Abstract
Individuals with a family history of alcohol and other drug use disorders (FH+) are at increased risk for developing substance use disorders themselves relative to those with no such histories (FH−). Here we sought to identify key characteristics associated with FH+ status and alcohol and other drug use disorder status in a large cohort of FH+ and FH− young adults.
We conducted principal component analyses on demographic, temperament, and cognitive measures differentiating 506 FH+ and 528 FH− young adults. Three principal components were identified, and these component scores were then used to predict the odds of being FH+ and the odds of having an alcohol or other drug use disorder. Component 1 consisted of measures indexing internalizing traits, with higher component scores indicating greater depressive, anxious, and emotional instability tendencies. Component 2 consisted of measures of externalizing traits as well as exposure to early life adversity (ELA), with higher scores indicating less impulse control, more antisocial behavior, and greater ELA exposure. Component 3 consisted of estimated intelligence, delay discounting, and demographic characteristics, with higher scores indicating lower estimated intelligence, greater discounting of delayed rewards, less education, and lower childhood socioeconomic status. For each 1-point increase in the Component 1, 2, and 3 scores, the odds of being classified FH+ increased by 2%, 8%, and 4%, respectively. Similar findings were observed when individuals with alcohol or other drug use disorders were removed from the analyses. Finally, greater Component 2 scores were also associated with increased odds of having an alcohol or other drug use disorder. Collectively, these findings provide a more comprehensive understanding of the FH+ phenotype in young adults and help form a basis for further studies on biological mechanisms underlying risk for substance use disorders. The present findings also provide further support for a prominent role of ELA in promoting risk for problem alcohol and other drug use.
Keywords: Family history, Early life adversity, Externalizing traits, Internalizing traits, Cognition, Delay discounting, Impulsivity, Risk
1. Introduction
Abuse of alcohol and other drugs results in substantial public harm, with alcohol misuse alone accounting for approximately 25% of deaths in young adults (WHO, 2014). The best-known risk factor for alcohol and other drug use disorders is having a positive family history (FH+) of substance use disorders relative to persons with no such histories (FH−; Cotton, 1979, Cloninger, Bohman, & Sigvardsson, 1981a). FH+ persons display a complex phenotypic pattern variously termed “behavioral undercontrol” or “neurobehavioral disinhibition,” characterized by biases in decision-making and cognition, variability in behavior and unstable mood regulation (Sher, Grekin, & Williams, 2004; Sher & Trull, 1994; Tarter et al., 2003). As much of this work has been done in youths, we developed the Family Health Patterns (FHP) project to extensively characterize and contrast nonabusing FH+ and FH− young adults. Our work allows us to study the FH+ phenotype in individuals with mature cognitive, temperament, and personality development while still minimizing confounding influences of excessive substance use.
Through the FHP project, we have shown that FH+ young adults have alterations in temperament and personality (Lovallo, Yechiam, Sorocco, Vincent, & Collins, 2006; Saunders et al., 2008), altered decision-making and cognitive functioning (Acheson, Vincent, Sorocco, & Lovallo, 2011; Lovallo et al., 2006), and increased exposure to early life adversity (ELA) (Lovallo et al., 2013; Sorocco, Carnes, Cohoon, Vincent, & Lovallo, 2015). One of our most robust findings is that FH+ young adults in the FHP cohort have increased antisocial tendencies as indexed by low scores on the Socialization scale of Gough’s California Psychological Inventory (CPI-So) (Gough, 1994a; Vincent, Sorocco, Carnes, Cohoon, & Lovallo, 2017). We were also the first to demonstrate that FH+ persons have clear increases in discounting of delayed rewards (Acheson et al., 2011), similar to findings in individuals with substance use disorders (see MacKillop, 2013, Gray & MacKillop, 2015 for reviews). Finally, another notable finding is the increased ELA exposure in FH+ is directly linked to differences observed on many measures including their increased anti-social tendencies and increased discounting of delayed rewards (Lovallo et al., 2013; Sorocco et al., 2015), consistent with findings demonstrating both a genetic contribution to the elevated risk in FH+ (Cloninger, Bohman, & Sigvardsson, 1981b; Merikangas, 1990; Reich et al., 1998; Slutske et al., 2002) and a strong role for early childhood trauma and adversity (Kendler et al., 2012; Svingen et al., 2016).
As part of screening for the FHP project, we have accumulated a large dataset from a cohort of 506 FH+ and 528 FH− young adults with and without alcohol and other drug use disorders on a battery of measures including demographics, estimated intelligence, ELA, measures of temperament and personality, and delay discounting. While our earlier reports have identified specific variables affected by FH status in non-abusing young adults, this dataset allows us to much more comprehensively compare these affected variables and extend our findings to individuals with substance use disorders. Here we first use this dataset to determine which variables are most robustly affected by FH status in young adults and how these variables relate to each other. Finally, we examined how these variables predicted both FH status and number of parents and grandparents with alcohol and other drug use disorders (a key risk index) as well as the presence of alcohol and other drug use disorders in the individuals themselves.
2. Method
2.1. Participants
We examined data from 1031 healthy young adults recruited from the local community who were 18–30 years of age and were screened for potential inclusion in the FHP study and had complete data for relevant study variables (77% of the full sample of screened participants). All participants signed consent forms approved by the Institutional Review Boards at the University of Oklahoma Health Sciences Center and the Veterans Affairs Medical Center in Oklahoma City, OK and at the University of Texas Health Sciences Center, San Antonio, TX and were paid for their participation. Privacy was further protected by a Certificate of Confidentiality from the U.S. Department of Health and Human Services.
2.2. Screening, inclusion and exclusion criteria
Subjects were recruited using advertisement in local newspapers, flyers posted in locations frequented by persons of the desired age range including college campuses, direct contact via campus job fairs and student activities, and electronic media including Craig’s List and campus list servers directed to students and staff. This multipronged approach to subject recruitment is preferable to a single source of volunteers, such as students or campus employees, and is superior to random telephone dialing in terms of attracting the needed numbers of volunteers (Sorocco, Vincent, Collins, Johnson, & Lovallo, 2006). Subjects were screened by telephone to ensure general conformity with entrance criteria followed by a laboratory visit for further evaluation. Physical health was assessed through a medical history checklist and self-report of current good health. Psychiatric history was assessed using the computerized version of the Diagnostic Interview Schedule updated for DSM-IV diagnoses (C-DIS-IV) (Blouin, Perez, & Blouin, 1988) administered by a trained interviewer working under the direction of a licensed clinical psychologist.
