Abstract
Background:
Per- and polyfluoroalkyl substances (PFASs) are a growing public health concern. Some longer chain PFASs bioaccumulate and many compounds persist in the environment for long time periods. Recent studies have established their ability to pass through placenta, yet data on the transplacental transfer efficiency and partitioning of short and long chain PFASs in blood matrices are limited.
Objectives:
To assess predictors of the partitioning of 17 PFAS compounds detected in the maternal serum, umbilical cord serum and whole cord blood samples from matched mother-newborn pairs from two Faroe Islands cohorts.
Methods:
We examined 151 mother-newborn pairs from two successive Faroese birth cohorts. cord:maternal serum (transplacental transfer) and serum:whole cord blood (blood partitioning) ratios were estimated for 17 PFAS compounds. We also examined the relationships of these ratios with maternal, newborns’, and physico-chemical properties using multivariable regression analyses.
Results:
Moderate to high correlations were observed between maternal and cord serum PFAS concentrations (ρ: 0.41 to 0.95), indicating significant transfer of these compounds from the mother to the fetus. Median transplacental transfer ratios were generally below 1, except for perfluorooctane sulfonamide (FOSA), and ranged between 0.36 for perfluorodecanoate (PFDA) and perfluoroundecanoate (PFUnDA) and 1.21 for FOSA. Most PFASs exhibited a preference to the serum component of the blood, except FOSA and perfluoroheptanoate (PFHpA), with blood partitioning ratios ranging from 0.36 for FOSA to 2.75 for PFUnDA. Both the functional groups and carbon chain length of different PFASs were important predictors of transplacental transfer and blood partitioning. We observed a U-shaped relationship between transplacental transfer ratios and carbon chain length for perfluorocarboxylates and perfluorosulfonates. Importantly, gestational diabetes was also a strong predictor of transplacental transfer ratios, with significantly higher transfer in mothers with gestational diabetes.
Conclusions:
Our findings provide a better understanding of the transplacental transfer and blood partitioning of a large number of PFAS compounds. Results elucidate the importance of chemical structure for future risk assessments and choice of appropriate blood matrices for measurement of PFAS compounds.
BACKGROUND
Per- and polyfluoroalkyl substances (PFASs) are a group of synthetically produced chemicals that have been widely used since the 1950s for industrial purposes and in consumer products (Fromme et al. 2009; Stahl et al. 2011). PFASs are ubiquitous in the environment and have been detected in both wildlife and humans worldwide (Birnbaum and Grandjean 2015). Human exposure sources for PFASs are diverse and include dust, consumer products, seafood consumption, and drinking water (Christensen et al. 2017; Hu et al. 2016; Schaider et al. 2017; Vestergren et al. 2012).
PFASs are highly persistent in the environment, some are known to bioaccumulate, and toxic effects associated with exposure have been widely reported (Birnbaum and Grandjean 2015; Liew et al. 2018). Indeed, the biological half-lives of the most studied PFAS compounds range between 3 and 7 years (Perfluorooctanoic acid [PFOA] 3.5 years; Perfluorononanoate [PFNA] 1.5; Perfluorodecanoate [PFDA] 4.2; Perfluoroundecanoate [PFUnDA] 4.4; Perfluorohexane sulfonate [PFHxS] 7.1; Perfluorooctane sulfate [PFOS] 4.8 years) (Li et al. 2018; Olsen et al. 2009; Papadopoulou et al. 2015). Recent studies have shown that shorter carbon chain PFASs such as Perfluorobutane sulfonate (PFBS) are generally excreted more rapidly than long-chain PFASs (Zhang et al. 2013), with the exception of PFHxS (Betts 2007). Their ubiquitous presence in humans and the environment along with their long half-lives represent a growing public health concern. Concerns about impacts of these chemicals on public health led to the phase out of production of PFOS and related compounds and PFOA by their major global manufacturer in 2002, and the inclusion of PFOS in the Stockholm Convention on Persistent Organic Pollutants (Buck et al. 2011). However, newer PFAS compounds, especially short chain ones have been introduced more recently (CECBP 2015; Wang et al. 2013).
Available evidence suggests that the toxicity of PFASs increases with increasing carbon chain length (Kudo et al. 2006; Stahl et al. 2011; Upham et al. 1998). Experimental studies on the toxicity of PFASs indicated the potential for developmental toxicity, immunotoxicity, hepatoxicity, and impacts on hormonal balance and carcinogenic potency (Fromme et al. 2009). In humans, a large number of epidemiologic studies investigated associations between exposure to PFASs and adverse health effects, especially in children. While there is a stronger evidence for potential effects of PFASs on immune responsiveness and dyslipidemia, findings are less conclusive for other health outcomes such as asthma, allergies, metabolic function, and cognitive and behavioral deficits (ATSDR 2018; Liew et al. 2018; Rappazzo et al. 2017).
Previous studies have measured concentration of PFASs in maternal and umbilical cord blood samples and concluded that PFASs cross the placental barrier and reach the fetus (Hanssen et al. 2013; Kim et al. 2011; Zhang et al. 2013; Zhao et al. 2017). Transplacental transfer of PFASs may depend on physical-chemical properties, and measurements of PFASs in matched maternal–fetal blood suggests that the transplacental transfer is lower for long-chain PFASs compared to short chain PFASs (Fromme et al. 2010; Zhang et al. 2013). However, most of the studies investigating transplacental transfer were based on relatively small sample sizes (n<70). Additionally, prior studies relied on maternal PFAS measured early in pregnancy, but transfer ratios measured at the time of delivery could reflect more accurately the transplacental effect of the entire pregnancy process (Zhao et al. 2017).
Serum (or plasma) concentrations of PFASs are commonly used in biomonitoring studies and are preferred to whole blood matrix (Olsen et al. 2007). However, a study comparing concentrations between blood components indicate that different compounds may be distributed differently between plasma and whole blood cells (Olsen et al. 2007). Hence, there is a need for clarifying the best blood compartments to be used for exposure assessment of different PFAS compounds.
The objective of this study was to compare the measurements of 17 PFAS concentrations in the maternal serum, umbilical cord sera and cord whole blood samples in 151 matched mother-child pairs from two Faroese birth cohorts. We additionally investigated the potential influence of PFASs physico-chemical properties on their transplacental transfer and partitioning between serum and whole blood. Finally, we investigated the potential influence of maternal and newborn characteristics on the transplacental transfer and partitioning of PFASs between serum and whole blood.
METHODS
Study Population
We included 151 mother-newborn pairs from two successive Faroese Birth Cohorts. The cohorts consisted of 100 singleton births from 1999 to 2001 (Cohort 3) and 51 singleton births from 2008 to 2009, (Cohort 5) respectively. Earlier investigations from these cohorts have determined that pilot whale and consumer products are the main source of exposure to persistent organic chemicals in this population (Dassuncao et al. 2018; Grandjean et al. 1995; Schlezinger et al. 2010; Weihe et al. 2008). Standard questionnaires were used to record past medical history, current health, and social factors during and before pregnancy. Relevant obstetric information, including birth weight, parity, maternal age, gestational diabetes, and gestational age were abstracted from hospital’s medical records. The study protocol was reviewed and approved by the Faroese ethics review committee and the institutional review board at the Harvard T.H. Chan School of Public Health. Details of these birth cohorts can be found elsewhere (Weihe and Debes Joensen 2012).
