROCHLORPERAZINE 0.1mg/kg i.v.; or PROCHLORPERAZINE 0.2mg/kg i.m. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; Y; Y; Y | |
| Participants | 143 children; no sex data; 1‐10; ENT or general | |
| Interventions | Preoperative Either: oral DIAZEPAM 0.25mg/kg or TRIMEPRAZINE 4mg/kg +/‐ oral DROPERIDOL 0.2mg/kg | |
| Outcomes | Vomiting. Postop 0‐12 hours. | |
| Notes | Male/female incidences not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; Y; Y; Y | |
| Participants | 149 children; no sex data; 1‐10 (mean 5); adenotonsillectomy | |
| Interventions | Preoperative Either: PLACEBO; or oral DIAZEPAM 0.5mg/kg; or oral PENTOBARBITONE 3mg/kg; or TRIMEPRAZINE 4mg/kg | |
| Outcomes | Vomiting; rescue antiemetic. Postop time unclear. | |
| Notes | Male/female incidences not recorded. No side effects. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; N; Y; Y | |
| Participants | 80 children; 44 girls; ASA1,2; strabismus; 1‐12 (mean 6); exc' antiemetic/N&V | |
| Interventions | Induction Either: PLACEBO; or DIMENHYDRINATE 0.5mg/kg (max 25mg) i.v. | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐5; 5‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. No side effects. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 148 adults; lap' chole' | |
| Interventions | Induction AND intraoperative Either: PLACEBO twice; or PLACEBO then ONDANSETRON 4mg i.v.; or ONDANSETRON 4mg then PLACEBO | |
| Outcomes | Vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Retching categorized as nausea. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; N; N | |
| Participants | 120 women; laparoscopies; ASA1,2; 20‐40 (mean 33); exc' opioids/antiemetic | |
| Interventions | Preoperative AND postoperative Either: PLACEBO twice; or PLACEBO AND DROPERIDOL 1.25mg i.v. then PLACEBO; or oral GINGER 1g AND PLACEBO i.v. then oral GINGER 1g; or oral GINGER 1g AND DROPERIDOL 1.25mg i.v. then oral GINGER 1g | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | No side effects. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 118 women; STOP; >18 (mean 26); exc' study drug allergy | |
| Interventions | Induction Either: PLACEBO; or METOCLOPRAMIDE 10 or 20mg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐half; half ‐1; 1‐2; 2‐4 hours. | |
| Notes | Outcomes commonest half an hour‐one hour. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 928 adults; 528 women; various surgeries; exc' liver, renal & cardiac failure, antiemetic | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 4mg i.v. | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 42 children; 23 girls; tonsil +/‐ adenoid; ASA1,2; 2‐12 (mean 5); exc' ASA>2, PONV, study drug allergy | |
| Interventions | Induction Either: PLACEBO; or DEXAMETHASONE 400 mcg/kg i.v. | |
| Outcomes | Vomiting; rescue antiemetics. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | High risk | C ‐ Inadequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 50 adults; 25 women; oral; ASA1,2 | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 4mg i.v. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | High risk | C ‐ Inadequate |
| Methods | Y; B; Y; Y; N; N | |
| Participants | 119 women; laparoscopies; ASA1,2; exc' pregnant/breastfeeding, antiemetic, study drug contraindication | |
| Interventions | Intraoperative Either: PLACEBO; or intranasal METOCLOPRAMIDE 20mg | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 118 children; 50 girls; strabismus; 2‐12 (mean 5); ASA1,2; exc' study drug allergy, POV/motion sickness, liver/renal/cardiac/CNS disease, antiemetic | |
| Interventions | Intraoperative Either: PLACEBO; or DOLASETRON 0.35mg/kg or 12.5mg iv | |
| Outcomes | Nausea; rescue antiemetic. Postop 0‐3; 3‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 50 women; gynaecological; mean 44 | |
| Interventions | Postoperative PCA Either: PLACEBO; or METOCLOPRAMIDE 0.5mg/ml | |
| Outcomes | All outcomes. Postop 0‐6; 6‐12; 12‐18; 18‐24 hours. | |
| Notes | Outcomes commonest 0‐6 hours. Side effects not recorded. Vomiting categorized severe nausea. Unclear if vomiters nauseated or categorized once or more. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 50 women; gynaecological; ASA1,2 | |
| Interventions | Intraoperative AND postoperative PCA Either: CYCLIZINE 50mg i.v. then 2mg/ml PCA; or DROPERIDOL 1.25mg then 0.05mg/ml | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 60 women; STOP | |
| Interventions | Induction Either: PLACEBO; or DOMPERIDONE 10mg i.v.; or METOCLOPRAMIDE 10mg | |
| Outcomes | Vomiting. Postop 0‐2; 0‐24 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized. Unclear if nauseated vomiter categorized once or twice | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; Y; N; N | |
| Participants | 90 adults; 50 women; ASA 1,2; 30‐55; lap' chole'; exc' motion sickness, antiemetic | |
| Interventions | Induction Either: PLACEBO; or DEXAMETHASONE 8mg i.v. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Retching categorized as vomiting. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; N; N | |
| Participants | 80 women; hysterectomy; ASA1,2; 35‐60 | |
| Interventions | Intraoperative Either: PLACEBO; or DEXAMETHASONE 8mg i.v. | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | No side effects. Unclear how retching categorized or if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; N; N | |
| Participants | 120 women; thyroidectomy; exc' antiemetic | |
| Interventions | Induction Either: PLACEBO; or DEXAMETHASONE 10mg i.v.; or DROPERIDOL 1.25mg | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Retching categorized as vomiting. Unclear if vomiters/retchers nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; N; N; N | |
| Participants | 90 women; gynaecological; ASA1,2; exc' breastfeeding/pregnant, >90kg, drugs | |
| Interventions | Induction Either: PLACEBO; or DEXAMETHASONE 10mg i.v. | |
| Outcomes | All outcomes. Postop 0‐4; 4‐24; 0‐24 hours. | |
| Notes | Side effects not recorded. Retching categorized as vomiting. Unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 120 women; hysterectomy; ASA1,2; 35‐45 (mean 41); exc' motion sickness, GI disease, antiemetic | |
| Interventions | Induction AND postoperative Either: PLACEBO twice; or PLACEBO then DEXAMETHASONE 10mg i.v.; or DEXAMETHASONE 10mg then PLACEBO | |
| Outcomes | All outcomes. Postop 0‐2; 2‐24 hours. | |
| Notes | Outcomes commonest 0‐2 hours. Side effects not recorded. Retching categorized as vomiting. Unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 225 women; thyroidectomy; excluded PONV, motion sickness, GI disease, antiemetic | |
| Interventions | Induction Either: PLACEBO; or DEXAMETHASONE 1.25 or 2.5 or 5 or 10mg i.v. | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | No side effects. Retching categorized as vomiting. Unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; N; N | |
| Participants | 180 pregnant women; Caesarean; ASA1,2; 20‐35 (mean 28); exc' PONV, motion sickness, GI disease, BM <50 >90kg | |
| Interventions | Intraoperative Either: PLACEBO; or DEXAMETHASONE 2.5 or 5 or 10mg i.v. | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | No side effects. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; A; Y; Y; N; N | |
| Participants | 120 women; hysterectomy; excluded PONV, motion sickness, disease, obese, antiemetic | |
| Interventions | Intraoperative Either: PLACEBO; or DEXAMETHASONE 5mg i.v.; or TROPISETRON 5mg | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; N; N | |
| Participants | 94 children; girls 42; various surgeries; 7‐16 (mean 11); exc' ASA>2, prematurity | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 10 mcg/kg i.v. | |
| Outcomes | All outcomes. Postop 0‐3; 3‐24 hours. | |
| Notes | Outcomes commonest 0‐3 hours. Male/female incidences not reported. Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; N; N | |
| Participants | 120 adults; 72 women; lap' chole'; ASA1,2; exc' PONV/motion sickness, disease, antiemetic | |
| Interventions | Induction Either: PLACEBO; or DEXAMETHASONE 5mg iv; or TROPISETRON 2mg | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. No side effects. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; N; N | |
| Participants | 120 adults; elective surgery | |
| Interventions | Either: ONDANSETRON 8mg iv/TROPISETRON 3mg iv +/‐ DEXAMETHASONE 10mg | |
| Outcomes | Nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 120 adults; 65 women; tympanomastoid; ASA1,2; 35‐55 (mean 39); exc' motion sickness/PONV, disease, antiemetic | |
| Interventions | Induction Either: PLACEBO; or DEXAMETHASONE 5mg iv; or TROPISETRON 2mg | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 70 adults; 37 women; craniotomy; ASA1,2; mean 40 | |
| Interventions | Induction Either: PLACEBO; or GRANISETRON 3mg iv | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐72 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; N; N | |
| Participants | 120 women; gynaecological; ASA1,2; exc' N&V/antiemetic, breastfeeding | |
| Interventions | Induction AND postoperative Either: DROPERIDOL 1.25mg i.v. +/‐ ONDANSETRON 4mg then PLACEBO; or DROPERIDOL 1.25mg +/‐ ONDANSETRON 4mg twice | |
| Outcomes | Nausea or vomiting; rescue antiemetic. Postop 0‐3.5; 3.5‐24; 0‐24 hours. | |
| Notes | Side effect "no difference". Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 374 women; hysterectomy; ASA1,2; 18‐70 (mean 43); exc' cardiac, lung, liver, renal disease, antiemetic | |
| Interventions | Preoperative Either: PLACEBO; or oral DOLASETRON 25 or 50 or 100 or 200mg | |
| Outcomes | All outcomes. Postop 0‐24 hours. | |
| Notes | Retching categorized as vomiting. Unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 120 children; girls 56; ASA1,2; 0.5‐12 (mean 5); strabismus | |
| Interventions | Induction NO TREATMENT; or DROPERIDOL 75 mcg/kg i.v. | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 113 children; no sex data; various surgeries; ASA1,2; exc' CNS/liver/renal/cardiac disease, reflux risk, study drug allergy | |
| Interventions | Intraoperative Either: PLACEBO; or NEOSTIGMINE 70 mcg/kg AND GLYCOPYRROLATE 10 mcg/kg i.v.; or EDROPHONIUM 1 mg/kg AND ATROPINE 10 mcg/kg | |
| Outcomes | Vomiting; rescue antiemetic. Postoperative recovery; 0‐24 hours. | |
| Notes | Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 130 children; 67 girls; 1.5‐15 (mean 6); ASA1,2; various surgeries; exc' N&V/antiemetic, study drug contraindication | |
| Interventions | Induction Either: PLACEBO; or ONDANSETRON 10 or 50 or 100 mcg/kg i.v. | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐3; 3‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 166 women; laparoscopies; exc' pregnant, opioid/antiemetic, study drug allergy | |
| Interventions | Induction Either: CYCLIZINE 50mg i.m.; or METOCLOPRAMIDE 10mg i.v.; or ONDANSETRON 4mg i.v. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | No side effects. Unclear how retching categorized. Nausea categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; N; N; N; N | |
| Participants | 80 women; breast; ASA1‐3; 19‐80 (mean 57); exc' steroid | |
| Interventions | Induction Either: DEXAMETHASONE 4mg iv; or ONDANSETRON 4mg | |
| Outcomes | All outcomes. Postop 0‐4; 4‐8; 8‐16; 16‐24; 0‐24 hours. | |
| Notes | No side effects. Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 301 children; no sex data; strabismus; 4‐10 (mean 6); ASA1,2; exc' fever, N&V, antiemetic, CNS disease | |
| Interventions | Preoperative Either: PLACEBO; or DIMENHYDRINATE 2‐3 mg/kg suppository | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐3; 0‐6; 0‐9; 0‐12; 0‐18 hours. | |
| Notes | Male/female incidences not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 99 children; Faden; ASA1,2; 4‐10; exc' PONV, antiemetic | |
| Interventions | Preoperative Either: PLACEBO; or DIMENHYDRINATE 40‐70mg suppository | |
| Outcomes | Vomiting. Postop 0‐18 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 60 adults; 24 women; ENT | |
| Interventions | Induction Either: PLACEBO; or METOCLOPRAMIDE 0.15 mg/kg i.v. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐3; 3‐6; 6‐24; 0‐24 hours. | |
| Notes | Unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; Y; Y | |
| Participants | 80 women; breast; ASA1,2; 18‐70 (mean 44); exc' antiemetic, ASA>2, reflux, liver/renal disease | |
| Interventions | Postoperative PCA Either: PLACEBO; or ONDANSETRON 1mg/hr | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 40 adults; 16 women; orthopaedic; ASA1‐3; mean 52; exc' opioid, epilepsy, renal failure, DM, neuropathy | |
| Interventions | Postoperative Either: PLACEBO; or epidural DROPERIDOL 2.5mg | |
| Outcomes | Nausea; vomiting. Postop 0‐5 hours. | |
| Notes | Male/female incidences not reported. Unclear if nauseated retching vomiter categorized once, twice or thrice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; Y; N; N | |
| Participants | 221 adults; no sex data; orthopaedic/plastic; ASA1,2; 16‐65; exc' reflux, antiemetic, anticholinergic | |
| Interventions | Preoperative Either: PLACEBO; or HYOSCINE patch | |
| Outcomes | Nausea; vomiting. Postop 0‐24; 24‐48 hours. | |
| Notes | Outcomes commonest 0‐24 hours. Male/female incidences not recorded. Side effects not reported. Unclear how retching categorized or if nauseated vomiter categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; B; Y; Y; N; N. | |
| Participants | 60 women; hysterectomy; ASA1,2; exc' phenothiazine | |
| Interventions | Postoperative PCA Either: PLACEBO; or DROPERIDOL 0.2mg per bolus | |
| Outcomes | All outcomes Postop 0‐4; 4‐12; 12‐24; 0‐24 hours. | |
| Notes | Vomiting commonest 12‐24 hours, nausea or vomiting 0‐4 hours. Unclear how retching categorized or if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; A; Y; Y; N; N | |
| Participants | 49 women; hysterectomy; ASA1‐3; 25‐60 (mean 41); exc' motion sickness/PONV, antiemetic, prochlorperazine, epilepsy, study drug allergy | |
| Interventions | Preoperative AND postoperative four times Either: PLACEBO; or buccal PROCHLORPERAZINE 6mg each time | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐4; 4‐8; 8‐12; 12‐16; 16‐20; 20‐24; 24‐28; 28‐32; 32‐36; 36‐40; 40‐44; 44‐48 hours. | |
| Notes | Nausea commonest 8‐12 hours, other outcomes 20‐24 hours. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | N; A; Y; N; Y; Y | |
| Participants | 156 adults; 116 women; gynaecological/oral | |
| Interventions | Preoperative Either: oral DIAZEPAM 10mg; or HEPTABARBITONE 400mg; or LORAZEPAM 3mg | |
| Outcomes | Nausea or vomiting. Postop 0‐18 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 60 women; gynaecological; 16‐60 | |
| Interventions | Preoperative Either: DOMPERIDONE 10 or 15mg i.m. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐6 hours. | |
| Notes | No side effects. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; N; Y; N | |
| Participants | 527 adults; women 507; various surgery; exc' ASA>3, obese, pregnant/breastfeeding, study drug allergy, N&V/antiemetic | |
| Interventions | Induction Either: PLACEBO; or GRANISETRON 0.1 or 1 or 3mg i.v. | |
| Outcomes | All outcomes. Postop 0‐6; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects "similar". Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; N; N | |
| Participants | 232 adults; 183 women; lap' chole''; ASA1‐3; 18‐73; exc' pregnancy, breastfeeding, obese, antiemetic, DM, post‐hoc conversion/admission | |
| Interventions | Induction Either: PLACEBO; or METOCLOPRAMIDE 10mg i.v.; or ONDANSETRON 8mg | |
| Outcomes | All outcomes. Postop 0‐4; 4‐24 hours. | |
| Notes | Outcomes commonest 0‐4 hours. Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 210 adults; 194 women; gynaecological/urological; mean 33 | |
| Interventions | Preoperative Either: PLACEBO; or DIPHENIDOL 40mg i.m.; or DROPERIDOL 5mg | |
| Outcomes | Nausea; vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Retching categorized as vomiting. Unclear if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; N; N | |
| Participants | 216 adults; 39 women; cardiac; ASA2,3; 18‐80; exc' >100kg, antiemetic/N&V | |
| Interventions | Preoperative Either: oral METOCLOPRAMIDE 10mg; or ONDANSETRON 16mg | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | A ‐ Adequate |
| Methods | Y; B; Y; Y; Y; Y | |
| Participants | 60 women; gynaecological; ASA1,2 | |
| Interventions | Intraoperative AND postoperative PCA Either: DROPERIDOL 1.25 mg i.v. then 3mg/60ml; or ONDANSETRON 4mg then 8mg/60ml; or both | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐6; 6‐12; 12‐18; 0‐18 hours. | |
| Notes | Nausea commonest 0‐6 hours. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | High risk | C ‐ Inadequate |
| Methods | Y; B; Y; Y; N; N | |
| Participants | 160 women; lap' chole'; ASA1‐3; 16‐65 (mean 33); exc' heart, blood, lung, renal, liver, CNS, endocrine disease, pregnancy, obese, drug abuse, antiemetic, allergy | |
| Interventions | Induction Either: PLACEBO; or DROPERIDOL 1.25mg i.v.; or ONDANSETRON 4mg; or both | |
| Outcomes | Nausea or vomiting; rescue antiemetic. Postop 0‐3.5; 3.5‐24; 0‐24 hours. | |
| Notes | Side effect "no difference". Retching categorized as vomiting. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 84 adults; no sex data; abdominal; ASA1,2 | |
| Interventions | Either: PLACEBO; or DEXAMETHASONE 10mg iv | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; A; Y; N; Y; Y | |
| Participants | 100 women; Caesarean; ASA1,2; exc' pre/eclampsia, liver, renal, heart disease, motion sickness, pruritus, study drug allergy | |
| Interventions | Intraoperative Either: PLACEBO; or ONDANSETRON 8mg i.v. | |
| Outcomes | Nausea or vomiting; rescue antiemetic. Postop 0‐24 hours. | |
| Notes | No side effects. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; Y; Y; Y | |
| Participants | 45 adults; no sex data; perianal; ASA1; exc' study drug allergy | |
| Interventions | Induction Either: PLACEBO; or intrathecal NEOSTIGMINE 25 or 50 mcg | |
| Outcomes | Nausea; vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Unclear if vomiters nauseated or categorized once or twice. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 75 women; gynaecological; ASA1,2; mean 32; | |
| Interventions | Intraoperative Either: PLACEBO; or DROPERIDOL 2.5mg iv; or GRANISETRON 3mg; or ONDANSETRON 4mg; or TROPISETRON 5mg | |
| Outcomes | All outcomes. Postop 0‐10; 10‐15; 15‐30; 30‐60 minutes; 0‐1 hour. | |
| Notes | Nausea and vomiting commonest 10‐15 minutes, nausea or vomiting 0‐10 minutes. Side effects not recorded. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 60 adults; 49 women; thyroidectomy; ASA1,2; 22‐60 (mean 46); exc' N&V, PONV, ASA>2 | |
| Interventions | Induction Either: PLACEBO; or METOCLOPRAMIDE 0.2mg/kg i.v.; or TROPISETRON 5mg | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐2; 2‐4 hours. | |
| Notes | Outcomes commonest 0‐2 hours. Male/female incidences not reported. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 60 women; thyroidectomy/breast; ASA1,2; mean 45; exc' N&V, PONV, ASA>2 | |
| Interventions | Induction Either: PLACEBO; or TROPISETRON 2 or 5mg i.v. | |
| Outcomes | Vomiting; rescue antiemetic. Postop 0‐2; 2‐4 hours. | |
| Notes | Outcomes commonest 0‐2 hours. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; Y; N; Y; Y | |
| Participants | 60 adults; 11 women; various surgeries; 18‐80 (mean 61); exc' ASA>3, hypotension, cardiac disease, clonidine | |
| Interventions | Preoperative Either: PLACEBO; or oral CLONIDINE 3 mcg/kg | |
| Outcomes | Nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not reported. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 200 adults; 88 women; ENT; ASA1,2; 20‐75 (mean 45); exc' antiemetic, pregnancy, cardiac/CNS/renal/liver/GI/endocrine disease, drug abuse, obese | |
| Interventions | Intraoperative Either: DOLASETRON 12.5 or 25mg i.v.; or ONDANSETRON 4 or 8mg | |
| Outcomes | Nausea; vomiting; rescue antiemetic. Postop 0‐5; 5‐24; 0‐24 hours. | |
| Notes | Male/female incidences not reported. Side effects not recorded. Retching categorized as vomiting. Unclear if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; Y; N; N | |
| Participants | 80 children; 50 girls; tonsil; 4‐15; ASA1,2; exc' conditions affecting outcome | |
| Interventions | Preoperative Either: oral ERYTHROMYCIN 1mg/kg; or METOCLOPRAMIDE 0.15mg/kg | |
| Outcomes | Nausea. Postop 0‐18 hours. | |
| Notes | Male/female incidences not reported. Side effects "no extrapyramidal". Unclear how many vomited. Unclear if retchers/vomiters nauseated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | Y; B; Y; N; Y; Y | |
| Participants | 70 women; gynaecological; 18‐75 (mean 47); exc' pregnancy, confounding conditions | |
| Interventions | Intraoperative Either: PLACEBO; or TROPISETRON 5mg i.v. | |
| Outcomes | Nausea; vomiting; rescue antiemetic. PACU; PACU‐24; 0‐24 hours. | |
| Notes | No side effects. Unclear how retching categorized or if vomiters nauseated or categorized once or twice. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
| Methods | N; B; N; N; Y; Y | |
| Participants | 155 adults; 67 women; general | |
| Interventions | Preoperative Either: PLACEBO; or DIAZEPAM 5mg i.m. | |
| Outcomes | Nausea; vomiting; nausea or vomiting. Postop 0‐24 hours. | |
| Notes | Male/female incidences not recorded. Side effects not recorded. Unclear how retching categorized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk | B ‐ Unclear |
"Methods": sample size calculation ('Y' yes, 'N' no); sequence generation ('A' adequate, 'B' unclear); outcome assessor blinded ('Y' yes, 'N' no); anaesthetist blinded ('Y' yes, 'N' no); intention to treat ('Y' yes, 'N' no); complete follow up ('Y' yes, 'N' no).
"Participants"; [numbers, unless specified, are ages in years];
'ASA' American Society of Anesthesiologists' grade; 'BMI' body mass index; 'chole'' cholecystectomy; 'CNS' central nervous system; 'COPD' chronic obstructive pulmonary disease; 'D&C' dilation and curettage; 'DM' diabetes mellitus; 'exc' excluded; 'ETT' endotracheal tube; 'GI' gastrointestinal; 'HH' hiatus hernia; 'ICU' intensive care unit; 'IHD' ischaemic heart disease; 'IM' intramuscular; 'IOP' intraophthalmic pressure; 'lap'' laparoscopic; 'MH' malignant hyperthermia; 'NG' nasogastric tube; 'NM' neuromuscular; 'NSAID' non‐steroidal anti‐inflammatory drug; 'NYHA' New York Heart Association angina grade; 'STOP' suction termination of pregnancy; 'steri' sterilization.
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Abouleish 1999 | Treatment; 50% of patients were sick before the intervention. |
| Alexander 1995b | Subsequently published in more detail as Alexander 1997. |
| Alon 1987b | Published in more detail as Alon 1987 (in which Placebo is compared to two additional groups; Droperidol and a higher dose of Haloperidol (5mg)). |
| Alon 1993 | Subsequently published in more detail as Alon 1993b. |
| Ambesh 1999 | Postoperative nausea or vomiting not assessed. |
| Apfel 2003 | Design paper for the 'IMPACT' study, the results of which were subsequently published. |
| April 1996b | Published as April 1996 |
| Arain 2002 | Postoperative nausea or vomiting not assessed. |
| Aromaa 1987 | Subsequently published in more detail as Tigerstedt 1988. |
| Arun 1987 | Observational study. |
| Assaf 1975b | Patients were not aware that they were in a study. Informed consent was not sought. |
| Assaf 1975 | Patients were not aware that they were in a study. Informed consent was not sought. |
| Badalaty 1990 | Intraoperative outcomes only assessed, not postoperative. |
| Beer 2001 | Operation performed without general anaesthesia, sedation or regional blockade. |
| Benhamou 1998 | Intraoperative outcomes only assessed, not postoperative |
| Boone 2002 | Unclear what number of women were nauseated or retched or vomited intraoperatively as opposed to postoperatively. |
| Calamandrei 1994b | Duplicate of Calamandrei 1994. |
| Campbell 1993 | Subsequently published as Lewis 1994. |
| Capouet 1996b | Same as Capouet 1996. |
| Cavanaugh 1996 | No data were reported. |
| Chalmers 1984 | Preoperative incidence of nausea, not postoperative. |
| Chestnut 1989 | Preintervention nausea: droperidol group 9/41, metoclopramide group 9/40. Intraoperative postintervention nausea: droperidol group 8/41, metoclopramide group 12/40; vomiting: droperidol group 2/41, metoclopramide group 1/40. Unclear whether the preintervention and postintervention intraoperative symptomatic women had postoperative symptoms. Prophylaxis or treatment? |
| Cieslak 1995 | Subsequently published as Cieslak 1996. |
| Cole 1982 | Patients allocated to groups on the basis of hospital number (odd or even). |
| Cook‐Sather 1998 | Published subsequently as Cook‐Sather 2002. |
| Costas 1985 | Postoperative nausea or vomiting not assessed. |
| Cramb 1988 | Subsequently published in more detail as Cramb 1989. |
| Cugini1997 | Trial of ketamine premedication versus no ketamine. |
| Danzer 1997 | Postoperative nausea or vomiting not assessed. |
| De‐Oliveira 2000 | The outcomes (nausea; vomiting; nausea or vomiting; rescue antiemetics) were not assessed in this study. |
| Dershwitz 1991 | Subsequently published in more detail as Dershwitz 1992. |
| Dershwitz 1992 | Subsequently published as part of McKenzie 1993b. |
| Dershwitz 1996 | Subsequently published in more detail as Dershwitz 1998. |
| Diemunsch 1994 | Subsequently published in more detail as Wilson 1996. |
| Diemunsch 1996 | Subsequently published in more detail as Diemunsch 1998. |
| Diemunsch 1999 | Treatment of established nausea and vomiting. |
| Dundee 1966 | Unclear whether the allocation to treatment was done in a random or pseudo‐randomized way. |
| Dundee 1974 | Patients were not aware that they were in a study. Informed consent was not sought. |
| Fabling 1999 | Subsequently published in more detail as Fabling 2000. |
| Fry 1974 | The intervention was a combination of drug (metoclopramide) and intravenous fluid. |
| Fujii 1998p | Intraoperative outcomes. |
| Furst 1993 | Subsequently published in more detail as Furst 1994. |
| Gackle 1999 | Part of the subsequently published and more complete Eberhart 1999d. |
| Gan 1993 | Subsequently published in more detail as Gan 1994. |
| Gan 1994b | Subsequently published in more detail as Gan 1995. |
| Gan 1996 | Individuals allocated placebo or intervention on a non‐random basis. |
| Gan 1997b | Published subsequently as Fortney 1998. This paper addresses patient satisfaction whilst Fortney 1998 records the incidences of nausea or vomiting. |
| Gesztesi 1998 | Subsequently published as part of the more detailed Gesztesi 2000. |
| Ghaly 1987 | The study was interrupted (droperidol not given after 12 administrations) and the results were only reported for acupuncture versus cyclizine. |
| Gift 1995 | No control for either the nasal cannulae or the face tent (both supplying oxygen in the intervention groups). |
| Graczyk 1997 | Part of the subsequently published and more complete Philip 2000. |
| Grasela 1994 | Observational study. |
| Gratz 1996 | Data forms part of Pearman 1994 and Kovac 1996b, which also contain data from McKenzie 1993 and Sung 1993 and Khalil 1994. |
| Gulhas 2001 | Subsequently published in more detail as Gulhas 2003. |
| Hamid 1996 | Subsequently published in more detail as Hamid 1998. |
| Hannalah 1995 | Subsequently published as Patel 1997. |
| Helmers1992 | Part of the subsequently published Rust 1994 (and same as Kenny 1992). |
| Hill 2000 | Same patients as Fortney 1998. |
| Houchin 1992 | The analgesic action of the local anaesthetic could indirectly contribute to a reduced incidence of emetic sequelae. No data were presented on what analgesic treatment was provided to the control group. |
| Imbeloni 1986 | The postoperative incidences of nausea and vomiting were not assessed, but the intraoperative incidences were. |
| Jakobsson 1991 | The allocation of people to either group was predictable and identifiable (even dates received ketobemidone, odd date midazolam). |
| Janknegt 1999b | Published previously as Janknegt 1999. |
| Jellish 1998 | Same study as Jellish 1997. |
| Kenny 1992 | Part of the subsequently published Rust 1994 (and same as Helmers 1992). |
| Khalil 1994 | Subgroup of patients included in Pearman 1994. |
| Kivalo 1976 | Treatment, not prevention, of postoperative nausea and vomiting. |
| Klamt 1997 | Postoperative nausea or vomiting not assessed. |
| Kostopanagiotou 1998 | Outcomes of interest not assessed in this study. |
| Kovac 1992 | Same as McKenzie 1993 (and data included in Pearman 1994 etc) |
| Kovac 1996 | Subsequently published in more detail as Kovac 1996b |
| Lawhorn 1994 | Part of the subsequently published Lawhorn 1997. |
| Lindblad 1990 | Subsequently published in full detail as Beattie 1993. |
| Liu 1998 | Subsequently published as part of Liu 1999. |
| Loers 1973 | Recruitment to the diazepam group was discontinued after twenty patients because of agitation. The method of allocation was not described. |
| Loper 1988 | Subsequently published as part of the more detailed Loper 1989. |
| Lopez 1995 | Subsequently published in more detail as Lopez 1996. |
| Lopez 2000 | Same as the previously published Lopez Herrera 1998. |
| Loughrey 2002 | Postoperative nausea or vomiting not assessed. |
| Lussos 1992 | Intraoperative, not postoperative, outcomes reported. |
| Manchikanti 1984 | Postoperative nausea or vomiting not assessed. |
| Maranhäo 1988 | Recorded the incidences of intraoperative outcomes. |
| Martins 1981 | Observational study. |
| Mattila 1983 | Preoperative nausea or vomiting was recorded, not postoperative. |
| McKenzie 1993 | Same data as Pearman 1994, numbers different as not intention to treat. |
| Michaloudis 1993 | This study compares two groups with different masses of Droperidol in each: Droperidol 2.5mg versus (Droperidol 0.5mg and Metoclopramide 5mg and Hyoscine 0.1mg). |
| Mirakhur 1981 | Historical controls. |
| Morrison 1970 | Patients were not aware that they were in a study. Informed consent was not sought. |
| Nelskyla 1998b | Subsequently published in more detail as Nelskyla 1998. |
| Nortcliffe 2001 | Published subsequently with 9 additional patients as Nortcliffe 2003. |
| Okamoto 1992 | Not a prospective interventional controlled trial (appears to be a prospective observational non‐allocated study). |
| Olsson 1982 | Postoperative nausea or vomiting not assessed. |
| Olvera 1997 | Postoperative nausea or vomiting not assessed. |
| Pan 1996 | Intraoperative outcomes. |
| Parlow 1999 | Some patients experienced nausea or vomiting before the administration of the antiemetic. |
| Polati 1997 | Treatment of postoperative nausea and vomiting. |
| Powell 2000 | Postoperative nausea or vomiting not assessed. |
| Pueyo 2003 | Results published previously as Sanchez 2002. |
| Ram 1999 | Postoperative nausea or vomiting not assessed. |
| Rose 1994 | Subsequently published as part of Rose 1996. |
| Rust 1995 | Subsequently published as part of Morris 1998. |
| Sadove 1971 | The outcomes for the different groups that received different premedications could not be determined from the paper. |
| Santos 1984 | Intraoperative outcomes. |
| Scholz 1996 | Subsequently published in more detail as Scholz 1998. |
| Scuderi 1994 | Part of subsequently published Scuderi 1997. |
| Scuderi 1997b | Subsequently published in more detail as Scuderi 1999. |
| Sen 2001 | Intraoperative, not postoperative, nausea and vomiting. |
| Senders 1999 | Postoperative nausea or vomiting not assessed. |
| Sjovall 1984 | Preoperative incidences measured, but not postoperative. |
| Spelina 1984 | Intraoperative incidences measured, but not postoperative. |
| Tan 1993 | Subsequently published as part of the more detailed Watcha 1995. |
| Tang 1995 | Part of subsequently published Tang 1996. |
| Tang 1996b | Subsequently published as Tang 1998. |
| Tang 1997 | Subsequently published as Tang 1998. |
| Thune 1995 | The interventions were not controlled for and the allocation of people to either group was predictable and identifiable(date of birth odd [metoclopramide] or even [hyoscine]) |
| Tolksdorf 1991 | The allocation to the intervention groups depended upon the preferences of the children and parents. |
| Tramer 1993 | The two groups systematically received different anaesthetics. In the ondansetron group anaesthesia was induced with thiopentone and maintained with isoflurane and air. In the lidocaine group anaesthesia was induced with propofol and maintained with propofol. |
| Trapp 1989 | Allocation to study groups was not random or pseudorandom, but sequential. |
| Ummenhofer 1993 | Subsequently published in more detail as Ummnhofer 1994. |
| Unlugenc 2003 | Treatment, not prevention, of PONV. |
| Ure 1999 | Intraoperative outcomes. |
| Vener 1994 | Subsequently published as Vener 1996. |
| Wallenborn 2003 | Not a comparative trial of interventions. |
| Warriner 1995 | Subsequently published as Warriner 1997. |
| Watcha 1994 | Intraoperative outcomes. |
| Williams 1995 | Reprint of the summary of Williams 1993. |
| Zarate 1999 | Subsequently published as EA 54. |
| Zomers 1993b | Subsquentlly published in full as Zomers 1993. |
Contributions of authors
Dr Carlisle (JBC) designed and wrote the protocol for this review, wrote the review methodology, designed and conducted the literature search, extracted data and categorized them according to the listed quality criteria, analysed the results, and wrote the review.
Dr Stevenson (CS) independently extracted data from studies and categorized them on the basis of the listed quality criteria.
Sources of support
Internal sources
Torbay Special Projects Fund, UK.
External sources
No sources of support supplied
Declarations of interest
None known.
