Abstract
Background
Transcatheter aortic valve-in-valve implantation (aVIV) has been used to treat bioprosthetic failure due to "stent creep", defined as inward flexion or bending of stent posts. The aim of this study was to develop quantitative three-dimensional transesophageal echocardiography (3D-TEE) geometric analysis of failed bioprostheses to determine the incidence of stent creep in patients undergoing aVIV and its contribution to the hemodynamics of those valves.
Methods
We retrospectively examined the 3D-TEE of 22 consecutive patients (age 74.4 ± 11.3 years; M/F = 12/10) who underwent aVIV for failed bioprostheses. The modes of bioprosthesis failure included stenosis (n = 8), regurgitation (n = 9), and combined (n = 5). The degree of stent creep was assessed by calculating the triangular area obtained by projecting the apex of stent posts on a reconstructed plane. This measured area was divided by that of the regular triangle defined by the base of stent posts to calculate a ratio, which we termed the "stent creep ratio" (SCR).
Results
The mean SCR was lower in the patients with failed prostheses than that in the controls (0.82 ± 0.16 vs. 0.96 ± 0.05, p = 0.02). The SCR was negatively correlated with the peak trans-aortic pressure gradient (r = -0.62, p < 0.01). An SCR cut-off point of 0.79 was associated with aortic peak velocity > 4 m/s (AUC = 0.81, sensitivity = 0.79, specificity = 0.83). Fourteen of the 22 patients had pre- and post-aVIV 3D-TEE, and the SCR was corrected satisfactorily from 0.81 ± 0.13 to 1.04 ± 0.19 (p < 0.01).
Conclusions
SCR measured by 3D-TEE is feasible to quantitatively evaluate stent creep. Stent creep is an important mode of structural deterioration in surgical bioprostheses, which can be treated by aVIV.
Keywords: Aortic valve-in-valve, Bioprosthetic valve dysfunction, Stent creep ratio
INTRODUCTION
Stent creep (SC), defined as inward bending of a stent post of stented bioprosthetic valves, was first identified in explanted Hancock valves, and was considered to be a possible etiology for intrinsic valvular stenosis.1 This led to the development of newer stent materials that are more resistant to such deformity. Currently, the diagnosis of SC is usually made from subjective observations of inward bending on echocardiography or fluoroscopy. However, due to the lack of feasible non-invasive quantification methods to assess the severity of SC, the clinical incidence of SC and its impacts on modern stented bioprostheses are unclear. Even though clinically significant SC is considered to be a rare type of bioprosthetic degeneration, it has recently been reported in some cases to be the only cause of prosthetic dysfunction, even in modern bioprostheses.7 As a result, there is an unmet need to develop a non-invasive quantification tool to diagnose SC.
In recent years, the durability of bioprostheses has been greatly improved, but it remains limited. Because of the relatively high surgical risk of redoing valve placement, some patients with failing bioprostheses are treated conservatively. Fortunately, as transcatheter aortic valvular implantation successfully improves the outcomes in patients with high-risk aortic stenosis and broaden its indication toward those with an intermediate risk,2,3 the transcatheter aortic valve-in-valve (aVIV) procedure has also emerged as a feasible and reasonable solution for those who havea high surgical risk of redo surgery.4-7 On the other hand, high-resolution three-dimensional transesophageal echocardiography (3D-TEE) is now a widely applied tool to assist in making decisions, guide procedures, and evaluate outcomes during transcatheter procedures.8 This provides an opportunity for in-vivo observations of the structure and function of failed bioprostheses.
The aim of the present study was to develop a quantitative 3D-TEE based geometric analysis of SC among failed bioprostheses to determine the percentage of SC in patients undergoing aVIV and its contribution to the hemodynamics of those valves.