Analysis of PFASs in serum and whole blood
Blood samples were collected and processed to sera at the data collection site. Maternal PFAS concentrations were measured in maternal serum collected from the mothers during late pregnancy (32nd week of pregnancy) in Cohort 3 and two weeks after parturition in Cohort 5.
The maternal serum, cord serum, and cord whole blood samples were analyzed for 17 PFAS concentrations (ng/mL), including 9 Perfluorocarboxylates (PFCAs): Perfluorobutanoate (PFBA), Perfluoro-n-pentanoic acid (PFPeA), Perfluorohexanoate (PFHxA), Perfluoroheptanoate (PFHpA), PFOA, PFNA, PFDA, PFUnDA, and Perfluorododecanoic acid (PFDoDA); 5 PerfluoroSulfonates (PFSAs): PFBS, PFHxS, Perfluorohexanesulfonate (PFHpS), PFOS (both linear [n] and branched [br] isomers), and Perfluorodecane sulfonate (PFDS); and 3 precursors (perfluoroalkane sulfonyl fluoride based substances): perfluorooctane sulfonamide (FOSA: both linear [n] and branched [br] isomers), perfluorooctane sulfonamidoacetic acid (NMeFOSAA), and N-ethyl derivative of perfluorooctane sulfonamidoacetic acid (NEtFOSAA). PFAS concentrations were measured using online solid-phase extraction (SPE) and analyzed using high-pressure liquid chromatography with tandem mass spectrometry (Haug et al. 2009). The limits of detection (LOD) ranged between 0.03 – 0.1 ng PFAS /mL. The whole blood samples were pretreated with the zinc-sulphate in order to precipitate proteins and blood cells prior to the online SPE. A volume of 150 μL whole blood and 150 μL 0.06 M ZnSO4 in methanol were whirl mixed for 30 sec with 30 μL of 20 ng/mL isotopically labelled PFAS analogues (internal standard). The sample was here after centrifuged at 13,500 RPM for 20 minutes, and 160 μL of the supernatant was transferred to a polypropylene vial with 400 μL 0.1 M formic acid and whirl mixed prior to injection of 400 μL onto the SPE column. The rest of the of the analysis were in accordance with the protocol for the serum samples. The accuracy and reliability of the data was ensured by including, in each analytical series, quality control serum samples, calibration standards, and reagent and serum blanks. Within-batch and between-batch coefficient of variations for the serum samples were better than 8.9% and 12.9 % for all analytes, whereas the whole blood analysis varied up to 14.9 %.
Statistical Analysis
All PFAS concentrations were log2-transformed to approximate a Gaussian distribution. Geometric means (GMs), geometric standard deviation (GSD), and Interquartile range (IQR) were used to summarize their distributions. Pearson correlations of log2-transformed PFAS concentrations were used to describe the correlations between maternal and cord serum, and between cord serum and whole cord blood PFAS concentrations.
The mother-child PFAS measurement pairs were summarized as cord:maternal serum concentration ratios for compounds detected in >10% in cohort 3 and >20% in cohort 5 (to insure a minimum of 10 samples) in both cord and maternal serum to determine their transplacental transfer. Cord serum:whole cord blood ratios were used to estimate PFASs partitioning between serum and whole blood to determine PFASs partitioning. All values below detection limit were replaced by LOD/√2. GMs and GSDs of the PFAS concentration ratios were used to summarize their distributions. Scatter plots were used to display correlations, whereas box plots were used to display cord:maternal serum and serum:whole cord blood PFAS ratios.
To investigate potential determinants of the estimated ratios, we ran multivariable regression models for the transplacental transfer and partitioning ratios for compound including newborn’s sex, birth weight (in grams), maternal age at delivery (in years), gestational age (in weeks), gestational diabetes, parity, smoking and alcohol consumption during pregnancy, and cohort. Additionally, to investigate physico-chemical properties as potential determinants of transplacental transfer and partitioning ratios, we ran multivariable regressions including transplacental transfer and partitioning ratios for all compounds (regardless of the compound) as outcomes, and PFAS functional group (PFCA, PFSA, or sulfonamide precursors) and carbon chain length as the predictors of interest, while adjusting for the same set of covariates above. To take into account any potential non-linearities in the relationship between carbon chain length and transplacental and partitioning ratios, we used generalized additive models (GAM) (Hastie and Tibshirani 1990) including carbon chain length as thin plate regression spline.
All statistical tests were two-sided, and significance was set at p<0.05. All analyses were performed using STATA and R software packages (StataCorp 2013; Team 2013).
RESULTS
Descriptive statistics.
Main characteristics of the study population are presented in supplemental material (Table S1). Newborns included in this study were predominantly male (58%). Mean birth weight was 3752 grams, and mean maternal age at delivery was 30.4 years. Mean gestational age was 39.8 weeks, prevalence of gestational diabetes was 7%, and most of the mothers had a previous child (77%). About 19% of mothers reported smoking and 28% reported having consumed alcohol during pregnancy. Table 1 shows the summary statistics of PFAS concentrations in maternal serum, cord serum, and whole cord blood in both cohorts 3 and 5. Overall, >20% detection frequencies were observed for 11 of the 17 PFASs in cohorts 3 and 5. PFBA and PFBS were not detected in any of the blood samples. PFPeA and PFHxA were detected in less than 10% of samples in both cohorts. PFDS was detected in less than 10% in cohort 3 samples, and in less than 20% in cohort 5 cord samples. Consequently, PFBA, PFBS, PFPeA, PFHxA, and PFDS were excluded from further analyses. All other PFASs had more than 10 mother-child pairs with quantified concentrations of PFAS for the analyses of both transplacental and partitioning ratios, except PFDoDA which had only 9 samples for transplacental transfer ratios (See tables S2 and S3 for detailed sample sizes).
Table 1.
