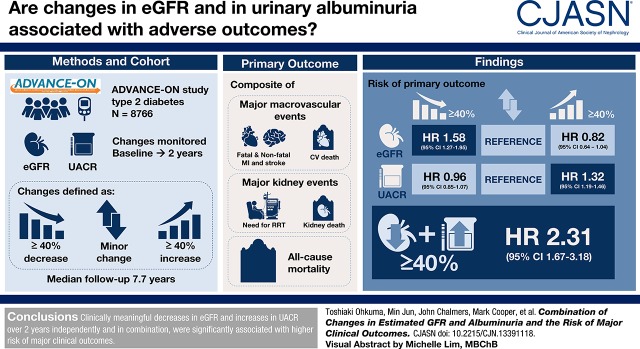
Visual Abstract
Keywords: albuminuria; cardiovascular disease; glomerular filtration rate; ESRD; diabetes mellitus; creatinine; ESKD; Diabetes Mellitus, Type 2; kidney; Stroke; Myocardial Infarction; Risk; Renal Replacement Therapy; Albumins
Abstract
Background and objectives
Whether combining changes in eGFR and urine albumin-to-creatinine ratio (UACR) is more strongly associated with outcomes compared with either change alone is unknown.
Design, setting, participants, & measurements
We analyzed 8766 patients with type 2 diabetes in the Action in Diabetes and Vascular Disease: Preterax and Diamicron Modified Release Controlled Evaluation Observational (ADVANCE-ON) study. Changes in eGFR and UACR (baseline to 2 years) were defined as ≥40% decrease, minor change, and ≥40% increase. The primary outcome was the composite of major macrovascular (nonfatal or fatal myocardial infarction, nonfatal or fatal stroke, or cardiovascular death), major kidney events (requirement for kidney replacement therapy or kidney death), and all-cause mortality.
Results
Over a median of 7.7 years of follow-up, 2191 primary outcomes were recorded. Strong linear associations between eGFR and UACR changes and subsequent risk of the outcome were observed. For eGFR, the hazard ratios were 1.58 (95% confidence interval [95% CI], 1.27 to 1.95) for a decrease ≥40% and 0.82 for an increase ≥40% (95% CI, 0.64 to 1.04) compared with minor change. For UACR, the hazard ratios were 0.96 (95% CI, 0.85 to 1.07) for a decrease ≥40% and 1.32 (95% CI, 1.19 to 1.46) for ≥40% increase compared with minor change. Compared with dual minor changes, both an eGFR decrease ≥40% and a UACR increase ≥40% had 2.31 (95% CI, 1.67 to 3.18) times the risk of the outcome, with evidence of interaction between the two markers.
Conclusions
Clinically meaningful decreases in eGFR and increases in UACR over 2 years, independently and in combination, were significantly associated with higher risk of major clinical outcomes.
Introduction
One of the most frequent complications of diabetes is CKD (1,2), and diabetic kidney disease is the leading cause of ESKD (1,2). Diabetic kidney disease also increases the risk of cardiovascular disease (3), which is the leading cause of morbidity and mortality among people with diabetes (2). As such, the development of novel treatments for the prevention and management of diabetic kidney disease has become crucial.
The number of randomized, controlled trials providing the basis for clinical decision making in nephrology is fewer than for any other specialty of internal medicine (4). This may well be because of the slow progressive nature of kidney disease, requiring a long duration to progress to the hard end point of ESKD. In response, CKD trials are being designed with surrogate kidney end points to shorten the trial duration, enroll fewer participants, reduce trial costs, and to study interventions in patients with earlier disease stages (5,6).
A decrease in eGFR of 40%, or even 30% in some circumstances, is now widely accepted as an end point for clinical trials of CKD (5,6). Change in proteinuria or albuminuria has also been widely used as a possible surrogate end point for kidney disease progression (7,8). In the Action in Diabetes and Vascular Disease: Preterax and Diamicron Modified Release Controlled Evaluation (ADVANCE) randomized, controlled trial and the Action in Diabetes and Vascular Disease: Preterax and Diamicron Modified Release Controlled Evaluation Observational (ADVANCE-ON) study, we previously reported that increases in urine albumin-to-creatinine ratio (UACR) over 2 years in patients with type 2 diabetes were independently associated with cardiovascular events and all-cause mortality, as well as kidney events (9). To our knowledge, no study has examined whether combining changes in eGFR and UACR will more accurately predict the subsequent risk of major clinical outcomes than either change alone.
The objective of this study was to examine the association between the combination of changes in eGFR and UACR and subsequent risk of major clinical outcomes in patients with type 2 diabetes in the ADVANCE and ADVANCE-ON study.
Materials and Methods
Study Design and Population
ADVANCE was a factorial, randomized, controlled trial evaluating the effects of BP-lowering and intensive blood glucose–lowering treatment on vascular outcomes in patients with type 2 diabetes. A detailed description of the design has been published previously (10–12). In brief, 11,140 individuals with type 2 diabetes, aged ≥55 years, and at high risk of cardiovascular events were enrolled from 215 centers in 20 countries. No inclusion or exclusion criterion was based on levels of BP or eGFR, but the presence of albuminuria was one of a number of potential inclusion criteria. Participants were randomized to a fixed-dose combination of perindopril and indapamide (2/0.625 mg) or matching placebo, and also to either a gliclazide (modified release)-based intensive glucose control regimen aiming to achieve a hemoglobin A1c (HbA1c) ≤6.5%, or standard glucose control according to local guidelines, after a 6-week active run-in period. The dose of randomized perindopril and indapamide treatment was doubled to 4 and 1.25 mg, respectively, 3 months after randomization. The ADVANCE-ON study was a post-trial follow-up study of the ADVANCE trial, comprising 8494 participants out of a total of 10,082 participants alive when the randomized treatment phase was completed (13). All participants discontinued the randomized treatments and returned to usual care through their physician. Participants were followed up for an overall median of 9.9 years (including both ADVANCE [4.4 and 5.0 years for BP-lowering and intensive blood glucose–lowering comparison, respectively] and ADVANCE-ON). Ethics approval of the study was obtained from the institutional review board of each center. All participants provided written informed consent. The study was conducted in accordance with the Declaration of Helsinki.
