Abstract
Melioidosis is a protean disease which is endemic to Southeast Asia and northern Australia. Here, we report a case of infected aortic aneurysm due to Burkholderia pseudomallei in an immunocompetent man 6 months after a trip to northern Malaysia. This patient initially received inappropriate surgical and antibiotic treatment, leading to a peri-prosthetic aortic infection with lumbar spondylitis and contiguous psoas muscle abscess. This case highlights the difficulty of diagnosing melioidosis given its diverse clinical manifestations and the limits of routine microbiological methods to identify B. pseudomallei. Melioidosis should be considered a possible diagnosis in individuals with unexplained fever subsequent to travel in an endemic area.
Keywords: cardiovascular system, infections, bone and joint infections, tropical medicine (infectious disease), urinary tract infections
Background
Melioidosis is a serious tropical bacterial infection due to Burkholderia pseudomallei, a Gram-negative bacillus discovered in 1912 by Captain Whitmore in Burma. This germ is endemic to Southeast Asia and northern Australia and is commonly found in soil and water. It has multiple clinical manifestations ranging from severe acute forms that include bacteraemia to localised subacute forms and latent forms. All the organs can potentially be affected.1 Here, we report a case of a B. pseudomallei infected aortic aneurysm, an extremely rare form of melioidosis.
Case presentation
A 57-year-old man was admitted to hospital for an unexplained fever in June 2017. His medical history was remarkable for high blood pressure and he was an active smoker. He had been through northern Malaysia 6 months previously and reported two superficial skin wounds: a scratch from a rock on the right leg and an urchin sting on the left foot. These two wounds healed normally after a few days. One week after returning to France, in December 2016, the patient presented with a high fever (40°C), dysuria and biological signs of inflammation (C reactive protein (CRP) 258 mg/L (n<5 mg/L)). Prostatitis was suspected though the bacteriological urine cultures were sterile. Blood cultures were negative. There was no imaging of the prostate. His condition improved following treatment with sulfamethoxazole/trimethoprim (800/160 mg two times per day) for 3 weeks. Over the two ensuing months, he progressively developed asthenia, anorexia and weight loss (10 kg) without fever. A bacteriological culture of urine was performed on 31 January 2017 and was considered contaminated (≥2 microorganisms). Two days later, he was admitted to hospital. The CRP was high again (160 mg/L) and a new bacteriological culture of urine was sterile, in the absence of any antibiotic treatment. Blood cultures were all negative. An abdominal CT-scan showed possible right pyelonephritis and a saccular infra-renal aortic aneurysm of 6 cm in diameter (figure 1). He was treated with levofloxacin (500 mg two times per day) for 8 days and amikacin (240 mg/day) for 2 days, and then cefixime (200 mg two times per day) and levofloxacin (500 mg two times per day) in association for the next 11 days, which resolved his clinical and biological symptoms. In March 2017, he underwent surgery for the aortic aneurysm with a bilateral subrenal aorto-iliac endoprosthesis. In June 2017, 3 months after surgery, he was admitted to the hospital for a relapse of fever (40°C) associated with significant asthenia and low back pain. The physical examination was unremarkable.
Figure 1.
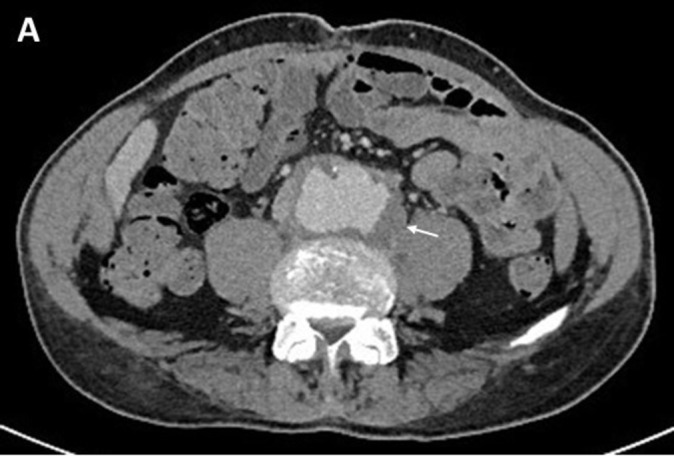
CT-scan showing a saccular infra-renal aortic aneurysm of 6 cm in diameter (white arrow).
