Abstract
Objective
To evaluate if the lowest target level for glycated haemoglobin (HbA1c) of <6.5% is associated with lower risk for retinopathy and nephropathy than less tight control in children and adults with type 1 diabetes.
Design
Population based cohort study.
Setting
Swedish National Diabetes Registry, 1 January 1998 to 31 December 2017.
Participants
10 398 children and adults with type 1 diabetes followed from diagnosis, or close thereafter, until end of 2017.
Main outcome measures
Relative risk (odds ratios) for retinopathy and nephropathy for different mean levels of HbA1c.
Results
Mean age of participants was 14.7 years (43.4% female), mean duration of diabetes was 1.3 years, and mean HbA1c level was 8.0% (63.4 mmol/mol). After adjustment for age, sex, duration of diabetes, blood pressure, blood lipid levels, body mass index, and smoking, the odds ratio for mean HbA1c <6.5% (<48 mmol/mol) compared with 6.5-6.9% (48-52 mmol/mol) for any retinopathy (simplex or worse) was 0.77 (95% confidence interval 0.56 to 1.05, P=0.10), for preproliferative diabetic retinopathy or worse was 3.29 (0.99 to 10.96, P=0.05), for proliferative diabetic retinopathy was 2.48 (0.71 to 8.62, P=0.15), for microalbuminuria or worse was 0.98 (0.60 to 1.61, P=0.95), and for macroalbuminuria was 2.47 (0.69 to 8.87, P=0.17). Compared with HbA1c levels 6.5-6.9%, HbA1c levels 7.0-7.4% (53-57 mmol/mol) were associated with an increased risk of any retinopathy (1.31, 1.05 to 1.64, P=0.02) and microalbuminuria (1.55, 1.03 to 2.32, P=0.03). The risk for proliferative retinopathy (5.98, 2.10 to 17.06, P<0.001) and macroalbuminuria (3.43, 1.14 to 10.26, P=0.03) increased at HbA1c levels >8.6% (>70 mmol/mol). The risk for severe hypoglycaemia was increased at mean HbA1c <6.5% compared with 6.5-6.9% (relative risk 1.34, 95% confidence interval 1.09 to 1.64, P=0.005).
Conclusions
Risk of retinopathy and nephropathy did not differ at HbA1c levels <6.5% but increased for severe hypoglycaemia compared with HbA1c levels 6.5-6.9%. The risk for severe complications mainly occurred at HbA1c levels >8.6%, but for milder complications was increased at HbA1c levels >7.0%.
Introduction
Type 1 diabetes is the predominant form of diabetes diagnosed in childhood and is characterised by increased blood glucose levels resulting from insulin deficiency. Treatment in developed countries generally comprises exogenous insulin by injections or insulin pump as guided by capillary testing or use of continuous glucose monitoring to estimate blood glucose levels. Intensive treatment with better glycaemic control has been shown to reduce the risk of retinopathy and nephropathy, with a strong association between glycated haemoglobin (HbA1c) level and diabetes complications.1 2 3 4
Multiple studies in several countries have confirmed that a higher HbA1c level is an independent risk factor for diabetes complications.1 2 3 4 5 6 7 8 9 10 Few studies have followed patients from diagnosis, therefore large contemporary population based studies evaluating the relation between HbA1c levels and diabetes complications would complement earlier studies to help understand the risks of complications during modern glucose lowering treatments.
The evidence for HbA1c levels and diabetes complications is interpreted differently by organisations such as the American Diabetes Association and International Society for Paediatric and Adolescent Diabetes, resulting in guidelines that differ between organisations and countries—current guidelines range between 6.5% (48 mmol/mol) and 7.5% (58 mmol/mol).11 12 13 14 HbA1c targets also differ within countries for children and adults. Reaching lower HbA1c levels requires greater effort from people with diabetes and their families and can be associated with more stress and possibly an increased risk of hypoglycaemia, which in itself can be burdensome and lead to unconsciousness.1 15 16 If major risk reductions of long term complications could be obtained with low HbA1c levels this might outweigh a potentially increased risk of hypoglycaemia and the efforts needed to achieve low levels.
To understand better the relation between HbA1c level and microvascular complications in people with type 1 diabetes, we utilised the paediatric and adult diabetes registries in Sweden. Participants were followed-up regarding retinopathy and nephropathy for 8-20 years from diagnosis.
Methods
This is a nationwide population based observational cohort study. Participants provided informed consent.
Data sources
The Swedish Diabetes Registry for adults recently merged with Swediabkids, the paediatric registry for diabetes established in 2000. Since 1996 the Swedish Diabetes Registry has included information on risk factors, drugs, and complications and is described in detail elsewhere.9 13 17 More than 95% of Swedish children and adolescents with type 1 diabetes are registered in the Swediabkids database, which consists of outpatient data from all paediatric diabetes centres in Sweden.18 Since 2007 the registry has included data on almost all (98%) children and adolescents with diabetes in Sweden. In this study we included information from the Swedish Diabetes Registry for 1998 onwards and from Swediabkids for 2000 onwards.
Study cohort
In the Swedish Diabetes Registry, type 1 diabetes was defined as treatment with insulin and diagnosis at age 30 years or younger, and this definition has been validated in 97% of cases.17 We also required that a clinician had recorded a diagnosis of type 1 diabetes in the registry. Swediabkids used the recorded diagnosis of type 1 diabetes. HLA and autoantibodies are determined in all children with newly diagnosed diabetes.
We included children and adults with a diagnosis of diabetes for five or less years when first recorded in the registries. Endpoints of retinopathy were classified as any retinopathy, preproliferative diabetic retinopathy or worse, and proliferative diabetic retinopathy. Any retinopathy included any signs of retinopathy (simplex, preproliferative, or proliferative). Proliferative diabetic retinopathy was defined as evidence of current proliferations or earlier laser photocoagulation. Microalbuminuria was defined as two positive test results from three samples taken within one year, with an albumin:creatinine ratio of 3-30 mg/mmol or urinary albumin of 20-200 µg/min (20-300 mg/L), and macroalbuminuria as an albumin:creatinine ratio >30 mg/mmol or urinary albumin >200 µg/min (>300 mg/L).
Procedures
As follow-up periods differed for participants because of being included in the register at different times, we categorised subcohorts by follow-up time from diabetes onset: 8-9, 10-11, 12-13, 14-15, and 16-20 years. We followed participants from the first observation until first retinopathy or nephropathy event, 31 December 2017, or end of each follow-up if censored earlier. Participants were categorised by glycaemic control based on area under the curve, a weighted mean value calculated using the trapezoidal method that takes into consideration the interval between HbA1c measurements.5 10 Descriptive data were analysed for each cohort. To increase study power and obtain overall effects of HbA1c categories we pooled estimates for the primary analysis of HbA1c levels and risk of complications.
Analyses of HbA1c are reported according to the International Federation of Clinical Chemistry standard, measured in mmol/mol. Laboratory methods at participating care units for analysing HbA1c level and albuminuria are regularly checked with central reference samples of HbA1c and albuminuria to ensure high accuracy.19 20 HbA1c values were converted to levels according to the National Glycohemoglobin Standardization Program for dual reporting.21 We evaluated HbA1c as a categorical variable (<6.5% (<48 mmol/mol), 6.5-6.9 (48-52 mmol/mol), 7.0-7.4 (53-57 mmol/mol), 7.5-8.6 (58-70 mmol/mol), and >8.6% (>70 mmol/mol) and estimated risk of complications in relation to HbA1c level as a continuous variable for each 1% (10 mmol/mol). We performed unadjusted analyses and in the main model we adjusted for age, sex, duration of diabetes, blood pressure, blood lipid levels, body mass index, and smoking.