For the present analysis, patients with major macrovascular or kidney events or death during the first 2 years, those with missing eGFR and UACR values at either of two occasions (study registration or 2 years after randomization), or those with missing values in covariates were excluded.
eGFR and UACR Evaluations
Our exposure variables were changes in eGFR and UACR from study registration to 2 years after randomization, i.e., over a period of 25.5 months or approximately 2 years. eGFR was calculated by the CKD Epidemiology Collaboration creatinine equation (14). For the primary analysis both of the kidney markers were classified into three groups: a decrease of ≥40%, minor change (decrease of <40% to increase <40%), and an increase of ≥40%.
Follow-Up and Study Outcomes
Patients were followed up from their 24-month visit until the earliest of the first study event, death, or the end of follow-up (Supplemental Figure 1). The primary study outcome was the composite of major macrovascular events (nonfatal or fatal myocardial infarction, nonfatal or fatal stroke, or cardiovascular death), major kidney events (requirement for kidney replacement therapy or kidney death), and all-cause mortality. Secondary outcomes were the three main components of the primary outcome.
Study outcomes recorded during the randomized treatment intervention period were reviewed and validated by an independent end point adjudication committee. The outcomes that occurred during the postrandomization observational period were reported by the study centers using the standardized definitions adopted during the trial, without adjudication (13).
Statistical Analyses
Continuous variables are presented as means (SD) for variables approximately symmetric distributed. Continuous variables with skewed distributions are presented as median (interquartile range). Linear trends of baseline characteristics across categories were tested by linear regression analysis and logistic regression analysis, as appropriate.
Cox regression models were used to estimate hazard ratios (HRs) and their corresponding 95% confidence intervals (95% CIs) for changes in eGFR and UACR. Models were adjusted for age, sex, region of residence (Asia or other), duration of diabetes mellitus, history of macrovascular disease, current smoking, current alcohol consumption, body mass index, HbA1c, total cholesterol, triglyceride, systolic BP, eGFR, UACR at registration, randomized BP-lowering intervention, randomized glucose control intervention, and percent 2-year changes in systolic BP, eGFR (for the analyses of change in UACR only), and UACR (for the analyses of change in eGFR only). We performed sensitivity analyses in which randomized BP and glucose control treatments and change in systolic BP were excluded from covariates, or change in HbA1c was added as a covariate. We also conducted sensitivity analyses, in which the cut-off point for changes in eGFR and UACR was defined as 30% instead of 40%. For change in UACR, other sensitivity analyses using change in UACR on the basis of log-transformed UACR, or a combination of change in albuminuria category (normo-, micro-, and macroalbuminuria) and percent change in UACR (Supplemental Figure 2) were also conducted. Further, we examined the association between changes in eGFR and log-transformed UACR as continuous variables and the outcomes using restricted cubic spline regression models with knots placed at −40%, −30%, 0%, 30%, and 40% change. In addition, it is a fact of nature that someone who has a high value at baseline will be likely to have a lower and closer value to the overall mean on a subsequent measurement and vice versa—so-called regression to the mean. To allow for this, we repeated the analyses, but only considered anyone in the highest or middle thirds of eGFR or UACR at baseline whose value went up by ≥40% or experienced minor change for the highest third at 2 years to have a “real” increase; that is, a residual increase after accounting for regression to the mean. Similarly, only patients in the middle or lowest thirds whose values went down by ≥40% or who experienced minor change for the lowest third were considered to have a “real” residual decrease, over and above regression to the mean (Supplemental Figure 3). A similar analysis was conducted by using a 10% cut-off point for eGFR and UACR instead of 40% (Supplemental Figure 4).
Prediction metrics for the primary outcome were also investigated. Discrimination was evaluated by c-statistics for 8-year risk, accounting for censoring, and compared between the above-mentioned conventional risk model and this model plus changes in eGFR and UACR individually and in combination. In addition, the ability to reclassify the 8-year risk was assessed by the integrated discrimination index and net reclassification improvement, using methods suitable for survival data.
Statistical analyses were performed with SAS 7.11 (SAS Institute, Cary, NC) and Stata software (release 13; StataCorp, College Station, TX). A two-sided P value <0.05 was considered statistically significant.
Results
Patient Characteristics at Registration
Of the 11,140 patients who participated in the ADVANCE trial, 8766 participants (79%) were included in this study; those excluded either had missing values of exposures or covariates or experienced the primary outcome during the initial 2-year period over which changes in kidney markers were quantified (Supplemental Figure 1). The mean age of included participants was 66 years (SD 6) and 43% were women. Mean duration of diabetes was 7.8 years (SD 6.3), mean eGFR was 75 ml/min per 1.73 m2 (SD 17), and median UACR was 14.1 µg/mg (interquartile range, 7.1–37.1).
Changes in eGFR and UACR
Over the 2-year change “window,” 276 (3%) participants experienced a decrease in eGFR of ≥40%, 8186 (93%) participants experienced minor change, and 304 (4%) participants experienced an increase of ≥40% (Table 1). For UACR, 2515 (29%) participants experienced a decrease of ≥40%, 3002 (34%) participants experienced minor change, and 3249 (37%) participants experienced an increase of ≥40% (Table 1). A total of 108 participants (1%) experienced both a decrease in eGFR of ≥40% and an increase in UACR of ≥40% (Supplemental Table 1A).
Table 1.
