Abstract
Background.
With appropriate protocols, F-18 fluorodeoxyglucose (FDG) positron emission tomography/computed tomography (PET/CT) can visualize myocardial inflammation. Optimal protocols and normative myocardial FDG uptake values are not well-established.
Methods.
We evaluated 111 patients referred for inflammation cardiac FDG PET/CT. Patients followed a low-carbohydrate, high-fat diet for 36 hours before imaging and received unfractionated heparin. Glucose and fatty acid metabolism biomarkers were obtained. Mean blood pool and maximum myocardial uptake (SUVmean, SUVmax) were measured, avoiding areas of abnormal FDG uptake or spillover.
Results.
Adequate suppression of myocardial FDG uptake occurred in 95% of patients (n = 106). Myocardial SUVmax was significantly below background blood pool SUVmean: septal myocardial to blood pool ratio 0.75 (95% CI 0.73–0.77; P < 0.001); lateral myocardial to blood pool ratio 0.70 (95% CI 0.68–0.72; P < 0.001). Glucose, insulin, and C-peptide correlated to blood pool SUVmean (Spearman rs = 0.39, P < 0.01; rs = 0.40, P < 0.01; rs = 0.35, P < 0.01) and myocardial SUVmax (Spearman rs = 0.31, P < 0.01; rs = 0.31, P < 0.01; rs = 0.26, P < 0.01). Fatty acid metabolism biomarkers did not correlate to myocardial SUVmax.
Conclusions.
Patients following intensive metabolic preparation have myocardial FDG SUVmax below background SUVmean. Biomarkers of glucose metabolism modestly correlate to FDG uptake.
Keywords: PET/CT, FDG, myocardial inflammation, metabolic preparation
INTRODUCTION
Positron emission tomography/computed tomography (PET/CT) using F-18 fluorodeoxyglucose (FDG) increasingly is used for imaging of inflammatory cardiac disease.1,2 However, because normal myocardium can metabolize both glucose and free fatty acids (FFAs), physiological accumulation of FDG in the myocardium can interfere with the recognition of abnormal FDG uptake.
Physiologic uptake of glucose (and thus FDG) in the myocardium depends on the patient’s metabolic state and can be modulated by patient preparation and imaging protocol, although there is no consensus on the optimal techniques. Dietary carbohydrate intake causes pancreatic insulin secretion, which promotes translocation of the insulin-dependent glucose transporter GLUT4 to the cell membrane, allowing glucose to enter myocytes in an insulin-dependent fashion. Under low-carbohydrate conditions, cardiac myocytes use free fatty acids for their energy source with minimal glucose uptake. Conversely, glucose enters inflammatory cells via GLUT1 and GLUT3 transporters, which are constitutively expressed and are not dependent on the presence of insulin. Ideally, for imaging cardiac inflammation, normal myocardial uptake of glucose would be completely suppressed, leaving only pathological FDG uptake in the heart.1,3
Methods used for FDG PET/CT inflammatory imaging with respect to suppressing myocardial glucose/FDG uptake vary, with guidelines offering only general protocol recommendations.3,4 Patient fasting, following a low-carbohydrate high fat diet (LCHFD), using unfractionated heparin (UFH) to increase lipolysis and suppress basal glucose uptake, and timing of imaging relative to FDG injection all affect FDG uptake in myocardium.5 UFH administration and LCHFD increase FFA levels in blood, thereby decreasing myocardial blood glucose uptake, and data suggests that a high-blood FFA level is associated with lower myocardial glucose uptake.6–8
Methods to assess adequacy of suppression of myocardial glucose uptake, including the maximal standard uptake value (SUVmax) of FDG and its ratio to background, have met with varying results.9,10 In order for FDG PET/CT to be used reliably for the diagnosis of inflammation, broadly applicable imaging standards and normative values for FDG uptake in myocardium are needed.
We have developed an imaging protocol which consists of a LCHFD, fasting, and UFH administration prior to FDG injection to maximally suppress physiologic myocardial glucose uptake and improve FDG PET/ CT imaging quality. In this work, we sought to characterize our imaging protocol; to determine whether there are correlations among biomarker values of glucose and FFA metabolism and adequate suppression of myocardial FDG uptake; and to develop a reliable method of evaluating for adequate myocardial glucose suppression.
MATERIALS AND METHODS
Study Population
We retrospectively identified 152 consecutive patients with suspected cardiac inflammation referred for cardiac FDG PET/CT at the University of Michigan (Ann Arbor, MI) between June 2015 and August 2016. We excluded patients who: (i) did not complete the LCHFD (2 patients); (ii) did not receive UFH prior to and after FDG injection (10 patients); and (iii) did not have a complete set of biomarkers [insulin, glucose, C-peptide, FFA, triglycerides (TG)] drawn prior to and after heparin administration (29 patients). The remaining 111 patients were included in the final analysis. This study was approved by the University of Michigan Institutional Review Board and was conducted in accordance with institutional guidelines.
Patient Preparation and Biomarker Collection
Several methods to suppress physiological myocardial glucose uptake have been proposed; however, few have been systematically studied or standardized.3 On the basis of a literature search and local expert consensus, we developed our patient preparation and biomarker collection protocol for cardiac inflammation FDG PET/CT scans. UFH has both anticoagulant and lipolytic actions. Asmal et al11 studied the effects of UFH on partial thromboplastin time (PTT) and lipolytic activity as a function of dose in healthy volunteers, and found that a dose of 10 IU/kg did not prolong significantly the PTT but induced a significant decrease in plasma TG and a significant increase in FFA. Additionally, administration of UFH resulted in an increase in plasma FFA and a decrease in plasma TG. In healthy volunteers, Geday12 demonstrated that intravenous administration of 12 IU/kg of UFH resulted in a uniform lipolytic response with maximum lipoprotein lipase activity between 6 and 15 minutes following intravenous UFH administration, with rapid decrease in lipolytic activity thereafter (Supplemental Figure 1). Administration of 5000 IU UFH intravenously to healthy volunteers was shown by Rogers et al to increase myocardial FFA concentrations (coronary sinus sampling, Supplemental Figure 2).13 Williams and Kolodny14 demonstrated that a very high-fat, low-carbohydrate, protein-permitted diet 3–6 hours before FDG administration effectively suppressed myocardial FDG uptake. Based on these data, we developed a patient preparation protocol consisting of a LCHFD, three infusions of 10 IU/kg UFH, a high-fat pre-test meal drink as described below, in addition to glucose and fatty acid biomarker sampling (Figure 1). Based on the prior literature relating biomarkers to fasting, carbohydrate restriction, and heparin infusion, we obtained glucose and fatty acid biomarkers to assist in the clinical determination of adherence to diet and quality of patient metabolic preparation.13‘15–17 Homeostatic Model Assessment of Insulin Resistance (HOMA-IR) was calculated as insulin concentration (μU/mL) × fasting glucose concentration (mg/dL)/405.18
Figure 1.
