Abstract
Stimulation of the bag cell neurons of Aplysiaactivates several biochemical pathways, including protein kinase C (PKC), and alters their excitability for many hours. After an ∼30 min afterdischarge, these neurons enter an ∼18 hr inhibited state during which additional stimulation fails to evoke discharges. In vivo, this refractory period limits the frequency of reproductive behaviors associated with egg laying. We have now examined the role of Ca2+-activated K+(BK) currents in the refractory period. Outward currents gated by both intracellular Ca2+ and depolarization, with pharmacological characteristics of BK currents, were recorded in isolated bag cell neurons. These currents were enhanced by the BK channel activators phloretin and 1,3-dihydro-1-[2-hydroxy-5-(trifluoro-methyl)phenyl]-5-trifluoromethyl-2H-benzimidazol-2-one and inhibited by the BK blocker paxilline. The BK component of K+ current was enhanced by 12-O-tetradecanoyl-phorbol-13-acetate, an activator of PKC, and this effect was blocked by sphinganine and PKC19–36, inhibitors of PKC in bag cell neurons. To test whether the BK current is altered during the refractory period, intact clusters were stimulated to afterdischarge, and neurons were isolated after the clusters had entered the refractory period. Compared with unstimulated cells, current density was almost doubled in refractory neurons. This increase in current was inhibited by preincubating clusters in sphinganine. Treatment of refractory clusters with paxilline significantly restored the ability of stimulation to evoke afterdischarges. Conversely, application of phloretin to previously unstimulated clusters inhibited the onset of afterdischarges. These results indicate that a prolonged increase in BK channel activity contributes to the prolonged refractory period of the bag cell neurons.
Keywords: mollusk, BK channels, refractory state, excitability, afterdischarge, TPA
Large-conductance Ca2+-activated K+ channels, also termed BK channels or Maxi-K channels, play an important role in the regulation of neuronal excitability. In addition to their large unitary conductance, the essential feature that distinguishes BK channels from other types of potassium channels, including the family of small-conductance Ca2+-activated K+ channels, is that they require depolarization and elevated cytosolic Ca2+levels to open at physiological membrane potentials (Latorre et al., 1989). This allows the electrical activity of a cell to be coupled to changes in intracellular Ca2+. In some instances, BK channels colocalize with voltage-gated Ca2+ channels, suggesting that they function as feedback regulators of intracellular Ca2+ concentration by hyperpolarizing the plasma membrane after Ca2+ entry (Robitaille and Charlton, 1992). In some neurons, this may allow BK channels to regulate the amount of neurotransmitter released from presynaptic nerve terminals by modulating the duration of the presynaptic action potential (Robitaille et al., 1993). Additionally, these channels may also contribute to firing patterns and the afterhyperpolarization of neurons and other excitable cells (MacDermott and Weight, 1982; Lancaster and Nicoll, 1987; Lancaster et al., 1991;Lovell and McCobb, 2001).
The activity of BK channels is known to be regulated by a variety of protein kinases and other messengers (Chung et al., 1991; Critz and Byrne, 1992; Sansom et al., 1997; Nara et al., 2000). Nevertheless, in only a few cases is the physiological significance of such modulation fully understood. In this study, we have analyzed their role in the peptidergic bag cell neurons of the marine mollusk Aplysia. These neurons control egg-laying behavior and have been used extensively as a model to investigate prolonged changes in excitability and neuropeptide secretion. Bag cell neurons form two homogeneous, electrotonically coupled clusters of 200–400 cells each at the rostral end of the abdominal ganglion (Kupfermann and Kandel, 1970; Conn and Kaczmarek, 1989). On electrical or hormonal stimulation, these normally silent neurons can be triggered to undergo an afterdischarge, a period of repetitive firing lasting ∼30 min that results in the secretion of egg-laying hormone. After the afterdischarge, the bag cell neurons enter an ∼18 hr period of relative inexcitability termed the refractory period (Kupfermann and Kandel, 1970; Kaczmarek and Kauer, 1983). During this refractory state, additional stimulation fails to trigger normal afterdischarges, although intense stimulation can sometimes cause lower-frequency discharges of short duration. The threshold for action potential generation is also increased in the refractory state, such that action potentials originating at the source of stimulation fail to propagate throughout the entire neuron (Dudek and Blankenship, 1977; Kaczmarek et al., 1978). Because the egg-laying behaviors triggered by the afterdischarge endure for several hours, the refractory period is believed to limit the frequency of these behaviors and to allow them to proceed to completion once they have been initiated. Our current findings suggest that a very prolonged increase in BK channel activity contributes to the change in the electrical properties of the bag cell neurons during the refractory period.
MATERIALS AND METHODS
Animals and cell culture. Adult Aplysia californica weighing 125–175 gm were obtained from Marine Specimens Unlimited (San Francisco, CA) or Marinus Inc. (Long Beach, CA). Animals were housed in an ∼400 l aquarium containing continuously circulating, aerated Instant Ocean (Aquarium Systems) at 14°C on a 12 hr light/dark cycle and fed romaine lettuce three times a week.
Primary cultures of isolated bag cell neurons were prepared as described by Kaczmarek et al. (1979). Animals were anesthetized by an injection of isotonic MgCl2 (50% of body weight); the abdominal ganglion was removed and incubated for 18 hr at 18–20°C in neutral protease (13.33 mg/ml; 165859; Roche Molecular Biochemicals, Indianapolis, IN) dissolved in normal artificial seawater (nASW) (containing, in mm: 460 NaCl, 10.4 KCl, CaCl2, 55 Mg Cl2, and 15 HEPES, plus 100 U/ml penicillin and 0.1 mg/ml streptomycin), pH 7.8. The ganglion was then transferred to fresh nASW, and the bag cell neuron clusters were dissected from their surrounding connective tissue. Using a fire-polished Pasteur pipette and gentle trituration, bag cell neurons were dispersed in nASW onto 35 × 10 mm polystyrene tissue culture dishes (25000; Corning, Corning, NY). Cultures were maintained in nASW for 1–3 d in a 14°C incubator. Single isolated bag cell neurons, which had no physical contact with adjacent neurons, were selected for whole-cell recording.
Whole-cell voltage clamp.Ca2+-activated K+ currents were measured at room temperature (18–20°C) using an EPC-8 amplifier (List Electronics) and the tight-seal, whole-cell method. Microelectrodes were pulled from 1.5-mm-external diameter borosilicate glass capillaries (TW 150 F-4;World Precision Instruments) and had a resistance of 0.9–1.2 MΩ when filled with intracellular saline. Intracellular saline contained (in mm): 500 K+ asparate, 70 KCl, 1.25 MgCl2, 10 HEPES, 11 glucose, 5 EGTA, 10 glutathione, 5 ATP (disodium salt), and 0.1 GTP (disodium salt), pH 7.3, with KOH. The free Ca2+ concentration of the intracellular saline was set at either 0 or 1 μmby adding the appropriate amount of CaCl2. The free Ca2+ concentration was calculated using the Ca Buffer program (courtesy of Dr. L. Schlichter, University of Toronto, Toronto, Ontario, Canada). The extracellular solution was designed to eliminate both contaminating Na+ currents and the activation of K+ currents through Ca2+ entry and contained (in mm): 471 N-methyld-glutamine, 55 MgCl2, 15 HEPES, 2 EGTA, and 10.4 KCl, pH 7.8. Pipette junction potentials were nulled immediately before seal formation. Pipette and neuronal capacitive currents were canceled, and the series resistance (range, 3–5 MΩ) was compensated 80–90% and monitored throughout the experiment. Current was filtered at 1 kHz with the EPC-8 built-in Bessel filter and was acquired using an IBM-compatible personal computer, a Digidata 1200 analog-to-digital converter (Axon Instruments), and pClamp version 6.0.2 software (Axon Instruments). The holding potential was set at –40 mV to reduce, as much as possible, contamination by both IA(A-current) and inactivating delayed rectifier (IK2/AplysiaShab) K+ currents (DeRiemer et al., 1985;Strong and Kaczmarek, 1986; Quattrocki et al., 1994), K+ currents were evoked by a series of 200 msec test pulses from −40 to + 60 mV. Leak subtraction was performed on-line using a P/4 protocol from −40 mV with subpulses of opposite polarity and an intersubpulse interval of 500 msec and 4 sec between actual test pulses. No evidence of changes in holding current or run-up and run-down of the voltage-activated outward current was detected throughout the time course of dialysis with 1 μm Ca2+ compared with dialysis with 0 Ca2+. This would suggest that under these conditions and over the time course of the recording period, 1 μmCa2+ did not cause significant changes to membrane currents of the bag cell neurons.
