Abstract
Species’ traits influence how populations respond to land-use change. However, even in well-characterized groups such as birds, widely studied traits explain only a modest proportion of the variance in response across species. Here, we show that associations with particular forest types strongly predict the sensitivity of forest-dwelling Amazonian birds to agriculture. Incorporating these fine-scale habitat associations into models of population response dramatically improves predictive performance and markedly outperforms the functional traits that commonly appear in similar analyses. Moreover, by identifying habitat features that support assemblages of unusually sensitive habitat-specialist species, our model furnishes straightforward conservation recommendations. In Amazonia, species that specialize on forests along a soil–nutrient gradient (i.e. both rich-soil specialists and poor-soil specialists) are exceptionally sensitive to agriculture, whereas species that specialize on floodplain forests are unusually insensitive. Thus, habitat specialization per se does not predict disturbance sensitivity, but particular habitat associations do. A focus on conserving specific habitats that harbour highly sensitive avifaunas (e.g. poor-soil forest) would protect a critically threatened component of regional biodiversity. We present a conceptual model to explain the divergent responses of habitat specialists in the different habitats, and we suggest that similar patterns and conservation opportunities probably exist for other taxa and regions.
Keywords: birds, conservation, Amazon, forest, habitat, traits
1. Introduction
The expansion of agriculture in the tropics is a pre-eminent threat to global biodiversity [1]. Tropical forests support the bulk of Earth's terrestrial species, and, depending on the taxonomic group, we stand to lose between 8% and 65% of those species if primary forests are subjected to wholesale degradation and conversion [2]. From 2016 to 2018, annual losses of primary tropical forest exceeded 4.5 million hectares on average [3]. Thus, in order to design effective conservation interventions, it is critical to understand how and why agriculture harms tropical forest species.
Functional traits provide an important framework for understanding species' responses to land-use change. Traits can predict both global extinction risk and local population declines across species [4–6], and can shed light on the mechanisms that cause species to be threatened [7]. For example, different functional vertebrate guilds are characteristically vulnerable to different forms of anthropogenic pressure (e.g. large-bodied birds are characteristically vulnerable to hunting, while small-bodied, forest-dwelling birds are characteristically vulnerable to habitat loss) [8–10]. The relationship between the functional traits of species and their population responses to land use is especially well characterized in birds, where key traits that have been investigated thus far include body mass, diet, migratory behaviour, and foraging stratum [11–15]. Coarse-scale habitat associations (i.e. forest versus non-forest) are also important, with elevated risk for forest-dwelling species [11,16] and for species that are restricted to a single broad habitat type as opposed to habitat generalists [8].
Recent work shows that land-use change tends to homogenize tropical forest communities across space [2,17,18]. This pattern suggests that tropical forest species with limited spatial distributions may undergo more severe population declines with land-use change [19]. For example, habitat specialists on humid (as opposed to dry) forests in Costa Rica and Brazil show elevated vulnerability to agriculture [18,20,21]. However, little is known about how myriad other forms of habitat specialization and range limitation might influence species vulnerability in tropical forests. Numerous tropical forest species specialize on particular forest types (e.g. forests distinguished by flooding regimes or soil nutrients) or have range limits along major rivers. Nevertheless, while coarse-scale associations with forest versus non-forest habitats are well known to predict avian responses to tropical agriculture [11], the influence of fine-scale habitat specialization and range restriction within forests remains underexplored.
In this paper, we examine the relationship between fine-scale habitat specialization and avian responses to land-use change at the global epicentre of terrestrial biodiversity, the western Amazon. In western Amazonia, smallholder agriculture (i.e. smallholder agricultural mosaics of intermingled cultivation, pasture and secondary-forest fallows) is a dominant driver of deforestation [22,23]. In this region, smallholder agriculture homogenizes bird communities across hydrologically and edaphically structured variation within the forest landscape [24]. These key regional habitats include (i) floodplain forest regularly inundated by the annual flood of the Amazon and its major tributaries and (ii) terra firme forest above the highest flood level. Within the terra firme, a further distinction exists between (a) rich-soil forest growing on nutrient-rich clays and (b) poor-soil forest growing on weathered clay terraces and sandy soils. Each of these habitats supports distinctive plant communities [25–27] and bird assemblages [28–30]. Another important feature of the Amazonian avifauna is the existence of numerous species with regional- or global-range limits at the Amazon River and its major tributaries [31,32]. Because agriculture homogenizes Amazonian bird communities across space [24], we hypothesized that both habitat-specialist species and species whose ranges are limited by river boundaries would display heightened sensitivities to forest conversion.
We test these hypotheses through a point-count inventory of birds at paired forest and agricultural sites in northeastern Peru, where agriculture is known to homogenize bird communities across forest types [24]. The dataset provides uniquely comprehensive coverage of the regional avifauna, with 455 species detected across floodplains, rich-soil terra firme and poor-soil terra firme on both banks of the Amazon River, and therefore supports the analysis of relationships between habitat associations and population change across multiple forest types.
2. Methods
(a). Bird surveys
We surveyed birds with four point counts (10 min duration, 100 m radius) at 235 points in Loreto Department, Peru, which retains most of its original primary-forest cover. The full dataset is published elsewhere, with a detailed description of the study area (including the forest habitats and agricultural activities that we studied) and sampling methodology [24]. In brief, we stratified our sampling points across floodplains (72 points) and terra firme (163 points), and across both banks of the Amazon River. Smallholder agriculture is regularly practised both on floodplains and in terra firme, and within each forest type, we situated half of our sampling points in extensive primary forest and half in smallholder agricultural mosaics of intermingled cultivation, pasture and secondary-forest fallows. We matched primary-forest and smallholder-agriculture points for flooding regime, soil texture and geographical proximity by identifying accessible, minimally disturbed primary-forest locations and searching for appropriate geographically proximate smallholder sites using a combination of aerial imagery, local knowledge and field examination of candidate sites. Within terra firme, we situated 48 points on white-sand soils (which are extremely nutrient poor), evenly divided between primary-forest and smallholder-agriculture sites. We situated the remaining 115 terra firme points on clay soils. We lacked explicit soil-nutrient data at these sites, but at our primary-forest terra firme points we ultimately recorded 50 bird species classified as rich-soil specialists, and 29 species classified as poor-soil specialists (habitat-specialist classifications based on independent datasets; see below). The occurrence of rich- and poor-soil specialist species was strongly negatively correlated across points (electronic supplementary material, figure S1).
