Abstract
Sudden cardiac arrest is the leading cause of cardiovascular mortality, posing a substantial public health burden. The incidence and epidemiology of sudden death are a function of age, with primary arrhythmia syndromes and inherited cardiomyopathies representing the predominant causes in younger patients, while coronary artery disease being the leading etiology in those who are 35 years of age and older. Internal cardioverter defibrillators remain the mainstay of primary and secondary prevention of sudden cardiac arrest. In the acute phase, cardiac chain of survival, early reperfusion, and therapeutic hypothermia are the key steps in improving outcomes. In the chronic settings, ventricular tachycardia ablation has been shown to improve patients’ quality of life by reducing frequency of defibrillator shocks. Moreover, recent studies have suggested that it may increase survival. Neuromodulation represents a novel therapeutic modality that has a great potential for improving treatment of ventricular arrhythmias.
Keywords: cardiac arrest, sudden cardiac death, ventricular tachycardia, ventricular arrhythmia, ablation, neuromodulation
INTRODUCTION
Sudden cardiac death (SCD) is defined as unexpected death due to cardiac causes, occurring within 1 hour from the onset of symptoms in individuals with no known prior conditions that would limit their lifespan. Autopsy studies demonstrate that approximately 85% of all unexplained SCDs are of cardiac etiology. SCD is the most common outcome of sudden cardiac arrest (SCA) and the leading cause of death worldwide. The annual rate of SCD in the United States is estimated to be on average 230,000–350,000.(1,2) Two-thirds of all cases of SCD occur in men. Overall, a decline in the rate of SCD, both in men and women, has been attributed to the success of primary and secondary prevention therapies and other competing causes of death.(2) SCD presents a tremendous public health burden due it its substantial fatality and short time from the onset of symptoms to death. SCD accounts for 50% of all cardiovascular mortality, being the first presentation of cardiac disease in 25–50% of cases.(1) Survival of individuals who experience SCA remains disappointing, estimated at 6–10% for outof-hospital cardiac arrest and up to 24% for in-hospital arrest.(1,2)
EPIDEMIOLOGY
Incidence of SCA and its etiology appears to be a function of age, but this association is not linear. In children and young adults (< 34 years of age), annual risk of SCA is estimated as 1 per 100,000. This is followed by a steep increase in the risk of SCA beginning at age 35, when the incidence increases to 1/1000 per year. The annual risk continues to rise, peaking at the age of 75, and declines thereafter.(1) In younger individuals (median age of 29 years), a structurally normal heart can be found in up to 42% of autopsies,(3) as compared to only 4% of more elderly subjects (mean age 54.7 years).(4) Causes in younger patients include primary arrhythmia syndromes due to congenital channelopathies, inherited dilated, hypertrophic, or arrhythmogenic right ventricular cardiomyopathies, as well as coronary artery anomalies.(2) The overwhelming majority of SCAs in the elderly are due to underlying structural heart disease (SHD), which is attributed to CAD in ≥ 80% of cases.(1) Other less prevalent etiologies include non-ischemic causes of SHD, channelopathies (5–15%), and idiopathic VT/VF (5%).(2) Overall, there is a trend toward a decline in proportion of SCD related to CAD and an increase in the share of nonischemic etiologies.(1) However, the cause of SCA remains unexplained in up to two-thirds of patients without SHD after routine diagnostic evaluation, such as electrocardiographic telemetry monitoring, transthoracic echocardiogram and coronary angiography.(1,5) The diagnostic approach to cases with normal ejection fraction was assessed in Cardiac Arrest Survivors With Preserved Ejection Fraction Registry (CASPER). This study showed that in patients with normal ejection fraction, additional testing, including signal-averaged electrocardiogram, exercise stress test, cardiac magnetic resonance imaging, provocative pharmacologic testing with intravenous infusion of epinephrine and procainamide, electrophysiologic study, endomyocardial biopsy and targeted genetic testing may provide an incremental benefit and lead to a definitive diagnosis in up to 56% of SCA patients.(5) Specifically, cardiac magnetic resonance imaging was shown to identify an underlying pathological substrate in nearly half of survivors of unexplained SCA and was conclusive in making the diagnosis in 30%.(6)
PATHOPHYSIOLOGY
The majority of SCA cases are due to malignant ventricular tachyarrhythmias (VT), often related to CAD. Mechanisms by which CAD results in arrhythmogenesis include acute ischemia leading to VT, myocardial scars that serve as the substrate for reentrant monomorphic VTs, and in rare cases, disease in the cardiac electrical conduction leading to bundle branch reentry VT (BBRVT). Coronary reperfusion and medical management of CAD have been credited for the declining rate of ventricular tachyarrhythmias (VAs) leading to SCA. Use of internal cardioverter defibrillators (ICDs), although not affecting burden of VT episodes, has led to the reduction in arrhythmic death owing to prompt and efficient termination VA episodes. At the same time, the incidence of pulseless electrical activity is increasing, which is largely attributed to the growing number of patients with advanced heart failure. Bradyarrhythmias, usually due to third degree atrioventricular block without sufficient escape, account for a small proportion (10–15%) of all SCDs.(1)
PREVENTION
Primary Prevention
Prevention of SCD in patients who are considered high risk for it often involves institution of medications to target the sympathetic nervous system (β-adrenergic receptor blockers and angiotensin converting enzyme inhibitors) as well as implantation of ICDs. Randomized controlled clinical trials demonstrated a reduction in mortality for patients LVEF ≤ 35% who received ICDs vs. those managed with conventional medical therapy only, with a relative risk reduction between 23% to 28% and absolute risk reduction ranging from 5.6% to 7.2%.(1) However, beneficial effects of the ICD may depend on etiology of cardiomyopathy. Data from the Danish Study to Assess the Efficacy of ICDs in Patients with Non-ischemic Systolic Heart Failure (DANISH) demonstrated that in subjects with NICM, primary prevention ICD did not reduce all-cause mortality in all patients even though it was associated with a 2-fold reduction in arrhythmic death. Arrhythmic death was offset by death from heart failure. It is important to note that this was not the case in younger patients (<68 years old) in whom ICD reduced mortality by 36%.(7)
ICDs neither prevent recurrent episodes of VA nor modify the underlying myocardial substrate, which tends to progress over time. Defibrillators save lives by attempting to abort arrhythmic death. Unfortunately rate of ICD-unresponsive SCD due to VT or VF has been shown to be between 5% and 17%.(8) Moreover, both appropriate and inappropriate ICD shocks have been reported to herald a poor prognosis and increased risk of death.(9) Several strategies for VT detection and programming of ICD therapies have been proposed to minimize ICD shocks.(10) A high detection-rate approach, programming the ICD to monitor VAs with slower heart rates without delivering therapy, and treating only VAs with faster heart rates (≥ 200 bpm) has been proposed. A delayed-therapy strategy, which consists of increasing the time from detection of the VA to onset of ICD therapy, has also been advocated. (10). This delay could range from 60 seconds for VTs of slower cycle lengths (170–199 bpm), to 12 seconds for VTs of faster cycle lengths (200–249 bpm), and 2.5 seconds for very fast VAs (≥ 250 bpm). A delayed-therapy approach allows for termination of non-sustained arrhythmias before ICD therapies are delivered. A combination of both strategies, with modifications as deemed appropriate by the treating physician based on the patients’ clinical VT and presentation, may prove beneficial in reducing ICD shocks.
