Abstract
Synaptic plasticity, the activity dependent change in synaptic strength, forms the molecular foundation of learning and memory. Synaptic plasticity includes structural changes, with spines changing their size to accomodate insertion and removal of postynaptic receptors, allowing for functional changes. Of particular relevance for memory storage are the long lasting forms of synaptic plasticity which are protein synthesis dependent. Given the importance of spine structural plasticity and protein synthesis, this review focuses on the signaling pathways that connect synaptic stimulation with regulation of protein synthesis and remodeling of the actin cytoskeleton. We also review computational models that implement novel aspects of molecular signaling in synaptic plasticity, such as the role of neuromodulators and spatial microdomains, as well as highlight the need for computational models that connect activation of memory kinases with spine actin dynamics.
Keywords: actin, LTP, LTD, translation, synaptic tagging, computational model
1.0. Introduction
Long term synaptic plasticity is the activity-dependent, long-lasting change in synaptic strength including both strengthening, called long term potentiation (LTP) and weakening, called long term depression (LTD). This change in synaptic strength is called functional plasticity as it influences the neuron’s output. A related phenomenon is spine morphological plasticity, an activity-dependent, long-lasting change in spine size, which is correlated with functional plasticity [1]. In fact, functional plasticity often involves enlargement and remodeling of the postsynaptic density of the spine (PSD) to allow glutamate receptor insertion. Thus, for the remainder of this article we include both morphological change and functional plasticity in the terms synaptic plasticity, LTP and LTD.
The two most commonly used methods to induce synaptic plasticity include frequency dependent protocols and spike timing dependent protocols. In frequency dependent protocols [2], the frequency of stimulation determines the outcome, e.g. high frequency stimulation produces LTP and low frequency stimulation may produce LTD. Spike timing dependent plasticity (STDP) involves pre-synaptic stimulation paired with precisely timed postsynaptic action potentials [3]. The timing of the pre- and postsynaptic stimulation determines the plasticity outcome: presynaptic stimulation preceeding postsynaptic action potential induces LTP; whereas the opposite timing causes timing dependent LTD. Numerous variations on these stimulation protocols have been developed, and the frequency or timing dependence of LTP and LTD vary by brain region and cell type [4]. One striking example is the observation that high frequency stimulation elicits LTP in hippocampal CA3 to CA1 pyramidal neuron synapses [5,6] versus LTD in striatal spiny projection neurons [7]. Another example is synaptic plasticity of parallel fiber synapses to cerebellar Purkinje cells, which is opposite that of CA3 to CA1 synapses, with low frequency stimulation producing LTP and higher frequency stimulation producing LTD [8].
Long term synaptic plasticity is an interesting phenomenon because of its relationship to learning and memory storage [9,10]. Several properties of synaptic plasticity resemble properties of learning and memory, such as the persistence of both memory and long term synaptic plasticity. The specificity of synaptic plasticity allows the neuron to change its response to the stimulation protocol (specific set or sequence of activated synaptic inputs) used to induce the plasticity, with minimal change in response to a different set of activated synaptic inputs [11]. This property is analogous to stimulus specificity observed in various types of associative learning [12].
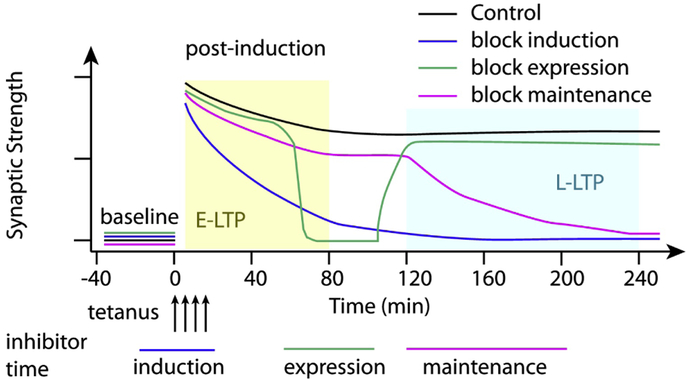
Synaptic plasticity is often subdivided into three processes: induction, expression and maintenance [13] (Fig 1). Induction refers to the brief time frame (in the range of minutes) during which the stimulation protocol is applied and also to the biochemical mechanisms activated during the stimulation that produce the plasticity measured at a later time frame. Expression refers to the measurement of the change in synaptic strength anytime after the induction period. Maintenance refers to mechanisms occurring sometime after induction, which allow the synaptic plasticity to persist.
Figure 1.
Three processes involved in L-LTP. Induction of L-LTP refers to molecules transiently activated during the stimulation protocol and is blocked by inhibitors applied during stimulation, yielding E-LTP. Maintenance of L-LTP refers to molecules persistently activated after the stimulation protocol, which can be blocked even after L-LTP has been observed. Expression refers to the molecules required for measurement of the change in synaptic strength, and re-appears after inhibitors are removed.
In addition to these three processes involved in synaptic plasticity, LTP is also subdivided into early phase (E-LTP) versus late phase (L-LTP), which differ in their underlying biochemical mechanisms. The former is a shorter lasting form of LTP, independent of protein synthesis [14]. The latter form typically endures for hours to days and is dependent on protein synthesis [15]. Induction of L-LTP is sometimes confused with maintenance of LTP. A distinguishing feature is that experiments that investigate mechanisms involved in induction of L-LTP apply inhibitor molecules during the induction period [15,16], whereas experiments that investigate mechanisms involved in maintenance of L-LTP apply inhibitor molecules as long as an hour after the induction period [17].
2.0. Signaling pathways underlying long term synaptic plasticity
Across the brain the biochemical mechanisms underlying long term synaptic plasticity differ by region, cell type, synapse type and plasticity polarity (i.e. depression versus potentiation). In this review we summarize a few of the better known forms for which some recent computational modeling papers highlight novel spatial aspects of synaptic plasticity mechanisms.
2.1. Calcium and coincidence detection
Almost all forms of synaptic plasticity at excitatory synapses are initiated by activation of glutamatergic synapses and subsequent calcium elevation [2]. In most cell types, the source of calcium is influx through NMDA receptors (e.g. hippocampal CA1, striatum, neocortex). The voltage dependence of the NMDA receptor, mediated by the magnesium block [18], provides a concise explanation of the frequency dependence and spike-timing dependence of LTP. Both high frequency stimulation and back-propagating action potentials (bAP) occuring shortly after glutamate binding cause sufficient depolarization to relieve the magnesium block and allow calcium influx through the receptor [2]. In some cases, LTD requires influx of calcium through voltage dependent calcium channels, either for frequency dependent LTD [19] or timing dependent LTD [20]. In some cell types, such as cerebellar Purkinje cells, the co-activation of metabotropic glutamate receptors and calcium influx through voltage dependent calcium channels is required for calcium release from intracellular stores [21,22].
Understanding the voltage dependence of LTP and LTD is the subject of numerous computational models [23], though only recently have spatial models begun to explain the role of inhibitory synapses, or how the timing dependence is influenced by location of the synapse on the dendrite [24]. Computational models of the striatum have demonstrated that, in the absence of GABA, NMDARs are activated during pre-post protocols that produce LTP, whereas VDCC are preferentially recruited during post-pre protocols that produce LTD [25,26]. These models illustrate that the distance dependence of STDP is caused by the back-propagating action potential. Both striatal [25,27] and neocortical [28] models show that inhibitory inputs modify the distance dependence of calcium concentration. In neocortical models, inhibitory inputs can block timing LTP or transform it to LTD depending on spatial location. In striatal models, GABA can switch the polarity of STDP both by shunting the bAP during pre-post pairings, and contributing to NMDAR depolarization during post-pre pairings.
2.2. Signaling pathways downstream of calcium
The observation that LTP and LTD both require calcium elevations has inspired numerous experimental and computational studies to investigate how calcium controls polarity of synaptic plasticity (reviewed in [29,30]). Key computational models highlight an issue with a commonly held view that high calcium concentrations produce LTP and moderate calcium elevations produce LTD. If plasticity depends solely on the calcium amplitude, STDP protocols using a long pre-then-post interval would exhibit LTD [31], which is rarely seen experimentally. Since several molecules downstream of calcium are sensitive to the dynamics of calcium elevation, a plausible refinement of that hypothesis is that direction of plasticity is determined by both amplitude and duration of calcium transients. The requirement by LTD for a calcium elevation of sufficiently long duration has experimental support (reviewed in [29]) and several computational models implement various forms of duration detection [25,31–33].
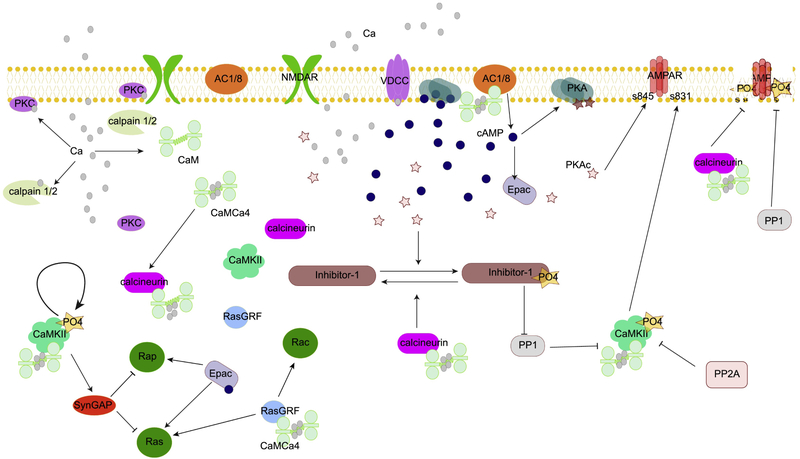
Calcium activates several key molecules that are critical for synaptic plasticity (Fig 2). The main calcium binding protein is calmodulin [34], which has numerous enzymatic and non-enzymatic targets that are required for various forms of synaptic plasticity. An important non-enzymatic target for synaptic plasticity is neurogranin [35,36]. Because neurogranin binds to apo-calmodulin with a higher affinity than to calcium-bound calmodulin, neurogranin acts as a source of calmodulin during high calcium conditions [37]. Among enzymatic targets the most prominent ones are [38,39]: calcium-calmodulin dependent protein kinase type II (CaMKII), adenylyl cyclase (AC) [40], and protein phosphatase 2B, also called calcineurin. CaMKII phosphorylates AMPA type glutamate receptor channels [41,42], producing a change in channel properties [43], as well as other molecules required for protein synthesis dependent L-LTP [44]. In contrast, calcineurin opposes some of the actions of CaMKII by dephosphorylating AMPA receptors [45]. AC produces the second messenger cAMP, which activates protein kinase A (PKA). PKA phosphorylates a number of targets, including AMPA receptor channels, increasing the number of such channels at the synapse [46]; DARPP32; and inhibitor 1, both of which inhibit protein phosphatase 1 (PP1)[47,48] to enhance net kinase activity.