Extracellular recording. For extracellular recording, abdominal ganglia were placed in a recording chamber at 14°C. A wide-bore, fire-polished glass suction electrode placed at the distal end of one connective, and a recording suction electrode was placed at the rostral end of the corresponding bag cell neuron cluster (Kaczmarek et al., 1978). Stimulating current pulses trains (20 V pulses of 2.5 msec duration at 6 Hz for 10–20 sec) were delivered with a Grass S88 stimulator and isolation unit, and voltage was recorded using a Warner DP-301 differential amplifier. Voltage was monitored using an oscilloscope and a permanent record made on a chart recorder. The compound action potentials of the bag cell neurons could be uniquely identified by their large amplitude and width (Kaczmarek and Strumwasser, 1981). The last compound action potential recorded was taken as the end of the afterdischarge. Within 15 min of the end of a first afterdischarge, a similar pulse train was applied one or two times to ensure that the cells had entered the refractory period. At this time, afterdischarge duration was scored as 0. To test the effect of the BK blocker paxilline, a 10 μm concentration of the drug was added to the bath once the cluster was determined to be refractory, and the cluster was then stimulated again within 10 min. For the BK activator phloretin, we pretreated clusters with 100 μm phloretin for 30 min and then tested the ability of the cluster to generate an afterdischarge by stimulating both in the continued presence of phloretin and after washout of the drug.
Drugs. ATP (disodium salt), GTP (disodium salt), iberiotoxin (I-2141), 1,3-dihydro-1-[2-hydroxy-5-(trifluoro-methyl)phenyl]-5-trifluoromethyl-2H-benzimidazol-2-one (NS 1619) (N-170), paxilline (P-2928), penitrem A (P-3053), phloretin (P-7912), 4α-phorbol (P-4888), PKC 19–36(P-8422), sphinganine (D-6908), 12-O-tetradecanoyl-phorbol-13-acetate (TPA; P-8139), and verruculogen (V-7755), were purchased from Sigma (St. Louis, MO). A rapid, single-cell perfusion system, consisting of a micromanipulator-controlled square-barreled glass pipette (∼300 μm bore) positioned close (300–500 μm) to the soma and connected to a series of gravity-driven reservoirs, was used to deliver all extracellularly applied drugs during whole-cell recording.
Analysis. Data are presented as the mean and SEM. Statistical analysis was performed by using Instat version 2.01 (GraphPad Software Inc., San Diego, CA). Student's t test was used to test for differences between two means. Data were considered significantly different at p < 0.05
RESULTS
Properties of Ca2+-activated K+ currents in isolated bag cell neurons
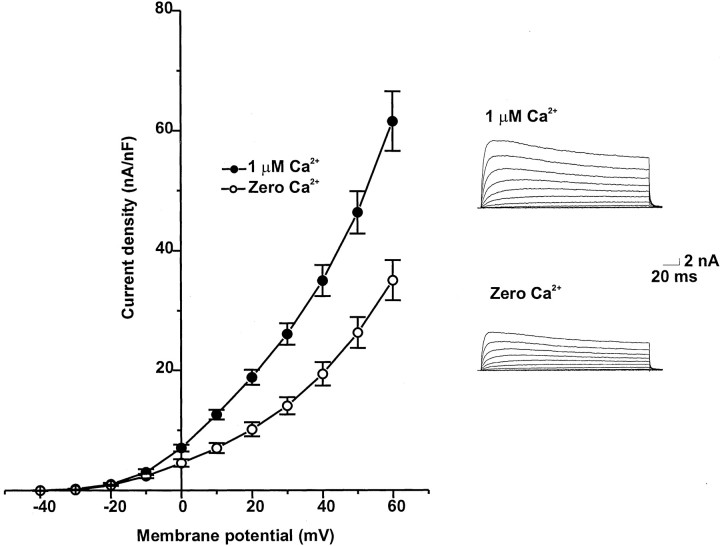
Figure 1 shows the mean current–voltage relationships for K+currents recorded from isolated bag cell neurons using the tight-seal, whole-cell configuration. K+ currents were evoked by depolarizing the membrane potential from a holding potential of −40 mV to test potentials between −40 and +60 mV in 10 mV increments. Steady-state currents were normalized by cell capacitance. To isolate Ca2+-activated K+ currents, we recorded the K+ currents from cultured bag cell neurons in a Ca2+- and Na+-free extracellular solution. When intracellular Ca2+ was set at 1 μm, large, outwardly rectifying currents were observed. Omission of Ca2+ from the internal solution reduced the outward current, particularly at more positive voltages.
Fig. 1.
Current–voltage relationships for whole-cell K+ currents of isolated bag cell neurons. Currents were recorded with Ca2+-free intracellular solutions or solutions containing 1 μm Ca2+ and evoked by depolarizing the membrane from a holding potential of –40 mV to test potentials from –40 to +60 mV. Peak K+currents were normalized by cell capacitance (n = 9).
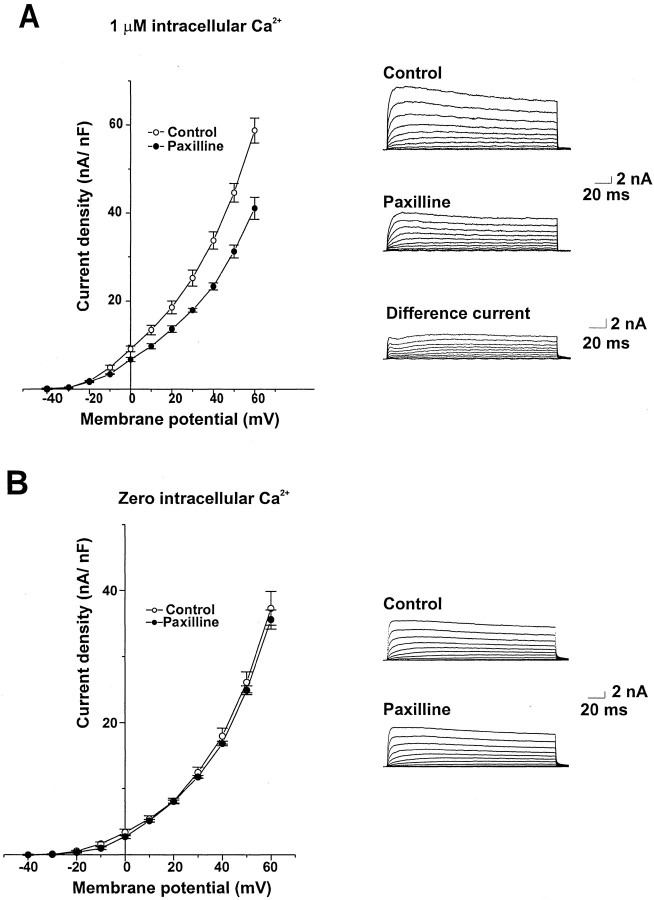
Figure 2 shows the effect of paxilline, a specific blocker of BK currents (Knaus et al., 1994a; Sanchez and McManus, 1996; Strobaek et al., 1996), on isolated bag cell neurons. At 10 μm, paxilline reduced outward currents in bag cell neurons with 1 μm Ca2+ in the intracellular solution but had no effect on outward currents with 0 Ca2+ in the intracellular solution. Figure2A also shows the difference currents obtained by subtracting traces obtained after application of paxilline from the control traces. The kinetics and voltage sensitivity of the difference current match those of BK currents, activating significantly with depolarizations to potentials >0 mV and undergoing little inactivation. It was not possible to establish whether the effect of paxilline was maximal at 10 μm, because higher concentrations had effects on currents recorded in 0 Ca2+ solution.
Fig. 2.