Even though ICDs play a major role in prevention of arrhythmic death, medications that target the sympathetic nervous system have been shown to reduce the risk of SCD. In the setting of SHD, β-blockers and angiotensin converting enzyme inhibitors have been demonstrated to reduce mortality and sudden death.(11,12) In addition, β-blocker medications are recommended as a first line therapy for patients with long QT syndrome and catecholaminergic polymorphic VT (CPVT).(13)
In certain population of channelopathies, anti-arrhythmic medications can be beneficial in reducing risk of VT. Flecainide can be used in addition to β-blockers in cases of CPVT with recurrent syncope or VAs.(13) Quinidine is beneficial for recurrent VAs in patients with Brugada syndrome who already have ICD and it may be considered in asymptomatic patients with spontaneous type 1 Brugada ECG.(13)
Secondary Prevention
Secondary prevention of SCA in subjects is implantation of an ICD. The overwhelming majority of patients in secondary prevention ICD clinical trials had underlying SHD, primarily due to CAD with reduced LVEF (< 35%).(1) Antiarrhythmics versus Implantable Defibrillators (AVID) trial demonstrated statistically significant relative risk reduction in all-cause mortality by 31% at 3 years in the ICD versus amiodarone groups.(14) Overall, based on a meta-analysis of 3 secondary prevention ICD trials, there was a 5% absolute risk reduction in mortality at 1 year and 8% at 2 years in the ICD compared to no ICD secondary prevention group.(14,15)
TREATMENT
Management of SCA in the acute setting
Survival from the SCA is a function of the presenting rhythm, which can be divided into shockable or nonshockable. An increasing proportion of pulseless electrical activity and asystole and decreasing rate of VT/VF pose a great challenge, as survival from nonshockable rhythms is very poor. This trend has been attributed to advances in preventative and therapeutic interventions for CAD, widespread use of ICDs, as well as the increase in age of the population. (16)
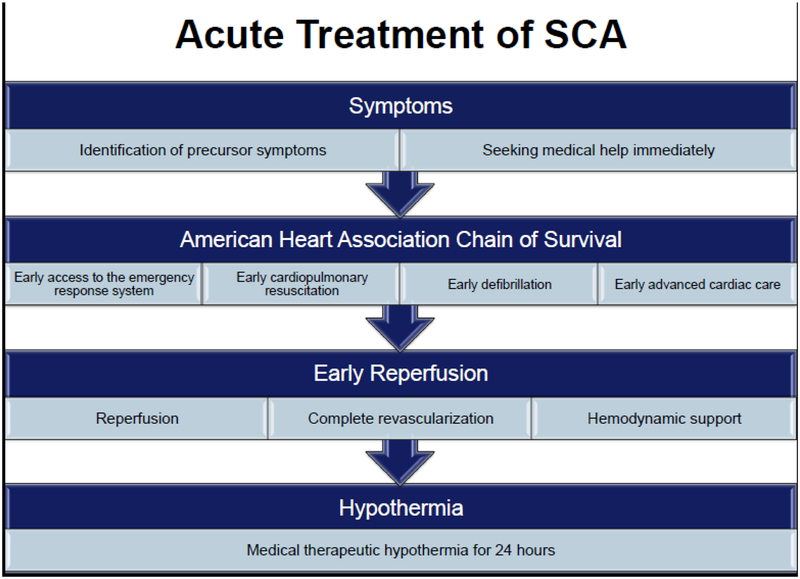
Steps of SCA management in the acute setting are outlined in Figure 1. Data from Oregon Sudden Unexpected Death Study indicated that half of all subjects who develop SCA have precursor symptoms, mainly chest pain and dyspnea, in the 4 weeks and particularly within 24 hours prior to the event. Individuals who called 911 at the onset of symptoms had a significantly better survival compared to those who did not (32.1% vs. 6.0%).(17). This emphasizes the importance of public education to take action when warning symptoms develop. Survival and functional outcome of SCA are also dependent upon implementation of the American Heart Association links of the chain of survival, including early access to emergency response systems, early cardiopulmonary resuscitation, and early defibrillation and advanced cardiac care. Early reperfusion, including percutaneous coronary intervention with the help of hemodynamic support devices, if needed, is essential in improving survival from SCA. In patients with ongoing hemodynamic instability, surgically implanted ventricular assist devices as a bridge to heart transplantation or destination therapy have been increasingly used to save lives.(18)
Figure 1.
Management of acute sudden cardiac arrest includes rapid resuscitation and hypothermia protocol. SCA = sudden cardiac arrest.
The only proven strategy to improve survival from out-of-hospital cardiac arrest after return of spontaneous circulation is therapeutic hypothermia.(19,20) The mechanism of therapeutic effect of hypothermia is not completely understood, but is believed to be due to a decrease in brain ischemia-reperfusion injury as a result of slowing of cellular metabolism and reduction in oxygen consumption. It appears that a longer duration of hypothermia (24 hours)(19) is superior to shorter duration (12 hours).(20)
Chronic therapy to reduce VT/VF
Because recurrent ICD shocks are associated with an increased mortality and decreased quality of life, a key aspect of the treatment of SCA is prevention and/or reduction of VT/VF episodes and ICD therapies.1(9) In this regard, catheter ablation of VT can be quite beneficial, particularly if the underlying substrate is ischemic cardiomyopathy (ICM) or the arrhythmia mechanism is BBRVT, (Figure 2).
