Abstract
Measurements of spin-lock relaxation rates in the rotating frame (R1ρ) at high magnetic fields afford the ability to probe not only relatively slow molecular motions, but also other dynamic processes, such as chemical exchange and diffusion. In particular, measurements of the variation (or dispersion) of R1ρ with locking field allow the derivation of quantitative parameters that describe these processes. Measurements in deuterated solutions demonstrate the manner and degree to which exchange dominates relaxation at high fields (4.7 T, 7 T) in simple solutions, whereas temperature and pH are shown to be very influential factors affecting the rates of proton exchange. Simulations and experiments show that multiple exchanging pools of protons in realistic tissues can be assumed to behave independently of each other. R1ρ measurements can be combined to derive an exchange rate contrast (ERC) that produces images whose intensities emphasize protons with specific exchange rates rather than chemical shifts. In addition, water diffusion in the presence of intrinsic susceptibility gradients may produce significant effects on R1ρ dispersions at high fields. The exchange and diffusion effects act independently of each other, as confirmed by simulation and experimentally in studies of red blood cells at different levels of oxygenation. Collectively, R1ρ measurements provide an ability to quantify exchange processes, to provide images that depict protons with specific exchange rates and to describe the microstructure of tissues containing magnetic inhomogeneities. As such, they complement traditional T1 or T2 measurements and provide additional insights from measurements of R1ρ at a single locking field.
Keywords: T1ρ, T1ρ dispersion, chemical exchange, diffusion, susceptibility, spin-locking
INTRODUCTION
In MRI, the spin–lattice relaxation rate in the rotating frame (R1ρ), measured using spin-locking pulse sequences, has been exploited previously to characterize tissues and to assess pathological changes in various research and clinical applications (1–5). The rate R1ρ is usually measured by application of an on-resonance spin-locking field along the direction of the bulk transverse magnetization in the rotating reference frame for a series of locking times. The decay of locked magnetization with increasing locking time may usually be well fitted to a mono-exponential with a characteristic time constant T1ρ (=1/R1ρ) (6,7). In conventional MRI, T1ρ-weighted imaging has been interpreted mainly as reflecting the relatively slow motional characteristics of macromolecules, especially when implemented at low static field strengths (8,9). Few previous reports of spin-lock imaging have emphasized its ability to quantify other dynamic processes, such as chemical exchange or diffusion at high fields. In this mini-review, we summarize some relevant theoretical developments and experimental results that focus on the use of spin-locking methods to study such exchange processes in biological tissues. In particular, we highlight recent work that illustrates how appropriate spin-locking measurements may be used to quantify exchange rates, to produce novel image contrast that selectively emphasizes protons characterized by specific exchange rates, and to characterize magnetically inhomogeneous media in new ways. Collectively, these studies suggest that spin-locking methods can be a powerful adjunct to the range of MRI methods available for studies of tissue and for the detection of pathological or physiological changes, especially at higher magnetic field strengths.
Most previous studies of T1ρ-based contrast in MRI have been performed at relatively low B0 fields, and these have emphasized the sensitivity of T1ρ measurements to slow molecular motions around the Larmor frequency corresponding to the locking field, typically from ~100 Hz to a few kHz (10–13). The low-frequency motions of macromolecules may affect water via dipolar interactions, and T1ρ may often provide a more specific probe of such effects than other relaxation measurements. Variations in R1ρ with locking field (R1ρ dispersion) then reflect changes in the spectral density of the local dipolar fields experienced by protons as a result of relatively slow molecular motions. However, at higher fields, there may be additional significant contributions to R1ρ (as well as R2) from chemical exchange between sites of different chemical shifts, and these increase rapidly with field strength and may dominate the dispersion curves measured experimentally (14). It is noteworthy that recent reports have shown that measured relaxation rates have a more complex dependence when spin-locking is achieved with a train of adiabatic pulses, as the modulation in the pulse as a function of time changes the exchange conditions through the time-dependent effective field (15,16). Mäkelä et al. (17) have investigated the B0 dependence of R1ρ dispersion in protein phantoms and rat brains in vivo and, although they claim that classical dipolar interactions are the most important mechanism, they also state that other effects, such as diffusion and exchange, probably influence the relaxation. Below, we consider the behavior under conditions of only uniform spin-locking pulses.
Chemical exchange describes the physical swapping of hydrogen nuclei from a bulk water pool to one or more smaller metabolite pools, as illustrated in Fig. 1. The exchange rate and chemical shift of the smaller pool determine the magnitude of such exchange contributions to transverse dephasing, but the application of appropriately strong locking fields can reduce these. This ability to reduce exchange contributions by varying the locking field gives rise to a dispersion in R1ρ values with locking field, which then provides information on the exchange processes in the intermediate to fast time scale regimes (18). This is of particular importance for the imaging of compounds with fast exchanging protons, such as in hydroxyl groups (−OH) found in compounds such as glucose or glutamic acid, key energy sources and neurotransmitters in the human brain, respectively. A corresponding dispersion of R2 values with pulse rate in Carr–Purcell–Meiboom–Gill (CPMG) sequences can, in principle, provide similar exchange information by comparing the different apparent T2 values obtained at long pulse spacings, where exchange will significantly influence the linewidth, and short pulse spacings, where exchange will have very little impact. However, the practicality of using multi-echo sequences in imaging is limited by technical constraints on the achievable pulse rates for probing relatively slower exchange (14). Moreover, although dipolar effects may still contribute to the dispersion of R1ρ, unlike chemical exchange effects, they do not increase rapidly with higher B0 fields.
Figure 1.
Schematic diagram of the chemical exchange of labile hydrogen nuclei between water and hydroxyls on a single glucose molecule. Water is represented as pool a and glucose as pool b, and so the corresponding exchange rate from water to glucose is kab and from glucose to water is kba.
Effects other than chemical exchange have also been reported to affect the dispersions of R2 and R1ρ, including water diffusion through internal field gradients caused by interfaces of unequal susceptibility. For example, Hills et al. (19) reported a double dispersion behavior of R2 as a function of reciprocal CPMG 90°–180° pulse spacing at 7 T in suspensions of packed Sephadex beads carrying hydroxyl protons in water. The dispersion curve was compared with theoretical simulations that took only chemical exchange into account, and accurately matched the short pulse spacing data, but failed to coincide with the long pulse spacing portion of the curve. This disparity for long pulse spacings was postulated to be a result of water diffusion into and out of the Sephadex beads, as well as through the intrinsic gradients produced immediately outside the beads. This diffusion produced a separate dispersion because the local field experienced by spins evolved on a much slower timescale (around 4 Hz) compared with the chemical exchange effects occurring at 2500 Hz. Figure 2 shows schematically the random paths of spins diffusing through the spaces between orderly packed microspheres. The low-frequency dispersion of R1ρ reflects the timescale over which magnetization is dephased, which depends directly on the magnetic microstructure of the medium. Similar findings were confirmed experimentally in plant tissues (20). More recently, we have shown how diffusion through susceptibility gradients, in the absence of chemical exchange, can produce significant R1ρ dispersion and reveal details of the inhomogeneities causing the gradients, and that such effects are measurable in biological tissues. Our experimental results have verified the new theoretical predictions of such effects (21,22). The diffusion of water through susceptibility gradients could be very relevant to studies of the tissue microvasculature, which may induce intrinsic gradients within tissues. However, some previous studies have claimed that T1ρ is not sensitive to changes in intravascular susceptibility. For example, Kettunen et al. (23) have shown that T1ρ is only marginally sensitive to intravascular effects after injection of the susceptibility agent AMI-227 using a locking field of 1.6 G (≈6800 Hz), whereas T2 is much more sensitive to these effects. We show here that significant effects are not expected using such a strong locking field, but that T1ρ is very sensitive to the presence of susceptibility agents when the locking field is much weaker. Our simulations and theory indicate that diffusion-related R1ρ dispersion effects depend on the relationship between the locking field frequency and the time needed for water molecules to diffuse distances of the order of the size of the vessels, which is typically on the order of many milliseconds. Recently, Rane et al. (5) have provided strong evidence that T1ρ is sensitive to intravascular susceptibility changes at low locking fields by detecting blood oxygenation level-dependent (BOLD) signals in the brain at different locking strengths. Thus, in addition to chemical exchange, R1ρ dispersion measurements can report on diffusion processes in inhomogeneous tissues.
Figure 2.
An example of four nuclei exhibiting random Brownian motion through local magnetic field gradients manifested from the presence of spherical perturbers of susceptibility χ in an external magnetic field. This illustrates the local field shifts experienced by a diffusing nucleus through the superposition of dipole fields.
