Abstract
Securing food supply for a growing population is a major challenge and heavily relies on the use of agrochemicals to maximize crop yield. It is increasingly recognized, that some neonicotinoid insecticides have a negative impact on non-target organisms, including important pollinators such as the European honeybee Apis mellifera. Toxicity of neonicotinoids may be enhanced through simultaneous exposure with additional pesticides, which could help explain, in part, the global decline of honeybee colonies. Here we examined whether exposure effects of the neonicotinoid thiamethoxam on bee viability are enhanced by the commonly used fungicide carbendazim and the herbicide glyphosate. We also analysed alternative splicing changes upon pesticide exposure in the honeybee. In particular, we examined transcripts of three genes: (i) the stress sensor gene X box binding protein-1 (Xbp1), (ii) the Down Syndrome Cell Adhesion Molecule (Dscam) gene and iii) the embryonic lethal/abnormal visual system (elav) gene, which are important for neuronal function. Our results showed that acute thiamethoxam exposure is not enhanced by carbendazim, nor glyphosate. Toxicity of the compounds did not trigger stress-induced, alternative splicing in the analysed mRNAs, thereby leaving dormant a cellular response pathway to these man-made environmental perturbations.
Subject terms: RNA splicing, Ecological genetics
Introduction
Worldwide honeybees and other insects encounter new man-made compounds at potentially harmful concentrations in agricultural landscapes. The combinatorial use of many herbicides, fungicides and pesticides is increasingly recognized for having a negative impact on many pollinators including the honeybee Apis mellifera1,2. During their foraging for nectar, pollen and water, forager bees can be exposed to agrochemicals which have been applied to crops3,4. Through the contaminated food harvested by bees and brought into the hive, the entire colony can be exposed to complex cocktails of xenobiotics5. Such exposure to sub-lethal mixtures of pesticides may cause a reduction in vigour and productivity of the hive5,6. Indeed, honeybee colonies are in decline in many parts of the world and numerous interacting factors drive the rates of loss, including pathogens, poor nutrition, environmental stress and crop protection chemicals [reviewed in7].
One class of insecticides used globally are the neonicotinoids. These nicotine-like neurotoxic insecticides have been linked to declining bee health8. At high levels, neonicotinoids lead to paralysis and death of target and non-target insects by binding to nicotinic acetylcholine receptors (nAChRs) which are expressed in the insect nervous system9,10. Thiamethoxam is one of the neonicotinoid compounds known to affect honeybees11–14.
Glyphosate is the most widely applied herbicide worldwide15 and often detected in honey, wax, pollen, and nectar16–18. Generally considered harmless to pollinating insects, glyphosate has been reported to affect larval development and feeding behaviour of honeybees19,20. Likewise, the fungicide carbendazim can persist in the environment due to its hydrolytically stable properties21,22 and is a frequent contaminant of bee hives23.
The honeybee genome encodes a comparatively small repertoire of xenobiotic detoxifying enzymes24. Consequently, honeybees may have only limited physiological and cellular response options when confronted with different mixtures of agrochemicals. A potential cellular strategy to rapidly respond to such environmental stressors would be differential expression and processing of messenger RNAs (mRNAs)25. Alternative splicing in particular enables cells of an organism to alter and expand availability of different transcripts and their encoding protein-isoforms in response to environmental perturbations26,27. Sub-lethal exposure of xenobiotics can, indeed, alter gene expression28 and induce modulation of splicing reactions29,30. Investigation of potential splicing effects mediated through the action of pesticides may help to clarify how toxic agents interfere with honeybee metabolism.
The X box binding protein-1 (Xbp1) mediates the unfolded protein response (UPR) as a reaction to cellular stress through a splicing mechanism31–35. The Xbp1 mRNA contains a retained intron that prevents expression of functional Xbp1 protein. This intron is spliced through a mechanism normally operative in tRNA genes leading to expression of the full length Xbp1 transcription factor, which then triggers the UPR.
Alternative mRNA splicing is particularly abundant in the brain and most elaborate in ion channels and cell adhesion molecule genes25,36,37. The most extraordinary example of an alternatively spliced gene is Down syndrome cell adhesion molecule (Dscam) gene in the fruitfly Drosophila melanogaster. Dscam can encode 38,016 alternatively spliced mRNAs. Dscam plays important roles in neuronal wiring and axon guidance in the nervous system and in phagocytosis of pathogens in the immune system38–42. Moreover, genetic variants of Dscam have been linked to insecticide resistance in Drosophila43. Dscam alternative splicing has been studied in exon-clusters 4, 6 and 9, which harbour an array of mutually exclusive variable exons and exon selection can be mediated by the splicing regulator Srrm234 in Drosophila44–48.
ELAV (Embryonic Lethal Abnormal Visual System)/HU proteins are important neuronal RNA binding proteins, highly conserved and extensively used as neuronal markers49–51. ELAV regulates alternative splicing by binding to AU-rich motifs, which are abundant in introns and untranslated regions52. The Drosophila genome has three members of the ELAV family of proteins, while the honeybee genome encodes only one ELAV protein30,53,54. ELAV proteins have prominent roles in regulating synaptic plasticity54–58.
Here, we analysed the combined effects of thiamethoxam, carbendazim and glyphosate on worker bee viability. Further, we examined the expression and alternative splicing of the genes Xbp1, Dscam and elav and investigated alternative splicing upon exposure of these commonly used agrochemicals. These experiments could reveal possible indicators of the toxicity of these pesticides. The search for biomarkers and information about the effects of pesticides on the neuronal system of bees is of great importance, aiming to contribute to the characterization of exposure to these xenobiotics at the molecular level.
