Abstract
Here, we report a simple, yet effective surface modification approach to impart hydrophobic surfaces with super-hydrophilicity using ultra-low fouling/functionalizable carboxybetaine (CB) copolymers via a dip-coating technique. A new series of CB random copolymers with varying amphiphilicities were synthesized and coated on hydrophobic polypropylene (PP) and polystyrene (PS) surfaces. Nonfouling capability of each coating was screened by enzyme-linked immunosorbent assay (ELISA), and further comprehensively assessed against 100% human serum by Micro BCA protein assay kit. The random copolymer containing ~30 mol% of CB unit showed superhydrophilicity with the highest air contact angle of more than 165° in DI water and the best nonfouling capability against 100% human blood serum. Surfaces of a 96-well plate coated with the optimal CB random copolymer showed significantly better nonfouling capability than those of a commercial 96-well plate with an ultra-low attachment surface. Adhesion of mouse embryonic fibroblast cells (NIH3T3) was completely inhibited on surfaces coated with CB random copolymers. Furthermore, the optimal nonfouling CB copolymer surface was functionalized with an antigen via covalent bonding where its specific interactions with its antibody were verified. Thus, this CB random copolymer is capable of imparting both ultra-low fouling and functionalizable capabilities to hydrophobic surfaces for blood-contacting devices.
Keywords: zwitterionic materials, carboxybetaine, random copolymer, dip-coating, nonfouling, undiluted blood serum
INTRODUCTION
Biofouling on blood-contacting medical device surfaces remains to be of major concern for thrombosis. Protein adsorption is the primary event that occurs on the surface of biomaterials in contact with biological media. Non-specific protein adsorption triggers biofouling culminating in serious adverse biological responses such as platelet activation, thrombus formation, complement activation, and inflammatory reaction among various others. Thus, anti-thrombogenic blood- contacting devices strive to effectively avoid non-specific protein adsorption.1–5 This is created through an ultra-low fouling interface between blood components and the surface they are in contact with. Synthetic polymer biomaterials with hydrophilic and charge-neutral characters are promising due to their high biocompatibility.6–9 Conventional biomaterials, such as poly(2-hydroxyethyl methacrylate) poly(HEMA) and poly(ethylene glycol) (PEG) have been historically used as the gold standards for surface modification. However, poly(HEMA) has low water content and poor resistance to protein adsorption while PEG can induce adverse reactions, including the emergence of anti-PEG antibodies and PEG-induced tissue histologic changes.10–15
It is known that an indestructible hydration layer formed on the polymer chain surface can repel the adsorption of biomolecules with high efficacy. Thus, hydration induced nonfouling capability is a predominant feature for a hydrophilic nonfouling polymer biomaterial.7, 16–19 Zwitterionic compounds comprising of super-hydrophilic zwitterionic groups, particularly phosphorylcholine (PC), carboxybetaine (CB) and sulfobetaine (SB), have become popular as blood-inert biomaterials over the past two decades. The zwitterionic groups, which are superhydrophilic and charge-neutral with an inner salt structure, can form a layer of strongly bound water molecules that cannot be displaced by bioactive species, thus inhibiting non-specific protein adsorption. Recently, CB-based polymers show excellent hydration-induced nonfouling capability for various applications, making them very attractive for blood-contacting surface use.20–24 Furthermore, CB groups have the unique capability to covalently immobilize biomolecules, such as proteins, enzymes and oligonucleartide, to their carboxyl groups.25–27
Surface modification using zwitterionic polymers can be achieved either through “grafting from” or “grafting to” methods. To achieve noninvasive surface coatings for massive molded medical devices with various shapes and sizes, “Grafting to” polymers are shown to be far more robust by simply applying various polymers with different architectures, such as homopolymer, random copolymer, and di-/tri-/multi-block copolymer, to various surfaces via silane, catechol, and photo-/heat-induced covalent bonding.28–33 Among them, random-type amphiphilic zwitterionic copolymers are a simple, yet effective nonfouling material that can effectively resist protein adsorption and platelet adhesion for a long term in blood-contacting surfaces. Poly(MPC- co-n-butyl methacrylate (BMA)) with ~30 mol% of hydrophilic unit has been applied to many surfaces of medical devices with excellent biocompatibility.31,34–35 However, comprehensive investigations of CB random copolymers are still lacking up to now. Furthermore, their capability for the surface functionalization of terminal carboxyl groups makes them an attractive surface modification material.
Here, we report a simple, yet effective modification approach for hydrophobic materials using amphiphilic CB random copolymers via a dip-coating technique. By adjusting the composition of the hydrophilic and the hydrophobic units in this new series of amphiphilic copolymers, we explored the effect of polymeric amphiphilicity on nonfouling capability, and established the optimal compositions for surface coating. 100% human serum adsorption was measured on our CB random copolymer coated surfaces. Result was compared to those of a commercial 96-well plate with an “ultra-low attachment surface”.36 Furthermore, the CB copolymer surface was subsequently functionalized with anti-fibrinogen by covalent bonding between the carboxyl group within CB and the amino group within the antigen (Scheme 1). Thus, this CB polymer material and modification technique is promising for a wide range of applications, including nonfouling medical devices and medical diagnostics.
Scheme 1.
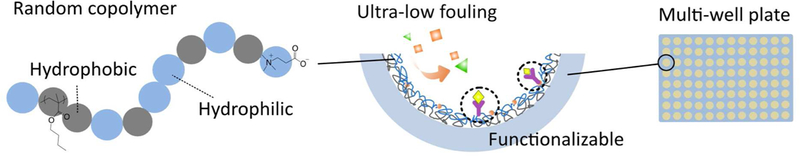
Schematic representation for the preparation of ultra-low fouling and functionalizable carboxybetaine (CB) polymer surfaces.
