Abstract
Background
Erysipelas and cellulitis (hereafter referred to as 'cellulitis') are common bacterial skin infections usually affecting the lower extremities. Despite their burden of morbidity, the evidence for different prevention strategies is unclear.
Objectives
To assess the beneficial and adverse effects of antibiotic prophylaxis or other prophylactic interventions for the prevention of recurrent episodes of cellulitis in adults aged over 16.
Search methods
We searched the following databases up to June 2016: the Cochrane Skin Group Specialised Register, the Cochrane Central Register of Controlled Trials (CENTRAL), MEDLINE, Embase, and LILACS. We also searched five trials registry databases, and checked reference lists of included studies and reviews for further references to relevant randomised controlled trials (RCTs). We searched two sets of dermatology conference proceedings, and BIOSIS Previews.
Selection criteria
Randomised controlled trials evaluating any therapy for the prevention of recurrent cellulitis.
Data collection and analysis
Two authors independently carried out study selection, data extraction, assessment of risks of bias, and analyses. Our primary prespecified outcome was recurrence of cellulitis when on treatment and after treatment. Our secondary outcomes included incidence rate, time to next episode, hospitalisation, quality of life, development of resistance to antibiotics, adverse reactions and mortality.
Main results
We included six trials, with a total of 573 evaluable participants, who were aged on average between 50 and 70. There were few previous episodes of cellulitis in those recruited to the trials, ranging between one and four episodes per study.
Five of the six included trials assessed prevention with antibiotics in participants with cellulitis of the legs, and one assessed selenium in participants with cellulitis of the arms. Among the studies assessing antibiotics, one study evaluated oral erythromycin (n = 32) and four studies assessed penicillin (n = 481). Treatment duration varied from six to 18 months, and two studies continued to follow up participants after discontinuation of prophylaxis, with a follow‐up period of up to one and a half to two years. Four studies were single‐centre, and two were multicentre; they were conducted in five countries: the UK, Sweden, Tunisia, Israel, and Austria.
Based on five trials, antibiotic prophylaxis (at the end of the treatment phase ('on prophylaxis')) decreased the risk of cellulitis recurrence by 69%, compared to no treatment or placebo (risk ratio (RR) 0.31, 95% confidence interval (CI) 0.13 to 0.72; n = 513; P = 0.007), number needed to treat for an additional beneficial outcome (NNTB) six, (95% CI 5 to 15), and we rated the certainty of evidence for this outcome as moderate.
Under prophylactic treatment and compared to no treatment or placebo, antibiotic prophylaxis reduced the incidence rate of cellulitis by 56% (RR 0.44, 95% CI 0.22 to 0.89; four studies; n = 473; P value = 0.02; moderate‐certainty evidence) and significantly decreased the rate until the next episode of cellulitis (hazard ratio (HR) 0.51, 95% CI 0.34 to 0.78; three studies; n = 437; P = 0.002; moderate‐certainty evidence).
The protective effects of antibiotic did not last after prophylaxis had been stopped ('post‐prophylaxis') for risk of cellulitis recurrence (RR 0.88, 95% CI 0.59 to 1.31; two studies; n = 287; P = 0.52), incidence rate of cellulitis (RR 0.94, 95% CI 0.65 to 1.36; two studies; n = 287; P = 0.74), and rate until next episode of cellulitis (HR 0.78, 95% CI 0.39 to 1.56; two studies; n = 287). Evidence was of low certainty.
Effects are relevant mainly for people after at least two episodes of leg cellulitis occurring within a period up to three years.
We found no significant differences in adverse effects or hospitalisation between antibiotic and no treatment or placebo; for adverse effects: RR 0.87, 95% CI 0.58 to 1.30; four studies; n = 469; P = 0.48; for hospitalisation: RR 0.77, 95% CI 0.37 to 1.57; three studies; n = 429; P = 0.47, with certainty of evidence rated low for these outcomes. The existing data did not allow us to fully explore its impact on length of hospital stay.
The common adverse reactions were gastrointestinal symptoms, mainly nausea and diarrhoea; rash (severe cutaneous adverse reactions were not reported); and thrush. Three studies reported adverse effects that led to discontinuation of the assigned therapy. In one study (erythromycin), three participants reported abdominal pain and nausea, so their treatment was changed to penicillin. In another study, two participants treated with penicillin withdrew from treatment due to diarrhoea or nausea. In one study, around 10% of participants stopped treatment due to pain at the injection site (the active treatment group was given intramuscular injections of benzathine penicillin).
None of the included studies assessed the development of antimicrobial resistance or quality‐of‐life measures.
With regard to the risks of bias, two included studies were at low risk of bias and we judged three others as being at high risk of bias, mainly due to lack of blinding.
Authors' conclusions
In terms of recurrence, incidence, and time to next episode, antibiotic is probably an effective preventive treatment for recurrent cellulitis of the lower limbs in those under prophylactic treatment, compared with placebo or no treatment (moderate‐certainty evidence). However, these preventive effects of antibiotics appear to diminish after they are discontinued (low‐certainty evidence). Treatment with antibiotic does not trigger any serious adverse events, and those associated are minor, such as nausea and rash (low‐certainty evidence). The evidence is limited to people with at least two past episodes of leg cellulitis within a time frame of up to three years, and none of the studies investigated other common interventions such as lymphoedema reduction methods or proper skin care. Larger, high‐quality studies are warranted, including long‐term follow‐up and other prophylactic measures.
Plain language summary
Preventive treatments for repeated episodes of cellulitis and erysipelas
Background
Cellulitis and erysipelas are both bacterial infections of the skin that most commonly affect the leg. Erysipelas affects the upper layers of the skin, and cellulitis affects its deeper parts, but in practice it is often hard to tell the difference between them, so we consider them together for this review (and refer to them as 'cellulitis'). Up to 50% of people with cellulitis experience repeated episodes. Despite the burden of this condition, there is a lack of high‐certainty, evidence‐based information about the desirable treatment for the prevention of recurrent cellulitis.
Review question
What are the best available treatments to prevent repeated episodes of cellulitis in adults aged over 16 years compared to no treatment, placebo, another intervention, or the same intervention with a different plan of treatment, and what are their side effects?
Study characteristics
We searched relevant databases and registers up to June 2016. We identified six trials, with 573 participants, who had an average age between 50 and 70. Both genders were included, but there were nearly twice as many women. Five trials used antibiotic treatment (four penicillin and one erythromycin), which was compared to either no treatment or placebo, and one trial used selenium compared to physiological salt solution. Treatments lasted from six to 18 months.
The most common setting was the hospital, and two studies were multicentre. The studies were conducted in the UK, Sweden, Tunisia, Israel, and Austria. There was a small number of previous episodes of cellulitis in those recruited to the trials, ranging between one and four episodes in each study. The antibiotic trials assessed prevention with antibiotics in people with cellulitis of the legs, and the selenium trial assessed people with cellulitis of the arms.
Key results
Our main outcome was prevention of repeated episodes of cellulitis. Our other outcomes included the number of repeated attacks of cellulitis, time to next attack, hospitalisation, quality of life, development of antibiotic resistance, adverse reactions and death.
Combining the results of all five trials that used antibiotics, we found moderate‐certainty evidence that for those people under preventative treatment, antibiotic treatment in general, and penicillin in particular, is probably both effective and safe for the prevention of repeated episodes of leg cellulitis when compared with no treatment or placebo.
The analyses showed that, compared with no treatment or placebo, taking antibiotics decreased the risk of future episodes by 69%, reduced their number by more than 50%, and significantly reduced the rate until the next attack (moderate‐certainty evidence). However, we found low‐certainty evidence that the protective effect of antibiotics for these three outcomes tailed off over time after treatment had been stopped. In addition, the beneficial effect of antibiotics was relevant for people with at least two past episodes of cellulitis within a time frame of up to three years.
We found low‐certainty evidence that there is no difference between antibiotics and no treatment/placebo for side effects and hospitalisation. The evidence did not allow for full exploration of the treatment's effect on length of hospital stay.
No serious adverse effects were reported, and common side effects included diarrhoea, nausea, rash (severe skin adverse reactions were not reported) and thrush. In three studies, adverse effects caused those taking part to stop taking the antibiotic. Three people taking erythromycin had abdominal pain and nausea, causing them to stop taking the treatment and to take penicillin instead. In one study, two people withdrew from treatment with penicillin because of diarrhoea or nausea. In another study, because of pain at the site of injection, around 10% of those taking part stopped taking intramuscular injections of benzathine penicillin.
None of the included studies measured quality of life or the development of antibiotic resistance.
Certainty of evidence
Evidence for the effects of antibiotics compared with no treatment or placebo on the recurrence, incidence rate and time to next episode of cellulitis under preventive treatment was of moderate certainty, and was limited by the small number of participants and events. Evidence for the remaining reported outcomes was of low certainty for the same reasons, as well as imprecise results.
Summary of findings
Summary of findings for the main comparison. Antibiotic prophylaxis compared to no treatment/placebo for the prevention of recurrent erysipelas and cellulitis.
| Antibiotic prophylaxis compared to no treatment/placebo for the prevention of recurrent erysipelas and cellulitis ‐ on prophylaxis | ||||||
| Patient or population: People with recurrent erysipelas or cellulitis Intervention: Antibiotic prophylaxis Comparison: No treatment/Placebo | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect (95% CI) | No of Participants (studies) | Quality of the evidence (GRADE) | Comments | |
| Assumed risk | Corresponding risk | |||||
| no treatment/placebo | Antibiotic prophylaxis | |||||
| Recurrence of cellulitis | Study population | RR 0.31 (0.13 to 0.72) | 513 (5 RCTs) | ⊕⊕⊕⊝ MODERATE 1 | Number needed to treat for 1 additional beneficial outcome (NNTB) is 6 | |
| 316 per 1000 | 98 per 1000 (41 to 227) | |||||
| Incidence rate of cellulitis | Study population | RR 0.44 (0.22 to 0.89) | 473 (4 RCTs) |
⊕⊕⊕⊝ MODERATE 1 | ‐ | |
| 43 fewer episodes of cellulitis per 1000 person‐months in treatment group (from 8 fewer to 60 fewer) | ||||||
| Time to next episode of cellulitis | Not estimable | HR 0.51 (0.34 to 0.78) | 437 (3 RCTs) | ⊕⊕⊕⊝ MODERATE 1 | ‐ | |
| Hospitalisation | Study population | RR 0.77 (0.37 to 1.57) | 429 (3 RCTs) | ⊕⊕⊝⊝ LOW 2 | ‐ | |
| 74 per 1000 | 57 per 1000 (27 to 116) | |||||
| Any adverse reactions | Study population | RR 0.87 (0.58 to 1.30) | 469 (4 RCTs) | ⊕⊕⊝⊝ LOW 3 | ‐ | |
| 287 per 1000 | 250 per 1000 (166 to 373) | |||||
| Quality of life | ‐ | |||||
| *The basis for the assumed risk (e.g. the median control group risk across studies) is provided in footnotes. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: Confidence interval; RR: risk ratio; HR: hazard ratio | ||||||
| GRADE Working Group grades of evidence High quality: Further research is very unlikely to change our confidence in the estimate of effect. Moderate quality: Further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate. Low quality: Further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate. Very low quality: We are very uncertain about the estimate. | ||||||
1We downgraded by one level because of imprecision due to the low number of events and the small sample size (optimal information size ‐ OIS).
2 We downgraded by two levels because of imprecision due to the low number of events and the small sample size (OIS) and the 95% confidence interval overlapping the line of no effect and ranging from benefit to harm.
3We downgraded by two levels because of imprecision due to the considerable low number of events and the small sample size (OIS).
We decided not to downgrade any of the outcomes for risk of bias as we decided it was not serious enough to affect the overall quality of the outcome.
Background
Description of the condition
Soft‐tissue infections are a common type of infection causing considerable disease. They account for up to 10% of all hospital admissions in western countries (Nathwani 2001; Vinh 2005) and are associated with high use of healthcare resources (Ostermann 2014). While these infections have many different causes, they are all the result of bacterial invasion through the skin barrier with a variable level of involvement of the soft tissue. Impetigo, boils, and erysipelas are superficial infections of the skin, while cellulitis involves deeper skin tissues (Stevens 2014). More advanced infections are myositis (muscle inflammation) and necrotising fasciitis (commonly known as the 'flesh‐eating disease') (Stevens 2014). Necrotising fasciitis is a severe, rapidly progressive bacterial inflammation that reaches the depth of the muscles and their surrounding tissues, leading to severe tissue destruction, which can be fatal (Stevens 2005; Stevens 2014).
Definition
Erysipelas is an infection that involves the superficial layer of the skin with significant inflammation of the lymphatic vessels (lymphangitis) (Bisno 1996). The resulting clinical picture is usually a skin area that is red, raised, and well‐demarcated from the surrounding normal skin (Raff 2016; Swartz 2004).
Cellulitis is a soft‐tissue infection that involves the deep subcutaneous (underlying) layer of the skin (Swartz 2004). The infected area, most commonly the leg, is characterised by local warmth, redness, swelling, and tenderness. Forty per cent of people with this infection can develop fever and illness (Morris 2008). Nevertheless, the lack of strict criteria or an optimal test for the diagnosis ('gold standard') of cellulitis and erysipelas can result in diagnostic errors, especially by non‐specialists (Arakaki 2014; David 2011; Levell 2011; Strazzula 2015; Weng 2016).
Although the two conditions are considered by some to be distinct, distinguishing erysipelas from cellulitis in daily clinical practice is challenging. Difficulties arise from a significant overlap between these two clinical patterns with regard to infective agents, risk factors, the areas of the body that are involved, and the depth of skin involvement (Bisno 1996). Some physicians use the terms 'erysipelas' and 'cellulitis' interchangeably, rendering this distinction even more problematic (Stevens 2005). Hence, in this review we will refer to both conditions as 'cellulitis', in accordance with the Cochrane Review on 'Interventions for cellulitis and erysipelas' (Kilburn 2010).
Epidemiology
Despite being a common medical problem, few studies provide good data on the incidence of cellulitis. Epidemiological surveys report an incidence that ranges from 0.2/1000 person‐years to 24.6/1000 person‐years in different populations (Ellis 2006; Lum 2002).
Although cellulitis affects the lower limbs in most cases, it can also affect other areas, such as the upper limbs, face, ears, and trunk (Ellis 2006). Upper limb cellulitis mostly occurs in women after surgical treatment for breast cancer following lymphatic system damage (Simon 1992). In this group, cellulitis affects up to 24% of women (Harris 2001). As more conservative breast surgery has been performed, this type of cellulitis tends to be more localised to the breast, rather than the entire upper limb (Mertz 1998; Miller 1998).
Microbiology
The most common causative agents of cellulitis are part of the skin's microbial flora (natural bacterial inhabitants). It is usually caused by streptococcal infection (Bruun 2015), but an infection by Staphylococcus aureus is also possible, and more commonly seen after surgery, penetrating trauma, or in wounds (Bernard 1987; Eriksson 1996; Jorup‐Rönström 1986; Siljander 2008; Swartz 2004). Unlike cellulitis, erysipelas is chiefly caused by Group A streptococci, infrequently involving other species of streptococci (groups B, C, or G) or Staphylococcus aureus (Mandell 2010).
Less common infective organisms include Streptococcus pneumoniae, Haemophilus influenzae, gram negative bacilli, and anaerobes. These pathogens are more likely to be involved in specific epidemiological settings, such as following injury, burns, and other co‐existing diseases (e.g. diabetes, cancer, malnutrition) (Bisno 1996; Brook 1995; Carratala 2003). Pseudomonas aeruginosa is involved in many types of soft tissue infections, frequently targeting vulnerable populations, and also those who have been exposed to contaminated hot tubs, loofah sponges, or nail punctures (Bowler 2001; Eron 2003). Pseudomonas aeruginosa has been shown to be the pathogen associated with the development of shock and death in some people with cellulitis, probably due to a combination of its high virulence and their poor health status (Carratala 2003).
Other cases involve unique types of bacteria. Vibrio vulnificus is associated with exposure to seawater, and Aeromonas hydrophila with freshwater exposure (Swartz 2004). Several organisms are characteristic of animal and human bites: Pasteurella multocida and Capnocytophaga canimorsus have been implicated following animal bites, and Eikenella corrodens following human bites or fist injuries (Stevens 2005).
The value of microbiological culture in the management of cellulitis is limited. Needle aspirations (samples), taken from the infected skin areas and then cultured, are positive for bacterial growth in only 10% of cases (CREST 2005). A higher yield is noticed in surgically‐removed full‐thickness skin biopsies or other tissue specimens from the lesion, with successful bacterial growth in 20% to 30% of cases (Duvanel 1989; Hook 1986). Blood cultures are even less productive, with only 2% to 4% of cultures showing positive bacterial growth. Furthermore, blood culture samples are often contaminated by irrelevant commensals (bacteria living at the same site, but with no harmful or beneficial effects) (Swartz 2004). The diagnosis of cellulitis is therefore based primarily on clinical features. Blood cultures might be useful when the risk of bacteraemia (bacteria in the blood) is high, such as in cases of severe infection or sepsis, or in cellulitis secondary to lymphoedema (an accumulation of lymph in the tissues) (Woo 2000).
Risk factors
Retrospective studies exploring risk factors for cellulitis have reported its association with several factors, including obesity, diabetes mellitus, immunosuppression, and alcoholism (Bartholomeeusen 2007; Koutkia 1999; Lazzarini 2005; Quirke 2016).
The physical condition of a person's skin may play a central role in predisposition to cellulitis, especially on the lower limb. Toe web intertrigo (skin maceration and inflammation, often due to a fungal infection), cracks on the soles of the feet, oedema (swelling that may be due to a damaged lymphatic system, e.g. lymphoedema), leg ulcers, prior trauma (injury), history of venous surgery, and venous insufficiency (caused by a malfunctioning of the venous system of the legs due to valves that are no longer able to pump blood back to the heart) have all been associated with an increased risk of cellulitis (Bjornsdottir 2005; Dupuy 1999; Halpern 2008; Mokni 2006; Pavlotsky 2004; Roujeau 2004). A previous episode of cellulitis is another important risk factor for leg cellulitis (Dupuy 1999; Halpern 2008; Roujeau 2004).
There is a strong association between cellulitis and foot dermatomycosis (fungal infection of the foot). A fungal infection causes breaching of the epidermal layer and fosters bacterial overgrowth, thus facilitating the entry of bacteria to cause skin infection (Roldan 2000; Semel 1996). This pathogenesis probably underlies the strong association between cellulitis and clinical and microscopically‐proven tinea pedis (athlete's foot) (Dupuy 1999; Roujeau 2004). This relationship, together with the high prevalence of tinea pedis in the general population, highlights the significant impact of fungal foot infection on the occurrence of cellulitis (Dupuy 1999; Roujeau 2004).
Recurrent cellulitis
Recurrent cellulitis is a frequent complication of single‐episode cellulitis (Cox 2006; Stoberl 1987). The incidence of recurrent cellulitis varies between studies. It has been noted that 10% to 30% of people who suffer an episode of cellulitis experience repeated attacks across different time intervals (Cox 1998; Dupuy 1999; Ellis 2006; Eriksson 1996; Jorup‐Rönström 1987). In more recent studies with a longer follow‐up period, there was a considerably higher percentage of recurrence. A retrospective analysis by Pavlotsky 2004 carried out on hospitalised patients during a three‐year period showed that more than 45% of admissions of people with cellulitis were due to recurrent episodes. Similar results were also reported by Cox 2006 in a community‐based survey. It is therefore remarkable that only a small number of studies have examined the cause of recurrent cellulitis.
Largely a disease of the lower limb, recurrent cellulitis is thought to be caused by repeated bacterial invasion of the skin through breaches in its protective barrier (Pavlotsky 2004). Accordingly, the most important risk factors for recurrent cellulitis are the local physical factors listed above (Cox 2006; Dupuy 1999; Jorup‐Rönström 1987; Karppelin 2010; Lewis 2006; McNamara 2007; Pavlotsky 2004; Wang 1997).
The primary general risk factors for recurrent cellulitis are lymphoedema, obesity, and a history of cancer (Cox 2006; Dupuy 1999; Karppelin 2010; Lewis 2006; McNamara 2007; Pavlotsky 2004). Contradictory results were obtained for the influence of smoking on recurrent cellulitis (Karppelin 2010; Lewis 2006; Pavlotsky 2004). Previously thought to encourage cellulitis and its recurrence, a weak association has been noted between diabetes as well as alcoholism and recurrent cellulitis (Karppelin 2010; Lewis 2006; Pavlotsky 2004). Nevertheless, a recent report by Karppelin 2013 underscores the significance of diabetes in recurrent cellulitis and also suggests an association between psoriasis and recurrence, as well as surgical removal of tonsils and recurrence of this infection.
Description of the intervention
Small case series and case reports have advocated the use of antibiotic treatment for cellulitis prophylaxis (Table 2) (Babb 1966; Bitnun 1985; Duvanel 1985; Ferrieri 1973; Thind 1985). According to these reports, the use of a long‐term preventive therapy with penicillin was effective in reducing the rate of cellulitis recurrence. While acknowledging the value of antibiotic prophylaxis, different experts have called for the rigorous control of predisposing factors (Baddour 2000; Cox 2006; Lewis 2006; McNamara 2007; Pavlotsky 2004; Swartz 2004). Scrupulous skin care, oedema reduction using compression stockings and diuretics (Table 2), antifungal therapy, and proper footwear have all been proposed as part of prophylactic regimens for recurrent cellulitis (Baddour 2000; Pauszek 1991; Pierce 1992; Stalbow 2004; Swartz 2004). Prophylaxis based on the anti‐inflammatory effects of selenium have been proposed by Kasseroller (Kasseroller 1996; Kasseroller 1998), who demonstrated the benefit of selenium in a randomised controlled trial among women following gynaecological cancer treatment, in most cases following mastectomy (Table 2).
1. Glossary of terms.
| Medical term | Explanation |
| Ambulatory | Ambulatory is when the patient can walk and is not bedridden. When referring to medical care it means that it is being provided to patients that are not hospitalised (outpatients) |
| Block randomisation | A method of randomisation that ensures allocation of participants into roughly equal sizes of comparison groups |
| Clostridium difficile | A bacterium that causes inflammation of the colon (colitis), typically resulting in diarrhoea, and is strongly associated with the use of antibiotics |
| Comorbidity | The presence of one or more diseases or conditions other than those of primary interest |
| Contralateral | On the opposite side of the body (e.g. a repeat episode of leg cellulitis can recur in the same leg [see 'ipsilateral'] or the other, contralateral leg |
| Control event rate (CER) | The rate at which events of interest (i.e. episodes of cellulitis in our review) occur in the control group of the study |
| Diuretics | Commonly known as "water pills" these are drugs that help the body to eliminate unneeded water and salt through the urine |
| Epidemiology | The study of the health of populations and communities, not just particular individuals |
| Erythema | Redness of the skin caused by increased blood flow. Often a sign of inflammation or infection |
| Filariasis | A disease caused by infection with worms, usually in tropical and subtropical areas of the world. The worms reside in the lymphatic system of the affected person and interfere with the drainage of the lymph, subsequently causing a significant swelling of the involved limb |
| Filarial lymphoedema | see Filiariasis |
| Folliculitis | Inflammation of hair follicles |
| Furunculosis | Deep form of inflammation of the hair follicles resulting in lumps caused by the accumulation of pus (boils) |
| Gastrointestinal | Relating to the stomach and the intestines |
| Incidence rate/Incidence rate ratio | The number of new occurrences of events in a population divided by its time period at risk; Incidence rate ratio is the ratio of two incidence rates |
| Ipsilateral | On the same side of the body; as opposed to 'contralateral' |
| Mastectomy | Surgical removal of one or both breasts |
| Outpatient/Inpatient | Outpatient is a person that is being treated without being hospitalised overnight and visits the physician in the clinic, hospital or other facility; compared with an inpatient who requires an overnight stay in hospital for medical treatment |
| Person‐months | The sum of the number of months each participant in the trial has been under observation (treated/followed) |
| 'Per protocol'/Intention‐to‐treat (ITT) analyses | 'Per protocol' analysis compares participants in a study based on the treatment they actually took and includes only those patients who completed the treatment originally allocated, as opposed to intention‐to‐treat analysis that compares participants on the basis of their random assignment to groups (treatment or placebo), regardless of adherence to treatment |
| Prophylaxis | Preventive treatment for disease |
| Retrospective cohort study | An observational study in which a defined group of people (the cohort) is followed over time. A retrospective cohort study identifies persons from past medical records and follows them from the time of those records to the present |
| Sensitivity analysis | An analysis used to determine how sensitive the results of a study or systematic review are to changes in how it was done |
| Tinea pedis | Fungal infection of the foot (athlete's foot) |
| Tonsillectomy | Surgical removal of tonsils |
To the best of our knowledge, 12 guidelines for cellulitis prophylaxis have so far been issued (ALA 2015; BLS 2016; CREST 2005; Draijer 2008; Duodecim 1999; Eron 2003; Esposito 2011; ILF 2006; ISL 2013; NICE 2005; SFD 2000; Stevens 2005).