Investigations
The biological tests showed raised inflammatory markers (CRP 240 mg/L, fibrinogen 6.2 g/L (N 2–4 g/L)) with a normal leucocyte count. Blood cultures were positive for a Gram-negative bacillus, which we were finally able to identify as B. pseudomallei with matrix-assisted laser desorption/ionisation-time of flight mass spectroscopy and use of a specific database of potential bioterrorism agents (Btyp2.0-sec-library1.0).
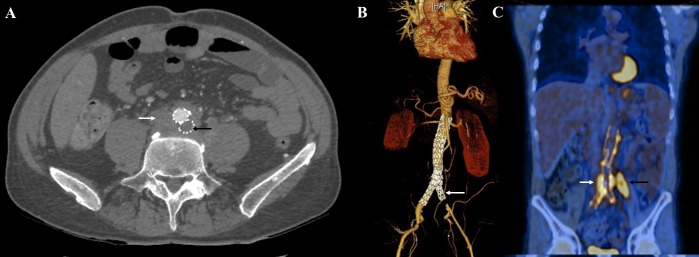
Abdominal CT-scan showed a septic thrombosis of the left common iliac portion of the endoprosthesis, a large peri-prosthetic infiltration, and multiple abscesses (left psoas muscle, aorto-iliac bifurcation and spinal paralumbar L4–L5) (figure 2A, B). Marked 18F-fluorodeoxyglucose (18F-FDG) uptake around the peri-prosthetic area and on left psoas muscle was found on a positron emission tomography (PET) scan (figure 2C). Lumbar MRI showed two prevertebral and paravertebral abscesses, between the endoprosthesis and the spine, with contiguous L4–L5 spondylitis. Prostate MRI and trans-oesophageal echocardiography were unremarkable.
Figure 2.
Abdominal CT-scan showing septic thrombosis of the left common iliac portion of the endoprosthesis (black arrow), and a large peri-prosthetic infiltration (white arrow) (A); three-dimensional reconstruction abdominal CT-scan, showing septic thrombosis of the left common iliac portion of the endoprosthesis (white arrow) (B) and positron emission tomography scan showing a marked aortic peri-prosthetic 18F-fluorodeoxyglucose uptake (white arrow) and a left psoas muscle collection (black arrow) (C).
The final diagnosis was bacteremic vascular melioidosis with peri-prosthetic infection, left psoas abscess and an L4–L5 spondylitis.
Treatment
The patient was treated with high doses of intravenous ceftazidime (2 g/6 hour). Apyrexia was obtained within 2 days and regression of the inflammatory biomarkers within 4 weeks. The first negative blood cultures were obtained after 1 week of treatment. He was treated with intravenous heparin for curative anticoagulation. After 4 weeks of antibiotic treatment, it was decided to remove the infected endoprosthesis. First, the abdominal aorta and the common right and left iliac arteries were resected. The endoprosthesis was removed and the aorto-iliac bifurcation was reconstructed using bilateral superficial femoral veins (figure 3). Despite the 4 weeks of antibiotic treatment, samples taken from the endoprosthesis were positive for B. pseudomallei.
Figure 3.
