Abstract
Background
Severe aortic valve stenosis (AS) is a major cause of morbidity and mortality worldwide. The definitive management for severe AS is aortic valve replacement (AVR). The choice of transcatheter approach versus open‐heart surgery for AVR in people with severe AS and low surgical risk remains a matter of debate.
Objectives
To assess the benefits and harms of transcatheter aortic valve implantation (TAVI) compared to surgical aortic valve replacement (SAVR) in people with severe AS and low surgical risk.
Search methods
We searched the following databases for randomised controlled trials (RCTs) on 29 April 2019: Cochrane Central Register of Controlled Trials (CENTRAL), MEDLINE, Embase, and Web of Science Core Collection. We also searched ClinicalTrials.gov and the World Health Organization International Clinical Trials Registry Platform. We searched all databases from inception to present and imposed no restriction on language or date of publication.
Selection criteria
We included RCTs that compared TAVI and SAVR in adults (18 years of age or older) with severe AS and low surgical risk.
Data collection and analysis
We used the standard methodological procedures expected by Cochrane. Two authors independently screened titles and abstracts for inclusion, performed data extraction, and assessed risk of bias in the studies included. We analysed dichotomous data using the risk ratio (RR) and continuous data using the mean difference (MD), with respective 95% confidence intervals (CI). We assessed the certainty of evidence for each outcome using the GRADE approach. Our outcomes of interest were assessed in the short term (i.e. during hospitalisation and up to 30 days of follow‐up). Primary outcomes were all‐cause mortality, stroke, and rehospitalisation. Secondary outcomes were myocardial infarction (MI), cardiac death, length of hospital stay (LOS), permanent pacemaker (PPM) implantation, new‐onset atrial fibrillation, acute kidney injury (AKI), and any bleeding.
Main results
We identified four studies (13 reports), with 2818 participants, and one ongoing study. Overall certainty of evidence ranged from high to very low.
There is probably little or no difference between TAVI and SAVR for the following short‐term outcomes: all‐cause mortality (RR 0.69, 95% CI 0.33 to 1.44; SAVR 11 deaths per 1000, TAVI 8 deaths per 1000 (95% CI 4 to 16); 2818 participants; 4 studies; moderate‐certainty evidence); stroke (RR 0.73, 95% CI 0.42 to 1.25; SAVR 21 strokes per 1000, TAVI 16 strokes per 1000 (95% CI 9 to 27); 2818 participants; 4 studies; moderate‐certainty evidence); MI (RR 0.82, 95% CI 0.42 to 1.58; SAVR 14 MI per 1000, TAVI 11 MI per 1000 (95% CI 6 to 21); 2748 participants; 3 studies; moderate‐certainty evidence); and cardiac death (RR 0.71, 95% CI 0.32 to 1.56; SAVR 10 cardiac deaths per 1000, TAVI 7 cardiac deaths per 1000 (95% CI 3 to 16); 2818 participants; 4 studies; moderate‐certainty evidence).
TAVI may reduce the risk of short‐term rehospitalisation, although the confidence interval also includes the possibility of no difference in risk between groups (RR 0.64, 95% CI 0.39 to 1.06; SAVR 30 cases per 1000, TAVI 19 cases per 1000 (95% CI 12 to 32); 2468 participants; 2 studies; low‐certainty evidence).
TAVI, compared with SAVR, probably increases the risk of PPM implantation (RR 3.65, 95% CI 1.50 to 8.87; SAVR 47 per 1000, TAVI 170 cases per 1000 (95% CI 70 to 413); number needed to treat for an additional harmful outcome (NNTH) = 7; 2683 participants; 3 studies; moderate‐certainty evidence). We are uncertain whether TAVI, compared with SAVR, affects the LOS in days, although it appears to be associated with shorter LOS.
TAVI, compared with SAVR, reduces the risk of atrial fibrillation (RR 0.21, 95% CI 0.15 to 0.30; 2683 participants; 3 studies), AKI (RR 0.30, 95% CI 0.16 to 0.58; 2753 participants; 4 studies), and bleeding (RR 0.31, 95% CI 0.16 to 0.62; 2753 participants; 4 studies) (all high‐certainty evidence).
Authors' conclusions
Our meta‐analysis indicates that, in the short term, TAVI probably has little or no mortality difference compared to SAVR for severe AS in individuals with low surgical risk. Similarly, there is probably little or no difference in risk of stroke, MI, and cardiac death between the two approaches. TAVI may reduce the risk of rehospitalisation, but we are uncertain about the effects on LOS. TAVI reduces the risk of atrial fibrillation, AKI, and bleeding. However, this benefit is offset by the increased risk of PPM implantation. Long‐term follow‐up data are needed to further assess and validate these outcomes, especially durability, in the low surgical risk population.
Keywords: Humans, Transcatheter Aortic Valve Replacement, Transcatheter Aortic Valve Replacement/adverse effects, Aortic Valve, Aortic Valve/surgery, Aortic Valve Stenosis, Aortic Valve Stenosis/mortality, Aortic Valve Stenosis/surgery, Postoperative Complications, Postoperative Complications/epidemiology, Randomized Controlled Trials as Topic, Risk Factors, Treatment Outcome
Plain language summary
Transcatheter aortic valve implantation versus surgical aortic valve replacement in people with severe aortic stenosis and low surgical risk
Review question
Does transcatheter approach, compared to open‐heart surgery, for aortic valve replacement improve outcomes in people with severe aortic stenosis and low surgical risk?
Background
Aortic stenosis (AS) is the narrowing of the exit of the left ventricle of the heart (where the aorta begins). It typically gets worse over time. Its severity can be divided into mild, moderate, severe, and very severe; and it is distinguishable by ultrasound scan of the heart and other clinical features. Once it has become severe, treatment primarily involves surgery to replace the valve. An alternative approach is to use transcatheter aortic valve implantation (TAVI). This approach improves outcomes in individuals who are inoperable or at a high to intermediate risk for surgery. However, it remains unclear if TAVI is beneficial to those who have severe AS and are at a low surgical risk.
Search Date
The evidence is current to April 2019. We searched the literature and found a total of 3092 citations that were potentially relevant. After reviewing each of these, we found 13 published articles describing four clinical trials that could help us answer our question.
Study characteristics
The four clinical trials included 2818 participants who were randomly allocated to undergo either TAVI or surgical aortic valve replacement (SAVR). The trials were multicentre and took place in Australia, Canada, France, Japan, the Netherlands, New Zealand, the USA, Denmark, and Sweden.
Key Results
Moderate‐certainty evidence from clinical trials shows that, in the short term (i.e. during hospitalisation and up to 30 days of follow‐up), there is probably little or no difference between TAVI compared with SAVR in risk of death due to any cause, stroke (insult to the brain), myocardial infarction (injury or death of heart muscle), or death due to cardiac causes (e.g. myocardial infarction or failure of the heart muscle pump). Low‐certainty evidence shows that TAVI may reduce the risk of rehospitalisation compared with SAVR. We are uncertain whether TAVI, compared with SAVR, affects the length of hospital stay, although it appears to be associated with shorter duration of hospitalisation. High‐certainty evidence shows that fewer people had atrial fibrillation (a type of irregular heart rhythm), acute kidney injury (insult to the kidney), and bleeding when they underwent TAVI, compared with SAVR. However, moderate‐certainty evidence shows that TAVI probably increases the risk of permanent pacemaker implantation (a device that is placed to artificially set the heart rhythm), compared with SAVR.
Quality of the evidence
We consider the overall quality of evidence to be moderate for most relevant outcomes (death, stroke, myocardial infarction, cardiac death, and risk of permanent pacemaker implantation), with the exception of rehospitalisation (low‐quality evidence) and length of hospital stay (very low quality evidence). The evidence for atrial fibrillation, acute kidney injury, and bleeding was of high quality.
Summary of findings
Summary of findings for the main comparison. Transcatheter aortic valve implantation compared to surgical aortic valve replacement for severe aortic stenosis in people with low surgical risk.
| TAVI compared to SAVR in people with severe AS and low surgical risk at short‐term follow‐up (up to 30 days) | ||||||
| Patient or population: adults with severe aortic stenosis who are at a low surgical risk. Setting: inpatient: Australia, Canada, France, Japan, the Netherlands, New Zealand, the USA, Denmark, and Sweden. Intervention: transcatheter aortic valve implantation. Comparison: surgical aortic valve replacement. | ||||||
| Outcomes (up to 30 days' follow‐up) | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Certainty of the evidence (GRADE) | Comments | |
| Risk with surgical aortic valve replacement (SAVR) | Risk with transcatheter aortic valve implantation (TAVI) | |||||
| All‐cause mortality | 11 per 1000 | 8 per 1000 (4 to 16) | RR 0.69 (0.33 to 1.44) | 2818 (4 RCTs) | ⊕⊕⊕⊝ MODERATE 1 | |
| Stroke | 21 per 1000 | 16 per 1000 (9 to 27) | RR 0.73 (0.42 to 1.25) | 2818 (4 RCTs) | ⊕⊕⊕⊝ MODERATE 1 | |
| Rehospitalisation | 30 per 1000 | 19 per 1000 (12 to 32) | RR 0.64 (0.39 to 1.06) | 2468 (2 RCTs) | ⊕⊕⊝⊝ LOW 1 2 | |
| Myocardial infarction | 14 per 1000 | 11 per 1000 (6 to 21) | RR 0.82 (0.42 to 1.58) | 2748 (3 RCTs) |
⊕⊕⊕⊝ MODERATE 1 | 1 additional trial (STACCATO 2012) had zero events in both arms. |
| Cardiac death | 10 per 1000 | 7 per 1000 (3 to 16) | RR 0.71 (0.32 to 1.56) | 2818 (4 RCTs) | ⊕⊕⊕⊝ MODERATE 1 | |
| Length of hospital stay | ‐ | ‐ | ‐ | ‐ | ⊕⊝⊝⊝ VERY LOW 2 3 4 5 | We did not pool the results due to the considerable heterogeneity. PARTNER 3 2019 reported shorter median LOS in days (interquartile range) in the TAVI group (TAVI 3 (2 to 3) days versus SAVR 7 (6 to 8) days, MD −4 (−4 to −3), P < 0.001). NOTION 2015 similarly reported a shorter mean LOS ± standard deviation (SD) with TAVI (TAVI 8.9 ± 6.2 days versus SAVR 12.9 ± 11.6 days, MD −4 (−6.2 to −1.8, P < 0.001). STACCATO 2012, on the other hand, reported a longer mean LOS after TAVI (TAVI 8.8 ± 6.7 days versus 7.6 ± 2.4 days, MD 1.2 (−1.18 to 3.58), P > 0.05). |
| Permanent pacemaker implantation | 47 per 1000 | 170 per 1000 (70 to 413) | RR 3.65 (1.50 to 8.87) | 2683 (3 RCTs) | ⊕⊕⊕⊝ MODERATE 5 | |
| All outcomes reported in the 'Summary of findings' table are short term (i.e. assessed during hospitalisation and up to 30 days of follow‐up). *The risk in the intervention group is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: Confidence interval; LOS: length of hospital stay; RR: Risk ratio; MD: Mean difference. | ||||||
| GRADE Working Group grades of evidence High certainty: we are very confident that the true effect lies close to that of the estimate of the effect. Moderate certainty: we are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low certainty: our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect. Very low certainty: we have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect. | ||||||
1 Confidence interval includes the null effect and appreciable benefit. Downgraded 1 level for concerns about imprecision.
2 High risk of detection bias due to lack of masking for all included trials. Downgraded 1 level for concerns about study design limitations (risk of bias).
3 Point estimates vary widely across studies and confidence intervals show no overlap. Downgraded 1 level for concerns about inconsistency.
4 Does not meet the optimal information size criterion (Schünemann 2013). Downgraded 1 level for concerns about imprecision.
5 Considerable unexplained heterogeneity. Downgraded 1 level for concerns about inconsistency.
Background
Description of the condition
Aortic valve stenosis (AS) is the most common heart valve disease and is a major cause of morbidity and mortality worldwide (Bhatia 2016; Maganti 2010). The aortic valve (AV) lies between the left ventricle and aorta, the major systemic blood vessel that provides blood supply to all body organs and tissues (Piazza 2008). Obstruction of the left ventricular outflow tract (LVOT), with ensuing left ventricular hypertrophy, eventually results in distressing symptoms including exertional dyspnoea, chest pain, and possible syncope (Grimard 2016). In addition to the magnitude of these clinical symptoms, the severity of the condition is determined by a number of echocardiographic parameters, including AS jet velocity, mean transvalvular pressure gradient, and AV area by continuity equation (Baumgartner 2017a).
Description of the intervention
The definitive management for severe AS is aortic valve replacement (AVR). This entails replacing the diseased heart valve with a new, functional valve, which may be made of mechanical or bioprosthetic material (Hirji 2018a; Leon 2010). Surgical AVR (SAVR) has long been the standard of care for severe, symptomatic AS, supported by current US and European guidelines (Baumgartner 2017b; Nishimura 2017). It has been shown to significantly improve symptoms and prolong survival (Leon 2010). Transcatheter aortic valve implantation (TAVI), also known as transcatheter aortic valve replacement, has recently emerged as a less invasive approach to AVR. Both procedures are utilised to achieve adequate haemodynamic parameters and relief of symptoms with improved survival (Hirji 2017).
How the intervention might work
Surgical approaches to AVR include a full sternotomy or minimally invasive surgical incisions, which have shown comparable outcomes (Hirji 2018b). The standard approach for TAVI is via the femoral artery (Grover 2017). However, in certain populations, such as people with severe peripheral arterial disease, alternative access sites are employed. These include the trans‐subclavian artery and, less commonly, transcarotid or transcaval approaches (Greenbaum 2017; Kolkailah 2018; Mylotte 2016). The benefit of such approaches is that they provide access in a less invasive manner, without having to open the chest cavity, which makes them an appealing choice in elderly, frail patients with a high surgical risk (Kolkailah 2018). Other common alternative access sites include the transapical and direct transaortic approaches (Thourani 2015). There is a growing momentum towards less invasive approaches, with SAVR now being put to the test in comparison to TAVI in different populations (Hirji 2018b; Leon 2010; Leon 2016).
Why it is important to do this review
TAVI has been established as an alternative to SAVR in people with severe AS who are deemed inoperable or at a high surgical risk (Leon 2010). More recently, TAVI indications have expanded to include people with severe AS and an intermediate surgical risk (Leon 2016). However, the choice of transcatheter approach versus open‐heart surgery for AVR in people with severe AS and low surgical risk remains a matter of debate (Witberg 2018).
Objectives
To assess the benefits and harms of TAVI compared to SAVR in people with severe AS and low surgical risk.
Methods
Criteria for considering studies for this review
Types of studies
We included all published, unpublished, and ongoing randomised controlled trials (RCTs). We did not identify cluster RCTs.
Types of participants
Adults (18 years of age or older) of both sexes with severe AS and low surgical risk undergoing AVR. We defined low surgical risk as the Society of Thoracic Surgery (STS) risk score — O'Brien 2009 — or European System for Cardiac Operative Risk Evaluation (EuroSCORE) II — Nashef 2012 — of less than 4%. This definition cut‐off is based on the most up‐to‐date European Society of Cardiology, European Association for Cardio‐Thoracic Surgery, American College of Cardiology, and American Heart Association guidelines on management of valvular heart disease (Baumgartner 2017b; Nishimura 2017).
In case of studies with mixed populations, and only a subset of the participants meeting our inclusion criteria, we attempted to obtain data for the subgroup of interest from the trialists in order to include the study. See Dealing with missing data for more details.
Types of interventions
TAVI versus SAVR for severe AS.
Types of outcome measures
Reporting one or more of the outcomes listed here in the trial was not an inclusion criterion of the review. Where a published report did not appear to report one of these outcomes, we accessed the trial protocol — if available — and contacted the trial authors to ascertain whether the outcomes were measured but not reported. This is described under the notes field for each trial in Characteristics of included studies.
Primary outcomes
All‐cause mortality
Stroke
Rehospitalisation (number of participants)
Secondary outcomes
Myocardial infarction
Cardiac death
Length of hospital stay
New‐onset atrial fibrillation
Permanent pacemaker implantation
Acute kidney injury
Any bleeding
Timing of outcome assessment
Short term; assessed during hospitalisation and up to 30 days of follow‐up.
Long term; assessed beyond 30 days of follow‐up.
The short‐term outcomes were of primary interest and were included in our Table 1.
Search methods for identification of studies
Electronic searches
We conducted systematic searches of the following bibliographic databases on 29 April 2019:
Cochrane Central Register of Controlled Trials (CENTRAL) in the Cochrane Library (Issue 4, April 2019)
MEDLINE and Epub Ahead of Print, In‐Process & Other Non‐Indexed Citations and Daily (Ovid, 1946 to 26 April 2019)
Embase and Embase Classic (Ovid, 1947 to 26 April 2019)
Web of Science Core Collection (Clarivate Analytics, 1900 to 26 April 2019)
We adapted the search strategy for MEDLINE (Ovid) (Appendix 1) for use in the other databases. We applied the Cochrane sensitivity‐maximising RCT filter to MEDLINE (Ovid) and adaptations of it to the other databases, except CENTRAL (Lefebvre 2011). We also conducted a search of the US National Institutes of Health Ongoing Trials Register ClinicalTrials.gov (www.clinicaltrials.gov) and the World Health Organization International Clinical Trials Registry Platform (WHO ICTRP) (apps.who.int/trialsearch) for ongoing or unpublished trials, on 29 April 2019. We searched all databases from inception to present and imposed no restriction on language of publication or publication status. We did not perform a separate search for adverse effects of interventions.
Searching other resources
We handsearched reference lists of all primary studies and review articles for additional references. We also contacted authors for missing data. There were no retraction statements or errata in our included studies.
Data collection and analysis
Selection of studies
Two review authors (AK, AN) independently screened titles and abstracts for inclusion of all the studies identified as a result of the search and coded them as 'retrieve' (eligible or potentially eligible/unclear) or 'do not retrieve'. We retrieved the full‐text study reports/publication, and two review authors (AK, AN) independently screened the full texts and identified studies for inclusion, and identified and recorded reasons for exclusion of the ineligible studies. There were no disagreements. We identified and excluded duplicates and collated multiple reports of the same study so that each study, rather than each report, is the unit of interest in the review. We recorded the selection process in sufficient detail to complete a PRISMA flow diagram (Figure 1) and 'Characteristics of excluded studies' table (Liberati 2009).
1.

PRISMA study flow diagram.
Data extraction and management
We piloted a data collection form that was used on all studies included in the review for study characteristics and outcome data. Two review authors (AK, AN) extracted the following study characteristics from the included studies:
Methods: study design, total duration of study, number of study centres and location, study setting, and date of study.
Participants: N randomised, N lost to follow‐up/withdrawn, N analysed, mean age, gender, surgical risk score at baseline, inclusion criteria, and exclusion criteria.
Interventions: intervention and comparison.
Outcomes: primary and secondary outcomes specified and collected, and time points reported.
Notes: funding for trial and notable conflicts of interest of trial authors.
Two review authors (AK, AN) independently extracted outcome data from the included studies and resolved any disagreements by discussion; or if agreement still could not be reached, reached consensus by involving a third review author to arbitrate. One review author (AK) transferred data into the Review Manager 5 file (Review Manager 2014). We double‐checked that data have been entered correctly by comparing the data presented in the systematic review with those in the data extraction form. A second review author (AN) spot‐checked study characteristics for accuracy against the trial report.
Assessment of risk of bias in included studies
Two review authors (AK, AN) independently assessed risk of bias for each study using the criteria outlined in the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2017). We resolved any disagreements by discussion. We assessed risk of bias according to the following domains:
Random sequence generation
Allocation concealment
Blinding of participants and personnel
Blinding of outcome assessment
Incomplete outcome data
Selective outcome reporting
Other bias
We graded each potential source of bias as high, low, or unclear and provide a quote from the study report together with a justification for our judgement in the 'Risk of bias in included studies' table. We summarised the 'Risk of bias' judgements across different studies for each of the domains listed. When considering treatment effects, we took into account the risk of bias for the studies that contribute to that outcome.
Assessment of bias in conducting the systematic review
We conducted the review according to our published protocol (Kolkailah 2019) with minor deviations as stated and justified in the 'Differences between protocol and review' section of the review.
