Abstract
Frequency of preeclampsia has no tendency to decrease, and it still takes the leading position in the structure of maternal mortality and morbidity worldwide. In this review, we present the “fibrotic concept” of the etiology and pathogenesis of preeclampsia which involves system consisting of Na/K-ATPase and its endogenous ligands including marinobufagenin. New therapy of preeclampsia includes modulation of the Na/K-ATPase system by immunoneutralization of the marinobufagenin and use of mineralocorticoid antagonists which are capable to impair marinobufagenin-Na/K-ATPase interactions.
1. Introduction
Hypertension is the most common medical condition encountered during pregnancy, with a frequency of approximately 6–8% [1]. The hypertensive disorders of pregnancy cover a spectrum of conditions, including preeclampsia-eclampsia, gestational hypertension, and chronic hypertension. Working Group on High Blood Pressure in Pregnancy defines hypertension in pregnancy as two blood pressure measurements of ≥140/90 mmHg measured≥six hours apart [1]. Despite ongoing prevention and repeated attempts to select a therapy, it was not possible to reduce the frequency of preeclampsia (PE). The diagnosis of PE is made by the combination of arterial hypertension (systolic blood pressure ≥ 140 mmHg and(or) diastolic blood pressure ≥ 90 mmHg [2]), proteinuria (presence of protein in the urine ≥ 0.3 g/l in the daily sample (24 h) or in two samples taken at intervals of 6 h; using the test strip (protein in the urine) [2], the indicator (0.3 g/l [3])), and generalized edema. Peripheral edema is observed in 70–80% of cases with physiological pregnancy, therefore, cannot be considered as a diagnostic criterion for PE [4].
The mechanisms implicated in the pathophysiology of PE include oxidative stress, placental steroid formation, and insulin resistance [5, 6]. The endpoint of this complex interplay between maternal and placental mechanisms is a maternal multisystem disorder, characterized by hypertension, proteinuria, and multiorgan dysfunction. During PE, the placental spiral arteries fail to lose their elastic lamellae ultimately leading to decreased placental perfusion [7]. Placental hypoxia is frequently viewed as an early trigger of placental production of soluble factors resulting in endothelial dysfunction [8], which may play the main role in the pathogenesis of PE.
2. Fibrosis and Preeclampsia
The first observation suggesting that fibrosis can underlie the development of PE is related to the works of Tenney and Montgomery in 1935–1936. It has been shown that collagen fibers in the villous stroma are first visible on light microscopy as early as the 2nd month of gestation. The authors found that the collagen content of the villi showed no appreciable increase between midpregnancy and midterm [9]. In 1968, Fox confirmed empirically the results of these works and suggested that placental fibrosis is not associated with any excess of fetal hypoxia complications. It is suggested that villous fibrosis is a result of a reduced fetal villous blood flow; such a reduction may be due, as in toxemia or diabetes, to obliterating endarteritis changes in the fetal stem arteries or, as in prolonged pregnancies, to a constriction of villous capillaries [9].
In later works, it was shown that women who have experienced PE are at increased risk of hypertension, coronary artery disease, and fatal stroke. Its effects do not end with childbirth and is associated with a higher cardiovascular risk lifelong. This increased risk could be partly explained by left ventricular systolic and diastolic impairment, arterial stiffening, and endothelial dysfunction which have been described during and after PE. These phenomena, which influence each other, are related to myocardial and vascular remodeling, a process leading to loss of function and fibrotic tissue deposition [10–12]. In early studies, it was suggested that the modified placental production of vasoactive substances appears to underlie the pathological mechanism of hypertension and vascular dysfunction in PE [13]. Over the past 20 years, it has been shown that endogenous digitalis-like factors or cardiotonic steroids (CS) of the Na/K-ATPase (NKA) are also among these substances, and elevated plasma levels of CS were detected in patients with PE [14–16].
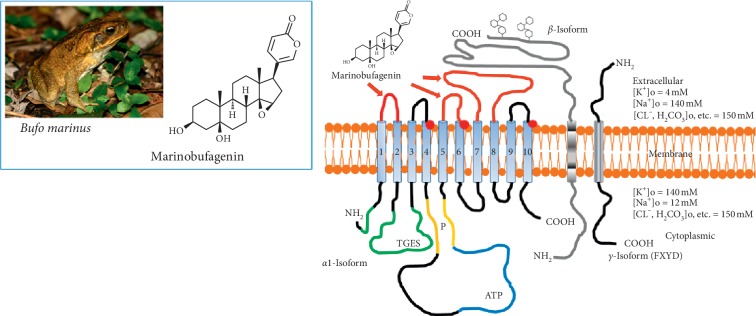
Recently, it has been demonstrated that nanomolar concentrations of bufadienolide CS, marinobufagenin (MBG) (Figure 1) stimulate the synthesis of collagen and induce fibrosis in cardiovascular tissues and in the kidney [17]. One of the mechanisms underlying the profibrotic effect of MBG is EGFR-Src-dependent inhibition of Fli-1, a nuclear transcription factor and a negative collagen-1 regulator, causing its reduction which resulted in activation of collagen-1 [15]. During the experiments, it was shown that CS (MBG, ouabain, and digoxin) cause an increase in the number of embedded proline residues and also increase collagen synthesis in the fibroblast culture, which was evidence of the presence of signaling function of the Na/K-ATPase-CS complex with activation of Src and MAPK [17, 18]. In vivo, the administration of MBG in a concentration that is observed in renal failure caused the development of cardiac fibrosis with activation of the signaling pathway mediated by NKA, which was confirmed by an increase in Src and phosphorylation of a mitogen-activated protein kinase (MAPK) in the myocardium [17, 18].