Characteristics at time of registration of 8766 people included in our analysis from the ADVANCE trial
| Variable | eGFR Change | UACR Change | ||||
|---|---|---|---|---|---|---|
| Decrease ≥40% | Minor Change | Increase ≥40% | Decrease ≥40% | Minor Change | Increase ≥40% | |
| No. of participants | 276 | 8186 | 304 | 2515 | 3002 | 3249 |
| Demographic factors | ||||||
| Age, yr | 65 (7) | 66 (6) | 65 (6) | 66 (6) | 65 (6) | 65 (6) |
| Women, % | 166 (60) | 3381 (41) | 183 (60) | 1065 (42) | 1246 (42) | 1419 (44) |
| Residence in Asia, % | 180 (65) | 3168 (38) | 174 (57) | 1058 (42) | 1179 (39) | 1285 (40) |
| Medical and lifestyle history | ||||||
| Duration of diabetes mellitus, yr | 9.2 (7.0) | 7.8 (6.2) | 7.8 (6.4) | 8.0 (6.4) | 7.5 (6.0) | 7.9 (6.4) |
| History of macrovascular disease at baseline, % | 73 (26) | 2529 (31) | 101 (33) | 779 (31) | 881 (29) | 1043 (32) |
| Current smoking, % | 28 (10) | 1219 (15) | 41 (13) | 364 (14) | 436 (15) | 488 (15) |
| Current alcohol drinking, % | 38 (14) | 2511 (31) | 47 (15) | 734 (29) | 939 (31) | 923 (28) |
| Risk factors | ||||||
| Systolic BP, mm Hg | 147 (22) | 144 (21) | 145 (22) | 147 (21) | 143 (21) | 144 (21) |
| Diastolic BP, mm Hg | 81 (12) | 81 (11) | 80 (11) | 82 (11) | 80 (11) | 80 (11) |
| Heart rate, bpm | 76 (12) | 74 (12) | 76 (12) | 75 (12) | 74 (12) | 74 (12) |
| HbA1c, % | 8.0 (1.9) | 7.5 (1.5) | 7.7 (1.8) | 7.5 (1.6) | 7.5 (1.5) | 7.5 (1.5) |
| Total cholesterol, mg/dl | 203 (46) | 200 (46) | 220 (51) | 203 (47) | 200 (44) | 200 (46) |
| Triglycerides, mg/dl | 147 (106–221) | 143 (106–204) | 151 (106–230) | 151 (106–213) | 142 (105–204) | 142 (106–204) |
| Body mass index, kg/m2 | 27.2 (4.6) | 28.3 (5.2) | 27.7 (4.8) | 28.3 (5.2) | 28.2 (5.1) | 28.2 (5.2) |
| Randomized treatments | ||||||
| Perindopril-indapamide | 159 (58) | 4066 (50) | 131 (43) | 1405 (56) | 1512 (50) | 1439 (44) |
| Intensive blood glucose control | 150 (54) | 4158 (51) | 150 (49) | 1340 (53) | 1524 (51) | 1594 (49) |
| Blood glucose–lowering treatments | ||||||
| Oral hypoglycemic agents, %a | 258 (93) | 7417 (91) | 279 (92) | 2285 (91) | 2703 (90) | 2966 (91) |
| Insulin, % | 4 (1) | 110 (1) | 11 (4) | 44 (2) | 37 (1) | 44 (1) |
| BP-lowering treatments | ||||||
| β-blocker, %s | 55 (20) | 1984 (24) | 73 (24) | 569 (23) | 715 (24) | 828 (25) |
| Calcium-channel blocker, % | 90 (33) | 2460 (30) | 116 (38) | 827 (33) | 840 (28) | 999 (31) |
| Diuretics, %b | 65 (24) | 1861 (23) | 88 (29) | 540 (21) | 657 (22) | 817 (25) |
| Angiotensin-converting enzyme inhibitors, %b | 107 (39) | 3457 (42) | 142 (47) | 1008 (40) | 1263 (42) | 1435 (44) |
| Angiotensin II receptor blockers, % | 16 (6) | 418 (5) | 7 (2) | 135 (5) | 144 (5) | 162 (5) |
| Other antihypertensive agents, % | 66 (24) | 967 (12) | 55 (18) | 318 (13) | 358 (12) | 412 (13) |
| Any BP-lowering agents, %b | 222 (80) | 6057 (74) | 243 (80) | 1900 (76) | 2171 (72) | 2451 (75) |
| Changes in risk factors | ||||||
| First UACR, mg/g | 21 (10–72) | 14 (7–35) | 14 (8–47) | 33 (16–92) | 13 (7–27) | 9 (4–19) |
| Second UACR, mg/g | 21 (11–66) | 14 (7–37) | 14 (7–45) | 9 (4–19) | 12 (7–27) | 28 (12–77) |
| First eGFR, ml/min per 1.73 m2 | 85 (21) | 76 (17) | 54 (11) | 76 (18) | 76 (17) | 75 (17) |
| Second eGFR, ml/min per 1.73 m2 | 44 (12) | 72 (17) | 85 (14) | 72 (18) | 72 (18) | 72 (18) |
| First systolic BP, mm Hg | 147 (22) | 144 (21) | 145 (21) | 147 (21) | 143 (21) | 144 (21) |
| Second systolic BP, mm Hg | 138 (19) | 138 (18) | 139 (18) | 137 (18) | 137 (18) | 139 (19) |
For continuous variables, mean values and their corresponding SD are presented, except that median values (interquartile interval [IQI[) are presented for triglycerides and UACR. Linear trends of triglycerides and UACR across categories were tested after log-transformation. Categorical variables are presented as n (%). ADVANCE, Action in Diabetes and Vascular Disease: Preterax and Diamicron Modified Release Controlled Evaluation; UACR, urine albumin-to-creatinine-ratio; HbA1c, hemoglobin A1c.
Randomized treatment with gliclazide was not included.
Randomized treatment with perindopril-indapamide was not included.
Participants with greater decreases in eGFR were more likely to have longer diabetes duration (Table 1), higher HbA1c levels, higher UACR and eGFR levels at registration, and to have been randomized to perindopril and indapamide treatment.
Participants with greater increases in UACR were more likely to be taking diuretics or angiotensin-converting enzyme inhibitors and to have lower UACR and systolic BP levels at registration, and were less likely to have been randomized to perindopril and indapamide or intensive blood glucose control.
Association of Changes in eGFR and UACR with Clinical Outcomes
During a median follow-up of 7.7 years, 2191 patients (25%) developed the primary outcome, 1392 (16%) experienced a major macrovascular event, 108 (1%) experienced a major kidney event, and 1416 (16%) died.
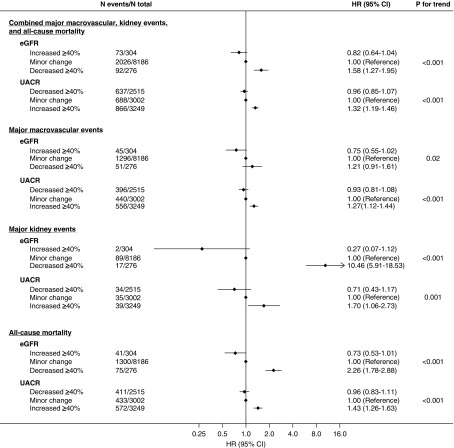
The HRs and 95% CIs for major clinical outcomes are shown in Figure 1. After adjustment (for eGFR, UACR, and other cardiovascular risk factors at study entry), an increase in eGFR was associated with a lower risk of the primary outcome. Compared with minor change, the HR for a decrease in eGFR of ≥40% was 1.58 (95% CI, 1.27 to 1.95), whereas the HR for an increase in eGFR of ≥40% was 0.82 (95% CI, 0.64 to 1.04).