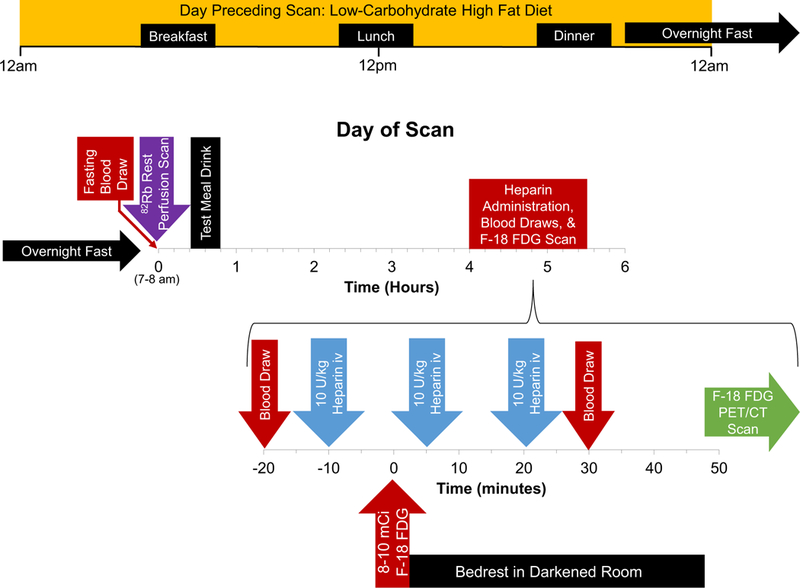
Cardiac FDG PET/CT imaging protocol. A high-fat, low-carbohydrate diet was followed for 36 hours prior to scanning. Rest myocardial persuasion imaging was performed with 82Rb. Following rest myocardial perfusion imaging, the test meal drink was consumed. Approximately 4 house later, 30 IU/kg of unfractionated heparin was administered as three boluses (10 IU/kg) at 10 minutes preceding and 5 and 20 minutes following FDG injection.
All patients were asked to follow a LCHFD prior to PET scanning (appendix). At least 36 hours prior to the scan, patients were instructed to consume a diet containing greater than 35 g of protein and less than 5 g of carbohydrates. Twelve hours prior to the scan (after dinner the preceding evening), patients were asked to refrain from any oral intake other than water and the test meal drink, which consisted of unsweetened almond milk mixed with two tablespoons (30 mL) of vegetable oil, providing 30 g of fat. This drink could be substituted with an Atkins™ Milkshake. This drink was given to patients for breakfast the day of the FDG PET/CT following rest 82Rb perfusion scan. Adherence to the LCHFD diet was verified by a patient diet questionnaire and reviewed by the supervising physician (Appendix). A total of 30 IU/kg of UFH was administered as three boluses (10 IU/kg) at 10 minutes before and 5 and 20 minutes after FDG injection (Figure 1). FFA, TG, and glucose were measured in the fasting state, immediately before UFH administration and on completion of UFH administration. C-peptide and insulin levels were measured before and after UFH administration.
Imaging Protocol
Images were acquired using a whole-body PET/CT scanner (Siemens Biograph mCT, Knoxville, TN). Rest myocardial perfusion images were obtained using 82Rb as previously described.19 PET/CT FDG imaging was performed 59 ± 13 minutes following injection of 9.1 ± 0.8 mCi of FDG. FDG images were acquired for 15 minutes in list mode using 3D ordered subset expectation maximization (3D-OSEM) according to manufacturer’s recommended protocol with 3 iterations and 24 subsets, matrix size of 128 × 128 and pixel size of 3.18 × 3.18 mm2.
Image Analysis
Images were analyzed using Corridor4DM (INVIA Medical Imaging Solutions, Ann Arbor, MI) by two expert readers (SRL or VLM and RLW). Using the fused CT attenuation corrected FDG images, a 1 cm diameter circular, three-dimensional region of interest (ROI) was drawn on fused FDG PET/CT images to measure the mean blood pool standardized uptake value (SUVmean) in the ascending aorta at the level of the pulmonary artery, excluding the vessel wall. The SUVmax for myocardium was measured using a circular three-dimensional ROI drawn in the mid-septal wall and mid-lateral wall, avoiding areas of FDG spillover or abnormal FDG uptake. The ROI diameter ranged from 0.7 to 1.0 cm depending on left ventricular wall thickness, and whenever possible, the ROI was drawn with the largest diameter (Figure 2).
Figure 2.
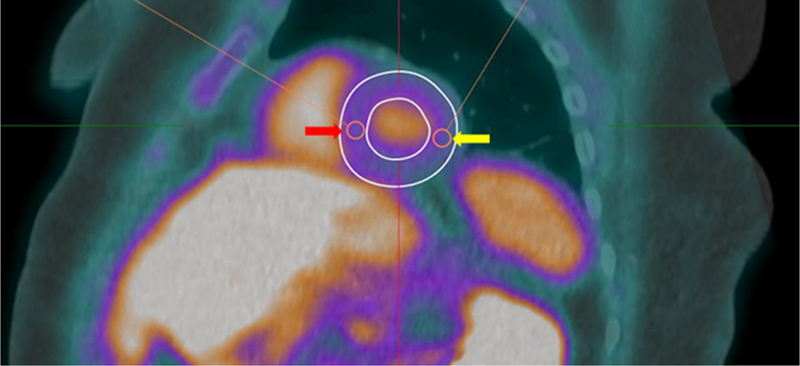
FDG PET/CT image analysis. The SUVmax for myocardium was measured using a three-dimensional ROI drawn in the mid-septal wall (red arrow) and mid-lateral wall (yellow arrow), avoiding areas of FDG spillover or abnormal FDG uptake.
