Distinctions in the nature and spatiotemporal expression of IL-2R subunits on conventional versus regulatory T cells are exploited to manipulate IL-2 immunomodulatory effects. Particularly, low-dose IL-2 and some recombinant derivatives are being evaluated to enhance/inhibit immune responses for therapeutic purposes.
Abstract
Historically, interleukin-2 (IL-2) was first described as an immunostimulatory factor that supports the expansion of activated effector T cells. A layer of sophistication arose when regulatory CD4+ T lymphocytes (Tregs) were shown to require IL-2 for their development, homeostasis, and immunosuppressive functions. Fundamental distinctions in the nature and spatiotemporal expression patterns of IL-2 receptor subunits on naive/memory/effector T cells versus Tregs are now being exploited to manipulate the immunomodulatory effects of IL-2 for therapeutic purposes. Although high-dose IL-2 administration has yielded discrete clinical responses, low-dose IL-2 as well as innovative strategies based on IL-2 derivatives, including “muteins,” immunocomplexes, and immunocytokines, are being explored to therapeutically enhance or inhibit the immune response.
Background
Clues supporting the existence of IL-2 emerged in 1976. That year, after successfully expanding T cells in vitro, Morgan et al. (1976) reported the presence of a T cell growth factor in the conditioned media of phytohemagglutinin-stimulated blood lymphocytes. In the early 1980s, human IL-2 was firmly identified as a variably glycosylated ∼15.5-kD protein (Robb and Smith, 1981), thereafter purified (Smith et al., 1983), and finally cloned (Taniguchi et al., 1983). At the same period, the IL-2 receptor (IL-2R) was discovered (Kuribayashi et al., 1981; Robb et al., 1981), thus solving the first type I cytokine/receptor complex.
By permitting a prolonged culture of T cells, the discovery of IL-2, initially called “T cell growth factor,” facilitated molecular and cellular investigations that precipitated, for example, the characterization of the TCR and its function (Allison et al., 1982; Haskins et al., 1983), or the identification of the first human retrovirus: human T cell leukemia virus (HTLV-1; Poiesz et al., 1980). Initial studies performed in vitro concluded to a critical role of IL-2 in the development of effector T lymphocytes. Moreover, experimental investigations conducted in a chicken model of autoimmune thyroiditis revealed a pro-autoimmune effect of IL-2 and IL-2R–expressing T lymphocytes (Krömer et al., 1985), an observation that was mechanistically explained by the capacity of IL-2 to reverse anergy of self-reactive T cells in mice (Gonzalo et al., 1993) and simultaneously validated by clinical studies in humans showing that cancer patients treated with high-dose (HD) IL-2 frequently developed autoimmune thyroiditis (Krouse et al., 1995).
However, in vivo studies conducted in the 1990s in mouse strains lacking IL-2 or IL-2R subunits led to a revision of the concept that the IL-2/IL-2R system would be solely involved in immunostimulatory circuities. Indeed, rather than harboring an immunodeficiency, these animals demonstrated lymphadenopathy, uncontrolled proliferation of peripheral activated T cells, and signs of autoimmunity (Sadlack et al., 1993; Suzuki et al., 1995; Willerford et al., 1995). Such observations unveiled the existence of immunosuppressive mechanisms critically relying on IL-2 and later attributed to regulatory CD4+ T cells (Tregs; Sakaguchi et al., 1995; Malek et al., 2000, 2002).
The immunomodulatory effects of IL-2, mainly on effector and regulatory T lymphocytes, have been exploited for treating various pathologies, though with limited clinical benefits so far. In this line, a recombinant human IL-2 called aldesleukin (brand name: Proleukin) was approved for the treatment of kidney cancer and melanoma as early as 1992 and 1998, respectively (Alva et al., 2016).
After introducing some fundamental aspects of IL-2 biology, the present review will summarize current strategies to introduce IL-2 into the immunotherapeutic armamentarium.
Biology of IL-2
TCR signaling and IL-2 production
IL-2 is mainly produced by CD4+ T lymphocytes (naive, memory, and T helper [Th] 1) following antigenic stimulation, by type 2 and 3 innate lymphoid cells in the small intestine, and to a lesser extent by activated CD8+ T cells, B cells, and by other innate immune entities such as natural killer (NK) and NKT lymphocytes, dendritic cells (DCs), monocytes, or mast cells (Malek, 2008; Wojciechowski et al., 2009; Hershko et al., 2011; Zelante et al., 2012; Zhou et al., 2019). In naive T lymphocytes, the engagement of the TCR and co-stimulatory molecules (e.g., CD28) within an immunological synapse activates activator protein 1 (AP-1), NFκB, and NFAT (Fig. 1). In cooperation with constitutive factors, these transcription factors promote the expression of the IL2 gene (Serfling et al., 1995). IL2 transcription occurs within 30 min after stimulation but is transient, declining to background levels within 24–48 h. Additionally, post-transcriptional regulatory mechanisms further restrict the availability of IL-2 mRNAs, the levels of which usually peak at 4–8 h after stimulation (Jain et al., 1995). The turnover of IL-2 mRNAs is mostly controlled by proteins interacting with an AU-rich cis element (ARE) in their 3′-untranslated region. Among these trans-acting factors figure nuclear factor 90 (NF90) and tristetraprolin. NF90 is activated by protein kinase (PK) B (best known as AKT) upon CD28 co-stimulation, or by PKC upon restimulation with PMA, and then exported from the nucleus to the cytosol. There, NF90 binds to ARE and stabilizes IL-2 mRNAs, thus allowing their translation (Pei et al., 2008; Zhu et al., 2010). In contrast, tristetraprolin is expressed in T lymphocytes following activation and plays a critical role in the rapid decay of IL-2 mRNAs, as its interaction with ARE promotes its degradation (Ogilvie et al., 2005; Yang et al., 2015). Altogether, these transcriptional and post-transcriptional mechanisms control the magnitude and duration of IL-2 production by activated T cells.
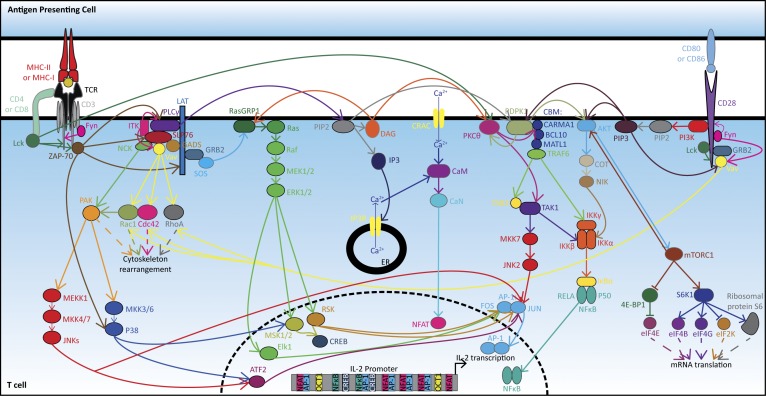
Figure 1.
IL-2 transcription following stimulation of the TCR and CD28 signaling cascades. Interactions between the CD4 (or CD8)/TCR complex at the surface of a T lymphocyte and an MHC-II (or MHC-I) molecule coupled to a cognate antigen at the surface of an APC triggers several intracellular signaling pathways leading to IL-2 production. Precisely, upon CD4/CD8 binding to MHC, lymphocyte-specific protein tyrosine kinase (Lck) is carried to the TCR proximity and is autophosphorylated and activated. Lck can phosphorylate CD3, which recruits the protein zeta-chain–associated protein kinase 70 (ZAP-70). ZAP-70 undergoes trans-autophosphorylation, leading to its activation, and phosphorylates the linker for the activation of T cells (LAT). Then, LAT plays as an adaptor protein supplying docking sites for: (i) growth factor receptor-bound protein 2 (GRB2); (ii) GRB2-related adaptor protein 2 (also known as GADS); and (iii) phospholipase C gamma1 (PLCγ). GRB2 binds to the son of sevenless protein (SOS), which activates Ras guanyl-releasing protein 1 (RasGRP1). RasGRP1 promotes the release of guanosine diphosphate (GDP) and the binding of guanosine triphosphate (GTP) on membrane-associated protein Ras, a small GTPase. GTP-bound Ras recruits the rapidly accelerated fibrosarcoma protein (Raf) and promotes its dimerization and activation by autophosphorylation. Activated Raf recruits and phosphorylates the mitogen-activated protein kinase kinases 1 and 2 (best known as MEK1/2), which in turn phosphorylate ERK1/2. Phosphorylated ERK1/2 translocates to the nucleus and activates ETS Like-1 protein (Elk1), mitogen and stress activated protein kinases 1 and 2 (MSK1/2), and p90 ribosomal S6 kinases (RSKs). The transcription factor Elk1, together with RSKs, promotes the expression of the gene FOS, while MSK1/2 activates c-AMP response element-binding protein (CREB). RSKs also phosphorylate JUN, enhancing the transcriptional activity of AP-1, which is constituted of a heterodimer of JUN and FOS. Both AP-1 and CREB translocate to the nucleus, bind to promoter of the IL2 gene, and stimulate its transcription. SH2 domain containing leukocyte protein of 76 kD (SLP-76) is recruited to GADS and phosphorylated by ZAP-70. Phosphorylated SLP-76 recruits PLCγ, the noncatalytic region of tyrosine kinase (NCK) adaptor protein, and the proteins Vav and interleukin-2-inducible T cell kinase (ITK). Fyn phosphorylates Vav, a guanine nucleotide exchange factor, which can activate Ras-related C3 botulinum toxin substrate 1 (Rac1), cell division control protein 42 homologue (Cdc42), and Ras homologue gene family, member A (RhoA), which are involved in cytoskeleton rearrangement. NCK recruits the p21-activated serine/threonine kinase (PAK) at the membrane, and the Cdc42/Rac1 complex activates PAK by disrupting PAK homodimerization and allowing its autophosphorylation. This leads to the activation of mitogen-activated protein kinase kinase kinase 1 (MEKK1) and dual specificity mitogen-activated protein kinase kinases (MKKs) 3 and 6 (MKK3/6). MEKK1 can further phosphorylate MKK4/7, and activated MKK4/7 activates in turn JNKs. Then, JNKs phosphorylate JUN leading to an enhanced transcriptional activity of the AP-1 complex. JNKs also activate the activating transcription factor 2 (ATF2) that phosphorylates and promotes the transcription of JUN. MKK3/6 phosphorylates P38 mitogen-activated kinases, which further activate ATF2 and MSK1/2. The recruitment of PLCγ by SLP-76 leads to its phosphorylation by ITK and ZAP-70. PLCγ detaches from SLP-76, re-translocates to the plasma membrane, binds to phosphatidylinositol 4,5-bisphosphate (PIP2), and hydrolyses PIP2 to produce diacylglycerol (DAG) and inositol 1,4,5-trisphosphate (IP3). IP3 binds to its receptor, IP3 receptor (IP3R), leading to the release of Ca2+ from ER, which activates calmodulin (CaM). This cytosolic influx of Ca2+ from ER stores ignites a prolonged opening of Ca2+ release–activated Ca2+ channels (CRAC) located at the plasma membrane, a requirement for sustaining T cell functions. Then, CaM binds to calcineurin (CaN), leading to its activation. CaN dephosphorylates NFAT, which can translocate into the nucleus. In the meantime, DAG can recruit to the plasma membrane and activate RasGRP1, leading to the downstream signaling pathway of Ras/Raf. Additionally, DAG also recruits the protein kinase C theta (PKCθ), leading to a conformational change and its phosphorylation by Lck. Simultaneously, the 3-phosphoinositide-dependent protein kinase 1 (PDPK1) is translocated to the membrane by binding to PIP2 or PIP3. Then, PDPK1 binds to caspase recruitment domain-containing protein 11 (CARMA1) and phosphorylates PKCθ; the latter autophosphorylates to achieve full activation. PKCθ phosphorylates CARMA1, which undergoes oligomerization and recruits BCL10. Afterwards, BCL10 oligomerizes and is phosphorylated by the receptor-interacting serine/threonine-protein kinase 2, leading to the recruitment of the mucosa-associated lymphoid tissue lymphoma translocation protein 1 (MATL1). In turn, MATL1 oligomerizes and binds to the TNF receptor-associated factor 6 (TRAF6). Then follows TRAF6 oligomerization, activation of its ubiquitin ligase activity, and auto-polyubiquitination. Ubiquitinated TRAF6 recruits the mitogen-activated protein kinase kinase kinase 7-interacting protein 2 (TAB2), which binds the mitogen-activated protein kinase kinase kinase 7 (best known as the transforming growth factor β-activated kinase 1 [TAK1]). The complex TAB2/TAK1 is ubiquitinated by TRAF6, and TAK1 undergoes autophosphorylation. Then, TAK1 phosphorylates MKK7, which activates JNK2. Then follows the JNK2-mediated phosphorylation of JUN, which associates with FOS to form the AP-1 complex. Activated TAK1 also phosphorylates NFκB inhibitor kinase subunit β (IKKβ), while TRAF6 ubiquitinates the γ subunit IKKγ (also known as NFκB essential modifier). Simultaneously, PDPK1 phosphorylates PKB (best known as AKT), allowing the latter to activate the mitogen-activated protein kinase kinase kinase 8 (also known as cancer osaka thyroid oncogene [COT]). COT activates the NFκB-inducing kinase (NIK, also known as MAP3K14), which phosphorylates IKKα. It results in an activated IKK complex composed of IKKα/β/γ. The NFκB inhibitor α (best known as IκBα), which otherwise sequesters the transcription factor NFκB in the cytoplasm, is phosphorylated by the IKK complex and then undergoes ubiquitination and degradation. The released NFκB, a heterodimer constituted of p50 (also known as NFκB subunit 1) and p65 (also known as v-Rel avian reticuloendotheliosis viral oncogene homologue A [RELA]), can therefore translocate to the nucleus. Ultimately, together with constitutive transcription factors like the octamer-binding protein 1 (OCT1, also known as POU domain class 2 transcription factor 1), the nuclear translocation of AP-1, NFκB, NFAT, and CREB, which all dispose of cis-regulatory elements within the promoter of the IL2 gene, will initiate its transcription. Moreover, AKT phosphorylates mTOR within the mTORC1 complex, composed of Raptor, proline-rich AKT substrate of 40 kD, DEP domain-containing mTOR-interacting protein, and mammalian lethal with SEC13 protein 8. mTORC1 can phosphorylate the eukaryotic translation initiation factor (eIF) 4E-binding protein 1 (4E-BP1), thus abrogating its inhibitory sequestration of eIF4E. mTORC1 also activates the ribosomal protein S6 kinase β-1 (S6K1). S6K1 phosphorylates and activates thereafter the ribosomal protein s6 as well as the translation initiation factors eIF4B and eIF4G, while inactivating eIF2K by phosphorylation. Altogether, these events up-regulate mRNA translation. Concurrent with the activation of the TCR signaling, a co-stimulatory signal, consecutive to the interaction of B7-1 (CD80) or B7-2 (CD86) on an APC with CD28 on the T cell, is triggered. The intracellular downstream signaling pathway starts with the phosphorylation of CD28 by Lck and Fyn, allowing the recruitment to CD28 of PI3Ks, GRB2, and GADS. PI3K can further phosphorylate PIP2 to PIP3, leading to the recruitment of PDPK1 and AKT to the membrane. The resulting stimulation of the AKT downstream signaling pathway activates NFκB and promotes the up-regulation of mRNA translation by mTORC1. In the meantime, Vav is recruited to the membrane through binding to GRB2 or PIP3 and phosphorylated by Fyn. Thereafter, Vav can stimulate Rac1, Cdc42, and RhoA-related cytoplasmic events. Sources: Reactome; KEGG pathway (Huse, 2009; Courtney et al., 2018).
