Abstract
Neurological diseases such as Alzheimer’s disease and Parkinson’s disease are growing problems, as average life expectancy is increasing globally. Drug discovery for neurological disease remains a major challenge. Poor understanding of disease pathophysiology and incomplete representation of human disease in animal models hinder therapeutic drug development. Recent advances with induced pluripotent stem cells (iPSCs) have enabled modeling of human diseases with patient-derived neural cells. Utilizing iPSC-derived neurons advances compound screening and evaluation of drug efficacy. These cells have the genetic backgrounds of patients that more precisely model disease-specific pathophysiology and phenotypes. Neural cells derived from iPSCs can be produced in a large quantity. Therefore, application of iPSC-derived human neurons is a new direction for neuronal drug discovery.
Keywords: Induced pluripotent stem cells, iPSC models, neuronal diseases, cell-based disease models, drug discovery and development
Teaser:
Patient-derived induced pluripotent stem cells are a useful model system for neural drug discovery.
Introduction
Globally, the increasing number of patients suffering from neurological and neuropsychiatric diseases is costing the healthcare industry billions of dollars. Affecting millions of individuals worldwide, these diseases present a massive economic, emotional and physical burden to individuals, their families and society [1]. Accordingly, the global demand for effective therapeutics against neuronal disorders has dramatically increased. Discovering ways to meet this need is a monumental challenge to 21st century medicine. Despite significant investments, there are still no cures or effective disease-modifying therapies for most neuronal diseases including Alzheimer’s disease (AD) and Parkinson’s disease (PD). The available therapies only help manage some symptoms of these disorders. To date, no treatments identified can halt or prevent their progression.
For drug development, animal disease models have crucial roles in the stages of lead discovery and preclinical development. Animal models are beneficial tools to identify the etiologies underlying human diseases and targets for drug development as well as for evaluating drug efficacy prior to conducting clinical trials. However, species differences could result in incomplete representation or misrepresentation of human diseases, especially when small animals are used for neuronal disease modeling [3]. Although many transgenic mouse models have been created, none has captured the full spectrum of human disease pathology [4]; and large animal models have their own limitations such as higher costs, longer experimental times and ethical constraints. Large animal models still retain the problematic characteristic of dissimilarity to humans [5]. Additionally, positive efficacy results observed in preclinical animal models are often not able to be reproduced in clinical trials [6]. Thus, there is an essential need to generate alternative human disease models for drug development that will be further discussed below.
Human induced pluripotent stem cell (iPSC) technology has opened a new path for disease modeling and drug discovery because patient-derived iPSCs and their derivative cells represent a more relevant disease system in the appropriate setting. Patient-derived iPSC models are more suitable for phenotypic-based drug discovery because they share the same genetic background with patients and can have the same disease phenotypes [7]. Differentiation of iPSCs into many cell types, including different types of neurons, enables disease modeling. These differentiated cells can be used to develop disease-relevant assays for drug screening. Thus, patient-derived iPSCs offer a new way to model and study sporadic (arising or occurring randomly without known cause as somatic mutations) diseases in addition to genetic diseases. Many neuronal disorders occur sporadically including in AD where 95% of patients show sporadic onset [8,9]. Although iPSCs can be used extensively in drug discovery and cell therapy without ethical constraints, there are still limitations such as incomplete cellular reprogramming and the genetic and epigenetic changes that can occur with prolonged culturing of iPSCs [10,11]. CRISPR- or TALEN-based DNA recombinant technology could be used as a beneficial and complementary tool for confirmation, because they enable the minimization of genetic variability. However, a major challenge in CRISPR–Cas9 technology is the possibility of off-target effects [3]. Another concern with using iPSCs is the lack of environmental factors in vitro that play an important part in neuropsychiatric diseases such as depression and anxiety [12]. Underrepresentation of iPSC-derived neurons for different developmental stages of the fetal brain could also be a disadvantage [13]. Nevertheless, the application of iPSCs in modeling neuronal diseases is an important alternative to animal disease models for drug discovery and development. The continuous development of iPSC technologies will help to overcome these shortcomings and improve the representation of human diseases using iPSC-derived models. In this review, we provide a brief overview of the applications of iPSC-derived neuronal disease models in drug discovery for neurodegenerative and neuropsychiatric diseases, as well as perspectives and highlights of emerging opportunities.
