Abstract
Basement membranes (BMs) are specialised extracellular matrix (ECM) structures and collagens are a key component required for BM function. While collagen IV is the major BM collagen, collagens VI, VII, XV, XVII and XVIII are also present. Mutations in these collagens cause rare multi-systemic diseases but these collagens have also been associated with major common diseases including stroke. Developing treatments for these conditions will require a collective effort to increase our fundamental understanding of the biology of these collagens and the mechanisms by which mutations therein cause disease. Novel insights into pathomolecular disease mechanisms and cellular responses to these mutations has been exploited to develop proof-of-concept treatment strategies in animal models. Combined, these studies have also highlighted the complexity of the disease mechanisms and the need to obtain a more complete understanding of these mechanisms. The identification of pathomolecular mechanisms of collagen mutations shared between different disorders represent an attractive prospect for treatments that may be effective across phenotypically distinct disorders.
Keywords: collagen, extracellular matrix, genetics, model organisms, molecular basis of health and disease, molecular mechanisms
Introduction
Basement membranes (BMs) are specialised extracellular matrix (ECM) structures that compartmentalise tissues, provide structural support and influence cell behaviour and signalling. BMs underlie epithelial and endothelial cells, surround smooth muscle, fat and Schwann cells, and occur in the synaptic cleft. They also link cells with the interstitial matrix which contains fibrillar collagens such as collagen I. BMs contain approximately 60–200 proteins and the composition of individual BMs differs to provide different biomechanical and biochemical properties to support their individual functions [1].
Vertebrates express 28 types of collagen, which are divided into classes based on protein domain structure and supramolecular assembly including fibrillar (e.g. collagen I), network forming (collagen IV), beaded microfibril (collagen VI), multiplexin (e.g. collagen XV and XVIII) and FACIT (fibril-associated collagens with interrupted triple helices, e.g. collagen VII and XVII) collagens. All collagens contain a triple helical collagen domain consisting of a Gly-X-Y repeat in which every third residue is a glycine and X-Y can be any amino acid. Collagens are folded within the endoplasmic reticulum (ER) and their secretion can require enlargement of COPII vesicles through TANGO1 (transport and Golgi organisation 1) and HSP47 (heat shock protein 47) proteins [2].
BM collagens are associated with a wide variety of diseases for which there is a need for treatments. Recent advances in elucidating disease mechanisms and gene/cell therapy-based approaches has identified therapeutic targets and guided proof-of-concept therapies. Here, we will provide a brief overview of recent progress in mechanisms of disease caused by mutations in BM collagens, and development of therapeutic strategies. For more in depth reviews on collagens and their diseases we refer the reader to the following: collagen IV [3–5], collagen VI [6], collagen VII and XVII [7,8], collagen XV and XVIII [9].
Collagen IV
Collagen IV is the most abundant structural BM component and is essential for BM integrity but not initial BM formation [10]. Vertebrates express six collagen IV α chains (α1(IV)–α6(IV)), encoded by the COL4A1–COL4A6 genes, forming three networks: α1α1α2(IV), α3α4α5(IV), and α5α5α6(IV). While α1α1α2(IV) is expressed in nearly every BM, α3α4α5(IV) and α5α5α6(IV) expression is more restricted [5]. Collagen IV alpha (α) chains contain a N-terminal 7S domain, a central collagen domain with approximately 20 interruptions, and a C-terminal NC1 domain, which initiates collagen folding in the ER. Following secretion, collagen IV molecules form a network in the BM that interacts with integrins, discoidin domain receptors (DDR) and G protein-coupled receptors [3,11].
Mutations affecting α3α4α5(IV) and α5α5α6(IV)
COL4A3–COL4A5 mutations cause glomerular BM (GBM) defects leading to Alport syndrome (AS) (OMIM # 301050, # 203780, # 104200), which causes renal disease, deafness and eye pathology [3]. AS now also covers thin BM nephropathy and familial benign haematuria phenotypes [12], while deletions spanning COL4A5 and COL4A6 cause AS with diffuse leiomyomatosis (OMIM # 308940). The production of auto-antibodies against the NC1 domain of α3(IV) underlies the autoimmune disorder Goodpasture syndrome [3]. Besides AS, COL4A3/COL4A4 mutations are associated with kidney disorders including diabetic kidney disease [13], focal segmental sclerosis [14] and steroid-resistant nephrotic syndrome [15], while COL4A5/COL4A6 mutations affect axogenesis in zebrafish [16], and COL4A6 mutations can cause non-syndromic hearing loss [17], indicating a growing role of these mutations in disease.
Autosomal dominant AS due to COL4A3/COL4A4 mutations is milder compared with autosomal recessive or X-linked AS (due to COL4A5 mutations), and nonsense mutations cause more severe disease compared with missense mutations, the majority of which affect the glycine residue in the Gly-X-Y repeat [3]. Reduced levels or absence of α3α4α5(IV) and associated GBM defects (Figure 1) is a major causative mechanism. Induced α5α5α6(IV) expression in Col4a3−/−mice (Table 1), a well-established model of AS, reduced disease severity [18] but mosaic α5α5α6(IV) expression in patients was not associated with improved outcome [19]. Similarly, X-linked AS patients exhibited persistent α1α1α2(IV) expression [3]. These data underscore the specific nature of the networks and potential species differences. Interestingly, a laminin β 2 missense variant which was not pathogenic of itself in mice, increased progression to kidney failure in Col4a3−/− mice and proteinuria in female Col4a5+/− mice, supporting the hypothesis that GBM components can act as genetic modifiers [20].
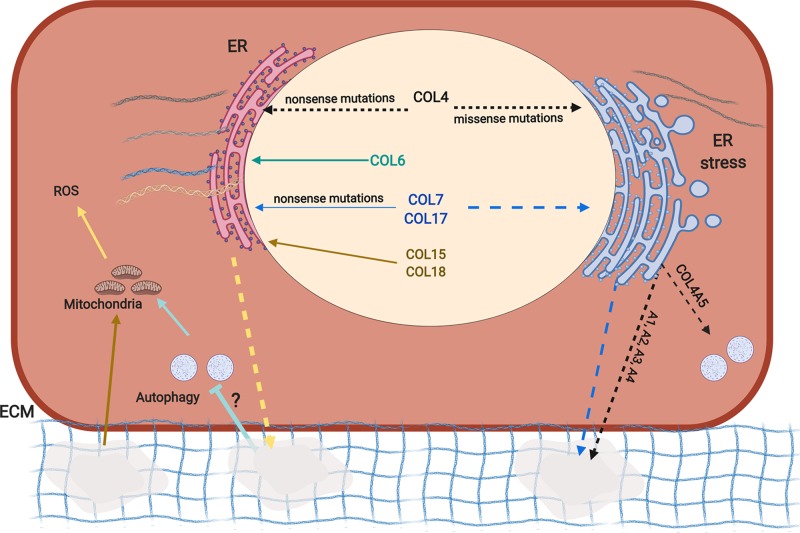
Figure 1. Overview of disease mechanisms caused by mutations in BM collagens.
Collagen IV (COL4, black arrows) proteins harbouring nonsense mutations are processed in the ER, resulting in reduced secretion of proteins that are incorporated in the ECM, causing matrix defects (indicated by holes). Missense mutations in collagen IV and VII (COL7) can result in their ER retention and ER stress, and subsequent reduction in secretion (dashed arrows). COL4A5 mutations can also induce autophagy. Mutant collagen IV may also be incorporated in the ECM, resulting in BM defects. Nonsense mutations in collagen VI (COL6), VII (COL7), XVII (COL17), XV (COL15) and XVIII (COL18) (solid arrows) do not result in ER retention but rather in reduced incorporation in the ECM (yellow dashed arrow). Matrix defects resulting from COL6 mutations (light green) lead to failure to induce autophagy (via an as yet unknown mechanism) and result in mitochondrial defects and production of reactive oxygen species (ROS). Matrix defects, resulting from COL15 deficiency (brown arrow) also cause mitochondrial defects and ROS production.
Table 1.
Frequently used animal models of collagen-related genetic diseases
| BM component | Affected gene | Animal model | Disease phenotype (or human equivalent) | References |
|---|---|---|---|---|
| Collagen IV | Col4a1 | Mouse missense mutations | Cerebrovascular disease intracerebral haemorrhage Kidney disease Myopathy Eye defects HANAC syndrome |
[21–24] |
| Col4a1/ Col4a2 double null | Mouse | Embryonically lethal, growth retardation,vascular defects | [10] | |
| Col4a1 (Cg25c) and Col4a2 (Vkg) | Drosophila missense and loss of function mutation | Intestinal defects, myopathy | [25] | |
| emb-9; let-2 (Cola4a1, Col4a2) | Caenorhabditis elegans misssense mutations | Embryonically lethal | [26] | |
| Col4a2 | Mouse missense mutations | Cerobrovascular, ocular, renal and muscle defects | [21] | |
| Col4a3 | Mouse knockout and missense mutation | Autosomal recessive and dominant AS | [27,28,29] | |
| Col4a3 & Col4a4 double null | Mouse | Juvenile form of AS | [30] | |
| Col4a4 | Mouse missense mutation | Autosomal recessive AS | [31] | |
| Col4a5 | Mouse knockout and nonsense mutation | X-linked AS | [32,33] | |
| Col4a5 | Zebrafish in-frame deletion | Defective retinal axon guiding | [34] | |
| Col4a6 | Zebrafish In-frame deletion |
Defective axon guiding, cerebellar granule cells defects | [16] | |
| Collagen VI | Col6a1 | Mouse knockout, heterozygous in frame deletion | Bethlem myopathy. Mitochondrial dysfunction, defective autophagy, fibre necrosis and osteoarthritis, abnormal collagen fibrillogenesis, CNS defect | [35,36] |
| Zebrafish morpholino knockdown | Bethlem myopathy, UCMD | [37] | ||
| Zebrafish knockdown | Bethlem myopathy, UCMD, myosclerosis | [38] | ||
| Col6a3 | Mouse in-frame deletion | Dominant mild myopathy with decreased muscle mass | [39] | |
| Zebrafish knockdown, in frame deletion | Bethlem myopathy (knockdown), Ullrich syndrome (in frame deletion) | [37,40] | ||
| Col6a4 | Zebrafish knockdown | Abnormal motoneuron axon growth | [38] | |
| Collagen VII | Col7a1 | Mouse knockout hypomorph mutation | Recessive dystrophic epidermolysis bullosa | [41,42] |
| Collagen XV | Col15a1 | Mouse | Mild skeletal myopathy Cardiomyopathy Vascular dysfunction Defects in nerve development and myelination |
[43] |
| Drosophila hypomorph mutant: piggybac transposon | Neuronal function defects, cardiomyocyte, skeletal muscle defects | [44,45] | ||
| Zebrafish morpholino knockdown of Col15a1a; Col15a1b knockdown | Defective notochord and muscle development; motor axon guidance defects and muscle atrophy | [46,47], | ||
| Collagen XVII | Col17a1 | Mouse knockout | Non-Herlitz epidermolysis bullosa, growth retardation, enamel hypoplasia | [48] |
| Zebrafish col17a1a knockdown; Col17a1b knockdown | Junctional epidermolysis bullosa (Col17a1a); neuronal defect (Col17a1b) | [49] | ||
| Collagen XVIII | Col18a1 | Mouse Col18a1 knockout | Knobloch syndrome; human pigment dispersion syndrome, hydrocephalus, kidney defect, adipocyte differentiation defect-metabolic defect | [50] |
| Col15a1 and Col18a1 knockout | [51] | |||
| Col18a1 isoform-specific knockout | [52] | |||
| cle-1 (Col18) | C. elegans | Defects in cell and axon migration and neuromuscular synapse function | [53] |
Due to space limitations, only the original references describing the animal model could be included. HANAC (hereditary angiopathy with nephropathy aneurysm and cramps), CNS (central nervous system) UCMD (Ullrich congenital muscular dystrophy)
Reduced α3α4α5(IV) GBM levels cause matrix defects, remodelling and fibrosis in AS. Recent evidence from Col4a3−/− mice supports a central role for mechanical strain [54] on the GBM in this process as it causes endothelin expression in mesangial cells, which results in endothelin A receptor expression in glomeruli. This leads to mesangial cell invasion [55], expression of laminin α 2-chain containing laminins, and focal adhesion kinase activation in podocytes, producing an inflammatory state and matrix metalloproteinase (MMP) activation [56], indicating matrix remodelling. In part, the fibrosis and MMP activation is influenced by LOXL2 (lysyl oxidase-like 2) collagen cross-linking activity [54], matrix-cell signalling via integrins α1β1 and α2β1, as well as DDR receptors [3,57].