Previous studies using spin-locking techniques have shown that R1ρ may be a versatile biomarker of pathological or physiological changes that occur, for example, in animal stroke and tumor models (3,24–27), with activation in human brain in functional MRI (fMRI) (4,12,28), in amyloid plaques in Alzheimer’s disease (2,13,29–31), or with articular cartilage degradation (1,32–34). However, although single measurements of rates may be useful, few of these studies have measured the dispersion of R1ρ with locking field at high static field, which we show here contains much additional information. We emphasize below that, by appropriate analysis of the dispersion of R1ρ, chemical exchange rates may be explicitly derived and these, in turn, may reflect other properties of interest, such as temperature or pH (27,35,36). Moreover, we show that appropriate combinations of spin-lock measurements can be used to create a novel form of image contrast that emphasizes populations characterized by specific exchange rates rather than other parameters. This sensitivity of measurements of R1ρ to exchange parameters implies that spin-locking methods may provide an alternative approach to the assessment of chemical exchange from chemical exchange saturation transfer (CEST), which is currently in widespread use for such purposes. Spin-locking methods are similar in nature to CEST methods (37,38), and recent studies have confirmed that each can potentially provide complementary information on tissues (39). However, the quantification of chemical exchange processes using CEST is not trivial because other processes, such as magnetization transfer with macromolecules and nuclear Overhauser effects (NOEs), can confound quantitative measurements. Moreover, CEST relies on distinguishing proton populations by their specific chemical shifts, and is well suited to study processes only in which the exchange rate (kex) is relatively slow compared with the chemical shift δ (kex/δ < 1). By contrast, spin-locking measurements are particularly sensitive to chemical exchange rates in the intermediate and fast exchange regimes (kex/δ >> 1), where CEST spectra begin to coalesce. Thus, spin-locking methods may have advantages in studies in which other approaches do not perform well. Examples of these applications are provided below.
THEORY
Chemical exchange
Labile hydrogen protons, such as hydroxyls (−OH), amides (NH+) or amines (NH2), may undergo physical exchange with solvent water protons at a rate dependent on several factors, such as temperature, pH and chemical environment. These exchange sites, in general, have different chemical shifts and intrinsic relaxation rates relative to water protons. The chemical shift differences correspond to different intrinsic precession rates, so that exchange leads to a mixture of different phases resulting in transverse signal losses. Spin-locking methods typically first nutate the bulk magnetization into the transverse plane with either an adiabatic or hard radiofrequency (RF) pulse, and subsequently ‘lock’ the magnetization by application of a relatively long-duration, low-amplitude RF pulse of strength B1 = ω1/γ along the direction of the magnetization. Figure 3a describes how the magnetization is tipped into the transverse plane and Fig. 3b illustrates the components of the effective field in the rotating frame. Transverse magnetization under the influence of this continuous, in phase and on-resonance RF locking field decays along the same axis at the rate R1ρ = 1/T1ρ. Chemical shift differences introduce a small longitudinal field component, but increasing the locking amplitude tends to ensure that the effective field remains in the transverse plane so that dephasing effects are reduced, which decreases the relaxation rate R1ρ in a manner that depends on the timescale of the relevant molecular interactions relative to the locking field period. This differs from CEST experiments in which saturation pulses are applied at a range of frequency offsets to partially saturate longitudinal magnetization to decrease the solvent signal after the exchange of saturated spins. Although the experiments are different, simulations have shown that off-resonance spin-locking and CEST are derived from the same eigenvalue of the Bloch–McConnell equations and the measurements are sensitive to the same exchange effects (37).
Figure 3.
(a) The application of an on-resonance B1 pulse tips the magnetization into the transverse plane. (b) The B1 spin-lock pulse, together with the field offset ΔΩ, comprises the effective field Beff, about which the magnetization precesses for the duration of the locking time.
The behavior of a system consisting of two pools (labeled pool a and pool b) undergoing chemical exchange may be analyzed using the Bloch–McConnell equations shown below (40).
| [1] |
Here, for pool a (bulk water pool), is the magnetization for i = x, y or z, are the longitudinal and transverse relaxation rates, Δωa is the chemical shift of pool a, is the exchange rate from pool a to pool b (the exchangeable solute), pa is the pool fraction size, ω1 is the magnitude of the spin-lock RF field (here applied along the x direction in the rotating frame) and is the equilibrium magnetization. The corresponding expressions for pool b are identical, except with switched indices. Trott and Palmer (41) solved these equations for R1ρ by finding the largest real eigenvalue of the determinant in the 6 × 6 matrix of Equation (1) using the assumptions that and for a dilute solution (pa >> pb) to derive the following expression:
| [2] |
Here θ = tan−1(ω1/Δω), δ = Δωb – Δωa, k = kab + kba, , and , where Δω is a population-averaged frequency offset. The first two terms on the right-hand side of Equation (2) are the effective relaxation rate governed by spin–lattice contributions at high locking field and the conventional transverse relaxation at very low locking field, and the third term is the chemical exchange contribution Rex.
Although the Trott and Palmer formalism has proven to be useful in some conditions, a more realistic analysis was provided by Chopra et al. (42), who derived a separate expression in a similar manner by assuming asymmetric pool fractions (pa >> pb), that RF pulses are applied on-resonance with the water pool (Δωa = 0) and that exchange is not slow compared with relaxation . Finding the dominant eigenvalue under these assumptions leads to the following equation:
| [3] |
Where the parametrs , , and . This accords well with the situation in biological tissues and reduces to Equation (2) for practical on-resonance conditions, when and . If is measured over a range of locking fields, this model may be used to fit the dispersion curve for the three variables R2, and Sρ. In the case in which (usually the case of interest in tissues), and the exchange rate kba can be explicitly found if the chemical shift is known.
When the second derivative with respect to ω1 of Equation (3) is equal to zero, the dispersion curve undergoes an inflection, and the corresponding locking field is then . The two models use slightly different assumptions in their respective derivations and give different functional forms, but produce almost identical results when simulated numerically with biologically relevant parameter values. They have been shown to model dynamic effects well for small exchanging proton pool fractions (pb << 1) in Bloch simulations as well as in model systems of various metabolites (18,35,43). The Chopra model shown in Equation (3) is slightly more general for the on-resonance spin-locking condition as the individual site relaxation rates do not need to be identical, and so this model is used for further data analyses in this article. The Trott and Palmer model may be needed when off-resonance locking pulses are used.
When there are two distinct exchanging proton pools with different chemical shifts and exchange rates, they may exhibit a double dispersion behavior that can be approximated very well by linearly adding the individual exchange contributions of the dispersions for small pool fractions. This can be extended to n exchanging pools by the following equation:
| [4] |
Equation (4) assumes that is the transverse relaxation rate of the bulk water pool which can be measured separately and that there is negligible exchange directly between any two minority pools not involving the bulk water pool (minor exchange, e.g. kbc, kcb). The term is subtracted from the summation so that only the exchange contributions from each dispersion are added and the curve is not incorrectly shifted to faster relaxation rates. Although this may be expanded for any number of pools, fitting dispersion curves in practice to more than two sets of dispersion parameters becomes too inexact to estimate the multiple parameters with any reasonable accuracy. Relaxation in most tissues exhibiting exchange in vivo may be typically described by one or two exchange sites, making the double dispersion method a relevant and useful extension of the theory. Bloch–McConnell simulations demonstrate the validity of this approximation for most realistic in vivo concentrations and show that, even in the presence of minor exchange, this model still agrees well for small pool fractions. Trott and Palmer (44) also derived an expression for n-pool exchange as an extension of their single exchange pool theory described above, which is practically identical to Equation (4) numerically and performs well in the fitting of experimental data. They also introduced another expression that accounts for minor exchange, but this consists of too many unknown variables to fit in practice, making it theoretically interesting but impractical for the estimation of exchange parameters experimentally.
Exchange rate contrast
In the case in which , the single exchange pool model may be approximately written in the following form:
| [5] |
By measuring three relaxation rates at judiciously selected locking strengths of ω1 ≈ 0, ∞ and any intermediate value, Equation (5) may be used to define an exchange rate contrast (ERC) by which images may be generated that exhibit novel contrast based on the dominant exchange rates within the system:
| [6] |
Here, , and , an intrinsic characteristic of the sample. The ERC value depends on the choice of intermediate locking field relative to this intrinsic parameter. The critical locking frequency, , corresponds to the field at which the dispersion displays an inflection point (maximum negative value of derivative). The ERC is a smooth and Lorenztian-like curve when plotted versus ω1 which displays a peak value of unity when . It falls off monotonically for values of ω1 different from this value, and the width of ERC is again described by the parameter α. ERC is thus a spectral response function in which the magnitude depends on the relationship of the locking field to the exchange rate and chemical shift frequency. This provides the basis for producing images that emphasize species with particular exchange rates. Cobb et al. (18) have shown that this can be used in an imaging context for single site exchange to emphasize specific exchange rates in studies of potential exchange-based contrast agents in vitro. The relaxation rates in Equation (6) may be replaced with image intensities from a single locking time at the respective locking amplitudes to produce parametric images that obey a similar behavior and emphasize the protons undergoing exchange at a rate corresponding to the selected intermediate locking field, i.e. exchange-weighted image contrast (EWIC). If this intermediate locking amplitude is chosen to be at the ERC peak of a specific exchanging species in the presence of single species exchange, the derived image intensity will scale with the local concentration of that species. By changing the locking field of the acquired images, individual proton pools may be emphasized over others and distinguished by their specific exchange properties. The EWIC image is much faster to derive as it requires only three images, but is sensitive to the spin-lock time used for each image and thus complicates the accurate estimation of the exchange rate. ERC maps calculated from R1ρ maps at three locking fields are much more robust for the estimation of exchange rates as there is no dependence on the locking time, but these require more images and longer acquisition times. Collectively, these methods provide a novel form of parametric image in which contrast does not depend on chemical shift, but explicitly on the exchange rates of protons within the sample.