Statistical Analysis
Continuous variables were summarized as mean ± standard deviation (SD). Categorical variables were summarized as numbers and percentages. Variables were assessed for normality by examination of histograms. Biomarker values were log transformed to improve normality. Correlation between myocardial FDG uptake (SUVmax) and background blood pool activity (SUVmean) was assessed using linear regression analysis and described using Pearson’s product-moment correlation coefficient. Correlation between imaging data (blood pool SUVmean or myocardial FDG uptake SUVmax) and markers of FFA and glucose metabolism were described using Spearman’s rank correlation. Ratios of tissue SUVmax to blood pool SUVmean were tested for deviation from a null hypothesis of 1 by evaluating Student’s t test of log-transformed ratios with a null hypothesis of 0. The type 1 error rate was set at 0.05. All statistical analyses were performed with R 3.5.3 (The R Foundation for Statistical Computing, Vienna, Austria).
RESULTS
Patient Characteristics
Table 1 summarizes the characteristics of the patient population. The analyzed data included 111 patients, of which 73 were male (66%), with a mean age of 56.3 ± 12.4 years. There were 31 diabetic patients (28%). Indications for FDG PET/CT imaging were to evaluate for cardiac sarcoidosis (n = 98, 88%), infection (n = 10, 9%) and myocarditis (n = 3, 3%). The average fasting blood glucose and blood glucose prior to FDG injection were 101 ± 40 and 97 ± 36 mg/dL, respectively (Table 2).
Table 1.
Baseline characteristics
| Characteristic | Mean ± SD N (%) |
|---|---|
| Age (years) | 56.3 ± 12.4 |
| Male | 73 (66%) |
| Diabetes mellitus | 31 (28%) |
| Inpatient | 26 (23.4%) |
| Outpatient | 85 (76.6%) |
| Ejection fraction (%) | 48 ± 15 |
| LVEF > 35% | 86 (77.5%) |
| LVEF ≤ 35% | 25 (22.5%) |
| Indications for FDG PET/CT | |
| Sarcoidosis | 98 (88%) |
| Infection | 10 (9%) |
| Myocarditis | 3 (3%) |
| Summed Rest Score | 4.8 ± 6.9 |
Table 2.
Protocol and Metabolic Parameters
| Parameter | Mean ± SD N (%) |
|---|---|
| Protocol parameters | |
| FDG dose (mCi) | 9.1 ± 0.8 |
| Time to imaging following FDG injection (min) | 59 ± 13 |
| Diet adherence | 111 (100%) |
| Heparin administration | 111 (100%) |
| Adequate myocardial suppression | 106 (95.5%) |
| Metabolic parameters | |
| Fasting glucose (mg/dL) | 100.9 ± 39.6 |
| Glucose baseline Pre-FDG injection (mg/dL) | 97.1 ± 36.5 |
| Glucose post-UFH (mg/dL) | 96.2 ± 37.1 |
| C-peptide baseline Pre-FDG injection (ng/mL) | 2.9 ± 2.5 |
| C-peptide post-UFH (ng/mL) | 2.8 ± 2.5 |
| Insulin level baseline Pre-FDG (μU/mL) | 12.8 ± 13.4 |
| Insulin level post-UFH (μU/mL) | 12.5 ± 13.3 |
| FFA fasting (mmol/L) | 0.74 ± 0.33 |
| FFA post-test meal drink/pre-UFH/pre-FDG injection (mmol/L) | 0.87 ± 0.32 |
| FFA post-UFH (mmol/L) | 1.48 ± 0.90 |
| TG fasting (mg/dL) | 154.7 ± 199.6 |
| TG post-test meal drink/pre-UFH/Pre-FDG injection (mg/dL) | 159.5 ± 178.8 |
| TG post-UFH (mg/dL) | 113.8 ± 178.4 |
| Background FDG activity aorta (SUVmean) | 2.1 ± 0.4 |
| Myocardial FDG activity mid-septal wall (SUVmax)a | 1.8 ± 1.0 |
| Myocardial FDG activity mid-lateral wall (SUVmax)a | 1.8 ± 1.3 |
Denotes SUVmax in ROI measuring normal myocardium
Myocardial and Blood Pool FDG Uptake
Of the 111 scans, only five scans were determined to be non-diagnostic after review by two expert readers. Using our patient preparation protocol, this yielded an adequate suppression of physiologic myocardial glucose uptake in 95% of subjects (Table 2, Figure 3). Among patients with adequate metabolic preparation and normal myocardial region of interest (defined as no pathologic FDG uptake in the measured region of interest), myocardial SUVmax in both the septal and lateral walls was strongly related to blood pool SUVmean (Pearson R = 0.749 (septal) and R = 0.695 (lateral); both P < 0.0001, Figure 3).
Figure 3.
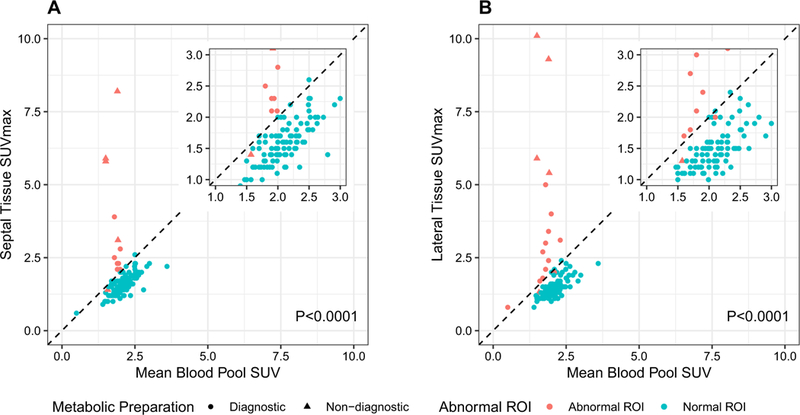
Relationship between septal (A) and lateral (B) myocardial tissue FDG uptake (SUVmax) and aortic blood pool activity (SUVmean). P-values estimated using linear regression among diagnostic scans with normal septal/lateral ROIs. Pearson correlation coefficients provided in text.
SUVmax in normal regions of myocardial FDG uptake in the septal wall (N=100, Figure 3A) and lateral wall (N = 95, Figure 3B) were consistently lower than SUVmean of blood pool, with ratios in the septal wall of 0.75 (95% CI 0.73–0.77, P < 0.001) and lateral wall of 0.70 (95% CI 0.68–0.72, P < 0.001), respectively. This finding extended to diabetics with ratios of in the septal wall of 0.73 (95% CI 0.69–0.78; P < 0.0001) and lateral wall of 0.69 (95% CI 0.65–0.73; P < 0.0001). Similarly, for patients with LVEF ≤ 35% the ratio in the septal wall was 0.80 (95% CI 0.75–0.85, P < 0.0001) and lateral wall of 0.75 (95% CI 0.69–0.81, P < 0.0001). (Supplemental Figures 3, 4).