Geometric means (GMs) and interquartile ranges (IQR) of PFAS concentrations (ng/mL) in maternal and cord serum, and cord whole blood for matched mother-child pairs (n=151). CS: cord serum; GSD: geometric standard deviation; LOD: limit of detection; MS: maternal serum; WCB: whole cord blood.
| Group | C Chain length |
PFAS | Matrix | Cohort 3 (1999-2001; n=100) | Cohort 5 (2008-2009; n=51) | ||||
|---|---|---|---|---|---|---|---|---|---|
| % >LOD |
GM(GSD), (ng/mL) |
IQR | % >LOD |
GM(GSD), (ng/mL) |
IQR | ||||
| PFCAs | C4 | PFBA | MS | 0 | - | - | 0 | - | - |
| CS | 0 | - | - | 0 | - | - | |||
| WCB | 0 | - | - | 0 | - | - | |||
| C5 | PFPeA | MS | 9 | - | - | 0 | - | - | |
| CS | 1 | - | - | 0 | - | - | |||
| WCB | 1 | - | - | 0 | - | - | |||
| C6 | PFHxA | MS | 1 | - | - | 0 | - | - | |
| CS | 3 | - | - | 0 | - | - | |||
| WCB | 2 | - | - | 0 | - | - | |||
| C7 | PFHpA | MS | 44 | 0.02 (0.001) | <LOD-0.11 | 6 | - | - | |
| CS | 35 | 0.02 (0.0003) | <LOD-0.06 | 0 | - | - | |||
| WCB | 29 | 0.02 (0.0004) | <LOD-0.08 | 2 | - | - | |||
| C8 | PFOA | MS | 100 | 2.33 (0.12) | 1.79-3.29 | 100 | 1.03 (0.08) | 0.75-1.41 | |
| CS | 100 | 1.97 (0.10) | 1.42-2.76 | 100 | 0.81 (0.07) | 0.56-1.26 | |||
| WCB | 100 | 1.08 (0.05) | 0.8-1.45 | 100 | 0.41 (0.03) | 0.29-0.59 | |||
| C9 | PFNA | MS | 100 | 0.48 (0.03) | 0.32-0.71 | 100 | 0.81 (0.04) | 0.62-1 | |
| CS | 100 | 0.27 (0.02) | 0.17-0.4 | 100 | 0.45 (0.03) | 0.34-0.65 | |||
| WCB | 100 | 0.14 (0.01) | 0.08-0.19 | 100 | 0.20 (0.01) | 0.15-0.26 | |||
| C10 | PFDA | MS | 100 | 0.29 (0.02) | 0.19-0.42 | 100 | 0.33 (0.02) | 0.25-0.41 | |
| CS | 100 | 0.11 (0.01) | 0.07-0.15 | 100 | 0.11 (0.01) | 0.09-0.14 | |||
| WCB | 86 | 0.05 (0.003) | 0.04-0.08 | 88 | 0.05 (0.004) | 0.04-0.07 | |||
| C11 | PFUnDA | MS | 100 | 0.55 (0.04) | 0.35-0.84 | 100 | 0.26 (0.02) | 0.2-0.39 | |
| CS | 100 | 0.19 (0.01) | 0.12-0.31 | 94 | 0.10 (0.01) | 0.06-0.15 | |||
| WCB | 73 | 0.05 (0.01) | <LOD-0.11 | 25 | 0.02 (0.002) | <LOD-0.03 | |||
| C12 | PFDoDA | MS | 41 | 0.03 (0.003) | <LOD-0.06 | 2 | - | - | |
| CS | 10 | 0.02 (0.001) | <LOD-<LOD | 57 | 0.05 (0.008) | <LOD-0.12 | |||
| WCB | 18 | 0.02 (0.001) | <LOD-<LOD | 33 | 0.03 (0.004) | <LOD-0.7 | |||
| PFSAs | C4 | PFBS | MS | 0 | - | - | 0 | - | - |
| CS | 0 | - | - | 0 | - | - | |||
| WCB | 0 | - | - | 0 | - | - | |||
| C6 | PFHxS | MS | 96 | 0.48 (0.04) | 0.39-0.69 | 100 | 0.47 (0.03) | 0.35-0.63 | |
| CS | 66 | 0.13 (0.02) | <LOD-0.52 | 100 | 0.27 (0.02) | 0.19-0.4 | |||
| WCB | 84 | 0.12 (0.01) | 0.1-0.21 | 92 | 0.12 (0.01) | 0.1-0.18 | |||
| C7 | PFHpS | MS | 100 | 0.19 (0.01) | 0.1-0.32 | 86 | 0.07 (0.01) | 0.05-0.13 | |
| CS | 100 | 0.11 (0.01) | 0.07-0.18 | 39 | 0.03 (0.003) | <LOD-0.05 | |||
| WCB | 65 | 0.04 (0.003) | <LOD-0.06 | 8 | - | - | |||
| C8 | Total PFOS | MS | 100 | 23.8 (1.2) | 15.8-36.9 | 100 | 8.82 (0.51) | 6.94-11.6 | |
| CS | 100 | 9.50 (0.49) | 6.34-13.89 | 100 | 3.09 (0.22) | 2.31-4.42 | |||
| WCB | 100 | 4.90 (0.26) | 3.33-6.94 | 100 | 1.60 (0.11) | 1.18-2.32 | |||
| brPFOS | MS | 100 | 8.15 (0.42) | 5.22-12.58 | 100 | 3.18 (0.2) | 2.35-4.33 | ||
| CS | 100 | 3.50 (0.18) | 2.38-4.94 | 100 | 1.17 (0.09) | 0.88-1.73 | |||
| WCB | 100 | 1.90 (0.10) | 1.35-2.73 | 100 | 0.63 (0.05) | 0.45-0.9 | |||
| nPFOS | MS | 100 | 15.6 (0.8) | 10.5-22.96 | 100 | 5.55 (0.33) | 4.16-7.45 | ||
| CS | 100 | 5.98 (0.31) | 3.97-8.71 | 100 | 1.89 (0.14) | 1.46-2.84 | |||
| WCB | 100 | 2.97 (0.16) | 1.98-4.36 | 100 | 0.96 (0.07) | 0.74-1.38 | |||
| C10 | PFDS | MS | 9 | - | - | 27 | 0.02 (0.002) | <LOD-0.04 | |
| CS | 3 | - | - | 18 | - | - | |||
| WCB | 4 | - | - | 12 | - | - | |||
| Precursors | C8 | Total FOSA | MS | 97 | 0.16 (0.01) | 0.10-0.27 | 14 | - | - |
| CS | 99 | 0.19 (0.01) | 0.12-0.30 | 10 | - | - | |||
| WCB | 100 | 0.55 (0.05) | 0.29-0.99 | 25 | 0.02 (0.002) | <LOD-0.03 | |||
| brFOSA | MS | 85 | 0.05 (0.004) | 0.03-0.09 | 14 | - | - | ||
| CS | 89 | 0.06 (0.005) | 0.04-0.09 | 10 | - | - | |||
| WCB | 99 | 0.26 (0.02) | 0.13-0.49 | 25 | 0.02 (0.002) | <LOD-0.03 | |||
| nFOSA | MS | 95 | 0.11 (0.01) | 0.08-0.18 | 14 | - | - | ||
| CS | 99 | 0.12 (0.01) | 0.08-0.2 | 10 | - | - | |||
| WCB | 99 | 0.29 (0.03) | 0.15-0.5 | 25 | 0.02 (0.001) | <LOD-0.02 | |||
| C11 | NMeFOSAA | MS | 100 | 0.22 (0.01) | 0.14-0.33 | 92 | 0.08 (0.01) | 0.05-0.16 | |
| CS | 100 | 0.20 (0.01) | 0.13-0.31 | 92 | 0.07 (0.01) | 0.05-0.11 | |||
| WCB | 93 | 0.10 (0.01) | 0.06-0.18 | 63 | 0.04 (0.005) | <LOD-0.08 | |||
| C12 | NEtFOSAA | MS | 100 | 0.79 (0.05) | 0.49-1.23 | 18 | - | - | |
| CS | 100 | 0.76 (0.05) | 0.46-1.16 | 25 | 0.02 (0.002) | <LOD-0.03 | |||
| WCB | 100 | 0.37 (0.03) | 0.24-0.59 | 12 | - | - | |||
ΣPFOS was predominant in all matrices with GMs of 23.8, 9.5, and 4.9 ng/mL, respectively for maternal serum, cord serum, and whole cord blood in cohort 3, and GMs of 8.8, 3.1, and 1.6 ng/mL, respectively for maternal serum, cord serum, and whole cord blood in cohort 5. PFOA was the second mostly predominant compound with GMs of 2.3, 2.0, and 1.1 ng/mL, respectively for maternal serum, cord serum, and whole cord blood in cohort 3, and GMs of 1.0, 0.8, and 0.4 ng/mL, respectively for maternal serum, cord serum, and whole cord blood in cohort 5. Other PFASs exhibited lower concentrations with GMs <1 ng/mL in all matrices. Of interest, we observed a decrease in PFAS concentrations in samples from cohort 5 in comparison to samples from cohort 3, except for PFNA, PFDA, PFDoDA, PFHxS, and PFDS.
