Abstract
Induction of vancomycin resistance in vancomycin-resistant enterococci (VRE) involves replacement of the d-Ala-d-Ala terminus of peptidoglycan (PG) stems with d-Ala-d-Lac, dramatically reducing the binding affinity of vancomycin to lipid II. Effects from vancomycin resistance induction on Enterococcus faecalis (ATCC 51299) were characterized using a combined solid-state NMR and LC-MS analysis. Solid-state NMR directly measured the total amounts of d-Lac and l,d-Ala metabolized from [2-13C]pyruvate, accumulated Park’s nucleotide, and changes to the PG bridge-linking density during early exponential growth phase (OD660nm 0.4) in intact whole cells of VRE. Large accumulation of depsipeptide substituted Park’s nucleotide consistent with the inhibition of transglycosylation step of PG biosynthesis during the initial phase of vancomycin resistance was observed, while no changes to the PG bridge-linking density following the induction of vancomycin resistance was detected. This indicated that the PG bridge attachment to lipid II by the peptidyl transferases was not inhibited by the d-Ala-d-Lac substituted PG-stem structure in VRE. Compositions of mutanolysin-digested isolated cell walls of VRE grown with and without vancomycin resistance induction were determined by LC-MS. Muropeptides with PG stems terminating in d-Ala-d-Lac were found only in VRE grown in presence of vancomycin. Percentages of muropeptides with a pentapeptide stem terminating in d-Ala-d-Lac for VRE grown in presence of vancomycin were 26% for mid-exponential (OD660nm 0.6) and 57% for stationary growth phase (OD660nm 1.0). These high percentages indicate that d-Ala-d-Lac substituted lipid II was efficiently utilized for PG biosynthesis in VRE.
Keywords: Vancomycin, REDOR, E. faecalis, Peptidoglycan, solid-state NMR
Graphical Abstract
Vancomycin-resistant enterococci (VRE) are opportunistic pathogens commonly found in the gastrointestinal tract. They can translocate across the intestinal lining into bloodstream and cause bacteremia, sepsis, endocarditis, and other potentially fatal infections. With increased vancomycin usage, various enterococcal infections have become common in nosocomial settings.1 Historically, Enterococcus faecalis alone has been responsible for 80-90% of all enterococcal infections.2 E. faecalis is one of the leading nosocomial agents for catheter-associated infections due to its ease of transmission through surface contacts. One of many factors that contribute to persistence of E. faecalis in healthcare settings is its robust cell wall approximately 30 nm in thickness. The primary component of this cell wall is peptidoglycan (PG), which is assembled from repeating subunits to form the lattice structure.3, 4
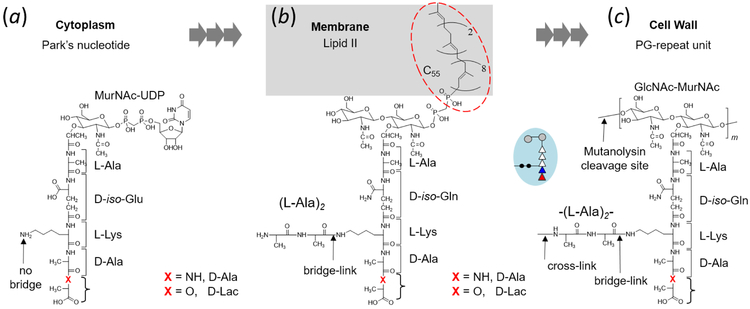
Biosynthesis of these PG subunits is carried out in three stages. The first stage, which takes place in the cytoplasm, is the sequential addition of amino acids to UDP-MurNAc for the synthesis of Park’s nucleotide (Scheme 1a). Resulting Park’s nucleotide is the cytoplasmic monosaccharide precursor of PG with the amino acid PG-stem structure (l-Ala-d-iso-Glu-l-Lys-d-Ala-d-Ala) attached to MurNAc. The second stage of PG biosynthesis is carried out on the interior side of cytoplasmic membrane with the transfer of Park’s nucleotide to the membrane transporter C55 (undecaprenyl phosphate). During this stage, PG disaccharide (GlcNAc-MurNAc) is first assembled by the addition of N-acetylglucosamine (GlcNAc), then two l-Ala of the crosslinking bridge are sequentially added by peptidyl transferases BppA1 and BppA25 to the ε-nitrogen side chain of l-Lys of the PG stem to form lipid II (Scheme 1b). The final stage of PG assembly is carried out by translocating lipid II to the exterior side of cytoplasmic membrane. Transglycosylase polymerizes PG disaccharides of lipid II to extend the glycan chain, and transpeptidase interconnects neighboring glycan chains through cross-links.6 A cross-link is the peptide bond between the N-terminus of l-Ala from the bridge in one PG unit to the C-terminus of penultimate d-Ala from the stem on a neighboring glycan chain. The terminal d-Ala (5th amino acid) from the acceptor stem is cleaved upon cross-link formation.
Scheme 1.
Peptidoglycan biosynthesis in Enterococcus faecalis. (a) The first stage of PG biosynthesis takes place in the cytoplasm with Park’s nucleotide (UDP-MurNAc-l-Ala-D-iso-Glu-l-Lys-d-Ala-d-Ala) as the end product. In vancomycin-resistant E. faecalis, the terminal d-Ala of Park’s nucleotide is replaced by d-Lac. (b) The second stage of PG biosynthesis occurs on the interior side of bacterial cytoplasmic membrane, where UDP-MurNAc-stem from Park’s nucleotide is transported by the lipid transporter C55 to form lipid II (N-acetylglucosamine-N-acetyl-muramyl-pentapeptide-pyrophosphoryl-undecaprenol). Attachment of l-Ala-l-Ala bridge to the PG-stem is carried out by peptidyl transferases BppA1 and BppA2. d-iso-Glu is amidated to d-iso-Gln. (c) The final stage of PG biosynthesis is carried out on the external side of cytoplasmic membrane with incorporation of the PG-repeat unit into the cell wall.
Vancomycin resistance is conferred upon VRE by substitution of the d-Ala-d-Ala terminus of the PG stem, the vancomycin binding site, with a depsipeptide d-Ala-d-Lac.7, 8 The substitution replaces the amide of final d-Ala with an oxygen from d-Lac, which introduces an electrostatic repulsion between oxygen in the ester bond of d-Ala-d-Lac to carbonyl from the fourth residue of vancomycin, and this reduces vancomycin’s binding affinity to the altered stem by a thousand fold.9, 10 VRE with the d-Ala-d-Lac PG-stem modification exhibit high-levels of vancomycin resistance with minimal inhibitory concentrations (MIC) that often exceed 100 μg/mL.11
E. faecalis strain ATCC 51299 used for this study is VRE of VanB type12 with the plasmid that includes vanRB, vanSB, vanHB, vanB, vanXB, vanYB, and vanW.13 VanSB and VanRB are part of a two-component regulatory system, where VanSB is a membrane-bound sensor kinase that as a response to vancomycin phosphorylates the response-regulator protein VanRB,14 which is a transcription factor that activates downstream elements vanHB, vanB, vanXB, and vanYB.13 VanHB and VanB are involved in biosynthesis of d-Ala-d-Lac, where VanHB is a dehydrogenase that catalyzes production of d-Lac from pyruvate,15 and VanB is a ligase that joins d-Lac and d-Ala together to form a depsipeptide.16 Biosynthesis of d-Ala-d-Lac is accompanied by breaking down of d-Ala-d-Ala by VanXB and VanYB. VanXB is a d,d-dipeptidase that cleaves the dipeptide d-Ala-d-Ala,17 while VanYB is a d,d-carboxypeptidase that removes terminal d-Ala from the PG stem of Park’s nucleotide.18, 19 The effect of both peptidases is rapid degradation of d-Ala-d-Ala in the PG stem of Park’s nucleotide, which is replaced by the depsipeptide d-Ala-d-Lac.