Of the 106 diagnostic scans, 52 (49.1%) were interpreted as having no myocardial FDG uptake (Supplemental Figure 5) and 54 (50.9%) scans were interpreted as having abnormal myocardial FDG uptake. Among the abnormal diagnostic scans (N = 54), 52 (96.3%) had focal FDG uptake (Supplemental Figure 6), 2 (3.7%) had focal on diffuse FDG uptake (Supplemental Figure 7), and there were no scans with diffuse FDG uptake. There were five scans interpreted as being non-diagnostic, of which 3 (60%) had diffuse myocardial FDG uptake and 2 (40%) had focal on diffuse myocardial FDG uptake (Table 3 and Supplemental Figures 7, 8). Representative cases of myocardial FDG uptake patterns are shown in Figure 4.
Table 3.
Distribution and patterns of myocardial FDG uptake
| Myocardial FDN uptake patterns N (%) |
||||
|---|---|---|---|---|
| No uptake | Focal | Focal on diffuse | Diffuse | |
| All scans (111) | 52 (46.8%) | 52 (46.8%) | 4 (3.6%) | 3 (2.7%) |
| Diagnostic scans (106) | 52 (49.1%) | 52 (49.1%) | 2 (1.8%) | 0 |
| Non-diagnostic scans (5) | 0 | 0 | 2 (40%) | 3 (60%) |
| Diabetes mellitus (31) | 19 (61.3%) | 11 (35.5%) | 1 (3.2%) | 0 |
| LVEF > 35% (86) | 41 (47.7%) | 39 (45.3%) | 3 (3.5%) | 3 (3.5%) |
| LVEF ≤ 35% (25) | 11 (44%) | 13 (52%) | 1 (4%) | 0 |
Figure 4.
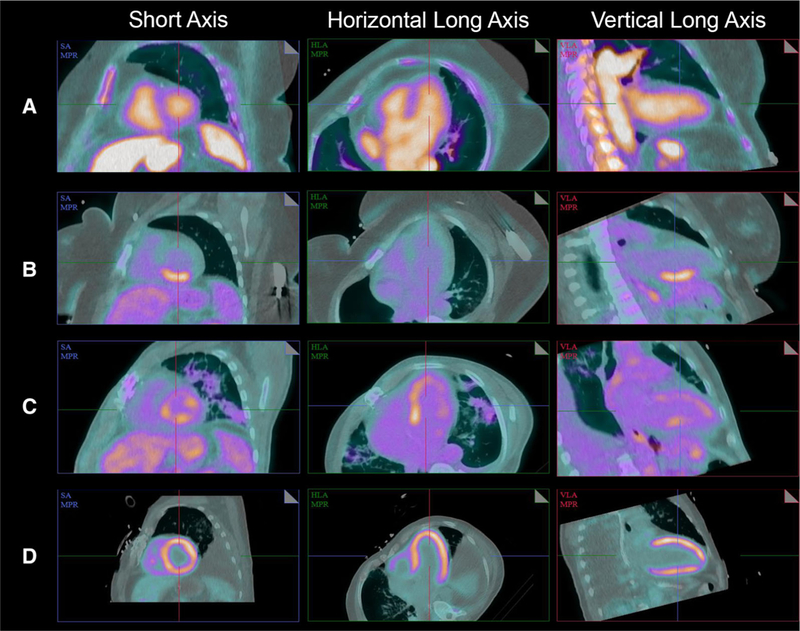
Representative cases illustrating different patterns of myocardial FDG uptake in short axis (SA), horizontal long axis (HLA), and vertical long axis (VLA). A No myocardial FDG uptake, B focal FDG uptake in the inferior wall of the left ventricle, C focal on diffuse left ventricular FDG uptake, and D diffuse left ventricular FDG uptake.
Biomarkers of Glucose and FFA Metabolism and Correlation to FDG Uptake
Following ingestion of the test meal drink FFA increased significantly compared to baseline (0.74 ± 0.33 mmol/L pre-test meal drink and 0.87 ± 0.32 mmol/L post-test meal drink, P = 0.0002) and continued to increase significantly following UFH administration (1.48 ± 0.90 mmol/L post-UFH administration, P < 0.0001, Figure 5A). TG levels increased significantly compared to baseline following the test drink ingestion (155 ± 199 mg/dL baseline and 159 ± 178 mg/dL post-test drink, P = 0.01, Figure 5B). Following UFH administration, TG levels significantly decreased (114 ± 178 mg/dL, P < 0.0001, Figure 5B). Glucose levels decreased significantly from baseline following UFH administration (101 ± 40 mg/dL base-line and 96 ± 37 mg/dL post-UFH, P = 0.0001) (Figure 5C); however, insulin levels did not significantly change pre- and post-UFH administration (12.8 ± 13.4 μU/mL pre-UFH and 12.5 ± 13.3 μU/mL post-UFH, P = 0.13) (Figure 5D).
Figure 5.