Intra- and Inter-matrices PFAS correlations.
Intra-matrices Spearman correlations ranged from −0.38 between cord serum NEtFOSAA and PFDoDA and 0.88 for the correlation between whole cord blood PFOA and PFOS. Overall, the patterns of correlations were comparable between the blood matrices (See Supplemental Material: Figure S1).
Figure 1 shows the correlations between maternal, cord serum, and whole cord blood PFAS concentrations for the mainly studied PFASs. Detailed correlations can be found in Supplemental Material (Figure S2) for additional PFASs. Overall, correlations between maternal and cord serum PFAS concentrations were high, and ranged between 0.50 (PFHpA) and 0.90 (NEtFOSAA) in cohort 3, and between 0.41 (PFHpS) and 0.95 (PFOA) in cohort 5 (Figure 1a and Figure S2a). Regarding correlations between cord serum and whole cord blood, Spearman correlation coefficients ranged between 0.53 (PFHxS) and 0.96 (PFOS) in cohort 3, and between −0.10 (PFUnDA) and 0.95 (PFOA and PFOS) in cohort 5 (Figure 1b and Figure S2b).
Figure 1.
Correlation plots for cord vs maternal serum and serum vs whole cord blood pairs for the most frequently detected PFAS compounds (n=151).
Transplacental transfer and partitioning ratios.
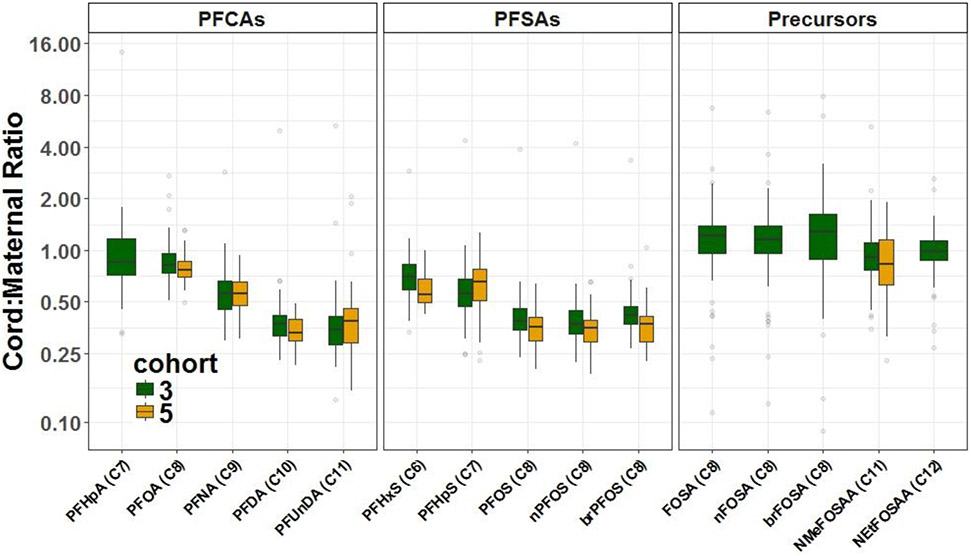
Figure 2 presents boxplots describing the distribution of transplacental transfer ratios in both cohorts. Overall, all PFASs had cord:maternal serum ratios below 1, except for total FOSA (both linear and branched isomers). Generally, these results suggest a significant transfer of PFASs from maternal blood to the fetus with transplacental transfer efficiencies ranging from a median efficiency of 36% (PFUnDA and PFDA) to 128% (branched FOSA isomer). Also, we observed a pattern of decreasing cord:maternal serum ratios with increasing carbon chain length for both PFCAs and PFSAs. Transplacental transfer also appeared to be higher in precursors FOSA, NMeFOSAA, and NEtFOSAA compared to both PFCAs and PFSAs. For instance, median cord:maternal serum ratios for total FOSA was 1.21 (IQR: 0.95-1.40), whereas the median cord:maternal serum ratios for PFOS and PFOA, both with the same carbon chain length, were 0.38 (IQR: 0.33-0.43) and 0.80 (IQR: 0.72-0.91), respectively. Detailed results can be found in Supplemental Material, Table S2.
Figure 2.
Distribution of the transplacental transfer ratios for PFAS compounds in the two cohorts, according to PFAS functional group and carbon chain length.
In regard to the blood partitioning of PFASs between serum and whole blood matrices, Figure 3 shows serum:whole blood in cord samples for both cohorts. Overall, all PFASs exhibited median ratios higher than 1, except for total FOSA (both linear and branched isomers), and PFHpA. These indicate a preference for the serum fraction for most PFASs. The highest serum:whole blood ratios were observed for PFUnDA and PFHpS with median ratios of 2.75 (IQR: 2.07-3.78) and 2.64 (IQR: 2.31-3.42), respectively. The lowest serum:whole blood ratios were observed for the branched isomer of FOSA with a median ratio of 0.24 (IQR: 0.16-0.50). Besides total FOSA and its isomers, PFDoDA and PFHpA also exhibited low ratios compared to other PFASs. Detailed results can be found in Supplemental Material, Table S3.
Figure 3.
Distribution of the blood partitioning ratios for PFAS compounds in the two cohorts, according to PFAS functional group and carbon chain length.
Predictors of transplacental transfer and partitioning ratios.
Associations between maternal and newborn characteristics and transplacental transfer and partitioning ratios.
In multivariable analyses investigation of potential determinants of transplacental and partitioning ratios, gestational diabetes appeared to be the strongest predictor of transplacental transfer ratios, with ratio increases ranging from 13% for PFOA (95% CI: −3, 29%) to 44% PFUnDA (95% CI: 15, 73%), depending on the compound, for mothers with gestational diabetes compared to mothers with no gestational diabetes (Figure 4 and Supplemental Material; Table S4). For instance, mothers with gestational diabetes had 33% (95% CI: 15, 51%) higher transplacental transfer ratios of PFOS compared with mothers with no gestational diabetes. Overall, there was no significant cohort effect on transplacental transfer ratios, except for PFHxS (β=−0.06, 95% CI: −0.11,0).