Effect of paxilline on Ca2+-activated K+ currents in bag cell neurons. A, Mean current density–voltage relationships for cells dialyzed with 1 μmCa2+ in the intracellular solution before and after exposure to 10 μm paxilline (n = 5). Also shown are representative current traces and the difference currents obtained by subtracting traces from a paxilline-treated cell from the control traces obtained before paxilline exposure. B, Mean current density–voltage relationships and representative current traces for cells dialyzed with Ca2+-free intracellular solution before and after exposure to 10 μm paxilline (n = 5).
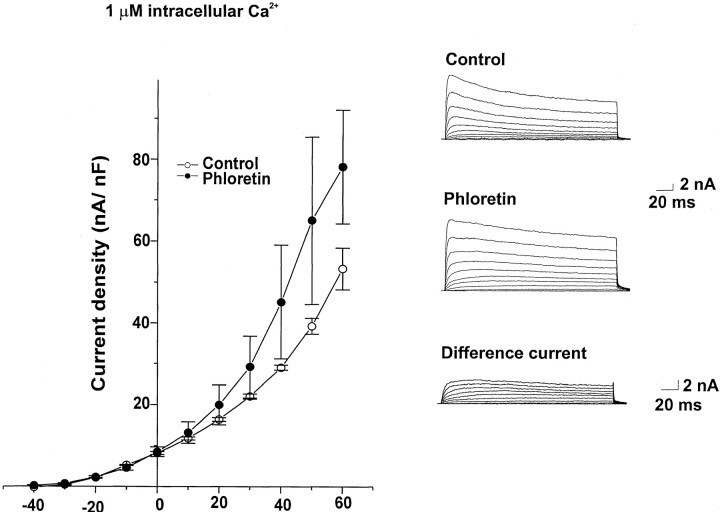
We also tested the effects of other reagents known to influence BK channels on the K+ current of isolated neurons. The BK channel activators phloretin (100 μm;Ransom and Sontheimer, 2001) and NS 1619 (30 μm;Holland et al., 1996) both enhanced the outward currents recorded with 1 μm Ca2+ in the intracellular solution but had no effect on outward currents recorded with a Ca2+-free intracellular solution (Fig.3, Table 1). Figure 3B also shows the difference currents obtained by subtracting control traces from those obtained after application of phloretin. In contrast, the BK channel blockers iberiotoxin (Joiner et al., 1998), penitrem A (Aoki and Baraban, 2000), and verruculogen (Aoki and Baraban, 2000) had no effect on outward currents recorded in bag cell neurons with either with 1 μm or 0 Ca2+ in the intracellular solution (Table1).
Fig. 3.
Effect of phloretin on Ca2+-activated K+ currents in bag cell neurons. Mean current density–voltage relationships are shown for cells dialyzed with 1 μm Ca2+ in an intracellular solution before and after exposure to 100 μm phloretin (n = 5). Also shown are representative current traces and the difference currents obtained by subtracting control traces from those obtained after phloretin exposure.
Table 1.
Effect of blockers and activators of BK channels on Ca2+-activated K+ currents in bag cell neurons
| Agent | Change (%) | |
|---|---|---|
| 1 μmCa2+ in intracellular solution | 0 Ca2+in intracellular solution | |
| Blockers | ||
| Paxilline | −30.0 ± 3.3* | −2.1 ± 0.2 |
| Iberiotoxin | −3.5 ± 0.2 | −1.5 ± 0.1 |
| Verruculogen | −5.6 ± 0.3 | −4.4 ± 0.1 |
| Penitrem A | −2.4 ± 0.2 | −2.1 ± 0.05 |
| Activators | ||
| Phloretin | +33.0 ± 4.0* | +2.8 ± 0.4 |
| NS 1619 | +35.0 ± 4.6* | +3.2 ± 0.3 |
Results shown are the means of five experiments for each condition. Currents were evoked by stepping from a holding potential of −40 mV to a test potential of 60 mV and were measured at the end of a 200 msec pulse.
p < 0.001.
Modulation of Ca2+-activated K+ current by protein kinase C
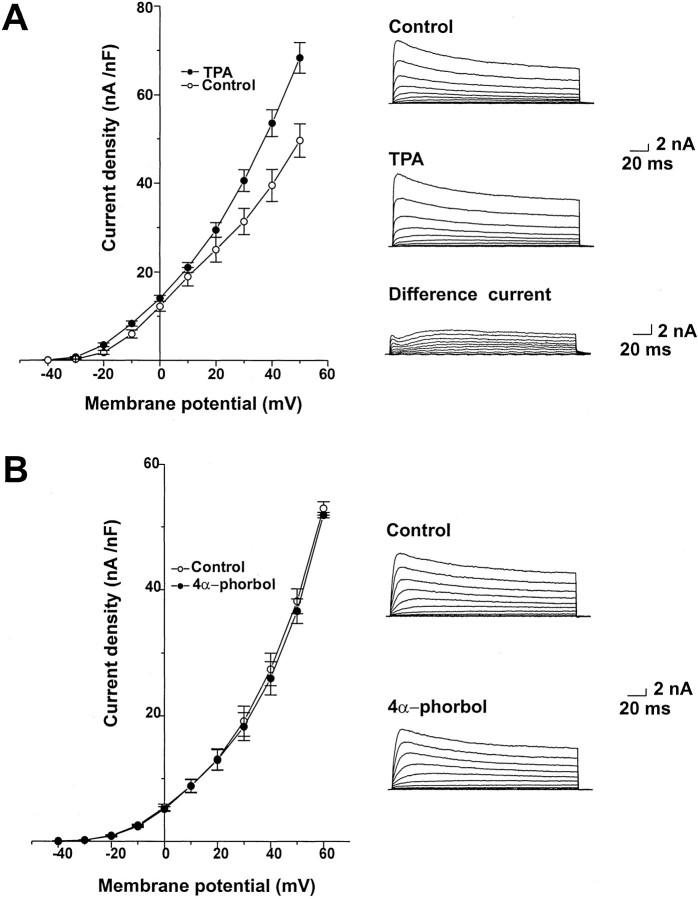
PKC is activated at the onset of afterdischarge and through the early part of the refractory period (Wayne et al., 1999). Activators of PKC produce an approximately twofold increase in the amplitude of the voltage-dependent Ca2+ currents in bag cell neurons but have been shown to have no effect on the purely voltage-dependent K+ currents in these cells (DeRiemer et al., 1985). We therefore tested the action of TPA (100 nm), a phorbol ester activator of PKC, on Ca2+-activated K+ currents in bag cell neurons. TPA produced an increase in the amplitude of the outward current recorded with 1 μm intracellular Ca2+(Fig. 4). The properties of the voltage-dependent difference current, obtained by subtracting control traces from those obtained after application of TPA (Fig.4A), generally match those of the paxilline-sensitive current (Fig. 2A).
Fig. 4.
K+ currents are increased by TPA in bag cell neurons. A, Mean values of peak current density for five cells dialyzed with 1 μmCa2+ before and after treatment with 100 nm TPA. Also shown are representative currenttraces and the difference currents obtained by subtracting control traces from those obtained after TPA exposure.B, Mean values of peak current density and representative traces for cells dialyzed with 1 μm Ca2+ before and after treatment with 4α-phorbol (100 nm; n = 5).