All documents call for reducing predisposing conditions. Avoiding dry and cracked skin, treating macerated skin and fungal infections, and using compressive dressings are agreed. Antibiotic prophylaxis is also advocated by the different groups. Long‐term antibiotic prophylaxis with oral or intramuscular penicillin or macrolides is recommended, while the British Lymphology Society (BLS 2016), the Australasian Lymphology Association (ALA 2015), the International Lymphoedema Framework (ILF 2006), the Dutch College of General Practitioners (Draijer 2008), the Clinical Resource Efficiency Support Team (CREST 2005) and NICE (National Institute for Health and Clinical Excellence) (NICE 2005) groups advise initiating treatment after the second cellulitis episode. The International Society of Lymphology (ISL 2013), the Infectious Diseases Society of America (Stevens 2005), the French Society of Dermatology (SFD 2000) and the working group set up by the Finnish Medical and the Finnish Dermatological Societies (Duodecim 1999) indicate that repeated, frequent or several episodes of treatment are necessary. The American guidelines are classified as grade B‐II recommendation (B refers to moderate evidence to support a recommendation for use that should generally be offered, and II means the evidence is available from well‐conducted non‐randomised studies). Seven groups propose an alternative to antibiotic prophylaxis with early self‐initiated antibiotic treatment (ALA 2015; BLS 2016; CREST 2005; Draijer 2008; Eron 2003; ILF 2006; Stevens 2005).
These recommendations are mostly supported by observational studies and expert opinion, and lack a systematic analysis of high‐quality research.
How the intervention might work
A plausible strategy aiming to prevent recurrent attacks of cellulitis is likely to rely on targeting the causative bacterial agents of cellulitis, i.e. streptococci and Staphylococcus aureus, or on controlling risk factors for recurrence that are amenable to change.
Antibiotic treatment is aimed at eradicating and preventing the growth of bacteria. However, the consistent difficulty isolating bacteria from the infected skin (Crisp 2015) might indicate that once cellulitis is initiated, it is promoted and perpetuated by bacterial toxins, pointing to another possible preventive mechanism: inhibition of bacterial toxins and their production. Such a mode of action is attributed to clindamycin (Goscinski 2006; Sawai 2007), an antibiotic that is often used in infections that are considered as toxin‐mediated. A cellulitis vaccine would naturally be the ultimate preventive measure for people at high risk for recurrent cellulitis, providing an acquired immunity against the disease.
Another preventive approach for recurrent cellulitis is to treat its underlying risk factors. The primary mechanism of action of different moisturisers and the treatment of fungal foot infection is probably by maintaining the skin barrier and preventing its breach by pathogenic bacteria.
Chronic lymphoedema and oedema, characterised by the accumulation of fluid in the tissues (usually of the limbs), foster the growth of bacteria and fungi and impair the body's ability to produce an appropriate local immune response (Mallon 1997; Mortimer 2014). Different methods have been described to treat lymphoedema and oedema, most of which are non‐operative and act principally by mechanical compression and increasing of blood and lymphatic flow: compressive bandages, elastic stockings, physical therapy and exercise. Diuretic treatment generally works through the production of urine and shifting of the body's fluids from the swollen tissues into the blood vessels, and weight loss works by reducing limb volume and the facilitation of vascular flow and lympathic drainage (Arsenault 2011; Ezzo 2015; Szolnoky 2014). Surgical techniques to treat lymphoedema have slowly been introduced, aiming to reconstruct a lymphatic drainage system and to remove overgrowing tissue, including the removal of fat tissue (liposuction) (Campisi 2015; Szolnoky 2014).
Why it is important to do this review
Cellulitis is a common disease treated in primary‐care settings, emergency rooms, and hospital departments by physicians of various specialties, including internal medicine, infectious diseases, dermatology, oncology, and different surgical subspecialties. Many people with cellulitis require hospitalisation with a relatively long length of stay for what can be regarded, in most cases, as a non‐fatal, simple‐to‐treat disease.
A significant proportion of all hospital admissions for cellulitis are for people experiencing recurrent episodes (Pavlotsky 2004). This population tends to occupy more bed days compared with the non‐relapsing group, which adds considerably to the costs and burden of this disease (Karppelin 2010). The economic burden of cellulitis was determined by a Dutch study, with an estimated total direct cost of more than EUR 17 million in 2001 (Goettsch 2006).
Despite this considerable burden, there is a lack of high‐quality, evidence‐based information regarding optimal treatments for the prevention of recurrent cellulitis. The purpose of this review is to summarise high‐quality research, giving clinicians the tools to provide more evidence‐based treatment for people with recurrent cellulitis, as well as delineating areas of focus for future research.
The plans for this review were published as a protocol 'Interventions for the prevention of recurrent erysipelas and cellulitis' (Dalal 2012).
Objectives
To assess the beneficial and adverse effects of antibiotic prophylaxis or other prophylactic interventions for the prevention of recurrent episodes of cellulitis in adults aged over 16.
Methods
Criteria for considering studies for this review
Types of studies
Randomised controlled trials (RCTs).
Types of participants
Adults older than 16 years after an episode of erysipelas and cellulitis (hereafter referred to as 'cellulitis').
We excluded people with cellulitis or erysipelas secondary to filarial lymphoedema.
When a study included participants with various types of skin infections, we included the study only if it had reported separately on cellulitis or if authors provided data on the subgroup with cellulitis. In cases where results had not been given separately, we excluded the study if more than 30% of participants had infections other than cellulitis.
Types of interventions
Interventions
Any intervention aimed at preventing cellulitis. Specifically, we aimed to assess the following interventions:
antibiotic prophylaxis;
anti‐inflammatory prophylaxis;
compression stockings;
treatments for toe web intertrigo (including antifungal treatments for tinea pedis);
treatments for venous insufficiency;
other interventions to reduce leg oedema.
If we found other relevant interventions, we also included them. We accepted any duration of intervention.
Control
No treatment, placebo, or another intervention as defined above, or the same intervention with a different treatment schedule (dose, frequency, timing, duration).
We contacted authors for results relating to cellulitis if they were not reported separately.
Types of outcome measures
Primary outcomes
Recurrence of cellulitis (number of participants with at least one recurrent episode within a follow‐up period of at least three months after randomisation) under prophylactic treatment. When specified, we considered a recurrence as a repeat episode of cellulitis in the same limb.
For all outcomes, we extracted data on physician‐diagnosed cellulitis, preferably that of a dermatologist. However, if the diagnoses were based on the assessment of other physicians, we accepted these diagnoses and documented the person assessing the outcomes. We accepted the study definitions of cellulitis and documented the differences between studies.
The primary time point for analysis of all outcomes was at the end of the treatment phase ('on prophylaxis'). Secondary time points for analysis were: after prophylaxis discontinuation ('post‐prophylaxis') and at the end of follow‐up ('overall').
Secondary outcomes
Incidence rate of cellulitis, i.e. episodes of recurrent cellulitis per person‐months of follow‐up in the trial under prophylactic treatment; also assessed after prophylaxis discontinuation ('post‐prophylaxis') and at the end of follow‐up.
Time to next episode of cellulitis among all trial participants under prophylactic treatment; also assessed after prophylaxis discontinuation ('post‐prophylaxis') and at end of follow‐up.
Number of participants requiring hospitalisation and number of hospital days.
Quality‐of‐life measures, using the score or scale reported in the study.
Development of resistance to antibiotics. In studies that used preventive antibiotic treatment, we evaluated this outcome by assessing laboratory‐proven growth of resistant bacteria to the study drug/s and infections (cellulitis or other) caused by such bacteria.
Adverse reactions, including allergic reactions, skin reactions, and pseudomembranous colitis (mainly for antibiotic prophylaxis interventions).
Mortality.
Search methods for identification of studies
We aimed to identify all relevant RCTs regardless of language or publication status (published, unpublished, in press, or in progress).
Electronic searches
We searched the following databases up to 23 June 2016:
the Cochrane Skin Group Specialised Register using the following terms: (erysipelas or cellulitis or "ignis sacer" or "holy fire" or "st anthony's fire" or impetigo or "soft tissue infection*" or staphylococc* or streptococc*);
the Cochrane Central Register of Controlled Trials (CENTRAL) 2015, Issue 2, in the Cochrane Library using the search strategy in Appendix 1;
MEDLINE via Ovid (from 1946) using the strategy in Appendix 2;
Embase via Ovid (from 1974) using the strategy in Appendix 3; and
Latin American and Caribbean Health Science Information database (LILACS) (from 1982) using the strategy in Appendix 4.
Trials registers
We searched the following trials registers up to 22 August 2016, using the terms “cellulitis” or “erysipelas”:
the ISRCTN registry (www.isrctn.com);
the World Health Organization International Clinical Trials Registry Platform (ICTRP) (apps.who.int/trialsearch/);
the Australian New Zealand Clinical Trials Registry (www.anzctr.org.au);
ClinicalTrials.gov (www.clinicaltrials.gov); and
the EU Clinical Trials Register (www.clinicaltrialsregister.eu/).
Searching other resources
Reference lists
We checked the reference lists of included studies and review articles for further references to relevant trials.
Conference proceedings
We searched the following major dermatological conference proceedings for relevant studies by searching the formal electronic journal of each academy up to 22 August 2016:
the American Academy of Dermatology annual conference proceedings (from 1990); and
the European Academy of Dermatology and Venereology annual conference proceedings (from 1991).
Grey literature
We searched BIOSIS Previews (from 1990) for relevant studies up to 10 March 2015, using the terms “cellulitis” or “erysipelas”.
Correspondence
We contacted the authors of potentially relevant and unpublished trials to obtain full trial results.
Data collection and analysis
Selection of studies
Two authors (AD and SR) independently reviewed the titles and abstracts identified by the literature search. The same authors later reviewed the full‐text versions of the eligible studies. We evaluated the studies according to our preset criteria, resolving disagreements by referral to a third author (MP).
Data extraction and management
AD and MS independently extracted data from the included studies onto data extraction forms, discussing any differences with a third author (MP). AD checked and entered data into Review Manager 5. We extracted the following data:
(1) Trial characteristics
publication status (published, published as abstract, unpublished);
year (defined as recruitment initiation year) and country/s of study;
setting (hospital/outpatient);
design (method of allocation generation and concealment, blinding);
intention‐to‐treat analysis (performed, possible to extract, efficacy analysis);
cellulitis case definition (diagnosis and by whom);
exclusion criteria (age of participants, comorbidities, risk factors or their severity, infective agents, previous use of antibiotics);
duration of study follow‐up (from intervention and from the end of intervention);
funding;
ethical committee approval and informed consent.
(2) Baseline participant characteristics
number of participants eligible;
number of participants randomised;
mean age and sex distribution;
area of body involved;
number of cellulitis attacks and observation period prior to intervention;
possible risk factors for cellulitis and estimation of their severity:
percentage of participants with diabetes and its level of control ‐ HbA1C at baseline and during the trial
percentage of participants with oedema of any cause at baseline and during the trial. Severity was extracted descriptively or by any other score
percentage of participants with venous drainage impairment, diagnosed clinically or by imaging studies. Severity was recorded for clinical findings and imaging studies
percentage of participants with diagnosis of peripheral vascular disease or arterial insufficiency. Severity was recorded by the ankle brachial pressure index
mean body mass index (weight in kilograms, divided by height in metres squared) at baseline and during the trial. Alternatively, we extracted the percentage of participants with a diagnosis of overweight/obesity
any type of skin injury (i.e. surgery, ulcers, insect bites, burns, etc) prior to and during the trial follow‐up
diagnosis of toe web intertrigo or tinea pedis and antifungal treatment
co‐existing skin diseases
immunodeficiency ‐ hereditary or acquired, secondary to medications, chronic infections, malignancies, or chronic diseases
smoking status and alcohol consumption
(3) Interventions
type of intervention (if antibiotics or other medications) ‐ drug, dosage, schedule, way of administration;
concomitant medical advice, by any means, regarding diet, weight loss, physical activity, skin care, oedema reduction, or any other recommendations;
adherence.
(4) Outcomes
As defined above in our Methods section. If they were not reported numerically, we extracted outcomes from graphs or figures presented in the publications (by counting pixels).
Assessment of risk of bias in included studies
In the quality assessment, AD and MS independently evaluated the following components individually, since there is evidence that these are associated with biased estimates of treatment effect (Jüni 2001). We discussed any differences with a third author (MP). We used the criteria described in theCochrane Handbook for Systematic Reviews of Interventions (Table 8.5.d) (Higgins 2011) for quality assessment, categorising each study as high risk of bias, low risk of bias, or unclear risk of bias.
(a) Randomisation generation (method of allocation generation): we defined this as low risk of bias when the allocation procedure protects against biased allocation to the comparison groups
(b) Randomisation concealment (method of allocation concealment): we defined this as low risk of bias when neither clinicians nor participants were aware of future allocation
(c) Masking as blinding of participants/investigator and of the outcome assessor: we defined this as low risk of bias when events were as follows:
Blinding of participants and key study personnel was ensured, and it is unlikely that the blinding could have been broken.
There was no blinding or incomplete blinding, but the review authors judged that the outcome was not likely to be influenced by lack of blinding.
Blinding of outcome assessment was ensured, and it is unlikely that the blinding could have been broken.
There was no blinding of outcome assessment, but the review authors judged that the outcome measurement was not likely to be influenced by lack of blinding.
(d) Loss to follow‐up (information about dropouts and withdrawals, and the analysis of these): we defined this as low risk of bias when it was clear that all of the participants in the trial were analysed (i.e. 0% lost to follow‐up). Additionally, when participants had been analysed based on the intention‐to‐treat principle (where participants were analysed based on the treatment to which they were randomised).
(e) Selective reporting (reporting bias due to selective outcome reporting): we defined this as low risk of bias when the study protocol was available, and all of the study's prespecified (primary and secondary) outcomes that were of interest to the review had been reported in the prespecified way. Or if the study protocol was not available, but it was clear that the published reports included all expected outcomes, and these were reported numerically (rather than as "significant" or "non‐significant").
(f) We also assessed the following items:
Baseline imbalance: we defined this as low risk when the baseline characteristics of the treatment groups were adequately addressed and there was no systematic imbalance between groups;
Early termination: we defined this as low risk when the trial ended by achieving a predefined sample size or stopped early according to predefined stopping rules. We defined high risk of bias as trials with premature termination of the trial, contrary to predefined stopping rules. In other cases (no sample size definition) we defined early termination as unclear risk of bias;
Other potential bias: we defined this as low risk of bias when the study appeared to be free of other sources of bias.
Measures of treatment effect
For dichotomous outcomes, we calculated risk ratios (RRs) and corresponding 95% confidence intervals (CIs) from individual studies. When the outcome to be assessed was episodes (outcomes occurring more than once per participant) we calculated rate ratios, defined as episodes/person‐month, and the corresponding 95% CI.
For time‐to‐event outcomes, we attempted to extract the hazard ratio (HRs) with a 95% CI from included trials. If this had not been reported, we estimated it from Kaplan‐Meier curves or other statistics reported in the study (Parmar 1998; Tierney 2007).
Unit of analysis issues
We considered participants as the unit of analysis and not limbs; i.e. recurrence included both ipsilateral and contralateral episodes of cellulitis. We did not expect cross‐over trials to be carried out, since this design would not be suitable for the evaluation of prophylactic treatment (mainly because the first treatment may significantly influence the course of the disease).
Dealing with missing data
We performed an intention‐to‐treat (ITT; see Table 2) analysis when possible, and only used per protocol data, documenting it accordingly, if information was not specified in the study or retrieved from trial investigators. One author (AD) contacted trials authors to ask for missing data.
Assessment of heterogeneity
Where we identified enough studies assessing the same intervention and performing the same comparison to combine them, we assessed heterogeneity visually by inspecting the forest plot and looking at the magnitude and direction of the study results; if relevant, we reported on the I² statistic value (Higgins 2011).
Assessment of reporting biases
The capacity of funnel plots to detect bias in a small number of included studies is limited (Egger 1997). As expected, we were unable to assess reporting bias using funnel plots, due to the small number of studies in each comparison.
Data synthesis
We performed a meta‐analysis if we found more than one study assessing the same intervention and outcome. We pooled data for dichotomous outcomes using the Mantel‐Haenszel fixed‐effect model to calculate a treatment effect across trials, when heterogeneity was low and the beneficial effects of small studies would be overestimated by the random‐effects analysis. If heterogeneity between trials was significant (I² > 50%) we used the random‐effects model. We expressed results as the RR with 95% CI. We pooled HRs with 95% CIs for time‐to‐event outcomes, using the inverse variance method in a fixed‐effect or random‐effects model, according to our previously‐mentioned evaluation.
Subgroup analysis and investigation of heterogeneity
We planned to explore reasons for substantial heterogeneity (I² > 50%) in any meta‐analyses we performed, using the following subgroups:
lower versus upper limb cellulitis;
participants with versus those without limb oedema at baseline (randomisation);
participants with a single episode of cellulitis versus those with at least two episodes at baseline (randomisation); and
different types of antibiotic.
When the planned analyses were irrelevant or data were scarce, we discuss narratively the potential factors contributing to heterogeneity.
Sensitivity analysis
Where we found substantial heterogeneity in the meta‐analyses, we planned to explore the reasons by sensitivity analyses, including the quality of included studies.
For all outcomes, we primarily extracted data on physician‐diagnosed cellulitis, and as a secondary analysis, we used the data on participant‐reported episodes.
'Summary of findings' table
We created a 'Summary of findings' table that reported the following main outcomes at the primary time point for analysis (end of prophylaxis phase ('on prophylaxis')): recurrence of cellulitis; incidence rate of cellulitis; time to next episode of cellulitis; hospitalisation; any adverse reactions; and quality of life.
We adopted the GRADE approach to assess the quality of evidence using five factors: study limitations, indirectness, inconsistency, imprecision, and publication bias. If we found a reason to change the grading of the quality of evidence, we detailed it in the footnotes.
We used methods and guidance described in chapter 12 of the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011) and in the GRADE handbook (Schünemann 2013), and we used the GRADEproGDT web‐based software (available at www.gradepro.org) to produce the 'Summary of findings' table.
Results
Description of studies
Results of the search
We identified 6139 records, using an inclusive and comprehensive search strategy. After removing duplicates, we had 5995 records. We excluded 5979 records based on titles and abstracts, and sought the full text of 16 studies. We retrieved the full text of 15 studies. For one (Ratnikova 1991) we had only an abstract, and we have listed the details of this study in Characteristics of studies awaiting classification. We hope to examine its full text in a future update of this review. Of the remaining 15 papers, we excluded eight (see Characteristics of excluded studies). We included six studies reported in seven publications (two reports represent one study and we included them under a single trial ID: Kasseroller 1998). We have summarised the screening process in the 'Study selection flow diagram' (Figure 1).
1.
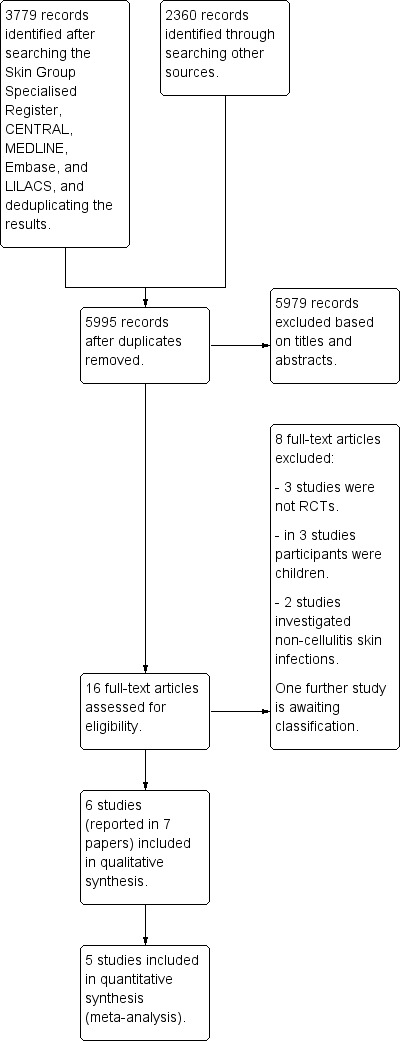
Study selection flow diagram.
Included studies
The six studies cover 595 participants, of which 573 were evaluated.
Design
All of the studies were randomised controlled trials using a two‐arm, parallel‐group design. Three studies had a placebo arm (Kasseroller 1998; Thomas 2012; Thomas 2013) and none of the included studies was a cross‐over trial.
Of the six included studies, four were single‐centre (Chakroun 1994; Kasseroller 1998; Kremer 1991; Sjöblom 1993) and two were multicentre with 20 (Thomas 2012) and 28 sites (Thomas 2013). The sample sizes of the studies ranged from 32 to 274 participants.
Setting
The included studies were carried out in five countries: UK (two studies: Thomas 2012; Thomas 2013), Sweden (Sjöblom 1993), Tunisia (Chakroun 1994), Israel (Kremer 1991) and Austria (Kasseroller 1998).
Four studies were hospital‐based (Chakroun 1994; Sjöblom 1993; Thomas 2012; Thomas 2013 ), one implied it was secondary‐care‐based (Kremer 1991), and one was conducted within a private clinic (Kasseroller 1998).
Most of the included studies were published in English. One study was printed in French (Chakroun 1994), and one report of a single trial (Kasseroller 1998) was published in German (Kasseroller 1996).
The Thomas 2012 and Thomas 2013 studies were led by the same research team.
Participants
Of the evaluable participants, 200 were men and 373 women.
The mean age of participants in the included studies was between 50 and 70 (Kasseroller 1998; Kremer 1991; Sjöblom 1993; Thomas 2012; Thomas 2013). In one study the mean age of participants was 46.2 (Chakroun 1994).
The number of previous episodes of cellulitis at recruitment to trial was at minimum: four episodes in one study (Kasseroller 1998), two episodes in three studies (Kremer 1991; Sjöblom 1993; Thomas 2013) and one episode in one study (Thomas 2012). The time interval to recurrence of cellulitis before trial entry was three years in two studies (Sjöblom 1993; Thomas 2013) and in other studies two years (Kasseroller 1998) and one year (Kremer 1991). Thomas 2012 included participants with one previous episode within 12 weeks from inclusion.These data were not reported in the Chakroun 1994 study.
In five of the included studies the vast majority of participants had past episodes of leg cellulitis at baseline (Chakroun 1994; Kremer 1991; Sjöblom 1993; Thomas 2012; Thomas 2013). In two of these studies three participants in the control group had upper limb cellulitis before entering the trial (two participants in the Kremer 1991 study and one participant in the Sjöblom 1993 study). Kasseroller 1998 investigated upper limb cellulitis in women after mastectomy (Table 2).