Figure 1.
Both a decrease in eGFR and an increase in UACR over 2 years were significantly associated with subsequent risk of major clinical outcomes. Models were adjusted for age, sex, region of residence, duration of diabetes, history of macrovascular disease, smoking habit, drinking habit, body mass index, HbA1c, total cholesterol, log-transformed triglycerides, eGFR, systolic BP, log-transformed UACR at registration, 2-year change in systolic BP, randomized BP-lowering intervention, and randomized glucose control intervention plus (for change in eGFR only) log-transformed 2-year change in UACR or (for change in UACR only) 2-year change in eGFR.
After adjustment, an increase in UACR was associated with a higher risk of the primary outcome. Compared with minor change, the HR for a decrease in UACR of ≥40% was 0.96 (95% CI, 0.85 to 1.07), whereas the HR for an increase in UACR of ≥40% was 1.32 (95% CI, 1.19 to 1.46). These results remained unchanged when (1) randomized BP and glucose control treatments and change in systolic BP were excluded from covariates, (2) change in HbA1c was added as a covariate (Supplemental Figure 5), (3) percent change was calculated on the basis of log-transformed UACR (Supplemental Figure 6), or (4) a combination of change in albuminuria category and percent change in UACR was used (Supplemental Figures 2 and 6). Findings were similar when changes in eGFR and UACR were assessed as continuous variables (Figure 2). There was also a statistically significant trend for major macrovascular events alone, major kidney events alone, and all-cause mortality when considered alone (Figure 1). These effects were not substantially changed in sensitivity analyses using a 30% cut-off point for the changes in eGFR and UACR (Supplemental Figure 7, Supplemental Table 1), and adjusting for regression to the mean (Supplemental Figures 8 and 9).
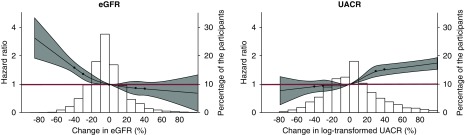
Figure 2.
The associations between changes in eGFR and UACR and subsequent risk of major clinical outcomes were similar when changes in these two markers were assessed as continuous variables. 2-Year changes in eGFR and UACR were both strongly associated with future risks of major clinical outcomes. Models were adjusted for age, sex, region of residence, duration of diabetes, history of macrovascular disease, smoking habit, drinking habit, body mass index, HbA1c, total cholesterol, log-transformed triglycerides, eGFR, systolic BP, log-transformed UACR at registration, 2-year change in systolic BP, randomized BP-lowering intervention, and randomized glucose control intervention plus (for change in eGFR only) 2-year change in UACR or (for change in UACR only) 2-year change in eGFR. The circles represent the points at which knots were placed (−40%, −30%, 0%, 30%, and 40%). The gray areas represent the 95% CIs. Values were trimmed at >100% change (0.16% and 3.0% of the participants for change in eGFR and change in UACR, respectively). The red lines represent the HRs of one.
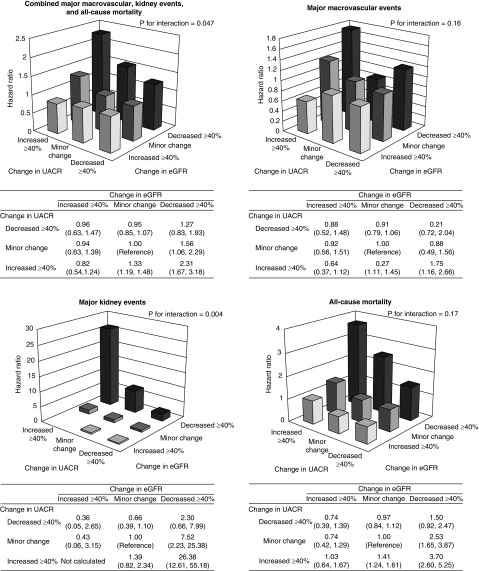
Combination of Changes in eGFR and UACR and Clinical Outcomes
Compared with minor changes in both eGFR and UACR, a combined decrease in eGFR and an increase in UACR were associated with a 2.31 (95% CI, 1.67 to 3.18) higher risk of the primary outcome, with evidence of interaction between the two kidney markers (P for interaction <0.05) (Figure 3). The corresponding HRs for major macrovascular events, major kidney events, and all-cause mortality were 1.75 (95% CI, 1.16 to 2.66), 26.38 (95% CI, 12.61 to 55.18), and 3.70 (95% CI, 2.60 to 5.25), respectively (P for interaction =0.16; 0.004; and 0.17, respectively). These effects were not substantially changed in sensitivity analyses using a 30% cut-off point for the combined changes in eGFR and UACR (Supplemental Figure 10) and those accounting for regression to the mean using a 40% cut-off point for the changes in eGFR and UACR (Supplemental Figure 11), although significant interaction between the two markers were no longer evident in the latter analysis.
Figure 3.
A combination of changes in eGFR and UACR was more strongly associated with subsequent risk of the outcomes than either change alone. Models were adjusted for age, sex, region of residence, duration of diabetes, history of macrovascular disease, smoking habit, drinking habit, body mass index, HbA1c, total cholesterol, log-transformed triglycerides, eGFR, systolic BP, log-transformed UACR at registration, change in systolic BP, randomized BP-lowering intervention, and randomized glucose control intervention.
Discrimination and Reclassification
The addition of change in eGFR to a model with conventional risk factors consistently and significantly improved a range of discrimination and reclassification of the 8-year risk of the primary outcome (Table 2). When change in UACR was added to the same base model, these prediction metrics were also improved significantly, with exception of categorical net reclassification improvement. Furthermore, the addition of a combination of both change in eGFR and UACR, and their interaction term provided better prognostic information, when compared with adding any of the change individually.
Table 2.
