Abstract
Calcium enters the outer segment of a vertebrate photoreceptor through a cGMP-gated channel and is extruded via a Na/Ca, K exchanger. We have identified another element in mammalian cones that might help to control cytoplasmic calcium. Reverse transcription-PCR performed on isolated photoreceptors identified mRNA for the SII− splice variant of the type I receptor for inositol 1,4,5-triphosphate (IP3), and Western blots showed that the protein also is expressed in outer segments. Immunocytochemistry showed type I IP3 receptor to be abundant in red-sensitive and green-sensitive cones of the trichromatic monkey retina, but it was negative or weakly expressed in blue-sensitive cones and rods. Similarly, the green-sensitive cones expressed the receptor in dichromatic retina (cat, rabbit, and rat), but the blue-sensitive cones did not. Immunostain was localized to disk and plasma membranes on the cytoplasmic face. To restore sensitivity after a light flash, cytoplasmic cGMP must rise to its basal level, and this requires cytoplasmic calcium to fall. Cessation of calcium release via the IP3 receptor might accelerate this fall and thus explain why the cone recovers much faster than the rod. Furthermore, because its own activity of the IP3 receptor depends partly on cytoplasmic calcium, the receptor might control the set point of cytoplasmic calcium and thus affect cone sensitivity.
Keywords: photoreceptor, Ca2+, S cone, M cone, L cone, phospholipase C, monkey
Sensitivity of vertebrate photoreceptors is regulated by cytoplasmic Ca2+([Ca2+]i) (Lamb and Torre, 1990; Koch, 1995; McNaughton, 1995; Koutalos and Yau, 1996) (for review, see Yau, 1994). Ca2+ enters the outer segment via cGMP-gated channels, is buffered by calcium binding proteins, and exits via the Na/Ca, K exchanger (Korenbrot, 1995) (for review, see Schnetkamp, 1995a). The outer segment contains an additional store of Ca2+ within membrane saccules (disks) that resemble smooth endoplasmic reticulum (Liebman, 1974; Fain and Schroder, 1985; Nicol et al., 1987; Schnetkamp and Bownds, 1987). Because the latter releases stored Ca2+ via an inositol 1,4,5-triphosphate (IP3) receptor or a ryanodine receptor (Berridge, 1993; Mikoshiba et al., 1994), so might the disks bear such a receptor and provide an additional source of cytoplasmic Ca2+.
Indeed, a light flash to bovine outer segment membranes releases IP3 (Ghalayini and Anderson, 1984; Hayashi and Amakawa, 1985; Brown et al., 1987), and an IP3 receptor has been identified biochemically (Day et al., 1993). Although an antibody against purified brain IP3 receptor did not bind to outer segments (Peng et al., 1991), the IP3 receptor is encoded by at least four genes, each of which might be spliced into several isoforms (Danoff et al., 1991; Nakagawa et al., 1991; Lin, 1995;Nucifora et al., 1995). Therefore, a negative immunoreaction could result simply from a mismatch between isoform and antibody. Here we show by RT-PCR, Western blot, and immunocytochemistry that photoreceptor outer segments express the SII−splice variant of the type I IP3 receptor on their plasma and disk membranes. The receptor is more abundant in red- and green-sensitive cones than in blue-sensitive cones and rods.
MATERIALS AND METHODS
Tissue sources
Eyes were enucleated under deep anesthesia from adult rat (Sprague Dawley), rabbit (Dutch-Belted), guinea pig (Dunkin Hartley), cat, and monkey (Macaca mulatta). All procedures complied with federal regulations and University of Pennsylvania policies.
Dissociating photoreceptors
Small pieces of rat retina were incubated in oxygenated Hank’s medium (Life Technologies, Gaithersburg, MD) containing 14.4 U/ml papain, 0.1 gm/ml cysteine, and 0.5 mm EDTA for 10 min at 28°C. After the retina was rinsed with papain-free Hank’s medium containing 0.5% bovine serum albumin, the retina was triturated gently with a wide-bore Pasteur pipette. An aliquot of dissociated cell suspension was diluted with Hank’s medium and dropped on a cover glass coated with concanavalin A (Sasaki and Kaneko, 1996). After 30 min, most cells attached to the coated cover glass, which then was washed with Hank’s medium at least five times to remove loose cells. Isolated photoreceptors were identified by their characteristic morphology (see Fig. 1C) and sucked into a patch pipette.
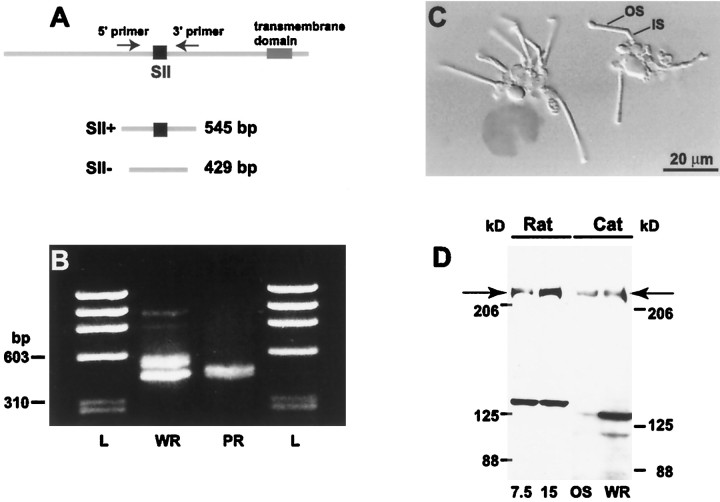
Fig. 1.
Mammalian retina expresses type I IP3receptor. A, Diagram of type I IP3 receptor mRNA and location of PCR primers (arrows).B, RT-PCR of the SII-containing region of type I IP3 receptor on rat whole retina (WR) and isolated photoreceptors (PR). L, DNA molecular weight ladder. C, Differential interference image of two groups of isolated rat photoreceptors used for RT-PCR. The outer segment (OS) and inner segment (IS) are indicated. D, Western blots of protein extracts from rat and cat probed with C-terminus antibody against type I IP3 receptor. For rat, two protein concentrations (7.5 and 15 μg) from whole retina were loaded. For cat, 15 μg was loaded.OS, Outer segments; WR, whole retina.Arrows point to type I IP3 receptor-positive band at the predicted molecular weight.
