Genetic and morphometric analysis of germinated ancient date seeds provide initial insights into origins of the Judean date palm.
Abstract
Germination of 2000-year-old seeds of Phoenix dactylifera from Judean desert archaeological sites provides a unique opportunity to study the Judean date palm, described in antiquity for the quality, size, and medicinal properties of its fruit, but lost for centuries. Microsatellite genotyping of germinated seeds indicates that exchanges of genetic material occurred between the Middle East (eastern) and North Africa (western) date palm gene pools, with older seeds exhibiting a more eastern nuclear genome on a gradient from east to west of genetic contributions. Ancient seeds were significantly longer and wider than modern varieties, supporting historical records of the large size of the Judean date. These findings, in accord with the region’s location between east and west date palm gene pools, suggest that sophisticated agricultural practices may have contributed to the Judean date’s historical reputation. Given its exceptional storage potentialities, the date palm is a remarkable model for seed longevity research.
INTRODUCTION
The date palm (Phoenix dactylifera), a dioecious species in the Arecaceae (formerly Palmae) family has a historical distribution stretching from Mauritania in the west to the Indus Valley in the east (1). A major fruit crop in hot and arid regions of North Africa and the Middle East and one of the earliest domesticated tree crops, archaeobotanical records suggest that the earliest exploitation and consumption of dates is from the Arabian Neolithic some 7000 years before the present (yr B.P.) (1). Evidence of cultivation in Mesopotamia and Upper Arabian Gulf approximately 6700 to 6000 yr B.P. support these centers as the ancient origin of date palm domestication in this region, with a later establishment of oasis agriculture in North Africa (1, 2).
The current date palm germplasm is constituted by two highly differentiated gene pools: an eastern population, consisting of cultivars extending from the Middle East and Arabian Peninsula to northwest India and Pakistan and a western population covering North Africa and sub-Saharan Africa (3, 4). Introgressive hybridization by a wild relative in North African date palms has been proposed as a source of this differentiation (2).
Date palms in the southern Levant (modern-day Israel, Palestine, and Jordan), situated between eastern and western domestication areas, have historically played an important economic role in the region and were also of symbolic and religious significance (5). The Kingdom of Judah (Judea) that arose in the southern part of the historic Land of Israel in the 11th century BCE was particularly renowned for the quality and quantity of its dates. These so-called “Judean dates” grown in plantations around Jericho and the Dead Sea were recognized by classical writers for their large size, sweet taste, extended storage, and medicinal properties (5). While evidence suggests that Judean date culture continued during the Byzantine and Arab periods (4th to 11th century CE), further waves of conquest proved so destructive that by the 19th century, no traces of these historic plantations remained (5).
In 2008, we reported the germination of a 1900-year-old date seed (6) recovered from the historical site of Masada overlooking the Dead Sea. In the current study, six additional ancient date seeds from archaeological sites in the Judean desert were germinated, bringing to seven the number of ancient genotypes genetically analyzed using molecular markers. In addition, morphometric analysis was used to compare the size and shape of ungerminated ancient date seeds with modern varieties and wild dates.
This study, which confirms the long-term survival of date palm seeds, provides a unique opportunity to rediscover the origins of a historic date palm population that existed in Judea 2000 years ago. The characteristics of the Judean date palm may shed light on aspects of ancient cultivation that contributed to the quality of its fruit and is thus of potential relevance to the agronomic improvement of modern dates.
RESULTS
Seed germination
Of the hundreds of ancient date seeds and other botanical material recovered from excavations carried out in the Judean desert between 1963 and 1991 (7, 8) (fig. S1), 32 well-preserved date seeds from the archaeological sites of Masada, Qumran, Wadi Makukh, and Wadi Kelt were planted in a quarantine site at Kibbutz Ketura (table S1). Of these, six ancient seeds germinated and were further identified by the following monikers: Masada: “Adam”; Qumran: “Jonah,” “Uriel,” “Boaz,” and “Judith”; and Wadi Makukh: “Hannah” (Figs. 1 and 2).
Fig. 1. Morphology of six germinated ancient date seeds before planting.

(A) Adam, (B) Jonah, (C) Uriel, (D) Boaz, (E) Judith, (F) Hannah, and (G) HU37A11, an unplanted ancient date seed from Qumran (Cave FQ37) used as a control. Scale bars, 0.5 cm (A, no bar size as unmeasured before planting). Photo credit: Guy Eisner.
Fig. 2. Germinated ancient date seedlings.

Ages in months at time of photograph (A to C) Adam (110 months), Jonah (63 months), and Uriel (54 months). (D to F) Boaz (54 months), Judith (47 months), and Hannah (88 months). Photo credit: Guy Eisner.
On visual inspection, no specific observation linked the ability of these seeds to germinate compared with those that failed to germinate. Before planting, the ancient date seeds had been weighted, and their length was measured, with the exception of those seeds from Masada, (including Adam, the germinated seed), which unfortunately were not measured (table S1). No statistically significant differences were found between germinated and ungerminated seeds in either weight {1.67 ± 0.55 and 1.61 ± 0.29 g, respectively [Student’s t test (t) = 0.348, degree of freedom (df) = 24, P = 0.731]} or length [27.60 ± 3.7 and 26.8 ± 3.7 mm, respectively (t = 0.455, df = 24, P = 0.653)].
Radiocarbon dating
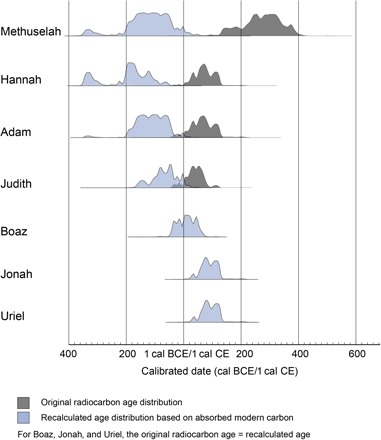
Radiocarbon ages are shown (Fig. 3 and table S2) for ancient date seeds germinated in the current study and also for the date seed (“seed 3”/“Methuselah”) germinated in our previous work (6). These ages were obtained from seed shell fragments found clinging to the rootlets of germinated seedlings during their transfer into larger pots (3 to 17 months of age). The values were recalculated to take into account contamination by modern carbon incorporated during seedling growth previously shown to reduce measured radiocarbon age by approximately 250 to 300 years, equivalent to 2 to 3% modern carbon (table S2) (6). On the basis of these calculations, Methuselah germinated in our previous study (6) and Hannah and Adam in the current study are the oldest samples (first to fourth centuries BCE), Uriel and Jonah are the youngest (first to second centuries CE), and Judith and Boaz are intermediate (mid-second century BCE to mid-first century CE) (Fig. 3).
