Abstract
Hyperpolarization-activated cyclic nucleotide-gated (HCN) channels are membrane proteins encoded by four genes (HCN1–4) and widely distributed in the central and peripheral nervous system and in the heart. HCN channels are involved in several physiological functions, including the generation of rhythmic activity, and are considered important drug targets if compounds with isoform selectivity are developed. At present, however, few compounds are known, which are able to discriminate among HCN channel isoforms. The inclusion of the three-methylene chain of zatebradine into a cyclohexane ring gave a compound (3a) showing a 5-fold preference for HCN4 channels, and ability to selectively modulate Ih in different tissues. Compound 3a has been tested for its ability to reduce Ih and to interact with other ion channels in the heart and the central nervous system. Its preference for HCN4 channels makes this compound useful to elucidate the contribution of this isoform in the physiological and pathological processes involving hyperpolarization-activated current.
Keywords: Hyperpolarization-activated current, HCN channels, isoform selectivity, phenylalkylamines, zatebradine analogues, rigid analogues
Hyperpolarization-activated cyclic nucleotide-gated (HCN) channels are members of the superfamilies of voltage-dependent K+ (Kv) and cyclic nucleotide-gated (CNG) channels. The architecture of these proteins resembles that of voltage-gated potassium channels, showing a tetrameric arrangement, each subunit being composed of six transmembrane domains including the voltage sensor S4, and a pore loop between S5 and S6. At variance with Kv channels, the C-terminal intracellular portion harbors a Cyclic Nucleotide Binding Domain (CNBD). HCN channels carry a mixed Na+/K+ current known as hyperpolarization-activated current (Ih), are activated at hyperpolarizing voltages, and are modulated by cyclic nucleotides (cAMP, cGMP, and others). Four isoforms have been cloned (HCN1–4), differing in localization and biophysical properties.1−3
HCN channels are involved in various physiological processes in the central and peripheral nervous systems and in non-neuronal districts.1 For instance, HCN channels control heart pace in the sino-atrial node (SAN), where HCN4 is the most abundant isoform.4 In the central nervous system (CNS), HCN channels regulate several functions, among which are intrinsic excitability and rhythmogenesis. All four isoforms are expressed, to different extent and with different distribution: HCN1 and HCN2 are more widely distributed than HCN4, which is mainly located in the thalamus.5 In the retina, where HCN channels shape the light response, all four isoform are expressed.6 In DRG, HCN1 and HCN2 are the most abundant isoforms.7
HCN channels can be considered important drug targets for cardiac pathologies and neurological diseases such as pain and epilepsy,8,9 but the modulation of these channels has been exploited so far only at the cardiac level. Ivabradine (Figure 1), the only drug developed as a specific HCN channel blocker, has been approved as a bradycardic agent in chronic stable angina pectoris and to reduce hospitalization from worsening heart failure.10,11 In addition, several lines of evidence have suggested a role of HCN channels in pain perception and in epilepsy.9,12−14 Indeed, ivabradine is able to attenuate neuropathic and inflammatory pain15,16 and to increase the threshold for maximal electroshock-induced seizures.17 However, although ivabradine is well tolerated, its use for new therapeutic applications may be restricted due to its lack of selectivity since it blocks the four human HCN isoforms with similar potency.18 Several clinically used drugs have been reported to modulate Ih,19 with few of them showing some isoform selectivity, such as the anesthetic propofol for HCN120 or the anticonvulsant gabapentin for HCN4.21 However, these molecules are not specific for HCN channels and interact with other targets, while isoform-selective inhibitors could have a safer pharmacological profile due to the reduction of side effects.
Figure 1.
Structure of ivabradine, zatebradine, and analogues 1, 2, and 3a endowed with isoform preference or selectivity.