IL-2R signaling
Once secreted, IL-2 is consumed in an autocrine/paracrine manner by neighboring cells that harbor its receptor, IL-2R (Fig. 2). The latter consists of a hetero-complex of up to three subunits: α, β, and γ, also known as CD25, CD122, and CD132, respectively. Although each receptor subunit may independently bind IL-2 with a weak affinity (Kd: ∼10−8–10−7 M), only the intermediate-affinity βγ dimeric (Kd: ∼10−9 M) and the strong-affinity αβγ trimeric IL-2R (Kd: ∼10−11 M) mediate intracellular signal transduction (Flynn and Hartley, 2017). The γ subunit is ubiquitously expressed on most hematopoietic cells. It is also referred to as the “common” chain (labeled “γc”), as it is shared with the receptors for IL-4, -7, -9, -15, and -21. The β subunit appears constitutively expressed at various levels on T and NK(T) lymphocytes. In contrast, the α chain/CD25 is detected on early thymocytes, absent on naive/memory T cells (particularly in mice, but detectable on a minor fraction of human naive/memory T cells), transiently exposed on activated/effector T lymphocytes, and preferentially/more stably expressed on Tregs (Kmieciak et al., 2009; Flynn and Hartley, 2017). Within the myeloid compartment, monocytes display the intermediate-affinity βγ receptor, whereas DC subtypes can present the three subunits of IL-2R (Bosco et al., 2000; Herr et al., 2014; Kitashima et al., 2018). Interestingly, DCs may also supply the α chain in trans, thus supporting high-affinity binding of IL-2 to naive T lymphocytes that undergo antigen priming (Wuest et al., 2011). Additionally, some nonhematopoietic cell types may harbor either (i) dimeric βγ receptors, as this applies to intestinal epithelial cells, dermal fibroblasts, or fibroblast-like synoviocytes; or (ii) the high-affinity αβγ IL-2R, as reported for endothelial cells, proximal tubular epithelial cells, or gingival fibroblasts. As an aside, IL-2R expression has also been documented for malignant cells such as melanoma and cervical tumor cells (Valle-Mendiola et al., 2016).
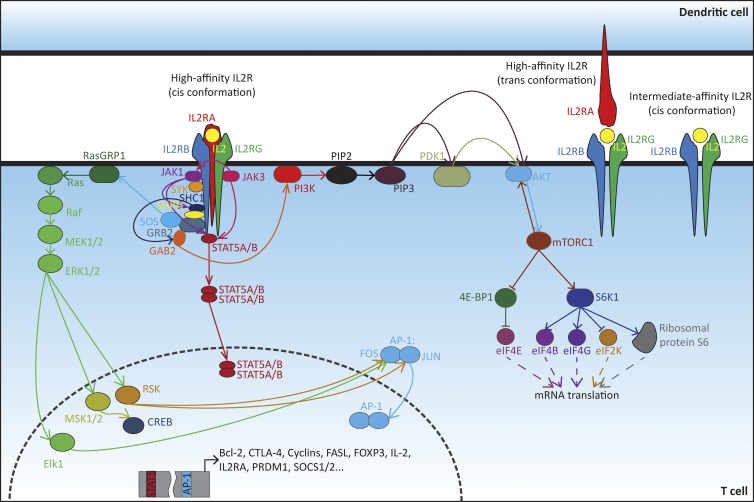
Figure 2.
IL-2R signaling and modulation of T cell activity. IL-2R is composed of up to three subunits: α (IL2RA), β (IL2RB), and γ (IL2RG). The intermediate-affinity IL-2R is composed of the IL2RB and IL2RG subunits. The high-affinity receptor consists either of the cis gathering of the three subunits, for instance, on a T cell, or of the cis assembly of IL2RB and IL2RG complemented in trans with IL2RA located at the surface of a DC. IL2RB and IL2RG are respectively bound to the JAK1 and JAK3 and responsible for transducing intracellular signaling. Upon ligation of IL-2, JAK3 phosphorylates JAK1, which in turn recruits to IL2RB the spleen-associated tyrosine kinase (SYK) and phosphorylates it. JAK1 also phosphorylates IL2RB, leading to the recruitment of the STAT5A or its paralog STAT5B. STAT5A/B is further phosphorylated by JAK3, but JAK1 could also be involved. Phosphorylated STAT5A/B is then released from IL-2R and dimerizes in the cytosol. The dimer can finally translocate to the nucleus and regulates genes encoding immune-related factors such as IL2RA itself, or again FOXP3, Fas-ligand (FASL), positive regulatory domain zinc finger protein 1 (PRDM1), or suppressor of cytokine signaling 1 or 2 (SOCS1/2). The phosphorylation of IL2RB also creates binding sites for the SHC-transforming protein 1 (SHC1), which is phosphorylated probably by JAK1 or Lck (still unclear). SHC1 then recruits the Src homology 2 domain containing inositol polyphosphate 5-phosphatase 1 (SHIP1). The SHC1/SHIP1 complex is stabilized through interaction with GRB2, itself associated with GRB2-associated binder 2 (GAB2). SHC1 promotes the phosphorylation of GAB2, but the kinase involved is not clear. Phosphorylated GAB2 can further recruit PI3Ks to the membrane, leading to the activation of the PI3K/PKB (best known as AKT)/mTOR pathway. JAK1 may also recruit PI3K. In the meantime, SHC1 recruits the complex GRB2/SOS. SOS interacts with, and activates, RasGRP1, resulting in the activation of the Ras/Raf pathway and the regulation of genes involved in T cell response. Sources: Reactome; KEGG pathway (Ross and Cantrell, 2018).
Binding of IL-2 to IL-2R activates JAK1/3. In turn, these kinases ignite the phosphoinositide-3-kinases (PI3Ks)/phosphatidylinositol 3,4,5-trisphosphate (PIP3)/AKT/mechanistic target of rapamycin (mTOR)/p70S6K, and Ras/Raf/mitogen-activated protein kinase kinases 1 and 2 (MAP2K1/2, also known as MEK1/2)/extracellular signal-regulated kinases 1 and 2 (ERK1/2) signaling cascades and phosphorylate STAT5. Activation of mTOR, p70S6K, AKT, and ERK1/2 modulates the activity and de novo expression of multiple downstream regulators involved in protein synthesis, autophagy, cell metabolism, survival, proliferation, and differentiation. Concomitantly, activated STAT5 determines the fate of the cell by transactivating numerous target genes (Fig. 2). While these three signaling cascades are stimulated in effector T cells, it is important to note that the STAT5 pathway is predominantly triggered in Tregs (Cheng et al., 2011). In T lymphocytes, depending on their subtype, STAT5-stimulated genes can encode the following: various cytokine receptors including IL-2Rα and IL-2Rβ (positive feedback), IL-4Rα or IL-12Rβ; diverse proteins involved in cell proliferation and survival (e.g., PIM1, MYC, cyclins, and B cell lymphoma (BCL) 2); some effector molecules such as granzyme B, CD178 (best known as Fas-ligand), and some cytokines like IFN-γ, TNF-α, or IL-4; and some regulators of immune cell functions such as the suppressor of cytokine signaling 1 and 2 (SOCS1/2), and the transcription factor forkhead box P3 (FOXP3). IL-2-activated STAT5 also represses the expression of particular effector genes such as IL17A or BCL6 (Kovanen et al., 2005; Grange et al., 2013; Knosp et al., 2013; Kanai et al., 2014; Ross and Cantrell, 2018). Phenotypic changes consecutive to IL-2 stimulation are described in the following paragraph.
Pleiotropic action of IL-2
IL-2 is a pleiotropic cytokine with immunostimulatory or immunoinhibitory activity depending on the target cell. Its effects on nonhematopoietic and innate immune cells remain poorly deciphered (Valle-Mendiola et al., 2016). Above all, IL-2 stands out as a well-established regulator of T cell development and homeostasis.
CD4+FOXP3+CD25+ Tregs are responsible for maintaining immunological self-tolerance and for down-regulating inflammatory and adaptive immune responses. As opposed to conventional T cells (Tconv cells), Tregs do not produce IL-2 but critically rely on its presence for their differentiation from immature single-positive CD4+ T cells in the thymus (referred to as natural/thymic Tregs) or from naive CD4+ T cells in the periphery (referred to as peripheral Tregs), when they acquire FOXP3, and hence for their expansion, survival, and immunoregulatory functions (including the secretion of the immunosuppressive cytokines IL-10 and TGF-β) in the periphery (Kündig et al., 1993; Sadlack et al., 1993; Suzuki et al., 1995; Willerford et al., 1995; Malek et al., 2002; D’Cruz and Klein, 2005; Fontenot et al., 2005; Burchill et al., 2007; Zheng and Rudensky, 2007; Malek, 2008; Tang et al., 2008; Campbell and Koch, 2011; Goldstein et al., 2013; Lin et al., 2013; Chinen et al., 2016; Ross and Cantrell, 2018).
Additionally, IL-2 regulates lineage commitment of CD4+ Th cell subsets. Th1 lymphocytes promote cellular immune responses against intracellular microbes and cancer cells. Th1 cell differentiation is triggered by IL-2–mediated expression of IL-12Rβ in naive CD4+ T cells. Then, together with IL-12 produced by APCs, IL-2 coordinates the expression of the transcription factor T-Box21 (TBX21, best known as T-bet), which orchestrates Th1 differentiation and the production of type 1 cytokines like IFN-γ (Reem and Yeh, 1984; Shi et al., 2008; Liao et al., 2011). Th2 lymphocytes regulate humoral immunity to extracellular parasites and bacteria, as well as allergic inflammation. In the presence of IL-4, IL-2 sensitizes cells to the Th2 program by up-regulating IL-4Rα expression and by inducing epigenetic changes that boost IL-4 secretion (Ben-Sasson et al., 1990; Zhu et al., 2003; Cote-Sierra et al., 2004; Le Gros et al., 2008; Liao et al., 2008). Th17 cells coordinate the adaptive defense against extracellular pathogens, mediate tissue inflammation, and participate in many autoimmune pathologies. IL-2 inhibits the differentiation of Th17 cells through various mechanisms including repression of retinoic acid receptor–related orphan nuclear receptor γt, IL-6Rα, and IL-17A. These three factors are required for the development and the proinflammatory activity of Th17 cells (Laurence et al., 2007; Liao et al., 2011; Yang et al., 2011). Accordingly, it has been reported that IL-2 consumption by Tregs facilitates Th17 differentiation in Candida albicans infection (Pandiyan et al., 2011). Similarly, IL-2 inhibits the differentiation of T follicular helper (Tfh) CD4+ T cells, which control B cell responses and germinal center formation. Thus, IL-2 activates the expression of positive regulatory domain zinc finger protein 1 (PRDM1, best known as B lymphocyte–induced maturation protein 1 [BLIMP-1]) which in turn trans-represses gene expression of BCL6, a transcriptional regulator required for Tfh development (Johnston et al., 2009; Ballesteros-Tato et al., 2012). Interestingly, in an influenza infection model, high levels of IL-2 prevented the development of T follicular regulatory (Tfr) cells in the course of the infection. However, once the virus was eliminated and the response resolved, some Tregs down-regulated IL-2Rα/CD25 and up-regulated BCL6 before differentiating into Tfr cells. These Tfr cells migrated to B cell follicles to prevent clonal expansion of self-reactive B lymphocytes (Botta et al., 2017).
In naive CD8+ T lymphocytes, IL-2 mediates the acquisition of the effector cytotoxic phenotype following antigen encounter by promoting the secretion of IFN-γ, TNF-α/β, granzyme B, and perforin. The intensity of the IL-2/IL-2R signaling activity shifts the fate of CD8+ T cells toward a short-lived effector or a long-lived memory phenotype. De facto, high levels of IL-2 stimulate the synthesis of BLIMP-1, which controls the effector transcriptional program and inhibits the expression of central memory markers such as BCL6, IL-7Rα/CD127, and CD62L. On the contrary, low levels of IL-2 impair the synthesis of effector molecules, while authorizing the (re)expression of BCL6, IL-7Rα/CD127, and CD62L, thus driving activated CD8+ T cells toward the memory compartment (Manjunath et al., 2001; Williams et al., 2006; Kalia et al., 2010; Pipkin et al., 2010; Ross and Cantrell, 2018; Spolski et al., 2018). Similarly, low doses of IL-2 drive a memory or Tfh-like phenotype in CD4+ T lymphocytes (Boyman and Sprent, 2012). Importantly, BLIMP-1 induction in effector cells ultimately leads to a reduced production of IL-2, accompanied by a contraction of the effector alcove and the appearance of an exhausted state (Gong and Malek, 2007; Beltra et al., 2016; Zhu et al., 2017). Interestingly, this exhausted phenotype, characterized by surface exposure of programmed cell death protein 1 (PD-1) or up-regulation of IL-2Rβ, is reversible. As evoked in the next section, this peculiarity may be exploited in immunostimulatory treatments by combining IL-2 and anti-PD1.