Notes
December 2016
The future of this review is being considered. This is because it includes a large number of retracted studies
Edited (no change to conclusions)
References
References to studies included in this review
- Aasboe V, Raeder JC, Groegaard B. Betamethasone reduces postoperative pain and nausea after ambulatory surgery. Anesthesia & Analgesia 1998 Aug;87(2):319‐23. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3219830
- Abdulatif M, Sanabary M. Caudal neostigmine, bupivacaine, and their combination for postoperative pain management after hypospadias surgery in children. Anesthesia & Analgesia 2002 Nov;95(5):1215‐8. [; MEDLINE: ; CN‐00410873] [DOI] [PubMed] [Google Scholar]; 3219832
- Abou Zeid H, Al‐Gahamdi A, Abdul‐Hadi M. Dolasetron decreases postoperative nausea and vomiting after breast surgery. Breast Journal of Anaesthesia 2002 Jul;8(4):216‐21. [; MEDLINE: ; CN‐00390043] [DOI] [PubMed] [Google Scholar]; 3219834
- Abramowitz MD, Oh TH, Epstein BS, Ruttimann UE, Friendly DS. The antiemetic effect of droperidol following outpatient strabismus surgery in children. Anesthesiology 1983 Dec;59(6):579‐83. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3219836
- Adducci E, Gorgoglione M, Aceto P, Congedo E, Clemente A, Gualtieri E, et al. The prophylaxis of postoperative nausea and vomiting after laparoscopic cholecystectomy. Acta Medica Romana 2002;40(4):331‐9. [; 2003425222 20031106 ISSN: 0001‐6098] [Google Scholar]; 3219838
- Agarwal A, Bose N, Gaur A, Singh U, Gupta MK, Singh D. Acupressure and ondansetron for postoperative nausea and vomiting after laparoscopic cholecystectomy. Canadian Journal of Anaesthesia 2002 Jun‐Jul;49(6):554‐60. [; MEDLINE: ; CN‐00389630] [DOI] [PubMed] [Google Scholar]; 3219840
- Ahmed AB, Hobbs GJ, Curran JP. Randomized, placebo‐controlled trial of combination antiemetic prophylaxis for day‐case gynaecological laparoscopic surgery. British Journal of Anaesthesia 2000;85(5):678‐82. [; MEDLINE: ; CN‐00334639] [DOI] [PubMed] [Google Scholar]; 3219842
- Akcabay M, Gunaydin B, Mahli A, Karadenizli Y. Ondansetron versus placebo to prevent postoperative nausea and vomiting in patients undergoing thyroidectomy. Gazi Medical Journal 1997;8(2):54‐7. [; MEDLINE: ] [Google Scholar]; 3219844
- Akkaya T, Sayin MM, Temizsoylu M, Kale O, Arik E, Alptekin A, et al. The comparison of the postoperative antiemetic and analgesic effects of granisetron and granisetron + dexamethasone [Granisetron ve granisetron + deksametazon'un postoperatif antiemetik ve analjezik ozelliklerinin karsilastirilmasi]. Turk Anesteziyoloji ve Reanimasyon 2001;29(3):113‐7. [; AN: 2001140535] [Google Scholar]; 3219846
- Alexander R, Lovell AT, Seingry D, Jones RM. Comparison of ondansetron and droperidol in reducing postoperative nausea and vomiting associated with patient‐controlled analgesia. Anaesthesia 1995;50(12):1086‐8. [; MEDLINE: ; EMBASE 1996004136] [DOI] [PubMed] [Google Scholar]; 3219848
- Alexander R, Fennelly M. Comparison of ondansetron, metoclopramide and placebo as premedicants to reduce nausea and vomiting after major surgery. Anaesthesia 1997 Jul;52(7):695‐8. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3219850
- Ali‐Melkkila T, Kanto J, Katevuo R. Tropisetron and metoclopramide in the prevention of postoperative nausea and vomiting. A comparative, placebo controlled study in patients undergoing ophthalmic surgery. Anaesthesia 1996 Mar;51(3):232‐5. [; MEDLINE: ; EMBASE 1996117379] [DOI] [PubMed] [Google Scholar]; 3219852
- Allen D, Jorgensen C, Sims C. Effect of tropisetron on vomiting during patient‐controlled analgesia in children. British Journal of Anaesthesia 1999 Oct;83(4):608‐10. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3219854
- Alon E, Caluori R, Bernoulli L, Axhausen C, Hossli G. Domperidone after ether anaesthesia. Prevention of postoperative hyperemesis. Anaesthesist 1982;31(4):185‐7. [; MEDLINE: ; EMBASE 1982114811] [PubMed] [Google Scholar]; 3219856
- Alon E, Zamboni P, Hossli G. ar. Acta Anaesthesiologica Scandinavica 1987;31:94. [; MEDLINE: ] [Google Scholar]; 3219858
- Alon E, Himmelseher S. Ondansetron in the treatment of postoperative vomiting: a randomized, double‐blind comparison with droperidol and metoclopramide. Anesthesia & Analgesia 1992 Oct;75(4):561‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3219860
- Alon E, Lenzlinger PM, Atanossoff PG. Ondansetron 4 mg versus 8 mg in the prophylaxis of postoperative nausea and vomiting after alfentanil‐supplemented inhalation anesthesia for minor gynecologic surgery. Anesthesiology 1993;79(3A):A8. [; SI:A1993LY10800008 ER] [Google Scholar]; 3219862
- Alon E, Biro P, Lenzlinger PM, Atanassoff PG. Ondansetron in the prophylaxis of postoperative nausea and vomiting: A randomized double‐blind comparison with droperidol. Anaesthesist 1994;43(8):500‐3. [; MEDLINE: ; EMBASE 1994273488] [DOI] [PubMed] [Google Scholar]; 3219864
- Alon E, Kocian R, Nett PC, Koechli OR, Baettig U, Grimaudo V. Tropisetron for the prevention of postoperative nausea and vomiting in women undergoing gynecologic surgery. Anesthesia and Analgesia 1996;82(2):338‐41. [; MEDLINE: ; AN: 1996046783] [DOI] [PubMed] [Google Scholar]; 3219866
- Alsner T, Brandt MR, Sorensen B. The postanesthetic antiemetic effect of premedication with dehydrobenzperidol before ether anesthesia. Acta Anaesthesiologica Scandinavica 1976;20(1):65‐9. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3219868
- Altintas F, Tunali Y, Utku T, Pozkurt P, Kaya G, Kose Y. Bupivacaine + neostigmine in spinal anaesthesia [Spinal anestezide bupivakain + neostigmin uygulamasi]. Turk Anesteziyoloji Ve Reanimasyon 1997;25(7):313‐7. [; EMBASE 1997305228] [Google Scholar]; 3219870
- Altunkaya H, Özer Y, Cihan A, Özkoçak I, Demirel CB. The effect of preoperative dexamethasone administration on nausea, vomiting and pain in thyroidectomy. Anestezi Dergisi 2003;11(2):131‐5. [; 2003298084 20030807 ISSN: 1300‐0578] [Google Scholar]; 3219872
- Anderson BJ, Exarchos H, Lee K, Brown TC. Oral premedication in children: a comparison of chloral hydrate, diazepam, alprazolam, midazolam and placebo for day surgery. Anaesthesia & Intensive Care 1990 May;18(2):185‐93. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3219874
- Ang C, Habre W, Sims C. Tropisetron reduces vomiting after tonsillectomy in children. British Journal of Anaesthesia 1998 Jun;80(6):761‐3. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3219876
- Aouad MT, Siddik SS, Rizk LB, Zaytoun GM, Baraka AS. The effect of dexamethasone on postoperative vomiting after tonsillectomy. Anesthesia & Analgesia 2001 Mar;92(3):636‐40. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3219878
- Apfel CC, Greim CA, Kranke P, Sefrin P, Roewer N. Propofol does more effectively reduce postoperative nausea and vomiting than volatile anesthetics with droperidol. Anesthesiology 1998 Sep;89:A52. [; SI:000075810900053 ER] [Google Scholar]; 3219880
- Apfel CC, Kranke P, Katz MH, Goepfert C, Papenfuss T, Rauch S, et al. Volatile anaesthetics may be the main cause of early but not delayed postoperative vomiting: a randomized controlled trial of factorial design. British Journal of Anaesthesia 2002 May;88(5):659‐68. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3219882
- Apfel CC, Korttila K, Abdalla M, Kerger H, Turan A, Vedder I et al [for the IMPACT investigators]. A Factorial Trial of Six Interventions for the Prevention of Postoperative Nausea and Vomiting. New England Journal of Medicine 2004;350(24):2441‐51. [; MEDLINE: ] [DOI] [PMC free article] [PubMed] [Google Scholar]; 3219884
- April MM, Callan ND, Nowak DM, Hausdorff MA. The effect of intravenous dexamethasone in pediatric adenotonsillectomy. Archives of otolaryngology ‐ head & neck surgery 1996 Feb;122(2):117‐20. [; MEDLINE: ; ISI:A1996TU72600002 ER] [DOI] [PubMed] [Google Scholar]; 3219886
- Arcioni R, della Rocca M, Romano S, Romano R, Pietropaoli P, Gasparetto A. Ondansetron inhibits the analgesic effects of tramadol: a possible 5‐HT(3) spinal receptor involvement in acute pain in humans. Anesthesia & Analgesia 2002 Jun;94(6):1553‐7. [; MEDLINE: ; CN‐00380270] [DOI] [PubMed] [Google Scholar]; 3219888
- Arfeen Z, Owen H, Plummer JL, Ilsley AH, Sorby‐Adams RA, Doecke CJ. A double‐blind randomized controlled trial of ginger for the prevention of postoperative nausea and vomiting. Anaesthesia & Intensive Care 1995 Aug;23(4):449‐52. [; MEDLINE: ; EMBASE 1995243557] [DOI] [PubMed] [Google Scholar]; 3219890
- Argiriadou H, Papaziogas B, Pavlidis T, Parlapani A, Georgiou M, Papagiannopoulou P, et al. Tropisetron vs ondansetron for prevention of postoperative nausea and vomiting after laparoscopic cholecystectomy: a randomized double‐blind, placebo‐controlled study. Surgical Endoscopy 2002 Jul;16(7):1087‐90. [; MEDLINE: ; CN‐00390727] [DOI] [PubMed] [Google Scholar]; 3219892
- Ascari C, Rhyne H, Durieux ME, Spiekermann BF. A comparison of ondansetron, droperidol and promethazine for post‐operative nausea and vomiting. British Journal of Anaesthesia 1999;82(Suppl 1):121. [; CN‐00304271] [Google Scholar]; 3219894
- Ascaso FJ, Ayala I, Carbonell P, Castro FJ, Palomar A. Prophylactic intravenous ondansetron in patients undergoing cataract extraction under general anesthesia. Ophthalmologica 1997;211(5):292‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3219896
- Awad IT, Murphy D, Stack D, Swanton BJ, Meeke RI, Shorten GD. A comparison of the effects of droperidol and the combination of droperidol and ondansetron on postoperative nausea and vomiting for patients undergoing laparoscopic cholecystectomy. Journal of Clinical Anesthesia 2002 Nov;14(7):481‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3219898
- Bach‐Styles T, Martin‐Sheridan D, Hughes C, Kaufman S. Comparison of ondansetron, metocloprarnide, and placebo in the prevention of postoperative emesis in children undergoing ophthalmic surgery. Clinical Forum for Nurse Anesthetists 1997;8(4):152‐6. [; EMBASE 1998010555] [PubMed] [Google Scholar]; 3219900
- Bacic A, Rumboldt Z, Gluncic I, Buklijas J. The impact of the menstrual cycle and ondansetron on postoperative nausea and vomiting. International Journal of Clinical Pharmacology Research 1998;18(4):153‐8. [; AN: 1999057684] [PubMed] [Google Scholar]; 3219902
- Badaoui R, Yagoubi A, Pouilly A, Carpenter F, Riboulot M, Ossart M. Comparison between ondansetron and droperidol in preventing postoperative nausea and vomiting after laparoscopic cholecystectomy [Comparaison de l'ondansetron et du droperidol pour la prevention des nausees et des vomissements apres cholecystectomie par voie laparoscopique]. Cahiers D'Anesthesiologie 1998;46(3):171‐4. [; EMBASE 1998236829] [Google Scholar]; 3219904
- Badaoui R, Pouilly A, Yagoubi A, Carpentier F, Riboulot M, Ossart M. Comparative study between ondansetron and droperidol in preventing postoperative nausea and vomiting after general surgery. British Journal of Anaesthesia 1999;82(1):A400. [; ISI:000081159400040 ER] [Google Scholar]; 3219906
- Barros‐de‐Oliveira CM, Iwata NM, Issy AM, Sakata RK, Amaral JLG. Evaluation of droperidol for relief of epidural morphine side effects [Avaliacao do droperidol no alivio dos efeitos colaterais provocados pela morfina por via peridural]. Revista Brasileira de Medicina 2002;59(1‐2):95‐8. [; AN: 2002131734] [Google Scholar]; 3219908
- Barrow PM, Hughes DG, Redfern N, Urie J. Influence of droperidol on nausea and vomiting during patient‐ controlled analgesia. British Journal of Anaesthesia 1994;72(4):460‐1. [; MEDLINE: ; EMBASE 1994127837] [DOI] [PubMed] [Google Scholar]; 3219910
- Barst SM, Leiderman JU, Markowitz A, Rosen AM, Abramson AL, Bienkowski RS. Ondansetron with propofol reduces the incidence of emesis in children following tonsillectomy. Canadian Journal of Anaesthesia 1999 Apr;46(4):359‐62. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3219912
- Batra YK, Arya VK, Mahajan R, Chari P. Dose response study of caudal neostigmine for postoperative analgesia in paediatric patients undergoing genitourinary surgery. Paediatric Anaesthesia 2003 Jul;13(6):515‐21. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3219914
- Baxendale BR, Vater M, Lavery KM. Dexamethasone reduces pain and swelling following extraction of third molar teeth. Anaesthesia 1993 Nov;48(11):961‐4. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3219916
- Beattie WS, Lindblad T, Buckley DN, Forrest JB. Menstruation increases the risk of nausea and vomiting after laparoscopy. A prospective randomized study. Anesthesiology 1993 Feb;78(2):272‐6. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3219918
- Benhamou D, Narchi P, Hamza J, Marx M, Peyrol MT, Sembeil F. Addition of oral clonidine to postoperative patient‐controlled analgesia with iv morphine. British Journal of Anaesthesia 1994 May;72(5):537‐40. [; MEDLINE: ; ISI:A1994NM26400008 ER] [DOI] [PubMed] [Google Scholar]; 3219920
- Bharti N, Shende D. Comparison of anti‐emetic effects of ondansetron and low‐dose droperidol in pediatric strabismus surgery. Journal of Pediatric Ophthalmology & Strabismus 2003 Jan‐Feb;40(1):23‐6. [; MEDLINE: ; CN‐00413222] [DOI] [PubMed] [Google Scholar]; 3219922
- Biedler A, Wilhelm W, Silomon M, Awwad R, Larsen R. [Ondansetron. Prophylaxis and therapy of nausea and vomiting following major gynecologic procedures. Results of a national multicenter study]. [German]. Anaesthesist 1998 Aug;47(8):638‐43. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3219924
- Bilgin H, Ozcan B, Bilgin T, Tokat O, Kimya Y, Kutlay O. Comparison of postoperatif antiemetic efficacy of ondansetron and tropisetron [Ondansetron ve tropisetronun postoperatif antiemetik etkilerinin karsilastirilmasi]. Turk Anesteziyoloji ve Reanimasyon 1998;26(3):134‐8. [; AN: 1998193887] [Google Scholar]; 3219926
- Bisgaard T, Klarskov B, Kehlet H, Rosenberg J. Preoperative dexamethasone improves surgical outcome after laparoscopic cholecystectomy: a randomized double‐blind placebo‐controlled trial. Annals of Surgery 2003 Nov;238(5):651‐60. [; MEDLINE: ] [DOI] [PMC free article] [PubMed] [Google Scholar]; 3219928
- Biswas BN, Rudra A. Comparison of granisetron and granisetron plus dexamethasone for the prevention of postoperative nausea and vomiting after laparoscopic cholecystectomy. Acta Anaesthesiologica Scandinavica 2003 Jan;47(1):79‐83. [; MEDLINE: ; CN‐00422346] [DOI] [PubMed] [Google Scholar]; 3219930
- Blanc VF, Ruest P, Milot J, Jacob JL, Tang A. Antiemetic prophylaxis with promethazine or droperidol in paediatric outpatient strabismus surgery. Canadian Journal of Anaesthesia 1991 Jan;38(1):54‐60. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3219932
- Bock M, Kunz P, Schreckenberger R, Graf BM, Martin E, Motsch J. Comparison of caudal and intravenous clonidine in the prevention of agitation after sevoflurane in children. British Journal of Anaesthesia 2002;88(6):790‐6. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3219934
- Boeke AJ, Lange JJ, Druenen B, Langemeijer JJ. Effect of antagonizing residual neuromuscular block by neostigmine and atropine on postoperative vomiting. British Journal of Anaesthesia 1994 Jun;72(6):654‐6. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3219936
- Bone ME, Young JR, McNeil J, Charlton S. Ginger root‐‐a new antiemetic. The effect of ginger root on postoperative nausea and vomiting after major gynaecological surgery. Anaesthesia 1990;45(8):669‐71. [; MEDLINE: ; CN‐00210142] [DOI] [PubMed] [Google Scholar]; 3219938
- Bonhomme V, Brichant JF, Wuilmart M, Dewandre PY, Hans P. Droperidol reduces nausea after caesarean section but alters the neurological status of the breastfed infants. Anesthesiology 2002;96:A1044. [; CN‐00433393] [Google Scholar]; 3219940
- Booij LH, Rachmat S, Bulder ER. Alizapride in prevention of postoperative nausea and vomiting. Netherlands Journal of Surgery 1988 Feb;40(1):6‐9. [; MEDLINE: ] [PubMed] [Google Scholar]; 3219942
- Borgeat A, Tazlari G, Balmer A, Tramer M, Rifat K. Does ondansetron decrease the incidence of postoperative nausea vomiting after strabismus surgery in children. British Journal of Anaesthesia 1995;74(1):99. [; ISI:A1995RA77700324 ER] [Google Scholar]; 3219944
- Bouderka MA, Al Harrar R, Bouaggad A, Harti A. Neostigmine added to bupivacaine in axillary plexus block: Which benefit? [Adjonction de la néostigmine au cours du bloc plexique par voie axillaire: quel bénéfice?]. Annales Francaises d'Anesthesie et de Reanimation 2003;22(6):510‐3. [; MEDLINE: ; 2003343276 20030904 ISSN: 0750‐7658] [DOI] [PubMed] [Google Scholar]; 3219946
- Boulanger M, Dubois A, Lecron L. Domperidone in the prevention of post‐operative nausea and vomiting. Postgraduate Medical Journal 1979;55(Suppl 1):45‐7. [; MEDLINE: ] [PubMed] [Google Scholar]; 3219948
- Bouly A, Nathan N, Feiss P. Preventing postoperative nausea and vomiting with ondansetron [Prevention des nausees et vomissements postoperatoires par l'ondansetron]. Annales Francaises d'Anesthesie et de Reanimation 1992;11(5):496‐501. [; MEDLINE: ; EMBASE 1992349370] [DOI] [PubMed] [Google Scholar]; 3219950
- Bouly A, Nathan N, Feiss P. Prevention of post‐operative nausea and vomiting using oral ondansetron. European Journal of Anaesthesiology 1993;10:71. [; CN‐00258149] [Google Scholar]; 3219952
- Bowhay AR, May HA, Rudnicka AR, Booker PD. A randomized controlled trial of the antiemetic effect of three doses of ondansetron after strabismus surgery in children. Paediatric Anaesthesia 2001;11(2):215‐21. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3219954
- Boyd JD, Manford ML. Premedication in children. A controlled clinical trial of oral triclofos and diazepam. British Journal of Anaesthesia 1973 May;45(5):501‐6. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3219956
- Brady M, Sollo D. Metoclopramide does not decrease the incidence of nausea after outpatient orthopedic surgery with propofol anesthesia. Anesthesiology 1994;81(3A):A20. [; none.] [Google Scholar]; 3219958
- Breivik H, Lind B. Anti‐emetic and propulsive peristaltic properties of metoclopramide. British Journal of Anaesthesia 1971 Apr;43(4):400‐3. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3219960
- Briggs LP, White M. The effects of premedication on anaesthesia with propofol ('Diprivan'). Postgraduate Medical Journal 1985;61(Suppl 3):35‐7. [; MEDLINE: ] [PubMed] [Google Scholar]; 3219962
- Broadman LM, Ceruzzi W, Patane PS, Hannallah RS, Ruttimann U, Friendly D. Metoclopramide reduces the incidence of vomiting following strabismus surgery in children. Anesthesiology 1990 Feb;72(2):245‐8. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3219964
- Brown RE Jr, James DJ, Weaver RG, Wilhoit RD, Bauman LA. Low‐dose droperidol versus standard‐dose droperidol for prevention of postoperative vomiting after pediatric strabismus surgery. Journal of Clinical Anesthesia 1991 Jul‐Aug;3(4):306‐9. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3219966
- Bugedo G, Gonzalez J, Asenjo C, Cuadra JC, Gajardo A, Castillo L, et al. Ondansetron and droperidol in the prevention of postoperative nausea and vomiting. British Journal of Anaesthesia 1999 Nov;83(5):813‐4. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3219968
- Burmeister MA, Standl TG, Wintruff M, Brauer P, Blanc I, Schulte am Esch J. Dolasetron prophylaxis reduces nausea and postanaesthesia recovery time after remifentanil infusion during monitored anaesthesia care for extracorporeal shock wave lithotripsy. British Journal of Anaesthesia 2003 Feb;90(2):194‐8. [; MEDLINE: ; CN‐00412838] [DOI] [PubMed] [Google Scholar]; 3219970
- Busoni P, Crescioli M, Agostino R, Sestini G. Vomiting and common paediatric surgery. Paediatric Anaesthesia 2000;10(6):639‐43. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3219972
- Butrón López F, Cardoso García LE, Gómez Sainos LA, Salmón Villalobos M. Postoperative nausea and vomiting prevention in nose surgery [Prevención de la náusea y vómito postoperatorio en rinoseptoplastía]. Revista mexicana anestesiologia 1996 Mar;19(1):6‐9. [; LILACS Id: 180458] [Google Scholar]; 3219974
- Cade L, King M, Ashley J. Lack of extrapyramidal side‐effects from droperidol in low dose. Anaesthesia & Intensive Care 1993 Aug;21(4):479‐80. [; MEDLINE: ] [PubMed] [Google Scholar]; 3219976
- Calamandrei M, Andreuccetti T, Crescioli M, Messeri A, Sarti A, Sestini G, et al. Effects of ondansetron and metoclopramide on postoperative nausea and vomiting after epidural anaesthesia in an infant [Effets de l'ondansétron et du métoclopramide sur les nausées et vomissements postopératoires après anesthésie péridurale chez l'enfant]. Cahiers d'anesthesiologie 1994;42(1):19‐23. [; MEDLINE: ; EMBASE 1994090573] [PubMed] [Google Scholar]; 3219978
- Campbell LC, Weis FR Jr. Comparison of three techniques on time to awakening, time to orientation and incidence of nausea and vomiting using alfentanil in balanced anesthesia in an outpatient surgical setting. AANA Journal 1990 Jun;58(3):241‐7. [; MEDLINE: ] [PubMed] [Google Scholar]; 3219980
- Campbell WI, Kendrick RW. Postoperative dental pain ‐ a comparative‐study of antiinflammatory and analgesic agents. Ulster Medical journal 1991 Apr;60(1):39‐43. [; ISI:A1991FP16300006 ER] [PMC free article] [PubMed] [Google Scholar]; 3219982
- Campbell C, Miller DD. Failure of ondansetron to control postoperative nausea and vomiting in ambulatory surgical patients. American Journal of Anesthesiology 1995;22(2):81‐6. [; EMBASE 1995116178] [Google Scholar]; 3219984
- Capouet V, Pauw C, Vernet B, Ivens D, Derijcke V, Versichelen L, et al. Single dose i.v. tropisetron in the prevention of postoperative nausea and vomiting after gynaecological surgery. British Journal of Anaesthesia 1996 Jan;76(1):54‐60. [; MEDLINE: ; EMBASE 1996034119] [DOI] [PubMed] [Google Scholar]; 3219986
- Carabine UA, Milligan KR, Mulholland D, Moore J. Extradural clonidine infusions for analgesia after total hip‐ replacement. British Journal of Anaesthesia 1992;68(4):338‐43. [; MEDLINE: ; SI:A1992HL95600003 ER] [DOI] [PubMed] [Google Scholar]; 3219988
- Carnahan D, Dato K, Hartsuff J. The safety and efficacy of granisetron in postoperative vomiting in pediatric patients undergoing tonsillectomy. AANA Journal 1997 Apr;65(2):154‐9. [; MEDLINE: ] [PubMed] [Google Scholar]; 3219990
- Caron E, Bussieres JF, Lebel D, Mathews S, Milot J, Jacob JL, et al. Ondansetron for the prevention and treatment of nausea and vomiting following pediatric strabismus surgery. Canadian Journal of Ophthalmology 2003 Apr;38(3):214‐22. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3219992
- Carr AS, Splinter WM, Bevan J, Reid CR, Stephenson CA, Lerman J. Ondansetron reduces postoperative vomiting in pediatric strabimus surgery. Anesthesiology 1994 Sep;81:A22. [; SI:A1994PJ09100022 ER] [Google Scholar]; 3219994
- Celik J, Reisli R, Tuncer S, Duman A, Okesli S. Prevention of postoperative nausea‐vomiting in children: Comparison of granisetron and droperidol plus metoclopramide [Cocuklarda postoperatif bulanti‐kusmanin onlenmesinde granisetron ile droperidol ve metoklopramid kombinasyonunun karsilastirilmasi]. Turk Anesteziyoloji ve Reanimasyon 2001;29(3):135‐9. [; AN: 2001140539] [Google Scholar]; 3219996
- Chan SC, Lo JR, Wong KC. Vomiting after anaesthesia for termination of pregnancy in Chinese. Singapore Medical Journal 1983 Dec;24(6):360‐2. [; MEDLINE: ] [PubMed] [Google Scholar]; 3219998
- Chan MT, Chui PT, Ho WS, King WW. Single‐dose tropisetron for preventing postoperative nausea and vomiting after breast surgery. Anesthesia & Analgesia 1998 Oct;87(4):931‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220000
- Chapa Alvarez JR, Navarro Muñíz C, Godoy Castro J, Barragán de la Torre R. Prohylaxis of postoperative nausea and vomiting with ranitidine [sic] [Profilaxis de náusea y vómito posoperatorio con ranitidina]. Investigacion médica internacional 1987 May;14(1):3‐8. [; LILACS Id: 46822] [Google Scholar]; 3220002
- Charuluxananan S, Kyokong O, Somboonviboon W, Narasethakamol A, Promlok P. Nalbuphine versus ondansetron for prevention of intrathecal morphine‐induced pruritus after cesarean delivery. Anesthesia & Analgesia 2003 Jun;96(6):1789‐93. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220004
- Chelly J, Melson T, Pollock J, Hantler C. Oral RS‐25259 prevents postoperative nausea and vomiting following laparoscopic surgery. Anesthesiology 1996 Sep;85:A21. [; ISI:A1996VM46600021 ER] [Google Scholar]; 3220006
- Chen PP, Chui PT, Gin T. Comparison of ondansetron and metoclopramide for the prevention of post‐operative nausea and vomiting after major gynaecological surgery. European Journal of Anaesthesiology 1996 Sep;13(5):485‐91. [; MEDLINE: ; EMBASE 1996299306] [DOI] [PubMed] [Google Scholar]; 3220008
- Chen JJ, Frame DG, Jeffery White T. Efficacy of ondansetron and prochlorperazine for the prevention of postoperative nausea and vomiting after total hip replacement or total knee replacement procedures: A randomized, double‐blind, comparative trial. Archives of Internal Medicine 1998 Oct;158(19):2124‐8. [; MEDLINE: ; AN: 1998356022] [DOI] [PubMed] [Google Scholar]; 3220010
- Chen X, Tang J, White PF, Wender RH, Quon R, Sloninsky A, et al. The effect of timing of dolasetron administration on its efficacy as a prophylactic antiemetic in the ambulatory setting. Anesthesia & Analgesia 2001 Oct;93(4):906‐11. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220012
- Cherian VT, Smith I. Prophylactic ondansetron does not improve patient satisfaction in women using PCA after Caesarean section. British Journal of Anaesthesia 2001 Sep;87(3):502‐4. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220014
- Chestnutt WN, Dundee JW. The influence of cyclizine and perphenazine on the emetic effect of meptazinol. European Journal of Anaesthesiology 1986 Jan;3(1):27‐32. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220016
- Chhibber AK, Lustik SJ, Thakur R, Francisco DR, Fickling KB. Effects of anticholinergics on postoperative vomiting, recovery, and hospital stay in children undergoing tonsillectomy with or without adenoidectomy. Anesthesiology 1999 Mar;90(3):697‐700. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220018
- Chisakuta AM, Mirakhur RK. Anticholinergic prophylaxis does not prevent emesis following strabismus surgery in children. Paediatric Anaesthesia 1995;5(2):97‐100. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220020
- Cholwill JM, Wright W, Hobbs GJ, Curran J. Comparison of ondansetron and cyclizine for prevention of nausea and vomiting after day‐case gynaecological laparoscopy. British Journal of Anaesthesia 1999 Oct;83(4):611‐4. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220022
- Christensen S, Farrow‐Gillespie A, Lerman J. Incidence of emesis and postanesthetic recovery after strabismus surgery in children: a comparison of droperidol and lidocaine. Anesthesiology 1989 Feb;70(2):251‐4. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220024
- Chung CJ, Kim JS, Park HS, Chin YJ. The efficacy of intrathecal neostigmine, intrathecal morphine, and their combination for post‐cesarean section analgesia. Anesthesia and Analgesia 1998;87(2):341‐6. [; MEDLINE: ; AN: 1998268961] [DOI] [PubMed] [Google Scholar]; 3220026
- Cieslak GD, Watcha MF, Phillips MB, Pennant JH. The dose‐response relation and cost‐effectiveness of granisetron for the prophylaxis of pediatric postoperative emesis. Anesthesiology 1996 Nov;85(5):1076‐85. [; MEDLINE: ; EMBASE 1996344323] [DOI] [PubMed] [Google Scholar]; 3220028
- Clyburn P, Kay NH, McKenzie PJ. Effects of diazepam and midazolam on recovery from anaesthesia in outpatients. British Journal of Anaesthesia 1986 Aug;58(8):872‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220030
- Coloma M, Duffy LL, White PF, Kendall Tongier W, Huber PJ Jr. Dexamethasone facilitates discharge after outpatient anorectal surgery. Anesthesia & Analgesia 2001 Jan;92(1):85‐8. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220032
- Coloma M, White PF, Markowitz SD, Whitten CW, Macaluso AR, Berrisford SB, et al. Dexamethasone in combination with dolasetron for prophylaxis in the ambulatory setting: Effect on outcome after laparoscopic cholecystectomy. Anesthesiology 2002;96(6):1346‐50. [; MEDLINE: ; AN: 1997034157] [DOI] [PubMed] [Google Scholar]; 3220034
- Conroy JM, Mahaffey JE, Wilson ME, Haynes GR, Dorman BH, Bailey MK. Prevention of emesis following strabismus surgery. Anesthesiology 1993;79(3A):A10. [; ?SI:A1993LY10800010 ER] [Google Scholar]; 3220036
- Cooke RD, Comijn DJ, Ball RW. Prevention of postoperative nausea and vomiting by domperidone: A double‐blind randomized study using domperidone, metoclopramide and a placebo. South African Medical Journal 1979;56(21):827‐9. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220038
- Cook‐Sather SD, Harris KA, Schreiner MS. Cisapride does not prevent postoperative vomiting in children. Anaesthesia & Analgesia 2002;94(1):50‐4. [; MEDLINE: ; ISI:000173082800009 ER] [DOI] [PubMed] [Google Scholar]; 3220040
- Cosar A, Dagli G, Kurt E, Acar HV, Suer AH. Do tropisetron prevent the postoperative nausea and vomiting? [Tropisetron postoperatif bulanti, kusmayi onler mi?]. Turk Anesteziyoloji ve Reanimasyon 1997;25(7):309‐12. [; AN: 1997305227] [Google Scholar]; 3220042
- Cote CJ, Cohen IT, Suresh S, Rabb M, Rose JB, Weldon BC, et al. A comparison of three doses of a commercially prepared oral midazolam syrup in children. Anesthesia & Analgesia 2002 Jan;94(1):37‐43. [; MEDLINE: ; CN‐00376708] [DOI] [PubMed] [Google Scholar]; 3220044
- Cozanitis D, Asantila R, Eklund P, Paloheimo M. A comparison of ranitidine, droperidol or placebo in the prevention of nausea and vomiting after hysterectomy. Canadian Journal of Anaesthesia 1996 Feb;43(2):106‐9. [; MEDLINE: ; EMBASE 1996103677] [DOI] [PubMed] [Google Scholar]; 3220046
- Cramb R, Fargas‐Babjak A, Hirano G. Intraoperative prochlorperazine for prevention of post‐operative nausea and vomiting. Canadian Journal of Anaesthesia 1989 Sep;36(5):565‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220048
- Culebras X, Corpataux JB, Gaggero G, Tramer MR. The antiemetic efficacy of droperidol added to morphine patient‐controlled analgesia: a randomized, controlled, multicenter dose‐finding study. Anesthesia & Analgesia 2003 Sep;97(3):816‐21. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220050
- D'Angelo R, Goldstein MJ, Gerancher JC, Owen MD. A comparison of bolus droperidol, metoclopramide; ondansetron and propofol on post operative nausea and vomiting following outpatient laparoscopic surgery. Anesthesiology 1997;87:A50. [] [Google Scholar]; 3220052
- Dabbous A, Itani M, Kawas N, Karam V, Aouad M, Baraka A, et al. Post‐laparoscopic vomiting in females versus males: comparison of prophylactic antiemetic action of ondansetron versus metoclopramide. Journal of the Society of Laparoendoscopic Surgeons 1998 Jul‐Sep;2(3):273‐6. [; MEDLINE: ] [PMC free article] [PubMed] [Google Scholar]; 3220054
- Daftary S, Jagtap SR, Saksena S. Intravenous Ondansetron in prevention of PONV following tonsillectomy under ether anaesthesia. Journal of Anaesthesiology Clinical Pharmacology 1998;14(1):51‐4. [; EMBASE 1998058517] [Google Scholar]; 3220056
- Danner K, Becker HG, Best B, Madler C. [Prophylaxis of nausea and vomiting after thyroid surgery: comparison of oral and intravenous dolasetron with intravenous droperidol and placebo]. [German]. Anasthesiologie Intensivmedizin Notfallmedizin Schmerztherapie 2001 Jul;36(7):425‐30. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220058
- Davies DR, Doughty AG. Premedication in children. A trial of intramuscular droperidol, droperidol‐phenoperidine, papaveretum‐hyoscine and normal saline. British Journal of Anaesthesia 1971 Jan;43(1):65‐75. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220060
- Davies AO. Oral diazepam premedication reduces the incidence of post‐succinylcholine muscle pains. Canadian Anaesthetists Society Journal 1983 Nov;30(6):603‐6. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220062
- Davies PRF, Warwick P, O CM. Antiemetic efficacy of ondansetron with patient‐controlled analgesia. Anaesthesia 1996;51(9):880‐2. [; MEDLINE: ; EMBASE 1996283679] [DOI] [PubMed] [Google Scholar]; 3220064
- Davis PJ, McGowan Jr FX, Landsman I, Maloney K, Hoffmann P. Effect of antiemetic therapy on recovery and hospital discharge time: A double‐blind assessment of ondansetron, droperidol, and placebo in pediatric patients undergoing ambulatory surgery. Anesthesiology 1995;83(5):956‐60. [; MEDLINE: ; AN: 1995339732] [DOI] [PubMed] [Google Scholar]; 3220066
- Davis A, Krige S, Moyes D. A double‐blind randomized prospective study comparing ondansetron with droperidol in the prevention of emesis following strabismus surgery. Anaesthesia & Intensive Care 1995 Aug;23(4):438‐43. [; MEDLINE: ; EMBASE 1995243555] [DOI] [PubMed] [Google Scholar]; 3220068
- Dershwitz M, Conant JA, Chang Y, Rosow CE, Connors PM. A randomized, double‐blind, dose‐response study of ondansetron in the prevention of postoperative nausea and vomiting. Journal of Clinical Anesthesia 1998;10(4):314‐20. [; MEDLINE: ; AN: 1998212659] [DOI] [PubMed] [Google Scholar]; 3220070
- Diamond MJ, Keeri‐Szanto M. Reduction of postoperative vomiting by preoperative administration of oral metoclopramide. Canadian Anaesthetists Society Journal 1980 Jan;27(1):36‐9. [; MEDLINE: ; EMBASE 1980111044] [DOI] [PubMed] [Google Scholar]; 3220072
- Diamond MJ, Bailey D, McPhee A. The Gender‐dependent pharmacodynamic difference in the antiemetic action of metoclopramide is a dose‐related phenomenon. Canadian Journal of Anaesthesia 1988;35(3 II):S65. [; EMBASE 1988130834] [Google Scholar]; 3220074
- Diemunsch P, D'Hollander A, Paxton L, Schoeffler P, Wessel P, Nave S, et al. Intravenous dolasetron mesilate in the prevention of postoperative nausea and vomiting in females undergoing gynecological surgery. Journal of Clinical Anesthesia 1997;9(5):365‐73. [; MEDLINE: ; AN: 1997227292] [DOI] [PubMed] [Google Scholar]; 3220076
- Diemunsch P, Korttila K, Leeser J, Helmers JH, Wilkey B, Nave S, et al. Oral dolasetron mesylate for prevention of postoperative nausea and vomiting: a multicenter, double‐blind, placebo‐controlled study. The Oral Dolasetron PONV Prevention Study Group. Journal of Clinical Anesthesia 1998 Mar;10(2):145‐52. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220078
- Dillier CM, Weiss M, Gerber AC. [Tropisetron for prevention of nausea and vomiting in children undergoing tonsillectomy and/or adenoidectomy]. [German] [Tropisetron zur prophylaxe von postoperativem erbrechen bei kindern. Wirksamkeit und vertraglichkeit nach tonsillektomien und adenotonsillektomien]. Anaesthesist 2000 Apr;49(4):275‐8. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220080
- Ding YF, Fredman B, White PF. Use of mivacurium during laparoscopic surgery ‐ effect of reversal drugs on postoperative recovery. Anesthesia & Analgesia 1994 Mar;78(3):450‐4. [; MEDLINE: ; ISI:A1994NA40300005 ER] [DOI] [PubMed] [Google Scholar]; 3220082