Reverse transcription-PCR (RT-PCR)
Total RNA from both whole retina and photoreceptors was isolated by acid guanidium and phenol-chloroform extraction (Chomzynski and Sacchi, 1987). The reverse transcription (RT) reaction was performed at 42°C for 50 min with 1–5 μg of total RNA in a 20 μl buffer containing (in mm) 50 Tris-HCl, pH 7.4, 60 KCl, 10 MgCl2, 1 DTT, and 0.5 of each dNTP plus 1 U/ml RNase inhibitor, 500 pmol random hexamer or 100 pmol of oligo dT, and 200 U of Super II M-MLV reverse transcriptase (Life Technologies). PCR reaction was performed in a buffer containing (in mm) 10 Tris, pH 8.3, 50 KCl, 2.5 MgCl2, and 0.4 dNTP plus 0.2 μm 5′ and 3′ primers, 2 μl of reverse-transcribed cDNA, and 2.5 U of AmpliTaq (Perkin-Elmer, Branchburg, NJ). Thirty cycles (94°C for 1 min, 52°C for 1 min, and 72°C for 2 min) were performed on a programmable thermocycler (Perkin-Elmer). The sequences of PCR primers (synthesized by Life Technologies) designed to amplify the SII region of type I IP3 receptor included the upstream primer 5′GAGCTGTCTGTGCTCGTG3′ and downstream primer 5′GTCGATGACCAGATTGGAG3′.
Isolating outer segment proteins for Western blot
Outer segment proteins were isolated by a protocol described previously (Panico et al., 1990). Briefly, retina was vortexed in 51% sucrose in MOPS buffer [(in mm) 20 MOPS, 2 MgCl2, 100 KCl, 0.1 EDTA, 1 DTT, and 0.1 PMSF plus 0.7 μg/ml aprotinin, 0.7 μg/ml leupeptin, 0.7 μg/ml pepstatin A, and 0.7 μg/ml benzamidine], layered with more MOPS buffer, and spun for 30 min at 27,000 × g. Outer segments floating at the interface were collected, diluted with MOPS buffer, and spun again. The pellet was resuspended in 38% sucrose in MOPS buffer and passed three times through an 18 gauge needle. The preparation was layered again with MOPS buffer and spun. The orange material at the interface (now mostly outer segments) then was diluted with MOPS buffer, spun again, and saved for analysis.
Western blot
Protein samples (5–10 μg/μl) were dissolved in SDS loading buffer and separated by 8% SDS-PAGE (Laemmli, 1970). Proteins were transferred to a nitrocellulose membrane, incubated with primary antibody against type I IP3 receptor (1:500 to 1:1000 dilution) for 2 hr at room temperature, washed, incubated 2 hr with 1:2000 dilution of horseradish peroxidase (HRP)-conjugated goat anti-rabbit IgG (Jackson ImmunoResearch, West Grove, PA), washed, and detected by chemiluminescence (Amersham, Arlington Heights, IL).
Immunohistochemistry
Posterior eyecups from cat, monkey, rat, and rabbit were fixed with 4% paraformaldehyde and 0.01% glutaraldehyde in phosphate buffer (PB), pH 7.3, at room temperature for 1 hr and cryoprotected overnight in PB containing 30% sucrose.
Light microscopy. Retina was embedded in Tissue Freezing Medium (Triangle Biomedical Sciences, Durham, NC) and sectioned radially at 10 μm in the cryostat. Sections were preincubated in PB containing 10% normal goat serum and 0.3% Triton X-100 for 1 hr and then in the same solution containing primary antibody (diluted 1:200 to 1:1000) overnight at 4°C. After being rinsed, the sections were incubated in goat anti-rabbit F(ab′)2 conjugated to a fluorescent dye for 2 hr at room temperature, mounted in Vectashield mounting medium (Vector Laboratories, Burlingame, CA), and coverslipped.
Double labeling. For type I IP3 receptor and blue-sensitive opsin (both antibodies raised in rabbit) the sections were incubated in antibody against type I IP3 receptor, rinsed and incubated in goat anti-rabbit F(ab′)2 conjugated to HRP, developed with 3,3′-diaminobenzidine (DAB) and 0.1% hydrogen peroxide, treated with glycine buffer, pH 2.2, for 5 min to elute antibodies (DAB reaction product remains), incubated in antiserum against blue opsin, and incubated with goat anti-rabbit F(ab′)2 conjugated to Cy3. For type I IP3receptor and red- and green-sensitive opsin (both antibodies raised in rabbit) the sections were incubated sequentially in antibody against type I IP3 receptor, an excess of goat anti-rabbit Fab′ fragments conjugated to FITC (to cover all rabbit epitopes), antiserum against red/green opsin, and goat anti-rabbit F(ab′)2conjugated to rhodamine. Control experiments were similar except that the antiserum against red/green opsin was omitted. Under regular fluorescent intensity the immunoreactivity of type I IP3receptor was detected only with the FITC filter set, indicating that the second secondary antibody did not react with the first primary antibody.
Immunoelectron microscopy. Radial vibratome sections (50–100 μm) were immunostained as described above except that Triton X-100 was omitted. Sections were incubated with HRP-conjugated secondary antibody, developed with DAB and hydrogen peroxide, and intensified by gold-substitution silver-intensification (Johnson and Vardi, 1998). Sections were osmicated with osmium tetroxide (2%; 1 hr), stained with uranyl acetate (1%, 1 hr), dehydrated in ethanol (70–100%), cleared in propylene oxide, and embedded in Epon 812. Ultrathin sections (70–90 nm) were stained with uranyl acetate and lead citrate and viewed with a transmission electron microscope (JEOL 1200EX).
Primary antibodies
We used three different polyclonal antibodies against type I IP3 receptor (all raised in rabbit). The first was raised against the C-terminal peptide (amino acid 2731–2749; from Dr. S. K. Joseph, Thomas Jefferson University, Philadelphia, PA). The specificity of this antibody was established (Mignery et al., 1989;Joseph and Samanta, 1993; Joseph et al., 1995). The second (M) was raised against a fusion protein containing amino acids 4466–5723 of type I IP3 receptor (Lin, 1995). The third (3′α) was raised against a fusion protein containing amino acids 7761–8027 (Lin, 1995). M and 3′α antibodies were obtained from Dr. William Agnew, Johns Hopkins University (Baltimore, MD). We also used a monoclonal antibody to type III IP3 receptor (Transduction Laboratories, Lexington, KY). Rabbit polyclonal antibodies against blue and red/green opsins were obtained from Dr. Jeremy Nathans, Johns Hopkins University (Baltimore, MD).