Since 2008 severe hypoglycaemias (defined as unconsciousness or seizures) have been recorded in Swediabkids and were analysed at each visit according to HbA1c categories in three steps. Firstly, participants were followed-up from first available hypoglycaemia data from 2008 and onwards, with HbA1c area under the curve counted from earliest one year before first registration of hypoglycaemic data (main hypoglycaemia analysis). Secondly, participants were included in the main hypoglycaemia analysis, including those with date of diabetes onset on or after first registration of hypoglycaemic data (first sensitivity hypoglycaemia analysis). Finally, only participants who were part of the main retinopathy and albuminuria analyses were included in the second sensitivity hypoglycaemia analysis.
Statistical analysis
We present numbers and percentages (with 95% confidence intervals) of complications for each category of HbA1c. Logistic regression was used to estimate the relation between HbA1c level and diabetes complications using odds ratios (95% confidence intervals) as risk estimates for different follow-up cohorts, and c statistics for goodness of fit. Analyses were performed by categories of HbA1c level, with 6.5-6.9% (48-52 mmol/mol) as the reference category and as a continuous variable for each 1% (10 mmol/mol) HbA1c, with splines also used to examine non-linear effects. We applied Firth’s penalised maximum likelihood estimation to reduce bias in the parameter estimates in the analyses with low number of events, leading to non-estimable coefficients or 95% confidence intervals. Generalised estimating equations modelling was used to estimate an overall effect of HbA1c categories on studied endpoints and to adjust for within patient correlation for repeated data over the follow-up cohorts.22 23 This model therefore allows a participant to be included in more than one cohort. We used unstructured covariance matrix and binomial distribution with logit link function, resulting in odds ratios (95% confidence intervals) as risk estimates.
Hypoglycaemic events were analysed using Poisson regression with log-link function and time from onset to event or last follow-up as an offset timing variable, to have event rates as the basis of the calculations instead of experience of an event only. Relative risks (95% confidence intervals) are provided as effect size.
No data were imputed. Tests were two tailed and conducted at 0.05 significance level. All statistical programming was performed using SAS Software version 9.4 (SAS Institute, Cary, NC).
Patient and public involvement
This research was done without patient involvement. Patients were not invited to comment on the study design and were not consulted to develop patient relevant outcomes or interpret the results. Patients were not invited to contribute to the writing or editing of this document for readability or accuracy.
Results
Participant characteristics
In total 10 398/11 009 (94.4%) children and adults with type 1 diabetes for more than eight years and early onset diabetes had information on HbA1c level and complications and were included in the analyses (see supplementary figure 1). The mean duration of diabetes at registration was 1.3 years (median 0.4 years) and at last follow-up was 11.9 years (median 10.4 years). Mean age at first visit was 14.7 years, 43.4% (4515/10 398) were female, and mean HbA1c area under the curve during follow-up was 8.0% (63.4 mmol/mol), with a mean number of HbA1c values per year of 2.7 (median 2.7). Table 1 presents the characteristic of participants by cohort (see supplementary table 1 for data used as covariates in analyses).
Table 1.
Patient characteristics for population with recorded retinopathy and albuminuria data at a clinical visit during selected years follow-up from diabetes onset. Values are numbers (percentages) unless stated otherwise
| Variables | Duration of follow-up (years) | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 8-9 | 10-11 | 12-13 | 14-15 | 16-20 | ||||||||||
| Retinopathy cohort (n=8049) | Albuminuria cohort (n=6476) | Retinopathy cohort (n=5324) | Albuminuria cohort (n=4490) | Retinopathy cohort (n=3044) | Albuminuria cohort (n=2642) | Retinopathy cohort (n=1927) | Albuminuria cohort (n=1959) | Retinopathy cohort (n=1172) | Albuminuria cohort (n=1262) | |||||
| Age at first visit (years): | ||||||||||||||
| Mean (SD) | 14.7 (7.9) | 18.1 (7.3) | 12.6 (7.1) | 15.1 (7.1) | 9.19 (3.67) | 10.1 (3.4) | 8.88 (3.40) | 9.34 (3.29) | 8.65 (3.12) | 8.84 (3.20) | ||||
| Median (min-max) | 13.0 (0.0-34.0) | 18.0 (0.6-34.0) | 11.1 (0.0-34.0) | 13.2 (0.0-34.0) | 9.24 (0.00-19.71) | 10.1 (0.4-19.9) | 8.91 (0.00-19.71) | 9.43 (0.66-19.89) | 8.65 (0.79-19.70) | 8.84 (0.79-19.71) | ||||
| Female | 3481 (43.2) | 2748 (42.4) | 2412 (45.3) | 2036 (45.3) | 1447 (47.5) | 1301 (49.2) | 941 (48.8) | 977 (49.9) | 566 (48.3) | 608 (48.2) | ||||
| Year of onset: | n=8046 | n=6476 | n=5319 | n=4490 | n=3042 | n=2642 | n=1925 | n=1959 | n=1166 | n=1262 | ||||
| Mean (SD) | 2003 (4) | 2004 (4) | 2003 (3) | 2003 (3) | 2002 (2) | 2002 (2) | 2000 (2) | 2000 (2) | 1999 (2) | 1999 (2) | ||||
| Median (min-max) | 2004 (1993-2009) | 2004 (1993-2009) | 2003 (1993-2007) | 2004 (1993-2007) | 2002 (1995-2005) | 2002 (1995-2005) | 2001 (1995-2003) | 2001 (1995-2003) | 1999 (1995-2001) | 1999 (1995-2001) | ||||
| HbA1c (mmol/mol): | ||||||||||||||
| Mean (SD) | 62.4 (11.2) | 62.2 (12.2) | 63.7 (10.7) | 63.9 (11.7) | 65.6 (10.0) | 66.3 (10.5) | 66.3 (9.9) | 67.0 (10.3) | 66.7 (10.3) | 67.0 (10.3) | ||||
| Median (min-max) | 61.5 (29.0-124.3) | 61.0 (29.0-124.3) | 62.9 (29.1-129.7) | 62.8 (29.1-129.7) | 64.5 (33.9-124.7) | 65.0 (33.9-124.7) | 65.0 (41.2-116.0) | 65.5 (41.2-120.9) | 65.4 (44.0-108.6) | 65.6 (44.0-109.9) | ||||
| HbA1c (%): | ||||||||||||||
| Mean (SD) | 7.86 (1.02) | 7.84 (1.11) | 7.98 (0.98) | 8.00 (1.07) | 8.16 (0.91) | 8.22 (0.96) | 8.22 (0.91) | 8.28 (0.94) | 8.26 (0.94) | 8.28 (0.94) | ||||
| Median (min-max) | 7.78 (4.81-13.53) | 7.73 (4.81-13.53) | 7.90 (4.81-14.02) | 7.90 (4.81-14.02) | 8.06 (5.25-13.57) | 8.10 (5.25-13.57) | 8.10 (5.92-12.77) | 8.15 (5.92-13.21) | 8.14 (6.18-12.09) | 8.15 (6.18-12.21) | ||||
| HbA1c AUC category*: | ||||||||||||||
| <6.5% | 625 (7.8) | 661 (10.2) | 271 (5.1) | 277 (6.2) | 49 (1.6) | 45 (1.7) | 20 (1.0) | 21 (1.1) | 10 (0.9) | 8 (0.6) | ||||
| 6.5-6.9% | 871 (10.8) | 789 (12.2) | 489 (9.2) | 452 (10.1) | 186 (6.1) | 157 (5.9) | 101 (5.2) | 89 (4.5) | 61 (5.2) | 62 (4.9) | ||||
| 7.0-7.4% | 1415 (17.6) | 1106 (17.1) | 858 (16.1) | 702 (15.6) | 434 (14.3) | 364 (13.8) | 244 (12.7) | 238 (12.1) | 152 (13.0) | 163 (12.9) | ||||
| 7.5-8.6% | 3420 (42.5) | 2429 (37.5) | 2408 (45.2) | 1862 (41.5) | 1534 (50.4) | 1268 (48.0) | 983 (51.0) | 974 (49.7) | 587 (50.1) | 623 (49.4) | ||||
| >8.6% | 1718 (21.3) | 1491 (23.0) | 1298 (24.4) | 1197 (26.7) | 841 (27.6) | 808 (30.6) | 579 (30.0) | 637 (32.5) | 362 (30.9) | 406 (32.2) | ||||
AUC=area under the curve.