Results
Thiamethoxam toxicity in bees is not enhanced by carbendazim and glyphosate
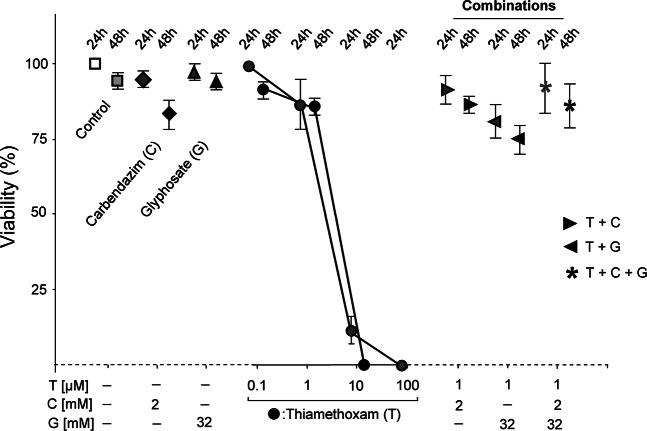
To determine the toxicity of thiamethoxam we injected this neonicotinoid into worker bees. Injection is the most accurate method of delivery and allowed us to administer an identical dose to each worker bee (Supplemental Fig. 2). We chose an injection volume of 2 µl based on our previous experience from injections with Drosophila, where an estimated 1/10 of the hemolymph volume is well tolerated59. LD50 for thiamethoxam was between 1 and 10 µM, and 100 µM resulted in complete lethality (Fig. 1). Intriguingly, the commonly used fungicide carbendazim and herbicide glyphosate, both at highest water soluble concentrations of 2 mM and 32 mM were not lethal (Fig. 1). Furthermore, combining these two compounds with a sub-lethal dose of thiamethoxam did not enhance its toxicity (Fig. 1). Initially, we assessed viability rates after 24 hours of exposure. We then repeated these injection experiments and determined viability also after 48 hours of exposure to these combinations of agrochemicals and did not observe significant differences between the two time points (Fig. 1).
Figure 1.
Viability of Apis mellifera exposed to xenobiotics. Means with standard errors from three experiments are represented. The percent viability of bees after 24 h and 48 his plotted against the concentration of xenobiotics. Bees were injected with 2 µl of water (Control), carbendazim (C, 2 mM), glyphosate (G, 32 mM) and/or thiamethoxam (T) individually or in combinations at indicated concentrations. The 24 h is indicated by lighter colour and the 48 h time-point by a darker colour.
Apis mellifera alternative splicing of the stress sensor Xbp1 does not change upon exposure to xenobiotics
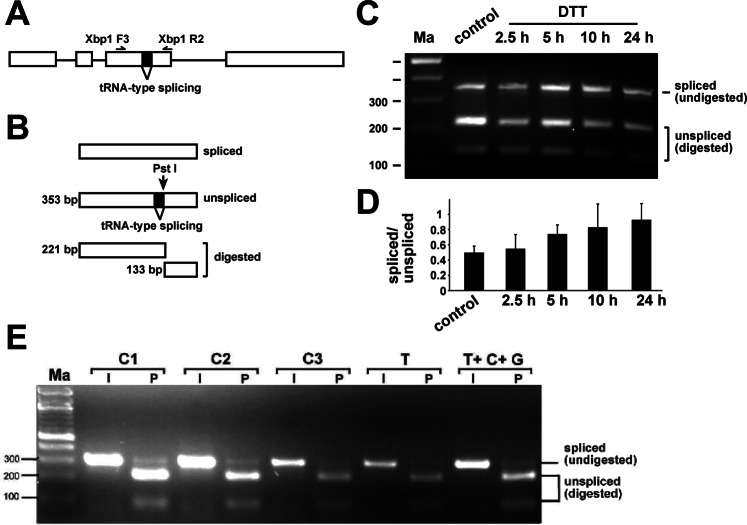
Upon cellular stress, the unfolded protein response (UPR) is triggered by non-spliceosomal tRNA-type alternative splicing of the Xbp1 gene31–35. To confirm that we can induce UPR35, we injected Dithiothreitol (DTT) and followed Xbp1 splicing by RT-PCR over 24 hours at selected time-points (Fig. 2A–D). Twenty four hours after injection of 2 µl of a 20 mM DTT solution, splicing of Xbp1 increased about two-fold (Fig. 2C,D). In contrast, injection of sub-lethal doses of thiamethoxam, or the combination of thiamethoxam with carbendazim and glyphosate did not result in apparent changes in the Xbp1 alternative splicing (Fig. 2E).
Figure 2.
Apis mellifera Xbp1 non-spliceosomal intron splicing in worker bees is unaffected by thiamethoxan, carbendazim and glyphosate. (A) Gene structure of Apis mellifera Xbp1 depicting the tRNA-type spliced intron and primers used to analyse its splicing (top). (B) To resolve similar sized spliced and unspliced isoforms, the RT-PCR product was digested with PstI, which only cuts the unspliced RT-PCR product. The size of the smaller fragment for return primer Xbp1 R1 is 91 bp (not shown). (C) Agarose gel showing the alternative splicing pattern of Xbp1 amplified with primers Xbp1 F3 and R2 by digestion of the RT-PCR product with PstI (P) at different time-points after injection of 2 µl 20 mM DTT. (D) Quantification of the changes in Xbp1 splicing shown in (C) as mean with the standard error from three replicates of the ration of spliced to unspliced. Only the large fragment of unspliced was used for quantification. (E) Agarose gel showing the alternative splicing pattern of Xbp1 amplified with primers Xbp1 F3 and R1 by digestion of the RT-PCR product with PstI (P) compared to undigested input (I) in control bees dissected immediately after collection (Control 1), control bees fed with water and sucrose for 24 h (Control 2) and control bees injected with water (Control 3) compared to bees injected with thiamethoxam (1 µM) and bees injected with a mixture of thiamethoxam (1 µM, T), carbendazim (2 mM, C) and glyphosate (32 mM, G) 24 h prior dissection. Samples were run on a 3% agarose gel. Ma: DNA marker. The undigested PCR product is shown at the bottom.