Figure 4.
Adjusted % difference and 95% confidence intervals in the PFAS transplacental transfer ratios between diagnosed gestational diabetes cases versus non-cases.
Regarding partitioning ratios, the ratios of PFNA, PFHxS, and FOSA were significantly different between the two cohorts, but there was no clear pattern in terms of direction. We also found significantly decreased (β=−0.51, 95% CI: −0.81, −0.21) serum:whole cord blood NEtFOSAA ratios in girls compared to boys (Supplemental Material; Table S5).
Associations between carbon chain length and PFAS’ functional group, and transplacental transfer and partitioning ratios.
In multivariable analyses investigating the associations between physico-chemical properties and transplacental and partitioning ratios, PFASs’ physico-chemical properties were significant predictors of transplacental transfer and partitioning ratios. For instance, the transplacental transfer ratios of PFSAs were 33% (95% CI: −41, −25%) lower in comparison to transplacental transfer of PFCAs. Concurrently, the transplacental transfer ratios of sulfonamide precursors were 55% (95% CI: 48, 62%) higher than transplacental transfer ratios of PFCAs. Finally, there was an 11% (95% CI: −13%, −9%) decrease in serum:maternal ratios per one carbon increase in chain length after adjustment for functional group and maternal and newborn characteristics.
Regarding PFASs partitioning, PFSAs’ partitioning ratios were 45% (95% CI: 35, 55%) higher than PFCAs’ partitioning ratios, whereas sulfonamide precursors partitioning ratios were 107% (95% CI: −117, −98%) lower than PFCAs partitioning ratios. Finally, there was a 29% (95% CI: 27, 32%) increase in serum: whole blood ratios per one carbon increase in chain length after adjustment for functional group and maternal and newborn characteristics.
In GAM analyses, we observed a non-linear dose-response relationship between carbon chain length and transplacental ratios with a steeper decrease from C6 to C10 PFASs (Figure 5a). When stratifying analyses by PFAS functional group (Figure 6a), we observed a U-shaped relationship for PFCAs with a decrease of ratios from C7 to C10, and an increase from C10 to C12 PFCAs.
Figure 5.
Dose-response relationship between a) transplacental transfer and b) blood partitioning ratios, and carbon chain length.
Figure 6.
Dose-response relationship between a) transplacental transfer and b) blood partitioning ratios, and carbon chain length, stratified by PFAS functional group. Shaded area represents the 95% confidence interval.
The same U-shaped relationship was observed for PFSAs with a decrease of ratios from C6 to C8, and an increase from C8 to C10 PFSAs. The relationship between carbon chain length and transplacental transfer ratios was linear for sulfonamide precursors for which transplacental transfer ratios decreased with increasing carbon chain length.
Regarding predictors of partitioning ratios, we observed a non-linear relationship with carbon chain length, with partitioning serum:whole blood ratios stable between C6 and C8, and increasing from C9 to C12 PFASs (Figure 5b). When stratifying by PFAS functional group, we observed a linear relationship between carbon chain length and partitioning serum:whole blood ratios with an increase of ratios with increasing carbon chain length for the three PFAS families (Figure 6b).
DISCUSSION
In the present study, we investigated the transplacental transfer ratios for PFASs characterized by measured cord:maternal serum ratios and blood partitioning patterns as measured by serum:whole cord blood ratios. Additionally, we assessed physico-chemical properties, newborn’, and maternal as potential predictors of these ratios. This is the largest study to date to model both transplacental transfer ratios and partitioning patterns of multiple PFAS compounds with different physico-chemical properties and maternal and newborn’s characteristics.
Overall, PFOS was the predominant compound in all maternal and cord blood matrices, followed by PFOA. Maternal serum concentrations of PFASs were higher than those in the cord serum concentrations, except for FOSA, whereas cord serum concentrations were higher than whole cord blood concentrations, except for FOSA again. Transplacental transfer ratios were higher in sulfonamide precursors, followed by PFCAs, and PFSAs. Partitioning ratios between serum and whole cord blood were higher in PFSAs, followed by PFCAs, and then sulfonamide precursors. Carbon chain length appeared to have a significant influence on both ratios, with transplacental transfer ratios decreasing with increasing chain length, whereas the opposite was observed for partitioning ratios.
Our study suggests that the overall concentrations of PFASs decreased over time since measured PFAS concentrations were lower in cohort 5 compared to cohort 3, except for PFNA, PFDA, PFDoDA, PFHxS, and PFDS. This decline may be attributable to large decreases in the legacy PFASs, PFOS and PFOA, which account for the dominant fractions of exposures (Dassuncao et al. 2018; Oulhote et al. 2016). Previous studies looking at the trends of PFAS exposures in the general population of U.S. show the same patterns of decline in PFOS and PFOA as the trends observed in the Faroe Island cohorts(Haug et al. 2009; Calafat et al. 2007; Olsen et al 2008; Kato et al 2011). The similar increase in PFNA and PFDA levels was also recorded for the U.S. general population between the years 1999-2008 (Glynn et al 2012, Calafat et al 2007, Gribble et al 2015).
Similar declines in PFASs exposure were reported in the U.S. (Harris et al. 2017), Sweden (Glynn et al. 2015), Australia (Eriksson et al. 2017), and Norway (Line S. Haug et al. 2009). The elimination of PFASs after parturition is another factor that may account for a part of the decreasing trend since the maternal blood was collected during late pregnancy in cohort 3 and two weeks after parturition in cohort 5 (Needham et al. 2011; Zhang et al. 2013).
Previous reports demonstrated that PFASs in human blood could cross the placental barrier and reach the fetus (Javins et al. 2013; Midasch et al. 2007; Mondal et al. 2012). However, these were limited by relatively small sample sizes and investigated fewer PFASs. The largest studies to date consisted of 66 and 100 mother-child pairs and investigated transplacental transfer of a limited number PFAS compounds in Spain (Manzano-Salgado et al. 2015) and China (Pan et al. 2017). Generally, our results are in line with findings from previously reported investigations showing decreasing transplacental transfer with increasing carbon chain from PFHpA (C7) to NEtFOSAA and PFDoDA (C12). Importantly, we found a U-shaped relationship for both PFCAs and PFSAs with a minimum value for transplacental transfer at C10 (PFDA) for PFCAs and C8 (PFOS) for PFSAs. A similar U-shaped relationship has already been reported ( Pan et al. 2017, Zhao et al. 2017). High transplacental values for long chain PFASs were also previously reported from studies in China, South Korea, and Norway (Gutzkow et al. 2012; Kim et al. 2011; Liu et al. 2011; Pan et al. 2017; Yang et al. 2013). To our knowledge, this is the first study to report on transplacental transfer of compounds such as PFHpS and NEtFOSAA.