Figure 2.
Calcium activates several key molecules that are critical for synaptic plasticity. Calcium directly binds to calmodulin, calpain and PKC. Calcium bound calmodulin activates adenylyl cyclase (AC1/8), CaMKII, RasGRF, and calcineurin. Adenylyl cyclase, synergistically activated by Gs coupled receptors, produces cAMP, which activates PKA and Epac. Both RasGRF and Epac activate monomeric GTP binding proteins. CaMKII, PKA and PKC all phosphorylate AMPA receptors, whereas PP1 and calcineurin dephosphorylate AMPA receptors. PKAc: catalytic subunit of PKA.
Several other calcium activated molecules play a role in various forms of synaptic plasticity. One is diacylglycerol lipase, required for LTD in many brain regions [7,49]. Diacylglycerol lipase produces the endocannabinoid 2-arachidonylgylcerol, which retrogradely targets the pre-synaptic cannabinoid type 1 receptor to reduce probability of neurotransmitter release [50]. Another calcium activated protein is protein kinase C (PKC), required for LTP in the striatum [51], LTD in the cerebellum [22,52] and LTP maintenance in the hippocampus [17]. The typical PKC isoform requires both calcium and diacylglycerol for activation [53], whereas a constitutively active form of PKC, called PKMζ, is the only kinase shown to be involved in LTP maintenance [17] and is produced either by synthesis or cleavage of other PKC forms. A third and more recently implicated calcium activated molecule is calpain, which is a protease activated by high calcium concentrations. Calpain has several targets, including molecules controlling protein synthesis [54] and actin cytoskeleton remodeling [55].
Though calcium is clearly critical for synaptic plasticity, signaling pathways downstream of G protein coupled receptors, activated by neuromodulators, are additionally required for synaptic plasticity in several brain regions, including amygdala [56], auditory cortex [57], and prefrontal cortex [58,59]. In vivo, neuromodulators are critically important as they carry information on reward, novelty, salience, emotion, and other factors that influence learning [60,61]. Although the neuromodulators dopamine, norepinephrine and acetylcholine are released in numerous brain regions, the downstream intracellular signaling molecules depend on the identity of the postsynaptic receptors and the type of triheteromeric GTP binding protein activated.
In the striatum, LTP is critically dependent on the dopamine D1 receptor in the direct pathway spiny projection neurons (SPN) and the adenosine A2A receptor in the indirect pathway SPNs [62]. Both types of receptors are coupled to Gs proteins, which lead to PKA activation, which is required for striatal LTP [51,63,64].
In the hippocampus, β-adrenergic receptor (βAR) stimulation and subsequent Gs activation is required for several forms of LTP [65]. In addition, βAR can switch their coupling from Gs to Gi [66], recruiting extracellular signal-regulated kinase types 1 and 2 (ERK) through an atypical signaling pathway. Experiments using novel βAR antagonists together with spatial stochastic modeling suggest that βAR may be required for all forms of hippocampal LTP [37,67].
Neuromodulators also are required in the neocortex. For example, STDP in visual cortex requires stimulation of both Gs and Gq coupled receptors [68,69]. Independent of spike timing, stimulation of βAR alone yields LTP, whereas stimulation of muscarinic M1 or adrenerginc α1 receptors produces LTD. Stimulation of both receptors simultaneously is required for the polarity of synaptic plasticity to be dependent on spike timing. In the prefrontal cortex dopamine is needed for both frequency dependent plasticity and STDP. Dopamine can change the temporal window of STDP [59], and with frequency dependent protocols, phasic dopamine release determines whether LTP will occur, whereas tonic dopamine controls the direction and magnitude of synaptic plasticity [70].
2.3. Models of signaling pathways: neuromodulators, spatial aspects and stochasticity
An early hypothesis proposed that information in the brain is stored in the form of molecular switches [71]: a molecular network that can enter (or exit) a long-lasting stable state in response to transient input such as calcium influx. In consequence, molecular activity is bistable with one state of low molecular activity (e.g. corresponding to no information stored or possibly corresponding to LTD) and one state of high molecular activity (e.g. corresponding to LTP). Support for this hypothesis was provided by CaMKII, a multi-subunit molecule highly expressed in the forebrain [72] and activated by calmodulin. More importantly, CaMKII can autophosphorylate itself at Thr-286 (i.e., an active CaMKII subunit can phosphorylate an adjacent calmodulin-bound subunit) preserving its activity after calcium concentration decreases and calmodulin dissociates. This property allows CaMKII to form a molecular switch even in the presence of phosphatases [73]. The ability to phosphorylate adjacent calmodulin bound subunits also conveys to CaMKII a strong dependence on frequency of calcium transients [74], which may explain how the direction of synaptic plasticity depends on stimulation frequency.
Numerous models have investigated the frequency dependence and other properties of CaMKII. Some recent models of synaptic plasticity [75–78] focus on understanding the biochemical mechanisms underlying the dependence of synaptic plasticity on stimulation frequency. These models show that with high calcium concentration (produced by high frequency stimulation), kinase activity dominates and elicits LTP, whereas for moderate calcium concentration (produced by low frequency stimulation) calmodulin preferentially binds to calcineurin and produces LTD [79]. More sophisticated models incorporate more complex interactions between CaMKII and phosphatases, such as calcineurin or PP1, as the foundation of a molecular switch [79–82]. All of these models can qualitatively reproduce results of plasticity outcomes in response to STDP and frequency dependent stimulation protocols. Some recent CaMKII models have incorporated an experiment that demonstrates activity of sub-saturated calmodulin. Though calmodulin requires four calcium ions for full activity, calmodulin bound to two calcium ions has partial activity, e.g. calmodulin bound to two calcium ions can activate CaMKII [83]. In the physiological range of calcium concentration, this sub-saturated calmodulin contributes significantly to CaMKII activation [75,76]. Since there are more calmodulin binding sites than calmodulin molecules, the competition for fully and partly calcium-bound calmodulin influences which downstream molecules are active [77]. Another question is how CaMKII could maintain its activity despite protein turnover, which would exchange a phosphorylated subunit for an unphosphorylated subunit. A recent discovery is that CaMKII holoenzymes exchange dimer subunits with unactivated holoenzymes [84,85]. A computational model [86] confirmed that CaMKII subunit exchange improves the stability of the active state in the PSD, and slows the decay of CaMKII activity in the spine cytosol, even in the presence of protein turnover. Further theoretical implications of diverse timescales of CaMKII activity in the synapse remain to be investigated.
Despite the elegance of these models, there is some question whether CaMKII actually functions as a bistable switch [87,88] and even the first models of CaMKII switching stated that only in the PSD could high CaMKII activity be maintained after calcium concentration returned to its resting value [65]. More importantly, experimental evidence, described below, shows that other molecules can play a pivotal role in synaptic plasticity, and that stochastic flucutations, molecule spatial location or spatial pattern of inputs are critical for accurate simulations of biochemical pathways in neurons.
Stochastic fluctuations are significant because the number of molecules in dendritic spines is quite small; thus the biochemical reactions underlying most forms of synaptic plasticity involve interactions between small numbers of molecules. Under these conditions, molecule concentration has less meaning, and interactions occur randomly, requiring stochastic simulation techniques. A stochastic model of the signaling pathways implicated in cerebellar plasticity [89] demonstrates the all-or-none nature of LTD in Purkinje cells and serves as an example of the critical role played by stochastic fluctuations. The switch like behavior is produced by a feedback loop involving phospholipase A2, PKC, and ERK (discussed more below). Simulations demonstate switch activation exhibits all or none behavior, with the probability of activation increasing with calcium concentration [90,91].
Research is increasingly showing that scaffolding or adaptor proteins are crucial for producing multi-protein complexes and co-localizing key molecules in specific spatial compartments [67,92,93]. Two recent models of cerebellar LTD have shown that molecules involved in AMPAR trafficking [94] or AMPAR interactions with PKC [91] can regulate LTD induction. Computational models of striatal [95] and hippocampal LTP [96] demonstrate that the role of A Kinase Anchoring Proteins is to colocalize the site of cAMP near its target PKA, due to the strong inactivation mechanisms for cAMP that prevent it from diffusing too far. These models also investigate spatial specificity of signaling models by simulating a dendrite with numerous spines. In the striatal signaling pathway models, LTP requires PKA and PKC, the latter of which binds to membrane lipids (e.g. diacylglcerol) as part of its activation; whereas LTD requires production of diffusible endocannabinoids. Model simulations reveal that activation of molecules required for LTP is mostly confined to the stimulated spines, whereas endocannabinoids are spatially diffuse, suggesting that spatially specific LTP induction would be accompanied by non-specific LTD.