Fig. 3. Original and recalculated radiocarbon age distribution of germinated date seeds [current and previous studies (six) taking into account the absorbed percentage of modern carbon].
Morphometric study of ungerminated ancient date seeds
Eighteen ancient date seeds that failed to germinate were recovered from the potting soil and compared with modern seeds derived from 57 current date palms of which 48 are cultivated varieties and 9 are wild individuals (9, 10). Ancient seeds were significantly larger in terms of both length and width (length, 27.62 ± 3.96 mm; width, 10.38 ± 0.71 mm) than both current cultivar (length, 20.60 ± 4.70 mm; width, 8.33 ± 1.02 mm) and wild date palm seeds (length, 16.69 ± 3.39 mm; width, 7.08 ± 0.46 mm) (Fig. 4). Ancient seeds were, on average, 27.69% wider (t = −11.923, df = 18.391, P = 2.157 × 10−10) and 38.37% longer than the combined current samples (wild and cultivated) (t = −7.422, df = 17.952, P = 3.564 × 10−7).
Fig. 4. Seed size comparison between ancient seeds from the Judean desert and modern date seeds.
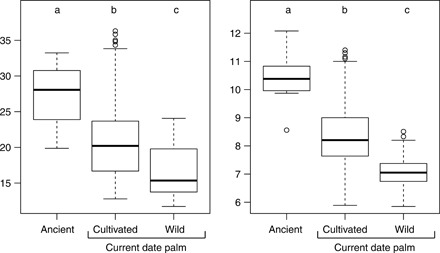
Length (millimeters) (left) and width (millimeters) (right) of ancient date seeds that failed to germinate (n = 18), 9 current wild individuals (n = 180), and 48 cultivated P. dactylifera varieties (n = 928). Letters a, b, and c above boxes indicate Tukey’s groups derived from HSD.test function and R package agricolae.
When only compared to the cultivars, the ancient date seeds were still larger: 24.55% wider (t = −11.923, df = 18.391, P = 2.157 × 10−10) and 34.06% longer (t = −7.422, df = 17.952, P = 3.564 × 10−7). However, the contrast in seed size is even more marked when comparing ancient seeds and current wild date palms: The Judean date palm seeds were, on average, 39.55% wider (t = −19.185, df = 18.471, P = 5.943 × 10−14) and 65.48% longer than current wild samples (t = −11.311, df = 19.574, P = 2.472 × 10−10) (tables S3 and S4).
Analysis of seed shape diversity in current and ancient date seeds using principal components analysis (PCA) (dudi.pca function) performed on seed outlines confirmed visual observation that modern cultivated seeds were more diverse in size than ancient ones but did not differentiate between the two groups [multivariate analysis of variance (MANOVA), P > 0.05]. Ancient seeds displayed an elongated shape similar to current cultivated samples (fig. S2).
Microsatellite genotyping
The sex of the six germinated ancient date seedlings in the current study identified using three sex-linked simple sequence repeats (SSR) (11) were as follows: Judith and Hannah are female genotypes and Uriel, Jonah, Boaz, Adam, and Methuselah (seed 3) from the previous study (6) are male genotypes. Through microsatellite genotyping, three levels of genetic inheritance were investigated to highlight geographic origins (Fig. 5, A and B): (i) inheritance transmitted by both parents to progeny, obtained by microsatellite markers showing western and eastern patterns of the ancient seed’s genomes (4), as presented in structure analysis and pie charts (Fig. 5A); (ii) inheritance transmitted from mother to progeny through the chloroplast genome, reflecting maternal lineage origin by reporting chloroplastic minisatellite eastern or western alleles (Fig. 5B, arrow) (12); and (iii) inheritance transmitted from father to son through the Y chromosome, reflecting paternal lineage origin by reporting male specific sex-linked eastern or western alleles (Fig. 5B, arrow) (11).
Fig. 5. Genetic analysis of the seven ancient seeds.
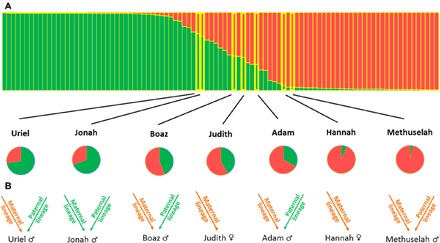
(A) Structure analysis results are shown for modern and ancient western (green) and eastern (orange) genotype contributions. Pie charts highlight eastern (orange) and western (green) ancient seeds nuclear genomes contributions. (B) Ancient seeds maternal and paternal lineages origin. Arrows represent clonally transmitted parental information, with maternal (chloroplastic) and paternal (Y chromosome) from western (green) and eastern (orange) origins.
Structure analysis revealed that distribution of the germinated ancient date seeds was within previously described eastern and western date palm gene pools (Fig. 5A). Methuselah, Hannah, and Adam are the most eastern genotypes, although they also show ancient western contributions requiring numerous generations and highlighting ancient crosses. Boaz and Judith are the most admixed, with almost equal eastern and western contributions reflecting more recent crossings. Jonah and Uriel are the most western genotypes with the most western parental lineages (Fig. 5B).
To shed light on genetic diversity of the ancient dates, basic population genetic parameters were estimated and compared to modern reference collections (tables S5 and S6). The ancient genotypes showed an allelic richness value (Ar) (i.e., the number of alleles) of 3.59, a relatively high diversity for such a small sample size (seven genotypes) compared to values of other countries sampled (table S6). Genetic relationships between the ancient date and current varieties (Fig. 6 and table S7) show Methuselah and Adam close to eastern modern varieties Fardh4 and Khalass, respectively, assigned to current Arabian Gulf varieties; Hannah and Judith related to modern Iraqi varieties Khastawi and Khyara, respectively; and Uriel, Boaz, and Jonah, the most western genotypes, related to modern Moroccan varieties, Mahalbit, Jihel, and Medjool, respectively.
Fig. 6. Genetic distances tree showing the relationships among modern date varieties and ancient germinated seedlings relying on neighbor-joining algorithm.