Measures of treatment effect
We analysed dichotomous data as risk ratios (RR) and continuous data as mean difference (MD) with respective 95% confidence intervals (CI). Risk difference (RD) was calculated in Review Manager 5 software using the Mantel‐Haenszel method (Review Manager 2014).
Unit of analysis issues
There were no included studies with multi‐arm interventions. We analysed rehospitalisation by participants and not episodes.
Cluster‐randomised trials
We did not identify any cluster‐randomised trials to be included in our meta‐analysis.
Dealing with missing data
For included studies, we noted levels of attrition or missing individual data for our subgroup of interest. We first attempted to obtain missing data or data regarding our subgroup of interest from the trialists. When were unable to obtain this information, we attempted to explore the impact of including studies with missing data in the overall assessment of treatment effect by using a sensitivity analysis (see Sensitivity analysis). However, the pre‐specified sensitivity analysis criteria were not met. For all outcomes, we followed intention‐to‐treat (ITT) principles to the greatest degree possible, that is we analysed participants in their randomised group regardless of what intervention they actually received. We used available‐case data for the denominator when ITT data were not available.
Assessment of heterogeneity
We inspected forest plots visually to consider the direction and magnitude of effects and the degree of overlap between confidence intervals. We assessed statistical heterogeneity in each meta‐analysis using Tau², the I² statistic, and the Chi² statistic. We regarded heterogeneity as substantial if Tau² was greater than zero and either I² was greater than or equal to 50% or there was a low P value (< 0.1) in the Chi² test for heterogeneity.
Assessment of reporting biases
We did not pool more than 10 trials, thus we did not create a funnel plot to explore possible reporting biases for the primary outcomes.
Data synthesis
We undertook meta‐analyses only where this was meaningful, that is if the treatments, participants, and the underlying clinical question were similar enough for pooling to make sense. We carried out statistical analyses using Review Manager 2014. We used fixed‐effect meta‐analysis for combining data where it was reasonable to assume that studies were estimating the same underlying treatment effect, that is where trials were examining the same intervention, and we judged the trials’ populations and methods to be sufficiently similar. In case of substantial heterogeneity that could not be explained clinically or methodologically, we used random‐effects meta‐analysis to produce an overall summary where an average treatment effect across trials was considered clinically meaningful. We treated the random‐effects summary as the average range of possible treatment effects, and we discussed the clinical implications of treatment effects differing between trials. When we used random‐effects analyses, we presented the results as the average treatment effect with its 95% CI and the estimates of Tau² and the I² statistic. We reported the number needed to treat for an additional beneficial outcome (NNTB) and the number needed to treat for an additional harmful outcome (NNTH) with 95% CI when appropriate, that is if the absolute risk reduction (ARR) did not include zero. We addressed all outcomes listed in the Types of outcome measures section in the Results section of the review under the heading 'Effects of interventions', with outcomes addressed in the order in which they are shown in Types of outcome measures. In addition, we have included a summary of the main outcomes in the 'Table 1'. We include the results of individual studies and any statistical summary of these in 'Data and analyses' tables in the review. Most included trials reported the long‐term outcomes at 12 months of follow‐up, which were included in the meta‐analyses. We report other time intervals separately in the narrative.
Subgroup analysis and investigation of heterogeneity
When we identified substantial heterogeneity, we checked the data for accuracy, and then checked for methodological or clinical explanations for the heterogeneity. We were planning to explore this further using subgroup analyses. However, none of our pre‐specified subgroups were reported, hence we could not perform the following pre‐specified subgroup analyses:
Women versus men
Young versus old age (75 years of age cut‐off)
TAVI versus mini‐AVR
We were planning to use the following outcomes in subgroup analyses (regardless of the presence of heterogeneity):
All‐cause mortality
Stroke
Rehospitalisation
Sensitivity analysis
We were planning to carry out the following sensitivity analyses to test whether key methodological factors or decisions have affected the main result:
Restrict analysis to only include studies with low risk of bias in the following domains: random sequence generation and allocation concealment (selection bias) as well as incomplete outcome data (attrition bias).
Explore the effects of fixed‐effect versus random‐effects analyses for outcomes with substantial statistical heterogeneity.
Restrict analysis to only include studies with lower surgical risk cut‐off for inclusion of participants (i.e. STS risk score or EuroSCORE II of < 3%).
We intended to restrict the above to the primary outcomes. Hence, we were unable to perform the above as the pre‐specified criteria were not applicable to our included studies.
Since STACCATO 2012 utilised the less contemporary transapical TAVI and was prematurely terminated due to an excess of adverse events in the TAVI group, we elected to perform a post hoc sensitivity analysis to test whether inclusion of this study has affected the main result. In line with our pre‐planned strategy, we restricted this to the primary outcomes.
Reaching conclusions
We based our conclusions only on findings from the quantitative or narrative synthesis of included studies for this review. We avoided making recommendations for practice and our implications for research suggest priorities for future research and outline the remaining uncertainties in the area.
Summary of findings and assessment of the certainty of the evidence
We created a Table 1 using the following short‐term outcomes:
All‐cause mortality
Stroke
Rehospitalisation
Myocardial infarction
Cardiac death
Length of hospital stay
Permanent pacemaker implantation
We created another table (Appendix 2) including GRADE quality of evidence for short‐term outcomes that were not included in the main Table 1 (new‐onset atrial fibrillation, AKI, and any bleeding). We used the five GRADE considerations (study limitations, consistency of effect, imprecision, indirectness, and publication bias) to assess the quality of a body of evidence as it relates to the studies that contribute data to the meta‐analyses for the prespecified outcomes. We used methods and recommendations described in Chapter 12 of the Cochrane Handbook for Systematic Reviews of Interventions (Schünemann 2017), employing GRADEpro GDT software (GRADEpro GDT 2015). We justified all decisions to downgrade the quality of evidence using footnotes and made comments to aid readers' understanding of the review where necessary.
Results
Description of studies
We provide descriptions of studies in the Characteristics of included studies, Characteristics of excluded studies, and Characteristics of ongoing studies tables.
Results of the search
We ran the searches for the review on 29 April 2019. Our search yielded 3092 records identified through database searches. We identified 2148 records after removal of duplicates. After an initial screening, we marked 22 records for retrieval and assessment of their full text for eligibility. We excluded four studies, identified one ongoing study, and four are awaiting classification. We included 13 reports of four studies (Figure 1).
Included studies
Four studies, from 13 reports, fulfilled our eligibility criteria. All included studies provided quantitative data from 2818 participants that we included in the meta‐analysis (see Characteristics of included studies table for details regarding characteristics for all included participants).
Design
All included studies were parallel group RCTs (Evolut 2019; PARTNER 3 2019; NOTION 2015; STACCATO 2012).
Sample size
Included studies had a varying sample size with the largest being Evolut 2019 and smallest being STACCATO 2012. Evolut 2019 randomised 1468 participants, PARTNER 3 2019 randomised 1000 participants, NOTION 2015 randomised 280 participants, and STACCATO 2012 randomised 72 participants.
Location
Most included studies were multicentre and conducted in different countries. Evolut 2019 was conducted in 86 centres across Australia, Canada, France, Japan, the Netherlands, New Zealand, and the USA. PARTNER 3 2019 was conducted in 71 sites spanning the USA, Canada, Australia, New Zealand, and Japan. NOTION 2015 was conducted in three centres in Denmark and Sweden. STACCATO 2012 was similarly planned as a multicentre study across the Nordic region, but was eventually conducted only in two centres in Denmark in light of its early termination.
Setting
All included studies were performed in the inpatient setting (Evolut 2019; PARTNER 3 2019; NOTION 2015; STACCATO 2012).
Participants
Clinical characteristics
All studies included participants with severe AS undergoing TAVI or SAVR. Each study gave details of inclusion/exclusion criteria, as summarised in the Characteristics of included studies tables.
Age
All included studies had predominantly elderly participants (i.e. aged 70 or older). Mean age was slightly different across studies, however, with relatively younger participants in Evolut 2019 and PARTNER 3 2019 as opposed to NOTION 2015 and STACCATO 2012. The mean age in Evolut 2019 was 74.0 ± 5.9 years (TAVI) versus 73.8 ± 6.0 years (SAVR) and in PARTNER 3 2019 mean age was 73.3 ± 5.8 years (TAVI) versus 73.6 ± 6.1 years (SAVR). On the other hand the mean age in NOTION 2015 was 79.2 ± 4.9 years (TAVI) versus 79.0 ± 4.7 years (SAVR) and in STACCATO 2012 mean age was 80 ± 3.6 years (TAVI) and 82 ± 4.4 years (SAVR).
Sex
Sex distribution was different across studies. Evolut 2019 and PARTNER 3 2019 included more men (65.1% and 69.3%, respectively). Conversely, STACCATO 2012 included more women than men (60% vs. 30%). NOTION 2015 had a slightly more balanced distribution (46.8% women and 53.2% men).
Surgical risk score
Most included participants in all studies were at a low surgical risk as per their baseline STS/EuroSCORE II and/or as deemed by the study investigators. In Evolut 2019, the mean baseline STS risk score was 1.9% ± 0.7% (TAVI) versus 1.9% ± 0.7% (SAVR). This was very similar to the population included in PARTNER 3 2019, with a mean STS risk score of 1.9% ± 0.7% versus 1.9% ± 0.6% (SAVR). Additionally, in PARTNER 3 2019, the mean EuroSCORE II score was 1.5% ± 1.2% (TAVI) versus 1.5% ± 0.9% (SAVR). As for NOTION 2015, the mean STS risk score was 2.9% ± 1.6% (TAVI) versus 3.1% ± 1.7% (SAVR) and the mean EuroSCORE II was 1.9% ± 1.2% (TAVI) versus 2.0% ± 1.3% (SAVR). STACCATO 2012 had, relatively, the highest mean baseline STS risk score of 3.1% ± 1.5% (TAVI) versus 3.4% ± 1.2% (SAVR).
Interventions
All included studies randomised participants to TAVI or SAVR. Most TAVI procedures were performed via transfemoral access except in STACCATO 2012, where TAVI was performed via transapical approach. A minority of participants underwent alternative access in Evolut 2019 (direct aortic (0.4%) and trans‐subclavian (0.6%)) as well as NOTION 2015 (trans‐subclavian (3.5%)). TAVI in PARTNER 3 2019 was performed exclusively via transfemoral access. The valve type used in the TAVI groups was different between studies, where Evolut 2019 and NOTION 2015 used self‐expandable valves (CoreValve System, Evolut R, or Evolut PRO; Medtronic, Minneapolis, MN, USA), while PARTNER 3 2019 and STACCATO 2012 used balloon‐expandable valves (SAPIEN 3 and SAPIEN heart valve system; Edwards Lifesciences, Irvine, CA, USA). Surgical approach slightly varied across studies. Evolut 2019 did not specify the surgical approach used. PARTNER 3 2019 performed full sternotomy in 74.2% of surgical participants, while mini‐sternotomy and right anterior thoracotomy incisions were allowed in accordance with the protocol and performed in 24.3% of participants at the surgeons' discretion. Surgical participants in NOTION 2015 and STACCATO 2012 all underwent full sternotomy.
Funding source
All included studies declared their funding/support. Evolut 2019 was funded and supported by Medtronic and Paradigm Biostatistics. PARTNER 3 2019 was funded by Edwards Lifesciences. NOTION 2015 received grants from The Danish Heart Foundation and Medtronic medical writer assisted with manuscript drafting. STACCATO 2012 was primarily funded by the participating hospitals, but also received a study grant from The Danish Heart Association. The study reported no industry involvement.
Excluded studies
We excluded three studies (NCT02628899; NCT02838199; SURTAVI 2017). NCT02628899 was a non‐randomised trial and NCT02838199 was withdrawn by the study investigators. We initially considered SURTAVI 2017 as it included participants with STS risk score less than 4%. However, all study participants were deemed intermediate risk according to the study investigators (see Characteristics of excluded studies table for details).
Risk of bias in included studies
We provided detailed descriptions of the risk of bias in included studies in the 'Risk of bias in included studies' tables. See Figure 2 and Figure 3 for a summary of risk of bias assessments.
2.
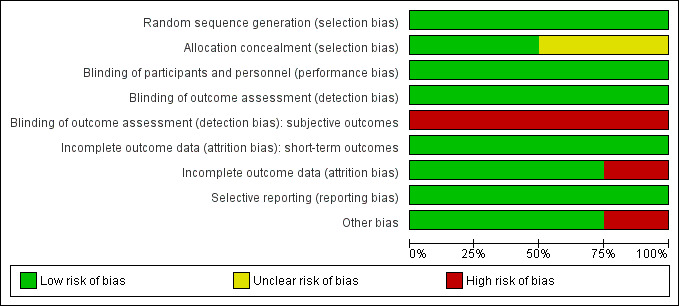
Risk of bias graph: review authors' judgements about each risk of bias item presented as percentages across all included studies.
3.

Risk of bias summary: review authors' judgements about each risk of bias item for each included study.
Allocation
All studies had low risk of bias in random sequence generation (Evolut 2019; PARTNER 3 2019; NOTION 2015; STACCATO 2012). Only PARTNER 3 2019 and NOTION 2015 had low risk of bias in allocation concealment, while the other two studies were at unclear risk (Evolut 2019; STACCATO 2012).
Blinding
All studies were unblinded or with unclear blinding due to the manner of the procedures. We deemed the risk of performance bias as low across all studies, however, since operative/interventional outcomes are unlikely to be influenced by lack of blinding of participants and personnel. As for detection bias, we deemed the risk of bias as low for most outcomes that are objective and unlikely to be affected by lack of blinding (Evolut 2019; PARTNER 3 2019; NOTION 2015; STACCATO 2012). The exception was for potentially subjective outcomes, such as rehospitalisation and LOS, which can be affected by lack of blinding and so we deemed risk of detection bias as high for these outcomes.
Incomplete outcome data
We classified attrition bias into short and long term for a more accurate assessment (i.e. within or beyond 30 days of follow‐up). All studies were at low risk for short‐term attrition bias (Evolut 2019; PARTNER 3 2019; NOTION 2015; STACCATO 2012). PARTNER 3 2019NOTION 2015 and STACCATO 2012 were at low risk for long‐term attrition bias, but Evolut 2019 was at high risk.
Selective reporting
We deemed all studies to be at low risk for selective reporting bias (Evolut 2019; PARTNER 3 2019; NOTION 2015; STACCATO 2012).
Other potential sources of bias
STACCATO 2012 utilised transapical TAVI and was prematurely terminated because of an overall excess of adverse events in the TAVI group as opposed to the SAVR group. Otherwise, we did not identify other sources of bias.
Effects of interventions
See: Table 1
See Table 1 for details of the main comparisons. Subgroup data were not available to perform our prespecified subgroup analyses. Pre‐specified criteria for sensitivity analyses were also not met.
Primary outcomes
All‐cause mortality
Four studies reported short‐term all‐cause mortality (Evolut 2019; PARTNER 3 2019; NOTION 2015; STACCATO 2012). There was probably little or no difference between TAVI and SAVR (RR 0.69, 95% CI 0.33 to 1.44; 2818 participants; 4 studies; Chi² = 2.30, df = 3 (P = 0.51); I² = 0.0%; moderate‐certainty evidence; Analysis 1.1). Please note that the data for Evolut 2019 were from Table S7 (ITT data) in the supplementary appendix, which slightly contradict data in Table S9 (causes of death) in the same appendix. Cause of death was given for four TAVI and eight SAVR participants. We included Table S7 ITT data as per our pre‐specified analysis plan. Three studies reported long‐term all‐cause mortality (Evolut 2019; PARTNER 3 2019; NOTION 2015). Similarly, there was little or no difference between both groups (RR 0.70, 95% CI 0.44 to 1.11; 2748 participants; 3 studies; Chi² = 1.12, df = 2 (P = 0.57); I² = 0.0%; Analysis 1.3). STACCATO 2012 reported 90‐day all‐cause mortality, where four of the 34 participants in the TAVI group had an event as opposed to none of the 36 in the SAVR group. NOTION 2015 reported longer term all‐cause mortality follow‐up with no difference between both groups at two years (TAVI 6.2% versus SAVR: 7.5%, RR 0.78, 95% CI 0.28 to 2.15) and five years (TAVI 22.3% versus SAVR 27.8%, RR 0.77, 95% CI 0.46 to 1.30). At six years, there was no difference in Kaplan‐Meier rates of all‐cause mortality (TAVI 42.5% versus SAVR 37.7, log‐rank P = 0.58).
1.1. Analysis.
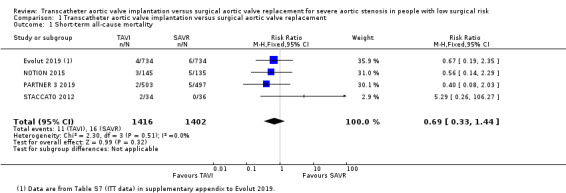
Comparison 1 Transcatheter aortic valve implantation versus surgical aortic valve replacement, Outcome 1 Short‐term all‐cause mortality.
1.3. Analysis.
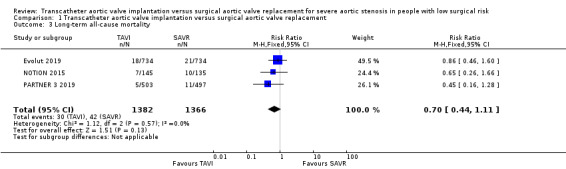
Comparison 1 Transcatheter aortic valve implantation versus surgical aortic valve replacement, Outcome 3 Long‐term all‐cause mortality.
Stroke
Four studies reported short‐term stroke with probably little or no difference between TAVI and SAVR (RR 0.73, 95% CI 0.42 to 1.25; 2818 participants; 4 studies; Chi² = 4.52, df = 3 (P = 0.21); I² = 34%; moderate‐certainty evidence; Analysis 1.4) (Evolut 2019; PARTNER 3 2019; NOTION 2015; STACCATO 2012). Three studies reported long‐term stroke also demonstrating little or no difference between both groups (RR 0.77, 95% CI 0.51 to 1.16; 2748 participants; 3 studies; Chi² = 2.44, df = 2 (P = 0.30); I² = 18%; Analysis 1.6) (Evolut 2019; PARTNER 3 2019; NOTION 2015). STACCATO 2012 reported 90‐day stroke, where three of the 34 participants in TAVI group had an event as opposed to one of the 36 in the SAVR group. NOTION 2015 reported longer‐term stroke follow‐up with no difference between both groups at two years (TAVI 3.6% versus SAVR 5.4%, log‐rank P = 0.46) and five years (TAVI 9.0% versus SAVR: 7.4%, log‐rank P = 0.65).
1.4. Analysis.
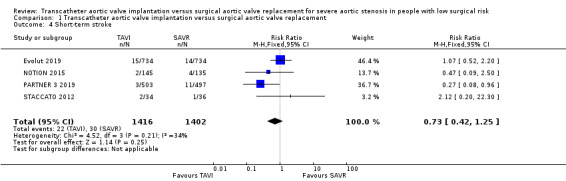
Comparison 1 Transcatheter aortic valve implantation versus surgical aortic valve replacement, Outcome 4 Short‐term stroke.
1.6. Analysis.
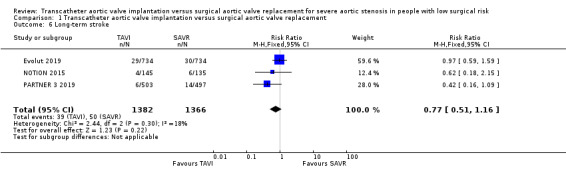
Comparison 1 Transcatheter aortic valve implantation versus surgical aortic valve replacement, Outcome 6 Long‐term stroke.
Rehospitalisation
Two studies reported short‐ and long‐term rehospitalisation (Evolut 2019; PARTNER 3 2019). TAVI may reduce the risk of short‐term rehospitalisation, although the confidence interval also includes the possibility of no difference in risk between groups (RR 0.64, 95% CI 0.39 to 1.06; 2468 participants; 2 studies; Chi² = 0.48, df = 1 (P = 0.49); I² = 0.0%; low‐certainty evidence; Analysis 1.7). The reduced risk of rehospitalisation with TAVI was sustained in the long term (RR 0.63, 95% CI 0.46 to 0.85; 2468 participants; 2 studies; Chi² = 0.98, df = 1 (P = 0.32); I² = 0.0%; Analysis 1.8).