Figure 1.
Chemical structures of bufadienolide, marinobufagenin and its pharmacological target, Na/K-ATPase.
Our observations confirmed and extended previously obtained findings demonstrating substantial elevations of MBG levels in PE. In our previous study, in patients with severe PE (161/104 mm Hg), plasma MBG concentration comprised averaged 2.63 nmol/L. Later, it was shown that in subjects with mild PE and lower arterial pressure, the mean level of MBG in plasma was 1.68 nmol/L [19]. Since plasma concentrations of MBG in subjects with noncomplicated pregnancy in these previous studies were similar and comprised 0.80 nmol/L and 0.63 nmol/L, respectively, the levels of MBG may be one of the markers of severity of PE. Our findings also indicate that MBG may not only be a marker but also a factor contributing to the pathogenesis of PE. In the “pathophysiologically relevant” range of concentrations (1–3 nmol/L), in vitro, MBG exhibits a 25% inhibition of NKA in the human vascular sarcolemma and induces a contractile response in the isolated rings of human mesenteric arteries [19]. Thus, the in vivo levels of MBG observed in patients with PE in our previous and present studies are capable of enhancing vascular tone and substantially inhibiting NKA.
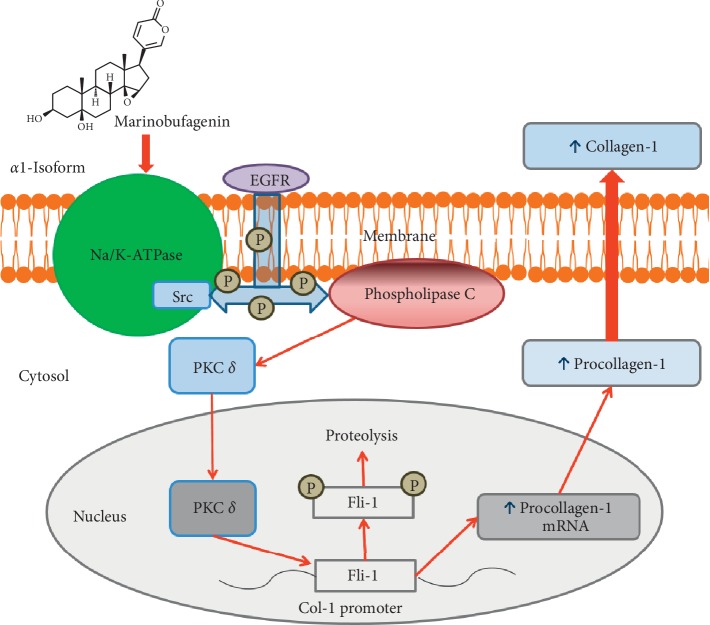
The mechanism of PE includes remodeling of uterine spiral arteries and making them fibrotic by a variety of vascular factors including cardiotonic steroids [20, 21]. Physiological function of CS is regulation of sodium excretion by means of inhibition of NKA in the epithelial cells of renal tubuli, i.e., an adaptive response [20]. Under pathological conditions, for example, in patients with PE, side effect of this primarily adaptive response becomes essential, and CS cause inhibition of sodium pumps in tissues including vessels [22]. Recent studies demonstrated that in addition to vasoconstrictor effect of CS, these compounds directly affect intracellular signaling resulting in the loss of vascular elasticity and vascular fibrosis [23]. Low concentrations of CS acting on PKC delta induce deactivation of Fli-1 and cause expression of collagen-1, which is a key factor of fibrosis in blood vessels, myocardium, and kidney [23, 24]. Recent studies demonstrate the clinical importance of fibrosis in the cardiovascular system and placenta [25, 26], and moreover, suggest that blood vessels exposed to PE environment exhibit greater responsiveness to injury despite resolution of PE [27]. The general scheme of the development of MBG-induced fibrosis is presented in Figure 2.
Figure 2.
Scheme of the Fli-1-dependent profibrotic pathway activated via binding of marinobufagenin (MBG) to Na/K-ATPase (NKA). The interaction of MBG with NKA leads to activation/phosphorylation of Src followed by activation/phosphorylation PLC and PKC via scaffolding function of NKA.