2.4. Signaling pathways converge onto MAPK/ERK
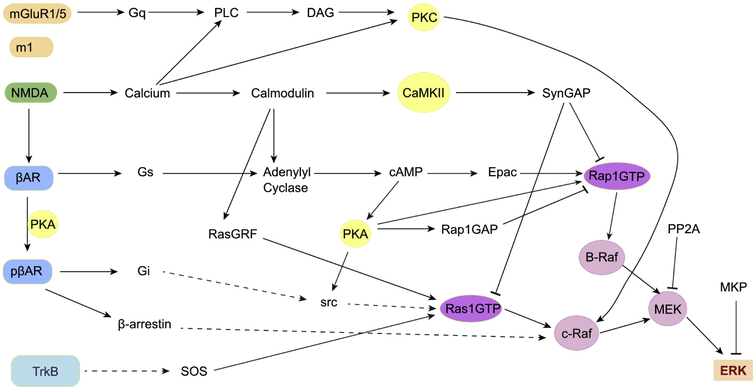
ERK, a member of the mitogen activated protein kinase (MAPK) family, seems to be the key integrative molecule required for all forms of LTP [97]. The canonical ERK pathway (Fig 3) is initiated by brain derived neurotrophic factor (BDNF) binding to a receptor tyrosine kinase (TrkB) [98], which, through several intermediary molecules, recruits a guanosine exchange factor (GEF) that activates a monomeric GTP binding protein called Ras [99]. A common motif in ERK activation is activation of a Ras family protein, followed by a series of three kinases: Ras binds to a MAP kinase kinase kinase (e.g. CRaf or BRaf) which phosphorylates a MAP kinase kinase (i.e. MEK) which then phosphorylates ERK itself [100]. In addition to BDNF activation of ERK, several other signaling pathways bypass TrkB receptors but nonetheless converge on ERK phosphorylation (Fig 3).
Figure 3.
Numerous signaling pathways contribute to ERK activation. cAMP contributes to ERK through activation of Epac and PKA. Calcium activated molecules such as RasGRF and PKC can activate ERK through activation of Ras family proteins or phosphorylation of Raf isoforms. BDNF binds to TrkB receptors and then activates Ras family proteins through several scaffolding proteins. In all cases, a series of three events activates ERK: Ras family protein binding to the kinase Raf, Raf phosphorylating MEK at two sites, and then MEK phosphorylating ERK at two sites. Dashed lines indicate that additional molecules may be intermediaries.
Several signaling pathways involving cAMP have been shown to contribute to ERK activation and play a role in synaptic plasticity. PKA can activate ERK through phosphorylation of the monomeric GTP binding protein Rap1 [101], src [102], Rap1Gap [103] or βAR [66], which then switches coupling to Gi and recruits ERK without BDNF. In addition, cAMP activates exchange factor activated by cAMP (Epac) which in turn activates Rap [104]. In Schaffer collateral synapses, Epac activation influences LTD [105]; and also contributes to βAR-dependent L-LTP [106]. Calcium activated pathways also contribute to ERK activation. CaMKII can enhance ERK through phosphorylation of SynGAP, which reduces its GTPase activity against Ras and Rap [107]. Calmodulin activates another guanosine exchange factor, called RasGRF contributing to ERK phosphorylation through Ras activation [108].
Several critical questions regarding ERK activation and spatial specificity of ERK signaling have been addressed with computational models. The first two questions address opposing aspects of spatial specificity of ERK signaling: how can spatial specificity of ERK phosphorylation be maintained with diffusible molecules and how can phospho-ERK propagate from the site of stimulation along the dendrite [109]. A model implementing PKA activation of ERK [110] showed that dendritic geometry and phosphodiesterase concentration controlled the steepness of the phospho-ERK gradient: with low phosphodiesterases, cAMP could diffuse widely, producing a small PKA gradient resulting in a small phospho-ERK gradient. A model of the feedback loop between phospho-ERK and cell excitability [111] showed that the diffusion of phospho-ERK is slow and for phospho-ERK to propagate along the dendrite (analogous to the dynamics of action potential propagation) a positive feedback loop is required: calcium leading to phospho-ERK, which then phosphorylates potassium channels increasing cell excitability and enhancing calcium influx. A later spatial deterministic model of these signaling pathways [112] showed that the same ultrasensitive behavior of ERK coupled with diffusion of upstream activators produced more ERK phosphorylation when nearby spines were stimulated in a spatial order as compared to a random order. Note that none of these models has evaluated the role of BDNF, RasGRF, SynGAP, Epac or βAR switching from Gs to Gi in ERK activation, yet experimental results suggest that the specific pathway recruiting ERK depends on stimulation pattern. Furthermore, it is likely that the different pools of ERK are activated by different signaling pathways [113,114], emphasizing the importance of modeling spatial aspects of neurons to fully understand the role played by ERK in synaptic plasticity.
3.0. From memory kinase to translation
Numerous experiments demonstrate that memory storage requires protein synthesis; thus to understand the synaptic plasticity underlying memory storage, it is necessary to elucidate how memory kinases regulate protein synthesis. Experiments have identified several points of control in both initiation and chain elongation, which are modified by ERK, CaMKII and PKMζ. ERK phosphorylates MAPK interacting kinase (MNK1), which phosphorylates several components of the critical protein complex eukaryotic initiation factor 4F (eIF4F) and S6 Kinase [115]. A second pathway regulating eIF4F is PI3 kinase, which activates Akt, which activates mTOR. Similar to ERK, PI3 kinase is activated by BDNF binding to TrkB receptors and subsequent Ras activation, and both mTOR and ERK additionally lead to phosphorylation of eukaryotic elongation factor 2 Kinase (eEF2K) which modifies the rate of chain elongation. CaMKII regulates protein synthesis through phosphorylation of cytoplasmic polyadenylation element (CPE)-binding protein (CPEB), which stimulates initiation of translation of some mRNAs in dendrites [116].
Jain and Bhalla [117] investigated how three activity dependent kinases: ERK, mTOR and CaMKIII, could control protein synthesis without producing the unstable situation of new proteins causing yet additional protein synthesis. Simulations using their carefully constructed model show that BDNF gates protein synthesis induced by LTP stimuli, and that calcium-calmodulin mediated inhibition of eEF2 (via CaMKIII) prevents more than transient increases in protein synthesis in the absence of BDNF. Another model [118] also investigates interactions between calcium activation of ERK, mTOR and eEF2K. In contrast to [117], ERK and mTOR inhibit eEF2K, whereas calcium-calmodulin directly activates eEF2K. Despite these differences, a similar conclusion is reached, that activation of these pathways produces only transient increases in protein synthesis. This second model further suggests that an additional regulator, i.e. AMP kinase, is required, demonstrating that the cell achieves tight control of protein synthesis by requiring coordinate regulation.
Several models have evaluated the biochemical mechanisms underlying the bistability of PKMζ, the constitutively active form of PKC that is responsible for LTP maintenance [17]. The candidate mechanisms include PKMζ initiating its own synthesis [119]; PKMζ autophosphorylation prolonging its residence time in synapses [120]; and phosphorylation of AMPAR, which increases AMPAR insertion in the membrane, which decreases either the degradation or diffusion of PKMζ [121]. All three mechanisms can reproduce experiments investigating time dependent effects of protein synthesis and PKMζ inhibitors. These models suggest that the PKMζ positive feedback loop is a very robust mechanism for plasticity maintenance. Only one model investigating the PKMζ feedback loop has implemented multiple spatial compartments [122] explaining mechanisms of synaptic tagging and capture [123], in which the synthesis of plasticity related proteins are targeted to stimulated spines (and not to non-stimulated spines). Simulations showed that the local, spine-specific increase in PKMζ maintains itself only in stimulated spines, whereas the concentration of PKMζ in non-stimulated spines never reaches the concentration required to switch to the high activity state.
4.0. From memory kinases to actin cytoskeleton
How is functional plasticity related to morphological plasticity? A well supported hypothesis is that LTP involves insertion of new AMPAR [124] at the PSD, into “slots” comprised of various structural and anchoring proteins, such as PSD-95. Thus, a change in spine size is needed to provide space for new AMPAR, and this requires remodeling of the branched network of actin filaments to re-build the spine actin cytoskeleton [125]. Synaptic plasticity induction protocols do indeed induce rapid remodelling of the actin cytoskeleton in the dendritic spines [126] to allow for increases in postsynaptic responses. A critical question remains unanswered: how do memory kinases or plasticity-related neuromodulators control remodeling of the actin cytoskeleton?
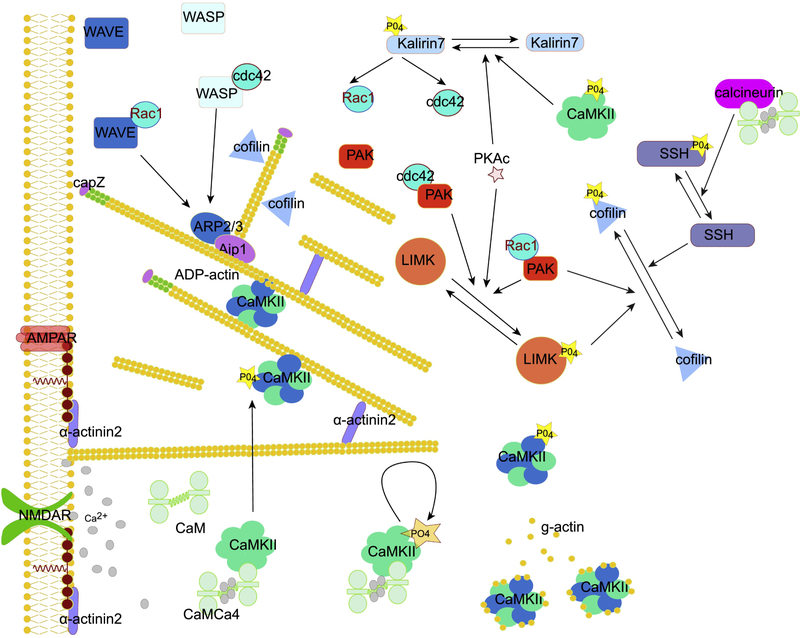
Actin filaments (f-actin) are dynamic structures, with actin monomers (known as g-actin) being added to the barbed end (polymerization) while simultaneously actin monomers are being removed from the pointed end (depolymerization) [127]. When these rates are balanced, the lengths of the actin filaments are maintained. f-actin is controlled directly by several sets of molecules including severing proteins such as cofilin, capping proteins such as capZ, branching proteins such as Arp2/3, and cross-linking/bundling proteins such as CaMKIIβ [127] (Fig 4). Both severing proteins and capping proteins accelerate f-actin disassembly: cofilin cuts the actin filament creating two shorter filaments; capZ attaches to the barbed end, and prevents addition of g-actin monomers, leading to shortening of f-actin because monomers continue to be removed from the pointed end. Paradoxically, cofilin can also promote f-actin lengthening by severing the capped actin and revealing new barbed ends to restart the polymerization process. The branching protein A complex (Arp2/3) allows for initiation of a new branch at an angle to an existing actin filament; thus it is instrumental in shaping the spine heads. Lastly, several cross-linking proteins, such as CamKIIβ and drebrin, stabilize the actin network by binding two filaments (or a filament and proteins at the PSD) and preventing binding of other actin binding proteins.