<6.5% (<48 mmol/mol); 6.5-6.9% (48-52 mmol/mol); 7.0-7.4% (53-57 mmol/mol); 7.5-8.6% (58-70 mmol/mol); >8.6% (>70 mmol/mol).
HbA1c level and microvascular complications
Table 2 shows the unadjusted and adjusted risk of retinopathy (simplex, preproliferative, and proliferative) in relation to HbA1c level of 6.5-6.9% (48-52 mmol/mol) for all follow-up cohorts using generalised estimating equation models and number of events. Overall, 9358 children and adults were included in analyses of retinopathy of whom 3115 (33.3%) had any retinopathy event. The number of participants with preproliferative diabetic retinopathy was 284 (3.0%) and with proliferative diabetic retinopathy was 106 (1.1%). The risk of any retinopathy (simplex or worse) for participants with an HbA1c level <6.5% (<48 mmol/mol) compared with 6.5-6.9% (48-52 mmol/mol) did not differ: unadjusted odds ratio 0.85 (95% confidence interval 0.63 to 1.13, P=0.25) and adjusted odds ratio 0.77 (0.56 to 1.05, P=0.10). The risk of retinopathy was increased in participants with HbA1c levels of 7.0-7.4% (53-57 mmol/mol), with an adjusted odds ratio of 1.31 (1.05 to 1.64, P=0.02), and the risk was higher in those with HbA1c levels of 7.5-8.6% (58-70 mmol/mol) and >8.6% (>70 mmol/mol) (table 2).
Table 2.
Generalised estimating equation models for association between glycated haemoglobin (HbA1c) categories on diabetic retinopathy and albuminuria endpoints pooled over all studied periods
| Endpoint by HbA1c category* | No of events | % (95% CI) | No of participants with events | % (95% CI) | Unadjusted | Adjusted† | |||
|---|---|---|---|---|---|---|---|---|---|
| Odds ratio (95% CI) | P value | Odds ratio (95% CI) | P value | ||||||
| Any retinopathy v none: | |||||||||
| <6.5% | 85 | 8.7 (7.0 to 10.7) | 75 | 11.0 (8.9 to 13.7) | 0.85 (0.63 to 1.13) | 0.25 | 0.77 (0.56 to 1.05) | 0.10 | |
| 6.5-6.9% | 214 | 12.5 (11.0 to 14.2) | 159 | 14.7 (12.7 to 17.0) | 1.00 (Ref) | 1.00 (Ref) | |||
| 7.0-7.4% | 480 | 15.5 (14.2 to 16.8) | 364 | 20.0 (18.2 to 21.9) | 1.25 (1.03 to 1.52) | 0.02 | 1.31 (1.05 to 1.64) | 0.02 | |
| 7.5-8.6% | 2199 | 24.6 (23.7 to 25.5) | 1445 | 33.1 (31.7 to 34.5) | 2.01 (1.68 to 2.40) | <0.001 | 2.05 (1.68 to 2.51) | <0.001 | |
| >8.6% | 2006 | 41.8 (40.4 to 43.2) | 1211 | 51.0 (49.0 to 53.0) | 4.11 (3.43 to 4.93) | <0.001 | 3.72 (3.02 to 4.58) | <0.001 | |
| PPDR/PDR/laser photocoagulation v none/simplex: | |||||||||
| <6.5% | 8 | 0.8 (0.4 to 1.6) | 7 | 1.0 (0.5 to 2.3) | 3.74 (1.19 to 11.76) | 0.02 | 3.29 (0.99 to 10.96) | 0.05 | |
| 6.5-6.9% | 5 | 0.3 (0.1 to 0.7) | 5 | 0.5 (0.2 to 1.2) | 1.00 (Ref) | 1.00 (Ref) | |||
| 7.0-7.4% | 12 | 0.4 (0.2 to 0.7) | 9 | 0.5 (0.3 to 1.0) | 1.12 (0.36 to 3.50) | 0.85 | 1.57 (0.49 to 5.03) | 0.45 | |
| 7.5-8.6% | 92 | 1.0 (0.8 to 1.3) | 78 | 1.8 (1.4 to 2.2) | 2.77 (1.08 to 7.08) | 0.03 | 3.98 (1.44 to 10.98) | 0.008 | |
| >8.6% | 250 | 5.2 (4.6 to 5.9) | 186 | 7.8 (6.8 to 9.0) | 13.44 (5.34 to 33.85) | <0.001 | 13.77 (5.08 to 37.35) | <0.001 | |
| PDR/laser photocoagulation v none/simplex/PPDR: | |||||||||
| <6.5% | 6 | 0.6 (0.2 to 1.3) | 6 | 0.9 (0.4 to 2.1) | 4.18 (1.17 to 14.90) | 0.03 | 2.48 (0.71 to 8.62) | 0.15 | |
| 6.5-6.9% | 4 | 0.2 (0.1 to 0.6) | 4 | 0.4 (0.2 to 1.1) | 1.00 (Ref) | 1.00 (Ref) | |||
| 7.0-7.4% | 7 | 0.2 (0.1 to 0.5) | 5 | 0.3 (0.1 to 0.7) | 0.85 (0.22 to 3.26) | 0.82 | 0.80 (0.20 to 3.22) | 0.75 | |
| 7.5-8.6% | 24 | 0.3 (0.2 to 0.4) | 20 | 0.5 (0.3 to 0.7) | 0.97 (0.32 to 2.96) | 0.96 | 1.14 (0.37 to 3.50) | 0.82 | |
| >8.6% | 93 | 1.9 (1.6 to 2.4) | 71 | 3.0 (2.4 to 3.8) | 6.32 (2.21 to 18.11) | <0.001 | 5.98 (2.10 to 17.06) | <0.001 | |
| Microalbuminuria/macroalbuminuria v none: | |||||||||
| <6.5% | 35 | 3.5 (2.4 to 4.8) | 28 | 3.9 (2.7 to 5.7) | 1.04 (0.66 to 1.66) | 0.86 | 0.98 (0.60 to 1.61) | 0.95 | |
| 6.5-6.9% | 53 | 3.4 (2.6 to 4.5) | 40 | 3.8 (2.8 to 5.3) | 1.00 (Ref) | 1.00 (Ref) | |||
| 7.0-7.4% | 130 | 5.1 (4.2 to 6.0) | 99 | 5.9 (4.9 to 7.2) | 1.41 (0.98 to 2.03) | 0.06 | 1.55 (1.03 to 2.32) | 0.03 | |
| 7.5-8.6% | 328 | 4.6 (4.1 to 5.1) | 256 | 6.2 (5.5 to 7.0) | 1.41 (1.01 to 1.98) | 0.04 | 1.33 (0.91 to 1.94) | 0.14 | |
| >8.6% | 481 | 10.6 (9.7 to 11.5) | 329 | 13.4 (12.1 to 14.9) | 3.16 (2.27 to 4.41) | <0.001 | 2.64 (1.81 to 3.86) | <0.001 | |
| Macroalbuminuria v none/microalbuminuria: | |||||||||
| <6.5% | 9 | 0.9 (0.4 to 1.7) | 7 | 1.0 (0.5 to 2.2) | 1.95 (0.67 to 5.74) | 0.22 | 2.47 (0.69 to 8.87) | 0.17 | |
| 6.5-6.9% | 6 | 0.4 (0.1 to 0.8) | 6 | 0.6 (0.3 to 1.4) | 1.00 (Ref) | 1.00 (Ref) | |||
| 7.0-7.4% | 18 | 0.7 (0.4 to 1.1) | 15 | 0.9 (0.5 to 1.5) | 1.70 (0.70 to 4.12) | 0.24 | 2.47 (0.83 to 7.35) | 0.10 | |
| 7.5-8.6% | 56 | 0.8 (0.6 to 1.0) | 49 | 1.2 (0.9 to 1.6) | 1.88 (0.82 to 4.31) | 0.14 | 2.11 (0.73 to 6.14) | 0.17 | |
| >8.6% | 74 | 1.6 (1.3 to 2.0) | 55 | 2.2 (1.7 to 3.0) | 3.67 (1.62 to 8.30) | 0.002 | 3.43 (1.14 to 10.26) | 0.03 | |
PPDR=preproliferative diabetic retinopathy; PDR=proliferative diabetic retinopathy.