3. Treatment of Preeclampsia
For millennia, the venomous secretions of amphibians, which contain a large number of bufadienolide derivatives, are widely used in the traditional medicine of different civilizations. One of the most well-known examples from the traditional Chinese medicine is Sen-So (or Ch`an Su), which was used for the treatment of heart failure [28]. Being highly active substances, bufadienolides often caused toxic effects. Human fatal poisonings from toad venom have been reported in the many countries by the venoms from the Colorado River toad (Bufo alvarius), the cane toad (Bufo marinus), and the Chinese toad (Bufo gargarizans) [29]. Over the past 20 years, the digoxin-specific Fab fragments (Digibind) have been successfully used in the poisoning of toad venom, and it has shown their effectiveness in animals [30] and humans [31]. It is well known also that in patients at the final stage of renal failure [32] or PE [33], as well as in Dahl-S (salt sensitive) rats with salt load [34, 35], the erythrocyte NKA is inhibited, which can be reversed incubation, ex vivo with antibodies to MBG or with Digibind, and bovine digoxin antibodies that interact with CS [33, 36].
4. Monoclonal Antibody
Immunoneutralization of the CS in patients with PE is not new. The similarity of the functional profile (inhibition of NKA) and the structure of cardenolides (digoxin and ouabain) and bufadienolides (MBG and resibufogenin) allowed clinical use of polyclonal antibodies against digoxin (Digibind) in the treatment of PE [37, 38]. Digibind and DigiFab have been used for many years to treat digoxin intoxication [39]. Digibind has been shown to induce a decrease in blood pressure in animals with volume-expanded hypertension by an interaction between Digibind and an endogenous digoxin-like factor [39]. Digibind demonstrates cross-reactivity against the bufadienolides and cardenolides [19]. In a rat model of PE, the Digibind treatment produced lowering of the blood pressure and proteinuria [40]. In a clinical study, it was shown that antibody therapy by Digibind prevents a decline in renal function with well tolerated in severe PE by neutralizing CS [37].
Based on the results of previous studies, the involvement of MBG was demonstrated in the development of fibrosis. This mechanism was also shown on the PE model [41]. In this study, the umbilical arteries, obtained from pregnant women suffering from PE, contained greater amounts of collagen, less Fli-1, and they also showed low sensitivity to the vasorelaxant effect of sodium nitroprusside. Our laboratory demonstrated that in PE plasma levels of MBG increase and that ex vivo anti-MBG antibody reverses PE-induced inhibition of erythrocyte NKA [19, 33]. In pregnant NaCl-supplemented rats, increased MBG levels accompanied the development of symptoms of PE including elevation of arterial pressure, proteinuria, and reduction of fetal weight and size [33, 42]. In this model, in vivo immunoneutralization of MBG with poly- and monoclonal antibodies produced antihypertensive effect associated with the increase in vascular NKA activity [33, 42]. Considering that, (i) MBG stimulates synthesis of collagen, (ii) fibrosis in placenta and umbilical arteries in PE patients is accompanied by elevated placental MBG and a dramatic decrease in Fli-1 level [43], and (iii) PE is associated with vascular stiffening [44, 45]; it has been showed that MBG is one of the factors, implicated in pathogenesis of PE via induction of vasoconstriction and vascular fibrosis [41]. In the recent study, it has been shown that the placental levels of Fli-1 were dramatically lower, while collagen-1 levels were higher in both placenta and umbilical arteries in PreE samples compared to the tissues from control noncomplicated pregnancy. The subsequent treatment of the umbilical artery explants from PE patients with anti-MBG mAb was accompanied by a significant decrease in collagen-1 abundance [43].
Furthermore, in comparative study of the efficacy of specific antibodies with antidigoxin (DigiFab) and monoclonal antibody against MBG (3E9), it was shown that monoclonal antibodies restore the activity of PE-inhibited NKA in erythrocytes better than DigiFab, which suggested the involvement of MBG in the pathogenesis of PE [46]. In a recent study, it was shown that the administration of humanized monoclonal antibodies (206–208, H1L2) against MBG attenuate MBG-induced downregulated PCNA and upregulated p38 phosphorylation in cytotrophoblast cells, indicating a possible therapeutic action in PE [47]. Therefore, immunoneutralization of MBG is a logical step in the treatment of PE.
5. Mineralocorticoid Antagonists
The fact that mineralocorticoid antagonists can offset the effects of digitoxin in rats has been first reported by Selye [48, 49]. Subsequently, spironolactone and its active metabolite, canrenone, were reported to reverse digitalis toxicity and to lower blood pressure in rat hypertension models, in which levels of CTS are elevated [50–52]. Most recently, spironolactone was reported to suppress cardiac fibrosis in rats chronically treated by MBG [53]. Notably, in this study, MBG exhibited profibrotic effect in the absence of changes in aldosterone levels [53]. Importantly, high levels of MBG were associated with hypertension [33], stiffening of umbilical vessels, and elevated vascular level collagen-1 in PE patients, and in vitro incubation of the healthy blood vessels in the presence of low MBG concentration produced similar phenotype [41]. We hypothesized that aldosterone antagonists can also reverse MBG-induced vascular fibrosis, in the explants of the thoracic aorta and in the cultured rat vascular smooth muscle cells, and we observed that canrenone suppressed the effect of MBG synthesis of collagen-1 [23]. This observation was confirmed by clinical data which showed that in patients with resistant hypertension receiving spironolactone as an addition to the conventional antihypertensive therapy, there was a decrease of aortic vascular stiffness in parallel with an increase in the erythrocyte NKA [23].