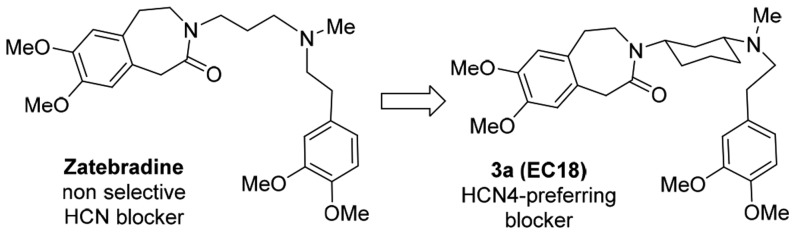
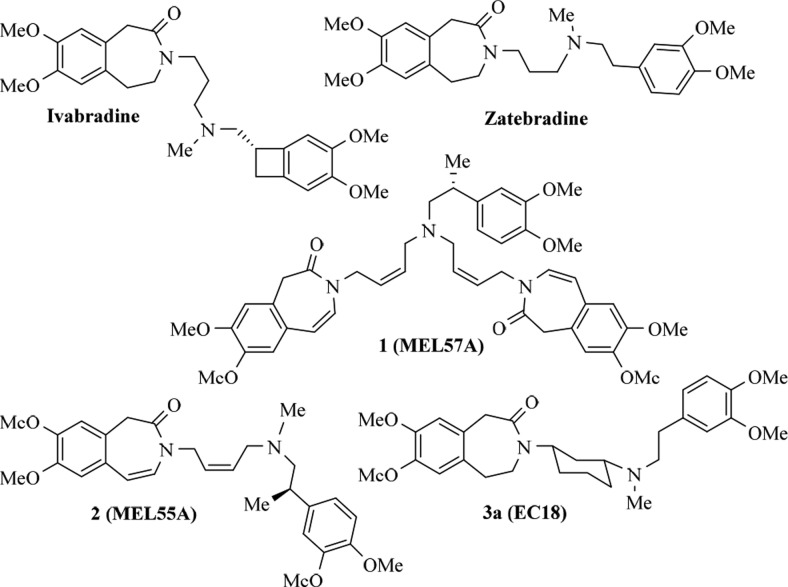
Recently some of us have described the design, synthesis, and characterization of phenylalkylamines endowed with isoform preference or selectivity (Figure 1). In HEK 293 cells stably expressing homomeric HCN1, HCN2, and HCN4 channels, MEL57A (1) selectively inhibited the HCN1 isoform.22,23 Importantly, this selectivity was also maintained in native tissues, where the channels may exist in the heteromeric form. Compound 1 was able to reduce Ih in DRG neurons at doses that did not influence heart rate;23 accordingly, 1 was effective in reducing hyperalgesia and allodynia in oxaliplatin-treated rats without cardiac effects.24 Compound 2 (MEL55A) is an example of isoform-preferring compound since it was shown to preferentially block HCN1 and HCN2 over HCN4;22 as its dimeric analogue 1, it was able to relieve chemotherapy-induced neuropathic pain.25 In this Letter we describe the design, synthesis, and characterization of 3a (EC18),23 a phenylalkylamine showing preference for HCN4 channels, and its usefulness to evaluate the physiological role of this isoform.
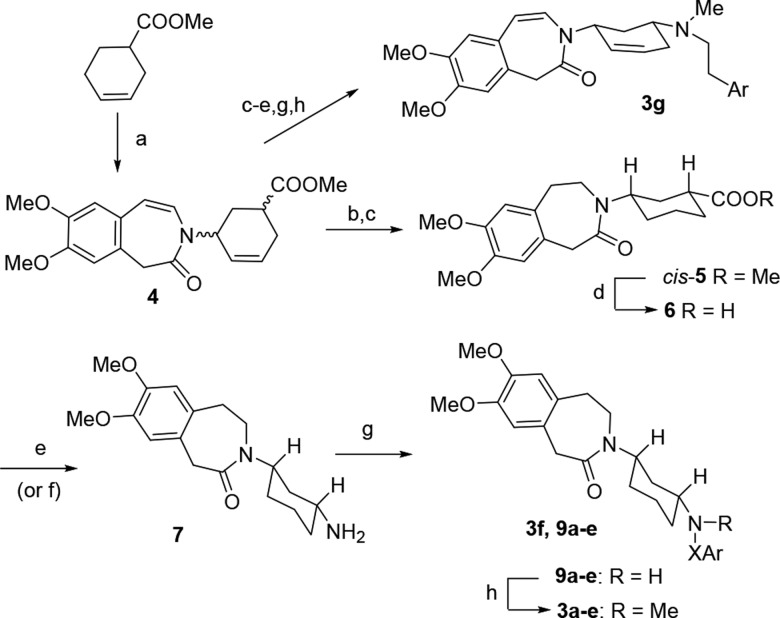
Compound 3a (EC18) represents a rigid analogue of zatebradine (Figure 1), the first “specific bradycardic agent” that entered clinical trials.19 The inclusion of the three-methylene chain into a cyclohexane ring sharply reduced zatebradine’s flexibility, as 3a is able to adopt different conformations only in the phenethyl moiety. By this way it was envisaged that some selectivity could be obtained; a similar modification made on verapamil, a structural analogue of zatebradine, caused a separation of vasodilator and cardiac depressant activities.26 A small set of cyclohexane derivatives (3a–f, see structure in Scheme 1 and Table 1), characterized by different spacers between the basic nitrogen and the dimethoxyphenyl ring, was designed and prepared. In addition, we thought it would be important to also synthesize 3g, the unsaturated analogue of 3a, since the presence of double bonds gave interesting properties to 1 and 2.
Scheme 1. Synthesis of Compounds 3a–g.
Reagents and conditions: (a) (1) NBS, AIBN, CCl4, heating; (2) t-BuOK, 7,8-dimethoxy-2,3-dihydro-1H-3-benzazepin-2-one; (b) H2/Pd/C; (c) chromatographic separation; (d) NaOH; (e) DPPA, toluene, then HCl; (f) (1) SOCl2, (2) NaN3, acetone; (3) Δ, toluene; (4) HCl; (g) arylalkyl halides 8a–e, Et3N; (h) CH2O, HCOOH. Ar = 3,4-dimethoxyphenyl. X = (CH2)2 (a); CH2 (b); (CH2)3 (c); (CH2)2O (d); CH2CH=CH (e); 3f, R = 3,4-dimethoxybenzyl, X = CH2.
Table 1. Percentage of Reduction of Ih at −80 mV after Application of Drugs at the Concentration of 5 μM on Human HCN4 Channel Expressed in HEK 293 Cellsa.