Four out of six studies described the clinical criteria for the diagnosis of past episodes of cellulitis at inclusion: in two studies the diagnosis of cellulitis was based on fever and local signs of skin infection/inflammation (Chakroun 1994;Sjöblom 1993); in Thomas 2012 and Thomas 2013 the diagnosis was either made by a physician or validated according to detailed clinical criteria of skin infection/inflammation, and any doubt over the certainty of the diagnosis was grounds for exclusion. One study mentioned parameters for diagnosis (physical examination and blood test markers) but it is unclear whether these were assigned to past episodes (Kasseroller 1998), and one study did not state criteria for the definition of cellulitis (Kremer 1991).
The baseline comorbidities of participants were reported in five out of six studies (Chakroun 1994; Kremer 1991; Sjöblom 1993; Thomas 2012; Thomas 2013), with Kasseroller 1998 not reporting any comorbidity data. The comorbidity profile of participants within the same study was similar. Variable data were reported for comorbidities from different studies: 5% to 25% of participants had diabetes mellitus (Chakroun 1994; Kremer 1991; Sjöblom 1993; Thomas 2012; Thomas 2013), 5.5% to 90% had venous insufficiency (Chakroun 1994; Sjöblom 1993; Thomas 2012; Thomas 2013), 10% to 68% had leg oedema (Chakroun 1994; Sjöblom 1993; Thomas 2012; Thomas 2013), 30% to 50% of participants had fungal foot infection (Chakroun 1994; Kremer 1991; Thomas 2012; Thomas 2013), and most participants were overweight (Chakroun 1994; Thomas 2012; Thomas 2013).
Interventions
Five studies evaluated antibiotic treatment (Chakroun 1994; Kremer 1991; Sjöblom 1993; Thomas 2012; Thomas 2013) and one study evaluated treatment with oral selenium (Kasseroller 1998).
1. Antibiotic therapy
Penicillin was used in four studies (Chakroun 1994; Sjöblom 1993; Thomas 2012; Thomas 2013) and erythromycin was used in one study (Kremer 1991). Three studies evaluated oral ingestion of penicillin (penicillin V) (Sjöblom 1993; Thomas 2012; Thomas 2013), at a dose of 250 mg twice a day in two studies (Thomas 2012; Thomas 2013) and 2 grams to 4 grams a day in one study (depending on participant's weight: 1 gr twice a day if < 90 kg; 1 gr + 2 gr a day if 90 kg to 120 kg; 2 gr twice a day if > 120 kg) (Sjöblom 1993). In Chakroun 1994 penicillin (benzathine penicillin) was injected into the muscle at a dose of 1.2 million units every 15 days. Kremer 1991 used erythromycin at a dose of 250 mg twice a day given by mouth. In two studies the control group received placebo (Thomas 2012; Thomas 2013).
In three out of four studies assessing penicillin, participants who were allergic to the drug were excluded from the trial (Chakroun 1994; Thomas 2012; Thomas 2013) and in one these participants received an alternative treatment with erythromycin at a dose of 250 mg to 500 mg twice a day, depending on participant's weight (Sjöblom 1993). Kremer 1991 did not refer participants with known allergy to erythromycin.
The duration of antibiotic therapy varied from six to 18 months across studies (Chakroun 1994; Kremer 1991; Sjöblom 1993; Thomas 2012; Thomas 2013). Treatment periods varied significantly among participants in the Chakroun 1994 study and lasted one to 38 months, with a mean duration of 11.6 months.
In four studies medical advice or treatment were given to both treatment and control arms: participants with fungal foot infection were treated with antifungal agents in one study (Kremer 1991), local skin care and the use of compression stockings/elastic bandages were recommended in one study (Sjöblom 1993), and treatment of predisposing factors, such as fungal foot infection, was given in two studies (stated as "normal clinical practice") (Thomas 2012; Thomas 2013).
Among the six included studies, two studies reported follow‐up, with follow‐up periods for the majority of participants of 18 months to two years after the treatment stopped (Thomas 2012; Thomas 2013).
2. Selenium therapy
Kasseroller 1998 used oral ingestion of sodium selenite solution given at daily doses of 1000 μg (micrograms) in the first week, 300 μg in the second and third weeks and 100 to 200 μg (depending on participant's weight) from the fourth to the 15th week. The participants in the control group were given physiological salt solution. In the first three weeks all participants were admitted to the medical centre for an intensive "congestive relief" therapy that consisted of manual lymph drainage, compression bandage, meticulous skin care, therapeutic exercises and high‐voltage therapy. No follow‐up period was reported after 15 weeks.
Outcomes
Primary outcome
All included studies reported the number of participants with recurrent episodes of cellulitis. Of the six studies, two reported on cellulitis recurrence after the discontinuation of treatment ('post‐prophylaxis') (Thomas 2012; Thomas 2013).
Five out of six studies mentioned the clinical findings for the diagnosis of cellulitis or its confirmation (Chakroun 1994; Kasseroller 1998; Sjöblom 1993; Thomas 2012; Thomas 2013).
In two studies the diagnosis of cellulitis was based on fever and local signs of skin infection/inflammation, and was established by an infectious diseases specialist (Chakroun 1994; Kremer 1991).
In Thomas 2012 and Thomas 2013 diagnosis of cellulitis before trial entry was established by a dermatologist, either by examination of the participant or by validation of the diagnosis from medical records according to prespecified criteria that indicated a skin infection/inflammation (such as pain, local warmth, tenderness, swelling, redness); during the trial period a new episode of cellulitis was defined as reported by the participant and confirmed by a medical practitioner. New episodes that were only self‐reported were included in sensitivity analysis (Table 2).
In Kasseroller 1998 diagnosis of cellulitis prior to enrolment was carried out in general or university hospitals; after enrolment the diagnosis was based on findings of the physical examination and blood test markers of inflammation. The report did not state who made the diagnosis or provide further data on its establishment.
One study (Kremer 1991) did not provide any details on how the diagnosis was made.
Secondary outcomes
Four studies reported incidence rate (Table 2) of cellulitis during treatment ('on prophylaxis') (Chakroun 1994; Kremer 1991; Thomas 2012; Thomas 2013); two reported the incidence rate after the treatment was stopped ('post‐prophylaxis') (Thomas 2012; Thomas 2013).
Three studies reported time to next episode of cellulitis 'on prophylaxis' (Sjöblom 1993; Thomas 2012; Thomas 2013); in Sjöblom 1993 these data were extracted from a survival curve; two of these three studies reported time to next episode of cellulitis 'post‐prophylaxis' (Thomas 2012; Thomas 2013).
Three studies provided data regarding hospitalisation of participants (Kremer 1991; Thomas 2012; Thomas 2013): all three reported the number of participants hospitalised, but only two stated the length of stay in hospital (Kremer 1991; Thomas 2013). A combined analysis of Thomas 2012 and Thomas 2013 for this outcome was described in a separate publication (Mason 2014).
Quality of life was not reported separately for any of the included studies. Quality‐of‐life measures were used in a combined analysis of Thomas 2012 and Thomas 2013 (Mason 2014).
The development of resistance to antibiotics was not examined in any of the included trials.
Adverse reactions were reported in five included studies (Chakroun 1994; Kremer 1991; Sjöblom 1993; Thomas 2012; Thomas 2013), but not in Kasseroller 1998.
Excluded studies
We excluded eight studies, reporting the reasons for their exclusion in the Characteristics of excluded studies tables. Three studies were not RCTs (Duvanel 1986; Haustein 1989; Wang 1997), two studies investigated preventive treatment for other skin infections (skin abscesses in Klempner 1988 and folliculitis/furunculosis (Table 2) in Raz 1996) and two studies were in children (Ferrieri 1973; Maddox 1985). After email communication with trial investigators we confirmed that another study focused on recurrent skin abscesses in children (more than 70% of participants were children) (Fritz 2011).
Studies awaiting classification
One study, Ratnikova 1991, is awaiting classification. For details, please see the Characteristics of studies awaiting classification table.
Risk of bias in included studies
We provide summaries of the risks of bias across the included studies in Figure 2 , Figure 3, and the Characteristics of included studies tables.
2.
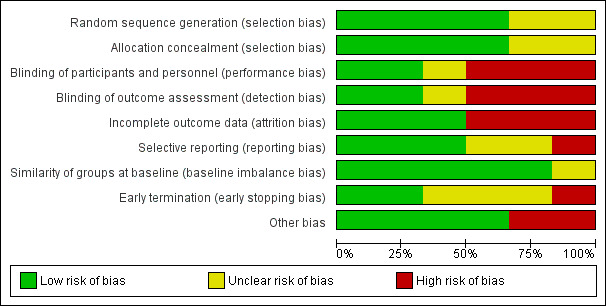
Risk of bias graph: review authors' judgements about each risk of bias item presented as percentages across all included studies.
3.
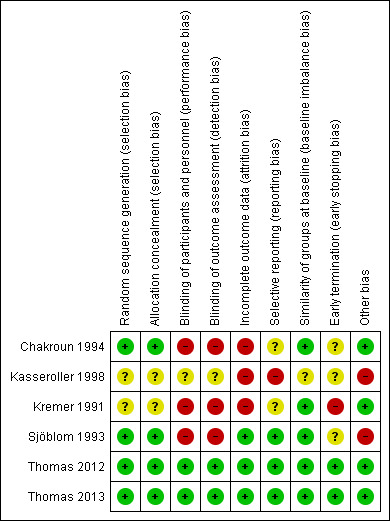
Risk of bias summary: review authors' judgements about each risk of bias item for each included study.
When the risk of bias information was not available in the study, we sought further data by correspondence with study investigators. We were answered by the investigators of five out of six studies (Chakroun 1994; Kremer 1991; Sjöblom 1993; Thomas 2012; Thomas 2013) (Table 3). For one of these studies the investigator could not provide details due to the age of the study, as the relevant data were not available (Kremer 1991). We sent emails, a letter and also attempted to contact the sole author of the Kasseroller 1998 study through his professional website but received no reply.
2. Contact with investigators of included studies.
| Study | Way of communication | Date | Information provided | Notes |
| Chakroun 1994 | 12/2013 | ‐ Allocation concealment ‐ Participants follow‐up ‐ Criteria for diagnosis ‐ Adherence ‐ Adverse reactions ‐ Informed consent ‐ Ethical committee approval ‐ Source of funding |
Full information was not available for all queries, but investigators responded to all of them | |
| Kasseroller 1998 | airmail, email, website | 2013 ‐ 2014 | ‐ | Investigator did not reply to our queries We also contacted a potential sponsor, not reported by the author, who confirmed their financial support for the conduct of this study (email correspondence with the head of medical‐scientific department of 'biosyn Arzneimittel GmbH' from January 2015) |
| Kremer 1991 | email and telephone | 12/2013 and 1/2014 | ‐ | Data were not available and the investigator did not remember any details |
| Sjöblom 1993 | 12/2013 | ‐ Allocation concealment ‐ Participants follow‐up ‐ By whom cellulitis was diagnosed ‐ Adherence ‐ Source of funding |
Full information was not available for all queries, but investigators responded to all of them | |
| Thomas 2012 | 1/2014 | ‐ Episodes of recurrent cellulitis per person‐months (incidence rate) ‐Time to next episode ‐ Adverse events by study arm ‐ Duration of hospitalisation ‐ Quality of life |
‐ | |
| Thomas 2013 | 1/2014 | ‐ Episodes of recurrent cellulitis per person‐months (incidence rate) ‐ Quality of life |
Hospitalisation and quality of life were not evaluated directly in this trial but were reported by indirect evaluation in Mason 2014 |
Allocation
Sequence generation
We judged four studies to be at low risk of bias for this domain (Chakroun 1994; Sjöblom 1993; Thomas 2012; Thomas 2013). In two of them investigators used an independent party to provide randomised codes, which were generated by computer programmes (Thomas 2012; Thomas 2013), one used stratified block randomisation (Table 2) (Sjöblom 1993) and in Chakroun 1994 the investigators drew lots to assign participants to treatment or control groups. Two studies did not state how they generated allocation sequences and we rated them at unclear risk of bias (Kasseroller 1998; Kremer 1991).
Allocation
In two studies allocation concealment was verified using a computer‐based allocation system by a central co‐ordinating team, and we judged them to be at low risk of bias (Thomas 2012; Thomas 2013). Following communication with investigators, we rated two additional studies at low risk of bias for this domain (Chakroun 1994; Sjöblom 1993). In Kasseroller 1998 and Kremer 1991 the method used to conceal the allocation sequence was not reported and we therefore judged them to be at unclear risk of bias for this domain.
Blinding
Three studies did not blind participants or personnel to the intervention being studied so we rated them at high risk of bias (Chakroun 1994; Kremer 1991; Sjöblom 1993).
We judged two studies to be at low risk for performance bias and detection bias (Thomas 2012; Thomas 2013). In these studies treatment allocation was concealed throughout the trial, with the randomisation code held by the trial centre and analysis of the results performed prior to breaking the code. In addition, participants in the control group were given placebo tablets that were of the same size and shape as the penicillin tablets. The investigators noted that the penicillin and placebo tablets were not identical, due to technical reasons, but this was balanced by the wide geographic area from which participants were recruited, thus minimising the possibility of comparison of the different kinds of tablets. The study also included an assessment of blinding effectiveness, as participants were asked to guess which treatment they had received. Thomas 2012 reported that only 13% of participants correctly guessed what treatment they were on, based on the smell, taste or look of the penicillin (or absence, for placebo). The potential for detection bias was further reduced by confirmation of cellulitis cases on the basis of medical records.
Kasseroller 1998 was reported to be a 'double‐blind study', yet did not provide information on the blinding of participants, personnel or assessors. In this study the participants in the control arm received physiological salt solution and in the treatment arm selenium dissolved in physiological salt solution. It is unclear whether there were differences between these two solutions by appearance, taste, smell or adverse reactions, and we rated the risk of bias due to blinding for this study as unclear.
Incomplete outcome data
We judged three studies to be at low risk of bias, with data presented for all participants and ITT analyses (Table 2) performed (Sjöblom 1993; Thomas 2012; Thomas 2013). Two of them provided complete flow charts of participants during the trial (CONSORT flow diagram) (Thomas 2012; Thomas 2013). There was high risk of attrition bias, associated with withdrawals or dropouts, in three studies (Chakroun 1994; Kasseroller 1998; Kremer 1991) and we therefore conducted a 'per protocol' analysis (Table 2).
Selective reporting
We judged three studies to be at low risk of bias: in Thomas 2012 and Thomas 2013 outcomes were reported as mentioned in the protocol, that had been registered and was available online, and any changes to the prespecified outcomes were explained. In Sjöblom 1993 a protocol was not available, but based on the data in the Methods section of the report, it appears to have reported all prespecified outcomes and we therefore judged it to be free of selective reporting. We judged that the Kasseroller 1998 study could have introduced an element of bias through selective outcome reporting, as it failed to include results that would have been expected to have been reported for such a study, i.e. adverse events. In addition, the investigator reported measurements of selenium blood levels but this outcome had not been prespecified in the Methods section or sufficiently explained.
Two studies did not provide enough information to determine if the prespecified outcomes had been adequately reported; we therefore judged them to be at an unclear risk of bias (Chakroun 1994; Kremer 1991).
Other potential sources of bias
Baseline imbalance
We judged the risk of bias as low in five out of six studies (Chakroun 1994; Kremer 1991; Sjöblom 1993; Thomas 2012; Thomas 2013). Kasseroller 1998 did not report on important baseline characteristics of the treatment groups (comorbidities, prior oncological treatment) and we rated it at unclear risk of bias.
Early termination
We judged three of the studies to be at unclear risk of bias (Chakroun 1994; Kasseroller 1998; Sjöblom 1993), as prespecified stopping rules or sample size calculations were not reported and the duration/termination of the trial was not explained. Two studies addressed the sample size calculation methods (Thomas 2012; Thomas 2013); in one of them recruitment was stopped before the target number was attained due to slow recruitment of participants (Thomas 2012), with reasons provided and well analysed (Thomas 2010); we therefore rated these studies at low risk of bias. Kremer 1991 reported termination of the trial based on the apparent efficacy of the intervention group, and was assessed as being at high risk of bias because its early stopping for benefit might have overestimated the treatment effect.
Other potential bias
We rated bias associated with other causes as low in four studies (Chakroun 1994; Kremer 1991; Thomas 2012; Thomas 2013), and high in two studies (Kasseroller 1998; Sjöblom 1993).
Kasseroller 1998 reported two periods of intervention for all the participants in the trial: the first three weeks of inpatient intensive therapy was followed by three months of outpatient follow‐up, both in parallel with the consumption of selenium or physiological salt solution. The article did not report the method of follow‐up after the first three weeks of inpatient care, having stated that many of the clinic's patients came from abroad. We thought that possible differences in follow‐up methods might introduce considerable bias, and deemed the available data on the method of follow‐up as insufficient. We therefore evaluated the study as being at high risk of bias.
In Sjöblom 1993 the investigators used fixed and known block sizes for randomisation which might allow prediction of treatment assignment, and so we classed this as being at high risk of bias.
Effects of interventions
See: Table 1
Please see our Glossary (Table 2) for an explanation of terms used in this section. For the exact definition of outcomes please see Types of outcome measures.
Antibiotics versus no treatment/placebo
See: Table 1 'Antibiotic prophylaxis compared to no treatment/placebo'
Primary outcome
1. Recurrence
All five studies, evaluating 513 participants, reported on recurrence of cellulitis, i.e. number of participants with at least one repeat episode of cellulitis during the study period/number of evaluable participants. Pooling of the studies showed that antibiotic prophylaxis significantly reduced the recurrence of cellulitis during treatment: risk ratio (RR) 0.31 (95% confidence interval (CI) 0.13 to 0.72; P = 0.007; moderate‐certainty evidence) with moderate heterogeneity (I² = 43%; Analysis 1.1). The number of participants needed to treat to prevent one episode of cellulitis when on treatment ('on prophylaxis') was 6 (95% CI 5 to 15) with a control event rate (CER) of 83/263 (32%).
1.1. Analysis.
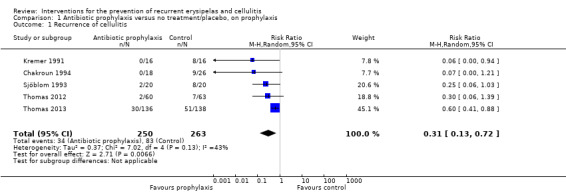
Comparison 1 Antibiotic prophylaxis versus no treatment/placebo, on prophylaxis, Outcome 1 Recurrence of cellulitis.
Only two studies (287 participants) continued to follow up participants after the cessation of treatment ('post‐prophylaxis') (Thomas 2012; Thomas 2013). Pooling the data did not show significant differences in recurrence after the treatment was stopped (RR 0.88, 95% CI 0.59 to 1.31; P = 0.52; I² = 0%; low‐certainty evidence; Analysis 2.1). We downgraded the evidence by two levels for imprecision (small sample size and low event rate, and a wide confidence interval including benefit and harm).
2.1. Analysis.
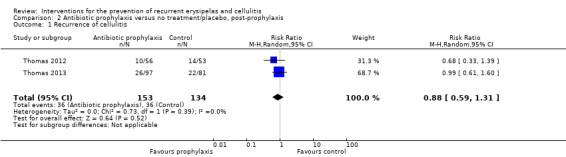
Comparison 2 Antibiotic prophylaxis versus no treatment/placebo, post‐prophylaxis, Outcome 1 Recurrence of cellulitis.
Similarly, evaluation of the overall effect of antibiotic prophylaxis ('overall trial') on cellulitis recurrence was available for Thomas 2012 and Thomas 2013, including 397 participants, with their results pooled into meta‐analysis that showed that antibiotic prophylaxis significantly reduced the recurrence of cellulitis: RR 0.75, 95% CI 0.59 to 0.95; P = 0.02; the number of participants needed to treat to prevent one episode of cellulitis was 8, 95% CI 5 to 42 (CER of 94/201 (47%)) with no heterogeneity (I² = 0%; Analysis 3.1).
3.1. Analysis.
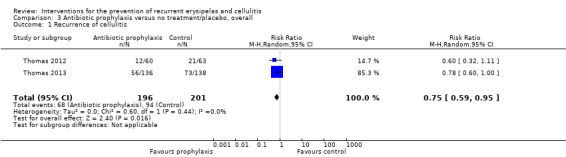
Comparison 3 Antibiotic prophylaxis versus no treatment/placebo, overall, Outcome 1 Recurrence of cellulitis.
Only one study included participants with a single episode of cellulitis at trial entry (Thomas 2012), and we were therefore unable to perform the planned analysis of participants with a single compared with at least two episodes of cellulitis at baseline. This study reported a reduction of recurrent episodes of cellulitis with six months treatment of antibiotic prophylaxis: 13/49 (27%) participants suffered a recurrent event in the control group and 8/48 (17%) participants in the treatment group. This result (RR 0.55, 95% CI 0.21 to 1.49; P = 0.24) was not statistically significant.
Secondary outcomes
1. Incidence rate
Data for this outcome were available from four studies (Chakroun 1994; Kremer 1991; Thomas 2012; Thomas 2013), including 256 episodes of cellulitis and 4375 person‐months. Antibiotic prophylaxis ('on prophylaxis') significantly reduced the incidence rate of cellulitis: RR 0.44, 95% CI 0.22 to 0.89; n = 473; P = 0.02; moderate‐certainty evidence, with moderate heterogeneity between studies (I² = 54%; Analysis 1.2). However, all studies indicated a consistent direction of benefit for use of antibiotics.
1.2. Analysis.
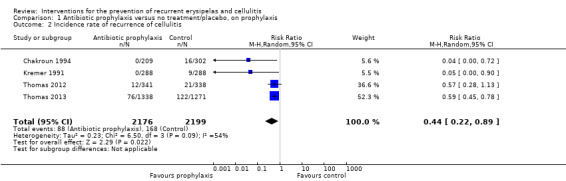
Comparison 1 Antibiotic prophylaxis versus no treatment/placebo, on prophylaxis, Outcome 2 Incidence rate of recurrence of cellulitis.
Subgroup analysis according to our predetermined subgroups (Subgroup analysis and investigation of heterogeneity) for this outcome was limited both by its irrelevance for the included studies and the small number of studies. Nevertheless, inspection of the forest plot for this analysis (Analysis 1.2) indicates a clear difference between the smaller trials (Chakroun 1994; Kremer 1991, including 44 and 40 evaluable participants, respectively) and the larger PATCH trials (Thomas 2012; Thomas 2013, including 123 and 274 participants, respectively), with the first group showing only few recurrent episodes in the control arm and no recurrence in the treatment arm. Possible reasons for this difference are the small number of participants in Chakroun 1994 and Kremer 1991, variable prophylactic regimens (including type, route and dosage of antibiotics) and methodological limitations of Chakroun 1994 and Kremer 1991 (mainly the lack of blinding and selective reporting).
As only two studies followed up participants after the treatment had stopped (Thomas 2012; Thomas 2013), a meta‐analysis of their results showed that incidence rates did not significantly change between groups after the cessation of antibiotics ('post‐prophylaxis'): RR 0.94, 95% CI 0.65 to 1.36; P = 0.74; I² = 0%; including 4566 person‐months and 109 episodes of cellulitis, low‐certainty evidence (Analysis 2.2). We downgraded the evidence by two levels for imprecision (small sample size and low event rate, and a wide confidence interval including benefit and harm). However, its overall benefit (both on and post‐prophylaxis) was still statistically significant (RR 0.69, 95% CI 0.56 to 0.85; P = 0.0005; I² = 0%; including 7854 person‐months and 340 episodes of cellulitis; Analysis 3.2), in agreement with the meta‐analyses for the primary outcome.