The pathomolecular mechanisms of dominant AS can also include primary intracellular responses to expressing dominant mutations as a Col4a3 glycine mutation in cultured podocytes and Col4a3 knockin mice caused ER stress, a stress response activated by misfolded protein accumulation (Figure 1) [29]. Similarly, COL4A5 glycine mutations can induce ER stress and activate autophagy in patient fibroblasts [58] (Figure 1). Intracellular signalling involving STAT3 and TGFβ1 [59,60], which leads to fibrosis, have also been implicated but how they are activated remains less clear. Similarly, the relative contribution of ER stress to dominant AS remains unknown, but it may influence disease expressivity, similar to COL4A2-associated stroke [61].
Mutations affecting α1α1α2(IV)
The role of COL4A1/COL4A2 in human disease was identified through analysis of mice with Col4a1/Col4a2 missense mutations (Table 1) [21–23]. These mutations cause a dominant multisystemic disorder including cerebral small vessel disease, intracerebral haemorrhage (ICH), glomerular and tubular kidney phenotypes, eye defects and myopathy [24,62,63] (OMIM 120130, 120090). The location and nature of mutations affects clinical outcome with mutations affecting the CB3 region of α1(IV) causing the clinical subentity HANAC (hereditary angiopathy, nephropathy, aneurysm and cramps) syndrome, [63] and those in the miRNA-29 binding site of the 3′-UTR of COL4A1 the ischaemic small vessel disease PADMAL (pontine autosomal dominant microangiopathy with leukoencephalopathy) [64]. In mice, Col4a1 mutations affecting the X or Y residue of the Gly-X-Y repeat are less severe compared with glycine mutations [23], as are Col4a2 mutations [4]. Importantly, data from patients, mice and Caenorhabditis elegans indicate that disease outcome is influenced by genetic and environmental modifiers such as matrix proteins, vaginal birth and exercise [4,65]. The vast majority (approximately 80%) of COL4A1/4A2 mutations are missense mutations but nonsense or 3′-UTR mutations also occur, supporting pathogenicity of altered levels [4,64]. Interestingly, common variants in COL4A1/COL4A2 are risk factors for major vascular disease such as ICH [66] and coronary artery disease [67], but the proportion of patients in which these mutations occur remains unclear. Insight is emerging regarding the cellular origin of the phenotypes whereby both endothelial cell and smooth muscle cells contribute to ICH [68], vascular defects to the myopathy [69] and the lens to the eye disease [70].
Several non-mutually exclusive pathomolecular disease mechanisms have been proposed: intracellular retention of misfolded protein causing ER stress; reduced collagen IV incorporation in the BM and/or incorporation of mutant protein (Figure 1). Structural matrix defects alongside reduced α1α1α2(IV) levels in the BM, which can be associated with MMP activity and fibrosis [24,68], are almost universally observed [4,61,22,23,62], and data from patients with nonsense mutations [4] indicate matrix defects can be sufficient to cause cerebrovascular disease. However, the mechanism for dominant missense mutations is potentially more complicated given the possible contribution of ER stress due to collagen retention. In a family with a COL4A2 mutation, we uncovered that ER stress and not matrix defects was associated with disease [61], while in mice ICH severity correlated with levels of ER retention [68]. However, not every glycine mutation induces ER retention and stress [4]. These data suggest that ER stress can have a modifier effect for ICH and that cellular consequences and, potentially, mechanisms are mutation dependent. Moreover, our analysis of renal disease in mice revealed that the glomerulopathy was associated with matrix defects but tubular disease with ER stress [62,71], indicating cell/tissue-specific disease mechanisms [62,71]. This was subsequently confirmed for myopathy and ICH [72]. Given this mechanistic interplay of mutation-, cell- and tissue-dependent mechanisms, delineating the relative contribution of cellular consequences and matrix defects to the different phenotypes for different mutations will be informative and important.
Collagen VI
Vertebrates express six collagen VI α chains, encoded by genes COL6A1–COL6A6, which form beaded microfibrils that anchor BMs to the interstitial matrix. The major collagen VI monomer is α1α2α3(VI) but the roles of α4(VI), α5(VI) or α6(VI) remain less clear [73]. The central collagen domain of collagen VI is flanked by a globular N- and C-terminal domains, containing motifs with homology to von Willebrand factor type A domains. Within the ER, α chains form antiparallel dimers and then tetramers, which after secretion generate beaded microfibrils [6].
COL6A1/COL6A2/COL6A3 mutations cause severe Ullrich congenital muscular dystrophy (UCMD) and milder Bethlem myopathy, which can be inherited as autosomal dominant or recessive disorders, but collagen VI mutations can also affect the skin and tendon [6]. The role of COL6A4–COL6A6 in disease remains unclear [6] but in mice increased Col6a4 levels causes Hirschsprung’s disease type defects [74]. Similar to collagen IV diseases, genetic and environmental modifiers contribute to the large variability in clinical presentation.
Collagen VI diseases are associated with reduced levels or aberrant incorporation into the matrix of α1α2α3(VI) fibrils with a complete absence causing severe forms of disease [75] (Figure 1). Some heterozygous premature termination codon (PTC) mutations in COL6A1 cause Bethlem myopathy [76] but they are non-pathogenic when they occur in COL6A2/COL6A3 [6]. There is also clustering of dominant glycine mutations within the N-terminal end and recessive glycine mutations in more C-terminal regions, suggesting functional domains in these regions [6,77]. The N-terminal dominant glycine mutations and in-frame deletions often affect tetramer and microfibril assembly [78], while more C-terminal in-frame deletions and recessive glycine mutations can affect trimer formation [79]. ER stress has not been reported, and these data strongly support absence of collagen VI or presence of mutant collagen VI in the ECM as being causal.
Analysis of Col6a1−/−mice (Table 1) that develop Bethlem myopathy provided key insights into the disease mechanisms, including a role for neuromuscular junction and muscle stem cell defects [80,81]. Key steps in the pathomolecular mechanism includes the failure to induce autophagy, associated with reduced Bnip3 (Bcl2 Interacting Protein 3) and Beclin levels, to remove defective mitochondria [82]. This affects the permeability transition pore, reducing ATP synthesis [83], and causing mitochondrial dysfunction and reactive oxygen species (ROS) generation. Further insight is needed into how matrix defects cause these cellular defects and the interplay between them (Figure 1).
Collagen VII
Collagen VII is encoded by the COL7A1 gene, and three pro-α1(VII) α chains interact to form α1α1α1(VII) monomers which contain a central collagen domain, with 19 interruptions in the Gly-X-Y sequence, flanked by N-terminal (NC-1) and C-terminal (NC-2) domains [8]. Secreted pro-collagen VII molecules form antiparallel dimers, which are cleaved at the NC-2 domain by bone morphogenic protein 1 during their aggregation to form anchoring fibrils [84]. These anchoring fibrils mediate dermal–epidermal adhesion via binding of collagen VII to laminin-332 and collagen IV in the BM, and collagen I in the interstitial matrix [8]. Auto-antibodies against epitopes in the NC-1 domain cause the autoimmune skin blistering disorders epidermolysis bullosa acquisita (EBA) [85] and bullous systemic lupus erythematosus [86].
Mutations in COL7A1 cause dystrophic epidermolysis bullosa (DEB) (OMIM # 131750 # 226600) whereby the generally milder dominant DEB (DDEB) and non-syndromic congenital nail disorder-8 (OMIM # 607523) are due to missense mutations. The generally more severe recessive form of DEB (RDEB) is due to homozygous or compound heterozygous COL7A1 mutations, most frequently leading to nonsense mediated decay (NMD) and absence of collagen VII [8]. This causes blisters, wounding, inflammation, reduced myofibroblast removal and fibrosis. Collagen VII also plays a direct role in would healing by affecting keratinocyte migration via organising laminin-332 and impacting integrin α6β4 signalling, and supporting fibroblast migration and controlling their cytokine production [87]. Disease severity may also be subject to genetic modifiers as increased severity has been associated with a functional variant in MMP1 [88], which degrades collagen VII, causing increased degradation and increased RDEB severity. However, this association has been questioned [89].
The pathomolecular mechanism of RDEB is the absence of α1α1α1(VII) in the matrix affecting anchoring fibrils, weakening the BM and causing blistering (Figure 1). However, dominant missense mutations can cause ER retention, suggesting that α1α1α1(VII) production, secretion and storage are altered in patients [90]. However, its effect on ER stress and downstream effects has not been reported.
Collagen XVII
Collagen XVII (aka BPAG2, BP180) is a transmembrane homotrimer α1α1α1(XVII), consisting of three α1(XVII) chains that is important for cell–matrix interactions as a component of hemidesmosomes, securing the attachment of epithelial cells to BMs through binding laminin-332 and potentially collagen IV [91]. Collagen XVII plays a role in autoimmune blistering disease as autoantibodies to collagen XVII cause Bullous Pemphigoid (BP), the most common autoimmune blistering skin disease [48] and linear IgA dermatosis [91]. The latter is caused by auto-antigens within the ectodomain of soluble collagen XVII, which can be generated by ADAM (A Disintegrin And Metalloproteinase) -mediated protease cleavage of collagen XVII [92].
A series of elegant papers has recently identified non-structural functions of collagen XVII. This includes a key role in stem cell maintenance/homeostasis as depletion of collagen XVII in skin, which occurs with normal ageing [93], causes stem cells in the hair follicle to terminally differentiate, causing hair loss [94]. An important role in ageing was also uncovered as replenishment of collagen XVII reverses the hyperproliferation of interfollicular epidermis induced with ageing skin [93], and promotes symmetrical division of epidermal stem cells, which outcompete asymmetrically dividing stem cells. This reversed skin ageing and improved wound healing [95].
COL17A1 mutations cause junctional epidermolysis bullosa (OMIM # 226650), which can also be caused by mutations in the laminin genes LAMA3, LAMB3, LAMC2 and integrin genes ITGA3, ITGA6 and ITGB4 [7,96,97], and is characterised by dermal–epidermal separation leading to skin blistering, and epithelial recurrent erosion dystrophy, alopecia and nail dystrophy [96]. The majority of mutations are nonsense mutations but some glycine missense mutations occur, which cause intracellular retention and thermal instability of the protein [98], indicating protein misfolding. However, it is unclear if this results in ER stress (Figure 1). The reduction or complete absence of collagen XVII in the matrix affects the hemidesmosomes and stability of the dermal BM zone leading to EB (Figure 1), and genotype–phenotype analysis revealed approximately 12–25% of normal protein is required for skin stability [96]. Dominant COL17A1 missense mutations have recently been identified as a cause of epithelial recurrent corneal erosion dystrophy (OMIM# 122400) [99] but no functional analysis was performed.