Neural iPSC-based models
iPSC generation
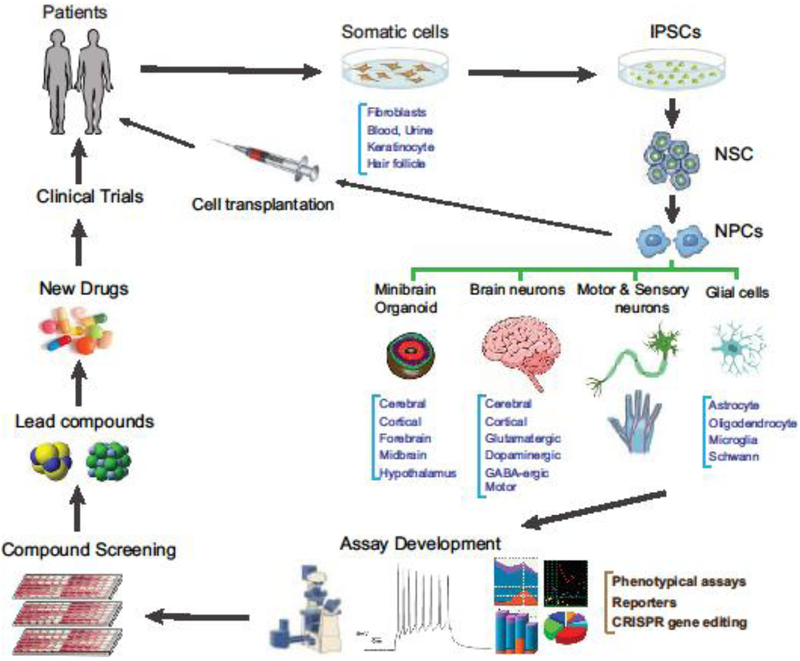
iPSCs can be generated from patient cell samples such as dermal fibroblasts, peripheral blood, urine, hair follicles and keratinocytes [3,14] (Figure 1). Several gene-delivery methods have been reported for generating iPSC lines: single cassette reprogramming vectors, reprogramming by nonintegrating viruses, nonviral reprogramming methods (mRNA transfection) and minicircle vectors [3,15]. However, Sendai virus technology and episomal plasmid vectors are integration free and most commonly used for efficient generation of iPSCs [15]. The Sendai-virus-based method has high efficiency for iPSC generation from patient samples [16]. iPSCs are easily proliferated and can be differentiated into many cell types.
Figure 1.
Applications of iPSCs in drug discovery and development. Cells easily obtained from actual patients are cultured and dedifferentiated into iPSCs. Next, the iPSCs can be re-differentiated into specific neural cell types and employed in assay development, drug screens, lead development, new drugs and clinical trials, leading to new therapies for neural diseases.
Neural stem cells and neuronal progenitor cells
Neural stem cells (NSCs) and neuronal progenitor cells (NPCs) can be quickly generated from iPSCs that are self-renewable. These cells can be produced in large quantities with high reproducibility. Depending on the disease types, NSCs and NPCs can have the relevant disease phenotypes that can be used as disease models for compound screening and efficacy tests [17]. NPCs have also been used as disease models for compound screening [6].
Neurons
Neuronal cells can be differentiated from the NSCs and NPCs or directly differentiated from iPSCs [18]. We have generated general neurons differentiated from iPSC-derived neural stem cells [17]. These neurons are relatively quick to obtain (usually in 2 weeks), exhibit disease phenotypes and can be used for evaluation of drug efficacy, although their purity and maturity are in question [19]. Finally, iPSCs can also be differentiated to more-specific neuron types, such as cortical neurons [20], glutamatergic neurons [21], GABAergic neurons [22], serotonergic neurons, dopaminergic neurons [21], motor neurons and sensory neurons [23], as well as astrocytes and oligodendrocytes [24].
Co-culture and neural organoids and minibrains
To better mimic brain histology and function, co-cultures of neurons with astrocytes and other cells (epithelial and endothelial cells) have been reported [24]. Limitations of dissociated neuronal cultures and the potential importance of cell–cell interactions for some neuronal diseases point the way toward 3D models. 3D neuronal cell culture systems have been reported; these recapitulate many of the cellular aspects of early brain development and permit the study of disease biology in more-complex environments, including cerebral organoids, cortical spheroids or forebrain organoids that mimic the organizational features of the human brain [25]. These 3D approaches have been used to study the disease biology of AD and microcephaly; they have yet to be used to study other neurological disorders. More-recent studies on familial AD have applied the 3D culture model to generate high-throughput models for drug screening against tau aggregation [26], or to compare efficacy of drug candidates in 2D versus 3D culture systems [27]. Raja et al. reported that brain organoids from familial AD patients recapitulate AD disease phenotypes and pathologies including amyloid aggregation, hyperphosphorylated tau and endosome abnormalities, all of which were reduced by treatment with secretase inhibitors [28].
Patient iPSC-based neural disease models
Alzheimer’s disease
AD is a highly prevalent neurodegenerative disorder involving the progressive loss of neurons in the brain. It has been estimated that by the year 2050 the population suffering from AD will reach ~100 million [29,30]. The majority of patients with AD suffer from sporadic AD (sAD) and ~0.5% of all AD patients suffer from familial AD (fAD) [31]. Reasons for the development of sAD are mostly unknown but some studies suggest that spontaneous germline mutations in APOE might explain some cases. A recent study using human iPSC (hiPSC)-derived neurons to test R33, a compound that stabilizes the retromer, indicated that retromer stabilization is a promising avenue for therapeutic development against sAD [32].
The major causes of fAD are mutations in PS1, PS2 and APP genes [29]. Using fAD patient-derived iPSC lines, the APP cleavage product β-CTF was found to be involved in the regulation of tau pathology [33]. Apigenin, an autophagy inducer, has been evaluated in fAD patient-derived neurons co-cultured with activated murine microglial cells. The treatment resulted in a reversal of morphological deficiencies, a reduction of hyperexcitability and protection against apoptosis [30]. Experiments with another line of iPSC-derived fAD neurons suggest that overactivated BRCA1 could lead to amyloid-β pathology and promote cell cycle reentry-driven cell death [34]. All of the above and following examples can be found in Table 1.
Table 1.