Figure 4.
Several memory kinases interact with actin binding proteins to remodel the spine actin cytoskeleton. CaMKIIβ both cross-links f-actin and buffers g-actin monomers. Binding of CaMKII to calmodulin decreases g-actin binding making g-actin monomers available and facilitating cofilin cleavage of f-actin. Both CaMKII and PKA enhance Kalirin7 activity through phosphorylation, allowing it to activate monomeric GTP binding proteins Rac1 and cdc42. Their targets include WAVE and WASP members of the ARP2/3 complex (needed for new actin branches) and PAK. Both PAK and PKA phosphorylate (to inactivate) LIMK, which phosphorylates (to inactivate) cofilin. Calcineurin dephosphorylates (to activate) SSH which then dephosphorylates (to activate) cofilin. PKAc: catalytic subunit of PKA.
How can actin filaments be dynamically stable -- maintaining their length while actin monomers are continually being added and removed? This key question was investigated by several computational models of actin dynamics (reviewed in [128]. Roland et al [129] reveal that both the concentration of actin monomers and the severing activity of cofilin are critical determinants of the steady state filament length. Berro et al. [130] demonstrate that binding of capping protein and Arp2/3 must be faster than measured in vitro to account for in vivo measurements of actin filament assembly and disassembly. Another model [131] explains spatial gradients of actin polymerization in lamelipodia of motile cells, showing that, when active WASP is confined to the leading edge of a lamelipodium, then Arp2/3 and its dependent branches exhibit elevated dissociation just behind the leading edge.
How do memory molecules remodel the actin cytoskeleton and exert control over spine morphology during and after plasticity induction? Live cell imaging has revealed transient changes in spine head concentration of several kinases and actin binding proteins during structural LTP induction [132]. The first step in remodeling the spine actin network is unbundling the actin filaments, which are normally crosslinked by unphosphorylated CamKIIβ [133], which also serves as a source of g-actin monomers. During LTP induction, CaMKIIβ is activated by calmodulin binding and CaMKIIα phosphorylation. CaMKIIβ then dissociates from g-actin making monomers available for growing the network. Dissociation of CaMKIIβ and α-actinin4, another cross-linking protein binding to actin and PSD proteins [134], allow access to actin by other actin binding proteins.
The next step after f-actin unbundling is f-actin severing by cofilin, which is controlled by LTP signaling networks through several points. Cofilin is activated during plasticity induction via dephosphorylation by slingshot phosphatase (SSH), which is a target of calcineurin. Subsequent to its activation and enhanced severing activity, cofilin is inactivated by phosphorylation by LIM Kinase (LIMK), either to allow more efficient actin branch creation (for LTP) or to stabilize the spine at a smaller size (for LTD). LIMK activity is controlled by both CaMKII and PKA, both directly and indirectly. PKA can directly phosphorylate (to activate) LIMK, and both PKA and CaMKII phosphorylate Kalirin7 and other GEFs [135,136], which in turn activate monomeric GTPases Rac1 and cdc42, which lead to LIMK phosphorylation through PAK [136].
Rac1 and cdc42 have the additional role of initiating the third part of actin remodling by activating the WAVE/WASP/IRSp35 complex directly upstream of Arp2/3 [137], which is necessary to increase polymerization and branching of the spine actin cytoskeleton. The fact that these two GTPases initiate both actin severing and actin branching suggests that the timing of activation of the different pathways is crucial for increases in spine size. A third monomeric GTPase protein, RhoA, opposing cdc42 and Rac1, activates RhoA Kinase to activate myosin [138] leading to decrease in spine size, due to the motor actions of myosin.
Following remodelling, the spine actin cytoskeleton needs to be stabilized. CaMKIIβ, inactivated due to the decrease in calcium-bound calmodulin, and α-actinin crosslink the new actin filaments. If CaMKIIβ has been phosphorylated on Thr287, then calmodulin may be trapped, and dephosphorylation would be required for crosslinking. Oligophrenin and SynGAP, both GTPase accelerating proteins enriched in the spine, inactivate Rac1 and cdc42 [139]. The importance of SynGAP has been demonstrated experimentally: spine enlargement can be mediated by CaMKII triggered SynGAP dispersion from dendritic spines, and the degree of SynGAP dispersion correlates with the maintenance of spine enlargement [140].
Even in the small volume of the spine, the spatial localization of molecules is critical. SynGAP interacts with PSD-95 to form highly concentrated liquid-like droplets representing a separation of the SynGAP/PSD-95 complex from the cytosol, reminiscent of the formation of ‘membrane-less’ intracellular compartments [141]. Localization of SynGAP to this compartment is critical for proper activity-dependent SynGAP dispersion from the PSD, AMPAR insertion and spine morphological plasticity. In addition, at the PSD CaMKII dephosphorylation may be impaired if PP1 is kept away from CaMKII [142].
Interaction between memory molecules and actin is not a one-way street: the state of the actin skeleton also influences the activation of signaling pathway molecules. Reaction rates between molecules are often limited by the diffusion rates, which control the frequency of encounter between reacting molecules. Diffusion within the cytosol can be dramatically hindered if the environment is crowded with numerous other molecules. For example, in the PSD, protein crowding can impede the diffusion of glutamate receptors [143]. Similarly, a dense actin network can impede diffusion of signaling molecules within the spine. Alternatively, molecular crowding may facilitate reactions by colocalizing reacting molecules and preventing them from diffusing away. Experimental evidence suggests that unbundling of actin transiently modifies diffusion of molecules in and out of the spine [132], with an increase in cofilin diffusion but a decrease in CaMKIIβ diffusion, and highlights the complexity of molecular interactions within a spine. Computational modeling of molecular crowding is a critical but nascent field, with new algorithms being developed to facilitate investigation [144–148].
The pioneering computational model bridging activation of signaling pathways with dynamics of the actin cytoskeleton [149] demonstrates how a single molecule, CaMKII, by virtue of different time constants for activation versus inhibition reactions, can produce LTP and a sustained increase in spine size. Extension of this model, to include control by cAMP activated pathways or to investigate heterosynaptic effects, promises to contribute greatly to our understanding of how synaptic plasticity depends on the interaction between memory kinases and the actin cytoskeleton.
5.0. Conclusions
Models of signaling pathways underlying long-term synaptic plasticity have revealed or confirmed interesting integrative and non-linear properties of diverse sets of molecules. However, more extensive modeling of the control of protein synthesis and actin reorganization is necessary to better understand the processes underlying memory storage. Increased efforts in modeling of spatial aspects of signaling pathways are especially important, such as interactions and differences between spine and dendrite, signaling from the synapse to the soma, and even emergence and function of sub-micron “membrane-less” compartments within the spine. Both experiment and theoretical studies have demonstrated that a crowded environment significantly changes both diffusion and reaction rates, which makes it crucial to include physical aspects of the spine environment such as molecular crowding in compulational models. The challenge to future models is to increase biophysical fidelity, as experiments continue to demonstrate increased complexity of neurons, while maintaining the elegance of simple models that illuminate principles of neuron function.