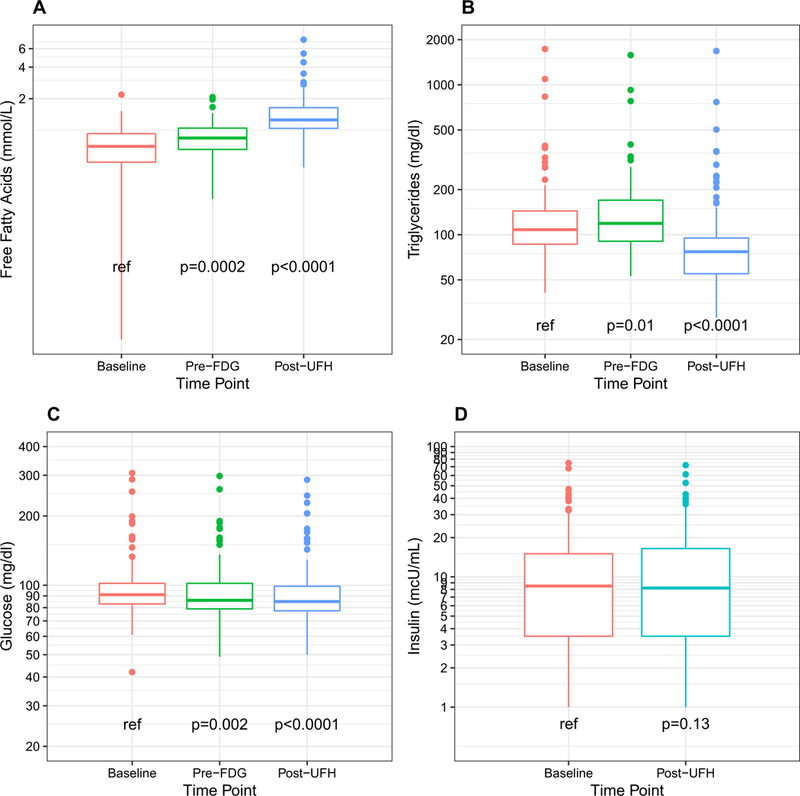
Concentration of free fatty acids (A), triglycerides (B), and glucose (C) at baseline fasting state, prior to FDG injection (following test meal drink) and post-UFH administration. D Concentration of insulin at baseline fasting state and post-UFH administration.
There were 15 patients with insulin levels greater than 1 standard deviation above the mean insulin level (range 26.1–75.0 μU/mL). All of these patients had adequate suppression of myocardial FDG uptake. Among these 15 patients, 8 had no evidence of abnormal myocardial FDG uptake. For patients with abnormal FDG scans, 5 patients had FDG uptake corresponding to myocardial perfusion defects; one patient had FDG uptake corresponding to a region of late gadolinium enhancement on cardiac MRI; and 1 patient had FDG uptake corresponding to a perfusion defect in the septum and also had FDG uptake in the lateral wall without an associated myocardial perfusion defect.
Markers of glucose metabolism (glucose, insulin, C-peptide, and HOMA-IR) were more closely related to blood pool SUVmean than myocardial SUVmax (Figure 6). Conversely, triglyceride and free fatty acid levels had weak, non-significant correlations with blood pool SUVmean and myocardial SUVmax.
Figure 6.
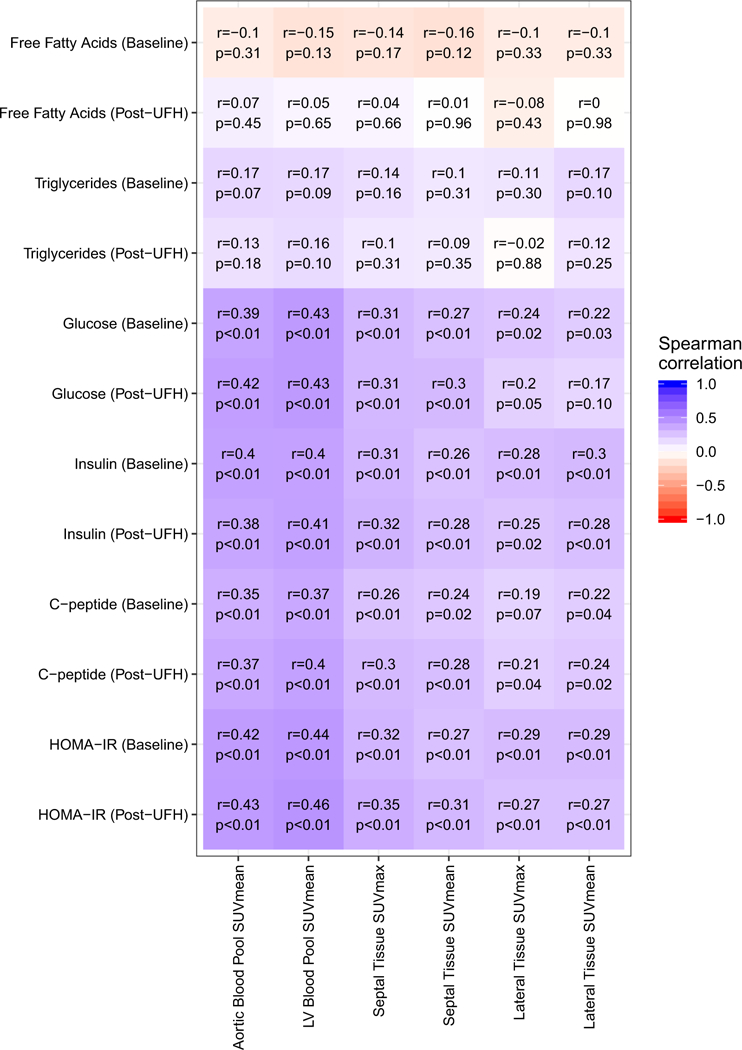
Heat map depicting correlation of biomarkers of glucose and fatty acid metabolism to blood pool and myocardial FDG uptake in diagnostic quality scans and excluding abnormal tissue ROIs.
DISCUSSION
A 36-hour metabolic preparation including a LCHFD, 12-hour fast, and UFH prior to and following FDG injection resulted in suppression of physiologic myocardial glucose uptake in 95% of patients undergoing FDG PET/CT for evaluation of myocardial inflammation. Using this protocol, we found myocardial FDG uptake to be consistently less than background (as measured by aortic blood pool SUVmean) and found myocardial FDG uptake to be strongly correlated to blood pool FDG uptake. The absolute value of myocardial FDG uptake, measured by SUVmax, varied widely in normal myocardium; however, remained consistently lower than blood pool background in patients with adequate myocardial glucose uptake suppression, with a ratio of SUVmax myocardium to SUVmean blood pool of 0.75 (septal) and 0.70 (lateral), respectively. This ratio of SUVmax in normal myocardium to blood pool background SUVmean held true both in patients with areas of abnormal FDG uptake in other regions of the myocardium and in patients without evidence of abnormal FDG uptake. The absolute values of myocardial FDG SUVmax and insulin level were not predictive of adequate myocardial suppression. Based on our findings, comparison of myocardial SUVmax to blood pool SUVmean may be useful for assessment of adequacy of metabolic preparation and developing quantitative thresholds for normal and abnormal FDG myocardial uptake.
In our study, insulin, C-peptide, and glucose levels were related to myocardial and blood pool FDG uptake. Baseline fasting TG were weakly and not significantly related to blood pool FDG uptake. TG and FFA levels were not significantly related to myocardial FDG uptake (SUVmax). Our study was underpowered to determine if any of these biomarkers could be used to distinguish diagnostic quality scans from those without adequate metabolic preparation.