Figure 2.
Ventricular Tachycardia Ablation-Approach and Outcomes. The approach to ablation as well as the outcomes of VT ablation are dependent on the presenting myocardial substrate and genetic abnormality. ICD – internal cardioverter defibrillator; PVC – premature ventricular complex; VF – ventricular fibrillation; VT – ventricular tachycardia
Two randomized controlled trials demonstrated that VT ablation reduces the rate of appropriate ICD shocks compared to medical therapy alone in patients with ICM who had already experienced a VT episode and/or ICD therapies. In the Substrate Mapping and Ablation in Sinus Rhythm to Halt Ventricular Tachycardia (SMASH-VT) study, patients with CAD and VT were randomized to ICD implantation or ICD implantation and catheter ablation. At 2 years of follow-up, there was a significant reduction in the incidence of all appropriate ICD therapies, including shocks or anti-tachycardia pacing (12% versus 33%, respectively; HR = 0.35; p = 0.007) and ICD shocks (9% versus 31%, respectively) in the ablation group compared to ICD only group.(21) Results of the Ventricular Tachycardia Ablation in Coronary Heart Disease (VTACH) trial demonstrated a statistically significant 39% decrease in the rate of ICD shocks at 2 years in the VT ablation group as compared to the ICD only group in patients with CAD, reduced LVEF and history of VT.(22) VA free survival was 47% in the ablation group as opposed to 29% in the control group (HR = 0.61, p = 0.045).(22) While both SMASH-VT and VTACH trials showed a reduction in ICD therapies for VAs in patients who had already had VT, neither study could demonstrate a decrease in mortality, as there was no difference in overall survival between the ablation and routine care arms of these studies.(21,22)
There are currently no prospective studies on the results of prophylactic catheter ablation of inducible VT in patients with primary prevention ICDs, but retrospective suggests that VT ablation in primary prevention ICD patients may decrease the rate of appropriate ICD shocks.(23) This decision has to be balanced with the lack of mortality data for VT ablation and the high cost and resources required for the procedure.
In addition to reducing the burden of VT and ICD shocks, successful VT ablation has been associated with mortality benefit in retrospective single center and multi-center studies.(24–26) VT ablation in earlier stages of the disease has also been associated with lower mortality compared to VT ablation performed in the setting of electrical storm.(27)
The Multicenter Thermocool Ventricular Tachycardia Ablation Trial was an observational study of VT ablation in patients with advanced SHD due to both ICM and NICM. Acute procedural success rate (no inducible VT at the end of the procedure) was 49% and 53% of patients were free from any VT at 6-month follow-up.(28) It also demonstrated a reduction in VT burden from a median of 11.5 episodes in the 6 months prior to ablation to 0 after ablation. However, as many as 20% of patients had an increase in the number of VT episodes. With regard to antiarrhythmic medications after ablation, 55% remained on these medications, while anti-arrhythmic medications were decreased in 26% decreased and increased 19% increased. Patients, who had successful ablation, were more likely to reduce their antiarrhythmic therapy, and 35% of them were not taking any at 6 months.(28) In a single center study of VT ablation in patients with SHD due to both ICM and NICM, overall VT recurrence rate was 34% at 24 months follow-up, ranging from 28.6% in patients with complete procedural success (no inducible VTs) to 66.7% in those with inducible clinical VT at the end of ablation. Non-inducibility of VT at the end of the ablation procedure was associated with a decreased risk of death and cardiac transplantation.(25) Most recently, non-inducibility of VT at the end of the procedure was associated with improved outcomes after VT ablation in a large multi-center study of 1064 patients.(29) This contradicted an earlier multicenter study (the Cooled RF Ablation System trial) of 106 patients which did not show prognostic utility to acute procedural success, though the latter study also demonstrated a high VT recurrence rate of 56% at 1 year.(30) The differences in the results of these retrospective studies may be related to advancement of catheter ablation techniques over the past two decades as well as the patient population studied.
It is also important to emphasize that catheter ablation of VT in the setting of SHD is a complex procedure. The rate of major perioperative complications can range from 3% (in the Multicenter Thermocool Ventricular Tachycardia Ablation Trial) to 8% (in the Cooled RF Multi Center trial).(28,30) The difference in the rates of complications for the different trials may again be related to both the advancement of mapping and ablation techniques over the past decade, increasing operator experience, as well as the patient population in each study.(21,22,28,30)
Data on outcomes of VT ablation in patients with NICM are limited. Many of the studies approach different etiologies of NICM as one group, making it difficult to assess results in specific etiologies. The International Ventricular Tachycardia Ablation Center Collaborative study and other single center studies have shown that the specific etiology of NICM affects rates of VT recurrence and combined endpoint of survival from VT, death, and heart transplantation, after VT ablation. Patients with dilated cardiomyopathy, myocarditis, and arrhythmogenic right ventricular cardiomyopathy appear to have superior outcomes after ablation compared to those with hypertrophic cardiomyopathy, valvular heart disease, and sarcoidosis.(31,32) In addition, underlying pathological substrate in NICM frequently involves epicardial surface of the heart, necessitating a combined epicardial and endocardial VT ablation approach.
VT ablation also has a role in primary idiopathic VF that is initiated by PVCs. PVC triggers are most frequently mapped to the distal Purkinje system in either the left or right ventricle and border zones of infarcts and less frequently, to the right ventricular outflow tract. Ablation of focal PVC triggers was reported to have a high success rate for prevention of further VF episodes.(33)
Analogous to idiopathic VF, electrical storm due to polymorphic VT or VF in the setting of acute ischemia may be triggered by PVCs originating from the His-Purkinje system, specifically the left posterior fascicle. Purkinje fibers are more resistant to ischemic injury compared to the rest of the myocardium. They are contained in surviving islands of myocardium within the scar tissue post myocardial infarction. Published ablation data on a small number of patients with incessant VT/VF triggered by monomorphic PVCs are very encouraging, with success rates between 80% and 100% for the treatment of electrical storm and prevention VF.(34)
BBRVT represents an infrequent but distinct form of VT, particularly in the setting of NICM, accounting for 5–8% of monomorphic VT in patients with SHD.(1) Given reported success rate of ablation approaching 100%, it is a class I recommendation to treat BBRVT with catheter ablation as the first-line therapy.(1) Nevertheless, given marked underlying myocardial disease, patients should still receive ICDs to prevent SCA from myocardial VTs.