Acknowledgements:
This work was supported by the joint NIH-NSF CRCNS program through NIAAA grant R01AA016022, NIDA grant R01DA03889 and NSF grant 1515686.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- [1].Yuste R, Bonhoeffer T, Morphological changes in dendritic spines associated with long-term synaptic plasticity, Annu.Rev.Neurosci 24 (2001) 1071–1089. [DOI] [PubMed] [Google Scholar]
- [2].Artola A, Singer W, Long-term depression of excitatory synaptic transmission and its relationship to long-term potentiation, Trends Neurosci. 16 (1993) 480–487. [DOI] [PubMed] [Google Scholar]
- [3].Dan Y, Poo MM, Spike Timing-Dependent Plasticity: From Synapse to Perception, Physiol. Rev 86 (2006) 1033–1048. doi: 10.1152/physrev.00030.2005. [DOI] [PubMed] [Google Scholar]
- [4].Malenka RC, Bear MF, LTP and LTD: an embarrassment of riches, Neuron. 44 (2004) 5–21. doi: 10.1016/j.neuron.2004.09.012. [DOI] [PubMed] [Google Scholar]
- [5].Debanne D, Gahwiler BH, Thompson SM, Cooperative interactions in the induction of long-term potentiation and depression of synaptic excitation between hippocampal CA3-CA1 cell pairs in vitro, Proc.Natl.Acad.Sci.U.S.A 93 (1996) 11225–11230. doi: 10.1073/pnas.93.20.11225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Dudek SM, Bear MF, Bidirectional long-term modification of synaptic effectiveness in the adult and immature hippocampus., J. Neurosci 13 (1993) 2910–8. doi: 10.1038/305719a0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].Lerner TN, Kreitzer AC, RGS4 Is Required for Dopaminergic Control of Striatal LTD and Susceptibility to Parkinsonian Motor Deficits, Neuron. 73 (2012) 347–359. doi: 10.1016/j.neuron.2011.11.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [8].Jörntell H, Hansel C, Synaptic memories upside down: bidirectional plasticity at cerebellar parallel fiber-Purkinje cell synapses., Neuron. 52 (2006) 227–38. doi: 10.1016/j.neuron.2006.09.032. [DOI] [PubMed] [Google Scholar]
- [9].Abraham WC, Williams JM, Properties and Mechanisms of LTP Maintenance, Neuroscientist. 9 (2003) 463–474. doi: 10.1177/1073858403259119. [DOI] [PubMed] [Google Scholar]
- [10].Nabavi S, Fox R, Proulx CD, Lin JY, Tsien RY, Malinow R, Engineering a memory with LTD and LTP, Nature. 511 (2014) 348–352. doi: 10.1038/nature13294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [11].Engert F, Bonhoeffer T, Synapse specificity of long-term potentiation breaks down at short distances, Nature. 388 (1997) 279–284. doi: 10.1038/40870. [DOI] [PubMed] [Google Scholar]
- [12].Berger TW, Orr WB, Hippocampectomy selectively disrupts discrimination reversal conditioning of the rabbit nictitating membrane response, Behav. Brain Res 8 (1983) 49–68. doi: 10.1016/0166-4328(83)90171-7. [DOI] [PubMed] [Google Scholar]
- [13].Citri A, Malenka RC, Synaptic plasticity: Multiple forms, functions, and mechanisms, Neuropsychopharmacology. 33 (2008) 18–41. doi: 10.1038/sj.npp.1301559. [DOI] [PubMed] [Google Scholar]
- [14].Abraham WC, Williams JM, LTP maintenance and its protein synthesis-dependence, Neurobiol. Learn. Mem 89 (2008) 260–268. doi: 10.1016/j.nlm.2007.10.001. [DOI] [PubMed] [Google Scholar]
- [15].Scharf MT, Woo NH, Lattal KM, Young JZ, Nguyen PV, Abel T, Protein synthesis is required for the enhancement of long-term potentiation and long-term memory by spaced training, J Neurophysiol. 87 (2002) 2770–2777. [DOI] [PubMed] [Google Scholar]
- [16].Woo NH, Duffy SN, Abel T, Nguyen PV, Genetic and pharmacological demonstration of differential recruitment of cAMP-dependent protein kinases by synaptic activity, J.Neurophysiol 84 (2000) 2739–2745. [DOI] [PubMed] [Google Scholar]
- [17].Tsokas P, Hsieh C, Yao Y, Lesburgueres E, Wallace EJ, Tcherepanov A, Jothianandan D, Hartley BR, Pan L, Rivard B, Farese RV, Sajan MP, Bergold PJ, Hernandez AI, Cottrell JE, Shouval HZ, Fenton AA, Sacktor TC, Compensation for PKMzeta in long-term potentiation and spatial long-term memory in mutant mice, Elife. 5 (2016) 14846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [18].Nowak L, Bregestovski P, Ascher P, Herbet A, Prochiantz A, Magnesium gates glutamate-activated channels in mouse central neurones, Nature. 307 (1984) 462–465. doi: 10.1038/307462a0. [DOI] [PubMed] [Google Scholar]
- [19].Ronesi J, Lovinger DM, Induction of striatal long-term synaptic depression by moderate frequency activation of cortical afferents in rat, J.Physiol 562 (2005) 245–256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20].Fino E, Paille V, Cui Y, Morera-Herreras T, Deniau JM, Venance L, Distinct coincidence detectors govern the corticostriatal spike timing-dependent plasticity, J. Physiol 588 (2010) 3045–3062. doi: 10.1113/jphysiol.2010.188466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [21].Khodakhah K, Armstrong CM, Induction of long-term depression and rebound potentiation by insitol triphosphate in cerebellar purkinje neurons, Proc.Natl.Acad.Sci.U.S.A 94 (1997) 14009–14014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [22].Ito M, Cerebellar long-term depression: characterization, signal transduction, and functional roles, Physiol Rev. 81 (2001) 1143–1195. [DOI] [PubMed] [Google Scholar]
- [23].Foncelle A, Mendes A, Jędrzejewska-Szmek J, Valtcheva S, Berry H, Blackwell KT, Venance L, Modulation of Spike-Timing Dependent Plasticity: Towards the Inclusion of a Third Factor in Computational Models, Front. Comput. Neurosci. 12 (2018) 49. doi: 10.3389/fncom.2018.00049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [24].Letzkus JJ, Kampa BM, Stuart GJ, Learning rules for spike timing-dependent plasticity depend on dendritic synapse location, J Neurosci. 26 (2006) 10420–10429. doi: 10.1523/JNEUROSCI.2650-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [25].Jędrzejewska-Szmek J, Damodaran S, Dorman DB, Blackwell KT, Jedrzejewska-Szmek J, Damodaran S, Dorman DB, Blackwell KT, Calcium dynamics predict direction of synaptic plasticity in striatal spiny projection neurons, Eur. J. Neurosci 45 (2017) 1044–1056. doi: 10.1111/ejn.13287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [26].Paille V, Fino E, Du K, Morera-Herreras T, Perez S, Kotaleski JH, Venance L, GABAergic circuits control spike-timing-dependent plasticity, J.Neurosci 33 (2013) 9353–9363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [27].Du K, Wu Y-W, Lindroos R, Liu Y, Rózsa B, Katona G, Ding JB, Kotaleski JH, Cell-type–specific inhibition of the dendritic plateau potential in striatal spiny projection neurons, Proc. Natl. Acad. Sci 114 (2017) E7612–E7621. doi: 10.1073/pnas.1704893114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [28].Bar-Ilan L, Gidon A, Segev I, The role of dendritic inhibition in shaping the plasticity of excitatory synapses, Front. Neural Circuits 6 (2013) 1–13. doi: 10.3389/fncir.2012.00118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [29].Evans RC, Blackwell KT, Calcium: Amplitude, duration, or location?, Biol. Bull 228 (2015) 75–83. doi: 10.1086/BBLv228n1p75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [30].Graupner M, Brunel N, Mechanisms of induction and maintenance of spike-timing dependent plasticity in biophysical synapse models, Front Comput Neurosci. 4 (2010) 1–19. doi: 10.3389/fncom.2010.00136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [31].Rubin JE, Gerkin RC, Bi GQ, Chow CC, Calcium time course as a signal for spike-timing-dependent plasticity, J.Neurophysiol 93 (2005) 2600–2613. doi: 10.1152/jn.00803.2004. [DOI] [PubMed] [Google Scholar]
- [32].Kumar A, Mehta MR, Frequency-Dependent Changes in NMDAR-Dependent Synaptic Plasticity, Front Comput. 5 (2011) 38. doi: 10.3389/fncom.2011.00038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [33].Graupner M, Brunel N, Calcium-based plasticity model explains sensitivity of synaptic changes to spike pattern, rate, and dendritic location, Proc.Natl.Acad.Sci.U.S.A 109 (2012) 3991–3996. doi: 10.1073/pnas.1109359109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [34].Faas GC, Raghavachari S, Lisman JE, Mody I, Calmodulin as a direct detector of Ca2+ signals, Nat. Neurosci 14 (2011) 301–304. doi: 10.1038/nn.2746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [35].Zhong L, Cherry T, Bies CE, Florence MA, Gerges NZ, Neurogranin enhances synaptic strength through its interaction with calmodulin., EMBO J. 28 (2009) 3027–39. doi: 10.1038/emboj.2009.236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [36].Hoffman L, Chandrasekar A, Wang X, Putkey JA, Waxham MN, Neurogranin alters the structure and calcium binding properties of calmodulin, J.Biol.Chem 289 (2014) 14644–14655. doi: 10.1074/jbc.M114.560656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [37].Jędrzejewska-Szmek J, Luczak V, Abel T, Blackwell KT, β-adrenergic signaling broadly contributes to LTP induction, PLoS Comput. Biol 13 (2017) e1005657. doi: 10.1371/journal.pcbi.1005657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [38].Lisman J, Yasuda R, Raghavachari S, Mechanisms of CaMKII action in long-term potentiation, Nat.Rev.Neurosci 13 (2012) 169–182. doi: 10.1038/nrn3192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [39].Mulkey RM, Endo S, Shenolikar S, Malenka RC, Involvement of a calcineurin/inhibitor-1 phosphatase cascade in hippocampal long-term depression, Nature. 369 (1994) 486–488. [DOI] [PubMed] [Google Scholar]
- [40].Masada N, Ciruela A, Macdougall DA, Cooper DM, Distinct mechanisms of regulation by Ca2+/calmodulin of type 1 and 8 adenylyl cyclases support their different physiological roles, J Biol Chem. 284 (2009) 4451–4463. doi:M807359200 [pii]\n 10.1074/jbc.M807359200 [doi]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [41].Roche KW, O’Brien RJ, Mammen AL, Bernhardt J, Huganir RL, Characterization of multiple phosphorylation sites on the AMPA receptor GluR1 subunit, Neuron. 16 (1996) 1179–1188. [DOI] [PubMed] [Google Scholar]