Although d-Ala-d-Lac substituted Park’s nucleotide has been identified from cytoplasmic fractions of vancomycin-resistant E. faecalis20 and E. faecium,21 d-Ala-d-Lac terminated PG stems have not yet been observed from isolated cell walls of E. faecalis22 and E. faecium.23 Their absence suggested that d-Ala-d-Lac substituted lipid II is not readily utilized for the cell wall biosynthesis and/or d-Lac is cleaved from PG stems in the cell wall by highly active d,d-carboxypeptidases.22, 23 d-Ala-d-Lac substituted PG stem is also thought to interfere with the attachment of bridge structure to lipid II, as treatment of vancomycin-resistant Staphylococcus aureus of VanA type (strain COLVA) with vancomycin resulted in increased concentrations of muropeptide species missing the bridge structure.24 In vitro assay has shown that addition of d-Ala-d-Lac substituted lipid II strongly inhibits FemX activity,25 where FemX is the peptidyl transferase that catalyzes the first committed step in the attachment of pentaglycine bridge to lipid II in S. aureus. In E. faecalis, l-Ala-l-Ala bridge attachment is carried out by peptidyl transferases BppA1 and BppA25 which belong to the same family of Fem proteins found in S. aureus.5, 26 Hence, d-Ala-d-Lac terminated lipid II is likely to be a poor substrate for peptidyl transferases in E. faecalis.
In this study, we characterized changes in the PG composition of vancomycin-resistant E. faecalis (ATCC 51299) grown with and without vancomycin using solid-state NMR and liquid chromatography-mass spectrometry (LC-MS). Solid-state NMR was used to directly measure in intact cells the PG bridge-link density and Park’s nucleotide accumulation in E. faecalis using l-[1-13C]Ala and l-[ε-15N]Lys, and characterize [2-13C]pyruvate utilization for the biosynthesis of d-[2-13C]Lac (Scheme 2). LC-MS analysis was performed on mutanolysin digested isolated cell walls of VRE. Mutanolysin is an N-acetylmuramidase which cleaves the MurNAc β1-4 glycosidic linkage in PG (Scheme 1), and resulting muropeptide fragments were analyzed by LC-MS to determine the incorporation efficiency of d-Ala-d-Lac substituted lipid II in the cell wall of VRE.
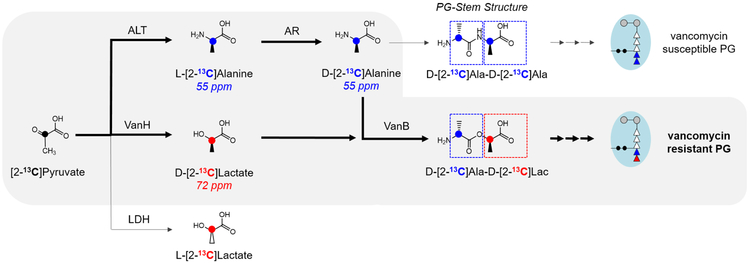
Scheme 2.
[2-13C]Pyruvate metabolism in vancomycin-resistant E. faecalis. In absence of vancomycin, [2-13C]pyruvate is utilized for the biosynthesis of l-[2-13C]Ala by alanine transaminase (ALT), then to d-[2-13C]Ala by alanine racemase (AR). [2-13C]pyruvate metabolism to l-[2-13C]Lac by lactate dehydrogenase (LDH) does not occur in VRE under aerobic growth. Following the induction of vancomycin resistance, VanHB catalyzes the biosynthesis of d-[2-13C]Lac.
EXPERIMENTAL METHODS
Bacterial growth and isotope labeling.
From overnight starter culture of vancomycin-resistant E. faecalis (ATCC 51299), 1% inoculum of the starter culture was added to one-liter flasks containing 330 mL of sterile Enhanced Standard Medium (ESM). ESM was modified from SASM27 to enable both staphylococcal and enterococcal bacteria to grow.28 PG bridge-links in intact E. faecalis were 13C-15N pair labeled by growing them in ESM where natural abundance l-Ala and l-Lys were replaced by l-[1-13C]Ala and l-[ε-15N]Lys. Alanine racemase inhibitor alaphosphin (5 μg/mL) and d-Ala (0.1 g/L) were added to prevent racemic scrambling of l-[1-13C]Ala and provide the source of d-Ala. To determine VanHB activity in VRE, E. faecalis were grown in ESM supplemented with Na-[2-13C]pyruvate (0.1 g/L). Vancomycin resistance was induced during the exponential growth (OD660nm 0.3) by addition of vancomycin to final concentration of 10 μg/mL. Cells were harvested after 1 hour of growth by centrifugation at 10,000 g for 10 minutes at 4 °C in Sorvall GS-3 rotor. Resulting pellets were washed twice with 100 mL of cold deionized water, then resuspended in approximately 25 mL of deionized water, followed by rapid freezing and lyophilization.
Rotational-echo double resonance (REDOR) NMR.
REDOR is a solid-state NMR method used to recouple dipolar interactions under magic-angle spinning (MAS).29, 30 REDOR experiment consists of two parts. In the first part, polarization transfer from the protons prepares 13C magnetization, and the spectrum is collected after N rotor periods without dipolar evolution; this observed 13C spectrum is referred to as the S0 spectrum. In the second part of REDOR experiment, 13C-15N dipolar coupling is reintroduced by applying rotor-synchronized dephasing π pulses to 15N nuclei. 15N pulses invert the sign of dipolar coupling and interfere with the MAS spatial refocusing, which result in net dipolar phase accumulation and reduced 13C signal intensity; this 13C spectrum with the dephasing pulses is referred to as the dephased spectrum with N rotor cycles of dipolar evolution. From these two spectra, the difference spectrum (ΔS = S - S0) with intensities only from 13C dipolar coupled to 15N is derived. Normalized REDOR difference (ΔS / S0) is the direct measure of dipolar coupling, and this quantity was calculated using modified Bessel function expressions given by Mueller et al.31 and de la Caillerie and Fretigny32 for an IS spin-1/2 pair.
Solid-state NMR spectrometer.
Solid-state NMR analyses were performed on lyophilized intact cells of E. faecalis packed into Chemagnetics/Varian 7-mm zirconia rotors. REDOR was performed at 4.7-T field (proton radio frequency of 200 MHz) provided by 89-mm bore Oxford (Cambridge, England) superconducting solenoids utilizing a four-frequency transmission-line probe with 17-mm long, 8.6-mm inside-diameter analytical coil and Chemagnetics/Varian ceramic stator. Rotors were spun at 5000 Hz with speed under active control to within ±2 Hz. π pulse lengths were 10 μs for both 13C and 15N. Matched proton-carbon cross-polarization transfers were made in 2 ms at 50 kHz. Proton dipolar decoupling was 98 kHz during data acquisition. Standard XY-8 phase cycling was used for all refocusing and dephasing pulses.33 All measurements were carried out at ambient room temperature.
Cell wall isolation.