2.2.1. Inclusion criteria
To qualify, participants were required to have current good health and no use of CNS-acting medications, and no history of neurological impairment or diabetes mellitus. They were also required to have normal intelligence based on Shipley Institute of Living verbal scale score ≥ 20 (John & Rattan, 1992), have been raised by at least one biological parent and be in contact with them, and be between 18 and 30 years old.
2.2.2. Exclusions
Participants were not allowed to participate if they had suspected maternal alcoholism during subject’s gestation, or were unable to provide credible report of family alcohol use patterns for two generations.
2.2.3. Family history of alcohol and other drug use disorders
FH classification was established using Family History Research Diagnostic Criteria (FH-RDC), which have a high degree of inter-rater reliability for reports of substance use disorders (Andreasen, Endicott, Spitzer, & Winokur, 1977). Inclusion criteria required that each pro-band be raised by at least one biological parent, be in touch with that parent, and adoptees were excluded from consideration. Persons were considered FH+ if either biological parent met FH-RDC criteria for alcohol or other drug use disorder. FH− were those reporting an absence of SUD in their biological parents and grandparents. The reliability of proband FH-RDC reports was verified by parent interview in 52% of the cases participating in the full study protocol, and these yielded 90% agreement between the two sources. In cases of disagreement between parent and proband reports, the subject’s data were excluded from analysis if parent reports did not allow a clear FH group assignment (8.8% of cases), and in the remaining cases the parent report was given precedence and the subject’s assignment was changed accordingly. FH− were coded 0 and FH+ were coded 1. A family history density score (FH density) was calculated by counting the number of biological parents and grandparents meeting criteria for any substance use disorder (including abuse, dependence, and/or withdrawal). Scores ranged from 0 (FH− participants) to a possible 6 (FH+ participants with both parents and all grandparents affected).
2.3. Analytic variables
Variables selected for inclusion in this analysis were determined from preliminary analyses examining differences between FH groups in the full screening sample (Table 2). Only those variables shown to differ between the FH groups (using independent t-tests) with a Cohen’s d effect size ≥0.2 were included in the current analyses. These variables are detailed below and consisted of demographic measures, estimated intelligence, ELA, temperament and personality measures, and a delay discounting questionnaire. Analyses for the current paper included only those individuals who had complete data across all of these variables (N = 1031).
Table 2.
Comparison of analytic variables.
| Measure | FH − | FH + | t | P | Holms-Bonferroni p* | Cohen’s d | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| N | Mean | SE | N | Mean | SE | |||||
| Education, yrs | 679 | 15.50 | 0.08 | 707 | 14.69 | 0.08 | 6.81 | 0.0000 | 0.0000 | 0.366 |
| Childhood SES | 631 | 48.87 | 0.49 | 645 | 42.05 | 0.54 | 9.30 | 0.0000 | 0.0000 | 0.523 |
| ELA | 588 | 0.51 | 0.03 | 586 | 1.17 | 0.03 | −15.34 | 0.0000 | 0.0000 | 0.906 |
| BDI | 543 | 5.13 | 0.24 | 629 | 9.12 | 0.34 | −9.59 | 0.0000 | 0.0000 | 0.579 |
| CPI-So | 677 | 31.59 | 0.20 | 703 | 27.35 | 0.23 | 14.10 | 0.0000 | 0.0000 | −0.766 |
| Shipley Mental Age | 616 | 17.47 | 0.07 | 677 | 17.06 | 0.06 | 4.41 | 0.0000 | 0.0004 | 0.246 |
| PPI Factor 1 | 665 | 16.82 | 0.13 | 685 | 16.54 | 0.12 | 1.57 | 0.1159 | 1.0000 | 0.086 |
| PPI Factor 2 | 665 | 12.28 | 0.09 | 685 | 13.21 | 0.10 | −6.88 | 0.0000 | 0.0000 | 0.376 |
| EPI-extraversion | 646 | 12.82 | 0.14 | 671 | 12.32 | 0.16 | 2.33 | 0.0200 | 0.4072 | 0.129 |
| EPI-neuroticism | 646 | 6.10 | 0.16 | 671 | 8.36 | 0.18 | −9.29 | 0.0000 | 0.0000 | 0.514 |
| EPI-impulsivity | 646 | 4.05 | 0.07 | 671 | 4.33 | 0.08 | −2.59 | 0.0097 | 0.2227 | 0.143 |
| TPQ-novelty seeking | 667 | 15.49 | 0.19 | 684 | 16.44 | 0.21 | −3.34 | 0.0009 | 0.0226 | 0.183 |
| TPQ-harm avoidance | 667 | 9.90 | 0.24 | 684 | 12.54 | 0.27 | −7.31 | 0.0000 | 0.0000 | 0.400 |
| TPQ-reward dependence | 667 | 19.26 | 0.18 | 684 | 18.92 | 0.17 | 1.39 | 0.1636 | 1.0000 | 0.076 |
| Delay discounting (log k) | 611 | −4.52 | 0.06 | 679 | −4.11 | 0.05 | −5.40 | 0.0000 | 0.0000 | 0.300 |
Notes. Cohen’s d values are expressed as FH+ > FH−. SE = standard error. SES = socioeconomic status. ELA = early life adversity. BDI = Beck Depression Inventory. CPISo = socialization scale from the California Personality Inventory. Shipley Mental Age = estimated mental age from the Shipley Institute of Living scale. PPI = Psychopathic Personality Inventory. EPI = Eysenck Personality Inventory. TPQ = Tridimensional Personality Inventory.
Variables with Cohen’s d values of 0.2 or greater are indicated with bold text.
Holms-Bonferroni is a p-value adjustment that corrects for familywise error rate.
Demographic measures consisted of years of education completed and childhood socioeconomic status. Childhood socioeconomic status using the Hollingshead scale (Hollingshead, 1975) based on the primary occupation of the main breadwinner in the household in which the participant grew up.
Estimated intelligence was based on the mental age score from the Revised Shipley Institute of Living scale (Zachary, 1986).
Early life adversity (ELA) scores were derived from items on the posttraumatic stress disorders module on the C-DIS-IV, which has a high degree of test-retest and inter-instrument reliability (Foa & Tolin, 2000). The items are closely similar to the life events assessed retrospectively in the studies by Caspi (Caspi et al., 2002) as follows: Physical or Sexual Adversity (Have you ever been mugged or threatened with a weapon or ever experienced a break-in or robbery? Have you ever been raped or sexually assaulted by a relative? Have you ever been raped or sexually assaulted by someone not related to you?), and Emotional Adversity (Before you were 15, was there a time when you did not live with your biological mother for at least 6 months? Before you were 15, was there a time when you did not live with your biological father for at least 6 months?). ELA scores ranged from 0 (no adverse events) to 5.