METHOD
Patient population (Table 1)
Table 1. Characteristics of aortic valve-in-valve for individual patient.
| Surgical valve brand/size (mm) | AVR or DVR | Post OP years | LogEuro | PPG, mmHg | MPG, mmHg | Failure type | Cusp pathology, P = prolapse, T = thickening | LVEF, % | SCRpre | SCRpost | THV | THV size, mm | ||
| 1 | Edwards S.A.V | 25 | AVR | 5 | 26.97 | 28 | 15 | 2 | P | 62 | 0.87 | 1.001 | CoreValve | 26 |
| 2 | Medtronic Hancock II | 25 | AVR | 12 | 22.34 | 55 | 34 | 3 | P+T | 62 | 0.64 | 1.269 | CoreValve | 26 |
| 3 | Edwards Standard | 21 | DVR | 12 | 42.11 | 76 | 43 | 1 | 53 | 0.60 | 0.933 | CoreValve | 26 | |
| 4 | Edwards S.A.V | 21 | AVR | 8 | 7.98 | 35 | 16 | 2 | P | 62 | 1.01 | 1.007 | CoreValve | 23 |
| 5 | Edwards perimount magna | 19 | DVR | 4 | 5.48 | 51 | 32 | 1 | 62 | 0.79 | 1.570 | CoreValve | 23 | |
| 6 | Edwards perimount magna | 23 | AVR | 3 | 13.64 | 23 | 12 | 2 | P | 62 | 0.82 | 1.078 | CoreValve | 26 |
| 7 | Medtronic Hancock II | 21 | AVR | 11 | 32.84 | 57 | 30 | 1 | T | 62 | 0.81 | 1.009 | CoreValve | 23 |
| 8 | Medtronic Hancock II | 25 | AVR | 11 | 20.57 | 26 | 16 | 2 | P+T | 25 | 0.79 | 0.986 | Sapien XT | 23 |
| 9 | Medtronic Hancock II | 23 | AVR | 13 | 40.32 | 50 | 25 | 2 | P+T | 23 | 0.76 | / | CoreValve | 23 |
| 10 | SJ. Epic | 21 | DVR | 6 | 14.84 | 56 | 29 | 3 | P | 59 | 0.69 | 0.763 | CoreValve | 23 |
| 11 | Edwards perimount magna | 25 | DVR | 3 | 4.84 | 16 | 9 | 2 | 50 | 0.80 | 0.918 | Sapien XT | 26 | |
| 12 | Edwards perimount | 21 | DVR | 7 | 30.49 | 41 | 25 | 1 | T | 35 | 1.07 | 1.002 | Sapien XT | 23 |
| 13 | Medtronic Hancock II | 25 | AVR | 12 | 37.06 | 28 | 16 | 2 | P | 62 | 1.09 | / | Sapien XT | 23 |
| 14 | Edwards perimount | 21 | AVR | 7 | 5.85 | 75 | 41 | 1 | 62 | 0.77 | / | Sapien XT | 23 | |
| 15 | Edwards Standard | 21 | DVR | 8 | 21.21 | 64 | 36 | 1 | 59 | 0.76 | 1.153 | Sapien XT | 23 | |
| 16 | Edwards perimount | 21 | DVR | 7 | 19.02 | 35 | 17 | 1 | T | 59 | 1.05 | / | Evolut R | 23 |
| 17 | Edwards S.A.V | 21 | AVR | 14 | 12.87 | 74 | 42 | 3 | P+T | 62 | 0.86 | / | Evolut R | 23 |
| 18 | Trifecta | 21 | AVR | 4 | 72.13 | 12 | 7 | 2 | P | 60 | 1.00 | / | Sapien XT | 23 |
| 19 | Edwards S.A.V | 19 | DVR | 12 | 12.54 | 82 | 52 | 3 | P+T | 64 | 0.63 | / | Evolut R | 23 |
| 20 | Edwards perimount magna | 19 | DVR | 4 | 16.17 | 53 | 32 | 3 | 66 | 0.62 | 0.921 | Evolut R | 23 | |
| 21 | Edwards perimount magna | 19 | DVR | 4 | 23.82 | 31 | 17 | 2+PVL | P | 40 | 0.96 | 0.960 | Evolut R | 23 |
| 22 | Edwards S.A.V | 27 | AVR | 10 | 4.84 | 127 | 78 | 1 | 73 | 0.70 | / | Sapien 3 | 26 |
AVR, aortic valve replacement; DVR, double valve replacement; LVEF, left ventricular ejection fraction; MPG, mean pressure gradient; PPG, peak pressure gradient; SCRpost, stent creep ratio after aortic valve-in-valve treatment; SCRpre, stent creep ratio before aortic valve-in-valve treatment; THV, transcatheter heart valve.