There have been varying reports in the literature regarding the effectiveness of using UFH to suppress myocardial glucose uptake. Elevation of plasma fatty acid concentrations after UFH administration is well described and been associated with impaired glucose utilization and a dose-dependent inhibition of insulinmediated glucose uptake.8,13 Masuda et al6 and Scholtens et al7 reported that UFH administration before FDG PET/CT more effectively suppressed myocardial FDG uptake compared to fasting alone. Manabe et al17 reported that higher baseline FFA levels were associated with reduced physiological myocardial FDG uptake. However, in their study, UFH administration significantly increased FFA concentration in both patients with diffuse LV FDG uptake and in those patients with adequate myocardial suppression of FDG uptake, suggesting the baseline metabolic state of the patient may be the primary driver for myocardial glucose utilization. Morooka et al16 compared myocardial FDG uptake among patients who underwent a 12 hour fast followed by intravenous injection of UFH (50 IU/kg) 15 minutes before FDG injection to patients who fasted for a minimum of 16 hours and did not receive heparin, and reported that FDG uptake was lower in subjects in the longer fasting group without heparin. Finally, Gormsen et al20 demonstrated that while somatostatin-induced insulin suppression combined with heparin administration increased circulating FFAs, myocardial FDG uptake was similar compared to patients who did not receive this metabolic preparation.
These studies highlight the ongoing question as to whether UFH administration aids in suppression of physiologic myocardial glucose uptake. Given the various patient preparation protocols used among these studies, it is difficult to draw conclusions regarding whether UFH administration improved FDG PET/CT image quality.1,21 In our study, using a combination of LCHFD to decrease myocardial glucose utilization and administration of UFH to increase lipolysis, we only found a mild correlation of TG levels to FDG blood pool uptake and no significant correlation of FFA and TG to myocardial FDG uptake. In contrast, markers of glucose metabolism more strongly correlated with blood pool and to a lesser extent, myocardial FDG uptake. How UFH administration, with its concomitant FFA increase, relates to suppression of myocardial glucose uptake requires further investigation, especially since the effects of UFH administration on FFA serum concentrations and myocardial glucose utilization may be altered as a function of the baseline insulin suppression of subjects and the length of patient fasting.
There is conflicting data on whether a high-fat drink prior to FDG injection improves suppression of myocardial FDG uptake. The Randle cycle describes that fatty acid loading suppresses myocardial glucose metabolism and that glucose loading suppresses myocardial fatty acid utilization.8,22 Conversely, fat loading prior to FDG imaging, particularly in patients with high sympathetic/adrenergic activity, may cause increased FDG uptake by increasing myocardial oxygen consumption and promoting glucose metabolism.23 Based on work by Williams and Kolodny14 which reported that a high fat, low-carbohydrate, protein-permitted meal eaten 3 to 6 hours prior to FDG injection suppressed myocardial uptake, we incorporated a high-fat drink approximately 4 hours prior to FDG injection into our protocol.
Studies by Soussan et al,24 Wykrzykowska et al,25 and Harisankar et al26 reported that a high fat, low-carbohydrate diet with a high fat meal prior to FDG injection adequately suppressed myocardial FDG uptake. Kobayashi et al15 demonstrated that a low-carbohydrate diet combined with a high fat beverage one hour before FDG administration improved myocardial FDG suppression compared to fasting alone; however, the level of FFA was not significantly higher than that achieved using conventional fasting. More recently, Lu et al27 reported that a 72-hour, high-fat, high-protein, very low-carbohydrate diet with a high-fat breakfast 4 hours prior to FDG injection effectively suppressed physiological myocardial FDG uptake compared to patients who followed a 24-hour or less dietary preparation. In contrast, Demeure et al28 described that suppression of myocardial FDG uptake was lower in patients who ate a high fat, low-carbohydrate meal followed by a 12 hour fast and received a high-fat drink one hour before FDG injection compared to patients who did not receive a high-fat solution prior to FDG injection. Finally, Cheng et al29 reported that a low-carbohydrate diet resulted in more effective myocardial suppression of FDG uptake compared to a high-fat, low-carbohydrate diet with a high-fat drink consumed one hour prior to FDG administration. Given the variation in patient preparation and dietary protocols, it remains unclear as to whether the addition of a high fat drink or meal one to four hours prior to FDG injection adds additional benefit with respect to suppression of myocardial FDG uptake, and merits further investigation.
Another aim of our study was to define our metabolic preparation protocol and determine how well it suppressed physiological myocardial FDG uptake. Our protocol involved a 36 hour LFHCD, a 12-hour fast, and UFH administration prior to and following FDG injection. In the 111 studies performed only five instances (4.5%) were determined to have inadequate myocardial suppression (based on the presence of diffuse myocardial FDG uptake adjudicated by expert readers). This level of myocardial FDG suppression is comparable to the more successful protocols reviewed by Osborne et al.3 A more recent study by Nensa et al30 evaluated a LCHFD for 24 hours and UFH administration (single dose of 50 IU/kg), but without fasting and found to have a myocardial suppression rate of 84% in a sample of 89 patients. Manabe et al17 also reported that SUVmax and the volume of myocardium with SUVmax greater than 1.5 times that of background blood pool are closely correlated with fasting, and subjects fasting greater than 18 hours had lower SUVmax than those who fasted for shorter periods of time. However, in this study, fasting blood glucose was not significantly associated with myocardial FDG uptake. With a myocardial suppression rate of 95%, our metabolic preparation protocol appears to have highly successful myocardial suppression, suggesting that a LCHFD, fasting, and possibly intravenous UFH administration are important aspects of metabolic preparation for FDG cardiac inflammation studies.
LIMITATIONS
The present study has several limitations. It is a retrospective study from a single center. Details of fasting times and dietary adherence were based on patient-self reporting, albeit with detailed physician review. Thus, we believe the information provided by our patients to be generally reliable, but cannot absolutely be confirmed.