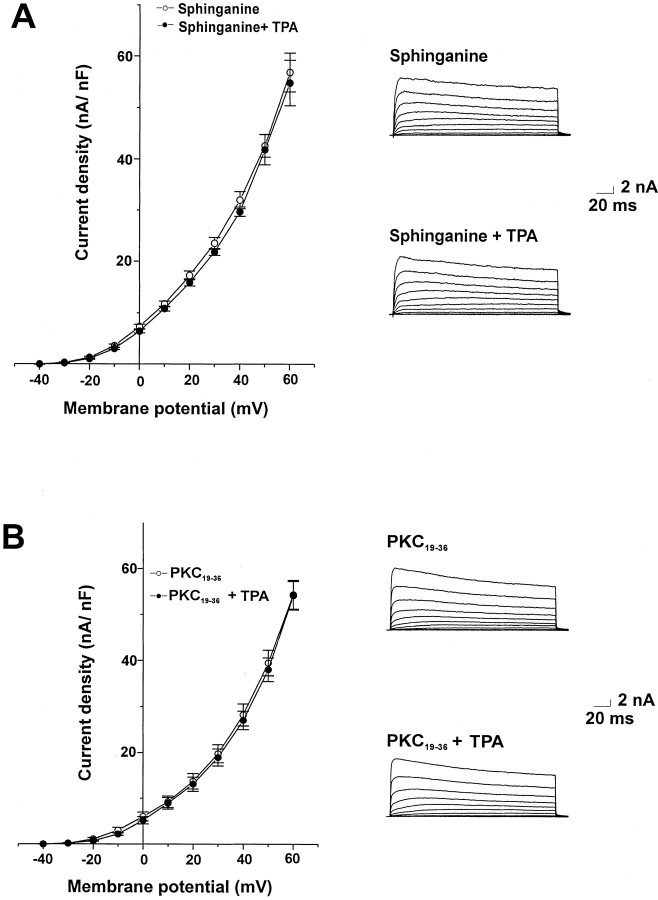
The inactive phorbol ester 4α-phorbol (100 nm), which does not activate PKC, had no significant effect on outward currents in bag cell neurons (Fig. 4B). As an additional test for the role of PKC in the actions of TPA on the outward current, we examined the effect of TPA in the presence of the PKC inhibitors sphinganine and PKC19–36. Sphinganine (10 μm), which has been shown previously to inhibit the action of PKC activators in bag cell neurons (Conn et al., 1989), was added to the recording chamber at least 15 min before voltage-clamp recording. This completely blocked the enhancement of Ca2+-activated K+ currents by 100 nm TPA (Fig.5A). The specific pseudosubstrate PKC inhibitor peptide PKC19–36(10 μm; House and Kemp, 1987) was added to the intracellular solution and allowed to dialyze for 10 min before application of TPA. This also fully blocked the increase in current produced by TPA (Fig. 5B).
Fig. 5.
Inhibitors of PKC prevent the increase in K+ current produced by TPA. Mean values of peak current density and representative current traces are shown for cells dialyzed with 1 μmCa2+ before and after treatment with 100 nm TPA in the presence of sphinganine (10 μm;n = 5; A) and after dialysis with the pseudosubstrate inhibitor PKC19–36 (50 μm; n = 5; B).
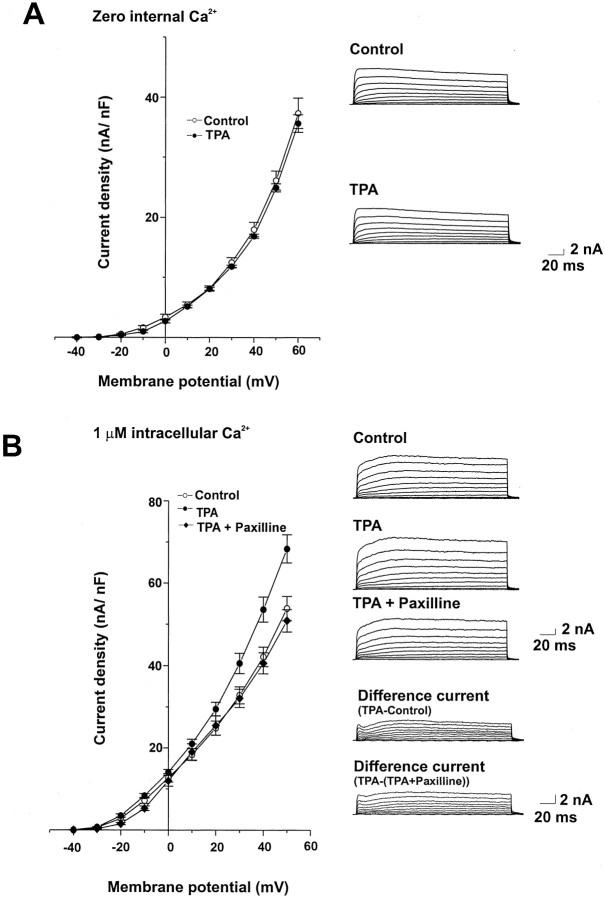
To confirm that activation of PKC influences the Ca2+-activated component of the K+ current, we tested the effect of 100 nm TPA with a Ca2+-free intracellular solution. TPA had no effect on current amplitude under these conditions (Fig.6A). Moreover, paxilline (10 μm) reversed the action of TPA (Fig. 6B). These findings are consistent with previous work demonstrating that TPA does not alter either the delayed rectifier or A current of bag cell neurons (DeRiemer et al., 1985).
Fig. 6.
TPA enhances Ca2+-activated K+ currents in bag cell neurons. A, Mean values of peak current density and representative currenttraces showing the lack of effect of 100 nmTPA in cells dialyzed with a Ca2+-free intracellular solution (n = 5). B, Mean values of peak current density and representative current tracesfor cells dialyzed with 1 μm Ca2+before and after treatment with 100 nm TPA and subsequent application of 10 μm paxilline (n = 5). Also shown are the difference currents.
Ca2+-activated K+ current increases after an afterdischarge
To determine whether Ca2+-activated K+ currents are altered during the refractory period that follows an afterdischarge in the bag cell neurons, abdominal ganglia were dissected and divided into three groups: (1) control unstimulated ganglia, (2) ganglia in which the pleuroabdominal nerve was stimulated to trigger afterdischarge in the bag cell neurons, and (3) ganglia that were incubated with sphinganine (25 μm) for at least 15 min before stimulation of an afterdischarge. Bag cell neurons were then isolated from clusters within these ganglia and maintained at room temperature for 3 hr, a time sufficient for the neurons to adhere firmly to the dish for whole-cell recording but still well within the ∼18 hr period of refractoriness.
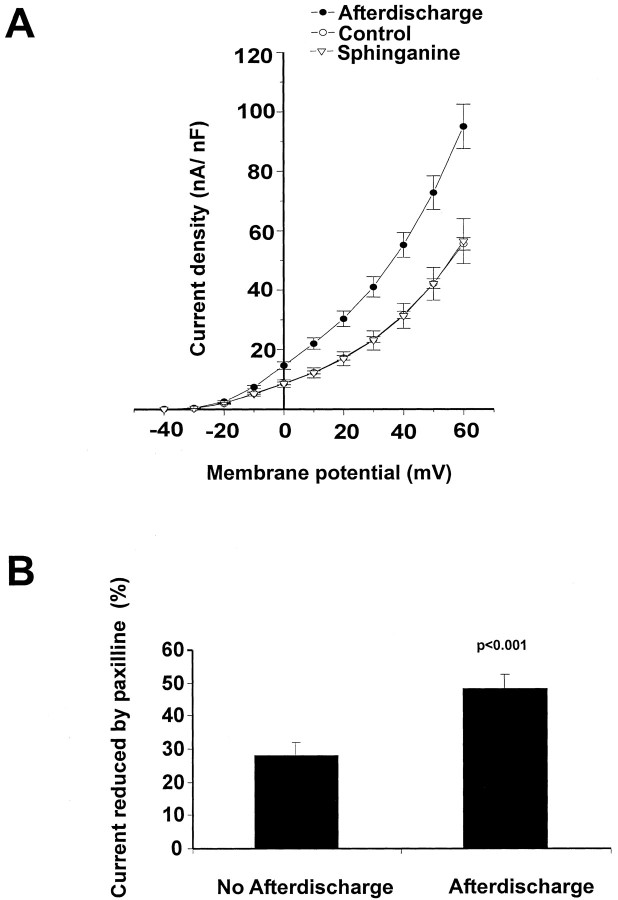
The K+ current density of cells that had undergone an afterdischarge was found to be significantly higher than that of the control unstimulated cells (Fig.7A). Moreover, no increase in current density occurred when bag cell neurons were stimulated in the presence of sphinganine (Fig. 7A). Evidence that the increased current in cells that have entered the refractory state represents an increase in BK current comes from the finding that the BK channel blocker paxilline (10 μm) decreased a significantly greater proportion of the current in stimulated refractory cells (48.4 ± 4.3%), compared with unstimulated controls (27.9 ± 3.9%) (Fig. 7B).
Fig. 7.