Examples of iPSC applications in neuronal diseases
| Disease | Genes & applications | iPSC model | Phenotypes discovered | Therapeutic target | Refs |
|---|---|---|---|---|---|
| AD | PS1, PS2 and APP | Genes reported to be related to AD but the iPSCs have not been reported | ND | ND | [29] |
| APP cleavage product (β-CTF) | Neuronsa | Tau pathology | ND | [33] | |
| Overactivated BRCA1 | Neuronsa | Amyloid-β pathology, cell death | ND | [34] | |
| Testing retromer | Neurons | ND | Retromer stabilizing compoundsb | [32] | |
| PD | 17 genes | DA neurons | Increased apoptosis, reduced neurites, impaired autophagy impaired mitophagy, irregular DA metabolism, mitochondrial deficits and oxidative stress | ND | [37] |
| GBA-N370S mutations | Midbrain DA neuronsa | Increased autophagosomes, higher expression of LAMP1, LAMP2 and cathepsin | ND | [38] | |
| PINK1 mutations | Midbrain DA neuronsa | Impaired mitophagy | ND | [39] | |
| R258Q mutation | Neuronsa | Defects in autophagosome assembly | ND | [40] | |
| ND | DA-neurons | ND | Dopaminergic p23 inhibitionb | [41] | |
| ALS | sALS patients | Sporadic neuronsa | TDP-43 pathology | ND | [44] |
| fALS mutation | iPSCsa | Proteinopathies, ER and oxidative stress | ND | [44] | |
| Phenotypic repurposing screen | Neuronsa | Bosutinib reduced phenotypes, increased survival of motor neurons Src/cAblb | ND | [43] | |
| HD | ND | Neuronsa | Increased cell death, sensitivity to stressors glutamate toxicity, reduced electrical firing |
NMDA & AMPA receptor inhibitorsb | [49] |
| ND | Neuronsa | MAPK and Wnt signaling pathwaysb | [50] | ||
| ND | Neuronsa | Increased lysosomes/autophagosomes, increased cell death, nuclear indentation, impaired nucleo-cytoplasmic transport | ND | [47] | |
| ND | Neuronsa | Activation of PPARγ by bexarotene or KD3010 rescued oxidative metabolism | ND | [52] | |
| Proteasome activity analysis | NPCs and neuronsa | FOXOs that modulate proteasome activityb | ND | [48] | |
| Epilepsy | Nav 1.1 mutation | GABAergic neurons | Influence channel currents and activation | ND | [53] |
| Expression profiling | iPSCsa | Upregulation of GRID1 (encodes glutamate D1 receptor) | ND | [54] | |
| CACNA1C mutation | Neuronsa | Action potential width, calcium current increased | Calcium channel blocker nimodipineb | [55] | |
| SZ | DISC1 mutation | iPSCs | Aberrant synaptic formation, synaptic vesicle release deficits | ND | [57] |
| 15q11.2 microdeletions |
NPCsa | Defects in adherent junctions and apical polarity | ND | [58] | |
| Gene expression analysis | Neuronsa | Altered expression in glutamate receptors and Wnt and cAMP signaling | ND | [56] | |
| Gene expression & proteomics | NPCsa | Abnormalities in cytoskeletal remodeling & oxidative stress | ND | [59] | |
| ND | DA neuronsa | Reduced neurite count, dopamine release, delayed maturation, mitochondria perturbations | ND | [61] | |
| ND | Neuronsa | Dopaminergic antagonist loxapineb | [12] | ||
| BD | ND | Neuronsa | Changes in WNT pathway & ion channel subunits genes | ND | [62] |
| ND | NPCsa | Impaired neural differentiation & decreased proliferation | GSK3β known target of lithium therapyb | [62] | |
| ND | Neuronsa | Altered gene expression of mitochondrial, calcium-signaling, neuronal excitability | Lithium treatmentb | [61] | |
| ND | Hippocampal dentate gyms granule cellsa | Changes in: PKA/PKC-signaling pathways genes, action potential firing-related Na and K channel subunits, mitochondrial system, hyperexcitability | ND | [61] | |
| ASD | ND | iPSCsa | Smaller soma size, reduced dendritic spine, decreased glutamatergic synapse, lower Ca2+ transient, abnormal excitatory synaptic transmission, fewer excitatory synapses | IGF-1 treatmentb | [64] |
| CDKL5 mutation | Neuronsa | Deficit in KCC2 expression & function | Rescue KCC2 functionb | [66] | |
| Depression | ND | Phenotypes reported to be related to depression but the iPSCs have not been reported | Dysregulation of GABAergic neurons, reduced GABA receptors in parahippocampal | ND | [68] |
| ND | iPSC | Deficiencies of BDNF plays a significant part | BDNFb | [12,70] |
From patient-derived iPSCs
Potential drug candidate.
Abbreviations: AD, Alzheimer’s disease; ND, not determined; PD, Parkinson’s disease;
Parkinson’s disease
PD is the second-most-common age-dependent complex neurodegenerative disease and is characterized by selective loss of dopamine neurons in the substantia nigra [36]. PD has been extensively modeled using patient-derived iPSCs [35]. To date, 17 genes with PD-causing mutations have been identified and many of these have been used to generate dopaminergic (DA) neurons from patient iPSCs [37]. Interestingly, many of the disease phenotypes such as increased apoptosis, reduced neurites, impaired autophagy, impaired mitophagy, irregular DA metabolism, mitochondrial deficits and oxidative stress are confirmed in familial and sporadic PD patient iPS-DA neurons [36,38]. Midbrain DA neurons were generated using iPSCs from PD patients with the GBA-N370S mutation that showed increased autophagosome numbers and higher expression of LAMP1, LAMP2 and cathepsin, indicating deficient autophagic flux in these cells [38]. Patient-derived midbrain DA neurons with mutations in the E3 ubiquitin ligase, Parkin or PINK1 showed impaired mitophagy in comparison with the controls [39]. Vanhauwaert et al. used iPSC-derived neurons from PD patients with the R258Q mutation and found defects in the translation of the PI3P signal into productive autophagosome assembly [40]. A recent iPSC-derived DA neuron study revealed a potential novel link between hsp90 and PHD2-HIF1α through an interaction with the hsp90 co-factor p23, suggesting that dopaminergic p23 inhibition could be considered as a novel therapeutic target for the disorder.