Our protocol to suppress myocardial glucose uptake differs from previously published methods, particularly in the administration of UFH.3 The high degree of myocardial suppression likely is a reflection of the stringency of the protocol. While the SUVmax of well-suppressed myocardium consistently was lower than background LV blood pool SUVmean, there was variation in the SUVmax of normal myocardium (Figure 3). This indicates that there likely is a range of normative myocardial SUVmax which is protocol-dependent, and that direct comparison of quantitative values among different protocols may not be possible. Comparison of our imaging and myocardial suppression data to those generated by other protocols suggests the method described in this paper is effective in imaging myocardial inflammation. Another limitation is that the repeatability of our protocol on serial patient examinations (without intervening treatment) has not been tested. Importantly, recent work by Alvi et al31 investigating the repeatability of FDG PET/CT for cardiac sarcoidosis in scans performed within 2 weeks of each other without intervening treatment (using a HFLC diet and fasting) showed different patterns of FDG uptake in 4 of 15 patients.
Finally, a sub-aim of our study was to correlate serum biomarkers of glucose and fatty acid metabolism to myocardial FDG uptake. Given the very low number of cases with inadequate myocardial FDG suppression (5% of the study population), it is possible that the range of myocardial glucose metabolism was not sufficient enough to identify tighter correlation coefficients.
CONCLUSIONS
Using an intensive metabolic preparation protocol consisting of a LCHFD, fasting, and UFH administration, we achieved an excellent rate of myocardial glucose suppression. In patients who followed this imaging preparation protocol, normal myocardial FDG SUVmax is consistently lower than the SUVmean of the blood pool. Biomarkers of glucose metabolism are modestly related to FDG uptake in the blood pool and myocardium. However, myocardial FDG uptake is strongly correlated to blood pool FDG uptake. Comparison of SUVmax in normal myocardium to blood pool SUVmean may be useful for assessment of adequacy of metabolic preparation and for identification of areas of abnormal myocardial FDG uptake.
NEW KNOWLEDGE GAINED
Following a low-carbohydrate high-fat diet for 36 hours, in combination with a 12-hour fast and unfractionated heparin administration prior to and following FDG administration resulted in excellent suppression of myocardial FDG uptake in 95% of patients presenting for clinical evaluation. Using this metabolic preparation, regions of normal myocardium had FDG uptake consistently lower than background FDG activity in the ascending aorta. Our findings describe important baseline characterization of FDG uptake in normal myocardium in the setting of effective suppression of myocardial glucose metabolism, and will be useful for future FDG PET/CT studies aimed at characterizing myocardial inflammation.
Supplementary Material
Abbreviations
- FDG
F-18 fluorodeoxyglucose
- PET/CT
Positron emission tomography/computed tomography
- SUVmax
Maximal standard uptake value
- SUVmean
Mean standardized uptake value
- ROI
Region of interest
Footnotes
Electronic supplementary material The online version of this article (https://doi.org/10.1007/s12350–019-01786-w) contains supplementary material, which is available to authorized users.
The authors of this article have provided a PowerPoint file, available for download at SpringerLink, which summarises the contents of the paper and is free for re-use at meetings and presentations. Search for the article DOI on SpringerLink.com.
Disclosure
S.R. Larson, J.A. Pieper, E. A. Hulten, and R. L. Weinberg have no disclosures or conflicts of interest related to this publication. E. P. Ficaro and J. R. Corbett have financial interest in INVIA Medical Imaging Solutions, which licenses the commercial software used for imaging processing. INVIA Medical Imaging Solutions did not provide direct support to this study. V. L. Murthy has received consulting fees and stock options from Ionetix, Inc., owns stock in General Electric and Cardinal Health, has a research grant from Siemens Medical Imaging, and has provided expert witness testimony on behalf of Jubilant Draximage. V. L. Murthy is supported by 1R01HL136685 from the National, Heart, Lung, Blood Institute and 1R01AG059729 from the National Institute on Aging. The views expressed are those of the author and do not reflect official policy of Fort Belvoir Community Hospital, the Defense Health Agency, the Department of Defense, or the United States Government.
Publisher’s Note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Chareonthaitawee P, Beanlands RS, Chen W, et al. Joint SNMMI-ASNC expert consensus document on the role of 18F-FDG PET/ CT in cardiac sarcoid detection and therapy monitoring. J Nucl Med 2017;58:1341–53. 10.2967/jnumed.117.196287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lawal I, Sathekge M. F-18 FDG PET/CT imaging of cardiac and vascular inflammation and infection. Br Med Bull 2016;120:55–74. 10.1093/bmb/ldw035. [DOI] [PubMed] [Google Scholar]
- 3.Osborne MT, Hulten EA, Murthy VL, et al. Patient preparation for cardiac fluorine-18 fluorodeoxyglucose positron emission tomography imaging of inflammation. J Nucl Cardiol 2016. 10.1007/s12350-016-0502-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Dilsizian V, Bacharach SL, Beanlands RS, et al. ASNC imaging guidelines/SNMMI procedure standard for positron emission tomography (PET) nuclear cardiology procedures. J Nucl Cardiol 2016. 10.1007/s12350-016-0522-3. [DOI] [PubMed] [Google Scholar]
- 5.Ohira H, Tsujino I, Yoshinaga K. 18F-Fluoro-2-deoxyglucose positron emission tomography in cardiac sarcoidosis. Eur J Nucl Med Mol Imaging 2011;38:1773–83. 10.1007/s00259-011-1832-y. [DOI] [PubMed] [Google Scholar]
- 6.Masuda A, Naya M, Manabe O, et al. Administration of unfractionated heparin with prolonged fasting could reduce physiological 18F-fluorodeoxyglucose uptake in the heart. Acta Radiol 2016;57:661–8. 10.1177/0284185115600916. [DOI] [PubMed] [Google Scholar]