Diffusive exchange
Brownian motion of water in the presence of inhomogeneous fields also gives rise to a spread in proton signal phases similar to the manner of exchange between different chemical environments. Water diffusion in the presence of gradients induced by intrinsic susceptibility variations may therefore also give rise to dispersion in R1ρ as a result of the fluctuating local fields experienced by the protons. The intrinsic gradients cause dephasing which, again, may be partially mitigated by strong locking fields. The variation in R1ρ with locking field then depends on the timescale of the local field fluctuations, which provides information on the mean spacing and dimensions of the inhomogeneities for a given diffusion rate. In general, the field variations experienced by nuclei are complex and usually random, but may be described by a correlation function with a characteristic correlation time (τc). A simple example that serves to approximate a medium with packed inhomogeneities can be analyzed by assuming that the local field fluctuates sinusoidally:
| [7] |
Here, x is the spatial location of the diffusing spin, g is the mean gradient strength and q is the spatial frequency of the gradient field. For this case, R1ρ can be estimated using a closed form expression for the diffusion-dependent dispersion (45):
| [8] |
In Equation (8), γ is the gyromagnetic ratio and D is the self-diffusion coefficient. It should be noted that this expression, derived for a specific geometry, hinges only on the identification of an appropriate correlation time for the field variations, which is given by the following equation:
| [9] |
For the more general case, as long as the correlation function decays exponentially, the same type of dependence is predicted, but with a more realistic and appropriate interpretation of the effective spatial frequency q. Other specific cases have been investigated for other relaxation processes (46–49).
Setting the second derivative of R1ρ to zero identifies the inflection point of the dispersion, . The correlation time is a parameter of primary interest because it describes the average time for a spin to diffuse through a characteristic dimension of the local field. For the sinusoidal field, this corresponds to one wavelength, a measure of the spatial distribution of the inhomogeneities, but not the strength of the gradients originating from the inhomogeneities. For more complex media, it is the time to diffuse a distance equal to the reciprocal of the width of the spatial frequency spectrum of the field distribution (22,50,51). Other factors, such as temperature, may influence the correlation time because of the dependence on the intrinsic diffusion coefficient.
Combined effects
Both chemical exchange and diffusion phenomena may occur simultaneously in real tissues, in which case these effects add independently for small exchange pool fractions:
| [10] |
This equation may be used to describe a system containing a dominant exchanging pool and with significant intrinsic gradients that affect water diffusion, and to fit for R2, , , g2D, and q2D. Models with multiple exchanging pools and diffusion usually will include too many variables to reliably fit in practice, and so only weighted averages of the various components may then be estimated. There are, however, several relevant examples of biological tissues for which precise parameters may be obtained. For example, in whole blood, diffusion through internal gradients caused by the presence of deoxyhemoglobin in red blood cells (RBCs) and chemical exchange effects caused by the existence of metabolites, such as glucose, in the plasma may concurrently affect the R1ρ dispersion as described above.
EVALUATION OF FACTORS AFFECTING R1ρ DISPERSION
Quantification of chemical exchange contributions
Dipolar effects are known to govern transverse relaxation in water and tissue at low fields (9), but chemical exchange between water molecules and labile protons, such as those on amide, amine or hydroxyl groups, are believed to be the dominant mechanism of R1ρ dispersion at high static fields where the chemical shift frequencies become larger (18,43). Although computer simulations and theory predict this trend (39,52), there has been no systematic experimental study to our knowledge that quantifies the evolution of dominant effects from dipolar interactions at low field strengths to chemical exchange at high field strengths. To verify and illustrate when exchange begins to dominate at high fields, experiments were performed at static fields of 4.7 and 7 T in solutions of constant concentrations of exchanging species with varying levels of deuteration. Substitution of protons with deuterons reduces dipolar coupling, but also reduces the number of protonated sites available for exchange. Solutions of 200 mm glucose were prepared in phosphate-buffered saline (PBS) with 0, 20, 40, 60 and 80% deuterium oxide, and proton R1ρ dispersions were measured with 10 logarithmically spaced locking fields ranging from 50 to 8000 Hz at each static field strength, each with six logarithmically spaced locking times ranging from 20 ms to 1 s. A simple mono-exponential fit was used to determine the relaxation rate R1ρ from the decay curves. The spin-locking pulse sequence used was a standard pulse sequence described by Witschey et al. (53) designed to mitigate B0 and B1 field inhomogeneities. Dispersions were fitted using the Chopra model of Equation (3) to estimate exchange parameters and to analyze the effects of B0 and deuteration. Although the low-frequency limit of the dispersion provides information about T2 values, T1 values corresponding to the asymptotic limit of the dispersion curve were more accurately measured using a standard inversion recovery sequence.
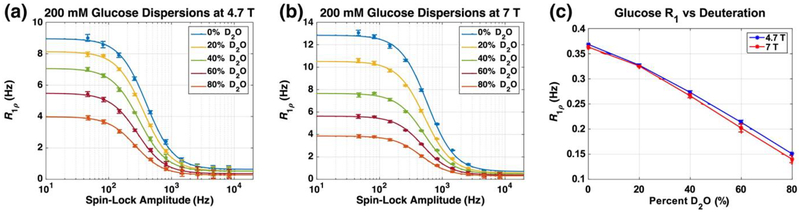
Figure 4 demonstrates that the substitution of protons by deuterons reduces the relaxation rates at low locking field and the magnitudes of the variations in R1ρ with locking field decrease in a concentration-dependent manner. These data are consistent with the loss of chemical exchange contributions to spin–lattice relaxation in the rotating frame as protons are substituted by deuterons. The dispersion magnitudes and the effects of deuteration on the dispersions also increase as the static field increases. The proton R1 values decrease significantly as the D2O concentration increases, as expected because the dipolar field of the deuteron is much less than that of the proton (54). The low locking field limits of the dispersions, R1ρ(0), decrease with D2O concentration, which is apparent in Fig. 5. Fitting the low limit R1ρ values versus proton concentration to a straight line allows one to elucidate information about the effect of exchange and dipolar interactions. The value of this fitted line as the proton concentration approaches zero provides insight into the dipolar contribution, which changes with the static field strength. The estimated contribution of dipolar effects is roughly 59% lower at 7 T than at 4.7 T, as expected, confirming that dipolar interactions decrease with field. The slope of the fitted line increases by a factor of 1.82 at 7 T compared with that at 4.7 T as a result of the effect of chemical exchange, close to the theoretical prediction (excluding all other factors) of 2.25. Moreover, the effective exchange rate is slowed by the introduction of deuterium as expected (55), which is evident by the shift of the inflection points of the dispersions to lower frequencies. The slowed exchange rate reduces the change in dispersion magnitude and is shown to have a larger effect at higher field.
Figure 4.
R1ρ dispersions of 200 mm glucose solutions (pH 7.4) in phosphate-buffered saline (PBS) with the specified concentration of D2O are shown at 4.7 T in (a) and at 7 T in (b). The R1 values of each concentration in (c) decrease at both 4.7 and 7 T.
Figure 5.
The low locking field limit of R1ρ versus the percentage of protons is plotted and fitted to a linear model. The increased slope at 7 T indicates how the chemical exchange effect becomes more prominent, whereas the decreased y-intercept value at 7 T reveals the decreased dipolar contribution to relaxation.
Effects of temperature
Temperature and pH may greatly affect the exchange parameters and therefore the corresponding R1ρ dispersion. In order to establish the effects of temperature, R1ρ dispersion curves were measured for a 200 mm glucose sample (pH 7.4) at five temperatures ranging from 19 to 40 °C. The sample was heated by blowing hot air down the bore of the magnet and the temperature was monitored using a thermocouple attached to an animal physiologic monitoring system (SA Instruments, Stony Brook, NY, USA). Dispersions were measured using 10 logarithmically spaced locking amplitudes ranging from 50 to 10 000 Hz and fitted to the Chopra model for analysis.