Failure type: 1 = stenosis, 2 = regurgitation, 3 = combined.
We retrospectively examined the 3D-TEE of 24 consecutive patients undergoing aVIV for failed bioprostheses. One homograft and 1 stentless valve were excluded due to essentially different valvular designs. The remaining 22 patients (age 74.4 ± 11.3 years; M/F = 12/10; log Euroscore = 22.2 ± 16.0) all had aortic valves replaced with stented bioprostheses, including 15 Edward valves (standard = 2, supra-annular valve = 5, perimount = 3, magna = 5), 5 Medtronic Handcock II valves, 1 Sj. Epic valve and 1 Trifecta valve. Ten patients had double valve replacement (DVR) (aortic and mitral), and 6 patients (case no. 3, 5, 10, 15, 19 and 20) had rheumatic heart disease (RHD). The modes of bioprosthetic failure included stenosis [n = 8 (36%)], regurgitation [n = 9 (41%)], and combined [n = 5 (23%)]. Seven newly implanted bioprostheses in patients who underwent surgical aortic valve replacement were also evaluated by 3D-TEE for comparison (Table 2). The study design, imaging retrieval and analyses were all approved by the ethics committee of our hospital.
Table 2. Characteristics of control group that just accepted surgical AVR.
| Surgical valve brand/size (mm) | Reason for AVR | SCR | PPG, mmHg | MPG, mmHg | ||
| 1 | Perimount magna ease | 25 | Regurgitation | 0.92 | 20 | 10 |
| 2 | Edwards perimount | 23 | Regurgitation | 0.98 | 10 | 6 |
| 3 | SJ medical | 21 | Stenosis | 1.00 | 16 | 9 |
| 4 | Perimount magna ease | 23 | Stenosis | 0.98 | 22 | 13 |
| 5 | Perimount magna | 21 | Regurgitation | 0.88 | 25 | 14 |
| 6 | Perimount magna ease | 23 | Stenosis | 1.02 | 18 | 10 |
| 7 | Perimount magna ease | 23 | Stenosis | 0.96 | 21 | 12 |
Abbreviations as in Table 1.
Three-dimensional transesophageal echocardiography
The 3D-TEE data were acquired using an EPIQ 7C system equipped with an X7-2t transducer (Philips Medical Systems, Andover, MA), and all of the patients underwent endotracheal intubation under general anesthesia. After optimization of the midesophageal long axis view, the studied bioprosthetic valve was enclosed into the 3D Zoom mode for further analysis. The multi-beat acquisition mode was selected to achieve a higher frame rate (> 25 Hz) in the patients with a regular rhythm.
Methods of quantifying the severity of SC
Stented valves are designed with three stent posts to provide commissural support for each cusp. As the stent posts are perpendicular to the annular plane, the two triangles defined by three tips and three bottoms of the stent posts should be congruent regular triangles. Once inward bending occurs in one or more of the stent posts, the top triangle will shrink. As a result the ratio between top and bottom triangles (i.e. stent creep ratio, SCR) will become less than one (Figure 1A).
Figure 1.
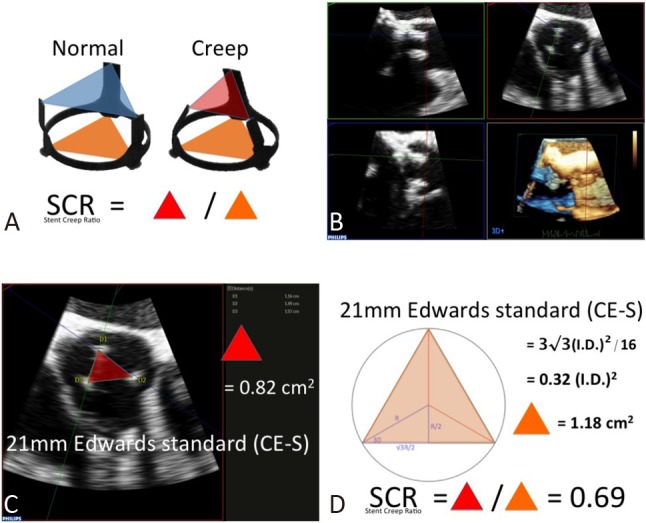
Calculation of stent creep ratio (SCR). (A) Ideally, three stent posts are perpendicular to the annular ring. As a results, the area of tip-triangle (blue) should equal the bottom-triangle (orange). Once stent creeping (SC) develops, tip-triangle shrinks and the ratio of tip- to bottom-triangle will be less than 1.0. (B) By three-dimensional transesophageal echocardiography and multi-planar reconstruction technique, the three tips of stent posts could be identified. (c) A case of SC. The tip-triangle area is calculated as 0.82 cm2 by Heron’s formula. (D) Bottom triangle is calculated basing the inner diameter of 21 mm Edwards standard (CE-S) bioprosthesis. The SCR is 0.82/1.18 = 0.69.