K+ currents are enhanced after an afterdischarge in the bag cell neurons. A, Mean current density–voltage relationships for cells isolated from unstimulated clusters (Control, n = 9) and from those that had been stimulated to afterdischarge at least 3 hr before recording (n = 9). Also shown are data for cells stimulated in the presence of 25 μm sphinganine applied 15 min before stimulation (n = 9).B, Comparison of the effect of paxilline (10 μm) on K+ currents recorded in cells isolated from unstimulated clusters (n = 9) and in those that had been stimulated to afterdischarge (n= 9). Paxilline reduced the currents to a significantly greater extent in the cells that had undergone afterdischarge.
Ca2+-activated K+ channels may contribute to prolonged inhibition in bag cell neurons
To investigate the possible contribution of enhanced BK current to the refractory state of bag cell neurons, we tested the actions of the BK current blocker paxilline and the BK activator phloretin on the ability of intact clusters of bag cell neurons within the abdominal ganglion to generate an afterdischarge.
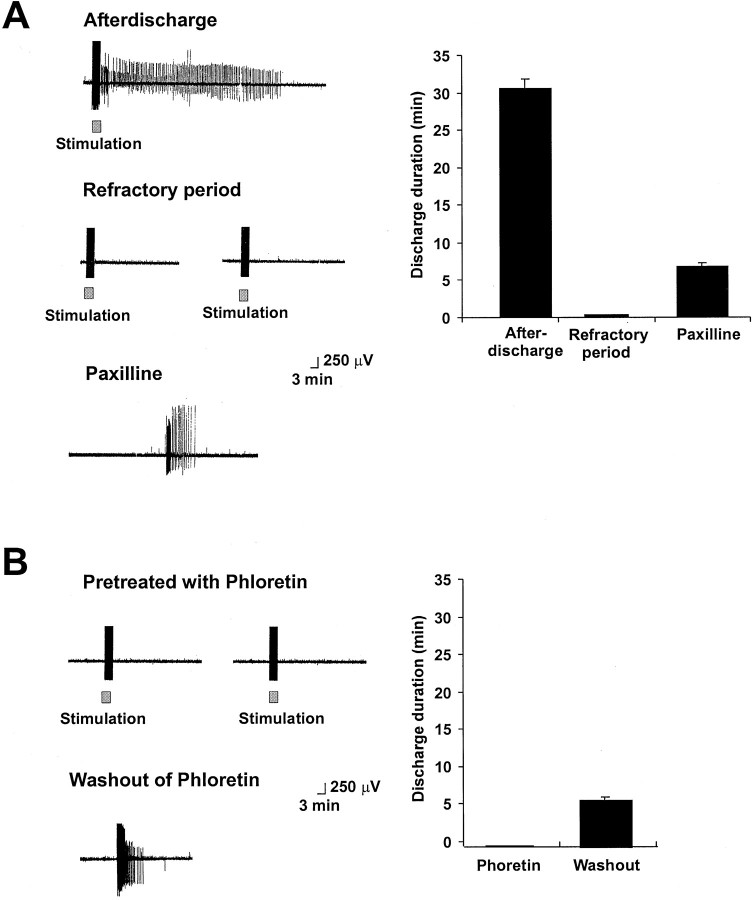
Control afterdischarges were elicited and found to have a mean duration of 30 ± 4.2 min (Fig.8A, left,top traces). Within 30 min of the end of the afterdischarge, repeated stimulation was again applied, verifying that the cells had entered the refractory state and that no additional afterdischarges could be evoked. Partial restoration of the afterdischarge occurred, however, after paxilline (10 μm) was introduced into the bath for 2–10 min. The mean duration of discharges in refractory bag cell clusters treated with paxilline was 6.8 ± 0.49 min. Moreover, in several instances, paxilline restored spontaneous firing of action potentials in bag cell neurons even without additional stimulation of the connective nerve (Fig.8A, left, bottom trace).
Fig. 8.
Inhibitors and activators of BK channels influence the refractory state of bag cell neurons in intact abdominal ganglia.A. Left, Extracellular recordings showing the onset of afterdischarge in response to stimulation of the pleuroabdominal connective nerve (20 V, 2.5 msec, 6 Hz). Subsequent stimulus trains fail to evoke afterdischarge (middle traces). Application of 10 μm paxilline restores spontaneous firing (bottom trace). Right, Histograms showing the mean duration of discharge in previously unstimulated clusters, refractory clusters, and refractory clusters treated with 10 μm paxilline (n = 6).B, The BK activator phloretin produces a refractory-like state in clusters of bag cell neurons. Left, Extracellular recordings showing the lack of response of previously unstimulated bag cell clusters exposed to phloretin (100 μm). When phloretin was washed out of the recording chamber, partial discharges were recovered (bottom trace). Right, Histograms showing the mean duration of discharge in previously unstimulated clusters exposed to 100 μm phloretin and after washout of phloretin (n = 5).
We also investigated the actions of the BK activator phloretin (100 μm) on the ability of previously unstimulated clusters to undergo an afterdischarge. Pretreatment with phloretin for 30 min severely attenuated the ability of stimulation to trigger afterdischarges. After washout of phloretin for 2–10 min, afterdischarges could again be evoked and in some cases began spontaneously (Fig. 8B, left, bottom traces). In the presence of phloretin, the outward current would likely be enhanced, which would prevent the cation channels that produce the depolarization of the afterdischarge (see Discussion) from initiating spikes in the cluster, even if these channels had become activated by stimulation. When phloretin was washed out, however, the previous activation of these channels in some clusters may have provided sufficient residual depolarization to elicit a discharge spontaneously. The duration of such afterdischarges was, however, 5.2 ± 0.7 min, which is less than the mean duration of discharges in control untreated clusters. This finding suggests that some of the mechanisms that induce the refractory state can be activated, at least in part, by stimulation of the bag cell neurons even when afterdischarges are inhibited by phloretin.
DISCUSSION
Modulation of K+ channels plays an important role in stimulus-evoked changes in the excitability of the bag cell neurons. For example, the onset of the afterdischarge is characterized by a marked frequency-dependent increase in the width of action potentials (Kaczmarek et al., 1982) that can be attributed to cumulative inactivation of K+ channels. In contrast, after the onset of the refractory period, the width of induced action potentials is diminished (Kaczmarek et al., 1978; Whim and Kaczmarek, 1998). The onset of the refractory state may be triggered by calcium entry during the afterdischarge (Kaczmarek and Kauer, 1983), and our present findings indicate that an increase in Ca2+-activated K+ current contributes to the properties of the neurons during refractoriness. The existence of the Ca2+-activated K+ current in bag cell neurons has been reported by Kaczmarek and Strumwasser (1984), Strong and Kaczmarek (1986), Nerbonne and Gurney (1987), Fink et al. (1988), and Fieber (2000). Moreover, previous work has found that the amplitude of the Ca2+-activated K+ current is high in juvenileAplysia that are incapable of afterdischarge and decreases throughout development toward adult levels that are characteristic of the mature electrophysiological phenotype (Nick et al., 1996).
The properties of the component of the voltage-dependent outward current that is dependent on intracellular Ca2+ match those expected for BK channels (Jan and Jan, 1997). In isolated cells, the amplitude of this current is increased by stimulation of PKC, an enzyme that is activated during and for a considerable time after an afterdischarge in the intact bag cell neuron cluster (Wayne et al., 1999). The amplitude of the current is also significantly elevated several hours after stimulation of an afterdischarge in intact clusters of bag cell neurons. The mechanisms that generate the refractory period are complex and not fully understood. Nevertheless, it is known that the cells are not only less responsive to both afferent synaptic input (connective stimulation) and chemical stimulation but are also intrinsically less excitable (Kaczmarek and Kauer, 1983; Wilson et al., 1996; Magoski et al., 2000). Our results have shown that bag cell neurons that have entered the prolonged refractory state can be induced to discharge if treated with a BK channel blocker. Conversely, treatment of unstimulated clusters with a BK activator renders them unable to discharge. Under these conditions, it is likely that modulation of other channels, such as nonselective cation channels that provide the depolarizing drive for an afterdischarge (Wilson and Kaczmarek, 1993; Wilson et al., 1996) still occurs but that the increase in the potassium current prevents the occurrence of afterdischarge.