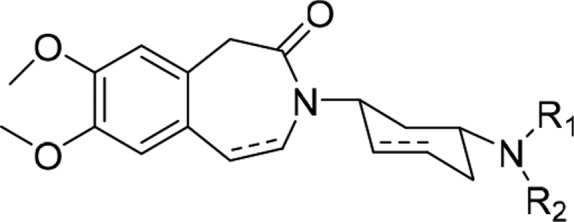
| N | R1 | R2 | Ih reduction (%) |
|---|---|---|---|
| 3a (EC18)b | Me | –CH2CH2Ar | 59 ± 14 (n = 5) |
| (+)-3a | Me | –CH2CH2Ar | 46 ± 7.7 (n = 3) |
| (−)-3a | Me | –CH2CH2Ar | 45 ± 20.1 (n = 4) |
| 3bb | Me | –CH2Ar | 21 ± 12 (n = 6) |
| 3cb | Me | –CH2CH2CH2Ar | 47 ± 16.1 (n = 4) |
| 3db | Me | –CH2CH2OAr | 48 ± 6.9 (n = 7) |
| 3eb | Me | –CH2CH=CHAr | 64 ± 8.0 (n = 4) |
| 3fb | CH2Ar | –CH2Ar | ∼4 (n = 4) |
| 3gc | Me | –CH2CH2Ar | 30 ± 11.6 (n = 4) |
| zatebradine | 59 ± 19 (n = 5)d |
Values are normalized to control and represent the mean ± SD of 3–7 experiments.
Single bond.
Double bond on both rings.
Obtained with a 10 μM concentration. Ar = 3,4-dimethoxyphenyl.
The synthesis of compounds 3a–g was performed as shown in Scheme 1. Following the procedure reported by Allan,27 methyl cyclohex-3-ene-1-carboxylate was reacted first with N-bromosuccinimide and then with the potassium salt of 7,8-dimethoxy-2,3-dihydro-1H-3-benzazepin-2-one,28 giving 4, which was hydrogenated on Pd/C yielding ester 5. This preparation was performed several times, obtaining 5 with similar yields (20–25%) and as mixtures of isomers, the most abundant one being the cis ester (85–95%), which was separated from the trans one by means of chromatography. The cis–trans configuration was assessed by means of NMR, as previously done for other cyclohexane derivatives:26 the width of the peak related to the cyclohexyl proton in position 1 was wider for the cis isomer (H axial) with respect to the trans one (H equatorial). The cis configuration was then confirmed by a NOESY experiment. Alkaline hydrolysis of cis-5 gave the corresponding acid 6, which was transformed into amine 7 through Curtius rearrangement of the acyl azide. This step was performed in two different ways. Treatment of the acid with DPPA (diphenyl phosphoryl azide) in toluene (method A), followed by acid hydrolysis of the intermediate isocyanate, gave 7 in about 50% yield. Alternatively, the acyl azide was prepared by reaction of the acyl chloride with sodium azide; heating in toluene and subsequent acid hydrolysis gave the amine 7 in slightly higher yields (method B, 60% yield). Reaction of 7 with the suitable arylalkyl halide (8a–e) gave secondary amines 9a–e, which were transformed into N-methyl-derivatives 3a–e by reaction with formaldehyde and formic acid; when using 3,4-dimethoxybenzyl bromide, some amount (4%) of double addition product 3f was obtained. Compound 4 was transformed into 3g using the same reaction sequence but avoiding catalytic hydrogenation.
As a preliminary screening, the compounds were tested at a 5 μM concentration to check their ability to block h-current on human HCN4 stably expressed in HEK 293 cells, at voltages (−80 mV) mimicking physiological membrane potential (Table 1). Under these conditions all the compounds were able to inhibit h-current, the majority of the compounds producing about 50% reduction. Representative traces are reported in Figure S1. The benzyl analogue 3b and the unsaturated derivative 3g were much less active, the extent of reduction being smaller (21% and 30%, respectively); the dibenzyl compound 3f was almost inactive. To understand the importance of chirality, the enantiomers of 3a were separated by means of HPLC (Figures S2 and S3), but no enantioselectivity was observed (Table 1). Although small, this set of compounds can suggest some structure–activity relationships: the insertion of two double bonds in the azepinone and cyclohexane rings is not productive, as well as the increase in size of both R1 and R2 substituents. On the contrary, the length and type of the spacer between the basic N atom and the dimethoxyphenyl moiety can be varied, still maintaining good potency.