We assembled an abundance dataset for each species by taking the maximum count of individuals obtained on any of the four visits to each point [33,34]. Elsewhere, we have shown that detection probabilities are at least as high (probably higher) in agriculture as in primary forest and discussed why N-mixture modelling is unlikely to provide accurate detection-corrected abundance estimates for our dataset [24]. Differences in detectability between species or across sites will not affect our conclusions in this paper as long as the detectability difference between forest and agriculture (i.e. across sites) is not strongly correlated with the species traits considered here. This assumption is likely to hold for the traits that we are examining (see below), and the assumption is ubiquitous in similar analyses (e.g. [11]).
(b). Morphological and behavioural traits
We compiled information on multiple morphological and behavioural traits that have been shown to influence responses to land-use change in tropical birds (table 1) [11,12]. We obtained information on migratory status (breeder versus non-breeder) from [35], and we obtained information on body mass, diet and foraging stratum from the EltonTraits 1.0 database [36]. The database allocates each species to one of five dietary guilds (frugivore, insectivore, nectarivore, carnivore/scavenger and omnivore) and provides percentage usage of seven foraging strata. Among the species in our dataset, four strata (ground, understorey, midstorey and canopy) account for over 97% of the total usage across species and thus sum approximately to one for almost all species. We therefore treated the percentage usage of ground, understorey and midstorey strata as three continuous variables that collectively capture nearly all of the variation in stratum. We were unable to compile data on nest construction and placement for the majority of species in our study and therefore did not include nesting traits.
Table 1.
Covariates used in models of species' sensitivity to agriculture.
| category | covariatea |
|---|---|
| coarse forest associations | forest user |
| forest specialist | |
| morphological and behavioural traits | stratum: ground |
| stratum: understorey | |
| stratum: midstorey | |
| diet: invertebrate | |
| diet: omnivore | |
| diet: granivore | |
| diet: carnivore | |
| body mass | |
| migratory | |
| fine-scale habitat specialization and range restriction | river-limited |
| floodplain specialist | |
| terra firme specialist | |
| poor-soil specialist | |
| rich-soil specialist |
aContinuous predictors (stratum and body mass) are standardized. Binary predictors are coded as ones relative to an intercept for non-forest, frugivore/nectivore, non-migratory, non-river-limited, and non-specialist.
(c). Habitat: data sources
We combined data from multiple sources to assess species habitat associations in our study landscape. To understand how functional traits influence the intrinsic sensitivity of forest-dwelling species to land-use change, we focused exclusively on the naturally occurring landscapes of our study region, and we did not use any prior information about species occurrence in anthropogenic habitats as predictors in our models. We compiled information both on coarse-scale associations with forest versus naturally occurring non-forest habitats and on fine-scale associations with floodplains versus terra firme and to poor versus rich soils within terra firme. Coarse-scale forest associations are well known to predict avian responses to land-use change, with forest-dwelling species less tolerant of disturbance than species associated with non-forest habitats [11,16]; the novel aspect of this study involves the fine-scale predictors.
We obtained information about habitat specialization from a canonical database of habitat associations for Neotropical birds [37] and from two more recent studies of edaphic specialization by birds within our study area [29,30]. To assess habitat associations in a regional context, we focused on 15 habitat classes in [37] that occur in western Amazonia. We modified the habitat classifications for several species based on information that was not available in 1996 (when the work [37] was published) or information specific to western Amazonia (see supplementary appendix S1 for the detailed justification of all changes). We then aggregated these 15 classes to four overarching categories that define the habitats of interest (table 2).
Table 2.
Habitat categories used to categorize coarse-scale forest associations, terra firme specialization and floodplain specialization.
| codea | habitat | habitat category for this study | natural | forest |
|---|---|---|---|---|
| F1 | upland forest | terra firme forest | yes | yes |
| F12 | white-sand forest | |||
| F2 | flooded forest | floodplain forest | yes | yes |
| F3 | river-edge forest | |||
| F13 | palm swamp | |||
| N11 | riparian thickets | floodplain non-forest | yes | no |
| N12 | river-island scrub | |||
| A1 | freshwater marshes | |||
| A5 | river beaches | |||
| A6 | lakes and ponds | |||
| A8 | rivers | |||
| A9 | streams | |||
| F15 | secondary forest | anthropogenic | no | no |
| N13 | pastures/agriculture | |||
| N14 | second-growth scrub |
aHabitat codes from [37].
(d). Habitat: forest associations
We define coarse-scale habitat associations in terms of forest and (natural) non-forest habitats, based on the habitat associations in [37]. All species in our dataset occur in at least one natural habitat, but not all species occur in forest. We treat species that occur in at least one natural forest habitat as forest users and species that occur exclusively in forest as forest specialists (figure 1). We refer to these categories as forest associations, distinct from the fine-scale habitat specializations described below.
Figure 1.