<6.5% (<48 mmol/mol); 6.5-6.9% (48-52 mmol/mol); 7.0-7.4% (53-57 mmol/mol); 7.5-8.6% (58-70 mmol/mol); >8.6% (>70 mmol/mol).
Adjusted for age; sex; mean systolic and diastolic blood pressure, body mass index, high and low density lipoprotein cholesterol, cholesterol, and triglycerides; and smoking.
A different pattern was observed for preproliferative diabetic retinopathy or worse, with risk increases in participants with low HbA1c levels (<6.5%, <48 mmol/mol) in unadjusted analysis (odds ratio 3.74, 1.19 to 11.76, P=0.02) and in adjusted analysis (3.29, 0.99 to 10.96, P=0.05), with a risk increase observed for higher HbA1c levels starting at 7.5-8.6% (58-70 mmol/mol): 3.98 (1.44 to 10.98, P=0.008). For the most advanced stage (proliferative diabetic retinopathy or earlier laser photocoagulation), risk increases were observed for participants with low HbA1c levels (<6.5%, <48 mmol/mol): odds ratio 4.18 (1.17 to 14.90, P=0.03) in unadjusted analysis and 2.48 (0.71 to 8.62, P=0.15) in adjusted analysis. This pattern became apparent at high HbA1c levels and was significant at >8.6% (>70 mmol/mol): 5.98 (2.10 to 17.06, P<0.001).
Overall, 9347 children and adults were included in analyses of microalbuminuria (n=737, 7.9%) and macroalbuminuria (n=132, 1.4%). The risk for microalbuminuria and macroalbuminuria did not differ between the two lowest (<6.5% (<48 mmol/mol) and 6.5-6.9% (48-52 mmol/mol)) HbA1c categories (table 2). The risk of microalbuminuria increased at HbA1c levels 7.0-7.4% (53-57 mmol/mol) and >8.6% (>70 mmol/mol), with adjusted odds ratios of 1.55 (95% confidence interval 1.03 to 2.32, P=0.03) and 2.64 (1.81 to 3.86, P<0.001), respectively. The risk of macroalbuminuria increased at HbA1c levels >8.6% (>70 mmol/mol) compared with HbA1c levels 6.5-6.9% (48-52 mmol/mol), with an adjusted odds ratio of 3.43 (1.14 to 10.26, P=0.03).
Supplementary tables 2.1 and 2.2 present descriptive data for each cohort in the pooled estimations of retinopathy and nephropathy.
Risk of complications over time
The risk of any retinopathy, preproliferative diabetic retinopathy, and proliferative diabetic retinopathy by 1% (10 mmol/mol) higher HbA1c level increased with longer follow-up (fig 1). The odds ratios for any retinopathy increased from 1.51 (1.42 to 1.62, P<0.001) for 8-10 years to 1.78 (1.51 to 2.09, P<0.001) for 16-20 years of follow-up and for proliferative diabetic retinopathy from 1.31 (0.97 to 1.77, P=0.07) to 2.87 (2.18 to 3.79, P<0.001), respectively. Supplementary table 2.1 shows the crude figures for unadjusted and adjusted odds ratios and corresponding 95% confidence intervals for the values reported in figure 1. Moreover, spline functions (ie, allowing non-linear effects of HbA1c as a continuous variable in relation to complications) indicated that the HbA1c level associated with severe retinopathy decreased over time (fig 2). This trend was most pronounced for the longest follow-up period, of 16-20 years.
Fig 1.

Adjusted odds ratios (95% confidence intervals) for retinopathy and nephropathy for 10 mmol/mol increase in glycated haemoglobin (HbA1c) area under curve. y axis is on logarithmic scale. Proliferative diabetic retinopathy/laser photocoagulation and macroalbuminuria adjusted only for age and sex owing to small number of events; other outcomes adjusted for age, sex, mean systolic and mean diastolic blood pressure, mean body mass index, mean high and mean low density lipoprotein cholesterol, mean cholesterol, mean triglycerides, and smoking. PPDR=preproliferative diabetic retinopathy; PDR=proliferative diabetic retinopathy. Whiskers represent 95% confidence intervals
Fig 2.