Immunosuppressive and immunostimulatory IL-2 therapies
The exquisite sensitivity of both effector and regulatory T lymphocytes to IL-2 has designated this cytokine as a potential ally for treating immune-related diseases, spurring interest in both its immunosuppressive and immunostimulatory effects. Fine-tuning of the balance between these two functionally contrasted T cell subsets is at the heart of IL-2–based immunotherapies. Immunoinhibitory IL-2 treatments aim at selectively expanding Tregs over effector T cells, whereas immunostimulatory IL-2 interventions should proceed inversely. To achieve these opposite goals, fundamental distinctions of the IL-2/IL-2R system within the targeted populations are being exploited. Thus, Tregs constitutively display the IL-2Rαβγ trimer, while activated effector T cells only transiently express IL-2Rα.
Strategies to improve immunosuppressive IL-2 therapy
When contemplating the addiction of Tregs to IL-2 and their preponderant role in maintaining immune tolerance and suppressing inflammation (Sakaguchi et al., 1995), it is a posteriori no surprise that inactivation of the IL2, IL2RA, IL2RB, STAT5, or FOXP3 genes in mice led to Treg cell depletion/dysfunction and to multi-organ autoimmune and inflammatory syndromes (Sadlack et al., 1993; Suzuki et al., 1995; Willerford et al., 1995; Malek et al., 2002; Fontenot et al., 2003; Snow et al., 2003; Burchill et al., 2007; Yao et al., 2007). Importantly, similar clinical manifestations have been detected in patients affected by mutations in IL2RA or FOXP3, leading to the immunodysregulation polyendocrinopathy enteropathy X-linked (IPEX) syndrome (Bennett et al., 2001; Wildin et al., 2001; Moraes-Vasconcelos et al., 2008; Goudy et al., 2013). Based on these observations, reestablishing Treg cell functions thanks to the administration of recombinant human IL-2 has been considered for the treatment of autoimmune, inflammatory, or graft-versus-host (GvHD) diseases (Krömer et al., 1985; Andreu-Sánchez et al., 1991; Koreth et al., 2011; Saadoun et al., 2011; Hartemann et al., 2013; Matsuoka et al., 2013; Castela et al., 2014; He et al., 2016).
For immunosuppression, the common strategy relies on the administration of low-dose (LD) IL-2 (as opposed to HD IL-2, which has been clinically approved for cancer therapy), based on the rationale that limited concentration of IL-2 restricts its capture to Tregs (rather than Tconv cells), as Tregs constitutively express high amounts of the high-affinity IL-2Rαβγ (Klatzmann and Abbas, 2015). The proof of concept of this approach was attained when LD IL-2 reverted and prevented experimental type 1 diabetes (T1D) in mice (Tang et al., 2008; Webster et al., 2009; Yu et al., 2009; Grinberg-Bleyer et al., 2010; Baeyens et al., 2013; Pérol and Piaggio, 2016). LD IL-2 also achieved disease control in other indications, but only when delivered chronically, like in Alzheimer’s disease (Dansokho et al., 2016), or when combined with other drugs such as glucocorticoids in GvHD (Pérol and Piaggio, 2016) and Trypanosoma cruzi infection (González et al., 2015) or rapamycin in skin transplantation (Pérol and Piaggio, 2016). In the clinic, LD IL-2 has been evaluated for the treatment of T1D (Long et al., 2012; Hartemann et al., 2013; Todd et al., 2016), chronic GvHD (Koreth et al., 2011, 2016; Matsuoka et al., 2013; Kennedy-Nasser et al., 2014), systemic lupus erythematosus (SLE; He et al., 2016; Humrich and Riemekasten, 2016), alopecia aereata (Castela et al., 2014), and vasculitis associated with chronic hepatitis C virus infection (Saadoun et al., 2011). Overall, LD IL-2 was well tolerated, not only due to the dosing (<3 × 106 IU/d) but also due to a preference for s.c., rather than systemic, administration. In all these pathological conditions, LD IL-2 led to an increase in circulating Tregs, and some clinical responses were observed. Nevertheless, to increase clinical efficacy, improvements are needed as IL-2 has a short half-life, requires repeated administration, and can unwantedly activate NK cells (Ye et al., 2018).
The short half-life of IL-2 (10–85 min in serum) is intrinsic to its small molecular weight that falls beneath the glomerular filtration cutoff estimated at 30–50 kD (Konrad et al., 1990; Ruggiero et al., 2010). Hence, to maintain efficient IL-2 bioavailability, administration must be repeated at close intervals. Alternatively, prodrug versions of recombinant human IL-2 decorated with releasable polyethylene glycol (PEG) chains have been synthesized and allowed: (i) a sustained release of the cytokine; (ii) a prolonged stimulation of the IL-2R signaling pathway; and (iii) a biased binding of IL-2 either to IL-2Rα/CD25 or IL-2Rβ/CD122, depending on the sites of PEGylation (Charych et al., 2017). For instance, a PEGylated IL-2 named NKTR-358 demonstrates a lower affinity for IL-2Rβ than for IL-2Rα and preferentially activates Tregs over Tconv cells. NKTR-358 restored Treg function in murine and simian models of SLE and cutaneous hypersensitivity, respectively, encouraging its clinical evaluation in patients (Cully, 2017; Table 1).
Table 1. Examples of innovative immunosuppressive IL-2 therapies undergoing clinical evaluation.
| IL-2 therapy | Clinical trial | |||||||
|---|---|---|---|---|---|---|---|---|
| Type | Agent (company) | Description | Delivery route | Indication | Co-therapy | Phase | Status | References |
| Immunocytokine | AMG-592/efavaleukin-alpha (Amgen) | Fc–IL-2 mutein fusion protein | s.c. | SLE | – | 1/2 | Recruiting | NCT03451422 (Tchao et al., 2017) |
| s.c. | RA | – | 1/2 | Recruiting | NCT03410056 (Tchao et al., 2017) | |||
| s.c. | Chronic GvHD | – | 1/2 | Recruiting | NCT03422627 (Tchao et al., 2017) | |||
| PEGylated IL-2 | NKTR-358 (Nektar Therapeutics) | PEG–IL-2 with IL-2Rα bias > IL-2Rβ bias | i.v. | SLE | – | 1 | Recruiting | NCT03556007 |
RA, rheumatoid arthritis.
IL-2/anti–IL-2 antibody immunocomplexes (referred to as “IL2Cxs”) are also designed to increase the half-life of IL-2 (Létourneau et al., 2010). Moreover, depending on the fine specificity of the antibody, IL2Cxs can redirect IL-2 toward IL-2Rαβγ+ Tregs (pro–Treg cell function) or IL-2Rβγ+ NK and naive/memory CD8+ T cells (pro-effector; Boyman et al., 2006). For instance, the mAb JES6-1 recognizes an epitope on murine IL-2 that contacts IL-2Rβ and γ. Interestingly, JES6-1 sterically blocks the interaction of IL-2 with IL-2Rβ and allosterically reduces IL-2 affinity for IL-2Rα. Experimentally, IL-2/JES6-1 selectively binds to and expands Tregs as they present sufficient IL-2Rα to displace the mAb (Spangler et al., 2015). Pro-Treg IL2Cxs have been successfully used in mouse models of experimental autoimmune encephalomyelitis (Webster et al., 2009), diabetes (Pérol and Piaggio, 2016), allergy (Smaldini et al., 2018), atherosclerosis (Dinh et al., 2012), and solid organ transplantation (Pérol and Piaggio, 2016). When complexed to recombinant human IL-2, the first clinical grade pro-Treg anti–IL-2, named F5111.2, demonstrated effectiveness in inducing T1D remission in diabetic mice, and in diminishing the severity of xeno-GvHD and experimental autoimmune encephalomyelitis (Trotta et al., 2018).
An alternative approach consisting in fusing IL-2 to the α chain of IL-2R has recently been evaluated in rodents (Ward et al., 2018). This IL-2–CD25 fusion protein exhibited an increased half-life and selectively expanded Tregs in vivo. In nonobese diabetic mice, administration of IL-2–CD25 enhanced Tregs in the endocrine pancreas and diminished the occurrence of diabetes. These results encourage the clinical evaluation of such a new class of IL-2 derivatives for treating autoimmunity and other pathologies that result from an exacerbated immune response (Ward et al., 2018).
Elucidation of the quaternary structure of IL-2 assembled to IL-2Rαβγ (Wang et al., 2005) has facilitated the engineering of a series of IL-2 “muteins” with variable affinity to IL-2Rα (Rao et al., 2003, 2005; Carmenate et al., 2013), IL-2Rβ (Liu et al., 2009; Levin et al., 2012; Mitra et al., 2015; Peterson et al., 2018), and IL-2Rγ (Liu et al., 2009; Mitra et al., 2015; Carmenate et al., 2018). These IL-2 analogues elicit graded (agonistic, mixed, or antagonistic) and differential signaling outputs downstream of IL-2Rαβγ and IL-2Rαβ, ultimately affecting the Treg/effector ratio. For instance, the IL-2 mutein H9-RETR (nine amino acid substitutions) was engineered to bind IL-2Rβ with a rather high affinity and block its heterodimerization with IL-2Rγ. H9-RETR antagonized signal transduction by native IL-2 and prevented ex vivo proliferation of pre-activated human CD8+ T cells, as well as the cytolytic activity of NK cells (Mitra et al., 2015).
Like their WT counterpart, IL-2 muteins suffer from limited bioavailability in vivo. As a remedy, they are frequently fused with an mAb or a crystallizable fragment (Fc) of an antibody, thus generating so called “immunocytokines.” Lately, a human IL-2 mutein harboring an N88D substitution responsible for a reduced affinity for IL-2Rβγ was fused to a nontargeted effector-function-silent human IgG1. In macaques, this long-lived IgG-(IL-2N88D)2 fusion protein sustained preferential amplification of Tregs (Peterson et al., 2018). In a GvHD murine model, the stabilized antagonist H9-RETR-Fc4 remarkably extended survival (Mitra et al., 2015). Preclinically, several IL-2–Fc molecules demonstrated therapeutic ability to induce transplantation tolerance (Zheng et al., 2003; Millington et al., 2012; Jindal et al., 2015; Mitra et al., 2015) or to prevent autoimmune disorders (Zheng et al., 1999; Bell et al., 2015). An Fc–IL-2 mutein called AMG-592 (Amgen) was designed to have greater half-life than native IL-2, as well as an increased affinity for IL-2Rα. In human peripheral blood mononuclear cell cultures, AMG-592 preferentially expanded Tregs over effector T cells and lowered the production of pro-inflammatory cytokines in comparison to native IL-2. In an ongoing first-in-human trial, AMG-592 was well tolerated without severe adverse events and increased the Treg/Tconv cell ratio (Tchao et al., 2017). AMG is now being evaluated in clinical trials for the treatment of rheumatoid arthritis, SLE, and GvHD (Table 1). Another Fc–IL-2 mutein with increased affinity for IL-2Rα, named DEL-106 (Delinia-Celgene-BMS), is under development (Cully, 2017).
Strategies to improve immunostimulatory IL-2 therapy
Mathematical prediction models, validated by experimentations, demonstrated that Tregs locally outcompete Tconv cells in consuming IL-2, as long as its autocrine/paracrine level does not reach the T cell activation threshold. Beyond this point, activated T cells engage a positive feedback loop that will temporarily up-regulate the high-affinity IL-2R, support their proliferation, and secure their effector program (Busse et al., 2010).
In line with these theoretical considerations, i.v. infusions of HD IL-2 (6–7.2 × 105 IU/kg/dose, 12–15 doses/d) have been conceived for the treatment of malignant and infectious diseases. In Western countries, HD IL-2 received approval for the care of metastatic renal cell carcinoma and melanoma in the 1990s (Alva et al., 2016). Seminal publications reported an overall response rate (ORR) of 14–23% (including 5–8% of complete responders [CRs]) in patients with kidney cancer (Fyfe et al., 1995, 1996) and a 16% ORR (6% CRs) in melanoma patients (Atkins et al., 1999). In the field of infectious disease, HD IL-2 has essentially been evaluated in AIDS as an adjuvant to antiretroviral therapy. A meta-analysis compiling data from 25 completed trials revealed an increase in the CD4+ T cell count in the presence of IL-2. However, IL-2 supplementation did not reduce mortality or the risk of opportunistic infections, and even tended to increase the rate of severe adverse events, thus discouraging further investigations in HIV-positive patients (Onwumeh et al., 2017).
The mitigated success of HD IL-2–based cancer therapy can be attributed to the following: (i) its short bioavailability; (ii) an undesired expansion of Tregs that dampens antitumor immunity; and (iii) its dose-dependent toxicity. Indeed, elevated systemic (endocrine) levels of cytokines are nonphysiological (because cytokines are by definition paracrine factors) and hence accompanied by deleterious effects. Thus, systemic injection of IL-2 causes vascular leak syndrome due to bystander damage of IL-2Rαβγ+ endothelial cells and due to the unwarranted release of pro-inflammatory cytokines from T and NK cells (Epstein et al., 2003; Boyman and Arenas-Ramirez, 2019).
On one hand, the deleterious side effects of HD IL-2 must be attenuated to enhance its therapeutic index. Lower dosages and alternative delivery routes, such as s.c., i.m., or intralesional injections, have been tested (Tang and Harding, 2019). Only intratumoral administration outperformed standard HD i.v. IL-2, reaching up to 62% CR in melanoma patients with skin and soft-tissue metastases (Konrad et al., 1990; Palmer et al., 1993; Ravaud et al., 2002; Radny et al., 2003; Yang et al., 2003; Geertsen et al., 2004).
On the other hand, the therapeutic index of HD IL-2 may be improved by the selective stimulation of effector cells instead of Tregs. Multiple groups are introducing IL-2 in combinatorial regimens alongside immunomodulatory regimens, with the hope of improving efficacy and eventually reducing IL-2 dosage. Thus, ≥45 trials are active, recruiting patients in diverse oncological indications (http://www.clinicaltrials.gov) to evaluate HD IL-2 in combination with surgery, chemotherapy, radiotherapy, adoptive T cell therapy, anti–PD-1 or anti–CTLA-4, tumor-targeting mAbs, or cancer vaccines.
