Abstract
The Drosophila Fog pathway represents one of the best-understood signaling cascades controlling epithelial morphogenesis. During gastrulation, Fog induces apical cell constrictions that drive the invagination of mesoderm and posterior gut primordia. The cellular mechanisms underlying primordia internalization vary greatly among insects and recent work has suggested that Fog signaling is specific to the fast mode of gastrulation found in some flies. On the contrary, here we show in the beetle Tribolium, whose development is broadly representative for insects, that Fog has multiple morphogenetic functions. It modulates mesoderm internalization and controls a massive posterior infolding involved in gut and extraembryonic development. In addition, Fog signaling affects blastoderm cellularization, primordial germ cell positioning, and cuboidal-to-squamous cell shape transitions in the extraembryonic serosa. Comparative analyses with two other distantly related insect species reveals that Fog’s role during cellularization is widely conserved and therefore might represent the ancestral function of the pathway.
Research organism: Other
Introduction
The Folded gastrulation (Fog) pathway is one of the few signaling pathways dedicated to epithelial morphogenesis (Gilmour et al., 2017; Manning and Rogers, 2014). Fog signaling was discovered in the fly Drosophila melanogaster, where it is required for the formation of two major epithelial folds during early embryogenesis: the ventral furrow, and the posterior gut fold (Costa et al., 1994; Parks and Wieschaus, 1991; Sweeton et al., 1991; Zusman and Wieschaus, 1985). The ventral furrow leads to the internalization of the mesoderm, while the posterior gut fold leads to internalization of the hindgut and posterior endoderm (Campos-Ortega and Hartenstein, 1997). These folds are formed by coordinated changes in cell shape that are driven by the reorganization of cytoskeleton components and the remodeling of cell junctions. Crucially, the inward directionality of the folding is caused by constrictions of the cells at their apical side, and it is this process that is coordinated by Fog signaling (Dawes-Hoang et al., 2005; Kölsch et al., 2007; Martin et al., 2009).
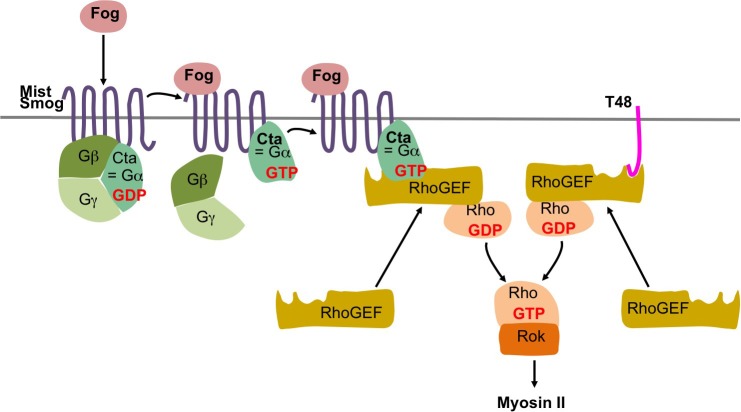
The molecular basis of the Fog signaling pathway has been extensively studied in Drosophila. Fog itself is an extracellular ligand that is secreted by future invaginating cells (Dawes-Hoang et al., 2005) and activates two G protein-coupled receptors (GPCRs): Mist (Mesoderm-invagination signal transducer, also known as Mthl1 [Methuselah-like1]) (Manning et al., 2013) and Smog (Jha et al., 2018; Kerridge et al., 2016). Activation of these receptors causes Concertina (Cta), the Gα12/13 subunit of a trimeric G protein, to recruit RhoGEF2 to the apical plasma membrane, where it promotes myosin II contractility (via Rho and Rho kinase), thereby triggering apical cell constrictions (Barrett et al., 1997; Dawes-Hoang et al., 2005; Kölsch et al., 2007) (Figure 1—figure supplement 1).
Although Fog is a secreted ligand, it appears to only act locally (Costa et al., 1994; Dawes-Hoang et al., 2005; Bailles et al., 2019). Because of this, the localized expression of fog and mist in the presumptive mesoderm and posterior endoderm provides the spatial specificity of the pathway (Costa et al., 1994; Manning et al., 2013).
It is important to note that in the absence of Fog signaling, some cells do still undergo apical constriction in the ventral furrow and posterior gut fold. However, fewer cells constrict, and the spatial and temporal coordination of those constrictions is disrupted. As such, Fog signaling is proposed to promote and coordinate apical constriction of cells across invaginating epithelia (Costa et al., 1994; Sweeton et al., 1991). Specifically, in fog mutants alone, ventral furrow formation is irregular and delayed compared with wildtype, but mesoderm internalization still occurs (Parks and Wieschaus, 1991; Seher et al., 2007; Sweeton et al., 1991). The transmembrane protein T48 also recruits RhoGEF2 apically and induces apical constrictions, in a Fog-independent manner (Kölsch et al., 2007) (Figure 1—figure supplement 1). Only deletion of both fog and T48 prevents mesoderm internalization (Kölsch et al., 2007). In contrast, posterior gut folding and endoderm internalization are completely dependent on Fog signaling (Seher et al., 2007; Zusman and Wieschaus, 1985). Here, Fog fulfills two functions: it induces apical constrictions locally, and it triggers a directional wave of Rho/MyoII activation that drives the propagation of cell invaginations outside of (anterior to) the fog expression domain (Bailles et al., 2019).
The Fog pathway is also involved in other morphogenetic events. During late embryogenesis, it is required during salivary gland morphogenesis and it affects the folding of imaginal discs in larvae (Chung et al., 2017; Manning et al., 2013; Nikolaidou and Barrett, 2004). Most recently, loss of Fog signaling was found to affect cell intercalation during germband extension (Jha et al., 2018; Kerridge et al., 2016), thus revealing functions for Fog signaling independent of apical constrictions.
The importance of Fog signaling during development in other insects is largely unknown. While the pathway components have been identified in many lineages, the morphogenetic basis of early development greatly varies between different species (Anderson, 1972a; Anderson, 1972b; Roth, 2004; Urbansky et al., 2016).
Recent molecular analysis in the midge Chironomus riparius has also cast doubts about the functional conservation of the pathway for early embryonic development. Rather than forming a highly stereotyped ventral furrow, Chironomus embryos internalize their mesoderm via cell ingression, and this event is only weakly affected by loss of Fog signaling (Urbansky et al., 2016). However, over-expression of fog and/or T48 causes the formation of a ventral furrow and invagination of mesoderm in a Drosophila-like mode. Based on their results, Urbansky et al. (2016) hypothesized that Fog signaling was recruited from a later role in development to an early role in gastrulation in the Drosophila lineage. However, as pointed out by the authors, an alternative hypothesis is that Fog signaling has a more widely conserved role in early development and this has been reduced in the lineage leading to Chironomus.
To test whether Fog signaling does have a more widely conserved role in early development, we have analyzed Fog pathway components in the beetle Tribolium castaneum. In contrast to Drosophila melanogaster and other dipteran species like Chironomus riparius, many features of Tribolium embryogenesis are more typical of insects in general, including the mechanism and timing of blastoderm cellularization (van der Zee et al., 2015), the mode of germ cell formation (Schröder, 2006), germband formation (Benton, 2018), extraembryonic tissue development (Hilbrant et al., 2016; Horn and Panfilio, 2016; van der Zee et al., 2005) and segmentation (Clark and Peel, 2018; Sommer and Tautz, 1993). Therefore, analyzing Fog signaling in Tribolium will reveal the role of the pathway within a developmental context that is more representative of insects.
Our analysis of Fog signaling in Tribolium reveals that, in contrast to Chironomus but like in Drosophila, this pathway contributes to mesoderm internalization and drives an early invagination at the posterior pole. In addition, Tribolium Fog signaling is involved in several aspects of development that have been lost or modified in the lineage leading to Drosophila, such as the extensive epithelial folding that leads to germband formation, the simultaneous spreading of the extraembryonic serosa, the apical-basal positioning of primordial germ cells, and even the cellularization of the blastoderm. The latter function is deeply conserved, as we also observed it in bug and cricket embryos representing distant branches of the insect phylogenetic tree.
Results
Tc-cta, Tc-mist and Tc-fog are expressed in morphogenetically active tissues
As a first step towards characterizing the Fog signaling pathway in Tribolium, we identified and cloned the pathway components (Figure 1—figure supplement 1) and characterized their expression during development. The Tribolium genome contains one ortholog each for fog, mist and cta (hereafter referred to as Tc-fog, Tc-mist and Tc-cta). Fog is a fast evolving protein with very low overall sequence conservation within insects and no detectable homologs in currently available non-insect genomes (Figure 1—figure supplement 2) (Urbansky et al., 2016). In contrast, previous research has shown mist and cta to be well conserved among insects (de Mendoza et al., 2016; Kozasa et al., 2011; Manning et al., 2013; Parks and Wieschaus, 1991; Urbansky et al., 2016).
In Drosophila, fog, mist and cta are all maternally expressed (Costa et al., 1994; Manning et al., 2013; Parks and Wieschaus, 1991; Urbansky et al., 2016). In Tribolium, we were unable to detect maternal expression for any of the three genes using conventional whole-mount RNA in-situ hybridization (ISH) (for staging and description of wildtype development see Benton et al., 2013; Handel et al., 2000). However, publically available RNA-sequencing data revealed the presence of maternal transcripts for Tc-cta and Tc-fog (Dönitz et al., 2018).
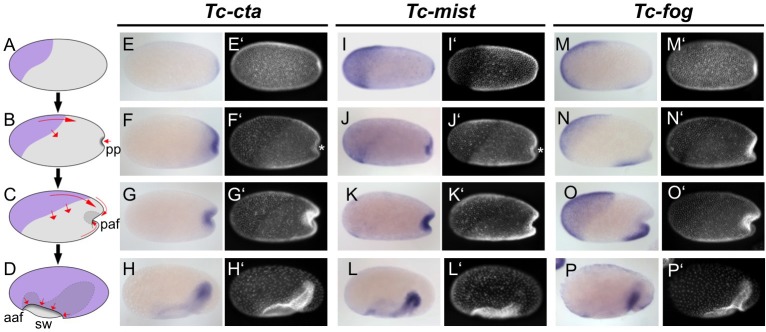
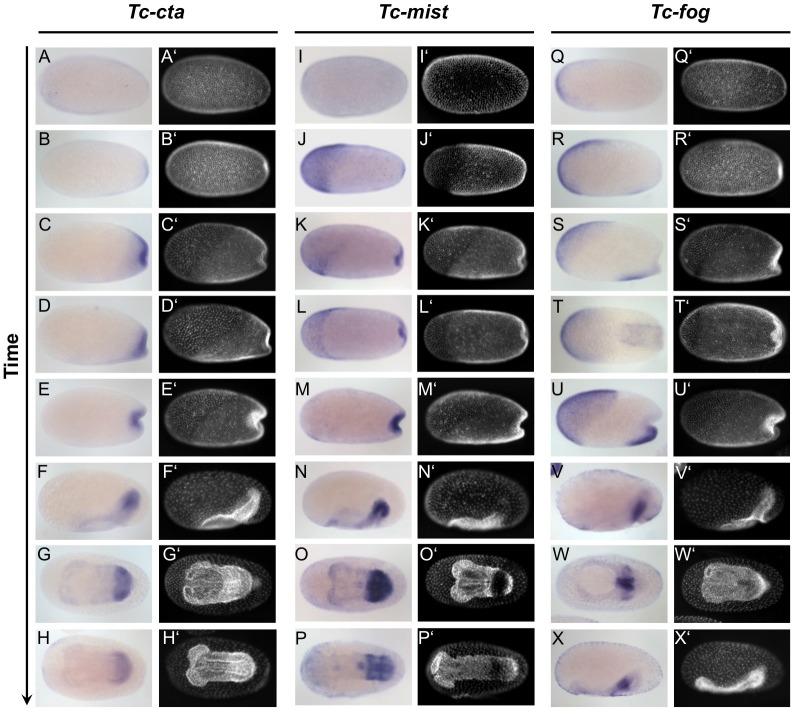
After blastoderm formation, Tc-cta and Tc-mist transcripts were uniformly distributed, while Tc-fog transcripts were enriched at the anterior pole (Figure 1—figure supplement 3A,I,Q). During later blastoderm stages, Tc-cta formed a shallow gradient with higher levels towards the posterior pole (Figure 1E), while Tc-mist and Tc-fog were strongly expressed in an oblique anterior-dorsal domain (the future serosa; Figure 1A,I,M). At the same time, weak Tc-mist expression became visible at the posterior pole, and weak Tc-fog expression was also visible in a patch of cells at the ventral side of the embryo (Figure 1I,M; Figure 1—figure supplement 4B).
Figure 1. Expression of Fog signaling components during early embryogenesis.
(A–D) Schematics showing embryo condensation as described in the text. Serosa is shown in purple, germ rudiment tissue is shown in gray, arrows display tissue movements. aaf: anterior amniotic fold, paf: posterior amniotic fold, pp: primitive pit, sw: serosal window. (E–P’) Whole mount ISH and DNA staining for Tc-cta (E–H), Tc-mist (I–L) and Tc-fog (M–P). (E’–P’) nuclear (DAPI) staining of respective embryos. Anterior is left, ventral is down (where possible to discern).
Figure 1—figure supplement 1. Fog and T48 pathway in Drosophila.
Figure 1—figure supplement 2. Insect Fog proteins.
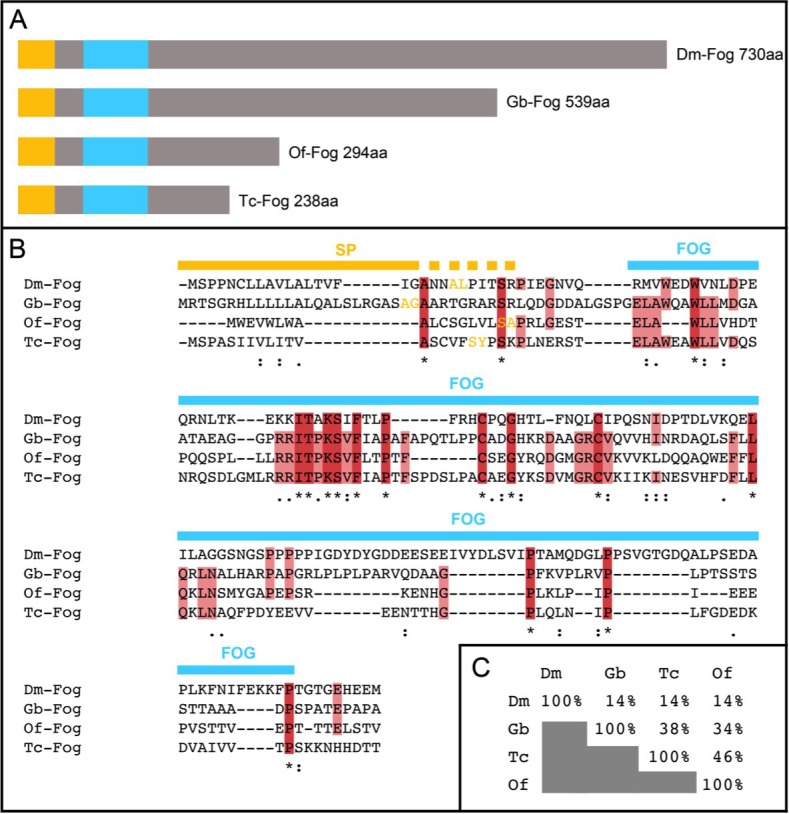
Figure 1—figure supplement 3. Expression of Tc-cta, Tc-mist and Tc-fog during early embryogenesis.
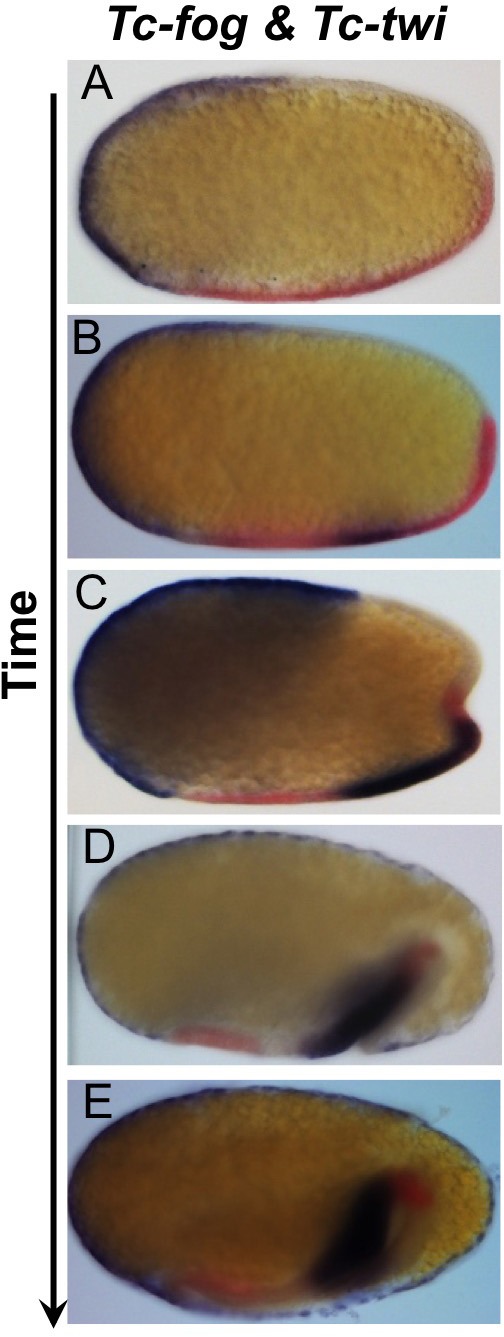
Figure 1—figure supplement 4. Tc-fog and Tc-twi are co-expressed only within the posterior presumptive mesoderm.