Owing to its similarity with zatebradine, compound 3a (EC18) was selected for further characterization, which has been partially disclosed previously.23 This compound was tested on the homomeric HCN1, HCN2, and HCN4 channels expressed in HEK293 cells: it showed a 5-fold preference for HCN4 (EC50 3.98 ± 1.16 μM) over HCN1 (EC50 21 ± 3.98 μM) and HCN2 (EC50 19.35 ± 4.48 μM). Consistently with its good activity on HCN4, in guinea pig SAN 10 μM 3a reduced Ih by ∼67% at −120 mV, and on dog cardiac Purkinje fibers it reduced the amplitude and slowed the slope of diastolic depolarization phase.23 Moreover, on guinea-pig spontaneously beating isolated atria, it produced a negative chronotropic effect (IC50 9.9 ± 5.5 μM), with potency comparable to zatebradine (IC50 13.7 ± 8.7 μM).29
In line with its lower activity on HCN1 and HCN2 channels, on mouse DRG neurons 3a did not affect Ih conductance, at a physiologically relevant voltage, up to a concentration of 100 μM.23 Indeed, 3a was unable to reduce hypersensitivity when tested in a mouse model of oxaliplatin-induced neuropathy, a finding in line with the low expression of this isoform in DRG neurons.25
EC18 (3a) represents the first HCN4-preferring compound, discovered in a project aimed to find zatebradine analogues endowed with isoform selectivity.22,23 In fact, when tested on a recombinant HCN4 channel, EC18 showed a potency in the same range as ivabradine, but a 5-fold preference for this isoform with respect to HCN1 and HCN2, while ivabradine reduced h-current with the same potency on the three isoforms.18 Notably, the preference obtained at recombinant channels is maintained in native tissues, thus making 3a a tool to study the individual function of HCN4 channels in cardiac cells and neurons. Other HCN4-selective/preferring compounds have been described, such as the already mentioned gabapentin21 and amiodarone.30 However, both compounds have been developed for interaction with other targets, and their multi-ion channels blocking properties can obscure selectivity for HCN4 channels, even if the activity on HCN4 channel may contribute to their pharmacological effect.
Although a complete characterization of 3a on other channels or receptors has not been done yet, some experiments on cardiac tissue suggest that 3a also interacts with other ion channels. In fact, in dog ventricular papillary muscle at a stimulation cycle length of 1000 ms, 3a (10 μM) significantly and strongly inhibited the Vmax and increased conduction time (Figure 2, Table S1). Compound 3a moderately shortened the action potential (AP) duration at 1 μM, then prolonged the action potential duration at 10 μM.
Figure 2.
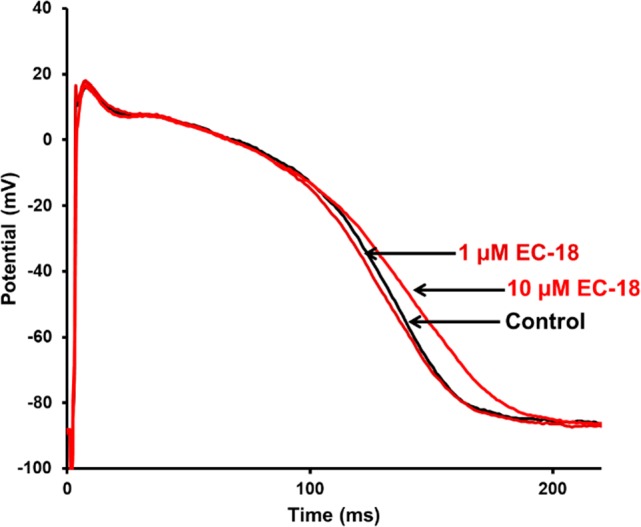
Effect of 3a (EC18) on the action potential repolarization in dog papillary muscle (n = 5) at stimulation cycle length of 1000 ms (representative action potential waveform recordings originate from the same preparation).
EC18 was found to have a cardiac electrophysiological profile similar to that of ivabradine in view of the ability to inhibit Vmax and to prolong repolarization at 10 μM concentration.31 The AP duration shortening exerted by 1 μM EC18 is probably attributable to Na+ channel blockade. The inhibitory effect of EC18 on Na+ channels might still exist at 10 μM, but regarding the effect of EC18 on the repolarization, the simultaneous K+ channel blockade could outweigh this AP shortening effect. Further studies are required for establishing the degree of the K+ current inhibition by EC18 in cardiomyocytes. Ivabradine, in addition to blocking the pacemaker current, was found able to inhibit hERG channels with a potency that is similar to that reported for HCN4 (see ref (1) and references therein). EC18 might have a similar action.
EC18 can be useful to study HCN channels not only in heart but also in the CNS. In the thalamus, a brain region mainly expressing HCN4 and HCN2 channels,33,34 compound 3a has been used to understand the contribution of the HCN4 isoform to Ih in small and large interneurons of the dorsal part of the lateral geniculate nucleus, which are classified based on differences in Ih current density. In these GABAergic neurons 30 μM 3a reduced Ih by about 31.4 ± 5.2%.35 As a further characterization, in this work we report the effect of 3a in thalamocortical relay (TC) neurons of the ventrobasal thalamic complex (VB), which belong to the principal thalamic neurons. Here, compound 3a revealed stronger effects with 30 μM blocking 91.3 ± 0.7% (n = 2) of Ih in these cells (Figure 3A). The construction of a dose–response relationship pointed to an EC50 value of around 11 μM (Figure 3B). These data suggest that in TC neurons Ih is mainly mediated by HCN4 or HCN2/4 heteromers and indicate that compound 3a is a valuable tool for analyzing the cell type-specific properties and function of HCN channels in native neurons.
Figure 3.