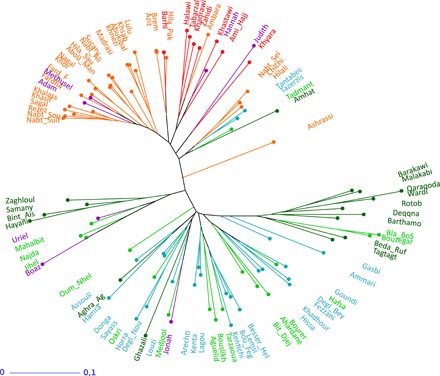
Modern varieties from United Arab Emirates (light orange), Iraq (red), Tunisia (blue), Morocco (light green), Egypt (dark green), and ancient genotypes (purple).
DISCUSSION
In the current study, six ancient date seeds, in addition to the seedling obtained in our previous study (6), were germinated. All the seeds were approximately 2000 years old and had been previously recovered from archaeological sites in the Judean desert, a rain shadow desert of ca. 1500 km2 located between the maquis-covered Judean Hills and the Dead Sea (fig. S1).
Little is known about the mechanisms determining seed longevity; however, it has been related to the ability to remain in a dry quiescent state (13). In the current study, low precipitation and very low humidity around the Dead Sea could have contributed to the longevity of the ancient date seeds, which may be an adaptation of date palms to extreme desert conditions fostering seed dispersion. Their remarkable durability, however, may also be connected to other extreme environmental conditions in this area; at 415 m below mean sea level, the Dead Sea and its surroundings have the thickest atmosphere on Earth, leading to a unique radiation regime and a complex haze layer associated with the chemical composition of the Dead Sea water (14). However, since no visible evidence in the current study was linked to seed germination and, accordingly, to their long term survival, further investigations are needed to understand the basis of date palm seed longevity.
Among the world’s oldest cultivated fruit trees, P. dactylifera is the emblematic of oasis agriculture and highly symbolic in Muslim, Christian, and Jewish religions (5). Closely connected to the history of human migrations, the first cultivated varieties of P. dactylifera are thought to have originated around Mesopotamia and the Upper Arabian Gulf some 6700 to 6000 yr B.P. (1, 2, 10). In Judea, an ancient geopolitical region that arose during the 11th century BCE in the southern part of the historic Land of Israel, and situated at the cross roads of Africa, Asia, and Europe, the origins of date palm cultivation are unknown. However, from historical records, a thriving Judean date culture was present around Jericho, the Dead Sea, and Jordan Valley from the fifth century BCE onward, benefitting from an optimal oasis agriculture environment of freshwater sources and subtropical climate (5).
Described by classical writers including Theophrastus, Herodotus, Galen, Strabo, Pliny the Elder, and Josephus, these valuable plantations produced dates attributed with various qualities including large size, nutritional and medicinal benefits, sweetness, and a long storage life, enabling them to be exported throughout the Roman Empire (5, 15, 16). Several types of Judean dates are also described in antiquity including the exceptionally large “Nicolai” variety measuring up to 11 cm (5, 15, 16).
In the current study, ancient seeds were significantly longer and wider than both modern date varieties and wild date palms. Previous research has established that both fruits and seeds are larger in domesticated fruit crops compared with their wild ancestors (17), suggesting that the ancient seeds were of cultivated origin (9, 18), most likely originating from the region’s date plantations. Furthermore, an increase in seed size has been linked allometrically to an increase in fruit size (19), corroborating the historical descriptions of the large fruits grown in this region.
Genotypes of the germinated ancient date seedlings cover a large part of present-day date palm distribution area, findings that reflect the variety, richness, and probable influences of the historic Judean date groves. Microsatellite genotyping shows a relatively high diversity, with eastern and western gene pool contributions, allelic richness, and genetic proximity to current varieties cultivated in the Arabian Peninsula, Iraq, and North Africa. Although the sample size is small, a predominance of eastern female lineages (six of seven) indicates that eastern female varieties grown from local germplasm were probably clonally propagated from offshoots to maintain desirable fruit qualities. Male lineages, mainly western (four of five), suggest that genetically different or “foreign” males were used for pollination. This assumption is supported by first century texts, indicating that substantial knowledge existed in ancient Judea 2000 years ago regarding the most suitable males for pollination of female date palms (20).
Our results reinforce the historical narrative that a highly sophisticated domestication culture existed in ancient Judea. Local farmers with an interest in maintaining genetic diversity in their date plantations and anthropogenic pressures leading to selection on fruit dimension and other desirable traits used cross-breeding with foreign (genetically different) males to develop a rich collection of varieties.
These findings suggest that Judean date culture was influenced by a variety of migratory, economic, and cultural exchanges that took place in this area over several millennia.
In Israel, the oldest remains of P. dactylifera are wood specimens 19,000 yr B.P. from Ohalo II site on the Sea of Galilee (21). Recovery of carbonized date seeds from Chalcolithic and Early Bronze Age sites (4500 to 2900 BCE) in the Judean desert, Jordan Valley, and Jericho (22, 23) and early Iron Age sites in Israel (12th to 11th century BCE) (24) suggest that human exploitation and consumption of dates occurred at this time. However, it is unclear whether these samples, which are relatively few in number and of very small size (22, 25, 26), are derived from ancient wild populations, as suggested by morphometric studies of modern wild date populations (18) or represent an early stage of the domestication process.
In the current study, although the sample size is too small to claim a trend, on a gradient from east to west genetic contributions, the older the germinated seeds are on radiocarbon dating (Fig. 3), the more eastern is the nuclear genome (Fig. 5, A and B ). In this respect, Methuselah, Adam, and Hannah (first to fourth centuries BCE) have a predominantly eastern nuclear genome and eastern maternal lineage, their relationship to modern varieties from the Arabian Gulf and Iraq suggesting that they belong to the same eastern genetic background.
The P. dactylifera cultivated by the inhabitants of Judea at that time therefore appears to be from the eastern gene pool, possibly growing locally and related to oasis populations, of which relict populations were recently found in Oman (9).
Elite female cultivars may also have been introduced to ancient Israel from these regions, consistent with a pattern of human intervention and possibly active acquisition of date palm varieties. Established trade links are documented with Arabia and the Persian Gulf from at least the 12th century BCE (27). Babylonian date palm cultivation in southern Mesopotamia (most of modern Iraq), originating some 6000 yr B.P. (1, 2), used deportees from ancient Judea following its conquest in the sixth century BCE (28). After the collapse of the Neo-Babylonian Empire, returning exiles may have brought this specialized knowledge and selected cultivars back to Judea; a date variety “Taali” cultivated in both Judea and Babylon is mentioned in the Talmud (29).