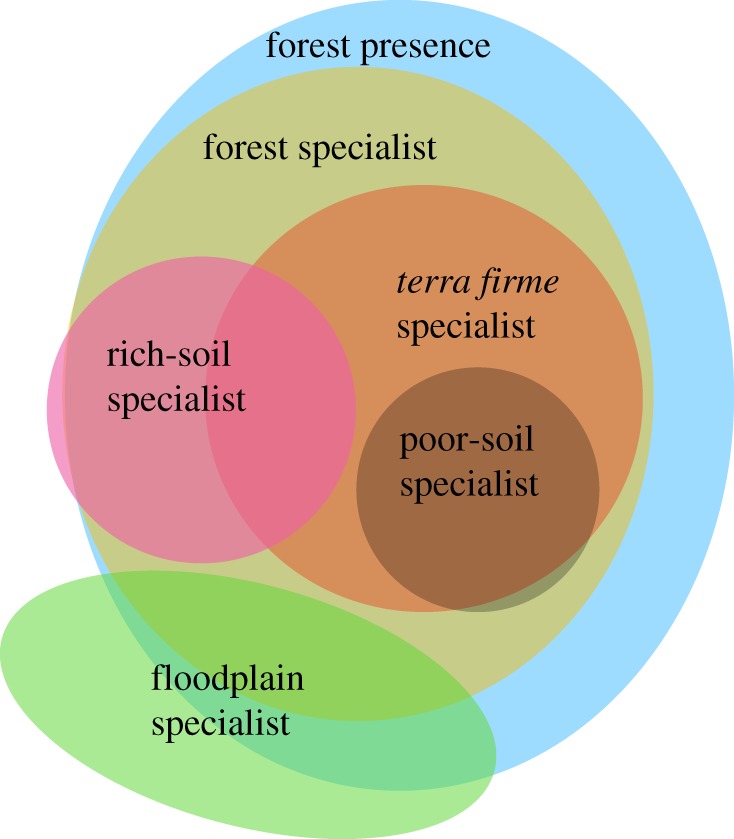
Habitat specializations used to predict population responses to agriculture. Regions of the diagram are not to scale but provide a rough idea of the relative size of each category. Forest presence and forest specialization are coarse-scale habitat associations, while the remaining habitat specializations are fine-scale. (Online version in colour.)
(e). Habitat: fine-scale specialization
We define fine-scale habitat associations in terms of edaphic and hydrological subdivisions of the Amazonian landscape (i.e. Grinnellian habitat specialization sensu [38]). Based on [37], we treat a species as a floodplain specialist if it occurs in at least one natural floodplain habitat but no natural terra firme habitats. Likewise, we treat a species as a terra firme specialist if it occurs in at least one natural terra firme habitat but no natural floodplain habitats. Every natural terra firme habitat is forested, and so all terra firme specialists are forest-specialist. Not every natural floodplain habitat is forested, and some floodplain specialists are not forest-users, while others are forest users but not forest specialists (figure 1). All species that are not forest users are floodplain specialists.
To assess edaphic specialization within terra firme (i.e. poor-soil versus rich-soil specialists), we relied on previous work within our study area. Pomara et al. [30] sampled birds and soil cation concentrations at multiple sites and used indicator species analysis to classify bird species as rich-soil or poor-soil indicators. Alvarez Alonso et al. [29] synthesized extensive fieldwork to compile a list of white-sand specialist species and a separate list of species that are absent or very rare on white sands compared with adjacent clay soils. We classified as rich-soil specialists those species previously identified as rich-soil indicators [30] or those that are rare/absent on white sands [29]. We classified as poor-soil specialists those species previously identified as poor-soil indicators [30] or those that are white-sand specialists [29]. Under this classification scheme, many rich-soil specialists are shared with floodplains, while most poor-soil specialists are restricted to terra firme (figure 1).
We refer to species that are not classified in any of our habitat-specialist categories as ‘habitat generalists’ and to species that, in particular, are not captured by either of our edaphic specialist categories as ‘edaphic generalists’.
(f). River limitation
We classified river-limited species based on their distributions with respect to the Amazon/Marañon, Napo and Ucayali rivers, which together split our study region into four quadrants. We define river-limited species as those that are absent (no documented Peruvian records) from at least one quadrant, based on published range maps [35] supplemented by recently published records [39,40] and well-documented eBird reports [41]. Because white-sand forest is absent from Peru east of the Napo and is not extensive west of the Ucayali, white-sand specialists (sensu [29]) that occur on both banks of the Amazon were never classified as river-limited, regardless of their distributions with respect to the Napo and Ucayali rivers.
(g). Modelling framework
We modelled species’ responses to agricultural conversion as a function of habitat specialization, river limitation, and functional traits. For each species, we assumed that the total counts of individuals sampled across all points in intact and agricultural landscapes (Ci and Ca) were drawn from Poisson distributions parametrized by the true abundances (Ai and Aa) in the respective landscapes.
We sought inference on the log-ratio of the true abundances R = log(Ai/Aa) [11]. Given observed counts Ci and Ca, the conditional likelihood for R can be re-expressed in a convenient form that permits direct inference on R via binomial regression. Briefly, the likelihood function for R that arises from assuming that Ci and Ca are Poisson variates is identical to the likelihood function for R that arises from, assuming that Ci is a binomial variate with the number of trials equal to Ci + Ca [42]. In the binomial model, R is equal to the logit of the binomial proportion, and so binomial logistic regression yields straightforward inference on R. We assumed that R would depend on species' traits and habitat associations, plus a homogeneous normal error term [11]. This assumption yields a generalized linear mixed model (GLMM) with fixed effects of traits and a random intercept for species.