The complex morphogenetic events that transform the Tribolium blastoderm into the germband (schematic in Figure 1A–D, wildtype in Videos 1 and 2) have been described in detail elsewhere (Handel et al., 2000; Benton et al., 2013), but we will briefly present them here for the benefit of the reader. After cellularization is complete, cells that will form the embryo proper and the extraembryonic amnion (together termed the germ rudiment) undergo mitosis and condense towards the posterior-ventral region of the embryo (Figure 1B,C). At the same time, a patch of cells at the posterior pole undergoes apical constrictions to form a cup-shaped indentation (termed the primitive pit, pp in Figure 1B) that then deepens into a fold (termed the posterior amniotic fold, paf in Figure 1C, Video 3). Cells constrict in a pulsatile manner (Video 4 showing close up of cells), as previously described for the Drosophila ventral furrow (Martin et al., 2009). Subsequently, the dorsal ‘lip’ of the posterior amniotic fold moves ventrally, progresses over the posterior pole while undergoing involution, and then moves anteriorly over the ventral face of the embryo. Differences in the relative timing of posterior folding, cell division and tissue condensation between embryos lead to high variability in the overall appearance and progression of the posterior fold (Video 5 showing four embryos from posterior). As this process occurs, the edges of the posterior amniotic fold spread anteriorly until they meet with the anterior amniotic fold (which forms independently, aaf in Figure 1D). During the above condensation and tissue folding, the presumptive serosa cells undergo a cuboidal-to-squamous transition and spread over the entire egg surface without any cell division (Video 6 showing wildtype serosa flattening). The boundary between serosa and germ rudiment is demarcated by a supracellular actin cable (sca) that may be involved in serosal window closure (sw in Figure 2D and wild type in Video 2) (Benton et al., 2013). Throughout this period, mesoderm internalization occurs along the ventral part of the germ rudiment via both cell ingression and furrow formation mediated by apical constriction (Handel et al., 2005).
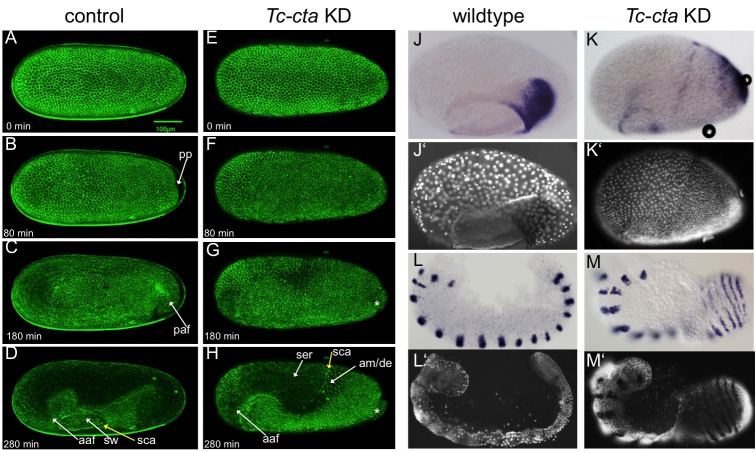
Figure 2. Fog signaling is required for posterior amniotic fold formation.
(A–H) Stills from live fluorescent imaging of LifeAct-eGFP transgenic embryos, ranging from late blastoderm to germband extension stages. (A–D) wildtype control. (E–H) Tc-cta KD. The asterisk marks a cluster of cells that becomes visible at the posterior pole. (J, K) Tc-pnr is expressed in a broad dorsal domain. (J’, K’) nuclear (DAPI) staining of respective embryos. (L, M) Tc-gsb expression marks forming and differentiating segments in elongating germ band embryos. (L’, M’) nuclear (DAPI) staining of respective embryos. (J, L) Wildtype. (K, M) Tc-cta KD. aaf: anterior amniotic fold, am/de: amnion dorsal ectoderm, paf: posterior amniotic fold, pp: primitive pit, sca: supracellular actin cable, sw: serosal window. Anterior is left, ventral is down.
Figure 2—figure supplement 1. KD of Tc-fog, Tc-mist and Tc-cta by RNAi results in similar phenotypes.
Figure 2—figure supplement 2. Fog signaling is required for posterior amniotic fold formation.
Figure 2—figure supplement 3. Morphogenetic defects in late Tc-cta KD embryos.
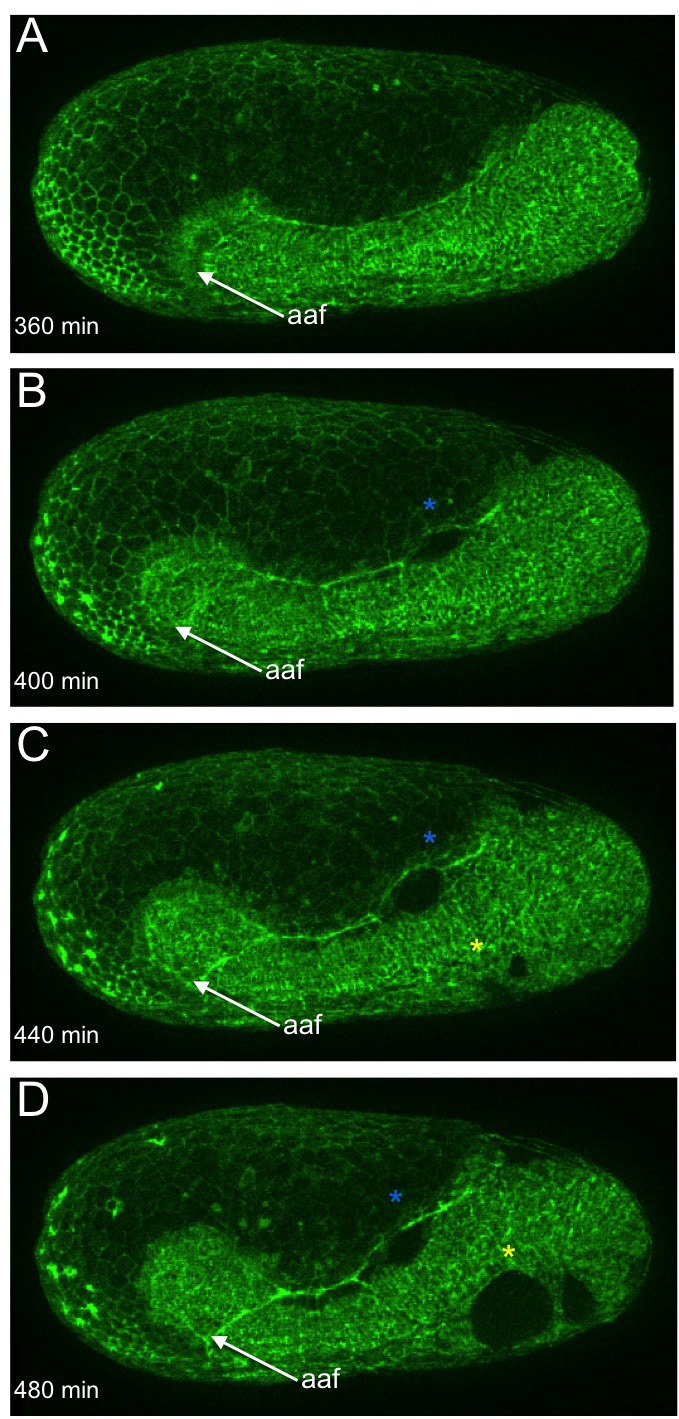
Figure 2—figure supplement 4. Frequencies of phenotypic defects upon Tc-fog KD.
Figure 2—figure supplement 5. Late development of Tc-cta KD embryos.
Video 1. Fluorescent live imaging of wildtype and Tc-fog RNAi nGFP transgenic embryos.
Maximum intensity projections of one egg hemisphere are shown with anterior to the left and ventral to the bottom.
Video 2. Fluorescent live imaging of wildtype and Tc-cta RNAi LifeAct-GFP transgenic embryos.
Maximum intensity projections of one egg hemisphere are shown with anterior to the left and ventral to the bottom.
Video 3. Fluorescent live imaging of the posterior pole of a wildtype embryo transiently expressing GAP43-YFP.
Apical constrictions are visible at the center of the forming fold. Embryo was mounted with the posterior pole towards the objective and the resulting movie was digitally rotated. Maximum intensity projection of posterior view is shown as well as a transverse section along the dorsal/ventral midline. Ventral is to the bottom.
Video 4. Single section through the subapical region of the cells at the posterior pole of a wildtype embryo transiently expressing GAP43-YFP.
Cells constrict over time and this occurs in a pulsatile manner, and cell intercalation is also visible. Ventral is to the bottom.
Video 5. Fluorescent live imaging of wildtype embryos transiently expressing GAP43-YFP.
Embryos were mounted with their posterior poles towards the objective. Maximum intensity projection of posterior view is shown. Ventral is to the bottom.
Video 6. Fluorescent live imaging of the presumptive serosa and germ rudiment epithelium of a wildtype embryo transiently expressing GAP43-YFP.
The cuboidal-to-squamous transition of the serosa cells during germband formation can be seen. Maximum intensity projection of the epithelium as well as transverse and sagittal sections along midpoints of the frame are shown. Anterior is to the left, ventral to the bottom.
Throughout embryo condensation, Tc-cta expression persisted in the posterior region of the germ rudiment/germ band (Figure 1F–H, Figure 1—figure supplement 3F–H). Tc-mist expression faded first from the dorsal serosal cells, then from the entire serosa, while expression in the primitive pit region/posterior end of the germ band strengthened (Figure 1J–L, Figure 1—figure supplement 3N–P). Tc-fog expression remained in the serosa throughout condensation and became upregulated in a posterior-ventral stripe of cells fated to become mesoderm (Figure 1N–P, Figure 1—figure supplement 3T–X; Figure 1—figure supplement 4C–E). Towards the end of embryo condensation, Tc-fog also became expressed in the ectoderm on either side of the mesoderm domain (Figure 1—figure supplement 3W).
Our expression analysis shows that Tribolium Fog signaling components are activated in a spatiotemporal pattern suggestive of a role in epithelial morphogenesis.
The Fog pathway is required for the posterior amniotic fold in Tribolium
To test whether the Fog signaling pathway is involved in early Tribolium embryogenesis, we disrupted Tc-cta, Tc-mist or Tc-fog function via parental RNAi (pRNAi) knockdown (KD) and analyzed both live and fixed embryos.
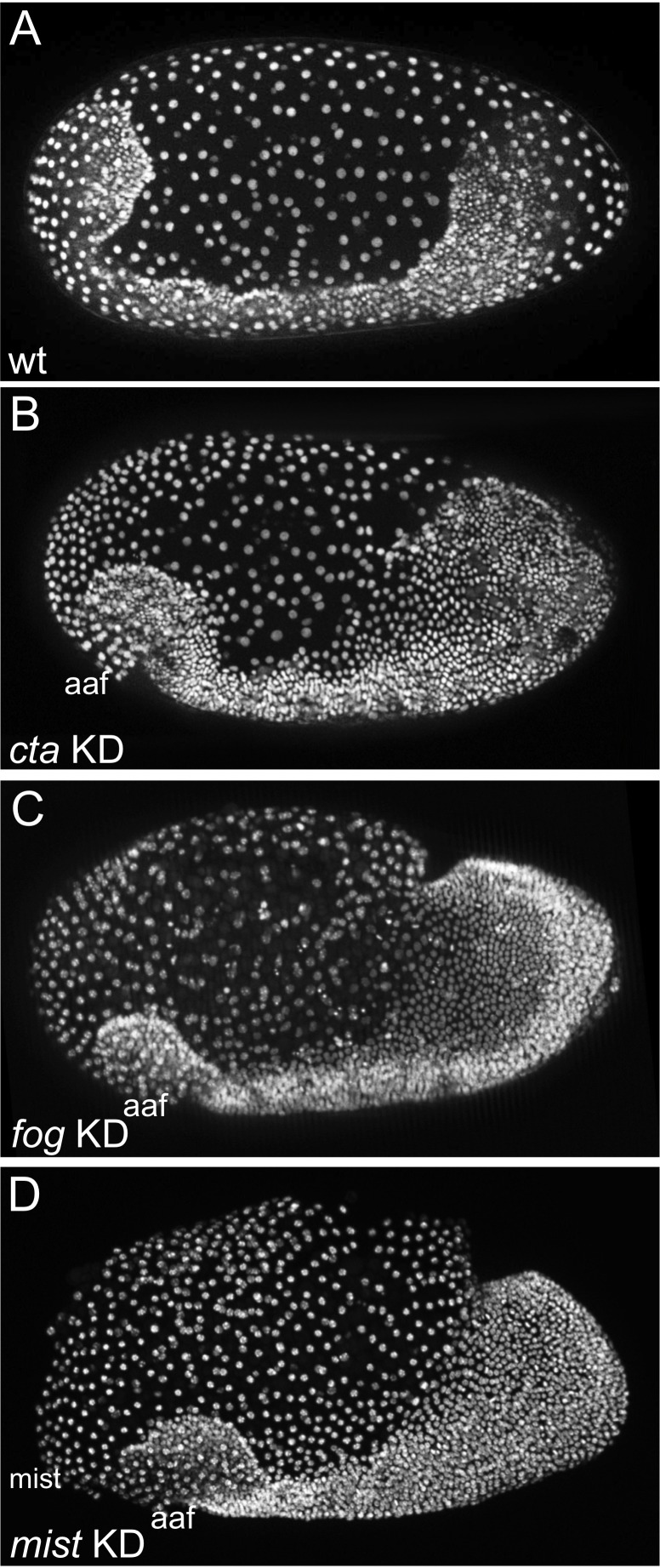
KD of each of the genes resulted in the same overall phenotype (Figure 2—figure supplement 1). We utilized fluorescent live imaging to better understand the underlying defects (Figure 2A–H; Videos 1, 2 and 7). The earliest and most prominent defect was the suppression of primitive pit and posterior amniotic fold formation (Figure 2B,C,F,G; Figure 2—figure supplements 2 and 4; Videos 2 and 7). Because of this lack of folding, the dorsal half of the germ rudiment remained at the dorsal side of the egg in KD embryos (Figure 2H,K; Figure 2—figure supplements 1 and 2; Figure 3).
Figure 3. Schematic representation of the embryonic phenotype produced by Fog pathway disruption.
Schematics showing wildtype development and the effects on embryo formation of RNAi disruption of Tc-fog, Tc-mist or Tc-cta. Anterior is left, ventral is down.
Video 7. Fluorescent live imaging of aTc-cta RNAi LifeAct-GFP transgenic embryo.
Maximum intensity projection of one egg hemisphere is shown with anterior to the left and ventral to the bottom.
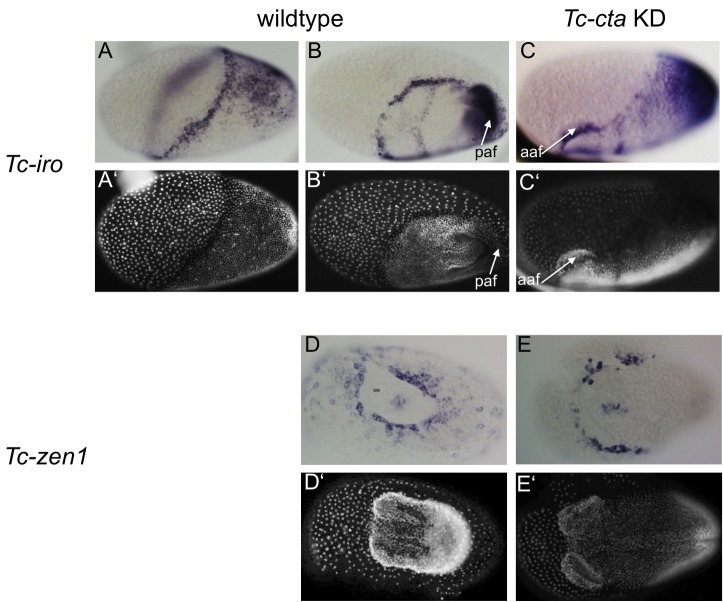
To investigate whether patterning of the dorsal half of the germ rudiment was disrupted in KD embryos, we analyzed the expression of two known marker genes. Despite the abnormal position of the relevant tissue, both Tc-pnr and Tc-iro, which are expressed in broad dorsal domains, appeared to be expressed normally in KD embryos (Figure 2J,K and Figure 2—figure supplement 2A–C). This finding is supported by the fact that a supracellular-actin cable formed between the serosa and germ rudiment tissues, as occurs in wildtype embryos at the same stage (sca in Figure 2D,H) (Benton et al., 2013).
In addition to the above defects, epithelial holes formed at the serosa/germ rudiment boundary and, during later stages of development, at posterior-ventral regions of the germband (Figure 2—figure supplements 3 and 4). In contrast to the major morphogenetic defects in the posterior of the embryo, anterior amniotic fold formation and head condensation still occurred in KD embryos (aaf in Figure 2H; Figure 2—figure supplement 1; Figure 2—figure supplement 2D, E and Figures 3 and 4 ).