Mineralocorticoid antagonists are known to reverse myocardial fibrosis [53]. It has been established that mineralocorticoid antagonists decrease blood pressure in rat hypertension models and suppress cardiac fibrosis, in which levels of CS including MBG are elevated [48, 49]. Thus, an interaction between Na/K-ATPase and MBG could be a target for aldosterone antagonists [53]. Considering the fact that MBG induces fibrosis in the cardiovascular system through the Fli-1-dependent mechanism, it was investigated the effects of spironolactone and its main metabolite, canrenone, for fibrosis in a series of experiments in vitro and in vivo. It has been shown that both spironolactone and canrenone weaken MBG-induced increase in collagen synthesis by myocardial fibroblasts, which was confirmed in vivo by a distinct decrease in cardiac fibrosis caused by experimental renal failure as a result of treatment with spironolactone [53]. As shown earlier, canrenone and spironolactone act as competing inhibitors of CS and NKA [50, 53–55]. Considering these, it can be assumed that short-term treatment with low doses of spironolactone or canrenone can be a very effective method of treating fibrotic manifestations of PE.
Spironolactone was introduced in 1960, and before 1980, this molecule was used for hypertension and PE [56, 57]. It was noted that when pregnant rats were treated with 40 mg spironolactone from 13 to 21 days of pregnancy, male foetuses showed signs of feminization [58]. Therefore, spironolactone is not advised for humans during pregnancy [59]. Considering that eplerenone has not been associated with adverse effects during pregnancy in animal studies, this drug is likely to be a better choice for use in pregnant women than spironolactone. However, no data on the use of these drugs in human pregnancy exist, and therefore, eplerenone should be used in pregnant women with heart failure only when treatment with other diuretics (such as furosemide) is noneffective [60].
Canrenone is the pharmacologically active metabolite of the spironolactone, used in hypertensive therapy. Canrenone exhibits its antialdosterone action because it blocks the binding of aldosterone to a cytosolic receptor in distal and collecting tubules of the nephron with subsequent inhibition of the synthesis of a specific protein that facilitates the entrance of Na+ ions into the cell and the consequent increment of NKA [61]. Several studies suggest that canrenone interacts with the ouabain-sensitive NKA competitively, by antagonizing the binding of 3H-ouabain [55] and MBG [53]. In addition to this well-known action, several studies have shown that canrenone was capable of inhibiting the NKA in vitro [50, 55, 62, 63]. Furthermore, if the pump was blocked by ouabain, canrenone could restimulate the pump [63, 64]. Interestingly, in experiment in rats, mineralocorticoid receptor antagonists also could reverse MBG-induced elevation of blood pressure associated with voluntary ethanol intake [65].
It has been shown that canrenone is a partial competitor of ouabain in the human placental membranes. Moreover, the analysis of the Scatchard plot shows a unique intercept suggesting that canrenone and ouabain compete for the same site [52]. The main findings of this study concern the effect of canrenone on the binding of ouabain to placental NKA and the observation that canrenone, at therapeutic concentrations (about 8 to 10 μM), is able to reduce the ability of ouabain to inhibit the sodium pump [52]. Considering the above data, it can be suggested that therapeutic effects of canrenone can be a successful pathway in the treatment of PE due to its well-established antialdosterone action and to its modulatory effects on the NKA activity.
6. Conclusion
Currently, it is known that MBG is actively involved in the pathogenesis of the development of PE. Being an endogenous ligand of the NKA, MBG via the ionic and signaling pathways participates in the development of placental fibrosis, which may be the main pathological mechanisms of PE. We suggest that the system of MBG and the NKA is a keystone of PE pathogenesis. Immunoneutralization of MBG, as well as antagonism against binding with the NKA, can be a new and effective direction in the treatment of PreE and further prevention of its complications. In conclusion, our results indicate that in preeclampsia, elevated levels of MBG, via a Fli-1-dependent mechanism, stimulate synthesis of collagen in umbilical arteries, which leads to an impairment of vasorelaxation, and MBG-induced Fli-1-dependent signaling pathway contributes to vascular stiffness in preeclampsia. Recent studies have provided evidence that an excess of signaling molecules released by the placenta to circulation, soluble FMS-like tyrosine kinase-1 (sFlt1) and endoglin (sEng), or decreased levels of placental growth factor (PlGF) and vascular endothelial growth factor A (VEGF-A) play an important role in the pathogenesis of PE. Numerous candidate biomarkers have been proposed for prediction of PE and measurement of maternal circulating factors, such as ratio of sFlt-1/PlGF and reflected antiangiogenic balance that characterizes PE [66]. In our earlier work, in patients with PE levels of sFlt1 and of sEng were elevated together with CS [42], but whether or not both are interconnected remains to be understood. Interestingly, most of the studies of CS and of antiangiogenic balance did not make a distinction between early- and late-onset PE, which also represents an important area for future investigations.
Acknowledgments
This work was supported by the Russian Scientific Foundation grant no. 18-15-00222 (AYB, NIA, VAR).