Amyotrophic lateral sclerosis
Amyotrophic lateral sclerosis (ALS) is a devastating neurodegenerative disease with a population of 223 000 in 2015 across the globe, which features progressive loss of motor neurons (MNs). There is no effective treatment. Several iPSC models have recently been reported [42,43]. Patient iPSC-derived MNs exhibited the specific pathophysiology of ALS subtypes [18]. Sporadic disease represents 90% of ALS patients and iPSCs from these patients have been differentiated into neurons that exhibited cytoplasmic aggregation of TDP-43 [44]. Furthermore, gene expression profiling of sporadic ALS iPSC-derived MNs suggests deficiencies in mitochondrial function [45]. Additionally, familial ALS mutation phenotypes have been recapitulated in iPSC disease models showing characteristic proteinopathies, endoplasmic reticulum stress and oxidative stress [42,46]. Isogenic corrections of known genetic mutations, such as SOD1, successfully rescued the disease phenotypes in these patient cells [46]. In another study, a phenotypic repurposing screen using ALS iPSC-derived motor neurons showed that bosutinib, an autophagy inducer, reduced the amount of misfolded mutant SOD1 protein and attenuated altered expression of mitochondrial genes. Bosutinib also increased survival of ALS iPSC-derived motor neurons from sporadic ALS and familial ALS patients, suggesting that Src/c-Abl could be a potentially useful target for new ALS drug development [43].
Huntington’s disease
Huntington’s disease (HD) is an inherited neurodegenerative disorder, primarily affecting GABAergic neurons in the striatum. Around the world, cases of HD are found in 5–10 out of 100 000 people [47,48]. Increased cell death, sensitivity to stressors, glutamate toxicity and reduced sporadic electrical firing have been reported in striatal-like neurons derived from HD patient iPSCs. Interestingly, the severity of several of these phenotypes increased with the number of CAG trinucleotide repeats in the HTT gene. Another study reported that cell death occurred following BDNF withdrawal, as well as increased susceptibility to glutamate toxicity, which could be blocked by NMDA and AMPA receptor inhibitors [49], suggesting novel therapeutic approaches for HD. Several potential HD-related pathways such as MAPK and Wnt have been identified using patient-derived iPSC neurons [50]. Neskarov et al. reported that HD iPSC neurons showed increased numbers of lysosomes and autophagosomes and exhibited increased cell death alongside nuclear indentation [51]. Furthermore, nuclear deficits including nucleoporin aggregation and impaired nucleocytoplasmic transport have recently been demonstrated in HD-patient-derived neurons [47]. One of the most recent studies showed an important role for the peroxisome-activated receptor PPAR-δ in the disease pathophysiology of HD-patient-derived iPSC neurons [52]. Indirect activation of PPAR-γ (another subtype of the PPAR family) by the small-molecule compounds bexarotene or KD3010 significantly rescued impaired oxidative metabolism in HD neurons. Another recent study analyzed proteasome activity and the expression of FOXO transcription factors in HD-iPSC-derived NPCs and neurons, suggesting that FOXOs modulated proteasome activity and thus represent a potentially valuable therapeutic target for HD [48].
Epilepsy
Epilepsy is a neurological disorder causing seizures or unusual behavior, sensations and sometimes loss of awareness. Approximately 50 million people worldwide suffer from epilepsy, making it one of the most common neurological diseases globally [53]. Mutations in SCN1A, which encodes the α-subunit of Nav1.1, a voltage-gated sodium channel, causes epilepsy with wide ranges of clinical phenotypes known as Dravet syndrome. In a study using iPSC-derived GABAergic neurons, a mutation of Nav 1.1 (c.A5768G) influenced the level of current produced by (and activation of) the channel. Additionally, whole postsynaptic activity was changed from the inhibition-dominated state to the excitation-dominated state in patient-derived neuronal networks, a reversal of the excitatory level of spontaneous postsynaptic activity [53]. A recent study tied together the two forms of Rett syndrome: classical and atypical, by showing that the only expression change in patient iPSCs was an upregulation of GRID1, which encodes the glutamate D1 receptor [54]. In neurons differentiated from Timothy syndrome patients, iPSC lines with a mutation in CACNA1C displayed action potential width increase and sustained calcium current; the increase in the action potential width and the sustained calcium current were blocked by the atypical L-type calcium channel blocker nimodipine [55].