EXPERIMENTAL SECTION
Materials
Carboxybetaine acrylamide, 1-Carboxy-N,N-dimethyl-N-(3’-acrylamidopropyl) ethanaminium inner salt (CB1) and carboxybetaine methacrylate, 2-carboxy-N,N-dimethyl-N-(2’-methacryloyloxyethyl) ethanaminium inner salt (CB2), were synthesized according to a previously reported method, respectively.37 The following materials and reagents were obtained from Sigma-Aldrich (St. Louis, MO, USA) and used without any further purification: 2,2’-Azobisisobutyronitrile (AIBN), human plasma fibrinogen (Fg), human serum albumin (HSA), human blood f-globulin, sodium acetate (SA), N-[Tris(hydroxymethyl)methyl]-3-aminopropanesulfonic acid (TAPS), 1-decanethiol, sodium γ-dodecyl sulfate (SDS), N-hydroxysuccinimide (NHS), N-ethyl-N’-(3-diethylaminopropyl) carbodiimide hydrochloride (EDC). n-Butyl methacrylate (BMA) was purchased from Tokyo Chemical Industry Co., Ltd. (Portland, Oregon, USA). Anti-fibrinogen antibody conjugated with horseradish peroxidase (HRP) was purchased from Novus Biologicals (Littleton, CO, USA). O-phenylenediamine dihydrochloride (OPD) was obtained from Pierce (Rockford, Illinois, USA). Phosphate-buffered saline (10×, solution), hydrogen peroxide (H2O2, 30% in water) and hydrochloric acid (HCl) were obtained from Fisher Scientific Co. (Fair Lawn, NJ, USA). Normal human blood serum (pooled mixed gender) was purchased from BioChemed Services (Winchester, VA, USA). Micro BCA protein assay kit and RBS™ 35 Concentrate were purchased from Thermo Scientific (Waltham, MA, USA). The multi-well plate with an ultra-low attachment surface was purchased from Corning Costar Corp. (Corning, NY, USA). Ethanol (200 proof) was purchased from Decon Labs (King of Prussia, PA, USA). The water was obtained from a Millipore water purification system with a minimum resistivity of 18.0 MΩ cm. Other organic reagents and solvents were commercially available as extra-pure grade reagents and were used as received.
Synthesis of Polymers
Amphiphilic random copolymers, poly(CBl-co-BMA) (PCB1) and poly(CB2-co-BMA) (PCB2) were synthesized by conventional free radical polymerization method using AIBN as an initiator, and similar method was reported previously.38–39 In brief, desired amounts of CB monomer, BMA (various molar ratio of CB/BMA: 2/8, 3/7, 4/6, 5/5, 6/4, 8/2), and AIBN were dissolved in ethanol. The solution was transferred to a Pyrex® Vista™ glass tube reactor and further purged with nitrogen gas for 30 min at room temperature. Polymerization was performed in the sealed glass tube under a protection atmosphere of nitrogen gas. After polymerization, the reaction solution was slightly dropped into a mixed solvent of ether/chloroform to precipitate copolymers. The copolymer was filtered off and collected as a white powder after vacuum desiccation for 24 h at room temperature. The residual CB monomer was removed by washing the collected white polymer powder with a large amount of Millipore water. Then, the copolymer was filtered off again, frozen by liquid nitrogen and treated with a lyophilizer (Labconco Co., Ltd., Kansas City, Missouri, USA) at −80 °C for 48 h to convert them into dried white powder. The chemical structures of purified copolymers were confirmed using 1H-NMR (AV-500, Bruker, German) and polymers were cryopreserved under −20°C.
Optimization of Coating Conditions
Polypropylene (PP) substrates (ePlastic, San Diego, CA, USA) were cut into 0.5 cm × 0.5 cm, ultrasonically washed in ethanol for 10 min and dried at room temperature. CB copolymers with varying amphiphilicities were dissolved in ethanol with 0.50 wt% concentration, respectively. Each substrate was immersed in a polymer solution for 10s, followed by solvent evaporation under atmospheric pressure in an ethanol vapor-protective environment at room temperature. All modified PP substrates were soaked in phosphate-buffered saline (PBS, 1×, pH 7.4) for 1 h at room temperature. Afterwards, the substrate was rinsed with DI water and dried in a vacuum desiccation for 24 h at room temperature. To determine the effect of polymer concentration on coating efficiency, cleaned PP substrates were coated with CB copolymers at different concentrations (0.03, 0.06, 0.13, 0.25, 0.5 and 1.00 wt%) for further testing.
To quickly evaluate nonfouling capability of each coating, adsorbed single protein (fibrinogen) was measured by enzyme-linked immunosorbent assay (ELISA). In brief, polymer- coated PP substrates were pre-wetted in PBS overnight, then were immersed in 1.0 mg/mL fibrinogen in PBS for 1 h at 25 °C. After rinsing with fresh PBS, PP substrates were soaked into solution of anti-fibrinogen antibody conjugated with HRP at room temperature for 30 min. Then, substrates were rinsed again and allowed to react with OPD/H2O2 mixture solution for another 15 min; mixture solution contained 1.0 mg/ml OPD and 1000 times diluted H2O2 in citrate buffer (1×, pH 5.0). After the reaction was quenched with 1.0 N HCl, absorbance of each solution at 492 nm was measured using a microplate reader (BioTek Instruments Inc., Winooski, VT, USA).
Surface Coating on Multi-well Plates and Gold Chip
Here, 96-well plate made of original polystyrene was simply modified with CB copolymers using dip-coating solvent evaporation method mentioned above. The anti-fouling capability was compared to both commercial 96-well plate with an ultra-low attachment surface and uncoated polystyrene 96-well plate. The gold chips were made of a BK7 glass slide coated with a first titanium film layer (−2 nm) and a second gold layer (−48 nm) using an electron beam evaporator. The chip was ultrasonically washed with acetone, DI water, and ethanol for 5 min in each solvent. Subsequently, they were treated by UV/ozone cleaner for 30 min and immersed into 0.2 mM 1-decanethiol for 24 h to form a hydrophobic self-assembled monolayer. Finally, chips were dip-coated with CB polymers using same procedure above. The thickness of coated polymer layer was measured under dry conditions with a spectroscopic ellipsometer (α-SE; J. A. Woolam Co., Inc., Tokyo, Japan).31
Protein Adsorption from Single-protein Solution and 100% Human Serum
Here, the nonfouling capability of CB random copolymer coatings was comprehensively tested against both single proteins and whole human blood serum. Single blood protein at 10% concentration has been frequently used as a standard to assess biofouling on various surfaces. Thus, evaluation of nonfouling capability against 100% single blood protein and 100% human blood serum is attractive and essential for blood contacting devices. The coated 96-well plate was pre-wetted with DI water at room temperature before testing protein adsorption. Micro BCA protein assay kit was used to evaluate protein adsorption against human plasma fibrinogen (Fg), human serum albumin (HSA), human blood γ-globulin, and human serum. The concentration of Fg, HSA and γ-globulin are 3.0, 45, and 16 mg/mL respectively, which is equivalent to the 100 % of the concentration found in human plasma. Normal human blood serum (100 %, pooled mixed gender) was used as received. In brief, human protein (dissolved in PBS, 1×, pH 7.4) or undiluted human serum were incubated within pre-wetted wells at 37 °C for 2 h, and were rinsed with fresh PBS (1×, pH 7.4). Adsorbed proteins were detached in 1.0 ml of 1.0 wt% sodium n-dodecyl sulfate (SDS) solution. 150 μL of the liquid supernatant were transferred into 96-well plate and gently mixed with another 150 μL of bicinchoninic acid (BCA) reagents. After incubation at 37 °C for 2 h, a purple-colored product is generally formed by the chelation of two molecules of BCA with one cuprous ion (Cu+1 ), which is reduced from Cu+2 by protein in an alkaline environment. Finally, a microplate reader was used to determine the absorbance at 562 nm. The absorbance at 562 nm shows a linear with increasing amount of adsorbed proteins.