Conflicts of Interest
All authors declare no conflicts of interest.
References
- 1.National High Blood Pressure Education Program Working Group on High Blood Pressure in Pregnancy. Report of the national high blood pressure education program working group on high blood pressure in pregnancy. American Journal of Obstetrics & Gynecology. 2000;183(1):s1–s22. doi: 10.1016/s0002-9378(00)40820-3. [DOI] [PubMed] [Google Scholar]
- 2.ACOG. Practice bulletin no. 125: chronic hypertension in pregnancy. Obstetrics and Gynecology. 2012;119(2):396–407. doi: 10.1097/aog.0b013e318249ff06. [DOI] [PubMed] [Google Scholar]
- 3.Thangaratinam S., Coomarasamy A., O’Mahony F., et al. Estimation of proteinuria as a predictor of complications of pre-eclampsia: a systematic review. BMC Medicine. 2009;7(1):p. 10. doi: 10.1186/1741-7015-7-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cunningham F. G. Williams Obstetrics. 25th. New York, USA: McGraw-Hill; 2018. [Google Scholar]
- 5.Wagner S. J., Craici I. M., Grande J. P., Garovic V. D. From placenta to podocyte: vascular and podocyte pathophysiology in preeclampsia. Clinical Nephrology. 2012;78(3):241–249. doi: 10.5414/cn107321. [DOI] [PubMed] [Google Scholar]
- 6.Herse F., LaMarca B. Angiotensin II type 1 receptor autoantibody (AT1-AA)-mediated pregnancy hypertension. American Journal of Reproductive Immunology. 2013;69(4):413–418. doi: 10.1111/aji.12072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Meekins J. W., Pijnenborg R., Hanssens M., McFadyen I. R., van Asshe A. A study of placental bed spiral arteries and trophoblast invasion in normal and severe pre-eclamptic pregnancies. British Journal of Obstetrics and Gynaecology. 1994;101(8):669–674. doi: 10.1111/j.1471-0528.1994.tb13182.x. [DOI] [PubMed] [Google Scholar]
- 8.Genbacev O., Zhou Y., Ludlow J. W., Fisher S. J. Regulation of human placental development by oxygen tension. Science. 1997;277(5332):1669–1672. doi: 10.1126/science.277.5332.1669. [DOI] [PubMed] [Google Scholar]
- 9.Fox H. Fibrosis of placental villi. The Journal of Pathology and Bacteriology. 1968;95(2):573–579. doi: 10.1002/path.1700950238. [DOI] [PubMed] [Google Scholar]
- 10.Hausvater A., Giannone T., Sandoval Y. H., et al. The association between preeclampsia and arterial stiffness. Journal of Hypertension. 2012;30(1):17–33. doi: 10.1097/hjh.0b013e32834e4b0f. [DOI] [PubMed] [Google Scholar]
- 11.Bellamy L., Casas J.-P., Hingorani A. D., Williams D. J. Pre-eclampsia and risk of cardiovascular disease and cancer in later life: systematic review and meta-analysis. BMJ (Clinical Research ed.) 2007;335(7627):p. 974. doi: 10.1136/bmj.39335.385301.be. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Orabona R., Sciatti E., Vizzardi E., et al. Endothelial dysfunction and vascular stiffness in women with previous pregnancy complicated by early or late pre-eclampsia. Ultrasound in Obstetrics & Gynecology. 2017;49(1):116–123. doi: 10.1002/uog.15893. [DOI] [PubMed] [Google Scholar]
- 13.Sibai B., Dekker G., Kupferminc M. Pre-eclampsia. The Lancet. 2005;365(9461):785–799. doi: 10.1016/s0140-6736(05)71003-5. [DOI] [PubMed] [Google Scholar]
- 14.Bagrov A. Y., Shapiro J. I. Endogenous digitalis: pathophysiologic roles and therapeutic applications. Nature Clinical Practice Nephrology. 2008;4(7):378–392. doi: 10.1038/ncpneph0848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Graves S. W., Valdes R., Jr., Brown B. A., Knight A. B., Craig H. R. Endogenous digoxin-immunoreactive substance in human pregnancies. The Journal of Clinical Endocrinology & Metabolism. 1984;58(4):748–751. doi: 10.1210/jcem-58-4-748. [DOI] [PubMed] [Google Scholar]
- 16.Gusdon J. P., Jr., Buckalew V. M., Jr., Hennessy J. F. A digoxin-like immunoreactive substance in preeclampsia. American Journal of Obstetrics and Gynecology. 1984;150(1):83–85. doi: 10.1016/s0002-9378(84)80114-3. [DOI] [PubMed] [Google Scholar]
- 17.Elkareh J., Kennedy D. J., Yashaswi B., et al. Marinobufagenin stimulates fibroblast collagen production and causes fibrosis in experimental uremic cardiomyopathy. Hypertension. 2007;49(1):215–224. doi: 10.1161/01.hyp.0000252409.36927.05. [DOI] [PubMed] [Google Scholar]