Apis mellifera Dscam exon 4 alternative splicing does not change during development, in adults and upon exposure to xenobiotics
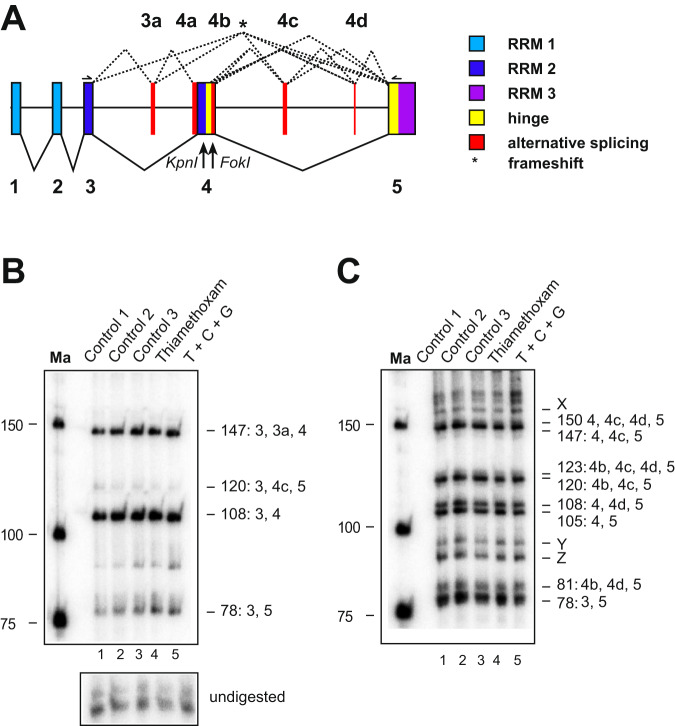
To examine potentially toxic effects of thiamethoxam on alternative splicing regulation in bees, we chose to analyse the splicing pattern in one of the most complex genes in arthopods, the Dscam gene39. Dscam in bees has three variable clusters of mutually exclusive exons which are the exon 4 cluster with 8 annotated variables, the exon 6 cluster with 45 variables and the exon 9 cluster with 17 variables60. We chose the exon 4 cluster because we could separate all variable exons after digestion based on annotated sequences with a combination of restriction enzymes on denaturing polyacrylamide gels, whereby exons 4.1, 4.2, 4.6, 4.7, 4.8 were resolved by Sau3AI, exons 4.3, 4.5 by HaeIII and exon 4.4 by MspI (Fig. 3A,B)47. Since the splicing pattern has not been characterized before, we then determined whether all eight annotated exons 4 were present in bees (Fig. 3A).
Figure 3.
Apis mellifera Dscam exon 4 alternative splicing during bee development and between castes. (A) Schematic depiction of Apis mellifera Dscam exon 4 variable cluster with primers indicated below orange markes corresponding to constant exons 3 and 5. Variable exons 4 are marked in light blue. Newly discovered exon 4.0 is spliced to exon 4.6 (dark blue). An alternative 5′ splice site discovered in exon 4.5 is indicated as a line. (B) Denaturing polyacrylamide gels (6%) showing the splicing pattern of Dscam exon 4 isoform variables on top by digestion of a 32P labeled RT-PCR product with a combination of HaeIII, MspI, and Sau3AI restriction enzymes in embryos (line1), larval brains (line 2), worker brains (line 3), drone brains (line 4) and queen brains (line 5). Exon 4.0, that is close to exon 4.6 in length and exon 4.0 + 4.6 are shown from an undigested control (bottom). (C) Table showing the length of variable exons and their length after restriction digest with indicated restriction enzymes. Exon 4.0 is close to exon 4.6 in length and is shown from na undigested control gel in (B). (D) Quantification of inclusion levels of individual exons are shown as means with standard error from three experiments for embryos, larval brains, worker brains, drone brains and queen brains.
Indeed, we could detect all annotated eight exons, but in addition, we also detected an additional exon, termed exon 4.0 (80 nts, CTGTTTAGAA…TACAGACACG), that is mostly spliced to exon 4.6 and a recessed alternative 5′ splice site in exon 4.5. In addition, a number of bands were evident (X-Z, Fig. 3B), that could not be further identified using separation and excision of bands on agarose gels for sequencing. In contrast to Drosophila, only eight of the 12 exons are present in the bee Apis mellifera (Fig. 3B,C).
Inclusion levels of variable exons were determined for bee embryos, larval brains, and brains from foragers, drones and queens. The inclusion of annotated exon 4 variants (exons 4.1–4.8) revealed no apparent differences in these five developmental stages (Fig. 3D).
After exposure to sub-lethal doses of thiamethoxam, or the combination of thiamethoxam with carbendazim and glyphosate, no apparent changes in the Dscam exon 4 splicing pattern in the brain were detected 5 h, 10 h or 24 h after injection (Fig. 4 and Supplemental Fig. 2).
Figure 4.
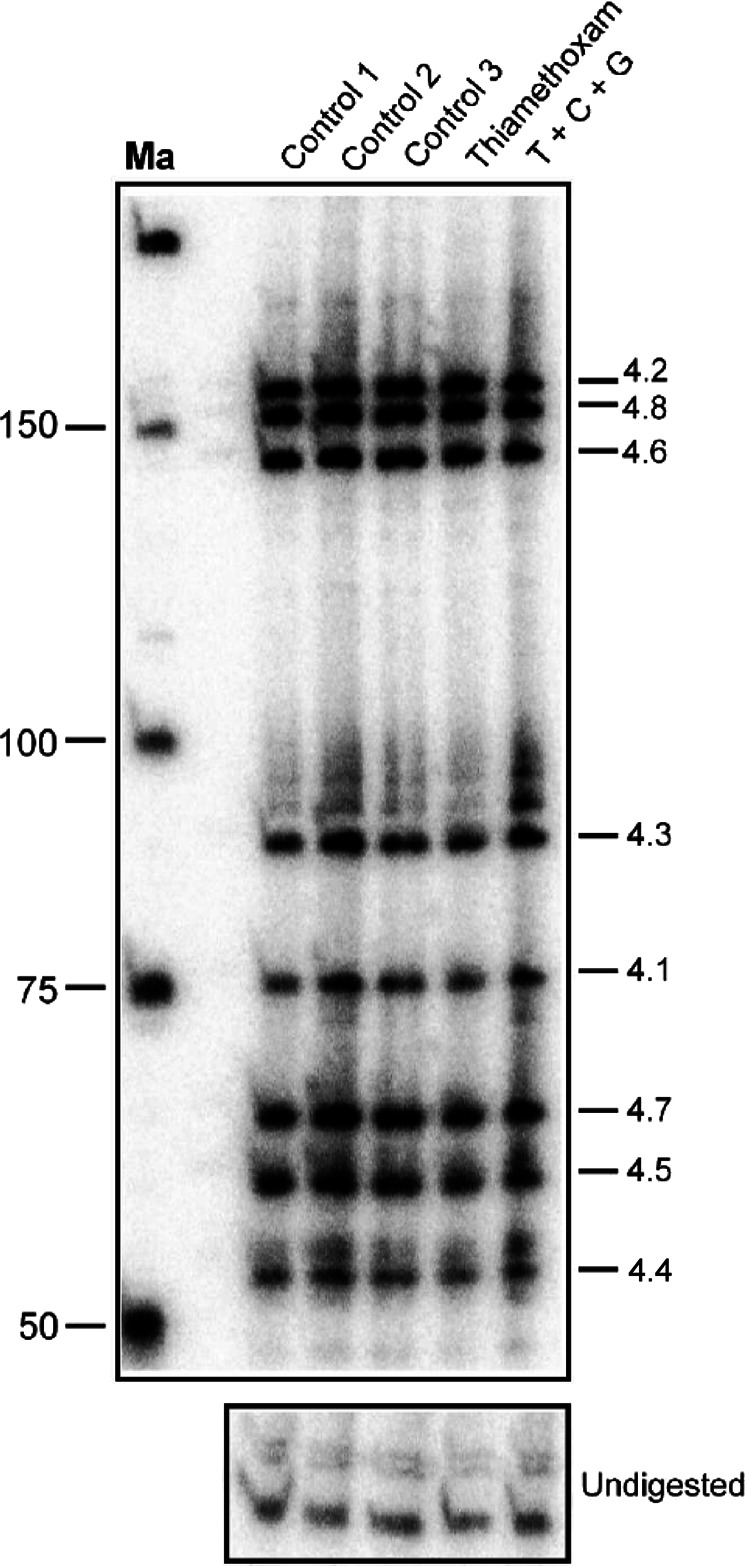
Apis mellifera Dscam exon 4 alternative splicing in brains of worker bees is unaffected by thiamethoxan, carbendazim and glyphosate. Denaturing polyacrylamide gels (6%) showing the splicing pattern of Dscam exon 4 isoform variables on top by digestion of a 32P labeled RT-PCR product with a combination of HaeIII, MspI, and Sau3AI restriction enzymes in control bees dissected immediately after collection (Control 1), control bees fed with water and sucrose for 24 h (Control 2) and control bees injected with water (Control 3) compared to bees injected with thiamethoxam (1 µM) and bees injected with a mixture of thiamethoxam (1 µM, T), carbendazim (2 mM, C) and glyphosate (32 mM, G) 24 h prior dissection. Samples were run on 8% polyacrylamide gel. Ma: DNA marker. The undigested PCR product is shown at the bottom.