2.2. Analysis.
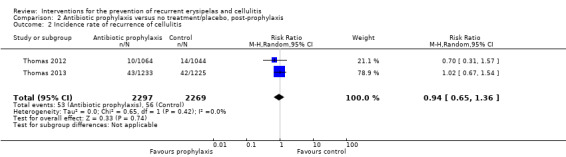
Comparison 2 Antibiotic prophylaxis versus no treatment/placebo, post‐prophylaxis, Outcome 2 Incidence rate of recurrence of cellulitis.
3.2. Analysis.
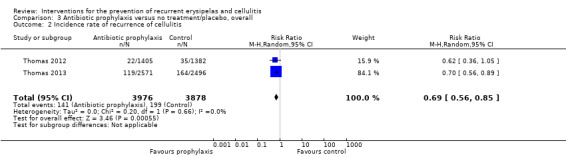
Comparison 3 Antibiotic prophylaxis versus no treatment/placebo, overall, Outcome 2 Incidence rate of recurrence of cellulitis.
2. Time to next episode
Time to next episode of cellulitis was reported in two studies (Thomas 2012; Thomas 2013) and extracted from a survival curve of another study (Sjöblom 1993). The Thomas 2013 article provided exact figures with median time to first recurrence of cellulitis of 1 year and 8.4 months in the treatment group and 1 year and 5.4 months in the control group. Overall, three studies, including 437 participants, contributed to this outcome with a 49% lower risk of an episode in the antibiotic prophylaxis group 'on prophylaxis' (pooled hazard ratio (HR) of 0.51, 95% CI 0.34 to 0.78; P = 0.002; I² = 0%; moderate‐certainty evidence; Analysis 1.3). Similar to other outcomes, the beneficial effects of antibiotics ceased after the treatment stopped ('post‐prophylaxis'), with a pooled HR of 0.78, 95% CI 0.39 to 1.56; P = 0.48; I² = 57%; low‐certainty evidence (Analysis 2.3), from two studies (Thomas 2012; Thomas 2013), including 287 participants. We downgraded the evidence by two levels for imprecision (small sample size and low event rate, and a wide confidence interval including benefit and harm). As with other outcomes, subgroup analysis was limited by the small number of trials included in the analysis.
1.3. Analysis.
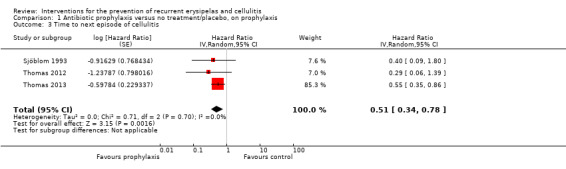
Comparison 1 Antibiotic prophylaxis versus no treatment/placebo, on prophylaxis, Outcome 3 Time to next episode of cellulitis.
2.3. Analysis.
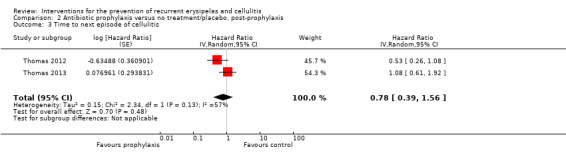
Comparison 2 Antibiotic prophylaxis versus no treatment/placebo, post‐prophylaxis, Outcome 3 Time to next episode of cellulitis.
3. Hospitalisation
Three studies (429 participants) reported on the number of participants who required hospital admission (Kremer 1991; Thomas 2012; Thomas 2013). Overall, those on antibiotic prophylaxis were less likely to be hospitalised, but this was not statistically significant: RR 0.77, 95% CI 0.37 to 1.57; P = 0.47; I² = 0%; low‐certainty evidence (Analysis 1.4).
1.4. Analysis.
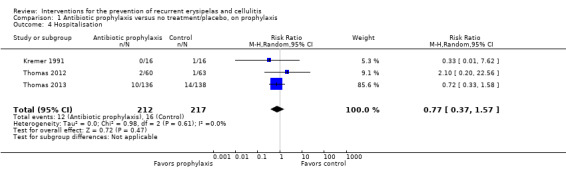
Comparison 1 Antibiotic prophylaxis versus no treatment/placebo, on prophylaxis, Outcome 4 Hospitalisation.
Only one study reported on the length of hospitalisation (Thomas 2013), finding no difference in duration of stay, with an overall mean of 10 days in the prophylaxis group (standard error of ± 7.1 days) compared with 9.2 days (standard error of ± 6.7 days) in the placebo group. A combined analysis of Thomas 2012 and Thomas 2013 did not identify a significant reduction in the number of hospital admissions or their duration (Mason 2014).
4. Quality of life
None of the six included studies reported directly on quality of life (QoL).
Mason 2014, which presented combined analysis of data from Thomas 2012 and Thomas 2013, indirectly assessed the influence of cellulitis on QoL using the European Quality of Life 5‐Dimensions questionnaire and the Dermatitis Quality of Life Index (EQ‐5D and DQLI, respectively).
The EQ‐D5 is a five‐dimension questionnaire used to measure health‐related QoL, consisting of two parts: self‐assessment questionnaire in five dimensions, i.e. mobility, self‐care, usual activities, pain/discomfort and anxiety/depression, and a self‐rated health status using a visual analogue scale. The DQLI is a 10‐item questionnaire used to measure QoL in people with a skin condition. The questions concern the influence of the disease on different aspects of life, including symptoms, self‐perception, social, interpersonal and daily activities. Each of the 10 questions is scored with a maximum of four points, which are then summed to a total score (0 to 30); higher scores mean greater impairment of quality of life (Finlay 1994).
In Mason 2014 the EQ‐D5 and the DQLI were used by participants during and 10 days after an episode of cellulitis, but before the start of trial prophylaxis, and were later used to approximate the detrimental effect of a recurrence of cellulitis on QoL. On average, an episode of cellulitis caused 26.3% reduction in QoL as measured by EQ‐5D (95% CI 18% to 35%, P < 0.001) and a deterioration of nearly 10 points as measured by DQLI (95% CI 7.83 to 11.5, P < 0.001). The investigators noted that they attempted to collect QoL data during the trials (Thomas 2012; Thomas 2013), but this was unsuccessful due to a time lag in being notified of the recurrent event.
5. Development of antibiotic resistance
We planned to evaluate the development of bacterial resistance according to the results of microbial cultures and antibiotic sensitivity testing in studies that used antibiotic prophylaxis. However, no trial properly examined the development of antibiotic resistance. Bacteriological surveillance cultures were performed in one study (Sjöblom 1993), but they were not taken from all the participants and no details of bacterial resistance were reported.
6. Adverse reactions
Four trials reported on the occurrence of any adverse reactions (Kremer 1991; Sjöblom 1993; Thomas 2012; Thomas 2013). These studies included 469 participants. There was no significant difference in adverse events between the two groups, with an overall pooled estimate of RR 0.87 (95% CI 0.58 to 1.30; P = 0.48; I² = 19%; low‐certainty evidence; Analysis 1.5). Synthesising the studies for adverse reactions did not show considerable heterogeneity, but we considered possible differences in adverse drug reaction profiles between studies, influenced by factors such as the type of antibiotic, its dosage and unblinding of trial participants to their treatment.
1.5. Analysis.
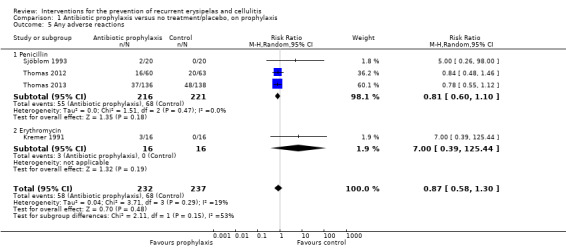
Comparison 1 Antibiotic prophylaxis versus no treatment/placebo, on prophylaxis, Outcome 5 Any adverse reactions.
Assessing the clinical applicability of our findings, we performed a post hoc subgroup analysis on the type of antibiotic, including three studies that used oral penicillin (Sjöblom 1993; Thomas 2012; Thomas 2013), which demonstrated similar results, albeit with higher precision and consistency (RR 0.81, 95% CI 0.60 to 1.10; n = 437; P = 0.18; I² = 0%; low‐certainty evidence; Analysis 1.5).The P value for the test for subgroup differences was 0.15 when comparing penicillin versus erythromycin.
The common adverse reactions were: gastrointestinal symptoms, mainly nausea and diarrhoea, rash (severe cutaneous adverse reactions were not reported) and thrush. The authors of Thomas 2013 noted that no clostridium difficile colitis infections were reported, but none of the four studies actively tested participants with gastrointestinal symptoms for clostridium difficile.
Adverse effects that required discontinuation of the assigned therapy were reported in three studies (Kremer 1991; Sjöblom 1993; Thomas 2012): in Kremer 1991 three participants developed abdominal pain and nausea while treated with erythromycin and their treatment was changed to penicillin; in Sjöblom 1993 two participants stopped treatment with penicillin because of diarrhoea or nausea; and in Thomas 2012 five participants stopped treatment (three in the placebo group and two in the penicillin group) because of adverse reactions that were not further described. Another study did not report on adverse reactions, but the investigator noted in an email correspondence that approximately 10% of participants stopped treatment due to pain at the injection site (the active treatment group was given intramuscular injections of benzathine penicillin) (Chakroun 1994).
7. Mortality
Three studies including 437 participants reported on mortality (Sjöblom 1993; Thomas 2012; Thomas 2013). Overall 20 deaths occurred: nine in the control group and 11 in the penicillin group, none of which was related to treatment, and when data were pooled there was no statistically significant difference in mortality between the two groups (RR 1.12, 95% CI 0.32 to 3.91; P = 0.86; I² = 37%; Analysis 1.6).
1.6. Analysis.
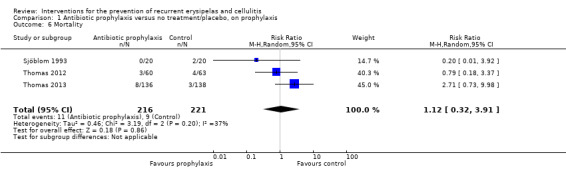
Comparison 1 Antibiotic prophylaxis versus no treatment/placebo, on prophylaxis, Outcome 6 Mortality.
Selenium versus physiological salt solution
Primary outcome
1. Recurrence
Only one study was included in this comparison (Kasseroller 1998). The study author reported that in the selenium group, 0 out of 29 participants had recurrent episodes of cellulitis. In the group of participants who received physiological salt solution, 15 out of 28 participants suffered recurrence (54%): one participant in the early inpatient‐care phase of the trial, followed by 14 participants in the ambulatory‐care phase.
Secondary outcomes
This study did not measure or report any of the relevant secondary outcomes: incidence rate, time to next episode, hospitalisation, quality of life, adverse reactions or mortality.
Sensitivity analysis
We had planned to explore heterogeneity through a sensitivity analysis; however, paucity of data for the relevant interventions made sensitivity analysis impractical.
Discussion
Summary of main results
The objective of this review was to summarise all available interventions for the prevention of cellulitis or erysipelas. A comprehensive search yielded six studies (573 evaluable participants), of which five assessed antibiotics versus no treatment (three studies) or placebo (two studies) in people with leg cellulitis, and one study evaluated prophylactic treatment with selenium solution against physiological salt solution in women after mastectomy.
A meta‐analysis of results from five studies, based on four studies evaluating penicillin and one evaluating erythromycin, demonstrated clear effectiveness of antibiotics compared to no treatment or placebo for the prevention of recurrent leg cellulitis, with a 69% reduction in the number of participants who were under prophylactic treatment (moderate‐certainty evidence; Table 1). However, when treatment stopped ('post‐prophylaxis'), the difference was no longer statistically significant (low‐certainty evidence).
Antibiotics were also shown to reduce the incidence rate of leg cellulitis by 56% when compared with no treatment or placebo (256 episodes of leg cellulitis over 4375 person‐months, n = 473, four studies; moderate‐quality evidence; Table 1) and significantly decreased the rate until the next episode under prophylactic treatment (n = 437, three studies, moderate‐certainty evidence; Table 1). Nevertheless, the protective effect of antibiotics seems to wane after the treatment stops ('post‐prophylaxis') for these two outcomes (low‐certainty evidence).
There was insufficient information to determine the role of antibiotic prophylaxis after a single episode of leg cellulitis, which was not statistically significant (one study). The effects are relevant mainly for people after at least two episodes of leg cellulitis occurring within a period up to three years.
With regard to other secondary outcomes, a meta‐analysis of results from three studies showed there was no difference in hospitalisation between participants on antibiotic prophylaxis and those given no treatment or placebo (three studies, n = 429, low‐certainty evidence; Table 1), but the current data did not allow us to explore its impact on length of hospital stay.
None of the studies reported severe side effects, with the main reactions being gastrointestinal symptoms, mainly nausea and diarrhoea; rash (no severe cutaneous adverse reactions were reported); and thrush. Participants discontinued treatment in three studies due to adverse events. Due to abdominal pain and nausea, three participants stopped treatment with erythromycin and instead took penicillin. A further two participants treated with penicillin stopped treatment due to diarrhoea or nausea. In one study, around 10% of participants stopped treatment because of pain at the injection site (the active treatment group was given intramuscular injections of benzathine penicillin).
Meta‐analyses looking at adverse reactions did not find a difference in tolerability or safety between antibiotic and no treatment or placebo (n = 469, four studies; low‐certainty evidence; Table 1).
None of the included studies reported quality of life or development of resistance to antibiotics.
Overall completeness and applicability of evidence
Included studies recruited participants that are representative of the target population in western countries, mostly people over the age of 50, who are overweight and suffer local predisposing conditions for recurrent cellulitis (e.g. leg oedema, fungal foot infection, etc). Although the interventions seem to be suitable for other health systems, the characteristics of people who suffer recurrent cellulitis (excluding filariasis) might be different in other parts of the world, such as in Africa, Asia and South America. In the these areas, HIV carrier rates, nutrition status, sanitation and hygiene conditions, possibly different comorbidity profiles of the population (with regard to weight, diabetes, vascular disease) and access to quality medical services may hamper generalisation of our conclusions. The six studies referred to all relate to the use of antibiotics or selenium; although we had planned to include non‐drug preventive treatments, we found no studies that sought to measure the effectiveness of these interventions.
Ascertaining the diagnosis of cellulitis is essential, as incorrect diagnoses might both introduce bias into the studies and limit the applicability of the evidence when studies are combined in a meta‐analysis.The importance of an accurate diagnosis of cellulitis is highlighted by recent publications, suggesting that it is commonly misdiagnosed both inside and outside the hospital (Arakaki 2014; Levell 2011; Strazzula 2015). Despite caveats, the vast majority of participants were diagnosed by specialists, either in dermatology or infectious disease clinics; the two larger studies in the review (Thomas 2012; Thomas 2013), including 77% of all evaluable participants, used consistent and rigorous criteria to diagnose cellulitis, enabling the extrapolation of data to the general population affected.
The period of cellulitis recurrence prior to trial entry varied among studies and ranged from one to three years, with most of the participants suffering at least two prior episodes of cellulitis. Thus, although pertinent to many cases, it is possible that the beneficial impact of antibiotic prophylaxis or its magnitude on the recurrence of cellulitis is not readily applicable for people at the extremes, either after a single or multiple episodes of cellulitis. Furthermore, most of the evidence relates to people with cellulitis of the leg.
It was disappointing to note that only two studies included in this review followed up participants after treatment was stopped. The follow‐up period lasted up to two years, during which the effectiveness of prophylaxis diminished. The duration of prophylactic therapy was also limited, with the longest mean duration of treatment up to 18 months. Considering the long‐lasting nature of local and systemic risk factors for recurrent cellulitis and increasing life expectancy, the duration of preventive treatment and its enduring effect, during and possibly after treatment, ought to be more adequately addressed.
We noted a clinically important heterogeneity in the type of antibiotic, dosage and route of administration. The PATCH trial team introduced low‐dose oral penicillin as preventive therapy, gathering an unprecedented number of almost 400 participants with leg cellulitis into carefully‐designed and scrupulously‐analysed RCTs. Higher doses of oral penicillin (2 grams to 4 grams a day) (Sjöblom 1993) or penicillin injections (Chakroun 1994) were used in two other studies, but with small numbers of participants and less stringent study designs limiting high‐quality evidence‐based selection of an alternative prophylaxis. Only one study (Kremer 1991), enrolling 32 participants, explored an alternative to penicillin by using erythromycin; however, it did not allow us to determine the role of antimicrobial prophylaxis for people with penicillin allergy.
Intramuscular injection of penicillin is commonly used in current practice, but this intervention was only assessed in a single small study. Moreover, a recent report of three deaths following benzathine penicillin injections (Israeli Ministry of Health 2015) together with previous cases of death associated with its intramuscular delivery (Arumugam 2012; WHO 2000) raise concerns about the safety of this practice. However, RCTs are not the optimal platform to assess rare adverse events.
With regard to secondary outcomes, a meta‐analysis of results from three studies showed there was no difference in hospitalisation between participants on antibiotic prophylaxis and those given no treatment or placebo (three studies, n = 429, P value = 0.47, low‐certainty evidence; Table 1), but the existing data did not allow us to explore its impact on length of hospital stay. A report by the PATCH trial team (Mason 2014) for their studies does not corroborate this analysis, and in this publication the preventive effect of antibiotic did not translate into a reduction in hospitalisations or the length of stay. The available studies did not permit assessment of the development of QoL or antimicrobial resistance.
Overall and without change with time, cellulitis is predominantly caused by group A streptococci and other β‐haemolytic streptococci (Jeng 2010). Staphylococcus aureus less frequently causes cellulitis, and is often associated with penetrating trauma and open wounds (Mandell 2010; Stevens 2014). Methicillin‐resistant Staphylococcus aureus (MRSA) is an unusual cause of typical cellulitis, and antibiotic coverage for this organism is usually unnecessary (Stevens 2014). The variable incidence of community‐acquired MRSA infections should therefore not limit the generalisability of our findings across countries with different incidence of MRSA‐associated skin infections. However, it would be prudent for future studies to examine the effectiveness of penicillin prophylaxis in geographical and other settings with a significant burden of MRSA infections.
Despite decades of use group A streptococci remained susceptible to penicillin (Gerber 1995; Horn 1998), making it an ideal antibiotic prophylaxis for cellulitis. Nonetheless, other bacteria are also known to be implicated in cellulitis (Chaniotakis 2016; Chira 2010; Garau 2015), raising a concern about the development of antimicrobial resistance. In addition to the possible development of resistant strains of bacteria in treated individuals, the introduction of antibiotic preventive therapy to a large population might further facilitate the evolution of antibiotic resistance and its relevance for public health must be addressed (Kunkel 2015). The high‐quality PATCH trials' preventive regimen (Thomas 2012; Thomas 2013) included low‐dose penicillin for a long treatment duration, which had been shown to induce the emergence of strains of drug‐resistant Streptococcus pneumoniae in another study (Guillemot 1998). However, the effect of any prophylactic regimen (with respect to dosage, duration or continuity) can not be easily predicted and should be based on future evidence (Kouyos 2014; Read 2011).
A number of attempts were made to develop a streptococcal vaccine but these were halted after serious safety concerns emerged (Massell 1969). Preliminary reports of later published trials indicating safe and effective results with newer streptococcal vaccines have revitalised efforts to develop a vaccine against group A streptococci (Kotloff 2004; McNeil 2005; Moreland 2014). We identified a single study assessing the use of streptococcal vaccine for cellulitis prevention (Haustein 1989) with a marked reduction in recurrence rate, but this was not a randomised controlled trial; larger well‐designed studies are necessary to establish the safety and effectiveness of a vaccine strategy.
Successful adherence to prophylactic treatment is a key element of any prevention strategy. The PATCH trial team reported good compliance, with more than 75% of participants adhering to treatment with penicillin tablets (see Included studies). Chakroun 1994 used penicillin injections into the muscle and mentioned a 10% dropout rate due to pain at the injection site. The benefits of antibiotic prophylaxis reported in these RCTs will only be replicated if people with cellulitis adhere to the prophylactic treatment. Possible barriers to adherence, except for adverse reactions, are the increasing age of those with recurrent cellulitis and the long‐term need for prophylaxis, as adherence would probably decrease with time. This emphasises the need to identify prophylactic regimens that are more convenient to both the person with recurrent cellulitis and the physician. These could include exploring other determinants of adherence, such as simplifying medication regimens, tailoring, and later monitoring the choice of prophylaxis to individuals, creating education and support programmes, and involving people with cellulitis and their families in clinical decision‐making as well as in research plans.
This review clearly demonstrates that current evidence falls far short of establishing the benefit of universally‐accepted treatments that mainly target local risk factors for recurrent cellulitis, including various methods to reduce lower or upper limb lymphoedema and oedema (Arsenault 2011; Brorson 2000; Campisi 2015; Damstra 2009; Didem 2005; Granzow 2014; Ko 1998; Szolnoky 2014; Yamamoto 2015), venous insufficiency and good local skin care, mainly antifungal treatment for tinea pedis (athlete's foot), but also proper local hygiene and maintenance of skin integrity.
Recurrent cellulitis of the upper limb was investigated by only one study evaluating preventive treatment with selenium, but data scarcity hinders its external validity.
Quality of the evidence
The five studies included in the quantitative analysis of the review can be classified into two groups of methodological quality: Thomas 2012 and Thomas 2013, conducted by the PATCH trial team, represent very high‐quality research characterised by a well‐drafted study protocol, meticulous study design with the use of placebo, and a complete report of predefined outcomes. The PATCH trial's data integrity proved to be solid, critically appraised by experts in several publications (Arasaratnam 2013; Durand 2013; Inghammar 2014; Oh 2014; Van Zuuren 2014) and with remarkable transparency and sharing of research data by the investigators. We judged these trials accordingly to be free of bias for all domains.
The other group of studies (Chakroun 1994; Kremer 1991; Sjöblom 1993) include small numbers of participants (range of 32 to 44 participants compared with 123 and 274 participants in the PATCH trials) with less stringent study design and reporting. They primarily lack blinding which might introduce bias, but they also raise concerns of incomplete reporting as detailed in the Risk of bias in included studies section and presented in Figure 3.
We graded the certainty of evidence for our primary outcome, the recurrence of leg cellulitis under preventive treatment, as moderate, reflecting the consistently clear beneficial effect of antibiotic prophylaxis on leg cellulitis recurrence across studies, and in particular the significant magnitude and large weight contributed by Thomas 2013 to the meta‐analysis. Similar reasons led to a moderate grading of other important comparisons: the incidence rate of cellulitis, and the time to next episode of cellulitis under treatment. However, we downgraded these outcomes due to imprecision because of a relatively low number of participants (small sample size).
Low certainty of evidence was attributed to the effect of antibiotic prophylaxis on hospital stay, adverse reactions and recurrence of cellulitis following the cessation of treatment, downgraded by imprecision of results, as indicated by the low number of participants and events (hospitalisations, adverse events and participants with recurrent episodes of cellulitis, respectively) and the 95% confidence interval overlapping the line of no effect (hospitalisations).
Three outcomes were assessed at 'post‐prophylaxis' (recurrence, incidence rate of recurrence, and time to next episode of cellulitis), and we rated them as low‐certainty evidence. We downgraded them two levels, due to serious imprecision from a small sample size/low event rate, wide confidence intervals and the confidence interval including both benefit and harm.
Potential biases in the review process
We conducted a highly inclusive search for published studies, and we further sought unpublished trials and abstracts submitted to major dermatological conferences and circulated in the informal grey literature. We scanned reference lists of all included studies, reviews and most of the other studies cited in the reference list of this review. We scrutinised many diverse guideline papers regarding recurrent cellulitis, from different countries and medical disciplines and published in different languages. We did not, however, search foreign‐language databases other than the Latin American and Caribbean Health Sciences Literature database (LILACS) (Appendix 4), so we cannot discount the possibility that relevant publications are to be found in the Chinese, Japanese or other non‐western medical literature. We planned in the protocol to search BIOSIS Previews from 1969 but only searched from 1990 in the review due to technical reasons and database availability.