Western genetic admixtures in the germinated seedlings and their proximity to current cultivated date varieties from Morocco also suggest that ancient Judean date palms were the result of germplasm exchanges with this area and of multiple crosses. Introgression of eastern genomes into western ones are common, detected in varieties from Algeria, Morocco, Mauritania, and particularly east-west junction areas like Egypt (1, 2, 4, 30). In the latter, eastern contributions from the Persian Gulf, detected in ancient Egypt date seeds from 1400 BCE to 800 CE, reveal a chronological pattern of change in agrobiodiversity and the possible emergence of a western form in the Roman period (10).
Introgression of date palm western genomes into eastern ones, however, is far lower (1, 2, 4, 12), their presence in the current study reflecting west to east exchanges.
The origins of these exchanges are unclear; however, archaeological evidence indicates that North Africa, Near East, and Mediterranean cultures were clearly linked during the Neolithic in the southern Levant (approximately 11,700 to 7300 B.P.) and were associated in Jericho with the earliest origins of food production and fundamental changes in human subsistence strategies (31).
Phoenicia, a maritime trading nation occupying the coastal areas of modern northern Israel, Lebanon, and Syria (1500 to 300 BCE), was also historically associated with cultivation and trade of date palms (32). We can speculate that later west to east germplasm exchanges to this region may have been associated with domesticated varieties originating in Phoenician City States in North African (e.g., Carthage in present-day Tunisia) (32), where oasis agriculture appeared relatively late in the archaeological record (3).
The most western genotypes in the current study (Uriel and Jonah) are also the youngest seeds (mid-first to mid-second CE), coinciding with established trade routes linking this region to North Africa and supporting evidence for date consumption in the latter 2000 years ago (2, 3). This period coincides with Judea’s well-documented wars against Rome (66 to 73 CE and 132 to 136 CE) and deportation and displacement of its population (16). The ancient seeds in the current study were found in the Judean desert, historically a place of refuge due to its steep cliffs and inaccessible caves (16, 23). The loss of political autonomy and the final collapse of Judea have been postulated as causing major disruption to labor intensive practices associated with date cultivation (33). Elite cultivars no longer conserved by vegetative propagation (offshoots) were gradually replaced by seedling date palms producing fruits displaying considerable variation within the progeny. Although P. dactylifera can live for more than 100 years (33) and date groves in this region are thought to have persisted for several more centuries, they were already rare by the 11th century and had been entirely replaced by seedling populations or feral, wild trees producing only low-quality fruit (5, 33), by the 19th century.
The current study sheds light on the origins of the Judean date palm, suggesting that its cultivation, benefitting from genetically distinct eastern and western populations, arose from local or introduced eastern varieties, which only later were crossed with western varieties. These findings are consistent with Judea’s location between east-west date palm diversification areas, ancient centers of date palm cultivation, and the impact of human dispersal routes at this crossroads of continents.
Given its exceptional storage potentialities, the date palm is a remarkable model for seed longevity research. Investigations on the molecular mechanisms involved in long-term protection in the dried state have important implications on plant adaptation to changing environments and for biodiversity conservation and seed banking. As new information on specific gene-associated traits (e.g., fruit color and texture) (3) is found, we hope to reconstruct the phenotypes of this historic date palm, identify genomic regions associated with selection pressures over recent evolutionary history, and study the properties of dates produced by using ancient male seedlings to pollinate ancient females. In doing so, we will more fully understand the genetics and physiology of the ancient Judean date palm once cultivated in this region.
MATERIALS AND METHODS
Experimental design
The objectives of this study and its design were as follows:
1) The origin and selection of ancient date seeds derived from archaeological sites in the Judean desert.
2) The germination of ancient date seeds in a quarantine site following a preparatory process.
3) Radiocarbon dating and recalculation of calendar ages of germinated ancient date seeds based on seed shell fragments and selected controls.
4) Seed morphometric studies: Comparing ungerminated ancient date seeds with seeds from modern date varieties and wild date palms.
5) Microsatellite analysis of seven germinated date seedlings.
(statistical methods are included in the respective sections)
Origins, inspection, and selection of ancient date seeds from archaeological sites in the Judean desert
The ancient date seeds in the current study were obtained from botanical material recovered from archaeological excavations and surveys carried out at the following sites in the Judean desert between 1963 and 1991 and stored at room temperature since their discovery (fig. S1).
1) Masada: An ancient fortress/palace complex built by King Herod the Great (37 to 4 BCE) at the southern end of the Dead Sea on the site of an earlier Hasmonean fortification (141 to 37 BCE) (7). The site, built on a plateau approximately 400 m above the Dead Sea, was first excavated by the late Y. Yadin (Institute of Archaeology, Hebrew University, Jerusalem, Israel) from 1963 to 1965 (7). Bioarchaeological material found at this time included large numbers of date seeds buried under rubble close to the remains of an area identified as a food storage site.
2) Qumran: An archaeological site situated at the northern end of the Dead Sea including an ancient settlement dating from the second century BCE destroyed in 68 CE and a number of caves located in the surrounding cliffs and marl terrace associated with the 1947 discovery of the Dead Sea Scrolls. Later excavations and surveys of caves in this area, carried out from 1986 to 1989, by J. Patrich and B. Arubas (The Institute of Archaeology, The Hebrew University, Jerusalem, Israel) (8) included the following: Qumran Cave 13: artifacts found included potsherds from period 1b Qumran (until 31 BCE), numerous date stones and dried dates in a pit, and a pottery juglet dated to approximately 67 to 79 CE containing an unknown viscid substance and wrapped in palm fibers (used as a control in radiocarbon analysis in the current study) (see below); and Qumran Cave FQ37: containing a number of date stones and first to second CE century artifacts from the late Second Temple period (60 to 70 CE) and Roman period.
3) Wadi Makukh: A winter water channel in the Judean desert surrounded by high cliffs and containing a number of caves, which were surveyed from 1986 to 1989 (above). Date seeds found in caves 1, 3, 6, and 24 in this area were included in the current study; Cave 1 was found to include a Chalcolithic burial site (fifth millennium B.P.) containing human skeletons as well as Roman period artifacts but with signs of considerable disturbance by grave robbers (8).
4) Wadi Kelt: A winter water channel running from Jerusalem to the Dead Sea containing a number of caves (8). Date seeds from Masada were provided to S.S. by M. Kislev (Faculty of Life Sciences, Bar Ilan University), initially in 2005 (6) and again in 2007 (germinated in the current study), following permission by the late E. Netzer (Department of Archaeology, Hebrew University of Jerusalem). Date seeds from Qumran, Wadi Makukh, and Wadi Kelt were provided to S.S. by J. Patrich in 2009.