We applied 3D-TEE to perform an in-vivo geometric analysis for SCR. Multi-planar reconstruction was performed offline with Qlab10 software to identify each stent tip of the top triangle (Figure 1B). Because Qlab10 software can only provide the area of "smoothed polygons", the top triangular area was calculated according to Heron’s formula, which uses the length of three sides to yield a triangular area (Figure 1C). Length measurements were conducted by one experienced echo lab technician who was blinded to the clinical scenario and brands of bioprostheses. We defined the bottom triangle as the largest regular triangle inscribed by a circle with a diameter equal to the internal diameter of the stented bioprosthesis. As a result, the bottom triangular area was calculated as 3√3 ÷·16 (internal diameter)2 (Figure 1D).
Statistical methods
Continuous variables are expressed as mean ± standard deviation (SD). On the basis of their distributions, continuous variables were compared using the Student’s t test or the Mann-Whitney U test. Pearson’s correlation tests were performed to determine correlations between SCR and clinical parameters. Receiver operating characteristic (ROC) curves were constructed to determine the optimal cutoff values of chosen variables and the area under the curve (AUC). All statistical analyses were performed using SPSS Statistics for Windows version 17.0 (IBM SPSS, Chicago, IL). All reported p values were two tailed, and p values < 0.05 were considered to indicate statistical significance.
RESULTS
Relationship between SCR and aortic prosthetic dysfunction
SCR was significantly lower in the failed prostheses than in the controls (0.82 ± 0.16 vs. 0.96 ± 0.05, p = 0.02) and negatively correlated with the trans-aortic peak pressure gradient (r = -0.62, p < 0.01) and mean pressure gradient (r = -0.54, p = 0.03) (Table 1). An SCR cut-off point of 0.79 was associated with aortic peak velocity > 4 m/s (Figure 2: AUC = 0.81, sensitivity = 0.79, specificity = 0.83). Patients in the stenosis and combined groups had a lower SCR (0.76 ± 0.16) in comparison with the regurgitation group (0.89 ± 0.12) (p = 0.10). Nine of the 22 (41%) patients had an SCR lower than 0.79.
Figure 2.
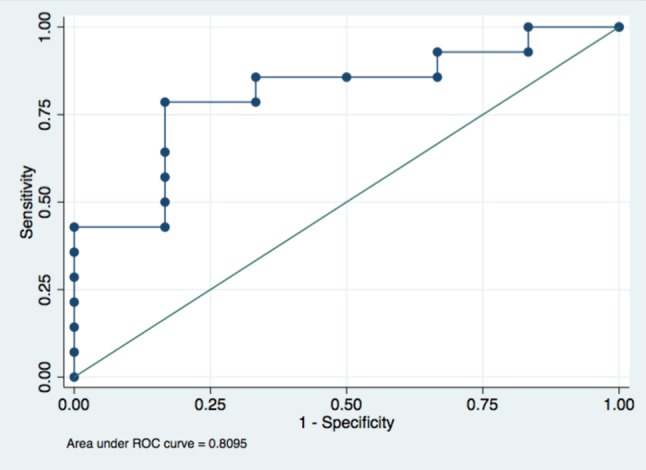
Predicting peak aortic pressure gradient > 4 m/s with SCR by receiver operating characteristic curve analysis.