We looked for all types of interventions, including local therapy, lifestyle modifications, surgery and systemic treatments. We placed no language restrictions and subsequently screened German‐published papers, a French‐published article, and we anticipate the evaluation of a Russian‐published study in a future update of this review. We included unpublished data for bias evaluation and further incorporation into meta‐analyses obtained from researchers (see Table 3).
We were not able to properly assess a single study that investigated the effectiveness of bemitil for cellulitis prophylaxis, for which the full text was not retrievable (Ratnikova 1991). This study is listed under studies awaiting classification. Despite incompleteness, we would not expect the missing information from this trial to influence the review's main findings on the value of antibiotic prophylaxis.
We cannot entirely rule out the risk of publication bias, especially when all studies exploring antibiotic prophylaxis point towards a positive effect of the intervention, but the small number of studies prevented us from formally assessing its probability. Nevertheless, the results of the recently‐published high‐quality PATCH trials (Thomas 2012; Thomas 2013), joining the other non‐industry‐funded RCTs in this review, and the fact that commercial investment in low‐cost preventive strategy with antibiotic are not likely to be profitable, minimise this risk.
Data from this review were insufficient to perform the subgroup analyses we had planned; we were therefore unable to investigate heterogeneity, due to the small number of included studies.
Agreements and disagreements with other studies or reviews
Two earlier systematic reviews evaluated the evidence for the prevention of recurrent cellulitis: Morris 2008 and Oh 2014.
Morris 2008 identified two RCTs (Kremer 1991; Sjöblom 1993), also included in our review. Examining these studies, the author of the review concluded that only limited evidence would support the use of prophylactic antibiotics for reduction of future attacks of cellulitis, and rated the certainty of evidence for the intervention as low, based on the GRADE classification. Our search retrieved four additional studies; two preceding the Morris 2008 review (Chakroun 1994; Kasseroller 1998), which were later followed by the two PATCH trials (Thomas 2012; Thomas 2013), which provide higher‐certainty evidence to further support the conclusion that prophylactic antibiotics reduce the incidence of future attacks of cellulitis.
Oh 2014 reviewed antibiotic prophylaxis for the prevention of recurrent cellulitis and found similar results to this review.
A number of guidance documents referring to the prevention of recurrent cellulitis have been issued up to this time and retrieved by our systematic search. These clinical practice guidelines and their major recommendations on antibiotic prophylaxis, its duration and adjunctive therapy are summarised in Table 4. Overall, most guidelines advocate antibiotic prophylaxis with penicillin, but differ widely on the best time to start treatment and its duration. There are also inconsistencies in alternatives to penicillin, as well as the means of delivery and dosage. Of note, only three groups, the Clinical Resources Efficiency Support Team, the National Institute for Health and Clinical Excellence and the Infectious Diseases Society of America, refer to the strength of their recommendations or the quality of evidence for the use prophylactic antibiotics (CREST 2005; NICE 2005; Stevens 2005; Stevens 2014).
3. Clinical guidelines on antibiotic prophylaxis for the prevention of recurrent cellulitis.
| Guideline | Organisation | Recommended antibiotic | Duration of Px |
No of episodes to initiate Px |
Adjunctive Tx | Quality of evidence † |
| BLS 2016 | BLS | penicillin by mouth; alternatives: cephalexin, erythromycin, clarithromycin, clindamycin, doxycycline |
2 y; life‐long Px if recurrence after Px stopped |
2 ≥/y | skin care, decongestive Tx, antifungal Tx, alcohol wipes; SIT |
NS |
| ALA 2015 | ALA | penicillin by mouth; alternatives: cephalexin, erythromycin, clindamycin |
2 y; life‐long Px if recurrence after Px stopped |
2 ≥/y | skin care, decongestive Tx, bacterial decolonisation Tx; SIT | NS |
| Stevens 2014 | IDSA | penicillin by mouth/IM; alternatives:erythromycin |
as long as RF persist |
3 – 4 /y | skin care, Tx of oedema, weight reduction | antibiotic Px ‐weak, moderate ‡ duration of Px ‐ strong, moderate ‡ skin care ‐ strong, moderate ‡ |
| Stevens 2005 | penicillin by mouth/IM; alternatives:erythromycin |
NS | frequent | skin care, Tx of oedema compression stockings, diuretics; SIT |
grade IIB § | |
| ISL 2013 | ISL | penicillin; alternatives: broad spectrum antibiotic |
NS | repeat despite physical Tx |
skin care, antifungal Tx | NS |
| Esposito 2011 | SIMIT and ISC | penicillin or macrolide | NS | recurrent | skin hygiene and compression stockings |
NS |
| Draijer 2008 | NHG | penicillin by mouth/IM | 1 ‐ 2 y | 2 ≥/y | skin care, compression stockings; SIT |
NS |
| ILF 2006 | ILF | penicillin by mouth; alternatives:erythromycin, clindamycin, clarithromycin |
2 y; life‐long Px if recurrence after Px stopped |
2 ≥/y | skin care, decongestive Tx, antifungal Tx; SIT |
NS |
| CREST 2005 | CREST | penicillin or erythromycin by mouth | 2 y | 2 ≥/y | SIT may be preferable | weak and inconclusive |
| NICE 2005 | NICE | a trial should be considered | NS | > 2/y | skin care, Tx of oedema, compression stockings, weight reduction |
weak and inconclusive |
| Eron 2003 | Other* | antibiotic may be needed; type of antibiotic NS |
long term | NS | skin care, Tx of oedema, antifungal Tx; SIT |
NS |
| SFD 2000 | SFD | penicillin by mouth/IM; alternatives: macrolide |
prolonged, probably indefinitely |
several/poorly controlled RF |
skin care, Tx of oedema | NS |
| Duodecim 1999 | FMSD | antibiotic should be considered; type of antibiotic NS |
long term | frequent | skin care | NS |
† Assessement of quality of evidence as defined and graded by the authors of the document.
‡ Strong recommendation, moderate quality ‐ desirable effects clearly outweigh undesirable effects; evidence from RCTs with important limitations or exceptionally strong evidence from unbiased observational studies; recommendation can apply to most patients in most circumstances and further research is likely to have an important impact on confidence in the estimate of effect and may change the estimate.
Weak recommendation, moderate quality ‐ desirable effects closely balanced with undesirable effects; evidence from RCTs with important limitations;recommendation may change when higher‐quality evidence becomes available; and further research is likely to have an important impact on confidence in the estimate of effect and may change the estimate.
§ Moderate evidence ‐ should generally be offered; II ‐ evidence from one well‐designed clinical trial.
Abbreviations
IM ‐ injections into the muscle (intramuscular) No ‐ number NS ‐ not specified Px ‐ preventive treatment (prophylaxis) RF‐ risk factors SIT ‐ self‐initiated treatment Tx ‐ treatment y ‐ year/s
Medical organisations
BLS ‐ British Lymphology Society ALA ‐ Australasian Lymphology Association IDSA ‐ Infectious Diseases Society of America ISL ‐ International Society of Lymphology SIMIT ‐ Società Italiana di Malattie Infettive e Tropicali ‐ Italian Society of Infectious Diseases ISC ‐ International Society of Chemotherapy NHG ‐ Nederlands Huisartsen Genootschap ‐ The Dutch College of General Practitioners ILF ‐ International Lymphoedema Framework CREST ‐ Clinical Resources Efficiency Support Team (UK) NICE ‐ National Institute for Health and Clinical Excellence (England) SFD ‐ La Société Française de Dermatologie ‐ The French Society of Dermatology MSD ‐ The Finnish Medical Society Duodecim
* 5 of 6 experts in this consensus paper were from North America; published in the Journal of the British Society for Antimicrobial Chemotherapy
Findings from this systematic review are in agreement with most of the aforementioned guidelines supporting the use of antibiotic prophylaxis (penicillin in particular) for people with recurrent lower limb cellulitis.
Authors of many of the major guidelines point out that prolonged or even life‐long antibiotic prophylaxis is warranted, in accordance with our meta‐analyses, showing that the preventive effects of antibiotics gradually wane after prophylaxis is stopped, corresponding with a similar observation reported as early as 1985 (Duvanel 1985). However, the evidence for this is lacking.
Authors' conclusions
Implications for practice.
Our review found moderate‐certainty evidence that when compared with no treatment or placebo antibiotic prophylaxis is probably effective for the prevention of recurrent cellulitis of the lower limbs in terms of reducing the risk of recurrent episodes of cellulitis and the number of episodes and prolonging the time to recurrence, based mainly on penicillin prophylaxis treatment. The preventative effect of antibiotic is supported both by its magnitude and by consistent improvement of the aforementioned patient‐outcomes.
Low‐certainty evidence found that antibiotic prophylaxis, based on four studies administering penicillin and one study administering oral erythromycin, may make little or no difference in terms of adverse effects or hospitalisation when compared with no treatment or placebo. The existing data did not allow us to explore its impact on length of hospital stay.
The characteristics of participants enrolled in the included studies, along with the magnitude of the preventive effect, favour its use in people with at least two past episodes of lower‐limb cellulitis within a time frame of up to three years. However, once discontinued, there was low‐certainty evidence to show that the protective effect of antibiotics does not seem to last.
The one study (Ratnikova 1991) in Studies awaiting classification may alter the conclusions of the review once assessed.
Implications for research.
We found no high‐quality research on interventions for the prevention of recurrent cellulitis, and much research remains to be undertaken. Cellulitis research is under‐represented in publicly‐funded medical research compared with the global burden it inflicts (Karimkhani 2014a; Karimkhani 2014b).
Of the five studies included in the quantitative analysis of this review, only two were well designed and rigorously conducted and reported. Further high‐quality sufficiently‐powered RCTs are thus needed measuring patient‐relevant outcomes such as hospitalisation and length of stay in hospital, and quality of life. To reduce bias, studies should ensure participants and outcome assessors are blinded to treatment allocation, and measures should be taken to prevent dropouts and losses to follow‐up.
We believe that the following clinical questions need to be answered:
What antibiotic should be used for prophylaxis?
RCTs published to date evaluated penicillin or erythromycin. There is no evidence for other antibiotics. The development of new antibiotics might inspire ideas for more trials, focusing on novel agents, such as very long‐acting antibiotics (Corey 2014) and their use when prophylaxis fails; however, induction of resistance has to be a main consideration in antibiotic studies.
When should antibiotic prophylaxis be started and for how long should it be maintained?
Trials should be designed to determine after how many episodes and for what time period between recurrences therapy should be initiated. Investigators must consider extending the follow‐up periods, during and after cessation of prophylaxis, to provide substantial evidence for its optimal duration and long‐lasting effects and safety. Data from several studies demonstrate early and irreversible lymphatic damage following cellulitis (Damstra 2008; De Godoy 2000; Soo 2008) that together with indications from Thomas 2012 might usher in a paradigm shift in the prevention of recurrent cellulitis, requiring more studies that focus on the early introduction of prophylaxis.
How should antibiotics be given?
Current evidence supports the use of low‐dose oral penicillin, but some evidence for weight‐dependent dosing of oral penicillin or its intramuscular delivery also exists (Chakroun 1994; Chen 2015; Wang 1997); this demonstrates the need for RCTs comparing different schedule and dosing regimens, preferably head‐to‐head trials. A concept of cyclic antibiotic prophylaxis was favoured by German investigators who used cycles of intravenous penicillin together with lymphatic decongestion (Allard 1999), but the value of this preventive approach should first be validated in a large RCT.
Who will benefit more from antibiotic prophylaxis?
Perhaps the most practical clinical approach for the prevention of cellulitis would be to identify and treat people with a high risk for recurrent cellulitis, initiating prophylaxis on an outpatient basis . A risk stratification approach should define and validate risk factors for recurrence (such as limb oedema, venous insufficiency, fungal foot infection, obesity and previous episodes of cellulitis) and create simple tools to enable its translation into clinical practice, such as risk‐scoring systems. A recently published work by Karppelin 2014 tried to identify clinical and laboratory predictors of recurrent cellulitis. Work by Tay 2015 sought to develop a clinical score to help guide physicians on when to intervene. These types of studies may pave the way for more targeted research efforts to define and improve methods for the prevention of cellulitis.
What are the alternatives?
In contrast to the key role of local factors in the pathogenesis of recurrent cellulitis and hence prevention, there was a striking absence of high‐quality evidence to support the prophylactic use of local skin care, antifungal treatment and lymphoedema reduction methods. We found an early observational study that reported an effective reduction of episodes of cellulitis with antifungal treatment (Young 1960) and two RCTs targeting recurrent cellulitis in people with filarial lymphoedema also reporting a clear benefit with the use of soap and proper foot care (Addis 2011; Shenoy 1999). These data indicate that further high‐quality research of these interventions for the prevention of recurrent cellulitis might be worth pursuing. Determining the contribution of these measures, both as combination therapy (with antibiotics) or single therapy (compared with antibiotics or placebo), should be of importance to physicians and patients alike and form a part of any cellulitis prevention strategy, in practice and research.
We welcome more carefully‐designed studies on the effectiveness of selenium, and on the specific value of the myriad of modalities for cellulitis prevention among women after breast cancer surgery.
Acknowledgements
We are grateful to the Cochrane Skin Group editorial team for their assistance and guidance.
We wish to thank the following investigators of the included studies, who answered our queries and provided additional details: Mohamed Chakroun and Christina Jorup‐Rönström. We particularly thank Kim Thomas, together with Angela Crook, James Mason and their team for providing us access to the primary data of the PATCH trials and kindly replying to our requests.
The Cochrane Skin editorial base wishes to thank Sam Gibbs, Cochrane Dermatology Editor for this review; Ben Carter, Statistical Editor; Esther van Zuuren, Methods Editor; the clinical referees, Laurence Le Cleach and Oh Choon Chiat; and the consumer referee, Peter Smart; as well as Kate Cahill, who copy edited the review.
Appendices
Appendix 1. CENTRAL (Cochrane Library) search strategy
#1 MeSH descriptor: [Cellulitis] explode all trees #2 MeSH descriptor: [Erysipelas] explode all trees #3 MeSH descriptor: [Soft Tissue Infections] explode all trees #4 MeSH descriptor: [Impetigo] explode all trees #5 MeSH descriptor: [Staphylococcus] explode all trees #6 MeSH descriptor: [Streptococcus] explode all trees #7 MeSH descriptor: [Skin] explode all trees #8 (cellulitis or erysipelas or impetigo or "soft tissue infection" or "soft tissue infections"):ti,ab,kw #9 staphylococc* or streptococc*:ti,ab,kw AND skin:ti,ab,kw #10 #5 or #6 #11 #7 and #10 #12 #1 or #2 or #3 or #4 or #8 or #9 or #11
Appendix 2. MEDLINE (Ovid) search strategy
1. randomized controlled trial.pt. 2. controlled clinical trial.pt. 3. randomized.ab. 4. placebo.ab. 5. clinical trials as topic.sh. 6. randomly.ab. 7. trial.ti. 8. 1 or 2 or 3 or 4 or 5 or 6 or 7 9. (animals not (humans and animals)).sh. 10. 8 not 9 11. exp Erysipelas/ or erysipelas.ti,ab. 12. exp Cellulitis/ or cellulitis.ti,ab. 13. impetigo.ti,ab. or exp *Impetigo/ 14. exp *Soft Tissue Infections/ 15. Ignis sacer.ti,ab. 16. holy fire.ti,ab. 17. st anthony's fire.ti,ab. 18. exp *Staphylococcus/ or staphylococc$.ti,ab. 19. exp *Streptococcus/ or streptococc$.ti,ab. 20. 18 or 19 21. exp *Skin/ 22. 20 and 21 23. 11 or 12 or 13 or 14 or 15 or 16 or 17 or 22 24. 10 and 23
[Lines 1‐10: Cochrane Highly Sensitive Search Strategy for identifying randomized trials in MEDLINE: sensitivity‐ and precision‐maximizing version (2008 revision)]
Appendix 3. EMBASE (Ovid) search strategy
1. exp Erysipelas/ or erysipelas.ti,ab. 2. exp Cellulitis/ or cellulitis.ti,ab. 3. impetigo.ti,ab. or exp *Impetigo/ 4. Ignis sacer.ti,ab. 5. holy fire.ti,ab. 6. st anthony's fire.ti,ab. 7. exp *Staphylococcus/ or staphylococc$.ti,ab. 8. exp *Streptococcus/ or streptococc$.ti,ab. 9. 7 or 8 10. exp *Skin/ 11. 9 and 10 12. exp *soft tissue infection/ 13. 1 or 2 or 3 or 4 or 5 or 6 or 11 or 12 14. random$.mp. 15. factorial$.mp. 16. (crossover$ or cross‐over$).mp. 17. placebo$.mp. or PLACEBO/ 18. (doubl$ adj blind$).mp. 19. (singl$ adj blind$).mp. 20. (assign$ or allocat$).mp. 21. volunteer$.mp. or VOLUNTEER/ 22. Crossover Procedure/ 23. Double Blind Procedure/ 24. Randomized Controlled Trial/ 25. Single Blind Procedure/ 26. 14 or 15 or 16 or 17 or 18 or 19 or 20 or 21 or 22 or 23 or 24 or 25 27. 13 and 26
Appendix 4. LILACS search strategy
(cellulitis or erysipelas or celulitis or "flemon difuso" or erisipela)
In LILACS we searched using the above terms and the Controlled clinical trials topic‐specific query filter.
Data and analyses
Comparison 1. Antibiotic prophylaxis versus no treatment/placebo, on prophylaxis.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Recurrence of cellulitis | 5 | 513 | Risk Ratio (M‐H, Random, 95% CI) | 0.31 [0.13, 0.72] |
| 2 Incidence rate of recurrence of cellulitis | 4 | 4375 | Risk Ratio (M‐H, Random, 95% CI) | 0.44 [0.22, 0.89] |
| 3 Time to next episode of cellulitis | 3 | Hazard Ratio (Random, 95% CI) | 0.51 [0.34, 0.78] | |
| 4 Hospitalisation | 3 | 429 | Risk Ratio (M‐H, Random, 95% CI) | 0.77 [0.37, 1.57] |
| 5 Any adverse reactions | 4 | 469 | Risk Ratio (M‐H, Random, 95% CI) | 0.87 [0.58, 1.30] |
| 5.1 Penicillin | 3 | 437 | Risk Ratio (M‐H, Random, 95% CI) | 0.81 [0.60, 1.10] |
| 5.2 Erythromycin | 1 | 32 | Risk Ratio (M‐H, Random, 95% CI) | 7.0 [0.39, 125.44] |
| 6 Mortality | 3 | 437 | Risk Ratio (M‐H, Random, 95% CI) | 1.12 [0.32, 3.91] |
Comparison 2. Antibiotic prophylaxis versus no treatment/placebo, post‐prophylaxis.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Recurrence of cellulitis | 2 | 287 | Risk Ratio (M‐H, Random, 95% CI) | 0.88 [0.59, 1.31] |
| 2 Incidence rate of recurrence of cellulitis | 2 | 4566 | Risk Ratio (M‐H, Random, 95% CI) | 0.94 [0.65, 1.36] |
| 3 Time to next episode of cellulitis | 2 | Hazard Ratio (Random, 95% CI) | 0.78 [0.39, 1.56] |
Comparison 3. Antibiotic prophylaxis versus no treatment/placebo, overall.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Recurrence of cellulitis | 2 | 397 | Risk Ratio (M‐H, Random, 95% CI) | 0.75 [0.59, 0.95] |
| 2 Incidence rate of recurrence of cellulitis | 2 | 7854 | Risk Ratio (M‐H, Random, 95% CI) | 0.69 [0.56, 0.85] |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Chakroun 1994.
| Methods | A randomised controlled, open‐label, parallel‐group trial | |
| Participants | 1. Setting: the trial recruited participants admitted to the infectious diseases service of the central university hospital in Monastir, Tunisia
2. Number of participants randomised: 58 3. Sex (men/women): 15/29 (14 participants were lost to follow‐up) 4. Mean age ± SD: 46.2 ± 19.4 5. Area of body involved: leg 6. Number of episodes of cellulitis prior to intervention: NR |
|
| Interventions | Study groups:
Duration of treatment: between 1 month and 38 months (average = 11.6 months) Follow‐up: during treatment phase‐ every 3 months; after treatment phase‐ NF |
|
| Outcomes | 1. The number of participants with repeat episodes of cellulitis 2. The number of repeat episodes of cellulitis 3. Costs |
|
| Notes | Criteria for diagnosis of cellulitis: fever + signs of local inflammation confirmed by a single infectious diseases specialist Funding source and Declaration of interest: NC and NR. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "A la sortie du service, un tirage au sort est effactué afin de classer le patient dans l'un des 2 groupes suivants". Comment: Randomisation was done by drawing lots |
| Allocation concealment (selection bias) | Low risk | The author reported using sealed enveloped in a separate email correspondence (Table 3) |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | This was an open‐label study |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | This was an open‐label study |
| Incomplete outcome data (attrition bias) All outcomes | High risk | 76% of participants were followed up (75% of the intervention group‐ 18/24, and 76% of the control group‐ 26/34) with low number of participants and events and 'per protocol' analysis (Table 2) |
| Selective reporting (reporting bias) | Unclear risk | There was insufficient information |
| Similarity of groups at baseline (baseline imbalance bias) | Low risk | Quote: "Les deux groupes sont statistiquement comparables pour les critères sus‐cités (tableau I)". Comment: Baseline characteristics were reported and balanced |
| Early termination (early stopping bias) | Unclear risk | Termination criteria or stopping rule were not reported |
| Other bias | Low risk | No other sources of potential bias were found |
Kasseroller 1998.
| Methods | A randomised, double‐blind, placebo‐controlled parallel‐group trial | |
| Participants | 1. Setting: participants with lymphoedema following mastectomy admitted to Wittlinger's therapy centre in Walchsee, Austria (private rehabilitation clinic)
2. Number of participants randomised: 60 3. Sex (men/women): presumably all women 4. Mean age: 60.5 5. Area of body involved: upper limb 6. Number of episodes of cellulitis prior to intervention: ≥ 4 |
|
| Interventions | Study groups:
Concomitant treatment: 3 weeks of inpatient care (Table 2) of congestion relief for both groups including daily treatment with manual lymph drainage; bandaging; exercise; skin care and high voltage therapy Duration of treatment:15 weeks: 3 weeks of intensive congestion relief treatment and 3 months of oral solution Follow‐up: during intensive treatment phase‐ inpatient care follow‐up, afterwards‐ NR |
|
| Outcomes | 1.The number of participants with repeat episodes of cellulitis 2. Blood selenium levels before and after treatment |
|
| Notes | 1. Criteria for diagnosis of cellulitis: prior to study enrolment diagnosis was made in general and university hospitals; after enrolment diagnosis was based on clinical examination and blood test markers of inflammation (erythrocyte sedimentation rate and CRP) but exact criteria are NR 2. No details reported on high voltage therapy Funding source and Declaration of interest: NR but on further examination we confirmed industrial sponsorship (Biosyn Arzneimittel GmbH ‐ Biosyn 2015, also in Table 3) |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | The paper did not detail the randomisation process |
| Allocation concealment (selection bias) | Unclear risk | The paper did not provide details |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Quote: "double‐blind study". Comment: the paper did not provide details on randomisation process, including similarity of treatment characteristics (possible different taste or colour of selenium solution versus physiological salt solution) or allocation schedule control (breaking of the code for analysis or for medical reasons) |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Quote: "double‐blind study" Comment: the investigator did not identify those blinded in the trial or other necessary data (specified above) to allow judgement of blinding of the participants, care‐givers, outcome assessors or others |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Quote: "During the treatment, some of the patients were excluded from the study since they did not meet the criteria for study inclusion, namely, too short period of stay". Comment: 3 participants were omitted from analyses. The study did not specify exclusion criteria nor the group to which these participants belonged or the reason for their short period of stay in the trial |
| Selective reporting (reporting bias) | High risk | The study report failed to include results for outcomes that would be expected to have been reported for such a study, such as number of episodes of cellulitis, severity of lymphoedema, adverse events. In addition, the investigator reported measurements of selenium blood levels, which had not been proposed prior to the results |
| Similarity of groups at baseline (baseline imbalance bias) | Unclear risk | The investigator did not report on baseline differences between study groups including previous treatment with radiation or other oncological treatment |
| Early termination (early stopping bias) | Unclear risk | The trial ended after 15 weeks but the investigator did not prespecify termination criteria nor did he explain the study's duration |
| Other bias | High risk | 1. Criteria for the diagnosis of cellulitis are unclear, especially in the ambulatory (Table 2) phase of the trial (this might cause differences in diagnosis of cellulitis between the intensive care phase and the ambulatory phase) 2.The investigator did not answer queries (sent by emails, post and professional website ‐ drkasseroller.at (accessed February 2014)) |
Kremer 1991.
| Methods | A randomised controlled, open‐label, parallel‐group trial | |
| Participants | 1. Setting: the trial was conducted in an outpatient (Table 2) setting in northern Israel. The recruitment process of participants was NR
2. Number of participants randomised: 40 3. Sex (men/women): 14/18 (8 participants were lost to follow‐up and their details are NR) 4. Mean age (range): treatment group‐ 63.2 (42 ‐ 75), control group‐ 65.5 (32 ‐ 75) 5. Area of body involved: leg. 2 participants from the control group suffered upper extremity infections 6. Number of episodes of cellulitis prior to intervention: ≥ 2 |
|
| Interventions | Study groups:
Concomitant treatment: local antifungal treatment for tinea pedis Duration of treatment: 18 months Follow‐up:during treatment phase‐monthly, after treatment phase‐ NR |
|
| Outcomes | 1. The number of participants with repeat episodes of cellulitis 2. The number of repeat episodes of cellulitis 3. The number of participants requiring hospitalisation 4. The number of adverse drug reactions |
|
| Notes | Criteria for diagnosis of cellulitis: NR.