(h). Model building and comparison
We considered multiple binomial GLMMs using different combinations of predictor variables, designed to test the ability of fine-scale habitat specialization and river limitation to (i) predict disturbance sensitivity, (ii) improve the predictive performance of models that also incorporate morphological and behavioural traits, and (iii) outperform models based on morphological and behavioural traits. We had a strong a priori expectation that coarse-scale forest associations would predict responses, with elevated sensitivity to smallholder agriculture in forest-specialist species. To ensure that we obtained a fair comparison between models incorporating fine-scale habitat specialization versus morphological and behavioural traits, all models (except a null model) included terms for coarse-scale forest associations.
In total, we fit six models using different combinations of predictor variables (electronic supplementary material, table S1). First, we fitted a global model using river-limitation, forest associations, fine-scale habitat specializations (figure 1) and morphological/behavioural traits. Second, we fitted a null model containing only the intercept and the random effect of species. Third, we fitted a ‘coarse’ model that additionally included coarse-scale forest associations (i.e. terms for forest user and forest specialist). Fourth, we fitted a ‘habitat’ model that included forest associations, river-limitation and all fine-scale habitat specializations. Fifth, we fitted a ‘traits’ model that included forest associations and all morphological/behavioural traits. Sixth, we fitted a ‘traits2’ model identical to the previous but excluding foraging stratum. We did so to ensure that the foraging stratum predictors, all with small effect sizes, did not substantially diminish the predictive performance of the traits model.
We fitted all models in Stan [43] via R package rstanarm [44] using weakly informative priors (electronic supplementary material, table S2) and four chains, each with 1000 iterations of burn-in and 1000 iterations of sampling. We compared model performance based on the expected log pointwise predictive density (ELPD) obtained via 10-fold cross-validation in rstanarm. The ELPD obtained through cross-validation is usually the most reliable metric of out-of-sample predictive performance for Bayesian models [45]. Additionally, we assessed the ability of the predictors to explain variation in the latent abundance log-ratios by computing per cent variance explained relative to the null model on the link scale via comparison of the random-effect variances. Following [46], we refer to this quantity as R2.
3. Results
Our field effort produced records of 8570 individuals of 455 species, providing unprecedented species coverage across multiple land-use categories of the western Amazon. The dataset includes substantial numbers of floodplain specialist species (167), terra firme specialists (155), rich-soil specialists (56), poor-soil specialists (30), river-limited species (60) and habitat-generalist species that do not belong to any of the above categories (100).
(a). Model performance
R-hat diagnostics [47] indicated model convergence (R-hat = 1.0 for all parameters). Graphical posterior and mixed predictive checks indicated adequate model fit (electronic supplementary material, figures S2 and S3).
All of our models for population change yielded substantial improvements over the null model in terms of both ELPD and variance explained (figure 2; electronic supplementary material, table S3) and yielded similar estimates for all shared parameters apart from the intercept, the interpretation of which differs between models (figure 3). The global model and the habitat model performed best. Models that excluded fine-scale specialization performed substantially worse and offered only modest improvement over the coarse model that used only coarse-scale forest associations (i.e. forest versus non-forest; figure 2; electronic supplementary material, table S3). These coarse-scale forest-association terms captured about 34% of the variance in species responses. Including fine-scale habitat specialization increased R2 to roughly 62% (figure 2; electronic supplementary material, table S3). Further including all morphological and behavioural traits more than doubled the model complexity, with nine additional free parameters, but increased R2 only by a further 4%.
Figure 2.
Model performance in terms of (a) ELPD and (b) R2. ELPD is expressed in terms of improvement over the null model, and significant differences are indicated above the bars: capitalization signifies a difference of at least 2 s.e. (95% confidence interval), while different letters signify a difference of at least 3 s.e. (99.7% confidence interval). R2 is defined following the study by Nakagawa et al. [46]; bars display posterior mean estimates.
Figure 3.
Parameter estimates (posterior means and 95% credible intervals) for the effects of habitat associations and traits on abundance ratios in agriculture versus forest. Shading groups broad categories of predictors (labelled at right). (Online version in colour.)
(b). Habitat specialization and trait effects
Forest associations, morphological/behavioural traits and fine-scale specialization generally had the expected effects on population change (figure 3; electronic supplementary material, table S4). In line with previous work, forest-specialist species tended to experience steep declines in response to land-use change, and these declines tended to be steeper in non-canopy species, insectivores, large-bodied species and sedentary species. With the coarse-scale, forest-specialist effect already included in the model, forest users tended to decline more than non-forest species, but the credible interval for this effect overlapped zero.
In addition to the effects of coarse-scale habitat specialization (forest versus non-forest), river limitation and fine-scale habitat specializations had substantial effects on species' population responses (figures 3 and 4). Declines were steepest in river-limited species, terra firme specialists, rich-soil specialists and especially poor-soil specialists. In stark contrast to these groups, floodplain specialists tended to increase substantially in agricultural areas. The high abundance of floodplain specialists at agricultural points involved not only an increase in abundance within the floodplain, but also widespread colonization of terra firme agricultural habitats (figure 5).
Figure 4.
Modelled abundance ratios in agriculture versus forest, based on the global model. Species are ordered by their posterior means (points) and plotted with 95% credible intervals (lines). The first panel shows all 451 species analysed; subsequent panels highlight different subsets of the species pool. Forest-dwelling floodplain specialists are species that are both forest users and floodplain specialists.
Figure 5.
Mean pointwise richness and abundance (from raw data) of floodplain specialists across the landscape. Floodplain specialists proliferate in agricultural landscapes due to moderate increases in the floodplain itself as well as tremendous increases across the terra firme landscape. Error bars show ±1 s.e.
4. Discussion
Ecological specialization is ubiquitous in highly diverse tropical forest communities where variation in traits such as diet, foraging stratum and habitat specialization facilitate diversity via niche-packing [48]. Thus, it makes sense to view the erosion of tropical forest diversity through the lens of these traits. Yet while diet and foraging stratum are routinely considered in studies of species responses to forest loss and degradation [11,12], fine-scale habitat specialization is not. Consistent with recent work on the homogenization of tropical forest habitats [24], we show that fine-scale habitat specialization is a major predictor of species declines in Amazonian agricultural mosaics.