1.7. Analysis.

Comparison 1 Transcatheter aortic valve implantation versus surgical aortic valve replacement, Outcome 7 Short‐term rehospitalisation.
1.8. Analysis.
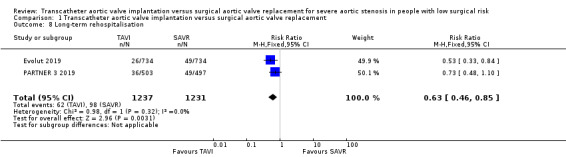
Comparison 1 Transcatheter aortic valve implantation versus surgical aortic valve replacement, Outcome 8 Long‐term rehospitalisation.
Secondary outcomes
Myocardial infarction (MI)
Four studies reported this outcome. Three trials (Evolut 2019; PARTNER 3 2019; NOTION 2015) had MI events and one trial (STACCATO 2012) had zero events in both arms. There was probably little or no difference between TAVI and SAVR in terms of short‐term MI (RR 0.82, 95% CI 0.42 to 1.58; 2748 participants; 3 studies; Chi² = 1.73, df = 2 (P = 0.42); I² = 0.0%; moderate‐certainty evidence; Analysis 1.9). In the long term, there was also little or no difference between both groups (RR 0.78, 95% CI 0.45 to 1.33; 2748 participants; 3 studies; Chi² = 1.22, df = 2 (P = 0.54); I² = 0.0%; Analysis 1.10). STACCATO 2012 reported zero events in both arms for this outcome in long‐term follow‐up at three months. NOTION 2015 reported a longer‐term MI follow‐up at two years (TAVI 5.1% vs SAVR 6.0%, log‐rank P = 0.69) and at five years (TAVI 7.7% vs SAVR 7.4%, log‐rank P = 0.96).
1.9. Analysis.
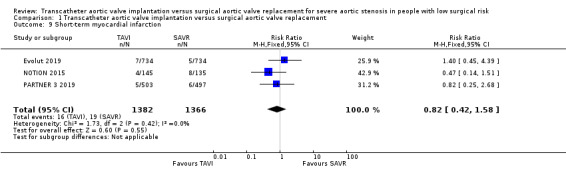
Comparison 1 Transcatheter aortic valve implantation versus surgical aortic valve replacement, Outcome 9 Short‐term myocardial infarction.
1.10. Analysis.
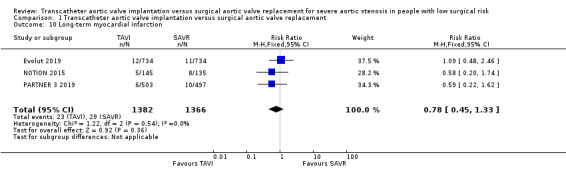
Comparison 1 Transcatheter aortic valve implantation versus surgical aortic valve replacement, Outcome 10 Long‐term myocardial infarction.
Cardiac death
Four studies reported short‐term cardiac death (Evolut 2019; PARTNER 3 2019; NOTION 2015; STACCATO 2012). There was probably little or no difference between TAVI and SAVR in short‐term cardiac death (RR 0.71, 95% CI 0.32 to 1.56; 2818 participants; 4 studies; Chi² = 1.18, df = 3 (P = 0.76); I² = 0.0%; moderate‐certainty evidence; Analysis 1.11). Long‐term cardiac death was reported by 3 studies and TAVI was associated with a reduced risk of long‐term cardiac death (RR 0.57, 95% CI 0.34 to 0.95; 2748 participants; 3 studies; Chi² = 0.27, df = 2 (P = 0.87); I² = 0.0%; Analysis 1.12) (Evolut 2019; PARTNER 3 2019; NOTION 2015). NOTION 2015 reported longer‐term cardiac death follow‐up with no difference between both groups at two years (TAVI 6.5% versus SAVR 9.1%, log‐rank P = 0.40) and five years (TAVI 20.8% versus SAVR 23.0%, log‐rank P = 0.62).
1.11. Analysis.
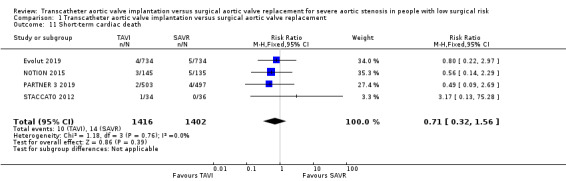
Comparison 1 Transcatheter aortic valve implantation versus surgical aortic valve replacement, Outcome 11 Short‐term cardiac death.
1.12. Analysis.

Comparison 1 Transcatheter aortic valve implantation versus surgical aortic valve replacement, Outcome 12 Long‐term cardiac death.
Length of hospital stay (LOS)
Three studies reported LOS (PARTNER 3 2019; NOTION 2015; STACCATO 2012). PARTNER 3 2019 reported shorter median LOS in days (interquartile range) in the TAVI group (TAVI 3 (2 to 3) days versus SAVR 7 (6 to 8) days, MD −4 (−4 to −3), P < 0.001). NOTION 2015 similarly reported a shorter mean LOS ± standard deviation (SD) with TAVI (TAVI 8.9 ± 6.2 days versus SAVR 12.9 ± 11.6 days, MD −4 (−6.2 to −1.8, P < 0.001). STACCATO 2012, on the other hand, reported a longer mean LOS after TAVI (TAVI 8.8 ± 6.7 days versus 7.6 ± 2.4 days, MD 1.2 (−1.18 to 3.58), P > 0.05). The different results of STACCATO 2012 are likely related to the higher frequency of complications associated with transapical TAVI which eventually resulted in early termination of the study. Given the considerable heterogeneity, we opted not to pool the results; Analysis 1.13). This outcome was deemed to have very low certainty of evidence, which means we are uncertain about the effect.
1.13. Analysis.

Comparison 1 Transcatheter aortic valve implantation versus surgical aortic valve replacement, Outcome 13 Length of hospital stay.
New‐onset atrial fibrillation
Three studies reported short‐ and long‐term atrial fibrillation (Evolut 2019; PARTNER 3 2019; NOTION 2015). TAVI was associated with a reduced risk of short‐term new‐onset atrial fibrillation (RR (random‐effects) 0.21, 95% CI 0.15 to 0.30; 2683 participants; 3 studies; Tau² = 0.06; Chi² = 5.66, df = 1 (P = 0.06); I² = 65; NNTB 3, 95% CI 4 to 3; high‐certainty evidence; Analysis 1.14). The risk of atrial fibrillation was similarly lower with TAVI in the long term (RR (random‐effects) 0.26, 95% CI 0.19 to 0.35; 2683 participants; 3 studies; Tau² = 0.05; Chi² = 5.69, df = 2 (P = 0.06); I² = 65%; NNTB 3, 95% CI 4 to 3; Analysis 1.15). We confirmed data accuracy and absence of methodological errors to investigate the observed heterogeneity. While all studies used the same Valve Academic Research Consortium (VARC) definitions, NOTION 2015 reported new‐onset or "worsening" atrial fibrillation. This may be a potential explanation for the observed heterogeneity. Hence, we used random‐effects model for pooling of results. NOTION 2015 reported longer‐term follow‐up with persistently lower frequency of new‐onset or worsening atrial fibrillation in the TAVI arm at two years (TAVI 22.7% versus SAVR 60.2%, log‐rank P < 0.001) and five years (TAVI 23.4% versus SAVR 60.8%, log‐rank P < 0.001).
1.14. Analysis.

Comparison 1 Transcatheter aortic valve implantation versus surgical aortic valve replacement, Outcome 14 Short‐term new‐onset atrial fibrillation.
1.15. Analysis.

Comparison 1 Transcatheter aortic valve implantation versus surgical aortic valve replacement, Outcome 15 Long‐term new‐onset atrial fibrillation.
Permanent pacemaker (PPM) implantation
Three studies reported short‐ and long‐term PPM implantation (Evolut 2019; PARTNER 3 2019; NOTION 2015). TAVI probably increased the risk of short‐term PPM implantation (RR (random‐effects) 3.65, 95% CI 1.50 to 8.87; 2683 participants; 3 studies; Tau² = 0.47; Chi² = 11.70, df = 2 (P = 0.003); I² = 83%; moderate‐certainty evidence; NNTH 7, 95% CI 4 to 43; Analysis 1.16). This increased risk with TAVI was sustained in the long term (RR (random‐effects) 3.48, 95% CI 1.40 to 8.62; 2683 participants; 3 studies; Tau² = 0.53; Chi² = 15.78, df = 2 (P = 0.0004); I² = 87%; NNTH 6, 95% CI 3 to 48; Analysis 1.17). There was, however, observed heterogeneity despite confirmation of data accuracy and absence of methodological errors. Possible explanations for such heterogeneity may include differences in the populations or interventions of trials. For instance, the valve type used in the TAVI groups was different between studies, where NOTION 2015 and Evolut 2019 used self‐expandable valves, while PARTNER 3 2019 used balloon‐expandable valves. Additionally, NOTION 2015 was conducted in Denmark and Sweden, while PARTNER 3 2019 was conducted in the USA, Canada, Australia, New Zealand, and Japan. Evolut 2019 had similar study centres to the latter with the addition of France and the Netherlands. Furthermore, NOTION 2015 participants were older on average. We also noted the lower number of events in the surgical arm of NOTION 2015 as compared to the other two studies; hence we used random‐effects model for pooling of results. STACCATO 2012 reported 90‐day PPM implantation, where two of the 34 participants in TAVI group had an event as opposed to one of the 36 in the SAVR group. NOTION 2015 reported longer‐term follow‐up for PPM implantation with a persistently increased risk in TAVI recipients at two years (TAVI 41.3% versus SAVR 4.2%, log‐rank P < 0.001) and five years (TAVI 41.7% versus SAVR: 7.8%, log‐rank P < 0.001).
1.16. Analysis.
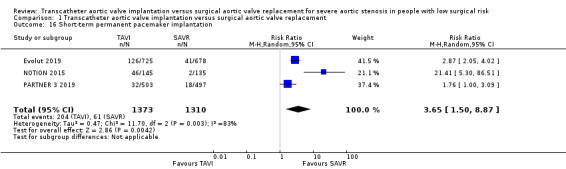
Comparison 1 Transcatheter aortic valve implantation versus surgical aortic valve replacement, Outcome 16 Short‐term permanent pacemaker implantation.
1.17. Analysis.

Comparison 1 Transcatheter aortic valve implantation versus surgical aortic valve replacement, Outcome 17 Long‐term permanent pacemaker implantation.
Acute kidney injury (AKI)
Four studies reported AKI (Evolut 2019; PARTNER 3 2019; NOTION 2015; STACCATO 2012) and TAVI was associated with a lower risk (RR 0.30, 95% CI 0.16 to 0.58; 2753 participants; 4 studies; Chi² = 3.32, df = 3 (P = 0.34); I² = 10%; NNTB 50, 95% CI 33 to 100; high‐certainty evidence; Analysis 1.18).
1.18. Analysis.
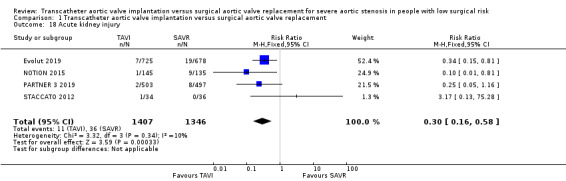
Comparison 1 Transcatheter aortic valve implantation versus surgical aortic valve replacement, Outcome 18 Acute kidney injury.
Any bleeding
Four studies reported short‐term bleeding and TAVI was associated with a reduced risk (RR (random‐effects) 0.31, 95% CI 0.16 to 0.62; 2753 participants; 4 studies; Tau² = 0.30; Chi² = 11.39, df = 3 (P = 0.010); I² = 74%; high‐certainty evidence; Analysis 1.19) (Evolut 2019; PARTNER 3 2019; NOTION 2015; STACCATO 2012). There was a slight variation in definition of bleeding across the four studies, which could potentially explain the observed heterogeneity. We pooled the results using a random‐effects model for this reason. Two studies reported long‐term bleeding (Evolut 2019; PARTNER 3 2019); and TAVI was similarly associated with a lower risk (RR 0.33, 95% CI 0.25 to 0.44; 2403 participants; 2 studies; Chi² = 0.14, df = 1 (P = 0.71); I² = 0%; NNTB 10, 95% CI 8 to 12; Analysis 1.20). The long‐term results included in the meta‐analysis are the one‐year follow‐up outcomes. The 90‐day bleeding events in STACCATO 2012 were similar to that of the one‐year follow‐up (1 event in TAVI and 1 event in SAVR arm).
1.19. Analysis.
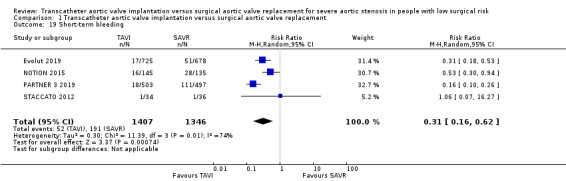
Comparison 1 Transcatheter aortic valve implantation versus surgical aortic valve replacement, Outcome 19 Short‐term bleeding.
1.20. Analysis.
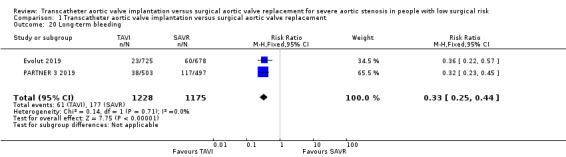
Comparison 1 Transcatheter aortic valve implantation versus surgical aortic valve replacement, Outcome 20 Long‐term bleeding.
Subgroup analysis
We were unable to perform subgroup analysis since none of the subgroups that we had pre‐specified was reported.
Sensitivity analysis
We were unable to carry out the sensitivity analyses that we had pre‐specified in our protocol since the criteria were not applicable to our included studies. However, since STACCATO 2012 utilised the less contemporary transapical TAVI and was prematurely terminated due to an excess of adverse events in the TAVI group, we elected to perform a post hoc sensitivity analysis on each primary outcome to test whether inclusion of this study has affected the main result. After exclusion of STACCATO 2012, there remained little or no difference between TAVI and SAVR in short‐term all‐cause mortality (RR 0.55, 95% CI 0.24 to 1.23; 2748 participants; 3 studies; Chi² = 0.25, df = 2 (P = 0.88); I² = 0.0%; Analysis 1.2), nor stroke (RR 0.68, 95% CI 0.39 to 1.20; 2748 participants; 3 studies; Chi² = 3.76, df = 2 (P = 0.15); I² = 47%; Analysis 1.5).
1.2. Analysis.
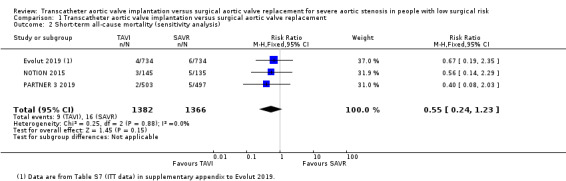
Comparison 1 Transcatheter aortic valve implantation versus surgical aortic valve replacement, Outcome 2 Short‐term all‐cause mortality (sensitivity analysis).
1.5. Analysis.

Comparison 1 Transcatheter aortic valve implantation versus surgical aortic valve replacement, Outcome 5 Short‐term stroke (sensitivity analysis).
Discussion
Summary of main results
We identified four studies that met our inclusion criteria and one ongoing study. The RCTs included participants with severe AS undergoing TAVI or SAVR who are deemed at a low surgical risk by the study investigators. TAVI and SAVR probably make little or no difference in short‐term all‐cause mortality as well as stroke risk (moderate‐certainty evidence), both of which remained similar at long‐term follow‐up. TAVI may reduce the risk of short‐term rehospitalisation (low‐certainty evidence) and this reduction in readmission rates was sustained at long‐term follow‐up. There is probably little or no difference between both groups in terms of incident MI in the short term (moderate‐certainty evidence) or long term. There is probably little or no difference between both groups in terms of short‐term cardiac death (moderate‐certainty evidence), but SAVR portends a higher risk of cardiac death in the long term. We are uncertain whether TAVI affects the LOS, although it appears to be associated with shorter LOS (very low certainty evidence). In both short and long terms, TAVI is associated with a reduced risk of atrial fibrillation, AKI, and bleeding (high‐certainty evidence). Conversely, TAVI probably increases the risk of short‐term PPM implantation, as compared to SAVR (moderate‐certainty evidence), and this increased risk is sustained in the long term.
Overall completeness and applicability of evidence
The four included studies, from 13 reports, enrolled 2818 participants and were conducted across various continents in countries with different levels of income. They included participants with severe AS undergoing TAVI or SAVR who are deemed at a low surgical risk. Most participants were elderly (i.e. aged 70 or older), so the results may not necessarily generalise to younger populations. More men than women were represented in the current available evidence. Furthermore, it is worth highlighting some of the trials' exclusion criteria for which the current evidence may not be applicable. For instance, PARTNER 3 2019 excluded participants with bicuspid valves and non‐transfemoral access. Evolut 2019 similarly excluded participants with bicuspid valves but included alternative access TAVI. Both trials excluded participants with unsuitable anatomy (e.g. LVOT calcification, too small or too large valves, etc.) Please refer to the full details on exclusion criteria of individual trials under Characteristics of included studies. There was not enough data to investigate our pre‐planned subgroup or sensitivity analyses in any meaningful way. Most of the evidence is limited to one year, although NOTION 2015 reported outcomes up to six years. Evolut 2019 and PARTNER 3 2019 are also still ongoing with longer planned follow‐up; all of these results will be included in the update of the current review. SAVR was mostly done via full sternotomy and only about one‐fourth of PARTNER 3 2019 underwent mini‐sternotomy or right anterior thoracotomy. The valve type used in the TAVI groups was different across studies (i.e. self‐expandable vs. balloon‐expandable valves) as described before. STACCATO 2012 utilised transapical TAVI and was prematurely terminated due to a high rate of adverse events in the TAVI group. Inclusion of this study did not, however, impact the main results as demonstrated by the post hoc sensitivity analysis. Regarding quality of life measures, PARTNER 3 2019 reported the mean relative change from baseline in the Kansas City Cardiomyopathy Questionnaire (KCCQ) at 30 days (TAVI 37.8% vs SAVR 12.8%) and one year (TAVI 39.7% vs SAVR 38.7%). Evolut 2019 reported the KCCQ overall summary score ± SD at 30 days (TAVI 88.7% ± 14.2% versus SAVR 78.6% ± 18.9%) with similar summary scores at 12 months (TAVI 90.3% ± 12.7% vs SAVR 90.8% ± 12.4%). These results suggest that quality of life was, more or less, comparable at one year. None of the studies reported cost or economic data. There are other relevant outcomes to be considered, such as paravalvular leak/regurgitation and aortic valve re‐intervention, which have not been included in the current analysis. These outcomes will likely be included in future updates of this review as we have longer follow‐up on valve performance and durability.
Quality of the evidence
Our review included four studies, from 13 reports, with 2818 participants. There was some concern for imprecision, however, since the number of events may have not been sufficient to detect a significant difference between groups. This was a consistent reason for downgrading the quality of evidence of all outcomes. All studies were adequately randomised and so were at a low risk of selection bias (in terms of random sequence generation domain), but half were unclear in the allocation concealment methods. In all studies, neither the participants nor the physicians were blinded (or it was unclear if they were) owing to the manner of the interventions, but we judged performance and detection biases as low risk for most outcomes that are objective and unlikely to be affected by lack of blinding. For potentially subjective outcomes, such as rehospitalisation and LOS, we deemed the risk of detection bias as high (knowledge of the intervention may 'subconsciously' affect physicians' decision to readmit participants or alter their LOS). We are confident that all included studies clearly address our review question, with no concern for indirectness in participants, interventions, comparators, or outcomes. We evaluated the quality of evidence using the GRADE approach. All of our main outcomes (all‐cause mortality, stroke, rehospitalisation, MI, cardiac death) had a moderate quality of evidence, except rehospitalisation and LOS which had low and very low quality of evidence, respectively. There were several reasons for further downgrading respective outcomes' quality of evidence. For instance, LOS point estimates varied widely across studies and confidence intervals did not show overlap, which raised concern for inconsistency of results. Additionally, for the same outcome there was a high risk of detection bias due to lack of blinding of included studies, which further downgraded the quality of evidence due to concern for study limitations. This also applied to the rehospitalisation outcome. In addition, LOS did not meet the optimal information size criterion (Schünemann 2013) and so we further downgraded the quality of evidence due to concern for imprecision. PPM implantation had considerable unexplained heterogeneity and so we downgraded the quality of evidence for inconsistency. We deemed the evidence for new‐onset atrial fibrillation, AKI, and any bleeding to be of high quality with no reasons for downgrading.