RESULTS
Photoreceptors express a splice variant of the type I IP3 receptor
To determine whether the transcript of the type I IP3receptor is expressed in photoreceptors, we performed RT-PCR. PCR primers were designed to flank the SII splicing region (Fig.1A). In whole retina, RT-PCR amplified two major DNA products with distinct molecular sizes: 545 and 429 bp (Fig. 1B). Direct DNA sequencing from these two bands showed that the larger product contained the exons A, B, and C in the SII region (SII+), whereas the smaller one lacked any of these exons (SII−). In isolated photoreceptors (Fig. 1C), RT-PCR amplified only the smaller splice variant (Fig. 1B) with a DNA sequence identical to the known sequence of the splice variant lacking the A, B, and C exons (SII−). This experiment was repeated three times with identical results.
To see if type I IP3 receptor is translated in photoreceptors, we prepared Western blots from whole retina (rat and cat) and from outer segments (cat) and probed them with an antibody against the C terminus. A prominent band at ∼220 kDa, corresponding to the approximate molecular weight of type I IP3 receptor, was detected in all blots (Fig. 1D). Sometimes an additional band at ∼130 kDa was labeled also; this is probably a degradation product (Joseph et al., 1995), but it could be a cross-reaction with a different protein. In the outer segments the major band of the expected molecular weight was prominent, and the smaller degradation product was always negligible. This suggests that the SII+ splice variant of type I IP3receptor is more likely to degrade.
Type I IP3 receptor is expressed strongly in cone outer segments
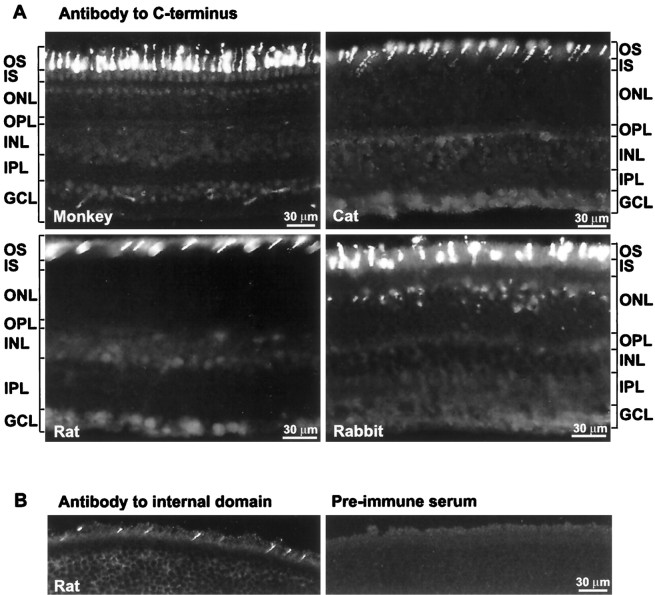
In all species tested (monkey, cat, rat, guinea pig, and rabbit), stain for type I IP3 receptor (against the C terminus) was strong in cone outer segments but very weak in the outer plexiform layer, the inner part of the inner nuclear layer, and the ganglion cell layer (Fig. 2A). No stain was observed in the inner plexiform layer. Rod outer segments were slightly positive (especially in rat), but this was evident only in semithin and ultrathin sections (see below).
Fig. 2.
Type I IP3 receptor is localized to cone outer segments. A, Frozen radial sections immunostained for type I IP3 receptor with C-terminus antibody. All species show strong staining in cone outer segments. In rabbit, the cells located in the outer layer of the ONL also stain strongly; their location and distribution suggest that these are cone somas. GCL, Ganglion cell layer; INL, inner nuclear layer; IPL, inner plexiform layer;IS, inner segments; ONL, outer nuclear layer; OPL, outer plexiform layer; OS, outer segments. B, Left, Rat section immunostained with antibody “M” directed against an internal domain of type I IP3 receptor. Cone outer segments are stained distinctly. B, Right, Rat section stained with the preimmune serum is devoid of stain.
To test whether staining was specific for type I IP3receptor, we applied two additional antibodies prepared against different domains of the rat receptor. Both antibodies gave distinct staining of cone outer segments, but the background was high. Control sections incubated with the preimmune serum were negative (Fig.2B, shown only for antibody “M”). A monoclonal antibody against type III IP3 receptor was negative for all retinal cells (tested in rat; data not shown).
Type I IP3 receptor is not detected in S cones
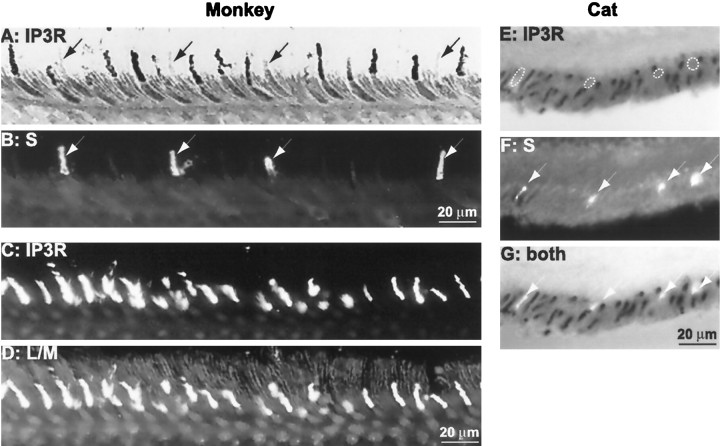
We noticed in monkey retina that the antibody to type I IP3 receptor failed to stain some cone outer segments (Fig.3A, arrows). Because the blue cone comprises only 5–10% of all cones, we surmised that it was unstained. To test this, we sequentially stained sections from monkey retina for type I IP3 receptor and blue-sensitive opsin. All cone outer segments negative for type I IP3 receptor were strongly positive for blue-sensitive opsin (Fig. 3A,B, three experiments). We also double-labeled retina for the IP3 receptor and red- and green-sensitive opsin. All labeled cones (i.e., red and green cones) were positive for type I IP3 receptor (Fig. 3C,D, two experiments). The blue cone in cat, rat, and rabbit (Fig.3E–G) was also negative for type I IP3receptor.
Fig. 3.
S cones do not stain for type I IP3 receptor. A, B, Monkey retina stained with antibodies against type I IP3 receptor (A; visualized with DAB reaction product) and blue-sensitive opsin (B; visualized with Cy3).Arrows point to S cone outer segments that are negative for type I IP3 receptor but are positive for blue-sensitive opsin. C, D, Monkey retina stained with antibodies against type I IP3 receptor (C; FITC) and red/green opsin (D; rhodamine). All cone outer segments stained for type I IP3 receptor are also positive for red- and green-sensitive opsin. E–G, Cat retina stained with antibodies against type I IP3 receptor (E; DAB) and blue-sensitive opsin (F; Cy3). Dotted outlines in E designate the location of the blue cone outer segments. G, Simultaneous visualization of both stainings: cone outer segments stained for the blue opsin do not stain for type I IP3receptor.