Out of a collection of many hundreds of ancient date seeds, a total of 34 were selected for the current study based on the specimens appearing visually to be intact whole seeds, in good condition, and without holes. They included Masada (8 seeds), Qumran (18 seeds), Wadi Makukh (7 seeds), and Wadi Kelt (1 seed). Ancient date seeds selected above were identified by code numbers and photographed, and measurements of weight and length were made before planting (with the exception of Masada seeds, which unfortunately were not measured) (table S1). One date seed, from the Qumran excavations (HU 37 A11), was selected as a control and left unplanted (table S1).
Preparatory process before planting ancient date seeds
The remaining 33 seeds were subjected to a preparatory process to increase the likelihood of seed germination using the following established methods to sprout delicate germplasm (34): seeds were initially soaked in water for 24 hours and in gibberellic acid (5.19 mM) (OrthoGrow, USA) for 6 hours to encourage embryonic growth. This was followed by Hormoril T8 solution (5 g/liter) (Asia-Riesel, Israel) for 6 hours to encourage rooting and KF-20 organic fertilizer (10 ml/liter) (VGI, Israel) for 12 hours. All solutions were maintained at 35°C.
Following the above procedure, one seed was found to be damaged and not planted. The remaining 32 seeds were separately potted in fresh sterile potting soil, 1 cm below the surface, and placed in a locked quarantine site at the Arava Institute of Environmental Sciences, Kibbutz Ketura, located in the southern Israel. Eight weeks after germination and periodically afterward, KF-20 (10 ml/liter) and iron chelate (10 g/liter) were added to the seedlings. Irrigation used desalinated water, as our previous study on germinating the first ancient date seed (6) indicated that using the region’s highly mineralized water produced “tip burn” (darkening and drying of leaves).
Radiocarbon dating
Radiocarbon ages in the current study were obtained for the following bioarchaeological material: (i) fragments of seed shell coat found clinging to the rootlets of six germinated ancient date seeds when these seedlings were transferred into larger pots, (ii) an unplanted ancient date seed from cave 37 Qumran (HU37 A11) (used as a control), and (iii) part of an ancient palm frond surrounding an oil juglet found in Qumran Cave 13 (used as a control). Radiocarbon ages of seed shell fragments from the germinated seedlings were recalculated to take into account modern carbon incorporated during seedling growth (6).
1) Methodology: Nonorganic carbon (carbonates) were removed from all samples with 10% HCl under reduced pressure followed by repeated washes in deionized water until neutral (pH 7). Organic acids formed during the rotting process were removed with 10% NaOH followed by repeated washes (as above). To prevent absorption of atmospheric CO2, all samples were placed again in 10% HCl and then washed in deionized water until neutral. To remove chemicals used in the germination process, a 7-mm-long shell fragment from the germinated date seed weighing 80 mg was cut into six cubes of 8 mm3 and subjected to an additional series of four boil washes. All samples were heated in an evacuated sealed quartz tube with CuO as an oxygen source. The resulting CO2 was mixed with hydrogen in the ratio 2.5:1 and catalytically reduced over cobalt powder at 550°C to elemental carbon (graphite). This mixture was pressed into a target and the 14C:12C ratio (for radiocarbon age) measured by accelerator mass spectrometry at the Institute for Particle Physics of the Swiss Federal Institute of Technology Zurich (ETHZ).
2) Calendar age: Calendar age was obtained using the OxCal 4.3 calibration program based on the latest IntCal 13 calibration curve (35). Calibrated calendar ages can be found with a probability of 68.3% in the 1σ-range and with a probability of 95.4% in the 2σ-range (table S2). The probability distribution P of individual ages is given for each sigma range. The 14C activity is reported as pMC (percentage of modern carbon) and corresponds to the ratio of the activity of the sample to the corrected activity of the oxalic acid standard, which has an age of 0 yr B.P.
3) Calculation of correction for pMC: The effect of contamination by modern carbon incorporated during seedling growth previously shown in our first germination of an ancient date seed to reduce measured age by 250 to 300 years (equivalent to 2 to 3% pMC) (6) was calculated using the following three groups based on the source of the ancient seeds in both the current and previous studies:
(i) Masada: Adam (current study), Methuselah (seed 3), and seed 1 [both from previous study (6) in which seed 1 was used as a control].
(ii) Qumran Cave 13: Judith and an ancient palm frond (used as a control)
(iii) Qumran Cave 37: Boaz, Jonah, Uriel, and seed HU37A11 (used as a control)
The germinated ancient seed Hannah from Wadi Makhukh was not assigned to a group due to the absence of a suitable control and considerable disruption to the site.
Using as age-controls the ancient palm frond (Qumran Cave 13), seed HU37A11 (Qumran Cave 37) from the current study and seed 1 (Masada) from the previous study (6), we assumed that a positive pMC difference between the germinated seeds and control sample could be attributed to modern carbon that was absorbed during germination. Ages of the germinated seeds were therefore recalculated (assuming that the measurement error remains unchanged) by adjusting the measured age to the control sample. For Hannah since no control exists, an average deviation (derived from the other samples) was taken into account.
Seed morphometric studies
Comparison of ancient date seeds that failed to germinate with modern date seeds
This was performed on the following groups:
1) Modern date seed (P. dactylifera) samples (n = 56): Being either from cultivated varieties (n = 47) or uncultivated and possibly wild individuals (n = 9) (9). Seeds from these sources (total n = 1108) were used as a current referential for seed morphometric analysis. The cultivated modern samples originated from 11 countries spanning date palm distribution from Spain to North Africa to the Middle-East. The candidate wild date palms originated from Oman and have been hypothesized as wild date palms based on seed shape, seed size (18), and genetic studies based on microsatellite and whole-genome resequencing data (9).
2) Ancient date seeds (n = 18): Of 26 ancient date seeds obtained from Qumran, Wadi Makukh, and Wadi Kelt archaeological sites (described above) that had been planted in the quarantine site, 21 failed to germinate and were retrieved from the potting soil. Of these, three were discarded as they had fragmented and were in poor condition. The remaining 18 retrieved ancient date seeds together with modern reference seeds (described above) were rephotographed on dorsal and lateral sides, and measurements of length and width were remade (table S3) [Neither current or previous (6) ancient date seeds from Masada that failed to germinate were used in the morphometric study as these seeds were not retrieved from the potting soil].
The following statistical analyses were performed using R software (36).
1) Size analysis of modern seeds: The length and width of a total of 1108 seeds obtained from 47 current cultivated varieties (928 seeds) and 9 current wild individuals (180 seeds) were measured using ImageJ (37) following the protocol previously established by Gros-Balthazard et al. (18). The thickness was not measured since it is highly correlated with width (18).