Schizophrenia
Schizophrenia (SZ) is a debilitating neuropsychiatric disorder with a worldwide prevalence of 1%. The symptoms vary in patients including abnormal behavior and speech, hallucinations, delusions and extremely disordered thinking [56]. Although the precise cause of this disease remains unclear, genetics and environmental factors have been linked to the disease. By generating hiPSCs from SZ patients with DISC1 mutations, a risk factor for SZ, researchers found that the mutant DISC1 gene causes aberrant synaptic formation and synaptic vesicle release deficits in SZ neurons in comparison with the isogenic hiPSCs [57]. The hiPSC-derived NPCs from SZ patients carrying 15q11.2 microdeletions also exhibited defects in adherent junctions and apical polarity [58]. A study of gene expression patterns of the SZ neurons revealed altered expression of genes involved in Wnt signaling, cAMP signaling and glutamate receptors [56]. A gene expression and proteomics study using NPCs found abnormalities in cytoskeletal remodeling and oxidative stress in SZ patient cells [59]. Dopaminergic neurons derived from SZ-iPSCs had a reduced neurite count and dopamine release and also showed delayed maturation [60]. Interestingly, these defective neurons exhibited perturbations in mitochondria, suggesting that the mitochondrial defect could play a key part in the pathogenesis of SZ [61]. In a recent study, treatment with the dopaminergic antagonist loxapine during the final 3 weeks of neuronal differentiation increased neuronal connectivity in SZ-iPSC-derived neurons.
Bipolar disorder
Bipolar disorder (BD) is a complex neuropsychiatric disorder characterized by intermittent episodes of mania and depression. Without treatment, ~15% of patients can commit suicide. Worldwide, the prevalence of BD is 2.4% [61]. BD-patient-derived neurons showed changes in expression of genes crucial for neuroplasticity, including Wnt pathway components and ion channel subunits. Interestingly, NPCs generated from BD patients showed impaired neural differentiation and decreased proliferation, both of which were rescued by a selective inhibitor of GSK3β that is a known target of lithium therapy [62]. Of note, BD-patient-derived neurons showed altered expression of mitochondrial, calcium-signaling and neuronal excitability genes, and a hyperexcitability phenotype with higher frequency of spontaneous action potentials. Remarkably, a 1-week treatment with lithium partially normalized the changes in mitochondrial gene expression and a hyperexcitability phenotype that only occurred in neurons derived from patients with BD who were responsive to lithium [61], suggesting a role of mitochondrial signaling in BD pathogenesis. Because BD patients often show atrophy in the hippocampus, a study with patient iPSC-derived hippocampal dentate gyrus granule cells revealed significant enhancement of expression in genes of the PKA/PKC signaling pathways, the action potential firing-related sodium and potassium channel subunit and the mitochondrial system. Patch-clamp recordings and calcium ion images of the BD patient neurons exhibited hyperexcitability in these neurons [63].
Autism spectrum disorder
Autism spectrum disorder (ASD; incidence of ~ 1% in the population) is a neuropsychiatric condition characterized by atypical development of social communication, and the presence of restrictive interests and repetitive behaviors [64]. Syndromic forms of autism spectrum diseases, such as Rett syndrome (RTT), Fragile X syndrome and Timothy Syndrome, have been modeled with hiPSCs [25]. For instance, hiPSCs generated from RTT patients were able to recapitulate several neurological phenotypes of the disease such as smaller soma size, reduced dendritic spine density, decreased glutamatergic synapse number, lower spontaneous calcium ion transient frequency, abnormal excitatory synaptic transmission and fewer excitatory synapses. A treatment with IGF-1 improved the neuronal growth and synaptogenesis, specifically glutamatergic synapses, in these patient iPSC-derived neurons [64]. hiPSC-derived neurons from patients with Rett-like syndrome with a mutation in CDKL5 [65] also showed a marked deficit in the expression of neuron-specific KCC2 and consequently a delayed GABA functional switch from excitation to inhibition [66], suggesting that rescue of KCC2 function in RTT neurons could serve as a potential therapeutic strategy for RTT.
Depression and anxiety disorders
Major depressive disorder (MDD) affects ~300 million people worldwide, ~4.5% of the global population [67]. Serotonin uptake modulators are currently the first-line treatment for MDD and anxiety disorders. Dysregulation of GABAergic neurons, reduced GABA receptors in the parahippocampal gyrus and reduction in the number and size of GABAergic neurons in the dorsolateral prefrontal cortex have also been implicated in depression and anxiety [68]. In addition, deficiency of BDNF plays a part in depression, anxiety and other neuropsychiatric illnesses [69] giving it great potential as a therapeutic agent [12,70]. It is conceivable that iPSC-derived cells with inducible BDNF expression might one day be used in a form of cell-based therapy for patients. Human embryonic stem cells and fibroblast-derived iPSCs have been used to generate serotonergic neurons that could lead to the discovery of new drug targets and new insights into the pathogenesis of depression [71].
Rare genetic diseases (neurological diseases)
The iPSC-based disease modeling system has added enormous value to modeling rare neuronal diseases caused by congenital gene mutations [72]. Disease-relevant animal models are usually unavailable for these diseases. For example, the neuronal cells from iPSCs of Niemann–Pick disease type 1C showed a cholesterol accumulation phenotype that is similar to human disease and responded to drug treatments [17]. The potency (IC50: half maximal inhibition) of methyl-β-cyclodextrin was 14.3 μM in neuronal cells compared with 60.5 μM in patient-derived fibroblasts, whereas the IC50 value of hydroxypropyl-β-cyclodextrin was 26.3 μM in neuronal cells compared with 1–3 mM in patient fibroblasts [17,73], indicating that neuronal cells are better as a model for evaluation of compound efficacy for the disease. In cystic fibrosis, mutation-specific iPSC lines were generated that were used to establish organoid models for evaluating and predicting drug clinical efficacies [74,75]. This approach has advantages because the appropriate animal models for evaluating drug efficacy to treat cystic fibrosis patients are not available. It is not easy to generate many individual mutation-based animal models to predict human drug efficacy. Another example is the NGLY-1 deficiency that is a rare neurological disease caused by mutations in the NLGY1 gene encoding N-glycanase 1, a hydrolase responsible for removing N-linked glycans from glycoproteins [76]. It has been difficult to generate a mouse model because NGLY1 knockout is lethal [77]. Therefore, generation of patient iPSCs and disease modeling with patient iPSC differentiated neurons will facilitate the study of disease pathophysiology and promote drug development [78].