Ultrastructural localization of type I IP3 receptor
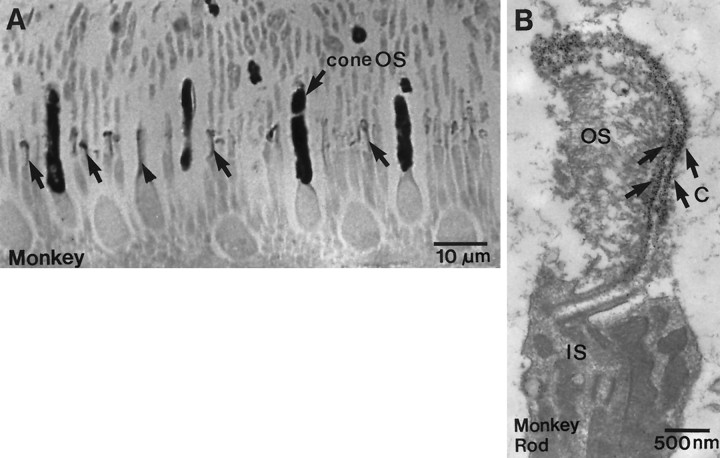
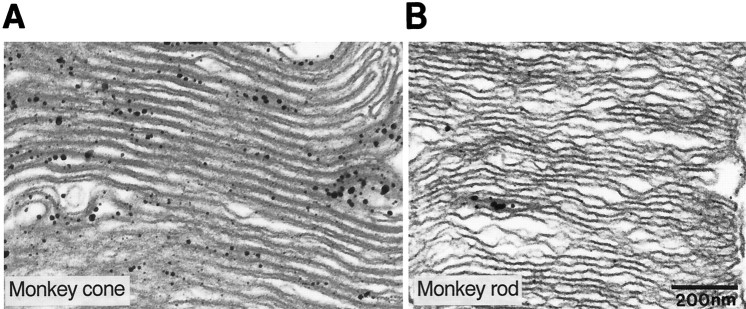
By light microscopy, stain for type I IP3 receptor was strong in cone outer segments but was barely detectable in rod outer segments. To determine whether this difference merely reflected the greater thickness of the cone or whether it represented a denser expression of receptor, we examined semithin sections cut parallel to the long axis of photoreceptor outer segments. Even in these ∼0.5-μm-thick sections, one-half the thickness of a rod outer segment, cones stained much more strongly than rods (Fig.4A). Furthermore, at the electron microscope level, gold particles representing immunostaining were denser in cone outer segments than in rods (Fig.5A,B). Stronger cone staining might occur if the disks communicate with the extracellular space, as they do in amphibians (Laties and Liebman, 1970), for this might render them more accessible to the antibody. However, disks in mammalian cones commonly are closed, as in rod (Cohen, 1970; Anderson and Fisher, 1976;Rodieck, 1988). Therefore, in cone and rod, access of antibody to the IP3 receptor may be similar. When equal access was assured by treating the tissue with a high concentration of detergent (0.5%), cone staining compared with rods was even more pronounced. Therefore, greater cone staining probably reflects their stronger expression of type I IP3 receptor.
Fig. 4.
Stain for type I IP3 receptor in cones is stronger than in rods, and it is also present in connecting cilia (monkey). A, Light micrograph of a 1 μm Epon section; cone outer segments (cone OS) are much stronger than rod outer segments. Arrows indicate stained connecting cilia in rods; the arrowhead indicates stained connecting cilium in a cone. B, Electron micrograph of a rod connecting cilium (c). Arrowsindicate the staining (gold deposits) along the tubular structures of the connecting cilium. IS, Inner segment;OS, outer segment.
Fig. 5.
Type I IP3 receptor is localized to cone and rod disk membranes (monkey). A, In the cone outer segment the immunodeposits are dense. In a fixed tissue the hypertonic condition often causes the disk membrane to collapse, which leads to a narrow intradisk lumen and wide interdisk space (or cytoplasmic space). Almost all of the immunodeposits are in the cytoplasmic space. B, In the rod outer segments the immunodeposits are scattered.
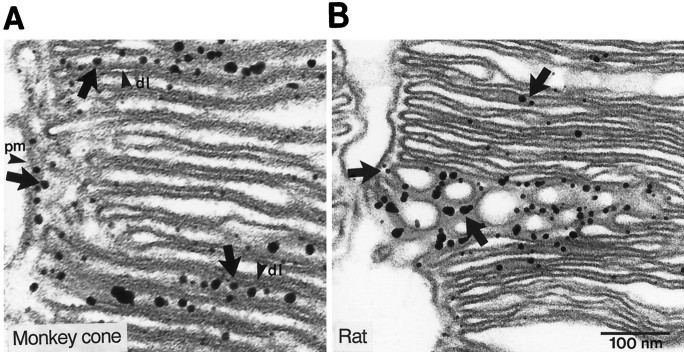
Most immunostain was localized to the cytoplasmic face of the disk and plasma membrane (Fig.6A). This was easiest to see where disks were swollen, because the disk lumen then could be distinguished clearly from the cytoplasmic space (Fig.6B). Staining of the cytoplasmic face matches the topology of the receptor, because its C terminus (target of the antibody) is thought to be cytoplasmic (Mikoshiba et al., 1994). In rods the sparse staining might be nonspecific; however, because the gold particles were located only on the cytoplasmic face of the membrane, they probably represent genuine, although weak, expression of IP3 receptor.
Fig. 6.
Staining for type I IP3 receptor is present on the cytoplasmic face of the plasma membrane.A, Monkey cone outer segment. Arrowheadsindicate disk lumen (dl) and plasma membrane (pm). Arrows indicate that the staining is associated at the cytoplasmic side of disk and plasma membranes. B, In rat it is difficult to discriminate cones from rods, but because rods are 100-fold more abundant and most neighboring outer segments appear similar to this one, we think that it is a rod outer segment. Arrows indicate that the staining is associated at the cytoplasmic side of disk membranes.
Thin sections also revealed stain over the rod cilium that connects the inner and outer segment (see Fig. 4A). Within the cilium, stain was concentrated along the microtubules (see Fig.4B). Staining also was observed in the cone cilium (see Fig. 4A), but microtubules were not discerned easily.