Apis mellifera elav alternative splicing does not change upon exposure to xenobiotics
Next, we examined alternative splicing of elav in honeybee workers upon exposure to xenobiotics. To determine alternative splice forms, 5′ and 3′ 32P labelled PCR products covering the variable region were digested with KpnI and FokI restriction enzymes, respectively, and resolved by denaturing polyacrylamide gels (Fig. 5A–C). After exposure for 24 hours to sub-lethal doses of thiamethoxam, or the combination of thiamethoxam with carbendazim and glyphosate, no apparent changes were detected in the elav splicing pattern in the brain (Fig. 5B,C).
Figure 5.
Apis mellifera elav alternative splicing in brains of worker bees is unaffected by thiamethoxan, carbendazim and glyphosate. (A) Gene structure of Apis mellifera elav depicting color-coded functional protein domains with constant exons (1–5, bottom, solid lines) and alternative splicing exons (3a and 4a–d, top, dashed lines). RNA Recognition Motiv 1 (RRM1): light blue, RRM2: dark blue, RRM3: purple, hinge region: red and alternatively spliced parts in red. KpnI and FokI restriction sites used to separate isoforms are indicated below the gene model. An asterisk indicates isoforms that encode truncated proteins by introducing a frameshift. (B,C) Denaturing polyacrylamide gels (6%) showing the alternative splicing pattern of elav by digestion of a 5′ (B) or 3′ (C) 32P labeled RT-PCR product with KpnI (B) and FokI (C) in control bees dissected immediately after collection (Control 1), control bees fed with water and sucrose for 24 h (Control 2) and control bees injected with water (Control 3) compared to bees injected with thiamethoxam (1 µM) and bees injected with a mixture of thiamethoxam (1 µM, T), carbendazim (2 mM, C) and glyphosate (32 mM, G) 24 h prior dissection. Samples were run on 6% polyacrylamide gel. Ma: DNA marker. The undigested PCR product is shown at the bottom.
Discussion
Like other insects, honeybees are exposed to complex combinations of agrochemicals present at potentially harmful concentrations in agricultural landscapes61. Honeybees display marked variation in sensitivity to different insecticides62,63, which may be influenced by different types of pesticide-pesticide cocktails24,61,64.
Here we investigated for the first time whether alternative splicing of important stress and neuronal genes is activated in the nervous system of the honeybee following acute exposure to xenobiotics. Injection into the hemolymph of forager bees ensured accurate and identical application of the tested pesticides. This is an initial approach to explore molecular effects of compounds on individual forager bees before evaluating exposure outcomes at the colony-level in cost- and labor-intensive field-experiments65.
Within a 48 hour window after acute exposure we did not observe any synergistic or additive effects on survival rates when sub-lethal doses of either glyphosate or carbendazim, or both, were administered along with the neonicotinoid thiamethoxam. Phenotypic outcomes of these particular combinations of insecticide, herbicide and fungicide have not been reported previously. As we only examined the viability of individual forager bees following short-term exposure we cannot rule out other, more subtle phenotypic effects nor predict what the synergistic impacts of chronic exposure to these pesticide combinations would be on individual bees or on a hive level. For example, gene expression alterations have been detected following 48 hour and 72 hour thiamethoxam exposures at concentrations which are similar to the sub-lethal dose tested here, however these changes after such prolonged time are likely indirect66.
Alternative splicing is a mechanism by which the exons are spliced in different ways to generate multiple transcripts from one mRNA precursor. This process contributes to protein diversity by generating different types of proteins and has been recognised as a rapid cellular mechanism in response to environmental perturbation27. From our experience with Drosophila, alternative splicing differences are most pronounced 24 hours after xenobiotic exposure30; for this reason we chose this time point to investigate RNA splicing in honeybees.
Alternative splicing is further thought to provide means for adaption to environmental changes, but given the complexity of the splicing process involving hundreds of proteins alternative splicing likely is also susceptible to interference by xenobiotics29,30. Since neonicotinoids show neurotoxic features in bees, we reasoned that alternative splicing of elav might be altered when bees are exposed to thiamethoxam alone, or in combination with glyphosate and carbendazim. Therefore, we analyzed the inclusion levels of elav variable exons through a novel method47. Our results revealed minimal changes in elav splicing in the presence of the abiotic pesticide stressors when compared to the control groups.
Similar to elav, the same pesticide dosages and exposure conditions revealed no significant changes in splicing patterns of Dscam exon 4. This lack of alternative splicing changes was unexpected as an enormous Dscam diversity is generated by mutually exclusive splicing in the Drosophila nervous and immune systems39.