There are some data indicating that triggers of electrical storm in Brugada syndrome are located in the right ventricular outflow tract. Epicardial and endocardial ablation in this region may treat electrical storm and prevent VF recurrences.(35)
AUTONOMIC MODULATION
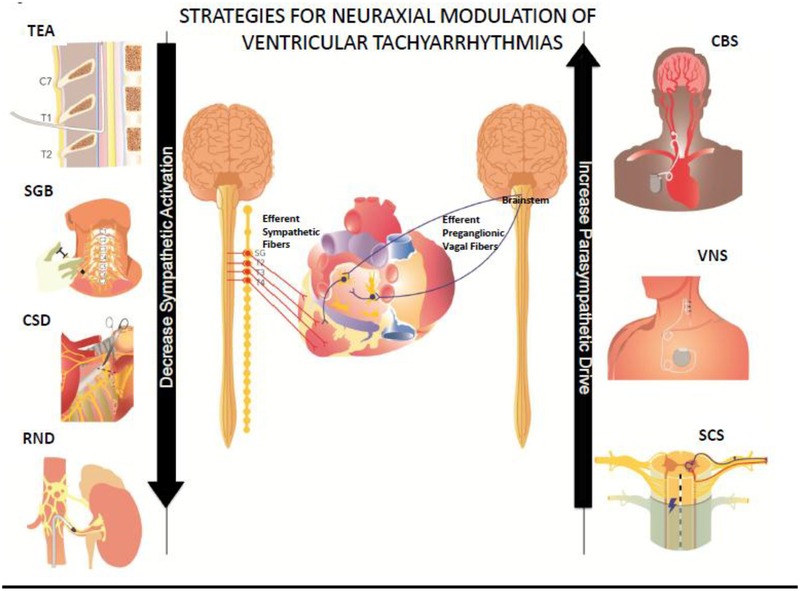
There is growing body of evidence to suggest that the autonomic nervous system is implicated in the genesis of cardiac arrhythmias.(36,37) Increased sympathetic activation has been linked to VAs and SCD in both animal studies and humans, while vagus nerve stimulation has shown protective effects.(36–38) Cardiac injury after myocardial infarction causes local denervation of sympathetic fibers, which is followed by non-homogeneous innervation, leading to electrical heterogeneity of the myocardium in the setting of sympathetic activation.(39,40) Sympathetic activation, even in normal hearts, can lead to triggered activity and increase dispersion of repolarization, a phenomenon that is further exaggerated in diseased hearts and can promote and sustain reentry.(41–43) The pathological neural remodeling, that follows adverse myocardial remodeling, affects not only the scar at the site of injury, but also the border zone regions and even the remote viable myocardium. Moreover, neural remodeling beyond the myocardium, at the level of cardiac and stellate ganglia has been described in association with both ICM and NICM.(44) Therefore, autonomic modulation, potentially at each level of the cardiac neuraxis, may provide an effective treatment modality to improve arrhythmia management. Currently available procedures of autonomic modulation are listed in Figure 3.
Figure 3.
Strategies for neuraxial modulation of ventricular tachyarrhythmias include methods to increase parasympathetic tone and decrease sympathetic activation. CBS – carotid body stimulation; CSD – cardiac sympathetic denervation; RND – renal denervation; SCS – spinal cord stimulation; SGB – stellate ganglion blockade; TEA – thoracic epidural anesthesia; VNS vagal nerve stimulation
Most cardioprotective medications currently used in the management of heart failure (β-blockers, angiotensin converting enzyme inhibitors, angiotensin receptor blockers, aldosterone antagonists) exert their beneficial effects by blocking sympathetic activation. Statins and fish oil, used for hyperlipidemia, have also been shown to reduce sympathetic and increase parasympathetic tone.(37) Of note, a nonselective β-blocker, propranolol, was shown to be superior to a selective β1-adrenergic receptor blocker, metoprolol, when used in combination with intravenous amiodarone for the management of patients with electrical storm. Subjects in the oral propranolol arm of the study had significantly lower incidence of VA and ICD discharges, suggesting that non-selective β-blocker therapy may be more beneficial in this population.(45) Deep sedation and general anesthesia can decrease sympathetic activation and have been proven effective in treating electrical storm.
Thoracic epidural anesthesia can be performed at bedside and provides bilateral complete sympathetic blockade between C8 and T4 segments of the spinal cord, proximal to both left and right stellate ganglia.(46) Encouraging results have been demonstrated in animal models and human studies. Thoracic epidural anesthesia was able to transiently but effectively terminate electrical storm, which was refractory to medical therapy.(46,47)
Cardiac sympathetic denervation (CSD) refers to surgical resection of 1/2 to 1/3 of the stellate and T1–T4 thoracic ganglia. Left CSD was initially found to be effective for long QT syndrome(48) and, subsequently, for CPVT.(49) CSD in the setting of SHD (both ICM and NICM) and refractory VT has been shown to reduce burden of VT/VF and ICD shocks, and in a porcine infarct model, to reduce VT inducibility acutely.(50–52) Studies in patients with structural heart disease also suggests that bilateral CSD may have more durable outcomes in reducing ICD shocks than left sided alone.(51) In the setting of electrical storm, sympathetic blockade (stellate ganglionic blockade or intravenous infusion of beta blockers) in addition to standard treatment with Advanced Cardiac Life Support (ACLS) was found to improve survival compared to standard ACLS alone.(53)
Spinal cord stimulation (SCS) is performed by placement of a stimulation catheter in epidural space at the level of T1–T4. There are conflicting data about effects of SCS in patients with heart failure, likely related to differences in dosing, duration and frequency of stimulation.(54,55) Reported positive effects of SCS in treatment of VAs are limited to animal studies. In a dog mode of myocardial infarction, SCS significantly reduced ischemic VAs.(56)
Vagal nerve stimulation (VNS) using bipolar electrodes placed around the cervical vagal trunk has shown promising results in animal models for treatment of VAs. It is noteworthy that VNS has significant ventricular electrophysiological effects, increasing action potential duration and effective refractory period.(57–59) In the setting of chronic myocardial infarction, VNS can stabilize infarct border zone and reduce VT inducibility acutely.(58,60) Furthermore, in a porcine infarct model, VNS after cardiac sympathetic denervation has been shown to further reduce inducible VAs.(60) It is important to understand that electrical stimulation of the vagal trunk can activate afferent as well as efferent fibers and reduce the effects of efferent stimulation.(57) Although VNS has not been evaluated for the treatment of VT in patients with heart disease, studies of VNS for the treatment of heart failure have produced mixed results.(38,61) In the Autonomic Regulation Therapy for the Improvement of Left Ventricular Function and Heart Failure Symptoms (ANTHEM-HF), an un-blinded study of heart failure patients with LVEF ≤ 40%, VNS group had an improvement in LVEF and functional heart failure class.(62) Conversely, the randomized trial, Neural Cardiac Therapy for Heart Failure (NECTAR-HF), consisting of individuals with LVEF ≤ 35% failed to show beneficial effects of VNS on cardiac function at 6 months and 18 months of follow up, but VNS implantation was associated with a better quality of life.(63,64) Finally, in another randomized study, the Increase in Vagal Tone in Heart Failure (INOVATE-HF), VNS did not reduce mortality or heart failure events in patients with LVEF ≤ 40%.(65) The difference in the results of these studies may be related to the different stimulation parameters utilized in each study.