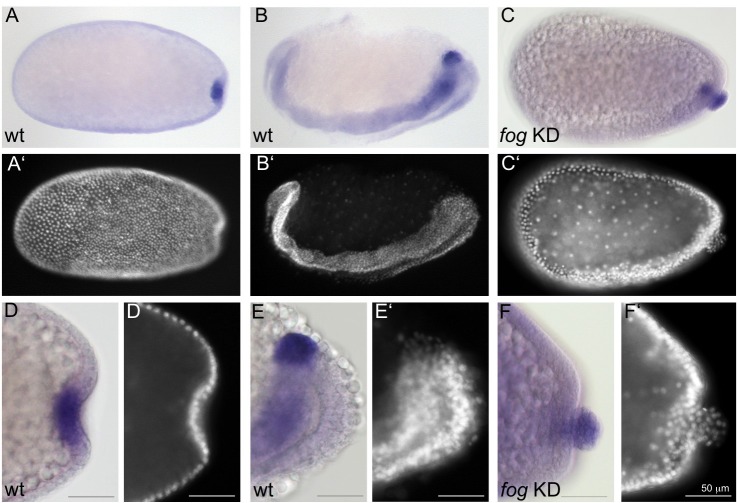
Figure 4. Fog signaling affects the positioning of the primordial germ cells.
Whole mount ISH for the germ cell marker Tc-tapas. (A, B, D, E) Wildtype. (C, F) Tc-fog KD. (A–C) Optical sagittal sections of whole embryos. (D–F) Optical sagittal sections of posterior regions. (A’–F’) DAPI staining of the respective embryos. (A, D) Wildtype embryo at primitive pit stage. (B, E) Wildtype embryo at early germ band extension stage. (C, F) Tc-fog KD embryos at stage corresponding to primitive pit stage in wildtype. Anterior is left, ventral is down.
Despite the severe changes in overall embryo topology, segmentation was not severely affected in KD embryos. In wildtype embryos, segmentation genes are often expressed in rings that completely encircle the epithelium of the germband (Benton et al., 2016; Sarrazin et al., 2012). In our KD embryos, these rings were readily visible due to the outward facing topology of the germband (e.g. Tc-gooseberry (Tc-gsb) (Davis et al., 2001) (Figure 2L,M).
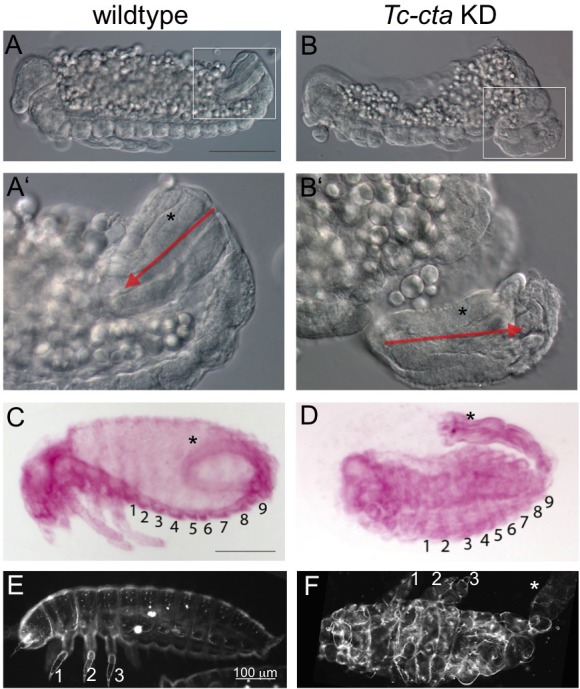
During germband retraction, KD embryos become highly distorted. However, segments could still be distinguished (Figure 2—figure supplement 5C,D). A conspicuous difference to control embryos is the wrong positioning of the hindgut. Instead of pointing inside the embryo, the hindgut points outwards, forming an external tube connected to the posterior tip of the embryo (Figure 2—figure supplement 5B, D). This suggests that primitive pit and posterior amniotic fold formation are also required for the correct internalization of the gut.
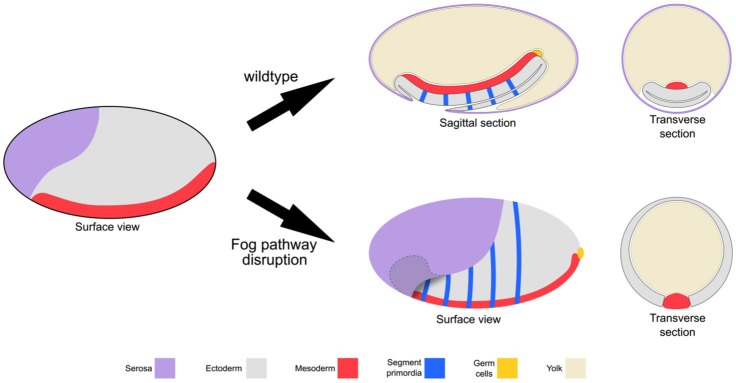
Taken together, Fog signaling is required for one of the most prominent morphogenetic movements during Tribolium gastrulation: the formation of the posterior amniotic fold that is essential both for extraembryonic membrane and gut development. In the absence of Fog the Tribolium embryo assumes a topology that is more like that of Drosophila: the dorsal ectoderm and extraembryonic tissues remain in a dorsal position (Figure 3).
Fog signaling controls the positioning of the primordial germ cells
Drosophila primordial germ cells (PGCs) are specified at the posterior pole of the early embryo and form as ‘pole cells’ above the surface of the blastoderm (Cinalli and Lehmann, 2013). In Tribolium, the PGCs are also specified at the posterior of the blastoderm, but they are integrated in the blastoderm cell layer and internalize beneath the blastoderm epithelium at around the same time as primitive pit formation (Schröder, 2006). In our live imaging analysis of KD embryos, we frequently observed a posterior ball of tissue (Figure 2G,H, white asterisk) and asked whether this tissue consisted of incorrectly localized PGCs.
To follow the development of Tribolium PGCs, we examined the expression of the gene Tc-tapas, which encodes a Tudor domain protein (Patil et al., 2014). Tc-tapas has a similar but more robust expression profile than the previously described PGC marker gene Tc-vasa (Schröder, 2006). In control embryos, the Tc-tapas expressing cells are located in the center of the forming primitive pit (Figure 4A,D). During early posterior amniotic fold formation, they leave the epithelium at its basal side by an unknown mechanism. Subsequently, the PGCs form a spherical cluster of cells that remains attached to the posterior end of the segment addition zone (SAZ) during germband extension (Figure 4B,E).
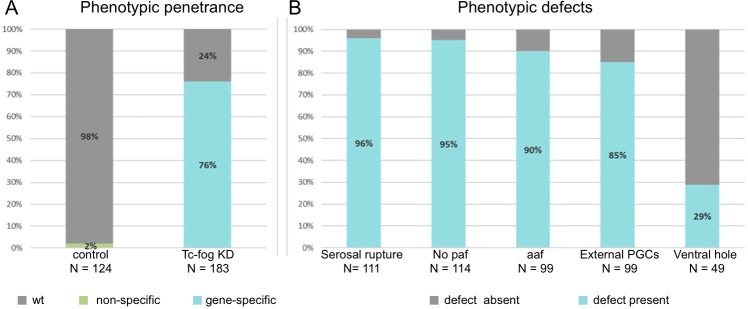
In Tc-fog pRNAi embryos, Tc-tapas was also expressed in a distinct cluster of putative PGCs at the posterior, but in most embryos (85% of KD embryos that displayed phenotypic defects [N = 99], Figure 2—figure supplement 4), the cell cluster was located at the apical side of the embryonic epithelium (Figure 4C,F). This cell cluster became visible in this location during embryo condensation, precisely when PGCs move beneath the epithelium in wildtype embryos (white asterisk in Figure 2G,H). Thus, in the absence of Fog signaling and primitive pit formation, the putative germ cells become mislocalized to the apical side of the embryonic epithelium (Figure 3).
Fog signaling is involved in, but not required for, mesoderm internalization
We next asked whether Fog signaling plays a role in mesoderm internalization in Tribolium. As described earlier, Drosophila Fog signaling is required for the formation of a deep ventral furrow, but mesodermal cells still internalize in Fog signaling mutant embryos (Seher et al., 2007; Sweeton et al., 1991; Zusman and Wieschaus, 1985).
In Tribolium, mesoderm internalization occurs at the ventral side of the embryo like in Drosophila, but the mode of internalization is less uniform (Handel et al., 2005). Shortly after primitive pit formation, mesodermal cells flatten and constrict apically (Handel et al., 2005), causing the formation of a ventral furrow that is shallow at the anterior (Figure 5A1 and A2) and deeper at the posterior (Figure 5A3, Figure 5—figure supplements 1 and 2A for outlines of apically constricting cells). After serosal window closure, the mesoderm is fully internalized and the left and right ectodermal plates fuse along the ventral midline (Figure 5B).
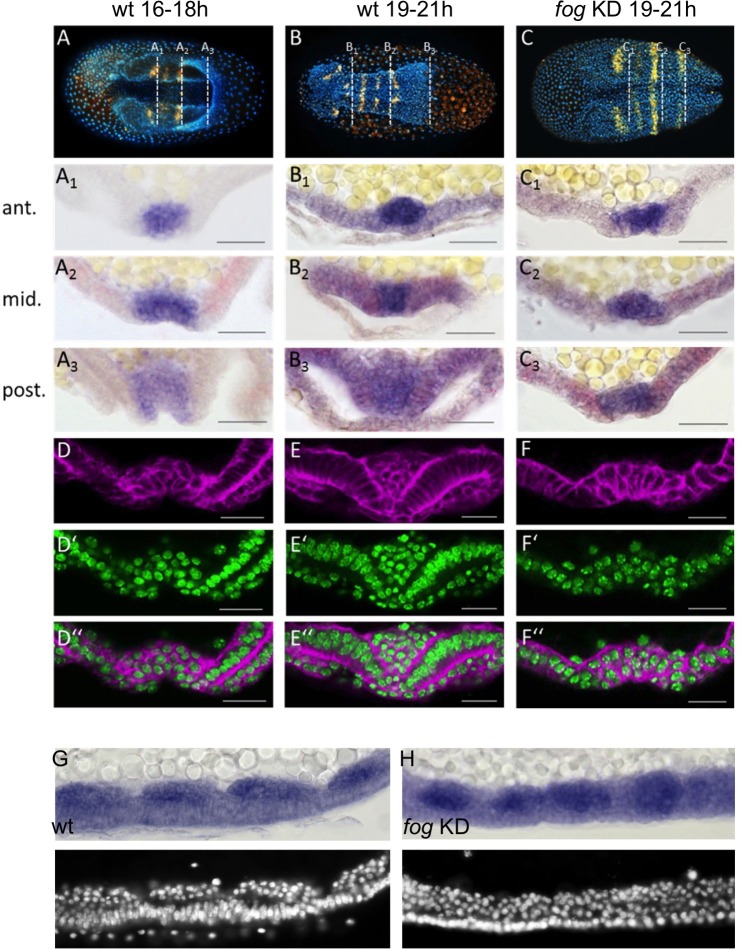
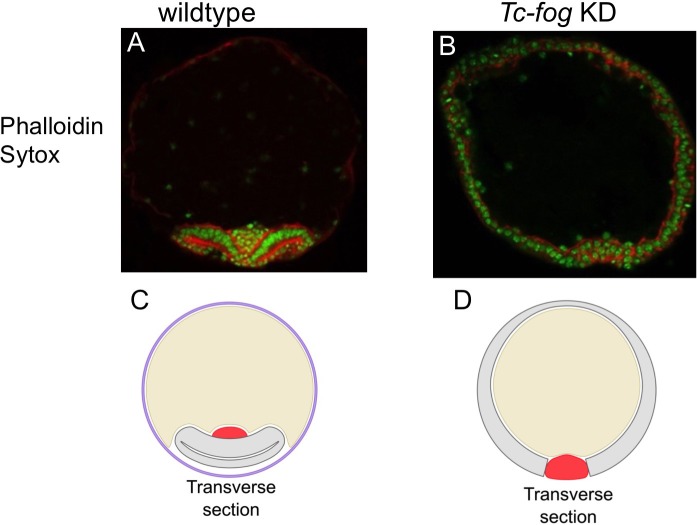
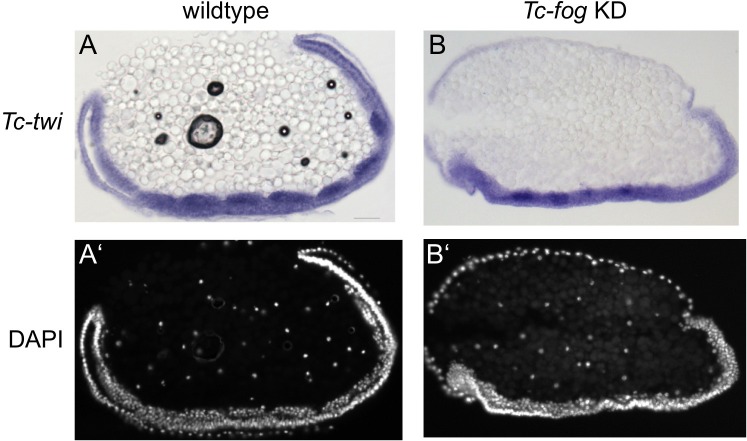
Figure 5. Tc-fog RNAi delays mesoderm internalization.
(A–C) Ventral views of whole mount embryos (anterior left) stained for the segmental marker Tc-gsb (yellow), nuclei (DAPI; blue). Embryos are also stained for Tc-twi expression but this is only visible in A (dark blue ventral domain). (A–B) Wildtype at horseshoe or early germband extension stage with two or four trunk Tc-gsb stripes, respectively. (C) Tc-fog KD at a stage corresponding to (B) with four trunk Tc-gsb stripes (the forth stripe is visible in lateral views). (A1–C3) transverse cryosections of the embryos shown in (A–C) with Tc-twi expression (blue) and Tc-gsb expression (red). The position of each section is indicated by a white dashed line in (A–C). (D–F’’) Transverse cryosections at posterior positions of embryos corresponding in age to those in (A–C) showing F-actin (phalloidin; magenta) and nuclei (sytox; green). (G, H) Sagittal cryosections of embryos during germband elongation (anterior left) showing Tc-twi expression (dark blue). (G’, H’) nuclear (DAPI) staining of respective embryos. Only a portion of the germband comprising four segments is shown. For corresponding sections showing the entire embryo see Figure 5—figure supplement 4. The different appearance of the mesoderm upon Tc-fog KD compared to wildtype is likely due to the delayed maturation of the mesoderm and the overall aberrant morphogenetic movements of KD embryos. For all embryos the serosa was removed during preparation. The cell sheet covering the ventral side of control embryos is the amnion (am) or amnion/dorsal ectoderm (am/de). Staging was done at 25°C.
Figure 5—figure supplement 1. Mesoderm internalization varies along the AP axis in Tribolium.
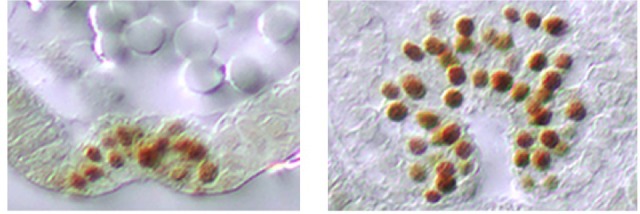
Figure 5—figure supplement 2. Apical constrictions during mesoderm internalization.
Figure 5—figure supplement 3. Delayed mesoderm internalization upon Tc-fog KD.
Figure 5—figure supplement 4. The mesoderm is internalized upon Tc-fog KD.
Due to the dynamic nature of mesoderm internalization, it was important for us to compare carefully stage matched control and KD embryos. To do this, we carried out timed embryo collections and, in addition, examined the number of segments specified in these embryos (via analysis of Tc-gsb expression, Davis et al., 2001). At 19–21 hr after egg lay (AEL) (at 25°C), control embryos had four trunk Tc-gsb stripes and had completely internalized their mesoderm (Figure 5B and E). Tc-fog KD embryos of the same age also had four trunk Tc-gsb stripes, and while some mesodermal cells exhibited apical constrictions, gastrulation was not complete (Figure 5C and F; Figure 5—figure supplements 2 and 3B,D). In anterior positions, gastrulation in KD embryos looked similar to 16–18 hr old control embryos (Figure 5A1, C1), while in middle and posterior regions, KD embryos showed a shallower furrow than that of control embryos at corresponding anterior-posterior (AP) positions (Figure 5D,F).
Despite the delay of mesoderm morphogenesis and the reduction in furrow depth in posterior positions, Tc-fog KD embryos eventually internalized the mesoderm. As in control embryos undergoing germ band extension, KD embryos possessed segmental clusters of Tc-twi expressing cells localized on the basal side of the ectoderm (Figure 5G,H; Figure 5—figure supplement 4). This situation is similar to Drosophila, where loss of fog affects coordination and speed of ventral furrow formation, but does not prevent mesoderm internalization.