- [42].Barria A, Muller D, Derkach V, Griffith LC, Soderling TR, Regulatory phosphorylation of AMPA-type glutamate receptors by CaM-KII during long-term potentiation, Science. 276 (1997) 2042–2044. [DOI] [PubMed] [Google Scholar]
- [43].Derkach V, Barria A, Soderling TR, Ca2+/calmodulin-kinase II enhances channel conductance of alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionate type glutamate receptors., Proc. Natl. Acad. Sci. U. S. A 96 (1999) 3269–74. doi: 10.1073/pnas.96.6.3269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [44].Redondo RL, Okuno H, Spooner PA, Frenguelli BG, Bito H, Morris RGM, Synaptic Tagging and Capture: Differential Role of Distinct Calcium/Calmodulin Kinases in Protein Synthesis-Dependent Long-Term Potentiation, J. Neurosci 30 (2010) 4981–4989. doi: 10.1523/JNEUROSCI.3140-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [45].Lee HK, Kameyama K, Huganir RL, Bear MF, NMDA induces long-term synaptic depression and dephosphorylation of the GluR1 subunit of AMPA receptors in hippocampus, Neuron. 21 (1998) 1151–1162. [DOI] [PubMed] [Google Scholar]
- [46].Esteban JA, Shi SH, Wilson C, Nuriya M, Huganir RL, Malinow R, PKA phosphorylation of AMPA receptor subunits controls synaptic trafficking underlying plasticity, Nat. Neurosci 6 (2003) 136–143. doi: 10.1038/nn997. [DOI] [PubMed] [Google Scholar]
- [47].Svenningsson P, Nishi A, Fisone G, Girault JA, Nairn AC, Greengard P, DARPP-32: an integrator of neurotransmission, Annu.Rev.Pharmacol.Toxicol 44 (2004) 269–296. [DOI] [PubMed] [Google Scholar]
- [48].Nairn AC, Hemmings HC Jr., Walaas SI, Greengard P, DARPP-32 and phosphatase inhibitor-1, two structurally related inhibitors of protein phosphatase-1, are both present in striatonigral neurons, J.Neurochem 50 (1988) 257–262. [DOI] [PubMed] [Google Scholar]
- [49].Kishimoto Y, Cagniard B, Yamazaki M, Nakayama J, Sakimura K, Kirino Y, Kano M, Task-specific enhancement of hippocampus-dependent learning in mice deficient in monoacylglycerol lipase, the major hydrolyzing enzyme of the endocannabinoid 2-arachidonoylglycerol, Front. Behav. Neurosci 9 (2015) 134. doi: 10.3389/fnbeh.2015.00134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [50].Gerdeman GL, Ronesi J, Lovinger DM, Postsynaptic endocannabinoid release is critical to long-term depression in the striatum, Nat.Neurosci 5 (2002) 446–451. doi: 10.1038/nn832. [DOI] [PubMed] [Google Scholar]
- [51].Hawes SL, Gillani F, Evans RC, Benkert EA, Blackwell KT, Sensitivity to theta-burst timing permits LTP in dorsal striatal adult brain slice, J.Neurophysiol 110 (2013) 2027–2036. doi: 10.1152/jn.00115.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [52].Hirono M, Sugiyama T, Kishimoto Y, Sakai I, Miyazawa T, Kishio M, Inoue H, Nakao K, Ikeda M, Kawahara S, Kirino Y, Katsuki M, Horie H, Ishikawa Y, Yoshioka T, Phospholipase Cbeta4 and protein kinase Calpha and/or protein kinase CbetaI are involved in the induction of long term depression in cerebellar Purkinje cells, J.Biol.Chem 276 (2001) 45236–45242. [DOI] [PubMed] [Google Scholar]
- [53].Huang K-P, The mechanism of protein kinase C activation, Trends Neurosci. 12 (1989) 425–432. doi: 10.1016/0166-2236(89)90091-X. [DOI] [PubMed] [Google Scholar]
- [54].Briz V, Hsu Y-T, Li Y, Lee E, Bi X, Baudry M, Calpain-2-Mediated PTEN Degradation Contributes to BDNF-Induced Stimulation of Dendritic Protein Synthesis, J. Neurosci 33 (2013) 4317–4328. doi: 10.1523/JNEUROSCI.4907-12.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [55].Baudry M, Zhu G, Liu Y, Wang Y, Briz V, Bi X, Multiple cellular cascades participate in long-term potentiation and in hippocampus-dependent learning, Brain Res. 1621 (2015) 73–81. doi: 10.1016/j.brainres.2014.11.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [56].Laing M, Bashir ZI, β-Adrenoceptors and synaptic plasticity in the perirhinal cortex, Neuroscience. 273 (2014) 163–173. doi: 10.1016/j.neuroscience.2014.04.070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [57].Shepard KN, Liles LC, Weinshenker D, Liu RC, Norepinephrine is necessary for experience-dependent plasticity in the developing mouse auditory cortex., J. Neurosci 35 (2015) 2432–7. doi: 10.1523/JNEUROSCI.0532-14.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [58].Zhou HC, Sun YY, Cai W, He XT, Yi F, Li BM, Zhang XH, Activation of β2-adrenoceptor enhances synaptic potentiation and behavioral memory via cAMP-PKA signaling in the medial prefrontal cortex of rats, Learn. Mem 20 (2013) 274–284. doi: 10.1101/lm.030411.113. [DOI] [PubMed] [Google Scholar]
- [59].Ruan H, Saur T, Yao W-D, Dopamine-enabled anti-Hebbian timing-dependent plasticity in prefrontal circuitry, Front. Neural Circuits 8 (2014) 38. doi: 10.3389/fncir.2014.00038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [60].Sara SJ, The locus coeruleus and noradrenergic modulation of cognition, Nat. Rev. Neurosci 10 (2009) 211–223. doi: 10.1038/nrn2573. [DOI] [PubMed] [Google Scholar]
- [61].Bromberg-Martin ES, Matsumoto M, Hikosaka O, Dopamine in Motivational Control: Rewarding, Aversive, and Alerting, Neuron. 68 (2010) 815–834. doi: 10.1016/j.neuron.2010.11.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [62].Pawlak V, Wickens JR, Kirkwood A, Kerr JN, Timing is not Everything: Neuromodulation Opens the STDP Gate, Front Synaptic.Neurosci 2 (2010) 146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [63].Ghiglieri V, Napolitano F, Pelosi B, Schepisi C, Migliarini S, Di Maio A, Pendolino V, Mancini M, Sciamanna G, Vitucci D, Maddaloni G, Giampà C, Errico F, Nisticò R, Pasqualetti M, Picconi B, Rhes influences striatal cAMP / PKA-dependent signaling and synaptic plasticity in a gender-sensitive fashion, Sci. Rep 5 (2015) 10933. doi: 10.1038/srep10933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [64].Yagishita S, Hayashi-Takagi A, Ellis-Davies GC, Urakubo H, Ishii S, Kasai H, A critical time window for dopamine actions on the structural plasticity of dendritic spines, Science. 345 (2014) 1616–1620. doi: 10.1126/science.1255514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [65].Hagena H, Hansen N, Manahan-vaughan D, β -Adrenergic Control of Hippocampal Function : Subserving the Choreography of Synaptic Information Storage and Memory, Cereb.Cortex (2018) 1349–1364. doi: 10.1093/cercor/bhv330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [66].Martin NP, Whalen EJ, Zamah MA, Pierce KL, Lefkowitz RJ, PKA-mediated phosphorylation of the beta1-adrenergic receptor promotes Gs/Gi switching, Cell Signal. 16 (2004) 1397–1403. [DOI] [PubMed] [Google Scholar]
- [67].Havekes R, Canton DA, Park AJ, Huang T, Nie T, Day JP, Guercio LA, Grimes Q, Luczak V, Gelman IH, Baillie GS, Scott JD, Abel T, Gravin orchestrates protein kinase A and beta2-adrenergic receptor signaling critical for synaptic plasticity and memory, J.Neurosci 32 (2012) 18137–18149. doi: 10.1523/JNEUROSCI.3612-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [68].Salgado H, Köhr G, Trevĩo M, Noradrenergic tone determines dichotomous control of cortical spike-timing-dependent plasticity, Sci. Rep 2 (2012) 417. doi: 10.1038/srep00417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [69].Huang S, Rozas C, Trevino M, Contreras J, Yang S, Song L, Yoshioka T, Lee H-K, Kirkwood A, Associative Hebbian Synaptic Plasticity in Primate Visual Cortex, J. Neurosci 34 (2014) 7575–7579. doi: 10.1523/JNEUROSCI.0983-14.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [70].Otani S, Daniel H, Roisin MP, Crepel F, Dopaminergic modulation of long-term synaptic plasticity in rat prefrontal, Cereb Cortex. 13 (2003) 1251–1256. [DOI] [PubMed] [Google Scholar]
- [71].Crick F, Memory and molecular turnover., Nature. 312 (n.d.) 101 http://www.ncbi.nlm.nih.gov/pubmed/6504122 (accessed December 19, 2018). [DOI] [PubMed] [Google Scholar]
- [72].Erondu NE, Kennedy MB, Regional distribution of type II Ca2+/calmodulin-dependent protein kinase in rat brain., J. Neurosci 5 (1985) 3270–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [73].Lisman JE, A mechanism for memory storage insensitive to molecular turnover: a bistable autophosphorylating kinase., Proc. Natl. Acad. Sci. U. S. A 82 (1985) 3055–7. http://www.ncbi.nlm.nih.gov/pubmed/2986148 (accessed December 18, 2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- [74].De Koninck P, Schulman H, Sensitivity of CaM Kinase II to the Frequency of Ca2+ Oscillations, Science. 279 (1998) 227–230. doi: 10.1126/science.279.5348.227. [DOI] [PubMed] [Google Scholar]
- [75].Li X, Holmes WR, Biophysical attributes that affect CaMKII activation deduced with a novel spatial stochastic simulation approach, PLoS Comput. Biol 14 (2018) e1005946. doi: 10.1371/journal.pcbi.1005946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [76].Pepke S, Kinzer-Ursem T, Mihalas S, Kennedy MB, A dynamic model of interactions of Ca2+, calmodulin, and catalytic subunits of Ca2+/calmodulin-dependent protein kinase II, PLoS Comput. Biol 6 (2010). doi: 10.1371/journal.pcbi.1000675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [77].Romano DR, Pharris MC, Patel NM, Kinzer-ursem TL, Competitive tuning : Competition ‘ s role in setting the frequency-dependence of Ca 2 + -dependent proteins, PLoS Comput. 13 (2017) e1005820. doi: 10.4231/R7154F7Q.PLOS. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [78].Li L, Stefan MI, Le Novere N, Calcium Input Frequency, Duration and Amplitude Differentially Modulate the Relative Activation of Calcineurin and CaMKII, PLoS One. 7 (2012). doi: 10.1371/journal.pone.0043810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [79].Castellani GC, Quinlan EM, Cooper LN, Shouval HZ, A biophysical model of bidirectional synaptic plasticity: dependence on AMPA and NMDA receptors, Proc.Natl.Acad.Sci.U.S.A 98 (2001) 12772–12777. doi: 10.1073/pnas.201404598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [80].Carlson KD, Giordano N, Interplay of the magnitude and time-course of postsynaptic Ca 2+ concentration in producing spike timing-dependent plasticity, J. Comput. Neurosci 30 (2011) 747–758. doi: 10.1007/s10827-010-0290-z. [DOI] [PubMed] [Google Scholar]