- 7.Scholtens AM, Verberne HJ, Budde RPJ, Lam MGEH. Additional heparin preadministration improves cardiac glucose metabolism suppression over low-carbohydrate diet alone in 18F-FDG PET imaging. J Nucl Med 2016;57:568–73. 10.2967/jnumed.115.166884. [DOI] [PubMed] [Google Scholar]
- 8.Boden G, Chen X, Ruiz J, et al. Mechanisms of fatty acid-induced inhibition of glucose uptake. J Clin Invest 1994;93:2438–46. 10.1172/JCI117252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Memmott MJ, James J, Armstrong IS, et al. The performance of quantitation methods in the evaluation of cardiac implantable electronic device (CIED) infection: A technical review. J Nucl Cardiol 2015. 10.1007/s12350-015-0106-7. [DOI] [PubMed] [Google Scholar]
- 10.Scholtens AM, Swart LE, te Kolste HJ, et al. Standardized uptake values in FDG PET/CT for prosthetic heart valve endocarditis: a call for standardization. J Nucl Cardiol 2017. 10.1007/s12350-017-0932-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Asmal AC, Leary WP, Thandroyen F, et al. A dose-response study of the anticoagulant and lipolytic activities of heparin in normal subjects. Br J Clin Pharmacol 1979;7:531–3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Geday E Lipolysis in plasma after subcutaneous, intramuscular and intravenous heparin in small doses. Acta Med Scand 1966;179:5–12. [PubMed] [Google Scholar]
- 13.Rogers WJ, McDaniel HG, Moraski RE, et al. Effect of heparin- induced free fatty acid elevation on myocardial oxygen consumption in man. Am J Cardiol 1977;40:365–72. [DOI] [PubMed] [Google Scholar]
- 14.Williams G, Kolodny GM. Suppression of myocardial 18F-FDG uptake by preparing patients with a high-fat, low-carbohydrate diet. Am J Roentgenol 2008;190:W151–6. 10.2214/AJR.07.2409. [DOI] [PubMed] [Google Scholar]
- 15.Kobayashi Y, Kumita S, Fukushima Y, et al. Significant suppression of myocardial 18F-fluorodeoxyglucose uptake using 24-h carbohydrate restriction and a low-carbohydrate, high-fat diet. J Cardiol 2013;62:314–9. 10.1016/j.jjcc.2013.05.004. [DOI] [PubMed] [Google Scholar]
- 16.Morooka M, Moroi M, Uno K, et al. Long fasting is effective in inhibiting physiological myocardial 18F-FDG uptake and for evaluating active lesions of cardiac sarcoidosis. EJNMMI Res 2014;4:1 10.1186/2191-219X-4-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Manabe O, Yoshinaga K, Ohira H, et al. The effects of 18-h fasting with low-carbohydrate diet preparation on suppressed physiological myocardial 18F-fluorodeoxyglucose (FDG) uptake and possible minimal effects of unfractionated heparin use in patients with suspected cardiac involvement sarcoidosis. J Nucl Cardiol 2016;23:244–52. 10.1007/s12350-015-0226-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Matthews DR, Hosker JP, Rudenski AS, et al. Homeostasis model assessment: insulin resistance and β-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985;28:412–9. 10.1007/BF00280883. [DOI] [PubMed] [Google Scholar]
- 19.Lee BC, Moody JB, Weinberg RL, et al. Optimization of temporal sampling for 82rubidium PET myocardial blood flow quantification. J Nucl Cardiol 2017;24:1517–29. 10.1007/s12350-017-0899-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gormsen LC, Christensen NL, Bendstrup E, et al. Complete somatostatin-induced insulin suppression combined with heparin loading does not significantly suppress myocardial 18F-FDG uptake in patients with suspected cardiac sarcoidosis. J Nucl Cardiol 2013;20:1108–15. 10.1007/s12350-013-9798-8. [DOI] [PubMed] [Google Scholar]
- 21.Bois JP, Chareonthaitawee P. Continuing evolution in preparation protocols for 18FDG PET assessment of inflammatory or malignant myocardial disease. J Nucl Cardiol 2017;24:989–92. 10.1007/s12350-016-0477-4. [DOI] [PubMed] [Google Scholar]
- 22.Frayn KN. The glucose-fatty acid cycle: a physiological perspective. Biochem Soc Trans 2003;31:1115–9. 10.1042/bst0311115. [DOI] [PubMed] [Google Scholar]
- 23.Vik-Mo H, Mjøs OD. Influence of free fatty acids on myocardial oxygen consumption and ischemic injury. Am J Cardiol 1981;48:361–5. 10.1016/0002-9149(81)90621-4. [DOI] [PubMed] [Google Scholar]
- 24.Soussan M, Brillet P-Y, Nunes H, et al. Clinical value of a high-fat and low-carbohydrate diet before FDG-PET/CT for evaluation of patients with suspected cardiac sarcoidosis. J Nucl Cardiol 2013;20:120–7. 10.1007/s12350-012-9653-3. [DOI] [PubMed] [Google Scholar]
- 25.Wykrzykowska J, Lehman S, Williams G, et al. Imaging of inflamed and vulnerable plaque in coronary arteries with 18F-FDG PET/CT in patients with suppression of myocardial uptake using a low-carbohydrate, high-fat preparation. J Nucl Med 2009;50:563–8. 10.2967/jnumed.108.055616. [DOI] [PubMed] [Google Scholar]
- 26.Harisankar CNB, Mittal BR, Agrawal KL, et al. Utility of high fat and low carbohydrate diet in suppressing myocardial FDG uptake. J Nucl Cardiol 2011;18:926–36. 10.1007/s12350-011-9422-8. [DOI] [PubMed] [Google Scholar]
- 27.Lu Y, Grant C, Xie K, Sweiss NJ. Suppression of myocardial 18F-FDG uptake through prolonged high-fat, high-protein, and very-low-carbohydrate diet before FDG-PET/CT for evaluation of patients with suspected cardiac sarcoidosis. Clin Nucl Med 2016;42:88–94. 10.1097/RLU.0000000000001465. [DOI] [PubMed] [Google Scholar]
- 28.Demeure F, Hanin F-X, Bol A, et al. A randomized trial on the optimization of 18F-FDG myocardial uptake suppression: Implications for vulnerable coronary plaque imaging. J Nucl Med 2014;55:1629–35. 10.2967/jnumed.114.138594. [DOI] [PubMed] [Google Scholar]
- 29.Cheng VY, Slomka PJ, Ahlen M, et al. Impact of carbohydrate restriction with and without fatty acid loading on myocardial 18F-FDG uptake during PET: A randomized controlled trial. J Nucl Cardiol 2010;17:286–91. 10.1007/s12350-009-9179-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Nensa F, Tezgah E, Schweins K, et al. Evaluation of a low-car-bohydrate diet-based preparation protocol without fasting for cardiac PET/MR imaging. J Nucl Cardiol 2017;24:980–8. 10.1007/s12350-016-0443-1. [DOI] [PubMed] [Google Scholar]
- 31.Alvi RM, Young BD, Shahab Z, et al. Repeatability and optimization of FDG positron emission tomography for evaluation of cardiac sarcoidosis. JACC Cardiovasc Imaging 2019. 10.1016/j.jcmg.2019.01.011. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