Regulation of Tc-fog and Tc-mist expression
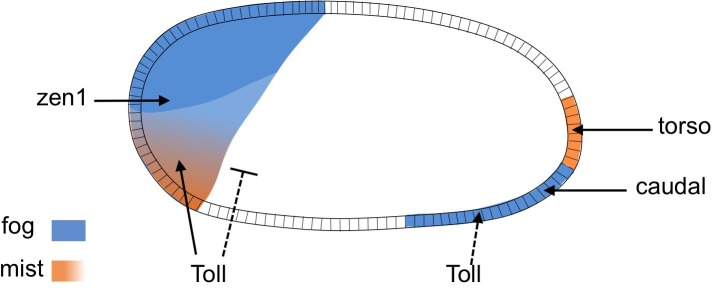
We next investigated how Fog signaling is regulated in Tribolium. Like in Drosophila, ventral tissue specification in Tribolium depends on Toll signaling; Tc-Toll KD leads to a complete loss of mesoderm and ventral ectoderm fates (Moussian and Roth, 2005; Nunes da Fonseca et al., 2008; Roth et al., 1989). Therefore, we reasoned that the ventral stripe of Tc-fog expression is likely also dependent on Toll signaling. Indeed, pRNAi for Tc-Toll resulted in the loss of ventral Tc-fog expression (Figure 6B). Tc-mist expression, on the other hand, remained in the primitive pit region (Figure 6I). The serosa expression of each gene was also affected by Tc-Toll KD; Tc-fog became expressed in an expanded, DV symmetric domain, while Tc-mist showed weak uniform expression (Figure 6B,I). These changes reflect the expansion and dorsalization of the serosa upon Tc-Toll KD (Nunes da Fonseca et al., 2008).
Figure 6. Regulation of Tc-fog and Tc-mist expression.
Whole mount ISH for Tc-fog (A–E) and Tc-mist (H–L) expression in wildtype embryos (A, H) and embryos in which DV and AP genes have been knocked down (B-E, I-L; specific KD shown in panels). All embryos are at primitive pit stage except A and C, which are at the early posterior amniotic fold stage. The asterisk in (D) indicates the appearance of weak Tc-fog expression within a posterior-dorsal domain.
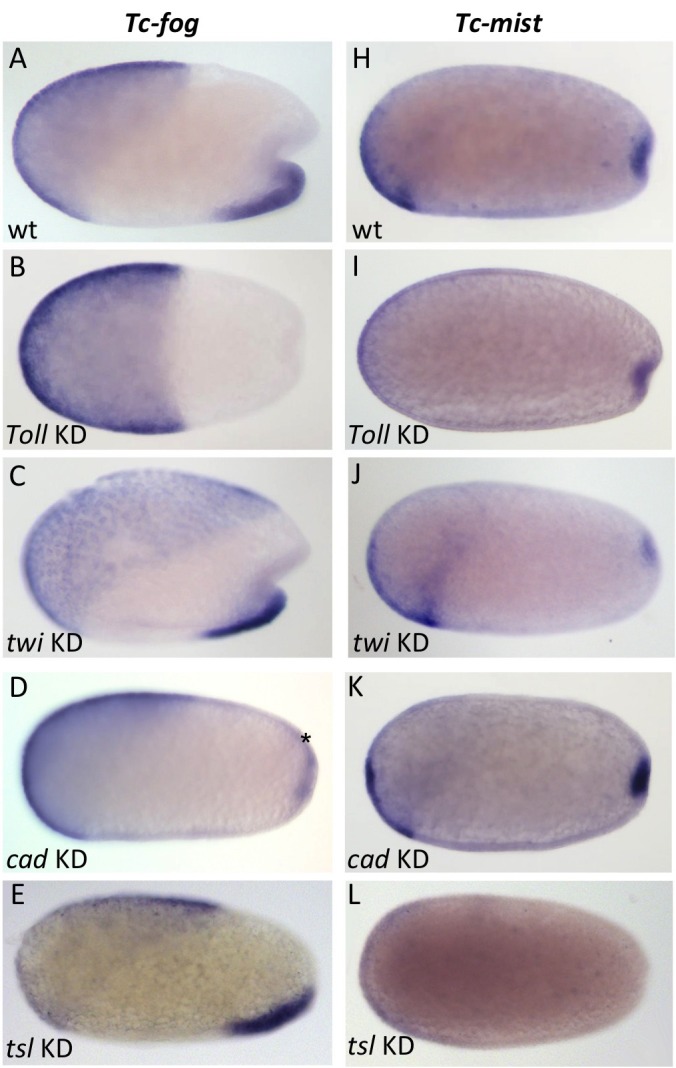
Figure 6—figure supplement 1. Regulation of Tc-fog and Tc-mist by DV and AP patterning genes.
To further dissect the ventral regulation of Tc-fog and Tc-mist, we analyzed the patterning genes downstream of Toll signaling. In both Drosophila and Tribolium, the transcription factors twi and snail (sna) are co-expressed in a ventral stripe (Leptin and Grunewald, 1990; Sommer and Tautz, 1994; Stappert et al., 2016). In Drosophila, both genes are required together to activate mesodermal fog expression (Costa et al., 1994), while sna alone is largely sufficient to activate mesodermal mist expression (Manning et al., 2013). In Tribolium we found that Tc-mist expression was unchanged following Tc-twi pRNAi (as expected based on their non-overlapping domains of expression) (Figure 6J), but Tc-fog was also unaffected (Figure 6C). Mesodermal Tc-sna expression is completely dependent on Tc-twi (von Levetzow, 2008), and, therefore, Tc-fog expression is not regulated by either Tc-twi or Tc-sna. Instead, ventral Tc-fog expression must depend on other zygotic factors in Tribolium.
To analyze the influence of the AP patterning system on Tc-fog and Tc-mist expression, we performed pRNAi for Tc-caudal (Tc-cad) (Copf et al., 2004; Schoppmeier et al., 2009) and Tc-torso-like (Tc-tsl) (Schoppmeier and Schröder, 2005). In Drosophila, caudal is required for the posterior, but not ventral, domain of fog expression (Wu and Lengyel, 1998). In contrast, KD of Tc-cad resulted in the loss of ventral Tc-fog expression and the appearance of a new domain of expression in the posterior-dorsal region of the embryo (asterisk in Figure 6D). Thus, Tc-cad both activates Tc-fog expression within the mesoderm and inhibits Tc-fog expression at the posterior of the embryo. Expression of Tc-mist was not notably altered after Tc-cad KD (Figure 6K).
Tc-tsl is a component of the terminal patterning system that specifies the anterior and posterior extremities of the AP axis (Schoppmeier and Schröder, 2005; Schroder et al., 2000). KD of Tc-tsl did not significantly affect mesodermal Tc-fog expression, but posterior Tc-mist expression was abolished (Figure 6E and L). This result matches published descriptions of Tc-tsl KD, which describe loss of normal posterior folding (Schoppmeier and Schröder, 2005).
Taken together, our results show that ventral expression of Tc-fog requires a combination of DV patterning (Tc-Toll) and AP patterning (Tc-cad) inputs, while the posterior expression of Tc-mist is controlled by the terminal patterning system (Tc-tsl) (Figure 6—figure supplement 1).
The role of local Tc-fog expression for posterior folding
In Drosophila, the timing and location of fog expression is tightly linked with its function (Bailles et al., 2019; Costa et al., 1994; Lim et al., 2017). In contrast, Tc-fog is highly expressed in the serosa and posterior mesoderm, while its expression is conspicuously absent (or not detectable) from the posterior of the embryo where it is most prominently required, for the initiation of primitive pit and posterior amniotic fold formation. To approach this problem, we analyzed how each Tc-fog expression domain contributes to primitive pit and posterior amniotic fold formation. Specifically, we removed each Tc-fog domain individually or simultaneously and monitored the impact on the morphogenetic movements of the respective embryos.
To start, we deleted the serosal domain of Tc-fog (without affecting the ventral domain; Figure 7—figure supplement 1) by knocking down Tc-zen1 to prevent serosa specification (van der Zee et al., 2005). This treatment had no detectable impact on primitive pit indentation or on the initiation of the posterior amniotic fold (Figure 7—figure supplement 1). During further development, posterior amniotic fold formation fails to achieve full encapsulation of the embryo, but this is likely due to the lack of a serosa in Tc-zen1 KD embryos (Panfilio et al., 2013; van der Zee et al., 2005). However, the presence of a primitive pit and early posterior amniotic fold suggest that serosal Tc-fog expression is not essential for early morphogenetic events at the posterior of the embryo. Therefore, the ventral expression of Tc-fog might provide the source for Fog ligand required for posterior folding.
To test this assumption, we knocked down Tc-Toll to delete the ventral Tc-fog expression domain while maintaining high levels of Tc-fog expression in the serosa. Such embryos nonetheless still form a primitive pit-like indentation, and then a deep rotationally symmetric invagination at the posterior pole (Figure 7A,B). The distinctive appearance of the posterior folded tissue in Tc-Toll KD embryos could represent a secondary morphological consequence of germ rudiment condensation in a rotationally symmetric embryo. To test whether these tissue folds are truly Fog-dependent, we simultaneously knocked down Tc-Toll and Tc-fog. In nearly all double-KD embryos (87%, N = 81), posterior folding was indeed abolished (Figure 7C, Video 8). Thus, Toll KD embryos possess morphogenetic movements corresponding to the primitive pit and posterior amniotic fold of control embryos although they lack detectable Tc-fog expression in the germ rudiment.
Figure 7. Local Tc-fog expression and posterior folding.
Nuclear (DAPI) staining of wildtype (A) and KD (B, C, D; specific KD shown in panels) embryos at the early posterior amniotic fold stage. Anterior is left, ventral is down (where possible to discern).
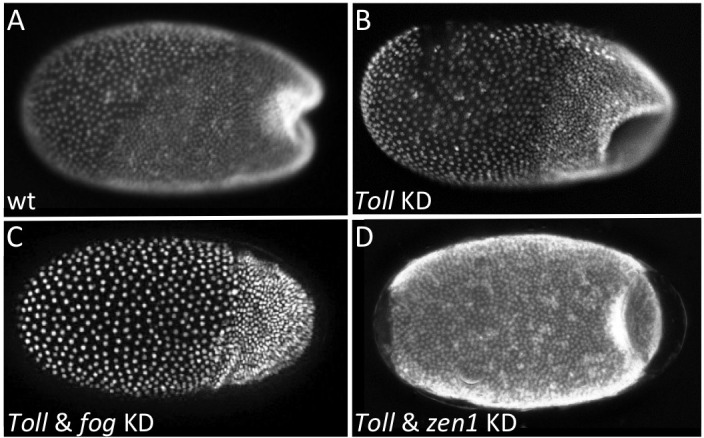
Figure 7—figure supplement 1. Tc-fog expression after Tc-zen1 KD.
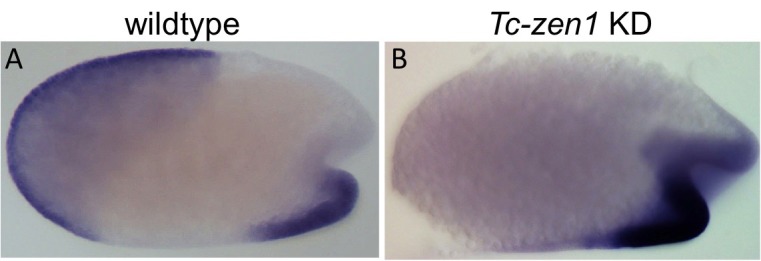
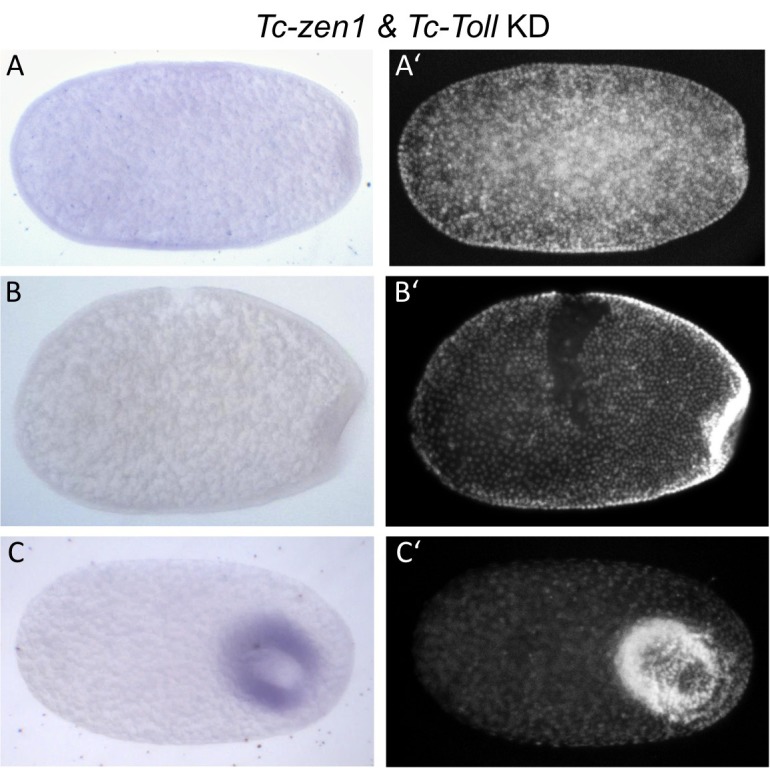
Figure 7—figure supplement 2. Tc-fog expression after Tc-Toll and Tc-zen1 KD.
Video 8. Fluorescent live imaging of Tc-Toll1 and Tc-fog double RNAi nGFP transgenic embryo.
Maximum intensity projection of one egg hemisphere is shown with anterior to the left.
As Tc-Fog is an extracellular ligand, it is possible in the Tc-Toll KD embryos that Tc-Fog protein diffuses from the serosal domain to the posterior of the embryo to activate Tc-Mist and initiate the posterior morphogenetic events. To test this hypothesis, we knocked down both Tc-Toll and Tc-zen1 to remove both domains of Tc-fog expression simultaneously. In such embryos, no Tc-fog expression was visible by RNA ISH (Figure 7—figure supplement 2). Nevertheless, these embryos formed a symmetric posterior invagination as in Tc-Toll single KD embryos (Figure 7D). Therefore, diffusion of Tc-Fog from the serosa domain to the posterior also does not account for posterior folding.
Two possibilities exist to explain this set of results. First, the primitive pit region of Tc-Toll KD embryos may harbor some Tc-fog transcript that, despite its low amounts, is sufficient to trigger the large-scale invagination of a symmetric posterior amniotic fold. Alternatively, there may be sufficient Tc-Fog protein remaining from the maternal expression of Tc-fog (Dönitz et al., 2018) to activate the Fog signaling pathway at the posterior of the embryo. However, both scenarios suggest that small amounts of Tc-Fog are sufficient to trigger large-scale folding specifically at the posterior pole.
A novel role for Fog signaling in serosal spreading
Our finding that Tribolium Fog signaling is involved in mesoderm internalization and posterior amniotic fold formation fits with the classic function of this pathway in apical cell constriction. However, Tc-fog and Tc-mist are also expressed in the serosa, and what function (if any) these genes may have here is unknown. As described earlier, serosal cells become squamous as they spread to encapsulate the germband and yolk, but they also undergo intercalation as occurs during the analogous process of epiboly in zebrafish (Benton, 2014; Köppen et al., 2006). As Fog signaling has been implicated in cell intercalation in the Drosophila germband (Kerridge et al., 2016), we asked whether Tribolium Fog signaling may have the same function in the serosa. We first describe the wildtype pattern of intercalation before testing for a role of Fog signaling in this process.
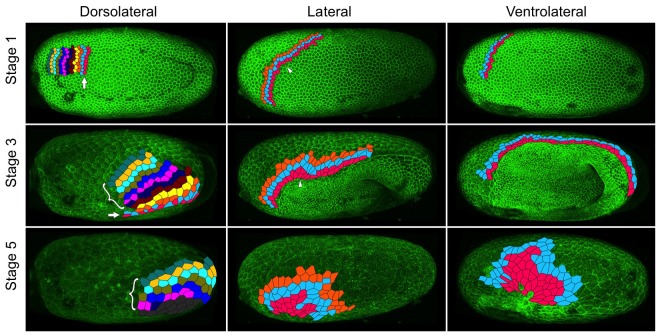
We observed extensive cell intercalation during serosal spreading (Figure 8, Videos 9–11), but the pattern of intercalation varied across the embryo. During the first half of germband formation (stages 1–3), the serosa/germ rudiment boundary increases in length as it moves over the posterior pole. During this period, intercalation occurred at the posterior half of the boundary to increase the number of cells at the boundary (Figure 8 arrow in left panels, Video 9). In contrast, intercalation at more anterior regions caused cells to leave the boundary during the same period (Figure 8 pink cells in middle and right panels, Videos 10–11). During the second half of germband formation (stages 4–5), serosal window closure causes the serosa/germ rudiment boundary to shrink to nothing. During this period, cells left the boundary all along its length (Figure 8 pink cells in middle and right panels, Videos 9–11). Cell intercalation also occurred further away from the boundary during all stages of spreading (Figure 8 bracketed cells in left panel, orange and pale blue cells in middle panel, and pale blue cells in right panel), and we observed the formation of multicellular rosettes (Video 12).
Figure 8. Serosal cells undergo intercalation during serosal expansion.