In parallel, attempts are ongoing to modify the pharmacological properties of IL-2. For example, pro-effector IL2Cxs in which an anti–IL-2 antibody sterically impedes the interaction between IL-2 and IL-2Rα, but allosterically enhances IL-2 affinity for IL-2Rβ, are being developed (Spangler et al., 2015). Antibodies with such characteristics include S4B6 (anti-mouse IL-2), as well as Mab602 and NARA-1 (anti-human IL-2; Boyman and Arenas-Ramirez, 2019). Preclinical studies in melanoma-bearing mice revealed superiority of S4B6 IL2Cxs over free IL-2 because: (i) the IL-2 interaction with IL-2Rα+ endothelial cells was disrupted and vascular leak syndrome prevented; and (ii) naive IL-2Rβγ+ CD8+ T and NK cells were preferentially amplified over IL-2Rαβγ+ Tregs. Encouragingly, IL2Cxs showed greater antitumor activity and some synergy with immune checkpoint inhibitors (Krieg et al., 2010; Létourneau et al., 2010; Arenas-Ramirez et al., 2016; Caudana et al., 2019).
A PEGylated IL-2 named NKTR-254 demonstrated a much more reduced affinity for IL-2Rα than for IL-2Rβ. A Phase 1/2 clinical trial has recently been completed in patients with late stage solid tumors (NCT02869295). Preliminary results indicate a favorable safety profile, a 10–30% shrinkage of the tumor burden in 23% of the patients, and an increase of tumor-infiltrating CD8+ T and NK cells with minimal impact on Tregs (Marin-Acevedo et al., 2018). Additional Phase 1–3 trials are now enrolling patients (Table 2).
Table 2. Examples of innovative immunostimulatory IL-2 therapies undergoing clinical evaluation.
| IL-2 therapy | Clinical trial | |||||||
|---|---|---|---|---|---|---|---|---|
| Type | Agent (company) | Description | Delivery route | Indication | Co-therapy | Phase | Status | References |
| Immunocytokine | ALKS 4230 (Alkermes) | IL-2–CD25 fusion protein | i.v. | Solid tumors | Pembrolizumab | 1/2 | Recruiting | NCT02799095 (Vaishampayan et al., 2017) |
| s.c. | Solid tumors | Pembrolizumab | 1 | Recruiting | NCT03861793 (Vaishampayan et al., 2017) | |||
| Darleukin (Philogen) | Anti-fibronectin diabody–IL-2 fusion protein | i.v. (?) | NSCLC | Radiotherapy; surgery | 2 | Not yet recruiting | NCT03705403 (Johannsen et al., 2010; Eigentler et al., 2011; Danielli et al., 2015) | |
| EMD273063/hu14.18-IL2 (Merck) | Anti–GD2–IL-2 fusion protein | i.v. | Melanoma | Surgery | 2 | Active, not recruiting | NCT00590824 | |
| i.t. | Melanoma | Radiotherapy; nivolumab; ipilimumab | 1/2 | Not yet recruiting | NCT03958383 | |||
| i.v. | Neuroblastoma | EEAHD NK cells | 1 | Recruiting | NCT03209869 | |||
| RG7461/RO6874281 (Roche) | Anti–FAP–IL-2 mutein (F42A, Y45A, and L72G) fusion protein | i.v. | NSCLC; SCCHN; ESCC; cervical cancer | Atezolizumab; gemcitabine; vinorelbine | 2 | Recruiting | NCT03386721 (Klein et al., 2017) | |
| i.v. | Pancreatic cancer | Atezolizumab | 1/2 | Recruiting | NCT03193190 (Klein et al., 2017) | |||
| i.v. | TNBC | Atezolizumab | 1/2 | Recruiting | NCT03424005 (Klein et al., 2017) | |||
| i.v. | Melanoma | Pembrolizumab | 1b | Recruiting | NCT03875079 (Klein et al., 2017) | |||
| i.v. | Breast cancer; HNC; other solid tumors | Trastuzumab; cetuximab | 1 | Recruiting | NCT02627274 (Klein et al., 2017) | |||
| i.v. | RCC | Atezolizumab; bevacizumab | 1 | Active, not recruiting | NCT03063762 (Klein et al., 2017) | |||
| RG7813/cergutuzumab amunaleukin (Roche) | Anti–CEA–IL-2 mutein (F42A, Y45A, and L72G) fusion protein | i.v. | Solid tumors | Atezolizumab | 1 | Active, not recruiting | NCT02350673 (Klein et al., 2017; Lo et al., 2017) | |
| PEGylated IL-2 | NKTR-214 (Nektar Therapeutics) | PEG–IL-2 with IL-2Rβ bias > IL-2Rα bias | i.v. | Melanoma | Nivolumab | 3 | Recruiting | NCT03635983 (Charych et al., 2016) |
| i.v. | RCC | Sunitinib; nivolumab; cabozantinib | 3 | Recruiting | NCT03729245 (Charych et al., 2016) | |||
| i.v. | Bladder cancer | Nivolumab | 2 | Recruiting | NCT03785925 (Charych et al., 2016) | |||
| i.v. | Sarcoma | Nivolumab | 2 | Recruiting | NCT03282344 (Charych et al., 2016) | |||
| i.v. | Melanoma; RCC; NSCLC; urothelial cancer; TNBC | Nivolumab; ipilimumab | 1/2 | Recruiting | NCT02983045 (Charych et al., 2016) | |||
| i.v. | Skin cancers (melanoma, MCC); TNBC; ovarian cancer; RCC; CRC; urothelial cancer; sarcoma | NKTR-262; Nivolumab | 1/2 | Recruiting | NCT03435640 (Charych et al., 2016) | |||
| i.v. | NHL | TAK-659 | 1 | Recruiting | NCT03772288 (Charych et al., 2016) | |||
| i.v. | NSCLC; bladder cancer; melanoma | Pembrolizumab; atezolizumab | 1 | Recruiting | NCT03138889 (Charych et al., 2016) | |||
| i.v. | Prostate cancer | Nivolumab | 1 | Recruiting | NCT03835533 (Charych et al., 2016) | |||
| i.v. | Solid tumors | Nivolumab | 1 | Recruiting | NCT03745807 (Charych et al., 2016) | |||
CEA, carcinoembryonic antigen; CRC, colorectal cancer; EEAHD, ex vivo expanded and activated haploidentical donor; ESCC, esophageal squamous cell carcinoma; FAP, fibroblast activation protein-alpha; GD2, ganglioside D2; HNC, head and neck cancer; i.t., intratumoral; MCC, Merkel cell carcinoma; NHL, Non-Hodgkin lymphoma; NSCLC, non-small cell lung cancer; RCC, renal cell carcinoma; SCCHN, squamous cell carcinoma of the head and neck; TNBC, triple negative breast cancer.
Based on in vitro evolution assays and protein crystallography, Levin et al. (2012) designed the IL-2 mutein H9 (the IL-2 agonist that sourced H9-RETR). H9 harbors five mutations affecting core residues (i.e., L80F, R80D, L85V, I86V, and I92F). These substitutions induce a natural conformational switch responsible for a much stronger interaction with IL-2Rβ and an optimal downstream signaling activation not requiring IL-2Rα (Levin et al., 2012). The so called “superkine” H9 favored the expansion of CD8+ T and NK cells, demonstrated lower toxicity, and improved antitumor activity in several murine cancer models. Interestingly, IL-2 superkines demonstrated similar therapeutic activity as immunocomplexed WT IL-2 (Levin et al., 2012; Tang and Harding, 2019). In the clinic, the IL-2 mutein BAY50-4798 (Bayer) failed to demonstrate sufficient efficacy in advanced melanoma and renal cancer (ORR < 5%), leading to its discontinuation (Margolin et al., 2007). Retrospectively, dual introduction of an IL-2Rα bias plus the N88R mutation (which disfavors IL-2/IL-2Rβ interaction; Peterson et al., 2018) likely conferred an undesired pro-Treg cell activity to BAY50-4798.
Immunocytokines, which are hybrid proteins composed of WT or mutant IL-2 fused to Fc domains or tumor-targeting mAbs, are being developed. Signs of efficacy were observed in preclinical melanoma models treated with IL-2–anti-ganglioside GD2 (Becker et al., 1996a,b). The humanized version of such a construct, called EMD273063 (Merck), demonstrated a weak efficacy in metastatic melanoma (ORR = 7.1%; Albertini et al., 2012). Additional immunocytokines already completed clinical evaluations such as an anti-EpCAM-IL-2 (EMD273066/tucotuzumab celmoleukin), an anti-single/double-stranded DNA-(D20T)IL-2 mutein (EMD521873), and a fibronectin-targeted L19 diabody-IL-2 (Darleukin), with no or only marginal responses (Johannsen et al., 2010; Eigentler et al., 2011; Albertini et al., 2012; Connor et al., 2013; Gillessen et al., 2013; Laurent et al., 2013; van den Heuvel et al., 2015). However, as a possible exception to this rule, intralesional therapy of metastatic melanoma with a combination of recombinant IL-2 protein with the human mAb fragment L19 (L19–IL-2) together with another similar protein in which IL-2 is replaced by TNF-α (L19-TNF-α) showed a 53.8% ORR and induced abscopal effects (Danielli et al., 2015). An alternative strategy consists in fusing IL-2 with IL-2Rα to sterically prevent interactions with the high-affinity receptor. Such a compound, ALKS4230, is being tested in two Phase 1/2 trials in combination with the anti-PD1 antibody pembrolizumab (Table 2).
Another procedure applied to chimeric antigen receptor T cell technology is exemplified by the ortho–IL-2/ortho–IL-2Rβ system. It consists of a mutated IL-2Rβ, ortho–IL-2Rβ, which no longer binds native IL-2 but its mutant form, ortho–IL-2. The expression of ortho–IL-2Rβ on chimeric antigen receptor T cells confers selective proliferation following ortho–IL-2 administration, avoiding bystander activation of other IL-2R+ cells like Tregs (Sockolosky et al., 2018).
Finally, Garcia’s team generated a computationally designed IL-2/15 hybrid molecule of 100 amino acids that differs from the natural IL-2 not only in sequence but also in topology, ultimately preserving its ligation to IL-2Rβγ but not to IL-2Rα. This so called “neoleukin” Neo-2/15 showed greater antitumor activity and tolerance than WT IL-2 in preclinical experiments (Silva et al., 2019).
Concluding remarks
Layer after layer, we keep uncovering the complexity of IL-2 biology and incrementing IL-2 therapy. Following decades of deceptions, we may finally meet broader success. In this line, standard therapy with nonmodified recombinant IL-2 protein still has room for improvement, as illustrated by its increased therapeutic index following an intralesional route. Regarding the innovative approaches embodied by IL-2 muteins such as superkines or antagonists, immunocomplexes, and immunocytokines, drawbacks have emerged. They include the intrinsic immunogenicity and poor bioavailability of muteins, the potential disassembly of immunocomplexes in vivo, or the unexpected depletion of T cell subsets targeted by IL-2 mutein-Fc immunocytokines (Vazquez-Lombardi et al., 2017). Additional rounds of preclinical research and clinical investigations will likely be necessary to unchain the full therapeutic potential of agents targeting the IL-2/IL-2R system. Most importantly, immunomodulatory combination therapies, particularly with immune checkpoint inhibitors, have proven preclinical efficiency. In the clinic, administration of HD IL-2 appears essential for the efficacy of cancer treatments relying on the adoptive transfer of ex vivo expanded autologous T cells (e.g., tumor-infiltrating lymphocytes, T cells with an engineered TCR, or a chimeric antigen receptor) with remarkable objective response rates witnessed in melanoma (34–56%), lymphoma (80–100%), and leukemia (67–100%; Rosenberg and Restifo, 2015; Boyiadzis et al., 2018). In this setting, IL-2–based therapeutics could emancipate as adjuvant agents that will enhance the efficacy of established immunotherapeutics.
Acknowledgments
G. Kroemer is supported by the Ligue contre le Cancer (équipe labelisée); Agence National de la Recherche—Projets blancs; Agence National de la Recherche under the frame of E-Rare-2, the ERA-Net for Research on Rare Diseases; Association pour la Recherche sur le Cancer; Association “Le Cancer du Sein, Parlons-en!,” Cancéropôle Ile-de-France; Chancellerie des Universités de Paris (Legs Poix), Fondation pour la Recherche Médicale; a donation by Elior; European Research Area Network on Cardiovascular Diseases Project on Metabolic Therapy for Managing Diastolic Heart Failure; Gustave Roussy Odyssea, the European Union Horizon 2020 Project Oncobiome; Fondation Carrefour; High-end Foreign Expert Program in China (GDW20171100085), Institut National du Cancer; Institut National de la Santé et de la Recherche Médicale Hétérogénéité Tumorale et Ecosystème; Institut Universitaire de France; LeDucq Foundation; the LabEx Immuno-Oncology (ANR-18-IDEX-0001); the Recherche Hospitalo-Universitaire en santé Torino Lumière; the Seerave Foundation; the Site de Recherche Intégrée sur le Cancer (SIRIC) Stratified Oncology Cell DNA Repair and Tumor Immune Elimination; and the Site de Recherche Intégrée sur le Cancer Cancer Research and Personalized Medicine.
G. Kroemer has been holding research contracts with Bayer Healthcare, Genentech, Glaxo Smyth Kline, Institut Mérieux, Lytix Pharma, PharmaMar, Sotio, and Vasculox. G. Kroemer is the chair of the Scientific Advisory Board of Medicenna and a member of the Board of Directors of the Bristol Myers Squibb Foundation France. G. Kroemer is a scientific co-founder of everImmune and Samsara Therapeutics. The other authors declare no competing financial interests.
Author contributions: J.G. Pol and P. Caudana wrote the manuscript. J.G. Pol and J. Paillet made the tables and figures and wrote the legends. E. Piaggio and G. Kroemer designed the plan of the review and edited the manuscript.