Discrimination and reclassification statistics (95% CIs) for 8-year risk of the primary outcome after addition of changes in eGFR or UACR to a model containing clinical risk factors
| Model | Δc-Statistic | Integrated Discrimination Improvement | Relative Integrated Discrimination Improvement, % | Net Reclassification Improvement | |
|---|---|---|---|---|---|
| Continuous | Categoricala | ||||
| Base model (reference)b | 0.6602 (0.6478 to 0.6726) | ||||
| Base model plus change in eGFR | 0.0041 (0.0010 to 0.0073) | 0.0046 (0.0036 to 0.0058) | 5.39 (4.11 to 6.80) | 0.114 (0.059 to 0.166) | 0.014 (0.0007 to 0.027) |
| P=0.009 | P<0.001 | P<0.001 | P=0.04 | ||
| Base model plus change in UACR | 0.0054 (0.0022 to 0.0086) | 0.0050 (0.0038 to 0.0062) | 5.85 (4.40 to 7.31) | 0.170 (0.116 to 0.224) | 0.0005 (−0.014 to 0.014) |
| P=0.001 | P<0.001 | P<0.001 | P=0.18 | ||
| Base model plus change in eGFR and UACR and interaction term | 0.0088 (0.0044 to 0.0131) | 0.0096 (0.0080 to 0.0114) | 11.21 (9.25 to 13.34) | 0.168 (0.112 to 0.219) | 0.017 (0.0003 to 0.033) |
| P<0.001 | P<0.001 | P<0.001 | P=0.05 | ||
| Base model plus change in eGFR (reference) | 0.6643 (0.6519 to 0.6768) | ||||
| Addition of change in UACR and interaction term | 0.0046 (0.0016 to 0.0077) | 0.0050 (0.0038 to 0.0062) | 5.55 (4.18 to 6.88) | 0.175 (0.121 to 0.226) | 0.002 (−0.011 to 0.015) |
| P=0.003 | P<0.001 | P<0.001 | P=0.56 | ||
| Base model plus change in UACR (reference) | 0.6656 (0.6533 to 0.6780) | ||||
| Addition of change in eGFR and interaction term | 0.0034 (0.0004 to 0.0063) | 0.0046 (0.0035 to 0.0058) | 5.08 (3.85 to 6.39) | 0.107 (0.051 to 0.159) | 0.017 (0.004 to 0.029) |
| P=0.03 | P<0.001 | P<0.001 | P=0.006 | ||
Change in UACR was log-transformed before the analyses. 95% CI, 95% confidence interval; UACR, urine albumin-to-creatinine ratio.
Using cut-off points of 10% and 20% 8-year risk.
Base model included age, sex, region of residence, duration of diabetes, history of macrovascular disease, smoking habit, drinking habit, body mass index, hemoglobin A1c, total cholesterol, log-transformed triglycerides, eGFR, systolic BP, log-transformed UACR at registration, 2-year change in systolic BP, randomized BP-lowering intervention, and randomized glucose control intervention.
Discussion
In our study cohort of 8766 patients with type 2 diabetes, 2-year changes in eGFR and UACR were both strongly associated with future risks of major clinical outcomes. Both decreases in eGFR and increases in UACR over 2 years, independently and in combination, predicted increased risk of the primary outcome. There was some evidence of an interaction between changes in eGFR and UACR such that, compared with minor eGFR and UACR changes, eGFR decreases of ≥40% and UACR increases of ≥40% was associated with 2.3 (95% CI, 1.7 to 3.2) and 26.4 (95% CI, 12.6 to 55.2) times increased risks of the primary outcome and major kidney events, respectively. Overall, our results suggest that a combined assessment of clinically meaningful changes in both eGFR and UACR compared with separate assessments of the two prognostic markers further improves risk stratification of people with type 2 diabetes at risk of developing major cardiovascular and kidney events.
The strong graded link between baseline levels of eGFR and albuminuria (and the interaction between these two prognostic markers) on subsequent risk of adverse cardiovascular and kidney outcomes is well established. We had previously reported that for every halving of baseline eGFR and every ten-fold increase in baseline UACR, the risk of a cardiovascular event was increased by a factor of 2.2 (95% CI, 1.1 to 4.4) and 2.5 (95% CI, 1.7 to 3.5), respectively (15). Other studies (16–18) have also suggested an independent predictive effect of baseline eGFR and UACR for future adverse outcomes, with some (17,18) also showing strong interaction between the two prognostic markers (e.g., the presence of both low eGFR and higher degrees of albuminuria further intensifying the risk of future adverse outcomes).
Accordingly, more recent studies have focused on assessing the prognostic effect of short- and long-term changes in eGFR (19,20) and UACR (9,21–23), to determine their utility as a therapeutic target and surrogate end points for cardiovascular and kidney outcomes—an area of intense debate (24–26). Although doubling of serum creatinine (equivalent to a 57% decrease in eGFR) from baseline has been widely used as a surrogate for ESKD in randomized trials, lesser magnitudes of eGFR decline (e.g., 30% and 40%) have recently been proposed (24), especially as the robust ascertainment of doubling of serum creatinine still requires substantially longer follow-up.
Since the publication of results from the National Kidney Foundation and US Food and Drug Administration–sponsored Scientific Workshop on GFR decline suggesting general concordance between 40% eGFR decline and established ESKD end points (ESKD or doubling of serum creatinine) (24), this end point has been increasingly used in randomized trials (27,28). Our results indicate that 40% changes in eGFR are strongly predictive of major clinical outcomes and, although our study did not aim to address the superiority of change in eGFR ≥40% as a surrogate end point compared with change in eGFR ≥30%, we observed similar effects across the two models, suggesting the potential utility of change in eGFR of ≥30% as an end point among certain high-risk groups, including type 2 diabetes. Indeed, the use of eGFR change of ≥30% resulted in more participants reaching the end point compared with eGFR change of ≥40% (higher across the assessed eGFR and UACR change categories).
Regarding UACR change, recent studies have tested the prognostic utility of 30%–100% changes in UACR, with a growing number of observational studies (9,21) showing positive association between changes in UACR and the risk of cardiovascular or kidney outcomes (particularly for increases in UACR and subsequently increased risk of adverse outcomes). We have previously reported a strong positive linear association between changes in UACR (defined as UACR decrease ≥30% or UACR increase ≥30% compared with minor change) and major macrovascular and major kidney events as well as all-cause mortality among patients with type 2 diabetes (9). In this study, there were more than one third of total participants in each category of increase or decrease in UACR, but only 3%–4% in corresponding categories for change in eGFR, due to the skewed distribution of UACR and subject to random variation. However, sensitivity analyses using change in log-transformed UACR or a combination of change in albuminuria category and percent change in UACR provided broadly similar results, which support our general conclusions.