DISCUSSION
We provide strong evidence that the type I IP3receptor is expressed in photoreceptor outer segments, especially in red- and green-sensitive cones: (1) mRNA of a particular splice variant (SII−) was amplified from isolated photoreceptors; (2) a single protein band with the expected molecular weight was demonstrated by Western blot of the outer segments; (3) strong staining for the receptor was detected with three different antibodies, whereas controls (preimmune serum) were negative; (4) antibody against the C terminus was localized to the cytoplasmic face of the disk and the plasma membrane, in accordance with the known receptor topology (Mikoshiba et al., 1994).
The finding of IP3 receptor on the outer face of the disk supports previous findings that IP3 can release Ca2+ from internal stores (Parker et al., 1986;Schnetkamp and Szerencsei, 1993; Schnetkamp, 1995b). Because IP3 usually is associated with smooth endoplasmic reticulum, localization of the type I IP3 receptor on theplasma membrane may seem surprising. However, IP3 receptor also localizes to plasma membrane in olfactory cilia, mast cells, and T-lymphocytes (Kuno and Gardner, 1987; Penner et al., 1988; Cunningham et al., 1993). This site can admit Ca2+ from the extracellular space, where the concentration (∼3 mm) is apparently the same as in the disk lumen (for review, see Schnetkamp, 1989). In mammals, because disk surface area is greater than plasma membrane surface area, the disks probably provide most of the IP3-mediated Ca2+ influx.
Possible function of type I IP3 receptor in red- and green-sensitive cones
Ca2+ plays a key role in terminating the light response and adaptation (Fig. 7). When light via the rhodopsin cascade reduces cGMP, Ca2+influx through the cGMP-gated channel decreases and, as extrusion continues, cytoplasmic Ca2+ declines. Lower Ca2+ (1) activates rhodopsin kinase, (2) inhibits phosphodiesterase, (3) activates guanylyl cyclase, and (4) increases the affinity of the cGMP-gated channel for its ligand. All of these effects help to terminate the light response and adapt the photoreceptor (i.e., reduce gain and restore sensitivity to a stronger light). A quantitative model of the rod response does not require an additional mechanism to modulate Ca2+ (Lamb and Pugh, 1992; Lyubarsky and Pugh, 1996; Nikonov et al., 1998). However, the cone recovers faster than the rod and is less sensitive. Conceivably, the abundant IP3 receptor on cone disk membranes might contribute to these response properties as we now explain.
Fig. 7.
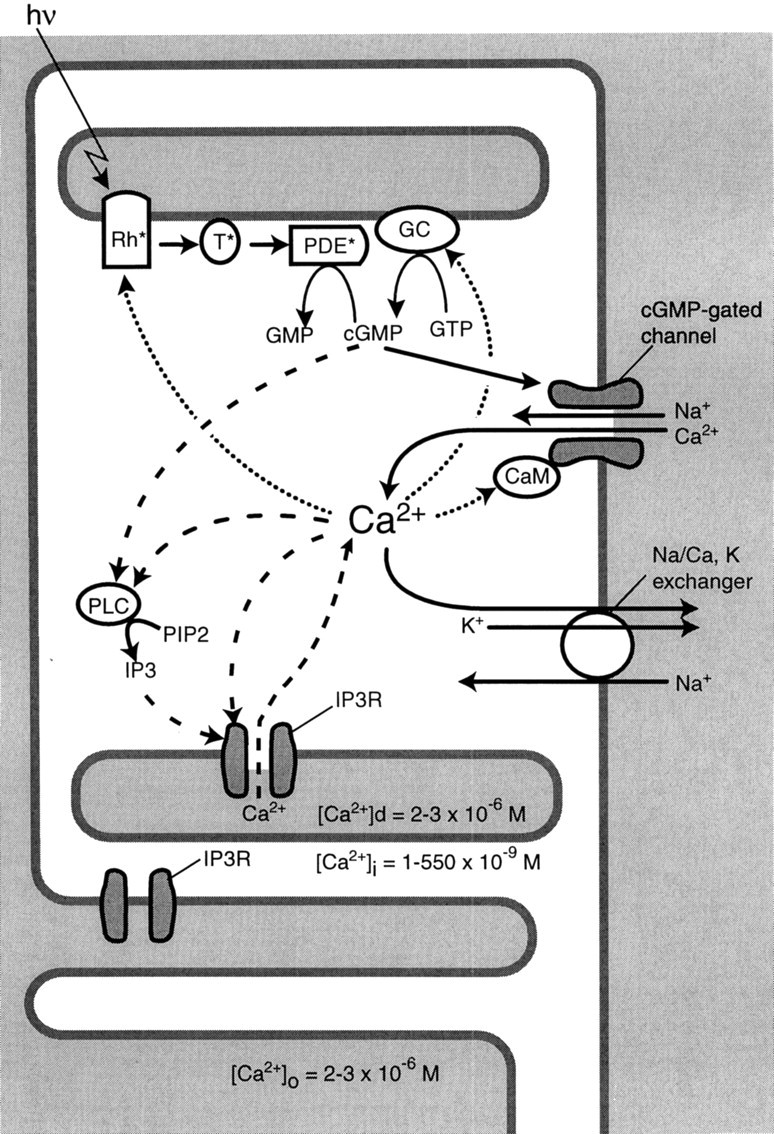
How the IP3 receptor might contribute to response recovery and adaptation. Solid arrows mark the phototransduction cascade leading from light (hυ) to the successive activation of opsin (Rh*), transducin (T*), phosphodiesterase (PDE*), and the hydrolysis of cGMP. Cation channels gated by cGMP close, thereby reducing Ca2+ influx, but Ca2+extrusion continues, so cytoplasmic Ca2+ falls. Low cytoplasmic Ca2+ affects several processes that terminate the light response and contribute to the response recovery (dotted arrows): opsin is phosphorylated, guanylyl cyclase (GC) is activated to synthesize cGMP, and channel affinity for cGMP is increased by binding Ca2+/calmodulin (CaM). The IP3 receptor (IP3R) on the disk and plasma membranes would accelerate changes in Ca2+i (dashed arrows). When Ca2+ and cGMP fall, phospholipase C (PLC) is suppressed, reducing IP3. Because both IP3 and Ca2+ regulate the IP3 receptor, their fall reduces Ca2+mobilization from the disks and extracellular space. This positive feedback loop via the IP3 receptor should accelerate the fall of Ca2+ after a light stimulus and its rise after a dark stimulus.