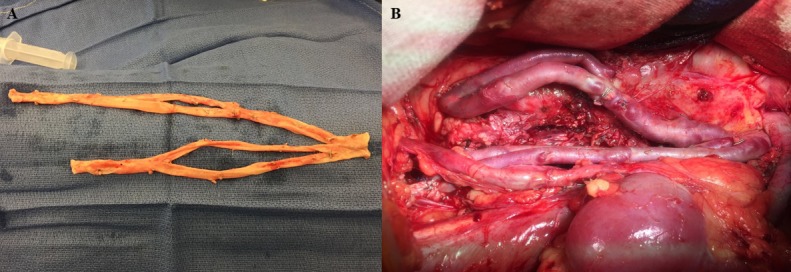
Superficial femoral veins were collected (A) and used for reconstruction of the aorto-iliac intersection (B).
There were no immediate complications. Abdominal CT angiography showed satisfactory permeability of the arterial axis. The treatment with intravenous ceftazidime was continued for 3 weeks after surgery, for a total of 7 weeks. Oral sulfamethoxazole/trimethoprim (800/160 mg, two tablets two times per day) was prescribed for an additional 6 months, in association with folinic acid (5 mg two times per day).
Outcome and follow-up
The clinical, biological and radiological outcomes were good, and there was no relapse 12 months after surgery. Two months after the end of the antibiotic treatment, the patient was called in for a PET scan to confirm the lack of 18F-FDG uptake around the abdominal peri-aortic, lumbar spine and left psoas muscle areas.
Discussion
Melioidosis is an emerging infectious disease that is caused by B. pseudomallei, a Gram-negative environmental bacterium endemic to tropical areas in Southeast Asia and northern Australia.1 Given its diverse clinical manifestations, and because this organism is often misidentified by methods routinely used in clinical laboratories, incidence is underestimated worldwide.1 Melioidosis has been described as ‘the great imitator’ because it is a protean disease that can vary from localised cutaneous forms at the bacterial entry site with no systemic manifestations, to septic shock with multi-organ failure. A large Australian cohort study found that most presentations were acute (85% of the 540 cases) and life-threatening (14% of mortality attributable to melioidosis).2 A majority of the patients had underlying conditions (80%), especially diabetes mellitus (39%).2 The main acute forms were pneumonia (50%), and then genitourinary, skin and osteoarticular infection.1 2 A smaller portion of cases reported chronic forms, defined as symptoms present for over 2 months. Finally, B. pseudomallei has a potential for latent infection and may emerge years after exposure. Our patient first presented a probable prostatitis and then pyelonephritis found on CT imaging. Therefore, we cannot exclude the possibility of an acute urinary form of the disease, which was initially misdiagnosed, subsequently developing into bacteraemia and then a chronic form with infected aortic aneurysm. A percutaneous inoculation, the most common way to acquire melioidosis, is also possible. It should be noted that more than half of the cases described in the literature were bacteraemic.2 Infected aneurysm is a rare presentation of melioidosis, found in no more than 1% of the cases and associated with high morbidity, mortality and relapse.1 2 As for the other causes of infected aneurysms, the aorta is the most common location,3 but middle-size vessels like femoral or iliac3 4 or the coronary arteries5 can also be affected. Aortic aneurysm secondary to melioidosis is commonly revealed by abdominal and/or low back pain.6 Because the symptoms are non-specific, and the potential absence of fever as in our case, the infectious origin of the aneurysm may be overlooked. However, no bacteriological sampling was performed during the surgery in our patient, who unfortunately had an endoprosthesis implanted in the infected site. Except in well-resourced laboratories with well-trained staff, the errors and delays in diagnosis can also be explained by the difficulty to identify B. pseudomallei in common bacterial cultures, though it remains the standard method for the diagnosis of melioidosis. The centers for disease control and prevention (CDC) recommends that blood, urine and throat cultures be performed for all patients with suspected melioidosis, and not only a specimen from the affected site.7 B. pseudomallei grows well on most laboratory media, but selective media such as Ashdown’s agar may increase the yield from sites with a normal flora.7
In the few reported cases, aneurysms secondary to melioidosis are often treated with long-term intravenous antibiotics (high dose ceftazidime or meropenem/imipenem 4–6 weeks) and then a course of oral antibiotics (sulfamethoxazole/trimethoprim 3–6 months).1 8 Some drug combinations, like sulfamethoxazole/trimethoprim and doxycycline, remain questionable, due to an in vitro antagonism between both drugs. However, given the rarity of cases, the level of evidence for managing melioidosis aneurysm remains very low. However, surgical removal of the aneurysm is always required.8 The timing is determined by estimating the risk of rupture and the surgical risk based on the patient’s general condition. Our case is unique in that melioidosis was discovered in the context of a vascular periprosthetic infection. Given the low probability of rupture, surgery was performed after an improvement in the clinical signs of infection and overall patient health.