Cell Adhesion
NIH3T3 mouse embryonic fibroblast cells obtained from American Type Culture Collection (ATCC, Rockville, MD, USA) were seeded in a polystyrene tissue culture dish (Φ = 10 cm, 5.0 × 104 cells/mL) in dulbecco’s modified eagle medium (DMEM) supplemented with 10% fetal bovine serum (FBS) at 37 °C in a humidified atmosphere containing 5.0% CO2. Sub-confluent cell cultures were passaged using 0.25% trypsin/EDTA. Sterilized CB polymer solution (0.5 wt%, ethanol) was dropped onto the surface of tissue culture plate and evaporated in the cell culture hood. Then, the coated surface was rinsed with PBS (1×, pH 7.4) and pre-wetted with DMEM overnight at room temperature. NIH3T3 cells were seeded into the partially coated dish at a concentration of 5.0 × 104 cells/mL in DMEM supplemented with 10 % FBS at 37 °C in a humidified atmosphere containing 5.0% CO2. After 3 days’ incubation, the medium was replaced and morphology of the cells on the partially coated surface was observed using a Nikon Eclipse TE2000-U microscope (Nikon Instruments, Melville, NY).
Surface Functionalization
Surface functionalization was performed on the PCB2–37 coated 96-well polystyrene plate. The surface modification procedure has been descripted above in detail using a dip-coating and solvent evaporation method. The carboxyl group on the polymer surface can be easily activated by EDC/NHS chemistry, and covalently bond with amino groups of, for example, proteins, enzyme, and aptamer/oligonucleotides. In brief, first, 0.15 mL of a freshly prepared solution containing 0.1 M NHS and 0.4 M EDC in DI water was added into coated 96-well plate for 30 min at 25 °C to activate the carboxylate group. Second, the solution of EDC and NHS was removed and the surface of well was rinsed with 10 mM SA buffer (pH 5.0) three times. Third, 0.15 mL Fg solution (1.0 mg/mL) in 10 mM TAPS (pH 8.2) was added into the activated well and allowed to react for 30 min at 25°C. Subsequently, the functionalized surface was washed three times with BA buffer (10 mM boric acid and 300 mM sodium chloride, pH 9.0) and then phosphate buffered saline (PBS, 1×, pH 7.4) before evaluation of antibody-antigen specific interaction. Along with above protein immobilization process the residual activated carboxyl groups were also deactivated. The Fg-functionalized 96-well plate was gently rinsed with fresh PBS three times. Subsequently 0.15 mL of anti-fibrinogen antibody conjugated with HRP in PBS solution were added into each well for 30 min at 25 °C. After that, the wells were rinsed again and allowed to react with 0.15 mL of OPD/H2O2 solution for another 15 min. The chromogenic reaction was quenched through adding 0.15 mL 1.0 N of hydrochloride acid solution. The absorbance at 492 nm in each solution was measured using a microplate reader mentioned above. For comparison, a non-activated CB polymer coated 96-well plate yet contacted with Fg, and an activated CB polymer surface but functionalized with HSA were utilized as control.
Statistical Analysis
All graphs and bar charts are expressed as the mean ± standard deviation (SD) of three or five repeated experiments as described above. Student’s t-test was carried out to determine whether the observed differences were statistically significant.
RESULTS & DISCUSSION
Synthesis of Random Copolymers
Polyolefins have been widely manufactured for biomedical applications. However, these hydrophobic polymeric hydrocarbon-based materials will trigger non-specific protein adsorption, platelet activation, blood clotting, thrombogenesis and other biofouling-associated issues. For practical applications, it is highly desirable to use the simplest approach to achieve the set goals. Free radical polymerization method is one of the most common and useful approaches for making polymers from small-scale laboratory trial to large-scale industrial application, particularly for polymerization of vinyl monomers. Synthesis of vinyl monomers, CB1 and CB2 were reported previously. Amphiphilic random copolymers were synthesized through a conventional free radical polymerization method using AIBN as a thermal free radical initiator. Viscosity of these reaction solutions gradually increased along with the processing of polymerization at 65 °C, indicating the conversion of monomers to copolymers. From the integral values of characteristic peaks in 1H-NMR spectra (Figure S-1a and Figure S-1b), the unit fraction of each monomer was obtained: 3.82 ppm (-CH2-, 2H) for the CB1 unit, 2.42 ppm (- CH2-, 2H) for the CB2 unit, and 1.45–1.63 ppm (-CH2-, 4H) for the BMA unit. The chemical structures and synthetic details about these polymers are shown in Figure 1 and Table 1, respectively. Hydrophilic CB unit and hydrophobic BMA unit in the polymer chain were randomly distributed, with a total composition approximately equal to that of the monomer feed solutions. The solubility of CB random copolymers in an aqueous solution mainly depends on the CB unit composition. Copolymers containing more than 30 mol% of CB unit will be easily dissolved in an aqueous solution, while less than 30 mol% will become water-insoluble (Table 1). Thus, water-insoluble CB copolymer containing 30 mol% CB unit is the best coating material among these copolymers. These results match with other random copolymers containing phosphorylcholine group, which has been widely utilized for surface modification of biomedical devices.
Figure 1.