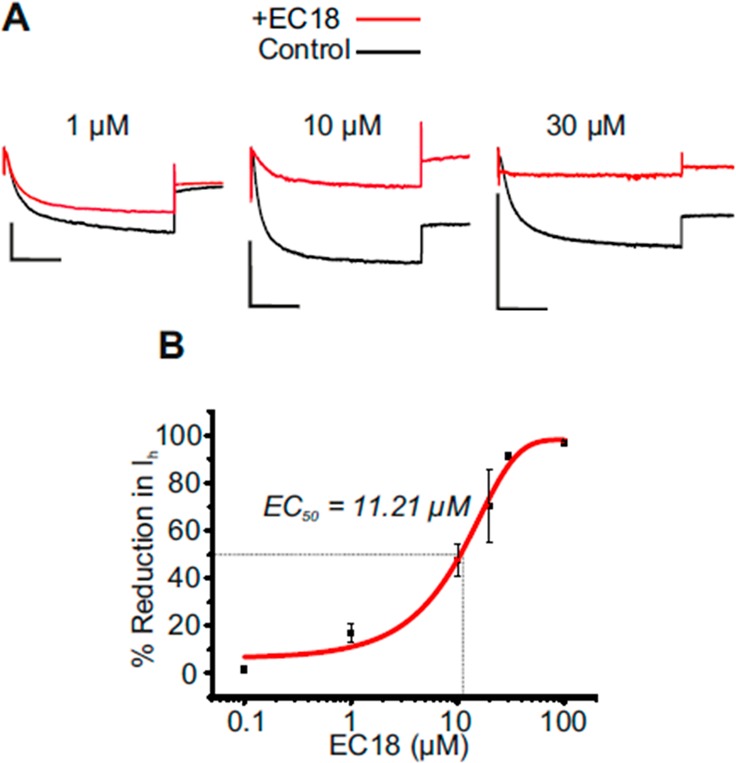
Effect of compound 3a on Ih in thalamic neurons. (A) Sample whole-cell patch clamp recordings showing the current reduction in VB TC neurons in brain slices32 by different 3a concentrations (1, 10, and 30 μM as indicated). Ih in the presence (red line) and absence (black lines) of 3a was elicited by standard voltage protocols (see Supporting Information 1). Scale bars represent 1 nA and 2 s. (B) Dose–response relationship of Ih amplitude reduction. The EC50 value was estimated by fitting a sigmoidal function (red line) to the data points (see Supporting Information 1).
An effect on K+ current was found also in VB TC neurons (Figure 4). When K+ outward currents were induced by step depolarizations (Figure 4A, lower inset) 10 μM 3a blocked 14.5 ± 2.2% (n = 8; Figure 4B) of the late component while leaving the early transient current unaffected. These findings indicate that compound 3a has selective effects on different types of voltage-dependent K+ channels, blocking slow delayed rectifier channels (Figure 4A upper inset) but not affecting K+ channels carrying A-type current in TC neurons.32,36
Figure 4.
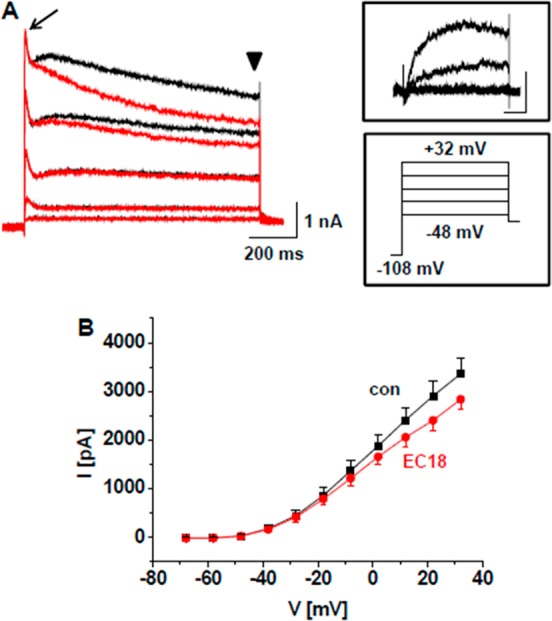
Effect of compound 3a on K+ outward currents in thalamic neurons. (A) Families of outward currents evoked by standard voltage protocols (see lower inset and Supporting Information 1) under control conditions (black traces) and in the presence of 10 μM 3a (red traces) following acute isolation.33 Late current components (arrowhead) but not the early transient currents (arrow) were affected. The 3a-sensitive component is shown in the upper inset (scale bars represent 200 ms and 500 pA). (B) Current–voltage relationship of the late current component (see arrowhead in A).
The data reported in this Letter suggest that compound 3a is a valuable tool to study HCN channel isoforms in the CNS and in heart, useful to clarify the contribution of different HCN isoforms in mediating Ih. As well as for compounds 1 and 2, the ability of 3a to discriminate among HCN channel isoforms is an advantage with respect to compounds such as ZD7288 or ivabradine, which are commonly used to block Ih but are not isoform-selective and, as well as 3a, nor absolutely specific for HCN channels.37,38
In conclusion, a compound has been discovered, 3a (EC18), showing some selectivity for HCN4 channels, and able to discriminate among HCN channel isoform in recombinant systems and in native tissues. Even if 3a shows some activity on Na+ and K+ currents, its HCN4 blocking properties make it a valuable tool to study HCN4 channels in a physiological environment or in pathologies, such as those of the CNS. In particular, the HCN4-blocking activity in the thalamus opens the way to test this compound in animal models of neurological disorders; the results of these tests will be presented in due time. Work is underway to improve potency and selectivity of the lead compound.
Acknowledgments
We want to thank Prof. Martin Biel (University of Munich, Germany) for providing stably transfected HCN1, HCN2, and HCN4 HEK293 cells. We wish to thank Prof. Andras Varrò (University of Szeged) and Prof. Alessandro Mugelli (University of Florence) for scientific support.