To our knowledge, no study to date has assessed the interaction between changes in eGFR and albuminuria and subsequent risk of major clinical outcomes including cardiovascular and kidney events. This assessment is critical for the appropriate identification of high-risk populations. For example, a study by Hemmelgarn et al. (18) reported that patients with heavy proteinuria (albumin-to-creatinine ratio >300 mg/g) without moderate CKD (eGFR>60 ml/min per 1.73 m2) experienced greater rates of cardiovascular and kidney outcomes compared with those who had moderate CKD but without proteinuria, highlighting the potential for misclassification of risk when changes in the two prognostic markers are considered in isolation.
To the best of our knowledge, our study represents the first assessment on the combined predictive value of eGFR and UACR change in assessing future cardiovascular and kidney risk in people with type 2 diabetes. Our overall results suggest that a combined approach of determining clinically meaningful magnitudes of earlier change in both eGFR and UACR in type 2 diabetes may add substantial prognostic value to that associated with eGFR or albuminuria change alone. Our results are largely consistent with those from a similar study reporting on the analysis of baseline eGFR and albuminuria from the Kidney Early Evaluation Program. However, the effect of changes in eGFR and albuminuria on outcomes or the interaction between these two prognostic markers were not assessed, although the improvements in predictive ability had been anticipated. Our data thus have important implications for risk stratification of individuals with type 2 diabetes at high risk of developing adverse cardiovascular and kidney outcomes. Although the degree of improvements in some prediction statistics were statistically significant, they were only small. However, we believe that even a modest improvement could be beneficial to prevent adverse events in these high-risk populations.
The strengths of our study include (1) the assessment of the relationship between change in eGFR and UACR and clinically important outcomes using separate (for eGFR and UACR) and combined approaches (i.e., analysis of the interaction between change in eGFR and UACR); (2) the large and diverse participant population (including Asia [40%], Australasia [14%], Europe [43%], and North America [3%]) derived from an international, multicenter, randomized trial; and (3) the long-term follow-up period, which included the 5-year post-trial phase. However, our study has limitations. First, our calculation of the percent change in eGFR and UACR was on the basis of only two eGFR and UACR measurements, respectively, with the second assessed 2 years after the initial measurement. Second, our study cohort was derived from a randomized trial of people with type 2 diabetes and therefore the results have limited generalizability to broader populations. Third, 84% of the participants alive at the end of the ADVANCE trial were enrolled in the post-trial follow-up (ADVANCE-ON). However, patient baseline characteristics of those included in the ADVANCE-ON trial were similar to those of the entire trial population (13). Finally, the ESKD event rate in the ADVANCE/ADVANCE-ON trials was relatively low (0.2% per year) compared with prior studies that included people with diabetes (0.7%–6.6% per year) (21,29,30).
In conclusion, we observed continuous associations between 2-year changes in eGFR and UACR and the risk of major clinical outcomes, with some evidence of significant interaction between changes in eGFR and UACR. Our results suggest that a combined assessment of eGFR and UACR change will add prognostic utility compared with separate assessments of the changes in the two prognostic markers in patients with type 2 diabetes.
Disclosures
Dr. Jun reports receiving grant support from the National Health and Medical Research Council of Australia (project grant: 1148060) and unrestricted grant support from VentureWise (a wholly owned commercial subsidiary of NPS MedicineWise) to conduct a commissioned project funded by AstraZeneca, outside the submitted work. Dr. Chalmers received research grants from the National Health and Medical Research Council of Australia and from Servier for the ADVANCE trial and ADVANCE-ON post-trial follow-up, and honoraria for speaking about these studies at scientific meetings, and reports grant support from Program Grant from the National Health and Medical Research Council of Australia. Dr. Cooper reports personal fees from Servier, grants from NovoNordisk, grants and personal fees from Boehringer Ingeleheim, personal fees from AstraZeneca, personal fees from Novartis, personal fees from Merck, and personal fees from Bayer, outside the submitted work. Dr. Hamet reports consulting fees from Servier, grant support from Quebec CQDM and Servier. Dr. Harrap reports grants from George Institute for International Health, during the conduct of the study; and other from Servier, outside the submitted work. Dr. Zoungas reports other from Sanofi, AstraZeneca, Novo Nordisk, and MSD Australia, outside the submitted work. Dr. Perkovic reports receiving personal fees for Advisory Boards or Scientific Presentations from Retrophin, Janssen, Merck, and Servier. Dr. Perkovic also reports receiving fees for Advisory Boards, Steering Committee roles, or Scientific Presentations from Abbvie, Astellas, Astra Zeneca, Bayer, Baxter, BMS, Boehringer Ingelheim, Dimerix, Durect, Eli Lilly, Gilead, GSK, Janssen, Merck, Mitsubishi Tanabe, Novartis, Novo Nordisk, Pfizer, Pharmalink, Relypsa, Retrophin, Sanofi, Servier, Vifor, and Tricida. Dr. Woodward reports personal fees from Amgen and Kirin, outside the submitted work. Dr. Ohkuma has nothing to disclose.
Supplemental Material
This article contains the following supplemental material online at http://cjasn.asnjournals.org/lookup/suppl/doi:10.2215/CJN.13391118/-/DCSupplemental.
Supplemental Table 1. Numbers of participants according to changes in eGFR and UACR.
Supplemental Figure 1. Study design and identification of the study cohort.
Supplemental Figure 2. Diagram showing the grouping of participants according to a combination of change in albuminuria category and percent change in UACR.
Supplemental Figure 3. Diagram showing the grouping of participants according to baseline and change in (A) eGFR and (B) UACR to account for regression to the mean, using a 40% cut-off point for the changes in eGFR and UACR.
Supplemental Figure 4. Diagram showing the grouping of participants according to baseline and change in (A) eGFR and (B) UACR to account for regression to the mean, using a 10% cut-off point for the changes in eGFR and UACR.
Supplemental Figure 5. Sensitivity analysis. Adjusted associations of 2-year changes in eGFR or UACR with the risk of subsequent major clinical outcomes, after exclusion of randomized BP and glucose control treatments and change in systolic BP were excluded from covariates, or additional adjustment for change in HbA1c.
Supplemental Figure 6. Sensitivity analysis. Adjusted associations of 2-year changes in UACR with the risk of subsequent major clinical outcomes, using different definitions.
Supplemental Figure 7. Sensitivity analysis. Adjusted associations of 2-year changes in eGFR or UACR with the risk of subsequent major clinical outcomes, using a 30% cut-off point for the changes in eGFR and UACR.