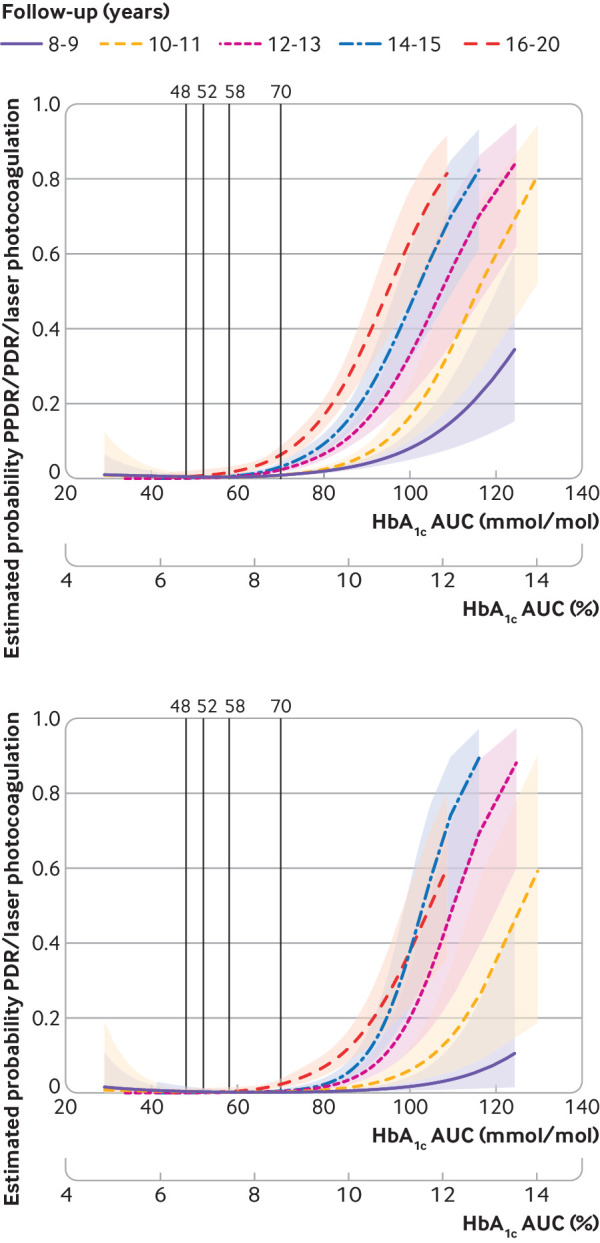
Estimated cumulative probability for preproliferative diabetic retinopathy or worse (PPDR), proliferative diabetic retinopathy (PDR), and laser photocoagulation for different area under the curve (AUC) glycated haemoglobin (HbA1c) values allowing for non-linear relation using spline functions for follow-up cohorts. Shaded areas represent 95% confidence intervals
The odds ratios for microalbuminuria and macroalbuminuria increased for each 1% (10 mmol/mol) higher HbA1c value during follow-up (fig 1, supplementary table 2.2), ranging from 1.31 (1.20 to 1.43, P<0.001) and 1.23 (1.04 to 1.46, P=0.02) for 8-10 years to 2.14 (1.71 to 2.68, P<0.001) and 3.83 (2.58 to 5.70, P<0.001) for 16-20 years, respectively.
Microvascular complications in relation to age and sex
After adjustment for HbA1c categories and sex, older age at first visit was associated with increased risk for retinopathy and nephropathy; odds ratio for each five years increase was 1.18 for any retinopathy; 1.72 for preproliferative diabetic retinopathy or worse, proliferative diabetic retinopathy, or laser photocoagulation; 1.95 for proliferative diabetic retinopathy or laser photocoagulation; 1.20 for microalbuminuria or macroalbuminuria; and 1.36 for macroalbuminuria; all P values <0.001.
Risk for retinopathy was not related to sex, but in females the risk was increased for microalbuminuria or macroalbuminuria (1.30, 1.11 to 1.53, P=0.001) and macroalbuminuria (1.51, 1.06 to 2.16, P=0.02).
Proliferative diabetic retinopathy and nephropathy at low HbA1c levels
Post hoc descriptive analyses were carried out on seven participants with macroalbuminuria and six with proliferative diabetic retinopathy (12 participants in total), within the HbA1c category <6.5% (<48 mmol/mol) (see supplementary table 1). Mean time from diabetes onset to first HbA1c measurement was 1.6 years (median 0.8 years) for those with HbA1c levels <6.5% (<48 mmol/mol) and no specified endpoints and 2.2 years (median 2.3 years) for those with any of the specified endpoints. Mean number of HbA1c measurements annually ranged between 1.7 and 2.7 among the cohorts, mean age at first visit 20.2 versus 22.7 years, smoking 80/769 (11.3%) versus 0/12 (0%), systolic blood pressure 117.9 versus 119.6 mm Hg, diastolic blood pressure 70.5 versus 72.0 mm Hg, body mass index 23.1 versus 21.9, high density lipoprotein cholesterol 1.6 versus 1.6 mmol/L, and low density lipoprotein cholesterol 2.4 versus 2.1 mmol/L, respectively.
Severe hypoglycaemias
The risk of severe hypoglycaemia was observed to be increased at mean HbA1c <6.5% (<48 mmol/mol) compared with 6.5-6.9% (48-52 mmol/mol, relative risk 1.34, 95% confidence interval 1.09 to 1.64, P=0.005). An association was also found between lower risk of severe hypoglycaemia with higher HbA1c level (0.79 (0.75 to 0.83) for each 1% (10 mmol/mol) higher mean HbA1c, P<0.001). Compared with the reference category 6.5-6.9% (48-52 mmol/mol), the relative risk for the highest category of HbA1c >8.6% (>70 mmol/mol) was 0.53 (95% confidence interval 0.43 to 0.64, P<0.001). The two sensitivity analyses led to similar conclusions to the main hypoglycaemia analysis (table 3).
Table 3.
Poisson regression for impact of continuous glycated haemoglobin (HbA1c) and categories on severe hypoglycaemic events
| Population/analysis and comparison | Relative risk (95% CI) | P value | |
|---|---|---|---|
| Swediabkids main: from first available hypoglycaemia data 2008 onwards with HbA1c counted from earliest 1 year before first registration of hypoglycaemic data n/N (%) events 1398/13413 (10.4) | |||
| HbA1c AUC category*: | |||
| By 1% increase | 0.79 (0.75 to 0.83) | <0.001 | |
| 6.5-6.9% v 6.5-6.9% | 1.00 (Ref) | ||
| <6.5% v 6.5-6.9% | 1.34 (1.09 to 1.64) | 0.005 | |
| 7.0-7.4% v 6.5-6.9% | 0.88 (0.74 to 1.06) | 0.19 | |
| 7.5-8.6% v 6.5-6.9% | 0.83 (0.71 to 0.98) | 0.03 | |
| >8.6% v 6.5-6.9% | 0.53 (0.43 to 0.64) | <0.001 | |
| Swediabkids first sensitivity analysis, main including those participants with onset date on or after first registration of hypoglycaemic data n/N (%) events725/8271 (8.8%) | |||
| By 1% increase | 0.88 (0.81 to 0.96) | 0.003 | |
| 6.5-6.9% v 6.5-6.9% | 1.00 (Ref) | ||
| <6.5% v 6.5-6.9% | 1.30 (1.04 to 1.63) | 0.02 | |
| 7.0-7.4% v 6.5-6.9% | 0.91 (0.73 to 1.14) | 0.40 | |
| 7.5-8.6% v 6.5-6.9% | 0.96 (0.78 to 1.18) | 0.68 | |
| >8.6% v 6.5-6.9% | 0.79 (0.56 to 1.12) | 0.18 | |
| Swediabkids second sensitivity analysis−first sensitivity+including only those participants from the main retinopathy/albuminuria analyses n/N (%) events: 217/1359 (16.0%) | |||
| By 1% increase | 0.75 (0.64 to 0.88) | <0.001 | |
| 6.5-6.9% v 6.5-6.9% | 1.00 (Ref) | ||
| <6.5% v 6.5-6.9% | 2.10 (1.32 to 3.34) | 0.002 | |
| 7.0-7.4% v 6.5-6.9% | 0.86 (0.55 to 1.34) | 0.51 | |
| 7.5-8.6% v 6.5-6.9% | 0.91 (0.62 to 1.36) | 0.66 | |
| >8.6% v 6.5-6.9% | 0.58 (0.31 to 1.10) | 0.10 | |
AUC=area under the curve.
<6.5% (<48 mmol/mol); 6.5-6.9% (48-52 mmol/mol); 7.0-7.4% (53-57 mmol/mol); 7.5-8.6% (58-70 mmol/mol); >8.6% (>70 mmol/mol).