The IP3 receptor might provide a positive feedback loop (Fig. 7). This would accelerate the fall of cytoplasmic Ca2+ after a light flash or its rise after a dark flash. Phospholipase C (PLC), the enzyme that produces IP3, is present in cones [Ferreira and Pak (1994), but see Peng et al. (1997) and discussion below] where it might be stimulated constitutively by cGMP and Ca2+(Ghalayini and Anderson, 1987; Rhee and Bae, 1997; Haque et al., 1998). Therefore, light ONset, by reducing cGMP and Ca2+, would inhibit PLC. This would reduce IP3 and thus the release of intradisk Ca2+. The IP3ligand binding of the receptor is increased by Ca2+up to ∼200 nm but is reduced above this level (Bezprozvanny et al., 1991; Li et al., 1995; Patel and Taylor, 1995;López-Colomè and Lee, 1996; Kaznacheyeva et al., 1998). In darkness, cytoplasmic Ca2+ is near this optimum for IP3 binding (Korenbrot, 1995), so the fall in Ca2+ after light stimulation would reduce IP3 binding and accelerate the fall in Ca2+. At light OFFset, Ca2+influx via the cGMP-gated channel rises. This would activate IP3 binding and accelerate the rise of cytoplasmic Ca2+. This loop for accelerating the rise of Ca2+ would cease as Ca2+ rises beyond the optimal concentration for IP3 binding.
Two points might seem inconsistent with the model. First, although biochemistry suggests an IP3 signaling system in purified photoreceptor outer segments, physiology finds no such effect on the light response of intact cells. However, most physiology has focused on rods in which the pathway is minor. Second, although light on isolated disk membranes increases IP3, our model shows light decreasing IP3. However, both PLC and the IP3receptor depend critically on the Ca2+ level, so the decisive test requires an intact cell.
It remains unclear which isoform of PLC is expressed by cones. Ferreira and Pak (1994) identified PLC-β4, but Peng et al. (1997) did not concur. The issue matters because PLC-β is activated by a member of the Gq family, whereas other PLC isoforms are activated differently, for example, by a different G-protein (Gh), a tyrosine kinase, or a lipid-derived second messenger (for review, see Rhee and Bae, 1997).
If the IP3 receptor accelerates the cone response, why is it absent from the blue-sensitive cone? Possibly the blue-sensitive cone expresses a different isoform; alternatively, the blue-sensitive cone and the rod both express the IP3 receptor at very low levels. Vision mediated by the blue cone does share several features with rod vision. For example, both have a longer integration time and a higher sensitivity than vision mediated by red- and green-sensitive cones (Brindley et al., 1966; Mollon and Polden, 1977a,b; Zrenner and Gouras, 1979, 1981; Williams et al., 1981; Nelson, 1985). Conceivably, the extra loop for rapidly driving cytoplasmic Ca2+through larger excursions is reduced or absent because it would ill serve a slower, more sensitive response.
Footnotes
This work was supported by National Institutes of Health Grants EY11105-0 and EY08124. We thank Drs. Paul Liebman, Edward Pugh, Robert Smith, William Agnew, and Suresh K. Joseph for insightful discussions. We also thank Yi-Jun Shi and Tina Geueke for excellent technical assistance and John Demb and Madeleine Johnson for reading this manuscript. Antibodies are kind gifts from Drs. Suresh K. Joseph, William Agnew, Dan Wu, and Jeremy Nathans.
Correspondence should be addressed to Dr. Noga Vardi, Department of Neuroscience, University of Pennsylvania, 122B Anatomy/Chemistry Building, Philadelphia, PA 19104.
Dr. Wang’s present address: Department of Pathology, Johns Hopkins University, School of Medicine, Baltimore, MD 21205.
REFERENCES
- 1.Anderson DH, Fisher SK. The photoreceptors of diurnal squirrels: outer segment structure disc shedding and protein renewal. J Ultrastruct Res. 1976;55:119–141. doi: 10.1016/s0022-5320(76)80087-1. [DOI] [PubMed] [Google Scholar]
- 2.Berridge MJ. Inositol triphosphate and calcium signaling. Nature. 1993;361:315–325. doi: 10.1038/361315a0. [DOI] [PubMed] [Google Scholar]
- 3.Bezprozvanny I, Watras J, Ehrlich BE. Bell-shaped calcium–response curves in Ins 1,4,5-P3- and calcium-gated channels from endoplasmic reticulum of cerebellum. Nature. 1991;351:751–754. doi: 10.1038/351751a0. [DOI] [PubMed] [Google Scholar]
- 4.Brindley GS, Croz JJD, Rushton WAH. The flicker fusion frequency of the blue-sensitive mechanism of colour vision. J Physiol (Lond) 1966;183:497–500. doi: 10.1113/jphysiol.1966.sp007879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Brown JE, Blazynski C, Cohen AI. Light induces a rapid and transient increase in inositol triphosphate in toad rod outer segments. Biochem Biophys Res Commun. 1987;146:1392–1396. doi: 10.1016/0006-291x(87)90804-7. [DOI] [PubMed] [Google Scholar]
- 6.Chomzynski P, Sacchi N. Single-step method of RNA isolation by acid guanidium thiocyanate-phenol-chloroform extraction. Anal Biochem. 1987;162:156–159. doi: 10.1006/abio.1987.9999. [DOI] [PubMed] [Google Scholar]
- 7.Cohen AI. Further studies on the question of the patency of saccules in outer segments of vertebrate photoreceptors. Vision Res. 1970;10:445–453. doi: 10.1016/0042-6989(70)90001-5. [DOI] [PubMed] [Google Scholar]
- 8.Cunningham AM, Ryugo DK, Sharp AH, Reed RR, Snyder SH, Ronnett GV. Neuronal inositol 1,4,5-triphosphate receptor localized to the plasma membrane of olfactory cilia. Neuroscience. 1993;57:339–352. doi: 10.1016/0306-4522(93)90067-p. [DOI] [PubMed] [Google Scholar]