The third gene we investigated for alternative splicing was Xbp1. Xbp1 is involved in the unfolded protein response (UPR), which is activated during stress conditions67. Johnston and colleagues had previously reported a robust UPR activation in the honeybee in response to multiple known stressors, including tunicamycin, a protein glycosylation inhibitor and DTT35. Based on these findings we reasoned that Xbp1 might serve as a key molecular component in mediating individual and combined effects of environmental stressors in honeybees. However, we did not detect changes in the characteristic tRNA-type cytoplasmic splicing event that processes Xbp1 transcripts in response to cellular accumulation of unfolded proteins.
Although our study is limited in that we evaluated potential alternative splicing events in transcripts of only three genes, namely Xbp1, Dscam and elav, these genes are important representatives of regulators of a stress-response, neuronal wiring, the immune response and synaptogenesis important for behavioural performance during foraging. In this study the acute exposure of bees to agrochemicals does not trigger the alternative splicing of the conserved stress-induced Xbp1, Dscam and elav genes in the brain. It remains to be shown by large-scale analysis of alternative splicing from whole genome RNA sequencing whether the lack of activation of this RNA processing mechanism is a general feature in honeybees upon acute exposure. However, long-term chronic low dose exposure to Thiamethoxam did not reveal significant changes in alternative splicing at global levels68. If alternative splicing remains underused as an adaptive response, this would leave the western honeybee more vulnerable to man-made environmental stresses.
Materials and Methods
Toxicity assays
For developmental expression studies, bees (Apis mellifera) of different castes and developmental stages were taken from the experimental apiary of the University campus in Toulouse (France), and cold-anesthetized by placing them on ice before dissection. Forager bees for toxicity assays were collected from colonies of the Winterbourne Garden of the University of Birmingham (UK) and kept in small round cages used for food storage (500 ml) with holes for air circulation. Bees were fed daily from a sucrose (1:1) filled 2 ml Eppendorf tube with small holes inserted into the lid of the container through a hole. Water was provided by a paper tissue saturated daily with water (Evian). For each experimental group 30 bees were collected (replicates with ten individuals in each group). To ensure that laboratory conditions were not stressful for bees, three groups of 10–12 bees were used. Bees from control group 1 were dissected immediately after collection, and their brains extracted. Bees from control group 2 were fed and dissected after 24 hours. Bees from control group 3 were injected with water into abdomen. Thiamethoxam (Sigma-Aldrich, #37942, PESTANAL analytical standard), carbendazim (Sigma-Aldrich, #45368, PESTANAL analytical standard) and glyphosate (Round-up, Bayer) were diluted in water at their maximal soluble concentration and then the minimum lethal dose was determined by injection. A 20 mM water-dissolved Dithiothreitol (DTT) solution was prepared and 2 µl used for injections.
For injections, bees were cold-anaesthetised by placing them on ice. Once immobilised, the bees were transferred onto a custom-made metal pad connected to a tube with circulating ice-water, which was driven by an aquarium pump. This set-up kept the animals anaesthetised during injections and allowed them to recover afterwards at 35oC. Injections were done with a 10 µl Hamilton syringe and each cold-anaesthetised bee was injected with 2 µl of individual xenobiotics are a mixture thereof with indicated concentrations into the left side of the abdomen between abdominal segment 2 and 3 (Supplemental Fig. 1). After injections, bees were kept in a humidified incubator at 32 °C and viability was scored after 24 hours.
RNA extraction, reverse transcription (RT) and polymerase chain reaction (PCR) and analysis of alternative splicing
RNA extraction was done using Tri-reagent (SIGMA) and reverse transcription was done with Superscript II (Invitrogen) as previously described69 using primer AM Dscam 13R2 (GCCGAGAGTCCTGCGCCGATTCCATTCACAG, 1 pmol/ 20 µl reaction) in combination with an oligo dT primer. 1–2 µl of the RT-PCR mix was used in 50 µl PCR reaction using Fermentas Taq (Thermo Scientific) according to the manufacturer’s instructions. Xbp1 was amplified with primers Xbp1 AM F3 (GAAGAAACTGTTCGAAGGTTAAGGGAAC) and Xbp1 AM R2 (GTTCGATATAATCATCTCCTTGGAG) or Xbp1 AM R1 (TCAAGAGGAAGTAGATGGTCAGAA). For the Pst I digestion of the PCR amplicon, 40 units of the restriction enzyme were added to the PCR amplicon-solution, following PCR amplification; the PCR-Pst I mix was digested for 1 hr at 37oC. PCR products were then analysed on ethidium bromide stained 3% agarose gels. To amplify the Dscam exon 4 cluster, PCR was performed using primers AM Dscam 3F1 (AGTTCACAGCCGAGATGTTAGCGTGAGAGC) and AM Dscam 5R1 (GGAAGGCAGTACCAAGTATTTTC) for 37 cycles with 1 µl of cDNA. New variables of Dscam exon 4 were gel purified and determined by sequencing (exon 4.0 and 4.0 + 4.6) or by the annotated sequence (Exon 4.5 recessed alternative 5′ splice site). Apis elav was amplified with primers elav AM F2 (GTCGCGGATACTTTGCGACAACATCAC) and elav AM R2 (CCCGGGTAGCATCGAGTTTGCCAATAGATC). For the analysis of Dscam and elav alternative splicing primers were labeled with 32P gamma-ATP (6000 Ci/ mmol, 25 mM, Perkin Elmer) with PNK to saturation and diluted as appropriate70. From a standard PCR reaction with a 32P labelled forward primer, 10–20% were sequentially digested with a mix of restriction enzymes according to the manufacturer’s instructions (NEB)70. PCR reaction and restriction digests were phenol/CHCl3 extracted, ethanol precipitated in the presence of glycogen (Roche) and analyzed on standard 6% sequencing type denaturing polyacrylamide gels. After exposure to a phosphoimager (BioRad), individual bands were quantified using ImageQuant (BioRad) and inclusion levels for individual variable exons were calculated from the summed up total of all variables. Statistical analysis was done by one-way ANOVA followed by Tukey–Kramer post-hoc analysis using Graphpad prism. Percent inclusion levels were calculated from the total sum of variables as described47.
Supplementary information
Acknowledgements
We thank Noel Parker and the Winterbourne garden for bees, Noel Parker for bee suits, V. Soller-Haussmann for help in bee collections, and L. Hotier for collecting bees at the Toulouse apiary. For this work we acknowledge funding from the Foundation for Research Support of the State of São Paulo, FAPESP (2012/13370-8; 2013/07251-9; 2015/22368-5; 2014/23197-7), the Biotechnology and Biological Sciences Research Council (BBSRC), the Nottingham Birmingham Fund, the Genetics and the Biochemical Society, and the Sukran Sinan Memory Fund.