The wide range of detected PFAS compounds in this study allows a better understanding of the maternal transfer pattern of PFASs with different carbon chain lengths and chemical structure. Available evidence suggests that the toxicity of PFASs increases with increasing carbon chain length (Kudo et al. 2006; Stahl et al. 2011; Upham et al. 1998). Our results suggesting a high transplacental transfer efficiency of certain long chain PFAS compounds raises serious concerns about their potential developmental toxicity. Although these PFASs are able to cross the placenta, the resulting levels of these compounds may not be indicative of a health risk.
We also report on serum:whole cord blood ratios which can be interpreted as a measure of blood partitioning of PFAS compounds. Our results indicate that the majority of the PFASs were present in the cord serum at higher concentrations than the cord whole blood samples. A major finding from our study is the differential partitioning of PFASs according to physico-chemical properties of the compound. Few previous studies investigated the distribution of PFASs in blood components, with equivocal findings. Ehresman et al. found that whole blood concentrations of PFOA, PFOS and PFHxS were almost half that of serum concentrations in a small sample of 18 3M company workers (Ehresman et al. 2007), whereas Karrman et al. reported plasma/whole blood concentration ratios ranging from 0.2 for FOSA to 1.4 for PFOA in a small sample (n=3-5). Finally, Jin et al. found partitioning ratios between 1.2 for PFOA and 1.6 for PFHxS in a small of 60 Chinese mother-child pairs (Jin et al. 2016). Biomonitoring studies usually measure PFAS compounds in serum or plasma because it is thought that these substances are largely predominant in serum/plasma, and when PFASs are measured in whole blood, whole blood data are usually converted to a serum/plasma basis by multiplying by a factor of 2. Our results suggesting higher affinity to the serum fraction with increasing carbon chain length indicate that conversion factors should be adapted to the PFAS compound. Moreover, FOSA showed a different pattern, with a higher affinity to the blood cells. Overall, most of PFASs were highly protein bound in blood, and this binding strength increases linearly with increasing carbon chain length in the three investigated PFAS functional groups. This is likely due to the fact that short carbon chain PFASs are more mobile with higher solubility in water and a lower potential for sorption to particles compared to long-chain analogues (EPA 2015).
We investigated main maternal and newborns’ characteristics as predictors of transplacental transfer and partitioning ratios. Overall, there were no consistent patterns of association between newborn’ and maternal characteristics and transplacental transfer ratios, except for gestational diabetes. The higher transplacental transfer of PFASs observed for mothers with gestational diabetes diagnosis may be due to functional and structural changes in the placenta under conditions of poor glycemic control (Desoye et al. 2011). No previous study investigated potential effect of gestational diabetes on transplacental transfer of PFAS compounds; however, such increased transplacental transfer was previously observed for the lidocaine metabolite monoethylglycinexylidide (Moises et al. 2015). Diabetes mellitus can alter the kinetic disposition and the metabolism of xenobiotics, and studies showed that diabetes mellitus may interfere with the placental capillarization index, causing delays in the mature placenta, and influencing the rate of transplacental transfer of xenobiotics during pregnancy (Calderon et al. 2007; Moises et al. 2015). No consistent pattern of associations was observed for partitioning ratios, although some compounds exhibited significantly different ratios between cohort 3 and 5. Some of these differences could be attributed to the timing of blood sampling in the mothers in the two cohorts.
A potential limitation of our study is the difference of the timing of maternal blood sampling between the two cohorts. Since parturition is a major route for PFAS clearance (Zhang et al. 2013), this difference in blood sampling could influence comparability of results in the two cohorts. However, all our analyses investigating predictors of transplacental transfer and partitioning ratios adjusted for the cohort effect, and we did not see a clear pattern demonstrating any substantial effect of the cohort. Additionally, we were not able to estimate transplacental transfer and partitioning ratios for PFBA, PFBS, PFPeA, and PFHxA because of the limited detection frequency in our sample.
Despite these limitations, the current study represents the largest study to date investigating both transplacental transfer and blood partitioning of PFASs and covered a wider number of PFAS compounds compared to previous investigations. The concordance of the findings between the two cohorts supports the validity of the estimated ratios. Our results provide a comprehensive characterization of the transplacental transfer and blood partitioning of PFAS compounds and deepen the knowledge on the effect of PFASs physico-chemical structure on these parameters. This provides a better understanding of these compounds and sheds light on the importance of considering the chemical structure of the compounds for future risk assessment purposes, and for the choice of appropriate blood matrices for measurement of PFAS compounds.
Supplementary Material
Highlights.
Per- and polyfluoroalkyl substances (PFASs) are a growing public health concern and can cross the placenta
Data on transplacental transfer efficiency and partitioning of short and long chain PFASs in blood matrices are limited.
We investigated predictors of transplacental transfer and blood partitioning of 17 PFAS compounds in151 matched mother-newborn pairs from two Faroe Islands cohorts.
We observed significantly higher transfer in mothers with gestational diabetes.
Functional groups and carbon chain length were important predictors of transplacental transfer and blood partitioning.
Acknowledgments:
We acknowledge financial support for this study from the NSF-NIH Oceans and Human Health Program (OCE-1321612).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Competing financial interests:
Dr. Philippe Grandjean served as a health expert for the State of Minnesota in a recent lawsuit against a PFAS-producing company.