Temperament and personality measures included were the Beck Depression Inventory II total score (BDI; Beck, Steer, Ball, & Ranieri, 1996), the Socialization scale of the California Personality Inventory (CPI-So; Gough, 1994b), the Psychopathic Personality Index (PPI; Lilienfeld & Andrews, 1996) Factor 1 (Fearless Dominance) and Factor 2 (Impulsive Antisociality) scales (Benning, Patrick, Blonigen, Hicks, & Iacono, 2005), Eysenck Personality Inventory (EPI; Eysenck & Eysenck, 1975), and the Tridimensional Personality Questionnaire (TPQ; Cloninger, Przybeck, & Åvrakic, 1991).
Delay discounting was assessed with a paper and pencil questionnaire (Acheson et al., 2011; Kirby, Petry, & Bickel, 1999). Participants completed 27 fixed-choice options between immediate, smaller and delayed, larger hypothetical amounts of money (e.g., “Would you prefer(a) $54 today or (b) $80 in 30 days?”). Delayed amounts of money ranged from small ($25–$35), medium ($50–$60), to large ($75–$85). Rates of delay discounting were characterized by calculating k values based on Mazur’s hyperbolic discounting function (Mazur, 1987) for choices in each of the three delayed monetary categories. For this study, k values were averaged across the three monetary categories and log transformed. Higher average k values indicated increased delay discounting.
2.4. Statistical analysis
All data analyses for this paper were generated using SAS software, Version 9.4 of the SAS System for Windows (SAS Institute Inc., Cary, NC). As noted above, variables were identified by conducting univariate FH+ and FH− comparisons using the independent samples t-test. Effect sizes were calculated using Cohen’s d (see Table 2). Variables with effect sizes of 0.2 or greater were included in subsequent analyses.
Data reduction was then conducted using principal components analysis (PCA). The principal axis method was used to extract the components followed by a varimax rotation. The goal was to reduce e set of variables into a smaller number of principal components for use in additional analyses. The number of retained components was determined using the following criteria: 1) eigenvalue > 1.0, 2) accounts for 10% or more of the total variance, and 3) substantive interpretability. To interpret the components, we focused on variables with factor loadings > 0.40 (Stevens, 2012). Simple component (or factor) scores from this principal components solution were created using a unit-weighting procedure that summed the variables with loadings >0.40. Finally, a series of logistic regressions were conducted to predict FH status and the presence of alcohol and other drug use disorders from the component scores.
3. Results
Demographic characteristics and substance use disorder classifications are shown in Table 1. The FH+ group has a disproportionate number of females, representing a greater rate of study participation among women, as well as a higher proportion of minorities relative to European whites. As expected the FH+ group represented more persons with an alcohol use disorder, a non-alcohol substance use disorder, or any substance use disorder. Table 2 shows the candidate predictor variables for FH status along with univariate contrasts and Cohen’s d effect sizes.
Table 1.
Study sample demographics (N = 1031).
| Variable | Total | FH − | FH + | p* |
|---|---|---|---|---|
| N | 1031 | 524 | 507 | |
| Male [N (%)] | 414 (40) | 247 (47) | 167 (33) | < 0.0001 |
| Non-Hispanic [N (%)] | 942 (91) | 478 (91.2) | 464 (91.5) | 0.87 |
| Race [N (%)] | 0.0007 | |||
| White | 806 (78) | 422 (80.5) | 384 (75.7) | |
| Black | 104 (10) | 42 (8.0) | 62 (12.3) | |
| Native American | 34 (3) | 8 (1.5) | 26 (5.1) | |
| Biracial | 32 (3) | 18 (3.4) | 14 (2.8) | |
| Asian | 10 (1) | 8 (1.5) | 2 (0.4) | |
| Hawaiian/Pacific Islander | 4 (< 1) | 4 (0.8) | 0 (0) | |
| Other | 41 (4) | 22 (4.2) | 19 (3.8) | |
| Age | 23.7 (3.3) | 23.5 (3.3) | 23.8 (3.4) | 0.10 |
| Alcohol use disorder (n = 994); [N (%)] | 160 (16) | 64 (12.2) | 96 (18.9) | 0.003 |
| Alcohol abuse [N (%)] | 84 (8.5) | 33 (6.3) | 51 (10.1) | 0.026 |
| Alcohol dependence [N (%)] | 73 (7.0) | 30 (5.7) | 43 (8.5) | 0.08 |
| Other substance use disorder (n = 994); [N (%)] | 39 (3.9) | 8 (1.5) | 31 (6.1) | 0.0001 |
| Drug abuse [N (%)] | 12 (1.2) | 5 (1) | 7 (1.4) | 0.56 |
| Drug dependence [N (%)] | 26 (2.6) | 2 (0.4) | 24 (4.7) | < 0.0001 |
| Any substance use disorder (n = 994); [N (%)] | 275 (28) | 99 (18.9) | 176 (34.7) | < 0.0001 |
| Alcohol and/or drug abuse [N (%)] | 111 (11.2) | 43 (8.2) | 68 (13.4) | 0.007 |
| Alcohol and/or drug dependence [N (%)] | 160 (16.1) | 54 (10.3) | 106 (20.9) | < 0.0001 |
Note. FH− = no alcohol or other substance use disorders in biological parents or grandparents.
p-value from independent t-test or chi-square analysis, as appropriate.
3.1. Principal components analysis
Measures from Table 2 with Cohen’s d ≥ 0.2 were included in the PCA analysis (Table 3). Using exploratory PCA, the top three components displayed eigenvalues λ > 1.0 and each accounted for > 10% of the total variance. Therefore, these 3 components were retained for rotation. The full 3-component solution accounted for 58% of the total variance of the observed variables with the component structure shown in Table 3. The eigenvalue (λ) of the first principal component (λ1 = 3.16) was almost 2 times greater than the next largest component (λ2 = 1.60). Component 1 accounted for 31.5% of the total variance, consistent with high correlations across these specific instruments both in the present dataset (Table 4) and previous reports (Meites, Lovallo, & Pishkin, 1980). Higher Component 1 scores indicated greater internalizing tendencies. Component 2 accounted for16.0% of the total variance, with higher scores indicating poorer impulse control, more antisocial behavior, and greater ELA exposure. Finally, Component 3 accounted for 10.8% of total variance and was more conceptually mixed and individual items were more modestly correlated (Table 4), with higher scores indicating greater discounting of delayed rewards, lower estimated intelligence, fewer years of education, and lower SES.