Potential biases in the review process
The methodological rigour of Cochrane Reviews aims to minimise different potential biases. We performed an exhaustive search to identify all eligible studies that addressed our review question. We did not apply language or date restrictions to the publications searched. However, any search strategy has an inherent risk of missing relevant studies. Additionally, there is always a pragmatic restriction to the number of searched resources and an English language bias. Lastly, as previously mentioned, there was an overall imbalance between men and women, surgical approaches varied slightly, and valve types/generations were different across studies. These issues may have also biased our review process.
Agreements and disagreements with other studies or reviews
We present the largest meta‐analysis conducted to date, comprising 2818 participants, that is solely based on RCTs comparing TAVI and SAVR for severe AS in individuals with low surgical risk. This includes the landmark trials published this year. We identified eight other reviews addressing our clinical question with similar inclusion criteria. Arora 2017 assessed the early outcomes of TAVI and SAVR from one RCT and three propensity‐score matched (PSM) studies demonstrating comparable results but suggesting the need for further long‐term studies. Despite our inclusion of three additional RCTs since their publication, we agree that longer‐term follow‐up is still needed. Witberg 2018 conducted a meta‐analysis of two RCTs and four PSM studies, which demonstrated an increased risk of mortality with TAVI, at a median follow‐up of two years, suggesting that SAVR should remain the mainstay of treatment for AS. This study included 3484 participants, only 350 of whom were RCT‐derived. Additionally, the majority of the current evidence stems from the studies included in our meta‐analysis, which were not published at the time, so we believe the conclusions drawn from that report may not be current. The same group, Witberg 2019, conducted a more recent meta‐analysis including four RCTs and five PSM studies with a larger sample size of 6124. However, this was again predominantly derived from the PSM studies. They concluded that TAVI has similar mortality to SAVR at two years of follow‐up and highlighted the need for long‐term follow‐up data, which we agree with. Overtchouk 2019 reported a comprehensive review of individual RCTs and observational studies with no conducted meta‐analyses. After meticulous review of available evidence, they concluded that TAVI, in the future, may replace SAVR in low‐risk populations. Junquera 2019 published a review discussing the future of TAVI in low‐risk participants. At the time, the major landmark trials included in our review were still ongoing. However, the authors "expected" the positive results demonstrated by TAVI in these trials, which they perceived as the basis to establish TAVI as default treatment for AS. We share both mentioned views on whether TAVI will withstand the test of time, durability being one of the main considerations for AVR. Kheiri 2019 conducted a meta‐analysis, including three RCTs with 604 participants. While their conclusions are similar to ours, our meta‐analysis is based on a substantially larger sample, including the two most recent landmark trials, and thus provides more accurate and contemporary estimates. Kolte 2019, on the other hand, included four RCTs with 2887 participants and definitively concluded that TAVI is associated with a lower risk of all‐cause mortality and cardiac death at one year. They further suggested that TAVI may be the preferred option over SAVR in low‐risk individuals. While Kheiri 2019 and Kolte 2019 have different conclusiveness, they both share one major difference in their methodology compared to ours: both reviews included SURTAVI 2017 in their meta‐analyses. Based on our correspondence with SURTAVI 2017's lead investigator, we confirmed that participants in this trial, including ones with STS risk score of less than 4%, are not representative of the low surgical risk population. Hence, as justified under Characteristics of excluded studies, we opted to exclude SURTAVI 2017 from our meta‐analysis. Al‐Abdouh 2019 published a recent meta‐analysis, with similar eligibility criteria to ours — they only included RCTs, comprising 2698 participants, and had similar results. However, they opted to exclude STACCATO 2012 given its relatively short follow‐up owing to its premature termination and utilization of transapical TAVI. We do not think this decision is unreasonable; however, we believe a negative outcome as such should still be presented with the available evidence. Notably, as discussed previously, we did not include the 3‐month follow‐up and only considered the short‐term STACCATO 2012 outcome data, which were before study termination. We further performed a post hoc sensitivity analysis for the primary outcomes, excluding STACCATO 2012, which did not change our main findings. Despite the mentioned differences between our reviews, our results both demonstrate no difference in short‐term mortality risk and a reduced risk of perioperative complications with TAVI, aside from PPM implantation. We agree that the current available evidence as well as the anticipated long‐term follow‐up results would warrant an update in the guidelines for management of AS in low‐risk individuals. There is a paradigm shift in management of individuals with severe AS who are at a low surgical risk with a growing momentum towards less invasive approaches. With the enhanced operator experience, continued valve systems technological advancements, and development of newer generation devices, TAVI indications continue to expand in the modern era, supported by the recent US Food and Drug Administration approval of certain TAVI devices in low‐risk individuals (FDA 2019). This further emphasises the indispensable role of a "heart team" towards fostering a clinical environment of well‐informed, shared decision‐making with special focus on the highlighted uncertainties in evidence and unclear durability of TAVI at this point in time.
Authors' conclusions
Implications for practice.
Our meta‐analysis indicates that, in the short term, TAVI probably has little or no mortality difference compared to SAVR for severe AS in individuals with low surgical risk. Similarly, there is probably little or no difference in risk of stroke, MI, and cardiac death between the two approaches. TAVI may reduce the risk of rehospitalisation, but we are uncertain about the effects on LOS. TAVI reduces the risk of atrial fibrillation, AKI, and bleeding. However, this benefit is offset by the increased risk of PPM implantation. Long‐term follow‐up data are needed to further assess and validate these outcomes, especially durability, in the low surgical risk population.
Implications for research.
In light of the current state of evidence, more RCTs with longer follow‐up data are needed to further assess and validate the durability of TAVI for severe AS in individuals with low surgical risk. It may also be prudent to emphasise patient‐centred outcomes, such as pain scores, quality of life measures, and recovery time, etc. Additionally, investigators should strive to include younger participants with a more equal distribution of men and women. The NOTION‐2 trial is underway with inclusion of younger participants and a planned minimum of five‐year follow‐up (NCT02825134). This would add to the current body of evidence and we would hope for an even longer follow‐up of ten years and beyond.
Acknowledgements
We would like to thank Nicole Martin and the editorial team of the Cochrane Heart Group for their support. The search strategy was designed by Charlene Bridges and peer reviewed by Robin Featherstone. Special thanks to Helen Wakeford, Sarah Hodgkinson, Rui Providencia, and the Cochrane Fast‐Track team for their productive feedback throughout the editorial process. We thank the peer referees Tomo Ando, Rajesh K Kharbanda, Antonio Miceli and Hemang Panchal, and consumer referee Terry Griffin, for all of their helpful comments. We would also like to thank Dr. Michael J Reardon for the correspondence and information provided on SURTAVI 2017 trial.
Appendices
Appendix 1. Search strategies
CENTRAL
#1 MeSH descriptor: [Aortic Valve Stenosis] explode all trees
#2 (aortic NEAR/3 stenos*)
#3 MeSH descriptor: [Heart Valve Diseases] this term only
#4 ((heart OR aortic) NEAR/2 (valv* NEAR/2 disease*))
#5 #1 OR #2 OR #3 OR #4
#6 MeSH descriptor: [Transcatheter Aortic Valve Replacement] this term only
#7 ((transapical OR transventricular OR percutaneous OR transcatheter*) NEAR/3 (valve* OR prosthe* OR bioprosthe*))
#8 (TAVI OR PAVR OR TAVR)
#9 #6 OR #7 OR #8
#10 MeSH descriptor: [Heart Valve Prosthesis Implantation] this term only
#11 ((surg* OR open‐heart) NEAR/3 (valve* OR prosthe* OR bioprosthe*))
#12 #10 OR #11
#13 #5 AND #9 AND #12
MEDLINE Ovid
1. exp Aortic Valve Stenosis/
2. (aortic adj3 stenos*).tw.
3. Heart Valve Diseases/
4. ((heart or aortic) adj2 (valv* adj2 disease*)).tw.
5. or/1‐4
6. Transcatheter Aortic Valve Replacement/
7. ((transapical or transventricular or percutaneous or transcatheter*) adj3 (valve* or prosthe* or bioprosthe*)).tw.
8. (TAVI or PAVR or TAVR).tw.
9. or/6‐8
10. Heart Valve Prosthesis Implantation/
11. ((surg* or open‐heart) adj3 (valve* or prosthe* or bioprosthe*)).tw.
12. 10 or 11
13. 9 and 12
14. 5 and 13
15. randomised controlled trial.pt.
16. controlled clinical trial.pt.
17. randomized.ab.
18. placebo.ab.
19. drug therapy.fs.
20. randomly.ab.
21. trial.ab.
22. groups.ab.
23. 15 or 16 or 17 or 18 or 19 or 20 or 21 or 22
24. exp animals/ not humans.sh.
25. 23 not 24
26. 14 and 25
Embase Ovid
1. aortic valve stenosis/
2. (aortic adj3 stenos*).tw.
3. valvular heart disease/
4. ((heart or aortic) adj2 (valv* adj2 disease*)).tw.
5. or/1‐4
6. transcatheter aortic valve implantation/
7. ((transapical or transventricular or percutaneous or transcatheter*) adj3 (valve* or prosthe* or bioprosthe*)).tw.
8. (TAVI or PAVR or TAVR).tw.
9. or/6‐8
10. exp heart valve replacement/
11. ((surg* or open‐heart) adj3 (valve* or prosthe* or bioprosthe*)).tw.
12. 10 or 11
13. 5 and 9 and 12
14. random$.tw.
15. factorial$.tw.
16. crossover$.tw.
17. cross over$.tw.
18. cross‐over$.tw.
19. placebo$.tw.
20. (doubl$ adj blind$).tw.
21. (singl$ adj blind$).tw.
22. assign$.tw.
23. allocat$.tw.
24. volunteer$.tw.
25. crossover procedure/
26. double blind procedure/
27. randomised controlled trial/
28. single blind procedure/
29. 14 or 15 or 16 or 17 or 18 or 19 or 20 or 21 or 22 or 23 or 24 or 25 or 26 or 27 or 28
30. (animal/ or nonhuman/) not human/
31. 29 not 30
32. 13 and 31
Web of Science Core Collection
#13 #12 AND #11
#12 TS=(random* or blind* or allocat* or assign* or trial* or placebo* or crossover* or cross‐over*)
#11 #10 AND #7 AND #4
#10 #9 OR #8
#9 TS=((surg* NEAR valve*) OR (surg* NEAR prosthe*) OR (surg* NEAR bioprosthe*) OR (open‐heart NEAR valve*) OR (open‐heart NEAR prosthe*) OR (open‐heart NEAR bioprosthe*))
#8 TS=Heart Valve Prosthesis Implantation
#7 #6 OR #5
#6 TS=(TAVI or PAVR or TAVR)
#5 TS=((transapical NEAR valve*) OR (transapical NEAR prosthe*) OR (transapical NEAR bioprosthe*) OR ( transventricular NEAR valve*) OR (transventricular NEAR prosthe*) OR (transventricular NEAR bioprosthe*) OR (percutaneous NEAR valve*) OR (percutaneous NEAR prosthe*) OR (percutaneous NEAR bioprosthe*) OR (transcatheter* NEAR valve*) OR (transcatheter* NEAR prosthe*) OR (transcatheter*NEAR bioprosthe*))
#4 #3 OR #2 OR #1
#3 TS=aortic valv* disease*
#2 TS=heart valv* disease*
#1 TS=aortic stenos*
Clinicaltrials.gov
Advanced search
Interventional Studies | Aortic Stenosis | aortic valve replacement | Adult, Older Adult
WHO ICTRP
Condition: aortic stenosis
Intervention: aortic valve replacement
Appendix 2. GRADE quality of evidence for other short‐term outcomes (not included in the main SoF table)
| TAVI compared to SAVR in people with severe AS and low surgical risk at short‐term follow‐up (up to 30 days) | |||||
| Patient or population: adults with severe aortic stenosis who are at low surgical risk. Setting: inpatient: Australia, Canada, France, Japan, the Netherlands, New Zealand, the USA, Denmark, and Sweden. Intervention: transcatheter aortic valve implantation. Comparison: surgical aortic valve replacement. | |||||
| Outcomes (up to 30 days' follow‐up) | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Certainty of the evidence (GRADE) | |
| Risk with surgical aortic valve replacement (SAVR) | Risk with transcatheter aortic valve implantation (TAVI) | ||||
| New‐onset atrial fibrillation | 353 per 1000 | 74 per 1000 (53 to 106) | RR 0.21 (0.15 to 0.30) | 2683 (3 RCTs) | ⊕⊕⊕⊕ HIGH |
| Acute kidney injury | 27 per 1000 | 8 per 1000 (4 to 16) | RR 0.30 (0.16 to 0.58) | 2753 (4 RCTs) | ⊕⊕⊕⊕ HIGH |
| Any bleeding | 142 per 1000 | 44 per 1000 (23 to 88) | RR 0.31 (0.16 to 0.62) | 2753 (4 RCTs) | ⊕⊕⊕⊕ HIGH |
All outcomes reported herein are short term (i.e. assessed during hospitalisation and up to 30 days of follow‐up).
*The risk in the intervention group is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI).
CI: Confidence interval; RR: Risk ratio; SoF: Summary of findings.
Data and analyses
Comparison 1. Transcatheter aortic valve implantation versus surgical aortic valve replacement.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Short‐term all‐cause mortality | 4 | 2818 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.69 [0.33, 1.44] |
| 2 Short‐term all‐cause mortality (sensitivity analysis) | 3 | 2748 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.55 [0.24, 1.23] |
| 3 Long‐term all‐cause mortality | 3 | 2748 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.70 [0.44, 1.11] |
| 4 Short‐term stroke | 4 | 2818 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.73 [0.42, 1.25] |
| 5 Short‐term stroke (sensitivity analysis) | 3 | 2748 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.68 [0.39, 1.20] |
| 6 Long‐term stroke | 3 | 2748 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.77 [0.51, 1.16] |
| 7 Short‐term rehospitalisation | 2 | 2468 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.64 [0.39, 1.06] |
| 8 Long‐term rehospitalisation | 2 | 2468 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.63 [0.46, 0.85] |
| 9 Short‐term myocardial infarction | 3 | 2748 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.82 [0.42, 1.58] |
| 10 Long‐term myocardial infarction | 3 | 2748 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.78 [0.45, 1.33] |
| 11 Short‐term cardiac death | 4 | 2818 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.71 [0.32, 1.56] |
| 12 Long‐term cardiac death | 3 | 2748 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.57 [0.34, 0.95] |
| 13 Length of hospital stay | 2 | Mean Difference (IV, Random, 95% CI) | Subtotals only | |
| 14 Short‐term new‐onset atrial fibrillation | 3 | 2683 | Risk Ratio (M‐H, Random, 95% CI) | 0.21 [0.15, 0.30] |
| 15 Long‐term new‐onset atrial fibrillation | 3 | 2683 | Risk Ratio (M‐H, Random, 95% CI) | 0.26 [0.19, 0.35] |
| 16 Short‐term permanent pacemaker implantation | 3 | 2683 | Risk Ratio (M‐H, Random, 95% CI) | 3.65 [1.50, 8.87] |
| 17 Long‐term permanent pacemaker implantation | 3 | 2683 | Risk Ratio (M‐H, Random, 95% CI) | 3.48 [1.40, 8.62] |
| 18 Acute kidney injury | 4 | 2753 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.30 [0.16, 0.58] |
| 19 Short‐term bleeding | 4 | 2753 | Risk Ratio (M‐H, Random, 95% CI) | 0.31 [0.16, 0.62] |
| 20 Long‐term bleeding | 2 | 2403 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.33 [0.25, 0.44] |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Evolut 2019.