Overnight cultures of E. faecalis were used to inoculate brain-heart infusion (BHI) media containing vancomycin (6 μg/mL). Cultures were grown for 24 hours at 37°C with 180 RPM shaking. Cells were harvested at exponential (OD600nm ~0.6) and stationary growth (OD600nm 1.0) phases by centrifugation (Beckman Coulter Allegra X-15R, SX4750 rotor) at 4750 rpm for 12 min at 4°C. Autolysins were inactivated by immersing pellets in boiling water baths for 30 minutes. Collected samples were divided equally into multiple microcentrifuge tubes with each containing approximately 400 μL (0.65 g) of 0.5 mm glass beads. Cells were agitated (Disruptor Genie, Scientific Industries) with beads for 15 minutes in alternating cycles of 1 minute of bead-beating and 1 minute of rest, giving a total of 8 cycles of 1-minute bead-beating and 7 cycles of 1-minute rest. Following the bead-beating, disrupted cells were resuspended in phosphate buffered saline (PBS) containing 8 mg/mL NaCl, 0.2 mg/mL KCl, 1.44 mg/mL Na2HPO4, and 0.24 mg/mL KH2PO4. Beads were filtered out from the cells using Steriflip 20 μm nylon net vacuum filter unit (EMD Millipore). Disrupted cells were washed once in PBS, boiled in 2% sodium dodecyl sulfate solution for 30 minutes, and washed 5 times with autoclaved deionized water. Resulting isolated cell walls were suspended in 20 mM Tris pH 8.0 buffer. To the samples, 200 μg of DNase (Sigma-Aldrich) was added and the mixture incubated for 24 hours at 37°C and 180 RPM. Trypsin (300 μg) (Sigma-Aldrich) was then added to the digest, and the sample incubated for additional 24 hours at 37°C and 180 RPM. Following the digestions, samples were centrifuged to spin down the isolated cell wall. Resulting pellets were resuspended in 20 mM Tris buffer, frozen, and lyophilized.
Mutanolysin digestion of isolated cell wall.
Lyophilized cell walls suspended in 2 mL of 20 mM Tris pH 8.0 buffer were first digested with 100 units of mutanolysin (Sigma-Aldrich) for 24 hours at room temperature, then additional 100 units were added for 24 more hours of digestion. Digested cell wall fragments were centrifuge filtered through 30 kDa molecular weight cutoff filters at 14800 RPM. Filtered muropeptides were reduced using sodium borohydride (10 mg/mL) in 1 mL 0.5 M borate pH 9.0 at room temperature for 30 minutes. Reduction was quenched by addition of 120 μL of 85% phosphoric acid, and the samples were frozen and lyophilized.
LC-MS analysis.
Mutanolysin-digested cell wall (20-40 mg) in 500 μL of 20 mM Tris pH 8.0 buffer was diluted 1:10 in methanol with 0.1% formic acid. Waters Synapt G2 High Definition Mass Spectrometer (HDMS) Time-of-Flight (TOF) mass analyzer and Waters C18 ACUITY Ultra Performance Liquid Chromatography (UPLC) were used to analyze mutanolysin digested fragments of peptidoglycan. Chromatographic separation of mutanolysin-digested peptidoglycan was carried out by injecting 1 μL of the sample from a 5 μL sample loop to the column under isocratic condition of 99% buffer A (99.8% anhydrous methanol with 0.1% formic acid) and 1 % buffer B (100% acetonitrile) for 5 minutes. Waters nanoACQUITY C18 reverse-phase column (75 μm X 100 mm, 1.7 μm beads with 130 Å pore size) with nanoACQUITY C18 trap column (180 μm X 20 mm, 5 μm beads with 100 Å pore size) was used for chromatographic separation. First, a linear gradient to 50% buffer B was applied for 60 minutes. The column was subsequently regenerated under isocratic condition with 85% buffer B for 5 minutes, a linear gradient to 98% buffer A for 1 minute, then isocratic at 98% buffer A for 23 minutes. The flow rate was kept constant (0.6 μL/min) throughout the analysis.
Resulting eluents were analyzed by Waters Synapt G2 HDMS-TOF mass analyzer operating in positive ion mode with the fragments ionized through electrospray ionization (ESI) with spray voltage of 35 V and capillary voltage of 3.5 kV. The mass analyzer was optimized for m/z range of 100-2000, with Fibrinopeptide B (Glu-Fib) as the lock mass for calibration. MS/MS fragmentation was accomplished through collision-induced dissociation with nitrogen gas at 120°C and 2.00 Bar. Transfer collision energy was 5 V, and trap collision energy was 30 V. MassLynx Mass Spectrometry Software (Waters) was used for data analysis. Values for calculating the exact mass for mutanolysin-digested PG fragments were obtained from the Molecular Mass Calculator on the Biological Magnetic Resonance Data Bank (www.bmrb.wisc.edu). The difference between observed and calculated masses needed to be less than 10 ppm for ions to be identified as positive matches.
RESULTS AND DISCUSSION
Park’s nucleotide accumulation in vancomycin-treated VRE.
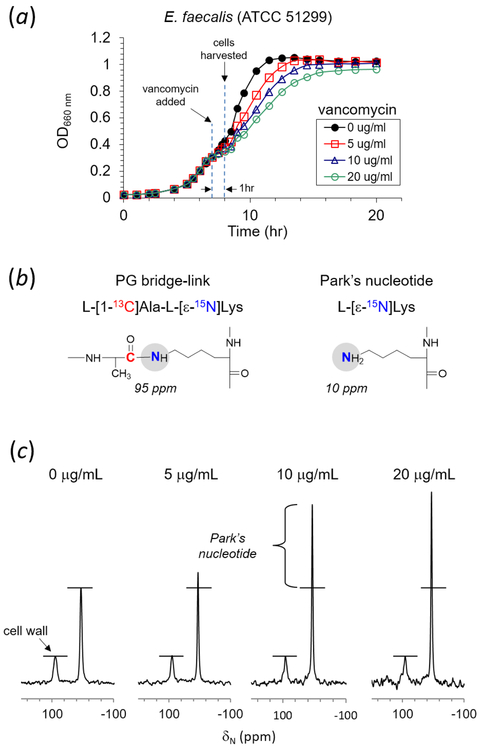
Growth curves of VRE (ATCC 51299) grown in ESM in presence of vancomycin as monitored by measuring the optical density at 660 nm (OD660nm) are shown in Figure 1a. ESM was supplemented with natural abundance d-Ala (0.1 g/L) and alaphosphin (5 μg/mL). Alaphosphin (l-alanyl-l-1-aminoethylphosphonic acid) is an alanine racemase inhibitor that prevents racemic scrambling between l-Ala and d-Ala. The MIC of alsphosphin against E. faecalis in the absence of supplemental d-Ala was < 5 μg/mL. Stock solutions of vancomycin were added during mid-growth (OD600nm 0.3) to final concentrations of 0, 5, 10 and 20 μg/mL. A dose-dependent growth delay immediately following the vancomycin addition was observed, but even high concentration of vancomycin (20 μg/mL) did not fully inhibit VRE growth.
Figure 1.
(a) Growth curves of vancomycin resistant E. faecalis (ATCC 51299) in ESM containing l-[1-13C]Ala and l-[ε-15N]Lys. Growth was monitored by measuring optical density at 660 nm (OD660nm). Vancomycin was added to final concentrations of 0, 5, 10, and 20 μg/mL during growth at OD660nm 0.3. Cells were harvested at OD660nm 0.4 for 15N-CPMAS NMR analysis. (b) Incorporation of l-[ε-15N]Lys into the PG bridge-link is visible as a lysyl-amide at 95 ppm, and into Park’s nucleotide as a lysyl-amine at 10 ppm in 15N-CPMAS spectra. (c) 15N-CPMAS spectra of E. faecalis labeled with l-[1-13C]Ala and l-[ε-15N]Lys. Spectra are normalized to 95-ppm intensity. l-[ε-15N]Lys incorporated into PG bridge-linked resonates at 95 ppm, and l-[ε-15N]Lys into proteins and Park’s nucleotide at 10 ppm. VRE grown in presence of vancomycin show Park’s nucleotide accumulation, indicating that vancomycin inhibited the transglycosylation step of PG biosynthesis. 15N-CPMAS spectra of VRE grown in vancomycin concentrations of 0, 5, 10, and 20 μg/mL are results of 74092, 80000, 70512, and 80000 accumulated scans, respectively. The magic angle spinning was at 5000 Hz. All measurements were carried out at ambient room temperature.