Stills from confocal live imaging of wildtype embryos with cell membranes marked via transient expression of GAP43-YFP. The tracked cells are colored as rows parallel to the serosa/germ rudiment boundary (pink closest to the boundary). The arrow indicates the region where cells intercalate to increase the number of cells at the serosa/germ rudiment boundary during stages 1–3. The arrowhead indicates one example region where cells intercalate to leave the serosa/germ rudiment boundary during stages 1–3. The bracket indicates cells located away from the serosa/germ rudiment boundary that undergo intercalation during stages 1–5. The dark gray cells in the lower left panel went out of the frame of view and could not be tracked for the full movie. In the right panels, new cells were tracked from when they became visible halfway through embryo formation (middle panel). Cell outlines were manually drawn using projection views of individual timepoints and individual z-sections. All panels show maximum intensity projections of one egg hemisphere. Anterior is left, ventral is down.
Video 9. Fluorescent live imaging of the dorsolateral side of a wildtype embryo transiently expressing GAP43-YFP.
Serosa cells at/near the serosa/germ rudiment boundary were tracked and colored as rows (pink cells closest to the boundary). Only cells that were visible from the beginning of the timelapse are shown. Maximum intensity projection of one egg hemisphere is shown with anterior to the left and ventral to the bottom.
Video 10. Fluorescent live imaging of the lateral side of a wildtype embryo transiently expressing GAP43-YFP.
Serosa cells at/near the serosa/germ rudiment boundary were tracked and colored as rows (pink cells closest to the boundary). Only cells that were visible from the beginning of the timelapse are shown. Maximum intensity projection of one egg hemisphere is shown with anterior to the left and ventral to the bottom.
Video 11. Fluorescent live imaging of the ventrolateral side of a wildtype embryo transiently expressing GAP43-YFP.
Serosa cells at/near the serosa/germ rudiment boundary were tracked and colored as rows (pink cells closest to the boundary). Roughly half the cells (at the anterior) are tracked from the beginning of the timelapse, while the rest are tracked from halfway through embryo formation (at 135 min) when they became visible. Maximum intensity projection of one egg hemisphere is shown with anterior to the left and ventral to the bottom.
Video 12. Fluorescent live imaging of part of the forming serosa in a wildtype embryo transiently expressing GAP43-YFP.
A group of cells intercalating via rosette formation are tracked. The field of view was manually stabilized to follow this group of cells. Anterior is to the left.
To test whether Fog signaling is involved in serosal cell intercalation, we used embryonic RNAi (eRNAi) to partially knock down Tc-fog to a level where phenotypic effects were visible but posterior amniotic fold formation and serosal expansion still occurred. We found that microinjection of Tc-fog dsRNA at 1 µg/µL caused roughly one-third of KD embryos (10 out of 29 embryos) to form a posterior amniotic fold and undergo serosal spreading. Embryos were co-injected with mRNA encoding the membrane marker GAP43-YFP to allow detailed live imaging and cell tracking (Benton et al., 2013). Cell intercalation was quantified by tracking roughly 70–100 cells at or near the serosa/germ rudiment boundary throughout serosa expansion and counting the number of intercalation events between tracked cells in four stage- and orientation-matched embryos (Video 13). Control embryos (injected with GAP43-YFP mRNA alone) showed 2.14 intercalation events per cell (171 cells tracked in two embryos) while Tc-fog partial KD embryos showed 2.56 intercalation events per cell (163 cells tracked in two embryos). This analysis shows that rather than decreasing intercalation in the serosa, reduction of Fog signaling led to a measurable increase in the relative amount of intercalation during serosal spreading.
Video 13. Fluorescent live imaging of the wildtype and Tc-fog weak KD embryos transiently expressing GAP43-YFP.
Serosa cells at/near the serosa/germ rudiment boundary were tracked. The top panels show cells coloured as rows (pink cells closest to the boundary). The bottom panels show cells colored randomly and intercalation events between tracked cells are shown with white triangles. Maximum intensity projection of one egg hemisphere is shown with anterior to the left and ventral to the bottom.
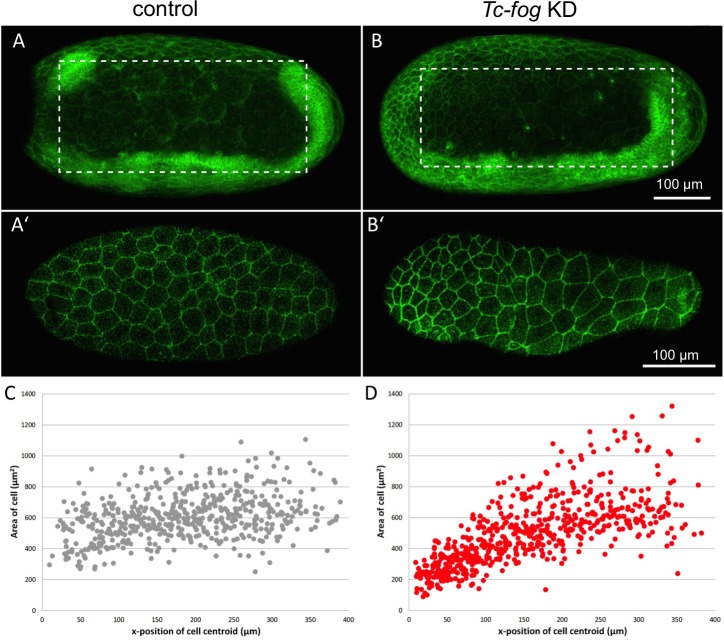
A second possible role for Fog signaling in the serosa is an involvement in the cuboidal-to-squamous cell shape change that occurs during serosal spreading (Benton et al., 2013; Handel et al., 2000). To test this possibility, we quantified the extent of serosal cell spreading in Tc-fog partial KD embryos (like those described above). In such embryos, serosal cells still became squamous but differences in the extent of flattening existed at anterior and posterior positions (Video 14). The highly attenuated nature of these cells prevented quantification of cell height/volume, so we measured apical cell area after the completion of cell spreading. In control embryos (injected with GAP43-YFP mRNA alone), serosal cell area was variable, but this variability was evenly distributed along the AP axis (σ = 145.96 µm2; n = 535 cells across eight embryos; Figure 9A,C). In Tc-fog partial KD embryos, the variability in final serosal cell area was significantly increased compared with control embryos (p<0.001 Fisher’s F test, σ = 215.9 µm2; n = 578 cells across seven embryos; Figure 9B,D). In addition, the serosal cells that covered the posterior half of the egg had larger surface areas than those in comparable positions in control embryos, while serosal cells in anterior regions showed the opposite pattern (Figure 9B,D). Based on our findings, we conclude that Tribolium Fog signaling has a novel role in serosal cells to coordinate the cuboidal-to-squamous cell shape transition that results in a uniformly thin layer of serosal cells surrounding the entire yolk and embryo.
Figure 9. Fog signaling affects serosal expansion.
(A, B) Stills from confocal live imaging of wildtype (A) and Tc-fog eRNAi (B) embryos with cell membranes marked via transient expression of GAP43-YFP. Embryos are undergoing germband elongation. (A’, B’) Single optical section of the serosa from the dashed box region in (A, B) showing cells whose areas were measured. Quantification was performed on single optical section to avoid artefacts caused by curvature of the egg. (C, D) scatter plots showing serosa cell areas (wildtype (gray): 535 cells across eight embryos, Tc-fog eRNAi (red): 578 cells across seven embryos) together with AP position of cell centroids. Measurements were performed manually. (A, B) are average intensity projections of one egg hemisphere. Anterior is left, ventral is down.
Video 14. Fluorescent live imaging of a Tc-fog weak KD embryo transiently expressing GAP43-YFP.
Maximum intensity projection of one egg hemisphere with anterior to the left and ventral to the bottom is shown in the center. A transverse section near the anterior pole is to the left, a transverse section near the posterior pole is to the right, a sagittal section along the middle of the egg is shown to the bottom.
T48 enhances Fog signaling in Tribolium
In Drosophila, fog and T48 both function during ventral furrow formation (Kölsch et al., 2007). We identified a single homolog of T48 in Tribolium (hereafter referred to as Tc-T48), and while we could not detect localized Tc-T48 expression by ISH, RNA-sequencing data suggested it is weakly expressed in embryos (Dönitz et al., 2018). Therefore, we tested whether Tc-T48 has an embryo-wide enhancement function on Fog signaling in Tribolium.
To test for such a Tc-T48 function, we microinjected embryos with Tc-fog dsRNA at 1 µg/µL (to partially KD Tc-fog) together with Tc-T48 dsRNA. As described above, roughly a third of embryos microinjected with Tc-fog double-stranded RNA (dsRNA) (at 1 µg/µL) alone still formed a posterior amniotic fold and underwent serosa spreading (Video 15). When embryos were injected with both Tc-fog dsRNA and Tc-T48 dsRNA (either by co-injection or sequential injections; n = 20 and 10, respectively) all embryos failed to form a posterior amniotic fold (Video 15). However, the double KD embryos did not show a more severe mesoderm internalization phenotype than the Tc-fog single KD embryos. Microinjection of Tc-T48 dsRNA alone had no detectable effect on development (n = 10).
Video 15. Fluorescent live imaging of wildtype and Tc-fog, Tc-T48, and Tc-fog and Tc-T48 double.
RNAi embryos transiently expressing GAP43-YFP. Average intensity projections of one egg hemisphere are shown with anterior to the left and ventral to the bottom.
The statistically significant difference (p<0.001, Chi-Square test) in the phenotype caused by Tc-fog eRNAi alone versus Tc-fog and TcT48 double RNAi, shows that Tc-T48 has a morphogenetic function in Tribolium. Given the apparent lack of localized Tc-T48 expression, we suggest that low levels of uniform expression play an embryo-wide role in enhancing Fog signaling.
Fog signaling is required for Tribolium blastoderm formation
In the course of analyzing the role of Fog signaling by embryonic RNAi, we injected different concentrations of dsRNA to vary the KD strength. While we recovered the phenotypes observed by parental RNAi with embryonic injections of 1 µg/µL dsRNA, injection of 3 µg/µL dsRNA yielded a phenotype that we had not obtained from pRNAi.
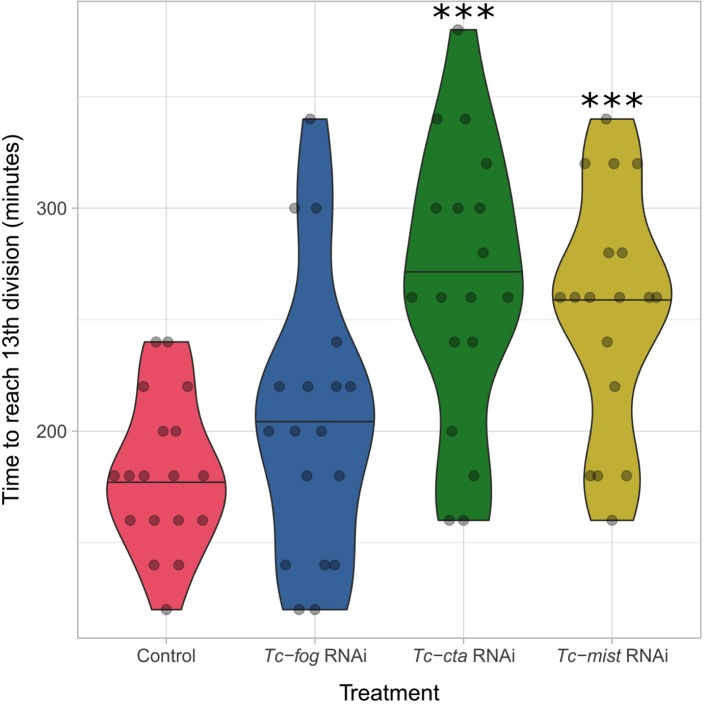
In the majority of KD embryos, major blastoderm-wide defects occurred during or prior to embryo condensation: 70% in Tc-cta KD, 70% in Tc-mist KD, 40% in Tc-fog KD versus 10% in control (n = 20 for each condition). Defects were highly variable in each KD and ranged from gaps in the blastoderm that became greatly enlarged during condensation to complete disintegration of the blastoderm prior to or during condensation (Figure 10B–D; Videos 16 and 17). In addition to the visible morphological defects, there was also a statistically significant delay in the development of Tc-cta and Tc-mist KD embryos (as measured using division 13 as a temporal landmark; Figure 10—figure supplement 1). This delay was not observed in Tc-fog KD embryos, which also had the lowest proportion of embryos with other blastoderm defects.
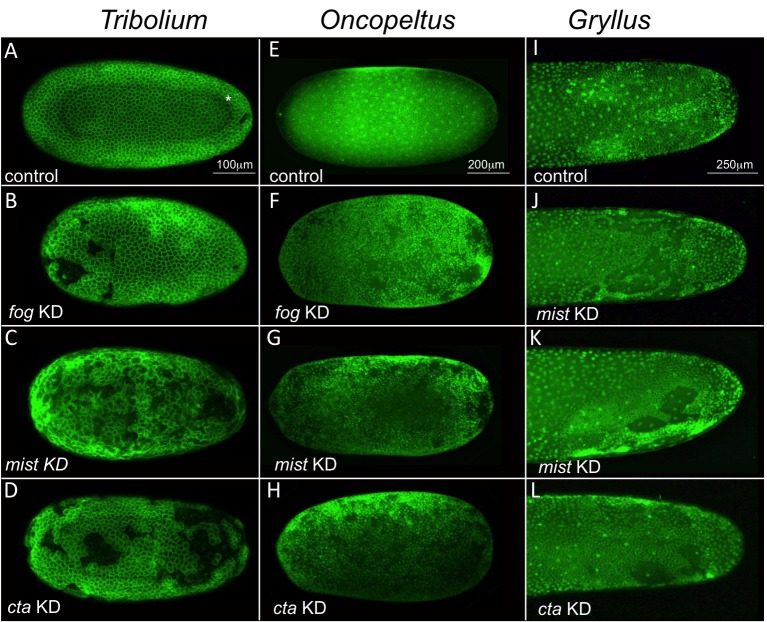
Figure 10. Fog signaling is required for blastoderm formation in Tribolium, Oncopeltus and Gryllus.
(A–D) Stills from confocal live imaging of wildtype (A), and Tc-fog, Tc-mist, Tc-cta eRNAi (B–D) Tribolium embryos with cell membranes marked via transient expression of GAP43-YFP. Embryos are at late blastoderm stage. White asterisk in (A) indicated hole within the blastoderm which later closes. (E–H) O. fasciatus blastoderm stage wildtype (E), and Of-fog, Of-mist, Of-cta pRNAi (F–G) embryos with nuclei stained (Sytox) to reveal their distribution. (I–L) Stills from fluorescent live imaging of control (I), and Gb-mist, Gb-cta pRNAi (J–L) G. bimaculatus embryos with nuclei labeled via a histone2B-eGFP transgene. The phenotype of Gb-mist pRNAi is stronger in (J) than in (K). The latter embryo recovered during later development. (A–D) are average intensity projections of one egg hemisphere, (I–L) are maximum focus projections of one egg hemisphere. Anterior is left for all embryos. (I, J, L) are ventral views, (K) is a ventrolateral view with ventral down.
Figure 10—figure supplement 1. Developmental delay upon KD of Fog pathway components.
Video 16. Fluorescent live imaging of wildtype and Tc-fog, Tc-cta, and Tc-mist RNAi embryos transiently expressing GAP43-YFP.
Average intensity projections of one egg hemisphere are shown with anterior to the left and ventral to the bottom (where possible to discern).
Video 17. Fluorescent live imaging of additional Tc-fog, Tc-cta, and Tc-mist RNAi embryos transiently expressing GAP43-YFP.
Further examples of blastoderm formation defects to demonstrate the variability in the phenotypes. Average intensity projections of one egg hemisphere are shown with anterior to the left and ventral to the bottom (where possible to discern).
These phenotypes are unlikely to be artifacts caused by embryo handling or microinjection as they were seen at different proportions upon KD of each of the genes and were never seen at such high rates in control injections. As such, components of the Fog signaling pathway must also function during the formation of the blastoderm cell layer in Tribolium.
The blastoderm function of Fog signaling is widely conserved
After finding that Fog signaling has key morphogenetic functions during embryonic development in Tribolium, we asked whether such functions are widely conserved in insects. To answer this question, we functionally analyzed Fog signaling pathway components in two distantly related (Misof et al., 2014) hemimetabolous insects: the milkweed bug Oncopeltus fasciatus and the cricket Gryllus bimaculatus.
We identified single orthologs for cta, mist and fog in both species (hereafter called Of-fog, Of-mist, Of-cta, and Gb-fog, Gb-mist, Gb-cta, Figure 1—figure supplement 2). KD of each of these genes via pRNAi was performed in Oncopeltus and in Gryllus (except for Gb-fog, the KD of which resulted in adult lethality) and led to highly penetrant early phenotypes in both species. While control embryos (from parental injections of GFP dsRNA [Oncopeltus] or buffer [Gryllus]) formed a uniform blastoderm layer (n = 29 for Oncopeltus, n = 15 for Gryllus), each KD resulted in blastoderms that were interrupted by large holes along the entire AP axis (Oncopeltus: Figure 10F–H, 65% in Of-fog KD [n = 29], 64% in Of-mist KD [n = 25], 88% in Of-cta KD [n = 26]; Gryllus: Figure 10J–L and Video 18, 100% in Gb-mist, and Gb-cta KD [n = 19 each]).
Video 18. Fluorescent live imaging of wildtype and Gb-mist RNAi histone2B-EGFP transgenic embryos.