| Methods | Study design: multinational, randomised, non‐inferiority clinical trial comparing the safety and efficacy of TAVI with those of surgery in patients with severe aortic stenosis who were deemed to be at low risk for death at 30 days with surgery. Total study duration: 24 months (median follow‐up in each group 12.2 months) Number of study centres and location: 86 centres in Australia, Canada, France, Japan, the Netherlands, New Zealand, and the USA. Study setting: inpatient. Date of study: March 2016 to November 2018 (for the current report). However, the study is ongoing with an anticipated end date of March 2026. |
|
| Participants | Eligible patients had severe aortic valve stenosis with suitable anatomy for transcatheter aortic valve implantation or surgery and no more than a predicted 3% risk of death by 30 days with surgery, as assessed by members of the local heart team. N‐randomised: 1468; 734 were assigned to TAVI and 734 were assigned to surgery. As‐treated cohort: 1403 patients; 725 in the TAVI group and 678 in the surgery group. N‐lost to follow‐up/withdrawn: 9 in the TAVI group and 27 in surgery group + crossovers. Final per protocol population: 1355; 706 in the TAVI group and 649 in the surgery group. Mean age: 74.1 years. Gender: 34.9% females. Surgical risk score at baseline: STS‐PROM: TAVI: 1.9% ± 0.7% versus SAVR: 1.9% ± 0.7% Inclusion criteria: 1. Severe aortic stenosis, defined as follows: a) For symptomatic patients: Aortic valve area ≤ 1.0 cm² (or aortic valve area index of ≤ 0.6 cm²/m²), OR mean gradient ≥ 40 mmHg, OR maximal aortic valve velocity ≥ 4.0 m/sec by transthoracic echocardiography at rest. b) For asymptomatic patients: i. Very severe aortic stenosis with an aortic valve area of ≤ 1.0 cm² (or aortic valve area index of ≤ 0.6 cm²/m²), AND maximal aortic velocity ≥ 5.0 m/sec, or mean gradient ≥ 60 mmHg by transthoracic echocardiography at rest, OR ii. Aortic valve area of ≤ 1.0 cm² (or aortic valve area index of ≤ 0.6 cm²/m²), AND a mean gradient ≥ 40 mmHg or maximal aortic valve velocity ≥ 4.0 m/sec by transthoracic echocardiography at rest, AND an exercise tolerance test that demonstrates a limited exercise capacity, abnormal blood pressure response, or arrhythmia OR iii. Aortic valve area of ≤ 1.0 cm² (or aortic valve area index of ≤ 0.6 cm²/m²), AND mean gradient ≥ 40 mmHg, or maximal aortic valve velocity ≥ 4.0 m/sec by transthoracic echocardiography at rest, AND a left ventricular ejection fraction < 50%. 2. Patient is considered low risk for surgery, where low risk is defined as predicted risk of mortality for surgery < 3% at 30 days per multidisciplinary local heart team assessment. 3. The patient and the treating physician agree that the patient will return for all required post‐procedure follow‐up visits. Exclusion criteria: Patients are NOT eligible for trial participation if they meet ANY of the following exclusion criteria: 1. Any condition considered a contraindication for placement of a bioprosthetic valve (e.g. patient is indicated for mechanical prosthetic valve). 2. A known hypersensitivity or contraindication to any of the following that cannot be adequately pre‐medicated: a. aspirin or heparin (HIT/HITTS) and bivalirudin b. ticlopidine and clopidogrel c. Nitinol (titanium or nickel) d. contrast media 3. blood dyscrasias as defined: leukopenia (white blood cell count < 1000 mm³), thrombocytopenia (platelet count < 50,000 cells/mm³), history of bleeding diathesis or coagulopathy, or hypercoagulable states. 4. Ongoing sepsis, including active endocarditis. 5. Any percutaneous coronary or peripheral interventional procedure with a bare metal stent within 30 days prior to randomisation, or drug‐eluting stent performed within 180 days prior to randomisation. 6. Multivessel coronary artery disease with a Synergy between Percutaneous Coronary Intervention with Taxus and Cardiac Surgery (SYNTAX) score > 22 and/or unprotected left main coronary artery. 7. Symptomatic carotid or vertebral artery disease or successful treatment of carotid stenosis within 10 weeks of heart team assessment. 8. Cardiogenic shock manifested by low cardiac output, vasopressor dependence, or mechanical haemodynamic support. 9. Recent (within 2 months of heart team assessment) cerebrovascular accident or transient ischaemic attack. 10. Gastrointestinal bleeding that would preclude anticoagulation. 11. Patient refuses a blood transfusion. 12. Severe dementia (resulting in either inability to provide informed consent for the trial/procedure, prevents independent lifestyle outside of a chronic care facility, or will fundamentally complicate rehabilitation from the procedure or compliance with follow‐up visits). 13. Estimated life expectancy of less than 24 months due to associated non‐cardiac co‐morbid conditions. 14. Other medical, social, or psychological conditions that in the opinion of the investigator precludes the patient from appropriate consent or adherence to the protocol‐required follow‐up exams. 15. Currently participating in an investigational drug or another device trial (excluding registries). 16. Evidence of an acute myocardial infarction ≤ 30 days before the trial procedure due to unstable coronary artery disease (World Health Organization criteria). 17. Need for emergency surgery for any reason. 18. Patient is pregnant or breast feeding. 19. Patient is less than legal age of consent, legally incompetent, or otherwise vulnerable. Anatomical exclusion criteria: 20. Pre‐existing prosthetic heart valve in any position. 21. Severe mitral regurgitation amenable to surgical replacement or repair. 22. Severe tricuspid regurgitation amenable to surgical replacement or repair. 23. Moderate or severe mitral stenosis amenable to surgical replacement or repair. 24. Hypertrophic obstructive cardiomyopathy with left ventricular outflow gradient. Timings of outcome reporting: 30 days, 1 year, 2 years, 5 years, and 6 years. 25. Bicuspid aortic valve verified by echocardiography, multidetector computed tomography or magnetic resonance imaging. 26. Prohibitive left ventricular outflow tract calcification. 27. Sinus of Valsalva diameter unsuitable for placement of the self‐expanding bioprosthesis. 28. Aortic annulus diameter of < 18 or > 30 mm. 29. Significant aortopathy requiring ascending aortic replacement. For transfemoral or transaxillary (subclavian) access: 30. Access vessel mean diameter < 5.0 mm for Evolut 23R, 26R, or 29R mm transcatheter aortic valves, or access vessel mean diameter < 5.5 mm for Evolut 34R mm or Evolut PRO transcatheter aortic valves. However, for transaxillary (subclavian) access in patients with a patent left internal mammary artery graft access vessel mean diameter < 5.5 mm for Evolut 23R, 26R, 29R mm transcatheter aortic valves, or access vessel mean diameter < 6.0 mm for the CoreValve 31 mm, Evolut R 34R or Evolut PRO transcatheter aortic valves. |
|
| Interventions | 1. Transcatheter aortic valve implantation (n = 734) 2. Surgical aortic valve replacement (n = 734) |
|
| Outcomes | The primary safety and effectiveness end point was a composite of death from any cause or disabling stroke at 24 months. Disabling stroke was defined by a score on the modified Rankin scale of 2 or more (with scores ranging from 0 (no symptoms) to 6 (death)) at 90 days and an increase of at least 1 category from baseline (i.e. before the stroke). There were 7 prespecified secondary end points that were tested hierarchically for either non‐inferiority or superiority. Additional secondary safety end points included a composite of death, disabling stroke, life‐threatening bleeding, major vascular complication, or stage 2 or 3 acute kidney injury at 30 days; and prosthetic valve endocarditis, prosthetic valve thrombosis, prosthetic valve dysfunction requiring a repeat procedure, stroke, and life‐threatening bleeding at 12 months. The full list of secondary end points as listed in the Methods section in the Supplementary Appendix: 1. Transvalvular mean gradient at 1 year (non‐inferiority). 2. Effective orifice area at 1 year (non‐inferiority). 3. Change in NYHA classification from baseline to 1 year (non‐inferiority). 4. Change in KCCQ score from baseline to 1 year (non‐inferiority). 5. Transvalvular mean gradient at 1 year (superiority). 6. Effective orifice area at 1 year (superiority). 7. Change in KCCQ score from baseline to 30 days (superiority). Secondary safety end points: ‐ The rate of the composite of death, disabling stroke, life‐threatening bleed, major vascular complications, or stage II or III acute kidney injury at 30 days ‐ The rate of new permanent pacemaker implantation at 30 days ‐ The rate of prosthetic valve endocarditis at 1 year ‐ The rate of prosthetic valve thrombosis at 1 year ‐ The rate of all stroke (disabling and non‐disabling) at 1 year ‐ The rate of life‐threatening bleeding at 1 year ‐ The rate of valve‐related dysfunction requiring repeat procedure at 1 year Secondary effectiveness end points: ‐ The rate of valve‐related dysfunction, defined as moderate or severe prosthetic valve stenosis, or moderate or severe prosthetic regurgitation at 1 year (per Valve Academic Research Consortium (VARC) II) ‐ Quality of life as assessed by Kansas City Cardiomyopathy Questionnaire (KCCQ) at 30 days and 1 year ‐ The rate of repeat hospitalisation for aortic valve disease at 1 year ‐ VARC II Device Success: o Absence of procedural mortality AND o Correct positioning of a single prosthetic heart valve into the proper anatomical location AND intended performance of the prosthetic heart valve, defined as the absence of patient‐prosthesis mismatch AND mean aortic valve gradient less than 20 mmHg (or peak velocity < 3 m/sec) AND absence of moderate or severe prosthetic valve regurgitation. ‐ Haemodynamic performance metrics by Doppler echocardiography o Mean aortic gradient at 1 year o Effective orifice area at 1 year o Degree of total, peri, and transvalvular prosthetic regurgitation at 1 year ‐ New York Heart Association (NYHA) functional classification at 1 year ‐ Health‐related quality of life at 1 year as assessed by EQ‐5D survey instrument |
|
| Notes | Ethical approval: the trial was conducted in compliance with the International Conference on Harmonisation and the Declaration of Helsinki. Local institutional review boards or medical ethics committees approved the protocol. Funding/support: Medtronic and Paradigm Biostatistics. Medtronic funded the trial and developed the protocol in collaboration with the executive committee. Medtronic was responsible for site selection, data monitoring, and trial management. Paradigm Biostatistics performed the Bayesian end‐point comparisons; an independent statistical consultant validated all end‐point analyses. Conflict of interest disclosure: attached as separate forms with full text of the trial. Correspondence notes: we sought to obtain ITT data on the following outcomes: atrial fibrillation, bleeding, AKI, and PPM implantation. We also attempted to clarify the exact numbers of all‐cause mortality and check whether LOS was measured but not reported. We were not able to obtain the mentioned information. LOS was not reported in the published protocol. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Randomization was performed in a 1:1 ratio, with variable block sizes, with an electronic randomisation system. Randomisation was stratified by site and the need for coronary artery revascularisation. |
| Allocation concealment (selection bias) | Unclear risk | Insufficient information to permit judgement of ‘low risk’ or ‘high risk’. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Although the study was unblinded due to the nature of the procedures, we deemed the risk of bias for this domain low since operative/interventional outcomes are unlikely to be influenced by lack of blinding of participants and personnel. |
| Blinding of outcome assessment (detection bias) Objective outcomes | Low risk | Although the study was unblinded due to the nature of the procedures, we deemed the risk of bias for this domain low due to the objective nature of the outcomes that are unlikely to be affected by lack of blinding. |
| Blinding of outcome assessment (detection bias): subjective outcomes Subjective outcomes | High risk | Detection bias for potentially subjective outcomes was deemed high since these outcomes may potentially be influenced by lack of blinding. |
| Incomplete outcome data (attrition bias): short‐term outcomes Short term outcomes | Low risk | Although there was an imbalance between the number of withdrawals across both groups (TAVI: 12 and SAVR: 53) in the intention‐to‐treat population, the study does not report final percentage follow‐up for short‐term outcomes as main analyses are reported only as percentages without actual number of participants. However, based on New York Heart Association outcome reporting at 30 days (Figure S4 in supplementary appendix), there was more loss to follow‐up in the SAVR group, but the difference remained less than 10%. |
| Incomplete outcome data (attrition bias) Long term outcomes | High risk | Quote: "12‐month follow‐up was available for 432 patients in the TAVI group and 352 in the surgery group; 24‐month follow‐up was available for 72 patients in the TAVI group and 65 patients in the surgery group. The median follow‐up time in each group was 12.2 months." Comment: the proportion of missing outcomes compared with observed event risk enough to induce clinically relevant bias in intervention effect estimate. Also, reason for missing outcome data likely to be related to true outcome, with imbalance in numbers and reasons for missing data across intervention groups. |
| Selective reporting (reporting bias) | Low risk | Study protocol available and all study’s prespecified (primary and secondary) outcomes of interest in review were reported in prespecified way. |
| Other bias | Low risk | None noted. |
NOTION 2015.
| Methods | Study design: the NOTION trial was an investigator‐initiated, multicentre, randomised, non‐blinded, superiority trial conducted at 2 centres in Denmark and 1 in Sweden. Patients were randomised in a 1:1 ratio to treatment with TAVI or SAVR. Randomisation was performed at the Copenhagen Trial Unit and was stratified according to trial site, age (70 to 74 years or older), and history of CAD (yes or no). Total study duration: 5 years (at least). Number of study centres and location: 3 centres in Denmark and Sweden. Study setting: inpatient. Date of study: December 2009 to April 2013. However, the study is still ongoing with an anticipated end date of April 2023. |
|
| Participants | A total of 280 patients were randomly assigned to TAVI using a self‐expanding prosthesis versus SAVR and were followed for 5 years. Patients ≧ 70 years of age with severe degenerative aortic valve stenosis referred for SAVR and also candidates for TAVI were eligible for inclusion regardless of their predicted risk of death after surgery. A heart team consisting of at least an imaging cardiologist, an interventional cardiologist, and a cardiac surgeon evaluated all patients. N‐randomised: 280; 145 TAVI and 135 SAVR N‐lost to follow‐up/withdrawn: 5; 3 TAVI and 2 SAVR + cross‐overs As‐treated population: 276; 142 TAVI and 134 SAVR N‐analysed: 274; 139 TAVI and 135 SAVR Mean age: 79.1 ± 4.8 years Gender: 53.2% males Surgical risk score at baseline: The overall STS‐PROM score was 3.0% ± 1.7% STS‐PROM: TAVI: 2.9% ± 1.6% SAVR 3.1% ± 1.7% EuroSCORE II TAVI: 1.9% ± 1.2% SAVR 2.0% ± 1.3% Inclusion criteria: Severe aortic valve stenosis was defined as an effective orifice area < 1 cm² or indexed for body surface area < 0.6 cm²/m² and a mean aortic valve gradient > 40 mmHg or peak systolic velocity > 4 m/s. Symptomatic patients had to have dyspnoea, New York Heart Association (NYHA) functional class II or higher, angina pectoris, or cardiac syncope to qualify for the trial. Asymptomatic patients could be included if they had left ventricular posterior wall thickness ≧ 17 mm, decreasing left ventricular ejection fraction, or new‐onset AF. To be eligible, patients were expected to survive for more than 1 year. Trial eligibility, choice of prosthesis size, and arterial access route were based on transthoracic and transoesophageal echocardiograms and an aorto‐iliofemoral angiogram and were confirmed by the primary interventional and surgical investigator at each site. Supplemental computed tomography (CT) studies were performed in patients with difficult aortic annular measurements or peripheral arterial disease. Exclusion criteria: Patients were excluded if they had another severe heart valve disease or coronary artery disease (CAD) requiring intervention. Other important exclusion criteria were previous cardiac surgery, myocardial infarction (MI) or stroke within 30 days, severe renal failure requiring dialysis, or pulmonary failure with a forced expiratory volume within 1 second or diffusion capacity < 40% of expected. |
|
| Interventions | 1. Transcatheter aortic valve replacement (n = 145) 2. Surgical aortic valve replacement (n = 135) |
|
| Outcomes | The primary outcome was the composite rate of all‐cause death, stroke, or MI 1 year post‐procedure. Exploratory outcomes were as follows: the rate of individual components of the composite outcome; the rate of cardiovascular death; prosthesis re‐intervention; cardiogenic shock; valve endocarditis; conduction abnormalities requiring permanent pacemaker; atrial fibrillation or flutter; and vascular, renal, and bleeding complications after 1 and 12 months. Clinical improvement was assessed according to NYHA functional classification. Echocardiographic outcomes included aortic valve effective orifice area, mean pressure gradient, and degree of total aortic valve regurgitation (graded as none/trace, mild, moderate, and severe) at 3 and 12 months. All outcomes were defined according to Valve Academic Research Consortium‐2 definitions. Timing of outcome reporting: 30 days, 1 year, 2 years, 5 years, and 6 years. |
|
| Notes | Ethical approval: The regional ethical review board at each site approved the trial protocol, and the trial was conducted according to the principles of the Declaration of Helsinki. All patients provided written informed consent. The investigators collected and stored all data, which were fully monitored by an independent monitoring unit. An independent clinical events committee adjudicated all clinical events. An independent statistician confirmed the statistical analysis. Funding/support: the Danish Heart Foundation provided financial support (grants: 09‐10‐AR76‐A2733‐25400, 12‐04‐R90‐A3879‐22733 and 13‐04‐R94‐A4473‐ 22762). Dr Anh Thuc Ngo, principal medical writer Molly Schiltgen, Medtronic, Inc. assisted with manuscript drafting. Conflict of interest disclosure: Dr. Steinbrüchel has received research contracts from Medtronic, Inc. Dr. Ihlemann has received speaker fees from Medtronic, Inc. Dr. Chang is an employee of Medtronic, Inc. Dr. Franzen has received research contracts from St. Jude Medical, Inc. Dr. Clemmensen has received research contracts and speaker fees from Medtronic, Inc. Dr. Hansen has received speaker fees from Medtronic, Inc. Dr. Olsen has received research contracts from Medtronic, Inc. and St. Jude Medical, Inc. Dr. Søndergaard is a proctor for Medtronic, Inc.; has received research contracts from Medtronic, Inc. and St. Jude Medical, Inc.; and has received speaker fees from Medtronic, Inc. All other authors have reported that they have no relationships relevant to the contents of this paper to disclose. Correspondence notes: We sought to obtain data on rehospitalisation outcome, with no success. The outcome was not part of the published protocol, although it was narratively mentioned as no difference between groups in the final report. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Randomisation is central at the Copenhagen Trial Unit, which has generated the allocation sequence in permuted blocks with unknown block size for the investigators. |
| Allocation concealment (selection bias) | Low risk | Randomisation is central at the Copenhagen Trial Unit, which has generated the allocation sequence in permuted blocks with unknown block size for the investigators. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Although the study was unblinded due to the nature of the procedures, we deemed the risk of bias for this domain low since operative/interventional outcomes are unlikely to be influenced by lack of blinding of participants and personnel. |
| Blinding of outcome assessment (detection bias) Objective outcomes | Low risk | Although the study was unblinded due to the nature of the procedures, we deemed the risk of bias for this domain low due to the objective nature of the outcomes that are unlikely to be affected by lack of blinding. |
| Blinding of outcome assessment (detection bias): subjective outcomes Subjective outcomes | High risk | Detection bias for potentially subjective outcomes was deemed high since these outcomes may potentially be influenced by lack of blinding. |
| Incomplete outcome data (attrition bias): short‐term outcomes Short term outcomes | Low risk | Study reported no loss to follow‐up or withdrawals. |
| Incomplete outcome data (attrition bias) Long term outcomes | Low risk | Study reported no loss to follow‐up or withdrawals. |
| Selective reporting (reporting bias) | Low risk | Study protocol available and all study’s prespecified (primary and secondary) outcomes of interest in review were reported in prespecified way. |
| Other bias | Low risk | None noted. |
PARTNER 3 2019.