Concluding remarks and future perspectives
Neural and neuropsychiatric diseases affect large populations globally. With improvement in healthcare, humans no longer succumb to historic causes of death such as infectious disease, accidents and now-easy-to-treat disorders like high blood pressure. Although the improvement in lifespan is welcome, the ensuing higher proportions of neural disorders is a challenge. There is an unmet need for relevant models of pathophysiology for neurological diseases. Major limitations in current neurological research and drug discovery include lack of valid disease models and unavailability of disease-specific human neuronal cells. The iPSC technology offers new opportunities to model human neuronal diseases using disease-relevant patient cells. In Figure 1 we present a schematic of how actual patients can provide tissues leading to the development of iPSCs, biologically relevant assays and finally drugs to address the unmet need in neural diseases. Although the method for iPSC generation has been well established, the technologies for iPSC-derived neuronal cells still need to be further developed to produce cost effective, matured and reproducible neuronal cells. The disease-relevant cell-based disease models are still to be established, optimized and developed. We believe that the drug development for neurological diseases will advance more quickly in parallel to the development of iPSC-based technologies.
Highlights:
Neural diseases and neuropsychiatric diseases are growing global problems
Effective therapies for neurological diseases are still unmet medical needs
Animal models do not often accurately represent human neurological diseases
Induced pluripotent stem cells are a good model system for human diseases
Acknowledgments
This work was supported by the Intramural Research Program of the Therapeutics for Rare and Neglected Diseases, the National Center for Advancing Translational Sciences and the National Institutes of Health. We would like to acknowledge Retrophin and NGLY1.org for support and helpful discussions. The authors would like to thank Dr DeeAnn Visk for her work in editing this manuscript.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Haggarty SJ and Perlis RH (2014) Translation: screening for novel therapeutics with disease-relevant cell types derived from human stem cell models. Biol. Psychiatry 75, 952–960 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Han C et al. (2018) Open science meets stem cells: a new drug discovery approach for neurodegenerative disorders. Front. Neurosci 12, 47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Shi Y et al. (2017) Induced pluripotent stem cell technology: a decade of progress. Nat. Rev. Drug Discov. 16, 115–130 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Onos KD et al. (2016) Toward more predictive genetic mouse models of Alzheimer’s disease. Brain Res. Bull 122, 1–11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Eaton SL and Wishart TM (2017) Bridging the gap: large animal models in neurodegenerative research. Mamm. Genome 28, 324–337 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Xu M et al. (2016) Disease models for the development of therapies for lysosomal storage diseases. Ann. N. Y. Acad. Sci 1371, 15–29 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mattis VB et al. (2012) Induced pluripotent stem cells from patients with Huntington’s disease show CAG-repeat-expansion-associated phenotypes. Cell Stem Cell 11, 264–278 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kondo T et al. (2013) Modeling Alzheimer’s disease with iPSCs reveals stress phenotypes associated with intracellular Abeta and differential drug responsiveness. Cell Stem Cell 12, 487–496 [DOI] [PubMed] [Google Scholar]
- 9.Adegbola A et al. (2017) Concise review: induced pluripotent stem cell models for neuropsychiatric diseases. Stem Cells Transl. Med 6, 2062–2070 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Raab S et al. (2014) A comparative view on human somatic cell sources for iPSC generation. Stem Cells Int. 2014, 768391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Sun W et al. (2017) Drug discovery and development for rare genetic disorders. Am. J. Med. Genet.A 173, 2307–2322 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Soliman MA et al. (2017) Pluripotent stem cells in neuropsychiatric disorders. Mol. Psychiatry 22, 1241–1249 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Deng B (2017) Mouse models and induced pluripotent stem cells in researching psychiatric disorders. Stem Cell Investig. 4, 62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Takahashi K et al. (2007) Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131, 861–872 [DOI] [PubMed] [Google Scholar]
- 15.Borgohain MP et al. (2018) An insight into DNA-free reprogramming approaches to generate integration-free induced pluripotent stem cells for prospective biomedical applications. Stem Cell Rev. Rep doi: 10.1007/s12015-018-9861-6 [DOI] [PubMed] [Google Scholar]
- 16.Beers J et al. (2015) A cost-effective and efficient reprogramming platform for large-scale production of integration-free human induced pluripotent stem cells in chemically defined culture. Sci. Rep 5, 11319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Yu D et al. (2014) Niemann–Pick disease type C: induced pluripotent stem cell-derived neuronal cells for modeling neural disease and evaluating drug efficacy. J. Biomol. Screen 19, 1164–1173 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ghaffari LT et al. (2018) Representing diversity in the dish: using patient-derived in vitro models to recreate the heterogeneity of neurological disease. Front. Neurosci 12, 56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Dai S et al. (2016) One-step seeding of neural stem cells with vitronectin-supplemented medium for high-throughput screening assays. J. Biomol. Screen 21, 1112–1124 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Shi Y et al. (2012) Human cerebral cortex development from pluripotent stem cells to functional excitatory synapses. Nat. Neurosci 15, 477–486, S471 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sanchez-Danes A et al. (2012) Disease-specific phenotypes in dopamine neurons from human iPS-based models of genetic and sporadic Parkinson’s disease. EMBO Mol. Med 4, 380–395 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yang N et al. (2017) Generation of pure GABAergic neurons by transcription factor programming. Nat. Methods 14, 621–628 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Boisvert EM et al. (2015) The specification and maturation of nociceptive neurons from human embryonic stem cells. Sci. Rep 5, 16821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Osaki T et al. (2018) In vitro microfluidic models for neurodegenerative disorders. Adv. Healthc. Mater 7, 1700489. [DOI] [PubMed] [Google Scholar]