- 9.Danoff SK, Ferris CD, Donath C, Fischer GA, Munemitsu S, Ullrich A, Snyder SH, Ross CA. Inositol 1,4,5-triphosphate receptors: distinct neuronal and nonneuronal forms derived by alternative splicing differ in phosphorylation. Proc Natl Acad Sci USA. 1991;88:2951–2955. doi: 10.1073/pnas.88.7.2951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Day NS, Koutz CA, Anderson RE. Inositol 1,4,5-triphosphate receptors in the vertebrate retina. Curr Eye Res. 1993;12:981–992. doi: 10.3109/02713689309029224. [DOI] [PubMed] [Google Scholar]
- 11.Fain GL, Schroder WH. Calcium content and calcium exchange in dark-adapted toad rods. J Physiol (Lond) 1985;368:641–665. doi: 10.1113/jphysiol.1985.sp015881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ferreira PA, Pak WL. Bovine phospholipase C highly homologous to the NorpA protein of Drosophila is expressed specifically in cones. J Biol Chem. 1994;269:3129–3131. [PubMed] [Google Scholar]
- 13.Ghalayini A, Anderson RE. Phosphatidylinositol 4,5-biphosphate: light-mediated breakdown in the vertebrate retina. Biochem Biophys Res Commun. 1984;124:503–506. doi: 10.1016/0006-291x(84)91582-1. [DOI] [PubMed] [Google Scholar]
- 14.Ghalayini AJ, Anderson RE. Activation of bovine rod outer segment phospholipase C by ATP and GTP. Neurosci Res Commun. 1987;1:119–127. [Google Scholar]
- 15.Haque R, Uchida K, Iuvone PM. 8-Br-cGMP and cGMP phosphodiesterase (PDE) inhibitors stimulate intracellular calcium levels and inositol phosphate accumulation in cultured chick photoreceptor cells. Invest Ophthalmol Vis Sci. 1998;39:S1056. [Google Scholar]
- 16.Hayashi F, Amakawa T. Light-mediated breakdown of phosphatidylinositol 4,5-bisphosphate in isolated rod outer segments of frog photoreceptor. Biochem Biophys Res Commun. 1985;128:954–959. doi: 10.1016/0006-291x(85)90139-1. [DOI] [PubMed] [Google Scholar]
- 17.Johnson MA, Vardi N. Regional differences in GABA and GAD immunoreactivity in rabbit horizontal cells. Vis Neurosci. 1998;15:743–753. doi: 10.1017/s0952523898154135. [DOI] [PubMed] [Google Scholar]
- 18.Joseph SK, Samanta S. Detergent solubility of the inositol triphosphate receptor in rat brain membranes. Evidence for association of the receptor with ankyrin. J Biol Chem. 1993;268:6477–6586. [PubMed] [Google Scholar]
- 19.Joseph SK, Lin C, Pierson S, Thomas AP, Maranto AR. Hetero-oligomers of type-I and type-III inositol triphosphate receptors in WB rat liver epithelial cells. J Biol Chem. 1995;270:23310–23316. doi: 10.1074/jbc.270.40.23310. [DOI] [PubMed] [Google Scholar]
- 20.Kaznacheyeva E, Lupu VD, Bezprozvanny I. Single-channel properties of inositol 1,4,5-triphosphate receptor heterologously expressed in HEK-293 cells. J Gen Physiol. 1998;111:847–856. doi: 10.1085/jgp.111.6.847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Koch K-W. Control of photoreceptor proteins by Ca2+. Cell Calcium. 1995;18:314–321. doi: 10.1016/0143-4160(95)90027-6. [DOI] [PubMed] [Google Scholar]
- 22.Korenbrot JI. Ca2+ flux in retinal rod and cone outer segments: differences in Ca2+ selectivity of the cGMP-gated ion channels and Ca2+ clearance rates. Cell Calcium. 1995;18:285–300. doi: 10.1016/0143-4160(95)90025-x. [DOI] [PubMed] [Google Scholar]
- 23.Koutalos Y, Yau K-W. Regulation of sensitivity in vertebrate rod photoreceptors by calcium. Trends Neurosci. 1996;19:73–81. doi: 10.1016/0166-2236(96)89624-x. [DOI] [PubMed] [Google Scholar]
- 24.Kuno M, Gardner P. Ion channels activated by inositol 1,4,5-triphosphate in plasma membrane of human T-lymphocytes. Nature. 1987;326:19–25. doi: 10.1038/326301a0. [DOI] [PubMed] [Google Scholar]
- 25.Laemmli EK. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970;227:680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- 26.Lamb TD, Pugh EN., Jr G-protein cascades: gain and kinetics. Trends Neurosci. 1992;15:291–298. doi: 10.1016/0166-2236(92)90079-n. [DOI] [PubMed] [Google Scholar]
- 27.Lamb TD, Torre V. The control of photoreceptor sensitivity. In: Ritchie JM, Magistretti PJ, Bolis L, editors. Progress in cell research. Elsevier Science; Amsterdam: 1990. pp. 109–120. [Google Scholar]
- 28.Laties AM, Liebman PA. Cones of living amphibian eye: selective staining. Science. 1970;168:1475–1476. doi: 10.1126/science.168.3938.1475. [DOI] [PubMed] [Google Scholar]
- 29.Li YX, Keizer J, Stojilkovic SS, Rinzel J. Ca2+ excitability of the ER membrane: an explanation for IP3-induced Ca2+ oscillations. Am J Physiol. 1995;269:C1079–C1092. doi: 10.1152/ajpcell.1995.269.5.C1079. [DOI] [PubMed] [Google Scholar]
- 30.Liebman PA. Light-dependent Ca2+ content of rod outer segment disc membranes. Invest Ophthalmol. 1974;13:700–701. [PubMed] [Google Scholar]
- 31.Lin DDM. Expression and characterization of two alternatively spliced isoforms of rat type I receptor for inositol 1,4,5-triphosphate. PhD thesis. The Johns Hopkins School of Medicine; 1995. [Google Scholar]
- 32.López-Colomé AM, Lee I. Pharmacological characterization of inositol 1,4,5-triphosphate binding to membranes from retina and retinal cultures. J Neurosci Res. 1996;44:149–156. doi: 10.1002/(SICI)1097-4547(19960415)44:2<149::AID-JNR7>3.0.CO;2-D. [DOI] [PubMed] [Google Scholar]
- 33.Lyubarsky AL, Pugh EN., Jr Recovery phase of the murine rod photoresponse reconstructed from electroretinographic recordings. J Neurosci. 1996;16:563–571. doi: 10.1523/JNEUROSCI.16-02-00563.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.McNaughton PA. Rods, cones, and calcium. Cell Calcium. 1995;18:275–284. doi: 10.1016/0143-4160(95)90024-1. [DOI] [PubMed] [Google Scholar]