2) Comparison of seed size between current and ancient samples: Measurements for current varieties were compared with those measured for the ancient date seeds using boxplots and Student’s and Tukey’s tests (table S4).
3) Analysis of seed shape diversity in current and ancient date seeds: PCA (dudi.pca function) was performed on seed outlines assessed by Fourier coefficients, a morphometric method applied to outline analysis.
Genotyping of seedlings germinated from ancient date seeds
DNA preparation
DNA of six ancient date seedlings from the current study and one (Methuselah) from the previous study (6) was analyzed. A set of 19 SSR was used for genotyping as described by Zehdi-Azouzi et al. (4). Gender was determined using date palm sex-linked microsatellite markers (11). Maternal lineages were traced back using the plastid intergenic spacer psbZ-trnf minisatellite (12, 38). Paternal lineages were studied through Y haplotypes using the three sex-linked SSRs (mPdIRDP80, mPdIRDP50, and mPdIRDP52) (11).
Total cellular DNA was extracted from lyophilized leaves using the TissueLyser and the DNeasy Plant Mini Kit (QIAGEN SA, Courtaboeuf, France) according to the manufacturer’s instructions. After purification, DNA concentrations were determined using a GeneQuant spectrometer (Amersham Pharmacia Biotech, France). The quality was checked by agarose minigel electrophoresis. The resulting DNA solutions were stored at −20°C.
Amplification and genotyping
Polymerase chain reactions were performed in an Eppendorf (AG, Hamburg, Germany) thermocycler. Reaction was performed in 20 μl and contained 10 ng of genomic DNA, 10× reaction buffer, 2 mM MgCl2, 200 μM deoxynucleotide triphosphates, 0.5 U polymerase, and 0.4 pmol of the forward primer labeled with a 5′M13 tail, 2 pmol of the reverse primer, and 2 pmol of the fluorochrome-marked M13 tail and MilliQ water. A touchdown polymerase chain reaction (PCR) was carried out with following parameters: denaturation for 2 min at 94°C, followed by six cycles of 94°C for 45 s, 60°C for 1 min, and 72°C for 1 min; then 30 cycles of 94°C for 45 s, 55°C for 1 min, and 72°C for 1.5 min; then 10 cycles of 94°C for 45 min, 53°C for 1 min, 72°C for 1.5 min; and a final elongation step at 72°C for 10 min. PCR products were analyzed using an ABI 3130XL Genetic Analyzer (Applied Biosystems, Foster City, CA, USA). Allele size scoring was performed with GeneMapper software v3.7 (Applied Biosystems).
Genetic analyses
The ancient genotypes were compared to a reference matrix (90 genotypes) containing genotyping data on current date palm varieties covering the two genetic pools defined by Zehdi-Azouzi et al. (4) and including 35 samples from the eastern pool and 55 samples from the western pool (table S5). The number of alleles per group (NA), the number of alleles with a frequency higher than 5% (NA,P), and the observed (Ho), the expected (He) heterozygosities, and the fixation index values (FIS) were estimated using the GenAlEx 6.5 program (table S6). The allelic richness of each group was also calculated via the divBasic function implemented in the R package diversity (table S6) (39).
The hierarchical classifications were generated using PHYLIP package by calculating Cavalli-Sforza and Edwards distances (40) between ancient genotypes and current varieties (table S7). The obtained distance was used to construct the dendrogram using the neighbor-joining algorithm (41). The tree was drawn using DARwin software (42).
The membership probabilities of the ancient genotypes were identified by using a model-based clustering algorithm implemented in the computer program STRUCTURE v.2.3.4 (43). This algorithm identifies clusters (K) with different allele frequencies and assigns portions of individual genotypes to these clusters. It assumes the Hardy-Weinberg equilibrium and linkage equilibrium within clusters. The STRUCTURE algorithm was run without previous information on the geographic origin of the accessions using a model with admixture and correlated allele frequencies with 10 independent replicate runs for each K value (K value ranging from 1 to 6). For each run, we used a burn period of 10,000 iterations followed by 1 million iterations. The optimal number of clusters was assigned by using the run with the maximum likelihood validated with an ad hoc quantity based on the second-order rate of change in the log probability of data between different K values (fig. S3).The optimal alignment of the independent iterations was obtained by CLUMPP v.1.1 implemented in the Pophelper software v.1.0.10 (44); Pophelper v.1.0.10 (44) was also used to plot the results for the optimal K.
Supplementary Material
Acknowledgments
We thank J. Patrich and the late E. Netzer for making available ancient date seeds from Judean desert excavations; R. Krueger (USDA-ARS, USA) for providing some current date palm varieties; and S. Zehdi (Faculty of Sciences, University of Tunis El Manar, Tunisia), A. Lemansour (UAEU, DPDRUD, United Arab Emirates), M. A. Elhoumaizi (Sciences Faculty, Morocco), and C. Newton for allowing the use of genotyping data on current date palm varieties in the reference matrix. M. Collin is acknowledged for the help in the figure preparation and T. Bdolah Abraham for the help in statistics. O. Fragman-Sapir is acknowledged for identification of ancient date seeds and C. Yeres and A. Rifkin for information on Midrashic and Talmudic Jewish source material. Funding: The study was supported by donations to NMRC from The Charles Wolfson Charitable Trust (UK), G. Gartner and the Louise Gartner Philanthropic Fund (USA), and the Morris Family Foundation (UK). Author contributions: S.S. initiated, designed, and coordinated the study, procured ancient date samples, researched historical and archaeological information and integrated it with scientific findings, and wrote the paper. E.C. and N.C. performed genetic analyses on germinated seedlings. E.S. germinated ancient date seeds. M.E. performed radiocarbon analysis. M.G.-B., S.I., and J.-F.T. performed morphometric analysis. F.A. supervised genetic analyses and with E.C., M.G.-B., and M.E. helped write the manuscript. Competing interests: The authors declare that they have no competing interests. Data and materials availability: All data needed to evaluate the conclusions in the paper are present in the paper and/or the Supplementary Materials. Additional data related to this paper may be requested from the authors.
SUPPLEMENTARY MATERIALS
Supplementary material for this article is available at http://advances.sciencemag.org/cgi/content/full/6/6/eaax0384/DC1
Fig. S1. Map of Judean desert sites where ancient seeds were discovered.
Fig. S2. PCA comparing current and ancient date (P. dactylifera) seeds.
Fig. S3. Log probabilities between K values according to (44).