- 18.Elkareh J., Periyasamy S. M., Shidyak A., et al. Marinobufagenin induces increases in procollagen expression in a process involving protein kinase C and Fli-1: implications for uremic cardiomyopathy. American Journal of Physiology-Renal Physiology. 2009;296(5):F1219–F1226. doi: 10.1152/ajprenal.90710.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Averina I. V., Tapilskaya N. I., Reznik V. A., et al. Endogenous Na/K-ATPase inhibitors in patients with preeclampsia. Cellular and Molecular Biology (Noisy-le-Grand, France) 2006;52(8):19–23. [PubMed] [Google Scholar]
- 20.Nikitina E. R., Mikhailov A. V., Nikandrova E. S., et al. In preeclampsia endogenous cardiotonic steroids induce vascular fibrosis and impair relaxation of umbilical arteries. Journal of Hypertension. 2011;29(4):769–776. doi: 10.1097/hjh.0b013e32834436a7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Orabona R., Sciatti E., Vizzardi E., et al. Ultrasound evaluation of left ventricular and aortic fibrosis after pre-eclampsia. Ultrasound in Obstetrics & Gynecology: The Official Journal of the International Society of Ultrasound in Obstetrics and Gynecology. 2018;52(5):648–653. doi: 10.1002/uog.18825. [DOI] [PubMed] [Google Scholar]
- 22.Blaustein M. P., Hamlyn J. M. Role of a natriuretic factor in essential hypertension: an hypothesis. Annals of Internal Medicine. 1983;98(5):785–792. doi: 10.7326/0003-4819-98-5-785. [DOI] [PubMed] [Google Scholar]
- 23.Fedorova O. V., Emelianov I. V., Bagrov K. A., et al. Marinobufagenin-induced vascular fibrosis is a likely target for mineralocorticoid antagonists. Journal of Hypertension. 2015;33(8):1602–1610. doi: 10.1097/hjh.0000000000000591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Bagrov A. Y., Shapiro J. I., Fedorova O. V. Endogenous cardiotonic steroids: physiology, pharmacology, and novel therapeutic targets. Pharmacological Reviews. 2009;61(1):9–38. doi: 10.1124/pr.108.000711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Orabona R., Sciatti E., Prefumo F., et al. Pre-eclampsia and heart failure: a close relationship. Ultrasound in Obstetrics & Gynecology: The Official Journal of the International Society of Ultrasound in Obstetrics and Gynecology. 2018;52(3):297–301. doi: 10.1002/uog.18987. [DOI] [PubMed] [Google Scholar]
- 26.Ohmaru-Nakanishi T., Asanoma K., Fujikawa M., et al. Fibrosis in preeclamptic placentas is associated with stromal fibroblasts activated by the transforming growth factor-β1 signaling pathway. The American Journal of Pathology. 2018;188(3):683–695. doi: 10.1016/j.ajpath.2017.11.008. [DOI] [PubMed] [Google Scholar]
- 27.Pruthi D., Khankin E. V., Blanton R. M., et al. Exposure to experimental preeclampsia in mice enhances the vascular response to future injury. Hypertension. 2015;65(4):863–870. doi: 10.1161/hypertensionaha.114.04971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Chen K. K., Kovaříková A. Pharmacology and toxicology of toad venom. Journal of Pharmaceutical Sciences. 1967;56(12):1535–1541. doi: 10.1002/jps.2600561202. [DOI] [PubMed] [Google Scholar]
- 29.Hitt M., Ettinger D. D. Toad toxicity. The New England Journal of Medicine. 1986;314(23):1517–1518. doi: 10.1056/nejm198606053142320. [DOI] [PubMed] [Google Scholar]
- 30.Brubacher J. R., Lachmanen D., Ravikumar P. R., Hoffman R. S. Efficacy of digoxin specific Fab fragments (Digibind®) in the treatment of toad venom poisoning. Toxicon. 1999;37(6):931–942. doi: 10.1016/s0041-0101(98)00224-4. [DOI] [PubMed] [Google Scholar]
- 31.Brubacher J. R., Ravikumar P. R., Bania T., Heller M. B., Hoffman R. S. Treatment of toad venom poisoning with digoxin-specific Fab fragments. Chest. 1996;110(5):1282–1288. doi: 10.1378/chest.110.5.1282. [DOI] [PubMed] [Google Scholar]
- 32.Lakatta E. G., Levy D. Arterial and cardiac aging: major shareholders in cardiovascular disease enterprises: part I: aging arteries: a “set up” for vascular disease. Circulation. 2003;107(1):139–146. doi: 10.1161/01.cir.0000048892.83521.58. [DOI] [PubMed] [Google Scholar]
- 33.Fedorova O. V., Simbirtsev A. S., Kolodkin N. I., et al. Monoclonal antibody to an endogenous bufadienolide, marinobufagenin, reverses preeclampsia-induced Na/K-ATPase inhibition and lowers blood pressure in NaCl-sensitive hypertension. Journal of Hypertension. 2008;26(12):2414–2425. doi: 10.1097/hjh.0b013e328312c86a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Fedorova O. V., Lakatta E. G., Bagrov A. Y. Endogenous Na,K pump ligands are differentially regulated during acute NaCl loading of Dahl rats. Circulation. 2000;102(24):3009–3014. doi: 10.1161/01.cir.102.24.3009. [DOI] [PubMed] [Google Scholar]