| Methods | Study design: the PARTNER 3 trial was a multicentre, randomised trial in which TAVI with transfemoral placement of a third‐generation balloon‐expandable valve was compared with standard surgical aortic‐valve replacement in patients with severe aortic stenosis and a low risk of death with surgery. Total study duration: the trial aims at recruiting 1328 participants and they plan to report the outcomes every year for 10 years. This publication reports the first year after recruiting 1000 participants. Number of study centres and location: 71 sites in the USA, Canada, Australia, New Zealand, and Japan. Study setting: inpatient. Date of study: March 2016 to October 2017 (for the current report). However, the study is still recruiting and ongoing with an anticipated end date of March 2027. |
|
| Participants | 1000 patients were enrolled at 71 sites; 979 of the patients were from the USA, 8 from Canada, 7 from Australia or New Zealand, and 6 from Japan. The patients were randomly assigned to undergo either TAVI (503 patients) or surgery (497 patients). The assigned procedure was performed in 950 patients (496 in the TAVI group and 454 in the surgery group), who composed the as‐treated population, and the intended valve was implanted in 948. Among the patients who did not undergo the assigned procedure (7 in the TAVI group and 43 in the surgery group), the most common reason was withdrawal from the trial (in 41 patients), mainly owing to the decision not to undergo surgery or the preference to undergo surgery at a non‐trial site. N‐randomised: 1000; TAVI (503 patients) and surgery (497 patients). N‐lost to follow‐up/withdrawn: (7 in the TAVI group and 43 in the surgery group). As‐treated population: TAVI (496 patient) and surgery (454 patients). N‐analysed: 950 TAVI (496 patient) and surgery (454 patients). Mean age: 73 years. Gender: 69.3% males. Surgical risk score at baseline: STS‐PROM: TAVI: 1.9% ± 0.7% SAVR: 1.9% ± 0.6% EuroSCORE II score: TAVI: 1.5% ± 1.2% SAVR: 1.5% ± 0.9% Inclusion criteria: All study participants must have met the following inclusion criteria: 1. Severe, calcific aortic stenosis meeting the following criteria: • AVA ≤ 1.0 cm² or AVA index ≤ 0.6 cm²/m² • Jet velocity ≥ 4.0 m/s or mean gradient ≥ 40 mmHg AND • NYHA Functional Class ≥ 2 OR • Exercise tolerance test that demonstrates a limited exercise capacity, abnormal BP response, or arrhythmia OR • Asymptomatic with LVEF < 50% Note: qualifying echo must be within the 90 days prior to randomisation. 2. Heart team agrees the patient has a low risk of operative mortality and an STS < 4. 3. The study patient has been informed of the nature of the study, agrees to its provisions and has provided written informed consent as approved by the Institutional Review Board (IRB)/Ethics Committee (EC) of the respective clinical site. Exclusion criteria: Candidates were excluded from the study if any of the following conditions were present: 1. Native aortic annulus size unsuitable for sizes 20, 23, 26, or 29 mm THV based on 3D imaging analysis 2. Iliofemoral vessel characteristics that would preclude safe passage of the introducer sheath 3. Evidence of an acute myocardial infarction ≤ 1 month (30 days) before randomisation 4. Aortic valve is unicuspid, bicuspid, or non‐calcified 5. Severe aortic regurgitation (> 3+) 6. Severe mitral regurgitation (> 3+) or ≥ moderate stenosis 7. Pre‐existing mechanical or bioprosthetic valve in any position. (Note: mitral ring is not an exclusion). 8. Complex coronary artery disease: a. Unprotected left main coronary artery b. Syntax score > 32 (in the absence of prior revascularisation) c. Heart team assessment that optimal revascularisation cannot be performed 9. Symptomatic carotid or vertebral artery disease or successful treatment of carotid stenosis within 30 days of randomisation 10. Leukopenia (WBC < 3000 cell/mL), anaemia (Hgb < 9 G/dL), Thrombocytopenia (Plt < 50,000 cell/mL), history of bleeding diathesis or coagulopathy, or hypercoagulable states 11. Haemodynamic or respiratory instability requiring inotropic support, mechanical ventilation or mechanical heart assistance within 30 days of randomisation 12. Hypertrophic cardiomyopathy with obstruction (HOCM) 13. Ventricular dysfunction with LVEF < 30% 14. Cardiac imaging (echo, CT, and/or MRI) evidence of intracardiac mass, thrombus or vegetation 15. Inability to tolerate, or condition precluding treatment with, antithrombotic/anticoagulation therapy during or after the valve implant procedure 16. Stroke or transient ischaemic attack (TIA) within 90 days of randomisation 17. Renal insufficiency (eGFR < 30 ml/min per the Cockcroft‐Gault formula) and/or renal replacement therapy at the time of screening. 18. Active bacterial endocarditis within 180 days of randomisation 19. Severe lung disease (FEV1 < 50% predicted) or currently on home oxygen 20. Severe pulmonary hypertension (e.g. PA systolic pressure ≥ 2/3 systemic pressure) 21. History of cirrhosis or any active liver disease 22. Significant frailty as determined by the heart team (after objective assessment of frailty parameters) 23. Significant abdominal or thoracic aortic disease (such as porcelain aorta, aneurysm, severe calcification, aortic coarctation, etc.) that would preclude safe passage of the delivery system or cannulation and aortotomy for surgical AVR 24. Hostile chest or conditions or complications from prior surgery that would preclude safe reoperation (i.e. mediastinitis, radiation damage, abnormal chest wall, adhesion of aorta or IMA to sternum, etc.) 25. Patient refuses blood products 26. BMI > 50 kg/m² 27. Estimated life expectancy < 24 months 28. Absolute contraindications or allergy to iodinated contrast that cannot be adequately treated with pre‐medication 29. Immobility that would prevent completion of study procedures (e.g. 6‐minute walk tests, etc.) 30. Patient is not a candidate for both arms of the study (not applicable to single‐arm registries) 31. Currently participating in an investigational drug or another device study. (Note: Trials requiring extended follow‐up for products that were investigational, but have since become commercially available, are not considered investigational trials. Observational studies are not considered exclusionary.) |
|
| Interventions | 1. Transcatheter aortic valve implantation (n = 503) 2. Surgical aortic valve replacement (n = 497) |
|
| Outcomes | The primary endpoint is the composite of all‐cause mortality, all stroke, and rehospitalisation (valve‐related or procedure‐related and including heart failure) at 1 year post‐procedure. Secondary endpoints: ‐ New onset atrial fibrillation at 30 days ‐ Length of index hospitalisation ‐ All‐cause death, all stroke, rehospitalisation at 1 year (primary endpoint – superiority testing) ‐ Death, KCCQ < 45 or KCCQ decrease from baseline ≥ 10 points at 30 days ‐ Death or stroke at 30 days ‐ Stroke at 30 days Other safety and effectiveness endpoints (not adjusted for multiple comparisons) Safety endpoints: • Mortality (all‐cause & cardiovascular) at 30 days and 1 year • Stroke (disabling & non‐disabling) at 30 days and 1 year • Death or stroke at 1 year • Death or disabling stroke at 30 days and 1 year • Vascular complications (major) at 30 days and 1 year • Bleeding complications (life threatening/disabling, or major) at 30 days and 1 year • Myocardial infarction at 30 days and 1 year • Acute kidney injury at 30 days • Requirement for renal replacement therapy at 1 year • New permanent pacemaker implantation resulting from new or worsened conduction disturbances at 30 days and 1 year • Coronary obstruction requiring intervention at 30 days and 1 year • New onset atrial fibrillation at 30 days and 1 year • Rehospitalisation (valve‐related or procedure‐related and including heart failure) at 30 days and 1 year. Effectiveness endpoints: • New York Heart Association class at 30 days and 1 year • Haemodynamic evaluations by echocardiography at 30 days and 1 year (e.g. aortic valve stenosis and aortic valve regurgitation (paravalvular & central)) • ICU days • Discharge location, ranked from 1 to 6 according to the level of required care assistance as follows: a) Discharge to Home or Self Care (Routine Discharge) b) Discharged/Transferred to Home Under Care of Organized Home Health Service Organization c) Discharged/Transferred to a Skilled Nursing Facility (SNF) or Inpatient Rehabilitation Facility d) Discharged/Transferred to Another Hospital e) Discharged/Transferred to a Hospice f) Expired (this code is used only when the patient dies) • Days alive and out of hospital (DAOH) at 1 year, computed using the following methodology: a) When computing days alive, the day of a subject’s death is excluded regardless of the time of day of the subject’s death. b) Beginning at Day 0, each day that the subject is alive is classified as a day in hospital or not; days of admission and discharge are counted as days in hospital. c) DAOH is defined to be the number of days for which the subject is not dead or in hospital, expressed as an integer. d) The maximum value DAOH can ever attain for a given subject is 366. DAOH would have this value if a subject had never been hospitalised, and was known to be alive at the 1‐year time point (Day 366) e) A given subject’s potential DAOH is 366 if the patient is alive more than 366 days post procedure, or died anytime. After the implant; the potential DAOH is the days between implant and termination if the patient terminated (withdrew or lost to follow‐up) before 366 days post procedure. f) To calculate Adjusted DAOH to 1 year, censoring must be accommodated Adjusted DAOH = DAOH * (366/potential DAOH) Adjusted DAOH is rounded to the nearest integer. This rounded value is used in all calculations. • 6‐minute walk test at 30 days and 1 year • Health status as evaluated by quality of life (QoL) questionnaires (including change from baseline value) a. KCCQ at 30 days and 1 year b. EQ‐5D‐5L at 30 days and 1 year c. SF‐36 at 30 days and 1 year • Composite of death, KCCQ < 45 or KCCQ decrease ≥ 10 points from baseline to 30 days • Structural valve deterioration (SVD) at 1 year VARC2 – EACTS – EACPI – EACVI definition: a) Structural and haemodynamic valve deterioration (moderate or severe prosthetic valve stenosis, AND/ OR moderate or severe transprosthetic valve regurgitation), AND b) Requiring repeat procedure (TAVI or surgery) Timings of outcome reporting: 30 days and 1 year. |
|
| Notes | Ethical approval: the protocol was approved by the institutional review board at each site. All the patients provided written informed consent. Funding/support: Edwards Lifesciences. The sponsor funded all trial‐related activities and participated in site selection, data collection and monitoring, and statistical analysis. The trial protocol was designed by the trial sponsor. Conflict of interest disclosure: Dr. Mack reports receiving consulting fees from Gore, serving as a trial coprimary investigator for Edwards Lifesciences and Abbott, and serving as a study chair for Medtronic; Dr. Leon reports receiving grant support, paid to his institution, and advisory board fees from Medtronic and Abbott, grant support, paid to his institution, advisory board fees, and equity from Boston Scientific, advisory board fees from Gore, and advisory board fees from Meril Life Sciences; Dr. Thourani reports receiving grant support and serving as an advisor for Edwards Lifesciences; Dr. Makkar reports receiving grant support from Abbott and Edwards Lifesciences; Dr. Kodali reports receiving equity from BioTrace Medical,Dura Biotech, and Thubrikar Aortic Valve, grant support from Medtronic and Boston Scientific, grant support and consulting fees from Abbott Vascular, and consulting fees from Claret Medical, Admedus, and Meril Life Sciences; Dr. Russo reports receiving consulting fees, lecture fees, and fees for serving as a proctor from Edwards Lifesciences, consulting fees and fees for serving as a proctor from Abbott, and consulting fees from Boston Scientific; Dr. Malaisrie reports receiving consulting fees from Medtronic and lecture fees from Abbott; Dr. Cohen reports receiving grant support, paid to his institution, and consulting fees from Edwards Lifesciences and Medtronic, and grant support, paid to his institution, from Boston Scientific and Abbott Vascular; Dr. Leipsic reports receiving grant support from Abbott and Medtronic, and consulting fees and stock options from Circle Cardiovascular Imaging; Dr. Hahn reports receiving lecture fees and consulting fees from Abbott Vascular and Siemens Healthineers, lecture fees from Boston Scientific and Bayliss, and consulting fees from Edwards Lifesciences, Philips Healthcare, 3Mensio, Medtronic, and Navigate; Dr. Blanke reports receiving consulting fees from Edwards Lifesciences, Tendyne (Abbott), Circle Cardiovascular Imaging, Neovasc, and Gore; Dr. McCabe reports receiving consulting fees from Edwards Lifesciences; Dr. Babaliaros reports receiving lecture fees and consulting fees from Edwards Lifesciences and Abbott; Dr. Goldman reports receiving advisory board fees from Edwards Lifesciences; Dr. Szeto reports receiving lecture fees and serving as an investigator for Edwards Lifesciences; Dr. Genereux reports receiving consulting fees and advisory board fees from Abbott Vascular, Boston Scientific, Cardiovascular Solutions, and Cordis, consulting fees and fees for serving as a proctor from Edwards Lifesciences, and consulting fees from Medtronic, Saranas, Pi‐Cardia, and Sig.Num; Dr. Alu reports receiving consulting fees from Claret Medical and Cardiac Dimensions; and Dr. Webb reports receiving consulting fees and fees for serving as a proctor from Edwards Lifesciences. No other potential conflict of interest relevant to this article was reported. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Randomisation was performed by an electronic randomisation system, BALANCE, with randomisation list generated using SAS. Randomization was stratified by site and used block sizes of 4. |
| Allocation concealment (selection bias) | Low risk | Randomization occurred centrally. To randomise a patient, the investigative site entered the subject into the designated electronic system and obtained the treatment assignment (TAVI or SAVR). |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Although the study was unblinded due to the nature of the procedures, we deemed the risk of bias for this domain low since operative/interventional outcomes are unlikely to be influenced by lack of blinding of participants and personnel. |
| Blinding of outcome assessment (detection bias) Objective outcomes | Low risk | Although the study was unblinded due to the nature of the procedures, we deemed the risk of bias for this domain low due to the objective nature of the outcomes that are unlikely to be affected by lack of blinding. |
| Blinding of outcome assessment (detection bias): subjective outcomes Subjective outcomes | High risk | We deemed detection bias for potentially subjective outcomes to be high since these outcomes may potentially be influenced by lack of blinding. |
| Incomplete outcome data (attrition bias): short‐term outcomes Short term outcomes | Low risk | Although the number of participants who did not receive the allocated intervention in the intention‐to‐treat population was unbalanced between both groups (TAVI: 7 and SAVR: 43), the difference was less than 10%. Additionally, the study reported an overall 99.6% patient follow‐up at 30 days for both groups in the as‐treated population, with only 4 participants withdrawn from the SAVR group and none from the TAVI group. |
| Incomplete outcome data (attrition bias) Long term outcomes | Low risk | The study reported an overall 98.4% of patients were available for primary endpoint analysis at 1 year. |
| Selective reporting (reporting bias) | Low risk | Study protocol available and all study’s prespecified (primary and secondary) outcomes of interest in review were reported in prespecified way. |
| Other bias | Low risk | None noted. |
STACCATO 2012.
| Methods | Study design: the study was planned as an academic prospective multicentre clinical trial in the Nordic region with a 1:1 randomisation of a total of 200 patients to a‐TAVI vs. SAVR. Total study duration: prematurely terminated before planned follow‐up. Number of study centres and location: 2 centres in Denmark. Study setting: inpatient. Date of study: November 2008 to May 2011. |
|
| Participants | A total of 72 patients were randomised. 2 patients were excluded after randomisation; 1 patient declined participation, and the other unexpectedly met the exclusion criteria of impaired pulmonary function. Thus, the study population consisted of 34 patients in the transapical transcatheter aortic valve implantation (a‐TAVI) and 36 patients in the SAVR group. N‐randomised: 72 N‐lost to follow‐up/withdrawn: 2 As treated population: 70 (34 a‐TAVI) and (36 SAVR) N‐analysed: 70 (34 a‐TAVI) and (36 SAVR) Mean age: 80 ± 3.6 (a‐TAVI) and 82 ± 4.4 (SAVR) Gender: 30% males. Surgical risk score at baseline (STS‐PROM): TAVI: 3.1% ± 1.5% SAVR: 3.4% ± 1.2% Inclusion criteria: Significant valvular aortic stenosis (valve area < 1 cm²), age initially ≥ 70, later ≥ 75 yrs; condition accessible both by SAVR and a‐TAVI; expected survival > 1 year following successful treatment; and patient acceptance of participation in the study as well as in the scheduled follow‐up investigations. We used age as our major criterion of inclusion, because age is a simple and well‐defined parameter closely related to surgical risk. Exclusion criteria: Coronary artery disease to be treated by percutaneous coronary intervention (PCI) or coronary artery bypass grafting (CABG); previous myocardial infarction, and previous PCI within 12 months. Previous heart surgery became a criterion of exclusion during the study. The need for other heart surgery (i.e. mitral or tricuspid valve surgery), emergency surgery (within 24 hours of indication for surgery), unstable cardiac condition (requiring an assist device, inotropes or i.v. nitrates in operating room), ongoing infection requiring antibiotics, stroke within 1 month, reduced pulmonary function (FEV1 < 1 l or < 40% of expected), renal failure to be treated by haemodialysis, allergy to acetylsalicylic acid, clopidogrel, prasugrel or x‐ray contrast material. |
|
| Interventions | 1. Transapical transcatheter aortic valve implantation (a‐TAVI) (n = 24) 2. Surgical aortic valve implantation (n = 36) |
|
| Outcomes | The primary endpoint was the composite of 30‐day all‐cause mortality, major stroke, and renal failure requiring dialysis. Secondary endpoints included: all‐cause death, cardiac death, stroke, myocardial infarction, New York Heart Association (NYHA) function class, SF‐36 composite physical and mental functional scores, echocardiographic parameters (aortic valve area, peak aortic valve gradient, aortic valve leakage, left ventricular ejection fraction), duration of hospital stay, operation for bleeding, and permanent pacemaker treatment. For endpoint definitions, we used the Valve Academic Research Consortium recommendations. All endpoints were adjudicated by an independent endpoint committee. Timing of outcome reporting: 30 days and 3 months. |
|
| Notes | Ethical approval: the study complied with the Declaration of Helsinki and was approved by the ethics committee of the Region of Midtjylland. All patients provided written, informed consent before participation in the trial. Funding/support: the study was an academic study, designed and carried out by the involved cardiac surgeons, cardiologists and anaesthesiologists at Aarhus University Hospital, and Odense University Hospital, and primarily funded by the participating hospitals. Further, there was a study grant from the Danish Heart Association. There was no industry involvement. Conflict of interest disclosures: KE Klaaborg and L Thuesen are part‐time physician proctors for Edwards Lifesciences. The other authors have no conflict of interest to declare. The present study was conducted without any relationship to industry. Correspondence notes: we sought to obtain information on our outcomes of interest (rehospitalisation, atrial fibrillation and PPM implantation) to check whether they were measured but not reported. No contact information was listed for the investigators, however; nor was there was a published protocol. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | The 1:1 randomisation between a‐TAVI and SAVR was implemented using the web‐based clinical trials support system TrialPartner (PCI Research, Aarhus University Hospital, Skejby, Denmark). TrialPartner permits, with a personal log‐in, 24‐hour randomisation. Data was entered in the electronic case report form of TrialPartner, a secure server‐based system with security that exceeds the demands and guidelines by the National Data Protection Agency. |
| Allocation concealment (selection bias) | Unclear risk | Insufficient information to permit judgement of ‘low risk’ or ‘high risk’. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Although it is unclear whether this was a blinded study, we deemed the risk of bias for this domain low since operative/interventional outcomes are unlikely to be influenced by lack of blinding of participants and personnel. |
| Blinding of outcome assessment (detection bias) Objective outcomes | Low risk | Although it is unclear whether this was a blinded study, we deemed the risk of bias for this domain low due to the objective nature of the outcomes that are unlikely to be affected by lack of blinding. |
| Blinding of outcome assessment (detection bias): subjective outcomes Subjective outcomes | High risk | We deemed detection bias for potentially subjective outcomes to be high since these outcomes may potentially be influenced by lack of blinding. |
| Incomplete outcome data (attrition bias): short‐term outcomes Short term outcomes | Low risk | Study reported no loss to follow‐up or withdrawals. |
| Incomplete outcome data (attrition bias) Long term outcomes | Low risk | Study reported no loss to follow‐up or withdrawals. |
| Selective reporting (reporting bias) | Low risk | No protocol available, but it is clear that the published report includes all expected outcomes. |
| Other bias | High risk | The STACCATO trial was prematurely terminated because of an overall excess of adverse events in transcatheter treated patients in comparison with patients receiving surgical aortic valve replacement. |
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| NCT02628899 | Non‐randomised trial. |
| NCT02838199 | The study status on ClinicalTrials.gov is "withdrawn". |
| SURTAVI 2017 | All study population was deemed intermediate risk as per correspondence with Dr. Reardon, on behalf of SURTAVI investigators, who stated: "SURTAVI patients were all intermediate risk patients. There are some who have a STS PROM that is < 4, but all were screened both by the local heart team and a national screening committee and confirmed to have additional factors that led to the designation of intermediate risk. The definition of intermediate risk in SURTAVI was a predicted surgical mortality of 3% to 8%. They are not appropriate to consider in low risk." |
Characteristics of studies awaiting assessment [ordered by study ID]
NCT03112980.
| Methods | Randomised, unblinded controlled trial |
| Participants | Ages eligible for study: 70 years to 85 years (older adult)
Sexes eligible for study: all
Accepts healthy volunteers: No Inclusion criteria: 1. Heart team consensus that TAVI and SAVR are both medically justified and advisable based on: * degenerative aortic valve stenosis with echocardiographically derived criteria: * mean gradient >40 mmHg or * jet velocity greater than 4.0 m/s or * aortic valve area (AVA) of < 1.0 cm² (indexed effective orifice area < 0.6 cm²/m²). * patient is symptomatic from his/her aortic valve stenosis * New York Heart Association Functional Class ≥ II or * angina pectoris or * syncope. * patient is classified as low to intermediate operative risk as assessed by the local heart team according to the variables outlined in the 2017 ESC/EACTS Guidelines for the management of valvular heart disease, taking into account cardiac and extracardiac patient characteristics and established risk scores (e.g. STS‐PROM, EuroSCORE). * a transfemoral or alternative (e.g. transapical, transaortic, transaxillary) access for TAVI seems feasible. Centres should follow a "transfemoral first" strategy for the primary route of access; however, other routes of access are also allowed, as decided by local heart team consensus. 2. Patient has provided written informed consent to participate in the trial. 3. Ability of the patient to understand the patient information and to personally sign and date the informed consent to participate in the study, before performing any study‐related procedures. 4. The patient agrees to undergo SAVR, if randomised to control treatment. 5. The patient and the treating physician agree that the patient will return for all required post‐procedure follow‐up visits. 6. Patients aged 70 to 85 years. 7. Male patients or females who are postmenopausal defined as no menses for 12 months without an alternative medical cause. Exclusion criteria: 1. Aortic valve is a congenital unicuspid or congenital bicuspid valve, or is non‐calcified. 2. Untreated clinically significant coronary artery disease considered a contraindication to an isolated aortic valve procedure (TAVI or SAVR) according to heart team consensus. 3. Previous cardiac surgery. 4. Any percutaneous coronary intervention performed within 1 month prior to the study procedure. 5. Untreated severe mitral or tricuspid regurgitation. 6. Untreated severe mitral stenosis. 7. Haemodynamic instability requiring inotropic support or mechanical circulatory support. 8. Ischemic stroke or intracranial bleeding within 1 month. 9. Severe ventricular dysfunction with left ventricular ejection fraction < 20% as measured by resting echocardiogram. 10. Hypertrophic obstructive cardiomyopathy or severe basal septal hypertrophy with outflow gradient. 11. Echocardiographic evidence of an intracardiac mass, thrombus, vegetation or endocarditis. 12. Any other condition considered a contraindication for an isolated aortic valve procedure. 13. Symptomatic carotid or vertebral artery disease. 14. Expected life expectancy < 12 months due to associated non‐cardiac comorbidities. 15. Currently participating in another investigational drug or device trial. |
| Interventions | Experimental: transcatheter aortic valve implantation.