Table S1. Judean desert archaeological sources of ancient date seeds and their preliminary measurements before planting.
Table S2. Calibrated calendar ages of germinated ancient date seeds and controls.
Table S3. Size parameters of 18 ungerminated ancient date seeds retrieved from potting soil.
Table S4. Comparison of 18 ungerminated ancient date seeds with current date seeds from cultivated varieties and wild individual date palms.
Table S5. Date palm varieties used for the genotyping reference matrix to compare ancient genotypes with current varieties.
Table S6. Genetic diversity within ancient genotypes and geographic date palm groups.
Table S7. Genetic distances (Cavalli-Sforza and Edwards) between ancient genotypes and current varieties.
REFERENCES AND NOTES
- 1.Tengberg M., Beginnings and early history of date palm garden cultivation in the Middle East. J. Arid Environ. 86, 139–147 (2012). [Google Scholar]
- 2.Flowers J. M., Hazzouri K. M., Gros-Balthazard M., Mo Z., Koutroumpa K., Perrakis A., Ferrand S., Khierallah H. S. M., Fuller D. Q., Aberlenc F., Fournaraki C., Purugganan M. D., Cross-species hybridization and the origin of North African date palms. Proc. Natl. Acad. Sci. U.S.A. 116, 1651–1658 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Gros-Balthazard M., Hazzouri K. M., Flowers J. M., Genomic insights into date palm origins. Genes 9, 502 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zehdi-Azouzi S., Cherif E., Moussouni S., Gros-Balthazard M., Abbas Naqvi S., Ludeña B., Castillo K., Chabrillange N., Bouguedoura N., Bennaceur M., Si-Dehbi F., Abdoulkader S., Daher A., Terral J.-F., Santoni S., Ballardini M., Mercuri A., Ben Salah M., Kadri K., Othmani A., Littardi C., Salhi-Hannachi A., Pintaud J.-C., Aberlenc-Bertossi F., Genetic structure of the date palm (Phoenix dactylifera) in the Old World reveals a strong differentiation between eastern and western populations. Ann. Bot. 116, 101–112 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Goor A., The history of the date through the ages in the holy land. Econ. Bot. 21, 320–340 (1967). [Google Scholar]
- 6.Sallon S., Solowey E., Cohen Y., Korchinsky R., Egli M., Woodhatch I., Simchoni O., Kislev M., Germination, genetics and growth of an ancient date seed. Science 320, 1464 (2008). [DOI] [PubMed] [Google Scholar]
- 7.Yadin Y., The Excavation of Masada—1963/64: Preliminary Report. Isr. Explor. J. 15, 1–120 (1965). [Google Scholar]
- 8.J. Patrich, B. Agor, B. Arubas, Judean desert cave survey 1986-1987. Hadashot Arkheologiyot: Excavations and Surveys in Israel, 65–67(1989).
- 9.Gros-Balthazard M., Galimberti M., Kousathanas A., Newton C., Ivorra S., Paradis L., Vigouroux Y., Carter R., Tengberg M., Battesti V., Santoni S., Falquet L., Pintaud J. C., Terral J. F., Wegmann D., The discovery of wild date palms in Oman reveals a complex domestication history involving centers in the Middle East and Africa. Curr. Biol. 27, 2211–2218.e8 (2017). [DOI] [PubMed] [Google Scholar]
- 10.Terral J.-F., Newton C., Ivorra S., Gros-Balthazard M., de Morais C. T., Picq S., Tengberg M., Pintaud J.-C., Insights into the historical biogeography of the date palm (Phoenix dactylifera L.) using geometric morphometry of modern and ancient seeds. J. Biogeogr. 39, 929–941 (2012). [Google Scholar]
- 11.Cherif E., Zehdi S., Castillo K., Chabrillange N., Abdoulkader S., Pintaud J.-C., Santoni S., Salhi-Hannachi A., Glémin S., Aberlenc-Bertossi F., Male-specific DNA markers provide genetic evidence of an XY chromosome system, a recombination arrest and allow the tracing of paternal lineages in date palm. New Phytol. 197, 409–415 (2013). [DOI] [PubMed] [Google Scholar]
- 12.J.-C. Pintaud, S. Zehdi, T. Couvreur, S. Barrow, S. Henderson, F. Aberlenc-Bertossi, J. Tregear, N. Billotte, Species delimitation in the genus Phoenix (Arecaceae) based on SSR markers, with Emphasis on the Identity of the Date Palm (Phoenix dactylifera), in Diversity, Phylogeny and Evolution in the Monocotyledons, O. Seberg, G. Petersen, A. S. Barfod, J. I. Davis, Eds. (Aarhus Univ. Press, 2010), pp. 267–286. [Google Scholar]
- 13.Leprince O., Pellizzaro A., Berriri S., Buitink J., Late seed maturation: Drying without dying. J. Exp. Bot. 68, 827–841 (2017). [DOI] [PubMed] [Google Scholar]
- 14.Kottmeier C., Agnon A., Al-Halbouni D., Alperti P., Corsmeier U., Dahm T., Eshel A., Geyer S., Haas M., Holohan E., Kalthoff N., Kishcha P., Krawczyk C., Lati J., Laronne J. B., Lott F., Mallast U., Merz R., Metzger J., Mohsen A., Morin E., Nied M., Rödiger T., Salameh E., Sawarieh A., Shannak B., Siebert C., Weber M., New perspectives on interdisciplinary earth science at the Dead Sea: The DESERVE project. Sci. Total Environ. 544, 1045–1058 (2016). [DOI] [PubMed] [Google Scholar]
- 15.Pliny the Elder, Natural History, The different varieties of Palm Trees and their characteristics, J. Bostock, H. T. Riley, Eds. Book XIII, chap. 9; www.perseus.tufts.edu/hopper/text.
- 16.Josepus, The Jewish War, G. Cornfeld, B. Mazar, P. Maier, Eds. (Zondervan, 1982), Book IV, chap. 8, pp. 296–299, and Book VII, chap. 8, pp. 488. [Google Scholar]
- 17.Fuller D. Q., Long and attenuated: Comparative trends in the domestication of tree fruits. Veg. Hist. Archaeobot. 27, 165–176 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Gros-Balthazard M., Newton C., Ivorra S., Pierre M.-H., Pintaud J.-C., Terral J.-F., The domestication syndrome in Phoenix dactylifera seeds: Toward the identification of wild date palm populations. PLOS ONE 11, e0152394 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bolmgren K., Eriksson O., Seed mass and the evolution of fleshy fruits in angiosperms. Oikos 119, 707–718 (2010). [Google Scholar]
- 20.Midrash Bamidbar Rabba (Numbers Rabbah): 3, 1, 31.