REFERENCES
- ATSDR. 2018. Toxicological profile for perfluoroalkyls. Atlanta, GA. [PubMed] [Google Scholar]
- Betts KS. 2007. Perfluoroalkyl acids what is the evidence telling us.Pdf. Environmental Health Perspectives 115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Birnbaum LS, Grandjean P. 2015. Alternatives to pfass: Perspectives on the science. Environ Health Perspect 123:A104–105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buck RC, Franklin J, Berger U, Conder JM, Cousins IT, de Voogt P, et al. 2011. Perfluoroalkyl and polyfluoroalkyl substances in the environment: Terminology, classification, and origins. Integr Environ Assess Manag 7:513–541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calafat AM, Wong LY, Kuklenyik Z, Reidy JA, Needham LL. 2007. Polyfluoroalkyl chemicals in the US population: Data from the National Health and Nutrition Examination Survey (NHANES) 2003–2004 and comparisons with NHANES 1999–2000. Environ. Health Perspect 115: 1596–1602 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calderon IM, Damasceno DC, Amorin RL, Costa RA, Brasil MA, Rudge MV. 2007. Morphometric study of placental villi and vessels in women with mild hyperglycemia or gestational or overt diabetes. Diabetes Res Clin Pract 78:65–71. [DOI] [PubMed] [Google Scholar]
- CECBP. 2015. Potential designated chemicals- perfluoroalkyl and polyfluoroalkyl substances (pfass)California Environmental Contaminant Biomonitoring Program.Christensen KY, [Google Scholar]; Raymond M, Blackowicz M, Liu Y, Thompson BA, Anderson HA, et al. 2017. Perfluoroalkyl substances and fish consumption. Environ Res 154:145–151. [DOI] [PubMed] [Google Scholar]
- Dassuncao C, Hu XC, Nielsen F, Weihe P, Grandjean P, Sunderland EM. 2018. Shifting global exposures to poly- and perfluoroalkyl substances (PFASs) evident in longitudinal birth cohorts from a seafood-consuming population. Environ Sci Technol 52:3738–3747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Desoye G, Gauster M, Wadsack C. 2011. Placental transport in pregnancy pathologies. Am J Clin Nutr 94:1896S–1902S. [DOI] [PubMed] [Google Scholar]
- Ehresman DJ, Froehlich JW, Olsen GW, Chang SC, Butenhoff JL. 2007. Comparison of human whole blood, plasma, and serum matrices for the determination of perfluorooctanesulfonate (pfos), perfluorooctanoate (pfoa), and other fluorochemicals. Environ Res 103:176–184. [DOI] [PubMed] [Google Scholar]
- EPA D. 2015. Short-chain polyfluoroalkyl substances (pfas): A literature review of information on human health effects and environmental fate and effect aspects of short-chain pfas. (Environmental Project).Danish Ministry of the Environement (EPA). [Google Scholar]
- Eriksson U, Mueller JF, Toms LL, Hobson P, Karrman A. 2017. Temporal trends of pfsas, pfcas and selected precursors in australian serum from 2002 to 2013. Environ Pollut 220:168–177. [DOI] [PubMed] [Google Scholar]
- Fromme H, Tittlemier SA, Volkel W, Wilhelm M, Twardella D. 2009. Perfluorinated compounds-exposure assessment for the general population in western countries. Int J Hyg Environ Health 212:239–270. [DOI] [PubMed] [Google Scholar]
- Fromme H, Mosch C, Morovitz M, Alba-Alejandre I, Boehmer S, Kiranoglu M, et al. 2010. Pre- and postnatal exposure to perfluorinated compounds (pfcs). Environmental Science & Technology 44:7123–7129. [DOI] [PubMed] [Google Scholar]
- Glynn A, Berger U, Bignert A, EUlah S, Aune M, Lignell S, Darnerud PA. 2012. Perfluorinated Alkyl Acids in Blood Serum from Primiparous Women in Sweden: Serial Sampling during Pregnancy and Nursing, And Temporal Trends 1996–2010. Environmental Science & Technology, 46: 9071–9079. [DOI] [PubMed] [Google Scholar]
- Glynn A, Benskin J, Lignell S, Gyllenhammar I, Aune M, Cantillana T, et al. 2015. Temporal trends of perfluoroalkyl substances in pooled serum samples from first-time mothers in uppsala 1997-2014. Report to the Swedish EPA.
- Grandjean P, Weihe P, Needham LL, Burse V, Patterson GDJ, Sampson E, et al. 1995. Relation of a seafood diet to mercury, selenium, arsenic, and polychlorinated biphenyl and other organochlorine concentrations in human milk. Environmental Research 71:29–38. [DOI] [PubMed] [Google Scholar]
- Gribble MO, Bartell SM, Kannan K, Wu Q, Fair PW, Kamen DL. 2015. Longitudinal measures of perfluoroalkyl substances (PFAS) in serum of Gullah African Americans in South Carolina: 2003–2013. Environ Res. 143(0 0): 82–88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gutzkow KB, Haug LS, Thomsen C, Sabaredzovic A, Becher G, Brunborg G. 2012. Placental transfer of perfluorinated compounds is selective-a norwegian mother and child sub-cohort study. Int J Hyg Environ Health 215:216–219. [DOI] [PubMed] [Google Scholar]
- Hanssen L, Dudarev AA, Huber S, Odland JO, Nieboer E, Sandanger TM. 2013. Partition of perfluoroalkyl substances (pfass) in whole blood and plasma, assessed in maternal and umbilical cord samples from inhabitants of arctic russia and Uzbekistan. Sci Total Environ 447:430–437. [DOI] [PubMed] [Google Scholar]
- Harris MH, Rifas-Shiman SL, Calafat AM, Ye X, Mora AM, Webster TF, et al. 2017. Predictors of per- and polyfluoroalkyl substance (pfas) plasma concentrations in 6-10 year old american children. Environ Sci Technol 51:5193–5204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hastie TJ, Tibshirani RJ. 1990. Generalized additive models. Chapman and Hall: CRC press. [Google Scholar]
- Haug LS, Thomsen C, Becher G. 2009. A sensitive method for determination of a broad range of perfluorinated compounds in serum suitable for large-scale human biomonitoring. J Chromatogr A 1216:385–393. [DOI] [PubMed] [Google Scholar]
- Haug LS, Thomsen C, Becher GB. 2009. Time trends and the influence of age and gender on serum concentrations of perfluorinated compounds in archived human samples. Environ Sci Technol 43:2131–2136. [DOI] [PubMed] [Google Scholar]
- Hu XC, Andrews DQ, Lindstrom AB, Bruton TA, Schaider LA, Grandjean P, et al. 2016. Detection of poly- and perfluoroalkyl substances (pfass) in u.S. Drinking water linked to industrial sites, military fire training areas, and wastewater treatment plants. Environ Sci Technol Lett 3:344–350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Javins B, Hobbs G, Ducatman AM, Pilkerton C, Tacker D, Knox SS. 2013. Circulating maternal perfluoroalkyl substances during pregnancy in the c8 health study. Environ Sci Technol 47:1606–1613. [DOI] [PubMed] [Google Scholar]
- Jin H, Zhang Y, Jiang W, Zhu L, Martin JW. 2016. Isomer-specific distribution of perfluoroalkyl substances in blood. Environ Sci Technol 50:7808–7815. [DOI] [PubMed] [Google Scholar]
- Kato K, Wong LY, Jia LT, Kuklenyik Z, Calafat AM. 2011. Trends in Exposure to Polyfluoroalkyl Chemicals in the U.S. Population: 1999–2008. Environmental Science & Technology, 45 (19): 8037–8045. [DOI] [PubMed] [Google Scholar]
- Kim S, Choi K, Ji K, Seo J, Kho Y, Park J, et al. 2011. Trans-placental transfer of thirteen perfluorinated compounds and relations with fetal thyroid hormones. Environ Sci Technol 45:7465–7472. [DOI] [PubMed] [Google Scholar]