- 35.Mignery GA, Südhof TC, Takei K, de Camilli P. Putative receptor for inositol 1,4,5-triphosphate similar to ryanodine receptor. Nature. 1989;342:192–194. doi: 10.1038/342192a0. [DOI] [PubMed] [Google Scholar]
- 36.Mikoshiba K, Furuichi T, Miyawaki A. Structure and function of IP3 receptors. Cell Biol. 1994;5:273–281. doi: 10.1006/scel.1994.1033. [DOI] [PubMed] [Google Scholar]
- 37.Mollon JD, Polden PG. An anomaly in the response of the eye to light of short wavelengths. Philos Trans R Soc Lond [Biol] 1977a;278:207–240. doi: 10.1098/rstb.1977.0038. [DOI] [PubMed] [Google Scholar]
- 38.Mollon JD, Polden PG. Saturation of a retinal cone mechanism. Nature. 1977b;265:243–246. doi: 10.1038/265243a0. [DOI] [PubMed] [Google Scholar]
- 39.Nakagawa T, Okano H, Furuichi T, Aruga J, Mikoshiba K. The subtypes of the mouse inositol 1,4,5-triphosphate receptor are expressed in a tissue-specific and developmentally specific manner. Proc Natl Acad Sci USA. 1991;88:6244–6248. doi: 10.1073/pnas.88.14.6244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Nelson R. Spectral properties of cat horizontal cells. Neurosci Res Suppl. 1985;2:S167–S183. doi: 10.1016/0921-8696(85)90015-5. [DOI] [PubMed] [Google Scholar]
- 41.Nicol GD, Kaupp UB, Bownds MD. Transduction persists in rod photoreceptors after depletion of intracellular calcium. J Gen Physiol. 1987;89:297–319. doi: 10.1085/jgp.89.2.297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Nikonov S, Engheta N, Pugh EN., Jr Kinetics of recovery of the dark-adapted salamander rod photoresponse. J Gen Physiol. 1998;111:7–37. doi: 10.1085/jgp.111.1.7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Nucifora FC, Li S-H, Danoff S, Ullrich A, Ross CA. Molecular cloning of a cDNA for the human inositol 1,4,5-triphosphate receptor type I, and the identification of a third alternatively spliced variant. Mol Brain Res. 1995;32:291–296. doi: 10.1016/0169-328x(95)00089-b. [DOI] [PubMed] [Google Scholar]
- 44.Panico J, Parkes JH, Liebman PA. The effect of GDP on rod outer segment G-protein interactions. J Biol Chem. 1990;265:18922–18927. [PubMed] [Google Scholar]
- 45.Parker KR, Briggs JA, Dratz EA. Inositol triphosphate stimulates Ca2+ release form toad retinal rod outer segment preparations. Vis Recept. 1986;49:31A. [Google Scholar]
- 46.Patel S, Taylor CW. Quantal responses to inositol 1,4,5-triphosphate are not a consequence of Ca2+ regulation of inositol 1,4,5-triphosphate receptors. Biochem J. 1995;312:789–794. doi: 10.1042/bj3120789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Peng Y-W, Sharp AH, Snyder SH, Yau K-W. Localization of the inositol 1,4,5-triphosphate receptor in synaptic terminals on the vertebrate retina. Neuron. 1991;6:525–531. doi: 10.1016/0896-6273(91)90055-5. [DOI] [PubMed] [Google Scholar]
- 48.Peng Y-W, Rhee SG, Yu W-P, Ho Y-K, Schoen T, Chader GJ, Yau K-W. Identification of components of a phosphoinositide signaling pathway in retinal rod outer segments. Proc Natl Acad Sci USA. 1997;94:1995–2000. doi: 10.1073/pnas.94.5.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Penner R, Matthews G, Neher E. Regulation of calcium influx by second messengers in rat mast cells. Nature. 1988;334:499–504. doi: 10.1038/334499a0. [DOI] [PubMed] [Google Scholar]
- 50.Rhee SG, Bae YS. Regulation of phosphoinositide-specific phospholipase C isozymes. J Biol Chem. 1997;272:15045–15048. doi: 10.1074/jbc.272.24.15045. [DOI] [PubMed] [Google Scholar]
- 51.Rodieck RW. Comparative primate biology, Vol 4, Neurosciences, pp 203–278. Liss; New York: 1988. The primate retina. [Google Scholar]
- 52.Sasaki T, Kaneko A. l-Glutamate-induced responses in OFF-type bipolar cells of the cat retina. Vision Res. 1996;36:787–795. doi: 10.1016/0042-6989(95)00176-x. [DOI] [PubMed] [Google Scholar]
- 53.Schnetkamp PPM. Na-Ca or Na-Ca-K exchange in rod photoreceptors. Prog Biophys Mol Biol. 1989;54:1–29. doi: 10.1016/0079-6107(89)90007-2. [DOI] [PubMed] [Google Scholar]
- 54.Schnetkamp PPM. How does the retinal rod Na-Ca+-K exchanger regulate cytosolic free Ca2+? J Biol Chem. 1995a;270:13231–13239. doi: 10.1074/jbc.270.22.13231. [DOI] [PubMed] [Google Scholar]
- 55.Schnetkamp PPM. Calcium homeostasis in vertebrate retinal rod outer segments. Cell Calcium. 1995b;18:322–330. doi: 10.1016/0143-4160(95)90028-4. [DOI] [PubMed] [Google Scholar]
- 56.Schnetkamp PP, Bownds MD. Na+- and cGMP-induced Ca2+ fluxes in frog rod photoreceptors. J Gen Physiol. 1987;89:481–500. doi: 10.1085/jgp.89.3.481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Schnetkamp PPM, Szerencsei RT. Intracellular Ca2+ sequestration and release in intact bovine retinal rod outer segments. J Biol Chem. 1993;268:12449–12457. [PubMed] [Google Scholar]
- 58.Williams DR, MacLeod DIA, Hayhoe MM. Punctate sensitivity of the blue-sensitive mechanism. Vision Res. 1981;21:1357–1375. doi: 10.1016/0042-6989(81)90242-x. [DOI] [PubMed] [Google Scholar]
- 59.Yau K-W. Phototransduction mechanism in retinal rods and cones. Invest Ophthalmol Vis Sci. 1994;35:9–32. [PubMed] [Google Scholar]
- 60.Zrenner E, Gouras P. Blue-sensitive cones of the cat produce a rod-like electroretinogram. Invest Ophthalmol Vis Sci. 1979;18:1076–1081. [PubMed] [Google Scholar]
- 61.Zrenner E, Gouras P. Characteristics of the blue-sensitive cone mechanism in primate retinal ganglion cells. Vision Res. 1981;21:1605–1609. doi: 10.1016/0042-6989(81)90042-0. [DOI] [PubMed] [Google Scholar]