- 21.N. Liphschitz, D. Nadel, Charred Wood Remains from Ohalo II (19,000 B.P.), Sea of Galilee, Israel, Mitekufat Haeven: Journal of the Israel Prehistoric Society, 5-18 (1997). [Google Scholar]
- 22.Western A. C., The ecological interpretation of ancient charcoals from Jericho. Levant 3, 31–40 (1971). [Google Scholar]
- 23.P. Bar-Adon, The Cave of the Treasure: The Finds from Nahal Mishmar (Israel Exploration Society, 1980). [Google Scholar]
- 24.Frumin S., Maeir A. M., Horwitz L. K., Weis E., Studying ancient anthropogenic impacts on current floral biodiversity in the Southern Levant as reflected by the Philistine migration. Sci. Rep. 5, 13308 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Liphschitz N., Bonani G., Wild and cultivated date palm (Phoenix Dactylifera) from Qumran Cave 24. Tel Aviv 28, 305–309 (2001). [Google Scholar]
- 26.R. Burleigh, K. M. Kenyon, T. A. Holland; British School of Archaeology, Excavations at Jericho. Vol. 5: The Pottery Phases of the Tell and other Finds (Jerusalem British School of Archaeology, 1983), appendix B, pp. 589. [Google Scholar]
- 27.Finkelstein I., Arabian trade and socio-political conditions in the Negev in the twelfth-eleventh centuries BCE. J. Near East Stud. 47, 241–252 (1988). [Google Scholar]
- 28.H. H. Ben Sasson, Lands of the Diaspora in A History of the Jewish People (Harvard Univ. Press, 1976), pp. 373–376. [Google Scholar]
- 29.H. Freedman, I. Epstein, Eds. Babylonian Talmud, Shabbath (Traditional Press, 1947), vol. 2, pp. 219. [Google Scholar]
- 30.Mathew L. S., Seidel M. A., George B., Mathew S., Spannagl M., Haberer G., Torres M. F., Al-Dous E. K., Al-Azwani E. K., Diboun I., Krueger R. R., Mayer K. F. X., Mohamoud Y. A., Suhre K., Malek J. A., A genome-wide survey of date palm cultivars supports two major subpopulations in Phoenix dactylifera. G3 5, 1429–1438 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Twiss K. C., The neolithic of the southern Levant evolutionary. Anthropol. Forum 16, 24–35 (2007). [Google Scholar]
- 32.M. E. Aubet, Phoenician colonies in the central Mediterranean, in The Phoenicians and the West: Politics, Colonies and Trade (Cambridge Univ. Press, 2001), pp. 212–259. [Google Scholar]
- 33.Y. Cohen, B. Glasner, Date Palm Status and Perspective in Israel, in Date Palm Genetic Resources and Utilization: Vol 2 Asia and Europe, J. M. Al-Khayri, S. M. Jain, D. V. Johnson, Eds. (Springer, 2015), chap. 8, pp. 265–268. [Google Scholar]
- 34.H. T. Hartman, D. Kester, Plant Propagation: Principals and Practice (Prentice-Hall, 1975), pp. 111–138, 212–231. [Google Scholar]
- 35.Reimer P. J., Bard E., Bayliss A., Beck J. W., Blackwell P. G., Bronk-Ramsey C., Buck C. E., Cheng H., Edwards R. L., Friedrich M., Grootes P. M., Guilderson T. P., Haflidason H., Hajdas I., Hatté C., Heaton T. J., Hoffmann D. L., Hogg A. G., Hughen K. A., Kaiser K. F., Kromer B., Manning S. W., Nui M., Reimer R. W., Richards D. A., Scott E. M., Southon J. R., Staff R. A., Turney C. S. M., van der Plicht J., IntCal13 and Marine13 radiocarbon age calibration curves 0–50,000 years cal BP. Radiocarbon 55, 1869–1887 (2013). [Google Scholar]
- 36.R Core Team, R: A language and environment for statistical computing (R Foundation for Statistical Computing) (2014).
- 37.Abramoff M. D., Magalhães P. J., Ram S. J., Image processing with ImageJ. Biophotonics Int. 11, 36–42 (2004). [Google Scholar]
- 38.Henderson S. A., Billotte N., Pintaud J.-C., Genetic isolation of Cape Verde Island Phoenix atlantica (Arecaceae) revealed by microsatellite markers. Conserv. Genet. 7, 213–223 (2006). [Google Scholar]
- 39.Keenan K., McGinnity P., Cross T. F., Crozier W. W., Prodöhl P. A., diveRsity: An R package for the estimation of population genetics parameters and their associated errors. Methods Ecol. Evol. 4, 782–788 (2013). [Google Scholar]
- 40.Cavalli-Sforza L. L., Edwards A. W. F., Phylogenetic analysis: Models and estimation procedures. Evolution 21, 550–570 (1967). [DOI] [PubMed] [Google Scholar]
- 41.Saitou N., Nei M., The neighbor-joining method: A new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 4, 406–425 (1987). [DOI] [PubMed] [Google Scholar]
- 42.X. Perrier, J. P. Jacquemoud-Collet, DARwin software (2006).
- 43.Pritchard J. K., Stephens M., Donnelly P., Inference of population structure using multilocus genotype data. Genetics 155, 945–959 (2000). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Francis R. M., POPHELPER: An R package and web app to analyze and visualize population structure. Mol. Ecol. Resour. 17, 27–32 (2017). [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary material for this article is available at http://advances.sciencemag.org/cgi/content/full/6/6/eaax0384/DC1
Fig. S1. Map of Judean desert sites where ancient seeds were discovered.
Fig. S2. PCA comparing current and ancient date (P. dactylifera) seeds.
Fig. S3. Log probabilities between K values according to (44).
Table S1. Judean desert archaeological sources of ancient date seeds and their preliminary measurements before planting.
Table S2. Calibrated calendar ages of germinated ancient date seeds and controls.
Table S3. Size parameters of 18 ungerminated ancient date seeds retrieved from potting soil.
Table S4. Comparison of 18 ungerminated ancient date seeds with current date seeds from cultivated varieties and wild individual date palms.
Table S5. Date palm varieties used for the genotyping reference matrix to compare ancient genotypes with current varieties.
Table S6. Genetic diversity within ancient genotypes and geographic date palm groups.
Table S7. Genetic distances (Cavalli-Sforza and Edwards) between ancient genotypes and current varieties.