Although BK channels are thought to be encoded by a single gene, theSlo gene, there is considerable heterogeneity in their pharmacological and electrophysiological properties in different cell types. Moreover, more than one isoform can be expressed in a single cell type, and these isoforms may be differentially regulated by protein kinases or have different pharmacological properties. Thus it is possible that more than one isoform is present in the bag cell neurons. For example, BK current in bag cell neurons can be blocked by paxilline, a member of a group of structurally related indole diterpenes that are among the most potent nonpeptidyl blockers of BK currents (Knaus et al., 1994a; Sanchez and McManus, 1996; Strobaek et al., 1996). Nevertheless, although 10 μmpaxilline produced a much greater inhibition of BK current in cells that had undergone afterdischarge than in unstimulated neurons (Fig.7B), this agent was not able to fully suppress the BK current after treatment of isolated cells with an activator of PKC (Fig. 6B). One possible explanation for this discrepancy is that the channels recruited during an afterdischarge represent a different isoform from those activated by PKC in isolated cells.
The BK channel activators phoretin and NS 1619 increase the BK current in bag cell neurons. In contrast, iberiotoxin, penitrem A, and verruculogen, blockers of BK channels in certain cells, had no effect on Ca2+-activated K+ currents in bag cell neurons. It is known that the pharmacological properties of BK currents are substantially altered by the presence of β subunits (Knaus et al., 1994b; Wallner et al., 1999; Wanner et al., 1999; Brenner et al., 2000;Meera et al., 2000; Weiger et al., 2000) or potentially by heteromultimerization of Slo with other α-subunits (Joiner et al., 1998). It is also possible that the pharmacological properties of the BK channels in bag cell neurons are influenced by the high ionic strength of the external medium.
Our present results, as well as previous studies (DeRiemer et al., 1985), have found that activators of protein kinase C have no apparent effect on the purely voltage-dependent K+currents in bag cell neurons. In cells dialyzed with the Ca2+-containing internal medium, however, treatment with the phorbol ester TPA produces an increase in the amplitude of a current whose properties closely match those of the paxilline-sensitive BK current. The fact that the effect of TPA could be blocked by sphinganine and by the specific pseudosubstrate inhibitor peptide PKC19–36 indicates that the enhancement of BK by TPA results from the activation of PKC.
The Slo potassium channel subunit contains multiple consensus phosphorylation sites for protein kinases (Atkinson et al., 1991), and in some cases the physiological effect of phosphorylation at a specific site is known (Levitan, 1999; Tian et al., 2001, Weiger et al., 2002). There are also multiple examples of modulation of BK currents by activators of PKC. For example, activation of PKC increases Ca2+-activated K+ currents in the pleural sensory neurons of Aplysia (Critz and Byrne, 1992). In certain glial cells, exposure to phorbol esters strongly reduces BK channel activity, whereas exposure to the protein kinase inhibitor staurosporine stimulates channel activity (Schopf et al., 1999). In the pituitary GH3 cell line, activation of PKC results in activation of BK channels (Jakab et al., 1997). It is not known, however, whether the actions of PKC in any of these cell types result from direct phosphorylation of the Slo protein or through indirect mechanisms.
The increase in K+ current that follows stimulation of an afterdischarge in bag cell neurons is an unusually long-lasting one, because it was recorded >3 hr after the end of stimulation. This prolonged time course readily falls within the duration of the refractory period, which endures for ∼18 hr. Although the increase in current was blocked by sphinganine, it is not yet known whether the mechanism of the enhancement is the same as that for the acute regulation of the BK current in isolated cells. In addition to acting on K+ currents, activation of PKC produces an increase in the voltage-dependent Ca2+ current in the bag cell neurons (DeRiemer et al., 1985; Strong et al., 1987). The enhancement of the Ca2+ current appears to result from the recruitment to the plasma membrane of α1A (CaV2.1) channels, which are normally localized on intracellular vesicles (White and Kaczmarek, 1997; White et al., 1998). It has been shown in a variety of neurons and other cell types that the activity of BK channels is tightly linked to that of voltage-dependent Ca2+ channels. Evidence for a physical link between these two types of channels has come from patch-clamp studies (Roberts et al., 1990; Robitaille et al., 1993;Marrion and Tavalin, 1998), colocalization at release sites (Knaus et al., 1996), and the finding that that direct specific binding of Ca2+ channel α1 subunits to Slo subunits occurs in transfected cells (Cibulsky et al., 2001). Thus it is possible that, in the bag cell neurons, BK channels are linked to voltage-dependent Ca2+ channels and that activation of PKC promotes the coinsertion of both channel types.
A number of independent properties of the bag cell neurons are altered during the transition from afterdischarge to the refractory state. The propagation of action potentials along neurites is attenuated during the refractory period, and the ability of action potentials to invade branch points is impaired (Dudek and Blankenship, 1977; Kaczmarek et al., 1978). Moreover, the frequency-dependent spike broadening that occurs during stimulation at the onset of afterdischarge may be lost after the cells enter the refractory cells (Whim and Kaczmarek, 1998). Each of these changes is consistent with an increase in K+ current, which would be expected to increase the threshold for action potential generation. Other changes are less readily attributable to an increase in the K+ current alone. For example, the mechanisms that produce release of Ca2+from intracellular stores may be attenuated in refractory neurons (Magoski et al., 2000). Moreover, stimulation of refractory cells fails to produce the prolonged depolarization that maintains the discharge (Kaczmarek and Kauer, 1983). This depolarization has been attributed to the activation of a nonselective cation current (Wilson and Kaczmarek, 1993; Wilson et al., 1996; Magoski et al., 2002). Nevertheless, an increase in the K+ current could also contribute in part to these events by indirect mechanisms. For example, it has been shown that, during stimulation, frequency-dependent spike broadening promotes the normal activation of the subsequent prolonged depolarization presumably by enhancing calcium entry (Whim and Kaczmarek, 1998). Our findings suggest, therefore, that a prolonged increase in the BK current is one of the central events in the transition from afterdischarge to the refractory state.
Footnotes
This research was supported by Human Frontiers Science Program Postdoctoral Fellowship LT-432–96 and Medical Research Council of Canada Postdoctoral Fellowship MFE-31315 (N.S.M.), Canadian Institutes of Health Research Operating Grant MOP-53348 (N.S.M.), and National Institutes of Health Grant NS 18492 (L.K.K.).
Correspondence should be addressed to Leonard K. Kaczmarek, Department of Pharmacology, Yale University School of Medicine, 333 Cedar Street, New Haven, CT 06520. E-mail: leonard.kaczmarek@yale.edu.