The difference in the results of these studies may be related to the different stimulation parameters utilized in each study.
Additional methods of increasing parasympathetic tone include transcutaneous vagal nerve stimulation and baroreceptor activation.(66,67) Both of these therapies remain to be tested for treatment of VT, but studies in a canine model and in patients with myocardial infarction suggest a benefit in reducing ischemia associated ventricular arrhythmias.
Renal denervation represents another method of modulation of sympathetic signaling. Catheter ablation of renal nerve fibers is performed through the bilateral renal arteries. In small case series, this treatment has shown reduction in burden of VT/VF episodes in patients with refractory VT, unresponsive to cardiac ablation and antiarrhythmic medications.(68,69)Renal denervation is limited by variable anatomy around the renal arteries that can change the efficacy of ablation, including veins and lymphoid tissue, as well as the location and distribution of the nerves around the renal arteries.(70,71) An objective marker that could signal adequate “denervation” is needed as procedural endpoint.
SUMMARY
SCA is the leading cause of death worldwide. Frequent presentation as the first manifestation of cardiac disease and poor survival rates after SCA emphasize importance of primary prevention. Secondary prevention and treatment of SCA focus primarily on VT/VF management. Catheter ablation of VT and PVC triggers of VF has been shown to reduce recurrent arrhythmia episodes and may improve survival, if successful. Further mechanistic and clinical studies are needed in this area to elucidate the best methods of neuromodulation, impact of these therapies, and their mechanisms of benefit.
Acknowledgments
Funding for Dr. Vaseghi: Grant from National Institutes of Health: NIHDP2HL132356
ABBREVIATIONS:
- BBRVT
bundle branch reentry ventricular tachycardia
- bpm
beats per minute
- CAD
coronary artery disease
- CPVT
catecholaminergic polymorphic ventricular tachycardia
- CSD
cardiac sympathetic denervation
- ICD
internal cardioverter defibrillator
- ICM
ischemic cardiomyopathy
- LVEF
left ventricular ejection fraction
- NICM
non-ischemic cardiomyopathy
- PVC
premature ventricular complex
- SCA
sudden cardiac arrest
- SCD
sudden cardiac death
- SCS
spinal cord stimulation
- SHD
structural heart disease
- VA
ventricular arrhythmia
- VF
ventricular fibrillation
- VNS
vagal nerve stimulation
- VT
ventricular tachycardia
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
REFERENCES
- 1.Al-Khatib SM, Stevenson WG, Ackerman MJ, Gillis AM, Bryant WJ, Hlatky MA, et al. 2017 AHA/ACC/HRS Guideline for Management of Patients With Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death: Executive Summary. Heart Rhythm. 2018;15(10):e190–e252. [DOI] [PubMed] [Google Scholar]
- 2.Benjamin EJ, Blaha MJ, Chiuve SE, Cushman M, Das SR, Deo R, et al. Heart Disease and Stroke Statistics-2017 Update: A Report From the American Heart Association. Circulation. 2017;135(10):e146–e603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Finocchiaro G, Papadakis M, Robertus J-L, Dhutia H, Steriotis AK, Tome M, et al. Etiology of Sudden Death in Sports. JACC. 2016;67(18):2108–15. [DOI] [PubMed] [Google Scholar]
- 4.Hookana E, Junttila MJ, Puurunen V-PP, Tikkanen JT, Kaikkonen KS, Kortelainen M-LL, et al. Causes of nonischemic sudden cardiac death in the current era. Heart Rhythm. 2011;8(10):1570–5. [DOI] [PubMed] [Google Scholar]
- 5.Krahn AD, Healey JS, Chauhan V, Birnie DH, Simpson CS, Champagne J, et al. Systematic Assessment of Patients With Unexplained Cardiac Arrest: Cardiac Arrest Survivors With Preserved Ejection Fraction Registry (CASPER). Circulation. 2009;120(4):278–85. [DOI] [PubMed] [Google Scholar]
- 6.Rodrigues P, Joshi A, Williams H, Westwood M, Petersen SE, Zemrak F, et al. Diagnosis and Prognosis in Sudden Cardiac Arrest Survivors Without Coronary Artery Disease: Utility of a Clinical Approach Using Cardiac Magnetic Resonance Imaging. Circ Cardiovasc Imaging. 2017;10(12):e006709. [DOI] [PubMed] [Google Scholar]
- 7.Køber L, Thune JJ, Nielsen JC, Haarbo J, Videbæk L, Korup E, et al. Defibrillator Implantation in Patients with Nonischemic Systolic Heart Failure. N Engl J Med. 2016;375(13):1221–30. [DOI] [PubMed] [Google Scholar]
- 8.Narayanan K, Reinier K, Uy-Evanado A, Teodorescu C, Chugh H, Marijon E, et al. Frequency and Determinants of Implantable Cardioverter Defibrillator Deployment Among Primary Prevention Candidates With Subsequent Sudden Cardiac Arrest in the Community. Circulation. 2013;128(16):1733–8. [DOI] [PubMed] [Google Scholar]
- 9.Poole JE, Johnson GW, Hellkamp AS, Anderson J, Callans DJ, Raitt MH, et al. Prognostic Importance of Defibrillator Shocks in Patients with Heart Failure. N Engl J Med. 2008;359(10):1009–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Moss AJ, Schuger C, Beck CA, Brown MW, Cannom DS, Daubert JP, et al. Reduction in Inappropriate Therapy and Mortality through ICD Programming. N Engl J Med. 2012;367(24):2275–83. [DOI] [PubMed] [Google Scholar]
- 11.Hjalmarson Å Effects of Beta Blockade on Sudden Cardiac Death During Acute Myocardial Infarction and the Postinfarction Period. Am J Cardiol. 1997;80(9):35J–39J. [DOI] [PubMed] [Google Scholar]