Discussion
This study of 10 398 children and adults followed for 8-20 years from diagnosis of type 1 diabetes or shortly thereafter did not find any statistically significant differences in risk for retinopathy or nephropathy for a mean glycated haemoglobin (HbA1c) level of <6.5% (<48 mmol/mol) compared with 6.5-6.9% (48-52 mmol/mol). Any retinopathy (defined as simplex or worse) and microalbuminuria became slightly more common at mean HbA1c levels greater than 7.0% (53 mmol/mol), whereas increasing risk of preproliferative diabetic retinopathy (moderately severe) started at HbA1c levels around 7.5% (58 mmol/mol). Severe complications in the form of proliferative diabetic retinopathy and macroalbuminuria first became apparent at mean HbA1c levels >8.6% (>70 mmol/mol). Severe hypoglycaemia increased with lower HbA1c levels and was greater at levels <6.5% (<48 mmol/mol) compared with 6.5-6.9% (48-52 mmol/mol).
Comparison with other studies
Multiple studies of HbA1c related to diabetes complications, both microvascular and macrovascular, have been performed.1 2 3 4 5 6 7 8 9 10 To fully understand the impact of HbA1c levels, studies should preferentially be performed in larger populations and follow participants from childhood, when onset of type 1 diabetes generally occurs, to adulthood, when complications most often exist.3 4 10 The Diabetes Control and Complications Trial is evaluating participants in the primary prevention cohort (n=726) from diagnosis or closely thereafter and showed the strongest relations between HbA1c and complications in this cohort compared with the secondary intervention cohort including people with longer duration of diabetes and existing complications.1 2 24 About half of the participants in the Diabetes Control and Complications Trial were treated with multiple dose insulin injections or insulin pumps, whereas the rest had only basal insulin. Because relatively few complications occurred at lower HbA1c levels, however, it is difficult to distinguish between differences in risk at low HbA1c levels, such as <6.5% (<48 mmol/mol) compared with <7.0% (<53 mmol/mol). A study that followed 451 participants from diagnosis to 20-24 years onwards found no cases of proliferative diabetic retinopathy at a mean HbA1c level of <7.6% (<60 mmol/mol), and no participant developed macroproteinuria at HbA1c <8.4% (<68 mmol/mol).4 The current study complements earlier studies as it is larger, population based, and includes participants who used modern glucose lowering treatments.
HbA1c targets
Currently, HbA1c targets vary in diabetes guidelines worldwide.11 12 13 14 In the US, the American Diabetes Association guidelines recommend an HbA1c of <7.5% (<58 mmol/mol) for children and <7.0% (<53 mmol/mol) for adults.11 In the UK, the National Institute for Health and Care Excellence recommends <6.5% (<48 mmol/mol) for both children and adults, whereas Swedish guidelines recommend <6.5% (<48 mmol/mol) for children and <7.0% (<53 mmol/mol) for adults.12 13 The International Society for Pediatric and Adolescent Diabetes has recently lowered the HbA1c target from <7.5% (<58 mmol/mol) to <7.0% (<53 mmol/mol).14
Clinical implications
Reaching lower HbA1c targets is associated with increased effort by people with type 1 diabetes15 and not least for carers of children with diabetes.16 Glucose levels must be checked often, insulin administered frequently, and certain things undertaken, such as following a suitable diet or ensuring physical activity. Both children and their parents often need to make substantial efforts in their daily lives, which can include overnight glucose monitoring and extra insulin doses to reach HbA1c targets. People with type 1 diabetes can be frustrated from having to monitor the disease, and diabetes can be associated with increased stress.15 16 It is therefore clinically important to know if a low HbA1c target of <6.5% (<48 mmol/mol) can prevent microvascular complications more than a HbA1c target of <7.0% (<53 mmol/mol).
In this study, preproliferative diabetic retinopathy or worse occurred in seven participants with a mean HbA1c level of <6.5% (<48 mmol/mol), which was associated with an increased risk of borderline significance (P=0.05) versus an HbA1c level of 6.5-6.9% (48-52 mmol/mol), and seven participants developed macroalbuminuria. It seems unlikely that low HbA1c levels indicating glucose levels close to normal should be harmful in themselves; however, preclinical studies have indicated that microvascular complications might be promoted by frequent hypoglycaemia, as is possibly the case with rapid glucose fluctuations that can be related to hypoglycaemia.25 26 Moreover, we observed an increased risk of severe hypoglycaemia with HbA1c levels <6.5% (<48 mmol/mol) compared with 6.5-6.9% (48-52 mmol/mol).
Hence the current findings suggest that clinicians should be extra vigilant about ensuring people with diabetes do not spend considerable time in hypoglycaemia and that treatment is related to good quality of life at HbA1c levels <6.5% (<48 mmol/mol) or else aim at higher levels of 6.5-6.9% (48-52 mmol/mol). The rationale being that we do not find any further reduction of risk for microvascular complications at HbA1c levels <6.5% (<48 mmol/mol) in this large cohort compared with levels of 6.5-6.9% (48-52 mmol/mol).
Future studies
In future studies it will be important to include data based on continuous and flash glucose monitors over a long period.27 In such studies it will also be important to evaluate complementary glycaemic metrics to HbA1c, including measures for euglycaemia, time in hypoglycaemia, and glycaemic variability.
HbA1c levels and cardiovascular disease
When discussing at what HbA1c levels clinicians should be aware that risk reductions exist and evidence from guidelines it is essential to also discuss relations between HbA1c level and cardiovascular disease. In a recent study of more than 30 000 people with type 1 diabetes, risk estimates for myocardial infarction were in principle identical for those with mean HbA1c levels <6.5% (<48 mmol/mol) compared with 6.5-7.0% (48-53 mmol/mol).9 Although information on mean level of HbA1c was available over about nine years, a limitation was that glycaemic control further back in the patient´s history was not known in many patients, known to be crucial for future complications termed metabolic memory or legacy effects.28
HbA1c levels and advanced microvascular complications
In the current study, complications were statistically significantly more advanced in participants with HbA1c levels >8.6% (>70 mmol/mol)—that is, proliferative diabetic retinopathy and macroalbuminuria at 10-20 years after diagnosis. Also, the tendency was for more advanced complications to appear after 16-20 years at lower HbA1c levels. Hence clinicians need to focus on lowering high HbA1c levels at least to moderately increased levels in addition to targeting HbA1c.
Exposure time and risk
The risk association between HbA1c level and complications increased with longer follow-up, and relative risks were generally higher than earlier described.1 2 3 4 5 6 7 8 During follow-up of 16-20 years, a 1% (10 mmol/mol) increase in HbA1c level resulted in an odds ratio of 2.87 for proliferative diabetic retinopathy and of 3.83 for macroalbuminuria. This is important for economic analyses and decision making about diabetes treatments as an essential part of health economic analyses within the specialty of diabetes is the association between HbA1c level and risk of complications.
Future treatments
Treatments for type 1 diabetes are emerging that could influence the possibilities of reaching low HbA1c levels with a low risk of hypoglycaemia in the future. Continuous glucose monitoring, in conjunction both with and without an insulin pump, have shown beneficial effects in reducing both HbA1c levels and time in hypoglycaemia and is becoming more commonly used among people with type 1 diabetes.29 30 31 32 Moreover, oral treatment with sodium-glucose cotransporter 2 inhibitors has shown beneficial effects in reducing HbA1c levels without increasing the risk of hypoglycaemia when added to insulin treatment in adults with type 1 diabetes.33 34 This treatment will likely soon be introduced in clinical practice. However, broad use might initially be limited owing to increased risk of diabetes ketoacidosis before it can be better understood who is at risk and how ketoacidosis can best be prevented.