Relationship between SCR and previous surgical AVR
SCR was not associated with the size of surgical valve (r = 0.14, p = 0.55) or the duration of implantation (r = -0.13, p = 0.57). There was no significant difference in SCR between the patients with DVR and a single AVR (SCR: 0.81 vs. 0.85, p = 0.56). The patients with a medical history of RHD had a lower SCR than the others (SCR: 0.70 vs. 0.87, p = 0.02).
Changes in SCR after aVIV
Six aVIV procedures were performed via the transapical approach and 16 via the transfemoral approach, and 11 CoreValve (2 needed a second valve), 5 Evolut R, 7 Sapien XT and 1 Sapien 3 were deployed. Fourteen of the 22 patients had pre- and post-aVIV 3D-TEE, and the SCR was corrected satisfactorily (Figure 3) from 0.81 ± 0.13 to 1.04 ± 0.19 (p < 0.01).
Figure 3.
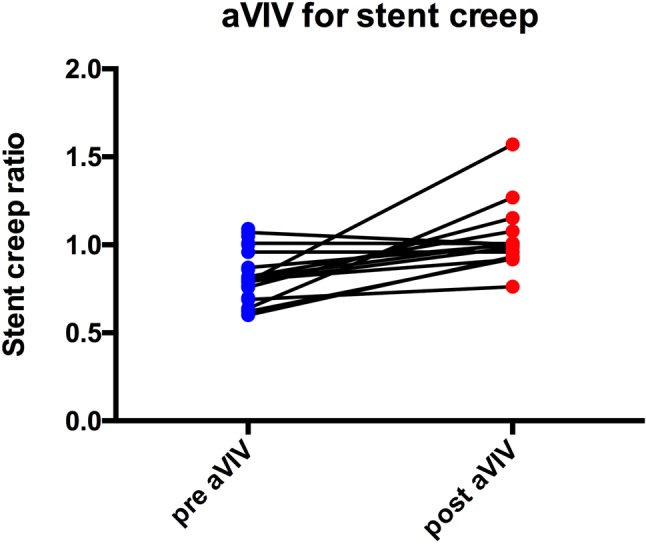
SCR of 14 failed bioprostheses before and after aVIV.
DISCUSSION
To the best of our knowledge, the current study is the first to non-invasively quantify SC in dysfunctional aortic bioprostheses. We found that SC was not an uncommon phenomenon in our aVIV population, among whom 41% had significant SC that was associated with an increased transaortic pressure gradient. This abnormal deformity was not time-related and could occur in valves with either stenosis or regurgitation failure. We attempted to define a possible cut-off value of SCR for clinically significant SC using ROC analysis, and we found that a SCR < 0.79 was associated with an increased transaortic flow rate > 4 m/s.
Comparison between SCR and other methods of quantifying creeping severity
SC was previously measured from explanted valves using quantitative methods such as inward bending angle of stent posts, projected outflow area ratio, and reduction of tip-projected triangular area.1,9,10 Measurements of inward bending angle from conventional chest X-rays may be skewed and can only be conducted in radiopaque stents. The interpretation of inward bending angle of each stent post can be even more difficult, as the correlation between stent post bending and valvular dysfunction is only modest in the aortic position.11 In large bioprosthetics, a valve with 15° inward bending of stent posts in-vitro could still have normal function.9 The projected outflow area and tip-projected triangular area are methods based on geometric changes in SC.10 However, some unknown factors during surgical removal may affect the severity of SC. Our method of SCR is similar to the idea of tip-projected triangular area. With 3D-TEE, we could measure the three sides of the tip-triangle using on-cart software and calculate the area using Heron’s formula. The main differences between SCR and Valente’s method10 are to diminish measurement errors and to simplify the steps. We used the mathematically derived area of a regular triangle on the annular level as the denominator to calculate SCR. Our results showed that an SCR < 0.79 was compatible with moderate deformity in Valente’s study. With an ideal acoustic window, our method could also be applied to 3D-TTE to derive SCR for longitudinal follow-up. However, the horizontal direction of aortic bioprostheses may hide one stent post behind the post-acoustic shadow of another post, especially on TTE where the tips of the stent post would be an inverted triangle (i.e. two-acoustic shadows over the lower stent post). Fortunately, our method could also be applied to multiple detector computed tomography images for patients in whom SC is highly suspected. We defined the tips in the center of echogenicity rather than inner edges, so that the erroneous effects of blooming artifacts could be minimized. The bottom triangle was calculated from reported stent inner diameters rather than true inner diameters, and such artifacts could be further reduced with new generations of transesophageal transducers.