References
- Albertini M.R., Hank J.A., Gadbaw B., Kostlevy J., Haldeman J., Schalch H., Gan J., Kim K., Eickhoff J., Gillies S.D., et al. . 2012. Phase II trial of hu14.18-IL2 for patients with metastatic melanoma. Cancer Immunol. Immunother. 61:2261–2271. 10.1007/s00262-012-1286-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allison J.P., McIntyre B.W., and Bloch D.. 1982. Tumor-specific antigen of murine T-lymphoma defined with monoclonal antibody. J. Immunol. 129:2293–2300. [PubMed] [Google Scholar]
- Alva A., Daniels G.A., Wong M.K., Kaufman H.L., Morse M.A., McDermott D.F., Clark J.I., Agarwala S.S., Miletello G., Logan T.F., et al. . 2016. Contemporary experience with high-dose interleukin-2 therapy and impact on survival in patients with metastatic melanoma and metastatic renal cell carcinoma. Cancer Immunol. Immunother. 65:1533–1544. 10.1007/s00262-016-1910-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andreu-Sánchez J.L., Moreno de Alborán I.M., Marcos M.A., Sánchez-Movilla A., Martínez-A C., and Kroemer G.. 1991. Interleukin 2 abrogates the nonresponsive state of T cells expressing a forbidden T cell receptor repertoire and induces autoimmune disease in neonatally thymectomized mice. J. Exp. Med. 173:1323–1329. 10.1084/jem.173.6.1323 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arenas-Ramirez N., Zou C., Popp S., Zingg D., Brannetti B., Wirth E., Calzascia T., Kovarik J., Sommer L., Zenke G., et al. . 2016. Improved cancer immunotherapy by a CD25-mimobody conferring selectivity to human interleukin-2. Sci. Transl. Med. 8:367ra166 10.1126/scitranslmed.aag3187 [DOI] [PubMed] [Google Scholar]
- Atkins M.B., Lotze M.T., Dutcher J.P., Fisher R.I., Weiss G., Margolin K., Abrams J., Sznol M., Parkinson D., Hawkins M., et al. . 1999. High-dose recombinant interleukin 2 therapy for patients with metastatic melanoma: Analysis of 270 patients treated between 1985 and 1993. J. Clin. Oncol. 17:2105–2116. 10.1200/JCO.1999.17.7.2105 [DOI] [PubMed] [Google Scholar]
- Baeyens A., Pérol L., Fourcade G., Cagnard N., Carpentier W., Woytschak J., Boyman O., Hartemann A., and Piaggio E.. 2013. Limitations of IL-2 and rapamycin in immunotherapy of type 1 diabetes. Diabetes. 62:3120–3131. 10.2337/db13-0214 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ballesteros-Tato A., León B., Graf B.A., Moquin A., Adams P.S., Lund F.E., and Randall T.D.. 2012. Interleukin-2 inhibits germinal center formation by limiting T follicular helper cell differentiation. Immunity. 36:847–856. 10.1016/j.immuni.2012.02.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Becker J.C., Pancook J.D., Gillies S.D., Furukawa K., and Reisfeld R.A.. 1996a T cell-mediated eradication of murine metastatic melanoma induced by targeted interleukin 2 therapy. J. Exp. Med. 183:2361–2366. 10.1084/jem.183.5.2361 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Becker J.C., Pancook J.D., Gillies S.D., Mendelsohn J., and Reisfeld R.A.. 1996b Eradication of human hepatic and pulmonary melanoma metastases in SCID mice by antibody-interleukin 2 fusion proteins. Proc. Natl. Acad. Sci. USA. 93:2702–2707. 10.1073/pnas.93.7.2702 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bell C.J., Sun Y., Nowak U.M., Clark J., Howlett S., Pekalski M.L., Yang X., Ast O., Waldhauer I., Freimoser-Grundschober A., et al. . 2015. Sustained in vivo signaling by long-lived IL-2 induces prolonged increases of regulatory T cells. J. Autoimmun. 56:66–80. 10.1016/j.jaut.2014.10.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beltra J.C., Bourbonnais S., Bédard N., Charpentier T., Boulangé M., Michaud E., Boufaied I., Bruneau J., Shoukry N.H., Lamarre A., et al. . 2016. IL2Rβ-dependent signals drive terminal exhaustion and suppress memory development during chronic viral infection. Proc. Natl. Acad. Sci. USA. 113:E5444–E5453. 10.1073/pnas.1604256113 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bennett C.L., Christie J., Ramsdell F., Brunkow M.E., Ferguson P.J., Whitesell L., Kelly T.E., Saulsbury F.T., Chance P.F., and Ochs H.D.. 2001. The immune dysregulation, polyendocrinopathy, enteropathy, X-linked syndrome (IPEX) is caused by mutations of FOXP3. Nat. Genet. 27:20–21. 10.1038/83713 [DOI] [PubMed] [Google Scholar]
- Ben-Sasson S.Z., Le Gros G., Conrad D.H., Finkelman F.D., and Paul W.E.. 1990. IL-4 production by T cells from naive donors. IL-2 is required for IL-4 production. J. Immunol. 145:1127–1136. [PubMed] [Google Scholar]
- Bosco M.C., Curiel R.E., Zea A.H., Malabarba M.G., Ortaldo J.R., and Espinoza-Delgado I.. 2000. IL-2 signaling in human monocytes involves the phosphorylation and activation of p59hck. J. Immunol. 164:4575–4585. 10.4049/jimmunol.164.9.4575 [DOI] [PubMed] [Google Scholar]
- Botta D., Fuller M.J., Marquez-Lago T.T., Bachus H., Bradley J.E., Weinmann A.S., Zajac A.J., Randall T.D., Lund F.E., León B., et al. . 2017. Dynamic regulation of T follicular regulatory cell responses by interleukin 2 during influenza infection. Nat. Immunol. 18:1249–1260. 10.1038/ni.3837 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boyiadzis M.M., Dhodapkar M.V., Brentjens R.J., Kochenderfer J.N., Neelapu S.S., Maus M.V., Porter D.L., Maloney D.G., Grupp S.A., Mackall C.L., et al. . 2018. Chimeric antigen receptor (CAR) T therapies for the treatment of hematologic malignancies: Clinical perspective and significance. J. Immunother. Cancer. 6:137 10.1186/s40425-018-0460-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boyman O., and Arenas-Ramirez N.. 2019. Development of a novel class of interleukin-2 immunotherapies for metastatic cancer. Swiss Med. Wkly. 149:w14697. [DOI] [PubMed] [Google Scholar]
- Boyman O., and Sprent J.. 2012. The role of interleukin-2 during homeostasis and activation of the immune system. Nat. Rev. Immunol. 12:180–190. 10.1038/nri3156 [DOI] [PubMed] [Google Scholar]
- Boyman O., Kovar M., Rubinstein M.P., Surh C.D., and Sprent J.. 2006. Selective stimulation of T cell subsets with antibody-cytokine immune complexes. Science. 311:1924–1927. 10.1126/science.1122927 [DOI] [PubMed] [Google Scholar]
- Burchill M.A., Yang J., Vogtenhuber C., Blazar B.R., and Farrar M.A.. 2007. IL-2 receptor β-dependent STAT5 activation is required for the development of Foxp3+ regulatory T cells. J. Immunol. 178:280–290. 10.4049/jimmunol.178.1.280 [DOI] [PubMed] [Google Scholar]
- Busse D., de la Rosa M., Hobiger K., Thurley K., Flossdorf M., Scheffold A., and Höfer T.. 2010. Competing feedback loops shape IL-2 signaling between helper and regulatory T lymphocytes in cellular microenvironments. Proc. Natl. Acad. Sci. USA. 107:3058–3063. 10.1073/pnas.0812851107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campbell D.J., and Koch M.A.. 2011. Phenotypical and functional specialization of FOXP3+ regulatory T cells. Nat. Rev. Immunol. 11:119–130. 10.1038/nri2916 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carmenate T., Pacios A., Enamorado M., Moreno E., Garcia-Martínez K., Fuente D., and León K.. 2013. Human IL-2 mutein with higher antitumor efficacy than wild type IL-2. J. Immunol. 190:6230–6238. 10.4049/jimmunol.1201895 [DOI] [PubMed] [Google Scholar]
- Carmenate T., Ortíz Y., Enamorado M., García-Martínez K., Avellanet J., Moreno E., Graça L., and León K.. 2018. Blocking IL-2 signal in vivo with an IL-2 antagonist reduces tumor growth through the control of regulatory T cells. J. Immunol. 200:3475–3484. 10.4049/jimmunol.1700433 [DOI] [PubMed] [Google Scholar]
- Castela E., Le Duff F., Butori C., Ticchioni M., Hofman P., Bahadoran P., Lacour J.P., and Passeron T.. 2014. Effects of low-dose recombinant interleukin 2 to promote T-regulatory cells in alopecia areata. JAMA Dermatol. 150:748–751. 10.1001/jamadermatol.2014.504 [DOI] [PubMed] [Google Scholar]
- Caudana P., Núñez N.G., De La Rochere P., Pinto A., Denizeau J., Alonso R., Niborski L.L., Lantz O., Sedlik C., and Piaggio E.. 2019. IL2/Anti-IL2 complex combined with CTLA-4, but not PD-1, blockade rescues antitumor NK cell function by regulatory T-cell modulation. Cancer Immunol. Res. 7:443–457. 10.1158/2326-6066.CIR-18-0697 [DOI] [PubMed] [Google Scholar]
- Charych D.H., Hoch U., Langowski J.L., Lee S.R., Addepalli M.K., Kirk P.B., Sheng D., Liu X., Sims P.W., VanderVeen L.A., et al. . 2016. NKTR-214, an engineered cytokine with biased IL2 receptor binding, increased tumor exposure, and marked efficacy in mouse tumor models. Clin. Cancer Res. 22:680–690. 10.1158/1078-0432.CCR-15-1631 [DOI] [PubMed] [Google Scholar]
- Charych D., Khalili S., Dixit V., Kirk P., Chang T., Langowski J., Rubas W., Doberstein S.K., Eldon M., Hoch U., and Zalevsky J.. 2017. Modeling the receptor pharmacology, pharmacokinetics, and pharmacodynamics of NKTR-214, a kinetically-controlled interleukin-2 (IL2) receptor agonist for cancer immunotherapy. PLoS One. 12:e0179431 10.1371/journal.pone.0179431 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng G., Yu A., and Malek T.R.. 2011. T-cell tolerance and the multi-functional role of IL-2R signaling in T-regulatory cells. Immunol. Rev. 241:63–76. 10.1111/j.1600-065X.2011.01004.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chinen T., Kannan A.K., Levine A.G., Fan X., Klein U., Zheng Y., Gasteiger G., Feng Y., Fontenot J.D., and Rudensky A.Y.. 2016. An essential role for the IL-2 receptor in Treg cell function. Nat. Immunol. 17:1322–1333. 10.1038/ni.3540 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Connor J.P., Cristea M.C., Lewis N.L., Lewis L.D., Komarnitsky P.B., Mattiacci M.R., Felder M., Stewart S., Harter J., Henslee-Downey J., et al. . 2013. A phase 1b study of humanized KS-interleukin-2 (huKS-IL2) immunocytokine with cyclophosphamide in patients with EpCAM-positive advanced solid tumors. BMC Cancer. 13:20 10.1186/1471-2407-13-20 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cote-Sierra J., Foucras G., Guo L., Chiodetti L., Young H.A., Hu-Li J., Zhu J., and Paul W.E.. 2004. Interleukin 2 plays a central role in Th2 differentiation. Proc. Natl. Acad. Sci. USA. 101:3880–3885. 10.1073/pnas.0400339101 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Courtney A.H., Lo W.L., and Weiss A.. 2018. TCR signaling: Mechanisms of initiation and propagation. Trends Biochem. Sci. 43:108–123. 10.1016/j.tibs.2017.11.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cully M. 2017. IL-2 focus switches to stimulating Tregs. Nat. Rev. Drug Discov. 16:595 10.1038/nrd.2017.171 [DOI] [PubMed] [Google Scholar]
- D’Cruz L.M., and Klein L.. 2005. Development and function of agonist-induced CD25+Foxp3+ regulatory T cells in the absence of interleukin 2 signaling. Nat. Immunol. 6:1152–1159. 10.1038/ni1264 [DOI] [PubMed] [Google Scholar]
- Danielli R., Patuzzo R., Di Giacomo A.M., Gallino G., Maurichi A., Di Florio A., Cutaia O., Lazzeri A., Fazio C., Miracco C., et al. . 2015. Intralesional administration of L19-IL2/L19-TNF in stage III or stage IVM1a melanoma patients: Results of a phase II study. Cancer Immunol. Immunother. 64:999–1009. 10.1007/s00262-015-1704-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dansokho C., Ait Ahmed D., Aid S., Toly-Ndour C., Chaigneau T., Calle V., Cagnard N., Holzenberger M., Piaggio E., Aucouturier P., et al. . 2016. Regulatory T cells delay disease progression in Alzheimer-like pathology. Brain. 139:1237–1251. 10.1093/brain/awv408 [DOI] [PubMed] [Google Scholar]
- Dinh T.N., Kyaw T.S., Kanellakis P., To K., Tipping P., Toh B.H., Bobik A., and Agrotis A.. 2012. Cytokine therapy with interleukin-2/anti-interleukin-2 monoclonal antibody complexes expands CD4+CD25+Foxp3+ regulatory T cells and attenuates development and progression of atherosclerosis. Circulation. 126:1256–1266. 10.1161/CIRCULATIONAHA.112.099044 [DOI] [PubMed] [Google Scholar]
- Eigentler T.K., Weide B., de Braud F., Spitaleri G., Romanini A., Pflugfelder A., González-Iglesias R., Tasciotti A., Giovannoni L., Schwager K., et al. . 2011. A dose-escalation and signal-generating study of the immunocytokine L19-IL2 in combination with dacarbazine for the therapy of patients with metastatic melanoma. Clin. Cancer Res. 17:7732–7742. 10.1158/1078-0432.CCR-11-1203 [DOI] [PubMed] [Google Scholar]
- Epstein A.L., Mizokami M.M., Li J., Hu P., and Khawli L.A.. 2003. Identification of a protein fragment of interleukin 2 responsible for vasopermeability. J. Natl. Cancer Inst. 95:741–749. 10.1093/jnci/95.10.741 [DOI] [PubMed] [Google Scholar]