- 12.Kober L, Torp-Pedersen C, Carlsen JE, Bagger H, Eliasen P, Lyngborg K, et al. A clinical trial of the angiotensin-converting-enzyme inhibitor trandolapril in patients with left ventricular dysfunction after myocardial infarction. Trandolapril Cardiac Evaluation (TRACE) Study Group. N Engl J Med. 1995;333(25):1670–6. [DOI] [PubMed] [Google Scholar]
- 13.Priori SG, Wilde AA, Horie M, Cho Y, Behr ER, Berul C, et al. HRS/EHRA/APHRS Expert Consensus Statement on the Diagnosis and Management of Patients with Inherited Primary Arrhythmia Syndromes. Heart Rhythm. 2013;10(12):1932–63. [DOI] [PubMed] [Google Scholar]
- 14.Connolly SJ, Hallstrom AP, Cappato R, Schron EB, KH K, Zipes DP, et al. Meta-analysis of the implantable cardioverter defibrillator secondary prevention trials. Eur Heart J. 2000;21(24):2071–8. [DOI] [PubMed] [Google Scholar]
- 15.Betts TR, Sadarmin PP, Tomlinson DR, Rajappan K, Wong KCK, de Bono JP, et al. Absolute risk reduction in total mortality with implantable cardioverter defibrillators: analysis of primary and secondary prevention trial data to aid risk/benefit analysis. Europace. 2013;15(6):813–9. [DOI] [PubMed] [Google Scholar]
- 16.Teodorescu C, Reinier K, Dervan C, Uy-Evanado A, Samara M, Mariani R, et al. Factors associated with pulseless electric activity versus ventricular fibrillation: the Oregon sudden unexpected death study. Circulation. 2010;122(21):2116–22. [DOI] [PubMed] [Google Scholar]
- 17.Marijon E, Uy-Evanado A, Dumas F, Karam N, Reinier K, Teodorescu C, et al. Warning Symptoms Are Associated With Survival From Sudden Cardiac Arrest. Annals of Internal Medicine. 2016;164(1):23–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Csepe TA, Kilic A. Advancements in mechanical circulatory support for patients in acute and chronic heart failure. J Thorac Dis. 2017;9(10):4070–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.The Hypothermia After Cardiac Arrest Group. Mild Therapeutic Hypothermia to Improve the Neurologic Outcome after Cardiac Arrest. N Engl J Med. 2002;346(8):549–56. [DOI] [PubMed] [Google Scholar]
- 20.Bernard SA, Gray TW, Buist MD, Jones BM, Silvester W, Gutteridge G, et al. Treatment of Comatose Survivors of Out-of-Hospital Cardiac Arrest with Induced Hypothermia. N Engl J Med. 2002;346(8):557–63. [DOI] [PubMed] [Google Scholar]
- 21.Reddy VY, Reynolds MR, Neuzil P, Richardson AW, Taborsky M, Jongnarangsin K, et al. Prophylactic Catheter Ablation for the Prevention of Defibrillator Therapy. N Engl J Med. 2007;357(26):2657–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kuck K-H, Schaumann A, Eckardt L, Willems S, Ventura R, Delacrétaz E, et al. Catheter ablation of stable ventricular tachycardia before defibrillator implantation in patients with coronary heart disease (VTACH): a multicentre randomised controlled trial. The Lancet. 2010;375(9708):31–40. [DOI] [PubMed] [Google Scholar]
- 23.Hayashi T, Fukamizu S, Hojo R, Komiyama K, Tanabe Y, Tejima T, et al. Prophylactic catheter ablation for induced monomorphic ventricular tachycardia in patients with implantable cardioverter defibrillators as primary prevention. Europace. 2013;15(10):1507–15. [DOI] [PubMed] [Google Scholar]
- 24.Tung R, Callans DJ, Frankel DS, Vakil K, Reddy M, Vaseghi M, et al. Freedom from recurrent ventricular tachycardia after catheter ablation is associated with improved survival in patients with structural heart disease: An International VT Ablation Center Collaborative Group study. Heart Rhythm. 2015;12(9):1997–2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Bella Della P, Baratto F, Tsiachris D, Trevisi N, Vergara P, Bisceglia C, et al. Management of ventricular tachycardia in the setting of a dedicated unit for the treatment of complex ventricular arrhythmias: long-term outcome after ablation. Circulation. 2013;127(13):1359–68. [DOI] [PubMed] [Google Scholar]
- 26.Bunch TJ, Weiss JP, Crandall BG, Day JD, May HT, Bair TL, et al. Patients treated with catheter ablation for ventricular tachycardia after an ICD shock have lower long-term rates of death and heart failure hospitalization than do patients treated with medical management only. Heart Rhythm. 2014;11(4):533–40. [DOI] [PubMed] [Google Scholar]
- 27.Aldhoon B, Wichterle D, Peichl P, Čihák R, Kautzner J. Outcomes of ventricular tachycardia ablation in patients with structural heart disease: The impact of electrical storm. Tolkacheva E, editor. PLoS ONE. 2017;12(2):e0171830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Stevenson WG, Wilber DJ, Natale A, Jackman WM, Marchlinski FE, Talbert T, et al. Irrigated radiofrequency catheter ablation guided by electroanatomic mapping for recurrent ventricular tachycardia after myocardial infarction: the multicenter thermocool ventricular tachycardia ablation trial. Circulation. 2008;118(25):2773–82. [DOI] [PubMed] [Google Scholar]
- 29.Yokokawa M, Kim HM, Baser K, Stevenson W, Nagashima K, Bella Della P, et al. Predictive value of programmed ventricular stimulation after catheter ablation of post-infarction ventricular tachycardia. JACC. 2015;65(18):1954–9. [DOI] [PubMed] [Google Scholar]
- 30.Calkins H, Epstein A, Packer D, Arria AM, Hummel J, Gilligan DM, et al. Catheter ablation of ventricular tachycardia in patients with structural heart disease using cooled radiofrequency energy. JACC. 2000;35(7):1905–14. [DOI] [PubMed] [Google Scholar]