In conclusion, our immunocompetent patient was diagnosed with infected aortic aneurysm, lumbar spondylitis and psoas abscess due to B. pseudomallei. The patient had no risk factors for melioidosis but had recently returned from endemic area with unexplained fever and deterioration of general health. Melioidosis should be considered as a possible cause of unexplained fever in individuals returning from an endemic area in order to avoid inappropriate surgical and antibiotic treatments.
Learning points.
Melioidosis should be considered in travellers returning from Southeast Asia, northern Australia or tropical areas with unexplained fever and deteriorating general health.
In the absence of early diagnosis and right management, systemic dissemination and secondary life-threatening complications such as aneurysms may arise.
Melioidosis aneurysms must be surgically removed and the patient put on long-term antibiotic therapy.
Acknowledgments
We thank Suzanne Rankin for reviewing the manuscript and Dr Olivier Gorgé from the Laboratory of highly pathogenic Burkholderia (Biomedical Research Institute of the Armed Forces—Bretigny—France) for his help in managing the patient.
Footnotes
Contributors: CA and MB wrote the case report. CN and LP reviewed the case report.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Patient consent for publication: Obtained
References
- 1. Wiersinga WJ, Virk HS, Torres AG, et al. Melioidosis. Nat Rev Dis Primers 2018;4:17107 10.1038/nrdp.2017.107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Currie BJ, Ward L, Cheng AC. The epidemiology and clinical spectrum of melioidosis: 540 cases from the 20 year Darwin prospective study. PLoS Negl Trop Dis 2010;4:e900 10.1371/journal.pntd.0000900 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Azizi ZA, Yahya M, Lee SK. Melioidosis and the vascular surgeon: Hospital Kuala Lumpur experience. Asian J Surg 2005;28:309–11. 10.1016/S1015-9584(09)60368-7 [DOI] [PubMed] [Google Scholar]
- 4. Amezyane T, Lecoules S, Algayres JP. Mycotic iliac aneurysm associated with Burkholderia pseudomallei. Int J Infect Dis 2010;14(Suppl 3):e381–2. 10.1016/j.ijid.2009.07.008 [DOI] [PubMed] [Google Scholar]
- 5. Yew KL, Choy CN, Kam JY, et al. Intracoronary blood sampling with a microcatheter for the diagnosis of giant infective coronary aneurysm: Melioidosis of coronary artery mycotic aneurysm. Int J Cardiol 2015;187:530–1. 10.1016/j.ijcard.2015.04.013 [DOI] [PubMed] [Google Scholar]
- 6. Low JG, Quek AM, Sin YK, et al. Mycotic aneurysm due to Burkholderia pseudomallei infection: case reports and literature review. Clin Infect Dis 2005;40:193–8. 10.1086/426590 [DOI] [PubMed] [Google Scholar]
- 7. Hemarajata P, Baghdadi JD, Hoffman R, et al. Burkholderia pseudomallei: challenges for the clinical microbiology laboratory. J Clin Microbiol 2016;54:2866–73. 10.1128/JCM.01636-16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Panginikkod S, Ramachandran A, Bollimunta P, et al. Burkholderia Aortic Aneurysm: A Case Report and Review of the Literature. Case Rep Infect Dis 2017;2017:1–4. 10.1155/2017/6206395 [DOI] [PMC free article] [PubMed] [Google Scholar]