Collagen XV and XVIII
Collagen XV and XVIII are multiplexin (multiple triple-helix domains with interruptions) collagens that are structurally closely related heparan sulphate proteoglycans. The collagen domain of collagen XVIII is flanked by a N-terminal non-collagenous domain containing thrombospondin-1-like and frizzled motifs [100], and a C-terminal trimerisation domain, which harbours the fragment endostatin that shares homology with restin in collagen XV [9]. The variable length of the N-terminal fragment of α1(XVIII) generates three isoforms with different expression patterns and functions [9]. For example, the short isoform is essential for retinal development while the medium and long isoforms are required for hepatocyte survival following injury [101] and adipose tissue formation [102]. Collagen XVIII is embedded into the BM by its C-terminal domain that binds perlecan and laminin, while its N-terminal domain extends into the ECM. Protease cleavage releases the anti-angiogenic fragment endostatin which inhibits endothelial cell migration and tumour growth. However, its efficacy for human cancer remains debated [9]. In addition, endostatin affects processes such as autophagy [103], its absence causes glomerular and tubular kidney defects, and exacerbates nephritis [104], and an endostatin-derived peptide can reduce fibrosis [105]. Elegant recent work using mouse models established that the frizzled motif in the N-terminal domain is required for preadipocyte differentiation and the ability of white adipose tissue to store fat by inhibiting Wnt signalling [102] (Table 1).
COL18A1 mutations, the vast majority of which are nonsense mutations, cause recessive Knobloch syndrome (OMIM # 267750) that is characterised by eye defects (e.g. myopia, vitreoretinal degeneration, macular abnormalities) and occipital encephalocele. Deficiency of Col18a1 in mice mimics Knobloch syndrome (Table 1 and Figure 1) but also affects the kidney and fat metabolism [9]. Many vascular defects of Knobloch syndrome, including persistence of the hyaloid vasculature, have been attributed to the absence of the short isoform and endostatin which generates a pro-angiogenic environment, although absence of the thrombospondin domain may also contribute [9,52]. This pro angiogenic environment due to the absence of endostatin, potentially combined with its effects on proteostasis and autophagy [106], are also associated with age-related retinal pigment epithelium degeneration in Col18a1−/− mice, a hallmark of age-related macular degeneration.
Collagen XV and XVII are structurally similar molecules with anti-angiogenic fragments, although data have challenged that for collagen XV this is mediated via its restin fragment [9,107]. Despite this structural similarity, these collagens have different functions. Collagen XV is highly expressed in tissues such as heart and skeletal smooth muscle, and structural analysis combined with muscle and vascular defects in Col15a1−/− deficient mice supports it acts as a spring between the BM and interstitial matrix to protect against contractile forces [108,43]. Investigation of Col15/18-deficient muscle defects in Drosophila uncovered they are due to altered integrin activity causing mitochondrial defects, reduced ATP generation and ROS production [44] (Figure 1). Treatment with cyclosporin A or losartan reduced these phenotypes [44], indicating a pathomolecular mechanism shared with COL6A1 mutations (see above) (Figure 1).
Analysis of zebrafish, which has two Col15a1 isoforms, Col15a1a and Col15a1b, suggests a potential developmental origin to this muscle phenotype as Col15a1a deficiency causes defective notochord and muscle development [46]. Deficiency of Col15a1b leads to motor axon guidance defects and muscle atrophy [47], establishing a role in neuromuscular development, while in mice α1α1α1(XV) contributes to peripheral nerve development and myelination [109]. In line with its high levels of expression in the heart, deficiency of collagen XV causes cardiomyopathy associated with matrix remodelling, defects in vascular permeability and haemodynamics, and increased stiffness [110]. Recent genetic analysis suggested COL15A1 can act as a modifier of thoracic aortic aneurysm severity [111] and has indicated a potential role in primary open angle glaucoma [112] and patients with Cuticular drusen (CD), a subtype of age-related macular degeneration [113]. These data indicate a growing role for collagen XV in human biology and disease.
Treatment intervention
The identification of underlying disease mechanisms has instigated a plethora of different approaches to modulate disease outcome. Broadly, these strategies can be divided into those that attempt to cure or overcome the genetic defect (gene therapy-based approaches), those that aim to modulate the pathomolecular disease mechanisms or cellular responses of mutations, and those that target further downstream targets (Figure 2 and Table 2). While an in-depth overview is beyond the scope of this review, a few examples of each class are discussed below.
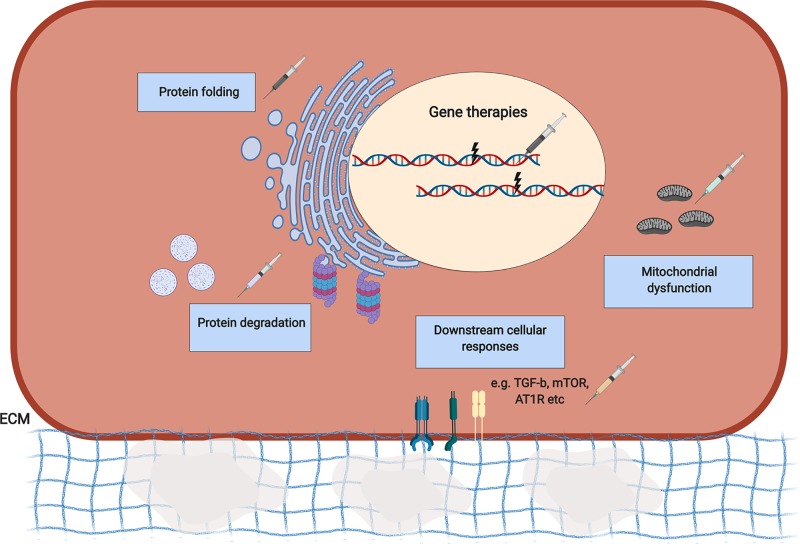
Figure 2. Overview of therapeutic strategies for BM collagen disorders.
Gene therapy approaches have been implemented to silence disease alleles of COL6A1 and COL7A1 mutations using AONs and siRNAs. RNA trans-splicing strategies have also been implemented. Targeted pathomolecular effects of mutations include intracellular retained misfolded proteins and ER stress, which has been used for collagen IV mutations via chemical chaperones to increase protein folding and increase secretion of proteins. Additionally, promoting autophagy and the proteasome may promote degradation of misfolded proteins. Autophagy and mitochondrial defects due to COL6A1/COL15A1 mutations have also been targeted pharmacologically and through diet. As an example of targeting downstream cellular responses or signalling to modulate disease, TGF-β signalling has been targeted in epidermolysis bullosa due to COL7A1 mutations. For a more detailed overview of therapeutic strategies, we refer the reader to Table 2. Abbreviation: AON, antisense oligonucleotide.
Table 2.
Mechanism-based therapeutic strategies for collagen-related disease
| Gene | Disease | Mechanism-target | Treatment | References |
|---|---|---|---|---|
| COL4A1, COL4A2, COL4A5 | Col4a1 disease and AS | ER retained protein, ER stress pathway | Chemical chaperones, e.g. 4-phenylbutyrate (4PBA) to reduce ER stress and increase secretion of correctly folded protein | [58,61,68,71] |
| COL4A5 | AS | Blood pressure by targeting renin–angiotensin system | Angiotensin-converting enzyme inhibitors, e.g. ramipril | * [114] |
| Angiotensin II type 1 receptor blockers, e.g. losartan | *[115] | |||
| Fibrosis-Transforming growth factor-β 1 (TGF-β), Connective tissue growth factor, miR-21 | HMG-CoA-reductase inhibitor (cerivastatin) | [116] | ||
| Vasopeptidase inhibitor AVE7688 | [117] | |||
| Anti-miR-21 oligonucleotides | [118] | |||
| Oxidative stress, inflammation and fibrosis: Nrf2 | Nrf2 activator, e.g. bardoxolone methyl (BARD) | *[119] | ||
| STAT3 signalling | STAT3 inhibitor, e.g. stattic | [59] | ||
| Functional correction | Gene therapy: restoration of network proof of concept | [120] | ||
| Cell therapy Bone marrow-derived stem cells |
[121] | |||
| Amniotic fluid stem cells | [122] | |||
| COL6A1 | Bethlem myopathy, Ullrich congenital muscular dystrophy | Reactivation of autophagy | mTOR inhibitor, e.g. Rapamycin | [82] |
| Low protein diet | *[123] | |||
| Spermidine | [124] | |||
| Mitochondrial defect: opening of Mitochondrial permeability transition pore (mPTP) | Cyclosporin A Cyclophilin inhibitor, e.g. NIM811, Debio25 (alisporivir) | *[125,83,37,126,127] | ||
| Metabolic defects | Adiponectin | [128] | ||
| Functional correction Collagen VI-producing cells | Cell therapy: fibroblast grafting | [81] | ||
| Adipose-derived stem cell transplant | [129] | |||
| Dominant negative mutation | Gene silencing with AONs or siRNAs | [130,131,132,133] | ||
| Splice mutations | AON-mediated exon skipping | [134] | ||
| COL7A1 | DEB | Wound healing | Injecting fibroblast cells | *[135] |
| Grafting revertant mosaicism skin-keratinocytes | *[136] | |||
| Genome editing patient-derived IPSC cells and transplant | [137] | |||
| Mesenchymal stromal cell therapy transplant | *[138] | |||
| Human placental‐derived stem cell transplant | [139] | |||
| Functional correction | Exon skipping | [140] | ||
| RDEB | Functional correction | Ex vivo TALEN gene editing | [141] | |
| Ex vivo CRISPR Genome editing keratinocytes | *[142] | |||
| RNA trans-splicing | [143] | |||
| Polymer-mediated cDNA delivery | [144] | |||
| Ex vivo retroviral transduction | [145] | |||
| AON-mediated exon skipping | [146] | |||
| Read through of Premature termination codons (PTCs) | See review [147] | |||
| Fibrosis: TGF-β | Angiotensin II type 1 receptor antagonist: losartan | [148] | ||
| DDEB | Functional correction | Allele-specific silencing via siRNA | [149] | |
| Gene editing using NHEJ to knockout mutant allele | [146,150] | |||
| Deficient collagen VII levels in ECM | Protein replacement therapy | [151,152] | ||
| COL15A1/COL18A1 | Muscular defect | Mitochondrial defect (opening permeability transition pore) and ROS production | Cyclosporine A Angiotensin II type 1 receptor antagonist, e.g. losartan | [44] |
Clinical trials are indicated by *. Abbreviation: AON, antisense oligonucleotide.
Gene therapy approaches
Gene therapy approaches are attractive as they are independent of disease mechanism and provide an actual cure. For missense mutations, allelic silencing using siRNA or gapmer antisense oligonucleotides (AONs) has been successfully applied to COL6A1 and COL7A1 mutations, with AONs affecting wild-type allele expression to a lesser extent [132,133], although this requires mutation specific compounds. Splice mutations in COL6A1/COL7A1 have also been countered using AONs that mediate exon skipping [134], but trans-splicing to obtain wild-type protein expression can be applied to splicing and PTC-generating mutations [143].