Table 3.
Varimax-rotated factor loadings (N = 1031).
| Component | |||
|---|---|---|---|
| Variable | 1 | 2 | 3 |
| TPQ harm avoidance | 0.87 | −0.06 | −0.08 |
| EPI neuroticism | 0.84 | 0.28 | 0.00 |
| Beck Depression Inventory | 0.70 | 0.40 | −0.12 |
| PPI Factor II (impulsive antisociality) | 0.27 | 0.80 | 0.14 |
| CPI-So | −0.21 | −0.79 | 0.23 |
| Early life adversity | 0.06 | 0.51 | −0.37 |
| Shipley Mental Age | −0.11 | −0.05 | 0.72 |
| Childhood SES | −0.08 | 0.09 | 0.69 |
| Years of education | −0.02 | −0.29 | 0.61 |
| Delay discounting (log k) | −0.09 | 0.32 | −0.42 |
Variables with Cohen’s d values of 0.2 or greater are indicated with bold text.
Table 4.
Pearson correlations between primary component variables.
| Variable | TPQ- harm avoidance | EPI-neuroticism | BDI | CPI-So | PPI Factor 2 | ELA | Education, yrs | Childhood SES | Shipley Mental Age |
|---|---|---|---|---|---|---|---|---|---|
| TPQ- harm avoidance | 1.0 | – | – | – | – | – | – | – | – |
| EPI-neuroticism | 0.60** | – | – | – | – | – | – | – | – |
| BDI | 0.45** | 0.60** | – | – | – | – | – | – | – |
| CPI-So | 0.19** | −0.34** | −0.46** | – | – | – | – | – | – |
| PPI Factor 2 | 0.17** | 0.44** | 0.43** | −0.54** | – | – | – | – | – |
| ELA | 0.14** | 0.18** | 0.22** | −0.43** | 0.22** | – | – | – | – |
| Education, yrs | −0.07* | −0.12* | −0.17** | 0.32** | −0.15** | −0.25** | – | – | – |
| Childhood SES | −0.07 | −0.05 | −0.10* | 0.15** | 0.02 | −0.22** | 0.22** | – | – |
| Shipley Mental Age | −0.10* | −0.10* | −0.19** | 0.23** | −0.04 | −0.19** | 0.35** | 0.23** | – |
| Delay discounting (log k) | 0.01 | 0.07 | 0.15** | −0.21** | 0.14** | 0.14** | −0.18** | −0.16** | −0.24** |
TPQ = Tridimensional Personality Inventory. BDI = Beck Depression Inventory. CPI-So = socialization scale from the California Personality Inventory. PPI = Psychopathic Personality Inventory. ELA = early life adversity. SES = socioeconomic status. Shipley Mental Age = estimated mental age from the Shipley Institute of Living scale.
Significant at p < 0.01.
Significant at p < 0.0001.
3.2. Prediction of FH status
We first predicted FH status using a 3-predictor logistic regression incorporating the component scores listed above. As shown in Table 5, the 3 individual predictors successfully differentiated the FH groups, as did the full model, indicated by a significant overall likelihood ratio, χ2(3) = 203.12, p < 0.0001, R2 = 18.0%, Nagelkerke’s R2 = 24.0%. According to the model, the odds of an individual being classified FH+ was predicted by higher scores on each component (Table 5; p’s < 0.001). For each 1-point increase in the Component 1, 2, and 3 scores, the odds of being classified FH+ increased by 2%, 8%, and 4%, respectively. The individual components were all significant univariate predictors of FH status (ps < 0.0001). The components share common underlying mechanisms, and they had small to moderate intercorrelations (rs = 0.13–0.44). Due to multicollinearity, variance inflation factors were calculated and were all < 1.3 and accordingly were not considered a significant influence on the logistic regression results.
Table 5.
Logistic regression analysis predicting FH using component scores.
| Predictor | β | SE β | Wald’s χ2 | df | p | OR (95% CI) |
|---|---|---|---|---|---|---|
| Constant | 3.87 | 0.49 | 62.49 | 1 | < 0.0001 | NA |
| Component 1 | 0.02 | 0.005 | 12.83 | 1 | 0.0003 | 1.02 (1.01–1.03) |
| Component 2 | 0.08 | 0.01 | 53.68 | 1 | < 0.0001 | 1.08 (1.06–1.10) |
| Component 3 | 0.04 | 0.005 | 52.47 | 1 | < 0.0001 | 1.04 (1.03–1.05) |
| Test | χ2 | df | p |
|---|---|---|---|
| Overall model evaluation | |||
| Likelihood ratio test | 203.12 | 3 | < 0.0001 |
| Score test | 185.03 | 3 | < 0.0001 |
| Wald test | 155.28 | 3 | < 0.0001 |
| Goodness-of-fit test | |||
| Hosmer & Lemeshow | 7.75 | 8 | 0.46 |
Note. Cox and Snell R2 = 0.18. Nagelkerke R2 (Max rescaled R2) = 0.24. Kendall’s Tau-a = 0.25. Goodman-Kruskal Gamma = 0.49. Somer’s Dxy = 0.49. c-statistic = 74.4%. OR = odds ratio; CI = confidence interval; NA = not applicable.
3.3. Associations of component scores with FH density
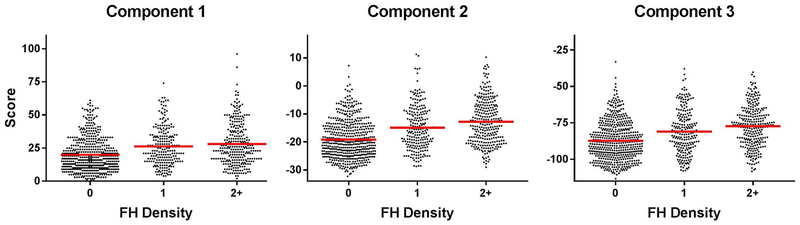
A series of one-way ANOVAs was conducted to examine the effect of FH density (0, 1, or 2+) on each of the component scores. A significant effect of FH density was observed for all three components (p < 0.0001). Those with 0 family members with substance use disorders were significantly different from those with a FH density of 1 or more for all three of the component scores (Fig. 1). In addition, Component 2 and 3 scores were lower in individuals with a FH density of 1 versus 2+ (Component 2: p = 0.003, Component 3: p = 0.01), but this difference was not observed for Component 1 scores (p = 0.31).
Fig. 1.