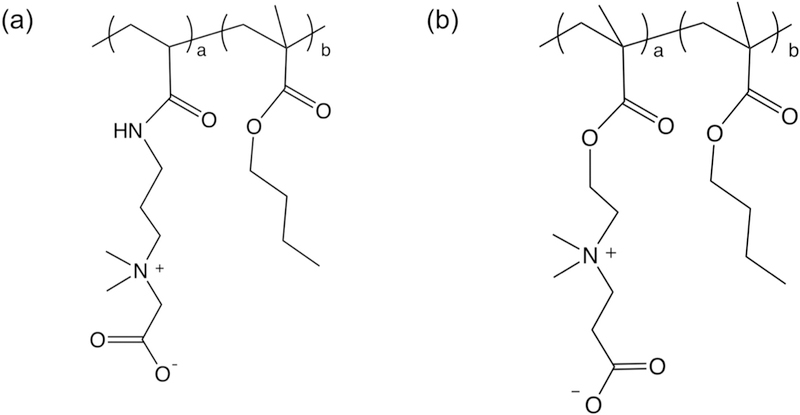
Chemical structures of synthetic carboxybetaine (CB) random copolymers. (a) poly(CB1-co-BMA) (PCB1), and (b) poly(CB2-co-BMA) (PCB2). 1 and 2 behind CB stand for number of carbon spacer between carboxyl groups and quaternary ammonium cations. The composition of each unit in the copolymer was calculated through integration of characteristic protons in 1H-NMR spectrum, where 3.82 ppm (-CH2-,2H) for the CB1 unit, 2.42 ppm (-CH2-,2H) for the CB2 unit, and 1.45–1.63 ppm (-CH2-,4H) for the BMA unit. The carboxyl groups in PCB2 can be activated by EDC/NHS chemistry, and covalently bond with amino groups of, for example, proteins, enzyme, and aptamer/oligonucleotides.
Table 1.
Characteristics of synthesized copolymers a
| monomer unit composition (mol %) |
molecular weightc |
solubility d |
|||||||
|---|---|---|---|---|---|---|---|---|---|
| in feed CB/BMA |
in copolymerb
CB/BMA |
initiator (mmol/L) |
polymerization time (h) |
yield (%) |
MW×104 | Mw/Mn | ethanol | water | |
| PCB1–28 | 20 / 80 | 17 / 83 | 5 | 16 | 80 | 7.4 | 2.6 | + + | – – |
| PCB1–37 | 30 / 70 | 31 / 69 | 5 | 16 | 87 | 8.3 | 2.4 | + + | – – |
| PCB1–46 | 40 / 60 | 38 / 62 | 5 | 16 | 88 | 6.7 | 2.8 | + + | + + |
| PCB1–55 | 50 / 50 | 50 / 50 | 5 | 16 | 78 | 7.1 | 2.3 | + + | + + |
| PCB1–64 | 60 / 40 | 57 / 43 | 5 | 16 | 80 | 7.5 | 2.7 | + + | + + |
| PCB1–82 | 80 / 20 | 82 / 18 | 5 | 16 | 85 | 6.9 | 2.5 | + + | + + |
| PCB2–37 | 30 / 70 | 27 / 73 | 5 | 16 | 86 | 7.9 | 2.8 | + + | – – |
[Monomer] = 0.5 mol/L; initiator for all the polymers was AIBN; polymerization temperature was 65 °C.
Determined by 1H NMR spectrum in CD3OD.
Molecular weights were determined by GPC in methanol/water = 7/3, [LiBr] = 10 mmol/L, and poly(ethylene oxide) standards. Mw and Mn represent weight-average molecular weight and number-average molecular weight, respectively.
Solubility was determined with 10 mg/mL each polymer sample and described as soluble (+) or insoluble (–) at 25 °C. PCB1 denotes poly(CBl-co-BMA) while PCB2 poly(CB2-co-BMA). Number in PCB1 and PCB2 stands for the molar ratio of two units, e.g., 28 in PCB1–28 indicates that the molar ratio of CB/BMA in this copolymer is 2/8.
Air Contact Angles and Screening of Coatings for Protein Adsorption
Amphiphilic CB copolymers with different hydrophobic/hydrophilic compositions can impart different nonfouling capabilities. Hydrophilic (CB) unit can promote ionic-induced hydration that can bind water molecules strongly to enhance nonfouling capability. However, strong hydration force may cause the detachment of coating materials from the surface. Hydrophobic (BMA) unit will bind the hydrophobic substrate surface strongly, to stabilize the CB unit on the surface in an aqueous solution and increase coating durability. Thus, it is necessary to prepare these coating random polymers with an appropriate molar ratio of CB/BMA so as to maximize both nonfouling and surface-binding properties. Results of air contact angle in Figure 2 showed that the copolymer containing ~30 mol% of CB unit exhibits superhydrophilicity with an air contact angle up to 165°. Its superhydrophilicity is related to its excellent performance as shown in Figure 3a, where the random polymer with ~30 mol% of CB unit has the strongest protein repelling capability. Other polymers with higher CB compositions are easily detached from PP surfaces due to their high solubility in an aqueous solution. Although PCB1–28 and PCB1–37 are both water-insoluble, higher CB compositions can increase their repelling capability against non-specific protein adsorption. In addition, the concentration of the polymer solution is another essential factor that affects the coated amount of zwitterionic polymer and thus nonfouling capability. As shown in Figure 3b, the absorbed amount of Fg decreased dramatically along with increasing of polymer concentration from 0.03 to 0.5 wt%, while above 0.5 wt% fouling reached a saturated relative low level that less than 15% compared to uncoated substrate.
Figure 2.
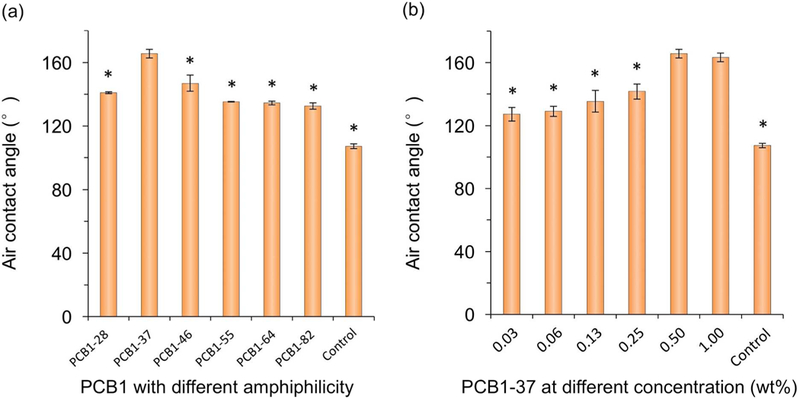
Air contact angle measured in distilled water on polypropylene (PP) substrates modified with (a) different CB random copolymers, and (b) PCB1–37 at different concentrations. Number stands for the molar ratio of two units in copolymers, e.g., 28 in PCB1–28 indicates that the molar ratio of CB/BMA in this copolymer is 2/8. Single asterisk (*) in (a) indicates statistically significant difference (p < 0.05, n = 5) compared to PCB1–37; and single asterisk (*) in (b) indicates statistically significant difference (p < 0.05, n = 5) compared to 0.50 wt%.