To determine the effects of vancomycin on PG biosynthesis, 15N-cross polarization at magic-angle spinning (CPMAS) NMR measurements were performed on intact cells of VRE grown in ESM containing l-[1-13C]Ala and l-[ε-15N]Lys. ESM was supplemented with alaphosphin (5 μg/mL), to prevent racemic scrambling of l-[1-13C]Ala to d-[1-13C]Ala, and provided with natural abundance d-Ala (0.1 g/L) to prevent inhibition of E. faecalis growth by alaphosphin.34 Vancomycin was added during growth at OD600nm 0.3, and the culture harvested for analysis approximately one hour following addition of vancomycin at OD660nm 0.4. Exogenous l-[ε-15N]Lys provided in ESM was readily taken up by E. faecalis and found incorporated into PG without scrambling. The l-[ε-15N]Lys incorporation efficiency into PG determined by mass spectrometry was 90%. In 15N-CPMAS echo spectra (Fig. 1c), l-[ε-15N]Lys incorporation to Park’s nucleotide as a lysyl-amine was observed at 10 ppm, and incorporation to PG-repeat units in the cell wall as a lysyl-amide was at 95 ppm.34 The 15N-natural abundance contribution to lysyl-amide and amine intensities are minor.35 Since the 15N lysyl-amide peak is unique to crosslinked PG, its intensity is directly proportional to the total number of PG-repeat units found in the cell wall.
15N-CPMAS echo spectrum of VRE treated with vancomycin (10 μg/mL) showed near doubling of the lysyl-amine peak intensity at 10 ppm from accumulation of Park’s nucleotide (Fig. 1c). The amount of Park’s nucleotide was estimated by back-extrapolating lysyl-amide and amine peak integrals to zero-contact time in order to account for differences in relaxation in rotating frame and cross-polarization transfer rates.34, 36 The extrapolated lysyl-amine to lysyl-amide peak integral ratio for VRE grown in absence of vancomycin was 3.2 to 1.0, and for VRE grown in presence of vancomycin (10 μg/mL) it was 4.8 to 1.0. Thus the relative amount of Park’s nucleotide accumulation in the cytoplasm of VRE following induction of vancomycin resistance is 1.6 (4.8 - 3.2) times the total number of PG-repeat units in the cell wall of VRE. The degree of Park’s nucleotide accumulation in VRE is comparable to those observed for vancomycin-susceptible Staphylococcus aureus when treated with vancomycin35 or oritavancin.37 The buildup of Park’s nucleotide in VRE is consistent with vancomycin inhibition of the transglycosylation step of PG biosynthesis.
Increased d-Lac biosynthesis in VRE.
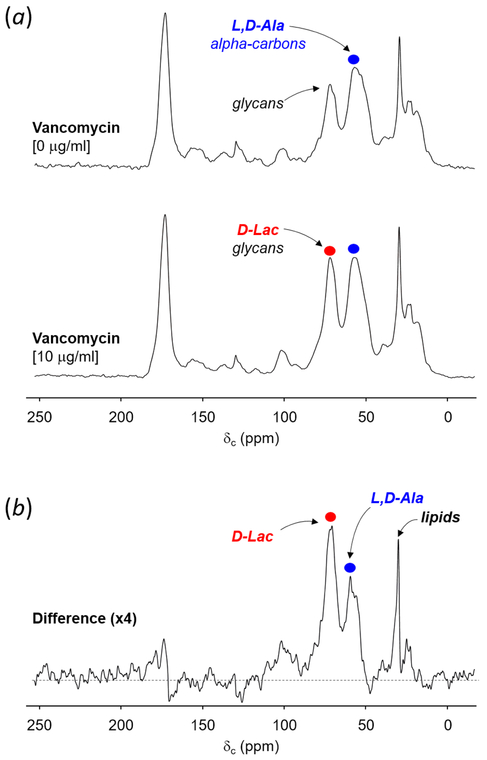
Pyruvate metabolism in VRE was investigated by growing E. faecalis in ESM containing natural abundance d-glucose (10 g/L) supplemented with sodium-[2-13C]pyruvate (0.1 g/L). At OD660nm 0.3, the culture was divided into two aliquots of 500 mL. Vancomycin was added to final concentration of 10 μg/mL to one aliquot, and cells were grown for 1 hour and harvested. 13C-CPMAS NMR was used to monitor the [2-13C]pyruvate utilization for [2-13C]Ala and [2-13C]Lac biosynthesis in intact cells. 13C-chemical shift assignments for [2-13C]pyruvate (206 ppm), [2-13C]Ala (55 ppm), and [2-13C]Lac (72 ppm) were determined based on the [2-13C]pyruvate metabolism in Propionibacterium freudenreichii.38 Absence of 206-ppm peak in the 13C-CPMAS spectrum (Fig. 2a, top) shows that [2-13C]pyruvate is readily taken up and metabolized by VRE.
Figure 2.
(a) 13C-CPMAS spectra of whole cells of E. faecalis grown in ESM supplemented with sodium-[2-13C]pyruvate (0.1 g/L). Vancomycin was added to final concentration of 10 μg/mL to one aliquot, and cells were grown for 1 hour and harvested. The 206-ppm peak assigned to [2-13C]pyruvate is absent from the 13C-CPMAS spectrum of VRE grown without vancomycin, due to [2-13C]pyruvate being fully utilized for the biosynthesis of [2-13C]Ala visible at 55 ppm (blue circle) and MurNAc at 72 ppm, which overlaps with the natural abundance 13C from glycans in PG. 13C-CPMAS spectra of VRE grown with and without vancomycin are results of 40000 and 80000 accumulated scans respectively. The magic angle spinning was at 5000 Hz. (b) The difference spectrum was obtained by subtracting 13C-CPMAS spectrum of VRE grown without vancomycin from the spectrum of VRE grown with vancomycin, and it shows increased d-[2-13C]Lac biosynthesis in VRE consistent with VanHB activity.
Multiple metabolic pathways for [2-13C]pyruvate exist in VRE. In absence of vancomycin, as E. faecalis lack the tricarboxylic cycle,39 [2-13C]pyruvate is readily routed for the biosynthesis of l-[2-13C]Ala by alanine transaminase, which in turn is converted into d-[2-13C]Ala by alanine racemase. This preferential utilization of pyruvate for alanine biosynthesis was evident by large 55-ppm peak in the 13C-CPMAS spectrum (Fig. 2a, top). Another metabolic pathway for [2-13C]pyruvate is the biosynthesis of MurNAc, where [2-13C]pyruvate is phosphorylated to phosphoenol-[2-13C]pyruvate, transferred to UDP-GlcNAc by MurA40, 41 as carboxyvinyl, then reduced to form UDP-MurNAc. UDP-MurNAc is the precursor for Park’s nucleotide and [2-13C]pyruvate is incorporated into MurNAc as lactate that appears as a 72 ppm peak in the 13C-CPMAS spectrum (Fig. 2a, top). Finally, lactate dehydrogenase can convert [2-13C]pyruvate to l-[2-13C]Lac; however, this conversion only occurs during anaerobic growth and is not observed under aerated growth conditions.39 Therefore, the 72-ppm peak in the 13C-CPMAS spectrum of VRE grown in absence of vancomycin (Fig. 2a, top) is from the 13C-labeled lactate moiety in MurNAc and contribution from 13C-natural abundance. The source of 13C-natural abundance is d-glucose (10 g/L) found in ESM, which is one hundred times the amount of 13C-labeled Na-[2-13C]pyruvate (0.1 g/L).