Figure 6a, b shows that increasing the temperature increases the exchange rates as derived from the fits to the dispersion data. The dispersion magnitudes increase to a maximum value and then decrease beyond a certain high exchange rate, but the inflection points of the dispersions increase monotonically with temperature.
Figure 6.
(a) R1ρ dispersions of 200 mm glucose (pH 7) at 7 T for various temperatures. (b) The estimated exchange rate, calculated from the parameters used in the dispersion fits, plotted versus temperature. (c) R1ρ dispersions of 200 mm glucose (T = 19 °C) at 7 T for various pH values. (d) The estimated exchange rates plotted versus pH.
Effects of pH
The effects of pH were determined by measuring R1ρ dispersions in two sets of samples of 200 mm glucose, one with seven samples with pH in the range pH 3–9 and one with five samples with pH in the range pH 6.00–7.40. All measurements were made at 7 T and the dispersions were acquired in the same manner as the temperature dispersions, but at bore temperature (19 °C). The Chopra model was again used to fit the dispersion data to determine the exchange rates, assuming a chemical shift of Δωb = 1 ppm.
Although temperature and pH both significantly affect the rates of exchange between proton pools, changing the pH has more complex effects because the exchange rate reaches a minimum just above pH 6, and increases with a change in pH in either direction, as shown by the estimated exchange rates in Fig. 6d which are derived from the dispersions shown in Fig. 6c. Figure 7 shows the dispersions over a smaller pH range to illustrate how small increases in pH around physiological levels may cause large changes in the exchange rate and a measurable shift and increase in the dispersion inflection.
Figure 7.
(a) R1ρ dispersions of 200 mm glucose (T = 19 °C) at 7 T over a more relevant range of pH values shows a more exponential trend in the estimated exchange rates shown in (b).
Effects of multiple pools
Multiple exchanging populations may each have an effect on both CEST spectra and R1ρ dispersions. Cobb et al. (39) verified the manner in which CEST spectra can be summed for the case of peptide and sugar exchange pools, and Trott et al. (44) have shown how the exchange-dependent contributions to R1ρ change as a function of the population-averaged chemical shift. The effects of chemical exchange on R1ρ dispersion in mixtures are further quantified here using Bloch–McConnell finite difference simulations. The exchange rates of two different species with water were selected to be close together for one scenario (1 and 3 kHz) and far apart in another (1 and 30 kHz) to visually demonstrate the nature in which the dispersion contribution from each species adds for small pool fractions. Simulations were performed using three exchange pools with pb = pc = 1%, Δωb = 0.75 ppm, Δωc = 0.5 ppm, R1a = R1b = R1c = 0.1 Hz, R2a = R2b = R2c = 0.4 Hz, kba = 1 kHz and kca = 3 kHz. The other scenario assumed Δωb = 0.75 ppm, Δωc = 1.75 ppm and kca = 30 kHz. Furthermore, simulations were conducted with various exchange rates up to 5 kHz between pools b and c to determine the degree to which exchange between minor pools affects R1ρ dispersion. Subsequently, solutions of PBS mixed with 100 mm creatine, 100 mm glucose, 50 mm uracil and mixtures of 100 mm creatine + 100 mm glucose and 100 mm creatine + 50 mm uracil were prepared and studied at 7 T to measure R1ρ dispersions and validate how the dispersions can be summed in practice.
Bloch–McConnell simulations were subsequently performed with two exchange pools for various pool fractions of one of the exchange pools. The exchange rates of the two sites were chosen to be kba = 30 000 Hz and kca = 800 Hz, which are disparate for illustrative purposes. The pool fraction for pool b remained constant at 1%, whereas pool c varied from 0% to 2%. Finite difference Bloch simulations were executed with time steps of 10 ns to generate dispersions depicting two inflections that were analyzed in terms of the ERC described in Equation (6). The ERC values were examined for each pool fraction at the spin-lock amplitude of 254 Hz, as this is the amplitude that generates the maximum ERC value of unity for the simulated dispersion of pool c alone. This was demonstrated in solutions of glucose and uracil by varying the concentration of glucose from 0 to 200 mm whilst holding the concentration of uracil constant at 40 mm. R1ρ dispersions were measured at 7 T by making R1ρ maps at 12 locking amplitudes (50–10 000 Hz) with a standard spin-lock-prepared fast spin-echo imaging sequence to demonstrate how the double dispersion changes in practice. ERC analysis was performed on a pixel-by-pixel basis using Equation (6) with the R1ρ maps at B1 = 50, 545 and 10 000 Hz to show image intensity changes based on glucose concentration.
The results of the simulations shown in Fig. 8a, c demonstrate the manner in which two exchanging pools, each with 1% pool fraction in 98% water, can contribute to an overall dispersion with two inflection points. The resulting double dispersions for both cases are very close to the linear addition of the individual exchange contributions of the dispersion curves of each single species using Equation (4); the small discrepancy arises from the fact that the linearly added curves do not take into account the 1% drop in the bulk water pool fraction when the simulation is run with two exchanging pools. These simulations do not account for the contributions to R1ρ that may occur from the natural abundance of H2O17 (0.037 atomic %), which may affect slightly the low B1 dispersion, although such effects should be small compared with the exchange effects caused by the size of the pool fractions used in the simulation (56–58). These systems were also analyzed allowing for exchange between the minor pools at various rates. Figure 8b, d shows that the effects of slow minor exchange are almost negligible, but the dispersions begin to shift and coalesce at very fast exchange rates. In practice, these exchange sites tend to interact much more frequently with bulk water than with each other, making the effects of inter-minor pool exchange insignificant in most cases. Environments containing multiple exchange pools with similar exchange rates will also, in principle, exhibit multiple dispersions, but may appear as a single dispersion as in Fig. 8a because the inflection points are sufficiently close that only a weighted average is observed. Both cases are also experimentally demonstrated in Fig. 9, which shows the R1ρ dispersions in solutions of creatine, glucose, uracil and corresponding mixtures. These dispersions exhibit a similar behavior to the simulations, as the exchange contributions are virtually independent of each other, even for these concentrations that are well above those found naturally in vivo. Fitting the double dispersions to the model described in Equation (4) derives the individual contributions (full blue and red lines) to the dispersion. Comparing the fitted estimates with the experimental dispersions (broken red and blue lines) shows that this method predicts the contributions poorly in Fig. 9b, where the dispersions overlap, but works well in Fig. 9d, where the individual dispersions are much more distinguishable.
Figure 8.
(a) R1ρ dispersions simulated using three pools with 1% pool fractions of the exchanging pools, ΔΩb = 0.75 ppm, ΔΩc = 0.5 ppm, kba = 1 kHz and kca = 3 kHz. The red and blue curves are single dispersions (one exchange pool), the yellow curve is the addition of the two single dispersions and the black curve is the simulated double dispersion (two exchange pools). (b) The effect of exchange between minor pools on the double dispersion. (c) Similar simulations with pool c exchanging much faster at 30 kHz. (d) The corresponding simulations with the addition of minor exchange.
Figure 9.
(a) Experimentally measured R1ρ dispersions at 7 T for creatine, glucose and a mixture. The single dispersions were fitted to the Chopra model in Equation ((3)), whereas the double dispersions were fitted to the double Chopra model in Equation ((4)). (b) The full blue lines indicate the individual dispersions estimated from the fitting of the black double dispersion curve, whereas the broken lines are the experimental single dispersions. The estimated curves cannot be accurately estimated when the exchange rates are too close. (c) R1ρ dispersions at 7 T for creatine, uracil and a mixture. (d) The estimated single dispersions are much more accurate when the exchange rates are very different. PBS, phosphate-buffered saline.
Exchange rate contrast
Values of ERC were simulated for two exchanging pools, one with a constant pool fraction and the other increasing from 0% to 2% (Fig. 10). The double dispersions are shown in Fig. 10a, with only the low-frequency dispersion increasing as a result of the pool size change. The simulated ERC curves are depicted in Fig. 10b and exhibit a broadening and shifting of the peak as the low-frequency dispersion becomes more dominant. The low-frequency dispersion alone would produce an ERC peak at B1 = 254 Hz, depicted by the thick vertical black line in Fig. 10b. The ERC intensity then scales as a function of concentration at that locking field, as shown in Fig. 10c. These results were replicated experimentally using solutions of uracil and glucose, as shown in Fig. 11. The R1ρ map in Fig. 11a is shown at the locking field of B1 = 545 Hz, as this is roughly the frequency at which glucose displays its ERC peak. The corresponding double dispersions are shown in Fig. 11b and behave similarly to the simulations, as only the low-frequency dispersion increases with glucose concentration. Figure 11c shows the corresponding ERC image at glucose’s critical frequency of 545 Hz, and the mean image intensities calculated from region of interest (ROI) analyses are plotted in Fig. 11d, with the last point being that of 200 mm glucose with no uracil present. The experimental ERC intensity curve behaves very similarly to the simulated data.