Offset scatter plots showing relationships between FH density (number of biological parents and grandparents with alcohol and other substance use disorders) and the three component scores with means for each FH density grouping shown in red. For a listing of measures comprising each component, please see Table 3. A significant effect of FH density was observed for all three components (p < 0.0001). Those with 0 family members with substance use disorders were significantly different from those with a FH density of 1 or more for all three of the component scores (Fig. 1). In addition, Component 2 and 3 scores were lower in individuals with a FH density of 1 versus 2+ (Component 2: p = 0.003, Component 3: p = 0.01), but this difference was not observed for Component 1 scores (p = 0.31).
3.4. Prediction of substance use disorders
We next used these same components in a logistic regression to predict alcohol use disorder, non-alcohol substance use disorder, or any substance use disorder status, as shown in Table 6. The estimated odds of having any of the three substance use disorder statuses were increased among those with higher Component 2 scores (χ2 = 28.8–92.7, p’s < 0.0001). Higher Component 1 scores decreased the odds of having a non-alcohol substance use disorder alone (χ2 = 4.14, p < 0.05), but did not alter the odds of having an alcohol use disorder or any substance use disorder. When each component score was examined individually, higher Component 2 scores increased the odds of having an alcohol use disorder, a non-alcohol substance use disorder, or any substance use disorders (p’s < 0.0001), and higher Component 1 scores increased the odds of alcohol use disorder or any substance use disorder (p’s < 0.001). Higher Component 3 scores increased the odds of non-alcohol substance use disorders (p = 0.03) as well as any substance use disorder (p = 0.047). As previously noted, the correlations among the components ranged from r = 0.13–0.44, indicating a degree of multicollinearity. Variance inflation factors relating to substance use disorder status were all < 1.3. However, shared variance may explain why the logistic regression examining all 3 components together showed only higher Component 2 scores increased odds of having any of the substance use disorder statuses, while higher Component 3 scores were no longer significant and higher Component 1 scores marginally decreased odds of having non-alcohol substance use disorders.
Table 6.
Component scores as predictors of alcohol and other substance-use disorders.
| Outcome – predictor | β | SE β | Wald’s χ2 | P | OR |
|---|---|---|---|---|---|
| Alcohol use disorder (N = 878) | R2 = 0.10 | ||||
| Constant | −0.86 | 0.61 | 1.98 | 0.16 | |
| Component 1 | 0.002 | 0.006 | 0.07 | 0.79 | 1.00 |
| Component 2 | 0.09 | 0.01 | 43.58 | < 0.0001 | 1.09 |
| Component 3 | −0.009 | 0.01 | 1.84 | 0.17 | 0.99 |
| Other substance use disorder (N = 758) | R2 = 0.13 | ||||
| Constant | 0.54 | 0.99 | 0.30 | 0.58 | |
| Component 1 | −0.03 | 0.01 | 4.14 | 0.04 | 0.97 |
| Component 2 | 0.12 | 0.02 | 28.83 | < 0.0001 | 1.13 |
| Component 3 | 0.01 | 0.01 | 1.04 | 0.31 | 1.01 |
| Any substance use disorder (N = 994) | R2 = 0.18 | ||||
| Constant | 0.74 | 0.50 | 2.16 | 0.14 | |
| Component 1 | −0.004 | 0.006 | 0.61 | 0.44 | 1.00 |
| Component 2 | 0.12 | 0.01 | 92.73 | < 0.0001 | 1.12 |
| Component 3 | −0.002 | 0.005 | 0.15 | 0.70 | 1.00 |
3.5. Tests of confounders: alcohol and other drug use disorders, race, and sex
In order to determine the impact of persons with alcohol and other drug use disorders in the initial FH-status comparison, we repeated the principal components analysis after excluding persons with any substance use disorder diagnosis. No difference in the internal structure of the components was found in this subset (N = 756), and each component accounted for a similar portion of the total variance as in the analysis with the full sample (Component 1 = 30.3%, Component 2 = 16.4%, and Component 3 = 10.5%). As before, the three-predictor logistic regression model resulted in a significant overall likelihood ratio, χ2(3) = 139.16, p < 0.0001, R2 = 16.8%, Nagelkerke’s R2 = 22.5%, indicating that the scores on these 3 components differentiated the FH+ and FH− groups in persons lacking any substance use disorders. In addition, the log of the odds of an individual being FH + was predicted by each of the individual component scores (p’s < 0.001). For each 1-point increase in the Component 1, Component 2, and Component 3 scores, the odds of being classified FH + increased by 2%, 7%, and 4%, respectively. Additional tests controlling for race and sex indicated that these had no significant impact on the above component structures or prediction of FH and substance use disorder status.
4. Discussion
In this study, we sought to comprehensively characterize young adults with family histories of alcohol and other drug use disorders on a broad array of measures. First, we identified demographic, temperament, cognitive, and life-history measures that best distinguished FH+ and FH− young adults. Next, we used these measures in a principal components analysis to identify sets of variables with shared variance, and three principal components emerged. Component 1 consisted of scores on TPQ Harm Avoidance, EPI Neuroticism, and Beck Depression Inventory (BDI), with higher component scores indicating greater internalizing tendencies. Component 2 consisted of PPI Factor II and CPI Socialization scores as well as ELA, with higher component scores indicating greater externalizing tendencies and more ELA exposure. Component 3 consisted of consisted of Shipley Mental Age, childhood socioeconomic status, years of education, and delay discounting, with higher component scores indicating lower estimated intelligence, more discounting of delayed rewards, and worse socioeconomic circumstances. Logistic regression then showed that the full three-component model, and each of the individual components, were significant predictors of FH status, and each component was associated with FH density. Similar findings were observed when individuals with alcohol or other drug use disorders were removed from the analyses. Next, we examined if the 3 components would identify the presence of an alcohol use disorder, a non-alcohol substance use disorder, or any substance use disorder in this population. Here, Component 2 scores predicted all three substance use disorder categories. Collectively, these findings help to more comprehensively understand the FH+ phenotype in young adults.