Glossary
ABBREVIATIONS
- AP
action potential
- HCN
hyperpolarization-activated cyclic nucleotide-gated
- DPPA
diphenyl phosphoryl azide
- DRG
dorsal root ganglion
- SAN
sino-atrial node
- TC
thalamocortical relay neurons
- VB
ventrobasal thalamic complex
Supporting Information Available
The Supporting Information is available free of charge on the ACS Publications website at DOI: 10.1021/acsmedchemlett.8b00587.
Author Present Address
∥ Stabilimento Chimico Farmaceutico Militare, Ministero della Difesa, Via Reginaldo Giuliani 201, I-50141 Firenze, Italy.
Author Contributions
The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript.
M.N.R., M.D.L., L.G., L.S., and E.C. have been supported by Normacor (Contract LSH M/CT/2006/018676). T.B. was supported by DFG (BU 1019/15-1/16-1). A.G., T.A.L., and I.K. were supported by the Hungarian Government-Ministry of Human Resources (grant EFOP-3.6.2-16-2017-00006, LIVE LONGER).
The authors declare no competing financial interest.
Supplementary Material
References
- Sartiani L.; Mannaioni G.; Masi A.; Romanelli M. N.; Cerbai E. The Hyperpolarization-Activated Cyclic Nucleotide–Gated Channels: from Biophysics to Pharmacology of a Unique Family of Ion Channels. Pharmacol. Rev. 2017, 69 (4), 354–395. 10.1124/pr.117.014035. [DOI] [PubMed] [Google Scholar]
- Wahl-Schott C.; Biel M. HCN channels: structure, cellular regulation and physiological function. Cell. Mol. Life Sci. 2009, 66 (3), 470–494. 10.1007/s00018-008-8525-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Datunashvili M.; Chaudhary R.; Zobeiri M.; Lüttjohann A.; Mergia E.; Baumann A.; Balfanz S.; Budde B.; van Luijtelaar G.; Pape H. C.; Koesling D.; Budde T. Modulation of hyperpolarization-activated inward current and thalamic activity modes by different cyclic nucleotides. Front. Cell. Neurosci. 2018, 369. 10.3389/fncel.2018.00369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herrmann S.; Schnorr S.; Ludwig A. HCN Channels—Modulators of Cardiac and Neuronal Excitability. Int. J. Mol. Sci. 2015, 16 (1), 1429–1447. 10.3390/ijms16011429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Notomi T.; Shigemoto R. Immunohistochemical localization of Ih channel subunits, HCN1–4, in the rat brain. J. Comp. Neurol. 2004, 471 (3), 241–276. 10.1002/cne.11039. [DOI] [PubMed] [Google Scholar]
- Müller F.; Scholten A.; Ivanova E.; Haverkamp S.; Kremmer E.; Kaupp U. B. HCN channels are expressed differentially in retinal bipolar cells and concentrated at synaptic terminals. Eur. J. Neurosci. 2003, 17, 2084–2096. 10.1046/j.1460-9568.2003.02634.x. [DOI] [PubMed] [Google Scholar]
- Chaplan S. R.; Guo H.-Q.; Lee D. H.; Luo L.; Liu C.; Kuei C.; Velumian A. A.; Butler M. P.; Brown S. M.; Dubin A. E. Neuronal Hyperpolarization-Activated Pacemaker Channels Drive Neuropathic Pain. J. Neurosci. 2003, 23, 1169–1178. 10.1523/JNEUROSCI.23-04-01169.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Postea O.; Biel M. Exploring HCN channels as novel drug targets. Nat. Rev. Drug Discovery 2011, 10, 903–913. 10.1038/nrd3576. [DOI] [PubMed] [Google Scholar]
- DiFrancesco J. C.; DiFrancesco D. Dysfunctional HCN ion channels in neurological diseases. Front. Cell. Neurosci. 2015, 9, 71. 10.3389/fncel.2015.00071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sulfi S.; Timmis A. D. Ivabradine – the first selective sinus node If channel inhibitor in the treatment of stable angina. Int. J. Clin.Pract. 2006, 60, 222–228. 10.1111/j.1742-1241.2006.00817.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bielecka-Dabrowa A.; von Haehling S.; Rysz J.; Banach M. Novel drugs for heart rate control in heart failure. Heart Failure Rev. 2018, 23 (4), 517–525. 10.1007/s10741-018-9696-x. [DOI] [PubMed] [Google Scholar]
- Emery E. C.; Young G. T.; McNaughton P. A. HCN2 ion channels: an emerging role as the pacemakers of pain. Trends Pharmacol. Sci. 2012, 33 (8), 456–463. 10.1016/j.tips.2012.04.004. [DOI] [PubMed] [Google Scholar]