- Dobkin AB, Evers W, Israel JS. Double‐blind evaluation of metoclopramide (MK 745, Sinemet), trimethobenzamide (Tigan), and a placebo as postanaesthetic anti‐emetics following methoxyflurane anaesthesia. Canadian Anaesthetists Society Journal 1968 Jan;15(1):80‐91. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220084
- Dodson ME, Eastley RJ. Comparative study of two long‐acting tranquillizers for oral premedication. British Journal of Anaesthesia 1978 Oct;50(10):1059‐64. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220086
- Doe EA, Jones P, O'Hara MA. A comparison of prophylactic ondansetron hydrochloride and droperidol for strabismus repair in adults. Journal of Pediatric Ophthalmology and Strabismus 1998;35(5):264‐9. [; MEDLINE: ; AN: 1998320870] [DOI] [PubMed] [Google Scholar]; 3220088
- Doenicke A, Roizen MF, Hoernecke R, Stodulka P, O'Connor MF. Premedication with h‐1 and h‐2 blocking reduces the incidence of postoperative nausea and vomiting. Anesthesia & Analgesia 1994 Feb;78(2):u61. [; ISI:A1994NA40600095 ER] [Google Scholar]; 3220090
- Doyle E, Byers G, McNicol LR, Morton NS. Prevention of postoperative nausea and vomiting with transdermal hyoscine in children using patient‐controlled analgesia. British Journal of Anaesthesia 1994;72(1):72‐6. [; MEDLINE: ; AN: 1994031870] [DOI] [PubMed] [Google Scholar]; 3220092
- Dresner M, Dean S, Lumb A, Bellamy M. High‐dose ondansetron regimen vs droperidol for morphine patient‐controlled analgesia. British Journal of Anaesthesia 1998 Sep;81(3):384‐6. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220094
- Duarte DF, Linhares S, Gesser N, Pederneiras SG. Effect of intramuscular clebopride on postoperative nausea and vomiting. Clinical Therapeutics 1985;7(3):365‐71. [; MEDLINE: ; SI:A1985AHW7100012 ER] [PubMed] [Google Scholar]; 3220096
- Dundee JW, Assaf RAE, Loan WB, Morrison JD. A comparison of the efficacy of cyclizine and perphenazine in reducing the emetic effects of morphine and pethidine. British Journal of Clinical Pharmacology 1975;2(1):81‐5. [; MEDLINE: ; CN‐00305545] [DOI] [PMC free article] [PubMed] [Google Scholar]; 3220098
- Dupeyron JP, Conseiller C, Levarlet M, Hemmingsen C, Schoeffler P, Pedersen FM, et al. The effect of oral ondansetron in the prevention of postoperative nausea and vomiting after major gynaecological surgery performed under general anaesthesia. Anaesthesia 1993 Mar;48(3):214‐8. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220100
- Eberhart LHJ, Holzrichter P, Roscher R. Transdermal hyoscine (scopolamine) in the prevention of postoperative nausea and vomiting. No therapeutic improvement in spite of reduced postoperative vomiting in general surgical and gynaecological patients [Transdermales scopolamin zur prophylaxe von ubelkeit und erbrechen in der postoperativen phase. kein klinisch relevanter fortschritt trotz verringerung postoperativen erbrechens bei allgemeinchirurgischen und gynakologischen patienten]. Anaesthesist 1996;45(3):259‐67. [; EMBASE 1996101306] [DOI] [PubMed] [Google Scholar]; 3220102
- Eberhart LH, Seeling W, Bopp TI, Morin AM, Georgieff M. Dimenhydrinate for prevention of post‐operative nausea and vomiting in female in‐patients. European Journal of Anaesthesiology 1999 May;16(5):284‐9. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220104
- Eberhart LH, Seeling W, Hartschuh T, Morin AM, Georgieff M. Droperidol and dimenhydrinate alone or in combination for the prevention of post‐operative nausea and vomiting after nasal surgery in male patients. European Journal of Anaesthesiology 1999 Nov;16(11):790‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220106
- Eberhart LH, Seeling W, Ulrich B, Morin AM, Georgieff M. [Dimenhydrinate and metoclopramide for prevention of nausea and vomiting following septorhinoplasties in women]. [German]. Anasthesiologie Intensivmedizin Notfallmedizin Schmerztherapie 1999 Aug;34(8):480‐4. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220108
- Eberhart LH, Lindenthal M, Seeling W, Gackle H, Georgieff M. [Dolasetron, droperidol and a combination of both in prevention of postoperative nausea and vomiting after extracapsular cataract extraction under general anesthesia]. [German]. Anasthesiologie Intensivmedizin Notfallmedizin Schmerztherapie 1999 Jun;34(6):345‐9. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220110
- Eberhart LH, Seeling W. Droperidol‐supplemented anaesthesia decreases post‐operative nausea and vomiting but impairs post‐operative mood and well‐being. European Journal of Anaesthesiology 1999 May;16(5):290‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220112
- Eberhart LH, Seeling W, Ulrich B, Morin AM, Georgieff M. Dimenhydrinate and metoclopramide alone or in combination for prophylaxis of PONV. Canadian Journal of Anaesthesia 2000 Aug;47(8):780‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220114
- Eberhart LH, Novatchkov N, Schricker T, Georgieff M, Baur CP. Clonidine compared to midazolam for intravenous premedication for ambulatory procedures. A controlled double blind study in ASA 1 patients. Anasthesiologie Intensivmedizin Notfallmedizin Schmerztherapie 2000;35(6):388‐93. [; CN‐00342868] [DOI] [PubMed] [Google Scholar]; 3220116
- Eberhart LH, Seeling W, Morin AM, Vogt N, Georgieff M. [Droperidol and dimenhydrinate alone or in combination for prevention of postoperative nausea and vomiting]. [German]. Anasthesiologie Intensivmedizin Notfallmedizin Schmerztherapie 2001 May;36(5):290‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220118
- Eberhart LH, Mayer R, Betz O, Tsolakidis S, Hilpert W, Morin AM, et al. Ginger does not prevent postoperative nausea and vomiting after laparoscopic surgery. Anesthesia & Analgesia 2003 Apr;96(4):995‐8. [; MEDLINE: ; CN‐00423085] [DOI] [PubMed] [Google Scholar]; 3220120
- Elhakim M, Ghalaab M, Soliman M. Effects of odansetron and balanced analgesia on postoperative nausea and vomiting in laparoscopic surgery. Acta Anaesthesiologica Italica 1995;46(Suppl 1):23‐8. [; EMBASE 1996027690] [Google Scholar]; 3220122
- Elhakim M, Nafie M, Mahmoud K, Atef A. Dexamethasone 8 mg in combination with ondansetron 4 mg appears to be the optimal dose for the prevention of nausea and vomiting after laparoscopic cholecystectomy. Canadian Journal of Anaesthesia 2002 Nov;49(9):922‐6. [; MEDLINE: ; CN‐00411173] [DOI] [PubMed] [Google Scholar]; 3220124
- Elhakim M, Ali NM, Rashed I, Riad MK, Refat M. Dexamethasone reduces postoperative vomiting and pain after pediatric tonsillectomy. Canadian Journal of Anaesthesia 2003 Apr;50(4):392‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220126
- Elliott RH, Graham SG, Curran JP. Sustained release metoclopramide for prophylaxis of post‐operative nausea and vomiting. European Journal of Anaesthesiology 1994 Nov;11(6):465‐7. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220128
- Ellis FR, Spence AA. Clinical trials of metoclopramide (Maxolon) as an antiemetic in anaesthesia. Anaesthesia 1970 Jul;25(3):368‐71. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220130
- Shobaki AM, Bondok RS, Yakoub AM. Efficacy of intravenous granisetron versus placebo in the prophylaxis of postoperative nausea and vomiting after infratentorial craniotomy: A double‐blind randomised study. Egyptian Journal of Anaesthesia 2003;19(3):297‐304. [; ISSN: 1110‐1849] [Google Scholar]; 3220132
- Ercelen O, Celiker V, Celebioglu B, Basgul E, Aypar U. Prevention from postoperative nausea and vomiting after strabismus surgery in children. Acta Anaesthesiologica Italica 1996;47(3):211‐4. [; AN: 1997107863] [Google Scholar]; 3220134
- Eriksson H, Korttila K. Recovery profile after desflurane with or without ondansetron compared with propofol in patients undergoing outpatient gynecological laparoscopy. Anesthesia & Analgesia 1996 Mar;82(3):533‐8. [; MEDLINE: ; EMBASE 1996073467] [DOI] [PubMed] [Google Scholar]; 3220136
- Eustis S, Lerman J, Smith DR. Effect of droperidol pretreatment on post‐anesthetic vomiting in children undergoing strabismus surgery: the minimum effective dose. Journal of Pediatric Ophthalmology and Strabismus 1987 Jul‐Aug;24(4):165‐9. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220138
- Fabling JM, Gan TJ, El‐Moalem HE, Warner DS, Borel CO. A randomized, double‐blinded comparison of ondansetron, droperidol, and placebo for prevention of postoperative nausea and vomiting after supratentorial craniotomy. Anesthesia & Analgesia 2000 Aug;91(2):358‐61. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220140
- Fabling JM, Gan TJ, Moalem HE, Warner DS, Borel CO. A randomized, double‐blind comparison of ondansetron versus placebo for prevention of nausea and vomiting after infratentorial craniotomy. Journal of Neurosurgical Anesthesiology 2002;14(2):102‐7. [; MEDLINE: ; AN: 2002122320] [DOI] [PubMed] [Google Scholar]; 3220142
- Fassolt A. The prophylaxis of nausea and vomiting after inhalation‐anaesthesia with droperidol (dehydrobenzperidol) [Droperidol (Dehydrobenzperidol) zur Prophylaxe von Nausea und Vomitus nach Inhalationsnarkosen]. Praktische Anasthesie Wiederbelebung und Intensivtherapie 1977;12:292‐7. [; CN‐00280257] [Google Scholar]; 3220144
- Fazi L, Jantzen EC, Rose JB, Kurth CD, Watcha MF. A comparison of oral clonidine and oral midazolam as preanesthetic medications in the pediatric tonsillectomy patient. Anesthesia & Analgesia 2001 Jan;92(1):56‐61. [; MEDLINE: ; ISI:000166044300011 ER] [DOI] [PubMed] [Google Scholar]; 3220146
- Fillinger MP, Rassias AJ, Guyre PM, Sanders JH, Beach M, Pahl J, et al. Glucocorticoid effects on the inflammatory and clinical responses to cardiac surgery. Journal of Cardiothoracic and Vascular Anesthesia 2002;16(2):163‐9. [; MEDLINE: ; AN: 2002169400] [DOI] [PubMed] [Google Scholar]; 3220148
- Flores Rivera I, Bosques Nieves G, Goiz Arenas CM. Comparative effects of ondansetron and metoclopramide in the prevention of postoperative nausea and vomiting in the pediatric ambulatory surgery [Estudio comparative de ondansetron y metoclopramida en la prevencion de nausea y vomito postoperatorio en cirugia ambulatoria pediatrica bajo anestesia general]. Revista Mexicana de Anestesiologia 1997 Jul;20(3):132‐5. [; EMBASE 1997330914; LILACS Id 225081] [Google Scholar]; 3220150
- Fogarty DJ, Carabine UA, Milligan KR. Comparison of the analgesic effects of intrathecal clonidine and intrathecal morphine after spinal anaesthesia in patients undergoing total hip replacement. British Journal of Anaesthesia 1993 Nov;71(5):661‐4. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220152
- Fonseca NM, Cardoso RCG. Nausea and vomiting prevention with metoclopramide in patients submitted to uterine curettage under general anesthesia [Prevençäo de náuseas e vômitos com metoclopramida em pacientes submetidos a curetagem uterina sob anestesia geral]. Revista Brasileira de Anestesiologia 2001 Mar;51(2):105‐11. [; LILACS Id: 282591] [Google Scholar]; 3220154
- Fortney JT, Gan TJ, Graczyk S, Wetchler B, Melson T, Khalil S, et al. A comparison of the efficacy, safety, and patient satisfaction of ondansetron versus droperidol as antiemetics for elective outpatient surgical procedures. S3A‐409 and S3A‐410 Study Groups. Anesthesia & Analgesia 1998;86(4):731‐8. [; MEDLINE: ; CN‐00296086] [DOI] [PubMed] [Google Scholar]; 3220156
- Foss JF, Moss J, O'Connor M, Roizen MF. Methylnaltrexone reduces morphine‐induced postoperative emesis by 30‐percent. Anesthesia & Analgesia 1994 Feb;78(2):73. [; ISI:A1994NA40600117 ER] 8267184 [Google Scholar]; 3220158
- Foster PN, Stickle BR, Laurence AS. Akathisia following low‐dose droperidol for antiemesis in day‐case patients. Anaesthesia 1996 May;51(5):491‐4. [; MEDLINE: ; EMBASE 1996149359] [DOI] [PubMed] [Google Scholar]; 3220160
- Fournier R, Gessel E, Weber A, Gamulin Z. Epinephrine and clonidine do not improve intrathecal sufentanil analgesia after total hip replacement. British Journal of Anaesthesia 2002 Oct;89(4):562‐6. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220162
- Fozard JR, Manford ML. A controlled clinical trial of oral droperidol and droperidol plus diazepam for premedication in children. British Journal of Anaesthesia 1977 Nov;49(11):1147‐51. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220164
- Frank T, Thieme V, Radow L. [Premedication in maxillofacial surgery under total intravenous anesthesia . Effects of clonidine compared to midazolam on the perioperative course]. [German]. Anasthesiologie Intensivmedizin Notfallmedizin Schmerztherapie 2000 Jul;35(7):428‐34. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220166
- Freedman GM, Kreitzer JM, Reuben SS, Eisenkraft JB. Improving patient‐controlled analgesia ‐ adding droperidol to morphine‐sulfate to reduce nausea and vomiting and potentiate analgesia. Mount Sinai Journal of Medicine 1995 May;62(3):221‐5. [; MEDLINE: ; SI:A1995RB84900011 ER] [PubMed] [Google Scholar]; 3220168
- Friesen RH, Lockhart CH. Oral transmucosal fentanyl citrate for preanesthetic medication of pediatric day surgery patients with and without droperidol as a prophylactic anti‐emetic. Anesthesiology 1992 Jan;76(1):46‐51. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220170
- Fujii Y, Tanaka H, Toyooka H. Reduction of postoperative nausea and vomiting with granisetron. Canadian Journal of Anaesthesia 1994 Apr;41(4):291‐4. [; MEDLINE: ; EMBASE 1994123086] [DOI] [PubMed] [Google Scholar]; 3220172
- Fujii Y, Tanaka H, Toyooka H. Optimal anti‐emetic dose of granisetron for preventing postoperative nausea and vomiting. Canadian Journal of Anaesthesia 1994 Sep;41(9):794‐7. [; MEDLINE: ; EMBASE 1994290979] [DOI] [PubMed] [Google Scholar]; 3220174
- Fujii Y, Tanaka H, Toyooka H. Granisetron‐dexamethasone combination reduces postoperative nausea and vomiting. Canadian Journal of Anaesthesia 1995 May;42(5):387‐90. [; MEDLINE: ; EMBASE 1995178643] [DOI] [PubMed] [Google Scholar]; 3220176
- Fujii Y, Tanaka H, Toyooka H. Prevention of postoperative nausea and vomiting with granisetron: a randomized, double‐blind comparison with droperidol. Canadian Journal of Anaesthesia 1995 Oct;42(10):852‐6. [; MEDLINE: ; EMBASE 1995321145] [DOI] [PubMed] [Google Scholar]; 3220178
- Fujii Y, Tanaka H, Toyooka H. Granisetron and dexamethasone provide more improved prevention of postoperative emesis than granisetron alone in children. Canadian Journal of Anaesthesia 1996 Dec;43(12):1229‐32. [; MEDLINE: ; EMBASE 1997060587] [DOI] [PubMed] [Google Scholar]; 3220180
- Fujii Y, Tanaka H, Toyooka H. Granisetron reduces vomiting after strabismus surgery and tonsillectomy in children. Canadian Journal of Anaesthesia 1996 Jan;43(1):35‐8. [; MEDLINE: ; EMBASE 1996065112] [DOI] [PubMed] [Google Scholar]; 3220182
- Fujii Y, Toyooka H, Tanaka H. Antiemetic efficacy of granisetron and metoclopramide in children undergoing ophthalmic or ENT surgery. Canadian Journal of Anaesthesia 1996 Nov;43(11):1095‐9. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220184
- Fujii Y, Toyooka H, Tanaka H. Antiemetic effects of granisetron on postoperative nausea and vomiting in patients with and without motion sickness. Canadian Journal of Anaesthesia 1996 Feb;43(2):110‐4. [; MEDLINE: ; EMBASE 1996103678] [DOI] [PubMed] [Google Scholar]; 3220186
- Fujii Y, Toyooka H, Tanaka H. Effective dose of granisetron for preventing postoperative emesis in children. Canadian Journal of Anaesthesia 1996 Jul;43(7):660‐4. [; MEDLINE: ; EMBASE 1996269133] [DOI] [PubMed] [Google Scholar]; 3220188
- Fujii Y, Tanaka H, Toyooka H. The effects of dexamethasone on antiemetics in female patients undergoing gynecologic surgery. Anesthesia & Analgesia 1997 Oct;85(4):913‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220190
- Fujii Y, Tanaka H, Toyooka H. Granisetron reduces the incidence and severity of nausea and vomiting after laparoscopic cholecystectomy. Canadian Journal of Anaesthesia 1997 Apr;44(4):396‐400. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220192
- Fujii Y, Toyooka H, Tanaka H. Prevention of PONV granisetron, droperidol and metoclopramide in female patients with history of motion sickness. Canadian Journal of Anaesthesia 1997 Aug;44(8):820‐4. [; MEDLINE: ; EMBASE 1997345411] [DOI] [PubMed] [Google Scholar]; 3220194
- Fujii Y, Tanaka H, Toyooka H. Granisetron reduces postoperative nausea and vomiting throughout menstrual cycle. Canadian Journal of Anaesthesia 1997 May;44(5 Pt 1):489‐93. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220196
- Fujii Y, Tanaka H, Toyooka H. Prophylactic antiemetic efficacy of granisetron in patients with and without previous postoperative emesis. Canadian Journal of Anaesthesia 1997 Mar;44(3):273‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220198
- Fujii Y, Tanaka H, Toyooka H. Effect of propofol for induction and ondansetron with or without dexamethasone for the prevention of nausea and vomiting after major gynecologic surgery. Acta Anaesthesiologica Scandinavica 1997 Oct;41(9):1167‐70. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220200
- Fujii Y, Tanaka H, Toyooka H. Granisetron reduces incidence of nausea and vomiting after breast surgery. Acta Anaesthesiologica Scandinavica 1997 Jun;41(6):746‐9. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220202
- Fujii Y, Toyooka H, Tanaka H. Granisetron reduces the incidence of nausea and vomiting after middle ear surgery. British Journal of Anaesthesia 1997 Oct;79(4):539‐40. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220204
- Fujii Y, Toyooka H, Tanaka H. Prophylactic antiemetic therapy with a combination of granisetron and dexamethasone in patients undergoing middle ear surgery. British Journal of Anaesthesia 1998 Nov;81(5):754‐6. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220206
- Fujii Y, Tanaka H, Toyooka H. Prophylactic antiemetic therapy with granisetron‐dexamethasone combination in women undergoing breast surgery. Acta Anaesthesiologica Scandinavica 1998;42(9):1038‐42. [; MEDLINE: ; CN‐00204291] [DOI] [PubMed] [Google Scholar]; 3220208
- Fujii Y, Saitoh Y, Tanaka H, Toyooka H. Prophylactic oral antiemetics for preventing postoperative nausea and vomiting: granisetron versus domperidone. Anesthesia & Analgesia 1998 Dec;87(6):1404‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220210
- Fujii Y, Saitoh Y, Tanaka H, Toyooka H. Comparison of granisetron and droperidol in the prevention of vomiting after strabismus surgery or tonsillectomy in children. Paediatric Anaesthesia 1998;8(3):241‐4. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220212
- Fujii Y, Saitoh Y, Tanaka H, Toyooka H. Anti‐emetic efficacy of prophylactic granisetron, droperidol and metoclopramide in the prevention of nausea and vomiting after laparoscopic cholecystectomy: a randomized, double‐blind, placebo‐controlled trial. European Journal of Anaesthesiology 1998 Mar;15(2):166‐71. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220214
- Fujii Y, Saitoh Y, Tanaka H, Toyooka H. Prophylactic antiemetic therapy with granisetron‐droperidol combination in patients undergoing laparoscopic cholecystectomy. Canadian Journal of Anaesthesia 1998 Jun;45(6):541‐4. [; MEDLINE: ; EMBASE 1998271578] [DOI] [PubMed] [Google Scholar]; 3220216
- Fujii Y, Saitoh Y, Tanaka H, Toyooka H. Prevention of PONV with granisetron, droperidol or metoclopramide in patients with postoperative emesis. Canadian Journal of Anaesthesia 1998 Feb;45(2):153‐6. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220218
- Fujii Y, Tanaka H. Prophylactic therapy with granisetron in the prevention of vomiting after paediatric surgery. A randomized, double‐blind comparison with droperidol and metoclopramide. Paediatric Anaesthesia 1998;8(2):149‐53. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220220
- Fujii Y, Tanaka H, Toyooka H. Prevention of nausea and vomiting with granisetron, droperidol and metoclopramide during and after spinal anaesthesia for caesarean section: a randomized, double‐blind, placebo‐controlled trial. Acta Anaesthesiologica Scandinavica 1998 Sep;42(8):921‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220222
- Fujii Y, Toyooka H, Tanaka H. Prevention of postoperative nausea and vomiting in female patients during menstruation: comparison of droperidol, metoclopramide and granisetron. British Journal of Anaesthesia 1998 Feb;80(2):248‐9. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220224
- Fujii Y, Toyooka H, Tanaka H. Granisetron‐droperidol combination for the prevention of postoperative nausea and vomiting in female patients undergoing breast surgery. British Journal of Anaesthesia 1998 Sep;81(3):387‐9. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220226
- Fujii Y, Toyooka H, Tanaka H. Prophylactic anti‐emetic therapy with granisetron, droperidol and metoclopramide in female patients undergoing middle ear surgery. Anaesthesia 1998 Dec;53(12):1165‐8. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220228
- Fujii Y, Toyooka H, Tanaka H. Prevention of postoperative nausea and vomiting with a combination of granisetron and droperidol. Anesthesia & Analgesia 1998 Mar;86(3):613‐6. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220230
- Fujii Y, Toyooka H, Tanaka H. A granisetron‐droperidol combination prevents postoperative vomiting in children. Anesthesia & Analgesia 1998 Oct;87(4):761‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220232
- Fujii Y, Saitoh Y, Tanaka H, Toyooka H. Effective dose of granisetron for the prevention of post‐operative nausea and vomiting in patients undergoing laparoscopic cholecystectomy. European Journal of Anesthesiology 1998 May;15(3):287‐91. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220234
- Fujii Y, Tanaka H, Toyooka H. Preoperative oral granisetron prevents postoperative nausea and vomiting. Acta Anaesthesiologica Scandinavica 1998 Jul;42(6):653‐7. [; MEDLINE: ; EMBASE 1998216260] [DOI] [PubMed] [Google Scholar]; 3220236
- Fujii Y, Toyooka H, Tanaka H. Granisetron in the prevention of nausea and vomiting after middle‐ear surgery: a dose‐ranging study. British Journal of Anaesthesia 1998 Jun;80(6):764‐6. [; MEDLINE: ; EMBASE 1998204642] [DOI] [PubMed] [Google Scholar]; 3220238
- Fujii Y, Toyooka H, Tanaka H. Oral granisetron prevents postoperative vomiting in children. British Journal of Anaesthesia 1998 Sep;81(3):390‐2. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220240
- Fujii Y, Saitoh Y, Tanaka H, Toyooka H. Prophylactic antiemetic therapy with granisetron in women undergoing thyroidectomy. British Journal of Anaesthesia 1998;81(4):526‐8. [; MEDLINE: ; CN‐00268706] [DOI] [PubMed] [Google Scholar]; 3220242
- Fujii Y, Tanaka H, Toyooka H. Prevention of nausea and vomiting in female patients undergoing breast surgery: a comparison with granisetron, droperidol, metoclopramide and placebo. Acta Anaesthesiologica Scandinavica 1998 Feb;42(2):220‐4. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220244
- Fujii Y, Saitoh Y, Tanaka H, Toyooka H. Prophylactic therapy with combined granisetron and dexamethasone for the prevention of post‐operative vomiting in children. European Journal of Anaesthesiology 1999 Jun;16(6):376‐9. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220246
- Fujii Y, Saitoh Y, Tanaka H, Toyooka H. Granisetron/dexamethasone combination for reducing nausea and vomiting during and after spinal anesthesia for cesarean section. Anesthesia & Analgesia 1999 Jun;88(6):1346‐50. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220248
- Fujii Y, Tanaka H, Kobayashi N. Granisetron, droperidol, and metoclopramide for preventing postoperative nausea and vomiting after thyroidectomy. Laryngoscope 1999 Apr;109(4):664‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220250
- Fujii Y, Saitoh Y, Tanaka H, Toyooka H. Combination of granisetron and droperidol for the prevention of vomiting after paediatric strabismus surgery. Paediatric Anaesthesia 1999;9(4):329‐33. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220252
- Fujii Y, Saitoh Y, Tanaka H, Toyooka H. Combination of granisetron and droperidol in the prevention of nausea and vomiting after middle ear surgery. Journal of Clinical Anesthesia 1999 Mar;11(2):108‐12. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220254
- Fujii Y, Saitoh Y, Tanaka H, Toyooka H. Prevention of post‐operative nausea and vomiting with combined granisetron and droperidol in women undergoing thyroidectomy. European Journal of Anaesthesiology 1999 Oct;16(10):688‐91. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220256
- Fujii Y, Saitoh Y, Tanaka H, Toyooka H. Preoperative oral antiemetics for reducing postoperative vomiting after tonsillectomy in children: granisetron versus perphenazine. Anesthesia & Analgesia 1999 Jun;88(6):1298‐1301. [; MEDLINE: ; ISI:000080505500018 ER] [DOI] [PubMed] [Google Scholar]; 3220258
- Fujii Y, Tanaka H, Kobayashi N. Prevention of nausea and vomiting after middle ear surgery: granisetron versus ramosetron. Laryngoscope 1999 Dec;109(12):1988‐90. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220260
- Fujii Y, Saitoh Y, Tanaka H, Toyooka H. Ramosetron vs granisetron for the prevention of postoperative nausea and vomiting after laparoscopic cholecystectomy. Canadian Journal of Anaesthesia 1999 Oct;46(10):991‐3. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220262
- Fujii Y, Saitoh Y, Tanaka H, Toyooka H. Comparison of ramosetron and granisetron for preventing postoperative nausea and vomiting after gynecologic surgery. Anesthesia & Analgesia 1999 Aug;89(2):476‐9. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220264
- Fujii Y, Saitoh Y, Tanaka H, Toyooka H. Anti‐emetic efficacy of prophylactic granisetron compared with perphenazine for the prevention of post‐operative vomiting in children. European Journal of Anaesthesiology 1999 May;16(5):304‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220266
- Fujii Y, Tanaka H. Granisetron reduces post‐operative vomiting in children: a dose‐ranging study. European Journal of Anaesthesiology 1999 Jan;16(1):62‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220268
- Fujii Y, Saitoh Y, Tanaka H, Toyooka H. Prevention of postoperative vomiting with granisetron in paediatric patients with and without a history of motion sickness. Paediatric Anaesthesia 1999;9(6):527‐30. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220270
- Fujii Y, Tanaka H, Ito M. Preoperative oral granisetron for the prevention of vomiting after strabismus surgery in children. Ophthalmology 1999 Sep;106(9):1713‐5. [; MEDLINE: ; AN: 2000129987] [DOI] [PubMed] [Google Scholar]; 3220272
- Fujii Y, Saitoh Y, Tanaka H, Toyooka H. Granisetron/dexamethasone combination for the prevention of postoperative nausea and vomiting after laparoscopic cholecystectomy. European Journal of Anaesthesiology 2000 Jan;17(1):64‐8. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220274
- Fujii Y, Tanaka H, Kobayashi N. Granisetron/dexamethasone combination for the prevention of postoperative nausea and vomiting after thyroidectomy. Anaesthesia and Intensive Care 2000 Jun;28(3):266‐9. [; MEDLINE: ; ISI:000087311000004 ER] [PubMed] [Google Scholar]; 3220276
- Fujii Y, Saitoh Y, Tanaka H, Toyooka H. Ramosetron for preventing postoperative nausea and vomiting in women undergoing gynecological surgery. Anesthesia & Analgesia 2000 Feb;90(2):472‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220278
- Fujii Y, Tanaka H, Kobayashi N. Prevention of postoperative nausea and vomiting with antiemetics in patients undergoing middle ear surgery: comparison of a small dose of propofol with droperidol or metoclopramide. Archives of Otolaryngology Head & Neck Surgery 2001 Jan;127(1):25‐8. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220280
- Fujii Y, Tanaka H, Kobayashi N. Small doses of propofol, droperidol, and metoclopramide for the prevention of postoperative nausea and vomiting after thyroidectomy. Otolaryngology ‐ Head & Neck Surgery 2001 Mar;124(3):266‐9. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220282
- Fujii Y, Tanaka H. Comparison of granisetron, droperidol, and metoclopramide for prevention of postoperative vomiting in children with a history of motion sickness undergoing tonsillectomy. Journal of Pediatric Surgery 2001;36(3):460‐2. [; MEDLINE: ; AN: 2001089760] [DOI] [PubMed] [Google Scholar]; 3220284
- Fujii Y, Saitoh Y, Kobayashi N. Prevention of vomiting after tonsillectomy in children: granisetron versus ramosetron. Laryngoscope 2001 Feb;111(2):255‐8. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220286
- Fujii Y, Tanaka H, Ito M. Ramosetron compared with granisetron for the prevention of vomiting following strabismus surgery in children. British Journal of Ophthalmology 2001 Jun;85(6):670‐2. [; MEDLINE: ] [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]; 3220288
- Fujii Y, Tanaka H, Kawasaki T. Preoperative oral granisetron for the prevention of postoperative nausea and vomiting after breast surgery. European Journal of Surgery 2001 Mar;167(3):184‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220290
- Fujii Y, Tanaka H, Kawasaki T. Prophylaxis with oral granisetron for the prevention of nausea and vomiting after laparoscopic cholecystectomy: a prospective randomized study. Archives of Surgery 2001 Jan;136(1):101‐4. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220292
- Fujii Y, Uemura A. Dexamethasone for the prevention of nausea and vomiting after dilatation and curettage: a randomized controlled trial. Obstetrics and Gynecology 2002 Jan;99(1):58‐62. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220294
- Fujii Y, Tanaka H. Preoperative oral granisetron for the prevention of vomiting following paediatric surgery. Paediatric Anaesthesia 2002 Mar;12(3):267‐71. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220296
- Fujii Y, Tanaka H. Comparison of granisetron and ramosetron for the prevention of nausea and vomiting after thyroidectomy. Clinical Therapeutics 2002;24(5):766‐72. [; MEDLINE: ; AN: 2002200225] [DOI] [PubMed] [Google Scholar]; 3220298
- Fujii Y, Uemura A, Tanaka H. Prophylaxis of nausea and vomiting after laparoscopic cholecystectomy with ramosetron: Randomised controlled trial. European Journal of Surgery 2002;168(11):583‐6. [; MEDLINE: ; 2003126485 20030403 ISSN: 1102‐4151] [DOI] [PubMed] [Google Scholar]; 3220300
- Fujii Y, Uemura A, Nakano M. Small dose of propofol for preventing nausea and vomiting after third molar extraction. Journal of Oral & Maxillofacial Surgery 2002 Nov;60(11):1246‐9. [; MEDLINE: ; CN‐00411188] [DOI] [PubMed] [Google Scholar]; 3220302
- Fujii Y, Tanaka H. Prevention of nausea and vomiting with ramosetron after total hip replacement. Clinical Drug Investigation 2003;23(6):405‐9. [; 2003243073 20030710] [DOI] [PubMed] [Google Scholar]; 3220304
- Furst SR, Rodarte A. Prophylactic antiemetic treatment with ondansetron in children undergoing tonsillectomy. Anesthesiology 1994;81(4):799‐803. [; MEDLINE: ; EMBASE 1994335709] [DOI] [PubMed] [Google Scholar]; 3220306
- Furst SR, Sullivan LJ, Soriano SG, McDermott JS, Adelson PD, Rockoff MA. Effects of ondansetron on emesis in the first 24 hours after craniotomy in children. Anesthesia & Analgesia 1996 Aug;83(2):325‐8. [; MEDLINE: ; ISI:A1996UZ99100021 ER] [DOI] [PubMed] [Google Scholar]; 3220308
- Galloon S, Gale GD, Lancee WJ. Comparison of lorazepam and diazepam as premedicants. British Journal of Anaesthesia 1977 Dec;49(12):1265‐9. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220310
- Gan TJ, Collis R, Hetreed M. Double‐blind comparison of ondansetron, droperidol and saline in the prevention of postoperative nausea and vomiting. British Journal of Anaesthesia 1994 May;72(5):544‐7. [; MEDLINE: ; EMBASE 1994157538] [DOI] [PubMed] [Google Scholar]; 3220312
- Gan TJ, Alexander R, Fennelly M, Rubin AP. Comparison of different methods of administering droperidol in patient‐controlled analgesia in the prevention of postoperative nausea and vomiting. Anesthesia & Analgesia 1995 Jan;80(1):81‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220314
- Gan TJ, Ginsberg B, Glass PS, Fortney J, Jhaveri R, Perno R. Opioid‐sparing effects of a low‐dose infusion of naloxone in patient‐administered morphine sulfate. Anesthesiology 1997 Nov;87(5):1075‐81. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220316
- Gan TJ, Franiak R, Reeves J. Ondansetron orally disintegrating tablet versus placebo for the prevention of postdischarge nausea and vomiting after ambulatory surgery. Anesthesia and Analgesia 2002;94(5):1199‐200. [; MEDLINE: ; AN: 2002154407] [DOI] [PubMed] [Google Scholar]; 3220318
- Ganem EM, Fabris P, Moro MZ, Castiglia YMM. Efficacy of ondansetron and alizapride in preventing gynecological laparoscopy nausea and vomiting [Eficacia do ondansetron e da alizaprida na prevencao de nausea e vomito em laparoscopia ginecologica]. Revista Brasileira de Anestesiologia 2001;51(5):401‐6. [; AN: 2001346835] [Google Scholar]; 3220320
- Ganem EM, Fukushima FB, Medeiros da Silva DS, Nakamura G, Machado Castiglia YM, Galvão Vianna PT. Efficacy of propofol and propofol plus dexamethasone in controlling postoperative nausea and vomiting of gynecologic laparoscopy. Revista Brasileira de Anestesiologia 2002;52(4):394‐401. [; 2002278207 20020829 ISSN: 0034‐7094] [PubMed] [Google Scholar]; 3220322
- Geens BJ, Genicot C, Primo DJ. Use of RO 5 4200 as hypnotic agent in neuroleptanalgesia. Comparison with methohexital and diazepam [Utilisation du ro 5 4200 comme agent hypnotique dans la neuroleptanalgesie: (comparaison avec le methohexital et le diazepam)]. Anesthesie, analgesie, reanimation 1974;31(5):687‐98. [; EMBASE 1975164617] [PubMed] [Google Scholar]; 3220324
- Geiger C, Kick O, Bohrer H, Motsch J, Martin E. Comparison of ondansetron, metoclopramide and droperidol for the prophylaxis of emetic symptoms after gynaecological laparoscopic procedures. British Journal of Anaesthesia 1993 May;70(suppl 1):A14. [] [Google Scholar]; 3220326
- Gentili M, Enel D, Szymskiewicz O, Mansour F, Bonnet F. Postoperative analgesia by intraarticular clonidine and neostigmine in patients undergoing knee arthroscopy. Regional anesthesia and pain medicine 2001 July;26(4):342‐7. [; MEDLINE: ; ISI:000170071500012 ER] [DOI] [PubMed] [Google Scholar]; 3220328
- Gesztesi Z, Scuderi PE, White PF, Wright W, Wender RH, D'Angelo R, et al. Substance P (neurokinin‐1) antagonist prevents postoperative vomiting after abdominal hysterectomy procedure. Anesthesiology 2000;93(4):931‐7. [; MEDLINE: ; AN: 2000360774] [DOI] [PubMed] [Google Scholar]; 3220330
- Giannoni C, White S, Enneking FK. Does dexamethasone with preemptive analgesic improve pediatric tonsillectomy pain?. Otolaryngology ‐ Head & Neck Surgery 2002 Mar;126(3):307‐16. [; MEDLINE: ; ISI:000175371100015 ER] [DOI] [PubMed] [Google Scholar]; 3220332
- Gilliland HEM, Prasad BK, Mirakhur RK, Fee JPH. An investigation of the potential morphine sparing effect of midazolam. Anaesthesia 1996;51(9):808‐11. [; EMBASE 1996283659] [DOI] [PubMed] [Google Scholar]; 3220334
- Godsiff L, Magee L, Park GR. Propofol versus propofol with midazolam for laryngeal mask insertion. European Journal of Anaesthesiology Supplement 1995 Nov;12:35‐40. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220336