- 25.Luo C et al. (2016) Cerebral organoids recapitulate epigenomic signatures of the human fetal brain. Cell Rep. 17, 3369–3384 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Medda X et al. (2016) Development of a scalable, high-throughput-compatible assay to detect tau aggregates using iPSC-derived cortical neurons maintained in a three-dimensional culture format. J. Biomol. Screen 21, 804–815 [DOI] [PubMed] [Google Scholar]
- 27.Lee HK et al. (2016) Three dimensional human neuro-spheroid model of Alzheimer’s disease based on differentiated induced pluripotent stem cells. PLoS One 11, e0163072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Raja WK et al. (2016) Self-organizing 3D human neural tissue derived from induced pluripotent stem cells recapitulate Alzheimer’s disease phenotypes. PLoS One 11, e0161969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Correa-Velloso JC et al. (2018) Pathophysiology in the comorbidity of bipolar disorder and Alzheimer’s disease: pharmacological and stem cell approaches. Prog. Neuropsychopharmacol. Biol. Psychiatry 80, 34–53 [DOI] [PubMed] [Google Scholar]
- 30.Balez R et al. (2016) Neuroprotective effects of apigenin against inflammation, neuronal excitability and apoptosis in an induced pluripotent stem cell model of Alzheimer’s disease. Sci. Rep 6, 31450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Puzzo D et al. (2015) Rodent models for Alzheimer’s disease drug discovery. Expert Opin. Drug Discov. 10, 703–711 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Young JE et al. (2018) Stabilizing the retromer complex in a human stem cell model of Alzheimer’s disease reduces TAU phosphorylation independently of amyloid precursor protein. Stem Cell Rep. 10, 1046–1058 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Moore S et al. (2015) APP metabolism regulates tau proteostasis in human cerebral cortex neurons. Cell Rep. 11, 689–696 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Wezyk M et al. (2018) Overactive BRCA1 affects presenilin 1 in induced pluripotent stem cell-derived neurons in Alzheimer’s disease. J. Alzheimers Dis. 62, 175–202 [DOI] [PubMed] [Google Scholar]
- 35.Cobb MM et al. (2018) iPS cells in the study of PD molecular pathogenesis. Cell Tissue Res. 373, 61–77 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hsieh CH et al. (2016) Functional impairment in miro degradation and mitophagy is a shared feature in familial and sporadic Parkinson’s disease. Cell Stem Cell 19, 709–724 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Surmeier DJ et al. (2017) Selective neuronal vulnerability in Parkinson disease. Nat. Rev. Neurosci 18, 101–113 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Fernandes HJ et al. (2016) ER stress and autophagic perturbations lead to elevated extracellular alpha-synuclein in GBA-N370S Parkinson’s iPSC-derived dopamine neurons. Stem Cell Rep. 6, 342–356 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Suzuki S et al. (2017) Efficient induction of dopaminergic neuron differentiation from induced pluripotent stem cells reveals impaired mitophagy in PARK2 neurons. Biochem. Biophys. Res. Commun 483, 88–93 [DOI] [PubMed] [Google Scholar]
- 40.Vanhauwaert R et al. (2017) The SAC1 domain in synaptojanin is required for autophagosome maturation at presynaptic terminals. EMBO J. 36, 1392–1411 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Rane A et al. (2018) Hsp90 co-chaperone p23 contributes to dopaminergic mitochondrial stress via stabilization of PHD2: implications for Parkinson’s disease. Neurotoxicology 65, 166–173 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Hall CE et al. (2017) Progressive motor neuron pathology and the role of astrocytes in a human stem cell model of VCP-related ALS. Cell Rep. 19, 1739–1749 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Imamura KIY et al. (2017) The Src/c-Abl pathway is a potential therapeutic target in amyotrophic lateral sclerosis. Sci. Transl. Med 9, eaaf3962. [DOI] [PubMed] [Google Scholar]
- 44.Qian K et al. (2017) Sporadic ALS astrocytes induce neuronal degeneration in vivo. Stem Cell Rep. 8, 843–855 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Alves CJ et al. (2015) Dysregulated expression of death, stress and mitochondrion related genes in the sciatic nerve of presymptomatic SOD1(G93A) mouse model of amyotrophic lateral sclerosis. Front. Cell Neurosci. 9, 332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Bhinge A et al. (2017) Genetic correction of SOD1 mutant iPSCs reveals ERK and JNK activated AP1 as a driver of neurodegeneration in amyotrophic lateral sclerosis. Stem Cell Rep. 8, 856–869 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Grima JC et al. (2017) Mutant huntingtin disrupts the nuclear pore complex. Neuron 94, 93–107, e106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Liu Y et al. (2017) FOXOs modulate proteasome activity in human-induced pluripotent stem cells of Huntington’s disease and their derived neural cells. Hum. Mol. Genet 26, 4416–4428 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Mattis VB et al. (2015) HD iPSC-derived neural progenitors accumulate in culture and are susceptible to BDNF withdrawal due to glutamate toxicity. Hum. Mol. Genet 24, 3257–3271 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Szlachcic WJ et al. (2015) Huntington disease iPSCs show