Skin disorders are particularly amenable to ex vivo genome editing using CRISPR-Cas9 from patient-derived induced pluripotent stem cells combined with skin grafting. This has been successful for junctional EB due to laminin mutations [153,145]. While, stem cell treatment employing iPSCs for some tissues/diseases may have its challenges, enormous progress has been made, making this an exciting approach [154]. For RDEB clinical trials employing ex vivo retroviral transduction of patient keratinocytes or fibroblasts to induce collagen VII expression followed by grafting or fibroblast injection, respectively, has shown promise [145,155]. These represent mutation-independent approaches for BM collagens.
ER stress and other disease mechanisms
Dominant missense mutations occur across collagen types and resultant protein misfolding, ER retention and ER stress represent potential convergent disease mechanisms. Promoting protein degradation, as performed for collagen X mutations [156] or protein folding through chaperones are potential strategies to address this. We and others established that the chemical chaperone 4-PBA reduces ER stress due to COL4A2 or COL4A5 mutation in patient cells [58,61]. Importantly, 4-PBA reduced cerebrovascular disease severity in mice carrying Col4a1 missense mutations at the C-terminal end of the collagen domain that cause intracellular retention [68,71], as a preventative approach and as a treatment for established disease [71]. However, we also established that 4-PBA was not effective for eye and kidney disease due to the same glycine mutation and that PBA increased secretion of collagen IV and weakened the BM [71]. Therefore, 4-PBA treatment may be contra-indicative for matrix-related phenotypes or missense mutations that do not cause ER stress [71]. This was subsequently confirmed for Col4a1 myopathy in mice carrying a more N-terminal mutation that does not induce ER stress [72]. Therefore, treatments will need stratification according to the mechanisms of mutations.
Mitochondrial dysfunction and dysregulation of autophagy resulting from collagen VI or XV/XVIII deficiency was ameliorated by dietary and pharmacological treatments when tested in mice and patient-derived cells [82,37,123,126], indicating a convergent mechanism and treatment. Re-instating collagen expression via cell graft or cell transplant represents a different approach and has shown promise for collagen VI and VII [81,145,155,129]. Re-instating Col6a1 expression also rescued the defective muscle stem cell renewal in Col6a1−/− mice, which was recalcitrant to autophagy treatment [81], suggesting a combinatorial therapy may be required.
While the above approaches target the intracellular consequences, due to our relatively poor understanding of the matrix defects and the subsequent aberrant matrix–cell signalling, limited progress has been made. An emerging example is the pharmacological inhibition of DDR1 to preserve renal function and reduce renal fibrosis in Col4a3−/− mice [157], illustrating the potential power of this approach.
Targeting downstream effect and treatment of symptoms
Alternatively, interventions have focused on particular disease symptoms or further downstream consequences. For example, inhibitors of the renin–angiotensin system (e.g. ramipril) are used to reduce blood pressure in AS to delay kidney failure [3].
Fibrosis is a common feature of matrix disorders and targeting TGF-β-induced matrix deposition has been undertaken via anti-miR-21 or pharmacological STAT3 inhibition in Alport mice [118,59]. Interestingly, inhibition of TGF-β signalling using losartan was also effective in RDEB, indicating that convergent downstream pathways represent a potential effective target for different phenotypes. Finally, the compound bardoxolone, which reduces inflammation and fibrosis in chronic kidney disease, is currently in a clinical trial for AS [119]. However as bardoxolone increases glomerular filtration rate, which may elevate the stress on the damaged GBM of Alport patients, this approach remains debated [158].
Outlook
BM collagen disorders are archetypical rare disorders for which treatment development can represent an economic challenge. Therefore, identifying convergent mechanisms and targets represent a very attractive proposition for both industry and patients, as it stands to accelerate treatment development. However, the multi-systemic nature, variable expressivity and disease severity pose a huge challenge. Therefore, there is an urgent need to increase our fundamental understanding of BM biology and acquire in-depth knowledge of disease mechanisms from gene to patient level. This includes, but is not limited to, the identification of genetic modifiers, a complete description of phenotypes in which BM collagens play a role, and establishing the relative contribution of intra/extracellular mechanisms including matrix–cell signalling to disease. In addition, the multi-systemic nature of these diseases with mutation-, cell- and tissue-specific mechanisms needs to be considered. However, while a ‘one-size-fits-all’ therapy is not possible, the identification of convergent aspects does raise the possibility that several diseases or mutation types may be amenable to manipulation of a shared mechanism. The stratified treatments would therefore focus on pathophysiological mechanisms and unite clinically disparate phenotypes.
Summary
Basement membrane collagens play an important role in a growing number of Mendelian disorders and common traits, and increasing our knowledge of basement membrane biology and disease mechanisms will help address the need for treatments.
Mechanistic studies have provided insight into the pathomolecular mechanisms of BM diseases, revealing that intra- and extracellular mechanisms are associated with disease.
The identification of disease mechanisms shared between distinct diseases raises the potential that several diseases or mutation types may be treated by manipulation of a shared mechanism.
Disease mechanism-based therapies are being explored using preclinical animal models and several have been taken forward to clinical trials.
Acknowledgments
We would like to apologise to our colleagues whose work we could not include due to space constraints. Images were generated using the BioRender software.
Abbreviations
- AON
antisense oligonucleotide
- AS
Alport syndrome
- BM
basement membrane
- DDR
discoidin domain receptor
- DEB
dystrophic epidermolysis bullosa
- ECM
extracellular matrix
- ER
endoplasmic reticulum
- GBM
glomerular BM
- ICH
intracerebral haemorrhage
- iPSC
induced Pluripotent Stem Cell
- NC
non-collagenous
- MMP
matrix metalloproteinase
- OMIM
Online Mendelian Inheritance in Man
- PTC
premature termination codon
- RDEB
recessive form of DEB
- ROS
reactive oxygen species
Funding
This work was supported by the MRC [grant number MR/R005567-1]; BHF [grant number PG/15/92/31813]; Stroke Association [grant number PPA 2016/02]; and Heart Research U.K. [grant number RG 2664/17/20] to T.V.A.
Competing Interests
The authors declare that there are no competing interests associated with the manuscript.
References
- 1.Randles M.J., Humphries M.J. and Lennon R. (2017) Proteomic definitions of basement membrane composition in health and disease. Matrix Biol. 57-58, 12–28 10.1016/j.matbio.2016.08.006 [DOI] [PubMed] [Google Scholar]
- 2.Ishikawa Y., Ito S., Nagata K., Sakai L.Y. and Bächinger H.P. (2016) Intracellular mechanisms of molecular recognition and sorting for transport of large extracellular matrix molecules. Proc. Natl. Acad. Sci. U.S.A. 113, E6036–E6044 10.1073/pnas.1609571113 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Chew C. and Lennon R. (2018) Basement membrane defects in genetic kidney diseases. Front. Pediatrics 6, 1–16, 10.3389/fped.2018.00011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Jeanne M. and Gould D.B. (2017) Genotype-phenotype correlations in pathology caused by collagen type IV alpha 1 and 2 mutations. Matrix Biol. 57–58, 29–44 10.1016/j.matbio.2016.10.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mao M., Alavi M.V., Labelle-Dumais C. and Gould D.B. (2015) Type IV collagens and basement membrane diseases: cell biology and pathogenic mechanisms. Curr. Top. Membr. 76, 61–116 [DOI] [PubMed] [Google Scholar]
- 6.Lamandé S.R. and Bateman J.F. (2018) Collagen VI disorders: insights on form and function in the extracellular matrix and beyond. Matrix Biol. 71–72, 348–367 10.1016/j.matbio.2017.12.008 [DOI] [PubMed] [Google Scholar]
- 7.Condrat I., He Y., Cosgarea R. and Has C. (2019) Junctional epidermolysis bullosa: allelic heterogeneity and mutation stratification for precision medicine. Front. Med. 5, 1–9, 10.3389/fmed.2018.00363 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Nyström A., Bernasconi R. and Bornert O. (2018) Therapies for genetic extracellular matrix diseases of the skin. Matrix Biol. 71–72, 330–347 10.1016/j.matbio.2017.12.010 [DOI] [PubMed] [Google Scholar]
- 9.Heljasvaara R., Aikio M., Ruotsalainen H. and Pihlajaniemi T. (2017) Collagen XVIII in tissue homeostasis and dysregulation—lessons learned from model organisms and human patients. Matrix Biol. 57–58, 55–75 10.1016/j.matbio.2016.10.002 [DOI] [PubMed] [Google Scholar]
- 10.Pöschl E., Schlötzer-Schrehardt U., Brachvogel B., Saito K., Ninomiya Y. and Mayer U. (2004) Collagen IV is essential for basement membrane stability but dispensable for initiation of its assembly during early development. Development 131, 1619–1628 10.1242/dev.01037 [DOI] [PubMed] [Google Scholar]
- 11.Paavola K.J., Sidik H., Zuchero J.B., Eckart M. and Talbot W.S. (2014) Type IV collagen is an activating ligand for the adhesion G protein–coupled receptor GPR126. Sci. Signal. 7, ra76 10.1126/scisignal.2005347 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kashtan C.E., Ding J., Garosi G., Heidet L., Massella L., Nakanishi K.. et al. (2018) Alport syndrome: a unified classification of genetic disorders of collagen IV α345: a position paper of the Alport Syndrome Classification Working Group. Kidney Int. 93, 1045–1051 10.1016/j.kint.2017.12.018 [DOI] [PubMed] [Google Scholar]
- 13.Guan M., Ma J., Keaton J.M., Dimitrov L., Mudgal P., Stromberg M.. et al. (2016) Association of kidney structure-related gene variants with type 2 diabetes-attributed end-stage kidney disease in African Americans. Hum. Genet. 135, 1251–1262 10.1007/s00439-016-1714-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Voskarides K., Damianou L., Neocleous V., Zouvani I., Christodoulidou S., Hadjiconstantinou V.. et al. (2007) COL4A3/COL4A4 mutations producing focal segmental glomerulosclerosis and renal failure in thin basement membrane nephropathy. J. Am. Soc. Nephrol. 18, 3004–3016 10.1681/ASN.2007040444 [DOI] [PubMed] [Google Scholar]