Figure 3.
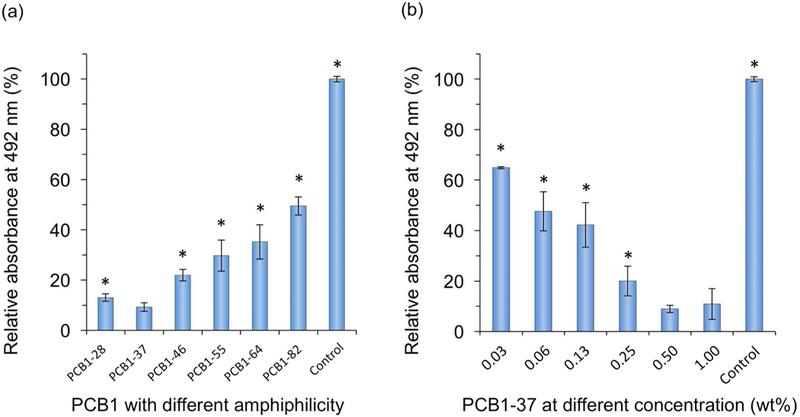
Relative adsorbed amount of fibrinogen (1.0 mg/mL, 1× PBS, pH 7.4) on a PP surface coated with (a) different CB random copolymers, and (b) PCB1–37 at different concentrations. Number stands for the molar ratio of two units in copolymers, e.g., 28 in PCB1–28 indicates that the molar ratio of CB/BMA in this copolymer is 2/8. Single asterisk (*) in (a) indicates statistically significant difference (p < 0.05, n = 5) compared to PCB1–37; and single asterisk (*) in (b) indicates statistically significant difference (p < 0.05, n = 5) compared to 0.50 wt%. Relative absorbance at 492 nm indicates the relative adsorbed amount of fibrinogen.
In addition, both PCB1–37 and PCB2–37 polymers showed similar nonfouling properties (Figure S-2). Importantly, the unique dual functionalities of nonfouling and surface ligand immobilization for CB polymers depend on two aspects: 1) the spacer groups between the positive quaternary amine group and the negative carboxyl group; 2) environmental factors (e.g., ionic strengths and pH values). The nonfouling capability of CB polymers tend to decrease for CB groups with a spacer longer than two carbon atoms in a solution, particularly with a lower ionic strength and a lower pH value, due to the additional hydrophobic spacer and anti-polyelectrolyte behavior of zwitterionic polymers.37 There is no significant difference in nonfouling capabilities between PCB1–37 and PCB2–37 under the same condition.
The stability of CB polymer (~30 mol% of CB unit) coating layer was confirmed under dry condition by comparing the polymer thickness of CB coated gold chips before and after soaking into PBS (1×, pH 7.4), using a spectroscopic ellipsometer. The thicknesses of modified CB polymer layers on gold substrates were calculated as 29.38±1.10 nm before soaking. This value did not significantly change after the substrates was soaked into PBS for two months when film thickness remained to be 28.80±0.73 nm.
Protein Adsorption from 100% Blood Proteins and Serums
The interactions between blood components and biomaterials may trigger a series of subsequent complex biological responses including protein adsorption, platelet adhesion/activation, blood coagulation and thrombosis. Rapid non-specific adsorption of plasma proteins has been considered as the first event occurring on biomaterial surfaces during blood/material interactions. Thus, it is crucial to evaluate the adsorption of main plasma proteins (e.g., fibrinogen, albumin, and γ-globulin) to understand the blood compatibility of biomaterials. Importantly, 10% diluted single-protein solutions are frequently utilized for this evaluation, but this testing condition is still far from the actual blood environment. In this work, both 100% single blood proteins and 100% human serums were used. Non-specific protein adsorption amount was tested by Micro BCA protein assay kit.
In Figure 4, for CB polymer-coated surfaces, the adsorbed amount of different types of proteins and undiluted serum was significantly reduced compared to uncoated surfaces both at 10 % and 100 % concentrations, indicating that zwiterionic CB polymer coating layer can impart ultra-low fouling capability to hydrophobic hydrocarbon-based surfaces. Although the commercial 96-well plate with an ultra-low attachment surface can repel protein adsorption from single-protein solutions at lower (10%) concentrations, there is still approximately 50% protein adsorption when protein concentration increases to 100% (Figure 4a and 4b). Importantly, although the commercial surface can repel the adsorption of single blood proteins at 10% concentrations, they lost nonfouling capability completely after being soaked in either 100% human serum or 100% γ-globulin environment (Figure 4c and 4d).
Figure 4.
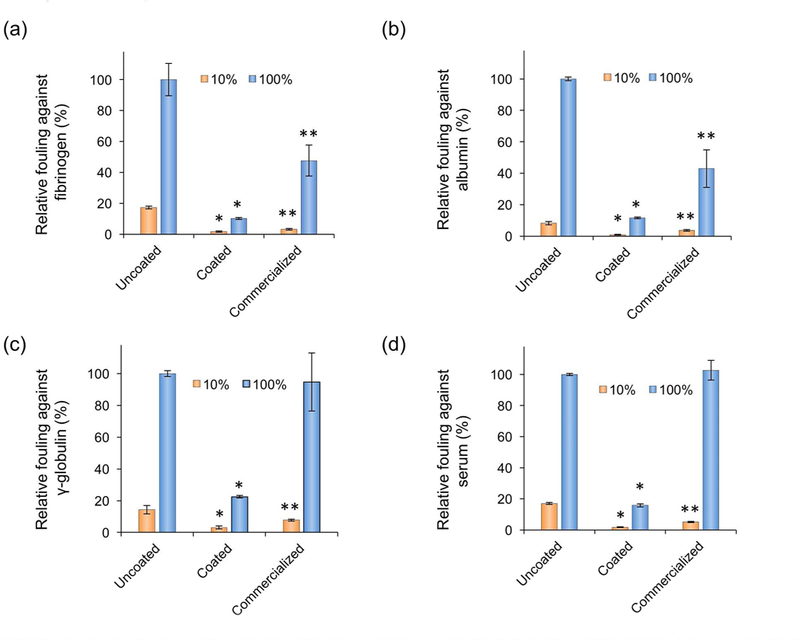
Non-specific protein adsorption against (a) human plasma fibrinogen (Fg), (b) human serum albumin (HSA), (c) human blood γ-globulin, and (d) human blood serum on 96-well plate with three kinds of surfaces: uncoated polystyrene surface, PCB1–37 coated polystyrene surface, and commercial plate with an ultra-low attachment surface (Corning Costar Corp., Corning, NY, USA). The corning ultra-low surface coating is a hydrophilic, neutrally charged coating covalently bound to the polystyrene surface. Both 10 % and 100 % of single blood proteins and human blood serums were used to evaluate their nonfouling capability. The uncoated surface and the commercial product with an ultra-low attachment surface were utilized as controls. Concentration of 100 % of Fg, HSA, and γ-globulin in blood are 3.0, 45, and 16 mg/mL, respectively. Normal human blood serum (pooled mixed gender) was used as received and without dilution. Single asterisk (*) indicates statistically significant difference (p < 0.01, n = 5) compared to either uncoated surface or commercialized ultra-low attachment surface; double asterisk (**) indicates statistically significant difference (p < 0.01, n = 5) compared to uncoated surface.