- Goksu S, Kocoglu H, Bayazit YA, Yuksek S, Karci Y, Kanlikama M, et al. Antiemetic effects of granisetron, droperidol and dexamethasone in otologic surgery. Auris Nasus Larynx 2002 Jul;29(3):253‐6. [; MEDLINE: ; CN‐00409407] [DOI] [PubMed] [Google Scholar]; 3220338
- Goll V, Akca O, Greif R, Freitag H, Arkilic CF, Scheck T, et al. Ondansetron is no more effective than supplemental intraoperative oxygen for prevention of postoperative nausea and vomiting. Anesthesia & Analgesia 2001 Jan;92(1):112‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220340
- Goodarzi M. A double blind comparison of droperidol and ondansetron for prevention of emesis in children undergoing orthopaedic surgery. Paediatric Anaesthesia 1998;8(4):325‐9. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220342
- Goode K, Laeder C. A comparison of the efficacy of intravenous granisetron and ondansetron in preventing postoperative vomiting in pediatric tonsillectomy and adenoidectomy procedures. Journal of the American Association of Nurse Anesthetists 1997;65(4):385‐6. [; AN: 1997266271] [Google Scholar]; 3220344
- Gordon NH, Turner DJ. Oral paediatric premedication. A comparative trial of either phenobarbitone, trimeprazine or diazepam with hyoscine, prior to guillotine tonsillectomy. British Journal of Anaesthesia 1969 Feb;41(2):136‐42. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220346
- Gougeon SD. Bromopride for prevention of nausea and vomiting caused by epidural morphine [Bromoprida na prevençäo de náuseas e vômitos causados por morfina peridural]. Revista Brasileira de Anestesiologia 1988 May;38(3):193‐6. [; LILACS Id: 63151] [Google Scholar]; 3220348
- Goyagi T, Nishikawa T. Oral clonidine premedication enhances the quality of postoperative analgesia by intrathecal morphine. Anesthesia & Analgesia 1996 Jun;82(6):1192‐6. [; MEDLINE: ; ISI:A1996UM65500016 ER] [DOI] [PubMed] [Google Scholar]; 3220350
- Goyagi T, Tanaka M, Nishikawa T. Oral clonidine premedication enhances postoperative analgesia by epidural morphine. Anesthesia & Analgesia 1999;89(6):1487‐91. [; MEDLINE: ; AN: 1999424043] [DOI] [PubMed] [Google Scholar]; 3220352
- Grace D, Bunting H, Milligan KR, Fee JPH. Postoperative analgesia after coadministration of clonidine and morphine by the intrathecal route in patients undergoing hip‐replacement. Anesthesia & Analgesia 1995 Jan;80(1):86‐91. [; MEDLINE: ; ISI:A1995PY27000015 ER] [DOI] [PubMed] [Google Scholar]; 3220354
- Grebenik CR, Allman C. Nausea and vomiting after cardiac surgery. British Journal of Anaesthesia 1996;77(3):356‐9. [; EMBASE 1996278666] [DOI] [PubMed] [Google Scholar]; 3220356
- Greif R, Laciny S, Rapf B, Hickle RS, Sessler DI. Supplemental oxygen reduces the incidence of postoperative nausea and vomiting. Anesthesiology 1999 Nov;91(5):1246‐52. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220358
- Grimsehl K, Whiteside JB, Mackenzie N. Comparison of cyclizine and ondansetron for the prevention of postoperative nausea and vomiting in laparoscopic day‐case gynaecological surgery. Anaesthesia 2002 Jan;57(1):61‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220360
- Grond S, Lynch J, Diefenbach C, Altrock K, Lehmann KA. Comparison of ondansetron and droperidol in the prevention of nausea and vomiting after inpatient minor gynecologic surgery. Anesthesia & Analgesia 1995 Sep;81(3):603‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220362
- Grottke O, Muller J, Dietrich PJ, Krause TH, Wappler F. Comparison of Premedication with Clonidine and Midazolam Combined with TCI for Orthopaedic Shoulder Surgery. Anasthesiologie Intensivmedizin Notfallmedizin Schmerztherapie 2003 Dec;38(12):772‐80. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220364
- Guard BC, Wiltshire SJ. The effect of glycopyrrolate on postoperative pain and analgesic requirements following laparoscopic sterilisation. Anaesthesia 1996;51(12):1173‐5. [; MEDLINE: ; AN: 1997034157] [DOI] [PubMed] [Google Scholar]; 3220366
- Guldager H, Jensen FM, Andersen KH, Arnfred I. Domperidone and metoclopramide in the prophylaxis of postoperative nausea and vomiting. Ugeskrift for Laeger 1983;145(23):1777‐9. [; MEDLINE: ; AN: 1983210928] [PubMed] [Google Scholar]; 3220368
- Guldogus F, Sarihasan B, Esener Z, Karakaya D, Tur A, Ustun ESO. The effect of a single dose of ondansetron on postoperative nausea and vomiting. Turk Anesteziyoloji ve Reanimasyon 1994;22(2):77‐80. [; no number] [Google Scholar]; 3220370
- Gulec S, Buyukkidan B, Oral N, Ozcan N, Tanriverdi B. Comparison of caudal bupivacaine, bupivacaine morphine and bupivacaine‐midazolam mixtures for post‐operative analgesia in children. European Journal of Anaesthesiology 1998 Mar;15(2):161‐5. [; MEDLINE: ; ISI:000073118800007 ER] [PubMed] [Google Scholar]; 3220372
- Gulhas N, Turkoz A, Durmus M, Togal T, Gedik E, Ersoy MO. Oral clonidine premedication does not reduce postoperative vomiting in children undergoing strabismus surgery. Acta Anaesthesiologica Scandinavica 2003 Jan;47(1):90‐3. [; MEDLINE: ; CN‐00422348] [DOI] [PubMed] [Google Scholar]; 3220374
- Günes Y, Ünlügenç H, Gündüz M, Özbek H, Özalevli M, Aliç V, et al. Comparison of Morphine‐Ondansetron and Morphine‐Dexamethasone Infusions as Patient‐Controlled Analgesia After Laminectomy. Turk Anesteziyoloji ve Reanimasyon Dernegi Dergisi May 2003;31(5):231‐6. [; 2003420712 20031106 ISSN: 1304‐0871] [Google Scholar]; 3220376
- Gurler T, Celik N, Totan S, Songur E, Sakarya M. Prophylactic use of ondansetron for emesis after craniofacial operations in children. Journal of Craniofacial Surgery 1999 Jan;10(1):45‐8. [; MEDLINE: ; ISI:000077966200010] [DOI] [PubMed] [Google Scholar]; 3220378
- Gurses E, Sungurtekin H, Tomatir E, Balci C, Gonullu M. The addition of droperidol or clonidine to epidural tramadol shortens onset time and increases duration of postoperative analgesia. Canadian Journal of Anaesthesia 2003 Feb;50(2):147‐52. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220380
- Hagemann E, Halvorsen A, Holgersen O, Tveit T, Raeder JC. Granisetron/dexamethasone combination for the prevention of postoperative nausea and vomiting after laparoscopic cholecystectomy. Acta Anaesthesiologica Scandinavica 2000 Jan;44(1):107‐11. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220382
- Halvorsen P, Raeder J, White PF, Almdahl SM, Nordstrand K, Saatvedt K, et al. The effect of dexamethasone on side effects after coronary revascularization procedures. Anesthesia & Analgesia 2003 Jun;96(6):1578‐83. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220384
- Hamid SK, Selby IR, Sikich N, Lerman J. Vomiting after adenotonsillectomy in children: a comparison of ondansetron, dimenhydrinate, and placebo. Anesthesia & Analgesia 1998 Mar;86(3):496‐500. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220386
- Hammas B, Thorn SE, Wattwil M. Superior prolonged antiemetic prophylaxis with a four‐drug multimodal regimen ‐ comparison with propofol or placebo. Acta Anaesthesiologica Scandinavica 2002 Mar;46(3):232‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220388
- Hammond J, Elwood R, Tarr T, Simpson D. Comparison of anti‐emetics. Anaesthesia 1985 Apr;40(4):382. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220390
- Handa F, Fujii Y. The efficacy of oral clonidine premedication in the prevention of postoperative vomiting in children following strabismus surgery. Paediatric Anaesthesia 2001;11(1):71‐4. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220392
- Handley AJ. Metoclopramide in the prevention of post‐operative nausea and vomiting. British Journal of Clinical Practice 1967 Sep;21(9):460‐2. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220394
- Hardy JF, Charest J, Girouard G, Lepage Y. Nausea and vomiting after strabismus surgery in preschool children. Canadian Anaesthetists Society Journal 1986 Jan;33(1):57‐62. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220396
- Harris SN, Sevarino FB, Sinatra RS, Preble L, O'Connor TZ, Silverman DG. Nausea prophylaxis using transdermal scopolamine in the setting of patient‐controlled analgesia. Obstetrics & Gynecology 1991 Oct;78(4):673‐7. [; MEDLINE: ; ISI:A1991GH11700020 ER] [PubMed] [Google Scholar]; 3220398
- Harti A, Bouaggad A, Benaguida M. Prevention of postoperative vomiting after epidural anesthesia (droperidol versus metoclopramide) [Prevention des vomissements postoperatoires apres anesthesies peridurale (droperidol versus metoclopramide)]. Urgences‐Medicales 1994;13(5):223‐5. [; AN: 1994347404] [Google Scholar]; 3220400
- Hechler A, Neumann S, Jehmlich M, Lange U, Hopf HB. A small dose of droperidol decreases postoperative nausea and vomiting in adults but cannot improve an already excellent patient satisfaction. Acta Anaesthesiologica Scandinavica 2001 Apr;45(4):501‐6. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220402
- Helmers JH, Briggs L, Abrahamsson J, Soni J, Moodley J, Forrler M, et al. A single i.v. dose of ondansetron 8 mg prior to induction of anaesthesia reduces postoperative nausea and vomiting in gynaecological patients. Canadian Journal of Anaesthesia 1993 Dec;40(12):1155‐61. [; MEDLINE: ; EMBASE 1994023531] [DOI] [PubMed] [Google Scholar]; 3220404
- Helmy SA. Prophylactic anti‐emetic efficacy of ondansetron in laparoscopic cholecystectomy under total intravenous anaesthesia. A randomised, double‐blind comparison with droperidol, metoclopramide and placebo. Anaesthesia 1999 Mar;54(3):266‐71. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220406
- Hildyard CL, Quinn A, Dresner M, Freeman J. Cyclizine reduces the incidence of nausea and vomiting after elective caesarean section with intrathecal diamorphine. British Journal of Anaesthesia 2001;87(4):672P. [; CN‐00370416] [Google Scholar]; 3220408
- Ho ST, Wang JJ, Tzeng JI, Liu HS, Ger LP, Liaw WJ. Dexamethasone for preventing nausea and vomiting associated with epidural morphine: A dose‐raging study. Anesthesia & Analgesia 2001;92(3):745‐8. [; MEDLINE: ; AN: 2001086757] [DOI] [PubMed] [Google Scholar]; 3220410
- Holt R, Rask P, Coulthard KP, Sinclair M, Roberts G, Walt J, et al. Tropisetron plus dexamethasone is more effective than tropisetron alone for the prevention of postoperative nausea and vomiting in children undergoing tonsillectomy. Paediatric Anaesthesia 2000;10(2):181‐8. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220412
- Honkavaara P, Saarnivaara L, Klemola UM. Prevention of nausea and vomiting with transdermal hyoscine in adults after middle‐ear surgery during general anesthesia. British Journal of Anaesthesia 1994 Dec;73(6):763‐6. [; MEDLINE: ; ISI:A1994PU35500008 ER] [DOI] [PubMed] [Google Scholar]; 3220414
- Honkavaara P, Saarnivaara L, Klemola UM. Effect of transdermal hyoscine on nausea and vomiting after surgical correction of prominent ears under general anaesthesia. British Journal of Anaesthesia 1995 Jun;74(6):647‐50. [; MEDLINE: ; EMBASE 1995168110] [DOI] [PubMed] [Google Scholar]; 3220416
- Honkavaara P. Effect of transdermal hyoscine on nausea and vomiting during and after middle ear surgery under local anaesthesia. British Journal of Anaesthesia 1996;76(1):49‐53. [; MEDLINE: ; EMBASE 1996034118] [DOI] [PubMed] [Google Scholar]; 3220418
- Honkavaara P. Effect of ondansetron on nausea and vomiting after middle ear surgery during general anaesthesia. British Journal of Anaesthesia 1996 Feb;76(2):316‐8. [; MEDLINE: ; EMBASE 1996050100] [DOI] [PubMed] [Google Scholar]; 3220420
- Honkavaara P, Pyykko I. Effects of atropine and scopolamine on bradycardia and emetic symptoms in otoplasty. Laryngoscope 1999 Jan;109(1):108‐12. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220422
- Horimoto Y, Tomie H, Hanzawa K, Nishida Y. Scopolamine patch reduces postoperative emesis in pediatric‐ patients following strabismus surgery. Canadian Journal of Anaesthesia 1991;38(4):441‐4. [; MEDLINE: ; ISI:A1991FL35100005 ER] [DOI] [PubMed] [Google Scholar]; 3220424
- Horta ML, Horta BL. Inhibition of epidural morphine‐induced pruritus by intravenous droperidol. Regional Anesthesia 1993 Mar‐Apr;18(2):118‐20. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220426
- Hovi‐Viander M, Kangas L, Kanto J. A comparative study of the clinical effects of pentobarbital and diazepam given orally as preoperative medication. Journal of Oral Surgery 1980 Mar;38(3):188‐90. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220428
- Hovorka J, Korttila K, Nelskyla K, Soikkeli A, Sarvela J, Paatero H, et al. Reversal of neuromuscular blockade with neostigmine has no effect on the incidence or severity of postoperative nausea and vomiting. Anesthesia & Analgesia 1997 Dec;85(6):1359‐61. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220430
- Huang JC, Shieh JP, Tang CS, Tzeng JI, Chu KS, Wang JJ. Low‐dose dexamethasone effectively prevents postoperative nausea and vomiting after ambulatory laparoscopic surgery. Canadian Journal of Anesthesia 2001;48(10):973‐7. [; MEDLINE: ; AN: 2002039197] [DOI] [PubMed] [Google Scholar]; 3220432
- Hunting A, Kjaersgaard P, Hjelle K, Skovlund E, Aune H. b. Acta Anaesthesiologica Scandinavica 1997;41(s110):s182. [] [Google Scholar]; 3220434
- Huston CL, Sheridan CA, Ungard SD, Selley K, Hagan A, Nagelhout J, et al. Comparison of oral granisetron, intravenous granisetron, and droperidol in the prevention of nausea and vomiting after outpatient laparoscopic procedures. Journal of the American Association of Nurse Anesthetists 1996;64(5):437‐8. [; AN: 1996345121] [Google Scholar]; 3220436
- Hyrkas T, Ylipaavalniemi P, Oikarinen VJ, Paakkari I. A comparison of diclofenac with and without single‐dose intravenous steroid to prevent postoperative pain after third molar removal. Journal of Oral and Maxillofacial Surgery 1993;51(6):634‐6. [; MEDLINE: ; AN: 1993163456] [DOI] [PubMed] [Google Scholar]; 3220438
- Iannuzzi E, Minichini A, Galluccio V, Perna S, Papa A, Ferraro F. Nausea and vomiting in biliary tract's surgery: Usage of ondansetron [Nausea e vomito nella chirurgia delle vie biliari: utilizzazione dell'ondasetron]. Rassegna Internazionale di Clinica e Terapia 1994;74(3):97‐101. [; AN: 1994193192] [Google Scholar]; 3220440
- Ilbeigi MS, Ponnudurani R, Liu P. A comparison of the efficacy of ondansetron or metoclopramide or ondansetron and metoclopramide in the prevention of postoperative nausea and vomiting. Anesthesia & Analgesia 1999 Feb;88:s348. [; ISI:000078507400344 ER] [Google Scholar]; 3220442
- Imbeloni L. Comparison between oral diazepam and sublingual flunitrazepam for premedication in peridural block [Comparaçäo entre Diazepam por via oral e flunitrazepam sublingual como medicaçäo pré‐anestésica em bloqueio peridural]. Revista Brasileira de Anestesiologia 1987 Jan;37(1):29‐33. [; LILACS Id: 40759] [Google Scholar]; 3220444
- Ismail OM, Said MA, Serag El Din MN. Vomiting after strabismus surgery in children: Propofol versus propofol and dexamethasone. Egyptian Journal of Anaesthesia 2003;19(3):305‐10. [; ISSN: 1110‐1849] [Google Scholar]; 3220446
- Iwamoto K, Schwartz H. Antiemetic effect of droperidol after ophthalmic surgery. Archives of Ophthalmology 1978 Aug;96(8):1378‐9. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220448
- Jakobsson J, Dahlgren G, Oddby E, Ryberg G. Nausea and vomiting after laparoscopic gynecologic surgery: a study of the incidence and the effects of tropisetron prophylaxis. Journal of Laparoendoscopic & Advanced Surgical Techniques. Part A 1999 Feb;9(2):141‐5. [; MEDLINE: ; ISI:000079145200007 ER] [DOI] [PubMed] [Google Scholar]; 3220450
- Janknegt R, Pinckaers JW, Rohof MH, Ausems ME, Arbouw ME, Velden RW, et al. Double‐blind comparative study of droperidol, granisetron and granisetron plus dexamethasone as prophylactic anti‐emetic therapy in patients undergoing abdominal, gynaecological, breast or otolaryngological surgery see comments. Anaesthesia 1999;54(11):1059‐68. [; CN‐00306678] [DOI] [PubMed] [Google Scholar]; 3220452
- Jeffs SA, Hall JE, Morris S. Comparison of morphine alone with morphine plus clonidine for postoperative patient‐controlled analgesia. British Journal of Anaesthesia 2002;89(3):424‐7. [; MEDLINE: ; IS: 0007‐0912] [PubMed] [Google Scholar]; 3220454
- Jellish WS, Leonetti JP, Murdoch JR, Fowles S. Propofol‐based anesthesia as compared with standard anesthetic techniques for middle ear surgery. Otolaryngology ‐ Head & Neck Surgery 1995 Feb;112(2):262‐7. [; MEDLINE: ; EMBASE 1995198772] [DOI] [PubMed] [Google Scholar]; 3220456
- Jellish WS, Thalji Z, Fluter E, Leonetti JP. Ondansetron versus droperidol or placebo when given prophylactically for the prevention of postoperative nausea and vomiting in patients undergoing middle ear procedures. Journal of Clinical Anesthesia 1997;9(6):451‐6. [; MEDLINE: ; AN: 1997256490] [DOI] [PubMed] [Google Scholar]; 3220458
- Jellish WS, Abodeely A, Fluder EM, Shea J. The effect of spinal bupivacaine in combination with either epidural clonidine and/or 0.5% bupivacaine administered at the incision site on postoperative outcome in patients undergoing lumbar laminectomy. Anesthesia & Analgesia 2003 Mar;96(3):874‐80. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220460
- Jensen AB, Christiansen DB, Coulthard K, Wilkins A, Roberts G, Walt JH, et al. Tropisetron reduces postoperative vomiting in children undergoing tonsillectomy. Paediatric Anaesthesia 2000;10(1):69‐75. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220462
- Jokela R, Koivuranta M. Tropisetron or droperidol in the prevention of postoperative nausea and vomiting. A comparative, randomised, double‐blind study in women undergoing laparoscopic cholecystectomy. Acta Anaesthesiologica Scandinavica 1999 Jul;43(6):645‐50. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220464
- Jokela RM, Kangas‐Saarela TA, Valanne JV, Koivuranta MK, Ranta PO, Alahuhta SM. Postoperative nausea and vomiting after sevoflurane with or without ondansetron compared with propofol in female patients undergoing breast surgery. Anesthesia & Analgesia 2000 Nov;91(5):1062‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220466
- Jokela R, Koivuranta M, Kangas‐Saarela T, Purhonen S, Alahuhta S. Oral ondansetron, tropisetron or metoclopramide to prevent postoperative nausea and vomiting: a comparison in high‐risk patients undergoing thyroid or parathyroid surgery. Acta Anaesthesiologica Scandinavica 2002 May;46(5):519‐24. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220468
- Jorgensen NH, Coyle JP. Intravenous droperidol decreases nausea and vomiting after alfentanil anesthesia without increasing recovery time. Journal of Clinical Anesthesia 1990 Sep‐Oct;2(5):312‐6. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220470
- Joris JL, Poth NJ, Djamadar AM, Sessler DI, Hamoir EE, Defechereux TR, et al. Supplemental oxygen does not reduce postoperative nausea and vomiting after thyroidectomy. British Journal of Anaesthesia 2003 Dec;91(6):857‐61. [; MEDLINE: ] [DOI] [PMC free article] [PubMed] [Google Scholar]; 3220472
- Joshi R, Sivaganesanathan A. Tiapride versus metoclopramide: comparison after minor gynaecological surgery. European Journal of Anaesthesiology 1993 Mar;10(2):109‐12. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220474
- Kafali IH, Mimaroglu C, Ozok U, Karsli B. Antiemetic effect of ondansetron on abdominal gynecologic operations. Turk Anesteziyoloji ve Reanimasyon 1994;22(5):261‐4. [; AN: 1994358269] [Google Scholar]; 3220476
- Kandler D, Lisander B. Analgesic action of metoclopramide in prosthetic hip surgery. Acta Anaesthesiologica Scandinavica 1993;37(1):49‐53. [; MEDLINE: ; AN:1993014654] [DOI] [PubMed] [Google Scholar]; 3220478
- Karabayirh S, Alver F, Alkis N. Comparision of the supplemental oxygen, dexametasone and ondansetrone for prevention of postoperative nausea and vomiting. Turk Anesteziyoloji Ve Reanimasyon 2003;31(3):110‐5. [; CN‐00431795] [Google Scholar]; 3220480
- Karamanlioglu B, Turan A, Memis D, Sut N. Comparison of oral dolasetron and ondansetron in the prophylaxis of postoperative nausea and vomiting in children. European Journal of Anaesthesiology 2003 Oct;20(10):831‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220482
- Karhunen U, Orko R. Nausea and vomiting after local anesthesia for cataract extraction in elderly female patients ‐‐ effect of droperidol premedication. Ophthalmic Surgery 1981 Nov;12(11):810‐2. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220484
- Karlsson E, Larsson LE, Nilsson K. The effects of prophylactic dixyrazine on postoperative vomiting after two different anaesthetic methods for squint surgery in children. Acta Anaesthesiologica Scandinavica 1993 Jan;37(1):45‐8. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220486
- Katayama M, Vieira JL, Aléssio FP de, Santos GV dos, Gomes CC. Importance of droperidol and metropol in anesthesia with propofol, alfentanil and nitrous oxide for septoplasties and turbinectomies [Importância do droperidol e do metoprolol na anestesia com profolol, alfentanil e óxido nitroso para septoplasias e turbinectomias]. Revista Brasileira de Anestesiologia 1995 Jul;45(4):225‐34. [; Id: 166852] [Google Scholar]; 3220488
- Kathirvel S, Balachundhar S, Bhatia A, Dash HH, Chaturvedi A. Postoperative nausea and vomiting after elective craniotomy. Anesthesiology 1998 Sep;89:A376. [; SI:000075810900376 ER] [DOI] [PubMed] [Google Scholar]; 3220490
- Kathirvel S, Shende D, Madan R. Comparison of anti‐emetic effects of ondansetron, metoclopromide or a combination of both in children undergoing surgery for strabismus. European Journal of Anaesthesiology 1999 Nov;16(11):761‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220492
- Kathirvel S, Dash HH, Bhatia A, Subramaniam B, Prakash A, Shenoy S. Effect of prophylactic ondansetron on postoperative nausea and vomiting after elective craniotomy. Journal of Neurosurgical Anesthesiology 2001 Jul;13(3):207‐12. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220494
- Kaufman SL, Martin SD. Effectiveness of ondansetron compared to metoclopramide and placebo in reducing postoperative emesis in children undergoing ophthalmic surgery. Journal of the American Association of Nurse Anesthetists 1996;64(5):438‐9. [; EMBASE 1996345122] [Google Scholar]; 3220496
- Kaufmann MA, Rosow C, Schnieper P, Schneider M. Prophylactic antiemetic therapy with patient‐controlled analgesia: a double‐blind, placebo‐controlled comparison of droperidol, metoclopramide, and tropisetron. Anesthesia & Analgesia 1994 May;78(5):988‐94. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220498
- Kaul HL, Rao U, Mandal NG, Rahman A. Comparative evaluation of single dose oral Ondansetron and Metoclopramide in a placebo controlled study for prevention of postoperative nausea and vomiting. Journal of Anaesthesiology Clinical Pharmacology 1996;12(1):27‐30. [; EMBASE 1996075560] [Google Scholar]; 3220500
- Kauste A, Tuominen M, Heikkinen H, Gordin A, Korttila K. Droperidol, alizapride and metoclopramide in the prevention and treatment of post‐operative emetic sequelae. European Journal of Anaesthesiology 1986;3(1):1‐9. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220502
- Kaya FN, Uçkunkaya N, Sahin S. Postoperative analgesic effects of intrathecal neostigmine, morphine and their combination and influence on spinal anaesthesia. Turk Anesteziyoloji ve Reanimasyon 2002;30(8):353‐9. [; 2002417374 20021212 ISSN: 1016‐515] [Google Scholar]; 3220504
- Khalil SN, Berry JM, Howard G, Lawson K, Hanis C, Mazow ML, et al. The antiemetic effect of lorazepam after outpatient strabismus surgery in children. Anesthesiology 1992;77(5):915‐9. [; MEDLINE: ; AN: 1992345445] [DOI] [PubMed] [Google Scholar]; 3220506
- Khalil S, Wells L, Rabb M, Villanueva V, Hanis C. Promethazine hydrochloride is the antiemetic of choice to prevent postoperative emesis in children after middle ear surgery. Anesthesiology 1996 Sep;85:A1102. [; ISI:A1996VM46601102 ER] [Google Scholar]; 3220508
- Khalil S, Baerenstecher J, Amhan I, Villanueva G, Hanis C. Promethazine hydrochloride may be the antiemetic of choice to prevent postoperative nausea and emesis after middle ear surgery in adults. Anesthesia & Analgesia 1997 Feb;84(SU 2):S40. [; ISI:A1997WF78000040 ER] [Google Scholar]; 3220510
- Khalil S, Philbrook L, Rabb M, Wells L, Aves T, Villanueva G, et al. Ondansetron/promethazine combination or promethazine alone reduces nausea and vomiting after middle ear surgery. Journal of Clinical Anesthesia 1999 Nov;11(7):596‐600. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220512
- Kimya Y, Tatlikazan S, Bilgin H, Bilgin T, Cengiz C. Ondansetron: The prevention of nausea and vomiting in gynecologic operations. Turkish Journal of Medical Sciences 1996;26(4):339‐42. [; AN: 1996283379] [Google Scholar]; 3220514
- King MJ, Milazkiewicz R, Carli F, Deacock AR. Influence of neostigmine on postoperative vomiting. British Journal of Anaesthesia. 1988;61(4):403‐6. [; MEDLINE: ; ISI:A1988Q479700007 ER] [DOI] [PubMed] [Google Scholar]; 3220516
- Klahsen AJ, O'Reilly D, McBride J, Ballantyne M, Parlow JL. Reduction of post‐operative nausea and vomiting with the combination of morphine and droperidol in patient‐controlled analgesia. Canadian Journal of Anaesthesia 1996 Nov;43(11):1100‐7. [; MEDLINE: ; EMBASE 1997044135] [DOI] [PubMed] [Google Scholar]; 3220518
- Klockgether‐Radke A, Feldmann M, Braun U, Muhlendyck H. Antiemetic effects of droperidol versus metoclopramide after strabismus surgery in children [Droperidol vs. Metoclopramid. Prophylaxe von erbrechen nach strabismus‐operationen bei kindern]. Anaesthesist 1992;41(5):254‐9. [; EMBASE 1992177987] [PubMed] [Google Scholar]; 3220520
- Klockgether‐Radke A, Demmel C, Braun U, Muhlendyck H. Vomiting and the oculocardiac reflex in children undergoing strabismus surgery. Prophylactic effects of droperidol and atropine [Erbrechen und okulokardialer reflex. Medikamentose prophylaxe mit droperidol und atropin bei kindern mit strabismusoperation]. Anaesthesist 1993;42(6):356‐60. [; EMBASE 1993189325] [PubMed] [Google Scholar]; 3220522
- Klockgether‐Radke A, Neumann S, Neumann P, Braun U, Muhlendyck H. Ondansetron, droperidol and their combination for the prevention of post‐operative vomiting in children. European Journal of Anaesthesiology 1997 Jul;14(4):362‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220524
- Knudsen K, Lisander B. Metoclopramide decreases emesis after spinal anesthesia supplemented with subarachnoid morphine. Regional Anesthesia 1994 Nov‐Dec;19(6):390‐4. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220526
- Koivuranta MK, Laara E, Ryhanen PT. Antiemetic efficacy of prophylactic ondansetron in laparoscopic cholecystectomy. A randomised, double‐blind, placebo‐controlled trial. Anaesthesia 1996 Jan;51(1):52‐5. [; MEDLINE: ; EMBASE 1996117356] [DOI] [PubMed] [Google Scholar]; 3220528
- Koivuranta M, Jokela R, Kiviluoma K, Alahuhta S. The anti‐emetic efficacy of a combination of ondansetron and droperidol. Anaesthesia 1997 Sep;52(9):863‐8. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220530
- Koivuranta M, Laara E, Ranta P, Ravaska P, Alahuhta S. Comparison of ondansetron and droperidol in the prevention of postoperative nausea and vomiting after laparoscopic surgery in women. A randomised, double‐blind, placebo‐controlled trial. Acta Anaesthesiologica Scandinavica 1997 Nov;41(10):1273‐9. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220532
- Koivuranta M, Ala‐Kokko TI, Jokela R, Ranta P. Comparison of ondansetron and tropisetron combined with droperidol for the prevention of emesis in women with a history of post‐operative nausea and vomiting. European Journal of Anaesthesiology 1999 Jun;16(6):390‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220534
- Kokinsky E, Thornberg E, Nilsson K, Larsson LE. Postoperative nausea and vomiting in children using patient‐controlled analgesia: the effect of prophylactic intravenous dixyrazine. Acta Anaesthesiologica Scandinavica 1999 Feb;43(2):191‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220536
- Korttila K, Kauste A, Auvinen J. Comparison of domperidone, droperidol, and metoclopramide in the prevention and treatment of nausea and vomiting after balanced general anesthesia. Anesthesia & Analgesia 1979 Sep‐Oct;58(5):396‐400. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220538
- Korttila K, Kauste A, Tuominen M, Salo H. Droperidol prevents and treats nausea and vomiting after enflurane anaesthesia. European Journal of Anaesthesiology 1985 Dec;2(4):379‐85. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220540
- Korttila K, Clergue F, Leeser J, Feiss P, Olthoff D, Payeur‐Michel C, et al. Intravenous dolasetron and ondansetron in prevention of postoperative nausea and vomiting: A multicenter, double‐blind, placebo‐controlled study. Acta Anaesthesiologica Scandinavica 1997;41(7):914‐22. [; MEDLINE: ; AN: 1997226699] [DOI] [PubMed] [Google Scholar]; 3220542
- Koski EMJ, Mattila MAK, Knapik D, Toivanen T, Ruusukallio H, Andersson P, et al. Double‐blind comparison of transdermal hyoscine and placebo for the prevention of postoperative nausea. British Journal of Anaesthesia 1990;64(1):16‐20. [; ISI:A1990CK10600005 ER] [DOI] [PubMed] [Google Scholar]; 3220544
- Kotake Y, Matsumoto M, Ai K, Morisaki H, Takeda J. Additional droperidol, not butorphanol, augments epidural fentanyl analgesia following anorectal surgery. Journal of Clinical Anesthesia 2000 Feb;12(1):9‐13. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220546
- Kothari SN, Boyd WC, Bottcher ML, Lambert PJ. Antiemetic efficacy of prophylactic dimenhydrinate (Dramamine) vs ondansetron (Zofran): A randomized, prospective trial in patients undergoing laparoscopic cholecystectomy. Surgical Endoscopy 2000;14(10):926‐9. [; MEDLINE: ; AN: 2000418786] [DOI] [PubMed] [Google Scholar]; 3220548
- Kovac AL, Pearman MH, Khalil SN, Scuderi PE, Joslyn AF, Prillaman BA, et al. Ondansetron prevents postoperative emesis in male outpatients. S3A‐379 Study Group. Journal of Clinical Anesthesia 1996 Dec;8(8):644‐51. [; PMID: 8982892] [DOI] [PubMed] [Google Scholar]; 3220550
- Kranke P, Apfel CC, Papenfuss T, Lengler R, Sefrin P, Roewer N. Effect of the timing of metoclopramide on postoperative nausea and vomiting after inhalational anesthesia. Anesthesia & Analgesia 1999 Feb;88:s17. [; ISI:000078507400018 ER] [Google Scholar]; 3220552
- Kranke P, Apfel CC, Papenfuss T, Ruebsam B, Sefrin P, Roewer N. Tropisetron for the prevention of postoperative nausea and vomiting after ENT‐ and strabismus‐surgery?. Anesthesia & Analgesia 1999 Feb;88:s18. [; ISI:000078507400019 ER] [Google Scholar]; 3220554
- Kraus GB, Giebner M, Palackal R. [The prevention of postoperative vomiting following strabismus surgery in children] [Die Prophylaxe des postoperativen Erbrechens nach Strabismusoperationen bei Kindern]. Anaesthesist 1991;40(2):92‐5. [; MEDLINE: ; CN‐00217779] [PubMed] [Google Scholar]; 3220556
- Kreisler NS, Spiekermann BF, Ascari CM, Rhyne HA, Kloth RL, Sullivan LM, et al. Small‐dose droperidol effectively reduces nausea in a general surgical adult patient population. Anesthesia & Analgesia 2000 Nov;91(5):1256‐61. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220558
- Ku PK, Tong MC, Lo P, Hasselt CA. Efficacy of ondansetron for prevention of postoperative nausea and vomiting after outpatient ear surgery under local anesthesia. The American Journal of Otology 2000 Jan;21(1):24‐7. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220560
- Kumar A, Bose S, Bhattacharya A, Tandon OP, Kundra P. Oral clonidine premedication for elderly patients undergoing intraocular surgery. Acta Anaesthesiologica Scandinavica 1992 Feb;36(2):159‐64. [; MEDLINE: ; ISI:000089776700006 ER] [DOI] [PubMed] [Google Scholar]; 3220562
- Kumar A, Agarwal M, Bhattacharya A. Effect of Ondansetron and metoclopramide on vigilance, cognition and recovery time following major gynaecological surgery. Journal of Anaesthesiology Clinical Pharmacology 1996;12(1):35‐8. [; EMBASE 1996075562] [Google Scholar]; 3220564
- Kymer PJ, Brown RE Jr, Lawhorn CD, Jones E, Pearce L. The effects of oral droperidol versus oral metoclopramide versus both oral droperidol and metoclopramide on postoperative vomiting when used as a premedicant for strabismus surgery. Journal of Clinical Anesthesia 1995 Feb;7(1):35‐9. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220566
- Kyokong O, Visalyaputra S, Saratan P, Somboonviboon W, Pausawadi S, Vongvises P. Comparison of ondansetron and placebo for preventing postoperative nausea and emesis in gastrointestinal tract surgery: A multicenter randomized controlled trial. Journal of the Medical Association of Thailand 1999;82(2):173‐7. [; MEDLINE: ; AN: 1999132993] [PubMed] [Google Scholar]; 3220568
- Lamond CT, Robinson DL, Boyd JD, Cashman JN. Addition of droperidol to morphine administered by the patient‐controlled analgesia method: what is the optimal dose?. European Journal of Anaesthesiology 1998 May;15(3):304‐9. [; MEDLINE: ; CN‐00152346] [DOI] [PubMed] [Google Scholar]; 3220570
- Laraki M, Mouknia M, Bouaguad A, Idali B, Benaguida M. Preventing postoperative nausea and vomiting with lorazepam [Prevention des nausees et vomissements postoperatoires par le lorazepam]. Cahiers D'Anesthesiologie 1996;44(2):159‐62. [; MEDLINE: ; EMBASE 1996168861] [PubMed] [Google Scholar]; 3220572
- Larsson S, Hagerdal M, Lundberg D. Premedication with intramuscular dixyrazine: (Esucos). A controlled double‐blind comparison with morphine‐scopolamine and placebo. Acta Anaesthesiologica Scandinavica 1988 Feb;32(2):131‐4. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220574
- Larsson S, Jonmarker C. Postoperative emesis after pediatric strabismus surgery: the effect of dixyrazine compared to droperidol. Acta Anaesthesiologica Scandinavia 1990 Apr;34(3):227‐30. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220576
- Latasch L, Muller B, Freye E. Postoperative routine use of physostigmine on vigilance, cardiovascular parameters and need of analgesics. Anasthesiologie Intensivmedizin Notfallmedizin Schmerztherapie 2003 Aug;38(8):528‐37. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220578
- Lauretti GR, Mattos AL, Gomes JM, Pereira NL. Postoperative analgesia and antiemetic efficacy after subarachnoid neostigmine in orthopedic surgery. Regional Anesthesia 1997 Jul‐Aug;22(4):337‐42. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220580
- Lauretti GR, Mattos AL, Gomes JM, Pereira NL. Postoperative analgesia and antiemetic efficacy after intrathecal neostigmine in patients undergoing abdominal hysterectomy during spinal anesthesia. Regional Anesthesia 1997 Nov‐Dec;22(6):527‐33. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220582
- Lauretti GR, Mattos AL, Reis MP, Prado WA. Intrathecal neostigmine for postoperative analgesia after orthopedic surgery. Journal of Clinical Anesthesia 1997;9(6):473‐7. [; MEDLINE: ; AN: 1997256494] [DOI] [PubMed] [Google Scholar]; 3220584
- Lauretti GR, Hood DD, Eisenach JC, Pfeifer BL. A multi‐center study of intrathecal neostigmine for analgesia following vaginal hysterectomy. Anesthesiology 1998 Oct;89(4):913‐8. [; MEDLINE: ; ISI:000076340300015 ER] [DOI] [PubMed] [Google Scholar]; 3220586