early molecular changes in intracellular signaling, the expression of oxidative stress proteins and the p53 pathway. Dis. Model. Mech 8, 1047–1057 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Nekrasov ED et al. (2016) Manifestation of Huntington’s disease pathology in human induced pluripotent stem cell-derived neurons. Mol. Neurodegener 11, 27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Dickey AS et al. (2017) PPARδ activation by bexarotene promotes neuroprotection by restoring bioenergetic and quality control homeostasis. Sci. Transl. Med 9, eaal2332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Liu J et al. (2016) CRISPR/Cas9 facilitates investigation of neural circuit disease using human iPSCs: mechanism of epilepsy caused by an SCN1A loss-of-function mutation. Transl. Psychiatry 6, e703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Livide G et al. (2015) GluD1 is a common altered player in neuronal differentiation from both MECP2-mutated and CDKL5-mutated iPS cells. Eur. J. Hum. Genet 23, 195–201 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Pasca SP et al. (2011) Using iPSC-derived neurons to uncover cellular phenotypes associated with Timothy syndrome. Nat. Med 17, 1657–1662 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Brennand KJ et al. (2011) Modelling schizophrenia using human induced pluripotent stem cells. Nature 473, 221–225 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Wen Z et al. (2014) Synaptic dysregulation in a human iPS cell model of mental disorders. Nature 515, 414–418 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Yoon KJ et al. (2014) Modeling a genetic risk for schizophrenia in iPSCs and mice reveals neural stem cell deficits associated with adherens junctions and polarity. Cell Stem Cell 15, 79–91 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Brennand K et al. (2015) Phenotypic differences in hiPSC NPCs derived from patients with schizophrenia. Mol. Psychiatry 20, 361–368 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Robicsek O et al. (2013) Abnormal neuronal differentiation and mitochondrial dysfunction in hair follicle-derived induced pluripotent stem cells of schizophrenia patients. Mol. Psychiatry 18, 1067–1076 [DOI] [PubMed] [Google Scholar]
- 61.Mertens J et al. (2015) Differential responses to lithium in hyperexcitable neurons from patients with bipolar disorder. Nature 527, 95–99 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Madison JM et al. (2015) Characterization of bipolar disorder patient-specific induced pluripotent stem cells from a family reveals neurodevelopmental and mRNA expression abnormalities. Mol. Psychiatry 20, 703–717 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Liu YN et al. (2017) Application of induced pluripotent stem cells to understand neurobiological basis of bipolar disorder and schizophrenia. Psychiatry Clin. Neurosci 71, 579–599 [DOI] [PubMed] [Google Scholar]
- 64.Yuen RK et al. (2016) Genome-wide characteristics of de novo mutations in autism. NPJ Genom. Med 1, 16027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Ricciardi S et al. (2012) CDKL5 ensures excitatory synapse stability by reinforcing NGL-1-PSD95 interaction in the postsynaptic compartment and is impaired in patient iPSC-derived neurons. Nat. Cell Biol. 14, 911–923 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Tang X et al. (2016) KCC2 rescues functional deficits in human neurons derived from patients with Rett syndrome. Proc. Natl. Acad. Sci. U. S. A 113, 751–756 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Vos T et al. (2012) Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990–2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet 380, 2163–2196 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Mohler H (2012) The GABA system in anxiety and depression and its therapeutic potential. Neuropharmacology 62, 42–53 [DOI] [PubMed] [Google Scholar]
- 69.Vithlani M et al. (2013) The ability of BDNF to modify neurogenesis and depressive-like behaviors is dependent upon phosphorylation of tyrosine residues 365/367 in the GABA(A)-receptor gamma2 subunit. J. Neurosci 33, 15567–15577 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Liu G et al. (2014) Use of induced pluripotent stem cell derived neurons engineered to express BDNF for modulation of stressor related pathology. Front. Cell Neurosci. 8, 316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Lu J et al. (2016) Generation of serotonin neurons from human pluripotent stem cells. Nat. Biotechnol 34, 89–94 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Hamazaki T et al. (2017) Concise review: induced pluripotent stem cell research in the era of precision medicine. Stem Cells 35, 545–550 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Swaroop M et al. (2012) Evaluation of cholesterol reduction activity of methyl-beta-cyclodextrin using differentiated human neurons and astrocytes. J. Biomol. Screen 17, 1243–1251 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Chen KG et al. (2018) Pluripotent stem cell platforms for drug discovery. Trends Mol. Med 24, 805–820 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Gorshkov K et al. (2018) Advancing precision medicine with personalized drug screening. Drug Discov. Today doi: 10.1016/j.drudis.2018.08.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Bi Y et al. (2017) Repurposing of proton pump inhibitors as first identified small molecule inhibitors of endo-beta-N-acetylglucosaminidase (ENGase) for the treatment of NGLY1 deficiency, a rare genetic disease. Bioorg. Med. Chem. Lett 27, 2962–2966 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Fujihira H et al. (2017) Lethality of mice bearing a knockout of the Ngly1-gene is partially rescued by the additional deletion of the Engase gene. PLoS Genet. 13, e1006696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Li R et al. (2018) Generation of an induced pluripotent stem cell line from a patient carrying compound heterozygous p.Q208X and p.G310G mutations in NGLY1 gene. Stem Cell Res. doi: 10.1016/j.scr.2018.101362 [DOI] [PMC free article] [PubMed] [Google Scholar]