Author contributions
R.S., T.C.R. and M.S. conceptualized the project. P.D. and P.U. performed the experiments, T.C.R., O.M., J.M.D., R.S. and M.S. supervised experiments and analyzed data, R.S. and M.S. wrote the manuscript with help from P.D., P.U. and J.M.D.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Competing interests
The authors declare no competing interests.
Footnotes
The original online version of this Article was revised: The original version of this Article contained an error in Figure 5A, where the gene structure was incorrectly labelled.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
These authors contributing equally: Pâmela Decio and Pinar Ustaoglu.
Contributor Information
Reinhard Stöger, Email: reinhard.stoger@nottingham.ac.uk.
Matthias Soller, Email: m.soller@bham.ac.uk.
Supplementary information
is available for this paper at 10.1038/s41598-019-55534-8.
References
- 1.Johnson RM, Ellis MD, Mullin CA, Frazier M. Pesticides and honey bee toxicity – USA. Apidologie. 2010;41:312–331. doi: 10.1051/apido/2010018. [DOI] [Google Scholar]
- 2.Woodcock BA, et al. Country-specific effects of neonicotinoid pesticides on honey bees and wild bees. Science. 2017;356:1393–1395. doi: 10.1126/science.aaa1190. [DOI] [PubMed] [Google Scholar]
- 3.Thompson HM. Behavioural effects of pesticides in bees–their potential for use in risk assessment. Ecotoxicology. 2003;12:317–30. doi: 10.1023/A:1022575315413. [DOI] [PubMed] [Google Scholar]
- 4.Desneux N, Decourtye A, Delpuech J-M. The Sublethal Effects of Pesticides on Beneficial Arthropods. Annu. Rev. Entomol. 2007;52:81–106. doi: 10.1146/annurev.ento.52.110405.091440. [DOI] [PubMed] [Google Scholar]
- 5.Devillers J, et al. Structure-toxicity modeling of pesticides to honey bees. SAR QSAR Environ. Res. 2002;13:641–8. doi: 10.1080/1062936021000043391. [DOI] [PubMed] [Google Scholar]
- 6.Blacquière T, Smagghe G, van Gestel CAM, Mommaerts V. Neonicotinoids in bees: a review on concentrations, side-effects and risk assessment. Ecotoxicology. 2012;21:973–992. doi: 10.1007/s10646-012-0863-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Steinhauer N, et al. Drivers of colony losses. Curr. Opin. insect Sci. 2018;26:142–148. doi: 10.1016/j.cois.2018.02.004. [DOI] [PubMed] [Google Scholar]
- 8.Tsvetkov N, et al. Chronic exposure to neonicotinoids reduces honey bee health near corn crops. Science. 2017;356:1395–1397. doi: 10.1126/science.aam7470. [DOI] [PubMed] [Google Scholar]
- 9.Matsuda K, et al. Neonicotinoids: insecticides acting on insect nicotinic acetylcholine receptors. Trends Pharmacol. Sci. 2001;22:573–80. doi: 10.1016/S0165-6147(00)01820-4. [DOI] [PubMed] [Google Scholar]
- 10.Goulson D. REVIEW: An overview of the environmental risks posed by neonicotinoid insecticides. J. Appl. Ecol. 2013;50:977–987. doi: 10.1111/1365-2664.12111. [DOI] [Google Scholar]
- 11.Iwasa T, Motoyama N, Ambrose JT, Roe RM. Mechanism for the differential toxicity of neonicotinoid insecticides in the honey bee, Apis mellifera. Crop Prot. 2004;23:371–378. doi: 10.1016/j.cropro.2003.08.018. [DOI] [Google Scholar]
- 12.Friol PS, Catae AF, Tavares DA, Malaspina O, Roat TC. Can the exposure of Apis mellifera (Hymenoptera, Apiadae) larvae to a field concentration of thiamethoxam affect newly emerged bees? Chemosphere. 2017;185:56–66. doi: 10.1016/j.chemosphere.2017.06.113. [DOI] [PubMed] [Google Scholar]
- 13.Tavares DA, Roat TC, Carvalho SM, Silva-Zacarin ECM, Malaspina O. In vitro effects of thiamethoxam on larvae of Africanized honey bee Apis mellifera (Hymenoptera: Apidae) Chemosphere. 2015;135:370–378. doi: 10.1016/j.chemosphere.2015.04.090. [DOI] [PubMed] [Google Scholar]
- 14.Tavares DA, Roat TC, Silva-Zacarin ECM, Nocelli RCF, Malaspina O. Exposure to thiamethoxam during the larval phase affects synapsin levels in the brain of the honey bee. Ecotoxicol. Environ. Saf. 2019;169:523–528. doi: 10.1016/j.ecoenv.2018.11.048. [DOI] [PubMed] [Google Scholar]
- 15.Benbrook CM. Trends in glyphosate herbicide use in the United States and globally. Environ. Sci. Eur. 2016;28:3. doi: 10.1186/s12302-016-0070-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Mullin CA, et al. High Levels of Miticides and Agrochemicals in North American Apiaries: Implications for Honey Bee Health. PLoS One. 2010;5:e9754. doi: 10.1371/journal.pone.0009754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Boily M, Sarrasin B, DeBlois C, Aras P, Chagnon M. Acetylcholinesterase in honey bees (Apis mellifera) exposed to neonicotinoids, atrazine and glyphosate: laboratory and field experiments. Environ. Sci. Pollut. Res. 2013;20:5603–5614. doi: 10.1007/s11356-013-1568-2. [DOI] [PubMed] [Google Scholar]
- 18.Calatayud-Vernich P, Calatayud F, Simó E, Picó Y. Pesticide residues in honey bees, pollen and beeswax: Assessing beehive exposure. Environ. Pollut. 2018;241:106–114. doi: 10.1016/j.envpol.2018.05.062. [DOI] [PubMed] [Google Scholar]
- 19.Vázquez DE, Ilina N, Pagano EA, Zavala JA, Farina WM. Glyphosate affects the larval development of honey bees depending on the susceptibility of colonies. PLoS One. 2018;13:e0205074. doi: 10.1371/journal.pone.0205074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Liao L-H, Wu W-Y, Berenbaum MR. Behavioral responses of honey bees (Apis mellifera) to natural and synthetic xenobiotics in food. Sci. Rep. 2017;7:15924. doi: 10.1038/s41598-017-15066-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hernandez P, Ballesteros Y, Galan F, Hernandez L. Determination of carbendazim with a graphite electrode modified with silicone OV-17. Electroanalysis. 1996;8:941–946. doi: 10.1002/elan.1140081018. [DOI] [Google Scholar]