Coarse-scale forest associations and fine-scale habitat specialization together explain roughly 62% of the variance (on the link scale) in species responses to agriculture, representing a large improvement over a model based on forest associations alone (the coarse model, which explains 34% of the variance). Fine-scale habitat specialization offers a markedly better improvement over the coarse model than do morphological and behavioural traits. Including fine-scale habitat specialization in the global model provides a major improvement over the traits models, whereas including traits in the global model provides marginal and non-significant improvement over the habitat model. Thus, habitat associations are crucial for predicting the response of Amazonian birds to forest disturbance.
(a). Covariate relationships
Terra firme specialists, poor-soil specialists and rich-soil specialists show substantially heightened disturbance sensitivity. Note that all terra firme specialists are forest specialists, and nearly all poor-soil specialists are terra firme specialists. The sum of these three effects—forest, terra firme and poor-soil—means that poor-soil specialists are sensitive indeed. Floodplain specialists, on the other hand, are uniquely insensitive to disturbance. Numerous floodplain specialists depend on non-forest habitats, and these species proliferate across agricultural habitats. Moreover, even forest-dwelling floodplain specialists are unusually insensitive to land-use change (figure 4). Note that the positive model-estimated effect of floodplain specialization is fully independent of the response of non-forest floodplain specialists, because the coarse forest-association terms fully capture the differential response of non-forest floodplain specialists and all other species. Because floodplain specialists are insensitive to forest conversion, we reject our initial hypothesis that habitat specialization per se predicts heightened disturbance sensitivity. The response of floodplain specialists to land-use change does contribute to biotic homogenization in our study landscape, but via proliferation across terra firme agriculture rather than disappearance from floodplains (i.e. additive homogenization sensu [19]).
As predicted, river limitation is associated with elevated disturbance sensitivity. However, the mechanism underlying this effect remains unclear (our initial hypothesis was based on the tendency for land-use change to homogenize tropical communities, not on any particular mechanism for this effect). One possibility is that river limitation is a proxy for dispersal limitation [32], which might result from a reluctance to enter or cross non-forest habitats such as those that occur along rivers. Because many riverine range limits are associated with competition with congeners on the opposite bank [49], an alternative explanation is that river-limited species tend to maintain stable niches that do not permit invasion by congeners but are erased by forest conversion.
(b). Conceptual model for habitat-specialist response
The divergent responses of habitat-specialist species can be understood in terms of the simple idea that habitat change produces larger population changes in habitat specialists than in habitat generalists. Thus, a given change in habitat is likely to produce large declines in some habitat specialists and large increases in others.
Importantly, Amazonian floodplain habitats are created and maintained by frequent disturbance [25]. Even mature floodplain forests contain large amounts of edge at the borders of oxbow lakes [50,51]. It appears that most floodplain specialists depend on structural features of the floodplain vegetation, including non-forest areas for some species, but also vines and tangles along gaps for forest-dwelling species. This connection to edge and disturbance explains their ability to persist and proliferate across the smallholder landscape. However, a few floodplain specialists (e.g. the antbird Myrmelastes hyperythra) appear to be associated with damp flood-prone soils rather than disturbance-dependent vegetation, and such species decline following agricultural conversion, sometimes severely.
(c). Caveats
We have shown that habitat associations strongly predict species responses to smallholder agriculture in the western Amazon. However, this effect might arise due to variation in agricultural practices across habitats rather than the intrinsic sensitivity of habitat-specialist birds. Several lines of evidence suggest that this is not the case, or at least is not a complete explanation. First, the proliferation of floodplain specialists across agriculture in terra firme demonstrates that their ability to tolerate agriculture is not restricted to a peculiar form of agriculture practiced on floodplains (figure 5). Second, rich-soil and poor-soil specialists both decline more severely than edaphic generalists, but the two terra firme forest types cannot simultaneously experience heavier agricultural impacts than terra firme on average. Third, secondary-forest cover, a major predictor of the abundance of disturbance-sensitive birds in the smallholder mosaic [24], is similar at north-bank and south-bank sites (electronic supplementary material, figure S4a). Thus, variation in smallholder activity is unlikely to explain the river-limitation effect. On the other hand, white-sand agricultural points had less secondary-forest cover than upland agricultural points (electronic supplementary material, figure S4b), probably as a result of the glacial pace of forest regeneration on poor soils (electronic supplementary material, figure S4c). This effect could partially account for the unique disturbance sensitivity of the poor-soil specialist avifauna.
(d). Conservation implications and generality
Fine-scale habitat specialization data are available for numerous species, including nearly all Neotropical birds [37], and might substantially improve our understanding of how agriculture threatens tropical biodiversity and which species are likely to be at risk. Importantly, our model's forest-association terms were based exclusively on natural habitats, and we did not incorporate any prior information on the use of anthropogenic habitats in our model. Therefore, the model can predict the response of species that have yet to be studied with respect to their responses to land-use change as long as their natural habitat associations are known.
Results from our model have a direct bearing on how to prioritize conservation interventions spatially in the western Amazon. The extreme disturbance sensitivity of poor-soil specialists suggests that these habitats should be a particularly high priority for conservation. Diverting agricultural expansion away from the poorest soils might represent a win–win–win for food production, local livelihoods and the most sensitive component of the regional avifauna. In some cases, farmers settling the region from elsewhere gravitate towards the worst soils because they are easy to clear and are the nearest unfarmed plots to villages (J.B.S. 2013, personal observation); achieving the win–win–win might be as simple as making new farmers aware of the vicissitudes of local soil quality. Thus, incorporating habitat specialization can provide an unusually direct link between trait-based models and conservation practice [7].