- Zhang H.; Dougherty P. M. Enhanced Excitability of Primary Sensory Neurons and Altered Gene Expression of Neuronal Ion Channels in Dorsal Root Ganglion in Paclitaxel-induced Peripheral Neuropathy. Anesthesiology 2014, 120 (6), 1463–1475. 10.1097/ALN.0000000000000176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campostrini G.; DiFrancesco J. C.; Castellotti B.; Milanesi R.; Gnecchi-Ruscone T.; Bonzanni M.; Bucchi A.; Baruscotti M.; Ferrarese C.; Franceschetti S.; Canafoglia L.; Ragona F.; Freri E.; Labate A.; Gambardella A.; Costa C.; Gellera C.; Granata T.; Barbuti A.; DiFrancesco D. A Loss-of-Function HCN4 Mutation Associated With Familial Benign Myoclonic Epilepsy in Infancy Causes Increased Neuronal Excitability. Front. Mol. Neurosci. 2018, 11, 269. 10.3389/fnmol.2018.00269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Descoeur J.; Pereira V.; Pizzoccaro A.; Francois A.; Ling B.; Maffre V.; Couette B.; Busserolles J.; Courteix C.; Noel J.; Lazdunski M.; Eschalier A.; Authier N.; Bourinet E. Oxaliplatin-induced cold hypersensitivity is due to remodelling of ion channel expression in nociceptors. EMBO Mol. Med. 2011, 3 (5), 266–278. 10.1002/emmm.201100134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young G. T.; Emery E. C.; Mooney E. R.; Tsantoulas C.; McNaughton P. A. Inflammatory and neuropathic pain are rapidly suppressed by peripheral block of hyperpolarisation-activated cyclic nucleotide-gated ion channels. Pain 2014, 155, 1708–1719. 10.1016/j.pain.2014.05.021. [DOI] [PubMed] [Google Scholar]
- Luszczki J. J.; Prystupa A.; Andres-Mach M.; Marzêda E.; Florek-Luszczki M. Ivabradine (a hyperpolarization activated cyclic nucleotide-gated channel blocker) elevates the threshold for maximal electroshock-induced tonic seizures in mice. Pharmacol. Rep. 2013, 65, 1407–1414. 10.1016/S1734-1140(13)71500-7. [DOI] [PubMed] [Google Scholar]
- Stieber J.; Wieland K.; Stockl G.; Ludwig A.; Hofmann F. Bradycardic and Proarrhythmic Properties of Sinus Node Inhibitors. Mol. Pharmacol. 2006, 69, 1328–1337. 10.1124/mol.105.020701. [DOI] [PubMed] [Google Scholar]
- Romanelli M. N.; Sartiani L.; Masi A.; Mannaioni G.; Manetti D.; Mugelli A.; Cerbai E. HCN channels modulators: the need for selectivity. Curr. Top. Med. Chem. 2016, 16, 1764–1791. 10.2174/1568026616999160315130832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tibbs G. R.; Rowley T. J.; Sanford R. L.; Herold K. F.; Proekt A.; Hemmings H. C.; Andersen O. S.; Goldstein P. A.; Flood P. D. HCN1 Channels as Targets for Anesthetic and Nonanesthetic Propofol Analogs in the Amelioration of Mechanical and Thermal Hyperalgesia in a Mouse Model of Neuropathic Pain. J. Pharmacol. Exp. Ther. 2013, 345 (3), 363–373. 10.1124/jpet.113.203620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tae H.-S.; Smith K. M.; Phillips A. M.; Boyle K. A.; Li M.; Forster I. C.; Hatch R. J.; Richardson R.; Hughes D. I.; Graham B. A.; Petrou S.; Reid C. A. Gabapentin Modulates HCN4 Channel Voltage-Dependence. Front. Pharmacol. 2017, 8, 554. 10.3389/fphar.2017.00554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melchiorre M.; Del Lungo M.; Guandalini L.; Martini E.; Dei S.; Manetti D.; Scapecchi S.; Teodori E.; Sartiani L.; Mugelli A.; Cerbai E.; Romanelli M. N. Design, Synthesis, and Preliminary Biological Evaluation of New Isoform-Selective f-Current Blockers. J. Med. Chem. 2010, 53 (18), 6773–6777. 10.1021/jm1006758. [DOI] [PubMed] [Google Scholar]
- Del Lungo M.; Melchiorre M.; Guandalini L.; Sartiani L.; Mugelli A.; Koncz I.; Szel T.; Varro A.; Romanelli M. N.; Cerbai E. Novel blockers of hyperpolarization-activated current with isoform selectivity in recombinant cells and native tissue. Br. J. Pharmacol. 2012, 166 (2), 602–616. 10.1111/j.1476-5381.2011.01782.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Resta F.; Micheli L.; Laurino A.; Spinelli V.; Mello T.; Sartiani L.; Di Cesare Mannelli L.; Cerbai E.; Ghelardini C.; Romanelli M. N.; Mannaioni G.; Masi A. Selective HCN1 block as a strategy to control oxaliplatin-induced neuropathy. Neuropharmacology 2018, 131, 403–413. 10.1016/j.neuropharm.2018.01.014. [DOI] [PubMed] [Google Scholar]