- 15.Bullich G., Trujillano D., Santín S., Ossowski S., Mendizábal S., Fraga G.. et al. (2014) Targeted next-generation sequencing in steroid-resistant nephrotic syndrome: mutations in multiple glomerular genes may influence disease severity. Eur. J. Hum. Genet. 23, 1192 10.1038/ejhg.2014.252 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Takeuchi M., Yamaguchi S., Yonemura S., Kakiguchi K., Sato Y., Higashiyama T.. et al. (2015) Type IV collagen controls the axogenesis of cerebellar granule cells by regulating basement membrane integrity in zebrafish. PLoS Genet. 11, e1005587 10.1371/journal.pgen.1005587 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Rost S., Bach E., Neuner C., Nanda I., Dysek S., Bittner R.E.. et al. (2013) Novel form of X-linked nonsyndromic hearing loss with cochlear malformation caused by a mutation in the type IV collagen gene COL4A6. Eur. J. Hum. Genet. 22, 208 10.1038/ejhg.2013.108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kang J.S., Wang X.-P., Miner J.H., Morello R., Sado Y., Abrahamson D.R.. et al. (2006) Loss of α3/α4(IV) collagen from the glomerular basement membrane induces a strain-dependent isoform switch to α5α;6(IV) collagen associated with longer renal survival in Col4a3−/− Alport mice. J. Am. Soc. Nephrol. 17, 1962–1969 10.1681/ASN.2006020165 [DOI] [PubMed] [Google Scholar]
- 19.Murata T., Katayama K., Oohashi T., Jahnukainen T., Yonezawa T., Sado Y.. et al. (2016) COL4A6 is dispensable for autosomal recessive Alport syndrome. Sci. Rep. 6, 29450 10.1038/srep29450 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Funk S.D., Bayer R.H., Malone A.F., McKee K.K., Yurchenco P.D. and Miner J.H. (2018) Pathogenicity of a human laminin β2 mutation revealed in models of Alport syndrome. J. Am. Soc. Nephrol. 29, 949–960, 10.1681/ASN.2017090997 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Favor J., Gloeckner C.J., Janik D., Klempt M., Neuhäuser-Klaus A., Pretsch W.. et al. (2007) Type IV procollagen missense mutations associated with defects of the eye, vascular stability, the brain, kidney function and embryonic or postnatal viability in the mouse, Mus musculus: an extension of the Col4a1 allelic series and the identification of the first two Col4a2 mutant alleles. Genetics 175, 725–736 10.1534/genetics.106.064733 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gould D.B., Phalan F.C., Breedveld G.J., van Mil S.E., Smith R.S., Schimenti J.C.. et al. (2005) Mutations in Col4a1 cause perinatal cerebral hemorrhage and porencephaly. Science 308, 1167–1171 10.1126/science.1109418 [DOI] [PubMed] [Google Scholar]
- 23.Van Agtmael T., Schlötzer-Schrehardt U., McKie L., Brownstein D.G., Lee A.W., Cross S.H.. et al. (2005) Dominant mutations of Col4a1 result in basement membrane defects which lead to anterior segment dysgenesis and glomerulopathy. Hum. Mol. Genet. 14, 3161–3168 10.1093/hmg/ddi348 [DOI] [PubMed] [Google Scholar]
- 24.Chen Z., Migeon T., Verpont M.-C., Zaidan M., Sado Y., Kerjaschki D.. et al. (2016) HANAC syndrome Col4a1 mutation causes neonate glomerular hyperpermeability and adult glomerulocystic kidney disease. J. Am. Soc. Nephrol. 27, 1042–1054 10.1681/ASN.2014121217 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kelemen-Valkony I., Kiss M., Csiha J., Kiss A., Bircher U., Szidonya J.. et al. (2012) Drosophila basement membrane collagen col4a1 mutations cause severe myopathy. Matrix Biol. 31, 29–37 10.1016/j.matbio.2011.09.004 [DOI] [PubMed] [Google Scholar]
- 26.Gupta M.C., Graham P.L. and Kramer J.M. (1997) Characterization of alpha1(IV) collagen mutations in Caenorhabditis elegans and the effects of alpha1 and alpha2(IV) mutations on type IV collagen distribution. J. Cell Biol. 137, 1185–1196 10.1083/jcb.137.5.1185 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Cosgrove D., Meehan D.T., Grunkemeyer J.A., Kornak J.M., Sayers R., Hunter W.J.. et al. (1996) Collagen COL4A3 knockout: a mouse model for autosomal Alport syndrome. Genes Dev. 10, 2981–2992 10.1101/gad.10.23.2981 [DOI] [PubMed] [Google Scholar]
- 28.Miner J.H. and Sanes J.R. (1996) Molecular and functional defects in kidneys of mice lacking collagen alpha 3(IV): implications for Alport syndrome. J. Cell Biol. 135, 1403–1413 10.1083/jcb.135.5.1403 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Pieri M., Stefanou C., Zaravinos A., Erguler K., Stylianou K., Lapathitis G.. et al. (2014) Evidence for activation of the unfolded protein response in collagen IV nephropathies. J. Am. Soc. Nephrol. 25, 260–275 10.1681/ASN.2012121217 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lu W., Phillips C.L., Killen P.D., Hlaing T., Harrison W.R., Elder F.F.B.. et al. (1999) Insertional mutation of the collagen genes Col4a3 and Col4a4 in a mouse model of Alport syndrome. Genomics 61, 113–124 10.1006/geno.1999.5943 [DOI] [PubMed] [Google Scholar]
- 31.Korstanje R., Caputo C.R., Doty R.A., Cook S.A., Bronson R.T., Davisson M.T.. et al. (2014) A mouse Col4a4 mutation causing Alport glomerulosclerosis with abnormal collagen α3α;4α5(IV) trimers. Kidney Int. 85, 1461–1468 10.1038/ki.2013.493 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Rheault M.N., Kren S.M., Thielen B.K., Mesa H.A., Crosson J.T., Thomas W.. et al. (2004) Mouse model of X-linked Alport syndrome. J. Am. Soc. Nephrol. 15, 1466–1474 10.1097/01.ASN.0000130562.90255.8F [DOI] [PubMed] [Google Scholar]
- 33.Hashikami K., Asahina M., Nozu K., Iijima K., Nagata M. and Takeyama M. (2019) Establishment of X-linked Alport syndrome model mice with a Col4a5 R471X mutation. Biochem. Biophys. Rep. 17, 81–86 10.1016/j.bbrep.2018.12.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Xiao T. and Baier H. (2007) Lamina-specific axonal projections in the zebrafish tectum require the type IV collagen Dragnet. Nat. Neurosci. 10, 1529 10.1038/nn2002 [DOI] [PubMed] [Google Scholar]
- 35.Bonaldo P., Braghetta P., Zanetti M., Piccolo S., Volpin D. and Bressan G.M. (1998) Collagen VI deficiency induces early onset myopathy in the mouse: an animal model for Bethlem myopathy. Hum. Mol. Genet. 7, 2135–2140 10.1093/hmg/7.13.2135 [DOI] [PubMed] [Google Scholar]
- 36.Pan T.-C., Zhang R.-Z., Sudano D.G., Marie S.K., Bönnemann C.G. and Chu M.-L. (2003) New molecular mechanism for ullrich congenital muscular dystrophy: a heterozygous in-frame deletion in the COL6A1 gene causes a severe phenotype. Am. J. Hum. Genet. 73, 355–369 10.1086/377107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Telfer W.R., Busta A.S., Bonnemann C.G., Feldman E.L. and Dowling J.J. (2010) Zebrafish models of collagen VI-related myopathies. Hum. Mol. Genet. 19, 2433–2444 10.1093/hmg/ddq126 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ramanoudjame L., Rocancourt C., Lainé J., Klein A., Joassard L., Gartioux C.. et al. (2015) Two novel COLVI long chains in zebrafish that are essential for muscle development. Hum. Mol. Genet. 24, 6624–6639 10.1093/hmg/ddv368 [DOI] [PubMed] [Google Scholar]
- 39.Pan T.-C., Zhang R.-Z., Arita M., Bogdanovich S., Adams S.M., Gara S.K.. et al. (2014) A mouse model for dominant collagen VI disorders: heterozygous deletion of Col6a3 exon 16. J. Biol. Chem. 289, 10293–10307 10.1074/jbc.M114.549311 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Zech M., Lam D.D., Francescatto L., Schormair B., Salminen A.V., Jochim A.. et al. (2015) Recessive mutations in the α3 (VI) collagen gene COL6A3 cause early-onset isolated dystonia. Am. J. Hum. Genet. 96, 883–893 10.1016/j.ajhg.2015.04.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Heinonen S., Mannikko M., Klement J.F., Whitaker-Menezes D., Murphy G.F. and Uitto J. (1999) Targeted inactivation of the type VII collagen gene (Col7a1) in mice results in severe blistering phenotype: a model for recessive dystrophic epidermolysis bullosa. J. Cell Sci. 112, 3641–3648 [DOI] [PubMed] [Google Scholar]
- 42.Fritsch A., Loeckermann S., Kern J.S., Braun A., Bösl M.R., Bley T.A.. et al. (2008) A hypomorphic mouse model of dystrophic epidermolysis bullosa reveals mechanisms of disease and response to fibroblast therapy. J. Clin. Invest. 118, 1669–1679 10.1172/JCI34292 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Eklund L., Piuhola J., Komulainen J., Sormunen R., Ongvarrasopone C., Fässler R.. et al. (2001) Lack of type XV collagen causes a skeletal myopathy and cardiovascular defects in mice. Proc. Natl. Acad. Sci. U.S.A. 98, 1194–1199 10.1073/pnas.98.3.1194 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Momota R., Narasaki M., Komiyama T., Naito I., Ninomiya Y. and Ohtsuka A. (2013) Drosophila type XV/XVIII collagen mutants manifest integrin mediated mitochondrial dysfunction, which is improved by cyclosporin A and losartan. Int. J. Biochem. Cell Biol. 45, 1003–1011 10.1016/j.biocel.2013.02.001 [DOI] [PubMed] [Google Scholar]
- 45.Momota R., Naito I., Ninomiya Y. and Ohtsuka A. (2011) Drosophila type XV/XVIII collagen, Mp, is involved in Wingless distribution. Matrix Biol. 30, 258–266 10.1016/j.matbio.2011.03.008 [DOI] [PubMed] [Google Scholar]
- 46.Pagnon-Minot A., Malbouyres M., Haftek-Terreau Z., Kim H.R., Sasaki T., Thisse C.. et al. (2008) Collagen XV, a novel factor in zebrafish notochord differentiation and muscle development. Dev. Biol. 316, 21–35 10.1016/j.ydbio.2007.12.033 [DOI] [PubMed] [Google Scholar]
- 47.Guillon E., Bretaud S. and Ruggiero F. (2016) Slow muscle precursors lay down a collagen XV Matrix fingerprint to guide motor axon navigation. J. Neurosci. 36, 2663–2676 10.1523/JNEUROSCI.2847-15.2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Nishie W., Sawamura D., Goto M., Ito K., Shibaki A., McMillan J.R.. et al. (2007) Humanization of autoantigen. Nat. Med. 13, 378 10.1038/nm1496 [DOI] [PubMed] [Google Scholar]
- 49.Kim S.H., Choi H.Y., So J.-H., Kim C.-H., Ho S.-Y., Frank M.. et al. (2010) Zebrafish type XVII collagen: gene structures, expression profiles, and morpholino “knock-down” phenotypes. Matrix Biol. 29, 629–637 10.1016/j.matbio.2010.07.002 [DOI] [PubMed] [Google Scholar]
- 50.Fukai N., Eklund L., Marneros A.G., Oh S.P., Keene D.R., Tamarkin L.. et al. (2002) Lack of collagen XVIII/endostatin results in eye abnormalities. EMBO J. 21, 1535–1544 10.1093/emboj/21.7.1535 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ylikärppä R., Eklund L., Sormunen R., Muona A., Fukai N., Olsen B.R.. et al. (2003) Double knockout mice reveal a lack of major functional compensation between collagens XV and XVIII. Matrix Biol. 22, 443–448 10.1016/S0945-053X(03)00074-X [DOI] [PubMed] [Google Scholar]