Transcatheter aortic valve implantation (TAVI) using the most appropriate CE (Conformité Européene)‐marked device available, with a minimum demand of experience of 30 implanted devices/type per centre. Active comparator: surgical aortic valve replacement. Surgical aortic valve replacement (SAVR) with free choice of surgical bioprosthesis and free choice of surgical access according to the surgeon's preference. |
| Outcomes | Primary outcome measures: 1. Overall survival (time frame: 5 years after last patient in) Efficacy endpoint . 2. Overall survival (time frame: after at least 1 year of follow‐up after last patient in and 196 deaths of any cause) Safety endpoint (event‐driven) . Secondary outcome measures: 1. Freedom from cardiovascular mortality (time frame: 5 years after last patient in) will be assessed at every study visit and compared between TAVI and SAVR groups . 2. Freedom from the composite of all‐cause mortality and stroke (time frame: 5 years after last patient in) will be assessed at every study visit and compared between TAVI and SAVR groups . 3. Freedom from myocardial infarction (time frame: 5 years after last patient in) will be assessed at every study visit and compared between TAVI and SAVR groups . 4. Freedom from stroke (time frame: 5 years after last patient in) will be assessed at every study visit and compared between TAVI and SAVR groups . 5. Freedom from major or life‐threatening/disabling bleeding (time frame: 5 years after last patient in) will be assessed at every study visit and compared between TAVI and SAVR groups . 6. Freedom from acute kidney injury (time frame: 5 years after last patient in) will be assessed at every study visit and compared between TAVI and SAVR groups . 7. Freedom from vascular access site and access‐related complications (time frame: 5 years after last patient in) will be assessed at every study visit and compared between TAVI and SAVR groups . 8. Freedom from conduction disturbances and arrhythmias, need for permanent pacemaker implantation (time frame: 5 years after last patient in) will be assessed at every study visit and compared between TAVI and SAVR groups . 9. Freedom from residual aortic regurgitation ≥ moderate (time frame: 5 years after last patient in) will be assessed at every study visit and compared between TAVI and SAVR groups . 10. Composite device success (time frame: 5 years after last patient in) ‒ Number of participants with freedom from procedural mortality and correct positioning of a single transcatheter heart valve (THV) in the proper position with intended performance (no prosthesis‐ patient mismatch and mean aortic valve gradient < 20 mmHg or peak velocity < 3 m/s, AND no moderate or severe prosthetic valve regurgitation) . 11. Composite early safety (time frame: 5 years after last patient in) ‒ Number of participants dying and/or number of participants with stroke (disabling and non‐disabling), and/or life‐threatening bleeding and/or acute kidney injury stages 2/3 and/or coronary artery obstruction requiring Intervention and/or major vascular complication and/or valve‐related dysfunction requiring repeat procedure. 12. Composite clinical efficacy (time frame: 5 years after last patient in) Number or participants dying and/or number of participants with stroke (disabling and non‐disabling) and/or rehospitalisation for worsening heart failure or valve‐related symptoms and/or New York Heart Association functional class (NYHA) III or IV and/or valve‐related dysfunction (mean aortic valve gradient > 20 mmHg, effective orifice area (EOA) < 0.9 to 1.1 cm² and/or Doppler Velocity Index (DVI) < 0.35 m/s, AND/OR moderate or severe prosthetic valve regurgitation) . 13. Freedom from prosthetic valve dysfunction (time frame: 5 years after last patient in) will be assessed at every study visit and compared between TAVI and SAVR groups . 14. Freedom from prosthetic aortic valve endocarditis (time frame: 5 years after last patient in) will be assessed at every study visit and compared between TAVI and SAVR groups . 15. Freedom from the composite time‐related valve safety (time frame: 5 years after last patient in) ‒ Number of participants with structural valve deterioration (including repeat procedures, prosthetic valve endocarditis and/or thrombosis) and/or number of participants with thromboembolic events (stroke) and/or Valve Academic Research Consortium (VARC‐2) bleeding (unless clearly unrelated to valve therapy). 16. Quality of life measures (time frame: 5 years after last patient in) ‒ Number of participants with reduced quality of life measures after valve replacement as compared to baseline levels prior to valve‐replacement, assessed using EuroQol 5 dimensions (EQ‐5D) questionnaire and/or Barthel Index and/or centre for epidemiologic studies depression (CES‐D) Scale. 17. Health economic analysis (time frame: 5 years after last patient in) ‒ Incremental cost‐effectiveness of TAVI compared to surgical valve replacement, by using quality adjusted life years (QALYs). |
| Notes | Contact: Moritz Seiffert, MD+49 (0) 40 7410 58206m.seiffert@uke.de
Contact: Stefan Blankenberg, MD+49 (0) 40 7410 53972s.blankenberg@uke.de Reason awaiting classification: inclusion criteria include participants at "low" to intermediate operative risk as assessed by the local heart team. The study is ongoing and we would need to see the final study population before making the final assessment of inclusion eligibility. |
NCT03383445.
| Methods | Randomised, unblinded controlled trial |
| Participants | Ages Eligible for Study: 65 years and older (older adult)
Sexes Eligible for Study: all
Accepts Healthy Volunteers: no
Inclusion criteria:
* Patients ≥ 65 years old diagnosed with severe AS (defined as: jet velocity ≥ 4.0 m/s or mean gradient ≥ 40 mmHg or velocity ratio < 0.25 AND aortic valve area ≤ 1.0 cm² or aortic valve area index ≤ 0.6 cm²/m²; OR mean gradient > 30 mmHg AND aortic valve area ≤ 1.0 cm² or aortic valve area index ≤ 0.6 cm²/m² AND > 1200 Agatston units for women or >2000 Agatston units for men as determined by non‐contrast CT).
* Small aortic annulus defined as a mean aortic annulus diameters ˂ 22 mm as measured by 3D‐computed tomography (CT) and/or 3D‐transoesophageal echocardiography (TEE). Exclusion criteria: * Prohibitive surgical risk as determined by the heart team * Severe pulmonary disease * Dialysis‐dependency * Porcelain aorta * Aortic root dilatation > 45 mm * Coronary artery disease (CAD) not treatable by percutaneous coronary intervention (PCI) or coronary artery bypass grafting (CABG), or SYNTAX score > 32 (in the absence of prior revascularization) 42 * Non‐calcific aortic stenosis * Severe mitral regurgitation * Moderate‐to‐severe tricuspid regurgitation requiring surgical repair |
| Interventions | TAVI
The TAVI procedure will be performed following the standards of each participating centre. No restriction or specific recommendation will be given regarding the approach, general vs. local anaesthesia, imaging guidance during the TAVI procedure, and post‐procedural TAVI management. Procedure: Edwards: the TAVI procedure will be performed with the Edwards SAPIEN XT or SAPIEN 3 valve (20 mm or 23 mm).
Procedure: CoreValve: the TAVI procedure will be performed with the CoreValve Evolut R valve system (23 mm or 26 mm). SAVR SAVR procedure will be performed using standard techniques, with no limitation in terms of type and size of the valve prosthesis or surgical procedure (e.g. enlargement of the aortic root). Procedure: standard. The choice of the type and size of valve, utilization of additional procedures such as root enlargement will be left to the discretion of heart team treating the patient. |
| Outcomes | Primary outcome measures: 1. Valve performance: rate of prothesis‐patient mismatch (PPM) and/or aortic regurgitation (AR) (time frame: 60 days) ‒ Severe PPM (defined as an indexed aortic valve area ≤ 0.65 cm²/m²) and/or ≥ moderate AR (Valve Academic Research Consortium‐2 (VARC‐2) definition). Secondary outcome measures: 1. Rate of PPM (time frame: 60 days, 1 year and 5 years) ‒ Rate of moderate or severe PPM 2. Rate of AR (time frame: 60 days, 1 year and 5 years) ‒ Rate of moderate or severe AR 3. Combined endpoints: rate of AR or PPM (time frame: 1 year and 5 years) ‒ Moderate or severe AR or severe PPM 4. Transvalvular gradient (time frame: 60 days, 1 year and 5 years) ‒ Mean transvalvular gradient 5. Combined endpoints: LVEF and LV (time frame: 60 days, 1 year and 5 years) ‒ Changes in LVEF and LV hypertrophy 6. Mortality (time frame: 30 days, 1 year and 5 years) ‒ Death 7. Stroke (time frame: 30 days, 1 year and 5 years) ‒ Stroke (Valve Academic Research Consortium‐2 (VARC‐2) definition) 8. Bleeding (time frame: 30 days, 1 year and 5 years) ‒ Major or life‐threatening bleeding 9. Rate of new atrial fibrillation (time frame: 30 days, 1 year and 5 years) ‒ Rate of new‐onset atrial fibrillation 10. Combined Safety endpoint (time frame: 30 days, 1 year and 5 years) ‒ Death, stroke, major/life threatening bleeding 11. Cardiac re‐hospitalization (time frame: 30 days, 1 year and 5 years) ‒ Need for cardiac re‐hospitalization 12. Day of hospital stay (time frame: for the duration of hospital stay) 13. Quality of life (time frame: 30 days, 1 year and 5 years) ‒Questionnaire, visual scale 14. Exercise capacity (time frame: 30 days, 1 year and 5 years) ‒Exercise capacity as evaluated by the 6‐minute walk test |
| Notes | Contact: Josep Rodés‐Cabau, MD 418‐656‐8711 josep.rodes@criucpq.ulaval.ca Contact: Emilie Pelletier Beaumont, MSc 418‐656‐8711 ext3929 emilie.pelletier‐beaumont@criucpq.ulaval.ca Reason awaiting classification: based on the study inclusion/exclusion criteria, the final study population may or may not be eligible for inclusion in our meta‐analysis. The study is still ongoing and we would need to see the final study population before making the final assessment of inclusion eligibility. |
PARTNER 2 2016.
| Methods | Randomised controlled trial |
| Participants | Inclusion criteria: 1. Patient had senile degenerative aortic valve stenosis with echocardiographically derived criteria: mean gradient > 40 mmHg or jet velocity greater than 4.0 m/s and an initial aortic valve area (AVA) of < 0.8 cm² or indexed EOA < 0.5 cm²/m². Qualifying echo was within 60 days of the date of the procedure. 2. Patient was symptomatic from his/her aortic valve stenosis, as demonstrated by NYHA Functional Class II or greater. 3. The heart team agreed (and verified in the case review process) that valve implantation would likely benefit the patient. 4. The study patient or the study patient’s legal representative was informed of the nature of the study, agreed to its provisions and had provided written informed consent as approved by the Institutional Review Board (IRB) of the respective clinical site. 5. The study patient agreed to comply with all required post‐procedure follow‐up visits including annual visits through 5 years and analysis close‐date visits, which was conducted as a phone follow‐up. Additional eligibility criteria specific to cohort A: 1. STS > 4 or < 4 if the heart team determines intermediate‐risk patient profile with important comorbidities not represented in the STS risk score algorithm. 2. Heart team (including examining cardiac surgeon) agree on eligibility including assessment that TAVI or AVR is appropriate. 3. Heart team agreed (a priori) on treatment strategy for concomitant coronary disease (if present). 4. Study patient agreed to undergo surgical aortic valve replacement (AVR) if randomised to control treatment. Exclusion criteria: 1. Heart team assessment of inoperability (including examining cardiac surgeon). 2. Evidence of an acute myocardial infarction ≤ 1 month (30 days) before the intended treatment (defined as: Q wave MI, or non‐Q wave MI with total CK elevation of CK‐MB ≥ twice normal in the presence of MB elevation and/or troponin level elevation (WHO definition)). 3. Aortic valve is a congenital unicuspid or congenital bicuspid valve, or is non‐calcified. 4. Mixed aortic valve disease (aortic stenosis and aortic regurgitation with predominant aortic regurgitation > 3+). 5. Pre‐existing mechanical or bioprosthetic valve in any position (NR3). 6. Complex coronary artery disease: a. unprotected left main coronary artery; b. syntax score > 32 (in the absence of prior revascularization). 7. Any therapeutic invasive cardiac procedure resulting in a permanent implant that is performed within 30 days of the index procedure (unless part of planned strategy for treatment of concomitant coronary artery disease). Implantation of a permanent pacemaker is not excluded. 8. Any patient with a balloon valvuloplasty (BAV) within 30 days of the procedure (unless BAV is a bridge to procedure after a qualifying ECHO). 9. Patients with planned concomitant surgical or transcatheter ablation for atrial fibrillation. 10. Leukopenia (WBC < 3000 cell/mL), acute anaemia (Hgb < 9 G/dL), Thrombocytopenia (Plt < 50,000 cell/mL). 11. Hypertrophic cardiomyopathy with or without obstruction. 12. Severe ventricular dysfunction with LVEF < 20%. 13. Echocardiographic evidence of intracardiac mass, thrombus or vegetation. 14. Active upper GI bleeding within 3 months (90 days) prior to procedure. 15. A known contraindication or hypersensitivity to all anticoagulation regimens, or inability to be anticoagulated for the study procedure. 16. Native aortic annulus size < 18 mm or > 27 mm as measured by echocardiogram. 17. Clinically (by neurologist) or neuroimaging confirmed stroke or transient ischaemic attack (TIA) within 6 months (180 days) of the procedure. 18. Renal insufficiency (creatinine > 3.0 mg/dL) and/or renal replacement therapy at the time of screening. 19. Estimated life expectancy < 24 months (730 days) due to carcinomas, chronic liver disease, chronic renal disease or chronic end stage pulmonary disease. 20. Expectation that patient will not improve despite treatment of aortic stenosis 21. Currently participating in an investigational drug or another device study. Note: trials requiring extended follow‐up for products that were investigational, but have since become commercially available, are not considered investigational trials. 22. Active bacterial endocarditis within 6 months (180 days) of procedure. 23. Patient refuses aortic valve replacement surgery. |
| Interventions | 1. Transcatheter aortic valve implantation 2. Surgical aortic valve replacement |
| Outcomes | The primary end point was a non‐hierarchical composite of death from any cause or disabling stroke at 2 years in the intention‐to‐treat population; all the patients were followed for at least 2 years. Death from any cause or disabling stroke Death from cardiac cause Neurologic event (transient ischaemic attack, disabling stroke, and non‐disabling stroke) Rehospitalization Myocardial infarction Major vascular complication Life‐threatening or disabling bleeding Acute kidney injury New atrial fibrillation New permanent pacemaker Endocarditis Aortic‐valve re‐intervention Coronary obstruction |
| Notes | Correspondence: Dr. Leon at Columbia University Medical Center, 161 Ft. Washington Ave., 6th Floor, New York, NY 10032, or at mleon@crf.org. Reason awaiting classification: the study may have had low‐risk participants, but we were unable to clarify this from the trialists or obtain data for our population of interest. |
Characteristics of ongoing studies [ordered by study ID]
NCT02825134.
| Trial name or title | Comparison of transcatheter versus surgical aortic valve replacement in younger low surgical risk patients with severe aortic stenosis (NOTION‐2) |
| Methods | The study is a randomised clinical multicentre trial. Central randomisation with variable block size and stratification by gender and coronary comorbidity will be used. An independent event committee blinded to treatment allocation will adjudicate safety endpoints. Interim analysis is planned after the first 20 events included in the primary end point (all‐cause mortality, stroke or myocardial infarction). |
| Participants | Younger low risk patients with severe aortic valve stenosis, who are scheduled for aortic valve replacement using a bioprosthesis. Subjects fulfilling the inclusion criteria, not having any exclusion criteria, and consenting to the trial will be randomised 1:1 to TAVI or SAVR with 496 patients in each group. |
| Interventions | TAVI: any CE‐Mark approved transcatheter aortic bioprosthesis may be used in the study, and the choice is at the discretion of the local TAVI team. The transfemoral TAVI procedure may be performed under general anaesthesia, local anaesthesia/conscious sedation, or local anaesthesia. Percutaneous coronary intervention (PCI) can be performed up to 30 days prior to TAVI or as a hybrid procedure. SAVR: the surgical SAVR technique follows standard protocol of the local department of cardio‐thoracic surgery. The operation is performed under general anaesthesia, which follows standard protocol of the department of anaesthesiology. A commercially available surgical aortic bioprosthesis will, at the surgeons discretion, be implanted. Concomitant coronary artery bypass graft (CABG) surgery may be performed. |
| Outcomes | The primary endpoint is the composite rate of all‐cause mortality, myocardial infarction and stroke within 1 year after the procedure (VARC‐2 definitions). Secondary endpoints: ‐ Device success (Absence of procedural mortality, correct positioning of a single valve into the proper anatomical location AND intended performance of the prosthetic heart valve) (time frame: at 30 days, 1 year and annually thereafter up to 10 years post‐procedure) VARC‐2 definitions ‐ Procedure time (time frame: intraoperative) ‐ Duration of index hospitalisation (time frame: number of days from admission to discharge (expected an average of 7 days)) ‐ Composite rate of all‐cause mortality, myocardial infarction and stroke (time frame: at 30 days, 1 year and annually thereafter up to 10 years post‐procedure) VARC‐2 definitions ‐ Cardiovascular mortality (time frame: at 30 days, 1 year and annually thereafter up to 10 years post‐procedure) VARC‐2 definitions ‐ Stroke or TIA (time frame: at 30 days, 1 year and annually thereafter up to 10 years post‐procedure) VARC‐2 definitions ‐ Bleeding (life‐threatening, major or minor) (time frame: at 30 days, 1 year and annually thereafter up to 10 years post‐procedure) VARC‐2 definitions ‐ Vascular complication (major or minor) (time frame: at 30 days, 1 year and annually thereafter up to 10 years post‐procedure) VARC‐2 definitions ‐ Acute kidney injury (stage 1, 2 or 3) (time frame: at 30 days, 1 year and annually thereafter up to 10 years post‐procedure) VARC‐2 definitions ‐ Echocardiographic aortic bioprosthesis performance (degree of paravalvular leakage, valve area, mean gradient) (time frame: before discharge from index hospitalisation (expected an average of 7 days), at 30 days, 1 year and annually thereafter up to 10 years post‐procedure) VARC‐2 definitions ‐ NYHA functional class (time frame: at 30 days, 1 year and annually thereafter up to 10 years post‐procedure] ‐ Need for permanent pacemaker (time frame: at 30 days, 1 year and annually thereafter up to 10 years post‐procedure] VARC‐2 definitions ‐ New onset atrial fibrillation captured on ECG (time frame: within discharge from index hospitalisation (expected an average of 7 days), at 30 days, 1 year and annually thereafter up to 10 years post‐procedure) VARC‐2 definitions ‐ Time‐related valve safety (echocardiographic structural valve deterioration, prosthetic valve endocarditis, prosthetic valve thrombosis, thrombo‐embolic events OR VARC bleeding) (time frame: at 30 days, 1 year and annually thereafter up to 10 years post‐procedure) VARC‐2 definitions ‐ Left ventricle remodeling as assessed by echocardiography [Time Frame: at 30 days, 1 year and annually thereafter up to 10 years post‐procedure] 1‐year overall costs in both treatment arms. (Time frame: 1 year) ‐ Duration of stay on ICU after index procedure. (Time frame: number of days from procedure to discharge from ICU) ‐ Incidence of early safety (all‐cause mortality, all‐stroke, life‐threatening bleeding, acute kidney injury, coronary artery obstruction requiring intervention, major vascular complication OR valve‐related dysfunction requiring repeat procedure) (time frame: at 30 days from index procedure) VARC‐II definitions ‐ Clinical efficacy (all‐cause mortality, all stroke, requiring hospitalisation for valve‐related symptoms or worsening congestive heart failure, NYHA class III or IV OR echocardiographic valve‐related dysfunction) (time frame: after 30 days of index procedure) VARC‐II definitions ‐ Quality of life change from baseline (time frame: at 30 days, 1 year and annually thereafter up to 10 years post‐procedure) assessed by SF‐36v2, EQ‐5d and KCCQ Follow‐up will be performed after 30 days, 3 months, 1 year and yearly thereafter for a minimum of 5 years. |
| Starting date | June 2016 |
| Contact information | Lars Søndergaard, MD; DMSc: Lars.Soendergaard.01@regionh.dk |
| Notes | Sponsors and Collaborators: ‐ Rigshospitalet, Denmark ‐ Symetis SA ‐ Boston Scientific Corporation ‐ Abbott |
Differences between protocol and review
We sought to conduct the review in accordance with our previously published protocol — Kolkailah 2019 — with no differences or deviations. However, we have made the following changes, as justified below:
1. We used the term "transcatheter aortic valve implantation, or TAVI" instead of "transcatheter aortic valve replacement, or TAVR" throughout the manuscript. This was based on Cochrane Heart Group editorial board's request to comply with the British/European spelling and to reflect a more accurate nomenclature for all stakeholders (Clegg 2012).
2. We added a post hoc sensitivity analysis excluding STACCATO 2012 since it utilised the less contemporary transapical TAVI and was prematurely terminated due to an excess of adverse events in the TAVI group.
Contributions of authors
AK: screened studies, extracted data, assessed the risk of bias, conducted meta‐analysis, drafted the full review, and approved the final version of the review.
RD: assessed the review critically for intellectual content and approved the final version of the review.