- [81].Pi HJ, Lisman JE, Coupled Phosphatase and Kinase Switches Produce the Tristability Required for Long-Term Potentiation and Long-Term Depression, J. Neurosci 28 (2008) 13132–13138. doi: 10.1523/JNEUROSCI.2348-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [82].Lisman JE, Zhabotinsky AM, A model of synaptic memory: a CaMKII/PP1 switch that potentiates transmission by organizing an AMPA receptor anchoring assembly, Neuron. 31 (2001) 191–201. [DOI] [PubMed] [Google Scholar]
- [83].Shifman JM, Choi MH, Mihalas S, Mayo SL, Kennedy MB, Ca2+/calmodulin-dependent protein kinase II (CaMKII) is activated by calmodulin with two bound calciums, Proc. Natl. Acad. Sci 103 (2006) 13968–13973. doi: 10.1073/pnas.0606433103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [84].Bhattacharyya M, Stratton MM, Going CC, McSpadden ED, Huang Y, Susa AC, Elleman A, Cao YM, Pappireddi N, Burkhardt P, Gee CL, Barros T, Schulman H, Williams ER, Kuriyan J, Molecular mechanism of activation-triggered subunit exchange in Ca(2+)/calmodulin-dependent protein kinase II., Elife. 5 (2016) e13405. doi: 10.7554/eLife.13405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [85].Stratton M, Lee I-H, Bhattacharyya M, Christensen SM, Chao LH, Schulman H, Groves JT, Kuriyan J, Activation-triggered subunit exchange between CaMKII holoenzymes facilitates the spread of kinase activity, Elife. 3 (2014) e01610. doi: 10.7554/eLife.01610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [86].Singh D, Bhalla US, Subunit exchange enhances information retention by CaMKII in dendritic spines, Elife. 7 (2018) e41412. doi: 10.7554/eLife.41412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [87].Michalski PJ, The delicate bistability of CaMKII, Biophys. J 105 (2013) 794–806. doi: 10.1016/j.bpj.2013.06.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [88].Chang JY, Parra-Bueno P, Laviv T, Szatmari EM, Lee SJR, Yasuda R, CaMKII Autophosphorylation Is Necessary for Optimal Integration of Ca2+Signals during LTP Induction, but Not Maintenance, Neuron. 94 (2017) 800–808.e4. doi: 10.1016/j.neuron.2017.04.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [89].Tanaka K, Augustine GJ, A positive feedback signal transduction loop determines timing of cerebellar long-term depression, Neuron. 59 (2008) 608–620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [90].Antunes G, De Schutter E, A Stochastic Signaling Network Mediates the Probabilistic Induction of Cerebellar Long-Term Depression, J. Neurosci 32 (2012) 9288–9300. doi: 10.1523/JNEUROSCI.5976-11.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [91].Hepburn I, Jain A, Gangal H, Yamamoto Y, A Model of Induction of Cerebellar Long-Term Depression Including RKIP Inactivation of Raf and MEK, 10 (2017) 1–11. doi: 10.3389/fnmol.2017.00019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [92].Sanderson JL, Gorski JA, Dell’Acqua ML, NMDA Receptor-Dependent LTD Requires Transient Synaptic Incorporation of Ca2+-Permeable AMPARs Mediated by AKAP150-Anchored PKA and Calcineurin, Neuron. 89 (2016) 1000–1015. doi: 10.1016/j.neuron.2016.01.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [93].Zhang M, Patriarchi T, Stein IS, Qian H, Matt L, Nguyen M, Xiang YK, Hell JW, Adenylyl cyclase anchoring by a kinase anchor protein AKAP5 (AKAP79/150) is important for postsynaptic β-adrenergic signaling, J. Biol. Chem 288 (2013) 17918–17931. doi: 10.1074/jbc.M112.449462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [94].Gallimore AR, Aricescu AR, Yuzaki M, Calinescu R, A Computational Model for the AMPA Receptor Phosphorylation Master Switch Regulating Cerebellar Long-Term Depression, PLoS Comput. Biol 12 (2016). doi: 10.1371/journal.pcbi.1004664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [95].Oliveira RF, Kim M, Blackwell KT, Subcellular Location of PKA Controls Striatal Plasticity: Stochastic Simulations in Spiny Dendrites, PLoS Comput Biol. 8 (2012) e1002383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [96].Kim M, Park AJ, Havekes R, Chay A, Guercio LA, Oliveira RF, Abel T, Blackwell KT, Colocalization of protein kinase a with adenylyl cyclase enhances protein kinase a activity during induction of Long-Lasting Long-Term-Potentiation, PLoS Comput. Biol 7 (2011) e1002084. doi: 10.1371/journal.pcbi.1002084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [97].Thomas GM, Huganir RL, MAPK cascade signalling and synaptic plasticity, Nat. Rev. Neurosci 5 (2004) 173–183. doi: 10.1038/nrn1346. [DOI] [PubMed] [Google Scholar]
- [98].Klein R, Nanduri V, Jing SA, Lamballe F, Tapley P, Bryant S, Cordon-Cardo C, Jones KR, Reichardt LF, Barbacid M, The trkB tyrosine protein kinase is a receptor for brain-derived neurotrophic factor and neurotrophin-3., Cell. 66 (1991) 395–403. http://www.ncbi.nlm.nih.gov/pubmed/1649702 (accessed December 18, 2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- [99].Nimnual AS, Yatsula BA, Bar-Sagi D, Coupling of Ras and Rac guanosine triphosphatases through the Ras exchanger Sos., Science. 279 (1998) 560–3. [DOI] [PubMed] [Google Scholar]
- [100].English J, Pearson G, Wilsbacher J, Swantek J, Karandikar M, Xu S, Cobb MH, New Insights into the Control of MAP Kinase Pathways, Exp. Cell Res 253 (1999) 255–270. doi: 10.1006/excr.1999.4687. [DOI] [PubMed] [Google Scholar]
- [101].Takahashi M, Dillon TJ, Liu C, Kariya Y, Wang Z, Stork PJS, Protein kinase a-dependent phosphorylation of Rap1 regulates its membrane localization and cell migration, J. Biol. Chem 288 (2013) 27712–27723. doi: 10.1074/jbc.M113.466904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [102].Li Y, Dillon TJ, Takahashi M, Earley KT, Stork XPJS, Protein Kinase A-independent Ras Protein Activation Cooperates with Rap1 Protein to Mediate Activation of the Extracellular Signal-regulated Kinases ( ERK ) by cAMP *, J. Biol. Chem 291 (2016) 21584–21595. doi: 10.1074/jbc.M116.730978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [103].McAvoy T, Zhou MM, Greengard P, Nairn AC, Phosphorylation of Rap1GAP, a striatally enriched protein, by protein kinase A controls Rap1 activity and dendritic spine morphology, Proc.Natl.Acad.Sci.U.S.A 106 (2009) 3531–3536. doi: 10.1073/pnas.0813263106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [104].De Rooij J, Zwartkruis FJT, Verheijen MHG, Cool RH, Nijman SMB, Wittinghofer A, Bos JL, Epac is a Rap1 guanine- nucleotide-exchange factor directly activated by cyclic AMP, Nature. 396 (1998) 474. [DOI] [PubMed] [Google Scholar]
- [105].Lee K, Kobayashi Y, Seo H, Kwak J-H, Masuda A, Lim C-S, Lee H-R, Kang SJ, Park P, Sim S-E, Kogo N, Kawasaki H, Kaang B-K, Itohara S, Involvement of cAMP-guanine nucleotide exchange factor II in hippocampal long-term depression and behavioral flexibility, Mol. Brain 8 (2015) 38. doi: 10.1186/s13041-015-0130-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [106].Gelinas JN, Banko JL, Peters MM, Klann E, Weeber EJ, Nguyen PV, Activation of exchange protein activated by cyclic-AMP enhances long-lasting synaptic potentiation in the hippocampus, Learn.Mem 15 (2008) 403–411. doi: 10.1101/lm.830008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [107].Walkup WG, Washburn L, Sweredoski MJ, Carlisle HJ, Graham RL, Hess S, Kennedy MB, Phosphorylation of synaptic GTPase-activating protein (synGAP) by Ca2+/Calmodulin-dependent protein kinase II (CaMKII) and cyclin- dependent kinase 5 (CDK5) alters the ratio of its GAP activity toward ras and rap GTPases, J. Biol. Chem 290 (2015) 4908–4927. doi: 10.1074/jbc.M114.614420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [108].Stacey D, Bilbao A, Maroteaux M, Jia T, Easton AC, Longueville S, Nymberg C, Banaschewski T, Barker GJ, Buchel C, Carvalho F, Conrod PJ, Desrivieres S, Fauth-Buhler M, Fernandez-Medarde A, Flor H, Gallinat J, Garavan H, Bokde ALW, Heinz A, Ittermann B, Lathrop M, Lawrence C, Loth E, Lourdusamy A, Mann KF, Martinot J-L, Nees F, Palkovits M, Paus T, Pausova Z, Rietschel M, Ruggeri B, Santos E, Smolka MN, Staehlin O, Jarvelin M-R, Elliott P, Sommer WH, Mameli M, Muller CP, Spanagel R, Girault J-A, Schumann G, RASGRF2 regulates alcohol-induced reinforcement by influencing mesolimbic dopamine neuron activity and dopamine release, Proc. Natl. Acad. Sci 109 (2012) 21128–21133. doi: 10.1073/pnas.1211844110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [109].Wiegert JS, Bengtson CP, Bading H, Diffusion and not active transport underlies and limits ERK1/2 synapse-to-nucleus signaling in hippocampal neurons, J. Biol. Chem 282 (2007) 29621–29633. doi: 10.1074/jbc.M701448200. [DOI] [PubMed] [Google Scholar]
- [110].Neves SR, Tsokas P, Sarkar A, Grace EA, Rangamani P, Taubenfeld SM, Alberini CM, Schaff JC, Blitzer RD, Moraru II, Iyengar R, Cell shape and negative links in regulatory motifs together control spatial information flow in signaling networks, Cell. 133 (2008) 666–680. doi: 10.1016/j.cell.2008.04.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [111].Ajay SM, Bhalla US, A propagating ERKII switch forms zones of elevated dendritic activation correlated with plasticity, HFSP.J 1 (2007) 49–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [112].Bhalla US, Synaptic input sequence discrimination on behavioral timescales mediated by reaction-diffusion chemistry in dendrites, Elife. 6 (2017) e25827. doi: 10.7554/eLife.25827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [113].Shalin SC, Hernandez CM, Dougherty MK, Morrison DK, Sweatt JD, Kinase suppressor of Ras1 compartmentalizes hippocampal signal transduction and subserves synaptic plasticity and memory formation, Neuron. 50 (2006) 765–779. [DOI] [PubMed] [Google Scholar]