Maximum focus projections of one egg hemisphere are shown as ventral views with anterior to left.
While these early blastoderm defects prevented us from studying Fog function during later development in Oncopeltus and Gryllus, these phenotypes show that Fog signaling components have a deeply conserved requirement during blastoderm formation in insects.
Discussion
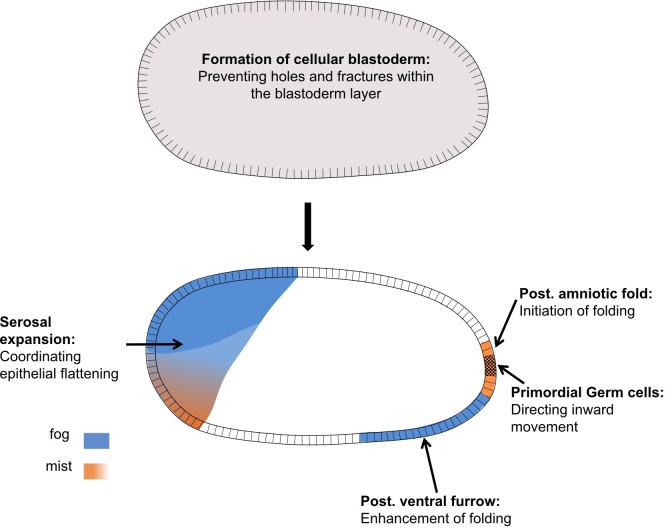
In this article we have shown that Fog signaling plays major morphogenetic roles during embryogenesis in the beetle Tribolium (Figures 3 and 11). Disruption of this pathway leads to severe defects during germband formation, including a complete loss of posterior amniotic fold formation, delayed mesoderm internalization, and mislocalization of PGCs. Fog signaling was also involved in the cuboidal-to-squamous cell shape change that occurs as the serosa spreads to cover the whole surface of the egg. Lastly, we found Tribolium Fog signaling to function during earlier stages of development: disruption of this pathway led to defects in the formation of a regular, continuous blastoderm epithelium. Functional analysis of Fog signaling in two distantly related insect species revealed this early blastoderm function to be widely conserved. In this discussion, we break down these diverse roles and discuss their importance for our understanding of the evolution of insect embryogenesis.
Figure 11. Distinct functions of Fog signaling in Tribolium.
Summary schematic showing the different roles of fog signaling during early Tribolium embryogenesis.
Fog signaling has local morphogenetic functions during gastrulation
Fog signaling was discovered for its functions during early morphogenesis in Drosophila (Zusman and Wieschaus, 1985; Sweeton et al., 1991), but doubts were raised about whether this pathway functions during early embryogenesis in other insects (Goltsev et al., 2007; Sweeton et al., 1991; Urbansky et al., 2016; Zusman and Wieschaus, 1985). Here, we have shown that Fog signaling also functions during early development in a beetle, and that disruption of this pathway causes severe embryo-wide defects.
The most severe effect caused by disruption of Fog signaling in Drosophila is the loss of posterior gut fold formation (Costa et al., 1994; Seher et al., 2007; Sweeton et al., 1991). This posterior folding event in Drosophila not only internalizes the posterior endoderm (the posterior midgut proper), but also the hindgut (proctodeum), and it is required for the correct dorsal and anteriorwards extension of the germband. Therefore, it has been named amnioproctodeal invagination (Campos-Ortega and Hartenstein, 1997). Tribolium Fog signaling is also required for a folding event at the posterior of the blastoderm. However, since gut differentiation in Tribolium takes place only after the fully segmented germband has formed, it is not known how many cells involved in this folding event will later contribute to the gut (Stanley and Grundmann, 1970; Berns et al., 2008). Classical morphological descriptions as well as molecular work, though, suggest that posterior midgut and proctodeum are derived from a region close to or encompassing the site of primitive pit formation (Anderson, 1972b; Benton, 2018; Berns et al., 2008; Handel et al., 2000; Johannsen and Butt, 1941; Kispert et al., 1994; Stanley and Grundmann, 1970; Ullmann, 1964). Indeed, one late phenotype of Tribolium embryos lacking Fog signaling concerns the hindgut, which points in the wrong direction, suggesting that Tribolium Fog is required for orienting the gut invagination towards the inside (Figure 2—figure supplement 5).
Although the role of the posterior amniotic fold for gut development in Tribolium needs further clarification, this fold has obvious consequences for early embryo topology: it causes the amnion primordium/dorsal ectoderm to cover the ventral side of the embryo to form the amniotic cavity (Handel et al., 2000). Upon Fog signaling disruption and loss of the posterior amniotic fold, most of the germ rudiment tissue remains in an open configuration (Figures 2 and 3). This defective topology is reminiscent to that of wildtype embryos of Drosophila and other (cyclorrhaphan) dipteran species that do not become covered by an amnion-like tissue (Schmidt-Ott, 2000). As such, reduction/loss of early posterior Fog signaling may have contributed to evolution of the Drosophila-like mode of development. To address this question, more detailed descriptions of the genetic and morphogenetic events occurring during posterior development in other insect species are required.
The mechanisms of Fog signaling at the posterior pole in Tribolium pose an interesting riddle. While Tc-mist is expressed in the region where posterior folding is initiated, this region lacks detectable Tc-fog expression (Figure 1; Figure 6A,H and Figure 6—figure supplement 1). The area closest to the primitive pit that harbors Tc-fog expression is the ventrally abutting mesoderm (Figure 1—figure supplement 4). However, even if mesodermal Tc-fog expression is removed, like in Tc-Toll KD embryos, posterior folding still takes place, suggesting that expression levels undetectable by our methods are sufficient to induce folding (Figure 7). This is in apparent contradiction to the extent of posterior folding, which results in a large-scale tissue re-arrangement. It is therefore possible that posterior folding in Tribolium involves some form of mechanical feedback amplification through which the folding event is less sensitive to the initial amount of Fog signaling, as was recently shown for Drosophila (Bailles et al., 2019).
Posterior folding in Drosophila is also driven by a cycle of cell deformations that require integrin-mediated cell adhesion to the inner eggshell (vitelline membrane) (Bailles et al., 2019). Such integrin-mediated anchoring was first described in Tribolium, where anterior anchoring exists during germband formation (Münster et al., 2019). The same integrin that is required for the anterior attachment point (PS2, Tc-inflated) in Tribolium is also expressed at the posterior pole (Münster et al., 2019) suggesting a further similarity between Drosophila and Tribolium posterior folding events. However, any attachment at the posterior pole in Tribolium must be transient, as our live imaging did not reveal a static posterior attachment point as we previously observed at the anterior using the same approach (Benton et al., 2013).
The second major role of Fog signaling in Drosophila is during mesoderm infolding, and this function also appears to be conserved in Tribolium. Disruption of Fog signaling has very similar consequences for mesoderm internalization in Tribolium and Drosophila. In both cases, the mechanism and timing of mesoderm internalization is affected by loss of signaling, but the mesoderm is still able to internalize (Seher et al., 2007; Zusman and Wieschaus, 1985). The same is also true for the dipteran Chironomus, where disruption of Fog signaling has a measurable impact on mesoderm internalization but the pathway is not strictly required for the process (Urbansky et al., 2016).
Further evidence for conservation of Fog signaling function in mesoderm internalization comes from the cell shape changes caused by Fog signaling. While anterior regions of the Tribolium mesoderm do not express Tc-fog and internalize by forming only a shallow furrow, the posterior half of the mesoderm expresses Tc-fog and does form a deep furrow during internalization (Figure 1M–P; Figure 5A and Figure 5—figure supplement 1; Figures 3 and 11) (Handel et al., 2005). This result suggests that mesodermal cells that experience high levels of Fog signaling enhance furrow formation, while those that may experience lower signaling (i.e. via diffusion of Fog ligand from neighbouring cells) do not. This hypothesis is also supported by research on Fog signaling in Chironomus and Drosophila. In Chironomus, the mesoderm forms a shallow furrow during internalization, and while fog is expressed in this tissue, expression is notably weaker in the ventralmost part of the domain. Experimental over-activation of Fog signaling triggers the formation of a deep ventral furrow (Urbansky et al., 2016), suggesting a quantitative response to levels of Fog ligand. In Drosophila, quantitative analyses have shown that the accumulation of Fog ligand directly correlates with the degree of change in the cytoskeleton required for cell shape changes (Lim et al., 2017).
An interesting difference between Drosophila and Tribolium consists of the function of T48. While mesodermal expression of T48 contributes to ventral furrow formation in Drosophila (Kölsch et al., 2007), Tc-T48 lacks local expression. Double KD with Tc-fog did not block mesoderm internalization, but enhanced the posterior folding phenotype. Thus, instead of the local mesoderm-specific function of T48 in Drosophila we suggest that Tc-T48 has a weak embryo-wide function, which we could only detect at the posterior pole.
Taken together posterior folding and mesoderm internalization show different requirements for Fog signaling in Tribolium, as is also true in Drosophila (Figure 11). Posterior folding is absolutely dependent on Fog, although apparently low levels of signaling are sufficient to induce massive folding. On the other hand mesoderm internalization is not strictly dependent on Fog, which rather acts quantitatively to modulate the depth of the invagination furrow along the anterior-posterior axis.
The role of Fog signaling in primordial germ cell positioning
One function for Tribolium Fog signaling that does not exist in Drosophila is the role in PGC positioning. We found that disruption of Fog signaling leads to Tribolium PGCs moving to the apical surface of the embryonic epithelium rather than being internalized basally (Figures 3, 4 and 11). This new aberrant localization is comparable to the positioning of Drosophila PGCs (the pole cells) at the apical surface of the blastoderm (Cinalli and Lehmann, 2013).
We propose two possible scenarios for Fog’s role in PGC internalization in Tribolium. One possibility is that PGC localization is due to a requirement for Fog signaling within the epithelial cells surrounding the PGCs. For instance, Fog-mediated apical constrictions of posterior blastoderm cells could bias the movement of PGCs to the basal side of the epithelium. When Fog signaling is disrupted, PGCs would carry out their normal developmental program and leave the epithelium, but the absence of apical constriction and primitive pit formation would cause the PGCs to localize to the apical side of the blastoderm. Alternatively, Fog signaling may directly control cell polarity within the PGCs, and it is the breakdown of this process that affects PGC localization. In addition, some combination of both sides of Fog activity may be true. Given that we observe a well-formed, albeit wrongly positioned, cluster of PGCs after loss of Fog signaling, it seems more likely that Fog signaling acts after PGC formation and without impairing cellular organization within the cluster.
Despite the lack of overt conservation of this PGC function, research in Drosophila does reveal a possibility as to how Fog signaling could be affecting Tribolium PGC development. In Drosophila, the GPCR Trapped in endoderm 1 (Tre1) is necessary in PGCs for their migration through the midgut epithelium (LeBlanc and Lehmann, 2017). Tre1 is activated by guidance cues and promotes germ cell migration by polarizing Rho1. In Tribolium, Fog signaling could potentially also act to polarize Rho1 via RhoGEF2 recruitment within PGCs and thereby effect their migration to the basal side of the epithelium.
Fog signaling has tissue-wide functions in the blastoderm and serosa
The functions for Tribolium Fog signaling discussed above fit with the traditional role for this pathway in apical constriction. In contrast, the involvement of Fog signaling in serosal spreading and blastoderm formation are two processes that do not involve apical constriction (Figure 11).
During serosal spreading, Fog signaling acts in a process that is effectively the opposite of apical constriction: the expansion of the apical (and basal) cell surface to cause the cuboidal-to-squamous transition. To analyse this function, partial KD of Fog was most informative, as posterior folding and serosal spreading still occurred, but were no longer uniform throughout the tissue. Rather, cells closest to the dorsal serosa/germ rudiment border acquired greater apical surface areas, while the remaining serosa cells in fact had reduced surface areas compared to wildtype cells at corresponding positions (Figure 9).
Tissue forces have not been directly measured in Tribolium, but indirect evidence indicates that folding and condensation of the germ rudiment exert pulling forces on the serosa (Münster et al., 2019). If true, these forces would likely be greatest at the dorsal serosa/germ rudiment border. Despite this tissue-level anisotropy, spreading of serosal cells appears fairly uniform in wildtype embryos. This uniformity may in part be due to the serosal cell intercalation we describe (Figure 8). Such intercalation events could be triggered by local tension in the epithelium as described in other systems (e.g. Aigouy et al., 2010) and could thereby serve to maintain tissue fluidity and reduce anisotropy in tissue tension (Tetley and Mao, 2018; Tetley et al., 2019). A recent preprint examines this process in further detail (Jain et al., 2019).
We found that reduced Fog signaling caused an increase in the relative number of intercalation events and decreased uniformity in serosal cell spreading (Figure 9). The increase in intercalation could be due to increased anisotropy in epithelial tension caused by the defects in serosal cell spreading. Thus, the role of Fog signaling in the serosa could be to regulate the cuboidal-to-squamous cell shape transition to evenly spread the propagation of forces between neighbouring serosal cells. The use of a paracrine signaling pathway such as Fog signaling for this function makes sense, as it would allow tissue-wide coarse-graining via the extracellular distribution of ligand, buffering the degree to which cells experience different forces across the tissue. At the intracellular level, Fog signaling could be influencing the distribution of myosin to affect cell spreading. As the mechanisms underlying cuboidal-to-squamous cell shape transitions are not well understood (Brigaud et al., 2015; Grammont, 2007; Wang and Riechmann, 2007), and only descriptive (Benton et al., 2013; Panfilio and Roth, 2010), but no mechanistic information on serosal spreading exists, substantial future work will be required to uncover the mechanism of Fog signaling during this important and widely conserved developmental event.
Fog signaling also appears to have a global tissue function during blastoderm formation in Tribolium (Figure 11) and two other species representing deep branches within the insects. In Tribolium we observed this phenotype only after embryonic injections of high amounts of dsRNA. Upon parental RNAi, which should affect both maternal and zygotic transcripts, cellularization was normal. However, parental KD also led to partial lethality and sterility of the injected individuals (e.g. for Tc-mist the survival rate was 77% (N = 200) and the surviving females produced a reduced number of eggs for only three days before becoming sterile). It is therefore likely that the eggs obtained following pRNAi represented an incomplete KD, despite the fact that the phenotypes we observed were highly penetrant (Figure 2—figure supplement 4). Embryonic RNAi circumvents the problem of adult lethality and sterility and apparently allows a more efficient KD of maternal and zygotic transcripts of Fog pathway components. In Drosophila problems with adult lethality and (certain types of) sterility can be overcome by producing mutant germ line clones. Using this technique Drosophila embryos were generated that completely lacked maternal and zygotic fog transcripts. However, these embryos did not show a stronger phenotype than embryos lacking only the zygotic transcripts (Costa et al., 1994; Zusman and Wieschaus, 1985). In particular, no defects during cellularization were observed.
The different requirement for Fog signaling during cellularization in Drosophila and three insects we have studied may be linked to variations in the modes of cellularization. The formation of a high-columnar blastoderm, which has been well studied in Drosophila, is an exception among insects and even among flies (Bullock et al., 2004; van der Zee et al., 2015). In many insects, cellularization generates a blastoderm of cuboidal cells, while some insects initially form individual cells that then migrate to form a continuous epithelium (Ho et al., 1997; Nakamura et al., 2010).
Little is known about the molecular mechanisms underlying the diverse modes of cellularization described above, but each mode will have its own mechanical requirements. For example, in Drosophila strong lateral adhesion between the highly columnar cells provides stability (Mazumdar and Mazumdar, 2002), while in Tribolium transitory holes and fractures between protocells are visible within the wildtype blastoderm (white asterix in Figure 10). Here, Tribolium Fog signaling may have a non-cell autonomous influence on actomyosin dynamics, increasing the stability and robustness of the epithelium to prevent fracture formation or heal existing fractures. However, the way in which this may occur at a mechanistic level is a completely open question. The discovery of special Innexin-based cell junctions that are essential for cellularization in Tribolium, but do not exist in Drosophila, highlights the potential diversity in mechanisms underlying cellularization in different insects (van der Zee et al., 2015). Thus, as with the spreading of the serosa, future work is required to uncover the mechanism of Fog signaling during blastoderm formation in Tribolium and other insects.
While the mechanism remains unknown, the requirement for Fog signaling during blastoderm formation in phylogenetically diverse insects suggests this to be a widely conserved function. Finally, while gastrulation is even more variable in non-insect arthropods than in insects, nearly all described arthropod species also form a cellular blastoderm that at least partially covers the surface of the egg (Anderson, 1973; Wanninger, 2015a; Wanninger, 2015b). As such, the early function of Fog signaling during blastoderm formation may be the most ancestral role of this pathway in arthropods.