13C-CPMAS spectrum of VRE treated with vancomycin is shown in Fig. 2a (bottom). The increase in 72-ppm peak intensity correlates with the induction of vanHB in VRE, which is a dehydrogenase that converts [2-13C]pyruvate to d-[2-13C]Lac. Metabolic changes associated with pyruvate utilization in VRE following induction of vancomycin resistance are clearly visible in the difference spectrum (Fig. 2b). The difference spectrum was obtained by subtracting the 13C-CPMAS spectrum of VRE grown without vancomycin (Fig. 2a, top) from the spectrum of VRE grown with vancomycin (Fig. 2a, bottom). The spectral subtraction removes contributions from natural abundance 13C and basal [2-13C]pyruvate metabolism prior to the induction of vancomycin resistance. In the difference spectrum, 72-ppm intensity for d-[2-13C]Lac is approximately twice that of 55 ppm assigned for d,l-[2-13C]Ala. This ratio indicates that VanHB activity highly increases during vancomycin-resistance induction, and suggests increase in the amount of depsipeptide-substituted Park’s nucleotide in VRE.
PG bridge-link density in VRE.
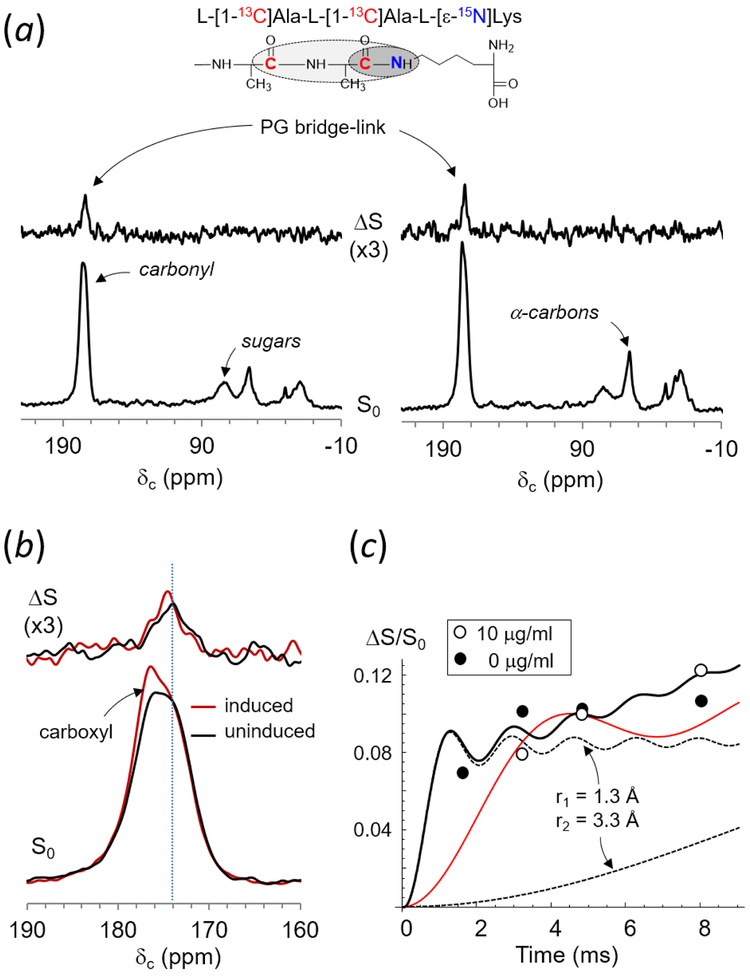
To determine the inhibitory effect of d-Ala-d-Lac on the attachment of PG bridge to lipid II,24, 25 the density of PG bridge-link in intact cells of VRE was measured using solid-state NMR. Figure 3 shows 13C{15N} rotational-echo double resonance (REDOR) NMR spectra at 4.8 ms dipolar evolution for whole cells of E. faecalis grown in ESM (Fig. 3a, left) and with vancomycin added to 10 μg/mL concentration (Fig. 3a, right). ESM contained isotope labels l-[1-13C]Ala and l-[ε-15N]Lys, and was supplemented with alaphosphin (5 μg/mL) and d-Ala (0.1 g/L). Growth in isotopically enriched ESM pair labeled 13C-15N of PG bridge-links (Fig. 3a, inset) in the cell wall of VRE. Full-echo (S0) spectra are shown at the bottom, and difference (ΔS) spectra are at the top. S0 spectra are normalized to the intensity of alanyl-carbonyl carbon from l-[1-13C]Ala at 174 ppm. Overlaid and enlarged S0 spectra centered at 174 ppm (Fig. 3b) show change in the lineshape for VRE grown in presence of vancomycin with the appearance of a 177-ppm shoulder. The shoulder is due to increased contribution from the alanyl-carboxyl carbon as result of VanXB (d,d-dipeptidase) and VanYB (d,d-carboxypeptidase) activities.
Figure 3.
(a) 13C{15N}REDOR of whole cells of E. faecalis labeled with l-[1-13C]Ala and l-[ε-15N]Lys at 4.8 ms dipolar evolution grown in absence (left) and presence of vancomycin at 10 μg/mL (right). 13C{15N}REDOR spectra of VRE grown in vancomycin concentrations of 0 and 20 μg/mL are results of 75248 and 320000 accumulated scans respectively. The magic angle spinning was at 5000 Hz. (b) Enlarged overlaid S0 and ΔS spectra centered at 174 ppm for VRE grown with vancomycin (red), and without (black). ΔS 174-ppm peak intensity, which was directly proportional to the bridge-link density, was independent of induction of vancomycin resistance in VRE. (c) 13C{15N}REDOR dephasings (ΔS/S0) of alanyl-carboxyl carbon at 174 ppm. Solid line is the calculated REDOR dephasing curve for two 13Cs in l-[1-13C]Ala-l-[1-13C]Ala bridge that are dephased by the 15N of l-[ε-15N]Lys with 13C-15N distances of 1.3 and 3.3 Å (dotted lines). 13C{15N}REDOR dephasing is highly selective for the PG-bridge structure, as demonstrated by the calculated dephasing curve for 13C-15N distances corresponding to 2.0 and 4.0 Å (red curve), which does not fit observed dephasings.
In 13C{15N} REDOR ΔS spectra at 4.8 ms dipolar evolution (Fig. 3a), only 13Cs from l-[1-13C]Ala-l-[1-13C]Ala bridge dipolar coupled to the ε-15N sidechain in l-[ε-15N]Lys within 4 Å are detected. Figure 3c shows 13C{15N}REDOR dephasing of 174 ppm plotted as a function of dipolar evolution time for VRE grown in absence (solid circle) and presence of vancomycin (open circle). Observed dephasings fit to a calculated REDOR dephasing curve for two distances of 1.3 and 3.3 Å (black solid line), but not to a single 13C-15N distance (not shown). Calculated dephasing curves for 13C-15N distances of 1.3 and 3.3 Å are shown as dotted black lines. These measured distances correspond to intra-molecular distances between 13Cs in (l-[1-13C]Ala)2-bridge structure to 15N of l-[ε-15N]Lys on the same PG-repeat unit. As inter-molecular 13C-15N couplings that exceed 4 Å distance do not contribute to the observed dephasing, 13C{15N} REDOR dephasings are highly specific for the PG bridge-link. A calculated REDOR dephasing curve for 13C-15N distances of 2.0 and 4.0 Å, shown as a red solid line, does not fit observed dephasings. Thus, the ΔS 174-ppm intensity at 4.8 ms is directly proportional to the total number of PG bridge-links in intact whole cells of VRE. Since the ΔS 174-ppm intensity does not change with the addition of vancomycin to VRE, induction of vancomycin resistance does not affect the PG bridge-link density. Therefore, the d-Ala-d-Lac substituted PG stem in lipid II did not inhibit sequential additions of l-Ala-l-Ala bridge structure by peptidyl transferases BppA1 and BppA2 in vancomycin-resistant E. faecalis.
Quantification of d-Ala-d-Lac terminated muropeptides in VRE cell walls by LC-MS.