- [114].Zhang L, Zhang P, Wang G, Lin L, Huganir RL, Zhu JJ, Ras and Rap Signal Bidirectional Synaptic Plasticity via Distinct Subcellular Microdomains, Neuron. 98 (2018) 783–800. doi: 10.1016/j.neuron.2018.03.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [115].Panja D, Bramham CR, BDNF mechanisms in late LTP formation: A synthesis and breakdown, Neuropharmacology. 76 (2014) 664–676. doi: 10.1016/j.neuropharm.2013.06.024. [DOI] [PubMed] [Google Scholar]
- [116].Atkins CM, Davare MA, Oh MC, Derkach V, Soderling TR, Bidirectional regulation of cytoplasmic polyadenylation element-binding protein phosphorylation by Ca2+/calmodulin-dependent protein kinase II and protein phosphatase 1 during hippocampal long-term potentiation, J.Neurosci 25 (2005) 5604–5610. doi: 10.1523/JNEUROSCI.5051-04.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [117].Jain P, Bhalla US, Signaling logic of activity-triggered dendritic protein synthesis: an mTOR gate but not a feedback switch, PLoS.Comput.Biol 5 (2009) e1000287. doi: 10.1371/journal.pcbi.1000287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [118].Kenney JW, Sorokina O, Genheden M, Sorokin A, Armstrong JD, Proud XCG, Dynamics of Elongation Factor 2 Kinase Regulation in Cortical Neurons in Response to Synaptic Activity, 35 (2015) 3034–3047. doi: 10.1523/JNEUROSCI.2866-14.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [119].Ogasawara H, Kawato M, The protein kinase M ζ network as a bistable switch to store neuronal memory, BMC Syst. Biol 4 (2010) 181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [120].Jalil SJ, Sacktor TC, Shouval HZ, Atypical PKCs in memory maintenance : the roles of feedback and redundancy, (2015) 344–353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [121].Helfer P, Shultz TR, Coupled feedback loops maintain synaptic long-term potentiation : A computational model of PKMzeta synthesis and AMPA receptor trafficking, PLoS Comput. 14 (2018) e1006147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [122].Smolen P, Baxter DA, Byrne JH, Molecular constraints on synaptic tagging and maintenance of long-term potentiation: a predictive model, PLoS.Comput.Biol 8 (2012) e1002620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [123].Frey U, Morris RGM, Weak before strong: Dissociating synaptic tagging and plasticity-factor accounts of late-LTP, Neuropharmacology. 37 (1998) 545–552. doi: 10.1016/S0028-3908(98)00040-9. [DOI] [PubMed] [Google Scholar]
- [124].Lüscher C, Frerking M, Restless AMPA receptors: Implications for synaptic transmission and plasticity, Trends Neurosci. 24 (2001) 665–70. doi: 10.1016/S0166-2236(00)01959-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [125].Saneyoshi T, Hayashi Y, The ca 2+ and rho gtpase signaling pathways underlying activity-dependent actin remodeling at dendritic spines, Cytoskeleton. 69 (2012) 545–554. doi: 10.1002/cm.21037. [DOI] [PubMed] [Google Scholar]
- [126].Okamoto K, Nagai T, Miyawaki A, Hayashi Y, Rapid and persistent modulation of actin dynamics regulates postsynaptic reorganization underlying bidirectional plasticity, Nat.Neurosci 7 (2004) 1104–1112. [DOI] [PubMed] [Google Scholar]
- [127].Pollard TD, Actin and Actin-Binding Proteins, Cold Spring Harb.Perspect.Biol 8 (2016) a018226. doi: 10.1101/cshperspect.a018226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [128].Danuser G, Allard J, Mogilner A, Mathematical modeling of eukaryotic cell migration: insights beyond experiments, Annu. Dev.Biol 29 (2013) 501–528. doi: 10.1146/annurev-cellbio-101512-122308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [129].Roland J, Berro J, Michelot A, Blanchoin L, Martie JL, Stochastic severing of actin filaments by actin depolymerizing factor/cofilin controls the emergence of a steady dynamical regime, Biophys. J 94 (2008) 2082–2094. doi: 10.1529/biophysj.107.121988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [130].Berro J, Sirotkin V, Pollard TD, Mathematical Modeling of Endocytic Actin Patch Kinetics in Fission Yeast: Disassembly Requires Release of Actin Filament Fragments, Mol. Biol. Cell 21 (2010) 2905–15. doi: 10.1091/mbc.E10-06-0494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [131].Ditlev JA, Mayer BJ, Loew LM, There is more than one way to model an elephant. Experiment-driven modeling of the actin cytoskeleton, Biophys. J 104 (2013) 520–532. doi: 10.1016/j.bpj.2012.12.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [132].Bosch M, Castro J, Saneyoshi T, Matsuno H, Sur M, Hayashi Y, Structural and molecular remodeling of dendritic spine substructures during long-term potentiation, Neuron. 82 (2014) 444–459. doi: 10.1016/j.neuron.2014.03.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [133].Hoffman L, Farley MM, Waxham MN, Calcium-calmodulin-dependent protein kinase II isoforms differentially impact the dynamics and structure of the actin cytoskeleton, Biochemistry. 52 (2013) 1198–1207. doi: 10.1021/bi3016586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [134].Borovac J, Bosch M, Okamoto K, Regulation of actin dynamics during structural plasticity of dendritic spines : Signaling messengers and actin-binding proteins, Mol. Cell. Neurosci 91 (2018) 122–130. doi: 10.1016/j.mcn.2018.07.001. [DOI] [PubMed] [Google Scholar]
- [135].Nadella KS, Saji M, Jacob NK, Pavel E, Ringel MD, Kirschner LS, Regulation of actin function by protein kinase A-mediated phosphorylation of Limk1, EMBO Rep. 10 (2009) 599–605. doi: 10.1038/embor.2009.58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [136].Xie Z, Srivastava DP, Photowala H, Kai L, Cahill ME, Woolfrey KM, Shum CY, Surmeier DJ, Penzes P, Kalirin-7 controls activity-dependent structural and functional plasticity of dendritic spines, Neuron. 56 (2007) 640–656. doi: 10.1016/j.neuron.2007.10.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [137].Leung DW, Otomo C, Chory J, Rosen MK, Genetically encoded photoswitching of actin assembly through the Cdc42-WASP-Arp2/3 complex pathway, Proc. Natl. Acad. Sci 105 (2008) 12797–12802. doi: 10.1073/pnas.0801232105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [138].Govek E, Newey SE, Van Aelst L, The role of the Rho GTPases in neuronal development The role of the Rho GTPases in neuronal development, Genes Dev. 19 (2005) 1–49. doi: 10.1101/gad.1256405. [DOI] [PubMed] [Google Scholar]
- [139].Nakano-Kobayashi A, Tai Y, Nadif Kasri N, Van Aelst L, The X-linked Mental Retardation Protein OPHN1 Interacts with Homer1b/c to Control Spine Endocytic Zone Positioning and Expression of Synaptic Potentiation, J. Neurosci 34 (2014) 8665–8671. doi: 10.1523/JNEUROSCI.0894-14.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [140].Araki Y, Zeng M, Zhang M, Huganir RL, Rapid Dispersion of SynGAP from Synaptic Spines Triggers AMPA Receptor Insertion and Spine Enlargement during LTP, Neuron. 85 (2015) 173–190. doi: 10.1016/j.neuron.2014.12.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [141].Zeng M, Shang Y, Araki Y, Guo T, Huganir RL, Zhang M, Phase Transition in Postsynaptic Densities Underlies Formation of Synaptic Complexes and Synaptic Plasticity., Cell. 166 (2016) 1163–1175.e12. doi: 10.1016/j.cell.2016.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [142].Mullasseril P, Dosemeci A, Lisman JE, Griffith LC, A structural mechanism for maintaining the “on-state” of the CaMKII memory switch in the post-synaptic density, J.Neurochem 103 (2007) 357–364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [143].Li TP, Song Y, MacGillavry HD, Blanpied TA, Raghavachari S, Protein Crowding within the Postsynaptic Density Can Impede the Escape of Membrane Proteins., J. Neurosci 36 (2016) 4276–95. doi: 10.1523/JNEUROSCI.3154-15.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [144].Gupta R, Self-crowding of AMPA receptors in the excitatory postsynaptic density can effectuate anomalous receptor sub-diffusion., PLoS Comput. Biol 14 (2018) e1005984. doi: 10.1371/journal.pcbi.1005984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [145].Meinecke L, Multiscale Modeling of Diffusion in a Crowded Environment., Bull. Math. Biol 79 (2017) 2672–2695. doi: 10.1007/s11538-017-0346-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [146].Ellery AJ, Baker RE, Simpson MJ, An analytical method for disentangling the roles of adhesion and crowding for random walk models on a crowded lattice., Phys. Biol 13 (2016) 05LT02. doi: 10.1088/1478-3975/13/5/05LT02. [DOI] [PubMed] [Google Scholar]
- [147].Marquez-Lago TT, Leier A, Burrage K, Anomalous diffusion and multifractional Brownian motion: simulating molecular crowding and physical obstacles in systems biology., IET Syst. Biol 6 (2012) 134–42. doi: 10.1049/iet-syb.2011.0049. [DOI] [PubMed] [Google Scholar]
- [148].Andrews SS, Smoldyn: particle-based simulation with rule-based modeling, improved molecular interaction and a library interface., Bioinformatics. 33 (2017) 710–717. doi: 10.1093/bioinformatics/btw700. [DOI] [PubMed] [Google Scholar]
- [149].Rangamani P, Levy MG, Khan S, Oster G, Paradoxical signaling regulates structural plasticity in dendritic spines, Proc. Natl. Acad. Sci 113 (2016) E5298–E5307. doi: 10.1073/pnas.1610391113. [DOI] [PMC free article] [PubMed] [Google Scholar]