Multiple Amazonian taxa in addition to birds display fine-scale specialization along the edaphic and hydrologic gradients that we studied [52], and fine-scale forest-habitat variation along comparable edaphic and hydrologic gradients is widespread in the African and Asian tropics. Thus, forest-habitat specialization may be an important trait for multiple taxa across multiple tropical realms. Moreover, although floodplain and white-sand habitats in Africa and Asia apparently support many fewer habitat-specialist bird species than comparable Amazonian habitats [28,53], other forms of forest-habitat specialization are ubiquitous and well characterized throughout the tropics. For example, habitat-specialist assemblages along elevation and precipitation gradients may display similar patterns to those that we uncovered. Deforestation might drive stronger declines in elevational specialists compared with generalists except at the highest elevations, where treeline specialists might proliferate downslope in a manner akin to Amazonian floodplain specialists. Likewise, deforestation might drive strong declines in wet-forest specialists while dry-forest specialists proliferate, a pattern that has already been noted in northwest Costa Rica [18]. At the same time, our results provide new impetus for detailed studies of habitat associations across various tropical forest types and across taxa. Incorporating these associations into models of species vulnerability to land-use change may yield a substantially updated and improved assessment of prospects for biodiversity retention and loss throughout the tropics.
Supplementary Material
Acknowledgements
We thank Z. Burialova, U. Srinivasan, X. Giam and P. Elsen for useful discussions. We thank numerous individuals and organizations named in [24] for logistical support in the field. We thank P. Saboya del Castillo for support and guidance, and D. Lane and B. Whitney for freely shared knowledge of Amazonian birdsong.
Ethics
We thank the Peruvian government ministry MINAGRI and the protected areas authority SERNANP for research permission (Resolución Directoral no. 422-2013-MINAGRI-DGFFS/DGEFFS and Resolución Jefatural no. 009-2013-SERNANP-RNAM-J).
Data accessibility
Data available from the Dryad Digital Repository: https://doi.org/10.5061/dryad.6944855 [54].
Authors' contributions
J.B.S. designed the study, collected and analysed the data, and drafted the manuscript. D.S.W. codesigned the study and extensively revised the manuscript. All authors gave final approval for publication and agree to be held accountable for the work performed therein.
Competing interests
We declare we have no competing interests
Funding
Funding for this study was provided by the High Meadows Foundation.
References
- 1.Laurance WF, Sayer J, Cassman KG. 2014. Agricultural expansion and its impacts on tropical nature. Trends Ecol. Evol. 29, 107–116. ( 10.1016/j.tree.2013.12.001) [DOI] [PubMed] [Google Scholar]
- 2.Alroy J. 2017. Effects of habitat disturbance on tropical forest biodiversity. Proc. Natl Acad. Sci. USA 114, 6056–6061. ( 10.1073/pnas.1611855114) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hansen MC, et al. 2013. High-resolution global maps of 21st-century forest cover change. Science 342, 850–853. ( 10.1126/science.1244693) [DOI] [PubMed] [Google Scholar]
- 4.Purvis A, Gittleman JL, Cowlishaw G, Mace GM. 2000. Predicting extinction risk in declining species. Proc. R. Soc. B 267, 1947–1952. ( 10.1098/rspb.2000.1234) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kotze DJ, O'Hara RB. 2003. Species decline—but why? Explanations of carabid beetle (Coleoptera, Carabidae) declines in Europe. Oecologia 135, 138–148. ( 10.1007/s00442-002-1174-3) [DOI] [PubMed] [Google Scholar]
- 6.Hanna E, Cardillo M. 2014. Island mammal extinctions are determined by interactive effects of life history, island biogeography and mesopredator suppression. Global Ecol. Biogeogr. 23, 395–404. ( 10.1111/geb.12103) [DOI] [Google Scholar]
- 7.Cardillo M, Meijaard E. 2012. Are comparative studies of extinction risk useful for conservation? Trends Ecol. Evol. 27, 167–171. ( 10.1016/j.tree.2011.09.013) [DOI] [PubMed] [Google Scholar]
- 8.Owens IP, Bennett PM. 2000. Ecological basis of extinction risk in birds: habitat loss versus human persecution and introduced predators. Proc. Natl Acad. Sci. USA 97, 12 144–12 148. ( 10.1073/pnas.200223397) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Murray KA, Rosauer D, McCallum H, Skerratt LF. 2011. Integrating species traits with extrinsic threats: closing the gap between predicting and preventing species declines. Proc. R. Soc. B 278, 1515–1523. ( 10.1098/rspb.2010.1872) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ripple WJ, Wolf C, Newsome TM, Hoffmann M, Wirsing AJ, McCauley DJ. 2017. Extinction risk is most acute for the world's largest and smallest vertebrates. Proc. Natl Acad. Sci. USA 114, 10 678–10 683. ( 10.1073/pnas.1702078114) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Newbold T, Scharlemann JPW, Butchart SHM, Sekercioglu CH, Alkemade R, Booth H, Purves DW. 2013. Ecological traits affect the response of tropical forest bird species to land-use intensity. Proc. R. Soc. B 280, 20122131 ( 10.1016/j.tree.2006.05.007) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hamer KC, Newton RJ, Edwards FA, Benedick S, Bottrell SH, Edwards DP. 2015. Impacts of selective logging on insectivorous birds in Borneo: the importance of trophic position, body size and foraging height. Biol. Conserv. 188, 82–88. ( 10.1016/j.biocon.2014.09.026) [DOI] [Google Scholar]