- Flynn M.J., and Hartley J.A.. 2017. The emerging role of anti-CD25 directed therapies as both immune modulators and targeted agents in cancer. Br. J. Haematol. 179:20–35. 10.1111/bjh.14770 [DOI] [PubMed] [Google Scholar]
- Fontenot J.D., Gavin M.A., and Rudensky A.Y.. 2003. Foxp3 programs the development and function of CD4+CD25+ regulatory T cells. Nat. Immunol. 4:330–336. 10.1038/ni904 [DOI] [PubMed] [Google Scholar]
- Fontenot J.D., Rasmussen J.P., Gavin M.A., and Rudensky A.Y.. 2005. A function for interleukin 2 in Foxp3-expressing regulatory T cells. Nat. Immunol. 6:1142–1151. 10.1038/ni1263 [DOI] [PubMed] [Google Scholar]
- Fyfe G., Fisher R.I., Rosenberg S.A., Sznol M., Parkinson D.R., and Louie A.C.. 1995. Results of treatment of 255 patients with metastatic renal cell carcinoma who received high-dose recombinant interleukin-2 therapy. J. Clin. Oncol. 13:688–696. 10.1200/JCO.1995.13.3.688 [DOI] [PubMed] [Google Scholar]
- Fyfe G.A., Fisher R.I., Rosenberg S.A., Sznol M., Parkinson D.R., and Louie A.C.. 1996. Long-term response data for 255 patients with metastatic renal cell carcinoma treated with high-dose recombinant interleukin-2 therapy. J. Clin. Oncol. 14:2410–2411. 10.1200/JCO.1996.14.8.2410 [DOI] [PubMed] [Google Scholar]
- Geertsen P.F., Gore M.E., Negrier S., Tourani J.M., and von der Maase H.. 2004. Safety and efficacy of subcutaneous and continuous intravenous infusion rIL-2 in patients with metastatic renal cell carcinoma. Br. J. Cancer. 90:1156–1162. 10.1038/sj.bjc.6601709 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gillessen S., Gnad-Vogt U.S., Gallerani E., Beck J., Sessa C., Omlin A., Mattiacci M.R., Liedert B., Kramer D., Laurent J., et al. . 2013. A phase I dose-escalation study of the immunocytokine EMD 521873 (Selectikine) in patients with advanced solid tumours. Eur. J. Cancer. 49:35–44. 10.1016/j.ejca.2012.07.015 [DOI] [PubMed] [Google Scholar]
- Goldstein J.D., Pérol L., Zaragoza B., Baeyens A., Marodon G., and Piaggio E.. 2013. Role of cytokines in thymus- versus peripherally derived-regulatory T cell differentiation and function. Front. Immunol. 4:155 10.3389/fimmu.2013.00155 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gong D., and Malek T.R.. 2007. Cytokine-dependent Blimp-1 expression in activated T cells inhibits IL-2 production. J. Immunol. 178:242–252. 10.4049/jimmunol.178.1.242 [DOI] [PubMed] [Google Scholar]
- González F.B., Villar S.R., Fernández Bussy R., Martin G.H., Pérol L., Manarin R., Spinelli S.V., Pilon C., Cohen J.L., Bottasso O.A., et al. . 2015. Immunoendocrine dysbalance during uncontrolled T. cruzi infection is associated with the acquisition of a Th-1-like phenotype by Foxp3+ T cells. Brain Behav. Immun. 45:219–232. 10.1016/j.bbi.2014.11.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gonzalo J.A., González-García A., Martínez C., and Kroemer G.. 1993. Glucocorticoid-mediated control of the activation and clonal deletion of peripheral T cells in vivo. J. Exp. Med. 177:1239–1246. 10.1084/jem.177.5.1239 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goudy K., Aydin D., Barzaghi F., Gambineri E., Vignoli M., Ciullini Mannurita S., Doglioni C., Ponzoni M., Cicalese M.P., Assanelli A., et al. . 2013. Human IL2RA null mutation mediates immunodeficiency with lymphoproliferation and autoimmunity. Clin. Immunol. 146:248–261. 10.1016/j.clim.2013.01.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grange M., Verdeil G., Arnoux F., Griffon A., Spicuglia S., Maurizio J., Buferne M., Schmitt-Verhulst A.M., and Auphan-Anezin N.. 2013. Active STAT5 regulates T-bet and eomesodermin expression in CD8 T cells and imprints a T-bet-dependent Tc1 program with repressed IL-6/TGF-β1 signaling. J. Immunol. 191:3712–3724. 10.4049/jimmunol.1300319 [DOI] [PubMed] [Google Scholar]
- Grinberg-Bleyer Y., Baeyens A., You S., Elhage R., Fourcade G., Gregoire S., Cagnard N., Carpentier W., Tang Q., Bluestone J., et al. . 2010. IL-2 reverses established type 1 diabetes in NOD mice by a local effect on pancreatic regulatory T cells. J. Exp. Med. 207:1871–1878. 10.1084/jem.20100209 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hartemann A., Bensimon G., Payan C.A., Jacqueminet S., Bourron O., Nicolas N., Fonfrede M., Rosenzwajg M., Bernard C., and Klatzmann D.. 2013. Low-dose interleukin 2 in patients with type 1 diabetes: A phase 1/2 randomised, double-blind, placebo-controlled trial. Lancet Diabetes Endocrinol. 1:295–305. 10.1016/S2213-8587(13)70113-X [DOI] [PubMed] [Google Scholar]
- Haskins K., Kubo R., White J., Pigeon M., Kappler J., and Marrack P.. 1983. The major histocompatibility complex-restricted antigen receptor on T cells. I. Isolation with a monoclonal antibody. J. Exp. Med. 157:1149–1169. 10.1084/jem.157.4.1149 [DOI] [PMC free article] [PubMed] [Google Scholar]
- He J., Zhang X., Wei Y., Sun X., Chen Y., Deng J., Jin Y., Gan Y., Hu X., Jia R., et al. . 2016. Low-dose interleukin-2 treatment selectively modulates CD4+ T cell subsets in patients with systemic lupus erythematosus. Nat. Med. 22:991–993. 10.1038/nm.4148 [DOI] [PubMed] [Google Scholar]
- Herr F., Lemoine R., Gouilleux F., Meley D., Kazma I., Heraud A., Velge-Roussel F., Baron C., and Lebranchu Y.. 2014. IL-2 phosphorylates STAT5 to drive IFN-γ production and activation of human dendritic cells. J. Immunol. 192:5660–5670. 10.4049/jimmunol.1300422 [DOI] [PubMed] [Google Scholar]
- Hershko A.Y., Suzuki R., Charles N., Alvarez-Errico D., Sargent J.L., Laurence A., and Rivera J.. 2011. Mast cell interleukin-2 production contributes to suppression of chronic allergic dermatitis. Immunity. 35:562–571. 10.1016/j.immuni.2011.07.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Humrich J.Y., and Riemekasten G.. 2016. Restoring regulation—IL-2 therapy in systemic lupus erythematosus. Expert Rev. Clin. Immunol. 12:1153–1160. 10.1080/1744666X.2016.1199957 [DOI] [PubMed] [Google Scholar]
- Huse M. 2009. The T-cell-receptor signaling network. J. Cell Sci. 122:1269–1273. 10.1242/jcs.042762 [DOI] [PubMed] [Google Scholar]
- Jain J., Loh C., and Rao A.. 1995. Transcriptional regulation of the IL-2 gene. Curr. Opin. Immunol. 7:333–342. 10.1016/0952-7915(95)80107-3 [DOI] [PubMed] [Google Scholar]
- Jindal R., Unadkat J., Zhang W., Zhang D., Ng T.W., Wang Y., Jiang J., Lakkis F., Rubin P., Lee W.P., et al. . 2015. Spontaneous resolution of acute rejection and tolerance induction with IL-2 fusion protein in vascularized composite allotransplantation. Am. J. Transplant. 15:1231–1240. 10.1111/ajt.13118 [DOI] [PubMed] [Google Scholar]
- Johannsen M., Spitaleri G., Curigliano G., Roigas J., Weikert S., Kempkensteffen C., Roemer A., Kloeters C., Rogalla P., Pecher G., et al. . 2010. The tumour-targeting human L19-IL2 immunocytokine: Preclinical safety studies, phase I clinical trial in patients with solid tumours and expansion into patients with advanced renal cell carcinoma. Eur. J. Cancer. 46:2926–2935. 10.1016/j.ejca.2010.07.033 [DOI] [PubMed] [Google Scholar]
- Johnston R.J., Poholek A.C., DiToro D., Yusuf I., Eto D., Barnett B., Dent A.L., Craft J., and Crotty S.. 2009. Bcl6 and Blimp-1 are reciprocal and antagonistic regulators of T follicular helper cell differentiation. Science. 325:1006–1010. 10.1126/science.1175870 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalia V., Sarkar S., Subramaniam S., Haining W.N., Smith K.A., and Ahmed R.. 2010. Prolonged interleukin-2Rα expression on virus-specific CD8+ T cells favors terminal-effector differentiation in vivo. Immunity. 32:91–103. 10.1016/j.immuni.2009.11.010 [DOI] [PubMed] [Google Scholar]
- Kanai T., Seki S., Jenks J.A., Kohli A., Kawli T., Martin D.P., Snyder M., Bacchetta R., and Nadeau K.C.. 2014. Identification of STAT5A and STAT5B target genes in human T cells. PLoS One. 9:e86790 10.1371/journal.pone.0086790 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kennedy-Nasser A.A., Ku S., Castillo-Caro P., Hazrat Y., Wu M.F., Liu H., Melenhorst J., Barrett A.J., Ito S., Foster A., et al. . 2014. Ultra low-dose IL-2 for GVHD prophylaxis after allogeneic hematopoietic stem cell transplantation mediates expansion of regulatory T cells without diminishing antiviral and antileukemic activity. Clin. Cancer Res. 20:2215–2225. 10.1158/1078-0432.CCR-13-3205 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kitashima D.Y., Kobayashi T., Woodring T., Idouchi K., Doebel T., Voisin B., Adachi T., Ouchi T., Takahashi H., Nishifuji K., et al. . 2018. Langerhans cells prevent autoimmunity via expansion of keratinocyte antigen-specific regulatory T cells. EBioMedicine. 27:293–303. 10.1016/j.ebiom.2017.12.022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klatzmann D., and Abbas A.K.. 2015. The promise of low-dose interleukin-2 therapy for autoimmune and inflammatory diseases. Nat. Rev. Immunol. 15:283–294. 10.1038/nri3823 [DOI] [PubMed] [Google Scholar]
- Klein C., Waldhauer I., Nicolini V.G., Freimoser-Grundschober A., Nayak T., Vugts D.J., Dunn C., Bolijn M., Benz J., Stihle M., et al. . 2017. Cergutuzumab amunaleukin (CEA-IL2v), a CEA-targeted IL-2 variant-based immunocytokine for combination cancer immunotherapy: Overcoming limitations of aldesleukin and conventional IL-2-based immunocytokines. OncoImmunology. 6:e1277306 10.1080/2162402X.2016.1277306 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kmieciak M., Gowda M., Graham L., Godder K., Bear H.D., Marincola F.M., and Manjili M.H.. 2009. Human T cells express CD25 and Foxp3 upon activation and exhibit effector/memory phenotypes without any regulatory/suppressor function. J. Transl. Med. 7:89 10.1186/1479-5876-7-89 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knosp C.A., Schiering C., Spence S., Carroll H.P., Nel H.J., Osbourn M., Jackson R., Lyubomska O., Malissen B., Ingram R., et al. . 2013. Regulation of Foxp3+ inducible regulatory T cell stability by SOCS2. J. Immunol. 190:3235–3245. 10.4049/jimmunol.1201396 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Konrad M.W., Hemstreet G., Hersh E.M., Mansell P.W., Mertelsmann R., Kolitz J.E., and Bradley E.C.. 1990. Pharmacokinetics of recombinant interleukin 2 in humans. Cancer Res. 50:2009–2017. [PubMed] [Google Scholar]
- Koreth J., Matsuoka K., Kim H.T., McDonough S.M., Bindra B., Alyea E.P. III, Armand P., Cutler C., Ho V.T., Treister N.S., et al. . 2011. Interleukin-2 and regulatory T cells in graft-versus-host disease. N. Engl. J. Med. 365:2055–2066. 10.1056/NEJMoa1108188 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koreth J., Kim H.T., Jones K.T., Lange P.B., Reynolds C.G., Chammas M.J., Dusenbury K., Whangbo J., Nikiforow S., Alyea E.P. III, et al. . 2016. Efficacy, durability, and response predictors of low-dose interleukin-2 therapy for chronic graft-versus-host disease. Blood. 128:130–137. 10.1182/blood-2016-02-702852 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kovanen P.E., Young L., Al-Shami A., Rovella V., Pise-Masison C.A., Radonovich M.F., Powell J., Fu J., Brady J.N., Munson P.J., et al. . 2005. Global analysis of IL-2 target genes: Identification of chromosomal clusters of expressed genes. Int. Immunol. 17:1009–1021. 10.1093/intimm/dxh283 [DOI] [PubMed] [Google Scholar]
- Krieg C., Létourneau S., Pantaleo G., and Boyman O.. 2010. Improved IL-2 immunotherapy by selective stimulation of IL-2 receptors on lymphocytes and endothelial cells. Proc. Natl. Acad. Sci. USA. 107:11906–11911. 10.1073/pnas.1002569107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krömer G., Sundick R.S., Schauenstein K., Hála K., and Wick G.. 1985. Analysis of lymphocytes infiltrating the thyroid gland of Obese strain chickens. J. Immunol. 135:2452–2457. [PubMed] [Google Scholar]
- Krouse R.S., Royal R.E., Heywood G., Weintraub B.D., White D.E., Steinberg S.M., Rosenberg S.A., and Schwartzentruber D.J.. 1995. Thyroid dysfunction in 281 patients with metastatic melanoma or renal carcinoma treated with interleukin-2 alone. J. Immunother. Emphasis Tumor Immunol. 18:272–278. 10.1097/00002371-199511000-00008 [DOI] [PubMed] [Google Scholar]
- Kündig T.M., Schorle H., Bachmann M.F., Hengartner H., Zinkernagel R.M., and Horak I.. 1993. Immune responses in interleukin-2-deficient mice. Science. 262:1059–1061. 10.1126/science.8235625 [DOI] [PubMed] [Google Scholar]
- Kuribayashi K., Gillis S., Kern D.E., and Henney C.S.. 1981. Murine NK cell cultures: Effects of interleukin-2 and interferon on cell growth and cytotoxic reactivity. J. Immunol. 126:2321–2327. [PubMed] [Google Scholar]