- Lawhorn CD, Brown RE, Schmidt ML, Vollers JM, Huggins DP, Kymer PJ, et al. A comparative‐evaluation of ondansetron, droperidol, and placebo in prevention of postoperative vomiting following tonsillectomy and adenoidectomy in the pediatric‐patient. Anesthesiology 1993;79(3A):A1195. [; ISI:A1993LY10801191 ER] [Google Scholar]; 3220588
- Lawhorn CD, Brown RE, Schmitz ML, Vollers JM, Huggins DP, Kymer PJ, et al. Prevention of postoperative vomiting in pediatric outpatient strabismus surgery. Anesthesiology 1993;79(3A):A1196. [; ISI:A1993LY10801192 ER] [Google Scholar]; 3220590
- Lawhorn CD, Bower C, Brown RE Jr, Schmitz ML, Kymer PJ, Stoner J, et al. Ondansetron decreases postoperative vomiting in pediatric patients undergoing tonsillectomy and adenoidectomy. International Journal of Pediatric Otorhinolaryngology 1996;36(2):99‐108. [; MEDLINE: ; ISI:A1993LY10801192 ER] [DOI] [PubMed] [Google Scholar]; 3220592
- Lawhorn CD, Kymer PJ, Stewart FC, Stoner JM, Shirey R, Volpe P. Ondansetron dose response curve in high‐risk pediatric patients. Journal of Clinical Anesthesia 1997 Dec;9(8):637‐42. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220594
- Lawrence CJ, Lange S. Effects of a single pre‐operative dexmedetomidine dose on isoflurane requirements and peri‐operative haemodynamic stability. Anaesthesia 1997;52(8):736‐44. [; MEDLINE: ; AN: 1997250792] [DOI] [PubMed] [Google Scholar]; 3220596
- Layfield DJ, Walker AK. Premedication for children with oral trimeprazine and droperidol. Anaesthesia 1984 Jan;39(1):32‐4. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220598
- Lazar RH, Alperin KM, Hughes GB. Efficacy of transdermal scopolamine as an antinauseant for stapes operation. Surgical Forum 1984;35:558‐61. [; AN: 1985193878] [Google Scholar]; 3220600
- Lee Y, Lin PC, Lai HY, Huang SJ, Lin YS, Cheng CR. Prevention of PONV with dexamethasone in female patients undergoing desflurane anesthesia for thyroidectomy. Acta Anaesthesiologica Sinica 2001 Dec;39(4):151‐6. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220602
- Lee SY, Lee JY, Park SY, Kim JH, Cho OG, Kim JS, et al. Prophylactic antiemetic efficacy of granisetron or ramosetron in patients undergoing thyroidectomy. Asian Journal of Surgery 2002 Oct;25(4):309‐14. [; MEDLINE: ; CN‐00412056] [DOI] [PubMed] [Google Scholar]; 3220604
- Lee Y, Lin YS, Chen YH. The effect of dexamethasone upon patient‐controlled analgesia‐related nausea and vomiting. Anaesthesia 2002 Jul;57(7):705‐9. [; MEDLINE: ; CN‐00390140] [DOI] [PubMed] [Google Scholar]; 3220606
- Lee Y, Lai HY, Lin PC, Huang SJ, Lin YS. Dexamethasone prevents postoperative nausea and vomiting more effectively in women with motion sickness. Canadian Journal of Anaesthesia 2003 Mar;50(3):232‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220608
- Leeser J, Lip H. Prevention of postoperative nausea and vomiting using ondansetron, a new, selective, 5‐HT3 receptor antagonist. Anesthesia & Analgesia 1991 Jun;72(6):751‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220610
- Lekprasert V, Meesangnil S, Pausawasdi S, Pongravee V. Efficacy of prophylactic ondansetron in Thai patients undergoing gastrointestinal tract surgery. Journal of the Medical Association of Thailand 1996 Jun;79(6):382‐7. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220612
- Lepouse C, Meriau G, Cousson J, Leon A. Evaluation of ondansetron on ondansetron plus dexamethasone for routine prophylaxis of postoperative nausea and vomiting (PONV) in cervicothoracic surgery. Anesthesiology 1999 Sep;91(3):1204. [; ISI:000082480601202 ER] [Google Scholar]; 3220614
- Roy I, Mortelmans B, Vandeput D, Deloof T, Vandenbroucke G. Prophylactic anti‐emetic therapy for PCA with morphine: A double‐blind placebo‐controlled comparison of two doses of Ondansetron. Acta Anaesthesiologica Belgica 1995;46(2):105‐6. [; AN: 1996051087] [Google Scholar]; 3220616
- Lessard MR, Trepanier CA, Rouillard JF. Neostigmine requirements for reversal of neuromuscular blockade following an infusion of mivacurium. Canadian Journal of Anaesthesia 1997 Aug;44(8):836‐42. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220618
- Lewis IH, Campbell DN, Barrowcliffe MP. Effect of nabilone on nausea and vomiting after total abdominal hysterectomy. British Journal of Anaesthesia 1994 Aug;73(2):244‐6. [; MEDLINE: ; EMBASE 1994248325] [DOI] [PubMed] [Google Scholar]; 3220620
- Liberman MA, Howe S, Lane M. Ondansetron versus placebo for prophylaxis of nausea and vomiting in patients undergoing ambulatory laparoscopic cholecystectomy. American Journal of Surgery 2000 Jan;179(1):60‐2. [; MEDLINE: ; ISI:000085562400021 ER] [DOI] [PubMed] [Google Scholar]; 3220622
- Lim KS, Lim BL, Tee CS, Vengadasalam D. Nausea and vomiting after termination of pregnancy as day surgery cases: comparison of 3 different doses of droperidol and metoclopramide as anti‐emetic prophylaxis. Singapore Medical Journal 1991 Oct;32(5):342‐3. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220624
- Lim BS, Pavy TJ, Lumsden G. The antiemetic and dysphoric effects of droperidol in the day surgery patient. Anaesthesia & Intensive Care 1999 Aug;27(4):371‐4. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220626
- Lin DM, Furst SR, Rodarte A. A double‐blinded comparison of metoclopramide and droperidol for prevention of emesis following strabismus surgery. Anesthesiology 1992 Mar;76(3):357‐61. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220628
- Lind B, Breivik H. Metoclopramide and perphenazine in the prevention of postoperative nausea and vomiting. British Journal of Anaesthesia 1970 Jul;42(7):614‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220630
- Litman RS, Wu CL, Catanzaro FA. Ondansetron decreases emesis after tonsillectomy in children. Anesthesia and Analgesia 1994;78(3):478‐81. [; MEDLINE: ; AN: 1994086863] [DOI] [PubMed] [Google Scholar]; 3220632
- Litman RS, Wu CL, Lee A, Griswold JD, Voisine R, Marshall C. Prevention of emesis after strabismus repair in children: a prospective, double‐blinded, randomized comparison of droperidol versus ondansetron. Journal of Clinical Anesthesia 1995 Feb;7(1):58‐62. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220634
- Liu K, Hsu CC, Chia YY. The effect of dose of dexamethasone for antiemesis after major gynecological surgery. Anesthesia & Analgesia 1999 Nov;89(5):1316‐8. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220636
- Liu YH, Li MJ, Wang PC, Ho ST, Chang CF, Ho CM, et al. Use of dexamethasone on the prophylaxis of nausea and vomiting after tympanomastoid surgery. Laryngoscope 2001;111(7):1271‐4. [; MEDLINE: ; AN: 2001243373] [DOI] [PubMed] [Google Scholar]; 3220638
- Loach A, Fisher A. Lorazepam as a premedicant for day‐case surgery: an assessment. Anaesthesia 1975 Jul;30(4):545‐9. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220640
- Loewen P, Lamb S, Clugston P. Randomized, double‐blind trial of dolasetron versus droperidol for prophylaxis of postoperative nausea and vomiting in patients undergoing TRAM flap breast reconstruction surgery. Annals of Plastic Surgery 2003 Nov;51(5):472‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220642
- Loo CC, Thomas E, Tan HM, Sia TH. A comparison of antiemetic efficacy of droperidol alone and in combination with metoclopramide in day surgery anaesthesia. Medical Journal of Malaysia 1997 Sep;52(3):264‐8. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220644
- Loper KA, Ready LB, Dorman BH. Prophylactic transdermal scopolamine patches reduce nausea in postoperative patients receiving epidural morphine. Anesthesia and Analgesia 1989;68(2):144‐6. [; MEDLINE: ; AN: 1989056358] [DOI] [PubMed] [Google Scholar]; 3220646
- López Herrera G, Sólís Soriano. Ondansetron versus tropisetron as antiemetic treatment in laparoscopic surgery [Valoración del ondansetrón vs tropisetrón en cirugía laparoscópica]. Revista Mexicana de Anestesiologia 1998 Jun;21(2):103‐8. [; LILACS Id: 248374] [Google Scholar]; 3220648
- Lopez‐Olaondo L, Carrascosa F, Pueyo FJ, Monedero P, Busto N, Saez A. Combination of ondansetron and dexamethasone in the prophylaxis of postoperative nausea and vomiting. British Journal of Anaesthesia 1996 Jun;76(6):835‐40. [; MEDLINE: ; EMBASE 1996180481] [DOI] [PubMed] [Google Scholar]; 3220650
- Lovstad RZ, Thagaard KS, Berner NS, Raeder JC. Neostigmine 50 < mu >g kg‐1 with glycopyrrolate increases postoperative nausea in women after laparoscopic gynaecological surgery. Acta Anaesthesiologica Scandinavica 2001;45(4):495‐500. [; MEDLINE: ; AN: 2001113787] [DOI] [PubMed] [Google Scholar]; 3220652
- Lunn DV, Lauder GR, Williams AR, Pickering RM, McQuillian PJ. Low‐dose droperidol reduces postoperative vomiting in paediatric day surgery. British Journal of Anaesthesia 1995 May;74(5):509‐11. [; MEDLINE: ; EMBASE 1995145293] [DOI] [PubMed] [Google Scholar]; 3220654
- Madan R, Perumal T, Subramaniam K, Shende D, Sadashivam S, Garg S. Effect of timing of ondansetron administration on incidence of postoperative vomiting in paediatric strabismus surgery. Anaesthesia & Intensive Care 2000 Feb;28(1):27‐30. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220656
- Madej TH, Simpson KH. Comparison of the use of domperidone, droperidol and metoclopramide in the prevention of nausea and vomiting following major gynaecological surgery. British Journal of Anaesthesia 1986 Aug;58(8):884‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220658
- Madej TH, Simpson KH. Comparison of the use of domperidone, droperidol and metoclopramide in the prevention of nausea and vomiting following gynaecological surgery in day cases. British Journal of Anaesthesia 1986 Aug;58(8):879‐83. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220660
- Madenoglu H, Yildiz K, Dogru K, Kurtsoy A, Guler G, Boyaci A. Randomized, double‐blinded comparison of tropisetron and placebo for prevention of postoperative nausea and vomiting after supratentorial craniotomy. Journal of Neurosurgical Anesthesiology 2003;15(2):82‐6. [; MEDLINE: ; CN‐00431256] [DOI] [PubMed] [Google Scholar]; 3220662
- Malins AF, Field JM, Nesling PM, Cooper GM. Nausea and vomiting after gynaecological laparoscopy: comparison of premedication with oral ondansetron, metoclopramide and placebo. British Journal of Anaesthesia 1994 Feb;72(2):231‐3. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220664
- Maltepe F, Gunerli A, Sagiroglu E, Kuvaki B, Mavioglu O. Comparison of the postoperative anti‐emetic effects of DHBP, meclopramide and ondansetron in pediatric anaesthesia. Turk Anesteziyoloji Ve Reanimasyon 1996;24(4):180‐3. [; EMBASE 1996205680] [Google Scholar]; 3220666
- Manani G, Cantele P, Dona B, Meroni M, Tiberio I, Ceschin C, et al. Oculo‐emetic and oculo‐cardiac reflex in the prevention of postoperative nausea and vomitus by diehydrobenzperidol and ondansetron after squint surgery. Acta Anaesthesiologica Italica 1996;47(3):201‐9. [; AN: 1997107862] [Google Scholar]; 3220668
- Mansfield M, Russell D, Kenny GN, Kinsella J. An investigation of the effect of ondansetron on time to induction of anaesthesia with thiopentone and propofol. European Journal of Anaesthesiology 1997 Jan;14(1):24‐8. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220670
- Marcus JR, Few JW, Chao JD, Fine NA, Mustoe TA. The prevention of emesis in plastic surgery: A randomized, prospective study. Plastic and Reconstructive Surgery 2002;109(7):2487‐94. [; MEDLINE: ; AN: 2002189579] [DOI] [PubMed] [Google Scholar]; 3220672
- Martin J, Williams D, Weis FR Jr. Comparison of three anesthetic techniques on emetic symptoms using sufentanil for outpatient surgery. AANA Journal 1987 Jun;55(3):245‐9. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220674
- Martínez Soberanis MI, Martínez Rodríguez JI. Sedation in pediatric dentistry. A comparison of intranasal midazolam and oral diazepam [Sedación en odontopediatría. Comparación del midazolam vía intranasal, con el diazepam vía oral]. Revista ADM: organo oficial de la Asociacion Dental Mexicana 1995 Oct;52(5):261‐5. [; Id: 166223] [Google Scholar]; 3220676
- Martins RS, Martins ALC, Grillo F, Bortolozzo CR. Prevention of postoperative nausea and vomiting with ondansetron: Comparison with prometazine [Prevencao de nauseas e vomitos no pos‐operatorio com ondansetron: comparacao com prometazina]. Revista Brasileira de Anestesiologia 1995;45(4):253‐8. [; AN: 1995264105] [Google Scholar]; 3220678
- Mathia WJ, Bell SK, Leak WD. Effects of preoperative metoclopramide and droperidol on postoperative nausea and vomiting in ambulatory surgery patients. AANA‐Journal 1988 Aug;56(4):325‐33. [; AN: 1988083838] [PubMed] [Google Scholar]; 3220680
- Mattila MA, Rajpar M, Pystynen P. Diazepam as an adjunct in propanidid anaesthesia for abortion. British Journal of Anaesthesia 1974 Jun;46(6):446‐8. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220682
- Mattila MA, Larni HM, Nummi SE, Pekkola PO. Effect of diazepam on emergence from ketamine anaesthesia. A double‐blind study. Anaesthesist 1979 Jan;28(1):20‐3. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220684
- Mattila MA, Saila K, Kokko T, Karkkainen T. Comparison of diazepam and flunitrazepam as adjuncts to general anaesthesia in preventing arousal following surgical stimuli. British Journal of Anaesthesia 1979 Apr;51(4):329‐37. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220686
- Mattila MAK, Hynynen KH, Eronen R, et al. Diazepam dosage and timing in ketamine combination anaesthesia. A double‐blind study. Anaesthesist 1981;30(10):500‐3. [; EMBASE 1981244263] [PubMed] [Google Scholar]; 3220688
- Mattila MA, Larni HM. The effect of diazepam on methohexitone short anaesthesia: a clinical double‐blind investigation. Current Medical Research and Opinion 1981;7(3):171‐8. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220690
- Mayson KV, Gofton EA, Chambers KG. Premedication with low dose oral clonidine does not enhance postoperative analgesia of intrathecal morphine. Canadian Journal of Anaesthesia 2000 Aug;47(8):752‐7. [; MEDLINE: ; ISI:000089776700006 ER] [DOI] [PubMed] [Google Scholar]; 3220692
- MayznerZawadzka E, Moraczewski W, Pankowska S. Ondansetron effectiveness in preventing nd therapy of postoperative nausea and vomiting after strabismus surgery. British Journal of Anaesthesia 1996 Jun;76(2):A297. [; ISI:A1996UR21500296 ER] [Google Scholar]; 3220694
- McAllister J, Abukhudair H, Hirshberg GE, Brown BA, Pence HC. Granisetron prevents postoperative vomiting after strabismus surgery. Anesthesiology 1996 SEP;85:A1077. [; ISI:A1996VM46601077 ER] [Google Scholar]; 3220696
- McCall JE, Stubbs K, Saylors S, Pohlman S, Ivers B, Smith S, et al. The search for cost‐effective prevention of postoperative nausea and vomiting in the child undergoing reconstructive burn surgery: ondansetron versus dimenhydrinate. Journal of Burn Care & Rehabilitation 1999 Jul‐Aug;20(4):309‐15. [; MEDLINE: ; SI: J14885000] [DOI] [PubMed] [Google Scholar]; 3220698
- McCartney CJL, Brill S, Rawson R, Sanandaji K, Iagounova A, Chan VWS. No anesthetic or analgesic benefit of neostigmine 1mg added to intravenous regional anesthesia with lidocaine 0.5% for hand surgery. Regional Anesthesia and Pain Medicine 2003 Sep;28(5):414‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220700
- McKenzie R, Wadhwa RK, Uy NT, Phitayakorn P, Tantisira B, Sinchioco C, et al. Antiemetic effectiveness of intramuscular hydroxyzine compared with intramuscular droperidol. Anesthesia & Analgesia 1982;60(11):783‐8. [; MEDLINE: ; CN‐00369344] [PubMed] [Google Scholar]; 3220702
- McKenzie R, Sharifi‐Azad S, Dershwitz M, Miguel R, Joslyn AF, Tantisira B, et al. A randomized, double‐blind pilot study examining the use of intravenous ondansetron in the prevention of postoperative nausea and vomiting in female inpatients. Journal of Clinical Anesthesia 1993 Jan‐Feb;5(1):30‐6. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220704
- McKenzie R, Tantisira B, Karambelkar DJ, Riley TJ, Abdelhady H. Comparison of ondansetron with ondansetron plus dexamethasone in the prevention of postoperative nausea and vomiting. Anesthesia & Analgesia 1994;79(5):961‐4. [; MEDLINE: ; AN: 1994329362] [DOI] [PubMed] [Google Scholar]; 3220706
- McKenzie R, Tantisira B, Jackson D, Bach T, Riley T. Antiemetic efficacy of a droperidol‐morphine combination in patient‐controlled analgesia. Journal of Clinical Anesthesia 1995 Mar;7(2):141‐7. [; MEDLINE: ; EMBASE 1995113573] [DOI] [PubMed] [Google Scholar]; 3220708
- McKenzie R, Lim NT, Uy NTL, Riley TJ, Hamilton DL. Droperidol/ondansetron combination controls nausea and vomiting after tubal banding. Anesthesia & Analgesia 1996 Ded;83(6):1218‐22. [; MEDLINE: ; ISI:A1996VV54300015 ER] [DOI] [PubMed] [Google Scholar]; 3220710
- McKenzie R, Riley TJ, Tantisira B, Hamilton DL. Effect of propofol for induction and ondansetron with or without dexamethasone for the prevention of nausea and vomiting after major gynecologic surgery. Journal of Clinical Anesthesia 1997 Feb;9(1):15‐20. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220712
- Mecklem DW. Propofol injection pain: comparing the addition of lignocaine or metoclopramide. Anaesthesia & Intensive Care 1994 Oct;22(5):568‐70. [; MEDLINE: ; EMBASE 1994327183] [DOI] [PubMed] [Google Scholar]; 3220714
- Memis D, Turan A, Karamanlioglu B, Kaya G, Süt N, Pamukçu Z. Caudal neostigmine for postoperative analgesia in paediatric surgery. Paediatric Anaesthesia 2003;13(4):324‐8. [; MEDLINE: ; 2003218822 20030612 ISSN: 1155‐5645] [DOI] [PubMed] [Google Scholar]; 3220716
- Migliavacca S, Speranza R, Cipolla M, Laveneziana D. [Clebopride in premedication in ambulatory interventions in general anesthesia]. [Italian]. Minerva Anestesiologica 1992 Mar;58(3):117‐20. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220718
- Mikawa K, Nishina K, Maekawa N, Asano M, Obara H. Oral clonidine premedication reduces vomiting in children after strabismus surgery. Canadian Journal of Anaesthesia 1995 Nov;42(11):977‐81. [; MEDLINE: ; EMBASE 1995342978] [DOI] [PubMed] [Google Scholar]; 3220720
- Mikawa K, Takao Y, Nishina K, Maekawa N, Obara H. The antiemetic efficacy of prophylactic granisetron in gynecologic surgery. Anesthesia and Analgesia 1995;80(5):970‐4. [; MEDLINE: ; AN: 1995141015] [DOI] [PubMed] [Google Scholar]; 3220722
- Mikawa K, Nishina K, Maekawa N, Shiga M, Obara H. Dose‐response of flurbiprofen on postoperative pain and emesis after paediatric strabismus surgery. Canadian Journal of Anaesthesia 1997 Jan;44(1):95‐8. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220724
- Mikawa K, Takao Y, Nishina K, Shiga M, Maekawa N, Obara H. Optimal dose of granisetron for prophylaxis against postoperative emesis after gynecological surgery. Anesthesia and Analgesia 1997;85(3):652‐6. [; MEDLINE: ; AN: 1997273805] [DOI] [PubMed] [Google Scholar]; 3220726
- Miles PJ, Ball DJ, Melchoir WR, Dosch MP, Hurt PD. Is ondansetron as effective as droperidol in prevention of postoperative nausea and vomiting?. Journal of the American Association of Nurse Anesthetists 1996;64(5):445. [; AN: 1996345129] [Google Scholar]; 3220728
- Millar JM, Hall PJ. Nausea and vomiting after prostaglandins in day case termination of pregnancy. The efficacy of low dose droperidol. Anaesthesia 1987 Jun;42(6):613‐8. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220730
- Miller CD, Anderson WG. Silent regurgitation in day case gynaecological patients. Anaesthesia 1988;43(4):321‐3. [; MEDLINE: ; AN: 1988125863] [DOI] [PubMed] [Google Scholar]; 3220732
- Millo J, Siddons M, Innes R, Laurie PS. Randomised double‐blind comparison of ondansetron and droperidol to prevent postoperative nausea and vomiting associated with patient‐controlled analgesia. Anaesthesia 2001 Jan;56(1):60‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220734
- Mjahed K, Harrar N, Idali B, Laouissi F, Benaguida M, Amraoui A. [Does oral ondansetron reduce the incidence of nausea and vomiting after surgery for strabismus in children?] [L'ondansetron oral reduit‐il l'incidence des nausees et vomissements apres chirurgie du strabisme chez l'enfant?]. Francaises d'anesthesie et de reanimation 1996;15(7):1018‐21. [; MEDLINE: ; EMBASE 1996352584; CN‐00217896] [DOI] [PubMed] [Google Scholar]; 3220736
- Moens P, Levarlet M, Hendrickx P, Guchteneere E. Single IV bolus dose of ondansetron in the prevention of postoperative nausea and emesis. Acta Anaesthesiologica Belgica 1997;48(4):245‐50. [; MEDLINE: ; AN: 1998083934] [PubMed] [Google Scholar]; 3220738
- Moerman I, Franck P, Camu F. Evaluation of methylnaltrexone for the reduction of postoperative vomiting and nausea incidences. Acta Anaesthesiologica Belgica 1995;46(3‐4):127‐32. [; MEDLINE: ; EMBASE 1996138759] [PubMed] [Google Scholar]; 3220740
- Mogensen F, Muller D, Valentin N. Glycopyrrolate during ketamine/diazepam anaesthesia. A double‐blind comparison with atropine. Acta Anaesthesiologica Scandinavica 1986 May;30(4):332‐6. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220742
- Monagle J, Barnes R, Goodchild C, Hewitt M. Ondansetron is not superior to moderate dose metoclopramide in the prevention of post‐operative nausea and vomiting after minor gynaecological surgery. European Journal of Anaesthesiology 1997 Nov;14(6):604‐9. [; MEDLINE: ; EMBASE 1998012589] [DOI] [PubMed] [Google Scholar]; 3220744
- Morin AM, Bezler T, Eberhart LH, Mayer R, Schreiber MN, Kilian J, et al. The effect of low droperidol dosages on postoperative anxiety, internal tension, general mood and PONV. Anaesthesist 1999;48(1):19‐25. [; MEDLINE: ; CN‐00214778] [DOI] [PubMed] [Google Scholar]; 3220746
- Morris RW, Aune H, Feiss P, Hanson A, Hasselstrom L, Maltby JR, et al. International, multicentre, placebo‐controlled study to evaluate the effectiveness of ondansetron vs. metoclopramide in the prevention of post‐operative nausea and vomiting. European Journal of Anaesthesiology 1998 Jan;15(1):69‐79. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220748
- Mortensen PT. Droperidol (dehydrobenzperidol(Reg.trademark)): Postoperative anti‐emetic effect when given intravenously to gynaecological patients. Acta Anaesthesiologica Scandinavia 1982;26(1):48‐52. [; MEDLINE: ; EMBASE 1982085185] [DOI] [PubMed] [Google Scholar]; 3220750
- Morton NS, Camu F, Dorman T, Knudsen KE, Kvalsvik O, Nellgard P, et al. Ondansetron reduces nausea and vomiting after paediatric adenotonsillectomy. Paediatric Anaesthesia 1997;7(1):37‐45. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220752
- Moscovici R, Prego G, Schwartz M, Steinfeld O. Epidural scopolamine administration in preventing nausea after epidural morphine. Journal of Clinical Anesthesia 1995 Sep;7(6):474‐6. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220754
- Movinsky BA, Gibbs RD, Pellegrini JE. The analgesia efficacy of metoclopramide on postoperative laparoscopic tubal ligation patients. AANA 1999;67:521(A32). [] [PubMed] [Google Scholar]; 3220756
- Muñoz Cuevas HJ, Palestino Luna S, Flores Sánchez VO, Amancio Chassin O. Use of clonidine and midazolam as premedication in ppthalmological surgery {sic} [Medicación preanéstesica en cirugía oftalmólogica: uso de clonidina y midazolam]. Revista mexicana anestesiologia 1992 July;15(3):131‐4. [; Localization: MX1.1] [Google Scholar]; 3220758
- Munro HM, D'Errico CC, Lauder GR, Wagner DS, Voepel‐Lewis T, Tait AR. Oral granisetron for strabismus surgery in children. Canadian Journal of Anaesthesia 1999 Jan;46(1):45‐8. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220760
- Munro FJ, Fisher S, Dickson U, Morton N. The addition of antiemetics to the morphine solution in patient controlled analgesia syringes used by children after an appendicectomy does not reduce the incidence of postoperative nausea and vomiting. Paediatric Anaesthesia 2002 Sep;12(7):600‐3. [; MEDLINE: ; CN‐00410336] [DOI] [PubMed] [Google Scholar]; 3220762
- Naguib M, Bakry AK, Khoshim MH, Channa AB, Gammal M, Gammal K, et al. Prophylactic antiemetic therapy with ondansetron, tropisetron, granisetron and metoclopramide in patients undergoing laparoscopic cholecystectomy: a randomized, double‐blind comparison with placebo. Canadian Journal of Anaesthesia 1996 Mar;43(3):226‐31. [; MEDLINE: ; AN: 1996132625] [DOI] [PubMed] [Google Scholar]; 3220764
- Naguib K, Osman HA, Al‐Khayat HC, Zikri AM. Prevention of post‐operative nausea and vomiting following laparoscopic surgery‐‐ephedrine vs propofol. Middle East Journal of Anesthesiology 1998 Feb;14(4):219‐30. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220766
- Najeeb R, Naqash I, Shah ZA, Habib M, Kant S. A comparative study of two antiemetics: Droperidol and Granisetron in the prevention of post anaesthesia nausea and vomiting. JK‐Practitioner 2000;7(1):52‐4. [; AN: 2000074626] [Google Scholar]; 3220768
- Najnigier B, Patkowski W, Zieniewicz K, Nyckowski P, Fraczek M, Kacka A, et al. Zofran (ondansetron) in preventing postoperative nausea and vomiting after laparoscopic cholecystectomy [Zofran w zapobieganiu nudnosciom i wymiotom po cholecystektomii laparoskopowej]. Acta Endoscopica Polona 1997;7(3‐4):125‐8. [; AN: 1998211599] [Google Scholar]; 3220770
- Nakata K, Mammoto T, Kita T, Taniguchi H, Kanbara N, Akamatsu T, et al. Continuous epidural, not intravenous, droperidol inhibits pruritus, nausea, and vomiting during epidural morphine analgesia. Journal of Clinical Anesthesia 2002;14(2):121‐5. [; MEDLINE: ; AN: 2002127564] [DOI] [PubMed] [Google Scholar]; 3220772
- Nawasreh O, Fraihat A, Maaita J. The effect of preoperative intravenous dexamethasone in pediatric adenotonsillectomy. Journal of the Bahrain Medical Society 2000;12(3):130‐3. [; AN: 2000203607] [Google Scholar]; 3220774
- Nelskyla K, Yli‐Hankala A, Soikkeli A, Korttila K. Neostigmine with glycopyrrolate does not increase the incidence or severity of postoperative nausea and vomiting in outpatients undergoing gynaecological laparoscopy. British Journal of Anaesthesia 1998 Nov;81(5):757‐60. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220776
- Ng KF, Tsui SL, Yang JC, Ho ET. Comparison of tramadol and tramadol/droperidol mixture for patient‐controlled analgesia. Canadian Journal of Anaesthesia 1997 Aug;44(8):810‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220778
- Nicolson SC, Kaya KM, Betts EK. The effect of preoperative oral droperidol on the incidence of postoperative emesis after paediatric strabismus surgery. Canadian Journal of Anaesthesia 1988 Jul;35(4):364‐7. [; MEDLINE: ; EMBASE 1988174489] [DOI] [PubMed] [Google Scholar]; 3220780
- Nortcliffe SA, Shah J, Buggy DJ. Prevention of postoperative nausea and vomiting after spinal morphine for Caesarean section: comparison of cyclizine, dexamethasone and placebo. British Journal of Anaesthesia 2003 May;90(5):665‐70. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220782
- O'Brien CM, Titley G, Whitehurst P. A comparison of cyclizine, ondansetron and placebo as prophylaxis against postoperative nausea and vomiting in children. Anaesthesia 2003 Jul;58(7):707‐11. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220784
- O'Donovan N, Shaw J. Nausea and vomiting in day‐case dental anaesthesia. The use of low‐dose droperidol. Anaesthesia 1984 Dec;39(12):1172‐6. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220786
- Oddby‐Muhrbeck E, Eksborg S, Bergendahl HT, Muhrbeck O, Lonnqvist PA. Effects of clonidine on postoperative nausea and vomiting in breast cancer surgery. Anesthesiology 2002 May;96(5):1109‐14. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220788
- Olutoye O, Jantzen EC, Alexis R, Rajchert D, Schreiner MS, Watcha MF. A comparison of the costs and efficacy of ondansetron and dolasetron in the prophylaxis of postoperative vomiting in pediatric patients undergoing ambulatory surgery. Anesthesia & Analgesia 2003 Aug;97(2):390‐6. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220790
- Omais M, Lauretti GR, Paccola CA. Epidural morphine and neostigmine for postoperative analgesia after orthopedic surgery. Anesthesia & Analgesia 2002 Dec;95(6):1698‐701. [; MEDLINE: ; CN‐00411824] [DOI] [PubMed] [Google Scholar]; 3220792
- Oshima T, Kasuya Y, Okumura Y, Terazawa E, Dohi S. Prevention of nausea and vomiting with tandospirone in adults after tympanoplasty. Anesthesia & Analgesia 2002 Nov;95(5):1442‐5. [; MEDLINE: ; CN‐00410888] [DOI] [PubMed] [Google Scholar]; 3220794
- Ostman PL, Faure E, Glosten B, Kemen M, Robert MK, Bedwell S. Is the antiemetic effect of the emulsion formulation of propofol due to the lipid emulsion?. Anesthesia and Analgesia 1990;71(5):536‐40. [; MEDLINE: ; AN: 1990389934] [PubMed] [Google Scholar]; 3220796
- Ozalp G, Kuru N, Guner F, Kadiogullari N. Prophylactic antiemetic therapy with patient‐controlled analgesia: Ondansetron and tropisetron [Hasta kontrollu analjezide profilaktik antiemetik kullanimi: ondansetron ve tropisetron]. Turk Anesteziyoloji Ve Reanimasyon 1997;25(4):173‐6. [; MEDLINE: ; EMBASE 1997190014] [Google Scholar]; 3220798
- Ozcan AA, Gunes Y, Haciyakupoglu G. Using diazepam and atropine before strabismus surgery to prevent postoperative nausea and vomiting: a randomized, controlled study. Journal of AAPOS: the official publication of the American Association for Pediatric Ophthalmology and Strabismus 2003 Jun;7(3):210‐2. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220800
- Ozmen S, Yavuz L, Ceylan BG, Tarhan O, Aydin C. Comparison of granisetron with granisetron plus droperidol combination prophylaxis in post‐operative nausea and vomiting after laparoscopic cholecystectomy. Journal of International Medical Research 2002 Sep‐Oct;30(5):520‐4. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220802
- Oztekin S, Ozzeybek D, Tasdogen A, Kilercik H, Kara HC. Comparison of the antiemetic efficacy of tropisetron and droperidol with patient‐given tramadol. Journal of International Medical Research 2003 Jul‐Aug;31(4):267‐71. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220804
- Paech MJ, Pavy TJ, Evans SF. Single‐dose prophylaxis for postoperative nausea and vomiting after major abdominal surgery: ondansetron versus droperidol. Anaesthesia & Intensive Care 1995 Oct;23(5):548‐54. [; MEDLINE: ; EMBASE 1995302739] [DOI] [PubMed] [Google Scholar]; 3220806
- Paech MJ, Pavy TJG, Orlikowski CEP, Lim W, Evans SF. Postoperative epidural infusion: A randomized, double‐blind, dose‐finding trial of clonidine in combination with bupivacaine and fentanyl. Anesthesia & Analgesia 1997 Jun;84(6):1323‐28. [; MEDLINE: ; ISI:A1997XB41400027 ER] [DOI] [PubMed] [Google Scholar]; 3220808
- Paech MJ, Lee BHS, Evans SF. The effect of anaesthetic technique on postoperative nausea and vomiting after day‐case gynaecological laparoscopy. Anaesthesia and Intensive Care 2002;30(2):153‐9. [; MEDLINE: ; AN: 2002143002] [DOI] [PubMed] [Google Scholar]; 3220810
- Paech MJ, Rucklidge MW, Banks SL, Gurrin LC, Orlikowski CE, Pavy TJ. The efficacy and cost‐effectiveness of prophylactic 5‐hydroxytryptamine3 receptor antagonists: tropisetron, ondansetron and dolasetron. Anaesthesia and Intensive Care 2003 Feb;31(1):11‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220812
- Palme CE, Tomasevic P, Pohl DV. Evaluating the effects of oral prednisolone on recovery after tonsillectomy: A prospective, double‐blind, randomized trial. Laryngoscope 2000;110(12):2000‐4. [; MEDLINE: ; ISI:000165856600003 ER] [DOI] [PubMed] [Google Scholar]; 3220814
- Pan PM, Huang CT, Wei TT, Mok MS. Enhancement of analgesic effect of intrathecal neostigmine and clonidine on bupivacaine spinal anesthesia. Regional Anesthesia and Pain Medicine 1998 Jan‐Feb;23(1):49‐56. [; MEDLINE: ; ISI:000072032800010 ER] [DOI] [PubMed] [Google Scholar]; 3220816
- Pan PH, Moore CH. Comparing the efficacy of prophylactic metoclopramide, ondansetron, and placebo in cesarean section patients given epidural anesthesia. Journal of Clinical Anesthesia 2001 Sep;13(6):430‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220818
- Pandit SK, Kothary SP, Pandit UA, Mirakhur RK. Premedication with cimetidine and metoclopramide. Effect on the risk factors of acid aspiration. Anaesthesia 1986 May;41(5):486‐92. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220820
- Pang WW, Wu HS, Lin CH, Chang DP, Huang MH. Metoclopramide decreases emesis but increases sedation in tramadol patient‐controlled analgesia. Canadian Journal of Anaesthesia 2002 Dec;49(10):1029‐33. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220822
- Papadimitriou L, Livanios S, Katsaros G, Hassiakos D, Koussi T, Demesticha T. Prevention of postoperative nausea and vomiting after laparoscopic gynaecological surgery. Combined antiemetic treatment with tropisetron and metoclopramide vs. metoclopramide alone. European Journal of Anaesthesiology 2001 Sep;18(9):615‐9. [; MEDLINE: ; AN: 2002203265] [DOI] [PubMed] [Google Scholar]; 3220824
- Pappas ALS, Sukhani R, Hotaling AJ, Mikat Stevens M, Javorski JJ, Donzelli J, et al. The effect of preoperative dexamethasone on the immediate and delayed postoperative morbidity in children undergoing adenotonsillectomy. Anesthesia & Analgesia 1998;87(1):57‐61. [; MEDLINE: ; AN: 1998227444] [DOI] [PubMed] [Google Scholar]; 3220826
- Park J, Forrest J, Kolesar R, Bhola D, Beattie S, Chu C. Oral clonidine reduces postoperative PCA morphine requirements. Canadian Journal of Anaesthesia 1996 Sep;43(9):900‐6. [; MEDLINE: ; CN‐00344892] [DOI] [PubMed] [Google Scholar]; 3220828
- Parks L, McNamee DA, Convery PN, Matthews E, Bell PF. A double‐blind placebo‐controlled study of rectal ondansetron for prophylactic antiemesis following total hip arthroplasty in the elderly. Anesthesiology 1999 Sep;91(3A):A50. [] [Google Scholar]; 3220830
- Parnis SJ, Foate JA, Walt JH, Short T, Crowe CE. Oral midazolam is an effective premedication for children having day‐stay anaesthesia. Anaesthesia & Intensive Care 1992 Feb;20(1):9‐14. [; MEDLINE: ; CN‐00084986] [DOI] [PubMed] [Google Scholar]; 3220832
- Pascucci G, Palmieri V, Miranda G, Buccarella A. Tropisetron versus ondansetron in the prevention and control of postoperative nausea and vomiting [Prevenzione e controllo dell'emesi post‐operatoria: confronto ondansetron‐tropisetron]. Acta Anaesthesiologica Italica 1996;47(3):173‐7. [; AN: 1997107861] [Google Scholar]; 3220834
- Patel RI, Davis PJ, Orr RJ, Ferrari LR, Rimar S, Hannallah RS, Cohen IT, Colingo K, Donlon JV, Haberkern CM, McGowan FX, Prillaman BA, Parasuraman TV, Creed MR. Single‐dose ondansetron prevents postoperative vomiting in pediatric outpatients. Anesthesia & Analgesia 1997 Sep;85(3):538‐45. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220836
- Patterson KW, Armstrong C, Timon D, O'Toole‐D, Keane PW. The value of buccal prochlorperazine in the prevention of post‐operative nausea and vomiting. Journal of the Irish Colleges of Physicians and Surgeons 1991;20(3):200‐2. [; MEDLINE: ; AN: 1991233463] [Google Scholar]; 3220838
- Patterson KW, Armstrong C, Timon D, O'Toole D, Keane PW. Buccal prochlorperazine as an antiemetic for day care surgery. Irish Medical Journal 1993 Nov‐Dec;86(6):186‐8. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220840