MP: assessed the review critically for intellectual content and approved the final version of the review.
AV: assessed the review critically for intellectual content and approved the final version of the review.
TK: assessed the review critically for intellectual content and approved the final version of the review.
AN: screened studies, verified the extracted data, assessed the risk of bias, shared in conducting meta‐analysis, produced the 'summary of findings' table, assessed the review critically for intellectual content, and approved the final version of the review.
Sources of support
Internal sources
-
Egyptian Center for Evidence Based Medicine, Egypt.
Advanced author training and methodological support.
External sources
This project was supported by the National Institute for Health Research (NIHR), via Cochrane Infrastructure and Cochrane Incentive funding to the Heart Group. The views and opinions expressed herein are those of the authors and do not necessarily reflect those of the Systematic Reviews Programme, NIHR, NHS, or the Department of Health and Social Care, UK.
Declarations of interest
AK: No conflicts of interest to disclose.
RD: No conflicts of interest to disclose.
MP: Consultancy work for Abbott Medical and LivaNova. Received funds to attend a Cryolife Investigators Meeting.
AV: No conflicts of interest to disclose.
TK: Speaker Bureau/Proctor for Edwards Lifesciences, Medtronic, and Abbott Vascular.
AN: No conflicts of interest to disclose.
New
References
References to studies included in this review
Evolut 2019 {published data only}
- NCT02701283. Medtronic Evolut transcatheter aortic valve replacement in low risk patients [Transcatheter aortic valve replacement with the medtronic transcatheter aortic valve replacement system in patients at low risk for surgical aortic valve replacement]. clinicaltrials.gov/ct2/show/NCT02701283 first posted March 8, 2016.
- Popma JJ, Deeb GM, Yakubov SJ, Mumtaz M, Gada H, O'Hair D, et al. Transcatheter aortic‐valve replacement with a self‐expanding valve in low‐risk patients. The New England Journal of Medicine 2019;380(18):1706‐715. [DOI] [PubMed] [Google Scholar]
NOTION 2015 {published data only}
- Jørgensen TH, Thyregod HG, Tarp JB, Svendsen JH, Søndergaard L. Temporal changes of new‐onset atrial fibrillation in patients randomized to surgical or transcatheter aortic valve replacement. International Journal of Cardiology 2017;234:16‐21. [DOI] [PubMed] [Google Scholar]
- NCT01057173. The Nordic Aortic Valve Intervention trial (NOTION) [Transcatheter versus surgical aortic valve implantation in patients with severe aortic valve stenosis]. clinicaltrials.gov/ct2/show/NCT01057173 first posted January 27, 2010.
- Søndergaard L, Ihlemann N, Capodanno D, Jørgensen T, Nissen H, Kjeldsen BJ, et al. Durability of transcatheter and surgical bioprosthetic aortic valves in patients at lower surgical Risk. Journal of the American College of Cardiology 2019;73(5):546–53. [DOI] [PubMed] [Google Scholar]
- Søndergaard L, Steinbrüchel DA, Ihlemann N, Nissen H, Kjeldsen BJ, Petursson P. Two‐year outcomes in patients with severe aortic valve stenosis randomized to transcatheter versus surgical aortic valve replacement: the all‐comers Nordic Aortic Valve Intervention randomized clinical trial. Circulation: Cardiovascular Interventions 2016;9(6):e003665. [DOI] [PubMed] [Google Scholar]
- Thyregod HG, Ihlemann N, Jørgensen TH, Nissen H, Kjeldsen BJ, Petursson P, et al. Five‐year clinical and echocardiographic outcomes from the Nordic Aortic Valve Intervention (NOTION) randomized clinical trial in lower surgical risk patients. Circulation 2019;139:2714–23. [DOI] [PubMed] [Google Scholar]
- Thyregod HG, Steinbrüchel DA, Ihlemann N, Nissen H, Kjeldsen BJ, Petursson P, et al. Transcatheter versus surgical aortic valve replacement in patients with severe aortic valve stenosis: 1‐Year results from the all‐comers NOTION randomized clinical trial. Journal of the American College of Cardiology 2015;65(20):2184‐94. [DOI] [PubMed] [Google Scholar]
- Thyregod HG, Søndergaard L, Ihlemann N, Franzen O, Andersen LW, Hansen PB, et al. The Nordic Aortic Valve Intervention (NOTION) trial comparing transcatheter versus surgical valve implantation: study protocol for a randomised controlled trial. Trials 2013;14:11. [DOI] [PMC free article] [PubMed] [Google Scholar]
PARTNER 3 2019 {published data only}
- Mack MJ, Leon MB, Thourani VH, Makkar R, Kodali SK, Russo M, et al. Transcatheter aortic‐valve replacement with a balloon‐expandable valve in low‐risk patients. The New England Journal of Medicine 2019;380(18):1695‐705. [DOI] [PubMed] [Google Scholar]
- NCT02675114. The PARTNER 3 ‐ trial ‐ the safety and effectiveness of the SAPIEN 3 transcatheter heart valve in low risk Patients with aortic stenosis (P3) [A prospective, randomized, controlled, multi‐center study to establish the safety and effectiveness of the SAPIEN 3 transcatheter heart valve in low risk patients who have severe, calcific, aortic stenosis requiring aortic valve replacement]. clinicaltrials.gov/ct2/show/NCT02675114 first posted February 5, 2016.
STACCATO 2012 {published data only}
- Nielsen HH, Klaaborg KE, Nissen H, Terp K, Mortensen PE, Kjeldsen BJ, et al. A prospective, randomised trial of transapical transcatheter aortic valve implantation vs. surgical aortic valve replacement in operable elderly patients with aortic stenosis: the STACCATO trial. EuroIntervention 2012;8:383‐89. [DOI] [PubMed] [Google Scholar]
- Ryhammer PK, Greisen J, Jakobsen CJ. Hemodynamic consequences following surgical aortic valve replacement versus transcatheter aortic valve implantation ‐ a randomized study. Acta Anaesthesiologica Scandinavica 2015;11:11. [Google Scholar]
References to studies excluded from this review
NCT02628899 {published data only}
- NCT02628899. Feasibility of transcatheter aortic valve replacement in low‐risk patients with symptomatic, severe aortic stenosis. clinicaltrials.gov/ct2/show/NCT02628899 first posted December 11, 2015.
NCT02838199 {published data only}
- NCT02838199. Transcatheter or surgIcal aortic valve replacement in all‐comers with severe aortic valve stenosis (TRANSIT) [Prospective, open label, multicenter, dual arm, randomized trial of transcatheter or surgIcal aortic valve replacement in all‐comers with severe aortic valve stenosis]. clinicaltrials.gov/ct2/show/NCT02838199 first posted July 20, 2016.
SURTAVI 2017 {published data only}
- Reardon MJ, Mieghem NM, Popma JJ, Kleiman NS, Søndergaard L, Mumtaz M, et al. Surgical or transcatheter aortic‐valve replacement in intermediate‐risk patients. The New England Journal of Medicine 2017;376(14):1321‐31. [DOI] [PubMed] [Google Scholar]
- Serruys PW, Modolo R, Reardon M, Miyazaki Y, Windecker S, Popma J, et al. One‐year outcomes of patients with severe aortic stenosis andan STS PROM of less than three percent in the SURTAVI trial. EuroIntervention 2018;14(8):877‐83. [DOI] [PubMed] [Google Scholar]
References to studies awaiting assessment
NCT03112980 {published data only}
- NCT03112980. Randomized trial of TAVI vs. SAVR in patients with severe aortic valve stenosis at intermediate risk of mortality (DEDICATE) [Randomized, multi‐center, event‐driven trial of TAVI versus SAVR in patients with symptomatic severe aortic valve stenosis and intermediate risk of mortality, as assessed by STS‐score ‐ DEDICATE]. clinicaltrials.gov/ct2/show/NCT03112980 first posted April 13, 2017.
NCT03383445 {published data only}
- NCT03383445. Transcatheter aortic valve replacement versus surgical aortic valve replacement for treating elderly patients with severe aortic stenosis and small aortic annuli: a prospective randomized study The VIVA trial. clinicaltrials.gov/ct2/show/NCT03383445 first posted December 26, 2017.
PARTNER 2 2016 {published data only}
- Baron SJ, Wang K, House JA, Magnuson EA, Reynolds MR, Makkar R, et al. Cost‐effectiveness of transcatheter versus surgical aortic valve replacement in patients with severe aortic stenosis at intermediate risk. Circulation 2019;139(7):877‐88. [DOI] [PubMed] [Google Scholar]
- Leon MB, Smith CR, Mack MJ, Makkar RR, Svensson LG, Kodali SK, et al. Transcatheter or surgical aortic‐valve replacement in intermediate‐risk Patients. The New England Journal of Medicine 2016;374(17):1609‐20. [DOI] [PubMed] [Google Scholar]
References to ongoing studies
NCT02825134 {published data only}
- NCT02825134. Comparison of transcatheter versus surgical aortic valve replacement in younger low surgical risk patients with severe aortic stenosis (NOTION‐2) [Nordic Aortic Valve Intervention trial 2 ‐ a randomized multicenter comparison of transcatheter versus surgical aortic valve replacement in younger low surgical risk patients with severe aortic stenosis]. clinicaltrials.gov/ct2/show/NCT02825134 first posted July 7, 2016.
Additional references
Al‐Abdouh 2019
Arora 2017
- Arora S, Strassle PD, Ramm CJ, Rhodes JA, Vaidya SR, Caranasos TG, et al. Transcatheter versus surgical aortic valve replacement in patients with lower surgical risk scores: a systematic review and meta‐analysis of early outcomes. Heart, Lung & Circulation 2017;26(8):840‐5. [DOI] [PubMed] [Google Scholar]
Baumgartner 2017a
- Baumgartner H, Hung J, Bermejo J, Chambers JB, Edvardsen T, Goldstein S, et al. Recommendations on the echocardiographic assessment of aortic valve stenosis: a focused update from the European Association of Cardiovascular Imaging and the American Society of Echocardiography. European Heart Journal 2017;18(3):254‐75. [DOI] [PubMed] [Google Scholar]
Baumgartner 2017b
- Baumgartner H, Falk V, Bax JJ, Bonis M, Hamm C, Holm PJ, et al. 2017 ESC/EACTS Guidelines for the management of valvular heart disease. European Heart Journal 2017;38(36):2739–91. [DOI] [PubMed] [Google Scholar]
Bhatia 2016
- Bhatia N, Basra SS, Skolnick AH, Wenger NK. Aortic valve disease in the older adult. Journal of Geriatric Cardiology 2016;13(12):941‐4. [DOI] [PMC free article] [PubMed] [Google Scholar]
Clegg 2012
- Clegg SD, Krantz MJ. Transcatheter aortic valve replacement: what's in a name?. Journal of the American College of Cardiology 2012;60(3):239. [DOI] [PubMed] [Google Scholar]
FDA 2019
- FDA expands indication for several transcatheter heart valves to patients at low risk for death or major complications associated with open‐heart surgery. www.fda.gov/news‐events/press‐announcements/fda‐expands‐indication‐several‐transcatheter‐heart‐valves‐patients‐low‐risk‐death‐or‐major 16 August 2019.
GRADEpro GDT 2015 [Computer program]
- McMaster University (developed by Evidence Prime). GRADEpro GDT. Version accessed 21 March 2019. Hamilton (ON): McMaster University (developed by Evidence Prime), 2015.
Greenbaum 2017
- Greenbaum AB, Babaliaros VC, Chen MY, Stine AM, Rogers T, O’Neill WW, et al. Transcaval access and closure for transcatheter aortic valve replacement: a prospective investigation. Journal of the American College of Cardiology 2017;69(5):511‐21. [DOI] [PMC free article] [PubMed] [Google Scholar]
Grimard 2016
- Grimard BH, Safford RE, Burns EL. Aortic stenosis: diagnosis and treatment. American Family Physician 2016;93(5):371‐8. [PubMed] [Google Scholar]
Grover 2017
- Grover FL, Vemulapalli S, Carroll JD, Edwards FH, Mack MJ, Thourani VH, et al. STS/ACC TVT Registry. 2016 Annual Report of The Society of Thoracic Surgeons/American College of Cardiology Transcatheter Valve Therapy Registry. Journal of the American College of Cardiology 2017;69(10):1215–30. [DOI] [PubMed] [Google Scholar]
Higgins 2017
- Higgins JP, Altman DG, Sterne JAC (editors). Chapter 8: Assessing risk of bias in included studies. In: Higgins JPT, Churchill R, Chandler J, Cumpston MS (editors). Cochrane Handbook for Systematic Reviews of Interventions version 5.2.0 (updated June 2017). The Cochrane Collaboration, 2017. Available at www.training.cochrane.org/handbook.
Hirji 2017
- Hirji SA, Ramirez‐Del Val F, Kolkailah AA, Ejiofor JI, McGurk S, Chowdhury R, et al. Outcomes of surgical and transcatheter aortic valve replacement in the octogenarians ‐ surgery still the gold standard?. Annals of Cardiothoracic Surgery 2017;6(5):453‐62. [DOI] [PMC free article] [PubMed] [Google Scholar]
Hirji 2018a
- Hirji SA, Kolkailah AA, Ramirez‐Del Val F, Lee J, McGurk S, Pelletier P, et al. Mechanical versus bioprosthetic aortic valve replacement in patients aged 50 years and younger. Annals of Thoracic Surgery 2018;106(4):1113‐21. [DOI] [PubMed] [Google Scholar]
Hirji 2018b
- Hirji SA, Funamoto M, Lee J, Ramirez Del Val F, Kolkailah AA, McGurk S, et al. Minimally invasive versus full sternotomy aortic valve replacement in low‐risk patients: which will stand against transcatheter aortic valve replacement?. Surgery 2018;164(2):282‐7. [DOI] [PubMed] [Google Scholar]
Junquera 2019
- Junquera L, Ferreira‐Neto A, Guimaraes L, Asmarats L, Val D, Wintzer‐Wehekind J. Transcatheter aortic valve replacement in low risk patients. Minerva Cardioangiologica 2019;67(1):19‐38. [DOI] [PubMed] [Google Scholar]
Kheiri 2019
- Kheiri B, Osman M, Abubakar H, Subahi A, Chahine A, Ahmed S, et al. Transcatheter versus surgical aortic valve replacement in low‐risk surgical patients: a meta‐analysis of randomized clinical trials. Cardiovascular Revascularization Medicine 2019;pii:S1553‐8389(18)30626‐2. [DOI] [PubMed] [Google Scholar]
Kolkailah 2018
- Kolkailah AA, Hirji SA, Ejiofor JI, Ramirez Del Val F, Lee J, Norman AV, et al. Novel fast‐track recovery protocol for alternative access transcatheter aortic valve replacement: application to non‐femoral approaches. Interactive CardioVascular and Thoracic Surgery 2018;26:938–43. [DOI] [PubMed] [Google Scholar]
Kolte 2019
- Kolte D, Vlahakes GJ, Palacios IF, Sakhuja R, Passeri JJ, Inglessis I, et al. Transcatheter versus surgical aortic valve replacement in low‐risk patients. Journal of the American College of Cardiology 2019;74(12):1532‐40. [DOI] [PubMed] [Google Scholar]
Lefebvre 2011
- Lefebvre C, Manheimer E, Glanville J. Chapter 6: Searching for studies. In: Higgins JP, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Leon 2010
- Leon MB, Smith CR, Mack M, Miller DC, Moses JW, Svensson LG. Transcatheter aortic‐valve implantation for aortic stenosis in patients who cannot undergo surgery. New England Journal of Medicine 2010;363(17):1597‐607. [DOI] [PubMed] [Google Scholar]
Leon 2016
- Leon MB, Smith CR, Mack MJ, Makkar RR, Svensson LG, Kodali SK, et al. Transcatheter or surgical aortic‐valve replacement in intermediate‐risk patients. New England Journal of Medicine 2016;374:1609‐20. [DOI] [PubMed] [Google Scholar]
Liberati 2009
- Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gotzsche PC, Ioannidis JP, et al. The PRISMA statement for reporting systematic reviews and meta‐analyses of studies that evaluate health care interventions: explanation and elaboration. PLOS Medicine 2009;6(7):e1000100. [DOI] [PMC free article] [PubMed] [Google Scholar]
Maganti 2010
- Maganti K, Rigolin VH, Sarano ME, Bonow RO. Valvular heart disease: diagnosis and management. Mayo Clinic Proceedings 2010;85(5):483‐500. [DOI] [PMC free article] [PubMed] [Google Scholar]
Mylotte 2016
- Mylotte D, Sudre A, Teiger E, Obadia JF, Lee M, Spence M, et al. Transcarotid transcatheter aortic valve replacement: feasibility and safety. Journal of the American College of Cardiology: Cardiovascular Interventions 2016;9(5):472–80. [DOI] [PubMed] [Google Scholar]
Nashef 2012
- Nashef SA, Roques F, Sharples LD, Nilsson J, Smith C, Goldstone AR, et al. EuroSCORE II. European Journal of Cardio‐thoracic Surgery 2012;41(4):734–45. [DOI] [PubMed] [Google Scholar]
Nishimura 2017
- Nishimura RA, Otto CM, Bonow RO, Carabello BA, Erwin JP 3rd, Fleisher LA, et al. 2017 AHA/ACC focused update of the 2014 AHA/ACC guideline for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 2017;135:e1159‐95. [DOI] [PubMed] [Google Scholar]
O'Brien 2009
- O'Brien SM, Shahian DM, Filardo G, Ferraris VA, Haan CK, Rich JB, et al. The Society of Thoracic Surgeons 2008 cardiac surgery risk models: part 2 ‐ isolated valve surgery. Annals of Thoracic Surgery 2009;88(Suppl 1):S23‐42. [DOI] [PubMed] [Google Scholar]
Overtchouk 2019
- Overtchouk P, Prendergast B, Modine T. Why should we extend transcatheter aortic valve implantation to low‐risk patients? A comprehensive review. Archives of Cardiovascular Diseases 2019;112(5):354‐62. [DOI] [PubMed] [Google Scholar]
Piazza 2008
- Piazza N, Jaegere P, Schultz C, Becker AE, Serruys PW, Anderson RH. Anatomy of the aortic valvar complex and its implications for transcatheter implantation of the aortic valve. Circulation: Cardiovascular Interventions 2008;1(1):74‐81. [DOI] [PubMed] [Google Scholar]
Review Manager 2014 [Computer program]
- Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager 5 (RevMan 5). Version 5.3. Copenhagen: Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Schünemann 2013
- Schünemann H, Brożek J, Guyatt G, Oxman A, editors. GRADE Handbook for grading quality of evidence and strength of recommendations. Updated October 2013. The GRADE Working Group, 2013. Available from guidelinedevelopment.org/handbook.
Schünemann 2017
- Schünemann HJ, Oxman AD, Vist GE, Higgins JP, Deeks JJ, Glasziou P, et al. Chapter 12: Interpreting results and drawing conclusions. In: Higgins JPT, Churchill R, Chandler J, Cumpston MS (editors). Cochrane Handbook for Systematic Reviews of Interventions version 5.2.0 (updated June 2017). The Cochrane Collaboration, 2017. Available from www.training.cochrane.org/handbook.
Thourani 2015
- Thourani VH, Li C, Devireddy C, Jensen HA, Kilgo P, Leshnower BG, et al. High‐risk patients with inoperative aortic stenosis: use of transapical, transaortic, and transcarotid techniques. Annals of Thoracic Surgery 2015;99(3):817–23; discussion 823‐5. [DOI] [PubMed] [Google Scholar]
Witberg 2018
- Witberg G, Lador A, Yahav D, Kornowski R. Transcatheter versus surgical aortic valve replacement in patients at low surgical risk: a meta‐analysis of randomized trials and propensity score matched observational studies. Catheterization and Cardiovascular Interventions 2018;92(2):408‐16. [DOI] [PubMed] [Google Scholar]
Witberg 2019
References to other published versions of this review
Kolkailah 2019
- Kolkailah AA, Doukky R, Pelletier MP, Kaneko T, Nabhan AF. Transcatheter versus surgical aortic valve replacement for severe aortic stenosis in people with low surgical risk. Cochrane Database of Systematic Reviews 2019, Issue 4. [DOI: 10.1002/14651858.CD013319] [DOI] [PMC free article] [PubMed] [Google Scholar]