Figure 10.

(a) Simulated R1ρ dispersions with three pools using various volume fractions of the slowly exchanging pool. (b) Exchange rate contrast (ERC) analysis of the previous double dispersions for various pool fractions with the full black line indicating the location of the peak in the case of no fast exchanging pool, which is the upper limit of the ERC shift. (c) The ERC values of each curve that intersect with the full back line in (b).
Figure 11.
(a) R1ρ map at 545 Hz for glucose and uracil samples of specified concentrations. (b) The entire R1ρ dispersion of each sample shown on the R1ρ map. (c) Exchange rate contrast (ERC) map at 545 Hz of the same samples; note the increased intensity of the 200 mm glucose sample as a result of the dependence on exchange rates, not relaxation rates. (d) Mean ERC values of each sample calculated by region of interest (ROI) analysis over each specific sample. PBS, phosphate-buffered saline.
Effects of diffusion
Diffusion effects on R1ρ dispersion were confirmed using modified Bloch simulations, wherein the chemical shift term (Δω0) included the susceptibility-induced ΔB0 in every voxel of a three-dimensional packed sphere structure. Spheres were assigned the volume magnetic susceptibility of polystyrene, −8.21 × 10−6, and were assumed to be impenetrable to simplify the system by excluding transmembrane diffusion. The local field shift induced by the spheres at each location was calculated from a linear superposition of single sphere perturbations described as follows (59):
| [11] |
Here, Δχ is the difference in susceptibility between the spheres and the surrounding medium, a is the radius of a sphere, is the distance away from the center of the sphere, θ = cos−1(z/r) is the azimuthal angle from the z axis and B0 is the static field strength. The finite difference simulations were incremented in time steps of 2 μs with a self-diffusion coefficient of D = 2.5 μm2/ms. Dispersions were fitted to the model in Equation (8) in a least-squares manner by solving for q2D, g2D, and a linear constant offset at high spin-lock amplitudes. The addition of chemical exchange to the system did, in fact, result in a simulated double dispersion with inflection frequencies determined by the timescales of the diffusion and exchange processes, as shown below.
Diffusion through susceptibility gradients induced by an ensemble of packed spheres gives rise to R1ρ dispersion as shown in Fig. 12a. The simulated dispersion magnitude increases with sphere radius and the inflection point shifts to lower frequencies, because the volume between the spheres increases, whereas the volume fraction stays constant. Both behaviors are consistent with theoretical expectations. Each dispersion curve was fitted to the model described in Equation (8) to estimate the correlation time that is shown to monotonically increase with sphere radius, which corresponds to a decrease in the spatial frequency spectral extent of the inhomogeneous field. Simulations were also performed with constant radius R = 5 μm and varying volume fraction. Changing the volume fraction generated smaller effects on the dispersion, but still modified the simulated curves in the expected manner. The decrease in inflection point is characteristic of the increase in space between spheres as the volume fraction decreases. Experiments have been performed previously on phantom samples of polystyrene in deionized water (21), and the resulting correlation time estimates were compared with the simulated correlation times in Fig. 12c. The correlation times agree well for small radii, but begin to diverge for larger radii, which may be a result of the packing structure of large spheres. It should be noted that the simulated results did not include radii below 3 μm because of the computation time necessary to carry out the simulations with these spatial resolutions.
Figure 12.
(a) Simulated R1ρ dispersions for an array of packed spheres for various radii. (b) The correlation time, calculated from the parameters of the fitted dispersion curves, increases with radius as there is more space between spheres. (c) Simulated correlation times are plotted together with the experimental correlation times estimated from the dispersions of polystyrene microspheres in pure water. The curves overlap well for small radii.
Effects of simultaneous exchange and diffusion
The effect of a system undergoing both chemical exchange and water diffusion in the presence of susceptibility gradients was simulated previously, where it was shown that these effects are linearly independent of each other (22). Bovine blood with varying levels of oxygen saturation provided a good model system to confirm these effects experimentally. Fresh whole bovine blood with sodium citrate anticoagulant (Lampire Biological Laboratories Pipersville, PA, USA) was used as a simple model system to validate these effects in vitro. Although the hematocrit (Hct) was not directly measured, nothing was done to alter the physiologic level, which should be ~40% based on previous studies. The blood was bubbled with pure O2 to oxygen saturations of 70, 73, 77, 85, 89 and 94%, as measured by a blood gas analyzer, and constantly rocked to hinder the precipitation of RBCs. The blood was then immediately placed in 5-mm NMR tubes and imaged at 7 T with a T1ρ-weighted fast spin-echo sequence using TR = 2 s, an echo train length of 4, a 64 × 64 voxel matrix and six logarithmically spaced spin-lock times from 10 to 100 ms, each measured at 10 logarithmically spaced spin-lock amplitudes ranging from 50 to 6000 Hz. The resulting double dispersions were fitted to the model in Equation (10) to estimate exchange and diffusion parameters simultaneously for each oxygen saturation level. The two components of the double dispersion fit were compared with the fitted dispersions from the single exchange pool measurements to demonstrate the accuracy of the fitting method.
The blood double dispersions calculated from R1ρ maps using Equation (10), shown in Fig. 13a, confirm that higher relaxation rates were measurable in the lower oxygen saturation samples because of the presence of more deoxyhemoglobin. The double dispersion phenomenon is apparent in these curves and the individual fitted contributions of diffusion and chemical exchange are plotted in Fig. 13b. It should be noted how the chemical exchange-based curves stay relatively constant, whereas the diffusion-based curves are greatly affected by oxygen saturation and diminish as the deoxyhemoglobin constituent in the blood decreases. The g2D parameter used in the fitting procedure represents the mean field offset magnitude for that dispersion, and is shown to decrease in the expected manner in Fig. 13c.
Figure 13.
(a) R1ρ double dispersions measured in whole bovine blood at 7 T for various oxygen saturation levels to vary the amount of deoxyhemoglobin present. (b) The individual dispersion components are plotted from the double dispersion fits in (a). Note how the exchange component stays relatively constant, whereas the low-frequency dispersion diminishes with higher oxygen saturation. (c) The gradient parameter, estimated from the dispersion fits, decreases with oxygen saturation.
DISCUSSION
Chemical exchange and water diffusion through internal susceptibility gradients may both be primary contributors to spin–lattice relaxation in the rotating frame at high fields, but previous studies have not often exploited the full potential of spin-locking methods to harness these mechanisms to characterize tissue quantitatively. Most in vivo studies have examined image intensities using T1ρ-weighted sequences or T1ρ maps only at a single locking field, usually at relatively low B0 field strengths (60–64). At higher field strengths, the dispersion of R1ρ with locking field amplitude provides much richer information on the dynamic properties of the system, allowing for a more comprehensive analysis of the parameters of exchange or intrinsic microstructure. Our studies of the effect of deuterium substitution show that chemical exchange between labile protons on solute molecules and water protons is the major determinant of the low locking field relaxation rate in solutions of glucose, as well as the change in rate with increasing field. Although deuteration slows the exchange process, and although dipolar interactions themselves are reduced by deuteration, the primary effect of deuteration is the dilution of proton-exchanging sites.
A potentially important conclusion from studies of the dispersion of tissues is that no single locking field is adequate to emphasize or quantify the presence of exchanging species. The low locking field values of T1ρ may reflect multiple contributions, whereas the chemical exchange contributions are substantially reduced at appropriate higher locking fields. Thus, it is essentially the difference in rates between low and higher locking fields that indicates the presence and magnitude of exchanging species, not the values at either one condition. This should be borne in mind when using spin-locking sequences to quantify, for example, the presence of a dominant exchangeable species, such as proteoglycans in cartilage (65).
Adequate knowledge of the complete dispersion curve enables the explicit derivation of exchange parameters according to the Chopra relationship. However, the location of the inflection point in the dispersion curve itself is sufficient to accurately estimate key intrinsic properties related to exchange. This inflection point varies monotonically with exchange rate, as shown for two different chemical shifts at multiple static field strengths in Fig. 14. For each chemical shift, the inflection point eventually begins to increase linearly with exchange rate at all fields and is linear at lower exchange rates for smaller chemical shifts. This relationship between the inflection point and the exchange rate is especially useful for faster exchanging metabolites possessing smaller chemical shifts, such as hydroxyls. The exchange rate has an explicit relationship to both temperature and pH, as shown in Figs 6b and 7b. Thus, in principle, changes in the inflection point can be used to detect changes in temperature and pH. Moreover, simply measuring the dispersion change from three R1ρ values, which is faster but more limited, may also be used to detect changes in temperature or pH, assuming that the concentration of exchanging species does not change. Figure 7a shows that the dispersion magnitude increases monotonically with exchange rate for a small range of pH values. Figure 15 shows the dependence of the ratio [R1ρ(0) – R1ρ(∞)]/[R1ρ(ω1) – R1ρ(∞)], which, according to Equation (3) can be simplified to . The curves in Fig. 15 assume Δωb = 1 ppm, , R2 = 5 Hz and B1 = 500 Hz. This ratio, which may be easily calculated from three R1ρ values, will have a direct relationship to the exchange rate and may be used for rapid estimations without explicit fitting, assuming that the chemical shift is known. The ratio shown in Fig. 15 depicts greater changes at lower fields, as the chemical shift dominates the equation at high fields. Figure 16b depicts how the dispersion magnitude changes versus the exchange rate for multiple field strengths. The magnitude becomes larger and the peak shifts to higher frequencies at higher fields, because the chemical shift term increases at higher fields.