Component 2 scores were most robustly linked to FH+ status, FH+ density, and having an alcohol or other drug use disorder. This component included measures of externalizing traits (impulsive and antisocial behaviors), consistent with previous studies identifying strong relationships between these traits and problem alcohol and other drug use (e.g.Barnow, Schuckit, Lucht, John, & Freyberger, 2002; El-Sheikh, 2001; Hasin et al., 2011; Stabenau, 1990). In addition to externalizing traits, Component 2 scores also included ELA, with notably the largest effect size difference between FH+ and FH− individuals (d = 0.906) of the measures we examined. Previous studies have found youths and adults exposed to ELA are more likely to display antisocial and externalizing behaviors (Abry et al., 2017; Biederman, Faraone, & Monuteaux, 2002; Carver, Johnson, McCullough, Forster, & Joormann, 2014; Eaves, Prom, & Silberg, 2010; Vaughn et al., 2011), and youths with alcohol problems are more likely to display aggressive and externalizing tendencies and to have experienced ELA (Barnow, Schuckit, Smith, Preuss, & Danko, 2002; Svingen et al., 2016). Further, ELA is more common among FH+ youths (Charles et al., 2014), ELA is a contributor to risk for alcohol and other drug problems among FH+ individuals (Kendler et al., 2012; Svingen et al., 2016), and the impact of ELA may be increased in relation to genetic vulnerabilities (Hicks, South, Dirago, Iacono, & McGue, 2009; Schilling, Aseltine, & Gore, 2008). Thus, ELA exposure may function to a greater degree in vulnerable individuals, such as FH+ persons, and that this level of exposure may itself contribute to development of externalizing behaviors promoting a heightened risk for alcohol and other drug use disorders. However, as greater levels of externalizing traits, problem substance use, and other psychiatric issues in parents can cause increased ELA experiences for their children, it is possible that the ELA relationships observed here may at least partially be a corollary of a more direct inheritance of these traits from parents rather than simply an environmental influence.
In contrast to Component 2 scores, Component 3 scores were more modestly linked with FH+ status and FH density and were not a significant predictor of substance use disorders in the logistic regression analyses. Component 3 measures were more diverse than the measures in Components 1 and 2, consisting of delay discounting, estimated intelligence, childhood socioeconomic status and years of education. While increased delay discounting has been previously conceptualized as an endophenotype for substance use disorders (Gray & MacKillop, 2015; MacKillop, 2013), our findings suggest increased delay discounting may be tied to a broader cognitive endophenotype that includes decreased estimated intelligence, which appears to be present already in pre-adolescent FH+ children (Dougherty et al., 2014, 2015). The association of poorer childhood socioeconomic status and increased delay discounting, lower estimated intelligence, and less education as an adult suggests that early experiences may have strong influences on cognitive processes underlying delay discounting and estimated intelligence as an adult, although it is not possible to rule out more direct genetic transmission of cognitive impairments (e.g., decision making and cognitive impairments in parents may reflect an inherited genetic diathesis in the offspring). Similarly, decision-making and cognitive impairments in FH+ individuals may limit their education achievements. The lack of significant independent associations with substance use disorders suggests this potential cognitive endophenotype is not sufficient to trigger problem substance use, however the strong association with FH+ status suggests that individuals displaying this phenotype who also have high levels of externalizing traits and increased ELA may be particularly vulnerable to developing problem substance use.
Component 1 comprised of measures indexing greater internalizing tendencies and while Component 1 scores were also associated with FH + status and density, these associations were weaker than for the other two component scores. These findings are consistent with previous studies identifying modest relationships between internalizing symptoms and family histories of alcohol and other drug use disorders that were often done in children (e.g., Iacono, Malone, & McGue, 2008; Preuss, Schuckit, Smith, Barnow, & Danko, 2002). While increased Component 1 scores were associated with increased odds of having an alcohol or other drug use disorders when considered independently, increased scores on this component were associated with a small decrease in odds of having a non-alcohol substance use disorder when considered along with the other components. This latter result is consistent with findings of modest protective effects of high internalizing symptoms on risk problem substance use risk after controlling for externalizing symptoms in adolescents (Colder et al., 2017). Plausibly, internalizing traits could offer some protection against substance use disorders by limiting social interactions that promote problem substance use and perhaps greater aversion to initiating alcohol and other substance use during adolescence and early adulthood due to increased harm avoidance behavior (TPQ; Cloninger et al., 1991).
This study has both strengths and limitations. This study had a large sample cohort that completed extensive measures of demographics, estimated intelligence, early life adversity (ELA), temperament and personality, substance use, and family history. Our sample included an increased representation of FH+ individuals, facilitating the analyses we performed but also potentially limiting how well these findings may generalize to the general population. Further, while this sample included a large number of individuals with substance use disorders, follow up analyses indicated these individuals were not driving our FH findings. We limited our FH density analysis to affected parents and grandparents as this is more directly related to substance use disorder risk in other relatives, however our findings may have changed had we examined density of substance use disorders in other relatives as well. There was an imbalance of women and minorities between the FH groups, although these imbalances did not appear to influence our results. Given the cross-sectional design, we are not able to determine causal relationships between study variables and substance use outcomes. Further, ELA was retrospectively reported and thus may be subject to recall biases, although the magnitude of events we are assessing makes this unlikely. Additionally, it is unclear to what degree ELA may be an environmental factor directly influencing outcomes versus being a corollary of parental psychopathology that may be passed on to offspring. Finally while our sample provides a comprehensive description of FH+ young adults, it is unclear how many of these characteristics may persist as these individuals grow to middle age and fully pass out of their peak risk period for developing substance use disorders.
In summary, we observed three primary components of traits and characteristics that distinguished FH+ and FH− young adults. By developing a more comprehensive understanding of FH+ phenotype in young adults, we establish a clearer framework for studying biological substrates that underlie this phenotype that could ultimately be targeted for prevention and intervention strategies. Of the 3 components we identified, one consisting of externalizing traits and early life adversity was most robustly associated with FH+ status, FH density, and the presence of substance use disorders. While it is not possible to determine causal relationships with our current data, these findings provide further support for a role of ELA in promoting risk for problem alcohol and other drug use. Thus, interventions to mitigate the impact of ELA may be a promising strategy to help attenuate the expression of the FH+ phenotype and prevent the development of substance use disorders.
HIGHLIGHTS.
Measures affected by family history of substance use disorders were compared.
From these measures three principal components were identified.
An externalizing behaviors and adversity component best predicted family history.
This same component also best predicted substance use disorders.
Acknowledgements
Supported by the Department of Veterans Affairs Medical Research Service, the National Institutes of Health/NIAAA (R01-AA12207), and the Arkansas Biosciences Institute. The authors have no conflicts of interest to declare.