- 13.Senior MJM, et al. 2012. Trait-dependent declines of species following conversion of rain forest to oil palm plantations. Biodivers. Conserv. 22, 253–268. ( 10.1007/s10531-012-0419-7) [DOI] [Google Scholar]
- 14.Burivalova Z, Lee TM, Giam X, Sekercioglu CH, Wilcove DS, Koh LP. 2015. Avian responses to selective logging shaped by species traits and logging practices. Proc. R. Soc. B 282, 20150164 ( 10.1098/rspb.2015.0164) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Jones MJ, Sullivan MS, Marsden SJ, Linsley MD. 2001. Correlates of extinction risk of birds from two Indonesian islands. Biol. J. Linnean Soc. 73, 65–79. ( 10.1111/j.1095-8312.2001.tb01347.x) [DOI] [Google Scholar]
- 16.Lees AC, Peres CA. 2008. Avian life-history determinants of local extinction risk in a hyper-fragmented neotropical forest landscape. Animal Conserv. 11, 128–137. ( 10.1111/j.1469-1795.2008.00162.x) [DOI] [Google Scholar]
- 17.Solar RRdeC, et al. 2015. How pervasive is biotic homogenization in human-modified tropical forest landscapes? Ecol. Lett. 18, 1108–1118. ( 10.1111/ele.12494) [DOI] [PubMed] [Google Scholar]
- 18.Karp DS, Frishkoff LO, Echeverri A, Zook J, Juárez P, Chan KMA. 2017. Agriculture erases climate-driven β-diversity in Neotropical bird communities. Glob. Change Biol. 24, 338–349. ( 10.1111/gcb.13821) [DOI] [PubMed] [Google Scholar]
- 19.Socolar JB, Gilroy JJ, Kunin WE, Edwards DP. 2016. How should beta-diversity inform biodiversity conservation? Trends Ecol. Evol. 31, 67–80. ( 10.1016/j.tree.2015.11.005) [DOI] [PubMed] [Google Scholar]
- 20.Frishkoff LO, Karp DS, Flanders JR, Zook J, Hadly EA, Daily GC, M'Gonigle LK. 2016. Climate change and habitat conversion favour the same species. Ecol. Lett. 19, 1081–1090. ( 10.1111/ele.12645) [DOI] [PubMed] [Google Scholar]
- 21.dos Anjos L, et al. 2011. Bird species abundance–occupancy patterns and sensitivity to forest fragmentation: implications for conservation in the Brazilian Atlantic forest. Biol. Conserv. 144, 2213–2222. ( 10.1016/j.biocon.2011.05.013) [DOI] [Google Scholar]
- 22.Ravikumar A, Sears RR, Cronkleton P, Menton M, Pérez-Ojeda del Arco M. 2017. Is small-scale agriculture really the main driver of deforestation in the Peruvian Amazon? Moving beyond the prevailing narrative. Conserv. Lett. 10, 170–177. ( 10.1111/conl.12264) [DOI] [Google Scholar]
- 23.Curtis PG, Slay CM, Harris NL, Tyukavina A, Hansen MC. 2018. Classifying drivers of global forest loss. Science 361, 1108–1111. ( 10.1126/science.aau3445) [DOI] [PubMed] [Google Scholar]
- 24.Socolar JB, Valderrama Sandoval EH, Wilcove DS. In press. Overlooked biodiversity loss in tropical smallholder agriculture. Conserv. Biol. 13344 ( 10.1111/cobi.13344) [DOI] [PubMed] [Google Scholar]
- 25.Puhakka M, Kalliola R, Rajasilta M, Salo J. 1992. River types, site evolution and successional vegetation patterns in Peruvian Amazonia. J. Biogeogr. 19, 651–665. ( 10.2307/2845707) [DOI] [Google Scholar]
- 26.Tuomisto H, Ruokolainen K, Kalliola R, Linna A, Danjoy W, Rodriguez Z. 1995. Dissecting Amazonian biodiversity. Science 269, 63–66. ( 10.1126/science.269.5220.63) [DOI] [PubMed] [Google Scholar]
- 27.Fine PVA, García-Villacorta R, Pitman NCA, Mesones I, Kembel SW. 2010. A floristic study of the white-sand forests of Peru. Ann. Missouri Bot. Gard. 97, 283–305. ( 10.3417/2008068) [DOI] [Google Scholar]
- 28.Remsen JV Jr, Parker TA III. 1983. Contribution of river-created habitats to bird species richness in Amazonia. Biotropica 15, 223–231. ( 10.2307/2387833) [DOI] [Google Scholar]
- 29.Alvarez Alonso J, Metz MR, Fine PV. 2013. Habitat specialization by birds in western Amazonian white-sand forests. Biotropica 45, 365–372. ( 10.1111/btp.12020) [DOI] [Google Scholar]
- 30.Pomara LY, Ruokolainen K, Tuomisto H, Young KR. 2012. Avian composition co-varies with floristic composition and soil nutrient concentration in Amazonian upland forests. Biotropica 44, 545–553. ( 10.1111/j.1744-7429.2011.00851.x) [DOI] [Google Scholar]
- 31.Wiley RH. 2010. Alfonso Olalla and his family: the ornithological exploration of Amazonian Peru. Bull. Am. Mus. Nat. Hist. 343, 1–68. ( 10.1206/677.1) [DOI] [Google Scholar]
- 32.Pomara LY, Ruokolainen K, Young KR. 2014. Avian species composition across the Amazon River: the roles of dispersal limitation and environmental heterogeneity. J. Biogeogr. 41, 784–796. ( 10.1111/jbi.12247) [DOI] [Google Scholar]