- 31.Tokuda M, Tedrow UB, Kojodjojo P, Inada K, Koplan BA, Michaud GF, et al. Catheter ablation of ventricular tachycardia in nonischemic heart disease. Circulation: Arrhythmia and Electrophysiology. 2012;5(5):992–1000. [DOI] [PubMed] [Google Scholar]
- 32.Vaseghi M, Hu TY, Tung R, Vergara P, Frankel DS, Di Biase L, et al. Outcomes of Catheter Ablation of Ventricular Tachycardia Based on Etiology in Nonischemic Heart Disease: An International Ventricular Tachycardia Ablation Center Collaborative Study. JACC Clin Electrophysiol. 2018;4(9):1141–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Haïssaguerre M, Shoda M, Jaïs P, Nogami A, Shah DC, Kautzner J, et al. Mapping and ablation of idiopathic ventricular fibrillation. Circulation. 2002;106(8):962–7. [DOI] [PubMed] [Google Scholar]
- 34.Bansch D, Oyang F, Antz M, Arentz T, Weber R, Val-Mejias JE, et al. Successful Catheter Ablation of Electrical Storm After Myocardial Infarction. Circulation. 2003;108(24):3011–6. [DOI] [PubMed] [Google Scholar]
- 35.Nademanee K, Veerakul G, Chandanamattha P, Chaothawee L, Ariyachaipanich A, Jirasirirojanakorn K, et al. Prevention of ventricular fibrillation episodes in Brugada syndrome by catheter ablation over the anterior right ventricular outflow tract epicardium. Circulation. 2011;123(12):1270–9. [DOI] [PubMed] [Google Scholar]
- 36.Shen MJ, Zipes DP. Role of the autonomic nervous system in modulating cardiac arrhythmias. Circulation Research. 2014;114(6):1004–21. [DOI] [PubMed] [Google Scholar]
- 37.Vaseghi M, Shivkumar K. The Role of the Autonomic Nervous System in Sudden Cardiac Death. Prog Cardiovasc Dis. 2008;50(6):404–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Huang WA, Shivkumar K, Vaseghi M. Device-Based Autonomic Modulation in Arrhythmia Patients: the Role of Vagal Nerve Stimulation. Curr Treat Options Cardiovasc Med. 2015;17(5):687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Cao J-MM, Fishbein MC, Han JB, Lai WW, Lai AC, Wu T-JJ, et al. Relationship between regional cardiac hyperinnervation and ventricular arrhythmia. Circulation. 2000;101(16):1960–9. [DOI] [PubMed] [Google Scholar]
- 40.Fallavollita JA, Heavey BM, Luisi AJ Jr., Michalek SM, Baldwa S, Mashtare TL Jr., et al. Regional Myocardial Sympathetic Denervation Predicts the Risk of Sudden Cardiac Arrest in Ischemic Cardiomyopathy. JACC. 2014;63(2):141–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Vaseghi M, Lux RL, Mahajan A, Shivkumar K. Sympathetic stimulation increases dispersion of repolarization in humans with myocardial infarction. AJP: Heart and Circulatory Physiology. 2012;302(9):H1838–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Opthof T, Misier AR, Coronel R, Vermeulen JT, Verberne HJ, Frank RG, et al. Dispersion of refractoriness in canine ventricular myocardium. Effects of sympathetic stimulation. Circulation Research. 1991;68(5):1204–15. [DOI] [PubMed] [Google Scholar]
- 43.Yagishita D, Chui RW, Yamakawa K, Rajendran PS, Ajijola OA, Nakamura K, et al. Sympathetic nerve stimulation, not circulating norepinephrine, modulates T-peak to T-end interval by increasing global dispersion of repolarization. Circulation: Arrhythmia and Electrophysiology. 2015;8(1):174–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Ajijola OA, Hoover DB, Simerly TM, Brown TC, Yanagawa J, Biniwale RM, et al. Inflammation, oxidative stress, and glial cell activation characterize stellate ganglia from humans with electrical storm. JCI Insight. 2017;2(18):1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Chatzidou S, Kontogiannis C, Tsilimigras DI, Georgiopoulos G, Kosmopoulos M, Papadopoulou E, et al. Propranolol Versus Metoprolol for Treatment of Electrical Storm in Patients With Implantable Cardioverter-Defibrillator. JACC. 2018;71(17):1897–906. [DOI] [PubMed] [Google Scholar]
- 46.Bourke T, Vaseghi M, Michowitz Y, Sankhla V, Shah M, Swapna N, et al. Neuraxial modulation for refractory ventricular arrhythmias: value of thoracic epidural anesthesia and surgical left cardiac sympathetic denervation. Circulation. 2010;121(21):2255–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Do DH, Bradfield J, Ajijola OA, Vaseghi M, Le J, Rahman S, et al. Thoracic Epidural Anesthesia Can Be Effective for the Short-Term Management of Ventricular Tachycardia Storm. Journal of the American Heart Association. 2017;6(11):e007080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Schwartz PJ, Priori SG, Cerrone M, Spazzolini C, Odero A, Napolitano C, et al. Left Cardiac Sympathetic Denervation in the Management of High-Risk Patients Affected by the Long-QT Syndrome. Circulation. 2004;109(15):1826–33. [DOI] [PubMed] [Google Scholar]
- 49.De Ferrari GM, Dusi V, Spazzolini C, Bos JM, Abrams DJ, Berul CI, et al. Clinical Management of Catecholaminergic Polymorphic Ventricular Tachycardia: TheRole of Left Cardiac Sympathetic Denervation. Circulation. 2015;131(25):2185–93. [DOI] [PubMed] [Google Scholar]