- 22.Veneziano A, Vacca G, Arana S, De Simone F, Rastrelli L. Determination of carbendazim, thiabendazole and thiophanate-methyl in banana (Musa acuminata) samples imported to Italy. Food Chem. 2004;87:383–386. doi: 10.1016/j.foodchem.2003.12.009. [DOI] [Google Scholar]
- 23.Lambert O, et al. Widespread Occurrence of Chemical Residues in Beehive Matrices from Apiaries Located in Different Landscapes of Western France. PLoS One. 2013;8:e67007. doi: 10.1371/journal.pone.0067007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Claudianos C, et al. A deficit of detoxification enzymes: pesticide sensitivity and environmental response in the honeybee. Insect Mol. Biol. 2006;15:615–36. doi: 10.1111/j.1365-2583.2006.00672.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Soller M. Pre-messenger RNA processing and its regulation: a genomic perspective. Cell. Mol. Life Sci. 2006;63:796–819. doi: 10.1007/s00018-005-5391-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Nilsen TW, Graveley BR. Expansion of the eukaryotic proteome by alternative splicing. Nature. 2010;463:457–63. doi: 10.1038/nature08909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Pai AA, Luca F. Environmental influences on RNA processing: Biochemical, molecular and genetic regulators of cellular response. Wiley Interdiscip. Rev. RNA. 2019;10:e1503. doi: 10.1002/wrna.1503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Derecka Kamila, Blythe Martin J., Malla Sunir, Genereux Diane P., Guffanti Alessandro, Pavan Paolo, Moles Anna, Snart Charles, Ryder Thomas, Ortori Catharine A., Barrett David A., Schuster Eugene, Stöger Reinhard. Transient Exposure to Low Levels of Insecticide Affects Metabolic Networks of Honeybee Larvae. PLoS ONE. 2013;8(7):e68191. doi: 10.1371/journal.pone.0068191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Sumanasekera C, Watt DS, Stamm S. Substances that can change alternative splice-site selection. Biochem. Soc. Trans. 2008;36:483–90. doi: 10.1042/BST0360483. [DOI] [PubMed] [Google Scholar]
- 30.Zaharieva E, Chipman JK, Soller M. Alternative splicing interference by xenobiotics. Toxicology. 2012;296:1–12. doi: 10.1016/j.tox.2012.01.014. [DOI] [PubMed] [Google Scholar]
- 31.Mitra S, Ryoo HD. The unfolded protein response in metazoan development. J. Cell Sci. 2019;132:jcs217216. doi: 10.1242/jcs.217216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Shen X, et al. Complementary signaling pathways regulate the unfolded protein response and are required for C. elegans development. Cell. 2001;107:893–903. doi: 10.1016/S0092-8674(01)00612-2. [DOI] [PubMed] [Google Scholar]
- 33.Yoshida H, Matsui T, Yamamoto A, Okada T, Mori K. XBP1 mRNA is induced by ATF6 and spliced by IRE1 in response to ER stress to produce a highly active transcription factor. Cell. 2001;107:881–91. doi: 10.1016/S0092-8674(01)00611-0. [DOI] [PubMed] [Google Scholar]
- 34.Calfon M, et al. IRE1 couples endoplasmic reticulum load to secretory capacity by processing the XBP-1 mRNA. Nature. 2002;415:92–96. doi: 10.1038/415092a. [DOI] [PubMed] [Google Scholar]
- 35.Johnston BA, Hooks KB, McKinstry M, Snow JW. Divergent forms of endoplasmic reticulum stress trigger a robust unfolded protein response in honey bees. J. Insect Physiol. 2016;86:1–10. doi: 10.1016/j.jinsphys.2015.12.004. [DOI] [PubMed] [Google Scholar]
- 36.Wang Z, Burge CB. Splicing regulation: from a parts list of regulatory elements to an integrated splicing code. RNA. 2008;14:802–13. doi: 10.1261/rna.876308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Fu X-D, Ares M. Context-dependent control of alternative splicing by RNA-binding proteins. Nat. Rev. Genet. 2014;15:689–701. doi: 10.1038/nrg3778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Watson FL, et al. Extensive diversity of Ig-superfamily proteins in the immune system of insects. Science. 2005;309:1874–8. doi: 10.1126/science.1116887. [DOI] [PubMed] [Google Scholar]
- 39.Schmucker D, et al. Drosophila Dscam is an axon guidance receptor exhibiting extraordinary molecular diversity. Cell. 2000;101:671–84. doi: 10.1016/S0092-8674(00)80878-8. [DOI] [PubMed] [Google Scholar]
- 40.Sun W, et al. Ultra-deep profiling of alternatively spliced Drosophila Dscam isoforms by circularization-assisted multi-segment sequencing. EMBO J. 2013;32:2029–38. doi: 10.1038/emboj.2013.144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Neves G, Zucker J, Daly M, Chess A. Stochastic yet biased expression of multiple Dscam splice variants by individual cells. Nat. Genet. 2004;36:240–246. doi: 10.1038/ng1299. [DOI] [PubMed] [Google Scholar]
- 42.Hemani Y, Soller M. Mechanisms of Drosophila Dscam mutually exclusive splicing regulation. Biochem. Soc. Trans. 2012;40:804–9. doi: 10.1042/BST20120060. [DOI] [PubMed] [Google Scholar]
- 43.Duneau D, et al. Signatures of Insecticide Selection in the Genome of Drosophila melanogaster. G3 Genes, Genomes, Genet. 2018;8:3469–3480. doi: 10.1534/g3.118.200537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Graveley BR. Mutually exclusive splicing of the insect Dscam pre-mRNA directed by competing intronic RNA secondary structures. Cell. 2005;123:65–73. doi: 10.1016/j.cell.2005.07.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Yang Y, et al. RNA secondary structure in mutually exclusive splicing. Nat. Struct. Mol. Biol. 2011;18:159–168. doi: 10.1038/nsmb.1959. [DOI] [PubMed] [Google Scholar]