Cell adhesion
NIH3T3 cells are able to adhere, proliferate and migrate on a normal tissue culture polystyrene (TCPS) plate. After seeding them onto the surface of TCPS plate with a zwitterionic CB copolymer partially coated, cells will gradually adhere and spread out on TCPS surface. In contrast, most of the cells will remain spherical shape and cannot adhere on the CB copolymer surface. After being cultured for 72 h and slightly rinsed with fresh medium, no adhered cells were observed on the CB copolymer coated surface, while cells proliferated and reached sub-confluent on the normal TCPS surface with a very clear border (Figure 5). For fibroblast cells, adhesion is essential in maintaining multicellular structure and function, which is important for subsequent proliferation and migration. Fibronectin plays a major role in the adhesion of many cell types, and the adsorption of fibronectin will directly affect cell adhesion to substrate surfaces. Superhydrophilic zwitterionic polymer surfaces have excellent nonfouling capability against non-specific protein, including fibronectin.31, 40 Thus, fibroblast cell adhesion was completely prevented by the CB polymer coating layer.
Figure 5.
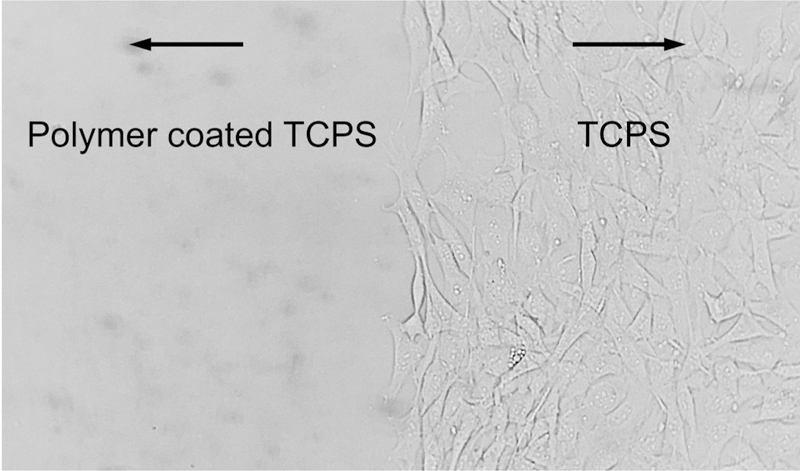
Adhesion and migration behavior of NIH3T3 mouse embryonic fibroblast cells on the CB polymer (PCB1–37) coated TCPS surface (left) and on the original TCPS surface (right).
Surface functionalization
To further evaluate the capability and versatility of the CB random copolymer-modified 96-well polystyrene plate for conjugating biomolecules, Fg and HSA were covalently immobilized via EDC/NHS coupling chemistry, respectively. The antibody detection was then conducted via enzyme chromogenic reaction. A typical antibody, anti-Fg conjugated with HRP was added into each well and ensured sufficient contact with surfaces. The degree of color reaction can indirectly show the amount of antibody detection. The non-activated CB surface will maintain nonfouling capability without any adhered biomolecules on the surface, leading to no subsequent antibody detection (Figure 6, Fg non-activated). For the surface covalently bonded with HSA yet contacted with anti-Fg, there is no specific antibody-antigen induced binding as well (Figure 6, HSA activated and HSA non-activated). The degree of surface functionalization could be adjusted by changing EDC/NHS concentration and the pH of the antibody conjugation buffer.41 Our previous study has shown that CB surfaces can still maintain nonfouling capability even being partially conjugated with biomolecules.42 Thus, this CB polymer modified 96-well plate is promising for the detection of biomolecules in complex media, including serum, plasma, and blood.
Figure 6.
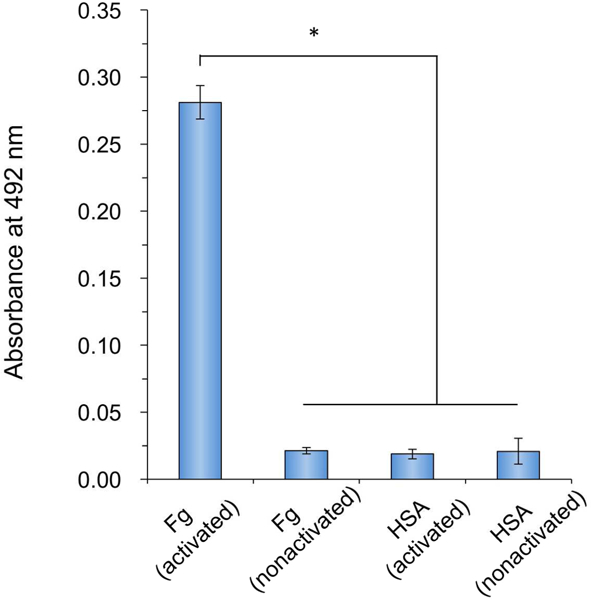
Surface functionalization and specific antibody-antigen interaction on the 96-well plate coated with the CB random copolymer, PCB2–37. Both Fg and HSA were conjugated to the CB polymer surface activated by EDC/NHS chemistry, respectively. Anti-Fg conjugated with HRP was utilized to evaluate specific binding, further to verify the feasibility for diagnosis using 96-well plates. Absorbance at 492 nm indirectly represents the amount of conjugated anti-Fg on the surface. The non-activated surface and the surface conjugated with HSA were utilized as controls. Single asterisk (*) indicates statistically significant difference (p < 0.001, n = 5).