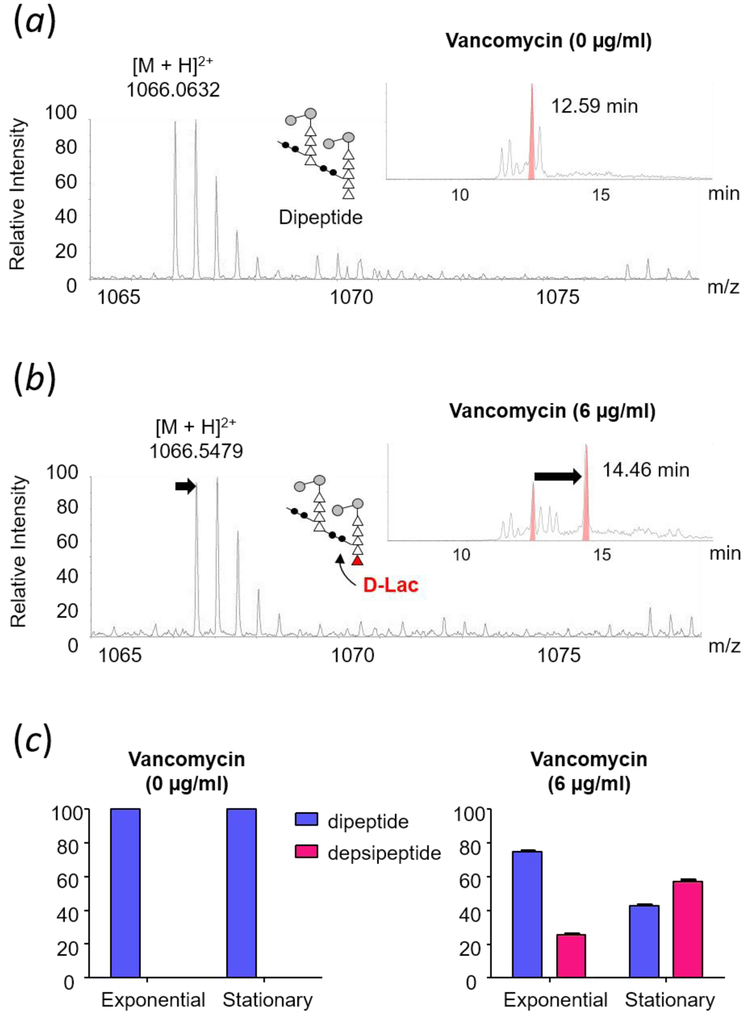
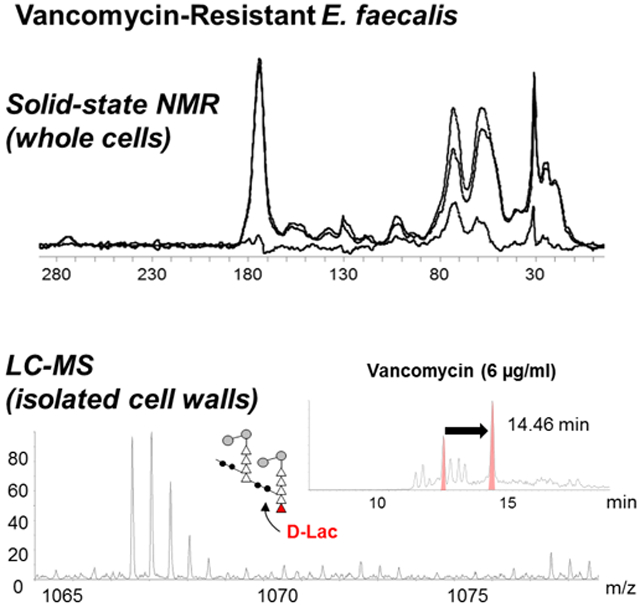
Although d-Ala-d-Lac terminated PG precursors have been routinely observed in the cytoplasm fraction of VRE grown in presence of vancomycin, depsipeptide terminated PG-stem structures have never been observed from isolated cell walls of vancomycin-resistant E. faecalis22 and E. faecium.23 This absence has led to suggestions that lipid II with a d-Ala-d-Lac stem is not readily incorporated into the cell wall or that d-Lac is cleaved from lipid II during PG biosynthesis.23 To determine incorporation of d-Ala-d-Lac terminated PG-stem structures into VRE’s cell wall, LC-MS analysis was performed on mutanolysin-digested cell walls of vancomycin-resistant E. faecalis (ATCC 51299). Figure 4 shows mass spectra for doubly-charged PG dimer from VRE at stationary growth phase (a) and with vancomycin added (b). The m/z of 1066.0632 in Fig. 4a corresponds to the chemical formula C87H153N21O402+, which is a PG dimer with pentapeptide stem terminating in d-Ala-d-Ala, while the m/z of 1066.5479 in Fig. 4b corresponds to the chemical formula C87H152N20O412+ for a PG dimer with pentapeptide stem terminating in d-Ala-d-Lac. The difference in mass for a PG dimer with the depsipeptide substitution is an increase of 1 Da, which shifts the m/z by 0.50 (Fig. 4b). The d-Ala-d-Lac modified PG dimer is chromatographically well resolved from its unmodified counterpart, as shown in the select ion chromatogram (Fig. 4b insets) by retention time increase of 2 minutes. In general, all d-Ala-d-Lac substituted muropeptide species were clearly resolved by the increase in retention time from unmodified muropeptides through chromatographic separation. Co-elution of mixed muropeptide species was not observed, and the deconvolution of mixed isotopic distributions for analysis was not needed as the observed distributions indicated presence of only a single muropeptide ion. Chemical structures and calculated exact masses for these PG dimers are provided in Supplementary Fig. S1.
Figure 4.
Mass spectra of doubly-charged PG dimers identified from mutanolysin-digested isolated cell wall of vancomycin-resistant E. faecalis grown in absence (a) and presence of vancomycin (b) at stationary growth phase. The chemical structure, formula, and exact mass for PG dimers are provided in Supplementary Fig. S1. Select ion chromatograms (inset) show that D-Ala-D-Lac substituted PG dimer has a longer retention time that resolves it from the unmodified dimer. (c) Changes in the PG composition of dipeptide and depsipeptide terminated PG stems in the cell wall of E. faecalis grown in absence (left) and presence of vancomycin (right) during exponential and stationary growth phases. Muropeptides with PG stems terminating in d-Ala-d-Lac are only found in the cell wall of VRE grown in presence of vancomycin. Error bars represent 95% confidence interval.
LC-MS provided direct evidence for the incorporation of d-Ala-d-Lac substituted PG-stem structures into cell walls of VRE. The incorporation efficiency of d-Ala-d-Lac terminated PG stem was determined by a quantitative PG compositional analysis on 88 of most abundant muropeptide-derived ions selected from 270 total ions identified through LC-MS. Each muropeptide species was quantified by integrating the ion-current chromatogram of selected ion. While this method of determining the abundance is an approximation due to possible differences in the ionization efficiency between muropeptides species, it nevertheless is a good approximation as such PG composition analysis on E. faecium by LC-MS using integration of ion current has yielded results that are in good agreement with solid-state NMR measurements.42 Chemical structure, calculated m/z, observed m/z, and the scaled integral for all identified muropeptide species are provided in Supplementary Table S1-S4.