References
- Abry T, Bryce CI, Swanson J, Bradley RH, Fabes RA, & Corwyn RF (2017).Classroom-level adversity: Associations with Children’s internalizing and externalizing behaviors across elementary school. Developmental Psychology, 53(3), 497–510. [DOI] [PubMed] [Google Scholar]
- Acheson A, Vincent AS, Sorocco KH, & Lovallo WR (2011). Greater discounting of delayed rewards in young adults with family histories of alcohol and drug use disorders: Studies from the Oklahoma family health patterns project. Alcoholism: Clinical and Experimental Research, 35, 1607–1613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andreasen NC, Endicott J, Spitzer RL, & Winokur G (1977). The family history method using diagnostic criteria. Reliability and validity. Archives of General Psychiatry, 34, 1229–1235. [DOI] [PubMed] [Google Scholar]
- Barnow S, Schuckit M, Smith TL, Preuss U, & Danko G (2002). The relationship between the family density of alcoholism and externalizing symptoms among 146 children. Alcohol and Alcoholism, 37, 383–387. [DOI] [PubMed] [Google Scholar]
- Barnow S, Schuckit MA, Lucht M, John U, & Freyberger HJ (2002). The importance of a positive family history of alcoholism, parental rejection and emotional warmth, behavioral problems and peer substance use for alcohol problems in teenagers: A path analysis. Journal of Studies on Alcohol, 63, 305–315. [DOI] [PubMed] [Google Scholar]
- Beck AT, Steer RA, Ball R, & Ranieri W (1996). Comparison of Beck Depression Inventories -IA and -II in psychiatric outpatiens. Journal of Personality Assessment, 67, 588–597. [DOI] [PubMed] [Google Scholar]
- Benning SD, Patrick CJ, Blonigen DM, Hicks BM, & Iacono WG (2005). Estimating facets of psychopathy from normal personality traits: A step toward community epidemiological investigations. Assessment, 12, 3–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Biederman J, Faraone SV, & Monuteaux MC (2002). Differential effect of environmental adversity by gender: Rutter’s index of adversity in a group of boys and girls with and without ADHD. The American Journal of Psychiatry, 159, 1556–1562. [DOI] [PubMed] [Google Scholar]
- Blouin AG, Perez EL, & Blouin JH (1988). Computerized administration of the diagnostic interview schedule. Psychiatry Research, 23, 335–344. [DOI] [PubMed] [Google Scholar]
- Carver CS, Johnson SL, McCullough ME, Forster DE, & Joormann J (2014). Adulthood personality correlates of childhood adversity. Frontiers in Psychology, 5, 1357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caspi A, McClay J, Moffitt TE, Mill J, Martin J, Craig IW, … Poulton R (2002).Role of genotype in the cycle of violence in maltreated children. Science, 297, 851–854. [DOI] [PubMed] [Google Scholar]
- Charles NE, Ryan SR, Acheson A, Mathias CW, Liang Y, & Dougherty DM (2014). Childhood stress exposure among preadolescents with and without family histories of substance use disorders. Psychology of Addictive Behaviors, 29(1), 192–200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cloninger CR, Bohman M, & Sigvardsson S (1981a). Inheritance of alcohol abuse. Cross-fostering analysis of adopted men. Archives of General Psychiatry, 38, 861–868. [DOI] [PubMed] [Google Scholar]
- Cloninger CR, Bohman M, & Sigvardsson S (1981b). Inheritance of alcohol abuse: Cross fostering analysis of adopted men. Archives of General Psychiatry, 38, 861–868. [DOI] [PubMed] [Google Scholar]
- Cloninger CR, Przybeck TR, & Åvrakic DM (1991). The tridimensional personality questionnaire: U.S. normative data. Psychological Reports, 69, 1047–1057. [DOI] [PubMed] [Google Scholar]
- Colder CR, Frndak S, Lengua LJ, Read JP, Hawk LW Jr., & Wieczorek WF (2017). Internalizing and externalizing problem behavior: A test of a latent variable interaction predicting a two-part growth model of adolescent substance use. Journal of Abnormal Child Psychology (Epub ahead of print). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cotton NS (1979). The familial incidence of alcoholism: A review. Journal of Studies onAlcohol, 40, 89–116. [DOI] [PubMed] [Google Scholar]
- Dougherty DM, Charles NE, Mathias CW, Ryan SR, Olvera RL, Liang Y, & Acheson A (2014). Delay discounting differentiates pre-adolescents at high and low risk for substance use disorders based on family history. Drug and Alcohol Dependence, 143, 105–111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dougherty DM, Lake SL, Mathias CW, Ryan SR, Bray BC, Charles NE, & Acheson A (2015). Behavioral impulsivity and risk-taking trajectories across early adolescence in youths with and without family histories of alcohol and other drug use disorders. Alcoholism, Clinical and Experimental Research, 39, 1501–1509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eaves LJ, Prom EC, & Silberg JL (2010). The mediating effect of parental neglect on adolescent and young adult anti-sociality: A longitudinal study of twins and their parents. Behavior Genetics, 40, 425–437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- El-Sheikh M (2001). Parental drinking problems and children’s adjustment: Vagal regulation and emotional reactivity as pathways and moderators of risk. Journal of Abnormal Psychology, 110, 499–515. [DOI] [PubMed] [Google Scholar]
- Eysenck HJ, & Eysenck SBG (1975). Manual of the Eysenck personality questionnaire. London: Hodder and Stoughton. [Google Scholar]
- Foa EB, & Tolin DF (2000). Comparison of the PTSD symptom scale-interview version and the clinician-administered PTSD scale. Journal of Traumatic Stress, 13, 181–191. [DOI] [PubMed] [Google Scholar]
- Gough HG (1994a). Theory, development, and interpretation of the CPI socialization scale. Psychological Reports, 75, 651–700. [DOI] [PubMed] [Google Scholar]
- Gough HG (1994b). Theory, development, and interpretation of the CPI socialization scale. Psychological Reports, 75, 651–700. [DOI] [PubMed] [Google Scholar]
- Gray JC, & MacKillop J (2015). Impulsive delayed reward discounting as a genetically-influenced target for drug abuse prevention: A critical evaluation. Frontiers in Psychology, 6, 1104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hasin D, Fenton MC, Skodol A, Krueger R, Keyes K, Geier T, … Grant B (2011). Personality disorders and the 3-year course of alcohol, drug, and nicotine use disorders. Archives of General Psychiatry, 68, 1158–1167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hicks BM, South SC, Dirago AC, Iacono WG, & McGue M (2009).Environmental adversity and increasing genetic risk for externalizing disorders. Archives of General Psychiatry, 66, 640–648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hollingshead AB (1975). Four factor index of social status Series four factor index of social status (pp. 22). New Haven, CT: Yale University. [Google Scholar]