Strengths and limitations of this study
The main strength of this study is follow-up of a large nationwide population from diagnosis of type 1 diabetes, with data collection up to 31 December 2017. Treatments have shifted over time and risk patterns might differ during recent decades, with more people receiving modern insulin analogues and continuous glucose monitoring known to reduce the risk of hypoglycaemia while lowering HbA1c levels.35 36 Although relatively few advanced complications occurred, the study was relatively highly powered to detect these, as shown by the narrow confidence intervals. Limitations include the decreasing number of people with longer follow-up and retrospective study design; the lack of more detailed information on hypoglycaemia, including continuous glucose monitoring results for time in hypoglycaemia; and information on insulin doses, pregnancy, and concurrent illnesses. Moreover, information on quality of life was not available. The current study did not include macrovascular endpoints since these events generally occur later in life and would require long follow-up. Microalbuminuria and macroalbuminuria evaluated here are some of the strongest risk factors for future cardiovascular risk and mortality.9 37 A further limitation is that we cannot exclude the influence of other renal diseases on albuminuria levels in certain cases. However, the strong risk gradients found between HbA1c level and nephropathy indicate a high overall quality of data, and the variables have been used in multiple earlier studies based on Swedish registry data.9 13 37
Conclusions
We found no difference in risk of retinopathy and nephropathy at HbA1c levels <6.5% (<48 mmol/mol) but an increased risk of severe hypoglycaemia compared with levels of 6.5-6.9% (48-52 mmol/mol). Severe complications mainly occurred at HbA1c levels >8.6% (>70 mmol/mol), whereas the risk of milder complications increased at HbA1c levels >7.0% (>53 mmol/mol). In people with HbA1c levels <6.5% (<48 mmol/mol), clinicians should focus on avoidance of frequent hypoglycaemia and obtaining good diabetes related quality of life, or else increase the target level to 6.5-6.9% (48-52 mmol/mol). Other measures, including time in range, time in hypoglycaemia, and variation in glycaemia will be essential complementary measures to HbA1c in clinical practice and future studies.
What is already known on this topic
Poor glycaemic control (high glycated haemoglobin (HbA1c) levels) is associated with increased risk of diabetes complications
Targets for glycaemic control in children and adults differ between developed countries
Low HbA1c levels could increase the risk of hypoglycaemia
What this study adds
This study found no decreased risk of diabetic retinopathy and nephropathy associated with HbA1c levels <6.5% (<48 mmol/mol) compared with 6.5-6.9% (48-52 mmol/mol) but an increased risk for severe hypoglycaemia—severe complications mainly occurred at HbA1c >8.6% and milder complications increased at >7.0%
The current findings support a general target of HbA1c <7.0% in people with type 1 diabetes
People who achieve HbA1c levels <6.5% should be vigilant about not spending too much time in hypoglycaemia and achieve a good diabetes related quality of life
Acknowledgments
We thank the clinicians and staff at Swediabkids and the Swedish Diabetes Register who collected data, and Ragnar Hanås for critical review of the manuscript.
Web extra.
Extra material supplied by authors
Supplementary information: Study flowchart, descriptive data for people with low HbA1c levels, and analyses on retinopathy/nephropathy endpoints for studied periods
Contributors: ML wrote the first draft of the manuscript. All authors were involved in the study design, interpretation of data, and review of the manuscript. ML, AP, and JL are the guarantors. ML attests that all listed authors meet authorship criteria and that no others meeting the criteria have been omitted.
Funding: This study was supported by the Swedish government (Agreement for Medical Education and Research) and Novonordisk Foundation. The funders had no role in the study design, data collection, data analysis, data interpretation, or writing of the report.
Competing interests: All authors have completed the ICMJE uniform disclosure form at www.icmje.org/coi_disclosure.pdf and declare: ML has received grants from AstraZeneca, Dexcom, Novo Nordisk, and Pfizer, and consulting fees from AstraZeneca, Dexcom, Eli Lilly, MSD, Novo Nordisk, and Rubin Medical; no financial relationships with any organisations that might have an interest in the submitted work in the previous three years, no other relationships or activities that could appear to have influenced the submitted work.
Ethical approval: This study was approved by the regional ethical and review board of the University of Gothenburg, Gothenburg, Sweden (Dnr 977-17).
Data sharing: Data may be obtained from the registries named in the methods section and are not publicly available.
The lead author (ML) affirms that this manuscript is an honest, accurate, and transparent account of the study being reported; that no important aspects of the study have been omitted; and that any discrepancies from the study as planned have been explained.
References
- 1. Nathan DM, Genuth S, Lachin J, et al. Diabetes Control and Complications Trial Research Group The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med 1993;329:977-86. 10.1056/NEJM199309303291401 [DOI] [PubMed] [Google Scholar]
- 2. The Diabetes Control and Complications Trial Research Group The relationship of glycemic exposure (HbA1c) to the risk of development and progression of retinopathy in the diabetes control and complications trial. Diabetes 1995;44:968-83. 10.2337/diab.44.8.968 [DOI] [PubMed] [Google Scholar]
- 3. Bojestig M, Arnqvist HJ, Hermansson G, Karlberg BE, Ludvigsson J. Declining incidence of nephropathy in insulin-dependent diabetes mellitus. N Engl J Med 1994;330:15-8. 10.1056/NEJM199401063300103 [DOI] [PubMed] [Google Scholar]
- 4. Nordwall M, Abrahamsson M, Dhir M, Fredrikson M, Ludvigsson J, Arnqvist HJ. Impact of HbA1c, followed from onset of type 1 diabetes, on the development of severe retinopathy and nephropathy: the VISS Study (Vascular Diabetic Complications in Southeast Sweden). Diabetes Care 2015;38:308-15. 10.2337/dc14-1203. [DOI] [PubMed] [Google Scholar]
- 5. Lind M, Odén A, Fahlén M, Eliasson B. A systematic review of HbA1c variables used in the study of diabetic complications. Diabetes Metab Syndr 2008;2:282-93 10.1016/j.dsx.2008.04.006. [DOI] [Google Scholar]
- 6. Wang SY, Andrews CA, Herman WH, Gardner TW, Stein JD. Incidence and Risk Factors for Developing Diabetic Retinopathy among Youths with Type 1 or Type 2 Diabetes throughout the United States. Ophthalmology 2017;124:424-30. 10.1016/j.ophtha.2016.10.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Grauslund J, Green A, Sjølie AK. Prevalence and 25 year incidence of proliferative retinopathy among Danish type 1 diabetic patients. Diabetologia 2009;52:1829-35. 10.1007/s00125-009-1450-4. [DOI] [PubMed] [Google Scholar]
- 8. Klein R, Knudtson MD, Lee KE, Gangnon R, Klein BE. The Wisconsin Epidemiologic Study of Diabetic Retinopathy: XXII the twenty-five-year progression of retinopathy in persons with type 1 diabetes. Ophthalmology 2008;115:1859-68. 10.1016/j.ophtha.2008.08.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Matuleviciene-Anängen V, Rosengren A, Svensson AM, et al. Glycaemic control and excess risk of major coronary events in persons with type 1 diabetes. Heart 2017;103:1687-95. 10.1136/heartjnl-2016-311050 [DOI] [PubMed] [Google Scholar]
- 10. Lind M, Odén A, Fahlén M, Eliasson B. The true value of HbA1c as a predictor of diabetic complications: simulations of HbA1c variables. PLoS One 2009;4:e4412. 10.1371/journal.pone.0004412 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. American Diabetes Association 6. Glycemic Targets: Standards of Medical Care in Diabetes-2018 . Diabetes Care 2018;41(Suppl 1):S55-64. [DOI] [PubMed] [Google Scholar]
- 12.National Institute for Health and Care Excellence. Type 1 diabetes in adults: diagnosis and management NICE guideline 26 August 2015 www.nice.org.uk/guidance/ng17 accessed 2019 Jul 9. [PubMed]
- 13.Gudbjörnsdottir S, Svensson A-M, Eliasson B. et al NDR report 2017 available online www.ndr.nu/pdfs/Arsrapport_NDR_2017.pdf accessed 2018 Oct 17.