Comment: the report mentioned that during follow‐up participants with "a relapse" came to the clinic and were treated according to "clinical findings" Funding source and Declaration of interest: NR. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | The paper did not provide details |
| Allocation concealment (selection bias) | Unclear risk | The paper did not provide details |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | This was an open‐label study |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | This was an open‐label study |
| Incomplete outcome data (attrition bias) All outcomes | High risk | 8 participants were lost to follow‐up (20% of participants in the study) and the paper did not detail to which groups they were assigned; we conducted a 'per protocol' analysis |
| Selective reporting (reporting bias) | Unclear risk | There was insufficient information |
| Similarity of groups at baseline (baseline imbalance bias) | Low risk | Baseline characteristics were reported and balanced (Table 1 in the article). |
| Early termination (early stopping bias) | High risk | Quote: "After I8 months' follow‐up the differences between the two groups were dramatic, and led us to conclude the study" Comment: the study was terminated based on results and did not report sample size calculation, formal interim analysis or a formal stopping rule |
| Other bias | Low risk | No other sources of potential bias were found |
Sjöblom 1993.
| Methods | A randomised controlled, open‐label, parallel‐group trial | |
| Participants | 1. Setting: the trial recruited participants admitted to the infectious diseases department of Roslagstull hospital in Stockholm, Sweden
2. Number of participants randomised: 40 3. Sex (men/women): 20/20 4. Mean age (range): treatment group‐ 67.5 (36 ‐ 87), control group‐ 62.6 (25 ‐ 84) 5. Area of body involved: leg. 1 participant from the control group suffered upper limb lymphoedema after having a mastectomy 6. Number of episodes of cellulitis prior to intervention: ≥ 2 |
|
| Interventions | Study groups:
5 participants that were allergic to penicillin received oral erythromycin 250 mg X 2/d; 250 mg + 500 mg/d and 500 mg X 2/d for the corresponding weight groups
Concomitant treatment:local skin care and compression stockings or elastic bandages Duration of treatment (mean (range), days) ‐ treatment group‐ 443 (50 ‐ 1047), control group‐ 436 (25 ‐ 84) Follow‐up: during treatment phase ‐ every 3 months, after treatment phase ‐ NF |
|
| Outcomes | 1. The number of participants with repeat episodes of cellulitis 2. The number of participants with adverse drug reactions requiring the interruption of treatment and other adverse events of interest (changes in blood cell count, liver enzyme disturbances, gastrointestinal symptoms, e.g. nausea, diarrhoea and rash) 3. Colonisation with streptococci and staphylococci (from blood, skin, nasopharynx and throat cultures) |
|
| Notes | Criteria for diagnosis of cellulitis:a febrile infection of acute onset with a sharply demarcated, warm, indurated and painful erythema (Table 2) accompanied by a temperature of at least 38° C. The diagnosis was made by 2 infectious diseases specialists Funding source and Declaration of interest: NC and NR. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | The investigators described using stratified block randomisation (Table 2) in a separate email correspondence (Table 3) |
| Allocation concealment (selection bias) | Low risk | Quote: "The patients were randomly assigned to treatment or control groups using sealed envelopes". Comment: investigators mentioned in correspondence that envelopes were sequentially numbered |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Quote: "This was an open study" Comment: trial was not blinded |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Quote: "This was an open study" Comment: trial was not blinded |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | 2 participants stopped treatment due to adverse drug reactions (95% follow‐up) and ITT analysis was performed |
| Selective reporting (reporting bias) | Low risk | The publication reported findings on all outcomes listed in the Methods section |
| Similarity of groups at baseline (baseline imbalance bias) | Low risk | Quote: "No major differences concerning predisposing factors were found between the groups". Comment: Baseline characteristics were reported and balanced |
| Early termination (early stopping bias) | Unclear risk | The trial stopped after a mean time of 14.4 months and the paper did not report sample size calculation, a formal stopping rule or results of an interim analysis (stated to be conducted every 6 months for at least 20 participants followed up for a minimum of 1 year) |
| Other bias | High risk | Randomisation included fixed‐size blocks (of 10) and the trial was open, thus allowing predictability |
Thomas 2012.
| Methods | A multicentre, randomised, double‐blind, placebo‐controlled trial | |
| Participants | 1. Setting: the trial recruited participants from 20 hospitals in the UK and Ireland, either in hospital setting or retrospectively via medical coding in records or poster adverts
2. Number of participants randomised: 123 3. Sex (men/women): 42/81 4. Mean age (range): treatment group‐ 56.7 (18 ‐ 81), placebo group‐ 59.5 (23 ‐ 84) 5. Area of body involved: leg 6. Number of episodes of cellulitis prior to intervention: ≥ 1 |
|
| Interventions | Study groups:
Duration of treatment: 6 months Follow‐up: on‐prophylaxis phase‐ telephone calls from co‐ordinating centre every 3 months , post‐prophylaxis phase‐ phone calls at 6‐month intervals. In addition participants were asked to complete a study diary and to immediately inform the trial staff of a repeat episode |
|
| Outcomes |
Primary outcomes: 1. Time to next episode of cellulitis Secondary outcomes: 1. The proportion of participants with repeat episodes of cellulitis at the end of the treatment phase, and at the end of the non‐intervention follow‐up phase 2. The number of repeat episodes of cellulitis 3. The proportion of participants with new oedema and/or ulceration at the end of the treatment phase, and at the end of the non‐intervention follow‐up phase 4. The number of nights in hospital for the treatment of repeat episodes of cellulitis 5. The number of adverse drug reactions and/or adverse events of interest (death, nausea, diarrhoea, thrush, rash) 6. Cost effectiveness 7. Predictors of response model to explore the impact of known risk factors in predicting the efficacy of prophylaxis (stated to be conducted only if a significant treatment effect was found) |
|
| Notes | 1. Criteria for diagnosis of cellulitis: ‐ Criteria for an episode of cellulitis for study eligibility (index episode): a confirmed diagnosis of cellulitis by the recruiting dermatologist; if the patient was not seen by the recruiting clinician, validation of diagnosis was sought from the medical records and interview with the patient. In this case specific criteria were required consistent with clinical signs and symptoms of cellulitis (specified in the report). Any doubt over the certainty of the diagnosis was grounds for exclusion ‐ Criteria for a repeat episode of cellulitis during treatment or follow‐up phases: next episode of cellulitis in either leg that had been reported by the participant and confirmed by a medical practitioner (sensitivity analysis was performed for patient‐reported cases that required antibiotic treatment and were not confirmed as stated) 2. The paper reported good and balanced adherence in both groups: "From self‐reported tablet counts, 97 (79%) patients fully adhered to treatment, defined as at least 75% of tablets taken.This was similar across treatment groups". Funding source and Declaration of interest: NC (The BUPA Health Foundation) and no conflicts of interests. The Thomas 2012 and Thomas 2013 report on 2 studies that were led by the same group of researchers under the title of: Prophylactic Antibiotics for the Treatment of Cellulitis at Home ‐ The PATCH trials (The PATCH II and PATCH I studies, respectively). Similar study designs were reported for both trials including randomisation process, allocation, blinding, definitions of outcomes, monitoring and analysis. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "participants were randomised by staff at the coordinating centre using a web‐based randomisation service provided by the Clinical Trials Unit (CTU)...". Comment: investigators used computer‐generated random list |
| Allocation concealment (selection bias) | Low risk | Quote: "Treatment allocations were concealed from all members of the trial team". Comments: central block randomisation was conducted with varying block sizes |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "Participants and all members of the study team were blinded to treatment allocation throughout the trial, and analysis was performed prior to breaking of the randomization code...Although the treatments were packaged in an identical way, and the placebo tablets were of the same size and shape as penicillin V, the tablets were not identical due to difficulties in obtaining a matched placebo product. Nevertheless, there was a low risk of unblinding...". Comment: The trial included placebo, randomisation list was held by the CTU, breaking of the allocation code was allowed according to decisions by the data monitoring committee and as prespecified in the protocol of the trial |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | As detailed for blinding of the participants and personnel: there was blinding, and it was unlikely that the blinding could have been broken |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Quote: "Of the 123 participants randomized, 20 (16%) participants (11 penicillin V and nine placebo) did not reach the end of the study... Participants in both groups had a similar study time experience, and approximately 80% had at least 2 years of follow‐up". Comment: most of participants completed follow‐up (84%); their number is balanced between groups; the reasons for withdrawal from study are probably not related to treatment or outcome;and ITT was performed |
| Selective reporting (reporting bias) | Low risk | Changes to outcomes, as prespecified in the protocol, were explained. Other outcomes were reported as mentioned in the protocol, that had been registered and available online (via ongoing trials registries and the trial website ‐Thomas 2012) |
| Similarity of groups at baseline (baseline imbalance bias) | Low risk | Quote:"...these stratification factors and other baseline variables were broadly similar across the two treatment groups". Comment: Baseline characteristics were reported and balanced |
| Early termination (early stopping bias) | Low risk | Quote:"...the identification of suitable participants was much slower than anticipated, and recruitment was therefore stopped after 2 years due to funding limitations. The possible reasons for this failure to recruit have been reported elsewhere" (Thomas 2010). Comment: Sample size calculation was stated (a sample of 400 participants) and the goal of recruitment was not achieved but carefully examined. Nevertheless, the study ended according to the protocol (see Thomas 2012) |
| Other bias | Low risk | No other sources of potential bias were found |
Thomas 2013.
| Methods | A multicentre, randomised, double‐blind, placebo‐controlled trial | |
| Participants | 1. Setting:the trial recruited participants from 28 hospitals in the UK and Ireland, either in hospital setting or retrospectively via medical coding in records or poster adverts
2. Number of participants randomised: 274 3. Sex (men/women):109/165 4. Mean age ± SD: treatment group ‐ 58.1 ± 12.6, placebo group ‐ 57.4 ± 14.4 5. Area of body involved: leg 6. Number of episodes of cellulitis prior to intervention: ≥ 2 |
|
| Interventions | Study groups:
Concomitant treatment: "normal clinical practice" for predisposing factors such as tinea pedis Duration of treatment‐ 12 months Follow‐up:on‐prophylaxis phase‐ telephone calls from co‐ordinating centre every 3 months, post‐prophylaxis phase‐ phone calls at 6‐month intervals. In addition participants were asked to complete a study diary and to immediately inform the trial staff on a repeat episode |
|
| Outcomes |
Primary outcomes: 1. Time to next episode of cellulitis Secondary outcomes: 1. The proportion of participants with repeat episodes of cellulitis at the end of the treatment phase, and at the end of the non‐intervention follow‐up phase 2. The number of repeat episodes of cellulitis 3. The proportion of participants with new oedema and/or ulceration at the end of the treatment phase, and at the end of the non‐intervention follow‐up phase 4. The number of nights in hospital for the treatment of repeat episodes of cellulitis 5. The number of adverse drug reactions and/or adverse events of interest (death, nausea, diarrhoea, thrush, rash, severe skin reactions, sepsis, and renal failure) 6. Cost effectiveness 7. Predictors of response model to explore the impact of known risk factors in predicting the efficacy of prophylaxis |
|
| Notes | 1. Criteria for diagnosis of cellulitis: ‐ Criteria for an episode of cellulitis for study eligibility (index episode):a confirmed diagnosis of cellulitis by the recruiting dermatologist; if the patient was not seen by the recruiting clinician, validation of diagnosis was sought from the medical records and interview with the patient. In this case specific criteria were required consistent with clinical signs and symptoms of cellulitis (specified in the report). Any doubt over the certainty of the diagnosis was grounds for exclusion ‐ Criteria for a repeat episode of cellulitis during treatment or follow‐up phases: next episode of cellulitis in either leg that had been reported by the participant and confirmed by a medical practitioner (sensitivity analysis was performed for patient‐reported cases that required antibiotic treatment and were not confirmed as stated). 2. The paper reported good and balanced adherence in both groups: "A total of 214 participants (78%) reported taking at least 75% of the study tablets; the proportion of patients who reported taking at least 75% of the tablets was similar in the two groups". Funding source and Declaration of interest: NC (Action Medical Research‐ medical research charity) and no conflicts of interests The Thomas 2012 and Thomas 2013 report on 2 studies that were led by the same group of researchers under the title of: Prophylactic Antibiotics for the Treatment of Cellulitis at Home ‐ The PATCH trials (The PATCH II and PATCH I studies, respectively). Similar study designs were reported for both trials including randomisation process, allocation, blinding, definitions of outcomes, monitoring and analysis. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "The coordinating center randomly assigned the participants with the use of the Nottingham Clinical Trials Unit (NCTU) Web‐based randomization service". Comment: Investigators used computer‐generated random list |
| Allocation concealment (selection bias) | Low risk | Quote: "The computer‐generated randomization list was produced before the start of recruitment, with the use of randomly varying block sizes, and was held by the NCTU". Comment: Central block randomisation was conducted with varying block sizes |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "Participants and all members of the study team were unaware of the treatment assignments throughout the trial, and the analysis was performed before the breaking of the randomization code". Comment: The trial included placebo, randomisation list was held by the CTU, breaking of the allocation code was allowed according to decisions by the data monitoring committee and as prespecified in the protocol of the trial |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | As detailed for blinding of the participants and personnel: there was blinding, and it was unlikely that the blinding could have been broken |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Quote: "A total of 247 patients (90%) underwent at least 18 months of follow‐up (median, 25)" Comment: attrition was low |
| Selective reporting (reporting bias) | Low risk | All the outcomes prespecified in the protocol were reported and any changes to its plan were explained (see Thomas 2013) |
| Similarity of groups at baseline (baseline imbalance bias) | Low risk | Quote: "The baseline characteristics of the participants were well balanced between the groups". Comment: Baseline characteristics were reported and balanced |
| Early termination (early stopping bias) | Low risk | Sample size calculation was stated (a sample of 260 participants), the goal of recruitment was achieved (274 participants randomised), and the study did not terminate prematurely |
| Other bias | Low risk | No other sources of potential bias were found. |
Abbreviations:
d ‐ Day g ‐ Microgram ITT ‐ Intention‐to‐treat (Table 2) NC ‐ Non‐commercial NF ‐ No follow‐up NR ‐ Not reported SD ‐ Standard deviation
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Duvanel 1986 | This was a retrospective cohort study (Table 2) |
| Ferrieri 1973 | Participants were children (ages 1 to 14) |
| Fritz 2011 | The study mainly investigated recurrent purulent abscesses in children |
| Haustein 1989 | This was a retrospective cohort study |
| Klempner 1988 | This study investigated prophylaxis for recurrent skin abscesses |
| Maddox 1985 | Participants were children (ages 2 to 5) |
| Raz 1996 | This study investigated prophylaxis for recurrent furunculosis and folliculitis (Table 2) |
| Wang 1997 | This was a non‐randomised study |
Characteristics of studies awaiting assessment [ordered by study ID]
Ratnikova 1991.
| Methods | Interventional study, exact methods are unclear |
| Participants | 66 participants with recurrent erysipelas, probably from Russia |
| Interventions | Study groups:
|
| Outcomes | Not reported |
| Notes |
|
Differences between protocol and review
Background: we updated the Background as we found other official guidelines during the search (the Australasian Lymphology Association consensus guideline, the International Society of Lymphology consensus document, the Italian Society of Infectious Diseases and International Society of Chemotherapy's consensus statement, the Dutch College of General Practitioners' practice guideline, the English National Institute for Health and Clinical Excellence (NICE) guideline, the International Lymphoedema Framework consensus document and the Finnish Dermatological Society's care guideline).
Objectives: the main objective of the review was changed and rephrased to reflect the importance of both the beneficial as well as the adverse effects of the intervention (therefore "effectiveness" was changed to "beneficial and adverse effects"). We also clarified the types of included participants.
Types of interventions: we contacted authors for results relating to cellulitis if they were not reported separately.
Types of outcome measures: we defined the relevant time points for extraction of data as it was not stated in original version of the protocol. The primary time point for analysis was the end of treatment phase ('on prophylaxis) and the secondary time points: after the cessation of treatment ('post‐prophylaxis') and at the end of follow‐up ('overall').
Types of outcome measures > Primary outcomes: we clarified that when data were available we defined recurrence as a repeat episode of cellulitis in the same limb.
Types of outcome measures > Secondary outcomes: we added another outcome of 'Mortality'. We intended to include this outcome under 'adverse reactions' but decided to separate mortality and morbidity outcomes for the reader's convenience.
Types of outcome measures > Secondary outcomes: for clarification, we slightly reworded and added extra information with regards to time points for secondary outcomes 1 and 2.
Electronic searches: we had planned in the protocol to search ISI Web of Science and NLM gateway but we chose not to search through the NLM Gateway as it had transitioned to a new pilot project. We did not search ISI Web of Science because of technical issues and database availability. We also updated the metaRegister of Controlled Trials website address
Data collection and analysis > Unit of analysis issues: we specifically referred to cross‐over trials and why their design would not be expected to be used.
Data collection and analysis > Dealing with missing data: we added the planned analysis of missing data that was omitted from the original protocol.
Data collection and analysis > Data synthesis: in our protocol we specified that we would use a Mantel‐Haenszel fixed‐effect model to calculate the treatment effect across trials. However, we decided instead to use a random‐effects models throughout, as we predicted there would be clinical and methodological heterogeneity between studies.
Data collection and analysis > Measures of treatment effect: we calculated the number needed to treat for an additional beneficial outcome (NNTB) for the primary outcome when the result was significant.
Data collection and analysis > 'Summary of findings' table: to adhere to Cochrane standards, we included a 'Summary of findings' table and described the basis on which it was created.
Searching other resources > Grey literature: we planned in the protocol to search BIOSIS Previews from 1969 but only searched from 1990 in the review, due to technical reasons and database availability.
Assessment of risk of bias in included studies: in the review we addressed other risks of bias under several headings which we had not planned and failed to include in the protocol. We included these because we thought them important: Baseline imbalance; Early termination; Other potential bias.
Subgroup analysis and investigation of heterogeneity: We planned to explore reasons for substantial heterogeneity (I² > 50%) in any meta‐analyses we performed but there were insufficient studies for the subgroups we planned. We performed a post hoc subgroup analysis with respect to the type of antibiotic because side effects are a critical outcome for the intervention assessed and side effects are drug‐specific.
Sensitivity analyses: We planned to explore heterogeneity through a sensitivity analysis; however, paucity of data for the relevant interventions made sensitivity analysis impractical.
Tables > 'Summary of findings' table: we included a 'Summary of findings' table as recommended by Cochrane guidelines. In addition, we also used the five GRADE considerations (study limitations, consistency of effect, imprecision, indirectness, and publication bias) to assess the quality of the body of evidence.
Contributions of authors
AD was the contact person with the editorial base, AD and MP co‐ordinated contributions from the co‐authors, and wrote the final draft of the review. AD and SR screened papers against eligibility criteria. AD obtained data on ongoing and unpublished studies. AD, MS and MP appraised the quality of papers. AD, MS and MP extracted data for the review and sought additional information about papers. AD entered data into Review Manager 5. AD, MS, DM, SR, WD, EH, LL and EH analysed and interpreted data. AD, LL and MP worked on the Methods sections. AD and MP drafted the clinical sections of the Background and responded to the clinical comments of the referees. AD, LL and MP responded to the methodology and statistics comments of the referees. WD was the consumer co‐author and checked the review for readability and clarity, as well as ensuring outcomes are relevant to consumers. AD is the guarantor of the update.
Disclaimer
This project was supported by the National Institute for Health Research, via Cochrane Infrastructure funding to the Cochrane Skin Group. The views and opinions expressed therein are those of the authors and do not necessarily reflect those of the Systematic Reviews Programme, NIHR, NHS or the Department of Health.
Sources of support
Internal sources
No sources of support supplied
External sources
-
The National Institute for Health Research (NIHR), UK.
The NIHR, UK, is the largest single funder of the Cochrane Skin Group.
Declarations of interest
Adam Dalal: nothing to declare. Marina Eskin‐Shwartz: nothing to declare. Daniel Mimouni: nothing to declare. Sujoy Ray: nothing to declare. Walford Days: nothing to declare. Emmilia Hodak: nothing to declare. Leonard Leibovici: nothing to declare. Mical Paul: nothing to declare. Oh Choon Chiat, who refereed this review, is the author/co‐author of two papers (Oh 2014; Tay 2015) cited in this review.
New
References
References to studies included in this review
Chakroun 1994 {published and unpublished data}
- Chakroun M, Ben‐Romdhane F, Battikh R, Souki A, Bouzouaia N. Benzathine penicillin prophylaxis in recurrent erysipelas [Intérêt de la benzathine pénicilline dans la prévention des récidives d'érysipèle]. Médecine et Maladies Infectieuses 1994;24(10):894‐7. [CENTRAL: CN‐00169390; EMBASE: 1994338740] [Google Scholar]
Kasseroller 1998 {published data only (unpublished sought but not used)}
- Kasseroller R. Prophylaxis of erysipelas in secondary lymphedema with selenium [Erysipelprophylaxe beim sekundären Lymphödem mit Selen]. Der Allgemeinarzt 1996;18(3):244‐7. [Google Scholar]
- Kasseroller R. Sodium selenite as prophylaxis against erysipelas in secondary lymphedema. Anticancer Research 1998;18(3C):2227‐30. [CENTRAL: CN‐00153878; EMBASE: 1998283262] [PubMed] [Google Scholar]
Kremer 1991 {published data only (unpublished sought but not used)}
- Kremer M, Zuckerman R, Avraham Z, Raz R. Long‐term antimicrobial therapy in the prevention of recurrent soft‐tissue infections. Journal of Infection 1991;22(1):37‐40. [CENTRAL: CN‐00073794; PUBMED: 2002231] [DOI] [PubMed] [Google Scholar]
Sjöblom 1993 {published and unpublished data}
- Sjöblom AC, Eriksson B, Jorup‐Rönström C, Karkkonen K, Lindqvist M. Antibiotic prophylaxis in recurrent erysipelas. Infection 1993;21(6):390‐3. [CENTRAL: CN‐00099842; PUBMED: 8132369] [DOI] [PubMed] [Google Scholar]
Thomas 2012 {published and unpublished data}
- Centre of Evidence Based Dermatology. Prophylactic Antibiotics for the Treatment of Cellulitis at Home (PATCH). www.nottingham.ac.uk/research/groups/cebd/projects/patch.aspx (accessed 6th June 2017).