SC and etiologies of bioprosthetic dysfunction
SC in the mitral position has previously been reported to not be related to the materials or designs of the stent post, but it tends to increase with time, and compression of the left ventricular wall is considered to be an important cause of mitral SC.9 In the aortic position, our results are consistent with previous reports, in that SC was not related to the time after implantation. Possible etiologies of SC in the aortic position include: 1. small aortic root implanted with a relatively large bioprosthesis, 2. traction of the stents due to overlying cloth-tissue ingrowth, 3. incompetence to diastolic flow, and 4. mechanical fatigue of the stent materials. In this study, 9 (41%) patients had a SCR < 0.79. Among these cases, prolapse and thickened cusps were noted in 4, which may provide evidence for the idea of tissue traction and diastolic impact force related to SC. However, the other 5 cases (case no. 3, 14, 15, 20 and 22) had no obvious calcification or fusion on cusps, and the increased pressure gradient could be only considered to be SC related. Of note, 3 (case no. 3, 15 and 20) of the 5 cases had undergone DVR for RHD before, which may support small aortic root-related SC. Even though Elgiloy materials have previously been reported to be more resistant to SC, we still observed 6 cases of significant creep with Elgiloy stents, even with the new generation of design.
Valve-in-valve
Transcatheter valve-in-valve (VIV) treatment has emerged as an alternative solution for bioprosthetic dysfunction, and it has been used in both the aortic position and pulmonic, mitral and tricuspid positions. In addition to bioprosthetic dysfunction of stenosis and regurgitation, VIV has also been applied to paravalvular leak and SC. Attia et al. identified three patients with pure SC-related prosthetic aortic stenosis, and demonstrated good responses to aVIV with Sapien XT.7 They proposed that balloon-expandable devices are particularly applicable for aVIV in SC. Even though the CoreValve system may have difficulty in device localization, the retrievable and repositionable Evolut R may be able to overcome this issue. Its supra-annular design may also provide some hemodynamic benefits. In the current study, 10 patients with significant SC (SCR < 0.8) were treated with 6 self-expandable (4 CoreValve, 2 Evolut R) and 4 balloon expandable (3 Sapien XT and 1 Sapien 3) THVs. Aortic pressure gradient and SCR were improved by aVIV with both types of devices. Similar to Attia’s work, 5 patients in our aVIV population had stenosis without obvious degenerative changes of the bioprostheses, however these 5 valves all had significant SC with an SCR from 0.60 to 0.79. Under fluoroscopy, balloon pre-dilatation helped splay open the inward bending stent posts, and THV implantation further provided resistance to the recoil force.
Limitations
There are several limitations to the present study. First, because our method is based on 3D-TEE, we did not have structural degeneration or pathological information that could be only provided by explanted valves. As a result, the causal relationship between SC and the aforementioned pathologic changes could not be determined in this retrospective study. However, as mentioned above, common etiologies of prosthetic degeneration may to some extent contribute to SC. This is also the reason why the SCR from 3D-TEE was only modestly correlated with trans-aortic peak pressure gradient. As artifacts after surgery may hinder detailed evaluation of degenerative prosthetic cusps, an SCR < 0.79 may serve as a common final endpoint of significant hemodynamic changes.
Second, four failed prostheses had a pre-aVIV SCR value > 1.0, which may raise concerns over accuracy of the measurements, however splay out of the stent post and deviation of the MPR plane during analysis could also have led to overestimation of the top triangular area and led to an increased SCR value. In addition, SC is only one of the possible etiologies of prosthetic stenosis, and it is possible to have a failed prosthesis with a normal SCR.
Third, because the original purpose of post-aVIV 3D-TEE of the aortic valve area is to examine the sphericity of the TAVI valve and increased artifacts after deploying the second valve, in some cases we could not calculate the SCR. Further studies are needed to verify the feasibility of our method in post-aVIV status.