- Iacono WG, Malone SM, & McGue M (2008). Behavioral disinhibition and the development of early-onset addiction: Common and specific influences. Annual Review of Clinical Psychology, 4, 325–348. [DOI] [PubMed] [Google Scholar]
- John KR, & Rattan G (1992). Shipley institute of living scale—Revised In Keyser DJ, & Sweetland RC (Vol. Eds.), Test Critiques. Vol. IX. Test Critiques (pp. 490–495). Austin, TX: Pro-Ed, Inc. [Google Scholar]
- Kendler KS, Sundquist K, Ohlsson H, Palmer K, Maes H, Winkleby MA, & Sundquist J (2012). Genetic and familial environmental influences on the risk for drug abuse: A national Swedish adoption study. Archives of General Psychiatry, 69, 690–697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kirby KN, Petry NM, & Bickel WK (1999). Heroin addicts have higher discount rates for delayed rewards than non-drug-using controls. Journal of Experimental Psychology. General, 128, 78–87. [DOI] [PubMed] [Google Scholar]
- Lilienfeld SO, & Andrews BP (1996). Development and preliminary validation of a self-report measure of psychopathic personality traits in noncriminal populations. Journal of Personality Assessment, 66, 488–524. [DOI] [PubMed] [Google Scholar]
- Lovallo WR, Farag NH, Sorocco KH, Acheson A, Cohoon AJ, & Vincent AS (2013). Early life adversity contributes to impaired cognition and impulsive behavior: Studies from the Oklahoma family health patterns project. Alcoholism, Clinical and Experimental Research, 37, 616–623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lovallo WR, Yechiam E, Sorocco KH, Vincent AS, & Collins FL (2006). Working memory and decision-making biases in young adults with a family history of alcoholism: Studies from the Oklahoma family health patterns project. Alcoholism, Clinical and Experimental Research, 30, 763–773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacKillop J (2013). Integrating behavioral economics and behavioral genetics: Delayed reward discounting as an endophenotype for addictive disorders. Journal of the Experimental Analysis of Behavior, 99, 14–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mazur JE (1987). An adjusting amount procedure for studying delayed reinforcement In Commons Jem ML, Nevin JA, & Rachlin H (Vol. Eds.), Quantitative Analysis of Behavior: The Effects of Delay and of Intervening Events on Reinforcement Value: . Vol. 5. Quantitative analysis of behavior: The effects of delay and of intervening events on reinforcement value (pp. 55–73). Hillsdale: Lawrence Erlbaum Associates. [Google Scholar]
- Meites K, Lovallo W, & Pishkin V (1980). A comparison of four scales for anxiety, depression, and neuroticism. Journal of Clinical Psychology, 36, 427–432. [DOI] [PubMed] [Google Scholar]
- Merikangas KR (1990). The genetic epidemiology of alcoholism. Psychological Medicine, 20, 11–22. [DOI] [PubMed] [Google Scholar]
- Preuss UW, Schuckit MA, Smith TL, Barnow S, & Danko GP (2002). Mood and anxiety symptoms among 140 children from alcoholic and control families. Drug and Alcohol Dependence, 67, 235–242. [DOI] [PubMed] [Google Scholar]
- Reich T, Edenberg HJ, Goate A, Williams JT, Rice JP, Van Eerdewegh P, … Begleiter H (1998). Genome-wide search for genes affecting the risk for alcohol dependence. American Journal of Medical Genetics, 81, 207–215. [PubMed] [Google Scholar]
- Saunders B, Farag N, Vincent AS, Collins FL Jr., Sorocco KH, & Lovallo WR (2008). Impulsive errors on a go-NoGo reaction time task: Disinhibitory traits in relation to a family history of alcoholism. Alcoholism, Clinical and Experimental Research, 32, 888–894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schilling EA, Aseltine RH, & Gore S (2008). The impact of cumulative childhood adversity on young adult mental health: Measures, models, and interpretations. Social Science & Medicine, 66, 1140–1151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sher KJ, Grekin ER, & Williams NA (2004). The development of alcohol use disorders. Annual reviews of clinical. Psychology, 22, 1–22. [DOI] [PubMed] [Google Scholar]
- Sher KJ, & Trull TJ (1994). Personality and disinhibitory psychopathology:Alcoholism and antisocial personality disorder. Journal of Abnormal Psychology, 103, 92–102. [DOI] [PubMed] [Google Scholar]
- Slutske WS, Heath AC, Madden PA, Bucholz KK, Statham DJ, & Martin NG (2002). Personality and the genetic risk for alcohol dependence. Journal of Abnormal Psychology, 111, 124–133. [PubMed] [Google Scholar]
- Sorocco KH, Carnes NC, Cohoon AJ, Vincent AS, & Lovallo WR (2015). Risk factors for alcoholism in the Oklahoma family health patterns project: Impact of early life adversity and family history on affect regulation and personality. Drug and Alcohol Dependence, 150, 38–45. [DOI] [PubMed] [Google Scholar]
- Sorocco KH, Vincent AS, Collins FL, Johnson CA, & Lovallo WR (2006). Recruitment of healthy participants for studies on risks for alcoholism: Effectiveness of random digit dialling. Alcohol and Alcoholism, 41, 349–352. [DOI] [PubMed] [Google Scholar]
- Stabenau JR (1990). Additive independent factors that predict risk for alcoholism. Journal of Studies on Alcohol, 51, 164–174. [DOI] [PubMed] [Google Scholar]
- Stevens JP (2012). Applied multivariate statistics for the social sciences. New York:Routledge. [Google Scholar]
- Svingen L, Dykstra RE, Simpson JL, Jaffe AE, Bevins RA, Carlo G, … Grant KM (2016). Associations between family history of substance use, childhood trauma, and age of first drug use in persons with methamphetamine dependence. Journal of Addiction Medicine, 10, 269–273. [DOI] [PubMed] [Google Scholar]
- Tarter RE, Kirisci L, Mezzich A, Cornelius JR, Pajer K, Vanyukov M, … Clark D (2003). Neurobehavioral disinhibition in childhood predicts early age at onset of substance use disorder. American Journal of Psychiatry, 160, 1078–1085. [DOI] [PubMed] [Google Scholar]
- Vaughn MG, Fu Q, Beaver KM, Delisi M, Perron BE, & Howard MO (2011). Effects of childhood adversity on bullying and cruelty to animals in the United States: Findings from a national sample. Journal of Interpersonal Violence, 26, 3509–3525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vincent AS, Sorocco KH, Carnes B, Cohoon AJ, & Lovallo WR (2017). Antisocial characteristics and early life adversity predict substance use disorders in young adults: The Oklahoma family health patterns project. Journal of Substance Abuse and Alcoholism, 5. [Google Scholar]
- WHO (2014). Global status report on alcohol and health 2014. World Health Organization. [Google Scholar]
- Zachary (1986). Shipley institute of living scale revised manual. Los Angeles: Western Psychological Services. [Google Scholar]