- Kudo N, Suzuki-Nakajima E, Mitsumoto A, Kawashima Y. 2006. Responses of the liver to perfluorinated fatty acids with different carbon chain length in male and female mice-in relation to induction of hepatomegaly, peroxisomal beta-oxidation and microsomal 1- acylglycerophosphocholine acyltransferase. Biological & pharmaceutical bulletin 29:1952–1957. [DOI] [PubMed] [Google Scholar]
- Li Y, Fletcher T, Mucs D, Scott K, Lindh CH, Tallving P, et al. 2018. Half-lives of pfos, pfhxs and pfoa after end of exposure to contaminated drinking water. Occup Environ Med 75:46–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liew Z, Goudarzi H, Oulhote Y. 2018. Developmental exposures to perfluoroalkyl substances (pfass): An update of associated health outcomes. Current environmental health reports 5:1–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu J, Li J, Liu Y, Chan HM, Zhao Y, Cai Z, et al. 2011. Comparison on gestation and lactation exposure of perfluorinated compounds for newborns. Environ Int 37:1206–1212. [DOI] [PubMed] [Google Scholar]
- Manzano-Salgado CB, Casas M, Lopez-Espinosa MJ, Ballester F, Basterrechea M, Grimalt JO, et al. 2015. Transfer of perfluoroalkyl substances from mother to fetus in a spanish birth cohort. Environ Res 142:471–478. [DOI] [PubMed] [Google Scholar]
- Midasch O, Drexler H, Hart N, Beckmann MW, Angerer J. 2007. Transplacental exposure of neonates to perfluorooctanesulfonate and perfluorooctanoate: A pilot study. Int Arch Occup Environ Health 80:643–648. [DOI] [PubMed] [Google Scholar]
- Moises EC, Duarte Lde B, Cavalli Rde C, Carvalho DM, Filgueira GC, Marques MP, et al. 2015. Transplacental distribution of lidocaine and its metabolite in peridural anesthesia administered to patients with gestational diabetes mellitus. Reprod Sci 22:791–797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mondal D, Lopez-Espinosa MJ, Armstrong B, Stein CR, Fletcher T. 2012. Relationships of perfluorooctanoate and perfluorooctane sulfonate serum concentrations between mother-child pairs in a population with perfluorooctanoate exposure from drinking water. Environ Health Perspect 120:752–757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Needham LL, Grandjean P, Heinzow B, Jorgensen PJ, Nielsen F, Patterson GDJ, et al. 2011. Partition of environmental chemicals between maternal and fetal blood and tissues. Environmental Science & Technology 45:1121–1126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olsen GW, Mair DC, Reagen WK, Ellefson ME, Ehresman DJ, Butenhoff JL, et al. 2007. Preliminary evidence of a decline in perfluorooctanesulfonate (pfos) and perfluorooctanoate (pfoa) concentrations in american red cross blood donors. Chemosphere 68:105–111. [DOI] [PubMed] [Google Scholar]
- Olsen GW, Mair DC, Church TR, Ellefson ME, Reagen WK, Boyd TM et al. 2008. Decline in perfluorooctanesulfonate and other polyfluoroalkyl chemicals in American red cross adult blood donors, 2000-2006. Environmental Science & Technology. 42:4989–4995. [DOI] [PubMed] [Google Scholar]
- Olsen GW, Butenhoff JL, Zobel LR. 2009. Perfluoroalkyl chemicals and human fetal development: An epidemiologic review with clinical and toxicological perspectives. Reprod Toxicol 27:212–230. [DOI] [PubMed] [Google Scholar]
- Oulhote Y, Steuerwald U, Debes F, Weihe P, Grandjean P. 2016. Behavioral difficulties in 7-year old children in relation to developmental exposure to perfluorinated alkyl substances. Environ Int 97:237–245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pan Y, Zhu Y, Zheng T, Cui Q, Buka S, Zhang B, et al. 2017. Novel Chlorinated Polyfluorinated Ether Sulfonates and Legacy Per-/Polyfluoroalkyl Substances: Placental Transfer and Relationship with Serum Albumin and Glomerular Filtration Rate. Environ. Sci. Technol 51:634–644. [DOI] [PubMed] [Google Scholar]
- Papadopoulou E, Haug LS, Sabaredzovic A, Eggesbo M, Longnecker MP. 2015. Reliability of perfluoroalkyl substances in plasma of 100 women in two consecutive pregnancies. Environ Res 140:421–429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rappazzo KM, Coffman E, Hines EP. 2017. Exposure to perfluorinated alkyl substances and health outcomes in children: A systematic review of the epidemiologic literature. Int J Environ Res Public Health 14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schaider LA, Balan SA, Blum A, Andrews DQ, Strynar MJ, Dickinson ME, et al. 2017. Fluorinated compounds in u.S. Fast food packaging. Environmental Science & Technology Letters 4:105–111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlezinger JJ, Bernard PL, Haas A, Grandjean P, Weihe P, Sherr DH. 2010. Direct assessment of cumulative aryl hydrocarbon receptor agonist activity in sera from experimentally exposed mice and environmentally exposed humans. Environ Health Perspect 118:693–698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stahl T, Mattern D, Brunn H. 2011. Toxicology of perfluorinated compounds. Environmental Sciences Europe 23. [Google Scholar]
- StataCorp. 2013. Stata statistical software. Part 13. College Station, TX:StataCorp LP. [Google Scholar]
- Team RC. 2013. R: A language and environment for statistical computing. Vienna, Austria:R Foundation for Statistical Computing. [Google Scholar]
- Upham BL, Deocampo ND, Wurl B, Trosko JE. 1998. Inhibition of gap junctional intercellular communication by perfluorinated fatty acids is dependent on the chain length of the fluorinated tail. International Journal of Cancer 78:491–495. [DOI] [PubMed] [Google Scholar]
- Vestergren R, Berger U, Glynn A, Cousins IT. 2012. Dietary exposure to perfluoroalkyl acids for the swedish population in 1999, 2005 and 2010. Environ Int 49:120–127. [DOI] [PubMed] [Google Scholar]
- Wang Z, Cousins IT, Scheringer M, Hungerbuhler K. 2013. Fluorinated alternatives to long-chain perfluoroalkyl carboxylic acids (pfcas), perfluoroalkane sulfonic acids (pfsas) and their potential precursors. Environment International 60:242–248. [DOI] [PubMed] [Google Scholar]
- Weihe P, Kato K, Calafat AM, Nielsen F, Wanigatunga AA, Needham LL, et al. 2008. Serum concentrations of polyfluoroalkyl compounds in faroese whale meat consumers. Environmental Science & Technology 42:6291–6295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weihe P, Debes Joensen H. 2012. Dietary recommendations regarding pilot whale meat and blubber in the faroe islands. International Journal of Circumpolar Health 71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang L, Li J, Lai J, Luan H, Cai Z, Wang Y, et al. 2016. Placental Transfer of Perfluoroalkyl Substances and Associations with Thyroid Hormones: Beijing Prenatal Exposure Study. Sci Rep 6: 21699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang T, Wu Q, Sun HW, Zhang XZ, Yun SH, Kannan K. 2010. Perfluorinated compounds in whole blood samples from infants, children, and adults in china. Environmental Science & Technology 44:4341 – 4347. [DOI] [PubMed] [Google Scholar]
- Zhang Y, Beesoon S, Zhu L, Martin JW. 2013. Biomonitoring of perfluoroalkyl acids in human urine and estimates of biological half-life. Environ Sci Technol 47:10619–10627. [DOI] [PubMed] [Google Scholar]
- Zhao L, Zhang Y, Zhu L, Ma X, Wang Y, Sun H, et al. 2017. Isomer-specific transplacental efficiencies of perfluoroalkyl substances in human whole blood. Environmental Science & Technology Letters 4:391–398. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.