- Dini L.; Del Lungo M.; Resta F.; Melchiorre M.; Spinelli V.; Di Cesare Mannelli L.; Ghelardini C.; Laurino A.; Sartiani L.; Coppini R.; Mannaioni G.; Cerbai E.; Romanelli M. N. Selective blockade of HCN1/HCN2 channels as a potential pharmacological strategy against pain. Front. Pharmacol. 2018, 9, 1252. 10.3389/fphar.2018.01252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dei S.; Romanelli M. N.; Scapecchi S.; Teodori E.; Gualtieri F.; Chiarini A.; Voigt W.; Mannhold R. Verapamil Analogues with Restricted Molecular Flexibility: Synthesis and Pharmacological Evaluation of the Four Isomers of α-1-[3-[1-N-[2-(3,4-Dimethoxy-phenyl)ethyl]-N-methylamino]cyclohexyl]-α-isopropyl-3,4-dimethoxybenzeneacetonitrile. J. Med. Chem. 1993, 36, 439–445. 10.1021/jm00056a003. [DOI] [PubMed] [Google Scholar]
- Allan R. D.; Johnston G. A. R.; Twichin B. Synthesis of analogues of GABA. VI* Stereoisomer of cis-3-Aminocyclohexanecarboxylic Acid. Aust. J. Chem. 1981, 34, 2231–2236. 10.1071/CH9812231. [DOI] [Google Scholar]
- Reiffen M.; Eberlien W.; Müller P.; Psiorz M.; Noll K.; Heider J.; Lillie C.; Kobinger W.; Luger P. Specific Bradycardic Agents. Chemistry, Pharmacology, and Structure-Activity Relationships of Substituted Benzazepinones, a New Class of Compounds Exerting Antiischemic Properties. J. Med. Chem. 1990, 33, 1496–1504. 10.1021/jm00167a033. [DOI] [PubMed] [Google Scholar]
- Romanelli M. N.; Cerbai E.; Dei S.; Guandalini L.; Martelli C.; Martini E.; Scapecchi S.; Teodori E.; Mugelli A. Design, synthesis and preliminary biological evaluation of Zatebradine analogues as potential blockers of the hyperpolarization-activated current. Bioorg. Med. Chem. 2005, 13, 1211–1220. 10.1016/j.bmc.2004.11.017. [DOI] [PubMed] [Google Scholar]
- Fan X.; Chen Y.; Wu P.; Xing J.; Chen H.; Song T.; Yang J.; Zhang J.; Huang C. Novel electropharmacological activity of amiodarone on human HCN channels heterologously expressed in the Xenopus oocytes. Eur. J. Pharmacol. 2011, 669 (1–3), 15–23. 10.1016/j.ejphar.2011.07.039. [DOI] [PubMed] [Google Scholar]
- Koncz I.; Szél T.; Bitay M.; Cerbai E.; Jaeger K.; Fülöp F.; Jost N.; Virág L.; Orvos P.; Tálosi L.; Kristóf A.; Baczkó I.; Papp J. G.; Varró A. Electrophysiological effects of ivabradine in dog and human cardiac preparations: Potential antiarrhythmic actions. Eur. J. Pharmacol. 2011, 668 (3), 419–426. 10.1016/j.ejphar.2011.07.025. [DOI] [PubMed] [Google Scholar]
- Kanyshkova T.; Broicher T.; Meuth S. G.; Pape H.-C.; Budde T. A-type K+ currents in intralaminar thalamocortical relay neurons. Pfluegers Arch. 2011, 461 (5), 545. 10.1007/s00424-011-0953-2. [DOI] [PubMed] [Google Scholar]
- Seifert R.; Scholten A.; Gauss R.; Mincheva A.; Lichter P.; Kaupp U. B. Molecular characterization of a slowly gating human hyperpolarization-activated channel predominantly expressed in thalamus, heart, and testis. Proc. Natl. Acad. Sci. U. S. A. 1999, 96 (16), 9391–9396. 10.1073/pnas.96.16.9391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ludwig A.; Budde T.; Stieber J.; Moosmang S.; Wahl C.; Holthoff K.; Langebartels A.; Wotjak C.; Munsch T.; Zong X.; Feil S.; Feil R.; Lancel M.; Chien K. R.; Konnerth A.; Pape H. C.; Biel M.; Hofmann F. Absence epilepsy and sinus dysrhythmia in mice lacking the pacemaker channel HCN2. EMBO J. 2003, 22 (2), 216–224. 10.1093/emboj/cdg032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leist M.; Datunashvilli M.; Kanyshkova T.; Zobeiri M.; Aissaoui A.; Cerina M.; Romanelli M. N.; Pape H.-C.; Budde T. Two types of interneurons in the mouse lateral geniculate nucleus are characterized by different h-current density. Sci. Rep. 2016, 6, 24904. 10.1038/srep24904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Budde T.; Mager R.; Pape H.-C. Different Types of Potassium Outward Current in Relay Neurons Acutely Isolated from the Rat Lateral Geniculate Nucleus. Eur. J. Neurosc. 1992, 4 (8), 708–722. 10.1111/j.1460-9568.1992.tb00180.x. [DOI] [PubMed] [Google Scholar]
- Melgari D.; Brack K. E.; Zhang Y.; El Harchi A.; Mitcheson J. S.; Dempsey C. E.; Ng G. A.; Hancox J. C. hERG potassium channel inhibition by ivabradine requires channel gating. J. Mol. Cell. Cardiol. 2015, 87, 126–128. 10.1016/j.yjmcc.2015.08.002. [DOI] [PubMed] [Google Scholar]
- Wu X.; Liao L.; Liu X.; Luo F.; Yang T.; Li C. Is ZD7288 a selective blocker of hyperpolarization-activated cyclic nucleotide-gated channel currents?. Channels 2012, 6, 1–5. 10.4161/chan.22209. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.