Materials and methods
Strains
Tribolium castaneum strains: San Bernandino wildtype (Brown et al., 2009), nuclear GFP (nGFP) (Sarrazin et al., 2012), LifeAct-GFP (van Drongelen et al., 2018) were cultured as described (Brown et al., 2009). Oncopeltus fasciatus was cultured as described (Ewen-Campen et al., 2011). Gryllus bimaculatus wildtype strain (Donoughe and Extavour, 2016) and pXLBGact Histone2B:eGFP (Nakamura et al., 2010) was kept as described (Donoughe and Extavour, 2016).
cDNA cloning
The primers used for in-situ hybridization and dsRNA synthesis were designed by using the new T. castaneum genome assembly (Dönitz et al., 2018). Primer design, RNA extraction and cDNA synthesis were carried out using standard protocols. For Gryllus bimaculatus, a new assembled transcriptome was used to design primers for Gb-cta and Gb-mist. The Advantage GC 2 PCR Kit (Takara) was used for gene cloning. All relevant genes, their corresponding Tc-identifiers and the primers used in this work are listed in the supplement (Supplementary file 1).
dsRNA synthesis, parental and embryonic
RNAi dsRNA preparation, pupae and adult injections followed Posnien et al. (2009). Embryonic RNAi was performed as described (Benton et al., 2013). For Gryllus bimaculatus, a 1184 bp fragment of Gb-cta and a 967 bp fragment of Gb-mist was used to knock down gene function. For both genes, the knockdown was performed in two independent experiments injecting 7 µg and 10 µg of dsRNA per animal, respectively. The dsRNA solution was injected into the proximal joint of the coxa of the second and third leg. For each experiment, four adult females of the pXLBGact Histone2B:eGFP line were injected and embryos of the second egg lay (collected about one week after injection) were analyzed via live imaging.
In-situ hybridisation, immunohistochemistry
Single and double ISH were performed essentially as described (Schinko et al., 2009). For staining of cell membranes, Alexa Fluor 555/568 Phalloidin (Molecular Probes, life technologies) was used. Nuclear counterstaining was performed using DAPI (Invitrogen) or Sytox Green (Thermo Fisher) as previously described (Nunes da Fonseca et al., 2008).
Cryosections
Embryos were embedded in a melted agarose/sucrose solution (2% agarose, 15% sucrose in PBS). After the agarose cooled down, blocks of agarose containing the embryos were cut and incubated in a solution of 15% sucrose in PBS overnight. The blocks were fixed to the specimen block using Tissue-Tek O.C.T. (Sakura). After shock freezing in −80°C isopentan, the blocks were transferred to a Cyrostat (Leica CM 1850) and sliced at −20°C (30μm-thick sections). The sections were mounted on Superfrost Ultra Plus microscope slides (Thermo Scientific) and dried over night at RT. Phalloidin and Sytox staining directly on the sections was performed following the standard protocols using a humidity chamber.
Live imaging
Confocal time-lapse imaging of Tribolium embryos injected with GAP43-YFP mRNA was performed as previously described (Benton et al., 2013; Benton, 2018) at 25–32°C at time intervals from 2 to 10 min between timepoints using 20x, 40x, or 63x objectives. For live imaging of the posterior poles, eggs were propped up vertically (resting against another egg for stability) on a glass-bottomed Petri dish (MatTek) with their posterior against the glass. Live imaging transgenic nuclear GFP (nGFP) (Sarrazin et al., 2012) or LifeAct-GFP (van Drongelen et al., 2018) embryos was done at room temperature using the Zeiss AxioImager.Z2 in combination with an Apotome.2 and movable stage (Zen2 Blue).
For imaging Gryllus bimaculatus embryos we used a Zeiss AxioZoom.V16, equipped with a movable stage. Gryllus embryos were placed on 1.5% agarose and were covered with Voltalef H10S oil (Sigma). Imaging was performed at 25–27°C.
Data analysis and software
Image analysis was performed in FIJI (Schindelin et al., 2012). Serosa cell areas were measured manually and cells at the periphery of the section were not included. Cell tracking and quantification of cell intercalation was performed manually on 4D hyperstacks and various projections (maximum/average intensity, sum slices) using mTrackJ (Meijering et al., 2012). Confocal data were rotated using TransformJ with quintic B-spline interpolation (Meijering et al., 2001). Additional plugins include Bioformats Importer (Linkert et al., 2010), Image Stabiliser (Li, 2008), StackReg (Thevenaz et al., 1998), and Bleach Correction (Miura et al., 2014).
Box/violin plots were generated using PlotsOfData (Postma and Goedhart, 2019). Figures and schematics were created using Powerpoint and the open source software Inkscape.
Acknowledgements
We thank Nathan Kenny for assembling the Gryllus bimaculatus transcriptome, S Noji and T Mito for providing Gryllus strains. NF, KHC and SR were supported by DFG CRC 680, MAB by a Humboldt Fellowship for Postdoctoral Researchers and DFG Research Fellowship 407643416, RNdF and CvL by the International Graduate School in Genetics and Functional Genomics, Cologne University. Research in the R NdF lab is supported by the following Brazilian agencies: CNPq, FAPERJ and CAPES. MP was supported by a UoC Postdoc Grant, MSH by DFG FOR 1234, DS by a Ph.D. fellowship of the Boehringer Ingelheim Fonds, KAP by the Emmy Noether Program of the DFG (grant PA 2044/1-1), JAL was supported by grant R03 HD078578 from the US National Institutes of Health.
Funding Statement
The funders had no role in study design, data collection and interpretation, or the decision to submit the work for publication.
Contributor Information
Siegfried Roth, Email: siegfried.roth@uni-koeln.de.
K VijayRaghavan, National Centre for Biological Sciences, Tata Institute of Fundamental Research, India.
K VijayRaghavan, National Centre for Biological Sciences, Tata Institute of Fundamental Research, India.
Funding Information
This paper was supported by the following grants:
Deutsche Forschungsgemeinschaft CRC 680 to Nadine Frey, Siegfried Roth.
University of Cologne International Graduate School in Genetics and Functional Genomics to Rodrigo Nunes da Fonseca, Cornelia von Levetzow.
Conselho Nacional de Desenvolvimento Científico e Tecnológico to Rodrigo Nunes da Fonseca.
Coordenação de Aperfeiçoamento de Pessoal de Nível Superior to Rodrigo Nunes da Fonseca.
Deutsche Forschungsgemeinschaft RU 1234 to Muhammad Salim Hakeemi.
Boehringer Ingelheim Fonds PhD fellowship to Dominik Stappert.
Deutsche Forschungsgemeinschaft Emmy Noether Program PA 2044/1-1) to Kristen A Panfilio.
National Institutes of Health R03 HD078578 to Jeremy A Lynch.
Alexander von Humboldt Foundation Postdoctoral Fellowship to Matthew Alan Benton.
Deutsche Forschungsgemeinschaft to Kai H Conrads.
University of Cologne Postdoctoral grant to Matthias Pechmann.
Deutsche Forschungsgemeinschaft DFG Research Fellowship 407643416 to Matthew Alan Benton.
Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro to Rodrigo Nunes da Fonseca.
Additional information
Competing interests
No competing interests declared.
Author contributions
Conceptualization, Formal analysis, Investigation, Visualization, Methodology, Writing—review and editing.
Formal analysis, Investigation, Methodology, Writing—review and editing.
Conceptualization, Formal analysis, Investigation, Methodology, Writing—review and editing.
Conceptualization, Formal analysis, Investigation, Methodology.
Formal analysis, Investigation.
Investigation, Writing—review and editing.
Investigation, Writing—review and editing.
Investigation, Methodology, Writing—review and editing.
Formal analysis, Investigation, Writing—review and editing.
Resources, Writing—review and editing.
Conceptualization, Formal analysis, Supervision, Funding acquisition, Writing—original draft, Project administration, Writing—review and editing.
Additional files
Primers used to produce anti-sense RNA for in-situ hybridization and dsRNA for RNAi-mediated gene knockdown.
Data availability
All data generated or analysed during this study are included in the manuscript and supporting files. Supplementary file 1 contains all primers used to amplify sequences for production of antisense RNA (ISH) and dsRNA (RNAi).
The following datasets were generated:
Nadine Frey, Matthew Alan Benton, Rodrigo Nunes da Fonseca, Cornelia von Levetzow, Dominik Stappert, Muhammad Salim Hakeemi, Kai H Conrads, Matthias Pechmann, Kristen A Panfilio, Jeremy A Lynch, Siegfried Roth. 2019. Gryllus bimaculatus mist mRNA, complete cds. NCBI. MK962881
Nadine Frey, Matthew Alan Benton, Rodrigo Nunes da Fonseca, Cornelia von Levetzow, Dominik Stappert, Muhammad Salim Hakeemi, Kai H Conrads, Matthias Pechmann, Kristen A Panfilio, Jeremy A Lynch, Siegfried Roth. 2019. Gryllus bimaculatus concertina mRNA, complete cds. NCBI. MK962880.1
Nadine Frey, Matthew Alan Benton, Rodrigo Nunes da Fonseca, Cornelia von Levetzow, Dominik Stappert, Muhammad Salim Hakeemi, Kai H Conrads, Matthias Pechmann, Kristen A Panfilio, Jeremy A Lynch, Siegfried Roth. 2019. Gryllus bimaculatus folded gastrulation mRNA, complete cds. NCBI. MK962882
References
- Aigouy B, Farhadifar R, Staple DB, Sagner A, Röper JC, Jülicher F, Eaton S. Cell flow reorients the axis of planar polarity in the wing epithelium of Drosophila. Cell. 2010;142:773–786. doi: 10.1016/j.cell.2010.07.042. [DOI] [PubMed] [Google Scholar]
- Anderson DT. The development of hemimetabolous insects. In: Counce SJ, Waddington CH, editors. Developmental Systems: Insects. London: Academic; 1972a. pp. 96–163. [Google Scholar]
- Anderson DT. The development of holometabolous insects. In: Counce SJ, Waddington CH, editors. Developmental Systems: Insects. London: Academic; 1972b. pp. 165–271. [Google Scholar]
- Anderson DT. Embryology and Phylogeny in Annelids and Arthropods. Oxford: Pergamon Press; 1973. [Google Scholar]
- Bailles A, Collinet C, Philippe JM, Lenne PF, Munro E, Lecuit T. Genetic induction and mechanochemical propagation of a morphogenetic wave. Nature. 2019;572:467–473. doi: 10.1038/s41586-019-1492-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barrett K, Leptin M, Settleman J. The rho GTPase and a putative RhoGEF mediate a signaling pathway for the cell shape changes in Drosophila gastrulation. Cell. 1997;91:905–915. doi: 10.1016/S0092-8674(00)80482-1. [DOI] [PubMed] [Google Scholar]
- Benton MA, Akam M, Pavlopoulos A. Cell and tissue dynamics during tribolium embryogenesis revealed by versatile fluorescence labeling approaches. Development. 2013;140:3210–3220. doi: 10.1242/dev.096271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benton MA. Cambridge, United Kingdom: University of Cambridge; 2014. Analysis of embryonic development in Tribolium castaneum using a versatile live fluorescent labelling technique.https://www.repository.cam.ac.uk/handle/1810/275921 [Google Scholar]
- Benton MA, Pechmann M, Frey N, Stappert D, Conrads KH, Chen YT, Stamataki E, Pavlopoulos A, Roth S. Toll genes have an ancestral role in axis elongation. Current Biology. 2016;26:1609–1615. doi: 10.1016/j.cub.2016.04.055. [DOI] [PubMed] [Google Scholar]
- Benton MA. A revised understanding of Tribolium morphogenesis further reconciles short and long germ development. PLOS Biology. 2018;16:e2005093. doi: 10.1371/journal.pbio.2005093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berns N, Kusch T, Schröder R, Reuter R. Expression, function and regulation of brachyenteron in the short germband insect Tribolium castaneum. Development Genes and Evolution. 2008;218:169–179. doi: 10.1007/s00427-008-0210-7. [DOI] [PubMed] [Google Scholar]
- Brigaud I, Duteyrat JL, Chlasta J, Le Bail S, Couderc JL, Grammont M. Transforming growth factor β/activin signalling induces epithelial cell flattening during Drosophila oogenesis. Biology Open. 2015;4:345–354. doi: 10.1242/bio.201410785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown SJ, Shippy TD, Miller S, Bolognesi R, Beeman RW, Lorenzen MD, Bucher G, Wimmer EA, Klingler M. The red flour beetle, Tribolium castaneum (Coleoptera): a model for studies of development and pest biology. Cold Spring Harbor Protocols. 2009;2009:pdb.emo126. doi: 10.1101/pdb.emo126. [DOI] [PubMed] [Google Scholar]
- Bullock SL, Stauber M, Prell A, Hughes JR, Ish-Horowicz D, Schmidt-Ott U. Differential cytoplasmic mRNA localisation adjusts pair-rule transcription factor activity to cytoarchitecture in dipteran evolution. Development. 2004;131:4251–4261. doi: 10.1242/dev.01289. [DOI] [PubMed] [Google Scholar]
- Campos-Ortega JA, Hartenstein V. The Embryonic Development of Drosophila melanogaster. Heidelberg: Springer; 1997. [Google Scholar]
- Chung S, Kim S, Andrew DJ. Uncoupling apical constriction from tissue invagination. eLife. 2017;6:e22235. doi: 10.7554/eLife.22235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cinalli RM, Lehmann R. A spindle-independent cleavage pathway controls germ cell formation in Drosophila. Nature Cell Biology. 2013;15:839–845. doi: 10.1038/ncb2761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark E, Peel AD. Evidence for the temporal regulation of insect segmentation by a conserved sequence of transcription factors. Development. 2018:dev.155580. doi: 10.1242/dev.155580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Copf T, Schröder R, Averof M. Ancestral role of caudal genes in axis elongation and segmentation. PNAS. 2004;101:17711–17715. doi: 10.1073/pnas.0407327102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Costa M, Wilson ET, Wieschaus E. A putative cell signal encoded by the folded gastrulation gene coordinates cell shape changes during Drosophila gastrulation. Cell. 1994;76:1075–1089. doi: 10.1016/0092-8674(94)90384-0. [DOI] [PubMed] [Google Scholar]
- Davis GK, Jaramillo CA, Patel NH. Pax group III genes and the evolution of insect pair-rule patterning. Development. 2001;128:3445–3458. doi: 10.1242/dev.128.18.3445. [DOI] [PubMed] [Google Scholar]
- Dawes-Hoang RE, Parmar KM, Christiansen AE, Phelps CB, Brand AH, Wieschaus EF. Folded gastrulation, cell shape change and the control of myosin localization. Development. 2005;132:4165–4178. doi: 10.1242/dev.01938. [DOI] [PubMed] [Google Scholar]
- de Mendoza A, Jones JW, Friedrich M. Methuselah/Methuselah-like G protein-coupled receptors constitute an ancient metazoan gene family. Scientific Reports. 2016;6:21801. doi: 10.1038/srep21801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dönitz J, Gerischer L, Hahnke S, Pfeiffer S, Bucher G. Expanded and updated data and a query pipeline for iBeetle-Base. Nucleic Acids Research. 2018;46:D831–D835. doi: 10.1093/nar/gkx984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Donoughe S, Extavour CG. Embryonic development of the cricket Gryllus bimaculatus. Developmental Biology. 2016;411:140–156. doi: 10.1016/j.ydbio.2015.04.009. [DOI] [PubMed] [Google Scholar]
- Ewen-Campen B, Shaner N, Panfilio KA, Suzuki Y, Roth S, Extavour CG. The maternal and early embryonic transcriptome of the milkweed bug Oncopeltus fasciatus. BMC Genomics. 2011;12:61. doi: 10.1186/1471-2164-12-61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gilmour D, Rembold M, Leptin M. From morphogen to morphogenesis and back. Nature. 2017;541:311–320. doi: 10.1038/nature21348. [DOI] [PubMed] [Google Scholar]
- Goltsev Y, Fuse N, Frasch M, Zinzen RP, Lanzaro G, Levine M. Evolution of the dorsal-ventral patterning network in the mosquito, anopheles gambiae. Development. 2007;134:2415–2424. doi: 10.1242/dev.02863. [DOI] [PubMed] [Google Scholar]
- Grammont M. Adherens junction remodeling by the notch pathway in Drosophila melanogaster oogenesis. The Journal of Cell Biology. 2007;177:139–150. doi: 10.1083/jcb.200609079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Handel K, Grünfelder CG, Roth S, Sander K. Tribolium embryogenesis: a SEM study of cell shapes and movements from blastoderm to serosal closure. Development Genes and Evolution. 2000;210:167–179. doi: 10.1007/s004270050301. [DOI] [PubMed] [Google Scholar]