Bar graphs shown in Figure 4c represent total sum normalized integrals of ion chromatograms from muropeptide species that have a pentapeptide stem structure. Integrals for muropeptide species with a PG-stem structure terminating in d-Ala-d-Ala are shown in blue and d-Ala-d-Lac in red. Muropeptides with d-Ala-d-Lac terminated PG-stem structures are found only from cell walls of VRE grown in presence of vancomycin. Percentages of pentapeptide fragments with a d-Ala-d-Lac terminated PG-stem structure to all pentapeptide fragments at exponential growth phase and stationary growth phase are 25.37 ± 1.94% and 57.33 ± 1.84 % respectively, while percentages of muropeptides with a PG stem terminating in d-Ala-d-Ala at exponential and stationary are 73.63% and 42.67%. Relatively high abundances of depsipeptide-terminated muropeptides confirmed that the depsipeptide-terminated lipid II is efficiently incorporated into the cell wall in VRE.
CONCLUSION
Addition of vancomycin to vancomycin resistant E. faecalis (ATCC 51299) during mid-exponential growth resulted in concentration-dependent perturbed growth (Fig. 1a). Large Park’s nucleotide accumulation in vancomycin-treated VRE (Fig. 1c) indicated that vancomycin inhibited the transglycosylation step of PG biosynthesis by targeting lipid II, suggesting that VRE grown with vancomycin continued to biosynthesize lipid II with a PG stem terminating in d-Ala-d-Ala. The amount of unutilized Park’s nucleotide in the cytoplasm was commensurate with the concentration of vancomycin present in the media for culturing VRE. The estimated amount of accumulated Park’s nucleotide for VRE grown with vancomycin (10 μg/mL) was approximately 1.6 times the total number of PG-repeat units found in the cell wall (Fig. 1c).
VRE grown in presence of vancomycin showed increased VanHB activity, as confirmed by the increased metabolic conversion of [2-13C]pyruvate to d-[2-13C]Lac (Fig. 2b), and VanXB and VanYB activities (Fig 4b) for the biosynthesis of d-Ala-d-Lac substituted lipid II. d-Ala-d-Lac substituted PG stem structure has been thought to interfere with the attachment of l-Ala-l-Ala bridge to lipid II that may result in reduced incorporation of lipid II with d-Ala-d-Lac substituted PG stem into the cell wall, but 13C{15N}REDOR NMR (Fig. 3) demonstrated that the bridge-link density in VRE was not affected by induction of vancomycin resistance. Thus, l-Ala-l-Ala bridge attachment to lipid II was not inhibited by the d-Ala-d-Lac substituted PG stem structure in VRE. LC-MS directly confirmed the presence of d-Ala-d-Lac terminated PG-stems in the cell wall of VRE (Fig. 4). Quantitative PG composition analysis determined that muropeptides with a d-Ala-d-Lac terminated PG stem constituted approximately 26% of all muropeptides with a pentapeptide stem at exponential and 57% at stationary growth phase. Thus, VRE continue to biosynthesize two variants of lipid II even after the induction of vancomycin resistance: one with a PG stem that terminates in d-Ala-d-Ala and the other in d-Ala-d-Lac. Both types of lipid II are efficiently utilized for incorporation into the cell wall of vancomycin resistant E. faecalis following the induction of vancomycin resistance.
Supplementary Material
ACKNOWLEDGMENT
The authors acknowledge the Baylor University Mass Spectrometry Center (BU-MSC) for support during this work, and thank Dr. Alejandro Ramirez for his technical support.
Funding Sources
This work was supported in part by the National Institutes of Health under grant number GM116130.
ABBREVIATIONS
- CPMAS
cross polarization at magic-angle spinning
- LC-MS
liquid chromatography-mass spectrometry
- lipid II
N-acetylglucosamine-N-acetyl-muramyl-pentapeptide-pyrophosphoryl-undecaprenol
- MIC
minimum inhibitory concentration
- NMR
nuclear magnetic resonance
- PG
peptidoglycan
- REDOR
rotational-echo double resonance
- VRE
vancomycin-resistant enterococci
- VRSA
vancomycin-resistant S. aureus
Footnotes
- Chemical structures of PG dimers with a pentapeptide stem terminating in d-Ala-d-Ala and d-Ala-d-Lac
- Observed peptidoglycan fragments by LC-MS for E. faecalis (ATCC 51299) grown in presence and absence of vancomycin during exponential and stationary growth phases
The authors declare no competing financial interest. The Supporting Information is available free of charge via the Internet at http://pubs.acs.org.
REFERENCES
- [1].Arias CA, and Murray BE (2012) The rise of the Enterococcus: beyond vancomycin resistance, Nat. Rev. Microbiol 10, 266–278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [2].Cetinkaya Y, Falk P, and Mayhall CG (2000) Vancomycin-resistant enterococci, Clin. Microbiol. Rev 13, 686–707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].Singh M, Kim SJ, Sharif S, Preobrazhenskaya M, and Schaefer J (2015) REDOR constraints on the peptidoglycan lattice architecture of Staphylococcus aureus and its FemA mutant, Biochim. Biophys. Acta 1848, 363–368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [4].Kim SJ, Singh M, Sharif S, and Schaefer J (2014) Cross-link formation and peptidoglycan lattice assembly in the FemA mutant of Staphylococcus aureus, Biochemistry 53, 1420–1427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].Bouhss A, Josseaume N, Allanic D, Crouvoisier M, Gutmann L, Mainardi JL, Mengin-Lecreulx D, van Heijenoort J, and Arthur M (2001) Identification of the UDP-MurNAc-pentapeptide:L-alanine ligase for synthesis of branched peptidoglycan precursors in Enterococcus faecalis, J. Bacteriol 183, 5122–5127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].van Heijenoort J (2001) Formation of the glycan chains in the synthesis of bacterial peptidoglycan, Glycobiology 11, 25R–36R. [DOI] [PubMed] [Google Scholar]
- [7].Arthur M, Molinas C, Bugg TD, Wright GD, Walsh CT, and Courvalin P (1992) Evidence for in vivo incorporation of D-lactate into peptidoglycan precursors of vancomycin-resistant enterococci, Antimicrob. Agents Chemother 36, 867–869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [8].Billot-Klein D, Gutmann L, Collatz E, and van Heijenoort J (1992) Analysis of peptidoglycan precursors in vancomycin-resistant enterococci, Antimicrob. Agents Chemother 36, 1487–1490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].Walsh CT, Fisher SL, Park IS, Prahalad M, and Wu Z (1996) Bacterial resistance to vancomycin: five genes and one missing hydrogen bond tell the story, Chem. Biol 3, 21–28. [DOI] [PubMed] [Google Scholar]
- [10].Bugg TD, Wright GD, Dutka-Malen S, Arthur M, Courvalin P, and Walsh CT (1991) Molecular basis for vancomycin resistance in Enterococcus faecium BM4147: biosynthesis of a depsipeptide peptidoglycan precursor by vancomycin resistance proteins VanH and VanA, Biochemistry 30, 10408–10415. [DOI] [PubMed] [Google Scholar]
- [11].Derlot E, and Courvalin P (1991) Mechanisms and implications of glycopeptide resistance in enterococci, Am. J. Med 91, 82S–85S. [DOI] [PubMed] [Google Scholar]
- [12].Patel R, Uhl JR, Kohner P, Hopkins MK, and Cockerill FR 3rd. (1997) Multiplex PCR detection of vanA, vanB, vanC-1, and vanC-2/3 genes in enterococci, J. Clin. Microbiol 35, 703–707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [13].Leclercq R, Derlot E, Duval J, and Courvalin P (1988) Plasmid-mediated resistance to vancomycin and teicoplanin in Enterococcus faecium, N. Engl. J. Med 319, 157–161. [DOI] [PubMed] [Google Scholar]