- Paul AK. Metoclopramide in prevention of postanaesthetic vomiting. Journal of the Indian Medical Association 1985;83(8):271‐3. [; MEDLINE: ; AN: 1986070877] [PubMed] [Google Scholar]; 3220842
- Paventi S, Santevecchi A, Ranieri R. Efficacy of a single‐dose ondansetron for preventing post‐operative nausea and vomiting after laparoscopic cholecystectomy with sevoflurane and remifentanil infusion anaesthesia. European review for medical and pharmacological sciences 2001 Mar‐Apr;5(2):59‐63. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220844
- Paxton LD, McKay AC, Mirakhur RK. Prevention of nausea and vomiting after day case gynaecological laparoscopy. A comparison of ondansetron, droperidol, metoclopramide and placebo. Anaesthesia 1995 May;50(5):403‐6. [; MEDLINE: ; EMBASE 1995141141] [DOI] [PubMed] [Google Scholar]; 3220846
- Paxton D, Taylor RH, Gallagher TM, Crean PM. Postoperative emesis following otoplasty in children. Anaesthesia 1995 Dec;50(12):1083‐5. [; MEDLINE: ; EMBASE 1996004135] [DOI] [PubMed] [Google Scholar]; 3220848
- Pearman MH. Single dose intravenous ondansetron in the prevention of postoperative nausea and vomiting. Anaesthesia 1994 Jan;49(Suppl):11‐5. [; MEDLINE: ; EMBASE 1994058324] [DOI] [PubMed] [Google Scholar]; 3220850
- Peixoto AJ, Peixoto Filho AJ, Leaes LF, Celich MF, Barros MA. Efficacy of prophylactic droperidol, ondansetron or both in the prevention of postoperative nausea and vomiting in major gynaecological surgery. A prospective, randomized, double‐blind clinical trial. European Journal of Anaesthesiology 2000 Oct;17(10):611‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220852
- Pendeville PE, Veyckemans F, Boven MJ, Steinier JR. Open placebo controlled comparison of the antiemetic effect of droperidol, metoclopramide or a combination of both in pediatric strabismus surgery. Acta Anaesthesiologica Belgica 1993;44(1):3‐10. [; MEDLINE: ; EMBASE 1994037717] [PubMed] [Google Scholar]; 3220854
- Pérez García A, Cruz Anguiano V. Ondansetron vs. metoclopramide for post operative nausea and vomiting prevention [Ondansetrón vs metoclopramida en la prevención del vómito postoperatorio]. Revista mexicana anestesiologia 2000 Oct;23(4):149‐154. [; MEDLINE: ] [Google Scholar]; 3220856
- Pertusa V, Bellver J, Marques A, Onrubia X, Vinals MP, Sanmiguel G, et al. Antiemetic prophylaxis after laparoscopic cholecystectomy: comparative study of dehydrobenzperidol, metoclopramide, ondansetron and placebo. Revista Espanola De Anestesiologia y Reanimacion 1996;43(7):239‐42. [; MEDLINE: ; EMBASE 1996313549] [PubMed] [Google Scholar]; 3220858
- Peters CG, Brunton JT. Comparative study of lorazepam and trimeprazine for oral premedication in paediatric anaesthesia. British Journal of Anaesthesia 1982 Jun;54(6):623‐8. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220860
- Philip BK, Pearman MH, Kovac AL, Chelly JE, Wetchler BV, McKenzie R, et al. Dolasetron for the prevention of postoperative nausea and vomiting following outpatient surgery with general anaesthesia: a randomized, placebo‐controlled study. The Dolasetron PONV Prevention Study Group. European Journal of Anaesthesiology 2000 Jan;17(1):23‐32. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220862
- Phillips S, Ruggier R, Hutchinson SE. Zingiber officinale (Ginger) ‐ An antiemetic for day case surgery. Anaesthesia 1993;48(8):715‐7. [; MEDLINE: ; EMBASE 1993240752] [DOI] [PubMed] [Google Scholar]; 3220864
- Piper SN, Triem JG, Maleck WH, Fent MT, Huttner I, Boldt J. Placebo‐controlled comparison of dolasetron and metoclopramide in preventing postoperative nausea and vomiting in patients undergoing hysterectomy. European Journal of Anaesthesiology 2001;18(4):251‐6. [; MEDLINE: ; AN: 2001149413] [DOI] [PubMed] [Google Scholar]; 3220866
- Piper SN, Suttner SW, Rohm KD, Maleck WH, Larbig E, Boldt J. Dolasetron, but not metoclopramide prevents nausea and vomiting in patients undergoing laparoscopic cholecystectomy. Canadian Journal of Anaesthesia 2002 Dec;49(10):1021‐8. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220868
- Piper SN, Triem JG, Rohm KD, Kranke P, Maleck WH, Boldt J. Prevention of post‐operative nausea and vomiting. Randomised comparison of dolasetron versus dolasetron plus dexamethasone [Prophylaxe von postoperativer Ubelkeit und postoperativem Erbrechen. Randomisierter Vergleich von Dolasetron versus Dolasetron mit Dexamethason]. Anaesthesist 2003 Feb;52(2):120‐6. [; MEDLINE: ; CN‐00422907] [DOI] [PubMed] [Google Scholar]; 3220870
- Pitkanen MT, Niemi L, Tuominen MK, Rosenberg PH. Effect of tropisetron, a 5‐HT3 receptor antagonist, on analgesia and nausea after intrathecal morphine. British Journal of Anaesthesia 1993 Nov;71(5):681‐4. [; MEDLINE: ; EMBASE 1993331013] [DOI] [PubMed] [Google Scholar]; 3220872
- Pitkanen MT, Numminen MK, Tuominen MK, Rosenberg PH. Comparison of metoclopramide and ondansetron for the prevention of nausea and vomiting after intrathecal morphine. European Journal of Anaesthesiology 1997 Mar;14(2):172‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220874
- Ploner F, Kainzwaldner A. [Evaluation of the administration time of ondansetron, a preventive for postoperative nausea and vomiting: prospective, randomized, double‐blind study in 120 patients]. [German] [Evaluierung der applikationszeit von ondansetron. Ein prophylaktikum fur postoperative ubelkeit und erbrechen: prospektive, randomisierte doppelblindstudie an 120 patienten]. Anaesthesist 1997 Jul;46(7):583‐7. [; MEDLINE: ; EMBASE 1997242657] [DOI] [PubMed] [Google Scholar]; 3220876
- Polati E, Finco G, Bartoloni A, Gottin L, Pinaroli AM, Zanoni L, et al. [Prevention of postoperative nausea and vomiting with ondansetron: a prospective, randomized, double‐blind study in 90 patients]. [Italian]. Minerva Anestesiologica 1995 Sep;61(9):373‐9. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220878
- Pongrojpaw D, Chiamchanya C. The efficacy of ginger in prevention of post‐operative nausea and vomiting after outpatient gynecological laparoscopy. Journal of the Medical Association of Thailand 2003 Mar;86(3):244‐50. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220880
- Ponnudurai R, Hurdley J. Bromazepam as oral premedication. A comparison with lorazepam.. Anaesthesia 1986 May;41(5):541‐3. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220882
- Prescott RJ, Espley AJ, Davie IT, Slawson KB, Ruckley CV. Double‐blind clinical trial of anaesthetic premedication for use in major day surgery. Lancet 1976 May;29(1):1148‐50. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220884
- Principi F, Di AP, Sofra M, Salerno S, Aloe L. Intravenous ondansetron in the prophylactic treatment of postoperative nausea and vomiting in gynaecological surgery. Acta Anaesthesiologica Italica 1996;47(2):147‐156. [; EMBASE 1997020652] [Google Scholar]; 3220886
- Pueyo FJ, Carrascosa F, Lopez L, Iribarren MJ, Garcia‐Pedrajas F, Saez A. Combination of ondansetron and droperidol in the prophylaxis of postoperative nausea and vomiting. Anesthesia & Analgesia 1996 Jul;83(1):117‐22. [; MEDLINE: ; EMBASE 1996203158] [DOI] [PubMed] [Google Scholar]; 3220888
- Pugh SC, Jones NC, Barsoum LZ. A comparison of prophylactic ondansetron and metoclopramide administration in patients undergoing major neurosurgical procedures. Anaesthesia 1996 Dec;51(12):1162‐4. [; MEDLINE: ; EMBASE 1997034153] [DOI] [PubMed] [Google Scholar]; 3220890
- Purhonen S, Kauko M, Koski EMJ, Nuutinen L. Comparison of tropisetron, droperidol, and saline in the prevention of postoperative nausea and vomiting after gynecologic surgery. Anesthesia and Analgesia 1997;84(3):662‐7. [; MEDLINE: ; AN: 1997071209] [DOI] [PubMed] [Google Scholar]; 3220892
- Purhonen S, Turunen M, Ruohoaho UM, Niskanen M, Hynynen M. Supplemental oxygen does not reduce the incidence of postoperative nausea and vomiting after ambulatory gynecologic laparoscopy. Anesthesia & Analgesia 2003 Jan;96(1):91‐6. [; MEDLINE: ; CN‐00412462] [DOI] [PubMed] [Google Scholar]; 3220894
- Purhonen S, Niskanen M, Wustefeld M, Mustonen P, Hynynen M. Supplemental oxygen for prevention of nausea and vomiting after breast surgery. British Journal of Anaesthesia 2003 Aug;91(2):284‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220896
- Quaynor H, Raeder JC. Incidence and severity of postoperative nausea and vomiting are similar after metoclopramide 20 mg and ondansetron 8 mg given by the end of laparoscopic cholecystectomies. Acta Anaesthesiologica Scandinavica 2002 Jan;46(1):109‐13. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220898
- Quiroga OJ, Hernández Santos JR, Tenopala S, Castillejos V, Porras R, Rivera MG, et al. Comparative study of bupivacaine‐morphine versus bupivacaine‐morphine‐clonidine peridurally administered for postoperative analgesia of patients undergoing cesareans. Revista de la Sociedad Espanola del Dolor 2003;10(3):135‐44. [; 2003220944 20030626 ISSN: 1134‐8046] [Google Scholar]; 3220900
- Rajeeva V, Bhardwaj N, Batra YK, Dhaliwal LK. Comparison of ondansetron with ondansetron and dexamethasone in prevention of PONV in diagnostic laparoscopy. Canadian Journal of Anaesthesia 1999 Jan;46(1):40‐4. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220902
- Ramirez DFJ, Mora RJ, Vazquez LM, Rodriguez LM, Martinez FMA, Morales VA. [Prevention of postoperative nausea and vomiting in gynecologic surgery with 3 fixed doses of metoclopramide, droperidol or placebo]. [Spanish]. Revista Espanola de Anestesiologia y Reanimacion 2001 Feb;48(2):65‐8. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220904
- Raphael JH, Norton AC. Antiemetic efficacy of prophylactic ondansetron in laparoscopic surgery: randomized, double‐blind comparison with metoclopramide. British Journal of Anaesthesia 1993 Dec;71(6):845‐8. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220906
- Ratra CK, Pandey K, Badola RP, Bhargava KP. Evaluation of the effect of halothane on postoperative vomiting. British Journal of Anaesthesia 1968 Apr;40(4):270‐8. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220908
- Reihner E, Grunditz R, Giesecke K, Gustafsson LL. Postoperative nausea and vomiting after breast surgery: efficacy of prophylactic ondansetron and droperidol in a randomized placebo‐controlled study. European Journal of Anaesthesiology 2000 Mar;17(3):197‐203. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220910
- Reinhart DJ, Klein KW, Schroff E. Transdermal scopolamine for the reduction of postoperative nausea in outpatient ear surgery: A double‐blind, randomized study. Anesthesia and Analgesia 1994;79(2):281‐4. [; MEDLINE: ; AN: 1994244871] [DOI] [PubMed] [Google Scholar]; 3220912
- Richardson FJ, Manford ML. Comparison of flunitrazepam and diazepam for oral premedication in older children. British Journal of Anaesthesia 1979 Apr;51(4):313‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220914
- Richardson MG, Wu CL, Hussain A. Midazolam premedication increases sedation but does not prolong discharge times after brief outpatient general anesthesia for laparoscopic tubal sterilization. Anesthesia & Analgesia 1997 Aug;85(2):301‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220916
- Riley TJ, McKenzie R, Tantisira BR, Hamilton DL. Droperidol‐ondansetron combination versus droperidol alone for postoperative control of emesis after total abdominal hysterectomy. Journal of Clinical Anesthesia 1998;10(1):6‐12. [; MEDLINE: ; AN: 1998082324] [DOI] [PubMed] [Google Scholar]; 3220918
- Rita L, Goodarzi M, Seleny F. Effect of low dose droperidol on postoperative vomiting in children. Canadian Anaesthetists' Society Journal 1981 May;28(3):259‐62. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220920
- Roberts CJ, Millar JM, Goat VA. The antiemetic effectiveness of droperidol during morphine patient‐controlled analgesia. Anaesthesia 1995 Jun;50(6):559‐62. [; MEDLINE: ; EMBASE 1995209335] [DOI] [PubMed] [Google Scholar]; 3220922
- Rodola F, Cannelli G, Barbi S, Forte E, Anastasio C, Vagnoni S. Cost effective prophylaxys of postoperative nausea and vomiting by anesthetic premedication. Rivista Europea Per Le Scienze Mediche e Farmacologiche 1995 Nov‐Dec;17(6):243‐6. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220924
- Rodrigo MRC, Campbell RCH, Chow J, Tong CKA, Hui E, Lueveswanij S. Ondansetron for prevention of postoperative nausea and vomiting following minor oral surgery: A double‐blind randomized study. Anaesthesia and Intensive Care 1994;22(5):576‐9. [; MEDLINE: ; EMBASE 1994327185] [DOI] [PubMed] [Google Scholar]; 3220926
- Rodrigo C, Campbell R, Chow J, Tong A. The effect of a 4‐mg preoperative intravenous dose of ondansetron in preventing nausea and vomiting after maxillofacial surgery. Journal of Oral & Maxillofacial Surgery 1996 Oct;54(10):1171‐5. [; MEDLINE: ; EMBASE 1997074860] [DOI] [PubMed] [Google Scholar]; 3220928
- Rohling R, Alon E, Atanassoff PG, Sailer H. Tropisetron vs metoclopramide and placebo for the prophylaxis of postoperative nausea and vomiting following maxillofacial surgery. British Journal of Anaesthesia 1995 May;74:91. [; ISI:A1995RA77700298 ER] [Google Scholar]; 3220930
- Rose JB, Martin TM, Corddry DH, Zagnoev M, Kettrick RG. Ondansetron reduces the incidence and severity of poststrabismus repair vomiting in children. Anesthesia & Analgesia 1994 Sep;79(3):486‐9. [; MEDLINE: ; EMBASE 1994277433] [DOI] [PubMed] [Google Scholar]; 3220932
- Rose JB, Martin TM. Posttonsillectomy vomiting. Ondansetron or metoclopramide during paediatric tonsillectomy: are two doses better than one?. Paediatric Anaesthesia 1996;6(1):39‐44. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220934
- Rose JB, Brenn BR, Corddry DH, Thomas PC. Preoperative oral ondansetron for pediatric tonsillectomy. Anesthesia & Analgesia 1996 Mar;82(3):558‐62. [; MEDLINE: ; EMBASE 1996073471] [DOI] [PubMed] [Google Scholar]; 3220936
- Rothenberg DM, Parnass SM, Litwack K, McCarthy RJ, Newman LM. Efficacy of ephedrine in the prevention of postoperative nausea and vomiting. Anesthesia & Analgesia 1991 Jan;72(1):58‐61. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220938
- Rothenberg DM, McCarthy RJ, Peng CC, Normoyle DA. Nausea and vomiting after dexamethasone versus droperidol following outpatient laparoscopy with a propofol‐based general anesthetic. Acta Anaesthesiologica Scandinavica 1998 Jul;42(6):637‐42. [; MEDLINE: ; EMBASE 1998216257] [DOI] [PubMed] [Google Scholar]; 3220940
- Ruiz de Adana J, Tobalina Bonis R, Garcia Galan F, Hernandez Matias A, Fernandez Luengas D, Ortega Deballon P, et al. Antiemetic efficacy of ondansetron in laparoscopic cholecystectomy. A randomized, double‐blind, placebo‐controlled study. Revista Espanola de Enfermedades Digestivas 1999 Sep;91(9):642‐4. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220942
- Rusch D, Bernhardt J, Wulf H. [Prophylaxis of nausea and vomiting after pelviscopy. Dolasetron or MCP in comparison with placebo].[German]. Anaesthesist 1999 Oct;48(10):705‐12. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220944
- Rusch D, Palm S, Sauerwald M, Romer T, Wulf H. [Prophylaxis of Postoperative Nausea and Vomiting FollowingGynaecological Laparoscopy] [Article in German]. Anasthesiol Intensivmed Notfallmed Schmerzther 2002 Jan;37(1):16‐23. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220946
- Russell D, Duncan LA, Frame WT, Higgins SP, Asbury AJ, Millar K. Patient‐controlled analgesia with morphine and droperidol following caesarean section under spinal anaesthesia. Acta Anaesthesiologica Scandinavica 1996 May;40(5):600‐5. [; MEDLINE: ; EMBASE 1996180412] [DOI] [PubMed] [Google Scholar]; 3220948
- Rust M, Cohen LA. Single oral dose ondansetron in the prevention of postoperative nausea and emesis. Anaesthesia 1994;49(SUPPL):16‐23. [; EMBASE 1994058325] [DOI] [PubMed] [Google Scholar]; 3220950
- Sadhasivam S, Saxena A, Kathirvel S, Kannan TR, Trikha A, Mohan V. The safety and efficacy of prophylactic ondansetron in patients undergoing modified radical mastectomy. Anesthesia & Analgesia 1999 Dec;89(6):1340‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220952
- Sadhasivam S, Shende D, Madan R. Prophylactic ondansetron in prevention of postoperative nausea and vomiting following pediatric strabismus surgery ‐ A dose‐ response study. Anesthesiology 2000 Apr;92(4):1035‐42. [; MEDLINE: ; ISI:000086172700017 ER] [DOI] [PubMed] [Google Scholar]; 3220954
- Sahjpaul RL, Mahon J, Wiebe S. Dexamethasone for morbidity after subdural electrode insertion‐‐a randomized controlled trial. Canadian journal of neurological sciences 2003 Nov;30(4):340‐8. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220956
- Salmenpera M, Kuoppamaki R, Salmenpera A. Do anticholinergic agents affect the occurrence of postanaesthetic nausea?. Acta Anaesthesiologica Scandinavica 1992 Jul;36(5):445‐8. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220958
- Sanansilp V, Areewatana S, Tonsukchai N. Droperidol and the side effects of epidural morphine after cesarean section. Anesthesia & Analgesia 1998 Mar;86(3):532‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220960
- Sanansilp V, Soontarinka S, Kantigal P, Visalyaputra S, Deesawat J, Phadermwongsa P, et al. Adding droperidol to morphine patient‐controlled analgesia: effect on nausea and vomiting. Journal of the Medical Association of Thailand 2002 Sep;85(Suppl 3):S923‐33. [; MEDLINE: ; CN‐00411731] [PubMed] [Google Scholar]; 3220962
- Sanchez Ledesma MJ, Lopez Olaondo L, Pueyo FJ, Carrascosa F, Ortega A. A comparison of three antiemetic combinations for the prevention of postoperative nausea and vomiting. Anesthesia & Analgesia 2002 Dec;95(6):1590‐5. [; MEDLINE: ; CN‐00411818] [DOI] [PubMed] [Google Scholar]; 3220964
- Sandhya, Yaddanapudi LN. Evaluation of two antiemetic agents during outpatient gynaecological surgery. Singapore Medical Journal 1994 Jun;35(3):271‐3. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220966
- Saur VP, Muhr C, Kazmaier S, Neumann P, Buhre W. Prophylaxis of postoperative nausea and vomiting using single and repetitive doses of ondansetron ‐ An overview of literature on application methods. Anaesthesiologie Und Reanimation 1996;21(5):131‐5. [; MEDLINE: ; EMBASE 1996350533] [PubMed] [Google Scholar]; 3220968
- Schettini G, Mastronardi P, Scanni E, Pinto M, Forgione A, Florio T, et al. [Antiemetic effect of the levo isomer of sulpiride (L‐sulpiride) in humans] [Article in Italian]. Minerva Anestesiologica 1989 May;55(5):239‐43. [; MEDLINE: ] [PubMed] [Google Scholar]; 3220970
- Schlager A, Mitterschiffthaler G, Puhringer F. Rectally administered dimenhydrinate reduces postoperative vomiting in children after strabismus surgery. British Journal of Anaesthesia 2000 Mar;84(3):405‐6. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220972
- Scholtes JL, Steinier JR, Gersdorff M. Is preloading useful for the complete efficacy of alizapride in highly emetogenic surgical procedures?. Current Therapeutic Research, Clinical and Experimental 1991;50(3):402‐11. [; MEDLINE: ] [Google Scholar]; 3220974
- Scholz J, Hennes HJ, Steinfath M, Farber L, Schweiger C, Dick W, et al. Tropisetron or ondansetron compared with placebo for prevention of postoperative nausea and vomiting. European journal of anaesthesiology 1998;15(6):676‐85. [; MEDLINE: ; CN‐00339487] [DOI] [PubMed] [Google Scholar]; 3220976
- Schuh R, Tolksdorf W, Hucke H. Transdermal scopolamine or droperidol for prevention of nausea and vomiting after cholecystectomy [Transdermales scopolamin oder droperidol zur prophylaxe von postoperativer ubelkeit und erbrechen bei cholecystektomie‐patientenau]. Intensivtherapie Notfallmedizin 1987 Dec;22(6):261‐6. [; MEDLINE: ; AN: 1988101022] [PubMed] [Google Scholar]; 3220978
- Schultz AA, Andrews AL, Goran SF, Mathew T, Sturdevant N. Comparison of acupressure bands and droperidol for reducing post‐operative nausea and vomiting in gynecologic surgery patients. Applied Nursing Research 2003 Nov;16(4):256‐65. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220980
- Schulz‐Stubner S, Wettmann G, Reyle‐Hahn SM, Rossaint R. Magnesium as part of balanced general anaesthesia with propofol, remifentanil and mivacurium: a double‐blind, randomized prospective study in 50 patients. European Journal of Anaesthesiology 2001 Nov;18(11):723‐9. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220982
- Scuderi PE, Weaver RG Jr, James RL, Mims G, Elliott WG, Weeks DB. A randomized, double‐blind, placebo controlled comparison of droperidol, ondansetron, and metoclopramide for the prevention of vomiting following outpatient strabismus surgery in children. Journal of Clinical Anesthesia 1997 Nov;9(7):551‐8. [; MEDLINE: ; AN: 1997324351] [DOI] [PubMed] [Google Scholar]; 3220984
- Scuderi PE, James RL, Harris L, Mims GR 3rd. Antiemetic prophylaxis does not improve outcomes after outpatient surgery when compared to symptomatic treatment. Anesthesiology 1999 Feb;90(2):360‐71. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220986
- Scuderi PE, James RL, Harris L, Mims GR 3rd. Multimodal antiemetic management prevents early postoperative vomiting after outpatient laparoscopy. Anesthesia & Analgesia 2000 Dec;91(6):1408‐14. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220988
- Semple P, Madej TH, Wheatley RG, Jackson IJ, Stevens J. Transdermal hyoscine with patient‐controlled analgesia. Anaesthesia 1992;47(5):399‐401. [; MEDLINE: ; CN‐00218069] [DOI] [PubMed] [Google Scholar]; 3220990
- Sennaraj B, Shende D, Sadhasivam S, Ilavajady S, Jagan D. Management of post‐strabismus nausea and vomiting in children using ondansetron: a value‐based comparison of outcomes. British Journal of Anaesthesia 2002 Sep;89(3):473‐8. [; MEDLINE: ; IS: 0007‐0912] [PubMed] [Google Scholar]; 3220992
- Serrano MY, Luna C. Ondansetron, metoclopramide and difenidol in the prevention of PONV, in ophtalmological surgery [Ondansetron, metoclopramida, difenidol en la prevencion del vomito y nausea postoperatoria en cirugia oftalmologica]. Revista Mexicana de Anestesiologia 1998;21(2):99‐102. [; AN: 1998283461] [Google Scholar]; 3220994
- Shah ZP, Wilson J. An evaluation of metoclopramide (Maxolon) as an anti‐emetic in minor gynaecological surgery. British Journal of Anaesthesia 1972 Aug;44(8):865‐8. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3220996
- Shah FR, Halbe AR, Panchal ID, Goodchild CS. Improvement in postoperative pain relief by the addition of midazolam to an intrathecal injection of buprenorphine and bupivacaine. European Journal of Anaesthesiology 2003 Nov;20(11):904‐10. [; MEDLINE: ; ISSN: 0265‐0215] [DOI] [PubMed] [Google Scholar]; 3220998
- Sharma SK, Davies MW. Patient‐controlled analgesia with a mixture of morphine and droperidol. British Journal of Anaesthesia 1993 Sep;71(3):435‐6. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221000
- Sharma S, Abdullah N. A comparison of commonly used anti‐emetics for the prevention of emetic sequelae after a major gynaecological surgery. Singapore Medical Journal 2000 Apr;41(4):147‐50. [; MEDLINE: ] [PubMed] [Google Scholar]; 3221002
- Shende D, Mandal NG. Efficacy of ondansetron and metoclopramide for preventing postoperative emesis following strabismus surgery in children. Anaesthesia 1997 May;52(5):496‐500. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221004
- Shende D, Haldar M. Prophylactic metoclopramide administered immediately after the induction of anesthesia has no effect on the incidence of postoperative emesis after strabismus surgery. Indian Pediatrics 1998 Mar;35(3):237‐41. [; MEDLINE: ; EMBASE 1998109858] [PubMed] [Google Scholar]; 3221006
- Shende D, Bharti N, Kathirvel S, Madan R. Combination of droperidol and ondansetron reduces PONV after pediatric strabismus surgery more than single drug therapy. Acta Anaesthesiologica Scandinavica 2001 Jul;45(6):756‐60. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221008
- Simpson KH, Dearden MJ, Ellis FR, Jack TM. Premedication with slow release morphine (MST) and adjuvants. British Journal of Anaesthesia 1988 Jun;60(7):825‐30. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221010
- Sinha PK, Tripathi M, Ambesh SP. Efficacy of ondansetron in prophylaxis of postoperative nausea and vomiting in patients following infratentorial surgery: a placebo‐controlled prospective double‐blind study. Journal of Neurosurgical Anesthesiology 1999 Jan;11(1):6‐10. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221012
- Sites BD, Beach M, Biggs R, Rohan C, Wiley C, Rassias A, et al. Intrathecal clonidine added to a bupivacaine‐morphine spinal anesthetic improves postoperative analgesia for total knee arthroplasty. Anesthesia and Analgesia 2003 Apr;96(4):1083‐8. [; MEDLINE: ; 2003129808 20030417] [DOI] [PubMed] [Google Scholar]; 3221014
- Sniadach MS, Alberts MS. A comparison of the prophylactic antiemetic effect of ondansetron and droperidol on patients undergoing gynecologic laparoscopy. Anesthesia & Analgesia 1997 Oct;85:797‐800. [; MEDLINE: ; ISI:A1997XY23200015 ER] [DOI] [PubMed] [Google Scholar]; 3221016
- Snow JC. Trimethobenzamide as an antiemetic in surgery for detached retina. American Journal of Ophthalmology 1967 Jul;64(1):149‐52. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221018
- So JBY, Cheong KF, Sng C, Cheah WK, Goh P. Ondansetron in the prevention of postoperative nausea and vomiting after laparoscopic cholecystectomy: A prospective randomized study. Endoscopy 2002;16(2):286‐8. [; MEDLINE: ; AN: 2002048964] [DOI] [PubMed] [Google Scholar]; 3221020
- Sohi HS, Heipel J, Inman KJ, Chinnick B, Cunningham DG, Holliday RL, et al. Preoperative transdermal scopolamine does not reduce the level of nausea and frequency of vomiting after laparoscopic cholecystectomy. Canadian Journal of Surgery 1994;37(4):307‐12. [; MEDLINE: ; AN: 1994289993] [PubMed] [Google Scholar]; 3221022
- Somri M, Vaida SJ, Sabo E, Yassain G, Gankin I, Gaitini LA. Acupuncture versus ondansetron in the prevention of postoperative vomiting. A study of children undergoing dental surgery. Anaesthesia 2001 Oct;56(10):927‐32. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221024
- Song D, Chung F, Yogendran S, Wong J. Evaluation of postural stability after low‐dose droperidol in outpatients undergoing gynaecological dilatation and curettage procedure. British Journal of Anaesthesia 2002 Jun;88(6):819‐23. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221026
- Spadafora SM, Moote CA. Postoperative nausea and vomiting after outpatient laparoscopy ‐ a random double‐blind placebo comparison of ondansetron, droperidol and metoclopramide. Anesthesia & Analgesia 1994 Feb;78(2):U219. [; ISI:A1994NA40600407 ER] [Google Scholar]; 3221028
- Splinter W, Noel LP, Roberts D, Rhine E, Bonn G, Clarke W. Antiemetic prophylaxis for strabismus surgery. Canadian Journal of Ophthalmology 1994 Oct;29(5):224‐6. [; MEDLINE: ; EMBASE 1994358370] [PubMed] [Google Scholar]; 3221030
- Splinter WM, Rhine EJ, Roberts DW, Baxter MR, Gould HM, Hall LE, et al. Ondansetron is a better prophylactic antiemetic than droperidol for tonsillectomy in children. Canadian Journal of Anaesthesia 1995 Oct;42(10):848‐51. [; MEDLINE: ; EMBASE 1995321144] [DOI] [PubMed] [Google Scholar]; 3221032
- Splinter WM, MacNeil HB, Menard EA, Rhine EJ, Roberts DJ, Gould MH. Midazolam reduces vomiting after tonsillectomy in children. Canadian Journal of Anaesthesia 1995 Mar;42(3):201‐3. [; MEDLINE: ; ISI:A1995QK02300005 ER] [DOI] [PubMed] [Google Scholar]; 3221034
- Splinter WM, Baxter MRN, Gould HM, Hall LE, MacNeill HB, Roberts DJ, et al. Oral ondansetron decreases vomiting after tonsillectomy in children. Canadian Journal of Anaesthesia 1995;42(4):277‐80. [; MEDLINE: ; EMBASE 1995129262] [DOI] [PubMed] [Google Scholar]; 3221036
- Splinter WM, Roberts DJ. Dexamethasone decreases vomiting by children after tonsillectomy. Anesthesia & Analgesia 1996 Nov;83(5):913‐6. [; MEDLINE: ; EMBASE 1996334069] [DOI] [PubMed] [Google Scholar]; 3221038
- Splinter W, Roberts DJ. Prophylaxis for vomiting by children after tonsillectomy: dexamethasone versus perphenazine. Anesthesia & Analgesia 1997 Sep;85(3):534‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221040
- Splinter WM, Rhine EJ. Prophylactic antiemetics in children undergoing tonsillectomy: high‐dose vs low‐dose ondansetron. Paediatric Anaesthesia 1997;7(2):125‐9. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221042
- Splinter WM, Roberts DJ. Perphenazine decreases vomiting by children after tonsillectomy. Canadian Journal of Anaesthesia 1997 Dec;44(12):1308‐10. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221044
- Splinter WM, Rhine EJ. Low‐dose ondansetron with dexamethasone more effectively decreases vomiting after strabismus surgery in children than does high‐dose ondansetron. Anesthesiology 1998 Jan;88(1):72‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221046
- Splinter WM, Rhine EJ. Prophylaxis for vomiting by children after tonsillectomy: ondansetron compared with perphenazine. British Journal of Anaesthesia 1998;80(2):155‐8. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221048
- Splinter WM. Prevention of vomiting after strabismus surgery in children: dexamethasone alone versus dexamethasone plus low‐dose ondansetron. Paediatric Anaesthesia 2001;11(5):591‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221050
- Stead SW, Beatie CD, Keyes M. Effects of droperidol dosage on postoperative emetic symptoms following pediatric strabismus surgery. Anesthesiology 1994 Sep;81:A23. [; SI:A1994PJ09100023 ER] [DOI] [PubMed] [Google Scholar]; 3221052
- Steinbrook RA, Freiberger D, Gosnell JL, Brooks DC. Prophylactic antiemetics for laparoscopic cholecystectomy: Ondansetron versus droperidol plus metoclopramide. Anesthesia and Analgesia 1996;83(5):1081‐3. [; MEDLINE: ; AN: 1996334097] [DOI] [PubMed] [Google Scholar]; 3221054
- Steinbrook RA, Gosnell JL, Freiberger D. Prophylactic antiemetics for laparoscopic cholecystectomy: A comparison of perphenazine, droperidol plus ondansetron, and droperidol plus metoclopramide. Journal of Clinical Anesthesia 1998;10(6):494‐8. [; MEDLINE: ; AN: 1998344118] [DOI] [PubMed] [Google Scholar]; 3221056
- Stene FN, Seay RE, Young LA, Bohnsack LE, Bostrom BC. Prospective, randomized, double‐blind, placebo‐controlled comparison of metoclopramide and ondansetron for prevention of posttonsillectomy or adenotonsillectomy emesis. Journal of Clinical Anesthesia 1996 Nov;8(7):540‐4. [; MEDLINE: ; EMBASE 1996319937] [DOI] [PubMed] [Google Scholar]; 3221058
- Stromberg BV, Reines DH, Ackerly J. Transderm scopolamine for the control of perioperative nausea. The American surgeon 1991;57(1):712‐5. [; MEDLINE: ; ISI:A1991GQ05400007 ER] [PubMed] [Google Scholar]; 3221060
- Subramaniam B, Madan R, Sadhasivam S, Sennaraj B, Tamilseluan P, Rajeshwari S, et al. Dexamethasone is a cost‐effective alternative to ondansertron in preventing PONV after paediatric strabismus repair. British Journal of Anaesthesia 2001;86(1):84‐9. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221062
- Suen TK, Gin TA, Chen PP, Rowbottom YM, Critchley LA, Ray AK. Ondansetron 4 mg for the prevention of nausea and vomiting after minor laparoscopic gynaecological surgery. Anaesthesia & Intensive Care 1994 Apr;22(2):142‐6. [; MEDLINE: ; EMBASE 1994112279] [DOI] [PubMed] [Google Scholar]; 3221064
- Sukhani R, Pappas AL, Lurie J, Hotaling AJ, Park A, Fluder E. Ondansetron and dolasetron provide equivalent postoperative vomiting control after ambulatory tonsillectomy in dexamethasone‐pretreated children. Anesthesia & Analgesia 2002 Nov;95(5):1230‐5. [; MEDLINE: ; CN‐00410876] [DOI] [PubMed] [Google Scholar]; 3221066
- Sun R, Klein K, White PF. Use of ondansetron, metoclopramide and droperidol for preventing postoperative nausea and vomiting after ent surgery. Anesthesiology 1995 Sep;83:A20. [; SI:A1995RX68500020 ER] [Google Scholar]; 3221068
- Sun R, Klein K, Skrivanek G, White PF. Postoperative nausea and vomiting: sevoflurane vs desflurane. Anesthesia & Analgesia 1997 Feb;84:S552. [; ISI:A1997WF78000551 ER] [Google Scholar]; 3221070
- Sun R, Klein K, Skrivanek G, White PF. Factors influencing emetic symptom after outpatient otolaryngologic procedures. Anesthesiology 1997;87(3A):A19. [] [Google Scholar]; 3221072
- Sun R, Klein KW, White PF. The effect of timing of ondansetron administration in outpatients undergoing otolaryngologic surgery. Anesthesia & Analgesia 1997 Feb;84(2):331‐6. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221074
- Sung YF, Wetchler BV, Duncalf D, Joslyn AF. A double‐blind, placebo‐controlled pilot study examining the effectiveness of intravenous ondansetron in the prevention of postoperative nausea and emesis. Journal of Clinical Anesthesia 1993 Jan‐Feb;5(1):22‐9. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221076
- Swiatkowski J, Goral A, Dzieciuch JA, Przesmycki K. Assessment of ondansetron and droperidol for the prevention of post‐operative nausea and vomiting after cholecystectomy and minor gynaecological surgery performed by laparoscopy. European Journal of Anaesthesiology 1999 Nov;16(11):766‐72. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221078
- Szarvas S, Chellapuri RS, Harmon DC, Owens J, Murphy D, Shorten GD. A comparison of dexamethasone, ondansetron, and dexamethasone plus ondansetron as prophylactic antiemetic and antipruritic therapy in patients receiving intrathecal morphine for major orthopedic surgery. Anesthesia & Analgesia 2003 Jul;97(1):259‐63. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221080
- Tan CH, Onsiong MK, Kua JSW. A comparison of droperidol and metoclopramide for the prevention of postoperative nausea and vomiting (PONV) associated with patient‐controlled analgesia (PCA) morphine. Anaesthesia and Intensive Care 1998 Mar;26(3):324‐5. [; MEDLINE: ; AN: 1998182169] [Google Scholar]; 3221082
- Tan PH, Kuo JH, Liu K, Hung CC, Tsai TC, Deng TY. Efficacy of intrathecal neostigmine for the relief of postinguinal hemiorrhaphy pain. Acta Anaesthesiologica Scandinavica 2000 Oct;44(9):1056‐60. [; MEDLINE: ; ISI:000089633600004 ER] [DOI] [PubMed] [Google Scholar]; 3221084
- Tan PH, Liu K, Peng CH, Yang LC, Lin CR, Lu CY. The effect of dexamethasone on postoperative pain and emesis after intrathecal neostigmine. Anesthesia & Analgesia 2001 Jan;92(1):228‐32. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221086
- Tang J, Watcha MF, White PF. A comparison of costs and efficacy of ondansetron and droperidol as prophylactic antiemetic therapy for elective outpatient gynecologic procedures. Anesthesia & Analgesia 1996 Aug;83(2):304‐13. [; MEDLINE: ; ISI:A1996UZ99100018 ER] [DOI] [PubMed] [Google Scholar]; 3221088
- Tang J, Wang B, White PF, Watcha MF, Qi J, Wender RH. The effect of timing of ondansetron administration on its efficacy, cost‐effectiveness, and cost‐benefit as a prophylactic antiemetic in the ambulatory setting. Anesthesia and Analgesia 1998;86(2):274‐82. [; MEDLINE: ; EMBASE 1998055396] [DOI] [PubMed] [Google Scholar]; 3221090