REFERENCES
- 1.Aoki T, Baraban SC. Properties of a calcium-activated K+ current on interneurons in the developing rat hippocampus. J Neurophysiol. 2000;83:3453–3461. doi: 10.1152/jn.2000.83.6.3453. [DOI] [PubMed] [Google Scholar]
- 2.Atkinson, Robertson GA, Ganetzky B. A component of calcium-activated potassium channels encoded by the Drosophila slo locus. Science. 1991;253:551–555. doi: 10.1126/science.1857984. [DOI] [PubMed] [Google Scholar]
- 3.Brenner R, Jegla TJ, Wickenden A, Liu Y, Aldrich RW. Cloning and functional characterization of novel large conductance calcium-activated potassium channel beta subunits, hkCNMB3 and hkCNMB4. J Biol Chem. 2000;275:6453–6461. doi: 10.1074/jbc.275.9.6453. [DOI] [PubMed] [Google Scholar]
- 4.Chung SK, Reinhart PH, Martin BH, Brautigan DL, Levitan IB. Protein kinase activity closely associated with a reconstituted calcium-activated potassium channel. Science. 1991;253:560–562. doi: 10.1126/science.1857986. [DOI] [PubMed] [Google Scholar]
- 5.Cibulsky SM, Zhou Y, Daniele LL, Yokoyama CT, Catterall WA, Levitan IB. Association between calcium-activated potassium channels and voltage-gated calcium channels. Soc Neurosci Abstr. 2001;31:139. [Google Scholar]
- 6.Conn PJ, Kaczmarek LK. The bag cell neurons of Aplysia: a model for the study of the molecular mechanisms involved in the control of prolonged animal behaviors. Mol Neurobiol. 1989;3:237–273. doi: 10.1007/BF02740607. [DOI] [PubMed] [Google Scholar]
- 7.Conn PJ, Strong JA, Kaczmarek LK. Inhibitors of protein kinase C prevent enhancement of calcium current and action potentials in peptidergic neurons of Aplysia. J Neurosci. 1989;9:480–487. doi: 10.1523/JNEUROSCI.09-02-00480.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Critz SD, Byrne JH. Modulation of IKCa by phorbol ester-mediated activation of PKC in pleural sensory neurons of Aplysia. J Neurophysiol. 1992;68:1079–1085. doi: 10.1152/jn.1992.68.4.1079. [DOI] [PubMed] [Google Scholar]
- 9.DeRiemer SA, Strong JA, Albert KA, Greengard P, Kaczmarek LK. Enhancement of calcium current in Aplysia neurons by phorbol ester and protein kinase C. Nature. 1985;313:313–316. doi: 10.1038/313313a0. [DOI] [PubMed] [Google Scholar]
- 10.Dudek FE, Blankenship JE. Neuroendocrine cells of Aplysia brasiliana. I. Bag cell action potentials and afterdischarge. J Neurophysiol. 1977;40:1301–1311. doi: 10.1152/jn.1977.40.6.1301. [DOI] [PubMed] [Google Scholar]
- 11.Fieber LA. The development of excitatory capability in Aplysia californica bag cells observed in cohorts. Brain Res Dev Brain Res. 2000;122:47–58. doi: 10.1016/s0165-3806(00)00053-5. [DOI] [PubMed] [Google Scholar]
- 12.Fink LA, Connor JA, Kaczmarek LK. Inositol triphosphate releases intracellularly stored calcium and modulates ion channels in molluscan neurons. J Neurosci. 1988;8:2544–2555. doi: 10.1523/JNEUROSCI.08-07-02544.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Holland M, Langton PD, Standen NB, Boyle JP. Effects of the BKca channel activator, NS1619, on rat cerebral artery smooth muscle. Br J Pharmacol. 1996;117:119–129. doi: 10.1111/j.1476-5381.1996.tb15163.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.House C, Kemp BE. Protein kinase C contains a pseudosubstrate prototope in its regulatory domain. Science. 1987;238:1726–1728. doi: 10.1126/science.3686012. [DOI] [PubMed] [Google Scholar]
- 15.Jakab M, Weiger TM, Hermann A. Ethanol activates maxi Ca2+-activated K+ channels of clonal pituitary (GH3) cells. J Membr Biol. 1997;157:237–245. doi: 10.1007/pl00005895. [DOI] [PubMed] [Google Scholar]
- 16.Jan LY, Jan YN. Ways and means for left shifts in the Maxi-K channel. Proc Natl Acad Sci USA. 1997;94:13383–13385. doi: 10.1073/pnas.94.25.13383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Joiner WJ, Tang MD, Wang L-Y, Dworetzky SI, Boissard CG, Gan L, Gribkoff VK, Kaczmarek LK. Formation of intermediate conductance calcium-activated potassium channels by interaction of Slack and Slo subunits. Nat Neurosci. 1998;1:462–469. doi: 10.1038/2176. [DOI] [PubMed] [Google Scholar]
- 18.Kaczmarek LK, Kauer JA. Calcium entry causes a prolonged refractory period in peptidergic neurons of Aplysia. J Neurosci. 1983;3:2230–2239. doi: 10.1523/JNEUROSCI.03-11-02230.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kaczmarek LK, Strumwasser F. The expression of long lasting afterdischarge by isolated Aplysia bag cell neurons. J Neurosci. 1981;1:626–634. doi: 10.1523/JNEUROSCI.01-06-00626.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kaczmarek LK, Strumwasser F. A voltage clamp analysis of currents underlying cyclic AMP-induced membrane modulation in isolated peptidergic neurons of Aplysia. J Neurophysiol. 1984;52:340–349. doi: 10.1152/jn.1984.52.2.340. [DOI] [PubMed] [Google Scholar]
- 21.Kaczmarek LK, Jennings K, Strumwasser F. Neurotransmitter modulation, phosphodiesterase inhibitor effects and cAMP correlates of afterdischarge in peptidergic neurites. Proc Natl Acad Sci USA. 1978;75:5200–5204. doi: 10.1073/pnas.75.10.5200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kaczmarek LK, Finbow M, Revel JP, Strumwasser F. The morphology and coupling of Aplysia bag cells within the abdominal ganglion and in cell culture. J Neurobiol. 1979;10:535–550. doi: 10.1002/neu.480100604. [DOI] [PubMed] [Google Scholar]
- 23.Kaczmarek LK, Jennings K, Strumwasser F. An early sodium and a late calcium phase in the afterdischarge of peptide secreting neurons of Aplysia. Brain Res. 1982;283:105–115. doi: 10.1016/0006-8993(82)90774-0. [DOI] [PubMed] [Google Scholar]
- 24.Knaus HG, McManus OB, Lee SH, Schmalhofer WA, Garcia-Calvo M, Helms LM, Sanchez M, Giangiacomo K, Reuben JP, Smith AB., III Tremorgenic indole alkaloids potently inhibit smooth muscle high-conductance calcium-activated potassium channels. Biochemistry. 1994a;33:5819–5828. doi: 10.1021/bi00185a021. [DOI] [PubMed] [Google Scholar]
- 25.Knaus HG, Folander K, Garcia-Calvo M, Garia ML, Kaczorowski GJ, Smith M, Swanson R. Primary sequence and immunological characterization of the beta-subunit of the high-conductance Ca2+-activated K+ channel from smooth muscle. J Biol Chem. 1994b;269:17274–17278. [PubMed] [Google Scholar]
- 26.Knaus HG, Schwarzer C, Koch RO, Eberhart A, Kaczorowski GJ, Glossmann H, Wunder F, Pongs O, Garcia ML, Sperk G. Distribution of high-conductance Ca2+-activated K+ channels in rat brain: targeting to axons and nerve terminals. J Neurosci. 1996;16:955–963. doi: 10.1523/JNEUROSCI.16-03-00955.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kupfermann I, Kandel ER. Electrophysiological properties and functional interconnections of two symmetrical neurosecretory clusters (bag cells) in abdominal ganglion of Aplysia. J Neurophysiol. 1970;33:865–876. doi: 10.1152/jn.1970.33.6.865. [DOI] [PubMed] [Google Scholar]
- 28.Lancaster B, Nicoll RA. Properties of two calcium-activated hyperpolarizations in rat hippocampal neurons. J Physiol (Lond) 1987;389:187–203. doi: 10.1113/jphysiol.1987.sp016653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lancaster B, Nicoll RA, Perkel DJ. Calcium activates two types of potassium channels in rat hippocampal neurons in culture. J Neurosci. 1991;11:23–30. doi: 10.1523/JNEUROSCI.11-01-00023.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Latorre R, Oberhauser A, Labarca P, Avarez O. Varieties of calcium-activated potassium channels. Annu Rev Physiol. 1989;51:385–399. doi: 10.1146/annurev.ph.51.030189.002125. [DOI] [PubMed] [Google Scholar]
- 31.Levitan IB. Modulation of ion channels by protein phosphorylation: how the brain works. Adv Second Messenger Phosphoprotein Res. 1999;33:3–22. doi: 10.1016/s1040-7952(99)80003-2. [DOI] [PubMed] [Google Scholar]