- 52.Aikio M., Hurskainen M., Brideau G., Hägg P., Sormunen R., Heljasvaara R.. et al. (2013) Collagen XVIII short isoform is critical for retinal vascularization, and overexpression of the Tsp-1 domain affects eye growth and cataract formation collagen XVIII short isoform in the eye. Invest. Ophthalmol. Vis. Sci. 54, 7450–7462 10.1167/iovs.13-13039 [DOI] [PubMed] [Google Scholar]
- 53.Ackley B.D., Crew J.R., Elamaa H., Pihlajaniemi T., Kuo C.J. and Kramer J.M. (2001) The Nc1/endostatin domain of Caenorhabditis elegans type XVIII collagen affects cell migration and axon guidance. J. Cell Biol. 152, 1219–1232 10.1083/jcb.152.6.1219 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Cosgrove D., Dufek B., Meehan D.T., Delimont D., Hartnett M., Samuelson G.. et al. (2018) Lysyl oxidase like-2 contributes to renal fibrosis in Col4α3/Alport mice. Kidney Int. 94, 303–314 10.1016/j.kint.2018.02.024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Dufek B., Meehan D.T., Delimont D., Cheung L., Gratton M.A., Phillips G.. et al. (2016) Endothelin A receptor activation on mesangial cells initiates Alport glomerular disease. Kidney Int. 90, 300–310 10.1016/j.kint.2016.02.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Delimont D., Dufek B.M., Meehan D.T., Zallocchi M., Gratton M.A., Phillips G.. et al. (2014) Laminin α2-mediated focal adhesion kinase activation triggers alport glomerular pathogenesis. PLoS ONE 9, e99083 10.1371/journal.pone.0099083 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Rubel D., Frese J., Martin M., Leibnitz A., Girgert R., Miosge N.. et al. (2014) Collagen receptors integrin alpha2beta1 and discoidin domain receptor 1 regulate maturation of the glomerular basement membrane and loss of integrin alpha2beta1 delays kidney fibrosis in COL4A3 knockout mice. Matrix Biol. 34, 13–21 10.1016/j.matbio.2014.01.006 [DOI] [PubMed] [Google Scholar]
- 58.Wang D., Mohammad M., Wang Y., Tan R., Murray L.S., Ricardo S.. et al. (2017) The chemical chaperone, PBA, reduces ER stress and autophagy and increases collagen IV α5 expression in cultured fibroblasts from men with X-linked Alport syndrome and missense mutations. Kidney Int. Rep. 2, 739–748 10.1016/j.ekir.2017.03.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Yokota T., Omachi K., Suico M.A., Kamura M., Kojima H., Fukuda R.. et al. (2017) STAT3 inhibition attenuates the progressive phenotypes of Alport syndrome mouse model. Nephrol. Dialysis Transplant. 33, 214–223 10.1093/ndt/gfx246 [DOI] [PubMed] [Google Scholar]
- 60.Cosgrove D., Rodgers K., Meehan D., Miller C., Bovard K., Gilroy A.. et al. (2000) Integrin α1β1 and transforming growth factor-β1 play distinct roles in Alport glomerular pathogenesis and serve as dual targets for metabolic therapy. Am. J. Pathol. 157, 1649–1659 10.1016/S0002-9440(10)64802-X [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Murray L.S., Lu Y., Taggart A., Van Regemorter N., Vilain C., Abramowicz M.. et al. (2013) Chemical chaperone treatment reduces intracellular accumulation of mutant collagen IV and ameliorates the cellular phenotype of a COL4A2 mutation that causes haemorrhagic stroke. Hum. Mol. Genet. 23, 283–292 10.1093/hmg/ddt418 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Jones F.E., Bailey M.A., Murray L.S., Lu Y., McNeilly S., Schlötzer-Schrehardt U.. et al. (2016) ER stress and basement membrane defects combine to cause glomerular and tubular renal disease resulting from Col4a1 mutations in mice. Dis. Models Mech. 9, 165–176 10.1242/dmm.021741 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Meuwissen M.E.C., Halley D.J.J., Smit L.S., Lequin M.H., Cobben J.M., de Coo R.. et al. (2015) The expanding phenotype of COL4A1 and COL4A2 mutations: clinical data on 13 newly identified families and a review of the literature. Genet. Med. 17, 843 10.1038/gim.2014.210 [DOI] [PubMed] [Google Scholar]
- 64.Verdura E., Hervé D., Bergametti F., Jacquet C., Morvan T., Prieto-Morin C.. et al. (2016) Disruption of a miR-29 binding site leading to COL4A1 upregulation causes pontine autosomal dominant microangiopathy with leukoencephalopathy. Ann. Neurol. 80, 741–753 10.1002/ana.24782 [DOI] [PubMed] [Google Scholar]
- 65.Gotenstein J.R., Koo C.C., Ho T.W. and Chisholm A.D. (2018) Genetic suppression of basement membrane defects in Caenorhabditis elegans by gain of function in extracellular matrix and cell-matrix attachment genes. Genetics 208, 1499–1512 10.1534/genetics.118.300731 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Rannikmäe K., Davies G., Thomson P.A., Bevan S., Devan W.J., Falcone G.J.. et al. (2015) Common variation in COL4A1/COL4A2 is associated with sporadic cerebral small vessel disease. Neurology 84, 918–926 10.1212/WNL.0000000000001309 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Yang W., Ng F.L., Chan K., Pu X., Poston R.N., Ren M.. et al. (2016) Coronary-heart-disease-associated genetic variant at the COL4A1/COL4A2 locus affects COL4A1/COL4A2 expression, vascular cell survival, atherosclerotic plaque stability and risk of myocardial infarction. PLoS Genet. 12, e1006127 10.1371/journal.pgen.1006127 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Jeanne M., Jorgensen J. and Gould D.B. (2015) Molecular and genetic analyses of collagen type IV mutant mouse models of spontaneous intracerebral hemorrhage identify mechanisms for stroke prevention. Circulation 131, 1555–1565 10.1161/CIRCULATIONAHA.114.013395 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Guiraud S., Migeon T., Ferry A., Chen Z., Ouchelouche S., Verpont M.-C.. et al. (2017) HANAC Col4a1 mutation in mice leads to skeletal muscle alterations due to a primary vascular defect. Am. J. Pathol. 187, 505–516 10.1016/j.ajpath.2016.10.020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Mao M., Kiss M., Ou Y. and Gould D.B. (2017) Genetic dissection of anterior segment dysgenesis caused by a Col4a1 mutation in mouse. Dis. Models Mech. 10, 475–485 10.1242/dmm.027888 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Jones F.E., Murray L.S., McNeilly S., Dean A., Aman A., Lu Y.. et al. (2018) 4-Sodium phenyl butyric acid has both efficacy and counter-indicative effects in the treatment of Col4a1 disease. Hum. Mol. Genet. 28, 628–638 10.1093/hmg/ddy369 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Labelle-Dumais C., Schuitema V., Hayashi G., Hoff K., Gong W., Dao D.Q.. et al. (2019) COL4A1 mutations cause neuromuscular disease with tissue-specific mechanistic heterogeneity. Am. J. Hum. Genet. 104, 847–860 10.1016/j.ajhg.2019.03.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Fitzgerald J., Holden P. and Hansen U. (2013) The expanded collagen VI family: new chains and new questions. Connect. Tissue Res. 54, 345–350 10.3109/03008207.2013.822865 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Soret R., Mennetrey M., Bergeron K.F., Dariel A., Neunlist M., Grunder F.. et al. (2015) A collagen VI–dependent pathogenic mechanism for Hirschsprung’s disease. J. Clin. Invest. 125, 4483–4496 10.1172/JCI83178 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Camacho Vanegas O., Bertini E., Zhang R.-Z., Petrini S., Minosse C., Sabatelli P.. et al. (2001) Ullrich scleroatonic muscular dystrophy is caused by recessive mutations in collagen type VI. Proc. Natl. Acad. Sci. U.S.A. 98, 7516–7521 10.1073/pnas.121027598 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Lamandé S.R., Bateman J.F., Hutchison W., Gardner R.J.M., Bower S.P., Byrne E.. et al. (1998) Reduced Collagen VI causes Bethlem myopathy: a heterozygous COL6A1 nonsense mutation results in mRNA decay and functional haploinsufficiency. Hum. Mol. Genet. 7, 981–989 10.1093/hmg/7.6.981 [DOI] [PubMed] [Google Scholar]
- 77.Butterfield R.J., Foley A.R., Dastgir J., Asman S., Dunn D.M., Zou Y.. et al. (2013) Position of Glycine substitutions in the triple helix of COL6A1, COL6A2, and COL6A3 is correlated with severity and mode of inheritance in collagen VI myopathies. Hum. Mutat. 34, 1558–1567 10.1002/humu.22429 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Baker N.L., Mörgelin M., Pace R.A., Peat R.A., Adams N.E., Gardner R. J.M.. et al. (2007) Molecular consequences of dominant Bethlem myopathy collagen VI mutations. Ann. Neurol. 62, 390–405 10.1002/ana.21213 [DOI] [PubMed] [Google Scholar]
- 79.Lamandé S.R., Mörgelin M., Selan C., Jöbsis G.J., Baas F. and Bateman J.F. (2002) Kinked collagen VI tetramers and reduced microfibril formation as a result of Bethlem myopathy and introduced triple helical glycine mutations. J. Biol. Chem. 277, 1949–1956 10.1074/jbc.M109932200 [DOI] [PubMed] [Google Scholar]
- 80.Cescon M., Gregorio I., Eiber N., Borgia D., Fusto A., Sabatelli P.. et al. (2018) Collagen VI is required for the structural and functional integrity of the neuromuscular junction. Acta Neuropathol. (Berl.) 136, 483–499 10.1007/s00401-018-1860-9 [DOI] [PubMed] [Google Scholar]
- 81.Urciuolo A., Quarta M., Morbidoni V., Gattazzo F., Molon S., Grumati P.. et al. (2013) Collagen VI regulates satellite cell self-renewal and muscle regeneration. Nat. Commun. 4, 1964 10.1038/ncomms2964 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Grumati P., Coletto L., Sabatelli P., Cescon M., Angelin A., Bertaggia E.. et al. (2010) Autophagy is defective in collagen VI muscular dystrophies, and its reactivation rescues myofiber degeneration. Nat. Med. 16, 1313 10.1038/nm.2247 [DOI] [PubMed] [Google Scholar]
- 83.Irwin W.A., Bergamin N., Sabatelli P., Reggiani C., Megighian A., Merlini L.. et al. (2003) Mitochondrial dysfunction and apoptosis in myopathic mice with collagen VI deficiency. Nat. Genet. 35, 367–371 10.1038/ng1270 [DOI] [PubMed] [Google Scholar]
- 84.Rattenholl A., Pappano W.N., Koch M., Keene D.R., Kadler K.E., Sasaki T.. et al. (2002) Proteinases of the bone morphogenetic protein-1 family convert procollagen VII to mature anchoring fibril collagen. J. Biol. Chem. 277, 26372–26378 10.1074/jbc.M203247200 [DOI] [PubMed] [Google Scholar]
- 85.Sitaru C., Chiriac M.T., Mihai S., Büning J., Gebert A., Ishiko A.. et al. (2006) Induction of complement-fixing autoantibodies against type VII collagen results in subepidermal blistering in mice. J. Immunol. 177, 3461–3468 10.4049/jimmunol.177.5.3461 [DOI] [PubMed] [Google Scholar]