- 35.Fedorova O. V., Agalakova N. I., Talan M. I., Lakatta E. G., Bagrov A. Y. Brain ouabain stimulates peripheral marinobufagenin via angiotensin II signalling in NaCl-loaded Dahl-S rats. Journal of Hypertension. 2005;23(8):1515–1523. doi: 10.1097/01.hjh.0000174969.79836.8b. [DOI] [PubMed] [Google Scholar]
- 36.Periyasamy S. M., Chen J., Cooney D., et al. Effects of uremic serum on isolated cardiac myocyte calcium cycling and contractile function. Kidney International. 2001;60(6):2367–2376. doi: 10.1046/j.1523-1755.2001.00053.x. [DOI] [PubMed] [Google Scholar]
- 37.Adair C. D., Buckalew V. M., Graves S. W., et al. Digoxin immune fab treatment for severe preeclampsia. American Journal of Perinatology. 2010;27(8):655–662. doi: 10.1055/s-0030-1249762. [DOI] [PubMed] [Google Scholar]
- 38.Adair C. D., Buckalew V., Taylor K., et al. Elevated endoxin-like factor complicating a multifetal second trimester pregnancy: treatment with digoxin-binding immunoglobulin. American Journal of Nephrology. 1996;16(6):529–531. doi: 10.1159/000169054. [DOI] [PubMed] [Google Scholar]
- 39.Krep H., Price D. A., Soszynski P., Tao Q.-F., Graves S. W., Hollenberg N. K. Volume sensitive hypertension and the digoxin-like factor reversal by a fab directed against digoxin in DOCA-salt hypertensive rats. American Journal of Hypertension. 1995;8(9):921–927. doi: 10.1016/0895-7061(95)00181-n. [DOI] [PubMed] [Google Scholar]
- 40.Agunanne E., Horvat D., Uddin M. N., Puschett J. The treatment of preeclampsia in a rat model employing Digibind®. American Journal of Perinatology. 2010;27(4):299–305. doi: 10.1055/s-0029-1241739. [DOI] [PubMed] [Google Scholar]
- 41.Agalakova N. I., Reznik V. A., Nadei O. V., et al. Endogenous cardiotonic steroids and vascular fibrosis in preeclampsia. Arterial Hypertension (in Russian) 2019;24(6):684–692. doi: 10.18705/1607-419x-2018-24-6-684-692. [DOI] [Google Scholar]
- 42.Fedorova O. V., Kolodkin N. I., Agalakova N. I., et al. Antibody to marinobufagenin lowers blood pressure in pregnant rats on a high NaCl intake. Journal of Hypertension. 2005;23(4):835–842. doi: 10.1097/01.hjh.0000163153.27954.33. [DOI] [PubMed] [Google Scholar]
- 43.Fedorova O., Ishkaraeva V., Grigorova Y., et al. Antibody to marinobufagenin reverses placenta-induced fibrosis of umbilical arteries in preeclampsia. International Journal of Molecular Sciences. 2018;19(8):p. 2377. doi: 10.3390/ijms19082377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Kaihura C., Savvidou M. D., Anderson J. M., McEniery C. M., Nicolaides K. H. Maternal arterial stiffness in pregnancies affected by preeclampsia. American Journal of Physiology-Heart and Circulatory Physiology. 2009;297(2):H759–H764. doi: 10.1152/ajpheart.01106.2008. [DOI] [PubMed] [Google Scholar]
- 45.Tihtonen K. M. H., Kööbi T., Uotila J. T. Arterial stiffness in preeclamptic and chronic hypertensive pregnancies. European Journal of Obstetrics & Gynecology and Reproductive Biology. 2006;128(1-2):180–186. doi: 10.1016/j.ejogrb.2005.12.026. [DOI] [PubMed] [Google Scholar]
- 46.Ishkaraeva-Yakovleva V. V., Fedorova O. V., Solodovnikova N. G., et al. DigiFab interacts with endogenous cardiotonic steroids and reverses preeclampsia-induced Na/K-ATPase inhibition. Reproductive Sciences. 2012;19(12):1260–1267. doi: 10.1177/1933719112447124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Uddin M. N., Wages J. M., Pantho A. F., et al. 316: novel anti-MBG antibodies protect cytotrophoblast cells from a marinobufagenin-induced preeclampsia phenotype. American Journal of Obstetrics and Gynecology. 2018;218(1):p. S199. doi: 10.1016/j.ajog.2017.10.252. [DOI] [Google Scholar]
- 48.Selye H., Krajny M., Savoie L. Digitoxin poisoning: prevention by spironolactone. Science. 1969;164(3881):842–843. doi: 10.1126/science.164.3881.842. [DOI] [PubMed] [Google Scholar]
- 49.Selye H. Spironolactone actions, independent of mineralocorticoid blockade. Steroids. 1969;13(6):803–808. doi: 10.1016/0039-128x(69)90075-0. [DOI] [PubMed] [Google Scholar]