CONCLUSIONS
In summary, a multifunctional random-type amphiphilic zwitterionic copolymer consisting of both super-hydrophilic carboxybetaine (CB) units and hydrophobic n-butyl methacrylate (BMA) units were successfully synthesized for use as a surface coating material. A CB random copolymer modified 96-well plate shows significantly better nonfouling property than the commercial 96-well plate with an ultra-low attachment surface. Furthermore, the specific detection of an antibody after surface functionalization with its antigen on a 96-well plate modified with this CB random copolymer was demonstrated. The novelty of this dual functional nonfouling and functionalizable surface coating also lies within its simple, yet effective for large-scale applications via a dip-coating method. Its utility is further verified through its ability to achieve noninvasive coating and long-term duration. Thus, this technique is promising for a wide range of medical and engineering applications.
Supplementary Material
ACKNOWLEDGMENTS
This work is supported by the National Institute of Health (R01HL089043) and the Center for Dialysis Innovation at the University of Washington.
Footnotes
Supporting Information
1H-NMR spectrum of (a) poly(CBl-co-BMA) (PCB1) and (b) poly(CB2-co-BMA) (PCB2). Relative adsorption of fibrinogen on PP surfaces coated with (a) PCB1–37 and (b) PCB2–37.
Notes
The authors declare the following competing financial 504 interest(s): S.J. is a cofounder of Taproot Medical Technologies LLC.
REFERENCES
- 1.Hoffman AS, Blood-biomaterial interactions: An overview. Biomaterials: Interfacial Phenomena and Applications, American Chemical Society, 1982, 199, 3–8. [Google Scholar]
- 2.Castner DG; Ratner BD, Biomedical surface science: Foundations to frontiers. SurSc. 2002, 500, 28–60. [Google Scholar]
- 3.Ratner BD; Bryant SJ, Biomaterials: where we have been and where we are going. Annu. Rev. Biomed. Eng. 2004, 6, 41–75. [DOI] [PubMed] [Google Scholar]
- 4.Williams DF, On the mechanisms of biocompatibility. Biomaterials 2008, 29, 2941–2953. [DOI] [PubMed] [Google Scholar]
- 5.Chen H; Yuan L; Song W; Wu Z; Li D, Biocompatible polymer materials: Role of protein-surface interactions. Prog. Polym. Sci. 2008, 33, 1059–1087. [Google Scholar]
- 6.Dalsin JL; Messersmith PB, Bioinspired antifouling polymers. Mater. Today 2005, 8, 38–46. [Google Scholar]
- 7.Chen S; Li L; Zhao C; Zheng J, Surface hydration: Principles and applications toward low-fouling/nonfouling biomaterials. Polymer 2010, 51, 5283–5293. [Google Scholar]
- 8.Thevenot P; Hu W; Tang L, Surface chemistry influence implant biocompatibility. Curr. Top. Med. Chem. 2008, 8, 270–280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lin X; Ishihara K, Water-soluble polymers bearing phosphorylcholine group and other zwitterionic groups for carrying DNA derivatives. J. Biomater. Sci., Polym. Ed. 2014, 25, 1461–1478. [DOI] [PubMed] [Google Scholar]
- 10.Tanaka M; Motomura T; Kawada M; Anzai T; Yuu K; Shiroya T; Shimura K; Onishi M; Akira M, Blood compatible aspects of poly(2-methoxyethylacrylate) (PMEA)-relationship between protein adsorption and platelet adhesion on PMEA surface. Biomaterials 2000, 21, 1471–1481. [DOI] [PubMed] [Google Scholar]
- 11.Keefe AJ; Jiang S, Poly(zwitterionic)protein conjugates offer increased stability without sacrificing binding affinity or bioactivity. Nat. Chem. 2012, 4, 59–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Li B; Yuan Z; Zhang P; Sinclair A; Jain P; Wu K; Tsao C; Xie J; Hung HC; Lin X; Bai T; Jiang S, Zwitterionic nanocages overcome the efficacy loss of biologic drugs. Adv. Mater. 2018, 30, 1705728. [DOI] [PubMed] [Google Scholar]
- 13.Li B; Xie J; Yuan Z; Jain P; Lin X; Wu K; Jiang S, Mitigation of inflammatory immune responses with hydrophilic nanoparticles. Angew. Chem. Int. Ed. 2018, 57, 4527–4531. [DOI] [PubMed] [Google Scholar]
- 14.Zhang P; Jain P; Tsao C; Yuan Z; Li W; Li B; Wu K; Hung H-C; Lin X; Jiang S, Polypeptide with high zwitterion density for safe and effective therapeutics. Angew. Chem. Int. Ed. 2018, D01: 10.1002/anie.201802452. [DOI] [PubMed] [Google Scholar]
- 15.Li B; Yuan Z; Hung H-C; Ma J; Jain P; Tsao C; Xie J; Zhang P; Lin X; Wu K; Jiang S, Revealing the immunogenic risk of polymers. Angew. Chem. Int. Ed. 2018, DOI: 10.1002/anie.201808615. [DOI] [PubMed] [Google Scholar]
- 16.Serrano Â; Sterner O; Mieszkin S; Zürcher S; Tosatti S; Callow ME; Callow JA; Spencer ND, Nonfouling response of hydrophilic uncharged polymers. Adv. Funct. Mater. 2013, 23, 5706–5718. [Google Scholar]
- 17.Jain P; Hung H-C; Lin X; Ma J; Zhang P; Sun F; Wu K; Jiang S, Poly(ectoine) hydrogels resist nonspecific protein adsorption. Langmuir 2017, 33, 11264–11269. [DOI] [PubMed] [Google Scholar]
- 18.Tanaka M; Sato K; Kitakami E; Kobayashi S; Hoshiba T; Fukushima K, Design of biocompatible and biodegradable polymers based on intermediate water concept. Polym. J. 2014, 47, 114. [Google Scholar]
- 19.Lin X; Nishio K; Konno T; Ishihara K, The effect of the encapsulation of bacteria in redox phospholipid polymer hydrogels on electron transfer efficiency in living cell-based devices. Biomaterials 2012, 33, 8221–8227. [DOI] [PubMed] [Google Scholar]
- 20.Ladd J; Zhang Z; Chen S; Hower JC; Jiang S, Zwitterionic polymers exhibiting high resistance to nonspecific protein adsorption from human serum and plasma. Biomacromolecules 2008, 9, 1357–1361. [DOI] [PubMed] [Google Scholar]