- 50.Vaseghi M, Gima J, Kanaan C, Ajijola OA, Marmureanu A, Mahajan A, et al. Cardiac sympathetic denervation in patients with refractory ventricular arrhythmias or electrical storm: Intermediate and long-term follow-up. Heart Rhythm. 2014;11(3):360–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Vaseghi M, Barwad P, Malavassi Corrales FJ, Tandri H, Mathuria N, Shah R, et al. Cardiac Sympathetic Denervation for Refractory Ventricular Arrhythmias. JACC. 2017;69(25):3070–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Irie T, Yamakawa K, Hamon D, Nakamura K, Shivkumar K, Vaseghi M. Cardiac sympathetic innervation via middle cervical and stellate ganglia and antiarrhythmic mechanism of bilateral stellectomy. AJP: Heart and Circulatory Physiology. 2017;312(3):H392–H405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Nademanee K, Taylor R, Bailey WE, Rieders DE, Kosar EM. Treating Electrical Storm : Sympathetic Blockade Versus Advanced Cardiac Life Support-Guided Therapy. Circulation. 2000;102(7):742–7. [DOI] [PubMed] [Google Scholar]
- 54.Zipes DP, Neuzil P, Theres H, Caraway D, Mann DL, Mannheimer C, et al. Determining the Feasibility of Spinal Cord Neuromodulation for the Treatment of Chronic Systolic Heart Failure: The DEFEAT-HF Study. JACC: Heart Failure. 2016;4(2):129–36. [DOI] [PubMed] [Google Scholar]
- 55.Ardell JL. Mechanisms of spinal cord neuromodulation for heart disease. Nat Rev Cardiol. 2016;13(3):127–8. [DOI] [PubMed] [Google Scholar]
- 56.Lopshire JC, Zhou X, Dusa C, Ueyama T, Rosenberger J, Courtney N, et al. Spinal cord stimulation improves ventricular function and reduces ventricular arrhythmias in a canine postinfarction heart failure model. Circulation. 2009;120(4):286–94. [DOI] [PubMed] [Google Scholar]
- 57.Yamakawa K, So EL, Rajendran PS, Hoang JD, Makkar N, Mahajan A, et al. Electrophysiological effects of right and left vagal nerve stimulation on the ventricular myocardium. AJP: Heart and Circulatory Physiology. 2014;307(5):H722–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Vaseghi M, Salavatian S, Rajendran PS, Yagishita D, Woodward WR, Hamon D, et al. Parasympathetic dysfunction and antiarrhythmic effect of vagal nerve stimulation following myocardial infarction. JCI Insight. 2017;2(16). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Ellenbogen KA, Smith ML, Eckberg DL. Increased vagal cardiac nerve traffic prolongs ventricular refractoriness in patients undergoing electrophysiology testing. Am J Cardiol. 1990;65(20):1345–50. [DOI] [PubMed] [Google Scholar]
- 60.Yamaguchi N, Yamakawa K, Rajendran PS, Takamiya T, Vaseghi M. Anti-Arrhythmic Effects of Vagal Nerve Stimulation After Cardiac Sympathetic Denervation in the Setting of Chronic Myocardial Infarction. Heart Rhythm. 2018;15(8):1214–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Ardell JL, Nier H, Hammer M, Southerland EM, Ardell CL, Beaumont E, et al. Defining the neural fulcrum for chronic vagus nerve stimulation: implications for integrated cardiac control. The Journal of Physiology. 2017;595(22):6887–903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Premchand RK, Sharma K, Mittal S, Monteiro R, Dixit S, Libbus I, et al. Extended Follow-Up of Patients With Heart Failure Receiving Autonomic Regulation Therapy in the ANTHEM-HF Study. Journal of Cardiac Failure. 2016;22(8):639–42. [DOI] [PubMed] [Google Scholar]
- 63.Zannad F, De Ferrari GM, Tuinenburg AE, Wright D, Brugada J, Butter C, et al. Chronic vagal stimulation for the treatment of low ejection fraction heart failure: results of the NEural Cardiac TherApy foR Heart Failure (NECTAR-HF) randomized controlled trial. Eur Heart J. 2014;36(7):425–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.De Ferrari GM, Stolen C, Tuinenburg AE, Wright DJ, Brugada J, Butter C, et al. Long-term vagal stimulation for heart failure: Eighteen month results from the NEural Cardiac TherApy foR Heart Failure (NECTAR-HF) trial. International Journal of Cardiology. 2017;244:229–34. [DOI] [PubMed] [Google Scholar]
- 65.Gold MR, Van Veldhuisen DJ, Hauptman PJ, Borggrefe M, Kubo SH, Lieberman RA, et al. Vagus Nerve Stimulation for the Treatment of Heart Failure. JACC. 2016;68(2):149–58. [DOI] [PubMed] [Google Scholar]
- 66.Yu L, Huang B, Po SS, Tan T, Wang M, Zhou L, et al. Low-Level Tragus Stimulation for the Treatment of Ischemia and Reperfusion Injury in Patients With ST-Segment Elevation Myocardial Infarction. JACC Cardiovasc Interv. 2017;10(15):1511–20. [DOI] [PubMed] [Google Scholar]
- 67.Liao K, Yu L, Yang K, Saren G, Wang S, Huang B, et al. Low-Level Carotid Baroreceptor Stimulation Suppresses Ventricular Arrhythmias during Acute Ischemia. Barbosa Moraes Resstel L, editor. PLoS ONE. 2014;9(10):e109313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Remo BF, Preminger M, Bradfield J, Mittal S, Boyle N, Gupta A, et al. Safety and efficacy of renal denervation as a novel treatment of ventricular tachycardia storm in patients with cardiomyopathy. Heart Rhythm. 2014;11(4):541–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Armaganijan LV, Staico R, Moreira DAR, Lopes RD, Medeiros PTJ, Habib R, et al. 6-Month Outcomes in Patients With Implantable Cardioverter-Defibrillators Undergoing Renal Sympathetic Denervation for the Treatment of Refractory Ventricular Arrhythmias. JACC Cardiovasc Interv. 2015;8(7):984–90. [DOI] [PubMed] [Google Scholar]
- 70.Tzafriri AR, Keating JH, Markham PM, Spognardi A-M, Stanley JRL, Wong G, et al. Arterial microanatomy determines the success of energy-based renal denervation in controlling hypertension. Science Translational Medicine. 2015;7(285):285ra65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Sakakura K, Ladich E, Cheng Q, Otsuka F, Yahagi K, Fowler DR, et al. Anatomic assessment of sympathetic peri-arterial renal nerves in man. JACC. 2014;64(7):635–43. [DOI] [PubMed] [Google Scholar]