- Tang J, D'Angelo R, White PF, Scuderi PE. The efficacy of RS‐25259, a long‐acting selective 5‐HT3 receptor antagonist, for preventing postoperative nausea and vomiting after hysterectomy procedures. Anesthesia & Analgesia 1998 Aug;87(2):462‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221092
- Tang J, Chen X, White PF, Wender RH, Ma H, Sloninsky A, et al. Antiemetic prophylaxis for office‐based surgery: are the 5‐HT3 receptor antagonists beneficial?. Anesthesiology 2003 Feb;98(2):293‐8. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221094
- Tarkkila P, Torn K, Tuominen M, Lindgren L. Premedication with promethazine and transdermal scopolamine reduces the incidence of nausea and vomiting after intrathecal morphine. Acta Anaesthesiologica Scandinavica 1995 Oct;39(7):983‐6. [; MEDLINE: ; EMBASE 1995294959] [DOI] [PubMed] [Google Scholar]; 3221096
- TerRiet MF, Jacobs JS, Nunez CM, Gold MI. Dose‐response effect of three antiemetics on alfentanil‐related postoperative nausea and vomiting (PONV). Anesthesiology 1997 Sep;87:A47. [; SI:A1997XV63600047 ER] [Google Scholar]; 3221098
- Tezcan C, Donmez A, Tulunay M, Asik I. The antiemetic effect of ephedrine in patients undergoing gynecologic laparoscopy. Turk Anesteziyoloji Ve Reanimasyon 1993;21(6):340‐4. [; EMBASE 1994078485] [Google Scholar]; 3221100
- Thagaard KS, Steine S, Raeder J. Ondansetron disintegrating tablets of 8 mg twice a day for 3 days did not reduce the incidence of nausea or vomiting after laparoscopic surgery. European Journal of Anaesthesiology 2003 Feb;20(2):153‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221102
- Thomas R, Jones N. Prospective randomized, double‐blind comparative study of dexamethasone, ondansetron, and ondansetron plus dexamethasone as prophylactic antiemetic therapy in patients undergoing day‐case gynaecological surgery. British Journal of Anaesthesia 2001 Oct;87(4):588‐92. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221104
- Tigerstedt I, Salmela L, Aromaa U. Double‐blind comparison of transdermal scopolamine, droperidol and placebo against postoperative nausea and vomiting. Acta Anaesthesiologica Scandinavica 1988 Aug;32(6):454‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221106
- Ting CK, Lin SM, Yang YW, Tsai HJ, Lao HC, Chu YC, et al. Reversal of mivacurium chloride: edrophonium of spontaneous recovery in microscopic laryngeal surgery. Acta Anaesthesiologica Sinica 2001 Dec;39(4):157‐62. [; MEDLINE: ; CN‐00377862 EMBASE 2002029114] [PubMed] [Google Scholar]; 3221108
- Tokat O, Ozcan B, Kahveci SF, Yilmazlar A. The effectivity of ondansetron on postoperative nausea and vomiting in the gynaecological operations [Jinekolojik operasyonlarda ondansetron'un post‐operatif bulanti‐kusmaya etkisi]. Turk Anesteziyoloji Ve Reanimasyon 1994;22(6):356‐8. [; EMBASE 1995093099] [Google Scholar]; 3221110
- Tolksdorf W, Meisel R, Muller P, Bender HJ. [Transdermal scopolamine (TTS‐scopolamine) for the prevention of postoperative nausea and vomiting]. [German]. Anaesthesist 1985 Dec;34(12):656‐62. [; MEDLINE: ] [PubMed] [Google Scholar]; 3221112
- Tolksdorf W, Baumann S, Heine C, Hettenbach A. Effects and side effects of transdermal scopolamine in premedication before general anaesthesia in elderly patients [Wirkungen und nebenwirkungen transdermalen scopolamins zur pramedikation vor allgemeinanasthesien bei alteren patienten]. Anasthesiol Intensivmed Notfallmed Schmerzther 1986;21(6):301‐6. [; MEDLINE: ; EMBASE 1987047923] [PubMed] [Google Scholar]; 3221114
- Tolksdorf W, Krug C, Hartung M, Hettenbach A. Effect and side effects of oral morphine, lormetazepam and placebos as premedication. Anasthesiol Intensivmed Notfallmed Schmerzther 1987;22(3):113‐7. [; MEDLINE: ; CN‐00212872] [PubMed] [Google Scholar]; 3221116
- Tom LWC, Templeton JJ, Thompson ME, Marsh RR. Dexamethasone in adenotonsillectomy. International Journal of Pediatric Otorhinolaryngology 1996 Oct;37(2):115‐20. [; IS: 0165‐5876] [DOI] [PubMed] [Google Scholar]; 3221118
- Top G, Baran O, Eroglu F, Akaltan A, Ertunc N. The effect of dexametasone to prophylactic ondansetron in prevention of postoperative nausea and vomiting [Postoperatif bulanti ve kusmanin onlenmesinde profilaktik ondansetrona deksametazonun etkisi]. Turk Anesteziyoloji ve Reanimasyon 1996;24(5):220‐4. [; AN: 1996245040] [Google Scholar]; 3221120
- Tosun Z, Akin A, Güler G, Mizrak A, Boyaci A. The effect of tropisetron on postoperative nausea and vomiting after strabismus surgery. Turk Anesteziyoloji ve Reanimasyon 2002;30(10):454‐7. [; 2003042373 20030207 ISSN: 1016‐5150] [Google Scholar]; 3221122
- Trakya A, Sahin N, Oznalcilar H, Atay O, Icel E, Ozmenoglu H. A comparison of the doses of prophylactic ondansetron administration in preventing postoperative nausea and vomiting [Postoperatif bulanti ve kusmanin onlenmesinde porfilaktik ondansetron kullaniminda doz karsilastirilmasi]. Turk Anesteziyoloji ve Reanimasyon 1996;24(8):375‐9. [; AN: 1996340313] [Google Scholar]; 3221124
- Tramer MR, Sansonetti A, Fuchs‐Buder T, Rifat K. Oculocardiac reflex and postoperative vomiting in paediatric strabismus surgery. A randomised controlled trial comparing four anaesthetic techniques. Acta Anaesthesiologica Scandinavica 1998 Jan;42(1):117‐23. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221126
- Triem JG, Piper SN, Maleck WH, Schenck A, Schmidt CC, Boldt J. [Prevention of postoperative nausea and vomiting after hysterectomy with oral dolasetron, intravenous dehydrobenzperidol or a combination of both substances]. [German]. Anasthesiologie Intensivmedizin Notfallmedizin Schmerztherapie 1999 Jun;34(6):340‐4. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221128
- Tripple GE, Holland MS, Hassanein K. Comparison of droperidol 0.01 mg/kg and 0.005 mg/kg as a premedication in the prevention of nausea and vomiting in the outpatient for laparoscopy. AANA Journal 1989 Oct;57(5):413‐6. [; MEDLINE: ] [PubMed] [Google Scholar]; 3221130
- Tsui SL, Ng KF, Wong LC, Tang GW, Pun TC, Yang JC. Prevention of postoperative nausea and vomiting in gynaecological laparotomies: a comparison of tropisetron and ondansetron. Anaesthesia & Intensive Care 1999 Oct;27(5):471‐6. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221132
- Tuncer S, Bariskaner H, Yosunkaya A, Tavlan A. Influence of Dexamethasone on Nausea and Vomiting during Patient‐Controlled Analgesia with Tramadol. Clinical Drug Investigation 2002;22(8):547‐52. [; IS: 1173‐2563] [Google Scholar]; 3221134
- Tur A, Ustun E, Esener Z, Gunaydin A, Guldogus F, Sarihasan B. The effect of oral ondansetron premedication on the postoperative emesis. Turk Anesteziyoloji ve Reanimasyon 1994;22(1):29‐33. [; AN: 1994078496] [Google Scholar]; 3221136
- Tur A, Ustun E, Esener Z, Gunaydin A. Comparison of the effects of 4, 8 and 16 mg of oral ondansetron premedication on postoperative emesis [Postoperatif emezise etkileri yonunden premedikasyonda 4, 8 ve 16 mg'lik oral ondansetron dozlarinin karsilastirilmasi]. Turk Anesteziyoloji ve Reanimasyon 1995;23(4):172‐5. [; AN: 1995240868] [Google Scholar]; 3221138
- Turan A, Memis D, Basaran UN, Karamanlioglu B, Sut N. Caudal ropivacaine and neostigmine in pediatric surgery. Anesthesiology 2003 Mar;98(3):719‐22. [; MEDLINE: ; CN‐00413387] [DOI] [PubMed] [Google Scholar]; 3221140
- Turhanoglu S, Ozyilmaz MA, Tok D, Olmez G, Cinar FS, Bayhan N. A comparison of the effects of ondansetron with or without dimenhydrinate in the prevention of nausea and vomiting after major gynaecological surgery. Acta Anaesthesiologica Italica/Anaesthesia and Intensive Care in Italy 1999;50(3):193‐9. [; AN: 2000203607] [Google Scholar]; 3221142
- Turkoglu M, Ozyar B, Kayaalti B, Ugur G. Comparison of the effects of droperidol, chlorpromazine and lidocaine on the incidence of postoperative nausea‐vomiting in children undergoing strabismus surgery [Sasilik operasyonu uygulanan cocuklarda postoperatif bulanti‐kusma uzerine droperidol, lidokain ve klorpromazinin etkilerinin karsilastirilmasi]. Turk Anesteziyoloji Ve Reanimasyon 1995;23(2):66‐9. [; EMBASE 1995326395] [Google Scholar]; 3221144
- Tzeng JI, Wang JJ, Ho ST, Tang CS, Liu YC, Lee SC. Dexamethasone for prophylaxis of nausea and vomiting after epidural morphine for post‐Caesarean section analgesia: Comparison of droperidol and saline. British Journal of Anaesthesia 2000;85(6):865‐8. [; MEDLINE: ; AN: 2000429964] [DOI] [PubMed] [Google Scholar]; 3221146
- Tzeng JI, Tswei TS, Tang CS, Ho ST, Wang JJ. Dexamethasone alone does not prevent postoperative nausea and vomiting in women undergoing dilatation and curettage: A comparison with droperidol and saline. Acta Anaesthesiologica Sinica 2000;38(3):137‐42. [; MEDLINE: ; AN: 2001015764] [PubMed] [Google Scholar]; 3221148
- Tzeng JI, Hsing CH, Chu CC, Chen YH, Wang JJ. Low‐dose dexamethasone reduces nausea and vomiting after epidural morphine: A comparison of metoclopramide with saline. Journal of Clinical Anesthesia 2002;14(1):19‐23. [; MEDLINE: ; AN: 2002078595] [DOI] [PubMed] [Google Scholar]; 3221150
- Tzeng JI, Chu KS, Ho ST, Cheng KI, Liu KS, Wang JJ. Prophylactic iv ondansetron reduces nausea, vomiting and pruritus following epidural morphine for postoperative pain control [L'administration prophylactique d'ondansetron iv reduit les nausees, les vomissements et le prurit qui suivent l'administration peridurale de morphine analgesique postoperatoire L'L'administration prophylactique d'ondansetron iv reduit les nausees, les vomissements et le prurit qui suivent l'administration peridurale de morphine analgesique postoperatoire]. Canadian Journal of Anaesthesia 2003 Dec;50(10):1023‐6. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221152
- Uerpairojkit K, Pavaves B, Nalawachai J. A placebo‐controlled, randomized trial of droperidol versus metoclopramide for outpatients undergoing gynecological laparoscopy under conscious sedation. Journal of the Medical Association of Thailand 2002 Apr;85(4):470‐6. [; MEDLINE: ] [PubMed] [Google Scholar]; 3221154
- Ummenhofer W, Frei FJ, Urwyler A, Kern C, Drewe J. Effects of ondansetron in the prevention of postoperative nausea and vomiting in children. Anesthesiology 1994;81(4):804‐10. [; MEDLINE: ; EMBASE 1994335710] [DOI] [PubMed] [Google Scholar]; 3221156
- Unlugenc H, Gunduz M, Ozalevli M, Akman H. A comparative study on the analgesic effect of tramadol, tramadol plus magnesium, and tramadol plus ketamine for postoperative pain management after major abdominal surgery. Acta Anaesthesiologica Scandinavica 2002 Sep;46(8):1025‐30. [; MEDLINE: ; CN‐00397858] [DOI] [PubMed] [Google Scholar]; 3221158
- Unseld H, Kahny B. Prevention of postoperative vomiting through the use of histamine receptor antagonists. Anasthesiologie Intensivmedizin Notfallmedizin Schmerztherapie 1996;37(1):29‐34. [; ISI:A1996VG36900005 ER] [Google Scholar]; 3221160
- Uppington J, Dunnett J, Blogg CE. Transdermal hyoscine and postoperative nausea and vomiting. Anaesthesia 1986;41(1):16‐20. [; MEDLINE: ; AN: 1986099664] [DOI] [PubMed] [Google Scholar]; 3221162
- Usha Rani P, Rama Raju GA, Naidu MUR, Jagga Rao P, Ramesh Kumar T, Shobha JC, et al. Randomised, double blind, placebo controlled study of Ondansetron in female patients undergoing day case surgery. Journal of Anaesthesiology Clinical Pharmacology 1996;12(1):31‐4. [; MEDLINE: ] [Google Scholar]; 3221164
- Usmani H, Quadir A, Siddiqui RA, Sharma SC. Ondansetron and dexamethasone in middle ear procedures. Indian Journal of Otolaryngology and Head and Neck Surgery 2003;55(2):97‐9. [; 2003309155 20030814 ISSN: 0019‐5421] [DOI] [PMC free article] [PubMed] [Google Scholar]; 3221166
- Valanne J, Korttila K. Effect of a small dose of droperidol on nausea, vomiting and recovery after outpatient enflurane anaesthesia. Acta Anaesthesiologica Scandinavica 1985 May;29(4):359‐62. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221168
- Valentine JMJ, Lyons G, Bellamy MC. The effect of intrathecal midazolam on post‐operative pain. European Journal of Anaesthesiology 1996 Nov;13(6):589‐93. [; MEDLINE: ; IS: 0265‐0215] [DOI] [PubMed] [Google Scholar]; 3221170
- Berg AA, Lambourne A, Yazji NS, Laghari NA. Vomiting after ophthalmic surgery. Effects of intra‐operative antiemetics and postoperative oral fluid restriction. Anaesthesia 1987;42(3):270‐6. [; MEDLINE: ; EMBASE 1987119913] [DOI] [PubMed] [Google Scholar]; 3221172
- Berg AA, Savva D, Honjol NM, Prabhu NV. Prevention of PONV following ent surgery: Controlled comparison of ondansetron and prochlorperazine. British Journal of Anaesthesia 1995 May;74:91 (A298). [; ISI:A1995RA77700298 ER] [Google Scholar]; 3221174
- Berg AA. A comparison of ondansetron and prochlorperazine for the prevention of nausea and vomiting after tympanoplasty. Canadian Journal of Anaesthesia 1996 Sep;43(9):939‐45. [; MEDLINE: ; EMBASE 1996297288] [DOI] [PubMed] [Google Scholar]; 3221176
- Berg AA. Comparison of ondansetron and prochlorperazine for the prevention of nausea and vomiting after adenotonsillectomy. British Journal of Anaesthesia 1996 Mar;76(3):449‐51. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221178
- Berg AA. The prophylactic antiemetic efficacy of prochlorperazine and ondansetron in nasal septal surgery: a randomized double‐blind comparison. Anaesthesia & Intensive Care 1996 Oct;24(5):538‐45. [; MEDLINE: ; EMBASE 1996343330] [DOI] [PubMed] [Google Scholar]; 3221180
- Walt JH, Nicholls B, Bentley M, Tomkins DP. Oral premedication in children. Anaesthesia & Intensive Care 1987 May;15(2):151‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221182
- Walt JH, Jacob R, Murrell D, Bentley M. The perioperative effects of oral premedication in children. Anaesthesia & Intensive Care 1990 Feb;18(1):5‐10. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221184
- Vener DF, Carr AS, Sikich N, Bissonnette B, Lerman J. Dimenhydrinate decreases vomiting after strabismus surgery in children. Anesthesia & Analgesia 1996 Apr;82(4):728‐31. [; MEDLINE: ; EMBASE 1996111715] [DOI] [PubMed] [Google Scholar]; 3221186
- Vimlati L, Pallos J, Nagy K, Muranyi M, Szabo Z, Karoliny V, et al. [Postoperative vomiting][Article in Hungarian]. Orvosi hetilap 2002 Apr;143(16):819‐24. [; MEDLINE: ] [PubMed] [Google Scholar]; 3221188
- Visalyaputra S, Petchpaisit N, Somcharoen K, Choavaratana R. The efficacy of ginger root in the prevention of postoperative nausea and vomiting after outpatient gynaecological laparoscopy. Anaesthesia 1998 May;53(5):506‐10. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221190
- Vollmer LB, Sonne N. The effect of metoclopramide on postoperative nausea and vomiting. Ugeskrift for Laeger 1988;150(3):154‐5. [; AN: 1988022198] [PubMed] [Google Scholar]; 3221192
- Volpe N, Gesini A, Collini S, Grassano MT, Guzzon D, Marino MR, et al. Single dose ondansetron for prevention of postoperative nausea and vomiting. Results from the Italian Multicentre Ondansetron Study. Drug Invest 1994;8(2):67‐72. [; EMBASE 1994264940] [Google Scholar]; 3221194
- Vosdoganis F, Baines DB. The effect of single dose intravenous dexamethasone in tonsillectomy in children. Anaesthesia & Intensive Care 1999 Oct;27(5):489‐92. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221196
- Wagley C, Hackett C, Haug RH. The effect of preoperative ondansetron on the incidence of postoperative nausea and vomiting in patients undergoing outpatient dentoalveolar surgery and general anesthesia. Journal of Oral & Maxillofacial Surgery 1999 Oct;57(10):1195‐200. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221198
- Wagner BKJ, Berman SL, Devitt PA, Halvorsen MB, OHara DA. A double‐blind, placebo‐controlled evaluation of intranasal metoclopramide in the prevention of postoperative nausea and vomiting. Pharmacotherapy 1996 Nov;16(6):1063‐9. [; MEDLINE: ; ISI:A1996VU95700013ER] [PubMed] [Google Scholar]; 3221200
- Wagner D, Pandit U, Voepel‐Lewis T, Weber M. Dolasetron for the prevention of postoperative vomiting in children undergoing strabismus surgery. Paediatric Anaesthesia 2003 Jul;13(6):522‐6. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221202
- Walder AD, Aitkenhead AR. Antiemetic efficacy of metoclopramide when included in a patient‐controlled analgesia infusion. Anaesthesia 1994 Sep;49(9):804‐6. [; MEDLINE: ; EMBASE 1994292995] [DOI] [PubMed] [Google Scholar]; 3221204
- Walder AD, Aitkenhead AR. A comparison of droperidol and cyclizine in the prevention of postoperative nausea and vomiting associated with patient‐controlled analgesia. Anaesthesia 1995;50(7):654‐6. [; MEDLINE: ; EMBASE 1995211697] [DOI] [PubMed] [Google Scholar]; 3221206
- Waldmann CS, Verghese C, Short SM, Goldhill DR, Evans SJ. The evaluation of domperidone and metoclopramide as antiemetics in day care abortion patients. British Journal of Clinical Pharmacology 1985 Mar;19(3):307‐10. [; MEDLINE: ] [DOI] [PMC free article] [PubMed] [Google Scholar]; 3221208
- Wang JJ, Ho ST, Liu YH, Lee SC, Liu YC, Liao YC, et al. Dexamethasone reduces nausea and vomiting after laparoscopic cholecystectomy. British Journal of Anaesthesia 1999 Nov;83(5):772‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221210
- Wang JJ, Ho ST, Liu YH, Ho CM, Liu K, Chia YY. Dexamethasone decreases epidural morphine‐related nausea and vomiting. Anesthesia & Analgesia 1999;89(1):117‐20. [; MEDLINE: ; ISI:000081101100020 ER] [DOI] [PubMed] [Google Scholar]; 3221212
- Wang JJ, Ho ST, Lee SC, Liu YC, Liu YH, Liao YC. The prophylactic effect of dexamethasone on postoperative nausea and vomiting in women undergoing thyroidectomy: A comparison of droperidol with saline. Anesthesia & Analgesia 1999 Jul;89(a):200‐3. [; MEDLINE: ; ISI:000081101100036 ER] [DOI] [PubMed] [Google Scholar]; 3221214
- Wang JJ, Ho ST, Liu HS, Ho CM. Prophylactic antiemetic effect of dexamethasone in women undergoing ambulatory laparoscopic surgery. British Journal of Anaesthesia 2000;84(4):459‐62. [; MEDLINE: ; CN‐00309929] [DOI] [PubMed] [Google Scholar]; 3221216
- Wang JJ, Ho ST, Tzeng JI, Tang CS. The effect of timing of dexamethasone administration on its efficacy as a prophylactic antiemetic for postoperative nausea and vomiting. Anesthesia & Analgesia 2000 Jul;91(1):136‐9. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221218
- Wang JJ, Ho ST, Lee SC, Liu YC, Ho CM. The use of dexamethasone for preventing postoperative nausea and vomiting in females undergoing thyroidectomy: A dose‐ranging study. Anesthesia & Analgesia 2000;91(6):1404‐7. [; MEDLINE: ; AN: 2000413860] [DOI] [PubMed] [Google Scholar]; 3221220
- Wang JJ, Ho ST, Wong CS, Tzeng JI, Liu HS, Ger LP. Dexamethasone prophylaxis of nausea and vomiting after epidural morphine for post‐Cesarean analgesia. Canadian Journal of Anaesthesia 2001 Feb;48(2):185‐90. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221222
- Wang JJ, Tzeng JI, Ho ST, Chen JY, Chu CC, So EC. The prophylactic effect of tropisetron on epidural morphine‐related nausea and vomiting: A comparison of dexamethasone with saline. Anesthesia & Analgesia 2002;94(3):749‐53. [; MEDLINE: ; AN: 2002081175] [DOI] [PubMed] [Google Scholar]; 3221224
- Wang SM, Kain ZN. P6 acupoint injections are as effective as droperidol in controlling early postoperative nausea and vomiting in children. Anesthesiology 2002 Aug;97(2):359‐66. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221226
- Wang JJ, Ho ST, Uen YH, Lin MT, Chen KT, Huang JC, et al. Small‐dose dexamethasone reduces nausea and vomiting after laparoscopic cholecystectomy: a comparison of tropisetron with saline. Anesthesia & Analgesia 2002 Jul;95(1):229‐32. [; MEDLINE: ; CN‐00389883] [DOI] [PubMed] [Google Scholar]; 3221228
- Wang C, Xu N, Xiong LZ, Liu HF, Yang XY, Lu ZH, et al. Dexamethasone enhances the effect of tropisetron and ondansetron against nausea and vomiting against nausea and vomiting after patient‐controlled analgesia. Di Yi Junyi Daxue Xuebao 2002 Feb;22(2):159‐61. [; MEDLINE: ; CN‐00410676] [PubMed] [Google Scholar]; 3221230
- Wang JJ, Wang PC, Liu YH, Chien CC. Low‐dose dexamethasone reduces nausea and vomiting after tympanomastoid surgery: a comparison of tropisetron with saline. American Journal of Otolaryngology 2002 Sep‐Oct;23(5):267‐71. [; MEDLINE: ; CN‐00410101] [DOI] [PubMed] [Google Scholar]; 3221232
- Wang YJ, Cheng ZG, Guo QL. Clinical observation of granisetron in preventing postoperative nausea and vomiting following supratentorial craniotomy. Hunan Yi Ke Da Xue Xue Bao 2002 Dec 28;27(6):545‐6. [; MEDLINE: ] [PubMed] [Google Scholar]; 3221234
- Warrick PD, Belo SE. Treating "rebound" emesis following outpatient gynecologic laparoscopy: the efficacy of a two‐dose regimen of droperidol and ondansetron. Journal of Clinical Anesthesia 1999 Mar;11(2):119‐25. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221236
- Warriner CB, Knox D, Belo S, Cole C, Finegan BA, Perreault L. Prophylactic oral dolasetron mesylate reduces nausea and vomiting after abdominal hysterectomy. The Canadian Dolasetron Study Group.. Canadian Journal of Anaesthesia 1997 Nov;44(11):1167‐73. [; MEDLINE: ; EMBASE 1998036931] [DOI] [PubMed] [Google Scholar]; 3221238
- Watcha MF, Simeon RM, White PF, Stevens JL. Effect of propofol on the incidence of postoperative vomiting after strabismus surgery in pediatric outpatients. Anesthesiology 1991 Aug;75(2):204‐9. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221240
- Watcha MF, Safavi FZ, McCulloch DA, Tan TS, White PF. Effect of antagonism of mivacurium‐induced neuromuscular block on postoperative emesis in children. Anesthesia & Analgesia 1995 Apr;80(4):713‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221242
- Watcha MF, Bras PJ, Cieslak GD, Pennant JH. The dose‐response relationship of ondansetron in preventing postoperative emesis in pediatric patients undergoing ambulatory surgery. Anesthesiology 1995 Jan;82(1):47‐52. [; MEDLINE: ; EMBASE 1995035248] [DOI] [PubMed] [Google Scholar]; 3221244
- Watts SA. A randomized double‐blinded comparison of metoclopramide, ondansetron and cyclizine in day‐case laparoscopy. Anaesthesia & Intensive Care 1996 Oct;24(5):546‐51. [; MEDLINE: ; EMBASE 1996343331] [DOI] [PubMed] [Google Scholar]; 3221246
- Wattwil M, Thorn SE, Lovqvist A, Wattwil L, Gupta A, Liljegren G. Dexamethasone is as effective as ondansetron for the prevention of postoperative nausea and vomiting following breast surgery. Acta Anaesthesiologica Scandinavica 2003 Aug;47(7):823‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221248
- Welters ID, Menges T, Graf M, Beikirch C, Menzebach A, Hempelmann G. Reduction of postoperative nausea and vomiting by dimenhydrinate suppositories after strabismus surgery in children. Anesthesia & Analgesia 2000 Feb;90(2):311‐4. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221250
- Welters ID, Graef M, Menges T, Beikirch C, Kaufmann H, Hempelmann G. Postoperative nausea and vomiting after Faden operation. Albrecht von Graefes Archiv fur klinische und experimentelle Ophthalmologie 2000 Jan;238(1):59‐63. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221252
- Whalley DG, AlHaddad S, Khalil I, Maurer W, Furgerson C. Metoclopramide does not decrease the incidence of nausea and vomiting after alfentanil for outpatient anaesthesia. Canadian Journal of Anaesthesia 1991 Nov;38(8):1023‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221254
- White LA, Vanarase M, Brockbank K, Barrett RF. Patient‐controlled analgesia and postoperative nausea and vomiting: efficacy of a continuous infusion of ondansetron. Anaesthesia 2001 Apr;56(4):365‐9. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221256
- Wildersmith CH, Wildersmith OHG, Farschtshian M, Naji P. Epidural droperidol reduces the side‐effects and duration of analgesia of epidural sufentanil. Anesthesia & Analgesia 1994 Jul;79(1):98‐104. [; MEDLINE: ; ISI:A1994NU61200019 ER] [DOI] [PubMed] [Google Scholar]; 3221258
- Wilkinson AR, Frampton CM, Glover PW, Davis FM. Preoperative transdermal hyoscine for the prevention of postoperative nausea and vomiting. Anaesthesia & Intensive Care 1989 Aug;17(3):285‐9. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221260
- Williams OA, Clarke FL, Harris RW, Smith P, Peacock JE. Addition of droperidol to patient‐controlled analgesia: effect on nausea and vomiting. Anaesthesia 1993 Oct;48(10):881‐4. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221262
- Williams PI, Smith M. An assessment of prochlorperazine buccal for the prevention of nausea and vomiting during intravenous patient‐controlled analgesia with morphine following abdominal hysterectomy. European Journal of Anaesthesiology 1999 Sep;16(9):638‐45. [; MEDLINE: ; MEDLINE 20017231] [DOI] [PubMed] [Google Scholar]; 3221264
- Wilson J. Lorazepam as a premedicant for general anaesthesia. Current Medical Research & Opinion 1973;1(5):308‐16. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221266
- Wilson DB, Dundee JW. Evaluation of the anti‐emetic action of domperidone. Anaesthesia 1979 Sep;34(8):765‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221268
- Wilson AJ, Diemunsch P, Lindeque BG, Scheinin H, Helbo‐Hansen HS, Kroeks MV, et al. Single‐dose i.v. granisetron in the prevention of postoperative nausea and vomiting. British Journal of Anaesthesia 1996 Apr;76(4):515‐8. [; MEDLINE: ; EMBASE 1996107544] [DOI] [PubMed] [Google Scholar]; 3221270
- Wilson EB, Bass CS, Abrameit W, Roberson R, Smith RW. Metoclopramide versus ondansetron in prophylaxis of nausea and vomiting for laparoscopic cholecystectomy. American Journal of Surgery 2001 Feb;181(2):138‐41. [; MEDLINE: ; ISI:000168416800010 ER] [DOI] [PubMed] [Google Scholar]; 3221272
- Winning TJ, Brock‐Utne JG, Downing JW. Nausea and vomiting after anesthesia and minor surgery. Anesthesia & Analgesia 1977 Sep‐Oct;56(5):674‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221274
- Woodward DK, Sherry KM, Harrison D. Antiemetic prophylaxis in cardiac surgery: comparison of metoclopramide and ondansetron. British Journal of Anaesthesia 1999 Dec;83(6):933‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221276
- Wrench IJ, Ward JEH, Walder AD, Hobbs GJ. The prevention of postoperative nausea and vomiting using a combination of ondansetron and droperidol. Anaesthesia 1996;51(8):776‐8. [; MEDLINE: ; EMBASE 1996256888] [DOI] [PubMed] [Google Scholar]; 3221278
- Wu O, Belo SE, Koutsoukos G. Additive anti‐emetic efficacy of prophylactic ondansetron with droperidol in out‐patient gynecological laparoscopy. Canadian Journal of Anaesthesia 2000 Jun;47(6):529‐36. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221280
- Wu YW, Xu JS, Chen ZQ, Gu MN, Wang YZ. Effects of dexamethasone on epidural morphine‐related nausea and vomiting. Di Yi Junyi Daxue Xuebao 2002 Aug;22(8):758‐9. [; MEDLINE: ; CN‐00410496] [PubMed] [Google Scholar]; 3221282
- Yazigi A, Chalhoub V, Madi‐Jebara S, Haddad F, Hayek G. Prophylactic ondansetron is effective in the treatment of nausea and vomiting but not on pruritus after cesarean delivery with intrathecal sufentanil‐morphine. Journal of Clinical Anesthesia 2002;14(3):183‐6. [; MEDLINE: ; AN: 2002190456] [DOI] [PubMed] [Google Scholar]; 3221284
- Yegin A, Yilmaz M, Karsli B, Erman M. Analgesic effects of intrathecal neostigmine in perianal surgery. European Journal of Anaesthesiology 2003 May;20(5):404‐8. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221286
- Yelken BB, Güleç S, Hedbe L, Ekemen S, Tanriverdi B. Comparison of Ondansetron, Tropisetron, Granisetron and Droperidol for Prevention of PONV After Gyneocological Laparoscopy in Patients Receiving TIVA with Remifentanyl and Propofol. Turk Anesteziyoloji ve Reanimasyon Dernegi Dergisi Apr 2003;31(4):189‐94. [; 2003420723 20031106 ISSN: 1304‐0871] [Google Scholar]; 3221288
- Yilmazlar A, Yilmazlar T, Gurpinar E, Korun N, Kutlay O. Anti‐emetic efficacy of tropisetron and metoclopramide. Journal of International Medical Research 1996 May‐Jun;24(3):266‐70. [; MEDLINE: ; EMBASE 1996174641] [DOI] [PubMed] [Google Scholar]; 3221290
- Yilmazlar A, Tokat O, Kutlay O, Yilmazlar T, Turker G. Comparison of the efficacy of 2 mg versus 5 mg tropisetron in the management of post‐operative nausea and vomiting. Journal of International Medical Research 2001;29(5):385‐8. [; AN: 2001403498] [DOI] [PubMed] [Google Scholar]; 3221292
- Yin YC, Chow LH, Tsao CM, Chu CC, Tsou MY, Chan KH, et al. Oral clonidine reduces myocardial ischemia in patients with coronary artery disease undergoing noncardiac surgery. Acta Anaesthesiologica Sinica 2002 Dec;40(4):197‐203. [; MEDLINE: ] [PubMed] [Google Scholar]; 3221294
- Zarate E, Watcha MF, White PF, Klein KW, Sa‐Rego M, Stewart DG. A comparison of the costs and efficacy of ondansetron versus dolasetron for antiemetic prophylaxis. Anesthesia and Analgesia 2000;90(6):1352‐8. [; MEDLINE: ; AN: 2000203082] [DOI] [PubMed] [Google Scholar]; 3221296
- Zatman TF, Hall JE, Harmer M. Gastric residual volume in children: a study comparing efficiency of erythromycin and metoclopramide as prokinetic agents. British Journal of Anaesthesia 2001 Jun;86(6):869‐71. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221298
- Zomers PJ, Langenberg CJ, Bruijn KM. Tropisetron for postoperative nausea and vomiting in patients after gynaecological surgery. British Journal of Anaesthesia 1993 Nov;71(5):677‐80. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221300
- Zsigmond EK. Double blind evaluation of the efficacy and safety of diazepam as preanesthetic medication [Evaluation en 'double aveugle' de l'efficacite et de la surete d'emploi du diazepam comme therapeutique pre anesthesique]. Anesthesie, analgesie, reanimation 1974;31(5):665‐72. [; MEDLINE: ; EMBASE 1975164618] [PubMed] [Google Scholar]; 3221302
References to studies excluded from this review
- Abouleish EI, Rashid S, Haque S, Giezentanner A, Joynton P, Chuang AZ. Ondansetron versus placebo for the control of nausea and vomiting during Caesarean section under spinal anaesthesia. Anaesthesia 1999 May;54(5):479‐82. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221304
- Alexander R, Fennelly M. Comparison of ondansetron and metoclopramide as premedicants to prevent postoperative nausea and vomiting following orthopedic‐ surgery. British Journal of Anaesthesia 1995 May;74:91. [; ISI:A1995RA77700296 ER] [Google Scholar]; 3221306
- Alon E, Zamboni P, Hossli G. Postoperative antiemetic prophylaxis with haloperidol and metoclopramid in differential dosages. Schweizerische medizinische Wochenschrift 1987 Mar;17(12):460. [; ISI:A1987G525100031 ER] [Google Scholar]; 3221308
- Alon E, Lenzlinger PM, Pasch T. Ondansetron 4 mg vs droperidol 1.25 mg in the prophylaxis of postoperative nausea and vomiting after alfentanil‐supplemented inhalation anesthesia. British journal of anaesthesia 1993 May;70:6. [; ISI:A1993LD22600012 ER] 8431336 [Google Scholar]; 3221310
- Ambesh SP, Dubey PK, Sinha PK. Ondansetron pretreatment to alleviate pain on propofol injection: a randomized, controlled, double‐blinded study. Anesthesia & Analgesia 1999 Jul;89(1):197‐9. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221312
- Apfel CC, Korttila K, Abdalla M, Biedler A, Kranke P, Pocock SJ, et al. An international multicenter protocol to assess the single and combined benefits of antiemetic interventions in a controlled clinical trial of a 2x2x2x2x2x2 factorial design (IMPACT). Controlled clinical trials 2003 Dec;24(6):736‐51. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221314
- April MM, Callan ND, Nowak DM, Hausdorff MA. The effect of intravenous dexamethasone in pediatric adenotonsillectomy. International Journal of Pediatric Otorhinolaryngology 1996 Aug;36(3):271. [; IS: 0165‐5876] [DOI] [PubMed] [Google Scholar]; 3221316
- Arain SR, Ebert TJ. The efficacy, side effects, and recovery characteristics of dexmedetomidine versus propofol when used for intraoperative sedation. Anesthesia & Analgesia 2002 Aug;95(2):461‐6. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221318
- Aromaa U, Salmela L, Tigerstedt I. Double‐blind comparison of transdermal scopolamine, droperidol and placebo against postoperative nausea and vomiting. Acta Anaesthesiol Scand 1987;31:94. [; ISI:A1987K100500092 ER] [DOI] [PubMed] [Google Scholar]; 3221320
- Arun Kumar Paul, Ghosh B. Prochlorperazine (STEMETIL) in pre‐anaesthetic medication. Medicine and Surgery 1987;27(1):8‐9+16A. [; AN: 1988192913] [Google Scholar]; 3221322
- Assaf RA, Dundee JW, Gamble JA. The influence of the route of administration on the clinical action of diazepam. Anaesthesia 1975 Mar;30(2):152‐8. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221324
- Assaf RA, Dundee JW, Bali IM. Studies of drugs given before anaesthesia XXV: Medazepam, a new benzodiazepine. British Journal of Anaesthesia 1975 Apr;47(4):464‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221326
- Badalaty MM, Houpt MI, Koenigsberg SR, Maxwell KC, DesJardins PJ. A comparison of chloral hydrate and diazepam sedation in young children. Pediatric Dentistry 1990 Feb;12(1):33‐7. [; MEDLINE: ] [PubMed] [Google Scholar]; 3221328
- Beer GM, Spicher I, Seifert B, Emanuel B, Kompatscher P, Meyer VE. Oral premedication for operations on the face under local anesthesia: A placebo‐controlled double‐blind trial. Plastic Reconstronstructive Surgery 2001 Sep;108(3):637‐43. [; MEDLINE: ; ISI:000170598700006 ER] [DOI] [PubMed] [Google Scholar]; 3221330
- Benhamou D, Thorin D, Brichant JF, Dailland P, Milon D, Schneider M. Intrathecal clonidine and fentanyl with hyperbaric bupivacaine improves analgesia during cesarean section. Anesthesia & Analgesia 1998 Sep;87(3):609‐13. [; MEDLINE: ; ISI:000075621200022 ER] [DOI] [PubMed] [Google Scholar]; 3221332
- Boone J, Gao G, Clark C, Chaudhuri S, Chaudhuri K. Dolasetron for prevention of perioperative nausea and vomiting during cesarean section under regional anesthesia. Anesthesiology 2002;96 Suppl:A1069. [; CN‐00420652] [Google Scholar]; 3221334
- Calamandrei M, Andreuccetti T, Crescioli M, Sarti A, Sestini G, Busoni P. Effect of ondansetron and metoclopramide on postoperative nausea and vomiting in children with epidural anaesthesia [Effetti di ondansetron e metoclopramide sulla nausea e il vomito postoperatorio in bambini in anestesia epidurale]. Minerva Anestesiologica 1994;60(suppl):37‐40. [] 8208450 [Google Scholar]; 3221336
- Campbell DN, Lewis IH, Barrowcliffe MP. Comparison of the effects of preoperative nabilone or metoclopramide on postoperative nausea and vomiting. British Journal of Anaesthesia 1993 Aug;71:305‐6. [; ISI:A1993LR04500029 ER] [DOI] [PubMed] [Google Scholar]; 3221338
- Capouet V, Pauw C, Ivens D, Ickx B, Hulstaert F. Single dose intravenous tropisetron in the prevention of post‐operative nausea and vomiting following gynaecological surgery: a double‐blind, multicentre, dose‐comparison study. European Journal of Anaesthesiology 1996;13:191‐2. [; CN‐00258460] [Google Scholar]; 3221340
- Cavanaugh JJ. Prophylaxis of postoperative nausea and vomiting: A clinical comparison of ondansetron and droperidol. Journal of the American Association of Nurse Anesthetists 1996;64(5):429‐30. [; EMBASE 1996345117] [Google Scholar]; 3221342