Figure 14.
Inflection points of R1ρ dispersions are plotted against the exchange rate for various B0 field strengths with a chemical shift of ΔΩb = 1 ppm in (a) and ΔΩb = 3.5 ppm in (b).
Figure 15.
The ratio of [R1ρ(0) – R1ρ(∞)]/[R1ρ(Ω1) – R1ρ(∞)] versus the exchange rate at three static field strengths. The maximum value of the ratio decreases at higher fields because of the dominance of the increase in chemical shift frequency offset.
Figure 16.
(a) R1ρ dispersions of 200 mm glucose at 4.7 T over the pH range 6–7.4. Note that the magnitude of the pH 7.4 dispersion does not continue to increase relative to the pH 7.02 dispersion. (b) The dispersion magnitudes plotted against exchange rate at various B0 fields; note that the peak shifts to higher frequencies at higher fields.
Multi-pool R1ρ dispersion analyses can be performed for two chemically exchanging species or when chemical exchange and diffusion occur simultaneously. It should be noted that, although double dispersion curves are observed when there are two exchanging species, they are not expected for diffusion amongst mixtures of differently sized inhomogeneities. The double dispersion for mixtures of compounds with different chemical shifts reflects the discrete possibilities of frequencies of the exchanging species. By contrast, dephasing by diffusion corresponds to motion through a continuously varying field, and the addition of differently sized inhomogeneities merely changes the scales and magnitudes of gradient values to still form a smoothly varying field distribution. The point of inflection for diffusion- or exchange-based dispersions elucidates the timescale of the respective interactions. This is evident in the double dispersion chemical exchange simulations, where the slower exchange process produces an inflection at a much lower frequency than the faster exchange process. This is also the case in hybrid systems where both diffusion through internal gradients and chemical exchange occur. The process of water molecules diffusing through a medium typically occurs with D= 2.5 μm2/ms, and so gradients that repeat every 10 μm translate to a correlation time of ~1 ms, which is much longer than the fast exchange times seen with hydroxyls. One point of caution with the quantification of double dispersion data as a result of two chemically exchanging pools is that the amount of minor exchange occurring cannot be known from spin-locking measurements alone, and significant minor exchange can greatly influence the double dispersion fit. Figure 8b, d illustrates this point, as there is a significant shift in the dispersion for fast minor exchange for both cases, in addition to a change in the low spin-lock amplitude relaxation rates. Most systems in vivo will have small pool fractions and are believed to exhibit very little minor exchange, and these effects are typically ignored, but conceivable problems would occur in a special case in which these effects become significant.
ERC analysis can generate parametric images in which the intensity scales with the concentration of specific types of protons, those with a specific range of exchange rates. The value of this approach can be seen by comparing Fig. 11a, c. Figure 11a shows that the relaxation rate on its own is not specific for glucose when other solutes, such as uracil, are present. The ERC map in Fig. 11c, however, emphasizes compounds with a specific exchange rate, and so the intensity scales in this case with the concentration of glucose unaffected by uracil. This approach does not possess the same spectral selectivity as CEST experiments, but could be advantageous when compounds exist that have the same side groups, e.g. amides, but different exchange rates. Moreover, for fast exchanging hydroxyls, the problem of separating the resonance from water does not arise. The derived ERC image contrast surpasses the available contrast available from standard CEST magnetization transfer ratio asymmetry techniques for many scenarios, and has potential for in vivo studies with an injectable contrast agent of an exchanging species. This technique eliminates common experimental problems encountered with CEST, such as the possibility of direct water saturation whilst attempting to saturate protons with small chemical shifts, and asymmetric CEST spectra that confound accurate magnetization transfer ratio asymmetry calculations as a result of NOE effects (66–68). CEST experiments have been used to examine glucose metabolism in a similar manner to fluorodeoxyglucose positron emission tomography by injecting natural d-glucose or an analog, such as 2-deoxy-d-glucose, as an exogenous contrast agent to analyze the increase in hydroxyl exchange after cellular glucose uptake (69–74). We hypothesize that spin-locking experiments would provide more robust measurements of either glucose analog as they are more adept in quantifying fast hydroxyl exchange.
Bovine blood samples provided characteristic double dispersions, reflecting diffusion and chemical exchange processes occurring together. The contributions of diffusion and chemical exchange dispersions clearly illustrate that changes in oxygen saturation only affect the low-frequency interactions that are thought to be diffusion effects. Under the assumption the RBCs are packed, the mean correlation time for the diffusion-based dispersions was estimated to be τc ≈ 0.7 ms, which approximates a spatial distribution of ≈4 μm between the RBCs. Silvennoinen et al. (75) performed more elaborate whole bovine blood R1ρ dispersion experiments at 4.7 T, examining multiple oxygen saturation levels as well as multiple Hct levels and a lysate sample. These experiments showed similar increases in the low locking field limit, but the locking fields used by these authors only went down to 0.2 G (≈850 Hz), which is much higher than the lower limit of 50 Hz reached in the experiment described above. The higher frequencies are unable to accurately detect the double dispersion phenomenon described in this article. Their study concluded that exchange and diffusion effects both influence the dispersion, but also stated that the difference in mobility of hemoglobin in cells and free solution may play a critical role. This may be the case, but, for situations in which only the oxygen saturation changes, as described above, the high-frequency dispersion stays constant and the low-frequency dispersion is clearly governed by the oxygenation level and intrinsic susceptibility. Clearly, this approach can be used to estimate the characteristic dimensions of tissue microstructure related to cell sizes or vascular spacing, depending on what dominates the variation of intrinsic susceptibility.
CONCLUSIONS
Spin–lattice relaxation in the rotating frame provides a powerful approach to measure the timescale of subvoxel interactions in MRI which has, to date, been used relatively infrequently by the MR community. R1ρ measurements alone may be of some advantage over traditional R1 and R2 measurements in certain situations, but R1ρ dispersions yield much more information. Chemical exchange and the diffusion of water through microscopic susceptibility gradients are the main effects influencing R1ρ at higher fields, and these may provide quantitative characteristics of the medium of interest. Exchange has been shown to become the dominant factor influencing R1ρ relaxation at high fields, and exchange rates, for example, can be used to estimate variations in temperature or pH under certain conditions. Double dispersion analysis may be used to estimate the concentrations of a specific metabolite in special cases of solute mixtures. ERC also provides a powerful means to produce a new image contrast that emphasizes specific exchanging metabolites based on their exchange rate. Finally, diffusion effects can provide information about the average spacing of cells or microvessels, for example, in otherwise homogeneous tissue, through the estimation of the diffusion correlation time τc. Diffusion and chemical exchange effects are independent of each other and can both be present in the same system. With the field strength of MRI magnets used both clinically and in research continuing to increase, the effects described in this article will probably become even more relevant.
Acknowledgement
The National Science Foundation (NSF) provided grant support (DGE-0909667).