- Mason JM, Thomas KS, Crook AM, Foster KA, Chalmers JR, Nunn AJ, et al. Prophylactic antibiotics to prevent cellulitis of the leg: economic analysis of the PATCH I & II Trials. PLoS One 2014;9(2):e82694. [DOI: 10.1371/journal.pone.0082694; PUBMED: 24551029] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas KS, UK Dermatology Clinical Trials Network's PATCH study group. Studying a disease with no home‐‐lessons in trial recruitment from the PATCH II study. Trials 2010;11(22):1‐5. [DOI: 10.1186/1745-6215-11-22; PUBMED: 20196846] [DOI] [PMC free article] [PubMed] [Google Scholar]
- UK Dermatology Clinical Trials Network’s PATCH Trial Team, Thomas K, Crook A, Foster K, Mason J, Chalmers J, Bourke J, et al. Prophylactic antibiotics for the prevention of cellulitis (erysipelas) of the leg: results of the UK Dermatology Clinical Trials Network's PATCH II trial. British Journal of Dermatology 2012;166(1):169‐78. [CENTRAL: CN‐00836571; PUBMED: 21910701] [DOI] [PMC free article] [PubMed] [Google Scholar]
Thomas 2013 {published and unpublished data}
- Centre of Evidence Based Dermatology. Prophylactic Antibiotics for the Treatment of Cellulitis at Home (PATCH). www.nottingham.ac.uk/research/groups/cebd/projects/patch.aspx (accessed 6th June 2017).
- Mason JM, Thomas KS, Crook AM, Foster KA, Chalmers JR, Nunn AJ, et al. Prophylactic antibiotics to prevent cellulitis of the leg: economic analysis of the PATCH I & II Trials. PLoS One 2014;9(2):e82694. [DOI: 10.1371/journal.pone.0082694; PUBMED: 24551029] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas KS, Crook AM, Nunn AJ, Foster KA, Mason JM, Chalmers JR, et al. Penicillin to prevent recurrent leg cellulitis. New England Journal of Medicine 2013;368(18):1695‐703. [CENTRAL: CN‐00859316; PUBMED: 23635049] [DOI] [PubMed] [Google Scholar]
- Williams HC, Crook AM, Mason JM, UK Dermatology Clinical Trials Network’s PATCH I Trial Team. Penicillin to prevent recurrent leg cellulitis. New England Journal of Medicine 2013;369(9):881‐2. [DOI: 10.1056/NEJMc1307321; PUBMED: 23984744] [DOI] [PubMed] [Google Scholar]
References to studies excluded from this review
Duvanel 1986 {published data only (unpublished sought but not used)}
- Duvanel T, Harms M, Merot Y, Saurat JH. Les récidives d’érysipèle et leur prévention par la benzathine‐penicilline. Annales de Dermatologie et de Vénéréologie 1986;113:174‐6. [Google Scholar]
Ferrieri 1973 {published data only}
- Ferrieri P, Dajani AS, Wannamaker LW. Benzathine penicillin in the prophylaxis of streptococcal skin infections: a pilot study. Journal of Pediatrics 1973;83(4):572‐7. [CENTRAL: CN‐00009022; PUBMED: 4581016] [DOI] [PubMed] [Google Scholar]
Fritz 2011 {published and unpublished data}
- Fritz SA, Camins BC, Eisenstein KA, Fritz JM, Epplin EK, Burnham CA, et al. Effectiveness of measures to eradicate Staphylococcus aureus carriage in patients with community‐associated skin and soft‐tissue infections: a randomized trial. Infection Control and Hospital Epidemiology 2011;32(9):872‐80. [CENTRAL: CN‐00804495; PUBMED: 21828967] [DOI] [PMC free article] [PubMed] [Google Scholar]
Haustein 1989 {published data only}
- Haustein UF, Biella U, Tausch I, Knöll H. Treatment of chronic recurrent erysipelas with streptococcal vaccine [Die behandlung des chronisch rezidivierenden erysipels mit Streptokokkenvakzine]. Hautarzt 1989;40(4):215‐21. [PUBMED: 2659550] [PubMed] [Google Scholar]
Klempner 1988 {published data only}
- Klempner MS, Styrt B. Prevention of recurrent staphylococcal skin infections with low‐dose oral clindamycin therapy. JAMA 1988;260(18):2682‐5. [CENTRAL: CN‐00056409; PUBMED: 3184334] [PubMed] [Google Scholar]
Maddox 1985 {published data only}
- Maddox JS, Ware JC, Dillon HC. The natural history of streptococcal skin infection: prevention with topical antibiotics. Journal of the American Academy of Dermatology 1985;13(2 Pt 1):207‐12. [CENTRAL: CN‐00039750; PUBMED: 2995463] [DOI] [PubMed] [Google Scholar]
Raz 1996 {published data only}
- Raz R, Miron D, Colodner R, Staler Z, Samara Z, Keness Y. A 1‐year trial of nasal mupirocin in the prevention of recurrent staphylococcal nasal colonization and skin infection. Archives of Internal Medicine 1996;156(10):1109‐12. [CENTRAL: CN‐00124903; PUBMED: 8638999] [PubMed] [Google Scholar]
Wang 1997 {published data only}
- Wang JH, Liu YC, Cheng DL, Yen MY, Chen YS, Wang JH, et al. Role of benzathine penicillin G in prophylaxis for recurrent streptococcal cellulitis of the lower legs. Clinical Infectious Diseases 1997;25(3):685‐9. [CENTRAL: CN‐00143909; PUBMED: 9314462] [DOI] [PubMed] [Google Scholar]
References to studies awaiting assessment
Ratnikova 1991 {published data only}
- Ratnikova LI. Effectiveness of bemitil in recurrent erysipelas [Эффективность бемитила при рецидивирующей роже]. Klinicheskaia Meditsina 1991;69(7):89‐90. [CENTRAL: CN‐00079309; EMBASE: 1942990] [PubMed] [Google Scholar]
Additional references
Addis 2011
- Addiss DG, Michel MC, Michelus A, Radday J, Billhimer W, Louis‐Charles J, et al. Evaluation of antibacterial soap in the management of lymphoedema in Leogane, Haiti. Transactions of the Royal Society of Tropical Medicine and Hygiene 2011;105(1):58‐60. [DOI] [PubMed] [Google Scholar]
ALA 2015
- Australasian Lymphology Association (ALA). Consensus Guideline: Management of Cellulitis in Lymphoedema. www.lymphoedema.org.au/public/7/files/ALA%20CELLULITIS%20IN%20LYMPHOEDEMA%202015.pdf (accessed May 2017).
Allard 1999
- Allard P, Stücker M, Kobyletzki G, Gammal S, Altmeyer P. Cyclical intravenous antibiosis as an effective therapy concept in chronic recurrent erysipelas [Zyklische intravenöse Antibiose als effizientes Therapiekonzept des chronisch‐rezidivierenden Erysipels]. Hautarzt 1999;50(1):34‐8. [PUBMED: 10068929] [DOI] [PubMed] [Google Scholar]
Arakaki 2014
- Arakaki RY, Strazzula L, Woo E, Kroshinsky D. The impact of dermatology consultation on diagnostic accuracy and antibiotic use among patients with suspected cellulitis seen at outpatient internal medicine offices: a randomized clinical trial. JAMA Dermatology 2014;150(10):1056‐61. [PUBMED: 25143179] [DOI] [PubMed] [Google Scholar]
Arasaratnam 2013
- Arasaratnam R. Penicillin to prevent recurrent leg cellulitis. New England Journal of Medicine 2013;369(9):881. [DOI: 10.1056/NEJMc1307321#SA2; PUBMED: 23984746] [DOI] [PubMed] [Google Scholar]
Arsenault 2011
- Arsenault K, Rielly L, Wise H. Effects of complete decongestive therapy on the incidence rate of hospitalization for the management of recurrent cellulitis in adults with lymphedema. Rehabilitation Oncology 2011;29(3):14‐20. [EMBASE: 2011656495] [Google Scholar]
Arumugam 2012
- Arumugam D, Atherton JJ, Martin PT. A lethal injection?. Lancet 2012;379(9814):492. [PUBMED: 22305227] [DOI] [PubMed] [Google Scholar]
Babb 1966
- Babb RR, Spittell JA Jr, Martin WJ, Schirger A. Prophylaxis of recurrent lymphangitis complicating lymphedema. JAMA 1966;195(10):871‐3. [PUBMED: 12608187] [DOI] [PubMed] [Google Scholar]
Baddour 2000
- Baddour LM. Cellulitis syndromes: an update. International Journal of Antimicrobial Agents 2000;14(2):113‐6. [PUBMED: 10720800] [DOI] [PubMed] [Google Scholar]
Bartholomeeusen 2007
- Bartholomeeusen S, Vandenbroucke J, Truyers C, Buntinx F. Epidemiology and comorbidity of erysipelas in primary care. Dermatology 2007;215(2):118‐22. [PUBMED: 17684373] [DOI] [PubMed] [Google Scholar]
Bernard 1987
- Bernard P, Toty L, Mounier M, Denis F, Bonnetblanc JM. Early detection of streptococcal group antigens in skin samples by latex particle agglutination. Archives of Dermatology 1987;123(4):468‐70. [PUBMED: 3827278] [PubMed] [Google Scholar]
Biosyn 2015
- Biosyn Selenase®. Selenium as a beacon of hope [Selen als Hoffnungsträge]. www.biosyn.de/innovation/selen‐als‐hoffnungstraeger (accessed January 2015).
Bisno 1996
- Bisno AL, Stevens DL. Streptococcal infections of skin and soft tissues. New England Journal of Medicine 1996;334(4):240‐5. [PUBMED: 8532002] [DOI] [PubMed] [Google Scholar]
Bitnun 1985
- Bitnun S. Prophylactic antibiotics in recurrent erysipelas. Lancet 1985;1(8424):345. [PUBMED: 2857399] [DOI] [PubMed] [Google Scholar]
Bjornsdottir 2005
- Bjornsdottir S, Gottfredsson M, Thorisdottir AS, Gunnarsson GB, Rikardsdottir H, Kristjansson M, et al. Risk factors for acute cellulitis of the lower limb: a prospective case‐control study. Clinical Infectious Diseases 2005;41(10):1416‐22. [PUBMED: 16231251] [DOI] [PubMed] [Google Scholar]
BLS 2016
- British Lymphology Society (BLS). Consensus Document on the Management of Cellulitis in Lymphoedema. Revised Cellulitis Guidelines 2016. www.lymphoedema.org/images/pdf/CellulitisConsensus.pdf (accessed May 2017).
Bowler 2001
- Bowler PG, Duerden BI, Armstrong DG. Wound microbiology and associated approaches to wound management. Clinical Microbiology Reviews 2001;14(2):244‐69. [PUBMED: 11292638] [DOI] [PMC free article] [PubMed] [Google Scholar]
Brook 1995
- Brook I, Frazier EH. Clinical features and aerobic and anaerobic microbiological characteristics of cellulitis. Archives of Surgery 1995;130(7):786‐92. [PUBMED: 7611872] [DOI] [PubMed] [Google Scholar]
Brorson 2000
- Brorson H. Liposuction gives complete reduction of chronic large arm lymphedema after breast cancer. Acta Oncologica 2000;39(3):407‐20. [PUBMED: 10987239] [DOI] [PubMed] [Google Scholar]
Bruun 2015
- Bruun T, Oppegaard O, Kittang BR, Mylvaganam H, Langeland N, Skrede S. Etiology of cellulitis and clinical prediction of streptococcal disease: a prospective study. Open Forum Infectious Diseases 2015;31(1):ofv181. [PUBMED: 26734653] [DOI] [PMC free article] [PubMed] [Google Scholar]
Campisi 2015
- Campisi CC, Ryan M, Boccardo F, Campisi C. A single‐site technique of multiple lymphatic‐venous anastomoses for the treatment of peripheral lymphedema: long‐term clinical outcome. Journal of Reconstructive Microsurgery 2016;32(1):42‐9. [PUBMED: 26029991] [DOI] [PubMed] [Google Scholar]
Carratala 2003
- Carratala J, Roson B, Fernandez‐Sabe N, Shaw E, Rio O, Rivera A, et al. Factors associated with complications and mortality in adult patients hospitalized for infectious cellulitis. European Journal of Clinical Microbiology & Infectious Diseases 2003;22(3):151‐7. [PUBMED: 12649712] [DOI] [PubMed] [Google Scholar]
Chaniotakis 2016
- Chaniotakis I, Gartzonika CG, Gaitanis G, Levidiotou‐Stefanou S, Bassukas ID. Causality evaluation of bacterial species isolated from patients with community‐acquired lower leg cellulitis. Journal of the European Academy of Dermatology and Venereology 2016;30(9):1583‐9. [PUBMED: 27400752] [DOI] [PubMed] [Google Scholar]
Chen 2015
- Chen HM, Li YL, Liu YM, Liu CE, Cheng YR, Chen CH, et al. The experience of intramuscular benzathine penicillin for prophylaxis of recurrent cellulitis: A cohort study. Journal of Microbiology, Immunology, & Infection 2015;S1684‐1182(15):00831‐2. [PUBMED: 26475200] [DOI] [PubMed] [Google Scholar]
Chira 2010
- Chira S, Miller LG. Staphylococcus aureus is the most common identified cause of cellulitis: a systematic review. Epidemiology and Infection 2010;138(3):313‐7. [PUBMED: 19646308] [DOI] [PubMed] [Google Scholar]
Corey 2014
- Corey GR, Kabler H, Mehra P, Gupta S, Overcash JS, Porwal A, et al. Single‐dose oritavancin in the treatment of acute bacterial skin infections. New England Journal of Medicine 2014;370(23):2180‐90. [PUBMED: 24897083] [DOI] [PubMed] [Google Scholar]
Cox 1998
- Cox NH, Colver GB, Paterson WD. Management and morbidity of cellulitis of the leg. Journal of The Royal Society of Medicine 1998;91(12):634‐7. [DOI] [PMC free article] [PubMed] [Google Scholar]
Cox 2006
- Cox NH. Oedema as a risk factor for multiple episodes of cellulitis/erysipelas of the lower leg: a series with community follow‐up. British Journal of Dermatology 2006;155(5):947‐50. [PUBMED: 17034523 ] [DOI] [PubMed] [Google Scholar]
CREST 2005
- CREST. Clinical Resource Efficiency Support Team (CREST). Guidelines on the management of cellulitis in adults. www.acutemed.co.uk/docs/Cellulitis%20guidelines,%20CREST,%2005.pdf 2005 (accessed 6th June 2017).
Crisp 2015
- Crisp JG, Takhar SS, Moran GJ, Krishnadasan A, Dowd SE, Finegold SM, et al. Inability of polymerase chain reaction, pyrosequencing, and culture of infected and uninfected site skin biopsy specimens to identify the cause of cellulitis. Clinical Infectious Diseases 2015;61(11):1679‐87. [PUBMED: 26240200] [DOI] [PubMed] [Google Scholar]
Damstra 2008
- Damstra RJ, Steensel MA, Boomsma JH, Nelemans P, Veraart JC. Erysipelas as a sign of subclinical primary lymphoedema: a prospective quantitative scintigraphic study of 40 patients with unilateral erysipelas of the leg. British Journal of Dermatology 2008;158(6):1210‐5. [PUBMED: 18363756] [DOI] [PubMed] [Google Scholar]
Damstra 2009
- Damstra RJ, Voesten HG, Schelven WD, Lei B. Lymphatic venous anastomosis (LVA) for treatment of secondary arm lymphedema. A prospective study of11 LVA procedures in 10 patients with breast cancer related lymphedema and a critical review of the literature. Breast Cancer Research & Treatment 2009;113(2):199‐206. [PUBMED: 18270813] [DOI] [PubMed] [Google Scholar]
David 2011
- David CV, Chira S, Eells SJ, Ladrigan M, Papier A, Miller LG, et al. Diagnostic accuracy in patients admitted to hospitals with cellulitis. Dermatology Online Journal 2011;17(3):1. [PUBMED: 21426867] [PubMed] [Google Scholar]
De Godoy 2000
- Godoy JM, Godoy MF, Valente A, Camacho EL, Paiva EV. Lymphoscintigraphic evaluation in patients after erysipelas. Lymphology 2000;33(4):177‐80. [PUBMED: 11191659] [PubMed] [Google Scholar]
Didem 2005
- Didem K, Ufuk YS, Serdar S, Zümre A. The comparison of two different physiotherapy methods in treatment of lymphedema after breast surgery. Breast Cancer Research & Treatment 2005;93(1):49‐54. [PUBMED: 16184458] [DOI] [PubMed] [Google Scholar]
Draijer 2008
- Draijer LW, Koning S, Wielink G, Boukes FS, Goudswaard AN. Summary of the practice guideline 'Bacterial skin infections' (first revision) from the Dutch College of General Practitioners [Samenvatting van de standaard ‘Bacteriële huidinfecties’(eerste herziening) van het Nederlands Huisartsen Genootschap]. Nederlands Tijdschrift voor Geneeskunde 2008;152(29):1619‐25. [PUBMED: 18998269] [PubMed] [Google Scholar]
Duodecim 1999
- Finnish Medical Society Duodecim and the Finnish Dermatological Society. Bacterial skin infections (online). Current care guideline. October 1999 [updated November 2010] [Ihon bakteeri‐infektiot]. www.kaypahoito.fi (accessed 11 February 2016):1‐3 (accessed December 2013).
Dupuy 1999
- Dupuy A, Benchikhi H, Roujeau JC, Bernard P, Vaillant L, Chosidow O, et al. Risk factors for erysipelas of the leg (cellulitis): case‐control study. BMJ 1999;318(7198):1591‐4. [PUBMED: 10364117] [DOI] [PMC free article] [PubMed] [Google Scholar]
Durand 2013
- Durand M, Wistaff R, Lelorier J. Penicillin to prevent recurrent leg cellulitis. New England Journal of Medicine 2013;369(9):880. [DOI: 10.1056/NEJMc1307321#SA1; PUBMED: 23984745] [DOI] [PubMed] [Google Scholar]
Duvanel 1985
- Duvanel T, Merot Y, Harms M, Saurat JH. Prophylactic antibiotics in erysipelas. Lancet 1985;1(8442):1401. [PUBMED: 2861358] [DOI] [PubMed] [Google Scholar]
Duvanel 1989
- Duvanel T, Auckenthaler R, Rohner P, Harms M, Saurat JH. Quantitative cultures of biopsy specimens from cutaneous cellulitis. Archives of Internal Medicine 1989;149(2):293‐6. [PUBMED: 2644902] [PubMed] [Google Scholar]
Egger 1997
- Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta‐analysis detected by a simple, graphical test. BMJ 1997;315(7109):629‐34. [PUBMED: 9310563] [DOI] [PMC free article] [PubMed] [Google Scholar]
Ellis 2006
- Ellis Simonsen SM, Orman ER, Hatch BE, Jones SS, Gren LH, Hegmann KT, et al. Cellulitis incidence in a defined population. Epidemiology & Infection 2006;134(2):293‐9. [PUBMED: 16490133] [DOI] [PMC free article] [PubMed] [Google Scholar]
Eriksson 1996
- Eriksson B, Jorup‐Rönström C, Karkkonen K, Sjöblom AC, Holm SE. Erysipelas: clinical and bacteriologic spectrum and serological aspects. Clinical Infectious Diseases 1996;23(5):1091‐8. [PUBMED: 8922808] [DOI] [PubMed] [Google Scholar]
Eron 2003
- Eron LJ, Lipsky BA, Low DE, Nathwani D, Tice AD, Volturo GA. Managing skin and soft tissue infections: expert panel recommendations on key decision points. Journal of Antimicrobial Chemotherapy 2003;52(Suppl 1):i3‐17. [PUBMED: 14662806] [DOI] [PubMed] [Google Scholar]
Esposito 2011
- Esposito S, Bassetti M, Borre S, Bouza E, Dryden M, Fantoni M, et al. Diagnosis and management of skin and soft‐tissue infections (SSTI): a literature review and consensus statement on behalf of the Italian Society of Infectious Diseases and International Society of Chemotherapy. Journal of Chemotherapy 2011;23(5):251‐62. [PUBMED: 22005055] [DOI] [PubMed] [Google Scholar]
Ezzo 2015
- Ezzo J, Manheimer E, McNeely ML, Howell DM, Weiss R, Johansson KI, et al. Manual lymphatic drainage for lymphedema following breast cancer treatment. Cochrane Database of Systematic Reviews 2015, Issue 5. [DOI: 10.1002/14651858.CD003475.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Finlay 1994
- Finlay AY, Khan GK. Dermatology Life Quality Index (DLQI)‐‐a simple practical measure for routine clinical use. Clinical & Experimental Dermatology 1994;19(3):210‐6. [PUBMED: 8033378] [DOI] [PubMed] [Google Scholar]
Garau 2015
- Garau J, Blasi F, Medina J, McBride K, Ostermann H, REACH study group. Early response to antibiotic treatment in European patients hospitalized with complicated skin and soft tissue infections: analysis of the REACH study. BMC Infectious Diseases 2015;15(78):1‐9. [PUBMED: 25879713] [DOI] [PMC free article] [PubMed] [Google Scholar]
Gerber 1995
- Gerber MA. Antibiotic resistance in group A streptococci. Pediatric Clinics of North America 1995;42(3):539‐51. [PUBMED: 7761140] [DOI] [PubMed] [Google Scholar]
Goettsch 2006
- Goettsch WG, Bouwes Bavinck JN, Herings RM. Burden of illness of bacterial cellulitis and erysipelas of the leg in the Netherlands. Journal of the European Academy of Dermatology and Venereology 2006;20(7):834‐9. [PUBMED: 16898907] [DOI] [PubMed] [Google Scholar]
Goscinski 2006
- Goscinski G, Tano E, Thulin P, Norrby‐Teglund A, Sjölin J. Release of SpeA from Streptococcus pyogenes after exposure to penicillin: dependency on dose and inhibition by clindamycin. Scandinavian Journal of Infectious Diseases 2006;38(11‐12):983‐7. [PUBMED: 17148065] [DOI] [PubMed] [Google Scholar]
Granzow 2014
- Granzow JW, Soderberg JM, Kaji AH, Dauphine C. Review of current surgical treatments for lymphedema. Annals of Surgical Oncology 2014;21(4):1195‐201. [PUBMED: 24558061] [DOI] [PubMed] [Google Scholar]
Guillemot 1998
- Guillemot D, Carbon C, Balkau B, Geslin P, Lecoeur H, Vauzelle‐Kervroëdan F, et al. Low dosage and long treatment duration of beta‐lactam: risk factors for carriage of penicillin‐resistant Streptococcus pneumoniae. JAMA 1998;279(5):365‐70. [PUBMED: 9459469] [DOI] [PubMed] [Google Scholar]
Halpern 2008
- Halpern J, Holder R, Langford NJ. Ethnicity and other risk factors for acute lower limb cellulitis: a UK‐based prospective case‐control study. British Journal of Dermatology 2008;158(6):1288‐92. [PUBMED: 18341662] [DOI] [PubMed] [Google Scholar]
Harris 2001
- Harris SR, Hugi MR, Olivotto IA, Levine M. Clinical practice guidelines for the care and treatment of breast cancer: 11. Lymphedema. CMAJ: Canadian Medical Association Journal 2001;164(2):191‐9. [PUBMED: 11332311] [PMC free article] [PubMed] [Google Scholar]
Higgins 2011
- Higgins JPT, Green S, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from www.handbook.cochrane.org.