Finally, the sample size was relatively small. Even though we demonstrated some degree of SC in patients undergoing aVIV, further large-scale investigations on the actual incidence of SC among bioprostheses are needed.
CONCLUSIONS
SCR measured by 3D-TEE is feasible to identify and quantitatively evaluate SC. We found that a SCR < 0.79 was associated with an increase in trans-aortic pressure gradient. SC is not an uncommon mode of structural deterioration in surgical bioprostheses, and it can be treated with aVIV. Further large-scale, prospective studies are warranted to investigate the value of SCR as a surrogate parameter or early sign of bioprosthetic dysfunction.
FUNDING
There is no funding for this study.
DECLARATION OF CONFLICT OF INTEREST
All the authors declare no conflict of interest.
REFERENCES
- 1.Schoen FJ, Schulman LJ, Cohn LH. Quantitative anatomic analysis of "stent creep" of explanted Hancock standard porcine bioprostheses used for cardiac valve replacement. Am J Cardiol. 1985;56:110–114. doi: 10.1016/0002-9149(85)90576-4. [DOI] [PubMed] [Google Scholar]
- 2.Chen YH, Chang HH, Chen PL, et al. Procedural characteristics and outcomes of transcatheter aortic valve implantation: a single-center experience of the first 100 inoperable or high surgical risk patients with severe aortic stenosis. Acta Cardiol Sin. 2017;33:339–349. doi: 10.6515/ACS20170620A. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Yin WH. Transcatheter aortic valve implantation in Taiwan: still evolving! Acta Cardiol Sin. 2017;33:350. doi: 10.6515/ACS20170707A. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Eggebrecht H, Schäfer U, Treede H, et al. Valve-in-valve transcatheter aortic valve implantation for degenerated bioprosthetic heart valves. JACC Cardiovasc Interv. 2011;4:1218–1227. doi: 10.1016/j.jcin.2011.07.015. [DOI] [PubMed] [Google Scholar]
- 5.Gurvitch R, Cheung A, Ye J, et al. Transcatheter valve-in-valve implantation for failed surgical bioprosthetic valves. J Am Coll Cardiol. 2011;58:2196–2209. doi: 10.1016/j.jacc.2011.09.009. [DOI] [PubMed] [Google Scholar]
- 6.Bapat V, Attia R, Redwood S, et al. Use of transcatheter heart valves for a valve-in-valve implantation in patients with degenerated aortic bioprosthesis: technical considerations and results. J Thorac Cardiovasc Surg. 2012;144:1372–1380. doi: 10.1016/j.jtcvs.2012.07.104. [DOI] [PubMed] [Google Scholar]
- 7.Attia R, Papalexopoulou N, Hancock J, et al. Successful treatment of failing biological prosthesis because of "stent creep" with valve-in-valve transcatheter aortic valve implantation: successful treatment of failing biological prosthesis with V-in-V TAVI. Catheter Cardiovasc Interv. 2015;86:E119–E125. doi: 10.1002/ccd.25452. [DOI] [PubMed] [Google Scholar]
- 8.Hahn RT, Nicoara A, Kapadia S, et al. Echocardiographic imaging for transcatheter aortic valve replacement. J Am Soc Echocardiogr Off Publ Am Soc Echocardiogr. 2018;31:405–433. doi: 10.1016/j.echo.2017.10.022. [DOI] [PubMed] [Google Scholar]
- 9.Akiyama K, Sawatani O, Imamura E, et al. Stent creep of porcine bioprosthesis in the mitral position. Ann Thorac Surg. 1988;46:73–78. doi: 10.1016/s0003-4975(10)65857-2. [DOI] [PubMed] [Google Scholar]
- 10.Valente M, Bortolotti U, Thiene G, et al. Post bending of the polypropylene flexible stent in mitral Hancock bioprostheses. Eur J Cardiothorac Surg. 1987;1:134–138. doi: 10.1016/1010-7940(87)90028-5. [DOI] [PubMed] [Google Scholar]
- 11.Hurlé A, Nistal JF, Revuelta JM. Stent fracture in Wessex porcine heart valve bioprostheses. Artif Organs. 1997;21:116–120. doi: 10.1111/j.1525-1594.1997.tb00347.x. [DOI] [PubMed] [Google Scholar]