Abbreviations used:
- BOLD
blood oxygenation level-dependent
- CEST
chemical exchange saturation transfer
- CPMG
Carr–Purcell–Meiboom–Gill
- ERC
exchange rate contrast
- EWIC
exchange-weighted image contrast
- fMRI
functional MRI
- Hct
hematocrit
- NOE
nuclear Overhauser effect
- PBS
phosphate-buffered saline
- RBC
red blood cell
- RF
radiofrequency
- ROI
region of interest
REFERENCES
- 1.Witschey W, Borthakur A, Fenty M, Kneeland B, Lonner J, McArdle E, Sochor M, Reddy R. T1ρ MRI quantification of arthroscopically confirmed cartilage degeneration. Magn. Reson. Med 2010; 63: 1376–1382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Borthakur A, Sochor M, Davatzikos C, Trojanowski J, Clark C. T1ρ MRI of Alzheimer’s disease. Neuroimage 2008; 41: 1199–1205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Duvvuri U, Poptani H, Feldman M, Nadal-Desbarats L, Gee M, Lee W, Reddy R, Leigh J, Glickson J. Quantitative T1ρ magnetic resonance imaging of RIF-1 tumors in vivo: detection of early response to cyclophosphamide therapy. Cancer Res. 2001; 61: 7747–7753. [PubMed] [Google Scholar]
- 4.Hulvershorn J, Borthakur A, Bloy L, Gualtieri E, Reddy R, Leigh J, Elliot M. T1ρ contrast in functional magnetic resonance imaging. Magn. Reson. Med 2005; 54: 1155–1162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rane S, Spear J, Zu Z, Donahue M, Gore J. Functional MRI using spin lock editing preparation pulses. Magn. Reson. Imaging 2014; 32: 813–818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Redfield A Nuclear magnetic resonance saturation and rotary saturation in solids. Phys. Rev 1955; 98: 1787–1809. [Google Scholar]
- 7.Abragam A Principles of Nuclear Magnetism. Oxford University Press, New York; 1961. [Google Scholar]
- 8.Davis D, Perlman M, London R. Direct measurements of the dissociation-rate constant for inhibitor–enzyme complexes via the T1ρ and T2 (CPMG) methods. J. Magn. Reson. Ser. B 1994; 104: 266–275. [DOI] [PubMed] [Google Scholar]
- 9.Korzhnev D, Billeter M, Arseniev A, Orekhov V. NMR studies of Brownian tumbling and internal motions in proteins. Prog. Nucl. Magn. Reson. Spectrosc 2001; 38: 197–266. [Google Scholar]
- 10.Virta A, Komu M, Kormano M. T1ρ of protein solutions at very low fields: dependence on molecular weight, concentration, and structure. Magn. Reson. Med 1997; 37: 53–57. [DOI] [PubMed] [Google Scholar]
- 11.Koskinen S, Niemi P, Kajander S, Komu M. T1ρ dispersion profile of rat tissues in vitro at very low locking fields. Magn. Reson. Imaging 2006; 24: 295–299. [DOI] [PubMed] [Google Scholar]
- 12.Borthakur A, Wheaton A, Gougoutas A, Akella S, Regatte R, Charagundla S, Reddy R. In vivo measurement of T1ρ dispersion in the human brain at 1.5 tesla. J. Magn. Reson. Imaging 2004; 19: 403–409. [DOI] [PubMed] [Google Scholar]
- 13.Haris M, McArdle E, Fenty M, Singh A, Davatzikos C, Trojanowski J, Melhem E, Clark C, Borthakur A. Early marker for Alzheimer’s disease: hippocampus T1ρ estimation. J. Magn. Reson. Imaging 2009; 29: 1008–1012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bain A Chemical exchange in NMR. Prog. Nucl. Magn. Reson. Spectrosc 2003; 43: 63–103. [Google Scholar]
- 15.Michaeli S, Sorce D, Idiyatullin D, Ugurbil K, Garwood M. Transverse relaxation in the rotating frame induced by chemical exchange. J. Magn. Reson 2004; 169: 293–299. [DOI] [PubMed] [Google Scholar]
- 16.Michaeli S, Sorce D, Springer C, Ugurbil K, Garwood M. T1ρ MRI contrast in the human brain: modulation of the longitudinal rotating frame relaxation shutter-speed during an adiabatic RF pulse. J. Magn. Reson 2006; 181: 135–147. [DOI] [PubMed] [Google Scholar]
- 17.Mäkelä H, De Vita E, Gröhn O, Kettunen M, Kavec M, Lythgoe M, Garwood M, Ordidge R, Kauppinen R. B0 dependence of the on-resonance longitudinal relaxation time in the rotating frame (T1ρ) in protein phantoms and rat brain in vivo. Magn. Reson. Med 2004; 51: 4–8. [DOI] [PubMed] [Google Scholar]
- 18.Cobb J, Xie J, Li K, Gochberg D, Gore J. Exchange-mediated contrast agents for spin-lock imaging. Magn. Reson. Med 2012; 67: 1427–1433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hills B, Wright K, Belton P. Proton N.M.R. studies of chemical and diffusive exchange in carbohydrate systems. Mol. Phys 1989; 67: 1309–1326. [Google Scholar]
- 20.Hills B, Duce S. The influence of chemical and diffusive exchange on water proton transverse relaxation in plant tissues. Magn. Reson. Imaging 1990; 8: 321–331. [DOI] [PubMed] [Google Scholar]
- 21.Spear J, Zu Z, Gore J. Dispersion of relaxation rates in the rotating frame under the action of spin-locking pulses and diffusion in inhomogeneous magnetic fields. Magn. Reson. Med 2014; 71: 1906–1911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Spear J, Gore J. Effects of diffusion in magnetically inhomogeneous media on rotating frame spin–lattice relaxation. J. Magn. Reson 2014; 249: 80–87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kettunen M, Gröhn O, Penttonen M, Kauppinen R. Cerebral T1ρ relaxation time increases immediately upon global ischemia in the rat independently of blood glucose and anoxic depolarization. Magn. Reson. Med 2001; 46: 565–572. [DOI] [PubMed] [Google Scholar]
- 24.Gröhn O, Kettunen M, Mäkelä H, Penttonen A, Pitkänen, Lukkarinen J, Kauppinen R. Early detection of irreversible cerebral ischemia in the rat using dispersion of the magnetic resonance imaging relaxation time, T1ρ. J. Cereb. Blood Flow Metab. 2000; 20: 1457–1466. [DOI] [PubMed] [Google Scholar]
- 25.Hakumäki J, Gröhn O, Tyynelä K, Valonen P, Ylä-Herttuala S, Kauppinen R. Early gene therapy-induced apoptotic response in BT4C gliomas by magnetic resonance relaxation contrast T1 in the rotating frame. Cancer Gene Ther. 2002; 9: 338–345. [DOI] [PubMed] [Google Scholar]
- 26.Sierra A, Michaeli S, Niskanen J, Valonen P, Gröhn H, Ylä-Herttuala S, Garwood M, Gröhn O. Water spin dynamics during apoptotic cell death in glioma gene therapy probed by T1ρ and T2ρ. Magn. Reson. Med 2008; 59: 1311–1319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Jin T, Wang P, Zong X, Kim S. Magnetic resonance imaging of the Amine–Proton EXchange (APEX) dependent contrast. Neuroimage 2012; 59: 1218–1227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Jin T, Kim S. Characterization of non-hemodynamic functional signal measured by spin-lock fMRI. Neuroimage 2013; 78C: 385–395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Haris M, Singh A, Cai K, McArdle E, Fenty M, Davatzikos C, Trojanowski J, Melhem E, Clark C, Borthakur A. T1ρ MRI in Alzheimer’s disease: detection of pathological changes in medial temporal lobe. J. Neuroimaging 2011; 21: e86–e90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Borthakur A, Gur T, Wheaton A, Corbo M, Trojanowski J, Lee V, Reddy R. In vivo measurement of plaque burden in a mouse model of Alzheimer’s disease. J. Magn. Reson. Imaging 2006; 24: 1011–1017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Jazrawi L, Alaia M, Chang G, Fitzgerald E, Recht M. Advances in magnetic resonance imaging of articular cartilage. J. Am. Acad. Orthop. Surg 2011; 19: 420–429. [DOI] [PubMed] [Google Scholar]
- 32.Duvvuri U, Reddy R, Patel S, Kaufman J, Kneeland J, Leigh J. T1ρ-relaxation in articular cartilage: effects of enzymatic degradation. Magn. Reson. Med 1997; 38: 863–867. [DOI] [PubMed] [Google Scholar]
- 33.Cobb J, Kan J, Gore J. T1ρ mapping of pediatric epiphyseal and articular cartilage in the knee. J. Magn. Reson. Imaging 2013; 38: 299–305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Duvvuri U, Goldberg A, Kranz J, Hoang L, Reddy R, Wehrli F, Wand A, Englander S, Leigh J. Water magnetic relaxation dispersion in biological systems: the contribution of proton exchange and implications for the noninvasive detection of cartilage degradation. Proc. Natl. Acad. Sci 2001; 98: 12 479–12 484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Jin T, Autio J, Obata T, Kim S. Spin-locking versus chemical exchange saturation transfer MRI for investigating chemical exchange process between water and labile metabolite protons. Magn. Reson. Med 2011; 65: 1448–1460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kettunen M, Gröhn O, Silvennoinen M, Penttonen M, Kauppinen R. Effects of intracellular pH, blood, and tissue oxygen tension on T1ρ relaxation in rat brain. Magn. Reson. Med 2002; 48: 470–477. [DOI] [PubMed] [Google Scholar]