- 86.Grabell D.A., Matthews L.A., Yancey K.B. and Chong B.F. (2015) Detection of type VII collagen autoantibodies before the onset of bullous systemic lupus erythematosus type VII collagen autoantibodies before BSLE onset type VII collagen autoantibodies before BSLE onset. JAMA Dermatol. 151, 539–543 10.1001/jamadermatol.2014.4409 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Nyström A., Velati D., Mittapalli V.R., Fritsch A., Kern J.S. and Bruckner-Tuderman L. (2013) Collagen VII plays a dual role in wound healing. J. Clin. Invest. 123, 3498–3509 10.1172/JCI68127 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Titeux M., Pendaries V., Tonasso L., Décha A., Bodemer C. and Hovnanian A. (2008) A frequent functional SNP in the MMP1 promoter is associated with higher disease severity in recessive dystrophic epidermolysis bullosa. Hum. Mutat. 29, 267–276 10.1002/humu.20647 [DOI] [PubMed] [Google Scholar]
- 89.Kern J.S., Grüninger G., Imsak R., Müller M.L., Schumann H., Kiritsi D.. et al. (2009) Forty-two novel COL7A1 mutations and the role of a frequent single nucleotide polymorphism in the MMP1 promoter in modulation of disease severity in a large European dystrophic epidermolysis bullosa cohort. Br. J. Dermatol. 161, 1089–1097 10.1111/j.1365-2133.2009.09333.x [DOI] [PubMed] [Google Scholar]
- 90.Watanabe M., Natsuga K., Shinkuma S. and Shimizu H. (2018) Epidermal aspects of type VII collagen: implications for dystrophic epidermolysis bullosa and epidermolysis bullosa acquisita. J. Dermatol. 45, 515–521 10.1111/1346-8138.14222 [DOI] [PubMed] [Google Scholar]
- 91.Natsuga K., Watanabe M., Nishie W. and Shimizu H. (2018) Life before and beyond blistering: the role of collagen XVII in epidermal physiology. Exp. Dermatol. 1–7 10.1111/exd.13550 [DOI] [PubMed] [Google Scholar]
- 92.Franzke C.W., Tasanen K., Schäcke H., Zhou Z., Tryggvason K., Mauch C.. et al. (2002) Transmembrane collagen XVII, an epithelial adhesion protein, is shed from the cell surface by ADAMs. EMBO J. 21, 5026–5035 10.1093/emboj/cdf532 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Watanabe M., Natsuga K., Nishie W., Kobayashi Y., Donati G., Suzuki S.. et al. (2017) Type XVII collagen coordinates proliferation in the interfollicular epidermis. eLife 6, e26635 10.7554/eLife.26635 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Matsumura H., Mohri Y., Binh N.T., Morinaga H., Fukuda M., Ito M.. et al. (2016) Hair follicle aging is driven by transepidermal elimination of stem cells via COL17A1 proteolysis. Science 351, aad4395 10.1126/science.aad4395 [DOI] [PubMed] [Google Scholar]
- 95.Liu N., Matsumura H., Kato T., Ichinose S., Takada A., Namiki T.. et al. (2019) Stem cell competition orchestrates skin homeostasis and ageing. Nature 568, 344–350 10.1038/s41586-019-1085-7 [DOI] [PubMed] [Google Scholar]
- 96.Has C., Nyström A., Saeidian A.H., Bruckner-Tuderman L. and Uitto J. (2018) Epidermolysis bullosa: Molecular pathology of connective tissue components in the cutaneous basement membrane zone. Matrix Biol. 71–72, 313–329 10.1016/j.matbio.2018.04.001 [DOI] [PubMed] [Google Scholar]
- 97.Fine J.-D., Bruckner-Tuderman L., Eady R.A.J., Bauer E.A., Bauer J.W., Has C.. et al. (2014) Inherited epidermolysis bullosa: Updated recommendations on diagnosis and classification. J. Am. Acad. Dermatol. 70, 1103–1126 10.1016/j.jaad.2014.01.903 [DOI] [PubMed] [Google Scholar]
- 98.Tasanen K., Floeth M., Schumann H. and Bruckner-Tuderman L. (2000) Hemizygosity for a glycine substitution in collagen XVII: unfolding and degradation of the ectodomain. J. Invest. Dermatol. 115, 207–212 10.1046/j.1523-1747.2000.00049.x [DOI] [PubMed] [Google Scholar]
- 99.Jonsson F., Byström B., Davidson A.E., Backman L.J., Kellgren T.G., Tuft S.J.. et al. (2015) Mutations in collagen, type XVII, alpha 1 (COL17A1) cause epithelial recurrent erosion dystrophy (ERED). Hum. Mutat. 36, 463–473 10.1002/humu.22764 [DOI] [PubMed] [Google Scholar]
- 100.Quélard D., Lavergne E., Hendaoui I., Elamaa H., Tiirola U., Heljasvaara R.. et al. (2008) A cryptic frizzled module in cell surface collagen 18 inhibits Wnt/β−Catenin signaling. PLoS ONE 3, e1878 10.1371/journal.pone.0001878 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Duncan M.B., Yang C., Tanjore H., Boyle P.M., Keskin D., Sugimoto H.. et al. (2013) Type XVIII collagen is essential for survival during acute liver injury in mice. Dis. Models Mech. 6, 942–951 10.1242/dmm.011577 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Aikio M., Elamaa H., Vicente D., Izzi V., Kaur I., Seppinen L.. et al. (2014) Specific collagen XVIII isoforms promote adipose tissue accrual via mechanisms determining adipocyte number and affect fat deposition. Proc. Natl. Acad. Sci. U.S.A. 111, E3043–E3052 10.1073/pnas.1405879111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Nguyen T.M.B., Subramanian I.V., Xiao X., Ghosh G., Nguyen P., Kelekar A.. et al. (2009) Endostatin induces autophagy in endothelial cells by modulating Beclin 1 and β-catenin levels. J. Cell. Mol. Med. 13, 3687–3698 10.1111/j.1582-4934.2009.00722.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Hamano Y., Okude T., Shirai R., Sato I., Kimura R., Ogawa M.. et al. (2010) Lack of collagen XVIII/endostatin exacerbates immune-mediated glomerulonephritis. J. Am. Soc. Nephrol. 21, 1445–1455 10.1681/ASN.2009050492 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Yamaguchi Y., Takihara T., Chambers R.A., Veraldi K.L., Larregina A.T. and Feghali-Bostwick C.A. (2012) A peptide derived from endostatin ameliorates organ fibrosis. Sci. Transl. Med. 4, 136ra171 10.1126/scitranslmed.3003421 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Kivinen N., Felszeghy S., Kinnunen A.I., Setälä N., Aikio M., Kinnunen K.. et al. (2016) Absence of collagen XVIII in mice causes age-related insufficiency in retinal pigment epithelium proteostasis. Biogerontology 17, 749–761 10.1007/s10522-016-9647-7 [DOI] [PubMed] [Google Scholar]
- 107.Mutolo M.J., Morris K.J., Leir S.-H., Caffrey T.C., Lewandowska M.A., Hollingsworth M.A.. et al. (2012) Tumor suppression by collagen XV is independent of the restin domain. Matrix Biol. 31, 285–289 10.1016/j.matbio.2012.03.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Myers J.C., Amenta P.S., Dion A.S., Sciancalepore J.P., Nagaswami C., Weisel J.W.. et al. (2007) The molecular structure of human tissue type XV presents a unique conformation among the collagens. Biochem. J. 404, 535–544 10.1042/BJ20070201 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Rasi K., Hurskainen M., Kallio M., Stavén S., Sormunen R., Heape A.M.. et al. (2010) Lack of Collagen XV impairs peripheral nerve maturation and, when combined with laminin-411 deficiency, leads to basement membrane abnormalities and sensorimotor dysfunction. J. Neurosci. 30, 14490–14501 10.1523/JNEUROSCI.2644-10.2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Rasi K., Piuhola J., Czabanka M., Sormunen R., Ilves M., Leskinen H.. et al. (2010) Collagen XV Is necessary for modeling of the extracellular matrix and its deficiency predisposes to cardiomyopathy. Circ. Res. 107, 1241–1252 10.1161/CIRCRESAHA.110.222133 [DOI] [PubMed] [Google Scholar]
- 111.Landis B.J., Schubert J.A., Lai D., Jegga A.G., Shikany A.R., Foroud T.. et al. (2017) Exome sequencing identifies candidate genetic modifiers of syndromic and familial thoracic aortic aneurysm severity. J. Cardiovasc. Transl. Res. 10, 423–432 10.1007/s12265-017-9753-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Wiggs J.L., Howell G.R., Linkroum K., Abdrabou W., Hodges E., Braine C.E.. et al. (2013) Variations in COL15A1 and COL18A1 influence age of onset of primary open angle glaucoma. Clin. Genet. 84, 167–174 10.1111/cge.12176 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Duvvari M.R., van de Ven J.P.H., Geerlings M.J., Saksens N.T.M., Bakker B., Henkes A.. et al. (2016) Whole exome sequencing in patients with the cuticular drusen subtype of age-related macular degeneration. PLoS ONE 11, e0152047 10.1371/journal.pone.0152047 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Gross O., Perin L. and Deltas C. (2014) Alport syndrome from bench to bedside: the potential of current treatment beyond RAAS blockade and the horizon of future therapies. Nephrol. Dialysis Transplant. 29, iv124–iv130 10.1093/ndt/gfu028 [DOI] [PubMed] [Google Scholar]
- 115.Webb N.J.A., Lam C., Shahinfar S., Strehlau J., Wells T.G., Gleim G.W.. et al. (2011) Efficacy and safety of losartan in children with Alport syndrome—results from a subgroup analysis of a prospective, randomized, placebo- or amlodipine-controlled trial. Nephrol. Dialysis Transplant. 26, 2521–2526 10.1093/ndt/gfq797 [DOI] [PubMed] [Google Scholar]
- 116.Koepke M.-L., Weber M., Schulze-Lohoff E., Beirowski B., Segerer S. and Gross O. (2007) Nephroprotective effect of the HMG-CoA-reductase inhibitor cerivastatin in a mouse model of progressive renal fibrosis in Alport syndrome. Nephrol. Dialysis Transplant. 22, 1062–1069 10.1093/ndt/gfl810 [DOI] [PubMed] [Google Scholar]
- 117.Gross O., Koepke M.-L., Beirowski B., Schulze-Lohoff E., Segerer S. and Weber M. (2005) Nephroprotection by antifibrotic and anti-inflammatory effects of the vasopeptidase inhibitor AVE7688. Kidney Int. 68, 456–463 10.1111/j.1523-1755.2005.00423.x [DOI] [PubMed] [Google Scholar]
- 118.Gomez I.G., MacKenna D.A., Johnson B.G., Kaimal V., Roach A.M., Ren S.. et al. (2015) Anti–microRNA-21 oligonucleotides prevent Alport nephropathy progression by stimulating metabolic pathways. J. Clin. Invest. 125, 141–156 10.1172/JCI75852 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Gross O., Appel G., Block G., Chin M., Goldsberry A., Inker L.. et al. (2018) SP121A phase 2/3 study of the efficacy and safety of bardoxolone methyl in patients with alport syndrome. Nephrol. Dialysis Transplant. 33, i384–i385 10.1093/ndt/gfy104.SP121 [DOI] [Google Scholar]