- 14. DiMeglio LA, Acerini CL, Codner E, et al. ISPAD Clinical Practice Consensus Guidelines 2018: Glycemic control targets and glucose monitoring for children, adolescents, and young adults with diabetes. Pediatr Diabetes 2018;19(Suppl 27):105-14; [Epub ahead of print]. 10.1111/pedi.12737. [DOI] [PubMed] [Google Scholar]
- 15. Fisher L, Hessler D, Polonsky W, Strycker L, Masharani U, Peters A. Diabetes distress in adults with type 1 diabetes: Prevalence, incidence and change over time. J Diabetes Complications 2016;30:1123-8. 10.1016/j.jdiacomp.2016.03.032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Lindström C, Åman J, Norberg AL, Forssberg M, Anderzén-Carlsson A. “Mission Impossible”; the Mothering of a Child With Type 1 Diabetes - From the Perspective of Mothers Experiencing Burnout. J Pediatr Nurs 2017;36:149-56. 10.1016/j.pedn.2017.06.002. [DOI] [PubMed] [Google Scholar]
- 17. Eeg-Olofsson K, Cederholm J, Nilsson PM, et al. Glycemic control and cardiovascular disease in 7,454 patients with type 1 diabetes: an observational study from the Swedish National Diabetes Register (NDR). Diabetes Care 2010;33:1640-6. 10.2337/dc10-0398 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Samuelsson U, Åkesson K, Peterson A, Hanas R, Hanberger L. Continued improvement of metabolic control in Swedish pediatric diabetes care. Pediatr Diabetes 2018;19:150-7. 10.1111/pedi.12467. [DOI] [PubMed] [Google Scholar]
- 19. Nordin G. Accuracy of HbA1c as Monitored by External Quality Assessment and Compared With Patient Mean Values. J Diabetes Sci Technol 2018;12:771-9. 10.1177/1932296818785622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Lindblad B, Nordin G. External quality assessment of HbA1c and its effect on comparison between Swedish pediatric diabetes clinics. Experiences from the Swedish pediatric diabetes quality register (Swediabkids) and Equalis. Clin Chem Lab Med 2013;51:2045-52. 10.1515/cclm-2013-0226. [DOI] [PubMed] [Google Scholar]
- 21. Hanas R, John G, International HBA1c Consensus Committee 2010 consensus statement on the worldwide standardization of the hemoglobin A1C measurement. Diabetes Care 2010;33:1903-4. 10.2337/dc10-0953 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Liang KY, Zeger SL. Longitudinal Data Analysis Using Generalized Linear Models. Biometrika 1986;73:13-22 10.1093/biomet/73.1.13. [DOI] [Google Scholar]
- 23.Description of GEE-model and SAS code in SAS: https://support.sas.com/documentation/cdl/en/statug/63347/HTML/default/viewer.htm#statug_genmod_sect049.htm accessed 2019 Jul 9.
- 24. The Diabetes Control and Complications Trial Research Group The absence of a glycemic threshold for the development of long-term complications: the perspective of the Diabetes Control and Complications Trial. Diabetes 1996;45:1289-98. 10.2337/diab.45.10.1289 [DOI] [PubMed] [Google Scholar]
- 25. Umino Y, Cuenca N, Everhart D, Fernandez-Sanchez L, Barlow RB, Solessio E. Partial rescue of retinal function in chronically hypoglycemic mice. Invest Ophthalmol Vis Sci 2012;53:915-23. 10.1167/iovs.11-8787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Khan MI, Barlow RB, Weinstock RS. Acute hypoglycemia decreases central retinal function in the human eye. Vision Res 2011;51:1623-6. 10.1016/j.visres.2011.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Danne T, Nimri R, Battelino T, et al. International consensus on use of continuous glucose monitoring. Diabetes Care 2017;40:1631-40. 10.2337/dc17-1600 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Lachin JM, Genuth S, Cleary P, Davis MD, Nathan DM, Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications Research Group Retinopathy and nephropathy in patients with type 1 diabetes four years after a trial of intensive therapy. N Engl J Med 2000;342:381-9. 10.1056/NEJM200002103420603 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Lind M, Polonsky W, Hirsch IB, et al. Continuous Glucose Monitoring vs Conventional Therapy for Glycemic Control in Adults With Type 1 Diabetes Treated With Multiple Daily Insulin Injections: The GOLD Randomized Clinical Trial. JAMA 2017;317:379-87. 10.1001/jama.2016.19976 [DOI] [PubMed] [Google Scholar]
- 30. Battelino T, Conget I, Olsen B, et al. SWITCH Study Group The use and efficacy of continuous glucose monitoring in type 1 diabetes treated with insulin pump therapy: a randomised controlled trial. Diabetologia 2012;55:3155-62. 10.1007/s00125-012-2708-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Bergenstal RM, Klonoff DC, Garg SK, et al. ASPIRE In-Home Study Group Threshold-based insulin-pump interruption for reduction of hypoglycemia. N Engl J Med 2013;369:224-32. 10.1056/NEJMoa1303576. [DOI] [PubMed] [Google Scholar]
- 32. Forlenza GP, Pinhas-Hamiel O, Liljenquist DR, et al. Safety Evaluation of the MiniMed 670G System in Children 7-13 Years of Age with Type 1 Diabetes. Diabetes Technol Ther 2019;21:11-9. 10.1089/dia.2018.0264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Mathieu C, Dandona P, Gillard P, et al. DEPICT-2 Investigators Efficacy and Safety of Dapagliflozin in Patients With Inadequately Controlled Type 1 Diabetes (the DEPICT-2 Study): 24-Week Results From a Randomized Controlled Trial. Diabetes Care 2018;41:1938-46. 10.2337/dc18-0623. [DOI] [PubMed] [Google Scholar]
- 34. Mathieu C, Dandona P, Phillip M, et al. DEPICT-1 and DEPICT-2 Investigators Glucose Variables in Type 1 Diabetes Studies With Dapagliflozin: Pooled Analysis of Continuous Glucose Monitoring Data From DEPICT-1 and -2. Diabetes Care 2019;42:1081-7. 10.2337/dc18-1983. [DOI] [PubMed] [Google Scholar]
- 35. Laranjeira FO, de Andrade KRC, Figueiredo ACMG, Silva EN, Pereira MG. Long-acting insulin analogues for type 1 diabetes: An overview of systematic reviews and meta-analysis of randomized controlled trials. PLoS One 2018;13:e0194801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Joubert M, Reznik Y. Personal continuous glucose monitoring (CGM) in diabetes management: review of the literature and implementation for practical use. Diabetes Res Clin Pract 2012;96:294-305. 10.1016/j.diabres.2011.12.010. [DOI] [PubMed] [Google Scholar]
- 37. Lind M, Svensson AM, Kosiborod M, et al. Glycemic control and excess mortality in type 1 diabetes. N Engl J Med 2014;371:1972-82. 10.1056/NEJMoa1408214 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary information: Study flowchart, descriptive data for people with low HbA1c levels, and analyses on retinopathy/nephropathy endpoints for studied periods