- 21.Hung H-C; Jain P; Zhang P; Sun F; Sinclair A; Bai T; Li B; Wu K; Tsao C; Liu EJ; Sundaram HS; Lin X; Farahani P; Fujihara T; Jiang S, A coating-free nonfouling polymeric elastomer. Adv. Mater. 2017, 29, 1700617. [DOI] [PubMed] [Google Scholar]
- 22.Shao Q; Jiang S, Molecular understanding and design of zwitterionic materials. Adv. Mater. 2015, 27, 15–26. [DOI] [PubMed] [Google Scholar]
- 23.Zhang L; Cao Z; Bai T; Carr L; Ella-Menye J-R; Irvin C; Ratner BD; Jiang S, Zwitterionic hydrogels implanted in mice resist the foreign-body reaction. Nat. Biotech. 2013, 31, 553–556. [DOI] [PubMed] [Google Scholar]
- 24.Jain P; Hung H-C; Li B; Ma J; Dong D; Lin X; Sinclair A; Zhang P; O’Kelly MB; Niu L; Jiang S, Zwitterionic hydrogels based on a degradable disulfide carboxybetaine cross-linker. Langmuir 2018, DOI: 10.1021/acs.langmuir.8b02100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sun F; Wu K; Hung H-C; Zhang P; Che X; Smith J; Lin X; Li B; Jain P; Yu Q; Jiang S, Paper sensor coated with a poly(carboxybetaine)-multiple DOPA conjugate via dip-coating for biosensing in complex media. Anal. Chem. 2017, 89, 10999–11004. [DOI] [PubMed] [Google Scholar]
- 26.Brault ND; White AD; Taylor AD; Yu Q; Jiang S, Directly functionalizable surface platform for protein arrays in undiluted human blood plasma. Anal. Chem. 2013, 85, 1447–1453. [DOI] [PubMed] [Google Scholar]
- 27.Jiang S; Cao Z, Ultralow-fouling, functionalizable, and hydrolyzable zwitterionic materials and their derivatives for biological applications. Adv. Mater. 2010, 22, 920–932. [DOI] [PubMed] [Google Scholar]
- 28.Lin X; Konno T; Takai M; Ishihara K, Redox phospholipid polymer microparticles as doubly functional polymer support for immobilization of enzyme oxidase. Colloids Surf. B. 2013, 102, 857–863. [DOI] [PubMed] [Google Scholar]
- 29.Chen H; Zou H; Paholak HJ; Ito M; Qian W; Che Y; Sun D, Thiol-reactive amphiphilic block copolymer for coating gold nanoparticles with neutral and functionable surfaces. Polym. Chem. 2014, 5, 2768–2773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Nagahashi K; Teramura Y; Takai M, Stable surface coating of silicone elastomer with phosphorylcholine and organosilane copolymer with cross-linking for repelling proteins. Colloids Surf. B. 2015, 134, 384–391. [DOI] [PubMed] [Google Scholar]
- 31.Lin X; Fukazawa K; Ishihara K, Photoreactive polymers bearing a zwitterionic phosphorylcholine group for surface modification of biomaterials. ACS Appl. Mater. Inter. 2015, 7, 17489–17498. [DOI] [PubMed] [Google Scholar]
- 32.Trel’ová D; Salgarella AR; Ricotti L; Giudetti G; Cutrone A; Šrámková P; Zahoranová A; Chorvát D; Haško D; Canale C; Micera S; Kronek J; Menciassi A; Lacík I, Soft hydrogel zwitterionic coatings minimize fibroblast and macrophage adhesion on polyimide substrates. Langmuir 2018, DOI: 10.1021/acs.langmuir.8b00765. [DOI] [PubMed] [Google Scholar]
- 33.Lin X; Konno T; Takai M; Ishihara K, Enzyme oxidase-immobilized phospholipid polymer microparticles for biofuel cell application. Trans. Mat. Res. Soc. Japan 2011, 36, 531–534. [Google Scholar]
- 34.Ishihara K; Oshida H; Endo Y; Ueda T; Watanabe A; Nakabayashi N, Hemocompatibility of human whole blood on polymers with a phospholipid polar group and its mechanism. J. Biomed. Mater. Res. 1992, 26, 1543–1552. [DOI] [PubMed] [Google Scholar]
- 35.Iwasaki Y; Ishihara K, Cell membrane-inspired phospholipid polymers for developing medical devices with excellent biointerfaces. Sci. Technol. Adv. Mat. 2012, 13, 064101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Eilenberger C; Rothbauer M; Ertl P; Küpcü S, A self-assembled antifouling nanobiointerface for the generation of spheroids. MethodsMol. Biol. 2018, 1771, 251–258. [DOI] [PubMed] [Google Scholar]
- 37.Zhang Z; Vaisocherová H; Cheng G; Yang W; Xue H; Jiang S, Nonfouling behavior of polycarboxybetaine-grafted surfaces: structural and environmental effects. Biomacromolecules 2008, 9, 2686–2692. [DOI] [PubMed] [Google Scholar]
- 38.Lin X; Konno T; Ishihara K, Cell-membrane-permeable and cytocompatible phospholipid polymer nanoprobes conjugated with molecular beacons. Biomacromolecules 2014, 15, 150–157. [DOI] [PubMed] [Google Scholar]
- 39.Lin X; Fukazawa K; Ishihara K, Photoinduced inhibition of DNA unwinding in vitro with water-soluble polymers containing both phosphorylcholine and photoreactive groups. Acta Biomater. 2016, 40, 226–234. [DOI] [PubMed] [Google Scholar]
- 40.Ishihara K; Ishikawa E; Iwasaki Y; Nakabayashi N, Inhibition of fibroblast cell adhesion on substrate by coating with 2-methacryloyloxyethyl phosphorylcholine polymers. J. Biomater. Sci. Polym. Ed. 1999, 10, 1047–1061. [DOI] [PubMed] [Google Scholar]
- 41.Yang W; Xue H; Li W; Zhang J; Jiang S, Pursuing “zero” protein adsorption of poly(carboxybetaine) from undiluted blood serum and plasma. Langmuir 2009, 25, 11911–11916. [DOI] [PubMed] [Google Scholar]
- 42.Vaisocherová H; Yang W; Zhang Z; Cao Z; Cheng G; Piliarik M; Homola J; Jiang S, Ultralow fouling and functionalizable surface chemistry based on a zwitterionic polymer enabling sensitive and specific protein detection in undiluted blood plasma. Anal. Chem. 2008, 80, 7894–7901. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