Hook 1986
- Hook EW 3rd, Hooton TM, Horton CA, Coyle MB, Ramsey PG, Turck M. Microbiologic evaluation of cutaneous cellulitis in adults. Archives of Internal Medicine 1986;146(2):295‐7. [PUBMED: 3947189] [PubMed] [Google Scholar]
Horn 1998
- Horn DL, Zabriskie JB, Austrian R, Cleary PP, Ferretti JJ, Fischetti VA. Why have group A streptococci remained susceptible to penicillin? Report on a symposium. Clinical Infectious Diseases 1998;26(6):1341‐5. [PUBMED: 9636860] [DOI] [PubMed] [Google Scholar]
ILF 2006
- International Lymphoedema Framework (ILF). Best Practice for the Management of Lymphoedema. International consensus 2006. http://www.lympho.org/portfolio/best‐practice‐for‐the‐management‐of‐lymphoedema/ (accessed 6th June 2017):1‐60.
Inghammar 2014
- Inghammar M, Rasmussen M, Linder A. Recurrent erysipelas‐risk factors and clinical presentation. BMC Infectious Diseases 2014 May;14:270. [DOI: 10.1186/1471-2334-14-270; PUBMED: 24884840] [DOI] [PMC free article] [PubMed] [Google Scholar]
ISL 2013
- International Society of Lymphology (ISL). The diagnosis and treatment of peripheral lymphedema: Consensus document. Lymphology 2013;46(1):1‐11. [PUBMED: 23930436] [PubMed] [Google Scholar]
Israeli Ministry of Health 2015
- Israeli Ministry of Health. Safety warning over the use of intramuscular Benzathine benzylpenicillin February 2015 [הנחיות המשך להפחתת הסיכון לתופעות לוואי במתן תוך שרירי של תכשירים המכילים בנזתין בנזילפניצלין]. www.health.gov.il/newsandevents/recall_drugs/doclib1/benzathine_benzylpenicillin.pdf (accessed August 2015).
Jeng 2010
- Jeng A, Beheshti M, Li J, Nathan R. The role of beta‐hemolytic streptococci in causing diffuse, nonculturable cellulitis: a prospective investigation. Medicine (Baltimore) 2010;89(4):217‐26. [PUBMED: 20616661] [DOI] [PubMed] [Google Scholar]
Jorup‐Rönström 1986
- Jorup‐Rönström C. Epidemiological, bacteriological and complicating features of erysipelas. Scandinavian Journal of Infectious Diseases 1986;18(6):519‐24. [PUBMED: 3810046] [DOI] [PubMed] [Google Scholar]
Jorup‐Rönström 1987
- Jorup‐Rönström C, Britton S. Recurrent erysipelas: predisposing factors and costs of prophylaxis. Infection 1987;15(2):105‐6. [PUBMED: 3110071] [DOI] [PubMed] [Google Scholar]
Jüni 2001
- Jüni P, Altman DG, Egger M. Systematic reviews in health care: Assessing the quality of controlled clinical trials. BMJ 2001;323(7303):42‐6. [PUBMED: 11440947 ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Karimkhani 2014a
- Karimkhani C, Boyers LN, Margolis DJ, Naghavi M, Hay RJ, Williams HC, et al. Comparing cutaneous research funded by the National Institute of Arthritis and Musculoskeletal and Skin Diseases with 2010 Global Burden of Disease results. PLoS One 2014;9(7):e102122. [PUBMED: 25003335] [DOI] [PMC free article] [PubMed] [Google Scholar]
Karimkhani 2014b
- Karimkhani C, Boyers LN, Prescott L, Welch V, Delamere FM, Nasser M, et al. Global burden of skin disease as reflected in Cochrane Database of Systematic Reviews. JAMA Dermatology 2014;150(9):945. [PUBMED: 24807687] [DOI] [PubMed] [Google Scholar]
Karppelin 2010
- Karppelin M, Siljander T, Vuopio‐Varkila J, Kere J, Huhtala H, Vuento R, et al. Factors predisposing to acute and recurrent bacterial non‐necrotizing cellulitis in hospitalized patients: a prospective case‐control study. Clinical Microbiology & Infection 2010;16(6):729‐34. [PUBMED: 19694769] [DOI] [PubMed] [Google Scholar]
Karppelin 2013
- Karppelin M, Siljander T, Huhtala H, Aroma A, Vuopio J, Hannula‐Jouppi K, et al. Recurrent cellulitis with benzathine penicillin prophylaxis associated with diabetes and psoriasis. European Journal of Clinical Microbiology & Infectious Diseases 2013;32(3):369‐72. [PUBMED: 23007460] [DOI] [PubMed] [Google Scholar]
Karppelin 2014
- Karppelin M, Siljander T, Aittoniemi J, Hurme M, Huttunen R, Huhtala H, et al. Predictors of recurrent cellulitis in five years. Clinical risk factors and the role of PTX3 and CRP. Journal of Infection 2015;70(5):467‐73. [DOI: 10.1016/j.jinf.2014.11.002; PUBMED: 25447714] [DOI] [PubMed] [Google Scholar]
Kasseroller 1996
- Kasseroller R. Erysipelprophylaxe beim sekundären Lymphödem mit Selen. Der Allgemeinartzt 1996;3:244‐7. [Google Scholar]
Kilburn 2010
- Kilburn SA, Featherstone P, Higgins B, Brindle R. Interventions for cellulitis and erysipelas. Cochrane Database of Systematic Reviews 2010, Issue 6. [DOI: 10.1002/14651858.CD004299.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Ko 1998
- Ko DS, Lerner R, Klose G, Cosimi AB. Effective treatment of lymphedema of the extremities. Archives of Surgery 1998;133(4):452‐8. [PUBMED: 9565129] [DOI] [PubMed] [Google Scholar]
Kotloff 2004
- Kotloff KL, Corretti M, Palmer K, Campbell JD, Reddish MA, Hu MC, et al. Safety and immunogenicity of a recombinant multivalent group a streptococcal vaccine in healthy adults: phase 1 trial. JAMA 2004;292(6):709‐15. [PUBMED: 15304468] [DOI] [PubMed] [Google Scholar]
Koutkia 1999
- Koutkia P, Mylonakis E, Boyce J. Cellulitis: evaluation of possible predisposing factors in hospitalized patients. Diagnostic Microbiology & Infectious Disease 1999;34(4):325‐7. [PUBMED: 10459485] [DOI] [PubMed] [Google Scholar]
Kouyos 2014
- Kouyos RD, Metcalf CJ, Birger R, Klein EY, Abel zur Wiesch P, Ankomah P, et al. The path of least resistance: aggressive or moderate treatment?. Proceedings. Biological Sciences / the Royal Society 2014;281(1794):1‐8. [PUBMED: 25253451] [DOI] [PMC free article] [PubMed] [Google Scholar]
Kunkel 2015
- Kunkel A, Colijn C, Lipsitch M, Cohen T. How could preventive therapy affect the prevalence of drug resistance? Causes and consequences. Philosopical Transactions of the Royal Society B 2015;370(1670):1‐9. [DOI: 10.1098/rstb.2014.0306; PUBMED: 25918446] [DOI] [PMC free article] [PubMed] [Google Scholar]
Lazzarini 2005
- Lazzarini L, Conti E, Tositti G, Lalla F. Erysipelas and cellulitis: clinical and microbiological spectrum in an Italian tertiary care hospital. Journal of Infection 2005;51(5):383‐9. [PUBMED: 16321649] [DOI] [PubMed] [Google Scholar]
Levell 2011
- Levell NJ, Wingfield CG, Garioch JJ. Severe lower limb cellulitis is best diagnosed by dermatologists and managed with shared care between primary and secondary care. British Journal of Dermatology 2011;164(6):1326‐8. [PUBMED: 21564054] [DOI] [PubMed] [Google Scholar]
Lewis 2006
- Lewis SD, Peter GS, Gomez‐Marin O, Bisno AL. Risk factors for recurrent lower extremity cellulitis in a U.S. Veterans Medical Center population. American Journal of the Medical Sciences 2006;332(6):304‐7. [PUBMED: 17170620] [DOI] [PubMed] [Google Scholar]
Lum 2002
- Lum GR. Cellulitis among active duty service members, US Armed Forces, 1998‐2001. Medical Surveillance Monthly Report (MSMR) 2002;8:6‐9. [Google Scholar]
Mallon 1997
- Mallon E, Powell S, Mortimer P, Ryan TJ. Evidence for altered cell‐mediated immunity in postmastectomy lymphoedema. British Journal of Dermatology 1997;137(6):928‐33. [PUBMED: 9470909] [PubMed] [Google Scholar]
Mandell 2010
- Mandell GL, Bennett JE, Dolin R. Mandell, Douglas, and Bennett's Principles and Practice of Infectious Diseases. 7. Vol. 1, Philadelphia: Churchill Livingstone, 2010. [Google Scholar]
Mason 2014
- Mason JM, Thomas KS, Crook AM, Foster KA, Chalmers JR, Nunn AJ, et al. Prophylactic antibiotics to prevent cellulitis of the leg: economic analysis of the PATCH I & II Trials. PLoS One 2014;9(2):e82694. [DOI: 10.1371/journal.pone.0082694; PUBMED: 24551029] [DOI] [PMC free article] [PubMed] [Google Scholar]
Massell 1969
- Massell BF, Honikman LH, Amezcua J. Rheumatic fever following streptococcal vaccination. Report of three cases. JAMA 1969;207(6):1115‐9. [PUBMED: 5818242 ] [PubMed] [Google Scholar]
McNamara 2007
- McNamara DR, Tleyjeh IM, Berbari EF, Lahr BD, Martinez J, Mirzoyev SA, et al. A predictive model of recurrent lower extremity cellulitis in a population‐based cohort. Archives of Internal Medicine 2007;167(7):709‐15. [PUBMED: 17420430] [DOI] [PubMed] [Google Scholar]
McNeil 2005
- McNeil SA, Halperin SA, Langley JM, Smith B, Warren A, Sharratt GP, et al. Safety and immunogenicity of 26‐valent group a streptococcus vaccine in healthy adult volunteers. Clinical Infectious Diseases 2005;41(8):1114‐22. [PUBMED: 16163629] [DOI] [PubMed] [Google Scholar]
Mertz 1998
- Mertz KR, Baddour LM, Bell JL, Gwin JL. Breast cellulitis following breast conservation therapy: a novel complication of medical progress. Clinical Infectious Diseases 1998;26(2):481‐6. [PUBMED: 9502474] [DOI] [PubMed] [Google Scholar]
Miller 1998
- Miller SR, Mondry T, Reed JS, Findley A, Johnstone PA. Delayed cellulitis associated with conservative therapy for breast cancer. Journal of Surgical Oncology 1998;67(4):242‐5. [PUBMED: 9579371] [DOI] [PubMed] [Google Scholar]
Mokni 2006
- Mokni M, Dupuy A, Denguezli M, Dhaoui R, Bouassida S, Amri M, et al. Risk factors for erysipelas of the leg in Tunisia: a multicenter case‐control study. Dermatology 2006;212(2):108‐12. [PUBMED: 16484815] [DOI] [PubMed] [Google Scholar]
Moreland 2014
- Moreland NJ, Waddington CS, Williamson DA, Sriskandan S, Smeesters PR, Proft T, et al. Working towards a group A streptococcal vaccine: report of a collaborative Trans‐Tasman workshop. Vaccine 2014;32(30):3713‐20. [PUBMED: 24837510] [DOI] [PubMed] [Google Scholar]
Morris 2008
- Morris AD. Cellulitis and erysipelas. Clinical Evidence 2008;01(1708):1‐8. [PUBMED: 19450336] [PMC free article] [PubMed] [Google Scholar]
Mortimer 2014
- Mortimer PS, Rockson SG. New developments in clinical aspects of lymphatic disease. Journal of Clinical Investigation 2014;124(3):915‐21. [PUBMED: 24590276] [DOI] [PMC free article] [PubMed] [Google Scholar]
Nathwani 2001
- Nathwani D. The management of skin and soft tissue infections: outpatient parenteral antibiotic therapy in the United Kingdom. Chemotherapy 2001;47(Suppl 1):17‐23. [PUBMED: 11096185] [DOI] [PubMed] [Google Scholar]
NICE 2005
- National Institute for Health and Care Excellence (NICE). Cellulitis ‐ acute 2005. //cks.nice.org.uk/cellulitis‐acute#!topicsummary (accessed March 2015).
Oh 2014
- Oh CC, Ko HC, Lee HY, Safdar N, Maki DG, Chlebicki MP. Antibiotic prophylaxis for preventing recurrent cellulitis: A systematic review and meta‐analysis. Journal of Infection 2014;69(1):26‐34. [DOI: 10.1016/j.jinf.2014.02.011; PUBMED: 24576824] [DOI] [PubMed] [Google Scholar]
Ostermann 2014
- Ostermann H, Blasi F, Medina J, Pascual E, McBride K, Garau J, REACH study group. Resource use in patients hospitalized with complicated skin and soft tissue infections in Europe and analysis of vulnerable groups: the REACH study. Journal of Medical Economics 2014;17(10):719‐29. [PUBMED: 24983206] [DOI] [PubMed] [Google Scholar]
Parmar 1998
- Parmar MK, Torri V, Stewart L. Extracting summary statistics to perform meta‐analyses of the published literature for survival endpoints. Statistics in Medicine 1998;17(24):2815‐34. [PUBMED: 9921604] [DOI] [PubMed] [Google Scholar]
Pauszek 1991
- Pauszek ME. Prophylaxis for recurrent cellulitis complicating venous and lymphatic insufficiency. Indiana Medicine 1991;84(4):252‐3. [PUBMED: 2037773] [PubMed] [Google Scholar]
Pavlotsky 2004
- Pavlotsky F, Amrani S, Trau H. Recurrent erysipelas: risk factors. Journal Der Deutschen Dermatologischen Gesellschaft 2004;2(2):89‐95. [PUBMED: 16279242] [DOI] [PubMed] [Google Scholar]
Pierce 1992
- Pierce RP, Daugird AJ. Recurrent leg cellulitis: pathogenesis, treatment, and prevention. Journal of the American Board of Family Medicine 1992;5(1):85‐7. [PUBMED: 1561927] [PubMed] [Google Scholar]
Quirke 2016
Raff 2016
- Raff AB, Kroshinsky D. Cellulitis: a review. JAMA 2016;316(3):325‐37. [PUBMED: 27434444] [DOI] [PubMed] [Google Scholar]
Read 2011
- Read AF, Day T, Huijben S. The evolution of drug resistance and the curious orthodoxy of aggressive chemotherapy. Proceedings of the National Academy of Sciences of the United States of America 2011;108(Suppl 2):10871‐7. [PUBMED: 21690376 ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Roldan 2000
- Roldan YB, Mata‐Essayag S, Hartung C. Erysipelas and tinea pedis. Mycoses 2000;43(5):181‐3. [PUBMED: 10948816] [DOI] [PubMed] [Google Scholar]
Roujeau 2004
- Roujeau JC, Sigurgeirsson B, Korting HC, Kerl H, Paul C. Chronic dermatomycoses of the foot as risk factors for acute bacterial cellulitis of the leg: a case‐control study. Dermatology 2004;209(4):301‐7. [PUBMED: 15539893] [DOI] [PubMed] [Google Scholar]
Sawai 2007
- Sawai J, Hasegawa T, Kamimura T, Okamoto A, Ohmori D, Nosaka N, et al. Growth phase‐dependent effect of clindamycin on production of exoproteins by Streptococcus pyogenes. Antimicrobial Agents & Chemotherapy 2007;51(2):461‐7. [DOI: 10.1128/AAC.00539-06; PUBMED: 17101685] [DOI] [PMC free article] [PubMed] [Google Scholar]
Schünemann 2013
- Schünemann H, Brożek J, Guyatt G, Oxman A. GRADE handbook for grading quality of evidence and strength of recommendation. The GRADE Working Group, 2013. Available from:http://gdt.guidelinedevelopment.org/app/handbook/handbook.html. [Google Scholar]
Semel 1996
- Semel JD, Goldin H. Association of athlete's foot with cellulitis of the lower extremities: diagnostic value of bacterial cultures of ipsilateral interdigital space samples. Clinical Infectious Diseases 1996;23(5):1162‐4. [PUBMED: 8922818] [DOI] [PubMed] [Google Scholar]
SFD 2000
- French Society of Dermatology [Société Française de Dermatologie]. Management of erysipelas and necrotizing fasciitis [Erysipèle et fasciite nécrosante:prise en charge]. Annales de Dermatologie et de Vénéréologie 2000;127(12):1118‐37. [PUBMED: 11173699] [PubMed] [Google Scholar]
Shenoy 1999
- Shenoy RK, Kumaraswami V, Suma TK, Rajan K, Radhakuttyamma G. A double‐blind, placebo‐controlled study of the efficacy of oral penicillin, diethylcarbamazine or local treatment of the affected limb in preventing acute adenolymphangitis in lymphoedema caused by brugian filariasis. Annals of Tropical Medicine & Parasitology 1999;93(4):367‐77. [PUBMED: 10656038] [DOI] [PubMed] [Google Scholar]
Siljander 2008
- Siljander T, Karppelin M, Vahakuopus S, Syrjanen J, Toropainen M, Kere J, et al. Acute bacterial, nonnecrotizing cellulitis in Finland: microbiological findings. Clinical Infectious Diseases 2008;46(6):855‐61. [PUBMED: 18260753] [DOI] [PubMed] [Google Scholar]
Simon 1992
- Simon MS, Cody RL. Cellulitis after axillary lymph node dissection for carcinoma of the breast. American Journal of Medicine 1992;93(5):543‐8. [PUBMED: 1364813] [DOI] [PubMed] [Google Scholar]
Soo 2008
- Soo JK, Bicanic TA, Heenan S, Mortimer PS. Lymphatic abnormalities demonstrated by lymphoscintigraphy after lower limb cellulitis. British Journal of Dermatology 2008;158(6):1350‐3. [PUBMED: 18241266] [DOI] [PubMed] [Google Scholar]
Stalbow 2004
- Stalbow J. Preventing cellulitis in older people with persistent lower limb oedema. British Journal of Nursing 2004;13(12):725‐32. [PUBMED: 15284636] [DOI] [PubMed] [Google Scholar]
Stevens 2005
- Stevens DL, Bisno AL, Chambers HF, Everett ED, Dellinger P, Goldstein EJ, et al. Practice guidelines for the diagnosis and management of skin and soft‐tissue infections. Clinical Infectious Diseases 2005;41(10):1373‐406. [PUBMED: 16231249] [DOI] [PubMed] [Google Scholar]
Stevens 2014
- Stevens DL, Bisno AL, Chambers HF, Dellinger EP, Goldstein EJ, Gorbach SL, et al. Practice guidelines for the diagnosis and management of skin and soft tissue infections: 2014 update by the Infectious Diseases Society of America. Clinical Infectious Diseases 2014;59(2):e10‐52. [DOI: 10.1093/cid/ciu444; PUBMED: 24973422] [DOI] [PubMed] [Google Scholar]
Stoberl 1987
- Stoberl C, Partsch H. Erysipelas and lymphedema‐‐egg or hen?. Zeitschrift für Hautkrankheiten 1987;62(1):56‐62. [PUBMED: 3577282] [PubMed] [Google Scholar]
Strazzula 2015
- Strazzula L, Cotliar J, Fox LP, Hughey L, Shinkai K, Gee SN, et al. Inpatient dermatology consultation aids diagnosis of cellulitis among hospitalized patients: A multi‐institutional analysis. Journal of the American Academy of Dermatology 2015;73(1):70‐5. [PUBMED: 26089048] [DOI] [PubMed] [Google Scholar]
Swartz 2004
- Swartz MN. Clinical practice. Cellulitis. New England Journal of Medicine 2004;350(9):904‐12. [PUBMED: 14985488] [DOI] [PubMed] [Google Scholar]
Szolnoky 2014
- Szolnoky G, Dobozy A, Kemény L. Towards an effective management of chronic lymphedema. Clinics in Dermatology 2014 Sep‐Oct;32(5):685‐91. [PUBMED: 25160111] [DOI] [PubMed] [Google Scholar]
Tay 2015
- Tay EY, Fook‐Chong S, Oh CC, Thirumoorthy T, Pang SM, Lee HY. Cellulitis Recurrence Score: A tool for predicting recurrence of lower limb cellulitis. Journal of the American Academy of Dermatology 2015;72(1):140‐5. [DOI: 10.1016/j.jaad.2014.08.043; PUBMED: 25443627] [DOI] [PubMed] [Google Scholar]
Thind 1985
- Thind P. Prophylactic antibiotics in recurrent erysipelas. Lancet 1985;1(8435):986. [PUBMED: 2859444] [DOI] [PubMed] [Google Scholar]
Thomas 2010
- Thomas KS, UK Dermatology Clinical Trials Network's PATCH study group. Studying a disease with no home‐‐lessons in trial recruitment from the PATCH II study. Trials 2010;11(2):1‐5. [DOI: 10.1186/1745-6215-11-22; PUBMED: 20196846] [DOI] [PMC free article] [PubMed] [Google Scholar]
Tierney 2007
- Tierney JF, Stewart LA, Ghersi D, Burdett S, Sydes MR. Practical methods for incorporating summary time‐to‐event data into meta‐analysis. Trials 2007;8:16. [PUBMED: 17555582] [DOI] [PMC free article] [PubMed] [Google Scholar]
Van Zuuren 2014
- Zuuren EJ, Fedorowicz Z, Alper B, Mitsuma SF. Penicillin to prevent recurrent leg cellulitis: a critical appraisal. British Journal of Dermatology 2014;171(6):1300‐3. [DOI: 10.1111/bjd.13461; PUBMED: 25523263] [DOI] [PubMed] [Google Scholar]
Vinh 2005
- Vinh DC, Embil JM. Rapidly progressive soft tissue infections. Lancet Infectious Diseases 2005;5(8):501‐13. [PUBMED: 16048719] [DOI] [PubMed] [Google Scholar]
Weng 2016
WHO 2000
- World Health Organization. Benzathine penicillin: three fatal reports following mega unit injections. Drug Information Bulletin 2000;4(2):1. [Google Scholar]
Woo 2000
- Woo PC, Lum PN, Wong SS, Cheng VC, Yuen KY. Cellulitis complicating lymphoedema. European Journal of Clinical Microbiology & Infectious Diseases 2000;19(4):294‐7. [PUBMED: 10834819] [DOI] [PubMed] [Google Scholar]
Yamamoto 2015
- Yamamoto T, Koshima I. Supermicrosurgical anastomosis of superficial lymphatic vessel to deep lymphatic vessel for a patient with cellulitis‐induced chronic localized leg lymphedema. Microsurgery 2015;35(1):68‐71. [PUBMED: 25197031] [DOI] [PubMed] [Google Scholar]
Young 1960
- Young JR, Dewolfe VG. Recurrent lymphangitis of the leg associated with dermatophytosis. Report of 25 consecutive cases. Cleveland Clinic quarterly 1960;27:19‐24. [PUBMED: 13846637] [DOI] [PubMed] [Google Scholar]
References to other published versions of this review
Dalal 2012
- Dalal A, Eskin‐Shwartz M, Mimouni D, Ray S, Days W, Hodak E, et al. Interventions for the prevention of recurrent erysipelas and cellulitis. Cochrane Database of Systematic Reviews 2012, Issue 4. [DOI: 10.1002/14651858.CD009758] [DOI] [PMC free article] [PubMed] [Google Scholar]