- 120.Lin X., Suh J.H., Go G. and Miner J.H. (2014) Feasibility of repairing glomerular basement membrane defects in Alport syndrome. J. Am. Soc. Nephrol. 25, 687–692 10.1681/ASN.2013070798 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Sugimoto H., Mundel T.M., Sund M., Xie L., Cosgrove D. and Kalluri R. (2006) Bone-marrow-derived stem cells repair basement membrane collagen defects and reverse genetic kidney disease. Proc. Natl. Acad. Sci. U.S.A. 103, 7321–7326 10.1073/pnas.0601436103 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Sedrakyan S., Da Sacco S., Milanesi A., Shiri L., Petrosyan A., Varimezova R.. et al. (2012) Injection of amniotic fluid stem cells delays progression of renal fibrosis. J. Am. Soc. Nephrol. 23, 661–673 10.1681/ASN.2011030243 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Castagnaro S., Pellegrini C., Pellegrini M., Chrisam M., Sabatelli P., Toni S.. et al. (2016) Autophagy activation in COL6 myopathic patients by a low-protein-diet pilot trial. Autophagy 12, 2484–2495 10.1080/15548627.2016.1231279 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Chrisam M., Pirozzi M., Castagnaro S., Blaauw B., Polishchuck R., Cecconi F.. et al. (2015) Reactivation of autophagy by spermidine ameliorates the myopathic defects of collagen VI-null mice. Autophagy 11, 2142–2152 10.1080/15548627.2015.1108508 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Merlini L., Angelin A., Tiepolo T., Braghetta P., Sabatelli P., Zamparelli A.. et al. (2008) Cyclosporin A corrects mitochondrial dysfunction and muscle apoptosis in patients with collagen VI myopathies. Proc. Natl. Acad. Sci. U.S.A. 105, 5225–5229 10.1073/pnas.0800962105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Zulian A., Rizzo E., Schiavone M., Palma E., Tagliavini F., Blaauw B.. et al. (2014) NIM811, a cyclophilin inhibitor without immunosuppressive activity, is beneficial in collagen VI congenital muscular dystrophy models. Hum. Mol. Genet. 23, 5353–5363 10.1093/hmg/ddu254 [DOI] [PubMed] [Google Scholar]
- 127.Tiepolo T., Angelin A., Palma E., Sabatelli P., Merlini L., Nicolosi L.. et al. (2009) The cyclophilin inhibitor Debio 025 normalizes mitochondrial function, muscle apoptosis and ultrastructural defects in Col6a1−/− myopathic mice. Br. J. Pharmacol. 157, 1045–1052 10.1111/j.1476-5381.2009.00316.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Gamberi T., Magherini F., Mannelli M., Chrisam M., Cescon M., Castagnaro S.. et al. (2019) Role of adiponectin in the metabolism of skeletal muscles in collagen VI–related myopathies. J. Mol. Med. 97, 793–801 10.1007/s00109-019-01766-0 [DOI] [PubMed] [Google Scholar]
- 129.Alexeev V., Arita M., Donahue A., Bonaldo P., Chu M.-L. and Igoucheva O. (2014) Human adipose-derived stem cell transplantation as a potential therapy for collagen VI-related congenital muscular dystrophy. Stem Cell Res. Ther. 5, 21 10.1186/scrt411 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Gualandi F., Manzati E., Sabatelli P., Passarelli C., Bovolenta M., Pellegrini C.. et al. (2012) Antisense-induced messenger depletion corrects a COL6A2 dominant mutation in ullrich myopathy. Hum. Gene Ther. 23, 1313–1318 10.1089/hum.2012.109 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Bolduc V., Zou Y., Ko D. and Bönnemann C.G. (2014) siRNA-mediated allele-specific silencing of a COL6A3 mutation in a cellular model of dominant ullrich muscular dystrophy. Mol. Ther. Nucleic Acids 3, e147, 10.1038/mtna.2013.74 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Noguchi S., Ogawa M., Kawahara G., Malicdan M.C. and Nishino I. (2014) Allele-specific gene silencing of mutant mRNA restores cellular function in ullrich congenital muscular dystrophy fibroblasts. Mol. Ther. Nucleic Acids 3, e171 10.1038/mtna.2014.22 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Marrosu E., Ala P., Muntoni F. and Zhou H. (2017) Gapmer antisense oligonucleotides suppress the mutant allele of COL6A3 and restore functional protein in ullrich muscular dystrophy. Mol. Ther. Nucleic Acids 8, 416–427 10.1016/j.omtn.2017.07.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Bolduc V., Foley A.R., Solomon-Degefa H., Sarathy A., Donkervoort S., Hu Y.. et al. (2019) A recurrent COL6A1 pseudoexon insertion causes muscular dystrophy and is effectively targeted by splice-correction therapies. JCI Insight 4, e124403, 10.1172/jci.insight.124403 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Venugopal S.S., Yan W., Frew J.W., Cohn H.I., Rhodes L.M., Tran K.. et al. (2013) A phase II randomized vehicle-controlled trial of intradermal allogeneic fibroblasts for recessive dystrophic epidermolysis bullosa. J. Am. Acad. Dermatol. 69, 898–908.e897 10.1016/j.jaad.2013.08.014 [DOI] [PubMed] [Google Scholar]
- 136.Gostyński A., Pasmooij A.M.G. and Jonkman M.F. (2014) Successful therapeutic transplantation of revertant skin in epidermolysis bullosa. J. Am. Acad. Dermatol. 70, 98–101 10.1016/j.jaad.2013.08.052 [DOI] [PubMed] [Google Scholar]
- 137.Sebastiano V., Zhen H.H., Haddad B., Bashkirova E., Melo S.P., Wang P.. et al. (2014) Human COL7A1-corrected induced pluripotent stem cells for the treatment of recessive dystrophic epidermolysis bullosa. Sci. Transl. Med. 6, 264ra163 10.1126/scitranslmed.3009540 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Petrof G., Lwin S.M., Martinez-Queipo M., Abdul-Wahab A., Tso S., Mellerio J.E.. et al. (2015) Potential of systemic allogeneic mesenchymal stromal cell therapy for children with recessive dystrophic epidermolysis bullosa. J. Invest. Dermatol. 135, 2319–2321 10.1038/jid.2015.158 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Liao Y., Ivanova L., Sivalenka R., Plumer T., Zhu H., Zhang X.. et al. (2018) Efficacy of human placental-derived stem cells in collagen VII knockout (recessive dystrophic epidermolysis bullosa) animal model. Stem Cells Transl. Med. 7, 530–542 10.1002/sctm.17-0182 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Bornert O., Kühl T., Bremer J., van den Akker P.C., Pasmooij A.M.G. and Nyström A. (2016) Analysis of the functional consequences of targeted exon deletion in COL7A1 reveals prospects for dystrophic epidermolysis bullosa therapy. Mol. Ther. 24, 1302–1311 10.1038/mt.2016.92 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Osborn M.J., Starker C.G., McElroy A.N., Webber B.R., Riddle M.J., Xia L.. et al. (2013) TALEN-based gene correction for epidermolysis bullosa. Mol. Ther. 21, 1151–1159 10.1038/mt.2013.56 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Bonafont J., Mencía Á., García M., Torres R., Rodríguez S., Carretero M.. et al. (2019) Clinically relevant correction of recessive dystrophic epidermolysis bullosa by dual sgRNA CRISPR/Cas9-mediated gene editing. Mol. Ther. 27, 986–998 10.1016/j.ymthe.2019.03.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Murauer E.M., Gache Y., Gratz I.K., Klausegger A., Muss W., Gruber C.. et al. (2011) Functional correction of type VII collagen expression in dystrophic epidermolysis bullosa. J. Invest. Dermatol. 131, 74–83 10.1038/jid.2010.249 [DOI] [PubMed] [Google Scholar]
- 144.Zhou D., Gao Y., Aied A., Cutlar L., Igoucheva O., Newland B.. et al. (2016) Highly branched poly(β-amino ester)s for skin gene therapy. J. Control. Release 244, 336–346 10.1016/j.jconrel.2016.06.014 [DOI] [PubMed] [Google Scholar]
- 145.Siprashvili Z., Nguyen N.T., Gorell E.S., Loutit K., Khuu P., Furukawa L.K.. et al. (2016) Safety and wound outcomes following genetically corrected autologous epidermal grafts in patients with recessive dystrophic epidermolysis bullosa. JAMA 316, 1808–1817 10.1001/jama.2016.15588 [DOI] [PubMed] [Google Scholar]
- 146.Turczynski S., Titeux M., Tonasso L., Décha A., Ishida-Yamamoto A. and Hovnanian A. (2016) Targeted exon skipping restores type VII collagen expression and anchoring fibril formation in an in vivo RDEB model. J. Invest. Dermatol. 136, 2387–2395 10.1016/j.jid.2016.07.029 [DOI] [PubMed] [Google Scholar]
- 147.Dabrowski M., Bukowy-Bieryllo Z. and Zietkiewicz E. (2018) Advances in therapeutic use of a drug-stimulated translational readthrough of premature termination codons. Mol. Med. 24, 25 10.1186/s10020-018-0024-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Nyström A., Thriene K., Mittapalli V., Kern J.S., Kiritsi D., Dengjel J.. et al. (2015) Losartan ameliorates dystrophic epidermolysis bullosa and uncovers new disease mechanisms. EMBO Mol. Med. 7, 1211–1228 10.15252/emmm.201505061 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Pendaries V., Gasc G., Titeux M., Tonasso L., Mejía J.E. and Hovnanian A. (2012) siRNA-mediated allele-specific inhibition of mutant type VII collagen in dominant dystrophic epidermolysis bullosa. J. Invest. Dermatol. 132, 1741–1743 10.1038/jid.2012.11 [DOI] [PubMed] [Google Scholar]
- 150.Shinkuma S., Guo Z. and Christiano A.M. (2016) Site-specific genome editing for correction of induced pluripotent stem cells derived from dominant dystrophic epidermolysis bullosa. Proc. Natl. Acad. Sci. U.S.A. 113, 5676–5681 10.1073/pnas.1512028113 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Remington J., Wang X., Hou Y., Zhou H., Burnett J., Muirhead T.. et al. (2009) Injection of recombinant human type VII collagen corrects the disease phenotype in a murine model of dystrophic epidermolysis bullosa. Mol. Ther. 17, 26–33 10.1038/mt.2008.234 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Woodley D.T., Wang X., Amir M., Hwang B., Remington J., Hou Y.. et al. (2013) Intravenously injected recombinant human type VII collagen homes to skin wounds and restores skin integrity of dystrophic epidermolysis bullosa. J. Invest. Dermatol. 133, 1910–1913 10.1038/jid.2013.10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Hirsch T., Rothoeft T., Teig N., Bauer J.W., Pellegrini G., De Rosa L.. et al. (2017) Regeneration of the entire human epidermis using transgenic stem cells. Nature 551, 327 10.1038/nature24487 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Blau H.M. and Daley G.Q. (2019) Stem cells in the treatment of disease. N. Engl. J. Med. 380, 1748–1760 10.1056/NEJMra1716145 [DOI] [PubMed] [Google Scholar]
- 155.Lwin S.M., Syed F., Di W.-L., Kadiyirire T., Liu L., Guy A.. et al. (2019) Safety and early efficacy outcomes for lentiviral fibroblast gene therapy in recessive dystrophic epidermolysis bullosa. JCI Insight 4, 10.1172/jci.insight.126243 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Mullan L.A., Mularczyk E.J., Kung L.H., Forouhan M., Wragg J.M.A., Goodacre R.. et al. (2017) Increased intracellular proteolysis reduces disease severity in an ER stress–associated dwarfism. J. Clin. Invest. 127, 3861–3865 10.1172/JCI93094 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Richter H., Satz A.L., Bedoucha M., Buettelmann B., Petersen A.C., Harmeier A.. et al. (2019) DNA-encoded library-derived DDR1 inhibitor prevents fibrosis and renal function loss in a genetic mouse model of Alport syndrome. ACS Chem. Biol. 14, 37–49 10.1021/acschembio.8b00866 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Baigent C. and Lennon R. (2018) Should We Increase GFR with Bardoxolone in Alport syndrome? J. Am. Soc. Nephrol. 29, 357–359 10.1681/ASN.2017101062 [DOI] [PMC free article] [PubMed] [Google Scholar]