- Handel K, Basal A, Fan X, Roth S. Tribolium castaneum twist: gastrulation and mesoderm formation in a short-germ beetle. Development Genes and Evolution. 2005;215:13–31. doi: 10.1007/s00427-004-0446-9. [DOI] [PubMed] [Google Scholar]
- Hilbrant M, Horn T, Koelzer S, Panfilio KA. The beetle amnion and serosa functionally interact as apposed epithelia. eLife. 2016;5:e13834. doi: 10.7554/eLife.13834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ho K, Dunin-Borkowski OM, Akam M. Cellularization in Locust embryos occurs before blastoderm formation. Development. 1997;124:2761–2768. doi: 10.1242/dev.124.14.2761. [DOI] [PubMed] [Google Scholar]
- Horn T, Panfilio KA. Novel functions for Dorsocross in epithelial morphogenesis in the beetle Tribolium castaneum. Development. 2016;143:3002–3011. doi: 10.1242/dev.133280. [DOI] [PubMed] [Google Scholar]
- Jain A, Ulman V, Mukherjee A, Prakash M, Pimpale L, Muenster S, Panfilio KA, Jug F, Grill SW, Tomancak P. Regionalized tissue fluidization by an actomyosin cable is required for epithelial gap closure during insect gastrulation. bioRxiv. 2019 doi: 10.1101/744193. [DOI] [PMC free article] [PubMed]
- Jha A, van Zanten TS, Philippe JM, Mayor S, Lecuit T. Quantitative control of GPCR organization and signaling by endocytosis in epithelial morphogenesis. Current Biology. 2018;28:1570–1584. doi: 10.1016/j.cub.2018.03.068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johannsen OA, Butt FH. Embryology of Insects and Myriapods. New York: McGraw-Hill; 1941. [Google Scholar]
- Kerridge S, Munjal A, Philippe JM, Jha A, de las Bayonas AG, Saurin AJ, Lecuit T. Modular activation of Rho1 by GPCR signalling imparts polarized myosin II activation during morphogenesis. Nature Cell Biology. 2016;18:261–270. doi: 10.1038/ncb3302. [DOI] [PubMed] [Google Scholar]
- Kispert A, Herrmann BG, Leptin M, Reuter R. Homologs of the mouse brachyury gene are involved in the specification of posterior terminal structures in Drosophila, Tribolium, and Locusta. Genes & Development. 1994;8:2137–2150. doi: 10.1101/gad.8.18.2137. [DOI] [PubMed] [Google Scholar]
- Kölsch V, Seher T, Fernandez-Ballester GJ, Serrano L, Leptin M. Control of Drosophila gastrulation by apical localization of adherens junctions and RhoGEF2. Science. 2007;315:384–386. doi: 10.1126/science.1134833. [DOI] [PubMed] [Google Scholar]
- Köppen M, Fernández BG, Carvalho L, Jacinto A, Heisenberg CP. Coordinated cell-shape changes control epithelial movement in zebrafish and Drosophila. Development. 2006;133:2671–2681. doi: 10.1242/dev.02439. [DOI] [PubMed] [Google Scholar]
- Kozasa T, Hajicek N, Chow CR, Suzuki N. Signalling mechanisms of RhoGTPase regulation by the heterotrimeric G proteins G12 and G13. Journal of Biochemistry. 2011;150:357–369. doi: 10.1093/jb/mvr105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LeBlanc MG, Lehmann R. Domain-specific control of germ cell polarity and migration by multifunction Tre1 GPCR. The Journal of Cell Biology. 2017;125:jcb.201612053. doi: 10.1083/jcb.201612053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leptin M, Grunewald B. Cell shape changes during gastrulation in Drosophila. Development. 1990;110:73–84. doi: 10.1242/dev.110.1.73. [DOI] [PubMed] [Google Scholar]
- Li K. The image stabilizer plugin for ImageJ. 2008 http://www.cs.cmu.edu/~kangli/code/Image_Stabilizer.html
- Lim B, Levine M, Yamazaki Y. Transcriptional Pre-patterning of Drosophila gastrulation. Current Biology. 2017;27:610. doi: 10.1016/j.cub.2017.01.067. [DOI] [PubMed] [Google Scholar]
- Linkert M, Rueden CT, Allan C, Burel JM, Moore W, Patterson A, Loranger B, Moore J, Neves C, Macdonald D, Tarkowska A, Sticco C, Hill E, Rossner M, Eliceiri KW, Swedlow JR. Metadata matters: access to image data in the real world. The Journal of Cell Biology. 2010;189:777–782. doi: 10.1083/jcb.201004104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manning AJ, Peters KA, Peifer M, Rogers SL. Regulation of epithelial morphogenesis by the G Protein-Coupled receptor Mist and its ligand Fog. Science Signaling. 2013;6:ra98. doi: 10.1126/scisignal.2004427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manning AJ, Rogers SL. The Fog signaling pathway: insights into signaling in morphogenesis. Developmental Biology. 2014;394:6–14. doi: 10.1016/j.ydbio.2014.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin AC, Kaschube M, Wieschaus EF. Pulsed contractions of an actin–myosin network drive apical constriction. Nature. 2009;457:495–499. doi: 10.1038/nature07522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mazumdar A, Mazumdar M. How one becomes many: blastoderm cellularization in Drosophila Melanogaster. BioEssays. 2002;24:1012–1022. doi: 10.1002/bies.10184. [DOI] [PubMed] [Google Scholar]
- Meijering EHW, Niessen WJ, Viergever MA. Quantitative evaluation of convolution-based methods for medical image interpolation. Medical Image Analysis. 2001;5:111–126. doi: 10.1016/S1361-8415(00)00040-2. [DOI] [PubMed] [Google Scholar]
- Meijering E, Dzyubachyk O, Smal I. Methods for cell and particle tracking. Methods in Enzymology. 2012;504:183–200. doi: 10.1016/B978-0-12-391857-4.00009-4. [DOI] [PubMed] [Google Scholar]
- Misof B, Liu S, Meusemann K, Peters RS, Donath A, Mayer C, Frandsen PB, Ware J, Flouri T, Beutel RG, Niehuis O, Petersen M, Izquierdo-Carrasco F, Wappler T, Rust J, Aberer AJ, Aspöck U, Aspöck H, Bartel D, Blanke A, Berger S, Böhm A, Buckley TR, Calcott B, Chen J, Friedrich F, Fukui M, Fujita M, Greve C, Grobe P, Gu S, Huang Y, Jermiin LS, Kawahara AY, Krogmann L, Kubiak M, Lanfear R, Letsch H, Li Y, Li Z, Li J, Lu H, Machida R, Mashimo Y, Kapli P, McKenna DD, Meng G, Nakagaki Y, Navarrete-Heredia JL, Ott M, Ou Y, Pass G, Podsiadlowski L, Pohl H, von Reumont BM, Schütte K, Sekiya K, Shimizu S, Slipinski A, Stamatakis A, Song W, Su X, Szucsich NU, Tan M, Tan X, Tang M, Tang J, Timelthaler G, Tomizuka S, Trautwein M, Tong X, Uchifune T, Walzl MG, Wiegmann BM, Wilbrandt J, Wipfler B, Wong TK, Wu Q, Wu G, Xie Y, Yang S, Yang Q, Yeates DK, Yoshizawa K, Zhang Q, Zhang R, Zhang W, Zhang Y, Zhao J, Zhou C, Zhou L, Ziesmann T, Zou S, Li Y, Xu X, Zhang Y, Yang H, Wang J, Wang J, Kjer KM, Zhou X. Phylogenomics resolves the timing and pattern of insect evolution. Science. 2014;346:763–767. doi: 10.1126/science.1257570. [DOI] [PubMed] [Google Scholar]
- Miura K, Rueden C, Hiner M, Schindelin J, Rietdorf J. ImageJ Plugin CorrectBleach V2.0.2. Zenodo 2014
- Moussian B, Roth S. Dorsoventral Axis formation in the Drosophila embryo--shaping and transducing a morphogen gradient. Current Biology. 2005;15:R887–R899. doi: 10.1016/j.cub.2005.10.026. [DOI] [PubMed] [Google Scholar]
- Münster S, Jain A, Mietke A, Pavlopoulos A, Grill SW, Tomancak P. Attachment of the blastoderm to the vitelline envelope affects gastrulation of insects. Nature. 2019;568:395–399. doi: 10.1038/s41586-019-1044-3. [DOI] [PubMed] [Google Scholar]
- Nakamura T, Yoshizaki M, Ogawa S, Okamoto H, Shinmyo Y, Bando T, Ohuchi H, Noji S, Mito T. Imaging of transgenic cricket embryos reveals cell movements consistent with a syncytial patterning mechanism. Current Biology. 2010;20:1641–1647. doi: 10.1016/j.cub.2010.07.044. [DOI] [PubMed] [Google Scholar]
- Nikolaidou KK, Barrett K. A rho GTPase signaling pathway is used reiteratively in epithelial folding and potentially selects the outcome of rho activation. Current Biology. 2004;14:1822–1826. doi: 10.1016/j.cub.2004.09.080. [DOI] [PubMed] [Google Scholar]
- Nunes da Fonseca R, von Levetzow C, Kalscheuer P, Basal A, van der Zee M, Roth S. Self-regulatory circuits in dorsoventral Axis formation of the short-germ beetle Tribolium castaneum. Developmental Cell. 2008;14:605–615. doi: 10.1016/j.devcel.2008.02.011. [DOI] [PubMed] [Google Scholar]
- Panfilio KA, Oberhofer G, Roth S. High plasticity in epithelial morphogenesis during insect dorsal closure. Biology Open. 2013;2:1108–1118. doi: 10.1242/bio.20136072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Panfilio KA, Roth S. Epithelial reorganization events during late extraembryonic development in a hemimetabolous insect. Developmental Biology. 2010;340:100–115. doi: 10.1016/j.ydbio.2009.12.034. [DOI] [PubMed] [Google Scholar]
- Parks S, Wieschaus E. The Drosophila gastrulation gene concertina encodes a G alpha-like protein. Cell. 1991;64:447–458. doi: 10.1016/0092-8674(91)90652-F. [DOI] [PubMed] [Google Scholar]
- Patil VS, Anand A, Chakrabarti A, Kai T. The Tudor domain protein Tapas, a homolog of the vertebrate Tdrd7, functions in the piRNA pathway to regulate retrotransposons in germline of Drosophila melanogaster. BMC Biology. 2014;12:61. doi: 10.1186/s12915-014-0061-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Posnien N, Schinko J, Grossmann D, Shippy TD, Konopova B, Bucher G. RNAi in the red flour beetle (Tribolium) Cold Spring Harbor Protocols. 2009;2009:pdb.prot5256. doi: 10.1101/pdb.prot5256. [DOI] [PubMed] [Google Scholar]
- Postma M, Goedhart J. PlotsOfData-A web app for visualizing data together with their summaries. PLOS Biology. 2019;17:e3000202. doi: 10.1371/journal.pbio.3000202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roth S, Stein D, Nüsslein-Volhard C. A gradient of nuclear localization of the dorsal protein determines dorsoventral pattern in the Drosophila embryo. Cell. 1989;59:1189–1202. doi: 10.1016/0092-8674(89)90774-5. [DOI] [PubMed] [Google Scholar]
- Roth S. Gastrulation in other insects. In: Stern C, editor. Gastrulation: From Cells to Embryos. New York: Coldspring Harbor Laboratory Press; 2004. pp. 105–121. [Google Scholar]
- Sarrazin AF, Peel AD, Averof M. A segmentation clock with two-segment periodicity in insects. Science. 2012;336:338–341. doi: 10.1126/science.1218256. [DOI] [PubMed] [Google Scholar]
- Schindelin J, Arganda-Carreras I, Frise E, Kaynig V, Longair M, Pietzsch T, Preibisch S, Rueden C, Saalfeld S, Schmid B, Tinevez JY, White DJ, Hartenstein V, Eliceiri K, Tomancak P, Cardona A. Fiji: an open-source platform for biological-image analysis. Nature Methods. 2012;9:676–682. doi: 10.1038/nmeth.2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schinko J, Posnien N, Kittelmann S, Koniszewski N, Bucher G. Single and double whole-mount in situ hybridization in red flour beetle (Tribolium) embryos. Cold Spring Harbor Protocols. 2009;2009:pdb.prot5258. doi: 10.1101/pdb.prot5258. [DOI] [PubMed] [Google Scholar]
- Schoppmeier M, Fischer S, Schmitt-Engel C, Löhr U, Klingler M. An ancient anterior patterning system promotes caudal repression and head formation in ecdysozoa. Current Biology. 2009;19:1811–1815. doi: 10.1016/j.cub.2009.09.026. [DOI] [PubMed] [Google Scholar]
- Schoppmeier M, Schröder R. Maternal torso signaling controls body axis elongation in a short germ insect. Current Biology. 2005;15:2131–2136. doi: 10.1016/j.cub.2005.10.036. [DOI] [PubMed] [Google Scholar]
- Schroder R, Eckert C, Wolff C, Tautz D. Conserved and divergent aspects of terminal patterning in the beetle Tribolium castaneum. PNAS. 2000;97:6591–6596. doi: 10.1073/pnas.100005497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schröder R. Vasa mRNA accumulates at the posterior pole during blastoderm formation in the flour beetle Tribolium castaneum. Development Genes and Evolution. 2006;216:277–283. doi: 10.1007/s00427-005-0054-3. [DOI] [PubMed] [Google Scholar]
- Seher TC, Narasimha M, Vogelsang E, Leptin M. Analysis and reconstitution of the genetic cascade controlling early mesoderm morphogenesis in the Drosophila embryo. Mechanisms of Development. 2007;124:167–179. doi: 10.1016/j.mod.2006.12.004. [DOI] [PubMed] [Google Scholar]
- Sommer RJ, Tautz D. Involvement of an orthologue of the Drosophila pair-rule gene hairy in segment formation of the short germ-band embryo of Tribolium (Coleoptera) Nature. 1993;361:448–450. doi: 10.1038/361448a0. [DOI] [PubMed] [Google Scholar]
- Sommer RJ, Tautz D. Expression patterns of twist and snail in tribolium (Coleoptera) suggest a homologous formation of mesoderm in long and short germ band insects. Developmental Genetics. 1994;15:32–37. doi: 10.1002/dvg.1020150105. [DOI] [PubMed] [Google Scholar]
- Stanley MSM, Grundmann AW. The embryonic development of Tribolium confusum. Annals of the Entomological Society of America. 1970;63:1248–1256. doi: 10.1093/aesa/63.5.1248. [DOI] [Google Scholar]
- Stappert D, Frey N, von Levetzow C, Roth S. Genome-wide identification of Tribolium dorsoventral patterning genes. Development. 2016;143:2443–2454. doi: 10.1242/dev.130641. [DOI] [PubMed] [Google Scholar]
- Sweeton D, Parks S, Costa M, Wieschaus E. Gastrulation in Drosophila: the formation of the ventral furrow and posterior midgut invaginations. Development. 1991;112:775–789. doi: 10.1242/dev.112.3.775. [DOI] [PubMed] [Google Scholar]
- Tetley RJ, Staddon MF, Heller D, Hoppe A, Banerjee S, Mao Y. Tissue fluidity promotes epithelial wound healing. Nature Physics. 2019;18 doi: 10.1038/s41567-019-0618-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tetley RJ, Mao Y. The same but different: cell intercalation as a driver of tissue deformation and fluidity. Philosophical Transactions of the Royal Society B: Biological Sciences. 2018;373:20170328. doi: 10.1098/rstb.2017.0328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thevenaz P, Ruttimann UE, Unser M. A pyramid approach to subpixel registration based on intensity. IEEE Transactions on Image Processing. 1998;7:27–41. doi: 10.1109/83.650848. [DOI] [PubMed] [Google Scholar]
- Ullmann SL. The origin and structure of the mesoderm and the formation of the coelomic sacs in Tenebrio molitor L. [Insecta, Coleoptera] Philosophical Transactions of the Royal Society of London Series B-Biological Sciences. 1964;248 doi: 10.1098/rstb.1964.0012. [DOI] [Google Scholar]
- Urbansky S, González Avalos P, Wosch M, Lemke S. Folded gastrulation and T48 drive the evolution of coordinated mesoderm internalization in flies. eLife. 2016;5:e18318. doi: 10.7554/eLife.18318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van der Zee M, Berns N, Roth S. Distinct functions of the Tribolium zerknüllt genes in serosa specification and dorsal closure. Current Biology. 2005;15:624–636. doi: 10.1016/j.cub.2005.02.057. [DOI] [PubMed] [Google Scholar]
- van der Zee M, Benton MA, Vazquez-Faci T, Lamers GE, Jacobs CG, Rabouille C. Innexin7a forms junctions that stabilize the basal membrane during cellularization of the blastoderm in Tribolium castaneum. Development. 2015;142:2173–2183. doi: 10.1242/dev.097113. [DOI] [PubMed] [Google Scholar]
- van Drongelen R, Vazquez-Faci T, Huijben T, van der Zee M, Idema T. Mechanics of epithelial tissue formation. Journal of Theoretical Biology. 2018;454:182–189. doi: 10.1016/j.jtbi.2018.06.002. [DOI] [PubMed] [Google Scholar]
- von Levetzow C. Köln, Germany: Institute of Developmental Biology, University of Cologne; 2008. Konservierte und divergente Aspekte der twist-, snail- und concertina-Funktion im Käfer Tribolium castaneum. [Google Scholar]
- Wang Y, Riechmann V. The role of the actomyosin cytoskeleton in coordination of tissue growth during Drosophila oogenesis. Current Biology. 2007;17:1349–1355. doi: 10.1016/j.cub.2007.06.067. [DOI] [PubMed] [Google Scholar]
- Wanninger A. Evolutionary Developmental Biology of Invertebrates. Ecdysozoa I: Non-Tetraconata. Vol. 3. Vienna, Austria: Springer-Verlag; 2015a. [Google Scholar]
- Wanninger A. Evolutionary Developmental Biology of Invertebrates. Ecdysozoa II: Crustacea. Vol. 4. Vienna, Austria: Springer-Verlag; 2015b. [Google Scholar]
- Wu LH, Lengyel JA. Role of caudal in hindgut specification and gastrulation suggests homology between Drosophila amnioproctodeal invagination and vertebrate blastopore. Development. 1998;125:2433–2442. doi: 10.1242/dev.125.13.2433. [DOI] [PubMed] [Google Scholar]
- Zusman SB, Wieschaus EF. Requirements for zygotic gene activity during gastrulation in Drosophila melanogaster. Developmental Biology. 1985;111:359–371. doi: 10.1016/0012-1606(85)90490-7. [DOI] [PubMed] [Google Scholar]