- 46.Olson S, et al. A regulator of Dscam mutually exclusive splicing fidelity. Nat. Struct. Mol. Biol. 2007;14:1134–40. doi: 10.1038/nsmb1339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Haussmann IU, et al. Plasmid-based gap-repair recombineered transgenes reveal a central role for introns in mutually exclusive alternative splicing in Down Syndrome Cell Adhesion Molecule exon 4. Nucleic Acids Res. 2018;47:1389–1403. doi: 10.1093/nar/gky1254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ustaoglu P, et al. Srrm234, but not canonical SR and hnRNP proteins, drive inclusion of Dscam exon 9 variable exons. RNA. 2019;25:1353–1365. doi: 10.1261/rna.071316.119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Soller M, White K. ELAV. Curr. Biol. 2004;14:R53. doi: 10.1016/j.cub.2003.12.041. [DOI] [PubMed] [Google Scholar]
- 50.Hinman MN, Lou H. Diverse molecular functions of Hu proteins. Cell. Mol. Life Sci. 2008;65:3168–3181. doi: 10.1007/s00018-008-8252-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Pascale A, Amadio M, Quattrone A. Defining a neuron: neuronal ELAV proteins. Cell. Mol. Life Sci. 2008;65:128–140. doi: 10.1007/s00018-007-7017-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Soller M, White K. ELAV multimerizes on conserved AU4-6 motifs important for ewg splicing regulation. Mol. Cell. Biol. 2005;25:7580–91. doi: 10.1128/MCB.25.17.7580-7591.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Haussmann IU, Li M, Soller M. ELAV-Mediated 3′-End Processing of ewg Transcripts Is Evolutionarily Conserved Despite Sequence Degeneration of the ELAV-Binding Site. Genetics. 2011;189:97–107. doi: 10.1534/genetics.111.131383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Zaharieva E, Haussmann IU, Bräuer U, Soller M. Concentration and Localization of Coexpressed ELAV/Hu Proteins Control Specificity of mRNA Processing. Mol. Cell. Biol. 2015;35:3104–15. doi: 10.1128/MCB.00473-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Soller M, Li M, Haussmann IU. Regulation of the ELAV target ewg: insights from an evolutionary perspective. Biochem. Soc. Trans. 2008;36:502–504. doi: 10.1042/BST0360502. [DOI] [PubMed] [Google Scholar]
- 56.Soller M, Li M, Haussmann IU. Determinants of ELAV gene-specific regulation. Biochem. Soc. Trans. 2010;38:1122–1124. doi: 10.1042/BST0381122. [DOI] [PubMed] [Google Scholar]
- 57.Haussmann IU, White K, Soller M. Erect wing regulates synaptic growth in Drosophila by integration of multiple signaling pathways. Genome Biol. 2008;9:R73. doi: 10.1186/gb-2008-9-4-r73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Haussmann IU, Soller M. Differential activity of EWG transcription factor isoforms identifies a subset of differentially regulated genes important for synaptic growth regulation. Dev. Biol. 2010;348:224–230. doi: 10.1016/j.ydbio.2010.09.006. [DOI] [PubMed] [Google Scholar]
- 59.Soller M, Bownes M, Kubli E. Mating and sex peptide stimulate the accumulation of yolk in oocytes of Drosophila melanogaster. Eur. J. Biochem. 1997;243:732–8. doi: 10.1111/j.1432-1033.1997.00732.x. [DOI] [PubMed] [Google Scholar]
- 60.Lee C, Kim N, Roy M, Graveley BR. Massive expansions of Dscam splicing diversity via staggered homologous recombination during arthropod evolution. RNA. 2010;16:91–105. doi: 10.1261/rna.1812710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Johnson RM, Dahlgren L, Siegfried BD, Ellis MD. Acaricide, Fungicide and Drug Interactions in Honey Bees (Apis mellifera) PLoS One. 2013;8:e54092. doi: 10.1371/journal.pone.0054092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Hardstone MC, Scott JG. Is Apis mellifera more sensitive to insecticides than other insects? Pest Manag. Sci. 2010;66:1171–1180. doi: 10.1002/ps.2001. [DOI] [PubMed] [Google Scholar]
- 63.Manjon C, et al. Unravelling the Molecular Determinants of Bee Sensitivity to Neonicotinoid Insecticides. Curr. Biol. 2018;28:1137–1143.e5. doi: 10.1016/j.cub.2018.02.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Berenbaum MR, Johnson RM. Xenobiotic detoxification pathways in honey bees. Curr. Opin. Insect Sci. 2015;10:51–58. doi: 10.1016/j.cois.2015.03.005. [DOI] [PubMed] [Google Scholar]
- 65.Henry Mickaël, Cerrutti Nicolas, Aupinel Pierrick, Decourtye Axel, Gayrard Mélanie, Odoux Jean-François, Pissard Aurélien, Rüger Charlotte, Bretagnolle Vincent. Reconciling laboratory and field assessments of neonicotinoid toxicity to honeybees. Proceedings of the Royal Society B: Biological Sciences. 2015;282(1819):20152110. doi: 10.1098/rspb.2015.2110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Christen V, Mittner F, Fent K. Molecular Effects of Neonicotinoids in Honey Bees (Apis mellifera) Environ. Sci. Technol. 2016;50:4071–4081. doi: 10.1021/acs.est.6b00678. [DOI] [PubMed] [Google Scholar]
- 67.Iwakoshi NN, et al. Plasma cell differentiation and the unfolded protein response intersect at the transcription factor XBP-1. Nat. Immunol. 2003;4:321–9. doi: 10.1038/ni907. [DOI] [PubMed] [Google Scholar]
- 68.Decio, P. et al. Thiamethoxam exposure in bees deregulates short ORF gene expression in the brain and compromises the immune response. BioRxi 10.1101/853291 (2019). [DOI] [PMC free article] [PubMed]
- 69.Koushika SP, Soller M, DeSimone SM, Daub DM, White K. Differential and Inefficient Splicing of a Broadly Expressed Drosophila erect wing Transcript Results in Tissue-Specific Enrichment of the Vital EWG Protein Isoform. Mol. Cell. Biol. 1999;19:3998–4007. doi: 10.1128/MCB.19.6.3998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Iglesias-Gato D, Martín-Marcos P, Santos MA, Hinnebusch AG, Tamame M. Guanine nucleotide pool imbalance impairs multiple steps of protein synthesis and disrupts GCN4 translational control in Saccharomyces cerevisiae. Genetics. 2011;187:105–22. doi: 10.1534/genetics.110.122135. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.