Supplemental Figure 8. Sensitivity analysis. Adjusted associations of 2-year changes in eGFR or UACR with the risk of subsequent major clinical outcomes, after adjusting for regression to the mean, using a 40% cut-off point for the changes in eGFR and UACR.
Supplemental Figure 9. Sensitivity analysis. Adjusted associations of 2-year changes in eGFR or UACR with the risk of subsequent major clinical outcomes, after adjusting for regression to the mean, using a 10% cut-off point for the changes in eGFR and UACR.
Supplemental Figure 10. Sensitivity analysis. Adjusted associations of combination of 2-year changes in eGFR and UACR with the risk of subsequent major clinical outcomes, using a 30% cut-off point for the changes in eGFR and UACR.
Supplemental Figure 11. Sensitivity analysis. Adjusted associations of combination of 2-year changes in eGFR and UACR with the risk of subsequent major clinical outcomes, after adjusting for regression to the mean, using a 40% cut-off point for the changes in eGFR and UACR.
Supplementary Material
Acknowledgments
The Action in Diabetes and Vascular Disease: Preterax and Diamicron Modified Release Controlled Evaluation trial was funded by the grants from the National Health and Medical Research Council (NHMRC) of Australia and Servier. T.O. is supported by the John Chalmers Clinical Research Fellowship of the George Institute. M.J. is supported by a Scientia Fellowship from the University of New South Wales (Sydney, Australia). M.W. is an NHMRC of Australia Principal Research Fellow (grant 1080206).
Footnotes
Published online ahead of print. Publication date available at www.cjasn.org.
See related editorial, “A Combination of Change in Albuminuria and GFR as a Surrogate End Point for Progression of CKD,” on pages 792–794.
References
- 1.Tuttle KR, Bakris GL, Bilous RW, Chiang JL, de Boer IH, Goldstein-Fuchs J, Hirsch IB, Kalantar-Zadeh K, Narva AS, Navaneethan SD, Neumiller JJ, Patel UD, Ratner RE, Whaley-Connell AT, Molitch ME: Diabetic kidney disease: A report from an ADA Consensus Conference. Diabetes Care 37: 2864–2883, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.American Diabetes Association: Standards of medical care in diabetes, 2018. Diabetes Care 41: S1–S159, 2018 [DOI] [PubMed] [Google Scholar]
- 3.Fox CS, Matsushita K, Woodward M, Bilo HJ, Chalmers J, Heerspink HJ, Lee BJ, Perkins RM, Rossing P, Sairenchi T, Tonelli M, Vassalotti JA, Yamagishi K, Coresh J, de Jong PE, Wen CP, Nelson RG; Chronic Kidney Disease Prognosis Consortium : Associations of kidney disease measures with mortality and end-stage renal disease in individuals with and without diabetes: A meta-analysis. Lancet 380: 1662–1673, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Strippoli GF, Craig JC, Schena FP: The number, quality, and coverage of randomized controlled trials in nephrology. J Am Soc Nephrol 15: 411–419, 2004 [DOI] [PubMed] [Google Scholar]
- 5.Mol PG, Maciulaitis R, Vetter T: GFR decline as an end point for clinical trials in CKD: A view from Europe. Am J Kidney Dis 64: 838–840, 2014 [DOI] [PubMed] [Google Scholar]
- 6.Thompson A, Lawrence J, Stockbridge N: GFR decline as an end point in trials of CKD: A viewpoint from the FDA. Am J Kidney Dis 64: 836–837, 2014 [DOI] [PubMed] [Google Scholar]
- 7.Jun M, Turin TC, Woodward M, Perkovic V, Lambers Heerspink HJ, Manns BJ, Tonelli M, Hemmelgarn BR: Assessing the validity of surrogate outcomes for ESRD: A meta-analysis. J Am Soc Nephrol 26: 2289–2302, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Heerspink HJ, Kröpelin TF, Hoekman J, de Zeeuw D; Reducing Albuminuria as Surrogate Endpoint (REASSURE) Consortium : Drug-induced reduction in albuminuria is associated with subsequent renoprotection: A meta-analysis. J Am Soc Nephrol 26: 2055–2064, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Jun M, Ohkuma T, Zoungas S, Colagiuri S, Mancia G, Marre M, Matthews D, Poulter N, Williams B, Rodgers A, Perkovic V, Chalmers J, Woodward M; ADVANCE Collaborative Group : Changes in albuminuria and the risk of major clinical outcomes in diabetes: Results from ADVANCE-ON. Diabetes Care 41: 163–170, 2018 [DOI] [PubMed] [Google Scholar]
- 10.ADVANCE Management Committee : Study rationale and design of ADVANCE: Action in diabetes and vascular disease--preterax and diamicron MR controlled evaluation. Diabetologia 44: 1118–1120, 2001 [DOI] [PubMed] [Google Scholar]
- 11.Patel A, MacMahon S, Chalmers J, Neal B, Woodward M, Billot L, Harrap S, Poulter N, Marre M, Cooper M, Glasziou P, Grobbee DE, Hamet P, Heller S, Liu LS, Mancia G, Mogensen CE, Pan CY, Rodgers A, Williams B; ADVANCE Collaborative Group : Effects of a fixed combination of perindopril and indapamide on macrovascular and microvascular outcomes in patients with type 2 diabetes mellitus (the ADVANCE trial): A randomised controlled trial. Lancet 370: 829–840, 2007 [DOI] [PubMed] [Google Scholar]
- 12.Patel A, MacMahon S, Chalmers J, Neal B, Billot L, Woodward M, Marre M, Cooper M, Glasziou P, Grobbee D, Hamet P, Harrap S, Heller S, Liu L, Mancia G, Mogensen CE, Pan C, Poulter N, Rodgers A, Williams B, Bompoint S, de Galan BE, Joshi R, Travert F; ADVANCE Collaborative Group : Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N Engl J Med 358: 2560–2572, 2008 [DOI] [PubMed] [Google Scholar]
- 13.Zoungas S, Chalmers J, Neal B, Billot L, Li Q, Hirakawa Y, Arima H, Monaghan H, Joshi R, Colagiuri S, Cooper ME, Glasziou P, Grobbee D, Hamet P, Harrap S, Heller S, Lisheng L, Mancia G, Marre M, Matthews DR, Mogensen CE, Perkovic V, Poulter N, Rodgers A, Williams B, MacMahon S, Patel A, Woodward M; ADVANCE-ON Collaborative Group : Follow-up of blood-pressure lowering and glucose control in type 2 diabetes. N Engl J Med 371: 1392–1406, 2014 [DOI] [PubMed] [Google Scholar]