- Chalmers P, Horton JN. Oral bromazepam in premedication. A comparison with diazepam. Anaesthesia 1984 Apr;39(4):370‐2. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221344
- Chestnut DH, Owen CL, Geiger M, Bates JN, Choi WW, Ostman PL. Metoclopramide versus droperidol for prevention of nausea and vomiting during epidural anesthesia for cesarean section. Southern medical journal 1989 Oct;82(10):1224‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221346
- Cieslak GD, Watcha MF, Phillips M, Pennant JH. The dose‐response relationship of granisetron in the prophylaxis of pediatric postoperative emesis. Anesthesiology 1995 Sep;83:A1161. [; ISI:A1995RX68501161 ER] [DOI] [PubMed] [Google Scholar]; 3221348
- Cole WH. Midazolam in paediatric anaesthesia. Anaesthesia & Intensive Care 1982 Feb;10(1):36‐9. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221350
- Cook‐Sather SD, Harris K, Schreiner MS. Cisapride does not prevent postoperative emesis in children. Anesthesiology 1998 Sep;9:A1284. [; ISI:000075810901278 ER] [Google Scholar]; 3221352
- Costas CD, Coronado Quesada E, Rubio H. Simple‐blind clinical study with sublingual lorazepam vs injectable diazepam as premedication in colonoscopies. Acta gastroenterologica latinoamericana 1985;15(1):43‐8. [; LILACS Id: 1599] [PubMed] [Google Scholar]; 3221354
- Cramb RJ, Fargas‐Babjak A. Intraoperative prochlorperazine for prevention of postoperative nausea and vomiting. Canadian‐Journal‐of‐Anaesthesia 1988;35(3 II):S64‐S65. [; AN: 1988130833] [DOI] [PubMed] [Google Scholar]; 3221356
- Cugini U, Lanzetta P, Nadbath P, Menchini U. Sedation with ketamine during cataract surgery. Journal of Cataract & Refractive Surgery 1997 Jun;23(5):784‐6. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221358
- Danzer BI, Birnbach DJ, Stein DJ, Kuroda MM, Thys DM. Does metoclopramide supplement postoperative analgesia using patient‐ controlled analgesia with morphine in patients undergoing elective cesarean delivery?. Regional Anesthesia 1997 Sep‐Oct;22(5):424‐7. [; MEDLINE: ; AN: 1997303323] [DOI] [PubMed] [Google Scholar]; 3221360
- Oliveira Filho GR, Teixeira Filho N, Pederneiras SG, Fonseca MHZ. Effects of previous administration of metoclopramide, droperidol or ondansetron on hemodynamics and hypnotic dose of propofol [Efeitos da administracao previa de metoclopramida, droperidol ou ondansetron sobre a hemodinamica e a dose hipnotica de propofol]. Revista Brasileira de Anestesiologia 2000;50(3):235‐8. [; AN: 2000196070] [Google Scholar]; 3221362
- Dershwitz M, Rosow CE, Dibiase PM, Joslyn AF, Sanderson PE. Prophylaxis of postoperative vomiting by ondansetron. Clinical Pharmacology and Therapeutics 1991;49(2):184. [; ISI:A1991EZ62000244 ER] [DOI] [PubMed] [Google Scholar]; 3221364
- Dershwitz M, Rosow CE, Biase PM, Joslyn AF, Sanderson PE. Ondansetron is effective in decreasing postoperative nausea and vomiting. Clinical Pharmacology and Therapeutics 1992 Jul;52(1):96‐101. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221366
- Dershwitz M, Conant JA, Rosow CE, Connors PM, Zaslavsky A. A dose‐response study of ondansetron in preventing postoperative nausea and vomiting in female inpatients. Anesthesiology 1996 Sep;85:A331. [; ISI:A1996VM46600331 ER] [Google Scholar]; 3221368
- Diemunsch P, Wilson J, Chitour K, Patterson J, Kong KL. A double‐blind, parallel‐group, placebo‐controlled dose‐ranging study of iv granisetron in the prevention of postoperative nausea and vomiting (ponv). Anesthesiology 1994 Sep;81:A1280. [; MEDLINE: ] [Google Scholar]; 3221370
- Diemunsch P, Leeser J, Helmers JH, Wilkey B, Radke J, Nave S, et al. Oral dolasetron mesylate for prevention of postoperative nausea and vomiting (PONV). Anesthesiology 1996 Sep;85:A3. [; ISI:A1996VM46600003ER] [DOI] [PubMed] [Google Scholar]; 3221372
- Diemunsch P, Schoeffler P, Bryssine B, Cheli‐Muller LE, Lees J, McQuade BA, et al. Antiemetic activity of the NK1 receptor antagonist GR205171 in the treatment of established postoperative nausea and vomiting after major gynaecological surgery. British Journal of Anaesthesia 1999 Feb;82(2):274‐6. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221374
- Dundee JW, Halliday F, Nicholl RM, Moore J. Studies of drugs given before anaesthesia. X. Two non‐phenothiazine anti‐emetics‐‐cyclizine and trimethobenzamide. British Journal of Anaesthesia 1966 Jan;38(1):50‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221376
- Dundee JW, Clarke RS, Howard PJ. Studies of drugs given before anaesthesia. XXII: Metoclopramide. British Journal of Anaesthesia 1974 Jul;46(7):509‐13. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221378
- Fabling JM, Guy J, Borel CO, Warner DS, Gan TJ. Ondansetron, droperidol, placebo: Prospective, randomized, double‐blind comparison of postoperative nausea & vomiting in supra‐tentorial craniotomy. Anesthesiology 1999 Sep;91(3A):A194. [; ISI:000082480600194 ER] [Google Scholar]; 3221380
- Fry EN. Metoclopramide and drinking before general anaesthesia. Anaesthesia 1974 Nov;29(6):754‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221382
- Fujii Y, Tanaka H, Toyooka H. Granisetron prevents nausea and vomiting during spinal anaesthesia for caesarean section. Acta Anaesthesiologica Scandinavica 1998 Mar;42(3):312‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221384
- Furst SR, Rodarte A, Demars P. Ondansetron reduces postoperative vomiting in children undergoing tonsillectomy. Anesthesiology 1993;79(3A):A1197. [; ISI:A1993LY10801193 ER] [Google Scholar]; 3221386
- Gackle H, Wagner P, Eberhart LH, Georgieff M, Lang GE, Lang GK. Prevention of postoperative nausea and vomiting after vitreo‐ retinal surgery under general anesthesia. Investigative ophthalmology & visual science 1999 Mar;40:4968B771. [; SI:000079269204964 ER] [Google Scholar]; 3221388
- Gan TJ, Collis R, Hetreed M. Ondansetron, droperidol and saline for the prevention of postoperative nausea and vomiting after major orthopaedic surgery. A double‐blind controlled study. British journal of anaesthesia 1993 May;71:768P. [; ISI:A1993LD22600013 ER] [DOI] [PubMed] [Google Scholar]; 3221390
- Gan TJ, Alexander R, Fennelly M, Rubin AP. Comparison of different methods of administering droperidol in pca morphine in the prevention of postoperative nausea and vomiting. Anesthesia & Analgesia 1994 Feb;78(2):U77. [; ISI:A1994NA40600126 ER] [DOI] [PubMed] [Google Scholar]; 3221392
- Gan TJ, Ginsberg B, Grant AP, Glass PSA. Double‐blind, randomized comparison of ondansetron and intraoperative propofol to prevent postoperative nausea and vomiting. Anesthesiology 1996;85(5):1036‐42. [; AN: 1996344318] [DOI] [PubMed] [Google Scholar]; 3221394
- Gan TJ, Fortney J, Parasuraman TV, Creed M, Graczyk S, Glass PSA. Patient satisfaction and pharmacoeconomic comparison of ondansetron vs. droperidol for the prevention of postoperative nausea and vomiting in ambulatory surgical patients. Anesthesiology 1997 Sep;87:A53. [; SI:A1997XV63600053 ER] [Google Scholar]; 3221396
- Gesztesi ZS, Song D, White PF. Comparison of a new NK‐1 antagonist (CP122,721) to ondansetron in the prevention of postoperative nausea and vomiting. Anesthesia & Analgesia 1998 Feb;86:s32. [; ISI:000071851500033 ER] [Google Scholar]; 3221398
- Ghaly RG, Fitzpatrick KT, Dundee JW. Antiemetic studies with traditional Chinese acupuncture. A comparison of manual needling with electrical stimulation and commonly used antiemetics. Anaesthesia 1987 Oct;42(10):1108‐10. [; MEDLINE: ; CN‐00210378] [DOI] [PubMed] [Google Scholar]; 3221400
- Gift AG, Stanik J, Karpenick J, Whitmore K, Bolgiano CS. Oxygen‐saturation in postoperative‐patients at low‐risk for hypoxemia ‐ is oxygen‐therapy needed. Anesthesia & Analgesia 1995 Feb;80(2):368‐72. [; MEDLINE: ; ISI:A1995QD73400028 ER] [DOI] [PubMed] [Google Scholar]; 3221402
- Graczyk SG, McKenzie R, Kallar S, Hickok CB, Melson T, Morrill B, et al. Intravenous dolasetron for the prevention of postoperative nausea and vomiting after outpatient laparoscopic gynecologic surgery. Anesthesia & Analgesia 1997 Feb;84(2):325‐30. [; MEDLINE: ; ISI:A1997WF15900015 ER] [DOI] [PubMed] [Google Scholar]; 3221404
- Grasela TH, McLeskey CH, Walawander MA, Kopman AF, Nabulsi A, Roizen MF, et al. Postoperative nausea and vomiting and the effect of anticholinesterase agents. Anesthesiology 1994 Sep;81:A1278. [; ISI:A1994PJ09101277 ER] [Google Scholar]; 3221406
- Gratz I, Allen E, Afshar M, Joslyn AF, Buxbaum J, Prilliman B. The effects of the menstrual cycle on the incidence of emesis and efficacy of ondansetron. Anesthesia & Analgesia 1996 Sep;83(3):565‐9. [; MEDLINE: ; EMBASE 1996274171] [DOI] [PubMed] [Google Scholar]; 3221408
- Gulhas N, Turkoz A, Bayramlar H, Durmus M, Gedik E, Daglioglu MC, et al. The effect of oral clonidine on postoperative nausea and vomiting in children undergoing strabismus surgery [Pediyatrik hastalarda sas< iota >l< iota >k cerrahisi oncesi oral klonidinin postoperatif bulant< iota > ve kusmaya etkisi]. Turk Anesteziyoloji ve Reanimasyon 2001;29(5):202‐5. [; AN: 2001253582] [Google Scholar]; 3221410
- Hamid SK, Selby IR, Sikich N, Lerman J. Prophylactic therapy for postoperative vomiting in children after adenotonsillectomy: A comparison of dimenhydrinate. Anesthesiology 1996 Sep;85:A1098. [; ISI:A1996VM46601098 ER] [Google Scholar]; 3221412
- Hannalah R, Davis PJ, Orr R, Ferrari L, Rimar S. Prophylactic administration of ondansetron effectively decreases postoperative emesis in children. Anesthesiology 1995 Sep;83:A1177. [; ISI:A1995RX68501177 ER] [Google Scholar]; 3221414
- Helmers JH. Oral ondansetron in the prevention of postoperative nausea and vomiting. European Journal of Anaesthesiology 1992 Nov;9(Suppl 6):49‐54. [; MEDLINE: ] [PubMed] [Google Scholar]; 3221416
- Hill RP, Lubarsky DA, Phillips‐Bute B, Fortney JT, Creed MR, Glass PSA, et al. Cost‐effectiveness of prophylactic antiemetic therapy with ondansetron, droperidol, or placebo. Anesthesiology 2000;92(4):958‐67. [; MEDLINE: ; AN: 2000121025] [DOI] [PubMed] [Google Scholar]; 3221418
- Houchin KW, Dunbar JA, Lingua RW. Reduction of postoperative emesis in medial rectus muscle surgery. Investigative ophthalmology & visual science 1992 Mar;33(4):1336. [; ISI:A1992HK13503203 ER] [Google Scholar]; 3221420
- Imbeloni LE, Santos JMM, Santos MM. Prophylactic use of metoclopramide for control of nausea and vomiting during epidural anesthesia for cesarean section [Uso profilático da metoclopramida no controle de náuseas e vômitos durante anestesia peridural para cesariana]. Revista Brasileira de Anestesiologia 1986 Jul;36(4):295‐8. [; LILACS Id: 38165] [Google Scholar]; 3221422
- Jakobsson J, Andersson L, Nilsson A, Davidson S, Askergren J. Premedication before elective breast surgery, a comparison between ketobemidone and midazolam. Acta Anaesthesiologica Scandinavica 1991 Aug;35(6):524‐8. [; MEDLINE: ; ISI:A1991FX80800012 ER] [DOI] [PubMed] [Google Scholar]; 3221424
- Janknegt R, Arbouw MEL, Velden RW, Pinckaers J, Rohof MHC, Ausems MEM, et al. Prophylaxis of postoperative nausea and vomiting. Cost effectiveness of drug therapy [Kosteneffectiviteit van de medicamenteuze behandeling. Profylaxe van postoperatieve misselijkheid en braken]. Pharmaceutisch Weekblad 1999 Dec;134(50):1736‐41. [; AN: 2000001502] [Google Scholar]; 3221426
- Jellish WS, Leonetti JP, Fluder E, Thalji Z. Ondansetron versus droperidol or placebo to prevent nausea and vomiting after otologic surgery. Otolaryngology ‐ Head & Neck Surgery 1998 Jun;118(6):785‐9. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221428
- Kenny GN, Oates JD, Leeser J, Rowbotham DJ, Lip H, Rust M, et al. Efficacy of orally administered ondansetron in the prevention of postoperative nausea and vomiting: a dose ranging study. British Journal of Anaesthesia 1992 May;68(5):466‐70. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221430
- Khalil SN, Kataria B, Pearson K, Conahan T, Kallar S, Zahl K, et al. Ondansetron prevents postoperative nausea and vomiting in women outpatients. Anesthesia and Analgesia 1994 Nov;79(5):845‐51. [; MEDLINE: ; AN: 1994329343] [DOI] [PubMed] [Google Scholar]; 3221432
- Kivalo I, Rosenberg P. The antiemetic effect of dixyrazine in postoperative patients‐‐ a double‐blind study. Annales Chirurgiae et Gynaecologiae 1976;65(4):28‐9. [; MEDLINE: ] [PubMed] [Google Scholar]; 3221434
- Klamt JG, Slullitel A, Garcia V, Prado WA. Postoperative analgesic effect of intrathecal neostigmine and its influence on spinal anaesthesia. Anaesthesia 1997 Jun;52(6):547‐51. [; MEDLINE: ; ISI: 0003‐2409] [DOI] [PubMed] [Google Scholar]; 3221436
- Kostopanagiotou G, Pouriezis T, Theodoraki K, Kottis G, Andreadou I, Smyrniotis V, et al. Influence of ondansetron on thiopental hypnotic requirements. Journal of Clinical Pharmacology 1998 Sep;38(9):825‐9. [; MEDLINE: ] [PubMed] [Google Scholar]; 3221438
- Kovac A, McKenzie R, O'Connor T, Duncalf D, Angel J, Gratz I, et al. Prophylactic intravenous ondansetron in female outpatients undergoing gynaecological surgery: a multicentre dose‐comparison study. European Journal of Anaesthesiology 1992 Nov;9(Suppl 6):37‐47. [; MEDLINE: ] [PubMed] [Google Scholar]; 3221440
- Kovac A, Chelly J, McKenzie R, Philip B, Pearman M. Multicenter intravenous dose response trial to assess the efficacy and safety of dolasetron mesylate in preventing postoperative nausea and vomiting. Anesthesiology 1996 Sep;85:A1. [; SI:A1996VM46600001 ER] 8870607 [Google Scholar]; 3221442
- Lawhorn CD, Brown RE, Schmitz ML, Vollers JM, Huggins DP, Kymer PJ, et al. Ondansetron dose response curve in pediatric T&A and strabismus patients with and without oral metoclopramide premedicant. Anesthesiology 1994 Sep;81(3A):A1354. [] [Google Scholar]; 3221444
- Lindblad T, Beattie WS, Forrest JB, Buckley DN. Loss of the antiemetic effect of droperidol in menstruating women. Canadian Journal of Anaesthesia 1990 May;37(4 Pt 2):S139. [; MEDLINE: ] [PubMed] [Google Scholar]; 3221446
- Liu K, Hsu CC, Chia YY. Effect of dexamethasone on postoperative emesis and pain. British Journal of Anaesthesia 1998 Jan;80(1):85‐6. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221448
- Loers JF. [Ketamine‐dehydrobenzperidol and ketamine‐diazepam combinations for anaesthesia in neuro‐radiology]. [German] [Ketamine‐Dehydrobenzperidol‐ und Ketamine‐ Diazepam‐Kombinationsnarkose in der Neuroradiologie]. Anaesthesist 1973;22(3):127‐30. [; MEDLINE: ; CN‐00353264] [PubMed] [Google Scholar]; 3221450
- Loper K, Dorman B, Ready LB. Evaluation of transdermal scopolamine for the reduction of nausea associated with epidural morphine in postoperative patients. Canadian Journal of Anaesthesia 1988;35(3 II):S65‐S66. [; AN: 1988130835] [Google Scholar]; 3221452
- Lopez Olaondo L, Pueyo J, Carrascosa F, Iribarren MJ, Panadero A, Saiz N. Combination ondansetron and dexamethasone in the prophylaxis of postoperative nausea and vomiting. British Journal of Anaesthesia 1995 May;74:90 (A296). [; ISI:A1995RA77700295 ER] [DOI] [PubMed] [Google Scholar]; 3221454
- Lopez Herrera G, Solis Soriano FJ. The efficacy of ondansetron versus tropisetron as antiemetics in the postoperatory of laparoscopic surgery [Valoracion del ondansetron versus tropisetron en cirugia laparoscopica como tratamiento antiemetico]. Revista Mexicana de Anestesiologia 2000;23(2):89‐93. [; AN: 2000249426] [Google Scholar]; 3221456
- Loughrey JPR, Walsh F, Gardiner J. Prophylactic intravenous bolus ephedrine for elective Caesarean section under spinal anaesthesia. European Journal of Anaesthesiology 2002 Jan;19(1):63‐8. [; MEDLINE: ; IS: 0265‐0215] [DOI] [PubMed] [Google Scholar]; 3221458
- Lussos SA, Bader AM, Thornhill ML, Datta S. The antiemetic efficacy and safety of prophylactic metoclopramide for elective cesarean delivery during spinal anesthesia. Regional Anesthesia 1992 May‐Jun;17(3):126‐30. [; MEDLINE: ] [PubMed] [Google Scholar]; 3221460
- Manchikanti L, Marrero TC, Roush JR. Preanesthetic cimetidine and metoclopramide for acid aspiration prophylaxis in elective surgery. Anesthesiology 1984 Jul;61(1):48‐54. [; MEDLINE: ] [PubMed] [Google Scholar]; 3221462
- Maranhäo MVM, Coelho VV, Ivo CMA, Maranhäo MHC, Amaral EB. A comparative study between metoclopramide and droperidol in the control of nauses and vomit in patients subjected to spinal blockade during cesarean section [Estudo comparativo entre a metoclopramida e o droperidol no controle de náuseas e vômitos na operaçäo cesariana em gestantes submetidas a bloqueio subaracnóideo]. Revista Brasileira de Anestesiologia 1988;38(4):245‐9. [; Id: 76897] [Google Scholar]; 3221464
- Martins AD, Paula AM, Polisseni F, Guttmayer E, Oliveira PA. Use of bromopride on nauseas and vomiting due to gynecologic surgery [Uso de bromopride nas nauseas e vomitos do pos‐operatorio de cirurgia ginecologica]. Jornal brasileiro ginecologia 1981;91(5):351‐2. [; LILACS Id: 5638] [Google Scholar]; 3221466
- Mattila MA, Suurinkeroinen S, Saila K, Himberg JJ. Midazolam and fat‐emulsion diazepam as intramuscular premedication. A double‐blind clinical trial. Acta Anaesthesiologica Scandinavica 1983 Oct;27(5):345‐8. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221468
- McKenzie R, Kovac A, O'Connor T, Duncalf D, Angel J, Gratz I, Tolpin E, McLeskey C, Joslyn A. Comparison of ondansetron versus placebo to prevent postoperative nausea and vomiting in women undergoing ambulatory gynecologic surgery. Anesthesiology 1993;78(1):21‐8. [; MEDLINE: ; AN: 1993038974] [DOI] [PubMed] [Google Scholar]; 3221470
- Michaloudis D, O'Keeffe N, O'Sullivan K, Healy TE. Postoperative nausea and vomiting: a comparison of anti‐emetic drugs used alone or in combination. Journal of the Royal Society of Medicine 1993 Mar;86(3):137‐8. [; MEDLINE: ] [DOI] [PMC free article] [PubMed] [Google Scholar]; 3221472
- Mirakhur RK, Dundee JW. Lack of anti‐emetic effect of glycopyrrolate. Anaesthesia 1981 Aug;36(8):819‐21. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221474
- Morrison JD, Clarke RS, Dundee JW. Studies of drugs given before anaesthesia. XXI. Droperidol. British Journal of Anaesthesia 1970 Aug;42(8):730‐5. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221476
- Nelskyla K, Yli‐hankala A, Soikkeli A, Korttila K. Neostigmine does not increase incidence or severity of postoperative nausea and vomiting in outpatients undergoing gynecological laparoscopy. Anesthesiology 1998 Sep;89:A2. [; SI:000075810900003 ER] [DOI] [PubMed] [Google Scholar]; 3221478
- Nortcliffe SA, Buggy DJ. Prophylaxis of nausea and vomiting after spinal morphine analgesia for caesarean section: comparison between cyclizine, dexamethasone, and placebo. British Journal of Anaesthesia 2001;87(4):657P. [; CN‐00370465] [DOI] [PubMed] [Google Scholar]; 3221480
- Okamoto GU, Duperon DF, Jedrychowski JR. Clinical evaluation of the effects of ketamine sedation on pediatric dental patients. Journal of clinical pediatric dentistry 1992;16(4):253‐7. [; MEDLINE: ] [PubMed] [Google Scholar]; 3221482
- Olsson GL, Hallen B. Pharmacological evacuation of the stomach with metoclopramide. Acta Anaesthesiologica Scandinavica 1982 Oct;26(5):417‐20. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221484
- Olvera DJH, Toranzo FJM, Beltrán SB. A comparative study between dexamethasone ibuprofen and betamethasone for the control of postoperative swelling after impacted third molars surgery [Estudio comparativo entre ibuprofeno dexametasona y betametasona en el control postoperatorio en cirugía de terceros molares retenidos]. Revista ADM: organo oficial de la Asociacion Dental Mexicana 1997 April;54(2):83‐7. [; MEDLINE: ] [Google Scholar]; 3221486
- Pan PH, Moore CH. Intraoperative antiemetic efficacy of prophylactic ondansetron versus droperidol for cesarean section patients under epidural anesthesia. Anesthesia & Analgesia 1996 Nov;83(5):982‐6. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221488
- Parlow JL, Meikle AT, Vlymen J, Avery N. Post discharge nausea and vomiting after ambulatory laparoscopy is not reduced by promethazine prophylaxis. Canadian Journal of Anaesthesia 1999 Aug;46(8):719‐24. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221490
- Polati E, Verlato G, Finco G, Mosaner W, Grosso S, Gottin L, Pinaroli AM, Ischia S. Ondansetron versus metoclopramide in the treatment of postoperative nausea and vomiting. Anesthesia & Analgesia 1997 Aug;85(2):395‐9. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221492
- Powell RM, Buggy DJ. Ondansetron given before induction of anesthesia reduces shivering after general anesthesia. Anesthesia & Analgesia 2000 Jun;90(6):1423‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221494
- Pueyo FJ, Lopez Olaondo L, Sanchez Ledesma MJ, Ortega A, Carrascosa F. Cost‐effectiveness of three combinations of antiemetics in the prevention of postoperative nausea and vomiting. British Journal of Anaesthesia 2003 Oct;91(4):589‐92. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221496
- Ram D, Mamber E, Chosack A, Fuks AB. The effect of metoclopramide and hydroxyzine in sedation of infants undergoing dental treatment. ASDC Journal of Dentistry for Children 1999 Jan‐Feb;66(1):49‐52. [; MEDLINE: ] [PubMed] [Google Scholar]; 3221498
- Rose J, Dooley BS, Martin T. A comparison of the antiemetic efficacy of one or two doses of metoclopramide or ondansetron in children undergoing tonsillectomy. Anesthesiology 1994 Sep;81(3A):A1352. [] [Google Scholar]; 3221500
- Rust M. Intravenous ondansetron versus metoclopramide for the prophylaxis of postoperative nausea and vomiting. Anaesthesist 1995;44(4):288‐90. [; EMBASE 1995149313] [DOI] [PubMed] [Google Scholar]; 3221502
- Sadove MS, Hatano S, Redlin T, Thomason R, Arastounejad P, Roman V. Clinical study of droperidol in the prevention of the side‐effects of ketamine anesthesia: a progress report. Anesthesia & Analgesia 1971 Jul‐Aug;50(4):526‐32. [; MEDLINE: ] [PubMed] [Google Scholar]; 3221504
- Santos A, Datta S. Prophylactic use of droperidol for control of nausea and vomiting during spinal anesthesia for cesarean section. Anesthesia & Analgesia 1984 Jan;63(1):85‐7. [; MEDLINE: ] [PubMed] [Google Scholar]; 3221506
- Scholz J, Hennes HJ, Schweiger C, Farber L. Tropisetron or ondansetron vs placebo for prevention of postoperative nausea and vomiting after abdominal and non‐ abdominal surgery. Anesthesiology 1996 Sep;85:A330. [; ISI:A1996VM46600330 ER] [Google Scholar]; 3221508
- Scuderi PE, Weaver RG, Mims GR, Weeks DB, James RL, McKinley AC. A comparison of droperidol, ondansetron and metoclopramide for the prevention of vomiting following strabismus surgery in children. Anesthesiology 1994 Sep;81:A24. [] [DOI] [PubMed] [Google Scholar]; 3221510
- Scuderi P, James R, Harris L, Mims G, Weeks D. Comparison of prophylaxis versus treatment of postoperative nausea and vomiting: Effect on outcome. Anesthesiology 1997 Sep;87:A48. [; SI:A1997XV63600048 ER] [Google Scholar]; 3221512
- Sen A, Rudra A, Sarkar SK, Biswas B. Intrathecal midazolam for postoperative pain relief in caesarean section delivery. Journal of the Indian Medical Association 2001 Dec;99(12):683‐4. [; MEDLINE: ; EMBASE 2002145924] [PubMed] [Google Scholar]; 3221514
- Senders CW, Mauro SM, Brodie HA, Emery BE, Sykes JM. The efficacy of perioperative steroid therapy in pediatric primary palatoplasty. Cleft palate ‐ craniofacial journal 1999 Jul;36(4):340‐4. [; AN: 1999065819] [DOI] [PubMed] [Google Scholar]; 3221516
- Sjovall S, Kanto J, Iisalo E, Kangas l, Mansikka M, Phlajamaki K. Use of atropine in connection with oral midazolam premedication. International Journal of Clinical Pharmacology Therapeutics and Toxicology 1984 Apr;22(4):184‐8. [; MEDLINE: ; EMBASE 1934102239] [PubMed] [Google Scholar]; 3221518
- Spelina KR, Gerber HR, Pagels IL. Nausea and vomiting during spinal anaesthesia. Effect of metoclopramide and domperidone: a double‐blind trial. Anaesthesia 1984 Feb;39(2):132‐7. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221520
- Tan TSH, Safavi F, Watcha MF, Bala A, White PF. Effect of antagonism of mivacurium induced neuromuscular blockade on postoperative emesis in children. Anesthesiology 1993;79(3A):A1123. [; ISI:A1993LY10801119 ER] [Google Scholar]; 3221522
- Tang J, Sun R, White PF. Clinical efficacy of prophylactic antiemetics for outpatients at high‐risk of postoperative emesis. Anesthesiology 1995 Sep;83:A9. [; SI:A1995RX68500009 ER] [Google Scholar]; 3221524
- Tang J, Wang B, Qi J, Wender RH, White PF. Effect of the timing of ondansetron administration on postoperative nausea and vomiting after outpatient laparoscopy. Anesthesiology 1996 Sep;85:A9. [; ISI:A1996VM46600009 ER] [Google Scholar]; 3221526
- Tang J, Angelo RD, White PF, Scuderi PE. Efficacy of RS‐25259, a novel 5‐HT3 antagonist, in the prevention of postoperative nausea and vomiting after major gynecologic surgery. Anesthesiology 1997 Sep;87:A329. [; ISI:A1997XV63600329 ER] [Google Scholar]; 3221528
- Thune A, Appelgren L, Haglind E. Prevention of postoperative nausea and vomiting after laparoscopic cholecystectomy. A prospective randomised study of metoclopramide and transdermal hyoscine. European Journal of Surgery Acta Chirurgica 1995;161(4):265‐8. [; MEDLINE: ; EMBASE 1995256343] [PubMed] [Google Scholar]; 3221530
- Tolksdorf W, Eick C. Rectal, oral, and nasal premedication with midazolam in children aged 1‐6 years. Anaesthesist 1991 Dec;40(12):661‐7. [; MEDLINE: ; ISI:A1991GV44000002 ER] [PubMed] [Google Scholar]; 3221532
- Tramer M, Borgeat A, Rifat K. Postoperative nausea and vomiting after strabismus surgery in children ‐ effects of propofol, ondansetron and lidocaine. Anesthesiology 1993;79(3A):A1193. [; ISI:A1993LY10801189 ER] [Google Scholar]; 3221534
- Trapp LD. An evaluation of Droperidol for preventing nausea and vomiting after deep intravenous sedation for ambulatory dental surgery. Anesthesia progress 1989 Jan‐Feb;36(1):9‐12. [; MEDLINE: ] [PMC free article] [PubMed] [Google Scholar]; 3221536
- Ummenhofer W, Frei FJ, Kern C, Urwyler A, Drewe J. Ondansetron reduces postoperative nausea and vomiting in children. Anesthesiology 1993;79(3A):A1192. [; ISI:A1993LY10801188 ER] [DOI] [PubMed] [Google Scholar]; 3221538
- Unlugenc H, Guler T, Gunes Y, Isik G. Comparative study of the antiemetic efficacy of ondansetron, propofol and midazolam in the early postoperative period. European Journal of Anaesthesiology 2003 Aug;20(8):668‐73. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221540
- Ure D, James KS, McNeill M, Booth JV. Glycopyrrolate reduces nausea during spinal anaesthesia for caesarean section without affecting neonatal outcome. British Journal of Anaesthesia 1999 Feb;82(2):277‐9. [; MEDLINE: ; CN‐00344276] [DOI] [PubMed] [Google Scholar]; 3221542
- Vener DF, Carr AS, Sikich N, Bissonette B, Lerman J. Does dimenhydrinate control vomiting in children after outpatient strabismus surgery?. Anesthesiology 1994 SEP;81(3A):A21. [] [Google Scholar]; 3221544
- Wallenborn J, Rudolph C, Gelbrich G, Goerlich TM, Dohnert J, Dorner J, et al. Metoclopramide and dexamethasone in prevention of postoperative nausea and vomiting after inhalational anaesthesia. Anasthesiologie Intensivmedizin Notfallmedizin Schmerztherapie 2003 Nov;38(11):695‐704. [; MEDLINE: ] [DOI] [PubMed] [Google Scholar]; 3221546
- Warriner CB, Knox D, Belo S, Cole C, Finnegan B, Perreault L, et al. Prophylactic oral dolasetron mesylate reduces postoperative nausea and vomiting following abdominal hysterectomy. Anesthesiology 1995 Sep;83:A312. [; ISI:A1995RX68500312 ER] [Google Scholar]; 3221548
- Watcha MF, Bras PJ, Pennant J, Cieslak G, Burnette D. The dose‐response relationship of ondansetron in the prophylaxis of pediatric postoperative emesis. Anesthesiology 1994 Sep;81:A1350. [; ISI:A1994PJ09101349 ER] [Google Scholar]; 3221550
- Williams OA, Clarke FL, Harris RW, Smith P, Peacock JE. Addition of droperidol to patient‐controlled analgesia: Effect on nausea and vomiting. Journal of Clinical Anesthesia 1995 March;7(2):147. [; IS: 0952‐8180] [DOI] [PubMed] [Google Scholar]; 3221552
- Zarate E, Watcha M, White PF, Klein K. Comparative cost‐effectiveness of dolasetron and ondansetron for prophylaxis of postoperative nausea and vomiting after ambulatory surgery. Anesthesiology 1999 Sep;91(3):A3. [; ISI:000082480600003 ER] [Google Scholar]; 3221554
- Zomers PJW, Langenberg CJM, Bruijn KM. Tropisetron in the prevention of postoperative nausea and vomiting in gynaecological patients. Annals of Oncology 1993;4(SUPPL 3):S47. [; AN: 1993257587] [Google Scholar]; 3221556
Additional references
- Altman DG, Bland JM. Interaction revisited: the difference between two estimates. British Medical Journal 2003;326(7382):219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Apfel CC, Kranke P, Greim CA, Roewer N. Non‐systematic serial publishing is not appropriate and ethically questionable. Acta Anaesthesiologica Scandinivica 1999;43(4):486‐7. [MEDLINE: ] [PubMed] [Google Scholar]
- Apfel CC, Kranke P, Eberhart LHJ, Roos A, Roewer N. Comparison of predictive models for postoperative nausea and vomiting. British Journal of Anaesthesia 2002;88(2):234‐40. [DOI] [PubMed] [Google Scholar]
- Cohen MM, Duncan PG, DeBoer DP, Tweed WA. The postoperative interview: assessing risk factors for nausea and vomiting. Anesthesia and Analgesia 1994;78:7‐16. [DOI] [PubMed] [Google Scholar]
- Eberhart LH, Mauch M, Morin AM, Wulf H, Geldner G. Impact of a multimodal anti‐emetic prophylaxis on patient satisfaction in high‐risk patients for postoperative nausea and vomiting. Anaesthesia 2002 Oct;57(10):1022‐7. [MEDLINE: ] [DOI] [PubMed] [Google Scholar]
- Engoren M, Steffel C. Patient perception of monetary value to avoiding unpleasant side effects of anesthesia and surgery. Journal of Clinical Anesthesia 2000 Aug;12(5):388‐91. [MEDLINE: ] [DOI] [PubMed] [Google Scholar]
- Figueredo ED, Canosa LG. Ondansetron in the prophylaxis of postoperative vomiting: a meta‐analysis.. Journal of Clinical Anesthesia 1998 1998;10(3):211‐21. [DOI] [PubMed] [Google Scholar]
- Gan T, Sloan F, Dear Gde L, El‐Moalem HE, Lubarsky DA. How much are patients willing to pay to avoid postoperative nausea and vomiting?. Anesthesia and Analgesia 2001 Feb;92(3):393‐400. [MEDLINE: ] [DOI] [PubMed] [Google Scholar]
- Gupta A, Wu CL, Elkassabany N, Krug CE, Parker SD, Fleisher LA. Does the routine prophylactic use of antiemetics affect the incidence of postdischarge nausea and vomiting following ambulatory surgery: a systematic review of randomized controlled trials. Anesthesiology 2003 2003;99(2):488‐95. [DOI] [PubMed] [Google Scholar]
- Henzi I, Walder B, Tramèr MR. Metoclopramide in the prevention of postoperative nausea and vomiting: a quantitative systematic review of randomized, placebo‐controlled studies. British Journal of Anaesthesia 1999 1999;83(5):761‐71. [DOI] [PubMed] [Google Scholar]
- Henzi I, Sonderegger T, Tramèr M R. Efficacy, dose‐response, and adverse effects of droperidol for prevention of postoperative nausea and vomiting. 2.Henzi I, Sonderegger T, Tramèr M R. Efficacy, dose‐response, and adverse effects of droperidol for prevention of postoperative nausea and vomiting. Canadian Journal of Anesthesia 2000;47(6):537‐51.. Canadian Journal of Anesthesia 2000;47(6):537‐51. [DOI] [PubMed] [Google Scholar]
- Hirayama T, Ishii F, Yago K, Ogata H. Evaluation of the effective drugs for the prevention of nausea and vomiting induced by morphine used for postoperative pain: a quantitative systematic review. Yakugaku Zasshi. Journal of the Pharmaceutical Society of Japan 2001;121(2):179‐85. [DOI] [PubMed] [Google Scholar]
- Kao Lw, Kirk MA, Evers SJ, Rosenfeld SH. Droperidol, QT prolongation, and sudden death: what is the evidence?. Annals of emergency medicine 2003;41(4):546‐58. [MEDLINE: ] [DOI] [PubMed] [Google Scholar]
- Kapur PA. Editorial: The big "little problem". Anesthesia and Analgesia 1991;73:243‐5. [DOI] [PubMed] [Google Scholar]
- Kenny G. Risk factors for postoperative nausea and vomiting. Anaesthesia 1994;49(suppl):6‐10. [DOI] [PubMed] [Google Scholar]
- Kortilla K. The study of postoperative nausea and vomiting. British Journal of Anaesthesia 1992;69(suppl 1):20S‐23S. [DOI] [PubMed] [Google Scholar]
- Kranke P, Apfel CC, Greim CA, Roewer N. Methodological problems arising from 'serial publishing' on the effectiveness of granisetron in PONV. British Journal of Anaesthesia 1999;82(3):481‐3. [MEDLINE: ] [DOI] [PubMed] [Google Scholar]
- Kranke P, Apfel CC, Roewer N. Reported data on granisetron and postoperative nausea and vomiting by Fujii et al. Are incredibly nice!. Anesthesia and Analgesia2000; Vol. 90, issue 4:1004‐6. [MEDLINE: ] [PubMed]
- Kranke P, Apfel CC, Eberhart LH, Georgieff M, Roewer N. The influence of a dominating centre on a quantitative systematic review of granisetron for preventing postoperative nausea and vomiting. Acta Anaesthesiologica Scandinavica 2001;45(6):659‐70. [MEDLINE: ] [DOI] [PubMed] [Google Scholar]
- Rashiq S, Bray P. Relative value to surgical patients and anesthesia providers of selected anesthesia related outcomes. BMC Medical Informatics and Decision Making 2003 Feb;3:3. [MEDLINE: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager (RevMan) [Computer program]. Version 4.2 for Windows. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2003.
- Rüsch D, Eberhart L, Biedler A, Dethling J, Apfel CC. Prospective application of a simplified risk score to prevent postoperative nausea and vomiting. Canadian Journal of Anaesthesia 2005 2005;52(5):478‐84. [DOI] [PubMed] [Google Scholar]
- Steward DL, Welge JA, Myer CM. Steroids for improving recovery following tonsillectomy in children. .. The Cochrane Database of Systematic Reviews 2002;3:CD003997. [CD003997] [DOI] [PubMed] [Google Scholar]
- Tramèr MR, Reynolds DJ, Moore RA, McQuay HJ. Impact of covert duplicate publication on meta‐analysis: a case study. British Medical Journal 1997;315(7):635‐40. [MEDLINE: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tramèr M, Moore A, McQuay H. Prevention of vomiting after paediatric strabismus surgery: a systematic review using the numbers‐needed‐to‐treat method. British Journal of Anaesthesia 1995;75(5):556‐61. [DOI] [PubMed] [Google Scholar]
- Tramèr MR, Reynolds JM, Moore RA, McQuay HJ. Efficacy, dose‐response, and safety of ondansetron in prevention of postoperative nausea and vomiting: a quantitative systematic review of randomized placebo‐controlled trials. Anesthesiology 1997 1997;87(6):1277‐89. [DOI] [PubMed] [Google Scholar]
- Tramèr M R, Walder B. Efficacy and adverse effects of prophylactic antiemetics during patient‐controlled analgesia therapy: a quantitative systematic review. Anesthesia and Analgesia 1999;88(6):1354‐61. [DOI] [PubMed] [Google Scholar]
- Bosch JE, Kalkman CJ, Vergouwe Y, Klei WA, Bonsel GJ, Grobbee DE, et al. Assessing the applicability of scoring systems for predicting postoperative nausea and vomiting. Anaesthesia 2005;60:323‐31. [DOI] [PubMed] [Google Scholar]
- Watcha MF, White PF. Postoperative nausea and vomiting. Its etiology, treatment and prevention. Anesthesiology 1992;77:162‐84. [DOI] [PubMed] [Google Scholar]
- Watcha MF, White PF. New antiemetic drugs. International Journal of Clinical Anesthesiology. 1995;33:1‐20. [PubMed] [Google Scholar]