- Laurence A., Tato C.M., Davidson T.S., Kanno Y., Chen Z., Yao Z., Blank R.B., Meylan F., Siegel R., Hennighausen L., et al. . 2007. Interleukin-2 signaling via STAT5 constrains T helper 17 cell generation. Immunity. 26:371–381. 10.1016/j.immuni.2007.02.009 [DOI] [PubMed] [Google Scholar]
- Laurent J., Touvrey C., Gillessen S., Joffraud M., Vicari M., Bertrand C., Ongarello S., Liedert B., Gallerani E., Beck J., et al. . 2013. T-cell activation by treatment of cancer patients with EMD 521873 (Selectikine), an IL-2/anti-DNA fusion protein. J. Transl. Med. 11:5 10.1186/1479-5876-11-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Le Gros G., Ben-Sasson S.Z., Seder R., Finkelman F.D., and Paul W.E.. 2008. Generation of interleukin 4 (IL-4)-producing cells in vivo and in vitro: IL-2 and IL-4 are required for in vitro generation of IL-4-producing cells. J. Immunol. 181:2943–2951. [PubMed] [Google Scholar]
- Létourneau S., van Leeuwen E.M., Krieg C., Martin C., Pantaleo G., Sprent J., Surh C.D., and Boyman O.. 2010. IL-2/anti-IL-2 antibody complexes show strong biological activity by avoiding interaction with IL-2 receptor α subunit CD25. Proc. Natl. Acad. Sci. USA. 107:2171–2176. 10.1073/pnas.0909384107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levin A.M., Bates D.L., Ring A.M., Krieg C., Lin J.T., Su L., Moraga I., Raeber M.E., Bowman G.R., Novick P., et al. . 2012. Exploiting a natural conformational switch to engineer an interleukin-2 ‘superkine’. Nature. 484:529–533. 10.1038/nature10975 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liao W., Schones D.E., Oh J., Cui Y., Cui K., Roh T.Y., Zhao K., and Leonard W.J.. 2008. Priming for T helper type 2 differentiation by interleukin 2-mediated induction of interleukin 4 receptor α-chain expression. Nat. Immunol. 9:1288–1296. 10.1038/ni.1656 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liao W., Lin J.X., Wang L., Li P., and Leonard W.J.. 2011. Modulation of cytokine receptors by IL-2 broadly regulates differentiation into helper T cell lineages. Nat. Immunol. 12:551–559. 10.1038/ni.2030 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin X., Chen M., Liu Y., Guo Z., He X., Brand D., and Zheng S.G.. 2013. Advances in distinguishing natural from induced Foxp3+ regulatory T cells. Int. J. Clin. Exp. Pathol. 6:116–123. [PMC free article] [PubMed] [Google Scholar]
- Liu D.V., Maier L.M., Hafler D.A., and Wittrup K.D.. 2009. Engineered interleukin-2 antagonists for the inhibition of regulatory T cells. J. Immunother. 32:887–894. 10.1097/CJI.0b013e3181b528da [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lo M., Kim H.S., Tong R.K., Bainbridge T.W., Vernes J.M., Zhang Y., Lin Y.L., Chung S., Dennis M.S., Zuchero Y.J., et al. . 2017. Effector-attenuating substitutions that maintain antibody stability and reduce toxicity in mice. J. Biol. Chem. 292:3900–3908. 10.1074/jbc.M116.767749 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Long S.A., Rieck M., Sanda S., Bollyky J.B., Samuels P.L., Goland R., Ahmann A., Rabinovitch A., Aggarwal S., Phippard D., et al. . 2012. Rapamycin/IL-2 combination therapy in patients with type 1 diabetes augments Tregs yet transiently impairs β-cell function. Diabetes. 61:2340–2348. 10.2337/db12-0049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malek T.R. 2008. The biology of interleukin-2. Annu. Rev. Immunol. 26:453–479. 10.1146/annurev.immunol.26.021607.090357 [DOI] [PubMed] [Google Scholar]
- Malek T.R., Porter B.O., Codias E.K., Scibelli P., and Yu A.. 2000. Normal lymphoid homeostasis and lack of lethal autoimmunity in mice containing mature T cells with severely impaired IL-2 receptors. J. Immunol. 164:2905–2914. 10.4049/jimmunol.164.6.2905 [DOI] [PubMed] [Google Scholar]
- Malek T.R., Yu A., Vincek V., Scibelli P., and Kong L.. 2002. CD4 regulatory T cells prevent lethal autoimmunity in IL-2Rβ-deficient mice. Implications for the nonredundant function of IL-2. Immunity. 17:167–178. 10.1016/S1074-7613(02)00367-9 [DOI] [PubMed] [Google Scholar]
- Manjunath N., Shankar P., Wan J., Weninger W., Crowley M.A., Hieshima K., Springer T.A., Fan X., Shen H., Lieberman J., et al. . 2001. Effector differentiation is not prerequisite for generation of memory cytotoxic T lymphocytes. J. Clin. Invest. 108:871–878. 10.1172/JCI13296 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Margolin K., Atkins M.B., Dutcher J.P., Ernstoff M.S., Smith J.W. II, Clark J.I., Baar J., Sosman J., Weber J., Lathia C., et al. . 2007. Phase I trial of BAY 50-4798, an interleukin-2-specific agonist in advanced melanoma and renal cancer. Clin. Cancer Res. 13:3312–3319. 10.1158/1078-0432.CCR-06-1341 [DOI] [PubMed] [Google Scholar]
- Marin-Acevedo J.A., Soyano A.E., Dholaria B., Knutson K.L., and Lou Y.. 2018. Cancer immunotherapy beyond immune checkpoint inhibitors. J. Hematol. Oncol. 11:8 10.1186/s13045-017-0552-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsuoka K., Koreth J., Kim H.T., Bascug G., McDonough S., Kawano Y., Murase K., Cutler C., Ho V.T., Alyea E.P., et al. . 2013. Low-dose interleukin-2 therapy restores regulatory T cell homeostasis in patients with chronic graft-versus-host disease. Sci. Transl. Med. 5:179ra43 10.1126/scitranslmed.3005265 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Millington T., Koulmanda M., Ng C., Boskovic S., Nadazdin O.M., Benichou G., Zheng X.X., Strom T.B., and Madsen J.C.. 2012. Effects of an agonist interleukin-2/Fc fusion protein, a mutant antagonist interleukin-15/Fc fusion protein, and sirolimus on cardiac allograft survival in non-human primates. J. Heart Lung Transplant. 31:427–435. 10.1016/j.healun.2012.01.864 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitra S., Ring A.M., Amarnath S., Spangler J.B., Li P., Ju W., Fischer S., Oh J., Spolski R., Weiskopf K., et al. . 2015. Interleukin-2 activity can be fine tuned with engineered receptor signaling clamps. Immunity. 42:826–838. 10.1016/j.immuni.2015.04.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moraes-Vasconcelos D., Costa-Carvalho B.T., Torgerson T.R., and Ochs H.D.. 2008. Primary immune deficiency disorders presenting as autoimmune diseases: IPEX and APECED. J. Clin. Immunol. 28(Suppl 1):11–19. 10.1007/s10875-008-9176-5 [DOI] [PubMed] [Google Scholar]
- Morgan D.A., Ruscetti F.W., and Gallo R.. 1976. Selective in vitro growth of T lymphocytes from normal human bone marrows. Science. 193:1007–1008. 10.1126/science.181845 [DOI] [PubMed] [Google Scholar]
- Ogilvie R.L., Abelson M., Hau H.H., Vlasova I., Blackshear P.J., and Bohjanen P.R.. 2005. Tristetraprolin down-regulates IL-2 gene expression through AU-rich element-mediated mRNA decay. J. Immunol. 174:953–961. 10.4049/jimmunol.174.2.953 [DOI] [PubMed] [Google Scholar]
- Onwumeh J., Okwundu C.I., and Kredo T.. 2017. Interleukin-2 as an adjunct to antiretroviral therapy for HIV-positive adults. Cochrane Database Syst. Rev. 5:CD009818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palmer P.A.J., Atzpodien J., Philip T., Negrier S., Kirchner H., Von der Maase H., Geertsen P., Evers P., Loriaux E., Oskam R., et al. . 1993. A comparison of 2 modes of administration of recombinant interleukin-2: Continuous intravenous infusion alone versus subcutaneous administration plus interferon alpha in patients with advanced renal cell carcinoma. Cancer Biother. 8:123–136. 10.1089/cbr.1993.8.123 [DOI] [PubMed] [Google Scholar]
- Pandiyan P., Conti H.R., Zheng L., Peterson A.C., Mathern D.R., Hernández-Santos N., Edgerton M., Gaffen S.L., and Lenardo M.J.. 2011. CD4+CD25+Foxp3+ regulatory T cells promote Th17 cells in vitro and enhance host resistance in mouse Candida albicans Th17 cell infection model. Immunity. 34:422–434. 10.1016/j.immuni.2011.03.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pei Y., Zhu P., Dang Y., Wu J., Yang X., Wan B., Liu J.O., Yi Q., and Yu L.. 2008. Nuclear export of NF90 to stabilize IL-2 mRNA is mediated by AKT-dependent phosphorylation at Ser647 in response to CD28 costimulation. J. Immunol. 180:222–229. 10.4049/jimmunol.180.1.222 [DOI] [PubMed] [Google Scholar]
- Pérol L., and Piaggio E.. 2016. New molecular and cellular mechanisms of tolerance: Tolerogenic actions of IL-2. Methods Mol. Biol. 1371:11–28. 10.1007/978-1-4939-3139-2_2 [DOI] [PubMed] [Google Scholar]
- Peterson L.B., Bell C.J.M., Howlett S.K., Pekalski M.L., Brady K., Hinton H., Sauter D., Todd J.A., Umana P., Ast O., et al. . 2018. A long-lived IL-2 mutein that selectively activates and expands regulatory T cells as a therapy for autoimmune disease. J. Autoimmun. 95:1–14. 10.1016/j.jaut.2018.10.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pipkin M.E., Sacks J.A., Cruz-Guilloty F., Lichtenheld M.G., Bevan M.J., and Rao A.. 2010. Interleukin-2 and inflammation induce distinct transcriptional programs that promote the differentiation of effector cytolytic T cells. Immunity. 32:79–90. 10.1016/j.immuni.2009.11.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poiesz B.J., Ruscetti F.W., Gazdar A.F., Bunn P.A., Minna J.D., and Gallo R.C.. 1980. Detection and isolation of type C retrovirus particles from fresh and cultured lymphocytes of a patient with cutaneous T-cell lymphoma. Proc. Natl. Acad. Sci. USA. 77:7415–7419. 10.1073/pnas.77.12.7415 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Radny P., Caroli U.M., Bauer J., Paul T., Schlegel C., Eigentler T.K., Weide B., Schwarz M., and Garbe C.. 2003. Phase II trial of intralesional therapy with interleukin-2 in soft-tissue melanoma metastases. Br. J. Cancer. 89:1620–1626. 10.1038/sj.bjc.6601320 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rao B.M., Girvin A.T., Ciardelli T., Lauffenburger D.A., and Wittrup K.D.. 2003. Interleukin-2 mutants with enhanced α-receptor subunit binding affinity. Protein Eng. 16:1081–1087. 10.1093/protein/gzg111 [DOI] [PubMed] [Google Scholar]
- Rao B.M., Driver I., Lauffenburger D.A., and Wittrup K.D.. 2005. High-affinity CD25-binding IL-2 mutants potently stimulate persistent T cell growth. Biochemistry. 44:10696–10701. 10.1021/bi050436x [DOI] [PubMed] [Google Scholar]
- Ravaud A., Delva R., Gomez F., Chevreau C., Douillard J.Y., Peny J., Coudert B., Négrier S., and Groupe Français d’Immunothérapie. 2002. Subcutaneous interleukin-2 and interferon α in the treatment of patients with metastatic renal cell carcinoma—Less efficacy compared with intravenous interleukin-2 and interferon α. Results of a multicenter Phase II trial from the Groupe Français d’Immunothérapie. Cancer. 95:2324–2330. 10.1002/cncr.10968 [DOI] [PubMed] [Google Scholar]
- Reem G.H., and Yeh N.H.. 1984. Interleukin 2 regulates expression of its receptor and synthesis of gamma interferon by human T lymphocytes. Science. 225:429–430. 10.1126/science.6429853 [DOI] [PubMed] [Google Scholar]
- Robb R.J., and Smith K.A.. 1981. Heterogeneity of human T-cell growth factor(s) due to variable glycosylation. Mol. Immunol. 18:1087–1094. 10.1016/0161-5890(81)90024-9 [DOI] [PubMed] [Google Scholar]
- Robb R.J., Munck A., and Smith K.A.. 1981. T cell growth factor receptors. Quantitation, specificity, and biological relevance. J. Exp. Med. 154:1455–1474. 10.1084/jem.154.5.1455 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosenberg S.A., and Restifo N.P.. 2015. Adoptive cell transfer as personalized immunotherapy for human cancer. Science. 348:62–68. 10.1126/science.aaa4967 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ross S.H., and Cantrell D.A.. 2018. Signaling and function of interleukin-2 in T lymphocytes. Annu. Rev. Immunol. 36:411–433. 10.1146/annurev-immunol-042617-053352 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruggiero A., Villa C.H., Bander E., Rey D.A., Bergkvist M., Batt C.A., Manova-Todorova K., Deen W.M., Scheinberg D.A., and McDevitt M.R.. 2010. Paradoxical glomerular filtration of carbon nanotubes. Proc. Natl. Acad. Sci. USA. 107:12369–12374. 10.1073/pnas.0913667107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saadoun D., Rosenzwajg M., Joly F., Six A., Carrat F., Thibault V., Sene D., Cacoub P., and Klatzmann D.. 2011. Regulatory T-cell responses to low-dose interleukin-2 in HCV-induced vasculitis. N. Engl. J. Med. 365:2067–2077. 10.1056/NEJMoa1105143 [DOI] [PubMed] [Google Scholar]
- Sadlack B., Merz H., Schorle H., Schimpl A., Feller A.C., and Horak I.. 1993. Ulcerative colitis-like disease in mice with a disrupted interleukin-2 gene. Cell. 75:253–261. 10.1016/0092-8674(93)80067-O [DOI] [PubMed] [Google Scholar]