- 14.Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF 3rd, Feldman HI, Kusek JW, Eggers P, Van Lente F, Greene T, Coresh J; CKD-EPI (Chronic Kidney Disease Epidemiology Collaboration) : A new equation to estimate glomerular filtration rate. Ann Intern Med 150: 604–612, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ninomiya T, Perkovic V, de Galan BE, Zoungas S, Pillai A, Jardine M, Patel A, Cass A, Neal B, Poulter N, Mogensen CE, Cooper M, Marre M, Williams B, Hamet P, Mancia G, Woodward M, Macmahon S, Chalmers J; ADVANCE Collaborative Group : Albuminuria and kidney function independently predict cardiovascular and renal outcomes in diabetes. J Am Soc Nephrol 20: 1813–1821, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Matsushita K, van der Velde M, Astor BC, Woodward M, Levey AS, de Jong PE, Coresh J, Gansevoort RT; Chronic Kidney Disease Prognosis Consortium : Association of estimated glomerular filtration rate and albuminuria with all-cause and cardiovascular mortality in general population cohorts: A collaborative meta-analysis. Lancet 375: 2073–2081, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Amin AP, Whaley-Connell AT, Li S, Chen SC, McCullough PA, Kosiborod MN; KEEP Investigators : The synergistic relationship between estimated GFR and microalbuminuria in predicting long-term progression to ESRD or death in patients with diabetes: Results from the Kidney Early Evaluation Program (KEEP). Am J Kidney Dis 61[Suppl 2]: S12–S23, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hemmelgarn BR, Manns BJ, Lloyd A, James MT, Klarenbach S, Quinn RR, Wiebe N, Tonelli M; Alberta Kidney Disease Network : Relation between kidney function, proteinuria, and adverse outcomes. JAMA 303: 423–429, 2010 [DOI] [PubMed] [Google Scholar]
- 19.Turin TC, James MT, Jun M, Tonelli M, Coresh J, Manns BJ, Hemmelgarn BR: Short-term change in eGFR and risk of cardiovascular events. J Am Heart Assoc 3: e000997, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Coresh J, Turin TC, Matsushita K, Sang Y, Ballew SH, Appel LJ, Arima H, Chadban SJ, Cirillo M, Djurdjev O, Green JA, Heine GH, Inker LA, Irie F, Ishani A, Ix JH, Kovesdy CP, Marks A, Ohkubo T, Shalev V, Shankar A, Wen CP, de Jong PE, Iseki K, Stengel B, Gansevoort RT, Levey AS: Decline in estimated glomerular filtration rate and subsequent risk of end-stage renal disease and mortality. JAMA 311: 2518–2531, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Schmieder RE, Mann JF, Schumacher H, Gao P, Mancia G, Weber MA, McQueen M, Koon T, Yusuf S; ONTARGET Investigators : Changes in albuminuria predict mortality and morbidity in patients with vascular disease. J Am Soc Nephrol 22: 1353–1364, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Inker LA, Levey AS, Pandya K, Stoycheff N, Okparavero A, Greene T; Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) : Early change in proteinuria as a surrogate end point for kidney disease progression: An individual patient meta-analysis. Am J Kidney Dis 64: 74–85, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Carrero JJ, Grams ME, Sang Y, Ärnlöv J, Gasparini A, Matsushita K, Qureshi AR, Evans M, Barany P, Lindholm B, Ballew SH, Levey AS, Gansevoort RT, Elinder CG, Coresh J: Albuminuria changes are associated with subsequent risk of end-stage renal disease and mortality. Kidney Int 91: 244–251, 2017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Levey AS, Inker LA, Matsushita K, Greene T, Willis K, Lewis E, de Zeeuw D, Cheung AK, Coresh J: GFR decline as an end point for clinical trials in CKD: A scientific workshop sponsored by the National Kidney Foundation and the US Food and Drug Administration. Am J Kidney Dis 64: 821–835, 2014 [DOI] [PubMed] [Google Scholar]
- 25.Lambers Heerspink HJ, Gansevoort RT: Albuminuria is an appropriate therapeutic target in patients with CKD: The pro view. Clin J Am Soc Nephrol 10: 1079–1088, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Fried LF, Lewis J: Albuminuria is not an appropriate therapeutic target in patients with CKD: The con view. Clin J Am Soc Nephrol 10: 1089–1093, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Neal B, Perkovic V, Mahaffey KW, de Zeeuw D, Fulcher G, Erondu N, Shaw W, Law G, Desai M, Matthews DR; CANVAS Program Collaborative Group : Canagliflozin and cardiovascular and renal events in type 2 diabetes. N Engl J Med 377: 644–657, 2017 [DOI] [PubMed] [Google Scholar]
- 28.Lv J, Zhang H, Wong MG, Jardine MJ, Hladunewich M, Jha V, Monaghan H, Zhao M, Barbour S, Reich H, Cattran D, Glassock R, Levin A, Wheeler D, Woodward M, Billot L, Chan TM, Liu ZH, Johnson DW, Cass A, Feehally J, Floege J, Remuzzi G, Wu Y, Agarwal R, Wang HY, Perkovic V; TESTING Study Group : Effect of oral methylprednisolone on clinical outcomes in patients with IgA nephropathy: The TESTING randomized clinical trial. JAMA 318: 432–442, 2017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Brenner BM, Cooper ME, de Zeeuw D, Keane WF, Mitch WE, Parving HH, Remuzzi G, Snapinn SM, Zhang Z, Shahinfar S; RENAAL Study Investigators : Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N Engl J Med 345: 861–869, 2001 [DOI] [PubMed] [Google Scholar]
- 30.Appel LJ, Wright JT Jr, Greene T, Agodoa LY, Astor BC, Bakris GL, Cleveland WH, Charleston J, Contreras G, Faulkner ML, Gabbai FB, Gassman JJ, Hebert LA, Jamerson KA, Kopple JD, Kusek JW, Lash JP, Lea JP, Lewis JB, Lipkowitz MS, Massry SG, Miller ER, Norris K, Phillips RA, Pogue VA, Randall OS, Rostand SG, Smogorzewski MJ, Toto RD, Wang X; AASK Collaborative Research Group : Intensive blood-pressure control in hypertensive chronic kidney disease. N Engl J Med 363: 918–929, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.