- 50.de Mendonça M., Grichois M.-L., Pernollet M.-G., et al. Antihypertensive effect of canrenone in a model where endogenous ouabain-like factors are present. Journal of Cardiovascular Pharmacology. 1988;11(1):75–83. doi: 10.1097/00005344-198801000-00012. [DOI] [PubMed] [Google Scholar]
- 51.Pamnani M. B., Whitehorn W. V., Clough D. L., Haddy F. J. Effects of canrenone on blood pressure in rats with reduced renal mass. American Journal of Hypertension. 1990;3(3):188–195. doi: 10.1093/ajh/3.3.188. [DOI] [PubMed] [Google Scholar]
- 52.Balzan S., Nicolini G., Bellitto L., Ghione S., Biver P., Montali U. Effect of canrenone on the digitalis site of Na+/K+-ATPase in human placental membranes and in erythrocytes. Journal of Cardiovascular Pharmacology. 2003;42(1):32–36. doi: 10.1097/00005344-200307000-00005. [DOI] [PubMed] [Google Scholar]
- 53.Tian J., Shidyak A., Periyasamy S. M., et al. Spironolactone attenuates experimental uremic cardiomyopathy by antagonizing marinobufagenin. Hypertension. 2009;54(6):1313–1320. doi: 10.1161/hypertensionaha.109.140038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Semplicini A., Serena L., Valle R., et al. Ouabain-inhibiting activity of aldosterone antagonists. Steroids. 1995;60(1):110–113. doi: 10.1016/0039-128x(94)00005-w. [DOI] [PubMed] [Google Scholar]
- 55.Finotti P., Palatini P. Canrenone as a partial agonist at the digitalis receptor site of sodium-potassium-activated adenosine triphosphatase. The Journal of Pharmacology and Experimental Therapeutics. 1981;217(3):784–790. [PubMed] [Google Scholar]
- 56.Morton A., Panitz B., Bush A. Eplerenone for gitelman syndrome in pregnancy. Nephrology. 2011;16(3):p. 349. doi: 10.1111/j.1440-1797.2010.01396.x. [DOI] [PubMed] [Google Scholar]
- 57.Awan S. Z., Lu J. Management of severe acne during pregnancy: a case report and review of the literature. International Journal of Women’s Dermatology. 2017;3(3):145–150. doi: 10.1016/j.ijwd.2017.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Aldactone® Spironolactone Tablets, USP (NDA 12-151/S-062), 2019, https://www.accessdata.fda.gov/drugsatfda_docs/label/2008/012151s062lbl.pdf.
- 59.Hecker A., Hasan S. H., Neumann F. Disturbances in sexual differentiation of rat foetuses following spironolactone treatment. Acta Endocrinologica. 1980;95(4):540–545. doi: 10.1530/acta.0.0950540. [DOI] [PubMed] [Google Scholar]
- 60.Regitz-Zagrosek V., Blomstrom Lundqvist C., Borghi C., et al. ESC guidelines on the management of cardiovascular diseases during pregnancy: the task force on the management of cardiovascular diseases during pregnancy of the European society of cardiology (ESC) European Heart Journal. 2011;32(24):3147–3197. doi: 10.1093/eurheartj/ehr218. [DOI] [PubMed] [Google Scholar]
- 61.Sadee W., Dagcioglu M., Schroder R. Pharmacokinetics of spironolactone, canrenone and canrenoate-K in humans. The Journal of Pharmacology and Experimental Therapeutics. 1973;185(3):686–695. [PubMed] [Google Scholar]
- 62.Tal D. M., Karlish S. J. Do canrenone and 6,7-dihydroxylated derivatives compete with ouabain at the same site on Na,K-ATPase? Molecular Pharmacology. 1988;34(3):245–249. [PubMed] [Google Scholar]
- 63.Garay R., Rosati C., Meyer P. Na+ transport in primary hypertension. Annals of the New York Academy of Sciences. 1986;488:187–195. doi: 10.1111/j.1749-6632.1986.tb46557.x. [DOI] [PubMed] [Google Scholar]
- 64.Garay R. P., Diez J., Nazaret C., Dagher G., Abitbol J. P. The interaction of canrenone with the Na+,K+ pump in human red blood cells. Naunyn-Schmiedeberg’s Archives of Pharmacology. 1985;329(3):311–315. doi: 10.1007/bf00501886. [DOI] [PubMed] [Google Scholar]
- 65.Kashkin V. A., Shekunova E. V., Egorov A. Y., Bagrov A. Y. Marinobufagenin in urine: a potential marker of predisposition to ethanol and a target for spironolactone. Current Hypertension Reviews. 2018;14(1):35–38. doi: 10.2174/1573402114666180212115518. [DOI] [PubMed] [Google Scholar]
- 66.Agrawal S., Cerdeira A. S., Redman C., Vatish M. Meta-analysis and systematic review to assess the role of soluble FMS-like tyrosine kinase-1 and placenta growth factor ratio in prediction of preeclampsia: the SaPPPhirE study. Hypertension. 2018;71(2):306–316. doi: 10.1161/hypertensionaha.117.10182. [DOI] [PubMed] [Google Scholar]