- Sakaguchi S., Sakaguchi N., Asano M., Itoh M., and Toda M.. 1995. Immunologic self-tolerance maintained by activated T cells expressing IL-2 receptor α-chains (CD25). Breakdown of a single mechanism of self-tolerance causes various autoimmune diseases. J. Immunol. 155:1151–1164. [PubMed] [Google Scholar]
- Serfling E., Avots A., and Neumann M.. 1995. The architecture of the interleukin-2 promoter: A reflection of T lymphocyte activation. Biochim. Biophys. Acta. 1263:181–200. 10.1016/0167-4781(95)00112-T [DOI] [PubMed] [Google Scholar]
- Shi M., Lin T.H., Appell K.C., and Berg L.J.. 2008. Janus-kinase-3-dependent signals induce chromatin remodeling at the Ifng locus during T helper 1 cell differentiation. Immunity. 28:763–773. 10.1016/j.immuni.2008.04.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silva D.A., Yu S., Ulge U.Y., Spangler J.B., Jude K.M., Labão-Almeida C., Ali L.R., Quijano-Rubio A., Ruterbusch M., Leung I., et al. . 2019. De novo design of potent and selective mimics of IL-2 and IL-15. Nature. 565:186–191. 10.1038/s41586-018-0830-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smaldini P.L., Trejo F., Cohen J.L., Piaggio E., and Docena G.H.. 2018. Systemic IL-2/anti-IL-2Ab complex combined with sublingual immunotherapy suppresses experimental food allergy in mice through induction of mucosal regulatory T cells. Allergy. 73:885–895. 10.1111/all.13402 [DOI] [PubMed] [Google Scholar]
- Smith K.A., Favata M.F., and Oroszlan S.. 1983. Production and characterization of monoclonal antibodies to human interleukin 2: Strategy and tactics. J. Immunol. 131:1808–1815. [PubMed] [Google Scholar]
- Snow J.W., Abraham N., Ma M.C., Herndier B.G., Pastuszak A.W., and Goldsmith M.A.. 2003. Loss of tolerance and autoimmunity affecting multiple organs in STAT5A/5B-deficient mice. J. Immunol. 171:5042–5050. 10.4049/jimmunol.171.10.5042 [DOI] [PubMed] [Google Scholar]
- Sockolosky J.T., Trotta E., Parisi G., Picton L., Su L.L., Le A.C., Chhabra A., Silveria S.L., George B.M., King I.C., et al. . 2018. Selective targeting of engineered T cells using orthogonal IL-2 cytokine-receptor complexes. Science. 359:1037–1042. 10.1126/science.aar3246 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spangler J.B., Tomala J., Luca V.C., Jude K.M., Dong S., Ring A.M., Votavova P., Pepper M., Kovar M., and Garcia K.C.. 2015. Antibodies to interleukin-2 elicit selective T cell subset potentiation through distinct conformational mechanisms. Immunity. 42:815–825. 10.1016/j.immuni.2015.04.015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spolski R., Li P., and Leonard W.J.. 2018. Biology and regulation of IL-2: From molecular mechanisms to human therapy. Nat. Rev. Immunol. 18:648–659. 10.1038/s41577-018-0046-y [DOI] [PubMed] [Google Scholar]
- Suzuki H., Kündig T.M., Furlonger C., Wakeham A., Timms E., Matsuyama T., Schmits R., Simard J.J., Ohashi P.S., Griesser H., et al. . 1995. Deregulated T cell activation and autoimmunity in mice lacking interleukin-2 receptor beta. Science. 268:1472–1476. 10.1126/science.7770771 [DOI] [PubMed] [Google Scholar]
- Tang A., and Harding F.. 2019. The challenges and molecular approaches surrounding interleukin-2-based therapeutics in cancer. Cytokine: X. 1:100001 [Google Scholar]
- Tang Q., Adams J.Y., Penaranda C., Melli K., Piaggio E., Sgouroudis E., Piccirillo C.A., Salomon B.L., and Bluestone J.A.. 2008. Central role of defective interleukin-2 production in the triggering of islet autoimmune destruction. Immunity. 28:687–697. 10.1016/j.immuni.2008.03.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taniguchi T., Matsui H., Fujita T., Takaoka C., Kashima N., Yoshimoto R., and Hamuro J.. 1983. Structure and expression of a cloned cDNA for human interleukin-2. Nature. 302:305–310. 10.1038/302305a0 [DOI] [PubMed] [Google Scholar]
- Tchao N., Gorski K.S., Yuraszeck T., Sohn S.J., Ishida K., Wong H., and Park K.. 2017. Amg 592 is an investigational IL-2 mutein that induces highly selective expansion of regulatory T cells. Blood. 130Suppl 1:696.28798058 [Google Scholar]
- Todd J.A., Evangelou M., Cutler A.J., Pekalski M.L., Walker N.M., Stevens H.E., Porter L., Smyth D.J., Rainbow D.B., Ferreira R.C., et al. . 2016. Regulatory T cell responses in participants with type 1 diabetes after a single dose of interleukin-2: A non-randomised, open label, adaptive dose-finding trial. PLoS Med. 13:e1002139 10.1371/journal.pmed.1002139 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trotta E., Bessette P.H., Silveria S.L., Ely L.K., Jude K.M., Le D.T., Holst C.R., Coyle A., Potempa M., Lanier L.L., et al. . 2018. A human anti-IL-2 antibody that potentiates regulatory T cells by a structure-based mechanism. Nat. Med. 24:1005–1014. 10.1038/s41591-018-0070-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaishampayan U.N., Ernstoff M.S., Velcheti V., Hoimes C.J., Fishman M.N., Cho D.C., McDermott D.F., Kurman M.R., Alvarez J., Sun L., et al. . 2017. A phase I trial of ALKS 4230, an engineered cytokine activator of NK and effector T cells, in patients with advanced solid tumors. J. Clin. Oncol. 35(15_suppl):TPS3111 10.1200/JCO.2017.35.15_suppl.TPS3111 [DOI] [Google Scholar]
- Valle-Mendiola A., Gutiérrez-Hoya A., Lagunas-Cruz M.C., Weiss-Steider B., and Soto-Cruz I.. 2016. Pleiotropic effects of IL-2 on cancer: Its role in cervical cancer. Mediators Inflamm. 2016:2849523 10.1155/2016/2849523 [DOI] [PMC free article] [PubMed] [Google Scholar]
- van den Heuvel M.M., Verheij M., Boshuizen R., Belderbos J., Dingemans A.M., De Ruysscher D., Laurent J., Tighe R., Haanen J., and Quaratino S.. 2015. NHS-IL2 combined with radiotherapy: Preclinical rationale and phase Ib trial results in metastatic non-small cell lung cancer following first-line chemotherapy. J. Transl. Med. 13:32 10.1186/s12967-015-0397-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vazquez-Lombardi R., Loetsch C., Zinkl D., Jackson J., Schofield P., Deenick E.K., King C., Phan T.G., Webster K.E., Sprent J., et al. . 2017. Potent antitumour activity of interleukin-2-Fc fusion proteins requires Fc-mediated depletion of regulatory T-cells. Nat. Commun. 8:15373 10.1038/ncomms15373 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang X., Rickert M., and Garcia K.C.. 2005. Structure of the quaternary complex of interleukin-2 with its alpha, beta, and gammac receptors. Science. 310:1159–1163. 10.1126/science.1117893 [DOI] [PubMed] [Google Scholar]
- Ward N.C., Yu A., Moro A., Ban Y., Chen X., Hsiung S., Keegan J., Arbanas J.M., Loubeau M., Thankappan A., et al. . 2018. IL-2/CD25: A long-acting fusion protein that promotes immune tolerance by selectively targeting the IL-2 receptor on regulatory T cells. J. Immunol. 201:2579–2592. 10.4049/jimmunol.1800907 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Webster K.E., Walters S., Kohler R.E., Mrkvan T., Boyman O., Surh C.D., Grey S.T., and Sprent J.. 2009. In vivo expansion of T reg cells with IL-2-mAb complexes: Induction of resistance to EAE and long-term acceptance of islet allografts without immunosuppression. J. Exp. Med. 206:751–760. 10.1084/jem.20082824 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wildin R.S., Ramsdell F., Peake J., Faravelli F., Casanova J.L., Buist N., Levy-Lahad E., Mazzella M., Goulet O., Perroni L., et al. . 2001. X-linked neonatal diabetes mellitus, enteropathy and endocrinopathy syndrome is the human equivalent of mouse scurfy. Nat. Genet. 27:18–20. 10.1038/83707 [DOI] [PubMed] [Google Scholar]
- Willerford D.M., Chen J., Ferry J.A., Davidson L., Ma A., and Alt F.W.. 1995. Interleukin-2 receptor α chain regulates the size and content of the peripheral lymphoid compartment. Immunity. 3:521–530. 10.1016/1074-7613(95)90180-9 [DOI] [PubMed] [Google Scholar]
- Williams M.A., Tyznik A.J., and Bevan M.J.. 2006. Interleukin-2 signals during priming are required for secondary expansion of CD8+ memory T cells. Nature. 441:890–893. 10.1038/nature04790 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wojciechowski W., Harris D.P., Sprague F., Mousseau B., Makris M., Kusser K., Honjo T., Mohrs K., Mohrs M., Randall T., et al. . 2009. Cytokine-producing effector B cells regulate type 2 immunity to H. polygyrus. Immunity. 30:421–433. 10.1016/j.immuni.2009.01.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wuest S.C., Edwan J.H., Martin J.F., Han S., Perry J.S., Cartagena C.M., Matsuura E., Maric D., Waldmann T.A., and Bielekova B.. 2011. A role for interleukin-2 trans-presentation in dendritic cell-mediated T cell activation in humans, as revealed by daclizumab therapy. Nat. Med. 17:604–609. 10.1038/nm.2365 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang C., Huang S., Wang X., and Gu Y.. 2015. Emerging roles of CCCH-type zinc finger proteins in destabilizing mRNA encoding inflammatory factors and regulating immune responses. Crit. Rev. Eukaryot. Gene Expr. 25:77–89. 10.1615/CritRevEukaryotGeneExpr.2015013022 [DOI] [PubMed] [Google Scholar]
- Yang J.C., Sherry R.M., Steinberg S.M., Topalian S.L., Schwartzentruber D.J., Hwu P., Seipp C.A., Rogers-Freezer L., Morton K.E., White D.E., et al. . 2003. Randomized study of high-dose and low-dose interleukin-2 in patients with metastatic renal cancer. J. Clin. Oncol. 21:3127–3132. 10.1200/JCO.2003.02.122 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang X.P., Ghoreschi K., Steward-Tharp S.M., Rodriguez-Canales J., Zhu J., Grainger J.R., Hirahara K., Sun H.W., Wei L., Vahedi G., et al. . 2011. Opposing regulation of the locus encoding IL-17 through direct, reciprocal actions of STAT3 and STAT5. Nat. Immunol. 12:247–254. 10.1038/ni.1995 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yao Z., Kanno Y., Kerenyi M., Stephens G., Durant L., Watford W.T., Laurence A., Robinson G.W., Shevach E.M., Moriggl R., et al. . 2007. Nonredundant roles for Stat5a/b in directly regulating Foxp3. Blood. 109:4368–4375. 10.1182/blood-2006-11-055756 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ye C., Brand D., and Zheng S.G.. 2018. Targeting IL-2: An unexpected effect in treating immunological diseases. Signal Transduct. Target. Ther. 3:2 10.1038/s41392-017-0002-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu A., Zhu L., Altman N.H., and Malek T.R.. 2009. A low interleukin-2 receptor signaling threshold supports the development and homeostasis of T regulatory cells. Immunity. 30:204–217. 10.1016/j.immuni.2008.11.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zelante T., Fric J., Wong A.Y., and Ricciardi-Castagnoli P.. 2012. Interleukin-2 production by dendritic cells and its immuno-regulatory functions. Front. Immunol. 3:161 10.3389/fimmu.2012.00161 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng Y., and Rudensky A.Y.. 2007. Foxp3 in control of the regulatory T cell lineage. Nat. Immunol. 8:457–462. 10.1038/ni1455 [DOI] [PubMed] [Google Scholar]
- Zheng X.X., Steele A.W., Hancock W.W., Kawamoto K., Li X.C., Nickerson P.W., Li Y., Tian Y., and Strom T.B.. 1999. IL-2 receptor-targeted cytolytic IL-2/Fc fusion protein treatment blocks diabetogenic autoimmunity in nonobese diabetic mice. J. Immunol. 163:4041–4048. [PubMed] [Google Scholar]
- Zheng X.X., Sánchez-Fueyo A., Sho M., Domenig C., Sayegh M.H., and Strom T.B.. 2003. Favorably tipping the balance between cytopathic and regulatory T cells to create transplantation tolerance. Immunity. 19:503–514. 10.1016/S1074-7613(03)00259-0 [DOI] [PubMed] [Google Scholar]
- Zhou L., Chu C., Teng F., Bessman N.J., Goc J., Santosa E.K., Putzel G.G., Kabata H., Kelsen J.R., Baldassano R.N., et al. . 2019. Innate lymphoid cells support regulatory T cells in the intestine through interleukin-2. Nature. 568:405–409. 10.1038/s41586-019-1082-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu J., Cote-Sierra J., Guo L., and Paul W.E.. 2003. Stat5 activation plays a critical role in Th2 differentiation. Immunity. 19:739–748. 10.1016/S1074-7613(03)00292-9 [DOI] [PubMed] [Google Scholar]
- Zhu L., Kong Y., Zhang J., Claxton D.F., Ehmann W.C., Rybka W.B., Palmisiano N.D., Wang M., Jia B., Bayerl M., et al. . 2017. Blimp-1 impairs T cell function via upregulation of TIGIT and PD-1 in patients with acute myeloid leukemia. J. Hematol. Oncol. 10:124 10.1186/s13045-017-0486-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu P., Jiang W., Cao L., Yu W., Pei Y., Yang X., Wan B., Liu J.O., Yi Q., and Yu L.. 2010. IL-2 mRNA stabilization upon PMA stimulation is dependent on NF90-Ser647 phosphorylation by protein kinase CbetaI. J. Immunol. 185:5140–5149. 10.4049/jimmunol.1000849 [DOI] [PubMed] [Google Scholar]