- 33.Edwards DP, Larsen TH, Docherty TDS, Ansell FA, Hsu WW, Derhe MA, Hamer KC, Wilcove DS. 2010. Degraded lands worth protecting: the biological importance of Southeast Asia's repeatedly logged forests. Proc. R. Soc. B 278, 82–90. ( 10.1098/rspb.2010.1062) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Gilroy JJ, Prescott GW, Cardenas JS, Castañeda PGDP, Sánchez A, Rojas-Murcia LE, Medina Uribe CA, Haugaasen T, Edwards DP. 2015. Minimizing the biodiversity impact of Neotropical oil palm development. Glob. Change Biol. 21, 1531–1540. ( 10.1111/gcb.12696) [DOI] [PubMed] [Google Scholar]
- 35.Schulenberg TS, Stotz DF, Lane DF, O'Neill JP, Parker TA III. 2010. Birds of Peru. Princeton, NJ: Princeton University Press. [Google Scholar]
- 36.Wilman H, Belmaker J, Simpson J, la Rosa dC. 2014. EltonTraits 1.0: species-level foraging attributes of the world's birds and mammals. Ecology 95, 2027 ( 10.1890/13-1917.1) [DOI] [Google Scholar]
- 37.Parker TA III, Stotz DF, Fitzpatrick JW. 1996. Ecological and distributional databases for Neotropical birds Chicago, IL: Chicago University Press. [Google Scholar]
- 38.Devictor V, Clavel J, Julliard R, Lavergne S, Mouillot D, Thuiller W, Venail P, Villéger S, Mouquet N. 2010. Defining and measuring ecological specialization. J. Appl. Ecol. 47, 15–25. ( 10.1111/j.1365-2664.2009.01744.x) [DOI] [Google Scholar]
- 39.Díaz-Alván J, Socolar JB, Álvarez Alonso J. 2017. The avifauna of the río Tigre basin, northern Perú. Ornitol. Neotrop. 28, 11–21. [Google Scholar]
- 40.Socolar JB, et al. 2018. Noteworthy bird records from northeastern Peru reveal connectivity and isolation in the western Amazonian avifauna. Wilson J. Ornithol. 130, 94–111. ( 10.1676/16-082.1) [DOI] [Google Scholar]
- 41.Sullivan BL, Wood CL, Iliff MJ, Bonney RE, Fink D, Kelling S. 2009. eBird: a citizen-based bird observation network in the biological sciences. Biol. Conserv. 142, 2282–2292. ( 10.1016/j.biocon.2009.05.006) [DOI] [Google Scholar]
- 42.Przyborowski J, Wilenski H. 1940. Homogeneity of results in testing samples from Poisson series: with an application to testing clover seed for dodder. Biometrika 31, 313–323. ( 10.1093/biomet/31.3-4.313) [DOI] [Google Scholar]
- 43.Carpenter B, et al. 2017. Stan: a probabilistic programming language. J. Stat. Softw. 76, 1–32. ( 10.18637/jss.v076.i01) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Stan Development Team. In press RStanArm: Bayesian applied regression modeling via Stan.
- 45.Gelman A, Hwang J, Vehtari A. 2013. Understanding predictive information criteria for Bayesian models. Stat. Comput. 24, 997–1016. ( 10.1007/s11222-013-9416-2) [DOI] [Google Scholar]
- 46.Nakagawa S, Johnson PCD, Schielzeth H. 2017. The coefficient of determination R2 and intra-class correlation coefficient from generalized linear mixed-effects models revisited and expanded. J. R. Soc. Interface 14, 20170213 ( 10.1098/rsif.2017.0213) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Gelman A, Rubin DB. 1992. Inference from iterative simulation using multiple sequences. Stat. Sci. 7, 457–472. ( 10.1214/ss/1177011136) [DOI] [Google Scholar]
- 48.Pigot AL, Trisos CH, Tobias JA. 2016. Functional traits reveal the expansion and packing of ecological niche space underlying an elevational diversity gradient in passerine birds. Proc. R. Soc. B 283, 20152013 ( 10.1098/rspb.2015.2013) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Pigot AL, Tobias JA. 2013. Species interactions constrain geographic range expansion over evolutionary time. Ecol. Lett. 16, 330–338. ( 10.1111/ele.12043) [DOI] [PubMed] [Google Scholar]
- 50.Räsänen M, Neller R, Salo J, Jungner H. 1992. Recent and ancient fluvial deposition systems in the Amazonian Foreland Basin, Peru. Geol. Mag. 129, 293–306. ( 10.1017/S0016756800019233) [DOI] [Google Scholar]
- 51.Robinson SK. 1997. Birds of a Peruvian oxbow lake: populations, resources, predation, and social behavior. Ornithol. Monogr. 48, 613–639. ( 10.2307/40157558) [DOI] [Google Scholar]
- 52.Tuomisto H, Ruokolainen K, Yli-Halla M. 2003. Dispersal, environment, and floristic variation of western Amazonian forests. Science 299, 241–244. ( 10.1126/science.1078037) [DOI] [PubMed] [Google Scholar]
- 53.Stattersfield AJ, Crosby MJ, Long AJ, Wege DC. 1998. Endemic bird areas of the world: priorities for biodiversity conservation. Cambridge, UK: Birdlife International. [Google Scholar]
- 54.Socolar JB, Wilcove DS. 2019. Data from: Forest-type specialization strongly predicts avian responses to tropical agriculture Dryad Digital Repository. ( 10.5061/dryad.6944855) [DOI] [PMC free article] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Citations
- Socolar JB, Wilcove DS. 2019. Data from: Forest-type specialization strongly predicts avian responses to tropical agriculture Dryad Digital Repository. ( 10.5061/dryad.6944855) [DOI] [PMC free article] [PubMed]
Supplementary Materials
Data Availability Statement
Data available from the Dryad Digital Repository: https://doi.org/10.5061/dryad.6944855 [54].