- 37.Zaiss M, Bachert P. Exchange-dependent relaxation in the rotating frame for slow and intermediate exchange – modeling off-resonant spin-lock and chemical exchange saturation transfer. NMR Biomed. 2013; 26: 507–518. [DOI] [PubMed] [Google Scholar]
- 38.van Zijl P, Yadav N. Chemical exchange saturation transfer (CEST): what is in a name and what isn’t? Magn. Reson. Med 2011; 65: 927–948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Cobb J, Li K, Xie J, Gochberg D, Gore J. Exchange-mediated contrast in CEST and spin-lock imaging. Magn. Reson. Imaging 2013; 32: 28–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.McConnell H Reaction rates by nuclear magnetic resonance. J. Chem. Phys 1958; 28: 430–431. [Google Scholar]
- 41.Trott O, Palmer A. R1ρ relaxation outside of the fast-exchange limit. J. Magn. Reson 2002; 154: 157–160. [DOI] [PubMed] [Google Scholar]
- 42.Chopra S, McClung R, Jordan R. Rotating-frame relaxation rates of solvent molecules in solutions of paramagnetic ions undergoing solvent exchange. J. Magn. Reson 1984; 59: 361–372. [Google Scholar]
- 43.Cobb J, Xie J, Gore J. Contributions of chemical exchange to T1ρ dispersion in a tissue model. Magn. Reson. Med 2011; 66: 1563–1571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Trott O, Palmer A. Theoretical study of R1ρ rotating-frame and R2 free-precession relaxation in the presence of n-site chemical exchange. J. Magn. Reson 2004; 170: 104–112. [DOI] [PubMed] [Google Scholar]
- 45.Deverell C, Morgan R, Strange J. Studies of chemical exchange by nuclear magnetic relaxation in the rotating frame. Mol. Phys 1970; 18: 553–559. [Google Scholar]
- 46.Jensen J, Chandra R. NMR relaxation in tissues with weak magnetic inhomogeneities. Magn. Reson. Med 2000; 44: 144–156. [PubMed] [Google Scholar]
- 47.Majumdar S, Gore J. Studies of diffusion in random fields produced by variations in susceptibility. J. Magn. Reson 1988; 78: 41–55. [Google Scholar]
- 48.Kiselev V Effect of magnetic field gradients induced by microvasculature on NMR measurements of molecular self-diffusion in biological tissues. J. Magn. Reson 2004; 170: 228–235. [DOI] [PubMed] [Google Scholar]
- 49.Audoly B, Sen P, Ryu S, Song Y. Correlation functions for inhomogeneous magnetic field in random media with application to a dense random pack of spheres. J. Magn. Reson 2003; 164: 154–159. [DOI] [PubMed] [Google Scholar]
- 50.Van Vleck J The dipolar broadening of magnetic resonance lines in crystals. Phys. Rev 1948; 74: 1168–1183. [Google Scholar]
- 51.Gore J, Leeman S. Transfer functions for describing ultrasound system performance. Phys. Med. Biol 1979; 24: 146–156. [DOI] [PubMed] [Google Scholar]
- 52.Cobb J, Xie J, Gore J. Contributions of chemical and diffusive exchange to T1ρ dispersion. Magn. Reson. Med 2013; 69: 1357–1366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Witschey W, Borthakur A, Elliot M, Mellon E, Niyogi S, Wallman D, Wang C, Reddy R. Artifacts in T1ρ-weighted imaging: compensation for B1 and B0 field imperfections. J. Magn. Reson 2007; 186: 75–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Anderson W, Arnold J. Proton relaxation times in H2O—D2O mixtures. Phys. Rev 1956; 101: 511–512. [Google Scholar]
- 55.Northrop D Steady-state analysis of kinetic isotope effects in enzymic reactions. Biochemistry 1975; 14: 2644–2651. [DOI] [PubMed] [Google Scholar]
- 56.Rizi R, Charagundla S, Song H, Reddy R, Stolpen A, Schnall M, Leigh J. Proton T1ρ-dispersion imaging of rodent brain at 1.9 T. J. Magn. Reson. Imaging 1998; 8: 1090–1096. [DOI] [PubMed] [Google Scholar]
- 57.Sood R Off-resonance binomial preparatory pulse technique for high sensitivity MRI of H2O17. Magn. Reson. Imaging 2004; 22: 181–195. [DOI] [PubMed] [Google Scholar]
- 58.McCommis K, He X, Abendschein D, Gupte P, Gropler R, Zheng J. Cardiac 17O MRI: toward direct quantification of myocardial oxygen consumption. Magn. Reson. Med 2010; 63: 1442–1447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Haacke E, Brown R, Thompson M, Venkatesan R. Magnetic Resonance Imaging: Physical Principles and Sequence Design. John Wiley & Sons, Hoboken, NJ; 1999. [Google Scholar]
- 60.Poptani H, Duvvuri U, Miller C, Mancuso A, Charagundla S, Fraser N, Glickson J, Leigh J, Reddy R. T1ρ imaging of murine brain tumors at 4 T. Acad. Radiol 2001; 8: 42–47. [DOI] [PubMed] [Google Scholar]
- 61.Wang L, Chang G, Xu J, Vieira R, Krasnokutsky S, Abramson S, Regatte R. T1ρ MRI of menisci and cartilage in patients with osteoarthritis at 3T. Eur. J. Radiol 2012; 81: 2329–2336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Wang Y, Zhao F, Griffith J, Mok G, Leung J, Ahuja A, Yuan J. T1ρ and T2 relaxation times for lumbar disc degeneration: an in vivo comparative study at 3.0-Tesla MRI. Eur. Radiol 2013; 23: 228–234. [DOI] [PubMed] [Google Scholar]
- 63.Watts R, Andrews T, Hipko S, Gonyea J, Filippi C. In vivo whole-brain T1ρ mapping across adulthood: normative values and age dependence. J. Magn. Reson. Imaging 2014; 40: 376–382. [DOI] [PubMed] [Google Scholar]
- 64.Mulkern R, Patz S, Brooks M, Metcalf D, Jolesz F. Spin-lock techniques and CPMG imaging sequences: a critical appraisal of T1p contrast at 0.15 T. Magn. Reson. Imaging 1989; 7: 437–444. [DOI] [PubMed] [Google Scholar]
- 65.Wang P, Block J, Gore J. Chemical exchange in knee cartilage assessed by R1ρ (1/T1ρ) dispersion at 3T. Magn. Reson. Imaging 2015; 33: 38–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Jones C, Huang A, Xu J, Edden A, Schär M, Hua J, Oskolkov N, Zacà D, Zhou J, McMahon M, Pillai J, van Zijl P. Nuclear Overhauser enhancement (NOE) imaging in the human brain at 7T. Neuroimage 2013; 77: 114–124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Jin T, Wang P, Zong X, Kim S. MR imaging of the amide-proton transfer effect and the pH-insensitive nuclear Overhauser effect at 9.4 T. Magn. Reson. Med 2013; 69: 760–770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Zaiss M, Kunz P, Goerke S, Radbruch A, Bachert P. MR imaging of protein folding in vitro employing nuclear-Overhauser-mediated saturation transfer. NMR Biomed. 2013; 26: 1815–1822. [DOI] [PubMed] [Google Scholar]
- 69.Walker-Samuel S, Johnson P, Pedley B, Lythgoe M, Golay X. Proceedings of the 19th Annual Meeting ISMRM, Montreal, QC, Canada, 2011; 962. [Google Scholar]
- 70.Chan K, McMahon M, Liu G, Yoshinori K, Bhujwalla Z, Artemov D, van Zijl P. Proceedings of the 19th Annual Meeting ISMRM, Montreal, QC, Canada, 2011; 551. [Google Scholar]
- 71.Walker-Samuel S, Ramasawmy S, Torrealdea F, Rega M, Johnson P, Rajkumar V, Richardson S, Thomas D, Pedley B, Lythgoe M, Golay X. Proceedings of the 20th Annual Meeting ISMRM, Melbourne, Vic., Australia, 2012; 182. [Google Scholar]
- 72.Walker-Samuel S, Ramasawmy R, Torrealdea F, Rega M, Rajkumar V, Johnson S, Richardson S, Gonçalves M, Parkes H, Årstad E, Thomas D, Pedley B, Lythgoe M, Golay X. In vivo imaging of glucose uptake and metabolism in tumors. Nat. Med 2013; 19: 1067–1072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Chan K, McMahon M, Kato Y, Liu G, Bulte J, Bhujwalla Z, Artemov D, van Zijl P. Natural D-glucose as a biodegradable MRI contrast agent for detecting cancer. Magn. Reson. Med 2012; 68: 1764–1773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Nasrallah F, Pagès G, Kuchel P, Golay X, Chuang K. Imaging brain deoxyglucose uptake and metabolism by glucoCEST MRI. J. Cereb. Blood Flow Metab. 2013; 33: 1270–1278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Silvennoinen M, Kettunen M, Clingman C, Kauppinen R. Blood NMR relaxation in the rotating frame: mechanistic implications. Arch. Biochem. Biophys 2002; 405: 78–86. [DOI] [PubMed] [Google Scholar]