- [14].Evers S, and Courvalin P (1996) Regulation of VanB-type vancomycin resistance gene expression by the VanS(B)-VanR (B) two-component regulatory system in Enterococcus faecalis V583, J. Bacteriol 178, 1302–1309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [15].Arthur M, Molinas C, Dutka-Malen S, and Courvalin P (1991) Structural relationship between the vancomycin resistance protein VanH and 2-hydroxycarboxylic acid dehydrogenases, Gene 103, 133–134. [DOI] [PubMed] [Google Scholar]
- [16].Evers S, Reynolds PE, and Courvalin P (1994) Sequence of the vanB and ddl genes encoding D-alanine:D-lactate and D-alanine: D-alanine ligases in vancomycin-resistant Enterococcus faecalis V583, Gene 140, 97–102. [DOI] [PubMed] [Google Scholar]
- [17].Reynolds PE, Depardieu F, Dutka-Malen S, Arthur M, and Courvalin P (1994) Glycopeptide resistance mediated by enterococcal transposon Tn1546 requires production of VanX for hydrolysis of D-alanyl-D-alanine, Mol. Microbiol 13, 1065–1070. [DOI] [PubMed] [Google Scholar]
- [18].Arthur M, Depardieu F, Snaith HA, Reynolds PE, and Courvalin P (1994) Contribution of VanY d,d-carboxypeptidase to glycopeptide resistance in Enterococcus faecalis by hydrolysis of peptidoglycan precursors, Antimicrob. Agents Chemother 38, 1899–1903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [19].Wright GD, Molinas C, Arthur M, Courvalin P, and Walsh CT (1992) Characterization of vanY, a DD-carboxypeptidase from vancomycin-resistant Enterococcus faecium BM4147, Antimicrob. Agents Chemother 36, 1514–1518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20].Handwerger S, Pucci MJ, Volk KJ, Liu J, and Lee MS (1992) The cytoplasmic peptidoglycan precursor of vancomycin-resistant Enterococcus faecalis terminates in lactate, J. Bacteriol 174, 5982–5984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [21].Gutmann L, Billot-Klein D, al-Obeid S, Klare I, Francoual S, Collatz E, and van Heijenoort J (1992) Inducible carboxypeptidase activity in vancomycin-resistant enterococci, Antimicrob. Agents Chemother 36, 77–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [22].de Jonge BL, Handwerger S, and Gage D (1996) Altered peptidoglycan composition in vancomycin-resistant Enterococcus faecalis, Antimicrob. Agents Chemother 40, 863–869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [23].Billot-Klein D, Shlaes D, Bryant D, Bell D, van Heijenoort J, and Gutmann L (1996) Peptidoglycan structure of Enterococcus faecium expressing vancomycin resistance of the VanB type, Biochem. J 313, 711–715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [24].Severin A, Tabei K, Tenover F, Chung M, Clarke N, and Tomasz A (2004) High level oxacillin and vancomycin resistance and altered cell wall composition in Staphylococcus aureus carrying the staphylococcal mecA and the enterococcal vanA gene complex, J. Biol. Chem 279, 3398–3407. [DOI] [PubMed] [Google Scholar]
- [25].Munch D, Engels I, Muller A, Reder-Christ K, Falkenstein-Paul H, Bierbaum G, Grein F, Bendas G, Sahl HG, and Schneider T (2015) Structural variations of the cell wall precursor lipid II and their influence on binding and activity of the lipoglycopeptide antibiotic oritavancin, Antimicrob Agents Chemother 59, 772–781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [26].Bouhss A, Josseaume N, Severin A, Tabei K, Hugonnet JE, Shlaes D, Mengin-Lecreulx D, Van Heijenoort J, and Arthur M (2002) Synthesis of the L-alanyl-L-alanine cross-bridge of Enterococcus faecalis peptidoglycan, J. Biol. Chem 277, 45935–45941. [DOI] [PubMed] [Google Scholar]
- [27].Kim SJ, Cegelski L, Studelska DR, O' Connor RD, Mehta AK, and Schaefer J (2002) Rotational-echo double resonance characterization of vancomycin binding sites in Staphylococcus aureus, Biochemistry 41, 6967–6977. [DOI] [PubMed] [Google Scholar]
- [28].Kim SJ, Matsuoka S, Patti GJ, and Schaefer J (2008) Vancomycin derivative with damaged d-Ala-d-Ala binding cleft binds to cross-linked peptidoglycan in the cell wall of Staphylococcus aureus, Biochemistry 47, 3822–3831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [29].Gullion T, and Schaefer J (1989) Rotational-echo double-resonance NMR, J. Magn. Reson 81, 196–200. [DOI] [PubMed] [Google Scholar]
- [30].Gullion T, and Schaefer J (1989) Detection of weak heteronuclear dipolar coupling by rotational-echo double-resonance nuclear magnetic resonance, Adv. Magn. Reson 13, 57–83. [Google Scholar]
- [31].Mueller KT, Jarvie TP, Aurentz DJ, and Roberts BW (1995) The REDOR transform: direct calculation of internuclear couplings from dipolar-dephasing NMR data, Chem. Phys. Lett 242, 535–542. [Google Scholar]
- [32].de la Caillerie J.-B. d. E., and Fretigny C (1998) Analysis of the REDOR signal and inversion, J. Magn. Reson 133, 273–280. [DOI] [PubMed] [Google Scholar]
- [33].Gullion T, Baker DB, and Conradi MS (1990) New, compensated Carr-Purcell sequences, J. Magn. Reson 89, 479–484. [Google Scholar]
- [34].Patti GJ, Kim SJ, and Schaefer J (2008) Characterization of the peptidoglycan of vancomycin-susceptible Enterococcus faecium, Biochemistry 47, 8378–8385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [35].Cegelski L, Kim SJ, Hing AW, Studelska DR, O' Connor RD, Mehta AK, and Schaefer J (2002) Rotational-echo double resonance characterization of the effects of vancomycin on cell wall synthesis in Staphylococcus aureus, Biochemistry 41, 13053–13058. [DOI] [PubMed] [Google Scholar]
- [36].McDowell LM, Schmidt A, Cohen ER, Studelska DR, and Schaefer J (1996) Structural constraints on the ternary complex of 5-enolpyruvylshikimate-3-phosphate synthase from rotational-echo double-resonance NMR, J. Mol. Biol 256, 160–171. [DOI] [PubMed] [Google Scholar]
- [37].Kim SJ, Cegelski L, Stueber D, Singh M, Dietrich E, Tanaka KS, Parr TR, Far AR, and Schaefer J (2008) Oritavancin exhibits dual mode of action to inhibit cell-wall biosynthesis in Staphylococcus aureus, J. Mol. Biol 377, 281–293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [38].Deborde C, and Boyaval P (2000) Interactions between pyruvate and lactate metabolism in Propionibacterium freudenreichii subsp. shermanii: in vivo (13)C nuclear magnetic resonance studies, Appl Environ Microbiol 66, 2012–2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [39].Leblanc D (2006) Enterococcus, In The prokaryotes : a handbook on the biology of bacteria (Dworkin M, Falkow S, Rosenberg E, Schleifer K-H, and Stackebrandt E, Eds.) 3rd ed., pp v. < 1–6 >, Springer, New York ; London. [Google Scholar]
- [40].van Heijenoort J (2001) Recent advances in the formation of the bacterial peptidoglycan monomer unit, Nat. Prod. Rep 18, 503–519. [DOI] [PubMed] [Google Scholar]
- [41].Gunetileke KG, and Anwar RA (1968) Biosynthesis of uridine diphospho-N-acetylmuramic acid. II. Purification and properties of pyruvate-uridine diphospho-N-acetylglucosamine transferase and characterization of uridine diphospho-N-acetylenopyruvylglucosamine, J. Biol. Chem 243, 5770–5778. [PubMed] [Google Scholar]
- [42].Patti GJ, Chen J, Schaefer J, and Gross ML (2008) Characterization of structural variations in the peptidoglycan of vancomycin-susceptible Enterococcus faecium: understanding glycopeptide-antibiotic binding sites using mass spectrometry, J. Am. Soc. Mass Spectrom 19, 1467–1475. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.