- 32.Lovell PV, McCobb DP. Pituitary control of BK potassium channel function and intrinsic firing properties of adrenal chromaffin cells. J Neurosci. 2001;21:3429–3442. doi: 10.1523/JNEUROSCI.21-10-03429.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.MacDermott AB, Weight FF. Action potential repolarization may involve a transient, Ca2+-sensitive outward current in a vertebrate neuron. Nature. 1982;300:185–188. doi: 10.1038/300185a0. [DOI] [PubMed] [Google Scholar]
- 34.Magoski NS, Knox RJ, Kaczmarek LK. Activation of a Ca2+-permeable cation channel produces a prolonged attenuation of intracellular Ca2+ release in Aplysia bag cell neurons. J Physiol (Lond) 2000;522:271–283. doi: 10.1111/j.1469-7793.2000.t01-2-00271.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Magoski NS, Wilson GF, Kaczmarek LK. Protein kinase modulation of a neuronal cation channel requires protein–protein interactions mediated by an Src homology 3 domain. J Neurosci. 2002;22:1–9. doi: 10.1523/JNEUROSCI.22-01-00001.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Marrion NV, Tavalin SJ. Selective activation of Ca2+-activated K+ channels by co-localized Ca2+ channels in hippocampal neurons. Nature. 1998;395:900–905. doi: 10.1038/27674. [DOI] [PubMed] [Google Scholar]
- 37.Meera P, Wallner M, Toro L. A neuronal β subunit (KCNM B4) makes the large conductance, voltage- and Ca2+-activated K+ channel resistant to charybdotoxin and iberiotoxin. Proc Natl Acad Sci USA. 2000;10:5562–5567. doi: 10.1073/pnas.100118597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Nara M, Dhulipala PD, Ji GJ, Kamaasani VR, Wang YX, Matalon S, Kotlikoff MI. Guanylyl cyclase stimulatory coupling to Kca channels. Am J Physiol. 2000;279:C1938–C1945. doi: 10.1152/ajpcell.2000.279.6.C1938. [DOI] [PubMed] [Google Scholar]
- 39.Nerbonne JM, Gurney AM. Blockade of Ca2+ and K+ currents in bag cell neurons of Aplysia californica by dihydropyridine Ca2+ antagonist. J Neurosci. 1987;7:882–893. doi: 10.1523/JNEUROSCI.07-03-00882.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Nick TA, Kaczmarek LK, Carew TJ. Ionic currents underlying developmental regulation of repetitive firing in Aplysia bag cell neurons. J Neurosci. 1996;16:7583–7598. doi: 10.1523/JNEUROSCI.16-23-07583.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Quattrocki EA, Marshall J, Kaczmarek LK. A Shab potassium channel contributes to action potential broadening in peptidergic neurons. Neuron. 1994;12:73–86. doi: 10.1016/0896-6273(94)90153-8. [DOI] [PubMed] [Google Scholar]
- 42.Ransom CB, Sontheimer H. BK channels in human glioma cells. J Neurophysiol. 2001;85:790–803. doi: 10.1152/jn.2001.85.2.790. [DOI] [PubMed] [Google Scholar]
- 43.Roberts WM, Jacobs RA, Hudspeth AJ. Colocalization of ion channels involved in frequently selectivity and synaptic transmission at presynaptic active zones of hair cells. J Neurosci. 1990;10:3664–3684. doi: 10.1523/JNEUROSCI.10-11-03664.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Robitaille R, Charlton MP. Presynaptic calcium signals and transmitter release are modulated by Ca2+-activated K+ channels. J Neurosci. 1992;12:297–305. doi: 10.1523/JNEUROSCI.12-01-00297.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Robitaille R, Garcia ML, Kaczorowski GJ, Charlton MP. Functional colocalization of calcium and calcium-gated potassium channels in control of transmitter release. Neuron. 1993;11:645–655. doi: 10.1016/0896-6273(93)90076-4. [DOI] [PubMed] [Google Scholar]
- 46.Sanchez M, McManus OB. Paxilline inhibition of the alpha-subunit of the high-conductance calcium-activated potassium channel. Eur Pharmacol. 1996;35:963–968. doi: 10.1016/0028-3908(96)00137-2. [DOI] [PubMed] [Google Scholar]
- 47.Sansom SC, Stockand JD, Hall D, Williams B. Regulation of large calcium-activated potassium channels by protein phosphatase 2A. J Biol Chem. 1997;272:9902–9906. doi: 10.1074/jbc.272.15.9902. [DOI] [PubMed] [Google Scholar]
- 48.Schopf S, Bringmann A, Reichenbach A. Protein kinase A and C are opponents in modulating glial Ca2+-activated K+ channels. NeuroReport. 1999;10:1323–1327. doi: 10.1097/00001756-199904260-00031. [DOI] [PubMed] [Google Scholar]
- 49.Strobaek D, Christophersen P, Holm NR, Moldt P, Ahring PK, Johansen TE, Olesen SP. Modulation of the Ca2+-activated K+ channel, hslo, by substituted dihenylurea NS 1608, paxilline and internal Ca2+. Neuropharmacology. 1996;35:903–914. doi: 10.1016/0028-3908(96)00096-2. [DOI] [PubMed] [Google Scholar]
- 50.Strong JA, Kaczmarek LK. Multiple components of delayed potassium current in peptidergic neurons of Aplysia: modulation by an activator of adenylate cyclase. J Neurosci. 1986;6:814–822. doi: 10.1523/JNEUROSCI.06-03-00814.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Strong JA, Fox AP, Tsien RW, Kaczmarek LK. Stimulation of protein kinase C recruits covert calcium channels in Aplysia bag cell neurons. Nature. 1987;325:714–717. doi: 10.1038/325714a0. [DOI] [PubMed] [Google Scholar]
- 52.Tian L, Duncan RR, Hammond MS, Coghill LS, Wen H, Rusinova R, Clark AG, Levitan IB, Shipston MJ. Alternative splicing switches potassium channel sensitivity to protein phosphorylation. J Biol Chem. 2001;276:7717–7720. doi: 10.1074/jbc.C000741200. [DOI] [PubMed] [Google Scholar]
- 53.Wallner M, Meera P, Toro L (1999) Determinant for β-subunit regulation in high-conductance voltage-activated and Ca2+ sensitive K+ channels: an additional transmembrane region at the N-terminus. Proc Natl Acad Sci USA 14922–14927. [DOI] [PMC free article] [PubMed]
- 54.Wanner SG, Koch RO, Kaschak A, Trieb M, Garcia ML, Kaczorowski GJ, Knaus HG. High conductance calcium-activated potassium channel in rat brain: pharmacology distribution and subunit composition. Biochemistry. 1999;38:5392–5400. doi: 10.1021/bi983040c. [DOI] [PubMed] [Google Scholar]
- 55.Wayne NL, Lee W, Kim YJ. Persistent activation of calcium-activated and calcium-independent protein kinase C in response to electrical afterdischarge from peptidergic neurons of Aplysia. Brain Res. 1999;834:211–213. doi: 10.1016/s0006-8993(99)01594-2. [DOI] [PubMed] [Google Scholar]
- 56.Weiger TM, Holmqvist MH, Levitan IB, Clark FT, Sprague S, Huang WJ, Ge P, Wang C, Lawson D, Jurman ME, Glucksmann MA, Silos-Santiago I, DiStefano PS, Curtis R. A novel nervous system subunit that downregulates human large-conductance calcium-dependent potassium channels. J Neurosci. 2000;20:3563–3570. doi: 10.1523/JNEUROSCI.20-10-03563.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Weiger TM, Hermann A, Levitan IB. Modulation of calcium-activated potassium channels. J Comp Physiol [A] 2002;188:79–87. doi: 10.1007/s00359-002-0281-2. [DOI] [PubMed] [Google Scholar]
- 58.Whim MD, Kaczmarek LK. Expression of Kv3.1 in bag cell neurons prevents the induction of a depolarizing afterpotential. J Neurosci. 1998;18:9171–9180. doi: 10.1523/JNEUROSCI.18-22-09171.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.White BH, Kaczmarek LK. Identification of a vesicular pool of calcium channels in the bag cell neurons of Aplysia. J Neurosci. 1997;17:1582–1595. doi: 10.1523/JNEUROSCI.17-05-01582.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.White BH, Nick TA, Carew TJ, Kaczmarek LK. Protein kinase C regulates a vesicular class of calcium channels in the bag cell neurons. J Neurophysiol. 1998;80:2514–2520. doi: 10.1152/jn.1998.80.5.2514. [DOI] [PubMed] [Google Scholar]
- 61.Wilson GF, Kaczmarek LK. Mode-switching of a voltage-gated cation channel is mediated by a protein kinase A-regulated tyrosine phosphatase. Nature. 1993;366:433–438. doi: 10.1038/366433a0. [DOI] [PubMed] [Google Scholar]
- 62.Wilson GF, Richardson FC, Fisher TE, Oliver BM, Kaczmarek LK. Identification and characterization of a Ca2+-sensitive nonspecific cation channel underlying prolonged repetitive firing in Aplysia neurons. J Neurosci. 1996;16:3661–3671. doi: 10.1523/JNEUROSCI.16-11-03661.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]